EP2849264A1 - Sekundärbatterie mit nichtwässrigem Elektrolyten - Google Patents
Sekundärbatterie mit nichtwässrigem Elektrolyten Download PDFInfo
- Publication number
- EP2849264A1 EP2849264A1 EP14003948.8A EP14003948A EP2849264A1 EP 2849264 A1 EP2849264 A1 EP 2849264A1 EP 14003948 A EP14003948 A EP 14003948A EP 2849264 A1 EP2849264 A1 EP 2849264A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- positive electrode
- active material
- secondary battery
- electrode active
- battery
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 239000011255 nonaqueous electrolyte Substances 0.000 title claims abstract description 48
- 239000007774 positive electrode material Substances 0.000 claims abstract description 105
- 239000003792 electrolyte Substances 0.000 claims abstract description 72
- 229910052751 metal Inorganic materials 0.000 claims abstract description 49
- 239000002184 metal Substances 0.000 claims abstract description 49
- 239000011888 foil Substances 0.000 claims abstract description 36
- 238000004806 packaging method and process Methods 0.000 claims abstract description 36
- 239000002253 acid Substances 0.000 claims abstract description 33
- 229910003002 lithium salt Inorganic materials 0.000 claims abstract description 28
- 159000000002 lithium salts Chemical class 0.000 claims abstract description 28
- 229910052723 transition metal Inorganic materials 0.000 claims abstract description 17
- 150000003624 transition metals Chemical class 0.000 claims abstract description 16
- -1 Lib4 Inorganic materials 0.000 claims description 54
- 229920000642 polymer Polymers 0.000 claims description 39
- 229910052782 aluminium Inorganic materials 0.000 claims description 29
- 239000000463 material Substances 0.000 claims description 29
- 229910052744 lithium Inorganic materials 0.000 claims description 27
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 26
- 150000001875 compounds Chemical class 0.000 claims description 19
- 229920005989 resin Polymers 0.000 claims description 17
- 239000011347 resin Substances 0.000 claims description 17
- 239000011248 coating agent Substances 0.000 claims description 15
- 238000000576 coating method Methods 0.000 claims description 15
- 239000002131 composite material Substances 0.000 claims description 9
- 229920005569 poly(vinylidene fluoride-co-hexafluoropropylene) Polymers 0.000 claims description 9
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 8
- 229920003171 Poly (ethylene oxide) Polymers 0.000 claims description 8
- 229910052759 nickel Inorganic materials 0.000 claims description 8
- 229910052748 manganese Inorganic materials 0.000 claims description 6
- 229910013191 LiMO2 Inorganic materials 0.000 claims description 5
- 229910013755 LiNiyCo1-yO2 Inorganic materials 0.000 claims description 5
- 229910013764 LiNiyCo1—yO2 Inorganic materials 0.000 claims description 5
- 229910002097 Lithium manganese(III,IV) oxide Inorganic materials 0.000 claims description 5
- 229920000058 polyacrylate Polymers 0.000 claims description 5
- 239000000565 sealant Substances 0.000 claims description 5
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 claims description 4
- 229910003005 LiNiO2 Inorganic materials 0.000 claims description 4
- 229910018698 LixCo1-yAlyO2 Inorganic materials 0.000 claims description 4
- 229910018705 LixCo1−yAlyO2 Inorganic materials 0.000 claims description 4
- 229910052796 boron Inorganic materials 0.000 claims description 4
- 150000002148 esters Chemical class 0.000 claims description 4
- 239000011737 fluorine Substances 0.000 claims description 4
- 229910052731 fluorine Inorganic materials 0.000 claims description 4
- 229910052733 gallium Inorganic materials 0.000 claims description 4
- 229910052738 indium Inorganic materials 0.000 claims description 4
- 229920000193 polymethacrylate Polymers 0.000 claims description 4
- 229910001560 Li(CF3SO2)2N Inorganic materials 0.000 claims description 3
- 229910001559 LiC4F9SO3 Inorganic materials 0.000 claims description 3
- 229910000552 LiCF3SO3 Inorganic materials 0.000 claims description 3
- 239000007864 aqueous solution Substances 0.000 claims description 3
- 229910001540 lithium hexafluoroarsenate(V) Inorganic materials 0.000 claims description 3
- 229910032387 LiCoO2 Inorganic materials 0.000 claims 2
- 229910001290 LiPF6 Inorganic materials 0.000 claims 1
- 229910003983 NiyM1-yO2 Inorganic materials 0.000 claims 1
- 239000007773 negative electrode material Substances 0.000 abstract description 19
- 239000002861 polymer material Substances 0.000 abstract description 10
- 239000003125 aqueous solvent Substances 0.000 abstract description 5
- 239000007787 solid Substances 0.000 abstract description 2
- 239000010408 film Substances 0.000 description 65
- 239000000203 mixture Substances 0.000 description 54
- 239000002245 particle Substances 0.000 description 52
- 230000000052 comparative effect Effects 0.000 description 40
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 25
- 239000008151 electrolyte solution Substances 0.000 description 24
- 229910021439 lithium cobalt complex oxide Inorganic materials 0.000 description 24
- 238000003860 storage Methods 0.000 description 18
- 238000006243 chemical reaction Methods 0.000 description 15
- 239000007789 gas Substances 0.000 description 15
- XGZVUEUWXADBQD-UHFFFAOYSA-L lithium carbonate Chemical compound [Li+].[Li+].[O-]C([O-])=O XGZVUEUWXADBQD-UHFFFAOYSA-L 0.000 description 14
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 13
- 239000004698 Polyethylene Substances 0.000 description 13
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 13
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 12
- 238000001035 drying Methods 0.000 description 12
- 229910052808 lithium carbonate Inorganic materials 0.000 description 12
- 238000000034 method Methods 0.000 description 12
- 239000005020 polyethylene terephthalate Substances 0.000 description 12
- 229920000139 polyethylene terephthalate Polymers 0.000 description 12
- 239000002904 solvent Substances 0.000 description 11
- 230000008859 change Effects 0.000 description 10
- 239000004743 Polypropylene Substances 0.000 description 9
- 229920000573 polyethylene Polymers 0.000 description 9
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 8
- 239000002033 PVDF binder Substances 0.000 description 8
- 229910002092 carbon dioxide Inorganic materials 0.000 description 8
- 239000011245 gel electrolyte Substances 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- 229920001155 polypropylene Polymers 0.000 description 8
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 8
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 8
- 239000002002 slurry Substances 0.000 description 8
- 229910045601 alloy Inorganic materials 0.000 description 7
- 239000000956 alloy Substances 0.000 description 7
- 238000002156 mixing Methods 0.000 description 7
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 229920001577 copolymer Polymers 0.000 description 6
- 239000011572 manganese Substances 0.000 description 6
- 239000004065 semiconductor Substances 0.000 description 6
- 239000007784 solid electrolyte Substances 0.000 description 6
- 238000003466 welding Methods 0.000 description 6
- SOGAXMICEFXMKE-UHFFFAOYSA-N Butylmethacrylate Chemical compound CCCCOC(=O)C(C)=C SOGAXMICEFXMKE-UHFFFAOYSA-N 0.000 description 5
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 5
- 239000004677 Nylon Substances 0.000 description 5
- 239000011230 binding agent Substances 0.000 description 5
- 239000001569 carbon dioxide Substances 0.000 description 5
- 238000000354 decomposition reaction Methods 0.000 description 5
- 230000006866 deterioration Effects 0.000 description 5
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 239000007772 electrode material Substances 0.000 description 5
- 229910000040 hydrogen fluoride Inorganic materials 0.000 description 5
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Chemical compound [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 5
- 229920001778 nylon Polymers 0.000 description 5
- 229920006254 polymer film Polymers 0.000 description 5
- YLZOPXRUQYQQID-UHFFFAOYSA-N 3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-1-[4-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]piperazin-1-yl]propan-1-one Chemical compound N1N=NC=2CN(CCC=21)CCC(=O)N1CCN(CC1)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F YLZOPXRUQYQQID-UHFFFAOYSA-N 0.000 description 4
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 4
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 4
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 4
- 230000006835 compression Effects 0.000 description 4
- 238000007906 compression Methods 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- HCDGVLDPFQMKDK-UHFFFAOYSA-N hexafluoropropylene Chemical group FC(F)=C(F)C(F)(F)F HCDGVLDPFQMKDK-UHFFFAOYSA-N 0.000 description 4
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 4
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 4
- 229910001416 lithium ion Inorganic materials 0.000 description 4
- 229920001684 low density polyethylene Polymers 0.000 description 4
- 239000004702 low-density polyethylene Substances 0.000 description 4
- 150000002739 metals Chemical class 0.000 description 4
- 230000000704 physical effect Effects 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 description 3
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 3
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 3
- 239000011149 active material Substances 0.000 description 3
- 239000006229 carbon black Substances 0.000 description 3
- 238000005266 casting Methods 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 239000010949 copper Substances 0.000 description 3
- 239000011889 copper foil Substances 0.000 description 3
- 230000007797 corrosion Effects 0.000 description 3
- 238000005260 corrosion Methods 0.000 description 3
- 238000007599 discharging Methods 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 230000004927 fusion Effects 0.000 description 3
- 229910002804 graphite Inorganic materials 0.000 description 3
- 239000010439 graphite Substances 0.000 description 3
- 150000004678 hydrides Chemical class 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 229920000092 linear low density polyethylene Polymers 0.000 description 3
- 239000004707 linear low-density polyethylene Substances 0.000 description 3
- 229910001496 lithium tetrafluoroborate Inorganic materials 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 239000007769 metal material Substances 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 229920006284 nylon film Polymers 0.000 description 3
- 230000002093 peripheral effect Effects 0.000 description 3
- 229920000098 polyolefin Polymers 0.000 description 3
- 229910001220 stainless steel Inorganic materials 0.000 description 3
- 239000010935 stainless steel Substances 0.000 description 3
- ZZXUZKXVROWEIF-UHFFFAOYSA-N 1,2-butylene carbonate Chemical compound CCC1COC(=O)O1 ZZXUZKXVROWEIF-UHFFFAOYSA-N 0.000 description 2
- GEWWCWZGHNIUBW-UHFFFAOYSA-N 1-(4-nitrophenyl)propan-2-one Chemical compound CC(=O)CC1=CC=C([N+]([O-])=O)C=C1 GEWWCWZGHNIUBW-UHFFFAOYSA-N 0.000 description 2
- CRMJLJFDPNJIQA-UHFFFAOYSA-N 2,4-difluoro-1-methoxybenzene Chemical compound COC1=CC=C(F)C=C1F CRMJLJFDPNJIQA-UHFFFAOYSA-N 0.000 description 2
- KBTMGSMZIKLAHN-UHFFFAOYSA-N 4-bromo-1,2-dimethoxybenzene Chemical compound COC1=CC=C(Br)C=C1OC KBTMGSMZIKLAHN-UHFFFAOYSA-N 0.000 description 2
- NLHHRLWOUZZQLW-UHFFFAOYSA-N Acrylonitrile Chemical compound C=CC#N NLHHRLWOUZZQLW-UHFFFAOYSA-N 0.000 description 2
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 2
- 229910012925 LiCoO3 Inorganic materials 0.000 description 2
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- 229910021383 artificial graphite Inorganic materials 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical compound CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 2
- 239000003575 carbonaceous material Substances 0.000 description 2
- 239000000571 coke Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 2
- 239000011808 electrode reactant Substances 0.000 description 2
- GAEKPEKOJKCEMS-UHFFFAOYSA-N gamma-valerolactone Chemical compound CC1CCC(=O)O1 GAEKPEKOJKCEMS-UHFFFAOYSA-N 0.000 description 2
- 229920001903 high density polyethylene Polymers 0.000 description 2
- 239000004700 high-density polyethylene Substances 0.000 description 2
- 238000010030 laminating Methods 0.000 description 2
- 239000011244 liquid electrolyte Substances 0.000 description 2
- MHCFAGZWMAWTNR-UHFFFAOYSA-M lithium perchlorate Chemical compound [Li+].[O-]Cl(=O)(=O)=O MHCFAGZWMAWTNR-UHFFFAOYSA-M 0.000 description 2
- 229910001486 lithium perchlorate Inorganic materials 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- 235000012771 pancakes Nutrition 0.000 description 2
- 238000004445 quantitative analysis Methods 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 238000004448 titration Methods 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- DHKHKXVYLBGOIT-UHFFFAOYSA-N 1,1-Diethoxyethane Chemical compound CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 1
- IOBWAHRFIPQEQL-UHFFFAOYSA-N 1,3-difluoro-2-methoxybenzene Chemical compound COC1=C(F)C=CC=C1F IOBWAHRFIPQEQL-UHFFFAOYSA-N 0.000 description 1
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 1
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 1
- HMZNNQMHGDXAHG-UHFFFAOYSA-N 1-cyanoethenyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC(=C)C#N HMZNNQMHGDXAHG-UHFFFAOYSA-N 0.000 description 1
- YENQKAGAGMQTRZ-UHFFFAOYSA-N 1-cyanoethenyl prop-2-enoate Chemical compound C=CC(=O)OC(=C)C#N YENQKAGAGMQTRZ-UHFFFAOYSA-N 0.000 description 1
- JAHNSTQSQJOJLO-UHFFFAOYSA-N 2-(3-fluorophenyl)-1h-imidazole Chemical compound FC1=CC=CC(C=2NC=CN=2)=C1 JAHNSTQSQJOJLO-UHFFFAOYSA-N 0.000 description 1
- JWUJQDFVADABEY-UHFFFAOYSA-N 2-methyltetrahydrofuran Chemical compound CC1CCCO1 JWUJQDFVADABEY-UHFFFAOYSA-N 0.000 description 1
- ZPLCXHWYPWVJDL-UHFFFAOYSA-N 4-[(4-hydroxyphenyl)methyl]-1,3-oxazolidin-2-one Chemical compound C1=CC(O)=CC=C1CC1NC(=O)OC1 ZPLCXHWYPWVJDL-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- 239000004925 Acrylic resin Substances 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- 229910004706 CaSi2 Inorganic materials 0.000 description 1
- 229920000049 Carbon (fiber) Polymers 0.000 description 1
- 239000004215 Carbon black (E152) Substances 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 229910021359 Chromium(II) silicide Inorganic materials 0.000 description 1
- 229910018999 CoSi2 Inorganic materials 0.000 description 1
- 239000004971 Cross linker Substances 0.000 description 1
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 229910005331 FeSi2 Inorganic materials 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- 229910015530 LixMO2 Inorganic materials 0.000 description 1
- 229910014514 LixNiyM1-yO2 Inorganic materials 0.000 description 1
- 229910014512 LixNiyM1−yO2 Inorganic materials 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- 229910019752 Mg2Si Inorganic materials 0.000 description 1
- 229910019743 Mg2Sn Inorganic materials 0.000 description 1
- 229910017025 MnSi2 Inorganic materials 0.000 description 1
- 229910020968 MoSi2 Inorganic materials 0.000 description 1
- 229910020044 NbSi2 Inorganic materials 0.000 description 1
- 229910005487 Ni2Si Inorganic materials 0.000 description 1
- 229910012990 NiSi2 Inorganic materials 0.000 description 1
- 229920000459 Nitrile rubber Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 229910003685 SiB4 Inorganic materials 0.000 description 1
- 229910003682 SiB6 Inorganic materials 0.000 description 1
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 1
- 229910004217 TaSi2 Inorganic materials 0.000 description 1
- 229910008479 TiSi2 Inorganic materials 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 1
- 229910008814 WSi2 Inorganic materials 0.000 description 1
- 229910007659 ZnSi2 Inorganic materials 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- 229940117913 acrylamide Drugs 0.000 description 1
- XECAHXYUAAWDEL-UHFFFAOYSA-N acrylonitrile butadiene styrene Chemical compound C=CC=C.C=CC#N.C=CC1=CC=CC=C1 XECAHXYUAAWDEL-UHFFFAOYSA-N 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- DFJQEGUNXWZVAH-UHFFFAOYSA-N bis($l^{2}-silanylidene)titanium Chemical compound [Si]=[Ti]=[Si] DFJQEGUNXWZVAH-UHFFFAOYSA-N 0.000 description 1
- 238000007664 blowing Methods 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 229940006460 bromide ion Drugs 0.000 description 1
- 238000001354 calcination Methods 0.000 description 1
- 239000004917 carbon fiber Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- GRFFKYTUNTWAGG-UHFFFAOYSA-N chloroethene;prop-2-enenitrile Chemical compound ClC=C.C=CC#N GRFFKYTUNTWAGG-UHFFFAOYSA-N 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 150000001993 dienes Chemical class 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 1
- AERPLAKASWUTNP-UHFFFAOYSA-N ethyl prop-2-enoate;hydrate Chemical compound O.CCOC(=O)C=C AERPLAKASWUTNP-UHFFFAOYSA-N 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 239000007849 furan resin Substances 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- GPRLSGONYQIRFK-UHFFFAOYSA-N hydron Chemical compound [H+] GPRLSGONYQIRFK-UHFFFAOYSA-N 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- KQNPFQTWMSNSAP-UHFFFAOYSA-N isobutyric acid Chemical compound CC(C)C(O)=O KQNPFQTWMSNSAP-UHFFFAOYSA-N 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- ZOSSPMWRLDTUKE-UHFFFAOYSA-N methyl prop-2-enoate;hydrate Chemical compound O.COC(=O)C=C ZOSSPMWRLDTUKE-UHFFFAOYSA-N 0.000 description 1
- LVHBHZANLOWSRM-UHFFFAOYSA-N methylenebutanedioic acid Natural products OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000011331 needle coke Substances 0.000 description 1
- 239000000615 nonconductor Substances 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 239000002006 petroleum coke Substances 0.000 description 1
- 239000006253 pitch coke Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920001197 polyacetylene Polymers 0.000 description 1
- 229920002239 polyacrylonitrile Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920005672 polyolefin resin Polymers 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- 238000004804 winding Methods 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/36—Accumulators not provided for in groups H01M10/05-H01M10/34
- H01M10/38—Construction or manufacture
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0565—Polymeric materials, e.g. gel-type or solid-type
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings; Jackets or wrappings
- H01M50/116—Primary casings; Jackets or wrappings characterised by the material
- H01M50/117—Inorganic material
- H01M50/119—Metals
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings; Jackets or wrappings
- H01M50/116—Primary casings; Jackets or wrappings characterised by the material
- H01M50/121—Organic material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings; Jackets or wrappings
- H01M50/116—Primary casings; Jackets or wrappings characterised by the material
- H01M50/124—Primary casings; Jackets or wrappings characterised by the material having a layered structure
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings; Jackets or wrappings
- H01M50/131—Primary casings; Jackets or wrappings characterised by physical properties, e.g. gas permeability, size or heat resistance
- H01M50/133—Thickness
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/10—Primary casings; Jackets or wrappings
- H01M50/14—Primary casings; Jackets or wrappings for protecting against damage caused by external factors
- H01M50/141—Primary casings; Jackets or wrappings for protecting against damage caused by external factors for protecting against humidity
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/50—Current conducting connections for cells or batteries
- H01M50/543—Terminals
- H01M50/547—Terminals characterised by the disposition of the terminals on the cells
- H01M50/55—Terminals characterised by the disposition of the terminals on the cells on the same side of the cell
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M50/00—Constructional details or processes of manufacture of the non-active parts of electrochemical cells other than fuel cells, e.g. hybrid cells
- H01M50/50—Current conducting connections for cells or batteries
- H01M50/543—Terminals
- H01M50/552—Terminals characterised by their shape
- H01M50/553—Terminals adapted for prismatic, pouch or rectangular cells
- H01M50/557—Plate-shaped terminals
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0567—Liquid materials characterised by the additives
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/058—Construction or manufacture
- H01M10/0587—Construction or manufacture of accumulators having only wound construction elements, i.e. wound positive electrodes, wound negative electrodes and wound separators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/42—Methods or arrangements for servicing or maintenance of secondary cells or secondary half-cells
- H01M10/4235—Safety or regulating additives or arrangements in electrodes, separators or electrolyte
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0065—Solid electrolytes
- H01M2300/0082—Organic polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0085—Immobilising or gelification of electrolyte
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/133—Electrodes based on carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/139—Processes of manufacture
- H01M4/1391—Processes of manufacture of electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/139—Processes of manufacture
- H01M4/1393—Processes of manufacture of electrodes based on carbonaceous material, e.g. graphite-intercalation compounds or CFx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M6/00—Primary cells; Manufacture thereof
- H01M6/04—Cells with aqueous electrolyte
- H01M6/06—Dry cells, i.e. cells wherein the electrolyte is rendered non-fluid
- H01M6/10—Dry cells, i.e. cells wherein the electrolyte is rendered non-fluid with wound or folded electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M6/00—Primary cells; Manufacture thereof
- H01M6/14—Cells with non-aqueous electrolyte
- H01M6/16—Cells with non-aqueous electrolyte with organic electrolyte
- H01M6/162—Cells with non-aqueous electrolyte with organic electrolyte characterised by the electrolyte
- H01M6/168—Cells with non-aqueous electrolyte with organic electrolyte characterised by the electrolyte by additives
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present invention relates to a battery in which a battery device having electrolyte as well as a positive electrode and a negative electrode is sealed in a film-state packaging member.
- each of such secondary batteries is generally constructed by interposing a liquid electrolyte (hereinbelow, also called electrolyte solution) obtained by dissolving a lithium salt into a nonaqueous solvent between a positive electrode and a negative electrode and accommodating them in a housing made of a metal.
- a liquid electrolyte hereinbelow, also called electrolyte solution
- a metal hard case cell (a positive electrode cover and a negative electrode can) having rigidity.
- a problem such as leakage occurs. It is therefore proposed to use, in place of the electrolyte solution, a gel electrolyte obtained by making a non-aqueous electrolyte solution containing a lithium salt held by a polymer compound, a solid electrolyte obtained by dispersing or mixing a lithium salt into a polymer compound having ion conductivity, or an electrolyte in which a lithium salt is held by a solid inorganic conductor.
- This non-aqueous gel polymer secondary battery has a positive electrode having a positive electrode collector on which a positive electrode active material layer is formed, and a negative electrode having a negative electrode collector on which a negative electrode active material is formed and has a structure that a gel layer containing an electrolyte is sandwiched between the positive electrode active material layer of the positive electrode and the negative electrode active layer of the negative electrode.
- an electrolyte solution is held in a gel matrix.
- the hard case cell becomes unnecessary.
- the degree of freedom in shape can be increased by using a film more flexible than a metal housing or the like as a packaging member. Further reduction in size, weight, and thickness can be realized.
- lithium hexafluorophosphate LiPF 6
- lithium tetrafluoroboric acid LiBF 4
- a problem such as a battery expansion occurs.
- a lithium salt is descomposed and a free acid component such as hydrogen fluoride (HF) or ion fluoride is generated.
- lithium-cobalt complex oxide is used as a positive electrode active material.
- a secondary battery using a non-aqueous gel electrolyte or solid electrolyte housed in a metal foil laminate case has a significant challenge to suppress expansion which is seen in a high temperature storage test or the like since a housing for accommodating the aluminum laminate pack may be broken due to the expansion.
- the positive electrode active material contains from 0.8% to 1.2% of lithium carbonate (Li 2 CO 3 ) so as to provide the function of generating CO 2 gas to shut down a safety valve in the case where the temperature of the battery becomes high when heated or excessively charged.
- Li 2 CO 3 lithium carbonate
- a conventionally used positive electrode active material includes about 500 ppm of water content by which a gas is generated when the battery is heated or excessively charged.
- a non-aqueous gel polymer secondary battery has improved safety against heating and excessive charging, and it is unnecessary to generate a gas when the temperature becomes high.
- the conventional non-aqueous gel polymer secondary battery uses lithium-cobalt complex oxide as a positive electrode active material.
- a non-aqueous gel polymer secondary battery using a metal foil laminate pack obtained by covering metal foil such as aluminum foil with a resin has a significant challenge to suppress expansion, which is seen in a high temperature storage test or the like since there is the possibility that an aluminum laminate pack is not housed in a set case due to the expansion.
- the invention has been achieved in consideration of the problems and its object is to provide a battery capable of suppressing shape change and suppressing deterioration in battery characteristics.
- Another object of the invention is to provide a positive electrode active material capable of suppressing expansion of a battery and a non-aqueous electrolyte secondary battery using the positive electrode active material.
- a non-aqueous electrolyte secondary battery includes a battery device having a positive electrode having a collector, on which a positive electrode active material layer containing a positive electrode material is formed, a negative electrode, and an electrolyte layer, the battery device being sealed in a film-state packaging member, and concentration in mass ratio of a free acid in the electrolyte layer is 60 ppm and less.
- the positive electrode active material is a composite oxide LiCoO 2 .
- the packaging member preferably, a metal foil laminate case or laminated film obtained by coating metal foil with a resin and having the structure of packaging resin layer/metal layer/sealant layer is used.
- a non-aqueous electrolyte secondary battery comprises a positive electrode having a positive electrode collector on which a positive electrode active material layer containing a positive electrode material is formed, a negative electrode having a negative electrode collector on which a negative electrode active material layer is formed, and a film-state case as a packaging member.
- average particle diameter of the positive electrode active material lies in a range from 10 to 22 ⁇ m.
- the positive electrode active material has minimum particle diameter of 5 ⁇ m or larger, maximum particle diameter of 50 ⁇ m and less, and specific surface area of 0.25 m 2 /g and less.
- the positive electrode active material is LiCoO 2 .
- a non-aqueous electrolyte secondary battery comprises a positive electrode having a positive electrode collector, on which a positive electrode active material layer containing a positive electrode material is formed, a negative electrode having a negative electrode collector on which a negative electrode active material layer is formed, and a film-state case as a packaging member.
- the positive electrode active material layer contains 0.15 percent by weight of carbonate compound.
- moisture contained in the positive electrode active material is 300 ppm and less.
- the positive electrode active material layer is made of a lithium and a transition metal complex oxide LiMO 2 (where, M is at least one material selected from Co, Ni, and Mn. More preferably, the positive electrode active material layer is made of LiCoO 2 , and the carbonate contained in the positive electrode active material is LiCoO 3 .
- the concentration in mass ratio of a free acid in the electrolyte is 60 ppm and less, even when the film-state packaging member is used, a change in shape is suppressed, and deterioration in battery characteristics is suppressed.
- the average particle diameter of the positive electrode active material lines in a range from 10 to 22 ⁇ m since the average particle diameter of the positive electrode active material lines in a range from 10 to 22 ⁇ m, the specific surface area of the positive electrode active material becomes narrow, a reaction area decreases and, as a result, generation of gas when the battery is stored at high temperature, is suppressed.
- the carbonate contained in the positive electrode active material is 0.15 percent by weight and less, decomposing reaction when the battery is stored at high temperature is suppressed, and generation of gas is suppressed.
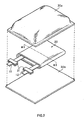
- Fig. 1 shows the appearance of a secondary battery according to an embodiment of the invention.
- Fig. 2 is an exploded view of the secondary battery shown in Fig. 1 .
- a battery device 20 to which a positive electrode lead 11 and a negative electrode lead 12 are attached is sealed in a film-state packaging member 30 (film-state case such as a laminated film in which metal foil such as aluminum foil and the like is coated with a resin, a polymer film, or a metal film).
- film-state packaging member 30 film-state case such as a laminated film in which metal foil such as aluminum foil and the like is coated with a resin, a polymer film, or a metal film.
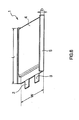
- Fig. 3 is a cross section taken along a III-III line of a sectional structure of the battery device 20 in Fig. 2 .
- the battery device 20 is a obtained by laminating a positive electrode 21 and a negative electrode 22 with, for example, a gel-state electrolyte layer 23 and a separator 24 in-between and rolling the laminated body.
- the outermost peripheral portion of the negative electrode 22 is protected by a protection tape 25.
- the positive electrode 21 has, for example, a positive electrode collector layer 21a and a positive electrode mixture layer 21b which is provided on one side or both sides of the positive electrode collector layer 21a.
- the positive electrode mixture layer 21b is not provided at one of the ends in the longitudinal direction of the positive electrode collector layer 21a, and there is a portion where the positive electrode collector layer 21a is exposed.
- the positive electrode lead 11 is attached to the exposed portion.
- the positive electrode collector layer 21a is made of metal foil such as aluminum (Al) foil, nickel (Ni) foil, or stainless steel foil.
- the positive electrode mixture layer 21b contains, for example, a positive electrode material, a conducting agent such as carbon black or graphite, and a binder such as polyvinylidene fluoride.
- Preferable positive electrode materials are, for example, a metallic oxide, a metallic sulfide, and a specific polymer material. One or more of the materials is/are selected according the application of the battery. To increase energy density, the most preferable material is a lithium composite oxide containing LixMO2 as a main component.
- M denotes one or more kinds of transition metal(s).
- At least one of cobalt (Co), nickel (Ni), and manganese (Mn) is preferable.
- the value (x) varies according to a charge/discharge state of the battery and usually satisfies 0.05 ⁇ x ⁇ 1.12.
- Examples of such lithium composite oxide are LiNiyCo1-yO 2 (where, 0 ⁇ y ⁇ 1) and LiMn 2 O 4 .
- the negative electrode 22 has, for example, in a manner similar to the positive electrode 21, a negative electrode collector layer 22a and a negative a positive electrode mixture layer 21b which is provided on one side or both sides of the negative electrode collector layer 22a.
- the negative electrode mixture layer 22b is not provided at one of the ends in the longitudinal direction of the negative electrode collector layer 22a, and there is a portion where the negative electrode collector layer 22a is exposed.
- the negative electrode lead 12 is attached to the exposed portion.
- the negative electrode collector layer 22a is made of metal foil such as copper (Cu) foil, nickel foil, or stainless steel foil.
- the negative electrode mixture layer 22b contains, for example, one or more of negative electrode materials capable of occluding and releasing a lithium metal or lithium.
- Negative electrode materials capable of occluding and releasing lithium are metals and semiconductors each can form an alloy or compound with lithium, and alloys and compounds of the metals and semiconductors.
- Each of the metals, alloys, and compounds is expressed by the chemical formula DsEtLiu.
- D denotes at least one of a metal element and a semiconductor element capable of forming an alloy or compound with lithium
- E denotes at least one of a metal element and a semiconductor element other than lithium and D.
- Each of values s, t, and u satisfies s > 0, t ⁇ 0, and u ⁇ 0.
- the metal or semiconductor elements each can form an alloy or compound with lithium
- Group 4B metal and semiconductor elements are preferable. More preferable elements are silicon and tin, and the most preferable element is silicon. Alloys and compounds of those elements are also preferable. Examples of the alloys and compounds are SiB 4 , SiB 6 , Mg 2 Si, Mg 2 Sn, Ni 2 Si, TiSi 2 , MoSi 2 , CoSi 2 , NiSi 2 , CaSi 2 , CrSi 2 , CusSi, FeSi 2 , MnSi 2 , NbSi 2 , TaSi 2 , VSi 2 , WSi 2 , and ZnSi 2 .
- negative electrode materials capable of occluding and releasing lithium are carbonaceous materials, metal oxides and polymer materials.
- the carbonaceous materials are pyrocarbons, cokes, graphites, glassy carbons, polymer organic compound calcined materials, carbon fiber, and activated carbon.
- the cokes include pitch coke, needle coke, and petroleum coke.
- the polymeric compound calcined material is a material obtained by calcining a polymeric material such as phynolic resin or furan resin at an appropriate temperature so as to be carbonated.
- a metal oxide tin oxide (SiO 2 ) or the like can be mentioned.
- the polymeric materials are polyacetylene, and polypyrrole.
- the electrolyte layer 23 is composed by, for example, a lithium salt, a non-aqueous solvent for dissolving the lithium salt, and a polymer material.
- a lithium salt a non-aqueous solvent for dissolving the lithium salt
- a polymer material e.g., polyethylene glycol, polypropylene glycol, polyethylene glycol, polypropylene glycol, polyethylene glycol, polypropylene glycol, poly(ethylene glycol), ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, ethylene glycol, hexane, LiN-(CF 3 SO 2 ) 2 N, LiC 4 F 9 SO 3 , LiCl and LiBr. Two or more of them may be mixed.
- non-aqueous solvents are, for example, ethylene carbonate, propylene carbonate, butylene carbonate, vinylene carbonate, ⁇ -butyrolactone, ⁇ -valerolactone, diethoxyethan, tetraphydrofuran, 2-methyltetrahydrofuran, 1,3-dioxolane, methyl acetate, methyl propionic acid, dimethyl carbonate, diethyl carbonate, methyl ethyl carbonate, 2,4-difluoroanisole, 2,6-difluoroanisole, and 4-bromoveratrole. Two or more kinds of the above materials may be mixed.
- any of the materials boiling at 150 °C or higher such as ethylene carbonate, propylene carbonate, butylene carbonate, ⁇ -butyrolactone, 2,4-difluoroanisole, 2,6-diffuoroanisole, and 4-bromoveratrole is used.
- the packaging member 30 is expanded and the outer shape deteriorates.
- Appropriate polymer materials are, for example, fluoride-contained polymers such as polyvinylidene fluoride and poly(vinylidene fluoride-co-hexafluoropropylene), ether-contained polymers such as polyacrylonitrile, acrylonitrile-butadiene rubber, acrylonitrile-butadien-styren resin, acrylonitrile polyethylene chloride diene styrene resin, acrylonitrile vinyl chloride resin, acrylonitrile methacrylate resin, acrylonitrile acrylate resin, and polyethylen oxide, and crosslinkers of the ethyl-contained polymers, and polyethyl modified siloxane.
- fluoride-contained polymers such as polyvinylidene fluoride and poly(vinylidene fluoride-co-hexafluoropropylene)
- ether-contained polymers such as polyacrylonitrile, acrylonit
- Two or more materials may be mixed. Copolymers with the following materials may be also used; acrylonitrile, vinyl acetate, methyl methacrylate, butyl methacrylate, methyl acrylate, butyl acrylate, itaconic acid, methyl acrylate hydroxide, ethyl acrylate hydroxide, acryl amide, vinyl chloride, and vinylidene fluoride. Further, copolymers with ethylene oxide, propylene oxide, methyl methacrylate, butyl methacrylate, methyl acrylate, and butyl acrylate may be also used.
- a copolymer of vinylidene fluoride and hexafluoropropylene, and a copolymer of ethyl modified siloxane may be used. More preferably, it is made by a micro porous polyolefin film.
- the separator 24 is made by, for example, a porous film made of a polyolefin-based material such as polypropylene or polyethylene or a porous film made of an inorganic material such as ceramic nonwoven cloth.
- a structure in which two or more kinds of porous films are stacked, may be also used. More preferably, it is made by a micro porous polyolefin film.
- the positive electrode lead 11 and the negative electrode lead 12 are led from the inside of the packaging member 30 to the outside, for example, in the same direction.
- a part of the positive electrode lead 11 is connected to an exposed portion in the positive electrode collector layer 21a in the packaging member 30.
- a part of the negative electrode lead 12 is connected to an exposed portion of the negative electrode collector layer 22a in the packaging member 30.
- the positive electrode lead 11 and the negative electrode lead 12 are made of a metal material such as aluminum, copper, nickel, or stainless steel in a thin film or mesh state.
- the packaging member 30 is constructed by, for example, two rectangular films 30a and 30b each having a thickness of about 90 ⁇ m to 110 ⁇ m.
- the peripheral portions of the films 30a and 30b adheres to each other by fusion or by using an adhesive.
- the packaging member 30 (films 30a and 30b) takes the form of a laminated film obtained by coating metal foil such as aluminum foil with a resin, the following materials can be used.
- Plastic materials to be used will be abbreviated as follows: polyethylene terephthalate:PET, fused polypropylane:PP, casting polypropylene:CPP, polyethylene:PE, low-density polyethylene:LDPE, high-density polyethylene:HDPE, linear low-density polyethylene:LLDPE, and nylon:Ny.
- Aluminum as a metal material used for a permeability-resistant barrier film is abbreviated as AL. SUS foil may be used in the same way.
- packaging layer/metal layer/sealant layer PET/AL/PE.
- packaging layer/metal film/sealant layer Ny/AL/CPP, PET/AL/CPP, PET/AL/PET/CPP, PET/Ny/AL/CPP, PET/Ny/AL/Ny/CPP, PET/Ny/AL/Ny/PE, Ny/PE/AL/LLDPE, PET/PE/AL/PET/LDPE, and PET/Ny/AL/LDPE/CPP.
- the metal film can be also made of any of metals other than AL.
- a laminated film in which, for example, a nylon film, aluminum foil, and a polyethylene film are laminated in this order, is used.
- the polyethylene film faces the battery device 20.
- the aluminum foil in the laminated film has moisture resistance for preventing intrusion of outside air.
- a laminated film having the other structure, a polymer film made of polypropylene or the like, or a metal film can be also used as the packaging member 30.
- the positive electrode lead 11, the negative electrode lead 12, and the packaging member 30 closely adheres to each other with, for example, a film 31 in-between so as to prevent intrusion of the outside air.
- the film 31 is made of a material which adheres to the positive electrode lead 11 and the negative electrode lead 12.
- the film 31 is made of a polyolefin resin such as polyethylene, polypropylene, modified polyethylene, or modified polypropylene.
- a non-aqueous electrolyte secondary battery having such a structure can be manufactured as follows.
- a positive electrode mixture is prepared by mixing a positive electrode material, a conducting agent, and a binder.
- the positive electrode mixture is dispersed in a solvent of N-methyl-pyrrolidone or the like to thereby obtain a positive electrode mixture slurry.
- the positive electrode mixture slurry is applied on one side or both sides of the positive electrode collector layer 21a, dried, and compression molded, thereby forming the positive electrode mixture layer 21b. In such a manner, the positive electrode 21 is fabricated.
- the positive electrode mixture is not applied to one end of the positive electrode collector layer 21a but the end is exposed.
- a negative electrode mixture is prepared by mixing a negative electrode material capable of occluding and releasing lithium with a binder and dispersing the mixture in a solvent of N-methyl-pyrrolidone or the like to thereby obtain a negative electrode mixture slurry.
- the negative electrode mixture slurry is applied on one side or both sides of the negative electrode collector layer 22a, dried, and compression molded, thereby forming the negative electrode mixture layer 21b. In such a manner, the negative electrode 21 is fabricated.
- the negative electrode mixture is not applied to one end of the negative electrode collector layer 22a but the end is exposed.
- the positive electrode lead 11 is attached to the exposed portion of the positive electrode collector layer 21a by resistance welding, ultrasonic welding, or the like, and the electrolyte is, for example, applied on the positive electrode mixture layer 21b to form the electrolyte layer 23.
- the negative electrode lead 12 is attached to the exposed portion of the negative electrode collector layer 22a by electric resistance welding, ultrasonic welding, or the like, and the electrolyte is, for example, applied on the negative electrode mixture layer 22b to form the electrolyte layer 23.
- the separator 24, the positive electrode 21 on which the electrolyte layer 23 is formed, the separator 24, and the negative electrode 22 on which the electrolyte layer 23 is formed are sequentially laminated and a laminated product is rolled, and the outermost portion is, for example, adhered by the protection tape 25. In such a manner, the battery device 20 is formed.
- the materials (that is, the mixture of the lithium salt, non-aqueous solvent, and polymer material) of the electrolyte stored in a dry atmosphere are heated to about 70 °C to be polymerized. While maintaining the temperature, the resultant is applied on the positive electrode mixture layer 21b or the negative electrode mixture layer 22b, thereby preventing generation of a free acid.
- the films 30a and 30b are prepared to sandwich the battery device 20 and are contact bonded to the battery device 20 in a reduced pressure atmosphere, and the outer peripheral portions of the films 30a and 30b are bonded to each other by thermal fusion bonding or the like.
- Films 31 are disposed so as to sandwich the positive electrode lead 11 and the negative electrode lead 12 at the end portions of the films 30a and 30b from which the positive electrode lead 11 and the negative electrode lead 12 are led, and the peripheries of the films 30a and 30b are bonded to each other via the film 31. In such a manner, the battery shown in Figs. 1 to 3 is completed.
- the secondary battery acts as follows.
- the secondary battery When the secondary battery is charged, for example, lithium ions are released from the positive electrode 21 and occluded by the negative electrode 22 via the electrolyte layer 23.
- the secondary battery When the secondary battery is discharged, for example, the lithium ions are released from the negative electrode 22 and occluded by the positive electrode 21 with the electrolyte layer 23 in-between. Since the concentration in mass ratio of a free acid in the electrolyte layer 23 is 60 ppm and less, the battery is prevented from being expanded. The reaction between the free acid and the lithium in the battery system is suppressed, so that excellent battery characteristics are attained.
- the concentration in mass ratio of the free acid in the electrolyte layer 23, that is, electrolyte is 60 ppm and less.
- the free acid denotes an acid generated when the lithium salt is decomposed, and ions generated when the acid is dissociated.
- the free acid is generated due to decomposition of the lithium salt, for example, when moisture exists or when the electrolyte is heated.
- hydrogen fluoride, fluoride ion, hydrogen chloride (HCl), chloride ion (Cl - ), hydrogen bromide (HBr), bromide ion (Br - ), and the like can be mentioned.
- the concentration in mass ratio of the free acid in the electrolyte is suppressed to 60 ppm and less.
- generation of a gaseous hydride in the battery and generation of gas by corrosion reaction in the battery can be suppressed. Consequently, a change in shape due to expansion can be prevented with the film-state packaging member 30. In the case where the battery is stored in high-temperature environment, the shape can be maintained.
- the consumption of lithium due to the reaction between the free acid and the lithium in the battery system can be also suppressed, and an increase in the internal resistance due to generation of lithium fluoride can be prevented.
- various battery characteristics such as capacity characteristic, shelf stability, and charge/discharge cycle can be prevented from deterioration.
- a positive electrode is fabricated by forming a positive electrode active material layer on a positive electrode collector. While heating the positive electrode to a temperature exceeding room temperature, a gel layer containing electrolyte is formed on the positive electrode active material layer of the positive electrode.
- the gel layer containing electrolyte may be applied on one side or on each of both sides by a single-side coater. Specifically, the electrode unwound from the wound role is heated by an electrode preheater. On the electrode active material layer on one side of the electrode, a composition for forming the gel layer containing electrolyte is applied from the coater head. The applied composition for forming the gel layer containing electrolyte is dried when passed through a dryer and becomes a gel layer containing electrolyte. The electrode on which the gel layer containing electrolyte is formed, is taken up by the wound role.
- the gel layer containing electrolyte can be also simultaneously coated on both sides by a double-side coater.
- the electrode unwound from the wound role is heated by the electrode preheater, and a composition for forming the gel layer containing electrolyte is applied from the coater head simultaneously on both sides of the electrode active material layer.
- the applied composition for forming the gel layer containing electrolyte is dried when passed through a dryer and becomes a gel layer containing electrolyte.
- the electrode on which the gel layer containing electrolyte is formed, is taken up by the wound role.
- the electrode When pressing is necessary, for example, after forming the electrode active material layer and before forming the gel layer containing the electrolyte, the electrode can be pressed by a general press roller.
- the negative electrode is fabricated. Subsequently, while heating the negative electrode to a temperature exceeding the room temperature, a gel layer containing electrolyte solution is formed on a negative electrode active layer of the negative electrode.
- the gel layer containing electrolyte on the positive electrode side and that on the negative electrode side adheres to each other, thereby obtaining an electrode body.
- the obtained electrode body may be assembled to thereby achieving a completed battery by any of methods such as; a method of forming a slit in the electrode on which the gel layer containing electrolyte is formed and assembling the electrode; a method of forming a slit in the electrode first, forming the gel layer containing the electrolyte solution, and assembling the electrode; and a method as a combination of the methods, of forming the gel layer containing the electrolyte solution and forming a slit in one of the electrodes, forming a slit and then forming the gel layer containing the electrolyte solution on the other electrode, and assembling the electrodes.
- methods such as; a method of forming a slit in the electrode on which the gel layer containing electrolyte is formed and assembling the electrode; a method of forming a slit in the electrode first, forming the gel layer containing the electrolyte solution, and assembling the electrode; and a method as a combination of the methods,
- a method of forming the gel layer containing electrolyte only one side of an electrode, forming a slit, forming the gel layer on the other face of the electrode, and assembling the electrode may be also used.
- the electrodes are laminated so that the active material layers of the electrodes face each other.
- the electrodes may be laminated by stacking electrodes which are cut in a desired size, winding stacked electrodes, and the like.
- the battery device fabricated in such a manner is sandwiched by the laminated films, the resultant is pressed to increase the adhesion of the gel layers containing electrolyte on both electrodes and is sealed, so that the battery device is not exposed to outside air.
- a non-aqueous gel polymer secondary battery using the aluminum laminate pack as shown in Fig. 1 is obtained.
- the invention is not limited to the method of preheating the electrode before coating the composition for forming the gel layer containing electrolyte in the invention.
- a method of passing a temperature-controlled roll, a method of blowing temperature-controlled air, a method of providing an infrared ray lamp, or the like can be mentioned.
- a copolymer of vinylidene fluoride and hexafluoropropylene as polymer materials was dissolved in a solvent obtained by mixing propylene carbonate and ethylene carbonate, and further, LiPF 6 was dissolved as a lithium salt.
- the mixing ratio in volume of the solvent and the polymeter material, specifically, propylene carbonate : ethylene carbonate : copolymer was set to 4 : 4 : 1. LiPF 6 was dissolved at the rate of 0.74 mol/dm 3 .
- the mixture solution was stored in a drying chamber for one week or longer and heated to about 70 °C so as to be gelled. In such a manner, electrolytes of the Examples 1-1 to 1-31 were obtained. The electrolytes of the Examples 1-1 to 1-31 were fabricated separately under the same conditions.
- the concentration of the free acid (hydrogen fluoride in this case) of the obtained electrolyte was measured.
- the electrolyte is dissolved in cold water of 1.5 °C or lower so as not to be hydrolyzed.
- acid-bace neutrization titration was carried out by using a sodium hydroxide (NaOH) solution of 0.01 mol/dm 3 , thereby measuring the concentration.
- Fig. 4 the vertical axis denotes concentration (unit: ppm) in mass ratio of the free acid, and the lateral axis denotes numbers of the example and comparative examples which will be described hereinlater.
- the concentration in mass ratio of the free acid in each of the electrolytes of the examples is 60 ppm and less.
- Comparative Examples 1-1 to 1-29 of the present examples electrolytes were fabricated in a manner similar to the examples except that the storage time in the dying chamber was set to one day and the heating temperature was set to 80 to 90 °C.
- the concentration of the free acid was measured with respect to each of Comparative Examples 1-1 to 1-29 in a manner similar to the examples. The result is also shown in Fig. 4 .
- the concentration at mass ratio of the free acid in each of the electrolytes in Comparative Examples 1-1 to 1-29 was 70 ppm or higher.
- a positive electrode mixture was prepared by mixing lithium-cobalt complex oxide (LiCoO 2 ) as a positive electrode material, graphite as a conducting agent, and polyvinylidene fluoride as a binder.
- the positive electrode mixture was dispersed in N-methyl-pyrrolidone as a solvent to thereby obtain a positive electrode mixture slurry.
- the positive electrode mixture slurry was applied on both sides of the positive electrode collector layer 21a made of aluminum foil, dried, and compression molded, thereby forming the positive electrode mixture layer 21b. In such a manner, the positive electrode 21 was fabricated.
- a negative electrode mixture was prepared by mixing graphite powders as a negative electrode material with polyvinylidene fluoride as a binder, the mixture was dispersed in a solvent of N-methyl-pyrrolidone to thereby obtain a negative electrode mixture slurry.
- the negative electrode mixture slurry was applied on both sides of the negative electrode collector layer 22a made of copper foil, dried, and compression molded, thereby forming the negative electrode mixture layer 22b. In such a manner, the negative electrode 22 was fabricated.
- the positive electrode lead 11 was attached to the positive electrode collector layer 21a and the electrolyte was applied on the positive electrode mixture layer 21b to form the electrolyte layer 23.
- the negative electrode lead 12 was attached to the negative electrode collector layer 22a and the electrolyte was applied on the negative electrode mixture layer 22b to form the electrolyte layer 23.
- a porous polypropylene film as the separator 24 was prepared, and the separator 24, the positive electrode 21, the separator 24, and the negative electrode 22 were sequentially laminated and a laminated product was rolled. The outermost portion was adhered by the protection tape 25. In such a manner, the battery device 20 was formed.
- the battery device 20 After forming the battery device 20, two metal foil laminated films each obtained by laminating a nylon film, aluminum foil, and a polyethylene film in this order were prepared, and the battery device 20 was sandwiched by the metal foil laminated films so that the film 31 for improving the adhesion was disposed at the end portions from which the positive electrode lead 11 and the negative electrode lead 12 were led. After that, the laminated films were contact bonded to the battery device 20, and the peripheries of the metal foil laminated films were fusion bonded to each other, thereby obtaining a secondary battery having a length of 62 mm, a width of 35 mm, and a thickness of about 3.8 mm.
- Each of the secondary batteries of examples and comparative examples were repeatedly charged and discharged, a change in shape after the charging was examined, and an initial discharge capacity was measured.
- the charging was performed with a constant current of 250 mA until the battery voltage reaches 4.2V and then by a constant voltage of 4.2V until the total charging time reached nine hours.
- the discharging was performed with a constant current of 250 mA until the battery voltage reaches 3V.
- Fig. 5 shows the results of the initial discharge capacity.
- the vertical axis denotes discharge capacity (unit; mAh), and the vertical axis denotes numbers of the examples and the comparative examples.
- the discharge capacity larger than 565 mAh was obtained in each of Examples 1-1 to 1-31.
- the discharge capacity smaller than 535 mAh was obtained in each of Comparative Examples 1-1 to 1-20.
- the average value of the examples is 586 mAh and that of the comparative examples is 512 mAh.
- the discharge capacity larger than that in the comparative examples by 14.5% was obtained. Variations in the values in the examples are smaller than those in the comparative examples. Thus, stable results were derived.
- each of the secondary batteries in the examples and the comparative examples was charged and discharged for 100 cycles.
- the ratio of the discharge capacity in the 100 th cycle to that in the 1 st cycle (that is, the capacity sustain ratio in the 100 th cycle) was calculated.
- the average value of the capacity sustain ratio of Examples 1-1 to 1-31 is 95%.
- the average value of the capacity sustain ratio of the comparative examples is 87%, which is lower than the average value of the examples.
- the electrolyte in fabrication of the electrolyte, after sufficiently drying the lithium salt, solvent, and polymer material, the electrolyte is gelled at low temperature of about 70 °C, the concentration of the free acid in the electrolyte can be suppressed to 60 ppm or lower at the mass ratio, a change in shape of the battery can be effectively prevented, and stable and excellent capacity characteristics and charge/discharge cycle characteristics can be obtained.
- Examples 2-1 to 2-3 secondary batteries were fabricated in a manner similar to Examples 1-1 to 1-31 except that the concentration in mass ratio of the free acid in the electrolyte was changed as shown in Table 1.
- Comparative Examples 2-1 to 2-3 of Examples 2-1 to 2-3 secondary batteries were fabricated in a manner similar to the examples except that the concentration of the free acid in the electrolyte as shown in Table 1.
- Example 2-3 60 571 93 hardly occurs Comparative Example 2-1 100 511 89 expanded Comparative Example 2-2 200 494 84 expanded Comparative Example 2-3 400 481 81 expanded
- Example 2-1 The concentration of the free acid in the electrolyte in each of Examples 2-1 to 2-3 and Comparative Examples 2-1 to 2-3 was controlled by adjusting the drying time in the drying chamber and the gelling temperature. Specifically, in Example 2-1, the drying time was set as one week and the gelling temperature was set as 70 °C. In Example 2-2, the drying time was set as 5 days, and the gelling temperature was set as 70 °C. In Example 2-3, the drying time was set as 5 days, and the gelling temperature was set as 75 °C. In Comparative Example 2-1, the drying time was set as one day, and the gelling temperature was set as 80 °C to 90 °C. In Comparative Example 2-2, there is no drying time, and the gelling temperature was set as 85 °C to 95 °C. In Comparative Example 2-3, there is no drying time, and the gelling temperature was set as 95 °C to 105 °C.
- composition of a positive electrode active material layer was mixed by a disper for four hours and was coated in a pattern on both sides of aluminum foil having a thickness of 20 ⁇ m.
- the coating pattern includes a coated portion having a length of 160 mm and an uncoated portion having a length of 30 mm, which are repeatedly provided on both sides. The start and end positions of coating on both sides were controlled to coincide with each other.
- Composition of positive electrode active material layer parts by weight LiCoO 2 100 polyvinylidene fluoride 5 (average molecular weight: 300,000) carbon black (average particle diameter: 15 nm) 10 N-methyl-2-pyrrolidone 100
- LiCoO 2 has, as shown in Table 2, average particle diameter of 10 ⁇ m, the minimum particle diameter of 5 ⁇ m, the maximum particle diameter of 18 ⁇ m, and specific surface area of 0.25 m2/g.
- Table 2 Particle size distribution and specific surface area of positive electrode active material average particle diameter (50% particle diameter) ⁇ m minimum particle diameter (5% particle diameter) ⁇ m minimum particle diameter (95% particle diameter) ⁇ m Specific surface area m 2 /g
- the row film of which both sides are coated with the positive electrode was pressed with linear pressure of 300 kg/cm. After the press, the thickness of the positive electrode was 100 ⁇ m and the density of the positive electrode active material layer was 3.45 g/cc.
- composition of a positive electrode active material layer was mixed by a disper for four hours and was coated in a pattern on both sides of copper foil having a thickness of 10 ⁇ m.
- the coating pattern includes a coated portion having a length of 160 mm and an uncoated portion having a length of 30 mm, which are repeatedly provided on both sides. The start and end positions of coating on both sides were controlled to coincide with each other.
- the row sheet of which both sides are coated with the negative electrode was pressed with linear pressure of 300 kg/cm. After the press, the thickness of the negative electrode was 90 ⁇ m and the density of the negative electrode active material layer was 1.30 g/cc.
- the composition for forming the gel layer containing the electrolyte solution was mixed by a disper for one hour in a heated state at 70 °C and was coated in a pattern on the negative electrode active material layers on both sides of the negative electrode collector so as to have a thickness of 20 ⁇ m and was coated in a pattern on the positive electrode collector active material layers on both sides of the positive electrode collector so as to have a thickness of 20 ⁇ m.
- a dryer was controlled so that only dimethyl carbonate evaporates substantially.
- the positive and negative electrodes were heated by setting an electrode preheater at a predetermined temperature 60 °C.
- the leads were welded to both the positive and negative electrodes, and the positive and negative electrodes were adhered to each other so that their electrode active material layers were in contact with each other and contact-bonded.
- the resultant was sent to an assembling section where the battery device was formed.
- the battery device was sandwiched so as to be covered with the laminated films.
- the non-aqueous gel polymer secondary battery as shown in Fig. 6 was fabricated.
- the non-aqueous gel polymer secondary battery of the embodiment used an aluminum laminate pack.
- the laminated film was obtained by stacking nylon, aluminum, and casting polypropylene (CPP) in accordance with the order from the outside.
- the thickness of nylon was 30 ⁇ m
- that of aluminum was 40 ⁇ m
- that of CPP was 30 ⁇ m.
- the thickness of the whole stack layers was 100 ⁇ m.
- LiCoO 2 as the positive electrode active material has, as shown in Table 2, average particle diameter of 16 ⁇ m, minimum particle diameter of 7 ⁇ m, maximum particle diameter of 40 ⁇ m, and specific surface area of 0.23 m 2 /g.
- LiCoO 2 as the positive electrode active material has, as shown in Table 2, average particle diameter of 22 ⁇ m, minimum particle diameter of 9 ⁇ m, maximum particle diameter of 50 ⁇ m, and specific surface area of 0.21 m 2 /g.
- LiCoO 2 as the positive electrode active material has, as shown in Table 2, average particle diameter of 6 ⁇ m, minimum particle diameter of 3 ⁇ m, maximum particle diameter of 12 ⁇ m, and specific surface area of 0.51 m 2 /g.
- LiCoO 2 as the positive electrode active material has, as shown in Table 2, average particle diameter of 8 ⁇ m, minimum particle diameter of 5 ⁇ m, maximum particle diameter of 16 ⁇ m, and specific surface area of 0.38 m 2 /g.
- the expansion ratio of a battery was measured as follows. A plurality of batteries of the examples and the comparative examples were prepared. Each battery was charged under the conditions of 4.2V, 500 mA, and two hours and thirty minutes, and the thickness of the battery was measured. After that, the batteries were stored under the conditions of constant temperature and fixed period such that the batteries were stored at 23 °C for one month, 35 °C for one month, 45 °C for one month, 60 °C for one month, and 90 °C for four hours. The thickness of each of the batteries one hour after the end of storage. A variation in thickness before and after the storage is used as an expansion amount.
- the expansion ratio is defined as follows.
- Expansion ratio % expansion amount / thickness of a battery before storage ⁇ 100
- the following method of measuring the thickness of a battery was used. Specifically, the battery was placed on a stand having a horizontal plane. A disc which is parallel to the plane and is larger than the surface portion of a battery was lowered to the battery. The thickness of the battery was measured in a state where a load of 300g was applied to the disc. When the surface portion of the battery was not a flat face, the highest part of the surface portion of the battery was used to measure thickness.
- each of the batteries was charged with constant current and constant voltage of hour rate of 5 (0.2C) for 15 hours to the upper limit of 4.2V and discharged with constant current of 0.2C, and the discharge was finished at the final voltage of 2.5V.
- the discharge capacity was determined in such a manner and was set as 100%.
- the batteries were stored under the conditions of 23 °C for one month, 35 °C for one month, 45 °C for one month, 60 °C for one month, and 90 °C for four hours.
- the batteries were discharged under the above-described discharging conditions.
- the charging and discharging was repeated five more times.
- the discharge capacity at the fifth time was measured and is displayed in % so as to be compared with the discharge capacity of 100%.
- the capacity of 100% in each of Examples 3 to 5 and Comparative Examples 3 and 4, that is, the capacity before storage was almost equal to each other.
- Example 3 has the expansion ratio ranging from 0 to 5% and proves itself excellent.
- Example 4 has the expansion ratio ranging from 0 to 3% and proves itself excellent.
- Example 5 has the expansion ratio ranging from 0 to 2% and proves itself excellent.
- Comparative Example 3 has a high expansion ratio of 10 to 25% except for the condition of 23 °C for one month.
- Comparative Example 4 has a high expansion ratio of 9 to 20% except for the condition of 23 °C for one month.
- the positive electrode active material used for Examples 3 to 5 produces an excellent result with respect to the expansion ratio.
- the average particle diameter of the positive electrode active material lies in a range from 10 to 22 ⁇ m.
- the positive electrode active material has the minimum particle diameter of 5 ⁇ m, the maximum particle diameter of 50 ⁇ m, and the specific surface area of 0.25 m2/g and less.
- the positive electrode active materials in Examples 3 to 5 obtain excellent results with respect to the expansion ratio for the following reason.
- the cause of expansion of a battery when the battery is stored at high temperature is regarded as generation of gas.
- the cause of the generation of gas is considered that, due to contact between the surface of the positive electrode active material with the electrolyte solution, reaction occurs on the surface, and cracked gas of CO 2 or hydrocarbon generates. Since the surface area of the positive electrode active material of each of Examples 3 to 5 is smaller than that of the positive electrode active material of each of Comparative Examples 3 and 4, it is presumed that due to the small surface area for reaction, decomposition reaction is suppressed. As a result, the expansion of the battery based on the cracked gas is suppressed.
- Example 3 has the capacity sustain ratio ranging from 94 to 98% and proves itself excellent.
- Example 4 has the capacity sustain ratio ranging from 96 to 98% and proves itself excellent.
- Example 5 has the capacity sustain ratio ranging from 97 to 98% and proves itself excellent.
- Comparative Example 3 has a capacity sustain ratio of 90 to 95% which is lower as compared with Examples 3 to 5.
- Comparative Example 4 has a capacity sustain ratio of 92 to 96% which is lower as compared with Examples 1 to 3.
- the positive electrode active material used for Examples 3 to 5 produces an excellent result with respect to the capacity sustain ratio after storage.
- the average particle diameter of the positive electrode active material lies in a range from 10 to 22 ⁇ m.
- the positive electrode active material has the minimum particle diameter of 5 ⁇ m, the maximum particle diameter of 50 ⁇ m, and the specific surface area of 0.25 m2/g and less.
- the positive electrode active materials in Examples 3 to 5 obtain excellent results with respect to the capacity sustain ratio for the following reason. Since the surface area of the positive electrode active material of each of Examples 3 to 5 is smaller than that of the positive electrode active material of each of Comparative Examples 3 and 4, it is presumed that due to the small surface area for reaction, decomposition reaction is suppressed. Due to reduction in the area for reaction, speed of deterioration in the positive electrode active material is also suppressed.
- the expansion which occurs when a non-aqueous gel or solid electrolyte polymer secondary battery using a metal foil case laminated electrical insulator material is stored at high temperature and is a conspicuous problem in the secondary battery can be suppressed.
- the discharge capacity sustain ratio can be improved.
- the positive electrode active material is a composite oxide made of Li and other metal, and the average particle diameter of the positive electrode active material lies within the range from 10 to 22 ⁇ m, the specific surface area is reduced, the reaction area is decreased and, as a result, the generation of gas is suppressed when the battery is stored at high temperature.
- the expansion of a battery which occurs when the battery is stored at a high temperature can be suppressed.
- the discharge capacity sustain ratio can be improved.
- composition of the positive electrode active material layer was mixed by a disper for four hours and was coated in a pattern on both sides of aluminum foil having a thickness of 20 ⁇ m.
- the coating pattern includes a coated portion having a length of 160 mm and an uncoated portion having a length of 30 mm, which are repeatedly provided on both sides. The start and end positions of coating on both sides were controlled to coincide with each other.
- the above-described positive electrode active material LiCoO 2 contains one part by weight of lithium carbonate (Li 2 CO 3 ).
- the positive electrode active material LiCoO 2 contains 400 ppm of moisture.
- the moisture in the positive electrode active material LiCoO 2 was reduced to 400 ppm by drying the positive electrode active material LiCoO 2 in vacuum and controlling the drying time.
- Quantitative analysis of the moisture was conducted as follows. 0.5g of the sample of the positive electrode active material was extracted and heated at 250 °C to vaporize the moisture, and the content of moisture was measured by a Karl Fischer measuring apparatus.
- Quantitative analysis of the content of lithium carbonate was made as follows. 2.0 g of the positive electrode active material was extracted, and analyzed by using the A.G.K. CO 2 analysis method (titration method described in JISR9101).
- the moisture is also contained in other materials such as the negative electrode material, gel, electrolyte, the moisture contained in each of them is very little.
- the moisture existing in a battery can be therefore determined by controlling the moisture contained in the positive electrode active material.
- the row sheet of which both sides were coated with the positive electrode was pressed with linear pressure of 300 kg/cm. After the press, the thickness of the positive electrode was 100 ⁇ m and the density of the positive electrode active material layer was 3.45 g/cc.
- composition of the negative electrode active material layer was mixed by a disper for four hours and was coated in a pattern on both sides of copper foil having a thickness of 10 ⁇ m.
- the coating pattern includes a coated portion having a length of 160 mm and an uncoated portion having a length of 30 mm, which are repeatedly provided on both sides. The start and end positions of coating on both sides were controlled to coincide with each other.
- the row sheet of which both sides are coated with the negative electrode was pressed with linear pressure of 300 kg/cm. After the press, the thickness of the negative electrode was 90 ⁇ m and the density of the negative electrode active material layer was 1.30 g/cc.
- the composition for forming the gel layer containing the electrolyte solution was mixed by a disper for one hour in a heated state at 70 °C and was coated in a pattern on the negative electrode active material layers on both sides of the negative electrode collector so as to have a thickness of 20 ⁇ m and was coated in a pattern on the positive electrode active material layers on both sides of the positive electrode collector so as to have a thickness of 20 ⁇ m.
- a dryer was controlled so that only dimethyl carbonate evaporates substantially.
- the positive and negative electrodes were heated by setting an electrode preheater at a predetermined temperature of 60 °C.
- the row negative electrode on which the gel layer containing the electrolyte solution was formed was cut into 40 mm width to fabricate a negative electrode band.
- the row positive electrode was cut into 38 mm width to fabricate a band-shaped positive electrode body.
- the leads were welded to both the positive and negative electrodes, and the positive and negative electrodes were adhered to each other so that their electrode active material layers were in contact with each other and contact-bonded.
- the resultant was sent to an assembling section where the battery device was formed.
- the battery device was sandwiched so as to be covered with the metal laminated films.
- the non-aqueous gel polymer secondary battery as shown in Fig. 6 was fabricated.
- the non-aqueous gel polymer secondary battery of the embodiment uses an aluminum laminate case.
- the metal laminated film was obtained by stacking nylon, aluminum, and casting polypropylene (CPP) in accordance with the order from the outside.
- the thickness of nylon is 30 ⁇ m
- that of aluminum is 40 ⁇ m
- that of CPP is 30 ⁇ m.
- the thickness of the whole stack layers is 100 ⁇ m.
- Examples 7 to 21 are similar to Example 1 except for the contents of the lithium carbonate and moisture in the positive electrode active material.
- the content of lithium carbonate in each of Examples 7 to 9 is 1 part by weight.
- the contents of moisture of Examples 7 to 9 are 300 ppm, 200 ppm, and 100 ppm, respectively.
- the content of lithium carbonate in each of Examples 10 to 13 is 0.15 percent by weight.
- the contents of moisture of Examples 10 to 13 are 400 ppm, 300 ppm, 200 ppm, and 100 ppm, respectively.
- the content of lithium carbonate of each of Examples 14 to 17 is 0.07 percent by weight.
- the contents of moisture of Examples 14 to 17 are 400 ppm, 300 ppm, 200 ppm, and 100 ppm, respectively.
- the content of lithium carbonate of each of Examples 18 to 21 is 0.01 percent by weight.
- the contents of moisture of Examples 18 to 21 are 400 ppm, 300 ppm, 200 ppm, and 100 ppm, respectively.
- Example 6 to 21 fabricated as described above were evaluated. Evaluation item is an expansion ratio.
- the expansion ratio will now be described.
- the expansion ratio was measured as follows. First, each of the batteries in the examples was charged under the conditions of 4.2V, 500 mA, and two hours and thirty minutes, and the thickness of the battery was measured. After that, the batteries were stored at 90 °C for four hours. The thickness of each of the batteries one hour after the end of storage was measured. A variation in thickness before and after the storage is used as an expansion amount.
- the following method of measuring the thickness of a battery is used. Specifically, the battery is placed on a stand having a horizontal plane. A disc which is parallel to the plane and is larger than the surface portion of a battery is lowered to the battery. The thickness of the battery was measured in a state where a load of 300g was applied to the disc. When the surface portion of the battery is not a flat face, the highest part of the surface portion of the battery is used to measure thickness.
- the expansion ratio of the battery can be suppressed to 4% or lower.
- the reason that the expansion ratio decreases is considered as follows.
- the lithium carbonate is contained in the positive electrode active material
- the lithium carbonate is decomposed by heat when the battery is stored at high temperature and carbon dioxide is resulted.
- an electrolyte such as LiPF 6
- HF electrolyte
- the decomposition of lithium carbonate is promoted, and carbon dioxide is generated.
- the generation of carbon dioxide is considered as a cause of the expansion of a battery.
- the contents of lithium carbonate and moisture as a cause of generation of carbon dioxide are reduced. Consequently, it is presumed that occurrence of carbon dioxide is suppressed, and the expansion of a battery is accordingly suppressed.
- the positive electrode active material is a composite oxide of Li and a transient metal, and carbonate contained in the positive electrode active material is equal to or lower than 0.15 percent by weight. Consequently, decomposition reaction when the battery is stored at high temperature is suppressed. Thus, expansion of the battery when the battery is stored at high temperature can be suppressed.
- the present invention is not limited to the embodiments and the examples but can be variously modified.
- the secondary batteries each using a gel electrolyte containing lithium salt, a non-aqueous solvent, and a polymer material has been described in the embodiments and examples, in place of the gel electrolyte, other electrolytes such as a liquid electrolyte obtained by dissolving a lithium salt into a solvent, a solid electrolyte obtained by dispersing lithium salt into polyethylene glycol or a polymer compound having ion conductivity such as acrylic polymer compound may be used.
- the two films 30a and 30b are used as the packaging member 30 and the battery device 20 is sealed in the two films 30a and 30b. It is also possible to fold a single film, closely adhere the peripheries of the film, and seal the battery device 20 in the folded film.
- the secondary batteries have been described as specific examples in the foregoing embodiments and examples.
- the present invention can be also applied to batteries of other shapes as long as a film-state packaging member is used.
- the non-aqueous secondary batteries have been described in the foregoing embodiments and examples, the present invention can be also applied to other batteries such as primary battery.
- the concentration in mass ratio of a free acid in a non-aqueous electrolyte is suppressed to 60 ppm. Consequently, generation of a gaseous hydride in a battery and generation of a gas due to corrosion reaction in the battery can be suppressed.
- effects such that a change in shape due to expansion can be prevented and the shape can be maintained even when the battery is stored in a high-temperature environment.
- the second aspect of the invention produces effects such that, since the positive electrode active material is a complex oxide of Li and transition metal, and the average particle diameter of the positive electrode active material lies in the range from 10 to 22 ⁇ m, the expansion of the battery when the battery is stored at high temperature can be suppressed.
- the discharge capacity sustain ratio can be also improved.
- the third aspect of the invention produces effects such that, since the positive electrode active material is a complex oxide of Li and transition metal, and carbonate compound contained in the positive electrode active material is 0.15 percent by weight. Thus, expansion of a non-aqueous electrolyte battery which occurs when the battery is stored at high temperature can be suppressed.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Dispersion Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Physics & Mathematics (AREA)
- Secondary Cells (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Sealing Battery Cases Or Jackets (AREA)
- Primary Cells (AREA)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP16178916.9A EP3101715A1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit wasserfreiem elektrolyt |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2000102624A JP4106644B2 (ja) | 2000-04-04 | 2000-04-04 | 電池およびその製造方法 |
| JP2000108412A JP2001291517A (ja) | 2000-04-04 | 2000-04-10 | 正極活物質および非水電解質二次電池 |
| JP2000111044A JP4062856B2 (ja) | 2000-04-04 | 2000-04-12 | 正極活物質及び非水電解質二次電池 |
| EP01108177A EP1143549A3 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP01108177A Division EP1143549A3 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16178916.9A Division EP3101715A1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit wasserfreiem elektrolyt |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP2849264A1 true EP2849264A1 (de) | 2015-03-18 |
Family
ID=27342987
Family Applications (6)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16178916.9A Withdrawn EP3101715A1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit wasserfreiem elektrolyt |
| EP01108177A Ceased EP1143549A3 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP05025373A Pending EP1675208A3 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP10009818A Expired - Lifetime EP2264813B1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP05025372A Revoked EP1672730B1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP14003948.8A Withdrawn EP2849264A1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigem Elektrolyten |
Family Applications Before (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP16178916.9A Withdrawn EP3101715A1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit wasserfreiem elektrolyt |
| EP01108177A Ceased EP1143549A3 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP05025373A Pending EP1675208A3 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP10009818A Expired - Lifetime EP2264813B1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
| EP05025372A Revoked EP1672730B1 (de) | 2000-04-04 | 2001-03-30 | Sekundärbatterie mit nichtwässrigen Elektrolyten |
Country Status (6)
| Country | Link |
|---|---|
| US (4) | US6967066B2 (de) |
| EP (6) | EP3101715A1 (de) |
| JP (3) | JP4106644B2 (de) |
| KR (1) | KR100812549B1 (de) |
| CN (3) | CN100359745C (de) |
| TW (1) | TW492210B (de) |
Families Citing this family (98)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4201459B2 (ja) * | 2000-03-31 | 2008-12-24 | 三洋電機株式会社 | 非水電解質二次電池及びその製造方法 |
| WO2003044882A1 (fr) | 2001-11-20 | 2003-05-30 | Tdk Corporation | Materiau actif d'electrode, electrode, element d'accumulateur au lithium-ion, procede de production de materiau actif d'electrode et procede de production d'element d'accumulateur au lithium-ion |
| KR100440933B1 (ko) * | 2002-02-06 | 2004-07-21 | 삼성에스디아이 주식회사 | 전지부와, 이를 채용한 리튬폴리머전지와, 그 제조방법 |
| US7563535B2 (en) * | 2002-02-06 | 2009-07-21 | Sony Corporation | Battery pack with insulating film sheath material and method of producing the same |
| JP3759908B2 (ja) * | 2002-02-19 | 2006-03-29 | 株式会社バーテックススタンダード | 無線通信機における緊急通報装置 |
| US20070264564A1 (en) | 2006-03-16 | 2007-11-15 | Infinite Power Solutions, Inc. | Thin film battery on an integrated circuit or circuit board and method thereof |
| US9793523B2 (en) | 2002-08-09 | 2017-10-17 | Sapurast Research Llc | Electrochemical apparatus with barrier layer protected substrate |
| US8404376B2 (en) | 2002-08-09 | 2013-03-26 | Infinite Power Solutions, Inc. | Metal film encapsulation |
| US8445130B2 (en) | 2002-08-09 | 2013-05-21 | Infinite Power Solutions, Inc. | Hybrid thin-film battery |
| US8431264B2 (en) | 2002-08-09 | 2013-04-30 | Infinite Power Solutions, Inc. | Hybrid thin-film battery |
| US8236443B2 (en) | 2002-08-09 | 2012-08-07 | Infinite Power Solutions, Inc. | Metal film encapsulation |
| US8394522B2 (en) | 2002-08-09 | 2013-03-12 | Infinite Power Solutions, Inc. | Robust metal film encapsulation |
| US8021778B2 (en) | 2002-08-09 | 2011-09-20 | Infinite Power Solutions, Inc. | Electrochemical apparatus with barrier layer protected substrate |
| JP4183472B2 (ja) * | 2002-10-10 | 2008-11-19 | 三洋電機株式会社 | 非水電解質二次電池 |
| DE60312116T2 (de) * | 2002-12-20 | 2007-06-28 | Toyota Jidosha Kabushiki Kaisha, Toyota | Aktives Material für eine positive Elektrode einer nichtwässrigen Sekundärbatterie und Verfahren zu deren Herstellung |
| US7348762B2 (en) * | 2003-05-01 | 2008-03-25 | Sony Corporation | Battery pack and method for producing battery pack |
| US8999566B2 (en) | 2003-05-22 | 2015-04-07 | Samsung Sdi Co., Ltd. | Pouch-type lithium secondary battery and fabrication method thereof |
| KR100496305B1 (ko) | 2003-05-22 | 2005-06-17 | 삼성에스디아이 주식회사 | 파우치형 리튬 이차 전지와 이의 제조 방법 |
| US8728285B2 (en) | 2003-05-23 | 2014-05-20 | Demaray, Llc | Transparent conductive oxides |
| GB2404781A (en) * | 2003-08-08 | 2005-02-09 | Alexander Gilmour | Electrochemical cell having "in-situ" generated component |
| KR101065307B1 (ko) | 2004-01-19 | 2011-09-16 | 삼성에스디아이 주식회사 | 리튬이차전지용 캐소드 활물질 및 이를 이용한 리튬이차전지 |
| JP4794866B2 (ja) * | 2004-04-08 | 2011-10-19 | パナソニック株式会社 | 非水電解質二次電池用正極活物質およびその製造方法ならびにそれを用いた非水電解質二次電池 |
| KR100601519B1 (ko) | 2004-06-22 | 2006-07-19 | 삼성에스디아이 주식회사 | 파우치형 리튬 폴리머 전지 및 그 제조 방법 |
| JP4117573B2 (ja) | 2004-07-21 | 2008-07-16 | ソニー株式会社 | 電池 |
| US8778529B2 (en) * | 2004-11-29 | 2014-07-15 | Samsung Sdi Co., Ltd. | Lithium secondary battery |
| US7959769B2 (en) | 2004-12-08 | 2011-06-14 | Infinite Power Solutions, Inc. | Deposition of LiCoO2 |
| ATE447777T1 (de) | 2004-12-08 | 2009-11-15 | Symmorphix Inc | Abscheidung von licoo2 |
| KR100766721B1 (ko) * | 2005-01-21 | 2007-10-11 | 주식회사 엘지화학 | 우수한 밀봉성의 이차전지 |
| DE102005005553A1 (de) * | 2005-02-07 | 2006-08-10 | Robert Bosch Gmbh | Elektrische Handwerkzeugmaschine |
| US20080032196A1 (en) | 2005-04-13 | 2008-02-07 | Lg Chem, Ltd. | Method of preparing material for lithium secondary battery of high performance |
| US20070298512A1 (en) * | 2005-04-13 | 2007-12-27 | Lg Chem, Ltd. | Material for lithium secondary battery of high performance |
| US20070292761A1 (en) | 2005-04-13 | 2007-12-20 | Lg Chem, Ltd. | Material for lithium secondary battery of high performance |
| KR100686812B1 (ko) | 2005-04-26 | 2007-02-26 | 삼성에스디아이 주식회사 | 이차 전지 |
| JP5181430B2 (ja) * | 2005-05-26 | 2013-04-10 | ソニー株式会社 | 二次電池 |
| CN100466340C (zh) * | 2005-06-17 | 2009-03-04 | 松下电器产业株式会社 | 非水电解质二次电池 |
| JP4867218B2 (ja) * | 2005-07-08 | 2012-02-01 | ソニー株式会社 | リチウムイオン二次電池 |
| EP1946396B1 (de) * | 2005-11-08 | 2018-02-07 | LG Chem, Ltd. | Sekundärbatterie mit verbesserter sicherheit |
| JP4967321B2 (ja) | 2005-11-21 | 2012-07-04 | ソニー株式会社 | リチウムイオン二次電池 |
| KR100906253B1 (ko) * | 2006-05-01 | 2009-07-07 | 주식회사 엘지화학 | 과전류의 인가시 파괴되는 파단부가 형성되어 있는전극단자를 포함하고 있는 이차전지 |
| JP4215078B2 (ja) | 2006-07-21 | 2009-01-28 | ソニー株式会社 | 非水電解質電池用正極及び非水電解質電池 |
| CN101523571A (zh) | 2006-09-29 | 2009-09-02 | 无穷动力解决方案股份有限公司 | 柔性基板上沉积的电池层的掩模和材料限制 |
| US8197781B2 (en) | 2006-11-07 | 2012-06-12 | Infinite Power Solutions, Inc. | Sputtering target of Li3PO4 and method for producing same |
| JP5230108B2 (ja) * | 2007-01-26 | 2013-07-10 | 三洋電機株式会社 | 非水電解質二次電池 |
| JP5188153B2 (ja) * | 2007-11-13 | 2013-04-24 | 三洋電機株式会社 | 非水電解質二次電池用正極活物質の評価方法及び非水電解質二次電池用正極の評価方法 |
| KR101349942B1 (ko) * | 2007-11-23 | 2014-01-15 | 삼성에스디아이 주식회사 | 리튬 이차 전지용 고분자 전해질 및 이를 포함하는 리튬이차 전지 |
| US8268488B2 (en) | 2007-12-21 | 2012-09-18 | Infinite Power Solutions, Inc. | Thin film electrolyte for thin film batteries |
| US9334557B2 (en) | 2007-12-21 | 2016-05-10 | Sapurast Research Llc | Method for sputter targets for electrolyte films |
| WO2009089417A1 (en) | 2008-01-11 | 2009-07-16 | Infinite Power Solutions, Inc. | Thin film encapsulation for thin film batteries and other devices |
| DE102008015965A1 (de) * | 2008-03-20 | 2009-09-24 | Varta Microbattery Gmbh | Galvanisches Element mit Foliendichtung |
| CN101983469B (zh) | 2008-04-02 | 2014-06-04 | 无穷动力解决方案股份有限公司 | 与能量采集关联的储能装置的无源过电压/欠电压控制和保护 |
| JP2012500610A (ja) | 2008-08-11 | 2012-01-05 | インフィニット パワー ソリューションズ, インコーポレイテッド | 電磁エネルギー獲得ための統合コレクタ表面を有するエネルギーデバイスおよびその方法 |
| WO2010030743A1 (en) | 2008-09-12 | 2010-03-18 | Infinite Power Solutions, Inc. | Energy device with integral conductive surface for data communication via electromagnetic energy and method thereof |
| JP5509561B2 (ja) * | 2008-09-17 | 2014-06-04 | 日産自動車株式会社 | 非水電解質二次電池 |
| JP5353151B2 (ja) * | 2008-09-25 | 2013-11-27 | 住友金属鉱山株式会社 | リチウムニッケル含有複合酸化物の製造方法 |
| WO2010042594A1 (en) | 2008-10-08 | 2010-04-15 | Infinite Power Solutions, Inc. | Environmentally-powered wireless sensor module |
| KR101094937B1 (ko) | 2009-02-16 | 2011-12-15 | 삼성에스디아이 주식회사 | 원통형 이차전지 |
| JP5195499B2 (ja) * | 2009-02-17 | 2013-05-08 | ソニー株式会社 | 非水電解質二次電池 |
| JP4835742B2 (ja) * | 2009-02-20 | 2011-12-14 | ソニー株式会社 | 電池および電池パック |
| EP2237346B1 (de) | 2009-04-01 | 2017-08-09 | The Swatch Group Research and Development Ltd. | Elektrisch leitfähiges Nanoverbundmaterial mit Opfer-Nanopartikeln sowie daraus hergestellte offen poröse Nanoverbundstoffe |
| EP2228855B1 (de) | 2009-03-12 | 2014-02-26 | Belenos Clean Power Holding AG | Offen poröses elektrisch leitfähiges Nanoverbundmaterial |
| JP5487676B2 (ja) * | 2009-03-30 | 2014-05-07 | Tdk株式会社 | 活物質、これを含む電極、当該電極及びリチウム塩を含む電解質溶液を備える電気化学デバイス |
| EP2287946A1 (de) * | 2009-07-22 | 2011-02-23 | Belenos Clean Power Holding AG | Neue Elektrodenmaterialien, insbesondere für wiederaufladbare Lithiumionen-Batterien |
| CN102576828B (zh) | 2009-09-01 | 2016-04-20 | 萨普拉斯特研究有限责任公司 | 具有集成薄膜电池的印刷电路板 |
| KR20110066448A (ko) * | 2009-12-11 | 2011-06-17 | 삼성에스디아이 주식회사 | 리튬 이차전지 |
| JP2011138623A (ja) * | 2009-12-25 | 2011-07-14 | Sanyo Electric Co Ltd | 非水系二次電池 |
| JP5329453B2 (ja) * | 2010-01-28 | 2013-10-30 | 三洋電機株式会社 | 非水系二次電池 |
| JP5329454B2 (ja) * | 2010-01-28 | 2013-10-30 | 三洋電機株式会社 | リチウムイオンポリマー電池 |
| EP2577777B1 (de) | 2010-06-07 | 2016-12-28 | Sapurast Research LLC | Wiederaufladbare elektrochemische vorrichtung von hoher dichte |
| WO2012020939A2 (ko) * | 2010-08-09 | 2012-02-16 | 주식회사 엘지화학 | 안전성이 향상된 이차전지 |
| US9413013B2 (en) * | 2010-12-28 | 2016-08-09 | Panasonic Intellectual Property Management Co., Ltd. | Non-aqueous electrolyte secondary battery and method for producing the same |
| JP5987336B2 (ja) * | 2011-03-25 | 2016-09-07 | 日本電気株式会社 | 二次電池 |
| JP6093516B2 (ja) * | 2011-09-30 | 2017-03-08 | 株式会社日本触媒 | 電解液及びその製造方法、並びに、これを用いた蓄電デバイス |
| CN104170153A (zh) * | 2012-03-02 | 2014-11-26 | 日本电气株式会社 | 二次电池 |
| JP5781219B2 (ja) * | 2012-03-15 | 2015-09-16 | 株式会社東芝 | 非水電解質二次電池および電池パック |
| JP5621869B2 (ja) * | 2012-03-27 | 2014-11-12 | Tdk株式会社 | リチウムイオン二次電池 |
| KR101511022B1 (ko) * | 2012-04-16 | 2015-04-10 | 주식회사 엘지화학 | 수분 제한 전극 활물질, 수분 제한 전극 및 이를 포함하는 리튬 이차전지 |
| WO2013172133A1 (ja) * | 2012-05-14 | 2013-11-21 | Necエナジーデバイス株式会社 | 二次電池用正極電極、二次電池、及びそれらの製造方法 |
| TWI665134B (zh) | 2013-01-25 | 2019-07-11 | 凸版印刷股份有限公司 | Flexible package |
| JP6076926B2 (ja) | 2013-03-25 | 2017-02-08 | 株式会社東芝 | 電池用活物質、非水電解質電池、電池パック及び自動車 |
| JP6283171B2 (ja) * | 2013-03-29 | 2018-02-21 | デンカ株式会社 | リチウムイオン二次電池用カーボンブラックおよびその用途 |
| JP6223760B2 (ja) | 2013-09-18 | 2017-11-01 | 株式会社東芝 | 非水電解質二次電池用活物質、非水電解質二次電池、電池パック及び車両 |
| JP6329888B2 (ja) | 2013-12-13 | 2018-05-23 | エルジー・ケム・リミテッド | 二次電池用負極材及びこれを用いた二次電池 |
| KR101762508B1 (ko) * | 2014-10-02 | 2017-07-27 | 주식회사 엘지화학 | 리튬 이차전지용 양극활물질, 이의 제조방법 및 이를 포함하는 리튬 이차전지 |
| JP2016177901A (ja) * | 2015-03-18 | 2016-10-06 | 株式会社東芝 | 二次電池用正極活物質、二次電池、及び、二次電池用正極活物質の製造方法 |
| JP6580886B2 (ja) | 2015-06-30 | 2019-09-25 | 株式会社エンビジョンAescジャパン | リチウムイオン二次電池 |
| KR102448295B1 (ko) * | 2015-07-16 | 2022-09-28 | 삼성에스디아이 주식회사 | 이차전지 |
| KR102515097B1 (ko) * | 2015-10-26 | 2023-03-27 | 삼성에스디아이 주식회사 | 리튬 이차 전지 |
| KR102479486B1 (ko) * | 2015-10-28 | 2022-12-19 | 삼성에스디아이 주식회사 | 파우치형 이차 전지 |
| JP6642471B2 (ja) * | 2017-02-02 | 2020-02-05 | トヨタ自動車株式会社 | 複合活物質粒子、正極、全固体リチウムイオン電池及びこれらの製造方法 |
| JP6989322B2 (ja) * | 2017-08-30 | 2022-01-05 | 株式会社エンビジョンAescジャパン | リチウムイオン二次電池用正極 |
| CN108680701A (zh) * | 2018-04-04 | 2018-10-19 | 珠海市赛纬电子材料股份有限公司 | 一种六氟磷酸锂电解液中含草酸类无机盐化合物的检测方法 |
| CN208226027U (zh) * | 2018-04-12 | 2018-12-11 | 宁德新能源科技有限公司 | 电芯 |
| CN108922991A (zh) * | 2018-06-26 | 2018-11-30 | 苏州英诺达自动化科技有限公司 | 一种具有环保性能的锂电池 |
| CN110003627B (zh) * | 2019-04-04 | 2021-01-19 | 苏州思达奇环保新材料有限公司 | 可降解固态电解质膜及其制备方法以及由其制成的锂电池 |
| CN114730910B (zh) * | 2020-08-14 | 2023-08-29 | 宁德时代新能源科技股份有限公司 | 二次电池及其制备方法与包含二次电池的电池模块、电池包及装置 |
| US20230317941A1 (en) * | 2020-09-04 | 2023-10-05 | Sanyo Electric Co., Ltd. | Positive electrode active material for nonaqueous electrolyte secondary batteries, and nonaqueous electrolyte secondary battery |
| CN112479271A (zh) * | 2020-12-09 | 2021-03-12 | 山东丰元化学股份有限公司 | 一种高镍三元正极材料制备方法 |
| JP2022111526A (ja) * | 2021-01-20 | 2022-08-01 | Fdk株式会社 | ラミネート型蓄電素子 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0848444A2 (de) * | 1996-12-14 | 1998-06-17 | VARTA Batterie Aktiengesellschaft | Verfahren zur Herstellung von Polymerelektrolyten |
| WO1998026469A1 (en) * | 1996-12-09 | 1998-06-18 | Valence Technology, Inc. | Stabilized electrochemical cell |
| FR2761531A1 (fr) * | 1997-03-25 | 1998-10-02 | Ube Industries | Solution electrolytique pour pile au lithium |
| EP0895296A1 (de) * | 1997-07-23 | 1999-02-03 | Sanyo Electric Co., Ltd. | Dünnschicht verschlossene elektrochemische Zelle mit nichtwässrigem Elektrolyten |
| WO1999034471A1 (fr) * | 1997-12-26 | 1999-07-08 | Tonen Corporation | Electrolyte pour piles au lithium et procede de production dudit electrolyte |
| JPH11185811A (ja) * | 1997-12-16 | 1999-07-09 | Tonen Corp | リチウム電池用電解液及びその製造方法 |
| JPH11283668A (ja) * | 1998-03-30 | 1999-10-15 | Sanyo Electric Co Ltd | リチウムイオン電池 |
| WO1999056336A1 (fr) * | 1998-04-27 | 1999-11-04 | Sony Corporation | Accumulateur electrique a electrolyte solide |
| EP1041658A1 (de) * | 1998-09-01 | 2000-10-04 | Sony Corporation | Verfahren zur herstellung einer elektrolytischen zelle mit nichtwässrigem gel |
Family Cites Families (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4913988A (en) * | 1987-06-11 | 1990-04-03 | Eveready Battery Company, Inc. | Li2 CO3 -Ca(OH)2 additive for cathodes in nonaqueous cells |
| JP2615854B2 (ja) * | 1988-06-01 | 1997-06-04 | ソニー株式会社 | 非水電解液二次電池 |
| JP2934449B2 (ja) * | 1989-03-23 | 1999-08-16 | 株式会社リコー | 二次電池 |
| JP3067165B2 (ja) * | 1990-06-20 | 2000-07-17 | ソニー株式会社 | リチウム2次電池の正極活物質LiCoO2およびその製造方法、並びに、リチウム2次電池 |
| JP3010781B2 (ja) * | 1991-04-26 | 2000-02-21 | ソニー株式会社 | 非水電解質二次電池 |
| US5427875A (en) * | 1991-04-26 | 1995-06-27 | Sony Corporation | Non-aqueous electrolyte secondary cell |
| JP3051506B2 (ja) * | 1991-05-21 | 2000-06-12 | 東芝電池株式会社 | 非水溶媒二次電池 |
| JP3231813B2 (ja) * | 1991-09-13 | 2001-11-26 | 旭化成株式会社 | 有機電解液電池 |
| US5393622A (en) * | 1992-02-07 | 1995-02-28 | Matsushita Electric Industrial Co., Ltd. | Process for production of positive electrode active material |
| JP3726163B2 (ja) * | 1994-10-27 | 2005-12-14 | 宇部興産株式会社 | 非水二次電池とその製造方法 |
| JP3385516B2 (ja) * | 1995-01-18 | 2003-03-10 | 日本電池株式会社 | 非水系ポリマー電池及び非水系ポリマー電池用ポリマー膜の製造方法 |
| JPH0959026A (ja) * | 1995-08-21 | 1997-03-04 | Nikki Kagaku Kk | ニッケル酸リチウムの製造方法 |
| JPH09288996A (ja) * | 1996-04-23 | 1997-11-04 | Sumitomo Electric Ind Ltd | 非水電解質電池 |
| JPH1027613A (ja) * | 1996-07-08 | 1998-01-27 | Sumitomo Metal Mining Co Ltd | 非水系電解質二次電池用正極活物質およびその製造方法 |
| JP3713900B2 (ja) * | 1996-07-19 | 2005-11-09 | ソニー株式会社 | 負極材料及びこれを用いた非水電解液二次電池 |
| JPH1040900A (ja) * | 1996-07-25 | 1998-02-13 | Toyota Motor Corp | リチウムイオン二次電池用正極 |
| US5976731A (en) * | 1996-09-03 | 1999-11-02 | Fuji Photo Film Co., Ltd. | Non-aqueous lithium ion secondary battery |
| JP3575582B2 (ja) * | 1996-09-12 | 2004-10-13 | 同和鉱業株式会社 | 非水二次電池用正極活物質とその製造方法 |
| JPH10144315A (ja) * | 1996-09-13 | 1998-05-29 | Sumitomo Metal Mining Co Ltd | 非水系電解質二次電池用正極活物質およびその製造方法 |
| US5928812A (en) * | 1996-11-18 | 1999-07-27 | Ultralife Batteries, Inc. | High performance lithium ion polymer cells and batteries |
| JPH10149832A (ja) * | 1996-11-20 | 1998-06-02 | Sony Corp | 非水電解液二次電池 |
| JPH10162826A (ja) | 1996-11-28 | 1998-06-19 | Sanyo Electric Co Ltd | 非水電解液二次電池 |
| EP0953218B1 (de) * | 1996-12-20 | 2003-03-26 | Danionics A/S | Lithium sekundarbatterie |
| JP3223824B2 (ja) * | 1996-12-26 | 2001-10-29 | 三菱電機株式会社 | リチウムイオン二次電池 |
| JP3562187B2 (ja) * | 1996-12-27 | 2004-09-08 | ソニー株式会社 | 非水電解液二次電池 |
| CN1194278A (zh) * | 1997-01-20 | 1998-09-30 | 松下电器产业株式会社 | 聚合物电解质和使用该电解质的锂-聚合物电池 |
| JPH10255860A (ja) * | 1997-03-13 | 1998-09-25 | Asahi Chem Ind Co Ltd | 非水系電池 |
| JP3706718B2 (ja) * | 1997-04-01 | 2005-10-19 | 日本化学工業株式会社 | リチウムイオン二次電池用コバルト酸リチウム系正極活物質、この製造方法及びリチウムイオン二次電池 |
| JP3907282B2 (ja) * | 1997-04-04 | 2007-04-18 | 昭和電工株式会社 | 複合電解質及びその用途 |
| JP3769871B2 (ja) * | 1997-04-25 | 2006-04-26 | ソニー株式会社 | 正極活物質の製造方法 |
| US6127065A (en) * | 1997-04-25 | 2000-10-03 | Sony Corporation | Method of manufacturing cathode active material and nonaqueous electrolyte secondary battery |
| CN1120533C (zh) | 1997-05-27 | 2003-09-03 | Tdk株式会社 | 非水电解质电池用电极 |
| JP3817034B2 (ja) | 1997-07-09 | 2006-08-30 | 株式会社スズテック | 育苗箱並列敷設装置 |
| JPH1186907A (ja) | 1997-09-05 | 1999-03-30 | Asahi Chem Ind Co Ltd | 非水系二次電池 |
| JPH11111266A (ja) | 1997-09-30 | 1999-04-23 | Yuasa Corp | 高分子電解質二次電池 |
| JPH11135119A (ja) | 1997-10-27 | 1999-05-21 | Matsushita Electric Ind Co Ltd | 非水電解液二次電池用活物質と正極板および非水電解液二次電池 |
| US6195251B1 (en) * | 1997-10-29 | 2001-02-27 | Asahi Glass Company Ltd. | Electrode assembly and electric double layer capacitor having the electrode assembly |
| JP4071342B2 (ja) * | 1998-02-16 | 2008-04-02 | 富士通株式会社 | リチウム二次電池及びそれに用いる正極合剤 |
| JPH11260346A (ja) * | 1998-03-06 | 1999-09-24 | Hitachi Maxell Ltd | ポリマー電解質電池 |
| JPH11260368A (ja) * | 1998-03-13 | 1999-09-24 | Mitsubishi Cable Ind Ltd | リチウム二次電池用の正極活物質 |
| JP4127892B2 (ja) | 1998-03-31 | 2008-07-30 | 日立マクセル株式会社 | リチウムイオン二次電池 |
| KR100274236B1 (ko) * | 1998-05-13 | 2001-02-01 | 김순택 | 리튬 이차 전지용 양극 활물질 및 그의 제조방법 |
| JP3690122B2 (ja) * | 1998-07-03 | 2005-08-31 | 宇部興産株式会社 | 非水二次電池 |
| JP4003298B2 (ja) * | 1998-07-01 | 2007-11-07 | 宇部興産株式会社 | 非水二次電池 |
| JP2000040505A (ja) * | 1998-07-24 | 2000-02-08 | Mitsubishi Cable Ind Ltd | リチウム二次電池用の正極体 |
| JP2000058259A (ja) * | 1998-08-07 | 2000-02-25 | Mitsubishi Electric Corp | 共振器型エレクトロルミネッセンス素子 |
| JP2000067869A (ja) * | 1998-08-26 | 2000-03-03 | Nec Corp | 非水電解液二次電池 |
| CN1089193C (zh) * | 1998-10-09 | 2002-08-14 | 清华大学 | 喷雾干燥法制备锂离子电池活性材料超细粉的方法 |
| JP4193247B2 (ja) * | 1998-10-30 | 2008-12-10 | ソニー株式会社 | 非水電解質電池及びその製造方法 |
| JP2000200605A (ja) * | 1998-10-30 | 2000-07-18 | Sanyo Electric Co Ltd | 非水電解質電池およびその製造方法 |
| TW439309B (en) * | 1999-01-22 | 2001-06-07 | Toshiba Corp | Nonaquous electrolyte secondary battery |
| JP3308232B2 (ja) * | 1999-05-17 | 2002-07-29 | 三菱電線工業株式会社 | Li−Co系複合酸化物およびその製造方法 |
| JP4075259B2 (ja) * | 1999-05-26 | 2008-04-16 | ソニー株式会社 | 固体電解質二次電池 |
| JP2001052747A (ja) * | 1999-08-06 | 2001-02-23 | Matsushita Electric Ind Co Ltd | リチウム二次電池 |
| GB9929698D0 (en) * | 1999-12-15 | 2000-02-09 | Danionics As | Non-aqueous electrochemical cell |
| JP2001273898A (ja) * | 2000-01-20 | 2001-10-05 | Japan Storage Battery Co Ltd | 非水電解質二次電池用正極活物質およびその製造方法並びにそれを使用した非水電解質二次電池 |
-
2000
- 2000-04-04 JP JP2000102624A patent/JP4106644B2/ja not_active Expired - Lifetime
- 2000-04-10 JP JP2000108412A patent/JP2001291517A/ja active Pending
- 2000-04-12 JP JP2000111044A patent/JP4062856B2/ja not_active Expired - Lifetime
-
2001
- 2001-03-30 EP EP16178916.9A patent/EP3101715A1/de not_active Withdrawn
- 2001-03-30 TW TW090107595A patent/TW492210B/zh not_active IP Right Cessation
- 2001-03-30 EP EP01108177A patent/EP1143549A3/de not_active Ceased
- 2001-03-30 EP EP05025373A patent/EP1675208A3/de active Pending
- 2001-03-30 EP EP10009818A patent/EP2264813B1/de not_active Expired - Lifetime
- 2001-03-30 EP EP05025372A patent/EP1672730B1/de not_active Revoked
- 2001-03-30 EP EP14003948.8A patent/EP2849264A1/de not_active Withdrawn
- 2001-04-04 CN CNB011214384A patent/CN100359745C/zh not_active Expired - Lifetime
- 2001-04-04 CN CNB200410056794XA patent/CN100394639C/zh not_active Expired - Lifetime
- 2001-04-04 US US09/825,988 patent/US6967066B2/en not_active Expired - Lifetime
- 2001-04-04 KR KR1020010017881A patent/KR100812549B1/ko active IP Right Grant
- 2001-04-04 CN CNB2004100567954A patent/CN1310369C/zh not_active Expired - Lifetime
-
2005
- 2005-03-07 US US11/073,509 patent/US7563539B2/en not_active Expired - Lifetime
- 2005-03-07 US US11/073,511 patent/US7223495B2/en not_active Expired - Lifetime
- 2005-03-07 US US11/073,510 patent/US20050175891A1/en not_active Abandoned
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998026469A1 (en) * | 1996-12-09 | 1998-06-18 | Valence Technology, Inc. | Stabilized electrochemical cell |
| EP0848444A2 (de) * | 1996-12-14 | 1998-06-17 | VARTA Batterie Aktiengesellschaft | Verfahren zur Herstellung von Polymerelektrolyten |
| FR2761531A1 (fr) * | 1997-03-25 | 1998-10-02 | Ube Industries | Solution electrolytique pour pile au lithium |
| EP0895296A1 (de) * | 1997-07-23 | 1999-02-03 | Sanyo Electric Co., Ltd. | Dünnschicht verschlossene elektrochemische Zelle mit nichtwässrigem Elektrolyten |
| JPH11185811A (ja) * | 1997-12-16 | 1999-07-09 | Tonen Corp | リチウム電池用電解液及びその製造方法 |
| WO1999034471A1 (fr) * | 1997-12-26 | 1999-07-08 | Tonen Corporation | Electrolyte pour piles au lithium et procede de production dudit electrolyte |
| US6383688B1 (en) * | 1997-12-26 | 2002-05-07 | Tonen Corporation | Electrolyte for lithium cells and method of producing the same |
| JPH11283668A (ja) * | 1998-03-30 | 1999-10-15 | Sanyo Electric Co Ltd | リチウムイオン電池 |
| WO1999056336A1 (fr) * | 1998-04-27 | 1999-11-04 | Sony Corporation | Accumulateur electrique a electrolyte solide |
| EP1001476A1 (de) * | 1998-04-27 | 2000-05-17 | Sony Corporation | Festelektrolyt-sekundärbatterie |
| EP1041658A1 (de) * | 1998-09-01 | 2000-10-04 | Sony Corporation | Verfahren zur herstellung einer elektrolytischen zelle mit nichtwässrigem gel |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1310369C (zh) | 2007-04-11 |
| EP1675208A2 (de) | 2006-06-28 |
| JP2001291517A (ja) | 2001-10-19 |
| JP2001283910A (ja) | 2001-10-12 |
| CN1322026A (zh) | 2001-11-14 |
| JP4106644B2 (ja) | 2008-06-25 |
| EP2264813B1 (de) | 2013-01-02 |
| EP3101715A1 (de) | 2016-12-07 |
| EP2264813A1 (de) | 2010-12-22 |
| CN1571211A (zh) | 2005-01-26 |
| TW492210B (en) | 2002-06-21 |
| US6967066B2 (en) | 2005-11-22 |
| CN100359745C (zh) | 2008-01-02 |
| US20050153210A1 (en) | 2005-07-14 |
| KR20010095309A (ko) | 2001-11-03 |
| US20050186472A1 (en) | 2005-08-25 |
| CN1574445A (zh) | 2005-02-02 |
| US20020031703A1 (en) | 2002-03-14 |
| KR100812549B1 (ko) | 2008-03-13 |
| EP2264813A8 (de) | 2011-03-23 |
| US7563539B2 (en) | 2009-07-21 |
| EP1143549A2 (de) | 2001-10-10 |
| EP1143549A3 (de) | 2004-12-29 |
| US20050175891A1 (en) | 2005-08-11 |
| EP1672730A3 (de) | 2007-04-11 |
| JP2001297764A (ja) | 2001-10-26 |
| EP1672730B1 (de) | 2011-10-19 |
| CN100394639C (zh) | 2008-06-11 |
| EP1675208A3 (de) | 2007-04-11 |
| EP1672730A2 (de) | 2006-06-21 |
| US7223495B2 (en) | 2007-05-29 |
| JP4062856B2 (ja) | 2008-03-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1672730B1 (de) | Sekundärbatterie mit nichtwässrigen Elektrolyten | |
| EP1195826B1 (de) | Festelektrolytzelle | |
| KR101032290B1 (ko) | 파우치 및 이를 포함하는 이차전지 | |
| US20100209757A1 (en) | Nonaqueous electrolyte secondary battery | |
| US20060222955A1 (en) | Battery | |
| EP1041658A1 (de) | Verfahren zur herstellung einer elektrolytischen zelle mit nichtwässrigem gel | |
| EP1445806B1 (de) | Elektrische zelle | |
| JP2004227818A (ja) | 非水電解質電池 | |
| KR20100071814A (ko) | 이차전지용 파우치 및 파우치형 이차전지 | |
| JP2000100399A (ja) | ポリマーリチウム二次電池の製造方法 | |
| US20050186481A1 (en) | Lithium-ion secondary battery | |
| JP4815845B2 (ja) | ポリマー電池 | |
| JP2004152619A (ja) | 非水電解質電池 | |
| JP2000277066A (ja) | 非水電解質二次電池 | |
| EP2157639B1 (de) | Aktive Masse einer positiven Elektrode, positive Elektrode, die diese Masse einsetzt, und nicht wässrige Elektrolyt-Sekundärbatterie | |
| JP2005322634A (ja) | 電解質およびそれを用いた電池 | |
| JP2003346786A (ja) | 非水電解質電池及びその製造方法 | |
| JP2002050402A (ja) | 非水電解液二次電池 | |
| KR20240019562A (ko) | 리튬 이차 전지 | |
| JPH11307066A (ja) | 薄形電池 | |
| JP2000277158A (ja) | 非水電解質二次電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20141124 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 1143549 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): DE FI FR GB SE |
|
| 17Q | First examination report despatched |
Effective date: 20160901 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: TOHOKU MURATA MANUFACTURING CO., LTD |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: MURATA MANUFACTURING CO., LTD. |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20180925 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20190206 |