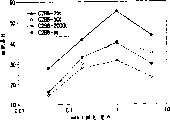CN1555411A - 抗体-依赖性细胞毒性增大的抗体糖基化变体 - Google Patents
抗体-依赖性细胞毒性增大的抗体糖基化变体 Download PDFInfo
- Publication number
- CN1555411A CN1555411A CNA028181735A CN02818173A CN1555411A CN 1555411 A CN1555411 A CN 1555411A CN A028181735 A CNA028181735 A CN A028181735A CN 02818173 A CN02818173 A CN 02818173A CN 1555411 A CN1555411 A CN 1555411A
- Authority
- CN
- China
- Prior art keywords
- antibody
- host cell
- cell
- human
- gntiii
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 title claims abstract description 67
- 230000013595 glycosylation Effects 0.000 title claims abstract description 59
- 238000006206 glycosylation reaction Methods 0.000 title claims abstract description 57
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 54
- 102000004169 proteins and genes Human genes 0.000 claims abstract description 23
- 230000001225 therapeutic effect Effects 0.000 claims abstract description 19
- 230000001976 improved effect Effects 0.000 claims abstract description 8
- 210000004027 cell Anatomy 0.000 claims description 266
- 150000002482 oligosaccharides Chemical class 0.000 claims description 105
- 229920001542 oligosaccharide Polymers 0.000 claims description 90
- 238000000034 method Methods 0.000 claims description 87
- 230000014509 gene expression Effects 0.000 claims description 63
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 43
- 229920001184 polypeptide Polymers 0.000 claims description 41
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 41
- 150000007523 nucleic acids Chemical class 0.000 claims description 38
- 238000002360 preparation method Methods 0.000 claims description 38
- 108020004707 nucleic acids Proteins 0.000 claims description 37
- 102000039446 nucleic acids Human genes 0.000 claims description 37
- 230000004927 fusion Effects 0.000 claims description 23
- 108060003951 Immunoglobulin Proteins 0.000 claims description 20
- 102000018358 immunoglobulin Human genes 0.000 claims description 20
- 239000000203 mixture Substances 0.000 claims description 18
- 238000011282 treatment Methods 0.000 claims description 18
- 102000008394 Immunoglobulin Fragments Human genes 0.000 claims description 17
- 108010021625 Immunoglobulin Fragments Proteins 0.000 claims description 17
- 206010028980 Neoplasm Diseases 0.000 claims description 17
- 206010035226 Plasma cell myeloma Diseases 0.000 claims description 16
- 241000699802 Cricetulus griseus Species 0.000 claims description 15
- 230000008859 change Effects 0.000 claims description 15
- 201000000050 myeloid neoplasm Diseases 0.000 claims description 15
- 210000001672 ovary Anatomy 0.000 claims description 15
- 230000001472 cytotoxic effect Effects 0.000 claims description 14
- 238000001890 transfection Methods 0.000 claims description 14
- 101100230376 Acetivibrio thermocellus (strain ATCC 27405 / DSM 1237 / JCM 9322 / NBRC 103400 / NCIMB 10682 / NRRL B-4536 / VPI 7372) celI gene Proteins 0.000 claims description 13
- 239000000427 antigen Substances 0.000 claims description 13
- 108091007433 antigens Proteins 0.000 claims description 13
- 102000036639 antigens Human genes 0.000 claims description 13
- 238000000816 matrix-assisted laser desorption--ionisation Methods 0.000 claims description 13
- 231100000433 cytotoxic Toxicity 0.000 claims description 12
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 12
- 239000012634 fragment Substances 0.000 claims description 12
- 201000011510 cancer Diseases 0.000 claims description 10
- 210000003719 b-lymphocyte Anatomy 0.000 claims description 9
- 201000010099 disease Diseases 0.000 claims description 9
- -1 N-acetylamino Chemical group 0.000 claims description 8
- 210000004408 hybridoma Anatomy 0.000 claims description 8
- 239000008194 pharmaceutical composition Substances 0.000 claims description 7
- 101001133056 Homo sapiens Mucin-1 Proteins 0.000 claims description 6
- 102100034256 Mucin-1 Human genes 0.000 claims description 6
- 241001529936 Murinae Species 0.000 claims description 6
- 210000000349 chromosome Anatomy 0.000 claims description 6
- 206010009944 Colon cancer Diseases 0.000 claims description 5
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 4
- 201000005202 lung cancer Diseases 0.000 claims description 4
- 208000020816 lung neoplasm Diseases 0.000 claims description 4
- 201000001441 melanoma Diseases 0.000 claims description 4
- 241001430294 unidentified retrovirus Species 0.000 claims description 4
- 208000001333 Colorectal Neoplasms Diseases 0.000 claims description 3
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 claims description 3
- 102000003781 Inhibitor of growth protein 1 Human genes 0.000 claims description 3
- 108090000191 Inhibitor of growth protein 1 Proteins 0.000 claims description 3
- 206010029260 Neuroblastoma Diseases 0.000 claims description 3
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 claims description 3
- 208000006265 Renal cell carcinoma Diseases 0.000 claims description 3
- 201000010989 colorectal carcinoma Diseases 0.000 claims description 3
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 3
- 102100031585 ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 Human genes 0.000 claims description 2
- 102100038080 B-cell receptor CD22 Human genes 0.000 claims description 2
- 102100024217 CAMPATH-1 antigen Human genes 0.000 claims description 2
- 229940124292 CD20 monoclonal antibody Drugs 0.000 claims description 2
- 101150013553 CD40 gene Proteins 0.000 claims description 2
- 108010065524 CD52 Antigen Proteins 0.000 claims description 2
- 101710132601 Capsid protein Proteins 0.000 claims description 2
- 101150029707 ERBB2 gene Proteins 0.000 claims description 2
- 108010066687 Epithelial Cell Adhesion Molecule Proteins 0.000 claims description 2
- 102000018651 Epithelial Cell Adhesion Molecule Human genes 0.000 claims description 2
- 102000016359 Fibronectins Human genes 0.000 claims description 2
- 108010067306 Fibronectins Proteins 0.000 claims description 2
- 102100041003 Glutamate carboxypeptidase 2 Human genes 0.000 claims description 2
- 102000006354 HLA-DR Antigens Human genes 0.000 claims description 2
- 108010058597 HLA-DR Antigens Proteins 0.000 claims description 2
- 101000777636 Homo sapiens ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 Proteins 0.000 claims description 2
- 101000884305 Homo sapiens B-cell receptor CD22 Proteins 0.000 claims description 2
- 101000892862 Homo sapiens Glutamate carboxypeptidase 2 Proteins 0.000 claims description 2
- 101000934338 Homo sapiens Myeloid cell surface antigen CD33 Proteins 0.000 claims description 2
- 101000738771 Homo sapiens Receptor-type tyrosine-protein phosphatase C Proteins 0.000 claims description 2
- 101000851376 Homo sapiens Tumor necrosis factor receptor superfamily member 8 Proteins 0.000 claims description 2
- 102100025243 Myeloid cell surface antigen CD33 Human genes 0.000 claims description 2
- 102100037422 Receptor-type tyrosine-protein phosphatase C Human genes 0.000 claims description 2
- 102100040245 Tumor necrosis factor receptor superfamily member 5 Human genes 0.000 claims description 2
- 102100036857 Tumor necrosis factor receptor superfamily member 8 Human genes 0.000 claims description 2
- 108010087914 epidermal growth factor receptor VIII Proteins 0.000 claims description 2
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 claims description 2
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 claims description 2
- 230000002068 genetic effect Effects 0.000 claims description 2
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical compound FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 claims description 2
- 229920001481 poly(stearyl methacrylate) Polymers 0.000 claims description 2
- 102100022019 Pregnancy-specific beta-1-glycoprotein 2 Human genes 0.000 claims 1
- 230000000694 effects Effects 0.000 description 46
- 239000000523 sample Substances 0.000 description 44
- 108020004999 messenger RNA Proteins 0.000 description 21
- 238000005516 engineering process Methods 0.000 description 20
- 238000012986 modification Methods 0.000 description 16
- 230000004048 modification Effects 0.000 description 14
- 102000003886 Glycoproteins Human genes 0.000 description 13
- 108090000288 Glycoproteins Proteins 0.000 description 13
- NWXMGUDVXFXRIG-WESIUVDSSA-N (4s,4as,5as,6s,12ar)-4-(dimethylamino)-1,6,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carboxamide Chemical compound C1=CC=C2[C@](O)(C)[C@H]3C[C@H]4[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]4(O)C(=O)C3=C(O)C2=C1O NWXMGUDVXFXRIG-WESIUVDSSA-N 0.000 description 12
- 108700008625 Reporter Genes Proteins 0.000 description 12
- 239000013604 expression vector Substances 0.000 description 12
- 230000003013 cytotoxicity Effects 0.000 description 11
- 231100000135 cytotoxicity Toxicity 0.000 description 11
- 230000006870 function Effects 0.000 description 11
- 230000009089 cytolysis Effects 0.000 description 10
- 102100022005 B-lymphocyte antigen CD20 Human genes 0.000 description 9
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 9
- 101000897405 Homo sapiens B-lymphocyte antigen CD20 Proteins 0.000 description 9
- 108020004684 Internal Ribosome Entry Sites Proteins 0.000 description 9
- 239000012636 effector Substances 0.000 description 9
- 238000011534 incubation Methods 0.000 description 9
- 230000001404 mediated effect Effects 0.000 description 9
- 238000004519 manufacturing process Methods 0.000 description 8
- 206010043554 thrombocytopenia Diseases 0.000 description 8
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 210000004369 blood Anatomy 0.000 description 7
- 239000008280 blood Substances 0.000 description 7
- 239000006143 cell culture medium Substances 0.000 description 7
- 230000007935 neutral effect Effects 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- 108020004414 DNA Proteins 0.000 description 6
- 102000004190 Enzymes Human genes 0.000 description 6
- 108090000790 Enzymes Proteins 0.000 description 6
- 241000238631 Hexapoda Species 0.000 description 6
- 108090001090 Lectins Proteins 0.000 description 6
- 102000004856 Lectins Human genes 0.000 description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 6
- 239000003814 drug Substances 0.000 description 6
- 229940088598 enzyme Drugs 0.000 description 6
- 108020001507 fusion proteins Proteins 0.000 description 6
- 102000037865 fusion proteins Human genes 0.000 description 6
- 230000012010 growth Effects 0.000 description 6
- 239000002523 lectin Substances 0.000 description 6
- 239000013612 plasmid Substances 0.000 description 6
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 5
- 201000009030 Carcinoma Diseases 0.000 description 5
- 108700023372 Glycosyltransferases Proteins 0.000 description 5
- 102000051366 Glycosyltransferases Human genes 0.000 description 5
- OVRNDRQMDRJTHS-UHFFFAOYSA-N N-acelyl-D-glucosamine Natural products CC(=O)NC1C(O)OC(CO)C(O)C1O OVRNDRQMDRJTHS-UHFFFAOYSA-N 0.000 description 5
- OVRNDRQMDRJTHS-RTRLPJTCSA-N N-acetyl-D-glucosamine Chemical compound CC(=O)N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-RTRLPJTCSA-N 0.000 description 5
- MBLBDJOUHNCFQT-LXGUWJNJSA-N N-acetylglucosamine Natural products CC(=O)N[C@@H](C=O)[C@@H](O)[C@H](O)[C@H](O)CO MBLBDJOUHNCFQT-LXGUWJNJSA-N 0.000 description 5
- 230000001684 chronic effect Effects 0.000 description 5
- 239000002131 composite material Substances 0.000 description 5
- 150000001875 compounds Chemical class 0.000 description 5
- 238000004520 electroporation Methods 0.000 description 5
- 238000011156 evaluation Methods 0.000 description 5
- 230000003993 interaction Effects 0.000 description 5
- 239000003550 marker Substances 0.000 description 5
- 230000002018 overexpression Effects 0.000 description 5
- 238000012545 processing Methods 0.000 description 5
- 210000002966 serum Anatomy 0.000 description 5
- 239000011734 sodium Substances 0.000 description 5
- 208000011580 syndromic disease Diseases 0.000 description 5
- 238000001262 western blot Methods 0.000 description 5
- 210000005253 yeast cell Anatomy 0.000 description 5
- 201000004624 Dermatitis Diseases 0.000 description 4
- 241000700605 Viruses Species 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 238000004113 cell culture Methods 0.000 description 4
- 239000003623 enhancer Substances 0.000 description 4
- 238000009396 hybridization Methods 0.000 description 4
- 230000001900 immune effect Effects 0.000 description 4
- 210000004698 lymphocyte Anatomy 0.000 description 4
- 239000002609 medium Substances 0.000 description 4
- 238000012207 quantitative assay Methods 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- 102000005962 receptors Human genes 0.000 description 4
- 230000009467 reduction Effects 0.000 description 4
- 229960004641 rituximab Drugs 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- 206010001052 Acute respiratory distress syndrome Diseases 0.000 description 3
- 102000004127 Cytokines Human genes 0.000 description 3
- 108090000695 Cytokines Proteins 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 239000012981 Hank's balanced salt solution Substances 0.000 description 3
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 3
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 206010037549 Purpura Diseases 0.000 description 3
- 241001672981 Purpura Species 0.000 description 3
- 241000723873 Tobacco mosaic virus Species 0.000 description 3
- 239000002671 adjuvant Substances 0.000 description 3
- 238000001042 affinity chromatography Methods 0.000 description 3
- 230000003321 amplification Effects 0.000 description 3
- 239000005557 antagonist Substances 0.000 description 3
- 238000013459 approach Methods 0.000 description 3
- 208000010668 atopic eczema Diseases 0.000 description 3
- 230000004071 biological effect Effects 0.000 description 3
- 238000005119 centrifugation Methods 0.000 description 3
- 238000010367 cloning Methods 0.000 description 3
- 239000002299 complementary DNA Substances 0.000 description 3
- 230000010013 cytotoxic mechanism Effects 0.000 description 3
- 238000004043 dyeing Methods 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 229930182830 galactose Natural products 0.000 description 3
- 238000010353 genetic engineering Methods 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- 230000036039 immunity Effects 0.000 description 3
- 238000010166 immunofluorescence Methods 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 210000004962 mammalian cell Anatomy 0.000 description 3
- 238000001819 mass spectrum Methods 0.000 description 3
- 239000011159 matrix material Substances 0.000 description 3
- 238000003199 nucleic acid amplification method Methods 0.000 description 3
- 230000037361 pathway Effects 0.000 description 3
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 3
- 210000000664 rectum Anatomy 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 229930182490 saponin Natural products 0.000 description 3
- 150000007949 saponins Chemical class 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 238000002560 therapeutic procedure Methods 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- DIGQNXIGRZPYDK-WKSCXVIASA-N (2R)-6-amino-2-[[2-[[(2S)-2-[[2-[[(2R)-2-[[(2S)-2-[[(2R,3S)-2-[[2-[[(2S)-2-[[2-[[(2S)-2-[[(2S)-2-[[(2R)-2-[[(2S,3S)-2-[[(2R)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[2-[[(2S)-2-[[(2R)-2-[[2-[[2-[[2-[(2-amino-1-hydroxyethylidene)amino]-3-carboxy-1-hydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1,5-dihydroxy-5-iminopentylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]hexanoic acid Chemical compound C[C@@H]([C@@H](C(=N[C@@H](CS)C(=N[C@@H](C)C(=N[C@@H](CO)C(=NCC(=N[C@@H](CCC(=N)O)C(=NC(CS)C(=N[C@H]([C@H](C)O)C(=N[C@H](CS)C(=N[C@H](CO)C(=NCC(=N[C@H](CS)C(=NCC(=N[C@H](CCCCN)C(=O)O)O)O)O)O)O)O)O)O)O)O)O)O)O)N=C([C@H](CS)N=C([C@H](CO)N=C([C@H](CO)N=C([C@H](C)N=C(CN=C([C@H](CO)N=C([C@H](CS)N=C(CN=C(C(CS)N=C(C(CC(=O)O)N=C(CN)O)O)O)O)O)O)O)O)O)O)O)O DIGQNXIGRZPYDK-WKSCXVIASA-N 0.000 description 2
- 208000031261 Acute myeloid leukaemia Diseases 0.000 description 2
- 208000026872 Addison Disease Diseases 0.000 description 2
- 240000006108 Allium ampeloprasum Species 0.000 description 2
- 235000005254 Allium ampeloprasum Nutrition 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- 108700031308 Antennapedia Homeodomain Proteins 0.000 description 2
- 208000023275 Autoimmune disease Diseases 0.000 description 2
- 241000208199 Buxus sempervirens Species 0.000 description 2
- 102100024423 Carbonic anhydrase 9 Human genes 0.000 description 2
- 102000011022 Chorionic Gonadotropin Human genes 0.000 description 2
- 108010062540 Chorionic Gonadotropin Proteins 0.000 description 2
- 108091026890 Coding region Proteins 0.000 description 2
- 241000186216 Corynebacterium Species 0.000 description 2
- 241000699800 Cricetinae Species 0.000 description 2
- SHZGCJCMOBCMKK-UHFFFAOYSA-N D-mannomethylose Natural products CC1OC(O)C(O)C(O)C1O SHZGCJCMOBCMKK-UHFFFAOYSA-N 0.000 description 2
- 101150074155 DHFR gene Proteins 0.000 description 2
- 102000003951 Erythropoietin Human genes 0.000 description 2
- 108090000394 Erythropoietin Proteins 0.000 description 2
- 241000206602 Eukaryota Species 0.000 description 2
- PNNNRSAQSRJVSB-SLPGGIOYSA-N Fucose Natural products C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C=O PNNNRSAQSRJVSB-SLPGGIOYSA-N 0.000 description 2
- 206010018364 Glomerulonephritis Diseases 0.000 description 2
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 2
- 101100165850 Homo sapiens CA9 gene Proteins 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 206010061218 Inflammation Diseases 0.000 description 2
- 102100026720 Interferon beta Human genes 0.000 description 2
- 108090000467 Interferon-beta Proteins 0.000 description 2
- SHZGCJCMOBCMKK-DHVFOXMCSA-N L-fucopyranose Chemical compound C[C@@H]1OC(O)[C@@H](O)[C@H](O)[C@@H]1O SHZGCJCMOBCMKK-DHVFOXMCSA-N 0.000 description 2
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 2
- 102000003792 Metallothionein Human genes 0.000 description 2
- 108090000157 Metallothionein Proteins 0.000 description 2
- 208000033776 Myeloid Acute Leukemia Diseases 0.000 description 2
- 208000015914 Non-Hodgkin lymphomas Diseases 0.000 description 2
- 108020004711 Nucleic Acid Probes Proteins 0.000 description 2
- 102000052812 Ornithine decarboxylases Human genes 0.000 description 2
- 108700005126 Ornithine decarboxylases Proteins 0.000 description 2
- 206010033128 Ovarian cancer Diseases 0.000 description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 description 2
- 206010060862 Prostate cancer Diseases 0.000 description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 2
- 208000013616 Respiratory Distress Syndrome Diseases 0.000 description 2
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 2
- 239000004098 Tetracycline Substances 0.000 description 2
- 102000006601 Thymidine Kinase Human genes 0.000 description 2
- 108020004440 Thymidine kinase Proteins 0.000 description 2
- 102000003978 Tissue Plasminogen Activator Human genes 0.000 description 2
- 108090000373 Tissue Plasminogen Activator Proteins 0.000 description 2
- 206010067584 Type 1 diabetes mellitus Diseases 0.000 description 2
- 108010084455 Zeocin Proteins 0.000 description 2
- 238000009825 accumulation Methods 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 230000000172 allergic effect Effects 0.000 description 2
- 238000011091 antibody purification Methods 0.000 description 2
- SQVRNKJHWKZAKO-UHFFFAOYSA-N beta-N-Acetyl-D-neuraminic acid Natural products CC(=O)NC1C(O)CC(O)(C(O)=O)OC1C(O)C(O)CO SQVRNKJHWKZAKO-UHFFFAOYSA-N 0.000 description 2
- 230000003570 biosynthesizing effect Effects 0.000 description 2
- 230000006696 biosynthetic metabolic pathway Effects 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 210000003169 central nervous system Anatomy 0.000 description 2
- 208000025302 chronic primary adrenal insufficiency Diseases 0.000 description 2
- 239000012228 culture supernatant Substances 0.000 description 2
- 210000004748 cultured cell Anatomy 0.000 description 2
- 238000012258 culturing Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 201000001981 dermatomyositis Diseases 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- VLCYCQAOQCDTCN-UHFFFAOYSA-N eflornithine Chemical compound NCCCC(N)(C(F)F)C(O)=O VLCYCQAOQCDTCN-UHFFFAOYSA-N 0.000 description 2
- 206010014599 encephalitis Diseases 0.000 description 2
- 229940105423 erythropoietin Drugs 0.000 description 2
- 208000021045 exocrine pancreatic carcinoma Diseases 0.000 description 2
- 239000012997 ficoll-paque Substances 0.000 description 2
- 230000004907 flux Effects 0.000 description 2
- 238000001415 gene therapy Methods 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 208000007475 hemolytic anemia Diseases 0.000 description 2
- 229920000669 heparin Polymers 0.000 description 2
- 229940022353 herceptin Drugs 0.000 description 2
- 229940084986 human chorionic gonadotropin Drugs 0.000 description 2
- 238000003018 immunoassay Methods 0.000 description 2
- 239000007943 implant Substances 0.000 description 2
- 230000008676 import Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 230000004054 inflammatory process Effects 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- 210000003292 kidney cell Anatomy 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 239000003068 molecular probe Substances 0.000 description 2
- 150000002772 monosaccharides Chemical group 0.000 description 2
- 229950006780 n-acetylglucosamine Drugs 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 239000002853 nucleic acid probe Substances 0.000 description 2
- CWCMIVBLVUHDHK-ZSNHEYEWSA-N phleomycin D1 Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC[C@@H](N=1)C=1SC=C(N=1)C(=O)NCCCCNC(N)=N)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C CWCMIVBLVUHDHK-ZSNHEYEWSA-N 0.000 description 2
- 208000005987 polymyositis Diseases 0.000 description 2
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 2
- 229940002612 prodrug Drugs 0.000 description 2
- 239000000651 prodrug Substances 0.000 description 2
- RXWNCPJZOCPEPQ-NVWDDTSBSA-N puromycin Chemical compound C1=CC(OC)=CC=C1C[C@H](N)C(=O)N[C@H]1[C@@H](O)[C@H](N2C3=NC=NC(=C3N=C2)N(C)C)O[C@@H]1CO RXWNCPJZOCPEPQ-NVWDDTSBSA-N 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 229940061969 rheumatrex Drugs 0.000 description 2
- 238000009666 routine test Methods 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 230000002269 spontaneous effect Effects 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 235000019364 tetracycline Nutrition 0.000 description 2
- 150000003522 tetracyclines Chemical class 0.000 description 2
- 229960000187 tissue plasminogen activator Drugs 0.000 description 2
- 229960005267 tositumomab Drugs 0.000 description 2
- 230000002103 transcriptional effect Effects 0.000 description 2
- 230000009261 transgenic effect Effects 0.000 description 2
- 229960000575 trastuzumab Drugs 0.000 description 2
- 241000701447 unidentified baculovirus Species 0.000 description 2
- 239000013598 vector Substances 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 230000003442 weekly effect Effects 0.000 description 2
- ASWBNKHCZGQVJV-UHFFFAOYSA-N (3-hexadecanoyloxy-2-hydroxypropyl) 2-(trimethylazaniumyl)ethyl phosphate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(O)COP([O-])(=O)OCC[N+](C)(C)C ASWBNKHCZGQVJV-UHFFFAOYSA-N 0.000 description 1
- UFBJCMHMOXMLKC-UHFFFAOYSA-N 2,4-dinitrophenol Chemical compound OC1=CC=C([N+]([O-])=O)C=C1[N+]([O-])=O UFBJCMHMOXMLKC-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 101150094949 APRT gene Proteins 0.000 description 1
- 108010024223 Adenine phosphoribosyltransferase Proteins 0.000 description 1
- 102100029457 Adenine phosphoribosyltransferase Human genes 0.000 description 1
- 101000689231 Aeromonas salmonicida S-layer protein Proteins 0.000 description 1
- 206010027654 Allergic conditions Diseases 0.000 description 1
- 206010001889 Alveolitis Diseases 0.000 description 1
- 206010002556 Ankylosing Spondylitis Diseases 0.000 description 1
- 208000003343 Antiphospholipid Syndrome Diseases 0.000 description 1
- 208000031104 Arterial Occlusive disease Diseases 0.000 description 1
- 108010002913 Asialoglycoproteins Proteins 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 102100025475 Carcinoembryonic antigen-related cell adhesion molecule 5 Human genes 0.000 description 1
- 241000701489 Cauliflower mosaic virus Species 0.000 description 1
- 206010008909 Chronic Hepatitis Diseases 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- 206010012438 Dermatitis atopic Diseases 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 208000002087 Endarteritis Diseases 0.000 description 1
- 208000004232 Enteritis Diseases 0.000 description 1
- 206010015218 Erythema multiforme Diseases 0.000 description 1
- 206010015226 Erythema nodosum Diseases 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- 208000007465 Giant cell arteritis Diseases 0.000 description 1
- 102000000340 Glucosyltransferases Human genes 0.000 description 1
- 108010055629 Glucosyltransferases Proteins 0.000 description 1
- 208000003807 Graves Disease Diseases 0.000 description 1
- 208000015023 Graves' disease Diseases 0.000 description 1
- 208000030836 Hashimoto thyroiditis Diseases 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- 208000035186 Hemolytic Autoimmune Anemia Diseases 0.000 description 1
- 201000004331 Henoch-Schoenlein purpura Diseases 0.000 description 1
- 206010019617 Henoch-Schonlein purpura Diseases 0.000 description 1
- 206010019755 Hepatitis chronic active Diseases 0.000 description 1
- 101000787903 Homo sapiens Transmembrane protein 200C Proteins 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010020850 Hyperthyroidism Diseases 0.000 description 1
- 208000031814 IgA Vasculitis Diseases 0.000 description 1
- 208000010159 IgA glomerulonephritis Diseases 0.000 description 1
- 206010021263 IgA nephropathy Diseases 0.000 description 1
- ZQISRDCJNBUVMM-UHFFFAOYSA-N L-Histidinol Natural products OCC(N)CC1=CN=CN1 ZQISRDCJNBUVMM-UHFFFAOYSA-N 0.000 description 1
- ZQISRDCJNBUVMM-YFKPBYRVSA-N L-histidinol Chemical compound OC[C@@H](N)CC1=CNC=N1 ZQISRDCJNBUVMM-YFKPBYRVSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- 201000010743 Lambert-Eaton myasthenic syndrome Diseases 0.000 description 1
- 208000005777 Lupus Nephritis Diseases 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 201000009906 Meningitis Diseases 0.000 description 1
- 206010027336 Menstruation delayed Diseases 0.000 description 1
- 101100261636 Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) trpB2 gene Proteins 0.000 description 1
- 208000034578 Multiple myelomas Diseases 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- 206010028424 Myasthenic syndrome Diseases 0.000 description 1
- SQVRNKJHWKZAKO-LUWBGTNYSA-N N-acetylneuraminic acid Chemical group CC(=O)N[C@@H]1[C@@H](O)CC(O)(C(O)=O)O[C@H]1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-LUWBGTNYSA-N 0.000 description 1
- 102000005348 Neuraminidase Human genes 0.000 description 1
- 108010006232 Neuraminidase Proteins 0.000 description 1
- 206010029240 Neuritis Diseases 0.000 description 1
- 206010029491 Nodular vasculitis Diseases 0.000 description 1
- 108091028043 Nucleic acid sequence Proteins 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- 229940122060 Ornithine decarboxylase inhibitor Drugs 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 241001524178 Paenarthrobacter ureafaciens Species 0.000 description 1
- 206010034277 Pemphigoid Diseases 0.000 description 1
- 241000721454 Pemphigus Species 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 102000057297 Pepsin A Human genes 0.000 description 1
- 108090000284 Pepsin A Proteins 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 208000031845 Pernicious anaemia Diseases 0.000 description 1
- 241000276498 Pollachius virens Species 0.000 description 1
- 206010065159 Polychondritis Diseases 0.000 description 1
- 208000025237 Polyendocrinopathy Diseases 0.000 description 1
- 206010036105 Polyneuropathy Diseases 0.000 description 1
- NPYPAHLBTDXSSS-UHFFFAOYSA-N Potassium ion Chemical compound [K+] NPYPAHLBTDXSSS-UHFFFAOYSA-N 0.000 description 1
- 102100025067 Potassium voltage-gated channel subfamily H member 4 Human genes 0.000 description 1
- 101710163352 Potassium voltage-gated channel subfamily H member 4 Proteins 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 206010037660 Pyrexia Diseases 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 208000035977 Rare disease Diseases 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 208000033464 Reiter syndrome Diseases 0.000 description 1
- 206010039710 Scleroderma Diseases 0.000 description 1
- 208000034189 Sclerosis Diseases 0.000 description 1
- 241000270295 Serpentes Species 0.000 description 1
- 241000700584 Simplexvirus Species 0.000 description 1
- 206010041067 Small cell lung cancer Diseases 0.000 description 1
- 201000002661 Spondylitis Diseases 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- 101000748795 Thermus thermophilus (strain ATCC 27634 / DSM 579 / HB8) Cytochrome c oxidase polypeptide I+III Proteins 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 102100025939 Transmembrane protein 200C Human genes 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- 239000006035 Tryptophane Substances 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 206010046851 Uveitis Diseases 0.000 description 1
- 241000700618 Vaccinia virus Species 0.000 description 1
- LPQOADBMXVRBNX-UHFFFAOYSA-N ac1ldcw0 Chemical compound Cl.C1CN(C)CCN1C1=C(F)C=C2C(=O)C(C(O)=O)=CN3CCSC1=C32 LPQOADBMXVRBNX-UHFFFAOYSA-N 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000012082 adaptor molecule Substances 0.000 description 1
- 238000009098 adjuvant therapy Methods 0.000 description 1
- 201000000028 adult respiratory distress syndrome Diseases 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 229910021502 aluminium hydroxide Inorganic materials 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 229940126575 aminoglycoside Drugs 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- 208000007502 anemia Diseases 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000001857 anti-mycotic effect Effects 0.000 description 1
- 230000001905 anti-neuroblastoma Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 230000009227 antibody-mediated cytotoxicity Effects 0.000 description 1
- 239000002543 antimycotic Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 208000021328 arterial occlusion Diseases 0.000 description 1
- 206010003230 arteritis Diseases 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- 230000001363 autoimmune Effects 0.000 description 1
- 201000000448 autoimmune hemolytic anemia Diseases 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 210000000941 bile Anatomy 0.000 description 1
- 230000000975 bioactive effect Effects 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000031018 biological processes and functions Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 208000002352 blister Diseases 0.000 description 1
- 210000001772 blood platelet Anatomy 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000002798 bone marrow cell Anatomy 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 244000309466 calf Species 0.000 description 1
- 229940112129 campath Drugs 0.000 description 1
- 238000002619 cancer immunotherapy Methods 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 238000005277 cation exchange chromatography Methods 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000006037 cell lysis Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 238000002659 cell therapy Methods 0.000 description 1
- 230000005889 cellular cytotoxicity Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000011097 chromatography purification Methods 0.000 description 1
- 208000019425 cirrhosis of liver Diseases 0.000 description 1
- 210000003109 clavicle Anatomy 0.000 description 1
- 239000000701 coagulant Substances 0.000 description 1
- 206010009887 colitis Diseases 0.000 description 1
- 210000001072 colon Anatomy 0.000 description 1
- 239000013068 control sample Substances 0.000 description 1
- 230000001276 controlling effect Effects 0.000 description 1
- 238000012937 correction Methods 0.000 description 1
- 201000003278 cryoglobulinemia Diseases 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 238000013016 damping Methods 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 229960001776 edrecolomab Drugs 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 238000013100 final test Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 238000013467 fragmentation Methods 0.000 description 1
- 238000006062 fragmentation reaction Methods 0.000 description 1
- 230000033581 fucosylation Effects 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 208000005017 glioblastoma Diseases 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 125000003147 glycosyl group Chemical group 0.000 description 1
- 201000003911 head and neck carcinoma Diseases 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- YQYJSBFKSSDGFO-FWAVGLHBSA-N hygromycin A Chemical compound O[C@H]1[C@H](O)[C@H](C(=O)C)O[C@@H]1Oc1ccc(\C=C(/C)C(=O)N[C@@H]2[C@@H]([C@H]3OCO[C@H]3[C@@H](O)[C@@H]2O)O)cc1O YQYJSBFKSSDGFO-FWAVGLHBSA-N 0.000 description 1
- 230000008076 immune mechanism Effects 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 208000015446 immunoglobulin a vasculitis Diseases 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 230000002637 immunotoxin Effects 0.000 description 1
- 229940051026 immunotoxin Drugs 0.000 description 1
- 239000002596 immunotoxin Substances 0.000 description 1
- 231100000608 immunotoxin Toxicity 0.000 description 1
- 210000003000 inclusion body Anatomy 0.000 description 1
- 150000002475 indoles Chemical class 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 230000028709 inflammatory response Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 238000009413 insulation Methods 0.000 description 1
- 229960001388 interferon-beta Drugs 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 230000000366 juvenile effect Effects 0.000 description 1
- 108010045069 keyhole-limpet hemocyanin Proteins 0.000 description 1
- 229950000518 labetuzumab Drugs 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 206010025135 lupus erythematosus Diseases 0.000 description 1
- 210000003563 lymphoid tissue Anatomy 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 238000012269 metabolic engineering Methods 0.000 description 1
- HPNSFSBZBAHARI-UHFFFAOYSA-N micophenolic acid Natural products OC1=C(CC=C(C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-UHFFFAOYSA-N 0.000 description 1
- 238000005497 microtitration Methods 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- ZAHQPTJLOCWVPG-UHFFFAOYSA-N mitoxantrone dihydrochloride Chemical compound Cl.Cl.O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO ZAHQPTJLOCWVPG-UHFFFAOYSA-N 0.000 description 1
- 229950003063 mitumomab Drugs 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 208000037890 multiple organ injury Diseases 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 231100000350 mutagenesis Toxicity 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 231100000219 mutagenic Toxicity 0.000 description 1
- 230000003505 mutagenic effect Effects 0.000 description 1
- 206010028417 myasthenia gravis Diseases 0.000 description 1
- 229960000951 mycophenolic acid Drugs 0.000 description 1
- HPNSFSBZBAHARI-RUDMXATFSA-N mycophenolic acid Chemical compound OC1=C(C\C=C(/C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-RUDMXATFSA-N 0.000 description 1
- 201000008383 nephritis Diseases 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 210000004940 nucleus Anatomy 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- 238000011275 oncology therapy Methods 0.000 description 1
- 239000002818 ornithine decarboxylase inhibitor Substances 0.000 description 1
- 230000007918 pathogenicity Effects 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 229940111202 pepsin Drugs 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000000505 pernicious effect Effects 0.000 description 1
- 238000009522 phase III clinical trial Methods 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 230000008488 polyadenylation Effects 0.000 description 1
- 229920000447 polyanionic polymer Polymers 0.000 description 1
- 201000006292 polyarteritis nodosa Diseases 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 230000007824 polyneuropathy Effects 0.000 description 1
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 1
- 229910001414 potassium ion Inorganic materials 0.000 description 1
- 239000012716 precipitator Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 208000008128 pulmonary tuberculosis Diseases 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 229950010131 puromycin Drugs 0.000 description 1
- 238000004445 quantitative analysis Methods 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 208000002574 reactive arthritis Diseases 0.000 description 1
- 238000003259 recombinant expression Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 208000020615 rectal carcinoma Diseases 0.000 description 1
- 230000008521 reorganization Effects 0.000 description 1
- 238000009256 replacement therapy Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 201000000306 sarcoidosis Diseases 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 239000006152 selective media Substances 0.000 description 1
- 238000013207 serial dilution Methods 0.000 description 1
- 239000012679 serum free medium Substances 0.000 description 1
- 125000005629 sialic acid group Chemical group 0.000 description 1
- 208000000587 small cell lung carcinoma Diseases 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- 230000010473 stable expression Effects 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 206010043207 temporal arteritis Diseases 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 229940040944 tetracyclines Drugs 0.000 description 1
- 229940126622 therapeutic monoclonal antibody Drugs 0.000 description 1
- 206010043778 thyroiditis Diseases 0.000 description 1
- 208000005057 thyrotoxicosis Diseases 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000010474 transient expression Effects 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 101150081616 trpB gene Proteins 0.000 description 1
- 101150111232 trpB-1 gene Proteins 0.000 description 1
- 229960004799 tryptophan Drugs 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 230000029812 viral genome replication Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2887—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against CD20
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K49/00—Preparations for testing in vivo
- A61K49/06—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations
- A61K49/08—Nuclear magnetic resonance [NMR] contrast preparations; Magnetic resonance imaging [MRI] contrast preparations characterised by the carrier
- A61K49/10—Organic compounds
- A61K49/14—Peptides, e.g. proteins
- A61K49/16—Antibodies; Immunoglobulins; Fragments thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2896—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against molecules with a "CD"-designation, not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
- C07K16/3038—Kidney, bladder
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/10—Transferases (2.)
- C12N9/1048—Glycosyltransferases (2.4)
- C12N9/1051—Hexosyltransferases (2.4.1)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P21/00—Preparation of peptides or proteins
- C12P21/005—Glycopeptides, glycoproteins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/10—Immunoglobulins specific features characterized by their source of isolation or production
- C07K2317/14—Specific host cells or culture conditions, e.g. components, pH or temperature
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/40—Immunoglobulins specific features characterized by post-translational modification
- C07K2317/41—Glycosylation, sialylation, or fucosylation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
- C07K2317/732—Antibody-dependent cellular cytotoxicity [ADCC]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/52—Improvements relating to the production of bulk chemicals using catalysts, e.g. selective catalysts
Abstract
本发明涉及蛋白质糖基化工程领域。更具体地说,本发明涉及糖基化工程产生具有提高的治疗性能的蛋白质,包括具有提高的抗体-依赖性细胞毒性的抗体。
Description
发明领域
本发明涉及蛋白质糖基化工程领域。更具体地说,本发明涉及生产治疗性能得到改进的蛋白质,包括抗体-依赖性细胞毒性增大的抗体的糖基化工程。
背景技术
糖蛋白介导人、其它真核生物和一些原核生物的许多重要功能,包括催化作用,发出信号,细胞-细胞转染和分子识别和缔合。它们构成真核生物中非胞质蛋白的大部分(Lis等,Eur.J.Biochem.218:1-27(1993))。已经开发了很多用于治疗目的的糖蛋白,近二十年来,天然存在的重组型的分泌型糖蛋白已经是生物技术产业的主要产物。例子包括红细胞生成素(EPO),治疗性单克隆抗体(治疗性mAb),组织纤溶酶原激活剂(tPA),干扰素-β,(IFN-β),粒细胞-巨噬细胞集落刺激因子(GM-CSF),和人绒毛膜促性腺素(hCG)(Cumming等,Glycobiology1:115-130(1991))。
寡糖成分能显著影响与治疗性糖蛋白效力相关的性质,包括物理稳定性,对蛋白酶作用的抗性,与免疫系统的相互作用,药物动力学和特异生物活性。这样的性质可能不仅仅取决于存在或不存在寡糖,而且还取决于寡糖的具体结构。可以进行寡糖结构与糖蛋白功能之间的一些总结。例如,一些寡糖结构通过与特异性碳水化合物结合蛋白质的相互作用而介导血流中糖蛋白的快速清除,而其它的能被抗体结合并且引起不希望的免疫反应(Jenkins等,Nature Biotechnol.14:975-81(1996))。
哺乳动物细胞由于它们能将蛋白质糖基化成最适于人应用的形式,所以成为生产治疗性糖蛋白的优选宿主(Gumming等,Glycobiology 1:115-30(1991);Jenkins等,Nature Biotechnol.14:975-81(1996))。细菌很少将蛋白质糖基化,而且就象酵母,丝状真菌,昆虫和植物细胞这样的其它类型的普通宿主一样,得到与从血流中快速清除,不期望的免疫相互作用,和在某些特殊情况下,降低的生物活性相关的糖基化模式。近二十年中,在哺乳动物细胞中,最常使用中国仓鼠卵巢(CHO)细胞。除了给出合适的糖基化模式之外,这些细胞持续产生遗传上稳定的,高产率的无性繁殖细胞系。在简单的生物反应器中使用无血清培养基能将它们培养至高密度,使得发展安全而可重复的生物加工过程成为可能。其它常用动物细胞包括幼仓鼠肾(BHK)细胞,NSO-和SP2/O-小鼠骨髓瘤细胞。最近,也试验了由转基因动物进行生产(Jenkins等,Nature Biotechnol.14:975-81(1996))。
所有的抗体在重链恒定区中的保守位置含有糖结构,每个同种型具有不同的N-连接糖结构排列方式,它们不定地影响蛋白质组装,分泌或功能活性(Wright,A.和Morrison,S.L.,Trends Biotech.15:26-32(1997))。被连接的N-键接糖的结构变化很大,这取决于处理程度,并且可包括高-甘露糖,多支链的以及双触角的复合寡糖(Wright,A.,和Morrison,S.L.,Trends Biotech.15:26-32(1997))。典型地,在特定的糖基化位点存在着对连接的核心寡糖结构的多种不同加工以致于即使单克隆抗体也以多糖基化形式存在。同样,已经证明抗体糖基化作用中的主要差异出现在细胞系之间,甚至不同培养条件下培养的特定细胞系也可以见到十分细微的差别(Lifely,M.R.等,Glycobiology 5(8):813-22(1995))。
由美国食品与药物管理局批准用于治疗CD20阳性B-细胞,低级或囊状非霍奇金淋巴瘤的利妥希玛(Rituxan TM;IDECPharmaceuticals,San Diego,CA,and Genentech Inc.,SanFrancisco,CA)和用于治疗晚期乳房癌(Grillo-Lopez,A.-J.,等,Semin.Oncol.26:66-73(1999);Goldenberg,M.M.,Clin.Ther.21:309-18(1999))的Trastuzumab(HerceptinTM;Genentech Inc,)证明,未缀合的单克隆抗体(mAb)能成为有用的治疗癌症的药物。这些产品的成功不仅仅依赖它们的药效,而且还依赖它们出色的安全性(Grillo-Lopez,A.-J.,等,Semin.Oncol.26:66-73(1999);Goldenberg,M.M.,Clill.Ther.21:309-18(1999))。虽然完成了这两种药物,最近人们在获得比通过未缀合的mAb治疗通常提供的特异性抗体活性更高的特异性抗体活性方面有很大兴趣。
效力获得大幅提高并且保留简单的生产过程并且潜在避免了明显的不期望的副作用的一条途径是通过对它们的寡糖成分进行技术处理来提高mAb的天然的,由细胞介导的效应子功能(Umana,P.等,NatureBiotechnol.17:176-180(1999))。IgGI型抗体,癌症免疫治疗中最常用的抗体,是在各个CH2结构域中的Asn297处具有保守的N-连接糖基化位点的糖蛋白。CH2结构域之间有与Asn297连接的两个复合双触角寡糖,与多肽主链形成充分接触,而且它们的存在对于抗体介导效应子功能例如抗体依赖性细胞毒性(ADCC)是必需的(Lifely,M.R.等,Glycobiology 5:813-822(1995);Jefferis,R.等,Immunol Rev.163:59-76(1998);Wright,A.和Morrison,S.L.,Trends Biotechnol.15:26-32(1997))。
本发明人先前证明中国仓鼠卵巢(CHO)细胞过量表达β(1,4)-N-乙酰基氨基葡糖基转移酶III(GnTIII),一种催化等分寡糖的形成的糖基转移酶,显著提高工程CHO细胞产生的抗成神经细胞瘤嵌合单克隆抗体(chCE7)的体外ADCC活性(参见Umana,P.等,NatureBiotechnol.17:176-180(1999),国际公开No.WO 99/54342,其全部内容在此全文引作参考)。抗体chCE7属于一大类未缀合的mAb,它们具有高肿瘤亲和性和特异性,但是当在没有GnTIII酶的常用工业细胞系中产生时其效力太小以致于不能进行临床使用(Umana,P.,等,Nature BiotechnoL 17:176-180(1999))。该项研究首次证明通过将恒定区(Fc)-结合的等分寡糖的比例增大到高于天然存在抗体中的水平能大幅度提高最大体外的ADCC活性。为了测定该项发现是否能外推到未缀合的mAb,它在没有等分寡糖存在下已经具有显著的ADCC活性,本发明人已经将该项技术应用于利妥希玛,抗-CD20,IDEC-C2B8嵌合抗体。本发明人还同样将该项技术应用于未缀合的抗-癌mAbchG250。
本发明概述
本发明人现在利用遗传工程处理的以四环素调节方式过量表达N-乙酰基氨基葡糖基转移酶III(GnTIII;EC 2.1.4.144)的产生mAb的细胞系制备了新的抗-CD20单克隆抗体(mAb)IDEC-C2B8(利妥希玛)和抗-癌mAb chG250的糖基化变体。等分寡糖的合成需要GnTIII,发现它在天然存在的人抗体中以低水平至中等水平存在,但是在常规工业细胞系中产生的mAb中则不存在。新的糖基化变型在生物(ADCC)活性方面超过了MabtheraTM(欧洲市场出售的Rixtuximab版本)和小鼠-骨髓瘤衍生的chG250。例如,达到与MabtheraTM一样的最大ADCC活性需要低10倍量的具有最高水平的等分寡糖的变体。对于chG250,具有最高水平的等分寡糖变体在比检测未改构的对照chG250的低ADCC活性所需浓度低125-倍的浓度下介导显著ADCC活性。发现GnTIII表达水平与ADCC活性之间存在明确的关系。
因此,本发明的一方面涉及通过表达至少一种编码β(1,4)-N-乙酰基氨基葡糖基转移酶III(GnTIII)的核酸而产生Fc-介导的细胞毒性提高的多肽的工程宿主细胞,其中由宿主细胞产生的多肽选自完整抗体分子,抗体片段,和包括与免疫球蛋白的Fc区等价的区域的融合蛋白,并且其中以足以提高Fc区中带有等分杂合寡糖或半乳糖基化复合寡糖或其混合物的所述多肽相对于Fc区中带有等分复合体寡糖的多肽的比例的量表达GnTIII。
在优选的实施方案中,所述多肽是IgG或其片段,最优选地是IgGl或其片段。在另一个优选的实施方案中,所述多肽是包括与人IgG的Fc区等价的区域的融合蛋白。
在本发明的另一个方面中,将含有至少一个编码GnTIII的基因的核酸分子导入宿主细胞。在优选的实施方案中,将至少一个编码GnTIII的基因导入宿主细胞的染色体。
或者,对宿主细胞进行基因工程处理致使内源GnTIII基因被激活,例如,通过将提高基因表达的DNA元件插入宿主染色体。在优选的实施方案中,通过将启动子,一个增强子,一个转录因子结合位点,转座子,或逆转录病毒元件或其组合插入到宿主细胞染色体中来将内源性GnTIII激活。另一方面,选择带有内源性GnTIII的突变引发表达的宿主细胞。优选地,宿主细胞是CHO细胞突变体lec10。
在本发明另一个优选的实施方案中,编码GnTIII的至少一种核酸与组成型启动子元件操作性连接。
在另一个优选的实施方案中,宿主细胞是CHO细胞,BHK细胞,NSO细胞,SP2/0细胞,或杂交瘤细胞,Y0骨髓瘤细胞,P3X63小鼠骨髓瘤细胞,PER细胞或PER。C6细胞和所述多肽是抗CD20抗体。在另一个优选的实施方案中,宿主细胞是SP2/0细胞并且多肽是单克隆抗体chG250。
另一方面,本发明涉及宿主细胞,该宿主细胞进一步含有编码抗体分子,抗体片段,或者包括与免疫球蛋白的Fc区等价的区域的融合蛋白的至少一种转染的核酸。在优选的实施方案中,宿主细胞含有编码下面蛋白的至少一种转染的核酸:抗CD20抗体,嵌合抗-人成神经细胞瘤单克隆抗体chCE7,嵌合抗-人肾细胞癌单克隆抗体chG250,嵌合抗-人结肠癌,肺癌和乳房癌单克隆抗体ING-1,人源化抗-人17-1A抗原单克隆抗体3622W94,人源化抗-人结肠直肠肿瘤抗体A33,针对GD3神经节苷脂R24的抗-人黑素瘤抗体,或者嵌合抗-人鳞状细胞癌单克隆抗体SF-25,抗-人EGFR抗体,抗-人EGFRvIII抗体,抗-人PSMA抗体和抗-人PSCA抗体,抗-人CD22抗体,抗-人CD30抗体,抗-人CD33抗体,抗-人CD38抗体,抗-人CD40抗体,抗-人CD45抗体,抗-人CD52抗体,抗-人CD138抗体,抗-人HLA-DR变体抗体,抗-人EpCAM抗体,抗-人CEA抗体,抗-人MUC1抗体,抗-人MUC1核心蛋白抗体,抗-人异常糖基化MUC1抗体,针对包含ED-B结构域的人纤连蛋白变体的抗体和抗-人HER2/neu抗体。
另一方面,本发明涉及在宿主细胞中产生多肽的方法,包括在允许产生所述Fc-介导的细胞毒性提高的多肽的条件下培养上述宿主细胞。在优选的实施方案中,所述方法进一步包括分离所述Fc-介导的细胞毒性提高的多肽。
在进一步优选的实施方案中,宿主细胞含有至少一种编码包括与免疫球蛋白的糖基化Fc区等价的区域的融合蛋白的核酸。
在优选的实施方案中,所述多肽的Fc区中的等分寡糖的比例大于50%,更优选地大于70%。在另一个实施方案中,Fc区中的等分杂合寡糖或半乳糖基化复合寡糖或其混合物的比例大于所述多肽的Fc区中的等分复合寡糖的比例。
在所述方法的一个优选方面中,所述多肽是抗CD20抗体并且由所述宿主细胞产生的抗-CD20抗体具有糖基化分布,如MALDI/TOF-MS所分析的,与图2E所示的基本上相当。
在所述方法的一个优选方面中,所述多肽是chG250单克隆抗体并且由所述宿主细胞产生的chG250抗体具有糖基化分布,如MALDI/TOF-MS分析的,与图7D所示的基本上相当。
另一方面,本发明涉及通过上述任何方法产生的具有提高的抗体依赖性细胞毒性(ADCC)的抗体。在优选的实施方案中,所述抗体选自抗-CD20抗体,chCE7,ch-G250,人源化抗-HER2单克隆抗体,ING-1,3622W94,SF-25,A33和R24。或者,所述多肽可以是通过上述任何方法制备的具有提高的Fc-介导的细胞毒性的包含相当于免疫球蛋白的Fc区的区域的抗体片段。
另一方面,本发明涉及通过上述任何方法制备的包含相当于免疫球蛋白的Fc区的区域的并且具有提高的Fc-介导的细胞毒性的融合蛋白。
另一方面,本发明涉及含有本发明的抗体,抗体片段或融合蛋白和药学可接受载体的药物组合物。
另一方面,本发明涉及一种治疗癌症的方法,包括对需要药物组合物的患者施用治疗有效量的所述药物组合物。
另一方面,本发明涉及一种治疗全部或部分由以B-细胞缺失为基础的病原性自身抗体引起的自身免疫疾病的改进方法,包括对需要的人受试者施用治疗有效量的免疫活性抗体,所述改进方法包括施用治疗有效量的如上所述制备的具有提高的ADCC的抗体。在优选的实施方案中,所述抗体是抗-CD20抗体。自身免疫疾病或病症的例子包括但不限于,免疫-介导的血小板减少症,例如急性特发性血小板减少紫癜和慢性特发性血小板减少紫癜,皮肤肌炎,舞蹈病,狼疮肾炎,风湿病发烧,多腺综合征,Henoch-Schonlein紫癜,链球菌肾炎,erythemanodosum,锁骨下静动脉闭合性血栓动脉炎,青铜色皮病,多形红斑,结节性多动脉炎,僵硬脊柱炎(ankylosing spondylitis),古德帕斯丘综合征,血栓性脉管炎ubiterans,原发性胆汁肝硬化,Hashimoto′s甲状腺炎,甲状腺毒症,慢性活动性肝炎,多肌炎/皮肌炎,多发性软骨炎,pamphigus vulgaris,韦格内氏肉芽肿病,膜肾病,肌萎缩性侧索硬化,脊髓痨,多肌痛,恶性贫血,快速进行性肾小球性肾炎和纤维组织形成肺泡炎,炎性反应例如皮炎,包括牛皮癣和皮肤炎(例如特应性皮炎);全身硬皮病和硬化病;与肠炎相关的反应(例如节段性回肠炎和溃疡性结肠炎);呼吸窘迫综合征(包括成年人呼吸窘迫综合征;ARDS);皮炎;脑膜炎;脑炎;葡萄膜炎;结肠炎;肾小球肾炎;变应性病症例如湿疹和哮喘以及其它病症,包括T细胞浸润和慢性炎症反应;动脉粥样硬化;白细胞粘着不足;类风湿性关节炎;全身红斑狼疮(SLE);糖尿病(例如I型糖尿病或胰岛素依赖性糖尿病);多发性硬化病;Reynaud′s综合征;自身免疫甲状腺炎;变应性脑脊髓炎;Sjorgen′s综合征;少年开始的糖尿病;和一般在肺结核,结节病,多肌炎,肉芽肿病和结节性脉管炎中发现的由细胞因子和T-淋巴细胞介导的急性和延迟性过敏症相关的免疫应答;恶性经闭(青铜色皮病);涉及白细胞渗出的疾病;中枢神经系统(CNS)炎症;多器官损伤综合征;溶血性贫血(包括但不限于冷球蛋白血症或Coombs阳性贫血);重症肌无力;抗原-抗体复合体介导的疾病;抗-血管小球肌膜病;抗磷脂综合征;变应性神经炎;格雷夫斯病;Lambert-Eaton肌无力综合征;类天疱疮大疱;天疱疮;自身免疫多内分泌腺病;莱特尔氏病;强直一人(stiff-man)综合征;被切特氏病;巨大细胞动脉炎;免疫复合体肾炎;IgA肾病;IgM多神经病;免疫血小板减少紫癜(ITP)或自身免疫血小板减少症等。在本发明的这方面,本发明的抗体被用来长时间排除血液中的正常B-细胞。
附图简要描述
图1.间接免疫荧光测试表明抗体制剂C2B8-25t对CD20阳性SB细胞的反应性。阴性对照物,包括HSB CD20阴性细胞系和只用二抗FITC-缀合的抗-人Fc多克隆抗体处理的细胞。
图2A-2E.从MabtheraTM(图2A),C2B8-nt(图2B),C2B8-2000t(图2C),C2B8-50t(图2D)和C2B8-25t(图2E)抗体样品衍生的寡糖的MALDI/TOF-MS图谱。寡糖作为[M+Na+]和[M+K+]离子出现。在头两个图谱中出现的寡糖是从不表达GnTIII的细胞培养物中得到的,而C,D,和E中的寡糖是从以不同水平(即四环素浓度)表达GnTIII的单一细胞系得到的。
图3A和3B.说明典型的人IgG Fc-结合寡糖结构(A)和部分N-连接糖基化途径(B)。(图3A)寡糖的核心由与Asn297连接的三个甘露糖(M)和两个N-乙酰基氨基葡糖(Gn)单糖残基组成。半乳糖(G),岩藻糖(F)和等分N-乙酰基氨基葡糖(Gn,boxed)可以存在或不存在。也可以存在末端N-乙酰基神经氨糖酸,但是图中没有包括。(图3B)导致形成主要寡糖类的部分N-连接糖基化途径(虚线框)。等分N-乙酰基氨基葡糖被表示为Gb n。下标数字表明每个寡糖中存在多少个单糖残基。每个结构给出了它的钠-结合[M+Na+]质谱。也包括包含岩藻糖(f)的那些结构的质谱。
图4A和4B.利妥希玛糖基化变体的ADCC活性。通过不同mAb浓度介导的人淋巴细胞(E∶T之比例是100∶1)对51Cr标记的CD20阳性SB细胞的溶胞作用测定细胞毒性百分比。(图4A)从单细胞得到但以递增GnTIII表达水平(即,递减的四环素浓度)产生的C2B8样品的活性。样品是C2B8-2000t,C2B8-50t,C2B8-25t和C2B8-nt(对照mAb来自不表达GnTIII的克隆)。(图4B)与MabtheraTM相当的C2B8-50t和C2B8-25t的ADCC活性。
图5.七个GnTIII表达克隆和野生型的蛋白质印迹分析。将30微克各种样品加载到8.75%SDS凝胶上,转移到PVDF膜并且用抗-c-myc单克隆抗体(9E10)探测。WT指wt-chG250-SP2/0细胞。
图6.分离的纯化抗体样品的SDS聚丙烯酰胺凝胶电泳。
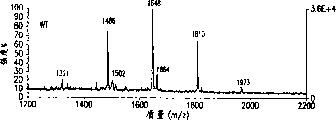
图7A-7D.得自由表达不同GnTIII水平的克隆和wt-chG250-SP2/0细胞产生的chG250 mAb样品的中性寡糖混合物的MALDI/TOF-MS图谱:WT(图7A),2F1(图7B),3D3(图7C),4E6(图7D)。
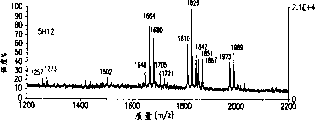
图8A-8D.得自由表达不同GANTE水平的克隆产生的chG250mAb样品的中性寡糖混合物的MALDI/TOF-MS图谱:4E8,(图8A);5G2,(图8B);4G3,(图8C);5H12,(图8D)。
图9.来自对照wt-chG250-SP2/-细胞和GnTIII转染克隆3D3和5H12的抗体样品的体外ADCC测定。
本发明的详细描述
这里使用的术语和本领域一般使用的一样,除非另外如下定义:
本文使用的术语抗体意在包括所有的抗体分子,抗体片段或者包括等价于免疫球蛋白的Fc区的区域的融合蛋白。
本文使用的术语等价于免疫球蛋白的Fc区的区域意在包括天然存在的免疫球蛋白的Fc区的等位变体以及具有取代,添加或缺失改变但是不实质性降低免疫球蛋白介导抗体依赖性细胞毒性能力的变体。例如,免疫球蛋白Fc区的N-末端或C-末端可以缺失一个或几个氨基酸而不实质性损失生物学功能。可以根据本领域公知的一般原则选择这样的变体以使对活性的影响最小(参见,例如,Bowie,J.U.等,Science 247:1306-10(1990)。
本文使用的术语糖蛋白-修饰的糖基转移酶指β(1,4)-N-乙酰基氨基葡糖基转移酶III(GnTIII)。
本文使用的术语工程和糖基化工程被认为包括天然存在的多肽或者其片段的糖基化作用方式的任何操作。糖基化工程包括细胞糖基化机构的代谢工程,包括实现改变在细胞中表达的糖蛋白糖基化作用的寡糖合成途径的遗传操作。此外,糖基化工程包括突变和细胞环境对糖基化作用的影响。
本文使用的术语宿主细胞覆盖所有类型的细胞体系,它们经工程处理产生令人感兴趣的修饰的糖基化形式的蛋白质,蛋白质片段,或肽,包括抗体和抗体片段。典型地,操纵宿主细胞表达最佳水平的GnTIII。宿主细胞包括培养的细胞,例如,哺乳动物培养细胞,例如CHO细胞,BHK细胞,NS0细胞,SP2/0细胞,Y0骨髓瘤细胞,P3X63小鼠骨髓瘤细胞,PER细胞,PER.C6细胞或杂交瘤细胞,酵母细胞和昆虫细胞,以及很少的,但是还有包含在转基因动物或培养组织中的细胞。
本文使用的术语Fc-介导的细胞毒性包括抗体-依赖性细胞毒性和包含人Fc-区的可溶性Fc-融合蛋白介导的细胞毒性。它是一种导致″人免疫效应子细胞″对″抗体-定向细胞″进行溶胞的免疫机理,其中:
″人免疫效应子细胞″是一群白细胞,它们展示其表面上的Fc受体,它们通过该受体与抗体的或Fc-融合蛋白的Fc区结合并且实施效应子功能。这样的细胞群可以包括但不限于外周血单核细胞(PBMC)和/或自然杀伤(NK)细胞。
″抗体-定向细胞″是通过抗体或Fc融合蛋白结合的细胞。抗体或Fc融合蛋白通过蛋白质Fc区的部分N-末端与靶细胞结合。
本文使用的术语提高的Fc-介导的细胞毒性被定义为通过上文定义的Fc-介导的细胞毒性的机理,在靶细胞周围的培养基中的给定浓度的抗体或Fc-融合蛋白下,于给定时间下溶解的″抗体-定向细胞″数目的增加,和/或通过Fc-介导的细胞毒性的机理,在给定时间下,实现给定数目的″抗体-定向细胞″的溶解需要的在靶细胞周围的培养基中抗体或Fc-融合蛋白浓度的降低。Fc-介导的细胞毒性的提高是相对于应用本领域技术人员公知的相同标准制备,纯化,配制和贮存方法由相同类型的宿主细胞产生的而不是通过本文描述的方法经工程处理而表达糖基转移酶GnTIII的宿主细胞产生的相同抗体或Fc-融合蛋白介导的细胞毒性。
所谓具有提高的抗体依赖性细胞毒性(ADCC)的抗体是指根据本领域技术人员公知的任何合适的方法测定具有提高的ADCC的抗体。
一种被接受的体外ADCC测定方法如下所示:
1)测定使用已知表达被抗体的抗原结合区识别的靶抗原的靶细胞;
2)测定使用从随机选择的健康供血者的血液中分离的人外周血单核细胞(PBMC)作为效应细胞;
3)根据下面的程序进行测定:
i)利用标准密度离心方法分离PBMC,并且以5×106细胞/毫升悬浮于RPMI细胞培养基;
ii)通过标准组织培养方法培养靶细胞,从成活率高于90%的指数成长期收获靶细胞,用RPMI细胞培养基洗涤,用100微居里的51Cr标记,用细胞培养基洗涤两次,并且以105细胞/毫升的密度再次悬浮于细胞培养基;
iii)将100微升上面最终的靶细胞悬浮液转移到96-孔微量滴定板的每个孔中;
iv)在细胞培养基中对抗体从4000ng/ml系列稀释,至0.04ng/ml,并且向96-孔微量滴定板中的靶细胞加入50微升得到的抗体溶液,以覆盖上面整个浓度范围的各种抗体浓度重复三次试验;
v)关于最大释放(MR)对照,板中含有标记的靶细胞的另外的3个孔接受50微升的2%(V/V)非离子洗涤剂水溶液(Nonidet,Sigma,St.Louis),以代替抗体溶液(上面的第iv点);
vi)关于自发释放(SR)对照,板中含有标记的靶细胞的另外的3个孔接受50微升的RPMI细胞培养基以代替抗体溶液(上面的第iv点);
vii)然后以50xg将96-孔微量滴定板离心1分钟,并且在4℃下温育1小时;
viii)向每个孔加入50微升的PBMC悬浮液(上面的第i点)以使效应物:靶细胞之比为25∶1,并且将板放在保温箱中在5%CO2的大气中在37℃下温育4小时;
ix)从各个孔收集不含细胞的上清液,并且用γ计数器定量测定试验上释放的放射性(ER);
x)根据公式(ER-MR)/(MR-SR)×100针对于每一个抗体浓度计算特异溶胞作用百分比,其中ER是针对那个抗体浓度定量测定的平均放射性(参见上面的第ix点),MR是针对MR对照物(参见上面的第v点)定量测定的平均放射性(参见上面的第ix点),和SR是对于SR对照物(参见上面的第vi点)定量测定的平均放射性(参见上面的第ix点);
4)″提高的ADCC″被定义为在上面测试的抗体浓度范围内发现的特异溶胞作用的最大百分比提高,和/或实现在上面测试的抗体浓度范围内发现的特异溶胞作用的最大百分比的一半所需抗体浓度的减小。ADCC的提高是相对于用上面的分析测定法的、相同抗体介导的、应用本领域技术人员公知的相同的标准制备,纯化,配制和贮存方法由相同类型的宿主细胞产生的而不是通过经工程处理而过量表达糖基转移酶GnTIII的宿主细胞产生的ADCC。
如本文使用的术语抗-CD20抗体意指特异性识别细胞表面35,000道尔顿的非糖基化磷蛋白的抗体,一般被命名为人B淋巴细胞限制性分化抗原Bp35,一般称作CD20。
需要糖基化模式修饰的蛋白质的编码核酸的鉴定和产生
本发明提供用于产生具有提高的抗体-依赖性细胞毒性的糖基化形式的抗体或抗体片段或包括抗体片段的融合蛋白的宿主细胞系统的制备和使用的方法。靶表位的鉴定和需要糖基化模式修饰的具有潜在治疗价值的抗体的制备,它们各自的编码核酸序列的分离都在本发明的范围内。
可以应用本领域公知的各种方法来制备抗令人感兴趣的靶表位的抗体。这样的抗体包括但不限于多克隆,单克隆,嵌合的,单链,Fab片段和Fab表达文库产生的片段。这样的抗体可以被用作,例如,作为诊断或治疗药物。作为治疗药物,中和抗体,即,与配体、底物或衔接分子结合相竞争的那些抗体是特别优选令人感兴趣的。
关于抗体的制备,通过用令人感兴趣的靶蛋白注射来免疫各种宿主动物,包括但不限于兔,小鼠,大鼠等。可以根据宿主物种使用各种佐剂来增强免疫应答,包括但不限于弗氏佐剂(完全的和不完全的),矿物凝胶,例如氢氧化铝,表面活性物质如溶血卵磷脂,多聚醇,聚阴离子,肽类,皂甙,油乳状液,匙孔血蓝蛋白,二硝基苯酚和可能有用的人佐剂如BCG(卡介苗)和小型棒状杆菌(Corynebacterium parvum)。
利用通过连续细胞系培养产生抗体分子的任何技术可以制备令人感兴趣的针对靶向物的单克隆抗体。这些包括但不限于最初由Kohler和Milstein,Nature 256:495-97(1975)描述的杂交瘤技术,人B-细胞杂交瘤技术(Kosbor等,Immunology Today 4:72(1983);Cote等,Proc.Natl.Acad.Sci.U.S.A.80:2026-30(1983)和EBV-杂交瘤技术(Cole等,Monoclonal Antibodies and Cancer Therapy 77-96(Alan R.Liss,Inc.,1985))。另外,可以利用为了制备″嵌合抗体″而开发的技术(Morrison等,Proc.Natl.Acad Sci.U.S.A.81:6851-55(1984);Neuberger等,Nature 312:604-08(1984);Takeda等,Nature 314:452-54(1985),通过拼接合适抗原特异性的小鼠抗体分子的基因与合适的生物活性的人抗体分子的基因。或者,可以采用被记载用于制备单链抗体的技术(美国专利US 4,946,778)制备具有所需特异性的单链抗体。
通过公知技术可以制备包含令人感兴趣的靶蛋白的特异结合位点的抗体片段。例如,这样的片段包括但不限于通过抗体分子的胃蛋白酶消化产生的F(ab′)2片段和通过将F(ab′)2片段的二硫桥还原而产生的Fab片段。或者,可以构建Fab表达文库(Huse等,Science 246:1275-81(1989),从而快速而容易地鉴定对令人感兴趣的靶蛋白具有所需特异性的单克隆Fab片段。
一旦鉴定出对需要以糖基化模式进行修饰的抗体或抗体片段,应用本领域公知的技术鉴定和分离核酸编码序列。
a.用于产生具有改变的糖基化模式的蛋白质的细胞系的产生
本发明提供了用于产生具有修饰的糖基化模式的蛋白质的宿主细胞表达系统。特别是,本发明提供了用于产生具有提高的治疗价值的糖基形式的蛋白质的宿主细胞系统。因此,本发明提供了经选择或工程处理而提高了糖蛋白-修饰糖基转移酶即β(1,4)-N-乙酰基氨基葡糖基转移酶III(GnTIII)的表达水平的宿主细胞表达系统。具体地说,可以工程处理这样的宿主细胞表达系统,使其包含与组成型或调节的启动子系统操作连接的编码GnTIII的重组核酸分子。或者,宿主细胞表达系统可以使用那种自然产生,经诱导产生,和/或被选择用来产生GnTIII的那些细胞表达系统。
在一个具体的实施方案中,本发明提供经工程处理以表达至少一种编码GnTIII的核酸的宿主细胞。一方面,用包含至少一种编码GnTIII的基因的核酸分子转化或转染宿主细胞。另一方面,以这样的方式工程处理和/或选择宿主细胞使得内源GnTIII被激活。例如,可以选择宿主细胞进行内源GnTIII的诱变触发表达。在一个具体的实施方案中,宿主细胞是CHOlecl0突变体。或者,可以工程处理宿主细胞使得内源GnTIII被激活。或者,通过向宿主细胞染色体插入组成型启动子元件,转座子,或逆转录病毒元件来工程处理宿主细胞使得内源GnTIII被激活。
一般来说,培养的细胞系类型可以被用作工程处理本发明的宿主细胞系的背景。在优选实施方案中,CHO细胞,BHK细胞,NS0细胞,SP2/0细胞,Y0骨髓瘤细胞,P3X63小鼠骨髓瘤细胞,PER细胞,PER.C6细胞或杂交瘤细胞,酵母细胞,或昆虫细胞被用作产生本发明的工程宿主细胞的背景细胞系。
本发明涉及包括本文定义的表达GnTIII的任何工程宿主细胞。
在组成型启动子的控制下,或者在调控表达系统的控制下,可以表达编码GnTIII的一种或几种核酸。合适的调节的表达系统包括但不限于四环素-调节的表达系统,蜕皮素-可诱导表达系统,lac-开关表达系统,糖皮质激素-可诱导表达系统,温度-可诱导表达系统,和金属硫蛋白金属-可诱导表达系统。如果宿主细胞系统中包含编码GnTIII的几种不同的核酸,它们中的一些可以在组成型启动子的控制下进行表达,而其余的在调节启动子控制下表达。认为最大表达水平是对细胞生长速率没有明显副作用的稳定的GnTIII表达的可能最高水平,利用常规试验测定。通过本领域一般公知的方法测定表达水平,包括使用GnTIII特异抗体的Western印迹分析,使用GnTIII特异核酸探针的Northern印迹分析,或者测定酶活性。或者,可以使用凝集素,该凝集素结合GnTIII的生物生物合成产物,例如,E4-PHA凝集素。或者,核酸还可以操作连接报道基因;通过测定与报道基因的表达水平相关的信号测定GnTIII表达水平。报道基因可以与编码所述GnTIII的核酸一起转录成单一mRNA分子;它们各自的编码序列可以通过内部核糖体进入位点(IRES)或者通过帽-独立性翻译增强子(CITE)来连接。报道基因可以与编码所述GnTIII的至少一种核酸一起进行翻译从而形成单链多肽。编码GnTIII的核酸在单一启动子的控制下可以与报道基因操作连接,这样编码GnTIII的核酸和报道基因被转录到RNA分子中,其另外被剪接成两个独立的信使RNA(mRNA)分子;得到的mRNA中的一个被翻译成所述报道蛋白质,另一个被翻译成所述GnTIII。
如果表达编码GnTIII的几种不同的核酸,它们可以以这样的方式排列以使它们被转录成一种或几种mRNA分子。如果它们被转录成单一mRNA分子,他们各自的编码序列可以通过内部核糖体进入位点(IRES)或者通过帽-独立性翻译增强子(CITE)来连接。它们可以从单一启动子被转录到RNA分子中,其另外被剪接成几个分开的信使RNA(mRNA)分子,其然后各自被翻译成它们各自编码的GnTIII。
在其它实施方案中,本发明提供了用于产生具有提高的抗体-依赖性细胞毒性的治疗性抗体的宿主细胞表达体系,和在其表面展示促进Fc-介导的细胞毒性的IgG Fc区的细胞。一般情况下,宿主细胞表达体系经工程处理和/或选择来表达编码抗体的核酸,该抗体需要产生改变的糖基化形式,以及至少一种编码GnTIII的核酸。在一个实施方案中,用编码GnTIII的至少一种基因转染宿主细胞体系。一般情况下,选择鉴定转染的细胞并且分离稳定表达GnTIII的克隆。在另一个实施方案中,选择表达内源GnTIII的宿主细胞。例如,选择带有触发另外沉默GnTIII进行表达的突变的细胞。例如,已知CHO细胞带有沉默GnTIII基因,该基因在一些突变体中是有活性的,例如,在突变体Lecl0中。此外,可以利用本领域公知的方法激活沉默GnTIII,包括插入调节型或组成型启动子,使用转座子,逆转录病毒元件等。也可以应用基因剔除技术或者应用核糖体方法调节宿主细胞的GnTIII表达水平,并且因此在本发明的范围内。
任何类型的培养细胞系能被用作对本发明的宿主细胞系进行工程处理的背景。在优选的实施方案中,可以使用例如CHO细胞,BHK细胞,NS0细胞,SP2/0细胞,Y0骨髓瘤细胞,P3X63小鼠骨髓瘤细胞,PER细胞,PER.C6细胞或杂交瘤细胞,酵母细胞,或昆虫细胞。典型地,工程处理这样的细胞系以便进一步包含编码整个抗体分子,抗体片段,或者包括等价于免疫球蛋白的Fc区的区域的融合蛋白中的至少一种转染的核酸,在另一个实施方案中,表达令人感兴趣的特定抗体的杂交瘤细胞系被用作产生本发明的工程宿主细胞的背景细胞系。
典型地,宿主细胞系中的至少一种核酸编码GnTIII。
在组成型启动子或者调节表达体系的控制下可以表达编码GnTIII的一种或几种核酸。合适的调节表达系统包括但不限于四环素-调节的表达系统,蜕皮素-可诱导表达系统,lac-开关表达系统,糖皮质激素-可诱导表达系统,温度-可诱导表达系统和金属硫蛋白金属-可诱导表达系统。如果宿主细胞系统中包括编码GnTIII的几种不同的核酸,它们中的一些可以在组成型启动子的控制下表达,而其它的在调节启动子控制下表达。认为最大表达水平是对细胞生长速度没有明显副作用的稳定GnTIII表达的可能最高水平,并能利用常规试验测定。通过本领域中公知的方法测定表达水平,包括使用GnTIII特异抗体的Western印迹分析,使用GnTIII特异核酸探针的Northern印迹分析或者测定酶活性。或者,可以使用凝集素,该凝集素结合GnTIII的生物生物合成产物,例如,E4-PHA凝集素。或者,核酸可以操作连接报道基因;通过测定与报道基因的表达水平相关的信号测定糖蛋白修饰的糖基化转移酶的表达水平。报道基因可以与编码所述糖蛋白修饰的糖基化转移酶的核酸一起转录成单一mRNA分子;它们各自的编码序列可以通过内部核糖体进入位点(IRES)或者通过帽-独立性翻译增强子(CITE)而连接。报道基因可以与编码GnTIII的至少一种核酸一起翻译以便形成单链多肽。编码GnTIII的核酸在单一启动子的控制下可以与报道基因操作连接,这样编码GnTIII的核酸和报道基因被转录到RNA分子中,其另外被剪接成两个分开的信使RNA(mRNA)分子;得到的mRNA中的一个被翻译成所述报道蛋白质,另一个被翻译成所述GnTIII。
如果表达编码GnTIII的几种不同的核酸,它们可以以这样的方式排列使得它们被转录成一种或几种mRNA分子。如果它们被转录成单一mRNA分子,它们各自的编码序列可以通过内部核糖体进入位点(IRES)或者通过帽-独立性翻译增强子(CITE)来连接。他们可以从单一启动子被转录到RNA分子中,其另外被剪接成几个分开的信使RNA(mRNA)分子,然后各自被翻译成他们各自编码的GnTIII。
i.表达体系
可以应用本领域技术人员公知的方法构建包含令人感兴趣的蛋白质的编码序列和GnTIII的编码序列和合适的转录/翻译控制信号的编码系列的表达载体。这些方法包括体外重组DNA技术,合成技术和体内重组/基因重组。参见,例如,Maniais等,Molecular Cloning ALaboratory Manual,Cold Spring Harbor Laboratory,N.Y.(1989)和Ausubel等,Current Protocols in Molecular Biology,GreenePublishing Associates and Wiley Interscience,N.Y(1989)中记载的技术。
可以利用各种宿主表达载体系统来表达令人感兴趣的蛋白质的编码序列和GnTIII的编码序列。优选地,哺乳动物细胞被用作采用包含令人感兴趣的蛋白质的编码序列和GnTIII的编码序列的重组质粒DNA或粘粒DNA表达载体转染的宿主细胞系统。
最优选地,CHO细胞,BHK细胞,NS0细胞,SP2/0细胞,Y0骨髓瘤细胞,P3X63小鼠骨髓瘤细胞,PER细胞,PER.C6细胞或杂交瘤细胞,酵母细胞或昆虫细胞被用作宿主细胞体系。在另一个实施方案中,可以包括其它真核宿主细胞体系,包括,用包含令人感兴趣的蛋白质的编码序列和GnTIII的编码序列的重组酵母表达载体转化的酵母细胞;用包含令人感兴趣的蛋白质的编码序列和GnTIII的编码序列的重组病毒表达载体(例如杆状病毒)转染的昆虫细胞体系;用包含令人感兴趣的蛋白质的编码序列和GnTIII的编码序列的重组病毒表达载体(例如,花椰菜花叶病毒,CaMV;烟草花叶病毒,TMV)转染的或者用包含令人感兴趣的蛋白质的编码序列和GnTIII的编码序列的重组质粒表达载体(例如,Ti质粒)转化的植物细胞体系;或者用重组病毒表达载体(例如腺病毒,牛痘病毒)转染的动物细胞体系,包括经工程处理而包含在双微染色体中稳定扩增的(CHO/dhfr)或者不稳定扩增的(例如,鼠细胞系)编码令人感兴趣的蛋白质的DNA的多个拷贝和GnTIII的编码序列的细胞系。
对于本发明的方法,稳定表达一般比瞬时表达优选,这是因为它通常能或得多个可重复结果,而且更适合大规模生产。与其使用包含病毒复制起点的表达载体,不如用各个通过合适的表达调控元件(例如,启动子,增强子,序列,转录终止子,多腺苷酸化位点等)控制的编码核酸和可选择标记转化的宿主细胞。导入外源DNA之后,可以让工程细胞在营养充分的培养基中生长1-2天,然后换成选择性培养基。重组质粒中的可选择标记带来对选择的抗性并且使得选择质粒稳定整合到它们的染色体中的细胞,并且生长形成中心,接着进行克隆并且被扩展成细胞系。
可以使用很多选择体系,包括但不限于单纯疱疹病毒胸苷激酶(Wigler等,Cell 11:223(1977)),次黄嘌呤-鸟嘌呤磷酸核糖基转移酶(Szybalska & Szybalski,Proc.Natl.Acad.Sci.USA 48:2026(1962))和腺嘌呤磷酸核糖基转移酶(Lowy等,Cell 22:817(1980))基因,其可以分别在tk-,hgprt-或aprt-细胞中进行使用。还有,抗代谢物抗性可以被用作选择dhfr的基础,它带来对甲氨喋呤的抗性(Wigler等,Natl.Acad.Sci.USA 77:3567(1989);O′Hare等,Proc.Natl.Acad.Sci.USA78:1527(1981));gpt,它带来对霉酚酸的抗性(Mulligan & Berg,Proc.Natl.Acad.Sci.USA 78:2072(1981));neo,它带来对氨基糖苷G-418的抗性(Colberre-Garapin等,J.MoL Biol.150:1(1981));和hygro,它带来对潮霉素的抗性(Santerre等,Gene 30:147(1984))基因。最近,有人描述了其它可选择基因,即trpB,它使得细胞利用吲哚代替色氨酸;hisD,它使得细胞利用组氨醇代替组氨酸(Hartman &Mulligan,Proc.Natl.Acad.Sci.USA85:8047(1988));谷酰胺合酶体系;和ODC(鸟氨酸脱羧酶),它带来对鸟氨酸脱羧酶抑制剂,2-(二氟甲基)-DL鸟氨酸,DFMO的抗性(McConlogue,in:CurrentCommunications in Molecular Biology,Cold Spring HarborLaboratory ed.(1987))。
ii.表达具有修饰的糖基化模式的蛋白质的转染体或转化体的鉴定
通过至少四种一般方法可以鉴定包含编码序列并且表达生物活性基因产物的宿主细胞:(a)DNA-DNA或DNA-RNA杂交;(b)存在或不存在″标记″基因功能;(c)根据各个mRNA转录本在宿主细胞中的表达评价转录水平;和(d)根据免疫测定或者通过其生物活性测定检测基因产物。
在第一种方法中,通过DNA-DNA或DNA-RNA杂交,使用包含分别与各个编码序列或其部分或其衍生物同源的核苷酸序列的探针,能检测表达载体中插入的令人感兴趣的蛋白质的编码序列和GnTIII的编码序列的存在。
在第二种方法中,根据存在或不存在某些″标记″基因功能(例如,胸苷激酶激酶活性,抗生素抗性,甲氨喋呤抗性,转化表型,杆状病毒中包函体形成等),能鉴定和选择重组表达载体/宿主体系。例如,如果在载体的标记基因序列中插入令人感兴趣的蛋白质的编码序列和GnTIII的编码序列,通过不存在标记基因功能能鉴定包含各个编码序列的重组体。或者,标记基因可以与编码序列串联放置,处于用来控制编码序列表达的相同的或不同的启动子的控制下。标记基因响应诱导或选择而进行表达表明对令人感兴趣的蛋白质的编码序列和GnTIII的编码序列进行表达。
在第三种方法中,通过杂交测定能评价令人感兴趣的蛋白质的编码区和GnTIII的编码序列的转录活性。例如,使用与令人感兴趣的蛋白质的编码序列和GnTIII的编码序列或者其特定部分同源的探针通过Northern印迹能分离和分析RNA。或者,可以提取宿主细胞的完整核酸并且分析与这种探针的杂交。
在第四种方法中,能免疫学测定令人感兴趣的蛋白质的蛋白质产物的表达和GnTIII的编码序列的表达,例如通过蛋白质印迹,免疫分析,如放射免疫-沉淀法,酶联免疫测试等。但是表达系统成功的最终试验包括检测生物学活性基因产物。
b.具有改变的糖基化模式的蛋白质和蛋白质片段的产生和用途
i.
具有提高的抗体-依赖性细胞毒性的抗体的产生和用途
在优选的实施方案中,本发明提供具有提高的抗体-依赖性细胞毒性的糖基化形式的抗体和抗体片段。
用于治疗某些类型癌症的未缀合单克隆抗体(mAb)的临床试验最近得到了令人鼓舞的结果。Dillman,CancerBiother.& Radiopharm.12:223-25(1997);Deo等,Immunology Today 18:127(1997)。已经批准嵌合的未缀合IgGl用于低级或滤泡B-细胞非何杰金氏淋巴组织瘤,Dillman,CancerBiother. & Radiopham.12:223-25(1997),而另一种未缀合的mAb,一种人源化IgG1定向实体乳房肿瘤,在三期临床试验中也证明有希望的结果。Deo等,Immunology Today 18:127(1997)。这两种mAbs的抗原在他们各自的肿瘤细胞中高度表达并且抗体在体外和体内通过效应细胞介导有力的肿瘤破坏。相反,很多其它有小的肿瘤特异性的未缀合的mAb不能触发足以有效用于临床应用的效应物功能。Frost等,Cancer 80:317-33(1997);Surfus等,J.Immunother.19:184-91(1996)。对于这些较弱的mAb中的一些,最近测试了辅助细胞因子治疗。细胞因子的加入通过提高循环淋巴细胞的活性和数目能刺激抗体-依赖性细胞毒性(ADCC)。Frost等,Cancer80:317-33(1997);Surfus等,J.Immunother.19:184-91(1996)。ADCC,对抗体-靶向细胞进行的溶胞作用攻击触发白细胞受体与抗体的恒定区(Fc)结合。Deo等,Immunology Today 18:127(1997)。
与提高未缀合IgG1的ADCC活性的不同的但起补充作用的方法是对抗体的Fc区进行工程处理以提高他们对淋巴细胞受体(FcγRs)的亲和性。蛋白质工程研究表明FcγRs与IgG CH2结构域的较低绞合区相互作用。Lund等,J.Immunol.157:4963-69(1996)。但是,FcγR结合还需要与CH2区中保守的Asn 297供价连接的寡糖的存在。Lund等,J.Immunol.157:4963-69(1996);Wright和Morrison,Trends Biotech.15:26-31(1997),提出寡糖和多肽两者都直接影响相互作用位点,并且需要寡糖来保持活性CH2多肽构象。因此,寡糖结构的修饰能被开发成提高相互作用亲和性的一种方法。
IgG分子在它的Fc区带有两个N-连接寡糖,每条重链上一个。和任何糖蛋白一样,将抗体制备成多糖基化形式,它们共有相同的多肽主链但是在糖基化位点处连接不同的寡糖。一般在血清IgG的Fc区中发现的寡糖是复合体双触角型(Wormald等,Biochemistry 36:130-38(1997),有低水平的末端唾液酸和等分N-乙酰氨基葡糖(GIcNAc)和可变程度的末端半乳糖基化和核心岩藻糖基化作用。一些研究表明FcγR结合需要的最小糖结构位于寡糖核心中。Lund等,J.Immunol.157:4963-69(1996)。去除末端半乳糖导致ADCC活性降低大约两倍,表明这些残基在FcγR受体结合中的作用。Lund等,J.Immunol.157:4963-69(1996)。
工业和学术中用于制备未缀合治疗性mAb的得自小鼠或仓鼠的细胞系一般将所需寡糖决定簇与Fc位点连接。但是这些细胞系中表达的IgG缺少血清IgG中少量发现的等分GlcNAc。Lifely等,Glycobiology 318:813-22(1995)。相反,最近发现大鼠骨髓瘤产生的人源化IgGl(CAMPATH-1H)在它的一些糖基化形式中带有GlcNAc。Lifely等,Glycobiology 318:813-22(1995)。大鼠细胞产生的抗体达到和标准细胞系中产生的CAMPATH-1H抗体类似的体外ADCC活性,但是抗体浓度低得多。
CAMPATH抗原一般以高浓度存在于淋巴组织瘤细胞上,这种嵌合的mAb在没有等分GlcNAc存在下具有高ADCC活性。Lifely等,Glycobiology318:813-22(1995)。在N-连接糖基化途径中,通过酶(1,4)-N-乙酰氨基葡糖转移酶III(GnTIII)加上等分GlcNAc。Schachter,Biochem.Cell Biol.64:163-81(1986)。
本发明人使用单一的产生抗体的CHO细胞系,它事先经工程处理以外部调节方式表达不同水平的克隆GnTIII基因。这种方法首次建立了GnTIII表达与修饰的抗体ADCC活性之间的精确关系。
本发明人先前表明根据公开的方法修饰的C2B8抗体比相同细胞培养和纯化条件下产生的标准未修饰的C2B8抗体ADCC活性高大约16倍。简要地说,如人淋巴细胞对SB细胞(CD20+)的体外溶胞作用所测定的,在没有GnTIII表达的CHO-tTA-C2B8细胞中表达的C2B8抗体样品证明细胞毒素活性大约31%(1微克/毫升抗体浓度)。相反,在1微克/毫升抗体浓度下,从基础性大量表达水平表达GnTIII的CHO细胞培养物中得到的C2B8抗体表明相对于相同抗体浓度的对照物来说ADCC活性提高33%。此外,与相同抗体浓度的对照物相比,提高GnTIII表达,将最大ADCC活性(在1微克/毫升抗体浓度下)提高将近80%(参见国际公开WO 99/54342,其全文在此引作参考)。
本发明的具有提高的抗体-依赖性细胞毒性的其它抗体包括但不限于本发明方法产生的抗-人成神经细胞瘤单克隆抗体(chCE7),本发明方法产生的嵌合抗-人肾细胞癌单克隆抗体(ch-G250),本发明方法产生的人源化抗-HER2单克隆抗体(例如,Trastuzumab(HERCEPTIN)),本发明方法产生的嵌合抗-人结肠癌,肺癌和乳房癌单克隆抗体(ING-1),人源化抗-人17-1A抗原单克隆抗体(3622W94),本发明方法产生的人源化抗-人结肠直肠肿瘤抗体(A33),本发明方法产生的针对GD3神经节苷脂的抗-人黑素瘤抗体(R24),本发明方法产生的嵌合抗-人鳞状细胞癌单克隆抗体(SF-25),本发明方法产生的抗-人小细胞肺癌单克隆抗体(BEC2,ImClone Systems,Merck KgaA),本发明方法产生的抗-人非何杰金氏淋巴瘤单克隆抗体(Bexxar(tositumomab,Coulter Pharmaceuticals),Oncolym(Techniclone,Alpha Therapeutic)),本发明方法产生的抗-人鳞状细胞头和颈癌单克隆抗体(C225,ImClone Systems),本发明方法制备的抗-人直肠和结肠癌单克隆抗体(帕罗瑞星(edrecolomab),Centocor,GlaxoWellcome),本发明方法制备的抗-人卵巢癌单克隆抗体(啥拉宗钠,Antisoma),本发明方法制备的抗-人急性骨髓性白血病癌单克隆抗体(SmartM195,Protein Design Labs,Kanebo),本发明方法制备的抗-人恶性神经胶质瘤单克隆抗体(Cotara,Techniclone,CambridgeAntibody Technology),本发明方法制备的抗-人B细胞非-何杰金氏淋巴瘤单克隆抗体(IDEC-Y2B8,IDEC Pharmaceuticals),本发明方法制备的抗-人实体肿瘤单克隆抗体(CEA-Cide,Immunomedics),本发明方法制备的抗-人直肠和结肠癌单克隆抗体(碘131-MN-14,knmunomedics),本发明方法制备的抗-人卵巢癌,肾癌,乳房癌和前列腺癌单克隆抗体(MDX-210,Medarex,Novartis),本发明方法制备的抗-人直肠结肠和胰腺癌单克隆抗体(TTMA,Pharmacie &Upjohn),本发明方法制备的抗-人TAG-72-表达癌单克隆抗体(MDX-220,Medarex),本发明方法制备的抗-人EGFr-表达癌单克隆抗体(MDX-447),本发明方法制备的抗-VEGF单克隆抗体(Genentech),本发明方法制备的抗-人乳房癌,肺癌,前列腺癌和胰腺癌和恶性黑素瘤单克隆抗体(BrevaRex,AltaRex),本发明方法制备的抗-人急性骨髓性白血病单克隆抗体(单克隆抗体结合物,Immunex)。另外,本发明涉及包括等价于免疫球蛋白的Fc区的区域的抗体片段和融合蛋白。
ii.包含促进Fc-介导的细胞毒性的等价于免疫球蛋白的Fc区的区域的融合蛋白的产生和使用
如上文讨论的,本发明涉及用于提高治疗性抗体的ADCC活性的方法。这通过对这样的抗体的Fc区的糖基化形式进行工程处理来实现,特别是通过将在它们的Fc区中带有与保守糖基化位点N-连接的等分复合体寡糖和等分杂合寡糖的抗体分子的比例最大化来实现。这个侧略能用来提高带有等价于免疫球蛋白的Fc区的区域的任何分子介导的而不只是治疗性抗体介导的抗不需要细胞的Fc-介导的细胞毒性,因为糖基化工程所导入的变化只影响Fc区,以及它与ADCC机理中涉及的效应细胞表面上的Fc受体相互作用。对其可以应用本发明公开的方法的包含Fc的分子包括但不限于,(a)由与Fc-区的N-末端融合的靶向蛋白质结构域构成的可溶性融合蛋白(Chamov和Ashkenazi,Trends Biotech.14:52(1996)和(b)由定位于与Fc-区的N-末端融合的质膜的II型跨膜结构域构成的质膜-固定的融合蛋白(Stabila,P.F.,NatureBiotech.16:1357(1998))。
对于可溶性融合蛋白(a)来说,靶向结构域指导融合蛋白与不期望的细胞例如癌细胞结合,即,以与治疗性抗体类似的方式。因此,本发明公开的提高这些分子介导的Fc-介导的细胞毒素活性的方法的应用与对治疗性抗体应用的方法相同。
对于质膜-固定的融合蛋白(b)来说,体内不期望的细胞不得不表达编码融合蛋白的基因。或者通过基因治疗方法,即通过用指导融合蛋白编码基因表达的质粒或病毒载体体内转染细胞,或者通过在体内植入经遗传工程处理而在他们表面上表达融合蛋白的细胞来实现。后面这种细胞一般以聚合物胶囊的形式被植入体内(胶囊化细胞治疗),Fc-介导的细胞毒性机理不能破坏它们。但是,一旦胶囊破碎,逃脱的细胞就变成不期望的了,他们之后由Fc-介导的细胞毒性消除。Stabila等,Nature Biotech.16:1357(1998)。在这种情况下,应用本发明公开的方法,或者向基因治疗载体中插入另外的基因表达盒指导GnTIII适当的或最大表达水平,或者通过工程处理要植入的细胞以适合的或最大水平表达GnTIII。在这两种情况下,公开的方法的目的在于将表面展示的带有等分复合体寡糖和/或等分杂合寡糖的Fc区的比例提高或最大化。
下面的实施例更详细地解释本发明。给出下面的制备和实施例使得本领域技术人员更清楚地理解和实施本发明。但是,本发明范围不受例举的实施方案的限制,实施例只是为了详细说明本发明的一方面,而功能等价的方法是在本发明的范围之内的。事实上,除了这里所述的之外的本发明的各种各样的修饰对于本领域技术人员来说根据上面的描述和附图而变得显而易见。这样的修饰是在后面的权利要求书范围内的。
实施例1
通过产生IDEC-CEB8的细胞系的糖基化工程获得的具有提高的抗体-
依赖性细胞毒性的嵌合式抗-CD20抗体IDEC-C2B8的新变型
IDEC-C2B8的VH和VL编码区的合成和哺乳动物表达载体的构建
利用PCR(Kobayashi,N.,等,Biotechniques 23:500-503(1997))以一步法从一组重叠的单链寡核苷酸组装编码IDEC-C2B8抗体的VH和VI区的cDNA。从公开的国际专利申请(国际公开号:WO 94/11026)获得IDEC-C2B8 VL和VH的原始序列数据。将组装的VL和VHcDNA片段亚克隆到pBluescriptIIKS(+)中,测序并且通过分别与人恒定轻链(Igκ)和重链(IgGl)cDNA连接而直接连接,使用的是可变区和恒定区接点处导入的而不改变原来氨基酸残基序列的独特限制性位点(Umana,P.等,Nat Biotechnol.17:176-180(1999);Reff,M.E.,等,Blood53:435-445(1994))。各个全长cDNA被分别亚克隆到pcDNA3.1(+)(Invitrogen,Leek,The Netherlands)中,得到用于嵌合C2B8轻链(pC2B8L)和重链(pC2B8H)的哺乳动物表达载体。
在CHO细胞中表达不同水平GnTIII的IDEC-C2Bf的制备
建立两个CHO细胞系,根据四环素浓度在培养基中表达不同水平GnTIII的CHO-tet-GnTIII;和CHO-tTA,先前描述过的不表达GnTIII的亲代细胞系(Umana,P.等,Nat Biotechnol.17:176 180(1999);Umana,P.,等,BiotechnolBioeng.65:542-549(1999))。利用磷酸钙方法,每个细胞系采用载体pC2B8L,pC2B8H和pZeoSV2(+)(对Zeocin抗性;Invitrogen,Leek,TheNetherlands)进行共同转染。将Zeocin抗性克隆转移到96-孔板中并且利用对人恒定区(4)特异性的ELISA测试来测定IDEC-C2B8产生。从选择的克隆(CHO-tet-GnTIII-C2B8)的平行培养物获得三个IDEC-C2B8样品,只是加给培养基的四环素浓度不同(分别是25,50和2000ng/mL)。在指数期后期收集培养物上清液。从产生CHO-tTA的克隆获得另外的抗体样品,CHO-tTA-C2B8,在相同的条件下培养,但是培养基中不加入四环素。通过蛋白质A亲和层析法从培养基中纯化抗体样品,并且如先前所述将缓冲液换成阳离子交换柱上的PBS(Umana,P.等,NatBiotechnol.17:176-180(1999))。使用从Molecular Probes(Leiden,The Netherlands)得到的以荧光为基础的试剂盒以利妥希玛作为标准物测定抗体浓度。
间接免疫荧光法
CD20-阳性细胞(SB细胞;ATCC保藏号ATCC CCL120)和CD20-阴性细胞(HSB细胞;ATCC保藏号ATCCCCL120.1)分别与Hank′s平衡盐溶液(GibcoBRL,Basel,Switzerland)中2.5微克/毫升的CHO-tet GnTIII-衍生的IDEC-C2B8抗体和2%牛血清白蛋白级分V(Roche Diagnostics,Rotkreuz,瑞士)(HBSSB)温育1小时。作为阴性对照物,使用HBSSB代替C2B8抗体。对于所有样品,PITC-结合的抗-人Fc多克隆抗体被用作二抗(SIGMA,圣路易斯)。使用Leica荧光显微镜(Wetzlar,德国)检查细胞。
通过MALDIlTOF-MS分析寡糖分布
如前所述(Umana,P.等,Nat Biotechnol.17:176-180(1999)),从C2B8抗体样品,MabTheraTM(利妥希玛欧洲对应物;RStahel,Universittspital,瑞士,馈赠),C2B8-25t,C2B8-50t,C2B8-2000t和C2B8-nt,(各100微克)衍生中性的、N-连接寡糖。简要地说,首先用Arthrobacter ureafaciens唾液酸酶(Oxford Glycosciences,Abingson,UK)处理抗体样品,去除所有的唾液酸单糖残基。然后使用肽-N-糖苷酶F(Oxford Glycosciences)从去唾液酸抗体样品释放中性N-l连接寡糖,利用微量柱子纯化,并且利用Elite Voyager 400分光计(PerseptiveBiosystems,Farmingham,MA)通过MALDI/TOF-MS进行分析。
ADCC活性测定
通过用Ficoll-Paque(Pharmacia Biotech,Dübendorf,瑞士)梯度离心从用肝素盐化的新鲜人血中分离外周血单核细胞(PBMC)(在从相同的健康供血者获得的所有的实验中)。通过塑料附着从PBMC(效应物)中排除单核细胞。37℃下用100μCi51Cr(Amersham,Dubendorf,瑞士)将CD20-阳性SB(靶)细胞标记90分钟,用RPMI(GibcoBRL,Basel,瑞士)洗涤两次并且以105细胞/毫升的浓度再次悬浮。将50微升用RPMI培养基中稀释的C2B8mAb加到96-孔圆底微量滴定板(Greiner,Langenthal,瑞士)中的100微升SB细胞(10,000细胞/孔)中,以50xg离心1分钟,并且在4℃下温育1小时。接着,在96-孔的每个孔中加入50微升效应细胞(以2×107细胞/毫升悬浮于RPMI培养基中),得到最终E∶T之比为100。37℃和5%CO2下将板温育4小时,用Skatron收集系统(Skatron Instruments,Sterling,VA)收集上清液,并且利用Cobra 05005γ计数器(Canberra Packard,Meriden,CT)计数(ER,实验释放)。通过取代C2B8 mAb而向100微升标记的靶细胞分别加入100微升的1%Nonidet(Sigma,St.Louis)或100微升的RPMI培养基从而获得的最大(MR)和自发(SR)释放。所有的数据点重复进行三次。用下面的公式计算特异溶胞作用(%):(ER-SR)/(MR-SR)×100。
结果和讨论
IDEC-C2B8的产生和特异性抗原结合的验证
建立具有稳定的四环素-调节表达GnTIII和稳定的组成型表达IDEC-C2B8的CHO-tet-GnTIII细胞,并且规模放大达到生产一组抗体样品。放大规模期间,相同克隆的平行培养物在三个不同的四环素浓度,25,50和2000ng/ml下生长。先前已经证明这些四环素水平导致不同水平的GnTIII和等分寡糖(Umana,P.等,Nat Biotechnol.17:176-180(1999);Umana,P.等,Biotechnol Bioeng.65:542-549(1999))。还建立了不表达GnTIII的产生C2B8-的对照细胞系,并且在与三个CHO-tet-GnTIII平行培养相同的条件下培养。蛋白质A-亲和层析之后,通过SDS-PAGE和考马斯兰染色评估mAb纯度高于95%。根据加到用于它们制备的培养基中的四环素浓度命名样品:C2B8-25t,C2B8-50t,C2B8-2000t和C2B8-nt(即,对非等分对照物没有四环素)。通过使用CD20-阳性和CD20-阴性细胞(图1)的间接免疫荧光法,样品C2B8-25t表现出特异性抗原结合性,表明合成的VL和VH基因片段功能是正确的。
用MALDI/TOF-MS分析寡糖分布
通过释放的中性寡糖混合物的MALDI/TOF-MS分析各个抗体样品的糖基化作用分布。在该项技术中,不同质量的寡糖表现为谱图中的各个峰以及它们的比例通过相对峰高来定量反映(Harvey,D.J.,Rapid Common MassSpectrom.7:614-619(1993);Harvey,D.J.,等,Glycoconj J.15:333-338(1998))。以它们预期的分子量为基础将寡糖结构分配对应给不同的峰,先前寡糖的结构数据来自相同宿主产生的IgGI mAb,和有关N-连接寡糖生物合成途径的信息。
发现GnTIII表达水平(即,四环素浓度)与来自不同抗体样品的等分寡糖的量之间存在清楚的关系。正如所预料的,MabTheraTM和C2B8-nt,它们来自不表达GnTIII的宿主,不带有等分寡糖(图2A和2B)。相反,等分结构的量最多达到样品C2B8-2000t中寡糖混合物的35%,即,GnTIII表达的基础水平。在这种情况下,主要等分寡糖峰是复合型的,在m/z 1689和m/z 1851峰之间不明确(图2C)。下一个较高的GnTIII表达水平,样品C2B8-50t,导致这些峰增大(包括m/z 1705和1861处它们的相关钠加成物)20%左右。这样的增大伴随它们的非等分对应物分别在m/z 1486和1648处的减小(图2D)。在最高GnTIII表达水平,样品C2B8-25t,GnTIII的主要底物,m/z 1486,几乎减小至基础水平,而复合体等分结构(m/z 1689和1851)减少,而m/z1664,1810和1826峰增大(图2E)。这些峰能分配给等分杂合化合物,给半乳糖基化复合体寡糖,或者给两者的混合物。但是它们的相对增大与等分杂合化合物的积累相吻合,因为CnTIII过量表达能在该途径的早期转移生物合成通量(参见图3A和3B)。对于该样品,等分寡糖结构(复合体和杂合型)的量达到大约80%。
IDEG-C2B8糖基化变体的ADCC活性
测定CD20-阳性SB细胞的体外溶胞作用,比较不同的C2B8 mAb糖基化变体的ADCC活性。还研究了来自缺乏GnTIII的亲代细胞系的其它mAb样品,C2B8-nt。
以基础GnTIII表达水平产生的并且带有低含量等分寡糖的C2B8-2000t样品比C2B8-nt活性稍微高一些(图4A)。在接下来的较高水平的GnTIII-表达下,样品C2B8-50t带有大约相等含量的等分和非等分寡糖,但是不显著介导更高的靶细胞溶胞作用。但是,在最低四环素浓度下,样品C2B8-25t,它含有高达80%的等分寡糖结构,在整个抗体浓度范围内,比其余样品的活性高得多。在抗体浓度低10倍下达到样品C2B8-nt的ADCC活性的最大水平(图4A)。样品C2B8-25t还被证明相对对照物来说最大ADCC活性显著提高(50%相对30%溶胞作用)。
样品C2B8-50t和C2B8-25t,带有最高比例的等分寡糖,进一步与MabtheraTM比较ADCC活性,MabtheraTM是最近在欧洲上市的RituxanTM型(图4B)。样品C2B8-50t表现出活性稍微增加,而样品C2B8-25t在所有的抗体浓度下明显超过MabtheraTM。要求低大约5-10倍浓度的C2B8-25t达到MabtheraTM的最大ADCC活性,并且的最大活性比MabtheraTM高大约25%。
这些结果表明,一般来说,C2B8抗体的体外ADCC活性与Fc区带有等分寡糖的分子的比例有关。我们以前报道过,对于chCE7,具有低基线水平的ADCC活性的抗体来说,通过将等分寡糖的成分增加到高于天然存在的抗体中发现的水平来获得活性的显著提高(Umana,P.等,Nat Biotechnol.17:176-180(1999))。对于C2B8 mAb也是如此,它在没有等分寡糖存在下已经具有高ADCC活性。但是对chCE7来说,在等分寡糖主要是复合体型的情况下,在GnTIII表达水平下发现ADCC活性有了非常大的提高(Umana,P.等,Vat Biotechnol.17:176-180(1999))。对于有效的C2B8 mAb来说,只是在所研究的GnTIII的最高表达水平下发现活性有如此的加强,这种情况下等分寡糖主要转换成杂合型(图2)。对于两种mAb来说,具有最高活性的样品等分寡糖比非等分寡糖的最高活性高得多。总的来说,这些观察表明可能两种复合体和杂合等分寡糖对于ADCC活性都是重要的。
在复合体和杂合寡糖中,等分GlcNAc导致寡糖构象发生很大变化(Balaji,P.V.等,Int.J.Biol.Macromol.18:101-114(1996))。寡糖的一部分中发生变化,该变化与CH2结构域中的多肽广泛作用(Jefferis,R.等,Immunol Rev.163:59-76(1998))。因为多肽在这个位置相对柔顺(Jefferis,R.,等,Immunol Rev.163:59-76(1998)),有可能的是,等分N-乙酰氨基葡糖通过Fc区中的构象变化介导它的生物学作用。可能改变的构象可能已经存在于自然界中,因为所有的血清IgG带有等分寡糖。工程抗体和天然抗体之间的主要区别是展示更具活性的构象的分子的比例。
用于提高未缀合mAb的活性的各种方法最近正在进行临床评价,包括放射-免疫治疗,抗体依赖性酶/前体药物治疗,采用细胞因子的免疫毒素和佐剂治疗(Hjelm Skog A.,等,Cancer ImmunolImmunother.48:463-470(1999);Blakey,D.C.等,Cell Biophys.25:175-183(1994);Wiseman,G.A.等,Clin Cancer Res.5:3281s-3296s(1999);Hank,J.A.,等,Cancer Res.50:5234-5239(1990))。这些技术都使活性大大提高,但是它们还能导致副作用显著增加,与未缀合mAb相比,它们存在升高的生产成本和从制备到对患者给药复杂的后勤。这里给出的技术为获得效能增加而保持简单的生产过程提供了一条替代途径,并且应该适用于很多未缀合的mAb。
实施例2
通过产生chG250的细胞系的糖基化工程而获得的具有提高的抗体-依
赖性细胞毒性的抗-肾细胞癌抗体chG250的新变型
1.细胞培养
在补加有1∶100(v/v)青霉素/链霉素/抗真菌溶液(SIGMA,Buchs,瑞士)的标准细胞培养基中培养产生chG250嵌合mAb的SP2/0小鼠骨髓瘤细胞(wt-chG250-SP2/0细胞)。细胞在37℃和5%CO2潮湿气氛下在组织培养瓶中培养。每3-4天更换一次培养基。在含有10%DMSO的培养基中冷冻细胞。
2.表达pGnTIII-puro的SP2/0细胞的产生
通过电穿孔,采用用于通过IRES与嘌呤霉素抗性基因操作连接的组成型GnTIII表达的载体转染wt-chG250-SP2/0骨髓瘤细胞。电穿孔之前24小时,更换培养基,并且以5×105细胞/毫升接种细胞。4℃下以1300rpm将七百万个细胞离心4分钟。用3mL新培养基洗涤细胞并且再次离心。将细胞重新悬浮于0.3-0.5ml体积的反应混合物中,培养基中含有1.25%(v/v)DMSO和20-30微克DNA。然后将电穿孔混合物转移到0.4cm比色杯中并且使用购自Bio Rad的GenePulser在低压(250-300V)和高电容(960μF)下进行脉冲。电穿孔之后,将细胞快速转移给T25培养瓶中的6mL 1.25%(v/v)DMSO培养基并且在37℃下温育。通过电穿孔2天后对培养基施加2微克/毫升嘌呤霉素来选择稳定整合体。2-3周之后获得稳定的嘌呤霉素-抗性混合集落。通过FACS获得单细胞产生克隆并且接着进行扩冲并且保持在嘌呤霉素选择条件下。
3.蛋白质印迹
通过蛋白质印迹筛选用于GnTIII表达的嘌呤霉素-抗性克隆。蛋白质印迹清楚地表明克隆5H12,4E6和4E8表达最高水平的GnTIII。5G2还表现出中等强度的GnTIII带,而2F1,3D3和4G3具有最低的带强度,因此表达最低量的GnTIII(图5)。
4.由包括野生型在内的七个GnTIII-表达克隆制备和纯化chG250单克隆抗体
以3×105细胞/毫升将克隆2F1,3D3,4E6,4E8,4G3,SG2,5H12和野生型(wt-chG250-SP2/0细胞)接种到总体积是130毫升的培养基中,并且在一个三联体瓶中培养。用于接种的细胞都处于完全指数生长期,因此当开始生产批次时认为细胞处于相同的生长状态。将细胞培养4天在晚指数生长期收集含有抗体的上清液保证再现性。用两步色谱法纯化chG250单克隆抗体。首先用HiTrap蛋白质A亲和层析法纯化每一批产生的含有chG250单克隆抗体的培养上清液。蛋白质A对于人IgGFc区是高度特异性的。来自蛋白质A洗出液的合并样品通过在Resource Slml柱子(AmershamPharmaciaBiotech)上进行的阳离子交换层析法缓冲液交换成PBS。通过SDS-染色和考马斯兰染色判断最终纯度高于95%(图6)。使用已知浓度的野生型抗体用标准校正曲线测定各个样品的浓度。
5.来自表达不同GnTIII水平的七个克隆的mAb制剂的寡糖分
布
通过飞行质谱(MALDI/TOF-MS)的矩阵辅助激光解吸/电离时间获得寡糖分布,MALDI/TOF-MS精确提供了不同寡糖结构的分子量。该项技术定量分析混合物中不同寡糖结构之间的比例。中性寡糖主要表现为[M+Na+]离子,但是有时它们伴有较小的[M+K+]离子,导致m/z质量增加16。作为钾离子加成物出现的结构百分比取决于基体含量,因此各个样品之间是不同的。使用2,5-脱水苯甲酸(2,5-DHB)作为基体分析从各个抗体制剂得到的中性N-连接寡糖的混合物。谱图中的一些峰明确地对应于特定寡糖结构,这是因为已知的单糖组成和独特质量。但是,有时多重结构对应于特定质量。MALDI能确定质量但是不能区别异构体。生物合成途径知识和以前的结构数据在大多数情况下能将寡糖结构与谱图中的峰对应起来。
来自wt-chG250-SP2/0细胞系产生的mAb样品的寡糖,它不表达GnTIII,包含非等分双触角复合体(m/z 1486)和单-或双-半乳糖基化非等分双触角复合体结构(图7A),核心区中的两个α(1,6)-岩藻糖基化结构(峰分别是m/z 1648和1810)。
GnTIII的表达产生两种类型的等分Fc-结合寡糖结构:复合体或杂合体。复合体等分寡糖明确对应于m/z 1543,1689,1705,1851和1867的峰([M+K+]加成物)。正如所预期的,等分寡糖的增加伴随m/z1486和1648峰的减小,那相应于非等分复合体寡糖。对于来自GnTIII表达克隆的所有样品,GnTIII的主要底物(m/z 1486)大大减少。根据预期的,对应于m/z 1648峰的非等分复合体寡糖型的百分比对于表达最高水平GnTIII的克隆(克隆4E6,4B8,5G2和SH12)有最低值。这两个减小的峰有利于等分复合体和等分杂合体型寡糖的积累(图7A-7D和8A-8D)。对于来自表达较低量的GnTIII的克隆的样品,等分复合体寡糖的百分比较高。这与下面的事实相符合:较高的GnTIII表达水平可能将生物合成通量改变为等分杂合体结构,从而减小复合体和复合体等分化合物的相对比例。对于等分杂合体结构,两种可能的结构有时被对应于单一峰。因此,为了接近这些结构相对全部寡糖集合物的百分比而进行一些假定。峰m/z 1664,1680,1810和1826可能、对应于等分杂合体型,对应于半乳糖化的复合体寡糖,或者它们的混合物。由于wt-抗体制剂具有相对低百分比峰1664的事实,假设这个峰,以从不同克隆得到的抗体样品中明显量出现,完全对应于等分杂合体结构(图7A-7D和8A-8D)。但是为了使具体结构对应于峰m/z 1810和1826,必须进行进一步的定性。总之,通过过量表达GnTIII,产生等分寡糖结构,并且他们相对的比例与GnTIII表达水平有关。
6.通过钙黄绿素-AM滞留测定抗体介导的细胞毒性
测定细胞毒性的钙黄绿素-AM滞留方法测定细胞与抗体温育之后残留在细胞内的染料荧光。在补加有10%胎牛血清的1.8毫升RPMI-1640细胞培养基(GIBCO BRL,Basel,瑞士)中用10μM钙黄绿素-AM(Molecular Probes,Eugene,OR)在37℃和5%CO2潮湿气氛中将四百万G250抗原-阳性细胞(靶物)标记30分钟。用培养基将细胞洗涤两次并且再次悬浮于12毫升AIMV无血清培养基(GIBCO BRL,Basel,瑞士)中。然后将标记的细胞转移到U-底96-孔(30,000细胞/孔)并且一式三份与不同浓度的抗体在4℃下温育1小时。通过用Ficoll-Paque(Pharmacia Biotech,Dübendorf,瑞士)梯度离心从用肝素盐处理的新鲜人血中分离外周血单核细胞(PBMC)(在从相同的健康供血者获得的所有的实验中)。在三个孔中在50微升体积中加入PBMC,得到的效应物与靶物之比(E∶T之比)是25∶1并且最终体积是200微升。然后在37℃和5%CO2气氛中将96-孔板温育4小时。然后将96-孔板以700xg离心5分钟并且弃除上清液。细胞沉降物用Hank′s平衡盐溶液(HBSS)洗涤两次,并且溶解于200微升0.05M硼酸钠,pH9,0.1%TritonX-100中。用FLUOstar微板读数器(BMG LabTechnologies,Offenburg,德国)测定靶细胞中荧光染料的残留。相对总的溶解对照物计算具体的溶胞作用,是从靶细胞接触皂甙(200微克/毫升AIMV;SIGMA,Buchs,瑞士)代替接触抗体而得出的。用下面的公式计算特定的溶胞作用(%):
其中Fmed代表单独用培养基处理的靶细胞的荧光并且认为是PMBC的非特异性溶解,Fexp代表用抗体处理的细胞的荧光,而Fdet代表用皂甙代替抗体处理过的细胞的荧光。
为了测定chG250的修饰的糖基化变体对体外ADCC活性的作用,将G250抗原-阳性靶细胞与有和没有不同浓度的chG250抗体样品的PBMC培养。将来自野生型细胞系的未修饰chG250抗体的细胞毒性分别与来自表达中等和高水平GnTIII的两个细胞系(3D3,5H12)的两种抗体制剂相比较(参见图5)。
在分析中使用的整个浓度范围中,未修饰的chG250抗体不介导显著ADCC活性(活性与背景没有显著不同)。用来自克隆3D3的抗体样品观察到在2微克/毫升下提高了的ADCC活性(接近20%,参见图9),克隆3D3表达中等水平的GnTIII。这个抗体样品的细胞毒性不随较高抗体浓度而提高。正如所预料的,来自克隆5H12的抗体制剂表现出其介导抗靶细胞的ADCC能力比样品3D3和未修饰抗体高得多。这种抗体制剂的最大ADCC活性是50%左右,并且当与未修饰对照样品比较时以低125-倍的浓度显著介导显著的ADCC活性。
实施例3
对慢性移植物-抗-宿主疾病患者治疗免疫-介导的血小板减少症
慢性移植物-抗-宿主疾病中的自身免疫血小板减少症代表导致临床疾病的B-细胞失调的例子。为了对患有慢性移植物-抗-宿主疾病患者的免疫-介导的血小板减少症进行治疗,根据Ratanatharathorn,V.等,Ann.Intern.Med.133(4):275 79(2000)(其全文在此引作参考)所述,对患者施用根据本发明方法制备的并且具有提高的ADCC的抗-CD20嵌合单克隆抗体。具体地说,每周对患者输注抗体,375mg/m2,持续四周。抗体治疗导致在外周血中显著消耗B细胞并且降低血小板缔合抗体的水平。
实施例4
治疗严重的免疫-介导的纯红细胞发育不全和溶血性贫血
免疫-介导的后天纯红细胞发育不全(PRCA)是一种罕见的疾病,经常与其他自身免疫现象相关。为了治疗免疫-介导的后天纯红细胞发育不全患者,根据Zecca,M.等,Blood 12:3995-97(1997)(其全文在此引作参考)所述,对患者施用根据本发明方法制备的并且具有提高的ADCC的抗-CD20嵌合单克隆抗体。具体地说,每周对PRCA和自身免疫溶血性贫血患者施用两个剂量的抗体,375mg/m2。抗体治疗之后,开始静脉施用免疫球蛋白的替代治疗。该项治疗导致B细胞显著消耗并且伴随提高的血红蛋白水平,网状细胞数显著增加。
清楚的是,除了上文具体描述和实施例之外可以以其它方式实施本发明。着眼于上面的教导对本发明进行多种修饰和改变是可能的,因此在所附权利要求书的保护范围内。
这里引述的所有的公开物(包括专利,专利申请,期刊文章,实验手册,书和其它文献)的全部公开内容在此引作参考。
Claims (38)
1.一种通过表达至少一种编码β(1,4)-N-乙酰基氨基葡糖转移酶III(GnTIII)的核酸而产生具有提高的Fc-介导的细胞毒性的多肽的工程宿主细胞,其中由所述宿主细胞产生的所述多肽选自整个抗体分子,抗体片段,和包含与免疫球蛋白的Fc区等价的区域的融合蛋白,并且其中以足以提高Fc区中带有等分杂合寡糖或半乳糖基化复合体寡糖或其混合物的所述多肽相对于Fc区中带有等分复合体寡糖的多肽的比例的量表达所述GnTIII。
2.权利要求1的宿主细胞,其中所述多肽是IgG或其片段。
3.权利要求1的宿主细胞,其中所述多肽是IgG1或其片段。
4.权利要求1的宿主细胞,其中所述多肽是包含与人IgG的Fc区等价的区域的融合蛋白。
5.权利要求1的宿主细胞,其中包含至少一种编码GnTIII的基因的核酸分子已被导入所述宿主细胞中。
6.权利要求1的宿主细胞,其中所述宿主细胞经工程处理使得内源GnTIII基因被激活。
7.权利要求6的宿主细胞,其中所述内源GnTIII已经通过将提高基因表达的DNA元件插入到宿主染色体中而被激活。
8.权利要求6的宿主细胞,其中选择带有触发内源GnTIII表达的突变的所述宿主细胞。
9.权利要求8的宿主细胞,其中所述宿主细胞是CHO细胞突变体lec 10。
10.权利要求1的宿主细胞,其中所述宿主细胞是CHO细胞,BHK细胞,NS0细胞,SP2/0细胞,Y0骨髓瘤细胞,P3X63小鼠骨髓瘤细胞,PER细胞,PER.C6细胞或杂交瘤细胞。
11.权利要求10的宿主细胞,其中所述多肽是抗-CD20抗体。
12.权利要求11的宿主细胞,其中所述抗-CD20抗体是IDEC-C2B8。
13.权利要求10的宿主细胞,其中所述宿主细胞是SP2/0细胞。
14.权利要求13的宿主细胞,其中所述抗体是嵌合式抗-人肾细胞癌单克隆抗体chG250。
15.权利要求5的宿主细胞,其中所述编码GnTIII的至少一种基因已被导入所述宿主细胞染色体中。
16.权利要求6的宿主细胞,其中所述内源GnTIII已经通过将启动子元件,转座子,或逆转录病毒元件插入到宿主细胞的染色体中而被激活。
17.权利要求1的宿主细胞,进一步包含至少一种编码抗体分子,抗体片段或者含有等价于免疫球蛋白的Fc区的区域的融合蛋白的转染的核酸。
18.权利要求1的宿主细胞,其中所述至少一种编码GnTIII的核酸与组成型启动子元件操作性连接。
19.权利要求17的宿主细胞,其中所述宿主细胞包含编码下面蛋白质中的至少一种转染的核酸:抗-CD20抗体,嵌合式抗-人成神经细胞瘤单克隆抗体chCE7,嵌合式抗-人肾细胞癌单克隆抗体chG250,嵌合式抗-人结肠癌,肺癌,和乳房癌单克隆抗体ING-1,人源化抗-人17-1A抗原单克隆抗体3622W94,人源化抗-人结肠直肠肿瘤抗体A33,抗GD3神经节苷脂R24的抗-人黑素瘤抗体,或者嵌合式抗-人鳞状细胞癌单克隆抗体SF-25,抗-人EGFR抗体,抗-人EGFRvIII抗体,抗-人PSMA抗体,抗-人PSCA抗体,抗-人CD22抗体,抗-人CD30抗体,抗-人CD33抗体,抗-人CD38抗体,抗-人CD40抗体,抗-人CD45抗体,抗-人CD52抗体,抗-人CD138抗体,抗-人HLA-DR变体抗体,抗-人EpCAM抗体,抗-人CEA抗体,抗-人MUC1抗体,抗-人MUC1核心蛋白抗体,抗-人畸变糖基化MUC1抗体,抗包含ED-B结构域的人纤连蛋白变体的抗体或抗-人HER2/neu抗体。
20.一种在宿主细胞中产生多肽的方法,包括在允许产生具有提高的Fc-介导的细胞毒性的所述多肽的条件下培养权利要求1-19任一项的宿主细胞。
21.权利要求20的方法,进一步包括分离所述具有提高的Fc-介导的细胞毒性的多肽。
22.权利要求20的方法,其中所述宿主细胞包含编码含有等价于免疫球蛋白的Fc区的区域的融合蛋白的至少一种核酸。
23.权利要求20的方法,其中所述多肽的Fc区中50%以上的寡糖是等分的。
24.权利要求20的方法,其中所述多肽的Fc区中70%以上的寡糖是等分的。
25.权利要求20的方法,其中Fc区中等分杂合寡糖或半乳糖基化复合体寡糖或其混合物的比例大于所述多肽的Fc区中等分复合体寡糖的比例。
26.权利要求20的方法,其中所述多肽是抗CD20抗体IDEC-C2B8,并且由所述宿主细胞产生的IDEC-C2B8抗体具有糖基化分布,如MALDI/TOF-MS分析的,与图2E所示的基本上相当。
27.权利要求20的方法,其中所述多肽是chG250单克隆抗体并且所述宿主细胞产生的chG250抗体具有糖基化分布,如MALDI/TOF-MS分析的,与图7D所示的基本上相当。
28.通过权利要求21的方法制备的具有提高的抗体依赖性细胞毒性(ADCC)的抗体。
29.权利要求28的抗体,其中所述抗体选自IDEC-C2B8,chCE7,ch-G250,人源化抗-HER2单克隆抗体,ING-1,3622W94,SF-25,A33,和R24。
30.通过权利要求21的方法制备的包含等价于免疫球蛋白的Fc区的区域的并且具有提高的Fc-介导的细胞毒性的抗体片段。
31.通过权利要求21的方法制备的包含等价于免疫球蛋白的Fc区的区域的并且具有提高的Fc-介导的细胞毒性的融合蛋白。
32.含有权利要求28的抗体和药学可接受载体的药物组合物。
33.含有权利要求30的抗体片段和药学可接受载体的药物组合物。
34.含有权利要求31的融合蛋白和药学可接受载体的药物组合物。
35.一种治疗癌症的方法,包括对需要治疗的患者施用治疗有效量的权利要求32-34任一项的药物组合物。
36.一种改进的以B-细胞排除为基础的疾病治疗方法,包括对需要治疗的人患者施用治疗有效量的抗体,所述改进包括施用治疗有效量的由权利要求28的方法制备的抗体。
37.权利要求36的改进的方法,其中所述抗体是抗-CD20单克隆抗体。
38.权利要求37的改进的方法,其中所述抗-CD20抗体是IDEC-C2B8。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US30951601P | 2001-08-03 | 2001-08-03 | |
| US60/309,516 | 2001-08-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN1555411A true CN1555411A (zh) | 2004-12-15 |
Family
ID=23198536
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNA028181735A Pending CN1555411A (zh) | 2001-08-03 | 2002-08-05 | 抗体-依赖性细胞毒性增大的抗体糖基化变体 |
Country Status (15)
| Country | Link |
|---|---|
| US (5) | US20030175884A1 (zh) |
| EP (2) | EP1423510A4 (zh) |
| JP (2) | JP2005524379A (zh) |
| KR (2) | KR20100018071A (zh) |
| CN (1) | CN1555411A (zh) |
| AU (1) | AU2002339845B2 (zh) |
| CA (2) | CA2838062C (zh) |
| HU (1) | HUP0700103A3 (zh) |
| IL (2) | IL160170A0 (zh) |
| MX (1) | MXPA04001072A (zh) |
| NO (1) | NO332457B1 (zh) |
| NZ (5) | NZ581474A (zh) |
| PL (1) | PL217751B1 (zh) |
| RU (1) | RU2321630C2 (zh) |
| WO (1) | WO2003011878A2 (zh) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8021856B2 (en) | 1998-04-20 | 2011-09-20 | Roche Glycart Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
Families Citing this family (917)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6136311A (en) | 1996-05-06 | 2000-10-24 | Cornell Research Foundation, Inc. | Treatment and diagnosis of cancer |
| EP2180007B2 (en) * | 1998-04-20 | 2017-08-30 | Roche Glycart AG | Glycosylation engineering of antibodies for improving antibody-dependent cellular cytotoxicity |
| CA2354377C (en) | 1998-12-09 | 2007-04-10 | Tatsuji Seki | A method for manufacturing glycoproteins having human-type glycosylation |
| JP5084984B2 (ja) | 1999-02-17 | 2012-11-28 | シーエスエル、リミテッド | 免疫原複合体およびそれに関する方法 |
| MXPA02004142A (es) | 1999-10-26 | 2003-08-20 | Plant Res Int Bv | Glucosilacion tipo mamifero en plantas. |
| AU5345901A (en) * | 2000-04-13 | 2001-10-30 | Univ Rockefeller | Enhancement of antibody-mediated immune responses |
| US7598055B2 (en) | 2000-06-28 | 2009-10-06 | Glycofi, Inc. | N-acetylglucosaminyltransferase III expression in lower eukaryotes |
| EP2339013B1 (en) * | 2000-06-28 | 2014-07-02 | GlycoFi, Inc. | Methods for producing modified glycoproteins |
| US7449308B2 (en) | 2000-06-28 | 2008-11-11 | Glycofi, Inc. | Combinatorial DNA library for producing modified N-glycans in lower eukaryotes |
| US8697394B2 (en) * | 2000-06-28 | 2014-04-15 | Glycofi, Inc. | Production of modified glycoproteins having multiple antennary structures |
| CA2434364C (en) | 2001-01-19 | 2013-03-19 | The Dow Chemical Company | Method for secretory production of glycoprotein having human-type sugar chain using plant cell |
| WO2002079255A1 (en) * | 2001-04-02 | 2002-10-10 | Idec Pharmaceuticals Corporation | RECOMBINANT ANTIBODIES COEXPRESSED WITH GnTIII |
| EP1467755A1 (en) * | 2001-12-21 | 2004-10-20 | Antigenics Inc. | Compositions comprising immunoreactive reagents and saponins, and methods of use thereof |
| US20070148171A1 (en) * | 2002-09-27 | 2007-06-28 | Xencor, Inc. | Optimized anti-CD30 antibodies |
| US8188231B2 (en) | 2002-09-27 | 2012-05-29 | Xencor, Inc. | Optimized FC variants |
| US20040132101A1 (en) | 2002-09-27 | 2004-07-08 | Xencor | Optimized Fc variants and methods for their generation |
| US20080254027A1 (en) * | 2002-03-01 | 2008-10-16 | Bernett Matthew J | Optimized CD5 antibodies and methods of using the same |
| US7662925B2 (en) * | 2002-03-01 | 2010-02-16 | Xencor, Inc. | Optimized Fc variants and methods for their generation |
| US7317091B2 (en) * | 2002-03-01 | 2008-01-08 | Xencor, Inc. | Optimized Fc variants |
| US20080260731A1 (en) * | 2002-03-01 | 2008-10-23 | Bernett Matthew J | Optimized antibodies that target cd19 |
| EP1485486B1 (en) | 2002-03-19 | 2012-11-21 | Stichting Dienst Landbouwkundig Onderzoek | Optimizing glycan processing in plants |
| JP5350573B2 (ja) | 2002-03-19 | 2013-11-27 | スティヒティング ディーンスト ランドバウクンディフ オンデルズーク | 植物におけるgntiii発現 |
| US20060235208A1 (en) * | 2002-09-27 | 2006-10-19 | Xencor, Inc. | Fc variants with optimized properties |
| AR042145A1 (es) | 2002-11-27 | 2005-06-08 | Dow Agrociences Llc | Produccion de inmunoglobulinas en plantas con una fucocilacion reducida |
| US7638270B2 (en) * | 2003-01-24 | 2009-12-29 | Agensys, Inc. | Nucleic acids and corresponding proteins entitled 254P1D6B useful in treatment and detection of cancer |
| US7332299B2 (en) | 2003-02-20 | 2008-02-19 | Glycofi, Inc. | Endomannosidases in the modification of glycoproteins in eukaryotes |
| US8084582B2 (en) | 2003-03-03 | 2011-12-27 | Xencor, Inc. | Optimized anti-CD20 monoclonal antibodies having Fc variants |
| US8388955B2 (en) * | 2003-03-03 | 2013-03-05 | Xencor, Inc. | Fc variants |
| US20070275460A1 (en) * | 2003-03-03 | 2007-11-29 | Xencor.Inc. | Fc Variants With Optimized Fc Receptor Binding Properties |
| US20090010920A1 (en) | 2003-03-03 | 2009-01-08 | Xencor, Inc. | Fc Variants Having Decreased Affinity for FcyRIIb |
| US9051373B2 (en) | 2003-05-02 | 2015-06-09 | Xencor, Inc. | Optimized Fc variants |
| AR044388A1 (es) | 2003-05-20 | 2005-09-07 | Applied Molecular Evolution | Moleculas de union a cd20 |
| US8101720B2 (en) | 2004-10-21 | 2012-01-24 | Xencor, Inc. | Immunoglobulin insertions, deletions and substitutions |
| US9714282B2 (en) | 2003-09-26 | 2017-07-25 | Xencor, Inc. | Optimized Fc variants and methods for their generation |
| SG10202008722QA (en) * | 2003-11-05 | 2020-10-29 | Roche Glycart Ag | Cd20 antibodies with increased fc receptor binding affinity and effector function |
| US20050249723A1 (en) * | 2003-12-22 | 2005-11-10 | Xencor, Inc. | Fc polypeptides with novel Fc ligand binding sites |
| DK1716181T3 (da) | 2004-02-19 | 2010-03-01 | Genentech Inc | CDR-reparerede antistoffer |
| AR048098A1 (es) * | 2004-03-15 | 2006-03-29 | Wyeth Corp | Conjugados de caliqueamicina |
| AU2005227326B2 (en) * | 2004-03-24 | 2009-12-03 | Xencor, Inc. | Immunoglobulin variants outside the Fc region |
| US20060002930A1 (en) * | 2004-04-16 | 2006-01-05 | Genentech, Inc. | Treatment of disorders |
| MXPA06014069A (es) | 2004-06-04 | 2007-04-25 | Genentech Inc | Metodo para tratar esclerosis multiple. |
| KR101413402B1 (ko) * | 2004-07-09 | 2014-06-27 | 츄가이 세이야꾸 가부시키가이샤 | 항 글리피칸 3 항체 |
| EP1765871A1 (en) * | 2004-07-14 | 2007-03-28 | Igeneon Krebs-Immuntherapie Forschungs- und Entwicklungs-AG | N-glycosylated antibody |
| US20150010550A1 (en) | 2004-07-15 | 2015-01-08 | Xencor, Inc. | OPTIMIZED Fc VARIANTS |
| US20060074225A1 (en) * | 2004-09-14 | 2006-04-06 | Xencor, Inc. | Monomeric immunoglobulin Fc domains |
| RU2411956C2 (ru) | 2004-10-05 | 2011-02-20 | Дженентек, Инк. | Способ лечения васкулита |
| JO3000B1 (ar) | 2004-10-20 | 2016-09-05 | Genentech Inc | مركبات أجسام مضادة . |
| BRPI0518279A2 (pt) * | 2004-10-26 | 2008-11-11 | Chugai Pharmaceutical Co Ltd | anticorpo antiglipicam 3 tendo cadeia de aÇécar modificada |
| DK1817340T3 (da) * | 2004-11-12 | 2012-08-13 | Xencor Inc | Fc-varianter med ændret binding til fcrn |
| US8802820B2 (en) | 2004-11-12 | 2014-08-12 | Xencor, Inc. | Fc variants with altered binding to FcRn |
| US8546543B2 (en) | 2004-11-12 | 2013-10-01 | Xencor, Inc. | Fc variants that extend antibody half-life |
| US8367805B2 (en) | 2004-11-12 | 2013-02-05 | Xencor, Inc. | Fc variants with altered binding to FcRn |
| EP1858925A2 (en) * | 2005-01-12 | 2007-11-28 | Xencor, Inc. | Antibodies and fc fusion proteins with altered immunogenicity |
| JP2008528486A (ja) | 2005-01-21 | 2008-07-31 | ジェネンテック・インコーポレーテッド | Her抗体の一定投薬 |
| BRPI0607315B1 (pt) | 2005-02-07 | 2022-05-17 | Roche Glycart Ag | Molécula de ligação ao antígeno anti-egfr humanizada e seus usos, composição farmacêutica, e método para detectar a presença de egfr em uma amostra |
| AU2006214033B2 (en) * | 2005-02-18 | 2011-08-11 | Medarex, L.L.C. | Monoclonal antibodies against CD30 lacking fucosyl residues |
| ZA200707078B (en) | 2005-02-23 | 2008-11-26 | Genentech Inc | Extending time to disease progression or survival in cancer patients |
| KR20070114324A (ko) * | 2005-03-25 | 2007-11-30 | 글리카트 바이오테크놀로지 아게 | Mcsp 에 향하며 증가된 fc 수용체 결합 친화도 및효과기 작용을 갖는 항원 결합 분자 |
| PE20061324A1 (es) | 2005-04-29 | 2007-01-15 | Centocor Inc | Anticuerpos anti-il-6, composiciones, metodos y usos |
| MX2007013924A (es) * | 2005-05-09 | 2008-01-28 | Glycart Biotechnology Ag | Moleculas que unen antigeno que tienen regiones fc modificadas y union alterada a receptores fc. |
| CA2610234A1 (en) * | 2005-06-02 | 2006-12-07 | Astrazeneca Ab | Antibodies directed to cd20 and uses thereof |
| CA2608818A1 (en) * | 2005-06-03 | 2006-12-14 | Genentech, Inc. | Method of producing antibodies with modified fucosylation level |
| WO2007005955A2 (en) | 2005-06-30 | 2007-01-11 | Centocor, Inc. | Anti-il-23 antibodies, compositions, methods and uses |
| KR20080030673A (ko) | 2005-07-21 | 2008-04-04 | 젠맵 에이/에스 | Fc 수용체와 결합하는 항체 약품 성분에 대한 효력 검정 |
| WO2007024743A2 (en) * | 2005-08-19 | 2007-03-01 | Centocor, Inc. | Proteolysis resistant antibody preparations |
| TWI615407B (zh) * | 2005-08-26 | 2018-02-21 | 羅齊克雷雅公司 | 具有經改變細胞傳訊活性之改質抗原結合分子 |
| CA2624189A1 (en) * | 2005-10-03 | 2007-04-12 | Xencor, Inc. | Fc variants with optimized fc receptor binding properties |
| JP4860703B2 (ja) * | 2005-10-06 | 2012-01-25 | ゼンコー・インコーポレイテッド | 最適化された抗cd30抗体 |
| US20070087005A1 (en) * | 2005-10-14 | 2007-04-19 | Lazar Gregory A | Anti-glypican-3 antibody |
| MY149159A (en) | 2005-11-15 | 2013-07-31 | Hoffmann La Roche | Method for treating joint damage |
| KR101453570B1 (ko) | 2005-12-02 | 2014-10-22 | 제넨테크, 인크. | Il-22 및 il-22r에 결합하는 항체를 포함하는,사이토카인 신호 전달과 관련된 질환 및 장애 치료용조성물 및 치료 방법 |
| SI2548577T1 (sl) | 2005-12-29 | 2017-05-31 | Janssen Biotech, Inc. | Človeška protitelesa proti il-23, sestavki, postopki in uporabe |
| CA2638785C (en) | 2006-01-05 | 2017-02-21 | Genentech, Inc. | Anti-ephb4 antibodies and methods using same |
| US20070166306A1 (en) * | 2006-01-17 | 2007-07-19 | Fey Georg H M | Anti-CD19 antibody composition and method |
| AU2007205939B2 (en) | 2006-01-17 | 2012-12-13 | Synthon Biopharmaceuticals B.V. | Compositions and methods for humanization and optimization of N-glycans in plants |
| US8716033B2 (en) * | 2006-02-10 | 2014-05-06 | Life Technologies Corporation | Oligosaccharide modification and labeling of proteins |
| AR059851A1 (es) | 2006-03-16 | 2008-04-30 | Genentech Inc | Anticuerpos de la egfl7 y metodos de uso |
| ATE509033T1 (de) | 2006-03-20 | 2011-05-15 | Univ California | Manipulierte anti-prostatastammzellenantigen (psca)-antikörper für krebs-targeting |
| MX2008015132A (es) | 2006-05-30 | 2008-12-10 | Genentech Inc | Anticuerpos e inmunoconjugados y sus usos. |
| KR20090027227A (ko) | 2006-06-06 | 2009-03-16 | 제넨테크, 인크. | 항-dll4 항체 및 이의 사용 방법 |
| KR20190003862A (ko) | 2006-07-14 | 2019-01-09 | 에이씨 이뮨 에스.에이. | 아밀로이드 베타에 대해 인간화된 항체 |
| CA2657681C (en) | 2006-07-14 | 2019-03-19 | Ac Immune S.A. | Humanized antibodies against beta amyloid protein |
| EP2046809B1 (en) | 2006-07-19 | 2016-12-07 | The Trustees Of The University Of Pennsylvania | Wsx-1/il-27 as a target for anti-inflammatory responses |
| AR062223A1 (es) * | 2006-08-09 | 2008-10-22 | Glycart Biotechnology Ag | Moleculas de adhesion al antigeno que se adhieren a egfr, vectores que los codifican, y sus usos de estas |
| PL2383297T3 (pl) | 2006-08-14 | 2013-06-28 | Xencor Inc | Zoptymalizowane przeciwciała ukierunkowane na CD19 |
| MY161866A (en) | 2006-09-13 | 2017-05-15 | Abbvie Inc | Cell culture improvements |
| US8911964B2 (en) | 2006-09-13 | 2014-12-16 | Abbvie Inc. | Fed-batch method of making human anti-TNF-alpha antibody |
| WO2008036688A2 (en) * | 2006-09-18 | 2008-03-27 | Xencor, Inc. | Optimized antibodies that target hm1.24 |
| RU2486201C2 (ru) | 2006-10-12 | 2013-06-27 | Дженентек, Инк. | Антитела к лимфотоксину-альфа |
| LT2845866T (lt) | 2006-10-27 | 2017-07-10 | Genentech, Inc. | Antikūnai ir imunokonjugatai bei jų panaudojimas |
| CA2671457C (en) * | 2006-12-01 | 2017-09-26 | Medarex, Inc. | Human antibodies that bind cd22 and uses thereof |
| BRPI0806403A2 (pt) | 2007-02-09 | 2011-09-06 | Genentech Inc | anticorpo anti-robo4, usode um anticorpo e método de obtenção de imagem |
| PL2132573T3 (pl) | 2007-03-02 | 2014-09-30 | Genentech Inc | Prognozowanie odpowiedzi na inhibitor dimeryzacji HER oparte na niskiej ekspresji HER3 |
| CN101679934B (zh) * | 2007-03-07 | 2014-04-02 | 格利科菲公司 | 具有经修饰的岩藻糖基化的糖蛋白的生产 |
| US7960139B2 (en) | 2007-03-23 | 2011-06-14 | Academia Sinica | Alkynyl sugar analogs for the labeling and visualization of glycoconjugates in cells |
| EP2147108B1 (en) | 2007-04-17 | 2014-03-05 | Stichting Dienst Landbouwkundig Onderzoek | Mammalian-type glycosylation in plants by expression of non-mammalian glycosyltransferases |
| NO3072525T3 (zh) | 2007-05-14 | 2018-06-30 | ||
| ES2417148T3 (es) | 2007-06-08 | 2013-08-06 | Genentech, Inc. | Marcadores de expresión génica de la resistencia tumoral al tratamiento con inhibidor de HER2 |
| ES2529174T3 (es) | 2007-06-12 | 2015-02-17 | Ac Immune S.A. | Anticuerpos humanizados para amiloide beta |
| US7580304B2 (en) * | 2007-06-15 | 2009-08-25 | United Memories, Inc. | Multiple bus charge sharing |
| PL2155880T3 (pl) * | 2007-06-15 | 2017-06-30 | Medicago Inc. | Modyfikacja wytwarzania glikoproteiny w roślinach |
| SG10202005450PA (en) | 2007-07-09 | 2020-07-29 | Genentech Inc | Prevention of disulfide bond reduction during recombinant production of polypeptides |
| JP6126773B2 (ja) | 2007-09-04 | 2017-05-10 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | 癌のターゲッティングおよび検出のための高親和性抗前立腺幹細胞抗原(psca)抗体 |
| KR101922788B1 (ko) | 2007-09-26 | 2018-11-27 | 추가이 세이야쿠 가부시키가이샤 | 항체 정상영역 개변체 |
| ES2609918T3 (es) | 2007-10-05 | 2017-04-25 | Genentech, Inc. | Uso de anticuerpo anti-amiloide beta en enfermedades oculares |
| WO2009086320A1 (en) | 2007-12-26 | 2009-07-09 | Xencor, Inc | Fc variants with altered binding to fcrn |
| PT2514436T (pt) | 2007-11-07 | 2018-03-21 | Genentech Inc | Il-22 para utilização no tratamento de distúrbios microbianos |
| AR069501A1 (es) | 2007-11-30 | 2010-01-27 | Genentech Inc | Anticuerpos anti- vegf (factor de crecimiento endotelial vascular) |
| EP2231183A2 (en) * | 2007-12-21 | 2010-09-29 | Genentech, Inc. | Therapy of rituximab-refractory rheumatoid arthritis patients |
| AR069979A1 (es) * | 2007-12-26 | 2010-03-03 | Biotest Ag | Metodo para disminuir los efectos secundarios citotoxicos y mejorar la eficacia de los inmunoconjugados |
| CN101952315B (zh) * | 2007-12-26 | 2015-04-01 | 生物测试股份公司 | 靶向cd138的试剂及其应用 |
| KR101654822B1 (ko) | 2007-12-26 | 2016-09-06 | 바이오테스트 아게 | Cd138 표적성 면역접합체 및 이의 용도 |
| CA2710483C (en) * | 2007-12-26 | 2018-05-08 | Biotest Ag | Methods and agents for improving targeting of cd138 expressing tumor cells |
| TWI472339B (zh) | 2008-01-30 | 2015-02-11 | Genentech Inc | 包含結合至her2結構域ii之抗體及其酸性變異體的組合物 |
| US20090269339A1 (en) * | 2008-04-29 | 2009-10-29 | Genentech, Inc. | Responses to immunizations in rheumatoid arthritis patients treated with a cd20 antibody |
| KR101054362B1 (ko) * | 2008-07-03 | 2011-08-05 | 재단법인 목암생명공학연구소 | 재조합 단백질의 푸코스 함량을 감소시키는 방법 |
| DK2318832T3 (da) | 2008-07-15 | 2014-01-20 | Academia Sinica | Glycan-arrays på PTFE-lignende aluminiumcoatede objektglas og relaterede fremgangsmåder |
| TW201438738A (zh) | 2008-09-16 | 2014-10-16 | Genentech Inc | 治療進展型多發性硬化症之方法 |
| US8080415B2 (en) * | 2008-09-26 | 2011-12-20 | Eureka Therapeutics, Inc. | Modified host cells and uses thereof |
| SG10201605250SA (en) | 2008-10-14 | 2016-08-30 | Genentech Inc | Immunoglobulin variants and uses thereof |
| CA2932207A1 (en) | 2008-10-20 | 2010-12-09 | Abbvie Inc. | Isolation and purification of antibodies using protein a affinity chromatography |
| CA2738499A1 (en) | 2008-10-20 | 2010-04-29 | Abbott Laboratories | Viral inactivation during purification of antibodies |
| DK2752189T3 (en) | 2008-11-22 | 2017-01-16 | Hoffmann La Roche | APPLICATION OF ANTI-VEGF ANTIBODY IN COMBINATION WITH CHEMOTHERY TO TREAT CANCER CANCER |
| AR074777A1 (es) | 2008-12-19 | 2011-02-09 | Glaxo Group Ltd | Proteinas de union a antigeno |
| WO2010075249A2 (en) | 2008-12-22 | 2010-07-01 | Genentech, Inc. | A method for treating rheumatoid arthritis with b-cell antagonists |
| CN102264759B (zh) | 2008-12-23 | 2016-05-11 | 弗·哈夫曼-拉罗切有限公司 | 与蛋白a的结合改变的免疫球蛋白变体 |
| ES2712732T3 (es) | 2009-02-17 | 2019-05-14 | Cornell Res Foundation Inc | Métodos y kits para el diagnóstico de cáncer y la predicción de valor terapéutico |
| US9454640B2 (en) * | 2009-02-26 | 2016-09-27 | Intrexon Ceu, Inc. | Mammalian cell line models and related methods |
| US8597652B2 (en) | 2009-03-20 | 2013-12-03 | Genentech, Inc. | Multispecific anti-HER antibodies |
| BRPI1006270B1 (pt) | 2009-03-25 | 2022-08-16 | Genentech, Inc | Anticorpo anti-a5ss1, imunoconjugado, composição farmacêutica, método in vitro ou ex vivo para detectar a proteína a5ss1, uso de um anticorpo e kit para detectar a proteína a5ss1 |
| HRP20221471T1 (hr) | 2009-03-25 | 2023-02-03 | Genentech, Inc. | Protutijela anti-fgfr3 i postupci za njihovu uporabu |
| MX2011011684A (es) * | 2009-05-06 | 2012-01-20 | Biotest Ag | Usos de inmunoconjugados dirigidos a cd138. |
| WO2010138184A2 (en) | 2009-05-27 | 2010-12-02 | Synageva Biopharma Corp. | Avian derived antibodies |
| US20100316639A1 (en) | 2009-06-16 | 2010-12-16 | Genentech, Inc. | Biomarkers for igf-1r inhibitor therapy |
| US9676845B2 (en) | 2009-06-16 | 2017-06-13 | Hoffmann-La Roche, Inc. | Bispecific antigen binding proteins |
| WO2011014457A1 (en) | 2009-07-27 | 2011-02-03 | Genentech, Inc. | Combination treatments |
| US20110027275A1 (en) | 2009-07-31 | 2011-02-03 | Napoleone Ferrara | Inhibition of tumor metastasis |
| RU2017138926A (ru) | 2009-08-11 | 2019-02-11 | Дженентек, Инк. | Получение белков в культуральных средах без глутамина |
| AR077848A1 (es) | 2009-08-15 | 2011-09-28 | Genentech Inc | Terapia anti-angiogenesis para el tratamiento de cancer de mama previamente tratado |
| TWI412375B (zh) * | 2009-08-28 | 2013-10-21 | Roche Glycart Ag | 人類化抗cdcp1抗體 |
| SG178567A1 (en) | 2009-08-31 | 2012-04-27 | Roche Glycart Ag | Affinity-matured humanized anti cea monoclonal antibodies |
| WO2011028952A1 (en) | 2009-09-02 | 2011-03-10 | Xencor, Inc. | Compositions and methods for simultaneous bivalent and monovalent co-engagement of antigens |
| WO2011028950A1 (en) | 2009-09-02 | 2011-03-10 | Genentech, Inc. | Mutant smoothened and methods of using the same |
| JP5814925B2 (ja) | 2009-10-22 | 2015-11-17 | ジェネンテック, インコーポレイテッド | 抗ヘプシン抗体及びその使用方法 |
| WO2011056494A1 (en) | 2009-10-26 | 2011-05-12 | Genentech, Inc. | Activin receptor-like kinase-1 antagonist and vegfr3 antagonist combinations |
| WO2011056502A1 (en) | 2009-10-26 | 2011-05-12 | Genentech, Inc. | Bone morphogenetic protein receptor type ii compositions and methods of use |
| WO2011056497A1 (en) | 2009-10-26 | 2011-05-12 | Genentech, Inc. | Activin receptor type iib compositions and methods of use |
| WO2011056997A1 (en) | 2009-11-04 | 2011-05-12 | Fabrus Llc | Methods for affinity maturation-based antibody optimization |
| CA2780143A1 (en) | 2009-11-05 | 2011-05-12 | Genentech, Inc. | Methods and composition for secretion of heterologous polypeptides |
| US11377485B2 (en) | 2009-12-02 | 2022-07-05 | Academia Sinica | Methods for modifying human antibodies by glycan engineering |
| RU2673908C2 (ru) | 2009-12-02 | 2018-12-03 | Имэджинэб, Инк. | Мини-антитела j591 и цис-диатела для направленной доставки простата-специфичного мембранного антигена (psma) человека и способы их применения |
| US10087236B2 (en) | 2009-12-02 | 2018-10-02 | Academia Sinica | Methods for modifying human antibodies by glycan engineering |
| WO2011071577A1 (en) | 2009-12-11 | 2011-06-16 | Genentech, Inc. | Anti-vegf-c antibodies and methods using same |
| DK2516465T3 (en) | 2009-12-23 | 2016-06-06 | Hoffmann La Roche | ANTI-BV8 ANTIBODIES AND APPLICATIONS THEREOF |
| WO2011091078A2 (en) | 2010-01-19 | 2011-07-28 | Xencor, Inc. | Antibody fc variants with enhanced complement activity |
| AR080027A1 (es) | 2010-01-28 | 2012-03-07 | Glaxo Group Ltd | Proteinas de union a cd127 |
| MX2012009167A (es) | 2010-02-09 | 2012-08-23 | Glaxo Group Ltd | Tratamiento de un trastorno metabolico. |
| JP5841072B2 (ja) | 2010-02-10 | 2016-01-06 | イミュノジェン・インコーポレーテッド | Cd20抗体およびその使用 |
| SG183333A1 (en) | 2010-02-18 | 2012-09-27 | Genentech Inc | Neuregulin antagonists and use thereof in treating cancer |
| US20110200595A1 (en) | 2010-02-18 | 2011-08-18 | Roche Glycart | TREATMENT WITH A HUMANIZED IgG CLASS ANTI EGFR ANTIBODY AND AN ANTIBODY AGAINST INSULIN LIKE GROWTH FACTOR 1 RECEPTOR |
| EP2539367A2 (en) | 2010-02-23 | 2013-01-02 | F. Hoffmann-La Roche AG | Anti-angiogenesis therapy for the treatment of ovarian cancer |
| UA108227C2 (xx) | 2010-03-03 | 2015-04-10 | Антигензв'язуючий білок | |
| EP2550295A1 (en) | 2010-03-24 | 2013-01-30 | F. Hoffmann-La Roche AG | Anti-lrp6 antibodies |
| WO2011124635A1 (en) | 2010-04-07 | 2011-10-13 | Humalys | Binding molecules against chikungunya virus and uses thereof |
| EP2374816B1 (en) | 2010-04-07 | 2016-09-28 | Agency For Science, Technology And Research | Binding molecules against Chikungunya virus and uses thereof |
| US10338069B2 (en) | 2010-04-12 | 2019-07-02 | Academia Sinica | Glycan arrays for high throughput screening of viruses |
| CN104777301B (zh) | 2010-05-10 | 2018-04-20 | 中央研究院 | 具有抗流感活性的扎那米韦膦酸酯同类物及流感病毒的奥司他韦易感性的测定 |
| WO2011146568A1 (en) | 2010-05-19 | 2011-11-24 | Genentech, Inc. | Predicting response to a her inhibitor |
| WO2011147834A1 (en) | 2010-05-26 | 2011-12-01 | Roche Glycart Ag | Antibodies against cd19 and uses thereof |
| WO2011153243A2 (en) | 2010-06-02 | 2011-12-08 | Genentech, Inc. | Anti-angiogenesis therapy for treating gastric cancer |
| AR081556A1 (es) | 2010-06-03 | 2012-10-03 | Glaxo Group Ltd | Proteinas de union al antigeno humanizadas |
| CA2795972A1 (en) | 2010-06-03 | 2011-12-08 | Genentech, Inc. | Immuno-pet imaging of antibodies and immunoconjugates and uses therefor |
| MX336001B (es) | 2010-06-18 | 2016-01-07 | Genentech Inc | Anticuerpos anti-axl y metodos de uso. |
| WO2011161119A1 (en) | 2010-06-22 | 2011-12-29 | F. Hoffmann-La Roche Ag | Antibodies against insulin-like growth factor i receptor and uses thereof |
| WO2011161189A1 (en) | 2010-06-24 | 2011-12-29 | F. Hoffmann-La Roche Ag | Anti-hepsin antibodies and methods of use |
| AU2011274528B2 (en) | 2010-07-09 | 2015-04-23 | Genentech, Inc. | Anti-neuropilin antibodies and methods of use |
| EP2409993A1 (en) | 2010-07-19 | 2012-01-25 | International-Drug-Development-Biotech | Anti-CD19 antibody having ADCC function with improved glycosylation profile |
| EP2409989A1 (en) | 2010-07-19 | 2012-01-25 | International-Drug-Development-Biotech | Method to improve glycosylation profile for antibody |
| EP2409712A1 (en) | 2010-07-19 | 2012-01-25 | International-Drug-Development-Biotech | Anti-CD19 antibody having ADCC and CDC functions and improved glycosylation profile |
| WO2012010582A1 (en) | 2010-07-21 | 2012-01-26 | Roche Glycart Ag | Anti-cxcr5 antibodies and methods of use |
| WO2012012750A1 (en) | 2010-07-23 | 2012-01-26 | Trustees Of Boston University | ANTI-DEsupR INHIBITORS AS THERAPEUTICS FOR INHIBITION OF PATHOLOGICAL ANGIOGENESIS AND TUMOR CELL INVASIVENESS AND FOR MOLECULAR IMAGING AND TARGETED DELIVERY |
| JP2013541501A (ja) | 2010-08-03 | 2013-11-14 | エフ・ホフマン−ラ・ロシュ・アクチェンゲゼルシャフト | 慢性リンパ性白血病(cll)のバイオマーカー |
| CA2805564A1 (en) | 2010-08-05 | 2012-02-09 | Stefan Jenewein | Anti-mhc antibody anti-viral cytokine fusion protein |
| NZ703653A (en) | 2010-08-13 | 2016-09-30 | Roche Glycart Ag | Anti-fap antibodies and methods of use |
| KR101653030B1 (ko) | 2010-08-13 | 2016-08-31 | 로슈 글리카트 아게 | 항-테나신-c a2 항체 및 이의 사용 방법 |
| JP5813114B2 (ja) | 2010-08-25 | 2015-11-17 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | Il−18r1に対する抗体およびその使用 |
| CA2808236A1 (en) | 2010-08-31 | 2012-03-08 | Genentech, Inc. | Biomarkers and methods of treatment |
| AU2011312205B2 (en) | 2010-10-05 | 2015-08-13 | Curis, Inc. | Mutant smoothened and methods of using the same |
| JP2013541594A (ja) | 2010-11-08 | 2013-11-14 | ジェネンテック, インコーポレイテッド | 皮下投与される抗il−6受容体抗体 |
| PE20140238A1 (es) | 2010-11-10 | 2014-03-07 | Genentech Inc | Metodos y composiciones para inmunoterapia para enfermedad neural |
| LT2643352T (lt) | 2010-11-23 | 2018-08-10 | Glaxo Group Limited | Antigeną onkostatiną m (osm) surišantys baltymai |
| US20130236467A1 (en) | 2010-11-24 | 2013-09-12 | Jeremy Griggs | Multispecific antigen binding proteins targeting hgf |
| CN110251668A (zh) | 2010-11-30 | 2019-09-20 | 霍夫曼-拉罗奇有限公司 | 低亲和力血脑屏障受体抗体及其用途 |
| MY188365A (en) | 2010-12-16 | 2021-12-06 | Genentech Inc | Diagnosis and treatments relating to th2 inhibition |
| UA115641C2 (uk) | 2010-12-20 | 2017-11-27 | Дженентек, Інк. | Виділене антитіло, яке зв'язує мезотелін, та імунокон'югат, що його містить |
| KR20130118925A (ko) | 2010-12-22 | 2013-10-30 | 제넨테크, 인크. | 항-pcsk9 항체 및 사용 방법 |
| WO2012092539A2 (en) | 2010-12-31 | 2012-07-05 | Takeda Pharmaceutical Company Limited | Antibodies to dll4 and uses thereof |
| EP2482074A1 (en) * | 2011-01-27 | 2012-08-01 | Medizinische Hochschule Hannover | Methods and means for diagnosing vasculitis |
| TWI666027B (zh) | 2011-02-10 | 2019-07-21 | 羅齊克雷雅公司 | 突變介白素-2多肽 |
| MX2013009151A (es) | 2011-02-10 | 2013-08-29 | Roche Glycart Ag | Inmunoterapia mejorada. |
| CA2824824A1 (en) | 2011-02-28 | 2012-09-07 | F. Hoffmann-La Roche Ag | Monovalent antigen binding proteins |
| CA2825081A1 (en) | 2011-02-28 | 2012-09-07 | Birgit Bossenmaier | Antigen binding proteins |
| US8642742B2 (en) | 2011-03-02 | 2014-02-04 | Roche Glycart Ag | Anti-CEA antibodies |
| KR20140021589A (ko) | 2011-04-07 | 2014-02-20 | 제넨테크, 인크. | 항-fgfr4 항체 및 사용 방법 |
| US9062106B2 (en) | 2011-04-27 | 2015-06-23 | Abbvie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| EA201892619A1 (ru) | 2011-04-29 | 2019-04-30 | Роше Гликарт Аг | Иммуноконъюгаты, содержащие мутантные полипептиды интерлейкина-2 |
| WO2012146630A1 (en) | 2011-04-29 | 2012-11-01 | F. Hoffmann-La Roche Ag | N-terminal acylated polypeptides, methods for their production and uses thereof |
| WO2012155019A1 (en) | 2011-05-12 | 2012-11-15 | Genentech, Inc. | Multiple reaction monitoring lc-ms/ms method to detect therapeutic antibodies in animal samples using framework signature pepides |
| LT2710035T (lt) | 2011-05-16 | 2017-06-26 | F. Hoffmann-La Roche Ag | Fgfr1 agonistai ir jų naudojimo būdai |
| CN106279418A (zh) | 2011-05-27 | 2017-01-04 | 葛兰素集团有限公司 | Bcma(cd269/tnfrsf17)结合蛋白 |
| EP2721067B1 (en) | 2011-06-15 | 2019-07-31 | F.Hoffmann-La Roche Ag | Anti-human epo receptor antibodies and methods of use |
| EA201400046A1 (ru) | 2011-06-22 | 2014-07-30 | Ф. Хоффманн-Ля Рош Аг | Удаление клеток-мишеней с помощью циркулирующих вирусспецифических цитотоксических т-клеток с использованием содержащих гкгс класса i комплексов |
| CN107090038A (zh) | 2011-06-30 | 2017-08-25 | 霍夫曼-拉罗奇有限公司 | 抗c‑met抗体配制剂 |
| US20130022551A1 (en) | 2011-07-22 | 2013-01-24 | Trustees Of Boston University | DEspR ANTAGONISTS AND AGONISTS AS THERAPEUTICS |
| EA028886B1 (ru) | 2011-07-27 | 2018-01-31 | Глаксо Груп Лимитед | ОТДЕЛЬНЫЕ ВАРИАБЕЛЬНЫЕ ДОМЕНЫ ПРОТИВ VEGF, СЛИТЫЕ С Fc-ДОМЕНАМИ |
| MX2014001736A (es) | 2011-08-17 | 2014-03-31 | Genentech Inc | Inhibicion de angiogenesis en tumores refractarios. |
| MX2014001766A (es) | 2011-08-17 | 2014-05-01 | Genentech Inc | Anticuerpos de neuregulina y sus usos. |
| CA2844141A1 (en) | 2011-08-23 | 2013-02-28 | Roche Glycart Ag | Anti-mcsp antibodies |
| WO2013026831A1 (en) | 2011-08-23 | 2013-02-28 | Roche Glycart Ag | Bispecific antigen binding molecules |
| WO2013040433A1 (en) | 2011-09-15 | 2013-03-21 | Genentech, Inc. | Methods of promoting differentiation |
| AR087918A1 (es) | 2011-09-19 | 2014-04-23 | Genentech Inc | Tratamientos combinados que comprenden antagonistas de c-met y antagonistas de b-raf |
| AU2012319150B2 (en) | 2011-10-05 | 2017-08-17 | Genentech, Inc. | Methods of treating liver conditions using Notch2 antagonists |
| ES2687951T3 (es) | 2011-10-14 | 2018-10-30 | F. Hoffmann-La Roche Ag | Anticuerpos anti-HtrA1 y procedimientos de uso |
| BR112014009178A2 (pt) | 2011-10-19 | 2018-09-04 | Roche Glycart Ag | métodos, uso, receptor e invenção |
| WO2013059531A1 (en) | 2011-10-20 | 2013-04-25 | Genentech, Inc. | Anti-gcgr antibodies and uses thereof |
| IN2014CN03062A (zh) | 2011-10-28 | 2015-07-31 | Hoffmann La Roche | |
| RU2014124842A (ru) | 2011-11-21 | 2015-12-27 | Дженентек, Инк. | Очистка анти-с-мет антител |
| EP2788024A1 (en) | 2011-12-06 | 2014-10-15 | F.Hoffmann-La Roche Ag | Antibody formulation |
| MX358680B (es) | 2011-12-08 | 2018-08-31 | Biotest Ag | Usos de inmunoconjugados dirigidos a cd138. |
| CN113896787A (zh) | 2011-12-22 | 2022-01-07 | 弗·哈夫曼-拉罗切有限公司 | 表达载体元件组合、新的生产用细胞产生方法及其在重组产生多肽中的用途 |
| SG11201403223PA (en) | 2011-12-22 | 2014-07-30 | Hoffmann La Roche | Expression vector organization, novel production cell generation methods and their use for the recombinant production of polypeptides |
| MX2014007262A (es) | 2011-12-22 | 2014-08-01 | Hoffmann La Roche | Sistema de exhibicion de anticuerpos de longitud completa para celulas eucarioticas y su uso. |
| WO2013096791A1 (en) | 2011-12-23 | 2013-06-27 | Genentech, Inc. | Process for making high concentration protein formulations |
| WO2013101771A2 (en) | 2011-12-30 | 2013-07-04 | Genentech, Inc. | Compositions and method for treating autoimmune diseases |
| CN104411717A (zh) | 2012-01-09 | 2015-03-11 | 斯克利普斯研究所 | 具有超长cdr3s的人源化抗体 |
| US10774132B2 (en) | 2012-01-09 | 2020-09-15 | The Scripps Research Instittue | Ultralong complementarity determining regions and uses thereof |
| PE20141561A1 (es) | 2012-01-18 | 2014-11-12 | Genentech Inc | Anticuerpos anti-lrp5 y metodos de uso |
| EP2804630B1 (en) | 2012-01-18 | 2017-10-18 | F. Hoffmann-La Roche AG | Methods of using fgf19 modulators |
| WO2013113641A1 (en) | 2012-01-31 | 2013-08-08 | Roche Glycart Ag | Use of nkp46 as a predictive biomarker for cancer treatment with adcc- enhanced antibodies |
| EP2812350B1 (en) | 2012-02-11 | 2019-04-03 | F.Hoffmann-La Roche Ag | R-spondin translocations and methods using the same |
| TR201808458T4 (tr) | 2012-02-15 | 2018-07-23 | Hoffmann La Roche | FC-reseptör bazlı afinite kromatografisi. |
| BR112014018374A8 (pt) | 2012-03-02 | 2017-07-11 | Roche Glycart Ag | Método para predizer a resposta d eum paciente com câncer, kit, anticorpo, método para o tratamento do câncer e composição farmacêutica |
| SG11201406079TA (en) | 2012-03-27 | 2014-10-30 | Genentech Inc | Diagnosis and treatments relating to her3 inhibitors |
| AR090549A1 (es) | 2012-03-30 | 2014-11-19 | Genentech Inc | Anticuerpos anti-lgr5 e inmunoconjugados |
| US10130714B2 (en) | 2012-04-14 | 2018-11-20 | Academia Sinica | Enhanced anti-influenza agents conjugated with anti-inflammatory activity |
| US9067990B2 (en) | 2013-03-14 | 2015-06-30 | Abbvie, Inc. | Protein purification using displacement chromatography |
| WO2013158279A1 (en) | 2012-04-20 | 2013-10-24 | Abbvie Inc. | Protein purification methods to reduce acidic species |
| WO2013158273A1 (en) | 2012-04-20 | 2013-10-24 | Abbvie Inc. | Methods to modulate c-terminal lysine variant distribution |
| KR20150006000A (ko) | 2012-05-01 | 2015-01-15 | 제넨테크, 인크. | 항-pmel17 항체 및 면역접합체 |
| WO2013170191A1 (en) | 2012-05-11 | 2013-11-14 | Genentech, Inc. | Methods of using antagonists of nad biosynthesis from nicotinamide |
| CA2873646C (en) | 2012-05-18 | 2022-04-26 | Genentech, Inc. | High-concentration monoclonal antibody formulations |
| BR112014028838A2 (pt) | 2012-05-21 | 2020-05-12 | Genentech Inc | Usos de um anticorpo, método para fazer um anticorpo e anticorpos |
| DK2852610T3 (en) | 2012-05-23 | 2018-09-03 | Glykos Finland Oy | PRODUCTION OF FUCOSYLED GLYCOPROTEIN |
| WO2013177470A1 (en) | 2012-05-23 | 2013-11-28 | Genentech, Inc. | Selection method for therapeutic agents |
| WO2013176754A1 (en) | 2012-05-24 | 2013-11-28 | Abbvie Inc. | Novel purification of antibodies using hydrophobic interaction chromatography |
| CN103463633B (zh) * | 2012-06-07 | 2016-03-30 | 复旦大学 | 一种靶向的嵌合乙肝核心抗原治疗性疫苗及其用途 |
| EP2861624A1 (en) | 2012-06-15 | 2015-04-22 | F. Hoffmann-La Roche AG | Anti-pcsk9 antibodies, formulations, dosing, and methods of use |
| WO2014004549A2 (en) | 2012-06-27 | 2014-01-03 | Amgen Inc. | Anti-mesothelin binding proteins |
| PL2869837T3 (pl) | 2012-07-04 | 2017-03-31 | F.Hoffmann-La Roche Ag | Przeciwciała przeciw teofilinie i sposoby ich stosowania |
| EP3138580B1 (en) | 2012-07-04 | 2021-03-03 | F. Hoffmann-La Roche AG | Covalently linked antigen-antibody conjugates |
| KR20150030755A (ko) | 2012-07-04 | 2015-03-20 | 에프. 호프만-라 로슈 아게 | 항-바이오틴 항체 및 사용 방법 |
| JP6309518B2 (ja) | 2012-07-05 | 2018-04-11 | ジェネンテック, インコーポレイテッド | 発現及び分泌システム |
| MX2015000314A (es) | 2012-07-09 | 2015-04-10 | Genentech Inc | Anticuerpos anti-cd79b e inmunoconjugados. |
| EP2869849A1 (en) | 2012-07-09 | 2015-05-13 | Genentech, Inc. | Immunoconjugates comprising anti-cd22 antibodies |
| KR20150030753A (ko) | 2012-07-09 | 2015-03-20 | 제넨테크, 인크. | 항-cd79b 항체를 포함하는 면역접합체 |
| EA201590173A1 (ru) | 2012-07-09 | 2015-09-30 | Дженентек, Инк. | Иммуноконъюгаты, содержащие антитела к cd22 |
| SI3495387T1 (sl) | 2012-07-13 | 2021-12-31 | Roche Glycart Ag | Bispecifična protitelesa proti VEGF/proti ANG-2 in njihova uporaba pri zdravljenju bolezni očesnih žil |
| CN110256559B (zh) | 2012-07-25 | 2023-05-26 | 塞尔德克斯医疗公司 | 抗kit抗体及其用途 |
| MY175687A (en) | 2012-08-07 | 2020-07-06 | Roche Glycart Ag | Composition comprising two antibodies engineered to have reduced and increased effector function |
| WO2014031498A1 (en) | 2012-08-18 | 2014-02-27 | Academia Sinica | Cell-permeable probes for identification and imaging of sialidases |
| US9547009B2 (en) | 2012-08-21 | 2017-01-17 | Academia Sinica | Benzocyclooctyne compounds and uses thereof |
| WO2014029752A1 (en) | 2012-08-22 | 2014-02-27 | Glaxo Group Limited | Anti lrp6 antibodies |
| KR20190121874A (ko) | 2012-08-29 | 2019-10-28 | 에프. 호프만-라 로슈 아게 | 혈액 뇌 장벽 셔틀 |
| US9512214B2 (en) | 2012-09-02 | 2016-12-06 | Abbvie, Inc. | Methods to control protein heterogeneity |
| EP2890782A1 (en) | 2012-09-02 | 2015-07-08 | AbbVie Inc. | Methods to control protein heterogeneity |
| RU2648476C2 (ru) | 2012-09-07 | 2018-03-26 | Дженентек, Инк. | Комбинированная терапия с использованием антитела против cd20 типа ii и селективного ингибитора bcl-2 |
| LT2839860T (lt) | 2012-10-12 | 2019-07-10 | Medimmune Limited | Pirolobenzodiazepinai ir jų konjugatai |
| US10100102B2 (en) | 2012-10-29 | 2018-10-16 | The University Of North Carolina At Chapel Hill | Compositions and methods for inhibiting pathogen infection |
| CA2890207A1 (en) | 2012-11-05 | 2014-05-08 | Foundation Medicine, Inc. | Novel ntrk1 fusion molecules and uses thereof |
| PE20150956A1 (es) | 2012-11-08 | 2015-06-20 | Hoffmann La Roche | Proteinas ligantes de antigeno her3 de union a la horquilla beta de her3 |
| EA201590731A1 (ru) | 2012-11-13 | 2015-11-30 | Дженентек, Инк. | Антитела к гемагглютинину и способы применения |
| JP6475167B2 (ja) | 2012-12-21 | 2019-02-27 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | ジスルフィド結合した多価mhcクラスiを含む多機能タンパク質 |
| CA2898326C (en) | 2013-01-18 | 2022-05-17 | Foundation Medicine, Inc. | Methods of treating cholangiocarcinoma |
| WO2014114595A1 (en) | 2013-01-23 | 2014-07-31 | Roche Glycart Ag | Predictive biomarker for cancer treatment with adcc-enhanced antibodies |
| WO2014116749A1 (en) | 2013-01-23 | 2014-07-31 | Genentech, Inc. | Anti-hcv antibodies and methods of using thereof |
| TW201446962A (zh) | 2013-02-13 | 2014-12-16 | Lab Francais Du Fractionnement | 具有修飾的糖化作用之蛋白質及其製造方法 |
| CN105308068A (zh) | 2013-02-13 | 2016-02-03 | 法国化学与生物科技实验室 | 高度半乳糖基化的抗TNF-α抗体及其用途 |
| CN104994879A (zh) | 2013-02-22 | 2015-10-21 | 霍夫曼-拉罗奇有限公司 | 治疗癌症和预防药物抗性的方法 |
| CA2896259A1 (en) | 2013-02-26 | 2014-09-04 | Roche Glycart Ag | Anti-mcsp antibodies |
| CA2902263A1 (en) | 2013-03-06 | 2014-09-12 | Genentech, Inc. | Methods of treating and preventing cancer drug resistance |
| AU2013381687A1 (en) | 2013-03-12 | 2015-09-24 | Abbvie Inc. | Human antibodies that bind human TNF-alpha and methods of preparing the same |
| US20160031887A1 (en) | 2013-03-13 | 2016-02-04 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| AU2014239903A1 (en) | 2013-03-14 | 2015-09-17 | Genentech, Inc. | Combinations of a MEK inhibitor compound with an HER3/EGFR inhibitor compound and methods of use |
| RU2015139054A (ru) | 2013-03-14 | 2017-04-19 | Дженентек, Инк. | Способы лечения рака и профилактики лекарственной резистентности рака |
| US9499614B2 (en) | 2013-03-14 | 2016-11-22 | Abbvie Inc. | Methods for modulating protein glycosylation profiles of recombinant protein therapeutics using monosaccharides and oligosaccharides |
| US9017687B1 (en) | 2013-10-18 | 2015-04-28 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same using displacement chromatography |
| US9562099B2 (en) | 2013-03-14 | 2017-02-07 | Genentech, Inc. | Anti-B7-H4 antibodies and immunoconjugates |
| US8921526B2 (en) | 2013-03-14 | 2014-12-30 | Abbvie, Inc. | Mutated anti-TNFα antibodies and methods of their use |
| KR20150127199A (ko) | 2013-03-14 | 2015-11-16 | 제넨테크, 인크. | 항-b7-h4 항체 및 면역접합체 |
| EP2968537A1 (en) | 2013-03-15 | 2016-01-20 | Genentech, Inc. | Methods of treating cancer and preventing cancer drug resistance |
| US9598485B2 (en) | 2013-03-15 | 2017-03-21 | Ac Immune S.A. | Anti-tau antibodies and methods of use |
| MX2015012326A (es) | 2013-03-15 | 2016-03-08 | Genentech Inc | Anticuerpos anti-crth2 y su uso. |
| SG11201507333XA (en) | 2013-03-15 | 2015-10-29 | Genentech Inc | Biomarkers and methods of treating pd-1 and pd-l1 related conditions |
| SI2970422T1 (en) | 2013-03-15 | 2018-07-31 | F. Hoffmann-La Roche Ag | IL-22 polypeptides and IL-22-Fc fusion proteins and procedures for use |
| KR20150128707A (ko) | 2013-03-15 | 2015-11-18 | 제넨테크, 인크. | 간암의 진단 및 치료를 위한 조성물 및 방법 |
| ME03796B (me) | 2013-03-15 | 2021-04-20 | Glaxosmithkline Ip Dev Ltd | Anti-lag-3 vezujući proteini |
| UA118028C2 (uk) | 2013-04-03 | 2018-11-12 | Рош Глікарт Аг | Біспецифічне антитіло, специфічне щодо fap і dr5, антитіло, специфічне щодо dr5, і спосіб їх застосування |
| EP3327034A1 (en) | 2013-04-29 | 2018-05-30 | F. Hoffmann-La Roche AG | Fcrn-binding abolished anti-igf-1r antibodies and their use in the treatment of vascular eye diseases |
| MX2015015060A (es) | 2013-04-29 | 2016-02-25 | Hoffmann La Roche | Anticuerpos asimetricos modificados que se unen al receptor fc y metodos de uso. |
| TWI747803B (zh) | 2013-04-29 | 2021-12-01 | 瑞士商赫孚孟拉羅股份公司 | 人類結合fcrn之經修飾抗體及使用方法 |
| PL3594240T3 (pl) | 2013-05-20 | 2024-04-02 | F. Hoffmann-La Roche Ag | Przeciwciała przeciwko receptorowi transferyny i sposoby ich zastosowania |
| WO2014210397A1 (en) | 2013-06-26 | 2014-12-31 | Academia Sinica | Rm2 antigens and use thereof |
| EP3013347B1 (en) | 2013-06-27 | 2019-12-11 | Academia Sinica | Glycan conjugates and use thereof |
| CN111518199A (zh) | 2013-07-18 | 2020-08-11 | 图鲁斯生物科学有限责任公司 | 具有超长互补决定区的人源化抗体 |
| US20160168231A1 (en) | 2013-07-18 | 2016-06-16 | Fabrus, Inc. | Antibodies with ultralong complementarity determining regions |
| MX2016001236A (es) | 2013-08-01 | 2016-05-24 | Five Prime Therapeutics Inc | Anticuerpos anti-fgfr2iiib afucosilados. |
| SG11201601044XA (en) | 2013-08-12 | 2016-03-30 | Genentech Inc | Compositions and method for treating complement-associated conditions |
| JP6486368B2 (ja) | 2013-09-06 | 2019-03-20 | アカデミア シニカAcademia Sinica | 改変されたグリコシル基を含む糖脂質を用いたヒトiNKT細胞の活性化 |
| CA2922889A1 (en) | 2013-09-17 | 2015-03-26 | Genentech, Inc. | Methods of using anti-lgr5 antibodies |
| EP3052640A2 (en) | 2013-10-04 | 2016-08-10 | AbbVie Inc. | Use of metal ions for modulation of protein glycosylation profiles of recombinant proteins |
| KR20160044060A (ko) | 2013-10-11 | 2016-04-22 | 에프. 호프만-라 로슈 아게 | 다중특이적 도메인 교환된 통상의 가변 경쇄 항체 |
| JP2016537965A (ja) | 2013-10-11 | 2016-12-08 | ジェネンテック, インコーポレイテッド | Nsp4阻害剤及び使用方法 |
| MX2016003744A (es) | 2013-10-11 | 2016-08-11 | Us Health | Anticuerpos tem8 y su uso. |
| US9181337B2 (en) | 2013-10-18 | 2015-11-10 | Abbvie, Inc. | Modulated lysine variant species compositions and methods for producing and using the same |
| CA2925598A1 (en) | 2013-10-18 | 2015-04-23 | Genentech, Inc. | Anti-rspo antibodies and methods of use |
| US9085618B2 (en) | 2013-10-18 | 2015-07-21 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US8946395B1 (en) | 2013-10-18 | 2015-02-03 | Abbvie Inc. | Purification of proteins using hydrophobic interaction chromatography |
| MX2016005159A (es) | 2013-10-23 | 2016-07-05 | Genentech Inc | Metodos de diagnostico y tratamiento de trastornos eosinofilicos. |
| WO2015073884A2 (en) | 2013-11-15 | 2015-05-21 | Abbvie, Inc. | Glycoengineered binding protein compositions |
| RU2697098C1 (ru) | 2013-11-21 | 2019-08-12 | Ф.Хоффманн-Ля Рош Аг | Антитела к альфа-синуклеину и способы применения |
| SG10201808835QA (en) | 2013-12-09 | 2018-11-29 | Allakos Inc | Anti-siglec-8 antibodies and methods of use thereof |
| MX2016007576A (es) | 2013-12-13 | 2016-10-03 | Genentech Inc | Anticuerpos e inmunoconjugados anti-cd33. |
| US20160333063A1 (en) | 2013-12-13 | 2016-11-17 | The General Hospital Corporation | Soluble high molecular weight (hmw) tau species and applications thereof |
| EP3527587A1 (en) | 2013-12-17 | 2019-08-21 | F. Hoffmann-La Roche AG | Combination therapy comprising ox40 binding agonists and pd-l1 binding antagonists |
| RU2719487C2 (ru) | 2013-12-17 | 2020-04-17 | Дженентек, Инк. | Способы лечения рака с использованием антагонистов, связывающихся с компонентами сигнального пути pd-1, и таксанов |
| CN105899535A (zh) | 2013-12-17 | 2016-08-24 | 豪夫迈·罗氏有限公司 | 用pd-1轴结合拮抗剂和抗cd20抗体治疗癌症的方法 |
| PE20210107A1 (es) | 2013-12-17 | 2021-01-19 | Genentech Inc | Anticuerpos anti-cd3 y metodos de uso |
| TWI728373B (zh) | 2013-12-23 | 2021-05-21 | 美商建南德克公司 | 抗體及使用方法 |
| JP6554473B2 (ja) | 2013-12-24 | 2019-07-31 | アルゲン−エックス ビーブイビーエー | FcRnアンタゴニスト及び使用方法 |
| CN105873616B (zh) | 2014-01-03 | 2020-06-05 | 豪夫迈·罗氏有限公司 | 共价连接的多肽毒素-抗体缀合物 |
| CN105873615B (zh) | 2014-01-03 | 2020-12-25 | 豪夫迈·罗氏有限公司 | 共价连接的helicar-抗helicar抗体缀合物及其用途 |
| JP6476194B2 (ja) | 2014-01-03 | 2019-02-27 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | 二重特異性抗ハプテン/抗血液脳関門受容体抗体、それらの複合体、及び血液脳関門シャトルとしてのそれらの使用 |
| WO2015103549A1 (en) | 2014-01-03 | 2015-07-09 | The United States Of America, As Represented By The Secretary Department Of Health And Human Services | Neutralizing antibodies to hiv-1 env and their use |
| MX2016008190A (es) | 2014-01-06 | 2016-10-21 | Hoffmann La Roche | Modulos de lanzadera de barrera cerebral sanguinea monovalente. |
| KR20160107190A (ko) | 2014-01-15 | 2016-09-13 | 에프. 호프만-라 로슈 아게 | 변형된 FcRn-결합 및 유지된 단백질 A-결합 성질을 갖는 Fc-영역 변이체 |
| US10150818B2 (en) | 2014-01-16 | 2018-12-11 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| CA2937123A1 (en) | 2014-01-16 | 2015-07-23 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| US20170043034A1 (en) | 2014-01-24 | 2017-02-16 | Genentech, Inc. | Methods of using anti-steap1 antibodies and immunoconjugates |
| CA2937539A1 (en) | 2014-02-04 | 2015-08-13 | Genentech, Inc. | Mutant smoothened and methods of using the same |
| AU2015214058B2 (en) | 2014-02-08 | 2020-07-09 | Genentech, Inc. | Methods of treating Alzheimer's Disease |
| EP3102230B1 (en) | 2014-02-08 | 2021-04-28 | F. Hoffmann-La Roche AG | Methods of treating alzheimer's disease |
| ES2685424T3 (es) | 2014-02-12 | 2018-10-09 | F. Hoffmann-La Roche Ag | Anticuerpos anti-Jagged1 y procedimientos de uso |
| WO2015127405A2 (en) | 2014-02-21 | 2015-08-27 | Genentech, Inc. | Anti-il-13/il-17 bispecific antibodies and uses thereof |
| EP3110446B1 (en) | 2014-02-28 | 2021-12-01 | Allakos Inc. | Methods and compositions for treating siglec-8 associated diseases |
| CN106103730B (zh) | 2014-03-14 | 2021-06-08 | 豪夫迈·罗氏有限公司 | 用于分泌异源多肽的方法和组合物 |
| WO2015140591A1 (en) | 2014-03-21 | 2015-09-24 | Nordlandssykehuset Hf | Anti-cd14 antibodies and uses thereof |
| BR112016021383A2 (pt) | 2014-03-24 | 2017-10-03 | Genentech Inc | Método para identificar um paciente com câncer que é susceptível ou menos susceptível a responder ao tratamento com um antagonista de cmet, método para identificar um paciente apresentando câncer previamente tratado, método para determinar a expressão do biomarcador hgf, antagonista anti-c-met e seu uso, kit de diagnóstico e seu método de preparo |
| WO2015148915A1 (en) | 2014-03-27 | 2015-10-01 | Academia Sinica | Reactive labelling compounds and uses thereof |
| UA121112C2 (uk) | 2014-03-31 | 2020-04-10 | Дженентек, Інк. | Антитіло до ox40 та його застосування |
| JP6588461B2 (ja) | 2014-03-31 | 2019-10-09 | ジェネンテック, インコーポレイテッド | 抗血管新生剤及びox40結合アゴニストを含む併用療法 |
| WO2015164615A1 (en) | 2014-04-24 | 2015-10-29 | University Of Oslo | Anti-gluten antibodies and uses thereof |
| KR20170003582A (ko) | 2014-05-22 | 2017-01-09 | 제넨테크, 인크. | 항-gpc3 항체 및 면역접합체 |
| JP2017524371A (ja) | 2014-05-23 | 2017-08-31 | ジェネンテック, インコーポレイテッド | Mitバイオマーカーとその使用方法 |
| JP6894239B2 (ja) | 2014-05-27 | 2021-06-30 | アカデミア シニカAcademia Sinica | 増強された抗体の有効性のための普遍的グリコフォームに関する組成物および方法 |
| US10005847B2 (en) | 2014-05-27 | 2018-06-26 | Academia Sinica | Anti-HER2 glycoantibodies and uses thereof |
| JP2017518989A (ja) | 2014-05-27 | 2017-07-13 | アカデミア シニカAcademia Sinica | 抗cd20糖操作抗体群およびその使用 |
| US10118969B2 (en) | 2014-05-27 | 2018-11-06 | Academia Sinica | Compositions and methods relating to universal glycoforms for enhanced antibody efficacy |
| US11332523B2 (en) | 2014-05-28 | 2022-05-17 | Academia Sinica | Anti-TNF-alpha glycoantibodies and uses thereof |
| WO2015191715A1 (en) | 2014-06-11 | 2015-12-17 | Genentech, Inc. | Anti-lgr5 antibodies and uses thereof |
| US20230190750A1 (en) | 2014-06-13 | 2023-06-22 | Genentech, Inc. | Methods of treating and preventing cancer drug resistance |
| MX2016015280A (es) | 2014-06-26 | 2017-03-03 | Hoffmann La Roche | Anticuerpos anti-bromodesoxiuridina(brdu) y metodos de uso. |
| AU2015288232B2 (en) | 2014-07-11 | 2020-10-15 | Ventana Medical Systems, Inc. | Anti-PD-L1 antibodies and diagnostic uses thereof |
| WO2016007775A1 (en) | 2014-07-11 | 2016-01-14 | Genentech, Inc. | Notch pathway inhibition |
| AU2015293949B2 (en) | 2014-07-21 | 2019-07-25 | Teknologian Tutkimuskeskus Vtt Oy | Production of glycoproteins with mammalian-like N-glycans in filamentous fungi |
| CN107148430B (zh) | 2014-08-19 | 2021-08-27 | 默沙东公司 | 抗tigit抗体 |
| EP4074735A1 (en) | 2014-08-28 | 2022-10-19 | BioAtla, Inc. | Conditionally active chimeric antigen receptors for modified t-cells |
| TWI751102B (zh) | 2014-08-28 | 2022-01-01 | 美商奇諾治療有限公司 | 對cd19具專一性之抗體及嵌合抗原受體 |
| WO2016040369A2 (en) | 2014-09-08 | 2016-03-17 | Academia Sinica | HUMAN iNKT CELL ACTIVATION USING GLYCOLIPIDS |
| EP3193940A1 (en) | 2014-09-10 | 2017-07-26 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| AR101848A1 (es) | 2014-09-12 | 2017-01-18 | Genentech Inc | Anticuerpos de anti-b7-h4 e inmunoconjugados |
| PE20170935A1 (es) | 2014-09-12 | 2017-07-13 | Genentech Inc | Anticuerpos anti-her2 e inmunoconjugados |
| SG10201804931QA (en) | 2014-09-12 | 2018-07-30 | Genentech Inc | Anti-cll-1 antibodies and immunoconjugates |
| CA2957148A1 (en) | 2014-09-17 | 2016-03-24 | Genentech, Inc. | Immunoconjugates comprising anti-her2 antibodies and pyrrolobenzodiazepines |
| DK3262071T3 (da) | 2014-09-23 | 2020-06-15 | Hoffmann La Roche | Fremgangsmåde til anvendelse af anti-CD79b-immunkonjugater |
| MA40764A (fr) | 2014-09-26 | 2017-08-01 | Chugai Pharmaceutical Co Ltd | Agent thérapeutique induisant une cytotoxicité |
| CN107074938A (zh) | 2014-10-16 | 2017-08-18 | 豪夫迈·罗氏有限公司 | 抗‑α‑突触核蛋白抗体和使用方法 |
| WO2016059602A2 (en) | 2014-10-16 | 2016-04-21 | Glaxo Group Limited | Methods of treating cancer and related compositions |
| EP3223865A4 (en) | 2014-10-31 | 2018-10-03 | Jounce Therapeutics, Inc. | Methods of treating conditions with antibodies that bind b7-h4 |
| BR112017009159A2 (pt) | 2014-11-03 | 2018-03-06 | Genentech, Inc. | métodos e biomarcadores para predizer a eficácia e avaliação de um tratamento com agonista de ox40 |
| CA2966523A1 (en) | 2014-11-03 | 2016-05-12 | Genentech, Inc. | Assays for detecting t cell immune subsets and methods of use thereof |
| US10208120B2 (en) | 2014-11-05 | 2019-02-19 | Genentech, Inc. | Anti-FGFR2/3 antibodies and methods using same |
| US20160152720A1 (en) | 2014-11-06 | 2016-06-02 | Genentech, Inc. | Combination therapy comprising ox40 binding agonists and tigit inhibitors |
| RS59340B1 (sr) | 2014-11-06 | 2019-10-31 | Hoffmann La Roche | Varijante fc regiona sa modifikovanim vezivanjem za fcrn i metode upotrebe |
| MX2017005150A (es) | 2014-11-06 | 2017-08-08 | Hoffmann La Roche | Variantes de region fc con propiedades modificadas de union a receptor neonatal fc (fcrn) y proteina a. |
| WO2016077369A1 (en) | 2014-11-10 | 2016-05-19 | Genentech, Inc. | Animal model for nephropathy and agents for treating the same |
| JP6929771B2 (ja) | 2014-11-10 | 2021-09-01 | ジェネンテック, インコーポレイテッド | 抗インターロイキン−33抗体及びその使用 |
| WO2016075278A1 (en) | 2014-11-14 | 2016-05-19 | F. Hoffmann-La Roche Ag | Antigen binding molecules comprising a tnf family ligand trimer |
| US10160795B2 (en) | 2014-11-14 | 2018-12-25 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to Ebola virus glycoprotein and their use |
| EP3221360A1 (en) | 2014-11-17 | 2017-09-27 | F. Hoffmann-La Roche AG | Combination therapy comprising ox40 binding agonists and pd-1 axis binding antagonists |
| PL3221349T3 (pl) | 2014-11-19 | 2021-05-17 | Axon Neuroscience Se | Humanizowane przeciwciała tau w chorobie Alzheimera |
| EP3221361B1 (en) | 2014-11-19 | 2021-04-21 | Genentech, Inc. | Anti-transferrin receptor / anti-bace1 multispecific antibodies and methods of use |
| WO2016081643A1 (en) | 2014-11-19 | 2016-05-26 | Genentech, Inc. | Anti-transferrin receptor antibodies and methods of use |
| EP3221364B1 (en) | 2014-11-19 | 2020-12-16 | Genentech, Inc. | Antibodies against bace1 and use thereof for neural disease immunotherapy |
| RU2753902C2 (ru) | 2014-11-20 | 2021-08-24 | Ф.Хоффманн-Ля Рош Аг | Комбинированная терапия на основе активирующих т-клетки биспецифических антигенсвязывающих молекул против cd3 и фолатного рецептора 1 (folr1) и антагонистов, связывающихся с осью pd-1 |
| JP6721590B2 (ja) | 2014-12-03 | 2020-07-15 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | 多重特異性抗体 |
| CN107108739B (zh) | 2014-12-05 | 2022-01-04 | 豪夫迈·罗氏有限公司 | 抗CD79b抗体和使用方法 |
| CN107207591A (zh) | 2014-12-10 | 2017-09-26 | 豪夫迈·罗氏有限公司 | 血脑屏障受体抗体及使用方法 |
| SI3233921T1 (sl) | 2014-12-19 | 2022-01-31 | Chugai Seiyaku Kabushiki Kaisha | Protitelesa proti C5 in postopki za uporabo |
| WO2016111947A2 (en) | 2015-01-05 | 2016-07-14 | Jounce Therapeutics, Inc. | Antibodies that inhibit tim-3:lilrb2 interactions and uses thereof |
| CN107428833B (zh) | 2015-01-16 | 2021-11-02 | 朱诺治疗学股份有限公司 | Ror1特异性抗体和嵌合抗原受体 |
| US10495645B2 (en) | 2015-01-16 | 2019-12-03 | Academia Sinica | Cancer markers and methods of use thereof |
| US9975965B2 (en) | 2015-01-16 | 2018-05-22 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| CN113956354A (zh) | 2015-01-22 | 2022-01-21 | 中外制药株式会社 | 两种以上抗-c5抗体的组合与使用方法 |
| US10342858B2 (en) | 2015-01-24 | 2019-07-09 | Academia Sinica | Glycan conjugates and methods of use thereof |
| CN107407677B (zh) | 2015-01-28 | 2020-07-17 | 豪夫迈·罗氏有限公司 | 多发性硬化的基因表达标志和治疗 |
| CN107636170A (zh) | 2015-02-04 | 2018-01-26 | 健泰科生物技术公司 | 突变型Smoothened及其使用方法 |
| KR20170110129A (ko) | 2015-02-05 | 2017-10-10 | 추가이 세이야쿠 가부시키가이샤 | 이온 농도 의존적 항원 결합 도메인을 포함하는 항체, Fc 영역 개변체, IL-8에 결합하는 항체, 및 그들의 사용 |
| KR20170140180A (ko) | 2015-02-24 | 2017-12-20 | 더 유나이티드 스테이츠 오브 어메리카, 애즈 리프리젠티드 바이 더 세크러테리, 디파트먼트 오브 헬쓰 앤드 휴먼 서비씨즈 | 중동 호흡기 증후군 코로나 바이러스 면역원, 항체 및 그 용도 |
| AU2016230827B2 (en) | 2015-03-09 | 2021-10-28 | Argenx Bvba | Methods of reducing serum levels of fc-containing agents using fcrn antagonists |
| WO2016149276A1 (en) | 2015-03-16 | 2016-09-22 | Genentech, Inc. | Methods of detecting and quantifying il-13 and uses in diagnosing and treating th2-associated diseases |
| WO2016146833A1 (en) | 2015-03-19 | 2016-09-22 | F. Hoffmann-La Roche Ag | Biomarkers for nad(+)-diphthamide adp ribosyltransferase resistance |
| US10562960B2 (en) | 2015-03-20 | 2020-02-18 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to gp120 and their use |
| PT3274370T (pt) | 2015-03-23 | 2020-01-30 | Bayer Pharma AG | Anticorpos anti-ceacam6 e utilizações dos mesmos |
| JP6944924B2 (ja) | 2015-03-23 | 2021-10-06 | ジョンス セラピューティクス, インコーポレイテッド | Icosに対する抗体 |
| MY186708A (en) | 2015-04-03 | 2021-08-11 | Eureka Therapeutics Inc | Constructs targeting afp peptide/mhc complexes and uses thereof |
| EP3286315B1 (en) | 2015-04-24 | 2021-05-26 | F. Hoffmann-La Roche AG | Methods of identifying bacteria comprising binding polypeptides |
| EP3288981A1 (en) | 2015-05-01 | 2018-03-07 | Genentech, Inc. | Masked anti-cd3 antibodies and methods of use |
| WO2016179194A1 (en) | 2015-05-04 | 2016-11-10 | Jounce Therapeutics, Inc. | Lilra3 and method of using the same |
| EA201792451A1 (ru) | 2015-05-07 | 2018-05-31 | Агенус Инк. | Антитела к ox40 и способы их применения |
| EP3936524A3 (en) | 2015-05-11 | 2022-06-15 | F. Hoffmann-La Roche AG | Compositions and methods of treating lupus nephritis |
| SI3294770T1 (sl) | 2015-05-12 | 2021-01-29 | F. Hoffmann-La Roche Ag | Terapevtski in diagnostični postopki za raka |
| JP2018520658A (ja) | 2015-05-29 | 2018-08-02 | ジェネンテック, インコーポレイテッド | ヒト化抗エボラウイルス糖タンパク質抗体及びその使用 |
| AU2016270625B2 (en) | 2015-05-29 | 2022-10-06 | Genentech, Inc. | Therapeutic and diagnostic methods for cancer |
| EP3763827A1 (en) | 2015-05-29 | 2021-01-13 | F. Hoffmann-La Roche AG | Pd-l1 promoter methylation in cancer |
| WO2016196679A1 (en) | 2015-06-02 | 2016-12-08 | Genentech, Inc. | Compositions and methods for using anti-il-34 antibodies to treat neurological diseases |
| WO2016196975A1 (en) | 2015-06-03 | 2016-12-08 | The United States Of America, As Represented By The Secretary Department Of Health & Human Services | Neutralizing antibodies to hiv-1 env and their use |
| WO2016193497A1 (en) | 2015-06-04 | 2016-12-08 | Ospedale San Raffaele Srl | Inhibitor of igfbp3/tmem219 axis and diabetes |
| ES2834823T3 (es) | 2015-06-04 | 2021-06-18 | Ospedale San Raffaele Srl | IGFBP3 y usos del mismo |
| SG10201911349YA (en) | 2015-06-05 | 2020-01-30 | Genentech Inc | Anti-tau antibodies and methods of use |
| CN107810011A (zh) | 2015-06-08 | 2018-03-16 | 豪夫迈·罗氏有限公司 | 使用抗ox40抗体治疗癌症的方法 |
| AU2016274584A1 (en) | 2015-06-08 | 2018-01-04 | Genentech, Inc. | Methods of treating cancer using anti-OX40 antibodies and PD-1 axis binding antagonists |
| AR104987A1 (es) | 2015-06-15 | 2017-08-30 | Genentech Inc | Inmunoconjugados anticuerpo-fármaco unidos por enlazadores no peptídicos |
| WO2016204966A1 (en) | 2015-06-16 | 2016-12-22 | Genentech, Inc. | Anti-cd3 antibodies and methods of use |
| LT3310814T (lt) | 2015-06-16 | 2023-10-10 | F. Hoffmann-La Roche Ag | Humanizuoti ir subrendusio giminingumo antikūnai prieš fcrh5 ir jų naudojimo būdai |
| WO2016205200A1 (en) | 2015-06-16 | 2016-12-22 | Genentech, Inc. | Anti-cll-1 antibodies and methods of use |
| CN107771076A (zh) | 2015-06-17 | 2018-03-06 | 豪夫迈·罗氏有限公司 | 使用pd‑1轴结合拮抗剂和紫杉烷治疗局部晚期或转移性乳腺癌的方法 |
| MX2017016169A (es) | 2015-06-17 | 2018-08-15 | Genentech Inc | Anticuerpos anti-her2 y metodos de uso. |
| CA2987797A1 (en) | 2015-06-17 | 2016-12-22 | Christopher Robert Bebbington | Methods and compositions for treating fibrotic diseases |
| ES2908009T3 (es) | 2015-06-24 | 2022-04-27 | Hoffmann La Roche | Anticuerpos anti-receptor de transferrina con afinidad adaptada |
| ES2898065T3 (es) | 2015-06-29 | 2022-03-03 | Ventana Med Syst Inc | Materiales y procedimientos para realizar ensayos histoquímicos para proepirregulina y anfirregulina humanas |
| MX2017016645A (es) | 2015-06-29 | 2018-11-09 | Genentech Inc | Anticuerpo anti-cd20 de tipo ii para su uso en el trasplante de órganos. |
| RU2611685C2 (ru) * | 2015-07-20 | 2017-02-28 | Илья Владимирович Духовлинов | Гуманизированное моноклональное антитело, специфичное к синдекану-1 |
| EP4137158A1 (en) | 2015-08-07 | 2023-02-22 | Imaginab, Inc. | Antigen binding constructs to target molecules |
| CN105384825B (zh) | 2015-08-11 | 2018-06-01 | 南京传奇生物科技有限公司 | 一种基于单域抗体的双特异性嵌合抗原受体及其应用 |
| JP6914919B2 (ja) | 2015-08-28 | 2021-08-04 | ジェネンテック, インコーポレイテッド | 抗ヒプシン抗体及びその使用 |
| JP7074341B2 (ja) | 2015-09-02 | 2022-05-24 | イムテップ エス.アー.エス. | 抗lag-3抗体 |
| CA2999369C (en) | 2015-09-22 | 2023-11-07 | Spring Bioscience Corporation | Anti-ox40 antibodies and diagnostic uses thereof |
| IL257565B1 (en) | 2015-09-23 | 2024-04-01 | Genentech Inc | Improved variants of anti-VEGF antibodies |
| EP3662930A1 (en) | 2015-09-24 | 2020-06-10 | AbVitro LLC | Hiv antibody compositions and methods of use |
| AU2016332900C1 (en) | 2015-09-29 | 2024-02-01 | Amgen Inc. | ASGR inhibitors |
| AR106188A1 (es) | 2015-10-01 | 2017-12-20 | Hoffmann La Roche | Anticuerpos anti-cd19 humano humanizados y métodos de utilización |
| JP6657392B2 (ja) | 2015-10-02 | 2020-03-04 | エフ・ホフマン−ラ・ロシュ・アクチェンゲゼルシャフト | 二重特異性抗ヒトcd20/ヒトトランスフェリン受容体抗体及び使用方法 |
| AR106189A1 (es) | 2015-10-02 | 2017-12-20 | Hoffmann La Roche | ANTICUERPOS BIESPECÍFICOS CONTRA EL A-b HUMANO Y EL RECEPTOR DE TRANSFERRINA HUMANO Y MÉTODOS DE USO |
| AU2016329120B2 (en) | 2015-10-02 | 2023-04-13 | F. Hoffmann-La Roche Ag | Bispecific antibodies specific for a costimulatory TNF receptor |
| EP3150636A1 (en) | 2015-10-02 | 2017-04-05 | F. Hoffmann-La Roche AG | Tetravalent multispecific antibodies |
| DK3356411T3 (da) | 2015-10-02 | 2021-09-06 | Hoffmann La Roche | Bispecifikke antistoffer, der er specifikke for PD1 og TIM3 |
| MA43345A (fr) | 2015-10-02 | 2018-08-08 | Hoffmann La Roche | Conjugués anticorps-médicaments de pyrrolobenzodiazépine et méthodes d'utilisation |
| JP2018529747A (ja) | 2015-10-06 | 2018-10-11 | ジェネンテック, インコーポレイテッド | 多発性硬化症を治療するための方法 |
| CR20180171A (es) | 2015-10-07 | 2018-05-03 | Hoffmann La Roche | Anticuerpos biespecíficos con tetravalencia para un receptor de fnt coestimulador |
| US10392441B2 (en) | 2015-10-07 | 2019-08-27 | United States Of America, As Represented By The Secretary, Department Of Health And Human Services | IL-7R-alpha specific antibodies for treating acute lymphoblastic leukemia |
| US10604577B2 (en) | 2015-10-22 | 2020-03-31 | Allakos Inc. | Methods and compositions for treating systemic mastocytosis |
| EP3365372A1 (en) | 2015-10-22 | 2018-08-29 | Jounce Therapeutics, Inc. | Gene signatures for determining icos expression |
| MX2018005036A (es) | 2015-10-29 | 2018-08-01 | Hoffmann La Roche | Anticuerpos y metodos de uso de anti-regiones de fragmentos cristalizables (fc) variantes. |
| EP3184547A1 (en) | 2015-10-29 | 2017-06-28 | F. Hoffmann-La Roche AG | Anti-tpbg antibodies and methods of use |
| JO3555B1 (ar) | 2015-10-29 | 2020-07-05 | Merck Sharp & Dohme | جسم مضاد يبطل فعالية فيروس الالتهاب الرئوي البشري |
| CN115160439A (zh) | 2015-10-30 | 2022-10-11 | 豪夫迈·罗氏有限公司 | 抗-HtrA1抗体及其使用方法 |
| EP3368074A2 (en) | 2015-10-30 | 2018-09-05 | Hoffmann-La Roche AG | Anti-factor d antibodies and conjugates |
| EP4011911A1 (en) | 2015-11-03 | 2022-06-15 | The United States of America as represented by The Secretary Department of Health and Human Services | Neutralizing antibodies to hiv-1 gp41 and their use |
| EP3371217A1 (en) | 2015-11-08 | 2018-09-12 | H. Hoffnabb-La Roche Ag | Methods of screening for multispecific antibodies |
| SG11201804134YA (en) | 2015-11-23 | 2018-06-28 | Five Prime Therapeutics Inc | Fgfr2 inhibitors alone or in combination with immune stimulating agents in cancer treatment |
| BR112018003984A2 (pt) | 2015-12-09 | 2018-09-25 | Hoffmann La Roche | anticorpos |
| AU2016372930B2 (en) | 2015-12-18 | 2020-10-08 | Chugai Seiyaku Kabushiki Kaisha | Anti-C5 antibodies and methods of use |
| CN115400220A (zh) | 2015-12-30 | 2022-11-29 | 豪夫迈·罗氏有限公司 | 减少聚山梨酯降解的制剂 |
| WO2017118675A1 (en) | 2016-01-08 | 2017-07-13 | F. Hoffmann-La Roche Ag | Methods of treating cea-positive cancers using pd-1 axis binding antagonists and anti-cea/anti-cd3 bispecific antibodies |
| BR112018014762A2 (pt) | 2016-01-20 | 2018-12-26 | Genentech Inc | método de tratamento da doença de alzheimer (da) precoce |
| BR112018015259A2 (pt) * | 2016-01-27 | 2018-12-18 | Medimmune Llc | métodos para preparação de anticorpos com um padrão de glicosilação definido |
| AU2017213826A1 (en) | 2016-02-04 | 2018-08-23 | Curis, Inc. | Mutant smoothened and methods of using the same |
| WO2017145166A1 (en) * | 2016-02-25 | 2017-08-31 | B. G. Negev Technologies And Applications Ltd., At Ben-Gurion University | Composition and method for treating amyotrophic lateral sclerosis |
| CN109196121B (zh) | 2016-02-29 | 2022-01-04 | 基因泰克公司 | 用于癌症的治疗和诊断方法 |
| WO2017156192A1 (en) | 2016-03-08 | 2017-09-14 | Academia Sinica | Methods for modular synthesis of n-glycans and arrays thereof |
| CA3016552A1 (en) | 2016-03-15 | 2017-09-21 | Genentech, Inc. | Methods of treating cancers using pd-1 axis binding antagonists and anti-gpc3 antibodies |
| US20170315132A1 (en) | 2016-03-25 | 2017-11-02 | Genentech, Inc. | Multiplexed total antibody and antibody-conjugated drug quantification assay |
| KR20180128471A (ko) | 2016-03-29 | 2018-12-03 | 얀센 바이오테크 인코포레이티드 | 항-il12 및/또는 항-il23 항체의 증가된 투여 간격을 사용한 건선의 치료 |
| KR20180131557A (ko) | 2016-04-05 | 2018-12-10 | 글락소스미스클라인 인털렉츄얼 프로퍼티 디벨로프먼트 리미티드 | 면역요법에서의 tgf베타의 억제 |
| EP3443004A1 (en) | 2016-04-14 | 2019-02-20 | H. Hoffnabb-La Roche Ag | Anti-rspo3 antibodies and methods of use |
| EP3443120A2 (en) | 2016-04-15 | 2019-02-20 | H. Hoffnabb-La Roche Ag | Methods for monitoring and treating cancer |
| KR20190003958A (ko) | 2016-04-15 | 2019-01-10 | 제넨테크, 인크. | 암의 치료 및 모니터링 방법 |
| CA3021086C (en) | 2016-04-15 | 2023-10-17 | Bioatla, Llc | Anti-axl antibodies, antibody fragments and their immunoconjugates and uses thereof |
| MA56474A (fr) | 2016-05-02 | 2022-05-11 | Hoffmann La Roche | Contorsbody - liant de cible à chaîne unique |
| WO2017192589A1 (en) | 2016-05-02 | 2017-11-09 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to influenza ha and their use and identification |
| EP3455254B1 (en) | 2016-05-11 | 2021-07-07 | F. Hoffmann-La Roche AG | Antigen binding molecules comprising a tnf family ligand trimer and a tenascin binding moiety |
| JP7089483B2 (ja) | 2016-05-11 | 2022-06-22 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | 修飾された抗テネイシン抗体及び使用方法 |
| EP3243836A1 (en) | 2016-05-11 | 2017-11-15 | F. Hoffmann-La Roche AG | C-terminally fused tnf family ligand trimer-containing antigen binding molecules |
| EP3243832A1 (en) | 2016-05-13 | 2017-11-15 | F. Hoffmann-La Roche AG | Antigen binding molecules comprising a tnf family ligand trimer and pd1 binding moiety |
| HRP20221298T1 (hr) | 2016-05-13 | 2022-12-23 | Bioatla, Inc. | Anti-ror2 protutijela, fragmenti protutijela, njihovi imunokonjugati i njihove uporabe |
| EP3458101B1 (en) | 2016-05-20 | 2020-12-30 | H. Hoffnabb-La Roche Ag | Protac antibody conjugates and methods of use |
| US20170370906A1 (en) | 2016-05-27 | 2017-12-28 | Genentech, Inc. | Bioanalytical analysis of site-specific antibody drug conjugates |
| CN110603266A (zh) | 2016-06-02 | 2019-12-20 | 豪夫迈·罗氏有限公司 | 用于治疗癌症的ii型抗cd20抗体和抗cd20/cd3双特异性抗体 |
| EP3252078A1 (en) | 2016-06-02 | 2017-12-06 | F. Hoffmann-La Roche AG | Type ii anti-cd20 antibody and anti-cd20/cd3 bispecific antibody for treatment of cancer |
| CN116143918A (zh) | 2016-06-24 | 2023-05-23 | 豪夫迈·罗氏有限公司 | 抗聚泛素多特异性抗体 |
| JP6983824B2 (ja) | 2016-07-04 | 2021-12-17 | エフ.ホフマン−ラ ロシュ アーゲーF. Hoffmann−La Roche Aktiengesellschaft | 新規抗体フォーマット |
| WO2018014260A1 (en) | 2016-07-20 | 2018-01-25 | Nanjing Legend Biotech Co., Ltd. | Multispecific antigen binding proteins and methods of use thereof |
| KR102553195B1 (ko) | 2016-07-29 | 2023-07-07 | 주노 쎄러퓨티크스 인코퍼레이티드 | 항-cd19 항체에 대한 항-이디오타입 항체 |
| MX2018015721A (es) | 2016-07-29 | 2019-05-27 | Chugai Pharmaceutical Co Ltd | Anticuerpos biespecificos que exhiben actividad de funcion de cofactor fviii alternativa mejorada. |
| NL2017270B1 (en) | 2016-08-02 | 2018-02-09 | Aduro Biotech Holdings Europe B V | New anti-hCTLA-4 antibodies |
| EP3494991A4 (en) | 2016-08-05 | 2020-07-29 | Chugai Seiyaku Kabushiki Kaisha | COMPOSITION FOR PREVENTING OR TREATING DISEASES RELATING TO IL-8 |
| CN109476748B (zh) | 2016-08-08 | 2023-05-23 | 豪夫迈·罗氏有限公司 | 用于癌症的治疗和诊断方法 |
| US10538592B2 (en) | 2016-08-22 | 2020-01-21 | Cho Pharma, Inc. | Antibodies, binding fragments, and methods of use |
| AU2017321973A1 (en) | 2016-09-02 | 2019-03-07 | Dana-Farber Cancer Institute, Inc. | Composition and methods of treating B cell disorders |
| EP3510046A4 (en) | 2016-09-07 | 2020-05-06 | The Regents of the University of California | ANTIBODIES AGAINST OXIDATION-SPECIFIC EPITOPES |
| SG10201607778XA (en) | 2016-09-16 | 2018-04-27 | Chugai Pharmaceutical Co Ltd | Anti-Dengue Virus Antibodies, Polypeptides Containing Variant Fc Regions, And Methods Of Use |
| EP3515932B1 (en) | 2016-09-19 | 2023-11-22 | F. Hoffmann-La Roche AG | Complement factor based affinity chromatography |
| SI3528838T1 (sl) | 2016-09-23 | 2023-10-30 | F. Hoffmann-La Roche Ag | Uporabe IL-13 antagonistov za zdravljenje atopijskega dermatitisa |
| JOP20190055A1 (ar) | 2016-09-26 | 2019-03-24 | Merck Sharp & Dohme | أجسام مضادة ضد cd27 |
| MA46366A (fr) | 2016-09-30 | 2019-08-07 | Janssen Biotech Inc | Procédé sûr et efficace de traitement du psoriasis avec un anticorps spécifique contre l'il-23 |
| EP3519433A1 (en) | 2016-10-03 | 2019-08-07 | Juno Therapeutics, Inc. | Hpv-specific binding molecules |
| JP7050770B2 (ja) | 2016-10-05 | 2022-04-08 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | 抗体薬物コンジュゲートの調製方法 |
| TWI775781B (zh) | 2016-10-06 | 2022-09-01 | 美商建南德克公司 | 癌症之治療性及診斷性方法 |
| EP3525829A1 (en) | 2016-10-11 | 2019-08-21 | Medimmune Limited | Antibody-drug conjugates with immune-mediated therapy agents |
| WO2018068201A1 (en) | 2016-10-11 | 2018-04-19 | Nanjing Legend Biotech Co., Ltd. | Single-domain antibodies and variants thereof against ctla-4 |
| US11555076B2 (en) | 2016-10-29 | 2023-01-17 | Genentech, Inc. | Anti-MIC antibodies and methods of use |
| HUE057559T2 (hu) | 2016-11-02 | 2022-06-28 | Jounce Therapeutics Inc | PD-1 elleni antitestek és alkalmazásaik |
| AU2017361081A1 (en) | 2016-11-15 | 2019-05-23 | Genentech, Inc. | Dosing for treatment with anti-CD20/anti-CD3 bispecific antibodies |
| MA46861A (fr) | 2016-11-16 | 2019-09-25 | Janssen Biotech Inc | Procédé de traitement du psoriasis avec un anticorps anti-il-23 spécifique |
| JOP20190100A1 (ar) | 2016-11-19 | 2019-05-01 | Potenza Therapeutics Inc | بروتينات ربط مولد ضد مضاد لـ gitr وطرق استخدامها |
| EP4015532A1 (en) | 2016-11-21 | 2022-06-22 | cureab GmbH | Anti-gp73 antibodies and immunoconjugates |
| US10759855B2 (en) | 2016-12-02 | 2020-09-01 | Rigel Pharmaceuticals, Inc. | Antigen binding molecules to TIGIT |
| BR112019011582A2 (pt) | 2016-12-07 | 2019-10-22 | Agenus Inc. | anticorpos e métodos de utilização dos mesmos |
| AU2017373884A1 (en) | 2016-12-07 | 2019-05-30 | Ac Immune Sa | Anti-tau antibodies and methods of their use |
| AU2017373889A1 (en) | 2016-12-07 | 2019-06-06 | Ac Immune Sa | Anti-Tau antibodies and methods of use |
| KR20190095921A (ko) | 2016-12-12 | 2019-08-16 | 제넨테크, 인크. | 항-pd-l1 항체 및 안티안드로겐을 사용하여 암을 치료하는 방법 |
| KR20190097039A (ko) | 2016-12-19 | 2019-08-20 | 에프. 호프만-라 로슈 아게 | 표적화된 4-1bb(cd137) 작용물질과의 병용 요법 |
| CA3039446A1 (en) | 2016-12-20 | 2018-06-28 | F. Hoffmann-La Roche Ag | Combination therapy of anti-cd20/anti-cd3 bispecific antibodies and 4-1bb (cd137) agonists |
| JOP20190134A1 (ar) | 2016-12-23 | 2019-06-02 | Potenza Therapeutics Inc | بروتينات رابطة لمولد ضد مضادة لنيوروبيلين وطرق استخدامها |
| MX2019007795A (es) | 2017-01-03 | 2019-08-16 | Hoffmann La Roche | Moleculas de union a antigeno biespecificas que comprenden el clon 20h4.9 anti-4-1bb. |
| WO2018129029A1 (en) | 2017-01-04 | 2018-07-12 | Immunogen, Inc. | Met antibodies and immunoconjugates and uses thereof |
| EP3568468A4 (en) | 2017-01-12 | 2020-12-30 | Eureka Therapeutics, Inc. | RECOMBINATION PRODUCTS TARGETING PEPTIDE HISTONE H3 / MHC COMPLEXES AND THEIR USES |
| US11266745B2 (en) | 2017-02-08 | 2022-03-08 | Imaginab, Inc. | Extension sequences for diabodies |
| CA3051700A1 (en) | 2017-02-10 | 2018-08-16 | Genentech, Inc. | Anti-tryptase antibodies, compositions thereof, and uses thereof |
| US11021535B2 (en) | 2017-02-10 | 2021-06-01 | The United States Of America As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to plasmodium falciparum circumsporozoite protein and their use |
| CN110546277A (zh) | 2017-03-01 | 2019-12-06 | 豪夫迈·罗氏有限公司 | 用于癌症的诊断和治疗方法 |
| EP3600442A1 (en) | 2017-03-22 | 2020-02-05 | Genentech, Inc. | Optimized antibody compositions for treatment of ocular disorders |
| EP3601337A1 (en) | 2017-03-28 | 2020-02-05 | Genentech, Inc. | Methods of treating neurodegenerative diseases |
| EP3601346A1 (en) | 2017-03-29 | 2020-02-05 | H. Hoffnabb-La Roche Ag | Bispecific antigen binding molecule for a costimulatory tnf receptor |
| JP7196094B2 (ja) | 2017-03-29 | 2022-12-26 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | 共刺激tnf受容体のための二重特異性抗原結合分子 |
| JOP20190203A1 (ar) | 2017-03-30 | 2019-09-03 | Potenza Therapeutics Inc | بروتينات رابطة لمولد ضد مضادة لـ tigit وطرق استخدامها |
| CN110494452B (zh) | 2017-04-03 | 2023-08-25 | 豪夫迈·罗氏有限公司 | 结合steap-1的抗体 |
| CR20190427A (es) | 2017-04-04 | 2019-11-01 | Hoffmann La Roche | Nuevas moléculas de unión a antígeno biespecíficas capaces de unirse específicamente a cd40 y a fap |
| CN110392698B (zh) | 2017-04-05 | 2022-01-25 | 豪夫迈·罗氏有限公司 | 抗lag3抗体 |
| JP6997212B2 (ja) | 2017-04-05 | 2022-02-04 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | Pd1及びlag3に特異的に結合する二重特異性抗体 |
| CA3058478A1 (en) | 2017-04-14 | 2018-10-18 | Genentech, Inc. | Diagnostic and therapeutic methods for cancer |
| CA3059615A1 (en) | 2017-04-21 | 2018-10-25 | Genentech, Inc. | Use of klk5 antagonists for treatment of a disease |
| KR20200012860A (ko) | 2017-04-26 | 2020-02-05 | 유레카 쎄라퓨틱스, 인코포레이티드 | 글리피칸 3을 특이적으로 인식하는 구축물 및 그의 용도 |
| US20220135670A1 (en) | 2017-04-27 | 2022-05-05 | Tesaro, Inc. | Antibody agents directed against lymphocyte activation gene-3 (lag-3) and uses thereof |
| EA201992626A1 (ru) | 2017-05-05 | 2020-04-24 | Аллакос Инк. | Способы и композиции для лечения аллергических заболеваний глаз |
| CA3062177A1 (en) | 2017-05-16 | 2018-11-22 | Five Prime Therapeutics, Inc. | Anti-fgfr2 antibodies in combination with chemotherapy agents in cancer treatment |
| CN110945028B (zh) | 2017-07-10 | 2023-09-08 | 国际药物发展生物技术公司 | 用非岩藻糖基化促凋亡抗cd19抗体与抗cd20抗体或化疗剂联合治疗b细胞恶性肿瘤 |
| JP2020527351A (ja) | 2017-07-21 | 2020-09-10 | ジェネンテック, インコーポレイテッド | がんの治療法及び診断法 |
| AU2018308364C1 (en) | 2017-07-26 | 2023-02-16 | Forty Seven, Inc. | Anti-SIRP-alpha antibodies and related methods |
| WO2019040780A1 (en) | 2017-08-25 | 2019-02-28 | Five Prime Therapeutics Inc. | ANTI-B7-H4 ANTIBODIES AND METHODS OF USE |
| WO2019059411A1 (en) | 2017-09-20 | 2019-03-28 | Chugai Seiyaku Kabushiki Kaisha | DOSAGE FOR POLYTHERAPY USING PD-1 AXIS BINDING ANTAGONISTS AND GPC3 TARGETING AGENT |
| TW201922780A (zh) | 2017-09-25 | 2019-06-16 | 美商健生生物科技公司 | 以抗il12/il23抗體治療狼瘡之安全且有效之方法 |
| KR20200104284A (ko) | 2017-10-03 | 2020-09-03 | 주노 쎄러퓨티크스 인코퍼레이티드 | Hpv-특이적 결합 분자 |
| US11230601B2 (en) | 2017-10-10 | 2022-01-25 | Tilos Therapeutics, Inc. | Methods of using anti-lap antibodies |
| EP3694890A4 (en) | 2017-10-12 | 2021-11-03 | Immunowake Inc. | LIGHT CHAIN ANTIBODY FUSION PROTEIN WITH VEGFR |
| AU2018357923A1 (en) | 2017-11-01 | 2020-03-05 | F. Hoffmann-La Roche Ag | Bispecific 2+1 contorsbodies |
| CN111246884A (zh) | 2017-11-01 | 2020-06-05 | 豪夫迈·罗氏有限公司 | 新颖的含有tnf家族配体三聚体的抗原结合分子 |
| AU2018359506A1 (en) | 2017-11-01 | 2020-04-23 | F. Hoffmann-La Roche Ag | Combination therapy with targeted OX40 agonists |
| MX2020004243A (es) | 2017-11-01 | 2020-09-25 | Juno Therapeutics Inc | Receptores de anticuerpos y de antigenos quimericos especificos para el antigeno de maduracion de celulas b. |
| TW201923089A (zh) | 2017-11-06 | 2019-06-16 | 美商建南德克公司 | 癌症之診斷及治療方法 |
| CN111615520A (zh) | 2017-12-01 | 2020-09-01 | 辉瑞大药厂 | 抗cxcr5抗体及其组合物和用途 |
| MA51032A (fr) | 2017-12-08 | 2021-03-17 | Argenx Bvba | Utilisation d'antagonistes de fcrn pour le traitement de la myasthénie grave généralisée |
| KR20200110745A (ko) | 2017-12-15 | 2020-09-25 | 주노 쎄러퓨티크스 인코퍼레이티드 | 항 - cct5 결합 분자 및 이의 사용 방법 |
| EP3502140A1 (en) | 2017-12-21 | 2019-06-26 | F. Hoffmann-La Roche AG | Combination therapy of tumor targeted icos agonists with t-cell bispecific molecules |
| AU2018390881A1 (en) | 2017-12-21 | 2020-07-02 | F. Hoffmann-La Roche Ag | Antibodies binding to HLA-A2/WT1 |
| TW201929907A (zh) | 2017-12-22 | 2019-08-01 | 美商建南德克公司 | Pilra結合劑用於治療疾病之用途 |
| EP4219559A3 (en) | 2017-12-22 | 2023-10-18 | Jounce Therapeutics, Inc. | Antibodies for lilrb2 |
| EP3732202A4 (en) | 2017-12-28 | 2022-06-15 | Nanjing Legend Biotech Co., Ltd. | SINGLE DOMAIN ANTIBODIES AND VARIANTS THEREOF AGAINST TIGIT |
| US20220135687A1 (en) | 2017-12-28 | 2022-05-05 | Nanjing Legend Biotech Co., Ltd. | Antibodies and variants thereof against pd-l1 |
| WO2019129679A1 (en) | 2017-12-29 | 2019-07-04 | F. Hoffmann-La Roche Ag | Method for improving vegf-receptor blocking selectivity of an anti-vegf antibody |
| WO2019136029A1 (en) | 2018-01-02 | 2019-07-11 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to ebola virus glycoprotein and their use |
| KR20200106525A (ko) | 2018-01-05 | 2020-09-14 | 에이씨 이뮨 에스.에이. | 미스폴딩된 tdp-43 결합 분자 |
| EP3508499A1 (en) | 2018-01-08 | 2019-07-10 | iOmx Therapeutics AG | Antibodies targeting, and other modulators of, an immunoglobulin gene associated with resistance against anti-tumour immune responses, and uses thereof |
| CA3084518A1 (en) | 2018-01-15 | 2019-07-18 | Nanjing Legend Biotech Co., Ltd. | Single-domain antibodies and variants thereof against pd-1 |
| CA3088649A1 (en) | 2018-01-16 | 2019-07-25 | Lakepharma, Inc. | Bispecific antibody that binds cd3 and another target |
| CR20200327A (es) | 2018-01-26 | 2020-11-05 | Genentech Inc | Proteínas de fusión fc il-22 y métodos de uso |
| ES2932861T3 (es) | 2018-01-26 | 2023-01-27 | Hoffmann La Roche | Composiciones de IL-22 Fc y procedimientos de uso |
| JP7438953B2 (ja) | 2018-02-01 | 2024-02-27 | イノベント バイオロジックス (スウツォウ) カンパニー,リミテッド | 完全ヒト化抗b細胞成熟抗原(bcma)の単鎖抗体およびその応用 |
| SG11202007390YA (en) | 2018-02-08 | 2020-08-28 | Genentech Inc | Bispecific antigen-binding molecules and methods of use |
| KR102417088B1 (ko) | 2018-02-09 | 2022-07-07 | 제넨테크, 인크. | 비만 세포 매개 염증성 질환에 대한 치료 및 진단 방법 |
| TWI829667B (zh) | 2018-02-09 | 2024-01-21 | 瑞士商赫孚孟拉羅股份公司 | 結合gprc5d之抗體 |
| EP3752530A1 (en) | 2018-02-14 | 2020-12-23 | ABBA Therapeutics AG | Anti-human pd-l2 antibodies |
| RU2020130795A (ru) | 2018-02-21 | 2022-03-21 | Дзе Юнайтед Стэйтс Оф Америка, Эс Репрезентед Бай Дзе Секретэри, Департмент Оф Хелт Энд Хьюман Сервисиз | Нейтрализующие антитела к env вич-1 и их применение |
| WO2019165075A1 (en) | 2018-02-21 | 2019-08-29 | Five Prime Therapeutics, Inc. | B7-h4 antibody dosing regimens |
| CA3091174A1 (en) | 2018-02-21 | 2019-08-29 | Five Prime Therapeutics, Inc. | B7-h4 antibody formulations |
| EP3755364A1 (en) | 2018-02-21 | 2020-12-30 | F. Hoffmann-La Roche AG | Dosing for treatment with il-22 fc fusion proteins |
| TW202000702A (zh) | 2018-02-26 | 2020-01-01 | 美商建南德克公司 | 用於抗tigit拮抗劑抗體及抗pd-l1拮抗劑抗體治療之投藥 |
| JP2021516051A (ja) | 2018-03-02 | 2021-07-01 | ファイブ プライム セラピューティクス, インコーポレイテッド | B7−h4抗体及びその使用方法 |
| JP2021515770A (ja) | 2018-03-05 | 2021-06-24 | ヤンセン バイオテツク,インコーポレーテツド | 抗il−23特異的抗体を用いたクローン病の治療方法 |
| KR20200132913A (ko) | 2018-03-13 | 2020-11-25 | 에프. 호프만-라 로슈 아게 | 4-1bb 작용제와 항-cd20 항체의 치료 조합 |
| TW202003561A (zh) | 2018-03-13 | 2020-01-16 | 瑞士商赫孚孟拉羅股份公司 | 使用靶向4-1bb (cd137)之促效劑的組合療法 |
| US20200040103A1 (en) | 2018-03-14 | 2020-02-06 | Genentech, Inc. | Anti-klk5 antibodies and methods of use |
| AU2019235033A1 (en) | 2018-03-14 | 2020-07-30 | Beijing Xuanyi Pharmasciences Co., Ltd. | Anti-claudin 18.2 antibodies |
| CA3093729A1 (en) | 2018-03-15 | 2019-09-19 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies having cross-reactivity to zika virus and methods of use |
| PE20210290A1 (es) | 2018-03-21 | 2021-02-11 | Five Prime Therapeutics Inc | ANTICUERPOS DE UNION A VISTA A pH ACIDO |
| AU2019245243A1 (en) | 2018-03-29 | 2020-09-03 | Genentech, Inc | Modulating lactogenic activity in mammalian cells |
| TW202011029A (zh) | 2018-04-04 | 2020-03-16 | 美商建南德克公司 | 偵測及定量fgf21之方法 |
| MA52193A (fr) | 2018-04-05 | 2021-02-17 | Juno Therapeutics Inc | Récepteurs de lymphocytes t et cellules modifiées les exprimant |
| JP7418346B2 (ja) | 2018-04-13 | 2024-01-19 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | 4-1BBLを含むHer2標的化抗原結合分子 |
| US20190345245A1 (en) | 2018-05-11 | 2019-11-14 | Janssen Biotech, Inc. | Methods of Treating Crohn's Disease with Anti-IL23 Specific Antibody |
| CA3100007A1 (en) | 2018-05-14 | 2019-11-21 | Werewolf Therapeutics, Inc. | Activatable interleukin-2 polypeptides and methods of use thereof |
| WO2019222294A1 (en) | 2018-05-14 | 2019-11-21 | Werewolf Therapeutics, Inc. | Activatable cytokine polypeptides and methods of use thereof |
| EP3796942A1 (en) | 2018-05-23 | 2021-03-31 | ADC Therapeutics SA | Molecular adjuvant |
| CN112566938A (zh) | 2018-06-03 | 2021-03-26 | 拉姆卡普生物测试有限公司 | 针对ceacam5和cd47的双特异性抗体 |
| EP3805400A4 (en) | 2018-06-04 | 2022-06-29 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule showing changed half-life in cytoplasm |
| WO2019246557A1 (en) | 2018-06-23 | 2019-12-26 | Genentech, Inc. | Methods of treating lung cancer with a pd-1 axis binding antagonist, a platinum agent, and a topoisomerase ii inhibitor |
| JP2021528988A (ja) | 2018-07-04 | 2021-10-28 | エフ・ホフマン−ラ・ロシュ・アクチェンゲゼルシャフト | 新規の二重特異性アゴニスト4−1bb抗原結合分子 |
| SG11202100096XA (en) | 2018-07-09 | 2021-02-25 | Five Prime Therapeutics Inc | Antibodies binding to ilt4 |
| WO2020014306A1 (en) | 2018-07-10 | 2020-01-16 | Immunogen, Inc. | Met antibodies and immunoconjugates and uses thereof |
| BR112021000303A2 (pt) | 2018-07-11 | 2021-04-13 | Five Prime Therapeutics, Inc. | Anticorpos que se ligam a vista em ph ácido |
| US20200025776A1 (en) | 2018-07-18 | 2020-01-23 | Janssen Biotech, Inc. | Sustained Response Predictors After Treatment With Anti-IL23 Specific Antibody |
| KR20210034622A (ko) | 2018-07-18 | 2021-03-30 | 제넨테크, 인크. | Pd-1 축 결합 길항제, 항 대사제, 및 백금 제제를 이용한 폐암 치료 방법 |
| TW202017944A (zh) | 2018-07-20 | 2020-05-16 | 美商表面腫瘤學公司 | 抗cd112r組合物及方法 |
| SG11202101152QA (en) | 2018-08-03 | 2021-03-30 | Chugai Pharmaceutical Co Ltd | Antigen-binding molecule containing two antigen-binding domains that are linked to each other |
| EP3608674A1 (en) | 2018-08-09 | 2020-02-12 | Regeneron Pharmaceuticals, Inc. | Methods for assessing binding affinity of an antibody variant to the neonatal fc receptor |
| SG11202003531WA (en) | 2018-08-10 | 2020-05-28 | Chugai Pharmaceutical Co Ltd | Anti-cd137 antigen-binding molecule and utilization thereof |
| JP2021534196A (ja) | 2018-08-23 | 2021-12-09 | シージェン インコーポレイテッド | 抗tigit抗体 |
| GB201814281D0 (en) | 2018-09-03 | 2018-10-17 | Femtogenix Ltd | Cytotoxic agents |
| AU2019342099A1 (en) | 2018-09-19 | 2021-04-08 | Genentech, Inc. | Therapeutic and diagnostic methods for bladder cancer |
| EP4249917A3 (en) | 2018-09-21 | 2023-11-08 | F. Hoffmann-La Roche AG | Diagnostic methods for triple-negative breast cancer |
| WO2020061560A1 (en) | 2018-09-21 | 2020-03-26 | The University Of North Carolina At Chapel Hill | Synthetic binding agents for limiting permeation through mucus |
| CN113164596A (zh) | 2018-09-24 | 2021-07-23 | 詹森生物科技公司 | 用抗il12/il23抗体治疗溃疡性结肠炎的安全且有效的方法 |
| CN113286812A (zh) | 2018-09-27 | 2021-08-20 | 西里欧发展公司 | 掩蔽型细胞因子多肽 |
| SG11202102477QA (en) | 2018-10-01 | 2021-04-29 | Hoffmann La Roche | Bispecific antigen binding molecules comprising anti-fap clone 212 |
| CN112654641A (zh) | 2018-10-01 | 2021-04-13 | 豪夫迈·罗氏有限公司 | 具有与cd40的三价结合的双特异性抗原结合分子 |
| EP3632929A1 (en) | 2018-10-02 | 2020-04-08 | Ospedale San Raffaele S.r.l. | Antibodies and uses thereof |
| US20220010020A1 (en) | 2018-10-05 | 2022-01-13 | Five Prime Therapeutics, Inc. | Anti-FGFR2 Antibody Formulations |
| US11130802B2 (en) | 2018-10-10 | 2021-09-28 | Tilos Therapeutics, Inc. | Anti-lap antibody variants |
| MX2021004226A (es) | 2018-10-15 | 2021-07-15 | Five Prime Therapeutics Inc | Terapia combinada contra el cáncer. |
| WO2020081493A1 (en) | 2018-10-16 | 2020-04-23 | Molecular Templates, Inc. | Pd-l1 binding proteins |
| CN113196061A (zh) | 2018-10-18 | 2021-07-30 | 豪夫迈·罗氏有限公司 | 肉瘤样肾癌的诊断和治疗方法 |
| US20210395390A1 (en) | 2018-10-31 | 2021-12-23 | Bayer Aktiengesellschaft | Reversal agents for neutralizing the therapeutic activity of anti-fxia antibodies |
| SG11202105010UA (en) | 2018-11-16 | 2021-06-29 | Memorial Sloan Kettering Cancer Center | Antibodies to mucin-16 and methods of use thereof |
| EP3883607A4 (en) | 2018-11-20 | 2022-08-17 | Janssen Biotech, Inc. | SAFE AND EFFECTIVE PROCESS FOR TREATING PSORIASIS WITH A SPECIFIC ANTI-IL-23 ANTIBODY |
| MX2021006578A (es) | 2018-12-05 | 2021-07-07 | Genentech Inc | Procedimientos de diagnostico y composiciones para la inmunoterapia contra el cancer. |
| US20220025051A1 (en) | 2018-12-07 | 2022-01-27 | Ono Pharmaceutical Co., Ltd. | Immunosuppresive agent |
| CN113227119A (zh) | 2018-12-10 | 2021-08-06 | 基因泰克公司 | 用于与含Fc的蛋白质进行位点特异性缀合的光交联肽 |
| MA54562A (fr) | 2018-12-18 | 2021-10-27 | Janssen Biotech Inc | Méthode sûre et efficace de traitement du lupus avec un anticorps anti-il12/il23 |
| US20220089694A1 (en) | 2018-12-20 | 2022-03-24 | The U.S.A., As Represented By The Secretary, Department Of Health And Human Services | Ebola virus glycoprotein-specific monoclonal antibodies and uses thereof |
| EP3898667A2 (en) | 2018-12-20 | 2021-10-27 | F. Hoffmann-La Roche AG | Modified antibody fcs and methods of use |
| EP3898671A1 (en) | 2018-12-21 | 2021-10-27 | F. Hoffmann-La Roche AG | Antibody that binds to vegf and il-1beta and methods of use |
| WO2020127618A1 (en) | 2018-12-21 | 2020-06-25 | F. Hoffmann-La Roche Ag | Tumor-targeted agonistic cd28 antigen binding molecules |
| SG11202102859TA (en) | 2018-12-21 | 2021-04-29 | Hoffmann La Roche | Antibodies binding to cd3 |
| AR117728A1 (es) | 2018-12-21 | 2021-08-25 | Hoffmann La Roche | Moléculas superagonistas de unión al antígeno cd28 con diana tumoral |
| MX2021007769A (es) | 2018-12-26 | 2021-09-23 | Xilio Dev Inc | Proteínas de unión anti-ctla4 enmascaradas activables. |
| JP2022515543A (ja) | 2018-12-30 | 2022-02-18 | エフ.ホフマン-ラ ロシュ アーゲー | 抗ウサギcd19抗体および使用方法 |
| CN115120716A (zh) | 2019-01-14 | 2022-09-30 | 健泰科生物技术公司 | 用pd-1轴结合拮抗剂和rna疫苗治疗癌症的方法 |
| CA3126778A1 (en) | 2019-01-17 | 2020-07-23 | Bayer Aktiengesellschaft | Methods to determine whether a subject is suitable of being treated with an agonist of soluble guanylyl cyclase (sgc) |
| CN113329770A (zh) | 2019-01-24 | 2021-08-31 | 中外制药株式会社 | 新型癌抗原及所述抗原的抗体 |
| GB201901197D0 (en) | 2019-01-29 | 2019-03-20 | Femtogenix Ltd | G-A Crosslinking cytotoxic agents |
| US20220096651A1 (en) | 2019-01-29 | 2022-03-31 | Juno Therapeutics, Inc. | Antibodies and chimeric antigen receptors specific for receptor tyrosine kinase like orphan receptor 1 (ror1) |
| CA3130695A1 (en) | 2019-02-27 | 2020-09-03 | Genentech, Inc. | Dosing for treatment with anti-tigit and anti-cd20 or anti-cd38 antibodies |
| MX2021010565A (es) | 2019-03-08 | 2021-10-13 | Genentech Inc | Metodos para detectar y cuantificar proteinas asociadas a la membrana en vesiculas extracelulares. |
| TW202100556A (zh) | 2019-03-14 | 2021-01-01 | 美商建南德克公司 | 使用her2 t細胞依賴性雙特異性抗體之治療 |
| CN113825765A (zh) | 2019-03-14 | 2021-12-21 | 詹森生物科技公司 | 用于制备抗il12/il23抗体组合物的制造方法 |
| AU2020243588A1 (en) | 2019-03-18 | 2021-10-07 | Janssen Biotech, Inc. | Method of treating psoriasis in pediatric subjects with anti-IL12/IL23 antibody |
| GB2589049C (en) | 2019-04-11 | 2024-02-21 | argenx BV | Anti-IgE antibodies |
| WO2020208049A1 (en) | 2019-04-12 | 2020-10-15 | F. Hoffmann-La Roche Ag | Bispecific antigen binding molecules comprising lipocalin muteins |
| AU2020257748A1 (en) | 2019-04-19 | 2021-11-18 | Chugai Seiyaku Kabushiki Kaisha | Chimeric receptor recognizing modification site of antibody |
| AU2020258480A1 (en) | 2019-04-19 | 2021-10-21 | Genentech, Inc. | Anti-mertk antibodies and their methods of use |
| WO2020227228A2 (en) | 2019-05-03 | 2020-11-12 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to plasmodium falciparum circumsporozoite protein and their use |
| CN114269376A (zh) | 2019-05-03 | 2022-04-01 | 豪夫迈·罗氏有限公司 | 用抗pd-l1抗体治疗癌症的方法 |
| CN114450022A (zh) | 2019-05-14 | 2022-05-06 | 狼人治疗公司 | 分离部分及其使用方法 |
| CN114206340A (zh) | 2019-05-14 | 2022-03-18 | 豪夫迈·罗氏有限公司 | 使用抗cd79b免疫缀合物治疗滤泡性淋巴瘤的方法 |
| US20230085439A1 (en) | 2019-05-21 | 2023-03-16 | University Of Georgia Research Foundation, Inc. | Antibodies that bind human metapneumovirus fusion protein and their use |
| AU2020278907A1 (en) | 2019-05-23 | 2022-01-20 | Ac Immune Sa | Anti-TDP-43 binding molecules and uses thereof |
| US11780911B2 (en) | 2019-05-23 | 2023-10-10 | Janssen Biotech, Inc. | Method of treating inflammatory bowel disease with a combination therapy of antibodies to IL-23 and TNF alpha |
| US11591388B2 (en) | 2019-06-07 | 2023-02-28 | argenx BV | Pharmaceutical formulations of FcRn inhibitors suitable for subcutaneous administration |
| JP2022538075A (ja) | 2019-06-26 | 2022-08-31 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | 抗体結合cea及び4-1bblの融合 |
| WO2020260326A1 (en) | 2019-06-27 | 2020-12-30 | F. Hoffmann-La Roche Ag | Novel icos antibodies and tumor-targeted antigen binding molecules comprising them |
| WO2021003297A1 (en) | 2019-07-02 | 2021-01-07 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Monoclonal antibodies that bind egfrviii and their use |
| CN114401992A (zh) | 2019-07-05 | 2022-04-26 | 艾欧麦克斯治疗股份公司 | 结合igsf11(vsig3)的igc2的抗体及其用途 |
| US20220267452A1 (en) | 2019-07-12 | 2022-08-25 | Chugai Seiyaku Kabushiki Kaisha | Anti-mutation type fgfr3 antibody and use therefor |
| AR119382A1 (es) | 2019-07-12 | 2021-12-15 | Hoffmann La Roche | Anticuerpos de pre-direccionamiento y métodos de uso |
| AR119393A1 (es) | 2019-07-15 | 2021-12-15 | Hoffmann La Roche | Anticuerpos que se unen a nkg2d |
| WO2021021605A1 (en) | 2019-07-26 | 2021-02-04 | Vanderbilt University | Human monoclonal antibodies to enterovirus d68 |
| CN114174338A (zh) | 2019-07-31 | 2022-03-11 | 豪夫迈·罗氏有限公司 | 与gprc5d结合的抗体 |
| CA3144524A1 (en) | 2019-07-31 | 2021-02-04 | F. Hoffmann-La Roche Ag | Antibodies binding to gprc5d |
| KR102509648B1 (ko) | 2019-08-06 | 2023-03-15 | 아프리노이아 테라퓨틱스 리미티드 | 병리학적 타우 종에 결합하는 항체 및 이의 용도 |
| CN114340675A (zh) | 2019-09-12 | 2022-04-12 | 豪夫迈·罗氏有限公司 | 治疗狼疮性肾炎的组合物和方法 |
| CA3150999A1 (en) | 2019-09-18 | 2021-03-25 | James Thomas Koerber | Anti-klk7 antibodies, anti-klk5 antibodies, multispecific anti-klk5/klk7 antibodies, and methods of use |
| JP2022548292A (ja) | 2019-09-19 | 2022-11-17 | ブリストル-マイヤーズ スクイブ カンパニー | 酸性pHでVISTAと結合する抗体 |
| JP2022549218A (ja) | 2019-09-20 | 2022-11-24 | ジェネンテック, インコーポレイテッド | 抗トリプターゼ抗体の投薬 |
| MX2022003610A (es) | 2019-09-27 | 2022-04-20 | Genentech Inc | Administracion de dosis para tratamiento con anticuerpos antagonistas anti-tigit y anti-pd-l1. |
| CN114945386A (zh) | 2019-10-18 | 2022-08-26 | 基因泰克公司 | 使用抗CD79b免疫缀合物治疗弥漫性大B细胞淋巴瘤的方法 |
| KR20220092580A (ko) | 2019-11-06 | 2022-07-01 | 제넨테크, 인크. | 혈액암의 치료를 위한 진단과 치료 방법 |
| JP2023501229A (ja) | 2019-11-15 | 2023-01-18 | エンテラ・エッセ・エッレ・エッレ | Tmem219抗体及びその治療的使用 |
| EP3822288A1 (en) | 2019-11-18 | 2021-05-19 | Deutsches Krebsforschungszentrum, Stiftung des öffentlichen Rechts | Antibodies targeting, and other modulators of, the cd276 antigen, and uses thereof |
| JP2023503105A (ja) | 2019-11-21 | 2023-01-26 | エンテラ・エッセ・エッレ・エッレ | Igfbp3抗体及びその治療的使用 |
| EP3831849A1 (en) | 2019-12-02 | 2021-06-09 | LamKap Bio beta AG | Bispecific antibodies against ceacam5 and cd47 |
| KR20220122653A (ko) | 2019-12-06 | 2022-09-02 | 주노 쎄러퓨티크스 인코퍼레이티드 | Bcma-표적 결합 도메인에 대한 항-이디오타입 항체 및 관련 조성물 및 방법 |
| EP4069742A1 (en) | 2019-12-06 | 2022-10-12 | Juno Therapeutics, Inc. | Anti-idiotypic antibodies to gprc5d-targeted binding domains and related compositions and methods |
| CR20220330A (es) | 2019-12-13 | 2022-08-04 | Genentech Inc | Anticuerpos anti-ly6g6d y métodos de uso. |
| PE20221282A1 (es) | 2019-12-18 | 2022-09-05 | Hoffmann La Roche | Anticuerpos que se unen a hla-a2/mage-a4 |
| PE20221585A1 (es) | 2019-12-27 | 2022-10-06 | Chugai Pharmaceutical Co Ltd | Anticuerpo anti antigeno-4 asociado al linfocito t citotoxico (ctla-4) y uso del mismo |
| CN113045655A (zh) | 2019-12-27 | 2021-06-29 | 高诚生物医药(香港)有限公司 | 抗ox40抗体及其用途 |
| BR112022013554A2 (pt) | 2020-01-08 | 2022-09-06 | argenx BV | Métodos para tratar distúrbios do pênfigo |
| AU2021206523A1 (en) | 2020-01-09 | 2022-05-26 | F. Hoffmann-La Roche Ag | New 4-1BBL trimer-containing antigen binding molecules |
| CN110818795B (zh) | 2020-01-10 | 2020-04-24 | 上海复宏汉霖生物技术股份有限公司 | 抗tigit抗体和使用方法 |
| WO2021194481A1 (en) | 2020-03-24 | 2021-09-30 | Genentech, Inc. | Dosing for treatment with anti-tigit and anti-pd-l1 antagonist antibodies |
| WO2022050954A1 (en) | 2020-09-04 | 2022-03-10 | Genentech, Inc. | Dosing for treatment with anti-tigit and anti-pd-l1 antagonist antibodies |
| JP2023513059A (ja) | 2020-01-31 | 2023-03-30 | ザ クリーブランド クリニック ファウンデーション | 抗ミューラー管ホルモン受容体2抗体及び使用方法 |
| JP2023512654A (ja) | 2020-01-31 | 2023-03-28 | ジェネンテック, インコーポレイテッド | Pd-1軸結合アンタゴニストおよびrnaワクチンを用いてネオエピトープ特異的t細胞を誘導する方法 |
| KR20220140568A (ko) | 2020-02-11 | 2022-10-18 | 벤더르빌트 유니버시티 | 중증 급성 호흡기 증후군 코로나바이러스 2(SARS-CoV-2)에 대한 인간 단일클론 항체 |
| TW202144395A (zh) | 2020-02-12 | 2021-12-01 | 日商中外製藥股份有限公司 | 用於癌症之治療的抗cd137抗原結合分子 |
| IL295023A (en) | 2020-02-14 | 2022-09-01 | Jounce Therapeutics Inc | Antibodies and fusion proteins that bind to ccr8 and their uses |
| EP4093762A1 (en) | 2020-02-20 | 2022-11-30 | The United States of America, as represented by the Secretary, Department of Health and Human Services | Epstein-barr virus monoclonal antibodies and uses thereof |
| EP3868396A1 (en) | 2020-02-20 | 2021-08-25 | Enthera S.R.L. | Inhibitors and uses thereof |
| AU2021225920A1 (en) | 2020-02-28 | 2022-09-15 | Shanghai Henlius Biotech, Inc. | Anti-CD137 construct and use thereof |
| CA3169910A1 (en) | 2020-02-28 | 2021-09-02 | Shanghai Henlius Biotech, Inc. | Anti-cd137 constructs, multispecific antibody and uses thereof |
| CA3174103A1 (en) | 2020-03-06 | 2021-09-10 | Go Therapeutics, Inc. | Anti-glyco-cd44 antibodies and their uses |
| CN115605507A (zh) | 2020-03-13 | 2023-01-13 | 基因泰克公司(Us) | 抗白介素-33抗体及其用途 |
| CN117510630A (zh) | 2020-03-19 | 2024-02-06 | 基因泰克公司 | 同种型选择性抗TGF-β抗体及使用方法 |
| JP2023518815A (ja) | 2020-03-23 | 2023-05-08 | ジェネンテック, インコーポレイテッド | Il6アンタゴニストによるcovid-19肺炎を含む肺炎の治療方法 |
| EP4107185A1 (en) | 2020-03-23 | 2022-12-28 | Genentech, Inc. | Biomarkers for predicting response to il-6 antagonist in covid-19 pneumonia |
| US20230174656A1 (en) | 2020-03-23 | 2023-06-08 | Genentech, Inc. | Tocilizumab and remdesivir combination therapy for covid-19 pneumonia |
| CR20220489A (es) | 2020-03-24 | 2022-10-31 | Genentech Inc | Agentes de fijación a tie2 y métodos de uso. |
| EP4127153A2 (en) | 2020-03-26 | 2023-02-08 | Genentech, Inc. | Modified mammalian cells having reduced host cell proteins |
| DK4045533T3 (da) | 2020-03-26 | 2024-02-12 | Univ Vanderbilt | Humane monoklonale antistoffer mod svær akut respiratorisk syndrom-coronavirus 2 (sars-cov-2) |
| WO2021195385A1 (en) | 2020-03-26 | 2021-09-30 | Vanderbilt University | HUMAN MONOCLONAL ANTIBODIES TO SEVERE ACUTE RESPIRATORY SYNDROME CORONAVIRUS 2 (SARS-GoV-2) |
| AR121706A1 (es) | 2020-04-01 | 2022-06-29 | Hoffmann La Roche | Moléculas de unión a antígeno biespecíficas dirigidas a ox40 y fap |
| WO2021202959A1 (en) | 2020-04-03 | 2021-10-07 | Genentech, Inc. | Therapeutic and diagnostic methods for cancer |
| EP4132971A1 (en) | 2020-04-09 | 2023-02-15 | Merck Sharp & Dohme LLC | Affinity matured anti-lap antibodies and uses thereof |
| WO2021207662A1 (en) | 2020-04-10 | 2021-10-14 | Genentech, Inc. | Use of il-22fc for the treatment or prevention of pneumonia, acute respiratory distress syndrome, or cytokine release syndrome |
| IL297541A (en) | 2020-04-24 | 2022-12-01 | Genentech Inc | Methods for using anti-cd79b immunoconjugates |
| JP2023523145A (ja) | 2020-04-27 | 2023-06-02 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | リポ蛋白(a)に対するイソ型非依存性抗体 |
| EP4143345A1 (en) | 2020-04-28 | 2023-03-08 | Genentech, Inc. | Methods and compositions for non-small cell lung cancer immunotherapy |
| EP4146283A1 (en) | 2020-05-03 | 2023-03-15 | Levena (Suzhou) Biopharma Co., Ltd. | Antibody-drug conjugates (adcs) comprising an anti-trop-2 antibody, compositions comprising such adcs, as well as methods of making and using the same |
| JP2023528235A (ja) | 2020-05-17 | 2023-07-04 | アストラゼネカ・ユーケイ・リミテッド | SARS-CoV-2抗体、並びにこれを選択及び使用する方法 |
| US20230220057A1 (en) | 2020-05-27 | 2023-07-13 | Staidson (Beijing) Biopharmaceuticals Co., Ltd. | Antibodies specifically recognizing nerve growth factor and uses thereof |
| CN116529260A (zh) | 2020-06-02 | 2023-08-01 | 当康生物技术有限责任公司 | 抗cd93构建体及其用途 |
| AU2021283345A1 (en) | 2020-06-02 | 2023-02-02 | Dynamicure Biotechnology Llc | Anti-CD93 constructs and uses thereof |
| AU2021288916A1 (en) | 2020-06-08 | 2022-11-24 | F.Hoffmann-La Roche Ag | Anti-HBV antibodies and methods of use |
| WO2021252977A1 (en) | 2020-06-12 | 2021-12-16 | Genentech, Inc. | Methods and compositions for cancer immunotherapy |
| WO2021257503A1 (en) | 2020-06-16 | 2021-12-23 | Genentech, Inc. | Methods and compositions for treating triple-negative breast cancer |
| MX2022015881A (es) | 2020-06-18 | 2023-01-24 | Genentech Inc | Tratamiento con anticuerpos anti-tigit y antagonistas de union al eje de pd-1. |
| WO2021255146A1 (en) | 2020-06-19 | 2021-12-23 | F. Hoffmann-La Roche Ag | Antibodies binding to cd3 and cea |
| KR20230025673A (ko) | 2020-06-19 | 2023-02-22 | 에프. 호프만-라 로슈 아게 | CD3 및 FolR1에 결합하는 항체 |
| WO2021255155A1 (en) | 2020-06-19 | 2021-12-23 | F. Hoffmann-La Roche Ag | Antibodies binding to cd3 and cd19 |
| IL296089A (en) | 2020-06-19 | 2022-11-01 | Hoffmann La Roche | Antibodies bind to cd3 |
| US20220017637A1 (en) | 2020-06-23 | 2022-01-20 | Hoffmann-La Roche Inc. | Agonistic cd28 antigen binding molecules targeting her2 |
| TW202216756A (zh) | 2020-06-24 | 2022-05-01 | 美商建南德克公司 | 具細胞凋亡抗性之細胞株 |
| WO2021260064A1 (en) | 2020-06-25 | 2021-12-30 | F. Hoffmann-La Roche Ag | Anti-cd3/anti-cd28 bispecific antigen binding molecules |
| WO2022008027A1 (en) | 2020-07-06 | 2022-01-13 | Iomx Therapeutics Ag | Antibodies binding igv of igsf11 (vsig3) and uses thereof |
| KR20230037578A (ko) | 2020-07-10 | 2023-03-16 | 에프. 호프만-라 로슈 아게 | 암 세포에 결합하고 방사성 핵종을 상기 세포로 표적화하는 항체 |
| TW202216780A (zh) | 2020-07-17 | 2022-05-01 | 美商建南德克公司 | 抗notch2抗體及其使用方法 |
| GB2597532A (en) | 2020-07-28 | 2022-02-02 | Femtogenix Ltd | Cytotoxic compounds |
| KR20230133832A (ko) | 2020-07-29 | 2023-09-19 | 다이내믹큐어 바이오테크놀로지 엘엘씨 | 항-cd93 구축물 및 그의 용도 |
| WO2022031749A1 (en) | 2020-08-03 | 2022-02-10 | Genentech, Inc. | Diagnostic and therapeutic methods for lymphoma |
| WO2022029660A1 (en) | 2020-08-05 | 2022-02-10 | Juno Therapeutics, Inc. | Anti-idiotypic antibodies to ror1-targeted binding domains and related compositions and methods |
| AU2021322239A1 (en) | 2020-08-07 | 2023-02-23 | Genentech, Inc. | Flt3 ligand fusion proteins and methods of use |
| KR20230045613A (ko) | 2020-08-10 | 2023-04-04 | 아스트라제네카 유케이 리미티드 | Covid-19의 치료 및 예방을 위한 sars-cov-2 항체 |
| AR123254A1 (es) | 2020-08-14 | 2022-11-16 | Ac Immune Sa | Moléculas de unión anti-tdp-43 humanizadas y usos de las mismas |
| WO2022043517A2 (en) | 2020-08-27 | 2022-03-03 | Cureab Gmbh | Anti-golph2 antibodies for macrophage and dendritic cell differentiation |
| CA3192344A1 (en) | 2020-08-28 | 2022-03-03 | Genentech, Inc. | Crispr/cas9 multiplex knockout of host cell proteins |
| EP4208201A1 (en) | 2020-09-04 | 2023-07-12 | F. Hoffmann-La Roche AG | Antibody that binds to vegf-a and ang2 and methods of use |
| CN116249719A (zh) | 2020-09-15 | 2023-06-09 | 拜耳公司 | 新的抗a2ap抗体及其用途 |
| EP4213877A1 (en) | 2020-09-17 | 2023-07-26 | Genentech, Inc. | Results of empacta: a randomized, double-blind, placebo-controlled, multicenter study to evaluate the efficacy and safety of tocilizumab in hospitalized patients with covid-19 pneumonia |
| JP2023544407A (ja) | 2020-10-05 | 2023-10-23 | ジェネンテック, インコーポレイテッド | 抗FcRH5/抗CD3二重特異性抗体による処置のための投与 |
| WO2022079211A1 (en) | 2020-10-16 | 2022-04-21 | Adc Therapeutics Sa | Glycoconjugates |
| EP4232475A1 (en) | 2020-10-20 | 2023-08-30 | Kantonsspital St. Gallen | Antibodies or antigen-binding fragments specifically binding to gremlin-1 and uses thereof |
| KR20230091871A (ko) | 2020-10-20 | 2023-06-23 | 에프. 호프만-라 로슈 아게 | Pd-1 축 결합 길항제 및 lrrk2 억제제의 병용 요법 |
| AR123855A1 (es) | 2020-10-20 | 2023-01-18 | Genentech Inc | Anticuerpos anti-mertk conjugados con peg y métodos de uso |
| WO2022093981A1 (en) | 2020-10-28 | 2022-05-05 | Genentech, Inc. | Combination therapy comprising ptpn22 inhibitors and pd-l1 binding antagonists |
| KR20230098317A (ko) | 2020-11-03 | 2023-07-03 | 도이체스크레브스포르슝스젠트룸스티프퉁데스외펜트리헨레크츠 | 표적 세포가 제한되는, 공동자극, 이중특이적 및 2가 항-cd28 항체 |
| IL302400A (en) | 2020-11-04 | 2023-06-01 | Genentech Inc | Subcutaneous dosing of bispecific anti-CD20/anti-CD3 antibodies |
| WO2022098638A2 (en) | 2020-11-04 | 2022-05-12 | Genentech, Inc. | Dosing for treatment with anti-cd20/anti-cd3 bispecific antibodies |
| EP4240493A2 (en) | 2020-11-04 | 2023-09-13 | Genentech, Inc. | Dosing for treatment with anti-cd20/anti-cd3 bispecific antibodies and anti-cd79b antibody drug conjugates |
| WO2022101458A1 (en) | 2020-11-16 | 2022-05-19 | F. Hoffmann-La Roche Ag | Combination therapy with fap-targeted cd40 agonists |
| JP2023551935A (ja) | 2020-12-02 | 2023-12-13 | グラクソスミスクライン、インテレクチュアル、プロパティー、ディベロップメント、リミテッド | Il-7結合タンパク質および医学的療法におけるそれらの使用 |
| TW202237639A (zh) | 2020-12-09 | 2022-10-01 | 日商武田藥品工業股份有限公司 | 鳥苷酸環化酶c(gcc)抗原結合劑之組成物及其使用方法 |
| TW202237638A (zh) | 2020-12-09 | 2022-10-01 | 日商武田藥品工業股份有限公司 | 烏苷酸環化酶c(gcc)抗原結合劑之組成物及其使用方法 |
| WO2022132904A1 (en) | 2020-12-17 | 2022-06-23 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Human monoclonal antibodies targeting sars-cov-2 |
| JP2024503826A (ja) | 2021-01-06 | 2024-01-29 | エフ・ホフマン-ラ・ロシュ・アクチェンゲゼルシャフト | Pd1-lag3二重特異性抗体及びcd20 t細胞二重特異性抗体を用いる併用療法 |
| EP4277705A1 (en) | 2021-01-12 | 2023-11-22 | F. Hoffmann-La Roche AG | Split antibodies which bind to cancer cells and target radionuclides to said cells |
| AU2022207624A1 (en) | 2021-01-13 | 2023-07-13 | F. Hoffmann-La Roche Ag | Combination therapy |
| WO2022162587A1 (en) | 2021-01-27 | 2022-08-04 | Centre Hospitalier Universitaire Vaudois (C.H.U.V.) | Anti-sars-cov-2 antibodies and use thereof in the treatment of sars-cov-2 infection |
| WO2022162203A1 (en) | 2021-01-28 | 2022-08-04 | Vaccinvent Gmbh | Method and means for modulating b-cell mediated immune responses |
| CN117120084A (zh) | 2021-01-28 | 2023-11-24 | 维肯芬特有限责任公司 | 用于调节b细胞介导的免疫应答的方法和手段 |
| EP4284422A1 (en) | 2021-01-28 | 2023-12-06 | Vaccinvent GmbH | Method and means for modulating b-cell mediated immune responses |
| EP4288458A1 (en) | 2021-02-03 | 2023-12-13 | Genentech, Inc. | Multispecific binding protein degrader platform and methods of use |
| IL304955A (en) | 2021-02-09 | 2023-10-01 | Us Health | Antibodies targeting the spike protein of a coronavirus |
| CN117396502A (zh) | 2021-02-09 | 2024-01-12 | 佐治亚大学研究基金会有限公司 | 针对肺炎球菌抗原的人类单克隆抗体 |
| CA3208365A1 (en) | 2021-02-15 | 2022-08-18 | Chantal KUHN | Cell therapy compositions and methods for modulating tgf-b signaling |
| GB202102396D0 (en) | 2021-02-19 | 2021-04-07 | Adc Therapeutics Sa | Molecular adjuvant |
| IL305131A (en) | 2021-02-26 | 2023-10-01 | Bayer Ag | IL-11 or IL-11Ra inhibitors for use in the treatment of abnormal uterine bleeding |
| CA3209364A1 (en) | 2021-03-01 | 2022-09-09 | Jennifer O'neil | Combination of masked ctla4 and pd1/pdl1 antibodies for treating cancer |
| US20220306743A1 (en) | 2021-03-01 | 2022-09-29 | Xilio Development, Inc. | Combination of ctla4 and pd1/pdl1 antibodies for treating cancer |
| JP2024509169A (ja) | 2021-03-03 | 2024-02-29 | ソレント・セラピューティクス・インコーポレイテッド | 抗bcma抗体を含む抗体-薬物コンジュゲート |
| JP2024512324A (ja) | 2021-03-05 | 2024-03-19 | ジーオー セラピューティクス,インコーポレイテッド | 抗グリコcd44抗体およびその使用 |
| JP2024509191A (ja) | 2021-03-05 | 2024-02-29 | ダイナミキュア バイオテクノロジー エルエルシー | 抗vista構築物およびその使用 |
| CA3212729A1 (en) | 2021-03-12 | 2022-09-15 | Janssen Biotech, Inc. | Safe and effective method of treating psoriatic arthritis with anti-il23 specific antibody |
| AR125074A1 (es) | 2021-03-12 | 2023-06-07 | Genentech Inc | Anticuerpos anti-klk7, anticuerpos anti-klk5, anticuerpos multiespecíficos anti-klk5 / klk7 y métodos de uso |
| CA3213278A1 (en) | 2021-03-12 | 2022-09-15 | Janssen Biotech, Inc. | Method of treating psoriatic arthritis patients with inadequate response to tnf therapy with anti-il23 specific antibody |
| CA3213599A1 (en) | 2021-03-15 | 2022-09-22 | Genentech, Inc. | Compositions and methods of treating lupus nephritis |
| WO2022197877A1 (en) | 2021-03-19 | 2022-09-22 | Genentech, Inc. | Methods and compositions for time delayed bio-orthogonal release of cytotoxic agents |
| TW202300648A (zh) | 2021-03-25 | 2023-01-01 | 美商當康生物科技有限公司 | 抗-igfbp7構築體及其用途 |
| US20220389096A1 (en) | 2021-03-30 | 2022-12-08 | Bayer Aktiengesellschaft | Novel anti-sema3a antibodies and uses thereof |
| AR125344A1 (es) | 2021-04-15 | 2023-07-05 | Chugai Pharmaceutical Co Ltd | Anticuerpo anti-c1s |
| TW202305122A (zh) | 2021-04-19 | 2023-02-01 | 美商建南德克公司 | 經修飾之哺乳動物細胞 |
| EP4326271A1 (en) | 2021-04-23 | 2024-02-28 | F. Hoffmann-La Roche AG | Prevention or mitigation of nk cell engaging agent-related adverse effects |
| WO2022228705A1 (en) | 2021-04-30 | 2022-11-03 | F. Hoffmann-La Roche Ag | Dosing for combination treatment with anti-cd20/anti-cd3 bispecific antibody and anti-cd79b antibody drug conjugate |
| CA3218170A1 (en) | 2021-05-12 | 2022-11-17 | Jamie Harue HIRATA | Methods of using anti-cd79b immunoconjugates to treat diffuse large b-cell lymphoma |
| US20220389097A1 (en) | 2021-05-14 | 2022-12-08 | Genentech Inc. | Agonists of TREM2 |
| WO2022243261A1 (en) | 2021-05-19 | 2022-11-24 | F. Hoffmann-La Roche Ag | Agonistic cd40 antigen binding molecules targeting cea |
| KR20240010469A (ko) | 2021-05-21 | 2024-01-23 | 제넨테크, 인크. | 관심 재조합 생성물의 생성을 위한 변형된 세포 |
| EP4347656A1 (en) | 2021-05-28 | 2024-04-10 | GlaxoSmithKline Intellectual Property Development Limited | Combination therapies for treating cancer |
| AR126009A1 (es) | 2021-06-02 | 2023-08-30 | Hoffmann La Roche | Moléculas agonistas de unión al antígeno cd28 que se dirigen a epcam |
| TW202306994A (zh) | 2021-06-04 | 2023-02-16 | 日商中外製藥股份有限公司 | 抗ddr2抗體及其用途 |
| CA3216220A1 (en) | 2021-06-09 | 2022-12-15 | F. Hoffmann-La Roche Ag | Combination of a particular braf inhibitor (paradox breaker) and a pd-1 axis binding antagonist for use in the treatment of cancer |
| CA3221924A1 (en) | 2021-06-11 | 2022-12-15 | Genentech, Inc. | Method for treating chronic obstructive pulmonary disease with an st2 antagonist |
| WO2022263638A1 (en) | 2021-06-17 | 2022-12-22 | Centre Hospitalier Universitaire Vaudois (C.H.U.V.) | Anti-sars-cov-2 antibodies and use thereof in the treatment of sars-cov-2 infection |
| WO2022266660A1 (en) | 2021-06-17 | 2022-12-22 | Amberstone Biosciences, Inc. | Anti-cd3 constructs and uses thereof |
| WO2022270611A1 (ja) | 2021-06-25 | 2022-12-29 | 中外製薬株式会社 | 抗ctla-4抗体 |
| BR112023023480A2 (pt) | 2021-06-25 | 2024-01-30 | Chugai Pharmaceutical Co Ltd | Uso de anticorpo anti-ctla-4 |
| TW202317633A (zh) | 2021-07-08 | 2023-05-01 | 美商舒泰神(加州)生物科技有限公司 | 特異性識別tnfr2的抗體及其用途 |
| AU2022306973A1 (en) | 2021-07-09 | 2024-02-22 | Janssen Biotech, Inc. | Manufacturing methods for producing anti-il12/il23 antibody compositions |
| WO2023288182A1 (en) | 2021-07-12 | 2023-01-19 | Genentech, Inc. | Structures for reducing antibody-lipase binding |
| CN115812082A (zh) | 2021-07-14 | 2023-03-17 | 舒泰神(北京)生物制药股份有限公司 | 特异性识别cd40的抗体及其应用 |
| US20230049152A1 (en) | 2021-07-14 | 2023-02-16 | Genentech, Inc. | Anti-c-c motif chemokine receptor 8 (ccr8) antibodies and methods of use |
| WO2023004386A1 (en) | 2021-07-22 | 2023-01-26 | Genentech, Inc. | Brain targeting compositions and methods of use thereof |
| WO2023001884A1 (en) | 2021-07-22 | 2023-01-26 | F. Hoffmann-La Roche Ag | Heterodimeric fc domain antibodies |
| CN117794953A (zh) | 2021-08-03 | 2024-03-29 | 豪夫迈·罗氏有限公司 | 双特异性抗体及使用方法 |
| WO2023014863A1 (en) | 2021-08-05 | 2023-02-09 | Go Therapeutics, Inc. | Anti-glyco-muc4 antibodies and their uses |
| WO2023019092A1 (en) | 2021-08-07 | 2023-02-16 | Genentech, Inc. | Methods of using anti-cd79b immunoconjugates to treat diffuse large b-cell lymphoma |
| AU2022327742A1 (en) | 2021-08-13 | 2024-03-14 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for ccr2-expressing cells |
| CA3227835A1 (en) | 2021-08-13 | 2023-02-16 | Peiling CHEN | Cytotoxicity targeting chimeras |
| WO2023019239A1 (en) | 2021-08-13 | 2023-02-16 | Genentech, Inc. | Dosing for anti-tryptase antibodies |
| WO2023021055A1 (en) | 2021-08-19 | 2023-02-23 | F. Hoffmann-La Roche Ag | Multivalent anti-variant fc-region antibodies and methods of use |
| AU2022332285A1 (en) | 2021-08-23 | 2024-02-15 | Immunitas Therapeutics, Inc. | Anti-cd161 antibodies and uses thereof |
| IL310382A (en) | 2021-08-27 | 2024-03-01 | Genentech Inc | Methods for treating tau pathologies |
| WO2023034750A1 (en) | 2021-08-30 | 2023-03-09 | Genentech, Inc. | Anti-polyubiquitin multispecific antibodies |
| WO2023034569A1 (en) | 2021-09-03 | 2023-03-09 | Go Therapeutics, Inc. | Anti-glyco-cmet antibodies and their uses |
| TW202325733A (zh) | 2021-09-03 | 2023-07-01 | 美商Go治療公司 | 抗醣化-lamp1抗體及其用途 |
| AU2022345251A1 (en) | 2021-09-17 | 2024-03-28 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Synthetic humanized llama nanobody library and use thereof to identify sars-cov-2 neutralizing antibodies |
| WO2023056403A1 (en) | 2021-09-30 | 2023-04-06 | Genentech, Inc. | Methods for treatment of hematologic cancers using anti-tigit antibodies, anti-cd38 antibodies, and pd-1 axis binding antagonists |
| CA3233953A1 (en) | 2021-10-05 | 2023-04-13 | Matthew Bruce | Combination therapies for treating cancer |
| WO2023073615A1 (en) | 2021-10-29 | 2023-05-04 | Janssen Biotech, Inc. | Methods of treating crohn's disease with anti-il23 specific antibody |
| WO2023081818A1 (en) | 2021-11-05 | 2023-05-11 | American Diagnostics & Therapy, Llc (Adxrx) | Monoclonal antibodies against carcinoembryonic antigens, and their uses |
| TW202342095A (zh) | 2021-11-05 | 2023-11-01 | 英商阿斯特捷利康英國股份有限公司 | 用於治療和預防covid—19之組成物 |
| WO2023086807A1 (en) | 2021-11-10 | 2023-05-19 | Genentech, Inc. | Anti-interleukin-33 antibodies and uses thereof |
| WO2023084488A1 (en) | 2021-11-15 | 2023-05-19 | Janssen Biotech, Inc. | Methods of treating crohn's disease with anti-il23 specific antibody |
| TW202337494A (zh) | 2021-11-16 | 2023-10-01 | 美商建南德克公司 | 用莫蘇妥珠單抗治療全身性紅斑狼瘡(sle)之方法及組成物 |
| WO2023088959A1 (en) | 2021-11-16 | 2023-05-25 | Ac Immune Sa | Novel molecules for therapy and diagnosis |
| WO2023095000A1 (en) | 2021-11-23 | 2023-06-01 | Janssen Biotech, Inc. | Method of treating ulcerative colitis with anti-il23 specific antibody |
| WO2023094569A1 (en) | 2021-11-26 | 2023-06-01 | F. Hoffmann-La Roche Ag | Combination therapy of anti-tyrp1/anti-cd3 bispecific antibodies and tyrp1-specific antibodies |
| AR127887A1 (es) | 2021-12-10 | 2024-03-06 | Hoffmann La Roche | Anticuerpos que se unen a cd3 y plap |
| TW202340248A (zh) | 2021-12-20 | 2023-10-16 | 瑞士商赫孚孟拉羅股份公司 | 促效性ltbr抗體及包含其之雙特異性抗體 |
| US20230227545A1 (en) | 2022-01-07 | 2023-07-20 | Johnson & Johnson Enterprise Innovation Inc. | Materials and methods of il-1beta binding proteins |
| US20230322958A1 (en) | 2022-01-19 | 2023-10-12 | Genentech, Inc. | Anti-Notch2 Antibodies and Conjugates and Methods of Use |
| WO2023154824A1 (en) | 2022-02-10 | 2023-08-17 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Human monoclonal antibodies that broadly target coronaviruses |
| WO2023156549A1 (en) | 2022-02-16 | 2023-08-24 | Ac Immune Sa | Humanized anti-tdp-43 binding molecules and uses thereof |
| WO2023159182A1 (en) | 2022-02-18 | 2023-08-24 | Rakuten Medical, Inc. | Anti-programmed death-ligand 1 (pd-l1) antibody molecules, encoding polynucleotides, and methods of use |
| WO2023161876A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for cxcr3-expressing cells |
| WO2023161879A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for fibroblast activation protein-expressing cells |
| WO2023161875A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for prostate specific membrane antigen-expressing cells |
| WO2023161877A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for integrin avb6-expressing cells |
| WO2023161881A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for ccr2-expressing cells |
| WO2023161874A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for c-c chemokine receptor 2-expressing cells |
| WO2023161878A1 (en) | 2022-02-25 | 2023-08-31 | Glaxosmithkline Intellectual Property Development Limited | Cytotoxicity targeting chimeras for folate receptor-expressing cells |
| WO2023173026A1 (en) | 2022-03-10 | 2023-09-14 | Sorrento Therapeutics, Inc. | Antibody-drug conjugates and uses thereof |
| TW202346365A (zh) | 2022-03-23 | 2023-12-01 | 瑞士商赫孚孟拉羅股份公司 | 抗cd20/抗cd3雙特異性抗體及化學療法之組合治療 |
| WO2023192827A1 (en) | 2022-03-26 | 2023-10-05 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Bispecific antibodies to hiv-1 env and their use |
| WO2023192881A1 (en) | 2022-03-28 | 2023-10-05 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to hiv-1 env and their use |
| WO2023186756A1 (en) | 2022-03-28 | 2023-10-05 | F. Hoffmann-La Roche Ag | Interferon gamma variants and antigen binding molecules comprising these |
| WO2023187707A1 (en) | 2022-03-30 | 2023-10-05 | Janssen Biotech, Inc. | Method of treating mild to moderate psoriasis with il-23 specific antibody |
| GB202204813D0 (en) | 2022-04-01 | 2022-05-18 | Bradcode Ltd | Human monoclonal antibodies and methods of use thereof |
| WO2023191816A1 (en) | 2022-04-01 | 2023-10-05 | Genentech, Inc. | Dosing for treatment with anti-fcrh5/anti-cd3 bispecific antibodies |
| WO2023194565A1 (en) | 2022-04-08 | 2023-10-12 | Ac Immune Sa | Anti-tdp-43 binding molecules |
| WO2023198727A1 (en) | 2022-04-13 | 2023-10-19 | F. Hoffmann-La Roche Ag | Pharmaceutical compositions of anti-cd20/anti-cd3 bispecific antibodies and methods of use |
| WO2023201299A1 (en) | 2022-04-13 | 2023-10-19 | Genentech, Inc. | Pharmaceutical compositions of therapeutic proteins and methods of use |
| WO2023203177A1 (en) | 2022-04-20 | 2023-10-26 | Kantonsspital St. Gallen | Antibodies or antigen-binding fragments pan-specifically binding to gremlin-1 and gremlin-2 and uses thereof |
| WO2023209177A1 (en) | 2022-04-29 | 2023-11-02 | Astrazeneca Uk Limited | Sars-cov-2 antibodies and methods of using the same |
| US20230348604A1 (en) | 2022-04-29 | 2023-11-02 | 23Andme, Inc. | Antigen binding proteins |
| WO2023215737A1 (en) | 2022-05-03 | 2023-11-09 | Genentech, Inc. | Anti-ly6e antibodies, immunoconjugates, and uses thereof |
| WO2023217933A1 (en) | 2022-05-11 | 2023-11-16 | F. Hoffmann-La Roche Ag | Antibody that binds to vegf-a and il6 and methods of use |
| WO2023219613A1 (en) | 2022-05-11 | 2023-11-16 | Genentech, Inc. | Dosing for treatment with anti-fcrh5/anti-cd3 bispecific antibodies |
| US20230374122A1 (en) | 2022-05-18 | 2023-11-23 | Janssen Biotech, Inc. | Method for Evaluating and Treating Psoriatic Arthritis with IL23 Antibody |
| WO2023227641A1 (en) | 2022-05-27 | 2023-11-30 | Glaxosmithkline Intellectual Property Development Limited | Use of tnf-alpha binding proteins and il-7 binding proteins in medical treatment |
| WO2023235699A1 (en) | 2022-05-31 | 2023-12-07 | Jounce Therapeutics, Inc. | Antibodies to lilrb4 and uses thereof |
| WO2023240058A2 (en) | 2022-06-07 | 2023-12-14 | Genentech, Inc. | Prognostic and therapeutic methods for cancer |
| WO2023237706A2 (en) | 2022-06-08 | 2023-12-14 | Institute For Research In Biomedicine (Irb) | Cross-specific antibodies, uses and methods for discovery thereof |
| WO2024015897A1 (en) | 2022-07-13 | 2024-01-18 | Genentech, Inc. | Dosing for treatment with anti-fcrh5/anti-cd3 bispecific antibodies |
| WO2024020407A1 (en) | 2022-07-19 | 2024-01-25 | Staidson Biopharma Inc. | Antibodies specifically recognizing b- and t-lymphocyte attenuator (btla) and uses thereof |
| WO2024020432A1 (en) | 2022-07-19 | 2024-01-25 | Genentech, Inc. | Dosing for treatment with anti-fcrh5/anti-cd3 bispecific antibodies |
| WO2024020564A1 (en) | 2022-07-22 | 2024-01-25 | Genentech, Inc. | Anti-steap1 antigen-binding molecules and uses thereof |
| WO2024020579A1 (en) | 2022-07-22 | 2024-01-25 | Bristol-Myers Squibb Company | Antibodies binding to human pad4 and uses thereof |
| WO2024030829A1 (en) | 2022-08-01 | 2024-02-08 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Monoclonal antibodies that bind to the underside of influenza viral neuraminidase |
| WO2024028732A1 (en) | 2022-08-05 | 2024-02-08 | Janssen Biotech, Inc. | Cd98 binding constructs for treating brain tumors |
| WO2024028731A1 (en) | 2022-08-05 | 2024-02-08 | Janssen Biotech, Inc. | Transferrin receptor binding proteins for treating brain tumors |
| WO2024042112A1 (en) | 2022-08-25 | 2024-02-29 | Glaxosmithkline Intellectual Property Development Limited | Antigen binding proteins and uses thereof |
| WO2024044779A2 (en) | 2022-08-26 | 2024-02-29 | Juno Therapeutics, Inc. | Antibodies and chimeric antigen receptors specific for delta-like ligand 3 (dll3) |
| WO2024049949A1 (en) | 2022-09-01 | 2024-03-07 | Genentech, Inc. | Therapeutic and diagnostic methods for bladder cancer |
| WO2024054929A1 (en) | 2022-09-07 | 2024-03-14 | Dynamicure Biotechnology Llc | Anti-vista constructs and uses thereof |
| WO2024054822A1 (en) | 2022-09-07 | 2024-03-14 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Engineered sars-cov-2 antibodies with increased neutralization breadth |
| WO2024064826A1 (en) | 2022-09-22 | 2024-03-28 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Neutralizing antibodies to plasmodium falciparum circumsporozoite protein and their use |
| WO2024068996A1 (en) | 2022-09-30 | 2024-04-04 | Centre Hospitalier Universitaire Vaudois (C.H.U.V.) | Anti-sars-cov-2 antibodies and use thereof in the treatment of sars-cov-2 infection |
Family Cites Families (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4215051A (en) * | 1979-08-29 | 1980-07-29 | Standard Oil Company (Indiana) | Formation, purification and recovery of phthalic anhydride |
| KR850004274A (ko) * | 1983-12-13 | 1985-07-11 | 원본미기재 | 에리트로포이에틴의 제조방법 |
| US4946778A (en) | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| US4978745A (en) * | 1987-11-23 | 1990-12-18 | Centocor, Inc. | Immunoreactive heterochain antibodies |
| US5047335A (en) * | 1988-12-21 | 1991-09-10 | The Regents Of The University Of Calif. | Process for controlling intracellular glycosylation of proteins |
| WO1990014104A1 (en) * | 1989-05-25 | 1990-11-29 | Sloan-Kettering Institute For Cancer Research | Anti-idiotypic antibody which induces an immune response against a glycosphingolipid and use thereof |
| DE4028800A1 (de) | 1990-09-11 | 1992-03-12 | Behringwerke Ag | Gentechnische sialylierung von glykoproteinen |
| US5665569A (en) | 1991-08-22 | 1997-09-09 | Nissin Shokuhin Kabushiki Kaisha | HIV immunotherapeutics |
| US5753229A (en) * | 1991-09-25 | 1998-05-19 | Mordoh; Jose | Monoclonal antibodies reactive with tumor proliferating cells |
| US5958403A (en) * | 1992-02-28 | 1999-09-28 | Beth Israel Hospital Association | Methods and compounds for prevention of graft rejection |
| EP0752248B1 (en) * | 1992-11-13 | 2000-09-27 | Idec Pharmaceuticals Corporation | Therapeutic application of chimeric and radiolabeled antibodies to human B lymphocyte restricted differentiation antigen for treatment of B cell lymphoma |
| US5736137A (en) * | 1992-11-13 | 1998-04-07 | Idec Pharmaceuticals Corporation | Therapeutic application of chimeric and radiolabeled antibodies to human B lymphocyte restricted differentiation antigen for treatment of B cell lymphoma |
| DK0678122T3 (da) | 1993-01-12 | 2000-03-06 | Biogen Inc | Rekombinante anti-VLA4 antistofmolekyler |
| WO1995024494A1 (en) | 1994-03-09 | 1995-09-14 | Abbott Laboratories | Humanized milk |
| US5811524A (en) * | 1995-06-07 | 1998-09-22 | Idec Pharmaceuticals Corporation | Neutralizing high affinity human monoclonal antibodies specific to RSV F-protein and methods for their manufacture and therapeutic use thereof |
| JP3606536B2 (ja) * | 1995-11-17 | 2005-01-05 | タカラバイオ株式会社 | ウイルス複製抑制剤 |
| GB9603256D0 (en) * | 1996-02-16 | 1996-04-17 | Wellcome Found | Antibodies |
| AU4231897A (en) | 1996-08-16 | 1998-03-06 | Southwest Foundation For Biomedical Research | Compositions and methods for delivery of nucleic acids to hepatocytes |
| US6183744B1 (en) * | 1997-03-24 | 2001-02-06 | Immunomedics, Inc. | Immunotherapy of B-cell malignancies using anti-CD22 antibodies |
| US6306393B1 (en) | 1997-03-24 | 2001-10-23 | Immunomedics, Inc. | Immunotherapy of B-cell malignancies using anti-CD22 antibodies |
| US5952203A (en) * | 1997-04-11 | 1999-09-14 | The University Of British Columbia | Oligosaccharide synthesis using activated glycoside derivative, glycosyl transferase and catalytic amount of nucleotide phosphate |
| EP2180007B2 (en) | 1998-04-20 | 2017-08-30 | Roche Glycart AG | Glycosylation engineering of antibodies for improving antibody-dependent cellular cytotoxicity |
| US6946292B2 (en) | 2000-10-06 | 2005-09-20 | Kyowa Hakko Kogyo Co., Ltd. | Cells producing antibody compositions with increased antibody dependent cytotoxic activity |
| WO2002079255A1 (en) * | 2001-04-02 | 2002-10-10 | Idec Pharmaceuticals Corporation | RECOMBINANT ANTIBODIES COEXPRESSED WITH GnTIII |
| NZ581474A (en) | 2001-08-03 | 2011-04-29 | Glycart Biotechnology Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| MXPA05007781A (es) | 2003-01-22 | 2005-09-30 | Glycart Biotechnology Ag | Constructos de fusion y uso de los mismos para producir anticuerpos con afinidad incrementada de enlace al receptor fc y funcion efectora. |
| SG10202008722QA (en) | 2003-11-05 | 2020-10-29 | Roche Glycart Ag | Cd20 antibodies with increased fc receptor binding affinity and effector function |
| BRPI0607315B1 (pt) | 2005-02-07 | 2022-05-17 | Roche Glycart Ag | Molécula de ligação ao antígeno anti-egfr humanizada e seus usos, composição farmacêutica, e método para detectar a presença de egfr em uma amostra |
| TWI615407B (zh) | 2005-08-26 | 2018-02-21 | 羅齊克雷雅公司 | 具有經改變細胞傳訊活性之改質抗原結合分子 |
| AR062223A1 (es) | 2006-08-09 | 2008-10-22 | Glycart Biotechnology Ag | Moleculas de adhesion al antigeno que se adhieren a egfr, vectores que los codifican, y sus usos de estas |
| SG178567A1 (en) | 2009-08-31 | 2012-04-27 | Roche Glycart Ag | Affinity-matured humanized anti cea monoclonal antibodies |
-
2002
- 2002-08-05 NZ NZ581474A patent/NZ581474A/en not_active IP Right Cessation
- 2002-08-05 NZ NZ603111A patent/NZ603111A/en not_active IP Right Cessation
- 2002-08-05 NZ NZ571596A patent/NZ571596A/en not_active IP Right Cessation
- 2002-08-05 US US10/211,554 patent/US20030175884A1/en not_active Abandoned
- 2002-08-05 KR KR1020107000746A patent/KR20100018071A/ko active Search and Examination
- 2002-08-05 WO PCT/US2002/024739 patent/WO2003011878A2/en active Application Filing
- 2002-08-05 CA CA2838062A patent/CA2838062C/en not_active Expired - Lifetime
- 2002-08-05 AU AU2002339845A patent/AU2002339845B2/en not_active Expired
- 2002-08-05 CA CA2455365A patent/CA2455365C/en not_active Expired - Lifetime
- 2002-08-05 PL PL374178A patent/PL217751B1/pl unknown
- 2002-08-05 JP JP2003517069A patent/JP2005524379A/ja active Pending
- 2002-08-05 EP EP02778191A patent/EP1423510A4/en not_active Withdrawn
- 2002-08-05 NZ NZ592087A patent/NZ592087A/xx not_active IP Right Cessation
- 2002-08-05 IL IL16017002A patent/IL160170A0/xx unknown
- 2002-08-05 CN CNA028181735A patent/CN1555411A/zh active Pending
- 2002-08-05 HU HU0700103A patent/HUP0700103A3/hu not_active Application Discontinuation
- 2002-08-05 KR KR10-2004-7001617A patent/KR20040054669A/ko not_active Application Discontinuation
- 2002-08-05 MX MXPA04001072A patent/MXPA04001072A/es active IP Right Grant
- 2002-08-05 RU RU2004106559/13A patent/RU2321630C2/ru active
- 2002-08-05 EP EP20100000043 patent/EP2180044A1/en not_active Ceased
- 2002-08-05 NZ NZ531219A patent/NZ531219A/en not_active IP Right Cessation
-
2004
- 2004-02-02 NO NO20040453A patent/NO332457B1/no not_active IP Right Cessation
- 2004-02-02 IL IL160170A patent/IL160170A/en unknown
-
2005
- 2005-08-09 US US11/199,232 patent/US8021856B2/en not_active Expired - Fee Related
-
2008
- 2008-12-25 JP JP2008331038A patent/JP2009114201A/ja active Pending
-
2011
- 2011-08-02 US US13/196,724 patent/US8999324B2/en not_active Expired - Fee Related
-
2015
- 2015-03-23 US US14/665,191 patent/US9321843B2/en not_active Expired - Fee Related
-
2016
- 2016-03-24 US US15/080,020 patent/US9631023B2/en not_active Expired - Fee Related
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8021856B2 (en) | 1998-04-20 | 2011-09-20 | Roche Glycart Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| US8999324B2 (en) | 1998-04-20 | 2015-04-07 | Roche Glycart Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| US9321843B2 (en) | 1998-04-20 | 2016-04-26 | Roche Glycart Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
| US9631023B2 (en) | 1998-04-20 | 2017-04-25 | Roche Glycart Ag | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1555411A (zh) | 抗体-依赖性细胞毒性增大的抗体糖基化变体 | |
| AU2002339845A1 (en) | Antibody glycosylation variants having increased antibody-dependent cellular cytotoxicity | |
| CN102782131B (zh) | 修饰抗体组合物 | |
| JP2002512014A (ja) | 抗体依存性細胞傷害性を改善するための抗体のグリコシル化操作 | |
| CN103540600A (zh) | 融合构建体及其用来生产Fc受体结合亲和性和效应子功能提高的抗体的用途 | |
| JP7216806B2 (ja) | アフコシル化抗体およびその製造法 | |
| WO2018041067A1 (en) | Anti-cd20 antibody | |
| JP7292377B2 (ja) | アフコシル化抗体およびその製造法 | |
| TWI748124B (zh) | 去岩藻醣基化抗體的製造方法 | |
| TWI745615B (zh) | 去岩藻醣基化抗體及表現此抗體的細胞株 | |
| CN116903738A (zh) | 一种低甘露糖型抗人肿瘤坏死因子-α单抗及其用途 | |
| CN116615231A (zh) | Fab高甘露糖糖型 | |
| Costa | Expression and characterization of a therapeutic monoclonal antibody in mammalian cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C12 | Rejection of a patent application after its publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20041215 |