EP0651663B1 - Verfahren für nadellose subkutaninjektionen - Google Patents
Verfahren für nadellose subkutaninjektionen Download PDFInfo
- Publication number
- EP0651663B1 EP0651663B1 EP93918344A EP93918344A EP0651663B1 EP 0651663 B1 EP0651663 B1 EP 0651663B1 EP 93918344 A EP93918344 A EP 93918344A EP 93918344 A EP93918344 A EP 93918344A EP 0651663 B1 EP0651663 B1 EP 0651663B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- ampule
- valve
- pressure
- poppet
- reservoir
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/30—Syringes for injection by jet action, without needle, e.g. for use with replaceable ampoules or carpules
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/31—Details
- A61M5/315—Pistons; Piston-rods; Guiding, blocking or restricting the movement of the rod or piston; Appliances on the rod for facilitating dosing ; Dosing mechanisms
- A61M5/31511—Piston or piston-rod constructions, e.g. connection of piston with piston-rod
- A61M2005/31516—Piston or piston-rod constructions, e.g. connection of piston with piston-rod reducing dead-space in the syringe barrel after delivery
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M2205/00—General characteristics of the apparatus
- A61M2205/58—Means for facilitating use, e.g. by people with impaired vision
- A61M2205/583—Means for facilitating use, e.g. by people with impaired vision by visual feedback
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M5/00—Devices for bringing media into the body in a subcutaneous, intra-vascular or intramuscular way; Accessories therefor, e.g. filling or cleaning devices, arm-rests
- A61M5/178—Syringes
- A61M5/20—Automatic syringes, e.g. with automatically actuated piston rod, with automatic needle injection, filling automatically
- A61M5/2053—Media being expelled from injector by pressurised fluid or vacuum
Definitions
- the present invention relates to a needleless hypodermic injection method.
- Various needleless hypodermic injection devices have been known and used in the past for performing the method according to the invention. These devices, also known as jet injectors, typically use spring or compressed gas driven plungers to accelerate an injectant to a velocity sufficient to pierce through the skin and enter the underlying tissues.
- US-A-4722728 discloses an injector which pressurizes a fluid injectant within an ampule by driving a plunger into the ampule with sufficient force to expel the desired volume of fluid.
- Jet injection avoids or diminishes these disadvantages.
- the characteristics of needleless or jet injections typically vary with the pressures exerted by the injection device, the nozzle diameter of the ampule, the patient's size, age and weight, the nature of the injection site, and the viscosity of the injectant.
- a long standing basic difficulty with jet injection has been the complex problem of determining which are the preferred injection variables. These variables include: 1) pressure profile, 2) nozzle size, 3) patient factors, i.e., age, sex and size, 4) injection site, and 5) medication viscosity.
- the repeated failures of the prior art to adequately solve these complex variables problems has contributed to the lack of acceptance of a handheld and portable jet injector in the medical community.
- the pressure profile is the pressure exerted on the liquid injectant, typically measured over time, from the beginning to the end of the injection.
- the pressure profile must be selected, in combination with the nozzle size and other factors, to deliver the injectant through the skin to the desired depth, preferably with minimum pain.
- the patient factors are also important. Gender is significant as women typically have a different adipose distribution than men. Men also typically have tougher tissue that women. The patient's age is important because infants are born with very little muscle, thick layers of adipose, and very easily penetrated skin. As infants age and become mobile the adipose is gradually replaced by muscle. At adolescence the introduction of hormones changes tissue composition. Aging through mid-life is usually associated with gradual weight gain and decrease in tissue strength.
- Injection sites are very significant because in all patients the thickness of the skin and adipose tissue varies at different regions of the body.
- the medical profession has established generally accepted injection sites for conventional needle syringes that are best suited for specific types of injection.
- the subcutaneous sites typically have a thick adipose layer and are free of major nerves and vasculature.
- Intramuscular sites typically have a thin adipose layer, a thick muscle layer, and are free of major nerves and vasculature.
- the viscosity of the injectant must be considered as it effects characteristics of the jet injection.
- viscosity effects have been widely misunderstood in the prior art.
- jet injection despite its great potential advantages, remains virtually unused. Accordingly, it is an object of the invention to provide an improved method for needleless injection, so That the advantages of jet injection may be brought into use.
- the pressure profiles of the injectant, nozzle diameter, patient and injection site parameters, as well as injectant viscosity, are selected to achieve desired injection characteristics.
- the injectant is purposely deposited on the deep fascia in a thin sheet. This provides rapid absorption into the blood stream, without the invasiveness, injection discomfort, and occasional post injection soreness associated with injection deep into the muscle.
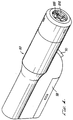
- an injector or needleless injection device 20 which performs the method of the present invention has a front end 22 , a back end 24 , a top surface 26 and a bottom surface 28 .
- a trigger 30 is slidably mounted on the injector 20 adjacent the bottom surface 28 .
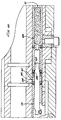
- the injector 20 includes an upper housing 42 and a shorter lower housing 44 attached to the upper housing 42 .
- the lower housing 44 has a flat upper surface 82 which lies against a flat lower surface 84 of the upper housing 42 .
- the upper housing 42 and lower housing 44 are attached together with four (4) pins 86 .
- the upper housing 42 and lower housing 44 together are sized and shaped to readily fit the user's hand, with the user's palm resting over the top surface 26 and side of the injector 20 , and with the user's index finger easily positionable over the trigger 30 .
- the top surface 26 has a step or incline 34 at approximately the center of the injector 20 .
- the upper and lower housings may alternatively be formed as a single housing.
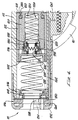
- the lower housing 44 is substantially hollow and defines a lower housing space 48 .
- the upper housing 42 defines an upper housing space 46 (Fig. 6).
- a cartridge chamber 50 for receiving and holding a compressed gas cartridge 54 , e.g., a CO 2 cartridge.
- a cartridge seat 52 at the forward end of the cartridge chamber 50 supports the back of the cartridge 54 .
- a generally u-shaped plastic cartridge chamber cover 56 snaps into place on the lower housing 44 over the cartridge chamber 50 .
- a generally cylindrical piercing housing 58 is slidably positioned behind the cartridge chamber 50 within the lower housing 44 .
- O-rings 60 seal the piercing housing 58 against the lower housing 44 while allowing the piercing housing 58 to slide within the lower housing 44 .
- An annulus 62 extends around the circumference of the piercing housing 58 in between the o-rings 60 .
- a cylindrical piercing body 66 is positioned within the piercing housing 58 and sealed against the piercing housing 58 by o-rings 88 .
- a piercing point 68 extends forward from the front surface of the piercing body 66 and is centrally aligned with the neck of the cartridge 54 .
- a seal 64 on the front end of the piercing body 66 surrounds the piercing point 68 . The seal 64 extends sufficiently forward to seal against the neck of the cartridge 54 before the piercing point 68 penetrates into the cartridge 54 .
- a bore 70 extends through the piercing point 68 and piercing body 66 connecting to the annulus 62 .
- a piercing body nut 74 threads into the back end of the piercing housing 58 , to secure the piercing body 66 and seal 64 in position within and against the forward end of the piercing housing 58 .
- a piercing housing nut 76 threads into the back of the lower housing 44 . Spanner tool openings are provided in the piercing body nut 74 and the piercing housing nut 76 for assembly purposes.
- a threaded shaft 72 extends through and engages threads in the piercing housing nut 76 .
- a knob 78 attached to the threaded shaft 72 has a flip handle 80 which can be flipped up perpendicular to the plane of the knob 78 to allow the knob 78 and threaded shaft 72 to be more easily tuned by hand.
- the forward end of the Threaded shaft 72 bears against the back surface of the piercing body 66 .
- a hole 92 extends through the upper surface 82 of the lower housing to connect the annulus 62 to a bore 96 leading into the upper housing space 46 .
- An o-ring 94 seals the connection of the hole 92 and bore 96 .
- a transparent window lens 98 secured to an end nut 108 by a rubber window retainer 100 .
- a Bourdon tube 116 is soldered into a gauge base 114 and has an open end 124 extending into a gauge chamber 122 .
- the pointer 102 extends perpendicularly from the back end of the Bourdon tube 116 .
- a gauge label 104 applied to the back end of a gauge body 106 around the Bourdon tube 116 provides a calibrated pressure scale with the scale and pointer visible through the lens 98 . Stop pins extending from the back end of the gauge body 106 provide high and low pressure end point stops for the pointer 102 .
- the end nut 108 has threads 110 at its forward end which engage the upper housing 42 .
- the gauge body 106 is rotated relative to the gauge base 114 .
- the gauge body 106 and gauge base 114 are adhered together.
- a guiding pin 112 extends from the upper housing 42 into a keyway groove and holds the gauge body 106 in place while the end nut 108 is tightened.
- Shims 118 are provided at the front surface at the gauge base 114 , as required, for proper stack up and positioning of components in the upper housing 42 .
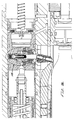
- An initiator valve housing 142 is spaced apart from the gauge base 114 by a filter retainer ring 120 .
- a sandwiched assembly of filter disks 130 and synthetic filters 132 are contained within the back end of the housing 142 .
- O-rings 140 seal the filter disks 130 against the retainer 140 and synthetic filter 132 .
- O-ring 126 seals the filter retainer 140 within the upper housing 42.
- O-ring 126 and o-ring 150 seal the gauge chamber 122 such that compressed gas provided Through the bore 96 can flow out of the gauge chamber 122 only through the filters.
- a port 148 extends through the back wall of the initiator valve housing 142 into an initiator valve chamber 146 within the housing 142 .
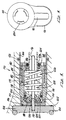
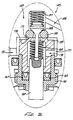
- An initiator valve 144 within the initiator valve chamber 146 controls gas flow from the port 148 through the initiator valve chamber 146 to a reservoir port 154 formed through the forward wall of the initiator valve housing 142 .
- a regulation valve 156 includes a regulation seat 158 formed around the reservoir port 154 .
- a dart 160 moves into and out of the regulation seat 158 .
- the dart 160 has a threaded dart shaft 162 Threaded into the narrower tube section at the back end of a poppet body 172 .
- a dart pin 164 extending through the tube section of the poppet body 172 and the threaded dart shaft 162 secures the adjustment of the longitudinal position of the dart 160 in relation to the regulation seat 158 .
- a reservoir spacer 166 within the upper housing 42 extends from the forward end of the initiator valve housing 142 to a poppet housing 178 , forming a reservoir 168 around the tube section of the poppet body 172 .
- O-rings 126 seal the reservoir spacer 166 against the upper housing 42 and seal the initiator valve housing 142 to the reservoir spacer 166 .
- a poppet valve 170 within the poppet housing 178 has a conical plastic poppet seat 188 centered within and positioned against a forward wall of the poppet housing 178 .
- the poppet body 172 has a sharp sealing edge 200 biased against the poppet seat 188 by a compression spring 186 held in position within the poppet housing 178 by a poppet nut 180 .
- the sealing edge 200 and poppet seat 188 may be configured with unlike angles selected so that the inner diameter contacts first, to minimize creep effects.
- the poppet nut 180 has a threaded forward section 184 engaged to a threaded rear section 182 of the poppet housing 178 .
- the poppet nut 180 is tuned to adjust the compression on the spring 186 and correspondingly set the cracking pressure of the poppet valve 170 .
- the diameter of the poppet seat 188 exposed to reservoir pressure prior to crack remains constant although the conical seat may creep, as the sealing surface, facing reservoir pressure, is parallel to the axis of poppet movement.
- the conical seat is attached to the poppet housing 178 rather than the poppet body 172 while all hard (poppet) parts are made concentric and perpendicular.
- all hard (poppet) parts are made concentric and perpendicular.
- irregularities in the seat 188 or soft part will creep to conform to hard parts.
- the hard parts are free to rotate but will still conform to the existing soft part deformation.
- the seal 206 used with the back up ring 204 may be a low friction seal.
- this seal is pressurized only after cracking due to the poppet body being pressurized internally before cracking, seal friction is greatly minimized.
- the poppet body begins to move during opening before this seal is pressurized. Thus, breakway friction is not increased by gas pressure.
- the poppet and regulation valves can act as a low pressure regulator.
- a cannula 176 is attached to and extends back from a drive piston 212 in front of the poppet valve 170 through the poppet housing 178 and poppet seat 188 and into the back section of the poppet body 172 .
- Poppet body supply holes 174 extend through the poppet body 172 (Fig. 3).
- a cannula exhaust hole is provided through the cannula 176 at a position just slightly behind the o-ring 207 which slidably seals the cannula 176 .
- radially spaced apart drive bores 194 extend through the poppet housing 178 and connect a poppet annulus 198 to the front surface of the poppet housing 178 .
- the poppet annulus 198 a ring-shaped space, is formed by the inside walls of the poppet housing 178 , the front surface of the poppet 172 and the conical surface of the poppet seat 188 .
- the front ends of the drive bores 194 are sealed by a preferably rubber disk drive bore seal 196 adhered to the back surface of the drive piston 212 .
- a joggle 192 in the poppet housing 178 which engages a corresponding lip within the upper housing 42 , acts as a stop for the poppet housing 178 .
- the reservoir spacer 166 , initiator valve housing 142 , filter ring, shims and the gauge body 106 are then subsequently installed within the upper housing 42 and stack up against the poppet housing 178 , with the end nut 108 clamping these components in place.
- o-rings 206 slidably seal the poppet body 172 against the poppet housing 178 and poppet nut 180 .
- the o-rings 206 and back up rings 204 prevent metal to metal contact during movement of the poppet body 172 and also act as pivots and guides to allow slight eccentricity between the poppet body 172 and poppet nut 180 .
- a ring-shaped plenum 202 is formed between the poppet housing 178 and the drive piston 212 , or the o-ring 214 which slidably seals the drive piston 212 Within the upper housing 42 .
- the plenum 202 is just wide enough to insure compression on the face seal 195 .
- a backup ring 218 is provided adjacent to the drive piston seal 214 which is preferably a low friction U-seal.
- a clap piston 210 is slidably positioned within the drive piston 212 and slidably seals against the drive piston 212 with a clamp piston o-ring 222 .
- the back surface of the clamp piston 210 and the front vertical wall of the drive piston 212 form a clamp piston plenum 216 (Fig. 3).
- An o-ring joggle 220 adjacent the back end of the drive piston 212 acts as a stop for the clamp piston o-ring 222 .
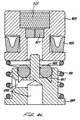
- a clap piston spring 224 within the clamp piston 210 biases forward a jaw plate 228 butting against two opposing flange walls 229 (shown in phantom in Fig. 4) extending from a jaw retainer nut 242 , allowing just enough clearance for the jaws to move freely.
- the force of the clamp piston spring 224 is accordingly transferred from the plate 228 to the flange walls 229 to the jaw retainer nut 242 and bypasses the clamp jaws 236 .
- the clamp jaws 236 are biased outwardly or apart and away from each other by a pair of spaced apart jaw springs 238 .
- the clamp jaws 236 have fine teeth 240 .
- Each clamp jaw 236 has a planar ramp surface 234 flatly engaged by a corresponding planar ramp drive surface 232 on the forward end of the clamp piston 210 .
- the jaw retainer nut 242 is threaded into the front end of the drive piston 212 .
- a return spring 244 is compressed in between the jaw retainer nut 242 and a pressure plate 248 .
- An indicator ring 250 is rotatably positioned in between the front end of the upper housing 42 and a front collar 252 threaded onto the front end of the upper housing 42 .
- the indicator ring 250 has colored sections on its outside edge visible through view ports 256 in the front collar 252 , when the indicator ring 250 is turned to a ready to actuate position signifying that the ampule lugs are fully engaged with the injector lugs,
- a detent pin 288 biased against the back surface of the indicator ring 250 holds the indicator ring in either the ampule loading/unloading position or the ready position, and provides a positive tactile (and optionally an audible click) indication that the ampule is correctly and fully installed, Referring to Fig. 13a, the detent pin 288 slides in or slides against a track 324 cut into the back of the indicator ring.
- the return spring 244 biases the pressure plate 248 forward, to clamp an ampule behind the lugs 254 on the front collar 252 , and it also acts to return the drive piston after an injection.
- the indicator ring 250 has three equally spaced apart tuning lugs 258 extending inwardly, for engaging the lugs 382 at the back of an ampule 360 (Fig. 10).
- the front collar 252 has three equally spaced apart retaining lugs 254 extending radially inwardly, for engaging the front surfaces of the ampule lugs 382 , to hold the ampule into the injector 20 .
- an actuator link 262 has a forward hook 264 in front of the indicator ring 250 .
- a rear hook 260 on the actuator link 262 is attached to an actuator slide block 266 slidably mounted in between the upper housing 42 and lower housing 44 .
- a slide block spring 268 pushing off of the lower housing 44 forwardly biases the actuator slide block 266 .
- the forward surface of the actuator slide block 266 forms the trigger 30 .
- an exhaust valve fork 270 extends laterally and upwardly from the actuator slide block 266 to engage a collar on a spool valve 286 .
- the slide block 266 has a rounded back end 272 facing an initiator valve cam 274 pivotally attached to a holder with a roll pivot pin 278 . Together they are held in a cavity in the upper housing by the upper surface of the lower housing.
- a gap 280 separates the rounded slide block end 272 and the initiator valve cam 274 (Fig. 3).
- a set screw 276 threaded into the initiator valve cam 274 engages an initiator pin in the initiator valve 144 .
- an orifice 282 in the upper housing 42 connects to a drive plenum exhaust bore 284 to continuously vent or bleed the drive plenum 202 to ambient pressure.
- the orifice has an approximately 0.10 mm diameter opening.
- the spool valve 286 attached to the exhaust valve fork 270 is slidably positioned within a spool housing 294 secured within an exhaust passage 296 in the upper housing 42 .
- the spool valve 286 fits within a spool bore 302 in the spool housing 294 with a very close tolerance. While the spool valve 286 does not absolutely seal against the spool bore 302 , leakage between them is very low.
- a reservoir exhaust bore 290 links the reservoir 168 to a spool valve plenum 300 around the spool valve 286 .
- a spool valve hole 301 leads from the spool valve plenum 300 to an exhaust duct 304 behind the spool valve 286 .
- O-rings 292 are positioned on either side of the spool valve plenum 300 to seal the stationary spool valve housing 294 around the reservoir exhaust bore 290 .
- Muffler seals 306 seal the forward end of the spool valve housing 294 against a muffler tube 308 filled with fiberglass wool 310 or other acoustic material and leading to an exhaust port 316 open to ambient pressure.
- a muffler retainer 312 and set screw 314 secure the spool valve housing 294 , muffler seals 306 and muffler tube 308 within the exhaust passage 296 .
- the initiator valve 144 has an initiator valve pin 330 extending from a pin socket 332 .
- a socket spring 334 overlying the pin socket 332 biases the initiator valve pin 330 outwardly or downwardly into engagement with the set screw 276 in the initiator valve cam 274 .
- a valve stem 336 spaced slightly apart from the pin socket 332 has a stem collar 342 with a rubber seat ring 340 sealably engaging a seat neck 350 , within an upper chamber 344 of the initiator valve 144 .
- a stem collar spring 346 positioned in between a valve nut 348 and the stem collar 342 biases the seat ring 340 into engagement with the seat nut 350 to maintain the valve 144 in a closed position.
- the seat nut 350 is supported by, or part of a valve seat 352 sealed within the initiator valve chamber 146 by an o-ring 338 .
- the housing is a single piece housing 303 , rather than the two-piece housing shown in Fig. 2.
- FIG. 6b An alternative preferred design to the exhaust valve shown in Fig. 6 is illustrated in Fig. 6b wherein a valve stem 291 slides inside of a front seal 293 and a rear seal 295 .
- a seal spacer 297 separates the front seal 293 and the rear seal 295 .
- the rear end of the valve stem 291 has two narrow slots 305 which provide a channel for flow of gas when the valve is opened, while giving support to the pressurized rear seal 295 to prevent it from collapsing inwardly,
- the slots 305 form a gradual angle with the rear seal 295 to prevent it from catching on an abrupt edge which could damage the seal.
- valve stem 291 When actuated, the valve stem 291 is pushed forward and the front edge of the valve slots 305 moves forward to the forward edge of the rear seal 295 .
- This allows pressurized exhaust gas to flow from an inlet port 307 , through the seal spacer 297 , out of the valve slots 305 , through a muffler 309 and into an outlet port 311 .
- the front and rear seals 293 and 295 are both u-cup type seals to provide for low friction.
- the exhaust valve is virtually gas tight and requires very little force for actuation. The only significant force that is translated to the valve stem is after opening, the stem is forced to open further which assists in returning the actuator of the injector.
- Fig. 6c shows a piston plenum shut-off valve 321 used in the housing 303 , as an alternative to the continuously venting orifice 282 and drive plenum exhaust bore 284 shown in Fig. 6.
- Shut-off valve 321 includes a piston 323 which has a filter 325 , an orifice 327 and a seal 329 .
- the piston 323 is biased upwardly and into an open position via a spring 331 .
- the force keeping the piston 323 down against the seal is provided by the pressure acting on the area of the annulus created by the piston seal 329 and the shut-off seal 333 .
- the shut-off seal 333 is supported by a valve base 335 which has a vent 337 beneath the shut-off seal 333 to prevent seal escape. Passageways 339 are provided for venting gas.
- the pressure acting on the valve is reduced, the piston 323 moves away from the shut-off seal 333 due to force provided by a spring 331 , and gas flows freely through the filter 325 , the orifice 327 , and through the passages 339 in the valve base 335 .
- Figs. 7a and 7b show an alternate preferred embodiment initiator valve 145 (illustrated in the closed position).
- the initiator valve 145 includes an initiator valve body 147 having an inlet 149 and an outlet 151 .
- a valve poppet 153 is biased against a valve seat 155 by a spring 157 .
- the valve seat 155 is preferably ethylene-propylene which resists absorption by carbon dioxide.
- a valve seat retainer 159 supports the valve seat 155 .
- a valve stem 169 passes through a valve stem guide 161 and a valve stem seal 163 .
- a valve stem spring 165 biases the valve stem into a closed position.
- a valve stem seal 167 slidably seals the valve stem against the valve stem guide 161 .
- an ampule 360 has three spaced apart lugs 382 at its back end.
- a flare 380 leads into an ampule chamber 384 to guide a contoured end 364 of a plunger 362 to engage the ampule 360 .
- the plunger shaft 372 has a cruciform cross section to provide a high moment of inertia using minimum material for the disposable plunger and ampule.
- a collar 374 on the plunger 362 is spaced apart from the tip of the contoured end 364 so that the collar 374 contacts the back surface 388 of the ampule 360 just before the contoured end 364 of the plunger 362 reaches the front end of the ampule 360 . This prevents the contoured end 364 from colliding with the front end of the ampule 360 and overstressing the ampule or buckling the plunger shaft 372 .
- Webs 376 extending from the plunger shaft 372 support the collar 374 .
- the back section 390 of the plunger shaft 372 may have teeth or ridges 378 matching the teeth or ridges 240 on the inside surfaces of the clap jaws 236 , a smooth back section 390 is preferred to avoid variations.
- the cartridge 54 is loaded into the injector 20 by removing or unsnapping the plastic cartridge chamber cover 56 , placing the cartridge 54 into the cartridge chamber 50 , with the neck of the cartridge 54 facing the piercing point 68 , and then replacing the cartridge chamber cover 56 .
- the cartridge chamber cover 56 snaps into position on the lower housing 44 .
- a wavy brass liner 32 say be provided in the cartridge chamber 50 to increase thermal conductivity between the cartridge 54 and the injector 20 .
- the flip handle 80 on the knob 78 is flipped outwardly so that the knob 78 can be more easily tuned.
- the knob 78 is turned by hand causing the threaded shaft 72 to advance forwardly and drive the piercing body 66 and housing 58 towards the cartridge 54 .
- the seal 64 engages and seals against a perimeter on the flat end surface of the cartridge 54 .
- the piercing point 68 engages and pierces the cartridge seal. Compressed gas from the cartridge 54 flows through the bore 70 , into the annulus 62 , through the hole 92 and moves through the bore 96 into the gauge chamber 122 .
- the seal 64 prevents leakage of compressed gas into the cartridge chamber 50 which remains at ambient pressure.
- the cartridge seat 52 supports the cartridge 54 longitudinally against the force exerted by the seal 64 and piercing pin 68 .
- O-rings 60 , 88 and 94 prevent leakage from the passageways from the cartridge 54 to the gauge chamber 122 .
- the knob 78 moves forward towards the piercing housing nut 76 .
- the piercing body 66 and housing are in a fully forward position and the back surface of the knob 78 is approximately flush with the back surface of the upper housing 42 .
- Compressed gas fills the gauge chamber 122 , passes through the filters 130 and 132 , flows through the port 148 (Fig. 3) and into the upper chamber 344 of the initiator valve 144 (Fig. 7).
- the stem collar spring 346 biases the seat ring 340 on the stem collar 342 against the seat neck 350 , thereby sealing the upper chamber 344 and preventing the compressed gas from moving forward.
- the cartridge 54 contains a saturated propellant gas, such as CO 2 , in both liquid and gas states, at temperatures near room temperature.
- a saturated propellant gas such as CO 2
- the filters 130 and 132 substantially prevent any liquid from the cartridge 54 from passing. This allows the device to be used in any orientation without affecting injection characteristics. Without the filters, liquid CO 2 could pass into the initiator valve 144 and reservoir 168 and flash into gas during actuation of the injector 20 , causing unpredictable injection characteristics.
- the Bourdon tube 116 which opens into the gauge chamber 122 is also pressurized, The pressure within the Bourdon tube 116 causes it to spiral outwardly resulting in movement of the pointer 102 to indicate the gas pressure on the gauge label 104 (after the gauge body 106 and gauge base 114 have been properly calibrated), The user can then check the available gas pressure within the injector 20 by looking at the pointer 102 through the lens 98 , as shown in Fig. 8.
- the ampule 360 , plunger 362 and a filling needle may be provided in a sterile package.
- the filling needle has a fitting to engage the Luer fitting 392 on the ampule.
- the ampule may be filled in the same way as a conventional needle and syringe, The filling needle is inserted into a vial of injectant and the injectant is drawn up into the ampule by pulling back on the plunger. Dosage is read by the alignment of the red o-ring 366 with volume graduations on the transparent ampule. The filling needle is removed and safely discarded.
- the ampule is then ready to be placed into the injector, Variable dosage injections are accordingly achieved by loading the ampule in the same manner as for a needle and syringe,
- the present injector 20 can inject various dosages without adjusting the injector.
- the ampule 360 may be filled to e.g., 1/3, 1/2, 3/4, etc. of its full volume capacity, Referring to Fig. 10, loading the ampule 360 with differing volumes of injectant will cause the plunger 362 to extend from the ampule 360 by varying amounts.
- the injector 20 can successfully drive the plunger 362 from any plunger starting position, a single size ampule 360 can be used for various dosage injections. Ampules of varying volumes are not required.
- the plunger and ampule are installed into the injector 20 .
- the lugs 382 on the ampule 360 are aligned to pass through the lugs 254 on the front collar 252 .
- the back end of the plunger 362 is passed Through the front collar 252 , through the return spring 44 and through the clamp piston spring 224 . Since the teeth or ridges 378 on the plunger 362 extend continuously in between the webs 376 and the back end of the plunger, regardless of the dosage carried by the ampule 360 , the teeth 240 of the clamp jaws 236 will over lie the plunger 362 .
- the back surface 388 of the ampule 360 comes to rest against the pressure plate 248 .
- the lugs 382 on the ampule 360 fit in between the lugs 258 on the indicator ring 250 .
- the user then turns the ampule (clockwise as viewed from the front) through an acute angle e.g., approximately 45°, from an ampule loading position to an ampule ready position.
- the ampule causes the indicator ring 250 to turn with it as the sides of the ampule lugs 382 push against the sides of the indicator ring lugs 258 .
- a step on each ampule lug prevents the indicator ring and ampule from being turned beyond range.
- the track on which the detent pin 288 acts is deep enough that the detent cannot be forced out of the track.
- the two ends of the track act as detent stops.
- the indicator ring 250 turns and locks into an injection ready position (Fig. 2a)
- the colored or painted sections on the outside perimeter of the indicator ring 250 moves into view through the view ports 256 . This indicates to the user that the ampule is properly installed in the injector 20 and ready for injection.
- a cut out 320 in the indicator ring moves into alignment with the hook 264 on the actuator link 262 .
- the trigger 30 can then be pulled back to actuate the injector 20 to provide an injection to a patient.
- the actuator link 262 prevents the trigger 30 from moving to actuate the device. Therefore, the injector 20 cannot be actuated unless an ampule is properly installed and aligned in the ready position.
- the nozzle 386 of the ampule 360 is placed against the patient's skin and the trigger 30 on the actuator slide block 266 is pulled back by the user's index finger.
- the exhaust valve fork 270 slides the spool valve 286 from an open position (which allows the reservoir 168 to bleed or exhaust through the exhaust bore to ambient) to a closed position wherein the spool valve 286 substantially seals off the reservoir exhaust bore 290 .
- the reservoir 168 is accordingly sealed off before the slide block end 272 engages the initiator valve cam 274 .
- the spool valve serves as an exhaust control valve.
- the slide block end 272 pushes against the initiator valve cam 274 levering the set screw 276 against the initiator valve pin 330 .
- the sliding movement of the trigger performs three functions: It controls the initiator valve, it controls the spool valve, and it provides an interlock when disabled by the actuator link 262 .
- the set screw 276 pushes up on the initiator valve pin 330 .
- the pin socket 332 is driven up against the valve stem 336 causing the stem collar to shift upwardly and separate the seat ring 340 from the seat neck 350 , thereby opening the initiator valve 144 .
- the valve poppet spring 157 biases the valve poppet 153 toward the valve seat 155 . Gas pressure from the gas inlet 149 drives the poppet 153 into the valve seat 155 creating a gas tight meal.
- the valve seat 155 is vented on the bottom side 171 to prevent the seat from escaping from the groove 173 .
- the valve seat retainer 159 retains and vents the valve seal 155 .
- the valve stem 169 is mechanically isolated from the poppet 153 to assure that the poppet closes without interference from the stem.
- valve stem 169 slides up and contacts the valve poppet 153 , pushing it away from the valve seat 155 .
- valve stem spring 165 returns the valve stem to the neutral position and the valve poppet 153 also returns to the closed position.
- compressed gas flows from the cartridge 54 through the filters and initiator valve 144 , through the reservoir port 154 past the dart 160 and into the reservoir 168 .
- gas pressure also builds within the poppet chamber 208 , as gas flows from the reservoir 168 through the poppet body supply holes 174 .
- the clamp piston 210 is driven forward compressing the clamp piston spring 224 and driving the clamp jaws 236 together, through the interaction of the ramp drive 232 on the clamp piston 210 and the clamp piston ramps 234 on the clamp jaws 236 .
- the teeth 240 on the clamp jaws 236 clamp down and around the plunger 362 .
- the clamp jaws 236 and their driving mechanism perform two functions: They grab onto the plunger at whatever position the plunger is in, and they transfer driving force from the drive piston to the plunger.
- the plunger 362 will be fully extended to the rear such that the clamp jaws 236 will engage the plunger 362 close behind the webs 376 .
- the plunger 362 will extend a shorter distance behind the ampule 360 and the clamp jaws 236 will engage the plunger 362 towards the back end of the plunger.
- the clamp jaws 236 securely clamp and engage the plunger 362 with the teeth 240 on the clamp jaws 236 locked into the teeth 378 on the plunger 362 .
- the gas pressure in the clamp piston plenum 216 maintains the engagement of the clamp jaws 236 to the plunger 362 during the injection sequence. As represented in Fig. 12, the clap jaws clap onto the plunger before the poppet valve opens.
- pressure in the poppet chamber 208 continues to build until it is sufficient to crack the poppet valve 170 open.
- the poppet spring chamber 226 is sealed from the reservoir 168 and the poppet chamber 208 and is vented to ambient pressure.
- the rearward acting force resulting from the gas pressure acting on the incline surfaces 152 of the poppet body 172 will exceed the forward acting force of the poppet spring 186 .
- this "cracking point" is reached, the poppet valve 170 snaps open.
- the poppet body 172 shifts or slides rearwardly,
- the sealing edge surface 200 separates from its sealing engagement against the conical poppet seat 188 allowing gas from the reservoir 168 to flow through the poppet chamber 208 to the drive bores 194 .
- the poppet valve 170 begins to open and the poppet body 172 moves away from the conical poppet seal 188 , the annular front surface 230 of the poppet body 172 is acted on by gas pressure now in the poppet annulus 198 . Since the surface areas acted on by the compressed gas are vastly increased with the addition of the front surface 230 of the poppet body, the force acting on the poppet body 172 rapidly escalates.
- the poppet valve 170 therefore opens with an "over-center" or hard-over action.
- the regulation valve 156 closes down via the dart 160 engaging and sealing against the regulation seat 158 .
- additional gas supply to the reservoir 168 is, at least initially, restricted by the regulation valve 156 , with substantially only the reservoir 168 then acting as a source of compressed gas.
- pressure regulation of the reservoir is provided through poppet area ratios and spring forces (which may be readily determined for various capacity injectors by those skilled in the art).
- the reservoir pressure reaches a desired minimum pressure.
- the drive piston plenum has been supplied by a fixed supply of gas from the reservoir.
- the spring force acting forwardly on the poppet body, overcomes the net pressure force, acting rearwardly on the poppet body.
- the poppet body moves forward, lessening the regulation valve restriction to incoming flow.
- the dart 160 moves with the poppet body away from the seat 158 to allow commencement or increase of gas flow.
- the opening of the regulator valve consequently increases gas flow into the reservoir and increases the reservoir pressure.
- the poppet body again moves rearwardly to restrict the incoming flow,
- the poppet valve and regulator valve act together as a reservoir pressure regulator (and consequently drive piston plenum pressure and ampule pressure).
- regulation movement when present, occurs generally during the last half of the injection.
- the CO 2 cartridge is filled with saturated CO 2 .
- the peak ampule pressure is determined by the poppet valve cracking pressure which is independent of source pressure.
- the minimum delivery pressure, governed by the pressure regulation is also independent of source pressure. Both of these features are controlled by area ratios and spring rates.
- the injector is substantially temperature independent.
- Fig. 12 illustrates the effect of pressure regulation.
- a smaller dosage of e.g., 1/2 ml or less generally there is no pressure regulation.
- larger dosages of e.g., over 3/4 ml pressure regulation occurs.
- intermediate range dosages of e.g., between 1/2 and 3/4 ml some pressure regulation may occur.
- the rubber or elastomeric face seal 196 adhered to the back of the drive piston 212 assists to rapidly open the poppet valve 170 .
- the face seal 196 encourages the build up of pressure in the drive bores 194 and poppet annulus 198 before pressurizing the drive plenum 202 . Accordingly, the rapid pressure increase within the drive bores 194 and poppet annulus 198 shorten the time required for opening the poppet valve 170 providing a quick ampule pressure rise time and a more uniform ampule peak pressure.
- the poppet body supply holes 174 have a large diameter to minimize pressure drop from the reservoir 168 to the poppet chamber 208 .
- the trigger 30 is held back for longer than necessary for the injection, only a small amount of gas is wasted since all spaces within the injector, except the drive plenum, remain virtually sealed while the trigger is held back.
- the drive plenum is opened to ambient pressure, but only through orifice 282 which severely restricts flow.
- the regulation valve 156 restricts flow while the trigger is held back.
- the trigger is released.
- the slide block spring 268 assisted by exhaust gas pressure returns the slide block 266 to its forward position.
- the initiator valve then closes, Then the exhaust valve fork 270 moving with the slide block 266 pulls the spool valve 286 forward reconnecting the spool valve bore 302 and spool plenum 300 to the reservoir exhaust bore 290 .
- the spool valve and exhaust passage allow the injector to be quickly and quietly reset for another injection, Gas in the reservoir exhausts out through the reservoir exhaust bore 290 and exhaust passage 296 .
- the exhaust gas pressure in the exhaust passage 296 pushes on the back of the spool valve 286 and helps to return the spool valve and slide block forward to their original ready positions.
- the slide block spring 268 consequently need only exert a slight force, thereby helping to reduce the finger force necessary to pull the trigger 30 .
- the drive piston 212 is in the forward position (Fig. 2c), with the plunger shoulder in contact with and exerting a large force on the back end 388 of the ampule 360 .
- the drive piston return spring 244 , clamp piston spring 224 and jaw springs 238 are compressed.
- the jaws 236 are engaged with the plunger and the clamp piston 210 is forward. Each part must then return to the ready position.
- the reservoir 168 Upon release of the trigger 30 , the reservoir 168 is able to rapidly vent to atmosphere.
- Drive piston plenum gas vents into the reservoir, in part, through the poppet body, until the poppet valve closes, Gas also vents into the reservoir through the cannula 176 , until the holes in the cannula are sealed by the o-ring 190 contained within the poppet seat 188 .
- This remaining gas which occupies a relatively small volume, and is at a very low pressure, vents through the bleed orifice 282 connecting the drive piston plenum directly to the atmosphere through the drive plenum exhaust bore 284 . Since the orifice 282 is always open, even during the injection, some beneficial drive gas is lost, thus it is a very small, restrictive orifice. Because the orifice 282 is small, if it was the only vent for drive piston plenum gas (i.e., if there were no cannula side holes), venting and reset time would be unacceptably long.
- the following reset sequence occurs and is controlled by component areas and spring forces, which may be readily determined by those skilled in the art.
- the clap jaws 236 and clap piston 210 release. This must occur before the drive piston is released so that the plunger is not pulled back.
- the clamp piston spring force overcomes the opposing pressure force. This release occurs when the drive piston 212 is close to a force equilibrium condition. The pressure force must be close to the opposing spring force. If not, then the drive piston 212 will rapidly return (if the spring force is larger) or plunge forward (if pressure force is larger) causing noise and possible damage to the injector, Thus a force balance is established at the point of plunger release, regardless of the dosage.
- the drive piston 212 After the plunger release, the drive piston 212 returns as the reservoir bleeds. The drive piston 212 is forced rearward by the drive piston return spring against the opposing pressure force. Gas exhaust and reset occurs quietly and quickly.
- O-ring 222 serves as a seal and a bumper to quiet the clamp piston return.
- the plunger 362 is driven forward until the collar 374 contacts the back surface 388 of the ampule 360 . Accordingly, if the trigger 30 is squeezed once and an injection given, released and squeezed again after some delay (i.e., "second fire") without replacing the ampule, the jaws will grab the plunger with the plunger collar in the forward most position, i.e., in contact with the rear ampule face. Thus no forward movement of the drive piston will occur. A second fire does not damage the ampule, plunger or injector.
- the cannula 176 is attached to and moves with the drive piston 212 .
- the cannula exhaust hole 190 in the cannula 176 speeds the return stroke of the piston 212 .
- the poppet valve closes before the drive piston begins its return, Thus a bleed hole in the cannula is required for gas to flow from the drive piston plenum to the reservoir.
- gas in the drive plenum 202 flows through the cannula exhaust hole 190 through the cannula 176 , back into the reservoir 168 and out through the relatively unobstructed exhaust system of the reservoir exhaust bore 290 and the exhaust passage 296 .
- the spent ampule and plunger are turned and removed from the injector 20 which is then prepared for the next injection sequence.
- the ampule and plunger are preferably a single use disposable unit.
- the plunger may have tapered sections at the front or back which engage a generally complimentary tapered section in the ampule.
- the injector exerts hundreds of pounds of force on the plunger which drives the tapered section of the plunger of Figs. 10a and 10b into an interference fit with the tapered section of the ampule.
- the used and non sterile plunger and ampule cannot easily then be re-used.
- the tapered sections can also act as a plunger stop, in place of the collar on the plunger of Fig. 10, The taper on the plunger and ampule are slightly mismatched and lock together only with high forces (at the end of an injection) and not at low forces (during filling of the ampule).
- Fig. 10c shows another non-reusable ampule and plunger having a detent. The detent is dimensioned so that only a large force will cause engagement.
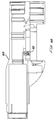
- the injector can be modified to give multiple sequential injections to the same patient.
- a drive piston stop 394 is added, and acts to stop the drive piston, as the plunger shoulder does in variable delivery.
- the jaws then disengage and the injector resets.
- the plunger will automatically be in a "ready” position for the next shot, and the injector may be fired again to deliver the same small dosage.
- This sequence may be repeated to deliver several small dosage injections until the plunger shoulder contacts the ampule. Dosage may be adjusted by rotating the outer ring 396 to the desired value, indicated by graduations 398 on the injector housing. A longer ampule can be provided to allow for more sequential shots.
- the present method of needleless injection uses a system of an injector and compatible ampules.
- the injector is designed to apply a specific force on the plunger of the ampules.
- the force applied to the plunger by the injector is varied, forming a force - displacement curve.
- the force applied to the plunger is quite high.
- the applied force is reduced substantially linearly until the volume injected reaches approximately 0.5 ml, and thereafter the force is held substantially constant.
- This force displacement curve is independent of the ampule nozzle size.
- This force - displacement curve translates directly to an ampule pressure - volume injected curve.
- the injection system employs a singular pressure profile and a family of ampules with various orifice sizes to achieve various depths of penetration.
- Fig. 17 shows preferred uses of various diameter nozzles with the pressure profile described below.
- the preferred pressure profile has the following properties: First the pressure rapidly increases from 0 to a value of about 26900-29670 kPa, and preferably about 28290 kPa, (4100 psi) in less than 6 milliseconds (and preferably less than 1 ms). This quick pressure rise avoids "splash-back" and loss of injectant. This pressure range is sufficient to pierce the tissues, but not so high as to cause the excessive pain associated with higher pressures. The pressure is gradually reduced to approximately 8280 - 13800 kPa, and preferably 12420 kPa, (1800 psi) in a generally linear (pressure - volume) fashion corresponding with volume discharged of 0.5 ml.
- the curve forms an exponential decay.
- the pressure is held constant until the end of the injection, at which time the pressure abruptly goes to 0 within 5 ms.
- Final pressures below about 8280 kPa tend to result in "leak-back" of the injectant after the injection,
- the pressure profile is defined as the pressure immediately proximal to the nozzle.
- the above- described pressure profile covers an injection larger than approximately 0.5 ml. If the injection is less than this amount, the pressure - profile curve is simply truncated at the end of the delivered volume.
- Medication viscosity affects penetration of intramuscular injections in a direction contrary to prior art.
- Experimental data shows that more viscous medications, in the range of from 0.01 to 0.70 poise, have greater fascia penetrating capability, apparently because of reduced turbulence and lower Reynold's number.
- the present invention also includes the appropriate guidelines for selection of nozzle size with viscous medications.
- Viscous medications preferably use the same size orifice as for water based medications. Nearly all viscous medications are intramuscular injections, Testing shows that viscous medications have more energy to penetrate the deep fascia than water based medications, but do not go substantially deeper into the muscle. Therefore, the deposition into the muscle is comparable independent of medication viscosity.
- the present peri-fascial injection is provided by using a nozzle diameter which is smaller than that which would ordinarily be used for an intramuscular injection.
- the peri-fascial injection is provided by using a SC nozzle (0.10 mm) at an IM injection site preferably with less than 5 mm adipose. This works well because IM sites tend to have very thin layers of adipose tissue.
- the SC nozzle has sufficient penetrating energy to deposit the medication on the deep fascia when injected into a thin layer of adipose.
- a peri-fascial injection can also be given at an IM injection site having a 10-15 mm adipose layer using a 0.15 mm diameter nozzle and the above-described pressure profile.
- the injectant 800 in a peri-fascial injection bores through the skin 802 and adipose 804 , but not the fascia 806 . Rather, the injectant forms a thin layer 808 over the fascia, The thin layer 808 may provide the same pharmacological effect as an IM injection, without penetrating the muscle.
Landscapes
- Health & Medical Sciences (AREA)
- Vascular Medicine (AREA)
- Engineering & Computer Science (AREA)
- Anesthesiology (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Hematology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Infusion, Injection, And Reservoir Apparatuses (AREA)
Claims (5)
- Verfahren zum Liefern eines Einspritzstrahls durch unter Druck setzen eines fluiden Injektats innerhalb einer Ampulle (360), welches Verfahren die Schritte aufweist:einen Tauchkolben (362) mit ausreichender Kraft in eine Ampulle (360) treiben, so daß innerhalb von 6 Millisekunden ein Druck von 26000-30000 kPa erzeugt wird;den Druck im allgemeinen linear reduzieren, bis 0,5 ml herausgetrieben werden und der Druck 8000-14000 kPa beträgt;den Druck so lange bei 8000-14000 kPa halten, bis ein gewünschtes Volumen an Injektat aus der Ampulle (360) herausgetrieben worden ist; undden Druck innerhalb von 5.0 Millisekunden auf ungefähr 0.0 kPa reduzieren.
- Verfahren gemäß Anspruch 1, welches weiter den Schritt des Regulierens des Durchmessers der Mündungsöffnung der Ampulle (360) aufweist.
- Verfahren gemäß Anspruch 1 oder 2, welches weiter den Schritt des Regulierens der Viskosität des Injektats aufweist.
- Verfahren gemäß Anspruch 3, wobei die Viskosität des Injektats im Bereich von 0,01 bis 0,70 Poise liegt.
- Verfahren gemäß einem der Ansprüche 1-4, welches weiter den Schritt des Filterns des komprimierten Gases, bevor es in ein Reservoir abgelassen wird, aufweist.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US920106 | 1992-07-24 | ||
| US07/920,106 US5383851A (en) | 1992-07-24 | 1992-07-24 | Needleless hypodermic injection device |
| PCT/US1993/006940 WO1994002188A1 (en) | 1992-07-24 | 1993-07-23 | Needleless hypodermic injection methods and device |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0651663A1 EP0651663A1 (de) | 1995-05-10 |
| EP0651663A4 EP0651663A4 (de) | 1995-08-30 |
| EP0651663B1 true EP0651663B1 (de) | 1999-12-01 |
Family
ID=25443171
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP93918344A Expired - Lifetime EP0651663B1 (de) | 1992-07-24 | 1993-07-23 | Verfahren für nadellose subkutaninjektionen |
Country Status (8)
| Country | Link |
|---|---|
| US (3) | US5383851A (de) |
| EP (1) | EP0651663B1 (de) |
| JP (2) | JP3633615B2 (de) |
| AT (1) | ATE187083T1 (de) |
| AU (1) | AU676490B2 (de) |
| CA (1) | CA2140772C (de) |
| DE (1) | DE69327165T2 (de) |
| WO (1) | WO1994002188A1 (de) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| USRE43824E1 (en) | 2001-01-11 | 2012-11-20 | Powder Pharmaceuticals Inc. | Needleless syringe |
| US8540665B2 (en) | 2007-05-04 | 2013-09-24 | Powder Pharmaceuticals Inc. | Particle cassettes and processes therefor |
Families Citing this family (1182)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2689017B1 (fr) * | 1992-03-31 | 1994-05-27 | Mtc Medical | Appareil d'injection medicale ou veterinaire. |
| US5383851A (en) * | 1992-07-24 | 1995-01-24 | Bioject Inc. | Needleless hypodermic injection device |
| US5405333A (en) * | 1992-12-28 | 1995-04-11 | Richmond; Frank M. | Liquid medicament bag with needleless connector fitting using boat assembly |
| US6206860B1 (en) | 1993-07-28 | 2001-03-27 | Frank M. Richmond | Spikeless connection and drip chamber with valve |
| US5540657A (en) * | 1994-07-15 | 1996-07-30 | Collagen Corporation | Delivery device for injectable materials |
| DE19519279A1 (de) * | 1994-12-07 | 1996-08-22 | Wolfgang Dr Med Wagner | Verfahren und Einrichtung zur Saug-Druckinjektion |
| US5599302A (en) | 1995-01-09 | 1997-02-04 | Medi-Ject Corporation | Medical injection system and method, gas spring thereof and launching device using gas spring |
| DE29507987U1 (de) * | 1995-05-15 | 1996-09-19 | Ferton Holding, Delemont | Ejektionsgerät zur Hochdruckejektion einer Flüssigkeit |
| US5893397A (en) * | 1996-01-12 | 1999-04-13 | Bioject Inc. | Medication vial/syringe liquid-transfer apparatus |
| US5800388A (en) | 1996-02-29 | 1998-09-01 | Medi-Ject Corporation | Plunger/ram assembly adapted for a fluid injector |
| US5697917A (en) | 1996-02-29 | 1997-12-16 | Medi-Ject Corporation | Nozzle assembly with adjustable plunger travel gap |
| US5722953A (en) | 1996-02-29 | 1998-03-03 | Medi-Ject Corporation | Nozzle assembly for injection device |
| US5643211A (en) | 1996-02-29 | 1997-07-01 | Medi-Ject Corporation | Nozzle assembly having a frangible plunger |
| US5921967A (en) | 1996-02-29 | 1999-07-13 | Medi-Ject Corporation | Plunger for nozzle assembly |
| US5865795A (en) | 1996-02-29 | 1999-02-02 | Medi-Ject Corporation | Safety mechanism for injection devices |
| US5785688A (en) * | 1996-05-07 | 1998-07-28 | Ceramatec, Inc. | Fluid delivery apparatus and method |
| KR100376646B1 (ko) | 1996-07-01 | 2003-03-15 | 파마시아 에이비 | 전달 장치 및 이것의 작동 방법 |
| US6458574B1 (en) | 1996-09-12 | 2002-10-01 | Transkaryotic Therapies, Inc. | Treatment of a α-galactosidase a deficiency |
| US6106502A (en) * | 1996-12-18 | 2000-08-22 | Richmond; Frank M. | IV sets with needleless fittings and valves |
| US5875976A (en) | 1996-12-24 | 1999-03-02 | Medi-Ject Corporation | Locking mechanism for nozzle assembly |
| US5993412A (en) * | 1997-05-19 | 1999-11-30 | Bioject, Inc. | Injection apparatus |
| US5911703A (en) * | 1997-05-22 | 1999-06-15 | Avant Drug Delivery Systems, Inc. | Two-stage fluid medicament jet injector |
| US5947928A (en) * | 1997-06-19 | 1999-09-07 | Mile Creek Capital, Llc | Drug delivery system |
| USD399951S (en) | 1997-06-30 | 1998-10-20 | Bioject, Inc. | Injection apparatus |
| US5964735A (en) * | 1997-07-01 | 1999-10-12 | Medisys Technologies, Inc. | Fine needle aspiration safety syringe |
| EP0888790A1 (de) * | 1997-07-04 | 1999-01-07 | PowderJect Research Limited | Vorrichtung zur Abgabe von Medikamententeilchen |
| US6569127B1 (en) * | 1997-07-18 | 2003-05-27 | Liebel-Flarsheim Company | Adapter and syringe for front-loading medical fluid injector |
| US6171276B1 (en) | 1997-08-06 | 2001-01-09 | Pharmacia & Upjohn Ab | Automated delivery device and method for its operation |
| US6071272A (en) * | 1997-10-28 | 2000-06-06 | Hoffman; Alan S. | Method for treating erectile dysfunctionality |
| US6610042B2 (en) | 1997-12-05 | 2003-08-26 | Felton Medical, Inc. | Disposable unit-dose jet-injection syringe for pre-filled and/or transfilled liquid injectable medical drug or vaccine products and method thereof |
| US6541606B2 (en) * | 1997-12-31 | 2003-04-01 | Altus Biologics Inc. | Stabilized protein crystals formulations containing them and methods of making them |
| US5993418A (en) * | 1998-02-24 | 1999-11-30 | Medisys Technologies, Inc. | Safety syringe |
| US20020147143A1 (en) | 1998-03-18 | 2002-10-10 | Corixa Corporation | Compositions and methods for the therapy and diagnosis of lung cancer |
| AU3489499A (en) | 1998-04-14 | 1999-11-01 | Merck & Co., Inc. | Needleless administration of polynucleotide formulations |
| JP4294868B2 (ja) | 1998-07-27 | 2009-07-15 | アンテアリーズ ファーマ インコーポレイテッド | 医療用注射器組立体用の注射補助プローブ |
| WO2000006227A1 (en) * | 1998-07-27 | 2000-02-10 | Medi-Ject Corporation | Loading mechanism for medical injector assembly |
| GB9818110D0 (en) * | 1998-08-19 | 1998-10-14 | Weston Medical Ltd | Needleless injectors and other devices |
| US6532386B2 (en) | 1998-08-31 | 2003-03-11 | Johnson & Johnson Consumer Companies, Inc. | Electrotransort device comprising blades |
| US20030235557A1 (en) | 1998-09-30 | 2003-12-25 | Corixa Corporation | Compositions and methods for WT1 specific immunotherapy |
| US6783509B1 (en) | 1998-11-18 | 2004-08-31 | Bioject Inc. | Single-use needle-less hypodermic jet injection apparatus and method |
| US6132395A (en) * | 1998-12-08 | 2000-10-17 | Bioject, Inc. | Needleless syringe with prefilled cartridge |
| US6383168B1 (en) | 1998-12-08 | 2002-05-07 | Bioject Medical Technologies Inc. | Needleless syringe with prefilled cartridge |
| US6406455B1 (en) * | 1998-12-18 | 2002-06-18 | Biovalve Technologies, Inc. | Injection devices |
| DE19859137C1 (de) * | 1998-12-21 | 2000-05-18 | Ferton Holding Sa | Ejektionsgerät zur Hochdruckejektion einer Flüssigkeit |
| US6328714B1 (en) * | 1999-01-29 | 2001-12-11 | Powderject Research Limited | Particle delivery device |
| US6849060B1 (en) | 1999-01-29 | 2005-02-01 | Powderject Research Limited | Particle delivery device |
| JP4180244B2 (ja) | 1999-04-16 | 2008-11-12 | ジョンソン・アンド・ジョンソン・コンシューマー・カンパニーズ・インコーポレイテッド | 内部センサを有する電気的な移動による送出装置系 |
| US7060048B1 (en) * | 1999-04-16 | 2006-06-13 | Powerject Research Limited | Needleless syringe |
| WO2000064514A1 (en) * | 1999-04-22 | 2000-11-02 | Gilbert Garitano | Needleless permanent makeup and tattoo device |
| US6689103B1 (en) | 1999-05-07 | 2004-02-10 | Scimed Life System, Inc. | Injection array apparatus and method |
| US6319230B1 (en) | 1999-05-07 | 2001-11-20 | Scimed Life Systems, Inc. | Lateral needle injection apparatus and method |
| US7192713B1 (en) | 1999-05-18 | 2007-03-20 | President And Fellows Of Harvard College | Stabilized compounds having secondary structure motifs |
| US7147633B2 (en) * | 1999-06-02 | 2006-12-12 | Boston Scientific Scimed, Inc. | Method and apparatus for treatment of atrial fibrillation |
| WO2000072908A1 (en) * | 1999-06-02 | 2000-12-07 | Microheart, Inc. | Devices and methods for delivering a drug |
| AU6076200A (en) | 1999-07-08 | 2001-01-30 | Johnson & Johnson Consumer Companies, Inc. | Exothermic bandage |
| US6890553B1 (en) | 1999-07-08 | 2005-05-10 | Johnson & Johnson Consumer Companies, Inc. | Exothermic topical delivery device |
| US20050027239A1 (en) * | 1999-08-20 | 2005-02-03 | Stout Richard R. | Intradermal injection system for injecting DNA-based injectables into humans |
| US6319224B1 (en) * | 1999-08-20 | 2001-11-20 | Bioject Medical Technologies Inc. | Intradermal injection system for injecting DNA-based injectables into humans |
| AU7064700A (en) * | 1999-08-20 | 2001-03-19 | Bioject, Inc. | Intramuscular injection system for injecting dna-based injectables into humans |
| EP1212422B1 (de) | 1999-08-24 | 2007-02-21 | Medarex, Inc. | Humane antikörper gegen ctla-4 und deren verwendungen |
| US7113821B1 (en) | 1999-08-25 | 2006-09-26 | Johnson & Johnson Consumer Companies, Inc. | Tissue electroperforation for enhanced drug delivery |
| US7133717B2 (en) * | 1999-08-25 | 2006-11-07 | Johnson & Johnson Consumer Companies, Inc. | Tissue electroperforation for enhanced drug delivery and diagnostic sampling |
| ATE285806T1 (de) | 1999-10-11 | 2005-01-15 | Felton International Inc | Universelle antiinfektionsschutzvorrichtung für nadellose injektoren |
| US7074210B2 (en) * | 1999-10-11 | 2006-07-11 | Felton International, Inc. | Universal protector cap with auto-disable features for needle-free injectors |
| US6776776B2 (en) * | 1999-10-14 | 2004-08-17 | Becton, Dickinson And Company | Prefillable intradermal delivery device |
| US20020193740A1 (en) | 1999-10-14 | 2002-12-19 | Alchas Paul G. | Method of intradermally injecting substances |
| US6494865B1 (en) | 1999-10-14 | 2002-12-17 | Becton Dickinson And Company | Intradermal delivery device including a needle assembly |
| US6569143B2 (en) | 1999-10-14 | 2003-05-27 | Becton, Dickinson And Company | Method of intradermally injecting substances |
| US6843781B2 (en) * | 1999-10-14 | 2005-01-18 | Becton, Dickinson And Company | Intradermal needle |
| US6569123B2 (en) | 1999-10-14 | 2003-05-27 | Becton, Dickinson And Company | Prefillable intradermal injector |
| CA2392598C (en) * | 1999-11-23 | 2010-06-08 | Needleless Ventures, Inc. | Injector assembly with driving means and locking means |
| US7887506B1 (en) | 1999-11-23 | 2011-02-15 | Pulse Needlefree Systems, Inc. | Safety mechanism to prevent accidental patient injection and methods of same |
| US7029457B2 (en) * | 1999-11-23 | 2006-04-18 | Felton International, Inc. | Jet injector with hand piece |
| US6770054B1 (en) | 1999-11-23 | 2004-08-03 | Felton International, Inc. | Injector assembly with driving means and locking means |
| US6344027B1 (en) | 1999-12-08 | 2002-02-05 | Scimed Life Systems, Inc. | Needle-less injection apparatus and method |
| US6613026B1 (en) * | 1999-12-08 | 2003-09-02 | Scimed Life Systems, Inc. | Lateral needle-less injection apparatus and method |
| CA2396569C (en) * | 2000-01-07 | 2010-03-23 | Biovalve Technologies, Inc. | Injection device |
| IL151097A0 (en) | 2000-02-23 | 2003-04-10 | Smithkline Beecham Biolog | Tumour-specific animal proteins |
| FR2805749B1 (fr) * | 2000-03-01 | 2002-05-17 | Poudres & Explosifs Ste Nale | Seringue sans aiguille a deux niveaux de vitesse d'injection |
| US7137968B1 (en) * | 2000-03-13 | 2006-11-21 | Nucryst Pharmaceuticals Corp. | Transcutaneous medical device dressings and method of use |
| WO2001070309A1 (en) * | 2000-03-23 | 2001-09-27 | Antares Pharma, Inc. | Single use disposable jet injector |
| US6719987B2 (en) | 2000-04-17 | 2004-04-13 | Nucryst Pharmaceuticals Corp. | Antimicrobial bioabsorbable materials |
| FR2807946B1 (fr) | 2000-04-19 | 2002-06-07 | Poudres & Explosifs Ste Nale | Seringue sans aiguille fonctionnant avec un chargement pyrotechnique bicomposition |
| US6716190B1 (en) * | 2000-04-19 | 2004-04-06 | Scimed Life Systems, Inc. | Device and methods for the delivery and injection of therapeutic and diagnostic agents to a target site within a body |
| US7404815B2 (en) * | 2000-05-01 | 2008-07-29 | Lifescan, Inc. | Tissue ablation by shear force for sampling biological fluids and delivering active agents |
| US6689101B2 (en) * | 2000-05-22 | 2004-02-10 | Pharmacia Ab | Medical arrangement |
| US6645169B1 (en) * | 2000-06-08 | 2003-11-11 | Avant Drug Delivery Systems, Inc. | Air-in-tip jet injector |
| US6406456B1 (en) | 2000-06-08 | 2002-06-18 | Avant Drug Delivery Systems, Inc. | Jet injector |
| DE10029325A1 (de) | 2000-06-20 | 2002-01-03 | Peter Lell | Nadellose Injektionsvorrichtung mit pyrotechnischem Antrieb |
| AR029540A1 (es) | 2000-06-28 | 2003-07-02 | Corixa Corp | COMPOSICIONES Y METODOS PARA EL DIAGNoSTICO Y LA TERAPIA DE CA NCER DE PULMoN |
| US6989157B2 (en) * | 2000-07-27 | 2006-01-24 | Nucryst Pharmaceuticals Corp. | Dry powders of metal-containing compounds |
| US20030206966A1 (en) * | 2000-07-27 | 2003-11-06 | Burrell Robert E. | Methods of inducing apoptosis and modulating metalloproteinases |
| US20040191329A1 (en) * | 2000-07-27 | 2004-09-30 | Burrell Robert E. | Compositions and methods of metal-containing materials |
| US7001617B2 (en) | 2001-04-23 | 2006-02-21 | Nueryst Pharmaceuticals Corp. | Method of induction of apoptosis and inhibition of matrix metalloproteinases using antimicrobial metals |
| US20030180379A1 (en) * | 2000-07-27 | 2003-09-25 | Burrell Robert E. | Solutions and aerosols of metal-containing compounds |
| US7255881B2 (en) * | 2000-07-27 | 2007-08-14 | Nucryst Pharmaceuticals Corp. | Metal-containing materials |
| AU2001278322B2 (en) | 2000-07-27 | 2005-12-22 | Smith & Nephew (Overseas) Limited | Treatment of hyperproliferative skin disorders and diseases |
| US7008647B2 (en) * | 2001-04-23 | 2006-03-07 | Nucryst Pharmaceuticals Corp. | Treatment of acne |
| US7427416B2 (en) * | 2000-07-27 | 2008-09-23 | Nucryst Pharmaceuticals Corp. | Methods of treating conditions using metal-containing materials |
| US20030185901A1 (en) | 2000-07-27 | 2003-10-02 | Burrell Robert E. | Methods of treating conditions with a metal-containing material |
| GB0022742D0 (en) | 2000-09-15 | 2000-11-01 | Smithkline Beecham Biolog | Vaccine |
| CA2324045A1 (fr) * | 2000-10-20 | 2002-04-20 | Universite De Sherbrooke | Seringue sans aiguille pour l'injection sous-cutanee de poudres medicamenteuses |
| FR2815544B1 (fr) | 2000-10-23 | 2003-02-14 | Poudres & Explosifs Ste Nale | Seringue sans aiguille securisee a architecture compacte |
| US6652483B2 (en) * | 2000-11-07 | 2003-11-25 | Avant Drug Delivery Systems, Inc. | Needleless jet injector system with separate drug reservoir |
| US6626863B1 (en) | 2000-11-22 | 2003-09-30 | Nusaf, L.L.C. | Safety syringe |
| US7931614B2 (en) * | 2000-11-30 | 2011-04-26 | Valeritas, Inc. | Injection systems |
| EP1351730B1 (de) * | 2001-01-18 | 2006-06-28 | Medrad, Inc. | Spritzenanschlussflächen und adaptern für verwendung mit medizinische injektoren |
| US6758837B2 (en) | 2001-02-08 | 2004-07-06 | Pharmacia Ab | Liquid delivery device and method of use thereof |
| EP2281573A3 (de) | 2001-02-23 | 2011-12-07 | GlaxoSmithKline Biologicals s.a. | Influenza Vakzinzusammensetzungen zur intradermaler Verabreichung |
| US6471669B2 (en) * | 2001-03-05 | 2002-10-29 | Bioject Medical Technologies Inc. | Disposable needle-free injection apparatus and method |
| US6645170B2 (en) * | 2001-03-05 | 2003-11-11 | Bioject Medical Technologies, Inc. | Simplified disposable needle-free injection apparatus and method |
| US20020172615A1 (en) * | 2001-03-08 | 2002-11-21 | Archie Woodworth | Apparatus for and method of manufacturing a prefilled sterile container |
| US20020139088A1 (en) | 2001-03-08 | 2002-10-03 | Archie Woodworth | Polymeric syringe body and stopper |
| CA2440983A1 (en) | 2001-03-14 | 2002-09-26 | Penjet Corporation | System and method for removing dissolved gas from a solution |
| ZA200200808B (en) | 2001-03-22 | 2002-08-12 | Roche Diagnostics Gmbh | Needleless hypodermic injection system, application device and medication cartridge therefor. |
| US7795218B2 (en) | 2001-04-12 | 2010-09-14 | Bioaxone Therapeutique Inc. | ADP-ribosyl transferase fusion variant proteins |
| GB0109297D0 (en) | 2001-04-12 | 2001-05-30 | Glaxosmithkline Biolog Sa | Vaccine |
| US20050192530A1 (en) * | 2001-04-13 | 2005-09-01 | Penjet Corporation | Method and apparatus for needle-less injection with a degassed fluid |
| US6613010B2 (en) * | 2001-04-13 | 2003-09-02 | Penjet Corporation | Modular gas-pressured needle-less injector |
| JP2004529929A (ja) * | 2001-04-23 | 2004-09-30 | ニュクリスト ファーマシューティカルズ コーポレーション | アポトーシスの誘導およびマトリックスメタロプロテイナーゼの阻害のための金属の使用 |
| US6755220B2 (en) | 2001-04-27 | 2004-06-29 | Penjet Corporation | Method and apparatus for filling or refilling a needle-less injector |
| JP2005504513A (ja) | 2001-05-09 | 2005-02-17 | コリクサ コーポレイション | 前立腺癌の治療及び診断のための組成物及び方法 |
| MY134424A (en) | 2001-05-30 | 2007-12-31 | Saechsisches Serumwerk | Stable influenza virus preparations with low or no amount of thiomersal |
| US20100221284A1 (en) | 2001-05-30 | 2010-09-02 | Saech-Sisches Serumwerk Dresden | Novel vaccine composition |
| US6648850B2 (en) * | 2001-06-08 | 2003-11-18 | Bioject, Inc. | Durable needle-less jet injector apparatus and method |
| CN1253220C (zh) * | 2001-06-29 | 2006-04-26 | 贝克顿迪肯森公司 | 通过微管在真皮内输入疫苗和基因治疗剂 |
| US20060018877A1 (en) * | 2001-06-29 | 2006-01-26 | Mikszta John A | Intradermal delivery of vacccines and therapeutic agents |
| US7544188B2 (en) * | 2001-07-19 | 2009-06-09 | Intelliject, Inc. | Medical injector |
| US7767872B2 (en) * | 2001-09-06 | 2010-08-03 | Mpg Biotechnologies, Llc | Thimerosal removal device |
| US8440791B2 (en) * | 2001-09-06 | 2013-05-14 | Mgp Biotechnologies, Llc | Thimerosal removal device |
| US6669664B2 (en) | 2001-09-07 | 2003-12-30 | Avant Drug Delivery Systems, Inc. | Vacuum control cycle for jet injector |
| MXPA04002631A (es) * | 2001-09-20 | 2004-07-08 | Glaxo Group Ltd | Vacunas de adn optimizadas por codon gag-vih. |
| US6824526B2 (en) | 2001-10-22 | 2004-11-30 | Penjet Corporation | Engine and diffuser for use with a needle-less injector |
| GB0125506D0 (en) | 2001-10-24 | 2001-12-12 | Weston Medical Ltd | Needle free injection method and apparatus |
| DE60236949D1 (de) * | 2001-11-09 | 2010-08-19 | Alza Corp | Pneumatisch angetriebener autoinjektor |
| US6607510B2 (en) | 2001-11-09 | 2003-08-19 | Bioject Medical Technologies Inc. | Disposable needle-free injection apparatus and method |
| CA2465901C (en) * | 2001-11-09 | 2010-10-26 | Pedro De La Serna | Collapsible syringe cartridge |
| TWI222370B (en) * | 2001-12-14 | 2004-10-21 | Roche Diagnostics Gmbh | Needleless hypodermic injection device |
| DK2224012T3 (da) | 2001-12-17 | 2013-05-13 | Corixa Corp | Sammensætninger og fremgangsmåder til terapi og diagnose af inflammatoriske tarmsygdomme |
| WO2003074102A2 (en) * | 2002-03-04 | 2003-09-12 | Nano Pass Technologies Ltd. | Devices and methods for transporting fluid across a biological barrier |
| US6912417B1 (en) | 2002-04-05 | 2005-06-28 | Ichor Medical Systmes, Inc. | Method and apparatus for delivery of therapeutic agents |
| MXPA04009959A (es) | 2002-04-12 | 2004-12-13 | Schering Corp | Ligandos del receptor acoplado a la proteina g y metodos. |
| DK1503788T3 (da) | 2002-04-25 | 2011-10-17 | Shire Human Genetic Therapies | Behandling af alpha-galactosidase A-deficiens |
| EP1506286B1 (de) | 2002-05-24 | 2014-03-19 | Merck Sharp & Dohme Corp. | Neutralisierung eines menschlichen anti-igfr-antikörpers |
| US6676630B2 (en) | 2002-06-04 | 2004-01-13 | Bioject Medical Technologies, Inc. | Needle-free injection system |
| US7238167B2 (en) * | 2002-06-04 | 2007-07-03 | Bioject Inc. | Needle-free injection system |
| US7156823B2 (en) * | 2002-06-04 | 2007-01-02 | Bioject Inc. | High workload needle-free injection system |
| US20040132058A1 (en) | 2002-07-19 | 2004-07-08 | Schering Corporation | NPC1L1 (NPC3) and methods of use thereof |
| US6883222B2 (en) * | 2002-10-16 | 2005-04-26 | Bioject Inc. | Drug cartridge assembly and method of manufacture |
| BRPI0315295C1 (pt) | 2002-10-17 | 2021-05-25 | Genmab As | anticorpo monoclonal humano isolado, célula hospedeira procariótica, composição farmacêutica, molécula biespecífica, usos de um anticorpo, métodos in vitro de detectar a presença de antígeno de cd20 ou uma célula que expressa cd20 em uma amostra, kit, e, vetor de expressão |
| US20040110738A1 (en) * | 2002-10-22 | 2004-06-10 | Gillis Scott H. | Prophylactic treatment methods |
| US7018356B2 (en) | 2002-10-31 | 2006-03-28 | Wise Roger R | Method and apparatus for adjusting the contents of a needle-less injector |
| DE10254601A1 (de) | 2002-11-22 | 2004-06-03 | Ganymed Pharmaceuticals Ag | Differentiell in Tumoren exprimierte Genprodukte und deren Verwendung |
| AU2003275895B2 (en) | 2002-11-25 | 2009-02-05 | Tecpharma Licensing Ag | Auto-injector comprising a resettable releasing safety device |
| CN101863930A (zh) | 2003-01-06 | 2010-10-20 | 科里克萨有限公司 | 一些氨烷基氨基葡糖苷磷酸酯化合物和它们的用途 |
| US7960522B2 (en) | 2003-01-06 | 2011-06-14 | Corixa Corporation | Certain aminoalkyl glucosaminide phosphate compounds and their use |
| DK1587907T3 (da) | 2003-01-07 | 2011-04-04 | Dyax Corp | Kunitz-domænebibliotek |
| EP2857036B1 (de) | 2003-01-31 | 2016-12-21 | Mount Sinai School of Medicine of New York University | Kombinationstherapie zur behandlung von proteinmangelstörungen |
| US6935384B2 (en) * | 2003-02-19 | 2005-08-30 | Bioject Inc. | Needle-free injection system |
| FR2852517B1 (fr) * | 2003-03-21 | 2005-11-11 | Dispositif d'injection sans aiguille a cartouche pyrotechnique et procede d'assemblage d'un tel dispositif | |
| EP1620147A4 (de) * | 2003-04-21 | 2008-06-11 | Corium Internat Inc | Gerät und verfahren für die repetitive mikrostrahl-arzneimittelabgabe |
| US20060184101A1 (en) * | 2003-04-21 | 2006-08-17 | Ravi Srinivasan | Microjet devices and methods for drug delivery |
| JP2007525458A (ja) | 2003-04-23 | 2007-09-06 | メダレックス インコーポレイテッド | インターフェロンα受容体−1(IFNAR−1)に対するヒト化抗体 |
| WO2004110533A1 (ja) * | 2003-05-09 | 2004-12-23 | Anges Mg, Inc. | 薬剤が収容された針無注射器 |
| WO2005026375A2 (en) | 2003-05-22 | 2005-03-24 | Fraunhofer Usa, Inc. | Recombinant carrier molecule for expression, delivery and purification of target polypeptides |
| WO2005034949A1 (en) * | 2003-09-09 | 2005-04-21 | University Of Florida | Desferrithiocin derivatives and their use as iron chelators |
| IL157981A (en) | 2003-09-17 | 2014-01-30 | Elcam Medical Agricultural Cooperative Ass Ltd | Auto injector |
| GB0325836D0 (en) * | 2003-11-05 | 2003-12-10 | Celltech R&D Ltd | Biological products |
| EP1685865A4 (de) * | 2003-11-21 | 2009-12-30 | Zhongshan Botai Pharmaceutic I | Spritze zum vormischen eines pulverförmigen medikaments |
| HUE026260T2 (en) | 2003-11-21 | 2016-06-28 | Ucb Biopharma Sprl | A method for treating multiple sclerosis by inhibiting IL-17 activity |
| PL1691837T3 (pl) | 2003-12-10 | 2012-11-30 | Squibb & Sons Llc | IP-10 przeciwciała i ich zastosowanie |
| KR101225299B1 (ko) | 2003-12-10 | 2013-01-24 | 메다렉스, 인코포레이티드 | 인터페론 알파 항체 및 그의 용도 |
| EP1713900A4 (de) * | 2004-01-27 | 2009-06-17 | Compugen Ltd | Verfahren und systeme zur kommentierung biomolekularer sequenzen |
| HUE025052T2 (en) | 2004-03-08 | 2016-01-28 | Ichor Medical Systems Inc | A sharpened device for electrically mediated delivery of medical substances |
| IL160891A0 (en) | 2004-03-16 | 2004-08-31 | Auto-mix needle | |
| DE102004024617A1 (de) | 2004-05-18 | 2005-12-29 | Ganymed Pharmaceuticals Ag | Differentiell in Tumoren exprimierte Genprodukte und deren Verwendung |
| EP1753458A4 (de) * | 2004-05-19 | 2009-07-22 | Wyeth Corp | Modulation der immunglobulinproduktion und atopische erkrankungen |
| US7717874B2 (en) * | 2004-05-28 | 2010-05-18 | Bioject, Inc. | Needle-free injection system |
| US7662381B2 (en) | 2004-06-21 | 2010-02-16 | Medarex, Inc. | Interferon alpha receptor 1 antibodies and their uses |
| CA2575791A1 (en) | 2004-08-03 | 2006-02-16 | Dyax Corp. | Hk1-binding proteins |
| GB0417852D0 (en) | 2004-08-11 | 2004-09-15 | Biotica Tech Ltd | Production of polyketides and other natural products |
| US20060040895A1 (en) * | 2004-08-19 | 2006-02-23 | Kipling Thacker | Aesthetic use of hyaluronan |
| CA2478458A1 (en) * | 2004-08-20 | 2006-02-20 | Michael Panzara | Treatment of pediatric multiple sclerosis |
| NZ553776A (en) | 2004-09-22 | 2010-05-28 | Glaxosmithkline Biolog Sa | Immunogenic composition comprising staphylococcal PNAG and Type 5 and/or 8 Capsular polysaccharide or oligosaccharide. |
| WO2007001448A2 (en) * | 2004-11-04 | 2007-01-04 | Massachusetts Institute Of Technology | Coated controlled release polymer particles as efficient oral delivery vehicles for biopharmaceuticals |
| CA2585719A1 (en) | 2004-11-08 | 2006-05-18 | Schering Corporation | Tumor association of mdl-1 and methods |
| CA2629751A1 (en) | 2004-11-12 | 2006-05-18 | The University Of British Columbia | Antimicrobial peptides |
| CA2587597A1 (en) | 2004-11-19 | 2006-05-26 | Biogen Idec Ma Inc. | Treatment for multiple sclerosis |
| US10737028B2 (en) | 2004-11-22 | 2020-08-11 | Kaleo, Inc. | Devices, systems and methods for medicament delivery |
| US11590286B2 (en) | 2004-11-22 | 2023-02-28 | Kaleo, Inc. | Devices, systems and methods for medicament delivery |
| US7947017B2 (en) * | 2004-11-22 | 2011-05-24 | Intelliject, Inc. | Devices, systems and methods for medicament delivery |
| US7648483B2 (en) | 2004-11-22 | 2010-01-19 | Intelliject, Inc. | Devices, systems and methods for medicament delivery |
| EP1814611A4 (de) | 2004-11-22 | 2010-06-02 | Intelliject Inc | Vorrichtungen, systeme und verfahren zur abgabe eines medikaments |
| US7648482B2 (en) * | 2004-11-22 | 2010-01-19 | Intelliject, Inc. | Devices, systems, and methods for medicament delivery |
| BRPI0515759B8 (pt) * | 2004-12-01 | 2021-06-22 | Acushot Inc | dispositivo de injeção sem agulha e kit para utilização do mesmo |
| NZ581497A (en) * | 2004-12-03 | 2012-07-27 | Biogen Idec Inc | Delaying or preventing onset of multiple sclerosis by vla-4 ginding antibody |
| PT1828249E (pt) | 2004-12-03 | 2011-02-25 | Schering Corp | Biomarcadores para a pré-selecção de pacientes para terapêutica anti-igf1r |
| US7351226B1 (en) * | 2004-12-07 | 2008-04-01 | Glenn Herskowitz | Medical infusion pump |
| JP2008527996A (ja) * | 2005-01-25 | 2008-07-31 | スカイ、ジェネティクス、インク | 癌細胞のアポトーシスのための核酸 |
| US20060183893A1 (en) * | 2005-01-25 | 2006-08-17 | North Don A | Nucleic acids for apoptosis of cancer cells |
| US8361026B2 (en) | 2005-02-01 | 2013-01-29 | Intelliject, Inc. | Apparatus and methods for self-administration of vaccines and other medicaments |
| GB2451769B (en) | 2005-02-01 | 2009-12-09 | Intelliject Llc | Devices, systems, and methods for medicament delivery |
| US9022980B2 (en) | 2005-02-01 | 2015-05-05 | Kaleo, Inc. | Medical injector simulation device |
| US8231573B2 (en) | 2005-02-01 | 2012-07-31 | Intelliject, Inc. | Medicament delivery device having an electronic circuit system |
| US7731686B2 (en) | 2005-02-01 | 2010-06-08 | Intelliject, Inc. | Devices, systems and methods for medicament delivery |
| US8206360B2 (en) | 2005-02-01 | 2012-06-26 | Intelliject, Inc. | Devices, systems and methods for medicament delivery |
| WO2006089095A2 (en) | 2005-02-17 | 2006-08-24 | Biogen Idec Ma Inc. | Treating neurological disorders |
| ES2498794T3 (es) | 2005-02-18 | 2014-09-25 | Medarex, L.L.C. | Anticuerpos monoclonales contra CD30 que carecen de restos fucosilo |
| US20100061994A1 (en) * | 2005-03-11 | 2010-03-11 | Rose Mary Sheridan | Medical uses of 39-desmethoxyrapamycin and analogues thereof |
| GB0504994D0 (en) * | 2005-03-11 | 2005-04-20 | Biotica Tech Ltd | Novel compounds |
| JP5225069B2 (ja) | 2005-03-23 | 2013-07-03 | ゲンマブ エー/エス | 多発性骨髄腫の治療のためのcd38に対する抗体 |
| AR054020A1 (es) | 2005-03-23 | 2007-05-30 | Glaxosmithkline Biolog Sa | Nuevo uso |
| EP1863519B1 (de) | 2005-03-31 | 2013-09-25 | The General Hospital Corporation | Modulation von hgf/hgfr-aktivität zur behandlung von lymphödem |
| BRPI0610644B8 (pt) | 2005-04-04 | 2021-05-25 | Univ Florida | composto e composição farmacêutica que compreende tal composto e um veículo ou diluente farmaceuticamente aceitável. |
| DK2439273T3 (da) | 2005-05-09 | 2019-06-03 | Ono Pharmaceutical Co | Humane monoklonale antistoffer til programmeret død-1(pd-1) og fremgangsmåder til behandling af cancer ved anvendelse af anti-pd-1- antistoffer alene eller i kombination med andre immunterapeutika |
| EP3782655A1 (de) | 2005-05-17 | 2021-02-24 | Amicus Therapeutics, Inc. | Verfahren zur behandlung der pompe-krankheit unter verwendung von 1-deoxynojirimycin und derivaten |
| MEP35408A (en) | 2005-05-27 | 2011-02-10 | Biogen Idec Inc | Tweak binding antibodies |
| US20060271014A1 (en) * | 2005-05-31 | 2006-11-30 | Mallinckrodt Inc. | Heat retention device for a syringe and methods of use |
| US20070118093A1 (en) * | 2005-06-20 | 2007-05-24 | Von Muhlen Marcio G | High-speed jet devices for drug delivery |
| PT1907424E (pt) | 2005-07-01 | 2015-10-09 | Squibb & Sons Llc | Anticorpos monoclonais humanos para o ligando 1 de morte programada (pd-l1) |
| US20100119525A1 (en) * | 2005-08-01 | 2010-05-13 | Mount Sinai Schoool Of Medicine Of New York University | Method for extending longevity using npc1l1 antagonists |
| JP2009502207A (ja) * | 2005-08-03 | 2009-01-29 | フラウンホーファー ユーエスエー, インコーポレイテッド | イムノグロブリンの産生のための組成物および方法 |
| US8591457B2 (en) * | 2005-08-10 | 2013-11-26 | Alza Corporation | Method for making a needle-free jet injection drug delivery device |
| EP1762575A1 (de) * | 2005-09-12 | 2007-03-14 | Ganymed Pharmaceuticals AG | Identifizierung Tumor-assoziierter Antigene für Diagnoseund Therapie |
| NZ566395A (en) | 2005-09-26 | 2012-03-30 | Medarex Inc | Human monoclonal antibodies to CD70 |
| WO2007045477A2 (en) | 2005-10-21 | 2007-04-26 | Novartis Ag | Human antibodies against il-13 and therapeutic uses |
| US20070099246A1 (en) * | 2005-11-03 | 2007-05-03 | Sandy John D | Antibodies, assays and kits to quantitate cartilage destruction |
| CN103071209A (zh) * | 2005-11-17 | 2013-05-01 | 周吉尼克斯股份有限公司 | 用无针注射递送粘稠制剂 |
| EP1790664A1 (de) | 2005-11-24 | 2007-05-30 | Ganymed Pharmaceuticals AG | Monoklonale Antikörper gegen Claudin-18 zur Behandlung von Krebs |
| US7695449B2 (en) * | 2005-12-05 | 2010-04-13 | Xinming Wang | Automatic syringe |
| US8383118B2 (en) | 2005-12-08 | 2013-02-26 | Medarex, Inc. | Human monoclonal antibodies to fucosyl-GM1 and methods for using anti-fucosyl-GM1 |
| TWI457133B (zh) | 2005-12-13 | 2014-10-21 | Glaxosmithkline Biolog Sa | 新穎組合物 |
| WO2007070682A2 (en) * | 2005-12-15 | 2007-06-21 | Massachusetts Institute Of Technology | System for screening particles |
| KR101515078B1 (ko) | 2005-12-22 | 2015-04-24 | 글락소스미스클라인 바이오로지칼즈 에스.에이. | 백신 |
| GB0607088D0 (en) | 2006-04-07 | 2006-05-17 | Glaxosmithkline Biolog Sa | Vaccine |
| EP2441846A3 (de) | 2006-01-09 | 2012-07-25 | The Regents Of the University of California | Immunstimulierende Kombinationen aus TNFRSF, TLR, NLR, RHR, purinergischem Rezeptor, und Zytokin-Rezeptor-Antagonisten für Impfstoffe und die Tumorimmuntherapie |
| US20070166774A1 (en) * | 2006-01-17 | 2007-07-19 | Groman Ernest V | Functional immunoassay |
| HRP20120994T1 (hr) | 2006-02-07 | 2012-12-31 | Shire Human Genetic Therapies, Inc. | Stabilizirana smjesa glukocerebrozidaze |
| CN104162200B (zh) * | 2006-02-09 | 2018-03-27 | 德卡产品有限公司 | 外围系统 |
| KR20080106434A (ko) * | 2006-02-13 | 2008-12-05 | 프라운호퍼 유에스에이, 인코포레이티드 | Hpv 항원, 백신 조성물 및 관련된 방법 |
| US8277816B2 (en) * | 2006-02-13 | 2012-10-02 | Fraunhofer Usa, Inc. | Bacillus anthracis antigens, vaccine compositions, and related methods |
| BRPI0707733B1 (pt) * | 2006-02-13 | 2019-12-31 | Ibio, Inc. | antígeno isolado, composição de vacina, uso da referida composição e método para produção de uma proteína de antígeno |
| AU2007218014A1 (en) * | 2006-02-17 | 2007-08-30 | Alan L. Buchman | Catheter cleaning devices |
| WO2007106415A2 (en) * | 2006-03-10 | 2007-09-20 | Massachusetts Institute Of Technology | Triggered self-assembly conjugates and nanosystems |
| WO2007111806A2 (en) | 2006-03-23 | 2007-10-04 | Massachusetts Eye And Ear Infirmary | Cyclopentane heptanoic acid compounds for reducing body fat |
| WO2007121054A2 (en) * | 2006-03-27 | 2007-10-25 | Sky Genetics, Inc. | Nucleic acids for apoptosis of cancer cells |
| EP2476433A1 (de) | 2006-03-30 | 2012-07-18 | GlaxoSmithKline Biologicals S.A. | Immunogene Zusammensetzung |
| CA2648099C (en) * | 2006-03-31 | 2012-05-29 | The Brigham And Women's Hospital, Inc | System for targeted delivery of therapeutic agents |
| US20090325944A1 (en) * | 2006-04-12 | 2009-12-31 | Suzanne Walker Kahne | Methods and Compositions for Modulating Glycosylation |
| AU2007240732B2 (en) * | 2006-04-21 | 2013-07-04 | Amgen, Inc. | Buffering agents for biopharmaceutical formulations |
| US20070258941A1 (en) * | 2006-05-02 | 2007-11-08 | Pfister Brian E | Methods and compositions for remediation of disc herniation by modifying structure |
| JP5630998B2 (ja) | 2006-05-15 | 2014-11-26 | マサチューセッツ インスティテュート オブ テクノロジー | 機能的粒子のためのポリマー |
| WO2007137117A2 (en) * | 2006-05-17 | 2007-11-29 | Massachusetts Institute Of Technology | Aptamer-directed drug delivery |
| PL2034830T3 (pl) | 2006-05-25 | 2015-04-30 | Biogen Ma Inc | Przeciwciało anty-VLA-1 do leczenia udaru |
| EP2030015B1 (de) * | 2006-06-02 | 2016-02-17 | President and Fellows of Harvard College | Neugestaltung von proteinoberflächen |
| WO2007150030A2 (en) | 2006-06-23 | 2007-12-27 | Massachusetts Institute Of Technology | Microfluidic synthesis of organic nanoparticles |
| WO2008005469A2 (en) | 2006-06-30 | 2008-01-10 | Schering Corporation | Igfbp2 biomarker |
| CA2656620C (en) | 2006-07-04 | 2018-03-13 | Genmab A/S | Cd20 binding molecules for the treatment of copd |
| PT2422810E (pt) | 2006-07-17 | 2014-12-03 | Glaxosmithkline Biolog Sa | Vacina da gripe |
| US20080033347A1 (en) * | 2006-08-03 | 2008-02-07 | Becton, Dickinson And Company | Detachable needle syringe having reduced dead space |
| US7618396B2 (en) * | 2006-08-09 | 2009-11-17 | Avant Medical Corp. | Injection system with hidden needles |
| JP5244785B2 (ja) | 2006-08-11 | 2013-07-24 | 小野薬品工業株式会社 | ストローマ由来因子−1(sdf−1)に対するモノクローナル抗体 |
| US9115358B2 (en) * | 2006-08-11 | 2015-08-25 | President And Fellows Of Harvard College | Moenomycin biosynthesis-related compositions and methods of use thereof |
| WO2008073161A2 (en) * | 2006-08-14 | 2008-06-19 | Massachusetts Institute Of Technology | Hemagglutinin polypeptides, and reagents and methods relating thereto |
| US20090269342A1 (en) * | 2006-08-14 | 2009-10-29 | Massachusetts Institute Of Technology | Hemagglutinin Polypeptides, and Reagents and Methods Relating Thereto |
| CA2662350A1 (en) | 2006-09-05 | 2008-03-13 | Medarex, Inc. | Antibodies to bone morphogenic proteins and receptors therefor and methods for their use |
| US7942845B2 (en) * | 2006-09-19 | 2011-05-17 | Bioject, Inc. | Needle-free injector and process for providing serial injections |
| MX2009003306A (es) | 2006-10-02 | 2009-04-23 | Medarex Inc | Anticuerpos humanos que se unen a cxcr4 y sus usos. |
| TW200831129A (en) * | 2006-10-06 | 2008-08-01 | Amgen Inc | Stable formulations |
| US7547293B2 (en) | 2006-10-06 | 2009-06-16 | Bioject, Inc. | Triggering mechanism for needle-free injector |
| JO3598B1 (ar) | 2006-10-10 | 2020-07-05 | Infinity Discovery Inc | الاحماض والاسترات البورونية كمثبطات اميد هيدروليز الحامض الدهني |
| EP2433648A3 (de) | 2006-10-12 | 2012-04-04 | GlaxoSmithKline Biologicals S.A. | Impfstoff mit einem Öl-in-wasser-emulsionshilfsstoff |
| EA015817B1 (ru) | 2006-10-12 | 2011-12-30 | Глаксосмитклайн Байолоджикалс С.А. | Иммуногенная композиция, содержащая адъювант в виде эмульсии "масло в воде" |
| AU2007319605B2 (en) | 2006-10-19 | 2011-02-17 | Csl Limited | Anti-IL-13R alpha 1 antibodies and their uses thereof |
| EP2094247B1 (de) * | 2006-10-20 | 2022-06-29 | Amgen Inc. | Stabile polypeptid-formulierungen |
| CN101600450A (zh) | 2006-10-20 | 2009-12-09 | 比奥根艾迪克Ma公司 | 利用可溶性淋巴毒素β受体的脱髓鞘病的治疗 |
| US8618248B2 (en) | 2006-10-31 | 2013-12-31 | President And Fellows Of Harvard College | Phosphopeptide compositions and anti-phosphopeptide antibody compositions and methods of detecting phosphorylated peptides |
| US20090124996A1 (en) * | 2006-11-03 | 2009-05-14 | Scott Heneveld | Apparatus and methods for injecting high viscosity dermal fillers |
| EP2097447A4 (de) | 2006-11-15 | 2010-12-29 | Medarex Inc | Monoklonale menschliche antikörper gegen btla und verfahren zu ihrer verwendung |
| US20100303723A1 (en) * | 2006-11-20 | 2010-12-02 | Massachusetts Institute Of Technology | Drug delivery systems using fc fragments |
| US8481683B2 (en) | 2006-12-01 | 2013-07-09 | Medarex, Inc. | Human antibodies that bind CD22 and uses thereof |
| SG177184A1 (en) | 2006-12-01 | 2012-01-30 | Anterios Inc | Amphiphilic entity nanoparticles |
| WO2008073856A2 (en) * | 2006-12-08 | 2008-06-19 | Massachusetts Institute Of Technology | Delivery of nanoparticles and/or agents to cells |
| CL2007003622A1 (es) | 2006-12-13 | 2009-08-07 | Medarex Inc | Anticuerpo monoclonal humano anti-cd19; composicion que lo comprende; y metodo de inhibicion del crecimiento de celulas tumorales. |
| MX2009006277A (es) | 2006-12-14 | 2009-07-24 | Medarex Inc | Anticuerpos humanos que se enlazan a cd70 y usos de los mismos. |
| CA2790018C (en) * | 2006-12-21 | 2015-02-03 | Amgen Inc. | Formulations |
| ES2405655T3 (es) | 2006-12-26 | 2013-05-31 | Lantheus Medical Imaging, Inc. | N-[3-bromo-4-(3-[18F]fluoropropoxi)-bencil]-guanidina para la formación de imágenes de la inervación cardíaca |
| WO2008083209A2 (en) * | 2006-12-29 | 2008-07-10 | Amir Genosar | Hypodermic drug delivery reservoir and apparatus |
| WO2008091838A2 (en) | 2007-01-22 | 2008-07-31 | Intelliject, Inc. | Medical injector with compliance tracking and monitoring |
| DK2118123T3 (en) | 2007-01-31 | 2016-01-25 | Dana Farber Cancer Inst Inc | Stabilized p53 peptides and uses thereof |
| EP2134830A2 (de) * | 2007-02-09 | 2009-12-23 | Massachusetts Institute of Technology | Oszillations-zellkultur-bioreaktor |
| KR101598229B1 (ko) | 2007-02-16 | 2016-02-26 | 메리맥 파마슈티컬즈, 인크. | Erbb3에 대한 항체 및 이의 용도 |
| US7744563B2 (en) * | 2007-02-23 | 2010-06-29 | Bioject, Inc. | Needle-free injection devices and drug delivery systems therefor |
| EP1970384A1 (de) | 2007-03-14 | 2008-09-17 | Ganymed Pharmaceuticals AG | Monoklonale Antikörper zur Krebsbehandlung |
| JP5439193B2 (ja) | 2007-03-15 | 2014-03-12 | ユニバーシティー オブ フロリダ リサーチ ファンデーション, インク. | デスフェリチオシンポリエーテル類似体 |
| US7960139B2 (en) | 2007-03-23 | 2011-06-14 | Academia Sinica | Alkynyl sugar analogs for the labeling and visualization of glycoconjugates in cells |
| CN101679974B (zh) | 2007-03-27 | 2015-09-30 | 航道生物技术有限责任公司 | 包含抗体替代轻链序列的构建体和文库 |
| US8592377B2 (en) | 2007-03-28 | 2013-11-26 | President And Fellows Of Harvard College | Stitched polypeptides |
| JP2010523595A (ja) | 2007-04-04 | 2010-07-15 | マサチューセッツ インスティテュート オブ テクノロジー | ポリ(アミノ酸)ターゲッティング部分 |
| WO2008124634A1 (en) | 2007-04-04 | 2008-10-16 | Massachusetts Institute Of Technology | Polymer-encapsulated reverse micelles |
| EP2152301A4 (de) * | 2007-04-28 | 2010-07-28 | Fraunhofer Usa Inc | Trypanosomaantigene, impfstoffzusammensetzungen und zugehörige verfahren |
| EP2167480A2 (de) | 2007-05-23 | 2010-03-31 | Vical Incorporated | Zusammensetzungen und verfahren zur verbesserung der immunreaktionen auf impfstoffe |
| EP1997832A1 (de) | 2007-05-29 | 2008-12-03 | Ganymed Pharmaceuticals AG | Monoklonale Antikörper gegen Claudin-18 zur Behandlung von Krebs |
| CA2687896A1 (en) | 2007-06-05 | 2008-12-18 | Yale University | Antibody directed against the d4 ig-like domain of the kit receptor |
| KR20100045413A (ko) * | 2007-06-14 | 2010-05-03 | 크루셀 스위츨랜드 아게 | 피내 인플루엔자 백신 |
| NZ595526A (en) * | 2007-06-14 | 2013-03-28 | Biogen Idec Inc | Pharmaceutical composition comprising a vla-4 binding antibody, a phosphate buffer and a surfactant |
| US8524444B2 (en) | 2007-06-15 | 2013-09-03 | President And Fellows Of Harvard College | Methods and compositions for detections and modulating O-glycosylation |
| CA2690708A1 (en) | 2007-06-26 | 2008-12-31 | Glaxosmithkline Biologicals S.A. | Vaccine |
| WO2009009759A2 (en) | 2007-07-11 | 2009-01-15 | Fraunhofer Usa, Inc. | Yersinia pestis antigens, vaccine compositions, and related methods |
| DE102007034871A1 (de) * | 2007-07-24 | 2009-01-29 | Lts Lohmann Therapie-Systeme Ag | Einweginjektor mit handbetätigbarem Kolben |
| JP5588866B2 (ja) | 2007-08-10 | 2014-09-10 | メダレックス エル.エル.シー. | Hco32およびhco27、ならびに関連実施例 |
| WO2009026397A2 (en) * | 2007-08-20 | 2009-02-26 | Fraunhofer Usa, Inc. | Prophylactic and therapeutic influenza vaccines, antigens, compositions, and methods |
| JP5607530B2 (ja) | 2007-09-04 | 2014-10-15 | コンピュゲン エルティーディー. | ポリペプチド並びにポリヌクレオチド、並びに薬剤および生物製剤生産のための薬剤標的としてのその利用 |
| GB0717337D0 (en) | 2007-09-06 | 2007-10-17 | Ucb Pharma Sa | Method of treatment |
| EP2205074A4 (de) * | 2007-10-04 | 2013-07-31 | Harvard College | Moenomycin-analoga, syntheseverfahren und verwendung |
| JP2011500569A (ja) * | 2007-10-12 | 2011-01-06 | マサチューセッツ インスティテュート オブ テクノロジー | ワクチンナノテクノロジー |
| TWI489993B (zh) | 2007-10-12 | 2015-07-01 | Novartis Ag | 骨硬化素(sclerostin)抗體組合物及使用方法 |
| JP2011500036A (ja) * | 2007-10-15 | 2011-01-06 | ザ ユニバーシティー オブ クイーンズランド | 構築物系およびその使用 |
| BRPI0820327A2 (pt) | 2007-11-02 | 2020-10-06 | Novartis Ag | moléculas e métodos para modulação de proteína relacionada ao receptor de lipoproteína de baixa densidade 6 (lrp6) |
| CN101888856B (zh) | 2007-11-07 | 2014-08-27 | 塞尔德克斯医疗公司 | 结合人树突和上皮细胞205(dec-205)的抗体 |
| US8617099B2 (en) * | 2007-11-26 | 2013-12-31 | Bioject Inc. | Injection device plunger auto-disable |
| US20090137949A1 (en) * | 2007-11-26 | 2009-05-28 | Bioject Inc. | Needle-free injection device with nozzle auto-disable |
| BRPI0822049B1 (pt) | 2007-12-17 | 2021-11-16 | Pfizer Limited | Composiqao farmaceutica compreendendo anticorpo antagonista anti-ngf, kit e uso de um anticorpo antingf |
| WO2009086250A1 (en) | 2007-12-21 | 2009-07-09 | Aesthetic Sciences Corporation | Self-contained pressurized injection device |
| WO2009086463A1 (en) | 2007-12-28 | 2009-07-09 | Aktivpak, Inc. | Dispenser and therapeutic package suitable for administering a therapeutic substance to a subject |
| DE102008011873A1 (de) | 2007-12-30 | 2009-07-09 | Idelevich, Evgeny | Applikationsvorrichtung und -verfahren zur automatisierten Medikamentenabgabe bei arteriellem Hypertonus |
| WO2009089119A2 (en) * | 2008-01-03 | 2009-07-16 | Massachusetts Institute Of Technology | Decoy influenza therapies |
| US8193182B2 (en) | 2008-01-04 | 2012-06-05 | Intellikine, Inc. | Substituted isoquinolin-1(2H)-ones, and methods of use thereof |
| WO2009093189A2 (en) | 2008-01-22 | 2009-07-30 | Fundacao De Amparo A Pesquisa Do Estado De Sao Paulo - Fapesp | Peptides, compositions, and uses thereof |
| EP2650017A3 (de) | 2008-02-05 | 2014-01-22 | Bristol-Myers Squibb Company | Alpha-5-beta-1-Antikörper und deren Verwendungen |
| KR20160070165A (ko) | 2008-02-08 | 2016-06-17 | 메디뮨 엘엘씨 | Fc 리간드 친화성이 감소된 항-IFNAR1 항체 |
| CA2721060A1 (en) * | 2008-04-09 | 2009-10-15 | Infinity Pharmaceuticals, Inc. | Inhibitors of fatty acid amide hydrolase |
| ES2553113T3 (es) | 2008-04-16 | 2015-12-04 | Glaxosmithkline Biologicals S.A. | Vacuna |
| JP2011523353A (ja) * | 2008-04-28 | 2011-08-11 | プレジデント アンド フェロウズ オブ ハーバード カレッジ | 細胞透過のための過剰に荷電されたタンパク質 |
| ES2563061T3 (es) * | 2008-04-28 | 2016-03-10 | Zogenix, Inc. | Nuevas formulaciones para el tratamiento de la migraña |
| EA201001734A1 (ru) | 2008-05-02 | 2011-10-31 | Новартис Аг | Улучшенные связывающие молекулы на основе фибронектина и их применение |
| US8021344B2 (en) | 2008-07-28 | 2011-09-20 | Intelliject, Inc. | Medicament delivery device configured to produce an audible output |
| USD994111S1 (en) | 2008-05-12 | 2023-08-01 | Kaleo, Inc. | Medicament delivery device cover |
| CA2725911A1 (en) | 2008-05-30 | 2009-12-23 | Dana-Farber Cancer Institute, Inc. | Methods of treating a meiotic kinesin-associated disease |
| WO2009158145A2 (en) * | 2008-05-30 | 2009-12-30 | Allergan, Inc. | Injection device for soft-tissue augmentation fillers, bioactive agents and other biocompatible materials in liquid or gel form |
| WO2009150623A1 (en) | 2008-06-13 | 2009-12-17 | Pfizer Inc | Treatment of chronic prostatitis |
| WO2009155513A2 (en) | 2008-06-20 | 2009-12-23 | Novartis Ag | Immunoglobulins with reduced aggregation |
| KR101848095B1 (ko) | 2008-06-26 | 2018-04-11 | 안테리오스, 인코퍼레이티드 | 경피 운반 |
| ES2442024T3 (es) | 2008-07-15 | 2014-02-07 | Academia Sinica | Matrices de glucano sobre portaobjetos de vidrio revestidos con aluminio de tipo PTFE y métodos relacionados |
| KR20110033304A (ko) * | 2008-07-23 | 2011-03-30 | 메사추세츠 인스티튜트 오브 테크놀로지 | Dna 손상에 대해 보호하고 신경세포 생존을 증가시키는 히스톤 디아세틸라제 1 (hdac1)의 활성화 |
| EP2356139A4 (de) | 2008-07-23 | 2013-01-09 | Harvard College | Ligieren zusammengehefteter polypeptide |
| EP2316030B1 (de) | 2008-07-25 | 2019-08-21 | Wagner, Richard W. | Proteinscreeningverfahren |
| AR072999A1 (es) | 2008-08-11 | 2010-10-06 | Medarex Inc | Anticuerpos humanos que se unen al gen 3 de activacion linfocitaria (lag-3) y los usos de estos |
| EP2166021A1 (de) * | 2008-09-16 | 2010-03-24 | Ganymed Pharmaceuticals AG | Monoklonale Antikörper zur Krebsbehandlung |
| WO2010033882A2 (en) * | 2008-09-18 | 2010-03-25 | Becton, Dickinson And Company | Medical injector with slidable sleeve activation |
| US8552154B2 (en) | 2008-09-26 | 2013-10-08 | Emory University | Anti-PD-L1 antibodies and uses therefor |
| US8734803B2 (en) | 2008-09-28 | 2014-05-27 | Ibio Inc. | Humanized neuraminidase antibody and methods of use thereof |
| US8343498B2 (en) | 2008-10-12 | 2013-01-01 | Massachusetts Institute Of Technology | Adjuvant incorporation in immunonanotherapeutics |
| US8277812B2 (en) * | 2008-10-12 | 2012-10-02 | Massachusetts Institute Of Technology | Immunonanotherapeutics that provide IgG humoral response without T-cell antigen |
| US8343497B2 (en) | 2008-10-12 | 2013-01-01 | The Brigham And Women's Hospital, Inc. | Targeting of antigen presenting cells with immunonanotherapeutics |
| US8591905B2 (en) | 2008-10-12 | 2013-11-26 | The Brigham And Women's Hospital, Inc. | Nicotine immunonanotherapeutics |
| CA2738243C (en) | 2008-10-29 | 2020-09-29 | Wyeth Llc | Formulations of single domain antigen binding molecules |
| CA2743904A1 (en) | 2008-11-17 | 2010-05-20 | The Regents Of The University Of Michigan | Cancer vaccine compositions and methods of using the same |
| EP2193817A1 (de) * | 2008-12-02 | 2010-06-09 | Sanofi-Aventis Deutschland GmbH | Zur Verwendung in Arzneimittelabgabevorrichtungen geeignete Antriebsanordnung und Arzneimittelabgabevorrichtung |
| EP3187219B2 (de) * | 2008-12-02 | 2026-03-04 | Allergan, Inc. | Injektionsvorrichtung |
| US20110311450A1 (en) | 2008-12-08 | 2011-12-22 | Zurit Levine | Polypeptides and polynucleotides, and uses thereof as a drug target for producing drugs and biologics |
| WO2010067345A1 (en) * | 2008-12-08 | 2010-06-17 | Perf-Action Technologies Ltd. | Erosion prevention for o-rings in a high pressure injection device |
| US9637746B2 (en) | 2008-12-15 | 2017-05-02 | Greenlight Biosciences, Inc. | Methods for control of flux in metabolic pathways |
| EP2380023A2 (de) | 2008-12-19 | 2011-10-26 | Medirista Biotechnologies AB | Oxidiertes cardiolipin als neuer pro-entzündungsfaktor |
| CN105132387A (zh) * | 2008-12-22 | 2015-12-09 | 绿光生物科学公司 | 用于制造化合物的组合物和方法 |
| CN102341412B (zh) | 2009-03-05 | 2018-01-05 | 梅达雷克斯有限责任公司 | 特异于cadm1 的全人抗体 |
| GB0904540D0 (en) | 2009-03-17 | 2009-04-29 | Biotica Tech Ltd | Novel compounds and methods for their production |
| WO2010118159A1 (en) | 2009-04-07 | 2010-10-14 | Infinity Pharmaceuticals, Inc. | Inhibitors of fatty acid amide hydrolase |
| WO2010118155A1 (en) | 2009-04-07 | 2010-10-14 | Infinity Pharmaceuticals, Inc. | Inhibitors of fatty acid amide hydrolase |
| BRPI1010517A2 (pt) | 2009-04-09 | 2017-01-31 | Amicus Therapeutics Inc | métodos para prevenção e/ou tratamento de doenças de armazenamento lisossomal |
| JP2013525260A (ja) * | 2009-04-17 | 2013-06-20 | バイオジェン・アイデック・エムエイ・インコーポレイテッド | 急性骨髄性白血病を治療するための組成物および方法 |
| DK2421898T3 (en) | 2009-04-20 | 2016-05-30 | Oxford Biotherapeutics Ltd | Cadherin-17 SPECIFIC ANTIBODIES |
| US9242040B2 (en) * | 2009-04-23 | 2016-01-26 | Bayer Healthcare Llc | Syringe assemblies, methods of forming syringe assemblies and adapters for forming syringe assemblies |
| PE20120532A1 (es) | 2009-04-27 | 2012-05-18 | Novartis Ag | ANTICUERPOS ANTI-ActRIIB |
| JP2012524524A (ja) | 2009-04-27 | 2012-10-18 | ノバルティス アーゲー | IL−12レセプターβ1サブユニットに特異的な治療用抗体の組成物および使用方法 |
| EP2424877A4 (de) | 2009-04-28 | 2013-01-02 | Harvard College | Suprageladene proteine zur zellpenetration |
| SG175436A1 (en) | 2009-05-04 | 2011-12-29 | Abbott Res Bv | Antibodies against nerve growth factor (ngf) with enhanced in vivo stability |
| CA2761681A1 (en) | 2009-05-13 | 2010-11-18 | Sea Lane Biotechnologies, Llc | Neutralizing molecules to influenza viruses |
| US8765735B2 (en) * | 2009-05-18 | 2014-07-01 | Infinity Pharmaceuticals, Inc. | Isoxazolines as inhibitors of fatty acid amide hydrolase |
| US8927551B2 (en) * | 2009-05-18 | 2015-01-06 | Infinity Pharmaceuticals, Inc. | Isoxazolines as inhibitors of fatty acid amide hydrolase |
| US9149465B2 (en) * | 2009-05-18 | 2015-10-06 | Infinity Pharmaceuticals, Inc. | Isoxazolines as inhibitors of fatty acid amide hydrolase |
| CA2769999C (en) | 2009-06-01 | 2020-02-18 | Shriners Hospitals For Children | A cloned transmembrane receptor for 24-hydroxylated vitamin d compounds and uses thereof |
| US8974423B2 (en) * | 2009-06-01 | 2015-03-10 | Sanofi-Aventis Deutschland Gmbh | Resettable drug delivery device |
| WO2010141074A2 (en) | 2009-06-01 | 2010-12-09 | President And Fellows Of Harvard College | O-glcnac transferase inhibitors and uses thereof |
| MX2011012665A (es) | 2009-06-04 | 2012-03-07 | Novartis Ag | Metodos para identificacion de sitios para conjugacion de igg. |
| WO2010146511A1 (en) | 2009-06-17 | 2010-12-23 | Pfizer Limited | Treatment of overactive bladder |
| AU2010273220B2 (en) | 2009-07-13 | 2015-10-15 | President And Fellows Of Harvard College | Bifunctional stapled polypeptides and uses thereof |
| BR112012007875A2 (pt) | 2009-07-31 | 2016-11-22 | Medarex Inc | anticorpos totalmente humanos para btla |
| US8394922B2 (en) | 2009-08-03 | 2013-03-12 | Medarex, Inc. | Antiproliferative compounds, conjugates thereof, methods therefor, and uses thereof |
| GB0913681D0 (en) | 2009-08-05 | 2009-09-16 | Glaxosmithkline Biolog Sa | Immunogenic composition |
| GB0914589D0 (en) | 2009-08-20 | 2009-09-30 | Biotica Tech Ltd | Novel compounds and methods for their production |
| WO2011029823A1 (en) | 2009-09-09 | 2011-03-17 | Novartis Ag | Monoclonal antibody reactive with cd63 when expressed at the surface of degranulated mast cells |
| GB0916630D0 (en) | 2009-09-22 | 2009-11-04 | Secr Defence | Antibody |
| CN102712675A (zh) | 2009-09-22 | 2012-10-03 | 爱勒让治疗公司 | 拟肽大环化合物 |
| US8784819B2 (en) | 2009-09-29 | 2014-07-22 | Ibio Inc. | Influenza hemagglutinin antibodies, compositions and related methods |
| GB0917817D0 (en) | 2009-10-12 | 2009-11-25 | Biotica Tech Ltd | Novel compounds and methods for their production |
| GB0917816D0 (en) | 2009-10-12 | 2009-11-25 | Biotica Tech Ltd | Novel compounds and methods for their production |
| EP2470569A1 (de) | 2009-10-13 | 2012-07-04 | Oxford Biotherapeutics Ltd. | Antikörper gegen epha10 |
| AU2010306774A1 (en) | 2009-10-14 | 2012-05-03 | Merrimack Pharmaceuticals, Inc. | Bispecific binding agents targeting IGF-1R and ErbB3 signalling and uses thereof |
| KR101880411B1 (ko) | 2009-10-19 | 2018-07-19 | 아미쿠스 세라퓨틱스, 인코포레이티드 | 중추신경계의 퇴행성 질환을 예방 및/또는 치료하는 신규 조성물 |
| SI2995306T1 (sl) | 2009-10-19 | 2019-04-30 | Amicus Therapeutics, Inc. | Novi sestavki za preprečevanje in/ali zdravljenje motenj lizosomskega shranjevanja |
| WO2011051327A2 (en) | 2009-10-30 | 2011-05-05 | Novartis Ag | Small antibody-like single chain proteins |
| WO2011051466A1 (en) | 2009-11-02 | 2011-05-05 | Novartis Ag | Anti-idiotypic fibronectin-based binding molecules and uses thereof |
| US20120282276A1 (en) | 2009-11-05 | 2012-11-08 | The Regents Of The University Of Michigan | Biomarkers predictive of progression of fibrosis |
| EP2507265B1 (de) | 2009-12-01 | 2016-05-11 | Compugen Ltd. | Heparanase Spice-Variante T5 spezifischer Antikörper und dessen Verwendung. |
| US11377485B2 (en) | 2009-12-02 | 2022-07-05 | Academia Sinica | Methods for modifying human antibodies by glycan engineering |
| US10087236B2 (en) | 2009-12-02 | 2018-10-02 | Academia Sinica | Methods for modifying human antibodies by glycan engineering |
| US20110137260A1 (en) | 2009-12-07 | 2011-06-09 | Allergan, Inc. | Slotted syringe |
| EP3000885B1 (de) | 2009-12-18 | 2018-07-25 | Arrowhead Pharmaceuticals, Inc. | Organische zusammensetzungen zur behandlung von hsf1-bedingten erkrankungen |
| BR112012017164A2 (pt) | 2009-12-22 | 2019-09-24 | Novartis Ag | proteína de fusão de região constante de anticorpo-cd47 tetravalente |
| WO2011090738A2 (en) | 2009-12-29 | 2011-07-28 | Dana-Farber Cancer Institute, Inc. | Type ii raf kinase inhibitors |
| AU2011210840B2 (en) | 2010-01-27 | 2014-12-11 | Massachusetts Institute Of Technology | Engineered polypeptide agents for targeted broad spectrum influenza neutralization |
| WO2011092233A1 (en) | 2010-01-29 | 2011-08-04 | Novartis Ag | Yeast mating to produce high-affinity combinations of fibronectin-based binders |
| RU2015143910A (ru) | 2010-02-03 | 2018-12-28 | Инфинити Фармасьютикалз, Инк. | Ингибиторы амид-гидролазы жирных кислот |
| US20120296403A1 (en) | 2010-02-10 | 2012-11-22 | Novartis Ag | Methods and compounds for muscle growth |
| AU2011224457B2 (en) | 2010-03-08 | 2014-11-06 | Sloan-Kettering Institute For Cancer Research | Cdc7 kinase inhibitors and uses thereof |
| GB201003922D0 (en) | 2010-03-09 | 2010-04-21 | Glaxosmithkline Biolog Sa | Conjugation process |
| GB201003920D0 (en) | 2010-03-09 | 2010-04-21 | Glaxosmithkline Biolog Sa | Method of treatment |
| AU2011227335B2 (en) | 2010-03-17 | 2014-11-06 | Abbott Research B.V. | Anti-nerve growth factor (NGF) antibody compositions |
| EP2550285B1 (de) | 2010-03-22 | 2017-07-19 | President and Fellows of Harvard College | Trioxacarcine und deren verwendungen |
| EP2371864A1 (de) | 2010-03-23 | 2011-10-05 | Ganymed Pharmaceuticals AG | Monoklonale Antikörper zur Krebsbehandlung |
| US20150231215A1 (en) | 2012-06-22 | 2015-08-20 | Randolph J. Noelle | VISTA Antagonist and Methods of Use |
| US10745467B2 (en) | 2010-03-26 | 2020-08-18 | The Trustees Of Dartmouth College | VISTA-Ig for treatment of autoimmune, allergic and inflammatory disorders |
| MX374075B (es) | 2010-03-26 | 2025-03-05 | Dartmouth College | Proteina mediadora de celula t regulatoria vista, agentes de enlace de vista y uso de los mismos. |
| US10338069B2 (en) | 2010-04-12 | 2019-07-02 | Academia Sinica | Glycan arrays for high throughput screening of viruses |
| EA201690310A1 (ru) | 2010-04-13 | 2016-12-30 | Селлдекс Терапьютикс Инк. | Антитела, связывающие cd27 человека, и их применение |
| HRP20171045T1 (hr) | 2010-04-16 | 2017-10-06 | Biogen Ma Inc. | Protutijela anti-vla-4 |
| US9795658B2 (en) | 2010-04-20 | 2017-10-24 | Admedus Vaccines Pty Ltd | Expression system for modulating an immune response |
| DK2561077T3 (en) | 2010-04-23 | 2016-08-01 | Arrowhead Res Corp | Organic compositions for the treatment of beta-ENaC-related diseases |
| WO2011140151A1 (en) | 2010-05-04 | 2011-11-10 | Dyax Corp. | Antibodies against epidermal growth factor receptor (egfr) |
| EP2566512A1 (de) | 2010-05-04 | 2013-03-13 | Merrimack Pharmaceuticals, Inc. | Antikörper gegen den epidermalen wachstumsfaktorrezeptor (egfr) und ihre verwendung |
| US8450350B2 (en) | 2010-05-05 | 2013-05-28 | Infinity Pharmaceuticals, Inc. | Triazoles as inhibitors of fatty acid synthase |
| CA2798330A1 (en) | 2010-05-05 | 2011-11-10 | Infinity Pharmaceuticals, Inc. | Tetrazolones as inhibitors of fatty acid synthase |
| EP2566953B1 (de) | 2010-05-07 | 2019-01-02 | Greenlight Biosciences, Inc. | Verfahren zur flusssteuerung bei stoffwechselwegen druch enzymumpositionierung |
| ES2763815T3 (es) | 2010-05-11 | 2020-06-01 | Lantheus Medical Imaging Inc | Composiciones, métodos y sistemas para la síntesis y uso de agentes de formación de imágenes |
| EP2756857B1 (de) | 2010-05-19 | 2016-05-18 | Allergan, Inc. | Modulare Injektionsvorrichtung |
| WO2011160042A2 (en) | 2010-06-18 | 2011-12-22 | Makoto Life Sciences, Inc. | Prpk-tprkb modulators and uses thereof |
| AU2011272941B2 (en) | 2010-06-30 | 2014-05-29 | Compugen Ltd. | C10RF32 for the treatment of multiple sclerosis, rheumatoid arthritis and other autoimmune disorders |
| US9072713B2 (en) | 2010-07-01 | 2015-07-07 | The Texas A&M University System | Perlecan domain V protects, repairs and restores ischemic brain stroke injury and motor function |
| CN103119062A (zh) | 2010-07-16 | 2013-05-22 | 埃博灵克斯股份有限公司 | 修饰的单结构域抗原结合分子及其应用 |
| WO2012019168A2 (en) | 2010-08-06 | 2012-02-09 | Moderna Therapeutics, Inc. | Engineered nucleic acids and methods of use thereof |
| ES2711526T3 (es) | 2010-08-13 | 2019-05-06 | Aileron Therapeutics Inc | Macrociclos peptidomiméticos |
| WO2012022734A2 (en) | 2010-08-16 | 2012-02-23 | Medimmune Limited | Anti-icam-1 antibodies and methods of use |
| EP2611922A1 (de) | 2010-08-31 | 2013-07-10 | Greenlight Biosciences, Inc. | Verfahren zur flusssteuerung bei stoffwechselwegen durch proteasemanipulation |
| JP5856621B2 (ja) | 2010-09-15 | 2016-02-10 | ゾゲニクス インコーポレーティッド | 無針注射器および注射性能を最適化する同注射器の設計パラメータ |
| WO2012035518A1 (en) | 2010-09-17 | 2012-03-22 | Compugen Ltd. | Compositions and methods for treatment of drug resistant multiple myeloma |
| JP2013541528A (ja) | 2010-09-21 | 2013-11-14 | マサチューセッツ インスティテュート オブ テクノロジー | ヒト適応haポリペプチド、ワクチン、およびインフルエンザの処置 |
| WO2012040459A2 (en) | 2010-09-22 | 2012-03-29 | President And Fellows Of Harvard College | Beta-catenin targeting peptides and uses thereof |
| AU2011309689B2 (en) | 2010-09-28 | 2015-01-15 | Hadasit Medical Research Services & Development Ltd. | Compositions and methods for treatment of hematological malignancies |
| EP2625189B1 (de) | 2010-10-01 | 2018-06-27 | ModernaTX, Inc. | Manipulierte nukleinsäuren und anwendungsverfahren dafür |
| EP3828205A1 (de) | 2010-10-01 | 2021-06-02 | Oxford BioTherapeutics Ltd | Anti-ror1-antikörper |
| MX2013003599A (es) | 2010-10-01 | 2013-07-29 | Biogen Idec Inc | Interferon-beta para uso como monoterapia o en combinacion con otras terapias de cancer. |
| JP6029586B2 (ja) | 2010-10-04 | 2016-11-24 | マサチューセッツ インスティテュート オブ テクノロジー | 血球凝集素ポリペプチド、ならびにそれに関連する試薬および方法 |
| EP2625203A1 (de) | 2010-10-05 | 2013-08-14 | Novartis AG | Anti-il12rbeta1-antikörper und ihre verwendung zur behanldung von autoimmun- und entzündungserkrankungen |
| AU2011325865B2 (en) | 2010-11-03 | 2015-01-22 | Construct Medical Pty Ltd | Portable vacuum device |
| JP2013545749A (ja) | 2010-11-10 | 2013-12-26 | インフィニティー ファーマシューティカルズ, インコーポレイテッド | 複素環化合物及びその使用 |
| EP2643456B1 (de) | 2010-11-24 | 2016-05-11 | Novartis International Pharmaceutical Ltd. | Fusionsproteine mit n-acetylglucosaminyltransferase aktivitàt |
| US10208349B2 (en) | 2011-01-07 | 2019-02-19 | Ucb Biopharma Sprl | Lipocalin 2 as a biomarker for IL-17 inhibitor therapy efficacy |
| GB201100282D0 (en) | 2011-01-07 | 2011-02-23 | Ucb Pharma Sa | Biological methods |
| ES2637113T3 (es) | 2011-01-10 | 2017-10-10 | Infinity Pharmaceuticals, Inc. | Procedimientos para preparar isoquinolinonas y formas sólidas de isoquinolinonas |
| EP3378480A1 (de) | 2011-01-19 | 2018-09-26 | Topokine Therapeutics, Inc. | Verfahren und zusammensetzungen zur behandlung des stoffwechselsyndroms |
| US20120328549A1 (en) | 2011-01-24 | 2012-12-27 | Anterios, Inc. | Oil compositions |
| US20120328701A1 (en) | 2011-01-24 | 2012-12-27 | Anterios, Inc. | Nanoparticle compositions, formulations thereof, and uses therefor |
| BR112013018918A2 (pt) | 2011-01-24 | 2016-10-04 | Anterios Inc | composições de nanopartículas |
| US8627816B2 (en) | 2011-02-28 | 2014-01-14 | Intelliject, Inc. | Medicament delivery device for administration of opioid antagonists including formulations for naloxone |
| US9084849B2 (en) | 2011-01-26 | 2015-07-21 | Kaleo, Inc. | Medicament delivery devices for administration of a medicament within a prefilled syringe |
| US8939943B2 (en) | 2011-01-26 | 2015-01-27 | Kaleo, Inc. | Medicament delivery device for administration of opioid antagonists including formulations for naloxone |
| CN103889219A (zh) | 2011-03-03 | 2014-06-25 | 特萨斯制药有限责任公司 | 包含C16:1n7-棕榈油酸酯的组合物和方法 |
| WO2012119989A2 (en) | 2011-03-04 | 2012-09-13 | Oryzon Genomics, S.A. | Methods and antibodies for the diagnosis and treatment of cancer |
| GB201103836D0 (en) | 2011-03-07 | 2011-04-20 | Glaxosmithkline Biolog Sa | Conjugation process |
| US20130281932A1 (en) * | 2011-03-30 | 2013-10-24 | Ziv Harish | Palm-based injector actuation and safety surfaces |
| WO2012135615A2 (en) | 2011-03-30 | 2012-10-04 | Brown University | Enopeptins, uses thereof, and methods of synthesis thereto |
| AU2012236099A1 (en) | 2011-03-31 | 2013-10-03 | Moderna Therapeutics, Inc. | Delivery and formulation of engineered nucleic acids |
| NZ713461A (en) | 2011-04-15 | 2017-02-24 | Compugen Ltd | Polypeptides and polynucleotides, and uses thereof for treatment of immune related disorders and cancer |
| RU2013151159A (ru) * | 2011-04-19 | 2015-05-27 | ИНВИЗИДЕРМ, ЭлЭлСи | Способ создания веществ, перенасыщенных газом, устройство трансдермальной доставки и его использование |
| CN103533953A (zh) | 2011-05-17 | 2014-01-22 | 葛兰素史密丝克莱恩生物有限公司 | 针对肺炎链球菌的疫苗 |
| US8691231B2 (en) | 2011-06-03 | 2014-04-08 | Merrimack Pharmaceuticals, Inc. | Methods of treatment of tumors expressing predominantly high affinity EGFR ligands or tumors expressing predominantly low affinity EGFR ligands with monoclonal and oligoclonal anti-EGFR antibodies |
| BR112013031762A2 (pt) | 2011-06-16 | 2016-09-13 | Novartis Ag | proteínas solúveis para utilização como terapêuticos |
| CA2837651A1 (en) | 2011-06-21 | 2012-12-27 | Oncofactor Corporation | Compositions and methods for the therapy and diagnosis of cancer |
| CA2840224C (en) | 2011-06-22 | 2019-08-13 | The General Hospital Corporation | Treatment of proteinopathies |
| PL2726508T3 (pl) | 2011-06-28 | 2017-12-29 | Oxford Biotherapeutics Ltd | Przeciwciała przeciwko cyklazie ADP-rybozylowej 2 |
| US9428574B2 (en) | 2011-06-30 | 2016-08-30 | Compugen Ltd. | Polypeptides and uses thereof for treatment of autoimmune disorders and infection |
| JP6472999B2 (ja) | 2011-07-01 | 2019-02-20 | ノバルティス アーゲー | 代謝障害を治療するための方法 |
| AR088218A1 (es) | 2011-07-19 | 2014-05-21 | Infinity Pharmaceuticals Inc | Compuestos heterociclicos utiles como inhibidores de pi3k |
| HK1198443A1 (en) | 2011-07-19 | 2015-04-24 | 无限药品股份有限公司 | Heterocyclic compounds and uses thereof |
| SI2734510T1 (sl) | 2011-07-22 | 2019-05-31 | Massachusetts Institute Of Technology | Aktivatorji histon deacetilaz (HDACS) razreda I in njihove uporabe |
| EP2736928B1 (de) | 2011-07-28 | 2019-01-09 | i2 Pharmaceuticals, Inc. | Gegen erbb3-gerichtete sur-bindende proteine |
| MX2014002542A (es) | 2011-08-29 | 2014-07-09 | Infinity Pharmaceuticals Inc | Compuestos heterociclicos y usos de los mismos. |
| CA3185394A1 (en) | 2011-09-02 | 2013-03-07 | Arrowhead Pharmaceuticals, Inc. | Organic compositions to treat hsf1-related diseases |
| CN104093848A (zh) | 2011-09-09 | 2014-10-08 | 绿光生物科学公司 | 碳青霉烯(carbapenem)的无细胞制备 |
| CN108484449A (zh) | 2011-09-09 | 2018-09-04 | 蓝瑟斯医学影像公司 | 用于合成和使用显像剂的组合物、方法和系统 |
| WO2013039861A2 (en) | 2011-09-12 | 2013-03-21 | modeRNA Therapeutics | Engineered nucleic acids and methods of use thereof |
| WO2013043569A1 (en) | 2011-09-20 | 2013-03-28 | Vical Incorporated | Synergistic anti-tumor efficacy using alloantigen combination immunotherapy |
| MX353958B (es) | 2011-09-22 | 2018-02-07 | Amgen Inc | Proteinas de union al antigeno cd27l. |
| WO2013049332A1 (en) | 2011-09-29 | 2013-04-04 | Infinity Pharmaceuticals, Inc. | Inhibitors of monoacylglycerol lipase and methods of their use |
| DE19216461T1 (de) | 2011-10-03 | 2021-10-07 | Modernatx, Inc. | Modifizierte nukleoside, nukleotide und nukleinsäuren und verwendungen davon |
| RU2474437C1 (ru) * | 2011-10-13 | 2013-02-10 | Борис Владимирович Смоляров | Способ осуществления безыгольной инъекции |
| US20130096170A1 (en) | 2011-10-14 | 2013-04-18 | Hospira, Inc. | Methods of treating pediatric patients using dexmedetomidine |
| TW201806968A (zh) | 2011-10-18 | 2018-03-01 | 艾利倫治療公司 | 擬肽巨環化合物 |
| CN104039320B (zh) | 2011-11-09 | 2019-10-15 | 百时美施贵宝公司 | 血液恶性肿瘤用抗-cxcr4抗体的治疗 |
| TWI679212B (zh) | 2011-11-15 | 2019-12-11 | 美商安進股份有限公司 | 針對bcma之e3以及cd3的結合分子 |
| US9382239B2 (en) | 2011-11-17 | 2016-07-05 | Dana-Farber Cancer Institute, Inc. | Inhibitors of c-Jun-N-terminal kinase (JNK) |
| US8603028B2 (en) | 2011-11-18 | 2013-12-10 | Allergan, Inc. | Injection device having an angled tip portion |
| GB201119999D0 (en) | 2011-11-20 | 2012-01-04 | Glaxosmithkline Biolog Sa | Vaccine |
| GB201120000D0 (en) | 2011-11-20 | 2012-01-04 | Glaxosmithkline Biolog Sa | Vaccine |
| JP6539046B2 (ja) | 2011-12-16 | 2019-07-03 | ユニバーシティー オブ フロリダ リサーチ ファンデーション, インク. | 4’−デスフェリチオシン類似体の使用 |
| CA3018046A1 (en) | 2011-12-16 | 2013-06-20 | Moderna Therapeutics, Inc. | Modified nucleoside, nucleotide, and nucleic acid compositions |
| US8426471B1 (en) | 2011-12-19 | 2013-04-23 | Topokine Therapeutics, Inc. | Methods and compositions for reducing body fat and adipocytes |
| US9975956B2 (en) | 2011-12-22 | 2018-05-22 | I2 Pharmaceuticals, Inc. | Surrogate binding proteins which bind DR4 and/or DR5 |
| GB201122305D0 (en) | 2011-12-23 | 2012-02-01 | Biotica Tech Ltd | Novel compound |
| US8242158B1 (en) | 2012-01-04 | 2012-08-14 | Hospira, Inc. | Dexmedetomidine premix formulation |
| RU2645252C2 (ru) | 2012-01-05 | 2018-02-19 | Глюкос Финланд Ой | Клетка нитчатых грибов с дефицитом протеаз и способы ее применения |
| EP2802658A2 (de) | 2012-01-09 | 2014-11-19 | Novartis AG | Rnai-wirkstoffe zur behandlung von erkrankungen im zusammenhang mit beta-catenin |
| US10130800B2 (en) | 2012-01-27 | 2018-11-20 | Invisiderm, Llc | Method of producing substances with supersaturated gas, transdermal delivery device thereof, and uses thereof |
| CA2848985A1 (en) | 2012-02-01 | 2013-08-08 | Compugen Ltd. | C10rf32 antibodies, and uses thereof for treatment of cancer |
| HUE033704T2 (en) | 2012-02-13 | 2017-12-28 | Bristol Myers Squibb Co | Enediyne compounds, conjugates thereof, and uses and methods therefor |
| BR112014020103A2 (pt) | 2012-02-15 | 2018-10-09 | Aileron Therapeutics, Inc. | macrociclos peptidomiméticos |
| EP2819688A4 (de) | 2012-02-15 | 2015-10-28 | Aileron Therapeutics Inc | Triazol- und thioethervernetzte peptidomimetische makrozyklen |
| WO2013142796A2 (en) | 2012-03-23 | 2013-09-26 | Bristol-Myers Squibb Company | Methods of treatments using ctla4 antibodies |
| EP2830659A1 (de) | 2012-03-27 | 2015-02-04 | Novartis AG | Behandlung von fibrose |
| CA2868459A1 (en) | 2012-03-27 | 2013-10-03 | Amicus Therapeutics, Inc. | Polymorphs of (3r,4r,5s)-5-(difluoromethyl)piperidine-3,4-diol fumarate and uses thereof |
| US9902985B2 (en) | 2012-04-06 | 2018-02-27 | President And Fellows Of Harvard College | Chemoenzymatic methods for synthesizing moenomycin analogs |
| US9273084B2 (en) | 2012-04-06 | 2016-03-01 | President And Fellows Of Harvard College | Moenomycin analogs, methods of synthesis, and uses thereof |
| EP2850091A1 (de) | 2012-04-06 | 2015-03-25 | President and Fellows of Harvard College | Verfahren und verbindungen zur identifizierung von glycosyltransferaseinhibitoren |
| US8940742B2 (en) | 2012-04-10 | 2015-01-27 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| US10130714B2 (en) | 2012-04-14 | 2018-11-20 | Academia Sinica | Enhanced anti-influenza agents conjugated with anti-inflammatory activity |
| WO2013158273A1 (en) | 2012-04-20 | 2013-10-24 | Abbvie Inc. | Methods to modulate c-terminal lysine variant distribution |
| US9597385B2 (en) | 2012-04-23 | 2017-03-21 | Allertein Therapeutics, Llc | Nanoparticles for treatment of allergy |
| US20130304017A1 (en) * | 2012-05-09 | 2013-11-14 | Bioject, Inc. | Peformance of needle-free injection according to known relationships |
| WO2013167153A1 (en) | 2012-05-09 | 2013-11-14 | Ganymed Pharmaceuticals Ag | Antibodies useful in cancer diagnosis |
| US9969794B2 (en) | 2012-05-10 | 2018-05-15 | Visterra, Inc. | HA binding agents |
| SG11201406769RA (en) | 2012-05-10 | 2014-11-27 | Massachusetts Inst Technology | Agents for influenza neutralization |
| US9399672B2 (en) | 2012-05-17 | 2016-07-26 | The United States Of America, As Represented By The Secretary Department Of Health And Human Services | Hepatitis C virus neutralizing antibody |
| AR091069A1 (es) | 2012-05-18 | 2014-12-30 | Amgen Inc | Proteinas de union a antigeno dirigidas contra el receptor st2 |
| US9522235B2 (en) | 2012-05-22 | 2016-12-20 | Kaleo, Inc. | Devices and methods for delivering medicaments from a multi-chamber container |
| EP2852610B1 (de) | 2012-05-23 | 2018-07-11 | Glykos Finland Oy | Herstellung von fucosylierten glykoproteinen |
| WO2013176754A1 (en) | 2012-05-24 | 2013-11-28 | Abbvie Inc. | Novel purification of antibodies using hydrophobic interaction chromatography |
| EP3530283A1 (de) | 2012-06-05 | 2019-08-28 | The Australian National University | Impfung mit interleukin-4-antagonisten |
| GB201210082D0 (en) * | 2012-06-07 | 2012-07-25 | Consort Medical Plc | Improved syringe |
| US9890215B2 (en) | 2012-06-22 | 2018-02-13 | King's College London | Vista modulators for diagnosis and treatment of cancer |
| JP6285923B2 (ja) | 2012-06-22 | 2018-02-28 | トラスティーズ・オブ・ダートマス・カレッジ | 新規VISTA−Igコンストラクト及び自己免疫、アレルギー性及び炎症性障害の処置のためのVISTA−Igの使用 |
| UY34887A (es) | 2012-07-02 | 2013-12-31 | Bristol Myers Squibb Company Una Corporacion Del Estado De Delaware | Optimización de anticuerpos que se fijan al gen de activación de linfocitos 3 (lag-3) y sus usos |
| USD695892S1 (en) | 2012-07-23 | 2013-12-17 | Pharmajet, Inc. | Needle-free syringe |
| AU2013301312A1 (en) | 2012-08-06 | 2015-03-19 | Glaxosmithkline Biologicals S.A. | Method for eliciting in infants an immune response against RSV and B. pertussis |
| US20140037680A1 (en) | 2012-08-06 | 2014-02-06 | Glaxosmithkline Biologicals, S.A. | Novel method |
| AU2013203000B9 (en) | 2012-08-10 | 2017-02-02 | Lantheus Medical Imaging, Inc. | Compositions, methods, and systems for the synthesis and use of imaging agents |
| US10309430B2 (en) * | 2012-08-10 | 2019-06-04 | Confluent Surgical, Inc. | Pneumatic actuation assembly |
| WO2014028463A1 (en) | 2012-08-14 | 2014-02-20 | Allergan, Inc. | Syringe for mixing and dispensing adipose tissue |
| WO2014031498A1 (en) | 2012-08-18 | 2014-02-27 | Academia Sinica | Cell-permeable probes for identification and imaging of sialidases |
| US9381244B2 (en) | 2012-09-07 | 2016-07-05 | King's College London | VISTA modulators for diagnosis and treatment of cancer |
| WO2014052647A2 (en) | 2012-09-26 | 2014-04-03 | President And Fellows Of Harvard College | Proline-locked stapled peptides and uses thereof |
| US20150225471A1 (en) | 2012-10-01 | 2015-08-13 | President And Fellows Of Harvard College | Stabilized polypeptide insulin receptor modulators |
| RS58700B1 (sr) | 2012-10-12 | 2019-06-28 | Broad Inst Inc | Gsk3 inhibitori i postupci njihove primene |
| EP2909194A1 (de) | 2012-10-18 | 2015-08-26 | Dana-Farber Cancer Institute, Inc. | Hemmer der cyclinabhängigen kinase 7 (cdk7) |
| WO2014063054A1 (en) | 2012-10-19 | 2014-04-24 | Dana-Farber Cancer Institute, Inc. | Bone marrow on x chromosome kinase (bmx) inhibitors and uses thereof |
| USRE48175E1 (en) | 2012-10-19 | 2020-08-25 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged small molecules as inducers of protein degradation |
| US9675754B2 (en) | 2012-10-24 | 2017-06-13 | Nuance Designs, LLC | Autoinjector |
| WO2014065822A1 (en) | 2012-10-26 | 2014-05-01 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Hepatitis c virus neutralizing epitopes, antibodies, and methods |
| AU2013337717B2 (en) | 2012-11-01 | 2018-10-25 | Infinity Pharmaceuticals, Inc. | Treatment of cancers using PI3 kinase isoform modulators |
| WO2014071241A1 (en) | 2012-11-01 | 2014-05-08 | Aileron Therapeutics, Inc. | Disubstituted amino acids and methods of preparation and use thereof |
| EP2917203B1 (de) | 2012-11-02 | 2019-04-03 | Dana-Farber Cancer Institute, Inc. | Verfahren zur identifizierung von myc inhibitoren |
| CN104936632B (zh) * | 2012-11-13 | 2017-08-04 | 康斯特鲁科特医疗有限公司 | 便携式抽吸装置 |
| WO2014081941A1 (en) | 2012-11-21 | 2014-05-30 | Topokine Therapeutics, Inc. | Methods and compositions for locally increasing body fat |
| PL2922554T3 (pl) | 2012-11-26 | 2022-06-20 | Modernatx, Inc. | Na zmodyfikowany na końcach |
| JP6408478B2 (ja) | 2012-11-26 | 2018-10-17 | プレジデント アンド フェローズ オブ ハーバード カレッジ | トリオキサカルシン、トリオキサカルシン−抗体複合体、およびその用途 |
| AU2013359419B2 (en) | 2012-12-10 | 2018-03-15 | Biogen Ma Inc. | Anti-blood dendritic cell antigen 2 antibodies and uses thereof |
| USD690416S1 (en) | 2012-12-12 | 2013-09-24 | Pharmajet, Inc. | Needle-free syringe |
| PL2935320T3 (pl) | 2012-12-18 | 2020-03-31 | Novartis Ag | Stabilizowane polipeptydy insulinopodobnego czynnika wzrostu |
| CA2899363A1 (en) | 2012-12-21 | 2014-06-26 | Epizyme, Inc. | Prmt5 inhibitors and uses thereof |
| CA2894126A1 (en) | 2012-12-21 | 2014-06-26 | Epizyme, Inc. | Prmt5 inhibitors and uses thereof |
| US9745291B2 (en) | 2012-12-21 | 2017-08-29 | Epizyme, Inc. | PRMT5 inhibitors containing a dihydro- or tetrahydroisoquinoline and uses thereof |
| DK2935222T3 (en) | 2012-12-21 | 2019-01-07 | Epizyme Inc | PRMT5 INHIBITORS AND APPLICATIONS THEREOF |
| WO2014106096A1 (en) | 2012-12-27 | 2014-07-03 | Kaleo, Inc. | Devices, systems and methods for locating and interacting with medicament delivery systems |
| AU2013374345A1 (en) | 2013-01-17 | 2015-08-06 | Moderna Therapeutics, Inc. | Signal-sensor polynucleotides for the alteration of cellular phenotypes |
| EP2948478B1 (de) | 2013-01-25 | 2019-04-03 | Amgen Inc. | Antikörper gegen cdh19 für melanome |
| JO3519B1 (ar) | 2013-01-25 | 2020-07-05 | Amgen Inc | تركيبات أجسام مضادة لأجل cdh19 و cd3 |
| US11576958B2 (en) | 2013-02-07 | 2023-02-14 | Children's Medical Center Corporation | Protein antigens that provide protection against pneumococcal colonization and/or disease |
| US20140335504A1 (en) | 2013-02-07 | 2014-11-13 | Massachusetts Institute Of Technology | Human adaptation of h5 influenza |
| LT2953969T (lt) | 2013-02-08 | 2019-12-10 | Novartis Ag | Anti-il-17a antikūnai ir jų panaudojimas autoimuninių ir uždegiminių ligų gydymui |
| LT2956173T (lt) | 2013-02-14 | 2017-06-26 | Bristol-Myers Squibb Company | Tubulizino junginiai, gavimo ir panaudojimo būdai |
| CA2902068C (en) | 2013-02-28 | 2023-10-03 | Caprion Proteomics Inc. | Tuberculosis biomarkers and uses thereof |
| EP3391898A3 (de) | 2013-03-13 | 2019-02-13 | President and Fellows of Harvard College | Gestapelte und geheftete polypeptide und verwendungen davon |
| EP2968391A1 (de) | 2013-03-13 | 2016-01-20 | Moderna Therapeutics, Inc. | Langlebige polynukleotidmoleküle |
| BR112015017307A2 (pt) | 2013-03-14 | 2017-11-21 | Abbvie Inc | composições de espécies com baixa acidez e métodos para produção e uso das mesmas |
| CA2899449A1 (en) | 2013-03-14 | 2014-10-02 | Abbvie Inc. | Low acidic species compositions and methods for producing the same using displacement chromatography |
| US9580486B2 (en) | 2013-03-14 | 2017-02-28 | Amgen Inc. | Interleukin-2 muteins for the expansion of T-regulatory cells |
| WO2014144466A1 (en) | 2013-03-15 | 2014-09-18 | Biogen Idec Ma Inc. | Anti-alpha v beta 6 antibodies and uses thereof |
| US9308236B2 (en) | 2013-03-15 | 2016-04-12 | Bristol-Myers Squibb Company | Macrocyclic inhibitors of the PD-1/PD-L1 and CD80(B7-1)/PD-L1 protein/protein interactions |
| PL2970449T3 (pl) | 2013-03-15 | 2020-04-30 | Amgen Research (Munich) Gmbh | Jednołańcuchowe cząsteczki wiążące zawierające N-końcowy ABP |
| WO2014151386A1 (en) | 2013-03-15 | 2014-09-25 | Infinity Pharmaceuticals, Inc. | Salts and solid forms of isoquinolinones and composition comprising and methods of using the same |
| WO2014143739A2 (en) | 2013-03-15 | 2014-09-18 | Biogen Idec Ma Inc. | Anti-alpha v beta 6 antibodies and uses thereof |
| EP2970446A1 (de) | 2013-03-15 | 2016-01-20 | Amgen Research (Munich) GmbH | Antikörperkonstrukte für influenza m2 und cd3 |
| JP2016515120A (ja) | 2013-03-15 | 2016-05-26 | バイオジェン・エムエイ・インコーポレイテッドBiogen MA Inc. | 抗アルファvベータ5抗体を用いた急性腎損傷の治療及び予防 |
| KR102233251B1 (ko) | 2013-04-03 | 2021-03-26 | 엔-폴드 엘엘씨 | 신규 나노입자 조성물 |
| JP6437523B2 (ja) | 2013-04-04 | 2018-12-12 | プレジデント アンド フェローズ オブ ハーバード カレッジ | マクロライドならびにそれらの調製および使用の方法 |
| WO2014169073A1 (en) | 2013-04-09 | 2014-10-16 | Massachusetts Institute Of Technology | Drug delivery polymer and uses thereof |
| WO2014179464A1 (en) | 2013-04-30 | 2014-11-06 | Massachusetts Institute Of Technology | Human adaptation of h3 influenza |
| WO2014179562A1 (en) | 2013-05-01 | 2014-11-06 | Massachusetts Institute Of Technology | 1,3,5-triazinane-2,4,6-trione derivatives and uses thereof |
| NO2753788T3 (de) | 2013-05-10 | 2018-06-16 | ||
| WO2014186504A1 (en) | 2013-05-15 | 2014-11-20 | Topokine Therapeutics, Inc. | Methods and compositions for topical delivery of prostaglandins to subcutaneous fat |
| US20140350516A1 (en) | 2013-05-23 | 2014-11-27 | Allergan, Inc. | Mechanical syringe accessory |
| US20140350518A1 (en) | 2013-05-23 | 2014-11-27 | Allergan, Inc. | Syringe extrusion accessory |
| HUE056580T2 (hu) | 2013-05-30 | 2022-02-28 | Kiniksa Pharmaceuticals Ltd | Onkosztatin-M-receptor antigénjét kötõ fehérjék |
| CA2914284A1 (en) | 2013-05-30 | 2014-12-04 | Infinity Pharmaceuticals, Inc. | Treatment of cancers using pi3 kinase isoform modulators |
| US10526375B2 (en) | 2013-06-05 | 2020-01-07 | Massachusetts Institute Of Technology | Human Adaptation of H7 HA |
| CA2914815A1 (en) | 2013-06-11 | 2014-12-18 | Kala Pharmaceuticals, Inc. | Urea derivatives and uses thereof |
| JP2016523241A (ja) | 2013-06-14 | 2016-08-08 | プレジデント アンド フェローズ オブ ハーバード カレッジ | 安定化されたポリペプチドインスリン受容体調節剤 |
| ES2909438T3 (es) | 2013-06-26 | 2022-05-06 | Helmholtz Zentrum Muenchen Deutsches Forschungszentrum Gesundheit & Umwelt Gmbh | Enantiómero (S) de mepacina como inhibidor de paracaspasa (MALT1) para tratar cáncer |
| EP3013365B1 (de) | 2013-06-26 | 2019-06-05 | Academia Sinica | Rm2-antigene und verwendung davon |
| EP3013347B1 (de) | 2013-06-27 | 2019-12-11 | Academia Sinica | Glykankonjugate und verwendung davon |
| US9975896B2 (en) | 2013-07-25 | 2018-05-22 | Dana-Farber Cancer Institute, Inc. | Inhibitors of transcription factors and uses thereof |
| AU2014306074B2 (en) | 2013-08-05 | 2018-08-30 | Greenlight Biosciences, Inc. | Engineered proteins with a protease cleavage site |
| MX2016001695A (es) | 2013-08-05 | 2016-05-02 | Glaxosmithkline Biolog Sa | Composiciones inmunogenas de combinacion. |
| TWI532511B (zh) * | 2013-08-16 | 2016-05-11 | 京華堂實業股份有限公司 | 無針型注射系統及其注射方法 |
| JP6486368B2 (ja) | 2013-09-06 | 2019-03-20 | アカデミア シニカAcademia Sinica | 改変されたグリコシル基を含む糖脂質を用いたヒトiNKT細胞の活性化 |
| AR097648A1 (es) | 2013-09-13 | 2016-04-06 | Amgen Inc | Combinación de factores epigenéticos y compuestos biespecíficos que tienen como diana cd33 y cd3 en el tratamiento de leucemia mieloide |
| WO2015050959A1 (en) | 2013-10-01 | 2015-04-09 | Yale University | Anti-kit antibodies and methods of use thereof |
| US20160271265A1 (en) | 2013-10-02 | 2016-09-22 | Novartis Ag | Insulin-like growth factor mimetics for use in therapy |
| WO2015051234A2 (en) | 2013-10-04 | 2015-04-09 | Biogen Idec Ma Inc. | Tweak antagonists for treating lupus nephritis and muscle atrophy |
| US9751888B2 (en) | 2013-10-04 | 2017-09-05 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| PT3052485T (pt) | 2013-10-04 | 2021-10-22 | Infinity Pharmaceuticals Inc | Compostos heterocíclicos e suas utilizações |
| CN105745224B (zh) | 2013-10-11 | 2019-11-05 | 牛津生物疗法有限公司 | 用于治疗癌症的针对ly75的偶联抗体 |
| WO2015057894A1 (en) | 2013-10-15 | 2015-04-23 | Massachusetts Institute Of Technology | Methods for treating polycystic kidney disease and polycystic liver disease |
| US9902986B2 (en) | 2013-10-16 | 2018-02-27 | Massachusetts Institute Of Technology | Enterobactin conjugates and uses thereof |
| WO2015057939A1 (en) | 2013-10-18 | 2015-04-23 | Biogen Idec Ma Inc. | Anti-s1p4 antibodies and uses thereof |
| EP3057956B1 (de) | 2013-10-18 | 2021-05-05 | Dana-Farber Cancer Institute, Inc. | Polycyclische inhibitoren der cyclin-dependent-kinase 7 (cdk7) |
| WO2015058126A1 (en) | 2013-10-18 | 2015-04-23 | Syros Pharmaceuticals, Inc. | Heteroaromatic compounds useful for the treatment of prolferative diseases |
| US20160244452A1 (en) | 2013-10-21 | 2016-08-25 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| US9458169B2 (en) | 2013-11-01 | 2016-10-04 | Kala Pharmaceuticals, Inc. | Crystalline forms of therapeutic compounds and uses thereof |
| UY35874A (es) | 2013-12-12 | 2015-07-31 | Novartis Ag | Un proceso para la preparación de una composición de proteínas pegiladas |
| CN106029697B (zh) | 2013-12-20 | 2021-06-04 | 英特维特国际股份有限公司 | 具有经修饰的ch2-ch3序列的犬抗体 |
| TWI670283B (zh) | 2013-12-23 | 2019-09-01 | 美商建南德克公司 | 抗體及使用方法 |
| AU2014369834B2 (en) | 2013-12-24 | 2018-12-20 | President And Fellows Of Harvard College | Cortistatin analogues and syntheses and uses thereof |
| WO2015097536A2 (en) | 2013-12-24 | 2015-07-02 | Janssen Pharmaceutical Nv | Anti-vista antibodies and fragments |
| US11014987B2 (en) | 2013-12-24 | 2021-05-25 | Janssen Pharmaceutics Nv | Anti-vista antibodies and fragments, uses thereof, and methods of identifying same |
| AU2015206370A1 (en) | 2014-01-16 | 2016-07-07 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| US10150818B2 (en) | 2014-01-16 | 2018-12-11 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| US10793571B2 (en) | 2014-01-31 | 2020-10-06 | Dana-Farber Cancer Institute, Inc. | Uses of diazepane derivatives |
| US9598438B2 (en) | 2014-02-12 | 2017-03-21 | Amicus Therapeutics, Inc. | Sugar derivatives comprising sulfur-containing moieties and methods of making same and methods of using the same for the treatment of MPS IIIC |
| MX2016011517A (es) | 2014-03-07 | 2017-04-13 | Univ Arizona | Peptidos de crmp2 no narcoticos que seleccionan como diana canales de sodio para el dolor cronico. |
| MX382033B (es) | 2014-03-19 | 2025-03-13 | Infinity Pharmaceuticals Inc | Compuestos heterocíclicos para utilizarlos en el tratamiento de trastornos mediados por pi3k-gamma. |
| CN106415244B (zh) | 2014-03-27 | 2020-04-24 | 中央研究院 | 反应性标记化合物及其用途 |
| DK3122375T3 (da) | 2014-03-28 | 2021-05-25 | Univ Washington Through Its Center For Commercialization | Vacciner mod bryst- og ovariecancer |
| US9675627B2 (en) | 2014-04-14 | 2017-06-13 | Amicus Therapeutics, Inc. | Dosing regimens for treating and/or preventing cerebral amyloidoses |
| US9862688B2 (en) | 2014-04-23 | 2018-01-09 | Dana-Farber Cancer Institute, Inc. | Hydrophobically tagged janus kinase inhibitors and uses thereof |
| WO2015164614A1 (en) | 2014-04-23 | 2015-10-29 | Dana-Farber Cancer Institute, Inc. | Janus kinase inhibitors and uses thereof |
| WO2015168079A1 (en) | 2014-04-29 | 2015-11-05 | Infinity Pharmaceuticals, Inc. | Pyrimidine or pyridine derivatives useful as pi3k inhibitors |
| WO2015168380A1 (en) | 2014-04-30 | 2015-11-05 | Massachusetts Institute Of Technology | Siderophore-based immunization against gram-negative bacteria |
| US10029048B2 (en) | 2014-05-13 | 2018-07-24 | Allergan, Inc. | High force injection devices |
| CA2949535C (en) | 2014-05-21 | 2023-09-12 | President And Fellows Of Harvard College | Ras inhibitory peptides and uses thereof |
| CA2950415A1 (en) | 2014-05-27 | 2015-12-03 | Academia Sinica | Anti-cd20 glycoantibodies and uses thereof |
| US10118969B2 (en) | 2014-05-27 | 2018-11-06 | Academia Sinica | Compositions and methods relating to universal glycoforms for enhanced antibody efficacy |
| CN106661099A (zh) | 2014-05-27 | 2017-05-10 | 中央研究院 | 抗her2醣抗体及其用途 |
| JP7093612B2 (ja) | 2014-05-27 | 2022-06-30 | アカデミア シニカ | Bacteroides由来のフコシダーゼおよびそれを使用する方法 |
| WO2015184001A1 (en) | 2014-05-28 | 2015-12-03 | Academia Sinica | Anti-tnf-alpha glycoantibodies and uses thereof |
| JP6612017B2 (ja) * | 2014-06-03 | 2019-11-27 | 株式会社ダイセル | 無針注射器による注射システム |
| PE20170441A1 (es) | 2014-06-06 | 2017-04-26 | Bristol Myers Squibb Co | Anticuerpos contra el receptor del factor de necrosis tumoral inducido por glucocorticoides (gitr) y sus usos |
| US11123426B2 (en) | 2014-06-11 | 2021-09-21 | The Trustees Of Dartmouth College | Use of vista agonists and antagonists to suppress or enhance humoral immunity |
| MX384992B (es) | 2014-06-13 | 2025-03-14 | Glaxosmithkline Biologicals Sa | Combinaciones inmunógenas. |
| EP3778584A1 (de) | 2014-06-19 | 2021-02-17 | ARIAD Pharmaceuticals, Inc. | Herstellungsverfahren von 2-chloro-4-heteroaryl-pyrimine derivaten |
| EP2959931A1 (de) | 2014-06-24 | 2015-12-30 | LTS LOHMANN Therapie-Systeme AG | Nadellose Injektionsvorrichtung |
| EP3161001A2 (de) | 2014-06-25 | 2017-05-03 | Novartis AG | Il17-a spezifische antikörper die mit hyaluronan bindenden peptid-marker fusioniert sind |
| WO2015200425A1 (en) | 2014-06-27 | 2015-12-30 | Topokine Therapeutics, Inc. | Topical dosage regimen |
| US9840479B2 (en) | 2014-07-02 | 2017-12-12 | Massachusetts Institute Of Technology | Polyamine-fatty acid derived lipidoids and uses thereof |
| WO2016004197A1 (en) | 2014-07-03 | 2016-01-07 | Abbvie Inc. | Methods for modulating protein glycosylation profiles of recombinant protein therapeutics using cobalt |
| WO2016007764A1 (en) | 2014-07-09 | 2016-01-14 | Abbvie Inc. | Methods for modulating the glycosylation profile of recombinant proteins using non-commonly used sugars |
| US9517307B2 (en) | 2014-07-18 | 2016-12-13 | Kaleo, Inc. | Devices and methods for delivering opioid antagonists including formulations for naloxone |
| US10759836B2 (en) | 2014-07-18 | 2020-09-01 | University Of Washington | Cancer vaccine compositions and methods of use thereof |
| MX384418B (es) | 2014-07-31 | 2025-03-14 | Amgen Res Munich Gmbh | Constructo de anticuerpo de cadena individual biespecífico con distribución mejorada de tejido. |
| UY36245A (es) | 2014-07-31 | 2016-01-29 | Amgen Res Munich Gmbh | Constructos de anticuerpos para cdh19 y cd3 |
| EP3174901B1 (de) | 2014-07-31 | 2019-06-26 | Amgen Research (Munich) GmbH | Optimierte artenübergreifende spezifische bispezifische einkettige antikörperkonstrukte |
| EP2979714A1 (de) | 2014-08-02 | 2016-02-03 | LTS LOHMANN Therapie-Systeme AG | Nadellose Injektionsvorrichtung aufweisend eine Membran |
| EP4108256A1 (de) | 2014-08-11 | 2022-12-28 | University of Massachusetts | Anti-ospa-antikörper und verfahren zur verwendung |
| US10736966B2 (en) | 2014-08-12 | 2020-08-11 | Massachusetts Institute Of Technology | Brush-poly (glycoamidoamine)-lipids and uses thereof |
| JO3663B1 (ar) | 2014-08-19 | 2020-08-27 | Merck Sharp & Dohme | الأجسام المضادة لمضاد lag3 وأجزاء ربط الأنتيجين |
| MD4733C1 (ro) | 2014-08-19 | 2021-07-31 | Merck Sharp & Dohme Corp | Anticorpi anti-TIGIT |
| EP3185885B1 (de) | 2014-08-26 | 2020-08-19 | Compugen Ltd. | Polypeptide und verwendungen davon als arzneimittel zur behandlung von autoimmunerkrankungen |
| EP2993174A1 (de) | 2014-09-08 | 2016-03-09 | Helmholtz Zentrum München Deutsches Forschungszentrum für Gesundheit und Umwelt GmbH | Pyrazolopyridinderivate und deren Verwendung in der Therapie |
| TWI745275B (zh) | 2014-09-08 | 2021-11-11 | 中央研究院 | 使用醣脂激活人類iNKT細胞 |
| AU2015320545C1 (en) | 2014-09-24 | 2020-05-14 | Aileron Therapeutics, Inc. | Peptidomimetic macrocycles and formulations thereof |
| EP3197478A4 (de) | 2014-09-24 | 2018-05-30 | Aileron Therapeutics, Inc. | Peptidomimetische makrozyklen und verwendungen davon |
| US10226585B2 (en) | 2014-10-01 | 2019-03-12 | Allergan, Inc. | Devices for injection and dosing |
| JP2017531642A (ja) | 2014-10-03 | 2017-10-26 | マサチューセッツ インスティテュート オブ テクノロジー | エボラ糖タンパク質に結合する抗体およびその使用 |
| WO2016054491A1 (en) | 2014-10-03 | 2016-04-07 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| US9732119B2 (en) | 2014-10-10 | 2017-08-15 | Bristol-Myers Squibb Company | Immunomodulators |
| UA119794C2 (uk) | 2014-10-21 | 2019-08-12 | Аріад Фармасьютикалз, Інк. | Кристалічна форма 5-хлор-n4-[2-(диметилфосфорил)феніл]-n2-{2-метокси-4-[4-(4-метилпіперазин-1-іл)піперидин-1-іл]}піримідин-2,4-діаміну |
| EP3011988A1 (de) | 2014-10-22 | 2016-04-27 | LTS LOHMANN Therapie-Systeme AG | Nadellose Injektionsvorrichtung aufweisend ein Gel und eine Membran |
| EP3212222A2 (de) | 2014-10-28 | 2017-09-06 | INSERM - Institut National de la Santé et de la Recherche Médicale | Zusammensetzungen und verfahren für antigenspezifische toleranz |
| WO2016077125A1 (en) | 2014-11-10 | 2016-05-19 | Moderna Therapeutics, Inc. | Alternative nucleic acid molecules containing reduced uracil content and uses thereof |
| WO2016075137A1 (en) | 2014-11-10 | 2016-05-19 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Spiropyrazine derivatives as inhibitors of non-apoptotic regulated cell-death |
| US10077287B2 (en) | 2014-11-10 | 2018-09-18 | Bristol-Myers Squibb Company | Tubulysin analogs and methods of making and use |
| US9856292B2 (en) | 2014-11-14 | 2018-01-02 | Bristol-Myers Squibb Company | Immunomodulators |
| AU2015349878A1 (en) | 2014-11-21 | 2017-05-25 | Bristol-Myers Squibb Company | Antibodies against CD73 and uses thereof |
| JP6668345B2 (ja) | 2014-11-21 | 2020-03-18 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | 修飾された重鎖定常領域を含む抗体 |
| US10253045B2 (en) | 2014-11-26 | 2019-04-09 | Kala Pharmaceuticals, Inc. | Crystalline forms of a therapeutic compound and uses thereof |
| WO2016087514A1 (en) | 2014-12-02 | 2016-06-09 | Cemm - Forschungszentrum Für Molekulare Medizin Gmbh | Anti-mutant calreticulin antibodies and their use in the diagnosis and therapy of myeloid malignancies |
| CA2969730A1 (en) | 2014-12-05 | 2016-06-09 | Immunext, Inc. | Identification of vsig8 as the putative vista receptor and its use thereof to produce vista/vsig8 modulators |
| EP3230272B1 (de) | 2014-12-10 | 2020-08-19 | Kala Pharmaceuticals, Inc. | 1-amino-triazol(1,5-a)pyridin-substituiertes harnstoffderivat und verwendungen davon |
| US9861680B2 (en) | 2014-12-18 | 2018-01-09 | Bristol-Myers Squibb Company | Immunomodulators |
| WO2016097865A1 (en) | 2014-12-19 | 2016-06-23 | Regenesance B.V. | Antibodies that bind human c6 and uses thereof |
| JP6180663B2 (ja) | 2014-12-23 | 2017-08-16 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Tigitに対する抗体 |
| EP3237066B1 (de) | 2014-12-23 | 2020-10-21 | Sloan-Kettering Institute for Cancer Research | Polymorph von granaticin b |
| JP6854762B2 (ja) | 2014-12-23 | 2021-04-07 | ダナ−ファーバー キャンサー インスティテュート, インコーポレイテッド | サイクリン依存性キナーゼ7(cdk7)の阻害剤 |
| ES2924394T3 (es) | 2015-01-09 | 2022-10-06 | Adalta Ltd | Moléculas de unión a CXCR4 |
| BR112017014937A2 (pt) | 2015-01-14 | 2018-03-13 | Bristol-Myers Squibb Company | dímeros de benzodiazepina ligados em ponte a heteroarileno, conjugados dos mesmos, e métodos de preparação e uso |
| US10495645B2 (en) | 2015-01-16 | 2019-12-03 | Academia Sinica | Cancer markers and methods of use thereof |
| US9975965B2 (en) | 2015-01-16 | 2018-05-22 | Academia Sinica | Compositions and methods for treatment and detection of cancers |
| JP6779887B2 (ja) | 2015-01-24 | 2020-11-04 | アカデミア シニカAcademia Sinica | 新規なグリカンコンジュゲートおよびその使用方法 |
| US20160222060A1 (en) | 2015-02-04 | 2016-08-04 | Bristol-Myers Squibb Company | Immunomodulators |
| US10983128B2 (en) | 2015-02-05 | 2021-04-20 | Bristol-Myers Squibb Company | CXCL11 and SMICA as predictive biomarkers for efficacy of anti-CTLA4 immunotherapy |
| WO2016144773A1 (en) | 2015-03-06 | 2016-09-15 | Abbvie Inc. | Arabinosylated glycoproteins |
| WO2016145085A2 (en) | 2015-03-09 | 2016-09-15 | Celldex Therapeutics, Inc. | Cd27 agonists |
| BR112017019272A2 (pt) | 2015-03-10 | 2018-05-02 | Allergan Pharmaceuticals Holdings Ireland Unlimited Company | injetor de múltiplas agulhas |
| US9809625B2 (en) | 2015-03-18 | 2017-11-07 | Bristol-Myers Squibb Company | Immunomodulators |
| WO2016154058A1 (en) | 2015-03-20 | 2016-09-29 | Aileron Therapeutics, Inc. | Peptidomimetic macrocycles and uses thereof |
| WO2016154010A1 (en) | 2015-03-20 | 2016-09-29 | Makidon Paul | Immunogenic compositions for use in vaccination against bordetella |
| WO2016154427A2 (en) | 2015-03-24 | 2016-09-29 | Kaleo, Inc. | Devices and methods for delivering a lyophilized medicament |
| USRE50776E1 (en) | 2015-03-27 | 2026-02-03 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| BR112017020690A2 (pt) | 2015-03-30 | 2018-06-26 | Greenlight Biosciences Inc | produção livre de células de ácido ribonucleico |
| CA2980087A1 (en) | 2015-04-02 | 2016-10-06 | Intervet International B.V. | Antibodies to canine interleukin-4 receptor alpha |
| WO2016162405A1 (en) | 2015-04-10 | 2016-10-13 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Pyrone derivatives for use as antiviral agents |
| CR20170510A (es) | 2015-04-10 | 2018-02-26 | Amgen Inc | Muteínas de interuquina 2 para la expansión de células t regulatorias |
| CN107750255B (zh) | 2015-04-17 | 2022-08-30 | 安进研发(慕尼黑)股份有限公司 | 用于cdh3和cd3的双特异性抗体构建体 |
| EP3288557A4 (de) | 2015-04-27 | 2018-11-07 | University of Florida Research Foundation, Inc. | Metabolisch programmierte metallchelatoren und verwendungen davon |
| TW201702271A (zh) | 2015-04-30 | 2017-01-16 | 哈佛大學校長及研究員協會 | 治療代謝病症之抗-ap2抗體及抗原結合劑 |
| WO2016178591A2 (en) | 2015-05-05 | 2016-11-10 | Gene Predit, Sa | Genetic markers and treatment of male obesity |
| CN115043943A (zh) | 2015-05-15 | 2022-09-13 | 综合医院公司 | 拮抗性抗肿瘤坏死因子受体超家族抗体 |
| EP3297676A2 (de) | 2015-05-22 | 2018-03-28 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Kombination aus trpm8-agonist und nachr-agonist zur behandlung von obesitas und verwandten erkrankungen sowie zur gewichtsreduzierung |
| JP6851322B2 (ja) | 2015-05-27 | 2021-03-31 | ユーシービー バイオファルマ エスアールエル | 神経疾患を治療する方法 |
| WO2016196895A1 (en) * | 2015-06-05 | 2016-12-08 | Joslin Diabetes Center, Inc. | Methods amd compositions for promoting thermogenic potential |
| WO2016201370A1 (en) | 2015-06-12 | 2016-12-15 | Dana-Farber Cancer Institute, Inc. | Combination therapy of transcription inhibitors and kinase inhibitors |
| US20160361496A1 (en) * | 2015-06-15 | 2016-12-15 | Nuance Designs Of Ct, Llc | Autoinjector |
| JP2017000667A (ja) | 2015-06-16 | 2017-01-05 | 国立大学法人三重大学 | 無針注射器及びそれを用いた注射対象領域へのdna導入方法 |
| WO2016205691A1 (en) | 2015-06-19 | 2016-12-22 | Massachusetts Institute Of Technology | Alkenyl substituted 2,5-piperazinediones and their use in compositions for delivering an agent to a subject or cell |
| CA2990360C (en) | 2015-06-24 | 2024-02-13 | Janssen Pharmaceutica Nv | Anti-vista antibodies and fragments |
| UY36751A (es) | 2015-06-26 | 2017-01-31 | Novartis Ag | Anticuerpos de factor xi y métodos de uso |
| CN107922500A (zh) | 2015-06-29 | 2018-04-17 | 洛克菲勒大学 | 具有增强的激动剂活性的抗cd40抗体 |
| US10576206B2 (en) | 2015-06-30 | 2020-03-03 | Kaleo, Inc. | Auto-injectors for administration of a medicament within a prefilled syringe |
| US10059741B2 (en) | 2015-07-01 | 2018-08-28 | Aileron Therapeutics, Inc. | Peptidomimetic macrocycles |
| US20180305451A1 (en) | 2015-07-13 | 2018-10-25 | Compugen Ltd. | Hide1 compositions and methods |
| TWI796283B (zh) | 2015-07-31 | 2023-03-21 | 德商安美基研究(慕尼黑)公司 | Msln及cd3抗體構築體 |
| EA039859B1 (ru) | 2015-07-31 | 2022-03-21 | Эмджен Рисерч (Мюник) Гмбх | Биспецифические конструкты антител, связывающие egfrviii и cd3 |
| TWI793062B (zh) | 2015-07-31 | 2023-02-21 | 德商安美基研究(慕尼黑)公司 | Dll3及cd3抗體構築體 |
| TWI717375B (zh) | 2015-07-31 | 2021-02-01 | 德商安美基研究(慕尼黑)公司 | Cd70及cd3抗體構築體 |
| TWI829617B (zh) | 2015-07-31 | 2024-01-21 | 德商安美基研究(慕尼黑)公司 | Flt3及cd3抗體構築體 |
| TWI744242B (zh) | 2015-07-31 | 2021-11-01 | 德商安美基研究(慕尼黑)公司 | Egfrviii及cd3抗體構築體 |
| JO3620B1 (ar) | 2015-08-05 | 2020-08-27 | Amgen Res Munich Gmbh | مثبطات نقطة فحص مناعية للاستخدام في علاج سرطانات محمولة عبر الدم |
| US10179128B2 (en) | 2015-08-31 | 2019-01-15 | Amicus Therapeutics, Inc. | Regimens for treating and preventing lysosomal disorders and degenerative disorders of the central nervous system |
| AU2016319125B2 (en) | 2015-09-09 | 2021-04-08 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinases |
| WO2017044633A1 (en) | 2015-09-10 | 2017-03-16 | Aileron Therapeutics, Inc. | Peptidomimetic macrocycles as modulators of mcl-1 |
| WO2017048702A1 (en) | 2015-09-14 | 2017-03-23 | Infinity Pharmaceuticals, Inc. | Solid forms of isoquinolinone derivatives, process of making, compositions comprising, and methods of using the same |
| EP3350333B2 (de) | 2015-09-17 | 2025-08-06 | ModernaTX, Inc. | Polynukleotide mit stabilisierendem schwanzbereich |
| JP6948313B6 (ja) | 2015-09-17 | 2022-01-14 | モデルナティエックス インコーポレイテッド | 治療剤の細胞内送達のための化合物および組成物 |
| RU2638457C2 (ru) | 2015-09-28 | 2017-12-13 | Общество С Ограниченной Ответственностью "Онкомакс" | Антитела, специфически связывающие рецептор 1 типа фактора роста фибробластов, применение антител для лечения онкологического заболевания, способ получения антител |
| EP3355914B1 (de) | 2015-09-29 | 2024-03-06 | The General Hospital Corporation | Eine bcg-haltige zusammensetzung zur cholesterolsenkung. |
| PL3356386T3 (pl) | 2015-09-30 | 2024-08-05 | Rhythm Pharmaceuticals, Inc. | Sposób leczenia zaburzeń związanych ze szlakiem receptora melanokortyny 4 |
| WO2017059389A1 (en) | 2015-10-01 | 2017-04-06 | Kythera Biopharmaceuticals, Inc. | Compositions comprising a statin for use in methods of adipolysis |
| GB201518684D0 (en) | 2015-10-21 | 2015-12-02 | Glaxosmithkline Biolog Sa | Vaccine |
| JO3555B1 (ar) | 2015-10-29 | 2020-07-05 | Merck Sharp & Dohme | جسم مضاد يبطل فعالية فيروس الالتهاب الرئوي البشري |
| CN118725134A (zh) | 2015-11-08 | 2024-10-01 | 豪夫迈·罗氏有限公司 | 筛选多特异性抗体的方法 |
| EP3374390A1 (de) | 2015-11-13 | 2018-09-19 | Visterra, Inc. | Zusammensetzungen und verfahren zur behandlung und vorbeugung von grippe |
| AU2016356780A1 (en) | 2015-11-19 | 2018-06-28 | Bristol-Myers Squibb Company | Antibodies against glucocorticoid-induced tumor necrosis factor receptor (GITR) and uses thereof |
| UY36990A (es) | 2015-11-21 | 2017-11-30 | Fundació Privada Inst De Recerca De La Sida-Caixa (Irsicaixa) | Derivados de anticuerpos contra el vih con actividad dual antiviral e inmunomodulatoria |
| WO2017095875A1 (en) | 2015-11-30 | 2017-06-08 | Bristol-Myers Squibb Company | Anti human ip-10 antibodies and their uses |
| SI3383920T1 (sl) | 2015-11-30 | 2024-06-28 | The Regents Of The University Of California | Dostava tumorsko specifičnega koristnega tovora in imunska aktivacija z uporabo humanega protitelesa, ki cilja visoko specifičen površinski antigen tumorskih celic |
| WO2017096432A1 (en) | 2015-12-09 | 2017-06-15 | Admedus Vaccines Pty Ltd | Immunomodulating composition for treatment |
| CA3005696A1 (en) | 2015-12-18 | 2017-06-22 | Intervet International B.V. | Caninized human antibodies to human and canine il-4r alpha |
| LT3394030T (lt) | 2015-12-22 | 2022-04-11 | Modernatx, Inc. | Junginiai ir kompozicijos terapinei medžiagai teikti intraceliuliniu būdu |
| RU2757314C2 (ru) | 2016-01-22 | 2021-10-13 | Мерк Шарп И Доум Корп. | Антитела против фактора свертывания xi |
| JOP20170017B1 (ar) | 2016-01-25 | 2021-08-17 | Amgen Res Munich Gmbh | تركيب صيدلي يتضمن تركيبات جسم مضاد ثنائي الاختصاص |
| IL313507A (en) | 2016-02-03 | 2024-08-01 | Amgen Res Munich Gmbh | Constructs of bispecific antibodies to BCMA and CD3 that bind to T cells, preparations containing the same and uses thereof |
| EA201891753A1 (ru) | 2016-02-03 | 2019-01-31 | Эмджен Рисерч (Мюник) Гмбх | Биспецифические конструкции антител к psma и cd3, вовлекающие т-клетки |
| WO2017137830A1 (en) | 2016-02-12 | 2017-08-17 | Janssen Pharmaceutica Nv | Anti-vista (b7h5) antibodies |
| AU2017219254B2 (en) | 2016-02-17 | 2019-12-12 | Novartis Ag | TGFbeta 2 antibodies |
| AU2017222564A1 (en) | 2016-02-24 | 2018-09-06 | Visterra, Inc. | Formulations of antibody molecules to influenza virus |
| ES2985566T3 (es) | 2016-03-04 | 2024-11-06 | Univ Rockefeller | Anticuerpos contra CD40 con actividad agonista mejorada |
| US10143746B2 (en) | 2016-03-04 | 2018-12-04 | Bristol-Myers Squibb Company | Immunomodulators |
| EP3423494A1 (de) | 2016-03-04 | 2019-01-09 | Bristol-Myers Squibb Company | Kombinationstherapie mit anti-cd73-antikörpern |
| EP3426693A4 (de) | 2016-03-08 | 2019-11-13 | Academia Sinica | Verfahren zur modularen synthese von n-glykanen und anordnungen davon |
| WO2017161116A1 (en) | 2016-03-17 | 2017-09-21 | Infinity Pharmaceuticals, Inc. | Isotopologues of isoquinolinone and quinazolinone compounds and uses thereof as pi3k kinase inhibitors |
| AU2017241925B2 (en) | 2016-03-28 | 2022-01-13 | Ichor Medical Systems, Inc. | Method and apparatus for delivery of therapeutic agents |
| US10358463B2 (en) | 2016-04-05 | 2019-07-23 | Bristol-Myers Squibb Company | Immunomodulators |
| CA3020312A1 (en) | 2016-04-06 | 2017-10-12 | Greenlight Biosciences, Inc. | Cell-free production of ribonucleic acid |
| US10596321B2 (en) | 2016-04-08 | 2020-03-24 | Allergan, Inc. | Aspiration and injection device |
| KR20230119259A (ko) | 2016-04-15 | 2023-08-16 | 이뮤넥스트, 인크. | 항-인간 vista 항체 및 이의 용도 |
| EP3445385A4 (de) | 2016-04-18 | 2019-11-20 | The Trustees of Columbia University in the City of New York | An der progression nichtalkoholischer steatohepatitis (nash) beteiligte therapeutische ziele |
| US12297266B2 (en) | 2016-04-18 | 2025-05-13 | Celldex Therapeutics, Inc. | Agonistic antibodies that bind human CD40 and uses thereof |
| JOP20170091B1 (ar) | 2016-04-19 | 2021-08-17 | Amgen Res Munich Gmbh | إعطاء تركيبة ثنائية النوعية ترتبط بـ cd33 وcd3 للاستخدام في طريقة لعلاج اللوكيميا النخاعية |
| CA3019588A1 (en) | 2016-04-20 | 2017-10-26 | Merck Sharp & Dohme Corp. | Cmv neutralizing antigen binding proteins |
| SI3445850T1 (sl) | 2016-04-22 | 2021-12-31 | BioNTech SE | Postopki za zagotavljanje enoverižne RNA |
| EP3932453B1 (de) * | 2016-04-29 | 2025-11-05 | Altaviz LLC | Selbständig energieversorgte spritze |
| JP7422480B2 (ja) | 2016-05-04 | 2024-01-26 | アムジエン・インコーポレーテツド | 制御性t細胞の増殖のためのインターロイキン-2変異タンパク質 |
| MX392069B (es) | 2016-05-09 | 2025-03-21 | Bristol Myers Squibb Co | Anticuerpos del ligando similar al factor de necrosis tumoral 1a (tl1a) y usos de los mismos. |
| TW201802121A (zh) | 2016-05-25 | 2018-01-16 | 諾華公司 | 抗因子XI/XIa抗體之逆轉結合劑及其用途 |
| EA201892385A1 (ru) | 2016-06-03 | 2019-06-28 | Санофи Пастер Инк. | Модификация сконструированных полипептидов гемагглютинина вируса гриппа |
| US10105449B2 (en) | 2016-06-07 | 2018-10-23 | Massachusetts Institute Of Technology | Drug delivery polymers and uses thereof |
| WO2017214269A1 (en) | 2016-06-08 | 2017-12-14 | Infinity Pharmaceuticals, Inc. | Heterocyclic compounds and uses thereof |
| IL315266A (en) | 2016-06-14 | 2024-10-01 | Merck Sharp ַ& Dohme Llc | Anti-coagulation factor xi antibodies |
| WO2017218704A1 (en) | 2016-06-14 | 2017-12-21 | Modernatx, Inc. | Stabilized formulations of lipid nanoparticles |
| US20180002417A1 (en) | 2016-06-15 | 2018-01-04 | Celimmune Llc | Methods and Compositions for the Treatment of Celiac Disease, Non-Celiac Gluten Sensitivity, and Refractory Celiac Disease |
| GB201610599D0 (en) | 2016-06-17 | 2016-08-03 | Glaxosmithkline Biologicals Sa | Immunogenic Composition |
| US10328205B2 (en) * | 2016-07-07 | 2019-06-25 | Shl Medical Ag | Drug delivery device with pneumatic power pack |
| CN109757103B (zh) | 2016-07-14 | 2024-01-02 | 百时美施贵宝公司 | 针对tim3的抗体及其用途 |
| WO2018018082A1 (en) | 2016-07-26 | 2018-02-01 | The Australian National University | Immunostimulatory compositions and uses therefor |
| US20180028755A1 (en) * | 2016-07-28 | 2018-02-01 | Portal Instruments, Inc. | Connected health platform including needle-free injector system |
| NL2017267B1 (en) | 2016-07-29 | 2018-02-01 | Aduro Biotech Holdings Europe B V | Anti-pd-1 antibodies |
| NL2017270B1 (en) | 2016-08-02 | 2018-02-09 | Aduro Biotech Holdings Europe B V | New anti-hCTLA-4 antibodies |
| WO2018027042A1 (en) | 2016-08-03 | 2018-02-08 | Bio-Techne Corporation | Identification of vsig3/vista as a novel immune checkpoint and use thereof for immunotherapy |
| CA3031797A1 (en) | 2016-08-05 | 2018-02-08 | Sanofi Pasteur, Inc. | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| NL2017294B1 (en) | 2016-08-05 | 2018-02-14 | Univ Erasmus Med Ct Rotterdam | Natural cryptic exon removal by pairs of antisense oligonucleotides. |
| CA3031799A1 (en) | 2016-08-05 | 2018-02-08 | Sanofi Pasteur, Inc. | Multivalent pneumococcal polysaccharide-protein conjugate composition |
| CN109641911B (zh) | 2016-08-19 | 2023-02-21 | 百时美施贵宝公司 | seco-环丙吡咯并吲哚化合物和其抗体-药物缀合物以及制备和使用方法 |
| CA3034057A1 (en) | 2016-08-22 | 2018-03-01 | CHO Pharma Inc. | Antibodies, binding fragments, and methods of use |
| WO2018035710A1 (en) | 2016-08-23 | 2018-03-01 | Akeso Biopharma, Inc. | Anti-ctla4 antibodies |
| US10981976B2 (en) | 2016-08-31 | 2021-04-20 | University Of Rochester | Human monoclonal antibodies to human endogenous retrovirus K envelope (HERV-K) and use thereof |
| JOP20190055A1 (ar) | 2016-09-26 | 2019-03-24 | Merck Sharp & Dohme | أجسام مضادة ضد cd27 |
| EP3305781A1 (de) | 2016-10-07 | 2018-04-11 | Deutsches Krebsforschungszentrum | Chemische substanzen welche die enzymatische aktivität der humanen kallikrein-related peptidase 6 (klk6) hemmen. |
| MX2019004371A (es) | 2016-10-13 | 2019-11-18 | Massachusetts Inst Technology | Anticuerpos que se unen a la proteina de envoltura del virus zika y usos de los mismos. |
| CN110352073A (zh) | 2016-10-28 | 2019-10-18 | 机敏医药股份有限公司 | 针对timp-2的抗体用于改善肾功能的用途 |
| KR102526034B1 (ko) | 2016-11-07 | 2023-04-25 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| US11583504B2 (en) | 2016-11-08 | 2023-02-21 | Modernatx, Inc. | Stabilized formulations of lipid nanoparticles |
| WO2018089829A1 (en) | 2016-11-10 | 2018-05-17 | Fortis Therapeutics, Inc. | Cd46-specific effector cells and uses thereof |
| CA3043277A1 (en) | 2016-11-11 | 2018-05-17 | The Regents Of The University Of California | Anti-cd46 antibodies and methods of use |
| AU2017361856A1 (en) | 2016-11-18 | 2019-05-23 | Neurovive Pharmaceutical Ab | Use of sanglifehrin macrocyclic analogues as anticancer compounds |
| WO2018106738A1 (en) | 2016-12-05 | 2018-06-14 | Massachusetts Institute Of Technology | Brush-arm star polymers, conjugates and particles, and uses thereof |
| MX2019007161A (es) | 2016-12-21 | 2020-02-12 | Mereo Biopharma 3 Ltd | Uso de anticuerpos anti-esclerostina en el tratamiento de osteogenesis imperfecta. |
| US10961305B2 (en) | 2016-12-21 | 2021-03-30 | Mereo Biopharma 3 Limited | Use of anti-sclerostin antibodies in the treatment of osteogenesis imperfecta |
| EP3558420B1 (de) | 2016-12-23 | 2024-09-18 | Kaleo, Inc. | Medikamentenabgabevorrichtung und verfahren zur abgabe von arzneimitteln an säuglinge und kinder |
| AU2017383232B2 (en) | 2016-12-23 | 2024-09-12 | Novartis Ag | Factor XI antibodies and methods of use |
| IL267689B2 (en) | 2016-12-31 | 2025-05-01 | Bioxcel Therapeutics Inc | Use of sublingual dexmedetomidine for the treatment of agitation |
| RU2769769C2 (ru) | 2017-01-05 | 2022-04-05 | Кахр Медикал Лтд. | СЛИТЫЙ БЕЛОК SIRPα-4-1BBL И СПОСОБЫ ЕГО ПРИМЕНЕНИЯ |
| CN110536693B (zh) | 2017-01-05 | 2023-12-22 | 卡尔医学有限公司 | Pd1-41bbl融合蛋白及使用其的方法 |
| TW201833140A (zh) | 2017-01-09 | 2018-09-16 | 美商莫瑞麥克製藥公司 | 抗fgfr抗體及使用方法 |
| AU2018210313A1 (en) | 2017-01-17 | 2019-06-20 | Kaleo, Inc. | Medicament delivery devices with wireless connectivity and event detection |
| US11377497B2 (en) | 2017-01-23 | 2022-07-05 | Suzhou Alphamab Co., Ltd. | PD-L1 binding polypeptide or composite |
| WO2018140510A1 (en) | 2017-01-25 | 2018-08-02 | Biogen Ma Inc. | Compositions and methods for treatment of stroke and other cns disorders |
| SMT202500367T1 (it) | 2017-02-02 | 2025-11-10 | Amgen Res Munich Gmbh | Composizione farmaceutica a basso ph comprendente costrutti di anticorpo che reclutano le cellule t |
| GB2559553B (en) * | 2017-02-06 | 2021-10-13 | Consort Medical Plc | A medicament delivery device |
| GB201702144D0 (en) | 2017-02-09 | 2017-03-29 | Annexin Pharmaceuticals Ab | Therapeutic compositions |
| JP2020507632A (ja) | 2017-02-10 | 2020-03-12 | マウント タム セラピューティクス, インコーポレイテッドMount Tam Therapeutics, Inc. | ラパマイシン類似体 |
| CA3051839A1 (en) | 2017-02-17 | 2018-08-23 | Bristol-Myers Squibb Company | Antibodies to alpha-synuclein and uses thereof |
| GB201703876D0 (en) | 2017-03-10 | 2017-04-26 | Berlin-Chemie Ag | Pharmaceutical combinations |
| CA3055653A1 (en) | 2017-03-15 | 2018-09-20 | Modernatx, Inc. | Lipid nanoparticle formulation |
| EP4186888B1 (de) | 2017-03-15 | 2025-11-26 | ModernaTX, Inc. | Verbindung und zusammensetzungen zur intrazellulären verabreichung von therapeutischen wirkstoffen |
| US11555031B2 (en) | 2017-03-20 | 2023-01-17 | The Broad Institute, Inc. | Compounds and methods for regulating insulin secretion |
| USD867582S1 (en) | 2017-03-24 | 2019-11-19 | Allergan, Inc. | Syringe device |
| CA3058198A1 (en) | 2017-04-05 | 2018-10-11 | The Broad Institute, Inc. | Tricyclic compounds as glycogen synthase kinase 3 (gsk3) inhibitors and uses thereof |
| TWI788340B (zh) | 2017-04-07 | 2023-01-01 | 美商必治妥美雅史谷比公司 | 抗icos促效劑抗體及其用途 |
| TWI796329B (zh) | 2017-04-07 | 2023-03-21 | 美商默沙東有限責任公司 | 抗-ilt4抗體及抗原結合片段 |
| KR102702926B1 (ko) | 2017-04-13 | 2024-09-06 | 사이로파 비.브이. | 항-sirp 알파 항체 |
| CN110573492A (zh) | 2017-04-25 | 2019-12-13 | 阿米库斯治疗学公司 | 用于预防和/或治疗中枢神经系统退行性失调和/或溶酶体贮积失调的新颖组合物 |
| UY37726A (es) | 2017-05-05 | 2018-11-30 | Amgen Inc | Composición farmacéutica que comprende constructos de anticuerpos biespecíficos para almacenamiento y administración mejorados |
| EP3621649A2 (de) | 2017-05-10 | 2020-03-18 | Albajuna Therapeutics, S.L. | Fc-fusionsproteinderivate mit hoher dualer hiv-antiviraler und immunmodulatorischer aktivität |
| EP3625251A1 (de) | 2017-05-15 | 2020-03-25 | University Of Rochester | Breit neutralisierende monoklonale anti-influenza-antikörper und verwendungen davon |
| US20200299400A1 (en) | 2017-05-25 | 2020-09-24 | Bristol-Myers Squibb Company | Antibodies comprising modified heavy constant regions |
| WO2018232120A1 (en) | 2017-06-14 | 2018-12-20 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of agents |
| MX2019015524A (es) | 2017-06-23 | 2020-09-10 | Univ Maryland | Composiciones inmunogenicas. |
| JP2020524527A (ja) | 2017-06-23 | 2020-08-20 | スージョウ ボジュホワ バイオメディカル デクノロジー カンパニー リミテッドSuzhou Bojuhua Biomedical Technology Co. Ltd | ヒト腫瘍幹細胞をターゲティングするモノクローナル抗体及びその使用 |
| EP3642220A1 (de) | 2017-06-23 | 2020-04-29 | Bristol-Myers Squibb Company | Immunmodulatoren als antagonisten von pd-1 |
| WO2019013789A1 (en) | 2017-07-12 | 2019-01-17 | Curza Global, Llc | ANTIMICROBIAL COMPOUNDS |
| WO2019013790A1 (en) | 2017-07-12 | 2019-01-17 | Curza Global, Llc | ANTIMICROBIAL COMPOUNDS AND USES THEREOF |
| GB201711635D0 (en) | 2017-07-19 | 2017-08-30 | Glaxosmithkline Biologicals Sa | Immunogenic composition |
| AU2018322475A1 (en) | 2017-08-22 | 2020-03-05 | Biogen Ma Inc. | Pharmaceutical compositions and dosage regimens containing anti-alpha(v)beta(6) antibodies |
| GB2584009B (en) | 2017-08-28 | 2022-03-23 | Revagenix Inc | Aminoglycosides and uses thereof |
| JP2020531041A (ja) | 2017-08-28 | 2020-11-05 | 上海易楽生物技術有限公司 | ポリペプチドとポリペプチドに結合される抗体 |
| EP3675817A1 (de) | 2017-08-31 | 2020-07-08 | Modernatx, Inc. | Verfahren zur herstellung von lipidnanopartikeln |
| CN111051332B (zh) | 2017-10-03 | 2024-09-06 | 百时美施贵宝公司 | 免疫调节剂 |
| EP3692061A1 (de) | 2017-10-04 | 2020-08-12 | Amgen Inc. | Transthyretinimmunoglobulinfusionen |
| EP3694552A1 (de) | 2017-10-10 | 2020-08-19 | Tilos Therapeutics, Inc. | Anti-lap-antikörper und verwendungen davon |
| KR102894284B1 (ko) | 2017-10-11 | 2025-12-03 | 그린라이트 바이오사이언시스, 아이엔씨. | 뉴클레오시드 트리포스페이트 및 리보핵산 생산을 위한 방법 및 조성물 |
| US12441998B2 (en) | 2017-10-12 | 2025-10-14 | The Trustees Of Columbia University In The City Of New York | SLC2A1 lncRNA as a biologic and related treatments and methods |
| WO2019080883A1 (en) | 2017-10-26 | 2019-05-02 | Immuneonco Biopharmaceuticals (Shanghai) Co., Ltd. | NOVEL RECOMBINANT FUSION PROTEINS, PREPARATION THEREOF AND USE THEREOF |
| KR20240171186A (ko) | 2017-10-26 | 2024-12-06 | 다케다 야쿠힌 고교 가부시키가이샤 | 글루코세레브로시다제 및 이소파고민을 포함하는 제제 |
| JP2021501801A (ja) | 2017-11-01 | 2021-01-21 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | 癌の処置に用いるための免疫刺激アゴニスト抗体 |
| CN111712518B (zh) | 2017-11-17 | 2025-03-25 | 默沙东有限责任公司 | 对免疫球蛋白样转录物3(ilt3)具有特异性的抗体及其用途 |
| WO2019109079A1 (en) | 2017-12-01 | 2019-06-06 | North Carolina State University | Fibrin particles and methods of making the same |
| AU2018383679B2 (en) | 2017-12-11 | 2025-10-09 | Amgen Inc. | Continuous manufacturing process for bispecific antibody products |
| EP3727333A1 (de) * | 2017-12-21 | 2020-10-28 | Taiwan Liposome Company Ltd | Triptanzusammensetzungen mit verzögerter freisetzung und verfahren zur herstellung davon auf subdermalem weg oder dergleichen |
| SG11202004971YA (en) | 2017-12-21 | 2020-06-29 | Gliapharm Sa | Compositions and methods of treatment for neurological disorders comprising motor neuron diseases |
| US20200339591A1 (en) | 2017-12-21 | 2020-10-29 | Gliapharm Sa | Compositions and methods of treatment for neurological disorders comprising a dementia |
| GB201721576D0 (en) | 2017-12-21 | 2018-02-07 | Glaxosmithkline Biologicals Sa | Hla antigens and glycoconjugates thereof |
| GB201721582D0 (en) | 2017-12-21 | 2018-02-07 | Glaxosmithkline Biologicals Sa | S aureus antigens and immunogenic compositions |
| CN111788227B (zh) | 2017-12-27 | 2025-02-25 | 百时美施贵宝公司 | 抗cd40抗体及其用途 |
| TW201940518A (zh) | 2017-12-29 | 2019-10-16 | 美商安進公司 | 針對muc17和cd3之雙特異性抗體構建體 |
| US12247060B2 (en) | 2018-01-09 | 2025-03-11 | Marengo Therapeutics, Inc. | Calreticulin binding constructs and engineered T cells for the treatment of diseases |
| EP3737700A1 (de) | 2018-01-12 | 2020-11-18 | Bristol-Myers Squibb Company | Antikörper gegen tim3 und verwendungen davon |
| WO2019148412A1 (en) | 2018-02-01 | 2019-08-08 | Merck Sharp & Dohme Corp. | Anti-pd-1/lag3 bispecific antibodies |
| US11787857B2 (en) | 2018-02-02 | 2023-10-17 | Bio-Techne Corporation | Compounds that modulate the interaction of VISTA and VSIG3 and methods of making and using |
| BR112020014978A2 (pt) | 2018-02-05 | 2020-12-22 | Sanofi Pasteur, Inc. | Composição de conjugado polissacarídeo-proteína pneumocócico multivalente |
| EP3749357A4 (de) | 2018-02-05 | 2022-04-20 | Sanofi Pasteur, Inc. | Polyvalente pneumokokken-polysaccharid-proteinkonjugatzusammensetzung |
| KR101859735B1 (ko) * | 2018-02-07 | 2018-05-21 | 주식회사 피테크 | 무통 주사 장치 |
| MA51747A (fr) | 2018-02-08 | 2020-12-16 | Amgen Inc | Formulation d'anticorps pharmaceutique à ph faible |
| NL2020520B1 (en) | 2018-03-02 | 2019-09-12 | Labo Bio Medical Invest B V | Multispecific binding molecules for the prevention, treatment and diagnosis of neurodegenerative disorders |
| EP3765517A1 (de) | 2018-03-14 | 2021-01-20 | Elstar Therapeutics, Inc. | An calreticulin bindende multifunktionsmoleküle und ihre verwendungen |
| KR20200143391A (ko) | 2018-03-15 | 2020-12-23 | 비온테크 알엔에이 파마슈티컬스 게엠베하 | 5'-캡-트리뉴클레오티드- 또는 고차의 올리고뉴클레오티드 화합물 및 이들의 rna 안정화, 단백질 발현 및 치료에서의 사용 |
| EP3768715A1 (de) | 2018-03-23 | 2021-01-27 | Bristol-Myers Squibb Company | Antikörper gegen mica und/or micb und deren verwendungen |
| CN112165957A (zh) | 2018-03-26 | 2021-01-01 | 上海易乐生物技术有限公司 | proBDNF调节剂在B细胞相关疾病中的用途 |
| WO2019195126A1 (en) | 2018-04-02 | 2019-10-10 | Bristol-Myers Squibb Company | Anti-trem-1 antibodies and uses thereof |
| WO2019194858A1 (en) | 2018-04-03 | 2019-10-10 | Achaogen, Inc. | Modular synthesis of aminoglycosides |
| PT3773713T (pt) | 2018-04-06 | 2025-07-29 | Regeneron Pharma | Anticorpo agonista do recetor de leptina para usar no aumento da massa óssea num sujeito que sofre de disfunção metabólica ou de hipoleptinemia |
| US20210169969A1 (en) | 2018-04-06 | 2021-06-10 | Leonardus H.T. Van Der Ploeg | Compositions for treating kidney disease |
| BR112020020826A2 (pt) | 2018-04-12 | 2021-01-19 | Bristol-Myers Squibb Company | Terapia de combinação anticâncer com anticorpo antagonista de cd73 e anticorpo antagonista do eixo pd-1/pd-l1 |
| CA3097369A1 (en) | 2018-04-17 | 2019-10-24 | Celldex Therapeutics, Inc. | Anti-cd27 and anti-pd-l1 antibodies and bispecific constructs |
| US12139480B2 (en) | 2018-04-20 | 2024-11-12 | Iomx Therapeutics Ag | 5-thiazolecarboxamide kinase inhibitor and uses thereof |
| KR102107578B1 (ko) * | 2018-05-02 | 2020-05-07 | 주식회사 피테크 | 무통 주사기용 약액주입 제어장치 및 이를 구비하는 무통 주사기 |
| KR102104080B1 (ko) * | 2018-05-02 | 2020-04-23 | 주식회사 피테크 | 무통 주사기용 음파발진장치 및 이를 구비하는 무통 주사기 |
| KR101907552B1 (ko) | 2018-05-03 | 2018-10-12 | 주식회사 피테크 | 무바늘 무통 주사 장치 |
| CA3096703A1 (en) | 2018-05-03 | 2019-11-07 | University Of Rochester | Anti-influenza neuraminidase monoclonal antibodies and uses thereof |
| CA3101364A1 (en) | 2018-05-25 | 2019-11-28 | Primmune Therapeutics, Inc. | Tlr7 agonists |
| CN119080931A (zh) | 2018-06-04 | 2024-12-06 | 马萨诸塞州渤健公司 | 具有降低的效应功能的抗vla-4抗体 |
| US11951286B2 (en) * | 2018-06-12 | 2024-04-09 | Banza Stamping Industry Corp. | Automatic injection device for fluid |
| GB201809746D0 (en) | 2018-06-14 | 2018-08-01 | Berlin Chemie Ag | Pharmaceutical combinations |
| BR112020025987A2 (pt) | 2018-06-21 | 2021-03-23 | Yumanity Therapeutics, Inc. | composições e métodos para tratamento e prevenção de distúrbios neurológicos |
| EP3810132A4 (de) | 2018-06-25 | 2022-06-22 | Dana-Farber Cancer Institute, Inc. | Kinaseinhibitoren der taire und ihre verwendungen |
| BR112020026672A2 (pt) | 2018-06-27 | 2021-03-30 | Bioxcel Therapeutics, Inc. | Formulações de filme contendo dexmedetomidina e métodos para produzi-las |
| CA3105448A1 (en) | 2018-07-03 | 2020-01-09 | Elstar Therapeutics, Inc. | Anti-tcr antibody molecules and uses thereof |
| KR102945860B1 (ko) | 2018-07-11 | 2026-03-31 | 카 메디컬 리미티드 | SIRPalpha-4-1BBL 변이체 융합 단백질 및 이의 사용 방법 |
| US11929160B2 (en) | 2018-07-16 | 2024-03-12 | Kaleo, Inc. | Medicament delivery devices with wireless connectivity and compliance detection |
| CA3107186A1 (en) | 2018-07-30 | 2020-02-06 | Amgen Research (Munich) Gmbh | Prolonged administration of a bispecific antibody construct binding to cd33 and cd3 |
| CA3105729A1 (en) | 2018-07-31 | 2020-02-06 | Amgen Research (Munich) Gmbh | Dosing regimen for bcma-cd3 bispecific antibodies |
| SG11202013248YA (en) | 2018-07-31 | 2021-02-25 | Amgen Inc | Pharmaceutical formulations of masked antibodies |
| WO2020025792A1 (en) | 2018-08-03 | 2020-02-06 | Amgen Research (Munich) Gmbh | Antibody constructs for cldn18.2 and cd3 |
| JP7193058B2 (ja) | 2018-08-08 | 2022-12-20 | イミューンオンコ バイオファーマシューティカルズ (シャンハイ) インコーポレイテッド | Cd47およびher2を標的とする組換え二機能性タンパク質 |
| WO2020047251A1 (en) | 2018-08-29 | 2020-03-05 | President And Fellows Of Harvard College | O-glcnac transferase inhibitors and uses thereof |
| WO2020049208A1 (es) | 2018-09-09 | 2020-03-12 | Fundacio Privada Institut De Recerca De La Sida - Caixa | Aurora cinasa como diana para tratar, prevenir o curar una infección por vih o sida |
| EP3853252A1 (de) | 2018-09-18 | 2021-07-28 | Merrimack Pharmaceuticals, Inc. | Anti-tnfr2-antikörper und verwendungen davon |
| WO2020061367A1 (en) | 2018-09-19 | 2020-03-26 | Modernatx, Inc. | Compounds and compositions for intracellular delivery of therapeutic agents |
| WO2020061457A1 (en) | 2018-09-20 | 2020-03-26 | Modernatx, Inc. | Preparation of lipid nanoparticles and methods of administration thereof |
| KR102199724B1 (ko) * | 2018-10-05 | 2021-01-07 | 주식회사 피테크 | 무바늘 무통 주사 장치 |
| CN113164780A (zh) | 2018-10-10 | 2021-07-23 | 泰洛斯治疗公司 | 抗lap抗体变体及其用途 |
| JP7644706B2 (ja) | 2018-10-11 | 2025-03-12 | アムジエン・インコーポレーテツド | 二重特異性抗体コンストラクトの下流プロセシング |
| DK3643713T3 (da) | 2018-10-23 | 2025-11-03 | Iomx Therapeutics Ag | Heterocykliske kinaseinhibitorer og deres anvendelse |
| WO2020084005A1 (en) | 2018-10-23 | 2020-04-30 | Myelopro Diagnostics And Research Gmbh | Compounds targeting mutant calreticulin |
| US11160791B2 (en) | 2018-11-01 | 2021-11-02 | Medefil, Inc. | Dexmedetomidine injection premix formulation in ready to use (RTU) bags |
| CA3117945A1 (en) | 2018-11-09 | 2020-05-14 | University Of Massachusetts | Anti-cfae antibodies and methods of use |
| CN113316590B (zh) | 2018-11-16 | 2025-02-28 | 百时美施贵宝公司 | 抗nkg2a抗体及其用途 |
| EP3887397A1 (de) | 2018-11-28 | 2021-10-06 | Bristol-Myers Squibb Company | Antikörper mit modifizierten schweren konstanten regionen |
| WO2020113063A1 (en) | 2018-11-30 | 2020-06-04 | Aleta Biotherapeutics Inc. | Single domain antibodies against cll-1 |
| WO2020118011A1 (en) | 2018-12-06 | 2020-06-11 | Alexion Pharmaceuticals, Inc. | Anti-alk2 antibodies and uses thereof |
| KR20210104744A (ko) | 2018-12-18 | 2021-08-25 | 리제너론 파마슈티칼스 인코포레이티드 | 렙틴 수용체, gdf8 및 액티빈 a에 대한 길항제를 사용하여 체중 및 제지방 근육량을 향상시키기 위한 조성물 및 방법 |
| TW202039554A (zh) | 2018-12-19 | 2020-11-01 | 瑞士商諾華公司 | 抗TNF-α抗體 |
| WO2020132378A2 (en) | 2018-12-22 | 2020-06-25 | Gliapharm Sa | Compositions and methods of treatment for neurological disorders comprising depression |
| US12281126B2 (en) | 2018-12-28 | 2025-04-22 | Dana-Farber Cancer Institute, Inc. | Inhibitors of cyclin-dependent kinase 7 and uses thereof |
| EP3902584A4 (de) | 2018-12-29 | 2022-09-07 | Kaleo, Inc. | Vorrichtungen und verfahren zur abgabe von substanzen in einer vorgefüllten spritze |
| WO2020144535A1 (en) | 2019-01-08 | 2020-07-16 | Aduro Biotech Holdings, Europe B.V. | Methods and compositions for treatment of multiple myeloma |
| AU2020209167B2 (en) | 2019-01-16 | 2025-09-04 | Curza Global, Llc | Antimicrobial compounds and methods |
| AU2020209170B2 (en) | 2019-01-16 | 2025-09-04 | Curza Global, Llc | Antimicrobial compounds and methods |
| CN109762067B (zh) | 2019-01-17 | 2020-02-28 | 北京天广实生物技术股份有限公司 | 结合人Claudin 18.2的抗体及其用途 |
| BR112021014276A2 (pt) | 2019-01-22 | 2021-09-28 | Genentech, Inc. | Anticorpos iga isolados, moléculas de fusão igg-iga isoladas, ácido nucleico isolado, célula hospedeira, método para produzir um anticorpo, para tratar um indivíduo, para aumentar a expressão de dímeros, trímeros ou tetrâmeros, para aumentar a produção de polímeros, para aumentar a produção de dímeros, para aumentar a produção de um polímero, para diminuir a produção de polímeros, para aumentar a expressão transitória de um anticorpo, para expressar dímeros de moléculas de fusão, para expressar dímeros, trímeros ou tetrâmeros, para purificar um anticorpo, para purificar um estado oligomérico de um anticorpo, composição farmacêutica e uso do anticorpo |
| IL285067B2 (en) | 2019-01-22 | 2025-02-01 | Aeovian Pharmaceuticals Inc | MTORC Modulators and Their Uses |
| CN113396162B (zh) | 2019-01-22 | 2024-08-16 | 百时美施贵宝公司 | 抗IL-7Rα亚基的抗体及其用途 |
| CN118697900A (zh) | 2019-01-31 | 2024-09-27 | 摩登纳特斯有限公司 | 制备脂质纳米颗粒的方法 |
| WO2020168466A1 (en) | 2019-02-19 | 2020-08-27 | Stemirna Therapeutics Co., Ltd. | Modified nucleoside and synthetic methods thereof |
| CN119039441A (zh) | 2019-02-21 | 2024-11-29 | 马伦戈治疗公司 | 与nkp30结合的抗体分子及其用途 |
| GB2599228B (en) | 2019-02-21 | 2024-02-07 | Marengo Therapeutics Inc | Multifunctional molecules that bind to T cell related cancer cells and uses thereof |
| CA3131953A1 (en) | 2019-03-01 | 2020-09-10 | Merrimack Pharmaceuticals, Inc. | Anti-tnfr2 antibodies and uses thereof |
| US10570210B1 (en) | 2019-03-04 | 2020-02-25 | Beijing Mabworks Biotech Co.Ltd | Antibodies binding CD40 and uses thereof |
| MA55298A (fr) | 2019-03-11 | 2022-01-19 | Biogen Ma Inc | Compositions pharmaceutiques contenant des anticorps anti-lingo-1 |
| US11230593B2 (en) | 2019-03-25 | 2022-01-25 | Visterra, Inc. | Compositions and methods for treating and preventing influenza |
| KR20220005471A (ko) | 2019-04-08 | 2022-01-13 | 바이오젠 엠에이 인코포레이티드 | 항-인테그린 항체 및 이의 용도 |
| US12404331B2 (en) | 2019-04-19 | 2025-09-02 | Tcrcure Biopharma Corp. | Anti-PD-1 antibodies and uses thereof |
| KR20220010535A (ko) | 2019-05-21 | 2022-01-25 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| EP3983015A1 (de) | 2019-06-11 | 2022-04-20 | Regeneron Pharmaceuticals, Inc. | Pcrv-bindende anti-pcrv-antikörper, zusammensetzungen mit anti-pcrv-antikörpern und verfahren zu deren verwendung |
| MX2021014644A (es) | 2019-06-13 | 2022-04-06 | Amgen Inc | Control de perfusion basado en biomasa automatizado en la fabricacion de productos biologicos. |
| EP3997112A1 (de) | 2019-07-08 | 2022-05-18 | Amgen, Inc | Multispezifische transthyretin-immunglobulin-fusionen |
| US20220267409A1 (en) | 2019-07-11 | 2022-08-25 | Kahr Medical Ltd. | Heterodimers and methods of use thereof |
| CN114174536B (zh) | 2019-07-15 | 2026-03-27 | 百时美施贵宝公司 | 抗trem-1抗体及其用途 |
| WO2021011681A1 (en) | 2019-07-15 | 2021-01-21 | Bristol-Myers Squibb Company | Antibodies against human trem-1 and uses thereof |
| CA3145345A1 (en) | 2019-07-15 | 2021-01-21 | Intervet International B.V. | Caninized antibodies against canine ctla-4 |
| WO2021009188A1 (en) | 2019-07-15 | 2021-01-21 | Intervet International B.V. | Caninized antibodies to human and canine ctla-4 |
| BR112022000992A2 (pt) | 2019-07-19 | 2022-06-14 | Arx Llc | Regimes de tratamento de dexmedetomidina não sedantes |
| PH12022550110A1 (en) | 2019-07-31 | 2022-12-12 | Sk Bioscience Co Ltd | Multivalent pneumococcal polysaccharide-protein conjugate compositions and methods of using the same |
| US20220280639A1 (en) | 2019-07-31 | 2022-09-08 | Modernatx, Inc. | Compositions and methods for delivery of rna interference agents to immune cells |
| CA3145580A1 (en) | 2019-08-09 | 2021-02-18 | Kaleo, Inc. | Devices and methods for delivery of substances within a prefilled syringe |
| PH12022550326A1 (en) | 2019-08-13 | 2023-03-13 | Amgen Inc | Interleukin-2 muteins for the expansion of t-regulatory cells |
| AU2020346062A1 (en) | 2019-09-13 | 2022-03-24 | Rutgers, The State University Of New Jersey | AAV-compatible laminin-linker polymerization proteins |
| KR20220088868A (ko) | 2019-10-30 | 2022-06-28 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| LT3819007T (lt) | 2019-11-11 | 2024-10-10 | Amgen Research (Munich) Gmbh | Dozavimo režimas, skirtas anti-bcma agentams |
| US20220396599A1 (en) | 2019-11-13 | 2022-12-15 | Amgen Inc. | Method for Reduced Aggregate Formation in Downstream Processing of Bispecific Antigen-Binding Molecules |
| CA3158103A1 (en) | 2019-11-13 | 2021-05-20 | Curza Global, Llc | Antimicrobial compounds and methods |
| WO2021102143A1 (en) | 2019-11-19 | 2021-05-27 | Bristol-Myers Squibb Company | Macrocyclic peptides as pd-l1 inhibitors and immunomodulators for the treatment of cancer and infectious diseases |
| WO2021119467A1 (en) | 2019-12-11 | 2021-06-17 | Visterra, Inc. | Compositions and methods for treating and preventing influenza |
| IL293471A (en) | 2019-12-17 | 2022-08-01 | Amgen Inc | Dual interleukin-2 / tnf receptor agonist for use in medicine |
| IL293640A (en) | 2019-12-20 | 2022-08-01 | Amgen Inc | CD40 antagonistic mesothelin-targeted multispecific antibody constructs for the treatment of solid tumors |
| CN121108343A (zh) | 2019-12-20 | 2025-12-12 | 英特维特国际股份有限公司 | 犬白介素-4受体α抗体 |
| MX2022007676A (es) | 2019-12-20 | 2022-07-19 | Intervet Int Bv | Anticuerpos caninizados biespecificos para tratar dermatitis atopica. |
| WO2021127500A1 (en) | 2019-12-20 | 2021-06-24 | Momenta Pharmaceuticals, Inc. | Antibodies against integrin alpha 11 beta 1 |
| WO2021136308A1 (en) | 2020-01-03 | 2021-07-08 | Biosion Inc. | Antibodies binding bcma and uses thereof |
| AU2020416273A1 (en) | 2020-01-03 | 2022-07-28 | Marengo Therapeutics, Inc. | Anti-TCR antibody molecules and uses thereof |
| CN114929714B (zh) | 2020-01-06 | 2024-05-31 | 百时美施贵宝公司 | 用于治疗癌症和感染性疾病的作为pd-l1抑制剂和免疫调节剂的大环肽 |
| EP4087857B1 (de) | 2020-01-06 | 2023-11-01 | Bristol-Myers Squibb Company | Immunmodulatoren |
| CN110818795B (zh) | 2020-01-10 | 2020-04-24 | 上海复宏汉霖生物技术股份有限公司 | 抗tigit抗体和使用方法 |
| US20230093169A1 (en) | 2020-01-22 | 2023-03-23 | Amgen Research (Munch) Gmbh | Combinations of antibody constructs and inhibitors of cytokine release syndrome and uses thereof |
| CA3169669A1 (en) | 2020-01-31 | 2021-08-05 | Modernatx, Inc. | Methods of preparing lipid nanoparticles |
| BR112022017174A2 (pt) | 2020-02-27 | 2022-10-18 | Chia Tai Tianqing Pharmaceutical Group Co Ltd | Anticorpo monoclonal isolado, ou uma porção de ligação ao antígeno do mesmo, nucleotídeo, vetor de expressão, célula hospedeira, composição farmacêutica e métodos de tratamento de uma doença alérgica e de um tumor |
| US11642407B2 (en) | 2020-02-28 | 2023-05-09 | Massachusetts Institute Of Technology | Identification of variable influenza residues and uses thereof |
| CN115485302A (zh) | 2020-03-09 | 2022-12-16 | 百时美施贵宝公司 | 具有增强的激动剂活性的针对cd40的抗体 |
| KR20220155332A (ko) | 2020-03-16 | 2022-11-22 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| WO2021188851A1 (en) | 2020-03-19 | 2021-09-23 | Amgen Inc. | Antibodies against mucin 17 and uses thereof |
| WO2021195089A1 (en) | 2020-03-23 | 2021-09-30 | Sorrento Therapeutics, Inc. | Fc-coronavirus antigen fusion proteins, and nucleic acids, vectors, compositions and methods of use thereof |
| KR20220161407A (ko) | 2020-03-30 | 2022-12-06 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| WO2021202235A1 (en) | 2020-04-01 | 2021-10-07 | University Of Rochester | Monoclonal antibodies against the hemagglutinin (ha) and neuraminidase (na) of influenza h3n2 viruses |
| US20230272056A1 (en) | 2020-04-09 | 2023-08-31 | Merck Sharp & Dohme Llc | Affinity matured anti-lap antibodies and uses thereof |
| EP3901151B1 (de) | 2020-04-21 | 2026-04-01 | iOmx Therapeutics AG | Halogenierte heteroaryl- und andere heterocyclische kinaseinhibitoren und verwendungen davon |
| EP4139341A1 (de) | 2020-04-21 | 2023-03-01 | Regeneron Pharmaceuticals, Inc. | Il-2-varianten mit reduzierter bindung an il-2-rezeptor-alpha und verwendungen davon |
| MX2021015024A (es) | 2020-04-28 | 2022-01-18 | Univ Rockefeller | Anticuerpos anti-sars-cov-2 ampliamente neutralizantes y métodos de uso de los mismos. |
| AU2021262482A1 (en) | 2020-04-28 | 2023-01-05 | Iomx Therapeutics Ag | Bicyclic kinase inhibitors and uses thereof |
| CN115461080A (zh) | 2020-04-29 | 2022-12-09 | 安进公司 | 药物配制品 |
| CA3173706A1 (en) | 2020-04-29 | 2021-11-04 | Twinkle R. Christian | Pharmaceutical formulation |
| EP4143227A2 (de) | 2020-04-30 | 2023-03-08 | Sairopa B.V. | Anti-cd103 antikörper |
| WO2021226460A1 (en) | 2020-05-08 | 2021-11-11 | Bristol-Myers Squibb Company | Immunomodulators |
| WO2021231729A1 (en) | 2020-05-13 | 2021-11-18 | Sanofi | Adjuvanted stabilized stem hemagglutinin nanoparticles and methods of using the same to induce broadly neutralizing antibodies against influenza |
| US20230192867A1 (en) | 2020-05-15 | 2023-06-22 | Bristol-Myers Squibb Company | Antibodies to garp |
| CA3183756A1 (en) | 2020-05-19 | 2021-11-25 | Amgen Inc. | Mageb2 binding constructs |
| US20230277524A1 (en) | 2020-05-22 | 2023-09-07 | Trailhead Biosystems Inc. | Combination therapy for treatment of viral infections |
| CN111529830B (zh) * | 2020-05-22 | 2024-10-15 | 江阴市机电技术有限公司 | 一种启动稳定的给药装置 |
| US20240209085A1 (en) | 2020-05-29 | 2024-06-27 | Amgen Inc. | Adverse effects-mitigating administration of a bispecific construct binding to cd33 and cd3 |
| WO2021252708A1 (en) | 2020-06-11 | 2021-12-16 | President And Fellows Of Harvard College | Stabilized trioxacarcin antibody drug conjugates and uses thereof |
| FR3112939B1 (fr) | 2020-07-31 | 2024-01-05 | Univ Montpellier | Produit universel de thérapie cellulaire et son utilisation |
| EP4192511A1 (de) | 2020-08-07 | 2023-06-14 | Fortis Therapeutics, Inc. | Gegen cd46 gerichtete immunkonjugate und verfahren zur verwendung davon |
| EP4200333A1 (de) | 2020-08-24 | 2023-06-28 | Amgen Inc. | Pharmazeutische formulierung mit einem biss, bispezifischem antikörper und methionin |
| WO2022061219A1 (en) * | 2020-09-21 | 2022-03-24 | Boston Scientific Scimed, Inc. | Fluid pressure indicators for medical devices |
| CN116322735A (zh) | 2020-10-02 | 2023-06-23 | 百时美施贵宝公司 | 大环肽硼酸盐免疫调节剂 |
| US20230374073A1 (en) | 2020-10-08 | 2023-11-23 | Bristol-Myers Squibb Company | Dimeric milla immunomodulators |
| KR20230084211A (ko) | 2020-10-09 | 2023-06-12 | 브리스톨-마이어스 스큅 컴퍼니 | 마크로시클릭 면역조정제 |
| US20220267450A1 (en) | 2020-10-14 | 2022-08-25 | Viridian Therapeutics, Inc. | Compositions and methods for treatment of thyroid eye disease |
| JP2023545182A (ja) | 2020-10-15 | 2023-10-26 | インターベット インターナショナル ベー. フェー. | イヌインターロイキン-31受容体アルファに対するイヌ化ラット抗体 |
| EP4238979A4 (de) | 2020-10-30 | 2024-09-25 | Xeno-Interface Inc. | Vernetztes beta-strang-peptid |
| CN117083065A (zh) | 2020-10-30 | 2023-11-17 | 庆应义塾 | 肌肉减少症相关疾病等的新颖治疗和预防 |
| TW202227481A (zh) | 2020-11-04 | 2022-07-16 | 美國洛克菲勒大學 | 中和抗sars-cov-2抗體 |
| WO2022096704A1 (en) | 2020-11-06 | 2022-05-12 | Amgen Inc. | Antigen binding domain with reduced clipping rate |
| CR20230229A (es) | 2020-11-06 | 2023-09-05 | Amgen Res Munich Gmbh | Moléculas de unión a antígeno biespecíficas con múltiples dianas de selectividad aumentada |
| UY39507A (es) | 2020-11-06 | 2022-03-31 | Amgen Res Munich Gmbh | Construcciones polipeptídicas que se unen selectivamente a cldn6 y cd3 |
| TW202225188A (zh) | 2020-11-06 | 2022-07-01 | 德商安美基研究(慕尼黑)公司 | 與cd3結合的多肽構建體 |
| EP4243936A1 (de) | 2020-11-10 | 2023-09-20 | Amgen Inc. | Verfahren zur verabreichung eines bcma x cd3-bindenden moleküls |
| MX2023005386A (es) | 2020-11-11 | 2023-05-23 | BioNTech SE | Anticuerpos monoclonales dirigidos contra la proteina de muerte programada-1 y su uso en medicina. |
| WO2022116877A1 (en) | 2020-12-02 | 2022-06-09 | Shanghai Henlius Biotech, Inc. | ANTI-GARP/TGFβ ANTIBODIES AND METHODS OF USE |
| TW202237068A (zh) | 2020-12-09 | 2022-10-01 | 美商建南德克公司 | 製備脂質奈米顆粒的高通量方法及其用途 |
| DE102020134181A1 (de) | 2020-12-18 | 2022-06-23 | Henke-Sass, Wolf Gmbh | Vorrichtung zum Applizieren eines Fluids |
| CN114685669A (zh) | 2020-12-30 | 2022-07-01 | 和铂医药(苏州)有限公司 | 结合trop2的抗体及其用途 |
| WO2022155324A1 (en) | 2021-01-15 | 2022-07-21 | The Rockefeller University | Neutralizing anti-sars-cov-2 antibodies |
| US12268847B1 (en) | 2021-02-10 | 2025-04-08 | Kaleo, Inc. | Devices and methods for delivery of substances within a medicament container |
| US12433847B2 (en) | 2021-03-09 | 2025-10-07 | Massachusetts Institute Of Technology | Branched poly(-amino esters) for the delivery of nucleic acids |
| JP2024510207A (ja) | 2021-03-12 | 2024-03-06 | ブリストル-マイヤーズ スクイブ カンパニー | Kras阻害剤 |
| KR20230160321A (ko) | 2021-03-24 | 2023-11-23 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| CA3214853A1 (en) | 2021-04-09 | 2022-10-13 | Celidex Therapeutics, Inc. | Antibodies against ilt4, bispecific anti-ilt4/pd-l1 antibody and uses thereof |
| WO2022221284A2 (en) | 2021-04-12 | 2022-10-20 | Bristol-Myers Squibb Company | Cyclic peptide immunomodulators |
| JP2024518369A (ja) | 2021-05-06 | 2024-05-01 | アムジェン リサーチ (ミュニック) ゲゼルシャフト ミット ベシュレンクテル ハフツング | 増殖性疾患で使用されるcd20及びcd22標的化抗原結合分子 |
| EP4334343A2 (de) | 2021-05-06 | 2024-03-13 | The Rockefeller University | Neutralisierende anti-sars-cov-2-antikörper und verfahren zur verwendung davon |
| EP4333805A1 (de) | 2021-05-06 | 2024-03-13 | Amgen Inc. | Etelcalcetidformulierungen |
| JP2024517903A (ja) | 2021-05-10 | 2024-04-23 | イッサム リサーチ ディベロップメント カンパニー オブ ザ ヘブライ ユニバーシティー オブ エルサレム リミテッド | 神経学的状態を治療するための薬学的組成物 |
| CN117769434A (zh) | 2021-05-20 | 2024-03-26 | 黛安瑟斯医疗运营公司 | 与C1s结合的抗体和其用途 |
| AU2022282609A1 (en) | 2021-05-26 | 2023-11-30 | Oxford Biotherapeutics Ltd | Pharmaceutical combination comprising an anti-cd205 antibody and an immune checkpoint inhibitor |
| TW202313065A (zh) | 2021-05-28 | 2023-04-01 | 美商季卡尼醫療公司 | 用於治療遺傳疾病之化合物 |
| JP2024526072A (ja) | 2021-06-09 | 2024-07-17 | ブリストル-マイヤーズ スクイブ カンパニー | 環状ペプチド免疫調節剤 |
| WO2022258792A1 (en) | 2021-06-09 | 2022-12-15 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Novel anti-fibrotic drugs |
| AU2022304662A1 (en) | 2021-06-30 | 2023-11-23 | Amgen Inc. | Method of reconstituting lyophilized formulation |
| US20240360178A1 (en) | 2021-07-12 | 2024-10-31 | Bristol-Myers Squibb Company | Macrocyclic immunomodulators |
| WO2023004240A1 (en) | 2021-07-19 | 2023-01-26 | Bristol-Myers Squibb Company | Macrocylic immunomodulators |
| AU2022316209A1 (en) | 2021-07-20 | 2024-02-22 | Regeneron Pharmaceuticals, Inc. | Butyrophilin-like 2 agents for treating inflammatory disorders |
| CN113398365B (zh) * | 2021-07-21 | 2022-10-28 | 南京瑞淇卓越医疗美容诊所有限公司 | 一种心血管内科用的临床造影注射装置及注射方法 |
| EP4384219A4 (de) | 2021-08-10 | 2025-06-18 | Viridian Therapeutics, Inc. | Zusammensetzungen, dosen und verfahren zur behandlung von schilddrüsenaugenkrankheit |
| CN118176204A (zh) | 2021-08-11 | 2024-06-11 | 圣诺菲·帕斯图尔公司 | 截短的流感神经氨酸酶及其使用方法 |
| JP2024532158A (ja) | 2021-08-20 | 2024-09-05 | インターベット インターナショナル ベー. フェー. | アトピー性皮膚炎を治療するためのホモ二量体融合タンパク質 |
| AU2022329459A1 (en) | 2021-08-20 | 2024-02-29 | Intervet International B.V. | Antibodies and igg fusion proteins with an extended half-life |
| WO2023039108A1 (en) | 2021-09-08 | 2023-03-16 | Affinivax, Inc. | Coronavirus vaccine |
| CN113773401B (zh) | 2021-09-15 | 2023-06-20 | 宜明昂科生物医药技术(上海)股份有限公司 | 靶向cd47和pd-l1的重组融合蛋白及其制备和用途 |
| WO2023056240A2 (en) | 2021-09-28 | 2023-04-06 | Frontaim Biomedicines, Inc. | Multiple formats of molecular complexes |
| AU2022361432A1 (en) | 2021-10-08 | 2024-05-23 | Sanofi Pasteur Inc. | Multivalent influenza vaccines |
| CN113956363B (zh) | 2021-10-13 | 2023-03-31 | 宜明昂科生物医药技术(上海)股份有限公司 | 靶向cd47和cd24的重组融合蛋白及其制备和用途 |
| CN113754769B (zh) | 2021-10-13 | 2023-06-06 | 宜明昂科生物医药技术(上海)股份有限公司 | 靶向cd70的抗体及其制备和用途 |
| CN113831412B (zh) | 2021-10-13 | 2023-06-20 | 宜明昂科生物医药技术(上海)股份有限公司 | 靶向cd24的抗体及其制备和用途 |
| AU2022364888A1 (en) | 2021-10-14 | 2024-05-30 | Latticon (Suzhou) Biopharmaceuticals Co., Ltd. | Novel antibody-cytokine fusion protein, preparation method therefor and use thereof |
| KR20240089796A (ko) | 2021-10-20 | 2024-06-20 | 브리스톨-마이어스 스큅 컴퍼니 | 면역조정제 |
| US20260070986A1 (en) | 2021-11-03 | 2026-03-12 | Affimed Gmbh | Bispecific cd16a binders |
| IL312515A (en) | 2021-11-03 | 2024-07-01 | Affimed Gmbh | Bispecific cd16a binders |
| AU2022379948A1 (en) | 2021-11-05 | 2024-06-20 | Sanofi Pasteur Inc. | Multivalent influenza vaccines comprising recombinant hemagglutinin and neuraminidase and methods of using the same |
| WO2023079113A1 (en) | 2021-11-05 | 2023-05-11 | Sanofi | Hybrid multivalent influenza vaccines comprising hemagglutinin and neuraminidase and methods of using the same |
| WO2023077521A1 (en) | 2021-11-08 | 2023-05-11 | Celldex Therapeutics, Inc | Anti-ilt4 and anti-pd-1 bispecific constructs |
| US20250073346A1 (en) | 2021-11-18 | 2025-03-06 | Oxford Bio Therapeutics Ltd | Pharmaceutical combinations |
| WO2023096904A2 (en) | 2021-11-24 | 2023-06-01 | President And Fellows Of Harvard College | C-16 modified trioxacarcins, antibody drug conjugates, and uses thereof |
| JP2024544009A (ja) | 2021-12-03 | 2024-11-26 | ブリストル-マイヤーズ スクイブ カンパニー | 大員環免疫調節剤 |
| US12171779B2 (en) | 2021-12-08 | 2024-12-24 | University Of South Florida | Regulated drug delivery via controlled degradable chondroitin sulfate particles |
| WO2023105281A1 (en) | 2021-12-11 | 2023-06-15 | Fundaciò Privada Institut De Recerca De La Sida-Caixa | Soluble tigit recombinant proteins |
| IL313473A (en) | 2021-12-15 | 2024-08-01 | Interius Biotherapeutics Inc | Pseudotyped viral particles, compositions comprising the same, and uses thereof |
| MX2024007090A (es) | 2021-12-16 | 2024-08-22 | Intervet Int Bv | Anticuerpos caninizados para el receptor alfa ii de interleucina-31 canino. |
| CA3240904A1 (en) | 2021-12-16 | 2023-06-22 | Intervet International B.V. | Caninized and felinized antibodies to human ngf |
| WO2023114889A1 (en) | 2021-12-16 | 2023-06-22 | Modernatx, Inc. | Processes for preparing lipid nanoparticles |
| KR20240122784A (ko) | 2021-12-17 | 2024-08-13 | 상하이 헨리우스 바이오테크, 인크. | 항-ox40 항체 및 사용 방법 |
| KR20240116755A (ko) | 2021-12-17 | 2024-07-30 | 상하이 헨리우스 바이오테크, 인크. | 항-ox40 항체, 다중 특이적 항체 및 이의 사용 방법 |
| US20250084041A1 (en) | 2021-12-30 | 2025-03-13 | Curza Global, Llc | Antimicrobial compounds and methods |
| JPWO2023145735A1 (de) | 2022-01-25 | 2023-08-03 | ||
| US20250333485A1 (en) | 2022-01-27 | 2025-10-30 | The Rockefeller University | Broadly neutralizing anti-sars-cov-2 antibodies targeting the n-terminal domain of the spike protein and methods of use thereof |
| JP2025503207A (ja) | 2022-01-27 | 2025-01-30 | サノフィ・パスツール | 修飾されたVero細胞、及びウイルス産生にこれを使用する方法 |
| AU2023223413A1 (en) | 2022-02-24 | 2024-10-10 | Sinomab Bioscience Limited | Bispecific binding proteins against alarmins and uses thereof |
| CN119522229A (zh) | 2022-03-04 | 2025-02-25 | 瑞瓦吉尼克斯股份有限公司 | 广谱氨基糖苷及其用途 |
| EP4494146A1 (de) | 2022-03-14 | 2025-01-22 | Sanofi Pasteur, Inc. | Maschinenlernverfahren beim proteinentwurf zur impfstofferzeugung |
| WO2023173393A1 (zh) | 2022-03-18 | 2023-09-21 | 北京天广实生物技术股份有限公司 | 结合b7-h3的抗体及其用途 |
| WO2023175171A1 (en) | 2022-03-18 | 2023-09-21 | Inserm (Institut National De La Sante Et De La Recherche Medicale) | Bk polyomavirus antibodies and uses thereof |
| CA3245762A1 (en) | 2022-03-25 | 2023-09-28 | Shanghai Henlius Biologics Co., Ltd. | ANTI-MSLN ANTIBODIES AND METHODS OF USE |
| US20250250299A1 (en) | 2022-03-28 | 2025-08-07 | Bristol-Myers Squibb Company | Macrocyclic immunomodulators |
| US20250214046A1 (en) | 2022-04-01 | 2025-07-03 | Modernatx, Inc. | Cross mixers for lipid nanoparticle production, and methods of operating the same |
| EP4257132A1 (de) | 2022-04-08 | 2023-10-11 | iOmx Therapeutics AG | Sik3-inhibitoren zum behandeln von krankheiten, die gegen death-rezeptor -signaltransduktion resistent sind |
| EP4257609A1 (de) | 2022-04-08 | 2023-10-11 | iOmx Therapeutics AG | Kombinationstherapien basierend auf pd-1-inhibitoren und sik3-inhibitoren |
| AU2023259350A1 (en) | 2022-04-26 | 2024-10-17 | Amgen Inc. | Lyophilization method |
| EP4522651A1 (de) | 2022-05-12 | 2025-03-19 | Amgen Research (Munich) GmbH | Mehrkettige, multitargeting, bispezifische antigenbindende moleküle mit erhöhter selektivität |
| EP4522654A1 (de) | 2022-05-12 | 2025-03-19 | BioNTech SE | Monoklonale antikörper gegen protein mit programmiertem tod-1 und ihre verwendung in der medizin |
| GB202207263D0 (en) | 2022-05-18 | 2022-06-29 | Annexin Pharmaceuticals Ab | Therapeutic compositions |
| US20260022142A1 (en) | 2022-05-20 | 2026-01-22 | Bristol-Myers Squibb Company | Macrocyclic immunomodulators |
| CN117186238A (zh) | 2022-05-31 | 2023-12-08 | 宜明昂科生物医药技术(上海)股份有限公司 | 靶向pd-l1和vegf的重组融合蛋白及其制备和用途 |
| WO2023235380A1 (en) | 2022-06-01 | 2023-12-07 | Zikani Therapeutics, Inc. | Macrolides for treating genetic diseases |
| CN119630676A (zh) | 2022-06-10 | 2025-03-14 | 百时美施贵宝公司 | 作为kras抑制剂的吡啶并[4,3-d]嘧啶衍生物 |
| CN119317629A (zh) | 2022-06-10 | 2025-01-14 | 百时美施贵宝公司 | 作为kras抑制剂的四氢吡啶并3,4-d嘧啶衍生物 |
| WO2023250513A1 (en) | 2022-06-24 | 2023-12-28 | Zikani Therapeutics, Inc. | 13-membered macrolide compounds for treating diseases mediated by abnormal protein translation |
| KR20250057777A (ko) | 2022-07-25 | 2025-04-29 | 인테리우스 바이오테라퓨틱스, 인코포레이티드 | 돌연변이된 폴리펩티드, 이를 포함하는 조성물, 및 그 용도(mutated polypeptides, compositions comprising the same, and uses thereof) |
| WO2024026482A1 (en) | 2022-07-29 | 2024-02-01 | Modernatx, Inc. | Lipid nanoparticle compositions comprising surface lipid derivatives and related uses |
| US20250375389A1 (en) | 2022-07-29 | 2025-12-11 | Modernatx, Inc. | Lipid nanoparticle compositions comprising phospholipid derivatives and related uses |
| WO2024026475A1 (en) | 2022-07-29 | 2024-02-01 | Modernatx, Inc. | Compositions for delivery to hematopoietic stem and progenitor cells (hspcs) and related uses |
| JP2025526744A (ja) | 2022-08-11 | 2025-08-15 | ブリストル-マイヤーズ スクイブ カンパニー | Kras阻害剤 |
| JP2025527530A (ja) | 2022-08-16 | 2025-08-22 | ブリストル-マイヤーズ スクイブ カンパニー | Kras阻害剤 |
| CN120152721A (zh) | 2022-09-01 | 2025-06-13 | 齐卡尼治疗股份有限公司 | 使用13元大环内酯治疗家族性腺瘤性息肉病 |
| EP4584276A1 (de) | 2022-09-07 | 2025-07-16 | Bristol-Myers Squibb Company | Kras-g12d-hemmer |
| US20260070945A1 (en) | 2022-09-12 | 2026-03-12 | Bristol-Myers Squibb Company | Macrocyclic immunomodulators |
| TW202421650A (zh) | 2022-09-14 | 2024-06-01 | 美商安進公司 | 雙特異性分子穩定組成物 |
| WO2024091918A2 (en) | 2022-10-25 | 2024-05-02 | Modernatx, Inc. | Methods of lipid nanoparticle production in cross-mixers |
| WO2024112734A1 (en) | 2022-11-21 | 2024-05-30 | Dianthus Therapeutics Opco, Inc. | Antibodies that bind to c1s and uses thereof |
| CN115737992B (zh) * | 2022-11-24 | 2024-06-07 | 深圳市本尚医疗科技有限公司 | 无针注射器 |
| EP4626863A1 (de) | 2022-11-30 | 2025-10-08 | The Trustees of The University of Pennsylvania | Inhibitoren des brisc-deubiquitinierenden enzyms |
| WO2024121380A1 (en) | 2022-12-08 | 2024-06-13 | Pierre Fabre Medicament | Vaccinal composition and adjuvant |
| GB202219154D0 (en) | 2022-12-19 | 2023-02-01 | Metacurum Biotech Ab | Antibodies and uses thereof |
| US11806334B1 (en) | 2023-01-12 | 2023-11-07 | Bioxcel Therapeutics, Inc. | Non-sedating dexmedetomidine treatment regimens |
| EP4658687A1 (de) | 2023-01-31 | 2025-12-10 | University of Rochester | Immuncheckpoint-blockadetherapie zur behandlung von staphylococcus-aureus-infektionen |
| EP4665756A1 (de) | 2023-02-13 | 2025-12-24 | Intervet International B.V. | Antikörper gegen hunde-il-4 |
| JP2026508149A (ja) | 2023-02-13 | 2026-03-10 | インターベット インターナショナル ベー. フェー. | イヌil-13に対するイヌ抗体 |
| KR20250167153A (ko) | 2023-03-03 | 2025-11-28 | 셀덱스 쎄라퓨틱스, 인크. | 항-줄기 세포 인자(scf) 및 항-흉선 기질 림포포이에틴(tslp) 항체, 및 이중특이적 구축물 |
| KR20250157375A (ko) | 2023-03-08 | 2025-11-04 | 암젠 인크 | 이중특이적 분자에 대한 제어된 빙핵 형성 동결건조 공정 |
| JP2026510891A (ja) | 2023-03-16 | 2026-04-10 | インマージーン プライベート リミテッド | Ilt7標的化抗体及びそれらの使用 |
| CN121263432A (zh) | 2023-04-12 | 2026-01-02 | 上海康抗生物技术有限公司 | 包含掩蔽型白介素12的多功能分子及使用方法 |
| WO2024218322A1 (en) | 2023-04-21 | 2024-10-24 | Helmholtz Zentrum München Deutsches Forschungszentrum Für Gesundheit Und Umwelt (Gmbh) | Ferroptosis inhibitors |
| EP4705319A1 (de) | 2023-05-04 | 2026-03-11 | Affinivax, Inc. | Rhizavidinvarianten |
| CN121194795A (zh) | 2023-05-18 | 2025-12-23 | 安尼欣制药有限公司 | 膜联蛋白a5蛋白质与抗vegf药剂作为组合疗法用于治疗黄斑水肿或视网膜静脉阻塞 |
| CN121241137A (zh) | 2023-05-24 | 2025-12-30 | 南京金斯瑞生物科技有限公司 | 用于制备环状rna的载体和方法 |
| CN121620357A (zh) | 2023-06-01 | 2026-03-06 | 赛诺菲巴斯德有限公司 | 包含mRNA脂质纳米颗粒的热稳定组合物 |
| EP4725969A1 (de) | 2023-06-09 | 2026-04-15 | Latticon (Suzhou) Biopharmaceuticals Co., Ltd. | Komplementäres bispezifisches anti-her2-antikörper-wirkstoff-konjugat, herstellungsverfahren dafür und verwendung davon |
| WO2024255753A1 (zh) | 2023-06-12 | 2024-12-19 | 南京蓬勃生物科技有限公司 | 结合人ccr8的抗体及其用途 |
| UY40797A (es) | 2023-06-14 | 2024-12-31 | Amgen Inc | Moléculas captadoras de enmascaramiento de células t |
| WO2024259373A1 (en) | 2023-06-14 | 2024-12-19 | Modernatx, Inc. | Compounds and compositions for delivery of therapeutic agents |
| IL324670A (en) | 2023-06-30 | 2026-01-01 | Bristol Myers Squibb Co | KRAS inhibitors |
| AU2024287547A1 (en) | 2023-07-07 | 2026-01-22 | Viridian Therapeutics, Inc. | Methods of treating chronic thyroid eye disease |
| WO2025046492A1 (en) | 2023-08-31 | 2025-03-06 | Revagenix, Inc. | Broad spectrum aminoglycosides and uses thereof |
| WO2025051975A1 (en) | 2023-09-06 | 2025-03-13 | Sanofi | Modified influenza b hemagglutinin polypeptides and nucleic acids and uses thereof |
| TW202519262A (zh) | 2023-09-06 | 2025-05-16 | 美商維里迪恩醫療股份有限公司 | 抗igf-1r抗體之醫藥組合物 |
| WO2025082594A1 (en) | 2023-10-17 | 2025-04-24 | BioNTech SE | Antigen binding proteins specifically binding to cd3 and their use in medicine |
| AU2024364781A1 (en) | 2023-10-17 | 2025-04-24 | BioNTech SE | Antigen binding proteins specifically binding to cd3 and their use in medicine |
| WO2025122619A1 (en) | 2023-12-06 | 2025-06-12 | Bristol-Myers Squibb Company | Kras g12c inhibitors |
| WO2025136985A1 (en) | 2023-12-17 | 2025-06-26 | Viridian Therapeutics, Inc. | Compositions, doses, and methods for treatment of thyroid eye disease |
| WO2025151502A1 (en) | 2024-01-09 | 2025-07-17 | Viridian Therapeutics, Inc. | Modified antibodies |
| WO2025151492A1 (en) | 2024-01-09 | 2025-07-17 | Viridian Therapeutics, Inc. | Compositions and methods for treatment of thyroid eye disease |
| WO2025151496A1 (en) | 2024-01-09 | 2025-07-17 | Viridian Therapeutics, Inc. | Compositions and methods for treatment of thyroid eye disease |
| WO2025157389A1 (en) | 2024-01-22 | 2025-07-31 | Iomx Therapeutics Ag | Combinations of halogenated heterocyclic kinase inhibitors and vegfr inhibitors |
| WO2025160127A1 (en) | 2024-01-23 | 2025-07-31 | Modernatx, Inc. | Ionizable lipids and compositions thereof for delivery of therapeutic agents |
| WO2025160381A1 (en) | 2024-01-26 | 2025-07-31 | Modernatx, Inc. | Methods of preparing lipid nanoparticles |
| WO2025166202A1 (en) | 2024-01-31 | 2025-08-07 | Modernatx, Inc. | Lipid nanoparticle compositions comprising sialic acid derivatives and the uses thereof |
| WO2025168716A1 (en) | 2024-02-07 | 2025-08-14 | Eyebiotech Limited | Compositions, doses, and methods for treatment of ocular diseases |
| US20250281608A1 (en) | 2024-03-05 | 2025-09-11 | Zenas Biopharma, Inc. | STABLE FORMULATIONS FOR TNF-alpha ANTIBODIES AND USES THEREOF |
| WO2025193574A1 (en) | 2024-03-11 | 2025-09-18 | Bristol-Myers Squibb Company | Macrocyclic peptides useful as immunomodulators |
| WO2025193583A1 (en) | 2024-03-11 | 2025-09-18 | Bristol-Myers Squibb Company | Macrocyclic peptides useful as immunomodulators |
| WO2025193573A1 (en) | 2024-03-11 | 2025-09-18 | Bristol-Myers Squibb Company | Macrocyclic peptides useful as immunomodulators |
| WO2025193571A1 (en) | 2024-03-11 | 2025-09-18 | Bristol-Myers Squibb Company | Macrocyclic peptides useful as immunomodulators |
| WO2025193903A1 (en) | 2024-03-15 | 2025-09-18 | Affinivax, Inc. | Pneumococcal polysaccharide compositions and uses thereof |
| EP4643861A1 (de) | 2024-04-30 | 2025-11-05 | iOmx Therapeutics AG | Halogenierter heteroarylkinasehemmer zur behandlung von weichgewebesarkom und gefässtumoren |
| WO2025229127A1 (en) | 2024-04-30 | 2025-11-06 | Iomx Therapeutics Ag | A halogenated-heteroaryl kinase inhibitor for treating soft tissue sarcoma and vascular tumors |
| WO2025242910A1 (en) | 2024-05-23 | 2025-11-27 | Egle Therapeutics | Il18r agonist antibodies and uses thereof |
| EP4653460A1 (de) | 2024-05-23 | 2025-11-26 | Egle Therapeutics | Il18r-agonist-antikörper und verwendungen davon |
| WO2025257588A1 (en) | 2024-06-10 | 2025-12-18 | Affimed Gmbh | Cd16a/tumor antigen polyspecific binder for use in the treatment of immune checkpoint inhibitor resistance |
| EP4670733A1 (de) | 2024-06-27 | 2025-12-31 | Sanofi | Verfahren zur herstellung von viralen proteinen und deren verwendungen |
| WO2026027944A1 (en) | 2024-07-30 | 2026-02-05 | Sairopa B.V. | Anti-sirp alpha antibody formulations and uses thereof |
| WO2026027730A1 (en) | 2024-07-31 | 2026-02-05 | Sanofi | Modified h5 influenza hemagglutinin polypeptides and nucleic acids and uses thereof |
| WO2026039512A1 (en) | 2024-08-14 | 2026-02-19 | Bristol-Myers Squibb Company | Kras inhibitors |
| WO2026044176A1 (en) | 2024-08-23 | 2026-02-26 | University Of Massachusetts | Anti-etec adhesin protein antibodies and methods of use |
| WO2026052773A1 (en) | 2024-09-06 | 2026-03-12 | Sanofi | Modified influenza a hemagglutinin polypeptides and nucleic acids and uses thereof |
| WO2026064599A1 (en) | 2024-09-19 | 2026-03-26 | Zenas Biopharma, Inc. | Stable formulations for ctla4-ig fusion proteins and uses thereof |
Family Cites Families (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2704543A (en) * | 1955-03-22 | Hypo jet injector | ||
| US2101140A (en) * | 1935-03-23 | 1937-12-07 | Raymond W Hege | Automatic injecting syringe device |
| US2380534A (en) * | 1941-04-26 | 1945-07-31 | Marshall L Lockhart | Hypodermic injector |
| US2605763A (en) * | 1948-01-31 | 1952-08-05 | Becton Dickinson Co | Injection device |
| US2547099A (en) * | 1948-03-11 | 1951-04-03 | Becton Dickinson Co | Injection device and ampoule |
| US2680439A (en) * | 1948-09-08 | 1954-06-08 | Arnold K Sutermeister | High-pressure injection device |
| US2670121A (en) * | 1951-01-31 | 1954-02-23 | Scherer Corp R P | Flexible follower for metal ampoules |
| US2816543A (en) * | 1954-07-12 | 1957-12-17 | Scherer Corp R P | Hypodermic injector |
| NL133848C (de) * | 1960-08-15 | |||
| US3292621A (en) * | 1963-07-19 | 1966-12-20 | Oscar H Banker | Jet type protable inoculator |
| US3292622A (en) * | 1964-09-21 | 1966-12-20 | Oscar H Banker | Power operated inoculator |
| US3424154A (en) * | 1965-11-08 | 1969-01-28 | Charles W Kinsley | Injection system |
| US3561443A (en) * | 1968-09-06 | 1971-02-09 | Oscar H Banker | Inoculator gun with delayed action |
| US3688765A (en) * | 1969-10-03 | 1972-09-05 | Jack S Gasaway | Hypodermic injection device |
| US3945379A (en) * | 1974-08-08 | 1976-03-23 | Smithkline Corporation | Injection device |
| JPS51130094A (en) * | 1975-05-08 | 1976-11-12 | Asahi Chemical Ind | Twoostage pressure injector |
| CA1178503A (en) * | 1982-05-27 | 1984-11-27 | Health-Mor Personal Care Corporation | Needleless hypodermic injector |
| HU186718B (en) * | 1983-06-29 | 1985-09-30 | Radelkis Electrokemiai | Automatic needleless syringe operable by cardox cylinder suitable for personal giving injection e.g. insulin |
| US4596556A (en) * | 1985-03-25 | 1986-06-24 | Bioject, Inc. | Hypodermic injection apparatus |
| US4680027A (en) * | 1985-12-12 | 1987-07-14 | Injet Medical Products, Inc. | Needleless hypodermic injection device |
| EP0250022A3 (de) * | 1986-06-17 | 1988-06-22 | SICIM SpA | Tragbarer hypodermatischer Injektor |
| US4722728A (en) * | 1987-01-23 | 1988-02-02 | Patents Unlimited, Ltd. | Needleless hypodermic injector |
| US4940460A (en) * | 1987-06-19 | 1990-07-10 | Bioject, Inc. | Patient-fillable and non-invasive hypodermic injection device assembly |
| US4941880A (en) * | 1987-06-19 | 1990-07-17 | Bioject, Inc. | Pre-filled ampule and non-invasive hypodermic injection device assembly |
| US4790824A (en) * | 1987-06-19 | 1988-12-13 | Bioject, Inc. | Non-invasive hypodermic injection device |
| DE3814023A1 (de) * | 1987-07-13 | 1989-01-26 | Medizin Labortechnik Veb K | Injektionsvorrichtung mit medikamentendepot |
| CA1310870C (en) * | 1987-11-16 | 1992-12-01 | Medhi-Tek International Limited | Apparatus for hypodermic injection of liquids |
| US5073165A (en) * | 1988-02-05 | 1991-12-17 | Marpam International, Inc. | Hypodermic jet injector and cartridge therefor |
| US4913699A (en) * | 1988-03-14 | 1990-04-03 | Parsons James S | Disposable needleless injection system |
| US5024656A (en) * | 1988-08-30 | 1991-06-18 | Injet Medical Products, Inc. | Gas-pressure-regulated needleless injection system |
| CA1325149C (en) * | 1989-08-31 | 1993-12-14 | Gavin Mcgregor | Variable intensity remote controlled needleless injector |
| US5064413A (en) * | 1989-11-09 | 1991-11-12 | Bioject, Inc. | Needleless hypodermic injection device |
| US5062830A (en) * | 1990-04-04 | 1991-11-05 | Derata Corporation | Dry disposable nozzle assembly for medical jet injector |
| US5383851A (en) * | 1992-07-24 | 1995-01-24 | Bioject Inc. | Needleless hypodermic injection device |
-
1992
- 1992-07-24 US US07/920,106 patent/US5383851A/en not_active Expired - Lifetime
-
1993
- 1993-07-23 CA CA002140772A patent/CA2140772C/en not_active Expired - Lifetime
- 1993-07-23 AT AT93918344T patent/ATE187083T1/de not_active IP Right Cessation
- 1993-07-23 DE DE69327165T patent/DE69327165T2/de not_active Expired - Lifetime
- 1993-07-23 WO PCT/US1993/006940 patent/WO1994002188A1/en not_active Ceased
- 1993-07-23 JP JP50471494A patent/JP3633615B2/ja not_active Expired - Lifetime
- 1993-07-23 AU AU47827/93A patent/AU676490B2/en not_active Expired
- 1993-07-23 EP EP93918344A patent/EP0651663B1/de not_active Expired - Lifetime
- 1993-07-23 US US08/097,266 patent/US5399163A/en not_active Expired - Lifetime
-
1995
- 1995-03-21 US US08/407,867 patent/US5520639A/en not_active Expired - Lifetime
-
2003
- 2003-12-24 JP JP2003426219A patent/JP2004141667A/ja active Pending
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| USRE43824E1 (en) | 2001-01-11 | 2012-11-20 | Powder Pharmaceuticals Inc. | Needleless syringe |
| US8540665B2 (en) | 2007-05-04 | 2013-09-24 | Powder Pharmaceuticals Inc. | Particle cassettes and processes therefor |
| US9044546B2 (en) | 2007-05-04 | 2015-06-02 | Powder Pharmaceuticals Incorporated | Particle cassettes and processes therefor |
| US9358338B2 (en) | 2007-05-04 | 2016-06-07 | Powder Pharmaceuticals Incorporated | Particle cassettes and processes therefor |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0651663A4 (de) | 1995-08-30 |
| JP3633615B2 (ja) | 2005-03-30 |
| DE69327165T2 (de) | 2000-08-24 |
| CA2140772C (en) | 2006-07-11 |
| US5399163A (en) | 1995-03-21 |
| CA2140772A1 (en) | 1994-02-03 |
| EP0651663A1 (de) | 1995-05-10 |
| US5520639A (en) | 1996-05-28 |
| US5383851A (en) | 1995-01-24 |
| AU676490B2 (en) | 1997-03-13 |
| AU4782793A (en) | 1994-02-14 |
| JPH07509161A (ja) | 1995-10-12 |
| DE69327165D1 (de) | 2000-01-05 |
| WO1994002188A1 (en) | 1994-02-03 |
| ATE187083T1 (de) | 1999-12-15 |
| JP2004141667A (ja) | 2004-05-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0651663B1 (de) | Verfahren für nadellose subkutaninjektionen | |
| WO1994002188A9 (en) | Needleless hypodermic injection methods and device | |
| US10918791B2 (en) | Devices, systems and methods for medicament delivery | |
| EP0427457B1 (de) | Ampulle für nadelloses, hypodermisches Injektionsgerät | |
| US6939319B1 (en) | Process and device for single use, needle-free intradermal, subcutaneous, or intramuscular injections | |
| US8361029B2 (en) | Devices, systems and methods for medicament delivery | |
| US5312335A (en) | Needleless hypodermic injection device | |
| US4194505A (en) | Containerized hypodermic module | |
| US5049125A (en) | Needleless injection apparatus of a liquid, notably for dental care | |
| CA2476076C (en) | Injector with bypass channel | |
| US5024656A (en) | Gas-pressure-regulated needleless injection system | |
| AU675534B2 (en) | Multiple medication injection apparatus and method | |
| US4623332A (en) | Needleless jet injector | |
| WO2004101025A2 (en) | Needle free hypodermic injector and ampule for intradermal, subcutaneous and intramuscular injection | |
| US20020099329A1 (en) | Gas power sources for needle-less injector and needle-less injectors incorporating the same | |
| JP2003529417A (ja) | 1回限り使用の使い捨て型ジェット注射器 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19950223 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| A4 | Supplementary search report drawn up and despatched | ||
| AK | Designated contracting states |
Kind code of ref document: A4 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| 17Q | First examination report despatched |
Effective date: 19980507 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI LU MC NL PT SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19991201 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19991201 Ref country code: LI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19991201 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 19991201 Ref country code: GR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19991201 Ref country code: ES Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 19991201 Ref country code: CH Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19991201 Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19991201 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 19991201 |
|
| REF | Corresponds to: |
Ref document number: 187083 Country of ref document: AT Date of ref document: 19991215 Kind code of ref document: T |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REF | Corresponds to: |
Ref document number: 69327165 Country of ref document: DE Date of ref document: 20000105 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20000301 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20000302 |
|
| ET | Fr: translation filed | ||
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000723 Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20000723 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 20000731 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20120723 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20120809 Year of fee payment: 20 Ref country code: DE Payment date: 20120731 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R071 Ref document number: 69327165 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 Expiry date: 20130722 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20130724 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20130722 |