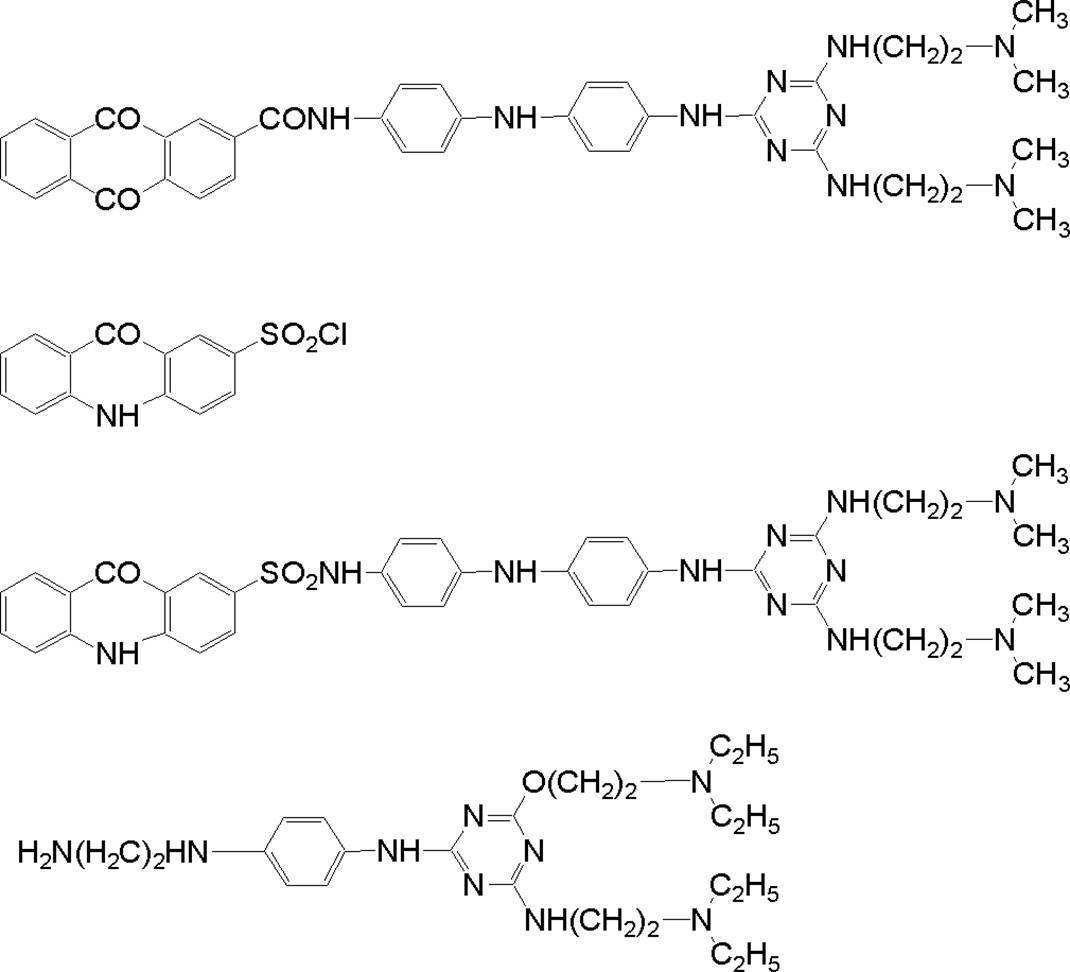
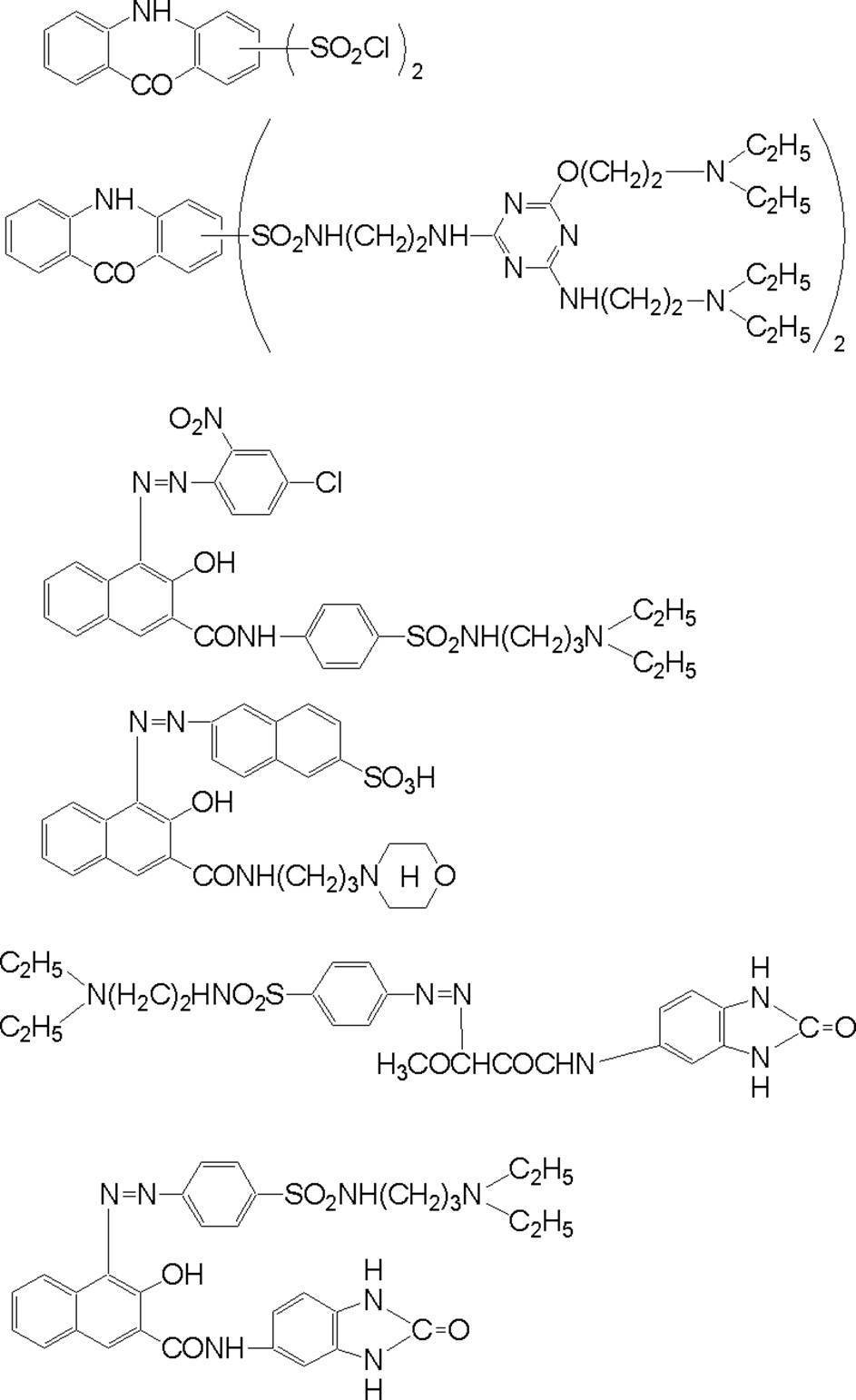
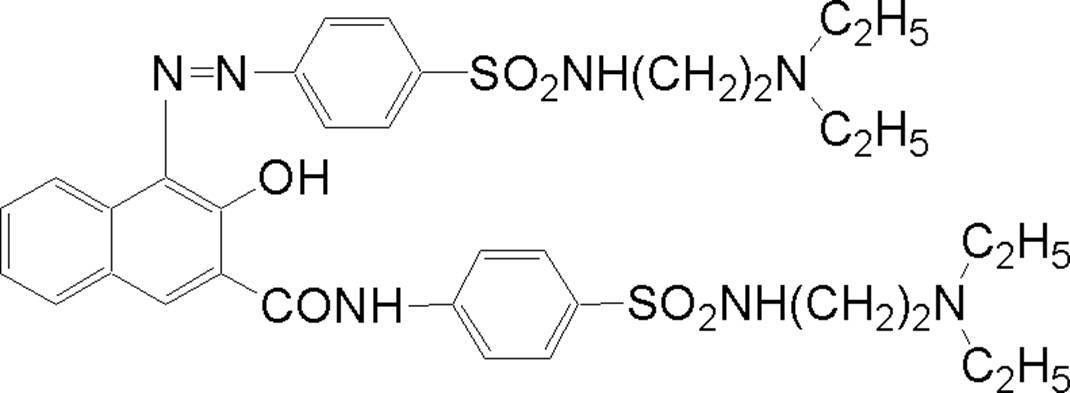
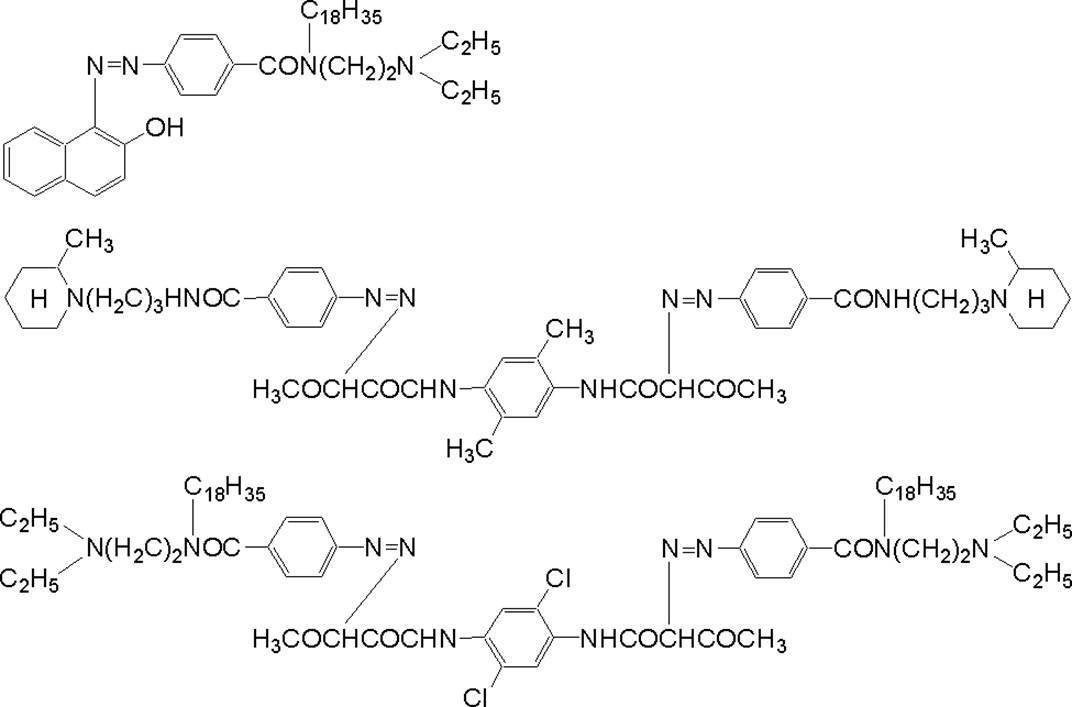
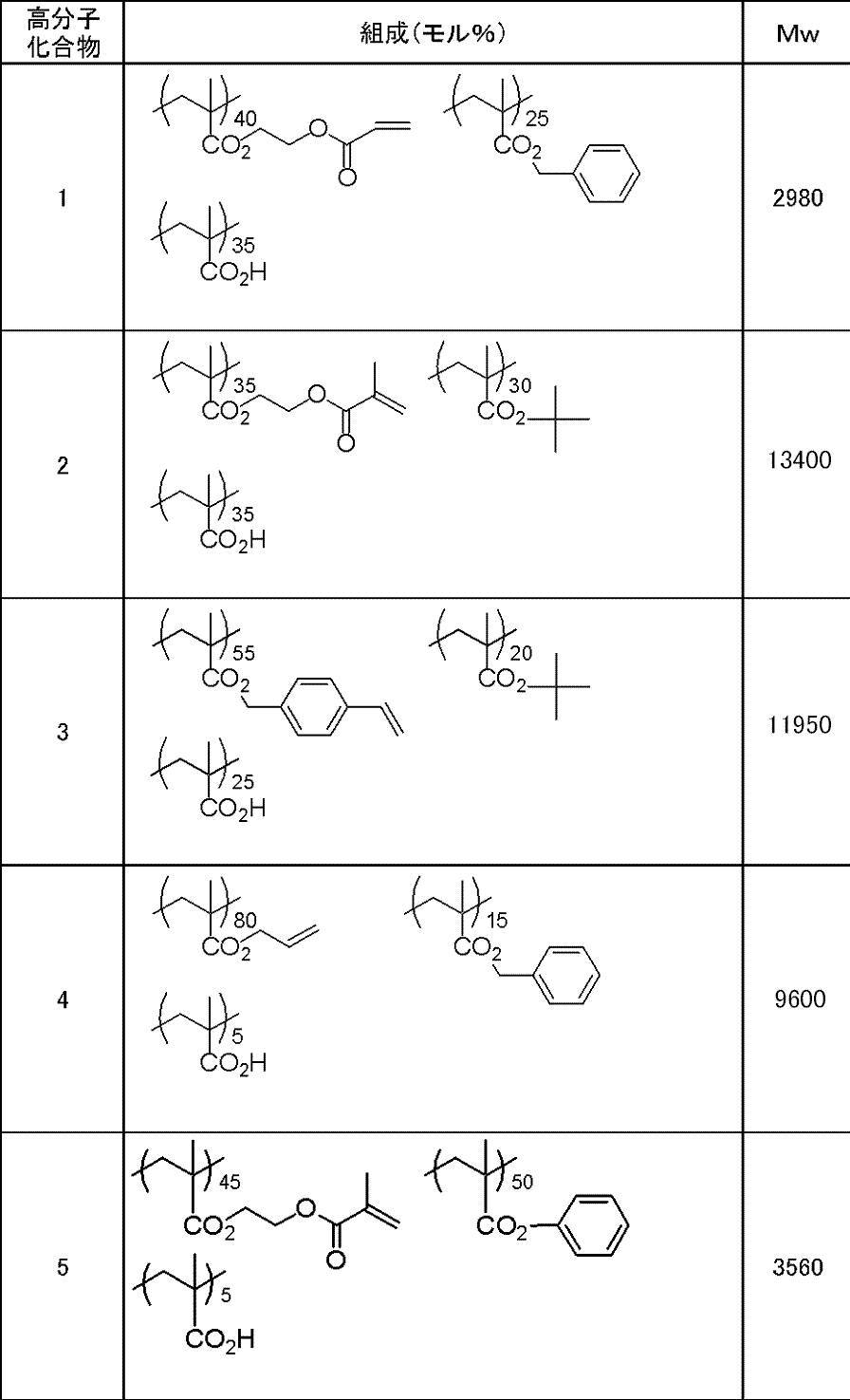
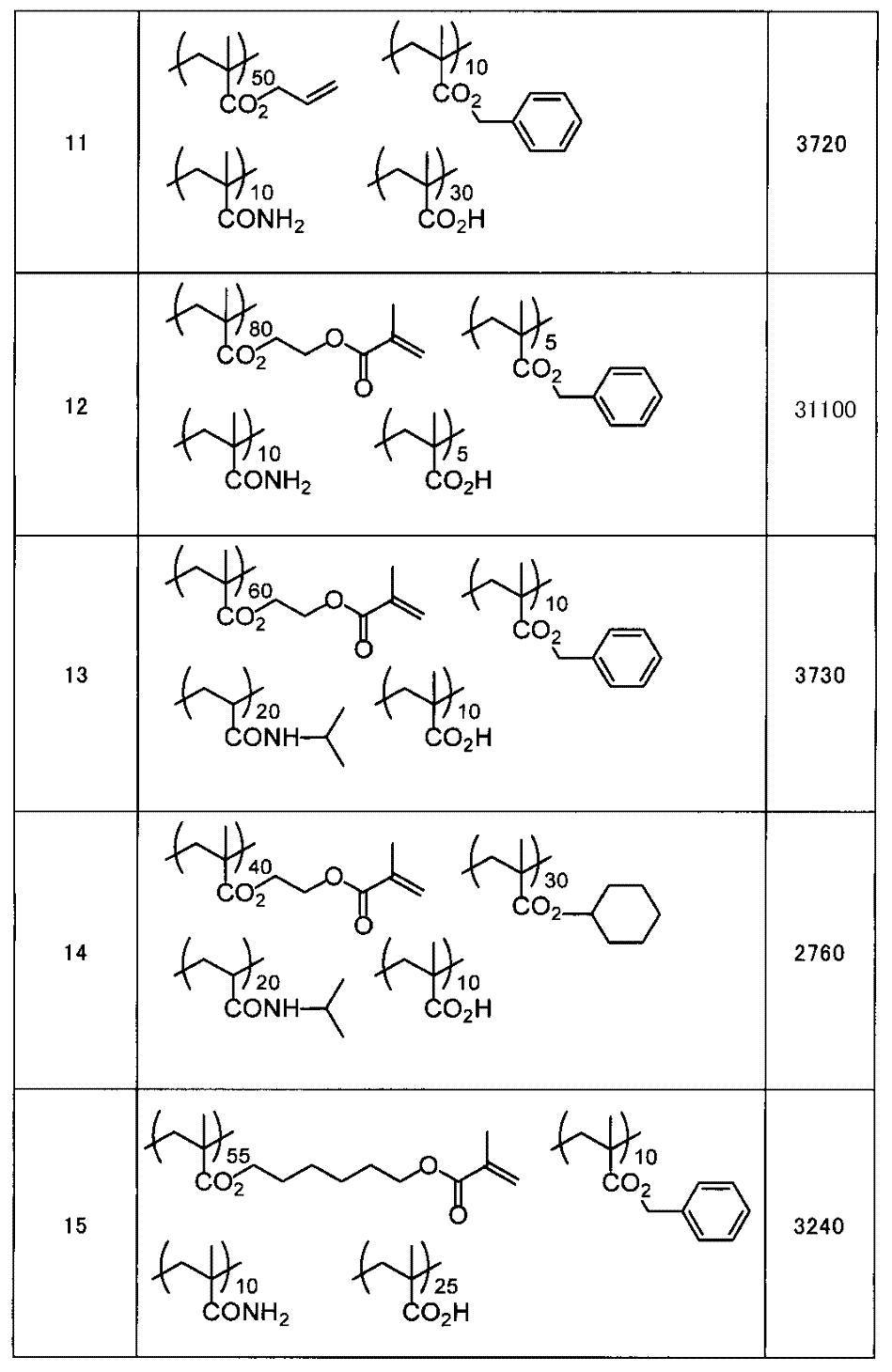
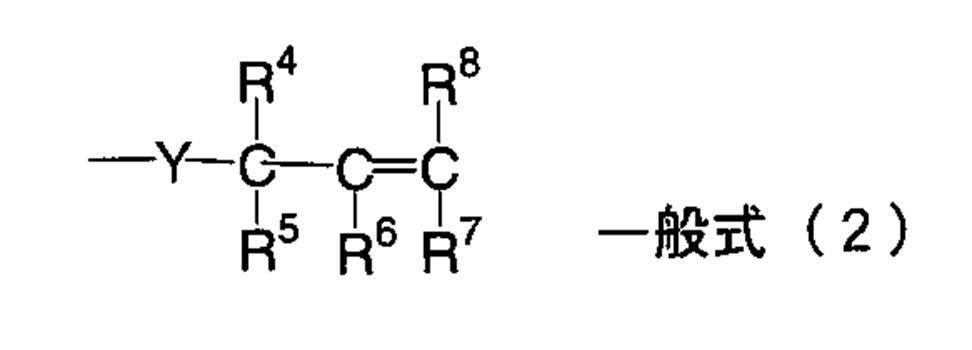
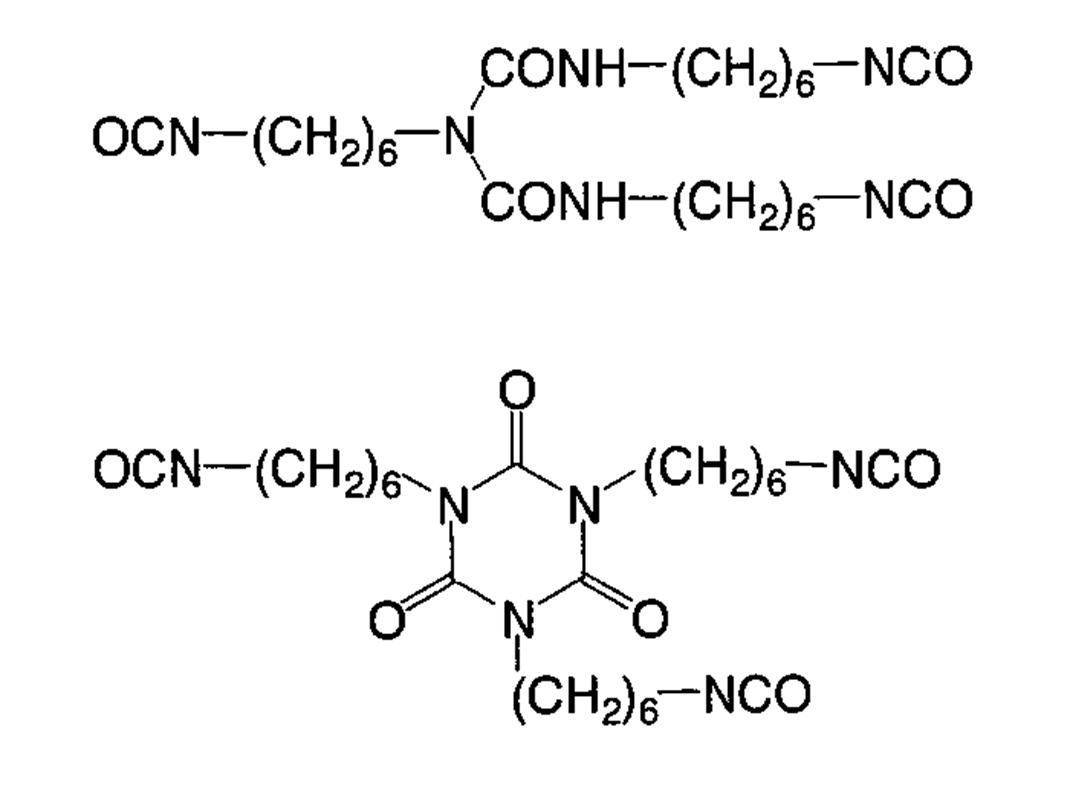
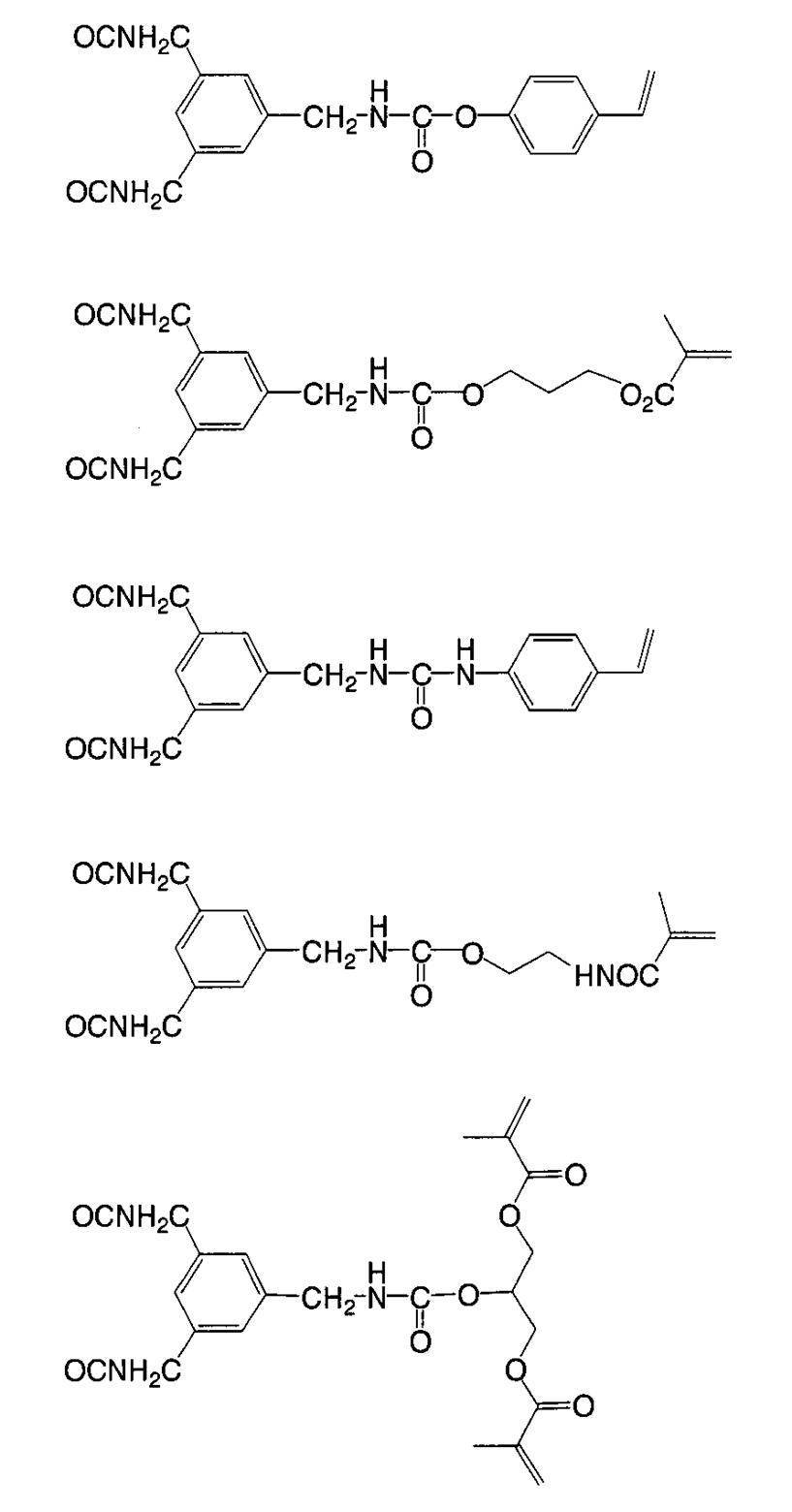
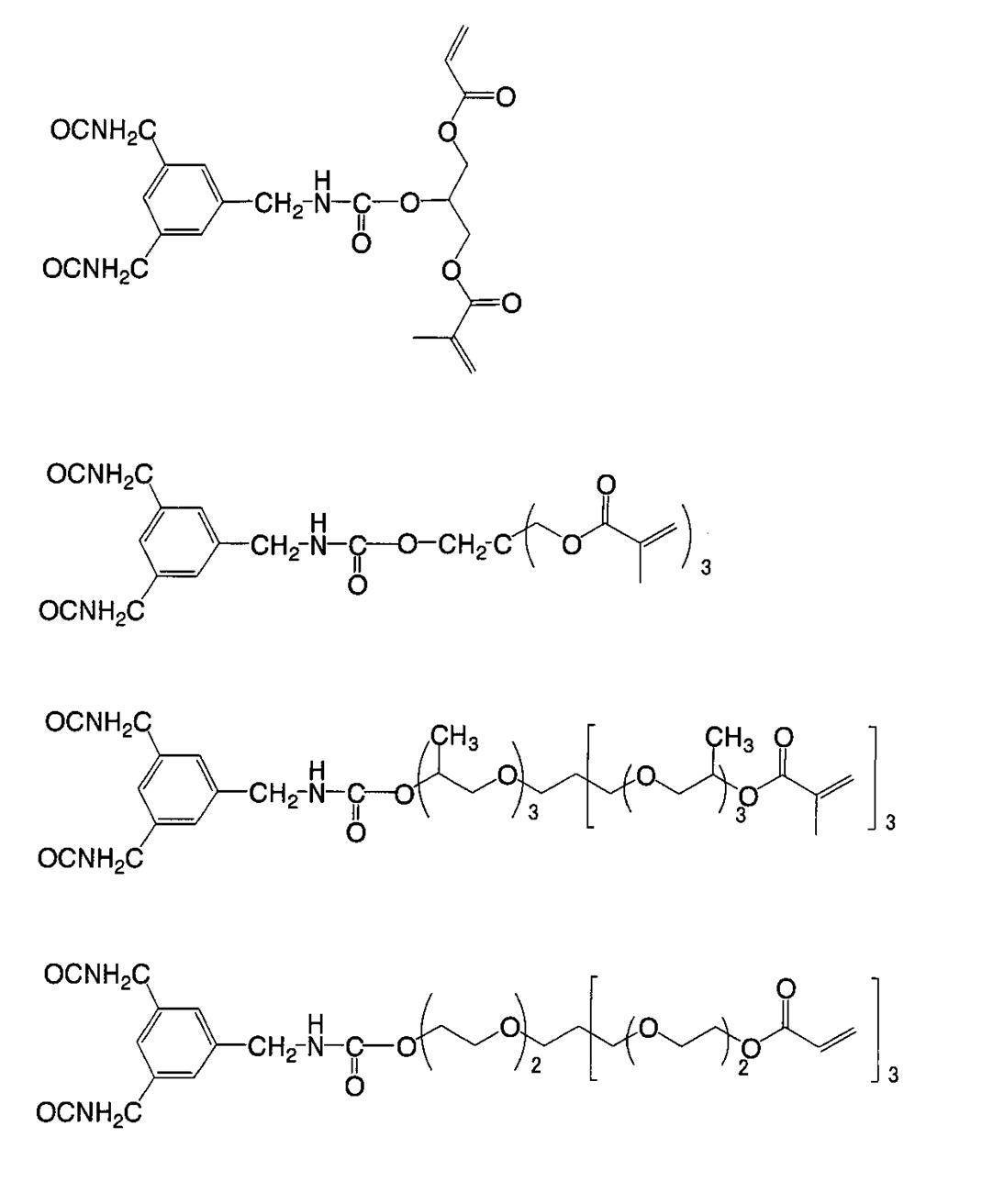
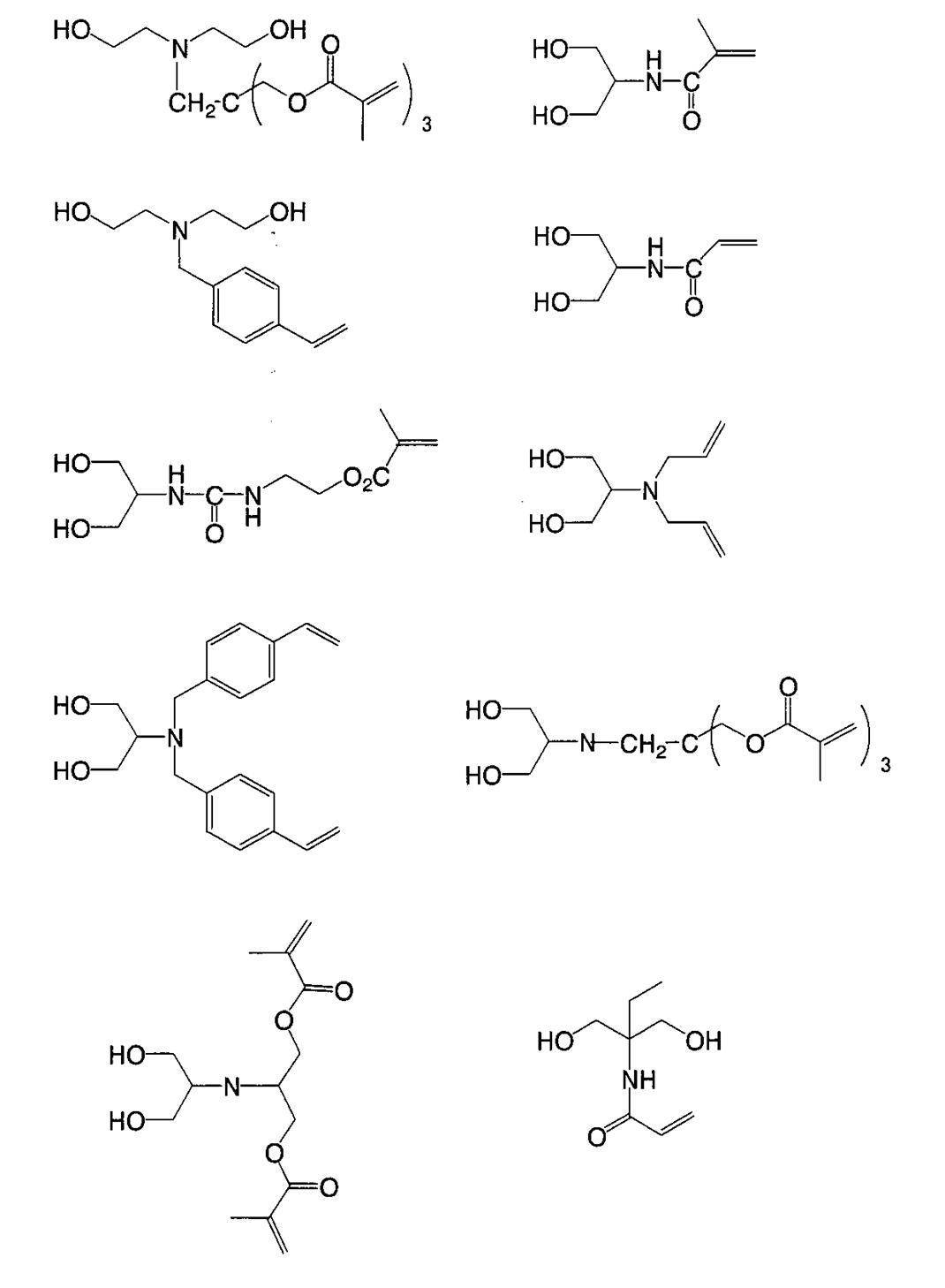
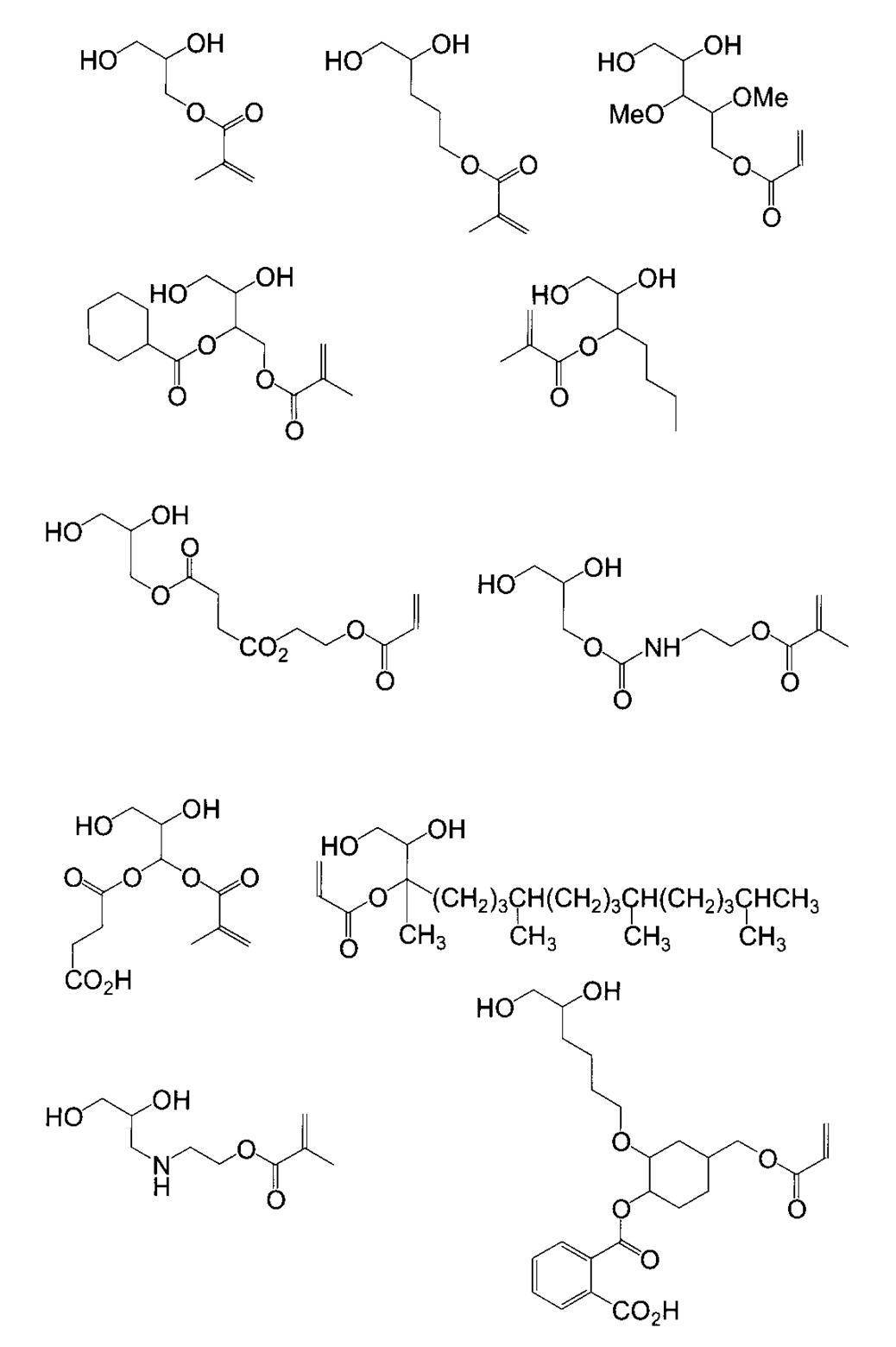
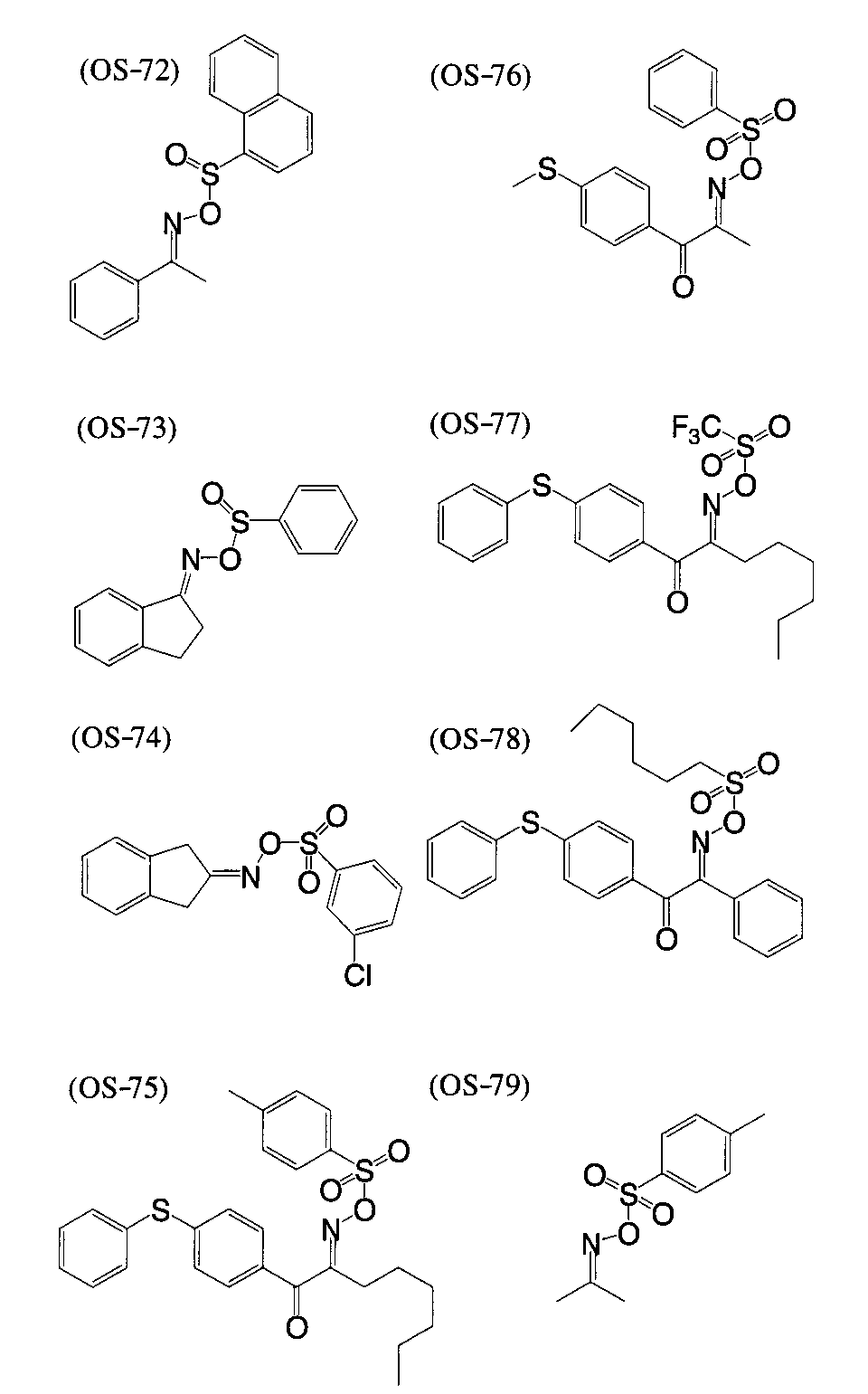
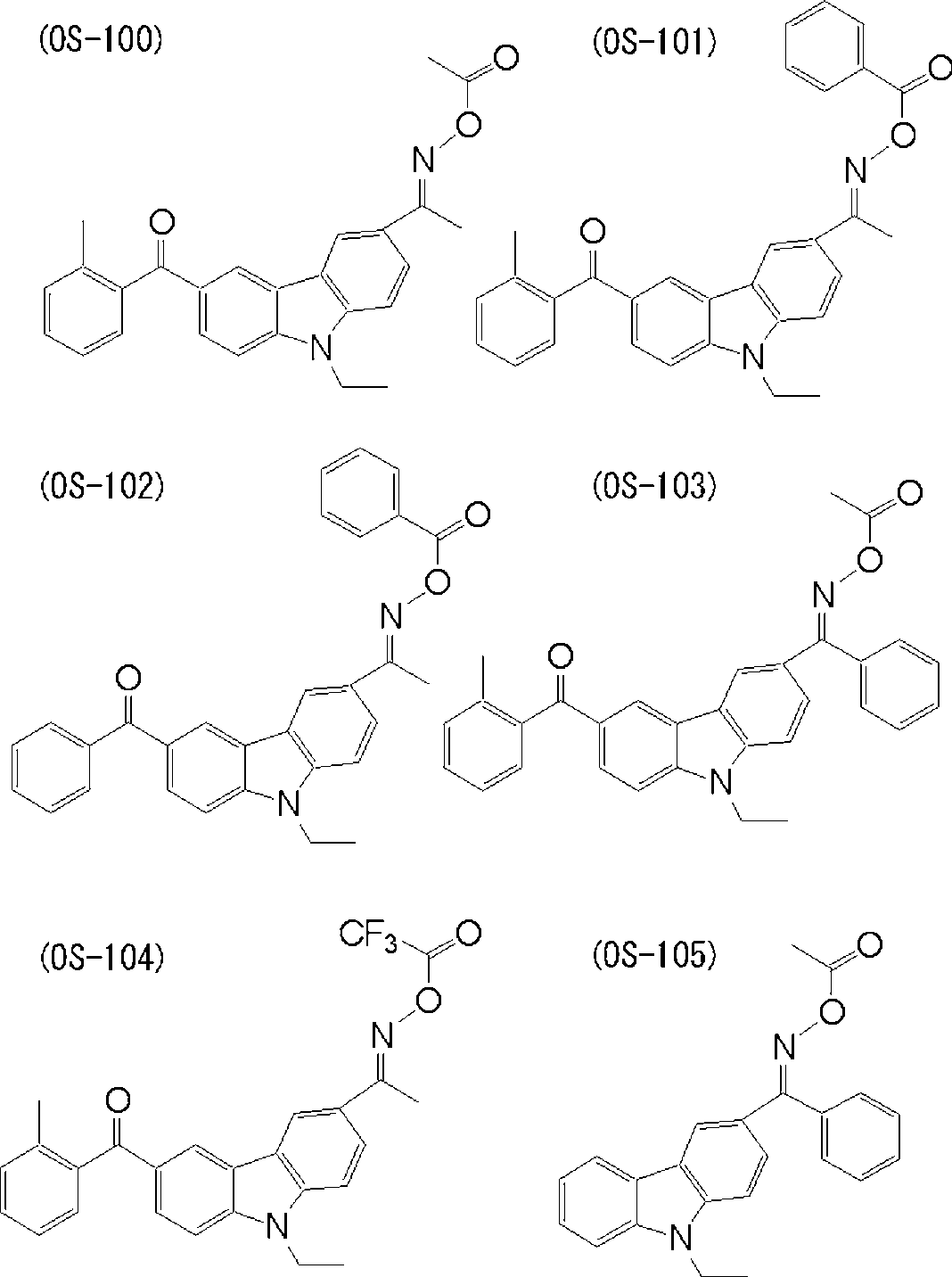
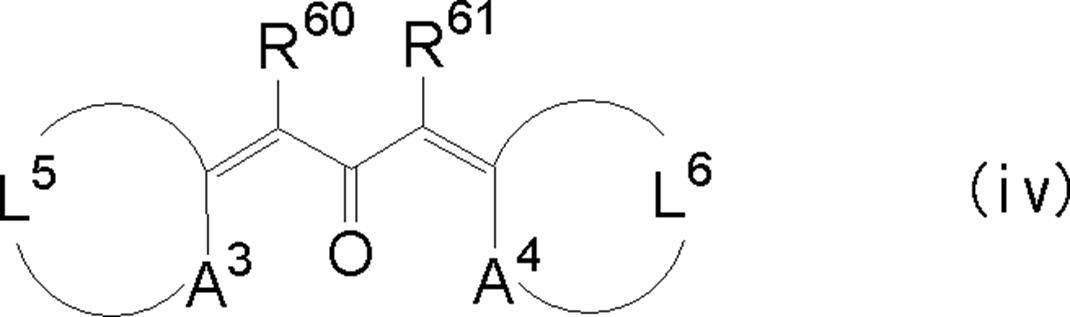
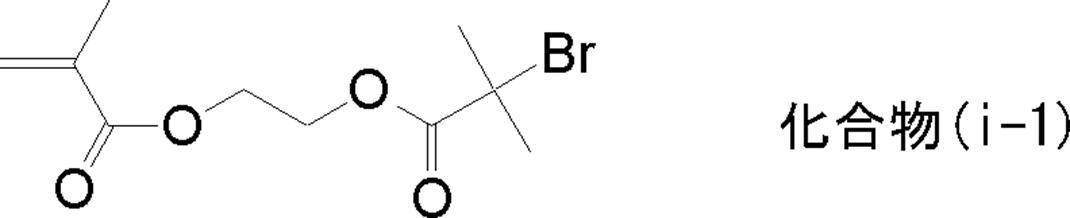
WO2009113447A1 - 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 - Google Patents
着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 Download PDFInfo
- Publication number
- WO2009113447A1 WO2009113447A1 PCT/JP2009/054216 JP2009054216W WO2009113447A1 WO 2009113447 A1 WO2009113447 A1 WO 2009113447A1 JP 2009054216 W JP2009054216 W JP 2009054216W WO 2009113447 A1 WO2009113447 A1 WO 2009113447A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- pigment
- acid
- compound
- substituent
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 178
- 238000003384 imaging method Methods 0.000 title claims description 24
- 239000000049 pigment Substances 0.000 claims abstract description 276
- -1 oxime ester Chemical class 0.000 claims abstract description 216
- 150000001875 compounds Chemical class 0.000 claims abstract description 188
- 239000006185 dispersion Substances 0.000 claims abstract description 184
- 229920005989 resin Polymers 0.000 claims abstract description 134
- 239000011347 resin Substances 0.000 claims abstract description 134
- 239000002253 acid Substances 0.000 claims abstract description 59
- 239000003999 initiator Substances 0.000 claims abstract description 51
- 125000003277 amino group Chemical group 0.000 claims abstract description 31
- 150000001408 amides Chemical group 0.000 claims description 18
- 125000000623 heterocyclic group Chemical group 0.000 claims description 14
- 239000007788 liquid Substances 0.000 claims description 12
- XSQUKJJJFZCRTK-UHFFFAOYSA-N urea group Chemical group NC(=O)N XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 claims description 10
- 125000001424 substituent group Chemical group 0.000 description 145
- 229920000642 polymer Polymers 0.000 description 86
- 125000003118 aryl group Chemical group 0.000 description 68
- 238000000034 method Methods 0.000 description 56
- 125000000217 alkyl group Chemical group 0.000 description 51
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 50
- 239000000758 substrate Substances 0.000 description 49
- 239000010408 film Substances 0.000 description 44
- 238000000576 coating method Methods 0.000 description 43
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 42
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 42
- 230000035945 sensitivity Effects 0.000 description 42
- 125000004432 carbon atom Chemical group C* 0.000 description 40
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 40
- 239000000178 monomer Substances 0.000 description 39
- 239000011248 coating agent Substances 0.000 description 38
- 229920005749 polyurethane resin Polymers 0.000 description 38
- 239000000243 solution Substances 0.000 description 38
- 238000011161 development Methods 0.000 description 29
- 230000018109 developmental process Effects 0.000 description 29
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 28
- 230000015572 biosynthetic process Effects 0.000 description 28
- 230000000052 comparative effect Effects 0.000 description 26
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 25
- 238000002360 preparation method Methods 0.000 description 25
- 229910052717 sulfur Inorganic materials 0.000 description 25
- 125000003545 alkoxy group Chemical group 0.000 description 23
- 125000005843 halogen group Chemical group 0.000 description 23
- 125000004434 sulfur atom Chemical group 0.000 description 23
- 239000011230 binding agent Substances 0.000 description 22
- 125000000524 functional group Chemical group 0.000 description 22
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 21
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 21
- 125000000962 organic group Chemical group 0.000 description 21
- 239000000126 substance Substances 0.000 description 21
- 229920001577 copolymer Polymers 0.000 description 20
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 20
- 239000004973 liquid crystal related substance Substances 0.000 description 20
- 238000003786 synthesis reaction Methods 0.000 description 20
- 125000004430 oxygen atom Chemical group O* 0.000 description 19
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 18
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 18
- 238000006243 chemical reaction Methods 0.000 description 18
- 239000000047 product Substances 0.000 description 18
- 125000001931 aliphatic group Chemical group 0.000 description 17
- 238000001723 curing Methods 0.000 description 17
- 239000002270 dispersing agent Substances 0.000 description 17
- 150000002148 esters Chemical class 0.000 description 17
- 239000010703 silicon Substances 0.000 description 17
- 229910052710 silicon Inorganic materials 0.000 description 17
- 239000007787 solid Substances 0.000 description 17
- 239000002904 solvent Substances 0.000 description 17
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 16
- 238000005227 gel permeation chromatography Methods 0.000 description 16
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 16
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 15
- 239000004593 Epoxy Substances 0.000 description 15
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 15
- 125000002947 alkylene group Chemical group 0.000 description 15
- 239000000975 dye Substances 0.000 description 15
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 15
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 14
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 14
- 125000004104 aryloxy group Chemical group 0.000 description 14
- 125000004093 cyano group Chemical group *C#N 0.000 description 14
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 14
- 235000011118 potassium hydroxide Nutrition 0.000 description 14
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 14
- 239000000600 sorbitol Substances 0.000 description 14
- 239000012860 organic pigment Substances 0.000 description 13
- 150000002923 oximes Chemical class 0.000 description 13
- 238000006116 polymerization reaction Methods 0.000 description 13
- 230000008569 process Effects 0.000 description 13
- 150000003254 radicals Chemical class 0.000 description 13
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 13
- 239000004094 surface-active agent Substances 0.000 description 13
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 12
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 12
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 12
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 12
- 239000011521 glass Substances 0.000 description 12
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 12
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 11
- 239000003513 alkali Substances 0.000 description 11
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 11
- 239000003086 colorant Substances 0.000 description 11
- 238000010438 heat treatment Methods 0.000 description 11
- GTDPSWPPOUPBNX-UHFFFAOYSA-N ac1mqpva Chemical compound CC12C(=O)OC(=O)C1(C)C1(C)C2(C)C(=O)OC1=O GTDPSWPPOUPBNX-UHFFFAOYSA-N 0.000 description 10
- 238000004040 coloring Methods 0.000 description 10
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 description 10
- 239000003960 organic solvent Substances 0.000 description 10
- 150000003839 salts Chemical class 0.000 description 10
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 9
- 229920001890 Novodur Polymers 0.000 description 9
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 9
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 9
- 150000008065 acid anhydrides Chemical class 0.000 description 9
- 125000003342 alkenyl group Chemical group 0.000 description 9
- 150000001735 carboxylic acids Chemical class 0.000 description 9
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 9
- 230000000694 effects Effects 0.000 description 9
- 239000011976 maleic acid Substances 0.000 description 9
- 239000002245 particle Substances 0.000 description 9
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 8
- SOGAXMICEFXMKE-UHFFFAOYSA-N Butylmethacrylate Chemical compound CCCCOC(=O)C(C)=C SOGAXMICEFXMKE-UHFFFAOYSA-N 0.000 description 8
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 8
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 8
- 239000004721 Polyphenylene oxide Substances 0.000 description 8
- 239000004793 Polystyrene Substances 0.000 description 8
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 8
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 8
- 125000000304 alkynyl group Chemical group 0.000 description 8
- 125000003368 amide group Chemical group 0.000 description 8
- 150000001412 amines Chemical class 0.000 description 8
- 239000011324 bead Substances 0.000 description 8
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 8
- 125000000753 cycloalkyl group Chemical group 0.000 description 8
- 238000011156 evaluation Methods 0.000 description 8
- 238000001914 filtration Methods 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- LVHBHZANLOWSRM-UHFFFAOYSA-N methylenebutanedioic acid Natural products OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 8
- 238000002156 mixing Methods 0.000 description 8
- 229920000728 polyester Polymers 0.000 description 8
- 229920000570 polyether Polymers 0.000 description 8
- 229920002223 polystyrene Polymers 0.000 description 8
- 230000009257 reactivity Effects 0.000 description 8
- 239000001054 red pigment Substances 0.000 description 8
- 229920002803 thermoplastic polyurethane Polymers 0.000 description 8
- LDHQCZJRKDOVOX-UHFFFAOYSA-N trans-crotonic acid Natural products CC=CC(O)=O LDHQCZJRKDOVOX-UHFFFAOYSA-N 0.000 description 8
- 239000001052 yellow pigment Substances 0.000 description 8
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 7
- 239000002202 Polyethylene glycol Substances 0.000 description 7
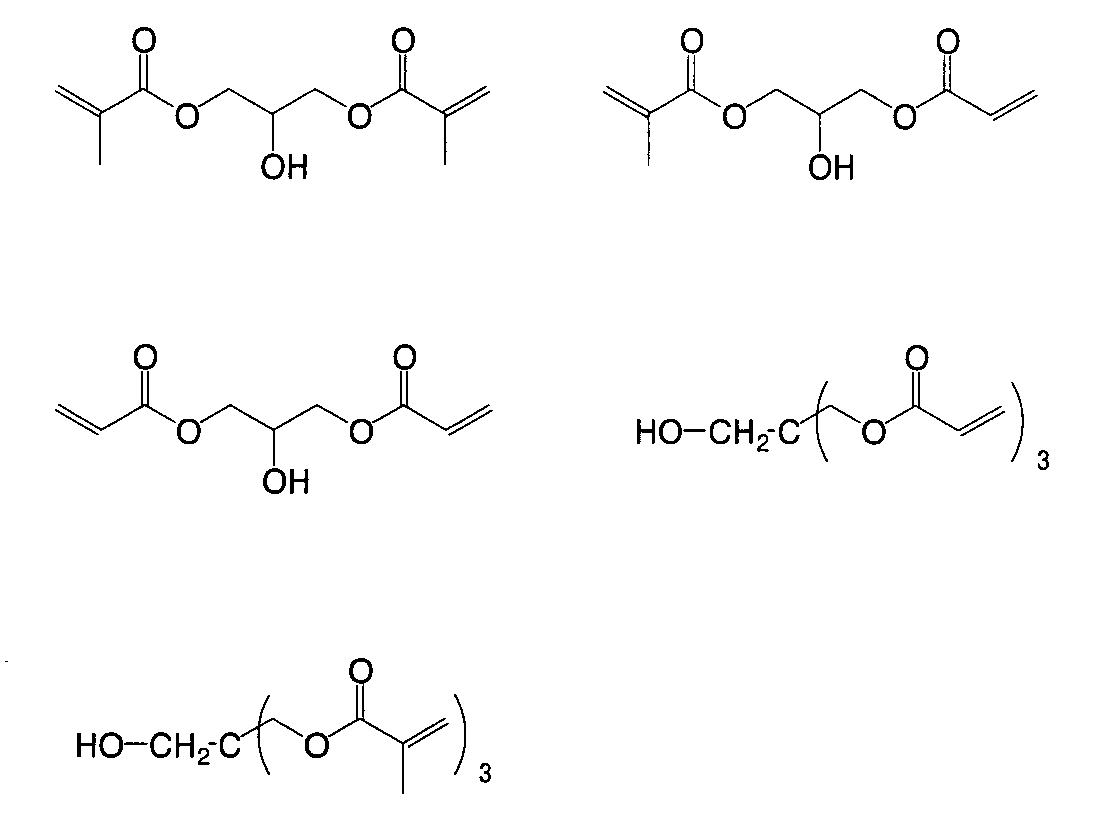
- MPIAGWXWVAHQBB-UHFFFAOYSA-N [3-prop-2-enoyloxy-2-[[3-prop-2-enoyloxy-2,2-bis(prop-2-enoyloxymethyl)propoxy]methyl]-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(COC(=O)C=C)(COC(=O)C=C)COCC(COC(=O)C=C)(COC(=O)C=C)COC(=O)C=C MPIAGWXWVAHQBB-UHFFFAOYSA-N 0.000 description 7
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 7
- 150000004056 anthraquinones Chemical class 0.000 description 7
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 description 7
- 125000003710 aryl alkyl group Chemical group 0.000 description 7
- AOJOEFVRHOZDFN-UHFFFAOYSA-N benzyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC1=CC=CC=C1 AOJOEFVRHOZDFN-UHFFFAOYSA-N 0.000 description 7
- LDHQCZJRKDOVOX-NSCUHMNNSA-N crotonic acid Chemical compound C\C=C\C(O)=O LDHQCZJRKDOVOX-NSCUHMNNSA-N 0.000 description 7
- 125000000392 cycloalkenyl group Chemical group 0.000 description 7
- 125000005442 diisocyanate group Chemical group 0.000 description 7
- VOZRXNHHFUQHIL-UHFFFAOYSA-N glycidyl methacrylate Chemical compound CC(=C)C(=O)OCC1CO1 VOZRXNHHFUQHIL-UHFFFAOYSA-N 0.000 description 7
- 125000005842 heteroatom Chemical group 0.000 description 7
- 230000003993 interaction Effects 0.000 description 7
- 125000005647 linker group Chemical group 0.000 description 7
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 7
- 229920000620 organic polymer Polymers 0.000 description 7
- 229920000647 polyepoxide Polymers 0.000 description 7
- 229920001223 polyethylene glycol Polymers 0.000 description 7
- 229920001451 polypropylene glycol Polymers 0.000 description 7
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 7
- MYWOJODOMFBVCB-UHFFFAOYSA-N 1,2,6-trimethylphenanthrene Chemical compound CC1=CC=C2C3=CC(C)=CC=C3C=CC2=C1C MYWOJODOMFBVCB-UHFFFAOYSA-N 0.000 description 6
- UZKWTJUDCOPSNM-UHFFFAOYSA-N 1-ethenoxybutane Chemical compound CCCCOC=C UZKWTJUDCOPSNM-UHFFFAOYSA-N 0.000 description 6
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 6
- JAHNSTQSQJOJLO-UHFFFAOYSA-N 2-(3-fluorophenyl)-1h-imidazole Chemical compound FC1=CC=CC(C=2NC=CN=2)=C1 JAHNSTQSQJOJLO-UHFFFAOYSA-N 0.000 description 6
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical group S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 6
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 6
- 239000000654 additive Substances 0.000 description 6
- 125000004391 aryl sulfonyl group Chemical group 0.000 description 6
- 125000004429 atom Chemical group 0.000 description 6
- 125000000751 azo group Chemical group [*]N=N[*] 0.000 description 6
- 239000002585 base Substances 0.000 description 6
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical class C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 238000007334 copolymerization reaction Methods 0.000 description 6
- 125000003700 epoxy group Chemical group 0.000 description 6
- 239000003822 epoxy resin Substances 0.000 description 6
- 239000001530 fumaric acid Substances 0.000 description 6
- 229910017053 inorganic salt Inorganic materials 0.000 description 6
- 239000011159 matrix material Substances 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- 229920003145 methacrylic acid copolymer Polymers 0.000 description 6
- 229940117841 methacrylic acid copolymer Drugs 0.000 description 6
- 229960004063 propylene glycol Drugs 0.000 description 6
- 238000007670 refining Methods 0.000 description 6
- 238000003860 storage Methods 0.000 description 6
- 229940058015 1,3-butylene glycol Drugs 0.000 description 5
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 5
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 5
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 5
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 5
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 5
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 5
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical class C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 238000007259 addition reaction Methods 0.000 description 5
- 239000000853 adhesive Substances 0.000 description 5
- 230000001070 adhesive effect Effects 0.000 description 5
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 125000001951 carbamoylamino group Chemical group C(N)(=O)N* 0.000 description 5
- 239000007795 chemical reaction product Substances 0.000 description 5
- 125000002993 cycloalkylene group Chemical group 0.000 description 5
- 125000004663 dialkyl amino group Chemical group 0.000 description 5
- 239000000539 dimer Substances 0.000 description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 5
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 5
- 238000000227 grinding Methods 0.000 description 5
- 239000003112 inhibitor Substances 0.000 description 5
- 239000000976 ink Substances 0.000 description 5
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 5
- FQPSGWSUVKBHSU-UHFFFAOYSA-N methacrylamide Chemical compound CC(=C)C(N)=O FQPSGWSUVKBHSU-UHFFFAOYSA-N 0.000 description 5
- 229910052757 nitrogen Inorganic materials 0.000 description 5
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 5
- 239000002736 nonionic surfactant Substances 0.000 description 5
- 229920003986 novolac Polymers 0.000 description 5
- NWVVVBRKAWDGAB-UHFFFAOYSA-N p-methoxyphenol Chemical compound COC1=CC=C(O)C=C1 NWVVVBRKAWDGAB-UHFFFAOYSA-N 0.000 description 5
- 239000003505 polymerization initiator Substances 0.000 description 5
- 238000012545 processing Methods 0.000 description 5
- 238000012719 thermal polymerization Methods 0.000 description 5
- 239000010936 titanium Substances 0.000 description 5
- 229910052719 titanium Inorganic materials 0.000 description 5
- 238000004448 titration Methods 0.000 description 5
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 4
- JLIDVCMBCGBIEY-UHFFFAOYSA-N 1-penten-3-one Chemical compound CCC(=O)C=C JLIDVCMBCGBIEY-UHFFFAOYSA-N 0.000 description 4
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 4
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 4
- GOXQRTZXKQZDDN-UHFFFAOYSA-N 2-Ethylhexyl acrylate Chemical compound CCCCC(CC)COC(=O)C=C GOXQRTZXKQZDDN-UHFFFAOYSA-N 0.000 description 4
- HCLJOFJIQIJXHS-UHFFFAOYSA-N 2-[2-[2-(2-prop-2-enoyloxyethoxy)ethoxy]ethoxy]ethyl prop-2-enoate Chemical compound C=CC(=O)OCCOCCOCCOCCOC(=O)C=C HCLJOFJIQIJXHS-UHFFFAOYSA-N 0.000 description 4
- VVBLNCFGVYUYGU-UHFFFAOYSA-N 4,4'-Bis(dimethylamino)benzophenone Chemical compound C1=CC(N(C)C)=CC=C1C(=O)C1=CC=C(N(C)C)C=C1 VVBLNCFGVYUYGU-UHFFFAOYSA-N 0.000 description 4
- IRQWEODKXLDORP-UHFFFAOYSA-N 4-ethenylbenzoic acid Chemical compound OC(=O)C1=CC=C(C=C)C=C1 IRQWEODKXLDORP-UHFFFAOYSA-N 0.000 description 4
- 239000004925 Acrylic resin Substances 0.000 description 4
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 4
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- WOBHKFSMXKNTIM-UHFFFAOYSA-N Hydroxyethyl methacrylate Chemical compound CC(=C)C(=O)OCCO WOBHKFSMXKNTIM-UHFFFAOYSA-N 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 4
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 4
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 4
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 4
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 4
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 4
- 230000002378 acidificating effect Effects 0.000 description 4
- 150000007513 acids Chemical class 0.000 description 4
- 125000002723 alicyclic group Chemical group 0.000 description 4
- 150000001336 alkenes Chemical class 0.000 description 4
- 125000003282 alkyl amino group Chemical group 0.000 description 4
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 4
- 125000001769 aryl amino group Chemical group 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N benzo-alpha-pyrone Natural products C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 4
- 239000001055 blue pigment Substances 0.000 description 4
- FUSUHKVFWTUUBE-UHFFFAOYSA-N buten-2-one Chemical compound CC(=O)C=C FUSUHKVFWTUUBE-UHFFFAOYSA-N 0.000 description 4
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical compound CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 4
- 239000006229 carbon black Substances 0.000 description 4
- 125000002603 chloroethyl group Chemical group [H]C([*])([H])C([H])([H])Cl 0.000 description 4
- NEHMKBQYUWJMIP-UHFFFAOYSA-N chloromethane Chemical compound ClC NEHMKBQYUWJMIP-UHFFFAOYSA-N 0.000 description 4
- 238000004132 cross linking Methods 0.000 description 4
- 230000007423 decrease Effects 0.000 description 4
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 4
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 4
- FJKIXWOMBXYWOQ-UHFFFAOYSA-N ethenoxyethane Chemical compound CCOC=C FJKIXWOMBXYWOQ-UHFFFAOYSA-N 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 239000001056 green pigment Substances 0.000 description 4
- JTHNLKXLWOXOQK-UHFFFAOYSA-N hex-1-en-3-one Chemical compound CCCC(=O)C=C JTHNLKXLWOXOQK-UHFFFAOYSA-N 0.000 description 4
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 4
- 230000006872 improvement Effects 0.000 description 4
- 230000000977 initiatory effect Effects 0.000 description 4
- 238000004898 kneading Methods 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 230000007246 mechanism Effects 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 239000011259 mixed solution Substances 0.000 description 4
- SWPMNMYLORDLJE-UHFFFAOYSA-N n-ethylprop-2-enamide Chemical compound CCNC(=O)C=C SWPMNMYLORDLJE-UHFFFAOYSA-N 0.000 description 4
- BPCNEKWROYSOLT-UHFFFAOYSA-N n-phenylprop-2-enamide Chemical compound C=CC(=O)NC1=CC=CC=C1 BPCNEKWROYSOLT-UHFFFAOYSA-N 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 4
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical class N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 4
- 229920000515 polycarbonate Polymers 0.000 description 4
- 239000004417 polycarbonate Substances 0.000 description 4
- 229920001721 polyimide Polymers 0.000 description 4
- FYNROBRQIVCIQF-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole-5,6-dione Chemical compound C1=CN=C2C(=O)C(=O)N=C21 FYNROBRQIVCIQF-UHFFFAOYSA-N 0.000 description 4
- 238000007142 ring opening reaction Methods 0.000 description 4
- 229920006395 saturated elastomer Polymers 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 125000005504 styryl group Chemical group 0.000 description 4
- 125000005420 sulfonamido group Chemical group S(=O)(=O)(N*)* 0.000 description 4
- 238000001308 synthesis method Methods 0.000 description 4
- 125000005309 thioalkoxy group Chemical group 0.000 description 4
- 125000005296 thioaryloxy group Chemical group 0.000 description 4
- 150000003573 thiols Chemical class 0.000 description 4
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea group Chemical group NC(=S)N UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 4
- 238000002834 transmittance Methods 0.000 description 4
- KOZCZZVUFDCZGG-UHFFFAOYSA-N vinyl benzoate Chemical compound C=COC(=O)C1=CC=CC=C1 KOZCZZVUFDCZGG-UHFFFAOYSA-N 0.000 description 4
- 229920001567 vinyl ester resin Polymers 0.000 description 4
- 229920002554 vinyl polymer Polymers 0.000 description 4
- JAMNSIXSLVPNLC-UHFFFAOYSA-N (4-ethenylphenyl) acetate Chemical compound CC(=O)OC1=CC=C(C=C)C=C1 JAMNSIXSLVPNLC-UHFFFAOYSA-N 0.000 description 3
- JIHQDMXYYFUGFV-UHFFFAOYSA-N 1,3,5-triazine Chemical group C1=NC=NC=N1 JIHQDMXYYFUGFV-UHFFFAOYSA-N 0.000 description 3
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 3
- JVYDLYGCSIHCMR-UHFFFAOYSA-N 2,2-bis(hydroxymethyl)butanoic acid Chemical compound CCC(CO)(CO)C(O)=O JVYDLYGCSIHCMR-UHFFFAOYSA-N 0.000 description 3
- QTUVQQKHBMGYEH-UHFFFAOYSA-N 2-(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC=NC=N1 QTUVQQKHBMGYEH-UHFFFAOYSA-N 0.000 description 3
- TXBCBTDQIULDIA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol Chemical compound OCC(CO)(CO)COCC(CO)(CO)CO TXBCBTDQIULDIA-UHFFFAOYSA-N 0.000 description 3
- IWTYTFSSTWXZFU-UHFFFAOYSA-N 3-chloroprop-1-enylbenzene Chemical compound ClCC=CC1=CC=CC=C1 IWTYTFSSTWXZFU-UHFFFAOYSA-N 0.000 description 3
- JLBJTVDPSNHSKJ-UHFFFAOYSA-N 4-Methylstyrene Chemical compound CC1=CC=C(C=C)C=C1 JLBJTVDPSNHSKJ-UHFFFAOYSA-N 0.000 description 3
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 3
- 229920000178 Acrylic resin Polymers 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 3
- GYCMBHHDWRMZGG-UHFFFAOYSA-N Methylacrylonitrile Chemical compound CC(=C)C#N GYCMBHHDWRMZGG-UHFFFAOYSA-N 0.000 description 3
- CNCOEDDPFOAUMB-UHFFFAOYSA-N N-Methylolacrylamide Chemical compound OCNC(=O)C=C CNCOEDDPFOAUMB-UHFFFAOYSA-N 0.000 description 3
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- XYFCBTPGUUZFHI-UHFFFAOYSA-N Phosphine Natural products P XYFCBTPGUUZFHI-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 3
- FHLPGTXWCFQMIU-UHFFFAOYSA-N [4-[2-(4-prop-2-enoyloxyphenyl)propan-2-yl]phenyl] prop-2-enoate Chemical compound C=1C=C(OC(=O)C=C)C=CC=1C(C)(C)C1=CC=C(OC(=O)C=C)C=C1 FHLPGTXWCFQMIU-UHFFFAOYSA-N 0.000 description 3
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 3
- 150000008062 acetophenones Chemical class 0.000 description 3
- 125000005396 acrylic acid ester group Chemical group 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 125000000129 anionic group Chemical group 0.000 description 3
- 125000000732 arylene group Chemical group 0.000 description 3
- KKEYFWRCBNTPAC-UHFFFAOYSA-N benzene-dicarboxylic acid Natural products OC(=O)C1=CC=C(C(O)=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-N 0.000 description 3
- 150000008366 benzophenones Chemical class 0.000 description 3
- DZBUGLKDJFMEHC-UHFFFAOYSA-N benzoquinolinylidene Natural products C1=CC=CC2=CC3=CC=CC=C3N=C21 DZBUGLKDJFMEHC-UHFFFAOYSA-N 0.000 description 3
- 238000012662 bulk polymerization Methods 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 150000001733 carboxylic acid esters Chemical class 0.000 description 3
- 239000001913 cellulose Substances 0.000 description 3
- 229920002678 cellulose Polymers 0.000 description 3
- 235000001671 coumarin Nutrition 0.000 description 3
- 150000004775 coumarins Chemical class 0.000 description 3
- 239000012975 dibutyltin dilaurate Substances 0.000 description 3
- 150000002009 diols Chemical class 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 125000006575 electron-withdrawing group Chemical group 0.000 description 3
- 238000003379 elimination reaction Methods 0.000 description 3
- 125000004185 ester group Chemical group 0.000 description 3
- MEGHWIAOTJPCHQ-UHFFFAOYSA-N ethenyl butanoate Chemical compound CCCC(=O)OC=C MEGHWIAOTJPCHQ-UHFFFAOYSA-N 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 239000011737 fluorine Substances 0.000 description 3
- 229920001519 homopolymer Polymers 0.000 description 3
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical class I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 239000001023 inorganic pigment Substances 0.000 description 3
- 239000012948 isocyanate Substances 0.000 description 3
- 150000002513 isocyanates Chemical class 0.000 description 3
- ZFSLODLOARCGLH-UHFFFAOYSA-N isocyanuric acid Chemical compound OC1=NC(O)=NC(O)=N1 ZFSLODLOARCGLH-UHFFFAOYSA-N 0.000 description 3
- GWVMLCQWXVFZCN-UHFFFAOYSA-N isoindoline Chemical compound C1=CC=C2CNCC2=C1 GWVMLCQWXVFZCN-UHFFFAOYSA-N 0.000 description 3
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 125000005397 methacrylic acid ester group Chemical group 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 description 3
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 3
- PNJWIWWMYCMZRO-UHFFFAOYSA-N pent‐4‐en‐2‐one Natural products CC(=O)CC=C PNJWIWWMYCMZRO-UHFFFAOYSA-N 0.000 description 3
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 3
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 3
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 3
- 229910000073 phosphorus hydride Inorganic materials 0.000 description 3
- 229920006122 polyamide resin Polymers 0.000 description 3
- 239000009719 polyimide resin Substances 0.000 description 3
- 229910052700 potassium Inorganic materials 0.000 description 3
- FBCQUCJYYPMKRO-UHFFFAOYSA-N prop-2-enyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC=C FBCQUCJYYPMKRO-UHFFFAOYSA-N 0.000 description 3
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 3
- 238000010298 pulverizing process Methods 0.000 description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 3
- IZMJMCDDWKSTTK-UHFFFAOYSA-N quinoline yellow Chemical compound C1=CC=CC2=NC(C3C(C4=CC=CC=C4C3=O)=O)=CC=C21 IZMJMCDDWKSTTK-UHFFFAOYSA-N 0.000 description 3
- 238000010526 radical polymerization reaction Methods 0.000 description 3
- 150000003440 styrenes Chemical class 0.000 description 3
- 239000010409 thin film Substances 0.000 description 3
- DVKJHBMWWAPEIU-UHFFFAOYSA-N toluene 2,4-diisocyanate Chemical compound CC1=CC=C(N=C=O)C=C1N=C=O DVKJHBMWWAPEIU-UHFFFAOYSA-N 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- 125000005409 triarylsulfonium group Chemical group 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- ORTVZLZNOYNASJ-OWOJBTEDSA-N (e)-but-2-ene-1,4-diol Chemical compound OC\C=C\CO ORTVZLZNOYNASJ-OWOJBTEDSA-N 0.000 description 2
- FFJCNSLCJOQHKM-CLFAGFIQSA-N (z)-1-[(z)-octadec-9-enoxy]octadec-9-ene Chemical compound CCCCCCCC\C=C/CCCCCCCCOCCCCCCCC\C=C/CCCCCCCC FFJCNSLCJOQHKM-CLFAGFIQSA-N 0.000 description 2
- DHKHKXVYLBGOIT-UHFFFAOYSA-N 1,1-Diethoxyethane Chemical compound CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 2
- YXIWHUQXZSMYRE-UHFFFAOYSA-N 1,3-benzothiazole-2-thiol Chemical compound C1=CC=C2SC(S)=NC2=C1 YXIWHUQXZSMYRE-UHFFFAOYSA-N 0.000 description 2
- XXCVIFJHBFNFBO-UHFFFAOYSA-N 1-ethenoxyoctane Chemical compound CCCCCCCCOC=C XXCVIFJHBFNFBO-UHFFFAOYSA-N 0.000 description 2
- OVGRCEFMXPHEBL-UHFFFAOYSA-N 1-ethenoxypropane Chemical compound CCCOC=C OVGRCEFMXPHEBL-UHFFFAOYSA-N 0.000 description 2
- OSSNTDFYBPYIEC-UHFFFAOYSA-N 1-ethenylimidazole Chemical compound C=CN1C=CN=C1 OSSNTDFYBPYIEC-UHFFFAOYSA-N 0.000 description 2
- WDRZVZVXHZNSFG-UHFFFAOYSA-N 1-ethenylpyridin-1-ium Chemical group C=C[N+]1=CC=CC=C1 WDRZVZVXHZNSFG-UHFFFAOYSA-N 0.000 description 2
- KUIZKZHDMPERHR-UHFFFAOYSA-N 1-phenylprop-2-en-1-one Chemical compound C=CC(=O)C1=CC=CC=C1 KUIZKZHDMPERHR-UHFFFAOYSA-N 0.000 description 2
- PTBDIHRZYDMNKB-UHFFFAOYSA-N 2,2-Bis(hydroxymethyl)propionic acid Chemical compound OCC(C)(CO)C(O)=O PTBDIHRZYDMNKB-UHFFFAOYSA-N 0.000 description 2
- STMDPCBYJCIZOD-UHFFFAOYSA-N 2-(2,4-dinitroanilino)-4-methylpentanoic acid Chemical compound CC(C)CC(C(O)=O)NC1=CC=C([N+]([O-])=O)C=C1[N+]([O-])=O STMDPCBYJCIZOD-UHFFFAOYSA-N 0.000 description 2
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical compound ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 2
- INQDDHNZXOAFFD-UHFFFAOYSA-N 2-[2-(2-prop-2-enoyloxyethoxy)ethoxy]ethyl prop-2-enoate Chemical compound C=CC(=O)OCCOCCOCCOC(=O)C=C INQDDHNZXOAFFD-UHFFFAOYSA-N 0.000 description 2
- FDSUVTROAWLVJA-UHFFFAOYSA-N 2-[[3-hydroxy-2,2-bis(hydroxymethyl)propoxy]methyl]-2-(hydroxymethyl)propane-1,3-diol;prop-2-enoic acid Chemical compound OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OC(=O)C=C.OCC(CO)(CO)COCC(CO)(CO)CO FDSUVTROAWLVJA-UHFFFAOYSA-N 0.000 description 2
- VUIWJRYTWUGOOF-UHFFFAOYSA-N 2-ethenoxyethanol Chemical compound OCCOC=C VUIWJRYTWUGOOF-UHFFFAOYSA-N 0.000 description 2
- WDQMWEYDKDCEHT-UHFFFAOYSA-N 2-ethylhexyl 2-methylprop-2-enoate Chemical compound CCCCC(CC)COC(=O)C(C)=C WDQMWEYDKDCEHT-UHFFFAOYSA-N 0.000 description 2
- OMIGHNLMNHATMP-UHFFFAOYSA-N 2-hydroxyethyl prop-2-enoate Chemical compound OCCOC(=O)C=C OMIGHNLMNHATMP-UHFFFAOYSA-N 0.000 description 2
- VHSHLMUCYSAUQU-UHFFFAOYSA-N 2-hydroxypropyl methacrylate Chemical compound CC(O)COC(=O)C(C)=C VHSHLMUCYSAUQU-UHFFFAOYSA-N 0.000 description 2
- GWZMWHWAWHPNHN-UHFFFAOYSA-N 2-hydroxypropyl prop-2-enoate Chemical compound CC(O)COC(=O)C=C GWZMWHWAWHPNHN-UHFFFAOYSA-N 0.000 description 2
- XLLIQLLCWZCATF-UHFFFAOYSA-N 2-methoxyethyl acetate Chemical compound COCCOC(C)=O XLLIQLLCWZCATF-UHFFFAOYSA-N 0.000 description 2
- HGJDNBZIDQOMEU-UHFFFAOYSA-N 2-methyl-n,n-bis(prop-2-enyl)prop-2-enamide Chemical compound CC(=C)C(=O)N(CC=C)CC=C HGJDNBZIDQOMEU-UHFFFAOYSA-N 0.000 description 2
- VRWOCLJWLOZDAI-UHFFFAOYSA-N 2-methyl-n-propanoylprop-2-enamide Chemical compound CCC(=O)NC(=O)C(C)=C VRWOCLJWLOZDAI-UHFFFAOYSA-N 0.000 description 2
- WXUAQHNMJWJLTG-UHFFFAOYSA-N 2-methylbutanedioic acid Chemical compound OC(=O)C(C)CC(O)=O WXUAQHNMJWJLTG-UHFFFAOYSA-N 0.000 description 2
- XBIPWOJKRZBTQK-UHFFFAOYSA-N 2-methylidenebut-3-enamide Chemical compound NC(=O)C(=C)C=C XBIPWOJKRZBTQK-UHFFFAOYSA-N 0.000 description 2
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 2
- RUMACXVDVNRZJZ-UHFFFAOYSA-N 2-methylpropyl 2-methylprop-2-enoate Chemical compound CC(C)COC(=O)C(C)=C RUMACXVDVNRZJZ-UHFFFAOYSA-N 0.000 description 2
- CFVWNXQPGQOHRJ-UHFFFAOYSA-N 2-methylpropyl prop-2-enoate Chemical compound CC(C)COC(=O)C=C CFVWNXQPGQOHRJ-UHFFFAOYSA-N 0.000 description 2
- CWZWBNXFJNGGRL-UHFFFAOYSA-N 2-phenylethenyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC=CC1=CC=CC=C1 CWZWBNXFJNGGRL-UHFFFAOYSA-N 0.000 description 2
- LUDSLRVDTMIOHA-UHFFFAOYSA-N 2-phenylethenyl prop-2-enoate Chemical compound C=CC(=O)OC=CC1=CC=CC=C1 LUDSLRVDTMIOHA-UHFFFAOYSA-N 0.000 description 2
- ZCYIYBNDJKVCBR-UHFFFAOYSA-N 2-prop-2-enoxyethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCOCC=C ZCYIYBNDJKVCBR-UHFFFAOYSA-N 0.000 description 2
- AKSBCQNPVMRHRZ-UHFFFAOYSA-N 2-prop-2-enoxyethyl prop-2-enoate Chemical compound C=CCOCCOC(=O)C=C AKSBCQNPVMRHRZ-UHFFFAOYSA-N 0.000 description 2
- KUDUQBURMYMBIJ-UHFFFAOYSA-N 2-prop-2-enoyloxyethyl prop-2-enoate Chemical compound C=CC(=O)OCCOC(=O)C=C KUDUQBURMYMBIJ-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- UYEMGAFJOZZIFP-UHFFFAOYSA-N 3,5-dihydroxybenzoic acid Chemical compound OC(=O)C1=CC(O)=CC(O)=C1 UYEMGAFJOZZIFP-UHFFFAOYSA-N 0.000 description 2
- KEPGNQLKWDULGD-UHFFFAOYSA-N 3-(3-prop-2-enoyloxypropoxy)propyl prop-2-enoate Chemical compound C=CC(=O)OCCCOCCCOC(=O)C=C KEPGNQLKWDULGD-UHFFFAOYSA-N 0.000 description 2
- REEBWSYYNPPSKV-UHFFFAOYSA-N 3-[(4-formylphenoxy)methyl]thiophene-2-carbonitrile Chemical compound C1=CC(C=O)=CC=C1OCC1=C(C#N)SC=C1 REEBWSYYNPPSKV-UHFFFAOYSA-N 0.000 description 2
- GNSFRPWPOGYVLO-UHFFFAOYSA-N 3-hydroxypropyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCO GNSFRPWPOGYVLO-UHFFFAOYSA-N 0.000 description 2
- QZPSOSOOLFHYRR-UHFFFAOYSA-N 3-hydroxypropyl prop-2-enoate Chemical compound OCCCOC(=O)C=C QZPSOSOOLFHYRR-UHFFFAOYSA-N 0.000 description 2
- XMIIGOLPHOKFCH-UHFFFAOYSA-N 3-phenylpropionic acid Chemical compound OC(=O)CCC1=CC=CC=C1 XMIIGOLPHOKFCH-UHFFFAOYSA-N 0.000 description 2
- MUPJJZVGSOUSFH-UHFFFAOYSA-N 4-(2-cyanoethyl)-4-nitroheptanedinitrile Chemical compound N#CCCC([N+](=O)[O-])(CCC#N)CCC#N MUPJJZVGSOUSFH-UHFFFAOYSA-N 0.000 description 2
- MPFIISCRTZAMEQ-UHFFFAOYSA-N 4-chloro-n-(2-methylprop-2-enoyl)benzamide Chemical compound CC(=C)C(=O)NC(=O)C1=CC=C(Cl)C=C1 MPFIISCRTZAMEQ-UHFFFAOYSA-N 0.000 description 2
- YKXAYLPDMSGWEV-UHFFFAOYSA-N 4-hydroxybutyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCO YKXAYLPDMSGWEV-UHFFFAOYSA-N 0.000 description 2
- NDWUBGAGUCISDV-UHFFFAOYSA-N 4-hydroxybutyl prop-2-enoate Chemical compound OCCCCOC(=O)C=C NDWUBGAGUCISDV-UHFFFAOYSA-N 0.000 description 2
- FUGYGGDSWSUORM-UHFFFAOYSA-N 4-hydroxystyrene Chemical compound OC1=CC=C(C=C)C=C1 FUGYGGDSWSUORM-UHFFFAOYSA-N 0.000 description 2
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 2
- DGQOZCNCJKEVOA-UHFFFAOYSA-N 5-(2,5-dioxooxolan-3-yl)-7-methyl-3a,4,5,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1C(C(OC2=O)=O)C2C(C)=CC1C1CC(=O)OC1=O DGQOZCNCJKEVOA-UHFFFAOYSA-N 0.000 description 2
- FYYIUODUDSPAJQ-UHFFFAOYSA-N 7-oxabicyclo[4.1.0]heptan-4-ylmethyl 2-methylprop-2-enoate Chemical compound C1C(COC(=O)C(=C)C)CCC2OC21 FYYIUODUDSPAJQ-UHFFFAOYSA-N 0.000 description 2
- DPTGFYXXFXSRIR-UHFFFAOYSA-N 7-oxabicyclo[4.1.0]heptan-4-ylmethyl prop-2-enoate Chemical compound C1C(COC(=O)C=C)CCC2OC21 DPTGFYXXFXSRIR-UHFFFAOYSA-N 0.000 description 2
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 description 2
- 229920002126 Acrylic acid copolymer Polymers 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 2
- BRLQWZUYTZBJKN-UHFFFAOYSA-N Epichlorohydrin Chemical compound ClCC1CO1 BRLQWZUYTZBJKN-UHFFFAOYSA-N 0.000 description 2
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 2
- JIGUQPWFLRLWPJ-UHFFFAOYSA-N Ethyl acrylate Chemical compound CCOC(=O)C=C JIGUQPWFLRLWPJ-UHFFFAOYSA-N 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N Ethylbenzene Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 description 2
- 239000005977 Ethylene Substances 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- 235000000177 Indigofera tinctoria Nutrition 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 229920002396 Polyurea Polymers 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- NRCMAYZCPIVABH-UHFFFAOYSA-N Quinacridone Chemical compound N1C2=CC=CC=C2C(=O)C2=C1C=C1C(=O)C3=CC=CC=C3NC1=C2 NRCMAYZCPIVABH-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- UWHCKJMYHZGTIT-UHFFFAOYSA-N Tetraethylene glycol, Natural products OCCOCCOCCOCCO UWHCKJMYHZGTIT-UHFFFAOYSA-N 0.000 description 2
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 2
- DAKWPKUUDNSNPN-UHFFFAOYSA-N Trimethylolpropane triacrylate Chemical compound C=CC(=O)OCC(CC)(COC(=O)C=C)COC(=O)C=C DAKWPKUUDNSNPN-UHFFFAOYSA-N 0.000 description 2
- 239000007877 V-601 Substances 0.000 description 2
- 235000010724 Wisteria floribunda Nutrition 0.000 description 2
- CQHKDHVZYZUZMJ-UHFFFAOYSA-N [2,2-bis(hydroxymethyl)-3-prop-2-enoyloxypropyl] prop-2-enoate Chemical compound C=CC(=O)OCC(CO)(CO)COC(=O)C=C CQHKDHVZYZUZMJ-UHFFFAOYSA-N 0.000 description 2
- HVVWZTWDBSEWIH-UHFFFAOYSA-N [2-(hydroxymethyl)-3-prop-2-enoyloxy-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(CO)(COC(=O)C=C)COC(=O)C=C HVVWZTWDBSEWIH-UHFFFAOYSA-N 0.000 description 2
- YPCHGLDQZXOZFW-UHFFFAOYSA-N [2-[[4-methyl-3-[[3-prop-2-enoyloxy-2,2-bis(prop-2-enoyloxymethyl)propoxy]carbonylamino]phenyl]carbamoyloxymethyl]-3-prop-2-enoyloxy-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound CC1=CC=C(NC(=O)OCC(COC(=O)C=C)(COC(=O)C=C)COC(=O)C=C)C=C1NC(=O)OCC(COC(=O)C=C)(COC(=O)C=C)COC(=O)C=C YPCHGLDQZXOZFW-UHFFFAOYSA-N 0.000 description 2
- HSZUHSXXAOWGQY-UHFFFAOYSA-N [2-methyl-3-prop-2-enoyloxy-2-(prop-2-enoyloxymethyl)propyl] prop-2-enoate Chemical compound C=CC(=O)OCC(C)(COC(=O)C=C)COC(=O)C=C HSZUHSXXAOWGQY-UHFFFAOYSA-N 0.000 description 2
- 239000011354 acetal resin Substances 0.000 description 2
- 235000011054 acetic acid Nutrition 0.000 description 2
- DPKHZNPWBDQZCN-UHFFFAOYSA-N acridine orange free base Chemical compound C1=CC(N(C)C)=CC2=NC3=CC(N(C)C)=CC=C3C=C21 DPKHZNPWBDQZCN-UHFFFAOYSA-N 0.000 description 2
- 150000001251 acridines Chemical class 0.000 description 2
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 125000004450 alkenylene group Chemical group 0.000 description 2
- 150000005215 alkyl ethers Chemical class 0.000 description 2
- 125000004419 alkynylene group Chemical group 0.000 description 2
- XXROGKLTLUQVRX-UHFFFAOYSA-N allyl alcohol Chemical compound OCC=C XXROGKLTLUQVRX-UHFFFAOYSA-N 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- QMKYBPDZANOJGF-UHFFFAOYSA-N benzene-1,3,5-tricarboxylic acid Chemical compound OC(=O)C1=CC(C(O)=O)=CC(C(O)=O)=C1 QMKYBPDZANOJGF-UHFFFAOYSA-N 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- ISAOCJYIOMOJEB-UHFFFAOYSA-N benzoin Chemical class C=1C=CC=CC=1C(O)C(=O)C1=CC=CC=C1 ISAOCJYIOMOJEB-UHFFFAOYSA-N 0.000 description 2
- 239000012965 benzophenone Substances 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical class C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- GCTPMLUUWLLESL-UHFFFAOYSA-N benzyl prop-2-enoate Chemical compound C=CC(=O)OCC1=CC=CC=C1 GCTPMLUUWLLESL-UHFFFAOYSA-N 0.000 description 2
- 239000004305 biphenyl Substances 0.000 description 2
- 235000010290 biphenyl Nutrition 0.000 description 2
- 235000019437 butane-1,3-diol Nutrition 0.000 description 2
- GKRVGTLVYRYCFR-UHFFFAOYSA-N butane-1,4-diol;2-methylidenebutanedioic acid Chemical compound OCCCCO.OC(=O)CC(=C)C(O)=O.OC(=O)CC(=C)C(O)=O GKRVGTLVYRYCFR-UHFFFAOYSA-N 0.000 description 2
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 2
- 150000001728 carbonyl compounds Chemical class 0.000 description 2
- 125000002843 carboxylic acid group Chemical group 0.000 description 2
- 239000012295 chemical reaction liquid Substances 0.000 description 2
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 2
- XCJYREBRNVKWGJ-UHFFFAOYSA-N copper(II) phthalocyanine Chemical compound [Cu+2].C12=CC=CC=C2C(N=C2[N-]C(C3=CC=CC=C32)=N2)=NC1=NC([C]1C=CC=CC1=1)=NC=1N=C1[C]3C=CC=CC3=C2[N-]1 XCJYREBRNVKWGJ-UHFFFAOYSA-N 0.000 description 2
- 229960000956 coumarin Drugs 0.000 description 2
- 229930003836 cresol Natural products 0.000 description 2
- 150000003950 cyclic amides Chemical group 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- OIWOHHBRDFKZNC-UHFFFAOYSA-N cyclohexyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC1CCCCC1 OIWOHHBRDFKZNC-UHFFFAOYSA-N 0.000 description 2
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 2
- 125000000664 diazo group Chemical group [N-]=[N+]=[*] 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- UKMSUNONTOPOIO-UHFFFAOYSA-N docosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCC(O)=O UKMSUNONTOPOIO-UHFFFAOYSA-N 0.000 description 2
- SNRUBQQJIBEYMU-UHFFFAOYSA-N dodecane Chemical compound CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 2
- 239000012776 electronic material Substances 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- NHOGGUYTANYCGQ-UHFFFAOYSA-N ethenoxybenzene Chemical compound C=COC1=CC=CC=C1 NHOGGUYTANYCGQ-UHFFFAOYSA-N 0.000 description 2
- XJELOQYISYPGDX-UHFFFAOYSA-N ethenyl 2-chloroacetate Chemical compound ClCC(=O)OC=C XJELOQYISYPGDX-UHFFFAOYSA-N 0.000 description 2
- FFYWKOUKJFCBAM-UHFFFAOYSA-N ethenyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC=C FFYWKOUKJFCBAM-UHFFFAOYSA-N 0.000 description 2
- BLCTWBJQROOONQ-UHFFFAOYSA-N ethenyl prop-2-enoate Chemical compound C=COC(=O)C=C BLCTWBJQROOONQ-UHFFFAOYSA-N 0.000 description 2
- UIWXSTHGICQLQT-UHFFFAOYSA-N ethenyl propanoate Chemical compound CCC(=O)OC=C UIWXSTHGICQLQT-UHFFFAOYSA-N 0.000 description 2
- LZCLXQDLBQLTDK-UHFFFAOYSA-N ethyl 2-hydroxypropanoate Chemical compound CCOC(=O)C(C)O LZCLXQDLBQLTDK-UHFFFAOYSA-N 0.000 description 2
- SUPCQIBBMFXVTL-UHFFFAOYSA-N ethyl 2-methylprop-2-enoate Chemical compound CCOC(=O)C(C)=C SUPCQIBBMFXVTL-UHFFFAOYSA-N 0.000 description 2
- 229940116333 ethyl lactate Drugs 0.000 description 2
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 2
- RRAMGCGOFNQTLD-UHFFFAOYSA-N hexamethylene diisocyanate Chemical compound O=C=NCCCCCCN=C=O RRAMGCGOFNQTLD-UHFFFAOYSA-N 0.000 description 2
- LNCPIMCVTKXXOY-UHFFFAOYSA-N hexyl 2-methylprop-2-enoate Chemical compound CCCCCCOC(=O)C(C)=C LNCPIMCVTKXXOY-UHFFFAOYSA-N 0.000 description 2
- LNMQRPPRQDGUDR-UHFFFAOYSA-N hexyl prop-2-enoate Chemical compound CCCCCCOC(=O)C=C LNMQRPPRQDGUDR-UHFFFAOYSA-N 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 150000003949 imides Chemical class 0.000 description 2
- 229940097275 indigo Drugs 0.000 description 2
- COHYTHOBJLSHDF-UHFFFAOYSA-N indigo powder Natural products N1C2=CC=CC=C2C(=O)C1=C1C(=O)C2=CC=CC=C2N1 COHYTHOBJLSHDF-UHFFFAOYSA-N 0.000 description 2
- 150000002484 inorganic compounds Chemical class 0.000 description 2
- 229910010272 inorganic material Inorganic materials 0.000 description 2
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 2
- LDHQCZJRKDOVOX-IHWYPQMZSA-N isocrotonic acid Chemical compound C\C=C/C(O)=O LDHQCZJRKDOVOX-IHWYPQMZSA-N 0.000 description 2
- QQVIHTHCMHWDBS-UHFFFAOYSA-N isophthalic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 2
- 150000002736 metal compounds Chemical class 0.000 description 2
- 125000005641 methacryl group Chemical group 0.000 description 2
- 125000005395 methacrylic acid group Chemical group 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 229940050176 methyl chloride Drugs 0.000 description 2
- UAEPNZWRGJTJPN-UHFFFAOYSA-N methylcyclohexane Chemical compound CC1CCCCC1 UAEPNZWRGJTJPN-UHFFFAOYSA-N 0.000 description 2
- 150000002762 monocarboxylic acid derivatives Chemical class 0.000 description 2
- BLYOHBPLFYXHQA-UHFFFAOYSA-N n,n-bis(prop-2-enyl)prop-2-enamide Chemical compound C=CCN(CC=C)C(=O)C=C BLYOHBPLFYXHQA-UHFFFAOYSA-N 0.000 description 2
- UUORTJUPDJJXST-UHFFFAOYSA-N n-(2-hydroxyethyl)prop-2-enamide Chemical compound OCCNC(=O)C=C UUORTJUPDJJXST-UHFFFAOYSA-N 0.000 description 2
- OJBZOTFHZFZOIJ-UHFFFAOYSA-N n-acetyl-2-methylprop-2-enamide Chemical compound CC(=O)NC(=O)C(C)=C OJBZOTFHZFZOIJ-UHFFFAOYSA-N 0.000 description 2
- PMJFVKWBSWWAKT-UHFFFAOYSA-N n-cyclohexylprop-2-enamide Chemical compound C=CC(=O)NC1CCCCC1 PMJFVKWBSWWAKT-UHFFFAOYSA-N 0.000 description 2
- MRUJSNSDPBZNHO-UHFFFAOYSA-N n-ethenyl-2-methylprop-2-enamide Chemical compound CC(=C)C(=O)NC=C MRUJSNSDPBZNHO-UHFFFAOYSA-N 0.000 description 2
- FYCBGURDLIKBDA-UHFFFAOYSA-N n-hexyl-2-methylprop-2-enamide Chemical compound CCCCCCNC(=O)C(C)=C FYCBGURDLIKBDA-UHFFFAOYSA-N 0.000 description 2
- CHDKQNHKDMEASZ-UHFFFAOYSA-N n-prop-2-enoylprop-2-enamide Chemical compound C=CC(=O)NC(=O)C=C CHDKQNHKDMEASZ-UHFFFAOYSA-N 0.000 description 2
- CNPHCSFIDKZQAK-UHFFFAOYSA-N n-prop-2-enylprop-2-enamide Chemical compound C=CCNC(=O)C=C CNPHCSFIDKZQAK-UHFFFAOYSA-N 0.000 description 2
- LQNUZADURLCDLV-UHFFFAOYSA-N nitrobenzene Chemical compound [O-][N+](=O)C1=CC=CC=C1 LQNUZADURLCDLV-UHFFFAOYSA-N 0.000 description 2
- BDJRBEYXGGNYIS-UHFFFAOYSA-N nonanedioic acid Chemical compound OC(=O)CCCCCCCC(O)=O BDJRBEYXGGNYIS-UHFFFAOYSA-N 0.000 description 2
- 230000000269 nucleophilic effect Effects 0.000 description 2
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 2
- 229940065472 octyl acrylate Drugs 0.000 description 2
- ANISOHQJBAQUQP-UHFFFAOYSA-N octyl prop-2-enoate Chemical compound CCCCCCCCOC(=O)C=C ANISOHQJBAQUQP-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- RPQRDASANLAFCM-UHFFFAOYSA-N oxiran-2-ylmethyl prop-2-enoate Chemical compound C=CC(=O)OCC1CO1 RPQRDASANLAFCM-UHFFFAOYSA-N 0.000 description 2
- 239000003973 paint Substances 0.000 description 2
- FZUGPQWGEGAKET-UHFFFAOYSA-N parbenate Chemical compound CCOC(=O)C1=CC=C(N(C)C)C=C1 FZUGPQWGEGAKET-UHFFFAOYSA-N 0.000 description 2
- UCUUFSAXZMGPGH-UHFFFAOYSA-N penta-1,4-dien-3-one Chemical class C=CC(=O)C=C UCUUFSAXZMGPGH-UHFFFAOYSA-N 0.000 description 2
- GYDSPAVLTMAXHT-UHFFFAOYSA-N pentyl 2-methylprop-2-enoate Chemical compound CCCCCOC(=O)C(C)=C GYDSPAVLTMAXHT-UHFFFAOYSA-N 0.000 description 2
- ULDDEWDFUNBUCM-UHFFFAOYSA-N pentyl prop-2-enoate Chemical compound CCCCCOC(=O)C=C ULDDEWDFUNBUCM-UHFFFAOYSA-N 0.000 description 2
- YNPNZTXNASCQKK-UHFFFAOYSA-N phenanthrene Chemical compound C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- ABLZXFCXXLZCGV-UHFFFAOYSA-N phosphonic acid group Chemical group P(O)(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 2
- 230000000704 physical effect Effects 0.000 description 2
- WLJVNTCWHIRURA-UHFFFAOYSA-N pimelic acid Chemical compound OC(=O)CCCCCC(O)=O WLJVNTCWHIRURA-UHFFFAOYSA-N 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 229920000259 polyoxyethylene lauryl ether Polymers 0.000 description 2
- 229920006324 polyoxymethylene Polymers 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 239000004814 polyurethane Substances 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 239000011164 primary particle Substances 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- UHDJLJWVPNZJJO-UHFFFAOYSA-N prop-1-enyl 2-methylprop-2-enoate Chemical compound CC=COC(=O)C(C)=C UHDJLJWVPNZJJO-UHFFFAOYSA-N 0.000 description 2
- UJEZSMGEGJNRNV-UHFFFAOYSA-N prop-1-enyl prop-2-enoate Chemical compound CC=COC(=O)C=C UJEZSMGEGJNRNV-UHFFFAOYSA-N 0.000 description 2
- QTECDUFMBMSHKR-UHFFFAOYSA-N prop-2-enyl prop-2-enoate Chemical compound C=CCOC(=O)C=C QTECDUFMBMSHKR-UHFFFAOYSA-N 0.000 description 2
- WPBNLDNIZUGLJL-UHFFFAOYSA-N prop-2-ynyl prop-2-enoate Chemical compound C=CC(=O)OCC#C WPBNLDNIZUGLJL-UHFFFAOYSA-N 0.000 description 2
- 235000019260 propionic acid Nutrition 0.000 description 2
- NHARPDSAXCBDDR-UHFFFAOYSA-N propyl 2-methylprop-2-enoate Chemical compound CCCOC(=O)C(C)=C NHARPDSAXCBDDR-UHFFFAOYSA-N 0.000 description 2
- PNXMTCDJUBJHQJ-UHFFFAOYSA-N propyl prop-2-enoate Chemical compound CCCOC(=O)C=C PNXMTCDJUBJHQJ-UHFFFAOYSA-N 0.000 description 2
- 239000001057 purple pigment Substances 0.000 description 2
- BBEAQIROQSPTKN-UHFFFAOYSA-N pyrene Chemical compound C1=CC=C2C=CC3=CC=CC4=CC=C1C2=C43 BBEAQIROQSPTKN-UHFFFAOYSA-N 0.000 description 2
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 2
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 description 2
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 230000027756 respiratory electron transport chain Effects 0.000 description 2
- 239000004576 sand Substances 0.000 description 2
- CXMXRPHRNRROMY-UHFFFAOYSA-N sebacic acid Chemical compound OC(=O)CCCCCCCCC(O)=O CXMXRPHRNRROMY-UHFFFAOYSA-N 0.000 description 2
- 230000001235 sensitizing effect Effects 0.000 description 2
- 239000002002 slurry Substances 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- 235000017550 sodium carbonate Nutrition 0.000 description 2
- 235000011121 sodium hydroxide Nutrition 0.000 description 2
- 239000006104 solid solution Substances 0.000 description 2
- 238000001179 sorption measurement Methods 0.000 description 2
- 238000004528 spin coating Methods 0.000 description 2
- 238000001694 spray drying Methods 0.000 description 2
- TYFQFVWCELRYAO-UHFFFAOYSA-N suberic acid Chemical compound OC(=O)CCCCCCC(O)=O TYFQFVWCELRYAO-UHFFFAOYSA-N 0.000 description 2
- 238000006467 substitution reaction Methods 0.000 description 2
- 229940014800 succinic anhydride Drugs 0.000 description 2
- 150000005846 sugar alcohols Polymers 0.000 description 2
- 125000000542 sulfonic acid group Chemical group 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 description 2
- HWCKGOZZJDHMNC-UHFFFAOYSA-M tetraethylammonium bromide Chemical compound [Br-].CC[N+](CC)(CC)CC HWCKGOZZJDHMNC-UHFFFAOYSA-M 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 description 2
- 125000003396 thiol group Chemical group [H]S* 0.000 description 2
- ANRHNWWPFJCPAZ-UHFFFAOYSA-M thionine Chemical compound [Cl-].C1=CC(N)=CC2=[S+]C3=CC(N)=CC=C3N=C21 ANRHNWWPFJCPAZ-UHFFFAOYSA-M 0.000 description 2
- KQTIIICEAUMSDG-UHFFFAOYSA-N tricarballylic acid Chemical compound OC(=O)CC(C(O)=O)CC(O)=O KQTIIICEAUMSDG-UHFFFAOYSA-N 0.000 description 2
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 2
- ARCGXLSVLAOJQL-UHFFFAOYSA-N trimellitic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 ARCGXLSVLAOJQL-UHFFFAOYSA-N 0.000 description 2
- AAAQKTZKLRYKHR-UHFFFAOYSA-N triphenylmethane Chemical compound C1=CC=CC=C1C(C=1C=CC=CC=1)C1=CC=CC=C1 AAAQKTZKLRYKHR-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- RSJKGSCJYJTIGS-UHFFFAOYSA-N undecane Chemical compound CCCCCCCCCCC RSJKGSCJYJTIGS-UHFFFAOYSA-N 0.000 description 2
- 238000001291 vacuum drying Methods 0.000 description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 2
- ZTWTYVWXUKTLCP-UHFFFAOYSA-N vinylphosphonic acid Chemical compound OP(O)(=O)C=C ZTWTYVWXUKTLCP-UHFFFAOYSA-N 0.000 description 2
- WRXCBRHBHGNNQA-UHFFFAOYSA-N (2,4-dichlorobenzoyl) 2,4-dichlorobenzenecarboperoxoate Chemical compound ClC1=CC(Cl)=CC=C1C(=O)OOC(=O)C1=CC=C(Cl)C=C1Cl WRXCBRHBHGNNQA-UHFFFAOYSA-N 0.000 description 1
- VMHYWKBKHMYRNF-UHFFFAOYSA-N (2-chlorophenyl)-phenylmethanone Chemical compound ClC1=CC=CC=C1C(=O)C1=CC=CC=C1 VMHYWKBKHMYRNF-UHFFFAOYSA-N 0.000 description 1
- CKGKXGQVRVAKEA-UHFFFAOYSA-N (2-methylphenyl)-phenylmethanone Chemical compound CC1=CC=CC=C1C(=O)C1=CC=CC=C1 CKGKXGQVRVAKEA-UHFFFAOYSA-N 0.000 description 1
- HGXJDMCMYLEZMJ-UHFFFAOYSA-N (2-methylpropan-2-yl)oxy 2,2-dimethylpropaneperoxoate Chemical compound CC(C)(C)OOOC(=O)C(C)(C)C HGXJDMCMYLEZMJ-UHFFFAOYSA-N 0.000 description 1
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- FEIQOMCWGDNMHM-KBXRYBNXSA-N (2e,4e)-5-phenylpenta-2,4-dienoic acid Chemical compound OC(=O)\C=C\C=C\C1=CC=CC=C1 FEIQOMCWGDNMHM-KBXRYBNXSA-N 0.000 description 1
- NAWXUBYGYWOOIX-SFHVURJKSA-N (2s)-2-[[4-[2-(2,4-diaminoquinazolin-6-yl)ethyl]benzoyl]amino]-4-methylidenepentanedioic acid Chemical compound C1=CC2=NC(N)=NC(N)=C2C=C1CCC1=CC=C(C(=O)N[C@@H](CC(=C)C(O)=O)C(O)=O)C=C1 NAWXUBYGYWOOIX-SFHVURJKSA-N 0.000 description 1
- BJZYYSAMLOBSDY-QMMMGPOBSA-N (2s)-2-butoxybutan-1-ol Chemical compound CCCCO[C@@H](CC)CO BJZYYSAMLOBSDY-QMMMGPOBSA-N 0.000 description 1
- OHWZQGOJLRVRIW-UHFFFAOYSA-N (3-carbamoyloxy-4-methylphenyl) carbamate Chemical compound CC1=CC=C(OC(N)=O)C=C1OC(N)=O OHWZQGOJLRVRIW-UHFFFAOYSA-N 0.000 description 1
- LECIENHMQJKJMI-UHFFFAOYSA-N (3-methoxy-3-methylbutoxy)carbonyloxyperoxy (3-methoxy-3-methylbutyl) carbonate Chemical compound COC(C)(C)CCOC(=O)OOOOC(=O)OCCC(C)(C)OC LECIENHMQJKJMI-UHFFFAOYSA-N 0.000 description 1
- URBLVRAVOIVZFJ-UHFFFAOYSA-N (3-methylphenyl)-phenylmethanone Chemical compound CC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 URBLVRAVOIVZFJ-UHFFFAOYSA-N 0.000 description 1
- KMOUUZVZFBCRAM-OLQVQODUSA-N (3as,7ar)-3a,4,7,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1C=CC[C@@H]2C(=O)OC(=O)[C@@H]21 KMOUUZVZFBCRAM-OLQVQODUSA-N 0.000 description 1
- AKOGNYJNGMLDOA-UHFFFAOYSA-N (4-acetyloxyphenyl) acetate Chemical compound CC(=O)OC1=CC=C(OC(C)=O)C=C1 AKOGNYJNGMLDOA-UHFFFAOYSA-N 0.000 description 1
- KEOLYBMGRQYQTN-UHFFFAOYSA-N (4-bromophenyl)-phenylmethanone Chemical compound C1=CC(Br)=CC=C1C(=O)C1=CC=CC=C1 KEOLYBMGRQYQTN-UHFFFAOYSA-N 0.000 description 1
- TVXNKQRAZONMHJ-UHFFFAOYSA-M (4-ethenylphenyl)methyl-trimethylazanium;chloride Chemical compound [Cl-].C[N+](C)(C)CC1=CC=C(C=C)C=C1 TVXNKQRAZONMHJ-UHFFFAOYSA-M 0.000 description 1
- WXPWZZHELZEVPO-UHFFFAOYSA-N (4-methylphenyl)-phenylmethanone Chemical compound C1=CC(C)=CC=C1C(=O)C1=CC=CC=C1 WXPWZZHELZEVPO-UHFFFAOYSA-N 0.000 description 1
- 239000001124 (E)-prop-1-ene-1,2,3-tricarboxylic acid Substances 0.000 description 1
- 229920002818 (Hydroxyethyl)methacrylate Polymers 0.000 description 1
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 1
- FGTUGLXGCCYKPJ-SPIKMXEPSA-N (Z)-but-2-enedioic acid 2-[2-(2-hydroxyethoxy)ethoxy]ethanol Chemical compound OC(=O)\C=C/C(O)=O.OC(=O)\C=C/C(O)=O.OCCOCCOCCO FGTUGLXGCCYKPJ-SPIKMXEPSA-N 0.000 description 1
- SORHAFXJCOXOIC-CCAGOZQPSA-N (z)-4-[2-[(z)-3-carboxyprop-2-enoyl]oxyethoxy]-4-oxobut-2-enoic acid Chemical compound OC(=O)\C=C/C(=O)OCCOC(=O)\C=C/C(O)=O SORHAFXJCOXOIC-CCAGOZQPSA-N 0.000 description 1
- ORTVZLZNOYNASJ-UPHRSURJSA-N (z)-but-2-ene-1,4-diol Chemical compound OC\C=C/CO ORTVZLZNOYNASJ-UPHRSURJSA-N 0.000 description 1
- WBYWAXJHAXSJNI-VOTSOKGWSA-M .beta-Phenylacrylic acid Natural products [O-]C(=O)\C=C\C1=CC=CC=C1 WBYWAXJHAXSJNI-VOTSOKGWSA-M 0.000 description 1
- NALFRYPTRXKZPN-UHFFFAOYSA-N 1,1-bis(tert-butylperoxy)-3,3,5-trimethylcyclohexane Chemical compound CC1CC(C)(C)CC(OOC(C)(C)C)(OOC(C)(C)C)C1 NALFRYPTRXKZPN-UHFFFAOYSA-N 0.000 description 1
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical compound C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 1
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 1
- GJZFGDYLJLCGHT-UHFFFAOYSA-N 1,2-diethylthioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=C(CC)C(CC)=CC=C3SC2=C1 GJZFGDYLJLCGHT-UHFFFAOYSA-N 0.000 description 1
- ZXHZWRZAWJVPIC-UHFFFAOYSA-N 1,2-diisocyanatonaphthalene Chemical compound C1=CC=CC2=C(N=C=O)C(N=C=O)=CC=C21 ZXHZWRZAWJVPIC-UHFFFAOYSA-N 0.000 description 1
- MSAHTMIQULFMRG-UHFFFAOYSA-N 1,2-diphenyl-2-propan-2-yloxyethanone Chemical compound C=1C=CC=CC=1C(OC(C)C)C(=O)C1=CC=CC=C1 MSAHTMIQULFMRG-UHFFFAOYSA-N 0.000 description 1
- KEQXNNJHMWSZHK-UHFFFAOYSA-L 1,3,2,4$l^{2}-dioxathiaplumbetane 2,2-dioxide Chemical compound [Pb+2].[O-]S([O-])(=O)=O KEQXNNJHMWSZHK-UHFFFAOYSA-L 0.000 description 1
- BPXVHIRIPLPOPT-UHFFFAOYSA-N 1,3,5-tris(2-hydroxyethyl)-1,3,5-triazinane-2,4,6-trione Chemical compound OCCN1C(=O)N(CCO)C(=O)N(CCO)C1=O BPXVHIRIPLPOPT-UHFFFAOYSA-N 0.000 description 1
- VDYWHVQKENANGY-UHFFFAOYSA-N 1,3-Butyleneglycol dimethacrylate Chemical compound CC(=C)C(=O)OC(C)CCOC(=O)C(C)=C VDYWHVQKENANGY-UHFFFAOYSA-N 0.000 description 1
- RTTZISZSHSCFRH-UHFFFAOYSA-N 1,3-bis(isocyanatomethyl)benzene Chemical compound O=C=NCC1=CC=CC(CN=C=O)=C1 RTTZISZSHSCFRH-UHFFFAOYSA-N 0.000 description 1
- YHMYGUUIMTVXNW-UHFFFAOYSA-N 1,3-dihydrobenzimidazole-2-thione Chemical compound C1=CC=C2NC(S)=NC2=C1 YHMYGUUIMTVXNW-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- OHLKMGYGBHFODF-UHFFFAOYSA-N 1,4-bis(isocyanatomethyl)benzene Chemical compound O=C=NCC1=CC=C(CN=C=O)C=C1 OHLKMGYGBHFODF-UHFFFAOYSA-N 0.000 description 1
- SBJCUZQNHOLYMD-UHFFFAOYSA-N 1,5-Naphthalene diisocyanate Chemical compound C1=CC=C2C(N=C=O)=CC=CC2=C1N=C=O SBJCUZQNHOLYMD-UHFFFAOYSA-N 0.000 description 1
- VZXPHDGHQXLXJC-UHFFFAOYSA-N 1,6-diisocyanato-5,6-dimethylheptane Chemical compound O=C=NC(C)(C)C(C)CCCCN=C=O VZXPHDGHQXLXJC-UHFFFAOYSA-N 0.000 description 1
- 229940008841 1,6-hexamethylene diisocyanate Drugs 0.000 description 1
- DKEGCUDAFWNSSO-UHFFFAOYSA-N 1,8-dibromooctane Chemical compound BrCCCCCCCCBr DKEGCUDAFWNSSO-UHFFFAOYSA-N 0.000 description 1
- AZUXKVXMJOIAOF-UHFFFAOYSA-N 1-(2-hydroxypropoxy)propan-2-ol Chemical compound CC(O)COCC(C)O AZUXKVXMJOIAOF-UHFFFAOYSA-N 0.000 description 1
- OGBWMWKMTUSNKE-UHFFFAOYSA-N 1-(2-methylprop-2-enoyloxy)hexyl 2-methylprop-2-enoate Chemical compound CCCCCC(OC(=O)C(C)=C)OC(=O)C(C)=C OGBWMWKMTUSNKE-UHFFFAOYSA-N 0.000 description 1
- QOZZIBSMAKODRZ-UHFFFAOYSA-M 1-[(4-ethenylphenyl)methyl]pyridin-1-ium;chloride Chemical compound [Cl-].C1=CC(C=C)=CC=C1C[N+]1=CC=CC=C1 QOZZIBSMAKODRZ-UHFFFAOYSA-M 0.000 description 1
- VLDPXPPHXDGHEW-UHFFFAOYSA-N 1-chloro-2-dichlorophosphoryloxybenzene Chemical compound ClC1=CC=CC=C1OP(Cl)(Cl)=O VLDPXPPHXDGHEW-UHFFFAOYSA-N 0.000 description 1
- YNSNJGRCQCDRDM-UHFFFAOYSA-N 1-chlorothioxanthen-9-one Chemical compound S1C2=CC=CC=C2C(=O)C2=C1C=CC=C2Cl YNSNJGRCQCDRDM-UHFFFAOYSA-N 0.000 description 1
- UAJRSHJHFRVGMG-UHFFFAOYSA-N 1-ethenyl-4-methoxybenzene Chemical class COC1=CC=C(C=C)C=C1 UAJRSHJHFRVGMG-UHFFFAOYSA-N 0.000 description 1
- OTHANJXFOXIOLL-UHFFFAOYSA-N 1-hydroxy-1-(2-methylphenyl)propan-2-one Chemical compound CC(=O)C(O)C1=CC=CC=C1C OTHANJXFOXIOLL-UHFFFAOYSA-N 0.000 description 1
- 239000012956 1-hydroxycyclohexylphenyl-ketone Substances 0.000 description 1
- ICLCCFKUSALICQ-UHFFFAOYSA-N 1-isocyanato-4-(4-isocyanato-3-methylphenyl)-2-methylbenzene Chemical compound C1=C(N=C=O)C(C)=CC(C=2C=C(C)C(N=C=O)=CC=2)=C1 ICLCCFKUSALICQ-UHFFFAOYSA-N 0.000 description 1
- RTBFRGCFXZNCOE-UHFFFAOYSA-N 1-methylsulfonylpiperidin-4-one Chemical compound CS(=O)(=O)N1CCC(=O)CC1 RTBFRGCFXZNCOE-UHFFFAOYSA-N 0.000 description 1
- XLPJNCYCZORXHG-UHFFFAOYSA-N 1-morpholin-4-ylprop-2-en-1-one Chemical compound C=CC(=O)N1CCOCC1 XLPJNCYCZORXHG-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- VOBUAPTXJKMNCT-UHFFFAOYSA-N 1-prop-2-enoyloxyhexyl prop-2-enoate Chemical compound CCCCCC(OC(=O)C=C)OC(=O)C=C VOBUAPTXJKMNCT-UHFFFAOYSA-N 0.000 description 1
- YIKSHDNOAYSSPX-UHFFFAOYSA-N 1-propan-2-ylthioxanthen-9-one Chemical compound S1C2=CC=CC=C2C(=O)C2=C1C=CC=C2C(C)C YIKSHDNOAYSSPX-UHFFFAOYSA-N 0.000 description 1
- WJFKNYWRSNBZNX-UHFFFAOYSA-N 10H-phenothiazine Chemical compound C1=CC=C2NC3=CC=CC=C3SC2=C1 WJFKNYWRSNBZNX-UHFFFAOYSA-N 0.000 description 1
- KGRVJHAUYBGFFP-UHFFFAOYSA-N 2,2'-Methylenebis(4-methyl-6-tert-butylphenol) Chemical compound CC(C)(C)C1=CC(C)=CC(CC=2C(=C(C=C(C)C=2)C(C)(C)C)O)=C1O KGRVJHAUYBGFFP-UHFFFAOYSA-N 0.000 description 1
- CDPPYCZVWYZBJH-UHFFFAOYSA-N 2,2,3,3-tetramethylbutanedioic acid Chemical compound OC(=O)C(C)(C)C(C)(C)C(O)=O CDPPYCZVWYZBJH-UHFFFAOYSA-N 0.000 description 1
- JCTXKRPTIMZBJT-UHFFFAOYSA-N 2,2,4-trimethylpentane-1,3-diol Chemical compound CC(C)C(O)C(C)(C)CO JCTXKRPTIMZBJT-UHFFFAOYSA-N 0.000 description 1
- SCBGJZIOPNAEMH-UHFFFAOYSA-N 2,2-bis(4-hydroxyphenyl)acetic acid Chemical compound C=1C=C(O)C=CC=1C(C(=O)O)C1=CC=C(O)C=C1 SCBGJZIOPNAEMH-UHFFFAOYSA-N 0.000 description 1
- HQOVXPHOJANJBR-UHFFFAOYSA-N 2,2-bis(tert-butylperoxy)butane Chemical compound CC(C)(C)OOC(C)(CC)OOC(C)(C)C HQOVXPHOJANJBR-UHFFFAOYSA-N 0.000 description 1
- PIZHFBODNLEQBL-UHFFFAOYSA-N 2,2-diethoxy-1-phenylethanone Chemical compound CCOC(OCC)C(=O)C1=CC=CC=C1 PIZHFBODNLEQBL-UHFFFAOYSA-N 0.000 description 1
- KWVGIHKZDCUPEU-UHFFFAOYSA-N 2,2-dimethoxy-2-phenylacetophenone Chemical compound C=1C=CC=CC=1C(OC)(OC)C(=O)C1=CC=CC=C1 KWVGIHKZDCUPEU-UHFFFAOYSA-N 0.000 description 1
- RIZUCYSQUWMQLX-UHFFFAOYSA-N 2,3-dimethylbenzoic acid Chemical compound CC1=CC=CC(C(O)=O)=C1C RIZUCYSQUWMQLX-UHFFFAOYSA-N 0.000 description 1
- QWQNFXDYOCUEER-UHFFFAOYSA-N 2,3-ditert-butyl-4-methylphenol Chemical compound CC1=CC=C(O)C(C(C)(C)C)=C1C(C)(C)C QWQNFXDYOCUEER-UHFFFAOYSA-N 0.000 description 1
- WCVOGSZTONGSQY-UHFFFAOYSA-N 2,4,6-trichloroanisole Chemical compound COC1=C(Cl)C=C(Cl)C=C1Cl WCVOGSZTONGSQY-UHFFFAOYSA-N 0.000 description 1
- RICRAVHJCLFPFF-UHFFFAOYSA-N 2,4,6-tris(chloromethyl)-1,3,5-triazine Chemical compound ClCC1=NC(CCl)=NC(CCl)=N1 RICRAVHJCLFPFF-UHFFFAOYSA-N 0.000 description 1
- LNRJBPCTMHMOFA-UHFFFAOYSA-N 2,4,6-tris(dichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)C1=NC(C(Cl)Cl)=NC(C(Cl)Cl)=N1 LNRJBPCTMHMOFA-UHFFFAOYSA-N 0.000 description 1
- URJAUSYMVIZTHC-UHFFFAOYSA-N 2,4,6-tris(tribromomethyl)-1,3,5-triazine Chemical compound BrC(Br)(Br)C1=NC(C(Br)(Br)Br)=NC(C(Br)(Br)Br)=N1 URJAUSYMVIZTHC-UHFFFAOYSA-N 0.000 description 1
- DXUMYHZTYVPBEZ-UHFFFAOYSA-N 2,4,6-tris(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 DXUMYHZTYVPBEZ-UHFFFAOYSA-N 0.000 description 1
- BRKORVYTKKLNKX-UHFFFAOYSA-N 2,4-di(propan-2-yl)thioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC(C(C)C)=CC(C(C)C)=C3SC2=C1 BRKORVYTKKLNKX-UHFFFAOYSA-N 0.000 description 1
- BTJPUDCSZVCXFQ-UHFFFAOYSA-N 2,4-diethylthioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC(CC)=CC(CC)=C3SC2=C1 BTJPUDCSZVCXFQ-UHFFFAOYSA-N 0.000 description 1
- HGEFWFBFQKWVMY-DUXPYHPUSA-N 2,4-dihydroxy-trans cinnamic acid Chemical compound OC(=O)\C=C\C1=CC=C(O)C=C1O HGEFWFBFQKWVMY-DUXPYHPUSA-N 0.000 description 1
- BEVWMRQFVUOPJT-UHFFFAOYSA-N 2,4-dimethyl-1,3-thiazole-5-carboxamide Chemical compound CC1=NC(C)=C(C(N)=O)S1 BEVWMRQFVUOPJT-UHFFFAOYSA-N 0.000 description 1
- LCHAFMWSFCONOO-UHFFFAOYSA-N 2,4-dimethylthioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC(C)=CC(C)=C3SC2=C1 LCHAFMWSFCONOO-UHFFFAOYSA-N 0.000 description 1
- DMWVYCCGCQPJEA-UHFFFAOYSA-N 2,5-bis(tert-butylperoxy)-2,5-dimethylhexane Chemical compound CC(C)(C)OOC(C)(C)CCC(C)(C)OOC(C)(C)C DMWVYCCGCQPJEA-UHFFFAOYSA-N 0.000 description 1
- BFQFFNWLTHFJOZ-UHFFFAOYSA-N 2-(1,3-benzodioxol-5-yl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=2C=C3OCOC3=CC=2)=N1 BFQFFNWLTHFJOZ-UHFFFAOYSA-N 0.000 description 1
- VJBPOYPFDHMOSM-UHFFFAOYSA-N 2-(2,2-dihydroxyethylsulfonyl)ethane-1,1-diol Chemical compound OC(O)CS(=O)(=O)CC(O)O VJBPOYPFDHMOSM-UHFFFAOYSA-N 0.000 description 1
- GBOJZXLCJZDBKO-UHFFFAOYSA-N 2-(2-chlorophenyl)-2-[2-(2-chlorophenyl)-4,5-diphenylimidazol-2-yl]-4,5-diphenylimidazole Chemical compound ClC1=CC=CC=C1C1(C2(N=C(C(=N2)C=2C=CC=CC=2)C=2C=CC=CC=2)C=2C(=CC=CC=2)Cl)N=C(C=2C=CC=CC=2)C(C=2C=CC=CC=2)=N1 GBOJZXLCJZDBKO-UHFFFAOYSA-N 0.000 description 1
- SBASXUCJHJRPEV-UHFFFAOYSA-N 2-(2-methoxyethoxy)ethanol Chemical compound COCCOCCO SBASXUCJHJRPEV-UHFFFAOYSA-N 0.000 description 1
- FNHQLSVILKHZNI-UHFFFAOYSA-N 2-(2-nitrophenyl)-2-[2-(2-nitrophenyl)-4,5-diphenylimidazol-2-yl]-4,5-diphenylimidazole Chemical compound [O-][N+](=O)C1=CC=CC=C1C1(C2(N=C(C(=N2)C=2C=CC=CC=2)C=2C=CC=CC=2)C=2C(=CC=CC=2)[N+]([O-])=O)N=C(C=2C=CC=CC=2)C(C=2C=CC=CC=2)=N1 FNHQLSVILKHZNI-UHFFFAOYSA-N 0.000 description 1
- DQMOHZLFVGYNAN-UHFFFAOYSA-N 2-(2-phenylethenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=CC=2C=CC=CC=2)=N1 DQMOHZLFVGYNAN-UHFFFAOYSA-N 0.000 description 1
- XMNIXWIUMCBBBL-UHFFFAOYSA-N 2-(2-phenylpropan-2-ylperoxy)propan-2-ylbenzene Chemical compound C=1C=CC=CC=1C(C)(C)OOC(C)(C)C1=CC=CC=C1 XMNIXWIUMCBBBL-UHFFFAOYSA-N 0.000 description 1
- OEPOKWHJYJXUGD-UHFFFAOYSA-N 2-(3-phenylmethoxyphenyl)-1,3-thiazole-4-carbaldehyde Chemical compound O=CC1=CSC(C=2C=C(OCC=3C=CC=CC=3)C=CC=2)=N1 OEPOKWHJYJXUGD-UHFFFAOYSA-N 0.000 description 1
- WJKHYAJKIXYSHS-UHFFFAOYSA-N 2-(4-chlorophenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(Cl)=CC=C1C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 WJKHYAJKIXYSHS-UHFFFAOYSA-N 0.000 description 1
- CTNICFBTUIFPOE-UHFFFAOYSA-N 2-(4-hydroxyphenoxy)ethane-1,1-diol Chemical compound OC(O)COC1=CC=C(O)C=C1 CTNICFBTUIFPOE-UHFFFAOYSA-N 0.000 description 1
- MPNIGZBDAMWHSX-UHFFFAOYSA-N 2-(4-methylphenyl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(C)=CC=C1C1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 MPNIGZBDAMWHSX-UHFFFAOYSA-N 0.000 description 1
- XLQIRWWNVOTDQS-UHFFFAOYSA-N 2-(7-oxabicyclo[4.1.0]hepta-1(6),2,4-trien-4-yl)-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=2C=C3OC3=CC=2)=N1 XLQIRWWNVOTDQS-UHFFFAOYSA-N 0.000 description 1
- CWQZLVPKEVXEOB-UHFFFAOYSA-N 2-(tribromomethyl)-1,3,5-triazine Chemical compound BrC(Br)(Br)C1=NC=NC=N1 CWQZLVPKEVXEOB-UHFFFAOYSA-N 0.000 description 1
- OXQGTIUCKGYOAA-UHFFFAOYSA-N 2-Ethylbutanoic acid Chemical compound CCC(CC)C(O)=O OXQGTIUCKGYOAA-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- PTTPXKJBFFKCEK-UHFFFAOYSA-N 2-Methyl-4-heptanone Chemical compound CC(C)CC(=O)CC(C)C PTTPXKJBFFKCEK-UHFFFAOYSA-N 0.000 description 1
- RFCQDOVPMUSZMN-UHFFFAOYSA-N 2-Naphthalenethiol Chemical compound C1=CC=CC2=CC(S)=CC=C21 RFCQDOVPMUSZMN-UHFFFAOYSA-N 0.000 description 1
- APJRQJNSYFWQJD-GGWOSOGESA-N 2-[(e)-but-2-enoyl]oxyethyl (e)-but-2-enoate Chemical compound C\C=C\C(=O)OCCOC(=O)\C=C\C APJRQJNSYFWQJD-GGWOSOGESA-N 0.000 description 1
- APJRQJNSYFWQJD-GLIMQPGKSA-N 2-[(z)-but-2-enoyl]oxyethyl (z)-but-2-enoate Chemical compound C\C=C/C(=O)OCCOC(=O)\C=C/C APJRQJNSYFWQJD-GLIMQPGKSA-N 0.000 description 1
- MXPYJVUYLVNEBB-UHFFFAOYSA-N 2-[2-(2-carboxybenzoyl)oxycarbonylbenzoyl]oxycarbonylbenzoic acid Chemical compound OC(=O)C1=CC=CC=C1C(=O)OC(=O)C1=CC=CC=C1C(=O)OC(=O)C1=CC=CC=C1C(O)=O MXPYJVUYLVNEBB-UHFFFAOYSA-N 0.000 description 1
- YJGHMLJGPSVSLF-UHFFFAOYSA-N 2-[2-(2-octanoyloxyethoxy)ethoxy]ethyl octanoate Chemical compound CCCCCCCC(=O)OCCOCCOCCOC(=O)CCCCCCC YJGHMLJGPSVSLF-UHFFFAOYSA-N 0.000 description 1
- MCNPOZMLKGDJGP-UHFFFAOYSA-N 2-[2-(4-methoxyphenyl)ethenyl]-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound C1=CC(OC)=CC=C1C=CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 MCNPOZMLKGDJGP-UHFFFAOYSA-N 0.000 description 1
- HWSSEYVMGDIFMH-UHFFFAOYSA-N 2-[2-[2-(2-methylprop-2-enoyloxy)ethoxy]ethoxy]ethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCOCCOCCOC(=O)C(C)=C HWSSEYVMGDIFMH-UHFFFAOYSA-N 0.000 description 1
- VIYWVRIBDZTTMH-UHFFFAOYSA-N 2-[4-[2-[4-[2-(2-methylprop-2-enoyloxy)ethoxy]phenyl]propan-2-yl]phenoxy]ethyl 2-methylprop-2-enoate Chemical compound C1=CC(OCCOC(=O)C(=C)C)=CC=C1C(C)(C)C1=CC=C(OCCOC(=O)C(C)=C)C=C1 VIYWVRIBDZTTMH-UHFFFAOYSA-N 0.000 description 1
- HDDQXUDCEIMISH-UHFFFAOYSA-N 2-[[4-[1,2,2-tris[4-(oxiran-2-ylmethoxy)phenyl]ethyl]phenoxy]methyl]oxirane Chemical compound C1OC1COC(C=C1)=CC=C1C(C=1C=CC(OCC2OC2)=CC=1)C(C=1C=CC(OCC2OC2)=CC=1)C(C=C1)=CC=C1OCC1CO1 HDDQXUDCEIMISH-UHFFFAOYSA-N 0.000 description 1
- XGYZGXHSCJSEBK-UHFFFAOYSA-N 2-[[4-[3,5-dimethyl-4-(oxiran-2-ylmethyl)phenyl]-2,6-dimethylphenyl]methyl]oxirane Chemical group CC1=CC(C=2C=C(C)C(CC3OC3)=C(C)C=2)=CC(C)=C1CC1CO1 XGYZGXHSCJSEBK-UHFFFAOYSA-N 0.000 description 1
- IGZBSJAMZHNHKE-UHFFFAOYSA-N 2-[[4-[bis[4-(oxiran-2-ylmethoxy)phenyl]methyl]phenoxy]methyl]oxirane Chemical compound C1OC1COC(C=C1)=CC=C1C(C=1C=CC(OCC2OC2)=CC=1)C(C=C1)=CC=C1OCC1CO1 IGZBSJAMZHNHKE-UHFFFAOYSA-N 0.000 description 1
- FGTYTUFKXYPTML-UHFFFAOYSA-N 2-benzoylbenzoic acid Chemical compound OC(=O)C1=CC=CC=C1C(=O)C1=CC=CC=C1 FGTYTUFKXYPTML-UHFFFAOYSA-N 0.000 description 1
- RXJXDPDHNAYULH-UHFFFAOYSA-N 2-benzyl-2-(dimethylamino)-4-morpholin-4-yl-1-phenylbutan-1-one Chemical compound C=1C=CC=CC=1CC(C(=O)C=1C=CC=CC=1)(N(C)C)CCN1CCOCC1 RXJXDPDHNAYULH-UHFFFAOYSA-N 0.000 description 1
- QEMUVGKPNFMGAZ-UHFFFAOYSA-N 2-benzylsulfanyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(SCC=2C=CC=CC=2)=N1 QEMUVGKPNFMGAZ-UHFFFAOYSA-N 0.000 description 1
- 125000006276 2-bromophenyl group Chemical group [H]C1=C([H])C(Br)=C(*)C([H])=C1[H] 0.000 description 1
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 description 1
- ZCDADJXRUCOCJE-UHFFFAOYSA-N 2-chlorothioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC(Cl)=CC=C3SC2=C1 ZCDADJXRUCOCJE-UHFFFAOYSA-N 0.000 description 1
- KRDXTHSSNCTAGY-UHFFFAOYSA-N 2-cyclohexylpyrrolidine Chemical compound C1CCNC1C1CCCCC1 KRDXTHSSNCTAGY-UHFFFAOYSA-N 0.000 description 1
- ZVUNTIMPQCQCAQ-UHFFFAOYSA-N 2-dodecanoyloxyethyl dodecanoate Chemical compound CCCCCCCCCCCC(=O)OCCOC(=O)CCCCCCCCCCC ZVUNTIMPQCQCAQ-UHFFFAOYSA-N 0.000 description 1
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 description 1
- VGZZAZYCLRYTNQ-UHFFFAOYSA-N 2-ethoxyethoxycarbonyloxy 2-ethoxyethyl carbonate Chemical compound CCOCCOC(=O)OOC(=O)OCCOCC VGZZAZYCLRYTNQ-UHFFFAOYSA-N 0.000 description 1
- LZDXRPVSAKWYDH-UHFFFAOYSA-N 2-ethyl-2-(prop-2-enoxymethyl)propane-1,3-diol Chemical compound CCC(CO)(CO)COCC=C LZDXRPVSAKWYDH-UHFFFAOYSA-N 0.000 description 1
- YJQMXVDKXSQCDI-UHFFFAOYSA-N 2-ethylthioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC(CC)=CC=C3SC2=C1 YJQMXVDKXSQCDI-UHFFFAOYSA-N 0.000 description 1
- MIRQGKQPLPBZQM-UHFFFAOYSA-N 2-hydroperoxy-2,4,4-trimethylpentane Chemical compound CC(C)(C)CC(C)(C)OO MIRQGKQPLPBZQM-UHFFFAOYSA-N 0.000 description 1
- RLHGFJMGWQXPBW-UHFFFAOYSA-N 2-hydroxy-3-(1h-imidazol-5-ylmethyl)benzamide Chemical compound NC(=O)C1=CC=CC(CC=2NC=NC=2)=C1O RLHGFJMGWQXPBW-UHFFFAOYSA-N 0.000 description 1
- 125000000954 2-hydroxyethyl group Chemical group [H]C([*])([H])C([H])([H])O[H] 0.000 description 1
- FLFWJIBUZQARMD-UHFFFAOYSA-N 2-mercapto-1,3-benzoxazole Chemical compound C1=CC=C2OC(S)=NC2=C1 FLFWJIBUZQARMD-UHFFFAOYSA-N 0.000 description 1
- BQZJOQXSCSZQPS-UHFFFAOYSA-N 2-methoxy-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(OC)C(=O)C1=CC=CC=C1 BQZJOQXSCSZQPS-UHFFFAOYSA-N 0.000 description 1
- LWRBVKNFOYUCNP-UHFFFAOYSA-N 2-methyl-1-(4-methylsulfanylphenyl)-2-morpholin-4-ylpropan-1-one Chemical compound C1=CC(SC)=CC=C1C(=O)C(C)(C)N1CCOCC1 LWRBVKNFOYUCNP-UHFFFAOYSA-N 0.000 description 1
- AKVUWTYSNLGBJY-UHFFFAOYSA-N 2-methyl-1-morpholin-4-ylprop-2-en-1-one Chemical compound CC(=C)C(=O)N1CCOCC1 AKVUWTYSNLGBJY-UHFFFAOYSA-N 0.000 description 1
- GOTIJEQQGSMAIN-UHFFFAOYSA-N 2-methyl-4,6-bis(tribromomethyl)-1,3,5-triazine Chemical compound CC1=NC(C(Br)(Br)Br)=NC(C(Br)(Br)Br)=N1 GOTIJEQQGSMAIN-UHFFFAOYSA-N 0.000 description 1
- LETDRANQSOEVCX-UHFFFAOYSA-N 2-methyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound CC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 LETDRANQSOEVCX-UHFFFAOYSA-N 0.000 description 1
- TURITJIWSQEMDB-UHFFFAOYSA-N 2-methyl-n-[(2-methylprop-2-enoylamino)methyl]prop-2-enamide Chemical compound CC(=C)C(=O)NCNC(=O)C(C)=C TURITJIWSQEMDB-UHFFFAOYSA-N 0.000 description 1
- YBKWKURHPIBUEM-UHFFFAOYSA-N 2-methyl-n-[6-(2-methylprop-2-enoylamino)hexyl]prop-2-enamide Chemical compound CC(=C)C(=O)NCCCCCCNC(=O)C(C)=C YBKWKURHPIBUEM-UHFFFAOYSA-N 0.000 description 1
- GDHSRTFITZTMMP-UHFFFAOYSA-N 2-methylidenebutanedioic acid;propane-1,2-diol Chemical compound CC(O)CO.OC(=O)CC(=C)C(O)=O.OC(=O)CC(=C)C(O)=O GDHSRTFITZTMMP-UHFFFAOYSA-N 0.000 description 1
- HHCHLHOEAKKCAB-UHFFFAOYSA-N 2-oxaspiro[3.5]nonane-1,3-dione Chemical compound O=C1OC(=O)C11CCCCC1 HHCHLHOEAKKCAB-UHFFFAOYSA-N 0.000 description 1
- HAZQZUFYRLFOLC-UHFFFAOYSA-N 2-phenyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(C=2C=CC=CC=2)=N1 HAZQZUFYRLFOLC-UHFFFAOYSA-N 0.000 description 1
- SCZHWMVQZILSGJ-UHFFFAOYSA-N 2-phenylacetic acid;2-phenylpropanoic acid Chemical compound OC(=O)CC1=CC=CC=C1.OC(=O)C(C)C1=CC=CC=C1 SCZHWMVQZILSGJ-UHFFFAOYSA-N 0.000 description 1
- LVFFZQQWIZURIO-UHFFFAOYSA-N 2-phenylbutanedioic acid Chemical compound OC(=O)CC(C(O)=O)C1=CC=CC=C1 LVFFZQQWIZURIO-UHFFFAOYSA-N 0.000 description 1
- AGBXYHCHUYARJY-UHFFFAOYSA-N 2-phenylethenesulfonic acid Chemical compound OS(=O)(=O)C=CC1=CC=CC=C1 AGBXYHCHUYARJY-UHFFFAOYSA-N 0.000 description 1
- XLLXMBCBJGATSP-UHFFFAOYSA-N 2-phenylethenol Chemical compound OC=CC1=CC=CC=C1 XLLXMBCBJGATSP-UHFFFAOYSA-N 0.000 description 1
- ONPJWQSDZCGSQM-UHFFFAOYSA-N 2-phenylprop-2-enoic acid Chemical compound OC(=O)C(=C)C1=CC=CC=C1 ONPJWQSDZCGSQM-UHFFFAOYSA-N 0.000 description 1
- RCUOAHPSDFRHOR-UHFFFAOYSA-N 2-phenylsulfanyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound ClC(Cl)(Cl)C1=NC(C(Cl)(Cl)Cl)=NC(SC=2C=CC=CC=2)=N1 RCUOAHPSDFRHOR-UHFFFAOYSA-N 0.000 description 1
- VFZKVQVQOMDJEG-UHFFFAOYSA-N 2-prop-2-enoyloxypropyl prop-2-enoate Chemical compound C=CC(=O)OC(C)COC(=O)C=C VFZKVQVQOMDJEG-UHFFFAOYSA-N 0.000 description 1
- KTALPKYXQZGAEG-UHFFFAOYSA-N 2-propan-2-ylthioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC(C(C)C)=CC=C3SC2=C1 KTALPKYXQZGAEG-UHFFFAOYSA-N 0.000 description 1
- DOSGQNSHFPTAOA-UHFFFAOYSA-N 2-propyl-4,6-bis(trichloromethyl)-1,3,5-triazine Chemical compound CCCC1=NC(C(Cl)(Cl)Cl)=NC(C(Cl)(Cl)Cl)=N1 DOSGQNSHFPTAOA-UHFFFAOYSA-N 0.000 description 1
- RVBUGGBMJDPOST-UHFFFAOYSA-N 2-thiobarbituric acid Chemical class O=C1CC(=O)NC(=S)N1 RVBUGGBMJDPOST-UHFFFAOYSA-N 0.000 description 1
- KGIGUEBEKRSTEW-UHFFFAOYSA-N 2-vinylpyridine Chemical compound C=CC1=CC=CC=N1 KGIGUEBEKRSTEW-UHFFFAOYSA-N 0.000 description 1
- XBNVWXKPFORCRI-UHFFFAOYSA-N 2h-naphtho[2,3-f]quinolin-1-one Chemical compound C1=CC=CC2=CC3=C4C(=O)CC=NC4=CC=C3C=C21 XBNVWXKPFORCRI-UHFFFAOYSA-N 0.000 description 1
- KKAJSJJFBSOMGS-UHFFFAOYSA-N 3,6-diamino-10-methylacridinium chloride Chemical compound [Cl-].C1=C(N)C=C2[N+](C)=C(C=C(N)C=C3)C3=CC2=C1 KKAJSJJFBSOMGS-UHFFFAOYSA-N 0.000 description 1
- GOLORTLGFDVFDW-UHFFFAOYSA-N 3-(1h-benzimidazol-2-yl)-7-(diethylamino)chromen-2-one Chemical compound C1=CC=C2NC(C3=CC4=CC=C(C=C4OC3=O)N(CC)CC)=NC2=C1 GOLORTLGFDVFDW-UHFFFAOYSA-N 0.000 description 1
- FRIBMENBGGCKPD-UHFFFAOYSA-N 3-(2,3-dimethoxyphenyl)prop-2-enal Chemical compound COC1=CC=CC(C=CC=O)=C1OC FRIBMENBGGCKPD-UHFFFAOYSA-N 0.000 description 1
- SBWOBTUYQXLKSS-UHFFFAOYSA-N 3-(2-methylprop-2-enoyloxy)propanoic acid Chemical compound CC(=C)C(=O)OCCC(O)=O SBWOBTUYQXLKSS-UHFFFAOYSA-N 0.000 description 1
- XYFRHHAYSXIKGH-UHFFFAOYSA-N 3-(5-methoxy-2-methoxycarbonyl-1h-indol-3-yl)prop-2-enoic acid Chemical compound C1=C(OC)C=C2C(C=CC(O)=O)=C(C(=O)OC)NC2=C1 XYFRHHAYSXIKGH-UHFFFAOYSA-N 0.000 description 1
- HTZFLOOLLIJZEQ-UHFFFAOYSA-N 3-butyl-5-(dimethylamino)-3-phenyl-4-(1,3,5-triazin-2-ylamino)-4h-chromen-2-one Chemical compound C1=CC=C(N(C)C)C2=C1OC(=O)C(CCCC)(C=1C=CC=CC=1)C2NC1=NC=NC=N1 HTZFLOOLLIJZEQ-UHFFFAOYSA-N 0.000 description 1
- ULMZOZMSDIOZAF-UHFFFAOYSA-N 3-hydroxy-2-(hydroxymethyl)propanoic acid Chemical compound OCC(CO)C(O)=O ULMZOZMSDIOZAF-UHFFFAOYSA-N 0.000 description 1
- VISOTGQYFFULBK-UHFFFAOYSA-N 3-hydroxy-4-phenylpyrrole-2,5-dione Chemical compound O=C1C(=O)NC(O)=C1C1=CC=CC=C1 VISOTGQYFFULBK-UHFFFAOYSA-N 0.000 description 1
- OFNISBHGPNMTMS-UHFFFAOYSA-N 3-methylideneoxolane-2,5-dione Chemical compound C=C1CC(=O)OC1=O OFNISBHGPNMTMS-UHFFFAOYSA-N 0.000 description 1
- IYMZEPRSPLASMS-UHFFFAOYSA-N 3-phenylpyrrole-2,5-dione Chemical compound O=C1NC(=O)C(C=2C=CC=CC=2)=C1 IYMZEPRSPLASMS-UHFFFAOYSA-N 0.000 description 1
- FQMIAEWUVYWVNB-UHFFFAOYSA-N 3-prop-2-enoyloxybutyl prop-2-enoate Chemical compound C=CC(=O)OC(C)CCOC(=O)C=C FQMIAEWUVYWVNB-UHFFFAOYSA-N 0.000 description 1
- CYUZOYPRAQASLN-UHFFFAOYSA-N 3-prop-2-enoyloxypropanoic acid Chemical compound OC(=O)CCOC(=O)C=C CYUZOYPRAQASLN-UHFFFAOYSA-N 0.000 description 1
- JIGUICYYOYEXFS-UHFFFAOYSA-N 3-tert-butylbenzene-1,2-diol Chemical compound CC(C)(C)C1=CC=CC(O)=C1O JIGUICYYOYEXFS-UHFFFAOYSA-N 0.000 description 1
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 1
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 1
- ZEHOVWPIGREOPO-UHFFFAOYSA-N 4,5,6,7-tetrachloro-2-[2-(4,5,6,7-tetrachloro-1,3-dioxoinden-2-yl)quinolin-8-yl]isoindole-1,3-dione Chemical compound O=C1C(C(=C(Cl)C(Cl)=C2Cl)Cl)=C2C(=O)N1C(C1=N2)=CC=CC1=CC=C2C1C(=O)C2=C(Cl)C(Cl)=C(Cl)C(Cl)=C2C1=O ZEHOVWPIGREOPO-UHFFFAOYSA-N 0.000 description 1
- IICCLYANAQEHCI-UHFFFAOYSA-N 4,5,6,7-tetrachloro-3',6'-dihydroxy-2',4',5',7'-tetraiodospiro[2-benzofuran-3,9'-xanthene]-1-one Chemical compound O1C(=O)C(C(=C(Cl)C(Cl)=C2Cl)Cl)=C2C21C1=CC(I)=C(O)C(I)=C1OC1=C(I)C(O)=C(I)C=C21 IICCLYANAQEHCI-UHFFFAOYSA-N 0.000 description 1
- XOJWAAUYNWGQAU-UHFFFAOYSA-N 4-(2-methylprop-2-enoyloxy)butyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCCCOC(=O)C(C)=C XOJWAAUYNWGQAU-UHFFFAOYSA-N 0.000 description 1
- AVCOFPOLGHKJQB-UHFFFAOYSA-N 4-(3,4-dicarboxyphenyl)sulfonylphthalic acid Chemical compound C1=C(C(O)=O)C(C(=O)O)=CC=C1S(=O)(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 AVCOFPOLGHKJQB-UHFFFAOYSA-N 0.000 description 1
- MKTOIPPVFPJEQO-UHFFFAOYSA-N 4-(3-carboxypropanoylperoxy)-4-oxobutanoic acid Chemical compound OC(=O)CCC(=O)OOC(=O)CCC(O)=O MKTOIPPVFPJEQO-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- HLLSOEKIMZEGFV-UHFFFAOYSA-N 4-(dibutylsulfamoyl)benzoic acid Chemical compound CCCCN(CCCC)S(=O)(=O)C1=CC=C(C(O)=O)C=C1 HLLSOEKIMZEGFV-UHFFFAOYSA-N 0.000 description 1
- BGNGWHSBYQYVRX-UHFFFAOYSA-N 4-(dimethylamino)benzaldehyde Chemical compound CN(C)C1=CC=C(C=O)C=C1 BGNGWHSBYQYVRX-UHFFFAOYSA-N 0.000 description 1
- LBSXSAXOLABXMF-UHFFFAOYSA-N 4-Vinylaniline Chemical compound NC1=CC=C(C=C)C=C1 LBSXSAXOLABXMF-UHFFFAOYSA-N 0.000 description 1
- KTZOPXAHXBBDBX-FCXRPNKRSA-N 4-[(e)-but-2-enoyl]oxybutyl (e)-but-2-enoate Chemical compound C\C=C\C(=O)OCCCCOC(=O)\C=C\C KTZOPXAHXBBDBX-FCXRPNKRSA-N 0.000 description 1
- GEYAGBVEAJGCFB-UHFFFAOYSA-N 4-[2-(3,4-dicarboxyphenyl)propan-2-yl]phthalic acid Chemical compound C=1C=C(C(O)=O)C(C(O)=O)=CC=1C(C)(C)C1=CC=C(C(O)=O)C(C(O)=O)=C1 GEYAGBVEAJGCFB-UHFFFAOYSA-N 0.000 description 1
- HMMIRWHBSMBXTH-UHFFFAOYSA-N 4-[bis(2-hydroxyethyl)amino]-4-oxobutanoic acid Chemical compound OCCN(CCO)C(=O)CCC(O)=O HMMIRWHBSMBXTH-UHFFFAOYSA-N 0.000 description 1
- DBCAQXHNJOFNGC-UHFFFAOYSA-N 4-bromo-1,1,1-trifluorobutane Chemical compound FC(F)(F)CCCBr DBCAQXHNJOFNGC-UHFFFAOYSA-N 0.000 description 1
- KFDVPJUYSDEJTH-UHFFFAOYSA-N 4-ethenylpyridine Chemical compound C=CC1=CC=NC=C1 KFDVPJUYSDEJTH-UHFFFAOYSA-N 0.000 description 1
- DNFUJUFGVPNZMP-UHFFFAOYSA-N 4-hydroxy-2-(2-hydroxyethyl)-2-methylbutanoic acid Chemical compound OCCC(C)(CCO)C(O)=O DNFUJUFGVPNZMP-UHFFFAOYSA-N 0.000 description 1
- JHWGFJBTMHEZME-UHFFFAOYSA-N 4-prop-2-enoyloxybutyl prop-2-enoate Chemical compound C=CC(=O)OCCCCOC(=O)C=C JHWGFJBTMHEZME-UHFFFAOYSA-N 0.000 description 1
- CDSULTPOCMWJCM-UHFFFAOYSA-N 4h-chromene-2,3-dione Chemical compound C1=CC=C2OC(=O)C(=O)CC2=C1 CDSULTPOCMWJCM-UHFFFAOYSA-N 0.000 description 1
- FVCSARBUZVPSQF-UHFFFAOYSA-N 5-(2,4-dioxooxolan-3-yl)-7-methyl-3a,4,5,7a-tetrahydro-2-benzofuran-1,3-dione Chemical compound C1C(C(OC2=O)=O)C2C(C)=CC1C1C(=O)COC1=O FVCSARBUZVPSQF-UHFFFAOYSA-N 0.000 description 1
- ZWAPMFBHEQZLGK-UHFFFAOYSA-N 5-(dimethylamino)-2-methylidenepentanamide Chemical compound CN(C)CCCC(=C)C(N)=O ZWAPMFBHEQZLGK-UHFFFAOYSA-N 0.000 description 1
- QQGYZOYWNCKGEK-UHFFFAOYSA-N 5-[(1,3-dioxo-2-benzofuran-5-yl)oxy]-2-benzofuran-1,3-dione Chemical compound C1=C2C(=O)OC(=O)C2=CC(OC=2C=C3C(=O)OC(C3=CC=2)=O)=C1 QQGYZOYWNCKGEK-UHFFFAOYSA-N 0.000 description 1
- LIUKKCUBVYIQBU-MSUUIHNZSA-N 5-[(z)-2-bromo-1-fluoro-2-(3,4,5-trimethoxyphenyl)ethenyl]-2-methoxyphenol Chemical compound C1=C(O)C(OC)=CC=C1C(\F)=C(\Br)C1=CC(OC)=C(OC)C(OC)=C1 LIUKKCUBVYIQBU-MSUUIHNZSA-N 0.000 description 1
- QBEAJGUCDQJJOV-UHFFFAOYSA-N 5-amino-3-methyl-3-phenyl-4-(1,3,5-triazin-2-ylamino)-4h-chromen-2-one Chemical compound C12=C(N)C=CC=C2OC(=O)C(C)(C=2C=CC=CC=2)C1NC1=NC=NC=N1 QBEAJGUCDQJJOV-UHFFFAOYSA-N 0.000 description 1
- IDIMQYQWJLCKLC-UHFFFAOYSA-N 5-hydroxy-2-(3-hydroxypropyl)-2-methylpentanoic acid Chemical compound OCCCC(C)(CCCO)C(O)=O IDIMQYQWJLCKLC-UHFFFAOYSA-N 0.000 description 1
- CGLVZFOCZLHKOH-UHFFFAOYSA-N 8,18-dichloro-5,15-diethyl-5,15-dihydrodiindolo(3,2-b:3',2'-m)triphenodioxazine Chemical compound CCN1C2=CC=CC=C2C2=C1C=C1OC3=C(Cl)C4=NC(C=C5C6=CC=CC=C6N(C5=C5)CC)=C5OC4=C(Cl)C3=NC1=C2 CGLVZFOCZLHKOH-UHFFFAOYSA-N 0.000 description 1
- LPEKGGXMPWTOCB-UHFFFAOYSA-N 8beta-(2,3-epoxy-2-methylbutyryloxy)-14-acetoxytithifolin Natural products COC(=O)C(C)O LPEKGGXMPWTOCB-UHFFFAOYSA-N 0.000 description 1
- RZVHIXYEVGDQDX-UHFFFAOYSA-N 9,10-anthraquinone Chemical group C1=CC=C2C(=O)C3=CC=CC=C3C(=O)C2=C1 RZVHIXYEVGDQDX-UHFFFAOYSA-N 0.000 description 1
- YDTZWEXADJYOBJ-UHFFFAOYSA-N 9-(7-acridin-9-ylheptyl)acridine Chemical compound C1=CC=C2C(CCCCCCCC=3C4=CC=CC=C4N=C4C=CC=CC4=3)=C(C=CC=C3)C3=NC2=C1 YDTZWEXADJYOBJ-UHFFFAOYSA-N 0.000 description 1
- MTRFEWTWIPAXLG-UHFFFAOYSA-N 9-phenylacridine Chemical compound C1=CC=CC=C1C1=C(C=CC=C2)C2=NC2=CC=CC=C12 MTRFEWTWIPAXLG-UHFFFAOYSA-N 0.000 description 1
- GJCOSYZMQJWQCA-UHFFFAOYSA-N 9H-xanthene Chemical compound C1=CC=C2CC3=CC=CC=C3OC2=C1 GJCOSYZMQJWQCA-UHFFFAOYSA-N 0.000 description 1
- RZVAJINKPMORJF-UHFFFAOYSA-N Acetaminophen Chemical compound CC(=O)NC1=CC=C(O)C=C1 RZVAJINKPMORJF-UHFFFAOYSA-N 0.000 description 1
- 102100033806 Alpha-protein kinase 3 Human genes 0.000 description 1
- 101710082399 Alpha-protein kinase 3 Proteins 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 101001101476 Bacillus subtilis (strain 168) 50S ribosomal protein L21 Proteins 0.000 description 1
- 235000021357 Behenic acid Nutrition 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- KYNSBQPICQTCGU-UHFFFAOYSA-N Benzopyrane Chemical compound C1=CC=C2C=CCOC2=C1 KYNSBQPICQTCGU-UHFFFAOYSA-N 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- 229930185605 Bisphenol Natural products 0.000 description 1
- FIPWRIJSWJWJAI-UHFFFAOYSA-N Butyl carbitol 6-propylpiperonyl ether Chemical compound C1=C(CCC)C(COCCOCCOCCCC)=CC2=C1OCO2 FIPWRIJSWJWJAI-UHFFFAOYSA-N 0.000 description 1
- DKJOHIMWQYGXFU-UHFFFAOYSA-N C(N)(OCC1=CC(=C(C=C1CCO)CCO)COC(N)=O)=O Chemical compound C(N)(OCC1=CC(=C(C=C1CCO)CCO)COC(N)=O)=O DKJOHIMWQYGXFU-UHFFFAOYSA-N 0.000 description 1
- GZLYSDJGEXGQDO-UHFFFAOYSA-N C1=CC=CC2=NCNC=C21 Chemical compound C1=CC=CC2=NCNC=C21 GZLYSDJGEXGQDO-UHFFFAOYSA-N 0.000 description 1
- BIUPSKBOERYLIO-WZGDRKDQSA-N CCCCN(CCCC)CCCN/C(/NNc(cc1)ccc1/N=N\C(C(C)=O)C(Nc(cc1N2)ccc1NC2=O)=O)=N/C(NCCCN(CCCC)CCCC)=N Chemical compound CCCCN(CCCC)CCCN/C(/NNc(cc1)ccc1/N=N\C(C(C)=O)C(Nc(cc1N2)ccc1NC2=O)=O)=N/C(NCCCN(CCCC)CCCC)=N BIUPSKBOERYLIO-WZGDRKDQSA-N 0.000 description 1
- JBNBJGRBKKEGQI-YTCTUYNNSA-N CCN(CC)CCCS/C(/NNc(cc1)ccc1/N=N\C(C(C)=O)C(Nc(cc1)cc(N2)c1NC2=O)=O)=N/C(SCCCN(CC)CC)=N Chemical compound CCN(CC)CCCS/C(/NNc(cc1)ccc1/N=N\C(C(C)=O)C(Nc(cc1)cc(N2)c1NC2=O)=O)=N/C(SCCCN(CC)CC)=N JBNBJGRBKKEGQI-YTCTUYNNSA-N 0.000 description 1
- 101150012716 CDK1 gene Proteins 0.000 description 1
- LAKGQRZUKPZJDH-GLIMQPGKSA-N C\C=C/C(=O)OCC(CO)(CO)COC(=O)\C=C/C Chemical compound C\C=C/C(=O)OCC(CO)(CO)COC(=O)\C=C/C LAKGQRZUKPZJDH-GLIMQPGKSA-N 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- LSPHULWDVZXLIL-UHFFFAOYSA-N Camphoric acid Natural products CC1(C)C(C(O)=O)CCC1(C)C(O)=O LSPHULWDVZXLIL-UHFFFAOYSA-N 0.000 description 1
- 239000005635 Caprylic acid (CAS 124-07-2) Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- WBYWAXJHAXSJNI-SREVYHEPSA-N Cinnamic acid Chemical compound OC(=O)\C=C/C1=CC=CC=C1 WBYWAXJHAXSJNI-SREVYHEPSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- MQIUGAXCHLFZKX-UHFFFAOYSA-N Di-n-octyl phthalate Natural products CCCCCCCCOC(=O)C1=CC=CC=C1C(=O)OCCCCCCCC MQIUGAXCHLFZKX-UHFFFAOYSA-N 0.000 description 1
- PYGXAGIECVVIOZ-UHFFFAOYSA-N Dibutyl decanedioate Chemical compound CCCCOC(=O)CCCCCCCCC(=O)OCCCC PYGXAGIECVVIOZ-UHFFFAOYSA-N 0.000 description 1
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 1
- VKOUCJUTMGHNOR-UHFFFAOYSA-N Diphenolic acid Chemical compound C=1C=C(O)C=CC=1C(CCC(O)=O)(C)C1=CC=C(O)C=C1 VKOUCJUTMGHNOR-UHFFFAOYSA-N 0.000 description 1
- ORAWFNKFUWGRJG-UHFFFAOYSA-N Docosanamide Chemical compound CCCCCCCCCCCCCCCCCCCCCC(N)=O ORAWFNKFUWGRJG-UHFFFAOYSA-N 0.000 description 1
- FPVVYTCTZKCSOJ-UHFFFAOYSA-N Ethylene glycol distearate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCCOC(=O)CCCCCCCCCCCCCCCCC FPVVYTCTZKCSOJ-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 239000005057 Hexamethylene diisocyanate Substances 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- NQSMEZJWJJVYOI-UHFFFAOYSA-N Methyl 2-benzoylbenzoate Chemical compound COC(=O)C1=CC=CC=C1C(=O)C1=CC=CC=C1 NQSMEZJWJJVYOI-UHFFFAOYSA-N 0.000 description 1
- 101100400378 Mus musculus Marveld2 gene Proteins 0.000 description 1
- OKIZCWYLBDKLSU-UHFFFAOYSA-M N,N,N-Trimethylmethanaminium chloride Chemical compound [Cl-].C[N+](C)(C)C OKIZCWYLBDKLSU-UHFFFAOYSA-M 0.000 description 1
- FSVCELGFZIQNCK-UHFFFAOYSA-N N,N-bis(2-hydroxyethyl)glycine Chemical compound OCCN(CCO)CC(O)=O FSVCELGFZIQNCK-UHFFFAOYSA-N 0.000 description 1
- UEEJHVSXFDXPFK-UHFFFAOYSA-N N-dimethylaminoethanol Chemical compound CN(C)CCO UEEJHVSXFDXPFK-UHFFFAOYSA-N 0.000 description 1
- 125000000815 N-oxide group Chemical group 0.000 description 1
- NPKSPKHJBVJUKB-UHFFFAOYSA-N N-phenylglycine Chemical compound OC(=O)CNC1=CC=CC=C1 NPKSPKHJBVJUKB-UHFFFAOYSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- HJRBEHGGICYUDN-UHFFFAOYSA-N O=S.[B+3] Chemical class O=S.[B+3] HJRBEHGGICYUDN-UHFFFAOYSA-N 0.000 description 1
- YDMUKYUKJKCOEE-SPIKMXEPSA-N OC(=O)\C=C/C(O)=O.OC(=O)\C=C/C(O)=O.OCC(CO)(CO)CO Chemical compound OC(=O)\C=C/C(O)=O.OC(=O)\C=C/C(O)=O.OCC(CO)(CO)CO YDMUKYUKJKCOEE-SPIKMXEPSA-N 0.000 description 1
- BEAWHIRRACSRDJ-UHFFFAOYSA-N OCC(CO)(CO)CO.OC(=O)CC(=C)C(O)=O.OC(=O)CC(=C)C(O)=O Chemical compound OCC(CO)(CO)CO.OC(=O)CC(=C)C(O)=O.OC(=O)CC(=C)C(O)=O BEAWHIRRACSRDJ-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010034972 Photosensitivity reaction Diseases 0.000 description 1
- LGRFSURHDFAFJT-UHFFFAOYSA-N Phthalic anhydride Natural products C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 description 1
- RVGRUAULSDPKGF-UHFFFAOYSA-N Poloxamer Chemical compound C1CO1.CC1CO1 RVGRUAULSDPKGF-UHFFFAOYSA-N 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 239000005062 Polybutadiene Substances 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- MZZSDCJQCLYLLL-UHFFFAOYSA-N Secalonsaeure A Natural products COC(=O)C12OC3C(CC1=C(O)CC(C)C2O)C(=CC=C3c4ccc(O)c5C(=O)C6=C(O)CC(C)C(O)C6(Oc45)C(=O)OC)O MZZSDCJQCLYLLL-UHFFFAOYSA-N 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- 239000006087 Silane Coupling Agent Substances 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- YSMRWXYRXBRSND-UHFFFAOYSA-N TOTP Chemical compound CC1=CC=CC=C1OP(=O)(OC=1C(=CC=CC=1)C)OC1=CC=CC=C1C YSMRWXYRXBRSND-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- OKKRPWIIYQTPQF-UHFFFAOYSA-N Trimethylolpropane trimethacrylate Chemical compound CC(=C)C(=O)OCC(CC)(COC(=O)C(C)=C)COC(=O)C(C)=C OKKRPWIIYQTPQF-UHFFFAOYSA-N 0.000 description 1
- SLGBZMMZGDRARJ-UHFFFAOYSA-N Triphenylene Natural products C1=CC=C2C3=CC=CC=C3C3=CC=CC=C3C2=C1 SLGBZMMZGDRARJ-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- FLRQOWAOMJMSTP-JJTRIOAGSA-N [(2s)-2-[(2r)-3,4-dihydroxy-5-oxo-2h-furan-2-yl]-2-hydroxyethyl] (6z,9z,12z)-octadeca-6,9,12-trienoate Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCC(=O)OC[C@H](O)[C@H]1OC(=O)C(O)=C1O FLRQOWAOMJMSTP-JJTRIOAGSA-N 0.000 description 1
- ORLQHILJRHBSAY-UHFFFAOYSA-N [1-(hydroxymethyl)cyclohexyl]methanol Chemical compound OCC1(CO)CCCCC1 ORLQHILJRHBSAY-UHFFFAOYSA-N 0.000 description 1
- GQPVFBDWIUVLHG-UHFFFAOYSA-N [2,2-bis(hydroxymethyl)-3-(2-methylprop-2-enoyloxy)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(CO)(CO)COC(=O)C(C)=C GQPVFBDWIUVLHG-UHFFFAOYSA-N 0.000 description 1
- ULQMPOIOSDXIGC-UHFFFAOYSA-N [2,2-dimethyl-3-(2-methylprop-2-enoyloxy)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(C)(C)COC(=O)C(C)=C ULQMPOIOSDXIGC-UHFFFAOYSA-N 0.000 description 1
- JUDXBRVLWDGRBC-UHFFFAOYSA-N [2-(hydroxymethyl)-3-(2-methylprop-2-enoyloxy)-2-(2-methylprop-2-enoyloxymethyl)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(CO)(COC(=O)C(C)=C)COC(=O)C(C)=C JUDXBRVLWDGRBC-UHFFFAOYSA-N 0.000 description 1
- LAKGQRZUKPZJDH-GGWOSOGESA-N [2-[[(e)-but-2-enoyl]oxymethyl]-3-hydroxy-2-(hydroxymethyl)propyl] (e)-but-2-enoate Chemical compound C\C=C\C(=O)OCC(CO)(CO)COC(=O)\C=C\C LAKGQRZUKPZJDH-GGWOSOGESA-N 0.000 description 1
- VZTQQYMRXDUHDO-UHFFFAOYSA-N [2-hydroxy-3-[4-[2-[4-(2-hydroxy-3-prop-2-enoyloxypropoxy)phenyl]propan-2-yl]phenoxy]propyl] prop-2-enoate Chemical compound C=1C=C(OCC(O)COC(=O)C=C)C=CC=1C(C)(C)C1=CC=C(OCC(O)COC(=O)C=C)C=C1 VZTQQYMRXDUHDO-UHFFFAOYSA-N 0.000 description 1
- SWHLOXLFJPTYTL-UHFFFAOYSA-N [2-methyl-3-(2-methylprop-2-enoyloxy)-2-(2-methylprop-2-enoyloxymethyl)propyl] 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(C)(COC(=O)C(C)=C)COC(=O)C(C)=C SWHLOXLFJPTYTL-UHFFFAOYSA-N 0.000 description 1
- BWVAOONFBYYRHY-UHFFFAOYSA-N [4-(hydroxymethyl)phenyl]methanol Chemical compound OCC1=CC=C(CO)C=C1 BWVAOONFBYYRHY-UHFFFAOYSA-N 0.000 description 1
- XQAXGZLFSSPBMK-UHFFFAOYSA-M [7-(dimethylamino)phenothiazin-3-ylidene]-dimethylazanium;chloride;trihydrate Chemical compound O.O.O.[Cl-].C1=CC(=[N+](C)C)C=C2SC3=CC(N(C)C)=CC=C3N=C21 XQAXGZLFSSPBMK-UHFFFAOYSA-M 0.000 description 1
- RJRZPEGSCQEPNL-UHFFFAOYSA-N [B+3].P Chemical class [B+3].P RJRZPEGSCQEPNL-UHFFFAOYSA-N 0.000 description 1
- IRXUPISPXFFGEO-UHFFFAOYSA-N [B+3].S Chemical class [B+3].S IRXUPISPXFFGEO-UHFFFAOYSA-N 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- YRKCREAYFQTBPV-UHFFFAOYSA-N acetylacetone Natural products CC(=O)CC(C)=O YRKCREAYFQTBPV-UHFFFAOYSA-N 0.000 description 1
- 229940091181 aconitic acid Drugs 0.000 description 1
- 229940023020 acriflavine Drugs 0.000 description 1
- 229920006243 acrylic copolymer Polymers 0.000 description 1
- 238000012644 addition polymerization Methods 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 150000004703 alkoxides Chemical class 0.000 description 1
- 125000005370 alkoxysilyl group Chemical group 0.000 description 1
- 150000001346 alkyl aryl ethers Chemical class 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-N ammonia Natural products N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O ammonium group Chemical group [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- JFCQEDHGNNZCLN-UHFFFAOYSA-N anhydrous glutaric acid Natural products OC(=O)CCCC(O)=O JFCQEDHGNNZCLN-UHFFFAOYSA-N 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 239000003146 anticoagulant agent Substances 0.000 description 1
- 229940053200 antiepileptics fatty acid derivative Drugs 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- 229940111121 antirheumatic drug quinolines Drugs 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- 239000004760 aramid Substances 0.000 description 1
- 229920003235 aromatic polyamide Polymers 0.000 description 1
- VUEDNLCYHKSELL-UHFFFAOYSA-N arsonium Chemical class [AsH4+] VUEDNLCYHKSELL-UHFFFAOYSA-N 0.000 description 1
- 150000008378 aryl ethers Chemical class 0.000 description 1
- 125000005110 aryl thio group Chemical group 0.000 description 1
- 150000007656 barbituric acids Chemical class 0.000 description 1
- WDIHJSXYQDMJHN-UHFFFAOYSA-L barium chloride Chemical compound [Cl-].[Cl-].[Ba+2] WDIHJSXYQDMJHN-UHFFFAOYSA-L 0.000 description 1
- 229910001626 barium chloride Inorganic materials 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 229940116226 behenic acid Drugs 0.000 description 1
- JGFLAAWSLCPCDY-UHFFFAOYSA-N benzene;cyclopenta-1,3-diene;iron Chemical class [Fe].C1C=CC=C1.C1=CC=CC=C1 JGFLAAWSLCPCDY-UHFFFAOYSA-N 0.000 description 1
- MYONAGGJKCJOBT-UHFFFAOYSA-N benzimidazol-2-one Chemical compound C1=CC=CC2=NC(=O)N=C21 MYONAGGJKCJOBT-UHFFFAOYSA-N 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 239000001518 benzyl (E)-3-phenylprop-2-enoate Substances 0.000 description 1
- NGHOLYJTSCBCGC-QXMHVHEDSA-N benzyl cinnamate Chemical compound C=1C=CC=CC=1\C=C/C(=O)OCC1=CC=CC=C1 NGHOLYJTSCBCGC-QXMHVHEDSA-N 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000000649 benzylidene group Chemical group [H]C(=[*])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- BJQHLKABXJIVAM-UHFFFAOYSA-N bis(2-ethylhexyl) phthalate Chemical compound CCCCC(CC)COC(=O)C1=CC=CC=C1C(=O)OCC(CC)CCCC BJQHLKABXJIVAM-UHFFFAOYSA-N 0.000 description 1
- ULCGAWLDXLEIIR-UHFFFAOYSA-N bis(2-hydroxyethyl) benzene-1,3-dicarboxylate Chemical compound OCCOC(=O)C1=CC=CC(C(=O)OCCO)=C1 ULCGAWLDXLEIIR-UHFFFAOYSA-N 0.000 description 1
- HSUIVCLOAAJSRE-UHFFFAOYSA-N bis(2-methoxyethyl) benzene-1,2-dicarboxylate Chemical compound COCCOC(=O)C1=CC=CC=C1C(=O)OCCOC HSUIVCLOAAJSRE-UHFFFAOYSA-N 0.000 description 1
- ZFMQKOWCDKKBIF-UHFFFAOYSA-N bis(3,5-difluorophenyl)phosphane Chemical compound FC1=CC(F)=CC(PC=2C=C(F)C=C(F)C=2)=C1 ZFMQKOWCDKKBIF-UHFFFAOYSA-N 0.000 description 1
- MQDJYUACMFCOFT-UHFFFAOYSA-N bis[2-(1-hydroxycyclohexyl)phenyl]methanone Chemical compound C=1C=CC=C(C(=O)C=2C(=CC=CC=2)C2(O)CCCCC2)C=1C1(O)CCCCC1 MQDJYUACMFCOFT-UHFFFAOYSA-N 0.000 description 1
- FAMJVEVTWNPFSF-UHFFFAOYSA-N bis[2-(2-hydroxypropan-2-yl)-4-propan-2-ylphenyl]methanone Chemical compound CC(O)(C)C1=CC(C(C)C)=CC=C1C(=O)C1=CC=C(C(C)C)C=C1C(C)(C)O FAMJVEVTWNPFSF-UHFFFAOYSA-N 0.000 description 1
- LZZMTLWFWQRJIS-UHFFFAOYSA-N bis[2-(4-propan-2-ylphenyl)propan-2-yl] 4-[3,4-bis[2-(4-propan-2-ylphenyl)propan-2-ylperoxycarbonyl]benzoyl]benzene-1,2-dicarboperoxoate Chemical compound C1=CC(C(C)C)=CC=C1C(C)(C)OOC(=O)C1=CC=C(C(=O)C=2C=C(C(C(=O)OOC(C)(C)C=3C=CC(=CC=3)C(C)C)=CC=2)C(=O)OOC(C)(C)C=2C=CC(=CC=2)C(C)C)C=C1C(=O)OOC(C)(C)C1=CC=C(C(C)C)C=C1 LZZMTLWFWQRJIS-UHFFFAOYSA-N 0.000 description 1
- 125000006367 bivalent amino carbonyl group Chemical group [H]N([*:1])C([*:2])=O 0.000 description 1
- 229910021538 borax Inorganic materials 0.000 description 1
- KGBXLFKZBHKPEV-UHFFFAOYSA-N boric acid Chemical class OB(O)O KGBXLFKZBHKPEV-UHFFFAOYSA-N 0.000 description 1
- 150000001638 boron Chemical class 0.000 description 1
- 229910052796 boron Inorganic materials 0.000 description 1
- 150000001642 boronic acid derivatives Chemical class 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- OCWYEMOEOGEQAN-UHFFFAOYSA-N bumetrizole Chemical compound CC(C)(C)C1=CC(C)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O OCWYEMOEOGEQAN-UHFFFAOYSA-N 0.000 description 1
- OZQCLFIWZYVKKK-UHFFFAOYSA-N butane-1,3-diol 2-methylidenebutanedioic acid Chemical compound CC(O)CCO.OC(=O)CC(=C)C(O)=O.OC(=O)CC(=C)C(O)=O OZQCLFIWZYVKKK-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- JHIWVOJDXOSYLW-UHFFFAOYSA-N butyl 2,2-difluorocyclopropane-1-carboxylate Chemical compound CCCCOC(=O)C1CC1(F)F JHIWVOJDXOSYLW-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- LSPHULWDVZXLIL-QUBYGPBYSA-N camphoric acid Chemical compound CC1(C)[C@H](C(O)=O)CC[C@]1(C)C(O)=O LSPHULWDVZXLIL-QUBYGPBYSA-N 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 239000003093 cationic surfactant Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- NEHMKBQYUWJMIP-NJFSPNSNSA-N chloro(114C)methane Chemical compound [14CH3]Cl NEHMKBQYUWJMIP-NJFSPNSNSA-N 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 229930016911 cinnamic acid Natural products 0.000 description 1
- 235000013985 cinnamic acid Nutrition 0.000 description 1
- CCRCUPLGCSFEDV-UHFFFAOYSA-N cinnamic acid methyl ester Natural products COC(=O)C=CC1=CC=CC=C1 CCRCUPLGCSFEDV-UHFFFAOYSA-N 0.000 description 1
- FEIQOMCWGDNMHM-UHFFFAOYSA-N cinnamylideneacetic acid Natural products OC(=O)C=CC=CC1=CC=CC=C1 FEIQOMCWGDNMHM-UHFFFAOYSA-N 0.000 description 1
- GTZCVFVGUGFEME-IWQZZHSRSA-N cis-aconitic acid Chemical compound OC(=O)C\C(C(O)=O)=C\C(O)=O GTZCVFVGUGFEME-IWQZZHSRSA-N 0.000 description 1
- NGHOLYJTSCBCGC-UHFFFAOYSA-N cis-cinnamic acid benzyl ester Natural products C=1C=CC=CC=1C=CC(=O)OCC1=CC=CC=C1 NGHOLYJTSCBCGC-UHFFFAOYSA-N 0.000 description 1
- HNEGQIOMVPPMNR-IHWYPQMZSA-N citraconic acid Chemical compound OC(=O)C(/C)=C\C(O)=O HNEGQIOMVPPMNR-IHWYPQMZSA-N 0.000 description 1
- 229940018557 citraconic acid Drugs 0.000 description 1
- 238000004581 coalescence Methods 0.000 description 1
- 239000011362 coarse particle Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 239000006103 coloring component Substances 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 239000007859 condensation product Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- AFYCEAFSNDLKSX-UHFFFAOYSA-N coumarin 460 Chemical compound CC1=CC(=O)OC2=CC(N(CC)CC)=CC=C21 AFYCEAFSNDLKSX-UHFFFAOYSA-N 0.000 description 1
- SPTHWAJJMLCAQF-UHFFFAOYSA-M ctk4f8481 Chemical compound [O-]O.CC(C)C1=CC=CC=C1C(C)C SPTHWAJJMLCAQF-UHFFFAOYSA-M 0.000 description 1
- 125000004956 cyclohexylene group Chemical group 0.000 description 1
- PESYEWKSBIWTAK-UHFFFAOYSA-N cyclopenta-1,3-diene;titanium(2+) Chemical class [Ti+2].C=1C=C[CH-]C=1.C=1C=C[CH-]C=1 PESYEWKSBIWTAK-UHFFFAOYSA-N 0.000 description 1
- WOSVXXBNNCUXMT-UHFFFAOYSA-N cyclopentane-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1CC(C(O)=O)C(C(O)=O)C1C(O)=O WOSVXXBNNCUXMT-UHFFFAOYSA-N 0.000 description 1
- 229960002887 deanol Drugs 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 125000005520 diaryliodonium group Chemical group 0.000 description 1
- 239000012954 diazonium Substances 0.000 description 1
- 150000001989 diazonium salts Chemical class 0.000 description 1
- KORSJDCBLAPZEQ-UHFFFAOYSA-N dicyclohexylmethane-4,4'-diisocyanate Chemical compound C1CC(N=C=O)CCC1CC1CCC(N=C=O)CC1 KORSJDCBLAPZEQ-UHFFFAOYSA-N 0.000 description 1
- PUFGCEQWYLJYNJ-UHFFFAOYSA-N didodecyl benzene-1,2-dicarboxylate Chemical compound CCCCCCCCCCCCOC(=O)C1=CC=CC=C1C(=O)OCCCCCCCCCCCC PUFGCEQWYLJYNJ-UHFFFAOYSA-N 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- 229940028356 diethylene glycol monobutyl ether Drugs 0.000 description 1
- XXJWXESWEXIICW-UHFFFAOYSA-N diethylene glycol monoethyl ether Chemical compound CCOCCOCCO XXJWXESWEXIICW-UHFFFAOYSA-N 0.000 description 1
- 229940075557 diethylene glycol monoethyl ether Drugs 0.000 description 1
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 1
- 229960001760 dimethyl sulfoxide Drugs 0.000 description 1
- 239000012972 dimethylethanolamine Substances 0.000 description 1
- OREAFAJWWJHCOT-UHFFFAOYSA-N dimethylmalonic acid Chemical compound OC(=O)C(C)(C)C(O)=O OREAFAJWWJHCOT-UHFFFAOYSA-N 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- CZZYITDELCSZES-UHFFFAOYSA-N diphenylmethane Chemical compound C=1C=CC=CC=1CC1=CC=CC=C1 CZZYITDELCSZES-UHFFFAOYSA-N 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- KGGOIDKBHYYNIC-UHFFFAOYSA-N ditert-butyl 4-[3,4-bis(tert-butylperoxycarbonyl)benzoyl]benzene-1,2-dicarboperoxoate Chemical compound C1=C(C(=O)OOC(C)(C)C)C(C(=O)OOC(C)(C)C)=CC=C1C(=O)C1=CC=C(C(=O)OOC(C)(C)C)C(C(=O)OOC(C)(C)C)=C1 KGGOIDKBHYYNIC-UHFFFAOYSA-N 0.000 description 1
- DDXLVDQZPFLQMZ-UHFFFAOYSA-M dodecyl(trimethyl)azanium;chloride Chemical compound [Cl-].CCCCCCCCCCCC[N+](C)(C)C DDXLVDQZPFLQMZ-UHFFFAOYSA-M 0.000 description 1
- ODQWQRRAPPTVAG-GZTJUZNOSA-N doxepin Chemical compound C1OC2=CC=CC=C2C(=C/CCN(C)C)/C2=CC=CC=C21 ODQWQRRAPPTVAG-GZTJUZNOSA-N 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- IINNWAYUJNWZRM-UHFFFAOYSA-L erythrosin B Chemical compound [Na+].[Na+].[O-]C(=O)C1=CC=CC=C1C1=C2C=C(I)C(=O)C(I)=C2OC2=C(I)C([O-])=C(I)C=C21 IINNWAYUJNWZRM-UHFFFAOYSA-L 0.000 description 1
- 229940011411 erythrosine Drugs 0.000 description 1
- 235000012732 erythrosine Nutrition 0.000 description 1
- 239000004174 erythrosine Substances 0.000 description 1
- DAOJMFXILKTYRL-UHFFFAOYSA-N ethane-1,2-diol;2-methylidenebutanedioic acid Chemical compound OCCO.OC(=O)CC(=C)C(O)=O.OC(=O)CC(=C)C(O)=O DAOJMFXILKTYRL-UHFFFAOYSA-N 0.000 description 1
- AFSIMBWBBOJPJG-UHFFFAOYSA-N ethenyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC=C AFSIMBWBBOJPJG-UHFFFAOYSA-N 0.000 description 1
- NKSJNEHGWDZZQF-UHFFFAOYSA-N ethenyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)C=C NKSJNEHGWDZZQF-UHFFFAOYSA-N 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- XPKFLEVLLPKCIW-UHFFFAOYSA-N ethyl 4-(diethylamino)benzoate Chemical compound CCOC(=O)C1=CC=C(N(CC)CC)C=C1 XPKFLEVLLPKCIW-UHFFFAOYSA-N 0.000 description 1
- 229940093499 ethyl acetate Drugs 0.000 description 1
- UHESRSKEBRADOO-UHFFFAOYSA-N ethyl carbamate;prop-2-enoic acid Chemical class OC(=O)C=C.CCOC(N)=O UHESRSKEBRADOO-UHFFFAOYSA-N 0.000 description 1
- OYQYHJRSHHYEIG-UHFFFAOYSA-N ethyl carbamate;urea Chemical compound NC(N)=O.CCOC(N)=O OYQYHJRSHHYEIG-UHFFFAOYSA-N 0.000 description 1
- STVZJERGLQHEKB-UHFFFAOYSA-N ethylene glycol dimethacrylate Substances CC(=C)C(=O)OCCOC(=O)C(C)=C STVZJERGLQHEKB-UHFFFAOYSA-N 0.000 description 1
- UKFXDFUAPNAMPJ-UHFFFAOYSA-N ethylmalonic acid Chemical compound CCC(C(O)=O)C(O)=O UKFXDFUAPNAMPJ-UHFFFAOYSA-N 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthrene Natural products C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 1
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 1
- 125000003709 fluoroalkyl group Chemical group 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 238000004817 gas chromatography Methods 0.000 description 1
- 238000010574 gas phase reaction Methods 0.000 description 1
- 229940100608 glycol distearate Drugs 0.000 description 1
- 229920000578 graft copolymer Polymers 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 150000008282 halocarbons Chemical class 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- MNWFXJYAOYHMED-UHFFFAOYSA-M heptanoate Chemical compound CCCCCCC([O-])=O MNWFXJYAOYHMED-UHFFFAOYSA-M 0.000 description 1
- 150000002391 heterocyclic compounds Chemical class 0.000 description 1
- IIRDTKBZINWQAW-UHFFFAOYSA-N hexaethylene glycol Chemical compound OCCOCCOCCOCCOCCOCCO IIRDTKBZINWQAW-UHFFFAOYSA-N 0.000 description 1
- XXMIOPMDWAUFGU-UHFFFAOYSA-N hexane-1,6-diol Chemical compound OCCCCCCO XXMIOPMDWAUFGU-UHFFFAOYSA-N 0.000 description 1
- 238000001093 holography Methods 0.000 description 1
- 239000000852 hydrogen donor Substances 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 230000003301 hydrolyzing effect Effects 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- NBZBKCUXIYYUSX-UHFFFAOYSA-N iminodiacetic acid Chemical compound OC(=O)CNCC(O)=O NBZBKCUXIYYUSX-UHFFFAOYSA-N 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 239000011256 inorganic filler Substances 0.000 description 1
- 229910003475 inorganic filler Inorganic materials 0.000 description 1
- 238000007733 ion plating Methods 0.000 description 1
- 238000012690 ionic polymerization Methods 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- PXZQEOJJUGGUIB-UHFFFAOYSA-N isoindolin-1-one Chemical compound C1=CC=C2C(=O)NCC2=C1 PXZQEOJJUGGUIB-UHFFFAOYSA-N 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- JMMWKPVZQRWMSS-UHFFFAOYSA-N isopropanol acetate Natural products CC(C)OC(C)=O JMMWKPVZQRWMSS-UHFFFAOYSA-N 0.000 description 1
- 229940011051 isopropyl acetate Drugs 0.000 description 1
- 125000005928 isopropyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- GWYFCOCPABKNJV-UHFFFAOYSA-M isovalerate Chemical compound CC(C)CC([O-])=O GWYFCOCPABKNJV-UHFFFAOYSA-M 0.000 description 1
- 229940070765 laurate Drugs 0.000 description 1
- 239000011133 lead Substances 0.000 description 1
- QDLAGTHXVHQKRE-UHFFFAOYSA-N lichenxanthone Natural products COC1=CC(O)=C2C(=O)C3=C(C)C=C(OC)C=C3OC2=C1 QDLAGTHXVHQKRE-UHFFFAOYSA-N 0.000 description 1
- 230000031700 light absorption Effects 0.000 description 1
- CDOSHBSSFJOMGT-UHFFFAOYSA-N linalool Chemical compound CC(C)=CCCC(C)(O)C=C CDOSHBSSFJOMGT-UHFFFAOYSA-N 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- DZVCFNFOPIZQKX-LTHRDKTGSA-M merocyanine Chemical compound [Na+].O=C1N(CCCC)C(=O)N(CCCC)C(=O)C1=C\C=C\C=C/1N(CCCS([O-])(=O)=O)C2=CC=CC=C2O\1 DZVCFNFOPIZQKX-LTHRDKTGSA-M 0.000 description 1
- AUHZEENZYGFFBQ-UHFFFAOYSA-N mesitylene Substances CC1=CC(C)=CC(C)=C1 AUHZEENZYGFFBQ-UHFFFAOYSA-N 0.000 description 1
- 125000001827 mesitylenyl group Chemical group [H]C1=C(C(*)=C(C([H])=C1C([H])([H])[H])C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 150000002734 metacrylic acid derivatives Chemical class 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- AYLRODJJLADBOB-QMMMGPOBSA-N methyl (2s)-2,6-diisocyanatohexanoate Chemical compound COC(=O)[C@@H](N=C=O)CCCCN=C=O AYLRODJJLADBOB-QMMMGPOBSA-N 0.000 description 1
- YDKNBNOOCSNPNS-UHFFFAOYSA-N methyl 1,3-benzoxazole-2-carboxylate Chemical compound C1=CC=C2OC(C(=O)OC)=NC2=C1 YDKNBNOOCSNPNS-UHFFFAOYSA-N 0.000 description 1
- 229940057867 methyl lactate Drugs 0.000 description 1
- WBYWAXJHAXSJNI-UHFFFAOYSA-N methyl p-hydroxycinnamate Natural products OC(=O)C=CC1=CC=CC=C1 WBYWAXJHAXSJNI-UHFFFAOYSA-N 0.000 description 1
- CCRCUPLGCSFEDV-BQYQJAHWSA-N methyl trans-cinnamate Chemical compound COC(=O)\C=C\C1=CC=CC=C1 CCRCUPLGCSFEDV-BQYQJAHWSA-N 0.000 description 1
- XJRBAMWJDBPFIM-UHFFFAOYSA-N methyl vinyl ether Chemical compound COC=C XJRBAMWJDBPFIM-UHFFFAOYSA-N 0.000 description 1
- GYNNXHKOJHMOHS-UHFFFAOYSA-N methyl-cycloheptane Natural products CC1CCCCCC1 GYNNXHKOJHMOHS-UHFFFAOYSA-N 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- ZIYVHBGGAOATLY-UHFFFAOYSA-N methylmalonic acid Chemical compound OC(=O)C(C)C(O)=O ZIYVHBGGAOATLY-UHFFFAOYSA-N 0.000 description 1
- 229960000907 methylthioninium chloride Drugs 0.000 description 1
- JESXATFQYMPTNL-UHFFFAOYSA-N mono-hydroxyphenyl-ethylene Natural products OC1=CC=CC=C1C=C JESXATFQYMPTNL-UHFFFAOYSA-N 0.000 description 1
- ZIUHHBKFKCYYJD-UHFFFAOYSA-N n,n'-methylenebisacrylamide Chemical compound C=CC(=O)NCNC(=O)C=C ZIUHHBKFKCYYJD-UHFFFAOYSA-N 0.000 description 1
- BZFWSDQZPYVFHP-UHFFFAOYSA-N n,n-dimethyl-4-methylsulfanylaniline Chemical compound CSC1=CC=C(N(C)C)C=C1 BZFWSDQZPYVFHP-UHFFFAOYSA-N 0.000 description 1
- 229940088644 n,n-dimethylacrylamide Drugs 0.000 description 1
- YLGYACDQVQQZSW-UHFFFAOYSA-N n,n-dimethylprop-2-enamide Chemical compound CN(C)C(=O)C=C YLGYACDQVQQZSW-UHFFFAOYSA-N 0.000 description 1
- MVBJSQCJPSRKSW-UHFFFAOYSA-N n-[1,3-dihydroxy-2-(hydroxymethyl)propan-2-yl]prop-2-enamide Chemical compound OCC(CO)(CO)NC(=O)C=C MVBJSQCJPSRKSW-UHFFFAOYSA-N 0.000 description 1
- WDQKICIMIPUDBL-UHFFFAOYSA-N n-[2-(dimethylamino)ethyl]prop-2-enamide Chemical compound CN(C)CCNC(=O)C=C WDQKICIMIPUDBL-UHFFFAOYSA-N 0.000 description 1
- YQCFXPARMSSRRK-UHFFFAOYSA-N n-[6-(prop-2-enoylamino)hexyl]prop-2-enamide Chemical compound C=CC(=O)NCCCCCCNC(=O)C=C YQCFXPARMSSRRK-UHFFFAOYSA-N 0.000 description 1
- DAHPIMYBWVSMKQ-UHFFFAOYSA-N n-hydroxy-n-phenylnitrous amide Chemical compound O=NN(O)C1=CC=CC=C1 DAHPIMYBWVSMKQ-UHFFFAOYSA-N 0.000 description 1
- RIWRFSMVIUAEBX-UHFFFAOYSA-N n-methyl-1-phenylmethanamine Chemical compound CNCC1=CC=CC=C1 RIWRFSMVIUAEBX-UHFFFAOYSA-N 0.000 description 1
- KKFHAJHLJHVUDM-UHFFFAOYSA-N n-vinylcarbazole Chemical compound C1=CC=C2N(C=C)C3=CC=CC=C3C2=C1 KKFHAJHLJHVUDM-UHFFFAOYSA-N 0.000 description 1
- DOBFTMLCEYUAQC-UHFFFAOYSA-N naphthalene-2,3,6,7-tetracarboxylic acid Chemical compound OC(=O)C1=C(C(O)=O)C=C2C=C(C(O)=O)C(C(=O)O)=CC2=C1 DOBFTMLCEYUAQC-UHFFFAOYSA-N 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- YTVNOVQHSGMMOV-UHFFFAOYSA-N naphthalenetetracarboxylic dianhydride Chemical compound C1=CC(C(=O)OC2=O)=C3C2=CC=C2C(=O)OC(=O)C1=C32 YTVNOVQHSGMMOV-UHFFFAOYSA-N 0.000 description 1
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- ZCYXXKJEDCHMGH-UHFFFAOYSA-N nonane Chemical compound CCCC[CH]CCCC ZCYXXKJEDCHMGH-UHFFFAOYSA-N 0.000 description 1
- 229910052755 nonmetal Inorganic materials 0.000 description 1
- BKIMMITUMNQMOS-UHFFFAOYSA-N normal nonane Natural products CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- ZWLPBLYKEWSWPD-UHFFFAOYSA-N o-toluic acid Chemical compound CC1=CC=CC=C1C(O)=O ZWLPBLYKEWSWPD-UHFFFAOYSA-N 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- GLZWNFNQMJAZGY-UHFFFAOYSA-N octaethylene glycol Chemical compound OCCOCCOCCOCCOCCOCCOCCOCCO GLZWNFNQMJAZGY-UHFFFAOYSA-N 0.000 description 1
- OTLDLKLSNZMTTA-UHFFFAOYSA-N octahydro-1h-4,7-methanoindene-1,5-diyldimethanol Chemical compound C1C2C3C(CO)CCC3C1C(CO)C2 OTLDLKLSNZMTTA-UHFFFAOYSA-N 0.000 description 1
- NOUWNNABOUGTDQ-UHFFFAOYSA-N octane Chemical compound CCCCCCC[CH2+] NOUWNNABOUGTDQ-UHFFFAOYSA-N 0.000 description 1
- 229960002446 octanoic acid Drugs 0.000 description 1
- ZPIRTVJRHUMMOI-UHFFFAOYSA-N octoxybenzene Chemical compound CCCCCCCCOC1=CC=CC=C1 ZPIRTVJRHUMMOI-UHFFFAOYSA-N 0.000 description 1
- 229920002114 octoxynol-9 Polymers 0.000 description 1
- 150000004010 onium ions Chemical class 0.000 description 1
- 125000005375 organosiloxane group Chemical group 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- IJFKEIUILIUSEV-UHFFFAOYSA-N oxane-3-carbonyl oxane-3-carboperoxoate Chemical compound C1CCOCC1C(=O)OOC(=O)C1CCCOC1 IJFKEIUILIUSEV-UHFFFAOYSA-N 0.000 description 1
- 150000002916 oxazoles Chemical group 0.000 description 1
- 125000003566 oxetanyl group Chemical group 0.000 description 1
- MPQXHAGKBWFSNV-UHFFFAOYSA-N oxidophosphanium Chemical class [PH3]=O MPQXHAGKBWFSNV-UHFFFAOYSA-N 0.000 description 1
- UFOIOXZLTXNHQH-UHFFFAOYSA-N oxolane-2,3,4,5-tetracarboxylic acid Chemical compound OC(=O)C1OC(C(O)=O)C(C(O)=O)C1C(O)=O UFOIOXZLTXNHQH-UHFFFAOYSA-N 0.000 description 1
- JCGNDDUYTRNOFT-UHFFFAOYSA-N oxolane-2,4-dione Chemical compound O=C1COC(=O)C1 JCGNDDUYTRNOFT-UHFFFAOYSA-N 0.000 description 1
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 description 1
- CKMXAIVXVKGGFM-UHFFFAOYSA-N p-cumic acid Chemical compound CC(C)C1=CC=C(C(O)=O)C=C1 CKMXAIVXVKGGFM-UHFFFAOYSA-N 0.000 description 1
- 238000002161 passivation Methods 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- JLFNLZLINWHATN-UHFFFAOYSA-N pentaethylene glycol Chemical compound OCCOCCOCCOCCOCCO JLFNLZLINWHATN-UHFFFAOYSA-N 0.000 description 1
- 125000005010 perfluoroalkyl group Chemical group 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- ZZSIDSMUTXFKNS-UHFFFAOYSA-N perylene red Chemical compound CC(C)C1=CC=CC(C(C)C)=C1N(C(=O)C=1C2=C3C4=C(OC=5C=CC=CC=5)C=1)C(=O)C2=CC(OC=1C=CC=CC=1)=C3C(C(OC=1C=CC=CC=1)=CC1=C2C(C(N(C=3C(=CC=CC=3C(C)C)C(C)C)C1=O)=O)=C1)=C2C4=C1OC1=CC=CC=C1 ZZSIDSMUTXFKNS-UHFFFAOYSA-N 0.000 description 1
- 150000002988 phenazines Chemical class 0.000 description 1
- 229950000688 phenothiazine Drugs 0.000 description 1
- 150000002990 phenothiazines Chemical class 0.000 description 1
- 239000013034 phenoxy resin Substances 0.000 description 1
- 229920006287 phenoxy resin Polymers 0.000 description 1
- BOTNYLSAWDQNEX-UHFFFAOYSA-N phenoxymethylbenzene Chemical compound C=1C=CC=CC=1COC1=CC=CC=C1 BOTNYLSAWDQNEX-UHFFFAOYSA-N 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 150000003009 phosphonic acids Chemical group 0.000 description 1
- 150000004714 phosphonium salts Chemical class 0.000 description 1
- 238000000016 photochemical curing Methods 0.000 description 1
- 230000036211 photosensitivity Effects 0.000 description 1
- 125000000612 phthaloyl group Chemical group C(C=1C(C(=O)*)=CC=CC1)(=O)* 0.000 description 1
- 229940110337 pigment blue 1 Drugs 0.000 description 1
- CLYVDMAATCIVBF-UHFFFAOYSA-N pigment red 224 Chemical compound C=12C3=CC=C(C(OC4=O)=O)C2=C4C=CC=1C1=CC=C2C(=O)OC(=O)C4=CC=C3C1=C42 CLYVDMAATCIVBF-UHFFFAOYSA-N 0.000 description 1
- 229940067265 pigment yellow 138 Drugs 0.000 description 1
- 229960005235 piperonyl butoxide Drugs 0.000 description 1
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 description 1
- 238000009832 plasma treatment Methods 0.000 description 1
- 229920001495 poly(sodium acrylate) polymer Polymers 0.000 description 1
- 239000004584 polyacrylic acid Substances 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920002857 polybutadiene Polymers 0.000 description 1
- 229920001225 polyester resin Polymers 0.000 description 1
- 239000004645 polyester resin Substances 0.000 description 1
- 229920001228 polyisocyanate Polymers 0.000 description 1
- 239000005056 polyisocyanate Substances 0.000 description 1
- 239000002952 polymeric resin Substances 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 150000004032 porphyrins Chemical class 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229960003975 potassium Drugs 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000011181 potassium carbonates Nutrition 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 229940086066 potassium hydrogencarbonate Drugs 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 230000002250 progressing effect Effects 0.000 description 1
- BAVIDSCVXNQEBA-UHFFFAOYSA-N prop-2-enoyl prop-2-eneperoxoate Chemical compound C=CC(=O)OOC(=O)C=C BAVIDSCVXNQEBA-UHFFFAOYSA-N 0.000 description 1
- KCTAWXVAICEBSD-UHFFFAOYSA-N prop-2-enoyloxy prop-2-eneperoxoate Chemical compound C=CC(=O)OOOC(=O)C=C KCTAWXVAICEBSD-UHFFFAOYSA-N 0.000 description 1
- BWJUFXUULUEGMA-UHFFFAOYSA-N propan-2-yl propan-2-yloxycarbonyloxy carbonate Chemical compound CC(C)OC(=O)OOC(=O)OC(C)C BWJUFXUULUEGMA-UHFFFAOYSA-N 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- MCSKRVKAXABJLX-UHFFFAOYSA-N pyrazolo[3,4-d]triazole Chemical class N1=NN=C2N=NC=C21 MCSKRVKAXABJLX-UHFFFAOYSA-N 0.000 description 1
- 239000005297 pyrex Substances 0.000 description 1
- 229940079877 pyrogallol Drugs 0.000 description 1
- 229940030966 pyrrole Drugs 0.000 description 1
- GZTPJDLYPMPRDF-UHFFFAOYSA-N pyrrolo[3,2-c]pyrazole Chemical compound N1=NC2=CC=NC2=C1 GZTPJDLYPMPRDF-UHFFFAOYSA-N 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 150000003248 quinolines Chemical class 0.000 description 1
- 239000007870 radical polymerization initiator Substances 0.000 description 1
- 229920005604 random copolymer Polymers 0.000 description 1
- 230000007261 regionalization Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 229940043267 rhodamine b Drugs 0.000 description 1
- 229940081623 rose bengal Drugs 0.000 description 1
- 229930187593 rose bengal Natural products 0.000 description 1
- STRXNPAVPKGJQR-UHFFFAOYSA-N rose bengal A Natural products O1C(=O)C(C(=CC=C2Cl)Cl)=C2C21C1=CC(I)=C(O)C(I)=C1OC1=C(I)C(O)=C(I)C=C21 STRXNPAVPKGJQR-UHFFFAOYSA-N 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- SPVXKVOXSXTJOY-UHFFFAOYSA-O selenonium Chemical class [SeH3+] SPVXKVOXSXTJOY-UHFFFAOYSA-O 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 125000005372 silanol group Chemical group 0.000 description 1
- 150000003376 silicon Chemical class 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 1
- NVIFVTYDZMXWGX-UHFFFAOYSA-N sodium metaborate Chemical compound [Na+].[O-]B=O NVIFVTYDZMXWGX-UHFFFAOYSA-N 0.000 description 1
- NNMHYFLPFNGQFZ-UHFFFAOYSA-M sodium polyacrylate Chemical compound [Na+].[O-]C(=O)C=C NNMHYFLPFNGQFZ-UHFFFAOYSA-M 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 235000010339 sodium tetraborate Nutrition 0.000 description 1
- 150000003413 spiro compounds Chemical class 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 125000000565 sulfonamide group Chemical group 0.000 description 1
- 150000003459 sulfonic acid esters Chemical class 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-O sulfonium group Chemical group [SH3+] RWSOTUBLDIXVET-UHFFFAOYSA-O 0.000 description 1
- 150000003464 sulfur compounds Chemical class 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 230000002123 temporal effect Effects 0.000 description 1
- BWSZXUOMATYHHI-UHFFFAOYSA-N tert-butyl octaneperoxoate Chemical compound CCCCCCCC(=O)OOC(C)(C)C BWSZXUOMATYHHI-UHFFFAOYSA-N 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- 125000001302 tertiary amino group Chemical group 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- JRMUNVKIHCOMHV-UHFFFAOYSA-M tetrabutylammonium bromide Chemical compound [Br-].CCCC[N+](CCCC)(CCCC)CCCC JRMUNVKIHCOMHV-UHFFFAOYSA-M 0.000 description 1
- 125000006158 tetracarboxylic acid group Chemical group 0.000 description 1
- YMBCJWGVCUEGHA-UHFFFAOYSA-M tetraethylammonium chloride Chemical compound [Cl-].CC[N+](CC)(CC)CC YMBCJWGVCUEGHA-UHFFFAOYSA-M 0.000 description 1
- 229940073455 tetraethylammonium hydroxide Drugs 0.000 description 1
- LRGJRHZIDJQFCL-UHFFFAOYSA-M tetraethylazanium;hydroxide Chemical compound [OH-].CC[N+](CC)(CC)CC LRGJRHZIDJQFCL-UHFFFAOYSA-M 0.000 description 1
- 150000004897 thiazines Chemical class 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 125000000101 thioether group Chemical group 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- YRHRIQCWCFGUEQ-UHFFFAOYSA-N thioxanthen-9-one Chemical class C1=CC=C2C(=O)C3=CC=CC=C3SC2=C1 YRHRIQCWCFGUEQ-UHFFFAOYSA-N 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 229950003937 tolonium Drugs 0.000 description 1
- HNONEKILPDHFOL-UHFFFAOYSA-M tolonium chloride Chemical compound [Cl-].C1=C(C)C(N)=CC2=[S+]C3=CC(N(C)C)=CC=C3N=C21 HNONEKILPDHFOL-UHFFFAOYSA-M 0.000 description 1
- RUELTTOHQODFPA-UHFFFAOYSA-N toluene 2,6-diisocyanate Chemical compound CC1=C(N=C=O)C=CC=C1N=C=O RUELTTOHQODFPA-UHFFFAOYSA-N 0.000 description 1
- 125000005424 tosyloxy group Chemical group S(=O)(=O)(C1=CC=C(C)C=C1)O* 0.000 description 1
- GTZCVFVGUGFEME-UHFFFAOYSA-N trans-aconitic acid Natural products OC(=O)CC(C(O)=O)=CC(O)=O GTZCVFVGUGFEME-UHFFFAOYSA-N 0.000 description 1
- IAQRGUVFOMOMEM-ONEGZZNKSA-N trans-but-2-ene Chemical compound C\C=C\C IAQRGUVFOMOMEM-ONEGZZNKSA-N 0.000 description 1
- OVTCUIZCVUGJHS-VQHVLOKHSA-N trans-dipyrrin Chemical class C=1C=CNC=1/C=C1\C=CC=N1 OVTCUIZCVUGJHS-VQHVLOKHSA-N 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 1
- 229960002622 triacetin Drugs 0.000 description 1
- 150000003918 triazines Chemical class 0.000 description 1
- PWBHRVGYSMBMIO-UHFFFAOYSA-M tributylstannanylium;acetate Chemical compound CCCC[Sn](CCCC)(CCCC)OC(C)=O PWBHRVGYSMBMIO-UHFFFAOYSA-M 0.000 description 1
- 125000004953 trihalomethyl group Chemical group 0.000 description 1
- 239000013638 trimer Substances 0.000 description 1
- FZGFBJMPSHGTRQ-UHFFFAOYSA-M trimethyl(2-prop-2-enoyloxyethyl)azanium;chloride Chemical compound [Cl-].C[N+](C)(C)CCOC(=O)C=C FZGFBJMPSHGTRQ-UHFFFAOYSA-M 0.000 description 1
- RRHXZLALVWBDKH-UHFFFAOYSA-M trimethyl-[2-(2-methylprop-2-enoyloxy)ethyl]azanium;chloride Chemical compound [Cl-].CC(=C)C(=O)OCC[N+](C)(C)C RRHXZLALVWBDKH-UHFFFAOYSA-M 0.000 description 1
- 150000004072 triols Chemical class 0.000 description 1
- 125000005580 triphenylene group Chemical group 0.000 description 1
- BSVBQGMMJUBVOD-UHFFFAOYSA-N trisodium borate Chemical compound [Na+].[Na+].[Na+].[O-]B([O-])[O-] BSVBQGMMJUBVOD-UHFFFAOYSA-N 0.000 description 1
- 238000001132 ultrasonic dispersion Methods 0.000 description 1
- 125000004417 unsaturated alkyl group Chemical group 0.000 description 1
- 150000003673 urethanes Chemical class 0.000 description 1
- 238000001771 vacuum deposition Methods 0.000 description 1
- 238000009489 vacuum treatment Methods 0.000 description 1
- 229940005605 valeric acid Drugs 0.000 description 1
- NLVXSWCKKBEXTG-UHFFFAOYSA-N vinylsulfonic acid Chemical compound OS(=O)(=O)C=C NLVXSWCKKBEXTG-UHFFFAOYSA-N 0.000 description 1
- 239000002351 wastewater Substances 0.000 description 1
- 238000001238 wet grinding Methods 0.000 description 1
- 150000003732 xanthenes Chemical class 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 125000006839 xylylene group Chemical group 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0034—Mixtures of two or more pigments or dyes of the same type
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/0033—Blends of pigments; Mixtured crystals; Solid solutions
- C09B67/0041—Blends of pigments; Mixtured crystals; Solid solutions mixtures containing one azo dye
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B67/00—Influencing the physical, e.g. the dyeing or printing properties of dyestuffs without chemical reactions, e.g. by treating with solvents grinding or grinding assistants, coating of pigments or dyes; Process features in the making of dyestuff preparations; Dyestuff preparations of a special physical nature, e.g. tablets, films
- C09B67/006—Preparation of organic pigments
- C09B67/0063—Preparation of organic pigments of organic pigments with only macromolecular substances
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/20—Filters
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/20—Filters
- G02B5/201—Filters in the form of arrays
-
- G—PHYSICS
- G02—OPTICS
- G02B—OPTICAL ELEMENTS, SYSTEMS OR APPARATUS
- G02B5/00—Optical elements other than lenses
- G02B5/20—Filters
- G02B5/22—Absorbing filters
- G02B5/223—Absorbing filters containing organic substances, e.g. dyes, inks or pigments
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/0005—Production of optical devices or components in so far as characterised by the lithographic processes or materials used therefor
- G03F7/0007—Filters, e.g. additive colour filters; Components for display devices
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/0045—Photosensitive materials with organic non-macromolecular light-sensitive compounds not otherwise provided for, e.g. dissolution inhibitors
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/0047—Photosensitive materials characterised by additives for obtaining a metallic or ceramic pattern, e.g. by firing
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/027—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds
- G03F7/028—Non-macromolecular photopolymerisable compounds having carbon-to-carbon double bonds, e.g. ethylenic compounds with photosensitivity-increasing substances, e.g. photoinitiators
- G03F7/031—Organic compounds not covered by group G03F7/029
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/038—Macromolecular compounds which are rendered insoluble or differentially wettable
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/09—Photosensitive materials characterised by structural details, e.g. supports, auxiliary layers
- G03F7/105—Photosensitive materials characterised by structural details, e.g. supports, auxiliary layers having substances, e.g. indicators, for forming visible images
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L27/00—Devices consisting of a plurality of semiconductor or other solid-state components formed in or on a common substrate
- H01L27/14—Devices consisting of a plurality of semiconductor or other solid-state components formed in or on a common substrate including semiconductor components sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation and specially adapted either for the conversion of the energy of such radiation into electrical energy or for the control of electrical energy by such radiation
- H01L27/144—Devices controlled by radiation
- H01L27/146—Imager structures
- H01L27/14601—Structural or functional details thereof
- H01L27/1462—Coatings
- H01L27/14621—Colour filter arrangements
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2323/00—Functional layers of liquid crystal optical display excluding electroactive liquid crystal layer characterised by chemical composition
Definitions
- the present invention has a colored curable composition suitable for producing a color filter for use in a liquid crystal display (LCD), a solid-state imaging device (CCD, CMOS, etc.) and the like, and has a colored region produced by the composition.
- the present invention relates to a color filter and a solid-state imaging device provided with the color filter.
- a color filter is an integral component of a liquid crystal display or a solid-state imaging device.
- a liquid crystal display is more compact than a CRT as a display device, and is equal to or more than in performance, and thus is being replaced by a CRT as a television screen, a personal computer screen, or another display device. Further, in recent years, the development trend of liquid crystal displays is moving from conventional monitor applications in which the screen has a relatively small area to TV applications in which the screen is large and high image quality is required.
- liquid crystal displays In color filter applications for liquid crystal displays (LCDs), the substrate size is increasing for large-screen TV production, and curing with low energy is desired to improve productivity when using a large-sized substrate. Further, liquid crystal displays for TVs are required to have higher image quality than those for conventional monitors. That is, the improvement of contrast and color purity. With respect to a curable composition used for producing a color filter to improve contrast, a finer particle size of a colorant (such as an organic pigment) to be used is required (for example, JP-A-2006-30541). No. 2). Along with this, the amount of dispersant added for pigment dispersion tends to increase.
- a colorant such as an organic pigment
- the content of the photopolymerization initiator and the photopolymerizable monomer, which are components necessary for curing the curable composition is limited in both cases for liquid crystal displays and solid imaging devices.
- the colorant concentration is high, it is difficult to make a thin layer, and it is difficult to achieve the desired sensitivity because the ratio of polymerization components is low, and there is no room for adding a development regulator, etc. It is difficult to ensure developability.
- the ratio of the pigment and the pigment dispersant in the curable composition increases with the thinning of the colored region, in particular, the colored pattern, and the content ratio of the curing component (polymerized material) relatively decreases. While it becomes difficult to maintain sensitivity, the amount of other components not related to coloring or curing, such as high acid value polymers and development accelerators added for the purpose of securing developability, is also inevitable. In addition, the problem of causing a development failure also occurs.
- the stability of the pigment dispersion becomes low and aging Cause an increase in viscosity, causing a new problem that it is difficult to form a uniform coating film.
- the colored curable composition when used to form a colored region in a color filter, particularly a color filter for a solid-state imaging device, it is necessary to satisfy both thinning and color value, and the pigment concentration is high. And, a colored curable composition capable of achieving both sensitivity and developability is highly desired.
- the present invention has been made in consideration of the above problems associated with the thinning of the colored region in the color filter, and the object of the present invention is to provide high sensitivity even when the pigment content is high.
- a colored curable composition capable of curing and having excellent developability and capable of forming a thin layer colored area with good hue and high definition, in particular, a colored curable composition useful for forming a colored area of a color filter. It is.
- a further object of the present invention is a solid-state imaging device provided with a color filter comprising a thin layer formed of the above-mentioned colored curable composition and a high-definition colored area excellent in color characteristics, and a color filter excellent in color characteristics. It is to provide.
- the present inventors have found that a pigment having a high dispersion stability can be obtained by combining a compound having a pigment mother core structure with an amino group and a resin having an acid group with a polymerizable group.
- the inventors have found that the above problems can be solved by combining a composition and an oxime ester initiator, and the present invention has been completed. That is, the constitution of the present invention is as follows.
- ⁇ 1> (A) (a-1) pigment, (a-2) a compound having a pigment mother core structure and an amino group in the molecule, and (a-3) a resin having an acid group and a polymerizable group, Colored curable composition containing the pigment dispersion liquid containing, (B) oxime ester type initiator, and (C) polymeric compound.
- ⁇ 3> The coloring according to ⁇ 1> or ⁇ 2>, wherein the compound having a pigment mother core structure and an amino group in the (a-2) molecule is a compound having a heterocyclic structure.
- Curable composition. ⁇ 4> The colored curable composition according to any one of ⁇ 1> to ⁇ 3>, which is used for forming a colored region of a color filter.
- ⁇ 5> A color filter having a colored region formed using the colored curable composition according to ⁇ 4>.
- ⁇ 6> A solid-state imaging device comprising the color filter according to ⁇ 5>.
- the action of the present invention is not clear but is estimated as follows.
- (a-1) pigment, (a-2) a compound having a pigment mother core structure and an amino group in the molecule, and (a-3) an acid group (A) A pigment dispersion comprising a resin having a polymerizable group is obtained, and the (A) pigment dispersion is mixed with (B) an oxime ester initiator and (C) a polymerizable compound to be colored.
- a curable composition A pigment dispersion comprising a resin having a polymerizable group is obtained, and the (A) pigment dispersion is mixed with (B) an oxime ester initiator and (C) a polymerizable compound to be colored.
- the pigment mother core structure in a compound having a pigment mother core structure and an amino group in the (a-2) molecule contained in the pigment dispersion (hereinafter appropriately referred to as (a-2) specific pigment derivative) is a similar mother mother.
- An interaction is formed with the (a-1) pigment having a core structure, and the (a-2) specific pigment derivative is adsorbed on the (a-1) pigment surface.
- the amino group in the (a-2) specific pigment derivative is between the coexisting (a-3) acid group and the resin having a polymerizable group (hereinafter appropriately referred to as (a-3) specific dispersion resin).
- the specific dispersion resin (a-3) is firmly bonded to the pigment surface via the (a-2) specific pigment derivative in the pigment dispersion (A) in order to form an interaction between For this reason, (a-3) the specific dispersion resin is less likely to be detached from the surface of the pigment, and the aggregation of fine pigments is suppressed for a long period of time.
- the oxime ester compound is neutral, the compatibility with the polymerizable compound in the photosensitive layer and the dispersant is good, and therefore, it is considered that the sensitivity is particularly good in this system.
- the pigment concentration in the photosensitive layer becomes high, a large amount of polymerizable compound can not be added to the composition in order to control to a desired color value and film thickness, which causes a decrease in sensitivity.
- a resin having a polymerizable group as the resin for dispersing the pigment, high sensitivity can be achieved.
- the “pigment mother core structure” is a structure containing a coloring atom group in an organic pigment, and is usually a heterocyclic structure containing a nitrogen atom, an oxygen atom, a sulfur atom or the like, an amide group, a urea structure, etc. It refers to the main skeleton structure of the pigment contained. Furthermore, the “pigment mother core structure” in the present invention has a structure similar to such a pigment main skeleton, or a partial structure of the pigment main skeleton, so as to interact with the pigment skeleton of the organic pigment. It is also intended to encompass “similar structures of pigment skeleton” and “partial structures of pigment skeleton” that can be formed.
- a colored curable composition capable of curing with high sensitivity and having excellent developability and capable of forming a thin layer colored region with good hue and high definition even when the pigment content is high.
- a colored curable composition useful for forming a colored region of a color filter.
- a solid-state imaging device provided with a color filter provided with a high-definition colored region excellent in color characteristics in a thin layer formed of the colored curable composition, and a color filter excellent in color characteristics.
- the colored curable composition of the present invention comprises (A) (a-1) a pigment, (a-2) a compound having a pigment mother core structure and an amino group in the molecule, and (a-3) an acid group and polymerization. It is characterized by containing the pigment dispersion liquid containing resin which has a property group, (B) oxime ester system initiator, and (C) polymeric compound. That is, first, (A) a pigment dispersion having excellent dispersion stability of a specific pigment is prepared, and further, (B) an oxime ester initiator and (C) a polymerizable compound are added to form a thin layer. Even if, it can form a colored region excellent in color characteristics.
- each component contained in a colored curable composition is demonstrated.
- the pigment dispersion (A) contained in the colored curable composition of the present invention is (a-1) a pigment, (a-2) a compound having a pigment mother core structure and an amino group in the molecule, (a-3) And d) a resin having an acid group and a polymerizable group, and further (a-4) a solvent.
- R 1 and R 2 each independently represent a hydrogen atom or a monovalent organic group.
- the monovalent organic group is preferably a saturated or unsaturated alkyl group having 1 to 20 carbon atoms, a saturated or unsaturated cycloalkyl group having 3 to 20 carbon atoms, or an aryl group.
- organic groups may further have a substituent, and as the substituent which can be introduced, a halogen atom, a hydroxyl group, an alkyl group, an alkenyl group, an alkynyl group, a cycloalkyl group, a cycloalkenyl group, a cycloalkynyl group , Aryl group, heterocyclic group, cyano group, alkoxy group, aryloxy group, thioalkoxy group, thioaryloxy group, carboxy group, alkoxycarbonyl group, sulfo group, sulfonamido group, urea group, thiourea group, amino group, amide And substituents having a group, a carbonyl group, a nitro group or these groups.
- R 1 and R 2 may be bonded to each other to form a ring.
- the compound represented by the above general formula (A) is preferably a compound having at least one of an amide structure and a urea structure in the molecule. Moreover, it is preferable that the compound represented by General formula (A) is a compound which has a heterocyclic structure.
- X is an m-valent group containing a pigment mother core structure.
- the pigment mother core structure include the coloring atom group in the organic pigment, the similar structure, or the partial structure as described above, and specifically, a skeleton having an azo group, a skeleton having a urea structure, an amide structure A structure having at least one selected from a skeleton having a cyclic amide structure, a skeleton having a cyclic amide structure, an aromatic ring having a hetero atom-containing 5-membered ring, and an aromatic ring having a hetero atom-containing 6-membered ring X is a substituent containing these pigment mother core structures.
- X preferably has a pigment mother nucleus structure, or a pigment mother nucleus structure and an aromatic ring, or a nitrogen-containing aromatic ring, or an oxygen-containing aromatic ring, or a sulfur-containing aromatic ring, and the amino group is a pigment mother nucleus It is bonded directly or via a linking group to any of a structure, an aromatic ring, a nitrogen-containing aromatic ring, an oxygen-containing aromatic ring and a sulfur-containing aromatic ring. Most preferably, it has a pigment mother nucleus structure and an aromatic ring, or a nitrogen-containing aromatic ring, and is preferably bonded to an amino group and a divalent linking group.
- n is an integer of 1 to 8, preferably 1 to 6 from the viewpoint of dispersibility and storage stability of the dispersion, and more preferably 2 to 6.
- Preferred specific examples of the (a-2) specific pigment derivative used in the present invention are shown below, but the invention is not limited thereto.
- the content of the (a-2) specific pigment derivative in the pigment dispersion is 0.5% by mass or more and 40% by mass or less as solid content. Is preferable, and 1% by mass or more and 15% by mass or less is more preferable.
- the amount is preferably 0.5 to 50 parts by mass and more preferably 1 to 25 parts by mass with respect to 100 parts by mass of the (a-1) pigment described later.
- a pigment which can be used for the (A) pigment dispersion liquid which concerns on this invention a conventionally well-known various inorganic pigment or organic pigment can be used.
- the pigment is preferably an inorganic pigment or an organic pigment, it is preferable to use a pigment with a small particle size and a small particle size as much as possible, in consideration of handling properties as well.
- it is preferably a pigment having an average primary particle size of 0.01 ⁇ m to 0.3 ⁇ m, more preferably 0.01 ⁇ m to 0.15 ⁇ m.
- the average primary particle size is determined by observation with SEM or TEM, measuring 100 particle sizes in a portion where the particles are not aggregated, and calculating the average value.
- metal compounds such as metal oxides and metal complex salts can be mentioned, and specifically, metals such as iron, cobalt, aluminum, cadmium, lead, copper, titanium, magnesium, chromium, zinc, antimony and the like And oxides of the above metals, and complex oxides of the above metals.
- Examples of the organic pigment include C. I. Pigment Red 1, 2, 3, 4, 5, 6, 7, 9, 10, 14, 17, 22, 23, 31, 38, 41, 48: 1, 48: 2, 48: 3, 48: 4, 49, 49: 1, 49: 2, 52: 1, 52: 2, 53: 1, 57: 1, 60: 1, 63: 1, 66, 67, 81: 1, 81: 2, 81: 3, 83, 88, 90, 105, 112, 119, 122, 123, 144, 146, 149, 150, 155, 166, 168, 169, 170, 171, 172, 175, 176, 177, 178, 179, 184, 185, 187, 188, 190, 200, 202, 206, 208, 209, 210, 210, 216, 220, 224, 242, 246, 254, 255, 264, 270, 272, 279, C.
- fine and sized organic pigments can be used as needed. Fineness of the pigment is prepared by preparing a highly viscous liquid composition together with the pigment, the water-soluble organic solvent and the water-soluble inorganic salt, and applying a stress and grinding using a wet grinding apparatus or the like. To be achieved.
- the water-soluble organic solvent used in the step of refining the pigment includes methanol, ethanol, isopropanol, n-propanol, isobutanol, n-butanol, ethylene glycol, diethylene glycol, diethylene glycol monomethyl ether, diethylene glycol monoethyl ether, diethylene glycol monobutyl ether And propylene glycol, propylene glycol monoglycol ether and the like.
- solvents which have low water solubility or do not have water solubility as long as they are adsorbed to the pigment by using a small amount and are not washed away in waste water such as benzene, toluene, xylene, ethylbenzene, chlorobenzene, Nitrobenzene, aniline, pyridine, quinoline, tetrahydrofuran, dioxane, ethyl acetate, isopropyl acetate, butyl acetate, butyl acetate, hexane, heptane, octane, nonane, decane, undecane, dodecane, cyclohexane, methylcyclohexane, halogenated hydrocarbon, acetone, methyl ethyl ketone, methyl An isobutyl ketone, cyclohexanone, dimethylformamide, dimethylsulfoxide, N-methyl
- Examples of the water-soluble inorganic salt used in the step of refining the pigment in the present invention include sodium chloride, potassium chloride, calcium chloride, barium chloride, sodium sulfate and the like.
- the amount of the water-soluble inorganic salt used in the refining step is 1 to 50 times by mass that of the pigment, and the larger the amount is, the more effective there is attrition effect, but the more preferable amount is 1 to 10 times by mass in terms of productivity.
- the amount of the water-soluble organic solvent used in the micronization step is in the range of 50 parts by mass to 300 parts by mass, preferably 100 parts by mass to 200 parts by mass with respect to 100 parts by mass of the pigment.
- the operating conditions of the wet pulverizing apparatus in the pigment refining process are the number of rotations of the blades in the apparatus in order to effectively advance the grinding by the pulverizing media. 10 to 200 rpm is preferable, and the one having a relatively large rotational ratio of two axes is preferable because the grinding effect is large.
- the operation time is preferably 1 to 8 hours in combination with the dry pulverization time, and the internal temperature of the apparatus is preferably 50 to 150.degree.
- the water-soluble inorganic salt, which is a grinding media preferably has a grinding particle size of 5 to 50 ⁇ m, a sharp particle size distribution, and a spherical shape.
- organic pigments can be used alone or in various combinations in order to increase the color purity.
- the specific example of the said combination is shown below.
- red pigments anthraquinone pigments, perylene pigments, diketopyrrolopyrrole pigments alone or at least one of them, disazo yellow pigments, isoindoline yellow pigments, quinophthalone yellow pigments or perylene red pigments And mixtures thereof with anthraquinone red pigments and diketopyrrolopyrrole red pigments.
- anthraquinone pigments C.I. I. Pigment red 177
- perylene pigments include C.I. I.
- Pigment red 155, C.I. I. Pigment red 224, and examples of diketopyrrolopyrrole pigments include C.I. I. Pigment red 254, and C.I. I. Pigment yellow 83, C.I. I. Pigment yellow 139 or C.I. I. A mixture with pigment red 177 is preferred.
- the mass ratio of the red pigment to the other pigment is preferably 100: 5 to 100: 80. In this range, the light transmittance of 400 nm to 500 nm can be suppressed, the color purity can be improved, and sufficient coloring power can be achieved. In particular, the range of 100: 10 to 100: 65 is optimum as the above mass ratio. In addition, in the case of the combination of red pigments, it can adjust according to a chromaticity.
- the green pigment it is possible to use one halogenated phthalocyanine-based pigment alone or a mixture thereof with a disazo-based yellow pigment, a quinophthalone-based yellow pigment, an azomethine-based yellow pigment or an isoindoline-based yellow pigment .
- a disazo-based yellow pigment e.g., C.I. I. Pigment green 7, 36, 37 and C.I. I. Pigment yellow 83, C.I. I. Pigment yellow 138, C.I. I. Pigment yellow 139, C.I. I. Pigment yellow 150, C.I. I. Pigment yellow 180 or C.I. I.
- the mixing with pigment yellow 185 is preferred.
- the mass ratio of the green pigment to the yellow pigment is preferably 100: 5 to 100: 200.
- the light transmittance of 400 to 450 nm can be suppressed, the color purity can be improved, and the main wavelength does not become close to the long wavelength, and a hue near the NTSC target hue as set position.
- You can get The mass ratio is particularly preferably in the range of 100: 20 to 100: 150.
- one phthalocyanine pigment may be used alone, or a mixture of this with a dioxazine purple pigment may be used.
- C.I. I. Pigment blue 15: 6 and C.I. I. A mixture with pigment violet 23 can be mentioned.
- the mass ratio of the blue pigment to the purple pigment is preferably 100: 0 to 100: 100, more preferably 100: 70 or less.
- carbon black, graphite, titanium black, iron oxide, titanium oxide can be used alone or in combination, and a combination of carbon black and titanium black is preferable.
- the mass ratio of carbon black to titanium black is preferably in the range of 100: 0 to 100: 60. If it is 100: 61 or more, dispersion stability may be reduced.
- a dye together with a pigment as a colorant in order to adjust the color tone.
- the dye that can be used as a colorant is not particularly limited, and known dyes conventionally used for color filter applications can be used.
- JP-A-8-62416 JP-A-2002-14220, JP-A-2002-14221, JP-A-2002-14222, JP-A-2002-14223, JP-A-8-302224. JP-A-8-73758, JP-A-8-179120, JP-A-8-151531, and the like.
- the chemical structure includes pyrazole azo type, anilino azo type, triphenylmethane type, anthraquinone type, anthrapyridone type, benzylidene type, oxonol type, pyrazolotriazole azo type, pyridone azo type, cyanine type, phenothiazine type, pyrrolopyrazole azomethine type, Dyes of xanthene type, phthalocyanine type, benzopyran type, indigo type, etc. can be used.
- the content of the pigment (a-1) in the pigment dispersion (A) in the present invention is preferably 10 to 60% by mass with respect to the total solid content (mass) in the pigment dispersion (A), -50% by mass is more preferable.
- the content of the pigment is in the above-mentioned range, it is effective for securing sufficient color density and securing excellent color characteristics.
- the pigment dispersion (A) in the present invention contains at least one of (a-3) a resin having an acid group and a polymerizable group [(a-3) specific dispersion resin]. By the inclusion of this resin, high dispersibility is exhibited, and sensitivity can be improved when combined with an initiator and a polymerizable compound.
- the acid group in the present application is a group having a dissociative group with a pKa of 14 or less, and specifically, for example, -COOH, -SO 3 H, -PO 3 H 2 , -OSO 3 H, -OPO 2 H 2, -PhOH, -SO 2 H, -SO 2 NH 2, -SO 2 NHCO -, - SO 2 NHSO 2 - and the like, among -COOH, -SO 3 H, is -PO 3 H 2 preferably , -COOH is most preferred.
- the (a-3) specific dispersion resin of the present invention has a double bond (ethylenically unsaturated bond) in a side chain, and the side chain double bond is, for example, a carboxyl group of an alkali-soluble resin having a carboxyl group. It can be obtained by adding an ethylenically unsaturated group-containing epoxy compound.
- a resin having a carboxyl group 1) a resin obtained by radically or ionically polymerizing a monomer having a carboxyl group, and 2) a radical or ionic polymerization of a monomer having an acid anhydride to hydrolyze or half esterify an acid anhydride unit And 3) epoxy acrylates obtained by modifying an epoxy resin with unsaturated monocarboxylic acid and acid anhydride.
- a vinyl-based resin having a carboxyl group as a monomer having a carboxyl group, (meth) acrylic acid, 2-succinoroyloxyethyl methacrylate, 2-maleinoroyloxyethyl methacrylate, methacrylic acid 2 -Resins obtained by homopolymerizing unsaturated carboxylic acids such as phthaloyl oxyethyl, 2-hexahydrophthaloyl oxyethyl methacrylate, maleic acid, fumaric acid, itaconic acid and crotonic acid, and styrenes of these unsaturated carboxylic acids , ⁇ -methylstyrene, methyl (meth) acrylate, ethyl (meth) acrylate, propyl (meth) acrylate, isopropyl (meth) acrylate, butyl (meth) acrylate, vinyl acetate, acrylonitrile,
- unsaturated carboxylic acids
- maleic anhydride is copolymerized with styrene, ⁇ -methylstyrene etc.
- maleic anhydride unit part is half-esterified with monohydric alcohol such as methanol, ethanol, propanol, butanol, hydroxyethyl (meth) acrylate or by water Also included are hydrolyzed resins.
- the resin of the present invention can also be obtained by reacting a resin having an epoxy group with a compound having a nucleophilic group and a polymerizable group.
- the resin of the present invention can also be obtained by reacting acrylic acid, methacrylic acid, hydroxyethyl methacrylate and the like with a polymer obtained by copolymerizing glycidyl methacrylate. In this case, it is also considered that the acid value is lowered, but acid anhydrides such as succinic acid, maleic anhydride, cyclohexanedicarboxylic acid anhydride, itaconic acid anhydride, etc. are considered as hydroxyl groups generated by the reaction of epoxy and carboxylic acid.
- a resin having a desired acid value can be obtained.
- novolac epoxy acrylate resin bisphenol epoxy resin, etc. (meth) acrylic acid, 2-succinoroyloxyethyl methacrylate, 2-maleinoroyloxyethyl methacrylate, 2-phthaloyloxyethyl methacrylate, methacrylic acid 2
- Unsaturated carboxylic acid such as hexahydrophthaloyl oxyethyl, maleic acid, fumaric acid, itaconic acid, crotonic acid or saturated carboxylic acid such as acetic acid, propionic acid or stearic acid, and then maleic anhydride or anhydrous
- a resin modified with an acid anhydride such as itaconic acid, tetrahydrophthalic anhydride, phthalic anhydride and the like can also be mentioned.
- resins having a carboxyl group in particular, (meth) acrylic acid (co) polymer resins containing (meth) acrylic acid are preferable, and as these copolymers, specific ones may be used.
- methyl methacrylate / methacrylic acid copolymer described in JP-A 60-208748 methyl methacrylate / methyl acrylate / methacrylic acid copolymer described in JP-A 60-214354, and the like.
- epoxy unsaturated compound although what has a glycidyl group as epoxy groups, such as glycidyl (meth) acrylate and allyl glycidyl ether, can also be used, a preferable thing is an unsaturated compound which has an alicyclic epoxy group. As such a thing, the following compounds can be illustrated, for example.
- the reaction introduced into the resin side chain is catalyzed by, for example, tertiary amines such as triethylamine and benzylmethylamine, quaternary ammonium salts such as dodecyltrimethylammonium chloride, tetramethylammonium chloride and tetraethylammonium chloride, pyridine, triphenylphosphine and the like.
- tertiary amines such as triethylamine and benzylmethylamine
- quaternary ammonium salts such as dodecyltrimethylammonium chloride, tetramethylammonium chloride and tetraethylammonium chloride
- pyridine triphenylphosphine and the like.
- triphenylphosphine triphenylphosphine and the like.
- the weight average molecular weight of the (a-3) specific dispersion resin is preferably in the range of 500 to 5,000,000, and more preferably 1,000 to 500,000.
- the (a-3) specific dispersion resin in the present invention is a polymer having at least one structural unit selected from structural units represented by any of the following general formulas (1) to (3) as an unsaturated double bond moiety. Compounds are preferred.
- each of A 1 , A 2 and A 3 independently represents an oxygen atom, a sulfur atom or -N (R 21 )-, and R 21 represents a substituent Represents an alkyl group which may be possessed.
- G 1 , G 2 and G 3 each independently represent a divalent organic group.
- X and Z each independently represent an oxygen atom, a sulfur atom, or -N (R 22 )-, and R 22 represents an alkyl group which may have a substituent.
- Y represents an oxygen atom, a sulfur atom, a phenylene group which may have a substituent, or -N (R 23 )-, and R 23 represents an alkyl group which may have a substituent.
- R 1 to R 20 each independently represent a monovalent substituent.
- each R 1 ⁇ R 3 are independently represent a monovalent substituent, a hydrogen atom, further including an alkyl group which may have a substituent, inter alia, R 1 And R 2 is preferably a hydrogen atom, and R 3 is preferably a hydrogen atom or a methyl group.
- R 4 to R 6 each independently represent a monovalent substituent, and examples of R 4 include a hydrogen atom and an alkyl group which may further have a substituent, and among them, a hydrogen atom and a methyl group And ethyl are preferred.
- R 5 and R 6 each independently further have a hydrogen atom, a halogen atom, an alkoxycarbonyl group, a sulfo group, a nitro group, a cyano group, an alkyl group which may further have a substituent, a substituent Aryl group, an alkoxy group which may further have a substituent, an aryloxy group which may further have a substituent, an alkylsulfonyl group which may further have a substituent, further having a substituent And the like.
- a hydrogen atom, an alkoxycarbonyl group, an alkyl group which may further have a substituent, and an aryl group which may further have a substituent are preferable.
- examples of the substituent which can be introduced include a methoxycarbonyl group, an ethoxycarbonyl group, an isopropyloxycarbonyl group, a methyl group, an ethyl group, a phenyl group and the like.
- a 1 represents an oxygen atom, a sulfur atom or -N (R 21 )-
- X represents an oxygen atom, a sulfur atom or -N (R 22 )-.
- R 21 and R 22 an alkyl group which may have a substituent can be mentioned.
- G 1 represents a divalent organic group, preferably an alkylene group which may have a substituent.
- an alkylene group which may have a substituent of 1 to 20 carbon atoms a cycloalkylene group which may have a substituent of 3 to 20 carbon atoms, a substituent having 6 to 20 carbon atoms
- Aromatic group and the like and among them, a linear or branched alkylene group having 1 to 10 carbon atoms which may have a substituent, and cyclo which may have a substituent having 3 to 10 carbon atoms
- An alkylene group and an aromatic group which may have a substituent having 6 to 12 carbon atoms are preferable in terms of performance such as strength and developability.
- a substituent in G 1 a hydroxyl group is preferable.
- R 7 to R 9 each independently represent a monovalent substituent, and examples thereof include a hydrogen atom and an alkyl group which may further have a substituent, and among them, R 7 R 8 is preferably a hydrogen atom, and R 9 is preferably a hydrogen atom or a methyl group.
- R 10 to R 12 each independently represent a monovalent substituent, and specific examples of the substituent include a hydrogen atom, a halogen atom, a dialkylamino group, an alkoxycarbonyl group, a sulfo group and a nitro group.
- Examples thereof include an oxy group, an alkylsulfonyl group which may further have a substituent, an arylsulfonyl group which may further have a substituent, and the like, and among them, it may further have a hydrogen atom, an alkoxycarbonyl group and a substituent.
- Preferred is an alkyl group or an aryl group which may further have a substituent.
- substituents which can be introduced those mentioned in the general formula (1) are exemplified in the same manner.
- a 2 represents an oxygen atom, a sulfur atom, or —N (R 21 ) —, wherein R 21 includes a hydrogen atom, an alkyl group which may have a substituent, and the like.
- G 2 represents a divalent organic group, preferably an alkylene group which may have a substituent. Preferably, it has an alkylene group which may have a substituent of 1 to 20 carbon atoms, a cycloalkylene group which may have a substituent of 3 to 20 carbon atoms, and a substituent having 6 to 20 carbon atoms.
- aromatic groups and the like and among them, linear or branched alkylene groups having 1 to 10 carbon atoms which may have a substituent, and cycloalkylenes which may have a substituent having 3 to 10 carbon atoms
- a group and an aromatic group which may have a substituent having 6 to 12 carbon atoms are preferable in terms of performance such as strength and developability.
- the substituent for G 2 is preferably a hydroxyl group.
- Y represents an oxygen atom, a sulfur atom, -N (R 23 )-or a phenylene group which may have a substituent.
- R 23 a hydrogen atom, an alkyl group which may have a substituent, and the like can be mentioned.
- R 13 to R 15 each independently represent a monovalent substituent, and examples thereof include a hydrogen atom and an alkyl group which may have a substituent, and among them, R 13 R 14 is preferably a hydrogen atom, and R 15 is preferably a hydrogen atom or a methyl group.
- R 16 to R 20 each independently represent a monovalent substituent
- R 16 to R 20 each represent, for example, a hydrogen atom, a halogen atom, a dialkylamino group, an alkoxycarbonyl group, a sulfo group, a nitro group or a cyano group
- a 3 represents an oxygen atom, a sulfur atom, or -N (R 21 )-
- Z represents an oxygen atom, a sulfur atom, or -N (R 22 )-.
- R 21 and R 22 the same as in General Formula (1) can be mentioned.
- G 3 represents a divalent organic group, preferably an alkylene group which may have a substituent.
- it has an alkylene group which may have a substituent of 1 to 20 carbon atoms, a cycloalkylene group which may have a substituent of 3 to 20 carbon atoms, and a substituent having 6 to 20 carbon atoms.
- a group and an aromatic group which may have a substituent having 6 to 12 carbon atoms are preferable in terms of performance such as strength and developability.
- the substituent for G 3, is preferably a hydroxyl group.
- the structural units represented by the above general formulas (1) to (3) are contained in an amount of 20 mol% in one molecule from the viewpoint of curability improvement and development It is preferable that it is a compound which contains in more than 95 mol% of range. More preferably, it is 25 to 90 mol%. More preferably, it is in the range of 30 mol% or more and less than 85 mol%.
- the synthesis of the polymer compound having a structural unit represented by the general formulas (1) to (3) is carried out according to the synthesis method described in paragraph Nos. [0027] to [0057] of JP-A No. 2003-226258. It can be done. Among these, the synthesis method 1) in the same publication is preferable, and this is shown in the following (1).
- polymer compound having a structural unit represented by the general formulas (1) to (3) As specific examples of the polymer compound having a structural unit represented by the general formulas (1) to (3), the following polymer compounds 1 to 17 can be mentioned.
- the resin obtained by the synthesis method of following (1) or (2) can be mentioned suitably.
- a base is used to extract a proton, L is eliminated, and a structure represented by the above general formula (1)
- R 3 to R 6 , A 1 , G 1 and X are as defined in the general formula (1), and either an inorganic compound or an organic compound is used as a base for causing the elimination reaction. You may use it. Further, details and preferable embodiments of this method are described in paragraph Nos. [0028] to [0033] of JP-A No. 2003-26298.
- Preferred inorganic compound bases include sodium hydroxide, potassium hydroxide, sodium carbonate, sodium hydrogencarbonate, potassium carbonate, potassium hydrogencarbonate and the like
- organic compound bases include sodium methoxide, sodium ethoxide, potassium t- Examples include metal alkoxides such as butoxide, and organic amine compounds such as triethylamine, pyridine and diisopropylethylamine.
- a 5 represents an oxygen atom, a sulfur atom, or -N (R 54) - represents
- a 6 represents an oxygen atom, a sulfur atom, or -NR 58 - represents, R 51, R 52 , R 53 , R 54 , R 55 , R 56 , R 57 and R 58 each independently represent hydrogen or a monovalent organic group
- X 10 represents a group to be removed by elimination reaction
- G 5 represents an organic linking group.
- n represents an integer of 1 to 10.
- a polymer compound described in JP-A-2003-335814 specifically, for example, (i) polyvinyl polymer compound, (ii) polyurethane polymer Compound, (iii) polyurea polymer, (iv) poly (urethane-urea) polymer, (v) polyester polymer, (vi) polyamide polymer, (vii) acetal-modified polyvinyl alcohol Polymer compounds of the system, and specific compounds obtained from the description of each of these can be suitably mentioned.
- Examples of the compound represented by the general formula (4) include the following compounds (M-1) to (M-12), but are not limited thereto.
- the specific dispersion resin (a-3) in the present invention needs to have a photopolymerizable unsaturated bond from the viewpoint of improving photosensitivity, and from the viewpoint of enabling alkali development, COOH, SO 3 H It is necessary to have an acid group such as, PO 3 H 2 , OSO 3 H, OPO 2 H 2 or the like.
- the (a-3) specific dispersion resin in the present invention has an acid value of 20 to 300 KOH ⁇ mg / g, preferably 40 to 200 KOH ⁇ mg / g, from the viewpoint of the dispersion stability and the balance between developability and sensitivity. More preferably, it is in the range of 60 to 150 KOH ⁇ mg / g.
- Examples of the monomer which can be used as a copolymerizable monomer when synthesizing the (a-3) specific dispersion resin in the present invention include the following (1) to (13).
- a monomer having a carboxy group such as acrylic acid, methacrylic acid, itaconic acid, crotonic acid, maleic acid, fumaric acid and 4-carboxystyrene, and an acidic cellulose derivative having a carboxy group in the side chain can also be used.
- Vinyl ethers such as ethyl vinyl ether, 2-chloroethyl vinyl ether, hydroxyethyl vinyl ether, propyl vinyl ether, butyl vinyl ether, octyl vinyl ether, phenyl vinyl ether and the like.
- Vinyl esters such as vinyl acetate, vinyl chloroacetate, vinyl butyrate and vinyl benzoate.
- Styrenes such as styrene, ⁇ -methylstyrene, methylstyrene, chloromethylstyrene and p-acetoxystyrene.
- Vinyl ketones such as methyl vinyl ketone, ethyl vinyl ketone, propyl vinyl ketone, phenyl vinyl ketone and the like.
- Olefins such as ethylene, propylene, isobutylene, butadiene and isoprene.
- N-vinyl pyrrolidone acrylonitrile, methacrylonitrile and the like.
- Unsaturated imides such as maleimide, N-acryloyl acrylamide, N-acetyl methacrylamide, N-propionyl methacrylamide, N- (p-chlorobenzoyl) methacrylamide and the like.
- a methacrylic acid-based monomer in which a hetero atom is bonded to the ⁇ position For example, compounds described in JP-A-2002-309057, JP-A-2002-311569, and the like.
- non-acrylic resins having a polymerizable group in the side chain can also be used.
- urethane resins include urethane resins, novolac resins, acetal resins, styrene resins, polyester resins, polyamide resins, polyurea resins, polyimide resins, etc., each having an ethylenically unsaturated bond in the side chain.
- urethane resins hereinafter, appropriately referred to as “specified urethane resin”
- styrene resins hereinafter, appropriately referred to as “specified styrene resin” are particularly preferable from the viewpoint of the effect.
- the specific urethane resin which is a preferred embodiment of the present invention, will be described in detail.
- Specific urethane resins used in the present invention include those having at least one of functional groups represented by the following general formulas (1) to (3) in the side chain. First, functional groups represented by the following general formulas (1) to (3) will be described.
- R 1 to R 3 each independently represent a hydrogen atom or a monovalent organic group, but as R 1 , preferably, a hydrogen atom or an alkyl which may have a substituent And the like, and among them, a hydrogen atom and a methyl group are preferable because of high radical reactivity.
- R 2 and R 3 each independently represent a hydrogen atom, a halogen atom, an amino group, a carboxyl group, an alkoxycarbonyl group, a sulfo group, a nitro group, a cyano group, an alkyl group which may have a substituent, a substitution An aryl group which may have a group, an alkoxy group which may have a substituent, an aryloxy group which may have a substituent, an alkylamino group which may have a substituent, and a substituent And arylamino group which may be substituted, alkylsulfonyl group which may have a substituent, arylsulfonyl group which may have a substituent, etc., among which a hydrogen atom, a carboxyl group, an alkoxycarbonyl group, a substituent Alkyl groups which may have one or more substituents and aryl groups which may have one or more substituents are preferable because of high radical
- X represents an oxygen atom, a sulfur atom, or -N (R 12 )-
- R 12 represents a hydrogen atom or a monovalent organic group.
- R 12 is, for example, an alkyl group which may have a substituent. Among them, a hydrogen atom, a methyl group, an ethyl group or an isopropyl group is preferable because of high radical reactivity.
- an alkyl group an alkenyl group, an alkynyl group, an aryl group, an alkoxy group, an aryloxy group, a halogen atom, an amino group, an alkylamino group, an arylamino group, a carboxyl group, an alkoxycarbonyl group, A sulfo group, a nitro group, a cyano group, an amido group, an alkylsulfonyl group, an arylsulfonyl group etc. are mentioned.
- R 4 to R 8 each independently represent a hydrogen atom or a monovalent organic group, but R 4 to R 8 are preferably a hydrogen atom, a halogen atom, an amino group, a dialkyl Amino group, carboxyl group, alkoxycarbonyl group, sulfo group, nitro group, cyano group, alkyl group which may have a substituent, aryl group which may have a substituent, alkoxy which may have a substituent A group, an aryloxy group which may have a substituent, an alkylamino group which may have a substituent, an arylamino group which may have a substituent, an alkylsulfonyl group which may have a substituent, The arylsulfonyl group which may have a substituent is mentioned, Especially, a hydrogen atom, a carboxyl group, an alkoxycarbonyl group, an alkyl group which may have a substituent, ary
- Y represents an oxygen atom, a sulfur atom, or -N (R 12 )-.
- R 12 has the same meaning as that of R 12 in formula (1), and preferred examples are also the same.
- R 9 preferably includes a hydrogen atom or an alkyl group which may have a substituent, and among them, a hydrogen atom and a methyl group have high radical reactivity. It is preferable from R 10 and R 11 each independently represent a hydrogen atom, a halogen atom, an amino group, a dialkylamino group, a carboxyl group, an alkoxycarbonyl group, a sulfo group, a nitro group, a cyano group, or an alkyl group which may have a substituent An aryl group which may have a substituent, an alkoxy group which may have a substituent, an aryloxy group which may have a substituent, an alkylamino group which may have a substituent, Examples thereof include an arylamino group which may have, an alkylsulfonyl group which may have a substituent, and an arylsulfonyl group which may have a substituent. Among them,
- Z represents an oxygen atom, a sulfur atom, -N (R 13 )-, or a phenylene group which may have a substituent.
- R 13 include an alkyl group which may have a substituent, and the like. Among them, a methyl group, an ethyl group and an isopropyl group are preferable because of high radical reactivity.
- the specific urethane resin according to the present invention is a reaction product of at least one kind of diisocyanate compound represented by the following general formula (4) and at least one kind of diol compound represented by the general formula (5)
- Polyurethane resin (hereinafter referred to as “specific polyurethane resin” as appropriate) having a basic structural unit as a structural unit.
- X 0 and Y 0 each independently represent a divalent organic residue.
- At least one of the diisocyanate compound represented by the general formula (4) or the diol compound represented by the general formula (5) is at least one of the groups represented by the general formulas (1) to (3) If it has one, as a reaction product of the diisocyanate compound and the diol compound, a specific polyurethane resin in which the groups represented by the general formulas (1) to (3) are introduced into the side chain is formed. Be done. According to this method, it is possible to easily produce the specific polyurethane resin according to the present invention, rather than substituting and introducing a desired side chain after reaction formation of the polyurethane resin.
- Diisocyanate Compound The diisocyanate compound represented by the above general formula (4) is obtained, for example, by the addition reaction of a triisocyanate compound and an equivalent of a monofunctional alcohol or monofunctional amine compound having an unsaturated group.
- a triisocyanate compound Although the thing shown below, for example is mentioned, it is not limited to this.
- Examples of the monofunctional alcohol or monofunctional amine compound having an unsaturated group include, but are not limited to, those shown below.
- a method of introducing an unsaturated group into the side chain of the polyurethane resin a method of using a diisocyanate compound containing an unsaturated group in the side chain as a raw material for producing a polyurethane resin is preferable.
- a diisocyanate compound obtainable by subjecting a triisocyanate compound and a monofunctional alcohol or unsaturated monofunctional alcohol or an equivalent of a monofunctional amine compound to an addition reaction, which has an unsaturated group in the side chain For example, although what is shown below is mentioned, it is not limited to this.
- the specific polyurethane resin used in the present invention is, for example, a diisocyanate compound containing the above-mentioned unsaturated group from the viewpoint of improving the compatibility with other components in the polymerizable composition and improving the storage stability.
- Other diisocyanate compounds can be copolymerized.
- diisocyanate compound to be copolymerized Preferred is a diisocyanate compound represented by the following general formula (6).
- L 1 represents a divalent aliphatic or aromatic hydrocarbon group which may have a substituent. If necessary, L 1 may have other functional groups which do not react with isocyanate groups, such as ester, urethane, amide and ureido groups.
- diisocyanate compound represented by the above general formula (6) examples include those shown below. That is, 2,4-tolylene diisocyanate, dimer of 2,4-tolylene diisocyanate, 2,6-tolylene diisocyanate, p-xylylene diisocyanate, m-xylylene diisocyanate, 4,4'-diphenylmethane diisocyanate, Aromatic diisocyanate compounds such as 1,5-naphthylene diisocyanate, 3,3'-dimethylbiphenyl-4,4'-diisocyanate, etc .; Aliphatic diisocyanate compounds such as hexamethylene diisocyanate, trimethylhexamethylene diisocyanate, lysine diisocyanate, dimer acid diisocyanate etc .; Cycloaliphatic diisocyanate compounds such as isophorone diisocyanate, 4,4'-methylenebis (cyclohexyl is
- Diol compound As a diol compound represented by the said General formula (5), a polyether diol compound, a polyester diol compound, a polycarbonate diol compound etc. are mentioned widely.
- a method of using a diol compound containing an unsaturated group in the side chain is also suitable as a raw material for polyurethane resin production.
- Such diol compounds may be commercially available, for example, trimethylolpropane monoallyl ether, or halogenated diol compounds, triol compounds, aminodiol compounds, carboxylic acids containing an unsaturated group, acids It may be a compound easily produced by reaction with a chloride, isocyanate, alcohol, amine, thiol, halogenated alkyl compound. Although the compound shown below is mentioned as a specific example of these compounds, It is not limited to this.
- a more preferable binder resin in the present invention it is obtained by using a diol compound represented by the following general formula (G) as at least one of diol compounds having an ethylenically unsaturated bonding group in the synthesis of polyurethane.
- G a diol compound represented by the following general formula (G) as at least one of diol compounds having an ethylenically unsaturated bonding group.
- Polyurethane resins can be mentioned.
- R 1 to R 3 each independently represent a hydrogen atom or a monovalent organic group
- A represents a divalent organic residue
- X is an oxygen atom, a sulfur atom, or N (R 12 ) — is represented
- R 12 represents a hydrogen atom or a monovalent organic group.
- R 1 ⁇ R 3 and X in the general formula (G) said a general formula (1) the same meaning as R 1 ⁇ R 3 and X in preferred embodiments versa.
- the specific polyurethane resin used in the present invention is, for example, a diol compound containing the above-mentioned unsaturated group from the viewpoint of improving the compatibility with other components in the polymerizable composition and improving the storage stability.
- Other diol compounds can be copolymerized.
- the polyether diol compound mentioned above, a polyester diol compound, and a polycarbonate diol compound can be mentioned, for example.
- polyether diol compounds compounds represented by the following formulas (7), (8), (9), (10) and (11), and random coweights of ethylene oxide and propylene oxide having a hydroxyl group at the terminal Coalescence is mentioned.
- R 14 represents a hydrogen atom or a methyl group
- X 1 represents the following group.
- a, b, c, d, e, f and g each represent an integer of 2 or more, preferably an integer of 2 to 100.
- polyether diol compounds represented by the above formulas (7) and (8) include those shown below. That is, diethylene glycol, triethylene glycol, tetraethylene glycol, pentaethylene glycol, hexaethylene glycol, heptaethylene glycol, octaethylene glycol, di-1,2-propylene glycol, tri-1,2-propylene glycol, tetra-1, 2-propylene glycol, hexa-1,2-propylene glycol, di-1,3-propylene glycol, tri-1,3-propylene glycol, tetra-1,3-propylene glycol, di-1,3-butylene glycol, Tri-1,3-butylene glycol, hexa-1,3-butylene glycol, polyethylene glycol having a weight-average molecular weight of 1,000, polyethylene glycol having a weight-average molecular weight of 1,500, a poly of weight-average molecular weight of 2,000 Tyrene glycol, polyethylene glycol having
- polyether diol compound represented by the above formula (9) examples include those shown below. That is, it is Sanyo Chemical Industries, Ltd. make (brand name) PTMG650, PTMG1000, PTMG2000, PTMG3000 grade
- polyether diol compound represented by the above formula (10) examples include those shown below. That is, Sanyo Chemical Industries, Ltd. (trade name) New Pole PE-61, New Pole PE-62, New Pole PE-64, New Pole PE-68, New Pole PE-71, New Pole PE-74, It is New Pole PE-75, New Pole PE-78, New Pole PE-108, New Pole PE-128, New Pole PE-61, and the like.
- polyether diol compound represented by the above formula (11) examples include those shown below. That is, Sanyo Chemical Industries, Ltd. (trade name) New Pole BPE-20, New Pole BPE-20 F, New Pole BPE-20 NK, New Pole BPE-20 T, New Pole BPE-20 G, New Pole BPE-40, Newpole BPE-60, Newpole BPE-100, Newpole BPE-180, Newpole BPE-2P, Newpole BPE-23P, Newpole BPE-3P, Newpole BPE-5P, and the like.
- random copolymer of ethylene oxide having a hydroxyl group at the end and propylene oxide examples include those shown below. That is, Sanyo Chemical Industries, Ltd. (trade name) New Pole 50HB-100, New Pole 50 HB-260, New Pole 50 HB-400, New Pole 50 HB-660, New Pole 50 HB-2000, New Pole 50 HB, etc. It is.
- L 2 , L 3 and L 4 may be the same or different and each represents a divalent aliphatic or aromatic hydrocarbon group, and L 5 is a divalent aliphatic carbon Represents a hydrogen group.
- L 2 to L 4 each represent an alkylene group, an alkenylene group, an alkynylene group or an arylene group, and L 5 represents an alkylene group.
- other functional groups which do not react with the isocyanate group such as ether, carbonyl, ester, cyano, olefin, urethane, amide, ureido group or halogen atom may be present.
- n1 and n2 is an integer of 2 or more, preferably an integer of 2 to 100.
- a polycarbonate diol compound there is a compound represented by Formula (14).
- L 6 may be the same or different and each represents a divalent aliphatic or aromatic hydrocarbon group.
- L 6 represents an alkylene group, an alkenylene group, an alkynylene group or an arylene group.
- n3 is an integer of 2 or more, preferably an integer of 2 to 100.
- diol compound represented by the above formula (12), (13) or (14) examples include (Exemplary compound No. 1) to (Exemplary compound No. 18) shown below.
- n represents an integer of 2 or more.
- a diol compound having a substituent which does not react with an isocyanate group can be used in combination.
- diol compounds include, for example, those shown below.
- L 7 and L 8 may be the same or different, and a substituent (for example, an alkyl group, an aralkyl group, an aryl group, an alkoxy group, an aryloxy group, -F,- It represents a divalent aliphatic hydrocarbon group, an aromatic hydrocarbon group or a heterocyclic group which may have each group such as halogen atoms such as Cl, -Br and -I. If necessary, L 7 and L 8 may have other functional groups which do not react with the isocyanate group, such as a carbonyl group, an ester group, a urethane group, an amide group, a ureido group and the like. L 7 and L 8 may form a ring.
- a substituent for example, an alkyl group, an aralkyl group, an aryl group, an alkoxy group, an aryloxy group, -F,- It represents a divalent aliphatic hydrocarbon group, an aromatic hydrocarbon group or
- a diol compound having a carboxyl group can be used in combination.
- diol compounds include those shown in the following formulas (17) to (19).
- R 15 is a hydrogen atom or a substituent (eg, a cyano group, a nitro group, -F, -Cl, -Br, -I or another halogen atom, -CONH 2 , -COOR 16 , -OR 16 , -NHCONHR 16 , -NHCOOR 16 , -NHCOR 16 , -OCONHR 16 (wherein, R 16 represents an alkyl group having 1 to 10 carbon atoms or an aralkyl group having 7 to 15 carbon atoms).
- a substituent eg, a cyano group, a nitro group, -F, -Cl, -Br, -I or another halogen atom, -CONH 2 , -COOR 16 , -OR 16 , -NHCONHR 16 , -NHCOOR 16 , -NHCOR 16 , -OCONHR 16 (wherein, R 16 represents an alkyl group having 1
- each group represents an alkyl group, an aralkyl group, an aryl group, an alkoxy group, or an aryloxy group, each of which preferably has a hydrogen atom, an alkyl group having 1 to 8 carbon atoms, or 6 to 6 carbon atoms. It represents 15 aryl groups.
- L 9 , L 10 and L 11 may be the same as or different from each other, and may have a single bond or a substituent (eg, each of alkyl, aralkyl, aryl, alkoxy and halogeno groups is preferable).
- L 9 to L 11 may have other functional groups which do not react with isocyanate groups, such as carbonyl, ester, urethane, amide, ureido and ether groups. Note that two or three of R 15 and L 9 to L 11 may form a ring.
- Ar represents a trivalent aromatic hydrocarbon group which may have a substituent, and preferably represents an aromatic group having 6 to 15 carbon atoms.
- diol compound having a carboxyl group represented by the above formulas (17) to (19) include those shown below. 3,5-dihydroxybenzoic acid, 2,2-bis (hydroxymethyl) propionic acid, 2,2-bis (2-hydroxyethyl) propionic acid, 2,2-bis (3-hydroxypropyl) propionic acid, Bis (hydroxymethyl) acetic acid, bis (4-hydroxyphenyl) acetic acid, 2,2-bis (hydroxymethyl) butyric acid, 4,4-bis (4-hydroxyphenyl) pentanoic acid, tartaric acid, N, N-dihydroxyethylglycine , N, N-bis (2-hydroxyethyl) -3-carboxy-propionamide and the like.
- the polyurethane resin having an ethylenically unsaturated bond group in the side chain is a resin further having a carboxyl group in the side chain, and more specifically, the ethylenically unsaturated bond group is A polyurethane resin having 0.3 meq / g or more and having a carboxyl group of 0.4 meq / g or more in the side chain is particularly preferably used as the binder polymer of the present invention.
- a compound obtained by ring-opening a tetracarboxylic acid dianhydride represented by the following formulas (20) to (22) with a diol compound may also be used in the synthesis of the specific polyurethane resin. it can.
- L 12 may have a single bond or a substituent (for example, each group of alkyl, aralkyl, aryl, alkoxy, halogeno, ester and amide is preferable).
- a substituent for example, each group of alkyl, aralkyl, aryl, alkoxy, halogeno, ester and amide is preferable.
- an aliphatic or aromatic hydrocarbon group -CO-, -SO-, -SO 2- , -O- or S-, preferably a single bond and a divalent aliphatic carbon having 1 to 15 carbon atoms It represents a hydrogen group, -CO-, -SO 2- , -O- or S-.
- R 17 and R 18, which may be the same or different, each represents a hydrogen atom, an alkyl group, an aralkyl group, an aryl group, an alkoxy group or a halogeno group, preferably a hydrogen atom or an alkyl having 1 to 8 carbon atoms And a group having 6 to 15 carbon atoms, an alkoxy group having 1 to 8 carbon atoms, or a halogeno group. Also, two of L 12 , R 17 and R 18 may combine to form a ring.
- R 19 and R 20 which may be the same or different, each represents a hydrogen atom, an alkyl group, an aralkyl group, an aryl group or a halogeno group, preferably a hydrogen atom, an alkyl having 1 to 8 carbon atoms, or 6 carbon atoms It represents -15 aryl groups. Also, two of L 12 , R 19 and R 20 may combine to form a ring.
- A represents a mononuclear or polynuclear aromatic ring. Preferably, it represents an aromatic ring having 6 to 18 carbon atoms.
- Specific examples of the compound represented by the above formula (20), (21) or (22) include those shown below. That is, pyromellitic dianhydride, 3,3 ', 4,4'-benzobenzoone tetracarboxylic dianhydride, 3,3', 4,4'-diphenyltetracarboxylic dianhydride, 2, 3,6,7-Naphthalenetetracarboxylic acid dianhydride, 1,4,5,8-Naphthalenetetracarboxylic acid dianhydride, 4,4'-sulfonyldiphthalic acid dianhydride, 2,2-bis (3 , 4-Dicarboxyphenyl) propane dianhydride, bis (3,4-dicarboxyphenyl) ether dianhydride, 4,4 '-[3,3'-(alkyl phosphoryl diphenylene) -bis (iminocarbonyl) ] Diphthalic dianhydride,
- Aromatic tetracarboxylic acid dianhydrides such as adducts of hydroquinone diacetate and trimet anhydride, adducts of diacetyl diamine and trimet anhydride, and the like; 5- (2,5-dioxotetrahydrofuryl) -3-methyl- 3-cyclohexene-1,2-dicarboxylic acid anhydride (manufactured by Dainippon Ink and Chemicals, Inc., Epiclon B-4400), 1,2,3,4-cyclopentane tetracarboxylic acid dianhydride, 1,2 Alicyclic tetracarboxylic acid dianhydrides such as 1,4,5-cyclohexanetetracarboxylic acid dianhydride, tetrahydrofuran tetracarboxylic acid dianhydride; 1,2,3,4-butanetetracarboxylic acid dianhydride, 1, And aliphatic tetracarboxylic acid
- a method for introducing a compound obtained by ring-opening these tetracarboxylic acid dianhydrides with a diol compound into a polyurethane resin there are, for example, the following methods. a) A method of reacting an alcohol-terminated compound obtained by ring-opening a tetracarboxylic acid dianhydride with a diol compound and a diisocyanate compound. b) A method of reacting an alcohol-terminated urethane compound obtained by reacting a diisocyanate compound under an excess of a diol compound with tetracarboxylic acid dianhydride.
- diol compound used for the ring opening reaction at this time examples include those shown below. That is, ethylene glycol, diethylene glycol, triethylene glycol, tetraethylene glycol, propylene glycol, dipropylene glycol, polyethylene glycol, polypropylene glycol, neopentyl glycol, 1,3-butylene glycol, 1,6-hexanediol, 2-butene- 1,4-diol, 2,2,4-trimethyl-1,3-pentanediol, 1,4-bis- ⁇ -hydroxyethoxycyclohexane, cyclohexanedimethanol, tricyclodecanedimethanol, hydrogenated bisphenol A, hydrogenated Bisphenol F, ethylene oxide adduct of bisphenol A, propylene oxide adduct of bisphenol A, ethylene oxide adduct of bisphenol F, bisphenol F pro Olefin oxide adduct, Ethylene oxide adduct of hydrogenated bisphenol A,
- the specific polyurethane resin which can be used in the present invention is synthesized by heating the above-mentioned diisocyanate compound and diol compound in an aprotic solvent by adding a known catalyst having an activity according to the respective reactivity.
- the molar ratio (M a : M b ) of diisocyanate and diol compound used in the synthesis is preferably 1: 1 to 1.2: 1, and the desired molecular weight or viscosity is desired by treating with alcohols or amines, etc.
- the product of physical properties of is finally synthesized in the form in which the isocyanate group does not remain.
- the ethylenically unsaturated bond group is 0.3 meq / g or more, further 0.35 to the side chain. It is preferable to contain 1.50 meq / g. Furthermore, a polyurethane resin containing 0.4 meq / g or more, more preferably 0.45 to 1.00 meq / g of carboxyl group in the side chain together with the ethylenically unsaturated bond group is particularly preferable as the binder polymer of the present invention.
- the molecular weight of the specific polyurethane resin according to the present invention is preferably 10,000 or more, and more preferably 40,000 to 200,000, in terms of weight average molecular weight.
- the weight average molecular weight is excellent in the strength of the image area within this range, and the developability of the non-image area by the alkaline developer Excellent.
- the specific polyurethane resin according to the present invention those having an unsaturated group at the polymer end and the main chain are also suitably used.
- the crosslinking reactivity is further improved between the polymerizable compound and the specific polyurethane resin or between the specific polyurethane resin, and the photocured product strength is increased.
- the specific polyurethane resin is used for the colored curable composition, it is possible to form a colored region excellent in strength.
- the unsaturated group it is particularly preferable to have a carbon-carbon double bond in view of the easiness of occurrence of a crosslinking reaction.
- an unsaturated group to the polymer end
- the unsaturated group is preferably introduced into the polymer side chain rather than the polymer end from the viewpoint that the introduction amount can be easily controlled, the introduction amount can be increased, and the crosslinking reaction efficiency is improved.
- the ethylenic unsaturated bond group to be introduced is preferably a methacryloyl group, an acryloyl group or a styryl group, more preferably a methacryloyl group or an acryloyl group, from the viewpoint of the formation of a crosslinked cured film.
- a methacryloyl group is more preferable from the viewpoint of coexistence with the formation property of a crosslinked cured film and the raw storage property.
- the introduction amount of methacryloyl group is preferably 0.30 meq / g or more, and more preferably in the range of 0.35 to 1.50 meq / g. That is, as the binder polymer of the present invention, a polyurethane resin having a methacryloyl group introduced in the range of 0.35 to 1.50 meq / g in the side chain is the most preferable embodiment.
- a method of introducing an unsaturated group into the main chain there is a method of using a diol compound having an unsaturated group in the main chain direction for the synthesis of a polyurethane resin.
- the diol compound having an unsaturated group in the main chain direction include the following compounds. That is, cis-2-butene-1,4-diol, trans-2-butene-1,4-diol, polybutadiene diol and the like.
- the specific polyurethane resin according to the present invention can also be used in combination with an alkali-soluble polymer containing a polyurethane resin having a structure different from that of the specific polyurethane resin.
- an alkali-soluble polymer containing a polyurethane resin having a structure different from that of the specific polyurethane resin for example, as the specific polyurethane resin, it is possible to use a polyurethane resin containing an aromatic group in the main chain and / or the side chain in combination.
- the specific styrenic resin used in the present invention is a styrenic double bond (styrene and ⁇ -methyl styrenic double bond) represented by the following general formula (23) in its side chain, What has at least one among the vinyl pyridinium groups represented by 24) is mentioned.
- R 21 represents a hydrogen atom or a methyl group.
- R 22 represents any substitutable atom or atomic group.
- k represents an integer of 0 to 4;
- the styrenic double bond represented by the general formula (23) is linked to the polymer main chain via a single bond or a linking group consisting of arbitrary atoms or atomic groups There is no limit.
- Preferred examples of the repeating unit of the polymer compound having a functional group represented by the general formula (23) are shown below, but the present invention is not limited to these examples.
- R 23 represents a hydrogen atom or a methyl group.
- R 24 represents any substitutable atom or atomic group.
- m represents an integer of 0 to 4;
- a - represents an anion.
- the pyridinium ring may be in the form of benzopyridinium in which a benzene ring is condensed as a substituent, and in this case, it includes a quinolium group and an isoquinolium group.
- the vinyl pyridinium group represented by the general formula (24) is linked to the polymer main chain via a single bond or a linking group consisting of any atom or atomic group, and there is no particular restriction on the bonding method Absent.
- Preferred examples of the repeating unit of the polymer compound having a functional group represented by the general formula (24) are shown below, but the present invention is not limited to these examples.
- the specific styrenic resin As one of the methods for synthesizing the specific styrenic resin according to the present invention, it has a functional group represented by the above general formula (23) or (24) and can be copolymerized with other copolymerization components.
- the method of copolymerizing the monomers which have a functional group using a well-known copolymerization method is mentioned.
- the specific styrenic resin may be a homopolymer having only one of any one of functional groups represented by the general formulas (23) and (24), or any one of them Alternatively, it may be a copolymer having two or more of each of the two functional groups.
- copolymers with other copolymerizable monomers not containing these functional groups.
- a copolymerization monomer it is preferable to select a carboxyl group-containing monomer, for example, for the purpose of imparting solubility to an aqueous alkali solution to the polymer, and acrylic acid, methacrylic acid and acrylic acid 2-carboxyethyl ester. Examples thereof include methacrylic acid 2-carboxyethyl ester, crotonic acid, maleic acid, fumaric acid, maleic acid monoalkyl ester, fumaric acid monoalkyl ester, 4-carboxystyrene and the like.
- styrene 4-methylstyrene, 4-hydroxystyrene, 4-acetoxystyrene, 4-carboxystyrene, 4-aminostyrene, chloromethylstyrene, 4-methoxystyrene Derivatives of styrene, vinylphosphonic acid, vinylsulfonic acid and their salts, styrenesulfonic acid and their salts, 4-vinylpyridine, 2-vinylpyridine, N-vinylimidazole, N-vinylcarbazole, 4-vinylbenzyltrimethylammonium chloride N-vinylimidazole quaternized with methyl chloride, 4-vinylbenzyl
- the above-mentioned copolymer When used as the specific styrenic resin according to the present invention, it has a functional group represented by the general formula (23) and / or the general formula (24) occupied in the entire copolymer composition As a ratio of a repeating unit, it is preferable that it is 20 mass% or more, and it is more preferable that it is 40 mass% or more. Within this range, a highly sensitive crosslinking system excellent in the effects of the present invention is provided.
- the specific styrenic resin according to the present invention may become water-soluble by the inclusion of a quaternary salt structure in its repeating unit.
- the colored curable composition of the present invention containing such a resin is used to form a colored area of a color filter, it can be developed with water after exposure.
- a quaternary salt structure is contained in a linking group having a functional group represented by the general formula (23) in a repeating unit and connecting the main chain and the functional group represented by the general formula (23)
- a linking group having a functional group represented by the general formula (23) in a repeating unit and connecting the main chain and the functional group represented by the general formula (23)
- it may be a homopolymer having such a structure, but in other cases, it may be a copolymer with other copolymerizable monomers listed below preferable.
- 4-vinylbenzyl trimethyl ammonium chloride, acryloyloxyethyl trimethyl ammonium chloride, methacryloyl oxyethyl trimethyl ammonium chloride, quaternized by methyl chloride of dimethylaminopropyl acrylamide, quaternized by methyl chloride of N-vinyl imidazole, 4- 4- Vinyl benzyl pyridinium chloride and the like are preferably used.
- the functional group represented by the general formula (24) is contained in the repeating unit, it may be a homopolymer or may be a copolymer with the other copolymerizable monomer. .
- the proportion of the repeating unit having a functional group represented by the general formula (23) and / or the general formula (24) is preferably 20% by mass or more, and the introduction of other repeating units Can be selected freely according to the purpose.
- the molecular weight of the specific styrenic resin according to the present invention is preferably in the range of 10,000 to 300,000 in weight average molecular weight, more preferably in the range of 15,000 to 200,000, and most preferably in the range of 20,000 to 150,000. .
- the weight average molecular weight is excellent in the strength of the image area within this range, and the developability of the non-image area by the alkaline developer Excellent.
- (a-3) a specific dispersion resin other than the specific polyurethane resin and the specific styrenic resin will be described.
- the novolak resin having an ethylenically unsaturated group in the side chain for example, the polymer described in JP-A No. 9-269596, the method described in JP-A No. 2002-62648 can be used to obtain an ethylenically non-ethylenically unsaturated group.
- transduced the saturated bond are mentioned.
- an acetal resin which has an ethylenically unsaturated bond in a side chain resin etc. which are described in Unexamined-Japanese-Patent No.
- polyamide resins having an ethylenically unsaturated bond in the side chain include the resins described in Japanese Patent Application No. 2003-321022 or the polyamide resins cited therein, as described in JP-A-2002-62648.
- transduced the ethylenically unsaturated bond to the side chain by the method are mentioned.
- a polyimide resin having an ethylenically unsaturated bond in a side chain for example, the method described in JP-A-2002-62648 can be used for the resin described in Japanese Patent Application No. 2003-339785 or the polyimide resin cited therein. And resins having an ethylenically unsaturated bond introduced in the side chain.
- the (a-3) specific dispersion resin has a function as a binder polymer, and when preparing the (A) pigment dispersion, it is added and dispersed together with the (a-1) pigment, but the content is A)
- the solid content of the pigment dispersion liquid is 2 to 70% by mass, more preferably 4 to 50% by mass, and still more preferably 5 to 40% by mass.
- dispersants for the purpose of improving the dispersion stability and / or adjusting the developability, dispersants other than the above-mentioned specific dispersion resin can be used.
- dispersants for example, known pigment dispersants and surfactants can be appropriately selected and used.
- organosiloxane polymer KP 341 manufactured by Shin-Etsu Chemical Co., Ltd.
- Cationic surfactants such as 95 (manufactured by Kyoeisha Chemical Industry Co., Ltd.), W001 (manufactured by Yusho Co., Ltd.), etc .; polyoxyethylene lauryl ether, polyoxyethylene stearyl ether, polyoxyethylene oleyl ether, polyoxyethylene Nonionic surfactants such as octyl phenyl ether, polyoxyethylene nonyl phenyl ether, polyethylene glycol dilaurate, polyethylene glycol distearate, sorbitan fatty acid ester; and anionic systems such as W004, W005, W017 (manufactured by Yusho Co., Ltd.) Surfactants: EFKA-46, EFKA-47, EFKA-47EA, EFKA Polymer 100, EFKA Polymer 400, EFKA Polymer 401, EFKA Polymer 450 (all manufactured by Ciba Specialty Chemicals) Polymer dispersants such as Dispers Aid 6, Dispers
- Disperbyk 101, 103, 106, 108, 109, 111, 112, 116, 130, 140, 142 162,163,164,166,167,170,171,174,176,180,182,2000,2001,2050,2150 BYK Chemie Co., Ltd.
- Other examples include oligomers or polymers having a polar group at the molecular terminal or side chain, such as acrylic copolymers.
- the content of the other dispersant in the pigment dispersion is preferably 1 to 100 parts by mass, and more preferably 3 to 70 parts by mass with respect to 100 parts by mass of the (a-1) pigment described above.
- pigment derivatives other than the (a-2) specific pigment derivative according to the present invention can be added to the (A) pigment dispersion in the present invention.
- a pigment derivative having a part having affinity with the dispersant or a polar group introduced can be adsorbed on the pigment surface and used as an adsorption point of the dispersant, so the pigment can be used as fine particles. It can be dispersed in a curable composition to prevent its reaggregation, and is effective in constructing a color filter having high contrast and excellent transparency.
- the other pigment derivative is a compound in which an organic pigment is used as a matrix and an acid group, a basic group or an aromatic group is introduced as a substituent to a side chain.
- organic pigments are quinacridone pigments, phthalocyanine pigments, azo pigments, quinophthalone pigments, isoindoline pigments, isoindolinone pigments, quinoline pigments, diketopyrrolopyrrole pigments, benzimidazolone pigments, etc.
- light yellow aromatic polycyclic compounds such as naphthalenes, anthraquinones, triazines and quinolines which are not called dyes are also included.
- the materials described in Japanese Patent Application Publication No. 2005-234478, Japanese Patent Application Publication No. 2003-240938, Japanese Patent Application Publication No. 2001-356210, etc. can be used.
- the content of the other pigment derivative according to the present invention in the pigment dispersion is preferably 0 to 30 parts by mass, and more preferably 3 to 20 parts by mass with respect to 100 parts by mass of the pigment.
- the dispersion can be favorably performed while the viscosity is suppressed low, and the dispersion stability after dispersion can be improved, and excellent color characteristics with high transmittance can be obtained, and color When making the filter, it can be configured to have high contrast with good color characteristics.
- (A) Pigment Dispersion As a method of preparing a pigment dispersion containing the above (a-1) to (a-3) and optional components, for example, (a-1) pigment and (a-3) specific dispersion resin are mixed in advance, It can be carried out by finely dispersing one dispersed by a homogenizer or the like using a bead disperser (for example, Dispermat manufactured by GETZMANN Co., Ltd.) using zirconia beads or the like. The dispersion time is preferably about 3 to 6 hours.
- the specific pigment derivative can be added at any step for forming the pigment dispersion, but it is preferable to add it at the time of the micronization step and / or the fine dispersion.
- the coloring and curing of the present invention is achieved by blending the (A) pigment dispersion prepared as described above with (B) an oxime ester initiator, (C) a polymerizable compound described later, and other components.
- the amount of the pigment dispersion (A) contained in the colored curable composition of the present invention is preferably in the range of 5 to 90% by mass, and more preferably in the range of 25 to 85% by mass.
- the amount of the pigment (a-1) contained in the colored curable composition of the present invention is preferably in the range of 25 to 65% by mass in the solid content, by containing the pigment dispersion (A) in the above-mentioned blending amount. Even in a thin layer, colored regions having a sufficient hue can be formed.
- ⁇ (B) oxime ester initiator> In the colored curable composition of the present invention, particularly when forming a thin film, since it is necessary to increase the pigment concentration as described above, it is not possible to add a large amount of initiator for generating radicals. In addition, since the concentration of the pigment is high, the concentrations of the sensitizer and the initiator can not be sufficiently obtained, and the effect of improving the curability by the initiation mechanism by electron transfer and energy transfer using the sensitizer can not be sufficiently obtained. Particularly when using the curable composition of the present invention to form a high-definition fine-colored area, such as a solid-state imaging device, a triazine-based initiation which generates a halogen upon exposure even with high sensitivity.
- the agent may contaminate a light source such as a stepper exposure apparatus and can not be used. For this reason, under the conditions, it is possible to cope with a wide range of light sources of 300 nm or more, and an oxime compound which is directly decomposed by light is used as an initiator, ie, an oxime ester initiator (hereinafter referred to as oxime initiator). It may be preferable to use the In particular, the use of the oxime type initiator is more effective in a curable composition containing a pigment in an amount of 30% by mass or more, further 40% by mass or more based on the total solid content of the curable composition and having a low ratio of polymerizable components.
- R 1 is a monovalent organic group, preferably an organic group having a carbonyl group or a sulfo group in the structure, and from the viewpoint of sensitivity and stability, R 1 is preferably a monovalent organic group represented by the following Formula (III) or Formula (IV).
- R 4 represents a monovalent organic group, and specifically, an alkyl group having 1 to 20 carbon atoms, an alkenyl group, an alkynyl group, a cycloalkyl group, a cycloalkenyl group, It is preferable that it is a cycloalkynyl group, an aryl group, a heterocyclic group, or an alkoxy group.
- substituents may further have a substituent, and as the substituent which can be introduced, a halogen atom, a hydroxyl group, an alkyl group, an alkenyl group, an alkynyl group, a cycloalkyl group, a cycloalkenyl group, a cycloalkynyl group, an aryl Group, heterocyclic group, cyano group, alkoxy group, aryloxy group, thioalkoxy group, thioaryloxy group, carboxyl group, alkoxycarbonyl group, sulfo group, sulfonamido group, urea group, thiourea group, amino group, amido group, A carbonyl group, a nitro group or a substituent having these groups can be mentioned.
- the substituent is most preferably a substituent represented by General Formula (III), and R 4 is an alkyl group or an aryl group.
- R 2 represents a hydrogen atom or a monovalent organic group
- examples of the organic group include, for example, 1 to 20 carbon atoms
- examples thereof include an alkyl group, an alkenyl group, an alkynyl group, a cycloalkyl group, a cycloalkenyl group, a cycloalkynyl group, an aryl group, a heterocyclic group and an alkoxy group.
- These may further have a substituent, and as the substituent which can be introduced, a halogen atom, a hydroxyl group, an alkyl group, an alkenyl group, an alkynyl group, a cycloalkyl group, a cycloalkenyl group, a cycloalkynyl group, cyano Group, aryl group, heterocyclic group, alkoxy group, aryloxy group, thioalkoxy group, thioaryloxy group, carboxyl group, alkoxycarbonyl group, sulfo group, sulfonamido group, urea group, thiourea group, amino group, amido group, A carbonyl group, a nitro group or a substituent having these groups can be mentioned.
- R 3 represents a monovalent organic group, and examples thereof include alkyl group having 1 to 20 carbon atoms, alkenyl group, alkynyl group, cycloalkyl group, cycloalkenyl group, cycloalkynyl group, aryl group, heterocyclic group and alkoxy group It can be mentioned.
- These may further have a substituent, and as the substituent which can be introduced, a halogen atom, a hydroxyl group, an alkyl group, an alkenyl group, an alkynyl group, a cycloalkyl group, a cycloalkenyl group, a cycloalkynyl group, an aryl Group, cyano group, heterocyclic group, alkoxy group, aryloxy group, thioalkoxy group, thioaryloxy group, carboxyl group, alkoxycarbonyl group, sulfo group, sulfonamido group, urea group, thiourea group, amino group, amido group, A carbonyl group, a nitro group or a substituent having these groups can be mentioned.
- R 3 is preferably an aryl group or a heterocyclic group, and in the case of an aryl group, it is preferable that R 3 be substituted with a substituent having a nitrogen atom or a sulfur atom, When it is a hetero ring, it preferably contains a nitrogen atom, an oxygen atom or a sulfur atom.
- R 3 is preferably an aryl group or a heterocyclic group, and in the case of an aryl group, it is preferable that R 3 be substituted with a substituent having a nitrogen atom or a sulfur atom, When it is a hetero ring, it preferably contains a nitrogen atom, an oxygen atom or a sulfur atom.
- Specific examples of the (B) oxime type initiator which can be preferably used in the present invention are shown below, but the present invention is not limited thereto.
- R 3 in the above general formula (I) or (II) is an aryl substituted with a substituent having a sulfur atom, or a nitrogen-containing aromatic ring
- R 1 Is a compound which is an aryl group or an alkyl group.
- R 3 is a nitrogen-containing aromatic ring
- R 1 is an alkyl group having 1 to 3 carbon atoms.
- the content of the (B) oxime type initiator in the colored curable composition of the present invention is preferably in the range of 0.3 to 30% by mass, preferably 1 to 20% by mass, with respect to the total solids of the composition. More preferably, it is in the range of (B) Since the oxime type initiator has high sensitivity, in the colored curable composition of the present invention containing a large amount of pigment, it is possible to form an excellent cured film even in the above compounding amount.
- (C) a polymerizable compound is used together with the (B) oxime type initiator.
- the polymerizable compound that can be used in the present invention include general radically polymerizable compounds, and a compound widely known as a compound having an ethylenically unsaturated double bond in the relevant industrial field can be used without particular limitation. These have chemical forms such as monomers, prepolymers, ie, dimers, trimers and oligomers, or mixtures thereof and copolymers thereof.
- Examples of the monomer and the copolymer thereof include unsaturated carboxylic acids (eg, acrylic acid, methacrylic acid, itaconic acid, crotonic acid, isocrotonic acid, maleic acid etc.), esters thereof and amides thereof.
- unsaturated carboxylic acids eg, acrylic acid, methacrylic acid, itaconic acid, crotonic acid, isocrotonic acid, maleic acid etc.
- esters thereof and amides thereof for this, an ester of an unsaturated carboxylic acid and an aliphatic polyhydric alcohol compound, and an amide of an unsaturated carboxylic acid and an aliphatic polyhydric amine compound are used.
- addition reaction products of unsaturated carboxylic acid esters or amides having a nucleophilic substituent such as hydroxyl group, amino group, mercapto group etc.
- monomers that are esters of aliphatic polyhydric alcohol compounds and unsaturated carboxylic acids include, as acrylic acid esters, ethylene glycol diacrylate, triethylene glycol diacrylate, 1,3-butanediol diacrylate, tetramethylene Glycol diacrylate, propylene glycol diacrylate, neopentyl glycol diacrylate, trimethylolpropane triacrylate, trimethylolpropane tri (acryloyloxypropyl) ether, trimethylolethane triacrylate, hexanediol diacrylate, 1,4-cyclohexanediol diacrylate Acrylate, tetraethylene glycol diacrylate, pentaerythritol diacrylate, pentaerythritol triac Rate, pentaerythritol tetraacrylate, dipentaerythritol diacrylate, dipentaerythritol hexa
- tetramethylene glycol dimethacrylate triethylene glycol dimethacrylate, neopentyl glycol dimethacrylate, trimethylolpropane trimethacrylate, trimethylolethane trimethacrylate, ethylene glycol dimethacrylate, 1,3-butanediol dimethacrylate, Hexanediol dimethacrylate, pentaerythritol dimethacrylate, pentaerythritol trimethacrylate, pentaerythritol tetramethacrylate, dipentaerythritol dimethacrylate, dipentaerythritol hexamethacrylate, sorbitol trimethacrylate, sorbitol tetramethacrylate, bis [p- (3-methacryloxy-) 2-hydroxy group Epoxy) phenyl] dimethyl methane, bis - [p- (
- crotonic acid esters include ethylene glycol diitaconate, tetramethylene glycol dicrotonate, pentaerythritol dicrotonate, and sorbitol tetradicrotonate.
- isocrotonic acid esters include ethylene glycol diisocrotonate, pentaerythritol diisocrotonate, and sorbitol tetraisocrotonate.
- maleic esters include ethylene glycol dimaleate, triethylene glycol dimaleate, pentaerythritol dimaleate, and sorbitol tetramaleate.
- esters for example, aliphatic alcohol-based esters described in JP-B-51-47334, JP-A-57-196231, JP-A-59-5240, JP-A-59-5241, JP-A-2-226149.
- Those having an aromatic skeleton as described above, those containing an amino group as described in JP-A-1-165613, and the like are also suitably used.
- the aforementioned ester monomers can also be used as a mixture.
- monomers of amides of aliphatic polyvalent amine compounds and unsaturated carboxylic acids include methylene bis-acrylamide, methylene bis-methacrylamide, 1,6-hexamethylene bis-acrylamide, 1,6-hexamethylene bis -Methacrylamide, diethylene triamine tris acrylamide, xylylene bis acrylamide, xylylene bis methacrylamide and the like.
- examples of other preferred amide-based monomers include those having a cyclohexylene structure described in JP-B-54-21726.
- urethane addition polymerization compounds produced by using an addition reaction of an isocyanate and a hydroxyl group are also suitable, and as such specific examples, for example, one molecule described in JP-B-48-41708.
- a vinyl urethane compound containing two or more polymerizable vinyl groups in one molecule in which a vinyl monomer containing a hydroxyl group represented by the following general formula (E) is added to a polyisocyanate compound having two or more isocyanate groups in Compounds etc. may be mentioned.
- CH 2 C (R 4) COOCH 2 CH (R 5) OH (E) (Wherein, R 4 and R 5 each represents H or CH 3.)
- urethane acrylates as described in JP-A-51-37193, JP-B-2-32293 and JP-B-2-16765, JP-B-58-49860 and JP-B-56-17654
- Urethane compounds having an ethylene oxide-based skeleton as described in JP-B-62-39417 and JP-B-62-39418 are also suitable.
- addition polymerizable compounds having an amino structure or a sulfide structure in the molecule as described in JP-A-63-277653, JP-A-63-260909, and JP-A-1-105238, It is possible to obtain a curable composition which is extremely excellent in photosensitive speed.
- polyester acrylates as described in JP-A-48-64183, JP-B-49-43191, JP-B-52-30490, and respective publications, epoxy resins and (meth) acrylic acid. Mention may be made of polyfunctional acrylates and methacrylates such as reacted epoxy acrylates.
- specific unsaturated compounds described in JP-B-46-43946, JP-B-1-44037, JP-B-1-40336, and vinyl phosphonic acid compounds described in JP-A-2-25493 can also be mentioned. .
- a structure containing a perfluoroalkyl group described in JP-A-61-22048 is preferably used.
- Japan Adhesiveness Association magazine vol. 20, no. also those introduced as photocurable monomers and oligomers on pages 7, 300-308 (1984) can be used.
- one molecule preferably contains two or more ethylenic unsaturated bonds, and more preferably three or more.
- urethane oligomers UAS-10, UAB-140 (Sanyo Gokusoku Pulp Co., Ltd.) , DPHA-40H (manufactured by Nippon Kayaku Co., Ltd.), UA-306H, UA-306T, UA-306I, AH-600, T-600, AI-600 (manufactured by Kyoeisha Co., Ltd.).
- bisphenol A diacrylate EO modified product pentaerythritol triacrylate, pentaerythritol tetraacrylate, dipentaerythritol pentaacrylate, dipentaerythritol hexaacrylate, tri (acryloyloxyethyl) isocyanurate, pentaerythritol tetraacrylate EO modified product
- Examples of commercially available dipentaerythritol hexaacrylate EO modified products include DPHA-40H (manufactured by Nippon Kayaku Co., Ltd.), UA-306H, UA-306T, UA-306I, AH-600, T-600 and AI-600. (Made by Kyoeisha) is more preferable.
- ethylenically unsaturated compounds having an acid group are also suitable, and examples of commercially available products include TO-756, which is a carboxyl group-containing trifunctional acrylate manufactured by Toagosei Co., Ltd., and a carboxyl group-containing pentafunctional acrylate One example is TO-1382.
- the polymerizable compound (C) may be used alone or in combination of two or more.
- the content of the (C) polymerizable compound in the colored curable composition of the present invention is, in terms of solid content, The range is preferably 4 to 80% by mass, and more preferably 7 to 50% by mass. In particular, when it is intended to form a cured film having a thickness of 0.8 ⁇ m or less, the content of the polymerizable compound (C) is in the range of 7 to 40% by mass, particularly 6 to 25% by mass in the total solid content. It is valid.
- a binder polymer can be further used, as necessary, for the purpose of improving film properties, adjusting developability, and the like. It is preferable to use a linear organic polymer as the binder.
- linear organic polymers known ones can be optionally used.
- a linear organic polymer which is soluble or swellable in water or weakly alkaline water is selected to enable water development or weakly alkaline water development.
- the linear organic polymer is selected and used not only as a film-forming agent but also according to the use as a water, weak alkaline water or organic solvent developer. For example, water development is possible with water-soluble organic polymers.
- radical polymers having a carboxylic acid group in the side chain such as JP-A-59-44615, JP-B-54-34327, JP-B-58-12577, JP-B-54-25957 Nos.
- monomers having a carboxyl group include acrylic acid, methacrylic acid, itaconic acid, crotonic acid, maleic acid, fumaric acid, 4-carboxystyrene and the like, and monomers having an acid anhydride include maleic anhydride and the like.
- monomers having an acid anhydride include maleic anhydride and the like.
- acidic cellulose derivatives having a carboxylic acid group in the side chain there are acidic cellulose derivatives having a carboxylic acid group in the side chain.
- a polymer having a hydroxyl group to which a cyclic acid anhydride is added is useful.
- alkali-soluble resin When used as a copolymer, monomers other than the monomers listed above can also be used as a compound to be copolymerized. Examples of other monomers include the following compounds (1) to (12).
- Vinyl ethers such as ethyl vinyl ether, 2-chloroethyl vinyl ether, hydroxyethyl vinyl ether, propyl vinyl ether, butyl vinyl ether, octyl vinyl ether, phenyl vinyl ether and the like.
- Vinyl esters such as vinyl acetate, vinyl chloroacetate, vinyl butyrate and vinyl benzoate.
- Styrenes such as styrene, ⁇ -methylstyrene, methylstyrene, chloromethylstyrene and p-acetoxystyrene.
- Vinyl ketones such as methyl vinyl ketone, ethyl vinyl ketone, propyl vinyl ketone, phenyl vinyl ketone and the like.
- Olefins such as ethylene, propylene, isobutylene, butadiene and isoprene.
- N-vinylpyrrolidone acrylonitrile, methacrylonitrile and the like.
- Unsaturated imides such as maleimide, N-acryloyl acrylamide, N-acetyl methacrylamide, N-propionyl methacrylamide, N- (p-chlorobenzoyl) methacrylamide and the like.
- a methacrylic acid-based monomer in which a hetero atom is bonded to the ⁇ position For example, compounds described in Japanese Patent Application No. 2001-115595, Japanese Patent Application No. 2001-115598, etc. can be mentioned.
- (meth) acrylic resins having an allyl group, a vinyl ester group and a carboxyl group in a side chain and double side chains described in JP-A-2000-187322 and JP-A-2002-62698 are preferable because it is excellent in the balance between film formability, sensitivity and developability.
- Japanese Examined Patent Publication No. 7-12004 Japanese Examined Patent Publication No. 7-120041, Japanese Examined Publication No. 7-120042, Japanese Examined Patent Publication No. 8-12424, Japanese Patent Laid-Open Publication 63-287944, Japanese Patent Laid-Open Publication 63-287947, Japanese Patent Laid-Open Publication 1- Urethane-based binder polymers containing an acid group as described in JP-A-271741 and Japanese Patent Application No. 10-116232, and urethane-based binder polymers having an acid group and a double bond as described in JP-A-2002-107918 in the side chain Is very excellent in strength, so it is advantageous in terms of printing durability and low exposure suitability.
- an acetal-modified polyvinyl alcohol-based binder polymer having an acid group described in European Patent 993966, European Patent 1204000, JP-A 2001-318463, etc. is excellent in the balance between film strength and developability and is preferable.
- polyvinyl pyrrolidone, polyethylene oxide and the like are useful as the water-soluble linear organic polymer.
- alcohol-soluble nylon, polyether of 2,2-bis- (4-hydroxyphenyl) -propane and epichlorohydrin, and the like are also useful for increasing the strength of the cured film.
- the weight average molecular weight of the binder polymer that can be used in the present invention is preferably 3,000 or more, more preferably 5,000 to 300,000, and the number average molecular weight is preferably 1,000 or more. And more preferably in the range of 2,000 to 250,000.
- the polydispersity (weight-average molecular weight / number-average molecular weight) is preferably 1 or more, and more preferably in the range of 1.1 to 10.
- These binder polymers may be any of random polymers, block polymers, graft polymers and the like.
- the binder polymer which can be used in the present invention can be synthesized by a conventionally known method.
- the solvent used in the synthesis include tetrahydrofuran, ethylene dichloride, cyclohexanone, methyl ethyl ketone, acetone, methanol, ethanol, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, 2-methoxyethyl acetate, diethylene glycol dimethyl ether, 1-methoxy -2-propanol, 1-methoxy-2-propyl acetate, N, N-dimethylformamide, N, N-dimethylacetamide, toluene, ethyl acetate, methyl lactate, ethyl lactate, dimethyl sulfoxide, water and the like.
- solvents may be used alone or in combination of two or more.
- combining the binder polymer which can be used in this invention well-known compounds, such as an azo initiator and a peroxide initiator, are mentioned.
- the binder polymer can also be added at the time of pigment dispersion, and can also be added at the time of preparing a curable composition using a dispersed dispersion.
- the amount of binder polymer added is preferably 0% by mass to 50% by mass, more preferably 2% by mass to 30% by mass, based on the total solid content of the curable composition.
- the preferred acid group of the binder polymer used for the purpose is a carboxyl group, a sulfonamide group, a sulfonic acid group, a phosphonic acid group or a phenol group, and the acid value is preferably 0.1 mmol / l from the viewpoint of developability and sensitivity. It is g to 10 mmol / g, more preferably 0.2 mmol / g to 5 mmol / g, and most preferably 0.3 mmol / g to 3 mmol / g.
- the colored curable composition of the present invention may further contain (B-2) another photopolymerization initiator other than the (B) oxime type initiator, as necessary.
- the other photopolymerization initiator (B-2) in the present invention is a compound which is decomposed by light to start or accelerate the polymerization of the polymerizable compound (C), and has absorption in a wavelength range of 300 to 500 nm. Is preferred.
- a photoinitiator can be used individually or in combination of 2 or more types.
- photopolymerization initiator for example, organic halogenated compounds, oxydiazole compounds, carbonyl compounds, ketal compounds, benzoin compounds, acridine compounds, organic peroxide compounds, azo compounds, coumarin compounds, azide compounds, metallocene compounds, hexaaryl compounds Rubiimidazole compounds, organic boric acid compounds, disulfonic acid compounds, onium salt compounds, acyl phosphine (oxide) compounds may be mentioned.
- an s-triazine derivative in which at least one mono-, di- or trihalogen-substituted methyl group is bonded to an s-triazine ring specifically, for example, 2,4,6- Tris (monochloromethyl) -s-triazine, 2,4,6-tris (dichloromethyl) -s-triazine, 2,4,6-tris (trichloromethyl) -s-triazine, 2-methyl-4,6- Bis (trichloromethyl) -s-triazine, 2-n-propyl-4,6-bis (trichloromethyl) -s-triazine, 2- ( ⁇ , ⁇ , ⁇ -trichloroethyl) -4,6-bis (trichloro) Methyl) -s-triazine, 2-phenyl-4,6-bis (trichloromethyl) -s-triazine, 2- (
- Examples of oxydiazole compounds include 2-trichloromethyl-5-styryl-1,3,4-oxodiazole, 2-trichloromethyl-5- (cyanostyryl) -1,3,4-oxodiazole Trichloromethyl-5- (naphth-1-yl) -1,3,4-oxodiazole, 2-trichloromethyl-5- (4-styryl) styryl-1,3,4-oxodiazole and the like .
- benzophenone derivatives such as benzophenone, Michler's ketone, 2-methylbenzophenone, 3-methylbenzophenone, 4-methylbenzophenone, 2-chlorobenzophenone, 4-bromobenzophenone, 2-carboxybenzophenone and the like, 2, 2-dimethoxy-2 -Phenylacetophenone, 2,2-diethoxyacetophenone, 1-hydroxycyclohexyl phenyl ketone, ⁇ -hydroxy-2-methylphenylpropanone, 1-hydroxy-1-methylethyl- (p-isopropylphenyl) ketone, 1-hydroxy -1- (p-dodecylphenyl) ketone, 2-methyl- (4 '-(methylthio) phenyl) -2-morpholino-1-propanone, 1,1,1-trichloromethyl- (p-butylphenyl) Ketones, acetophenone derivatives such as 2-benzyl-2-di
- ketal compounds include benzyl methyl ketal, benzyl- ⁇ -methoxyethylethyl acetal and the like.
- benzoin compounds include m benzoin isopropyl ether, benzoin isobutyl ether, benzoin methyl ether, methyl o-benzoyl benzoate and the like.
- acridine compound examples include 9-phenylacridine, 1,7-bis (9-acridinyl) heptane and the like.
- organic peroxide compound examples include trimethylcyclohexanone peroxide, acetylacetone peroxide, 1,1-bis (tert-butylperoxy) -3,3,5-trimethylcyclohexane, 1,1-bis (tert-butylperoxy) Oxy) cyclohexane, 2,2-bis (tert-butylperoxy) butane, tert-butyl hydroperoxide, cumene hydroperoxide, diisopropylbenzene hydroperoxide, 2,5-dimethylhexane-2,5-dihydroperoper Oxide, 1,1,3,3-tetramethylbutyl hydroperoxide, tert-butylcumyl peroxide, dicumyl peroxide, 2,5-dimethyl-2,5-di (tert-butylperoxy) hexane, 2 5-oxanoyl peroxide, succinic acid peroxide, benzoyl peroxide,
- Examples of the azo compound include azo compounds described in JP-A-8-108621.
- the coumarin compound for example, 3-methyl-5-amino-((s-triazin-2-yl) amino) -3-phenylcoumarin, 3-chloro-5-diethylamino-((s-triazin-2-yl) Etc. may be mentioned) amino) -3-phenylcoumarin, 3-butyl-5-dimethylamino-((s-triazin-2-yl) amino) -3-phenylcoumarin and the like.
- organic azide compounds described in U.S. Pat. No. 2,848,328, U.S. Pat. No. 2,852,379 and U.S. Pat. No. 2,940,853, 2,6-bis (4-azidobenzylidene) -4-ethyl Examples include cyclohexanone (BAC-E) and the like.
- metallocene compounds there can be mentioned, for example, JP-A-59-152396, JP-A-61-151197, JP-A-63-41484, JP-A-2-249, JP-A-2-4705, JP-A-H02-147.
- titanocene compounds described in JP-A 5-83588 for example, di-cyclopentadienyl-Ti-bis-phenyl, di-cyclopentadienyl-Ti-bis-2,6-difluorophen-1-yl, di-cyclopentadienyl -Cyclopentadienyl-Ti-bis-2,4-di-fluoropheny-l-yl, di-cyclopentadienyl-Ti-bis-2,4,6-trifluoropheny-l-yl, di- Cyclopentadienyl-Ti-bis-2,3,5,6-tetrafluoropheny-1-yl, di-cyclopentadienyl-Ti-bis-2,3,4,5,6-pentafu Orophen-1-yl, di-methylcyclopentadienyl-Ti-bis-2,6-difluoropheny-1-yl, di-methylcyclopent
- hexaarylbiimidazole compounds include the respective specifications of Japanese Patent Publication No. 6-29285, U.S. Pat. Nos. 3,479,185, 4,311,783, and 4,622,286. And various compounds described in the above, specifically, 2,2'-bis (o-chlorophenyl) -4,4 ', 5,5'-tetraphenylbiimidazole, 2,2'-bis (o-bromophenyl) )) 4,4 ′, 5,5′-tetraphenylbiimidazole, 2,2′-bis (o, p-dichlorophenyl) -4,4 ′, 5,5′-tetraphenylbiimidazole, 2,2 ′ -Bis (o-chlorophenyl) -4,4 ', 5,5'-tetra (m-methoxyphenyl) biidazole, 2,2'-bis (o, o'-dichlorophen
- organic borate compounds for example, JP-A-62-143044, JP-A-62-150242, JP-A-9-188685, JP-A-9-188686, JP-A-9-188710 and JP-A 2000 No. 2002-107916, Japanese Patent No. 2764769, Japanese Patent Application No. 2000-310808, etc., and Kunz, Martin "Rad Tech'98. Proceeding April 19-22, 1998, Chicago", etc.
- Examples of the disulfone compound include compounds described in JP-A-61-166544, JP-A-2001-132318, and the like.
- onium salt compounds examples include S.I. I. Schlesinger, Photogr. Sci. Eng. , 18, 387 (1974), T.S. S. Diazonium salts described in Bal et al, Polymer, 21, 423 (1980), ammonium salts described in U.S. Pat. No. 4,069,055, JP-A-4-365049, etc., U.S. Pat. No. 4,069, The phosphonium salt described in each specification of 055 and 4,069,056, each specification of European Patent No. 104,143, US Patent Nos. 339,049 and 410,201, Japanese Patent Laid-Open No. 2 And iodonium salts described in JP-A-150848 and JP-A-2-296514.
- the iodonium salts that can be suitably used in the present invention are diaryliodonium salts, and from the viewpoint of stability, it is preferable that two or more of them be substituted with an electron donating group such as an alkyl group, an alkoxy group or an aryloxy group. .
- one substituent of the triarylsulfonium salt has a coumarin, an iodonium salt having an antoaquinone structure, and having absorption at 300 nm or more.
- the sulfonium salts described in each specification of Patent Nos. 2,904,626, 3,604,580, and 3,604,581 can be mentioned, and in view of stability sensitivity point, electron-withdrawing groups are preferred. Preferably it is substituted.
- the electron withdrawing group preferably has a Hammett value greater than 0.
- a halogen atom, a carboxylic acid and the like can be mentioned.
- Other preferable sulfonium salts include sulfonium salts in which one substituent of the triarylsulfonium salt has a coumarin or anthraquinone structure and absorbs at 300 nm or more.
- Another preferable sulfonium salt is a sulfonium salt having absorption at 300 nm or more, in which the triarylsulfonium salt has an aryloxy group or arylthio group as a substituent.
- onium salt compound J.I. V. Crivello et al, Macromolecules, 10 (6), 1307 (1977), J. Org. V. Crivello et al, J. Mol. Polymer Sci. , Polymer Chem. Ed. , Selenonium salts described in C., 17, 1047 (1979), C.I. S. Wen et al, Teh, Proc. Conf. Rad. Curing ASIA, p 478 Tokyo, Oct (1988), and onium salts such as arsonium salts.
- acyl phosphine (oxide) compound examples include Irgacure 819, Darocure 4265, Darocure TPO and the like manufactured by Ciba Specialty Chemicals.
- photopolymerization initiators used in the present invention include the purpose of use of the colored curable composition of the present invention, and the relationship with the (a-1) pigment and (C) polymerizable compound used.
- one or two or more suitable ones may be selected and used, but from the viewpoint of exposure sensitivity, trihalomethyl triazine compounds, benzyl dimethyl ketal compounds, ⁇ -hydroxy ketone compounds, ⁇ -amino ketone compounds, acyl phosphines Compounds, phosphine oxide compounds, metallocene compounds, triallylimidazole dimers, onium compounds, benzothiazole compounds, benzophenone compounds, acetophenone compounds and their derivatives, cyclopentadiene-benzene-iron complexes and their salts, halomethyl Oxadiazole compounds, 3-aryl substituted coumarin compounds Preferred are compounds selected from the group consisting of
- the content of the other photopolymerization initiator (B-2) which can be contained in the colored curable composition of the present invention is preferably 0 to 20% by mass with respect to the total solid content of the curable composition. More preferably, it is 0 to 15% by mass, still more preferably 0 to 10% by mass.
- a compound having an adhesive group can be used for the purpose of improving the adhesion of the cured film to the support or the substrate.
- a compound having an adhesive group is preferably used from the viewpoint of improving the adhesion between the colored region and the substrate.
- the adhesive group contained in such a compound may be a substituent having adhesiveness to a substrate used for a color filter, but a compound having adsorptivity to a glass substrate or a silicon substrate is particularly preferable.
- Preferred adhesion groups are acid groups such as sulfonic acid groups, phosphonic acid groups and carboxyl groups; ester groups of these acids; metal salts of these acids; onium salts of these metals; onium groups such as ammonium groups and pyridinium groups;
- the substituent is preferably a group selected from a substituent capable of generating a silanol group by hydrolysis such as a silyl group; a zwitterionic group such as a phenolic hydroxyl group and an N-oxide group; and a chelating group such as iminodiacetic acid. From the viewpoint of adhesion, an alkoxysilyl group and / or a hydrolyzed group thereof is preferable.
- the compound having an adhesive group in the present invention optionally uses a compound having a group capable of improving the developability, a carbon-carbon double bond group, a cyclic alkoxy group, etc. in the molecule. It is possible.
- a group for improving developability functional groups having a hetero atom are preferable, and alkoxy group, alkoxycarbonyl group, carboxyl group, hydroxyl group, amino group, urethane group, amido group, thiol group, sulfo group, urea group and the like can be mentioned. It is preferable to use a compound having such a functional group in combination.
- Preferred examples of the group for improving sensitivity include methacryl group, acryl group, styryl group, vinyl ether group, allyl group, cyclic alkenyl group, furyl group, oxetane group, epoxy group and tetrahydrofuran group, and methacryl group from the viewpoint of sensitivity. It is preferable to use a compound having an acryl group or a styryl group in combination.
- the curable composition of the present invention may contain a sensitizer for the purpose of improving the radical generation efficiency of the radical initiator and lengthening the photosensitive wavelength.
- a sensitizer which can be used for this invention what sensitizes the above-mentioned photoinitiator by an electron transfer mechanism or an energy transfer mechanism is preferable.
- the sensitizers that can be used in the present invention include those belonging to the compounds listed below and having an absorption wavelength in the wavelength range of 300 nm to 450 nm.
- Examples of preferable sensitizers include those belonging to the following compounds and having an absorption wavelength in the range of 330 nm to 450 nm.
- polynuclear aromatics eg, phenanthrene, anthracene, pyrene, perylene, triphenylene, 9, 10-dialkoxyanthracene
- xanthenes eg, fluorescein, eosin, erythrosine, rhodamine B, rose bengal
- thioxanthones Isopropylthioxanthone, diethylthioxanthone, chlorothioxanthone
- cyanines eg, thiacarbocyanine, oxacarbocyanine
- merocyanines eg, merocyanine, carbomerocyanine
- phthalocyanines thiazines (eg, thionine, methylene blue, toluidine blue)
- Acridines eg, acridine orange, chloroflavin, acriflavine
- anthraquinones eg, anthra
- Examples of more preferable sensitizers include compounds represented by the following formulas (i) to (v).
- a 1 represents a sulfur atom or NR 50
- R 50 represents an alkyl group or an aryl group
- L 2 cooperates with adjacent A 1 and adjacent carbon atoms to form a basic nucleus of the dye
- R 51 and R 52 each independently represent a hydrogen atom or a monovalent nonmetal atomic group
- R 51 and R 52 are mutually bonded to form an acidic nucleus of the dye.
- W may represent an oxygen atom or a sulfur atom.
- Ar 1 and Ar 2 each independently represent an aryl group, and they are linked via a bond of -L 3- , wherein L 3 represents -O- or -S-. Moreover, W is synonymous with what was shown to general formula (i).)
- a 2 represents a sulfur atom or NR 59
- L 4 represents a nonmetallic atomic group forming a basic nucleus of a dye in cooperation with adjacent A 2 and carbon atom
- R 53 , R 54 , R 55 , R 56 , R 57 and R 58 each independently represent a monovalent nonmetallic atomic group group
- R 59 represents an alkyl group or an aryl group.
- a 3, A 4 each independently -S- or -NR 62 - or -NR 63 - and represents, R 63, R 64 each independently represent a substituted or unsubstituted alkyl group, a substituted L 5 and L 6 each independently represent a nonmetallic atomic group forming a basic nucleus of a dye in combination with adjacent A 3 , A 4 and adjacent carbon atoms, and R 5 and L 6 each independently represent an aryl group.
- 60 and R 61 each independently represent a monovalent nonmetallic radical or may be bonded to each other to form an aliphatic or aromatic ring).
- R 66 represents an aromatic ring or a hetero ring which may have a substituent
- a 5 represents an oxygen atom, a sulfur atom or —NR 67 — R 64 , R 65 and R 67 each independently represents a hydrogen atom or a monovalent nonmetallic atomic group, and R 67 and R 64 , and R 65 and R 67 may be bonded to each other to form an aliphatic or aromatic ring, respectively it can.
- the sensitizers may be used alone or in combination of two or more.
- the content of the sensitizer in the curable composition of the present invention is preferably 0.1 to 20% by mass in terms of solid content from the viewpoint of light absorption efficiency to the deep part and initiation decomposition efficiency, 5 to 15% by mass is more preferable.
- the curable composition of the present invention preferably also contains a co-sensitizer.
- the co-sensitizer has an action of further improving the sensitivity of the sensitizing dye or the initiator to the actinic radiation, or suppressing the inhibition of the polymerization of the polymerizable compound by oxygen.
- co-sensitizers include amines, such as M.I. R. Sander et al., "Journal of Polymer Society", 10, 3173 (1972), JP-B-44-20189, JP-A-51-82102, JP-A-52-134692, JP-A-59-138205.
- co-sensitizers include thiols and sulfides, such as thiol compounds described in JP-A-53-702, JP-B-55-500806, JP-A-5-142772, And the like. Specific examples thereof include 2-mercaptobenzothiazole, 2-mercaptobenzoxazole, 2-mercaptobenzoimidazole, 2-mercapto-4 (3H) -quinazoline and ⁇ -mercapto Naphthalene etc. are mentioned.
- an amino acid compound eg, N-phenylglycine etc.
- an organic metal compound eg, tributyltin acetate etc.
- hydrogen donors described in 34414
- sulfur compounds described in JP-A No. 6-308727 (eg, trithian etc.).
- the content of these co-sensitizers is in the range of 0.1 to 30% by mass with respect to the mass of the total solid content of the curable composition from the viewpoint of improving the curing rate by the balance between the polymerization growth rate and the chain transfer.
- the range of 1 to 25% by mass is more preferable, and the range of 0.5 to 20% by mass is more preferable.
- a small amount of a thermal polymerization inhibitor may be added to prevent unnecessary thermal polymerization of a compound having a polymerizable ethylenic unsaturated double bond during production or storage of a curable composition. desirable.
- thermal polymerization inhibitor which can be used in the present invention, hydroquinone, p-methoxyphenol, di-t-butyl-p-cresol, pyrogallol, t-butyl catechol, benzoquinone, 4,4'-thiobis (3-methyl-6) And -t-butylphenol), 2,2'-methylenebis (4-methyl-6-t-butylphenol), N-nitrosophenylhydroxyamine cerous salt and the like.
- the addition amount of the thermal polymerization inhibitor is preferably about 0.01% by mass to about 5% by mass with respect to the mass of the entire composition.
- higher fatty acid derivatives such as behenic acid and behenic acid amide may be added to cause uneven distribution on the surface of the photosensitive layer in the process of drying after coating.
- the amount of the higher fatty acid derivative added is preferably about 0.5% by mass to about 10% by mass of the total composition.
- Thermally polymerized component It is also effective to contain a thermal polymerization component in the colored curable composition of the present invention.
- an epoxy compound can be added to increase the strength of the coating.
- the epoxy compound is a compound having two or more epoxy rings in the molecule, such as bisphenol A type, cresol novolac type, biphenyl type and alicyclic epoxy compounds.
- bisphenol A type Epototh YD-115, YD-118T, YD-127, YD-128, YD-134, YD-8125, YD-7011R, ZX-1059, YDF-8170, YDF-170, etc.
- alicyclic epoxy compounds such as 3,5,3 ', 5'-tetramethyl-4,4'-diglycidylbiphenyl include: celoxide 2021, 2081, 2083, 2085, epolide GT-301, GT-302, GT- 401, GT-403, EHPE-3150 (manufactured by Daicel Chemical Industries, Ltd.), Suntote ST-3000, ST-4000, ST-5080, ST-5100, etc. (manufactured by Toto Kasei Co., Ltd.), and the like.
- 1,1,2,2-tetrakis p-glycidyloxyphenyl) ethane, tris (p-glycidyloxyphenyl) methane, triglycidyl tris (hydroxyethyl) isocyanurate, o-phthalic acid diglycidyl ester, terephthalic acid di-ester
- glycidyl esters, epototh YH-434 and YH-434L which are amine type epoxy resins, and glycidyl esters in which a dimer acid is modified in the skeleton of bisphenol A type epoxy resin can also be used.
- the colored curable composition of the present invention is preferably composed of various surfactants, from the viewpoint of improving coatability, and various surfactants of fluorine type surfactants, nonionic type, cationic type and anionic type are preferable. Agents can be used. Among them, fluorine surfactants and nonionic surfactants are preferable.
- a fluorosurfactant a compound having a fluoroalkyl or fluoroalkylene group at least at any one of the terminal, main chain and side chain can be suitably used.
- Specific commercial products include, for example, Megafac F142D, F172, F173, F176, F177, F183, 780, 781, R30, R08, F-472SF, BL20, and the like.
- R-61, R-90 (Dainippon Ink and Chemicals, Inc.), Florard FC-135, FC-170C, FC-430, FC-431, Novec FC-4430 (Sumitomo 3M) Asahi Guard AG 7105, 7000, 950, 7600, Surfron S-112, S-113, S-131, S-141, S-145, S-382, SC-101, SC-102 , SC-103, SC-104, SC-105, SC-106 (manufactured by Asahi Glass Co., Ltd.), F-Top EF 351, 352, 801, 802 (manufactured by JEMCO Co., Ltd.) and the like.
- the fluorine-based surfactant is particularly effective in preventing coating unevenness and thickness unevenness when forming a thin coating film using the colored curable composition of the present invention. Furthermore, it is also effective when applying the colored curable composition of the present invention to slit coating in which liquid breakage easily occurs.
- nonionic surfactants include nonionic surfactants such as polyoxyethylene alkyl ethers, polyoxyethylene alkyl aryl ethers, polyoxyethylene alkyl esters, sorbitan alkyl esters, and monoglyceride alkyl esters. Particularly preferred.
- polyoxyethylene alkyl ethers such as polyoxyethylene lauryl ether, polyoxyethylene stearyl ether, and polyoxyethylene oleyl ether; polyoxyethylene octyl phenyl ether, polyoxyethylene polysylated ether, polyoxyethylene trilyl ether
- Polyoxyethylene aryl ethers such as benzyl phenyl ether, polyoxyethylene-propylene polystyryl ether, polyoxyethylene nonyl phenyl ether
- polyoxyethylene dialkyl esters such as polyoxyethylene dilaurate, polyoxyethylene distearate, sorbitan fatty acid ester , Polyoxyethylene sorbitan fatty acid esters, ethylenediamine polyoxyethylene-polyoxypropyl ester
- Nonionic surfactants such as quinone condensates, which are commercially available from Kao Co., Ltd., Nippon Oil & Fats Co., Ltd., Takemoto Oil & Fat Co., Ltd.
- additives can be used in the colored curable composition of the present invention.
- the additive include UV absorbers such as 2- (3-t-butyl-5-methyl-2-hydroxyphenyl) -5-chlorobenzotriazole and alkoxybenzophenone, and anticoagulation agents such as sodium polyacrylate , Glass, fillers such as alumina; itaconic acid copolymer, crotonic acid copolymer, maleic acid copolymer, partially esterified maleic acid copolymer, acidic cellulose derivative, addition of acid anhydride to polymer having hydroxyl group And alkali-soluble resins such as phenoxy resins formed from bisphenol A and epichlorohydrin.
- an organic carboxylic acid preferably having a molecular weight of 1000 or less, is used in preparation of the pigment dispersion (A) for the purpose of promoting the alkali solubility of the uncured portion and further improving the developability of the colored curable composition.
- the addition of low molecular weight organic carboxylic acids can be carried out.
- aliphatic monocarboxylic acids such as formic acid, acetic acid, propionic acid, butyric acid, valeric acid, pivalic acid, caproic acid, diethylacetic acid, enanthate and caprylic acid; oxalic acid, malonic acid, succinic acid, Aliphatic dicarboxylic acids such as glutaric acid, adipic acid, pimelic acid, suberic acid, azelaic acid, sebacic acid, brassic acid, methylmalonic acid, ethylmalonic acid, dimethylmalonic acid, methylsuccinic acid, tetramethylsuccinic acid, citraconic acid and the like; Aliphatic tricarboxylic acids such as tricarballylic acid, aconitic acid, and camphoric acid; aromatic monocarboxylic acids such as benzoic acid, toluic acid, cuminic acid, hemellitic acid, mesitylene acid; phthalic acid,
- known additives such as an inorganic filler, a plasticizer, and a sensitizing agent capable of improving the ink receptivity on the surface of the photosensitive layer may be added.
- the plasticizer include dioctyl phthalate, didodecyl phthalate, triethylene glycol dicaprylate, dimethyl glycol phthalate, tricresyl phosphate, dioctyl adipate, dibutyl sebacate, triacetyl glycerin and the like, and when a binder is used, ethylene is used. 10 mass% or less can be added with respect to the total mass of the compound which has a sex unsaturated double bond, and a binder.
- the colored curable composition of the present invention cures with high sensitivity and also has good storage stability. It also exhibits high adhesion to the surface of hard materials such as substrates to which the curable composition is applied. Therefore, the curable composition of the present invention can be preferably used in the fields of imaging materials such as three-dimensional stereolithography and holography, color filters, inks, paints, adhesives, and coating agents.
- the colored curable composition of the present invention comprises the (A) pigment dispersion described above (B) an oxime ester initiator, (C) a polymerizable compound, and an alkali-soluble resin optionally used in combination (preferably It can be prepared by passing through a mixing and dispersing step of mixing with a solvent, adding additives such as surfactant if necessary, and mixing and dispersing them using various mixers and dispersers.
- the mixing and dispersing step preferably includes kneading and dispersing followed by fine dispersion treatment, but kneading and dispersing may be omitted.
- a pigment dispersion (A-1) A pigment, (a-3) a specific dispersion resin, (a-2) a specific pigment derivative, and a solvent are blended to carry out bead dispersion. Finely dispersed with beads made of glass with a particle diameter of 0.01 to 1 mm, zirconia, etc. mainly using a vertical or horizontal sand grinder, pin mill, slit mill, ultrasonic dispersion machine, etc. (A) pigment A dispersion is obtained. It is also possible to dispense with the process of refining the pigment. The details of kneading and dispersion are described in T.K. C.
- a step of applying the colored curable composition of the present invention obtained as described above onto a support to form a curable composition layer (hereinafter referred to simply as “curable composition layer forming step” as appropriate) And exposing the curable composition layer through a mask (hereinafter referred to as “exposure step” as appropriate) and developing the curable composition layer after exposure to form a colored region. And forming a step (hereinafter, appropriately abbreviated as a “development step”).
- a step hereinafter, appropriately abbreviated as a “development step”.
- the color filter of the present invention is formed on a substrate such as glass using the colored curable composition of the present invention described above, and the colored curable composition of the present invention is directly or via another layer.
- a coating film is formed on the substrate by, for example, slit coating, the coating film is dried, exposed to a pattern, and developed preferably by sequentially performing development processing using a developer.
- the colored curable composition of the present invention can be applied to a substrate directly or through another layer by a coating method such as spin coating, slit coating, cast coating, roll coating, bar coating and the like.
- a coating method such as spin coating, slit coating, cast coating, roll coating, bar coating and the like.
- the substrate for example, non-alkali glass used for liquid crystal display elements, soda glass, Pyrex (registered trademark) glass, quartz glass, and those obtained by attaching a transparent conductive film to these, solid imaging elements, etc.
- Photoelectric conversion element substrates such as silicon substrates, and plastic substrates. On these substrates, usually, a black matrix for separating each pixel is formed, or a transparent resin layer is provided for promoting adhesion and the like.
- the plastic substrate preferably has a gas barrier layer and / or a solvent resistant layer on its surface.
- the coloring curability of the present invention is also applied to a driving substrate (hereinafter referred to as "TFT liquid crystal driving substrate") on which thin film transistors (TFT) of a thin film transistor (TFT) type color liquid crystal display device are disposed.
- TFT liquid crystal driving substrate on which thin film transistors (TFT) of a thin film transistor (TFT) type color liquid crystal display device are disposed.
- a patterned film made of the composition can be formed to make a color filter.
- the photomask used at that time is also provided with a pattern for forming a through hole or a U-shaped depression.
- the substrate in the TFT type liquid crystal driving substrate include glass, silicon, polycarbonate, polyester, aromatic polyamide, polyamide imide, polyimide and the like.
- These substrates may be subjected to appropriate pretreatment such as chemical treatment with a silane coupling agent, plasma treatment, ion plating, sputtering, gas phase reaction method, vacuum deposition and the like, if desired.
- the method for applying the colored curable composition of the present invention to a substrate is not particularly limited, but a method using a slit nozzle such as a slit and spin method or a spinless coating method (hereinafter referred to as a slit nozzle coating method) Is preferred.
- a slit nozzle coating method although the conditions differ depending on the size of the coated substrate, the slit and spin coating method and the spinless coating method apply the fifth generation glass substrate (1100 mm ⁇ 1250 mm) by the spinless coating method, for example.
- the discharge rate of the photocurable composition from the slit nozzle is usually 500 to 2000 microliters / second, preferably 800 to 1500 microliters / second, and the coating speed is usually 50 to 300 mm / second.
- the thickness of the coating film is generally 0.3 to 5.0 ⁇ m, preferably 0.5 to 4 .0 ⁇ m, most preferably 0.8 to 3.0 ⁇ m.
- the film thickness is most preferably in the range of 0.4 ⁇ m to 2.0 ⁇ m.
- the present invention is particularly effective in forming a colored region having a thickness of 0.4 ⁇ m to 1.0 ⁇ m, and further, 0.45 ⁇ m to 0.8 ⁇ m.
- pre-baking treatment is performed after coating.
- vacuum treatment can be performed before prebaking.
- the degree of vacuum is usually about 0.1 to 1.0 torr, preferably about 0.2 to 0.5 torr.
- the pre-baking treatment can be performed using a hot plate, an oven, or the like at a temperature range of 50 to 140 ° C., preferably about 70 to 110 ° C., and under conditions of 10 to 300 seconds.
- High frequency processing may be used in combination. The high frequency processing can be used alone.
- the curable composition layer formed in the curable composition layer forming step is exposed through a mask having a predetermined mask pattern.
- the pattern exposure of the coating film in this process is performed by exposing through a predetermined mask pattern, curing only the coating film portion irradiated with light, and developing with a developer, and from the pixels of each color (three or four colors) It can carry out by forming the following pattern-like film.
- ultraviolet rays such as g-line and i-line are preferably used as radiation which can be used for exposure. Irradiation amount is more preferably 5 ⁇ 1500mJ / cm 2 is preferably 10 ⁇ 1000mJ / cm 2, and most preferably 10 ⁇ 500mJ / cm 2.
- the color filter of the present invention is used for a liquid crystal display device
- 5 to 200 mJ / cm 2 is preferable within the above range
- 10 to 150 mJ / cm 2 is more preferable
- 10 to 100 mJ / cm 2 is most preferable.
- more preferably 30 ⁇ 1500mJ / cm 2 is preferably 50 ⁇ 1000mJ / cm 2 within the above range, most preferably 80 ⁇ 500mJ / cm 2 .
- ⁇ Development process> In the development processing, the uncured part after exposure is dissolved in a developer and only the cured part remains.
- the development temperature is usually 20 to 30 ° C., and the development time is 20 to 90 seconds.
- dissolving the coating film of the photocurable photocurable composition in an unhardened part any thing can be used if it does not melt
- Examples of the organic solvent include the solvents described above which can be used when preparing the pigment dispersion composition or the photocurable composition of the present invention.
- Examples of the alkaline aqueous solution include sodium hydroxide, potassium hydroxide, sodium carbonate, sodium hydrogencarbonate, sodium borate, sodium metaborate, aqueous ammonia, ethylamine, diethylamine, dimethylethanolamine, tetramethylammonium hydroxide, Alkaline compounds such as tetraethylammonium hydroxide, choline, pyrrole, piperidine and 1,8-diazabicyclo- [5,4,0] -7-undecene are used at a concentration of 0.001 to 10% by mass, preferably 0.01 to 10% by mass.
- dissolved so that it may be 1 mass% is mentioned.
- An appropriate amount of a water-soluble organic solvent such as methanol or ethanol, a surfactant or the like can be added to the alkaline aqueous solution.
- the development method may be any of a dip method, a shower method, a spray method and the like, and may be combined with a swing method, a spin method, an ultrasonic method and the like. Before the developer is touched, the surface to be developed may be moistened with water or the like in advance to prevent uneven development. Also, the substrate can be inclined and developed. In the case of producing a color filter for a solid-state imaging device, paddle development may be used.
- the rinse process is usually performed with pure water, but pure water is used in the final washing, used pure water is used at the beginning of washing, the substrate is tilted and washed, and ultrasonic irradiation is used to save liquid. Can be used together.
- heat treatment is usually carried out at about 200.degree. C. to 250.degree.
- the heat treatment (post bake) is continuous or batchwise using a heating means such as a hot plate, convection oven (hot air circulation dryer), high frequency heater or the like so that the coated film after development is under the above conditions. It can be done by the formula.
- the application to the color filter has mainly been described, but the formation of a black matrix separating the colored pixels constituting the color filter Can also be applied.
- the colored curable composition of the present invention using a black pigment such as carbon black or titanium black as a pigment is exposed and developed, and then post-baked as required to accelerate the curing of the film. It can be formed by
- the colored curable composition of the present invention is excellent in pigment dispersion stability, is cured with high sensitivity, and is excellent in developability, so it is a thin layer even when it contains a high concentration of pigment.
- a colored region excellent in color characteristics can be formed with high resolution, and in particular, it is useful for forming a colored region of a color filter, and its application range is wide.
- Synthesis Example 1 Preparation of Specified Dispersion Resin Solution 1 800 parts of cyclohexanone is put in a reaction vessel, and heated to 100 ° C. while injecting nitrogen gas into the vessel, and at the same temperature, 60.0 parts of styrene, 80.0 parts of methacrylic acid, 68.0 parts of methyl methacrylate, butyl methacrylate A mixture of 62.0 parts and 9.0 parts of azobisisobutyronitrile was added dropwise over 1 hour to carry out a polymerization reaction. After dropping, the reaction is further allowed to react at 100 ° C.
- Synthesis Example 2 Preparation of a Specific Dispersion Resin Solution 2 10 parts by mass of glycidyl methacrylate and 1 part by mass of tetraethylammonium bromide were added to the specific dispersion resin solution 1 obtained in the above Synthesis Example 1, and the mixture was stirred at 90 ° C. for 2 hours to introduce a double bondable group in the side chain A solution of specific dispersion resin 2 having a molecular weight of about 32,000 and an acid value of 3.1 mmol / g was obtained. Cyclohexanone (solvent) was added to the solution of the specific dispersion resin 2 to prepare a specific dispersion resin solution 2 having a nonvolatile content of 20% by mass.
- Synthesis Example 3 Preparation of a Specific Dispersion Resin Solution 3 50 parts by weight of glycidyl methacrylate in 333 parts by weight of a propylene glycol monomethyl ether acetate solution containing 100 parts by weight of polyacrylic acid / methyl methacrylate (weight ratio 50/50) having a weight average molecular weight of 10,000 and an acid value of 6.9 mmol / g 1 part by mass of tetraethylammonium bromide was added, and the mixture was stirred at 90 ° C. for 3 hours, and a double bondable group was introduced into the side chain, having a weight average molecular weight of 12,000 and an acid value of 1.95 mmol / g A solution was obtained. Propylene glycol monomethyl ether acetate (solvent) was added to the solution of the specific dispersion resin 3 to prepare a specific dispersion resin solution 3 having a nonvolatile content of 20% by mass.
- reaction solution was cooled to room temperature and then poured into 7 L of water to precipitate a polymer compound.
- the precipitated polymer compound was collected by filtration, washed with water and dried to obtain 131 g of a polymer compound.
- the weight average molecular weight of the obtained polymer compound was measured by gel permeation chromatography (GPC) using polystyrene as a standard substance, and as a result, it was 11,800. Further, the acid value of this polymer compound was determined by titration, and it was 69.6 mg KOH / g (calculated value 69.7 mg KOH / g).
- the precipitated specific dispersion resin 4 was collected by filtration, washed with water and dried to obtain 105 g of the intended specific dispersion resin 4.
- the specific dispersion resin 4 was dissolved in propylene glycol monomethyl ether to obtain a specific dispersion resin solution 4 (20% solid solution).
- reaction solution was cooled to room temperature and then poured into 8 L of water (liter; the same in the following) to precipitate a polymer compound.
- the precipitated polymer compound was collected by filtration, washed with water and dried to obtain 114 g of a polymer compound (ii-1).
- the weight average molecular weight of the obtained polymer compound (ii-1) was measured by gel permeation chromatography (GPC) using polystyrene as a standard substance, and as a result, it was 12,000.
- this polymer compound (ii-1) was determined by titration, and it was 258 mg KOH / g (calculated value 260 mg KOH / g), and it was confirmed that the polymerization was carried out normally.
- the precipitated polymer compound was collected by filtration, washed with water and dried to obtain 54 g of a polymer compound (ii-2).
- the weight average molecular weight of the obtained polymer compound (ii-2) was measured to be 17,800 by gel permeation chromatography (GPC) using polystyrene as a standard substance.
- the acid value of the polymer compound (ii-2) was determined by titration and found to be 128 mg KOH / g (calculated value 129 mg KOH / g).
- the precipitated specific dispersion resin 5 was collected by filtration, washed with water and dried to obtain 54 g of the intended specific dispersion resin 5.
- the weight average molecular weight of the specific dispersion resin 5 was measured to be 20, 100 by gel permeation chromatography (GPC) using polystyrene as a standard substance. Furthermore, the acid value of this polymer compound was determined by titration, and it was 115 mg KOH / g (calculated value 117 mg KOH / g).
- the specific dispersion resin 5 was dissolved in propylene glycol monomethyl ether to obtain a specific dispersion resin solution 5 (20% solid solution).
- Synthesis Example 7 Preparation of Specific Dispersion Resin 7 5.9 g (0.04 mol) of 2,2-bis (hydroxymethyl) butanoic acid and 15.9 g (0.06 mol) of the following diol compound (2) in a 500 ml three-necked round bottom flask equipped with a condenser and a stirrer Mole) was dissolved in 100 ml of N, N-dimethylacetamide. To this, 20.4 g (0.082 mol) of 4,4-diphenylmethane diisocyanate, 3.4 g (0.02 mol) of 1,6-hexamethylene diisocyanate, and 0.3 g of dibutyltin dilaurate are added.
- the mixture was heated and stirred for 8 hours. Thereafter, it was diluted with 100 ml of N, N-dimethylformamide and 200 ml of methyl alcohol and stirred for 30 minutes.
- the reaction solution was poured into 3 liters of water with stirring to precipitate a white specific dispersion resin 7.
- the specific dispersion resin 7 was separated by filtration, washed with water and dried under vacuum to obtain 34 g of the specific dispersion resin 7.
- the molecular weight was measured by gel permeation chromatography (GPC), and the weight average molecular weight (polystyrene standard) was 50,000 acid number 0.88 mmol / g.
- Comparative resin 1 A resin having a weight average molecular weight of 12,000 and an acid value of 2.06 mmol / g obtained by radical polymerization according to a known method at a molar ratio of methyl methacrylate and methacrylic acid of 80/20 was used as Comparative Resin 1.
- Comparative resin 2 A resin having a weight average molecular weight of 30,000 and an acid value of 2.85 mmol / g obtained by radical polymerization according to a known method at a molar ratio of 60/40 of benzyl methacrylate and methacrylic acid was used as Comparative Resin 2.
- composition (1) The components of the following composition (1) are mixed, and the number of revolutions is 3,000 r.p.m. p. m. The mixture was stirred for 3 hours and mixed to prepare a mixed solution (AD, H-1 to H-2) containing a pigment.
- Composition (1) C. I. Pigment red 254 80 parts C.I. I. Pigment yellow 139 20 parts ⁇ specific dispersion resin or resin for comparison (described in Table 1 below) 275 parts ⁇ 1-methoxy-2-propyl acetate 630 parts ⁇ specific pigment derivative or comparative compound (described in Table 1 below) 8 parts
- the mixed solution (A to D, H-1 to H-2) obtained above is further dispersed for 6 hours with a bead disperser Dispermat (manufactured by GETZMANN) using 0.3 mm ⁇ zirconia beads.
- a vacuum mechanism-equipped high-pressure dispersing machine NANO-3000-10 manufactured by Nippon Biii Co.
- This dispersion process was repeated 10 times to obtain red pigment dispersions (A to D, H-1 to H-2).
- ⁇ Composition (2)> -19.20 parts of propylene glycol monomethyl ether acetate (PGMEA: solvent) -Ethyl lactate 36.67 parts-Binder polymer 30.51 parts [40% PGMEA solution of benzyl methacrylate / methacrylic acid / methacrylic acid-2-hydroxy ethyl methacrylate copolymer (molar ratio 60/22/18)] -Dipentaerythritol hexaacrylate 12.20 parts (photopolymerizable compound) Polymerization inhibitor (p-methoxyphenol) 0.0061 parts Fluorosurfactant 0.83 parts (F-475, manufactured by Dainippon Ink and Chemicals, Inc.) -0.586 parts of a photopolymerization initiator (TAZ-107: a trihalomethyl triazine based photopolymerization initiator, manufactured by Midori Kagaku Co., Ltd.)
- a 6 inch silicon wafer was heat treated in an oven at 200 ° C. for 30 minutes.
- the resist solution is coated on this silicon wafer to a dry film thickness of 2 ⁇ m, and further dried by heating in an oven at 220 ° C. for 1 hour to form an undercoat layer to obtain a silicon wafer substrate with an undercoat layer.
- Pigment dispersion 100 parts by mass DPHA (manufactured by Nippon Kayaku Co., Ltd.) 3.5 parts by mass Polymerization initiator [Oxime ester initiator or comparative initiator described in Table 2] 0.6 parts by mass of allyl methacrylate / methacrylic acid [molar ratio, 73/27, Weight average molecular weight 57,000] 1.0 parts by mass Surfactant (trade name: Tetranic 150R1, BASF Corporation) 0.2 parts by mass Solvent: PGMEA 100 parts by mass
- the exposure was performed at various exposure amounts of 50 to 1600 mJ / cm 2 so as to obtain an Island pattern.
- 50 mJ / cm 2 exposure was measured by a light illuminance system, and the exposure time was changed with reference to the exposure, and the integrated exposure (mJ / cm 2 ) was determined as sensitivity.
- the numerical value is smaller, it is possible to form a fine pattern of high resolution with a low exposure amount, which indicates high sensitivity.
- Table 2 The results are shown in Table 2 above.
- the silicon wafer substrate on which the irradiated coating film is formed is placed on the horizontal rotary table of a spin shower developing machine (DW-30 type, manufactured by Chemitronics Co., Ltd.), and CD-2000 (Fuji Film) Paddling development was performed at 23 ° C. for 60 seconds using Electronic Materials (manufactured by Shin-Etsu Materials Co., Ltd.) to form a colored pattern on a silicon wafer substrate.
- DW-30 type manufactured by Chemitronics Co., Ltd.
- CD-2000 Fluji Film
- the silicon wafer substrate on which a coloring pattern is formed is fixed to the horizontal rotary table by a vacuum chuck method, and the silicon wafer substrate is rotated at a number of revolutions of 50 r. p. m. While rotating at the same time, pure water was supplied in the form of a shower from a jet nozzle from above the rotation center to perform a rinse process, and then spray drying was performed. Thereafter, using a length measurement SEM “S-9260A” (manufactured by Hitachi High-Technologies Corp.), the formed 1.4 ⁇ m square colored pattern and its periphery were observed at a magnification of 20,000. The presence or absence of the residue in the area (unexposed area) where light was not irradiated in the exposure step was observed to evaluate the developability. The results are shown in Table 2 above.
- composition (3) C. I. Pigment green 36 50 parts by mass C.I. I. Pigment yellow 150 50 parts by mass specific dispersion resin or resin for comparison (described in Table 3 below) 200 parts by mass Solsperse 32000 GR (manufactured by Nippon Lubrizol; polyester based dispersant) 20 parts by mass Solvent: 520 parts by mass propylene glycol methyl ether acetate Specific pigment derivative or comparative compound (described in Table 3 below) 5 parts by mass
- the oxime type initiators (I-1) and (I-2) described in Table 4 above are the compounds described above.
- the structures of (B) the oxime type initiator (I-3) and the comparative polymerization initiator (IH-2) are shown below.
- the colorant-containing curable compositions of Examples 6 to 9 and Comparative Example 3 prepared as described above were coated on a 6-inch silicon substrate to form a curable composition layer (coated film). Then, heat treatment (pre-baking) was performed for 120 seconds using a hot plate at 100 ° C. so that the dry thickness of the coating film was 0.8 ⁇ m. Next, an i-line stepper exposure apparatus FPA-3000i5 + (Canon Co., Ltd.) is used to expose a 1.4 ⁇ m square island pattern at a wavelength of 365 nm and a 1.4 ⁇ m square pattern of the same size as the mask pattern. The exposure was performed at various exposure amounts of 50 to 1600 mJ / cm 2 so as to obtain an Island pattern.
- FPA-3000i5 + Canon Co., Ltd.
- the silicon wafer substrate on which a coloring pattern is formed is fixed to the horizontal rotary table by a vacuum chuck method, and the silicon wafer substrate is rotated at a number of revolutions of 50 r. p. m. While rotating at the same time, pure water was supplied in the form of a shower from a jet nozzle from above the rotation center to perform a rinse process, and then spray drying was performed. Thereafter, using a length measurement SEM “S-9260A” (manufactured by Hitachi High-Technologies Corp.), the formed 1.4 ⁇ m square colored pattern and its periphery were observed at a magnification of 20,000. The presence or absence of the residue in the area (unexposed area) where light was not irradiated in the exposure step was observed to evaluate the developability. The results are shown in Table 4 above.
- Pigment red 177 50 parts ⁇ specific dispersion resin (described in Table 5 below) 200 parts ⁇ BYK 111 (manufactured by Bick Chemie; phosphate ester dispersant) 10 parts ⁇ specific pigment derivative or comparative compound (described in Table 5 below) 4 parts -660 parts of 1-methoxy-2-propyl acetate
- the mixed solution obtained above is further subjected to dispersion treatment for 6 hours with a bead disperser Dispermat (manufactured by GETZMANN Co., Ltd.) using 0.3 mm ⁇ zirconia beads, and then a high pressure disperser NANO with a pressure reducing mechanism is further added.
- Dispersion treatment was performed using a flow rate of 500 g / min under a pressure of 2000 kg / cm 3 using -3000-10 (manufactured by Nippon Bei Co., Ltd.). This dispersion process was repeated 10 times to obtain a red pigment dispersion composition (H to K, H-5 to H-6).
- the specific pigment derivatives (Y-1), (Y-2) and (Y-6) according to the present invention described in Table 5 above are the same compounds as described above.
- the structures of the specific pigment derivatives (Y-8) and (Y-9) and the comparison compound (H-3) are shown below.
- Pigment dispersion 100 parts by mass DPHA (manufactured by Nippon Kayaku Co., Ltd.) 3.8 parts by mass Polymerization initiator 1.5 parts by mass (Oxime ester initiators described in Table 6 below or Comparison initiator) -Benzyl methacrylate / methacrylic acid (mol ratio, 70/30, weight average molecular weight: 70,000) 0.7 parts by mass-Surfactant (trade name: Tetranic 150R1, BASF Corporation) 0.2 parts by mass-Solvent: PGMEA 100 parts by mass
- the oxime type initiators (I-1), (I-2), (I-3) and (IH-1) described in Table 6 above are the compounds described above.
- Curable Composition layer A curable composition containing the above-mentioned pigment is slit-coated as a resist solution on a 550 mm ⁇ 650 mm glass substrate under the following conditions, and then allowed to stand still for 10 minutes for vacuum drying and prebaking ( Prebake (100 ° C. for 80 seconds) was applied to form a curable composition coating (curable composition layer).
- Prebake 100 ° C. for 80 seconds
- Curable composition layer Curable composition layer.
- Clearance at opening of tip of coating head 50 ⁇ m Coating speed: 100 mm / sec. Clearance between substrate and coating head: 150 ⁇ m Coating thickness (dry thickness): 2 ⁇ m Coating temperature: 23 ° C
- the photocurable coating film is exposed in a pattern using a test photomask with a line width of 20 ⁇ m using a 2.5 kw ultrahigh pressure mercury lamp, and after exposure, the entire surface of the coating film is Fujifilm Electronics It was covered with a 1% aqueous solution of Alkaline developer CDK-1, manufactured by Materials Co., and rested for 40 seconds.
- CDK-1 Alkaline developer
- ⁇ Liquid crystal display element> With respect to the above curable composition, except that the pigment alone was changed to the blue pigment and the green pigment described in the specification, the green and blue colored curable compositions were similarly prepared. First, a black matrix is formed by the usual method on the above-mentioned substrate, and 20 ⁇ 20 ⁇ m red, green and blue patterns are sequentially formed in the same manner as in the red curable composition to obtain a color filter for a liquid crystal display element. Made. When the obtained color filter was incorporated into a liquid crystal display element, it was confirmed that the liquid crystal display element is excellent in color characteristics and contrast.
- Solid-state imaging device With respect to the above curable composition, except that only the pigment was changed to a blue pigment and a green pigment, green and blue colored curable compositions were similarly prepared. First, 1.6.times.1.6 .mu.m red, green and blue patterns were sequentially formed on the substrate in the same manner as in the case of the red curable composition, to produce a color filter for a solid-state imaging device. When the obtained color filter was incorporated into a solid-state imaging device, it was confirmed that the solid-state imaging device has high resolution and excellent color separation.
Landscapes
- Physics & Mathematics (AREA)
- Chemical & Material Sciences (AREA)
- General Physics & Mathematics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Engineering & Computer Science (AREA)
- Optics & Photonics (AREA)
- Structural Engineering (AREA)
- Architecture (AREA)
- Power Engineering (AREA)
- Ceramic Engineering (AREA)
- Electromagnetism (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Computer Hardware Design (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Materials For Photolithography (AREA)
- Optical Filters (AREA)
- Solid State Image Pick-Up Elements (AREA)
Abstract
Description
液晶ディスプレイは、表示装置としてCRTと比較すると、コンパクトであり、且つ、性能面では同等以上であることから、テレビ画面、パソコン画面、その他の表示装置としてCRTに置き換わりつつある。また、近年では、液晶ディスプレイの開発の動向は、画面が比較的小面積であった従来のモニター用途から、画面が大型で高度な画質が求められるTV用途に向かいつつある。
このような要因により、液晶ディスプレイ用、固体撮像素子用いずれの場合においても、硬化性組成物を硬化させるために必要な成分である光重合開始剤および光重合性モノマーの含有量が制限される上に、着色剤濃度が高くなり、薄層化が困難であり、また、重合成分比率が低く所望の感度を達成することが難しい、又、現像調整剤等を入れる余裕が無く、所望とする現像性を確保することが難しい。
着色性を低下させずに感度を確保する目的で、顔料の含有量は維持しつつ、顔料分散剤の添加量を減らすことも試みられているが、顔料分散液の安定性が低くなり、経時で粘度上昇を引き起こし、均一な塗膜を形成しがたいという新たな問題が生じる。
このように、着色硬化性組成物を、カラーフィルタ、特に固体撮像素子用カラーフィルタにおける着色領域の形成に用いる場合、薄膜化と色価の双方を満足させることが必要であり、顔料濃度が高く、且つ、感度、現像性の両立を達成しうる着色硬化性組成物が熱望されている。
本発明のさらなる目的は、前記着色硬化性組成物により形成された薄層で色特性に優れた高精細の着色領域を備えるカラーフィルタ、及び、色特性に優れたカラーフィルタを備える固体撮像素子を提供することにある。
即ち、本発明の構成は以下に示すとおりである。
<2>前記(a-2)分子内に顔料母核構造とアミノ基とを有する化合物が、アミド構造及びウレア構造の少なくとも1つを有する化合物であることを特徴とする前記<1>に記載の着色硬化性組成物。
<4>カラーフィルタの着色領域形成に用いられることを特徴とする前記<1>~<3>のいずれかに記載の着色硬化性組成物。
<6>前記<5>に記載のカラーフィルタを備えることを特徴とする固体撮像素子。
本発明の硬化性組成物を調製するに際し、まず、(a-1)顔料、(a-2)分子内に顔料母核構造とアミノ基とを有する化合物、および(a-3)酸基と重合性基を有する樹脂、を含んでなる(A)顔料分散液を得て、その(A)顔料分散液を、(B)オキシムエステル系開始剤および(C)重合性化合物と混合して着色硬化性組成物を形成する。
顔料分散液に含まれる(a-2)分子内に顔料母核構造とアミノ基とを有する化合物〔以下適宜、(a-2)特定顔料誘導体と称する〕における顔料母核構造は、類似の母核構造を有する(a-1)顔料との間で相互作用を形成し、(a-2)特定顔料誘導体が(a-1)顔料表面に吸着する。また、この(a-2)特定顔料誘導体におけるアミノ基は、共存する(a-3)酸基と重合性基を有する樹脂〔以下適宜、(a-3)特定分散樹脂と称する〕との間で相互作用を形成するために、(A)顔料分散液中では、(a-1)顔料表面に(a-3)特定分散樹脂が、(a-2)特定顔料誘導体を介して強固に結合するために、(a-3)特定分散樹脂が顔料の表面から離脱し難くなり、長期間に亘り、微細な顔料の凝集が抑制される。
また、感光層中の顔料濃度が高くなる場合には、所望の色価、膜厚に制御する為に、組成物中へ重合性化合物を多く添加できず、感度低下を引き起こすが、本発明では、顔料を分散する樹脂に重合性基を有する樹脂を用いることで高感度化が可能となる。
なお、本発明において「顔料母核構造」とは、有機顔料における発色原子団を含む構造であり、通常、窒素原子、酸素原子、硫黄原子などを含むヘテロ環構造やアミド基、ウレア構造などを含む顔料の主骨格構造を指すものとする。さらに、本発明における「顔料母核構造」は、このような顔料主骨格と類似の構造、或いは、顔料主骨格の部分構造を有することで、有機顔料の有する顔料骨格との間で相互作用を形成しうる「顔料骨格の類似構造」、及び、「顔料骨格の部分構造」をも、包含するものとする。
また、前記着色硬化性組成物により形成された薄層で色特性に優れた高精細の着色領域を備えるカラーフィルタ、及び、色特性に優れたカラーフィルタを備える固体撮像素子を提供することができる。
本発明の着色硬化性組成物は、(A)(a-1)顔料、(a-2)分子内に顔料母核構造とアミノ基とを有する化合物、および(a-3)酸基と重合性基を有する樹脂、を含む顔料分散液と、(B)オキシムエステル系開始剤と、(C)重合性化合物と、を含有することを特徴とする。即ち、まず、特定の顔料の分散安定性に優れた(A)顔料分散液を調製し、さらに、(B)オキシムエステル系開始剤、及び、(C)重合性化合物を加えることで、薄層であっても色特性に優れた着色領域を形成しうる。以下、着色硬化性組成物に含まれる各成分について説明する。
本発明の着色硬化性組成物に含まれる(A)顔料分散液は、(a-1)顔料、(a-2)分子内に顔料母核構造とアミノ基とを有する化合物、(a-3)酸基と重合性基を有する樹脂、さらに(a-4)溶媒、を含有する。
<(a-2)分子内に顔料母核構造とアミノ基とを有する化合物((a-2)特定顔料誘導体)>
(a-2)特定顔料誘導体を用いることで、顔料母核構造と(a-1)顔料の間における相互作用による両者の吸着性の確保と、アミノ基による(a-3)特定分散樹脂との相互作用を発現することにより、(A)顔料分散液中での顔料の分散安定性が確保される。
本発明において用いられる(a-2)特定顔料誘導体は、下記一般式(A)を部分構造として有する化合物であることが好ましい。
これらの有機基は、さらに置換基を有していてもよく、導入可能な置換基としては、ハロゲン原子、水酸基、アルキル基、アルケニル基、アルキニル基、シクロアルキル基、シクロアルケニル基、シクロアルキニル基、アリール基、ヘテロ環基、シアノ基、アルコキシ基、アリーロキシ基、チオアルコキシ基、チオアリーロキシ基、カルボキシ基、アルコキシカルボニル基、スルホ基、スルホンアミド基、ウレア基、チオウレア基、アミノ基、アミド基、カルボニル基、ニトロ基またはこれらの基を有する置換基が挙げられる。
また、R1およびR2は互いに結合し、環を形成してもよい。
また、一般式(A)で表される化合物は、ヘテロ環構造を有する化合物であることが好ましい。
ここで、顔料母核構造としては、前記の如く有機顔料における発色原子団、その類似構造、或いは部分構造が挙げられ、具体的には、アゾ基を有する骨格、ウレア構造を有する骨格、アミド構造を有する骨格、環状アミド構造を有する骨格、ヘテロ原子含有5員環を有する芳香族環、及び、ヘテロ原子含有6員環を有する芳香族環から選択される1種以上の部分構造を含む構造が挙げられ、Xはこれらの顔料母核構造を含む置換基である。
本発明におけるXとしては、好ましくは顔料母核構造、又は顔料母核構造と芳香環、或いは含窒素芳香環、或いは含酸素芳香環、或いは含硫黄芳香環を有し、アミノ基は顔料母核構造、芳香環、含窒素芳香環、含酸素芳香環、含硫黄芳香環のいずれかに直接或いは連結基により結合されている。
最も好ましくは顔料母核構造と芳香環、或いは含窒素芳香環を有し、アミノ基と2価の連結基で結合されていることが好ましい。
mは、1~8の整数であり、分散性、分散液の保存安定性の観点から1~6が好ましく、更に好ましくは2~6である。
以下に、本発明に用いられる(a-2)特定顔料誘導体の好ましい具体例を示すが、これらに限定されるものではない。
また、後述する(a-1)顔料の100質量部に対して、0.5質量部以上50質量部以下であることが好ましく、1質量部以上25質量部以下であることがより好ましい。
本発明に係る(A)顔料分散液に用いることができる顔料としては、従来公知の種々の無機顔料又は有機顔料を用いることができる。また、顔料は、無機顔料または有機顔料を問わず、高透過率であることが好ましいことを考慮すると、できるだけ粒子径が小さく微少な粒子サイズの顔料を使用することが好ましく、ハンドリング性をも考慮すると、好ましくは平均一次粒子径0.01μm~0.3μm、より好ましくは0.01μm~0.15μmの顔料である。該粒径が前記範囲内であると、透過率が高く、色特性が良好であると共に、高いコントラストのカラーフィルタを形成するのに有効である。
平均一次粒子径は、SEMあるいはTEMで観察し、粒子が凝集していない部分で粒子サイズを100個計測し、平均値を算出することによって求める。
C.I.Pigment Red 1、2、3、4、5、6、7、9、10、14、17、22、23、31、38、41、48:1、48:2、48:3、48:4、49、49:1、49:2、52:1、52:2、53:1、57:1、60:1、63:1、66、67、81:1、81:2、81:3、83、88、90、105、112、119、122、123、144、146、149、150、155、166、168、169、170、171、172、175、176、177、178、179、184、185、187、188、190、200、202、206、207、208、209、210、216、220、224、226、242、246、254、255、264、270、272、279、
C.I.Pigment Yellow 1,2,3,4,5,6,10,11,12,13,14,15,16,17,18,20,24,31,32,34,35,35:1,36,36:1,37,37:1,40,42,43,53,55,60,61,62,63,65,73,74,77,81,83,86,93,94,95,97,98,100,101,104,106,108,109,110,113,114,115,116,117,118,119,120,123,125,126,127,128,129,137,138,139,147,148,150,151,152,153,154,155,156,161,162,164,166,167,168,169,170,171,172,173,174,175,176,177,179,180,181,182,185,187,188,193,194,199,213,214
C.I. Pigment Orange 2,5,13,16,17:1,31,34,36,38,43,46,48,49,51,52,55,59,60,61,62,64,71,73
C.I. Pigment Green 7,10,36,37、58
C.I.Pigment Blue 1,2,15,15:1,15:2,15:3,15:4,15:6,16,22,60,64,66,79,79のCl置換基をOHに変更したもの、80
C.I.Pigment Violet 1,19,23,27,32,37,42
C.I.Pigment Brown 25,28
C.I.Pigment Black 1,7等を挙げることができる。
C.I.Pigment Yellow 11,24,108,109,110,138,139,150,151,154,167,180,185,
C.I.Pigment Orange 36,71,
C.I.Pigment Red 122,150,171,175,177,209,224,242,254,255,264,
C.I.Pigment Violet 19,23,32,
C.I.Pigment Blue 15:1,15:3,15:6,16,22,60,66,
C.I.Pigment Green 7,36,37;
C.I.Pigment Black 1,7
本発明においては、必要に応じて、微細でかつ整粒化された有機顔料を用いることができる。顔料の微細化は、顔料と水溶性有機溶剤と水溶性無機塩類と共に高粘度な液状組成物を調製し、湿式粉砕装置等を使用して、応力を付加して摩砕する工程を経ることで達成される。
また、少量用いることで顔料に吸着して、廃水中に流失しない限りにおいては、水溶性は低いか、或いは、水溶性を有しない他の溶剤、例えば、ベンゼン、トルエン、キシレン、エチルベンゼン、クロロベンゼン、ニトロベンゼン、アニリン、ピリジン、キノリン、テトラヒドロフラン、ジオキサン、酢酸エチル、酢酸イソプロピル、酢酸ブチル、ヘキサン、ヘプタン、オクタン、ノナン、デカン、ウンデカン、ドデカン、シクロヘキサン、メチルシクロヘキサン、ハロゲン化炭化水素、アセトン、メチルエチルケトン、メチルイソブチルケトン、シクロヘキサノン、ジメチルホルムアミド、ジメチルスルホキシド、N-メチルピロリドン等を用いてもよい。
顔料の微細化工程に使用する溶剤は、1種のみでもよく、必要に応じて2種類以上を混合して使用してもよい。
微細化工程における水溶性無機塩の使用量は顔料の1~50質量倍であり、多い方が摩砕効果はあるが、より好ましい量は生産性の点で1~10質量倍である。また、水分が1%以下の無機塩類を用いることが好ましい。
微細化工程における水溶性有機溶剤の使用量は、顔料100質量部に対して50質量部から300質量部の範囲であり、好ましくは100質量部から200質量部の範囲である。
これら有機顔料は、単独もしくは色純度を上げるため種々組合せて用いることができる。上記組合せの具体例を以下に示す。例えば、赤の顔料として、アントラキノン系顔料、ペリレン系顔料、ジケトピロロピロール系顔料単独又はそれらの少なくとも1種と、ジスアゾ系黄色顔料、イソインドリン系黄色顔料、キノフタロン系黄色顔料又はペリレン系赤色顔料、アントラキノン系赤色顔料、ジケトピロロピロール系赤色顔料と、の混合などを用いることができる。例えば、アントラキノン系顔料としては、C.I.ピグメント・レッド177が挙げられ、ペリレン系顔料としては、C.I.ピグメント・レッド155、C.I.ピグメント・レッド224が挙げられ、ジケトピロロピロール系顔料としては、C.I.ピグメント・レッド254が挙げられ、色再現性の点でC.I.ピグメント・イエロー83、C.I.ピグメント・イエロー139またはC.I.ピグメント・レッド177との混合が好ましい。また、赤色顔料と他顔料との質量比は、100:5~100:80が好ましい。この範囲においては、400nmから500nmの光透過率を抑え、色純度の向上が図れ、且つ、十分な発色力が達成される。特に、上記質量比としては、100:10~100:65の範囲が最適である。なお、赤色顔料同士の組み合わせの場合は、色度に併せて調整することができる。
青色顔料と紫色顔料との質量比は、100:0~100:100が好ましく、より好ましくは100:70以下である。
また、カーボンブラックとチタンブラックとの質量比は、100:0~100:60の範囲が好ましい。100:61以上では、分散安定性が低下する場合がある。
本発明において、色調を合わせるために、着色剤として、顔料とともに染料を使用することが可能である。
着色剤として使用可能な染料としては、特に制限はなく、従来カラーフィルタ用途に用いられている公知の染料を使用できる。例えば、特開昭64-90403号公報、特開昭64-91102号公報、特開平1-94301号公報、特開平6-11614号公報、特登2592207号、米国特許第4,808,501号明細書、米国特許第5,667,920号明細書、米国特許第5,059,500号明細書、特開平5-333207号公報、特開平6-35183号公報、特開平6-51115号公報、特開平6-194828号公報、特開平8-211599号公報、特開平4-249549号公報、特開平10-123316号公報、特開平11-302283号公報、特開平7-286107号公報、特開2001-4823号公報、特開平8-15522号公報、特開平8-29771号公報、特開平8-146215号公報、特開平11-343437号公報、特開平8-62416号公報、特開2002-14220号公報、特開2002-14221号公報、特開2002-14222号公報、特開2002-14223号公報、特開平8-302224号公報、特開平8-73758号公報、特開平8-179120号公報、特開平8-151531号公報等に記載の色素である。
化学構造としては、ピラゾールアゾ系、アニリノアゾ系、トリフェニルメタン系、アントラキノン系、アンスラピリドン系、ベンジリデン系、オキソノール系、ピラゾロトリアゾールアゾ系、ピリドンアゾ系、シアニン系、フェノチアジン系、ピロロピラゾールアゾメチン系、キサンテン系、フタロシアニン系、ペンゾピラン系、インジゴ系等の染料が使用できる。
本発明における(A)顔料分散液は、(a-3)酸基と重合性基を含有する樹脂〔(a-3)特定分散樹脂〕の少なくとも1種を含有する。この樹脂の含有により、高い分散性を示し、又、開始剤、重合性化合物と組み合わせた際に感度を向上させることができる。
本願の酸基とは、pKaが14以下の解離性基を有するものであり、具体的には例えば、-COOH、-SO3H、-PO3H2、-OSO3H、-OPO2H2、-PhOH、-SO2H、-SO2NH2、-SO2NHCO-、-SO2NHSO2-等が挙げられ、なかでも-COOH、-SO3H、-PO3H2が好ましく、-COOHが最も好ましい。
R4~R6はそれぞれ独立に、1価の置換基を表すが、R4としては、水素原子または置換基を更に有してもよいアルキル基などが挙げられ、中でも、水素原子、メチル基、エチル基が好ましい。また、R5、R6は、それぞれ独立に、水素原子、ハロゲン原子、アルコキシカルボニル基、スルホ基、ニトロ基、シアノ基、置換基を更に有してもよいアルキル基、置換基を更に有してもよいアリール基、置換基を更に有してもよいアルコキシ基、置換基を更に有してもよいアリールオキシ基、置換基を更に有してもよいアルキルスルホニル基、置換基を更に有してもよいアリールスルホニル基などが挙げられ、中でも、水素原子、アルコキシカルボニル基、置換基を更に有してもよいアルキル基、置換基を更に有してもよいアリール基が好ましい。
ここで、導入しうる置換基としては、メトキシカルボニル基、エトキシカルボニル基、イソプロピオキシカルボニル基、メチル基、エチル基、フェニル基等が挙げられる。
G1は、2価の有機基を表すが、置換基を有してもよいアルキレン基が好ましい。より好ましくは、炭素数1~20の置換基を有してもよいアルキレン基、炭素数3~20の置換基を有してもよいシクロアルキレン基、炭素数6~20の置換基を有してもよい芳香族基などが挙げられ、中でも、置換基を有してもよい炭素数1~10の直鎖状あるいは分岐アルキレン基、炭素数3~10の置換基を有してもよいシクロアルキレン基、炭素数6~12の置換基を有してもよい芳香族基が強度、現像性等の性能上、好ましい。
ここで、G1における置換基としては、水酸基がこのましい。
R10~R12は、それぞれ独立に1価の置換基を表すが、この置換基としては、具体的には例えば、水素原子、ハロゲン原子、ジアルキルアミノ基、アルコキシカルボニル基、スルホ基、ニトロ基、シアノ基、置換基を更に有してもよいアルキル基、置換基を更に有してもよいアリール基、置換基を更に有してもよいアルコキシ基、置換基を更に有してもよいアリールオキシ基、置換基を更に有してもよいアルキルスルホニル基、置換基を更に有してもよいアリールスルホニル基などが挙げられ、中でも、水素原子、アルコキシカルボニル基、置換基を更に有してもよいアルキル基、置換基を更に有してもよいアリール基が好ましい。
ここで、導入可能な置換基としては、一般式(1)において挙げたものが同様に例示される。
G2は、2価の有機基を表すが、置換基を有してもよいアルキレン基が好ましい。好ましくは、炭素数1~20の置換基を有してもよいアルキレン基、炭素数3~20の置換基を有してもよいシクロアルキレン基、炭素数6~20の置換基を有してもよい芳香族基などが挙げられ、中でも、置換基を有してもよい炭素数1~10の直鎖状あるいは分岐アルキレン基、炭素数3~10の置換基を有してもよいシクロアルキレン基、炭素数6~12の置換基を有してもよい芳香族基が強度、現像性等の性能上、好ましい。
ここで、G2における置換基としては、水酸基が好ましい。
R16~R20は、それぞれ独立に1価の置換基を表すが、R16~R20は、例えば、水素原子、ハロゲン原子、ジアルキルアミノ基、アルコキシカルボニル基、スルホ基、ニトロ基、シアノ基、置換基を更に有してもよいアルキル基、置換基を更に有してもよいアリール基、置換基を更に有してもよいアルコキシ基、置換基を更に有してもよいアリールオキシ基、置換基を更に有してもよいアルキルスルホニル基、置換基を更に有してもよいアリールスルホニル基などが挙げられ、中でも、水素原子、アルコキシカルボニル基、置換基を更に有してもよいアルキル基、置換基を更に有してもよいアリール基が好ましい。導入しうる置換基としては、一般式(1)においてあげたものが例示される。
A3は、酸素原子、硫黄原子、又は-N(R21)-を表し、Zは、酸素原子、硫黄原子、又は-N(R22)-を表す。R21、R22としては、一般式(1)におけるのと同様のものが挙げられる。
ここで、G3における置換基としては、水酸基が好ましい。
(1)下記一般式(4)で表される化合物を共重合成分として用いた重合体に、塩基を用いてプロトンを引き抜き、Lを脱離させ、前記一般式(1)で表される構造を有する所望の高分子化合物を得る方法。
尚、一般式(4)中、Lはアニオン性脱離基を表し、好ましくはハロゲン原子、スルホン酸エステル等が挙げられる。R3~R6、A1、G1、及びXについては前記一般式(1)における場合と同義であり、脱離反応を生起させるために用いる塩基としては、無機化合物、有機化合物のどちらを使用してもよい。また、この方法の詳細及び好ましい態様については、特開2003-262958号公報の段落番号[0028]~[0033]に記載されている。
尚、一般式(5)中、A5は酸素原子、硫黄原子、又は-N(R54)-を表し、A6は酸素原子、硫黄原子、又は-NR58-を表し、R51、R52、R53、R54、R55、R56、R57、及びR58は、それぞれ独立に水素又は1価の有機基を表し、X10は脱離反応により除去される基を表し、G5は有機連結基を表す。nは、1~10の整数を表す。また、この方法の詳細及び好ましい態様については、特開2003-335814号公報に詳細に記載されている。
本発明における(a-3)特定分散樹脂を合成する際に、共重合モノマーとして用いることができるモノマーの例としては、下記(1)~(13)が挙げられる。
(2)2-ヒドロキシエチルアクリレート、2-ヒドロキシプロピルアクリレート、3-ヒドロキシプロピルアクリレート、4-ヒドロキシブチルアクリレート、2-ヒドロキシエチルメタクリレート、2-ヒドロキシプロピルメタクリレート、3-ヒドロキシプロピルメタクリレート、4-ヒドロキシブチルメタクリレート等の脂肪族水酸基を有するアクリル酸エステル類、及びメタクリル酸エステル類、及びこれらのエチレンオキシ変性体、ヒドロキシスチレンなどの水酸基を有するモノマー。
(4)メタクリル酸メチル、メタクリル酸エチル、メタクリル酸プロピル、メタクリル酸ブチル、メタクリル酸イソブチル、メタクリル酸アミル、メタクリル酸ヘキシル、メタクリル酸2-エチルヘキシル、メタクリル酸シクロヘキシル、メタクリル酸ベンジル、メタクリル酸-2-クロロエチル、グリシジルメタクリレート、3,4-エポキシシクロヘキシルメチルメタクリレート、ビニルメタクリレート、2-フェニルビニルメタクリレート、1-プロペニルメタクリレート、アリルメタクリレート、2-アリロキシエチルメタクリレート、プロパルギルメタクリレート等のアルキルメタクリレート。
(5)アクリルアミド、メタクリルアミド、N-メチロールアクリルアミド、N-エチルアクリルアミド、N-ヘキシルメタクリルアミド、N-シクロヘキシルアクリルアミド、N-ヒドロキシエチルアクリルアミド、N-フェニルアクリルアミド、N-ニトロフェニルアクリルアミド、N-エチル-N-フェニルアクリルアミド、ビニルアクリルアミド、ビニルメタクリルアミド、N,N-ジアリルアクリルアミド、N,N-ジアリルメタクリルアミド、アリルアクリルアミド、アリルメタクリルアミド等のアクリルアミド若しくはメタクリルアミド。
(7)ビニルアセテート、ビニルクロロアセテート、ビニルブチレート、安息香酸ビニル等のビニルエステル類。
(8)スチレン、α-メチルスチレン、メチルスチレン、クロロメチルスチレン、p-アセトキシスチレン等のスチレン類。
(9)メチルビニルケトン、エチルビニルケトン、プロピルビニルケトン、フェニルビニルケトン等のビニルケトン類。
(10)エチレン、プロピレン、イソブチレン、ブタジエン、イソプレン等のオレフィン類。
(12)マレイミド、N-アクリロイルアクリルアミド、N-アセチルメタクリルアミド、N-プロピオニルメタクリルアミド、N-(p-クロロベンゾイル)メタクリルアミド等の不飽和イミド。
(13)α位にヘテロ原子が結合したメタクリル酸系モノマー。例えば、特開2002-309057号公報、特開2002-311569号公報等に記載されている化合物。
まず、本発明の好ましい態様である、特定ウレタン樹脂について詳細に説明する。
本発明に用いられる特定ウレタン樹脂は、その側鎖に、下記一般式(1)~(3)で表される官能基のうち少なくとも1つを有するものが挙げられる。まず、下記一般式(1)~(3)で表される官能基について説明する。
本発明に係る特定ウレタン樹脂は、下記一般式(4)で表されるジイソシアネート化合物の少なくとも1種と、一般式(5)で表されるジオール化合物の少なくとも1種と、の反応生成物で表される構造単位を基本骨格とするポリウレタン樹脂(以下、適宜「特定ポリウレタン樹脂と称する)である。
HO-Y0-OH 一般式(5)
上記一般式(4)で表されるジイソシアネート化合物としては、例えば、トリイソシアネート化合物と、不飽和基を有する単官能のアルコール又は単官能のアミン化合物1当量とを付加反応させて得られる生成物がある。
トリイソシアネート化合物としては、例えば下記に示すものが挙げられるが、これに限定されるものではない。
すなわち、2,4-トリレンジイソシアネート、2,4-トリレンジイソシアネートの二量体、2,6-トリレンジイソシアネート、p-キシリレンジイソシアネート、m-キシリレンジイソシアネート、4,4’-ジフェニルメタンジイソシアネート、1,5-ナフチレンジイソシアネート、3,3’-ジメチルビフェニル-4,4’-ジイソシアネート等のような芳香族ジイソシアネート化合物;
ヘキサメチレンジイソシアネート、トリメチルヘキサメチレンジイソシアネート、リジンジイソシアネート、ダイマー酸ジイソシアネート等のような脂肪族ジイソシアネート化合物;
イソホロンジイソシアネート、4,4’-メチレンビス(シクロヘキシルイソシアネート)、メチルシクロヘキサン-2,4(又は2,6)ジイソシアネート、1,3-(イソシアネートメチル)シクロヘキサン等のような脂環族ジイソシアネート化合物;
1,3-ブチレングリコール1モルとトリレンジイソシアネート2モルとの付加体等のようなジオールとジイソシアネートとの反応物であるジイソシアネート化合物;等が挙げられる。
上記一般式(5)で表されるジオール化合物としては、広くは、ポリエーテルジオール化合物、ポリエステルジオール化合物、ポリカーボネートジオール化合物等が挙げられる。
なお、この一般式(G)におけるR1~R3及びXは、前記一般式(1)におけるR1~R3及びXと同義であり、好ましい態様もまた同様である。
このようなジオール化合物に由来するポリウレタン樹脂を用いることにより、立体障害の大きい2級アルコールに起因するポリマー主鎖の過剰な分子運動を抑制する効果により、層の被膜強度の向上が達成できるものと考えられる。
以下、特定ポリウレタン樹脂の合成に好適に用いられる一般式(G)で表されるジオール化合物の具体例を示す。
そのようなジオール化合物としては、例えば、上述したポリエーテルジオール化合物、ポリエステルジオール化合物、ポリカーボネートジオール化合物を挙げることできる。
すなわち、ジエチレングリコール、トリエチレングリコール、テトラエチレングリコール、ペンタエチレングリコール、ヘキサエチレングリコール、ヘプタエチレングリコール、オクタエチレングリコール、ジ-1,2-プロピレングリコール、トリ-1,2-プロピレングリコール、テトラ-1,2-プロピレングリコール、ヘキサ-1,2-プロピレングリコール、ジ-1,3-プロピレングリコール、トリ-1,3-プロピレングリコール、テトラ-1,3-プロピレングリコール、ジ-1,3-ブチレングリコール、トリ-1,3-ブチレングリコール、ヘキサ-1,3-ブチレングリコール、重量平均分子量1000のポリエチレングリコール、重量平均分子量1500のポリエチレングリコール、重量平均分子量2000のポリエチレングリコール、重量平均分子量3000のポリエチレングリコール、重量平均分子量7500のポリエチレングリコール、重量平均分子量400のポリプロピレングリコール、重量平均分子量700のポリプロピレングリコール、重量平均分子量1000のポリプロピレングリコール、重量平均分子量2000のポリプロピレングリコール、重量平均分子量3000のポリプロピレングリコール、重量平均分子量4000のポリプロピレングリコール等である。
すなわち、三洋化成工業(株)製、(商品名)PTMG650、PTMG1000、PTMG2000、PTMG3000等である。
すなわち、三洋化成工業(株)製、(商品名)ニューポールPE-61、ニューポールPE-62、ニューポールPE-64、ニューポールPE-68、ニューポールPE-71、ニューポールPE-74、ニューポールPE-75、ニューポールPE-78、ニューポールPE-108、ニューポールPE-128、ニューポールPE-61等である。
すなわち、三洋化成工業(株)製、(商品名)ニューポールBPE-20、ニューポールBPE-20F、ニューポールBPE-20NK、ニューポールBPE-20T、ニューポールBPE-20G、ニューポールBPE-40、ニューポールBPE-60、ニューポールBPE-100、ニューポールBPE-180、ニューポールBPE-2P、ニューポールBPE-23P、ニューポールBPE-3P、ニューポールBPE-5P等である。
すなわち、三洋化成工業(株)製、(商品名)ニューポール50HB-100、ニューポール50HB-260、ニューポール50HB-400、ニューポール50HB-660、ニューポール50HB-2000、ニューポール50HB-5100等である。
ポリカーボネートジオール化合物としては、式(14)で表される化合物がある。
HO-L8-CO-O-L7-OH (16)
このようなジオール化合物としては、例えば、以下の式(17)~(19)に示すものが含まれる。
Arは置換基を有していてもよい三価の芳香族炭化水素基を表し、好ましくは炭素数6~15個の芳香族基を表す。
すなわち、3,5-ジヒドロキシ安息香酸、2,2-ビス(ヒドロキシメチル)プロピオン酸、2,2-ビス(2-ヒドロキシエチル)プロピオン酸、2,2-ビス(3-ヒドロキシプロピル)プロピオン酸、ビス(ヒドロキシメチル)酢酸、ビス(4-ヒドロキシフェニル)酢酸、2,2-ビス(ヒドロキシメチル)酪酸、4,4-ビス(4-ヒドロキシフェニル)ペンタン酸、酒石酸、N,N-ジヒドロキシエチルグリシン、N,N―ビス(2-ヒドロキシエチル)-3-カルボキシ-プロピオンアミド等である。
すなわち、ピロメリット酸二無水物、3,3’,4,4’-べンゾフェノンテトラカルボン酸二無水物、3,3’,4,4’-ジフェニルテトラカルボン酸二無水物、2,3,6,7-ナフタレンテトラカルボン酸二無水物、1,4,5,8-ナフタレンテトラカルボン酸二無水物、4,4’-スルホニルジフタル酸二無水物、2,2-ビス(3,4-ジカルボキシフェニル)プロパン二無水物、ビス(3,4-ジカルボキシフェニル)エーテル二無水物、4,4’-[3,3’-(アルキルホスホリルジフェニレン)-ビス(イミノカルボニル)]ジフタル酸二無水物、
a)テトラカルボン酸二無水物をジオール化合物で開環させて得られたアルコール末端の化合物と、ジイソシアネート化合物とを反応させる方法。
b)ジイソシアネート化合物をジオール化合物過剰の条件下で反応させ得られたアルコール末端のウレタン化合物と、テトラカルボン酸二無水物とを反応させる方法。
すなわち、エチレングリコール、ジエチレングリコール、トリエチレングリコール、テトラエチレングリコール、プロピレングリコール、ジプロピレングリコール、ポリエチレングリコール、ポリプロピレングリコール、ネオペンチルグリコール、1,3-ブチレングリコール、1,6-ヘキサンジオール、2-ブテン-1,4-ジオール、2,2,4-トリメチル-1,3-ペンタンジオール、1,4-ビス-β-ヒドロキシエトキシシクロヘキサン、シクロヘキサンジメタノール、トリシクロデカンジメタノール、水添ビスフェノールA、水添ビスフェノールF、ビスフェノールAのエチレンオキサイド付加体、ビスフェノールAのプロピレンオキサイド付加体、ビスフェノールFのエチレンオキサイド付加体、ビスフェノールFのプロピレンオキサイド付加体、水添ビスフェノールAのエチレンオキサイド付加体、水添ビスフェノールAのプロピレンオキサイド付加体、ヒドロキノンジヒドロキシエチルエーテル、p-キシリレングリコール、ジヒドロキシエチルスルホン、ビス(2-ヒドロキシエチル)-2,4-トリレンジカルバメート、2,4-トリレン-ビス(2-ヒドロキシエチルカルバミド)、ビス(2-ヒドロキシエチル)-m-キシリレンジカルバメート、ビス(2-ヒドロキシエチル)イソフタレート等が挙げられる。
なお、不飽和基は、導入量の制御が容易で導入量を増やすことができ、また、架橋反応効率が向上するといった観点から、ポリマー末端よりもポリマー側鎖に導入されることが好ましい。
導入されるエチレン性不飽和結合基としては、架橋硬化膜形成性の観点から、メタクリロイル基、アクリロイル基、スチリル基が好ましく、より好ましくはメタクリロイル基、アクリロイル基である。架橋硬化膜の形成性と生保存性との両立の観点からは、メタクリロイル基がさらに好ましい。
また、メタクリロイル基の導入量としては、先に述べたように0.30meq/g以上であることが好ましく、0.35~1.50meq/gの範囲であることがさらに好ましい。即ち、本発明のバインダーポリマーとしては、側鎖にメタクリロイル基を0.35~1.50meq/gの範囲で導入したポリウレタン樹脂が最も好ましい態様である。
すなわち、cis-2-ブテン-1,4-ジオール、trans-2-ブテン-1,4-ジオール、ポリブタジエンジオール等である。
本発明に用いられる特定スチレン系樹脂とは、その側鎖に、下記一般式(23)で表されるスチレン性二重結合(スチレンおよびαメチルスチレン系二重結合)、及び、下記一般式(24)で表されるビニルピリジニウム基のうち少なくとも一方を有するものが挙げられる。
なお、一般式(23)で表されるスチレン性二重結合は、単結合、或いは、任意の原子または原子団からなる連結基を介してポリマー主鎖と連結しており、結合の仕方について特に制限はない。
以下に、一般式(23)で表される官能基を有する高分子化合物の繰り返し単位として好ましい例を示すが、本発明はこれらの例に限定されるものではない。
なお、一般式(24)で表されるビニルピリジニウム基は、単結合、或いは、任意の原子または原子団からなる連結基を介してポリマー主鎖と連結しており、結合の仕方について特に制限はない。
以下に、一般式(24)で表される官能基を有する高分子化合物の繰り返し単位として好ましい例を示すが、本発明はこれらの例に限定されるものではない。
特に、一般式(23)で表される官能基を繰り返し単位中に有し、且つ、主鎖と一般式(23)で表される官能基とを連結する連結基中に4級塩構造を有する場合(例えば、前記具体例等)には、このような構造を有するホモポリマーであっても良いが、こうした場合以外では、以下に挙げる他の共重合モノマーとの共重合体であることが好ましい。例えば、4-ビニルベンジルトリメチルアンモニウムクロライド、アクリロイルオキシエチルトリメチルアンモニウムクロライド、メタクリロイルオキシエチルトリメチルアンモニウムクロライド、ジメチルアミノプロピルアクリルアミドのメチルクロライドによる4級化物、N-ビニルイミダゾールのメチルクロライドによる4級化物、4-ビニルベンジルピリジニウムクロライド等が好ましく使用される。
また、一般式(24)で表される官能基を繰り返し単位中に含む場合に於いては、ホモポリマーであっても良いし、上記他の共重合モノマーとの共重合体であっても良い。
側鎖にエチレン性不飽和基を有するノボラック樹脂としては、例えば、特開平9-269596号公報に記載のポリマーに、特開2002-62648公報に記載の方法を用いて、側鎖にエチレン性不飽和結合を導入した樹脂等が挙げられる。
側鎖にエチレン性不飽和結合を有するアセタール樹脂としては、例えば、特開2002-162741公報に記載の樹脂等が挙げられる。
側鎖にエチレン性不飽和結合を有するポリアミド系樹脂としては、例えば、特願2003-321022公報に記載の樹脂、又はその中で引用されているポリアミド樹脂に、特開2002-62648公報に記載の方法で側鎖にエチレン性不飽和結合を導入した樹脂等が挙げられる。
側鎖にエチレン性不飽和結合を有するポリイミド樹脂としては、例えば、特願2003-339785公報に記載の樹脂、又はその中で引用されているポリイミド樹脂に、特開2002-62648公報に記載の方法で側鎖にエチレン性不飽和結合を導入した樹脂等が挙げられる。
-他の分散剤-
本願の分散液において、分散安定性向上、及び或いは現像性を調整する目的として、上述した特定分散樹脂以外の他の分散剤を使用することができる。
他の分散剤としては、例えば、公知の顔料分散剤や界面活性剤を適宜選択して用いることができる。
本発明における(A)顔料分散液には、必要に応じて、前記本発明に係る(a-2)特定顔料誘導体以外の、他の顔料誘導体を添加することができる。
他の顔料誘導体を用いることで、分散剤と親和性のある部分、あるいは極性基を導入した顔料誘導体を顔料表面に吸着させて分散剤の吸着点として用いうるため、顔料を微細な粒子として光硬化性組成物中に分散させ、その再凝集を防止することができ、コントラストが高く、透明性に優れたカラーフィルタを構成するのに有効である。
前記(a-1)~(a-3)及び任意成分を含む顔料分散液を調製する方法としては、例えば、(a-1)顔料と(a-3)特定分散樹脂を予め混合して、ホモジナイザー等で分散しておいたものを、ジルコニアビーズ等を用いたビーズ分散機(例えばGETZMANN社製のディスパーマット)等を用いて微分散させることによって行なえる。分散時間としては、3~6時間程度が好適である。(a-2)特定顔料誘導体の添加は、顔料分散液を形成する為のどの工程でも添加可能であるが、微細化工程及び/或いは微分散時に添加することが好ましい。
本発明の着色硬化性組成物に含まれる(A)顔料分散液の量としては、5~90質量%の範囲であることが好ましく、25~85質量%の範囲であることがより好ましい。
また、(A)顔料分散液を上記配合量で含むことで、本発明の着色硬化性組成物に含まれる(a-1)顔料の量は、好ましくは固形分中25~65質量%の範囲となり、薄層でも十分な色相を有する着色領域を形成しうる。
本発明の着色硬化性組成物において、特に薄膜を形成する際には、前述の如く顔料濃度を高くすることを要するため、ラジカルを発生させる開始剤を多く添加できない。また、顔料濃度が高いために、増感剤と開始剤の濃度を十分にとれず、増感剤を用いての電子移動、エネルギー移動による開始機構による硬化性向上効果は十分得られない。
特に固体撮像素子の如き、高精細で微細な着色領域を形成するために本発明の硬化性組成物を使用する場合には、高感度であっても、露光によりハロゲンを発生するトリアジン系の開始剤は、ステッパー露光装置等の光源を汚染する懸念があり、使用することができない。このため、該条件下で、300nm以上の光源に幅広く対応可能であり、且つ、光により直接分解するオキシム化合物を開始剤として用いる、即ちオキシムエステル系開始剤(以下、適宜、オキシム型開始剤と称する場合がある。)を用いることが好ましい。特に、顔料が硬化性組成物全固形分中30質量%以上、更に40質量%以上含み、重合性成分の比率が低い硬化性組成物において、オキシム型開始剤の使用はより効果的である。
これらは、さらに置換基を有するものであってもよく、導入可能な置換基としては、ハロゲン原子、水酸基、アルキル基、アルケニル基、アルキニル基、シクロアルキル基、シクロアルケニル基、シクロアルキニル基、アリール基、ヘテロ環基、シアノ基、アルコキシ基、アリーロキシ基、チオアルコキシ基、チオアリーロキシ基、カルボキシル基、アルコキシカルボニル基、スルホ基、スルホンアミド基、ウレア基、チオウレア基、アミノ基、アミド基、カルボニル基、ニトロ基或いはこれらの基を有する置換基が挙げられる。
これらのなかでも、経時安定性、感度、生産性の面から最も好ましくは一般式(III)で表される置換基であって、R4がアルキル基、またはアリール基である。
これらは、さらに置換基を有するものであってもよく、導入可能な置換基としては、ハロゲン原子、水酸基、アルキル基、アルケニル基、アルキニル基、シクロアルキル基、シクロアルケニル基、シクロアルキニル基、シアノ基、アリール基、ヘテロ環基、アルコキシ基、アリーロキシ基、チオアルコキシ基、チオアリーロキシ基、カルボキシル基、アルコキシカルボニル基、スルホ基、スルホンアミド基、ウレア基、チオウレア基、アミノ基、アミド基、カルボニル基、ニトロ基或いはこれらの基を有する置換基が挙げられる。
これらは、さらに置換基を有するものであってもよく、導入可能な置換基としては、ハロゲン原子、水酸基、アルキル基、アルケニル基、アルキニル基、シクロアルキル基、シクロアルケニル基、シクロアルキニル基、アリール基、シアノ基、ヘテロ環基、アルコキシ基、アリーロキシ基、チオアルコキシ基、チオアリーロキシ基、カルボキシル基、アルコキシカルボニル基、スルホ基、スルホンアミド基、ウレア基、チオウレア基、アミノ基、アミド基、カルボニル基、ニトロ基或いはこれらの基を有する置換基が挙げられる。
本発明に好ましく用いうる(B)オキシム型開始剤の具体例を以下に示すが、本発明はこれらに限定されるものではない。
(B)オキシム型開始剤は高感度であるため、顔料を多量に含む本発明の着色硬化性組成物において、上記配合量においても、優れた硬化膜を形成することが可能である。
本発明には、硬化成分として、前記(B)オキシム型開始剤とともに、(C)重合性化合物を用いる。本発明に用いうる重合性化合物としては、一般的なラジカル重合性化合物が挙げられ、当該産業分野においてエチレン性不飽和二重結合を有する化合物として広く知られる化合物を特に限定無く用いることができる。これらは、例えばモノマー、プレポリマー、すなわち2量体、3量体及びオリゴマー、又はそれらの混合物ならびにそれらの共重合体などの化学的形態をもつ。
モノマー及びその共重合体の例としては、不飽和カルボン酸(例えば、アクリル酸、メタクリル酸、イタコン酸、クロトン酸、イソクロトン酸、マレイン酸など)や、そのエステル類、アミド類が挙げられ、好ましくは、不飽和カルボン酸と脂肪族多価アルコール化合物とのエステル、不飽和カルボン酸と脂肪族多価アミン化合物とのアミド類が用いられる。また、ヒドロキシル基やアミノ基、メルカプト基等の求核性置換基を有する不飽和カルボン酸エステル或いはアミド類と単官能若しくは多官能イソシアネート類或いはエポキシ類との付加反応物、及び単官能若しくは、多官能のカルボン酸との脱水縮合反応物等も好適に使用される。また、イソシアネート基や、エポキシ基等の親電子性置換基を有する不飽和カルボン酸エステル或いはアミド類と単官能若しくは多官能のアルコール類、アミン類、チオール類との付加反応物、更にハロゲン基や、トシルオキシ基等の脱離性置換基を有する不飽和カルボン酸エステル或いはアミド類と単官能若しくは多官能のアルコール類、アミン類、チオール類との置換反応物も好適である。また、別の例として、上記の不飽和カルボン酸の代わりに、不飽和ホスホン酸、スチレン、ビニルエーテル等に置き換えた化合物群を使用することも可能である。
(ただし、R4及びR5は、H又はCH3を示す。)
本発明の着色硬化性組成物における(C)重合性化合物の含有量は、固形分換算で、
4~80質量%の範囲であることが好ましく、7~50質量%の範囲であることがさらに好ましい。
特に膜厚が0.8μm以下の硬化膜を形成しようとする場合には、(C)重合性化合物の含有量は、全固形分中7~40質量%、特に6~25質量%の範囲で有効である。
以下、本発明の着色硬化性組成物に含まれうる任意成分について説明する。
本発明の硬化性組成物においては、皮膜特性向上、現像性の調整などの目的で、必要に応じて、さらにバインダーポリマーを使用することができる。
バインダーとしては線状有機ポリマーを用いることが好ましい。このような「線状有機ポリマー」としては、公知のものを任意に使用できる。好ましくは水現像あるいは弱アルカリ水現像を可能とするために、水あるいは弱アルカリ水に可溶性又は膨潤性である線状有機ポリマーが選択される。線状有機ポリマーは、皮膜形成剤としてだけでなく、水、弱アルカリ水あるいは有機溶剤現像剤としての用途に応じて選択使用される。例えば、水可溶性有機ポリマーを用いると水現像が可能になる。このような線状有機ポリマーとしては、側鎖にカルボン酸基を有するラジカル重合体、例えば特開昭59-44615号、特公昭54-34327号、特公昭58-12577号、特公昭54-25957号、特開昭54-92723号、特開昭59-53836号、特開昭59-71048号に記載されているもの、すなわち、カルボキシル基を有するモノマーを単独あるいは共重合させた樹脂、酸無水物を有するモノマーを単独あるいは共重合させ酸無水物ユニットを加水分解もしくはハーフエステル化もしくはハーフアミド化させた樹脂、エポキシ樹脂を不飽和モノカルボン酸および酸無水物で変性させたエポキシアクリレート等が挙げられる。カルボキシル基を有するモノマーとしては、アクリル酸、メタクリル酸、イタコン酸、クロトン酸、マレイン酸、フマル酸、4-カルボキシルスチレン等があげられ、酸無水物を有するモノマーとしては、無水マレイン酸等が挙げられる。
また同様に側鎖にカルボン酸基を有する酸性セルロース誘導体がある。この他に水酸基を有する重合体に環状酸無水物を付加させたものなどが有用である。
(2)アクリル酸メチル、アクリル酸エチル、アクリル酸プロピル、アクリル酸ブチル、アクリル酸イソブチル、アクリル酸アミル、アクリル酸ヘキシル、アクリル酸2-エチルヘキシル、アクリル酸オクチル、アクリル酸ベンジル、アクリル酸-2-クロロエチル、グリシジルアクリレート、3,4-エポキシシクロヘキシルメチルアクリレート、ビニルアクリレート、2-フェニルビニルアクリレート、1-プロペニルアクリレート、アリルアクリレート、2-アリロキシエチルアクリレート、プロパルギルアクリレート等のアルキルアクリレート。
(4)アクリルアミド、メタクリルアミド、N-メチロールアクリルアミド、N-エチルアクリルアミド、N-ヘキシルメタクリルアミド、N-シクロヘキシルアクリルアミド、N-ヒドロキシエチルアクリルアミド、N-フェニルアクリルアミド、N-ニトロフェニルアクリルアミド、N-エチル-N-フェニルアクリルアミド、ビニルアクリルアミド、ビニルメタクリルアミド、N,N-ジアリルアクリルアミド、N,N-ジアリルメタクリルアミド、アリルアクリルアミド、アリルメタクリルアミド等のアクリルアミド若しくはメタクリルアミド。
(6)ビニルアセテート、ビニルクロロアセテート、ビニルブチレート、安息香酸ビニル等のビニルエステル類。
(7)スチレン、α-メチルスチレン、メチルスチレン、クロロメチルスチレン、p-アセトキシスチレン等のスチレン類。
(8)メチルビニルケトン、エチルビニルケトン、プロピルビニルケトン、フェニルビニルケトン等のビニルケトン類。
(9)エチレン、プロピレン、イソブチレン、ブタジエン、イソプレン等のオレフィン類。
(11)マレイミド、N-アクリロイルアクリルアミド、N-アセチルメタクリルアミド、N-プロピオニルメタクリルアミド、N-(p-クロロベンゾイル)メタクリルアミド等の不飽和イミド。
(12)α位にヘテロ原子が結合したメタクリル酸系モノマー。例えば、特願2001-115595号明細書、特願2001-115598号明細書等に記載されている化合物を挙げる事ができる。
また、欧州特許993966、欧州特許1204000、特開2001-318463等に記載の酸基を有するアセタール変性ポリビニルアルコール系バインダーポリマーは、膜強度、現像性のバランスに優れており、好適である。
さらにこの他に水溶性線状有機ポリマーとして、ポリビニルピロリドンやポリエチレンオキサイド等が有用である。また硬化皮膜の強度を上げるためにアルコール可溶性ナイロンや2,2-ビス-(4-ヒドロキシフェニル)-プロパンとエピクロロヒドリンのポリエーテル等も有用である。
これらのバインダーポリマーは、ランダムポリマー、ブロックポリマー、グラフトポリマー等いずれでもよい。
本発明において用いうるバインダーポリマーを合成する際に用いられるラジカル重合開始剤としては、アゾ系開始剤、過酸化物開始剤等公知の化合物が挙げられる。
また、本発明において、液晶用或いは固体撮像素子用カラーフィルタでアルカリ現像による画像形成に用いられる場合には、酸基及び/又は親水性基を有するバインダーポリマーを使用することが好ましく、このような目的で使用されるバインダーポリマーが有する好ましい酸基としては、カルボキシル基、スルホンアミド基、スルホン酸基、ホスホン酸基、フェノール基であり、現像性と感度の観点から好ましい酸価は0.1mmol/g~10mmol/gであり、より好ましくは0.2mmol/g~5mmol/g、最も好ましくは0.3mmol/g~3mmol/gである。
本発明の着色硬化性組成物は、必要に応じ、前記(B)オキシム型開始剤以外の、(B-2)他の光重合開始剤をさらに含有することができる。
本発明における(B-2)他の光重合開始剤は、光により分解し、前記(C)重合性化合物の重合を開始、促進する化合物であり、波長300~500nmの領域に吸収を有するものであることが好ましい。また、光重合開始剤は、単独で、又は2種以上を併用して用いることができる。
また、その他の好ましいスルホニウム塩としては、トリアリールスルホニウム塩の1つの置換基がクマリン、アントアキノン構造を有し、300nm以上に吸収を有するスルホニウム塩が挙げられる。別の好ましいスルホニウム塩としては、トリアリールスルホニウム塩が、アリロキシ基、アリールチオ基を置換基に有する300nm以上に吸収を有するスルホニウム塩が挙げられる。
本発明の着色硬化性組成物には、硬化皮膜の支持体や基板との密着性向上を目的として、密着性基を有する化合物を用いることができる。特に、本発明の着色硬化性組成物をカラーフィルタの着色領域の形成に用いる場合、着色領域と基板との密着性向上の観点から、このような化合物を用いることが好ましい。
このような化合物が有する密着性基とは、カラーフィルタに用いられる基板への密着性を有する置換基であればよいが、特にガラス基板またはシリコン基板への吸着性を有する化合物が好ましい。
好ましい密着性基は、スルホン酸基、ホスホン酸基、カルボキシル基などの酸基;これら酸のエステル基;これら酸の金属塩;これら金属のオニウム塩;アンモニウム基、ピリジニウム基などのオニウム基;アルコキシシリル基などの加水分解によりシラノール基を生成する置換基;フェノール性水酸基、N-オキシド基などの両性イオン性基;イミノ二酢酸などのキレート性基から選択される基であることが好ましい。
密着性の観点から好ましくは、アルコキシシリル基、及び/またはその加水分解した基が好ましい。
現像性を向上させる基として、ヘテロ原子を有する官能基が好ましく、アルコキシ基、アルコキシカルボニル基、カルボキシル基、水酸基、アミノ基、ウレタン基、アミド基、チオール基、スルホ基、ウレア基などが挙げられ、このような官能基を有する化合物を併用することが好ましい。
感度を向上させる基としては、メタクリル基、アクリル基、スチリル基、ビニルエーテル基、アリル基、環状アルケニル基、フリル基、オキセタン基、エポキシ基、テトラヒドロフラン基が好ましく挙げられ、感度の観点からメタクリル基、アクリル基、スチリル基を有する化合物を併用することが好ましい。
本発明の硬化性組成物は、ラジカル開始剤のラジカル発生効率の向上、感光波長の長波長化の目的で、増感剤を含有していてもよい。本発明に用いることができる増感剤としては、前記した光重合開始剤に対し、電子移動機構又はエネルギー移動機構で増感させるものが好ましい。
好ましい増感剤の例としては、以下の化合物類に属しており、かつ330nmから450nm域に吸収波長を有するものを挙げることができる。
例えば、多核芳香族類(例えば、フェナントレン、アントラセン、ピレン、ペリレン、トリフェニレン、9,10-ジアルコキシアントラセン)、キサンテン類(例えば、フルオレッセイン、エオシン、エリスロシン、ローダミンB、ローズベンガル)、チオキサントン類(イソプロピルチオキサントン、ジエチルチオキサントン、クロロチオキサントン)、シアニン類(例えばチアカルボシアニン、オキサカルボシアニン)、メロシアニン類(例えば、メロシアニン、カルボメロシアニン)、フタロシアニン類、チアジン類(例えば、チオニン、メチレンブルー、トルイジンブルー)、アクリジン類(例えば、アクリジンオレンジ、クロロフラビン、アクリフラビン)、アントラキノン類(例えば、アントラキノン)、スクアリウム類(例えば、スクアリウム)、アクリジンオレンジ、クマリン類(例えば、7-ジエチルアミノ-4-メチルクマリン)、ケトクマリン、フェノチアジン類、フェナジン類、スチリルベンゼン類、アゾ化合物、ジフェニルメタン、トリフェニルメタン、ジスチリルベンゼン類、カルバゾール類、ポルフィリン、スピロ化合物、キナクリドン、インジゴ、スチリル、ピリリウム化合物、ピロメテン化合物、ピラゾロトリアゾール化合物、ベンゾチアゾール化合物、バルビツール酸誘導体、チオバルビツール酸誘導体、アセトフェノン、ベンゾフェノン、チオキサントン、ミヒラーズケトンなどの芳香族ケトン化合物、N-アリールオキサゾリジノンなどのヘテロ環化合物などが挙げられる。更に欧州特許第568,993号明細書、米国特許第4,508,811号明細書、同5,227,227号明細書、特開2001-125255号公報、特開平11-271969号公報等に記載の化合物等などが挙げられる。
本発明の硬化性組成物中における増感剤の含有量は、深部への光吸収効率と開始分解効率の観点から、固形分換算で、0.1~20質量%であることが好ましく、0.5~15質量%がより好ましい。
本発明の硬化性組成物は、共増感剤を含有することも好ましい。本発明において共増感剤は、増感色素や開始剤の活性放射線に対する感度を一層向上させる、あるいは酸素による重合性化合物の重合阻害を抑制する等の作用を有する。
この様な共増感剤の例としては、アミン類、例えばM. R. Sanderら著「Journal of Polymer Society」第10巻3173頁(1972)、特公昭44-20189号公報、特開昭51-82102号公報、特開昭52-134692号公報、特開昭59-138205号公報、特開昭60-84305号公報、特開昭62-18537号公報、特開昭64-33104号公報、Research Disclosure 33825号記載の化合物等が挙げられ、具体的には、トリエタノールアミン、p-ジメチルアミノ安息香酸エチルエステル、p-ホルミルジメチルアニリン、p-メチルチオジメチルアニリン等が挙げられる。
本発明においては、硬化性組成物の製造中あるいは保存中において重合可能なエチレン性不飽和二重結合を有する化合物の不要な熱重合を阻止するために少量の熱重合防止剤を添加することが望ましい。
本発明に用いうる熱重合防止剤としては、ハイドロキノン、p-メトキシフェノール、ジ-t-ブチル-p-クレゾール、ピロガロール、t-ブチルカテコール、ベンゾキノン、4,4’-チオビス(3-メチル-6-t-ブチルフェノール)、2,2’-メチレンビス(4-メチル-6-t-ブチルフェノール)、N-ニトロソフェニルヒドロキシアミン第一セリウム塩等が挙げられる。
本発明の着色硬化性組成物には、熱重合成分を含有させることも有効である。必要によっては、塗膜の強度を上げるために、エポキシ化合物を添加することができる。エポキシ化合物としては、ビスフェノールA型、クレゾールノボラック型、ビフェニル型、脂環式エポキシ化合物などのエポキシ環を分子中に2個以上有する化合物である。例えばビスフェノールA型としては、エポトートYD-115、YD-118T、YD-127、YD-128、YD-134、YD-8125、YD-7011R、ZX-1059、YDF-8170、YDF-170など(以上東都化成製)、デナコールEX-1101、EX-1102、EX-1103など(以上ナガセ化成製)、プラクセルGL-61、GL-62、G101、G102(以上ダイセル化学製)の他に、これらの類似のビスフェノールF型、ビスフェノールS型も挙げることができる。またEbecryl 3700、3701、600(以上ダイセルユーシービー製)などのエポキシアクリレートも使用可能である。クレゾールノボラック型としては、エポトートYDPN-638、YDPN-701、YDPN-702、YDPN-703、YDPN-704など(以上東都化成製)、デナコールEM-125など(以上ナガセ化成製)、ビフェニル型としては3,5,3’,5’-テトラメチル-4,4’ジグリシジルビフェニルなど、脂環式エポキシ化合物としては、セロキサイド2021、2081、2083、2085、エポリードGT-301、GT-302、GT-401、GT-403、EHPE-3150(以上ダイセル化学製)、サントートST-3000、ST-4000、ST-5080、ST-5100など(以上東都化成製)などを挙げることができる。また1,1,2,2-テトラキス(p-グリシジルオキシフェニル)エタン、トリス(p-グリシジルオキシフェニル)メタン、トリグリシジルトリス(ヒドロキシエチル)イソシアヌレート、o-フタル酸ジグリシジルエステル、テレフタル酸ジグリシジルエステル、他にアミン型エポキシ樹脂であるエポトートYH-434、YH-434L、ビスフェノールA型エポキシ樹脂の骨格中にダイマー酸を変性したグリシジルエステル等も使用できる。
本発明の着色硬化性組成物は、塗布性を改良する観点から、各種の界面活性剤を用いて構成することが好ましく、フッソ系界面活性剤、ノニオン系、カチオン系、アニオン系の各種界面活性剤を使用できる。中でも、フッ素系界面活性剤、ノニオン系界面活性剤が好ましい。
具体的な市販品としては、例えば、メガファックF142D、同F172、同F173、同F176、同F177、同F183、同780、同781、同R30、同R08、同F-472SF、同BL20、同R-61、同R-90(大日本インキ(株)製)、フロラードFC-135、同FC-170C、同FC-430、同FC-431、Novec FC-4430(住友スリーエム(株)製)、アサヒガードAG7105,7000,950,7600、サーフロンS-112、同S-113、同S-131、同S-141、同S-145、同S-382、同SC-101、同SC-102、同SC-103、同SC-104、同SC-105、同SC-106(旭ガラス(株)製)、エフトップEF351、同352、同801、同802(JEMCO(株)製)などが挙げられる。
上記以外に、本発明の着色硬化性組成物には、各種の添加剤を用いることができる。添加剤の具体例としては、2-(3-t-ブチル-5-メチル-2-ヒドロキシフェニル)-5-クロロベンゾトリアゾール、アルコキシベンゾフェノン等の紫外線吸収剤、ポリアクリル酸ナトリウム等の凝集防止剤、ガラス、アルミナ等の充填剤;イタコン酸共重合体、クロトン酸共重合体、マレイン酸共重合体、部分エステル化マレイン酸共重合体、酸性セルロース誘導体、水酸基を有するポリマーに酸無水物を付加させたもの、アルコール可溶性ナイロン、ビスフェノールAとエピクロルヒドリンとから形成されたフェノキシ樹脂などのアルカリ可溶の樹脂などがある。
可塑剤としては例えばジオクチルフタレート、ジドデシルフタレート、トリエチレングリコールジカプリレート、ジメチルグリコールフタレート、トリクレジルホスフェート、ジオクチルアジペート、ジブチルセバケート、トリアセチルグリセリン等があり、結合剤を使用した場合、エチレン性不飽和二重結合を有する化合物と結合剤との合計質量に対し10質量%以下添加することができる。
本発明の着色硬化性組成物は、既述の(A)顔料分散液に(B)オキシムエステル系開始剤、(C)重合性化合物、及び、所望により併用されるアルカリ可溶性樹脂を(好ましくは溶剤と共に)含有させ、これに必要に応じて界面活性剤等の添加剤を混合し、各種の混合機、分散機を使用して混合分散する混合分散工程を経ることによって調製することができる。
なお、混合分散工程は、混練分散とそれに続けて行なう微分散処理からなるのが好ましいが、混練分散を省略することも可能である。
(1.微細化顔料の調製)
顔料と水溶性有機溶剤と水溶性無機塩類との混合物を、二本ロール、三本ロール、ボールミル、トロンミル、ディスパー、ニーダー、コニーダー、ホモジナイザー、ブレンダー、単軸もしくは2軸の押出機等の混練機を用いて、強い剪断力を与えながら顔料を摩砕した後、この混合物を水中に投入し、攪拌機等でスラリー状とする。次いで、このスラリーをろ過、水洗し、水溶性有機溶剤と水溶性無機塩を除去した後、乾燥し、微細化された顔料が得られる。
(a-1)顔料、(a-3)特定分散樹脂、(a-2)特定顔料誘導体、及び、溶剤を配合し、ビーズ分散を行なう。主として縦型もしくは横型のサンドグラインダー、ピンミル、スリットミル、超音波分散機等を使用し、0.01~1mmの粒径のガラス、ジルコニア等でできたビーズで微分散処理し、(A)顔料分散液を得る。また、顔料を微細化する処理を省くことも可能である。
なお、混練、分散についての詳細は、T.C.Patton著”Paint Flow and Pigment Dispersion”(1964年 John Wiley and Sons社刊)等に記載されている。
そして、上記のようにして得られた(A)顔料分散液に、(C)重合性化合物、(B)オキシムエステル系開始剤、及び、所望により併用される(B-2)他の重合開始剤、アルカリ可溶性樹脂などを添加して、本発明の着色硬化性組成物を得る。
以下、本発明のカラーフィルタについて、その製造方法(本発明のカラーフィルタの製造方法)を通じて詳述する。
以下、本発明の製造方法における各工程について説明する。
本発明のカラーフィルタは、既述の本発明の着色硬化性組成物を用いてガラスなどの基板上に形成されるものであり、本発明の着色硬化性組成物を直接若しくは他の層を介して基板上に例えばスリット塗布によって塗膜を形成した後、この塗膜を乾燥させ、パターン露光し、現像液を用いた現像処理を順次行なうことによって好適に作製することができる。これにより、液晶表示素子や固体撮像素子に用いられるカラーフィルタをプロセス上の困難性が少なく、高品質でかつ低コストに作製することができる。
前記基板としては、例えば、液晶表示素子等に用いられる無アルカリガラス、ソーダガラス、パイレックス(登録商標)ガラス、石英ガラス、及びこれらに透明導電膜を付着させたものや、固体撮像素子等に用いられる光電変換素子基板、例えばシリコン基板等、並びにプラスチック基板が挙げられる。これらの基板上には、通常、各画素を隔離するブラックマトリクスが形成されていたり、密着促進等のために透明樹脂層を設けたりしている。
プラスチック基板には、その表面にガスバリヤー層及び/又は耐溶剤性層を有していることが好ましい。このほかに、薄膜トランジスター(TFT)方式カラー液晶表示装置の薄膜トランジスター(TFT)が配置された駆動用基板(以下、「TFT方式液晶駆動用基板」という。)上にも本発明の着色硬化性組成物からなるパターン状皮膜を形成し、カラーフィルタを作成することができる。その際に使用されるフォトマスクには、画素を形成するためのパターンのほか、スルーホールあるいはコの字型の窪みを形成するためのパターンも設けられている。TFT方式液晶駆動用基板における基板としては、例えば、ガラス、シリコン、ポリカーボネート、ポリエステル、芳香族ポリアミド、ポリアミドイミド、ポリイミド等を挙げることができる。これらの基板には、所望により、シランカップリング剤等による薬品処理、プラズマ処理、イオンプレーティング、スパッタリング、気相反応法、真空蒸着等の適宜の前処理を施しておくこともできる。例えば、TFT方式液晶駆動用基板の表面上、あるいは該駆動基板の表面に窒化ケイ素膜等のパッシベーション膜を形成した基板等を挙げることができる。
なお、本発明の着色硬化性組成物を高解像度の固体撮像素子用カラーフィルタの形成に適用する場合には、膜厚は最も好ましくは0.4μm~2.0μmの範囲である。
本発明では、特に膜厚が0.4μm~1.0μm、更には0.45μm~0.8μmの着色領域を形成するのに、特に有効である。
プリベーク処理は、ホットプレート、オーブン等を用いて50~140℃の温度範囲で、好ましくは70~110℃程度であり、10~300秒の条件にて行なうことができる。高周波処理などを併用してもよい。高周波処理は単独でも使用可能である。
露光工程では、前記硬化性組成物層形成工程において形成された硬化性組成物層を、所定のマスクパターンを有するマスクを介して露光する。
本工程における塗布膜のパターン露光は、所定のマスクパターンを介して露光し、光照射された塗布膜部分だけを硬化させ、現像液で現像して、各色(3色あるいは4色)の画素からなるパターン状皮膜を形成することにより行うことができる。露光に際して用いることができる放射線としては、特に、g線、i線等の紫外線が好ましく用いられる。照射量は5~1500mJ/cm2が好ましく10~1000mJ/cm2がより好ましく、10~500mJ/cm2が最も好ましい。
現像処理では、露光後の未硬化部を現像液に溶出させ、硬化分のみを残存させる。現像温度としては、通常20~30℃であり、現像時間としては20~90秒である。
現像液としては、未硬化部における光硬化性の光硬化性組成物の塗膜を溶解する一方、硬化部を溶解しないものであれば、いずれのものも用いることができる。具体的には、種々の有機溶剤の組合せやアルカリ性の水溶液を用いることができる。
前記アルカリ性の水溶液としては、例えば、水酸化ナトリウム、水酸化カリウム、炭酸ナトリウム、炭酸水素ナトリウム、硅酸ナトリウム、メタ硅酸ナトリウム、アンモニア水、エチルアミン、ジエチルアミン、ジメチルエタノールアミン、テトラメチルアンモニウムヒドロキシド、テトラエチルアンモニウムヒドロキシド、コリン、ピロール、ピペリジン、1,8-ジアザビシクロ-[5,4,0]-7-ウンデセン等のアルカリ性化合物を、濃度が0.001~10質量%、好ましくは0.01~1質量%となるように溶解したアルカリ性水溶液が挙げられる。アルカリ性水溶液には、例えばメタノール、エタノール等の水溶性有機溶剤や界面活性剤等を適量添加することもできる。
現像方式は、デイップ方式、シャワー方式、スプレー方式などいずれでもよく、これにスウィング方式、スピン方式、超音波方式などを組み合わせてもよい。現像液に触れる前に、被現像面を予め水等で湿しておいて、現像むらを防ぐこともできる。また基板を傾斜させて現像することもできる。
固体撮像素子用カラーフィルタを作製する場合にはパドル現像を用いてもよい。
リンス工程は通常は純水で行うが、省液のために、最終洗浄で純水を用い、洗浄はじめは使用済の純水を使用したり、基板を傾斜させて洗浄したり、超音波照射を併用したりできる。
以上の操作を所望の色相数に合わせて各色毎に順次繰り返し行なうことにより、複数色の着色された硬化膜が形成されてなるカラーフィルタを作製することができる。
前記ブラックマトリックスは、顔料としてカーボンブラック、チタンブラックなどの黒色顔料を用いた本発明の着色硬化性組成物を露光、現像し、その後必要に応じて更にポストベークして膜の硬化を促進させることで形成できる。
なお、特に断りのない限り、「%」「部」は質量基準である。
反応容器にシクロヘキサノン800部を入れ、容器に窒素ガスを注入しながら100℃に加熱して、同温度で、スチレン60.0部、メタクリル酸80.0部、メチルメタクリレート68.0部、ブチルメタクリレート62.0部、アゾビスイソブチロニトリル9.0部の混合物を1時間かけて滴下して重合反応を行った。滴下後さらに100℃で3時間反応させた後、アゾビスイソブチロニトリル2.0部をシクロヘキサノン50部で溶解させたものを添加し、さらに100℃で1時間反応を続けて、重量平均分子量が約30,000、酸価が3.44mmol/gの特定分散樹脂1の溶液を得た。重量平均分子量は、ポリスチレンを標準物質としたゲルパーミエーションクロマトグラフィー法(GPC)により測定した。酸価は滴定により求めた。
室温まで冷却した後、樹脂溶液約2gをサンプリングして180℃、20分加熱乾燥して不揮発分を測定し、先に合成した樹脂溶液に不揮発分が20質量%になるようにシクロヘキサノンを添加して特定分散樹脂溶液1を調製した。
前記合成例1で得た特定分散樹脂溶液1にグリシジルメタクリレートを10質量部、テトラエチルアンモニウムブロミド1質量部を加え、90℃で2時間攪拌し、側鎖に二重結合性基を導入した重量平均分子量が約32,000、酸価が3.1mmol/gの特定分散樹脂2の溶液を得た。
特定分散樹脂2の溶液にシクロヘキサノン(溶媒)を添加して、不揮発分が20質量%の特定分散樹脂溶液2を調整した。
重量平均分子量10,000、酸価6.9mmol/gのポリアクリル酸/メタクリル酸メチル(重量比 50/50)100質量部を含むプロピレングリコールモノメチルエーテルアセテート溶液333質量部にグリシジルメタクリレートを50質量部、テトラエチルアンモニウムブロミド1質量部を加え、90℃で3時間攪拌し、側鎖に二重結合性基を導入した、重量平均分子量12,000、酸価1.95mmol/gの特定分散樹脂3の溶液を得た。
特定分散樹脂3の溶液にプロピレングリコールモノメチルエーテルアセテート(溶媒)を添加して、不揮発分が20質量%の特定分散樹脂溶液3を調整した。
1000ml三口フラスコに、1-メチル-2-ピロリドン100gを入れ、窒素気流下、90℃まで加熱した。これに、下記化合物(i-1)84g、メタクリル酸ベンジル33g、メタクリル酸14g、及びV-601(和光純薬製)5.2gを含む1-メチル-2-ピロリドン溶液100gを混合して得た溶液を、2時間かけて滴下した。滴下終了後、更に2時間撹拌した。その後、室温まで反応溶液を冷却した後、水7Lに投入し、高分子化合物を析出させた。析出した高分子化合物を濾取し、水で洗浄、乾燥し、高分子化合物131gを得た。
得られた高分子化合物を、ポリスチレンを標準物質としたゲルパーミエーションクロマトグラフィー法(GPC)により、重量平均分子量を測定した結果、11,800であった。また、滴定によりこの高分子化合物の酸価を求めたところ、69.6mgKOH/g(計算値69.7mgKOH/g)である。
1000ml三口フラスコに、プロピレングリコールモノメチルエーテル120gを入れ、窒素気流下、90℃まで加熱した。これに、メタクリル酸ベンジル75g、メタクリル酸40g、及びV-601(和光純薬社製)7.0gを含むN-メチルピロリドン溶液120gを混合して得た溶液を、2時間かけて滴下した。滴下終了後、更に2時間撹拌した。その後、室温まで反応溶液を冷却した後、水8L(リットル;以下同様)に投入し、高分子化合物を析出させた。析出した高分子化合物を濾取し、水で洗浄、乾燥し、高分子化合物(ii-1)114gを得た。
得られた高分子化合物(ii-1)を、ポリスチレンを標準物質としたゲルパーミエーションクロマトグラフィー法(GPC)により、重量平均分子量を測定した結果、12,000であった。また、滴定によりこの高分子化合物(ii-1)の酸価を求めたところ、258mgKOH/g(計算値260mgKOH/g)であり、正常に重合が行なわれたことが確認された。
得られた高分子化合物(ii-2)について、ポリスチレンを標準物質としたゲルパーミエーションクロマトグラフィー法(GPC)により、重量平均分子量を測定した結果、17,800であった。更に、滴定によりこの高分子化合物(ii-2)の酸価を求めたところ、128mgKOH/g(計算値129mgKOH/g)であった。
特定分散樹脂5について、ポリスチレンを標準物質としたゲルパーミエーションクロマトグラフィー法(GPC)により、重量平均分子量を測定した結果、20,100であった。更に、滴定によりこの高分子化合物の酸価を求めたところ、115mgKOH/g(計算値117mgKOH/g)であった。特定分散樹脂5をプロピレングリコールモノメチルエーテルに溶解させ、特定分散樹脂溶液5(固形分20%溶液)とした。
コンデンサー、撹拌機を備えた500mlの3つ口丸底フラスコに2,2-ビス(ヒドロキシメチル)ブタン酸8.2g(0.05モル)、下記ジオ-ル化合物(1)13.0g(0.05モル)をN,N-ジメチルアセトアミド100mlに溶解した。これに、4,4-ジフェニルメタンジイソシアネート25.5g(0.102モル)、ジブチル錫ジラウリレート0.1gを添加し、100℃にて、8時間加熱撹拌した。その後、N,N-ジメチルホルムアミド100ml及びメチルアルコール200mlにて希釈し30分撹拌した。反応溶液を水3リットル中に撹拌しながら投入し、白色の特定分散樹脂6を析出させた。この特定分散樹脂6を濾別し、水で洗浄後、真空下乾燥させることにより37gの特定分散樹脂6を得た。
ゲルパーミエーションクロマトグラフィー(GPC)にて分子量を測定したところ、重量平均分子量(ポリスチレン標準)で95,000、酸価1.06mmol/gであった。
コンデンサー、撹拌機を備えた500mlの3つ口丸底フラスコに2,2-ビス(ヒドロキシメチル)ブタン酸5.9g(0.04モル)、下記ジオール化合物(2)15.9g(0.06モル)をN,N-ジメチルアセトアミド100mlに溶解した。これに、4,4-ジフェニルメタンジイソシアネート20.4g(0.082モル)、1,6-ヘキサメチレンジイソシアネート3.4g(0.02モル)、ジブチル錫ジラウリレート0.3gを添加し、100℃にて、8時間加熱撹拌した。その後、N,N-ジメチルホルムアミド100ml及びメチルアルコール200mlにて希釈し30分撹拌した。反応溶液を水3リットル中に撹拌しながら投入し、白色の特定分散樹脂7を析出させた。この特定分散樹脂7を濾別し、水で洗浄後、真空下乾燥させることにより34gの特定分散樹脂7を得た。
ゲルパーミエーションクロマトグラフィー(GPC)にて分子量を測定したところ、重量平均分子量(ポリスチレン標準)で50,000 酸価0.88mmol/gであった。
コンデンサー、撹拌機を備えた500mlの3つ口丸底フラスコに2,2-ビス(ヒドロキシメチル)プロピオン酸5.4g(0.04モル)、下記ジオ-ル化合物(3)15.6g(0.06モル)をN,N-ジメチルアセトアミド100mlに溶解した。これに、ナフタレンジイソシアネート21.4g(0.102モル)、ジブチル錫ジラウリレート0.1gを添加し、100℃にて、4時間加熱撹拌した。その後、N,N-ジメチルホルムアミド100ml及びメチルアルコール200mlにて希釈し30分撹拌した。反応溶液を水3リットル中に撹拌しながら投入し、白色の特定分散樹脂8を析出させた。この特定分散樹脂8を濾別し、水で洗浄後、真空下乾燥させることにより34gの特定分散樹脂8を得た。
ゲルパーミエーションクロマトグラフィー(GPC)にて分子量を測定したところ、重量平均分子量(ポリスチレン標準)で28,000 酸価0.93mmol/gであった。
メチルメタクリレートとメタクリル酸を80/20のモル比率で、公知の方法によりラジカル重合で得られた重量平均分子量12,000、酸価2.06mmol/gの樹脂を比較用樹脂1とした。
ベンジルメタクリレートとメタクリル酸を60/40のモル比率で、公知の方法によりラジカル重合で得られた重量平均分子量30,000、酸価2.85mmol/gの樹脂を比較用樹脂2とした。
<顔料分散液の調製1>
下記組成(1)の成分を混合し、ホモジナイザーを用いて回転数3,000r.p.m.で3時間撹拌して混合し、顔料を含む混合溶液(A~D、H-1~H-2)を調製した。
〔組成(1)〕
・C.I.ピグメントレッド254 80部
・C.I.ピグメントイエロー139 20部
・特定分散樹脂または比較用樹脂(下記表1に記載) 275部
・1-メトキシ-2-プロピルアセテート 630部
・特定顔料誘導体または比較化合物(下記表1に記載) 8部
得られた本発明に係る顔料分散液(A~D)及び比較顔料分散液(H-1~H-2)について下記の評価を行った。
(1)粘度の測定、評価
得られた顔料分散組成物について、E型粘度計を用いて、分散直後の顔料分散液の粘度η1および分散後(室温25℃~28℃にて)1週間経過した後の顔料分散液の粘度η2を測定し、差を算出して増粘の程度を評価した。1週間後の粘度η2と分散後の粘度η1との差が5(mPa・s)以下であれば、分散安定性が良好であることを示す。結果を上記表1に併記した。
表1に明らかなように、(a-2)特定顔料誘導体および(a-3)特定分散樹脂を含んで調製された顔料分散液A~Dは、いずれも分散安定性に優れたものであった。
一方、酸型の誘導体と特定樹脂のみを用いた顔料分散液H-1は粘度変化が大きく安定性が低いことが解る。
〔下塗り層付シリコン基板の作製〕
下記組成(2)の成分を混合して溶解し、下塗り層用にレジスト液を調製した。
〈組成(2)〉
・プロピレングリコールモノメチルエーテルアセテート 19.20部
(PGMEA:溶剤)
・乳酸エチル 36.67部
・バインダーポリマー 30.51部
〔メタクリル酸ベンジル/メタクリル酸/メタクリル酸-2-ヒドロキシ
エチル共重合体(モル比=60/22/18)の40%PGMEA溶液〕
・ジペンタエリスリトールヘキサアクリレート 12.20部
(光重合性化合物)
・重合禁止剤(p-メトキシフェノール) 0.0061部
・フッ素系界面活性剤 0.83部
(F-475、大日本インキ化学工業(株)製)
・光重合開始剤 0.586部
(TAZ-107:トリハロメチルトリアジン系の光重合開始剤、みどり化学社製)
前記分散処理して得た顔料分散液に、更に下記成分を添加し、撹拌混合して硬化性組成物溶液を調製した。
・顔料分散液〔表2に記載の分散液〕 100質量部
・DPHA(日本化薬社製) 3.5質量部
・重合開始剤〔表2記載のオキシムエステル系開始剤又は比較開始剤〕
0.6質量部
・アリルメタクリレート/メタクリル酸 〔mol比、73/27、
重量平均分子量 57,000〕 1.0質量部
・界面活性剤(商品名:テトラニック150R1、BASF社) 0.2質量部
・溶剤:PGMEA 100質量部
-パターンの形成と感度の評価-
前述のように調製した実施例1~5および比較例1~2の着色剤含有硬化性組成物を、前記下塗り層付シリコン基板上に塗布し、硬化性組成物層(塗布膜)を形成した。そして、この塗布膜の乾燥膜厚が0.9μmになるように、100℃のホットプレートを用いて120秒間加熱処理(プリベーク)を行なった。
次いで、i線ステッパー露光装置FPA-3000i5+(Canon(株)製)を使用して365nmの波長でパターンが1.4μm四方のIslandパターンマスクを介する露光によってマスクパターンと同サイズの1.4μm四方のIslandパターンが得られるように50~1600mJ/cm2の種々の露光量で露光した。ここで、光照度系により50mJ/cm2露光量を測定して、その露光量を基準とし、照射時間を変更させて積算露光量(mJ/cm2)を求めて感度とした。この数値が小さいほど、低露光量で、高解像度の微細パターンを形成することができ、高感度であることを示す。結果を上記表2に併記した。
その後、照射された塗布膜が形成されているシリコンウエハー基板をスピン・シャワー現像機(DW-30型、(株)ケミトロニクス製)の水平回転テーブル上に載置し、CD-2000(富士フイルムエレクトロニクスマテリアルズ(株)製)を用いて23℃で60秒間パドル現像を行ない、シリコンウエハー基板に着色パターンを形成した。
他方、(B)オキシム型開始剤に代えてα-アミノアセトフェノン系化合物(IH-1)を用いた比較例2では十分な感度が得られなかった。
他方、(a-2)特定分散樹脂を用いない比較例1でも、感度が劣ることがわかる。
<顔料分散液の調製2>
下記組成(3)の成分を混合し、2本ロールで混練分散処理し、顔料混合物を得た。
〔組成(3)〕
・C.I.ピグメントグリーン36 50質量部
・C.I.ピグメントイエロー150 50質量部
・特定分散樹脂または比較用樹脂(下記表3に記載) 200質量部
・ソルスパース32000GR(日本ルーブリゾール社製;ポリエステル系分散剤)
20質量部
・溶剤:プロピレングリコールメチルエーテルアセテート 520質量部
・特定顔料誘導体または比較化合物(下記表3に記載) 5質量部
・溶剤 350質量部
〔プロピレングリコールメチルエーテルアセテート:PGMEA〕
得られた本発明に係る顔料分散液(E~G)および比較顔料分散液(H-3~H-4)について、顔料分散液A~Dと同様にして粘度の測定および評価をした。結果を上記表3に併記した。
表3に明らかなように、(a-2)特定顔料誘導体および(a-3)特定分散樹脂を含んで調製された顔料分散液E~Gは、分散安定性に優れたものであった。
前記分散処理して得た顔料分散液に、更に下記成分を添加し、撹拌混合して硬化性組成物溶液を調製した。
・顔料分散液(下記表4に記載) 100質量部
・ペンタエリスリトールテトラアクリレート 3.8質量部
・重合開始剤 0.5質量部
(下記表4に記載のオキシムエステル系開始剤または比較開始剤)
・上記特定分散樹脂4(粉体) 0.7質量部
・界面活性剤(商品名:テトラニック150R1、BASF社) 0.2質量部
・(3-アクリロキシプロピル)メチルジメトキシシラン 0.05質量部
・溶剤:PGMEA 100質量部
次いで、i線ステッパー露光装置FPA-3000i5+(Canon(株)製)を使用して365nmの波長でパターンが1.4μm四方のIslandパターンマスクを介する露光によってマスクパターンと同サイズの1.4μm四方のIslandパターンが得られるように50~1600mJ/cm2の種々の露光量で露光した。ここで、光照度系により50mJ/cm2露光量を測定して、その露光量を基準とし、照射時間を変更させて積算露光量(mJ/cm2)を求めて感度とした。この数値が小さいほど、低露光量で、高解像度の微細パターンを形成することができ、高感度であることを示す。結果を上記表4に併記した。
その後、照射された塗布膜が形成されているシリコンウエハー基板をスピン・シャワー現像機(DW-30型、(株)ケミトロニクス製)の水平回転テーブル上に載置し、CD-2000(富士フイルムエレクトロニクスマテリアルズ(株)製)を用いて23℃で60秒間パドル現像を行ない、シリコンウエハー基板に着色パターンを形成した。
<顔料分散組成物の調製>
下記組成(5)の成分を混合し、ホモジナイザーを用いて回転数3,000r.p.m.で3時間撹拌して混合し、顔料を含む混合溶液(H~K、H-5~H-6)を調製した。
〔組成(5)〕
・C.I.ピグメントレッド254 50部
・C.I.ピグメントレッド177 50部
・特定分散樹脂(下記表5に記載) 200部
・BYK111(ビックケミー社製;リン酸エステル系分散剤) 10部
・特定顔料誘導体または比較化合物(下記表5に記載) 4部
・1-メトキシ-2-プロピルアセテート 660部
得られた顔料分散組成物(H~K)及び比較顔料分散液(H-5~H-6)について、顔料分散液A~Dと同様にして分散安定性を評価した。結果を前記表5に併記する。
表5に明らかなように、(a-2)特定顔料誘導体および(a-3)特定分散樹脂を含んで調製された顔料分散液H~Kは、分散安定性に優れたものであった。
<硬化性組成物(塗布液)の調製>
前記分散処理して得た顔料分散液に、更に下記成分を添加し、撹拌混合して硬化性組成物溶液を調製した。
・顔料分散液(下記表6に記載) 100質量部
・DPHA(日本化薬社製) 3.8質量部
・重合開始剤 1.5質量部
(下記表6に記載のオキシムエステル系開始剤または比較開始剤)
・ベンジルメタクリレート/メタクリル酸
(mol比、70/30、重量平均分子量70,000) 0.7質量部
・界面活性剤(商品名:テトラニック150R1、BASF社) 0.2質量部
・溶剤:PGMEA 100質量部
3-2.硬化性組成物層の形成
上記顔料を含有する硬化性組成物をレジスト溶液として、550mm×650mmのガラス基板に下記条件でスリット塗布した後、10分間そのままの状態で待機させ、真空乾燥とプレベーク(prebake)(100℃80秒)を施して硬化性組成物塗膜(硬化性組成物層)を形成した。
(スリット塗布条件)
塗布ヘッド先端の開口部の間隙:50μm
塗布速度:100mm/秒
基板と塗布ヘッドとのクリヤランス:150μm
塗布厚(乾燥厚):2μm
塗布温度:23℃
その後、2.5kwの超高圧水銀灯を用いて光硬化性塗布膜を、線幅20μmのテスト用フォトマスクを用いてパターン状に露光し、露光後、塗布膜の全面を富士フイルムエレクトロニクスマテリアルズ社製 アルカリ現像液 CDK-1、の1%水溶液で被い、40秒間静止した。
静止後、純水をシャワー状に噴射して現像液を洗い流し、かかる光硬化処理及び現像処理を施した塗布膜を220℃のオーブンにて1時間加熱した(ポストベーク)。これにより、ガラス基板上に樹脂皮膜からなる着色領域を形成し、カラーフィルタの着色パターンとした。
上記で調整された着色硬化性組成物を用いてガラス基板上に形成された硬化性組成物塗布膜(着色層)の硬化に必要な露光量を実施例1と同様に測定し、感度の基準とした。この数値が小さいほど高感度であることを示す。結果を上記表6に併記した。
また、別途形成した着色層(20μm四方)を100mJ/cm2の露光量で露光したのち、上記条件で、現像、ポストベイクを行い、形成された着色領域及びその周辺部を実施例1と同様の条件で観察し、非画像部の残渣の有無を確認した。結果を上記表6に併記した。
上記硬化性組成物について、顔料のみを、明細書に記載の青色顔料と緑色顔料とに変更した他は同様にして緑色、青色の着色硬化性組成物を調整した。
上記基板にまず、常法によりブラックマトリックスを形成し、赤色硬化性組成物で実施したのと同様にして20×20μmの赤色、緑色、青色パターンを順次形成して液晶表示素子用のカラーフィルターを作製した。得られたカラーフィルターを液晶表示素子に組み込んだところ、該液晶表示素子は、色特性、コントラストに優れることが確認された。
上記硬化性組成物について、顔料のみを青色顔料と緑色顔料とに変更した他は同様にして緑色、青色の着色硬化性組成物を調整した。
上記基板にまず、赤色硬化性組成物で実施したのと同様にして1.6×1.6μmの赤色、緑色、青色パターンを順次形成して固体撮像素子用のカラーフィルターを作製した。得られたカラーフィルターを固体撮像素子に組み込んだところ、該固体撮像素子は、高解像度で、色分離性に優れることが確認された。
Claims (6)
- (A)(a-1)顔料、(a-2)分子内に顔料母核構造とアミノ基とを有する化合物、および(a-3)酸基と重合性基を有する樹脂、を含む顔料分散液と、(B)オキシムエステル系開始剤と、(C)重合性化合物と、を含有する着色硬化性組成物。
- 前記(a-2)分子内に顔料母核構造とアミノ基とを有する化合物が、アミド構造及びウレア構造の少なくとも1つを有する化合物であることを特徴とする請求項1に記載の着色硬化性組成物。
- 前記(a-2)分子内に顔料母核構造とアミノ基とを有する化合物が、ヘテロ環構造を有する化合物であることを特徴とする請求項1に記載の着色硬化性組成物。
- カラーフィルタの着色領域形成に用いられることを特徴とする請求項1に記載の着色硬化性組成物。
- 請求項4に記載の着色硬化性組成物を用いて形成された着色領域を有することを特徴とするカラーフィルタ。
- 請求項5に記載のカラーフィルタを備えることを特徴とする固体撮像素子。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2009801081785A CN101960380B (zh) | 2008-03-10 | 2009-03-05 | 着色固化性组合物、滤色器和固体摄像元件 |
| EP09719268.6A EP2270593B1 (en) | 2008-03-10 | 2009-03-05 | Colored curable composition, color filter and solid-state imaging device |
| US12/921,648 US8303860B2 (en) | 2008-03-10 | 2009-03-05 | Colored curable composition, color filter and solid-state imaging device |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008059741A JP5448352B2 (ja) | 2008-03-10 | 2008-03-10 | 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 |
| JP2008-059741 | 2008-03-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009113447A1 true WO2009113447A1 (ja) | 2009-09-17 |
Family
ID=41065116
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/054216 WO2009113447A1 (ja) | 2008-03-10 | 2009-03-05 | 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8303860B2 (ja) |
| EP (1) | EP2270593B1 (ja) |
| JP (1) | JP5448352B2 (ja) |
| KR (1) | KR101622996B1 (ja) |
| CN (1) | CN101960380B (ja) |
| WO (1) | WO2009113447A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102053494A (zh) * | 2009-10-28 | 2011-05-11 | 住友化学株式会社 | 着色感光性树脂组合物 |
| JP2017187587A (ja) * | 2016-04-05 | 2017-10-12 | 東洋インキScホールディングス株式会社 | カラーフィルタ用着色組成物およびカラーフィルタ |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5425575B2 (ja) | 2009-09-18 | 2014-02-26 | ダイハツ工業株式会社 | 火花点火式内燃機関の燃焼状態判定方法 |
| KR102105622B1 (ko) * | 2011-06-09 | 2020-04-29 | 스미또모 가가꾸 가부시키가이샤 | 착색 조성물 |
| JP5789847B2 (ja) * | 2011-10-12 | 2015-10-07 | 東洋インキScホールディングス株式会社 | カラーフィルタ用着色組成物、およびカラーフィルタ |
| EP2602654B1 (en) * | 2011-12-08 | 2023-04-19 | Essilor International | Ophthalmic filter |
| EP2602653B1 (en) | 2011-12-08 | 2020-09-16 | Essilor International | Method of determining the configuration of an ophthalmic filter |
| JP6192968B2 (ja) * | 2012-04-10 | 2017-09-06 | 住友化学株式会社 | 着色剤分散液 |
| JP5826135B2 (ja) * | 2012-08-23 | 2015-12-02 | 富士フイルム株式会社 | 着色感放射線性組成物、これを用いたカラーフィルタ |
| KR102281546B1 (ko) * | 2013-08-05 | 2021-07-27 | 도쿄 오카 고교 가부시키가이샤 | 안료 분산액, 그것을 포함하는 감광성 수지 조성물 및 분산 조제 |
| CN103901725B (zh) * | 2014-04-19 | 2017-07-28 | 长兴电子(苏州)有限公司 | 一种光固化树脂组合物 |
| JP6380723B1 (ja) * | 2017-02-15 | 2018-08-29 | 三菱ケミカル株式会社 | 感光性着色組成物、硬化物、着色スペーサー、画像表示装置 |
| JP7472521B2 (ja) | 2020-01-08 | 2024-04-23 | artience株式会社 | 感光性着色組成物、カラーフィルタ、および画像表示装置 |
| TWI767248B (zh) * | 2020-06-10 | 2022-06-11 | 新應材股份有限公司 | 白色感光性樹脂組成物、白色隔壁、光轉換層以及光發射裝置 |
| CN113777882A (zh) * | 2020-06-10 | 2021-12-10 | 新应材股份有限公司 | 白色感光性树脂组合物、白色隔壁、光转换层以及光发射装置 |
Citations (195)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US161811A (en) | 1875-04-06 | Improvement in mechanisms for feeding heel-stiffeners or counter-blanks | ||
| US339049A (en) | 1886-03-30 | Sole-edge-burnishing | ||
| US410201A (en) | 1889-09-03 | Bent for suspension-bridges | ||
| US2833827A (en) | 1955-01-17 | 1958-05-06 | Bayer Ag | Tri (3, 5-di lower alkyl-4-hydroxy phenyl)-sulfonium chlorides and method of preparing same |
| US2848328A (en) | 1954-06-16 | 1958-08-19 | Eastman Kodak Co | Light sensitive diazo compound and binder composition |
| US2852379A (en) | 1955-05-04 | 1958-09-16 | Eastman Kodak Co | Azide resin photolithographic composition |
| US2940853A (en) | 1958-08-21 | 1960-06-14 | Eastman Kodak Co | Azide sensitized resin photographic resist |
| US3479185A (en) | 1965-06-03 | 1969-11-18 | Du Pont | Photopolymerizable compositions and layers containing 2,4,5-triphenylimidazoyl dimers |
| JPS4643946B1 (ja) | 1967-11-09 | 1971-12-27 | ||
| JPS4836281A (ja) | 1971-09-03 | 1973-05-28 | ||
| JPS4864183A (ja) | 1971-12-09 | 1973-09-05 | ||
| JPS4841708B1 (ja) | 1970-01-13 | 1973-12-07 | ||
| JPS4842965B1 (ja) | 1968-04-02 | 1973-12-15 | ||
| JPS4943191B1 (ja) | 1969-07-11 | 1974-11-19 | ||
| US3905815A (en) | 1971-12-17 | 1975-09-16 | Minnesota Mining & Mfg | Photopolymerizable sheet material with diazo resin layer |
| JPS5137193A (ja) | 1974-09-25 | 1976-03-29 | Toyo Boseki | |
| JPS5182102A (en) | 1974-12-10 | 1976-07-19 | Basf Ag | Insatsuinki oyobi hifukuzairyonotameno hikarijugokanonaketsugozai |
| JPS5147334B1 (ja) | 1970-11-02 | 1976-12-14 | ||
| JPS5230490B2 (ja) | 1972-03-21 | 1977-08-09 | ||
| JPS52134692A (en) | 1976-05-04 | 1977-11-11 | Fuji Photo Film Co Ltd | Photopolymerizable composition |
| JPS53702A (en) | 1975-12-23 | 1978-01-06 | Dynachem Corp | Adhesion promoting agent for polymerized film |
| US4069055A (en) | 1974-05-02 | 1978-01-17 | General Electric Company | Photocurable epoxy compositions containing group Va onium salts |
| US4069056A (en) | 1974-05-02 | 1978-01-17 | General Electric Company | Photopolymerizable composition containing group Va aromatic onium salts |
| JPS5492723A (en) | 1977-12-30 | 1979-07-23 | Somar Mfg | Photosensitive material and use |
| DE2904626A1 (de) | 1978-02-08 | 1979-08-09 | Minnesota Mining & Mfg | Triarylsulfoniumkomplexsalze, verfahren zu ihrer herstellung und diese salze enthaltende photopolymerisierbare gemische |
| JPS5425957B2 (ja) | 1974-10-04 | 1979-08-31 | ||
| JPS5434327B1 (ja) | 1970-12-28 | 1979-10-26 | ||
| JPS5532070A (en) | 1978-08-29 | 1980-03-06 | Fuji Photo Film Co Ltd | Photosensitive resin composition |
| JPS5534414B2 (ja) | 1972-06-02 | 1980-09-06 | ||
| JPS55500806A (ja) | 1978-10-19 | 1980-10-16 | ||
| JPS5617654B2 (ja) | 1970-12-28 | 1981-04-23 | ||
| JPS5675643A (en) | 1979-11-07 | 1981-06-22 | Hoechst Ag | Photopolymerizable mixture and photopolymerizable copying material using same |
| US4311783A (en) | 1979-08-14 | 1982-01-19 | E. I. Du Pont De Nemours And Company | Dimers derived from unsymmetrical 2,4,5,-triphenylimidazole compounds as photoinitiators |
| JPS57196231A (en) | 1981-05-20 | 1982-12-02 | Hoechst Ag | Mixture able to be polymerized by radiation and copying material mainly composed thereof |
| JPS5812577B2 (ja) | 1973-12-21 | 1983-03-09 | ヘキスト アクチエンゲゼルシヤフト | 光重合可能な複写材料 |
| JPS5849860B2 (ja) | 1973-12-07 | 1983-11-07 | ヘキスト アクチェンゲゼルシャフト | コウジユウゴウセイフクシヤザイリヨウ |
| JPS595240A (ja) | 1982-06-21 | 1984-01-12 | ヘキスト・アクチエンゲゼルシヤフト | 放射線重合可能な混合物 |
| JPS595241A (ja) | 1982-06-21 | 1984-01-12 | ヘキスト・アクチエンゲゼルシヤフト | 放射線重合可能な混合物 |
| JPS5944615A (ja) | 1982-09-07 | 1984-03-13 | Furuno Electric Co Ltd | ジヤイロ装置 |
| EP0104143A1 (de) | 1982-09-18 | 1984-03-28 | Ciba-Geigy Ag | Diaryljodosylsalze enthaltende photopolymerisierbare Zusammensetzungen |
| JPS5953836A (ja) | 1982-09-21 | 1984-03-28 | Fuji Photo Film Co Ltd | 感光性平版印刷版 |
| JPS5971048A (ja) | 1982-10-18 | 1984-04-21 | Mitsubishi Chem Ind Ltd | 光重合系感光性組成物 |
| JPS59138205A (ja) | 1983-01-20 | 1984-08-08 | チバ−ガイギ− アクチエンゲゼルシヤフト | 被覆組成物およびその硬化方法 |
| JPS59152396A (ja) | 1983-02-11 | 1984-08-31 | チバ−ガイギ− アクチエンゲゼルシヤフト | メタロセン,その製造方法およびメタロセンを含む光重合性組成物 |
| US4508811A (en) | 1983-01-17 | 1985-04-02 | U.S. Philips Corporation | Recording element having a pyrylium or thiopyrylium-squarylium dye layer and new pyrylium or thiopyrylium-squarylium compounds |
| JPS6084305A (ja) | 1983-08-30 | 1985-05-13 | バスフ アクチェン ゲゼルシャフト | 光重合可能な混合物 |
| JPS60208748A (ja) | 1984-04-02 | 1985-10-21 | Hitachi Chem Co Ltd | 感光性樹脂組成物及びこれを用いた積層体 |
| JPS60214354A (ja) | 1984-04-10 | 1985-10-26 | Mitsubishi Rayon Co Ltd | 光重合性樹脂組成物 |
| JPS60239736A (ja) | 1984-05-14 | 1985-11-28 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS6122048A (ja) | 1984-06-08 | 1986-01-30 | ヘキスト・アクチエンゲゼルシヤフト | 重合可能な化合物、その製法、およびこれを含有する放射線感性複写層 |
| JPS61151197A (ja) | 1984-12-20 | 1986-07-09 | チバ‐ガイギー アーゲー | チタノセン類およびこれらのチタノセン類を含有する照射重合開始剤 |
| JPS61166544A (ja) | 1985-01-18 | 1986-07-28 | Fuji Photo Film Co Ltd | 光可溶化組成物 |
| JPS61169837A (ja) | 1985-01-22 | 1986-07-31 | Fuji Photo Film Co Ltd | 光可溶化組成物 |
| JPS61169835A (ja) | 1985-01-22 | 1986-07-31 | Fuji Photo Film Co Ltd | 光可溶化組成物 |
| US4622286A (en) | 1985-09-16 | 1986-11-11 | E. I. Du Pont De Nemours And Company | Photoimaging composition containing admixture of leuco dye and 2,4,5-triphenylimidazolyl dimer |
| JPS6218537A (ja) | 1985-07-16 | 1987-01-27 | ザ・ミ−ド・コ−ポレ−シヨン | 共開始剤として第三アミンを含有する感光性マイクロカプセルを用いた造像材料 |
| JPS6258241A (ja) | 1985-09-09 | 1987-03-13 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS62143044A (ja) | 1985-11-20 | 1987-06-26 | サイカラー・インコーポレーテッド | 染料−ほう素化合物錯体を含有する光硬化性組成物およびその組成物を使用する感光性材料 |
| JPS62150242A (ja) | 1985-11-20 | 1987-07-04 | サイカラー・インコーポレーテッド | イオン性染料化合物を開始剤として含む感光性材料 |
| DE3604581A1 (de) | 1986-02-14 | 1987-08-20 | Basf Ag | 4-acylbenzylsulfoniumsalze, ihre herstellung sowie sie enthaltende photohaertbare gemische und aufzeichnungsmaterialien |
| DE3604580A1 (de) | 1986-02-14 | 1987-08-20 | Basf Ag | Haertbare mischungen, enthaltend n-sulfonylaminosulfoniumsalze als kationisch wirksame katalysatoren |
| JPS6239417B2 (ja) | 1978-05-20 | 1987-08-22 | Hoechst Ag | |
| JPS6239418B2 (ja) | 1978-05-20 | 1987-08-22 | Hoechst Ag | |
| JPS62212401A (ja) | 1986-03-14 | 1987-09-18 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPS6341484A (ja) | 1986-08-01 | 1988-02-22 | チバ−ガイギ− ア−ゲ− | チタノセン、それらの製造法およびそれらを含有する組成物 |
| JPS6370243A (ja) | 1986-09-11 | 1988-03-30 | Fuji Photo Film Co Ltd | 感光性組成物 |
| US4760013A (en) | 1987-02-17 | 1988-07-26 | International Business Machines Corporation | Sulfonium salt photoinitiators |
| JPS63260909A (ja) | 1987-03-28 | 1988-10-27 | ヘキスト・アクチエンゲゼルシヤフト | 光重合性混合物及びこの混合物から製造される記録材料 |
| JPS63277653A (ja) | 1987-03-28 | 1988-11-15 | ヘキスト・アクチエンゲゼルシヤフト | 重合可能な化合物、これを含有する放射線重合可能な混合物及び放射線重合可能な記録材料 |
| JPS63287947A (ja) | 1987-05-21 | 1988-11-25 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS63287944A (ja) | 1987-05-21 | 1988-11-25 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS63298339A (ja) | 1987-05-29 | 1988-12-06 | Fuji Photo Film Co Ltd | 感光性組成物 |
| EP0297442A1 (de) | 1987-07-01 | 1989-01-04 | BASF Aktiengesellschaft | Sulfoniumsalze mit säurelabilen Gruppierungen |
| EP0297443A2 (de) | 1987-07-01 | 1989-01-04 | BASF Aktiengesellschaft | Strahlungsempfindliches Gemisch für lichtempfindliche Beschichtungsmaterialien |
| JPS6433104A (en) | 1987-06-25 | 1989-02-03 | Ciba Geigy Ag | Photopolymerizable composition |
| US4808501A (en) | 1985-10-15 | 1989-02-28 | Polaroid Corporation, Patent Dept. | Method for manufacturing an optical filter |
| JPS6490403A (en) | 1987-09-30 | 1989-04-06 | Konishiroku Photo Ind | Color mosaic filter |
| JPS6491102A (en) | 1987-10-01 | 1989-04-10 | Konishiroku Photo Ind | Color mosaic filter |
| JPH0194301A (ja) | 1987-10-06 | 1989-04-13 | Konica Corp | カラーモザイクフィルター |
| JPH01105238A (ja) | 1987-03-28 | 1989-04-21 | Hoechst Ag | 光重合可能な混合物および光重合可能な記録材料 |
| JPH01152109A (ja) | 1987-12-09 | 1989-06-14 | Toray Ind Inc | 光重合性組成物 |
| JPH01165613A (ja) | 1987-11-16 | 1989-06-29 | Hoechst Ag | 重合可能な化合物、その放射線重合性混合物および放射線重合記録材料 |
| JPH0140337B2 (ja) | 1979-12-29 | 1989-08-28 | Hoechst Ag | |
| JPH0140336B2 (ja) | 1979-12-29 | 1989-08-28 | Hoechst Ag | |
| JPH01271741A (ja) | 1988-04-25 | 1989-10-30 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPH01304453A (ja) | 1988-06-02 | 1989-12-08 | Toyobo Co Ltd | 光重合性組成物 |
| JPH02249A (ja) | 1987-12-01 | 1990-01-05 | Ciba Geigy Ag | チタノセンおよびそれを含有する光重合性組成物 |
| JPH024705A (ja) | 1988-03-24 | 1990-01-09 | Dentsply Internatl Inc | 光硬化性組成物用チタナート開始剤 |
| JPH0225493A (ja) | 1988-05-21 | 1990-01-26 | Hoechst Ag | アルケニルホスホン酸エステルおよびアルケニルホスフイン酸エルテル、その製法並びに当該化合物を含有する放射線重合性混合物および記録材料 |
| JPH0216765B2 (ja) | 1980-09-29 | 1990-04-18 | Hoechst Ag | |
| JPH02127602A (ja) | 1988-10-24 | 1990-05-16 | Polaroid Corp | カラーフィルターの製造方法 |
| EP0370693A2 (en) | 1988-11-21 | 1990-05-30 | Eastman Kodak Company | Novel onium salts and the use thereof as photoinitiators |
| JPH02150848A (ja) | 1988-12-02 | 1990-06-11 | Hitachi Ltd | 光退色性放射線感応性組成物およびそれを用いたパターン形成法 |
| US4933377A (en) | 1988-02-29 | 1990-06-12 | Saeva Franklin D | Novel sulfonium salts and the use thereof as photoinitiators |
| JPH0232293B2 (ja) | 1980-12-22 | 1990-07-19 | Hoechst Ag | |
| JPH02226149A (ja) | 1988-12-22 | 1990-09-07 | Hoechst Ag | 光重合性化合物、それを含む光重合性混合物及びそれから製造された光重合性複写材料 |
| EP0390214A2 (en) | 1989-03-31 | 1990-10-03 | E.F. Johnson Company | Method and apparatus for a distributive wide area network for a land mobile transmission trunked communication system |
| JPH02296514A (ja) | 1989-05-12 | 1990-12-07 | Matsushita Electric Ind Co Ltd | 車両用サスペンション制御装置 |
| US5059500A (en) | 1990-10-10 | 1991-10-22 | Polaroid Corporation | Process for forming a color filter |
| JPH04249549A (ja) | 1990-12-28 | 1992-09-04 | Orient Chem Ind Ltd | 着色樹脂及びその関連技術 |
| JPH04365049A (ja) | 1991-06-12 | 1992-12-17 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPH0536581A (ja) | 1991-07-30 | 1993-02-12 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPH0583588A (ja) | 1991-09-24 | 1993-04-02 | Omron Corp | 画像処理装置 |
| JPH05142772A (ja) | 1991-11-26 | 1993-06-11 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| US5227227A (en) | 1990-03-05 | 1993-07-13 | Johnson & Johnson Inc. | Non-woven fabric with a thermally activated adhesive surface, resulting product and applications thereof |
| EP0568993A2 (en) | 1992-05-06 | 1993-11-10 | Kyowa Hakko Kogyo Co., Ltd. | Chemical amplification resist composition |
| JPH05333542A (ja) | 1992-06-03 | 1993-12-17 | Nippon Synthetic Chem Ind Co Ltd:The | 感光性樹脂組成物 |
| JPH05333207A (ja) | 1992-05-29 | 1993-12-17 | Nippon Kayaku Co Ltd | カラーフィルター |
| JPH0611614A (ja) | 1992-02-19 | 1994-01-21 | Eastman Kodak Co | パターン化可能な上塗層を有するカラーフィルターアレイ素子の製造方法 |
| JPH0635183A (ja) | 1992-07-20 | 1994-02-10 | Sumitomo Chem Co Ltd | ポジ型レジスト組成物並びにそれを用いるカラーフィルターの製造方法 |
| JPH0651115A (ja) | 1992-07-31 | 1994-02-25 | Sumitomo Chem Co Ltd | カラーフィルター用色素 |
| JPH0629285B2 (ja) | 1983-10-14 | 1994-04-20 | 三菱化成株式会社 | 光重合性組成物 |
| JPH06157623A (ja) | 1992-08-14 | 1994-06-07 | Toyo Ink Mfg Co Ltd | 重合性組成物および重合方法 |
| JPH06175564A (ja) | 1992-12-04 | 1994-06-24 | Toyo Ink Mfg Co Ltd | ホログラム記録材料及びそれを用いた体積位相型ホログラムの製造方法 |
| JPH06175554A (ja) | 1992-12-03 | 1994-06-24 | Toyo Ink Mfg Co Ltd | 体積位相型ホログラムの製造方法 |
| JPH06175561A (ja) | 1992-12-04 | 1994-06-24 | Toyo Ink Mfg Co Ltd | ホログラム記録媒体及びそれを用いた体積位相型ホログラムの製造方法 |
| JPH06175553A (ja) | 1992-12-03 | 1994-06-24 | Toyo Ink Mfg Co Ltd | ホログラム記録媒体及びそれを用いた体積位相型ホログラムの製造方法 |
| JPH06194828A (ja) | 1992-12-24 | 1994-07-15 | Sumitomo Chem Co Ltd | カラーフィルター用レジスト組成物 |
| JPH06308727A (ja) | 1993-04-19 | 1994-11-04 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPH06348011A (ja) | 1993-06-04 | 1994-12-22 | Toyo Ink Mfg Co Ltd | 光重合性組成物 |
| JPH0712004B2 (ja) | 1988-10-19 | 1995-02-08 | 日本電装株式会社 | コイル用ボビン |
| JPH07128785A (ja) | 1993-11-02 | 1995-05-19 | Konica Corp | 画像形成材料及び画像形成方法 |
| JPH07140589A (ja) | 1993-11-19 | 1995-06-02 | Konica Corp | 画像形成材料および画像形成方法 |
| JPH07261407A (ja) | 1994-03-23 | 1995-10-13 | Mitsubishi Rayon Co Ltd | 可視光硬化型レジストの形成方法 |
| JPH07286107A (ja) | 1994-04-20 | 1995-10-31 | Ricoh Co Ltd | フタロシアニン化合物 |
| JPH07292014A (ja) | 1994-04-25 | 1995-11-07 | Nippon Paint Co Ltd | 近赤外光重合性組成物 |
| JPH07306527A (ja) | 1994-05-11 | 1995-11-21 | Konica Corp | 画像形成材料及び画像形成方法 |
| JPH07120041B2 (ja) | 1987-05-21 | 1995-12-20 | 富士写真フイルム株式会社 | 感光性組成物 |
| JPH07120042B2 (ja) | 1987-05-21 | 1995-12-20 | 富士写真フイルム株式会社 | 感光性組成物 |
| JPH0815522A (ja) | 1994-07-04 | 1996-01-19 | Canon Inc | カラーフィルタ、その製造方法、及びカラーフィルタを備えた液晶パネル |
| JPH0829771A (ja) | 1994-07-15 | 1996-02-02 | Canon Inc | カラーフィルタ、その製造方法、及びカラーフィルタを備えた液晶パネル |
| JPH0812424B2 (ja) | 1987-11-19 | 1996-02-07 | 富士写真フイルム株式会社 | 感光性組成物 |
| JPH0862416A (ja) | 1994-08-22 | 1996-03-08 | Sony Corp | カラーフィルタ用色素およびカラーフィルタの製造方法 |
| JPH0873758A (ja) | 1994-09-06 | 1996-03-19 | Nippon Kayaku Co Ltd | アンスラキノン化合物及びそれを用いたカラーフィルター |
| JPH08108621A (ja) | 1994-10-06 | 1996-04-30 | Konica Corp | 画像記録媒体及びそれを用いる画像形成方法 |
| JPH08146215A (ja) | 1994-09-21 | 1996-06-07 | Canon Inc | カラーフィルタおよびその製造方法、液晶パネルならびに情報処理装置 |
| JPH08151531A (ja) | 1994-09-30 | 1996-06-11 | Nippon Shokubai Co Ltd | キニザリン化合物、その製造方法およびその用途 |
| JPH08179120A (ja) | 1994-12-20 | 1996-07-12 | Nippon Kayaku Co Ltd | カラーフィルタ用色素及びこれを含有するカラーレジスト、カラーフィルタ |
| JPH08211599A (ja) | 1995-02-07 | 1996-08-20 | Hitachi Chem Co Ltd | 着色感光性樹脂組成物、着色画像形成材料、カラーフィルタの製造法及びカラーフィルタ |
| JPH08295810A (ja) | 1995-04-26 | 1996-11-12 | Toyo Ink Mfg Co Ltd | カラーフィルタ用顔料および着色組成物 |
| JPH08302224A (ja) | 1995-04-28 | 1996-11-19 | Yamamoto Chem Inc | フタロシアニン化合物、その中間体、該フタロシアニン化合物の製造方法及びこれを用いるカラーフィルター、油性インク組成物 |
| JPH09188685A (ja) | 1995-11-24 | 1997-07-22 | Ciba Geigy Ag | 光重合のためのボレート補助開始剤 |
| JPH09188710A (ja) | 1995-11-24 | 1997-07-22 | Ciba Geigy Ag | モノボランからのボレート光開始剤 |
| JPH09188686A (ja) | 1995-11-24 | 1997-07-22 | Ciba Geigy Ag | 光重合のための酸安定性硼酸塩 |
| US5667920A (en) | 1996-03-11 | 1997-09-16 | Polaroid Corporation | Process for preparing a color filter |
| JPH09269596A (ja) | 1996-03-29 | 1997-10-14 | Konica Corp | 感光性組成物、感光性平版印刷版及び感光性平版印刷版の製造方法 |
| JPH10110008A (ja) | 1996-10-08 | 1998-04-28 | Nippon Synthetic Chem Ind Co Ltd:The | 感光性樹脂組成物及びこれを用いたドライフィル ムレジスト |
| JPH10116232A (ja) | 1996-10-09 | 1998-05-06 | Niigata Seimitsu Kk | メモリシステム |
| JPH10123316A (ja) | 1996-10-16 | 1998-05-15 | Nippon Kayaku Co Ltd | カラーフィルター及び光学装置 |
| JP2764769B2 (ja) | 1991-06-24 | 1998-06-11 | 富士写真フイルム株式会社 | 光重合性組成物 |
| JPH10198031A (ja) | 1997-01-08 | 1998-07-31 | Nippon Synthetic Chem Ind Co Ltd:The | 感光性樹脂組成物およびその用途 |
| JPH10245501A (ja) | 1997-03-03 | 1998-09-14 | Toyo Ink Mfg Co Ltd | 顔料組成物およびその製造方法 |
| JPH1149974A (ja) | 1996-12-20 | 1999-02-23 | Seiko Epson Corp | 顔料塊状体及びその製造方法、顔料水系分散液、並びに水系インク組成物 |
| JPH11189732A (ja) | 1996-12-16 | 1999-07-13 | Dainichiseika Color & Chem Mfg Co Ltd | 顔料の分散剤、顔料分散液、カラーフイルター用顔料分散液 |
| JPH11199796A (ja) | 1997-11-17 | 1999-07-27 | Toyo Ink Mfg Co Ltd | 顔料分散剤、及びそれを含有する顔料組成物 |
| JPH11271969A (ja) | 1998-01-21 | 1999-10-08 | Mitsubishi Chemical Corp | 光重合性組成物及び感光性平版印刷版 |
| JPH11302283A (ja) | 1998-04-17 | 1999-11-02 | Canon Inc | フタロシアニン化合物、インク、カラーフィルタ、液晶パネル、コンピュータ及びカラーフィルタの製造方法 |
| JPH11343437A (ja) | 1998-03-31 | 1999-12-14 | Canon Inc | インク、カラ―フィルタ、液晶パネル、コンピュ―タ及びカラ―フィルタの製造方法 |
| EP0993966A1 (de) | 1998-10-13 | 2000-04-19 | Agfa-Gevaert AG | Negativ arbeitendes, strahlungsempfindliches Gemisch zur Herstellung eines mit Wärme oder Infrarotlaser bebilderbaren Aufzeichnungsmaterials |
| JP2000131837A (ja) | 1998-08-17 | 2000-05-12 | Mitsubishi Chemicals Corp | 光重合性組成物、光重合性平版印刷版及び画像形成方法 |
| JP2000187322A (ja) | 1998-10-15 | 2000-07-04 | Mitsubishi Chemicals Corp | 感光性組成物、画像形成材料及びそれを用いた画像形成方法 |
| JP2000310808A (ja) | 1999-04-26 | 2000-11-07 | Canon Inc | 照明装置及びそれを用いた撮影装置 |
| JP2001004823A (ja) | 1999-06-21 | 2001-01-12 | Nippon Kayaku Co Ltd | カラーフィルター |
| JP2001115598A (ja) | 1999-10-14 | 2001-04-24 | Misawa Homes Co Ltd | 格子パネル |
| JP2001115595A (ja) | 1999-10-15 | 2001-04-24 | Tagawa Tadahiro | 発光ブロック |
| JP2001125255A (ja) | 1999-10-27 | 2001-05-11 | Fuji Photo Film Co Ltd | 感光性平版印刷版 |
| JP2001132318A (ja) | 1999-10-29 | 2001-05-15 | Oi Seisakusho Co Ltd | 車両用開閉体の開閉装置 |
| JP2001242612A (ja) | 2000-03-01 | 2001-09-07 | Fuji Photo Film Co Ltd | 画像記録材料 |
| JP2001318463A (ja) | 2000-05-11 | 2001-11-16 | Fuji Photo Film Co Ltd | ネガ型平版印刷版原版 |
| JP2001356210A (ja) | 2000-06-14 | 2001-12-26 | Jsr Corp | カラーフィルタ用感放射線性組成物およびカラーフィルタ |
| JP2002014221A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 緑色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002014223A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 黄色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002014220A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 赤色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002014222A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 青色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002062698A (ja) | 2000-08-21 | 2002-02-28 | Kyocera Corp | 画像形成機 |
| JP2002062648A (ja) | 2000-08-21 | 2002-02-28 | Fuji Photo Film Co Ltd | 画像記録材料 |
| JP2002107916A (ja) | 2000-09-27 | 2002-04-10 | Fuji Photo Film Co Ltd | 平版印刷版原版 |
| JP2002107918A (ja) | 2000-10-03 | 2002-04-10 | Fuji Photo Film Co Ltd | 感光性平版印刷版 |
| EP1204000A1 (en) | 2000-11-06 | 2002-05-08 | Fuji Photo Film Co., Ltd. | Photosensitive lithographic printing plate |
| JP2002162741A (ja) | 2000-11-22 | 2002-06-07 | Fuji Photo Film Co Ltd | ネガ型感光性平版印刷版 |
| JP2002309057A (ja) | 2001-04-13 | 2002-10-23 | Fuji Photo Film Co Ltd | 酸分解型感光性組成物及び酸分解型平版印刷版 |
| JP2002311569A (ja) | 2001-04-13 | 2002-10-23 | Fuji Photo Film Co Ltd | 感光性組成物及びネガ型平版印刷版 |
| JP2003043680A (ja) * | 2001-07-30 | 2003-02-13 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2003240938A (ja) | 2002-02-22 | 2003-08-27 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2003262958A (ja) | 2002-03-11 | 2003-09-19 | Fuji Photo Film Co Ltd | 画像記録材料 |
| JP2003321022A (ja) | 2002-04-30 | 2003-11-11 | Toppan Printing Co Ltd | ひねって開封する紙箱 |
| JP2003335814A (ja) | 2002-03-11 | 2003-11-28 | Fuji Photo Film Co Ltd | 高分子化合物の製造方法 |
| JP2003339785A (ja) | 2002-05-24 | 2003-12-02 | France Bed Co Ltd | 起床式ベッド装置 |
| JP2005234478A (ja) | 2004-02-23 | 2005-09-02 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2006016545A (ja) * | 2004-07-02 | 2006-01-19 | Mitsubishi Chemicals Corp | 着色樹脂組成物、カラーフィルタ及び液晶表示装置 |
| JP2006030541A (ja) | 2004-07-15 | 2006-02-02 | Jsr Corp | カラーフィルタ用感放射線性組成物およびその調製法 |
| JP2006201730A (ja) * | 2004-12-24 | 2006-08-03 | Toyo Ink Mfg Co Ltd | カラーフィルタ用青色着色組成物およびカラーフィルタ |
| JP2006215452A (ja) * | 2005-02-07 | 2006-08-17 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2006265528A (ja) | 2005-02-22 | 2006-10-05 | Dainichiseika Color & Chem Mfg Co Ltd | 顔料分散剤およびその使用 |
| JP4420189B2 (ja) | 2003-10-28 | 2010-02-24 | 株式会社Ihi | X線検査装置 |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995018399A1 (fr) * | 1993-12-27 | 1995-07-06 | Hoechst Japan Limited | Composition de resine coloree photosensible |
| KR0140908B1 (ko) * | 1995-01-20 | 1998-06-15 | 박흥기 | 액정 디스플레이 칼라필터용 안료분산 포토 레지스트 조성물 |
| JP2002194244A (ja) | 2000-12-25 | 2002-07-10 | Fuji Photo Film Co Ltd | 顔料分散組成物、それを用いた着色感光性組成物及びカラーフィルタ |
| JP4146105B2 (ja) | 2001-05-30 | 2008-09-03 | 富士フイルム株式会社 | 紫外線吸収剤及びその製造方法、紫外線吸収剤を含有する組成物、ならびに画像形成方法 |
| US6848781B2 (en) * | 2002-09-30 | 2005-02-01 | Canon Kabushiki Kaisha | Image forming process, image-recorded article, liquid composition and ink-jet recording apparatus |
| JP2004347915A (ja) * | 2003-05-23 | 2004-12-09 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| WO2005080337A1 (ja) * | 2004-02-23 | 2005-09-01 | Mitsubishi Chemical Corporation | オキシムエステル系化合物、光重合性組成物及びこれを用いたカラーフィルター |
| JP4661384B2 (ja) * | 2005-06-16 | 2011-03-30 | Jsr株式会社 | 着色層形成用感放射線性組成物およびカラーフィルタ |
| JP2007058085A (ja) * | 2005-08-26 | 2007-03-08 | Fujifilm Holdings Corp | 着色剤含有硬化性組成物、カラーフィルタ及びその製造方法 |
| TWI406840B (zh) * | 2006-02-24 | 2013-09-01 | Fujifilm Corp | 肟衍生物、光聚合性組成物、彩色濾光片及其製法 |
| CN101473268A (zh) | 2006-06-20 | 2009-07-01 | 西巴控股有限公司 | 肟磺酸酯和其作为潜伏酸的用途 |
| JP2008033206A (ja) * | 2006-07-03 | 2008-02-14 | Fujifilm Corp | カラーフィルタ用インクジェットインク、カラーフィルタ、カラーフィルタの製造方法及び表示装置 |
| JP5050428B2 (ja) * | 2006-07-12 | 2012-10-17 | 東洋インキScホールディングス株式会社 | カラーフィルタ用黒色着色組成物およびカラーフィルタ |
| JP5082318B2 (ja) * | 2006-07-24 | 2012-11-28 | 東洋インキScホールディングス株式会社 | α型結晶変態ジクロロジケトピロロピロール顔料の製造方法、該方法で製造されたα型結晶変態ジクロロジケトピロロピロール顔料、およびそれを用いた着色組成物 |
| US7973097B2 (en) * | 2007-04-24 | 2011-07-05 | Canon Kabushiki Kaisha | Ink jet pigment ink and ink set |
| JP5213375B2 (ja) | 2007-07-13 | 2013-06-19 | 富士フイルム株式会社 | 顔料分散液、硬化性組成物、それを用いるカラーフィルタ及び固体撮像素子 |
| JP4990085B2 (ja) * | 2007-09-28 | 2012-08-01 | 富士フイルム株式会社 | 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 |
-
2008
- 2008-03-10 JP JP2008059741A patent/JP5448352B2/ja active Active
-
2009
- 2009-03-05 EP EP09719268.6A patent/EP2270593B1/en active Active
- 2009-03-05 KR KR1020107022533A patent/KR101622996B1/ko active IP Right Grant
- 2009-03-05 WO PCT/JP2009/054216 patent/WO2009113447A1/ja active Application Filing
- 2009-03-05 CN CN2009801081785A patent/CN101960380B/zh active Active
- 2009-03-05 US US12/921,648 patent/US8303860B2/en active Active
Patent Citations (198)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US161811A (en) | 1875-04-06 | Improvement in mechanisms for feeding heel-stiffeners or counter-blanks | ||
| US339049A (en) | 1886-03-30 | Sole-edge-burnishing | ||
| US410201A (en) | 1889-09-03 | Bent for suspension-bridges | ||
| US2848328A (en) | 1954-06-16 | 1958-08-19 | Eastman Kodak Co | Light sensitive diazo compound and binder composition |
| US2833827A (en) | 1955-01-17 | 1958-05-06 | Bayer Ag | Tri (3, 5-di lower alkyl-4-hydroxy phenyl)-sulfonium chlorides and method of preparing same |
| US2852379A (en) | 1955-05-04 | 1958-09-16 | Eastman Kodak Co | Azide resin photolithographic composition |
| US2940853A (en) | 1958-08-21 | 1960-06-14 | Eastman Kodak Co | Azide sensitized resin photographic resist |
| US3479185A (en) | 1965-06-03 | 1969-11-18 | Du Pont | Photopolymerizable compositions and layers containing 2,4,5-triphenylimidazoyl dimers |
| JPS4643946B1 (ja) | 1967-11-09 | 1971-12-27 | ||
| JPS4842965B1 (ja) | 1968-04-02 | 1973-12-15 | ||
| JPS4943191B1 (ja) | 1969-07-11 | 1974-11-19 | ||
| JPS4841708B1 (ja) | 1970-01-13 | 1973-12-07 | ||
| JPS5147334B1 (ja) | 1970-11-02 | 1976-12-14 | ||
| JPS5434327B1 (ja) | 1970-12-28 | 1979-10-26 | ||
| JPS5617654B2 (ja) | 1970-12-28 | 1981-04-23 | ||
| JPS4836281A (ja) | 1971-09-03 | 1973-05-28 | ||
| JPS4864183A (ja) | 1971-12-09 | 1973-09-05 | ||
| US3905815A (en) | 1971-12-17 | 1975-09-16 | Minnesota Mining & Mfg | Photopolymerizable sheet material with diazo resin layer |
| JPS5230490B2 (ja) | 1972-03-21 | 1977-08-09 | ||
| JPS5534414B2 (ja) | 1972-06-02 | 1980-09-06 | ||
| JPS5849860B2 (ja) | 1973-12-07 | 1983-11-07 | ヘキスト アクチェンゲゼルシャフト | コウジユウゴウセイフクシヤザイリヨウ |
| JPS5812577B2 (ja) | 1973-12-21 | 1983-03-09 | ヘキスト アクチエンゲゼルシヤフト | 光重合可能な複写材料 |
| US4069055A (en) | 1974-05-02 | 1978-01-17 | General Electric Company | Photocurable epoxy compositions containing group Va onium salts |
| US4069056A (en) | 1974-05-02 | 1978-01-17 | General Electric Company | Photopolymerizable composition containing group Va aromatic onium salts |
| JPS5137193A (ja) | 1974-09-25 | 1976-03-29 | Toyo Boseki | |
| JPS5425957B2 (ja) | 1974-10-04 | 1979-08-31 | ||
| JPS5182102A (en) | 1974-12-10 | 1976-07-19 | Basf Ag | Insatsuinki oyobi hifukuzairyonotameno hikarijugokanonaketsugozai |
| JPS53702A (en) | 1975-12-23 | 1978-01-06 | Dynachem Corp | Adhesion promoting agent for polymerized film |
| JPS52134692A (en) | 1976-05-04 | 1977-11-11 | Fuji Photo Film Co Ltd | Photopolymerizable composition |
| JPS5492723A (en) | 1977-12-30 | 1979-07-23 | Somar Mfg | Photosensitive material and use |
| DE2904626A1 (de) | 1978-02-08 | 1979-08-09 | Minnesota Mining & Mfg | Triarylsulfoniumkomplexsalze, verfahren zu ihrer herstellung und diese salze enthaltende photopolymerisierbare gemische |
| JPS6239417B2 (ja) | 1978-05-20 | 1987-08-22 | Hoechst Ag | |
| JPS6239418B2 (ja) | 1978-05-20 | 1987-08-22 | Hoechst Ag | |
| JPS5532070A (en) | 1978-08-29 | 1980-03-06 | Fuji Photo Film Co Ltd | Photosensitive resin composition |
| JPS55500806A (ja) | 1978-10-19 | 1980-10-16 | ||
| US4311783A (en) | 1979-08-14 | 1982-01-19 | E. I. Du Pont De Nemours And Company | Dimers derived from unsymmetrical 2,4,5,-triphenylimidazole compounds as photoinitiators |
| JPS5675643A (en) | 1979-11-07 | 1981-06-22 | Hoechst Ag | Photopolymerizable mixture and photopolymerizable copying material using same |
| JPH0140336B2 (ja) | 1979-12-29 | 1989-08-28 | Hoechst Ag | |
| JPH0140337B2 (ja) | 1979-12-29 | 1989-08-28 | Hoechst Ag | |
| JPH0216765B2 (ja) | 1980-09-29 | 1990-04-18 | Hoechst Ag | |
| JPH0232293B2 (ja) | 1980-12-22 | 1990-07-19 | Hoechst Ag | |
| JPS57196231A (en) | 1981-05-20 | 1982-12-02 | Hoechst Ag | Mixture able to be polymerized by radiation and copying material mainly composed thereof |
| JPS595241A (ja) | 1982-06-21 | 1984-01-12 | ヘキスト・アクチエンゲゼルシヤフト | 放射線重合可能な混合物 |
| JPS595240A (ja) | 1982-06-21 | 1984-01-12 | ヘキスト・アクチエンゲゼルシヤフト | 放射線重合可能な混合物 |
| JPS5944615A (ja) | 1982-09-07 | 1984-03-13 | Furuno Electric Co Ltd | ジヤイロ装置 |
| EP0104143A1 (de) | 1982-09-18 | 1984-03-28 | Ciba-Geigy Ag | Diaryljodosylsalze enthaltende photopolymerisierbare Zusammensetzungen |
| JPS5953836A (ja) | 1982-09-21 | 1984-03-28 | Fuji Photo Film Co Ltd | 感光性平版印刷版 |
| JPS5971048A (ja) | 1982-10-18 | 1984-04-21 | Mitsubishi Chem Ind Ltd | 光重合系感光性組成物 |
| US4508811A (en) | 1983-01-17 | 1985-04-02 | U.S. Philips Corporation | Recording element having a pyrylium or thiopyrylium-squarylium dye layer and new pyrylium or thiopyrylium-squarylium compounds |
| JPS59138205A (ja) | 1983-01-20 | 1984-08-08 | チバ−ガイギ− アクチエンゲゼルシヤフト | 被覆組成物およびその硬化方法 |
| JPS59152396A (ja) | 1983-02-11 | 1984-08-31 | チバ−ガイギ− アクチエンゲゼルシヤフト | メタロセン,その製造方法およびメタロセンを含む光重合性組成物 |
| JPS6084305A (ja) | 1983-08-30 | 1985-05-13 | バスフ アクチェン ゲゼルシャフト | 光重合可能な混合物 |
| JPH0629285B2 (ja) | 1983-10-14 | 1994-04-20 | 三菱化成株式会社 | 光重合性組成物 |
| JPS60208748A (ja) | 1984-04-02 | 1985-10-21 | Hitachi Chem Co Ltd | 感光性樹脂組成物及びこれを用いた積層体 |
| JPS60214354A (ja) | 1984-04-10 | 1985-10-26 | Mitsubishi Rayon Co Ltd | 光重合性樹脂組成物 |
| JPS60239736A (ja) | 1984-05-14 | 1985-11-28 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS6122048A (ja) | 1984-06-08 | 1986-01-30 | ヘキスト・アクチエンゲゼルシヤフト | 重合可能な化合物、その製法、およびこれを含有する放射線感性複写層 |
| JPS61151197A (ja) | 1984-12-20 | 1986-07-09 | チバ‐ガイギー アーゲー | チタノセン類およびこれらのチタノセン類を含有する照射重合開始剤 |
| JPS61166544A (ja) | 1985-01-18 | 1986-07-28 | Fuji Photo Film Co Ltd | 光可溶化組成物 |
| JPS61169835A (ja) | 1985-01-22 | 1986-07-31 | Fuji Photo Film Co Ltd | 光可溶化組成物 |
| JPS61169837A (ja) | 1985-01-22 | 1986-07-31 | Fuji Photo Film Co Ltd | 光可溶化組成物 |
| JPS6218537A (ja) | 1985-07-16 | 1987-01-27 | ザ・ミ−ド・コ−ポレ−シヨン | 共開始剤として第三アミンを含有する感光性マイクロカプセルを用いた造像材料 |
| JPS6258241A (ja) | 1985-09-09 | 1987-03-13 | Fuji Photo Film Co Ltd | 感光性組成物 |
| US4622286A (en) | 1985-09-16 | 1986-11-11 | E. I. Du Pont De Nemours And Company | Photoimaging composition containing admixture of leuco dye and 2,4,5-triphenylimidazolyl dimer |
| US4808501A (en) | 1985-10-15 | 1989-02-28 | Polaroid Corporation, Patent Dept. | Method for manufacturing an optical filter |
| JPS62150242A (ja) | 1985-11-20 | 1987-07-04 | サイカラー・インコーポレーテッド | イオン性染料化合物を開始剤として含む感光性材料 |
| JPS62143044A (ja) | 1985-11-20 | 1987-06-26 | サイカラー・インコーポレーテッド | 染料−ほう素化合物錯体を含有する光硬化性組成物およびその組成物を使用する感光性材料 |
| EP0233567A2 (de) | 1986-02-14 | 1987-08-26 | BASF Aktiengesellschaft | Härtbare Mischungen, enthaltend N-Sulfonylaminosulfoniumsalze als kationisch wirksame Katalysatoren |
| US4734444A (en) | 1986-02-14 | 1988-03-29 | Basf Aktiengesellschaft | Curable mixtures containing N-sulfonylaminosulfonium salts as cationically active catalysts |
| DE3604580A1 (de) | 1986-02-14 | 1987-08-20 | Basf Ag | Haertbare mischungen, enthaltend n-sulfonylaminosulfoniumsalze als kationisch wirksame katalysatoren |
| DE3604581A1 (de) | 1986-02-14 | 1987-08-20 | Basf Ag | 4-acylbenzylsulfoniumsalze, ihre herstellung sowie sie enthaltende photohaertbare gemische und aufzeichnungsmaterialien |
| JPS62212401A (ja) | 1986-03-14 | 1987-09-18 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPS6341484A (ja) | 1986-08-01 | 1988-02-22 | チバ−ガイギ− ア−ゲ− | チタノセン、それらの製造法およびそれらを含有する組成物 |
| JPS6370243A (ja) | 1986-09-11 | 1988-03-30 | Fuji Photo Film Co Ltd | 感光性組成物 |
| US4760013A (en) | 1987-02-17 | 1988-07-26 | International Business Machines Corporation | Sulfonium salt photoinitiators |
| JPS63260909A (ja) | 1987-03-28 | 1988-10-27 | ヘキスト・アクチエンゲゼルシヤフト | 光重合性混合物及びこの混合物から製造される記録材料 |
| JPS63277653A (ja) | 1987-03-28 | 1988-11-15 | ヘキスト・アクチエンゲゼルシヤフト | 重合可能な化合物、これを含有する放射線重合可能な混合物及び放射線重合可能な記録材料 |
| JPH01105238A (ja) | 1987-03-28 | 1989-04-21 | Hoechst Ag | 光重合可能な混合物および光重合可能な記録材料 |
| JPS63287947A (ja) | 1987-05-21 | 1988-11-25 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS63287944A (ja) | 1987-05-21 | 1988-11-25 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPH07120041B2 (ja) | 1987-05-21 | 1995-12-20 | 富士写真フイルム株式会社 | 感光性組成物 |
| JPH07120042B2 (ja) | 1987-05-21 | 1995-12-20 | 富士写真フイルム株式会社 | 感光性組成物 |
| JPS63298339A (ja) | 1987-05-29 | 1988-12-06 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPS6433104A (en) | 1987-06-25 | 1989-02-03 | Ciba Geigy Ag | Photopolymerizable composition |
| EP0297443A2 (de) | 1987-07-01 | 1989-01-04 | BASF Aktiengesellschaft | Strahlungsempfindliches Gemisch für lichtempfindliche Beschichtungsmaterialien |
| EP0297442A1 (de) | 1987-07-01 | 1989-01-04 | BASF Aktiengesellschaft | Sulfoniumsalze mit säurelabilen Gruppierungen |
| JPS6490403A (en) | 1987-09-30 | 1989-04-06 | Konishiroku Photo Ind | Color mosaic filter |
| JPS6491102A (en) | 1987-10-01 | 1989-04-10 | Konishiroku Photo Ind | Color mosaic filter |
| JPH0194301A (ja) | 1987-10-06 | 1989-04-13 | Konica Corp | カラーモザイクフィルター |
| JPH01165613A (ja) | 1987-11-16 | 1989-06-29 | Hoechst Ag | 重合可能な化合物、その放射線重合性混合物および放射線重合記録材料 |
| JPH0812424B2 (ja) | 1987-11-19 | 1996-02-07 | 富士写真フイルム株式会社 | 感光性組成物 |
| JPH02249A (ja) | 1987-12-01 | 1990-01-05 | Ciba Geigy Ag | チタノセンおよびそれを含有する光重合性組成物 |
| JPH01152109A (ja) | 1987-12-09 | 1989-06-14 | Toray Ind Inc | 光重合性組成物 |
| US4933377A (en) | 1988-02-29 | 1990-06-12 | Saeva Franklin D | Novel sulfonium salts and the use thereof as photoinitiators |
| JPH024705A (ja) | 1988-03-24 | 1990-01-09 | Dentsply Internatl Inc | 光硬化性組成物用チタナート開始剤 |
| JPH01271741A (ja) | 1988-04-25 | 1989-10-30 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JPH0225493A (ja) | 1988-05-21 | 1990-01-26 | Hoechst Ag | アルケニルホスホン酸エステルおよびアルケニルホスフイン酸エルテル、その製法並びに当該化合物を含有する放射線重合性混合物および記録材料 |
| JPH01304453A (ja) | 1988-06-02 | 1989-12-08 | Toyobo Co Ltd | 光重合性組成物 |
| JPH0712004B2 (ja) | 1988-10-19 | 1995-02-08 | 日本電装株式会社 | コイル用ボビン |
| JPH02127602A (ja) | 1988-10-24 | 1990-05-16 | Polaroid Corp | カラーフィルターの製造方法 |
| EP0370693A2 (en) | 1988-11-21 | 1990-05-30 | Eastman Kodak Company | Novel onium salts and the use thereof as photoinitiators |
| JPH02150848A (ja) | 1988-12-02 | 1990-06-11 | Hitachi Ltd | 光退色性放射線感応性組成物およびそれを用いたパターン形成法 |
| JPH02226149A (ja) | 1988-12-22 | 1990-09-07 | Hoechst Ag | 光重合性化合物、それを含む光重合性混合物及びそれから製造された光重合性複写材料 |
| EP0390214A2 (en) | 1989-03-31 | 1990-10-03 | E.F. Johnson Company | Method and apparatus for a distributive wide area network for a land mobile transmission trunked communication system |
| JPH02296514A (ja) | 1989-05-12 | 1990-12-07 | Matsushita Electric Ind Co Ltd | 車両用サスペンション制御装置 |
| US5227227A (en) | 1990-03-05 | 1993-07-13 | Johnson & Johnson Inc. | Non-woven fabric with a thermally activated adhesive surface, resulting product and applications thereof |
| US5059500A (en) | 1990-10-10 | 1991-10-22 | Polaroid Corporation | Process for forming a color filter |
| JPH04249549A (ja) | 1990-12-28 | 1992-09-04 | Orient Chem Ind Ltd | 着色樹脂及びその関連技術 |
| JPH04365049A (ja) | 1991-06-12 | 1992-12-17 | Fuji Photo Film Co Ltd | 感光性組成物 |
| JP2764769B2 (ja) | 1991-06-24 | 1998-06-11 | 富士写真フイルム株式会社 | 光重合性組成物 |
| JPH0536581A (ja) | 1991-07-30 | 1993-02-12 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPH0583588A (ja) | 1991-09-24 | 1993-04-02 | Omron Corp | 画像処理装置 |
| JPH05142772A (ja) | 1991-11-26 | 1993-06-11 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPH0611614A (ja) | 1992-02-19 | 1994-01-21 | Eastman Kodak Co | パターン化可能な上塗層を有するカラーフィルターアレイ素子の製造方法 |
| JP2592207B2 (ja) | 1992-02-19 | 1997-03-19 | イーストマン コダック カンパニー | パターン化可能な上塗層を有するカラーフィルターアレイ素子の製造方法 |
| EP0568993A2 (en) | 1992-05-06 | 1993-11-10 | Kyowa Hakko Kogyo Co., Ltd. | Chemical amplification resist composition |
| JPH05333207A (ja) | 1992-05-29 | 1993-12-17 | Nippon Kayaku Co Ltd | カラーフィルター |
| JPH05333542A (ja) | 1992-06-03 | 1993-12-17 | Nippon Synthetic Chem Ind Co Ltd:The | 感光性樹脂組成物 |
| JPH0635183A (ja) | 1992-07-20 | 1994-02-10 | Sumitomo Chem Co Ltd | ポジ型レジスト組成物並びにそれを用いるカラーフィルターの製造方法 |
| JPH0651115A (ja) | 1992-07-31 | 1994-02-25 | Sumitomo Chem Co Ltd | カラーフィルター用色素 |
| JPH06157623A (ja) | 1992-08-14 | 1994-06-07 | Toyo Ink Mfg Co Ltd | 重合性組成物および重合方法 |
| JPH06175554A (ja) | 1992-12-03 | 1994-06-24 | Toyo Ink Mfg Co Ltd | 体積位相型ホログラムの製造方法 |
| JPH06175553A (ja) | 1992-12-03 | 1994-06-24 | Toyo Ink Mfg Co Ltd | ホログラム記録媒体及びそれを用いた体積位相型ホログラムの製造方法 |
| JPH06175561A (ja) | 1992-12-04 | 1994-06-24 | Toyo Ink Mfg Co Ltd | ホログラム記録媒体及びそれを用いた体積位相型ホログラムの製造方法 |
| JPH06175564A (ja) | 1992-12-04 | 1994-06-24 | Toyo Ink Mfg Co Ltd | ホログラム記録材料及びそれを用いた体積位相型ホログラムの製造方法 |
| JPH06194828A (ja) | 1992-12-24 | 1994-07-15 | Sumitomo Chem Co Ltd | カラーフィルター用レジスト組成物 |
| JPH06308727A (ja) | 1993-04-19 | 1994-11-04 | Fuji Photo Film Co Ltd | 光重合性組成物 |
| JPH06348011A (ja) | 1993-06-04 | 1994-12-22 | Toyo Ink Mfg Co Ltd | 光重合性組成物 |
| JPH07128785A (ja) | 1993-11-02 | 1995-05-19 | Konica Corp | 画像形成材料及び画像形成方法 |
| JPH07140589A (ja) | 1993-11-19 | 1995-06-02 | Konica Corp | 画像形成材料および画像形成方法 |
| JPH07261407A (ja) | 1994-03-23 | 1995-10-13 | Mitsubishi Rayon Co Ltd | 可視光硬化型レジストの形成方法 |
| JPH07286107A (ja) | 1994-04-20 | 1995-10-31 | Ricoh Co Ltd | フタロシアニン化合物 |
| JPH07292014A (ja) | 1994-04-25 | 1995-11-07 | Nippon Paint Co Ltd | 近赤外光重合性組成物 |
| JPH07306527A (ja) | 1994-05-11 | 1995-11-21 | Konica Corp | 画像形成材料及び画像形成方法 |
| JPH0815522A (ja) | 1994-07-04 | 1996-01-19 | Canon Inc | カラーフィルタ、その製造方法、及びカラーフィルタを備えた液晶パネル |
| JPH0829771A (ja) | 1994-07-15 | 1996-02-02 | Canon Inc | カラーフィルタ、その製造方法、及びカラーフィルタを備えた液晶パネル |
| JPH0862416A (ja) | 1994-08-22 | 1996-03-08 | Sony Corp | カラーフィルタ用色素およびカラーフィルタの製造方法 |
| JPH0873758A (ja) | 1994-09-06 | 1996-03-19 | Nippon Kayaku Co Ltd | アンスラキノン化合物及びそれを用いたカラーフィルター |
| JPH08146215A (ja) | 1994-09-21 | 1996-06-07 | Canon Inc | カラーフィルタおよびその製造方法、液晶パネルならびに情報処理装置 |
| JPH08151531A (ja) | 1994-09-30 | 1996-06-11 | Nippon Shokubai Co Ltd | キニザリン化合物、その製造方法およびその用途 |
| JPH08108621A (ja) | 1994-10-06 | 1996-04-30 | Konica Corp | 画像記録媒体及びそれを用いる画像形成方法 |
| JPH08179120A (ja) | 1994-12-20 | 1996-07-12 | Nippon Kayaku Co Ltd | カラーフィルタ用色素及びこれを含有するカラーレジスト、カラーフィルタ |
| JPH08211599A (ja) | 1995-02-07 | 1996-08-20 | Hitachi Chem Co Ltd | 着色感光性樹脂組成物、着色画像形成材料、カラーフィルタの製造法及びカラーフィルタ |
| JPH08295810A (ja) | 1995-04-26 | 1996-11-12 | Toyo Ink Mfg Co Ltd | カラーフィルタ用顔料および着色組成物 |
| JPH08302224A (ja) | 1995-04-28 | 1996-11-19 | Yamamoto Chem Inc | フタロシアニン化合物、その中間体、該フタロシアニン化合物の製造方法及びこれを用いるカラーフィルター、油性インク組成物 |
| JPH09188685A (ja) | 1995-11-24 | 1997-07-22 | Ciba Geigy Ag | 光重合のためのボレート補助開始剤 |
| JPH09188710A (ja) | 1995-11-24 | 1997-07-22 | Ciba Geigy Ag | モノボランからのボレート光開始剤 |
| JPH09188686A (ja) | 1995-11-24 | 1997-07-22 | Ciba Geigy Ag | 光重合のための酸安定性硼酸塩 |
| US5667920A (en) | 1996-03-11 | 1997-09-16 | Polaroid Corporation | Process for preparing a color filter |
| JPH09269596A (ja) | 1996-03-29 | 1997-10-14 | Konica Corp | 感光性組成物、感光性平版印刷版及び感光性平版印刷版の製造方法 |
| JPH10110008A (ja) | 1996-10-08 | 1998-04-28 | Nippon Synthetic Chem Ind Co Ltd:The | 感光性樹脂組成物及びこれを用いたドライフィル ムレジスト |
| JPH10116232A (ja) | 1996-10-09 | 1998-05-06 | Niigata Seimitsu Kk | メモリシステム |
| JPH10123316A (ja) | 1996-10-16 | 1998-05-15 | Nippon Kayaku Co Ltd | カラーフィルター及び光学装置 |
| JPH11189732A (ja) | 1996-12-16 | 1999-07-13 | Dainichiseika Color & Chem Mfg Co Ltd | 顔料の分散剤、顔料分散液、カラーフイルター用顔料分散液 |
| JPH1149974A (ja) | 1996-12-20 | 1999-02-23 | Seiko Epson Corp | 顔料塊状体及びその製造方法、顔料水系分散液、並びに水系インク組成物 |
| JPH10198031A (ja) | 1997-01-08 | 1998-07-31 | Nippon Synthetic Chem Ind Co Ltd:The | 感光性樹脂組成物およびその用途 |
| JPH10245501A (ja) | 1997-03-03 | 1998-09-14 | Toyo Ink Mfg Co Ltd | 顔料組成物およびその製造方法 |
| JPH11199796A (ja) | 1997-11-17 | 1999-07-27 | Toyo Ink Mfg Co Ltd | 顔料分散剤、及びそれを含有する顔料組成物 |
| JPH11271969A (ja) | 1998-01-21 | 1999-10-08 | Mitsubishi Chemical Corp | 光重合性組成物及び感光性平版印刷版 |
| JPH11343437A (ja) | 1998-03-31 | 1999-12-14 | Canon Inc | インク、カラ―フィルタ、液晶パネル、コンピュ―タ及びカラ―フィルタの製造方法 |
| JPH11302283A (ja) | 1998-04-17 | 1999-11-02 | Canon Inc | フタロシアニン化合物、インク、カラーフィルタ、液晶パネル、コンピュータ及びカラーフィルタの製造方法 |
| JP2000131837A (ja) | 1998-08-17 | 2000-05-12 | Mitsubishi Chemicals Corp | 光重合性組成物、光重合性平版印刷版及び画像形成方法 |
| EP0993966A1 (de) | 1998-10-13 | 2000-04-19 | Agfa-Gevaert AG | Negativ arbeitendes, strahlungsempfindliches Gemisch zur Herstellung eines mit Wärme oder Infrarotlaser bebilderbaren Aufzeichnungsmaterials |
| JP2000187322A (ja) | 1998-10-15 | 2000-07-04 | Mitsubishi Chemicals Corp | 感光性組成物、画像形成材料及びそれを用いた画像形成方法 |
| JP2000310808A (ja) | 1999-04-26 | 2000-11-07 | Canon Inc | 照明装置及びそれを用いた撮影装置 |
| JP2001004823A (ja) | 1999-06-21 | 2001-01-12 | Nippon Kayaku Co Ltd | カラーフィルター |
| JP2001115598A (ja) | 1999-10-14 | 2001-04-24 | Misawa Homes Co Ltd | 格子パネル |
| JP2001115595A (ja) | 1999-10-15 | 2001-04-24 | Tagawa Tadahiro | 発光ブロック |
| JP2001125255A (ja) | 1999-10-27 | 2001-05-11 | Fuji Photo Film Co Ltd | 感光性平版印刷版 |
| JP2001132318A (ja) | 1999-10-29 | 2001-05-15 | Oi Seisakusho Co Ltd | 車両用開閉体の開閉装置 |
| JP2001242612A (ja) | 2000-03-01 | 2001-09-07 | Fuji Photo Film Co Ltd | 画像記録材料 |
| JP2001318463A (ja) | 2000-05-11 | 2001-11-16 | Fuji Photo Film Co Ltd | ネガ型平版印刷版原版 |
| JP2001356210A (ja) | 2000-06-14 | 2001-12-26 | Jsr Corp | カラーフィルタ用感放射線性組成物およびカラーフィルタ |
| JP2002014223A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 黄色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002014221A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 緑色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002014220A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 赤色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002014222A (ja) | 2000-06-30 | 2002-01-18 | Sumitomo Chem Co Ltd | 青色フィルタ層を有する色フィルタアレイおよびその製造方法 |
| JP2002062698A (ja) | 2000-08-21 | 2002-02-28 | Kyocera Corp | 画像形成機 |
| JP2002062648A (ja) | 2000-08-21 | 2002-02-28 | Fuji Photo Film Co Ltd | 画像記録材料 |
| JP2002107916A (ja) | 2000-09-27 | 2002-04-10 | Fuji Photo Film Co Ltd | 平版印刷版原版 |
| JP2002107918A (ja) | 2000-10-03 | 2002-04-10 | Fuji Photo Film Co Ltd | 感光性平版印刷版 |
| EP1204000A1 (en) | 2000-11-06 | 2002-05-08 | Fuji Photo Film Co., Ltd. | Photosensitive lithographic printing plate |
| JP2002162741A (ja) | 2000-11-22 | 2002-06-07 | Fuji Photo Film Co Ltd | ネガ型感光性平版印刷版 |
| JP2002309057A (ja) | 2001-04-13 | 2002-10-23 | Fuji Photo Film Co Ltd | 酸分解型感光性組成物及び酸分解型平版印刷版 |
| JP2002311569A (ja) | 2001-04-13 | 2002-10-23 | Fuji Photo Film Co Ltd | 感光性組成物及びネガ型平版印刷版 |
| JP2003043680A (ja) * | 2001-07-30 | 2003-02-13 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2003240938A (ja) | 2002-02-22 | 2003-08-27 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2003262958A (ja) | 2002-03-11 | 2003-09-19 | Fuji Photo Film Co Ltd | 画像記録材料 |
| JP2003335814A (ja) | 2002-03-11 | 2003-11-28 | Fuji Photo Film Co Ltd | 高分子化合物の製造方法 |
| JP2003321022A (ja) | 2002-04-30 | 2003-11-11 | Toppan Printing Co Ltd | ひねって開封する紙箱 |
| JP2003339785A (ja) | 2002-05-24 | 2003-12-02 | France Bed Co Ltd | 起床式ベッド装置 |
| JP4420189B2 (ja) | 2003-10-28 | 2010-02-24 | 株式会社Ihi | X線検査装置 |
| JP2005234478A (ja) | 2004-02-23 | 2005-09-02 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2006016545A (ja) * | 2004-07-02 | 2006-01-19 | Mitsubishi Chemicals Corp | 着色樹脂組成物、カラーフィルタ及び液晶表示装置 |
| JP2006030541A (ja) | 2004-07-15 | 2006-02-02 | Jsr Corp | カラーフィルタ用感放射線性組成物およびその調製法 |
| JP2006201730A (ja) * | 2004-12-24 | 2006-08-03 | Toyo Ink Mfg Co Ltd | カラーフィルタ用青色着色組成物およびカラーフィルタ |
| JP2006215452A (ja) * | 2005-02-07 | 2006-08-17 | Toyo Ink Mfg Co Ltd | カラーフィルタ用着色組成物およびカラーフィルタ |
| JP2006265528A (ja) | 2005-02-22 | 2006-10-05 | Dainichiseika Color & Chem Mfg Co Ltd | 顔料分散剤およびその使用 |
Non-Patent Citations (11)
| Title |
|---|
| C. S. WEN ET AL., TECH. PROC. CONF. RAD. CURING ASIA, October 1988 (1988-10-01), pages 478 |
| J. V. CRIVELLO ET AL., J. POLYMER SCI., vol. 17, 1979, pages 1047 |
| J. V. CRIVELLO ET AL., MACROMOLECULES, vol. 10, no. 6, 1977, pages 1307 |
| JOURNAL OF JAPANESE ADHESIVE SOCIETY, vol. 20, no. 7, 1984, pages 300 - 308 |
| JOURNAL OF POLYMER SOCIETY, vol. 10, 1972, pages 3,173 |
| KUNZ, MARTIN, RAD TECH '98, 19 April 1998 (1998-04-19) |
| S. I. SCHLESINGER, PHOTOGR. SCI. ENG., vol. 18, 1974, pages 387 |
| See also references of EP2270593A4 |
| T. C. PATTON: "Paint Flow and Pigment Dispersion", 1964, JOHN WILEY AND SONS |
| T. S. BAL ET AL., POLYMER, vol. 21, 1980, pages 423 |
| WAKABAYASHI, BULL CHEM. SOC JAPAN, vol. 42, 1969, pages 2924 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102053494A (zh) * | 2009-10-28 | 2011-05-11 | 住友化学株式会社 | 着色感光性树脂组合物 |
| CN102053494B (zh) * | 2009-10-28 | 2014-10-15 | 住友化学株式会社 | 着色感光性树脂组合物及涂膜、图样、滤色器 |
| JP2017187587A (ja) * | 2016-04-05 | 2017-10-12 | 東洋インキScホールディングス株式会社 | カラーフィルタ用着色組成物およびカラーフィルタ |
Also Published As
| Publication number | Publication date |
|---|---|
| US20110017962A1 (en) | 2011-01-27 |
| KR101622996B1 (ko) | 2016-05-20 |
| KR20100125395A (ko) | 2010-11-30 |
| EP2270593B1 (en) | 2014-04-30 |
| JP5448352B2 (ja) | 2014-03-19 |
| CN101960380A (zh) | 2011-01-26 |
| EP2270593A1 (en) | 2011-01-05 |
| JP2009216913A (ja) | 2009-09-24 |
| US8303860B2 (en) | 2012-11-06 |
| EP2270593A4 (en) | 2011-10-05 |
| CN101960380B (zh) | 2013-04-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5448352B2 (ja) | 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 | |
| JP5213375B2 (ja) | 顔料分散液、硬化性組成物、それを用いるカラーフィルタ及び固体撮像素子 | |
| JP5344790B2 (ja) | 硬化性組成物、カラーフィルタ及びその製造方法 | |
| JP5339781B2 (ja) | 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 | |
| JP5371449B2 (ja) | 樹脂、顔料分散液、着色硬化性組成物、これを用いたカラーフィルタ及びその製造方法 | |
| JP5512095B2 (ja) | 感光性組成物、固体撮像素子用感光性組成物、固体撮像素子用遮光性カラーフィルタ、及び固体撮像素子 | |
| JP4959411B2 (ja) | 着色光重合性組成物並びにそれを用いたカラーフィルタ及びカラーフィルタの製造方法 | |
| JP5878492B2 (ja) | 固体撮像素子用着色光硬化性組成物、カラーフィルタ及びその製造方法、並びに固体撮像素子 | |
| JP5528677B2 (ja) | 重合性組成物、固体撮像素子用遮光性カラーフィルタ、固体撮像素子および固体撮像素子用遮光性カラーフィルタの製造方法 | |
| JP4969189B2 (ja) | 固体撮像素子用途のカラーフィルタ形成用の硬化性組成物、固体撮像素子用途のカラーフィルタ及びその製造方法 | |
| JP4990085B2 (ja) | 着色硬化性組成物、カラーフィルタ、及び、固体撮像素子 | |
| JP2007316445A (ja) | 硬化性組成物、カラーフィルタ、及びその製造方法 | |
| JP2008032860A (ja) | 硬化性組成物、カラーフィルタ、及びその製造方法 | |
| JP5586828B2 (ja) | 顔料分散組成物、硬化性組成物、並びにカラーフィルタ及びその製造方法 | |
| JP5535444B2 (ja) | 固体撮像素子用緑色硬化性組成物、固体撮像素子用カラーフィルタ及びその製造方法 | |
| JP5523655B2 (ja) | 顔料分散組成物、硬化性組成物、並びにカラーフィルタ及びその製造方法 | |
| JP2008134613A (ja) | カラーフィルタ用着色硬化性組成物、カラーフィルタ、及びその製造方法 | |
| JP4805018B2 (ja) | 光重合性組成物、カラーフィルタ、及びその製造方法 | |
| JP5105867B2 (ja) | 硬化性組成物、カラーフィルタ及びその製造方法 | |
| JP2009109750A (ja) | 着色重合性組成物、カラーフィルタ、及び固体撮像素子 | |
| JP2008197285A (ja) | カラーフィルタ用硬化性組成物 | |
| JP2008222844A (ja) | 非水系用顔料分散剤、非水系顔料分散組成物、光硬化性組成物、並びにカラーフィルタおよびその製造方法 | |
| JP4837500B2 (ja) | 硬化性組成物、カラーフィルタ、及びその製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980108178.5 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09719268 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12921648 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009719268 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20107022533 Country of ref document: KR Kind code of ref document: A |