WO2013172064A1 - エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物及び当該硬化物を用いた電子・電気機器並びに当該硬化物の製造方法 - Google Patents
エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物及び当該硬化物を用いた電子・電気機器並びに当該硬化物の製造方法 Download PDFInfo
- Publication number
- WO2013172064A1 WO2013172064A1 PCT/JP2013/053920 JP2013053920W WO2013172064A1 WO 2013172064 A1 WO2013172064 A1 WO 2013172064A1 JP 2013053920 W JP2013053920 W JP 2013053920W WO 2013172064 A1 WO2013172064 A1 WO 2013172064A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- epoxy
- acid anhydride
- less
- resin composition
- epoxy resin
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/20—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the epoxy compounds used
- C08G59/22—Di-epoxy compounds
- C08G59/24—Di-epoxy compounds carbocyclic
- C08G59/245—Di-epoxy compounds carbocyclic aromatic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F22/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides or nitriles thereof
- C08F22/10—Esters
- C08F22/1006—Esters of polyhydric alcohols or polyhydric phenols, e.g. ethylene glycol dimethacrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F283/00—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G
- C08F283/10—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G on to polymers containing more than one epoxy radical per molecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/4007—Curing agents not provided for by the groups C08G59/42 - C08G59/66
- C08G59/4014—Nitrogen containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/42—Polycarboxylic acids; Anhydrides, halides or low molecular weight esters thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/68—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the catalysts used
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B3/00—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties
- H01B3/18—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances
- H01B3/30—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes
- H01B3/40—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/20—Applications use in electrical or conductive gadgets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/03—Polymer mixtures characterised by other features containing three or more polymers in a blend
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/05—Polymer mixtures characterised by other features containing polymer components which can react with one another
Definitions
- the present invention relates to an epoxy-vinyl copolymerization type liquid resin composition (hereinafter referred to as varnish), a cured product thereof, a method for producing the cured product, and an electronic material which is an insulating material and / or a structural material using the cured product. It relates to electrical equipment.
- Patent Document 4 has an example in which tetrafunctional methylcyclohexene tetracarboxylic dianhydride is used as a curing agent, and the glass transition temperature becomes higher than that in the case of using methylhexahydrophthalic anhydride as a curing agent. It is described.
- Patent Document 5 discloses that when 2 to 10% by weight of triallyl cyanate is blended with an epoxy resin composition containing SMA, which is a copolymer of maleic anhydride and styrene, as a curing agent, the glass transition temperature of the cured product increases. It is disclosed. However, if the heat resistance of the insulating material is increased using a polyfunctionalized, rigidized epoxy resin, a polyfunctionalized acid anhydride, or an acid anhydride having a large molecular weight, the melting temperature and viscosity of the varnish will increase. This leads to a decrease in moldability.
- SMA which is a copolymer of maleic anhydride and styrene
- the insulating material is also required to have high thermal conductivity, toughness, and high voltage resistance.
- a method of reforming by blending organic and inorganic fillers is required. Addition of organic and inorganic fillers to the varnish increases the melt viscosity. The deterioration of moldability prevents the formation of fine and complex insulating structures, so it helps to generate voids between wires and makes it difficult to defoam from the resin composition. Promotes the survival of Air gaps between wirings and air bubbles in the insulating layer impair the insulation reliability of electronic and electrical equipment, and must be avoided.
- a casting varnish for producing an insulating layer, a fixing layer, a housing and the like by impregnation and casting operations it is required to have a low viscosity as well as a high heat resistance.
- Patent Document 7 discloses an alumina ceramic comprising a thermosetting resin composition containing a polyfunctional epoxy resin having two or more epoxy groups per molecule, an acid anhydride curing agent, an inorganic filler, and a surfactant. Is a cured resin product molded with a stress safety factor of 7 or more, a strain safety factor of 10 or more, an allowable defect size of 0.1 mm or more, and a thermal conductivity of 0.7 to 2.5 W when cooled to room temperature. What is / m ⁇ K is disclosed.
- Patent Document 8 includes (a) an epoxy resin containing at least two epoxy groups in one molecule, an average molecular weight of 1000 or less, a phenoxy resin, and at least two maleimide groups in one molecule.
- An imide ring-containing epoxy compound obtained by reacting in advance with a polymaleimide compound, (b) a polyfunctional vinyl monomer containing two or more acrylic, methacrylic or allyl groups in one molecule, and (c) liquid
- a liquid thermosetting resin composition comprising an acid anhydride of the above has been disclosed, no investigation has been made on reducing the viscosity or improving crack resistance of the epoxy resin composition.
- JP-A-6-233486 JP-A-9-316167 JP-A-8-109316 JP 2010-193673 A Japanese National Patent Publication No. 10-505376 JP 2011-01424 A JP 2011-57734 A JP 2004-203955 A
- An object of the present invention is to obtain a cured product having excellent heat resistance while reducing the viscosity of the varnish, and to provide an electronic / electric apparatus using the cured product as an insulating material and / or a structural material.
- Another object of the present invention is to obtain a cured product that improves thermal conductivity, thermal expansibility, toughness and strength and is excellent in heat resistance and crack resistance, and the cured product is used as an insulating material or / And providing an electronic / electrical device as a structural material.
- Another object of the present invention is to improve the thermal conductivity, thermal expansibility and toughness while reducing the viscosity of the varnish, and to obtain a cured product having excellent heat resistance.
- Another object of the present invention is to provide an electronic / electrical device as a structural material.
- Another object of the present invention is to improve the thermal conductivity, thermal expansibility and strength while reducing the viscosity of the varnish, and to obtain a cured product having excellent heat resistance.
- Another object of the present invention is to provide an electronic / electrical device as a structural material.
- Another object of the present invention is to obtain a cured product that improves the thermal conductivity and thermal expansibility while reducing the viscosity of the varnish, and that is excellent in heat resistance. And providing an electronic / electrical device as a structural material.
- a polyfunctional vinyl monomer is blended with a varnish to reduce the varnish viscosity, and a cured product that is a copolymer of the polyfunctional vinyl monomer, an epoxy resin, and an acid anhydride can be formed.
- the heat resistance of the cured product is improved by curing the resin composition.
- the present invention further includes adding a predetermined amount of crushed crystalline silica, crosslinked rubber particles, and core-shell rubber particles to the resin composition of (I) above, whereby the thermal conductivity and thermal expansibility of the cured product are obtained. And improve toughness.
- the present invention further increases the thermal conductivity, thermal expansibility, and strength of the cured product by adding predetermined amounts of crushed crystalline silica and acicular inorganic filler to the resin composition of (I) above. Improve.
- this invention improves the thermal conductivity and thermal expansibility of hardened
- the varnish viscosity can be reduced as compared with the conventional varnish, and the heat resistance of the cured product can be improved. Furthermore, the hardened
- the varnish viscosity can be reduced as compared with conventional varnishes, the thermal conductivity, thermal expansibility, toughness and strength of the cured product can be improved, and the heat resistance and crack resistance can be improved. Can do.
- cured material as an insulating material or / and a structural material can be provided.
- the varnish viscosity can be reduced as compared with the conventional varnish, the thermal conductivity, thermal expansibility and toughness of the cured product can be improved, and the heat resistance can be improved.
- cured material which hardened this thermosetting resin composition, and the electronic / electrical apparatus which uses this hardened
- the varnish viscosity can be reduced as compared with the conventional varnish, the thermal conductivity, thermal expansion and strength of the cured product can be improved, and the heat resistance can be improved.
- cured material which hardened this thermosetting resin composition, and the electronic / electrical apparatus which uses this hardened
- the varnish viscosity can be reduced as compared with the conventional varnish, the thermal conductivity and thermal expansibility of the cured product can be improved, and the heat resistance can be improved.
- cured material as an insulating material or / and a structural material can be provided.
- FIG. 4B is a plan view of FIG. 4A.
- FIG. 4B is a side view of FIG. 4A. It is the ZZ sectional view taken on the line of FIG. 4B.
- It is a perspective schematic diagram of the C type washer made from SUS309S used for a crack resistance test. It is sectional drawing of the screw 6 of FIG. 5A.
- FIG. 5B is a schematic diagram showing a state in which the C-type washer made of SUS309S shown in FIG. It is sectional drawing of the cup 7 of FIG. 6A.
- FIG. 7B is a schematic diagram of the sample prepared in FIG. 7A (a C-type washer embedded in a cured product). It is a figure which shows the conditions of the thermal shock (temperature and time) in a crack-proof test.
- Patent Document 1 and Patent Document 2 Conventionally, a method of applying a diluent as described in Patent Document 1 and Patent Document 2 is generally used to lower the viscosity of a varnish.
- the addition of a diluent reduces the heat resistance of the cured product, so that the amount used should be suppressed.
- the copolymer refers to a polymer obtained by combining plural types of monomers or prepolymers by a polymerization reaction. Therefore, the copolymer consisting of the above acid anhydride, epoxy resin and polyfunctional vinyl monomer is a polymer obtained by combining an acid anhydride, epoxy resin and polyfunctional vinyl monomer by a polymerization reaction, Inevitably, a relatively small amount of other monomers or prepolymers (desirably 5% by weight or less of the total amount of plural types of monomers or prepolymers constituting the resin composition) are involved in the polymerization reaction, The monomer may be a constituent of the obtained polymer, but is essentially a polymer composed of structural units derived from the above-described plural types of monomers or prepolymers.
- the present inventor blends a predetermined amount of composite fine particles composed of crushed crystalline silica and rubber particles having a specific range of size into the resin composition. It has been found that the expandability and toughness can be improved and excellent heat resistance can be imparted.
- the present inventor blends a predetermined amount of composite fine particles composed of crushed crystalline silica having a size in a specific range and an acicular inorganic filler into the resin composition, whereby the thermal conductivity of the cured product is obtained. It has been found that the thermal expansion and strength can be improved and excellent heat resistance can be imparted.
- the present inventor improves the thermal conductivity and thermal expansibility of the cured product by blending a predetermined amount of crushed crystalline silica having a size in a specific range into the resin composition, and It has been found that excellent heat resistance can be imparted.
- thermosetting liquid resin composition of the present invention is referred to as an epoxy-vinyl copolymer liquid resin composition
- the cured product is referred to as an epoxy-vinyl copolymer insulating material.
- the mechanism for improving heat resistance is an increase in crosslink density due to copolymerization and a decrease in the thermal decomposition reaction rate, and the mechanism for improving crack resistance is stress relaxation and stress dispersion by rubber particles. .
- the cured resin composition is a cured product that is a copolymer of the epoxy resin and the acid anhydride (A) or the acid anhydride and maleic anhydride (B) and the polyfunctional vinyl monomer.
- An epoxy-vinyl copolymerization type liquid resin composition (I) which can be formed.
- This composition is more than the conventional acid anhydride curable epoxy resin composition by forming a copolymer comprising an epoxy resin, an acid anhydride (above (A) or (B)) and a polyfunctional vinyl monomer. High heat resistance can be realized.
- An acid anhydride having an unsaturated double bond is chemically bonded to each of the epoxy resin and the polyfunctional vinyl monomer to form a copolymer composed of the epoxy resin, the acid anhydride, and the polyfunctional vinyl monomer.
- Epoxy resins and polyfunctional vinyl monomers must not be modified products combined with other compounds (unmodified epoxy resins, unmodified vinyl monomers) in order to avoid an increase in molecular weight (increase in viscosity of the composition). preferable.
- maleic anhydride is solid at normal temperature, it dissolves in other components and becomes liquid, so it is used in the present invention.
- a cured product that is a copolymer comprising at least the epoxy resin, the acid anhydride (A) or the acid anhydride and maleic anhydride (B), and the polyfunctional vinyl monomer is formed. It is important to adjust the resin composition to be obtained to be liquid at 17 ° C., and this property makes the resin composition easy to handle.
- the resin composition further includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, (b) an average diameter of 0.1 ⁇ m or more and 3 ⁇ m or less, and an average length of 10 ⁇ m or more. (A) to (c) a needle-like inorganic filler having a particle diameter of 50 ⁇ m or less, (c) a crosslinked rubber particle having an average particle diameter of 10 nm or more and 100 nm or less, and (d) a core-shell rubber particle having an average particle diameter of 100 nm or more and 2000 nm or less.
- An epoxy-vinyl copolymer liquid resin composition (II) containing one or more fine particles of (d).
- the crushed crystalline silica can impart high thermal conductivity and low thermal expansibility
- the needle-like inorganic filler can impart high strength
- the rubber particles can impart high toughness.
- the resin composition further includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, (c) crosslinked rubber particles having an average particle diameter of 10 nm or more and 100 nm or less, and (d) an average.
- An epoxy-vinyl copolymerization type liquid resin composition (III) comprising composite fine particles composed of core-shell rubber particles having a particle size of 100 nm or more and 2000 nm or less.
- the resin composition further includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, and (b) an average diameter of 0.1 ⁇ m or more and 3 ⁇ m or less, and an average length of 10 ⁇ m or more.
- An epoxy-vinyl copolymerization type liquid resin composition (IV) comprising composite fine particles composed of an acicular inorganic filler having a size of 50 ⁇ m or less.
- the composite fine particles are composed of (a) 83 to 94% by weight of crushed crystalline silica, (b) 1 to 5% by weight of acicular inorganic filler, and (c) crosslinked rubber particles based on the whole composite fine particles.
- the viscosity of the base varnish composed of the epoxy resin of the present invention, acid anhydride or maleic anhydride, and polyfunctional vinyl monomer is low, and therefore contains composite fine particles in this range. Also in the varnish, the increase in viscosity is suppressed, and defoaming work and casting work can be carried out efficiently.
- These fine particles may be in contact with each other and agglomerated, but it is desirable that they are uniformly dispersed in the same varnish so that the varnish viscosity does not become non-uniform. Thereby, casting workability
- the resin composition further comprises (A) the acid anhydride or (B) 1 to 33 mol% or less of maleic anhydride based on the total amount of the acid anhydride and maleic anhydride.
- Maleic anhydride has a high radical polymerizability and facilitates the formation of a copolymer of an epoxy resin, an acid anhydride and a polyfunctional vinyl monomer.
- “1 to 33 mol%” means 1 mol% or more and 33 mol% or less.
- the ratio of the number of equivalents of (A) the acid anhydride or (B) the acid anhydride and maleic anhydride to the number of equivalents of the epoxy resin is 0.9 or more and less than 1.0 in the following formula 1.
- the polyfunctional vinyl monomer ranges from 10 parts by weight to 100 parts by weight with respect to 100 parts by weight of the total amount of the epoxy resin and (A) the acid anhydride or (B) the acid anhydride and maleic anhydride.
- the epoxy curing catalyst is contained in the range of 0.08 parts by weight or more and 1.0 parts by weight or less with respect to 100 parts by weight of the epoxy resin, and the radical polymerization catalyst is contained in 100 parts by weight of the polyfunctional vinyl monomer.
- An epoxy-vinyl copolymerization type liquid resin composition characterized by containing in an amount of 0.5 parts by weight or more and 2 parts by weight or less based on the weight.
- Equivalent ratio Equivalent number of total acid anhydride in resin composition / Equivalent number of total epoxy resin in resin composition. (However, the number of equivalents of acid anhydride having all unsaturated double bonds is the amount of total acid anhydride having unsaturated double bonds in the resin composition divided by the equivalent of acid anhydride having unsaturated double bonds. The total number of equivalents of the epoxy resin is a value obtained by dividing the total amount of epoxy resin in the resin composition by the equivalent of epoxy resin.) With this configuration, an insulating varnish capable of suppressing the remaining of unreacted epoxy resin and acid anhydride can be obtained.
- the resin composition is an inorganic or / and organic filler other than (a) crushed crystalline silica, (b) acicular inorganic filler, (c) crosslinked rubber particles, and (d) core-shell rubber particles.
- a cured product formed by curing a resin composition containing a radical polymerization catalyst that promotes The cured product is an epoxy-vinyl copolymer cured product comprising the epoxy resin, the acid anhydride or the acid anhydride and maleic anhydride, and the polyfunctional vinyl monomer. .
- the cured product is further (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, (b) an average diameter of 0.1 ⁇ m or more and 3 ⁇ m or less, and an average length of 10 ⁇ m or more, Including composite fine particles composed of acicular inorganic fillers having a diameter of 50 ⁇ m or less, (c) crosslinked rubber particles having an average particle diameter of 10 nm or more and 100 nm or less, and (d) core-shell rubber particles having an average particle diameter of 100 nm or more and 2000 nm or less.
- An epoxy-vinyl copolymer cured product characterized by the above.
- the cured product further includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, (c) crosslinked rubber particles having an average particle diameter of 10 nm or more and 100 nm or less, and (d) an average particle An epoxy-vinyl copolymer cured product comprising composite fine particles composed of core-shell rubber particles having a diameter of 100 nm or more and 2000 nm or less.
- the cured product further includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, and (b) an average diameter of 0.1 ⁇ m or more and 3 ⁇ m or less, and an average length of 10 ⁇ m or more.
- An epoxy-vinyl copolymer-type cured product comprising composite fine particles composed of acicular inorganic fillers of 50 ⁇ m or less.
- An epoxy-vinyl copolymer-type cured product wherein the cured product further comprises (a) composite fine particles made of crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less.
- An epoxy-vinyl copolymerization type insulating material characterized in that the activation energy based on a 5 wt% weight reduction temperature with respect to all organic components in the cured product is 30 kcal / mol or more. Thereby, an insulating material having excellent heat resistance can be provided.
- the epoxy composition characterized by heating the resin composition to form a copolymer comprising the epoxy resin, the acid anhydride or the acid anhydride and maleic anhydride, and the polyfunctional vinyl monomer.
- the cured product can have high heat resistance without causing an increase in the viscosity of the insulating varnish. Can contribute to the improvement of workability.
- the cured resin further includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, (b) an average diameter of 0.1 ⁇ m or more and 3 ⁇ m or less, and an average length of 10 ⁇ m or more.
- a method for producing an epoxy-vinyl copolymer cured product comprising:
- the cured product further comprises (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, (c) crosslinked rubber particles having an average particle diameter of 10 nm or more and 100 nm or less, and (d) average particles.
- a method for producing an epoxy-vinyl copolymer cured product comprising composite fine particles comprising core-shell rubber particles having a diameter of 100 nm or more and 2000 nm or less.
- the cured product is further (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, and (b) an average diameter of 0.1 ⁇ m or more and 3 ⁇ m or less, and an average length of 10 ⁇ m or more.
- a method for producing an epoxy-vinyl copolymer-type cured product comprising composite fine particles composed of needle-like inorganic fillers of 50 ⁇ m or less.
- Patent Document 8 The invention content described in Patent Document 8 (hereinafter referred to as Document 8) is compared with the present invention as follows.
- an imide ring-containing epoxy resin obtained by reacting an epoxy resin and polymaleimide is an essential component for increasing heat resistance.
- the imide ring-containing epoxy resin has a problem in that the molecular weight increases as compared with the raw material epoxy resin and polymaleimide. This is dissolved in a liquid polyfunctional vinyl monomer and an acid anhydride to form a liquid varnish, but the problem is that the varnish viscosity increases as the molecular weight increases. Also, in the step of synthesizing the imide ring-containing epoxy resin, if the modification with polymaleimide proceeds too much, the epoxy resin is crosslinked and cannot be varnished, so appropriate process management is necessary, and the varnish production process is complicated. This is also a problem.
- the present invention uses an unmodified epoxy resin, a polyfunctional vinyl monomer, and an acid anhydride having an unsaturated double bond, copolymerizes in the curing process, and increases the crosslink density of the cured product to increase the heat resistance. It is a design philosophy.
- the present invention does not add a modification that increases the molecular weight of general-purpose epoxy resins, polyfunctional vinyl monomers, and acid anhydrides at the varnish stage, thereby reducing varnish viscosity and facilitating management of the varnish production process. This is advantageous.
- the present invention provides a crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, an average diameter of which is reduced with respect to cracking shrinkage due to an increase in crosslinking density and a decrease in crack resistance due to an increase in residual stress.
- Acicular inorganic filler having an average length of 10 ⁇ m or more and 50 ⁇ m or less, a crosslinked rubber particle having a particle size of 10 nm or more and 100 nm or less, a particle size of 100 nm or more and 2000 nm or less.
- the crosslinking reaction between the acid anhydride having an unsaturated double bond and a polyfunctional vinyl monomer is advanced at the same time.
- This increases the crosslink density of the curing system and improves the heat resistance. That is, the present invention achieves a low viscosity in a varnish state and a high heat resistance of a cured product by a design philosophy that simultaneously performs a curing reaction of an epoxy resin and a modification of an acid anhydride having an unsaturated double bond. It is.
- Epoxy resin Preferably, it is a bisphenol A type or bisphenol F type epoxy resin.
- a trifunctional or higher polyfunctional epoxy resin or an epoxy resin having a rigid structure may be used, but in that case, the effect of improving the heat resistance is lowered and the viscosity of the varnish is also increased.
- More preferable epoxy resins are those having an epoxy equivalent of 200 g / eq or less from the viewpoint of reducing the varnish viscosity.
- EPICLON 840 (epoxy equivalent 180 to 190 g / eq, viscosity 9000 to 11000 mPa ⁇ s / 25 ° C.) manufactured by DIC Corporation
- EPICLON 850 (epoxy equivalent 183 to 193 g / eq, viscosity 11000 to 15000 mPa ⁇ s / 25 ° C.)
- EPICLON 830 epoxy equivalent 165 to 177 g / eq, viscosity 3000 to 4000 mP ⁇ s / 25 ° C.
- jER827 epoxy equivalent 180 to 190 g / eq, viscosity 9000 to 11000 mPa ⁇ s / 25 ° C.
- Mitsubishi Chemical Corporation jER828 (epoxy equivalent 184 to 194 g / eq, viscosity 12,000 to 15000 mPa ⁇ s / 25 ° C
- Acid anhydride It is preferable to use a general-purpose liquid acid anhydride having an unsaturated double bond in the structure. By using an acid anhydride having an unsaturated double bond, copolymerization with a polyfunctional vinyl monomer occurs during the curing process. It is considered that the polyfunctional vinyl monomer and the epoxy resin are copolymerized through the acid anhydride, and the heat resistance of the cured product is improved.
- HN-2000 (acid anhydride equivalent 166 g / eq, viscosity 30-50 mPa ⁇ s / 25 ° C.)
- HN-2200 (acid anhydride equivalent 166 g / eq, viscosity manufactured by Hitachi Chemical Co., Ltd.) 50-80 mPa ⁇ s / 25 ° C.
- MHAC-P (acid anhydride equivalent 178 g / eq, viscosity 150-300 mPa ⁇ s / 25 ° C.)
- DIC Corporation EPICLON B-570H (acid anhydride equivalent 166 g / eq , Viscosity 40 mPa ⁇ s / 25 ° C.).
- the acid anhydride may be used alone or in a mixture, and 1 to 33 mol% of the total acid anhydride in the system may be maleic anhydride.
- Maleic anhydride is solid at room temperature, but in this range, it can be dissolved in a liquid acid anhydride and a polyfunctional vinyl monomer, and can be handled in the same manner as a liquid acid anhydride.
- Maleic anhydride is preferred as a component of the epoxy-vinyl copolymerization type insulating material because of its high copolymerizability with polyfunctional vinyl monomers.
- Multifunctional vinyl monomer A compound having an unsaturated double bond such as a plurality of acrylate groups, methacrylate groups, styrene groups, and allyl groups in the molecule can be used. Of these, application of a compound that is liquid at room temperature is preferred. Examples include hexanediol diacrylate (Miramer M200, viscosity 15 mPa ⁇ s / 25 ° C.), hexanediol EO-modified diacrylate (Miramer M202, viscosity 30 mPa ⁇ s / 25 ° C.), tripropylene glycol, manufactured by Toyo Chemicals Co., Ltd.
- Diacrylate (Miramer M220, viscosity 20 mPa ⁇ s / 25 ° C), trimethylolpropane triacrylate (Miramer M300, viscosity 120 mPa ⁇ s / 25 ° C), trimethylolpropane EO modified triacrylate (Miramer M3130, viscosity 65 mPa ⁇ s / 25) ° C), ditrimethylolpropane tetraacrylate (Miramer M410, viscosity 750 mPa ⁇ s / 25 ° C), diethylene glycol dimethacrylate (Mi amer M231, viscosity 20 mPa ⁇ s / 25 ° C), trimethylolpropane trimethacrylate (Miramer M301, viscosity 60 mPa ⁇ s / 25 ° C), trial allyl isocyanate (viscosity 80-110 mPa
- polyfunctional vinyl monomers are preferably used in the range of 10 to 100 parts by weight, with the total amount of epoxy resin and acid anhydride being 100 parts by weight.
- the blending amount of the polyfunctional vinyl monomer is 10 parts by weight or less, the effect of improving heat resistance is lowered, and when it exceeds 100 parts by weight, cracks are easily generated in the insulating material due to curing shrinkage. From the viewpoint of improving crack resistance, it is more preferable that the total amount of maleic anhydride and polyfunctional vinyl monomer is in the range of 30 to 100 parts by weight with respect to 100 parts by weight of the epoxy resin.
- Epoxy resin curing catalyst The epoxy-vinyl copolymer liquid resin composition of the present invention accelerates the curing reaction of an epoxy resin curing catalyst and a polyfunctional vinyl monomer that accelerates the curing reaction between the epoxy resin and the acid anhydride. Containing a radical polymerization catalyst.
- epoxy resin curing catalysts include tertiary amines such as trimethylamine, triethylamine, tetramethylbutanediamine, triethylenediamine, dimethylaminoethanol, dimethylaminopentanol, tris (dimethylaminomethyl) phenol, N-methylmorpholine, etc.
- the addition amount is preferably 0.08 parts by weight or more and 1.0 part by weight or less with respect to 100 parts by weight of the epoxy resin. In this range, the gelation time at 100 ° C. and the cured epoxy- The glass transition temperature of the vinyl copolymer insulating material can be adjusted.
- (V) Radical polymerization catalyst examples include benzoin compounds such as benzoin and benzoin methyl, acetophenone, acetophenone compounds such as 2,2-dimethoxy-2-phenylacetophenone, thioxanthone, 2, 4 -Thioxanthone compounds such as diethylthioxanthone, 4,4'-diazidochalcone, 2,6-bis (4'-azidobenzal) cyclohexanone, bisazide compounds such as 4,4'-diazidobenzophenone, azobisisobuty Azo compounds such as rhonitrile, 2,2-azobispropane, m, m′-azoxystyrene, hydrazone, 2,5-dimethyl-2,5-di (t-butylperoxy) hexane, 5-dimethyl-2,5-di (t-butylperoxy) hexyne-3
- a radical polymerization catalyst having a one-hour half-life temperature exceeding at least 100 ° C.
- examples thereof include t-butyl peroxymaleic acid (1 half life temperature of 119 ° C., perbutyl MA manufactured by NOF Corporation), n-butyl-4,4-bis (t-butylperoxy) valerate ( 1 holding half-life temperature 126.5 ° C, NOF Corporation Perhexa V), 2,5-dimethyl-2,5-di (t-butylperoxy) hexyne-3 (1 holding half-life temperature 149 -9 ° C, NOF Corporation Perhexine 25B), Dicumyl Peroxyside (1 holding half-life temperature 175.2 ° C, NOF Corporation Park Mill D), and the like.
- the addition amount is preferably in the range of 0.5 parts by weight or more and 2 parts by weight or less with respect to 100 parts by weight of the polyfunctional vinyl monomer from the viewpoint of
- the epoxy-vinyl copolymerization type liquid resin composition of the present invention includes (a) crushed crystalline silica having an average particle diameter of 5 ⁇ m or more and 50 ⁇ m or less, and (b) a diameter of 0.1 ⁇ m. Or more, 3 ⁇ m or less, a needle-like inorganic filler having a length of 10 ⁇ m or more and 50 ⁇ m or less, (c) a crosslinked rubber particle having an average particle size of 10 nm or more and 100 nm or less, and (d) an average particle size of 100 nm or more, 1 type or more of core-shell rubber particles which are 2000 nm or less are included.
- (A) Crushed crystalline silica It is preferable as a main component of the composite fine particles because it has high thermal conductivity, low thermal expansion, and is inexpensive.
- the average particle size is preferably 5 ⁇ m or more and 50 ⁇ m or less, and more preferably has a wide particle size distribution of about 0.1 ⁇ m to 100 ⁇ m.
- Examples of such silica include Hayashi Kasei Co., Ltd. SQ-H22, SQ-H18, and Tatsumori CRYSTALITE series.
- the above average particle diameter is the value disclosed by the manufacturer, and the same applies to the average diameter and average length of acicular inorganic fillers described later, and the average particle diameters of the crosslinked rubber particles and the core-shell rubber particles.
- (B) Acicular inorganic filler In addition to suppressing curing shrinkage and increasing the strength of the cured product, it contributes to the improvement of crack resistance by the combined action with rubber particle components described later.
- the size is preferably from 0.1 ⁇ m to 3 ⁇ m in average diameter and from 10 ⁇ m to 50 ⁇ m in average length from the viewpoint of suppressing an increase in varnish viscosity.
- needle-shaped inorganic fillers include Arborex Y (aluminum borate whisker, diameter 0.10 ⁇ m or more, 1 ⁇ m or less, length 10 ⁇ m or more, 30 ⁇ m or less) manufactured by Shikoku Kasei Kogyo Co., Ltd., Otsuka Chemical Co., Ltd.
- Timos N potassium titanate whisker, diameter 0.3 ⁇ m or more, 0.6 ⁇ m or less, length 10 ⁇ m or more, 20 ⁇ m or less, Ube Industries Co., Ltd.
- Mosheiji magnesium sulfate whisker, diameter 0.10 ⁇ m, length 10 ⁇ m or more, 30 ⁇ m or less
- Whiscal A calcium carbonate whisker, diameter 0.10 ⁇ m or more, 1 ⁇ m or less, length 20 ⁇ m or more, 30 ⁇ m or less
- These acicular inorganic fillers are stirred during varnish preparation. In this case, a needle-like inorganic filler that has been crushed and shortened may be included.
- Cross-linked rubber particles or core-shell rubber particles imparts flexibility and stress relaxation to the cured resin and contributes to improvement of crack resistance.
- a crosslinked rubber particle having an average particle size of 10 nm or more and 100 nm or less and a core-shell rubber particle having an average particle size of 100 nm or more and 2000 nm or less are used in combination.
- Such a rubber particle component suppresses the growth of fine cracks in a small particle size rubber particle, and further relaxes stress that cannot be alleviated in a small particle size rubber particle in a large particle size rubber particle. Minimize progress.
- the amount added can be increased, and the elastic modulus of the cured product can be reduced to suppress the occurrence of cracks. In that case, however, the viscosity of the varnish is significantly increased.
- crack resistance is improved while suppressing a significant increase in varnish viscosity by using in combination with rubber particles having different particle sizes.
- the rubber particles having a large particle size it is preferable to use core-shell rubber particles having improved dispersibility with respect to the epoxy resin.
- constituents of the rubber particles include acrylic rubber, nitrile rubber, urethane rubber, ethylene propylene rubber, styrene rubber, silicone rubber, fluorine rubber, and other general synthetic rubbers, carboxylic acid-modified, acrylic acid-modified rubber, and the like. Those whose surface or inside is modified with a carboxyl group, acid anhydrides, amines or imidazoles are generally commercially available.
- Crosslinked rubber particles Crosslinked acrylonitrile butadiene rubber particles are particularly preferred from the viewpoint of cost and heat resistance. Moreover, as the crosslinking method, it is preferable to use a crosslinking method using radiation such as gamma rays. Radiation crosslinking can provide rubber particles having higher heat resistance than chemical crosslinking using a vulcanizing agent. Furthermore, the occurrence of migration due to the vulcanizing agent is suppressed, contributing to the improvement of insulation reliability.
- radiation crosslinking can provide rubber particles having higher heat resistance than chemical crosslinking using a vulcanizing agent. Furthermore, the occurrence of migration due to the vulcanizing agent is suppressed, contributing to the improvement of insulation reliability.
- (D) Core-shell rubber particles The above-mentioned crosslinked rubber particles are used as a core, and a shell layer obtained by graft polymerization of a different polymer is provided on the surface thereof. Thereby, the dispersibility in resin can be increased.
- Rohm & Haas trade name Paraloid EXL2655 (average particle size 200 nm), Gantz Kasei Co., Ltd., trade name Staphyloid AC3355 (average particle size 100-500 nm), Zefiac F351 (average particle size 300 nm), etc. Is commercially available.
- the preferable composition range of the composite fine particles is from 83 to 94% by weight of crushed crystalline silica, 1 to 5% by weight of acicular inorganic filler, based on the total amount of the composite fine particles, from the viewpoint of reducing the varnish viscosity and improving crack resistance.
- the range is 2 to 9% by weight of the crosslinked rubber particles and 1 to 5% by weight of the core-shell rubber particles, and the composite fine particles are preferably contained in an amount of 50 to 76% by weight based on the total amount of the resin composition. Since the base material composed of epoxy resin, liquid acid anhydride, and polyfunctional vinyl monomer has a low varnish viscosity, the varnish containing composite fine particles within this range is prevented from increasing in varnish, and the efficiency of casting work is improved. You can plan.
- the epoxy-vinyl copolymerization type liquid resin composition is cured by heating and can be used as an insulator or a structure.
- the cured epoxy-vinyl copolymer insulating material is excellent in heat resistance and crack resistance, and can contribute to improvement in heat resistance reliability of various electronic and electric devices.
- an insulating material having an activation energy based on a 5 wt% weight reduction of the organic component in the cured product of 30 kcal / mol or more, more preferably 40 kcal / mol or more. This is preferable because the effect of improving the heat resistance of various devices is high.
- the curing temperature is in the range of 100 ° C. to 180 ° C.
- the curing time is selected in the range of 1 hour to 24 hours.
- curing by multi-stage heating is preferable from the viewpoint of preventing cracks accompanying curing shrinkage.
- the activation energy based on the 5% weight reduction in the present invention is one of the factors governing the heat resistance of the insulating material, and indicates a value obtained by the following method.
- thermogravimetric measurement (TGA) of the cured resin is performed under conditions of a heating rate of 5 ° C., 10 ° C., and 20 ° C./min.
- the temperature (absolute temperature T5, T10, T20) at which the organic component in the cured resin is reduced by 5 wt% under each temperature rising condition is observed.
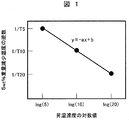
- FIG. 1 is a graph showing the relationship between the reciprocal of the 5% weight loss temperature and the logarithmic value of the rate of temperature increase in thermogravimetry of the cured product of the epoxy-vinyl copolymer resin composition according to the present invention.
- the logarithm of the rate of temperature increase (log (5), log (10), log (20)) is plotted on the horizontal axis and the reciprocal of the observed 5% weight loss temperature (1 / T5, 1 / T10, 1 / T20) is taken on the vertical axis and approximated by a straight line.
- the absolute value a of the slope of the straight line was determined.
- Heat-resistant temperature index In the present invention, the time when the weight reduction rate of the organic component in the insulating material reaches 5 wt% under a certain temperature environment is defined as the lifetime. In the present invention, the target lifetime is defined as 30 years. The temperature at which a 5 wt% weight loss occurs after 30 years was defined as the heat resistant index temperature. The heat resistant temperature index was determined from the experimentally determined 5% weight loss temperature at 230 ° C. and the activation energy. The contents are described in “(6) Calculation of heat-resistant temperature index” in Examples.
- the acid anhydride having an unsaturated double bond targeted by the present invention and liquid at room temperature has an unsaturated double bond in the structure and is liquid at room temperature.
- An acid anhydride with an acid anhydride equivalent of 166 g / eq to 178 g / eq is preferred.
- the polyfunctional vinyl monomer targeted by the present invention is a compound having a plurality of unsaturated double bonds such as an acrylate group, a methacrylate group, a styrene group, and an allyl group in the molecule.
- a compound that is liquid at normal temperature and has a lower viscosity than an epoxy resin is preferable.
- varnish generally transparent paint
- the varnish targeted by the present invention is a solventless varnish.
- the contained substance contains the above-described epoxy resin, acid anhydride, polyfunctional vinyl monomer, epoxy curing catalyst, and radical polymerization catalyst as essential components.
- an organic or inorganic filler may be contained as an additive.
- the high heat resistance method of the present invention is the curing of a conventional epoxy resin and an acid anhydride by copolymerization of an epoxy resin, an acid anhydride and a polyfunctional vinyl monomer. It refers to a technology that improves long-term heat-reliability by increasing the crosslink density over the product and delaying the thermal decomposition reaction rate.
- the present invention describes the reduction of the thermogravimetric reduction rate at high temperatures.
- One of the objects of the present invention is to reduce the viscosity of a conventional acid anhydride curable epoxy resin varnish while increasing the heat resistance of the cured product, a thermosetting resin composition using the same, and a cured product thereof. And providing an electronic / electrical device using the cured product as an insulating material and / or a structural material.
- Another object of the present invention is to reduce the viscosity of a conventional acid anhydride curable epoxy resin varnish, while providing a cured product with high heat resistance and high crack resistance, and thermosetting using the method.
- the present invention provides a conductive resin composition, a cured product thereof, and an electronic / electric device using the cured product as an insulating material or / and a structural material.
- the activation energy of 30 kcal / mol is described as an index when the product is applied. Therefore, Examples 1 and 2 in which the activation energy is increased as compared with Comparative Example 3 described later which is not copolymerized with the polyfunctional vinyl monomer are within the scope of the present invention even if the activation energy does not reach 30 kcal / mol. Is within. Similarly, Comparative Examples 1, 4 and 5 whose activation energy exceeds 30 kcal / mol are not included in the present invention.
- Equation 2 is a coefficient of an approximate expression for deriving activation energy by the Ozawa method (Source: Takeo Ozawa “Non-isothermal kinetics (1) In the case of a single elementary process”, Neto Sukutei Vol. 31, (3), pp125-132).
- the present invention relates to a technique for improving heat resistance by copolymerizing an acid anhydride having an unsaturated double bond, an epoxy resin, and a polyfunctional vinyl monomer, and a varnish and curing using the technique.
- the material composition ratios in Tables 1 to 6 are weight ratios.
- Varnish is poured into an aluminum cup having a diameter of 45 mm and a depth of 5 mm, and is heated in the air at 100 ° C./1 hour, 110 ° C./1 hour, 140 ° C./1 hour, 170 ° C./15 hours. Thus, a cured product was produced.
- Thermogravimetry (TGA) About 20 mg of resin was cut out from the cured product and used as a sample. Thermogravimetric measurement was performed in the atmosphere at each temperature rising rate of 5 ° C., 10 ° C., and 20 ° C., and the temperature at which the organic component in the cured product was reduced by 5 wt% (absolute temperatures T5, T10, T20). ) was observed.
- Equation 2 Calculation of activation energy As shown in FIG. 1, the logarithm of the rate of temperature increase (log (5), log (10), log (20)) is the reciprocal of the observed 5 wt% weight reduction temperature (1 / T5, 1 / T10, 1 / T20) is plotted on the vertical axis and approximated by a straight line.
- the activation energy (E) based on a 5% weight loss was determined by substituting the absolute value a of the slope of the straight line into Equation 2.
- FIG. 2 is a graph showing the relationship between the 5% weight reduction time and the reciprocal of deterioration temperature in thermogravimetric measurement of the cured product of the epoxy-vinyl copolymer resin composition according to the present invention. .
- the initial weight of the cured product prepared previously was observed.
- the sample was placed in a constant temperature bath at 230 ° C. under the atmosphere, and the relationship between the heating time and the weight reduction rate was examined, and the 5 wt% weight reduction time (t) of the organic component in the cured product was determined.
- the previously obtained activation energy (E) and 5 wt% weight reduction time (t) were substituted into the following formula 3 to obtain the intercept (b), and an Arrhenius plot was prepared.
- FIG. 4A is a schematic perspective view of a C-type washer made of SUS309S used for a crack resistance test.
- 4B is a plan view of FIG. 4A
- FIG. 4C is a side view of FIG. 4A
- FIG. 4D is a sectional view taken along the line ZZ of FIG. 4B.
- R represents the curvature
- ⁇ represents the diameter.
- the unit of the dimension in a figure is all mm. The same applies to FIGS. 5B and 6B described later.
- FIG. 5A is a schematic perspective view of a C-type washer made of SUS309S used for a crack resistance test.
- FIG. 5B is a cross-sectional view of the screw 6 of FIG. 5A.
- FIG. 6A is a schematic view showing a state where the C-type washer made of SUS309S shown in FIG. 6B is a cross-sectional view of the cup 7 of FIG. 6A.
- FIG. 7A is a schematic view showing a state in which a predetermined varnish is poured into a cup 7 provided with a C-type washer made of SUS309S used for a crack resistance test and subjected to a curing treatment.
- FIG. 7B is a schematic diagram of the sample prepared in FIG. 7A (a C-type washer embedded in the cured product).
- FIG. 8 is a diagram showing the conditions of thermal shock (temperature and time) in the heat-resistant crack test.
- FIG. 5A SUS309S screws 6 were adhered to both surfaces of the SUS309S C-type washer 5 shown in FIG. 4A.
- the C-type washer was subjected to a mold release treatment and installed in the center of the cup 7.
- a predetermined varnish was poured into the cup and cured in the atmosphere by multistage heating at 100 ° C./1 hour, 110 ° C./1 hour, 140 ° C./1 hour, 180 ° C./15 hours.
- FIGS. 7A and 7B a sample in which a C-type washer was embedded was taken out from a SUS309S cup, and the occurrence of cracks during curing was confirmed.
- the thermal shock shown in FIG. 8 was applied to the sample in which the C-type washer was embedded, and the lowest temperature at which no crack was generated was observed as the thermal crack resistance.
- this crack resistance test is referred to as a C-type washer test.
- Viscosity measurement The viscosity of the cured product of the epoxy-vinyl copolymerization resin composition according to the present invention was measured using an E-type viscometer manufactured by Tokimec Co., Ltd. The measurement conditions were a rotor rotational speed of 2.5 to 100 rpm and an observation temperature of 17 ° C.
- Comparative Example 1 The composition and evaluation results of the varnish of Comparative Example 1 are shown in Table 1.
- Comparative Example 1 is an example of an acid anhydride curable epoxy resin using an acid anhydride HN-5500 having no unsaturated double bond in the structure as a curing agent.
- the activation energy of the cured product was 32 kcal / mol.
- the viscosity of the varnish was 1946 mPa ⁇ s.
- Comparative Example 2 The composition and evaluation results of the varnish of Comparative Example 2 are shown in Table 1.
- Comparative Example 2 is an example in which M3130 was blended in the composition of Comparative Example 1 as a polyfunctional vinyl monomer.
- the activation energy of the cured product was 26 kcal / mol.
- a copolymer with a polyfunctional vinyl monomer does not form. Therefore, no improvement in activation energy was observed, and conversely, a decrease in activation energy was observed. From this, it was estimated that the heat resistance of the M3130 single cured product was lower than the cured product of the acid anhydride curable epoxy resin of Comparative Example 1.
- the viscosity of the varnish was 227 mPa ⁇ s.
- Comparative Example 3 is an example of an acid anhydride curable epoxy resin having an acid anhydride HN-2200 having an unsaturated double bond in the structure as a curing agent.
- the activation energy of the cured product was 25 kcal / mol.
- the viscosity of the varnish was 781 mPa ⁇ s (17 ° C., the same shall apply hereinafter).
- Examples 1 to 3 The compositions and evaluation results of the varnishes of Examples 1 to 3 are shown in Table 2.
- Examples 1 to 3 are examples in which M3130 was blended as a polyfunctional vinyl monomer in the composition of Comparative Example 3. It became clear that the activation energy increased with increasing amount of M3130.
- a copolymer of both is formed by blending a polyfunctional vinyl monomer in the system, The result that heat resistance is thought to be improved was obtained.
- the viscosity of the varnish of Example 1 was 576 mPa ⁇ s
- the viscosity of the varnish of Example 2 was 282 mPa ⁇ s
- the viscosity of the varnish of Example 3 was 205 mPa ⁇ s.
- Comparative Example 4 The composition and evaluation results of the varnish of Comparative Example 4 are shown in Table 3.
- Comparative Example 4 is an example of an acid anhydride curable epoxy resin having an acid anhydride MHAC-P having an unsaturated double bond in the structure as a curing agent.
- the activation energy of the cured product was 39 kcal / mol.
- the viscosity of the varnish was 2560 mPa ⁇ s.
- Example 4 The composition and evaluation results of the varnish of Example 4 are shown in Table 2.
- Example 4 is an example in which M3130 was blended as a polyfunctional vinyl monomer in the composition of Comparative Example 4, and 32 mol% of MHAC-P was replaced with maleic anhydride.
- the activation energy of Example 4 was 55 kcal / mol, which was much higher than that of Comparative Example 4 and Examples 1 to 3. It seemed that a lot of copolymer structure of epoxy resin and polyfunctional vinyl monomer was formed via acid anhydride by blending maleic anhydride with high radical polymerization property with M3130 which is polyfunctional vinyl monomer. .
- an acid anhydride curable epoxy resin that uses an acid anhydride having an unsaturated double bond in the structure as a curing agent
- the polyfunctional vinyl monomer and maleic anhydride are added to the system to mix the epoxy resin and the polyfunctional resin. Copolymers with vinyl monomers were formed efficiently, and the result was considered that the heat resistance was drastically improved.
- the viscosity of the varnish was 538 mPa ⁇ s at 17 ° C. and 20 mPa ⁇ s at 60 ° C.
- Comparative Example 5 The composition and evaluation results of the varnish of Comparative Example 5 are shown in Table 4.
- Comparative Example 5 is an example in which the composition of Comparative Example 1 is blended with 75 wt% of XJ-7, which is silica.
- the activation energy of the cured product was 32 kcal / mol.
- FIG. 2 shows the heat resistance evaluation results.
- the 5 wt% weight loss time for all organic components at 230 ° C. was 5 days, and the heat resistant temperature index was calculated to be 182 ° C.
- Example 5 The composition and evaluation results of the varnish of Example 5 are shown in Table 4.
- Example 5 is an example in which the composition of Example 4 was blended with 75 wt% of XJ-7, which is silica.
- the activation energy of the cured product was 55 kcal / mol.
- the 5 wt% weight loss time for all organic components at 230 ° C. was 7 days, and the heat resistant temperature index was calculated to be 202 ° C.
- the activation energy, 5 wt% weight loss time, and heat resistance temperature index all show large values, and the cured product of the epoxy-vinyl copolymer liquid resin composition has excellent heat resistance. confirmed.
- Example 6 Table 5 shows the compositions of the varnishes of Examples 6 to 8 and the results of the C-type washer test.
- Example 6 is an example in which only crushed crystalline silica is blended as a fine particle component
- Example 7 is an example in which crosslinked rubber particles and core-shell rubber particles are blended in the composition of Example 6,
- Example 8 is This is an example in which crushed crystalline silica and an acicular inorganic filler are blended as fine particle components. Due to the low viscosity of the base material of the varnish of the present invention, the liquid varnish containing each fine particle component showed good castability.
- Example 6 has high thermal conductivity and low thermal expansion
- Example 7 has high thermal conductivity
- Example 8 has high thermal conductivity. It has been shown that low thermal expansion and high strength can be imparted.
- Table 5 shows the compositions of the varnishes of Examples 9 to 12 and the results of the C-type washer test.
- Examples 9 to 12 are examples in which composite fine particles composed of crushed crystalline silica, acicular inorganic filler, crosslinked rubber particles, and core-shell rubber particles were blended as composite fine particles. By blending the composite fine particles, no cracks were observed during curing, and the thermal shock crack resistance was 0 ° C.
- Examples 13 to 17 Table 6 shows the compositions of the varnishes of Examples 13 to 17 and the results of the C-type washer test.
- Examples 13 to 17 are examples in which the total amount of maleic anhydride and M3130 as radical polymerization components was reduced to 36.84 parts by weight with respect to 100 parts by weight of the epoxy resin.
- extremely excellent thermal crack resistance of ⁇ 30 ° C. or lower was observed in the C-type washer test.
- the crack-resistant epoxy-vinyl copolymer liquid resin composition of the present invention that gives a cured product having excellent heat resistance and thermal shock crack resistance obtained results that seem to be suitable as an insulating material for electronic and electrical equipment. .
- FIG. 3 is a schematic cross-sectional view showing an example of a casting coil for a model transformer.
- 25 kg of the epoxy-vinyl copolymer liquid resin composition described in Example 17 was prepared.
- the mold of the liquid resin composition and the casting coil for the model transformer was heated to 90 ° C.
- 25 kg of the liquid resin composition was poured into the mold and vacuum degassed.
- the degassing conditions were 90 ° C., 20 Pa, and 1 hour.
- 1 is a casting resin
- 2 is a shield coil
- 3 is a secondary coil
- 4 is a primary coil. Since the transformer coil is well known, detailed description is omitted.
- the present invention is effective as a technique for increasing the heat resistance of acid anhydride curable epoxy resins used for insulating materials and structural materials for various electronic and electric devices.
- the epoxy-vinyl copolymerization type liquid resin composition of the present invention can maintain the low viscosity of the varnish and can increase the heat resistance of the cured product.
- the epoxy-vinyl copolymerization type liquid resin composition of this invention maintains the low viscosity property of a varnish, and has high heat resistance and high crack resistance of the hardened
- 1 casting resin, 2 ... shield coil, 3 ... secondary coil, 4 ... primary coil.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Epoxy Resins (AREA)
- Organic Insulating Materials (AREA)
- Macromonomer-Based Addition Polymer (AREA)
- Inorganic Insulating Materials (AREA)
Abstract
Description
前記樹脂組成物は硬化により、前記エポキシ樹脂と、前記酸無水物(A)又は前記酸無水物及び無水マレイン酸(B)と、前記多官能ビニルモノマーとからなる共重合体である硬化物を形成し得るものであることを特徴とするエポキシ‐ビニル共重合型液状樹脂組成物(I)。
(3)前記樹脂組成物が更に(a)平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ及び(c)平均粒径が10nm以上、100nm以下である架橋ゴム粒子及び(d)平均粒径が100nm以上、2000nm以下であるコアシェルゴム粒子からなる複合微粒子を含むことを特徴とするエポキシ‐ビニル共重合型液状樹脂組成物(III)。これにより熱伝導性、熱膨張性及び靭性を改善し、かつ耐熱性の優れたエポキシ‐ビニル共重型絶縁材料を得ることができる。
(4)前記樹脂組成物が更に(a)平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ及び(b)平均直径が0.1μm以上、3μm以下であり、平均長さが10μm以上、50μm以下である針状無機フィラーからなる複合微粒子を含むことを特徴とするエポキシ‐ビニル共重合型液状樹脂組成物(IV)。これにより、熱伝導性、熱膨張性及び強度を改善し、かつ耐熱性の優れたエポキシ‐ビニル共重型絶縁材料を得ることができる。
(5)前記樹脂組成物が更に(a)平均粒径が5μm以上、50μm以下の破砕状結晶質シリカからなる複合微粒子を含むことを特徴とするエポキシ‐ビニル共重合型液状樹脂組成物(V)。これにより、熱伝導性、熱膨張性を改善し、かつ耐熱性の優れたエポキシ‐ビニル共重型絶縁材料を得ることができる。
(6)前記複合微粒子は、複合微粒子全体に対して(a)破砕状結晶質シリカを83~94重量%、(b)針状無機フィラーを1~5重量%、(c)架橋ゴム粒子を2~9重量%、(d)コアシェルゴム粒子を1~5重量%含み、本複合微粒子を樹脂組成物全量に対して50~76重量%含有することを特徴とするエポキシ‐ビニル共重合型液状樹脂組成物。従来のエポキシ樹脂組成物に比べて、本発明のエポキシ樹脂と、酸無水物又は無水マレイン酸と、多官能ビニルモノマーからなる母剤のワニスの粘度は低いので、本範囲の複合微粒子を含有するワニスにおいても粘度の上昇が抑制され、脱泡作業、注型作業を効率的に実施できる。
(但し、全不飽和二重結合を有する酸無水物の当量数は樹脂組成物中の不飽和二重結合を有する全酸無水物量を、不飽和二重結合を有する酸無水物の当量で除算し、和した値であり、全エポキシ樹脂の当量数は、樹脂組成物中の全エポキシ樹脂量をエポキシ樹脂の当量で除算、和した値である。)
本構成によって、未反応のエポキシ樹脂、酸無水物の残存を抑制可能な絶縁ワニスを得ることができる。
該硬化物は前記エポキシ樹脂と、前記酸無水物又は前記酸無水物及び無水マレイン酸と、前記多官能ビニルモノマーとからなる共重合体であることを特徴とするエポキシ‐ビニル共重合型硬化物。
前記樹脂組成物を加熱して、前記エポキシ樹脂と、前記酸無水物又は前記酸無水物及び無水マレイン酸と、前記多官能ビニルモノマーとからなる共重合体を形成することを特徴とするエポキシ‐ビニル共重合型硬化物の製造方法。
(2)反応機構を比較した場合、文献8の場合は、イミド含有エポキシ樹脂が、(イ)マレイミド基を含有している場合、(ロ)マレイミド基が消失してエポキシ基のみを有する場合の2パターンが考えられる。但し、(ロ)の場合は、架橋により不溶化してしまうので、(イ)の場合のみを考えると、マレイミド基を介してエポキシ樹脂と多官能ビニルモノマーの共重合が生じる。このとき、酸無水物が二重結合を有していた場合においても、マレイミド基のラジカル重合性が高いので、マレイミド基との反応が優先して進み、酸無水物の不飽和二重結合と多官能ビニルモノマー間の反応は、十分に進行しないまま硬化するものと思われる。文献8の場合は、エポキシ樹脂を変性して、多官能化することによって架橋密度を増すという思想である。
即ち、本発明は、エポキシ樹脂の硬化反応と不飽和二重結合を有する酸無水物の変性を同時に実施する設計思想により、ワニス状態での低粘度化と硬化物の高耐熱化を実現する発明である。
(i)エポキシ樹脂:好ましくは、ビスフェノールA型またはビスフェノールF型エポキシ樹脂である。3官能以上の多官能エポキシ樹脂や剛直構造を有するエポキシ樹脂を用いても良いが、その場合は耐熱性の改善効果が低下するほか、ワニスの粘度も増加する。より好ましいエポキシ樹脂としては、エポキシ当量が200g/eq以下のものが、ワニス粘度の低減の観点から好ましい。
(v)ラジカル重合触媒:ラジカル重合触媒の例としては、ベンゾイン、ベンゾインメチルのようなベンゾイン系化合物、アセトフェノン、2、2‐ジメトキシ‐2‐フェニルアセトフェノンのようなアセトフェノン系化合物、チオキサントン、2、4‐ジエチルチオキサントンのようなチオキサンソン系化合物、4、4’‐ジアジドカルコン、2、6‐ビス(4’ ‐アジドベンザル)シクロヘキサノン、4、4’ ‐ジアジドベンゾフェノンのようなビスアジド化合物、アゾビスイソブチロニトリル、2、2‐アゾビスプロパン、m、m’‐アゾキシスチレン、ヒドラゾン、のようなアゾ化合物、2、5‐ジメチル‐2、5‐ジ(t-ブチルパーオキシ)ヘキサン、2、5‐ジメチル‐2、5‐ジ(t‐ブチルパーオキシ)ヘキシン‐3、ジクミルパーオキシドのような有機過酸化物等が挙げられる。
以下に本明細書におけるその他の用語の定義及び説明を示す。
本発明では、ある温度環境下において、絶縁材料中の有機成分の重量減少率が5wt%に達した時間を寿命時間と定義した。また、本発明では、目標の寿命時間を30年と定義した。30年後に5wt%の重量減少が生じる温度を耐熱指数温度と定義した。実験的に求めた230℃における5%重量減少温度と活性化エネルギーから耐熱温度指数を求めた。その内容を実施例の「(6)耐熱温度指数の算定」に記載した。
本発明が対象とする不飽和二重結合を有し、常温で液体である酸無水物は、構造中に不飽和二重結合を有し、常温で液体である酸無水物当量166g/eq~178g/eqの酸無水物が好ましい。
本発明が対象とする多官能ビニルモノマーは、分子内に複数のアクリレート基、メタクリレート基、スチレン基、アリル基等の不飽和二重結合を有する化合物であり、且つ、常温で液状であり、エポキシ樹脂よりも低粘度である化合物が好ましい。
本発明が対象とするワニスは、無溶剤ワニスである。含有物質は、先に記載のエポキシ樹脂、酸無水物、多官能ビニルモノマーと、エポキシ硬化触媒、ラジカル重合触媒を必須成分とする。更に添加物としては有機、無機フィラーを含有しても良い。
本発明の高耐熱化法とは、エポキシ樹脂と酸無水物と多官能ビニルモノマーの共重合化によって、従来のエポキシ樹脂と酸無水物との硬化物よりも架橋密度を増して、熱分解反応速度を遅延させることによって長期の耐熱信頼性を向上する技術を指す。特に本発明では、高温下における熱重量減少速度の低減について述べている。
上記方法は、本発明の範囲外である。これは、予備反応によって高分子量化した生成物がワニス粘度の増加を招くことを避けるためである。本発明における酸無水物-エポキシ-ビニル共重合体は、注型又は含浸後の熱硬化過程において形成される。
後述の比較例1、4に示したように従来材であっても活性化エネルギーが30kcal/mol以上の樹脂が存在する。よって本発明は活性化エネルギーが30kcal/mol以上であることに加えて、従来の酸無水物硬化型エポキシ樹脂の粘度、コストを増加させること無く、その耐熱性及び/又は耐クラック性を向上するための技術を付加したことに特徴を有する。30kcal/mol以上であるとの規定は、現製品群の主流であるF種相当の耐熱性を有する絶縁材料の値を示したものであり、製品適用時の最低条件である。
小澤法による活性化エネルギー導出の近似式の係数(出典:小澤丈夫「非等温的速度論(1)単一素過程の場合」,Netsu Sokutei Vol.31,(3), pp125-132)である。
本発明は、不飽和二重結合を有する酸無水物とエポキシ樹脂と多官能ビニルモノマーを共重合することによって耐熱性を向上する手法、およびその技術を用いたワニス、硬化物、機器を提供するものである。従って、先の活性化エネルギーの場合と同様に、耐熱温度指数の大きさによって限定される物ではなく、これも活性化エネルギーの場合と同様に、現行製品を基準として155℃が最低ラインと考えられる。その場合、後述の比較例5の樹脂組成物も耐熱指数的には合格となるが、低粘度な多官能ビニルモノマーを含有する本発明の方がワニス粘度の低減効果は高いといえる。
本発明では、材料系が同系統なので、硬化物の耐熱性の活性化エネルギーによる評価をしている。活性化エネルギーは図2のアレニウスプロットの傾きに相当しており、活性化エネルギーが大きいと傾きが大きくなり、高い耐熱性が見込まれる。しかし、材料系がまったく異なり、例えば230℃での寿命時間が極端に短い場合は、傾きが大きくても耐熱温度指数は低くなる。本発明の実施例では、230℃での寿命時間を求めているので耐熱温度指数序列に間違いは無いものと考えられる。また、求める耐熱性が、強度なのか、熱減量なのかによって、得られる活性化エネルギー、耐熱温度指数の値は大きく異なる。本発明では、熱減量に基づく耐熱性についてのみ、議論している。
以下に、実施例および比較例を示して本発明を具体的に説明する。なお、以下の実施例は本発明の具体的な説明のためのものであって、本発明の範囲がこれに限定されるものではなく、特許請求の範囲の発明思想の範囲内において自由に変更可能である。
(i)エポキシ樹脂
AER-260[旭化成エポキシ(株)製、ビスフェノールA型エポキシ樹脂、エポキシ等量約190g/eq]
(ii)酸無水物
HN-2200[日立化成(株)製、3又は4-メチル-1,2,3,6-テトラヒドロ無水フタル酸、酸無水物当量166g/eq、構造中に不飽和二重結合を有する酸無水物]
MHAC-P[日立化成(株)製、メチル-3,6-エンドメチレン-1,2,3,6-テトラヒドロ無水フタル酸、酸無水物当量178g/eq、構造中に不飽和二重結合を有する酸無水物]
HN-5500[日立化成(株)製、3又は4-メチル-ヘキサヒドロ無水フタル酸、酸無水物当量168g/eq、構造中に不飽和二重結合がない酸無水物]
無水マレイン酸[和光純薬(株)製、酸無水物当量98g/eq]
(iii)多官能ビニルモノマー
M3130[東洋ケミカルズ(株)製、トリメチロールプロパンEO変性トリアクリレート]
(iv)エポキシ樹脂硬化触媒
2E4MZ-CN[四国化成工業(株)製、1-シアノエチル-2-エチル-4-メチルイミダゾール]
(v)ラジカル重合触媒
パーヘキシン25B[日油(株)製、2、5-ジメチル-2、5-ジ(t-ブチルパーオキシ)ヘキシン-3]
(vi)破砕状結晶質シリカ
XJ-7[(株)龍森製、破砕状結晶質シリカ、平均粒径約6.3μm]
(vii)針状無機フィラー
アルボレックスY[四国化成工業(株)製、ホウ酸アルミニウムウイスカ、平均直径0.1~1.0μm、平均長さ10~30μm]
ティモスN[大塚化学(株)製、チタン酸カリウムウイスカ、平均直径0.3~0.6μm、平均長さ10~20μm]
ウィスカルA[丸尾カルシウム(株)製炭酸カルシウムウイスカ、平均直径0.1~1μm、平均長さ20~30μm]
(viii)架橋ゴム粒子
架橋アクリロニトリルブタジエンゴム粒子、平均粒径50~100nm。
コアシェルゴム粒子、平均粒径100~500nm。
S-180[日本曹達(株)製、(2-n-ブトキシカルボニルベンゾイルオキシ)トリブトキシチタン]
KBM-503[信越化学工業(株)製、3-メタクリロキシプロピルトリメトキシシラン]
KBM-403[信越化学工業(株)製、3-グリシドキシプロピルトリメトキシシラン]
(2)ワニスの調製
所定の配合比で各成分を配合し、(株)シンキー製AR-100型自転・公転式ミキサーで3分間攪拌してワニスを作製した。
ワニスをφ45mm、深さ5mmのアルミカップに注ぎ、大気中で100℃/1時間、110℃/1時間、140℃/1時間、170℃/15時間の多段階加熱により硬化物を作製した。
硬化物から約20mgの樹脂を切り出してサンプルとした。大気中で昇温速度5℃、10℃、20℃の各条件で熱重量測定を実施し、各昇温条件において硬化物中の有機成分が5wt%減量する温度(絶対温度T5、T10、T20)を観測した。
図1のように昇温速度の対数(log(5)、log(10)、log(20))を横軸に、観測した5wt%重量減少温度の逆数(1/T5、1/T10、1/T20)を縦軸にとり、直線で近似した。直線の傾きの絶対値aを式2に代入して5%重量減少に基づく活性化エネルギー(E)を求めた。
図2は本発明に係るエポキシ-ビニル共重合型樹脂組成物の硬化物の熱重量測定における5%重量減少時間と劣化温度の逆数との関係を示すグラフである。先に作製した硬化物の初期重量を観測した。次いで大気下、230℃の恒温槽内にサンプルを入れ、加熱時間と重量減少率の関係を調べ、硬化物中の有機成分の5wt%重量減少時間(t)を求めた。先に求めた活性化エネルギー(E)と5wt%重量減少時間(t)を下記式3に代入して切片(b)を求め、アレニウスプロットを作製した。これを図2に示した。本プロットから、硬化物中の有機成分が30年後に5wt%減量する温度を耐熱温度指数として求めた。
Log(t)=E/(RT)+b…式3
t:硬化物中の有機成分の重量減少が5wt%に達する時間(日)
E:活性化エネルギー(J/mol)
R:気体定数、8.3122621(J/k・mol)
b:アレニウスプロットの切片
なお、活性化エネルギーE(kcal/mol)は下記の式4により単位換算が可能である。
1kcal/mol=4184J/mol…式4
(7)耐クラック試験
図4A~図4D、図5A及び図5Bを用いて、本実施例及び比較例における耐クラック試験の方法について説明する。図4Aは耐クラック試験に用いるSUS309S製C型ワッシャーの斜視模式図である。また、図4Bは図4Aの平面図であり、図4Cは図4Aの側面図であり、図4Dは図4BのZ‐Z線断面図である。なお、図中Rは曲率を、φは直径を示す。また、図中の寸法の単位は全てmmである。後述する図5B及び図6Bにおいても同様である。
(株)トキメック製、E型粘度計を用いて、本発明に係るエポキシ-ビニル共重合型樹脂組成物の硬化物の粘度を測定した。測定条件は、ローター回転数2.5~100rpm、観測温度17℃とした。
比較例1のワニスの組成と評価結果を表1に示した。比較例1は構造中に不飽和二重結合を持っていない酸無水物HN-5500を硬化剤とする酸無水物硬化型エポキシ樹脂の例である。その硬化物の活性化エネルギーは、32kcal/molであった。ワニスの粘度は1946mPa・sであった。
比較例2のワニスの組成と評価結果を表1に示した。比較例2は、比較例1の組成物に多官能ビニルモノマーとしてM3130を配合した例である。その硬化物の活性化エネルギーは、26kcal/molであった。不飽和二重結合を持っていない酸無水物を硬化剤とする酸無水物硬化型エポキシ樹脂においては、多官能ビニルモノマーとの共重合体が生成しない。そのため活性化エネルギーの向上は認められず、逆に活性化エネルギーの低下が観測された。このことからM3130単独硬化物の耐熱性は、比較例1の酸無水物硬化型エポキシ樹脂の硬化物よりも低いものと推定された。ワニスの粘度は227mPa・sであった。
比較例3のワニスの組成と評価結果を表2に示した。比較例3は構造中に不飽和二重結合を有する酸無水物HN-2200を硬化剤とする酸無水物硬化型エポキシ樹脂の例である。その硬化物の活性化エネルギーは、25kcal/molであった。また、ワニスの粘度は781mPa・sであった(17℃、以下同じ)。
実施例1~3のワニスの組成と評価結果を表2に示した。実施例1~3は、比較例3の組成物に多官能ビニルモノマーとしてM3130を配合した例である。M3130の増量にともない活性化エネルギーが増大する傾向が明らかとなった。構造中に不飽和二重結合を有する酸無水物を硬化剤とする酸無水物硬化型エポキシ樹脂においては、系内に多官能ビニルモノマーを配合することにより、両者の共重合体が形成され、耐熱性が向上すると思われる結果を得た。実施例1のワニスの粘度は576mPa・s、実施例2のワニスの粘度は282mPa・s、実施例3のワニスの粘度は205mPa・sであった。
比較例4のワニスの組成と評価結果を表3に示した。比較例4は構造中に不飽和二重結合を有する酸無水物MHAC-Pを硬化剤とする酸無水物硬化型エポキシ樹脂の例である。その硬化物の活性化エネルギーは、39kcal/molであった。また、ワニスの粘度は2560mPa・sであった。
実施例4のワニスの組成と評価結果を表2に示した。実施例4は、比較例4の組成物に多官能ビニルモノマーとしてM3130を配合し、更にMHAC‐Pの32mol%を無水マレイン酸に置き換えた例である。実施例4の活性化エネルギーは、55kcal/molであり、比較例4や実施例1~3に比べて非常に高い値を示した。多官能ビニルモノマーであるM3130とともに、ラジカル重合性の高い無水マレイン酸を配合したことにより、酸無水物を介してエポキシ樹脂と多官能ビニルモノマーとの共重合構造が多く生成したものと思われた。構造中に不飽和二重結合を有する酸無水物を硬化剤とする酸無水物硬化型エポキシ樹脂においては、系内に多官能ビニルモノマー、無水マレイン酸を配合することにより、エポキシ樹脂と多官能ビニルモノマーとの共重合体が効率よく形成され、耐熱性が飛躍的に向上すると思われる結果を得た。ワニスの粘度は17℃において538mPa・s、60℃において20mPa・sであった。
比較例5のワニスの組成と評価結果を表4に示した。比較例5は、比較例1の組成物にシリカであるXJ-7を75wt%配合した例である。その硬化物の活性化エネルギーは、32kcal/molであった。図2に耐熱性評価結果を示した。230℃における全有機成分に対する5wt%減量時間は5日であり、耐熱温度指数は182℃と算出された。
実施例5のワニスの組成と評価結果を表4に示した。実施例5は、実施例4の組成物にシリカであるXJ-7を75wt%配合した例である。その硬化物の活性化エネルギーは、55kcal/molであった。230℃における全有機成分に対する5wt%減量時間は7日であり、耐熱温度指数は202℃と算出された。比較例5に比べて活性化エネルギー、5wt%減量時間、耐熱温度指数のいずれもが大きな値を示し、エポキシ-ビニル共重合型液状樹脂組成物の硬化物は、耐熱性が優れていることが確認された。
実施例6~8のワニスの組成とC型ワッシャー試験の結果を表5に示した。実施例6は微粒子成分として破砕状結晶質シリカのみを配合した例であり、実施例7は、実施例6の組成に架橋ゴム粒子とコアシェルゴム粒子を配合した例であり、実施例8は、微粒子成分として破砕状結晶質シリカと針状無機フィラーとを配合した例である。本発明のワニスの母材の粘度が低いことに起因して、各微粒子成分を配合した液状ワニスは良好な注型性を示した。このことから、低粘度性、高耐熱性に加えて実施例6では高熱伝導性及び低熱膨張性を、実施例7では高熱伝導性、低熱膨張性及び高靭性を、実施例8では高熱伝導性、低熱膨張性及び高い強度を付与可能であることが示された。
(実施例9~12)
実施例9~12のワニスの組成とC型ワッシャー試験の結果を表5に示した。実施例9~12は、複合微粒子として破砕状結晶質シリカと針状無機フィラーと架橋ゴム粒子とコアシェルゴム粒子からなる複合微粒子を配合した例である。複合微粒子を配合することによって硬化時のクラックが見られなくなり、冷熱衝撃クラック耐性は0℃であった。従来の液状エポキシ樹脂組成物である比較例5に記載の樹脂組成物の冷熱衝撃クラック耐性も0℃であり、エポキシ-ビニル共重合型絶縁材料の耐クラック性は、複合微粒子を配合したことにより実用上問題のないレベルに改善された。
実施例13~17のワニスの組成とC型ワッシャー試験の結果を表6に示した。実施例13~17は、エポキシ樹脂100重量部に対して、ラジカル重合成分である無水マレイン酸とM3130の総量を36.84重量部に低減した例である。本検討の組成範囲においては、C型ワッシャー試験において、-30℃以下の極めて優れた冷熱クラック耐性が認められた。耐熱性と冷熱衝撃クラック耐性が優れた硬化物を与える本発明の耐クラック性エポキシ-ビニル共重合型液状樹脂組成物は、電子、電気機器の絶縁材料として好適であると思われる結果を得た。
図3はモデル変圧器用注型コイルの一例を示す断面模式図である。実施例17に記載のエポキシ-ビニル共重合型液状樹脂組成物を25kg準備した。本液状樹脂組成物、モデル変圧器用注型コイルの型を90℃に加熱した。次いで型に液状樹脂組成物25kgを流し込み、真空脱気した。脱気条件は、90℃、20Pa、1時間とした。その後、大気中で100℃/5時間、110℃/2時間、140℃/2時間/、180℃/15時間の条件で硬化した。次いで、8時間かけて50℃に冷却し、型を外して図3に示すモデル変圧器用注型コイルを作製した。
また、本発明のエポキシ‐ビニル共重合型液状樹脂組成物は、ワニスの低粘度性を維持し、その硬化物の高耐熱化及び高耐クラック性を有する。このため、高耐熱化の要求が高い、電子機器の封止材料、モーターや発電機用コイルの含浸固着ワニス、モールド変圧器の注型ワニスとして好適である。
Claims (19)
- エポキシ当量が200g/eq以下であるエポキシ樹脂と、常温で液状の不飽和二重結合を有する酸無水物又は前記酸無水物及び無水マレイン酸と、常温で液状の多官能ビニルモノマーと、前記エポキシ樹脂と前記酸無水物又は前記酸無水物及び無水マレイン酸の硬化反応を促進するエポキシ樹脂硬化触媒と、前記多官能ビニルモノマーの硬化反応を促進するラジカル重合触媒とを含有する樹脂組成物であって、
前記樹脂組成物は、硬化により、前記エポキシ樹脂と、前記酸無水物又は前記酸無水物及び無水マレイン酸と、前記多官能ビニルモノマーとからなる共重合体である硬化物を形成し得るものであることを特徴とするエポキシ‐ビニル共重合型液状樹脂組成物。 - さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ、平均直径が0.1μm以上、3μm以下であり、平均長さが10μm以上、50μm以下である針状無機フィラー、平均粒径が10nm以上、100nm以下である架橋ゴム粒子及び平均粒径が100nm以上、2000nm以下であるコアシェルゴム粒子からなる複合微粒子を含むことを特徴とする請求項1に記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ及び平均粒径が10nm以上、100nm以下である架橋ゴム粒子及び平均粒径が100nm以上、2000nm以下であるコアシェルゴム粒子からなる複合微粒子を含むことを特徴とする請求項1に記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ及び平均直径が0.1μm以上、3μm以下であり、平均長さが10μm以上、50μm以下である針状無機フィラーからなる複合微粒子を含むことを特徴とする請求項1に記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカからなる複合微粒子を含むことを特徴とする請求項1に記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- 前記複合微粒子は、複合微粒子全量に対して前記破砕状結晶質シリカが83~94重量%、前記針状無機フィラーが1~5重量%、前記架橋ゴム粒子が2~9重量%、前記コアシェルゴム粒子が1~5重量%であり、該複合微粒子を前記樹脂組成物全量に対して50~76重量%含有することを特徴とする請求項2に記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- 前記エポキシ樹脂は、前記エポキシ当量が200g/eq以下であるビスフェノールA型エポキシ樹脂又は/及びビスフェノールF型エポキシ樹脂を含有することを特徴とする請求項1乃至請求項6のいずれかに記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- 前記酸無水物は、メチルテトラヒドロ無水フタル酸又は/及びメチルナジック酸無水物を含有することを特徴とする請求項1乃至請求項7のいずれかに記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- さらに、前記酸無水物及び無水マレイン酸の総量に対して1~33molの無水マレイン酸を含有することを特徴とする請求項1乃至請求項8のいずれかに記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- 前記酸無水物又は前記酸無水物及び無水マレイン酸の当量数と前記エポキシ樹脂の当量数との比が下記式1において0.9以上、1.0未満であり、前記エポキシ樹脂と前記酸無水物又は前記酸無水物及び無水マレイン酸の総量100重量部に対して、前記多官能ビニルモノマーを10重量部以上、100重量部以下の範囲で含有し、前記エポキシ樹脂硬化触媒を前記エポキシ樹脂100重量部に対して0.08重量部以上、1.0重量部以下の範囲で含有し、前記ラジカル重合触媒を前記多官能ビニルモノマー100重量部に対して0.5重量部以上、2重量部以下の範囲で含有することを特徴とする請求項1乃至請求項9のいずれかに記載のエポキシ‐ビニル共重合型液状樹脂組成物。
当量比=樹脂組成物中の全酸無水物の当量数÷樹脂組成物中の全エポキシ樹脂の当量数…式1
(但し、全酸無水物の当量数は樹脂組成物中の全酸無水物量を酸無水物の当量で除算し、和した値であり、全エポキシ樹脂の当量数は、樹脂組成物中の全エポキシ樹脂量をエポキシ樹脂の当量で除算し、和した値である。) - 100℃におけるゲル化時間が1時間以上であることを特徴とする請求項1乃至請求項10のいずれかに記載のエポキシ‐ビニル共重合型液状樹脂組成物。
- エポキシ樹脂の当量が200g/eq以下であるエポキシ樹脂と、常温で液状の不飽和二重結合を有する酸無水物又は前記酸無水物及び無水マレイン酸と、常温で液状の多官能ビニルモノマーと、前記エポキシ樹脂と前記酸無水物又は前記酸無水物及び無水マレイン酸との硬化反応を促進するエポキシ樹脂硬化触媒と、前記多官能ビニルモノマーの硬化反応を促進するラジカル重合触媒とを含有する樹脂組成物の硬化により形成された硬化物であって、
前記エポキシ樹脂と、前記酸無水物又は前記酸無水物及び無水マレイン酸と、前記多官能ビニルモノマーとからなる共重合体であることを特徴とするエポキシ‐ビニル共重合型硬化物。 - さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ、平均直径が0.1μm以上、3μm以下であり、平均長さが10μm以上、50μm以下である針状無機フィラー、平均粒径が10nm以上、100nm以下である架橋ゴム粒子及び平均粒径が100nm以上、2000nm以下であるコアシェルゴム粒子からなる複合微粒子を含むことを特徴とする請求項12に記載のエポキシ-ビニル共重合型硬化物。
- さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ及び平均粒径が10nm以上、100nm以下である架橋ゴム粒子及び平均粒径が100nm以上、2000nm以下であるコアシェルゴム粒子からなる複合微粒子を含むことを特徴とする請求項12に記載のエポキシ‐ビニル共重合型硬化物。
- さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカ及び平均直径が0.1μm以上、3μm以下であり、平均長さが10μm以上、50μm以下である針状無機フィラーからなる複合微粒子を含むことを特徴とする請求項12に記載のエポキシ‐ビニル共重合型硬化物。
- さらに、平均粒径が5μm以上、50μm以下の破砕状結晶質シリカからなる複合微粒子を含むことを特徴とする請求項12に記載のエポキシ‐ビニル共重合型硬化物。
- 全有機成分に対する5wt%重量減少温度に基づく活性化エネルギーが30kcal/mol以上であることを特徴とする請求項12乃至請求項16のいずれかに記載のエポキシ-ビニル共重合型硬化物。
- 請求項12乃至請求項17のいずれかに記載のエポキシ-ビニル共重合型硬化物を絶縁体又は/及び構造材料としたことを特徴とする電子・電気機器。
- エポキシ当量が200g/eq以下であるエポキシ樹脂と、常温で液状の不飽和二重結合を有する酸無水物又は前記酸無水物及び無水マレイン酸と、常温で液状の多官能ビニルモノマーと、前記エポキシ樹脂と前記酸無水物又は前記酸無水物及び無水マレイン酸との硬化反応を促進するエポキシ樹脂硬化触媒と、前記多官能ビニルモノマーの硬化反応を促進するラジカル重合触媒とを含有する樹脂組成物を調製し、前記樹脂組成物を加熱して、前記エポキシ樹脂と、前記酸無水物又は前記酸無水物及び前記無水マレイン酸と、前記多官能ビニルモノマーとからなる共重合体を形成することを特徴とするエポキシ‐ビニル共重合型硬化物の製造方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020147021464A KR101613385B1 (ko) | 2012-05-16 | 2013-02-19 | 에폭시-비닐 공중합형 액상 수지 조성물, 그의 경화물 및 당해 경화물을 이용한 전자·전기 기기 및 당해 경화물의 제조 방법 |
| CN201380007977.XA CN104093763A (zh) | 2012-05-16 | 2013-02-19 | 环氧-乙烯基共聚型液态树脂组合物、其固化物和使用该固化物的电子、电气机器以及该固化物的制造方法 |
| US14/374,778 US9493605B2 (en) | 2012-05-16 | 2013-02-19 | Epoxy-vinyl copolymerization type liquid resin composition, cured product of the same, electronic/electric apparatus using the cured product, and method of producing the cured product |
| EP13790677.2A EP2851382A4 (en) | 2012-05-16 | 2013-02-19 | EPOXY / VINYL COPOLYMER TYPE LIQUID RESIN COMPOSITION, ARTICLE CURED THEREFROM, ELECTRICAL / ELECTRONIC DEVICE USING THE CURED ARTICLE, AND METHOD FOR PRODUCING THE CURED ARTICLE |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-112461 | 2012-05-16 | ||
| JP2012112461 | 2012-05-16 | ||
| JP2012-254899 | 2012-11-21 | ||
| JP2012254899A JP5738261B2 (ja) | 2012-05-16 | 2012-11-21 | エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物、製造方法及び硬化物を用いた絶縁材料、電子・電気機器 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013172064A1 true WO2013172064A1 (ja) | 2013-11-21 |
Family
ID=49583489
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/053920 WO2013172064A1 (ja) | 2012-05-16 | 2013-02-19 | エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物及び当該硬化物を用いた電子・電気機器並びに当該硬化物の製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US9493605B2 (ja) |
| EP (1) | EP2851382A4 (ja) |
| JP (1) | JP5738261B2 (ja) |
| KR (1) | KR101613385B1 (ja) |
| CN (1) | CN104093763A (ja) |
| WO (1) | WO2013172064A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015132990A1 (ja) * | 2014-03-05 | 2015-09-11 | 株式会社 日立産機システム | エポキシ樹脂組成物およびそれを用いた電力機器 |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5738261B2 (ja) | 2012-05-16 | 2015-06-17 | 株式会社日立産機システム | エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物、製造方法及び硬化物を用いた絶縁材料、電子・電気機器 |
| WO2015062057A1 (en) * | 2013-10-31 | 2015-05-07 | Dow Global Technologies Llc | Curable compositions which form interpenetrating polymer networks |
| JP2016033197A (ja) * | 2014-07-31 | 2016-03-10 | 京セラケミカル株式会社 | 注形用エポキシ樹脂組成物、点火コイル及び点火コイルの製造方法 |
| JP2016121258A (ja) * | 2014-12-25 | 2016-07-07 | 株式会社日立製作所 | エポキシ樹脂組成物、およびそれを用いた硬化物、電力機器、真空遮断機 |
| KR20160140258A (ko) | 2015-05-29 | 2016-12-07 | 삼성전기주식회사 | 패키지용 수지 조성물, 이를 이용한 절연 필름 및 인쇄회로기판 |
| JP2017115132A (ja) * | 2015-12-22 | 2017-06-29 | 信越化学工業株式会社 | 液状エポキシ樹脂組成物 |
| JP6563823B2 (ja) * | 2016-01-18 | 2019-08-21 | 株式会社日立産機システム | 高電圧機器用モールド樹脂材料および高電圧機器の製造方法 |
| JP6909206B2 (ja) * | 2016-04-13 | 2021-07-28 | 株式会社カネカ | 強靭化エポキシ樹脂組成物、強靭化エポキシ樹脂組成物の製造方法、及び硬化物 |
| WO2018211799A1 (ja) * | 2017-05-17 | 2018-11-22 | 三菱電機株式会社 | 液状熱硬化性樹脂組成物、樹脂硬化物の製造方法、固定子コイル及び回転電機 |
| CN113227193A (zh) * | 2019-01-07 | 2021-08-06 | 日东新兴有限公司 | 树脂组合物、该树脂组合物的制造方法、以及导热性片 |
| JPWO2022039121A1 (ja) * | 2020-08-17 | 2022-02-24 | ||
| CN115612064A (zh) * | 2022-11-14 | 2023-01-17 | 国网辽宁省电力有限公司阜新供电公司 | 一种用于辐射降温涂料固化用的粘结剂及辐射降温涂料 |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06233486A (ja) | 1993-01-29 | 1994-08-19 | Hitachi Ltd | 絶縁電気線輪、回転電機及びその製造方法 |
| JPH08109316A (ja) | 1994-10-12 | 1996-04-30 | Meidensha Corp | 耐熱性滴下含浸樹脂 |
| JPH09249740A (ja) * | 1996-03-15 | 1997-09-22 | Toshiba Corp | 熱硬化性樹脂組成物 |
| JPH09316167A (ja) | 1996-05-30 | 1997-12-09 | Hitachi Ltd | 熱硬化性樹脂組成物,電機絶縁線輪,回転電機及びその製造方法 |
| JPH10505376A (ja) | 1994-09-08 | 1998-05-26 | アクゾ ノーベル ナムローゼ フェンノートシャップ | エチレン性不飽和無水物とビニル化合物との共重合体を含む、アリルを含むエポキシ樹脂組成物 |
| JPH11255864A (ja) * | 1998-03-09 | 1999-09-21 | Toshiba Corp | 液状エポキシ樹脂組成物および樹脂封止型半導体装置 |
| JP2003277471A (ja) * | 2002-03-27 | 2003-10-02 | Toray Ind Inc | エポキシ樹脂組成物、プリプレグおよび炭素繊維強化複合材料 |
| JP2004203955A (ja) | 2002-12-24 | 2004-07-22 | Mitsubishi Electric Corp | 液状熱硬化性樹脂組成物 |
| JP2010193673A (ja) | 2009-02-20 | 2010-09-02 | Hitachi Ltd | ドライマイカテープ、それを用いた電気絶縁線輪,固定子コイル及び回転電機 |
| JP2011001424A (ja) | 2009-06-17 | 2011-01-06 | Hitachi Industrial Equipment Systems Co Ltd | 電気機器用絶縁注型樹脂及びこれを用いた高電圧電気機器 |
| JP2011057734A (ja) | 2009-09-07 | 2011-03-24 | Hitachi Electric Systems Ltd | 高靭性高熱伝導性硬化性樹脂組成物、その硬化物及びモールド電機機器 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4503200A (en) * | 1982-07-19 | 1985-03-05 | Shell Oil Company | Heat curable polyepoxide resin blends |
| JPS6028422A (ja) * | 1983-07-26 | 1985-02-13 | Mitsubishi Electric Corp | 低粘度エポキシ含浸樹脂の製造方法 |
| JPS6112721A (ja) * | 1984-06-29 | 1986-01-21 | Mitsubishi Electric Corp | 熱硬化性樹脂組成物 |
| JPS61148230A (ja) * | 1984-12-21 | 1986-07-05 | Hitachi Chem Co Ltd | 熱硬化性樹脂組成物 |
| JPS6243412A (ja) * | 1985-08-21 | 1987-02-25 | Nippon Paint Co Ltd | 高エネルギ−線硬化樹脂組成物 |
| WO1992012900A1 (de) | 1991-01-26 | 1992-08-06 | Bühler AG Maschinenfabrik | Verfahren zur automatischen sackbereitstellung und sackanhängevorrichtung |
| JP2001244376A (ja) * | 2000-02-28 | 2001-09-07 | Hitachi Ltd | 半導体装置 |
| JP2003255864A (ja) | 2002-03-05 | 2003-09-10 | Cap Corporation | 内照式ディスプレイ装置 |
| JP2003277579A (ja) * | 2002-03-22 | 2003-10-02 | Nippon Kayaku Co Ltd | 高耐熱エポキシ樹脂組成物及びその硬化物 |
| JP3975984B2 (ja) * | 2003-08-11 | 2007-09-12 | 日立化成工業株式会社 | 銅と接着状態にある絶縁基材 |
| JP2009185232A (ja) * | 2008-02-08 | 2009-08-20 | Sumitomo Bakelite Co Ltd | 樹脂組成物、プリプレグおよび積層板 |
| JP5738261B2 (ja) | 2012-05-16 | 2015-06-17 | 株式会社日立産機システム | エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物、製造方法及び硬化物を用いた絶縁材料、電子・電気機器 |
-
2012
- 2012-11-21 JP JP2012254899A patent/JP5738261B2/ja active Active
-
2013
- 2013-02-19 US US14/374,778 patent/US9493605B2/en active Active
- 2013-02-19 WO PCT/JP2013/053920 patent/WO2013172064A1/ja active Application Filing
- 2013-02-19 EP EP13790677.2A patent/EP2851382A4/en not_active Withdrawn
- 2013-02-19 CN CN201380007977.XA patent/CN104093763A/zh active Pending
- 2013-02-19 KR KR1020147021464A patent/KR101613385B1/ko active IP Right Grant
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06233486A (ja) | 1993-01-29 | 1994-08-19 | Hitachi Ltd | 絶縁電気線輪、回転電機及びその製造方法 |
| JPH10505376A (ja) | 1994-09-08 | 1998-05-26 | アクゾ ノーベル ナムローゼ フェンノートシャップ | エチレン性不飽和無水物とビニル化合物との共重合体を含む、アリルを含むエポキシ樹脂組成物 |
| JPH08109316A (ja) | 1994-10-12 | 1996-04-30 | Meidensha Corp | 耐熱性滴下含浸樹脂 |
| JPH09249740A (ja) * | 1996-03-15 | 1997-09-22 | Toshiba Corp | 熱硬化性樹脂組成物 |
| JPH09316167A (ja) | 1996-05-30 | 1997-12-09 | Hitachi Ltd | 熱硬化性樹脂組成物,電機絶縁線輪,回転電機及びその製造方法 |
| JPH11255864A (ja) * | 1998-03-09 | 1999-09-21 | Toshiba Corp | 液状エポキシ樹脂組成物および樹脂封止型半導体装置 |
| JP2003277471A (ja) * | 2002-03-27 | 2003-10-02 | Toray Ind Inc | エポキシ樹脂組成物、プリプレグおよび炭素繊維強化複合材料 |
| JP2004203955A (ja) | 2002-12-24 | 2004-07-22 | Mitsubishi Electric Corp | 液状熱硬化性樹脂組成物 |
| JP2010193673A (ja) | 2009-02-20 | 2010-09-02 | Hitachi Ltd | ドライマイカテープ、それを用いた電気絶縁線輪,固定子コイル及び回転電機 |
| JP2011001424A (ja) | 2009-06-17 | 2011-01-06 | Hitachi Industrial Equipment Systems Co Ltd | 電気機器用絶縁注型樹脂及びこれを用いた高電圧電気機器 |
| JP2011057734A (ja) | 2009-09-07 | 2011-03-24 | Hitachi Electric Systems Ltd | 高靭性高熱伝導性硬化性樹脂組成物、その硬化物及びモールド電機機器 |
Non-Patent Citations (2)
| Title |
|---|
| See also references of EP2851382A4 |
| TAKEO OZAWA: "Non-isothermal Kinetics (1) Single Elementary Process", NETSU SOKUTEI, vol. 31, no. 3, pages 125 - 132 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015132990A1 (ja) * | 2014-03-05 | 2015-09-11 | 株式会社 日立産機システム | エポキシ樹脂組成物およびそれを用いた電力機器 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2851382A4 (en) | 2016-01-20 |
| KR20140119075A (ko) | 2014-10-08 |
| JP5738261B2 (ja) | 2015-06-17 |
| JP2013256637A (ja) | 2013-12-26 |
| EP2851382A1 (en) | 2015-03-25 |
| CN104093763A (zh) | 2014-10-08 |
| US20140378580A1 (en) | 2014-12-25 |
| KR101613385B1 (ko) | 2016-04-18 |
| US9493605B2 (en) | 2016-11-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5738261B2 (ja) | エポキシ‐ビニル共重合型液状樹脂組成物、その硬化物、製造方法及び硬化物を用いた絶縁材料、電子・電気機器 | |
| JP6310730B2 (ja) | エポキシ樹脂組成物およびそれを用いた電力機器 | |
| JP6405867B2 (ja) | 樹脂ペースト組成物及び半導体装置 | |
| JP5756082B2 (ja) | ダイレクトオーバーモールディング | |
| WO2011158753A1 (ja) | 樹脂ペースト組成物 | |
| JP2016501922A (ja) | 高温用途用の強靭化された硬化可能エポキシ組成物 | |
| JP7219123B2 (ja) | モールド機器 | |
| JP7100651B2 (ja) | ペースト組成物、半導体装置及び電気・電子部品 | |
| JP2015168687A (ja) | コイル含浸用樹脂組成物及びコイル装置 | |
| JP6563823B2 (ja) | 高電圧機器用モールド樹脂材料および高電圧機器の製造方法 | |
| JP2003041123A (ja) | 熱硬化性樹脂組成物、その製造方法及び懸濁液状混合物 | |
| JP6279161B1 (ja) | 硬化性組成物およびその硬化物ならびに回転機 | |
| WO2017104726A1 (ja) | モールドワニス、樹脂硬化物、電力機器及び真空遮断器 | |
| JP2016204530A (ja) | 熱硬化性樹脂組成物、固定子コイル及び回転電機 | |
| JP2002003699A (ja) | エポキシ樹脂組成物、その製造方法及びエポキシ樹脂硬化物 | |
| JP5359934B2 (ja) | リアクトル | |
| JP2002194057A (ja) | 熱硬化性樹脂組成物 | |
| JP4114048B2 (ja) | エポキシ樹脂組成物及びその製法 | |
| JP2016117869A (ja) | 半導体接着用樹脂組成物及び半導体装置 | |
| JP2006036862A (ja) | 電気絶縁性注型用エポキシ樹脂組成物及び硬化物、並びに熱硬化性接着剤組成物 | |
| JPH06248056A (ja) | エポキシ樹脂組成物 | |
| WO2016103842A1 (ja) | エポキシ樹脂組成物、およびそれを用いた硬化物、電力機器、真空遮断機 | |
| JP2016050221A (ja) | 硬化性エポキシ樹脂組成物 | |
| WO2022162805A1 (ja) | 絶縁樹脂組成物、硬化物、回転機用コイル、および回転機 | |
| KR102644123B1 (ko) | 열 전도성 포팅 조성물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13790677 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013790677 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14374778 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20147021464 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |