WO2018025982A1 - Il-8関連疾患の治療用又は予防用組成物 - Google Patents
Il-8関連疾患の治療用又は予防用組成物 Download PDFInfo
- Publication number
- WO2018025982A1 WO2018025982A1 PCT/JP2017/028346 JP2017028346W WO2018025982A1 WO 2018025982 A1 WO2018025982 A1 WO 2018025982A1 JP 2017028346 W JP2017028346 W JP 2017028346W WO 2018025982 A1 WO2018025982 A1 WO 2018025982A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino acid
- seq
- acid sequence
- antibody
- hvr
- Prior art date
Links
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 title claims abstract description 113
- 201000010099 disease Diseases 0.000 title claims abstract description 110
- 239000000203 mixture Substances 0.000 title claims description 71
- 238000011282 treatment Methods 0.000 title abstract description 34
- 238000011321 prophylaxis Methods 0.000 title 1
- 108090001007 Interleukin-8 Proteins 0.000 claims abstract description 399
- 239000003112 inhibitor Substances 0.000 claims abstract description 160
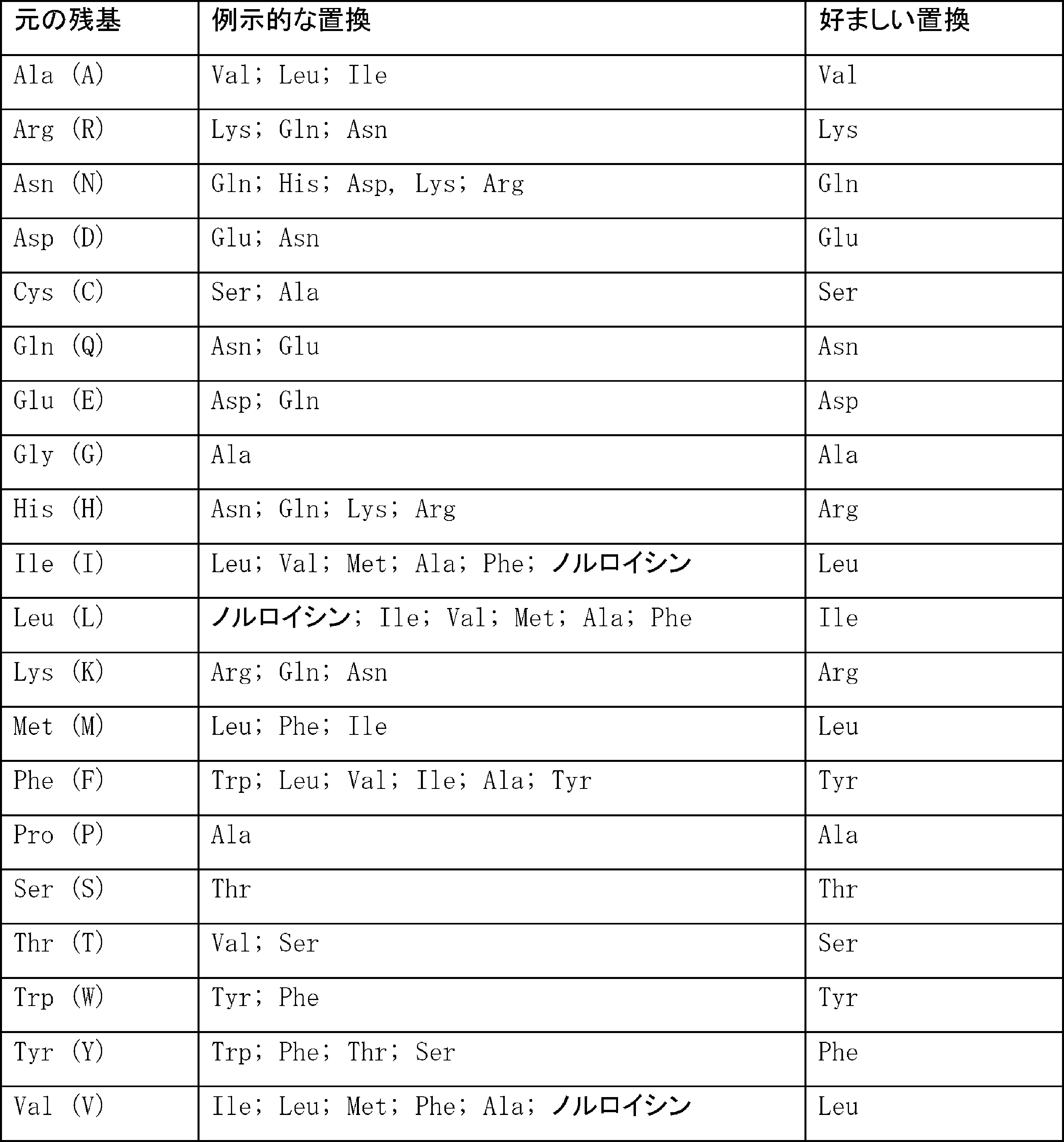
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 441
- 102000004890 Interleukin-8 Human genes 0.000 claims description 396
- 230000027455 binding Effects 0.000 claims description 304
- 238000009739 binding Methods 0.000 claims description 250
- 101001055222 Homo sapiens Interleukin-8 Proteins 0.000 claims description 162
- 241000282414 Homo sapiens Species 0.000 claims description 149
- 238000006467 substitution reaction Methods 0.000 claims description 144
- 201000009273 Endometriosis Diseases 0.000 claims description 142
- 235000001014 amino acid Nutrition 0.000 claims description 126
- 102100026120 IgG receptor FcRn large subunit p51 Human genes 0.000 claims description 115
- 101710177940 IgG receptor FcRn large subunit p51 Proteins 0.000 claims description 115
- 230000002378 acidificating effect Effects 0.000 claims description 83
- 229940024606 amino acid Drugs 0.000 claims description 82
- 210000004027 cell Anatomy 0.000 claims description 79
- 150000001413 amino acids Chemical class 0.000 claims description 78
- 201000009274 endometriosis of uterus Diseases 0.000 claims description 75
- 206010016654 Fibrosis Diseases 0.000 claims description 71
- 230000004761 fibrosis Effects 0.000 claims description 69
- 208000005171 Dysmenorrhea Diseases 0.000 claims description 67
- 206010013935 Dysmenorrhoea Diseases 0.000 claims description 64
- 108010018951 Interleukin-8B Receptors Proteins 0.000 claims description 60
- 102100036166 C-X-C chemokine receptor type 1 Human genes 0.000 claims description 56
- 101000947174 Homo sapiens C-X-C chemokine receptor type 1 Proteins 0.000 claims description 56
- 238000004519 manufacturing process Methods 0.000 claims description 56
- 208000002193 Pain Diseases 0.000 claims description 55
- 230000001419 dependent effect Effects 0.000 claims description 52
- 230000036407 pain Effects 0.000 claims description 48
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 46
- 208000005641 Adenomyosis Diseases 0.000 claims description 37
- 230000001225 therapeutic effect Effects 0.000 claims description 31
- 230000003902 lesion Effects 0.000 claims description 30
- 206010061218 Inflammation Diseases 0.000 claims description 29
- 230000004054 inflammatory process Effects 0.000 claims description 29
- 208000000509 infertility Diseases 0.000 claims description 26
- 230000036512 infertility Effects 0.000 claims description 26
- 102000005962 receptors Human genes 0.000 claims description 26
- 108020003175 receptors Proteins 0.000 claims description 26
- 231100000535 infertility Toxicity 0.000 claims description 25
- 239000012636 effector Substances 0.000 claims description 23
- 125000000404 glutamine group Chemical group N[C@@H](CCC(N)=O)C(=O)* 0.000 claims description 23
- 230000002401 inhibitory effect Effects 0.000 claims description 23
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 claims description 22
- 235000004554 glutamine Nutrition 0.000 claims description 22
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 claims description 21
- 230000002357 endometrial effect Effects 0.000 claims description 21
- 210000002865 immune cell Anatomy 0.000 claims description 21
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 claims description 20
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 claims description 19
- 235000009582 asparagine Nutrition 0.000 claims description 19
- 229960001230 asparagine Drugs 0.000 claims description 19
- 235000009697 arginine Nutrition 0.000 claims description 18
- 125000001909 leucine group Chemical group [H]N(*)C(C(*)=O)C([H])([H])C(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 18
- 125000000637 arginyl group Chemical class N[C@@H](CCCNC(N)=N)C(=O)* 0.000 claims description 17
- 239000004475 Arginine Substances 0.000 claims description 16
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Natural products NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 16
- 235000004279 alanine Nutrition 0.000 claims description 16
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 claims description 16
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 claims description 16
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 15
- 230000008595 infiltration Effects 0.000 claims description 14
- 238000001764 infiltration Methods 0.000 claims description 14
- 239000004471 Glycine Substances 0.000 claims description 13
- 230000000069 prophylactic effect Effects 0.000 claims description 13
- 208000005069 pulmonary fibrosis Diseases 0.000 claims description 13
- 230000001568 sexual effect Effects 0.000 claims description 13
- 125000000613 asparagine group Chemical group N[C@@H](CC(N)=O)C(=O)* 0.000 claims description 12
- 108010078554 Aromatase Proteins 0.000 claims description 10
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 claims description 10
- 239000004480 active ingredient Substances 0.000 claims description 10
- 206010003694 Atrophy Diseases 0.000 claims description 8
- 230000037444 atrophy Effects 0.000 claims description 8
- 201000002793 renal fibrosis Diseases 0.000 claims description 8
- 208000019425 cirrhosis of liver Diseases 0.000 claims description 7
- 210000002919 epithelial cell Anatomy 0.000 claims description 7
- 201000003883 Cystic fibrosis Diseases 0.000 claims description 6
- 201000004681 Psoriasis Diseases 0.000 claims description 6
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 claims description 6
- 210000004907 gland Anatomy 0.000 claims description 5
- 238000001356 surgical procedure Methods 0.000 claims description 5
- 102100028989 C-X-C chemokine receptor type 2 Human genes 0.000 claims description 3
- 208000021642 Muscular disease Diseases 0.000 claims 3
- 201000009623 Myopathy Diseases 0.000 claims 3
- 102000014654 Aromatase Human genes 0.000 claims 1
- 238000000034 method Methods 0.000 abstract description 100
- 102000002791 Interleukin-8B Receptors Human genes 0.000 description 58
- 239000000427 antigen Substances 0.000 description 57
- 108091007433 antigens Proteins 0.000 description 56
- 102000036639 antigens Human genes 0.000 description 56
- 230000004048 modification Effects 0.000 description 46
- 238000012986 modification Methods 0.000 description 46
- 238000010494 dissociation reaction Methods 0.000 description 44
- 230000001965 increasing effect Effects 0.000 description 44
- 241000699666 Mus <mouse, genus> Species 0.000 description 43
- 230000005593 dissociations Effects 0.000 description 43
- 230000000694 effects Effects 0.000 description 41
- 230000007935 neutral effect Effects 0.000 description 41
- 150000007523 nucleic acids Chemical class 0.000 description 37
- 102000039446 nucleic acids Human genes 0.000 description 36
- 108020004707 nucleic acids Proteins 0.000 description 36
- 238000005259 measurement Methods 0.000 description 34
- 230000005847 immunogenicity Effects 0.000 description 32
- 210000000440 neutrophil Anatomy 0.000 description 31
- 230000002829 reductive effect Effects 0.000 description 28
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 27
- 238000001727 in vivo Methods 0.000 description 26
- 108090000623 proteins and genes Proteins 0.000 description 26
- 230000003472 neutralizing effect Effects 0.000 description 25
- 210000001163 endosome Anatomy 0.000 description 24
- 230000014759 maintenance of location Effects 0.000 description 24
- 210000001519 tissue Anatomy 0.000 description 24
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Chemical group NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 23
- 241000282567 Macaca fascicularis Species 0.000 description 23
- 239000003814 drug Substances 0.000 description 23
- 230000035772 mutation Effects 0.000 description 23
- 125000000539 amino acid group Chemical group 0.000 description 22
- 239000000243 solution Substances 0.000 description 22
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 21
- 235000014304 histidine Nutrition 0.000 description 21
- 230000001976 improved effect Effects 0.000 description 21
- 102000004169 proteins and genes Human genes 0.000 description 21
- 230000008034 disappearance Effects 0.000 description 20
- 235000018102 proteins Nutrition 0.000 description 20
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 18
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 18
- 210000004369 blood Anatomy 0.000 description 18
- 239000008280 blood Substances 0.000 description 18
- 108010068617 neonatal Fc receptor Proteins 0.000 description 18
- 239000000523 sample Substances 0.000 description 18
- 208000024891 symptom Diseases 0.000 description 18
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 17
- 239000004472 Lysine Substances 0.000 description 17
- 230000014509 gene expression Effects 0.000 description 17
- 230000000670 limiting effect Effects 0.000 description 17
- 235000018977 lysine Nutrition 0.000 description 17
- 230000005012 migration Effects 0.000 description 17
- 238000013508 migration Methods 0.000 description 17
- 206010039073 rheumatoid arthritis Diseases 0.000 description 17
- 238000012360 testing method Methods 0.000 description 17
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 16
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 16
- 108060003951 Immunoglobulin Proteins 0.000 description 16
- 210000002744 extracellular matrix Anatomy 0.000 description 16
- 102000018358 immunoglobulin Human genes 0.000 description 16
- 230000002265 prevention Effects 0.000 description 16
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical group OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 15
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical group OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 15
- 108090000765 processed proteins & peptides Proteins 0.000 description 15
- 150000003668 tyrosines Chemical group 0.000 description 15
- 108091007491 NSP3 Papain-like protease domains Proteins 0.000 description 14
- 230000006870 function Effects 0.000 description 14
- 210000002966 serum Anatomy 0.000 description 14
- 238000003860 storage Methods 0.000 description 14
- 241000282412 Homo Species 0.000 description 13
- 239000013598 vector Substances 0.000 description 13
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 12
- 241000699670 Mus sp. Species 0.000 description 12
- 239000003937 drug carrier Substances 0.000 description 12
- 238000005516 engineering process Methods 0.000 description 12
- 238000011156 evaluation Methods 0.000 description 12
- 230000003176 fibrotic effect Effects 0.000 description 12
- 238000002360 preparation method Methods 0.000 description 12
- 241000282693 Cercopithecidae Species 0.000 description 11
- 241001465754 Metazoa Species 0.000 description 11
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 11
- 230000007423 decrease Effects 0.000 description 11
- 239000008194 pharmaceutical composition Substances 0.000 description 11
- 230000007704 transition Effects 0.000 description 11
- 235000003704 aspartic acid Nutrition 0.000 description 10
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 10
- 230000003247 decreasing effect Effects 0.000 description 10
- 230000005906 menstruation Effects 0.000 description 10
- 210000000056 organ Anatomy 0.000 description 10
- 229920001184 polypeptide Polymers 0.000 description 10
- 102000004196 processed proteins & peptides Human genes 0.000 description 10
- 239000012146 running buffer Substances 0.000 description 10
- 102100029361 Aromatase Human genes 0.000 description 9
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 229940079593 drug Drugs 0.000 description 9
- 230000006872 improvement Effects 0.000 description 9
- 230000005764 inhibitory process Effects 0.000 description 9
- 230000011664 signaling Effects 0.000 description 9
- 239000003981 vehicle Substances 0.000 description 9
- NFGXHKASABOEEW-UHFFFAOYSA-N 1-methylethyl 11-methoxy-3,7,11-trimethyl-2,4-dodecadienoate Chemical compound COC(C)(C)CCCC(C)CC=CC(C)=CC(=O)OC(C)C NFGXHKASABOEEW-UHFFFAOYSA-N 0.000 description 8
- 101710155857 C-C motif chemokine 2 Proteins 0.000 description 8
- 102000000018 Chemokine CCL2 Human genes 0.000 description 8
- 102000000989 Complement System Proteins Human genes 0.000 description 8
- 108010069112 Complement System Proteins Proteins 0.000 description 8
- 101001100327 Homo sapiens RNA-binding protein 45 Proteins 0.000 description 8
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 8
- 241000124008 Mammalia Species 0.000 description 8
- 101100288142 Mus musculus Klkb1 gene Proteins 0.000 description 8
- 102100038823 RNA-binding protein 45 Human genes 0.000 description 8
- 230000004071 biological effect Effects 0.000 description 8
- 238000011088 calibration curve Methods 0.000 description 8
- 230000008859 change Effects 0.000 description 8
- 239000003795 chemical substances by application Substances 0.000 description 8
- 239000012530 fluid Substances 0.000 description 8
- 230000002757 inflammatory effect Effects 0.000 description 8
- 238000003780 insertion Methods 0.000 description 8
- 230000037431 insertion Effects 0.000 description 8
- 210000003205 muscle Anatomy 0.000 description 8
- 201000008383 nephritis Diseases 0.000 description 8
- 230000001575 pathological effect Effects 0.000 description 8
- 230000009467 reduction Effects 0.000 description 8
- 238000002965 ELISA Methods 0.000 description 7
- 102000010781 Interleukin-6 Receptors Human genes 0.000 description 7
- 108010038501 Interleukin-6 Receptors Proteins 0.000 description 7
- 241000288906 Primates Species 0.000 description 7
- 102100024952 Protein CBFA2T1 Human genes 0.000 description 7
- 230000033115 angiogenesis Effects 0.000 description 7
- 201000011510 cancer Diseases 0.000 description 7
- 208000037976 chronic inflammation Diseases 0.000 description 7
- 238000003745 diagnosis Methods 0.000 description 7
- 230000008030 elimination Effects 0.000 description 7
- 238000003379 elimination reaction Methods 0.000 description 7
- 238000002474 experimental method Methods 0.000 description 7
- 229940125697 hormonal agent Drugs 0.000 description 7
- 210000003734 kidney Anatomy 0.000 description 7
- 210000002540 macrophage Anatomy 0.000 description 7
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 7
- 210000001616 monocyte Anatomy 0.000 description 7
- 230000008569 process Effects 0.000 description 7
- 238000005406 washing Methods 0.000 description 7
- 108010088751 Albumins Proteins 0.000 description 6
- 102000009027 Albumins Human genes 0.000 description 6
- 208000000094 Chronic Pain Diseases 0.000 description 6
- 102000008186 Collagen Human genes 0.000 description 6
- 108010035532 Collagen Proteins 0.000 description 6
- 101000916059 Homo sapiens C-X-C chemokine receptor type 2 Proteins 0.000 description 6
- 201000009794 Idiopathic Pulmonary Fibrosis Diseases 0.000 description 6
- 241000699660 Mus musculus Species 0.000 description 6
- 241000283973 Oryctolagus cuniculus Species 0.000 description 6
- 239000002202 Polyethylene glycol Substances 0.000 description 6
- RJKFOVLPORLFTN-LEKSSAKUSA-N Progesterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](C(=O)C)[C@@]1(C)CC2 RJKFOVLPORLFTN-LEKSSAKUSA-N 0.000 description 6
- 241000700159 Rattus Species 0.000 description 6
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 6
- 238000004458 analytical method Methods 0.000 description 6
- 150000001508 asparagines Chemical group 0.000 description 6
- 239000000872 buffer Substances 0.000 description 6
- 238000004587 chromatography analysis Methods 0.000 description 6
- 230000001684 chronic effect Effects 0.000 description 6
- 229920001436 collagen Polymers 0.000 description 6
- 238000012217 deletion Methods 0.000 description 6
- 230000037430 deletion Effects 0.000 description 6
- 210000000981 epithelium Anatomy 0.000 description 6
- 230000036541 health Effects 0.000 description 6
- 210000004408 hybridoma Anatomy 0.000 description 6
- 229920001223 polyethylene glycol Polymers 0.000 description 6
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 6
- 238000004321 preservation Methods 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 230000008929 regeneration Effects 0.000 description 6
- 238000011069 regeneration method Methods 0.000 description 6
- 238000002560 therapeutic procedure Methods 0.000 description 6
- 238000011830 transgenic mouse model Methods 0.000 description 6
- 210000004291 uterus Anatomy 0.000 description 6
- 210000003556 vascular endothelial cell Anatomy 0.000 description 6
- 108700028369 Alleles Proteins 0.000 description 5
- 102000019034 Chemokines Human genes 0.000 description 5
- 108010012236 Chemokines Proteins 0.000 description 5
- 102000004127 Cytokines Human genes 0.000 description 5
- 108090000695 Cytokines Proteins 0.000 description 5
- 229920001213 Polysorbate 20 Polymers 0.000 description 5
- 210000001744 T-lymphocyte Anatomy 0.000 description 5
- 238000003556 assay Methods 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 238000004364 calculation method Methods 0.000 description 5
- 230000006020 chronic inflammation Effects 0.000 description 5
- 229940011871 estrogen Drugs 0.000 description 5
- 239000000262 estrogen Substances 0.000 description 5
- 230000035558 fertility Effects 0.000 description 5
- 150000002333 glycines Chemical group 0.000 description 5
- 238000001794 hormone therapy Methods 0.000 description 5
- 102000055357 human CXCR2 Human genes 0.000 description 5
- 208000015181 infectious disease Diseases 0.000 description 5
- 102000010681 interleukin-8 receptors Human genes 0.000 description 5
- 238000002156 mixing Methods 0.000 description 5
- 238000006386 neutralization reaction Methods 0.000 description 5
- 230000007170 pathology Effects 0.000 description 5
- 238000002823 phage display Methods 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 5
- 210000002460 smooth muscle Anatomy 0.000 description 5
- 239000011780 sodium chloride Substances 0.000 description 5
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 4
- IVLXQGJVBGMLRR-UHFFFAOYSA-N 2-aminoacetic acid;hydron;chloride Chemical compound Cl.NCC(O)=O IVLXQGJVBGMLRR-UHFFFAOYSA-N 0.000 description 4
- 239000007991 ACES buffer Substances 0.000 description 4
- 208000004998 Abdominal Pain Diseases 0.000 description 4
- 241000282472 Canis lupus familiaris Species 0.000 description 4
- 206010014561 Emphysema Diseases 0.000 description 4
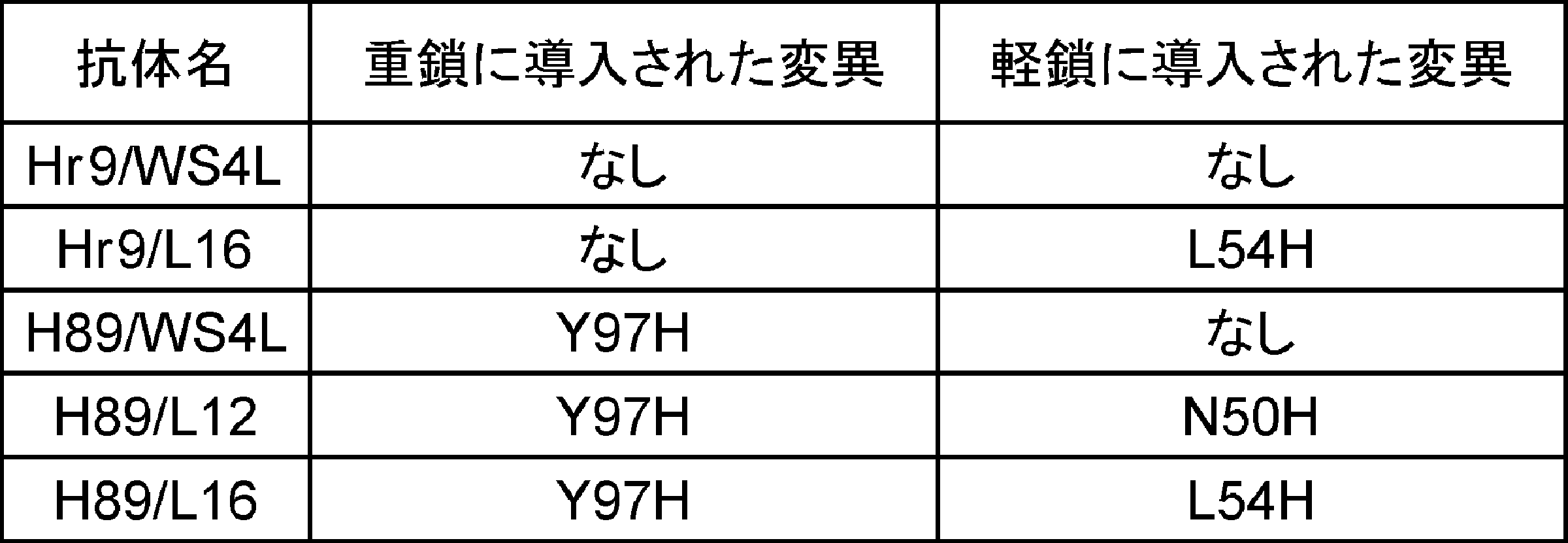
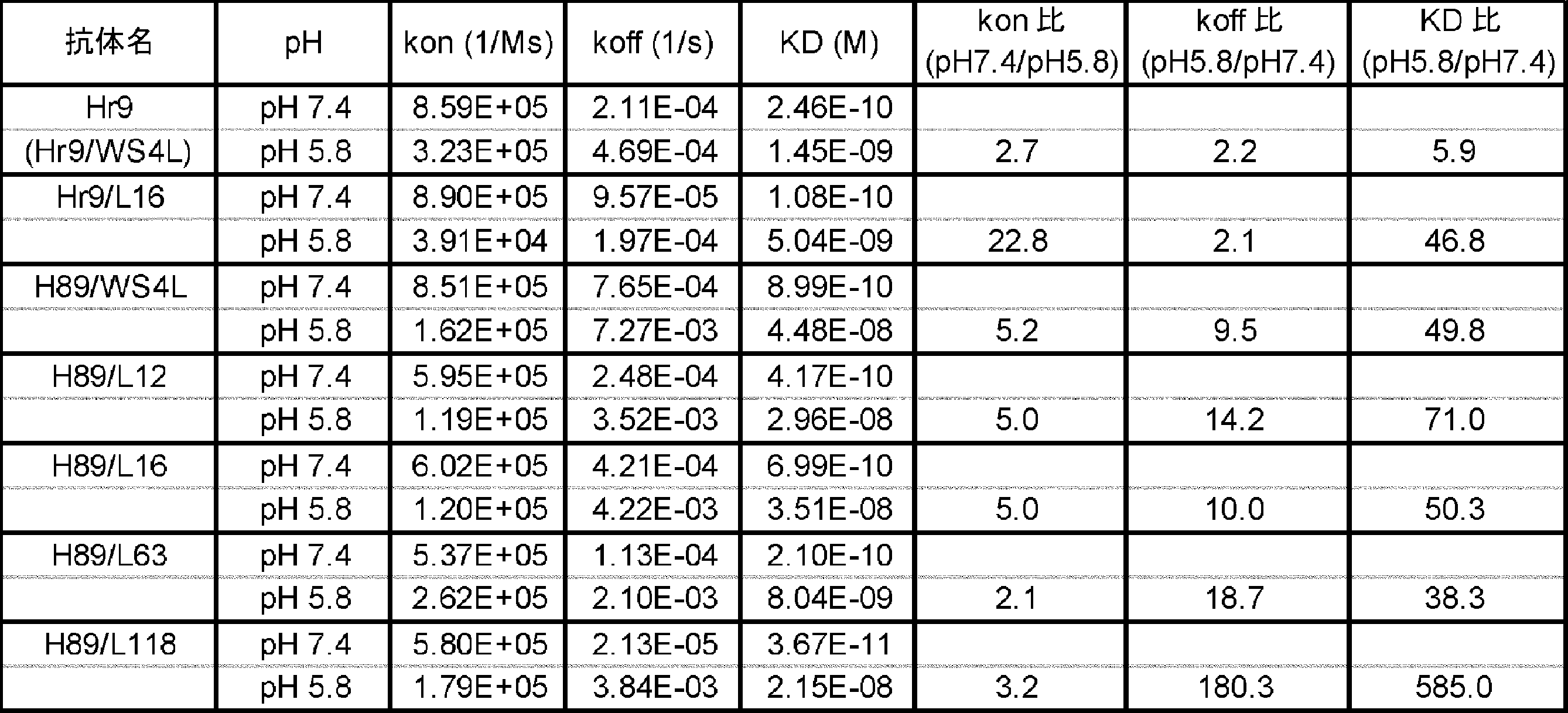
- 102220483804 High mobility group protein B1_Y97H_mutation Human genes 0.000 description 4
- 101000961156 Homo sapiens Immunoglobulin heavy constant gamma 1 Proteins 0.000 description 4
- 102100039345 Immunoglobulin heavy constant gamma 1 Human genes 0.000 description 4
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 4
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 4
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 4
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical group C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 4
- 206010028980 Neoplasm Diseases 0.000 description 4
- 206010037575 Pustular psoriasis Diseases 0.000 description 4
- 108010003723 Single-Domain Antibodies Proteins 0.000 description 4
- 108010090804 Streptavidin Proteins 0.000 description 4
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 4
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 4
- 210000000683 abdominal cavity Anatomy 0.000 description 4
- 238000002835 absorbance Methods 0.000 description 4
- 150000001412 amines Chemical class 0.000 description 4
- 229940125644 antibody drug Drugs 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 210000000038 chest Anatomy 0.000 description 4
- 208000011829 combined pulmonary fibrosis-emphysema syndrome Diseases 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 238000010168 coupling process Methods 0.000 description 4
- 235000018417 cysteine Nutrition 0.000 description 4
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 238000011161 development Methods 0.000 description 4
- 230000018109 developmental process Effects 0.000 description 4
- 208000027866 inflammatory disease Diseases 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 108010038415 interleukin-8 receptors Proteins 0.000 description 4
- 238000012004 kinetic exclusion assay Methods 0.000 description 4
- 210000004185 liver Anatomy 0.000 description 4
- 210000004072 lung Anatomy 0.000 description 4
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 4
- 230000007246 mechanism Effects 0.000 description 4
- 239000011259 mixed solution Substances 0.000 description 4
- 239000002773 nucleotide Substances 0.000 description 4
- 125000003729 nucleotide group Chemical group 0.000 description 4
- 150000002482 oligosaccharides Chemical class 0.000 description 4
- 230000036470 plasma concentration Effects 0.000 description 4
- 238000003752 polymerase chain reaction Methods 0.000 description 4
- 230000003449 preventive effect Effects 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 201000010914 pustulosis of palm and sole Diseases 0.000 description 4
- 208000011797 pustulosis palmaris et plantaris Diseases 0.000 description 4
- 239000012088 reference solution Substances 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- 102220124278 rs754450822 Human genes 0.000 description 4
- 238000012216 screening Methods 0.000 description 4
- 229940124597 therapeutic agent Drugs 0.000 description 4
- 230000001960 triggered effect Effects 0.000 description 4
- JQMFQLVAJGZSQS-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-N-(2-oxo-3H-1,3-benzoxazol-6-yl)acetamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)CC(=O)NC1=CC2=C(NC(O2)=O)C=C1 JQMFQLVAJGZSQS-UHFFFAOYSA-N 0.000 description 3
- 208000023275 Autoimmune disease Diseases 0.000 description 3
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 208000008439 Biliary Liver Cirrhosis Diseases 0.000 description 3
- 208000033222 Biliary cirrhosis primary Diseases 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 208000032843 Hemorrhage Diseases 0.000 description 3
- 206010019728 Hepatitis alcoholic Diseases 0.000 description 3
- 201000003838 Idiopathic interstitial pneumonia Diseases 0.000 description 3
- 102100029185 Low affinity immunoglobulin gamma Fc region receptor III-B Human genes 0.000 description 3
- 241000282560 Macaca mulatta Species 0.000 description 3
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 3
- OVRNDRQMDRJTHS-UHFFFAOYSA-N N-acelyl-D-glucosamine Natural products CC(=O)NC1C(O)OC(CO)C(O)C1O OVRNDRQMDRJTHS-UHFFFAOYSA-N 0.000 description 3
- MBLBDJOUHNCFQT-LXGUWJNJSA-N N-acetylglucosamine Natural products CC(=O)N[C@@H](C=O)[C@@H](O)[C@H](O)[C@H](O)CO MBLBDJOUHNCFQT-LXGUWJNJSA-N 0.000 description 3
- 208000000450 Pelvic Pain Diseases 0.000 description 3
- 208000012654 Primary biliary cholangitis Diseases 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 3
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Chemical group CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 3
- 208000002353 alcoholic hepatitis Diseases 0.000 description 3
- 208000007502 anemia Diseases 0.000 description 3
- 238000010171 animal model Methods 0.000 description 3
- 230000003110 anti-inflammatory effect Effects 0.000 description 3
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 description 3
- 210000004899 c-terminal region Anatomy 0.000 description 3
- 210000000349 chromosome Anatomy 0.000 description 3
- 230000004540 complement-dependent cytotoxicity Effects 0.000 description 3
- 229920001577 copolymer Polymers 0.000 description 3
- 239000012228 culture supernatant Substances 0.000 description 3
- 238000012258 culturing Methods 0.000 description 3
- 238000010790 dilution Methods 0.000 description 3
- 239000012895 dilution Substances 0.000 description 3
- 210000004696 endometrium Anatomy 0.000 description 3
- 229940088598 enzyme Drugs 0.000 description 3
- 210000002950 fibroblast Anatomy 0.000 description 3
- 239000012634 fragment Substances 0.000 description 3
- 239000001963 growth medium Substances 0.000 description 3
- 238000009396 hybridization Methods 0.000 description 3
- 230000001900 immune effect Effects 0.000 description 3
- 238000000126 in silico method Methods 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 208000014674 injury Diseases 0.000 description 3
- 238000007912 intraperitoneal administration Methods 0.000 description 3
- 230000002427 irreversible effect Effects 0.000 description 3
- 238000001155 isoelectric focusing Methods 0.000 description 3
- 208000007106 menorrhagia Diseases 0.000 description 3
- 210000000651 myofibroblast Anatomy 0.000 description 3
- 229920001542 oligosaccharide Polymers 0.000 description 3
- 230000016087 ovulation Effects 0.000 description 3
- 230000027758 ovulation cycle Effects 0.000 description 3
- 210000004197 pelvis Anatomy 0.000 description 3
- 239000002243 precursor Substances 0.000 description 3
- 230000035935 pregnancy Effects 0.000 description 3
- 239000003755 preservative agent Substances 0.000 description 3
- 239000000186 progesterone Substances 0.000 description 3
- 229960003387 progesterone Drugs 0.000 description 3
- 238000004393 prognosis Methods 0.000 description 3
- 230000002062 proliferating effect Effects 0.000 description 3
- 238000012552 review Methods 0.000 description 3
- 102200114090 rs121965037 Human genes 0.000 description 3
- 229910052707 ruthenium Inorganic materials 0.000 description 3
- 150000003354 serine derivatives Chemical group 0.000 description 3
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 230000001629 suppression Effects 0.000 description 3
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 3
- 210000000115 thoracic cavity Anatomy 0.000 description 3
- 230000009261 transgenic effect Effects 0.000 description 3
- 210000003462 vein Anatomy 0.000 description 3
- -1 αalpha Proteins 0.000 description 3
- 206010000084 Abdominal pain lower Diseases 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 208000028685 Asherman syndrome Diseases 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- 102100036153 C-X-C motif chemokine 6 Human genes 0.000 description 2
- 108010014423 Chemokine CXCL6 Proteins 0.000 description 2
- 102000012605 Cystic Fibrosis Transmembrane Conductance Regulator Human genes 0.000 description 2
- 108010079245 Cystic Fibrosis Transmembrane Conductance Regulator Proteins 0.000 description 2
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 2
- 229920002307 Dextran Polymers 0.000 description 2
- 206010061818 Disease progression Diseases 0.000 description 2
- 108010021468 Fc gamma receptor IIA Proteins 0.000 description 2
- 102000003886 Glycoproteins Human genes 0.000 description 2
- 108090000288 Glycoproteins Proteins 0.000 description 2
- 208000002777 Gynatresia Diseases 0.000 description 2
- 206010019799 Hepatitis viral Diseases 0.000 description 2
- 101000937544 Homo sapiens Beta-2-microglobulin Proteins 0.000 description 2
- 101100441536 Homo sapiens CXCR1 gene Proteins 0.000 description 2
- 108010073807 IgG Receptors Proteins 0.000 description 2
- 108090001005 Interleukin-6 Proteins 0.000 description 2
- 102000004889 Interleukin-6 Human genes 0.000 description 2
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 2
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 2
- 125000002068 L-phenylalanino group Chemical group [H]OC(=O)[C@@]([H])(N([H])[*])C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- 239000007987 MES buffer Substances 0.000 description 2
- OVRNDRQMDRJTHS-RTRLPJTCSA-N N-acetyl-D-glucosamine Chemical compound CC(=O)N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-RTRLPJTCSA-N 0.000 description 2
- 102000016387 Pancreatic elastase Human genes 0.000 description 2
- 108010067372 Pancreatic elastase Proteins 0.000 description 2
- 102000007079 Peptide Fragments Human genes 0.000 description 2
- 108010033276 Peptide Fragments Proteins 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Natural products OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 206010035664 Pneumonia Diseases 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- NBBJYMSMWIIQGU-UHFFFAOYSA-N Propionic aldehyde Chemical compound CCC=O NBBJYMSMWIIQGU-UHFFFAOYSA-N 0.000 description 2
- 206010037888 Rash pustular Diseases 0.000 description 2
- 108020004459 Small interfering RNA Proteins 0.000 description 2
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 2
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- 206010046798 Uterine leiomyoma Diseases 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 230000003213 activating effect Effects 0.000 description 2
- 238000001042 affinity chromatography Methods 0.000 description 2
- 229940035676 analgesics Drugs 0.000 description 2
- 239000000730 antalgic agent Substances 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 102000015736 beta 2-Microglobulin Human genes 0.000 description 2
- 108010081355 beta 2-Microglobulin Proteins 0.000 description 2
- 102000000072 beta-Arrestins Human genes 0.000 description 2
- 108010080367 beta-Arrestins Proteins 0.000 description 2
- 229960002685 biotin Drugs 0.000 description 2
- 235000020958 biotin Nutrition 0.000 description 2
- 239000011616 biotin Substances 0.000 description 2
- 208000034158 bleeding Diseases 0.000 description 2
- 231100000319 bleeding Toxicity 0.000 description 2
- 230000000740 bleeding effect Effects 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- YCIMNLLNPGFGHC-UHFFFAOYSA-N catechol Chemical compound OC1=CC=CC=C1O YCIMNLLNPGFGHC-UHFFFAOYSA-N 0.000 description 2
- 238000004113 cell culture Methods 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 208000026106 cerebrovascular disease Diseases 0.000 description 2
- 208000037893 chronic inflammatory disorder Diseases 0.000 description 2
- 208000020832 chronic kidney disease Diseases 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 238000004590 computer program Methods 0.000 description 2
- 230000002596 correlated effect Effects 0.000 description 2
- 210000002726 cyst fluid Anatomy 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000001212 derivatisation Methods 0.000 description 2
- 206010012601 diabetes mellitus Diseases 0.000 description 2
- 238000000502 dialysis Methods 0.000 description 2
- 230000006806 disease prevention Effects 0.000 description 2
- 230000005750 disease progression Effects 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 210000003162 effector t lymphocyte Anatomy 0.000 description 2
- 210000002889 endothelial cell Anatomy 0.000 description 2
- 230000002708 enhancing effect Effects 0.000 description 2
- 239000013604 expression vector Substances 0.000 description 2
- 210000003754 fetus Anatomy 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- 230000013595 glycosylation Effects 0.000 description 2
- 238000006206 glycosylation reaction Methods 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 229920001519 homopolymer Polymers 0.000 description 2
- 102000047279 human B2M Human genes 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- 230000036039 immunity Effects 0.000 description 2
- 230000002163 immunogen Effects 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- XKTZWUACRZHVAN-VADRZIEHSA-N interleukin-8 Chemical compound C([C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@@H](NC(C)=O)CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CCSC)C(=O)N1[C@H](CCC1)C(=O)N1[C@H](CCC1)C(=O)N[C@@H](C)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC=1C=CC(O)=CC=1)C(=O)N[C@H](CO)C(=O)N1[C@H](CCC1)C(N)=O)C1=CC=CC=C1 XKTZWUACRZHVAN-VADRZIEHSA-N 0.000 description 2
- 229940096397 interleukin-8 Drugs 0.000 description 2
- 230000021995 interleukin-8 production Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 238000004255 ion exchange chromatography Methods 0.000 description 2
- 238000012933 kinetic analysis Methods 0.000 description 2
- 238000002350 laparotomy Methods 0.000 description 2
- 201000010260 leiomyoma Diseases 0.000 description 2
- 239000007791 liquid phase Substances 0.000 description 2
- 208000019423 liver disease Diseases 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- 230000004199 lung function Effects 0.000 description 2
- 210000004698 lymphocyte Anatomy 0.000 description 2
- RLSSMJSEOOYNOY-UHFFFAOYSA-N m-cresol Chemical compound CC1=CC=CC(O)=C1 RLSSMJSEOOYNOY-UHFFFAOYSA-N 0.000 description 2
- 238000002595 magnetic resonance imaging Methods 0.000 description 2
- 230000008774 maternal effect Effects 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 201000001441 melanoma Diseases 0.000 description 2
- 230000002175 menstrual effect Effects 0.000 description 2
- 235000013336 milk Nutrition 0.000 description 2
- 210000004080 milk Anatomy 0.000 description 2
- 239000008267 milk Substances 0.000 description 2
- 210000000822 natural killer cell Anatomy 0.000 description 2
- 210000005036 nerve Anatomy 0.000 description 2
- 208000008338 non-alcoholic fatty liver disease Diseases 0.000 description 2
- 230000008506 pathogenesis Effects 0.000 description 2
- 230000007310 pathophysiology Effects 0.000 description 2
- AQIXEPGDORPWBJ-UHFFFAOYSA-N pentan-3-ol Chemical compound CCC(O)CC AQIXEPGDORPWBJ-UHFFFAOYSA-N 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 2
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 2
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 230000000750 progressive effect Effects 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 230000005180 public health Effects 0.000 description 2
- 208000029561 pustule Diseases 0.000 description 2
- 230000001850 reproductive effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- GHMLBKRAJCXXBS-UHFFFAOYSA-N resorcinol Chemical compound OC1=CC=CC(O)=C1 GHMLBKRAJCXXBS-UHFFFAOYSA-N 0.000 description 2
- 229960004641 rituximab Drugs 0.000 description 2
- 102200116209 rs387906935 Human genes 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 238000004659 sterilization and disinfection Methods 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 201000000596 systemic lupus erythematosus Diseases 0.000 description 2
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 description 2
- 238000002054 transplantation Methods 0.000 description 2
- 230000008733 trauma Effects 0.000 description 2
- 230000005740 tumor formation Effects 0.000 description 2
- 201000001862 viral hepatitis Diseases 0.000 description 2
- 229920003169 water-soluble polymer Polymers 0.000 description 2
- 230000029663 wound healing Effects 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- BGJSXRVXTHVRSN-UHFFFAOYSA-N 1,3,5-trioxane Chemical compound C1OCOCO1 BGJSXRVXTHVRSN-UHFFFAOYSA-N 0.000 description 1
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 1
- YQYFEGTYCUQBEI-UHFFFAOYSA-N 1-(2-chloro-3-fluorophenyl)-3-(4-chloro-2-hydroxy-3-piperazin-1-ylsulfonylphenyl)urea Chemical compound C1=CC(Cl)=C(S(=O)(=O)N2CCNCC2)C(O)=C1NC(=O)NC1=CC=CC(F)=C1Cl YQYFEGTYCUQBEI-UHFFFAOYSA-N 0.000 description 1
- VOXZDWNPVJITMN-ZBRFXRBCSA-N 17β-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-ZBRFXRBCSA-N 0.000 description 1
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 1
- ODHCTXKNWHHXJC-VKHMYHEASA-N 5-oxo-L-proline Chemical compound OC(=O)[C@@H]1CCC(=O)N1 ODHCTXKNWHHXJC-VKHMYHEASA-N 0.000 description 1
- CONKBQPVFMXDOV-QHCPKHFHSA-N 6-[(5S)-5-[[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]methyl]-2-oxo-1,3-oxazolidin-3-yl]-3H-1,3-benzoxazol-2-one Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C[C@H]1CN(C(O1)=O)C1=CC2=C(NC(O2)=O)C=C1 CONKBQPVFMXDOV-QHCPKHFHSA-N 0.000 description 1
- 206010000060 Abdominal distension Diseases 0.000 description 1
- 206010071198 Allergic hepatitis Diseases 0.000 description 1
- 241000233788 Arecaceae Species 0.000 description 1
- 206010003445 Ascites Diseases 0.000 description 1
- 201000001320 Atherosclerosis Diseases 0.000 description 1
- 230000003844 B-cell-activation Effects 0.000 description 1
- 208000008035 Back Pain Diseases 0.000 description 1
- 208000009137 Behcet syndrome Diseases 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 102100031151 C-C chemokine receptor type 2 Human genes 0.000 description 1
- 101710149815 C-C chemokine receptor type 2 Proteins 0.000 description 1
- 210000001239 CD8-positive, alpha-beta cytotoxic T lymphocyte Anatomy 0.000 description 1
- 101100289995 Caenorhabditis elegans mac-1 gene Proteins 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 102000016289 Cell Adhesion Molecules Human genes 0.000 description 1
- 108010067225 Cell Adhesion Molecules Proteins 0.000 description 1
- 102000000844 Cell Surface Receptors Human genes 0.000 description 1
- 108010001857 Cell Surface Receptors Proteins 0.000 description 1
- 206010008088 Cerebral artery embolism Diseases 0.000 description 1
- 206010008132 Cerebral thrombosis Diseases 0.000 description 1
- 102000016950 Chemokine CXCL1 Human genes 0.000 description 1
- 108010014419 Chemokine CXCL1 Proteins 0.000 description 1
- 102000009410 Chemokine receptor Human genes 0.000 description 1
- 108050000299 Chemokine receptor Proteins 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 1
- 108091035707 Consensus sequence Proteins 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 208000011231 Crohn disease Diseases 0.000 description 1
- 206010011732 Cyst Diseases 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- SHZGCJCMOBCMKK-UHFFFAOYSA-N D-mannomethylose Natural products CC1OC(O)C(O)C(O)C1O SHZGCJCMOBCMKK-UHFFFAOYSA-N 0.000 description 1
- 230000004544 DNA amplification Effects 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 206010012438 Dermatitis atopic Diseases 0.000 description 1
- 206010012442 Dermatitis contact Diseases 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 208000007342 Diabetic Nephropathies Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 208000010201 Exanthema Diseases 0.000 description 1
- 206010073306 Exposure to radiation Diseases 0.000 description 1
- 108010021472 Fc gamma receptor IIB Proteins 0.000 description 1
- 108010087819 Fc receptors Proteins 0.000 description 1
- 102000009109 Fc receptors Human genes 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- PNNNRSAQSRJVSB-SLPGGIOYSA-N Fucose Natural products C[C@H](O)[C@@H](O)[C@H](O)[C@H](O)C=O PNNNRSAQSRJVSB-SLPGGIOYSA-N 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 206010064571 Gene mutation Diseases 0.000 description 1
- 206010018364 Glomerulonephritis Diseases 0.000 description 1
- 206010018367 Glomerulonephritis chronic Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- NMJREATYWWNIKX-UHFFFAOYSA-N GnRH Chemical compound C1CCC(C(=O)NCC(N)=O)N1C(=O)C(CC(C)C)NC(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)CNC(=O)C(NC(=O)C(CO)NC(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)C(CC=1NC=NC=1)NC(=O)C1NC(=O)CC1)CC1=CC=C(O)C=C1 NMJREATYWWNIKX-UHFFFAOYSA-N 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 208000005176 Hepatitis C Diseases 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 102000008949 Histocompatibility Antigens Class I Human genes 0.000 description 1
- 108010088652 Histocompatibility Antigens Class I Proteins 0.000 description 1
- 101000929495 Homo sapiens Adenosine deaminase Proteins 0.000 description 1
- 101000935587 Homo sapiens Flavin reductase (NADPH) Proteins 0.000 description 1
- 101001046686 Homo sapiens Integrin alpha-M Proteins 0.000 description 1
- 101001076408 Homo sapiens Interleukin-6 Proteins 0.000 description 1
- 101000878605 Homo sapiens Low affinity immunoglobulin epsilon Fc receptor Proteins 0.000 description 1
- 101000917839 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-B Proteins 0.000 description 1
- 108010003272 Hyaluronate lyase Proteins 0.000 description 1
- 102000001974 Hyaluronidases Human genes 0.000 description 1
- 206010020649 Hyperkeratosis Diseases 0.000 description 1
- 206010020880 Hypertrophy Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 description 1
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 description 1
- 238000012404 In vitro experiment Methods 0.000 description 1
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 1
- 102100022338 Integrin alpha-M Human genes 0.000 description 1
- 108010002352 Interleukin-1 Proteins 0.000 description 1
- 102100026236 Interleukin-8 Human genes 0.000 description 1
- 108010018976 Interleukin-8A Receptors Proteins 0.000 description 1
- 208000029523 Interstitial Lung disease Diseases 0.000 description 1
- 201000001429 Intracranial Thrombosis Diseases 0.000 description 1
- 206010060840 Ischaemic cerebral infarction Diseases 0.000 description 1
- 208000001126 Keratosis Diseases 0.000 description 1
- 208000008839 Kidney Neoplasms Diseases 0.000 description 1
- 108010092694 L-Selectin Proteins 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- SHZGCJCMOBCMKK-DHVFOXMCSA-N L-fucopyranose Chemical compound C[C@@H]1OC(O)[C@@H](O)[C@H](O)[C@@H]1O SHZGCJCMOBCMKK-DHVFOXMCSA-N 0.000 description 1
- 125000003290 L-leucino group Chemical group [H]OC(=O)[C@@]([H])(N([H])[*])C([H])([H])C(C([H])([H])[H])([H])C([H])([H])[H] 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- LRQKBLKVPFOOQJ-YFKPBYRVSA-N L-norleucine Chemical compound CCCC[C@H]([NH3+])C([O-])=O LRQKBLKVPFOOQJ-YFKPBYRVSA-N 0.000 description 1
- 125000000174 L-prolyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])([H])[C@@]1([H])C(*)=O 0.000 description 1
- 102000016551 L-selectin Human genes 0.000 description 1
- 102100038007 Low affinity immunoglobulin epsilon Fc receptor Human genes 0.000 description 1
- 102100029204 Low affinity immunoglobulin gamma Fc region receptor II-a Human genes 0.000 description 1
- 102100029205 Low affinity immunoglobulin gamma Fc region receptor II-b Human genes 0.000 description 1
- 102100029193 Low affinity immunoglobulin gamma Fc region receptor III-A Human genes 0.000 description 1
- 101710099301 Low affinity immunoglobulin gamma Fc region receptor III-A Proteins 0.000 description 1
- 208000019693 Lung disease Diseases 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 102000043131 MHC class II family Human genes 0.000 description 1
- 108091054438 MHC class II family Proteins 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 208000003289 Meconium Ileus Diseases 0.000 description 1
- 206010054949 Metaplasia Diseases 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- OVRNDRQMDRJTHS-FMDGEEDCSA-N N-acetyl-beta-D-glucosamine Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-FMDGEEDCSA-N 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 241000238413 Octopus Species 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010033603 Pancreatic atrophy Diseases 0.000 description 1
- 208000035467 Pancreatic insufficiency Diseases 0.000 description 1
- 241000282520 Papio Species 0.000 description 1
- 206010033733 Papule Diseases 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 206010057249 Phagocytosis Diseases 0.000 description 1
- 241000233805 Phoenix Species 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 208000002151 Pleural effusion Diseases 0.000 description 1
- 206010060932 Postoperative adhesion Diseases 0.000 description 1
- 206010036790 Productive cough Diseases 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 108010076504 Protein Sorting Signals Proteins 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- ODHCTXKNWHHXJC-GSVOUGTGSA-N Pyroglutamic acid Natural products OC(=O)[C@H]1CCC(=O)N1 ODHCTXKNWHHXJC-GSVOUGTGSA-N 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 208000001647 Renal Insufficiency Diseases 0.000 description 1
- 206010038389 Renal cancer Diseases 0.000 description 1
- 206010063837 Reperfusion injury Diseases 0.000 description 1
- 208000004756 Respiratory Insufficiency Diseases 0.000 description 1
- 206010057190 Respiratory tract infections Diseases 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- 206010040047 Sepsis Diseases 0.000 description 1
- 206010040070 Septic Shock Diseases 0.000 description 1
- 108010071390 Serum Albumin Proteins 0.000 description 1
- 102000007562 Serum Albumin Human genes 0.000 description 1
- 101000857870 Squalus acanthias Gonadoliberin Proteins 0.000 description 1
- 208000005718 Stomach Neoplasms Diseases 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 208000032851 Subarachnoid Hemorrhage Diseases 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 101710120037 Toxin CcdB Proteins 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 208000007814 Unstable Angina Diseases 0.000 description 1
- 206010061401 Uterine injury Diseases 0.000 description 1
- 101150117115 V gene Proteins 0.000 description 1
- 208000036142 Viral infection Diseases 0.000 description 1
- 206010067505 Vulvovaginal adhesion Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 238000012452 Xenomouse strains Methods 0.000 description 1
- 210000003815 abdominal wall Anatomy 0.000 description 1
- 238000005299 abrasion Methods 0.000 description 1
- ODHCTXKNWHHXJC-UHFFFAOYSA-N acide pyroglutamique Natural products OC(=O)C1CCC(=O)N1 ODHCTXKNWHHXJC-UHFFFAOYSA-N 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 206010069351 acute lung injury Diseases 0.000 description 1
- 206010000891 acute myocardial infarction Diseases 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 238000005377 adsorption chromatography Methods 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 229960000548 alemtuzumab Drugs 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- 239000002870 angiogenesis inducing agent Substances 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 238000010913 antigen-directed enzyme pro-drug therapy Methods 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 208000006673 asthma Diseases 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- 230000002238 attenuated effect Effects 0.000 description 1
- 208000025341 autosomal recessive disease Diseases 0.000 description 1
- 229960000686 benzalkonium chloride Drugs 0.000 description 1
- 229960001950 benzethonium chloride Drugs 0.000 description 1
- UREZNYTWGJKWBI-UHFFFAOYSA-M benzethonium chloride Chemical compound [Cl-].C1=CC(C(C)(C)CC(C)(C)C)=CC=C1OCCOCC[N+](C)(C)CC1=CC=CC=C1 UREZNYTWGJKWBI-UHFFFAOYSA-M 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- CADWTSSKOVRVJC-UHFFFAOYSA-N benzyl(dimethyl)azanium;chloride Chemical compound [Cl-].C[NH+](C)CC1=CC=CC=C1 CADWTSSKOVRVJC-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- SQVRNKJHWKZAKO-UHFFFAOYSA-N beta-N-Acetyl-D-neuraminic acid Natural products CC(=O)NC1C(O)CC(O)(C(O)=O)OC1C(O)C(O)CO SQVRNKJHWKZAKO-UHFFFAOYSA-N 0.000 description 1
- 238000007413 biotinylation Methods 0.000 description 1
- HUTDDBSSHVOYJR-UHFFFAOYSA-H bis[(2-oxo-1,3,2$l^{5},4$l^{2}-dioxaphosphaplumbetan-2-yl)oxy]lead Chemical compound [Pb+2].[Pb+2].[Pb+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O HUTDDBSSHVOYJR-UHFFFAOYSA-H 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 239000010839 body fluid Substances 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 210000000621 bronchi Anatomy 0.000 description 1
- 206010006451 bronchitis Diseases 0.000 description 1
- LRHPLDYGYMQRHN-UHFFFAOYSA-N butyl alcohol Substances CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- ZTQSAGDEMFDKMZ-UHFFFAOYSA-N butyric aldehyde Natural products CCCC=O ZTQSAGDEMFDKMZ-UHFFFAOYSA-N 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 230000003185 calcium uptake Effects 0.000 description 1
- 229940112129 campath Drugs 0.000 description 1
- 238000005251 capillar electrophoresis Methods 0.000 description 1
- 239000002775 capsule Substances 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000004700 cellular uptake Effects 0.000 description 1
- 206010008118 cerebral infarction Diseases 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 230000035605 chemotaxis Effects 0.000 description 1
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 1
- 235000019504 cigarettes Nutrition 0.000 description 1
- 230000007882 cirrhosis Effects 0.000 description 1
- 230000004186 co-expression Effects 0.000 description 1
- 210000003022 colostrum Anatomy 0.000 description 1
- 235000021277 colostrum Nutrition 0.000 description 1
- 238000004891 communication Methods 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 208000010247 contact dermatitis Diseases 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 230000000875 corresponding effect Effects 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 208000031513 cyst Diseases 0.000 description 1
- 210000000805 cytoplasm Anatomy 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 229940127089 cytotoxic agent Drugs 0.000 description 1
- 239000002254 cytotoxic agent Substances 0.000 description 1
- 231100000599 cytotoxic agent Toxicity 0.000 description 1
- 229960002806 daclizumab Drugs 0.000 description 1
- POZRVZJJTULAOH-LHZXLZLDSA-N danazol Chemical compound C1[C@]2(C)[C@H]3CC[C@](C)([C@](CC4)(O)C#C)[C@@H]4[C@@H]3CCC2=CC2=C1C=NO2 POZRVZJJTULAOH-LHZXLZLDSA-N 0.000 description 1
- 229960000766 danazol Drugs 0.000 description 1
- 230000013872 defecation Effects 0.000 description 1
- 210000004443 dendritic cell Anatomy 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 208000033679 diabetic kidney disease Diseases 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 208000018459 dissociative disease Diseases 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- 125000002228 disulfide group Chemical group 0.000 description 1
- 230000003828 downregulation Effects 0.000 description 1
- 230000002500 effect on skin Effects 0.000 description 1
- 229950006357 elubrixin Drugs 0.000 description 1
- 210000003989 endothelium vascular Anatomy 0.000 description 1
- 230000007515 enzymatic degradation Effects 0.000 description 1
- 229930182833 estradiol Natural products 0.000 description 1
- 210000003527 eukaryotic cell Anatomy 0.000 description 1
- 230000005713 exacerbation Effects 0.000 description 1
- 201000005884 exanthem Diseases 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 210000000630 fibrocyte Anatomy 0.000 description 1
- 230000005714 functional activity Effects 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 206010017758 gastric cancer Diseases 0.000 description 1
- 238000001641 gel filtration chromatography Methods 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 210000004392 genitalia Anatomy 0.000 description 1
- 208000005017 glioblastoma Diseases 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 210000002443 helper t lymphocyte Anatomy 0.000 description 1
- 230000002008 hemorrhagic effect Effects 0.000 description 1
- 208000002672 hepatitis B Diseases 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 239000000833 heterodimer Substances 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 125000000487 histidyl group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C([H])=N1 0.000 description 1
- 239000000710 homodimer Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 102000043395 human ADA Human genes 0.000 description 1
- 102000052624 human CXCL8 Human genes 0.000 description 1
- 102000052611 human IL6 Human genes 0.000 description 1
- 229960002773 hyaluronidase Drugs 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 201000001421 hyperglycemia Diseases 0.000 description 1
- 208000022168 hypermenorrhea Diseases 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 230000003053 immunization Effects 0.000 description 1
- 238000002649 immunization Methods 0.000 description 1
- 230000016784 immunoglobulin production Effects 0.000 description 1
- 238000001114 immunoprecipitation Methods 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 231100000824 inhalation exposure Toxicity 0.000 description 1
- 208000036971 interstitial lung disease 2 Diseases 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 208000003243 intestinal obstruction Diseases 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 201000010849 intracranial embolism Diseases 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 208000028867 ischemia Diseases 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 230000007803 itching Effects 0.000 description 1
- 210000002510 keratinocyte Anatomy 0.000 description 1
- 201000010982 kidney cancer Diseases 0.000 description 1
- 208000017169 kidney disease Diseases 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 238000002357 laparoscopic surgery Methods 0.000 description 1
- 230000033001 locomotion Effects 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- 239000012931 lyophilized formulation Substances 0.000 description 1
- 210000003712 lysosome Anatomy 0.000 description 1
- 230000001868 lysosomic effect Effects 0.000 description 1
- 108010026228 mRNA guanylyltransferase Proteins 0.000 description 1
- 206010025482 malaise Diseases 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 235000008409 marco Nutrition 0.000 description 1
- 244000078446 marco Species 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 210000002752 melanocyte Anatomy 0.000 description 1
- 230000009245 menopause Effects 0.000 description 1
- 230000015689 metaplastic ossification Effects 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 210000000110 microvilli Anatomy 0.000 description 1
- 238000010232 migration assay Methods 0.000 description 1
- 238000002715 modification method Methods 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 210000003097 mucus Anatomy 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 230000003387 muscular Effects 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 231100000350 mutagenesis Toxicity 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 208000031225 myocardial ischemia Diseases 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 229950006780 n-acetylglucosamine Drugs 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 230000002956 necrotizing effect Effects 0.000 description 1
- 210000004126 nerve fiber Anatomy 0.000 description 1
- 210000002569 neuron Anatomy 0.000 description 1
- 230000011242 neutrophil chemotaxis Effects 0.000 description 1
- 206010053219 non-alcoholic steatohepatitis Diseases 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 238000011580 nude mouse model Methods 0.000 description 1
- 230000002611 ovarian Effects 0.000 description 1
- 229940094443 oxytocics prostaglandins Drugs 0.000 description 1
- LXCFILQKKLGQFO-UHFFFAOYSA-N p-hydroxybenzoic acid methyl ester Natural products COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 1
- 210000001819 pancreatic juice Anatomy 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 210000005259 peripheral blood Anatomy 0.000 description 1
- 239000011886 peripheral blood Substances 0.000 description 1
- 206010034674 peritonitis Diseases 0.000 description 1
- 230000008782 phagocytosis Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229940127557 pharmaceutical product Drugs 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 230000008884 pinocytosis Effects 0.000 description 1
- 239000000902 placebo Substances 0.000 description 1
- 229940068196 placebo Drugs 0.000 description 1
- 210000002826 placenta Anatomy 0.000 description 1
- 230000010118 platelet activation Effects 0.000 description 1
- 238000007747 plating Methods 0.000 description 1
- 210000003281 pleural cavity Anatomy 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 229920000191 poly(N-vinyl pyrrolidone) Polymers 0.000 description 1
- 229920001308 poly(aminoacid) Polymers 0.000 description 1
- 229920001583 poly(oxyethylated polyols) Polymers 0.000 description 1
- 102000040430 polynucleotide Human genes 0.000 description 1
- 108091033319 polynucleotide Proteins 0.000 description 1
- 239000002157 polynucleotide Substances 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 238000010837 poor prognosis Methods 0.000 description 1
- 230000002980 postoperative effect Effects 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000006013 primary lung bud formation Effects 0.000 description 1
- 238000011809 primate model Methods 0.000 description 1
- 210000001236 prokaryotic cell Anatomy 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 230000035752 proliferative phase Effects 0.000 description 1
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 150000003180 prostaglandins Chemical class 0.000 description 1
- 238000000159 protein binding assay Methods 0.000 description 1
- 230000004850 protein–protein interaction Effects 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 229920005604 random copolymer Polymers 0.000 description 1
- 206010037844 rash Diseases 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000008707 rearrangement Effects 0.000 description 1
- 238000003259 recombinant expression Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 238000004064 recycling Methods 0.000 description 1
- 230000010410 reperfusion Effects 0.000 description 1
- 201000004193 respiratory failure Diseases 0.000 description 1
- 208000023504 respiratory system disease Diseases 0.000 description 1
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 1
- 238000004007 reversed phase HPLC Methods 0.000 description 1
- 102220202173 rs148450358 Human genes 0.000 description 1
- 238000005185 salting out Methods 0.000 description 1
- 238000007790 scraping Methods 0.000 description 1
- 238000006748 scratching Methods 0.000 description 1
- 230000002393 scratching effect Effects 0.000 description 1
- 230000036303 septic shock Effects 0.000 description 1
- 238000013207 serial dilution Methods 0.000 description 1
- SQVRNKJHWKZAKO-OQPLDHBCSA-N sialic acid Chemical compound CC(=O)N[C@@H]1[C@@H](O)C[C@@](O)(C(O)=O)OC1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-OQPLDHBCSA-N 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 239000000779 smoke Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 210000003802 sputum Anatomy 0.000 description 1
- 208000024794 sputum Diseases 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- SFVFIFLLYFPGHH-UHFFFAOYSA-M stearalkonium chloride Chemical compound [Cl-].CCCCCCCCCCCCCCCCCC[N+](C)(C)CC1=CC=CC=C1 SFVFIFLLYFPGHH-UHFFFAOYSA-M 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 230000000707 stereoselective effect Effects 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 201000011549 stomach cancer Diseases 0.000 description 1
- 208000003265 stomatitis Diseases 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 230000000153 supplemental effect Effects 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229940126585 therapeutic drug Drugs 0.000 description 1
- 230000008467 tissue growth Effects 0.000 description 1
- 238000011200 topical administration Methods 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 230000005747 tumor angiogenesis Effects 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 238000000108 ultra-filtration Methods 0.000 description 1
- 230000002485 urinary effect Effects 0.000 description 1
- 208000019553 vascular disease Diseases 0.000 description 1
- 210000005166 vasculature Anatomy 0.000 description 1
- 230000009385 viral infection Effects 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 210000001325 yolk sac Anatomy 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
- C07K16/244—Interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/02—Drugs for genital or sexual disorders; Contraceptives for disorders of the vagina
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
- A61P15/08—Drugs for genital or sexual disorders; Contraceptives for gonadal disorders or for enhancing fertility, e.g. inducers of ovulation or of spermatogenesis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2866—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against receptors for cytokines, lymphokines, interferons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
- A61K2039/507—Comprising a combination of two or more separate antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/51—Complete heavy chain or Fd fragment, i.e. VH + CH1
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/515—Complete light chain, i.e. VL + CL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/567—Framework region [FR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Definitions
- This disclosure relates to a composition for treating or preventing IL-8 related diseases.
- Non-Patent Documents 1 and 2 Endometriosis is not only estrogen dependent (Non-Patent Documents 1 and 2), but also known as an inflammatory disease (Non-Patent Document 3).
- Endometriosis is not intended to be limiting, but in one aspect, endometrial tissue present in the uterus develops and grows ectopically (usually in the pelvis, sometimes the abdominal cavity, rarely the thoracic cavity) outside the uterus.
- Disease Although it is not a malignant tumor, it is a disease that progresses with time and causes tumor formation and surrounding adhesion, severe abdominal pain and pelvic pain during menstruation and other times, and infertility (fertility reduction). It occurs mainly in women over the age of 20 years of reproductive age, leading to decreased QOL due to pain, etc., and chronic pain and infertility due to severe adhesions.
- Uterine adenomyosis is not intended to be limiting, but in one aspect is a disease similar to endometriosis, but where the intima develops in the muscular layers within the uterus. Severe menstrual pain, excessive menstruation as well as anemia and chronic pain. (Note that the terms endometriosis and adenomyosis described herein are defined by the description given below in the detailed description.) Conventionally used treatment methods for endometriosis or adenomyosis include analgesics, hormonal therapy, and surgical therapy. Analgesics have limited effects and cannot prevent progression of the pathology.
- Hormonal therapy is effective in reducing pain and delaying disease progression, but it stops fertility during treatment to stop menstruation, and it is highly likely that the disease will progress again if treatment is stopped. Side effects of hormonal agents are problems. In addition, there is no report that fertility improves after administration even by administration of hormonal agents. Even in the case of surgical treatment, there is a high possibility of recurrence of about 50% in 3-5 years (Non-patent Documents 4 and 5), and in order to prevent postoperative recurrence, it is necessary to withstand side effects and continue to take hormonal agents There is.
- IL-8 (Interleukin 8, Interleukin 8) is a kind of chemokine, and is a protein known to have a 72 amino acid type and a 77 amino acid type. IL-8 is also called CXCL8. The IL-8 monomer has two disulfide bridges between cysteine 7 and cysteine 34 and between cysteine 9 and cysteine 50 when expressed in the 72 amino acid form. IL-8 is known to exist as a homodimer in solution, but there is no covalent bond between the molecules of these homodimers, and the two monomeric ⁇ Stabilized by non-covalent interactions between sheets.
- IL-8 is produced from various cells such as peripheral blood monocytes, tissue macrophages, NK cells, fibroblasts, vascular endothelial cells by stimulation with inflammatory cytokines (Non-patent Document 6).
- IL-8 is said to have the effect of mainly activating neutrophils, enhancing the expression of cell adhesion molecules, and enhancing adhesion of neutrophils to vascular endothelial cells.
- IL-8 also has neutrophil chemotaxis, and IL-8 produced in damaged tissues promotes migration of neutrophils attached to vascular endothelial cells to tissues and is associated with neutrophil infiltration Inflammation is caused.
- IL-8 is a potent angiogenic factor for endothelial cells and is also known to be associated with tumor angiogenesis.
- Non-patent Document 7 Non-patent Document 7
- the present invention has been made in view of various situations including the above, and in one non-limiting aspect, the object of the present invention is a novel method using an IL-8 signal inhibitor against IL-8 related diseases. It is to provide a treatment method and the like. More specifically, the present invention provides, in one non-limiting embodiment, a novel therapeutic method using an IL-8 signal inhibitor for a novel IL-8 related disease. Alternatively, in another non-limiting embodiment, the present invention provides a novel therapeutic method using a novel anti-IL-8 antibody against a novel or known IL-8 related disease.
- endometriosis While not necessarily intending to be bound by theory, the present inventors have already reported the true pathology of endometriosis (Odagiri, Fertil Steril. 2009; 92 (5): 1525-31.) It has been proposed to be a concept of chronic inflammatory proliferative disease.
- the pathophysiology of endometriosis is an immune response that follows chronic inflammation and manifests as fibrosis, smooth muscle metaplasia, nerve regeneration, angiogenesis, and the like.
- IL-8 is involved in an important part of the mechanism of chronic inflammation, and pathological and clinical evaluation of drug efficacy (laparoscope and MR Observation).
- the present invention is not limited to the creation of a non-human primate model for elucidating the pathophysiology of endometriosis, etc. It is based on a comprehensive study that deals with basic to animal model clinical practice. Since a true assessment of human uterine-related diseases is not possible in model experiments in rats, mice, and other small animals, we have created surgically-induced endometriosis model cynomolgus monkeys and created cynomolgus monkeys.
- IL-8 a major inflammatory cytokine in endometriosis, as a therapeutic drug that works in the end, it is approached from the anti-inflammatory side, and the pathology is improved by administering an IL-8 signal inhibitor Surprisingly found to do.
- the present inventors improved the adhesion by taking an anti-inflammatory approach to adhesion caused by surgical operation etc. and administering an IL-8 signal inhibitor. Surprisingly found to do.
- the present inventors have conceived that the highly functional anti-IL-8 antibody in the present disclosure is useful for treatment of a novel or known IL-8 related disease and the like.
- a highly functional anti-IL-8 antibody may be a pH-dependent anti-IL-8 antibody (an anti-IL-8 antibody that binds to IL-8 in a pH-dependent manner).
- the pH-dependent anti-IL-8 antibody has at least one or more of the following properties when administered to an individual when compared to a reference antibody: rapidly eliminate IL-8, neutralize IL-8 This is advantageous because it can have stable activity, low immunogenicity, and high expression level.
- such a highly functional anti-IL-8 antibody has an antibody having at least one or more of the following properties: the binding affinity for FcRn at acidic pH is greater than the binding affinity for FcRn in the native Fc region
- An anti-IL-8 antibody comprising an Fc region, an anti-IL-8 antibody comprising an Fc region in which the binding affinity to an existing ADA is lower than the binding affinity of the natural Fc region to an existing ADA
- the invention relates in one specific, non-limiting embodiment to the following: [1] Endometriosis; endometriosis; dysmenorrhea; adhesion; fibrosis; endometriosis containing an isolated anti-IL-8 antibody that binds to human IL-8 as an active ingredient
- a composition for treating or preventing IL-8 related diseases selected from the group consisting of pain in uterine adenomyosis or dysmenorrhea; infertility; and pain due to adhesion, fibrosis or inflammation
- the anti-IL-8 antibody is selected from the group consisting of the following (1) to (7): (1) An anti-IL-8 antibody that binds to IL-8 in a pH-dependent manner, including substitution of at least one amino acid in at least one of the following (a) to (f): (A) HVR-H1, comprising the amino acid sequence of SEQ ID NO: 23, (B) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 24, (C) HVR
- An anti-IL-8 antibody that binds to IL-8 in a pH-dependent manner, including at least one amino acid substitution in at least one of the following (a) to (f): (A) HVR-H1, comprising the amino acid sequence of SEQ ID NO: 23, (B) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 24, (C) HVR-H3 comprising the amino acid sequence of SEQ ID NO: 25, (D) HVR-L1, comprising the amino acid sequence of SEQ ID NO: 26, (E) HVR-L2 comprising the amino acid sequence of SEQ ID NO: 27, and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 28
- the anti-IL-8 antibody includes at least amino acid substitutions at the 9th and 11th arginines of the amino acid sequence of SEQ ID NO: 24, and the 3rd tyrosine of the amino acid sequence of SEQ ID NO: 25, respectively;
- composition according to [1], wherein the anti-IL-8 antibody is selected from the group consisting of (8) to (19) below: (8) (a) includes the amino acid sequence of SEQ ID NO: 23 as HVR-H1, (b) includes the amino acid sequence of SEQ ID NO: 29 as HVR-H2, and (c) includes the amino acid sequence of SEQ ID NO: 30 as HVR-H3.
- the anti-IL-8 antibody according to any one of (1) to (7), which comprises an amino acid sequence; (11) (d) includes the amino acid sequence of SEQ ID NO: 26 as HVR-L1, (e) includes the amino acid sequence of SEQ ID NO: 107 as HVR-L2, and (f) includes the amino acid sequence of SEQ ID NO: 32 as HVR-L3.
- the anti-IL-8 antibody according to any one of (1) to (7), which comprises an amino acid sequence; (12) (a) the amino acid sequence of SEQ ID NO: 23 as HVR-H1, (b) the amino acid sequence of SEQ ID NO: 29 as HVR-H2, and (c) the amino acid of SEQ ID NO: 30 as HVR-H3 D) the amino acid sequence of SEQ ID NO: 26 as HVR-L1, (e) the amino acid sequence of SEQ ID NO: 31 as HVR-L2, and (f) the amino acid sequence of SEQ ID NO: 32 as HVR-L3.
- the anti-IL-8 antibody according to any one of (1) to (7), which comprises an amino acid sequence; (13) (a) the amino acid sequence of SEQ ID NO: 23 as HVR-H1, (b) the amino acid sequence of SEQ ID NO: 24 as HVR-H2, and (c) the amino acid of SEQ ID NO: 30 as HVR-H3 (D) the amino acid sequence of SEQ ID NO: 26 as HVR-L1, (e) the amino acid sequence of SEQ ID NO: 107 as HVR-L2, and (f) the SEQ ID NO: 32 as HVR-L3.
- the anti-IL-8 antibody according to (1) or (2) which comprises the amino acid sequence of [1];
- the heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 34 and the light chain variable region comprising the amino acid sequence of SEQ ID NO: 35, according to any one of (1) to (7) of [1] Anti-IL-8 antibody;
- HVR-H2 including the amino acid sequence of SEQ ID NO: 29,
- HVR-H3 including the amino acid sequence of SEQ ID NO: 30
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 26,
- HVR-H1 including
- the antibody comprises (a) the amino acid sequence of SEQ ID NO: 23 as HVR-H1, and (b) the amino acid of SEQ ID NO: 29 as HVR-H2. (C) the amino acid sequence of SEQ ID NO: 30 as HVR-H3, (d) the amino acid sequence of SEQ ID NO: 26 as HVR-L1, and (e) the amino acid sequence of SEQ ID NO: 31 as HVR-L2.
- amino acid And (f) may be functionally equivalent to an antibody comprising the amino acid sequence of SEQ ID NO: 32 as HVR-L3); (17) (a) HVR-H1 including the amino acid sequence of SEQ ID NO: 23, (b) HVR-H2 including the amino acid sequence of SEQ ID NO: 24, (c) HVR-H3 including the amino acid sequence of SEQ ID NO: 30 (D) HVR-L1 comprising the amino acid sequence of SEQ ID NO: 26, (e) HVR-L2 comprising the amino acid sequence of SEQ ID NO: 107, and (f) HVR-L3 comprising the amino acid sequence of SEQ ID NO: 32 Any one of (1) to (7) of [1], including HVR-H1, HVR-H2, HVR-H3, HVR-L1, HVR-L2, and HVR-L3 each having at least 80% sequence identity (Wherein the antibody comprises (a) the amino acid sequence of SEQ ID NO: 23 as HVR-H1, and (b) the amino acid sequence of SEQ ID NO: 24
- an antibody comprising the amino acid sequence of SEQ ID NO: 32 as HVR-L3 may be functionally equivalent).
- a heavy chain variable region and a light chain variable region each having at least 80% sequence identity with a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 34 and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 35 The anti-IL-8 antibody according to any one of (1) to (7), wherein the antibody comprises a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 34 and SEQ ID NO: And may be functionally equivalent to an antibody comprising a light chain variable region comprising 35 amino acid sequences);
- a composition for treating or preventing IL-8 related diseases selected from the group consisting of pain in uterine adenomyosis or dysmenorrhea; infertility; and pain due to adhesion, fibrosis or inflammation is selected from the group consisting of the following (I) to (VIII): (I) Selected from the group consisting of 235, 236, 239, 327, 330, 331, 431, 428, 434, 436, 438, and 440 as represented by EU numbering An anti-IL-8 antibody comprising an Fc region containing an amino acid substitution at one or more positions; (II) an anti-IL-8 antibody comprising Fc regions containing amino acid substitutions at all positions selected from the group consisting
- a composition for treating or preventing IL-8 related diseases selected from the group consisting of pain in uterine adenomyosis or dysmenorrhea; infertility; and pain due to adhesion, fibrosis or inflammation is selected from the group consisting of the following (A) to (F): (A) an anti-IL-8 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 36 and a light chain comprising the amino acid sequence of SEQ ID NO: 38; (B) an anti-IL-8 antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 37 and a light chain comprising the amino acid sequence of SEQ ID NO: 38; (C) an anti-IL-8 antibody comprising a heavy chain comprising
- the antibody may be functionally equivalent to an antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 36 and a light chain comprising the amino acid sequence of SEQ ID NO: 38); (E) an anti-IL-8 antibody comprising a heavy chain comprising at least 80% sequence identity with a heavy chain comprising the amino acid sequence of SEQ ID NO: 37 and a light chain comprising the amino acid sequence of SEQ ID NO: 38, respectively.
- the antibody may be functionally equivalent to an antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 37 and a light chain comprising the amino acid sequence of SEQ ID NO: 38.); (F) an anti-IL-8 antibody comprising a heavy chain comprising at least 80% sequence identity to a heavy chain comprising the amino acid sequence of SEQ ID NO: 106 and a light chain comprising the amino acid sequence of SEQ ID NO: 44, respectively.
- the antibody may be functionally equivalent to an antibody comprising a heavy chain comprising the amino acid sequence of SEQ ID NO: 106 and a light chain comprising the amino acid sequence of SEQ ID NO: 44).
- Fibrosis consists of fibrosis in endometriosis or adenomyosis, chronic obstructive pulmonary disease (COPD), cystic fibrosis, psoriasis, liver fibrosis, renal fibrosis, and pulmonary fibrosis
- COPD chronic obstructive pulmonary disease
- IL-8 signal inhibitor preferably human IL-8 signal inhibitor
- a composition for treating or preventing IL-8-related diseases selected from the group consisting of pain in uterine adenomyosis or dysmenorrhea; infertility; and pain due to adhesion, fibrosis or inflammation.
- the composition according to [6] which suppresses infiltration of immune cells into a lesion site of an IL-8 related disease or its surrounding environment.
- the dysmenorrhea is endometriosis or dysmenorrhea having uterine adenomyosis, or dysmenorrhea in which endometriosis or uterine adenomyosis is suspected, [6] to [ 11] The composition according to any one of [11]. [13] The composition according to [6], wherein the adhesion is an adhesion generated after surgery. [14] IL-8 signal inhibitor is an IL-8 inhibitor (preferably human IL-8 inhibitor), CXCR1 inhibitor (preferably human CXCR1 inhibitor), or CXCR2 inhibitor (preferably human CXCR2 inhibitor) The composition according to any one of [6] to [13].
- the IL-8 inhibitor, CXCR1 inhibitor, or CXCR2 inhibitor is an anti-IL-8 antibody (preferably an anti-human IL-8 antibody), an anti-CXCR1 antibody (preferably an anti-human CXCR1 antibody), or an anti-IL, respectively.
- the composition according to [14] which is a CXCR2 antibody (preferably an anti-human CXCR2 antibody).
- composition according to any one of [1] to [5] which does not affect the sexual cycle.
- Dysmenorrhea is endometriosis or dysmenorrhea having uterine adenomyosis, or dysmenorrhea with suspected endometriosis or uterine adenomyosis, [1] to [ [5] The composition according to any one of [5]. [22] The composition according to any one of [1] to [5], wherein the adhesion is an adhesion produced after surgery. [23] The composition according to any one of [1] to [22], wherein the IL-8 related disease is responsive to an IL-8 signal. [24] The composition according to any one of [1] to [23], further comprising a pharmaceutically acceptable carrier.
- an anti-IL-8 antibody defined in any one of [1] to [5] and [16] to [25] or any one of [1] to [5] and [16] to [25] Endometriosis; uterine adenomyosis; dysmenorrhea; adhesion; fibrosis; endometriosis, adenomyosis, comprising administering the described composition to a subject in need thereof Or a method of treating or preventing an IL-8 related disease selected from the group consisting of pain in dysmenorrhea; infertility; and pain due to adhesions, fibrosis or inflammation (where the subject is in need) , Or a subject who is suffering from or may be suffering from the IL-8-related disease.
- IL-8 signal inhibitor defined in any of [6] to [15], [23] to [25] or any of [6] to [15], [23] to [25]
- FIG. 4 shows the degree of binding of Fv4-YTE to rheumatoid factor in the serum of RA patients.
- Fig. 6 shows the degree of binding of Fv4-LS to rheumatoid factor in the serum of RA patients.
- Fig. 4 shows the degree of binding of Fv4-N434H to rheumatoid factor in the serum of RA patients.
- Fig. 6 shows the degree of binding of Fv4-F1847m to rheumatoid factor in the serum of RA patients.
- FIG. 4 shows the degree of binding of Fv4-F1848m to rheumatoid factor in the serum of RA patients.
- FIG. 5 shows the degree of binding of Fv4-F1886m to rheumatoid factor in the serum of RA patients.
- Fig. 4 shows the degree of binding of Fv4-F1889m to rheumatoid factor in the serum of RA patients.
- Fig. 4 shows the degree of binding of Fv4-F1927m to rheumatoid factor in the serum of RA patients.
- Fig. 4 shows the degree of binding of Fv4-F1168m to rheumatoid factor in the serum of RA patients.
- the average value of the binding property to rheumatoid factor in the blood of RA patients is shown for each of the antibodies containing Fv4-IgG1 and various FcRn-binding increased Fc region variants.
- Anti-human IgE antibodies, natural human IgG1 having an Fc region of OHB-IgG1 and antibodies containing various FcRn binding-enhancing Fc region variants are administered to cynomolgus monkeys, respectively, and the changes in the concentrations of various anti-human IgE antibodies in the plasma of the cynomolgus monkeys are shown.
- Fv4-IgG1 which is an anti-human IL-6 receptor antibody
- Fv4-F1718 with increased binding to FcRn at acidic pH in the antibody are each administered to human FcRn transgenic mice
- the change of antibody concentration is shown.
- Sensorgrams obtained when measuring binding of H998 / L63 and Hr9 to IL-8 at pH 7.4 and pH 5.8 with Biacore are shown.
- the time course of human IL-8 concentration in the mouse plasma when H998 / L63 and H89 / L118 mixed with human IL-8 are each administered to mice at 2 mg / kg is shown.
- save in plasma of Hr9, H89 / L118, and H553 / L118 by an antibody concentration is shown.
- save in plasma of Hr9, H89 / L118, and H553 / L118 is shown.
- save in plasma of Hr9, H89 / L118, and H553 / L118 is shown.
- the ADA occurrence prediction frequency of each IL-8 antibody (hWS4, Hr9, H89 / L118, H496 / L118, H553 / L118) predicted by EpiMatrix and the ADA occurrence prediction frequency of other existing antibody drugs are shown.
- Predicted ADA occurrence frequency of each IL-8 antibody (H496 / L118, H496v1 / L118, H496v2 / L118, H496v3 / L118, H1004 / L118, H1004 / L395) predicted by EpiMatrix and other existing antibody drugs Indicates the frequency.
- save in plasma of Hr9, H89 / L118, and H1009 / L395-F1886s is shown.
- save in plasma of Hr9, H89 / L118, and H1009 / L395-F1886s is shown.
- save in plasma of Hr9, H89 / L118, and H1009 / L395-F1886s is shown.
- the transition of human IL-8 concentration in the mouse plasma when H1009 / L395, H553 / L118 and H998 / L63 mixed with human IL-8 are each administered to the mouse is shown.
- the amount of binding to the extracellular matrix when Hr9, H89 / L118 and H1009 / L395 are added alone to the extracellular matrix and when mixed with human IL-8 is shown.
- an antibody having a variable region of H1009 / L395 and having an Fc region that does not bind to FcRn (F1942m) is administered alone to human FcRn transgenic mice or mixed with human IL-8, human FcRn transgenic mice It shows the antibody concentration transition in the plasma of each mouse when administered.
- the ADA occurrence prediction frequency of H1009 / L395 and H1004 / L395 predicted by EpiMatrix and the ADA occurrence prediction frequency of other existing antibody drugs are shown.
- H89 / L118-IgG1 having the variable region of H89 / L118, having the Fc region of natural human IgG1, and antibodies having various FcRn binding-enhancing Fc region variants (H89 / L118-F1168m, H89 / L118-F1847m, H89 / L118-F1848m, H89 / L118-F1886m, H89 / L118-F1889m, H89 / L118-F1927m) were administered to cynomolgus monkeys, respectively. Show.
- FIG. 6 shows changes in the concentration of human IL-8 in the mouse plasma when an anti-IL-8 antibody mixed with human IL-8 is administered to a human FcRn transgenic mouse.
- the anti-IL-8 antibody includes H1009 / L395-IgG1 (2 mg / kg) containing the variable region of H1009 / L395 and the Fc region of natural human IgG1, and the variable region of H1009 / L395 and a modified Fc region.
- H1009 / L395-F1886s (2, 5 or 10 mg / kg).
- 3 is a graph showing changes in total ⁇ r-AFS score, adhesionhesr-AFS score, and size r-AFS score before and after administration of Vehicle or antibody H1009 / L395-F1974m.
- 2 is a graph showing changes in total r-AFS score before and after administration of Vehicle or antibody H1009 / L395-F1974m.
- 12 shows the histopathological image of the transplanted part in the Vehicle group and the antibody H1009 / L395-F1974m administration group 12 months after the attraction (6 months after the administration).
- antibody is used in the broadest sense, and is not limited, but may be a monoclonal antibody, a polyclonal antibody, a multispecific antibody (for example, a bispecific antibody) as long as it exhibits a desired antigen-binding activity. And antibody fragments.
- an antibody that binds to the same epitope with respect to a desired reference antibody (eg, a reference anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody), in one embodiment, refers to a desired antigen.
- a reference antibody that blocks, for example, 50%, 60%, 70%, 80%, 90%, 95% or more binding of a reference antibody that binds to (eg, IL-8, CXCR1, or CXCR2) to the antigen;
- a reference antibody inhibits binding of an antibody to its antigen, for example by 50%, 60%, 70%, 80%, 90%, 95% or more.
- a typical competition assay may be used, but is not limited thereto.
- monoclonal antibody means an antibody obtained from a substantially homogeneous population of antibodies, ie, contains, for example, naturally occurring mutations or occurs during the production of monoclonal antibody formulations. Thus, with the exception of potential variant antibodies, such as variants that are generally present in small amounts, the individual antibodies that make up the population are identical and / or bind to the same epitope. In contrast to polyclonal antibody preparations, which typically contain different antibodies to different determinants (epitopes), each monoclonal antibody of a monoclonal antibody preparation is against a single determinant on the antigen.
- the modifier “monoclonal” indicates the character of the antibody as being obtained from a substantially homogeneous population of antibodies, and is not to be construed as requiring production of the antibody by any particular method.
- monoclonal antibodies in the present disclosure can be obtained by a variety of techniques including, but not limited to, hybridoma methods, recombinant DNA methods, phage display methods, and methods that utilize transgenic animals containing all or part of a human immunoglobulin locus. May be made.
- the antibodies in this disclosure may be monoclonal antibodies.
- natural antibody refers to immunoglobulin molecules having various structures that occur in nature.
- a natural IgG antibody in one aspect is a heterotetrameric glycoprotein of about 150,000 daltons, consisting of, but not limited to, two identical light chains and two identical heavy chains that are disulfide bonded . From the N-terminus to the C-terminus, each heavy chain has a variable region (VH) followed by three constant regions (CH1, CH2 and CH3). Similarly, from the N-terminus to the C-terminus, each light chain has a variable region (VL) followed by a constant region (CL).
- the light chain of an antibody can be assigned to one of two types, called kappa ( ⁇ ) and lambda ( ⁇ ), based on the amino acid sequence of its constant region.
- ⁇ kappa
- ⁇ lambda
- the constant region used here any reported allotype (allele) or any subclass / isotype may be used.
- the heavy chain constant region is not limited, but a constant region of a natural IgG antibody (IgG1, IgG2, IgG3 or IgG4) can be used.
- IgG1, IgG2, IgG3 or IgG4 can be used.
- IGHG1 * 01-05 is known as an allele of IgG1 (see http://www.imgt.org/), and any of these can be used as a human natural IgG1 sequence.
- the constant region sequence may be derived from a single allele or subclass / isotype, or may be derived from a plurality of alleles or subclass / isotypes. That is, although not limited thereto, for example, antibodies such as CH1 derived from IGHG1 * 01, CH2 derived from IGHG1 * 02, and CH3 derived from IGHG1 * 01 are also included.
- Examples of the heavy chain constant region of the natural human IgG antibody include human IgG1 constant region (SEQ ID NO: 100), human IgG2 constant region (SEQ ID NO: 101), human IgG3 constant region (SEQ ID NO: 102), human IgG4 Examples thereof include a constant region (SEQ ID NO: 103).
- Examples of the light chain constant region of a natural human IgG antibody include a human ⁇ chain constant region (SEQ ID NO: 104), a human ⁇ chain constant region (SEQ ID NO: 105), and the like.
- variable region FR refers to variable region portions other than hypervariable region (HVR) residues.
- HVR hypervariable region
- the variable region FR consists of four FR domains: FR1, FR2, FR3, and FR4. Therefore, the HVR and FR sequences usually appear in VH (or VL) as follows: FR1-H1 (L1) -FR2-H2 (L2) -FR3-H3 (L3) -FR4.
- human consensus framework is a framework that represents the amino acid residues that usually appear most commonly in the selection of human immunoglobulin VL or VH framework sequences.
- human immunoglobulin VL or VH sequences are selected from a subgroup of variable region sequences.
- the subgroups of sequences are subgroups by Kabat et al., Sequences, of proteins, Immunological, Interest, 5th, Ed. Public, Health, Service, National Institutes of Health, Bethesda, MD, (1991).
- the subgroup for VL may be subgroup ⁇ I according to Kabat et al listed above.
- the subgroup for VH may be subgroup III by Kabat et al.
- acceptor human framework is a framework containing the amino acid sequence of a VL or VH framework obtained from a human immunoglobulin framework or a human consensus framework.
- An acceptor human framework “derived from” a human immunoglobulin framework or human consensus framework may contain the same amino acid sequence as a human immunoglobulin framework or human consensus framework, or may contain existing amino acid substitutions. Good. In one embodiment, the number of existing amino acid substitutions is 10, 9, 8, 7, 6, 5, 4, 3, or 2 or less.
- the VL acceptor human framework is identical in sequence to the VL human immunoglobulin framework sequence or human consensus framework sequence.
- variable region refers to a heavy or light chain domain of an antibody involved in binding of the antibody to a desired antigen.
- Natural antibody heavy and light chain variable regions typically have a similar structure with each domain containing four conserved framework regions (FRs) and three hypervariable regions (HVRs).
- FRs conserved framework regions
- HVRs hypervariable regions
- one VH or VL domain is sufficient to confer antigen binding specificity, but is not limited thereto.
- antibodies that bind to a particular antigen may be isolated from antibodies that bind to that antigen using a VH or VL domain to screen a complementary library of VL or VH domains (eg, Portolano et al., J. Immunol. 150: 880-887 (1993); Clarkson et al., Nature 1991 352: 624-628).
- hypervariable region is a loop that is hypervariable in sequence (“complementarity determining region” or “CDR”) and / or structurally determined.
- CDR complementarity determining region
- Hypervariable loop refers to each region of an antibody variable domain that forms (and / or contains) an antigen contact residue ("antigen contact”).
- an antibody contains 6 HVRs: 3 in VH (H1, H2, H3) and 3 in VL (L1, L2, L3).
- Exemplary HVRs include the following: (a) at amino acid residues 26-32 (L1), 50-52 (L2), 91-96 (L3), 26-32 (H1), 53-55 (H2), and 96-101 (H3) The resulting hypervariable loop (Chothia and Lesk, J. Mol. Biol. 196: 901-917 (1987)); (b) at amino acid residues 24-34 (L1), 50-56 (L2), 89-97 (L3), 31-35b (H1), 50-65 (H2), and 95-102 (H3) The resulting CDR (Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed.
- HVR residues and other residues in the variable region are numbered herein according to Kabat et al, supra.
- “individual” refers to mammals. Mammals include, but are not limited to, domesticated animals (eg, cattle, sheep, cats, dogs, horses), primates (eg, humans or non-human primates such as monkeys), rabbits, and animals. Includes teeth (such as mice and rats).
- the “individual” is preferably a mammal having IL-8 naturally in vivo, more preferably an animal having menstruation similar to humans, for example, a non-human primate, more preferably a human.
- an individual is used interchangeably with “subject” unless the context contradicts.
- an “isolated” antibody is one that has been separated from components of its natural environment.
- IEF isoelectric focusing
- capillary electrophoresis eg 95% or 99
- An “isolated” antibody in the present disclosure is, in one aspect, paraphrased as a “purified” antibody.
- isolated nucleic acid refers to a nucleic acid molecule that has been separated from a component of its natural environment.
- the nucleic acid usually includes the nucleic acid molecule contained in the cell containing the nucleic acid molecule, but also includes those that exist outside the chromosome and those that exist at a position different from the position of the natural chromosome.
- affinity typically refers to the sum total of non-covalent interactions between a single binding site of a molecule (eg, an antibody or compound) and its binding partner (eg, an antigen). May mean a specific strength.
- binding affinity means an inherent binding affinity that reflects a 1: 1 interaction between members of a binding pair (eg, an antibody or compound and an antigen).
- the affinity of a molecule X for its partner Y can generally be expressed as a dissociation constant (KD). Binding affinity may be measured by methods known to those skilled in the art, such as the methods described in this disclosure.
- the antibody that binds to an antigen such as IL-8, CXCR1 or CXCR2 is, for example, ⁇ 1000 nM, ⁇ 100 nM, ⁇ 10 nM, ⁇ 1 nM, ⁇ 0.1 nM, ⁇ 0.01 nM, or ⁇ 0.001 nM. (For example, 10 ⁇ 8 M or less, 10 ⁇ 8 M to 10 ⁇ 13 M, 10 ⁇ 9 M to 10 ⁇ 13 M, etc.).
- host cell and “host cell line” are used interchangeably and refer to cells into which foreign nucleic acid has been introduced (including progeny of such cells).
- Host cells include “transformants” and “transformed cells”, including the primary transformed cell and progeny derived from that cell regardless of passage number.
- the progeny may not be completely identical in nucleic acid content with the parent cell, and may contain mutations. Also included are mutant progeny that have the same function or biological activity as was used when the original transformed cells were screened or selected.
- vector refers to a nucleic acid molecule capable of amplifying another nucleic acid linked thereto, and is integrated into the vector as a self-replicating nucleic acid structure and the genome of the host cell into which it has been introduced.
- vector can provide for the expression of a nucleic acid operably linked to itself. Such vectors are also referred to herein as “expression vectors”.
- an antibody in the present disclosure may be an antibody fragment.
- the antibody fragment may include, for example, antibody fragments such as Fab, Fab ′, Fab′-SH, F (ab ′) 2 , Fv, scFv fragment, diabody, and single domain antibody. See Hudson et al. Nat. Med. 9: 129-134 (2003) for a review of antibody fragments. For a review of scFv fragments, see, for example, Pluckthun, in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., (Springer-Verlag, New York), pp. 269-315 (1994), WO93 / 16185, US See Patent No. 5,571,894, US Patent No. 5,587,458.
- a “diabody” is an antibody fragment that is bivalent or bispecific and has two antigen binding sites (eg, EP404,097; WO1993 / 01161; Hudson et al., Nat. Med. 9: 129-134 (2003); Hollinger et al., Proc. Natl. Acad. Sci. USA 90: 6644-6448 (1993)). Triabodies and tetrabodies are described, for example, in Hudson et al., Nat. Med. 9: 129-134 (2003).
- the “single domain antibody” is an antibody fragment containing all or part of the heavy chain variable region of the antibody, or all or part of the light chain variable region.
- the antibody in the present disclosure is a single domain antibody, it may be a human single domain antibody (eg, Domantis, Inc., Waltham, MA; US Patent No., 6, 248, 516).
- Antibody fragments can be produced by various methods including, but not limited to, production using the recombinant host cells described herein, such as enzymatic degradation of full length antibodies.
- chimeric antibody refers to an antibody in which a part of the heavy chain and / or light chain is of a specific origin or species and the remaining part is a different origin or species.
- humanized antibody refers to a chimeric antibody having amino acid residues derived from non-human HVR and amino acid residues derived from human FR.
- the humanized antibody comprises substantially at least one, typically two variable regions in which all (or substantially all) HVRs are of the non-human antibody. It corresponds to HVR (eg CDR) and all (or substantially all) FRs correspond to FRs of human antibodies.
- a humanized antibody may optionally include at least a portion of an antibody constant region derived from a human antibody.
- an antibody in the present disclosure may be a chimeric antibody.
- Chimeric antibodies are described, for example, in US Patent No. 4,816,567 and Morrison et al., Proc. Natl. Acad. Sci. USA, 81: 6851-6855 (1984).
- a chimeric antibody may comprise a non-human variable region (eg, a non-human primate such as a monkey, or a variable region derived from a mouse, rat, hamster, or rabbit) and a human constant region.
- an antibody in the present disclosure may be a humanized antibody.
- non-human antibodies are humanized to reduce their immunogenicity in humans while maintaining the specificity and affinity of the parent non-human antibody.
- a humanized antibody contains one or more variable regions, among which are HVRs, such as CDRs (or portions thereof) derived from non-human antibodies, and FRs (or portions thereof) derived from human antibody sequences. Some) and exist.
- a humanized antibody can optionally comprise at least a portion of a human constant region.
- the amino acid residues of FRs in a humanized antibody are those of a non-human antibody (eg, the antibody from which the HVR residue is derived), eg, to maintain or improve antibody specificity or affinity. It may be substituted with the corresponding amino acid residue.
- Humanized antibodies and methods for their production are reviewed, for example, in the following (Almagro and Fransson, Front. Biosci. 13: 1619-1633 (2008)) and are further described, for example: Riechmann et al., Nature 332: 323-329 (1988); Queen et al., Proc. Nat'l Acad. Sci. USA 86: 10029-10033 (1989); US Patent Nos.
- the human framework that would be used for humanization is, for example, a framework selected using the “best fit” method (Sims et al. J. Immunol. 151: 2296 (1993)), A framework derived from the consensus sequence of human antibodies of a particular subgroup with heavy or light chain variable regions (Carter et al. Proc. Natl. Acad. Sci. USA, 89: 4285 (1992) and Presta et al J. Immunol., 151: 26231993 (1993)) and may contain framework regions derived from FR library screening (Baca et al., J. Biol. Chem. 272: 10678-10684 (1997) And Rosok et al., J. Biol. Chem. 271: 22611-22618 (1996)).
- the antibody (eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody) in the present disclosure may be a human antibody.
- Human antibodies can be produced by various techniques. Human antibodies are reviewed, for example, in van Dijk and van de Winkel, Curr. Opin. Pharmacol. 5: 368-374 (2001) and Lonberg, Curr. Opin. Immunol. 20: 450-459 (2008). Human antibodies are prepared by administering an immunogen to a transgenic animal that has been modified to produce fully human antibodies or fully antibodies with human variable regions in response to an antigen (eg, IL-8, CXCR1, or CXCR2). May be.
- an antigen eg, IL-8, CXCR1, or CXCR2. May be.
- Such animals typically include all or part of a human immunoglobulin locus, which all or part of the human immunoglobulin locus replaces the endogenous immunoglobulin locus or is extrachromosomal or It exists in a state of being randomly incorporated in the chromosome of the animal. In such transgenic mice, the endogenous immunoglobulin locus is usually inactivated.
- a human immunoglobulin locus replaces the endogenous immunoglobulin locus or is extrachromosomal or It exists in a state of being randomly incorporated in the chromosome of the animal.
- the endogenous immunoglobulin locus is usually inactivated.
- human antibodies can also be generated by hybridoma-based methods.
- Human myeloma cells and mouse-human heteromyeloma cell lines for the production of human monoclonal antibodies are described below (eg, Kozbor J. Immunol., 133: 3001 (1984); Brodeur et al., Monoclonal Antibody Production). Techniques and Applications, pp. 51-63 (Marcel Dekker, Inc., New York, 1987); and Boerner et al., J. Immunol., 147: 86 (1991)).
- Human antibodies produced via human B cell hybridoma technology are described in Li et al., Proc. Natl. Acad. Sci. USA, 103: 3557-3562 (2006).
- human antibodies can also be generated by isolating Fv clone variable domain sequences selected from human-derived phage display libraries. Such variable region sequences can then be combined with the desired human constant region. See below for techniques to select human antibodies from antibody libraries.
- an antibody in the present disclosure eg, an anti-IL-8 antibody, an anti-CXCR1 antibody, or an anti-CXCR2 antibody
- methods for preparing phage display libraries, methods for screening such libraries for antibodies having desired binding characteristics, and the like are known in the art. Such methods are reviewed in Hoogenboom et al.
- the VH and VL repertoires can be cloned separately by polymerase chain reaction (PCR) and are randomly recombined in a phage library, Et al., Ann. Rev. Immunol., 12: 433-455 (1994) may be screened for antigen-binding phages.
- the phage displays antibody fragments such as scFv and Fab. Libraries from immunized sources can provide high affinity antibodies to the immunogen without the need to construct hybridomas.
- the na ⁇ ve repertoire has been cloned (eg, from a human) and extensively cloned without immunization, as described in Griffiths et al., EMBO J, 12: 725-734 (1993). It is also possible to provide antibodies derived from a single non-self or self-antigen.
- the naive library clones pre-rearranged V-gene segments from stem cells as described in Hoogenboom and Winter, J. Mol. Biol., 227: 381-388 (1992). It can also be made synthetically by using PCR primers that encode the hypervariable region CDR3 and contain random sequences to achieve rearrangement in vitro.
- Patent literatures describing human antibody phage libraries include, for example, US Patent No. 5,750,373, US 2005/0079574, US 2005/0119455, US 2005/0266000, US 2007/0117126, US 2007/0160598, US 2007/0237764, US 2007/0292936, US Can be mentioned.
- An antibody or antibody fragment isolated from a human antibody library is regarded as a human antibody or a human antibody fragment in this specification.
- the antibody (eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody) in this disclosure is a multispecific antibody (eg, bispecific antibody).
- Multispecific antibodies are antibodies (eg, monoclonal antibodies) that have binding specificities at at least two different sites.
- one of the binding specificities is for an antigen (eg, IL-8, CXCR1, or CXCR2) and the other is for any other antigen.
- the bispecific antibody may bind to two different epitopes of an antigen (eg, IL-8, CXCR1, or CXCR2).
- Bispecific antibodies may be used to localize cytotoxic agents to cells that express the antigen (eg, IL-8, CXCR1, or CXCR2).
- Bispecific antibodies may be prepared as full length antibodies or as antibody fragments.
- a method for producing a multispecific antibody is not limited, but includes recombinant co-expression of two immunoglobulin heavy chain-light chain pairs having different specificities (for example, Milstein and Cuello, Nature 305: 537 (1983), WO93). / 08829, and Traunecker et al., EMBO J. 10: 3655 (1991)), and the knob-in-hole technology (eg US Pat. No. 5,731,168).
- Multispecific antibodies are those that manipulate electrostatic steering effects to produce Fc heterodimeric molecules (eg, WO2009 / 089004A1); cross-link two or more antibodies or antibody fragments ( For example, US Pat. No.
- the antibody eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody
- antibody fragment thereof in this disclosure is different from the antigen (eg, IL-8, CXCR1, or CXCR2). It may be a “dual-acting Fab” or “DAF” containing one antigen-binding site that binds to (eg US2008 / 0069820).
- an amino acid sequence variant (variant) of an antibody in the present disclosure has an appropriate modification to the nucleic acid encoding the antibody molecule. It can be prepared by introduction or synthesis of a peptide. Such modification may be performed by appropriately combining one or more arbitrary deletions, insertions, and substitutions of arbitrary amino acids (residues) into the amino acid sequence. Any combination of deletion, insertion, substitution can be used as long as the final construct has the desired characteristics (eg, antigen binding).
- the target site for substitutional mutagenesis may include HVR and FR.
- Conservative substitutions in one embodiment are indicated as “preferred substitutions” in Table 1, more substantial changes are indicated as “exemplary substitutions” in Table 1, and further referring to the nature of the amino acid side chain In Amino acid substitutions may be introduced into the antibody of interest, and the product thereby screened for the desired activity, eg, retention / improved antigen binding, decreased immunogenicity, or improved ADCC or CDC. Also good.
- Amino acids can be divided into the following based on common side chain properties: (1) Hydrophobicity: norleucine, Met, Ala, Val, Leu, Ile; (2) Neutral hydrophilicity: Cys, Ser, Thr, Asn, Gln; (3) Acidity: Asp, Glu; (4) Basicity: His, Lys, Arg; (5) Residues that affect chain orientation: Gly, Pro; (6) Aromatic: Trp, Tyr, Phe Non-conservative substitutions mean exchanging one member of these classes for another class.
- an amino acid insertion may refer to the insertion of one or more amino acid residues into the amino acid sequence and is 1, 2, or 3-100 at the N-terminus and / or C-terminus, Alternatively, polypeptides containing more residues may be fused. Examples of terminal insertion include antibodies having a methionyl residue at the N-terminus. Examples of other insertion variants (variants) include an antibody (eg, an enzyme for ADEPT) or a polypeptide that increases the plasma half-life of the antibody fused to the N-terminus and / or C-terminus of the antibody. Things.
- percent (%) amino acid sequence identity refers to after aligning the sequences to introduce the maximum percent sequence identity and introducing gaps if necessary, and Defined as the percentage of amino acid residues in a candidate sequence that are identical to amino acid residues in a reference polypeptide sequence, given that any conservative substitution is not considered part of the sequence identity. Alignments for the purpose of determining% amino acid sequence identity are publicly available in various ways in the art, such as BLAST, BLAST-2, ALIGN, Megalign® (DNASTAR) ® software, or GENETYX® This can be achieved by using computer software. Those skilled in the art can determine appropriate parameters for aligning sequences, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared.
- the ALIGN-2 sequence comparison computer program is a work of Genentech, and its source code is submitted to the US Copyright Office along with user documents and registered under the US copyright registration number TXU510087.
- the ALIGN-2 program is publicly available from Genentech, Inc., South San Francisco, California, and may be compiled from source code.
- the ALIGN-2 program is compiled for use on UNIX® operating systems including Digital UNIX V4.0D. All sequence comparison parameters are set by the ALIGN-2 program and do not vary.
- % amino acid sequence identity of a given amino acid sequence A to, or with, or against a given amino acid sequence B (or a given amino acid sequence) A given amino acid sequence A, which has or contains some% amino acid sequence identity to, with or to B) is calculated as follows: fraction X / Y Hundredfold. Where X is the number of amino acid residues scored by the sequence alignment program ALIGN-2 as identical matches in the A and B alignments of the program, and Y is the total number of amino acid residues in B . When the length of amino acid sequence A is different from the length of amino acid sequence B, it is understood that the% amino acid sequence identity of A to B is different from the% amino acid sequence identity of B to A. Unless otherwise indicated, all% amino acid sequence identity values used herein are obtained using the ALIGN-2 computer program described above.
- the CDR region, heavy chain variable region, light chain variable region, heavy chain constant region, light chain constant region of an antibody in the present disclosure eg, an anti-IL-8 antibody, an anti-CXCR1 antibody, or an anti-CXCR2 antibody
- an antigen for example, IL-8, CXCR1, Alternatively, an amino acid sequence having binding activity to CXCR2
- a nucleic acid that hybridizes under stringent conditions to a nucleic acid comprising a base sequence encoding the amino acid sequence of such a region.
- Examples of stringent hybridization conditions for isolating nucleic acids that hybridize under stringent conditions include 6M urea, 0.4% SDS, 0.5 x SSC, 37 ° C, or equivalent or equivalent conditions. Is done. Isolation of nucleic acids with higher homology can be expected by using conditions with higher stringency, for example, 6M urea, 0.4% SDS, 0.1 ⁇ SSC, and 42 ° C.
- washing conditions after hybridization for example, 0.5xSSC (where 1xSSC is, for example, 0.15 M NaCL, 0.015 M sodium citrate, pH 7.0), 0.1% SDS, washing at 60 ° C, more preferably 0.2xSSC, 0.1% SDS, washing at 60 ° C, more preferably 0.2xSSC, 0.1% SDS, washing at 62 ° C, more preferably 0.2xSSC, 0.1% SDS, washing at 65 ° C, more preferably 0.1xSSC, 0.1% SDS, washing at 65 ° C.
- the washing time and number of times can be adjusted as appropriate. For example, 20 minutes may be carried out three times.
- the sequence of the isolated nucleic acid can be determined by a known method.
- the nucleic acid isolated in this manner is at least 50% or more, more preferably 70% or more, 75% or more, 80% or more, 85% or more of the entire nucleotide sequence relative to the nucleotide sequence of the target region. More preferably 90% or more (eg, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% or more) of sequence identity.
- An antibody having an amino acid sequence encoded by such an isolated nucleic acid is functionally (substantially) equivalent to an antibody having the amino acid sequence of the region of interest (eg, antigen binding or retention in blood). Any one or more indicators or characteristics such as gender are advantageously (substantially equivalent), but are not limited thereto.
- substantially is compared with the function of an antibody having the amino acid sequence of the target region (for example, attention may be paid to any one or more indicators or characteristics such as antigen-binding property or blood retention property).
- at least 50% or more, more preferably 70% or more, 75% or more, 80% or more, 85% or more, more preferably 90% or more for example, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% or more).
- an antibody in the present disclosure may be glycosylated. Addition or deletion of glycosylation sites to the antibody can be readily accomplished by altering the amino acid sequence to create or remove glycosylation sites.
- an antibody in the present disclosure for example, an anti-IL-8 antibody, an anti-CXCR1 antibody, or an anti-CXCR2 antibody
- the sugar chains that can be added to the region can vary.
- Naive antibodies produced by animal cells typically contain a branched biantennary oligosaccharide that is attached to the Asc 297 of the CH2 domain of the Fc region by N-linking (Wright et al. TIBTECH 15: 26-32 (1997)).
- Oligosaccharides include, for example, mannose, N-acetylglucosamine (GlcNAc), galactose, or sialic acid, in addition to fucose added to GlcNAc in the “trunk” of the branched oligosaccharide structure.
- modification of an antibody oligosaccharide in the present disclosure may be utilized to generate antibody variants with certain improved properties.
- effector function means a biological activity attributable to the Fc region of an antibody (eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody), and may vary depending on the antibody isotype.
- antibody effector functions include C1q binding or complement dependent cytotoxicity (CDC); Fc receptor binding; antibody dependent cell mediated cytotoxicity (ADCC); phagocytosis; cell surface receptors (eg, B cells Receptor) down-regulation; or B cell activation.
- the “Fc region” is used to define the C-terminal region of an immunoglobulin heavy chain containing at least a constant region part.
- the term includes both native Fc regions and variant Fc regions (Fc region variants).
- the natural Fc region refers to an Fc region of a natural antibody.
- Examples of the Fc region of natural human IgG include the above-described human IgG1 constant region (SEQ ID NO: 100), human IgG2 constant region (SEQ ID NO: 101), human IgG3 constant region (SEQ ID NO: 102), or human It may be an Fc region contained in an IgG4 constant region (SEQ ID NO: 103).
- the human IgG heavy chain Fc region extends from the amino acid residue at position Cys226 or at position Pro230 to the C-terminus of the heavy chain.
- lysine (Lys447) at the C-terminal of the Fc region, or glycine (Gly446) and lysine (Lys447) at the C-terminal may or may not be present.
- the numbering of amino acid residues in the Fc region or constant region herein is as follows: Kabat et al, Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, As described by Bethesda, MD (1991), according to the EU numbering system, also called the EU index.
- FcRn is different from Fc ⁇ R belonging to the immunoglobulin superfamily, and FcRn, particularly human FcRn, is structurally similar to major histocompatibility complex (MHC) class I polypeptides. It has 22-29% sequence identity with class I MHC molecules (Ghetie et al., Immunol. Today (1997) 18 (12), 592-598). FcRn is expressed as a heterodimer consisting of transmembrane ⁇ or heavy chain complexed with soluble ⁇ or light chain ( ⁇ 2 microglobulin).
- MHC major histocompatibility complex
- the ⁇ chain of FcRn consists of three extracellular domains ( ⁇ 1, ⁇ 2, ⁇ 3), and the short cytoplasmic domain tethers the protein to the cell surface.
- the ⁇ 1 and ⁇ 2 domains interact with the FcRn binding domain in the antibody Fc region (Raghavanghet al (Immunity (1994) 1, 303-315).
- FcRn is expressed in the maternal placenta or yolk sac of mammals, Involved in the transfer of IgG from the fetus to the fetus, and in the small intestine of rodent neonates that express FcRn, is involved in the movement of maternal IgG across the brush border epithelium from colostrum or milk ingested FcRn. It is expressed in many other tissues across many species, as well as in various endothelial cell lines, which are also expressed in human adult vascular endothelium, muscle vasculature, and liver sinusoidal capillaries.FcRn binds to IgG.
- the polynucleotide sequence and amino acid sequence of human FcRn may be derived from the precursors described in NM_004107.4 and NP_004098.1 (including the signal sequence), respectively (the RefSeq registration number is shown in parentheses).
- the precursor forms a complex with human ⁇ 2-microglobulin in vivo, so soluble human FcRn that can form a complex with human ⁇ 2-microglobulin is produced using known recombinant expression techniques.
- FcRn capable of forming a complex with ⁇ 2-microglobulin
- the binding activity of the antibody or Fc region variant to FcRn is evaluated.
- FcRn is not particularly limited as long as it is in a form capable of binding to the FcRn binding domain, and human FcRn is preferable.
- an antibody eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody
- FcRn binding domain Preferably having a human FcRn binding domain.
- FcRn binding domain is particularly limited if the antibody (eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody) has binding activity or affinity for FcRn at acidic and / or neutral pH Or a domain having binding activity to FcRn directly or indirectly.
- domains include, for example, Fc region of IgG type immunoglobulin having direct binding activity to FcRn, albumin, albumin domain3, anti-FcRn antibody, anti-FcRn peptide, anti-FcRn Scaffold molecule, etc., or indirectly FcRn Examples thereof include, but are not limited to, IgG having a binding activity to and molecules binding to albumin. Further, for example, a domain having FcRn binding activity at acidic pH and / or neutral pH may be used. The domain can be used as it is as long as it has FcRn binding activity at an acidic pH and / or neutral pH in advance.
- the amino acid residue in the FcRn binding domain in the antibody or Fc region variant is altered to change the acidic pH and / or medium FcRn binding activity at neutral pH may be obtained.
- amino acids in the domain having FcRn binding activity at acidic pH and / or neutral pH may be modified in advance to further increase FcRn binding activity.
- the desired modification can be found by comparing the FcRn-binding activity at acidic pH and / or neutral pH before and after the amino acid modification.
- the FcRn binding domain is preferably a region that directly binds to FcRn.
- the FcRn binding region include an antibody constant region or an Fc region.
- a region capable of binding to a polypeptide having binding activity to FcRn such as albumin or IgG can be indirectly bound to FcRn via albumin or IgG. Therefore, the FcRn binding region may be a region that binds to a polypeptide having binding activity to albumin or IgG.
- the FcRn-binding domain has a high FcRn-binding activity at neutral pH, and to improve the retention of antibodies in plasma.
- the FcRn binding domain preferably has a higher FcRn binding activity at acidic pH.
- an FcRn binding domain having a high FcRn binding activity at neutral pH or acidic pH may be selected and used in advance, or an amino acid in the antibody or Fc region may be modified to change FcRn at neutral pH or acidic pH. Binding activity may be imparted, or FcRn binding activity already present at neutral or acidic pH may be increased.
- the binding activity of an antibody (for example, an anti-IL-8 antibody, an anti-CXCR1 antibody, or an anti-CXCR2 antibody) or Fc region (variant) to FcRn is compared with the antibody or Fc region (variant) before being modified, Whether it is increased, (substantially) maintained, or decreased is not particularly limited as long as it is a method known to those skilled in the art, and the method described in this example may be used, for example, BIACORE, Scatchard Plots, flow cytometers, etc. can be used (WO2013 / 046722).
- the extracellular domain of human FcRn may be used as a soluble antigen.
- Conditions other than pH at the time of measuring the binding activity of the antibody or Fc region (modified) to FcRn can be appropriately selected by those skilled in the art and are not particularly limited.
- measurement can be performed under conditions of MES buffer and 37 ° C.
- the measurement of the binding activity between the antibody or Fc region (modified) and FcRn may be evaluated, for example, by flowing FcRn as an analyte to a chip on which the antibody is immobilized.
- the binding activity of the antibody or Fc region (modified) to FcRn is KD (Dissociation constant), apparent KD (Apparent dissociation constant), dissociation rate kd (Dissociation rate: dissociation) Velocity), or apparent kd (Apparent dissociation).
- the pH conditions for measuring the binding activity between the FcRn-binding domain and FcRn contained in the antibody (eg, anti-IL-8 antibody, anti-CXCR1 antibody, or anti-CXCR2 antibody) or Fc region (variant) can be acidic pH or neutral. pH conditions can be used as appropriate.
- the temperature used for the measurement conditions the binding activity (binding affinity) between the FcRn binding domain and FcRn can be evaluated at any temperature between 10 ° C. and 50 ° C.
- a temperature between 15 ° C. and 40 ° C. is used to determine the binding activity (binding affinity) between the human FcRn binding domain and FcRn. More preferably from 20 ° C.
- any of these temperatures is also used to determine, but is not limited to, the binding activity (binding affinity) between the FcRn binding domain and FcRn.
- a temperature of 25 ° C. is a non-limiting example.
- the amino acid sequence contained in the amino acid sequence of an antibody in the present disclosure is post-translationally modified (eg, by pyroglutamylation of N-terminal glutamine).
- the modification to pyroglutamic acid is a modification well known to those skilled in the art), but even if the amino acid is post-translationally modified in this way, it is equivalent to the amino acid sequence described in this disclosure Naturally included as a thing.
- an antibody in the present disclosure may be further derivatized by additionally including a non-proteinaceous moiety.
- Suitable moieties for antibody derivatization include, for example, water-soluble polymers.
- water-soluble polymers examples include polyethylene glycol (PEG), copolymers of ethylene glycol and propylene glycol, carboxymethyl cellulose, dextran, polyvinyl alcohol, polyvinyl pyrrolidone, poly 1,3 dioxolane, poly 1,3,6, trioxane, ethylene / Maleic anhydride copolymers, polyamino acids (either homopolymers or random copolymers), dextran or poly (n-vinylpyrrolidone) polyethylene glycol, polypropylene glycol homopolymers, polypropylene oxide / ethylene oxide copolymers, polyoxyethylated polyols (eg glycerol ), Polyvinyl alcohol, and any mixtures thereof.
- PEG polyethylene glycol
- copolymers of ethylene glycol and propylene glycol carboxymethyl cellulose
- dextran polyvinyl alcohol
- polyvinyl pyrrolidone poly 1,3 dioxolane
- polyethylene glycol propionaldehyde may be advantageous in production due to its water stability.
- the polymer may be of any molecular weight and may or may not be branched.
- the number of polymers added to the antibody can vary, and if more than one polymer is added, they can be the same molecule or different molecules. In general, the number and / or type of polymers used for derivatization will depend on, for example, the particular property or function of the antibody to be improved, whether the antibody derivative is used for therapy under defined conditions. , Etc. may be taken into consideration.
- a conjugate of an antibody in the present disclosure eg, an anti-IL-8 antibody, an anti-CXCR1 antibody, or an anti-CXCR2 antibody
- a non-protein portion that can be selectively heated by exposure to radiation
- the non-protein portion may be, for example, a carbon nanotube (Kam et al., Proc. Natl. Acad. Sci. USA 102: 11600-11605 (2005)).
- the radiation can be any wavelength as long as the desired purpose is achieved, e.g., such that the non-protein portion is heated to a temperature that does not harm normal cells but kills cells in close proximity to the antibody-non-protein portion. It's okay.
- IL-8 refers to any vertebrate, primate (eg, human, cynomolgus monkey, or rhesus monkey), or other mammal (eg, rabbit or dog, unless otherwise indicated, except for mouse or rat). Does not exist in IL-8).
- IL-8 includes full-length IL-8, any form of IL-8 modified in vivo, naturally occurring derivatives of IL-8, such as splice variants and allelic variants .
- a typical example of the amino acid sequence of human IL-8 is shown in SEQ ID NO: 22.
- the human interleukin-8 precursor is registered with NCBI as NP_000575.1.
- an example of a non-limiting cynomolgus monkey IL-8 amino acid sequence is registered with NCBI as XP_005555144.1.
- CXCR1 is a chemokine receptor also known as Interleukin-8 receptor, ⁇ alpha, IL8RA or CD181, and CXCR2 is also known as Interleukin-8 receptor, ⁇ beta, or IL8RB.
- IL-8 is known to produce physiological effects by binding to the receptors CXCR1 and CXCR2 (Science. 1991; 253: 1278-80, Science. 1991; 253: 1280-3.) .
- CXCR1 is activated only by IL-8 and granulocyte chemotactic protein-2
- CXCR2 is activated by binding to multiple molecules such as GRO ⁇ , ⁇ , ⁇ , neutrophil-activating peptide, and granulocyte chemotactic protein-2 It has been reported (Neuro-oncol. 2005; 7: 122-33), but no IL-8 receptor has been reported to date other than CXCR1 and CXCR2. As described in this example, the present inventors have confirmed that the migration ability to IL-8 in neutrophils is similarly inhibited by IL-8 neutralizing antibody and CXCR2 inhibitor.
- IL-8 signal inhibitors including CXCR1 inhibitors and CXCR2 inhibitors are useful not only for anti-IL-8 antibodies, but also for endometriosis, adhesions, and adenomyosis described in this Example It is easily predicted that.
- CXCR1 in this disclosure refers to natural CXCR1 present in any vertebrate, primate (eg, human, cynomolgus monkey, or rhesus monkey) or other mammal (eg, rabbit or dog) unless otherwise indicated.
- CXCR1 includes full-length CXCR1, any form of CXCR1 modified in vivo, naturally occurring derivatives of CXCR1, such as splice variants and allelic variants.
- An example of a non-limiting amino acid sequence of human CXCR1 is registered with NCBI as NP_000625.1.
- CXCR2 in this disclosure refers to natural CXCR2 present in any vertebrate, primate (eg, human, cynomolgus monkey, or rhesus monkey), or other mammal (eg, rabbit or dog) unless otherwise indicated.
- CXCR2 includes full-length CXCR2, any form of CXCR2 modified in vivo, and naturally occurring derivatives of CXCR2, such as splice variants and allelic variants.
- Non-limiting examples of human CXCR2 amino acid sequences are registered with NCBI as NP_001161770.1 and NP_001548.1.
- Immune cells such as monocytes, macrophages, neutrophils, lymphocytes, dermal fibroblasts, keratinocytes, vascular endothelial cells, melanocytes, hepatocytes, and various tumor cell lines produce IL-8.
- IL-8 is a powerful neutrophil chemokine that participates in the migration of neutrophils to the site of inflammation. When bound to its high-affinity receptors (CXCR1 and CXCR2) present on the surface of neutrophils, IL-8 promotes degranulation and increases the concentration of free Ca 2+ in the cytoplasm. It has been reported to activate spheres and induce neutrophil migration to destroy infiltrated tissues (WO2004 / 058797).
- CXCR1 and CXCR2 are expressed in immune cells such as neutrophils, monocytes, macrophages, endometriosis epithelial cells, and vascular endothelial cells.
- immune cells such as neutrophils, monocytes, macrophages, endometriosis epithelial cells, and vascular endothelial cells.
- stroma cells and myofibroblasts it was found not to be expressed in stroma cells and myofibroblasts.
- Iwabe's paper after removing epithelial cells from endometriotic lesions derived from human patients with endometriosis and making them stroma cells, IL-8 is added to examine cell proliferation, but it is weak at around 20% It was a reaction.
- an IL-8 signal inhibitor such as antibody H1009 / L395-F1974m in a non-human primate endometriosis model. It was demonstrated that atrophic changes of epithelial cells and stroma cells were observed.
- Iwabe's paper conducts experiments immediately after cell passage, so it is possible that immune cells are mixed.
- IL-8 signals via CXCR1 and CXCR2 do not act directly on stroma cells, but directly on endometrial epithelium and immune cells.
- the stroma cells were considered to act indirectly through these endometrial epithelium and immune cell signals.
- IL-8 signal inhibitor in the present disclosure is not particularly limited as long as it can directly or indirectly partially or completely inhibit IL-8 signal, and includes, for example, nucleic acids (for example, siRNA and the like). Heavy chain nucleic acid), protein (including antibody or antibody fragment), peptide, or other compound (eg, low molecular weight compound).
- the IL-8 signal inhibitor can be an IL-8 inhibitor, a CXCR1 inhibitor, or a CXCR2 inhibitor, in which case they are, for example, nucleic acids (eg, double-stranded nucleic acids such as siRNA), A protein (including an antibody or an antibody fragment, and more specifically, an anti-IL-8 antibody, an anti-CXCR1 antibody, an anti-CXCR2 antibody, or an antibody fragment that partially or completely retains the function thereof); It may be a peptide or other compound (for example, a low molecular weight compound).
- nucleic acids eg, double-stranded nucleic acids such as siRNA
- a protein including an antibody or an antibody fragment, and more specifically, an anti-IL-8 antibody, an anti-CXCR1 antibody, an anti-CXCR2 antibody, or an antibody fragment that partially or completely retains the function thereof
- It may be a peptide or other compound (for example, a low molecular weight compound).
- an IL-8 signal inhibitor partially or completely inhibits IL-8 binding to its receptor, CXCR1 and / or CXCR2
- inhibition of IL-8 causes IL-8 to be It is preferred that the normal level or type of activity that occurs when bound to the receptor is reduced or altered.
- the decrease or change is, for example, inhibition of IL-8 induced elastase release or calcium flux, inhibition of IL-8 induced increase in CD11b (Mac-1) expression, or inhibition of L-selectin expression decrease, etc. You can observe.
- Such inhibition is at least, for example, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90% when compared to the absence of an IL-8 signal inhibitor. It includes%, 95%, 99%, or 100% reduction or change in the above level or type of activity.
- a CXCR1 inhibitor in the present disclosure may be one that binds to CXCR1 (specific) but does not bind to CXCR2, or binds to both CXCR1 and CXCR2 (specific) ) (Also referred to as CXCR1 / CXCR2 inhibitor).
- a CXCR2 inhibitor in the present disclosure may be one that binds to CXCR2 (specific) but does not bind to CXCR1, or binds to both CXCR1 and CXCR2 (specific) ) (CXCR1 / CXCR2 inhibitor).
- CXCR1 inhibitor and / or CXCR2 inhibitor in the present disclosure include an anti-CXCR1 antibody disclosed in WO2010 / 056753, or repartaxin or a repartaxin derivative; disclosed in WO2011 / 042466 CXCR1 and / or CXCR2 inhibitors; CXCR1 or CXCR2 chemokine antagonists disclosed in WO2005 / 113534 and WO2003 / 057676; CXCR2 binding polypeptides disclosed in WO2012 / 062713 and WO2013 / 168108; WO2008 / 061740, WO2008 / 061741, Examples include CXCR2 inhibitors disclosed in WO2008 / 062026, WO2009 / 106539, WO2010 / 015613 and WO2013 / 030803.
- IL-8 inhibitors in the present disclosure include IL-8 mimetics described in JP2006-117633A; IL-8 expression inhibitors disclosed in JP2013-180992A (or JP5496232B); IL-8 production inhibitor disclosed in JPH9-2954A (or JP3008010B); antibody fragment-polymer complex disclosed in WO1998 / 037200; WO1995 / 023865, WO2009 / 026117, WO2013 / 166099, WO2006 / 113643, WO2004 / Anti-IL-8 antibodies disclosed in 058797, WO2008 / 130969, WO2011 / 100271, WO98 / 58671, WO2014 / 149733, US2003 / 0077283A1, etc., multispecific antibodies that bind to antigens containing IL-8, or modifications thereof Any body.
- these anti-IL-8 antibody variants may further be provided with one or more of the features described below as highly functional anti-
- an “anti-IL-8 antibody” or “an antibody that binds to IL-8” in the present disclosure is an antibody that can bind to IL-8 with sufficient affinity, so that the antibody Refers to antibodies that are useful as detection, diagnostic, therapeutic, and / or prophylactic compositions by targeting IL-8.
- the degree of anti-IL-8 antibody binding to a non-specific unrelated non-IL-8 protein may be, for example, less than 10% of the degree of antibody binding to IL-8.
- an anti-IL-8 antibody in the present disclosure has IL-8 neutralizing activity.
- IL-8 neutralizing activity may refer to the activity that inhibits the biological activity exhibited by IL-8, or it inhibits IL-8 from binding to its receptor (CXCR1 or CXCR2) You may point to the activity to do.
- the anti-IL-8 antibody in the present disclosure may be a chimeric antibody, a humanized antibody, or a human antibody.
- an anti-IL-8 antibody in the present disclosure is preferably cross-reactive with an animal (eg, a rabbit) that has IL-8 endogenously, and optionally, an anti-primate IL-8 antibody (preferably, it is an anti-non-human primate IL-8 antibody or an anti-human IL-8 antibody (for example, an anti-human IL-8 neutralizing antibody) having menstruation similar to that of humans, for example, cynomolgus monkeys or baboons .
- an amino acid modification technique that can add a high function to an antibody (for example, an anti-IL-8 antibody, an anti-CXCR1 antibody, or an anti-CXCR2 antibody), as embodied in this example, Based on that, I came up with a highly functional anti-IL-8 antibody, a non-limiting example of which is given below:
- the anti-IL-8 antibody in the present disclosure may be an anti-IL-8 antibody that may have a pH-dependent affinity for IL-8, such as the anti-IL-8 antibody.
- an amino acid may be substituted with any other amino acid unless otherwise specified.
- such an anti-IL-8 antibody of the present disclosure may be substituted with another amino acid at a position selected from the group consisting of the following positions, or with the amino acid specified below at that position: May each further include: (1) Serine at position 8 of the sequence of SEQ ID NO: 26, Asparagine at position 1 of the sequence of SEQ ID NO: 27 and leucine at position 5, and glutamine at position 1 of the sequence of SEQ ID NO: 28; (2) Substitution of alanine at position 6 in the sequence of SEQ ID NO: 24 to aspartic acid, substitution of arginine at position 11 to proline, and substitution of tyrosine at position 3 of tyrosine in the sequence of SEQ ID NO: 25 Replacement; (3) Replacement of glycine at position 8 with tyrosine and replacement of tyrosine at position 9 with histidine in the sequence of SEQ ID NO: 24; (4) Substitution of alanine at position 6 in the sequence
- the anti-IL-8 antibody that may have a pH-dependent affinity for IL-8 in the present disclosure is one or any plurality selected from the group consisting of: Contains the amino acid sequence: (A) HVR-H1 comprising the amino acid sequence of SEQ ID NO: 23, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, more preferably 95% or more HVR-H1 having sequence identity; (B) HVR-H2 comprising the amino acid sequence of SEQ ID NO: 24 or 29, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, more preferably 95% HVR-H2 having the above sequence identity; (C) HVR-H3 comprising the amino acid sequence of SEQ ID NO: 25 or 30, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, more preferably 95% HVR-H3 having the above sequence identity; (D) HVR-L1 comprising the amino acid sequence: (A
- an HVR having a specific% sequence identity with a reference amino acid sequence may be functionally equivalent to an HVR having a reference amino acid sequence.
- the anti-IL-8 antibody may include HVR-H2 including the amino acid sequence of SEQ ID NO: 29 and HVR-H3 including the amino acid sequence of SEQ ID NO: 30, and / or SEQ ID NO: HVR-L2 comprising 31 amino acid sequences and HVR-L3 comprising amino acid sequence SEQ ID NO: 32 may be included.
- the anti-IL-8 antibody further comprises at least a substitution with another amino acid at a position selected from the group consisting of the following positions, or a substitution with the amino acid specified below at the position.
- “functionally equivalent” in the present disclosure means, unless otherwise specified, a function (for example, a physicochemical property and / or biological activity) possessed by an antibody comprising a reference amino acid sequence.
- a function for example, a physicochemical property and / or biological activity
- at least 50% or more, more preferably 70 % Or more, 75% or more, 80% or more, 85% or more, more preferably 90% or more eg, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99 % Or more is intended.
- the anti-IL-8 antibody that may have a pH-dependent affinity for IL-8 in the present disclosure is one or any plurality selected from the group consisting of: Contains the amino acid sequence: (A) a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 33, 34, or 108, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, More preferably a heavy chain variable region having a sequence identity of 95% or more; and (B) a light chain variable region comprising the amino acid sequence of SEQ ID NO: 35 or 109, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, more preferably 95 A light chain variable region having at least% sequence identity.
- variable region having a certain percent sequence identity with a reference amino acid sequence may be functionally equivalent to a variable region having a reference amino acid sequence.
- the anti-IL-8 antibody may include a heavy chain variable region comprising the amino acid sequence of SEQ ID NO: 34 and a light chain variable region comprising the amino acid sequence of SEQ ID NO: 35. This may be advantageous because the IL-8 neutralizing activity is kept stable in plasma (for example in plasma), or it may be advantageous because of low immunogenicity.
- the antibody when the anti-IL-8 antibody contains a variant of the amino acid sequence of SEQ ID NO: 33, the antibody is directed to other amino acids at one or more positions selected from the group consisting of the following positions: It may further comprise at least a substitution or substitution at that position with the amino acid specified below: (I) substitution of alanine at position 55 of the amino acid sequence of SEQ ID NO: 33 with another amino acid, for example, substitution with aspartic acid; (Ii) substitution of glycine at position 57 of the amino acid sequence of SEQ ID NO: 33 with another amino acid, for example, substitution with tyrosine; (Iii) substitution of tyrosine at position 58 of the amino acid sequence of SEQ ID NO: 33 with another amino acid, for example, substitution with histidine; (Iv) substitution of arginine at position 60 in the amino acid sequence of SEQ ID NO: 33 with another amino acid, for example, substitution with proline; (V) substitution of glutamine at position 84 in the amino acid sequence of SEQ ID NO:
- the anti-IL-8 antibody in the present disclosure comprises an antibody comprising at least one to six amino acid sequences of (a) to (f) described in the following (1) to (7), respectively: Or an antibody comprising a substitution of at least one amino acid in at least one amino acid sequence of any one of (a) to (f) described above.
- the antibody may be an anti-IL-8 antibody having a pH dependent affinity for IL-8.
- An amino acid may be substituted with any other amino acid unless otherwise specified.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 58
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 59
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 60
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 61
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 62
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 63.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 64
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 65
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 66
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 67
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 68
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 69.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 70
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 71
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 72
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 73
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 74
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 75.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 76
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 77
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 78
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 79
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 80
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 81.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 82
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 83
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 84
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 85
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 86
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 87.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 88
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 89
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 90
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 91
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 92
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 93.
- HVR-H1 comprising the amino acid sequence of SEQ ID NO: 94
- B HVR-H2 comprising the amino acid sequence of SEQ ID NO: 95
- C HVR-H3 comprising the amino acid sequence of SEQ ID NO: 96
- D HVR-L1 comprising the amino acid sequence of SEQ ID NO: 97
- E HVR-L2 comprising the amino acid sequence of SEQ ID NO: 98
- f HVR-L3 comprising the amino acid sequence of SEQ ID NO: 99.
- “acidic pH” refers to a pH that can be selected from, for example, pH 4.0 to pH 6.5.
- neutral pH refers to a pH that can be selected from, for example, pH 6.7 to pH 10.0.
- the anti-IL-8 antibody in the present disclosure is an anti-IL-8 antibody that binds IL-8 in a pH-dependent manner.
- an anti-IL-8 antibody that binds to IL-8 in a pH-dependent manner refers to IL- at acidic pH compared to its binding affinity to IL-8 at neutral pH. Refers to an antibody with reduced binding affinity to 8.
- pH-dependent anti-IL-8 antibodies include antibodies that have a higher affinity for IL-8 at neutral pH than at acidic pH.
- the anti-IL-8 antibody in the present disclosure has at least a 2-fold, 3-fold, 5-fold, 10-fold affinity for IL-8 at neutral pH rather than affinity at acidic pH, 15 times, 20 times, 25 times, 30 times, 35 times, 40 times, 45 times, 50 times, 55 times, 60 times, 65 times, 70 times, 75 times, 80 times, 85 times, 90 times, 95 times , 100 times, 200 times, 400 times, 1000 times, 10000 times or higher.
- a surface plasmon resonance method such as BIACORE
- the binding rate constant (kon) and dissociation rate constant (koff) are calculated using the simple one-to-one Langmuir binding model Biacore T200 Evaluation Software (GE (Healthcare).
- the equilibrium dissociation constant (KD) is calculated as the koff / kon ratio. Screening for antibodies with different binding affinities depending on pH is not particularly limited. In addition to surface plasmon resonance (BIACORE, etc.), ELISA and binding equilibrium exclusion methods (Kinetic Exclusion Assay; KinExA TM ), etc. Can also be used.
- the pH-dependent IL-8 binding ability means the property of binding to IL-8 in a pH-dependent manner. Whether or not an antibody can bind to IL-8 multiple times can be determined by the method described in WO2009 / 125825.
- the anti-IL-8 antibody in the present disclosure preferably has a small dissociation constant (KD) for IL-8 at a neutral pH (eg, pH 7.4).
- KD small dissociation constant
- the antibody may have a dissociation constant for IL-8 at a neutral pH (eg, pH 7.4) of 0.3 nM or less, 0.1 nM or less, or 0.03 nM or less.
- the anti-IL-8 antibody in the present disclosure preferably has a large dissociation constant (KD) for IL-8 at an acidic pH (eg, pH 5.8).
- the antibody may have a dissociation constant for IL-8 at an acidic pH (eg, pH 5.8) of 3 nM or more, 10 nM or more, or 30 nM or more.
- the anti-IL-8 antibody in the present disclosure has a ratio of dissociation constant at acidic pH (eg, pH 5.8) to dissociation constant at neutral pH (eg, pH 7.4) [KD (acid pH) / KD ( Neutral pH)] (for example, [KD (pH 5.8) / KD (pH 7.4)]) is, for example, 30 or more, 50 or more, or for example 100 or more, for example, 200, 300, 400, 500, 600, 700, 800, 900, 1000, 1500, 2000, 2500, 3000, 3500, 4000, 4500, 5000, 5500, 6000, 6500, 7000, 7500, 8000, 8500, 9000, or 9500 or more.
- the anti-IL-8 antibody in the present disclosure preferably has a large dissociation rate constant (koff) at an acidic pH (eg, pH 5.8).
- the dissociation rate constant at an acidic pH (eg, pH 5.8) of the antibody may be, for example, 0.003 (1 / s) or more, 0.005 (1 / s) or more, or 0.01 (1 / s) or more.
- WO2013 / 046704 was also introduced with a predetermined mutation (typically a two-residue modification of Q438R / S440E represented by EU numbering). It has been reported that Fc region variants showed a significant reduction in rheumatoid factor binding. However, in WO2013 / 046704, it has been shown that a modified Fc region with reduced rheumatoid factor binding by modification of Q438R / S440E is superior in plasma retention compared to an antibody having a natural Fc region. I didn't.
- an anti-IL-8 antibody in the present disclosure comprises an Fc region variant comprising an FcRn binding domain, said FcRn binding domain comprising, for example, an EU compared to a native Fc region. It may be a modified Fc region containing Ala at position 434; Glu, Arg, Ser, or Lys at position 438; and Glu, Asp, or Gln at position 440, represented by numbering.
- the anti-IL-8 antibody can be an FcRn binding domain with increased FcRn binding activity at acidic and neutral pH, particularly at acidic pH.
- the antibody comprises an Fc region whose FcRn binding domain is represented by EU numbering, including Ala at position 434; Glu, Arg, Ser or Lys at position 438; and Glu, Asp or Gln at position 440 It is preferred to include a variant, more preferably Ala at position 434; Arg or Lys at position 438; and Glu or Asp at position 440.
- the Fc region variant in the anti-IL-8 antibody in the present disclosure is preferably a Fc region variant of a natural Ig antibody, and a natural IgG (IgG1, IgG2, IgG3, or IgG4 Type) antibody Fc region variant is more preferable, and it is more preferably derived from natural human IgG1, IgG2, IgG3, or IgG4. For example, it may be derived from human IgG1.
- Fc regions of natural IgG1, IgG2, IgG3, and IgG4 type antibodies the modified sites described above, represented by EU numbering, at positions 428, 434, 436, 438, and 440, except for position 436, all of the Fc regions of natural human IgG1, IgG2, IgG3, and IgG4 type antibodies are common.
- position 436 all of the Fc regions of natural human IgG1, IgG2, and IgG4 type antibodies are Tyr (Y), but the natural human IgG3 type antibody has Fhe region of Phe (F). is there.
- WO2013 / 046704 describes a 2-residue amino acid that could result in a significant reduction in rheumatoid factor binding when combined with an amino acid substitution capable of increasing binding to FcRn under acidic pH.
- substitutions specifically, Q438R / S440E, Q438R / S440D, Q438K / S440E, and Q438K / S440D, which are represented by EU numbering, have been reported.
- the amino acid sequence of the Fc region variant in the anti-IL-8 antibody in the present disclosure is represented by the EU numbering in the FcRn-binding domain as compared to the native Fc region.
- amino acid substitutions selected from the group consisting of L235R, G236R, S239K, A327G, A330S, P331S, M428L, N434A, Y436T, Q438R, and S440E (for example, six, Seven or all).
- the Fc region variant in the anti-IL-8 antibody of the present disclosure preferably has an increased FcRn binding activity under acidic pH as compared to the native IgG Fc region.
- Increased binding affinity of the FcRn binding domain for FcRn at a certain pH may correspond to an increase in measured FcRn binding affinity compared to the measured FcRn binding affinity for the native FcRn binding domain.
- KD (natural Fc region) / KD (an Fc region variant in the anti-IL-8 antibody in the present disclosure) that is a difference in binding affinity is at least 1.5 times, 2 times, 3 times, 4 times, It may be 5 times, 10 times, 15 times, 20 times, 50 times, 70 times, 80 times, 100 times, 500 times, or 1000 times.
- the increase in the binding affinity of the FcRn binding domain for FcRn may be at acidic pH and / or neutral pH, but it is particularly preferred that the binding activity for FcRn is increased under acidic pH.
- the Fc region variant in the anti-IL-8 antibody in the present disclosure in which the binding affinity to FcRn is increased under acidic pH has a binding affinity for FcRn higher than that of the native IgG Fc region, for example, pH.
- pH a binding affinity for FcRn higher than that of the native IgG Fc region, for example, pH.
- 6.0 and 25 ° C. for example, 1.5 times, 2 times, 3 times, 4 times, 5 times, 6 times, 7 times, 8 times, 9 times, 10 times, 20 times, 30 times, 50 times, 75 times, 100 times, 200 times, 500 times, 1000 times or more strong.
- anti-pharmaceutical antibody or “ADA” means an endogenous antibody that has binding affinity (binding activity) for an epitope located on a therapeutic antibody, and thus can bind to the therapeutic antibody.
- existing anti-pharmaceutical antibody or “existing ADA” means an anti-pharmaceutical antibody that can be present and detected in the blood of a patient before the therapeutic antibody is administered to the patient.
- the existing ADA is a rheumatoid factor, a polyclonal or monoclonal autoantibody directed against the Fc region of a human IgG antibody.
- the epitope for rheumatoid factor is located in the CH2 / CH3 interface region and CH3 domain, but may vary from clone to clone.
- the Fc region variant with a low binding affinity for an existing ADA is, for example, 1/10 or less, 1/50 or less, compared to the binding affinity of an antibody containing a natural IgG Fc region to ADA. Alternatively, it may be reduced to 1/100 or less.
- the binding affinity of the Fc region (variant) of an antibody for an existing ADA can be demonstrated by an electrochemiluminescence (ECL) reaction at acidic and / or neutral pH; however, known to those skilled in the art
- ECL electrochemiluminescence
- the ECL assay is described, for example, in Moxness et al (Clin Chem, 2005, 51: 1983-85) and in this example.
- the measurement can be performed, for example, under conditions of MES buffer and 37 ° C.
- Biacore may be used for the antigen-binding activity of the antibody.
- the binding affinity for an existing ADA can be assessed at any temperature between 10 ° C and 50 ° C.
- a temperature between 15 ° C. and 40 ° C. is used to determine the binding activity between the human Fc region and the existing human ADA.
- the temperature is preferably 20 ° C. to 25 ° C., more preferably 25 ° C. It may be measured at pH 7.4 (or pH 7.0) and 25 ° C.
- the Fc region variant in the anti-IL-8 antibody in the present disclosure binds to an existing ADA, preferably rheumatoid factor (RF), under neutral pH compared to the Fc region of native IgG.
- RF rheumatoid factor
- Affinity is not significantly increased and / or increased binding activity to FcRn under acidic pH results in decreased plasma clearance (CL) or increased plasma residence time.
- the plasma half-life (t1 / 2) is preferably increased. It is known to those skilled in the art that the antibody clearance in plasma (CL), plasma residence time, plasma half-life (t1 / 2) are correlated with each other.
- an Fc region variant in an anti-IL-8 antibody in the present disclosure comprises a reference Fc region variant comprising a combination of N434Y / Y436V / Q438R / S440E substituted amino acids represented by EU numbering
- the retention in plasma is improved.
- an Fc region variant called F1718 described in WO2013 / 046704 mutations at four sites N434Y / Y436V / Q438R / S440E are introduced into the Fc region
- an Fc region variant The plasma retention of two Fc region variants with F1848m (in which four mutations N434A / Y436V / Q438R / S440E have been introduced) is compared.
- the antibody includes Fc region variants containing one or more amino acid substitutions that increase binding of the Fc region to FcRn, and include 238, 256, 265, 272, 286, 303, 305, 307, 311, 312 , 317, 340, 356, 360, 362, 376, 378, 380, 382, 413, 424, and 434 (EU numbering), eg, position 434 of the Fc region Amino acid substitutions may be included.
- Fc region variants see Duncan & Winter, Nature 322: 738-40 (1988); US Patent No. 5,648,260; US Patent No. 5,624,821; WO 94/29351. Modifications of these amino acid residues may be appropriately incorporated into the Fc region (variant) of the anti-IL-8 antibody in the present disclosure.
- Fc region variants that do not bind to effector receptors may be safer and / or more effective.
- an anti-IL-8 antibody comprising an Fc region variant in the present disclosure has low binding activity for complement protein or does not bind to complement protein.
- the complement protein is C1q.
- the low binding activity to complement protein is, for example, 1/10 or less, 1/50 or less, compared to the binding activity to the complement protein of a natural IgG or an antibody containing a natural IgG Fc region, Alternatively, it may refer to the binding activity to complement proteins, which is reduced to 1/100 or less.
- the binding activity of the Fc region (modified) to the complement protein can be reduced by modifying the amino acid sequence such as amino acid substitution, insertion, or deletion.
- an anti-IL-8 antibody comprising an Fc region variant in the present disclosure preferably has low or no binding affinity for an effector receptor.
- effector receptors include activated Fc ⁇ R, in particular Fc ⁇ RI, Fc ⁇ RII, Fc ⁇ RIII.
- Fc ⁇ RI includes, for example, Fc ⁇ RIa, Fc ⁇ RIb, Fc ⁇ RIc, and subtypes thereof.
- Fc ⁇ RII includes, for example, Fc ⁇ RIIa (two allotypes R131 and H131) and Fc ⁇ RIIb.
- Fc ⁇ RIII includes, for example, Fc ⁇ RIIIa (two allotypes V158 and F158) and Fc ⁇ RIIIb (two allotypes Fc ⁇ IIIb-NA1 and Fc ⁇ IIIb-NA2).
- An antibody having low or no binding affinity for an effector receptor is, for example, 1/10 or less, 1/50 or less compared to the binding affinity of an antibody containing a natural IgG Fc region, or It may refer to an antibody comprising an Fc region variant that is reduced to 1/100 or less.
- an antibody containing a silent Fc region or an antibody not containing an Fc region eg, Fab, F (ab) ′ 2 , scFv, sc (Fv) 2 , diabody.
- Fc region variants with low or no binding affinity for effector receptors are described in Strohl et al. (Current Opinion in Biotechnology (2009) 20 (6), 685-691), e.g. Examples of silent Fc regions (IgG1-L234A / L235A, IgG1-H268Q /) that are glycosylated Fc regions (N297A, N297Q) or Fc regions that have been engineered to silence (or immunosuppress) effector functions A330S / P331S, IgG1-C226S / C229S, IgG1-C226S / C229S / E233P / L234V / L235A, IgG1-L234F / L235E / P331S, IgG2-V234A / G237A, IgG2-H268Q / V309L / A330S / A331S, IgG4
- WO2008 / 092117 discloses an antibody comprising a silent Fc region containing a substitution G236R / L328R, L235G / G236R, N325A / L328R, or N325L / L328R (by EU numbering).
- WO2000 / 042072 discloses an antibody comprising a silent Fc region containing a substitution at one or more of positions EU233, EU234, EU235 and EU237.
- WO2009 / 011941 discloses an antibody comprising a silent Fc region containing deletion of residues from EU231 to EU238.
- 332 7,332,581 describes the Fc region variant containing two or more amino acid substitutions of EU265, EU269, EU270, EU297, EU327 in addition to the so-called ⁇ DANA '' Fc region variant in which EU265 and EU297 are replaced by alanine Has been. Modification of these amino acid residues may be appropriately incorporated into the Fc region variant of the anti-IL-8 antibody in the present disclosure.
- “Low binding to an effector receptor” means, for example, 95% or less, preferably 90, 85, 80, or 75 of the binding activity of a natural IgG (or an antibody containing a natural IgG Fc region) to an effector receptor. %, More preferably 70, 65, 60, 55, 50, 45, 40, 35, 30, 25, 20, 15, 10, 9, 8, 7, 6, 5, 4, 3, 2, or 1 It means a binding activity that is less than or equal to%.
- the binding activity to Fc ⁇ R is, for example, 1/10 or less, 1/50 or less, or 100 compared to the binding activity of natural IgG (or an antibody containing a natural IgG-Fc region) to an effector receptor. It may be reduced to a fraction of a minute.
- a silent Fc region is an Fc region variant containing one or more amino acid substitutions, insertions, deletions, etc. that reduce binding to an effector receptor as compared to a native Fc region. .
- the Fc region variant no longer (substantially) binds to the effector receptor, since the binding activity to the effector receptor can be greatly reduced.
- silent Fc regions include EU234, EU235, EU236, EU237, EU238, EU239, EU265, EU266, EU267, EU269, EU270, EU271, EU295, EU296, EU297, EU298, EU300, EU324, EU325, EU327, EU328, Fc regions (variants) containing amino acid substitutions at one or more positions selected from the group consisting of EU329, EU331, and EU332 are included. In one embodiment, modification of these amino acid residues may be appropriately incorporated into the Fc region (variant) of the anti-IL-8 antibody in the present disclosure.
- the antibody constant region modification method for producing the Fc region variant of the anti-IL-8 antibody in the present disclosure includes, for example, examining a plurality of constant region isotypes (IgG1, IgG2, IgG3, IgG4). Then, by appropriately introducing, for example, an amino acid substitution into the sequence, an isotype that reduces the antigen binding activity under acidic pH and / or increases the dissociation rate under acidic pH may be selected.
- IgG1, IgG2, IgG3, IgG4 constant region isotypes
- the anti-IL-8 antibody in the present disclosure comprises (A) a heavy chain comprising the amino acid sequence of SEQ ID NO: 36, 37 or 106, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, more preferably 95 % Heavy chain with sequence identity of greater than or equal to; and / or (B) a light chain comprising the amino acid sequence of SEQ ID NO: 38 or 44, or at least 65%, 70%, or 75% or more, preferably 80%, 85% or 90% or more, more preferably 95% or more
- a heavy or light chain having a certain percentage of sequence identity with a reference amino acid sequence may be functionally equivalent to a heavy or light chain having a reference amino acid sequence.
- the anti-IL-8 antibody may comprise an Fc region variant having at least one of the following five properties: (a) The binding affinity of the Fc region variant to FcRn at acidic pH is higher than the binding affinity of the native Fc region to FcRn. (b) The binding affinity of the Fc region variant to the existing ADA is lower than the binding affinity of the natural Fc region to the existing ADA.
- the plasma half-life of the Fc region variant is greater than the plasma half-life of the native Fc region, (d) the plasma clearance of the Fc region variant is reduced compared to the plasma clearance of the native Fc region, (e) the binding affinity of the Fc region variant to the effector receptor is lower than the binding affinity of the native Fc region to the effector receptor; (f) It can bind to IL-8 in a pH-dependent manner.
- anti-IL-8 antibodies in the present disclosure can be made using, for example, the methods described in US Patent No. 4,816,567.
- an isolated nucleic acid encoding an anti-IL-8 antibody in the present disclosure is provided. Such a nucleic acid may encode an amino acid sequence comprising VL of the antibody and / or an amino acid sequence comprising VH (eg, the light chain and / or heavy chain of the antibody).
- the nucleic acid encoding the isolated anti-IL-8 antibody may be inserted into one or more vectors for further cloning and / or expression in the host cell.
- nucleic acids can be readily isolated and sequenced in a conventional manner (eg, by using an oligonucleic acid probe that specifically binds to the gene encoding the heavy and / or light chain of the antibody).
- one or more vectors comprising such nucleic acids are provided.
- a host cell comprising such a nucleic acid is provided. The host cell includes (1) a vector containing a nucleic acid encoding an amino acid sequence containing VL of an antibody and / or an amino acid sequence containing VH, or (2) a nucleic acid encoding an amino acid sequence containing the VL of an antibody.
- a second vector comprising a nucleic acid encoding an amino acid sequence comprising one vector and an antibody VH is included (eg, the cells have been transformed with these vectors).
- the host is eukaryotic (eg, CHO cells or lymphocytes (eg, Y0, NS0, SP20 cells)).
- a method for producing an anti-IL-8 antibody in the present disclosure comprises culturing a host cell containing the nucleic acid under conditions suitable for antibody expression, and optionally a host cell ( Or recovering the antibody from the culture medium of the host cells).
- Suitable host cells for expression and cloning of the antibody-encoding vector include, for example, prokaryotic cells or eukaryotic cells.
- Cells from multicellular organisms may be used for the expression of glycosylated antibodies. Examples of invertebrates are plant cells and insect cells.
- the antibody produced by culturing a host cell containing a nucleic acid encoding an anti-IL-8 antibody under conditions suitable for antibody expression can be produced in the host cell or extracellularly (medium, milk, etc.). And purified as a substantially pure and homogeneous antibody.
- separation and purification methods commonly used for purification of polypeptides can be preferably used, but are not limited thereto. For example, chromatography column, filter, ultrafiltration, salting out, solvent precipitation, solvent extraction, distillation, immunoprecipitation, SDS-polyacrylamide gel electrophoresis, isoelectric focusing, dialysis, recrystallization, etc. are appropriately selected, In combination, the antibodies can be suitably separated and purified.
- chromatography examples include affinity chromatography, ion exchange chromatography, hydrophobic chromatography, gel filtration chromatography, reverse phase chromatography, adsorption chromatography and the like. These chromatography can be performed using liquid phase chromatography, for example, liquid phase chromatography such as HPLC and FPLC.
- liquid phase chromatography such as HPLC and FPLC.
- columns used for affinity chromatography include a protein A column and a protein G column. For example, as a column using protein A, Hyper D, POROS, Sepharose F. F. (manufactured by Pharmacia) can be mentioned.
- the present disclosure addresses the properties of anti-IL-8 antibodies such as increased binding to extracellular matrix and increased cellular uptake of IL-8 and anti-IL-8 antibody complexes.
- the present invention relates to a method for selecting an antibody having increased binding to an extracellular matrix or an antibody having increased intracellular uptake.
- the present disclosure relates to a method for producing an antibody comprising a variable region whose IL-8 binding activity is pH-dependent, comprising the following steps: i) a step of evaluating the binding between the anti-IL-8 antibody and the extracellular matrix, (ii) a step of selecting an anti-IL-8 antibody having a high binding to the extracellular matrix, and (iii) a nucleic acid encoding the antibody.
- Evaluation of binding to the extracellular matrix is performed by, for example, ELISA that detects binding between the antibody and the extracellular matrix by adding an antibody to a plate on which the extracellular matrix is immobilized and adding a labeled antibody to the antibody. It can be measured by the system.
- an electrochemiluminescence method in which a mixture of an antibody and a ruthenium antibody is added to a plate on which an extracellular matrix is immobilized, and the binding between the antibody and the extracellular matrix is measured by electrochemiluminescence of ruthenium. ) Can be measured.
- the anti-IL-8 antibody in the present disclosure desirably maintains IL-8 neutralization activity stably in a solution (for example, in PBS). Whether the activity remains stable in solution depends on whether the IL-8 neutralizing activity of the disclosed antibodies added to the solution changes before and after storage at a temperature for a period of time. It can be confirmed by measuring.
- the storage period is, for example, 1-4 weeks, and the storage temperature is, for example, 25 degrees, 30 degrees, 35 degrees, 40 degrees, or 50 degrees.
- the anti-IL-8 antibody in the present disclosure stably maintains IL-8 neutralizing activity in vivo (for example, in plasma). Whether the activity is stably maintained in vivo is determined by determining whether the IL-8 neutralizing activity of the antibody of the present disclosure added to plasma of a non-human animal (eg, mouse) or human was stored at a certain temperature for a certain period of time. It can be confirmed by measuring whether it changes before and after.
- the storage period is, for example, 1 to 4 weeks, and the storage temperature is, for example, 25 degrees, 30 degrees, 35 degrees, or 40 degrees.
- the rate at which the anti-IL-8 antibody is taken into cells in the present disclosure is greater when complexed with IL-8 than when the antibody alone.
- the anti-IL-8 antibody in the present disclosure preferably has reduced immunogenicity expected in a human host.
- “Low immunogenicity” refers to, for example, that an administered anti-IL-8 antibody is immunized by a living body in at least a majority of individuals to which a sufficient amount of antibody is administered for a time sufficient to achieve a therapeutic effect. It may mean that no response is elicited. Eliciting an immune response can include the production of anti-pharmaceutical antibodies. “Low production of anti-pharmaceutical antibodies” can be rephrased as “low immunogenicity”.
- the level of immunogenicity in humans can also be predicted by a T cell epitope prediction program, which includes Epibase (Lonza), iTope / TCED (Antitope), EpiMatrix (EpiVax), etc. It is.
- a T cell epitope prediction program which includes Epibase (Lonza), iTope / TCED (Antitope), EpiMatrix (EpiVax), etc. It is.
- analysis can be performed using a T cell epitope prediction program, and a sequence with reduced immunogenicity can be designed.
- Non-limiting examples of such amino acid modification sites include positions 81 and / or 82b represented by Kabat numbering in the heavy chain sequence of the anti-IL-8 antibody represented by SEQ ID NO: 34.
- the reference antibody in the functional comparison with the anti-IL-8 antibody described in the present disclosure may be an antibody comprising the amino acid sequences of SEQ ID NOs: 39 and 40.
- the reference antibody for use in the PK test may be an antibody comprising the amino acid sequences of SEQ ID NOs: 43 and 45.
- an IL-8 signal inhibitor eg, an anti-IL-8 antibody in the present disclosure is identified and screened by various known methods using physicochemical properties and / or biological activities as indicators. Or characterized.
- antibodies in the present disclosure may be surface plasmons using devices such as ELISA, Western blot, Kinetic Exclusion Assay (KinExA TM ), or BIACORE (GE Healthcare). Antigen binding activity may be examined by methods such as resonance.
- binding affinity can be measured using Biacore® T200 (GE® Healthcare) as follows.
- An appropriate amount of a supplemental protein for example, protein A / G (PIERCE)
- CM4 GE Healthcare
- the target antibody is captured there.
- the diluted antigen solution and running buffer as reference solution: eg 0.05% tween20, 20 mM ACES, 150 mM NaCl, pH 7.4
- the antigen molecules are applied to the antibodies captured on the sensor chip.
- a 10 mM glycine-HCl solution (pH 1.5) is used, and the measurement is performed at a specified temperature (for example, 37 ° C, 25 ° C, or 20 ° C).
- a specified temperature for example, 37 ° C, 25 ° C, or 20 ° C.
- the KD (M) for each antibody antigen is calculated.
- IL-8 quantification can be performed as follows. A plate on which an anti-human IL-8 antibody having a mouse IgG constant region is immobilized is prepared. A solution containing IL-8 bound to a humanized IL-8 antibody that does not compete with the anti-human IL-8 antibody is dispensed to the solid phased plate and stirred. Thereafter, biotinylated anti-human Igk light chain antibody is added and allowed to react for a certain period of time, and further, SULFO-Tag labeled streptavidin is added and allowed to react for a certain period of time. Immediately after adding the measurement buffer, measure with SECTOR Imager 2400 (Meso Scale Discovery).
- a measurement is provided to identify an antibody having biological activity.
- the biological activity includes, for example, an activity of neutralizing an antigen (eg, IL-8) or an activity of blocking an antigen (eg, IL-8) signal.
- the method for determining the degree of IL-8 neutralization activity can be measured, for example, by the following method.
- PathHunter TM CHO-K1KCXCR2 ⁇ -Arrestin Cell Line expresses human CXCR2, known as one of the human IL-8 receptors, and human IL-8 It is a cell line created artificially so as to exhibit chemiluminescence when a signal by is transmitted.
- human IL-8 is added to the culture medium of the cells, the cells exhibit chemiluminescence depending on the concentration of the added human IL-8.
- the anti-human IL-8 antibody can block signal transduction by IL-8, so the chemiluminescence of the cell As compared with the case where no is added, it is weakened or not detected at all. That is, the stronger the human IL-8 neutralizing activity of the antibody, the weaker the chemiluminescence, and the weaker the human IL-8 neutralizing activity of the antibody, the stronger the chemiluminescence. By confirming this difference, it is possible to evaluate the neutralizing activity of anti-human IL-8 antibody against human IL-8.
- compositions are used interchangeably with “diagnostic agent”, “therapeutic agent”, and “prophylactic agent”, respectively.
- these compositions are also referred to as pharmaceutical compositions, Usually refers to a drug for the treatment or prevention of a disease or condition.
- pharmaceutically acceptable carrier refers to a component other than the active ingredient present in the pharmaceutical composition, and is typically selected to be harmless to the subject. It will be decided in consideration of the risks and benefits depending on the situation.
- compositions in the present disclosure includes a pharmaceutically acceptable carrier and can be prepared in the form of a lyophilized formulation or an aqueous solution formulation.
- the pharmaceutically acceptable carrier is typically non-toxic to the recipient in doses and concentrations used, eg, buffers such as phosphate, citrate, histidine; ascorbic acid and methionine. Containing antioxidants; preservatives (eg octadecyldimethylbenzylammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propylparaben; catechol; resorcinol; Cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptide; protein such as serum albumin, gelatin, or immunoglobulin; hydrophilic poly, such as polyvinylpyrrolidone -Amino acids such as glycine, glutamine, asparagine, histidine,
- an interstitial drug dispersing agent such as soluble hyaluronidase glycoprotein (sHASEGP) may be included (US2005 / 0260186, US2006 / 0104968).
- sHASEGP may be combined with one or more glycosaminoglycanases such as chondrokinase.
- an IL-8 signal inhibitor e.g., an IL-8 inhibitor (e.g., an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor
- an IL-8 signal inhibitor e.g., an IL-8 inhibitor (e.g., an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor
- an IL-8 signal inhibitor can be used as a pharmaceutical composition. based on.
- the IL-8 signal inhibitor for example, IL-8 inhibitor (for example, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor) in the present disclosure can be used for diagnosis of diseases in which IL-8 is excessively present, for example. Useful for treatment or prevention.
- an IL-8 signal inhibitor in the present disclosure is administered parenterally, intrapulmonary, It is administered by any suitable means, such as intranasal administration or, if local treatment is desired, intralesional administration.
- parenteral injection include intramuscular administration, intravenous administration, intraarterial administration, intraperitoneal administration, or subcutaneous administration. Depending on whether administration is short-term or long-term, it may be administered by an appropriate route such as injection such as intravenous injection or subcutaneous injection.
- Various dosing schedules can be performed.
- the IL-8 signal inhibitor in the present disclosure can be formulated, dosed, and administered in a manner tailored to medical utility.
- the present disclosure relates to products comprising IL-8 signaling inhibitors useful for diagnosis, treatment, and / or prevention for the diseases disclosed herein.
- the product includes a container and a label or package insert associated with the container. Suitable containers include, for example, bottles, vials, intravenous solution bags.
- IL-8 signal inhibitor for example, IL-8 inhibitor (for example, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor) in the present disclosure in the treatment or prevention of diseases
- IL-8 signal inhibitor for example, IL-8 inhibitor (for example, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor
- Appropriate dosage and administration interval of IL-8 signal inhibitor for example, IL-8 inhibitor (for example, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor) in the present disclosure in the treatment or prevention of diseases
- IL-8 signal inhibitor for example, IL-8 inhibitor (for example, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor
- the therapeutic effect may include, for example, prevention of disease onset or recurrence, amelioration of symptoms, reduction of any direct or indirect pathological consequences of the disease, prevention of metastasis, reduction of disease progression rate. Including recovery, alleviation, remission or improved prognosis of the disease state.
- the present disclosure encompasses a disease or condition in which IL-8 is present in excess, or a disease in which IL-8 signal is involved or can be involved in the onset, progression, exacerbation, recurrence, etc. of a disease state.
- IL-8 related disease it is called “IL-8 related disease”.
- the present disclosure contains an IL-8 signal inhibitor (eg, an IL-8 inhibitor (eg, an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor) as an active ingredient.
- the present invention relates to a composition for treating or preventing IL-8 related diseases.
- the composition may further comprise a pharmaceutically acceptable carrier.
- the present disclosure provides for use in the treatment or prevention of IL-8 related diseases (eg, endometriosis, adenomyosis, dysmenorrhea, adhesions, fibrotic diseases) It relates to an IL-8 signal inhibitor (eg, an IL-8 inhibitor (eg, an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor).
- IL-8 related diseases eg, endometriosis, adenomyosis, dysmenorrhea, adhesions, fibrotic diseases
- an IL-8 signal inhibitor eg, an IL-8 inhibitor (eg, an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor).
- the present disclosure provides diagnostic compositions, therapeutic compositions for IL-8 related diseases (eg, endometriosis, adenomyosis, dysmenorrhea, adhesions, fibrosis)
- IL-8 related diseases eg, endometriosis, adenomyosis, dysmenorrhea, adhesions, fibrosis
- an IL-8 signal inhibitor eg, an IL-8 inhibitor (eg, an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor
- the disclosure provides an IL-8 signal inhibitor (eg, an IL-8 inhibitor (eg, an anti-IL-8 antibody), a CXCR1 inhibitor, or a CXCR2 inhibitor) or a therapeutic comprising the same IL-8 related diseases (eg, endometriosis, uterine adenomyosis, dysmenorrhea, adhesions) comprising administering (in an effective amount) a composition or prophylactic composition to a subject in need thereof , Fibrotic diseases, etc.).
- the subject in need thereof may be a subject suffering from or likely to suffer from the IL-8 related disease.
- the composition may further comprise a pharmaceutically acceptable carrier.
- the present disclosure provides a medicament for the treatment or prevention of IL-8 related diseases (eg, endometriosis, adenomyosis, dysmenorrhea, adhesions, fibrotic diseases, etc.).
- IL-8 signal inhibitor of the present disclosure for example, IL-8 inhibitor (for example, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor) or a therapeutic or prophylactic composition containing the same in manufacture Concerning the use of things.
- the composition may further comprise a pharmaceutically acceptable carrier.
- an IL-8 that is subject to treatment or prevention with an IL-8 signal inhibitor (eg, an IL-8 inhibitor (eg, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor) in the present disclosure.
- an IL-8 signal inhibitor eg, an IL-8 inhibitor (eg, anti-IL-8 antibody), CXCR1 inhibitor, or CXCR2 inhibitor
- 8 Related diseases include, for example, endometriosis; adenomyosis; dysmenorrhea; adhesion of Asherman syndrome; infertility; pain in endometriosis, adenomyosis or dysmenorrhea; And pain caused by adhesion, fibrosis or inflammation; inflammatory skin diseases such as inflammatory keratosis, atopic dermatitis, contact dermatitis, palmoplantar pustulosis, psoriasis; rheumatoid arthritis, systemic lupus erythematosus (SLE
- the IL-8 related disease may be a disease in a mammal, preferably a disease in a primate, and a disease in humans (eg, human endometriosis for endometriosis; uterine adenomyosis) The same applies to human uterine adenomyosis and the like below.
- a patient (subject) of an IL-8 related disease to be treated or prevented by the IL-8 signal inhibitor of the present disclosure suffers from or is affected by the IL-8 related disease. It may be a subject of fear.
- the subject who may be suffering from IL-8 related disease is not limited, but may be a subject who has suffered from IL-8 related disease in the past, and whose symptoms may relapse, Alternatively, it may be a subject suspected of having developed an IL-8 related disease before a doctor or the like makes a definitive diagnosis with an IL-8 related disease, for example, by pathological findings.
- prevention and treatment of IL-8 related diseases may be understood interchangeably in some cases.
- prevention of IL-8 related diseases may be understood in general terms well known to those skilled in the art, where appropriate.
- the IL-8 related diseases in this disclosure are responsive to IL-8 signals.
- endometriosis is not intended to be limiting, but tissue having the same or similar form and function as endometrial tissue may be ectopic outside the uterus (usually in the pelvis, sometimes in the abdominal cavity). It rarely occurs in the thoracic cavity and proliferates to bring about symptoms (Japan Obstetrics and Gynecology Association, 1993). Although it is not a malignant tumor, it progresses over time, causing tumor formation and surrounding adhesions, severe abdominal pain and pelvic pain during menstruation and other times, sexual intercourse pain, defecation pain, and infertility (fertility) It is a coming disease.
- Fibrosis occurs when the pathological state of the part where adhesion has occurred in the lesioned part (disease site) of endometriosis progresses. Collagen is generated in the fibrotic part, which causes fibrosis.
- the progression of fibrosis in endometriosis is generally understood to be irreversible. Therefore, there has been no report on a method for stopping or improving the progression of fibrosis in endometriosis.
- endometriosis is a disease that presents various pathological findings in the process of development from the onset of the disease state and progresses through various stages.
- a non-human primate endometriosis in vivo model with menstruation similar to that of humans has recently been created, and the pathogenesis has finally been elucidated. is there.
- the present inventors have surgically induced endometriosis in a cynomolgus monkey endometriosis in vivo model having menstruation similar to that of humans, and administered an IL-8 signal inhibitor to the living body, thereby allowing endometriosis to occur.
- the epithelial cells and stroma cells in the lesions of the atrophy change atrophically, or the endometriosis interstitium decreases, so that even smooth muscles shrink and the growth of the endometriosis lesions can be suppressed.
- the present inventors confirmed regeneration of muscle tissue that was not seen in the vehicle group in the IL-8 signal inhibitor administration group, and the lesion that had infiltrated the muscle layer regressed and replaced with the muscle tissue and returned. I thought.
- the IL-8 signal inhibitor in the present disclosure can treat or prevent a condition that can be regarded as a fibrotic disease, i.e., fibrosis at a specific stage in endometriosis.
- IL-8 signal inhibitor is a promising therapeutic agent or prophylactic agent compared with existing therapeutic agents for endometriosis. Furthermore, it is surprising that endometriosis can be treated or prevented with a single agent of an IL-8 signal inhibitor without using in combination with a hormonal agent. In one embodiment, endometriosis in the present disclosure is responsive to IL-8 signal.
- uterine adenomyosis is a state in which endometrial tissue, that is, an endometrial gland and an endometrial stroma (stroma) surrounding the endometrial tissue exist in the uterine smooth muscle layer, although not limited thereto.
- endometrial tissue that is, an endometrial gland and an endometrial stroma (stroma) surrounding the endometrial tissue exist in the uterine smooth muscle layer, although not limited thereto.
- stroma endometrial stroma surrounding the endometrial tissue
- the present inventors confirmed and identified that the cynomolgus monkey in vivo model described above spontaneously developed uterine adenomyosis and administered an IL-8 signal inhibitor to the living body. Successfully demonstrated for the first time in the world that it can be controlled. In the cynomolgus monkey in vivo model, the IL-8 signal inhibitor did not affect the sexual cycle (see this example), so the IL-8 signal inhibitor was compared to the existing treatment for adenomyosis. It is a promising therapeutic or preventive agent.
- the uterine adenomyosis can be treated or prevented with a single agent of IL-8 signal inhibitor without using in combination with a hormonal agent.
- an IL-8 signal inhibitor By administering an IL-8 signal inhibitor, the enlarged uterine wall in uterine adenomyosis can be thinned, and a therapeutic or preventive effect on fibrosis in uterine adenomyosis is conceived.
- the uterine adenomyosis in the present disclosure is responsive to IL-8 signal.
- dysmenorrhea is not limited, but may refer to symptoms that cause trouble in daily life, particularly among menstrual symptoms. It is known that dysmenorrhea may involve endometriosis, adenomyosis, excessive menstruation, etc., and typical symptoms include, for example, menstrual abdominal pain, particularly lower abdominal pain In addition, back pain, headache, nausea, anemia, malaise and the like can be mentioned. Dysmenorrhea is roughly classified into two types according to its cause: functional (primary) dysmenorrhea and organic (secondary) dysmenorrhea.
- the cause of functional dysmenorrhea includes, for example, excessive secretion of prostaglandins, or those that occur with progression of diseases such as uterine fibroids and endometriosis.
- Examples of the causes of organic dysmenorrhea include those that occur with the progression of diseases such as uterine fibroids and endometriosis.
- dysmenorrhea is known to be caused by inflammation inside and outside the uterus, and IL-8 is known as an anti-inflammatory cytokine. Accordingly, in one embodiment, the IL-8 signal inhibitor in the present disclosure can treat or prevent dysmenorrhea.
- the IL-8 signaling inhibitor in the present disclosure comprises dysmenorrhea with endometriosis or adenomyosis, dysmenorrhea suspected of endometriosis or adenomyosis, Dysmenorrhea with inflammation or painful dysmenorrhea by adhesion can be treated or prevented.
- dysmenorrhea in the present disclosure is responsive to IL-8 signal.
- the term “dysmenorrhea in which endometriosis or adenomyosis is suspected” is not limited, but there is a risk that the symptoms may relapse in the past due to endometriosis or adenomyosis.
- the IL-8 signaling inhibitor in the present disclosure treats or prevents dysmenorrhea with endometriosis or adenomyosis, or dysmenorrhea suspected of endometriosis or adenomyosis.
- IL-8 signaling inhibitors do not affect the sexual cycle, and treat or prevent endometriosis and uterine adenomyosis with a single agent regardless of concomitant use with hormonal agents. This is advantageous.
- an IL-8 signal inhibitor can treat or prevent adhesions in addition to endometriosis and adenomyosis.
- Dysmenorrhea is characterized by pain such as abdominal pain. Not only adhesions in endometriosis, but also adhesions due to other symptoms or diseases cause pain to patients due to adhesion of tissues or organs. Naturally it is possible to cause.
- adhesions between genital appendages and pain do not correlate with adhesions in the pelvis, but Douglas fossa adhesions and pain are correlated (Porpora et al, The Journal of the American Association of Gynecologic Laparoscopists 1999; 6: 429-434 .), And it is known that pain is caused by the adhesion between organs. In endometriosis, production of ectopic nerve fibers and NGF is known to cause pain and exacerbation of pain (Anaf et al, Hum Reprod 2000; 15: 1744-1750., Berkley et al. al, Proc Natl Acad Sci USA 2004; 101: 11094-11098.
- an IL-8 signal inhibitor in the present disclosure is pain in endometriosis or adenomyosis (chronic pain, pain during menstruation, etc.), pain in dysmenorrhea, or adhesion, fibrosis Alternatively, pain due to inflammation can be treated or prevented.
- the chronic pain includes, but is not intended to be limited, chronic lower abdominal pain or chronic pelvic pain.
- “does not affect the sexual cycle” in the present disclosure means that the periodic menstrual cycle and ovulation in the subject (female for humans and female for non-human animals) substantially interfere.
- the periodicity of the cycle may be determined from the presence or absence of bleeding during menstruation as observed in this example, fluctuations in blood or urinary estrogen concentration and progesterone concentration You may judge from the fluctuation
- Hormone eg, estrogen / progesterone combinations, progesterone preparations, GnRH agonists, danazol
- whether or not it has been substantially interfered with is, for example, an IL-8 signal inhibitor compared to a control group receiving hormonal therapy known to affect the sexual cycle In the group that received the administration, it may be judged whether the menstrual cycle is not interfered statistically or as a trend.
- the IL-8 signaling inhibitor in the present disclosure does not affect the sexual cycle in a subject with endometriosis, adenomyosis, or dysmenorrhea administered it, thus ovulation It is recalled that it does not interfere with pregnancy and, unlike conventional hormone therapy, pregnancy is possible.
- a subject can avoid side effects such as menopause due to a low estrogen state by not using a hormonal agent.
- a living body has a wound healing function that attempts to heal a damaged or wounded site when cells, tissues, organs, or organs in the living body are damaged or wounded.
- adhesions between cells, tissues, organs or organs may occur which are otherwise undesirable during the wound healing process.
- typically such an adhesive state is referred to as adhesion. While there are patients who do not have subjective symptoms even when adhesions occur, there are many patients who need treatment with symptoms such as pain, infertility, and abdominal bloating.
- adhesions are adhesions that occur (occur) after surgery (for example, adhesions that occur (occur) directly or indirectly at the fracture surface during surgical operation, around the suture site, or at the site of abrasion such as gauze) Intrauterine vaginal adhesions (asherman syndrome)) caused by scraping treatment; adhesions due to drugs (eg topical administration of drugs to organs); diseases (eg endometriosis, uterine adenomyosis) Adhesion due to infectious or infiltrating cancer cells or tissues, inflammatory diseases associated with infection (eg tubitis, ovitis, pelvic peritonitis); intestinal obstruction with adhesion; spontaneously occurring adhesion, etc. Is mentioned.
- the present inventors have observed that adhesion to the laparotomy site different from endometriosis that occurred after laparotomy is performed by administering an IL-8 signal inhibitor to the living body. I accidentally found that adhesions decreased.
- the adhesions in the present disclosure are responsive to IL-8 signals.
- infertility is not intended to be limited, but it does not mean pregnancy for a certain period of time (generally a year) even though healthy men and women who want to conceive have sex without contraception. (Japan Society of Obstetrics and Gynecology, 2016).
- the infertility may be infertility that occurs after adhesion (particularly due to adhesions) or infertility due to inflammation in the abdominal cavity.
- the infertility in this disclosure is responsive to IL-8 signal.
- palmoplantar pustulosis refers to a disease in which countless rashes (pustules) can occur on the palms and soles of the feet, although not intended to be limited.
- Pustules are sterile and free of bacteria and mold, with neutrophils accumulating in the skin and high concentrations of IL-8 (Skov et al, J Immunol 2008; 181: 669-679) .
- Symptoms appear chronically after repeated symptoms over a long period of time.
- administration of anti-IL-8 antibody HuMab10F8 improved the pathology (Skov et al, J Immunol 2008; 181: 669-679).
- palmoplantar pustulosis in the present disclosure is responsive to IL-8 signal.
- ANCA-related nephritis refers to nephritis positive for ANCA (anti-neutrophiltrocytoplasmic antibody).
- ANCA-related nephritis is a disease that causes inflammation in kidney blood vessels such as glomeruli, and typical pathological findings indicate necrotizing crescent-forming nephritis. It leads to renal failure.
- RPGN Rapid Progressive Nephritis Syndrome
- IL-8 was highly expressed in the lesion, and the migration ability of patient neutrophils was increased by ANCA treatment, and the migration ability was suppressed by anti-IL-8 antibody treatment (Cockwell et al, Kidney Int. 1999 Mar; 55 (3): 852-63.).
- the ANCA associated nephritis in the present disclosure is responsive to IL-8 signal.
- cystic fibrosis is a systemic autosomal recessive disease caused by a gene mutation of cystic fibrosis transmembrane conductance regulator (CFTR).
- CFTR cystic fibrosis transmembrane conductance regulator
- Systemic secretions and mucus such as airway fluid, intestinal fluid, and pancreatic juice become extremely viscous, obstructing the lumen, making it susceptible to infection, meconium ileus, and pancreatic atrophy, resulting in poor digestion and absorption due to pancreatic exocrine insufficiency Symptoms such as respiratory failure appear after repeated respiratory infections.
- cystic fibrosis in the present disclosure is responsive to IL-8 signal.
- psoriasis is also known as an inflammatory disease, although not intended to be limited.
- a typical symptom is a well-defined red papule or aspect covered with silvery scales.
- Common triggers include trauma, infection, and certain drugs. Usually, there are few symptoms and sometimes mild itching, but a cosmetic point of view may be the main complaint. Some people become more severe with painful arthritis.
- US2003 / 0077283A1 discloses a local therapy for psoriasis using an anti-IL-8 neutralizing antibody.
- an anti-IL-8 neutralizing antibody treatment drug is sold under the name of Abcream (registered trademark).
- Abcream registered trademark
- fibrosis-related disease or condition is comprehensively referred to as a “fibrotic disease” by taking a form of the fibrosis.
- IL-8 signal IL-8 signal inhibitor suppressed immune cell infiltration, decreased collagen-rich interstitium sites (see this example), and in vitro assays
- IL-8 signal IL-8 signal inhibitor is a promising therapeutic agent for fibrosis because the inhibitor showed the migration inhibitory action of neutrophils producing MCP-1 (known as macrophage migration factor and fibrosis promoting factor) and the like Or it is a prophylactic agent as above-mentioned.
- various anti-IL-8 antibodies suppressed the increased expression of connecting tissue growth factor (CTGF), which is a fibrosis promoting factor, in in vitro tests. This result confirms that IL-8 signal inhibitors such as anti-IL-8 antibodies are promising therapeutic or preventive agents for fibrotic diseases.
- CTGF connecting tissue growth factor
- adhesion or fibrosis in endometriosis in the present disclosure occurs in the process in which uterine-derived tissue develops and proliferates outside the uterus (eg, pelvic organs, intraperitoneal cavity, or thoracic cavity). obtain.
- liver fibrosis is not intended to be limited, but it is triggered by chronic inflammation caused by viral infection or fattening, and the extracellular matrix (ECM) such as collagen is overproduced or degraded in the liver, resulting in fibrosis. It refers to the disease.
- ECM extracellular matrix
- liver fibrosis include cirrhosis caused by chronic inflammation such as viral hepatitis, alcoholic hepatitis, nonalcoholic fatty liver disease (NAFLD), and primary biliary cirrhosis (PBC).
- Increased IL-8 in chronic liver diseases such as type A and B viral hepatitis, alcoholic hepatitis, primary biliary cirrhosis (Mediators Inflamm.
- liver fibrosis in the present disclosure is responsive to IL-8 signal.
- renal fibrosis refers to a disease in which inflammation is triggered, but an extracellular matrix such as collagen is excessively produced or degraded in the kidney and fibrosis is not intended to be limited.
- Examples of renal fibrosis include chronic kidney disease caused by chronic inflammation such as chronic nephritis and diabetes. When chronic kidney disease progresses, it causes fibrosis of the kidneys regardless of the causative disease, and the fibrosis that occurs once is irreversible, and the kidney is forced to introduce dialysis or kidney transplantation. Renal fibroblasts enhanced IL-8 production via IL-1 (Kidney Int.
- renal fibrosis in the present disclosure is responsive to IL-8 signal.
- pulmonary fibrosis refers to a disease that is not intended to be limited, but is triggered by inflammation, resulting in excessive production or degradation of extracellular matrix such as collagen in the lung or bronchus, resulting in fibrosis.
- pulmonary fibrosis include chronic obstructive pulmonary disease (COPD), pulmonary fibrosis combined with emphysema (combined pulmonary fibrosis and emphysema; CPFE), idiopathic interstitial pneumonia (idiopathic interstitial pneumonias: IIPs) and idiopathic lung Fibrosis (idiopathic pulmonary fibrosis: IPF) is mentioned.
- COPD chronic obstructive pulmonary disease
- CPFE combined with emphysema
- IIPs idiopathic interstitial pneumonias
- IPF idiopathic lung Fibrosis
- COPD Chronic obstructive pulmonary disease
- COPD refers to an inflammatory disease of the lung caused by long-term inhalation exposure to harmful substances, mainly cigarette smoke, although not intended to be limited (COPD (chronic obstructive pulmonary disease)) Guidelines for diagnosis and treatment 4th edition).
- COPD chronic obstructive pulmonary disease
- Chronic airways and lung inflammation cause reduced alveolar elasticity and bronchial lumen constriction, resulting in poor air flow to the lungs and airflow obstruction, etc. Appears.
- IL-8 is high in patients and there are many neutrophils, and that there is a correlation between the chemotaxis index and lung function FEV1 / FVC in the patient neutrophil migration assay (Yamamoto et al , Chest.
- Pulmonary fibrosis (combined pulmonary fibrosis and emphysema; CPFE) is not intended to be a limitation, but refers to pulmonary emphysema, a lesion in which the lung breaks and spreads, and a poor prognosis disease associated with pulmonary fibrosis (Cottin et al , Eur Respir J. 2005 Oct; 26 (4): 586-93.).
- IL-8 is high in bronchoalveolar lavage fluid, and IL-8 concentration in bronchoalveolar lavage fluid correlates with low-concentration absorption area (LAA) indicating emphysematous lesions in chest CT (Respirology) .
- LAA low-concentration absorption area
- IIPs is a general term for various interstitial pneumonia whose cause cannot be identified, and is a disease in which the alveolar wall becomes inflamed or damaged, the alveolar wall becomes thick and hard and fibrosis, and the gas exchange function decreases (idiopathic Interstitial pneumonia diagnosis and treatment guide revised 2nd edition; Travis et al, Am J Respir Crit Care Med. 2013 Sep 15; 188 (6): 733-48).
- IPF is the most frequent and most difficult to treat of IIPs, and has a prognostic lung disease with a chronic and progressive course and advanced fibrosis leading to irreversible honeycomb lung formation (idiopathic stroma) Guideline for Diagnosis and Treatment of Pneumonia (2nd Edition; Raghu et al, Am J Respir Crit Care Med. 2011 Mar 15; 183 (6): 788-824).
- IPF requires high levels of IL-8 in serum and bronchoalveolar lavage fluid from patients with disease (Car et al, Am J Respir Crit Care Med 1994; 149: 655-659, Ziegenhagen et al, Am J Respir Crit Care Med 1998; 157: 762-768), and bronchoalveolar lavage fluid with increased neutrophils migrating by IL-8 has a high prognosis (Haslam et al, Thorax 1980; 35: 328- 339, Turner-Warwick M et al, Am Rev Respir Dis 1987; 135: 26-34), suggesting that IL-8 and neutrophils are deeply involved in the pathogenesis of IPF.
- IL-8 is the cause of the disease because of the presence of SNIP in IL-8 and high concentrations of local IL-8 (Ahn-8et al, Respir Res. 2011 Jun 8; 12: 73) Is inferred.
- pulmonary fibrosis, COPD, CPFE, IIPs or IPF in the present disclosure is responsive to IL-8 signals.
- the present disclosure relates to IL-8 signal inhibitors (eg, anti-IL-8 antibodies) for use in inhibiting fibrosis factor production.
- the present disclosure provides a method of inhibiting fibrosis factor production in a subject, or a use in the method, comprising administering to the subject an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody).
- IL-8 signal inhibitor for example, anti-IL-8 antibody.
- the present disclosure relates to a pharmaceutical composition for inhibiting fibrosis factor production comprising an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody).
- the present disclosure relates to the use of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) in the manufacture of a medicament for inhibiting fibrosis factor production.
- the present disclosure relates to the use of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) in inhibiting fibrosis factor production.
- the present disclosure relates to a method for producing a pharmaceutical composition for inhibiting fibrosis factor production, comprising a step of mixing an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) and a pharmaceutically acceptable carrier.
- the fibrosis factor may include, for example, MCP-1 (Monocyte Chemotactic Protein-1). Such production inhibition of fibrosis factor may occur, for example, in immune cells such as neutrophils.
- the present disclosure relates to IL-8 signal inhibitors (eg, anti-IL-8 antibodies) for use in inhibiting angiogenesis.
- the present disclosure provides a method of inhibiting angiogenesis in a subject comprising administering an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) to the subject, or IL- for use in the method.
- an IL-8 signal inhibitor eg, an anti-IL-8 antibody
- the present disclosure relates to a pharmaceutical composition for inhibiting angiogenesis comprising an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody).
- the present disclosure relates to the use of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) in the manufacture of a medicament for inhibiting angiogenesis.
- an IL-8 signal inhibitor eg, an anti-IL-8 antibody
- this indication is related with the manufacturing method of the pharmaceutical composition for angiogenesis inhibition including the process of mixing an IL-8 signal inhibitor (for example, anti-IL-8 antibody) and a pharmaceutically acceptable carrier.
- the present disclosure provides for use in the suppression of infiltration of immune cells into the lesion or surrounding environment of an IL-8 related disease (sometimes referred to as migration).
- IL-8 signal inhibitor for example, anti-IL-8 antibody.
- the present disclosure provides for administering to the subject an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) to the lesion of an IL-8 related disease in an immune cell or a surrounding environment thereof in the subject.
- the present invention relates to an IL-8 signal inhibitor (for example, an anti-IL-8 antibody) for use in a method for suppressing invasion of a bacterium, or for use in the method.
- the present disclosure provides a medicament for suppressing infiltration of an immune cell into a lesion site of an IL-8 related disease or its surrounding environment, comprising an effective amount of an IL-8 signal inhibitor (for example, an anti-IL-8 antibody).
- an IL-8 signal inhibitor for example, an anti-IL-8 antibody
- the present disclosure relates to the use of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) in the manufacture of a medicament for suppressing the infiltration of immune cells into the lesion site of an IL-8 related disease or the surrounding environment.
- the present disclosure relates to the use of an IL-8 signal inhibitor (for example, an anti-IL-8 antibody) in suppressing infiltration of an immune cell into a lesion site of an IL-8 related disease or its surrounding environment.
- the present disclosure provides a process for mixing an IL-8 signal inhibitor (for example, an anti-IL-8 antibody) and a pharmaceutically acceptable carrier into a lesion site of an IL-8 related disease in an immune cell or its surrounding environment.
- the present invention relates to a method for producing a pharmaceutical composition for the suppression of infiltration.
- immune cells include, for example, but are not limited to, neutrophils, macrophages, monocytes, helper T cells, killer T cells, dendritic cells, NK cells, and the like. Macrophages and monocytes are preferred.
- the IL-8-related disease may be any of the above-mentioned diseases, such as endometriosis, uterine adenomyosis, dysmenorrhea, Adhesion, fibrosis, etc. may be mentioned, and it is contemplated that immune cells infiltrate or migrate to the lesion (disease site) of such an IL-8 related disease or the vicinity thereof.
- endometriosis infiltrate or migrate to the lesion (disease site) of such an IL-8 related disease or the vicinity thereof.
- infiltration or migration into the abdominal cavity or ascites would be contemplated.
- pulmonary fibrosis Those skilled in the art will understand that infiltration or migration into the pleural cavity or pleural effusion would be contemplated.
- the present disclosure relates to an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) for use in inhibiting aromatase production in a lesion of endometriosis or adenomyosis.
- an IL-8 signal inhibitor eg, an anti-IL-8 antibody
- the present disclosure provides a method for inhibiting aromatase production in a subject, comprising administering an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) to the subject, or an IL for use in the method -8 signal inhibitor (eg, anti-IL-8 antibody).
- the present disclosure relates to a pharmaceutical composition for inhibiting aromatase production comprising an effective amount of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody).
- the present disclosure relates to the use of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) in the manufacture of a medicament for inhibiting aromatase production.
- the present disclosure relates to the use of an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) in inhibiting the production of aromatase in a lesion of endometriosis or adenomyosis.
- the present disclosure provides for the production of aromatase in a lesion of endometriosis or adenomyosis comprising mixing an IL-8 signal inhibitor (eg, an anti-IL-8 antibody) and a pharmaceutically acceptable carrier.
- the present invention relates to a method for producing a pharmaceutical composition for inhibition.
- first, second, third, fourth,... are used to represent various elements, these elements should not be limited by those terms Is understood. These terms are only used to distinguish one element from another, for example, the first element is referred to as the second element, and similarly, the second element is referred to as the first element. Is understood to be possible without departing from the scope of the present disclosure.
- Example 1 Preparation of a novel Fc region variant with increased FcRn binding under acidic pH conditions for improvement of plasma retention.
- IgG antibodies incorporated into cells are treated with FcRn under acidic pH conditions in endosomes. It is known to be returned to plasma by binding to. Therefore, IgG antibodies generally have a longer plasma half-life than proteins that do not bind to FcRn.
- we have already known a method to improve the retention of antibodies in plasma by introducing amino acid modifications into the Fc region of antibodies to increase the binding ability (binding affinity) to FcRn under acidic pH conditions. It has been.
- Fv4-IgG1 consisting of VH3-IgG1m (SEQ ID NO: 2) as the heavy chain and VL3-CK (SEQ ID NO: 110) as the light chain
- Fv4-YTE consisting of VH3-YTE (SEQ ID NO: 3) as the heavy chain and VL3-CK as the light chain
- Fv4-LS consisting of VH3-LS (SEQ ID NO: 4) as the heavy chain and VL3-CK as the light chain
- Fv4-N434H consisting of VH3-N434H (SEQ ID NO: 5) as the heavy chain and VL3-CK as the light chain
- VH3-F1847m SEQ ID NO: 6
- Fv4-F1847m consisting of VL3-CK as the light chain
- VH3-F1848m SEQ ID NO: 7
- Fv4-F1848m consisting of VL3-CK as the light chain
- a diluted FcRn solution and a running buffer (as a reference solution) were injected, and human FcRn was allowed to interact with the antibody captured on the sensor chip.
- a running buffer 50 mM sodium phosphate, 150 mM NaCl, 0.05% (w / v) Tween 20, pH 6.0 was used, and each buffer was also used for dilution of FcRn.
- 10 mM glycine-HCl, pH 1.5 was used. All measurements were performed at 25 ° C.
- ADA Anti-pharmaceutical antibody
- rheumatoid arthritis RA
- RF rheumatoid factor
- humanized anti-CD4 IgG1 antibody with N434H (Asn434His) mutation induces significant rheumatoid factor binding (Clin Pharmacol Ther. 2011 Feb; 89 (2): 283-90).
- N434H mutation in human IgG1 increases the binding of rheumatoid factor to the Fc region of the antibody compared to the parent human IgG1.
- Rheumatoid factors are polyclonal autoantibodies to human IgG, and their epitopes in human IgG vary from clone to clone, but they are located in the CH2 / CH3 interface region and in the CH3 domain that can overlap with the FcRn binding epitope. It seems to do. Therefore, a mutation that increases the binding activity (binding affinity) to FcRn may increase the binding activity (binding affinity) of a rheumatoid factor to a specific clone.
- the mixture was then added to a Streptavidin coated MULTI-ARRAY 96 well plate (Meso Scale Discovery) and the plate was incubated for 2 hours at room temperature and washed. After adding Read Buffer T ( ⁇ 4) (Meso Scale Discovery) to each well, the plate was immediately set in a SECTOR imager 2400 Reader (Meso Scale Discovery), and chemiluminescence was measured.
- FIGS. Fv4-IgG1 with native human IgG1 showed only weak rheumatoid factor binding
- Fv4-YTE FIG. 2
- Fv4-LS existing FcRn-binding increased Fc region variants
- FIG. 3 and Fv4-N434H both had significantly increased rheumatoid factor binding in multiple donors.
- Fv4-F1847m FIG. 5
- Fv4-F1848m Fig. 6
- Fv4-F1886m Fig. 7
- Fv4-F1889m Fig.
- FIG. 11 shows the average value of the binding property to rheumatoid factor in the blood of 30 RA patients for each variant. All six new variants show lower binding than the three existing variants (YTE, LS, N434H), and even lower rheumatoid factor compared to native human IgG1 The binding property of was shown.
- Example 3 PK evaluation in cynomolgus monkeys of a novel Fc region variant with increased FcRn binding under acidic pH conditions
- a novel Fc region variant confirmed to be inhibited from binding to rheumatoid factor
- the effect of improving plasma retention in cynomolgus monkeys was evaluated by the following method.
- OHB-IgG1 As anti-human IgE antibodies, 8 types of OHB-IgG1, OHB-LS, OHB-N434A, OHB-F1847m, OHB-F1848m, OHB-F1886m, OHB-F1889m, and OHB-F1927m were used.
- the concentration of anti-human IgE antibody in monkey plasma was calculated from the absorbance of the calibration curve using analysis software SOFTmax PRO (Molecular Devices). The measured concentration transition of anti-human IgE antibody in monkey plasma is shown in FIG. In addition, disappearance clearance was calculated by moment analysis from the measured changes in the concentration of anti-human IgE antibody in monkey plasma using Phoenix WinNonlin Ver.6.2 (Pharsight Corporation). The calculated pharmacokinetic parameters are shown in Table 3. Individuals who tested positive for anti-administered plasma antibodies in plasma were excluded from monkey plasma anti-human IgE antibody concentration transitions and clearance calculations.
- Fv4-F1718 consisting of VH3-F1718 (SEQ ID NO: 21) as the heavy chain
- VL3-CK VL3-CK
- a calibration curve sample containing anti-human IL-6 receptor antibody with a plasma concentration of 0.8, 0.4, 0.2, 0.1, 0.05, 0.025, 0.0125 ⁇ g / mL and a mouse plasma measurement sample diluted 100 times or more were prepared.
- the reaction of the reaction with Biotinylated Anti-human IL-6 R Antibody (R & D) for 1 hour at room temperature and Streptavidin-PolyHRP80 (Stereospecific Detection Technologies) for 1 hour at room temperature is the TMB One Component HRP reaction. This was performed using Microwell Substrate (BioFX Laboratories) as a substrate. The absorbance at 450 nm of the reaction solution in each well whose reaction was stopped by adding 1N-Sulfuric acid (Showa Chemical) was measured with a microplate reader. The antibody concentration in mouse plasma was calculated from the absorbance of the calibration curve using analysis software SOFTmax PRO (Molecular Devices).
- FIG. F1718 which is an Fc region variant for increasing FcRn binding at acidic pH described in WO2013 / 046704, did not show the effect of prolonging antibody PK and showed plasma retention equivalent to that of natural IgG1.
- F1718 described in WO2013 / 046704 four mutations N434Y / Y436V / Q438R / S440E are introduced into the Fc region.
- N434A / Y436V / Q438R / S440E have been introduced into F1848m newly found this time.
- F1848m is A (alanine).
- Example (3-2) F1848m showed an improvement in plasma retention compared to that of natural IgG1, whereas F1718 showed no improvement in plasma retention. From this, it is suggested that A (alanine) is preferable as the amino acid mutation introduced at position 434 as an amino acid mutation used for improving plasma retention, although not particularly limited. .
- Example 4 Production of humanized anti-human IL-8 antibody (4-1) Production of humanized anti-human IL-8 antibody hWS-4
- the humanized anti-IL-8 antibody disclosed in US6245894 (WO1996 / 002576) is human IL-8 (hereinafter referred to as hIL-8).
- the physiological action is blocked.
- a humanized anti-IL-8 antibody can be prepared by combining the variable region sequences of heavy and light chains disclosed in US6245894 with the constant region sequence of any human antibody.
- the constant region sequence of the human antibody is not particularly limited, and a natural human IgG1 sequence or a natural human IgG4 sequence can be used as the heavy chain constant region, and a natural human Kappa sequence can be used as the light chain constant region sequence.
- hWS4H-IgG1 which combines a heavy chain variable region RVHg and a natural human anti-IgG1 sequence as a heavy chain constant region.
- These genes were prepared by the method of Reference Example 1.
- a gene of hWS4L-k0MT (SEQ ID NO: 40) combining the light chain variable region RVLa and the natural human Kappa sequence as the light chain constant region was prepared by the method of Reference Example 1.
- An antibody combining the above heavy chain and light chain was prepared and used as a humanized WS-4 antibody (hereinafter, hWS-4).
- Hr9 the heavy chain variable region sequence is shown in SEQ ID NO: 33
- Hr9 / hWS4L the heavy chain variable region sequence is shown in SEQ ID NO: 33
- the antibody was expressed using FreeStyle 293F cells (Invitrogen) according to the protocol attached to the product. Purification of the antibody from the culture supernatant was performed by the method of Reference Example 2. As a result, the amount of antibody shown in Table 4 was obtained. Surprisingly, the expression level of Hr9 was about 8 times that of hWS-4.
- the running buffer a solution having the above composition was used, and this buffer was also used for dilution of human IL-8.
- 10 mM glycine-HCl, pH 1.5 was used for regeneration of the sensor chip. All measurements were performed at 37 ° C. Based on the association rate constant kon (1 / Ms) and the dissociation rate constant koff (1 / s), which are kinetic parameters calculated from the sensorgram obtained from the measurement, the KD for each antibody to human IL-8 ( M) was calculated. Biacore T200 Evaluation Software (GE Healthcare) was used for calculation of each parameter. The results are shown in Table 5. hWS-4 and Hr9 were confirmed to have equivalent binding affinity for human IL-8.
- Example 5 Acquisition of antibody having pH-dependent IL-8 binding ability (5-1) Preparation of Hr9-modified antibody for conferring pH dependence A study was conducted for the purpose of imparting pH-dependent IL-8 binding ability to Hr9 obtained in Example 4. Without being bound by a specific theory, it is considered that an antibody having a pH-dependent binding ability to IL-8 exhibits the following behavior in vivo.
- the antibody administered to the living body can strongly bind to IL-8 and block its function in an environment maintained at a neutral pH (for example, in plasma). A part of such a complex of IL-8 and antibody is taken into cells by non-specific interaction (pinocytosis) with the cell membrane (hereinafter referred to as non-specific uptake).
- the antibody dissociates IL-8 because the binding affinity of the antibody to IL-8 is weakened. Thereafter, the antibody that has dissociated IL-8 can return to the outside of the cell via FcRn. The antibody returning to the outside of the cell (in plasma) in this way can bind to another IL-8 again and block its function. It can be considered that an antibody having a pH-dependent binding ability to IL-8 can bind to IL-8 multiple times by the mechanism described above. On the other hand, in the case of an antibody that does not have such properties as the above antibody, one molecule of antibody can neutralize an antigen only once, and cannot neutralize an antigen multiple times.
- an IgG antibody has two Fabs, so one antibody molecule can neutralize two molecules of IL-8.
- an antibody that can bind to IL-8 multiple times can bind to IL-8 any number of times as long as it stays in the living body.
- a pH-dependent IL-8 binding antibody that has been taken up 10 times in a cell between administration and disappearance can neutralize up to 20 molecules of IL-8. is there. Therefore, an antibody that can bind to IL-8 multiple times has the advantage that a large amount of IL-8 can be neutralized even with a smaller amount of antibody.
- an antibody that can bind to IL-8 multiple times has the advantage that it can maintain a state that can neutralize IL-8 for a longer period of time when the same amount of antibody is administered.
- antibodies that can bind to IL-8 multiple times have the advantage of being able to block IL-8 biological activity more strongly when the same amount of antibody is administered. .
- amino acid modifications centered on histidine were introduced into the variable regions of Hr9-IgG1 and WS4L-k0MT in order to create antibodies that can bind to IL-8 multiple times. .
- the modifications shown in Table 6 were prepared by the methods of Reference Examples 1 and 2.
- the notation such as “Y97H” shown in Table 6 represents a mutation introduction position defined by Kabat numbering, an amino acid before mutation introduction, and an amino acid after mutation introduction.
- the expression “Y97H” indicates that the amino acid residue of Kabat numbering No. 97 was substituted from Y (tyrosine) to H (histidine).
- Y tyrosine
- H histidine
- Example 5-1 pH-dependent IL-8 binding ability
- the human IL-8 binding affinity of the antibody prepared in Example 5-1 was measured using Biacore T200 (GE Healthcare) as follows. The following two running buffers were used. ⁇ 0.05% tween20, 20 mM ACES, 150 mM NaCl, pH 7.4 ⁇ 0.05% tween20, 20 mM ACES, 150 mM NaCl, pH 5.8
- An appropriate amount of protein A / G (PIERCE) was immobilized on the sensor chip CM4 (GE Healthcare) by the amine coupling method, and the target antibody was captured there.
- Hr9 / L16 which contains L54H modification in the light chain, has a slightly enhanced human IL-8 binding affinity at neutral pH (pH 7.4) than Hr9, while human at acidic pH (pH 5.8). IL-8 binding affinity was reduced.
- anti-IL-8 antibodies H89 / WS4L, H89 / L12, and H89 / L16 that combine H89 containing Y97H modification in the heavy chain with various light chains all bind human IL-8 at acidic pH At the same time the affinity was reduced, the human IL-8 binding affinity at neutral pH was also reduced.
- H89 / L63 having H89-IgG1 (SEQ ID NO: 42) as the heavy chain and L63-k0MT (SEQ ID NO: 43) as the light chain has a human IL-8 binding affinity at neutral pH (pH 7.4) of Hr9
- the human IL-8 binding affinity at acidic pH (pH 5.8) was reduced.
- H89 / L63 had both koff (dissociation rate constant) and KD (dissociation constant) at pH 5.8 larger than Hr9.
- H89 / L63 has a property of easily releasing human IL-8 under acidic pH conditions in endosomes.
- H89 / L118 having H89-IgG1 as the heavy chain and L118-k0MT (SEQ ID NO: 44) as the light chain has a human IL-8 binding affinity (KD) of Hr9 at neutral pH conditions.
- human IL-8 binding affinity (KD) under acidic pH conditions was attenuated compared to Hr9.
- a pH-dependent antigen-binding antibody strongly neutralizes an antigen under neutral pH conditions (for example, in plasma).
- H89 / L118 acquired favorable properties compared with Hr9 at both these neutral pH and acidic pH. That is, useful amino acid modifications such as Y97H for the heavy chain of Hr9 and N50H / L54H / Q89K for the light chain were found. Although it is not particularly limited, it is shown that an excellent pH-dependent IL-8 binding antibody as a pharmaceutical can be prepared by introducing an amino acid modification selected from these alone or in combination.
- Example 6 Preparation of high-affinity antibody for mouse PK test
- the method for confirming the effect of an antibody on the disappearance rate of human IL-8 in a mouse is not particularly limited. There is a method of comparing the disappearance rate of human IL-8 from mouse plasma after administration to mice in a mixed state with IL-8.
- the reference antibody for use in the mouse PK test has a sufficiently strong binding affinity under conditions of neutral pH and acidic pH. Therefore, as a result of searching for a modification imparting high affinity to Hr9, H998 / L63 having H998-IgG1 (SEQ ID NO: 45) as a heavy chain and L63-k0MT as a light chain was created.
- H998 / L63 the binding affinity of human IL-8 was evaluated in the same manner as in Example 5-2.
- the resulting sensorgram is shown in FIG. H998 / L63 was shown to have a significantly slower dissociation rate and a stronger IL-8 binding affinity than Hr9 at both neutral and acidic pH conditions.
- analytical values such as dissociation rate constant (koff) and dissociation constant (KD) cannot be calculated accurately in the case of protein-protein interactions with such a slow dissociation rate. It is known.
- koff dissociation rate constant
- KD dissociation constant
- the analysis value is not shown here.
- the results of this experiment confirm that H998 / L63 has a very strong binding affinity at both neutral and acidic pH and is suitable as an antibody to be used as a comparison target in the mouse PK test. It was done.
- Example 7 Mouse PK test using pH-dependent IL-8 binding antibody H89 / L118 (7-1) Mouse PK test using H89 / L118 Evaluation of the disappearance rate of human IL-8 in vivo using H89 / L118 prepared in Example 5 and H998 / L63 prepared in Example 6 Carried out. The pharmacokinetics of human IL-8 after simultaneous administration of human IL-8 and anti-human IL-8 antibody to mice (C57BL / 6J, Charles river) was evaluated. A mixed solution of human IL-8 and anti-human IL-8 antibody (10 ⁇ g / mL and 200 ⁇ g / mL, respectively) was administered once at 10 mL / kg into the tail vein.
- Blood was collected 5 minutes, 2 hours, 4 hours, 7 hours, 1 day, 2 days, 3 days, 7 days, 14 days, 21 days, and 28 days after administration. The collected blood was immediately centrifuged at 4 ° C. and 15,000 rpm for 15 minutes to obtain plasma. The separated plasma was stored in a freezer set to ⁇ 20 ° C. or lower until measurement was performed.
- a calibration curve sample containing human IL-8 with a plasma concentration of 275, 91.7, 30.6, 10.2, 3.40, 1.13, 0.377 ng / mL and a mouse plasma measurement sample diluted 25-fold or more were prepared. After mixing, the mixture was reacted at 37 ° C. overnight, then dispensed at 50 ⁇ L into each well of the anti-human IL-8 antibody-immobilized plate, and then stirred at room temperature for 1 hour. The final concentration of hWS-4 was adjusted to 25 ⁇ g / mL.
- human IL-8 administered simultaneously with H89 / L118 is significantly faster in mouse plasma than human IL-8 administered simultaneously with H998 / L63. Indicated.
- H89 / L118 is about 19 times the rate of disappearance of human IL-8 compared to H998 / L63 It was shown to increase.
- Human IL-8 administered at the same time as the antibody is mostly bound to the antibody in the plasma and exists in a complex state.
- Human IL-8 bound to H998 / L63 exists in a state bound to an antibody with a strong affinity even in endosomes under acidic pH conditions. Later, H998 / L63 is returned to plasma via FcRn in a complex with human IL-8, so that human IL-8 is also returned to plasma at the same time. . Therefore, most of the human IL-8 taken up into the cells returns to the plasma again. That is, the rate of disappearance of human IL-8 from the plasma is markedly reduced when administered simultaneously with H998 / L63. On the other hand, as described above, human IL-8 incorporated into cells in the form of a complex with pH-dependent IL-8 binding antibody H89 / L118 is an antibody under acidic pH conditions in endosomes.
- IL-8 Dissociates from. Human IL-8 dissociated from the antibody migrates to the lysosome and is degraded. Therefore, pH-dependent IL-8 binding antibodies are significantly more potent than human IL-8 compared to IL-8 binding antibodies, such as H998 / L63, which have strong binding affinity at both acidic and neutral pH. It is possible to expedite the disappearance.
- the group administered with 8 mg / kg antibody was confirmed to have about twice the rate of disappearance of human IL-8. .
- the contents inferring one of the factors that are not intended to be bound by theory but that may bring about the above results will be described.
- the proportion of human IL-8 bound is preferably low.
- it is desirable that the percentage of free type not bound to antibodies is high.
- [Percentage of free human IL-8 in endosome (%)] [Free human IL-8 concentration in endosome] ⁇ [Total human IL-8 concentration in endosome] ⁇ 100
- the proportion of free human IL-8 in the endosome is desirably higher, for example, 20% is preferable to 0%, 40% is preferable to 20%, and 40% is preferable. Is preferably 60%, more preferably 80% over 60%, and more preferably 100% over 80%. It is thought that there is a correlation between the ratio of free human IL-8 in the endosome as described above and the binding affinity (KD) and / or dissociation rate constant (koff) for human IL-8 at acidic pH. It makes sense.
- KD binding affinity
- koff dissociation rate constant
- Example 8 Preparation and evaluation of pH-dependent IL-8 binding antibody H553 / L118 (8-1) Production of new H553 / L118 having pH-dependent IL-8 binding ability Therefore, an attempt was made to produce an antibody with a weaker human IL-8 binding affinity and / or faster dissociation rate under acidic pH conditions than H89 / L118. Based on H89 / L118, amino acid modifications centered on histidine were introduced, and modified antibodies shown in Table 10 were prepared in the same manner as in Example 5. In addition, the human IL-8 binding affinity of these antibodies was measured in the same manner as in Example 5-2. Some of the results are shown in Table 10.
- H553-IgG1 (SEQ ID NO: 46) as the heavy chain
- H553 / L118 having L118-k0MT as the light chain
- H496-IgG1 SEQ ID NO: 57
- H496 / L118 having L118-k0MT as the light chain
- H496 / L118 having L118-k0MT as the light chain
- H553 / L118 has two types of amino acid modifications Y55H and R57P introduced into the heavy chain of H89 / L118.
- H496 / L118 in which only R57P is introduced into the heavy chain of H89 / L118, has enhanced human IL-8 binding affinity at neutral pH compared to H89 / L118, but at acidic pH Human IL-8 binding affinity is almost unchanged. That is, the R57P modification introduced into H89 / L118 is a modification that enhances only the binding affinity at neutral pH without changing the human IL-8 binding affinity at acidic pH.
- H553 / L118 in which Y55H modification is introduced into the heavy chain of H496 / L118 maintains or slightly enhances the binding affinity at neutral pH compared to H89 / L118, while at acidic pH.
- the binding affinity was reduced.
- combining two types of amino acid modifications Y55H and R57P into H89 / L118 has the property that the binding affinity at acidic pH is reduced while maintaining or slightly increasing the binding affinity at neutral pH. It was possible to further enhance.
- H553 / L118 As a result, there was no significant difference between H553 / L118 and H89 / L118 in the group administered 2 mg / kg antibody, but in the group administered 8 mg / kg antibody. It was confirmed that H553 / L118 accelerated the disappearance of human IL-8 about 2.5 times compared with H89 / L118. From another point of view, H553 / L118 does not show a difference in the rate of elimination of human IL-8 in the comparison between 2 mg / kg and 8 mg / kg, and the antibody dose as seen in H89 / L118. There was no decrease in the rate of disappearance of the antigen when increased. Although not particularly limited, it is possible to consider the following as one of the reasons why such a result was obtained.
- H553 / L118 showed the same human IL-8 elimination rate when administered with 2 mg / kg antibody and when administered with 8 mg / kg antibody. This is because H553 / L118 has a sufficiently weak IL-8 binding at acidic pH, so that even in the condition of 8 mg / kg, the ratio of free IL-8 in the endosome is close to 100%. It shows that. In other words, H89 / L118 can achieve the maximum human IL-8 elimination effect at a dose of about 2 mg / kg, but the effect diminishes at a high dose of about 8 mg / kg. This suggests the possibility of On the other hand, H553 / L118 can achieve the maximum elimination effect of human IL-8 even at a high dose of about 8 mg / kg.
- H553 / L118 is an antibody capable of significantly accelerating the disappearance of human IL-8 in mice more significantly than H89 / L118.
- this antibody in order for this antibody to maintain the inhibitory effect of human IL-8 for a long period of time in vivo, during the period when the administered antibody is present in vivo (for example, in plasma) It is also important that the sum activity is kept stable (stability in IL-8 neutralizing activity of the antibody). Therefore, the stability of these antibodies in mouse plasma was evaluated by the following method. Mouse plasma was collected from blood of C57BL / 6J (Charles river) by methods known to those skilled in the art.
- CXCR1 and CXCR2 are known as receptors for human IL-8.
- PathHunter (r) CHO-K1 CXCR2 ⁇ -Arrestin Cell Line (DiscoveRx, Cat. # 93-0202C2) expresses human CXCR2 and exhibits chemiluminescence when a signal from human IL-8 is transmitted An artificially created cell line.
- DiscoveRx Cat. # 93-0202C2
- the degree of chemiluminescence when using the antibody after storage is increased compared to that before storage. Therefore, it was verified whether the antibody stored in mouse plasma maintained neutralizing activity using the above cell line.
- the cell line was suspended in AssayComplete (tm) Cell Plating 0 Reagent and seeded at 5000 cells / well on a 384 well plate.
- AssayComplete tm
- a test for determining the added concentration of human IL-8 was performed as follows. A serial dilution of the human IL-8 solution was added to the cell culture so that the final human IL-8 concentration included 45 nM (400 ng / mL) to 0.098 nM (0.1 ng / mL).
- a detection reagent was added according to the product protocol, and the amount of relative chemiluminescence was detected using a chemiluminescence detector.
- the reactivity of the cells to human IL-8 was confirmed, and an appropriate human IL-8 concentration was set to confirm the neutralizing activity of the anti-human IL-8 antibody.
- the human IL-8 concentration was 2 nM.
- the neutralization activity of the antibody contained therein was evaluated using mouse plasma supplemented with the aforementioned anti-human IL-8 antibody.
- Mouse plasma containing human IL-8 at the concentration determined above and the aforementioned anti-human IL-8 antibody was added to the cell culture medium.
- the amount of mouse plasma to be added was determined so as to be included stepwise in the range of 2 ⁇ g / mL (13.3 nM) to 0.016 ⁇ g / mL (0.1 nM) as the anti-human IL-8 antibody concentration.
- a detection reagent was added according to the product protocol, and the amount of relative chemiluminescence was detected using a chemiluminescence detector.
- the relative chemiluminescence average of wells not added with human IL-8 and antibody is 0%
- the average of relative chemiluminescence of wells added with only human IL-8 and no antibody is 100%
- the relative value of the relative chemiluminescence amount at each antibody concentration was calculated.
- the results of the human IL-8 inhibition assay using human CXCR2-expressing cells are shown in FIG. 18-1 for Initial (no storage treatment in mouse plasma), and in FIG. 18-2 for the sample stored at 40 ° C. for 1 week.
- the results of samples stored at 40 ° C. for 2 weeks are shown in FIG.
- Hr9 and H89 / L118 showed no difference in human IL-8 neutralizing activity before and after storage in mouse plasma.
- H553 / L118 showed a decrease in human IL-8 neutralizing activity after 2 weeks of storage. From this, H553 / L118 has a tendency to decrease human IL-8 neutralization activity in mouse plasma compared to Hr9 and H89 / L118, and has an unstable property in terms of IL-8 neutralization activity. It was shown to be an antibody.
- Example 9 Production of antibody with reduced immunogenicity prediction score by in silico system (9-1) Immunogenicity prediction scores of various IL-8 binding antibodies
- ADA anti-pharmaceutical antibodies
- the production of anti-pharmaceutical antibodies (ADA) affects the effects and pharmacokinetics of therapeutic antibodies, and sometimes causes serious side effects Thus, the usefulness and drug efficacy of therapeutic antibodies in the clinic can be limited by ADA production.
- the immunogenicity of therapeutic antibodies is known to be influenced by many factors, but many important effects of effector T cell epitopes possessed by therapeutic antibodies have been reported.
- Epibase Longza
- iTope / TCED Antitope
- EpiMatrix EpiMatrix
- EpiMatrix mechanically designed peptide fragment sequences by dividing the amino acid sequence of the protein whose immunogenicity is to be predicted every 9 amino acids, and against them, eight major MHC Class II alleles (DRB1 * 0101, DRB1 * 0301, DRB1 * 0401, DRB1 * 0701, DRB1 * 0801, DRB1 * 1101, DRB1 * 1301, DRB1 * 1501) and the ability to predict the immunogenicity of the target protein. (Clin Immunol. 2009 May; 131 (2): 189-201.) The immunogenicity scores of the heavy and light chains of each IL-8 antibody calculated as described above are shown in the column “EpiMatrix Score” in Table 12.
- Tregitope is a peptide fragment sequence that is mostly contained in naturally occurring antibody sequences, and is a sequence that is thought to suppress immunogenicity by activating suppressor T cells (Treg). is there. Moreover, about these scores, what totaled the score of the heavy chain and the light chain is shown in the total column.
- the immunogenicity scores of H89 / L118, H496 / L118, and H553 / L118 are known humanized anti-human IL-8 antibodies regardless of whether “EpiMatrix Score” or “tReg Adjusted Epx Score” is seen. It was lower than hWS-4.
- EpiMatrix can also compare the predicted ADA frequency of the whole antibody molecule with the actual ADA frequency of various commercially available antibodies, taking into account the heavy and light chain scores. The result of performing such an analysis is shown in FIG. In FIG.
- hWS-4 is “WS4”
- Hr9 is “HR9”
- H89 / L118 is “H89L118”
- H496 / L118 is “H496L118”
- H553 / L118 is “H553L118”.
- FIG. 19 the incidence of ADA in humans with various commercially available antibodies is 45% for Campath (Alemtuzumab), 27% for Rituxan (Rituximab), and 14% for Zenapax (Daclizumab). It is known.
- H496v1, H496v2, and H496v3, including single modifications all had an immunogenicity score lower than that of H496. Furthermore, in H1004 introduced by combining three types of modifications, a significant improvement in immunogenicity score was achieved.
- L395 has also been found as a suitable light chain to be combined with H1004. Therefore, in the calculation of the immunogenicity score, a combination of both L118 and L395 was calculated. As shown in Table 13, H1004 / L118 and H1004 / L395 also showed very low immunogenicity scores as the immunogenicity score when combining heavy and light chains.
- the ADA occurrence frequency was predicted in the same manner as in Example 9-1.
- H496v1 / L118 is represented as “V1”, H496v2 / L118 as “V2”, H496v3 / L118 as “V3”, H1004 / L118 as “H1004L118”, and H1004 / L395 as “H1004L395”.
- H1004 / L395 is an antibody having H1004-IgG1m (SEQ ID NO: 47) as a heavy chain and L395-k0MT (SEQ ID NO: 38) as a light chain was made.
- the binding affinity of H1004 / L395 to human IL-8 was measured using Biacore T200 (GE Healthcare) as follows. The following two types of running buffers were used, and measurement was performed by setting the respective temperatures.
- Example 10 Preparation and evaluation of pH-dependent IL-8 binding antibody H1009 / L395 (10-1) Preparation of various pH-dependent IL-8 binding antibodies H1004 / L395 having pH-dependent IL-8 binding ability and reduced immunogenicity score based on the study shown in Example 9 was acquired. Next, intensive studies were conducted with the aim of creating a modified body that satisfies both these desirable properties and stability in mouse plasma. Based on H1004 / L395, various modifications were introduced to produce the following modified antibodies.
- Biacore T200 GE Healthcare
- the amount of human IL-8 bound to the modified antibody was analyzed. Measurement was performed at 40 ° C. using 0.05% tween 20, 40 mM ACES, 150 mM NaCl, pH 7.4 as a running buffer.
- An appropriate amount of protein A / G (PIERCE) was immobilized on the sensor chip CM4 (GE Healthcare) by the amine coupling method, and the target antibody was captured there.
- H1009 / L395 an antibody having H1009-IgG1m (SEQ ID NO: 48) as the heavy chain and L395-k0MT as the light chain.
- H1009 / L395 has a slightly enhanced human IL-8 binding affinity at neutral pH compared to H89 / L118, but a reduced binding affinity at acidic pH. The pH dependence was further strengthened.
- H1009 / L395 was slightly improved over H89 / L118 in terms of stability in IL-8 binding when exposed to severe conditions of 50 ° C. in PBS. From these facts, H1009 / L395 was selected as an antibody that has a pH-dependent IL-8 binding ability and can also stably maintain neutralization activity in mouse plasma.
- Example 10-2 Stability evaluation of H1009 / L395
- H1009 / L395-F1886s whose details are described later in Example 15 was used.
- This antibody has the same variable region as H1009 / L395, and the constant region is modified to enhance FcRn binding under acidic pH conditions and to reduce binding to Fc ⁇ R compared to natural human IgG1. Having a constant region.
- H1009 / L395 to human IL-8 and the neutralizing activity of IL-8 are carried by the variable region of this antibody, especially the region centered on HVR, and the alteration introduced into the constant region affects it. It is thought that there is no.
- the stability evaluation in mouse plasma was performed as follows. 150 ⁇ L of 200 mM phosphate buffer (pH 6.7) was added to 585 ⁇ L of mouse plasma. Further, sodium azide was added as a preservative to a final concentration of 0.1%. Each antibody (Hr9, H89 / L118, H1009 / L395-F1886s) was added to the mouse plasma so as to have a final concentration of 0.4 mg / mL.
- H1009 / L395 has a human IL-8 elimination rate of 2 mg / kg in mice at the same level as H553 / L118, and can achieve free IL-8 in endosomes of nearly 100%. It was shown that It was also shown that the clearance (CL) value quantitatively representing the rate of disappearance of human IL-8 from mouse plasma was about 30 times higher than that of H998 / L63. Although there is no particular limitation, the effect of increasing the disappearance rate of human IL-8 can be interpreted as follows. In general, in the living body in which the antigen concentration is kept almost constant, the production rate and disappearance rate of the antigen are also kept almost constant.
- the disappearance rate of the antigen can be changed by forming a complex with the antibody.
- the rate of disappearance of the antigen is larger than the rate of disappearance of the antibody, and in such a case, the rate of disappearance of the antigen complexed with the antibody is decreased.
- the rate of disappearance of the antigen decreases, the concentration of the antigen in plasma increases, and the degree of increase can be defined by the ratio of the rate of disappearance when the antigen is alone and the rate of disappearance when the complex is formed.
- the antigen concentration in the plasma of the living body to which the antibody is administered is about 10 times that before the antibody administration.
- clearance (CL) can also be used as these disappearance rates. That is, it can be considered that the increase in antigen concentration (antigen accumulation) that occurs after antibody administration to a living body is defined by the antigen CL in each state before and after antibody administration.
- CL clearance
- H1009 / L395 can block the biological activity of plasma IL-8 with an extremely small amount of antibody, which is about 1/30 of that of H998 / L63.
- H1009 / L395 and H998 / L63 are administered to humans at the same dose, H1009 / L395 is stronger and blocks IL-8 biological activity for a longer period of time. Is possible.
- H1009 / L395 can maintain its human IL-8 neutralizing activity over a long period of time. It was shown that H1009 / L395 including these remarkable properties is an antibody having an excellent effect from the viewpoint of neutralizing IL-8 in vivo.
- Example 11 Evaluation of Extracellular Matrix Binding Using pH-Dependent IL-8 Binding Antibody H1009 / L395 As shown in Example 10, the human IL-8 elimination effect, which is 30 times better than H1009 / L395, It was a surprising effect. It is known that the rate of antigen disappearance upon administration of a pH-dependent antigen-binding antibody depends on the rate at which the antibody-antigen complex is taken into the cell. That is, if the rate of incorporation into cells increases when a complex with an antigen is formed compared to when the complex is not formed, it is possible to enhance the antigen elimination effect of the pH-dependent antibody.
- Methods for enhancing the rate of uptake of antibodies into cells include imparting FcRn binding ability to antibodies under neutral pH conditions (WO 2011/122011) and enhancing antibody binding ability to Fc ⁇ R (WO 2013) / 047752), a method using promotion of formation of a complex containing a multivalent antibody and a multivalent antigen (WO 2013/081143), and the like are known. However, the above technique is not used in the constant region of H1009 / L395.
- IL-8 is known to form a homo dimer, but since H1009 / L395 recognizes the homo dimer formation surface of human IL-8, human IL-8 bound to H1009 / L395 is a monomer It has become clear that Therefore, these antibodies do not form a multivalent complex. That is, although the above-mentioned technique is not used for H1009 / L395, H1009 / L395 showed 30 times as much human IL-8 elimination effect.
- H1009 / L395 showed 30 times as much human IL-8 elimination effect.
- Human IL-8 is a protein having a high isoelectric point (pI), and the theoretical isoelectric point calculated by a known method is about 10. That is, under conditions of neutral pH, human IL-8 is a protein having a charge biased toward the positive charge side.
- the pH-dependent IL-8 binding antibody represented by H1009 / L395 is also a protein having a charge biased toward the positive charge side, and the theoretical isoelectric point of H1009 / L395 is about 9.
- the complex formed by binding H1009 / L395, which is originally a protein with a high isoelectric point and rich in positive charge, to human IL-8 having a high isoelectric point is more isoelectric than H1009 / L395 alone. The point goes up.
- Increasing the isoelectric point of an antibody causes non-specific uptake of the antibody-antigen complex into the cell. It is also possible to think that it increases. It is possible that a complex of anti-IL-8 antibody and human IL-8 having a high isoelectric point has a higher isoelectric point than anti-IL-8 antibody alone and is likely to be taken up into cells. it was thought. Further, as described above, the binding property to the extracellular matrix is also one of the factors that may influence the intracellular uptake. Therefore, it was verified whether the binding property of the antibody alone to the extracellular matrix is different from the binding property of the complex of human IL-8 and the antibody.
- the antibody used for evaluation was prepared as follows. As a sample added with antibody alone, each antibody was diluted to 9 ⁇ g / mL with Buffer-1 as shown below, and further diluted with Buffer-2 to a final concentration of 3 ⁇ g / mL. .
- Buffer-1 20 mM ACES buffer containing 150 mM NaCl, 0.05% Tween 20, 0.01% NaN 3 , pH 7.4
- Buffer-2 150 mM NaCl, 0.05% Tween20, 0.1% BSA, 20 mM ACES buffer containing 0.01% NaN 3, pH7.4
- human IL-8 having a molar concentration 10 times that of the antibody was added to the antibody sample, and then the antibody concentration was 9 ⁇ g / ml using Buffer-1. After each dilution to mL, each was further diluted with Buffer-2 so that the final antibody concentration was 3 ⁇ g / mL.
- human IL-8 is about 0.6 ⁇ g / mL. Shake for 1 hour at room temperature to form the complex. Next, the antibody alone or the complex solution was added to the plate from which the blocking solution had been removed, and shaken at room temperature for 1 hour. Thereafter, the antibody alone or complex solution was removed, Buffer-1 containing 0.25% Glutaraldehyde was added, and the mixture was allowed to stand for 10 minutes, and then washed with DPBS containing 0.05% Tween20 (manufactured by Wako Pure Chemical Industries, Ltd.).
- the antibody for ECL detection was prepared by Sulfo-Tag using Goat anti-human IgG (gamma) (Zymed Laboratories) using Sulfo-Tag NHS Ester (MSD).
- the antibody for ECL detection was diluted with Buffer-2 to 1 ⁇ g / mL, added to the plate, and shaken at room temperature for 1 hour in the dark. ECL detection antibody was removed, MSD Read Buffer T (4x) (MSD) diluted twice with ultrapure water was added, and luminescence was measured using SECTOR Imager 2400 (MSD) did. The results are shown in FIG.
- Example 12 Mouse PK test using FcRn non-binding antibody
- a pH-dependent IL-8 binding antibody forms a complex with human IL-8, and the complex enters the cell. Whether or not uptake increased was confirmed by the following method.
- a mutant having a variable region of H1009 / L395 and an Fc region lacking binding affinity to various Fc receptors was prepared. Specifically, as a modification that deletes the ability to bind to human FcRn under acidic pH conditions, EU numbering 253rd isoleucine is used for alanine and 254th serine is used for asparagine against H1009-IgG1, which is a heavy chain. Substituted with acid.
- the collected blood was immediately centrifuged at 4 ° C. and 15,000 rpm for 15 minutes to obtain plasma.
- the separated plasma was stored in a freezer set to ⁇ 20 ° C. or lower until measurement was performed.
- Anti-human IL-8 antibody concentration in mouse plasma was measured by electrochemiluminescence method.
- the Anti-Human Kappa Light Chain Goat IgG Biotin (Meso Scale Discovery) was blocked on a Streptavidin Gold Multi-ARRAY plate (Meso Scale Discovery) that was blocked overnight at room temperature using a PBS-Tween solution containing 5% BSA (w / v). IBL) was reacted at room temperature for 1 hour to prepare an anti-human antibody-immobilized plate.
- Samples for calibration curves containing anti-human IL-8 antibodies with plasma concentrations of 3.20, 1.60, 0.800, 0.400, 0.200, 0.100, 0.0500 ⁇ g / mL and samples for mouse plasma measurement diluted 100 times or more were prepared. . Each sample was mixed with human IL-8, dispensed into each well of an anti-human antibody-immobilized plate at 50 ⁇ L, and stirred at room temperature for 1 hour. The final concentration of human IL-8 was adjusted to 333 ng / mL. Thereafter, an anti-human IL-8 antibody (in-house preparation) having a mouse IgG constant region was added to the plate, and allowed to react at room temperature for 1 hour.
- Anti-Mouse IgG (BECKMAN COULTER) rutheniumized with SULFO-TAG NHS Ester (Meso Scale Discovery) was added to the plate and allowed to react for 1 hour, and then Read Buffer T ( ⁇ 1) (Meso Scale Discovery) was separated. Note and immediately measured with SECTOR Imager 2400 (Meso Scale Discovery).
- the anti-human IL-8 antibody concentration was calculated from the response of the calibration curve using analysis software SOFTmax PRO (Molecular Devices). The resulting antibody concentration in mouse plasma is shown in FIG. 24, and antibody clearance under each condition is shown in Table 17.
- the uptake rate of the complex of H1009 / L395 and IL-8 into the cell increases more than the actual uptake rate of H1009 / L395 into the cell in vivo.
- the effect is not limited to a value such as 2.2 times.
- the following interpretation can be made from the knowledge obtained so far.
- the anti-IL-8 antibody when the anti-IL-8 antibody is pH-dependent, the anti-IL-8 antibody once taken into the cell returns to the outside of the cell again after dissociating the IL-8 molecule in the cell, and another IL- Since it becomes possible to bind to 8 molecules, it is possible to consider that the increased uptake into cells during complex formation has the additional effect of removing IL-8 more strongly. That is, selecting an anti-IL-8 antibody with increased binding to the extracellular matrix or an anti-IL-8 antibody with increased intracellular uptake is another aspect of the present disclosure.
- Example 13 Prediction of immunogenicity of pH-dependent IL-8 binding antibody H1009 / L395 by in silico system
- immunogenicity score and ADA frequency of H1009 / L395 were evaluated in the same manner as in Example 9-1. Predicted. The results are shown in Table 18 and FIG. In FIG. 25, H1009 / L395 is represented as “H1009L395”.
- H1009 / L395 has a low immunogenicity score as much as H1004 / L395. Further, from the results of FIG. 25, the ADA occurrence frequency predicted in H1009 / L395 was 0%, which was also the same as that in H1004 / L395. From the above, the immunogenicity predicted in H1009 / L395 was significantly reduced as compared with hWS-4 which is a known anti-human IL-8 antibody. From this, it is considered that H1009 / L395 has extremely low immunogenicity in humans and can safely maintain anti-IL-8 neutralizing activity over a long period of time.
- Example 14 Cynomolgus monkey PK test using H89 / L118 variant with enhanced FcRn binding ability under acidic pH conditions
- the pH-dependent IL-8 binding antibody H1009 / L395 In the case of having a natural IgG1 constant region, the antibody has very excellent properties. However, it can also be used as an antibody containing an amino acid substitution in the constant region, for example, an antibody containing an Fc region with enhanced FcRn binding at acidic pH exemplified in Example 5. Thus, it was confirmed using H89 / L118 that the Fc region with enhanced FcRn binding at acidic pH also functions in pH-dependent IL-8 binding antibodies.
- H89-IgG1m (SEQ ID NO: 50) as the heavy chain, H89 / L118-IgG1 having L118-K0MT as the light chain, H89-F1168m (SEQ ID NO: 51) as the heavy chain, H89 / L118-F1168m with L118-K0MT as the light chain, H89-F1847m (SEQ ID NO: 52) as the heavy chain, H89 / L118-F1847m with L118-K0MT as the light chain, H89-F1848m as heavy chain (SEQ ID NO: 53), H89 / L118-F1848m with L118-K0MT as light chain, H89-F1886m (SEQ ID NO: 54) as the heavy chain, H89 / L118-F1886m with L118-K0MT as the light chain, H89-F1889m (SEQ ID NO: 55) as the heavy chain, H89 / L118-F1889m with L118-K0MT as the light
- the concentration of anti-human IL-8 antibody in cynomolgus monkey plasma was measured by electrochemiluminescence method.
- Anti-hKappa Capture Ab (Antibody Solutions) was dispensed into MULTI-ARRAY 96-well plates (Meso Scale Discovery) and stirred at room temperature for 1 hour. Thereafter, blocking with a PBS-Tween solution containing 5% BSA (w / v) for 2 hours at room temperature produced an anti-human antibody-immobilized plate.
- Samples for calibration curves containing anti-human IL-8 antibodies with plasma concentrations of 40.0, 13.3, 4.44, 1.48, 0.494, 0.165, 0.0549 ⁇ g / mL and cynomolgus monkey plasma samples diluted more than 500 times were prepared, After 50 ⁇ L was dispensed into each well of the anti-human antibody-immobilized plate, the mixture was stirred at room temperature for 1 hour. Thereafter, Anti-hKappa Reporter Ab, Biotin conjugate (Antibody Solutions) was added to the plate and allowed to react at room temperature for 1 hour.
- Fc region with reduced Fc ⁇ R binding ability Natural human IgG1 binds to Fc ⁇ receptors (hereinafter referred to as Fc ⁇ R) on various immune cells, and It is known to show effector functions such as ADCC and ADCP for cells that do.
- Fc ⁇ R Fc ⁇ receptors
- IL-8 is a soluble cytokine
- anti-IL-8 antibodies used as pharmaceuticals neutralize their functions mainly at sites where IL-8 is present excessively, and have a pharmacological action. It is expected to show.
- the site where IL-8 is excessively present is not particularly limited, but for example, an inflammatory site can be assumed. In general, it is known that various immune system cells are gathered and activated in such an inflammatory site.
- the anti-IL-8 antibody administered to the living body preferably has a low binding ability to Fc ⁇ R from the viewpoint of safety.
- H1009-F1886m the EU numbering 235th L was replaced with R, the 236th G was replaced with R, and the region of EU numbering 327th to 331st was defined as a human natural IgG4 sequence, -F1974m (SEQ ID NO: 36) was prepared.
- H1009 / L395-F1886s and H1009 / L395-F1974m were prepared as antibodies having these heavy chains and L395-k0MT as the light chain.
- Soluble Fc ⁇ RIa and Fc ⁇ RIIIa of human and cynomolgus monkeys, respectively, were prepared as His-tagged molecular forms by methods known to those skilled in the art.
- An appropriate amount of rProtein L (BioVision) was immobilized on the sensor chip CM4 (GE Healthcare) by the amine coupling method, and the antibody was captured thereon.
- soluble Fc ⁇ RIa or Fc ⁇ RIIIa and a running buffer (as a reference solution) were injected to interact with the antibody captured on the sensor chip.
- HBS-EP + GE Healthcare
- HBS-EP + was also used for dilution of soluble Fc ⁇ RIa or Fc ⁇ RIIIa.
- H1009 / L395-F1886m was shown to bind to any Fc ⁇ R, whereas the newly produced H1009 / L395-F1886s and H1009 / L395-F1974m were able to bind to any Fc ⁇ R. It was confirmed that they do not combine.
- human IL-8 (10 ⁇ g / mL) and anti-human IL-8 antibody (200 ⁇ g / mL, 500 ⁇ g / mL or 1000 ⁇ g / mL) was administered once to the tail vein at 10 mL / kg. At this time, since a sufficient amount of anti-human IL-8 antibody exists in excess of human IL-8, it is considered that almost all human IL-8 is bound to the antibody.
- Blood was collected 5 minutes, 2 hours, 4 hours, 7 hours, 1 day, 2 days, 3 days, 7 days, 14 days, 21 days, and 28 days after administration.
- the collected blood was immediately centrifuged at 4 ° C. and 15,000 rpm for 15 minutes to obtain plasma.
- the separated plasma was stored in a freezer set to ⁇ 20 ° C. or lower until measurement was performed.
- Measurement of human IL-8 concentration in mouse plasma was carried out in the same manner as in Example 7.
- the resulting plasma human IL-8 concentration data is shown in FIG. 28, and human IL-8 clearance (CL) values from mouse plasma are shown in Table 20.
- H1009 / L395 having the Fc region of natural IgG1 and H1009 / L395-F1886s having the modified Fc region may have equivalent human IL-8 elimination effects. Indicated.
- human IL-8 clearance There was no significant difference between 2 mg / kg and 10 mg / kg. This strongly suggests that the antibody containing the variable region of H1009 / L395 shows a sufficient IL-8 elimination effect even when administered at a high dose.
- Cynomolgus monkey PK test of Fc region variant Next, using H1009 / L395-F1886s and H1009 / L395-F1974m, the plasma retention of antibodies in cynomolgus monkeys was verified by the following method. The pharmacokinetics of anti-human IL-8 antibody after simultaneous administration of anti-human IL-8 antibody alone or human IL8 and anti-human IL-8 antibody to cynomolgus monkeys was evaluated. Single human vein of anti-human IL-8 antibody solution (2 mg / mL) or mixed solution of human IL-8 (100 ⁇ g / kg) and anti-human IL-8 antibody (2 mg / kg) at 1 mL / kg It was administered internally.
- Blood was collected at 5 minutes, 4 hours, 1 day, 2 days, 3 days, 7 days, 10 days, 14 days, 21 days, 28 days, 35 days, 42 days, 49 days, and 56 days after administration.
- the collected blood was immediately centrifuged at 4 ° C. and 15,000 rpm for 10 minutes to obtain plasma.
- the separated plasma was stored in a freezer set to ⁇ 60 ° C. or lower until measurement.
- the measurement of anti-human IL-8 antibody concentration in cynomolgus monkey plasma was carried out by the method described in Example 14.
- the resulting plasma anti-human IL-8 antibody concentration data is shown in FIG. 29, and the half-life (t1 / 2) and clearance (CL) values of anti-human IL-8 antibody from cynomolgus monkey plasma are shown.
- H1009 / L395-F1886s which has an Fc region with improved function, has significantly improved plasma retention. Indicated. Furthermore, even when H1009 / L395-F1886s was administered at the same time as human IL-8, the plasma concentration transition was the same as when administered with the antibody alone. Although there is no particular limitation, it is possible to consider as follows. As described above, it has been shown that the uptake of the complex of H1009 / L395 and human IL-8 into the cell is higher than the uptake of H1009 / L395 alone.
- H1009 / L395-F1886s may be sufficiently salvaged by FcRn and returned to plasma even if the intracellular uptake rate of the antibody is increased.
- another type of Fc region variant H1009 / L395-F1974m, also showed the same plasma retention as H1009 / L395-F1886s.
- these Fc region variants have been introduced as different modifications to reduce the ability to bind to various Fc ⁇ Rs, but they do not affect the plasma retention of the antibody itself. It is shown that.
- H1009 / L395 has a pH-dependent IL-8 binding ability and a property that a complex with IL-8 is rapidly taken up into cells.
- This is the first antibody that can significantly increase the disappearance rate of human IL-8 in vivo.
- the binding affinity to IL-8 under neutral pH conditions is also increased compared to the known hWS-4 antibody, and it is more potent in neutralizing human IL-8 under neutral pH conditions such as in plasma. It is possible to sum.
- it is an antibody that has excellent stability under plasma conditions and does not decrease IL-8 neutralizing activity even after administration in vivo.
- H1009 / L395 produced based on Hr9 whose production amount is significantly improved compared to hWS-4, is an antibody suitable for production from the viewpoint of production amount. Further, in silico immunogenicity prediction shows a very low score for the immunogenicity, which is much lower than that of known hWS-4 antibodies and some of the existing commercially available antibodies. That is, H1009 / L395 is expected to be an antibody that is difficult to produce ADA in humans and can be used safely over a long period of time. From these facts, H1009 / L395 is an antibody that has been improved from various viewpoints as compared with known anti-human IL-8 antibodies, and is very useful as a pharmaceutical product.
- H1009 / L395 having the Fc region of natural IgG is sufficiently useful as described above, but a variant of H1009 / L395 containing an Fc region with improved function is also an antibody with enhanced utility Can be used as appropriate. Specifically, FcRn binding under acidic pH conditions is enhanced, plasma retention is improved, and the effect can be maintained for a longer period.
- a variant containing an Fc region into which a modification that reduces the ability to bind to Fc ⁇ R is introduced avoids the occurrence of unintended immune system cell activation or cytotoxic activity in the administered organism, It can be used as a highly safe antibody drug.
- F1886s or F1974m implemented in this specification, but it is not limited to these Fc, and other Fc may be used as long as they have similar functions.
- Antibody pharmaceuticals containing modified Fc regions are also used as one embodiment of the present disclosure.
- the antibodies in the present disclosure including H1009 / L395-F1886s, H1009 / L395-F1974m, etc. maintain a state capable of strongly inhibiting the biological activity of human IL-8 over a long period of time and safely. Is possible. This has achieved a level that cannot be achieved with existing anti-IL-8 antibodies, and is expected to be used as a highly complete anti-IL-8 antibody drug.
- Example 16 Measurement of IL-8 concentration in cyst fluid of endometriosis patients
- concentration of IL-8 in cyst fluid of endometriosis patients was determined. It was measured. Samples were collected from postoperative tissue cyst fluid from patients with endometriosis who were surgically treated at Jichi Medical University. After anonymization, Chugai Pharmaceutical performed secondary anonymization. IL-8 concentration in the cyst fluid was measured by electrochemiluminescence.
- the anti-human IL-8 antibody (Hycult Biotech) was labeled with biotin, and then MSD GOLD 96-Well Streptavidin SECTOR Plate (2% blocked with PBS-Tween solution containing 5% BSA (w / v)) ( An anti-human IL-8 antibody-immobilized plate was prepared by dispensing to Meso Scale Discovery and stirring for 1 hour at room temperature in the dark.
- Calibration curve samples containing human IL-8 (in-house adjustment with reference to NP_000575.1) with 900, 300, 100, 33.3, 11.1, 3.70, 1.23 pg / mL in cyst fluid and human cysts diluted more than 5 times
- a liquid measurement sample was prepared and dispensed at 25 ⁇ L into each well of an anti-human IL-8 antibody-immobilized plate, and then stirred at room temperature in the dark for 1 hour.
- Example 17 Preparation and pathological evaluation of endometriosis surgically attracted monkey model To evaluate the efficacy of anti-IL-8 antibody in endometriosis, an endometriosis surgical attraction model was prepared and evaluated. went.
- the pathological model was prepared as follows. An 8-14 year-old female cynomolgus monkey (obtained from the Primate Medical Science Research Center, National Institute of Biomedical Innovation, Health and Nutrition) with a regular estrous cycle is laparotomized under anesthesia during the luteal phase, and the uterus The body was incised into a V shape and excised.
- the uterine smooth muscle layer is left and trimmed to 5 mm to 10 mm square, with one end on the right abdominal wall and two on the left abdominal wall so that the endometrium is in contact with the peritoneum (head side, tail) Side) was sewn with an absorbent thread to obtain a transplanted part.
- the excised uterus was sutured with an absorbent thread.
- the remaining intimal tissue was minced and suspended in 5 mL of 2 ng / mL Hepatocyte Growth Factor solution (manufactured by R & D systems), seeded and transplanted intraperitoneally, and closed.
- Anesthesia was performed using ketamine xylazine balanced anesthesia (mixed with a ratio of 2: 1 as a guide), but was maintained with isoflurane depending on the duration of anesthesia. During the operation, the body temperature was maintained by heating pad, and the condition was controlled by electrocardiogram. At the time of awakening after surgery, the antagonist antisedan (with the same amount as xylazine) and the antibiotic cephalazine are administered, and the analgesic zaruban is administered for 2 days after the operation. Confirmed that there is no. Anesthesia and postoperative management were similarly performed during laparoscopic observation.
- the size of the lesion was measured using a graduated bar or graduated forceps. Measure the size (length, width, height) of the nodular lesion formed by the suture of the endometrial tissue, and calculate the volume from length (mm) x width (mm) x height (mm) did.
- the adhesion of the adhesion is determined according to the modified r-AFS score, which is a modified r-AFS score for monkeys. Location, extent, depth and lesion size were assessed during laparoscopic observation.
- the Modified r-AFS score was modified as follows for monkeys as shown in FIG. 1.
- the frequency of adhesions to the bladder was higher than that of humans, so an item for adhesion to the bladder uterus was added.
- the evaluation method for adhesion to the bladder was scoring similar to the adhesion of Douglas fossa. 2. Since monkeys are smaller in body and organ size than humans, the lesion size criteria were changed to ⁇ 3 mm, 3-10 mm,> 10 mm for scoring. Later, the recorded laparoscopic video was confirmed using a video system, and the laparoscopic evaluation was finalized. Based on the results of laparoscopic observation after 4 months of attraction, individuals satisfying the criteria were selected and divided into groups. The recruitment criteria are as follows. 1. 1. Laparoscopic observation after 4 months of attraction can confirm engraftment of the intima.
- H1009 / L395-F1974m was intravenously injected at 10 mg / kg as the antibody administration group, and His Buffer (20 mM His-Asp, 150 mM Arg-Asp (pH 6.0)) was subcutaneously administered as the Vehicle group.
- His Buffer (20 mM His-Asp, 150 mM Arg-Asp (pH 6.0)) was subcutaneously administered as the Vehicle group.
- Laparoscopy was performed at 3 months and 6 months after the first administration every 3 months from the start of administration to evaluate the pathological condition of endometriosis.
- the membrane epithelium was highly formed and formed even in the abdominal wall muscle layer.
- the periphery of the intimal epithelium is composed of multi-layered stroma cells and interstitium rich in collagen fibers, and a structural morphology similar to that of human endometriotic tissue structure was observed. From these results, it was confirmed that this model is useful as an endometriosis model (12 th World Congress on Endometriosis, 2014, P-221). Similarly, a method using baboons has also been reported as a surgical attraction model for endometriosis (Fertil Steril. 2013; 99 (3): 783-789, Fertil Steril. 2013; 100 (4): 1144). -50).
- the monkey IL-8 concentration was calculated from the response of the calibration curve using analysis software SOFT-Max-PRO (Molecular Devices). As a result, as shown in Table 23, in the cyst fluid of the endometriosis surgically attracted monkey model, it was confirmed that IL-8 had a very high concentration similarly to the cyst fluid of the human endometriosis patient. It was. This finding supports that this monkey model is highly extrapolated to human endometriosis. Furthermore, when the correlation between the IL-8 concentration in each cystic fluid and the adhesionadr-AFS score was examined, it was interesting to find that the IL-8 concentration in the cystic fluid and the adhesion r as shown in FIG. -There was a correlated trend in AFS score. In addition, when the IL-8 concentration in a plurality of cyst fluids could be measured in each individual, the highest value was used as a representative value. This result indicated the involvement of IL-8 in the adhesion of endometriosis.
- the drug efficacy was examined by laparoscopic observation 6 months after the start of administration.
- individuals whose menstrual bleeding was confirmed less than 3 times during the 6-month administration period were excluded from the analysis.
- one of the antibody H1009 / L395-F1974m-administered group # 117 (10 mg / kg group) corresponded and was excluded from the analysis.
- anti-antibodies were detected in the group administered with antibody H1009 / L395-F1974m according to a method known to those skilled in the art, anti-antibodies were detected in 2 out of 6 animals, and a decrease in blood trough API concentration was observed. It was. The following analysis was performed including individuals who were anti-antibody positive. First, the volume of each nodular lesion formed by suturing the endometrial tissue was calculated, and classified according to the change and size over time as shown in Table 24 below. When evaluation over time was not possible, such as inability to evaluate by adhesion, the relevant part was not included in the analysis.
- the frequency of type A and type B indicating proliferation was as high as 54% (7/13 lesions), and type D indicating the decrease was 15% (2/13 lesions).
- the frequency of type D which shows a decrease, was high at 58% (7/12 lesions). Nodularity caused by antibody H1009 / L395-F1974m administration The reduction effect of the lesion was shown.
- the relative volume of nodular lesions in the Vehicle group was 141% (67-331) (median (minimum-maximum)).
- the antibody H1009 / L395-F1974m administration group was 49% (15-156).
- the change of modified r-AFS score was calculated as follows to determine how much each individual changed before and after antibody administration, and the results are shown in FIG. 35-1.
- FIG. 35-1 shows change of modified r-AFS score evaluated based on images taken during laparoscopic surgery.
- the efficacy evaluation was performed in the surgically induced monkey model, and in the antibody H89 / L118-F22 administration group, Atrophy of proliferating epithelium and stroma cells, and decrease of interstitium mainly composed of collagen fibers were confirmed.
- anti-IL-8 antibody is effective for reducing endometriotic lesion volume, improving adhesion, atrophic changes of epithelium and stroma cells, reducing infiltration of immune cells, and improving fibrosis. It was confirmed that the anti-IL-8 antibody is useful as a therapeutic agent for endometriosis. From this result, those skilled in the art will understand that an IL-8 signal inhibitor is useful as an agent for treating or preventing endometriosis.
- Example 18 Pharmacologic effect of anti-IL-8 antibody on adenomyosis 10
- 10 months after the final laparoscopic observation 6 months after the start of antibody administration It was fixed with a neutral buffered formalin solution and observed with H ⁇ E staining.
- endometrial tissue similar to adenomyosis was strongly observed in one case in the Vehicle group and one in the antibody administration group.
- the endometrial tissue observed in these two cynomolgus monkeys has endometrial gland and surrounding endometrial stroma in the myometrium, and the pathological pathology of adenomyosis observed in the human uterus.
- CXCR1 and CXCR2 are known as receptors for human IL-8.
- mouse anti-human CXCR1 monoclonal antibody R & D Systems, catalog number: 42705
- mouse anti-human CXCR2 monoclonal antibody Abcam, catalog number: 19.
- immunohistochemical staining was performed using a paraffin-embedded block of surgically removed human endometriosis tissue.
- FIG. 38 shows representative intraperitoneal photographs of the Vehicle group and the antibody H1009 / L395-F1974m administration group.
- Example 21 Isolation of neutrophils and evaluation of neutrophil migration by IL-8 IL-8 has neutrophil chemotaxis, and IL-8 produced in damaged tissues is vascular endothelial cells It is said that the migration of neutrophils adhering to the tissue to the tissue is promoted and inflammation accompanying neutrophil infiltration is caused. As shown in Table 27, infiltration of neutrophils, macrophages, and mononuclear cells was also confirmed at the endometriotic lesion site, and CXCR1 and CXCR2 were both positively expressed in these immune cells.
- IL-8 has a direct pharmacological action on endometriosis epithelial cells and pharmacology via invasive immune cells such as neutrophils and macrophages It was suggested that there was a possibility of action. Therefore, first, migration to IL-8 in neutrophils was evaluated. Neutrophils were collected from the peripheral blood of healthy individuals, and isolated and purified as follows. 1/5 volume of HetaSep (manufactured by STEMCELL Technologies) was added to heparinized whole blood, and the supernatant containing nucleated cells was collected according to the product protocol, and then ACK solution (manufactured by GIBCO) was added to hemolyze.
- HetaSep manufactured by STEMCELL Technologies
- the cells were suspended in 0.1% BSA / PBS and the number of cells was counted. After counting the number of cells, adjust the number of cells and use the EasySep (registered trademark) Human Neutrophil Enrichment Kit (manufactured by STEMCELL technologies) and neutrophils with EasySep (registered trademark) Magnet (manufactured by STEMCELL technologies) according to the product protocol.
- EasySep registered trademark
- Human Neutrophil Enrichment Kit manufactured by STEMCELL technologies
- neutrophils with EasySep registered trademark
- Magnet manufactured by STEMCELL technologies
- the isolated neutrophils were evaluated for migration using a migration plate of CytoSelect (registered trademark) 96-Well Cell Migration Assay (manufactured by Cell Biolabs, catalog number: CBA-104).
- CytoSelect registered trademark
- 96-Well Cell Migration Assay manufactured by Cell Biolabs, catalog number: CBA-104.
- 150 ⁇ L of RPMI-1640 medium manufactured by SIGMA
- 1% FBS or 1% FBS + IL-8 100 ng / mL
- the anti-IL-8 neutralizing antibody Anti-IL-8 antibody [807] (manufactured by abcam, catalog number: ab18672), Anti-IL-8 antibody [ 6217] (manufactured by abcam, catalog number: ab10768), IgG1 (manufactured by Sigma, catalog number: I5154) as a negative control, each 10 ⁇ g / mL, the compound is CXCR2 inhibitor SRT3109 (manufactured by Shanghai Haoyuan Chemexpress, catalog No .: HY-15462) was 10 ⁇ M, and PBS and DMSO were placed as solvent controls.
- FIG. 39 shows the result of calculating the relative value of the relative chemiluminescence amount in the wells to which IL-8 was not added and calculating the relative value of the relative chemiluminescence amount when each reagent was added.
- IL-8 promoted neutrophil migration and various anti-IL-8 neutralizing antibodies and CXCR2 inhibitors inhibited neutrophil migration.
- IL-8 signal inhibitors such as CXCR1 inhibitor and CXCR2 inhibitor known to be involved in IL-8 signal inhibition widely suppress neutrophil migration.
- the IL-8 signal inhibitor is likely to suppress infiltration similarly to neutrophils that infiltrate lesions with high concentrations of IL-8, including endometriosis.
- IL-8 signaling inhibitors can prevent endometriosis and inflammatory diseases where IL-8 is known at high concentrations.
- Endometriosis cells were obtained as follows. The surgically removed human endometriosis tissue is minced in DMEM / F-12, HEPES (GIBCO) medium containing 0.5 mg / mL collagenase and 0.1 mg / mL DNase1, and the cells are incubated at 37 ° C. It was collected. The cell suspension was passed through a 100 ⁇ m cell strainer and then hemolyzed with Lysing Buffer (BD Biosciences).
- the cells were seeded, and the passaged cells were cryopreserved in liquid nitrogen as endometriosis cells.
- DMEM / F-12, HEPES supplemented with 10% FBS, 2.5 ⁇ g / mL amphoterici B, 100 U / mL penicillin, 100 ⁇ g / mL streptomycin were used.
- cryopreserved cells were used after sleeping. Neutrophil culture supernatant was added to the endometriosis cells obtained by the above method, and the reactivity in endometriosis cells was examined.
- the neutrophil culture supernatant is obtained by isolating neutrophils, suspending them in RPMI-1640 medium containing 1% FBS, seeding them on a 6- well plate at 3.0 x 10 6 cells / well, and culturing them for 1 day. It was collected. Endometriosis cells were seeded at 10000 cells / well in a 96-well plate, and half of the medium was added to the recovered neutrophil culture supernatant. Similarly, to the control group (also referred to as “Ctrl” in this example), half of the medium was added in an RPMI-1640 medium containing 1% FBS.
- cDNA was synthesized using SuperScript (registered trademark) VILO (registered trademark) MasterMix (manufactured by Invitrogen), and the mRNA expression level of aromatase was compared by real-time PCR.
- Taqman probe Hs00903411_m1 was used as the aromatase probe
- Human GAPD (GAPDH) Endogenous Control (Applied Biosystems, catalog number: 4326317E) was used for GAPDH measurement.
- Aromatase is an estrogen synthase, and estrogen is known to promote the growth of endometriotic epithelial cells. Since aromatase expression in endometriosis cells was enhanced by addition of neutrophil culture supernatant, aromatase in endometriosis cells by infiltration of neutrophils into endometriotic lesions The expression level was increased, suggesting that the proliferation of endometriotic epithelial cells was increased.
- Example 23 Analysis of neutrophil-producing chemokines and cytokines
- chemokines, cytokines, and growth factors are produced in neutrophils when IL-8 is added or not added, and anti-IL-8 Analysis was performed at the time of antibody addition.
- the isolated neutrophils were suspended in RPMI-1640 medium containing 1% FBS, and seeded at 1.5 ⁇ 10 6 cells / well on a 6- well plate.
- IL-8 was added or not added thereto, or IL-8 and various antibodies were added thereto, and cultured for 1 day.
- H1009 / L395-F1974m, Anti-IL-8 antibody [807] (ab18672), and IgG1 were added to a final concentration of 10 ⁇ g / mL, and IL-8 was added to a final concentration of 100 ng / mL.
- IL-8 was added to a final concentration of 100 ng / mL.
- the cell culture medium was collected, and using a Cytokine Human Magnetic 30-Plex Panel for the Luminex (registered trademark) platform (manufactured by Thermo Fisher), according to the product protocol, various cytokines, chemokines and chemokines in the cell culture medium were collected. The concentration of growth factor was examined. As a result, as shown in FIG.
- MCP-1 monocyte chemoattractant protein 1
- CCL2 CCL2 and has the ability to chemotactic monocytes and is said to promote monocyte migration and infiltration into the inflamed area.
- H1009 / L395-F1974m in the nodular lesion of the surgically induced monkey model of endometriosis, macrophages or monocytes were treated with the antibody. It was suggested that infiltration was suppressed.
- MCP-1 with monocyte chemotaxis was produced by IL-8 stimulation of neutrophils, so that neutrophils infiltrate the endometriotic lesion by IL-8, Furthermore, by producing MCP-1, it was shown that monocytes and macrophages may further migrate and invade the lesion site. Furthermore, MCP-1 is known to act on fibroblasts and promote collagen production (J Biol Chem. 1996 Jul 26; 271 (30): 17779-84.) And promotes tissue fibrosis. Many things have been reported in vivo (J Immunol. 1994 Nov 15; 153 (10): 4733-41, Am J Physiol Lung Cell Mol Physiol.
- IL-8 infiltrates neutrophils into the endometriotic lesions, and neutrophils produce MCP-1.
- endometriosis The possibility of promoting fibrosis of the lesion was also shown. This result suggests that suppression of neutrophil infiltration by an IL-8 signal inhibitor improves fibrosis in the endometriotic lesion.
- IL-8 signal inhibitors were useful for fibrosis of endometriosis. Those skilled in the art will be able to generalize and expand and fully understand that IL-8 signaling inhibitors can treat or prevent fibrosis of endometriosis, as well as various fibrosis. .
- Example 24 Preparation and disease state evaluation of postoperative adhesion-inducing monkey model (24-1) Preparation of postoperative adhesion-inducing monkey model To create a postoperative adhesion-inducing monkey model, female cynomolgus monkeys 9-16 years old (obtained from Primate Medical Science Research Center, National Institute of Biomedical Innovation, Health and Nutrition) ), The following work was performed under anesthesia. Anesthesia was performed using ketamine xylazine balanced anesthesia (mixed at a ratio of 2: 1) and isoflurane. The midline of the abdominal wall was incised 5 cm to 6 cm, and the abdomen was opened.
- the uterine body was incised 1 cm and sutured with 4 needles, and then the entire uterus was wiped 5 times with gauze.
- a 1 cm incision was made in the right abdominal wall peritoneum 2 cm from the incised abdominal wall midline, and four stitches were continuously sutured with an absorbent thread.
- the sutured part was wiped with gauze five times.
- the left abdominal wall peritoneum was wiped with an incision, suture, and gauze.
- 10-11 stitches were stitched from the midline of the abdominal wall with continuous stitches, and all operations were completed within 1 hour.
- antisedan antagonist (with the same amount of xylazine as a guide) and cefradine antibiotic were administered.
- the analgesic zaruban was administered for 2 days after the operation, and it was confirmed by daily regular observation that there was no abnormality after the operation. Anesthesia and postoperative management were similarly performed during laparoscopic observation.
- the laparoscope was connected to a video system (made by KARL STORZ) and a monitor, recording was performed with the video system, and intraperitoneal observation was performed with the monitor.
- the size of the adhesions was measured using a graduated bar or graduated forceps.
- the presence, extent, and location of adhesions were assessed during laparoscopic observation. The results are shown in Table 29.
- adhesion was confirmed at all incision sites in two monkeys in the untreated group.
- the anti-IL-8 antibody administration group although adhesion was observed in individual No. 206, no adhesion was observed in the left and right abdominal wall peritoneum and uterine incision in individual No. 201. Indicated.
- anti-IL-8 antibody is useful as an adhesion preventing agent.
- the anti-IL-8 antibody can exert a higher effect of preventing adhesion formation by appropriately changing the administration time point.
- an IL-8 signal inhibitor is useful as an adhesion preventing agent based on this result.
- Example 25 Functional analysis of IL-8 in macrophages differentiated from monocytes and evaluation of drug efficacy of anti-IL-8 antibodies
- Monocytes isolated from healthy human peripheral blood as a CD14 positive fraction (All Cells, catalog number) : PB011-PF-2) was purchased and stored at -80 ° C. When used in the experiment, the cells were thawed and used as follows. A medium containing DNaseI (manufactured by STEMCELL Technologies, catalog number: 07900) and 10% FBS was added to the cells thawed in a 37 ° C. water bath, and the supernatant was removed after centrifugation with a high-speed centrifuge.
- DNaseI manufactured by STEMCELL Technologies, catalog number: 07900
- CTGF connecting tissue growth factor
- Monocytes were suspended in RPMI-1640 (SIGMA) medium containing 10% FBS and 25 ng / mL GM-CSF (SIGMA) and seeded on a culture plate. Half of the culture supernatant was changed every 2 or 3 days and cultured for 7 days.
- the culture supernatant was removed and 100 ng / mL in RPMI-1640 medium containing 10% FBS and 20 ng / mL IFN- ⁇ (manufactured by SIGMA), 10 pg / mL LPS (manufactured by Wako Pure Chemical Industries, Ltd.) mL IL-8 and various antibodies were added and cultured for 3 days.
- the antibodies evaluated were H1009 / L395-F1974m antibody, Anti-IL-8 antibody [807] (abcam, catalog number: ab18672), Anti-IL-8 antibody [6217] (abcam, catalog number: ab10768) ), Anti-IL-8 antibody (Becton Dickinson, catalog number: BD554726) and Anti-IL-8 antibody (Becton Dickinson, catalog number: BD555717) were used at a concentration of 10 ⁇ g / mL.
- IgG1 manufactured by SIGMA, catalog number: I5154
- cDNA was synthesized using SuperScript VILO MasterMix (manufactured by Thermo Fisher), and the expression level of CTGF mRNA was evaluated by real-time PCR.
- Taqman probe Hs00170014_m1 manufactured by Thermo Fisher
- 18S rRNA Eukaryotic 18S rRNA Endogenous Control
- CTGF 42 shows values obtained by correcting the expression level of CTGF with 18S rRNA.
- IL-8 has a function of inducing increased expression of CTGF on macrophages differentiated from monocytes.
- anti-IL-8 antibody was added simultaneously with IL-8, no increase in CTGF expression was observed. It was shown that the increase in the expression of CTGF by IL-8 is suppressed by various anti-IL-8 antibodies.
- IL-8 is a factor involved in the progression of fibrosis, and that IL-8 signal inhibitors such as anti-IL-8 antibodies are useful as therapeutic or preventive agents for fibrotic diseases.
- IL-8 signal inhibitors such as anti-IL-8 antibodies are useful as therapeutic or preventive agents for fibrotic diseases.
- HEK293H strain derived from human fetal kidney cancer cells is suspended in DMEM medium (Invitrogen) containing 10% Fetal Bovine Serum (Invitrogen), and the dish (diameter) has a cell density of 5-6 ⁇ 10 5 cells / mL. 10 mL to each dish of 10 cm, CORNING) After incubating overnight in a CO 2 incubator (37 ° C, 5% CO 2 ), the medium is removed by suction and CHO-S-SFM-II (Invitrogen) medium 6.9 mL was added.
- the prepared plasmid was introduced into cells by the lipofection method. After collecting the obtained culture supernatant, the cells are removed by centrifugation (approximately 2000 g, 5 minutes, room temperature), and further sterilized through a 0.22 ⁇ m filter MILLEX (R) -GV (Millipore). Got.
- the obtained culture supernatant was purified by a method known to those skilled in the art using rProtein A Sepharose TM Fast Flow (Amersham Biosciences). The concentration of the purified antibody was measured by measuring absorbance at 280 nm using a spectrophotometer. The antibody concentration was calculated from the obtained value using the extinction coefficient calculated by the method described in Protein Science 1995; 4: 2411-2423.
- anti-IL-8 antibody and the like are useful as a therapeutic and / or prophylactic agent for endometriosis and the like.
- the present invention is useful in reducing endometriotic lesion volume, improving adhesion, atrophic changes of epithelium and stroma cells, reducing infiltration of immune cells, improving fibrosis, and the like.
Abstract
Description
従来用いられてきた、子宮内膜症又は子宮腺筋症の治療法として、鎮痛剤、ホルモン療法、手術療法がある。鎮痛剤は、効果が限定的である上に、病態の進行を防ぐことはできない。ホルモン療法の場合、痛みの軽減や病態進行の遅延には効果があるが、月経を止めるため治療中は妊孕能がなくなること、治療を中止すると再び病態が進行する可能性が高いこと、さらに、ホルモン剤の副作用などが問題点としてある。また、ホルモン剤の投与によっても、投与後に妊孕能が改善するとの報告はない。手術療法の場合も、3~5年で約50%と再発する可能性は高く(非特許文献4及び5)、術後再発を予防するには、副作用に耐えてホルモン剤を服用し続ける必要がある。
IL-8は、炎症性サイトカインなどの刺激によって、末梢血単球、組織マクロファージ、NK細胞、線維芽細胞、血管内皮細胞など種々の細胞から産生される(非特許文献6)。IL-8は主に好中球を活性化し、細胞接着分子の発現を亢進し、好中球の血管内皮細胞への接着を高める作用を有すると言われている。また、IL-8は好中球走化能も有し、損傷組織で産生されるIL-8は血管内皮細胞に接着した好中球の組織への遊走を促進し、好中球浸潤に伴う炎症が惹起される。更にIL-8は、内皮細胞に対する有力な血管新生因子であり、又、腫瘍の血管新生に関連していることも知られている。
しかし、ヒトと同様の月経は霊長類以外に実験動物モデルでは存在しないため、ヒト子宮内膜症の原因究明に向けて、ラット、マウス等ではヒト子宮内膜症の真の意味での動物モデルにはなり得ない。そもそも、ラット、マウスにはIL-8が存在していない。また、最近までヒト子宮内膜症を正しく評価できる非ヒト霊長類のin vivoモデルがなかったこともあり、これまで子宮内膜症に対するIL-8シグナルの影響を実際に非ヒト霊長類のin vivoモデルで確認した報告は存在しない。
より具体的には、本発明は、非限定的な一実施態様において、新規なIL-8関連疾患に対する、IL-8シグナル阻害剤を用いた、新規な治療方法等を提供することにある。あるいは、本発明は、非限定的な別の実施態様において、新規又は既知のIL-8関連疾患に対する、新規な抗IL-8抗体を用いた、新規な治療方法等を提供することにある。
限定を意図しないが、本発明は、子宮内膜症等の病態解明のための非ヒト霊長類モデルの作製から、一方で、高機能抗IL-8抗体の作成と改良、抗体の薬理効果の評価を行うという、基礎から動物モデル臨床まで扱った包括的研究に基づくものである。ヒト子宮関連疾患の真の評価はラット、マウス、その他の小動物でのモデル実験では不可能であるため、本発明者らは、外科的に誘引された子宮内膜症モデルカニクイザルを作製し、カニクイザルを含む霊長類の生殖基礎研究、並びに、ヒト子宮内膜症等の病理学的病態概念及び評価と臨床(腹腔鏡手術と観察、MRI(磁気共鳴画像)評価等)管理などの優れた技術と知見等を互いに持ち寄り、試行錯誤と鋭意検討の末に本発明を完成させた。
また、非限定的な別の実施態様において、本発明者らは、外科的手術等により生じる癒着に対し抗炎症の側面からアプローチを行い、IL-8シグナル阻害剤を投与することで癒着が改善することを驚くべきことに見出した。
さらに、非限定的な別の実施態様において、本発明者らは、本開示における高機能抗IL-8抗体が、新規又は既知のIL-8関連疾患の治療等に有用であることに想到した。そのような高機能抗IL-8抗体としてはpH依存性抗IL-8抗体(pH依存的にIL-8に結合する抗IL-8抗体)であってよい。当該pH依存性抗IL-8抗体は、個体に投与されたときに参照抗体と比較して、以下の少なくとも1つ以上の特性:IL-8を速やかに消失させる、IL-8を中和する活性を安定的に維持する、免疫原性が低い、発現量が高い、を有し得るため有利である。あるいは、そのような高機能抗IL-8抗体は、以下の少なくとも1つ以上の特性を有する抗体:酸性pHにおけるFcRnに対する結合親和性が、天然型のFc領域のFcRnに対する結合親和性よりも増大しているFc領域を含む抗IL-8抗体、既存のADAに対する結合親和性が天然型のFc領域の既存のADAに対する結合親和性よりも低下しているFc領域を含む抗IL-8抗体、血漿中半減期が天然型のFc領域の血漿中半減期よりも増大しているFc領域を含む抗IL-8抗体、エフェクター受容体に対する結合親和性が天然型のFc領域のエフェクター受容体に対する結合親和性よりも低下しているFc領域を含むpH依存性抗IL-8抗体、であってよい。
〔1〕 ヒトIL-8に結合する単離された抗IL-8抗体を有効成分として含有する、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物であって、
前記抗IL-8抗体は以下(1)から(7)からなる群から選択される、組成物:
(1)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3;
(2)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:25のアミノ酸配列の3位のチロシン、配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む;
(3)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の9位のチロシンと11位のアルギニン、及び配列番号:25のアミノ酸配列の3位のチロシンでアミノ酸の置換をそれぞれ少なくとも含む;
(4)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の6位のアラニン、8位のグリシン、9位のチロシンと11位のアルギニン、及び配列番号:25のアミノ酸配列の3位のチロシンでアミノ酸の置換をそれぞれ少なくとも含む;
(5)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、及び配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む;
(6)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の9位のチロシンと11位のアルギニン、配列番号:25のアミノ酸配列の3位のチロシン、配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、及び配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む;並びに、
(7)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の6位のアラニン、8位のグリシン、9位のチロシンと11位のアルギニン、配列番号:25のアミノ酸配列の3位のチロシン、配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、及び配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む。
〔2〕 抗IL-8抗体は以下(8)から(19)からなる群から選択される、〔1〕に記載の組成物:
(8)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:29のアミノ酸配列を含み、及び(c)HVR-H3として配列番号:30のアミノ酸配列を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体;
(9)(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:31のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体;
(10)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:24のアミノ酸配列を含み、及び(c)HVR-H3として配列番号:30のアミノ酸配列を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体;
(11)(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:107のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体;
(12)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:29のアミノ酸配列を含み、(c)HVR-H3として配列番号:30のアミノ酸配列を含み、d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:31のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体;
(13)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:24のアミノ酸配列を含み、(c)HVR-H3として配列番号:30のアミノ酸配列を含み、(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:107のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、〔1〕の(1)又は(2)に記載の抗IL-8抗体;
(14)配列番号:34のアミノ酸配列を含む重鎖可変領域及び配列番号:35のアミノ酸配列を含む軽鎖可変領域を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体;
(15)配列番号:108のアミノ酸配列を含む重鎖可変領域及び配列番号:109のアミノ酸配列を含む軽鎖可変領域を含む、〔1〕の(1)又は(2)に記載の抗IL-8抗体;
(16)(a)配列番号:23のアミノ酸配列を含むHVR-H1、(b)配列番号:29のアミノ酸配列を含むHVR-H2、(c)配列番号:30のアミノ酸配列を含むHVR-H3、(d)配列番号:26のアミノ酸配列を含むHVR-L1、(e)配列番号:31のアミノ酸配列を含むHVR-L2、及び(f)配列番号:32のアミノ酸配列を含むHVR-L3とそれぞれ少なくとも80%の(アミノ酸)配列同一性を有するHVR-H1、HVR-H2、HVR-H3、HVR-L1、HVR-L2、及びHVR-L3を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体(ここで、当該抗体は、(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:29のアミノ酸配列を含み、(c)HVR-H3として配列番号:30のアミノ酸配列を含み、(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:31のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む抗体と機能的に同等であってもよい。);
(17)(a)配列番号:23のアミノ酸配列を含むHVR-H1、(b)配列番号:24のアミノ酸配列を含むHVR-H2、(c)配列番号:30のアミノ酸配列を含むHVR-H3、(d)配列番号:26のアミノ酸配列を含むHVR-L1、(e)配列番号:107のアミノ酸配列を含むHVR-L2、及び(f)配列番号:32のアミノ酸配列を含むHVR-L3とそれぞれ少なくとも80%の配列同一性を有するHVR-H1、HVR-H2、HVR-H3、HVR-L1、HVR-L2、及びHVR-L3を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体(ここで、当該抗体は、(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:24のアミノ酸配列を含み、(c)HVR-H3として配列番号:30のアミノ酸配列を含み、(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:107のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む抗体と機能的に同等であってもよい。);
(18)配列番号:34のアミノ酸配列を含む重鎖可変領域及び配列番号:35のアミノ酸配列を含む軽鎖可変領域とそれぞれ少なくとも80%の配列同一性を有する重鎖可変領域及び軽鎖可変領域を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体(ここで、当該抗体は、配列番号:34のアミノ酸配列を含む重鎖可変領域及び配列番号:35のアミノ酸配列を含む軽鎖可変領域を含む抗体と機能的に同等であってもよい。);並びに、
(19)配列番号:108のアミノ酸配列を含む重鎖可変領域及び配列番号:109のアミノ酸配列を含む軽鎖可変領域とそれぞれ少なくとも80%の配列同一性を有する重鎖可変領域及び軽鎖可変領域を含む、〔1〕の(1)から(7)のいずれかに記載の抗IL-8抗体(ここで、当該抗体は、配列番号:108のアミノ酸配列を含む重鎖可変領域及び配列番号:109のアミノ酸配列を含む軽鎖可変領域を含む抗体と機能的に同等であってもよい。)。
〔3〕 ヒトIL-8に結合する単離された抗IL-8抗体を有効成分として含有する、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物であって、
当該抗IL-8抗体は以下(I)から(VIII)からなる群から選択される、組成物:
(I)EUナンバリングで表される、235位、236位、239位、327位、330位、331位、428位、434位、436位、438位、及び440位からなる群から選択される一つ以上の位置にアミノ酸の置換を含むFc領域を含む、抗IL-8抗体;
(II)EUナンバリングで表される、434位、438位、及び440位からなる群から選択される全ての位置にアミノ酸の置換を含むFc領域を含む、抗IL-8抗体;
(III)EUナンバリングで表される、434位にAla;438位にGlu、Arg、Ser若しくはLys;及び、440位にGlu、Asp若しくはGlnの全てのアミノ酸置換を含むFc領域を含む、抗IL-8抗体;
(IV)EUナンバリングで表される、434位にAla;438位にGlu、Arg、Ser若しくはLys;及び、440位にGlu、Asp若しくはGlnの全てのアミノ酸置換を含み、さらに、428位にIle若しくはLeu、及び/又は、436位にIle、Leu、Val、Thr若しくはPheを含むFc領域を含む、抗IL-8抗体;
(V)EUナンバリングで表される、L235R, G236R, S239K, A327G, A330S, P331S, M428L, N434A, Y436T, Q438R, 及びS440Eからなる群から選択されるアミノ酸置換を一つ以上含むFc領域を含む、抗IL-8抗体;
(VI)EUナンバリングで表される、
N434A/Q438R/S440E;N434A/Q438R/S440D;
N434A/Q438K/S440E;N434A/Q438K/S440D;
N434A/Y436T/Q438R/S440E;N434A/Y436T/Q438R/S440D;
N434A/Y436T/Q438K/S440E;N434A/Y436T/Q438K/S440D;
N434A/Y436V/Q438R/S440E;N434A/Y436V/Q438R/S440D;
N434A/Y436V/Q438K/S440E;N434A/Y436V/Q438K/S440D;
N434A/R435H/F436T/Q438R/S440E;N434A/R435H/F436T/Q438R/S440D;
N434A/R435H/F436T/Q438K/S440E;N434A/R435H/F436T/Q438K/S440D;
N434A/R435H/F436V/Q438R/S440E;N434A/R435H/F436V/Q438R/S440D;
N434A/R435H/F436V/Q438K/S440E;N434A/R435H/F436V/Q438K/S440D;
M428L/N434A/Q438R/S440E;M428L/N434A/ Q438R/S440D;
M428L/N434A/Q438K/S440E;M428L/N434A/ Q438K/S440D;
M428L/N434A/Y436T/Q438R/S440E;M428L/N434A/Y436T/Q438R/S440D;
M428L/N434A/Y436T/Q438K/S440E;M428L/N434A/Y436T/Q438K/S440D;
M428L/N434A/Y436V/Q438R/S440E;M428L/N434A/Y436V/Q438R/S440D;
M428L/N434A/Y436V/Q438K/S440E;及び、M428L/N434A/Y436V/Q438K/S440D
からなる群から選択されるアミノ酸置換の組み合わせを含むFc領域を含む、抗IL-8抗体;
(VII)EUナンバリングで表される、
L235R/G236R/S239K/M428L/N434A/Y436T/Q438R/S440E;又は、
L235R/G236R/A327G/A330S/P331S/M428L/N434A/Y436T/Q438R/S440E
のアミノ酸置換の組み合わせを含むFc領域を含む、抗IL-8抗体;並びに、
(VIII)前記(I)から(VII)のいずれかに記載のFc領域を含み、当該Fc領域が以下の(a)から(e)からなる群から選択される性質の少なくとも一つを有する抗IL-8抗体:
(a)酸性pHでの、当該Fc領域のFcRnに対する結合親和性が、天然型のFc領域のFcRnに対する結合親和性よりも増大している、
(b)既存のADAに対する当該Fc領域の結合親和性が天然型のFc領域の既存のADAに対する結合親和性よりも低下している、
(c) 当該Fc領域の血漿中半減期が天然型のFc領域の血漿中半減期よりも増大している、
(d) 当該Fc領域の血漿中クリアランスが天然型のFc領域の血漿中クリアランスよりも減少している、及び
(e) エフェクター受容体に対する当該Fc領域の結合親和性が天然型のFc領域のエフェクター受容体に対する結合親和性よりも低下している。
〔4〕 ヒトIL-8に結合する単離された抗IL-8抗体を有効成分として含有する、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物であって、
当該抗IL-8抗体は以下(A)から(F)からなる群から選択される、組成物:
(A)配列番号:36のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖を含む抗IL-8抗体;
(B)配列番号:37のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖を含む抗IL-8抗体;
(C)配列番号:106のアミノ酸配列を含む重鎖及び配列番号:44のアミノ酸配列を含む軽鎖を含む抗IL-8抗体;
(D)配列番号:36のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖とそれぞれ少なくとも80%の配列同一性を有する重鎖及び軽鎖を含む抗IL-8抗体(ここで、当該抗体は、配列番号:36のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖を含む抗体と機能的に同等であってもよい。);
(E)配列番号:37のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖とそれぞれ少なくとも80%の配列同一性を有する重鎖及び軽鎖を含む抗IL-8抗体(ここで、当該抗体は、配列番号:37のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖を含む抗体と機能的に同等であってもよい。);並びに、
(F)配列番号:106のアミノ酸配列を含む重鎖及び配列番号:44のアミノ酸配列を含む軽鎖とそれぞれ少なくとも80%の配列同一性を有する重鎖及び軽鎖を含む抗IL-8抗体(ここで、当該抗体は、配列番号:106のアミノ酸配列を含む重鎖及び配列番号:44のアミノ酸配列を含む軽鎖を含む抗体と機能的に同等であってもよい。)。
〔5〕 線維化疾患が、子宮内膜症又は子宮腺筋症における線維化、慢性閉塞肺疾患(COPD)、嚢胞性線維症、乾癬、肝線維症、腎線維症、及び肺線維症からなる群から選択される、〔1〕~〔4〕のいずれかに記載の組成物。
〔6〕 IL-8シグナル阻害剤(好ましくはヒトIL-8シグナル阻害剤)を有効成分として含有する、ヒト子宮内膜症;子宮腺筋症;月経困難症;癒着;ヒト子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物。
〔7〕 免疫細胞のIL-8関連疾患の病変部又はその周辺環境への浸潤を抑制するための、〔6〕に記載の組成物。
〔8〕 アロマターゼ又は線維化因子の産生を阻害するための、〔6〕又は〔7〕に記載の組成物。
〔9〕 性周期への影響を与えない、〔6〕~〔8〕のいずれかに記載の組成物。
〔10〕 子宮内膜症又は子宮腺筋症における癒着又は線維化を抑制させるための、〔6〕~〔9〕のいずれかに記載の組成物。
〔11〕 (1)子宮内膜症又は子宮腺筋症の患者において子宮内膜の上皮細胞若しくはstroma細胞を萎縮させるための、又は、(2)子宮内膜症において子宮内膜のinterstitiumを減少させるための、〔6〕~〔10〕のいずれかに記載の組成物。
〔12〕 月経困難症が子宮内膜症若しくは子宮腺筋症を有する月経困難症であるか、又は、子宮内膜症若しくは子宮腺筋症が疑われる月経困難症である、〔6〕~〔11〕のいずれかに記載の組成物。
〔13〕 癒着が手術後に生じた癒着である、〔6〕に記載の組成物。
〔14〕 IL-8シグナル阻害剤がIL-8阻害剤(好ましくはヒトIL-8阻害剤)、CXCR1阻害剤(好ましくはヒトCXCR1阻害剤)、又はCXCR2阻害剤(好ましくはヒトCXCR2阻害剤)である、〔6〕~〔13〕のいずれかに記載の組成物。
〔15〕 IL-8阻害剤、CXCR1阻害剤、又はCXCR2阻害剤が、それぞれ抗IL-8抗体(好ましくは抗ヒトIL-8抗体)、抗CXCR1抗体(好ましくは抗ヒトCXCR1抗体)、又は抗CXCR2抗体(好ましくは抗ヒトCXCR2抗体)である、〔14〕に記載の組成物。
〔16〕 免疫細胞のIL-8関連疾患の病変部又はその周辺環境への浸潤を抑制するための、〔1〕~〔5〕のいずれかに記載の組成物。
〔17〕 アロマターゼ又は線維化因子の産生を阻害するための、〔1〕~〔5〕のいずれかに記載の組成物。
〔18〕 性周期への影響を与えない、〔1〕~〔5〕のいずれかに記載の組成物。
〔19〕 子宮内膜症又は子宮腺筋症における癒着又は線維化を抑制させるための、〔1〕~〔5〕のいずれかに記載の組成物。
〔20〕 (1)子宮内膜症又は子宮腺筋症の患者において子宮内膜の上皮細胞若しくはstroma細胞を萎縮させるための、又は、(2)子宮内膜症において子宮内膜のinterstitiumを減少させるための、〔1〕~〔5〕のいずれかに記載の組成物。
〔21〕 月経困難症が子宮内膜症若しくは子宮腺筋症を有する月経困難症であるか、又は、子宮内膜症若しくは子宮腺筋症が疑われる月経困難症である、〔1〕~〔5〕のいずれかに記載の組成物。
〔22〕 癒着が手術後に生じた癒着である、〔1〕~〔5〕のいずれかに記載の組成物。
〔23〕 IL-8関連疾患がIL-8シグナルに応答性を示す、〔1〕~〔22〕のいずれかに記載の組成物。
〔24〕 さらに薬学的に許容される担体を含む、〔1〕~〔23〕のいずれかに記載の組成物。
〔25〕子宮内膜症が、ヒト子宮内膜症である、〔1〕~〔5〕、〔16〕~〔24〕のいずれかに記載の組成物。
〔A1〕 子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療又は予防における使用のための、〔1〕~〔5〕、〔16〕~〔25〕のいずれかに規定される抗IL-8抗体。
〔A2〕 ヒト子宮内膜症;子宮腺筋症;月経困難症;癒着;ヒト子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療又は予防における使用のための〔6〕~〔15〕、〔23〕~〔25〕のいずれかに規定されるIL-8シグナル阻害剤。
〔A3〕 〔1〕~〔5〕、〔16〕~〔25〕のいずれかに規定される抗IL-8抗体又は〔1〕~〔5〕、〔16〕~〔25〕のいずれかに記載される組成物を、それを必要とする対象に投与することを含む、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患を治療又は予防する方法(ここで、それを必要とする対象とは、当該IL-8関連疾患に罹患した、又は、罹患している恐れのある対象であってよい。)。
〔A4〕〔6〕~〔15〕、〔23〕~〔25〕のいずれかに規定されるIL-8シグナル阻害剤又は〔6〕~〔15〕、〔23〕~〔25〕のいずれかに記載される組成物を、それを必要とする対象に投与することを含む、ヒト子宮内膜症;子宮腺筋症;月経困難症;癒着;ヒト子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患を治療又は予防する方法(ここで、それを必要とする対象とは、当該IL-8関連疾患に罹患した、又は、罹患している恐れのある対象であってよい。)。
〔A5〕 子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療又は予防のための医薬の製造における、〔1〕~〔5〕、〔16〕~〔25〕のいずれかに規定される抗IL-8抗体の使用。
〔A6〕 ヒト子宮内膜症;子宮腺筋症;月経困難症;癒着;ヒト子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療又は予防のための医薬の製造における〔6〕~〔15〕、〔23〕~〔25〕のいずれかに規定されるIL-8シグナル阻害剤の使用。
これらのいずれかに記載の一又は複数の要素(element)の一部又は全部を任意に組み合わせたものも、当業者の技術常識に基づいて技術的に矛盾しない限り、本発明に含まれることが意図され、かつ、当業者には当然に理解されるものとして記載される。
(a) アミノ酸残基26-32 (L1)、50-52 (L2)、91-96 (L3)、26-32 (H1)、53-55 (H2)、及び96-101 (H3)のところで生じる超可変ループ (Chothia and Lesk, J. Mol. Biol. 196:901-917 (1987));
(b) アミノ酸残基24-34 (L1)、50-56 (L2)、89-97 (L3)、31-35b (H1)、50-65 (H2)、及び95-102 (H3)のところで生じるCDR (Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, MD (1991));
(c) アミノ酸残基27c-36 (L1)、46-55 (L2)、89-96 (L3)、30-35b (H1)、47-58 (H2)、及び93-101 (H3)のところで生じる抗原接触 (MacCallum et al. J. Mol. Biol. 262: 732-745 (1996));並びに、
(d) HVRアミノ酸残基46-56 (L2)、47-56 (L2)、48-56 (L2)、49-56 (L2)、26-35 (H1)、26-35b (H1)、49-65 (H2)、93-102 (H3)、及び94-102 (H3)を含む、(a)、(b)、及び/又は(c)の組合せ。
別段示さない限り、HVR残基及び可変領域中の他の残基(例えば、FR残基)は、本明細書では上記のKabat et alにしたがって番号付けされる。
一実施態様において、本開示における抗体(例えば抗IL-8抗体、抗CXCR1抗体、又は抗CXCR2抗体)はヒト化抗体であってよい。典型的には、非ヒト抗体は、親の非ヒト抗体の特異性と親和性を維持しつつ、ヒトでの免疫原性を減少させるためにヒト化される。典型的には、ヒト化抗体は一つ以上の可変領域を含み、その中にはHVR、例えば非ヒト抗体に由来するCDR(又はその一部)と、ヒト抗体配列に由来するFR(又はその一部)とが存在する。ヒト化抗体は、任意に、ヒト定常領域の少なくとも一部を含むことができる。一実施態様において、ヒト化抗体内のFRのアミノ酸残基は、例えば、抗体の特異性や親和性を維持又は改善させるために、非ヒト抗体(例えばHVR残基の由来となった抗体)の対応するアミノ酸残基と置換されていてもよい。
ヒト化抗体及びその作製方法は、例えば以下で総説されており(Almagro and Fransson, Front. Biosci. 13:1619-1633 (2008))、更に例えば以下に記載されている:Riechmann et al., Nature 332:323-329 (1988); Queen et al., Proc. Nat'l Acad. Sci. USA 86:10029-10033 (1989); US Patent Nos. 5, 821,337, 7,527,791, 6,982,321, and 7,087,409; Kashmiri et al., Methods 36:25-34 (2005) (describing specificity determining region (SDR) grafting); Padlan, Mol. Immunol. 28:489-498 (1991) (describing "resurfacing"); Dall'Acqua et al., Methods 36:43-60 (2005) (describing "FR shuffling"); and Osbourn et al., Methods 36:61-68 (2005) and Klimka et al., Br. J. Cancer, 83:252-260 (2000) (describing the "guided selection" approach to FR shuffling)。
別の実施態様において、ヒト抗体は、ヒト由来ファージディスプレイライブラリから選択されるFvクローン可変ドメイン配列を単離することでも生成できる。このような可変領域配列は、次に所望のヒト定常領域と組み合わせることができる。抗体ライブラリからヒト抗体を選択する手法は以下を参照。
一実施態様において、一又は複数のアミノ酸置換を行った抗体改変体(変異体)が提供される場合には、置換的変異導入の目的部位は、HVR及びFRを含み得る。
一実施態様においての保存的置換を、表1の「好ましい置換」として示し、より実質的な変更を、表1の「例示的な置換」として示し、さらにアミノ酸側鎖の性質に言及しつつ以下で述べる。アミノ酸置換は目的の抗体に導入されてもよく、それによる産物は、例えば、保持/改善された抗原結合性、減少した免疫原性、又は改善したADCC若しくはCDCなどの所望の活性についてスクリーニングされてもよい。
(1)疎水性:ノルロイシン、Met、Ala、Val、Leu、Ile;
(2)中性の親水性:Cys、Ser、Thr、Asn、Gln;
(3)酸性:Asp、Glu;
(4)塩基性:His、Lys、Arg;
(5)鎖配向に影響する残基:Gly、Pro;
(6)芳香族:Trp、Tyr、Phe
非保存的置換は、これらの分類の中の1つのメンバーを他の分類に交換することを意味する。
アミノ酸配列比較にALIGN-2が用いられる場合、所与のアミノ酸配列Aの、所与のアミノ酸配列Bへの、またはそれとの、またはそれに対する、%アミノ酸配列同一性(あるいは、所与のアミノ酸配列Bへの、またはそれとの、またはそれに対する、ある%アミノ酸配列同一性を有するまたは含む所与のアミノ酸配列A、ということもできる)は、次のように計算される:分率X/Yの100倍。ここで、Xは配列アラインメントプログラムALIGN-2によって、当該プログラムのA及びBのアラインメントにおいて同一である一致としてスコアされたアミノ酸残基の数であり、YはB中のアミノ酸残基の全数である。アミノ酸配列Aの長さがアミノ酸配列Bの長さと異なる場合、AのBへの%アミノ酸配列同一性はBのAへの%アミノ酸配列同一性と異なることが理解される。特に明示しない限り、本明細書で用いられるすべての%アミノ酸配列同一性値は、上記ALIGN-2コンピュータプログラムを用いて得られる。
別の実施態様において、このようなハイブリダイゼーション技術の代わりに、CDR領域、重鎖可変領域、軽鎖可変領域、重鎖定常領域、軽鎖定常領域、重鎖全長領域、軽鎖全長領域、又はこれらの任意の特定領域のアミノ酸配列をコードする塩基配列情報を基に合成したプライマーを用いる遺伝子増幅法、例えばPCR法を利用して、該領域のアミノ酸配列をコードする塩基配列からなる核酸とストリンジェントな条件下でハイブリダイズする核酸を単離することも可能である。
このようにして単離された核酸は、対象となる領域の塩基配列に対して、塩基配列全体で、少なくとも50%以上、より好ましくは70%以上、75%以上、80%以上、85%以上、さらに好ましくは90%以上(例えば、91%、92%、93%、94%、95%、96%、97%、98%、若しくは99%以上)の配列の同一性を有する。このような単離された核酸によってコードされるアミノ酸配列を有する抗体は、対象となった領域のアミノ酸配列を有する抗体と、機能的に(実質的に)同等(例えば抗原結合性又は血中滞留性といった任意の1つ以上の指標又は特性が(実質的に)同等)であると有利であるがそれに限定されない。ここで実質的とは、対象となった領域のアミノ酸配列を有する抗体が有する機能(例えば抗原結合性又は血中滞留性などの任意の1つ以上の指標又は特性に着目してよい)と比較して、当業者公知の方法で測定した場合に、少なくとも50%以上、より好ましくは70%以上、75%以上、80%以上、85%以上、さらに好ましくは90%以上(例えば、91%、92%、93%、94%、95%、96%、97%、98%、若しくは99%以上)の機能を保持していることを意図する。
FcRn結合ドメインは、直接FcRnと結合する領域であることが好ましい。FcRn結合領域の好ましい例として、抗体の定常領域又はFc領域を挙げることができる。しかしながら、アルブミンやIgGなどのFcRnとの結合活性を有するポリペプチドに結合可能な領域は、アルブミンやIgGなどを介して間接的にFcRnと結合することが可能である。そのため、FcRn結合領域は、アルブミンやIgGとの結合活性を有するポリペプチドに結合する領域であってもよい。限定はされないが、血漿中からの抗原の消失を促進するためには、FcRn結合ドメインは、中性pHにおけるFcRn結合活性が高いほうが好ましく、また、抗体の血漿中の滞留性を向上させるためには、FcRn結合ドメインは、酸性pHにおけるFcRn結合活性が高いほうが好ましい。例えば、予め、中性pH又は酸性pHにおけるFcRn結合活性が高いFcRn結合ドメインを選択して用いてもよいし、あるいは、抗体又はFc領域中のアミノ酸を改変して中性pH又は酸性pHにおけるFcRn結合活性を付与しても良いし、あるいは、中性pH又は酸性pHにおける既に存在するFcRn結合活性を増大させてもよい。
また、抗体又はFc領域(改変体)のFcRnに対する結合活性は、KD(Dissociation constant:解離定数)、見かけのKD(Apparent dissociation constant:見かけの解離定数)、解離速度であるkd(Dissociation rate:解離速度)、又は見かけのkd(Apparent dissociation:見かけの解離速度)等として評価されてよい。
本開示における「CXCR2」とは、特に示さない限り、あらゆる脊椎動物、霊長類(例えばヒト、カニクイザル、又はアカゲザル)、又はその他の哺乳類(例えば、ウサギ又はイヌ)に存在する天然のCXCR2を指す。CXCR2は、全長のCXCR2、生体内で修飾されたいかなる形態のCXCR2、天然に存在するCXCR2の派生物、例えば、スプライス変異体やアレル変異体のようなものも含む。非限定的なヒトCXCR2のアミノ酸配列の一例としてはNCBIにNP_001161770.1やNP_001548.1として登録されている。
本実施例に記載されるように、本発明者らは、CXCR1及びCXCR2が、好中球、単核球、マクロファージといった免疫細胞、子宮内膜症上皮細胞、及び、血管内皮細胞で発現しているが、stroma細胞及び筋線維芽細胞では発現していないことを見出した。さらに、子宮内膜症ヒト患者から採取し培養した子宮内膜症由来のstroma細胞および筋線維芽細胞にIL-8や抗IL-8抗体を添加しても、細胞の増殖や萎縮等の変化は何ら見られなかった(data not shown)。
これらの結果は、Ulukus et al., Human Reproduction 2005 20(3):794-801において、CXCR1が子宮内膜症組織の上皮、stroma、線維化した部分で強発現しているという報告とは一致しなかった。Ulukusの論文では、全体的に染色強度が高かった。CXCR2においても弱陽性ではあるが血管内皮細胞とともにstroma細胞も染色された。加えて、Iwabe et al., Fertility and Sterility 1998 69(5):924-930において、in vitro実験で子宮内膜症患者由来のstroma細胞の増殖がIL-8により促進され、抗IL-8抗体の添加により抑制された報告とは一致しなかった。Iwabeの論文では、子宮内膜症ヒト患者由来の内膜症病変から上皮細胞を除去し、stroma細胞とした後に、IL-8を添加して細胞増殖を調べているが、20%前後の弱い反応であった。
他方、本実施例に記載されるように、本発明者らは、非ヒト霊長類子宮内膜症モデルにおいて、抗体H1009/L395-F1974m投与といったIL-8シグナル阻害剤の投与により増殖性を示す上皮細胞及びstroma細胞の萎縮性変化が見られることを実証した。
理論に拘束されることを意図しないが、一側面において、Iwabeの論文では、細胞継代後すぐに実験を実施していることから、免疫細胞が混在している可能性が推定される。CXCR1、CXCR2はstroma細胞には発現していないことから、CXCR1、CXCR2を介してのIL-8シグナルは、stroma細胞を標的としては直接作用せず、子宮内膜上皮や免疫細胞を直接の標的として作用し、stroma細胞へはこれら子宮内膜上皮や免疫細胞のシグナルを介して間接的に作用しているものと考えられた。
IL-8シグナル阻害剤がIL-8のその受容体であるCXCR1及び/又はCXCR2への結合を部分的又は完全に阻害する場合には、IL-8の阻害により、阻害なしでIL-8がその受容体に結合したときに生じる正常なレベル又は種類の活性が低下又は変化することが好ましい。その低下又は変化は、例えば、IL-8誘導性エラスターゼ放出若しくはカルシウム流束の阻害、IL-8誘導性のCD11b(Mac-1)発現増加の阻害、又は、L-セレクチン発現減少の阻害などとして観察してよい。このような阻害は、IL-8シグナル阻害剤非存在下と比較した場合に、少なくとも、例えば、10%、20%、30%、40%、50%、60%、70%、80%、90%、95%、99%、又は100%の上述のレベル又は種類の活性の低下又は変化を包含する。
一実施態様において、本開示におけるCXCR2阻害剤は、CXCR2に結合する(特異的である)がCXCR1には結合しないものであってもよいし、CXCR1とCXCR2の両方に結合する(特異的である)もの(CXCR1/CXCR2阻害剤)であってもよい。
一実施態様において、本開示におけるCXCR1阻害剤及び/又はCXCR2阻害剤の具体例としては、WO2010/056753に開示される抗CXCR1抗体、又は、レパルタキシン(Repartaxin)若しくはレパルタキシン誘導体;WO2011/042466に開示されるCXCR1及び/又はCXCR2阻害薬;WO2005/113534やWO2003/057676に開示されるCXCR1又はCXCR2ケモカインアンタゴニスト;WO2012/062713及びWO2013/168108に開示されるCXCR2結合ポリペプチド;WO2008/061740、WO2008/061741、WO2008/062026、WO2009/106539、WO2010/015613及びWO2013/030803に開示されるCXCR2阻害剤が挙げられる。
一実施態様において、非特異的な無関係の非IL-8タンパク質に対する抗IL-8抗体の結合の程度は、IL-8に対する抗体の結合の程度の例えば10%未満であってもよい。
一実施態様において、本開示における抗IL-8抗体は、キメラ抗体、ヒト化抗体、又は、ヒト抗体であってよい。
一実施態様において、本開示における抗IL-8抗体は、IL-8を内在的に有する動物(例えばウサギ)と交差性を有するのが好ましく、さらに、場合により、抗霊長類IL-8抗体(好ましくはヒトと同様の月経を有する、例えば、カニクイザル又はヒヒなどに対する抗非ヒト霊長類IL-8抗体又は抗ヒトIL-8抗体(例えば抗ヒトIL-8中和抗体)であることがより好ましい。
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3。
アミノ酸は、特段の指定のない限り、他のいかなるアミノ酸に置換されてもよい。さらなる実施態様において、本開示のかかる抗IL-8抗体は、以下の位置からなる群から選択される位置での他のアミノ酸への置換、又は、当該位置での下記に指定のアミノ酸への置換をそれぞれ少なくともさらに含んでよい:
(1)配列番号:26の配列の8位のセリン、配列番号:27の配列の1位のアスパラギンと5位のロイシン、及び、配列番号:28の配列の1位のグルタミン;
(2)配列番号:24の配列の6位のアラニンのアスパラギン酸への置換と、11位のアルギニンのプロリンへの置換と、及び、配列番号:25の配列の3位のチロシンのヒスチジンへの置換;
(3)配列番号:24の配列の8位のグリシンのチロシンへの置換と9位のチロシンのヒスチジンへの置換;
(4)配列番号:24の配列の6位のアラニンのアスパラギン酸への置換と、8位のグリシンのチロシンへの置換と、9位のチロシンのヒスチジンへの置換と、11位のアルギニンのプロリンへの置換と、及び、配列番号:25の配列の3位のチロシンのヒスチジンへの置換;
(5)配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換;
(6)配列番号:26の配列の8位のセリンのグルタミン酸への置換と、配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換;
(7)配列番号:24の配列の9位のチロシンのヒスチジンへの置換と11位のアルギニンのプロリンへの置換と、配列番号:25の配列の3位のチロシンのヒスチジンへの置換と、配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換;及び、
(8)配列番号:24の配列の6位のアラニンのアスパラギン酸への置換と8位のグリシンのチロシンへの置換と9位のチロシンのヒスチジンへの置換と11位のアルギニンのプロリンへの置換と、配列番号:25の配列の3位のチロシンのヒスチジンへの置換と、配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換。
(a)配列番号:23のアミノ酸配列を含むHVR-H1、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有するHVR-H1;
(b)配列番号:24又は29のアミノ酸配列を含むHVR-H2、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有するHVR-H2;
(c)配列番号:25又は30のアミノ酸配列を含むHVR-H3、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有するHVR-H3;
(d)配列番号:26のアミノ酸配列を含むHVR-L1、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有するHVR-L1;
(e)配列番号:27、31、又は、107のアミノ酸配列を含むHVR-L2、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有するHVR-L2;及び、
(f)配列番号:28又は32のアミノ酸配列を含むHVR-L3、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有するHVR-L3。
ここで、参照アミノ酸配列と特定%の配列同一性を有するHVRは、参照アミノ酸配列を有するHVRと機能的に同等であってよい。また、例えば、当該抗IL-8抗体は、配列番号:29のアミノ酸配列を含むHVR-H2及び配列番号:30のアミノ酸配列を含むHVR-H3を含んでもよいし、及び/又は、配列番号:31のアミノ酸配列を含むHVR-L2及び配列番号:32のアミノ酸配列を含むHVR-L3を含んでもよい。さらに、当該抗IL-8抗体は、以下の位置からなる群から選択される位置での他のアミノ酸への置換、又は、当該位置での下記に指定のアミノ酸への置換をそれぞれ少なくともさらに含んでよい:
(1)配列番号:26の配列の8位のセリン、配列番号:27の配列の1位のアスパラギンと5位のロイシン、及び、配列番号:28の配列の1位のグルタミン;
(2)配列番号:24の配列の6位のアラニンのアスパラギン酸への置換と、11位のアルギニンのプロリンへの置換と、及び、配列番号:25の配列の3位のチロシンのヒスチジンへの置換;
(3)配列番号:24の配列の8位のグリシンのチロシンへの置換と9位のチロシンのヒスチジンへの置換;
(4)配列番号:24の配列の6位のアラニンのアスパラギン酸への置換と、8位のグリシンのチロシンへの置換と、9位のチロシンのヒスチジンへの置換と、11位のアルギニンのプロリンへの置換と、及び、配列番号:25の配列の3位のチロシンのヒスチジンへの置換;
(5)配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換;
(6)配列番号:26の配列の8位のセリンのグルタミン酸への置換と、配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換;
(7)配列番号:24の配列の9位のチロシンのヒスチジンへの置換と11位のアルギニンのプロリンへの置換と、配列番号:25の配列の3位のチロシンのヒスチジンへの置換と、配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換;及び、
(8)配列番号:24の配列の6位のアラニンのアスパラギン酸への置換と8位のグリシンのチロシンへの置換と9位のチロシンのヒスチジンへの置換と11位のアルギニンのプロリンへの置換と、配列番号:25の配列の3位のチロシンのヒスチジンへの置換と、配列番号:27の配列の1位のアスパラギンのリジンへの置換と5位のロイシンのヒスチジンへの置換と、及び、配列番号:28の配列の1位のグルタミンのリジンへの置換。
ここに記載される、想定されるあらゆるアミノ酸配列の組み合わせを当然に当業者は理解する。
(A)配列番号:33、34、若しくは、108のアミノ酸配列を含む重鎖可変領域、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有する重鎖可変領域;及び、
(B)配列番号:35若しくは109のアミノ酸配列を含む軽鎖可変領域、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有する軽鎖可変領域。
ここで、参照アミノ酸配列と特定%の配列同一性を有する可変領域は、参照アミノ酸配列を有する可変領域と機能的に同等であってよい。また、例えば、当該抗IL-8抗体は、配列番号:34のアミノ酸配列を含む重鎖可変領域及び配列番号:35のアミノ酸配列を含む軽鎖可変領域を含んでもよく、かかる抗体は生体内(例えば血漿中)でIL-8中和活性が安定に保たれるので有利であり得るし、或いは、免疫原性が低いので有利であり得る。さらに、当該抗IL-8抗体が配列番号:33のアミノ酸配列の改変体を含む場合には、当該抗体は以下の位置からなる群から選択される一つ以上の位置での他のアミノ酸への置換、又は、当該位置での下記に指定されるアミノ酸への置換を少なくともさらに含んでよい:
(i)配列番号:33のアミノ酸配列の55位のアラニンの他のアミノ酸への置換、例えば、アスパラギン酸への置換;
(ii)配列番号:33のアミノ酸配列の57位のグリシンの他のアミノ酸への置換、例えば、チロシンへの置換;
(iii)配列番号:33のアミノ酸配列の58位のチロシンの他のアミノ酸への置換、例えば、ヒスチジンへの置換;
(iv)配列番号:33のアミノ酸配列の60位のアルギニンの他のアミノ酸への置換、例えば、プロリンへの置換;
(v)配列番号:33のアミノ酸配列の84位のグルタミンの他のアミノ酸への置換、例えば、スレオニンへの置換;
(vi)配列番号:33のアミノ酸配列の87位のセリンの他のアミノ酸への置換、例えば、アスパラギン酸への置換;及び
(vii)配列番号:33のアミノ酸配列の103位のチロシンの他のアミノ酸への置換、例えば、ヒスチジンへの置換。
ここに記載される、想定されるあらゆるアミノ酸配列の組み合わせを当然に当業者は理解する。
(1)(a)配列番号:58のアミノ酸配列を含むHVR-H1、
(b)配列番号:59のアミノ酸配列を含むHVR-H2、
(c)配列番号:60のアミノ酸配列を含むHVR-H3、
(d)配列番号:61のアミノ酸配列を含むHVR-L1、
(e)配列番号:62のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:63のアミノ酸配列を含むHVR-L3。
(2)(a)配列番号:64のアミノ酸配列を含むHVR-H1、
(b)配列番号:65のアミノ酸配列を含むHVR-H2、
(c)配列番号:66のアミノ酸配列を含むHVR-H3、
(d)配列番号:67のアミノ酸配列を含むHVR-L1、
(e)配列番号:68のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:69のアミノ酸配列を含むHVR-L3。
(3)(a)配列番号:70のアミノ酸配列を含むHVR-H1、
(b)配列番号:71のアミノ酸配列を含むHVR-H2、
(c)配列番号:72のアミノ酸配列を含むHVR-H3、
(d)配列番号:73のアミノ酸配列を含むHVR-L1、
(e)配列番号:74のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:75のアミノ酸配列を含むHVR-L3。
(4)(a)配列番号:76のアミノ酸配列を含むHVR-H1、
(b)配列番号:77のアミノ酸配列を含むHVR-H2、
(c)配列番号:78のアミノ酸配列を含むHVR-H3、
(d)配列番号:79のアミノ酸配列を含むHVR-L1、
(e)配列番号:80のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:81のアミノ酸配列を含むHVR-L3。
(5)(a)配列番号:82のアミノ酸配列を含むHVR-H1、
(b)配列番号:83のアミノ酸配列を含むHVR-H2、
(c)配列番号:84のアミノ酸配列を含むHVR-H3、
(d)配列番号:85のアミノ酸配列を含むHVR-L1、
(e)配列番号:86のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:87のアミノ酸配列を含むHVR-L3。
(6)(a)配列番号:88のアミノ酸配列を含むHVR-H1、
(b)配列番号:89のアミノ酸配列を含むHVR-H2、
(c)配列番号:90のアミノ酸配列を含むHVR-H3、
(d)配列番号:91のアミノ酸配列を含むHVR-L1、
(e)配列番号:92のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:93のアミノ酸配列を含むHVR-L3。
(7)(a)配列番号:94のアミノ酸配列を含むHVR-H1、
(b)配列番号:95のアミノ酸配列を含むHVR-H2、
(c)配列番号:96のアミノ酸配列を含むHVR-H3、
(d)配列番号:97のアミノ酸配列を含むHVR-L1、
(e)配列番号:98のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:99のアミノ酸配列を含むHVR-L3。
また、WO2013/046704では、酸性pH下でのFcRnに対する結合を増大させることが可能なアミノ酸の置換と組み合わせた場合に、リウマトイド因子結合の有意な低下をもたらすことができた2残基のアミノ酸の置換として、具体的に、EUナンバリングで表される、Q438R/S440E、Q438R/S440D、Q438K/S440E、Q438K/S440Dを報告している。
N434A/Q438R/S440E;N434A/Q438R/S440D;
N434A/Q438K/S440E;N434A/Q438K/S440D;
N434A/Y436T/Q438R/S440E;N434A/Y436T/Q438R/S440D;
N434A/Y436T/Q438K/S440E;N434A/Y436T/Q438K/S440D;
N434A/Y436V/Q438R/S440E;N434A/Y436V/Q438R/S440D;
N434A/Y436V/Q438K/S440E;N434A/Y436V/Q438K/S440D;
N434A/R435H/F436T/Q438R/S440E;N434A/R435H/F436T/Q438R/S440D;
N434A/R435H/F436T/Q438K/S440E;N434A/R435H/F436T/Q438K/S440D;
N434A/R435H/F436V/Q438R/S440E;N434A/R435H/F436V/Q438R/S440D;
N434A/R435H/F436V/Q438K/S440E;N434A/R435H/F436V/Q438K/S440D;
M428L/N434A/Q438R/S440E;M428L/N434A/ Q438R/S440D;
M428L/N434A/Q438K/S440E;M428L/N434A/ Q438K/S440D;
M428L/N434A/Y436T/Q438R/S440E;M428L/N434A/Y436T/Q438R/S440D;
M428L/N434A/Y436T/Q438K/S440E;M428L/N434A/Y436T/Q438K/S440D;
M428L/N434A/Y436V/Q438R/S440E;M428L/N434A/Y436V/Q438R/S440D;
M428L/N434A/Y436V/Q438K/S440E;及び、M428L/N434A/Y436V/Q438K/S440D;
L235R/G236R/S239K/M428L/N434A/Y436T/Q438R/S440E;及び、
L235R/G236R/A327G/A330S/P331S/M428L/N434A/Y436T/Q438R/S440E
からなる群から選択される、置換されたアミノ酸の組み合わせを含んでよい。
あるいは別の実施態様において、本開示における抗IL-8抗体中のFc領域改変体は、天然型Fc領域と比較して、EUナンバリングで表される、235位、236位、239位、327位、330位、331位、428位、434位、436位、438位、及び440位からなる群から選択される一つ以上(例えば、六つ、七つ又は全て)の位置で他のアミノ酸への置換を含んでよく、例えば、L235R, G236R, S239K, A327G, A330S, P331S, M428L, N434A, Y436T, Q438R, 及びS440Eからなる群から選択されるアミノ酸置換を一つ以上(例えば、六つ、七つ又は全て)含んでよい。
あるpH下でのFcRnに対するFcRn結合ドメインの結合親和性の増大は、天然型のFcRn結合ドメインについて測定されたFcRn結合親和性と比較した、測定されたFcRn結合親和性の増大に相当してよい。かかる場合、結合親和性の差であるKD(天然型Fc領域)/KD(本開示における抗IL-8抗体中のFc領域改変体)は、少なくとも1.5倍、2倍、3倍、4倍、5倍、10倍、15倍、20倍、50倍、70倍、80倍、100倍、500倍、または1000倍であってよい。FcRnに対するFcRn結合ドメインの結合親和性の増大は、酸性pH及び/又は中性pHにおけるものであってよいが、特には酸性pH下でFcRnに対する結合活性が増大されていることが好ましい。
また、酸性pH下でFcRnに対する結合親和性が増大されている本開示における抗IL-8抗体中のFc領域改変体は、天然型のIgGのFc領域よりもFcRnに対する結合親和性が、例えばpH 6.0及び25℃において、例えば、1.5倍、2倍、3倍、4倍、5倍、6倍、7倍、8倍、9倍、10倍、20倍、30倍、50倍、75倍、100倍、200倍、500倍、1000倍以上強くてよい。
一実施態様において、本開示における抗IL-8抗体中のFc領域改変体は、天然型IgGのFc領域と比較して、中性pH下で既存のADA、好ましくはリウマトイド因子(RF)に対する結合親和性が有意に増大されておらず、及び/又は、酸性pH下でFcRnに対する結合活性が増大されていることで、血漿中クリアランス(CL)が減少されているか、血漿中滞留時間が増大されているか、又は、血漿中半減期(t1/2)が増大されていることが好ましい。抗体の血漿中クリアランス(CL)、血漿中滞留時間、血漿中半減期(t1/2)が互いに相関していることは当業者に公知である。
本実施例1~3では、WO2013/046704に記載のF1718というFc領域改変体(Fc領域にN434Y/Y436V/Q438R/S440Eという4か所の変異が導入されている。)と、Fc領域改変体F1848m(N434A/Y436V/Q438R/S440Eという4か所の変異が導入されている。)との2つのFc領域改変体の血漿中滞留性が比較されている。両者のFc領域改変体のアミノ酸変異の違いは、EUナンバリング434番に導入されているアミノ酸変異が、F1718はY(チロシン)であり、F1848mはA(アラニン)であるという点のみであった。それにも関わらず、F1848mは天然型IgG1の場合に比べて血漿中滞留性の向上が認められた一方で、F1718は血漿中滞留性の向上が認められなかった(特に、本実施例(3-2)を参照)。また、本実施例(1-2)及び本実施例(3-3)の結果より、各種Fc領域改変体のうち、F1848mよりも、F1847m、F1886m、F1889m、F1927mの方が血漿中滞留性の向上が認められた。したがって、F1848mのみならず、F1847m、F1886m、F1889m、F1927mを含むFc領域改変体は、EUナンバリングで表される、N434Y/Y436V/Q438R/S440Eの置換されたアミノ酸の組み合わせを含む当該参照Fc領域改変体と比較して、血漿中滞留性が向上していることを当業者であれば当然に予期できる。
さらなる又は別の実施態様において、本開示におけるFc領域改変体を含む抗IL-8抗体は、エフェクター受容体に対する結合親和性が低いか又は結合親和性を有さないのが好ましい。エフェクター受容体の例には、活性化FcγR、特にFcγRI、FcγRII、FcγRIIIが含まれる。FcγRIには、例えば、FcγRIa、FcγRIb、FcγRIc、並びにこれらのサブタイプが含まれる。FcγRIIには、例えば、FcγRIIa(2つのアロタイプR131、H131)及びFcγRIIbが含まれる。FcγRIIIには、例えば、FcγRIIIa(2つのアロタイプV158、F158)及びFcγRIIIb(2つのアロタイプFcγIIIb-NA1、FcγIIIb-NA2)が含まれる。エフェクター受容体に対する結合親和性が低いか又は有さない抗体は、天然型のIgG Fc領域を含む抗体の結合親和性と比較して、例えば、10分の1以下、50分の1以下、又は100分の1以下に低下しているFc領域改変体を含む抗体を指してよい。具体例としては、例えば、サイレントFc領域を含む抗体、またはFc領域を含まない抗体(例えば、Fab、F(ab)'2、scFv、sc(Fv)2、diabody)が挙げられる。
(A)配列番号:36、37又は106のアミノ酸配列を含む重鎖、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有する重鎖;並びに/あるいは、
(B)配列番号:38又は44のアミノ酸配列を含む軽鎖、又は、それと少なくとも65%、70%、若しくは75%以上、好ましくは80%、85%若しくは90%以上、より好ましくは95%以上の配列同一性を有する軽鎖
を含んでよい。ここで、参照アミノ酸配列と特定%の配列同一性を有する重鎖又は軽鎖は、参照アミノ酸配列を有する重鎖又は軽鎖と機能的に同等であってよい。さらに、当該抗IL-8抗体は、以下の5つの性質のうち少なくとも一つの性質を有するFc領域改変体を含んでよい:
(a) 酸性pHでのFc領域改変体のFcRnに対する結合親和性が、天然型のFc領域のFcRnに対する結合親和性よりも増大している、
(b) 既存のADAに対するFc領域改変体の結合親和性が天然型のFc領域の既存のADAに対する結合親和性よりも低下している、
(c) Fc領域改変体の血漿中半減期が天然型のFc領域の血漿中半減期よりも増大している、
(d) Fc領域改変体の血漿中クリアランスが天然型のFc領域の血漿中クリアランスよりも減少している、
(e) Fc領域改変体のエフェクター受容体に対する結合親和性が天然型のFc領域のエフェクター受容体に対する結合親和性よりも低下している、
(f) pH依存的にIL-8に結合し得る。
ここに記載される、想定されるあらゆるアミノ酸配列の組み合わせを当然に当業者は理解する。
さらなる実施態様では、このような核酸を含む一つ以上のベクター(例えば発現ベクター)が提供される。一実施態様において、このような核酸を含む宿主細胞が提供される。当該宿主細胞は、(1)抗体のVLを含むアミノ酸配列及び/又はVHを含むアミノ酸配列をコードする核酸を含むベクター、又は、(2)抗体のVLを含むアミノ酸配列をコードする核酸を含む第一のベクターと抗体のVHを含むアミノ酸配列をコードする核酸を含む第二のベクターを含む(例えば、これらベクターで細胞は形質転換されている)。
一実施態様において、宿主は真核生物(例えばCHO細胞やリンパ細胞(例えばY0, NS0, SP20細胞))である。
一実施態様において、本開示における抗IL-8抗体の製造方法が提供され、その製造方法は、例えば、上記核酸を含む宿主細胞を、抗体発現に適した条件で培養し、任意に宿主細胞(又は宿主細胞の培養培地)から当該抗体を回収することを含む。
ある側面では、本開示における抗体は、例えば、ELISAやウェスタンブロット、結合平衡除外法(Kinetic Exclusion Assay;KinExATM)、又はBIACORE(GE Healthcare)などの装置を用いた表面プラズモン共鳴法のような方法で抗原結合活性を調べてよい。
ある側面では、測定は生物学的活性を有する抗体を同定するために提供される。生物学的活性には、例えば、抗原(例えばIL-8)を中和する活性、又は、抗原(例えばIL-8)のシグナルを遮断する活性が含まれる。
本明細書において「薬学的に許容される担体」とは、医薬組成物中に存在する有効成分以外の構成要素を指し、対象に対して無害なものが典型的には選択されるが、目的に応じたリスク・ベネフィットを勘案して決定されよう。薬学的に許容される担体は、例えば、緩衝液、賦形剤、安定剤、又は保存剤を含む。
一実施態様において、本開示における治療用組成物又は予防用組成物は薬学的に許容される担体を含み、凍結乾燥製剤又は水溶液製剤の形態で調製され得る。
ある側面において、本開示は、IL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)が医薬組成物として利用可能であることに基づく。本開示におけるIL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)は、例えば、IL-8が過剰に存在する疾患の診断、治療又は予防に有益である。
一実施態様において、本開示におけるIL-8シグナル阻害剤は、医学的実用性に合わせた様式で、製剤化され、用量設定され、投与され得る。
一実施態様において、本開示は、本明細書に開示される疾患のための診断、治療、及び/又は予防に有用なIL-8シグナル阻害剤を含む製品に関する。この製品は、容器と、当該容器に付随するラベル又は添付文書を含む。適切な容器には、例えば、ビン、バイアル、静脈注射用溶液バッグが含まれる。
ある側面における一実施態様において、本開示は、IL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)を有効成分として含有する、IL-8関連疾患の治療用組成物又は予防用組成物に関する。さらなる実施態様において、当該組成物は、薬学的に許容される担体をさらに含んでよい。
ある側面における一実施態様において、本開示は、IL-8関連疾患(例えば、子宮内膜症、子宮腺筋症、月経困難症、癒着、線維化疾患)の治療又は予防における使用のための、IL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)に関する。
ある側面における一実施態様において、本開示は、IL-8関連疾患(例えば、子宮内膜症、子宮腺筋症、月経困難症、癒着、線維化疾患)に対する診断用組成物、治療用組成物、又は、予防用組成物としてのIL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)の使用に関する。
ある側面における一実施態様において、本開示は、IL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)又はそれを含む治療用組成物若しくは予防用組成物を、それを必要とする対象に(有効量で)投与することを含む、IL-8関連疾患(例えば、子宮内膜症、子宮腺筋症、月経困難症、癒着、線維化疾患など)を治療又は予防する方法に関する。ここで、それを必要とする対象とは、当該IL-8関連疾患に罹患した、又は、罹患している恐れのある対象であってよい。さらなる実施態様において、当該組成物は、薬学的に許容される担体をさらに含んでよい。
ある側面における一実施態様において、本開示は、IL-8関連疾患(例えば、子宮内膜症、子宮腺筋症、月経困難症、癒着、線維化疾患など)の治療又は予防のための医薬の製造における、本開示のIL-8シグナル阻害剤(例えば、IL-8阻害剤(例えば抗IL-8抗体)、CXCR1阻害剤、又はCXCR2阻害剤)又はそれを含む治療用組成物若しくは予防用組成物の使用に関する。さらなる実施態様において、当該組成物は、薬学的に許容される担体をさらに含んでよい。
一実施態様において、本開示におけるIL-8シグナル阻害剤による治療又は予防の対象となるIL-8関連疾患の患者(対象)は、当該IL-8関連疾患に罹患した、又は、罹患している恐れのある対象であってよい。ここで、IL-8関連疾患に罹患している恐れのある対象とは、限定はされないが、IL-8関連疾患に過去に罹患しており、症状が再燃する恐れのある対象でもよいし、あるいは、IL-8関連疾患との例えば病理所見による確定診断などが医師等によって下される前の、IL-8関連疾患を発症していると疑われている対象であってもよい。一実施態様において、IL-8関連疾患の予防と治療は場合によって同義に解されてもよい。あるいは、IL-8関連疾患の予防とは、適切な場合には、当業者に周知の一般的な観念で解されてもよい。
一実施態様において、本開示におけるIL-8関連疾患はIL-8シグナルに応答性を示す。
本発明者らは、上述のカニクイザルin vivoモデルが子宮腺筋症を自然発症したことを確認、同定し、IL-8シグナル阻害剤を生体に投与したところ、子宮腺筋症の病変部の増殖を抑制できることを世界で初めて実証することに成功した。カニクイザルin vivoモデルにおいて、IL-8シグナル阻害剤は性周期への影響を与えなかったことから(本実施例参照)、既存の子宮腺筋症治療剤と比較してIL-8シグナル阻害剤は有望な治療剤又は予防剤である。さらに、ホルモン剤との併用によらずともIL-8シグナル阻害剤単剤で子宮腺筋症を治療又は予防できることは驚くべきことである。IL-8シグナル阻害剤を投与することで、子宮腺筋症における肥大した子宮壁を薄くさせることが可能であり、子宮腺筋症における線維化に対する治療又は予防効果が想起される。一実施態様において、本開示における子宮腺筋症はIL-8シグナルに応答性を示す。
ここで、「子宮内膜症若しくは子宮腺筋症が疑われる月経困難症」とは、限定はされないが、子宮内膜症又は子宮腺筋症に過去に罹患しており、症状が再燃する恐れのある対象でもよいし、あるいは、子宮内膜症若しくは子宮腺筋症との例えば病理所見による確定診断などが医師等によって下される前の、子宮内膜症若しくは子宮腺筋症を発症していると疑われている対象であってもよい。
一実施態様において、本開示におけるIL-8シグナル阻害剤が子宮内膜症若しくは子宮腺筋症を伴う月経困難症、又は子宮内膜症若しくは子宮腺筋症が疑われる月経困難症の治療又は予防に用いられる場合、上述の通り、IL-8シグナル阻害剤は性周期への影響を与えず、ホルモン剤との併用によらずとも単剤で子宮内膜症や子宮腺筋症を治療又は予防することができるので有利である。
また、本実施例に記載のとおり、本発明者らは、IL-8シグナル阻害剤が、子宮内膜症、子宮腺筋症に加えて、癒着をも治療又は予防できることを実証又は想到した。月経困難症は腹痛などの疼痛によっても特徴付けられるところ、子宮内膜症における癒着のみならず、それ以外の症状又は疾患による癒着であっても組織あるいは臓器が接着等することによって患者に疼痛を引き起こすことが当然に考えられる。例えば骨盤内における癒着では生殖附属器同士の癒着と痛みは相関しないものの、ダグラス窩の癒着と痛みは相関すること(Porpora et al, The Journal of the American Association of Gynecologic Laparoscopists 1999; 6:429-434.)や、臓器同士が癒着することで引きつって疼痛が生じることが知られている。また、子宮内膜症では異所性の神経線維およびNGFの産生が疼痛および疼痛増悪の原因となることが知られている(Anaf et al, Hum Reprod 2000; 15:1744-1750., Berkley et al, Proc Natl Acad Sci U S A 2004; 101:11094-11098. Odagiri et al, Fertil Steril 2009; 92:1525-1531.)。また子宮内膜症周囲にある平滑筋が発達したinterstitiumが収縮時の痛みを増強することを示唆する報告もある(Odagiri et al, Fertil Steril 2009; 92:1525-1531.)。本実施例の抗IL-8抗体の投薬によって子宮内膜症組織のinterstitiumが減少していることが確認されたことに加えて、異所性神経の減少が期待できることから、疼痛改善が期待できる。
一実施態様において、本開示におけるIL-8シグナル阻害剤は、子宮内膜症又は子宮腺筋症における疼痛(慢性疼痛、月経時の疼痛など)、月経困難症における疼痛、又は、癒着、線維化若しくは炎症による疼痛を治療又は予防することができる。ここで、当該慢性疼痛としては、限定を意図しないが、慢性下腹部痛又は慢性骨盤痛が挙げられる。
本開示において、このような線維化を伴う疾患や状態を、その線維化の態様を捉えて、包括的に「線維化疾患」という。
カニクイザルin vivoモデルにおいて、IL-8シグナル阻害剤は免疫細胞の浸潤を抑制したこと、コラーゲン豊富なinterstitium部位が減少したこと(本実施例参照)に加えて、in vitroアッセイにおいて、IL-8シグナル阻害剤はMCP-1(マクロファージ遊走因子及び線維化促進因子として知られる。)などを産生する好中球の遊走阻害作用を示したことから、IL-8シグナル阻害剤は有望な線維化治療剤又は予防剤であることは上述の通りである。
加えて、実施例に示されるように、in vitro試験において様々な抗IL-8抗体が、線維化促進因子であるConnecting tissue growth factor(CTGF)の発現上昇を抑制した。この結果は、抗IL-8抗体等のIL-8シグナル阻害剤が線維化疾患に対する有望な治療剤又は予防剤であることを裏付けるものである。
A型及びB型ウイルス性肝炎、アルコール性肝炎、原発性胆汁性肝硬変など慢性肝疾患においてIL-8が上昇すること(Mediators Inflamm. 2015;2015:276850)、慢性肝疾患で血中IL-8濃度が高く病態進行した患者では特に高いこと、受容体であるCXCR1の発現が単球で高いこと、IL-8が肝臓での炎症を強く亢進すること、これらの結果からIL-8の線維化進行への寄与が示唆された(PLoS One. 2011;6(6):e21381)と報告されている。
またNASH患者の血漿中IL-8濃度は線維化を伴った患者で有意に高かった(Gastroenterology. 2015 Sep;149(3):635-48.e14)ことから、IL-8と線維化との関連が示唆された。一実施態様において、本開示における肝線維症はIL-8シグナルに応答性を示す。
腎線維芽細胞がIL-1を介してIL-8の産生を増強した(Kidney Int. 1995 Mar;47(3):845-54.)、MCP-1シグナルを抑制することで腎線維症が改善した(Kitagawa et al, Am J Pathol. 2004 Jul;165(1):237-46.)、ヒト線維細胞を用いて、糖尿病状態における活性化ならびに線維化機序を検討したところ、ヒト線維細胞は高血糖およびMCP-1/CCR2を介して糖尿病性腎症の進展に関与することが示唆された(Clin Exp Nephrol. 2013 Dec;17(6):793-804)などの報告がある。IL-8シグナル阻害剤はMCP-1を産生する好中球が病変部に遊走することを阻害する(本実施例参照)ので、MCP-1を介して腎線維症の進展を抑制、あるいは改善することが想起される。一実施態様において、本開示における腎線維症はIL-8シグナルに応答性を示す。
ある実施態様において、免疫細胞としては、例えば、好中球、マクロファージ、単球、ヘルパーT細胞、キラーT細胞、樹状細胞、NK細胞などが挙げられ、限定は意図しないが、好中球、マクロファージ、単球が好ましい。免疫細胞のIL-8関連疾患の病変部への浸潤という場合、IL-8関連疾患は上述のいずれの疾患であってもよく、例えば、子宮内膜症、子宮腺筋症、月経困難症、癒着、線維化疾患などが挙げられ、このようなIL-8関連疾患の病変部(疾患部位)又はその近傍へ免疫細胞が浸潤又は遊走等することが企図されよう。また、免疫細胞のIL-8関連疾患の周辺環境への浸潤という場合、例えば子宮内膜症であれば、腹腔内や腹水への浸潤又は遊走等が企図されるであろうし、例えば肺線維症であれば、胸腔内や胸水への浸潤又は遊走等が企図されるであろうことが当業者には理解できる。
細胞内に取り込まれたIgG抗体は、エンドソーム内の酸性pHの条件下において、FcRnに結合することにより血漿中に戻されることが知られている。そのため、IgG抗体は一般的に、FcRnに結合しないタンパク質に比べて、長い血漿中半減期を有している。その性質を利用し、抗体のFc領域にアミノ酸改変を導入することで、酸性pH条件におけるFcRnへの結合能(結合親和性)を増大し、抗体の血漿中滞留性を向上させる方法が既に知られている。具体的には、M252Y/S254T/T256E (YTE) 改変 (J Biol Chem 2006 281:23514-23524.)や、M428L/N434S (LS) 改変 (Nat Biotechnol, 2010 28:157-159.)、N434H改変(Clinical Pharmacology & Therapeutics (2011) 89(2):283-290.)などのアミノ酸改変により、酸性pH条件におけるFcRnへの結合能を増大し、抗体の血漿中滞留性を向上させる方法が知られている。
酸性pH条件におけるFcRn結合能を増大させたFc領域改変体として、公知の改変であるYTE、LS及び N434Hと、新たに見出されたいくつかのFc領域改変体(F1847m、F1848m、F1886m、F1889m、F1927m、F1168m)を、以下に示すように作製した。
抗ヒトIL-6レセプター抗体であるFv4-IgG1の重鎖(VH3-IgG1m)のFc領域に、アミノ酸改変を導入した遺伝子を、参考実施例1の方法で作製した。これらの重鎖を用いて、参考実施例2の方法により、以下の抗体を作製した。
重鎖としてVH3-IgG1m(配列番号:2)、軽鎖としてVL3-CK(配列番号:110)からなるFv4-IgG1、
重鎖としてVH3-YTE(配列番号:3)、軽鎖としてVL3-CKからなるFv4-YTE、
重鎖としてVH3-LS(配列番号:4)、軽鎖としてVL3-CKからなるFv4-LS、
重鎖としてVH3-N434H(配列番号:5)、軽鎖としてVL3-CKからなるFv4-N434H、
重鎖としてVH3-F1847m(配列番号:6)、軽鎖としてVL3-CKからなるFv4-F1847m、
重鎖としてVH3-F1848m(配列番号:7)、軽鎖としてVL3-CKからなるFv4-F1848m、
重鎖としてVH3-F1886m(配列番号:8)、軽鎖としてVL3-CKからなるFv4-F1886m、
重鎖としてVH3-F1889m(配列番号:9)、軽鎖としてVL3-CKからなるFv4-F1889m、
重鎖としてVH3-F1927m(配列番号:10)、軽鎖としてVL3-CKからなるFv4-F1927m、
重鎖としてVH3-F1168m(配列番号:11)、軽鎖としてVL3-CKからなるFv4-F1168m
重鎖としてVH3-IgG1mあるいは上記の改変体を含み、軽鎖としてL(WT)(配列番号:1)を含む抗体が参考実施例2に示した方法で作製され、下記のようにヒトFcRnに対する結合活性が評価された。
Biacore T100(GE Healthcare)を用いて、ヒトFcRnと各抗体との速度論的解析を行った。センサーチップCM4(GE Healthcare)上にアミンカップリング法でプロテインL(ACTIGEN)を適当量固定化し、そこへ目的の抗体を捕捉させた。次に、FcRn希釈液とランニングバッファー(参照溶液として)とをインジェクトし、センサーチップ上に捕捉させた抗体にヒトFcRnを相互作用させた。ランニングバッファーには50 mMリン酸ナトリウム、150 mM NaCl、0.05% (w/v) Tween20、pH6.0を用い、FcRnの希釈にもそれぞれのバッファーが使用された。センサーチップの再生には10 mMグリシン-HCl, pH1.5が用いられた。測定は全て25 ℃で実施された。測定で得られたセンサーグラムから算出されたカイネティクスパラメーターである結合速度定数 ka (1/Ms)、及び解離速度定数 kd (1/s)をもとに各抗体のヒトFcRnに対する KD (M) が算出された。各パラメーターの算出には Biacore T100 Evaluation Software(GE Healthcare)が用いられた。
その結果を表2に示す。
抗医薬品抗体(ADA)は治療用抗体の効果及び薬物動態に影響を及ぼし、時に重篤な副作用をもたらすことがあるため、臨床における治療用抗体の有用性と薬効はADAの産生によって制限され得る。治療用抗体の免疫原性には、多くの要因が影響を及ぼすが、エフェクターT細胞エピトープの存在がその要因の1つである。加えて、治療用抗体の投与前から患者が有しているADA(「Pre-existing ADA」ともいう)の存在もまた、同様に問題があると考えられる。特に、関節リウマチ(RA)などの自己免疫疾患の患者に対する治療用抗体の場合、ヒトIgGに対する自己抗体であるリウマトイド因子(RF)が「Pre-existing ADA」の問題となり得る。最近、N434H(Asn434His)変異を有するヒト化抗CD4 IgG1抗体が顕著なリウマトイド因子結合を誘発することが報告された(Clin Pharmacol Ther. 2011 Feb;89(2):283-90)。詳細な研究により、ヒトIgG1におけるN434H変異が、親ヒトIgG1と比較して抗体のFc領域に対するリウマトイド因子の結合を増大することが確認された。
一方で、同じくWO2013/046704の中で、FcRnに対する結合性には影響せずにリウマトイド因子への結合性を選択的に抑制するいくつかのアミノ酸改変が例示され、その中に2アミノ酸変異の組み合わせであるQ438R/S440E、Q438R/S440D、Q438K/S440E又はQ438K/S440Dが示されている。そこで、今回新たに作製された、酸性pH条件における結合能が増大されたFcに対しても、Q438R/S440Eを導入することでリウマトイド因子に対する結合性を低下することが可能かどうかを検証した。
リウマトイド因子に対する結合アッセイは、30名のRA患者の個々の血清(Proteogenex)を用いて、pH 7.4における電気化学発光(ECL)により行った。50倍希釈した血清試料、ビオチン標識した各試験抗体(1μg/mL)、及びSULFO-TAG NHS Ester(Meso Scale Discovery)標識した各試験抗体(1μg/mL)を混合し、室温で3時間インキュベートした。その後、混合物をStreptavidinでコーティングされたMULTI-ARRAY 96ウェルプレート(Meso Scale Discovery)に加え、プレートを室温で2時間インキュベートし、洗浄した。Read Buffer T(×4)(Meso Scale Discovery)を各ウェルに加えた後、直ちにプレートをSECTOR imager 2400 Reader(Meso Scale Discovery)にセットし、化学発光を測定した。
また、図11は、それぞれの改変体について、30名のRA患者の血液におけるリウマトイド因子への結合性の平均値を示したものである。6種類の新規の改変体はいずれも、3種の既存の改変体(YTE、LS、N434H)よりも低い結合性を示し、さらには天然型ヒトIgG1と比較しても、より低いリウマトイド因子への結合性を示した。以上のことから、関節リウマチなどの自己免疫疾患等について、FcRnに対する結合能が改善された治療用抗体の臨床開発を考慮する場合に、既存のFc領域改変体において懸念されるリウマトイド因子に関連するリスクは、今回新規に作製されたFc領域改変体においては抑制されており、より安全に使用可能であることが考えられる。
実施例3において、リウマトイド因子への結合が抑制されていることが確認された新規Fc領域改変体を含む抗体を用いて、カニクイザルにおける血漿中滞留性の改善効果を以下の方法で評価した。
以下の抗ヒトIgE抗体を作製した。
重鎖としてOHBH-IgG1(配列番号:12)、軽鎖としてOHBL-CK(配列番号:13)からなるOHB-IgG1、
重鎖としてOHBH-LS(配列番号:14)、軽鎖としてOHBL-CKからなるOHB-LS、
重鎖としてOHBH-N434A(配列番号:15)、軽鎖としてOHBL-CKからなるOHB-N434A
重鎖としてOHBH-F1847m(配列番号:16)、軽鎖としてOHBL-CKからなるOHB-F1847m、
重鎖としてOHBH-F1848m(配列番号:17)、軽鎖としてOHBL-CKからなるOHB-F1848m、
重鎖としてOHBH-F1886m(配列番号:18)、軽鎖としてOHBL-CKからなるOHB-F1886m、
重鎖としてOHBH-F1889m(配列番号:19)、軽鎖としてOHBL-CKからなるOHB-F1889m、
重鎖としてOHBH-F1927m(配列番号:20)、軽鎖としてOHBL-CKからなるOHB-F1927m
カニクイザルに抗ヒトIgE抗体を投与した後の、血漿中抗ヒトIgE抗体の体内動態を評価した。抗ヒトIgE抗体の溶液を2mg/kgで単回静脈内投与した。投与後5分、(2時間)、7時間、1日、2日、3日、(4日)、7日、14日、21日、28日、35日、42日、49日、56日で採血を行った。採取した血液はただちに4℃、15,000 rpmで5分間遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで、-80℃以下に設定された冷凍庫に保存した。抗ヒトIgE抗体としては、OHB-IgG1、OHB-LS、OHB-N434A、OHB-F1847m、OHB-F1848m、OHB-F1886m、OHB-F1889m、OHB-F1927mの8種類を使用した。
カニクイザル血漿中の抗ヒトIgE抗体濃度はELISA法にて測定した。まず抗ヒトIgG kappa chain抗体(Antibody Solution)をNunc-Immuno Plate, MaxiSoup (Nalge nunc International)に分注し、4℃で1晩静置し抗ヒトIgG kappa chain抗体固相化プレートを作製した。血漿中濃度として640、320、160、80、40、20、10 ng/mLの検量線試料と100倍以上希釈したカニクイザル血漿測定試料を調製した。これら検量線試料及び血漿測定試料にはカニクイザルIgE (社内調製品)が1 μg/mlの濃度で添加されるように作製された。その後、抗ヒトIgG kappa chain抗体固相化プレートに分注し室温で2時間静置した。その後HRP-抗ヒトIgG gamma chain抗体(Southern Biotech)を分注し室温で1時間静置した。その後、TMB Chromogen Solution (Life Technologies)を基質として用い発色反応を行い、1N-Sulfuric acid(Wako)で反応停止後、マイクロプレートリーダーにて450 nmの吸光度を測定した。サル血漿中抗ヒトIgE抗体濃度は検量線の吸光度から解析ソフトウェアSOFTmax PRO(Molecular Devices)を用いて算出した。測定したサル血漿中抗ヒトIgE抗体の濃度推移は図12に示した。また測定したサル血漿中抗ヒトIgE抗体の濃度推移からPhoenix WinNonlin Ver.6.2(Pharsight Corporation)を用いてモーメント解析により消失クリアランスを算出した。算出した薬物動態学的パラメータは表3に示した。血漿中抗投与検体抗体が陽性を示した個体はサル血漿中抗ヒトIgE抗体の濃度推移及びクリアランスの算出から除外した。
サルの血漿中抗投与検体抗体は電気化学発光法にて測定した。SULFO-TAG NHS Ester(Meso Scale Discovery)でルテニウム化した投与検体、EZ-Link Micro Sulfo-NHS-Biotinylation Kit(Pierce)でビオチン化した投与検体及びカニクイザル血漿測定試料を等量混合し、4℃で1晩静置した。MULTI-ARRAY 96-well Streptavidin Gold Plate (Meso Scale Discovery)に試料を添加後室温で2時間反応させ洗浄後、Read Buffer T(×4)(Meso Scale Discovery)を分注し、ただちにSECTOR Imager 2400(Meso Scale Discovery)で測定を行った。
酸性pHにおけるFcRn結合増大のためのFc領域改変体として、WO2013/046704に記載のFc領域改変体であるF1718と、今回新規に見出されたFc領域改変体であるF1848mを比較するために、以下のような実験を実施した。
重鎖としてVH3-IgG1、軽鎖としてVL3-CKからなるFv4-IgG1、
重鎖としてVH3-F1718(配列番号:21)、軽鎖としてVL3-CKからなるFv4-F1718
マウス血漿中の抗ヒトIL-6レセプター抗体の濃度はELISA法にて測定された。まず、Anti-Human IgG(gamma-chain specific)F(ab')2 Fragment of Antibody(SIGMA)をNunc-Immuno Plate, MaxiSoup (Nalge nunc International)に分注し、4℃で1晩静置することによってAnti-Human IgG固相化プレートが作製された。血漿中濃度として0.8、0.4、0.2、0.1、0.05、0.025、0.0125μg/mLの抗ヒトIL-6レセプター抗体を含む検量線試料と100倍以上希釈されたマウス血漿測定試料が調製された。これらの検量線試料及び血漿測定試料100μLに20 ng/mLの可溶型ヒトIL-6レセプターが200μL加えられた混合液を、室温で1時間静置させた。その後、当該混合液が各ウェルに分注されたAnti-Human IgG固相化プレートをさらに室温で1時間静置させた。その後、Biotinylated Anti-human IL-6 R Antibody(R&D)と室温で1時間反応させ、さらにStreptavidin-PolyHRP80(Stereospecific Detection Technologies)を室温で1時間反応させた反応液の発色反応が、TMB One Component HRP Microwell Substrate(BioFX Laboratories)を基質として用いて行われた。1N-Sulfuric acid(Showa Chemical)を添加することによって反応が停止された各ウェルの反応液の450 nmの吸光度が、マイクロプレートリーダーにて測定された。マウス血漿中の抗体濃度は検量線の吸光度から解析ソフトウェアSOFTmax PRO(Molecular Devices)を用いて算出された。
ここで、WO2013/046704に記載のF1718には、Fc領域にN434Y/Y436V/Q438R/ S440Eという4か所の変異が導入されている。一方で、今回新たに見出されたF1848mには、N434A/Y436V/Q438R/S440Eという4か所の変異が導入されている。これら二種類のFcに導入されているアミノ酸変異の違いは、EUナンバリング434番に導入されているアミノ酸変異が、F1718はY(チロシン)であり、F1848mはA(アラニン)であるという点のみである。実施例(3-2)において、F1848mは天然型IgG1の場合に比べて血漿中滞留性の向上が認められた一方で、F1718は血漿中滞留性の向上が認められなかった。このことから、特に限定されるわけではないが、血漿中滞留性の向上のために用いられるアミノ酸変異として、434番目に導入されるアミノ酸変異としては、A(アラニン)が好ましいことが示唆される。
(4-1)ヒト化抗ヒトIL-8抗体hWS-4の作製
US6245894 (WO1996/002576)の中で開示されているヒト化抗IL-8抗体は、ヒトIL-8(以下、hIL-8とも表記する)と結合することにより、その生理作用を遮断する。ヒト化抗IL-8抗体は、US6245894に開示されている重鎖及び軽鎖の可変領域配列と、任意のヒト抗体の定常領域配列を組み合わせて作成可能である。ヒト抗体の定常領域配列としては、特に限定はされないが、重鎖定常領域として天然型ヒトIgG1配列又は天然型ヒトIgG4配列、軽鎖定常領域配列として天然型ヒトKappa配列を用いることができる。
ここで、US6245894の中で開示されているヒト化IL-8抗体のうち、重鎖可変領域RVHgと、重鎖定常領域として天然型ヒト抗IgG1配列を組み合わせたhWS4H-IgG1(配列番号:39)の遺伝子を、参考実施例1の方法で作成した。更に、軽鎖可変領域RVLaと、軽鎖定常領域として天然型ヒトKappa配列を組み合わせたhWS4L-k0MT(配列番号:40)の遺伝子を、参考実施例1の方法で作成した。上記の重鎖及び軽鎖を組み合わせた抗体を作製し、ヒト化WS-4抗体(以下、hWS-4)とした。
hWS-4で使用されているFRとは異なるヒトコンセンサスフレームワーク配列を用いて、新たなヒト化抗体を作製した。
具体的には、重鎖のFR1としてVH3-23とVH3-64のハイブリッド配列、FR2としてVH3-15やVH3-49などに見られる配列、FR3としてVH3-72に見られる配列(ただしKabatナンバリング82aを除く)、FR4としてはJH1などに見られる配列を用いて、これらをhWS-4重鎖のCDR配列と連結し、新規のヒト化抗体の重鎖であるHr9-IgG1(配列番号:41)を作製した。
次に、重鎖としてhWS4H-IgG1、軽鎖としてhWS4L-k0MTを有するhWS-4と、重鎖としてHr9-IgG1、軽鎖としてhWS4L-k0MTを有するHr9の2種類の抗体を作製した。なお、本開示において、特に軽鎖を明記したい場合においては、Hr9(その重鎖可変領域の配列を配列番号:33に示す。)はHr9/hWS4Lとも表記される。抗体は、FreeStyle 293F細胞(Invitrogen)を用いて、製品に添付のプロトコールに従い発現させた。培養上清からの抗体の精製は参考実施例2の方法で行った。その結果、表4に示す量の抗体が取得された。驚くべきことに、Hr9の発現量は、hWS-4の発現量に比べて約8倍であった。
hWS-4及びHr9のヒトIL-8への結合親和性を、Biacore T200(GE Healthcare)を用いて以下の通り測定した。
ランニングバッファーは、0.05% tween20, 20 mM ACES, 150 mM NaCl(pH 7.4)の組成のものを用いた。
センサーチップCM4(GE Healthcare)上にアミンカップリング法でプロテインA/G(PEIRCE)を適当量固定化し、そこへ目的の抗体を捕捉させた。次に、ヒトIL-8希釈液とランニングバッファー(参照溶液として)とをインジェクトし、センサーチップ上に捕捉させた抗体にヒトIL-8を相互作用させた。なお、ランニングバッファーは上記の組成の溶液が用いられ、ヒトIL-8の希釈にも当バッファーが使用された。センサーチップの再生には10 mMグリシン-HCl, pH 1.5が用いられた。測定は全て37 ℃で実施された。測定で得られたセンサーグラムから算出されたカイネティクスパラメーターである結合速度定数 kon (1/Ms)、及び解離速度定数 koff (1/s)をもとに各抗体のヒトIL-8に対する KD (M) が算出された。各パラメーターの算出には Biacore T200 Evaluation Software(GE Healthcare)が用いられた。
結果を表5に示す。hWS-4とHr9は、ヒトIL-8に対して同等の結合親和性を有することが確認された。
(5-1)pH依存性付与のためのHr9改変抗体の作製
実施例4で得られたHr9に対してpH依存的IL-8結合能を付与することを目的として、検討を行った。
特定の理論に拘束されるわけではないが、IL-8に対するpH依存的結合能を有する抗体は、生体内において次のような挙動を示すと考え得る。生体に投与された当該抗体は、中性pHに維持されている環境(例えば血漿中)において、IL-8に対して強く結合し、その機能を遮断することができる。このようなIL-8と抗体の複合体の一部は、細胞膜との非特異的な相互作用(ピノサイトーシス)によって細胞内へと取り込まれる(以下、非特異的な取り込みという)。エンドソーム内の酸性pHの条件下においては、前記抗体のIL-8に対する結合親和性が弱まるため、前記抗体はIL-8を解離する。その後、IL-8を解離した前記抗体は、FcRnを介して細胞外に戻ってくることができる。このように細胞外(血漿中)に戻ってきた前記抗体は、再度別のIL-8に結合し、その機能を遮断することが可能である。IL-8に対してpH依存的結合能を有する抗体は、上記のようなメカニズムによっても、IL-8に対して複数回結合することが可能になると考え得る。
一方、前記抗体のような性質を有さない抗体の場合、1分子の抗体は1度だけ抗原を中和することが可能であり、抗原を複数回中和することはできない。通常、IgG抗体は2つのFabを有するため、1つの抗体分子は2分子のIL-8を中和できる。一方、IL-8に複数回結合することができる抗体は、生体内に滞留している限り、何度でもIL-8に結合することができる。例えば、投与されてから消失するまでの間に、細胞内に10回取り込まれたpH依存的IL-8結合抗体は、1分子でも最大で20分子のIL-8を中和することが可能である。そのため、複数回IL-8に結合できる抗体は、より少ない抗体量であっても、多くのIL-8を中和することができるという利点を有する。別の観点では、複数回IL-8に結合できる抗体は、同じ量の抗体を投与した場合に、より長期間にわたってIL-8を中和することが可能な状態を維持できるという利点を有する。また、さらに別の観点からは、複数回IL-8に結合できる抗体は、同じ量の抗体を投与した場合に、より強くIL-8の生物学的活性を遮断することができるという利点を有する。
これらの利点を実現するために、複数回IL-8に結合できる抗体を創製することを目的として、Hr9-IgG1及びWS4L-k0MTの可変領域に対して、ヒスチジンを中心としたアミノ酸改変を導入した。具体的には、表6に示す改変体を参考実施例1及び2の方法で作製した。
なお、表6において示されている「Y97H」のような表記は、Kabatナンバリングにより定義される変異導入箇所と、変異導入前のアミノ酸、変異導入後のアミノ酸を表したものである。具体的には、「Y97H」と表記した場合、Kabatナンバリング97番のアミノ酸残基を、Y(チロシン)からH(ヒスチジン)に置換したことを表している。更に、複数のアミノ酸置換を組み合わせて導入した場合には、「N50H/L54H」のように記載する。
実施例5-1で作成された抗体のヒトIL-8結合親和性を、Biacore T200(GE Healthcare)を用いて以下の通り測定した。 ランニングバッファーは以下の2種を用いた。
・0.05% tween20, 20 mM ACES, 150 mM NaCl, pH 7.4
・0.05% tween20, 20 mM ACES, 150 mM NaCl, pH 5.8
センサーチップCM4(GE Healthcare)上にアミンカップリング法でプロテインA/G(PIERCE)を適当量固定化し、そこへ目的の抗体を捕捉させた。次に、ヒトIL-8希釈液とランニングバッファー(参照溶液として)とをインジェクトし、センサーチップ上に捕捉させた抗体にヒトIL-8を相互作用させた。なお、ランニングバッファーは上記のいずれかが用いられ、ヒトIL-8の希釈にもそれぞれのバッファーが使用された。センサーチップの再生には10 mMグリシン-HCl, pH 1.5が用いられた。測定は全て37 ℃で実施された。測定で得られたセンサーグラムから算出されたカイネティクスパラメーターである結合速度定数 kon (1/Ms)、及び解離速度定数 koff (1/s)をもとに各抗体のヒトIL-8に対する KD (M) が算出された。各パラメーターの算出には Biacore T200 Evaluation Software(GE Healthcare)が用いられた。
結果を表7に示す。まず、軽鎖にL54H改変を含むHr9/L16は、中性pH(pH7.4)におけるヒトIL-8結合親和性がHr9よりやや増強している一方で、酸性pH(pH5.8)におけるヒトIL-8結合親和性が低下していた。一方で、重鎖にY97H改変を含むH89と各種軽鎖とを組み合わせた抗IL-8抗体(H89/WS4L、H89/L12、及びH89/L16)はいずれも、酸性pHにおけるヒトIL-8結合親和性は低下していると同時に、中性pHにおけるヒトIL-8結合親和性も低下していた。
そこで次に、5-2で見いだされた有望な改変の組み合わせと、新たなアミノ酸変異の探索を行い、その結果、以下の組み合わせが見いだされた。
その結果を表7に併せて記した。重鎖としてH89-IgG1(配列番号:42)、軽鎖としてL63-k0MT(配列番号:43)を有するH89/L63は、中性pH(pH7.4)におけるヒトIL-8結合親和性がHr9と同等のまま、酸性pH(pH5.8)におけるヒトIL-8結合親和性が低下していた。具体的には、H89/L63は、pH5.8におけるkoff(解離速度定数)及びKD(解離定数)の両方が、Hr9よりも大きくなっていた。このことは、エンドソーム内の酸性pH条件下において、H89/L63はヒトIL-8を解離しやすい性質を有していることを意味している。
更に、驚くべきことに、重鎖としてH89-IgG1、軽鎖としてL118-k0MT(配列番号:44)を有するH89/L118は、中性pH条件におけるヒトIL-8結合親和性(KD)はHr9よりも増強しているのに対し、酸性pH条件におけるヒトIL-8結合親和性(KD)はHr9よりも減弱していた。特に限定はされないが、一般的に抗原に複数回結合できる抗体を医薬品として使用する際には、pH依存的抗原結合抗体は、中性pH条件下(例えば、血漿中)で抗原を強く中和することが可能なように、強い結合親和性を持つ(KDが小さい)ことが好ましい。一方で、酸性pH条件下(例えば、エンドソーム内)で、抗原を速やかに解離することが可能なように、解離速度定数(koff)が大きいこと、及び/又は弱い結合親和性を持つ(KDが大きい)ことが好ましい。H89/L118は、これら中性pHと酸性pHのいずれにおいても、Hr9と比較して好ましい性質を獲得していた。
即ち、Hr9の重鎖に対してY97H、軽鎖に対してN50H/L54H/Q89Kといった有用なアミノ酸改変が見いだされた。特に限定されることはないが、これらの中から選ばれるアミノ酸改変を単独、あるいは複数組み合わせて導入することによっても、医薬品として優れたpH依存的IL-8結合抗体が作製可能であることが示された。
特定の理論に拘束されることはないが、pH依存的抗原結合抗体を医薬品として利用する際に重要な点は、生体内に投与された抗体が、エンドソーム内において抗原を解離することができるかどうかであると考えられる。そのためには酸性pH条件下において、十分に結合が弱い(解離定数;KDが大きい)こと、又は十分に解離速度が速い(解離速度定数;koffが大きい)こと、が重要であると考えられる。そこで、Biacoreにより得られたH89/L118のKD又はkoffが、生体内のエンドソーム内において抗原を解離するために十分なものかどうかを、次に示す実験で検証した。
抗体による、ヒトIL-8の消失速度への効果をマウスにおいて確認する方法としては、特に限定はされないが、例えば、抗体をヒトIL-8と混合した状態でマウスに投与し、その後のマウス血漿中からのヒトIL-8の消失速度を比較する方法がある。
ここで、マウスのPK試験に用いるための参照抗体は、中性pH及び酸性pHの条件下において、いずれも十分に強い結合親和性を持つことが望ましい。そこで、Hr9に対して高親和性を付与する改変を探索した結果、重鎖としてH998-IgG1(配列番号:45)、軽鎖としてL63-k0MTを有するH998/L63が創製された。
H998/L63を用いて、ヒトIL-8の結合親和性を実施例5-2と同様の方法で評価した。結果として得られたセンサーグラムを図14に示す。
H998/L63は、中性pHと酸性pHのいずれの条件においても、著しく解離速度が遅く、Hr9よりも強いIL-8結合親和性を有することが示された。ただし、Biacoreの装置上の限界から、このように解離速度が遅いタンパク質-タンパク質間相互作用の場合、解離速度定数(koff)、解離定数(KD)などの解析値を正確に算出することができないことが知られている。H998/L63においても、正確な解析値を取得することができなかったため、ここでは解析値を示していない。しかしながら、当実験の結果から、H998/L63が中性と酸性のいずれのpHにおいても非常に強い結合親和性を有しており、マウスPK試験における比較対象として用いる抗体として適切であることが確認された。
(7-1)H89/L118を用いたマウスPK試験
実施例5で作成したH89/L118と、実施例6で作成したH998/L63を用いて、in vivoでのヒトIL-8消失速度の評価を実施した。
マウス(C57BL/6J、Charles river)に、ヒトIL-8と、抗ヒトIL-8抗体を同時に投与した後のヒトIL-8の体内動態を評価した。ヒトIL-8、抗ヒトIL-8抗体の混合溶液(それぞれ10 μg/mL, 200 μg/mL)を尾静脈に10 mL/kgで単回投与した。このとき、ヒトIL-8に対して抗ヒトIL-8抗体は十分量過剰に存在することから、ヒトIL-8はほぼ全て抗体に結合していると考えられる。投与5分後、2時間後、4時間後、7時間後、1日後、2日後、3日後、7日後、14日後、21日後、28日後に血液を採取した。採取した血液は直ちに4℃、15,000 rpmで15分遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで-20℃以下に設定された冷凍庫に保存した。
マウス血漿中のヒトIL-8濃度は電気化学発光法にて測定された。まず、マウスIgGの定常領域を有する抗ヒトIL-8抗体(社内調製品)を、MULTI-ARRAY 96-well Plate(Meso Scale Discovery)に分注し、室温で1時間静置した後に、5%BSA(w/v)を含有したPBS-Tween溶液を用いて室温で2時間ブロッキングすることによって抗ヒトIL-8抗体固相化プレートが作成された。血漿中濃度として275、91.7、30.6、10.2、3.40、1.13、0.377 ng/mLのヒトIL-8を含む検量線試料と25倍以上に希釈されたマウス血漿測定試料が調製され、hWS-4と混合させてから37℃で1晩反応させた後、抗ヒトIL-8抗体固相化プレートの各ウェルに50 μLで分注されてから、室温で1時間撹拌させた。hWS-4の終濃度は25 μg/mLとなるよう調製された。その後、Biotin Mouse Anti-Human Ig k Light Chain (BD Pharmingen)を室温で1時間反応させ、さらにSULFO-TAG Labeled Streptavidin(Meso Scale Discovery)を室温で1時間反応させた後、Read Buffer T(×1)(Meso Scale Discovery)を分注し、ただちにSECTOR Imager 2400(Meso Scale Discovery)で測定を行った。ヒトIL-8濃度は検量線のレスポンスから解析ソフトウェアSOFT Max PRO(Molecular Devices)を用いて算出した。
結果として得られた、血漿中ヒトIL-8濃度のデータを図15に、またマウス血漿中からのヒトIL-8クリアランス(CL)の数値を表8に示す。
特定の理論に拘束されるわけではないが、今回得られたデータから、次のように考察することも可能である。抗体と同時に投与されたヒトIL-8は、血漿中において大部分が抗体と結合し、複合体状態で存在する。H998/L63と結合したヒトIL-8は、酸性pH条件下であるエンドソーム内においても、強い親和性により抗体と結合した状態で存在する。その後、H998/L63は、ヒトIL-8との複合体を形成したままの状態でFcRnを介して血漿中に戻されるため、その際にヒトIL-8も同時に血漿中に戻されることになる。そのため、細胞内に取り込まれたヒトIL-8のうちの大部分は、再び血漿中へと戻ってくることになる。つまり、ヒトIL-8の血漿中からの消失速度は、H998/L63と同時に投与することにより、顕著に低下する。一方で、先述の通り、pH依存的IL-8結合抗体であるH89/L118と複合体を形成した状態で細胞内に取り込まれたヒトIL-8は、エンドソーム内の酸性pH条件下において、抗体から解離する。抗体から解離したヒトIL-8は、ライソソームに移行して分解される。そのため、pH依存的IL-8結合抗体は、H998/L63のような、酸性pH及び中性pHで共に強い結合親和性を有するIL-8結合抗体と比較して、顕著にヒトIL-8の消失を早めることが可能である。
次に、H89/L118の投与量を変化させた場合の影響を検証する実験を、以下のように実施した。マウス(C57BL/6J、Charles river)に、ヒトIL-8と、H89/L118(2 mg/kgあるいは8 mg/kg)を同時に投与した後のヒトIL-8の体内動態を評価した。ヒトIL-8(2.5 μg/mL)、抗ヒトIL-8抗体(200 μg/mL又は800 μg/mL)の混合溶液を尾静脈に10 mL/kgで単回投与した。このとき、ヒトIL-8に対して抗ヒトIL-8抗体は十分量過剰に存在することから、ヒトIL-8はほぼ全て抗体に結合していると考えられる。投与5分後、7時間後、1日後、2日後、3日後、7日後、14日後、21日後、28日後に血液を採取した。採取した血液は直ちに4℃、15,000 rpmで15分遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで-20℃以下に設定された冷凍庫に保存した。
マウス血漿中のヒトIL-8濃度測定は、実施例7-2と同様の方法で実施した。結果として得られた、血漿中ヒトIL-8濃度のデータを図16に、またマウス血漿中からのヒトIL-8クリアランス(CL)の数値を表9に示す。
以下、理論に拘束されることは意図しないが前記の結果をもたらす可能性がある要因の一つを推察した内容を記述する。
エンドソーム内からFcRnを介して血漿中に戻される抗体のうち、ヒトIL-8が結合しているものの割合は低い方が好ましい。一方、エンドソーム内に存在するヒトIL-8に着目すると、こちらは抗体と結合していないフリー型の割合が高いことが望ましい。pH依存的IL-8結合能を有さない抗体と一緒に投与された場合は、エンドソーム内において、大部分の(100%に近い)ヒトIL-8は抗体と複合体を形成した状態で存在すると考えられ、フリー型は少ない(0%に近い)と考えられる。一方で、pH依存的IL-8結合抗体(例えばH89/L118)と一緒に投与された場合は、エンドソーム内において、ある程度の割合のヒトIL-8はフリー型として存在しているはずである。この際のフリー型の割合は、仮想的にではあるが、以下のように理解することも可能である。
[エンドソーム内におけるフリー型ヒトIL-8の割合(%)]=[エンドソーム内のフリー型ヒトIL-8濃度]÷[エンドソーム内の全ヒトIL-8濃度]×100
上式のように理解される、エンドソーム内におけるフリー型ヒトIL-8の割合は、より高い方が望ましく、例えば0%よりは20%が好ましく、20%よりは40%が好ましく、40%よりは60%が好ましく、60%よりは80%が好ましく、80%よりは100%が好ましい。
上記のようなエンドソーム内におけるフリー型ヒトIL-8の割合と、酸性pHにおけるヒトIL-8に対する結合親和性(KD)及び/又は解離速度定数(koff)には、相関があると考えるのが理に適っている。つまり、酸性pHにおけるヒトIL-8への結合親和性が弱いほど、及び/又は解離速度が速いほど、エンドソーム内においてフリー型ヒトIL-8の割合が増加するはずである。しかしながら、エンドソーム内におけるフリー型ヒトIL-8の割合が既に100%に近くなっているpH依存的IL-8結合抗体においては、それ以上に酸性pHにおける結合親和性を弱めること、及び/又は解離速度を速めることは、必ずしもフリー型ヒトIL-8の割合を効果的に増加させることにはつながらない。例えば、フリー型ヒトIL-8の割合を、99.9%の状態から99.99%に改善したとしても、その改善の度合いは顕著なものではないであろうことは容易に理解できる。
また、一般的な化学平衡の理論に則ると、抗IL-8抗体とヒトIL-8が共存し、それらの結合反応と解離反応が平衡状態に達している場合において、フリー型ヒトIL-8の割合は、抗体濃度と抗原濃度、解離定数(KD)の3者によって一義的に決定される。ここで、抗体濃度が高い場合、抗原濃度が高い場合、又は解離定数(KD)が小さい場合には、複合体の形成がされやすくなり、フリー型ヒトIL-8の割合は低下する。一方で、抗体濃度が低い場合、抗原濃度が低い場合、又は解離定数(KD)が大きい場合には、複合体の形成がされにくくなり、フリー型ヒトIL-8の割合は増加する。
ここで、今回の試験において、H89/L118を8 mg/kg投与した場合のヒトIL-8の消失速度は、2 mg/kgの抗体を投与した場合に比べて遅くなっていた。これはつまり、エンドソーム内において、8 mg/kgの抗体を投与した場合のフリー型ヒトIL-8の割合が、2 mg/kgの抗体を投与した場合に比べ低下したことを示唆している。その理由は、抗体の投与量を4倍に増加させたことにより、エンドソーム内の抗体濃度が増加し、エンドソーム内でのIL-8と抗体の複合体の形成がされやすくなったためだと推察される。つまり、抗体の投与量を増加させた投与群においては、エンドソーム内のフリー型ヒトIL-8の割合が低下したため、ヒトIL-8の消失速度が低下したと考えられる。これはまた、8 mg/kgの抗体投与時には、H89/L118の酸性pH条件下における解離定数(KD)の大きさが、フリー型ヒトIL-8を100%近くにするためには不十分であることを示唆している。すなわち、酸性pH条件下において、より大きな解離定数(KD)を有する(より結合が弱い)抗体であれば、8 mg/kgの抗体を投与した場合においても、フリー型IL-8の割合が100%に近い状態を実現し、2 mg/kgの抗体を投与した場合と同等のヒトIL-8消失速度を示すと考えられる。
以上のことから、目的のpH依存的IL-8結合抗体が、上記のようなフリー型ヒトIL-8の割合を100%近くまで達成できているかどうかを確認するためには、特に限定はされないが、in vivoにおける抗原消失効果の程度を上昇させる余地があるかどうかを検証することによっても可能である。例えば、H89/L118以上に酸性pHにおける結合親和性を弱めた、及び/又は酸性pHにおける解離速度を速めた新たなpH依存的IL-8抗体を用いたときのヒトIL-8消失速度を、H89/L118用いたときのIL-8消失速度と比較するという方法がある。前記の新たなpH依存的IL-8抗体が、H89/L118と同等のヒトIL-8消失速度を示した場合、H89/L118の酸性pHにおける結合親和性及び/又は解離速度は、エンドソーム内においてフリー型ヒトIL-8の割合を100%近くにするために既に十分なレベルにあることを示唆していると言える。一方で、前記の新たなpH依存的IL-8抗体の方が、より高いヒトIL-8消失速度を示した場合、H89/L118の酸性pHにおける結合親和性及び/又は解離速度は、改善の余地があることを示唆していると言える。
(8-1)pH依存的IL-8結合能を有する新たなH553/L118の作製
そこで、H89/L118よりもさらに、酸性pH条件下におけるヒトIL-8結合親和性を弱め、及び/又は解離速度を速めた抗体の作製を試みた。
H89/L118を基にして、ヒスチジンを中心としたアミノ酸改変を導入し、表10に示す改変抗体を実施例5と同様の方法で作製した。また、実施例5-2と同様の方法で、これらの抗体のヒトIL-8結合親和性を測定した。
結果の一部を表10に示す。重鎖としてH553-IgG1(配列番号:46)、軽鎖としてL118-k0MTを有するH553/L118と、重鎖としてH496-IgG1(配列番号:57)、軽鎖としてL118-k0MTを有するH496/L118が、H89/L118よりも更にpH依存性が増大していることが示された。
H553/L118を用いて、マウスにおけるヒトIL-8消失速度の評価を、実施例7-2と同様の方法で実施した。結果として得られた、血漿中ヒトIL-8濃度のデータを図17に、またマウス血漿中からのヒトIL-8クリアランス(CL)の数値を表11に示す。
特に限定はされないが、このような結果が得られた理由の一つとして、次のように考察することも可能である。
H553/L118は、2 mg/kgの抗体を投与した場合と8 mg/kgの抗体を投与した場合とで同等のヒトIL-8消失速度を示していた。これは、H553/L118は酸性pHにおけるIL-8結合が十分に弱いために、8 mg/kg投与の条件においても、エンドソーム内のフリー型IL-8の割合を100%に近いレベルで達成していることを示している。つまり、H89/L118は、2 mg/kg程度の用量においては、最大限のヒトIL-8消失効果を達成することが可能だが、8 mg/kg程度の高用量になると、その効果は減弱してしまう可能性を示唆している。一方、H553/L118は、8 mg/kg程度の高用量においてもなお、最大限のヒトIL-8消失効果を達成することが可能である。
H553/L118は、マウスにおいて顕著にH89/L118よりもヒトIL-8の消失を早めることが可能な抗体であることが示された。一方で、この抗体が生体内において長期間にわたってヒトIL-8の阻害効果を持続するためには、投与された抗体が生体内(例えば血漿中)に存在している期間中、IL-8中和活性が安定に保たれること(当該抗体のIL-8中和活性における安定性)もまた重要である。そこで、以下に示す方法で、これらの抗体のマウス血漿中における当該安定性を評価した。
マウス血漿は、C57BL/6J(Charles river)の血液から当業者公知の方法で採取した。マウス血漿800 μLに対し、200 mM PBS(Sigma, P4417)を200μL添加して1 mLとした。また、防腐剤としてアジ化ナトリウムを終濃度0.1%となるように添加した。更に、各抗体(Hr9, H89/L118, H553/L118)を終濃度0.2 mg/mLとなるように、上記のマウス血漿に添加した。この時点で一部を採取し、Initialサンプルとした。残りのサンプルは40℃で保管した。保管開始から1週間及び2週間が経過した時点で、それぞれ一部を採取し、1週間保存サンプル及び2週間保存サンプルとした。なお、全てのサンプルは各分析まで-80℃で凍結保管した。
次に、マウス血漿中に含まれる抗IL-8抗体の、ヒトIL-8に対する中和活性の評価を次のように行った。
ヒトIL-8の受容体として、CXCR1及びCXCR2が知られている。PathHunter(r) CHO-K1 CXCR2 β-Arrestin Cell Line (DiscoveRx社、Cat.# 93-0202C2)は、ヒトCXCR2を発現し、ヒトIL-8によるシグナルが伝達された際に化学発光を呈するように、人工的に作成された細胞株である。特に限定はされないが、この細胞を用いても、抗ヒトIL-8抗体が有するヒトIL-8に対する中和活性を評価することが可能である。まず、当該細胞の培養液中に、ヒトIL-8を添加すると、添加したヒトIL-8の濃度に依存して、ある量の化学発光を呈する。ここで、ヒトIL-8と抗ヒトIL-8抗体を併せて培養液中に添加した場合は、抗ヒトIL-8抗体はヒトIL-8と結合することにより、ヒトIL-8のシグナル伝達を遮断し得る。その結果として、ヒトIL-8の添加によって起こる化学発光は抗ヒトIL-8抗体により阻害され、抗体を添加していない場合に比べると弱い化学発光を示すか、あるいは全く化学発光を示さないことになる。そのため、抗体が有するヒトIL-8中和活性が強いほど化学発光の程度は弱まり、抗体が有するヒトIL-8中和活性が弱いほど化学発光の程度は強まることになる。
これは、マウス血漿中に添加して一定期間保存した抗体においても同様である。マウス血漿中で保存することによって、中和活性が変化しない抗体であれば、上記の化学発光の度合いは保存の前後で変化しないはずである。一方で、マウス血漿中で保存することによって、中和活性が低下する抗体の場合は、保存後の抗体を用いた場合の化学発光の度合いは、保存前に比べて増加することになる。
そこで、上記の細胞株を用いて、マウス血漿中に保存した抗体が、中和活性を維持しているかどうかを検証した。まず、細胞株をAssayComplete(tm) Cell Plating 0 Reagentに懸濁し、384well plateに5000 cells/wellずつ播種した。細胞の培養開始から1日後に、ヒトIL-8の添加濃度を決定するための試験を以下のように行った。最終ヒトIL-8濃度として45 nM(400 ng/mL)から0.098 nM(0.1 ng/mL)を含むように、ヒトIL-8溶液を段階希釈したものを細胞培養液に添加した。次に、製品プロトコールに従って検出試薬を添加し、化学発光検出装置を用いて相対化学発光量を検出した。それにより、細胞のヒトIL-8に対する反応性を確認し、抗ヒトIL-8抗体の中和活性を確認するために適切なヒトIL-8濃度を設定した。ここでは、ヒトIL-8濃度は2 nMとした。
次に、先述の抗ヒトIL-8抗体を添加したマウス血漿を用いて、そこに含まれる抗体の中和活性の評価を行った。上記で決定した濃度のヒトIL-8と、先述の抗ヒトIL-8抗体を含むマウス血漿を細胞培養液に添加した。ここで、添加するマウス血漿の量は、抗ヒトIL-8抗体濃度として2 μg/mL(13.3 nM)から0.016 μg/mL (0.1 nM)の範囲で段階的に含まれるように決定された。次に、製品プロトコールに従って検出試薬を添加し、化学発光検出装置を用いて相対化学発光量を検出した。
ここで、ヒトIL-8及び抗体を添加していないwellの相対化学発光量平均を0%、ヒトIL-8のみを添加し、抗体を添加していないwellの相対化学発光量平均を100%とした時の、各抗体濃度における相対化学発光量の相対値を算出した。
ヒトCXCR2発現細胞を用いたヒトIL-8阻害アッセイの結果を、Initial(マウス血漿中の保存処理なし)を図18-1に、40℃で1週間保存したサンプルの結果を図18-2に、40℃で2週間保存したサンプルの結果を図18-3に、それぞれ示した。
その結果、Hr9及びH89/L118は、マウス血漿中で保存した前後でヒトIL-8中和活性に差は見られなかった。一方、H553/L118は2週間の保存により、ヒトIL-8中和活性の低下が見られた。このことから、H553/L118は、Hr9やH89/L118と比較して、マウス血漿中においてヒトIL-8中和活性が低下しやすく、IL-8中和活性の面で不安定な性質を有する抗体であることが示された。
(9-1)各種IL-8結合抗体の免疫原性予測スコア
抗医薬品抗体(ADA)の産生は、治療用抗体の効果及び薬物動態に影響を及ぼし、時に重篤な副作用をもたらすことがあるため、臨床における治療用抗体の有用性や薬効は、ADAの産生によって制限され得る。治療用抗体の免疫原性は、多くの要因に影響を受けることが知られているが、特に治療用抗体が有するエフェクターT細胞エピトープの重要性が多数報告されている。
T細胞エピトープを予測するためのin silicoツールとしては、Epibase(Lonza)、iTope/TCED(Antitope)、及びEpiMatrix(EpiVax)などが開発されている。これらのin silicoツールを用いて、各アミノ酸配列中のT細胞エピトープを予測することができ(Expert Opin Biol Ther. 2007 Mar;7(3):405-18)、治療用抗体の潜在的な免疫原性評価が可能となる。
ここで、EpiMatrixを用いて、各抗IL-8抗体の免疫原性スコアを算出した。EpiMatrixは、免疫原性を予測したいタンパク質のアミノ酸配列を9アミノ酸ごとに区切ったペプチド断片の配列を機械的に設計し、それらに対して、8種類の主要なMHC Class IIアレル(DRB1*0101, DRB1*0301, DRB1*0401, DRB1*0701, DRB1*0801, DRB1*1101, DRB1*1301, DRB1*1501)に対する結合能を計算し、目的タンパク質の免疫原性を予測するシステムである。(Clin Immunol. 2009 May;131(2):189-201.)
上記のように算出された、各IL-8抗体の重鎖及び軽鎖の免疫原性スコアが、表12の「EpiMatrix Score」の欄に示されている。更に、EpiMatrix Scoreについて、Tregitopeの含有を考慮して補正された免疫原性スコアが、「tReg Adjusted Epx Score」の欄に示されている。Tregitopeとは、主に天然型の抗体配列中に多く含まれているペプチド断片配列であり、抑制性T細胞(Treg)を活性化することによって、免疫原性を抑制すると考えられている配列である。
また、これらのスコアについて、重鎖と軽鎖のスコアを合計したものが、合計の欄に示されている。
更に、EpiMatrixでは、重鎖と軽鎖のスコアを考慮したうえで、抗体分子全体として予測されるADA発生頻度を、各種市販抗体の実際のADA発生頻度と比較することも可能である。そのような解析を実施した結果が、図19に示されている。なお、システムの関係上、図19においては、hWS-4は「WS4」、Hr9は「HR9」、H89/L118は「H89L118」、H496/L118は「H496L118」、H553/L118は「H553L118」とそれぞれ表記されている。
図19に示されているように、各種の市販抗体のヒトにおけるADAの発生頻度は、Campath (Alemtuzumab)は45%、Rituxan (Rituximab)は27%、Zenapax (Daclizumab)は14%といった値であることが知られている。一方で、公知のヒト化抗ヒトIL-8抗体であるhWS-4のアミノ酸配列から予測されたADA発生頻度は10.42%であったが、今回新たに見出されたH89/L118(5.52%)、H496/L118(4.67%)、H553/L118(3.45%)は、hWS-4と比較すると有意に低下していた。
上記の通り、H89/L118、H496/L118及びH553/L118の免疫原性スコアは、hWS-4と比較して低下していたが、表12から明らかなように、重鎖の免疫原性スコアは軽鎖に比べると高く、特に重鎖のアミノ酸配列は、免疫原性の観点でまだ改善の余地があることを示唆している。そこで、H496の重鎖可変領域から、免疫原性スコアを低下させることが可能なアミノ酸改変の探索を行った。鋭意探索を行った結果、Kabatナンバリング52c番のアラニンがアスパラギン酸に置換されたH496v1、81番のグルタミンがスレオニンに置換されたH496v2、82b番のセリンがアスパラギン酸に置換されたH496v3の3種類の改変体が見いだされた。また、これら3種の改変を全て導入したH1004が作成された。
実施例9-1と同様の方法で、免疫原性スコアを算出した結果を表13に示す。
ここで、H1004と組み合わせる適切な軽鎖としては、L118に加え、L395もまた見いだされてきた。そのため、免疫原性スコアの算出においては、L118及びL395の両者を組み合わせたものを算出した。表13に示されているように、重鎖と軽鎖を組み合わせた際の免疫原性スコアとしても、H1004/L118及びH1004/L395は、非常に低い免疫原性スコアを示していた。
次に、これらについて実施例9-1と同様にADA発生頻度を予測した。その結果を図20に示す。なお、図20においては、H496v1/L118は「V1」、H496v2/L118は「V2」、H496v3/L118は「V3」、H1004/L118は「H1004L118」、H1004/L395は「H1004L395」とそれぞれ表記されている。
驚くべきことに、免疫原性スコアを顕著に低減させたH1004/L118及びH1004/L395は、ADA発生頻度の予測値についても改善しており、0%という予測値を示していた。
重鎖としてH1004-IgG1m(配列番号:47)、軽鎖としてL395-k0MT(配列番号:38)を有する抗体であるH1004/L395を作製した。H1004/L395のヒトIL-8への結合親和性を、Biacore T200(GE Healthcare)を用いて以下の通り測定した。
ランニングバッファーは、以下の2種を用い、それぞれの温度に設定して測定を行った。
・0.05% tween20, 40 mM ACES, 150 mM NaCl, pH 7.4, 40 ℃
・0.05% tween20, 40 mM ACES, 150 mM NaCl, pH 5.8, 37 ℃
センサーチップCM4(GE Healthcare)上にアミンカップリング法でプロテインA/G(PIERCE)を適当量固定化し、そこへ目的の抗体を捕捉させた。次に、ヒトIL-8希釈液とランニングバッファー(参照溶液として)とをインジェクトし、センサーチップ上に捕捉させた抗体にヒトIL-8を相互作用させた。なお、ランニングバッファーは上記のいずれかが用いられ、ヒトIL-8の希釈にもそれぞれのバッファーが使用された。センサーチップの再生には25 mM NaOH及び10 mMグリシン-HCl, pH 1.5が用いられた。測定で得られたセンサーグラムから算出されたカイネティクスパラメーターである結合速度定数 kon (1/Ms)、及び解離速度定数 koff (1/s)をもとに各抗体のヒトIL-8に対する KD (M) が算出された。各パラメーターの算出には Biacore T200 Evaluation Software(GE Healthcare)が用いられた。
測定結果を表14に示す。免疫原性スコアを低減させたH1004/L395は、H89/L118と比較して、中性pHにおけるヒトIL-8へのKDは同等であったが、酸性pHにおけるKD及びkoffは増加しており、エンドソーム内においてIL-8を解離しやすい性質を有していることが示された。
(10-1)各種pH依存的IL-8結合抗体の作製
実施例9に示した検討により、pH依存的IL-8結合能を有し、かつ免疫原性スコアが低減されたH1004/L395が取得された。次に、これらの好ましい性質と、マウス血漿中における安定性とを両立した改変体の創製を目指して、鋭意検討を行った。
H1004/L395を基に、各種の改変を導入し、以下の改変抗体を作製した。
中性及び酸性pH条件下におけるヒトIL-8結合親和性を、実施例9-3の方法と同様に測定した。その結果のうち、pH7.4におけるKD、pH5.8におけるKD及びkoffの値について、表15に示す。
次に、以下に示す方法でPBS中で保存した場合のIL-8の結合における安定性評価を実施した。
それぞれの抗体をDPBS (Sigma-Aldrich)に対して一晩透析を行った後、各抗体の濃度を0.1 mg/mLとなるように調製した。この時点で一部を採取し、Initialサンプルとした。残りのサンプルは50℃で1週間保管した後、回収して熱加速試験用サンプルとした。
次に、Initialサンプル及び熱加速試験用サンプルを用いて、BiacoreによるIL-8結合親和性の測定を以下のように実施した。
Biacore T200(GE Healthcare)を用いて、改変抗体へのヒトIL-8の結合量解析を行った。ランニングバッファーとして0.05% tween20, 40 mM ACES, 150 mM NaCl, pH 7.4を用い、40 ℃にて測定を行った。
センサーチップCM4(GE Healthcare)上にアミンカップリング法でプロテインA/G(PIERCE)を適当量固定化し、そこへ目的の抗体を捕捉させた。次に、ヒトIL-8希釈液とランニングバッファー(参照溶液として)とをインジェクトし、センサーチップ上に捕捉させた抗体にヒトIL-8を相互作用させた。ヒトIL-8の希釈にもランニングバッファーが使用された。センサーチップの再生には25 mM NaOH及び10 mMグリシン-HCl, pH 1.5が用いられた。測定で得られたヒトIL-8の結合量とその結合量を得たときの抗体捕捉量をBiacore T200 Evaluation Software(GE Healthcare)を用いて抽出した。
Initialサンプル及び熱加速試験用サンプルに関して、抗体捕捉量1000 RU当たりのヒトIL-8の結合量を算出した。さらに、InitialサンプルのヒトIL-8の結合量に対する熱加速試験用サンプルのヒトIL-8の結合量の比を算出した。
結果として得られた、Initialサンプルと熱加速試験用サンプルのIL-8結合量の比を、表15に併せて示す。
表15に示されているように、H1009/L395は、H89/L118と比べて中性pHにおけるヒトIL-8結合親和性はやや増強している一方で、酸性pHにおける結合親和性は低下しており、pH依存性がより強められていた。また、PBS中で50℃という過酷な条件に曝した場合の、IL-8結合における安定性において、H1009/L395はH89/L118よりもやや改善していた。
これらのことから、H1009/L395は、pH依存的IL-8結合能を有しつつ、マウス血漿中での中和活性も安定に保たれる可能性がある抗体として選定された。
次に、実施例8-3の方法と同様に、H1009/L395のIL-8中和活性が、マウス血漿中において安定に保たれるかどうかを評価した。ここでは、後に実施例15において、その詳細が記述されるH1009/L395-F1886sを用いた。この抗体は、H1009/L395と同じ可変領域を有し、定常領域は、天然型ヒトIgG1に比べて、酸性pH条件下におけるFcRn結合を増強する改変と、FcγRに対する結合を低減するための改変を有する定常領域を有する。H1009/L395のヒトIL-8に対する結合及びIL-8の中和活性は、この抗体の可変領域、とりわけHVRを中心とした領域が担っており、定常領域に導入された改変が影響を与えることは無いと考えられる。
マウス血漿中における安定性評価は、次のように実施した。マウス血漿585 μLに対し、200 mM リン酸緩衝液(pH6.7)を150 μL添加した。また、防腐剤としてアジ化ナトリウムを終濃度0.1%となるように添加した。各抗体(Hr9, H89/L118, H1009/L395-F1886s)について終濃度0.4 mg/mLとなるように、上記のマウス血漿に添加した。この時点で一部を採取し、Initialサンプルとした。残りのサンプルは40℃で保管した。保管開始から1週間及び2週間が経過した時点で、それぞれ一部を採取し、1週間保存サンプル及び2週間保存サンプルとした。なお、全てのサンプルは各分析まで-80℃で凍結保管した。
ヒトCXCR2発現細胞を用いたヒトIL-8中和活性測定は、実施例8-3と同様の方法で実施した。ただし、抗ヒトIL-8抗体の中和活性を確認するためのヒトIL-8濃度は、今回は1.2 nMで実施した。
上記の抗体を用いて得られた、ヒトCXCR2発現細胞を用いたヒトIL-8阻害アッセイの結果として、Initial(マウス血漿中の保存処理なし)の結果を図21-1に、40℃で1週間保存したサンプルの結果を図21-2に、40℃で2週間保存したサンプルの結果を図21-3に、それぞれ示した。
その結果、驚くべきことに、H1009/L395-F1886sは、マウス血漿中で40℃で2週間保存した場合においても、ヒトIL-8の中和活性が維持されており、H553/L118よりもIL-8中和活性が安定に保たれていた。
H1009/L395のマウスにおけるヒトIL-8消失速度の評価を、次に示す方法で実施した。抗体としてはH1009/L395、H553/L118及びH998/L63を用いた。マウスへの投与及び採血、マウス血漿中のヒトIL-8濃度測定は実施例7に示した方法で実施した。
結果として得られた、血漿中ヒトIL-8濃度のデータを図22に、またマウス血漿中からのヒトIL-8クリアランス(CL)の数値を表16に示す。
特に限定はされないが、ヒトIL-8の消失速度を増加させる効果については、次のように解釈することが可能である。一般に、抗原濃度がほぼ一定に保たれている生体内においては、抗原の産生速度と消失速度もまた、ほぼ一定に保たれていることになる。この状態に抗体を投与すると、抗原の産生速度は影響を受けない場合においても、抗原の消失速度は、抗原が抗体との複合体を形成することにより変化し得る。一般的には、抗原の消失速度は抗体の消失速度に比べて大きいため、そのような場合は、抗体と複合体を形成した抗原の消失速度は低下する。抗原の消失速度が低下すると、血漿中の抗原濃度が上昇するが、その際の上昇度合いは抗原単独時の消失速度と複合体形成時の消失速度の比によっても規定され得る。つまり、抗原単独時の消失速度に比べて、複合体形成時の消失速度が10分の1に低下した場合は、抗体が投与された生体の血漿中の抗原濃度は抗体投与前の約10倍にまで上昇し得る。ここで、これらの消失速度として、クリアランス(CL)を用いることも可能である。すなわち、生体に対して抗体を投与した後に起こる抗原濃度の上昇(抗原の蓄積)は、抗体投与前と投与後それぞれの状態における抗原CLによって規定されると考え得る。
ここで、H998/L63とH1009/L395を投与した際のヒトIL-8のCLに約30倍の差があったということは、ヒトにこれらの抗体を投与した際に起こる、血漿中ヒトIL-8濃度の上昇度合いに、約30倍の差が生じうることを示唆している。更に、血漿中ヒトIL-8濃度に30倍の差が生じるということは、それぞれの状況においてヒトIL-8の生物学的活性を完全に遮断するために必要な抗体の量もまた、約30倍の差が生じうるということになる。つまり、H1009/L395は、H998/L63に比べて、30分の1程度の極めて少量の抗体で、血漿中IL-8の生物学的活性を遮断することが可能である。また、H1009/L395とH998/L63それぞれを同じ投与量でヒトに投与した場合には、H1009/L395の方がより強く、かつ、より長期間にわたりIL-8の生物学的活性を遮断することが可能になる。また、長期間にわたりIL-8の生物学的活性を遮断するためには、そのIL-8中和活性が安定に維持されることが必要である。実施例10において示されたように、H1009/L395はマウス血漿を用いた実験から、長期間にわたってそのヒトIL-8中和活性を維持できることが明らかとなっている。これらの特筆すべき性質を含むH1009/L395は、生体内におけるIL-8を中和する効果という観点でも優れた効果を有する抗体であることが示された。
実施例10で示された、H1009/L395の30倍もの優れたヒトIL-8消失効果は、驚くべき効果であった。pH依存性抗原結合抗体を投与した際の抗原消失速度は、抗体と抗原の複合体が細胞内に取り込まれる速度に依存することが知られている。つまり、抗原との複合体を形成したときに、形成しないときと比較して細胞内に取り込まれる速度が増大すれば、pH依存性抗体による抗原消失効果を高めることも可能である。細胞内に抗体が取り込まれる速度を増強させる方法として、中性pH条件でのFcRn結合能を抗体に付与する方法(WO 2011/122011)や抗体のFcγRへの結合能を増強する方法(WO 2013/047752)、多価の抗体と多価の抗原を含む複合体の形成の促進を利用した方法(WO 2013/081143)などが知られている。
しかしながら、H1009/L395の定常領域において上記技術は用いられていない。また、IL-8はhomo dimerを形成することが知られているが、H1009/L395はヒトIL-8のhomo dimerの形成面を認識するため、H1009/L395が結合したヒトIL-8はmonomerの状態になることが明らかになっている。そのため、これらの抗体は多価の複合体を形成することはない。
つまり、H1009/L395に対しては、上記技術が使用されていないが、H1009/L395は30倍ものヒトIL-8消失効果を示していた。
以下、理論に拘束されることは意図しないが、H1009/L395に代表されるpH依存的IL-8結合抗体の前記特性をもたらす可能性のある要因の一つを挙げる。
ヒトIL-8は、高い等電点(pI)を有するタンパク質であり、公知の方法で計算される理論等電点はおよそ10である。つまり、中性pHの条件下においては、ヒトIL-8は正電荷側に偏った電荷を有するタンパク質である。H1009/L395に代表されるpH依存的IL-8結合抗体もまた、正電荷側に偏った電荷を有するタンパク質であり、H1009/L395の理論等電点はおよそ9である。つまり、元々高い等電点を有し、正電荷に富むタンパク質であるH1009/L395が、高い等電点を有するヒトIL-8と結合して生じる複合体は、H1009/L395単独よりも等電点が上昇する。
抗体の正電荷の数を増加させること、及び/又は、負電荷の数を減少させることを含む、抗体の等電点の増加は、抗体・抗原複合体の細胞内への非特異的な取り込みを増加させると考えることも可能である。抗IL-8抗体と高い等電点を有するヒトIL-8との複合体は、抗IL-8抗体単独よりも等電点が増大し、細胞内に取り込まれやすくなっているという可能性も考えられた。
また、先述の通り、細胞外マトリックスへの結合性もまた、細胞内取り込みに影響する可能性がある因子の一つである。そこで、細胞外マトリックスに対する抗体単独の結合性と、ヒトIL-8と抗体との複合体の結合性が異なるかどうかを検証した。
TBS(Takara, T903)を用いて、細胞外マトリックス(BDマトリゲル基底膜マトリックス/ BD社製)を2 mg/mLに希釈した。希釈した細胞外マトリックスをMULTI-ARRAY 96well Plate, High bind, Bare (Meso Scale Discovery: MSD社製) に1wellあたり5 μL分注し、4℃で一晩固相化した。その後、ブロッキングは、150 mM NaCl, 0.05% Tween20, 0.5% BSA, 0.01% NaN3を含む20 mM ACES buffer, pH7.4を用いて行った。
また、評価に供する抗体を次のように調整した。抗体単独の添加サンプルとしては、各抗体を以下に示すBuffer-1を用いて9 μg/mLにそれぞれ希釈した後、Buffer-2を用いてさらに希釈し、最終濃度をそれぞれ3 μg/mLとした。
Buffer-1: 150 mM NaCl, 0.05% Tween20, 0.01% NaN3を含む20 mM ACES buffer, pH7.4
Buffer-2: 150 mM NaCl, 0.05% Tween20, 0.1% BSA, 0.01% NaN3を含む20 mM ACES buffer, pH7.4
一方、抗体とヒトIL-8との複合体の添加サンプルは、抗体の10倍のモル濃度のヒトIL-8を抗体サンプルに添加した上で、Buffer-1を用いて抗体濃度が9 μg/mLになるようにそれぞれ希釈された後、最終抗体濃度がそれぞれ3 μg/mLとなるように、Buffer-2によって更に希釈された。なお、この時ヒトIL-8は約0.6 μg/mLとなっている。複合体を形成させるために、室温で1時間振盪した。
次に、ブロッキング溶液を除去したプレートに、抗体単独あるいは複合体の溶液を添加し、室温で1時間振盪した。その後、抗体単独あるいは複合体の溶液を除去し、0.25% Glutaraldehyde を含むBuffer-1を添加して10分間静置した後、0.05% Tween20を含むDPBS (和光純薬工業社製) で洗浄した。ECL検出用抗体は、Goat anti-human IgG (gamma) (Zymed Laboratories社製) をSulfo-Tag NHS Ester (MSD社製) を用いてSulfo-Tag化し調製した。ECL検出用抗体を1 μg/mLとなるようにBuffer-2で希釈してプレートに添加し、遮光下、室温で1時間振盪した。ECL検出用抗体を除去し、MSD Read Buffer T(4x)(MSD社製)を超純水で2倍希釈した溶液を添加した後、SECTOR Imager 2400 (MSD社製)を用いて発光量を測定した。
結果を図23に示す。興味深いことに、H1009/L395などの抗IL-8抗体はいずれも、抗体単独(-IL8)ではほとんど細胞外マトリックスへの結合が見られなかったが、ヒトIL-8(+hIL8)との複合体を形成して、細胞外マトリックスへの結合が見られることが明らかになった。
抗IL-8抗体が、上記のようにヒトIL-8と結合することにより、細胞外マトリックスに対して結合性を有するようになるという性質は、先行技術情報からは明らかになっていない。また、限定はされないが、このような性質をpH依存的IL-8結合抗体と組み合わせ、より効率的にIL-8の消失速度を増大させることも可能である。
マウスの生体内において、pH依存的IL-8結合抗体がヒトIL-8との複合体を形成して、その複合体の細胞内への取り込みが増加するかどうかを、以下に示す方法で確認した。
まず、H1009/L395の可変領域と、各種Fc受容体への結合親和性を欠損しているFc領域とを有する変異体を作成した。具体的には、酸性pH条件下におけるヒトFcRnへの結合能を欠失させる改変として、重鎖であるH1009-IgG1に対して、EUナンバリング253番目のイソロイシンをアラニンに、254番目のセリンをアスパラギン酸に置換した。また、マウスFcγRへの結合を欠失させる改変として、235番目のロイシンをアルギニンに、236番目のグリシンをアルギニンに、239番目のセリンをリジンに置換した。これら4つの改変を含む重鎖として、1009-F1942m(配列番号:49)を作製した。また、重鎖としてH1009-F1942m、軽鎖としてL395-k0MTを有する、H1009/L395-F1942mを作製した。
このFc領域を有する抗体は、酸性pH条件下におけるFcRn結合親和性を欠損しているため、エンドソーム内から血漿中への移行が起こらない。そのため、このような抗体は生体内において、天然型Fc領域を有する抗体に比べて、速やかに血漿中から消失する。このとき、天然型Fc領域を有する抗体は細胞内に取り込まれた後、FcRnによるサルベージを受けなかった一部の抗体のみがライソソームに移行して分解されるが、FcRnへの結合親和性を持たないFc領域を有する抗体の場合は、細胞内に取り込まれた抗体の全てがライソソームで分解を受ける。すなわち、このような改変Fc領域を有する抗体の場合は、投与した抗体の血漿中からの消失速度は、細胞内に取り込まれる速度と等しいと考えることもできる。つまり、FcRnへの結合親和性を欠損させた抗体の血漿中からの消失速度を測定することによっても、当該抗体の細胞内に取り込まれる速度を確認することが可能である。
そこで、このH1009/L395-F1942mとヒトIL-8が結合した複合体の細胞内への取り込みが、H1009/L395-F1942mの取り込みよりも増加するか否かを検証した。具体的には、当該抗体を単独で投与した場合と、ヒトIL-8との複合体を形成させて投与した場合とで、当該抗体の血漿中からの消失速度が変化するかどうかを検証した。
ヒトFcRnトランスジェニックマウス(B6.mFcRn-/-.hFcRn Tg line 32 +/+ mouse、Jackson Laboratories、Methods Mol Biol. (2010) 602, 93-104)に、抗ヒトIL-8抗体のみを投与した場合と、ヒトIL-8と抗ヒトIL-8抗体を同時に投与した場合のそれぞれで、抗ヒトIL-8抗体の体内動態を評価した。抗ヒトIL-8抗体溶液(200 μg/mL)及び、ヒトIL-8(10 μg/mL)と抗ヒトIL-8抗体(200 μg/mL)の混合溶液のそれぞれを、尾静脈に10mL/kgで単回投与した。このとき、ヒトIL-8に対して抗ヒトIL-8抗体は十分量過剰に存在することから、ヒトIL-8はほぼ全て抗体に結合していると考えられる。投与5分後、2時間後、7時間後、1日後、2日後に血液を採取した。採取した血液は直ちに4℃、15,000 rpmで15分遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで-20℃以下に設定された冷凍庫に保存した。
マウス血漿中の抗ヒトIL-8抗体濃度は電気化学発光法によって測定された。まず、5%BSA(w/v)を含有したPBS-Tween溶液を用いて室温で1晩ブロッキングされたStreptavidin Gold Multi-ARRAY Plate(Meso Scale Discovery)に、Anti-Human Kappa Light Chain Goat IgG Biotin (IBL)を室温で1時間反応させることによって抗ヒト抗体固相化プレートが作成された。血漿中濃度として3.20、1.60、0.800、0.400、0.200、0.100、0.0500 μg/mLの抗ヒトIL-8抗体を含む検量線用試料と100倍以上に希釈されたマウス血漿測定用試料が調製された。各試料は、ヒトIL-8と混合された後に抗ヒト抗体固相化プレートの各ウェルに50 μLで分注されて、室温で1時間撹拌された。ヒトIL-8の終濃度は333 ng/mLとなるよう調製された。
その後、前記プレートに、マウスIgGの定常領域を有する抗ヒトIL-8抗体(社内調製品)を加え、室温で1時間反応させた。さらにSULFO-TAG NHS Ester(Meso Scale Discovery)でルテニウム化したAnti-Mouse IgG(BECKMAN COULTER)を前記プレートに加えて1時間反応させた後、Read Buffer T(×1)(Meso Scale Discovery)を分注し、ただちにSECTOR Imager 2400 (Meso Scale Discovery)で測定を行った。抗ヒトIL-8抗体濃度は検量線のレスポンスから解析ソフトウェアSOFTmax PRO(Molecular Devices)を用いて算出した。
結果として得られた、マウス血漿中の抗体濃度を図24に、またそれぞれの条件における抗体のクリアランスを表17に示した。
特に限定はされないが、これまで得られた知見から、次のような解釈を行うこともできる。pH依存的IL-8結合抗体であるH1009/L395は、ヒトIL-8と複合体を形成すると、その複合体は、抗体単独で存在する場合と比べて、より等電点が高く、正電荷に偏った状態となる。同時に、その複合体の細胞外マトリックスへの結合性が抗体単独の結合性よりも増大している。等電点の上昇や、細胞外マトリックスへの結合増強といった性質は、生体内において、細胞内への取り込みを促進させる因子として考えることができる。更に、マウスを用いた実験から、H1009/L395とヒトIL-8との複合体の細胞内への取り込み速度がH1009/L395の取り込み速度よりも2.2倍以上に増大することも示された。以上のことから、理論的な説明付けとin vitroでの性質、in vivoでの現象とが一貫して、H1009/L395がヒトIL-8と複合体を形成して、細胞内への取り込みが促進され、ヒトIL-8の消失を顕著に増加させている仮説を支持している。
これまでにも、IL-8に対する抗体はいくつか報告があるが、IL-8との複合体を形成したときの細胞外マトリックスへの結合性の増大や、前記複合体の細胞内への取込みの増加はこれまでに報告がない。
また、抗IL-8抗体の細胞内への取込みの増加がIL-8との複合体を形成したときに見られるということは、血漿中においてIL-8と複合体を形成した抗IL-8抗体は速やかに細胞内に取り込まれる一方で、IL-8との複合体を形成していないフリー型の抗体は、細胞内には取り込まれずに滞留しやすいと考えることも可能である。この場合、抗IL-8抗体がpH依存性であるときは、一旦細胞内に取り込まれた抗IL-8抗体が細胞内でIL-8分子を解離後に再び細胞外に戻り、別なIL-8分子と結合することが可能となることから、複合体形成時の細胞内への取込み増加は、IL-8をより強く除去するというさらなる効果を有すると考えることも可能である。すなわち、細胞外マトリックスへの結合が増大した抗IL-8抗体や細胞内取り込みが増加した抗IL-8抗体を選択することも、本開示の別な側面である。
次に、H1009/L395について実施例9-1と同様の方法で免疫原性スコア及びADA発生頻度の予測を行った。その結果を表18及び図25に示す。なお、図25において、H1009/L395は、「H1009L395」と表記されている。
以上のことから、H1009/L395において予測される免疫原性は、公知の抗ヒトIL-8抗体であるhWS-4と比較して、大幅に低下していた。このことから、H1009/L395はヒトにおける免疫原性が極めて低く、長期にわたって安全に、抗IL-8中和活性を持続することが可能であると考えられる。
これまでの実施例に記載の通り、pH依存的IL-8結合抗体H1009/L395は、天然型IgG1の定常領域を有する場合において、非常に優れた性質を有する抗体である。しかしながら、アミノ酸置換を定常領域に含む抗体、例えば実施例5で例示された、酸性pHにおけるFcRn結合を増強させたFc領域を含む抗体としても利用可能である。そこで、酸性pHにおけるFcRn結合を増強させたFc領域がpH依存的IL-8結合抗体においても機能することを、H89/L118を用いて確認した。
H89/L118のFc領域に対して、実施例5-1に記載の、各種FcRn結合増強改変を導入した。具体的には、H89-IgG1のFc領域に対して、F1847m、F1848m、F1886m、F1889m、F1927m、F1168mに用いられている改変を導入して、以下の改変体を作製した。
重鎖としてH89-IgG1m(配列番号:50)、軽鎖としてL118-K0MTを有するH89/L118-IgG1、
重鎖としてH89-F1168m(配列番号:51)、軽鎖としてL118-K0MTを有するH89/L118-F1168m、
重鎖としてH89-F1847m(配列番号:52)、軽鎖としてL118-K0MTを有するH89/L118-F1847m、
重鎖としてH89-F1848m(配列番号:53)、軽鎖としてL118-K0MTを有するH89/L118-F1848m、
重鎖としてH89-F1886m(配列番号:54)、軽鎖としてL118-K0MTを有するH89/L118-F1886m、
重鎖としてH89-F1889m(配列番号:55)、軽鎖としてL118-K0MTを有するH89/L118-F1889m、
重鎖としてH89-F1927m(配列番号:56)、軽鎖としてL118-K0MTを有するH89/L118-F1927m、
これらを用いたカニクイザルPK試験を、次に示す方法で実施した。
なお、後述の実施例で記載のH89/L118-F22も同様に作製した(重鎖としてH89-F22(配列番号:106)、軽鎖としてL118-K0MTを有する)。
カニクイザルに、抗ヒトIL-8抗体を投与した後の抗ヒトIL-8抗体の体内動態を評価した。抗ヒトIL-8抗体溶液を2 mg/kgで単回静脈内投与した。投与5分後、4時間後、1日後、2日後、3日後、7日後、10日後、14日後、21日後、28日後、35日後、42日後、49日後、56日後に血液を採取した。採取した血液は直ちに4℃、15,000 rpmで10分遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで-60℃以下に設定された冷凍庫に保存した。
カニクイザル血漿中の抗ヒトIL-8抗体濃度は電気化学発光法によって測定された。まず、Anti-hKappa Capture Ab(Antibody Solutions)がMULTI-ARRAY 96-well Plate(Meso Scale Discovery)に分注され、室温で1時間撹拌された。その後、5% BSA(w/v)を含有したPBS-Tween溶液を用いて室温で2時間ブロッキングすることで抗ヒト抗体固相化プレートが作成された。血漿中濃度として40.0、13.3、4.44、1.48、0.494、0.165、0.0549 μg/mLの抗ヒトIL-8抗体を含む検量線用試料と500倍以上に希釈されたカニクイザル血漿測定用試料が調製され、抗ヒト抗体固相化プレートの各ウェルに50μLで分注されてから、室温で1時間撹拌された。その後、前記プレートに、Anti-hKappa Reporter Ab, Biotin conjugate(Antibody Solutions)を加え、室温で1時間反応させた。さらにSULFO-TAG Labeled Streptavidin(Meso Scale Discovery)を加えて室温で1時間反応させた後、Read Buffer T(×1)(Meso Scale Discovery)を分注し、ただちにSECTOR Imager 2400(Meso Scale Discovery)で測定を行った。抗ヒトIL-8抗体濃度は検量線のレスポンスから解析ソフトウェアSOFT Max PRO(Molecular Devices)を用いて算出した。
結果として得られた各抗体の半減期(t1/2)及びクリアランス(CL)を表19に、カニクイザル血漿中抗体濃度推移を図26にそれぞれ示した。
天然型ヒトIgG1は、そのFc領域が各種の免疫系細胞上のFcγ受容体(以下、FcγRと表記する)と結合して、対象とする細胞に対してADCCやADCPといったエフェクター機能を示すことが知られている。
一方、IL-8は可溶型サイトカインであり、医薬品として使用される抗IL-8抗体は、主にIL-8が過剰に存在している部位においてその機能を中和して、薬理作用を示すことが期待される。このようなIL-8が過剰に存在している部位としては、特に限定はされないが、例えば炎症部位が想定され得る。一般的に、このような炎症部位においては、各種の免疫系細胞が集まり、なおかつ活性化されていることが知られている。このような免疫系細胞に対して、Fc受容体を介して意図せぬ活性化シグナルを伝達してしまうことや、意図せぬ細胞に対してADCC、ADCPといった活性を引き起こしてしまうことは、必ずしも好ましくはない。そのため、特に限定はされないが、安全性の観点からは、生体に投与される抗IL-8抗体は、FcγRに対する結合能は低いほうが好ましいと考えられ得る。
各種のヒト及びカニクイザルFcγRに対する結合能を低下させることを目的として、H1009/L395-F1886mのFc領域に対して更にアミノ酸改変を導入した。具体的には、重鎖であるH1009-F1886mに対して、EUナンバリング235番目のLをRに、236番目のGをRに、239番目のSをKに、それぞれ置換を行ってH1009-F1886s(配列番号:37)を作製した。同様に、H1009-F1886mに対して、EUナンバリング235番目のLをRに、236番目のGをRに置換し、さらにEUナンバリング327番目から331番目までの領域をヒト天然型IgG4配列として、H1009-F1974m(配列番号:36)を作製した。これらの重鎖と、軽鎖としてL395-k0MTを有する抗体として、H1009/L395-F1886s及びH1009/L395-F1974mを作製した。
次に、作製されたH1009/L395-F1886s及びH1009/L395-F1974mの、ヒト及びカニクイザルそれぞれの可溶型FcγRIa及びFcγRIIIaに対する結合性を、次に示す方法で確認した。
作製されたH1009/L395-F1886s及びH1009/L395-F1974mに関して、Biacore T200(GE Healthcare)を用いて、ヒト及びカニクイザルそれぞれの可溶型FcγRIa及びFcγRIIIaとの結合実験を行った。ヒト及びカニクイザルそれぞれの可溶型FcγRIa及びFcγRIIIaは、当業者に公知の方法でHisタグを付与した分子形として作製した。センサーチップCM4(GE Healthcare)上にアミンカップリング法でrProtein L (BioVision)を適当量固定化し、そこへ抗体を捕捉させた。次に、可溶型FcγRIaあるいはFcγRIIIaとランニングバッファー(参照溶液として)とをインジェクトし、センサーチップ上に捕捉させた抗体に相互作用させた。なお、ランニングバッファーはHBS-EP+(GE Healthcare)を用い、可溶型FcγRIaあるいはFcγRIIIaの希釈にもHBS-EP+が用いられた。センサーチップの再生には10 mMグリシン-HCl, pH 1.5が用いられた。測定は全て20 ℃で実施された。
結果を図27に示す。ここで、ヒトFcγRIa、ヒトFcγRIIIa、カニクイザルFcγRIa、カニクイザルFcγRIIIaの順に、hFcγRIa、hFcγRIIIa、cynoFcγRIa、cynoFcγRIIIaと記載されている。H1009/L395-F1886mはいずれのFcγRに対しても結合することが示されたのに対して、新たに作製されたH1009/L395-F1886s及びH1009/L395-F1974mは、いずれのFcγRに対しても結合しないことが確認された。
次に、作製されたH1009/L395-F1886s及びH1009/L395-F1974mのマウスにおけるヒトIL-8消失速度や抗体の血漿中滞留性を、以下に示す実験で確認した。なお、ここでは、H1009/L395-F1886sの抗体投与量を増加させることによる影響も含めて評価するために、H1009/L395-F1886sに関しては投与量を2 mg/kg, 5 mg/kg, 10 mg/kgの3点で評価を行った。
ヒトFcRnトランスジェニックマウス(B6.mFcRn-/-.hFcRn Tg line 32 +/+ mouse、Jackson Laboratories、Methods Mol Biol. (2010) 602, 93-104)に、ヒトIL-8と、抗ヒトIL-8抗体を同時に投与した後のヒトIL-8の体内動態を評価した。ヒトIL-8(10 μg/mL)、抗ヒトIL-8抗体(200 μg/mL、500 μg/mL又は1000 μg/mL)の混合溶液を尾静脈に10 mL/kgで単回投与した。このとき、ヒトIL-8に対して抗ヒトIL-8抗体は十分量過剰に存在することから、ヒトIL-8はほぼ全て抗体に結合していると考えられる。投与5分後、2時間後、4時間後、7時間後、1日後、2日後、3日後、7日後、14日後、21日後、28日後に血液を採取した。採取した血液は直ちに4℃、15,000 rpmで15分遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで-20℃以下に設定された冷凍庫に保存した。
マウス血漿中のヒトIL-8濃度測定は、実施例7と同様の方法で実施した。結果として得られた、血漿中ヒトIL-8濃度のデータを図28に、またマウス血漿中からのヒトIL-8クリアランス(CL)の数値を表20に示す。
まず、2 mg/kgの投与群の比較において、天然型IgG1のFc領域を有するH1009/L395と、改変Fc領域を有するH1009/L395-F1886sは、同等のヒトIL-8消失効果を有することが示された。
次に、H1009/L395-F1886sの抗体投与量を変化させた場合は、投与後1日の時点での血漿中IL-8濃度には若干の差が見られたものの、ヒトIL-8クリアランスの値としては2 mg/kgと10 mg/kgとで有意な差は見られなかった。このことは、H1009/L395の可変領域を含む抗体は、高用量で投与された場合でも十分なIL-8消失効果を示すことを強く示唆する。
次に、H1009/L395-F1886s及びH1009/L395-F1974mを用いて、カニクイザルにおける抗体の血漿中滞留性を以下に示す方法で検証した。
カニクイザルに、抗ヒトIL-8抗体単独、又はヒトIL8及び抗ヒトIL-8抗体を同時に投与した後の、抗ヒトIL-8抗体の体内動態を評価した。抗ヒトIL-8抗体溶液(2 mg/mL)又は、ヒトIL-8(100 μg/kg)及び抗ヒトIL-8抗体(2 mg/kg)の混合溶液を1 mL/kgで単回静脈内投与した。投与5分後、4時間後、1日後、2日後、3日後、7日後、10日後、14日後、21日後、28日後、35日後、42日後、49日後、56日後に血液を採取した。採取した血液は直ちに4℃、15,000 rpmで10分遠心分離し、血漿を得た。分離した血漿は、測定を実施するまで-60℃以下に設定された冷凍庫に保存した。
カニクイザル血漿中の抗ヒトIL-8抗体濃度の測定は、実施例14に記載の方法で実施した。結果として得られた、血漿中抗ヒトIL-8抗体濃度のデータを図29に、またカニクイザル血漿中からの抗ヒトIL-8抗体の半減期(t1/2)及びクリアランス(CL)の数値を表21に示す。
まず、天然型ヒトIgG1のFc領域を有するHr9及びH89/L118と比較して、機能が改良されたFc領域を有するH1009/L395-F1886sは、血漿中滞留性が有意に向上していることが示された。
更に、H1009/L395-F1886sは、ヒトIL-8と同時に投与された場合においても、抗体単独で投与された場合と同等の血漿中濃度推移を示した。特に限定されることは無いが、このことから、次のように考察することも可能である。先述の通り、H1009/L395とヒトIL-8との複合体の細胞内への取込みはH1009/L395単独の取込みよりも増加していることが示されている。一般的に、高分子量のタンパク質は、非特異的あるいは受容体依存的に細胞内に取り込まれた後、ライソソームへと移行し、ライソソームに存在する各種の分解酵素によって分解されると考えられている。そのため、当該タンパク質の細胞内への取り込み速度が上昇すれば、その血漿中滞留性も悪化する方向に進むことが考えられる。しかしながら、抗体の場合は、エンドソーム内に存在するFcRnによって血漿中へと戻される性質を有するため、FcRnによるサルベージ機能が十分に働いている限りにおいて、細胞内取り込み速度が増加した場合においても、血漿中滞留性には影響しないことが考えられる。ここで、H1009/L395-F1886sは、ヒトIL-8と同時にカニクイザルに投与された場合においても、血漿中滞留性への影響は見られなかった。このことは、H1009/L395-F1886sは、抗体の細胞内取り込み速度は増加しても、FcRnによって十分にサルベージされ、血漿中へと戻ってくることができている可能性を示している。
更に、もう一種類のFc領域改変体であるH1009/L395-F1974mも、H1009/L395-F1886sと同等の血漿中滞留性を示していた。先述の通り、これらのFc領域改変体は各種FcγRへの結合能を低下させるための改変として、異なるものが導入されているが、それらは抗体自身の血漿中滞留性に影響を与えることがないことを示している。以上のことから、H1009/L395-F1886s及びH1009/L395-F1974mのいずれも、カニクイザルにおける血漿中滞留性は、天然型IgG1のFc領域を有する抗体と比較して顕著に改善し、極めて良好であることが示された。
天然型IgGのFc領域を有するH1009/L395は、上記の通りに十分に有用であるが、機能が改良されたFc領域を含むH1009/L395の改変体もまた、その有用性が高められた抗体として適宜使用することが可能である。具体的には、酸性pH条件下におけるFcRn結合を増強させ、血漿中滞留性を改善し、より長期間にわたり効果を持続することも可能になる。また、FcγRへの結合能を低下させる改変が導入されたFc領域を含む改変体は、投与された生体内での意図せぬ免疫系細胞の活性化や細胞傷害活性などの発生を回避し、安全性の高い抗体医薬品として使用することが可能となる。このようなFcとしては、当明細書中で実施されたF1886s又はF1974mを用いることが特に好ましいが、これらのFcに限定されるということはなく、同様の機能を有している限り、他の改変Fc領域含む抗体医薬品も本開示の一実施態様として使用される。
結果として、H1009/L395-F1886s、H1009/L395-F1974m等を含む本開示における抗体は、長期間にわたって、かつ安全に、ヒトIL-8の生物学的活性を強く阻害し得る状態を維持することが可能である。これは、既存の抗IL-8抗体では達成し得ないレベルを実現しており、極めて完成度の高い抗IL-8抗体医薬品として、その使用が期待されるものである。
子宮内膜症におけるIL-8の関与を確認するため、子宮内膜症患者の嚢胞液中のIL-8濃度を測定した。サンプルは、自治医科大学で手術適応の子宮内膜症患者から術後組織の嚢胞液を回収し、1次匿名化ののち、中外製薬にて2次匿名化を行った。嚢胞液中のIL-8濃度は電気化学発光法にて測定された。まず、抗ヒトIL-8抗体(Hycult Biotech)をビオチン標識した後に、5%BSA(w/v)を含有したPBS-Tween溶液を用いて2時間ブロッキングされたMSD GOLD 96-Well Streptavidin SECTOR Plate(Meso Scale Discovery)に分注して室温・暗所で1時間撹拌することによって、抗ヒトIL-8抗体固相化プレートが作成された。嚢胞液中濃度として900、300、100、33.3、11.1、3.70、1.23 pg/mLのヒトIL-8 (NP_000575.1を参照し社内調整)を含む検量線試料と5倍以上希釈されたヒト嚢胞液測定試料が調製され、抗ヒトIL-8抗体固相化プレートの各ウェルに25 μLで分注されてから、室温・暗所で1時間撹拌させた。その後、SULFO-TAG Labeled抗ヒトIL-8抗体(社内調製品)を室温・暗所で1時間反応させた後、Read Buffer T(×1)(Meso Scale Discovery)を分注し、ただちにSECTOR Imager 2400(Meso Scale Discovery)で測定を行った。ヒトIL-8濃度は検量線のレスポンスから解析ソフトウェアSOFT Max PRO(Molecular Devices)を用いて算出した。
その結果、図30に示すように、子宮内膜症患者の嚢胞液中のIL-8濃度は10000 pg/mL以上と非常に高濃度であることが判明し、子宮内膜症におけるIL-8の重要性が示唆された。
子宮内膜症における抗IL-8抗体の薬効を評価するため、子宮内膜症外科的誘引モデルを作製し、評価を行った。病態モデルの作製は以下のように行った。
性周期が定期的である8-14歳齢のメスカニクイザル(医薬基盤・健康・栄養研究所 霊長類医科学研究センターより入手)に対し、黄体期該当期間中に麻酔下で開腹を行い、子宮体部をV字に切開して切除した。次に子宮の平滑筋層を残して5 mm~10 mm角にトリミングし、子宮内膜部分が腹膜に接するように右腹壁腹膜に1か所、左腹壁腹膜に2か所(頭側、尾側)に吸収糸にて縫合し、移植部とした。切除後の子宮は吸収糸にて縫合した。さらに残りの内膜組織を細切し5 mLの2 ng/mL Hepatocyte Growth Factor溶液(R&D systems社製) に懸濁し、腹腔内に播種移植し、閉腹した。麻酔はケタミンキシラジンのバランス麻酔(2:1の比率を目安に混合)を用いるが、麻酔維持時間によってはイソフルレンで維持した。術中はヒーティングパットにより体温維持管理を行い、心電図により状態を管理した。術後の覚醒時には拮抗薬のアンチセダン(キシラジン量と同量を目安に)と抗生剤のセファラジンを投与、鎮痛剤のザルバンを術後2日間投与し、毎日の定期観察で術後の異常がないことを確認した。なお腹腔鏡観察時も同様に麻酔と術後管理を実施した。
腹腔鏡観察は、腹部正中を麻酔下で切開し、トローカーを刺入し、腹腔鏡を挿入した。腹腔鏡はビデオシステム(カールストルツ社製)並びにモニターに接続され、腹腔内の観察はモニターで行い、記録はビデオシステムで行った。次に、高速気腹装置(OLYMPUS社製)を用いて二酸化炭素ガスを腹腔内に充満させ腹部を拡張し、目盛り付バーや鉗子を腹側部から挿入した。病変のサイズは目盛りのついたバーあるいは目盛りのついた鉗子を用いて測定した。
子宮内膜組織の縫合によって形成された結節性病変(nodular lesion)のサイズ(縦、横、高さ)を測定し、縦(mm) x 横 (mm) x 高さ(mm)から体積を算出した。子宮内膜組織の細切後、播種によって形成された病変部位と癒着に対しては、臨床で使用されているr-AFS scoreをサル用に改変したmodified r-AFS scoreに則って、癒着の場所、範囲、深さおよび病変のサイズを腹腔鏡観察時に評価した。Modified r-AFS scoreは、図31に示すように、サル用に以下2点の改変を行った。
1.サルモデルでは膀胱への癒着頻度がヒトよりも高いため、膀胱子宮窩への癒着項目を追加した。膀胱への癒着の評価方法は、ダグラス窩の癒着と同様のスコアリングとした。
2.サルはヒトより体と臓器の大きさが小さいため、病変サイズのクライテリアを<3 mm, 3-10 mm, >10 mmに変更してスコアリングした。
後日ビデオシステムを用いて録画した腹腔鏡観察動画を確認して腹腔鏡による評価を最終化した。
誘引4ヶ月後の腹腔鏡観察結果をもとに、基準を満たした個体を選択し、群分けを行った。採用基準は以下の通りである。
1.誘引4ヶ月後に腹腔鏡観察を行い移植内膜の生着が確認できること
2.誘引後投与前期間中に3回以上の月経出血が確認できること
群分けは、2回に分けて実施し、個体ごとの結節性病変の平均体積と測定可能であった病変数をもとにランダマイズを行い、最終的にvehicle群に6頭, 抗体H1009/L395-F1974m投与群に7頭割り付けた。最終的に割り付けられた全個体のデータを用いて、群間に偏りが無いことをKraskal-Wallis検定により確認した。項目は結節性病変の体積(病変ごと)、体重、size r-AFS score, adhesion r-AFS scoreを確認した。
投与開始から6ヶ月後の最終腹腔鏡観察後、追加麻酔して放血、安楽死処置したのち、剖検を行い、移植部を含めた骨盤内臓器(左右卵巣、左右卵管、左右卵管間膜、左右卵管采、左右子宮広間膜、子宮、膣、ダグラス窩、膀胱子宮窩)を採取した。採取した組織は10%中性緩衝ホルマリン液で固定し、パラフィン包埋薄切組織標本 でヘマトキシリン・エオジン[H・E]染色を実施した。組織および縫合由来病変に割を入れて嚢胞液が貯留している場合は嚢胞液も採取した。
まず、子宮内膜組織の縫合によって形成された各結節性病変一つずつの体積を算出し、継時的な変化及び大きさによって以下の表24のように分類した。癒着によって評価できないなど、経時的な評価ができていない場合は、該当箇所を解析に含めなかった。
Change of total r-AFS score=total r-AFS score(投与後)- total r-AFS score(投与前)
Change of adhesion r-AFS score=adhesion r-AFS score(投与後) - adhesion r-AFS score(投与前)
Change of size r-AFS score=size r-AFS score(投与後) - size r-AFS score(投与前)
これらの結果から、抗IL-8抗体が子宮内膜症病変体積の縮小、癒着の改善、上皮およびstroma細胞の萎縮性変化、免疫細胞の浸潤減少、線維化改善に対して薬効を有することが確認され、抗IL-8抗体が子宮内膜症治療剤として有用であることが示された。この結果から、IL-8シグナル阻害剤が子宮内膜症の治療剤または予防剤として有用であることを当業者は当然に理解できよう。
子宮内膜症外科的誘引サルモデルにおいて、抗体投与開始から6ヶ月後の最終腹腔鏡観察後、剖検して採取した子宮を10%中性緩衝ホルマリン液で固定し、 H・E染色して観察した。子宮内において、子宮腺筋症と類似した子宮内膜組織がVehicle群で1例、抗体投与群で1例ずつ強く認められた。これら2例のカニクイザルで認められた子宮内膜組織は、子宮筋層内において子宮内膜腺とそれを囲む子宮内膜間質が存在し、ヒト子宮にみられる子宮腺筋症の病理学的特徴を有していたことからヒト子宮腺筋症併発モデルであると判断し、これら1例ずつの子宮筋層内の子宮内膜組織を比較した。図37に示すように、Vehicle群と比較し、抗体H1009/L395-F1974m投与群において子宮腺筋症病変部の内膜上皮の萎縮ならびにstroma細胞の減少と萎縮が認められた。本所見により、抗IL-8抗体が子宮腺筋症に対しても抑制効果があることが示された。この結果から、IL-8シグナル阻害剤が子宮腺筋症の治療剤または予防剤として有用であることを当業者は当然に理解できよう。
ヒトIL-8の受容体として、CXCR1およびCXCR2が知られている。子宮内膜症病変部において、IL-8が作用する細胞を調べるため、マウス抗ヒトCXCR1モノクローナル抗体(R&D Systems,カタログ番号:42705)ならびにマウス抗ヒトCXCR2モノクローナル抗体(Abcam, カタログ番号:19)を用いて、手術摘出ヒト子宮内膜症組織のパラフィン包埋ブロックを用いて免疫組織化学的染色を行った。これらのパラフィンブロックから薄切組織切片を作成し、上記抗CXCR1ならびに2抗体を一次抗体としてインキュベーションした後、二次抗体としてポリマー-HRPと結合したヤギ抗マウスIgG抗体(Dako)と反応させ、ジアミノベンジジン(和光純薬工業株式会社)で可視化した。その結果、表27に示すように、CXCR1とCXCR2がstroma細胞において陰性であることが確認された。一方で、組織内に浸潤している好中球やヘモジデリン沈着したマクロファージ、単核球においてはCXCR1, CXCR2ともに陽性であること、上皮細胞においても発現が弱陽性~陽性であることが確認された。また、血管内皮細胞においても、CXCR1, CXCR2ともに弱陽性が確認された。本所見は、子宮内膜症上皮細胞に対するIL-8の直接的な薬理作用と、好中球やマクロファージなどの浸潤免疫細胞を介した薬理作用がある可能性を示している。
上述の子宮内膜症外科的誘引カニクイザルモデルにおいて、誘引のための開腹手術後に生じた、子宮内膜症による癒着とは別の腹壁切開部位周囲への癒着が、誘引4ヶ月後(抗体投与2ヶ月前)の腹腔鏡観察時に全個体で見られた。外科的手術後の癒着に対する抗IL-8抗体の薬効を調べる為、個体番号を匿名化し、投与2ヶ月前の腹腔鏡観察所見と投与開始から6ヶ月後の最終腹腔鏡観察の写真を比較して、腹壁切開部位周囲への癒着の評価を実施した。具体的には腹壁切開部位周囲への癒着を投与前後で比較し、(1)改善(Improved)、(2)不変(Stable)、(3)悪化(Progressed)、の三段階評価を5人の評価者で行った。判定は評価者5人のうち3人以上で評価が一致したものを採用し、それ以外のものは(2)不変、とした。Vehicle群6頭と、抗体H1009/L395-F1974m投与群7頭について評価した。図38にVehicle群と抗体H1009/L395-F1974m投与群の代表的な腹腔内写真を示す。表28に示すように、Vehicle群では6頭のうち5頭が不変、1頭が悪化したのに対して、抗体H1009/L395-F1974m投与群では7頭のうち1頭が悪化したものの6頭が改善した。悪化した1頭は抗抗体が産生され血中のH1009/L395-F1974m抗体濃度が低下した個体であった。本所見により、抗IL-8抗体が外科的手術後の癒着改善治療薬あるいは癒着予防剤として有用であることが示唆された。この結果から、IL-8シグナル阻害剤が癒着治療剤または予防剤として有用であることを当業者は当然に理解できよう。
IL-8は好中球走化能を有し、損傷組織で産生されるIL-8は血管内皮細胞に接着した好中球の組織への遊走を促進し、好中球浸潤に伴う炎症が惹起されると言われている。表27で示す通り、子宮内膜症病変部位においても、好中球やマクロファージ、単核球の浸潤が確認され、これら免疫細胞においてCXCR1, CXCR2は発現がともに陽性であった。また、上皮細胞においても発現が弱陽性~陽性であったことから、子宮内膜症上皮細胞に対するIL-8の直接的な薬理作用と、好中球やマクロファージなどの浸潤免疫細胞を介した薬理作用がある可能性が示された。そこで、まず、好中球におけるIL-8に対する遊走性を評価した。好中球を健常人の末梢血から採取し、単離、精製を次のように行った。
ヘパリン処理した全血に1/5量のHetaSep(STEMCELL Technologies社製)を加え、製品プロトコルに従って、有核細胞を含む上清を回収後、ACK solution(GIBCO社製)を加えて溶血した。溶血後、0.1%BSA/PBSに懸濁し、細胞数を計測した。細胞数計測後、細胞数を調整し、EasySep(登録商標) Human Neutrophil Enrichment Kit (STEMCELL technologies社製)を使用して、製品プロトコルに従いEasySep(登録商標) Magnet(STEMCELL technologies社製)により好中球を単離した。単離後の好中球は、抗CD66b抗体、抗CD45RA抗体を使用し、FACSにより精製度に問題ないことを確認した。
単離した好中球は、CytoSelect(登録商標) 96-Well Cell Migration Assay(Cell Biolabs社製、カタログ番号:CBA-104)のMigration Plateを使用して遊走性を評価した。メンブレン下層のfeeder trayに1%FBS又は1%FBS+IL-8 (100 ng/mL)に、各種抗体、化合物、もしくは溶媒対照を添加したRPMI-1640培地(SIGMA社製)を150 μL加えた。評価対象として、抗体はH1009/L395-F1974mのほか、抗IL-8中和抗体であるAnti-IL-8抗体[807](abcam社製、カタログ番号:ab18672)、Anti-IL-8抗体[6217] (abcam社製、カタログ番号:ab10768)、陰性対照としてIgG1(シグマ社製、カタログ番号:I5154)をそれぞれ10 μg/mL、化合物はCXCR2阻害剤であるSRT3109(Shanghai Haoyuan Chemexpress社製、カタログ番号:HY-15462)を10 μM、溶媒対照としてPBS、DMSOを置いた。メンブレン上層のmembrane chamberには無血清のRPMI-1640培地に懸濁した好中球細胞を50000 cells/wellとなるよう播種した。2時間のincubation後、下層のfeeder trayに遊走した生細胞数(好中球)をCellTiter-Glo(登録商標)Luminescent Cell Viability Assay(Promega社製)を用いて相対化学発光量から検出した。IL-8を添加していないwellの相対化学発光量平均を1とし、各試薬添加時における相対化学発光量の相対値を算出した結果を図39に示した。
その結果、IL-8による好中球の遊走促進と、各種抗IL-8中和抗体およびCXCR2阻害剤による好中球の遊走阻害が確認された。本結果から、IL-8阻害剤のみならず、IL-8シグナル阻害に関与することが知られているCXCR1阻害剤やCXCR2阻害剤などのIL-8シグナル阻害剤が幅広く好中球遊走を抑制することが高い蓋然性をもってサポートされた。したがって、IL-8シグナル阻害剤は、子宮内膜症をはじめIL-8が高濃度である病変部に浸潤する好中球に対しても同様に浸潤抑制する事が高い蓋然性をもって示唆された。よって、当業者は、IL-8シグナル阻害剤が子宮内膜症ならびにIL-8が高濃度で知られる炎症性疾患を予防できることをこれらの結果から一般化し、拡張化して十分に理解できよう。
次に、子宮内膜症病変部に浸潤した好中球が子宮内膜症細胞に対しどのように働くかを調べた。子宮内膜症細胞は、次のように得た。手術摘出ヒト子宮内膜症組織を0.5 mg/mLコラーゲナーゼ、0.1 mg/mL DNase1を含むDMEM/F-12, HEPES(GIBCO社製)培地内で細切し、37℃でインキュベーションして細胞を回収した。細胞懸濁液を100 μmセルストレイナーに通した後、Lysing Buffer (BD Biosciences) により溶血した。溶血後、細胞を播種し、継代したものを子宮内膜症細胞として液体窒素中で凍結保存した。培養にはDMEM/F-12, HEPESに10%FBS, 2.5 μg/mLアムホテリシB, 100U/mLペニシリン, 100 μg/mLストレプトマイシンを添加したものを使用した。実験には、凍結保存した細胞を起眠して使用した。
上記の方法で得た子宮内膜症細胞に対し、好中球培養上清を添加し、子宮内膜症細胞における反応性を調べた。好中球培養上清は、好中球を単離後、1%FBSを含むRPMI-1640培地に懸濁し、6well plateに3.0 x106 cells/wellで播種し、1日培養した培養上清を回収した。96well plateに子宮内膜症細胞を10000 cells/wellで播種し、培地の半量を回収した好中球培養上清となるよう添加した。対照群(本実施例において、「Ctrl」ともいう。)には同様に培地の半量を、1%FBSを含むRPMI-1640培地となるよう添加した。8時間後、培地を除去し、RNeasy 96 Kit (QIAGEN社製)を使用し、製品プロトコルに従ってRNAを精製した。精製RNAはSuperScript(登録商標) VILO(登録商標) MasterMix(Invitrogen社製)を使用してcDNAを合成し、リアルタイムPCR法によりアロマターゼのmRNA発現量を比較した。アロマターゼのプローブには、TaqmanプローブのHs00903411_m1 (Thermo Fisher社製)を使用し、GAPDHの測定にはHuman GAPD(GAPDH) Endogenous Control(Applied Biosystems 社製、カタログ番号:4326317E)を使用した。
図40に、アロマターゼの発現量をGAPDHで補正後、Ctrlを1としたときのアロマターゼの相対発現量を示した。好中球培養上清を添加した子宮内膜症細胞では、アロマターゼの発現量の亢進が確認された。
アロマターゼはエストロゲン合成酵素であり、エストロゲンが子宮内膜症上皮細胞の増殖を促進することが知られている。好中球培養上清の添加により、子宮内膜症細胞中のアロマターゼ発現が亢進していたことから、好中球が子宮内膜症病変部に浸潤することで、子宮内膜症細胞におけるアロマターゼ発現量が亢進し、子宮内膜症上皮細胞の増殖が亢進する事が示唆された。したがって、IL-8シグナル阻害剤により、子宮内膜症病変部への好中球浸潤を抑制することで、子宮内膜症上皮細胞の増殖が抑制される事が推察される。子宮内膜症の外科的誘引サルモデルにおいても、抗体H1009/L395-F1974m投与群では増殖性を示す上皮細胞の萎縮が確認されたことからも、上記考察がサポートされた。
次に、好中球においてどのようなケモカインやサイトカイン、増殖因子が産生されているのかをIL-8添加・非添加時、抗IL-8抗体添加時で、解析した。単離した好中球を、1%FBSを含むRPMI-1640培地に懸濁し、6well plateに1.5 x106 cells/wellずつ播種した。これに、IL-8を添加・非添加、もしくはIL-8と各種抗体を添加し、1日培養した。抗体は、H1009/L395-F1974m、Anti-IL-8抗体[807](ab18672)、IgG1をそれぞれ終濃度10 μg/mL、IL-8は終濃度100 ng/mLとなるように添加した。1日後、細胞培養液を回収し、Cytokine Human Magnetic 30-Plex Panel for the Luminex(登録商標) platform(Thermo Fisher社製)を用いて、製品プロトコルに従い細胞培養液中の複数の各種サイトカイン、ケモカイン及び増殖因子の濃度を調べた。
その結果、図41に示すように、細胞培養液中のMCP-1(monocyte chemoattractant protein 1)濃度において、IL-8添加により強い上昇がみられ、抗IL-8中和抗体により上昇が抑制された。
MCP-1はCCL2とも呼ばれ、単球走化能を有し、炎症局所への単球の遊走及び浸潤を促進すると言われている。表26で示した通り、子宮内膜症の外科的誘引サルモデルの結節性病変部において、ヘモジデリンが抗体H1009/L395-F1974m投与群において減少していたことから、マクロファージあるいは単球が抗体投与によって浸潤抑制されている事が示唆された。本結果により、好中球をIL-8刺激することで単球走化能を持つMCP-1が産生されたことから、IL-8により子宮内膜症病変部に好中球が浸潤し、さらにMCP-1を産生することで、単球およびマクロファージがさらに病変部位へ遊走、浸潤する可能性が示された。
さらに、MCP-1は、線維芽細胞に働きコラーゲン産生を促進する働きが知られており(J Biol Chem. 1996 Jul 26;271(30):17779-84.)、組織の線維化を亢進する事がin vivoにおいても多数報告されている(J Immunol. 1994 Nov 15;153(10):4733-41、Am J Physiol Lung Cell Mol Physiol. 2004 May;286(5):L1038-44、J Invest Dermatol. 2003 Sep;121(3):510-6.)。IL-8により子宮内膜症病変部に好中球が浸潤し、さらに好中球がMCP-1を産生することで、単球およびマクロファージの遊走、浸潤の促進に加えて、子宮内膜症病変部の線維化に対しても促進する可能性が示された。本結果から、IL-8シグナル阻害剤により、好中球浸潤を抑制することにより、子宮内膜症病変部における線維化が改善される事が示唆された。実際に、子宮内膜症外科的誘引サルモデルの結節性病変部においても、図36および表26で示した通り、抗体H1009/L395-F1974m投与群において膠原線維を主体とするinterstitiumの減少が確認され、線維化改善に対して薬効を有することが確認された。以上のことから、IL-8シグナル阻害剤が子宮内膜症の線維化に対して有用であることが高い蓋然性をもってサポートされた。当業者は、IL-8シグナル阻害剤が、子宮内膜症の線維化、さらには各種線維化症に対しても治療または予防できることをこれらの結果から一般化し、拡張化して十分に理解できよう。
(24-1)術後癒着誘引サルモデルの作製
術後癒着誘引モデルを作製するために、9-16歳齢のメスのカニクイザル(医薬基盤・健康・栄養研究所 霊長類医科学研究センターより入手)に対し、麻酔下で以下の作業を行った。麻酔はケタミンキシラジンのバランス麻酔(2:1の比率を目安に混合)とイソフルランを用いた。腹壁正中線を5 cm~6 cm切開し開腹後、子宮体部を1 cm切開し単糸で4針縫合した後ガーゼで子宮全体を5回拭った。次に切開した腹壁正中線より2 cmの右腹壁腹膜部分を1 cm切開し、吸収糸にて4針連続縫合した。また、縫合後はガーゼで縫合部分を5回拭った。同様に左腹壁腹膜も切開、縫合、ガーゼで拭った。最後は腹壁正中線を連続縫合で10針~11針縫合し、すべての作業を1時間以内で終了した。術後の覚醒時には拮抗薬のアンチセダン(キシラジン量と同量を目安に)と抗生剤のセフラジンを投与した。また、鎮痛剤のザルバンを術後2日間投与し、毎日の定期観察で術後の異常がないことを確認した。なお腹腔鏡観察時も同様に麻酔と術後管理を実施した。
抗IL-8抗体投与群の動物に対し、上述の術後癒着誘引手術後、1時間以内に抗IL-8抗体であるH1009/L395-F1974mを10 mg/kg静脈内投与した。コントロール群は無処置とした。抗IL-8抗体の投与から24日~35日後に腹腔鏡観察を行い、切開、縫合、ガーゼの拭う作業によって形成された癒着を観察した。腹腔鏡観察は、腹部正中を麻酔下で切開し、トローカーを刺入し、腹腔鏡を挿入した。腹腔鏡はビデオシステム(カールストルツ社製)とモニターに接続され、記録はビデオシステムで、腹腔内の観察はモニターで行った。癒着のサイズは目盛りのついたバーあるいは目盛りのついた鉗子を用いて測定した。癒着の有無、範囲および癒着の場所を腹腔鏡観察時に評価した。結果を表29に示す。腹腔鏡観察の結果、無処置群の2頭のサルで、切開部位全てにおいて癒着が確認された。抗IL-8抗体投与群において、個体No.206では癒着が認められたものの、個体No.201において左右腹壁腹膜と子宮切開部で癒着が認められず、抗IL-8抗体による癒着予防効果が示された。この結果から、抗IL-8抗体が癒着予防剤として有用であることが示された。当業者であれば、投与時点等を適宜変更することで、抗IL-8抗体による、より高い癒着形成予防効果が発揮され得ることを当然に理解できよう。また、この結果により、IL-8シグナル阻害剤が癒着予防剤として有用であることを、当業者は当然に理解できよう。
健常人末梢血よりCD14陽性画分として分離された単球(All Cells社、カタログ番号:PB011-P-F-2)を購入し、-80 ℃で保管した。実験に使用する際、次のように細胞を解凍し使用した。37 ℃ウォーターバスで解凍した細胞にDNaseI(STEMCELL Technologies社製、カタログ番号:07900)および10 % FBSを含む培地を添加し、高速遠心機で遠心した後、上清を除いた。この作業を繰り返した後、適量の培地に細胞を懸濁し、細胞数を計測した。単球は、in vitroでマクロファージへの分化培養を行い、IL-8存在下および非存在下でのConnecting tissue growth factor(CTGF)の発現変化を調べた。CTGFは線維化を促進する因子である。単球は10 %FBS、および25 ng/mL GM-CSF(SIGMA社製)を含むRPMI-1640(SIGMA社製)培地に懸濁し、培養用プレートに播種した。2日または3日おきに培養上清を半量ずつ交換し、7日間培養した。7日後、培養上清を除去し、10 %FBSおよび20 ng/mL IFN-γ(SIGMA社製)、10 pg/mL LPS(和光純薬工業社製)を含むRPMI-1640培地に100 ng/mLのIL-8と各種抗体を添加して3日間培養した。
評価した抗体は、H1009/L395-F1974m抗体、Anti-IL-8抗体[807](abcam社製、カタログ番号:ab18672)、Anti-IL-8抗体[6217](abcam社製、カタログ番号:ab10768)、Anti-IL-8抗体(Becton Dickinson社製、カタログ番号:BD554726)、Anti-IL-8抗体(Becton Dickinson社製、カタログ番号:BD555717)で、10 μg/mLの濃度で使用した。陰性対象としてIgG1(SIGMA社製、カタログ番号:I5154)を10 μg/mLの濃度で使用した。
培養3日後、細胞を回収しRNeasy Mini Kit(QIAGEN社製)を使用し、製品プロトコルに従ってRNAを精製した。精製RNAはSuperScript VILO MasterMix(Thermo Fisher社製)を使用してcDNAを合成し、リアルタイムPCR法によりCTGFのmRNA発現量を評価した。CTGFのプローブには、TaqmanプローブのHs00170014_m1(Thermo Fisher社製)を使用し、18SrRNAの測定にはEukaryotic 18S rRNA Endogenous Control(Thermo Fisher社製、カタログ番号:E4319413E)を使用した。
図42に、CTGFの発現量を18S rRNAで補正した値を示す。IL-8無添加条件では、単球から分化させたマクロファージにおいてCTGFの発現が全く認められなかった。一方、IL-8を添加した条件では、分化させたマクロファージにおいてCTGF発現上昇が認められた。この結果から、IL-8は単球から分化させたマクロファージに対して、CTGFの発現上昇を誘導する機能を持つことが示された。さらに、IL-8と同時に抗IL-8抗体を加えると、CTGFの発現上昇は認められなかった。IL-8によるCTGFの発現上昇は、様々な抗IL-8抗体によって抑制されることが示された。この結果から、IL-8が線維化の進行に関与する因子であること、並びに、抗IL-8抗体等のIL-8シグナル阻害剤が線維化疾患に対する治療または予防剤として有用であることを、当業者は当然に理解できよう。
QuikChange Site-Directed Mutagenesis Kit(Stratagene)を用いて、添付説明書記載の方法で作製された変異体を含むプラスミド断片を動物細胞発現ベクターに挿入することによって、目的のH鎖発現ベクター及びL鎖発現ベクターが作製された。得られた発現ベクターの塩基配列は当業者に公知の方法で決定された。
抗体の発現は以下の方法を用いて行われた。ヒト胎児腎癌細胞由来HEK293H株(Invitrogen)を10 % Fetal Bovine Serum(Invitrogen)を含むDMEM培地(Invitrogen)へ懸濁し、5~6 × 105細胞/mLの細胞密度で接着細胞用ディッシュ(直径10 cm, CORNING)の各ディッシュへ10 mLずつ蒔きこみCO2インキュベーター(37℃、5 % CO2)内で一昼夜培養した後に、培地を吸引除去し、CHO-S-SFM-II(Invitrogen)培地6.9 mLを添加した。調製したプラスミドをlipofection法により細胞へ導入した。得られた培養上清を回収した後、遠心分離(約2000 g、5分間、室温)して細胞を除去し、さらに0.22μmフィルターMILLEX(R)-GV(Millipore)を通して滅菌して培養上清を得た。得られた培養上清にrProtein A SepharoseTM Fast Flow(Amersham Biosciences)を用いて当業者に公知の方法で精製した。精製抗体の濃度は、分光光度計を用いて280 nmでの吸光度を測定した。得られた値からProtein Science 1995 ; 4 : 2411-2423に記された方法により算出された吸光係数を用いて抗体濃度を算出した。
Claims (15)
- ヒトIL-8に結合する単離された抗IL-8抗体を有効成分として含有する、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物であって、
前記抗IL-8抗体は以下(1)から(7)からなる群から選択される、組成物:
(1)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3;
(2)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:25のアミノ酸配列の3位のチロシン、配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む;
(3)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の9位のチロシンと11位のアルギニン、及び配列番号:25のアミノ酸配列の3位のチロシンでアミノ酸の置換をそれぞれ少なくとも含む;
(4)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の6位のアラニン、8位のグリシン、9位のチロシンと11位のアルギニン、及び配列番号:25のアミノ酸配列の3位のチロシンでアミノ酸の置換をそれぞれ少なくとも含む;
(5)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、及び配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む;
(6)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の9位のチロシンと11位のアルギニン、配列番号:25のアミノ酸配列の3位のチロシン、配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、及び配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む;並びに、
(7)以下(a)から(f)の少なくとも一つにおいて、少なくとも一つのアミノ酸の置換を含む、pH依存的にIL-8に結合する、抗IL-8抗体:
(a)配列番号:23のアミノ酸配列を含むHVR-H1、
(b)配列番号:24のアミノ酸配列を含むHVR-H2、
(c)配列番号:25のアミノ酸配列を含むHVR-H3、
(d)配列番号:26のアミノ酸配列を含むHVR-L1、
(e)配列番号:27のアミノ酸配列を含むHVR-L2、及び
(f)配列番号:28のアミノ酸配列を含むHVR-L3
ここで、当該抗IL-8抗体は配列番号:24のアミノ酸配列の6位のアラニン、8位のグリシン、9位のチロシンと11位のアルギニン、配列番号:25のアミノ酸配列の3位のチロシン、配列番号:27のアミノ酸配列の1位のアスパラギンと5位のロイシン、及び配列番号:28のアミノ酸配列の1位のグルタミンでアミノ酸の置換をそれぞれ少なくとも含む。 - 抗IL-8抗体は以下(8)から(19)からなる群から選択される、請求項1に記載の組成物:
(8)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:29のアミノ酸配列を含み、及び(c)HVR-H3として配列番号:30のアミノ酸配列を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(9)(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:31のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(10)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:24のアミノ酸配列を含み、及び(c)HVR-H3として配列番号:30のアミノ酸配列を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(11)(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:107のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(12)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:29のアミノ酸配列を含み、(c)HVR-H3として配列番号:30のアミノ酸配列を含み、(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:31のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(13)(a)HVR-H1として配列番号:23のアミノ酸配列を含み、(b)HVR-H2として配列番号:24のアミノ酸配列を含み、(c)HVR-H3として配列番号:30のアミノ酸配列を含み、(d)HVR-L1として配列番号:26のアミノ酸配列を含み、(e)HVR-L2として配列番号:107のアミノ酸配列を含み、及び(f)HVR-L3として配列番号:32のアミノ酸配列を含む、請求項1の(1)又は(2)に記載の抗IL-8抗体;
(14)配列番号:34のアミノ酸配列を含む重鎖可変領域及び配列番号:35のアミノ酸配列を含む軽鎖可変領域を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(15)配列番号:108のアミノ酸配列を含む重鎖可変領域及び配列番号:109のアミノ酸配列を含む軽鎖可変領域を含む、請求項1の(1)又は(2)に記載の抗IL-8抗体;
(16)(a)配列番号:23のアミノ酸配列を含むHVR-H1、(b)配列番号:29のアミノ酸配列を含むHVR-H2、(c)配列番号:30のアミノ酸配列を含むHVR-H3、(d)配列番号:26のアミノ酸配列を含むHVR-L1、(e)配列番号:31のアミノ酸配列を含むHVR-L2、及び(f)配列番号:32のアミノ酸配列を含むHVR-L3とそれぞれ少なくとも80%の配列同一性を有するHVR-H1、HVR-H2、HVR-H3、HVR-L1、HVR-L2、及びHVR-L3を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(17)(a)配列番号:23のアミノ酸配列を含むHVR-H1、(b)配列番号:24のアミノ酸配列を含むHVR-H2、(c)配列番号:30のアミノ酸配列を含むHVR-H3、(d)配列番号:26のアミノ酸配列を含むHVR-L1、(e)配列番号:107のアミノ酸配列を含むHVR-L2、及び(f)配列番号:32のアミノ酸配列を含むHVR-L3とそれぞれ少なくとも80%の配列同一性を有するHVR-H1、HVR-H2、HVR-H3、HVR-L1、HVR-L2、及びHVR-L3を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;
(18)配列番号:34のアミノ酸配列を含む重鎖可変領域及び配列番号:35のアミノ酸配列を含む軽鎖可変領域とそれぞれ少なくとも80%の配列同一性を有する重鎖可変領域及び軽鎖可変領域を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体;並びに、
(19)配列番号:108のアミノ酸配列を含む重鎖可変領域及び配列番号:109のアミノ酸配列を含む軽鎖可変領域とそれぞれ少なくとも80%の配列同一性を有する重鎖可変領域及び軽鎖可変領域を含む、請求項1の(1)から(7)のいずれかに記載の抗IL-8抗体。 - ヒトIL-8に結合する単離された抗IL-8抗体を有効成分として含有する、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物であって、
当該抗IL-8抗体は以下(I)から(VIII)からなる群から選択される、組成物:
(I)EUナンバリングで表される、235位、236位、239位、327位、330位、331位、428位、434位、436位、438位、及び440位からなる群から選択される一つ以上の位置にアミノ酸の置換を含むFc領域を含む、抗IL-8抗体;
(II)EUナンバリングで表される、434位、438位、及び440位からなる群から選択される全ての位置にアミノ酸の置換を含むFc領域を含む、抗IL-8抗体;
(III)EUナンバリングで表される、434位にAla;438位にGlu、Arg、Ser若しくはLys;及び、440位にGlu、Asp若しくはGlnの全てのアミノ酸置換を含むFc領域を含む、抗IL-8抗体;
(IV)EUナンバリングで表される、434位にAla;438位にGlu、Arg、Ser若しくはLys;及び、440位にGlu、Asp若しくはGlnの全てのアミノ酸置換を含み、さらに、428位にIle若しくはLeu、及び/又は、436位にIle、Leu、Val、Thr若しくはPheを含むFc領域を含む、抗IL-8抗体;
(V)EUナンバリングで表される、L235R, G236R, S239K, A327G, A330S, P331S, M428L, N434A, Y436T, Q438R, 及びS440Eからなる群から選択されるアミノ酸置換を一つ以上含むFc領域を含む、抗IL-8抗体;
(VI)EUナンバリングで表される、
N434A/Q438R/S440E;N434A/Q438R/S440D;
N434A/Q438K/S440E;N434A/Q438K/S440D;
N434A/Y436T/Q438R/S440E;N434A/Y436T/Q438R/S440D;
N434A/Y436T/Q438K/S440E;N434A/Y436T/Q438K/S440D;
N434A/Y436V/Q438R/S440E;N434A/Y436V/Q438R/S440D;
N434A/Y436V/Q438K/S440E;N434A/Y436V/Q438K/S440D;
N434A/R435H/F436T/Q438R/S440E;N434A/R435H/F436T/Q438R/S440D;
N434A/R435H/F436T/Q438K/S440E;N434A/R435H/F436T/Q438K/S440D;
N434A/R435H/F436V/Q438R/S440E;N434A/R435H/F436V/Q438R/S440D;
N434A/R435H/F436V/Q438K/S440E;N434A/R435H/F436V/Q438K/S440D;
M428L/N434A/Q438R/S440E;M428L/N434A/ Q438R/S440D;
M428L/N434A/Q438K/S440E;M428L/N434A/ Q438K/S440D;
M428L/N434A/Y436T/Q438R/S440E;M428L/N434A/Y436T/Q438R/S440D;
M428L/N434A/Y436T/Q438K/S440E;M428L/N434A/Y436T/Q438K/S440D;
M428L/N434A/Y436V/Q438R/S440E;M428L/N434A/Y436V/Q438R/S440D;
M428L/N434A/Y436V/Q438K/S440E;及び、M428L/N434A/Y436V/Q438K/S440D
からなる群から選択されるアミノ酸置換の組み合わせを含むFc領域を含む、抗IL-8抗体;
(VII)EUナンバリングで表される、
L235R/G236R/S239K/M428L/N434A/Y436T/Q438R/S440E;又は、
L235R/G236R/A327G/A330S/P331S/M428L/N434A/Y436T/Q438R/S440E
のアミノ酸置換の組み合わせを含むFc領域を含む、抗IL-8抗体;並びに、
(VIII)前記(I)から(VII)のいずれかに記載のFc領域を含み、当該Fc領域が以下の(a)から(e)からなる群から選択される性質の少なくとも一つを有する抗IL-8抗体:
(a)酸性pHでの、当該Fc領域のFcRnに対する結合親和性が、天然型のFc領域のFcRnに対する結合親和性よりも増大している、
(b)既存のADAに対する当該Fc領域の結合親和性が天然型のFc領域の既存のADAに対する結合親和性よりも低下している、
(c) 当該Fc領域の血漿中半減期が天然型のFc領域の血漿中半減期よりも増大している、
(d) 当該Fc領域の血漿中クリアランスが天然型のFc領域の血漿中クリアランスよりも減少している、及び
(e) 当該Fc領域のエフェクター受容体に対する結合親和性が天然型のFc領域のエフェクター受容体に対する結合親和性よりも低下している。 - ヒトIL-8に結合する単離された抗IL-8抗体を有効成分として含有する、子宮内膜症;子宮腺筋症;月経困難症;癒着;線維化疾患;子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物であって、
当該抗IL-8抗体は以下(A)から(F)からなる群から選択される、組成物:
(A)配列番号:36のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖を含む抗IL-8抗体;
(B)配列番号:37のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖を含む抗IL-8抗体;
(C)配列番号:106のアミノ酸配列を含む重鎖及び配列番号:44のアミノ酸配列を含む軽鎖を含む抗IL-8抗体;
(D)配列番号:36のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖とそれぞれ少なくとも80%の配列同一性を有する重鎖及び軽鎖を含む抗IL-8抗体;
(E)配列番号:37のアミノ酸配列を含む重鎖及び配列番号:38のアミノ酸配列を含む軽鎖とそれぞれ少なくとも80%の配列同一性を有する重鎖及び軽鎖を含む抗IL-8抗体;並びに、
(F)配列番号:106のアミノ酸配列を含む重鎖及び配列番号:44のアミノ酸配列を含む軽鎖とそれぞれ少なくとも80%の配列同一性を有する重鎖及び軽鎖を含む抗IL-8抗体。 - 線維化疾患が、子宮内膜症又は子宮腺筋症における線維化、慢性閉塞肺疾患(COPD)、嚢胞性線維症、乾癬、肝線維症、腎線維症、及び肺線維症からなる群から選択される、請求項1~4のいずれか一項に記載の組成物。
- IL-8シグナル阻害剤を有効成分として含有する、ヒト子宮内膜症;子宮腺筋症;月経困難症;癒着;ヒト子宮内膜症、子宮腺筋症、若しくは月経困難症における疼痛;不妊症;及び、癒着、線維化若しくは炎症による疼痛からなる群から選択されるIL-8関連疾患の治療用又は予防用組成物。
- 免疫細胞のIL-8関連疾患の病変部又はその周辺環境への浸潤を抑制するための、請求項6に記載の組成物。
- アロマターゼ又は線維化因子の産生を阻害するための、請求項6又は7に記載の組成物。
- 性周期への影響を与えない、請求項6~8のいずれか一項に記載の組成物。
- 子宮内膜症又は子宮腺筋症における癒着又は線維化を抑制させるための、請求項6~9のいずれか一項に記載の組成物。
- (1)子宮内膜症又は子宮腺筋症の患者において子宮内膜の上皮細胞若しくはstroma細胞を萎縮させるための、又は、(2)子宮内膜症において子宮内膜のinterstitiumを減少させるための、請求項6~10のいずれか一項に記載の組成物。
- 月経困難症が子宮内膜症若しくは子宮腺筋症を有する月経困難症であるか、又は、子宮内膜症若しくは子宮腺筋症が疑われる月経困難症である、請求項6~11のいずれか一項に記載の組成物。
- 癒着が手術後に生じた癒着である、請求項6に記載の組成物。
- IL-8シグナル阻害剤がIL-8阻害剤、CXCR1阻害剤、又はCXCR2阻害剤である、請求項6~13のいずれか一項に記載の組成物。
- IL-8阻害剤、CXCR1阻害剤、又はCXCR2阻害剤が、それぞれ抗IL-8抗体、抗CXCR1抗体、又は抗CXCR2抗体である、請求項14に記載の組成物。
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IL264626A IL264626B (en) | 2016-08-05 | 2017-08-04 | A preparation for the prevention or treatment of diseases related to il-8 |
| JP2018531994A JP6527643B2 (ja) | 2016-08-05 | 2017-08-04 | Il−8関連疾患の治療用又は予防用組成物 |
| US16/323,142 US11053308B2 (en) | 2016-08-05 | 2017-08-04 | Method for treating IL-8-related diseases |
| MX2019001448A MX2019001448A (es) | 2016-08-05 | 2017-08-04 | Composicion para profilaxis o tratamiento de enfermedades relacionadas con interleucina 8 (il-8). |
| BR112019001902A BR112019001902A2 (pt) | 2016-08-05 | 2017-08-04 | composição para profilaxia ou tratamento de doenças relacionadas à il-8 |
| CN202310193929.XA CN116251182A (zh) | 2016-08-05 | 2017-08-04 | 用于预防或治疗il-8相关疾病的组合物 |
| CN201780046902.0A CN109689099B (zh) | 2016-08-05 | 2017-08-04 | 用于预防或治疗il-8相关疾病的组合物 |
| AU2017305073A AU2017305073B2 (en) | 2016-08-05 | 2017-08-04 | Composition for prevention or treatment of IL-8 related diseases |
| KR1020237017761A KR20230079499A (ko) | 2016-08-05 | 2017-08-04 | Il-8 관련 질환의 치료용 또는 예방용 조성물 |
| KR1020177027051A KR102102734B1 (ko) | 2016-08-05 | 2017-08-04 | Il-8 관련 질환의 치료용 또는 예방용 조성물 |
| KR1020207009872A KR102538749B1 (ko) | 2016-08-05 | 2017-08-04 | Il-8 관련 질환의 치료용 또는 예방용 조성물 |
| CN202310196594.7A CN116271014A (zh) | 2016-08-05 | 2017-08-04 | 用于预防或治疗il-8相关疾病的组合物 |
| RU2019105510A RU2766112C2 (ru) | 2016-08-05 | 2017-08-04 | Композиция для профилактики или лечения связанных с il-8 заболеваний |
| SG11201801024XA SG11201801024XA (en) | 2016-08-05 | 2017-08-04 | Therapeutic or preventive compositions for il-8-related diseases |
| EP17837083.9A EP3494991A4 (en) | 2016-08-05 | 2017-08-04 | COMPOSITION FOR PREVENTING OR TREATING DISEASES RELATING TO IL-8 |
| CA3026050A CA3026050A1 (en) | 2016-08-05 | 2017-08-04 | Composition for prophylaxis or treatment of il-8 related diseases |
| US17/333,256 US11780912B2 (en) | 2016-08-05 | 2021-05-28 | Composition for prophylaxis or treatment of IL-8 related diseases |
| US18/450,863 US20240083996A1 (en) | 2016-08-05 | 2023-08-16 | Composition for prophylaxis or treatment of il-8 related diseases |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2016154174 | 2016-08-05 | ||
| JP2016-154174 | 2016-08-05 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US16/323,142 A-371-Of-International US11053308B2 (en) | 2016-08-05 | 2017-08-04 | Method for treating IL-8-related diseases |
| US17/333,256 Division US11780912B2 (en) | 2016-08-05 | 2021-05-28 | Composition for prophylaxis or treatment of IL-8 related diseases |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018025982A1 true WO2018025982A1 (ja) | 2018-02-08 |
Family
ID=61072950
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2017/028346 WO2018025982A1 (ja) | 2016-08-05 | 2017-08-04 | Il-8関連疾患の治療用又は予防用組成物 |
Country Status (14)
| Country | Link |
|---|---|
| US (3) | US11053308B2 (ja) |
| EP (1) | EP3494991A4 (ja) |
| JP (3) | JP6527643B2 (ja) |
| KR (3) | KR102102734B1 (ja) |
| CN (3) | CN116271014A (ja) |
| AU (1) | AU2017305073B2 (ja) |
| BR (1) | BR112019001902A2 (ja) |
| CA (1) | CA3026050A1 (ja) |
| IL (1) | IL264626B (ja) |
| MX (2) | MX2019001448A (ja) |
| RU (1) | RU2766112C2 (ja) |
| SG (1) | SG11201801024XA (ja) |
| TW (2) | TWI693940B (ja) |
| WO (1) | WO2018025982A1 (ja) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10604561B2 (en) | 2016-09-16 | 2020-03-31 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies, polypeptides containing variant Fc regions, and methods of use |
| WO2020213706A1 (en) * | 2019-04-19 | 2020-10-22 | Chugai Seiyaku Kabushiki Kaisha | Antibodies and compositions for use in detecting or capturing a polypeptide in a sample, and methods for detecting or capturing a polypeptide in a sample |
| US10919953B2 (en) | 2012-08-24 | 2021-02-16 | Chugai Seiyaku Kabushiki Kaisha | FcgammaRIIB-specific Fc region variant |
| US11046784B2 (en) | 2006-03-31 | 2021-06-29 | Chugai Seiyaku Kabushiki Kaisha | Methods for controlling blood pharmacokinetics of antibodies |
| US11053308B2 (en) | 2016-08-05 | 2021-07-06 | Chugai Seiyaku Kabushiki Kaisha | Method for treating IL-8-related diseases |
| US11180548B2 (en) | 2015-02-05 | 2021-11-23 | Chugai Seiyaku Kabushiki Kaisha | Methods of neutralizing IL-8 biological activity |
| US11236168B2 (en) | 2012-08-24 | 2022-02-01 | Chugai Seiyaku Kabushiki Kaisha | Mouse FcγammaRII-specific Fc antibody |
| US11248053B2 (en) | 2007-09-26 | 2022-02-15 | Chugai Seiyaku Kabushiki Kaisha | Method of modifying isoelectric point of antibody via amino acid substitution in CDR |
| US11267868B2 (en) | 2013-04-02 | 2022-03-08 | Chugai Seiyaku Kabushiki Kaisha | Fc region variant |
| US11359194B2 (en) | 2008-04-11 | 2022-06-14 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule capable of binding two or more antigen molecules repeatedly |
| US11359009B2 (en) | 2015-12-25 | 2022-06-14 | Chugai Seiyaku Kabushiki Kaisha | Anti-myostatin antibodies and methods of use |
| US11454633B2 (en) | 2014-12-19 | 2022-09-27 | Chugai Seiyaku Kabushiki Kaisha | Anti-myostatin antibodies, polypeptides containing variant Fc regions, and methods of use |
| US11891434B2 (en) | 2010-11-30 | 2024-02-06 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule capable of binding to plurality of antigen molecules repeatedly |
| US11891432B2 (en) | 2018-03-15 | 2024-02-06 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies having cross-reactivity to Zika virus and methods of use |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4136106A1 (en) * | 2020-04-17 | 2023-02-22 | Zoetis Services LLC | Canine antibody variants |
| CA3176321A1 (en) * | 2020-04-22 | 2021-10-28 | Peng Zhang | Anti-cd73-anti-pd-1 bispecific antibody and use thereof |
| WO2022060736A1 (en) * | 2020-09-15 | 2022-03-24 | Aristea Therapeutics, Inc. | Compositions and methods for the treatment of palmoplantar pustulosis |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08217799A (ja) * | 1994-07-13 | 1996-08-27 | Chugai Pharmaceut Co Ltd | ヒトインターロイキン−8に対する再構成ヒト抗体 |
| WO2009026117A2 (en) * | 2007-08-16 | 2009-02-26 | Glaxo Group Limited | Novel compounds |
| JP2009541352A (ja) * | 2006-06-23 | 2009-11-26 | スミスクライン・ビーチャム・コーポレイション | 治療方法 |
| JP2013518606A (ja) * | 2010-02-09 | 2013-05-23 | グラクソスミスクライン・リミテッド・ライアビリティ・カンパニー | 新規使用 |
| WO2016125495A1 (en) * | 2015-02-05 | 2016-08-11 | Chugai Seiyaku Kabushiki Kaisha | Antibodies comprising an ion concentration dependent antigen-binding domain, fc region variants, il-8-binding antibodies, and uses therof |
| WO2017046994A1 (en) * | 2015-09-18 | 2017-03-23 | Chugai Seiyaku Kabushiki Kaisha | Il-8-binding antibodies and uses thereof |
Family Cites Families (490)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4769320A (en) | 1982-07-27 | 1988-09-06 | New England Medical Center Hospitals, Inc. | Immunoassay means and methods useful in human native prothrombin and human abnormal prothorombin determinations |
| ES521371A0 (es) | 1982-04-12 | 1984-05-16 | Hybritech Inc | Un procedimiento para la purificacion de un anticuerpo. |
| US4689299A (en) | 1982-09-30 | 1987-08-25 | University Of Rochester | Human monoclonal antibodies against bacterial toxins |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| JPS6132803A (ja) | 1984-07-25 | 1986-02-15 | Electric Power Dev Co Ltd | 架空線への光フアイバケ−ブル巻付機 |
| US4737456A (en) | 1985-05-09 | 1988-04-12 | Syntex (U.S.A.) Inc. | Reducing interference in ligand-receptor binding assays |
| US4676980A (en) | 1985-09-23 | 1987-06-30 | The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Target specific cross-linked heteroantibodies |
| US6548640B1 (en) | 1986-03-27 | 2003-04-15 | Btg International Limited | Altered antibodies |
| JPH06104071B2 (ja) | 1986-08-24 | 1994-12-21 | 財団法人化学及血清療法研究所 | 第▲ix▼因子コンホメ−シヨン特異性モノクロ−ナル抗体 |
| US4801687A (en) | 1986-10-27 | 1989-01-31 | Bioprobe International, Inc. | Monoclonal antibody purification process using protein A |
| IL85035A0 (en) | 1987-01-08 | 1988-06-30 | Int Genetic Eng | Polynucleotide molecule,a chimeric antibody with specificity for human b cell surface antigen,a process for the preparation and methods utilizing the same |
| DE3883899T3 (de) | 1987-03-18 | 1999-04-22 | Sb2 Inc | Geänderte antikörper. |
| US5004697A (en) | 1987-08-17 | 1991-04-02 | Univ. Of Ca | Cationized antibodies for delivery through the blood-brain barrier |
| US5606040A (en) | 1987-10-30 | 1997-02-25 | American Cyanamid Company | Antitumor and antibacterial substituted disulfide derivatives prepared from compounds possessing a methyl-trithio group |
| US5770701A (en) | 1987-10-30 | 1998-06-23 | American Cyanamid Company | Process for preparing targeted forms of methyltrithio antitumor agents |
| US5322678A (en) | 1988-02-17 | 1994-06-21 | Neorx Corporation | Alteration of pharmacokinetics of proteins by charge modification |
| AU634186B2 (en) | 1988-11-11 | 1993-02-18 | Medical Research Council | Single domain ligands, receptors comprising said ligands, methods for their production, and use of said ligands and receptors |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5202253A (en) | 1988-12-30 | 1993-04-13 | Oklahoma Medical Research Foundation | Monoclonal antibody specific for protein C and antibody purification method |
| CA2006684C (en) | 1988-12-30 | 1996-12-17 | Charles T. Esmon | Monoclonal antibody against protein c |
| DE3920358A1 (de) | 1989-06-22 | 1991-01-17 | Behringwerke Ag | Bispezifische und oligospezifische, mono- und oligovalente antikoerperkonstrukte, ihre herstellung und verwendung |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| CA2026147C (en) | 1989-10-25 | 2006-02-07 | Ravi J. Chari | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| US5959177A (en) | 1989-10-27 | 1999-09-28 | The Scripps Research Institute | Transgenic plants expressing assembled secretory antibodies |
| JPH0636741B2 (ja) | 1989-11-08 | 1994-05-18 | 帝人株式会社 | ヒト・プロテインcの分離方法 |
| US6075181A (en) | 1990-01-12 | 2000-06-13 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| US6150584A (en) | 1990-01-12 | 2000-11-21 | Abgenix, Inc. | Human antibodies derived from immunized xenomice |
| EP0515571B1 (en) | 1990-02-16 | 1998-12-02 | Boston Biomedical Research Institute | Hybrid reagents capable of selectively releasing molecules into cells |
| US5130129A (en) | 1990-03-06 | 1992-07-14 | The Regents Of The University Of California | Method for enhancing antibody transport through capillary barriers |
| JP2909971B2 (ja) | 1990-06-06 | 1999-06-23 | 三菱電機ホーム機器株式会社 | 布団乾燥機用シート付マット |
| US5770429A (en) | 1990-08-29 | 1998-06-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| GB9022547D0 (en) | 1990-10-17 | 1990-11-28 | Wellcome Found | Purified immunoglobulin |
| DE69129154T2 (de) | 1990-12-03 | 1998-08-20 | Genentech Inc | Verfahren zur anreicherung von proteinvarianten mit geänderten bindungseigenschaften |
| US5571894A (en) | 1991-02-05 | 1996-11-05 | Ciba-Geigy Corporation | Recombinant antibodies specific for a growth factor receptor |
| JP3370324B2 (ja) | 1991-04-25 | 2003-01-27 | 中外製薬株式会社 | ヒトインターロイキン−6受容体に対する再構成ヒト抗体 |
| EP0590058B1 (en) | 1991-06-14 | 2003-11-26 | Genentech, Inc. | HUMANIZED Heregulin ANTIBODy |
| GB9114948D0 (en) | 1991-07-11 | 1991-08-28 | Pfizer Ltd | Process for preparing sertraline intermediates |
| JPH06104071A (ja) | 1991-08-30 | 1994-04-15 | Jasco Corp | 発熱体セラミックス溶接加工方法 |
| JP3951062B2 (ja) | 1991-09-19 | 2007-08-01 | ジェネンテック・インコーポレーテッド | 少なくとも遊離のチオールとして存在するシステインを有する抗体フラグメントの大腸菌での発現、2官能性F(ab’)2抗体の産生のための使用 |
| US5587458A (en) | 1991-10-07 | 1996-12-24 | Aronex Pharmaceuticals, Inc. | Anti-erbB-2 antibodies, combinations thereof, and therapeutic and diagnostic uses thereof |
| WO1993008829A1 (en) | 1991-11-04 | 1993-05-13 | The Regents Of The University Of California | Compositions that mediate killing of hiv-infected cells |
| ATE419355T1 (de) | 1992-02-06 | 2009-01-15 | Novartis Vaccines & Diagnostic | Marker für krebs und biosynthetisches bindeprotein dafür |
| GB9203459D0 (en) | 1992-02-19 | 1992-04-08 | Scotgen Ltd | Antibodies with germ-line variable regions |
| EP0640094A1 (en) | 1992-04-24 | 1995-03-01 | The Board Of Regents, The University Of Texas System | Recombinant production of immunoglobulin-like domains in prokaryotic cells |
| JP2650574B2 (ja) | 1992-07-10 | 1997-09-03 | 株式会社島津製作所 | 磁場型質量分析装置 |
| US5639641A (en) | 1992-09-09 | 1997-06-17 | Immunogen Inc. | Resurfacing of rodent antibodies |
| DK0669836T3 (da) | 1992-11-13 | 1996-10-14 | Idec Pharma Corp | Terapeutisk anvendelse af kimære og radioaktivt mærkede antistoffer og humant B-lymfocytbegrænset differentieringsantigen til behandling af B-cellelymfom |
| US5635483A (en) | 1992-12-03 | 1997-06-03 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Tumor inhibiting tetrapeptide bearing modified phenethyl amides |
| US5780588A (en) | 1993-01-26 | 1998-07-14 | Arizona Board Of Regents | Elucidation and synthesis of selected pentapeptides |
| EP1333035A3 (en) | 1993-03-19 | 2004-07-07 | The Johns Hopkins University School Of Medicine | Growth differentiation factor-8 |
| US7393682B1 (en) | 1993-03-19 | 2008-07-01 | The Johns Hopkins University School Of Medicine | Polynucleotides encoding promyostatin polypeptides |
| ATE322547T1 (de) | 1993-06-10 | 2006-04-15 | Genetic Therapy Inc | Adenovirale vektoren für die behandlung der hämophilie |
| EP0714409A1 (en) | 1993-06-16 | 1996-06-05 | Celltech Therapeutics Limited | Antibodies |
| IL107742A0 (en) | 1993-11-24 | 1994-02-27 | Yeda Res & Dev | Chemically-modified binding proteins |
| JPH07177572A (ja) | 1993-12-20 | 1995-07-14 | Matsushita Electric Ind Co Ltd | コードレス電話の留守番方法 |
| US6074642A (en) | 1994-05-02 | 2000-06-13 | Alexion Pharmaceuticals, Inc. | Use of antibodies specific to human complement component C5 for the treatment of glomerulonephritis |
| US5773001A (en) | 1994-06-03 | 1998-06-30 | American Cyanamid Company | Conjugates of methyltrithio antitumor agents and intermediates for their synthesis |
| TW416960B (en) * | 1994-07-13 | 2001-01-01 | Chugai Pharmaceutical Co Ltd | Reshaped human antibody to human interleukin-8 |
| DE69535243T2 (de) | 1994-07-13 | 2007-05-10 | Chugai Seiyaku K.K. | Gegen menschliches interleukin-8 gerichteter, rekonstituierter menschlicher antikörper |
| US6048972A (en) | 1994-07-13 | 2000-04-11 | Chugai Pharmaceutical Co., Ltd. | Recombinant materials for producing humanized anti-IL-8 antibodies |
| CN1156460A (zh) | 1994-07-13 | 1997-08-06 | 中外制药株式会社 | 抗人白细胞介素-8的重构人抗体 |
| US6309636B1 (en) | 1995-09-14 | 2001-10-30 | Cancer Research Institute Of Contra Costa | Recombinant peptides derived from the Mc3 anti-BA46 antibody, methods of use thereof, and methods of humanizing antibody peptides |
| PT783893E (pt) | 1994-10-07 | 2012-05-24 | Chugai Pharmaceutical Co Ltd | Inibição do crescimento anormal das células sinoviais usando antagonistas de il-6 como componente ativo |
| CA2203182C (en) | 1994-10-21 | 2009-11-24 | Asao Katsume | Remedy for diseases caused by il-6 production |
| US5789199A (en) | 1994-11-03 | 1998-08-04 | Genentech, Inc. | Process for bacterial production of polypeptides |
| US6485943B2 (en) | 1995-01-17 | 2002-11-26 | The University Of Chicago | Method for altering antibody light chain interactions |
| US5840523A (en) | 1995-03-01 | 1998-11-24 | Genetech, Inc. | Methods and compositions for secretion of heterologous polypeptides |
| US5731168A (en) | 1995-03-01 | 1998-03-24 | Genentech, Inc. | Method for making heteromultimeric polypeptides |
| US5869046A (en) | 1995-04-14 | 1999-02-09 | Genentech, Inc. | Altered polypeptides with increased half-life |
| US5830478A (en) | 1995-06-07 | 1998-11-03 | Boston Biomedical Research Institute | Method for delivering functional domains of diphtheria toxin to a cellular target |
| US5712374A (en) | 1995-06-07 | 1998-01-27 | American Cyanamid Company | Method for the preparation of substantiallly monomeric calicheamicin derivative/carrier conjugates |
| US5714586A (en) | 1995-06-07 | 1998-02-03 | American Cyanamid Company | Methods for the preparation of monomeric calicheamicin derivative/carrier conjugates |
| US6267958B1 (en) | 1995-07-27 | 2001-07-31 | Genentech, Inc. | Protein formulation |
| US5783186A (en) | 1995-12-05 | 1998-07-21 | Amgen Inc. | Antibody-induced apoptosis |
| GB9603256D0 (en) | 1996-02-16 | 1996-04-17 | Wellcome Found | Antibodies |
| AU728657B2 (en) | 1996-03-18 | 2001-01-18 | Board Of Regents, The University Of Texas System | Immunoglobulin-like domains with increased half-lives |
| IL127872A0 (en) | 1996-07-19 | 1999-10-28 | Amgen Inc | Analogs of cationic proteins |
| US7247302B1 (en) | 1996-08-02 | 2007-07-24 | Bristol-Myers Squibb Company | Method for inhibiting immunoglobulin-induced toxicity resulting from the use of immunoglobulins in therapy and in vivo diagnosis |
| EP0918872B1 (en) | 1996-08-02 | 2008-02-20 | Bristol-Myers Squibb Company | A method for inhibiting immunoglobulin-induced toxicity resulting from the use of immunoglobulins in therapy and in vivo diagnosis |
| US5990286A (en) | 1996-12-18 | 1999-11-23 | Techniclone, Inc. | Antibodies with reduced net positive charge |
| US6025158A (en) | 1997-02-21 | 2000-02-15 | Genentech, Inc. | Nucleic acids encoding humanized anti-IL-8 monoclonal antibodies |
| US6277375B1 (en) | 1997-03-03 | 2001-08-21 | Board Of Regents, The University Of Texas System | Immunoglobulin-like domains with increased half-lives |
| US20070059302A1 (en) | 1997-04-07 | 2007-03-15 | Genentech, Inc. | Anti-vegf antibodies |
| US6884879B1 (en) | 1997-04-07 | 2005-04-26 | Genentech, Inc. | Anti-VEGF antibodies |
| ES2183351T3 (es) | 1997-04-17 | 2003-03-16 | Amgen Inc | Composicones que comprenden conjugados de proteina ob humana activa estable con una cadena fc de inmunoglobulinas y metodos. |
| US6171586B1 (en) | 1997-06-13 | 2001-01-09 | Genentech, Inc. | Antibody formulation |
| CN1068524C (zh) | 1997-06-23 | 2001-07-18 | 叶庆炜 | 一种治疗顽症牛皮癣的药物 |
| JP2002506353A (ja) | 1997-06-24 | 2002-02-26 | ジェネンテック・インコーポレーテッド | ガラクトシル化糖タンパク質の方法及び組成物 |
| US6040498A (en) | 1998-08-11 | 2000-03-21 | North Caroline State University | Genetically engineered duckweed |
| US7052873B2 (en) | 1997-10-03 | 2006-05-30 | Chugai Seiyaku Kabushiki Kaisha | Natural human antibody |
| ATE419009T1 (de) | 1997-10-31 | 2009-01-15 | Genentech Inc | Methoden und zusammensetzungen bestehend aus glykoprotein-glykoformen |
| US6610833B1 (en) | 1997-11-24 | 2003-08-26 | The Institute For Human Genetics And Biochemistry | Monoclonal human natural antibodies |
| PT1034298E (pt) | 1997-12-05 | 2012-02-03 | Scripps Research Inst | Humanização de anticorpo murino |
| US6458355B1 (en) | 1998-01-22 | 2002-10-01 | Genentech, Inc. | Methods of treating inflammatory disease with anti-IL-8 antibody fragment-polymer conjugates |
| DE69937291T2 (de) | 1998-04-02 | 2008-07-10 | Genentech, Inc., South San Francisco | Antikörpervarianten und fragmente davon |
| US6194551B1 (en) | 1998-04-02 | 2001-02-27 | Genentech, Inc. | Polypeptide variants |
| TR200101541T2 (tr) | 1998-04-03 | 2002-06-21 | Chugai Seiyaku Kabushiki Kaisha | İnsan doku faktörüne (TF) karşı insanlaştırılmış antikor. |
| DE69942021D1 (de) | 1998-04-20 | 2010-04-01 | Glycart Biotechnology Ag | Glykosylierungs-engineering von antikörpern zur verbesserung der antikörperabhängigen zellvermittelten zytotoxizität |
| GB9809951D0 (en) | 1998-05-08 | 1998-07-08 | Univ Cambridge Tech | Binding molecules |
| AU770555B2 (en) | 1998-08-17 | 2004-02-26 | Abgenix, Inc. | Generation of modified molecules with increased serum half-lives |
| US6475718B2 (en) | 1998-09-08 | 2002-11-05 | Schering Aktiengesellschaft | Methods and compositions for modulating the interaction between the APJ receptor and the HIV virus |
| EP1109545A4 (en) | 1998-09-11 | 2004-11-10 | Ilexus Pty Ltd | MODULATORS OF THE Fc RECEPTOR AND USE OF SUCH MODULATORS |
| RU2236222C2 (ru) | 1998-09-11 | 2004-09-20 | Айлексус Пти Лимитед | Модуляторы fc-рецептора и их применение |
| JP2002531466A (ja) | 1998-12-01 | 2002-09-24 | プロテイン デザイン ラブス, インコーポレイテッド | γ−インターフェロンに対するヒト化抗体 |
| US6737056B1 (en) | 1999-01-15 | 2004-05-18 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US7183387B1 (en) | 1999-01-15 | 2007-02-27 | Genentech, Inc. | Polypeptide variants with altered effector function |
| EP2386574A3 (en) | 1999-01-15 | 2012-06-27 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US6972125B2 (en) | 1999-02-12 | 2005-12-06 | Genetics Institute, Llc | Humanized immunoglobulin reactive with B7-2 and methods of treatment therewith |
| AU2006225302B2 (en) | 1999-03-25 | 2010-08-12 | AbbVie Deutschland GmbH & Co. KG | Human antibodies that bind human IL-12 and methods for producing |
| DK2270147T4 (da) | 1999-04-09 | 2020-08-31 | Kyowa Kirin Co Ltd | Fremgangsmåde til at kontrollere aktiviteten af immunologisk funktionelt molekyle |
| SK782002A3 (en) | 1999-07-21 | 2003-08-05 | Lexigen Pharm Corp | FC fusion proteins for enhancing the immunogenicity of protein and peptide antigens |
| PT1222292E (pt) | 1999-10-04 | 2005-11-30 | Medicago Inc | Metodo para regulacao da transcricao de genes exogenos na presenca de azoto |
| US7125978B1 (en) | 1999-10-04 | 2006-10-24 | Medicago Inc. | Promoter for regulating expression of foreign genes |
| AU7950400A (en) | 1999-10-19 | 2001-04-30 | Kyowa Hakko Kogyo Co. Ltd. | Process for producing polypeptide |
| SE9903895D0 (sv) | 1999-10-28 | 1999-10-28 | Active Biotech Ab | Novel compounds |
| AU784983B2 (en) | 1999-12-15 | 2006-08-17 | Genentech Inc. | Shotgun scanning, a combinatorial method for mapping functional protein epitopes |
| CA2395660A1 (en) | 1999-12-29 | 2001-07-12 | Immunogen, Inc. | Cytotoxic agents comprising modified doxorubicins and daunorubicins and their therapeutic use |
| RS50743B (sr) | 2000-03-22 | 2010-08-31 | OCTAPHARMA BIOPHARMACEUTICALS GmbH. | Proizvodnja rekombinantnih faktora koagulacije krvi u humanim ćelijama |
| JP2003531588A (ja) | 2000-04-11 | 2003-10-28 | ジェネンテック・インコーポレーテッド | 多価抗体とその用途 |
| FR2807767B1 (fr) | 2000-04-12 | 2005-01-14 | Lab Francais Du Fractionnement | Anticorps monoclonaux anti-d |
| EP1278512A2 (en) | 2000-05-03 | 2003-01-29 | MBT Munich Biotechnology AG | Cationic diagnostic, imaging and therapeutic agents associated with activated vascular sites |
| PT2275449T (pt) | 2000-06-16 | 2016-12-27 | Human Genome Sciences Inc | Anticorpos que se ligam imunoespecificamente a blys |
| AU2011244851A1 (en) | 2000-07-27 | 2011-11-24 | The John Hopkins University School Of Medicine | Promyostatin peptides and methods of using same |
| US6946292B2 (en) | 2000-10-06 | 2005-09-20 | Kyowa Hakko Kogyo Co., Ltd. | Cells producing antibody compositions with increased antibody dependent cytotoxic activity |
| ES2651952T3 (es) | 2000-10-06 | 2018-01-30 | Kyowa Hakko Kirin Co., Ltd. | Células que producen unas composiciones de anticuerpo |
| US7064191B2 (en) | 2000-10-06 | 2006-06-20 | Kyowa Hakko Kogyo Co., Ltd. | Process for purifying antibody |
| EP1325033B1 (en) | 2000-10-10 | 2009-11-25 | Genentech, Inc. | Inhibition of complement c5 activation for the treatment and prevention of delayed xenograft or acute vascular rejection |
| US6596541B2 (en) | 2000-10-31 | 2003-07-22 | Regeneron Pharmaceuticals, Inc. | Methods of modifying eukaryotic cells |
| JP3523245B1 (ja) | 2000-11-30 | 2004-04-26 | メダレックス,インコーポレーテッド | ヒト抗体作製用トランスジェニック染色体導入齧歯動物 |
| EP2357187A1 (en) | 2000-12-12 | 2011-08-17 | MedImmune, LLC | Molecules with extended half-lives, compositions and uses thereof |
| US20040001839A1 (en) | 2000-12-29 | 2004-01-01 | Avigdor Levanon | Multimers - isolated molecules comprising epitopes containing sulfated moieties, antibodies to such epitopes, and uses thereof |
| US20040001822A1 (en) | 2000-12-29 | 2004-01-01 | Avigdor Levanon | Y1-isolated molecules comprising epitopes containing sulfated moieties, antibodies to such epitopes, and uses thereof |
| US20040002450A1 (en) | 2000-12-29 | 2004-01-01 | Janette Lazarovits | Y17 - isolated molecules comprising epitopes containing sulfated moieties, antibodies to such epitopes, and uses thereof |
| AU2002258778C1 (en) | 2001-04-13 | 2008-12-04 | Biogen Ma Inc. | Antibodies to VLA-1 |
| US7667004B2 (en) | 2001-04-17 | 2010-02-23 | Abmaxis, Inc. | Humanized antibodies against vascular endothelial growth factor |
| DE60237929D1 (de) | 2001-06-22 | 2010-11-18 | Chugai Pharmaceutical Co Ltd | Zellproliferationshemmer, die Anti-Glypican 3 Antikörper enthalten |
| US20040161741A1 (en) | 2001-06-30 | 2004-08-19 | Elazar Rabani | Novel compositions and processes for analyte detection, quantification and amplification |
| EP1423510A4 (en) | 2001-08-03 | 2005-06-01 | Glycart Biotechnology Ag | ANTIBODY GLYCOSYLATION VARIANTS WITH INCREASED CELL CYTOTOXICITY DEPENDENT OF ANTIBODIES |
| WO2003015819A1 (en) | 2001-08-17 | 2003-02-27 | Tanox, Inc. | Complement pathway inhibitors binding to c5 and c5a without preventing formation of c5b |
| US20030049203A1 (en) | 2001-08-31 | 2003-03-13 | Elmaleh David R. | Targeted nucleic acid constructs and uses related thereto |
| US7320789B2 (en) | 2001-09-26 | 2008-01-22 | Wyeth | Antibody inhibitors of GDF-8 and uses thereof |
| KR100988949B1 (ko) | 2001-10-25 | 2010-10-20 | 제넨테크, 인크. | 당단백질 조성물 |
| ES2624547T3 (es) | 2001-11-14 | 2017-07-14 | Janssen Biotech, Inc. | Anticuerpos anti il 6, composiciones, métodos y usos |
| JP2005512044A (ja) | 2001-12-03 | 2005-04-28 | アブジェニックス・インコーポレーテッド | 結合特性に基づく抗体分類 |
| US20040093621A1 (en) | 2001-12-25 | 2004-05-13 | Kyowa Hakko Kogyo Co., Ltd | Antibody composition which specifically binds to CD20 |
| MXPA04007583A (es) | 2002-02-11 | 2005-04-25 | Genentech Inc | Variantes de anticuerpo con tasas mas rapidas de asociacion a antigeno. |
| AR038568A1 (es) | 2002-02-20 | 2005-01-19 | Hoffmann La Roche | Anticuerpos anti-a beta y su uso |
| US7317091B2 (en) | 2002-03-01 | 2008-01-08 | Xencor, Inc. | Optimized Fc variants |
| US8188231B2 (en) | 2002-09-27 | 2012-05-29 | Xencor, Inc. | Optimized FC variants |
| US20040132101A1 (en) | 2002-09-27 | 2004-07-08 | Xencor | Optimized Fc variants and methods for their generation |
| US20080199471A1 (en) | 2002-03-01 | 2008-08-21 | Bernett Matthew J | Optimized cd40 antibodies and methods of using the same |
| WO2003074679A2 (en) | 2002-03-01 | 2003-09-12 | Xencor | Antibody optimization |
| US7662925B2 (en) | 2002-03-01 | 2010-02-16 | Xencor, Inc. | Optimized Fc variants and methods for their generation |
| EP1502603A4 (en) | 2002-04-09 | 2006-12-13 | Kyowa Hakko Kogyo Kk | AN ANTIBODY COMPOSITION CONTAINING MEDICAMENT FOR PATIENTS WITH Fc gamma RIIIa POLYMORPHISM |
| EP1500698B1 (en) | 2002-04-09 | 2011-03-30 | Kyowa Hakko Kirin Co., Ltd. | Cell with depression or deletion of the activity of protein participating in gdp-fucose transport |
| EP1500400A4 (en) | 2002-04-09 | 2006-10-11 | Kyowa Hakko Kogyo Kk | MEDICAMENT WITH ANTIBODY COMPOSITION |
| EP1498491A4 (en) | 2002-04-09 | 2006-12-13 | Kyowa Hakko Kogyo Kk | METHOD FOR INCREASING THE ACTIVITY OF AN ANTIBODY COMPOSITION FOR BINDING TO THE FC GAMMA RECEPTOR IIIA |
| AU2003236015A1 (en) | 2002-04-09 | 2003-10-20 | Kyowa Hakko Kirin Co., Ltd. | Process for producing antibody composition |
| WO2003085107A1 (fr) | 2002-04-09 | 2003-10-16 | Kyowa Hakko Kogyo Co., Ltd. | Cellules à génome modifié |
| EP1509770B1 (en) | 2002-05-31 | 2014-07-09 | GE Healthcare Bio-Sciences AB | Method of coupling binding agents to a substrate surface |
| CA2488441C (en) | 2002-06-03 | 2015-01-27 | Genentech, Inc. | Synthetic antibody phage libraries |
| WO2003105757A2 (en) | 2002-06-12 | 2003-12-24 | Genencor International, Inc. | Methods and compositions for milieu-dependent binding of a targeted agent to a target |
| CA2489022C (en) | 2002-06-12 | 2012-10-16 | Genencor International, Inc. | Methods for improving a binding characteristic of a molecule |
| ITMI20021527A1 (it) | 2002-07-11 | 2004-01-12 | Consiglio Nazionale Ricerche | Anticorpi anti componente c5 del complemento e loro uso |
| US20050260213A1 (en) | 2004-04-16 | 2005-11-24 | Scott Koenig | Fcgamma-RIIB-specific antibodies and methods of use thereof |
| US8193318B2 (en) | 2002-08-14 | 2012-06-05 | Macrogenics, Inc. | FcγRIIB specific antibodies and methods of use thereof |
| JP2005535341A (ja) | 2002-08-15 | 2005-11-24 | エピトミスク インコーポレーティッド | ヒト化ウサギ抗体 |
| US20040138118A1 (en) | 2002-09-16 | 2004-07-15 | Neil Wolfman | Metalloprotease activation of myostatin, and methods of modulating myostatin activity |
| DK1553975T3 (da) | 2002-09-27 | 2012-05-07 | Xencor Inc | Optimerede Fc-varianter og fremgangsmåder til generering heraf. |
| RU2325186C2 (ru) | 2002-09-27 | 2008-05-27 | Ксенкор, Инк. | АНТИТЕЛО, СОДЕРЖАЩЕЕ Fc-ВАРИАНТНУЮ ЧАСТЬ (ВАРИАНТЫ), ФАРМАЦЕВТИЧЕСКАЯ КОМПОЗИЦИЯ, СОДЕРЖАЩАЯ АНТИТЕЛО, И СПОСОБ ЛЕЧЕНИЯ МЛЕКОПИТАЮЩЕГО |
| PT1562972E (pt) | 2002-10-15 | 2010-11-10 | Facet Biotech Corp | Alteração de afinidades de ligação ao fcrn ou semi-vidas séricas de anticorpos por mutagénese |
| US7361740B2 (en) | 2002-10-15 | 2008-04-22 | Pdl Biopharma, Inc. | Alteration of FcRn binding affinities or serum half-lives of antibodies by mutagenesis |
| US7217797B2 (en) | 2002-10-15 | 2007-05-15 | Pdl Biopharma, Inc. | Alteration of FcRn binding affinities or serum half-lives of antibodies by mutagenesis |
| US7261893B2 (en) | 2002-10-22 | 2007-08-28 | Wyeth | Neutralizing antibodies against GDF-8 and uses therefor |
| PT1572744E (pt) | 2002-12-16 | 2010-09-07 | Genentech Inc | Variantes de imunoglobulina e utilizações destas |
| JP4739763B2 (ja) | 2002-12-16 | 2011-08-03 | ゲンマブ エー/エス | インターロイキン8(il−8)に対するヒトモノクローナル抗体 |
| US7960512B2 (en) | 2003-01-09 | 2011-06-14 | Macrogenics, Inc. | Identification and engineering of antibodies with variant Fc regions and methods of using same |
| AU2004205631A1 (en) | 2003-01-16 | 2004-08-05 | Genentech, Inc. | Synthetic antibody phage libraries |
| WO2004068931A2 (en) | 2003-02-07 | 2004-08-19 | Protein Design Labs Inc. | Amphiregulin antibodies and their use to treat cancer and psoriasis |
| EP1601697B1 (en) | 2003-02-28 | 2007-05-30 | Lonza Biologics plc | Antibody purification by Protein A and ion exchange chromatography |
| KR20060026004A (ko) | 2003-02-28 | 2006-03-22 | 안티제닉스 아이엔씨 | 당단백질 및 항원성 분자의 올리고머화를 조장하는 렉틴의용도 |
| US8388955B2 (en) | 2003-03-03 | 2013-03-05 | Xencor, Inc. | Fc variants |
| US20090010920A1 (en) | 2003-03-03 | 2009-01-08 | Xencor, Inc. | Fc Variants Having Decreased Affinity for FcyRIIb |
| US7871607B2 (en) | 2003-03-05 | 2011-01-18 | Halozyme, Inc. | Soluble glycosaminoglycanases and methods of preparing and using soluble glycosaminoglycanases |
| US20060104968A1 (en) | 2003-03-05 | 2006-05-18 | Halozyme, Inc. | Soluble glycosaminoglycanases and methods of preparing and using soluble glycosaminogly ycanases |
| GB2401040A (en) | 2003-04-28 | 2004-11-03 | Chugai Pharmaceutical Co Ltd | Method for treating interleukin-6 related diseases |
| US9051373B2 (en) | 2003-05-02 | 2015-06-09 | Xencor, Inc. | Optimized Fc variants |
| RU2337107C2 (ru) | 2003-05-02 | 2008-10-27 | Ксенкор, Инк. | ОПТИМИЗИРОВАННЫЕ Fc-ВАРИАНТЫ, ИМЕЮЩИЕ ИЗМЕНЕННОЕ СВЯЗЫВАНИЕ С FcγR, И СПОСОБЫ ИХ ПОЛУЧЕНИЯ |
| BRPI0410927A (pt) | 2003-06-02 | 2006-06-27 | Wyeth Corp | métodos terapêuticos e profiláticos para distúrbios neuromusculares |
| WO2005005462A2 (en) | 2003-06-05 | 2005-01-20 | Genentech, Inc. | Blys antagonists and uses thereof |
| AU2004246744B2 (en) * | 2003-06-17 | 2008-04-03 | Xuning Wang | Crushing and grinding mill and soybean milk maker with the mill and method therefor |
| WO2005023193A2 (en) | 2003-09-04 | 2005-03-17 | Interleukin Genetics, Inc. | Methods of treating endometriosis |
| EP1663306A2 (en) | 2003-09-05 | 2006-06-07 | Genentech, Inc. | Antibodies with altered effector functions |
| US8101720B2 (en) | 2004-10-21 | 2012-01-24 | Xencor, Inc. | Immunoglobulin insertions, deletions and substitutions |
| EP1688439A4 (en) | 2003-10-08 | 2007-12-19 | Kyowa Hakko Kogyo Kk | HYBRID PROTEIN COMPOSITION |
| EP1705251A4 (en) | 2003-10-09 | 2009-10-28 | Kyowa Hakko Kirin Co Ltd | PROCESS FOR PRODUCING ANTIBODY COMPOSITION BY RNA INHIBITION OF FUNCTION OF $ G (A) 1,6-FUCOSYLTRANSFERASE |
| AU2003271174A1 (en) | 2003-10-10 | 2005-04-27 | Chugai Seiyaku Kabushiki Kaisha | Double specific antibodies substituting for functional protein |
| EP1673395A1 (en) | 2003-10-15 | 2006-06-28 | PDL BioPharma, Inc. | Alteration of fc-fusion protein serum half-lives by mutagenesis of positions 250, 314 and/or 428 of the heavy chain constant region of ig |
| DK1689777T3 (da) | 2003-11-05 | 2007-06-11 | Ares Trading Sa | Fremgangsmåde til oprensning af il18-bindende protein |
| EP1692182B1 (en) | 2003-11-05 | 2010-04-07 | Roche Glycart AG | Cd20 antibodies with increased fc receptor binding affinity and effector function |
| PT2489364E (pt) | 2003-11-06 | 2015-04-16 | Seattle Genetics Inc | Compostos de monometilvalina conjugados com anticorpos |
| US20050100965A1 (en) | 2003-11-12 | 2005-05-12 | Tariq Ghayur | IL-18 binding proteins |
| CA2545603A1 (en) | 2003-11-12 | 2005-05-26 | Biogen Idec Ma Inc. | Neonatal fc receptor (fcrn)-binding polypeptide variants, dimeric fc binding proteins and methods related thereto |
| EP1701979A2 (en) | 2003-12-03 | 2006-09-20 | Xencor, Inc. | Optimized antibodies that target the epidermal growth factor receptor |
| WO2005053742A1 (ja) | 2003-12-04 | 2005-06-16 | Kyowa Hakko Kogyo Co., Ltd. | 抗体組成物を含有する医薬 |
| CA2548817A1 (en) | 2003-12-04 | 2005-06-23 | Xencor, Inc. | Methods of generating variant proteins with increased host string content and compositions thereof |
| LT2418220T (lt) | 2003-12-10 | 2017-10-10 | E. R. Squibb & Sons, L.L.C. | Interferono-alfa antikūnai ir jų panaudojimas |
| US20050249723A1 (en) | 2003-12-22 | 2005-11-10 | Xencor, Inc. | Fc polypeptides with novel Fc ligand binding sites |
| SG153874A1 (en) | 2003-12-31 | 2009-07-29 | Schering Plough Ltd | Neutralizing epitope-based growth enhancing vaccine |
| CN1956738B (zh) | 2004-01-09 | 2013-05-29 | 辉瑞大药厂 | MAdCAM抗体 |
| US20050169921A1 (en) | 2004-02-03 | 2005-08-04 | Leonard Bell | Method of treating hemolytic disease |
| US20070116710A1 (en) | 2004-02-03 | 2007-05-24 | Leonard Bell | Methods of treating hemolytic anemia |
| BRPI0506679A (pt) | 2004-02-11 | 2007-05-15 | Warner Lambert Co | métodos de tratar osteoartrite com antagonistas de il-6 |
| ATE557042T1 (de) | 2004-03-23 | 2012-05-15 | Lilly Co Eli | Anti-myostatin-antikörper |
| AU2005227326B2 (en) | 2004-03-24 | 2009-12-03 | Xencor, Inc. | Immunoglobulin variants outside the Fc region |
| US20050260711A1 (en) | 2004-03-30 | 2005-11-24 | Deepshikha Datta | Modulating pH-sensitive binding using non-natural amino acids |
| RU2386638C2 (ru) | 2004-03-31 | 2010-04-20 | Дженентек, Инк. | Гуманизированные анти-тфр-бета-антитела |
| US7785903B2 (en) | 2004-04-09 | 2010-08-31 | Genentech, Inc. | Variable domain library and uses |
| WO2005123780A2 (en) | 2004-04-09 | 2005-12-29 | Protein Design Labs, Inc. | Alteration of fcrn binding affinities or serum half-lives of antibodies by mutagenesis |
| ES2403055T3 (es) | 2004-04-13 | 2013-05-13 | F. Hoffmann-La Roche Ag | Anticuerpos anti-P-selectina |
| WO2005112564A2 (en) | 2004-04-15 | 2005-12-01 | The Government Of The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Germline and sequence variants of humanized antibodies and methods of making and using them |
| KR100620554B1 (ko) | 2004-06-05 | 2006-09-06 | 한국생명공학연구원 | Tag-72에 대한 인간화 항체 |
| AR049390A1 (es) | 2004-06-09 | 2006-07-26 | Wyeth Corp | Anticuerpos contra la interleuquina-13 humana y usos de los mismos |
| JP2008503217A (ja) | 2004-06-18 | 2008-02-07 | アンブレツクス・インコーポレイテツド | 新規抗原結合ポリペプチド及びそれらの使用 |
| AU2005259992A1 (en) | 2004-06-25 | 2006-01-12 | Medimmune, Llc | Increasing the production of recombinant antibodies in mammalian cells by site-directed mutagenesis |
| WO2006085967A2 (en) | 2004-07-09 | 2006-08-17 | Xencor, Inc. | OPTIMIZED ANTI-CD20 MONOCONAL ANTIBODIES HAVING Fc VARIANTS |
| EP3342782B1 (en) | 2004-07-15 | 2022-08-17 | Xencor, Inc. | Optimized fc variants |
| ES2426816T3 (es) | 2004-08-04 | 2013-10-25 | Mentrik Biotech, Llc | Regiones Fc variantes |
| AU2005277567A1 (en) | 2004-08-16 | 2006-03-02 | Medimmune, Llc | Integrin antagonists with enhanced antibody dependent cell-mediated cytotoxicity activity |
| CN101052654A (zh) | 2004-08-19 | 2007-10-10 | 健泰科生物技术公司 | 具有改变的效应子功能的多肽变体 |
| WO2006031825A2 (en) | 2004-09-13 | 2006-03-23 | Macrogenics, Inc. | Humanized antibodies against west nile virus and therapeutic and prophylactic uses thereof |
| WO2006030200A1 (en) | 2004-09-14 | 2006-03-23 | National Institute For Biological Standards And Control | Vaccine |
| TWI380996B (zh) | 2004-09-17 | 2013-01-01 | Hoffmann La Roche | 抗ox40l抗體 |
| US7563443B2 (en) | 2004-09-17 | 2009-07-21 | Domantis Limited | Monovalent anti-CD40L antibody polypeptides and compositions thereof |
| DK1791565T3 (en) | 2004-09-23 | 2016-08-01 | Genentech Inc | Cysteingensplejsede antibodies and conjugates |
| WO2006036834A2 (en) | 2004-09-24 | 2006-04-06 | Amgen Inc. | MODIFIED Fc MOLECULES |
| JO3000B1 (ar) | 2004-10-20 | 2016-09-05 | Genentech Inc | مركبات أجسام مضادة . |
| WO2006047350A2 (en) | 2004-10-21 | 2006-05-04 | Xencor, Inc. | IgG IMMUNOGLOBULIN VARIANTS WITH OPTIMIZED EFFECTOR FUNCTION |
| JP2008520552A (ja) | 2004-10-22 | 2008-06-19 | メディミューン,インコーポレーテッド | Hmgb1に対する高親和性の抗体およびその使用法 |
| AU2005302453A1 (en) | 2004-10-29 | 2006-05-11 | Medimmune, Llc | Methods of preventing and treating RSV infections and related conditions |
| US7462697B2 (en) | 2004-11-08 | 2008-12-09 | Epitomics, Inc. | Methods for antibody engineering |
| EP1810035A4 (en) | 2004-11-10 | 2010-03-17 | Macrogenics Inc | EFFECTOR FUNCTION OBTAINED BY CREATION BY BIOLOGICAL GENE OF FC ANTIBODY REGIONS |
| US20070135620A1 (en) | 2004-11-12 | 2007-06-14 | Xencor, Inc. | Fc variants with altered binding to FcRn |
| EP1817340B1 (en) | 2004-11-12 | 2012-05-16 | Xencor, Inc. | Fc variants with altered binding to fcrn |
| US8802820B2 (en) | 2004-11-12 | 2014-08-12 | Xencor, Inc. | Fc variants with altered binding to FcRn |
| US8329186B2 (en) | 2004-12-20 | 2012-12-11 | Isu Abxis Co., Ltd | Treatment of inflammation using BST2 inhibitor |
| US20090061485A1 (en) | 2004-12-22 | 2009-03-05 | Chugai Seiyaku Kabushiki Kaisha | Method of Producing an Antibody Using a Cell in Which the Function of Fucose Transporter Is Inhibited |
| WO2006066598A2 (en) | 2004-12-23 | 2006-06-29 | Novo Nordisk A/S | Antibody binding affinity ligands |
| AU2005321974B2 (en) | 2004-12-27 | 2011-11-17 | Progenics Pharmaceuticals (Nevada), Inc. | Orally deliverable and anti-toxin antibodies and methods for making and using them |
| EA018897B1 (ru) | 2005-01-05 | 2013-11-29 | Ф-Стар Биотехнологише Форшунгс- Унд Энтвиклунгсгез.М.Б.Х. | Молекулы иммуноглобулина, содержащие модифицированные участки структурных петель, обладающие свойством связывания, и способ их получения |
| AU2006204791A1 (en) | 2005-01-12 | 2006-07-20 | Xencor, Inc | Antibodies and Fc fusion proteins with altered immunogenicity |
| GB0502358D0 (en) | 2005-02-04 | 2005-03-16 | Novartis Ag | Organic compounds |
| NZ538097A (en) | 2005-02-07 | 2006-07-28 | Ovita Ltd | Method and compositions for improving wound healing |
| AU2006227377B2 (en) | 2005-03-18 | 2013-01-31 | Medimmune, Llc | Framework-shuffling of antibodies |
| EP1871808A2 (en) | 2005-03-31 | 2008-01-02 | Xencor, Inc. | Fc VARIANTS WITH OPTIMIZED PROPERTIES |
| WO2006106905A1 (ja) | 2005-03-31 | 2006-10-12 | Chugai Seiyaku Kabushiki Kaisha | 会合制御によるポリペプチド製造方法 |
| ES2494921T3 (es) | 2005-04-08 | 2014-09-16 | Chugai Seiyaku Kabushiki Kaisha | Anticuerpo que sustituye la función del factor VIII de coagulación sanguínea |
| WO2006113643A2 (en) | 2005-04-20 | 2006-10-26 | Amgen Fremont Inc. | High affinity fully human monoclonal antibodies to interleukin-8 and epitopes for such antibodies |
| DOP2006000093A (es) | 2005-04-25 | 2007-01-31 | Pfizer | Anticuerpos contra miostatina |
| WO2006121852A2 (en) | 2005-05-05 | 2006-11-16 | Duke University | Anti-cd19 antibody therapy for autoimmune disease |
| WO2006130834A2 (en) | 2005-05-31 | 2006-12-07 | Board Of Regents, The University Of Texas System | IGGl ANTIBODIES WITH MUTATED FC PORTION FOR INCREASED BINDING TO FCRN RECEPTOR AND USES THEREOF |
| CA2652434A1 (en) | 2005-07-08 | 2007-01-18 | Xencor, Inc. | Optimized proteins that target ep-cam |
| US8008453B2 (en) | 2005-08-12 | 2011-08-30 | Amgen Inc. | Modified Fc molecules |
| WO2007022520A2 (en) | 2005-08-19 | 2007-02-22 | Cerus Corporation | Antibody-mediated enhancement of immune response |
| US7888486B2 (en) | 2005-08-19 | 2011-02-15 | Wyeth Llc | Antagonist antibodies against GDF-8 |
| CA2624189A1 (en) | 2005-10-03 | 2007-04-12 | Xencor, Inc. | Fc variants with optimized fc receptor binding properties |
| WO2007044616A2 (en) | 2005-10-06 | 2007-04-19 | Xencor, Inc. | Optimized anti-cd30 antibodies |
| CA2624976A1 (en) | 2005-10-06 | 2007-04-19 | Eli Lilly And Company | Anti-myostatin antibodies |
| UA92504C2 (en) | 2005-10-12 | 2010-11-10 | Эли Лилли Энд Компани | Anti-myostatin monoclonal antibody |
| CN101331223A (zh) | 2005-10-14 | 2008-12-24 | 米迪缪尼有限公司 | 细胞展示抗体文库 |
| EP1957531B1 (en) | 2005-11-07 | 2016-04-13 | Genentech, Inc. | Binding polypeptides with diversified and consensus vh/vl hypervariable sequences |
| AR057579A1 (es) | 2005-11-23 | 2007-12-05 | Merck & Co Inc | Compuestos espirociclicos como inhibidores de histona de acetilasa (hdac) |
| WO2007060411A1 (en) | 2005-11-24 | 2007-05-31 | Ucb Pharma S.A. | Anti-tnf alpha antibodies which selectively inhibit tnf alpha signalling through the p55r |
| AU2006330807A1 (en) | 2005-11-28 | 2007-07-05 | Medimmune, Llc | Antagonists of HMBG1 and/or rage and methods of use thereof |
| WO2007064919A2 (en) | 2005-12-02 | 2007-06-07 | Genentech, Inc. | Binding polypeptides with restricted diversity sequences |
| NZ568384A (en) | 2005-12-12 | 2011-09-30 | Ac Immune Sa | Therapeutic vaccine comprising Abeta peptide |
| SI3219328T1 (sl) | 2005-12-29 | 2020-10-30 | Janssen Biotech, Inc. | Človeška protitelesa proti-IL-23, sestavki, postopki in uporabe |
| CA2638811A1 (en) | 2006-02-03 | 2007-08-16 | Medimmune, Llc | Protein formulations |
| US20070190056A1 (en) | 2006-02-07 | 2007-08-16 | Ravi Kambadur | Muscle regeneration compositions and uses therefor |
| JP4179517B2 (ja) | 2006-02-21 | 2008-11-12 | プロテノバ株式会社 | イムノグロブリン親和性リガンド |
| WO2007097361A1 (ja) | 2006-02-21 | 2007-08-30 | Protenova Co., Ltd. | イムノグロブリン親和性リガンド |
| SG171578A1 (en) | 2006-03-02 | 2011-06-29 | Alexion Pharma Inc | Prolongation of survival of an allograft by inhibiting complement activity |
| KR101513308B1 (ko) | 2006-03-08 | 2015-04-28 | 아케믹스 엘엘씨 | 안질환의 치료에 유용한 보체 결합 앱타머 및 항-c5 제제 |
| EP4316465A3 (en) | 2006-03-15 | 2024-04-24 | Alexion Pharmaceuticals, Inc. | Treatment of paroxysmal nocturnal hemoglobinuria patients by an inhibitor of complement |
| CN103073639A (zh) | 2006-03-17 | 2013-05-01 | 比奥根艾迪克Ma公司 | 稳定的多肽组合物 |
| KR20080113268A (ko) | 2006-03-28 | 2008-12-29 | 바이오겐 아이덱 엠에이 인코포레이티드 | 항 igf-1r 항체 및 그의 용도 |
| EP3345616A1 (en) | 2006-03-31 | 2018-07-11 | Chugai Seiyaku Kabushiki Kaisha | Antibody modification method for purifying bispecific antibody |
| CN104761637B (zh) | 2006-03-31 | 2021-10-15 | 中外制药株式会社 | 调控抗体血液动力学的方法 |
| RU2466740C2 (ru) | 2006-04-05 | 2012-11-20 | Эбботт Байотекнолоджи Лтд. | Очистка антитела |
| EP2016101A2 (en) | 2006-05-09 | 2009-01-21 | Genentech, Inc. | Binding polypeptides with optimized scaffolds |
| EP2021029B1 (en) | 2006-05-26 | 2014-06-11 | MacroGenics, Inc. | Humanized fc gamma riib-specific antibodies and methods of use thereof |
| EP2371393B1 (en) | 2006-06-08 | 2015-08-05 | Chugai Seiyaku Kabushiki Kaisha | Preventive or remedy for inflammatory disease |
| EP2032159B1 (en) | 2006-06-26 | 2015-01-07 | MacroGenics, Inc. | Combination of fcgammariib antibodies and cd20-specific antibodies and methods of use thereof |
| AR062223A1 (es) | 2006-08-09 | 2008-10-22 | Glycart Biotechnology Ag | Moleculas de adhesion al antigeno que se adhieren a egfr, vectores que los codifican, y sus usos de estas |
| PL2383297T3 (pl) | 2006-08-14 | 2013-06-28 | Xencor Inc | Zoptymalizowane przeciwciała ukierunkowane na CD19 |
| DK2059533T3 (da) | 2006-08-30 | 2013-02-25 | Genentech Inc | Multispecifikke antistoffer |
| JP5058261B2 (ja) | 2006-09-05 | 2012-10-24 | イーライ リリー アンド カンパニー | 抗ミオスタチン抗体 |
| DK2066349T3 (da) | 2006-09-08 | 2012-07-09 | Medimmune Llc | Humaniseret anti-cd19-antistoffer og anvendelse heraf i behandling af tumorer, transplantation og autoimmune sygdomme |
| US8394374B2 (en) | 2006-09-18 | 2013-03-12 | Xencor, Inc. | Optimized antibodies that target HM1.24 |
| EP3424950A1 (en) | 2006-10-06 | 2019-01-09 | The Government of the United States of America as represented by the Secretary of the Department of Health and Human Services | Prevention of tissue ischemia, related methods and compositions |
| US20100034194A1 (en) | 2006-10-11 | 2010-02-11 | Siemens Communications Inc. | Eliminating unreachable subscribers in voice-over-ip networks |
| WO2008121160A2 (en) | 2006-11-21 | 2008-10-09 | Xencor, Inc. | Optimized antibodies that target cd5 |
| CN100455598C (zh) | 2006-11-29 | 2009-01-28 | 中国抗体制药有限公司 | 功能人源化抗人cd20抗体及其应用 |
| US8652466B2 (en) | 2006-12-08 | 2014-02-18 | Macrogenics, Inc. | Methods for the treatment of disease using immunoglobulins having Fc regions with altered affinities for FcγRactivating and FcγRinhibiting |
| US20080226635A1 (en) | 2006-12-22 | 2008-09-18 | Hans Koll | Antibodies against insulin-like growth factor I receptor and uses thereof |
| WO2008091798A2 (en) | 2007-01-22 | 2008-07-31 | Xencor, Inc. | Optimized ca9 antibodies and methods of using the same |
| EP2125893A2 (en) | 2007-01-23 | 2009-12-02 | Xencor, Inc. | Optimized cd40 antibodies and methods of using the same |
| WO2008092117A2 (en) | 2007-01-25 | 2008-07-31 | Xencor, Inc. | Immunoglobulins with modifications in the fcr binding region |
| WO2008098115A2 (en) | 2007-02-07 | 2008-08-14 | Xencor, Inc. | Optimized igf-1r antibodies and methods of using the same |
| WO2008114011A2 (en) | 2007-03-19 | 2008-09-25 | Medimmune Limited | Fc polypeptide variants obtained by ribosome display methodology |
| EA200901211A1 (ru) | 2007-03-22 | 2010-04-30 | Новартис Аг | Антигены белка с5 и их применение |
| US8337854B2 (en) | 2007-04-04 | 2012-12-25 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Monoclonal antibodies against dengue and other viruses with deletion in Fc region |
| CL2008001071A1 (es) | 2007-04-17 | 2009-05-22 | Smithkline Beecham Corp | Metodo para obtener anticuerpo penta-especifico contra il-8/cxcl8, gro-alfa/cxcl1, gro-beta/cxcl2), gro-gama/cxcl3 y ena-78/cxcl5 humanas; anticuerpo penta-especifico; proceso de produccion del mismo; vector, hbridoma o celela que lo comprende; composicion farmceutica; uso para tratar copd, otras enfermedades. |
| CN100592373C (zh) | 2007-05-25 | 2010-02-24 | 群康科技(深圳)有限公司 | 液晶显示面板驱动装置及其驱动方法 |
| CA2693053C (en) | 2007-05-30 | 2021-01-05 | Xencor, Inc. | Methods and compositions for inhibiting cd32b expressing cells |
| WO2009000098A2 (en) | 2007-06-25 | 2008-12-31 | Esbatech Ag | Sequence based engineering and optimization of single chain antibodies |
| RU2540150C2 (ru) | 2007-06-25 | 2015-02-10 | Эсбатек, Эн Элкон Биомедикал Рисёрч Юнит Ллк | Способы модификации антител и модифицированные антитела с улучшенными функциональными свойствами |
| EP2179282B1 (en) | 2007-06-29 | 2016-06-01 | Quest Diagnostics Investments Incorporated | Analysis of amino acids in body fluid by liquid chromatography-mass spectrometry |
| JOP20080381B1 (ar) | 2007-08-23 | 2023-03-28 | Amgen Inc | بروتينات مرتبطة بمولدات مضادات تتفاعل مع بروبروتين كونفيرتاز سيتيليزين ككسين من النوع 9 (pcsk9) |
| EP2190480A4 (en) | 2007-08-28 | 2013-01-23 | Biogen Idec Inc | ANTI-IGR-1R ANTIBODIES AND ITS USES |
| JP2010538012A (ja) | 2007-08-28 | 2010-12-09 | バイオジェン アイデック マサチューセッツ インコーポレイテッド | Igf−1rの複数のエピトープに結合する組成物 |
| AU2008304756B8 (en) | 2007-09-26 | 2015-02-12 | Chugai Seiyaku Kabushiki Kaisha | Anti-IL-6 receptor antibody |
| TWI464262B (zh) | 2007-09-26 | 2014-12-11 | 中外製藥股份有限公司 | 抗體固定區的變異 |
| EP3127921A1 (en) | 2007-09-26 | 2017-02-08 | Chugai Seiyaku Kabushiki Kaisha | Method of modifying isoelectric point of antibody via amino acid substition in cdr |
| WO2009041062A1 (ja) | 2007-09-28 | 2009-04-02 | Chugai Seiyaku Kabushiki Kaisha | 血漿中動態が改善されたグリピカン3抗体 |
| KR100888133B1 (ko) | 2007-10-02 | 2009-03-13 | 에스케이에너지 주식회사 | 4종의 금속성분으로 구성된 다성분계 비스무스몰리브데이트 촉매 제조방법 및 상기촉매를 이용하여1,3-부타디엔을 제조하는 방법 |
| PE20091163A1 (es) | 2007-11-01 | 2009-08-09 | Wyeth Corp | Anticuerpos para gdf8 |
| US20120039871A1 (en) | 2007-11-08 | 2012-02-16 | Pikamab, Inc. | Methods and compositions for antibody therapy |
| EP2261254A3 (en) | 2007-12-21 | 2011-04-13 | Amgen, Inc | Anti-amyloid antibodies and uses thereof |
| LT2808343T (lt) | 2007-12-26 | 2019-09-10 | Xencor Inc. | Fc variantai su pakitusiu prisijungimu prie fcrn |
| CA2709847C (en) | 2008-01-07 | 2018-07-10 | Amgen Inc. | Method for making antibody fc-heterodimeric molecules using electrostatic steering effects |
| US8551485B2 (en) | 2008-01-23 | 2013-10-08 | Xencor, Inc. | Anti-CD40 antibodies and methods of inhibiting proliferation of CD40 expressing cells |
| US20110183861A1 (en) | 2008-01-29 | 2011-07-28 | Ablynn N.V. | Methods to stabilize proteins and polypeptides |
| AU2015227424A1 (en) * | 2008-04-11 | 2015-10-01 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule capable of binding to two or more antigen molecules repeatedly |
| TWI564021B (zh) | 2008-04-11 | 2017-01-01 | Chugai Pharmaceutical Co Ltd | Repeated binding of antigen to antigen binding molecules |
| SG196839A1 (en) | 2008-04-25 | 2014-02-13 | Dyax Corp | Antibodies against fcrn and use thereof |
| ES2487846T3 (es) | 2008-05-01 | 2014-08-25 | Amgen, Inc. | Anticuerpos anti-hepcindina y métodos de uso |
| CN102088995B (zh) | 2008-05-14 | 2016-01-20 | 维多利亚农业服务控股公司 | 血管生成素和血管生成素激动剂用于治疗疾病和障碍的用途 |
| WO2009155513A2 (en) | 2008-06-20 | 2009-12-23 | Novartis Ag | Immunoglobulins with reduced aggregation |
| CA2732782C (en) | 2008-08-05 | 2019-02-26 | Novartis Ag | Compositions and methods for antibodies targeting complement protein c5 |
| CN102292353B (zh) | 2008-09-17 | 2015-04-29 | Xencor公司 | 用于治疗ige介导的疾患的新组合物和方法 |
| TWI440469B (zh) | 2008-09-26 | 2014-06-11 | Chugai Pharmaceutical Co Ltd | Improved antibody molecules |
| JP5229888B2 (ja) | 2008-09-30 | 2013-07-03 | 独立行政法人産業技術総合研究所 | 弱酸性域での易解離性を向上したプロテインa変異型タンパク質及び抗体捕捉剤 |
| NZ592054A (en) | 2008-10-13 | 2013-06-28 | Inst Research In Biomedicine | Dengue virus neutralizing antibodies and uses thereof |
| JP5913980B2 (ja) | 2008-10-14 | 2016-05-11 | ジェネンテック, インコーポレイテッド | 免疫グロブリン変異体及びその用途 |
| CA3035432A1 (en) | 2008-11-10 | 2010-05-14 | Alexion Pharmaceuticals, Inc. | Methods and compositions for treating complement-associated disorders |
| WO2010058860A1 (ja) | 2008-11-18 | 2010-05-27 | 株式会社シノテスト | 試料中のc反応性蛋白質の測定方法及び測定試薬 |
| JO3672B1 (ar) | 2008-12-15 | 2020-08-27 | Regeneron Pharma | أجسام مضادة بشرية عالية التفاعل الكيماوي بالنسبة لإنزيم سبتيليسين كنفرتيز بروبروتين / كيكسين نوع 9 (pcsk9). |
| AR074777A1 (es) | 2008-12-19 | 2011-02-09 | Glaxo Group Ltd | Proteinas de union a antigeno |
| WO2010098863A1 (en) | 2009-02-26 | 2010-09-02 | Lpath, Inc. | Humanized platelet activating factor antibody design using anti-lipid antibody templates |
| US20120071634A1 (en) | 2009-03-19 | 2012-03-22 | Chugai Seiyaku Kabushiki Kaisha | Antibody Constant Region Variant |
| EP2233500A1 (en) | 2009-03-20 | 2010-09-29 | LFB Biotechnologies | Optimized Fc variants |
| MA33405B1 (fr) | 2009-05-15 | 2012-07-03 | Chugai Pharmaceutical Co Ltd | Anticorps anti-axl |
| US8609097B2 (en) | 2009-06-10 | 2013-12-17 | Hoffmann-La Roche Inc. | Use of an anti-Tau pS422 antibody for the treatment of brain diseases |
| US8945511B2 (en) | 2009-06-25 | 2015-02-03 | Paul Weinberger | Sensitive methods for detecting the presence of cancer associated with the over-expression of galectin-3 using biomarkers derived from galectin-3 |
| EP2448972A4 (en) | 2009-06-30 | 2012-11-28 | Res Dev Foundation | IMMUNOGLOBULIN FC POLYPEPTIDES |
| GB0914691D0 (en) | 2009-08-21 | 2009-09-30 | Lonza Biologics Plc | Immunoglobulin variants |
| RU2646139C1 (ru) | 2009-09-03 | 2018-03-01 | Мерк Шарп И Доум Корп. | Анти-gitr-антитела |
| WO2011051350A1 (en) | 2009-10-27 | 2011-05-05 | Ucb Pharma S.A. | Function modifying nav 1.7 antibodies |
| CN101875696B (zh) | 2009-11-11 | 2012-02-08 | 中国人民解放军军事医学科学院生物工程研究所 | 一种抗体及其制备方法与应用 |
| EP2327725A1 (en) | 2009-11-26 | 2011-06-01 | InflaRx GmbH | Anti-C5a binding moieties with high blocking activity |
| WO2011091078A2 (en) | 2010-01-19 | 2011-07-28 | Xencor, Inc. | Antibody fc variants with enhanced complement activity |
| US9624290B2 (en) | 2010-01-28 | 2017-04-18 | Ab Biosciences, Inc. | Lowered affinity antibodies and methods of making the same |
| MX2012010116A (es) | 2010-03-01 | 2013-02-26 | Alexion Pharma Inc | Metodos y composiciones para el tratamiento de la enfermedad de degos. |
| TW201206466A (en) | 2010-03-11 | 2012-02-16 | Rinat Neuroscience Corp | Antibodies with pH dependent antigen binding |
| CN102844332B (zh) | 2010-03-11 | 2015-08-19 | 瑞纳神经科学公司 | 呈pH依赖性抗原结合的抗体 |
| TWI667346B (zh) | 2010-03-30 | 2019-08-01 | 中外製藥股份有限公司 | 促進抗原消失之具有經修飾的FcRn親和力之抗體 |
| US20140206849A1 (en) | 2010-04-30 | 2014-07-24 | Alexion Pharmaceuticals, Inc. | Antibodies having reduced immunogenicity in a human |
| JO3340B1 (ar) | 2010-05-26 | 2019-03-13 | Regeneron Pharma | مضادات حيوية لـعامل تمايز النمو 8 البشري |
| TW201210612A (en) | 2010-06-03 | 2012-03-16 | Glaxo Group Ltd | Humanised antigen binding proteins |
| CN105585630B (zh) | 2010-07-29 | 2020-09-15 | Xencor公司 | 具有修改的等电点的抗体 |
| MX2013001845A (es) | 2010-08-16 | 2013-08-27 | Amgen Inc | Polipetidos que enlazan miostatina, composiciones y metodos. |
| CN103261222B (zh) | 2010-09-10 | 2017-07-28 | 医疗免疫有限公司 | 抗体衍生物 |
| WO2012044831A1 (en) | 2010-09-30 | 2012-04-05 | Board Of Trustees Of Northern Illinois University | Library-based methods and compositions for introducing molecular switch functionality into protein affinity reagents |
| TWI761912B (zh) | 2010-11-30 | 2022-04-21 | 日商中外製藥股份有限公司 | 具有鈣依存性的抗原結合能力之抗體 |
| MX348152B (es) | 2010-12-14 | 2017-06-02 | Nat Univ Singapore | Anticuerpo monoclonal humano con especificidad para el virus del dengue serotipo 1 proteina e y sus usos. |
| WO2012088247A2 (en) | 2010-12-22 | 2012-06-28 | Medimmune, Llc | Anti-c5/c5a/c5adesr antibodies and fragments |
| US20140080153A1 (en) | 2011-01-07 | 2014-03-20 | Chugai Seiyaku Kabushiki Kaisha | Method for improving physical properties of antibody |
| WO2012132067A1 (ja) | 2011-03-30 | 2012-10-04 | 中外製薬株式会社 | 抗原結合分子の血漿中滞留性と免疫原性を改変する方法 |
| MX352889B (es) | 2011-02-25 | 2017-12-13 | Chugai Pharmaceutical Co Ltd | Anticuerpo de fc especifico para fcyriib. |
| AU2012233313C1 (en) | 2011-03-30 | 2017-08-03 | Chugai Seiyaku Kabushiki Kaisha | Method for altering plasma retention and immunogenicity of antigen-binding molecule |
| ES2709654T3 (es) | 2011-04-29 | 2019-04-17 | Apexigen Inc | Anticuerpos anti-CD40 y métodos de uso |
| JP5891298B2 (ja) | 2011-05-04 | 2016-03-22 | オメロス コーポレーション | Masp−2依存性の補体活性化を阻害するための組成物 |
| CN103826659A (zh) | 2011-06-20 | 2014-05-28 | 圣路易斯大学 | 用于治疗的靶向神经肌肉接头 |
| KR102049122B1 (ko) | 2011-06-30 | 2019-11-26 | 추가이 세이야쿠 가부시키가이샤 | 헤테로이량화 폴리펩티드 |
| US9738707B2 (en) | 2011-07-15 | 2017-08-22 | Biogen Ma Inc. | Heterodimeric Fc regions, binding molecules comprising same, and methods relating thereto |
| RU2722829C9 (ru) | 2011-09-30 | 2020-09-22 | Чугаи Сейяку Кабусики Кайся | Антигенсвязывающая молекула, индуцирующая иммунный ответ на антиген-мишень |
| US10024867B2 (en) | 2011-09-30 | 2018-07-17 | Chugai Seiyaku Kabushiki Kaisha | Ion concentration-dependent binding molecule library |
| SG11201401101XA (en) | 2011-09-30 | 2014-08-28 | Chugai Pharmaceutical Co Ltd | Antigen-binding molecule for promoting loss of antigens |
| TW201817745A (zh) | 2011-09-30 | 2018-05-16 | 日商中外製藥股份有限公司 | 具有促進抗原清除之FcRn結合域的治療性抗原結合分子 |
| WO2013047748A1 (ja) | 2011-09-30 | 2013-04-04 | 中外製薬株式会社 | 複数の生理活性を有する抗原の消失を促進する抗原結合分子 |
| JP6271251B2 (ja) | 2011-10-05 | 2018-01-31 | 中外製薬株式会社 | 糖鎖受容体結合ドメインを含む抗原の血漿中からの消失を促進する抗原結合分子 |
| CN104080909A (zh) | 2011-11-30 | 2014-10-01 | 中外制药株式会社 | 包含进入细胞内以形成免疫复合体的搬运体(载体)的药物 |
| US20140349321A1 (en) | 2011-12-16 | 2014-11-27 | Agency For Science, Technology And Research | Binding molecules against dengue virus and uses thereof |
| JP6226752B2 (ja) | 2012-02-09 | 2017-11-08 | 中外製薬株式会社 | 抗体のFc領域改変体 |
| TW202015731A (zh) | 2012-02-24 | 2020-05-01 | 日商中外製藥股份有限公司 | 經FcγRIIB促進抗原消失之抗原結合分子 |
| MY172730A (en) | 2012-03-16 | 2019-12-11 | Regeneron Pharma | Histidine engineered light chain antibodies and genetically modified non-human animals for generating the same |
| US9801362B2 (en) | 2012-03-16 | 2017-10-31 | Regeneron Pharmaceuticals, Inc. | Non-human animals expressing pH-sensitive immunoglobulin sequences |
| CN104302170B (zh) | 2012-03-16 | 2016-09-28 | 瑞泽恩制药公司 | 生产具有ph依赖性结合特性的抗原结合蛋白的小鼠 |
| HUE037613T2 (hu) | 2012-03-29 | 2018-09-28 | Novimmune Sa | Anti-TLR4 antitestek és azok felhasználása |
| CN104428312A (zh) | 2012-04-02 | 2015-03-18 | 北卡罗来纳-查佩尔山大学 | 用于登革病毒表位的方法和组合物 |
| TWI619729B (zh) | 2012-04-02 | 2018-04-01 | 再生元醫藥公司 | 抗-hla-b*27抗體及其用途 |
| JP5988659B2 (ja) | 2012-04-09 | 2016-09-07 | シャープ株式会社 | 送風機 |
| WO2013166099A1 (en) | 2012-05-01 | 2013-11-07 | Glaxosmithkline Llc | Novel antibodies |
| US9255154B2 (en) | 2012-05-08 | 2016-02-09 | Alderbio Holdings, Llc | Anti-PCSK9 antibodies and use thereof |
| WO2013173348A1 (en) | 2012-05-14 | 2013-11-21 | The Usa, As Represented By The Secretary, Dept. Of Health & Human Services | Cross-reactive antibodies against dengue virus and uses thereof |
| EP3795215A1 (en) | 2012-05-30 | 2021-03-24 | Chugai Seiyaku Kabushiki Kaisha | Target tissue-specific antigen-binding molecule |
| EP3892638A1 (en) | 2012-05-30 | 2021-10-13 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule for eliminating aggregated antigens |
| WO2013187495A1 (ja) | 2012-06-14 | 2013-12-19 | 中外製薬株式会社 | 改変されたFc領域を含む抗原結合分子 |
| MY177331A (en) | 2012-06-15 | 2020-09-12 | Pfizer | Improved antagonist antibodies against gdf-8 and uses therefor |
| AU2013285355A1 (en) | 2012-07-06 | 2015-01-29 | Genmab B.V. | Dimeric protein with triple mutations |
| US9499607B2 (en) | 2012-08-07 | 2016-11-22 | Massachusetts Institute Of Technology | Anti-dengue virus antibodies and uses thereof |
| AR092098A1 (es) | 2012-08-13 | 2015-03-25 | Regeneron Pharma | ANTICUERPOS ANTI-PCSK9 CON CARACTERISTICAS DE UNION DEPENDIENTES DEL pH |
| US9133269B2 (en) | 2012-08-24 | 2015-09-15 | Anaptysbio, Inc. | Humanized antibodies directed against complement protein C5 |
| CN113831406A (zh) | 2012-08-24 | 2021-12-24 | 中外制药株式会社 | FcγRIIb特异性Fc区变体 |
| WO2014030750A1 (ja) | 2012-08-24 | 2014-02-27 | 中外製薬株式会社 | マウスFcγRII特異的Fc抗体 |
| TW201922795A (zh) | 2012-09-10 | 2019-06-16 | 愛爾蘭商尼歐托普生物科學公司 | 抗mcam抗體及相關使用方法 |
| NZ628446A (en) | 2012-09-13 | 2017-06-30 | Bristol Myers Squibb Co | Fibronectin based scaffold domain proteins that bind to myostatin |
| TW201418707A (zh) | 2012-09-21 | 2014-05-16 | Alexion Pharma Inc | 補體組分c5拮抗劑之篩選分析 |
| SG11201503271XA (en) | 2012-11-06 | 2015-05-28 | Scholar Rock Inc | Compositions and methods for modulating cell signaling |
| WO2015089492A2 (en) | 2013-12-13 | 2015-06-18 | The Trustees Of The University Of Pennsylvania | Dna antibody constructs and method of using same |
| AR094271A1 (es) | 2012-12-21 | 2015-07-22 | Aveo Pharmaceuticals Inc | Anticuerpos anti-gdf15 |
| DK2940135T5 (da) | 2012-12-27 | 2021-09-20 | Chugai Pharmaceutical Co Ltd | Heterodimeriseret polypeptid |
| US9701759B2 (en) | 2013-01-14 | 2017-07-11 | Xencor, Inc. | Heterodimeric proteins |
| WO2014114651A1 (en) | 2013-01-24 | 2014-07-31 | Glaxosmithkline Intellectual Property Development Limited | Tnf-alpha antigen-binding proteins |
| JP6608702B2 (ja) | 2013-01-31 | 2019-11-20 | ソウル大学校産学協力団 | 補体関連疾患の予防及び治療のためのc5抗体及び方法 |
| JO3532B1 (ar) | 2013-03-13 | 2020-07-05 | Regeneron Pharma | الأجسام المضادة لمضاد انترلوكين-33 واستعمالاتها |
| US9481725B2 (en) | 2013-03-14 | 2016-11-01 | Alderbio Holdings, Llc | Antibodies to HGF and compositions containing |
| US20160038588A1 (en) | 2013-03-15 | 2016-02-11 | Amgen Inc. | Myostatin Antagonism in Human Subjects |
| WO2014150983A2 (en) | 2013-03-15 | 2014-09-25 | Amgen Inc. | Human antigen binding proteins that bind to proprotein convertase subtilisin kexin type 9 |
| US20160031985A1 (en) | 2013-03-15 | 2016-02-04 | Katherine S. Bowdish | Charge-engineered antibodies or compositions of penetration-enhanced targeting proteins and methods of use |
| CA2906128A1 (en) | 2013-03-15 | 2014-09-18 | Bayer Healthcare Llc | Anti-tfpi antibody variants with differential binding across ph range for improved pharmacokinetics |
| WO2014144080A2 (en) | 2013-03-15 | 2014-09-18 | Amgen Inc. | Human antigen binding proteins that bind to proprotein convertase subtilisin kexin type 9 |
| KR102561553B1 (ko) | 2013-03-15 | 2023-07-31 | 젠코어 인코포레이티드 | 이형이량체 단백질 |
| WO2014140366A1 (en) | 2013-03-15 | 2014-09-18 | Affibody Ab | New polypeptides |
| US10858417B2 (en) | 2013-03-15 | 2020-12-08 | Xencor, Inc. | Heterodimeric proteins |
| US9321686B2 (en) | 2013-03-15 | 2016-04-26 | Forta Corporation | Reinforcement fiber coating compositions, methods of making and treating, and uses for improved adhesion to asphalt and portland cement concrete |
| WO2014160958A1 (en) | 2013-03-29 | 2014-10-02 | Alexion Pharmaceuticals, Inc. | Compositions and methods for increasing the serum half-life of a therapeutic agent targeting complement c5 |
| CA2908350C (en) | 2013-04-02 | 2023-08-08 | Futa Mimoto | Fc region variant |
| CA2911514A1 (en) | 2013-05-06 | 2014-11-13 | Scholar Rock, Inc. | Compositions and methods for growth factor modulation |
| KR20160021125A (ko) | 2013-05-17 | 2016-02-24 | 쌩뜨레 나티오날 데 라 르세르쉬 생띠끄 (씨. 엔. 알. 에스) | 항-cxcl1, cxcl7 및 cxcl8 항체 및 이들의 용도 |
| US10111953B2 (en) | 2013-05-30 | 2018-10-30 | Regeneron Pharmaceuticals, Inc. | Methods for reducing remnant cholesterol and other lipoprotein fractions by administering an inhibitor of proprotein convertase subtilisin kexin-9 (PCSK9) |
| DK3004174T3 (da) | 2013-05-31 | 2019-07-22 | Zymeworks Inc | Heteromultimerer med reduceret eller nedreguleret effektorfunktion |
| EP3406633B1 (en) | 2013-07-25 | 2022-03-02 | Cytomx Therapeutics Inc. | Multispecific antibodies, multispecific activatable antibodies and methods of using the same |
| SG10201801063TA (en) | 2013-08-14 | 2018-04-27 | Novartis Ag | Methods of treating sporadic inclusion body myositis |
| JP2016531910A (ja) | 2013-08-16 | 2016-10-13 | アレクシオン ファーマシューティカルズ, インコーポレイテッド | 移植前の臓器への補体阻害剤の投与による移植拒絶の処置 |
| EP3042912A4 (en) | 2013-09-04 | 2017-05-03 | Protenova Co., Ltd. | Immunoglobulin-binding domain multimer |
| KR102150414B1 (ko) | 2013-09-18 | 2020-09-02 | 리제너론 파마슈티칼스 인코포레이티드 | 히스티딘 엔지니어링된 경쇄 항체 및 그것을 생성하기 위한 유전자 변형된 비-인간 동물 |
| EP2853898B1 (en) | 2013-09-27 | 2017-01-04 | Medizinische Hochschule Hannover | Analysis of myostatin in serum |
| US10155811B2 (en) | 2013-11-20 | 2018-12-18 | Regeneron Pharmaceuticals, Inc. | APLNR modulators and uses thereof |
| JP2017510622A (ja) | 2014-01-27 | 2017-04-13 | ノバルティス アーゲー | 筋萎縮を予測するバイオマーカー、方法および使用 |
| MX2016009991A (es) | 2014-02-11 | 2016-10-07 | Massachusetts Inst Technology | Nuevo anticuerpo anti-dengue de espectro completo. |
| WO2015122995A1 (en) | 2014-02-11 | 2015-08-20 | Visterra, Inc. | Antibody moleules to dengue virus and uses thereof |
| US9701743B2 (en) | 2014-02-20 | 2017-07-11 | Allergan, Inc. | Complement component C5 antibodies |
| NZ711451A (en) | 2014-03-07 | 2016-05-27 | Alexion Pharma Inc | Anti-c5 antibodies having improved pharmacokinetics |
| JP6451279B2 (ja) | 2014-03-31 | 2019-01-16 | 信越化学工業株式会社 | フルオロポリエーテル基含有ポリマー変性シラン、表面処理剤及び物品 |
| TW201622746A (zh) | 2014-04-24 | 2016-07-01 | 諾華公司 | 改善或加速髖部骨折術後身體復原之方法 |
| GB201413086D0 (en) | 2014-07-23 | 2014-09-03 | Imp Innovations Ltd And Inst Pasteur | Methods |
| US9688717B2 (en) | 2014-09-10 | 2017-06-27 | Purdue Research Foundation | Raman tag |
| US10307480B2 (en) | 2014-11-06 | 2019-06-04 | Scholar Rock, Inc. | Anti-pro/latent-myostatin antibodies and uses thereof |
| WO2016073906A2 (en) | 2014-11-06 | 2016-05-12 | Scholar Rock, Inc. | Transforming growth factor-related immunoassays |
| CA2969800A1 (en) | 2014-12-08 | 2016-06-16 | Novartis Ag | Myostatin or activin antagonists for the treatment of sarcopenia |
| PE20171111A1 (es) | 2014-12-19 | 2017-08-07 | Chugai Pharmaceutical Co Ltd | Anticuerpos antimiostatina, polipeptidos que contienen regiones fc variantes, y metodos de uso |
| TW201809008A (zh) | 2014-12-19 | 2018-03-16 | 日商中外製藥股份有限公司 | 抗c5抗體及使用方法 |
| US20180016327A1 (en) | 2015-01-22 | 2018-01-18 | Chugai Seiyaku Kabushiki Kaisha | A Combination of Two or More Anti-C5 Antibodies and Methods of Use |
| EP3271390B1 (en) | 2015-03-17 | 2023-09-27 | Agency For Science, Technology And Research | A serotype cross-reactive, dengue neutralizing antibody and uses thereof |
| JP6944375B2 (ja) | 2015-03-31 | 2021-10-06 | アレクシオン ファーマシューティカルズ, インコーポレイテッド | 発作性夜間血色素尿症(pnh)患者の亜集団の同定および処置 |
| CA2982810A1 (en) | 2015-04-15 | 2016-10-20 | Regeneron Pharmaceuticals, Inc. | Methods of increasing strength and functionality with gdf8 inhibitors |
| US10940126B2 (en) | 2015-07-03 | 2021-03-09 | Camilla Svensson | Inhibition of IL-8 in the treatment of pain and/or bone loss |
| CN113896789A (zh) | 2015-09-15 | 2022-01-07 | 供石公司 | 抗-原肌生长抑制素/潜伏肌生长抑制素抗体及其用途 |
| AU2016372934B2 (en) | 2015-12-18 | 2023-10-05 | Chugai Seiyaku Kabushiki Kaisha | Anti-myostatin antibodies, polypeptides containing variant Fc regions, and methods of use |
| SG11201610782YA (en) | 2015-12-18 | 2017-07-28 | Chugai Pharmaceutical Co Ltd | Anti-c5 antibodies and methods of use |
| US10233252B2 (en) | 2015-12-21 | 2019-03-19 | Wisconsin Alumni Research Foundation | pH-dependent antibodies targeting the transferrin receptor and methods of use thereof to deliver a therapeutic agent |
| US11359009B2 (en) | 2015-12-25 | 2022-06-14 | Chugai Seiyaku Kabushiki Kaisha | Anti-myostatin antibodies and methods of use |
| JP2019504064A (ja) | 2016-01-08 | 2019-02-14 | スカラー ロック インコーポレイテッドScholar Rock,Inc. | 抗プロ/潜在型ミオスタチン抗体およびその使用方法 |
| US20190023775A1 (en) | 2016-01-11 | 2019-01-24 | Alexion Pharmaceuticals, Inc. | Dosage and administration of anti-c5 antibodies for treatment |
| GB201610162D0 (en) | 2016-06-10 | 2016-07-27 | Imp Innovations Ltd And Inst Pasteur | Methods |
| JP6823167B2 (ja) | 2016-06-13 | 2021-01-27 | スカラー ロック インコーポレイテッドScholar Rock,Inc. | ミオスタチン阻害剤の使用および併用療法 |
| IL309975A (en) | 2016-06-14 | 2024-03-01 | Regeneron Pharma | Antibodies against C5 and their uses |
| TWI789369B (zh) | 2016-06-17 | 2023-01-11 | 日商中外製藥股份有限公司 | 抗c5抗體及使用方法 |
| TW202228778A (zh) | 2016-06-17 | 2022-08-01 | 日商中外製藥股份有限公司 | 抗肌抑素抗體及使用方法 |
| WO2018025982A1 (ja) | 2016-08-05 | 2018-02-08 | 中外製薬株式会社 | Il-8関連疾患の治療用又は予防用組成物 |
| SG10201607778XA (en) | 2016-09-16 | 2018-04-27 | Chugai Pharmaceutical Co Ltd | Anti-Dengue Virus Antibodies, Polypeptides Containing Variant Fc Regions, And Methods Of Use |
| SG11201801401UA (en) | 2017-01-31 | 2018-09-27 | Chugai Pharmaceutical Co Ltd | A pharmaceutical composition for use in the treatment or prevention of a c5-related disease and a method for treating or preventing a c5-related disease |
| JP7211961B2 (ja) | 2017-03-14 | 2023-01-24 | ファイヴ プライム セラピューティクス インク | 酸性pHでVISTAに結合する抗体 |
| CA3055251A1 (en) | 2017-03-16 | 2018-09-20 | Medimmune Limited | Anti-par2 antibodies and uses thereof |
| JP2020515643A (ja) | 2017-04-03 | 2020-05-28 | インフラルクス・ゲゼルシャフト・ミット・ベシュレンクテル・ハフツングInflaRx GmbH | C5a活性のインヒビターでの炎症性疾患の処置 |
| CN116327926A (zh) | 2018-03-15 | 2023-06-27 | 中外制药株式会社 | 对寨卡病毒具有交叉反应性的抗登革热病毒抗体及使用方法 |
| KR20200033348A (ko) | 2018-08-01 | 2020-03-27 | 추가이 세이야쿠 가부시키가이샤 | C5-관련 질환의 치료 또는 예방용 의약 조성물 및 c5-관련 질환을 치료 또는 예방하기 위한 방법 |
| KR20210149779A (ko) | 2019-04-10 | 2021-12-09 | 추가이 세이야쿠 가부시키가이샤 | Fc 영역 개변 항체의 정제 방법 |
-
2017
- 2017-08-04 WO PCT/JP2017/028346 patent/WO2018025982A1/ja active Application Filing
- 2017-08-04 IL IL264626A patent/IL264626B/en unknown
- 2017-08-04 BR BR112019001902A patent/BR112019001902A2/pt unknown
- 2017-08-04 SG SG11201801024XA patent/SG11201801024XA/en unknown
- 2017-08-04 MX MX2019001448A patent/MX2019001448A/es unknown
- 2017-08-04 AU AU2017305073A patent/AU2017305073B2/en active Active
- 2017-08-04 TW TW106126372A patent/TWI693940B/zh active
- 2017-08-04 RU RU2019105510A patent/RU2766112C2/ru active
- 2017-08-04 CN CN202310196594.7A patent/CN116271014A/zh active Pending
- 2017-08-04 KR KR1020177027051A patent/KR102102734B1/ko active IP Right Grant
- 2017-08-04 CN CN201780046902.0A patent/CN109689099B/zh active Active
- 2017-08-04 US US16/323,142 patent/US11053308B2/en active Active
- 2017-08-04 CN CN202310193929.XA patent/CN116251182A/zh active Pending
- 2017-08-04 CA CA3026050A patent/CA3026050A1/en active Pending
- 2017-08-04 KR KR1020237017761A patent/KR20230079499A/ko not_active Application Discontinuation
- 2017-08-04 EP EP17837083.9A patent/EP3494991A4/en active Pending
- 2017-08-04 TW TW111130380A patent/TW202300168A/zh unknown
- 2017-08-04 KR KR1020207009872A patent/KR102538749B1/ko not_active Application Discontinuation
- 2017-08-04 JP JP2018531994A patent/JP6527643B2/ja active Active
-
2019
- 2019-02-01 MX MX2022010229A patent/MX2022010229A/es unknown
- 2019-05-10 JP JP2019089724A patent/JP7193803B2/ja active Active
-
2021
- 2021-05-28 US US17/333,256 patent/US11780912B2/en active Active
-
2022
- 2022-12-01 JP JP2022192775A patent/JP2023022238A/ja active Pending
-
2023
- 2023-08-16 US US18/450,863 patent/US20240083996A1/en active Pending
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08217799A (ja) * | 1994-07-13 | 1996-08-27 | Chugai Pharmaceut Co Ltd | ヒトインターロイキン−8に対する再構成ヒト抗体 |
| JP2009541352A (ja) * | 2006-06-23 | 2009-11-26 | スミスクライン・ビーチャム・コーポレイション | 治療方法 |
| JP2014055145A (ja) * | 2006-06-23 | 2014-03-27 | Glaxosmithkline Llc | Il−8受容体アンタゴニスト |
| WO2009026117A2 (en) * | 2007-08-16 | 2009-02-26 | Glaxo Group Limited | Novel compounds |
| JP2013518606A (ja) * | 2010-02-09 | 2013-05-23 | グラクソスミスクライン・リミテッド・ライアビリティ・カンパニー | 新規使用 |
| WO2016125495A1 (en) * | 2015-02-05 | 2016-08-11 | Chugai Seiyaku Kabushiki Kaisha | Antibodies comprising an ion concentration dependent antigen-binding domain, fc region variants, il-8-binding antibodies, and uses therof |
| WO2017046994A1 (en) * | 2015-09-18 | 2017-03-23 | Chugai Seiyaku Kabushiki Kaisha | Il-8-binding antibodies and uses thereof |
Non-Patent Citations (1)
| Title |
|---|
| IWABE, T. ET AL.: "Pathogenetic significance of increased levels of interleukin-8 in the peritoneal fluid of patients with endometriosis", FERTILITY AND STERILITY, vol. 69, no. 5, 1998, pages 924 - 930, XP055460270, DOI: doi:10.1016/S0015-0282(98)00049-1 * |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11046784B2 (en) | 2006-03-31 | 2021-06-29 | Chugai Seiyaku Kabushiki Kaisha | Methods for controlling blood pharmacokinetics of antibodies |
| US11248053B2 (en) | 2007-09-26 | 2022-02-15 | Chugai Seiyaku Kabushiki Kaisha | Method of modifying isoelectric point of antibody via amino acid substitution in CDR |
| US11371039B2 (en) | 2008-04-11 | 2022-06-28 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule capable of binding to two or more antigen molecules repeatedly |
| US11359194B2 (en) | 2008-04-11 | 2022-06-14 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule capable of binding two or more antigen molecules repeatedly |
| US11891434B2 (en) | 2010-11-30 | 2024-02-06 | Chugai Seiyaku Kabushiki Kaisha | Antigen-binding molecule capable of binding to plurality of antigen molecules repeatedly |
| US11236168B2 (en) | 2012-08-24 | 2022-02-01 | Chugai Seiyaku Kabushiki Kaisha | Mouse FcγammaRII-specific Fc antibody |
| US10919953B2 (en) | 2012-08-24 | 2021-02-16 | Chugai Seiyaku Kabushiki Kaisha | FcgammaRIIB-specific Fc region variant |
| US11267868B2 (en) | 2013-04-02 | 2022-03-08 | Chugai Seiyaku Kabushiki Kaisha | Fc region variant |
| US11454633B2 (en) | 2014-12-19 | 2022-09-27 | Chugai Seiyaku Kabushiki Kaisha | Anti-myostatin antibodies, polypeptides containing variant Fc regions, and methods of use |
| US11180548B2 (en) | 2015-02-05 | 2021-11-23 | Chugai Seiyaku Kabushiki Kaisha | Methods of neutralizing IL-8 biological activity |
| US11359009B2 (en) | 2015-12-25 | 2022-06-14 | Chugai Seiyaku Kabushiki Kaisha | Anti-myostatin antibodies and methods of use |
| US11053308B2 (en) | 2016-08-05 | 2021-07-06 | Chugai Seiyaku Kabushiki Kaisha | Method for treating IL-8-related diseases |
| US11780912B2 (en) | 2016-08-05 | 2023-10-10 | Chugai Seiyaku Kabushiki Kaisha | Composition for prophylaxis or treatment of IL-8 related diseases |
| US10844113B2 (en) | 2016-09-16 | 2020-11-24 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies, polypeptides containing variant Fc regions, and methods of use |
| US10604561B2 (en) | 2016-09-16 | 2020-03-31 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies, polypeptides containing variant Fc regions, and methods of use |
| US11780908B2 (en) | 2016-09-16 | 2023-10-10 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies, polypeptides containing variant FC regions, and methods of use |
| US11891432B2 (en) | 2018-03-15 | 2024-02-06 | Chugai Seiyaku Kabushiki Kaisha | Anti-dengue virus antibodies having cross-reactivity to Zika virus and methods of use |
| CN113544157A (zh) * | 2019-04-19 | 2021-10-22 | 中外制药株式会社 | 用于检测或捕获样品中的多肽的抗体和组合物,以及用于检测或捕获样品中的多肽的方法 |
| WO2020213706A1 (en) * | 2019-04-19 | 2020-10-22 | Chugai Seiyaku Kabushiki Kaisha | Antibodies and compositions for use in detecting or capturing a polypeptide in a sample, and methods for detecting or capturing a polypeptide in a sample |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7193803B2 (ja) | Il-8関連疾患の治療用又は予防用組成物 | |
| KR102473544B1 (ko) | 항체-fynomer 접합체 | |
| CA2960297A1 (en) | Anti-interleukin-33 antibodies and uses thereof | |
| JP2021502961A (ja) | 高リスク多発性骨髄腫の治療方法 | |
| WO2011031695A1 (en) | High affinity human antibodies to human protease-activated receptor-2 | |
| KR102513989B1 (ko) | 간 질환을 치료하거나 예방하는 방법 | |
| MX2014004662A (es) | Antagonistas de il17c para el tratamiento de trastornos inlflamatorios. | |
| JP2023548878A (ja) | 中和抗sars-cov-2抗体 | |
| JP2023516727A (ja) | コロナウイルス誘発性急性呼吸窮迫症候群の処置および/または予防のためにmasp-2を阻害する方法 | |
| US20230279129A1 (en) | Method of treating or preventing ischemia-reperfusion injury | |
| TWI831965B (zh) | Il-8相關疾病之治療用或預防用組成物 | |
| TWI834025B (zh) | 用於治療和/或預防冠狀病毒誘導的急性呼吸窘迫症候群的抑制masp-2的方法 | |
| US20220281962A1 (en) | Tetranectin-targeting monoclonal antibodies to fight against lethal sepsis and other pathologies | |
| JP2024512279A (ja) | 異常子宮出血の処置における使用のためのil-11又はil-11raの阻害剤 | |
| WO2023168087A1 (en) | Methods and compositions for treating and preventing fibrosis | |
| CA3206789A1 (en) | Biomarker for assessing the risk of developing acute covid-19 and post-acute covid-19 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 20177027051 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11201801024X Country of ref document: SG |
|
| ENP | Entry into the national phase |
Ref document number: 2018531994 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17837083 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3026050 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2017305073 Country of ref document: AU Date of ref document: 20170804 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 122023002253 Country of ref document: BR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112019001902 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2017837083 Country of ref document: EP Effective date: 20190305 |
|
| ENP | Entry into the national phase |
Ref document number: 112019001902 Country of ref document: BR Kind code of ref document: A2 Effective date: 20190130 |