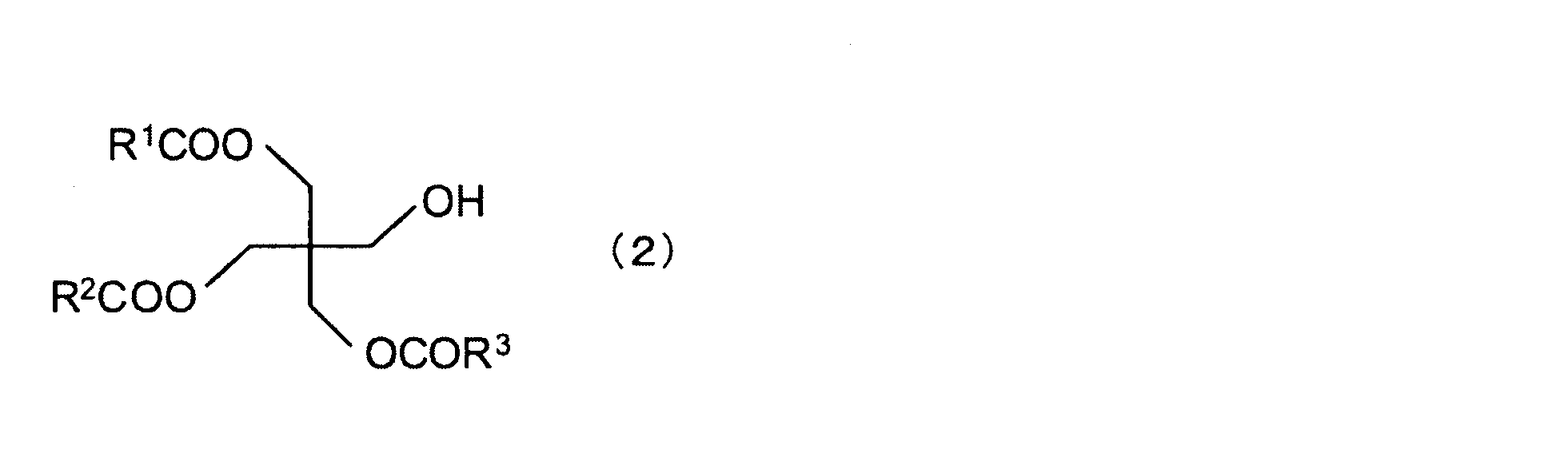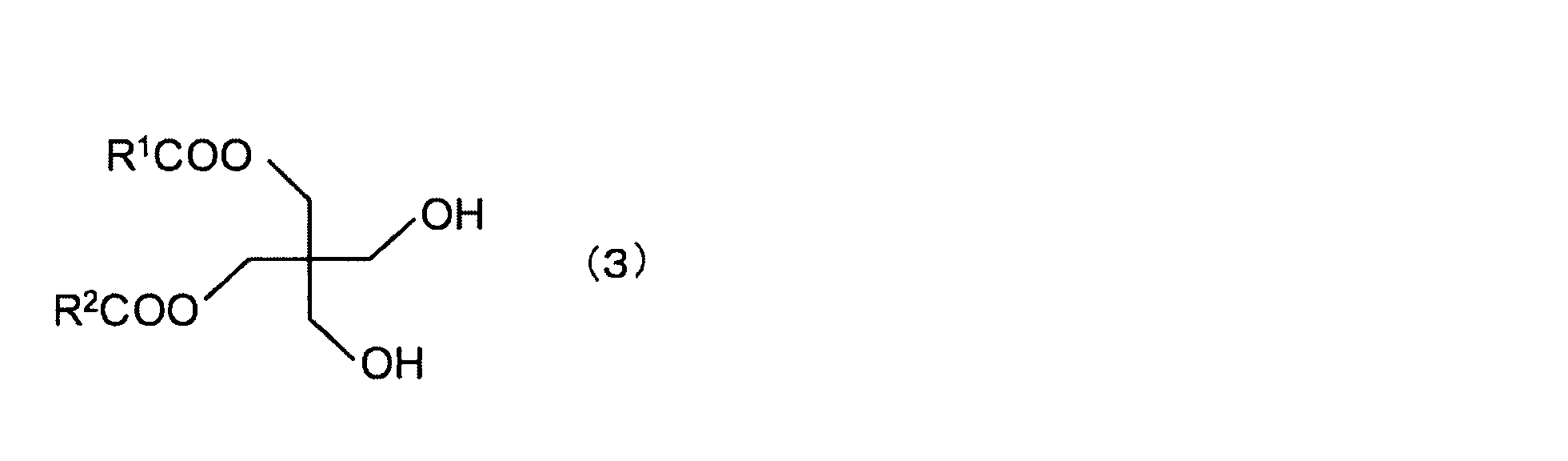WO2013129236A1 - 吸収性物品 - Google Patents
吸収性物品 Download PDFInfo
- Publication number
- WO2013129236A1 WO2013129236A1 PCT/JP2013/054382 JP2013054382W WO2013129236A1 WO 2013129236 A1 WO2013129236 A1 WO 2013129236A1 JP 2013054382 W JP2013054382 W JP 2013054382W WO 2013129236 A1 WO2013129236 A1 WO 2013129236A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- acid
- chain hydrocarbon
- hydrocarbon moiety
- blood
- group
- Prior art date
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/45—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the shape
- A61F13/47—Sanitary towels, incontinence pads or napkins
- A61F13/472—Sanitary towels, incontinence pads or napkins specially adapted for female use
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/45—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the shape
- A61F13/49—Absorbent articles specially adapted to be worn around the waist, e.g. diapers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/51—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the outer layers
- A61F13/511—Topsheet, i.e. the permeable cover or layer facing the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/51—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the outer layers
- A61F13/511—Topsheet, i.e. the permeable cover or layer facing the skin
- A61F13/51104—Topsheet, i.e. the permeable cover or layer facing the skin the top sheet having a three-dimensional cross-section, e.g. corrugations, embossments, recesses or projections
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/51—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the outer layers
- A61F13/511—Topsheet, i.e. the permeable cover or layer facing the skin
- A61F13/51113—Topsheet, i.e. the permeable cover or layer facing the skin comprising an additive, e.g. lotion or odour control
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/84—Accessories, not otherwise provided for, for absorbent pads
- A61F13/8405—Additives, e.g. for odour, disinfectant or pH control
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/20—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons containing organic materials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/50—Lubricants; Anti-adhesive agents
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B3/00—Layered products comprising a layer with external or internal discontinuities or unevennesses, or a layer of non-planar form; Layered products having particular features of form
- B32B3/26—Layered products comprising a layer with external or internal discontinuities or unevennesses, or a layer of non-planar form; Layered products having particular features of form characterised by a particular shape of the outline of the cross-section of a continuous layer; characterised by a layer with cavities or internal voids ; characterised by an apertured layer
- B32B3/30—Layered products comprising a layer with external or internal discontinuities or unevennesses, or a layer of non-planar form; Layered products having particular features of form characterised by a particular shape of the outline of the cross-section of a continuous layer; characterised by a layer with cavities or internal voids ; characterised by an apertured layer characterised by a layer formed with recesses or projections, e.g. hollows, grooves, protuberances, ribs
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/02—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by structural features of a fibrous or filamentary layer
- B32B5/022—Non-woven fabric
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/02—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by structural features of a fibrous or filamentary layer
- B32B5/024—Woven fabric
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B5/00—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts
- B32B5/22—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by the presence of two or more layers which are next to each other and are fibrous, filamentary, formed of particles or foamed
- B32B5/24—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by the presence of two or more layers which are next to each other and are fibrous, filamentary, formed of particles or foamed one layer being a fibrous or filamentary layer
- B32B5/26—Layered products characterised by the non- homogeneity or physical structure, i.e. comprising a fibrous, filamentary, particulate or foam layer; Layered products characterised by having a layer differing constitutionally or physically in different parts characterised by the presence of two or more layers which are next to each other and are fibrous, filamentary, formed of particles or foamed one layer being a fibrous or filamentary layer another layer next to it also being fibrous or filamentary
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/84—Accessories, not otherwise provided for, for absorbent pads
- A61F13/8405—Additives, e.g. for odour, disinfectant or pH control
- A61F2013/8455—Additives, e.g. for odour, disinfectant or pH control being lubricants
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2250/00—Layers arrangement
- B32B2250/20—All layers being fibrous or filamentary
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/02—Synthetic macromolecular fibres
- B32B2262/0253—Polyolefin fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/02—Synthetic macromolecular fibres
- B32B2262/0276—Polyester fibres
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2262/00—Composition or structural features of fibres which form a fibrous or filamentary layer or are present as additives
- B32B2262/06—Vegetal fibres
- B32B2262/062—Cellulose fibres, e.g. cotton
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/726—Permeability to liquids, absorption
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/70—Other properties
- B32B2307/73—Hydrophobic
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2555/00—Personal care
- B32B2555/02—Diapers or napkins
Definitions
- This disclosure relates to absorbent articles.
- Absorbent articles such as sanitary napkins, panty liners, etc. have improved basic performance due to many years of technical development, and leaks etc. after absorbing excretion such as menstrual blood compared to before.
- the research is on further enhancement of functionality, for example, having a feeling of wearing close to underwear, and that the topsheet is smooth even after absorbing excretion such as menstrual blood. Has been done.
- menstrual menstrual blood may contain components such as endometrium, and the viscosity thereof is high.
- the top sheet has a sticky feeling. It is preferable that the material is smooth.
- highly viscous menstrual blood often remains in a lump state on the top sheet, and the user often feels visually uncomfortable. It is preferable not to leave it on the top sheet.
- menstrual menstrual discharge is not constant, and depending on the period after menstruation started, there are waves in menstrual discharge, and when a large amount of menstrual blood is discharged at once, There are cases where a small amount of menstrual blood is discharged at once. Even during menstruation, menstrual blood is not always discharged, and there are times when menstrual blood is not discharged.
- menstrual blood when considering from the top sheet side, there is a time when a large amount of menstrual blood reaches at once, a time when a small amount of menstrual blood reaches at once, and a time when menstrual blood does not reach, regardless of the amount of menstrual blood, It is preferable to transfer menstrual blood to the absorbent body without remaining on the top sheet.
- Patent Document 1 discloses a lotion composition containing a polypropylene glycol material, an inner surface of a top sheet (clothing side surface), an inner surface of a back sheet (body side surface), an inner surface of the top sheet, and a back sheet.
- An absorbent article disposed on a base material or the like between the inner surfaces is disclosed.
- Patent Document 2 discloses an absorbent article in which a lotion composition containing a polypropylene glycol material is applied to the outer surface (surface on the body side) of a top sheet.
- Patent Documents 1 and 2 are not designed to transfer menstrual blood to the absorbent without remaining on the top sheet, regardless of the amount of menstrual blood.
- the relationship with the shape of the sheet, particularly the top sheet, is not mentioned. Therefore, the present disclosure provides an absorbent article in which the top sheet is not sticky and the top sheet is smooth not only when absorbing a large amount of menstrual blood but also when absorbing a small amount of menstrual blood. For the purpose.
- liquid-permeable top sheet has a concavo-convex structure including a convex part and a concave part on the skin contact surface
- liquid-permeable article has a kinematic viscosity of 0.01 to 80 mm 2 / s at 40 ° C., a water retention of 0.01 to 4.0% by mass
- An absorbent article characterized by containing a blood slipperiness imparting agent having a weight average molecular weight of less than 000 has been found.
- the absorbent article of the present disclosure is not sticky to the top sheet when it absorbs a large amount of menstrual blood but also a small amount of menstrual blood, and the top sheet is smooth.
- FIG. 1 is a front view of a sanitary napkin which is one embodiment of the absorbent article of the present invention.
- FIG. 2 is a cross-sectional view of a portion A of the sanitary napkin 1 shown in FIG.
- FIG. 3 is an electron micrograph of the skin contact surface of the top sheet in a sanitary napkin in which the top sheet contains tri-C2L oil fatty acid glycerides.
- FIG. 4 is a micrograph of menstrual blood with or without a blood slipping agent.
- FIG. 5 is a diagram for explaining a method of measuring the surface tension.
- the absorbent article of the present disclosure will be described in detail below.
- the liquid-permeable top sheet has a concavo-convex structure including a convex portion and a concave portion on the skin contact surface.
- menstrual blood that has reached the convex portion is transferred to the concave portion and then to the absorbent body, and therefore, between the height of the convex portion and the height of the concave portion. It is preferable that there is a certain difference.
- the height of the convex portion is preferably about 0.1 to about 15.0 mm higher than the height of the concave portion, more preferably about 0.5 to about 5.0 mm higher, and more preferably about 0.5 to about 5.0 mm. 2.0 mm higher.
- the heights of the convex portions and the concave portions can be measured with a laser displacement meter such as a high-precision two-dimensional laser displacement meter LJ-G series (model: LJ-G030) manufactured by Keyence Corporation.
- Examples of the top sheet having a concavo-convex structure include, for example, a liquid-permeable top sheet having a ridge groove structure including a plurality of ridges and a plurality of grooves on the skin contact surface, for example, 7-84597, JP-A-2-229255, JP-A-2001-328191, JP-A-2008-002034, JP-A-2008-023311, JP-A-2008-025078 to JP-A-2008-025085, JP-A-2008.
- the height of the ridge is preferably about 0.1 to about 15.0 mm higher than the height of the groove, and more preferably about 0.5 to about 5.0 mm. High, and more preferably about 0.5 to about 2.0 mm high.
- the pitch of the heel portion is preferably about 1.5 to about 17 mm, more preferably about 2.0 to about 12 mm, and even more preferably about 3 to about 8 mm. This is because menstrual blood is slid down from the convex portion to the concave portion and then quickly transferred to the absorber.
- the liquid-permeable top sheet can have at least a squeezed portion formed by squeezing the liquid-permeable top sheet.
- the liquid-permeable top sheet can have a compressed part formed by pressing a liquid-permeable top sheet and an absorbent body, and the absorbent article of the present disclosure includes a second sheet.
- the liquid permeable top sheet may have a squeezed portion formed by squeezing the liquid permeable top sheet, the second sheet, and the absorber.
- the liquid-permeable top sheet has a kinematic viscosity of about 0.01 to about 80 mm 2 / s at 40 ° C. and about 0.05 to about 4.0% by mass at least on the protrusions.
- a blood slipping agent having a water retention rate of less than about 1,000 and a weight average molecular weight of less than about 1,000.
- the blood slipperiness imparting agent has a kinematic viscosity at 40 ° C. of about 0 to about 80 mm 2 / s, preferably about 1 to about 70 mm 2 / s, preferably about 3 to about 60 mm 2. More preferably, it has a kinematic viscosity of about / s, more preferably about 5 to about 50 mm 2 / s, and even more preferably about 7 to about 45 mm 2 / s.
- the kinematic viscosity is as follows: a) as the molecular weight of the blood slipperiness agent increases, b) polar groups such as carbonyl bond (—CO—), ether bond (—O—), carboxyl group (—COOH), hydroxyl group There is a tendency that the higher the ratio of (—OH) and the like, and c) the higher the IOB, the higher the ratio.
- the melting point of the blood slipperiness imparting agent is preferably 45 ° C. or less. This is because when the blood slipperiness-imparting agent contains crystals at 40 ° C., the kinematic viscosity tends to increase. In this specification, the kinematic viscosity at 40 ° C. may be simply referred to as “kinematic viscosity”.
- the kinematic viscosity in the blood slipperiness-imparting agent is so high that it has reached the skin contact surface of the top sheet. Along with blood, it tends to be difficult to slide down from the convex portion to the concave portion and then move into the absorber.
- the kinematic viscosity can be measured at a test temperature of 40 ° C. using a Cannon-Fenske reverse flow viscometer according to “5. Kinematic viscosity test method” of JIS K 2283: 2000.
- the blood slipperiness imparting agent has a water retention of about 0.01 to about 4.0% by mass, preferably about 0.02 to about 3.5% by mass, More preferably, it has a water content of 0.03 to about 3.0% by weight, more preferably about 0.04 to about 2.5% by weight, and about 0.05 to about 2 More preferably, it has a water retention of 0.0% by mass.
- water retention means the proportion of water that a substance can retain and can be measured as follows. (1) Place a test tube, a rubber stopper, a substance to be measured, and deionized water in a constant temperature room at 40 ° C. overnight. (2) In the temperature-controlled room, put 5.0 g of the substance to be measured and 5.0 g of deionized water into a 20 mL test tube. (3) In the above-mentioned temperature-controlled room, the mouth of the test tube is plugged with a rubber stopper, rotated once, and allowed to stand for 5 minutes.
- the affinity between the blood slipperiness-imparting agent and menstrual blood is reduced, and the skin contact surface of the top sheet is reduced. There is a tendency that it becomes difficult to transfer to the absorber together with the reached menstrual blood.
- the affinity with menstrual blood becomes very high like a surfactant, and the absorbed blood remains on the skin contact surface of the top sheet, and the skin contact of the top sheet is increased. The contact surface tends to be red and easily colored.
- the water retention rate is as follows: a) the molecular weight of the blood slipperiness-imparting agent decreases, and b) polar groups such as carbonyl bond (—CO—), ether bond (—O—), carboxyl group (—COOH), The higher the ratio of hydroxyl groups (—OH) and the like, the larger the value tends to be. This is because the blood slipperiness imparting agent is more hydrophilic. Further, the water retention rate tends to increase as the IOB increases, that is, as the inorganic value increases and as the organic value decreases. This is because the blood slipperiness-imparting agent is more hydrophilic.
- FIG. 1 is a front view of a sanitary napkin that is one embodiment of the absorbent article of the present invention, and is a view observed from the skin contact surface side.
- the sanitary napkin 1 shown in FIG. 1 is the front side on the left side.
- a sanitary napkin 1 shown in FIG. 1 includes a liquid-permeable top sheet 2, an absorbent body 3, and a liquid-impermeable back sheet (not shown).
- the sanitary napkin 1 shown in FIG. 1 also shows a side sheet 4 and a pressing part 5.
- the top sheet has a plurality of ridges and a plurality of grooves extending in the longitudinal direction of the absorbent article on the skin contact surface. Are omitted for convenience. Moreover, in the sanitary napkin 1 shown in FIG. 1, the ridges and the grooves are alternately arranged in the width direction of the absorbent article. Moreover, although the side sheet 4 and the pressing part 5 are shown by the sanitary napkin 1 shown by FIG. 1, in another embodiment of the absorbent article of this indication, an absorbent article is a side sheet and a pressing part. Does not have.
- FIG. 2 is a cross-sectional view of the A portion of the sanitary napkin 1 shown in FIG.
- the sanitary napkin 1 shown in FIG. 2 includes a liquid-permeable top sheet 2, a liquid-impermeable back sheet 6, and absorption between the liquid-permeable top sheet 2 and the liquid-impermeable back sheet 6. And a body 3.
- the top sheet 2 has a convex portion 7 and a concave portion 8 on the skin contact surface 9, and a blood slipperiness imparting agent 10 is applied to the skin contact surface 9 of the top sheet 2.
- the blood slipperiness imparting agent 10 is shown as a droplet on the skin contact surface 9 of the top sheet 2 for convenience.
- the blood slipperiness imparting agent is shown.
- the shape and distribution are not limited to those shown in the drawings.
- the highly viscous menstrual blood 11 that has reached the convex portion 7 of the skin contact surface 9 of the top sheet 2 comes into contact with at least the blood slipperiness imparting agent 10 present on the convex portion 7.
- the blood slipperiness imparting agent 10 having a predetermined water retention rate and kinematic viscosity slides into the recess 8 together with the menstrual blood 11 to become menstrual blood 11 ′, and then the menstrual blood 11 ′ mainly passes through the recess 8, It reaches the absorber 3 to become menstrual blood 11 ′′ and is rapidly absorbed by the absorber 3.
- the blood slipperiness-imparting agent 10 having a kinematic viscosity of about 0.01 to about 80 mm 2 / s at 40 ° C. has a very low viscosity around the wearer's body temperature and is consistent with menstrual blood 11. Since it has affinity, it slides from the convex portion 7 to the concave portion 8 together with the menstrual blood 11, and the menstrual blood 11 'passes through the concave portion 8 of the top sheet 2 by utilizing the momentum at the time of the sliding, and the absorbent body It is thought that it can shift to 3 quickly.
- the blood slipperiness imparting agent 10 present on the convex portion 7 has a water retention of about 0.01 to about 4.0% by mass, it is mainly a hydrophilic component (eg plasma) in the menstrual blood 11. It is considered that menstrual blood 11 is unlikely to remain on the top sheet.
- a hydrophilic component eg plasma
- menstrual blood 11 When menstrual blood 11 is a large amount of menstrual blood, the kinetic energy of menstrual blood 11 itself is large, the kinematic viscosity value of blood slipperiness imparting agent 10 is relatively high, and it is difficult to slide with menstrual blood 11. Even when the water retention value is relatively high and the affinity with the hydrophilic component of menstrual blood 11 is high, even when the weight molecular weight value is relatively high and difficult to slide with menstrual blood 11, And even if there is no uneven structure on the skin contact surface of the top sheet, it is considered that menstrual blood 11 is easily transferred to the absorber 3.
- the menstrual blood 11 when the menstrual blood 11 is a small amount of menstrual blood, the kinetic energy of the menstrual blood 11 tends to be small and the menstrual blood that has reached the skin contact surface 9 of the top sheet 2 tends to stay on the spot. Accordingly, a predetermined blood slipperiness-imparting agent slides from the convex portion 7 to the concave portion 8 together with the menstrual blood 11, and draws the menstrual blood 11 ′ into the top sheet 2 and then into the absorbent body 3, whereby the menstrual blood 11 Blood can be quickly transferred to the absorber.
- the blood slipperiness imparting agent has a weight average molecular weight of less than about 1,000, and preferably has a weight average molecular weight of less than about 900. This is because when the weight average molecular weight is about 1,000 or more, the blood slipperiness-imparting agent itself is tacky and tends to give the wearer discomfort. In addition, since the viscosity of the blood slipperiness-imparting agent tends to increase as the weight average molecular weight increases, it becomes difficult to lower the viscosity of the blood slipperiness-imparting agent to a viscosity suitable for coating by heating, As a result, the blood slipping agent may have to be diluted with a solvent.
- the blood slipperiness-imparting agent preferably has a weight average molecular weight of about 100 or more, and more preferably has a weight average molecular weight of about 200 or more. This is because when the weight average molecular weight is small, the vapor pressure of the blood slipperiness-imparting agent is high and vaporizes during storage, which may cause problems such as a decrease in the amount and odor when worn.
- weight average molecular weight refers to a polydispersed compound (for example, a compound produced by sequential polymerization, a plurality of fatty acids, and an ester produced from a plurality of aliphatic monohydric alcohols).
- a weight average molecular weight means the value of polystyrene conversion calculated
- GPC measurement conditions include the following. Model: Hitachi High-Technologies Corporation High-Performance Liquid Chromatogram Lachrom Elite Column: SHODEX KF-801, KF-803 and KF-804 manufactured by Showa Denko K.K. Eluent: THF Flow rate: 1.0 mL / min Driving amount: 100 ⁇ L Detection: RI (differential refractometer)
- the weight average molecular weight described in the Example of this specification is measured on the said conditions.
- the blood slipperiness agent may have an IOB of about 0.00 to about 0.60.
- IOB Inorganic Organic Balance
- IOB is an index indicating a balance between hydrophilicity and lipophilicity.
- Oda et al. IOB value calculated by inorganic value / organic value.
- the inorganic value and the organic value are represented by Fujita Minoru, “Prediction of organic compounds and conceptual diagram of organic compounds”, chemistry area Vol. 11, no. 10 (1957) p. 719-725).
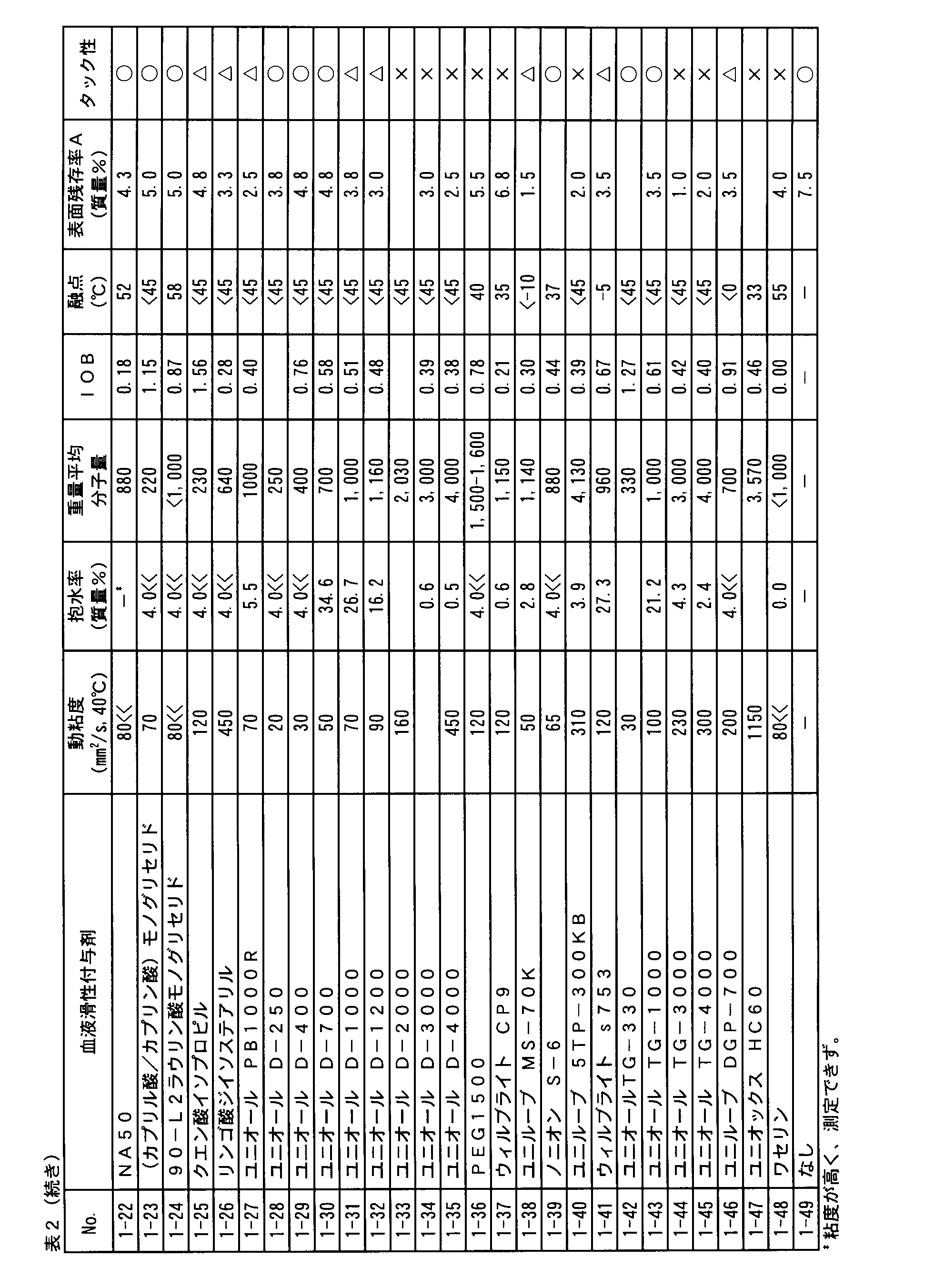
- Table 1 summarizes the organic and inorganic values of the major groups by Mr. Fujita.
- the IOB is preferably about 0.00 to about 0.60, more preferably about 0.00 to about 0.50, and about 0.00 to about 0.00. More preferred is 40, and more preferred is from about 0.00 to about 0.30. This is because when the IOB is in the above range, the water holding power and kinematic viscosity easily satisfy the above requirements.
- the blood slipperiness-imparting agent preferably has a melting point of 45 ° C. or lower. This is because when the blood slipperiness-imparting agent has a melting point of 45 ° C. or less, the blood slipperiness-imparting agent tends to have a kinematic viscosity in the above range.
- melting point means a peak top temperature of an endothermic peak when changing from a solid state to a liquid state when measured with a differential scanning calorimeter at a heating rate of 10 ° C./min.
- the melting point can be measured using, for example, a DSC-60 type DSC measuring apparatus manufactured by Shimadzu Corporation.
- the blood slipperiness-imparting agent may be liquid at room temperature (about 25 ° C.) or solid as long as it has a melting point of about 45 ° C. or lower, that is, even if the melting point is about 25 ° C. or higher. Alternatively, it may be less than about 25 ° C. and may have a melting point of, for example, about ⁇ 5 ° C., about ⁇ 20 ° C., etc.
- the blood slipperiness-imparting agent has a lower melting point, but preferably has a low vapor pressure.
- the blood slipping agent has a vapor pressure of preferably about 0 to about 200 Pa, more preferably about 0 to about 100 Pa, and about 0 to about 10 Pa at 25 ° C. (1 atm). Is more preferably from about 0 to about 1 Pa, and even more preferably from about 0.0 to about 0.1 Pa.
- the vapor pressure is preferably about 0 to about 700 Pa at 40 ° C. (1 atm), and is about 0 to about 100 Pa. And more preferably from about 0 to about 10 Pa, even more preferably from about 0 to about 1 Pa, and even more preferably from about 0.0 to about 0.1 Pa. This is because if the vapor pressure is high, vaporization may occur during storage, and problems such as a decrease in the amount of the blood slipperiness-imparting agent and odor during wearing may occur.
- the melting point of the blood slipperiness-imparting agent can be selected according to the climate, the length of wearing time, and the like. For example, in an area where the average temperature is about 10 ° C. or less, by adopting a blood slipperiness imparting agent having a melting point of about 10 ° C. or less, menstrual blood is excreted and then cooled by the ambient temperature. However, it is considered that the blood slipperiness-imparting agent is easy to function.
- the melting point of the blood slipperiness imparting agent is preferably higher in the range of about 45 ° C. or less. This is because the blood slipperiness-imparting agent is not easily biased even when worn for a long time, and is not easily affected by sweat, friction at the time of wearing.
- the skin contact surface of the top sheet is coated with a surfactant for the purpose of changing the surface tension of menstrual blood and the like and quickly absorbing menstrual blood.
- the top sheet coated with a surfactant has a high affinity with hydrophilic components (such as plasma) in menstrual blood, and tends to attract them and rather cause menstrual blood to remain on the top sheet.
- the blood slipperiness-imparting agent has a low affinity for menstrual blood, and can rapidly transfer menstrual blood to the absorbent body without remaining on the top sheet.
- the blood slipperiness imparting agent is preferably the following (i) to (iii), (I) hydrocarbons, (Ii) from (ii-1) a hydrocarbon moiety and (ii-2) a carbonyl group (—CO—) and an oxy group (—O—) inserted between the CC single bonds of the hydrocarbon moiety.
- the hydrocarbon moiety A compound having one or a plurality of the same or different groups selected from the group consisting of a carboxyl group (—COOH) and a hydroxyl group (—OH), which replaces a hydrogen atom; As well as any combination thereof.
- hydrocarbon means a compound composed of carbon and hydrogen, and a chain hydrocarbon, for example, a paraffinic hydrocarbon (also referred to as an alkane, which does not include a double bond and a triple bond).
- a paraffinic hydrocarbon also referred to as an alkane, which does not include a double bond and a triple bond.
- Olefinic hydrocarbons including one double bond, also referred to as alkene
- acetylenic hydrocarbons including one triple bond, also referred to as alkyne
- hydrocarbons containing two or more bonds selected from the above and cyclic hydrocarbons such as aromatic hydrocarbons and alicyclic hydrocarbons.
- the hydrocarbon is preferably a chain hydrocarbon and an alicyclic hydrocarbon, more preferably a chain hydrocarbon, a paraffinic hydrocarbon, an olefinic hydrocarbon, and two double bonds. More preferred are hydrocarbons (not including triple bonds), and more preferred are paraffinic hydrocarbons.
- the chain hydrocarbon includes a straight chain hydrocarbon and a branched chain hydrocarbon.
- each oxy group (—O—) is not adjacent. Therefore, the compounds (ii) and (iii) do not include compounds having a continuous oxy group (so-called peroxides).
- At least one hydrogen atom in the hydrocarbon moiety is more hydroxyl group (—OH) than in the compound in which at least one hydrogen atom in the hydrocarbon moiety is substituted with a carboxyl group (—COOH).
- a carboxyl group —COOH
- IOB is preferred. This is because the carboxyl group binds to a metal or the like in menstrual blood, increases the water retention rate of the blood slipperiness imparting agent, and may exceed a predetermined range. This is the same from the viewpoint of IOB. As shown in Table 1, the carboxyl group binds to menstrual metals and the like, and the inorganic value increases significantly from 150 to 400 or more. Sometimes the value of IOB can be greater than about 0.60.
- the blood slipperiness imparting agent is more preferably the following (i ′) to (iii ′), (I ′) hydrocarbon, (Ii ′) (ii′-1) a hydrocarbon moiety and (ii′-2) a carbonyl bond (—CO—), an ester bond (—COO) inserted between the C—C single bonds of the hydrocarbon moiety.
- the blood slipperiness imparting agent is more preferably about 1.8 or less carbonyl bonds (—CO—) and 2 or less ester bonds (—COO—) per 10 carbon atoms in the hydrocarbon moiety.
- About 1.5 or less carbonate bonds (—OCOO—), about 6 or less ether bonds (—O—), about 0.8 or less carboxyl groups (—COOH), and / or hydroxyl groups (—OH) ) Can be about 1.2 or less.
- the blood slipperiness imparting agent is more preferably the following (A) to (F), (A) (A1) a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups replacing hydrogen atoms in the chain hydrocarbon moiety, (A2) a chain hydrocarbon moiety, and the chain An ester with a compound having one carboxyl group for substituting a hydrogen atom in the hydrocarbon moiety, (B) (B1) a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups replacing hydrogen atoms in the chain hydrocarbon moiety, (B2) a chain hydrocarbon moiety, and the chain An ether with a compound having one hydroxyl group replacing a hydrogen atom of the hydrocarbon moiety, (C) (C1) a carboxylic acid, a hydroxy acid, an alkoxy acid or an oxo acid containing a chain hydrocarbon moiety and 2 to 4 carboxyl groups replacing a hydrogen atom in the chain hydrocarbon moiety; C2) an ester of a compound having a chain
- (A) (A1) a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups substituting a hydrogen atom of the chain hydrocarbon moiety, (A2) a chain hydrocarbon moiety, Esters with compounds having one carboxyl group replacing a hydrogen atom of a chain hydrocarbon moiety]
- (A) (A1) a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups replacing hydrogen atoms in the chain hydrocarbon moiety;
- (A2) a chain hydrocarbon moiety;
- Ester with a compound having one carboxyl group that substitutes a hydrogen atom of the hydrocarbon-like moiety hereinafter sometimes referred to as “compound (A)” has the above-mentioned kinematic viscosity, water retention and weight average. All hydroxyl groups may not be esterified as long as they have a molecular weight.
- (A1) A compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups replacing hydrogen atoms in the chain hydrocarbon moiety (hereinafter sometimes referred to as “compound (A1)”)
- a chain hydrocarbon tetraol such as an alkanetetraol, such as pentaerythritol
- a chain hydrocarbon triol such as an alkanetriol, such as glycerine
- a chain hydrocarbon diol such as an alkanediol, such as, for example, Glycol.
- Examples of the compound having a chain hydrocarbon moiety and one carboxyl group that replaces a hydrogen atom of the chain hydrocarbon moiety include, for example, one hydrogen atom on a hydrocarbon having one carboxyl group
- Examples include compounds substituted with (—COOH), for example, fatty acids.
- Examples of the compound (A) include (a 1 ) an ester of a chain hydrocarbon tetraol and at least one fatty acid, (a 2 ) an ester of a chain hydrocarbon triol and at least one fatty acid, and (a 3 ) Esters of chain hydrocarbon diols with at least one fatty acid.
- ester of (a 1 ) chain hydrocarbon tetraol and at least one fatty acid examples include the following formula (1): Tetraesters of pentaerythritol and fatty acids of the following formula (2): Triesters of pentaerythritol and fatty acids of the following formula (3): Diester of pentaerythritol and fatty acid of the following formula (4): And monoesters of pentaerythritol and fatty acids. (Wherein R 1 to R 4 are each a chain hydrocarbon)
- the ester of pentaerythritol and fatty acid has the kinematic viscosity, water retention rate and There is no particular limitation as long as the weight average molecular weight requirement is satisfied, but for example, a saturated fatty acid, for example, a C 2 to C 30 saturated fatty acid, for example, acetic acid (C 2 ) (C 2 represents the number of carbon atoms, R 1 C, R 2 C, R 3 C or R 4 C, the same applies hereinafter), propanoic acid (C 3 ), butanoic acid (C 4 ) and isomers thereof such as 2-methylpropane Acids (C 4 ), pentanoic acid (C 5 ) and isomers thereof such as 2-methylbutanoic acid (C 5 ), 2,2-dimethylpropanoic acid (C 5 ),
- the fatty acid can also be an unsaturated fatty acid.
- unsaturated fatty acid include C 3 to C 20 unsaturated fatty acids such as monounsaturated fatty acids such as crotonic acid (C 4 ), myristoleic acid (C 14 ), palmitoleic acid (C 16 ), Oleic acid (C 18 ), elaidic acid (C 18 ), vaccenic acid (C 18 ), gadoleic acid (C 20 ), eicosenoic acid (C 20 ), etc., diunsaturated fatty acids such as linoleic acid (C 18 ), Triunsaturated fatty acids such as eicosadienoic acid (C 20 ), such as linolenic acid, such as ⁇ -linolenic acid (C 18 ) and ⁇ -linolenic acid (C 18 ), pinolenic acid (C 18 ), eleostearic acid, For example, ⁇ -eleostearic acid (C
- the ester of pentaerythritol and fatty acid is an ester of pentaerythritol and a fatty acid derived from a saturated fatty acid, that is, an ester of pentaerythritol and a saturated fatty acid, considering the possibility of modification by oxidation or the like. preferable.
- the ester of pentaerythritol and fatty acid is preferably a diester, triester or tetraester, more preferably a triester or tetraester, from the viewpoint of reducing the value of water retention. More preferably, it is a tetraester.
- the total number of carbon atoms of the fatty acid constituting the tetraester of pentaerythritol and fatty acid that is, in the formula (1), the total number of carbon atoms of the R 1 C, R 2 C, R 3 C and R 4 C moieties is preferably about 15 (when the total number of carbon atoms is 15, the IOB is 0.60).
- tetraester of pentaerythritol and fatty acid for example, pentaerythritol, hexanoic acid (C 6 ), heptanoic acid (C 7 ), octanoic acid (C 8 ), for example, 2-ethylhexanoic acid (C 8 ), And tetraesters with nonanoic acid (C 9 ), decanoic acid (C 10 ) and / or dodecanoic acid (C 12 ).
- the total number of carbon atoms of the fatty acid constituting the triester of pentaerythritol and fatty acid that is, the above-mentioned In the formula (2), the total number of carbon atoms of the R 1 C, R 2 C and R 3 C moieties is preferably about 19 or more (when the total number of carbon atoms is 19, the IOB is 0.58). Becomes).
- the total number of carbon atoms of the fatty acid constituting the diester of pentaerythritol and fatty acid that is, the above formula ( In 3)
- the total number of carbon atoms in the R 1 C and R 2 C moieties is preferably about 22 or more (when the total number of carbon atoms is 22, IOB is 0.59).
- the carbon number of the R 1 C moiety is preferably about 25 or more (when the carbon number is 25, IOB is 0.60).
- the effects of double bonds, triple bonds, iso branches, and tert branches are not considered (the same applies hereinafter).
- esters of pentaerythritol and fatty acids examples include Unistar H-408BRS, H-2408BRS-22 (mixed product) and the like (manufactured by NOF Corporation).
- ester of (a 2 ) chain hydrocarbon triol and at least one fatty acid examples include the following formula (5): Triester of glycerin and fatty acid of the following formula (6): Diesters of glycerin and fatty acids and the following formula (7): (Wherein R 5 to R 7 are each a chain hydrocarbon) Monoester of glycerin and fatty acid.
- the ester of glycerin and the fatty acid satisfies the requirements for the kinematic viscosity, water retention and weight average molecular weight.
- the ester of glycerin and fatty acid is preferably a diester or triester, and more preferably a triester, from the viewpoint of reducing the water retention value.
- the triester of glycerin and a fatty acid is also referred to as a triglyceride.
- triester of glycerin and octanoic acid C 8
- triester of glycerin and decanoic acid C 10
- glycerin and dodecanoic acid C 12
- triesters of glycerin and 2 or 3 fatty acids and mixtures thereof.
- Examples of the triesters of glycerin and two or more fatty acids include triesters of glycerin and octanoic acid (C 8 ) and decanoic acid (C 10 ), glycerin, octanoic acid (C 8 ), and decane.
- Examples thereof include triesters with (C 16 ) and octadecanoic acid (C 18 ).
- the triester of glycerin and fatty acid is the total number of carbon atoms of fatty acids constituting the triester of glycerin and fatty acid, that is, in formula (5), R 5
- the total carbon number of the C, R 6 C and R 7 C moieties is preferably about 40 or less.
- the triester of glycerin and fatty acid is the total number of carbon atoms of the fatty acid constituting the triester of glycerin and fatty acid, that is, the formula (5 ),
- the total number of carbon atoms in the R 5 C, R 6 C and R 7 C moieties is preferably about 12 or more (when the total number of carbon atoms is 12, IOB is 0.60).
- the triester of glycerin and a fatty acid is a so-called fat and is a component that can constitute a human body, and thus is preferable from the viewpoint of safety.
- triesters of glycerin and fatty acids include tricoconut oil fatty acid glyceride, NA36, panacet 800, panacet 800B and panacet 810S, and tri-C2L oil fatty acid glyceride and tri-CL oil fatty acid glyceride (above, manufactured by NOF Corporation). ) And the like.
- the diester of glycerin and fatty acid is also called diglyceride.
- diester of glycerin and decanoic acid (C 10 ) diester of glycerin and dodecanoic acid (C 12 ), glycerin and hexadecanoic acid (C 16 ) Examples include diesters, diesters of glycerin and two fatty acids, and mixtures thereof.
- the total number of carbon atoms of the fatty acid constituting the diester of glycerin and fatty acid that is, in the formula (6) , R 5 C and R 6 C are preferably about 16 or more in total (when the total number of carbons is 16, IOB is 0.58).
- Monoesters of glycerin and fatty acids are also known as monoglycerides, for example, octadecanoic acid glycerin (C 18) monoesters, docosanoate glycerin (C 22) monoesters, and the like.
- the carbon number of the fatty acid constituting the monoester of glycerin and fatty acid that is, in the formula (7)
- the carbon number of the R 5 C moiety is preferably about 19 or more (when the carbon number is 19, the IOB is 0.59).
- ester of (a 3 ) chain hydrocarbon diol and at least one fatty acid examples include C 2 to C 6 chain hydrocarbon diols such as C 2 to C 6 glycols such as ethylene glycol, propylene glycol, butylene. Examples thereof include monoesters or diesters of glycol, pentylene glycol or hexylene glycol and a fatty acid.
- ester of the chain hydrocarbon diol and at least one fatty acid for example, the following formula (8): R 8 COOC k H 2k OCOR 9 (8) (Wherein k is an integer from 2 to 6 and R 8 and R 9 are each a chain hydrocarbon) A diester of a C 2 -C 6 glycol with a fatty acid and the following formula (9): R 8 COOC k H 2k OH (9) (Wherein k is an integer from 2 to 6 and R 8 is a chain hydrocarbon) And monoesters of C 2 -C 6 glycols and fatty acids.
- the fatty acid to be esterified (corresponding to R 8 COOH and R 9 COOH in formula (8) and formula (9)) is C 2 -C 6 glycol.
- an ester of a fatty acid are not particularly limited as long as the ester satisfies the above requirements of kinematic viscosity, water retention and weight average molecular weight.
- “(a 1 ) chain hydrocarbon tetraol and at least one fatty acid Fatty acids listed in "Ester with” that is, saturated fatty acids and unsaturated fatty acids, and saturated fatty acids are preferred in consideration of the possibility of modification by oxidation or the like.
- the carbon number of the R 8 C and R 9 C moieties Is preferably about 6 or more (when the total number of carbon atoms is 6, IOB is 0.60).
- the carbon number of the R 8 C moiety is about It is preferably 12 or more (when the number of carbon atoms is 12, IOB is 0.57).
- the ester of a C 2 ⁇ C 6 glycols and fatty acid in view of the potential for degradation by oxidation and the like, derived from saturated fatty acids, esters of C 2 ⁇ C 6 glycols and fatty acid, Nachi Suwa, C 2 An ester of ⁇ C 6 glycol and saturated fatty acid is preferred.
- an ester of glycol and fatty acid derived from glycol having a large carbon number for example, butylene glycol, pentylene, etc. It is preferably an ester of glycol and fatty acid derived from lenglycol or hexylene glycol.
- the ester of the C 2 -C 6 glycol and the fatty acid is preferably a diester from the viewpoint of reducing the water retention value. Examples of commercial products of the ester of C 2 -C 6 glycol and fatty acid include Compol BL and Compol BS (manufactured by NOF Corporation).
- compound (B1) As a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups substituting hydrogen atoms of the chain hydrocarbon moiety (hereinafter sometimes referred to as “compound (B1)”) And those listed as the compound (A1) in “Compound (A)”, for example, pentaerythritol, glycerin, and glycol.
- Examples of the compound (B2) having a chain hydrocarbon moiety and one hydroxyl group replacing the hydrogen atom of the chain hydrocarbon moiety include: , Compounds in which one hydrogen atom of a hydrocarbon is replaced by one hydroxyl group (—OH), for example, aliphatic monohydric alcohols, for example, saturated aliphatic monohydric alcohols and unsaturated aliphatic monohydric alcohols Is mentioned.
- saturated aliphatic monohydric alcohol examples include C 1 to C 20 saturated aliphatic monohydric alcohols such as methyl alcohol (C 1 ) (C 1 represents the number of carbon atoms, the same shall apply hereinafter), ethyl alcohol ( C 2 ), propyl alcohol (C 3 ) and isomers thereof such as isopropyl alcohol (C 3 ), butyl alcohol (C 4 ) and isomers thereof such as sec-butyl alcohol (C 4 ) and tert-butyl alcohol (C 4 ), pentyl alcohol (C 5 ), hexyl alcohol (C 6 ), heptyl alcohol (C 7 ), octyl alcohol (C 8 ) and isomers thereof such as 2-ethylhexyl alcohol (C 8 ), nonyl alcohol (C 9), decyl alcohol (C 10), dodecyl alcohol (C 12), tetradecyl alcohol (C 14), Hexadecyl alcohol (C 16), to
- Examples of the unsaturated aliphatic monohydric alcohol include those obtained by substituting one of the C—C single bonds of the saturated aliphatic monohydric alcohol with a C ⁇ C double bond, such as oleyl alcohol. It is commercially available from Shin Nippon Rika Co., Ltd. under the names of the Jamaica Coal series and the Angelo All series.
- Examples of the compound (B) include (b 1 ) ethers of chain hydrocarbon tetraol and at least one aliphatic monohydric alcohol, such as monoether, diether, triether and tetraether, preferably diether, triether.
- Ethers and tetraethers more preferably triethers and tetraethers, and even more preferably tetraethers, ethers of (b 2 ) chain hydrocarbon triols and at least one aliphatic monohydric alcohol, such as monoethers, diethers and Triethers, preferably diethers and triethers, and more preferably triethers, and (b 3 ) ethers of chain hydrocarbon diols with at least one aliphatic monohydric alcohol, such as monoethers and diethers, and preferably Diether It is below.
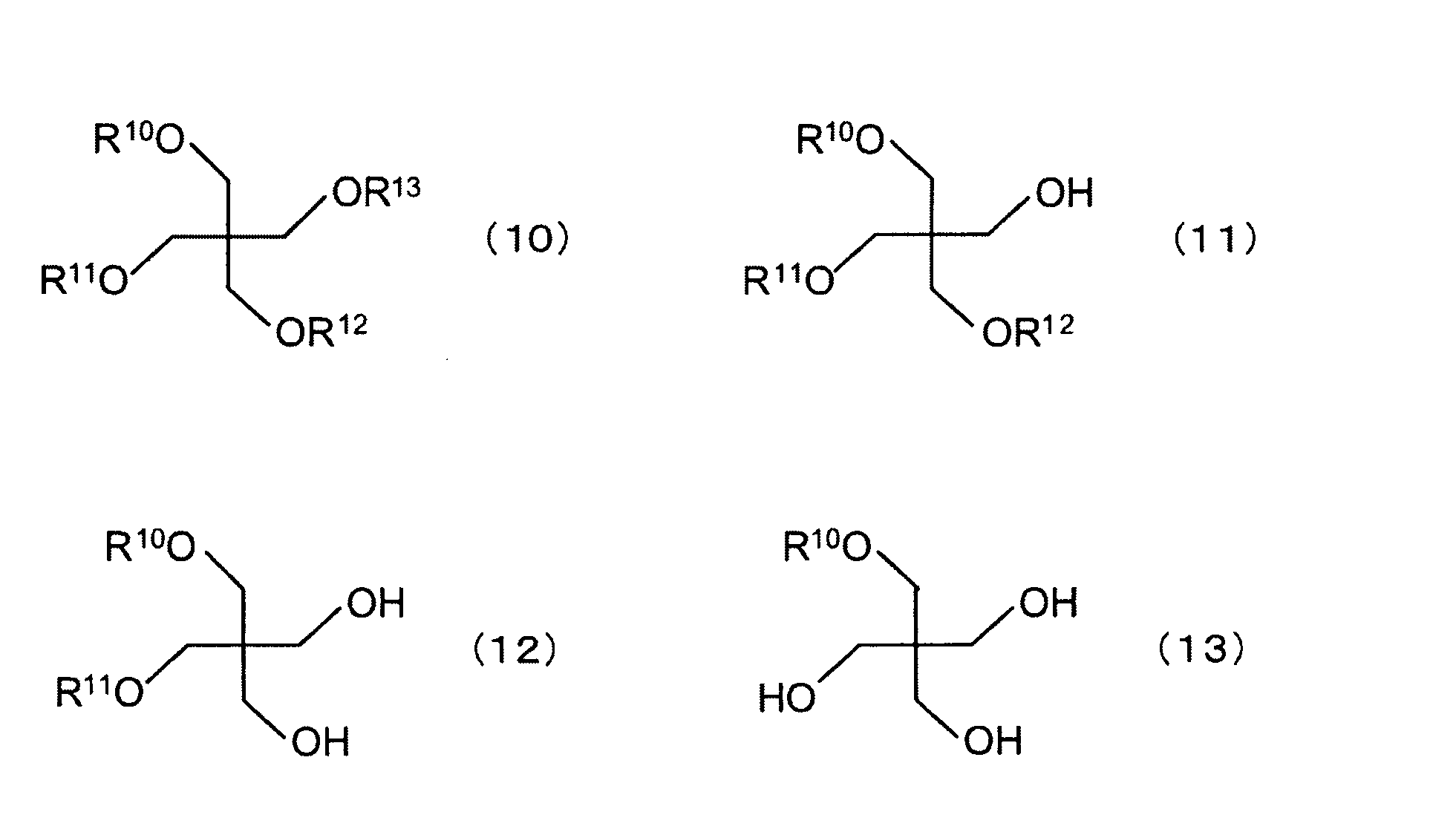
- Examples of the ether of the chain hydrocarbon tetraol and at least one aliphatic monohydric alcohol include, for example, the following formulas (10) to (13): (In the formula, R 10 to R 13 are each a chain hydrocarbon.) And tetraethers, triethers, diethers and monoethers of pentaerythritol and aliphatic monohydric alcohols.
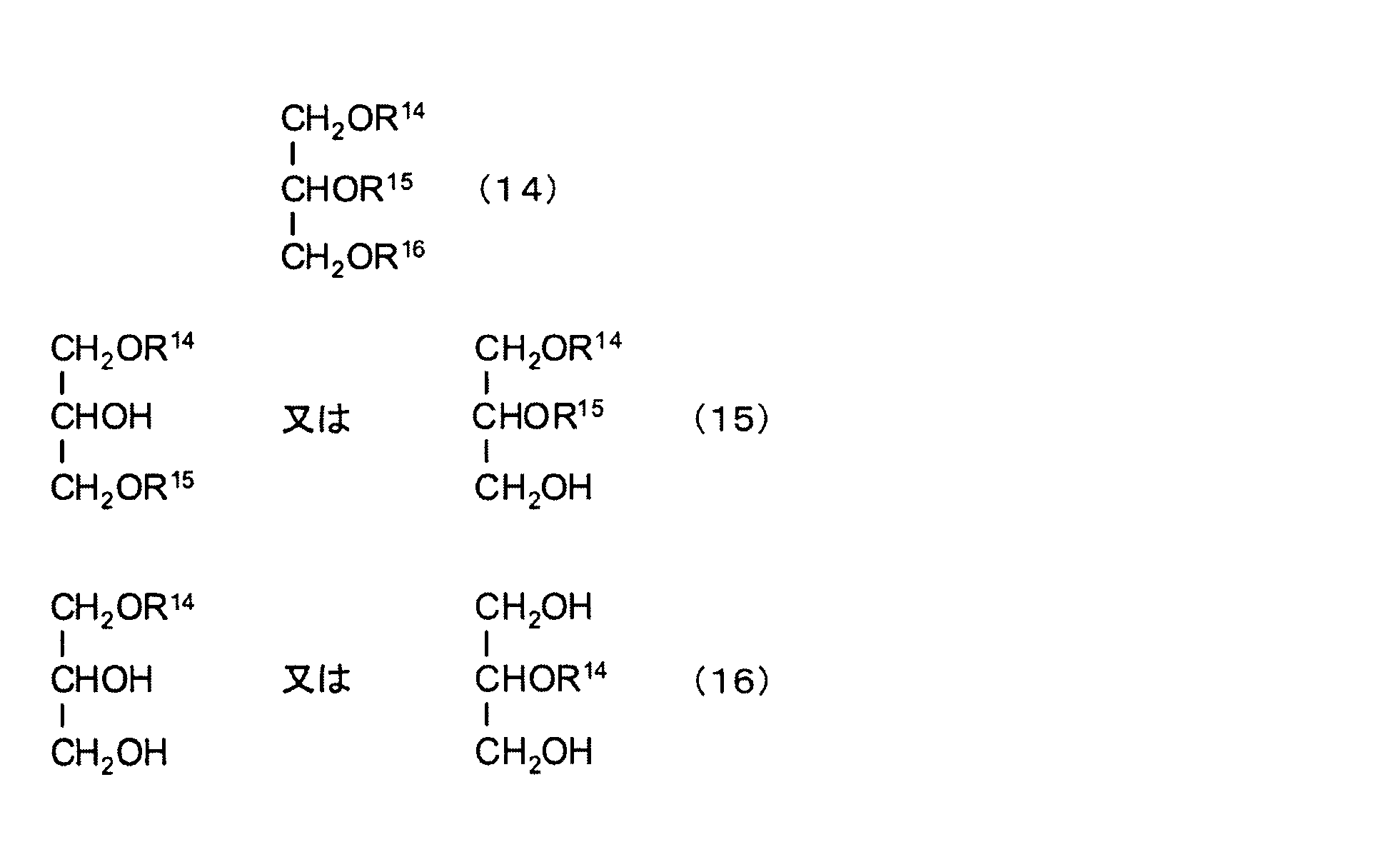
- Examples of the ether of the chain hydrocarbon triol and at least one aliphatic monohydric alcohol include, for example, the following formulas (14) to (16): (Wherein R 14 to R 16 are each a chain hydrocarbon.) And triether, diether and monoether of glycerin and aliphatic monohydric alcohol.
- Examples of the ether of the chain hydrocarbon diol and at least one aliphatic monohydric alcohol include the following formula (17): R 17 OC n H 2n OR 18 (17) (Wherein n is an integer from 2 to 6 and R 17 and R 18 are each a chain hydrocarbon) A diether of a C 2 -C 6 glycol and an aliphatic monohydric alcohol, and the following formula (18): R 17 OC n H 2n OH (18) (Wherein n is an integer from 2 to 6 and R 17 is a chain hydrocarbon) And monoethers of C 2 -C 6 glycols and aliphatic monohydric alcohols.
- the tetraether of pentaerythritol and aliphatic monohydric alcohol described above is an aliphatic constituting the tetraether of pentaerythritol and aliphatic monohydric alcohol.
- the total number of carbon atoms of the monohydric alcohol, that is, in the above formula (10), the total number of carbon atoms of the R 10 , R 11 , R 12 and R 13 portions is preferably about 4 or more (of the above carbon number). If the sum is 4, the IOB is 0.44).
- the above-mentioned triether of pentaerythritol and aliphatic monohydric alcohol is an aliphatic constituting a triether of pentaerythritol and aliphatic monohydric alcohol.
- the total number of carbon atoms of the monohydric alcohol, that is, in the above formula (11), the total number of carbon atoms in the R 10 , R 11 and R 12 portions is preferably about 9 or more (the total number of carbon atoms is 9 In this case, the IOB is 0.57).
- the diether of pentaerythritol and aliphatic monohydric alcohol described above is an aliphatic monovalent constituting a diether of pentaerythritol and aliphatic monohydric alcohol.
- the total number of carbon atoms of the alcohol that is, the total number of carbon atoms in the R 10 and R 11 moieties in the formula (12) is preferably about 15 or more (when the total number of carbon atoms is 15, IOB becomes 0.60).
- the monoether of pentaerythritol and aliphatic monohydric alcohol described above is an aliphatic constituting a monoether of pentaerythritol and aliphatic monohydric alcohol.
- the carbon number of the monohydric alcohol, that is, the carbon number of the R 10 moiety is preferably about 22 or more (when the carbon number is 22, IOB is 0.59).
- the above-described triether of glycerin and aliphatic monohydric alcohol is an aliphatic constituting the triether of glycerin and aliphatic monohydric alcohol. It is preferable that the total number of carbon atoms of the monohydric alcohol, that is, the total number of carbon atoms in the R 14 , R 15 and R 16 moieties in the formula (14) is about 3 or more (the total number of carbon atoms is 3). In this case, IOB becomes 0.50).
- the diether of glycerin and aliphatic monohydric alcohol described above is an aliphatic monohydric alcohol constituting the diether of glycerin and aliphatic monohydric alcohol.
- the total number of carbon atoms that is, the total number of carbon atoms in the R 14 and R 15 moieties in the formula (15) is preferably about 9 or more (when the total number of carbon atoms is 9, the IOB is 0.1. 58).
- the monoether of glycerin and aliphatic monohydric alcohol described above is an aliphatic monovalent constituting a monoether of glycerin and aliphatic monohydric alcohol.
- the carbon number of the alcohol, that is, the carbon number of the R 14 portion is preferably about 16 or more (when the carbon number is 16, the IOB is 0.58).
- R 17 and R 18 moieties
- the total number of carbons is preferably about 2 or more (when the total number of carbons is 2, IOB is 0.33).
- the R 17 moieties The number of carbon atoms is preferably about 8 or more (when the number of carbon atoms is 8, IOB is 0.60).
- Compound (B) can be produced by dehydrating condensation of compound (B1) and compound (B2) in the presence of an acid catalyst.
- C1 Carboxylic acids, hydroxy acids, alkoxy acids or oxo acids (hereinafter referred to as “compounds”) containing a chain hydrocarbon moiety and 2 to 4 carboxyl groups that replace the hydrogen atoms of the chain hydrocarbon moiety.
- C1) may be referred to as, for example, a chain hydrocarbon carboxylic acid having 2 to 4 carboxyl groups, such as a chain hydrocarbon dicarboxylic acid such as an alkanedicarboxylic acid such as ethanedioic acid.
- Examples include rubonic acids such as alkanetetracarboxylic acids such as butanetetraacid, pentanetetraacid, hexanetetraacid, heptanetetraacid, octanetetraacid, nonanetetraacid and decanetetra
- the compound (C1) includes a chain hydrocarbon hydroxy acid having 2 to 4 carboxyl groups, for example, a chain chain having 2 to 4 carboxyl groups such as malic acid, tartaric acid, citric acid, isocitric acid and the like. Hydrocarbon alkoxy acids such as O-acetylcitric acid and chain hydrocarbon oxoacids having 2 to 4 carboxyl groups are included.
- C2 Examples of the compound having a chain hydrocarbon moiety and one hydroxyl group substituting a hydrogen atom of the chain hydrocarbon moiety include those listed in the section of “Compound (B)”, for example, An aliphatic monohydric alcohol is mentioned.
- compound (D) a compound having any one bond selected from the group consisting of a carbonate bond (—OCOO—)
- compound (D) a compound having any one bond selected from the group consisting of a carbonate bond (—OCOO—)
- compound (D) aliphatic
- examples include ethers of monohydric alcohols and aliphatic monohydric alcohols, (d 2 ) dialkyl ketones, esters of (d 3 ) fatty acids and aliphatic monohydric alcohols, and (d 4 ) dialkyl carbonates.
- the ether As the aliphatic monohydric alcohol constituting the ether (corresponding to R 19 OH and R 20 OH in the formula (19)), the ether has the above-mentioned requirements for kinematic viscosity, water retention and weight average molecular weight. If satisfy
- dialkylketone As the dialkyl ketone, the following formula (20): R 21 COR 22 (20) (Wherein R 21 and R 22 are each an alkyl group) The compound which has is mentioned.
- the dialkyl ketone can be obtained by a known method, for example, by oxidizing a secondary alcohol with chromic acid or the like.
- ester of the fatty acid and the aliphatic monohydric alcohol include the following formula (21): R 23 COOR 24 (21) (Wherein R 23 and R 24 are each a chain hydrocarbon) The compound which has is mentioned.
- Examples of the fatty acid constituting the ester include, for example, the fatty acids listed in “(a 1 ) ester of chain hydrocarbon tetraol and fatty acid”, that is, Saturated fatty acids and unsaturated fatty acids are mentioned, and saturated fatty acids are preferred in consideration of the possibility of modification by oxidation or the like.
- Examples of the aliphatic monohydric alcohol constituting the ester include the aliphatic monohydric alcohols listed in the section “Compound (B)”.
- ester of the fatty acid and the aliphatic monohydric alcohol examples include, for example, an ester of dodecanoic acid (C 12 ) and dodecyl alcohol (C 12 ), tetradecanoic acid (C 14 ), and dodecyl alcohol (C 12 ).
- ester of the above fatty acids and aliphatic monohydric alcohols examples include Electol WE20 and Electol WE40 (manufactured by NOF Corporation).
- the dialkyl carbonate can be synthesized by a reaction between phosgene and an alcohol, a reaction between a chlorinated formate and an alcohol or an alcoholate, and a reaction between silver carbonate and an alkyl iodide.
- (d 1 ) an ether of an aliphatic monohydric alcohol and an aliphatic monohydric alcohol, (d 2 ) a dialkyl ketone, (d 3 ) a fatty acid and an aliphatic monohydric alcohol And (d 4 ) dialkyl carbonates preferably have a weight average molecular weight of about 100 or more, and more preferably about 200 or more.
- dialkyl ketone when the total number of carbon atoms is about 8, for example, for 5-nonanone, the melting point is about ⁇ 50 ° C., and the vapor pressure is about 230 Pa at 20 ° C.
- (E) Polyoxy C 3 -C 6 alkylene glycol, or alkyl ester or alkyl ether thereof includes (e 1 ) polyoxy C 3 -C 6 alkylene glycol, (e 2 ) Esters of polyoxy C 3 -C 6 alkylene glycol and at least one fatty acid, (e 3 ) Ethers of polyoxy C 3 -C 6 alkylene glycol and at least one aliphatic monohydric alcohol. This will be described below.
- the polyoxy C 3 -C 6 alkylene glycol has the following formula (23): HO- (C m H 2m O) n -H (23) (Where m is an integer from 3 to 6) Is represented by
- Examples of commercially available poly C 3 -C 6 alkylene glycols include Uniol (trademark) PB-500 and PB-700 (manufactured by NOF Corporation).
- ester of polyoxy C 3 -C 6 alkylene glycol and at least one fatty acid examples include the OH terminal of the polyoxy C 3 -C 6 alkylene glycol described in the section “(e 1 ) polyoxy C 3 -C 6 alkylene glycol”. In which one or both of them are esterified with a fatty acid, that is, monoesters and diesters.
- fatty acid to be esterified in the ester of polyoxy C 3 -C 6 alkylene glycol and at least one fatty acid are listed in “Ester of (a 1 ) chain hydrocarbon tetraol and at least one fatty acid”.
- Fatty acid that is, saturated fatty acid or unsaturated fatty acid, and saturated fatty acid is preferable in consideration of possibility of modification by oxidation or the like.
- Examples of the aliphatic monohydric alcohol to be etherified in the ether of polyoxy C 3 -C 6 alkylene glycol and at least one aliphatic monohydric alcohol include, for example, the aliphatic enumerated in the section of “Compound (B)”. A monohydric alcohol is mentioned.
- chain hydrocarbon examples include (f 1 ) chain alkanes such as straight chain alkanes and branched chain alkanes.
- the linear alkane has a carbon number of about 22 or less when the melting point is about 45 ° C. or less, and about 13 or less when the vapor pressure is about 0.01 Pa or less at 1 atm and 25 ° C. That's it.
- Branched-chain alkanes tend to have lower melting points at the same carbon number than straight-chain alkanes. Therefore, the branched chain alkane can include those having 22 or more carbon atoms even when the melting point is about 45 ° C. or lower.
- Pearl Ream 6 (NOF Corporation) can be mentioned.
- any one that is usually used in the art can be adopted as long as it has a concavo-convex structure including convex portions and concave portions on the skin contact surface.
- Examples thereof include a sheet-like material having a structure that allows liquid to permeate, such as a film, a woven fabric, and a non-woven fabric.
- the fibers constituting the woven fabric and the nonwoven fabric include natural fibers and chemical fibers.
- natural fibers include cellulose such as pulverized pulp and cotton.
- chemical fibers include rayon and fibrillar rayon. And regenerated cellulose, semi-synthetic cellulose such as acetate and triacetate, thermoplastic hydrophobic chemical fibers, and thermoplastic hydrophobic chemical fibers subjected to hydrophilic treatment.
- thermoplastic hydrophobic chemical fiber examples include single fibers such as polyethylene (PE), polypropylene (PP), and polyethylene terephthalate (PET), and fibers made of a graft polymer of PE and PP.
- nonwoven fabric examples include air-through nonwoven fabric, spunbond nonwoven fabric, point bond nonwoven fabric, spunlace nonwoven fabric, needle punch nonwoven fabric, melt blown nonwoven fabric, and combinations thereof (for example, SMS).
- liquid-impermeable back sheet examples include films containing PE, PP, etc., resin films having air permeability, those obtained by bonding a resin film having air permeability to a nonwoven fabric such as spunbond or spunlace, and composite materials such as SMS.
- a layer nonwoven fabric etc. are mentioned.
- a low density polyethylene (LDPE) film having a basis weight of about 15 to about 30 g / m 2 is preferred.
- the absorbent article may include a second sheet between the liquid-permeable top sheet and the absorbent body.
- the second sheet include the same examples as the liquid-permeable top sheet.
- Constituent elements of the absorbent core include, for example, hydrophilic fibers such as cellulose such as pulverized pulp and cotton, regenerated cellulose such as rayon and fibril rayon, semi-synthetic cellulose such as acetate and triacetate, particulate polymer, and fibrous polymer. , Thermoplastic hydrophobic chemical fibers, hydrophobized thermoplastic hydrophobic chemical fibers, and combinations thereof.
- a superabsorbent polymer for example, a granular material such as sodium acrylate copolymer may be mentioned.
- the core wrap is not particularly limited as long as it is liquid permeable and has a barrier property that does not allow the polymer absorber to permeate, and examples thereof include woven fabric and nonwoven fabric.
- examples of the woven fabric and non-woven fabric include natural fibers, chemical fibers, and tissues.
- the absorbent body As a second example of the absorbent body, one formed from an absorbent sheet or a polymer sheet can be cited, and the thickness is preferably about 0.3 to about 5.0 mm.
- the absorbent sheet and polymer sheet can be used without particular limitation as long as they are usually used in absorbent articles such as sanitary napkins.
- the liquid-permeable top sheet is in the excretory opening contact region. Contains a blood lubricity-imparting agent.
- the liquid-permeable top sheet includes a blood slipperiness-imparting agent in a place other than the excretory opening contact area in addition to the excretion opening contact area, for example, the top A blood slipperiness-imparting agent is included on the entire surface of the sheet.
- the liquid permeable top sheet includes a planar region containing a blood slipperiness agent
- the liquid permeable top sheet is a skin contact surface.
- a blood slipperiness imparting agent is included in at least the convex portion of the concavo-convex structure. The presence of a blood slipperiness imparting agent at least in the convex portion causes the blood slipperiness imparting agent present in the convex portion to slide down into the concave portion together with the menstrual blood that has reached the convex portion, and then transfer menstrual blood to the absorber. be able to.
- the liquid-permeable top sheet includes a blood slipperiness imparting agent in both the convex and concave portions of the concavo-convex structure.
- the presence of the blood slipperiness imparting agent in both the convex part and the concave part causes the blood slipperiness imparting agent present in the convex part to slide down into the concave part together with menstrual blood that has reached the convex part, and then the blood present in the concave part
- lubricity imparting agent can transfer the menstrual blood which slid down to the recessed part to an absorber.
- the liquid-permeable topsheet has its skin-side surface, i.e. The skin contact surface contains a blood slipperiness imparting agent.
- the skin contact surface contains a blood slipperiness imparting agent.
- a blood slipperiness-imparting agent is contained in the interior between the surface and the clothing side surface.
- the liquid-permeable top sheet has an entire thickness direction thereof, i.e., the inside of the skin contact surface and the skin contact surface and the clothing side surface, A blood slipping agent is included on the clothing side surface. The presence of the blood slipperiness-imparting agent inside the top sheet and / or the clothing side surface makes it possible to rapidly transfer menstrual blood present on the skin contact surface to the absorbent body.
- the blood slipperiness-imparting agent preferably does not block the gaps between the fibers of the nonwoven fabric or the woven fabric.
- the imparting agent may be attached to the surface of the fiber of the nonwoven fabric or the woven fabric in the form of droplets or particles, or may cover the surface of the fiber.
- the blood slipperiness-imparting agent when the liquid-permeable top sheet is formed from an apertured film, the blood slipperiness-imparting agent preferably does not block the aperture of the apertured film, For example, it can be adhered to the surface of the apertured film in the form of droplets or particles. This is because when the blood slipperiness-imparting agent closes the aperture of the aperture film, the absorbed liquid may be inhibited from transferring to the absorber.
- the blood slipperiness-imparting agent preferably has a large surface area in order to slide with absorbed menstrual blood, and the blood slipperiness-imparting agent present in the form of droplets or particles may have a small particle size. preferable.
- the absorbent article has a second sheet containing a blood slipperiness imparting agent.
- an absorbent article has an absorber containing a blood slipperiness
- the top sheet preferably contains about 1 to about 30 g / m 2 , more preferably about 2 to about 20 g / m 2 , and even more preferably about 3 to about 10 g of the blood slipperiness imparting agent.
- / m including at a basis weight in the range of 2.
- the basis weight of the blood slipperiness-imparting agent is less than about 1 g / m 2 , absorbed menstrual blood tends to remain in the top sheet, and the basis weight of the blood slipperiness-imparting agent exceeds about 30 g / m 2 . And there is a tendency for the stickiness during wearing to increase.
- the top sheet is a non-woven fabric or woven fabric made of a synthetic resin, an apertured film, etc.
- these are coated with a hydrophilic agent on the surface, or It is preferable that the material is hydrophilized by being mixed with a synthetic resin or film. Since the original material has hydrophilicity, the lipophilic region derived from the blood slipperiness-imparting agent and the hydrophilic region derived from the hydrophilic agent coexist sparsely on the top sheet. It is because it becomes easy to slide down from the convex part of a top sheet to a recessed part, and to transfer to an absorber next.
- the method for applying the blood slipperiness-imparting agent is not particularly limited, and is heated as necessary.
- a contactless coater such as a spiral coater, curtain coater, spray coater, dip coater, etc. It can be applied by a coater or the like.
- a non-contact type coater is preferable from the viewpoint that the droplet-like or particulate blood slipperiness-imparting agent is uniformly dispersed throughout and the material is not damaged.
- the blood slipperiness-imparting agent is applied from the control seam HMA gun as it is when it is liquid at room temperature, or heated to lower the viscosity, and heated so as to be liquefied when it is solid at room temperature. can do. By increasing the air pressure of the control seam HMA gun, a particulate blood slipping agent can be applied.
- the blood slipperiness-imparting agent can be applied when a top sheet material, for example, a non-woven fabric is manufactured, or can be applied in a manufacturing line for manufacturing an absorbent article. From the viewpoint of suppressing capital investment, it is preferable to apply a blood slipperiness-imparting agent in the production line for absorbent articles, and to prevent the blood slipperiness-imparting agent from dropping and contaminating the line. It is preferable to apply the blood slipperiness imparting agent immediately downstream of the production line, specifically, immediately before the product is enclosed in the individual package.
- the blood slipperiness-imparting agent can also act as a lubricant. Therefore, when the top sheet is a nonwoven fabric, the blood slipperiness-imparting agent can reduce friction between fibers and improve the flexibility of the entire nonwoven fabric. Moreover, when a top sheet is a resin film, the blood slipperiness
- the absorbent article is one intended to absorb blood, such as a sanitary napkin, a panty liner, or the like.
- the absorbent article of the present disclosure is different from the absorbent article containing a known skin care composition, lotion composition, etc., and does not require components such as an emollient and a fixing agent. It can be applied to the top sheet by itself.
- Tri-C2L oil fatty acid glyceride manufactured by NOF Corporation C 8 fatty acid: C 10 fatty acid: C 12 fatty acid containing approximately 37: 7: 56 weight ratio, glycerol and fatty acid triester, Weight average molecular weight: about 570
- fatty acids manufactured by NOF Corporation C 8 fatty to C 10 are contained in approximately 85:15 weight ratio of triesters of glycerol with fatty acids, the weight average molecular weight: about 480 ⁇ Panasate 800, manufactured by NOF Corporation All fatty acids are octanoic acid (C 8 ), triester of glycerin and fatty acid, weight average molecular weight: about 470
- Panaceate 800B manufactured by NOF Corporation All fatty acids are 2-ethylhexanoic acid (C 8 ), triester of glycerin and fatty acid, weight average molecular weight: about 470 ⁇ NA36, manufactured by NOF Corporation C 16 fatty acid: fatty acid C 18: (including both saturated and unsaturated fatty acids) fatty acids to C 20 is approximately 5: contained in a weight ratio of 3: 92 Triester of glycerin and fatty acid, weight average molecular weight: about 880
- C 8 fatty C 10: fatty acid
- C 12 fatty acid
- C 14 (including both saturated and unsaturated fatty acids)
- C 16 is approximately 4 : Triester of glycerin and fatty acid, contained in a weight ratio of 8: 60: 25: 3, weight average molecular weight: 670 -Caprylic acid diglyceride, manufactured by NOF Corporation
- Fatty acid is octanoic acid, diester of glycerin and fatty acid, weight average molecular weight: 340
- [Other materials] -NA50 manufactured by NOF Corporation Hydrogen added to NA36 to reduce the ratio of double bonds derived from the unsaturated fatty acid as a raw material Triester of glycerin and fatty acid, weight average molecular weight: about 880
- PEG 1500 manufactured by NOF Corporation, polyethylene glycol, weight average molecular weight: about 1,500 to about 1,600 -Wilbright cp9, manufactured by NOF Co., Ltd.
- Nonionic S-6 polyoxyethylene monostearate manufactured by NOF Corporation, about 7 repeating units, weight average molecular weight: about 880 ⁇ Unilube 5TP-300KB Polyoxyethylene polyoxypropylene pentaerythritol ether produced by adding 5 mol of ethylene oxide and 65 mol of propylene oxide to 1 mol of pentaerythritol, weight average molecular weight: 4,130
- Example 1 [A surface residual rate A of menstrual blood when a large amount of blood is absorbed] An experiment was conducted to evaluate the absorbability when a sanitary napkin absorbed a large amount of blood at one time.
- Top sheet formed from air-through nonwoven fabric (composite fiber made of polyester and polyethylene terephthalate, basis weight: 35 g / m 2 ) treated with a hydrophilic agent, and air-through nonwoven fabric (composite fiber made of polyester and polyethylene terephthalate, basis weight: 30 g) / M 2 ), a pulp (basis weight: 150 to 450 g / m 2 , more in the center), an acrylic superabsorbent polymer (basis weight: 15 g / m 2 ), and a tissue as a core wrap.
- An absorbent body, a side sheet treated with a water repellent, and a back sheet made of a polyethylene film were prepared.
- the top sheet is a top sheet having a ridge groove structure manufactured according to the method described in Japanese Patent Application Laid-Open No. 2008-2034.
- the ridge portion has a thickness of about 1.5 mm and the groove portion has a thickness of about 0. 4 mm, the pitch of the ridge groove structure (width of the ridge portion + width of the groove portion) was about 4 mm, and an opening portion having an opening ratio of about 15% was formed in the groove portion.
- Unistar H-408BRS (manufactured by NOF Corporation, tetraester of pentaerythritol and fatty acid) is selected as a blood slipperiness-imparting agent, and the skin contact surface ( ⁇ ⁇ ⁇ ) of the top sheet from a control seam HMA gun at room temperature.
- the groove surface was coated with a basis weight of 5.0 g / m 2 .
- H-408BRS was in the form of fine particles and adhered to the fiber surface.
- the back sheet, the absorbent body, the second sheet, and the top sheet were sequentially laminated with the groove surface facing up, whereby a sanitary napkin no. 1-1 was formed.
- the blood slipperiness-imparting agent was changed from Unistar H-408BRS to that shown in Table 2, and sanitary napkin no. 1-2 to No. 1-49 was produced.
- the blood slipperiness-imparting agent is a liquid at room temperature
- the blood slipperiness-imparting agent is a solid at room temperature
- it is heated to the melting point + 20 ° C., and then a control seam HMA gun is used.
- the blood slipperiness imparting agent was atomized and applied to the skin contact surface of the top sheet so that the basis weight was approximately 5 g / m 2 .
- lubricity imparting agent was applied to the substantially whole surface of the skin contact surface of a top sheet, and to both a collar part and a groove part.
- tackiness of the skin contact surface of the top sheet was measured at 35 ° C. according to the following criteria. ⁇ : No tackiness ⁇ : Some tackiness is present ⁇ : There is tackiness
- Table 2 below shows the surface residual ratio A, tackiness, and characteristics of each blood slipperiness imparting agent of each absorbent article. Moreover, in FIG. 3, the electron micrograph of the skin contact surface of a top sheet in the sanitary napkin in which a top sheet contains tri C2L oil fatty-acid glyceride is shown.
- Sanitary napkin No. which does not have a blood slipperiness imparting agent.
- the surface residual rate A was 7.5% by mass, but the kinematic viscosity and water retention rate were within the predetermined ranges. 1-1-No. In 1-21, the surface residual ratio A was 2.5 mass% or less.
- sanitary napkins no. 1-1-No. 1-21 suggests that when a large amount of menstrual blood reaches the top sheet at a time, menstrual blood can be rapidly transferred from the top sheet to the absorber.
- the top sheet is a top sheet having a ridge groove structure manufactured according to the method described in Japanese Patent Application Laid-Open No. 2008-2034.
- the ridge portion has a thickness of about 1.5 mm and the groove portion has a thickness of about 0. 4 mm, the pitch of the ridge groove structure (width of the ridge portion + width of the groove portion) was about 4 mm, and an opening portion having an opening ratio of about 15% was formed in the groove portion.
- Unistar H-408BRS (manufactured by NOF Corporation, tetraester of pentaerythritol and fatty acid) is selected as a blood slipperiness-imparting agent, and the skin contact surface ( ⁇ ⁇ ⁇ ) of the top sheet from a control seam HMA gun at room temperature.
- the groove surface was coated with a basis weight of 5.0 g / m 2 .
- H-408BRS was in the form of fine particles and adhered to the fiber surface.
- the back sheet, the absorbent body, the second sheet, and the top sheet were sequentially laminated with the groove surface facing up, whereby a sanitary napkin no. 2-1 (i) was formed.
- a top sheet made of a flat, air-through nonwoven fabric composite fiber made of polyester and polyethylene terephthalate, basis weight: 35 g / m 2
- flat hydrophilic agent
- the blood slipperiness-imparting agent was changed from Unistar H-408BRS to that shown in Table 3, and sanitary napkin no. 2-2 (i) -No. 2-11 (i) and No. 2-11. 2-2 (ii) -No. 2-11 (ii) was prepared.
- the blood slipperiness-imparting agent is a liquid at room temperature
- the blood slipperiness-imparting agent is a solid at room temperature
- it is heated to the melting point + 20 ° C., and then a control seam HMA gun is used.
- the blood slipperiness imparting agent was atomized and applied to the skin contact surface of the top sheet so that the basis weight was approximately 5 g / m 2 .
- lubricity imparting agent was apply
- Example 3 [Viscosity of blood containing blood slipping agent] The viscosity of blood containing a blood slipping agent was measured using Rheometric Expansion System ARES (Rheometric Scientific, Inc). 2% by mass of panacet 810s was added to equine defibrinated blood, lightly stirred to form a sample, the sample was placed on a parallel plate with a diameter of 50 mm, the gap was 100 ⁇ m, and the viscosity was measured at 37 ⁇ 0.5 ° C. . Because of the parallel plate, the sample did not have a uniform shear rate, but the average shear rate displayed on the instrument was 10 s ⁇ 1 .
- the viscosity of equine defibrinated blood containing 2% by mass of panacet 810s was 5.9 mPa ⁇ s, while the viscosity of equine defibrinated blood not containing a blood slipperiness agent was 50.4 mPa ⁇ s. Therefore, it can be seen that equine defibrinated blood containing 2% by mass of panacet 810s decreases the viscosity by about 90% compared to the case where no blood slipperiness-imparting agent is contained.
- blood contains components such as blood cells and has thixotropic properties, but the blood slipperiness imparting agent of the present disclosure has an action of lowering the viscosity of blood such as menstrual blood in a low viscosity region. It is thought to have. By reducing the viscosity of blood, it is considered that absorbed menstrual blood is easily transferred from the top sheet to the absorber.
- Example 4 [Micrograph of blood containing blood slipping agent] A healthy volunteer's menstrual blood is collected on a wrap film for food protection, and a portion of the panacet 810s dispersed in 10 times the mass of phosphate buffered saline is used. It added so that it might become. Menstrual blood was appropriately applied to a slide glass, covered with a cover glass, and the state of red blood cells was observed with an optical microscope. A photomicrograph of menstrual blood containing no blood slipperiness agent is shown in FIG. 4 (a), and a photomicrograph of menstrual blood containing panacet 810s is shown in FIG. 4 (b).
- red blood cells form aggregates such as remuneration, but in menstrual blood that includes panacet 810s, red blood cells are each stably dispersed. I understand that. Therefore, it is suggested that the blood slipperiness imparting agent also has a function of stabilizing red blood cells in blood.
- Example 5 [Surface tension of blood containing blood slipperiness agent]
- the surface tension of blood containing a blood slipperiness imparting agent was measured by a pendant drop method using a contact angle meter Drop Master 500 manufactured by Kyowa Interface Science Co., Ltd.
- the surface tension was measured after adding a predetermined amount of blood slipperiness-imparting agent to sheep defibrinated blood and shaking sufficiently. Although the measurement is automatically performed by the instrument, the surface tension ⁇ is obtained by the following equation (see FIG. 5).
- the density ⁇ is defined in 5. of “Density Test Method and Density / Mass / Capacity Conversion Table” of JIS K 2249-1995. Based on the vibration density test method, the temperature was measured as shown in Table 4 below. For measurement, DA-505 of Kyoto Electronics Industry Co., Ltd. was used. The results are shown in Table 4 below.
- the blood slipperiness-imparting agent also has an action of lowering the surface tension of blood.
- the absorbed blood can be quickly transferred to the absorbent body without being held between the fibers of the top sheet.
- the present disclosure relates to the following J1 to J10.
- An absorbent article having a liquid-permeable top sheet, a liquid-impermeable back sheet, and an absorbent body between the liquid-permeable top sheet and the liquid-impermeable back sheet,
- the liquid-permeable top sheet has a concavo-convex structure including a convex part and a concave part on the skin contact surface, and the liquid-permeable top sheet is at least the convex part in the excretory opening contact area.
- a blood slipping agent having a kinematic viscosity at 40 ° C. of 0.01 to 80 mm 2 / s, a water retention of 0.01 to 4.0% by mass, and a weight average molecular weight of less than 1,000.
- the said absorbent article characterized by the above-mentioned.
- the blood slipperiness-imparting agent comprises the following (i) to (iii), (I) hydrocarbons, (Ii) from (ii-1) a hydrocarbon moiety and (ii-2) a carbonyl group (—CO—) and an oxy group (—O—) inserted between the CC single bonds of the hydrocarbon moiety.
- the hydrocarbon moiety A compound having one or a plurality of the same or different groups selected from the group consisting of a carboxyl group (—COOH) and a hydroxyl group (—OH), which replaces a hydrogen atom;
- —COOH carboxyl group
- —OH hydroxyl group
- the blood slipperiness-imparting agent comprises the following (i ′) to (iii ′), (I ′) hydrocarbon, (Ii ′) (ii′-1) a hydrocarbon moiety and (ii′-2) a carbonyl bond (—CO—), an ester bond (—COO) inserted between the C—C single bonds of the hydrocarbon moiety.
- the above-mentioned blood slipperiness-imparting agent comprises the following (A) to (F), (A) (A1) a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups replacing hydrogen atoms in the chain hydrocarbon moiety, (A2) a chain hydrocarbon moiety, and the chain An ester with a compound having one carboxyl group for substituting a hydrogen atom in the hydrocarbon moiety, (B) (B1) a compound having a chain hydrocarbon moiety and 2 to 4 hydroxyl groups replacing hydrogen atoms in the chain hydrocarbon moiety, (B2) a chain hydrocarbon moiety, and the chain An ether with a compound having one hydroxyl group replacing a hydrogen atom of the hydrocarbon moiety, (C) (C1) a carboxylic acid, a hydroxy acid, an alkoxy acid or an oxo acid containing a chain hydrocarbon moiety and 2 to 4 carboxyl groups replacing a hydrogen atom in the chain hydrocarbon moiety; C2) an ester
- the blood lubricity-imparting agent comprises (a 1 ) an ester of a chain hydrocarbon tetraol and at least one fatty acid, (a 2 ) an ester of a chain hydrocarbon triol and at least one fatty acid, (a 3 ) chain Ester of linear hydrocarbon diol and at least one fatty acid, (b 1 ) ether of chain hydrocarbon tetraol and at least one aliphatic monohydric alcohol, (b 2 ) chain hydrocarbon triol and at least one fat Ethers with aliphatic monohydric alcohols, (b 3 ) ethers of chain hydrocarbon diols with at least one aliphatic monohydric alcohol, (c 1 ) chain hydrocarbon tetracarboxylic acids having four carboxyl groups, hydroxy acid, alkoxy acid or an oxo acid, at least one ester of an aliphatic monohydric alcohol, (c 2) a chain hydrocarbon bets with 3 carboxyl groups Carboxy
- the liquid-permeable top sheet has a ridge groove structure including a plurality of ridges and a plurality of grooves on the skin contact surface, and at least the ridge includes the blood slipperiness-imparting agent, J1
Abstract
Description
従って、トップシート側から考察すると、一度に大量の経血が到達する時間、一度に少量の経血が到達する時間、及び経血が到達しない時間とがあり、経血の量を問わず、経血をトップシート上に残存させずに吸収体に移行させることが好ましい。
例えば、特許文献1には、ポリプロピレングリコール材料を含むローション組成物を、トップシートの内表面(着衣側の面)、バックシートの内表面(身体側の面)、トップシートの内表面及びバックシートの内表面の間の基材等に配置した吸収性物品が開示されている。
また、特許文献2には、ポリプロピレングリコール材料を含むローション組成物を、トップシートの外表面(身体側の面)に適用した吸収性物品が開示されている。
従って、本開示は、大量の経血を吸収した場合だけでなく、少量の経血を吸収した場合にも、トップシートにべたつき感がなく、トップシートがサラサラしている吸収性物品を提供することを目的とする。
[液透過性のトップシート]
本開示の吸収性物品では、液透過性のトップシートが、肌当接面に、凸部と、凹部とを含む凹凸構造を有する。本開示のメカニズムについては後述するが、本開示の吸収性物品では、凸部に到達した経血を凹部、次いで吸収体に移行させるため、凸部の高さと、凹部の高さとの間には、一定の差があることが好ましい。
なお、凸部及び凹部の高さは、キーエンス株式会社製 高精度2次元レーザ変位計 LJ-Gシリーズ(型式:LJ-G030)等のレーザ変位計により測定することができる。
本開示の吸収性物品において、液透過性のトップシートが、少なくとも凸部に、40℃における約0.01~約80mm2/sの動粘度と、約0.05~約4.0質量%の抱水率と、約1,000未満の重量平均分子量とを有する血液滑性付与剤を含む。
上記動粘度は、a)血液滑性付与剤の分子量が大きくなるほど、b)極性基、例えば、カルボニル結合(-CO-)、エーテル結合(-O-)、カルボキシル基(-COOH)、ヒドロキシル基(-OH)等の比率が高いほど、そしてc)IOBが大きくなるほど、高くなる傾向がある。
なお、本明細書では、40℃における動粘度を、単に「動粘度」と称する場合がある。
(1)40℃の恒温室に、試験管、ゴム栓、測定すべき物質及び脱イオン水を一昼夜静置する。
(2)上記恒温室で、20mLの試験管に、測定すべき物質5.0gと、脱イオン水5.0gを投入する。
(3)上記恒温室で、試験管の口をゴム栓にて栓をし、1回転させ、5分間静置する。
(5)上記シャーレを、オーブン内で、105℃で3時間加熱し、水分を蒸発させ、シャーレごと、質量を測定する(質量:W1)。
(6)抱水率を、以下の式に従って算出する。
抱水率(%)=100×(W0-W1)/3.0
測定は3回実施し、平均値を採用する。
図1は、本発明の吸収性物品の実施形態の1つである、生理用ナプキンの正面図であり、肌当接面側から観察した図である。図1に示される生理用ナプキン1は、向かって左側が、前方である。図1に示される生理用ナプキン1は、液透過性のトップシート2と、吸収体3と、液不透過性のバックシート(図示せず)とを有する。図1に示される生理用ナプキン1にはまた、サイドシート4、及び圧搾部5が示されている。
Mw=ΣNiMi 2/ΣNiMi
により求められるMwを意味する。
GPCの測定条件としては、例えば、以下が挙げられる。
機種:(株)日立ハイテクノロジーズ製 高速液体クロマトグラム Lachrom Elite
カラム:昭和電工(株)製 SHODEX KF-801、KF-803及びKF-804
溶離液:THF
流量 :1.0mL/分
打込み量:100μL
検出:RI(示差屈折計)
なお、本明細書の実施例に記載される重量平均分子量は、上記条件により測定したものである。
IOB(Inorganic Organic Balance)は、親水性及び親油性のバランスを示す指標であり、本明細書では、小田らによる次式:
IOB=無機性値/有機性値
により算出される値を意味する。
藤田氏による、主要な基の有機性値及び無機性値を、下記表1にまとめる。
(i)炭化水素、
(ii) (ii-1)炭化水素部分と、(ii-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル基(-CO-)及びオキシ基(-O-)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、及び
(iii) (iii-1)炭化水素部分と、(iii-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル基(-CO-)及びオキシ基(-O-)から成る群から選択される、一又は複数の、同一又は異なる基と、(iii-3)上記炭化水素部分の水素原子を置換する、カルボキシル基(-COOH)及びヒドロキシル基(-OH)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、
並びにそれらの任意の組み合わせから成る群から選択される。
上記鎖状炭化水素には、直鎖状炭化水素及び分岐鎖状炭化水素が含まれる。
(i’)炭化水素、
(ii’) (ii’-1)炭化水素部分と、(ii’-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル結合(-CO-)、エステル結合(-COO-)、カーボネート結合(-OCOO-)、及びエーテル結合(-O-)から成る群から選択される、一又は複数の、同一又は異なる結合とを有する化合物、及び
(iii’) (iii’-1)炭化水素部分と、(iii’-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル結合(-CO-)、エステル結合(-COO-)、カーボネート結合(-OCOO-)、及びエーテル結合(-O-)から成る群から選択される、一又は複数の、同一又は異なる結合と、(iii’-3)上記炭化水素部分の水素原子を置換する、カルボキシル基(-COOH)及びヒドロキシル基(-OH)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、
並びにそれらの任意の組み合わせから成る群から選択される。
(A) (A1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(A2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のカルボキシル基とを有する化合物とのエステル、
(B) (B1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(B2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエーテル、
(C) (C1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する、2~4個のカルボキシル基とを含むカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、(C2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエステル、
(D)鎖状炭化水素部分と、上記鎖状炭化水素部分のC-C単結合間に挿入された、エーテル結合(-O-)、カルボニル結合(-CO-)、エステル結合(-COO-)、及びカーボネート結合(-OCOO-)から成る群から選択されるいずれか1つの結合とを有する化合物、
(E)ポリオキシC3~C6アルキレングリコール、又はそのアルキルエステル若しくはアルキルエーテル、及び
(F)鎖状炭化水素、
並びにそれらの任意の組み合わせから成る群から選択される。
以下、(A)~(F)に従う血液滑性付与剤について詳細に説明する。
(A)(A1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(A2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のカルボキシル基とを有する化合物とのエステル(以下、「化合物(A)」と称する場合がある)は、上述の動粘度、抱水率及び重量平均分子量を有する限り、全てのヒドロキシル基がエステル化されていなくともよい。
化合物(A)としては、例えば、(a1)鎖状炭化水素テトラオールと少なくとも1の脂肪酸とのエステル、(a2)鎖状炭化水素トリオールと少なくとも1の脂肪酸とのエステル、及び(a3)鎖状炭化水素ジオールと少なくとも1の脂肪酸とのエステルが挙げられる。
上記鎖状炭化水素テトラオールと少なくとも1の脂肪酸とのエステルとしては、例えば、次の式(1):
(式中、R1~R4は、それぞれ、鎖状炭化水素である)
また、上記ペンタエリトリトールと脂肪酸とのエステルとしては、抱水率の値を小さくする観点から、ジエステル、トリエステル又はテトラエステルであることが好ましく、トリエステル又はテトラエステルであることがより好ましく、そしてテトラエステルであることがさらに好ましい。
なお、上記IOBの計算に当たっては、二重結合、三重結合、iso分岐、及びtert分岐の影響は、考慮していない(以下、同様である)。
上記鎖状炭化水素トリオールと少なくとも1の脂肪酸とのエステルとしては、例えば、次の式(5):
のグリセリンと脂肪酸とのモノエステルが挙げられる。
上記グリセリンと脂肪酸とのトリエステルは、いわゆる、脂肪であり、人体を構成しうる成分であるため、安全性の観点から好ましい。
上記鎖状炭化水素ジオールと少なくとも1の脂肪酸とのエステルとしては、例えば、C2~C6の鎖状炭化水素ジオール、例えば、C2~C6のグリコール、例えば、エチレングリコール、プロピレングリコール、ブチレングリコール、ペンチレングリコール又はヘキシレングリコールと、脂肪酸とのモノエステル又はジエステルが挙げられる。
R8COOCkH2kOCOR9 (8)
(式中、kは、2~6の整数であり、そしてR8及びR9は、それぞれ、鎖状炭化水素である)
のC2~C6グリコールと脂肪酸とのジエステル、及び次の式(9):
R8COOCkH2kOH (9)
(式中、kは、2~6の整数であり、そしてR8は、鎖状炭化水素である)
のC2~C6グリコールと脂肪酸とのモノエステルが挙げられる。
さらに、上記C2~C6グリコールと脂肪酸とのエステルとしては、抱水率の値を小さくする観点から、ジエステルであることが好ましい。
上記C2~C6グリコールと脂肪酸とのエステルの市販品としては、例えば、コムポールBL、コムポールBS(以上、日油株式会社製)等が挙げられる。
(B) (B1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(B2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエーテル(以下、「化合物(B)」と称する場合がある)は、上述の動粘度、抱水率及び重量平均分子量を有する限り、全てのヒドロキシル基がエーテル化されていなくともよい。
の、ペンタエリトリトールと脂肪族1価アルコールとのテトラエーテル、トリエーテル、ジエーテル及びモノエーテルが挙げられる。
の、グリセリンと脂肪族1価アルコールとのトリエーテル、ジエーテル及びモノエーテルが挙げられる。
R17OCnH2nOR18 (17)
(式中、nは、2~6の整数であり、そしてR17及びR18は、それぞれ、鎖状炭化水素である)
のC2~C6グリコールと脂肪族1価アルコールとのジエーテル、及び次の式(18):
R17OCnH2nOH (18)
(式中、nは、2~6の整数であり、そしてR17は、鎖状炭化水素である)
のC2~C6グリコールと脂肪族1価アルコールとのモノエーテルが挙げられる。
また、IOBを約0.00~約0.60とする観点から考察すると、式(18)に示されるエチレングリコール(n=2)と脂肪族1価アルコールとのモノエーテルでは、R17部分の炭素数が、約8以上であることが好ましい(上記炭素数が8の場合に、IOBが0.60となる)。
(C) (C1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する、2~4個のカルボキシル基とを含むカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、(C2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエステル(以下、「化合物(C)」と称する場合がある)は、上述の動粘度、抱水率及び重量平均分子量を有する限り、全てのカルボキシル基がエステル化されていなくともよい。
(C2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物としては、「化合物(B)」の項で列挙されるもの、例えば、脂肪族1価アルコールが挙げられる。
化合物(C)の例としては、アジピン酸ジオクチル、O-アセチルクエン酸トリブチル等が挙げられ、そして市販されている。
(D)鎖状炭化水素部分と、上記鎖状炭化水素部分のC-C単結合間に挿入された、エーテル結合(-O-)、カルボニル結合(-CO-)、エステル結合(-COO-)、及びカーボネート結合(-OCOO-)から成る群から選択されるいずれか1つの結合とを有する化合物(以下、「化合物(D)」と称する場合がある)としては、(d1)脂肪族1価アルコールと脂肪族1価アルコールとのエーテル、(d2)ジアルキルケトン、(d3)脂肪酸と脂肪族1価アルコールとのエステル、及び(d4)ジアルキルカーボネートが挙げられる。
上記脂肪族1価アルコールと脂肪族1価アルコールとのエーテルとしては、次の式(19):
R19OR20 (19)
(式中、R19及びR20は、それぞれ、鎖状炭化水素である)
を有する化合物が挙げられる。
上記ジアルキルケトンとしては、次の式(20):
R21COR22 (20)
(式中、R21及びR22は、それぞれ、アルキル基である)
を有する化合物が挙げられる。
上記ジアルキルケトンは、市販されている他、公知の方法、例えば、第二級アルコールを、クロム酸等で酸化することにより得ることができる。
上記脂肪酸と脂肪族1価アルコールとのエステルとしては、例えば、次の式(21):
R23COOR24 (21)
(式中、R23及びR24は、それぞれ、鎖状炭化水素である)
を有する化合物が挙げられる。
上記ジアルキルカーボネートとしては、次の式(22):
R25OC(=O)OR26 (22)
(式中、R25及びR26は、それぞれ、アルキル基である)
を有する化合物が挙げられる。
上記ジアルキルカーボネートは、市販されている他、ホスゲンとアルコールとの反応、塩化ギ酸エステルとアルコール又はアルコラートとの反応、及び炭酸銀とヨウ化アルキルとの反応により合成することができる。
なお、(d2)ジアルキルケトンにおいて、上記炭素数の合計が約8の場合、例えば、5-ノナノンでは、融点は約-50℃であり、蒸気圧は20℃で約230Paである。
(E)ポリオキシC3~C6アルキレングリコール、又はそのアルキルエステル若しくはアルキルエーテル(以下、化合物(E)と称する場合がある)としては、(e1)ポリオキシC3~C6アルキレングリコール、(e2)ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪酸とのエステル、(e3)ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪族1価アルコールとのエーテルが挙げられる。以下、説明する。
上記ポリオキシC3~C6アルキレングリコールは、i)オキシC3~C6アルキレン骨格、すなわち、オキシプロピレン骨格、オキシブチレン骨格、オキシペンチレン骨格、及びオキシヘキシレン骨格から成る群から選択されるいずれか1種の骨格を有し且つ両末端にヒドロキシ基を有するホモポリマー、ii)上記群から選択される2種以上の骨格を有し且つ両末端にヒドロキシ基を有するブロックコポリマー、又はiii)上記群から選択される2種以上の骨格を有し且つ両末端にヒドロキシ基を有するランダムコポリマーを意味する。
HO-(CmH2mO)n-H (23)
(式中、mは3~6の整数である)
により表わされる。
上記ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪酸とのエステルとしては、「(e1)ポリオキシC3~C6アルキレングリコール」の項で説明したポリオキシC3~C6アルキレングリコールのOH末端の一方又は両方が、脂肪酸によりエステル化されているもの、すなわち、モノエステル及びジエステルが挙げられる。
上記ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪族1価アルコールとのエーテルとしては、「(e1)ポリオキシC3~C6アルキレングリコール」の項で説明したポリオキシC3~C6アルキレングリコールのOH末端の一方又は両方が、脂肪族1価アルコールによりエーテル化されているもの、すなわち、モノエーテル及びジエーテルが挙げられる。
ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪族1価アルコールとのエーテルにおいて、エーテル化すべき脂肪族1価アルコールとしては、例えば、「化合物(B)」の項で列挙されている脂肪族1価アルコールが挙げられる。
上記鎖状炭化水素としては、例えば、(f1)鎖状アルカン、例えば、直鎖アルカン及び分岐鎖アルカンが挙げられる。直鎖アルカンは、融点が約45℃以下の場合には、炭素数が約22以下となり、そして蒸気圧が1気圧及び25℃で約0.01Pa以下である場合には、炭素数が約13以上となる。分岐鎖アルカンは、直鎖アルカンよりも、同一炭素数において融点が低い傾向がある。従って、分岐鎖アルカンは、融点が約45℃以下の場合でも、炭素数が22以上のものも含むことができる。
上記炭化水素の市販品としては、例えば、パールリーム6(日油株式会社)が挙げられる。
上記不織布の例としては、例えば、エアスルー不織布、スパンボンド不織布、ポイントボンド不織布、スパンレース不織布、ニードルパンチ不織布、メルトブローン不織布、及びこれらの組み合わせ(例えば、SMS等)等が挙げられる。
上記吸収コアの構成要素としては、例えば、親水性繊維、例えば、粉砕パルプ、コットン等のセルロース、レーヨン、フィブリルレーヨン等の再生セルロース、アセテート、トリアセテート等の半合成セルロース、粒子状ポリマー、繊維状ポリマー、熱可塑性疎水性化学繊維、及び親水化処理された熱可塑性疎水性化学繊維、並びにこれらの組み合わせ等が挙げられる。また、上記吸収コアの構成要素として、高吸収性ポリマー、例えば、アクリル酸ナトリウムコポリマー等の粒状物が挙げられる。
また、上記血液滑性付与剤は、吸収した経血とともに滑落するために、その表面積が大きいことが好ましく、液滴状又は粒子状で存在する血液滑性付与剤は、粒径が小さいことが好ましい。
なお、本開示の吸収性物品は、公知のスキンケア組成物、ローション組成物等を含む吸収性物品とは異なり、エモリエント剤、固定化剤等の成分が不要であり、血液滑性付与剤は、単体で、トップシートに適用されうる。
以下に、実験に用いられた血液滑性付与剤を列挙する。
[(a1)鎖状炭化水素テトラオールと少なくとも1の脂肪酸とのエステル]
・ユニスター H-408BRS,日油株式会社製
テトラ2-エチルヘキサン酸ペンタエリトリトール,重量平均分子量:約640
・ユニスター H-2408BRS-22,日油株式会社製
テトラ2-エチルヘキサン酸ペンタエリトリトールと、ジ2-エチルヘキサン酸ネオペンチルグリコールとの混合物(58:42、重量比),重量平均分子量:約520
・トリC2L油脂肪酸グリセリド,日油株式会社製
C8の脂肪酸:C10の脂肪酸:C12の脂肪酸がおおよそ37:7:56の重量比で含まれている、グリセリンと脂肪酸とのトリエステル,重量平均分子量:約570
・トリCL油脂肪酸グリセリド,日油株式会社製
C8の脂肪酸:C12の脂肪酸がおおよそ44:56の重量比で含まれている、グリセリンと脂肪酸とのトリエステル,重量平均分子量:約570
C8の脂肪酸:C10の脂肪酸がおおよそ85:15の重量比で含まれている、グリセリンと脂肪酸とのトリエステル,重量平均分子量:約480
・パナセート800,日油株式会社製
脂肪酸が全てオクタン酸(C8)である、グリセリンと脂肪酸とのトリエステル,重量平均分子量:約470
脂肪酸が全て2-エチルヘキサン酸(C8)である、グリセリンと脂肪酸とのトリエステル,重量平均分子量:約470
・NA36,日油株式会社製
C16の脂肪酸:C18の脂肪酸:C20の脂肪酸(飽和脂肪酸及び不飽和脂肪酸の両方を含む)がおおよそ5:92:3の重量比で含まれている、グリセリンと脂肪酸とのトリエステル,重量平均分子量:約880
C8の脂肪酸:C10の脂肪酸:C12の脂肪酸:C14の脂肪酸:C16の脂肪酸(飽和脂肪酸及び不飽和脂肪酸の両方を含む)がおおよそ4:8:60:25:3の重量比で含まれている、グリセリンと脂肪酸とのトリエステル,重量平均分子量:670
・カプリル酸ジグリセリド,日油株式会社製
脂肪酸がオクタン酸である、グリセリンと脂肪酸とのジエステル,重量平均分子量:340
・ユニスター H-208BRS,日油株式会社製
ジ2-エチルヘキサン酸ネオペンチルグリコール,重量平均分子量:約360
・コムポールBL,日油株式会社製
ブチレングリコールのドデカン酸(C12)モノエステル,重量平均分子量:約270
・コムポールBS,日油株式会社製
ブチレングリコールのオクタデカン酸(C18)モノエステル,重量平均分子量:約350
・O-アセチルクエン酸トリブチル,東京化成工業株式会社製
重量平均分子量:約400
・クエン酸トリブチル,東京化成工業株式会社製
重量平均分子量:約360
・アジピン酸ジオクチル,和光純薬工業製
重量平均分子量:約380
・エレクトールWE20,日油株式会社製
ドデカン酸(C12)と、ドデシルアルコール(C12)とのエステル,重量平均分子量:約360
・エレクトールWE40,日油株式会社製
テトラデカン酸(C14)と、ドデシルアルコール(C12)とのエステル,重量平均分子量:約390
・ユニオールPB500,日油株式会社製
ポリブチレングリコール,重量平均分子量:約500
・ユニオールPB700,日油株式会社製
ポリオキシブチレンポリオキシプロピレングリコール,重量平均分子量:約700
・パールリーム6,日油株式会社製
流動イソパラフィン、イソブテン及びn-ブテンを共重合し、次いで水素を付加することにより生成された分岐鎖炭化水素、重合度:約5~約10,重量平均分子量:約330
・NA50,日油株式会社製
NA36に水素を付加し、原料である不飽和脂肪酸に由来する二重結合の比率を下げたグリセリンと脂肪酸とのトリエステル,重量平均分子量:約880
・(カプリル酸/カプリン酸)モノグリセリド,日油株式会社製
オクタン酸(C8)及びデカン酸(C10)がおおよそ85:15の重量比で含まれている、グリセリンと脂肪酸とのモノエステル,重量平均分子量:約220
・Monomuls 90-L2ラウリン酸モノグリセリド,コグニスジャパン株式会社製
重量平均分子量:約230
・リンゴ酸ジイソステアリル
重量平均分子量:約640
・ユニオールPB1000R,日油株式会社製
ポリブチレングリコール,重量平均分子量:約1,000
・ユニオールD-250,日油株式会社製
ポリプロピレングリコール,重量平均分子量:約250
ポリプロピレングリコール,重量平均分子量:約400
・ユニオールD-700,日油株式会社製
ポリプロピレングリコール,重量平均分子量:約700
・ユニオールD-1000,日油株式会社製
ポリプロピレングリコール,重量平均分子量:約1,000
・ユニオールD-1200,日油株式会社製
ポリプロピレングリコール,重量平均分子量:約1,160
ポリプロピレングリコール,重量平均分子量:約2,030
・ユニオールD-3000,日油株式会社製
ポリプロピレングリコール,重量平均分子量:約3,000
・ユニオールD-4000,日油株式会社製
ポリプロピレングリコール,重量平均分子量:約4,000
ポリエチレングリコール,重量平均分子量:約1,500~約1,600
・ウィルブライトcp9,日油株式会社製
ポリブチレングリコールの両末端のOH基が、ヘキサデカン酸(C16)によりエステル化された化合物,重量平均分子量:約1,150
・ユニルーブMS-70K,日油株式会社製
ポリプロピレングリコールのステアリルエーテル,約15の繰返し単位,重量平均分子量:約1,140
ポリオキシエチレンモノステアレート、約7の繰返し単位、重量平均分子量:約880
・ユニルーブ5TP-300KB
ペンタエリトリトール1モルに、エチレンオキシド5モルと、プロピレンオキシド65モルとを付加させることにより生成した、ポリオキシエチレンポリオキシプロピレンペンタエリスリトールエーテル,重量平均分子量:4,130
ポリオキシエチレンポリオキシプロピレンポリオキシブチレングリセリン,重量平均分子量:約960
・ユニオール TG-330,日油株式会社製
ポリプロピレングリコールのグリセリルエーテル,約6の繰返し単位,重量平均分子量:約330
ポリプロピレングリコールのグリセリルエーテル,約16の繰返し単位,重量平均分子量:約1,000
・ユニオール TG-3000,日油株式会社製
ポリプロピレングリコールのグリセリルエーテル,約16の繰返し単位,重量平均分子量:約3,000
・ユニオール TG-4000,日油株式会社製
ポリプロピレングリコールのグリセリルエーテル,約16の繰返し単位,重量平均分子量:約4,000
ポリプロピレングリコールのジグリセリルエーテル,約9の繰返し単位,重量平均分子量:約700
・ユニオックスHC60,日油株式会社製
ポリオキシエチレン硬化ヒマシ油,重量平均分子量:約3,570
・ワセリン,コグニスジャパン株式会社製
石油に由来する炭化水素、半固形
[大量の血液を吸収した際の経血の表面残存率A]
生理用ナプキンが一度に大量の血液を吸収した場合の吸収性を評価する実験を行った。
親水剤で処理されたエアスルー不織布(ポリエステル及びポリエチレンテレフタレートから成る複合繊維、坪量:35g/m2)から形成されたトップシートと、エアスルー不織布(ポリエステル及びポリエチレンテレフタレートから成る複合繊維、坪量:30g/m2)から形成されたセカンドシートと、パルプ(坪量:150~450g/m2、中央部ほど多い)、アクリル系高吸収ポリマー(坪量:15g/m2)及びコアラップとしてのティッシュを含む吸収体と、撥水剤処理されたサイドシートと、ポリエチレンフィルムから成るバックシートとを準備した。
次いで、バックシート、吸収体、セカンドシート、そして畝溝面を上にしてトップシートを順に重ね合わせることにより、生理用ナプキンNo.1-1を形成した。
また、血液滑性付与剤は、トップシートの肌当接面のほぼ全面に、そして畝部及び溝部の両方に塗工された。
トップシートの質量W2(試験前のトップシートの質量)を測定した後、吸収性物品の長手方向及び幅方向の中央部且つトップシートの上に、穴の開いたアクリル板(200mm×100mm,125g,中央に、40mm×10mmの穴が開いている)を置き、上記穴から、37±1℃のウマEDTA血(ウマの血液に、凝結防止のため、エチレンジアミン四酢酸(以下、「EDTA」と称する)が添加されたもの)4.0gを、ピペットを用いて滴下した。
表面残存率A(質量%)
=100×(W3-W2)/4.0
○:タック性なし
△:若干のタック性有り
×:タック性有り
[少量の血液を吸収した際の経血の表面残存率B]
生理用ナプキンが少量の血液を吸収した場合の吸収性を評価する実験を行った。
親水剤で処理されたエアスルー不織布(ポリエステル及びポリエチレンテレフタレートから成る複合繊維、坪量:35g/m2)から形成されたトップシート(以下、「畝溝を有するトップシート」と称する場合がある)と、エアスルー不織布(ポリエステル及びポリエチレンテレフタレートから成る複合繊維、坪量:30g/m2)から形成されたセカンドシートと、パルプ(坪量:150~450g/m2、中央部ほど多い)、アクリル系高吸収ポリマー(坪量:15g/m2)及びコアラップとしてのティッシュを含む吸収体と、撥水剤処理されたサイドシートと、ポリエチレンフィルムから成るバックシートとを準備した。
次いで、バックシート、吸収体、セカンドシート、そして畝溝面を上にしてトップシートを順に重ね合わせることにより、生理用ナプキンNo.2-1(i)を形成した。
また、血液滑性付与剤は、トップシートの肌当接面のほぼ全面に、そして畝溝構造を有するトップシートでは、畝部及び溝部の両方に塗工された。
トップシートの質量W4(試験前のトップシートの質量)を測定した後、吸収性物品の長手方向及び幅方向の中央のトップシートの上に、37±1℃のウマEDTA血約0.25g(2滴)をピペットから滴下した。なお、畝溝を有するトップシートでは、畝部の頂部にウマEDTA血を滴下した。
表面残存率B(質量%)
=100×(W5-W4)/W6
なお、W6は、滴下前後のピペットの質量から算出した、滴下されたウマEDTA血の質量である。
結果を、下記表3に示す。
[血液滑性付与剤を含む血液の粘性]
血液滑性付与剤を含む血液の粘性を、Rheometric Expansion System ARES(Rheometric Scientific,Inc)を用いて測定した。ウマ脱繊維血に、パナセート810sを2質量%添加し、軽く撹拌して試料を形成し、直径50mmのパラレルプレートに試料を載せ、ギャップを100μmとし、37±0.5℃で粘度を測定した。パラレルプレートゆえ、試料に均一なせん断速度はかかっていないが、機器に表示された平均せん断速度は、10s-1であった。
[血液滑性付与剤を含む血液の顕微鏡写真]
健常ボランティアの経血を、食品保護用ラップフィルム上に採取し、その一部に、10倍の質量のリン酸緩衝生理食塩水中に分散されたパナセート810sを、パナセート810sの濃度が1質量%となるように添加した。経血を、スライドグラスに適下し、カバーグラスをかけ、光学顕微鏡にて、赤血球の状態を観察した。血液滑性付与剤を含まない経血の顕微鏡写真を図4(a)に、そしてパナセート810sを含む経血の顕微鏡写真を図4(b)に示す。
[血液滑性付与剤を含む血液の表面張力]
血液滑性付与剤を含む血液の表面張力を、協和界面科学社製接触角計 Drop Master500を用い、ペンダントドロップ法にて測定した。表面張力は、ヒツジ脱繊維血に、所定の量の血液滑性付与剤を添加し、十分振とうした後に測定した。
測定は、機器が自動で行うが、表面張力γは、以下の式により求められる(図5を参照)。
g:重力定数
1/H:ds/deから求められる補正項
ρ:密度
de:最大直径
ds:滴下端よりdeだけ上がった位置での径
測定には、京都電子工業株式会社のDA-505を用いた。
結果を、下記表4に示す。
血液の表面張力を下げることにより、吸収した血液をトップシートの繊維間に保持せず、速やかに吸収体に移行させることができると考えられる。
[J1]
液透過性のトップシートと、液不透過性のバックシートと、上記液透過性のトップシート及び液不透過性のバックシートの間の吸収体とを有する吸収性物品であって、
上記液透過性のトップシートが、肌当接面に、凸部と、凹部とを含む凹凸構造を有し、そして
上記液透過性のトップシートが、排泄口当接域において、少なくとも上記凸部に、40℃における0.01~80mm2/sの動粘度と、0.01~4.0質量%の抱水率と、1,000未満の重量平均分子量とを有する血液滑性付与剤を含む、
ことを特徴とする、上記吸収性物品。
上記血液滑性付与剤が、0.00~0.60のIOBをさらに有する、J1に記載の吸収性物品。
上記血液滑性付与剤が、次の(i)~(iii)、
(i)炭化水素、
(ii) (ii-1)炭化水素部分と、(ii-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル基(-CO-)及びオキシ基(-O-)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、及び
(iii) (iii-1)炭化水素部分と、(iii-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル基(-CO-)及びオキシ基(-O-)から成る群から選択される、一又は複数の、同一又は異なる基と、(iii-3)上記炭化水素部分の水素原子を置換する、カルボキシル基(-COOH)及びヒドロキシル基(-OH)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、
並びにそれらの任意の組み合わせから成る群から選択され、
ここで、(ii)又は(iii)の化合物において、オキシ基が2つ以上挿入されている場合には、各オキシ基は隣接していない、
J1又はJ2に記載の吸収性物品。
上記血液滑性付与剤が、次の(i’)~(iii’)、
(i’)炭化水素、
(ii’) (ii’-1)炭化水素部分と、(ii’-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル結合(-CO-)、エステル結合(-COO-)、カーボネート結合(-OCOO-)、及びエーテル結合(-O-)から成る群から選択される、一又は複数の、同一又は異なる結合とを有する化合物、及び
(iii’) (iii’-1)炭化水素部分と、(iii’-2)上記炭化水素部分のC-C単結合間に挿入された、カルボニル結合(-CO-)、エステル結合(-COO-)、カーボネート結合(-OCOO-)、及びエーテル結合(-O-)から成る群から選択される、一又は複数の、同一又は異なる結合と、(iii’-3)上記炭化水素部分の水素原子を置換する、カルボキシル基(-COOH)及びヒドロキシル基(-OH)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、
並びにそれらの任意の組み合わせから成る群から選択され、
ここで、(ii’)又は(iii’)の化合物において、2以上の同一又は異なる結合が挿入されている場合には、各結合は隣接していない、
J1~J3のいずれか一項に記載の吸収性物品。
上記血液滑性付与剤が、次の(A)~(F)、
(A) (A1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(A2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のカルボキシル基とを有する化合物とのエステル、
(B) (B1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(B2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエーテル、
(C) (C1)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する、2~4個のカルボキシル基とを含むカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、(C2)鎖状炭化水素部分と、上記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエステル、
(D)鎖状炭化水素部分と、上記鎖状炭化水素部分のC-C単結合間に挿入された、エーテル結合(-O-)、カルボニル結合(-CO-)、エステル結合(-COO-)、及びカーボネート結合(-OCOO-)から成る群から選択されるいずれか1つの結合とを有する化合物、
(E)ポリオキシC3~C6アルキレングリコール、又はそのアルキルエステル若しくはアルキルエーテル、及び
(F)鎖状炭化水素、
並びにそれらの任意の組み合わせから成る群から選択される、J1~J4のいずれか一項に記載の吸収性物品。
上記血液滑性付与剤が、(a1)鎖状炭化水素テトラオールと少なくとも1の脂肪酸とのエステル、(a2)鎖状炭化水素トリオールと少なくとも1の脂肪酸とのエステル、(a3)鎖状炭化水素ジオールと少なくとも1の脂肪酸とのエステル、(b1)鎖状炭化水素テトラオールと少なくとも1の脂肪族1価アルコールとのエーテル、(b2)鎖状炭化水素トリオールと少なくとも1の脂肪族1価アルコールとのエーテル、(b3)鎖状炭化水素ジオールと少なくとも1の脂肪族1価アルコールとのエーテル、(c1)4個のカルボキシル基を有する鎖状炭化水素テトラカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、少なくとも1の脂肪族1価アルコールとのエステル、(c2)3個のカルボキシル基を有する鎖状炭化水素トリカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、少なくとも1の脂肪族1価アルコールとのエステル、(c3)2個のカルボキシル基を有する鎖状炭化水素ジカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、少なくとも1の脂肪族1価アルコールとのエステル、(d1)脂肪族1価アルコールと脂肪族1価アルコールとのエーテル、(d2)ジアルキルケトン、(d3)脂肪酸と脂肪族1価アルコールとのエステル、(d4)ジアルキルカーボネート、(e1)ポリオキシC3~C6アルキレングリコール、(e2)ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪酸とのエステル、(e3)ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪族1価アルコールとのエーテル、及び(f1)鎖状アルカン、並びにそれらの任意の組み合わせから成る群から選択される、J1~J5のいずれか一項に記載の吸収性物品。
上記液透過性のトップシートが、肌当接面に、複数の畝部と、複数の溝部とを含む畝溝構造を有し、少なくとも上記畝部が、上記血液滑性付与剤を含む、J1~J6のいずれか一項に記載の吸収性物品。
上記液透過性のトップシートが、少なくとも上記液透過性のトップシートを圧搾することにより形成された圧搾部を有する、J1~J7のいずれか一項に記載の吸収性物品。
[J9]
上記液透過性のトップシートが、不織布又は織布であり、上記血液滑性付与剤が、上記不織布又は織布の繊維の表面に付着している、J1~J8のいずれか一項に記載の吸収性物品。
[J10]
生理用ナプキン又はパンティーライナーである、J1~J9のいずれか一項に記載の吸収性物品。
2 トップシート
3 吸収体
4 サイドシート
5 圧搾部
6 バックシート
7 凸部
8 凹部
9 肌当接面
10 血液滑性付与剤
11,11’,11’’ 経血
Claims (10)
- 液透過性のトップシートと、液不透過性のバックシートと、前記液透過性のトップシート及び液不透過性のバックシートの間の吸収体とを有する吸収性物品であって、
前記液透過性のトップシートが、肌当接面に、凸部と、凹部とを含む凹凸構造を有し、そして
前記液透過性のトップシートが、排泄口当接域において、少なくとも前記凸部に、40℃における0.01~80mm2/sの動粘度と、0.01~4.0質量%の抱水率と、1,000未満の重量平均分子量とを有する血液滑性付与剤を含む、
ことを特徴とする、前記吸収性物品。 - 前記血液滑性付与剤が、0.00~0.60のIOBをさらに有する、請求項1に記載の吸収性物品。
- 前記血液滑性付与剤が、次の(i)~(iii)、
(i)炭化水素、
(ii) (ii-1)炭化水素部分と、(ii-2)前記炭化水素部分のC-C単結合間に挿入された、カルボニル基(-CO-)及びオキシ基(-O-)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、及び
(iii) (iii-1)炭化水素部分と、(iii-2)前記炭化水素部分のC-C単結合間に挿入された、カルボニル基(-CO-)及びオキシ基(-O-)から成る群から選択される、一又は複数の、同一又は異なる基と、(iii-3)前記炭化水素部分の水素原子を置換する、カルボキシル基(-COOH)及びヒドロキシル基(-OH)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、
並びにそれらの任意の組み合わせから成る群から選択され、
ここで、(ii)又は(iii)の化合物において、オキシ基が2つ以上挿入されている場合には、各オキシ基は隣接していない、
請求項1又は2に記載の吸収性物品。 - 前記血液滑性付与剤が、次の(i’)~(iii’)、
(i’)炭化水素、
(ii’) (ii’-1)炭化水素部分と、(ii’-2)前記炭化水素部分のC-C単結合間に挿入された、カルボニル結合(-CO-)、エステル結合(-COO-)、カーボネート結合(-OCOO-)、及びエーテル結合(-O-)から成る群から選択される、一又は複数の、同一又は異なる結合とを有する化合物、及び
(iii’) (iii’-1)炭化水素部分と、(iii’-2)前記炭化水素部分のC-C単結合間に挿入された、カルボニル結合(-CO-)、エステル結合(-COO-)、カーボネート結合(-OCOO-)、及びエーテル結合(-O-)から成る群から選択される、一又は複数の、同一又は異なる結合と、(iii’-3)前記炭化水素部分の水素原子を置換する、カルボキシル基(-COOH)及びヒドロキシル基(-OH)から成る群から選択される、一又は複数の、同一又は異なる基とを有する化合物、
並びにそれらの任意の組み合わせから成る群から選択され、
ここで、(ii’)又は(iii’)の化合物において、2以上の同一又は異なる結合が挿入されている場合には、各結合は隣接していない、
請求項1~3のいずれか一項に記載の吸収性物品。 - 前記血液滑性付与剤が、次の(A)~(F)、
(A) (A1)鎖状炭化水素部分と、前記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(A2)鎖状炭化水素部分と、前記鎖状炭化水素部分の水素原子を置換する1個のカルボキシル基とを有する化合物とのエステル、
(B) (B1)鎖状炭化水素部分と、前記鎖状炭化水素部分の水素原子を置換する2~4個のヒドロキシル基とを有する化合物と、(B2)鎖状炭化水素部分と、前記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエーテル、
(C) (C1)鎖状炭化水素部分と、前記鎖状炭化水素部分の水素原子を置換する、2~4個のカルボキシル基とを含むカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、(C2)鎖状炭化水素部分と、前記鎖状炭化水素部分の水素原子を置換する1個のヒドロキシル基とを有する化合物とのエステル、
(D)鎖状炭化水素部分と、前記鎖状炭化水素部分のC-C単結合間に挿入された、エーテル結合(-O-)、カルボニル結合(-CO-)、エステル結合(-COO-)、及びカーボネート結合(-OCOO-)から成る群から選択されるいずれか1つの結合とを有する化合物、
(E)ポリオキシC3~C6アルキレングリコール、又はそのアルキルエステル若しくはアルキルエーテル、及び
(F)鎖状炭化水素、
並びにそれらの任意の組み合わせから成る群から選択される、請求項1~4のいずれか一項に記載の吸収性物品。 - 前記血液滑性付与剤が、(a1)鎖状炭化水素テトラオールと少なくとも1の脂肪酸とのエステル、(a2)鎖状炭化水素トリオールと少なくとも1の脂肪酸とのエステル、(a3)鎖状炭化水素ジオールと少なくとも1の脂肪酸とのエステル、(b1)鎖状炭化水素テトラオールと少なくとも1の脂肪族1価アルコールとのエーテル、(b2)鎖状炭化水素トリオールと少なくとも1の脂肪族1価アルコールとのエーテル、(b3)鎖状炭化水素ジオールと少なくとも1の脂肪族1価アルコールとのエーテル、(c1)4個のカルボキシル基を有する鎖状炭化水素テトラカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、少なくとも1の脂肪族1価アルコールとのエステル、(c2)3個のカルボキシル基を有する鎖状炭化水素トリカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、少なくとも1の脂肪族1価アルコールとのエステル、(c3)2個のカルボキシル基を有する鎖状炭化水素ジカルボン酸、ヒドロキシ酸、アルコキシ酸又はオキソ酸と、少なくとも1の脂肪族1価アルコールとのエステル、(d1)脂肪族1価アルコールと脂肪族1価アルコールとのエーテル、(d2)ジアルキルケトン、(d3)脂肪酸と脂肪族1価アルコールとのエステル、(d4)ジアルキルカーボネート、(e1)ポリオキシC3~C6アルキレングリコール、(e2)ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪酸とのエステル、(e3)ポリオキシC3~C6アルキレングリコールと少なくとも1の脂肪族1価アルコールとのエーテル、及び(f1)鎖状アルカン、並びにそれらの任意の組み合わせから成る群から選択される、請求項1~5のいずれか一項に記載の吸収性物品。
- 前記液透過性のトップシートが、肌当接面に、複数の畝部と、複数の溝部とを含む畝溝構造を有し、少なくとも前記畝部が、前記血液滑性付与剤を含む、請求項1~6のいずれか一項に記載の吸収性物品。
- 前記液透過性のトップシートが、少なくとも前記液透過性のトップシートを圧搾することにより形成された圧搾部を有する、請求項1~7のいずれか一項に記載の吸収性物品。
- 前記液透過性のトップシートが、不織布又は織布であり、前記血液滑性付与剤が、前記不織布又は織布の繊維の表面に付着している、請求項1~8のいずれか一項に記載の吸収性物品。
- 生理用ナプキン又はパンティーライナーである、請求項1~9のいずれか一項に記載の吸収性物品。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/381,948 US9775751B2 (en) | 2012-02-29 | 2013-02-21 | Absorbent article |
| CN201380011717.XA CN104144664B (zh) | 2012-02-29 | 2013-02-21 | 吸收性物品 |
| AU2013227547A AU2013227547B2 (en) | 2012-02-29 | 2013-02-21 | Absorbent article |
| KR1020147026759A KR102013009B1 (ko) | 2012-02-29 | 2013-02-21 | 흡수성 물품 |
| EA201400844A EA034433B1 (ru) | 2012-02-29 | 2013-02-21 | Абсорбирующее изделие |
| ES13755663T ES2702377T3 (es) | 2012-02-29 | 2013-02-21 | Artículo absorbente |
| EP13755663.5A EP2821041B1 (en) | 2012-02-29 | 2013-02-21 | Absorbent article |
| SG11201404804SA SG11201404804SA (en) | 2012-02-29 | 2013-02-21 | Absorbent article |
| US15/691,791 US10772770B2 (en) | 2012-02-29 | 2017-08-31 | Absorbent article |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012044354A JP5717672B2 (ja) | 2012-02-29 | 2012-02-29 | 吸収性物品 |
| JP2012-044354 | 2012-02-29 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/381,948 A-371-Of-International US9775751B2 (en) | 2012-02-29 | 2013-02-21 | Absorbent article |
| US15/691,791 Continuation US10772770B2 (en) | 2012-02-29 | 2017-08-31 | Absorbent article |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013129236A1 true WO2013129236A1 (ja) | 2013-09-06 |
Family
ID=49082436
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/054382 WO2013129236A1 (ja) | 2012-02-29 | 2013-02-21 | 吸収性物品 |
Country Status (14)
| Country | Link |
|---|---|
| US (2) | US9775751B2 (ja) |
| EP (1) | EP2821041B1 (ja) |
| JP (1) | JP5717672B2 (ja) |
| KR (1) | KR102013009B1 (ja) |
| CN (1) | CN104144664B (ja) |
| AR (1) | AR091799A1 (ja) |
| AU (1) | AU2013227547B2 (ja) |
| EA (1) | EA034433B1 (ja) |
| ES (1) | ES2702377T3 (ja) |
| MY (1) | MY168929A (ja) |
| SG (1) | SG11201404804SA (ja) |
| TR (1) | TR201819034T4 (ja) |
| TW (1) | TWI573577B (ja) |
| WO (1) | WO2013129236A1 (ja) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014050600A1 (ja) * | 2012-09-30 | 2014-04-03 | ユニ・チャーム株式会社 | 不織布及び吸収性物品 |
| WO2014050368A1 (ja) * | 2012-09-28 | 2014-04-03 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2014054649A1 (ja) * | 2012-10-03 | 2014-04-10 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2014196670A1 (ja) * | 2013-09-30 | 2014-12-11 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2014200121A1 (ja) * | 2013-09-30 | 2014-12-18 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2016002351A1 (ja) * | 2014-06-30 | 2016-01-07 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP2017214694A (ja) * | 2016-05-31 | 2017-12-07 | 花王株式会社 | 不織布 |
| WO2017209008A1 (ja) * | 2016-05-31 | 2017-12-07 | 花王株式会社 | 不織布 |
| US20210000659A1 (en) * | 2017-11-24 | 2021-01-07 | Daio Paper Corporation | Absorbent article |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6092508B2 (ja) | 2011-09-30 | 2017-03-08 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP5717672B2 (ja) * | 2012-02-29 | 2015-05-13 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP5717685B2 (ja) * | 2012-04-02 | 2015-05-13 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP5717686B2 (ja) * | 2012-04-02 | 2015-05-13 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP6062199B2 (ja) | 2012-09-28 | 2017-01-18 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP6021565B2 (ja) * | 2012-09-28 | 2016-11-09 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP5685234B2 (ja) | 2012-09-30 | 2015-03-18 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP6684880B2 (ja) * | 2018-10-04 | 2020-04-22 | ユニ・チャーム株式会社 | 吸収性物品 |
| US20210298369A1 (en) * | 2018-10-23 | 2021-09-30 | Clearview Systems, Llc | Athletic garment |
| USD919084S1 (en) * | 2019-03-19 | 2021-05-11 | Johnson & Johnson Consumer Inc. | Absorbent article |
| USD917692S1 (en) | 2019-03-19 | 2021-04-27 | Johnson & Johnson Consumer Inc. | Absorbent article |
| JP7101636B2 (ja) * | 2019-04-26 | 2022-07-15 | ユニ・チャーム株式会社 | 吸収性物品、清浄化用シート、及び拭取性向上剤の使用 |
| USD978342S1 (en) * | 2019-09-06 | 2023-02-14 | Kimberly-Clark Worldwide, Inc. | Feminine pad |
| JP7272945B2 (ja) * | 2019-12-25 | 2023-05-12 | ユニ・チャーム株式会社 | 吸収性物品、清浄化用シート、及び拭取性向上組成物の使用 |
| JP2023025797A (ja) * | 2021-08-11 | 2023-02-24 | ユニ・チャーム株式会社 | 吸収性物品 |
Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5717081B2 (ja) | 1974-12-20 | 1982-04-08 | ||
| JPS6434365A (en) | 1987-07-30 | 1989-02-03 | Kao Corp | Surface material for sanitary supplies |
| JPH02229255A (ja) | 1989-03-01 | 1990-09-12 | Kao Corp | 凹凸を有する不織布の製造方法 |
| JPH0784697B2 (ja) | 1984-06-13 | 1995-09-13 | チコピー | 孔あき不織布およびその製造方法 |
| JP2001328191A (ja) | 2000-03-13 | 2001-11-27 | Uni Charm Corp | 開孔シートとこの開孔シートを使用した吸収性物品および前記開孔シートの製造方法。 |
| JP2006255051A (ja) * | 2005-03-16 | 2006-09-28 | Kao Corp | 吸収性物品の表面シート |
| JP2008002034A (ja) | 2006-06-23 | 2008-01-10 | Uni Charm Corp | 不織布、不織布製造方法及び不織布製造装置 |
| JP2008025078A (ja) | 2006-06-23 | 2008-02-07 | Uni Charm Corp | 不織布 |
| JP2008023311A (ja) | 2006-06-23 | 2008-02-07 | Uni Charm Corp | 吸収体、多層吸収体及び吸収性物品 |
| JP2008025085A (ja) | 2006-06-23 | 2008-02-07 | Uni Charm Corp | 不織布及び不織布製造方法 |
| JP2008307179A (ja) | 2007-06-13 | 2008-12-25 | Uni Charm Corp | 吸収性物品及び不織布シート |
| JP2009005767A (ja) * | 2007-06-26 | 2009-01-15 | Kao Corp | 吸収性物品 |
| JP2009030218A (ja) | 2007-06-22 | 2009-02-12 | Uni Charm Corp | 不織布およびその製造方法 |
| JP2010518918A (ja) | 2007-02-16 | 2010-06-03 | ザ プロクター アンド ギャンブル カンパニー | ポリプロピレングリコール材料を含むローションを備える吸収性物品 |
| JP2010285735A (ja) | 2009-05-14 | 2010-12-24 | Uni Charm Corp | 透液性繊維不織布 |
| JP2011038211A (ja) | 2009-08-11 | 2011-02-24 | Uni Charm Corp | 不織布およびその製造方法 |
| JP2011510801A (ja) | 2008-02-15 | 2011-04-07 | ザ プロクター アンド ギャンブル カンパニー | ポリプロピレングリコール材料を含むローションを備える吸収性物品 |
| JP2011074515A (ja) | 2009-09-29 | 2011-04-14 | Unicharm Corp | 不織布およびその製造方法 |
| JP2011080178A (ja) | 2009-10-09 | 2011-04-21 | Unicharm Corp | 不織布 |
Family Cites Families (110)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0304617B1 (en) | 1987-07-30 | 1995-01-11 | Kao Corporation | Sanitary article |
| US4759754A (en) | 1987-08-26 | 1988-07-26 | Personal Products Company | Sanitary napkin |
| JPH082364B2 (ja) | 1987-12-16 | 1996-01-17 | 花王株式会社 | 衛生用品の表面材 |
| JPH0720856B2 (ja) | 1988-12-01 | 1995-03-08 | 株式会社大塚製薬工場 | 皮膚の殺菌・清拭用組成物 |
| US5334176A (en) | 1991-07-23 | 1994-08-02 | The Procter & Gamble Company | Absorbent core for use in catamenial products |
| US5591149A (en) * | 1992-10-07 | 1997-01-07 | The Procter & Gamble Company | Absorbent article having meltblown components |
| ES2092691T5 (es) | 1991-07-23 | 2003-04-16 | Procter & Gamble | Metodo de fabricar un articulo absorbente curvado y conformado. |
| MA22660A1 (fr) | 1991-10-01 | 1993-04-01 | Procter & Gamble | Article absorbant comportant des rabats et des zones d'extensibilite differentielle . |
| JP3091283B2 (ja) | 1991-12-09 | 2000-09-25 | 花王株式会社 | 生理用ナプキン |
| JPH065614A (ja) | 1992-06-17 | 1994-01-14 | Murata Mfg Co Ltd | 熱処理装置 |
| JP2559049Y2 (ja) | 1992-06-30 | 1998-01-14 | ユニ・チャーム株式会社 | 使い捨て衛生吸収物品 |
| JPH08510665A (ja) | 1993-05-21 | 1996-11-12 | ザ、プロクター、エンド、ギャンブル、カンパニー | 染み透り阻止用圧着接合部を有する吸収物品 |
| US5643588A (en) | 1994-11-28 | 1997-07-01 | The Procter & Gamble Company | Diaper having a lotioned topsheet |
| US5693337A (en) * | 1994-07-13 | 1997-12-02 | Wakamoto Pharmaceutical Co., Ltd. | Stable lipid emulsion |
| US6120488A (en) * | 1994-11-28 | 2000-09-19 | The Procter & Gamble Company | Absorbent articles having cuffs and topsheet with skin care composition(s) disposed thereon |
| US5614283A (en) | 1994-12-22 | 1997-03-25 | Tredegar Industries | Absorbent composite with three-dimensional film surface for use in absorbent disposable products |
| JP2974934B2 (ja) | 1995-06-02 | 1999-11-10 | ザ、プロクター、エンド、ギャンブル、カンパニー | 生理用ナプキン |
| US5609587A (en) * | 1995-08-03 | 1997-03-11 | The Procter & Gamble Company | Diaper having a lotioned topsheet comprising a liquid polyol polyester emollient and an immobilizing agent |
| US5650214A (en) | 1996-05-31 | 1997-07-22 | The Procter & Gamble Company | Web materials exhibiting elastic-like behavior and soft, cloth-like texture |
| US5891126A (en) | 1996-08-30 | 1999-04-06 | The Procter & Gamble Company | Absorbent interlabial device treated with a polysiloxane emollient |
| JPH1095810A (ja) | 1996-09-24 | 1998-04-14 | Oji Paper Co Ltd | 高吸水性樹脂及びその製造方法 |
| ZA985679B (en) | 1997-06-30 | 1999-01-26 | Procter & Gamble | Absorbent article with multi-layered extensible wings |
| JP2000510376A (ja) | 1997-12-05 | 2000-08-15 | ザ、プロクター、エンド、ギャンブル、カンパニー | フラップと延伸性ゾーンとを有する体にフィットする複合生理用ナプキン |
| US6262331B1 (en) | 1998-06-05 | 2001-07-17 | The Procter & Gamble Company | Absorbent article having a topsheet that includes selectively openable and closable openings |
| EP1034804A1 (en) | 1999-03-05 | 2000-09-13 | The Procter & Gamble Company | Articles comprising an oxidising agent and a hemolytic agent |
| US6730819B1 (en) | 1999-03-05 | 2004-05-04 | The Procter & Gamble Company | Articles comprising oxidizing and hemolytic agents |
| US6153209A (en) | 1999-09-28 | 2000-11-28 | The Procter & Gamble Company | Article having a transferable breathable skin care composition thereon |
| JP3748743B2 (ja) | 1999-10-04 | 2006-02-22 | ユニ・チャーム株式会社 | 吸収性物品およびその製造方法 |
| JP3611761B2 (ja) | 1999-11-02 | 2005-01-19 | 花王株式会社 | 吸収性物品 |
| US20010044614A1 (en) | 1999-12-23 | 2001-11-22 | Damay Emmanuelle Cecile | Reducing agents for feminine care products |
| CA2357516A1 (en) * | 2000-09-28 | 2002-03-28 | Uni-Charm Corporation | Absorbent article and barrier agent for absorbent article |
| EP1250940B1 (en) | 2001-04-17 | 2010-01-06 | The Procter & Gamble Company | An absorbent article comprising an agent able to convey a perception to the wearer |
| US7005557B2 (en) | 2001-07-03 | 2006-02-28 | The Procter & Gamble Company | Film-forming compositions for protecting skin from body fluids and articles made therefrom |
| JP3926587B2 (ja) | 2001-07-12 | 2007-06-06 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP4855606B2 (ja) | 2001-08-10 | 2012-01-18 | ユニ・チャーム株式会社 | 吸収性物品 |
| SE519783C2 (sv) | 2001-08-31 | 2003-04-08 | Sca Hygiene Prod Ab | Absorberande alster innefattande öppningar i absorptionskroppen |
| US20030082219A1 (en) | 2001-10-01 | 2003-05-01 | The Procter & Gamble Company | Skin care compositions comprising low concentrations of skin treatment agents |
| JP2003192563A (ja) | 2001-12-26 | 2003-07-09 | Lion Corp | 含浸用組成物、皮膚保護用含浸体 |
| SG103382A1 (en) | 2002-01-11 | 2004-04-29 | Uni Charm Corp | Colored absorbent article |
| JP4693847B2 (ja) | 2002-03-26 | 2011-06-01 | ユニ・チャーム株式会社 | 吸収性物品およびその製造方法 |
| JP4278963B2 (ja) | 2002-03-26 | 2009-06-17 | ユニ・チャーム株式会社 | 吸収性物品 |
| US8283515B2 (en) * | 2002-05-03 | 2012-10-09 | Sca Hygiene Products Ab | Absorbent article containing a skin conditioning agent |
| DE60221639T2 (de) * | 2002-05-03 | 2008-05-08 | Sca Hygiene Products Ab | Absorbierender Artikel mit Hautpflegemittel |
| EP1371379B2 (en) * | 2002-06-12 | 2012-06-20 | SCA Hygiene Products AB | Absorbent article containing a skincare composition |
| JP3949020B2 (ja) | 2002-07-19 | 2007-07-25 | 花王株式会社 | 吸収性物品 |
| US9035123B2 (en) | 2002-10-01 | 2015-05-19 | The Procter & Gamble Company | Absorbent article having a lotioned topsheet |
| US7060867B2 (en) * | 2002-11-27 | 2006-06-13 | Kimberly-Clark Worldwide, Inc. | Absorbent article with a body facing liner having discretely placed lotion deposits |
| US8030535B2 (en) | 2002-12-18 | 2011-10-04 | The Procter & Gamble Company | Sanitary napkin for clean body benefit |
| EP1444970B1 (en) * | 2003-02-05 | 2011-08-17 | Kao Corporation | Absorbent article |
| JP2005095759A (ja) | 2003-09-24 | 2005-04-14 | San-Dia Polymer Ltd | 吸収剤とこれを用いてなる吸収性物品 |
| US20050096614A1 (en) | 2003-10-29 | 2005-05-05 | Perez Roberto C. | Cover layer for an absorbent article |
| JP4476102B2 (ja) | 2003-12-11 | 2010-06-09 | 花王株式会社 | 吸収性物品の表面シート |
| BRPI0512331A (pt) | 2004-06-21 | 2008-03-04 | Procter & Gamble | artigo absorvente com camada superior contendo loção |
| JP4459013B2 (ja) | 2004-10-20 | 2010-04-28 | 花王株式会社 | 吸収性物品 |
| ATE519467T1 (de) * | 2004-12-17 | 2011-08-15 | Procter & Gamble | Unregelmässiger auftrag einer lotion auf die obere lage eines absorbierenden artikels |
| US8211078B2 (en) | 2005-02-17 | 2012-07-03 | The Procter And Gamble Company | Sanitary napkins capable of taking complex three-dimensional shape in use |
| JP4523866B2 (ja) | 2005-03-31 | 2010-08-11 | ユニ・チャーム株式会社 | 吸収性物品 |
| CN101184459B (zh) * | 2005-06-02 | 2012-12-12 | 宝洁公司 | 具有横向加强元件的吸收制品 |
| JP4792249B2 (ja) | 2005-07-11 | 2011-10-12 | ユニ・チャーム株式会社 | 体液吸収性物品 |
| JP2006233405A (ja) | 2005-08-16 | 2006-09-07 | Asahi Kasei Chemicals Corp | 繊維処理剤 |
| EP1982697A1 (en) * | 2005-10-03 | 2008-10-22 | Taisho Pharmaceutical Co., Ltd | Emulsion lotion |
| MX2008011419A (es) | 2006-03-14 | 2008-09-22 | Procter & Gamble | Articulos absorbentes con lociones. |
| EP2258408A1 (en) | 2006-04-05 | 2010-12-08 | The Procter & Gamble Company | Absorbent articles including odour control system |
| KR20090012247A (ko) * | 2006-05-26 | 2009-02-02 | 가부시키가이샤 시세이도 | 계면활성제 및 이를 함유하는 조성물 |
| WO2007148799A1 (ja) | 2006-06-23 | 2007-12-27 | Uni-Charm Corporation | 吸収性物品 |
| JP5069890B2 (ja) | 2006-06-23 | 2012-11-07 | ユニ・チャーム株式会社 | 不織布 |
| JP5123512B2 (ja) | 2006-06-23 | 2013-01-23 | ユニ・チャーム株式会社 | 不織布 |
| JP5328088B2 (ja) | 2006-06-23 | 2013-10-30 | ユニ・チャーム株式会社 | 不織布 |
| JP5328089B2 (ja) | 2006-06-23 | 2013-10-30 | ユニ・チャーム株式会社 | 多層不織布及び多層不織布の製造方法 |
| JP5069891B2 (ja) | 2006-06-23 | 2012-11-07 | ユニ・チャーム株式会社 | 不織布 |
| JP5123511B2 (ja) | 2006-06-23 | 2013-01-23 | ユニ・チャーム株式会社 | 不織布 |
| JP4986673B2 (ja) | 2007-03-27 | 2012-07-25 | 花王株式会社 | 吸収性物品 |
| JP5021380B2 (ja) | 2006-07-05 | 2012-09-05 | 花王株式会社 | 吸収性物品 |
| MY154960A (en) | 2006-07-05 | 2015-08-28 | Kao Corp | Absorbent article |
| JP4980685B2 (ja) | 2006-09-29 | 2012-07-18 | 大王製紙株式会社 | 吸収性物品 |
| WO2008069199A1 (ja) | 2006-12-05 | 2008-06-12 | Uni-Charm Corporation | 不織布、不織布の製造方法及び吸収性物品 |
| JP4939196B2 (ja) | 2006-12-12 | 2012-05-23 | ユニ・チャーム株式会社 | 不織布、不織布の製造方法及び吸収性物品 |
| JP4939192B2 (ja) | 2006-12-05 | 2012-05-23 | ユニ・チャーム株式会社 | 不織布、不織布の製造方法及び吸収性物品 |
| KR20090084923A (ko) | 2006-12-13 | 2009-08-05 | 유니챰 가부시키가이샤 | 흡수성 물품 |
| JP5311607B2 (ja) * | 2007-02-23 | 2013-10-09 | 株式会社 資生堂 | 皮膚用または毛髪用組成物 |
| JP4979423B2 (ja) | 2007-03-20 | 2012-07-18 | 花王株式会社 | 吸収性物品 |
| JP4979424B2 (ja) | 2007-03-20 | 2012-07-18 | 花王株式会社 | 吸収性物品 |
| JP4879074B2 (ja) | 2007-04-17 | 2012-02-15 | ユニ・チャーム株式会社 | 不織布製造方法 |
| JP5114087B2 (ja) | 2007-04-17 | 2013-01-09 | ユニ・チャーム株式会社 | 不織布、及び、吸収性物品 |
| WO2008133067A1 (ja) | 2007-04-17 | 2008-11-06 | Uni-Charm Corporation | 不織布、不織布製造方法、及び、吸収性物品 |
| US20080281287A1 (en) * | 2007-05-08 | 2008-11-13 | Marcelo Ana Maria Elena R | Sanitary napkin including body-facing protrusions for preventing side leakage and obliquely arranged embossed channels |
| EP1992366B1 (en) | 2007-05-15 | 2011-07-06 | The Procter & Gamble Company | Use of a lotion composition on an absorbent article for reducing adherence of feces or menses to the skin |
| JP5161491B2 (ja) | 2007-05-30 | 2013-03-13 | ユニ・チャーム株式会社 | 吸収性物品 |
| US20090030391A1 (en) | 2007-07-25 | 2009-01-29 | John Lee Hammons | Absorbent article |
| JP4652387B2 (ja) | 2007-10-03 | 2011-03-16 | 王子製紙株式会社 | 折り返し部を有する吸収体製品 |
| JP2009201878A (ja) | 2008-02-29 | 2009-09-10 | Uni Charm Corp | 吸収性物品 |
| JP5268416B2 (ja) | 2008-05-01 | 2013-08-21 | 大王製紙株式会社 | 吸収性物品の製造方法 |
| JP5173616B2 (ja) | 2008-06-10 | 2013-04-03 | 花王株式会社 | 吸収性物品 |
| JP5371361B2 (ja) | 2008-10-10 | 2013-12-18 | ユニ・チャーム株式会社 | 吸収性物品及び吸収性物品の製造方法 |
| JP5455363B2 (ja) | 2008-12-25 | 2014-03-26 | ユニ・チャーム株式会社 | 薄型吸収性物品 |
| WO2010134456A1 (ja) | 2009-05-20 | 2010-11-25 | 花王株式会社 | 吸収体および吸収性物品 |
| JP5401177B2 (ja) | 2009-06-04 | 2014-01-29 | 花王株式会社 | 吸収性物品 |
| JP2011067484A (ja) | 2009-09-28 | 2011-04-07 | Kao Corp | 吸収性物品 |
| JP5277131B2 (ja) | 2009-09-29 | 2013-08-28 | 松本油脂製薬株式会社 | 透水性付与剤、透水性繊維および不織布の製造方法 |
| JP5530693B2 (ja) | 2009-09-30 | 2014-06-25 | 大王製紙株式会社 | 吸収性物品 |
| JP5395630B2 (ja) | 2009-11-13 | 2014-01-22 | 花王株式会社 | 吸収性物品 |
| JP5466489B2 (ja) | 2009-11-16 | 2014-04-09 | 花王株式会社 | 吸収性物品 |
| JP2011110122A (ja) | 2009-11-24 | 2011-06-09 | Kao Corp | 吸収性物品 |
| JP2011131044A (ja) * | 2009-11-24 | 2011-07-07 | Kao Corp | 吸収性物品 |
| JP5551926B2 (ja) | 2009-12-09 | 2014-07-16 | 花王株式会社 | 吸収性物品 |
| JP5506519B2 (ja) | 2010-04-16 | 2014-05-28 | ユニ・チャーム株式会社 | 凹凸を有する不織布を簡易に製造する方法、及び不織布を簡易に加工する方法 |
| JP5506520B2 (ja) | 2010-04-16 | 2014-05-28 | ユニ・チャーム株式会社 | 伸長性繊維と伸縮性繊維とを含む、凹凸を有する不織布、及び当該不織布を製造する方法 |
| JP5749907B2 (ja) | 2010-08-31 | 2015-07-15 | ユニ・チャーム株式会社 | 吸収性物品及び生理用ナプキン |
| TWI448277B (zh) | 2011-03-31 | 2014-08-11 | Uni Charm Corp | Absorbent items |
| JP5717672B2 (ja) * | 2012-02-29 | 2015-05-13 | ユニ・チャーム株式会社 | 吸収性物品 |
-
2012
- 2012-02-29 JP JP2012044354A patent/JP5717672B2/ja active Active
-
2013
- 2013-02-21 EP EP13755663.5A patent/EP2821041B1/en active Active
- 2013-02-21 TR TR2018/19034T patent/TR201819034T4/tr unknown
- 2013-02-21 SG SG11201404804SA patent/SG11201404804SA/en unknown
- 2013-02-21 WO PCT/JP2013/054382 patent/WO2013129236A1/ja active Application Filing
- 2013-02-21 CN CN201380011717.XA patent/CN104144664B/zh active Active
- 2013-02-21 KR KR1020147026759A patent/KR102013009B1/ko active IP Right Grant
- 2013-02-21 AU AU2013227547A patent/AU2013227547B2/en not_active Ceased
- 2013-02-21 US US14/381,948 patent/US9775751B2/en active Active
- 2013-02-21 EA EA201400844A patent/EA034433B1/ru not_active IP Right Cessation
- 2013-02-21 ES ES13755663T patent/ES2702377T3/es active Active
- 2013-02-21 MY MYPI2014702237A patent/MY168929A/en unknown
- 2013-02-26 TW TW102106714A patent/TWI573577B/zh active
- 2013-02-28 AR ARP130100641A patent/AR091799A1/es active IP Right Grant
-
2017
- 2017-08-31 US US15/691,791 patent/US10772770B2/en active Active
Patent Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5717081B2 (ja) | 1974-12-20 | 1982-04-08 | ||
| JPH0784697B2 (ja) | 1984-06-13 | 1995-09-13 | チコピー | 孔あき不織布およびその製造方法 |
| JPS6434365A (en) | 1987-07-30 | 1989-02-03 | Kao Corp | Surface material for sanitary supplies |
| JPH02229255A (ja) | 1989-03-01 | 1990-09-12 | Kao Corp | 凹凸を有する不織布の製造方法 |
| JP2001328191A (ja) | 2000-03-13 | 2001-11-27 | Uni Charm Corp | 開孔シートとこの開孔シートを使用した吸収性物品および前記開孔シートの製造方法。 |
| JP2006255051A (ja) * | 2005-03-16 | 2006-09-28 | Kao Corp | 吸収性物品の表面シート |
| JP2008023311A (ja) | 2006-06-23 | 2008-02-07 | Uni Charm Corp | 吸収体、多層吸収体及び吸収性物品 |
| JP2008025078A (ja) | 2006-06-23 | 2008-02-07 | Uni Charm Corp | 不織布 |
| JP2008002034A (ja) | 2006-06-23 | 2008-01-10 | Uni Charm Corp | 不織布、不織布製造方法及び不織布製造装置 |
| JP2008025085A (ja) | 2006-06-23 | 2008-02-07 | Uni Charm Corp | 不織布及び不織布製造方法 |
| JP2010518918A (ja) | 2007-02-16 | 2010-06-03 | ザ プロクター アンド ギャンブル カンパニー | ポリプロピレングリコール材料を含むローションを備える吸収性物品 |
| JP2008307179A (ja) | 2007-06-13 | 2008-12-25 | Uni Charm Corp | 吸収性物品及び不織布シート |
| JP2009030218A (ja) | 2007-06-22 | 2009-02-12 | Uni Charm Corp | 不織布およびその製造方法 |
| JP2009005767A (ja) * | 2007-06-26 | 2009-01-15 | Kao Corp | 吸収性物品 |
| JP2011510801A (ja) | 2008-02-15 | 2011-04-07 | ザ プロクター アンド ギャンブル カンパニー | ポリプロピレングリコール材料を含むローションを備える吸収性物品 |
| JP2010285735A (ja) | 2009-05-14 | 2010-12-24 | Uni Charm Corp | 透液性繊維不織布 |
| JP2011038211A (ja) | 2009-08-11 | 2011-02-24 | Uni Charm Corp | 不織布およびその製造方法 |
| JP2011074515A (ja) | 2009-09-29 | 2011-04-14 | Unicharm Corp | 不織布およびその製造方法 |
| JP2011080178A (ja) | 2009-10-09 | 2011-04-21 | Unicharm Corp | 不織布 |
Non-Patent Citations (1)
| Title |
|---|
| FUJITA A.: "Organic compound predictions and organic paradigms", KAGAKU NO RYOIKI, vol. 11, no. 10, 1957, pages 719 - 725 |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9504767B2 (en) | 2012-09-28 | 2016-11-29 | Unicharm Corporation | Absorbent article |
| WO2014050368A1 (ja) * | 2012-09-28 | 2014-04-03 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP2014068944A (ja) * | 2012-09-28 | 2014-04-21 | Uni Charm Corp | 吸収性物品 |
| US10092463B2 (en) | 2012-09-30 | 2018-10-09 | Unicharm Corporation | Nonwoven and absorbent article having a blood slipping agent |
| WO2014050600A1 (ja) * | 2012-09-30 | 2014-04-03 | ユニ・チャーム株式会社 | 不織布及び吸収性物品 |
| WO2014054649A1 (ja) * | 2012-10-03 | 2014-04-10 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2014196670A1 (ja) * | 2013-09-30 | 2014-12-11 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2014200121A1 (ja) * | 2013-09-30 | 2014-12-18 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP2015066373A (ja) * | 2013-09-30 | 2015-04-13 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP2015066383A (ja) * | 2013-09-30 | 2015-04-13 | ユニ・チャーム株式会社 | 吸収性物品 |
| CN105228569A (zh) * | 2013-09-30 | 2016-01-06 | 尤妮佳股份有限公司 | 吸收性物品 |
| JP2016013153A (ja) * | 2014-06-30 | 2016-01-28 | ユニ・チャーム株式会社 | 吸収性物品 |
| WO2016002351A1 (ja) * | 2014-06-30 | 2016-01-07 | ユニ・チャーム株式会社 | 吸収性物品 |
| JP2017214694A (ja) * | 2016-05-31 | 2017-12-07 | 花王株式会社 | 不織布 |
| WO2017209009A1 (ja) * | 2016-05-31 | 2017-12-07 | 花王株式会社 | 不織布 |
| WO2017209008A1 (ja) * | 2016-05-31 | 2017-12-07 | 花王株式会社 | 不織布 |
| JP2017214695A (ja) * | 2016-05-31 | 2017-12-07 | 花王株式会社 | 不織布 |
| CN109219679A (zh) * | 2016-05-31 | 2019-01-15 | 花王株式会社 | 无纺布 |
| RU2735533C2 (ru) * | 2016-05-31 | 2020-11-03 | Као Корпорейшн | Нетканый материал |
| US20210000659A1 (en) * | 2017-11-24 | 2021-01-07 | Daio Paper Corporation | Absorbent article |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102013009B1 (ko) | 2019-08-21 |
| SG11201404804SA (en) | 2014-10-30 |
| EA034433B1 (ru) | 2020-02-07 |
| EP2821041A4 (en) | 2015-10-07 |
| AR091799A1 (es) | 2015-03-04 |
| US20170360624A1 (en) | 2017-12-21 |
| JP2013179982A (ja) | 2013-09-12 |
| US9775751B2 (en) | 2017-10-03 |
| US20150032074A1 (en) | 2015-01-29 |
| AU2013227547A1 (en) | 2014-09-11 |
| JP5717672B2 (ja) | 2015-05-13 |
| AU2013227547B2 (en) | 2017-08-24 |
| TW201408267A (zh) | 2014-03-01 |
| CN104144664B (zh) | 2015-12-09 |
| US10772770B2 (en) | 2020-09-15 |
| EP2821041B1 (en) | 2018-11-14 |
| MY168929A (en) | 2019-01-09 |
| TWI573577B (zh) | 2017-03-11 |
| EP2821041A1 (en) | 2015-01-07 |
| ES2702377T3 (es) | 2019-02-28 |
| EA201400844A1 (ru) | 2015-05-29 |
| TR201819034T4 (tr) | 2019-01-21 |
| KR20140130722A (ko) | 2014-11-11 |
| CN104144664A (zh) | 2014-11-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5717672B2 (ja) | 吸収性物品 | |
| JP5122007B1 (ja) | 吸収性物品 | |
| JP5717686B2 (ja) | 吸収性物品 | |
| JP6021567B2 (ja) | 吸収性物品 | |
| JP5717685B2 (ja) | 吸収性物品 | |
| JP6116179B2 (ja) | 吸収性物品 | |
| JP6116177B2 (ja) | 吸収性物品 | |
| JP5685234B2 (ja) | 吸収性物品 | |
| JP6116178B2 (ja) | 吸収性物品 | |
| JP5726121B2 (ja) | 吸収性物品 | |
| JP6092508B2 (ja) | 吸収性物品 | |
| JP6057648B2 (ja) | 吸収性物品 | |
| JP6012380B2 (ja) | 不織布及び吸収性物品 | |
| JP5988810B2 (ja) | 吸収性物品 | |
| JP6104551B2 (ja) | 吸収性物品 | |
| JP2017109125A (ja) | 吸収性物品 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13755663 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 201400844 Country of ref document: EA Ref document number: 2013755663 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14381948 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2013227547 Country of ref document: AU Date of ref document: 20130221 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20147026759 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112014021344 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112014021344 Country of ref document: BR Kind code of ref document: A2 Effective date: 20140828 |