WO2012011286A1 - 半有機絶縁被膜付き電磁鋼板 - Google Patents
半有機絶縁被膜付き電磁鋼板 Download PDFInfo
- Publication number
- WO2012011286A1 WO2012011286A1 PCT/JP2011/004123 JP2011004123W WO2012011286A1 WO 2012011286 A1 WO2012011286 A1 WO 2012011286A1 JP 2011004123 W JP2011004123 W JP 2011004123W WO 2012011286 A1 WO2012011286 A1 WO 2012011286A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- steel sheet
- semi
- silica
- insulating coating
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/61—Additives non-macromolecular inorganic
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D163/00—Coating compositions based on epoxy resins; Coating compositions based on derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/08—Anti-corrosive paints
- C09D5/082—Anti-corrosive paints characterised by the anti-corrosive pigment
- C09D5/084—Inorganic compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/66—Additives characterised by particle size
- C09D7/67—Particle size smaller than 100 nm
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/66—Additives characterised by particle size
- C09D7/68—Particle size between 100-1000 nm
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/70—Additives characterised by shape, e.g. fibres, flakes or microspheres
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C22/00—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals
- C23C22/73—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals characterised by the process
- C23C22/74—Chemical surface treatment of metallic material by reaction of the surface with a reactive liquid, leaving reaction products of surface material in the coating, e.g. conversion coatings, passivation of metals characterised by the process for obtaining burned-in conversion coatings
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F1/00—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties
- H01F1/01—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials
- H01F1/03—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity
- H01F1/12—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials
- H01F1/14—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials metals or alloys
- H01F1/16—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials metals or alloys in the form of sheets
- H01F1/18—Magnets or magnetic bodies characterised by the magnetic materials therefor; Selection of materials for their magnetic properties of inorganic materials characterised by their coercivity of soft-magnetic materials metals or alloys in the form of sheets with insulating coating
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C2222/00—Aspects relating to chemical surface treatment of metallic material by reaction of the surface with a reactive medium
- C23C2222/20—Use of solutions containing silanes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/25—Web or sheet containing structurally defined element or component and including a second component containing structurally defined particles
- Y10T428/259—Silicic material
Definitions
- the present invention has no corrosion resistance and water resistance deterioration even without the inclusion of a chromium compound, is excellent in powder blowing resistance, scratch resistance, sticking property, Tig weldability and punching property, and has a uniform coating appearance after annealing.
- the present invention relates to a magnetic steel sheet with a semi-organic insulating coating that is excellent in properties.
- various characteristics such as convenience during processing and storage, and stability during use are required for insulating coatings on electrical steel sheets used for motors and transformers. Since electrical steel sheets are used for various applications, various insulating coatings have been developed according to the applications. When a magnetic steel sheet is punched, sheared, bent, etc., the magnetic properties deteriorate due to residual strain. In order to eliminate this, strain relief smelting is often performed at a temperature of about 700 to 800 ° C. Therefore, in this case, the insulating coating must be able to withstand strain relief annealing.
- Insulation coating is roughly divided (1) An inorganic coating that emphasizes weldability and heat resistance and resists strain relief annealing, (2) Resin-containing inorganic coating (ie, semi-organic coating) that can withstand strain relief annealing to achieve both punchability and weldability, (3) Although it is classified into three types of organic coatings that cannot be strain-relieved and annealed for special applications, the coatings containing inorganic components shown in (1) and (2) above can withstand strain relief annealing as general-purpose products. Yes, both contained chromium compounds.
- Patent Document 1 discloses that a dichromate-based aqueous solution containing at least one divalent metal has a vinyl acetate / veova ratio of 90/10 to 90% as an organic resin with respect to 100 parts by weight of CrO 3 in the aqueous solution.
- a treatment liquid containing a resin emulsion having a ratio of 40/60 in a proportion of 5 to 120 parts by weight of resin solids and an organic reducing agent in a ratio of 10 to 60 parts by weight is applied to the surface of the base iron plate and baked by a conventional method.
- An electrical steel sheet having an electrical insulating coating obtained by applying is described.
- Patent Document 2 discloses an insulating coating that does not contain chromium and has good punchability, and is composed of resin and colloidal silica (alumina-containing silica). Are listed. Further, an insulating coating comprising one or more of colloidal silica, alumina sol, and zirconia sol and containing a water-soluble or emulsion resin is described in Patent Document 3, and is mainly composed of a phosphate containing no chromium. Patent Document 4 discloses an insulating coating containing bismuth.
- the electrical steel sheet with an insulating coating that does not contain these chromium compounds has a problem in that the bonding between inorganic substances is relatively weak and the corrosion resistance is inferior as compared with the case containing chromium compounds. Further, when the back tension is applied by rubbing the surface of the steel sheet with felt in slit processing (use of a tension pad), there is a problem of powder blowing. Furthermore, there is a problem that the film becomes weak after the strain relief annealing, and scratches are easily generated.
- the inventors have made extensive studies to solve the above problems, and as a result, a compound containing a Zr compound and a Si compound containing plate-like silica, as well as a B compound, as an inorganic component in the semi-organic coating.
- a compound containing a Zr compound and a Si compound containing plate-like silica, as well as a B compound as an inorganic component in the semi-organic coating.
- the gist configuration of the present invention is as follows. 1.
- a nitric acid compound in terms of NO 3
- a silane coupling agent in terms of solid content
- a phosphorus compound in terms of P 2 O 5
- the present invention is an electrical steel sheet having a semi-organic insulating coating made of an inorganic component and an organic resin on its surface, (1) Zr compound and Si compound containing plate-like silica are each in a ratio in the dry film, Zr compound (ZrO 2 conversion): 20 to 70% by mass, Si compound containing plate-like silica (SiO 2 conversion): 10-50% by mass, (2) If necessary, the B compound is further added in a ratio in the dry film to the B compound (in terms of B 2 O 3 ): 0.1 to 5% by mass, (3) If necessary, dry one or more selected from nitric acid compounds (NO 3 conversion), silane coupling agents (solid content conversion) and phosphorus compounds (P 2 O 5 conversion).
- the remainder is a semi-organic insulating coated electrical steel sheet that is substantially an organic resin (that is, permitting the inclusion of impurities and known additives), and the plate-like silica has an average particle diameter of 10 to 600 nm.
- the aspect ratio (average length / average thickness ratio) of the plate-like silica is preferably 2 to 400.
- the present invention it is needless to say that it is excellent in various properties such as powder blowing resistance, scratch resistance, sticking property, Tig weldability and punchability, and even if it does not contain a chromium compound, water resistance and It is possible to obtain an electrical steel sheet with a semi-organic insulating coating that is not deteriorated in corrosion resistance and is excellent in the uniformity of the coating appearance after annealing.
- the present invention will be specifically described below. First, in the present invention, the reason for limiting the blending ratio of the Z compound, which is an inorganic component of the semi-organic coating, and the Si compound including plate-like silica and further the B compound to the above range will be described. In addition, the mass% of these components is a ratio in a dry film.
- Zr compound 20 to 70% by mass in terms of ZrO 2
- the Zr compound has a strong binding force with oxygen and can be strongly bonded to an oxide, hydroxide, or the like on the Fe surface.
- the Zr compound has three or more bonds, it is possible to form a tough film without using chromium by forming a network with Zr or other inorganic compounds.
- the ratio of the Zr compound in the dry film is less than 20% by mass in terms of ZrO 2 , the adhesiveness deteriorates, and not only the corrosion resistance and powder blowing resistance deteriorate, but also the appearance after annealing caused by the Si compound. Degradation occurs.
- the Zr compound is limited to the range of 20 to 70% by mass in terms of ZrO 2 .
- Zr compounds include zirconium acetate, zirconium propionate, zirconium oxychloride, zirconium nitrate, ammonium zirconium carbonate, zirconium carbonate potassium, hydroxy zirconium chloride, zirconium sulfate, zirconium phosphate, sodium zirconium phosphate, hexafluoride.
- Zr compounds include potassium zirconate, tetranormal propoxyzirconium, tetranormalbutoxyzirconium, zirconium tetraacetylacetonate, zirconium tributoxyacetylacetonate, zirconium tributoxy systemate and the like. These can be used alone or in combination of two or more.
- the plate-like silica of the present invention is also called leaf-like silica or scaly silica, and has a layered silicic acid structure in which many thin layers of SiO 2 are laminated, and many of them have non-crystalline or microcrystalline properties.
- Such plate-like silica has a layered form as compared with general silica particles such as colloidal silica, and is excellent in corrosion substance permeation suppression, and further has excellent adhesion due to a large number of hydroxyl groups. Excellent softness due to its softness. For this reason, it is more effective in improving corrosion resistance and punchability.
- Plate-like silica can be obtained by preparing aggregated particles in which primary particles of a thin layer are laminated and pulverizing the aggregated particles.
- the average particle diameter of the plate-like silica is preferably about 10 to 600 nm. More preferably, it is in the range of 100 to 450 nm.
- the aspect ratio (length / thickness ratio) of the plate-like silica is preferably about 2 to 400. More preferably, it is in the range of 10-100.
- the length of the plate-like silica (length in the major axis direction) can be obtained by observing the coating surface with SEM. it can.
- the thickness of the plate-like silica can be determined by SEM observation of a cross section such as freeze fracture or FIB (Focused Ion Beam). Therefore, the length and thickness of the plate-like silica are obtained at several places by SEM observation, and the aspect ratio (average length / average thickness ratio) is obtained from the average value thereof.
- the silica is elliptical or polygonal
- the SEM is observed at several places to obtain the average particle diameter in the plane and the average thickness in the cross section, and the aspect ratio is calculated by (average particle diameter) / (average thickness). Find the ratio.
- the Si compound other than the plate-like silica examples include colloidal silica, fumed silica, alkoxysilane, and siloxane, which contribute to improvement of corrosion resistance and adhesion after strain relief annealing.
- colloidal silica and fumed silica are spherical or an ellipsoid having an aspect ratio of less than 2, and alkoxysilane and siloxane are amorphous. If the blending ratio of the plate-like silica in the entire Si compound is less than 50% by mass, good corrosion resistance and punching performance as aimed by the present invention cannot be obtained. Therefore, the blending ratio of the plate-like silica is 50% by mass. The above is preferable.
- all the Si compounds may be plate-like silica.
- a particularly preferred range is from 50 to 100% by mass.
- the mixing ratio of the plate-like silica is determined by measuring the SiO 2 amount (B) of the plate-like silica portion and the SiO 2 amount (A) in other regions, and B / (A + B) ⁇ 100 It can ask for. Further, the amount of SiO 2 in each region can be obtained from the average Si concentration obtained by EDS or the like and the region area.
- the Si compound containing plate-like silica described above is useful for solving the problem when a Zr compound is added alone. That is, when the Zr compound was used alone, the corrosion resistance and powder blowing resistance deteriorated, and the scratch resistance on the strain relief annealed plate tended to deteriorate significantly. By doing so, the powder blowing resistance and scratch resistance can be greatly improved.
- the content of the Si compound in the dry film is less than 10% by mass in terms of SiO 2 , sufficient corrosion resistance cannot be obtained, while if it exceeds 50% by mass, the powder blowing resistance deteriorates. Since scratch resistance in the strain relief annealed plate also deteriorates, the Si compound is limited to the range of 10 to 50% by mass.
- a B compound in addition to the above-described Zr compound and Si compound containing plate-like silica, a B compound can be further contained as an inorganic component.
- B compound 0.1 to 5% by mass in terms of B 2 O 3
- the B compound advantageously contributes to solving the problem when the Zr compound is added alone. That is, when the Zr compound was added alone, the corrosion resistance and powder blowing resistance deteriorated, and the scratch resistance on the strain relief annealed plate tended to deteriorate significantly. The reason for this is considered that the Zr compound alone has a large volume shrinkage when baked, so that coating cracks are likely to occur, and a portion where the substrate is partially exposed is generated.
- the film cracking that has occurred in the case of Zr alone can be effectively alleviated, and the powder blowing resistance can be remarkably improved.
- the ratio of the B compound in the dry film is 0.1% by mass or more in terms of B 2 O 3 , the addition effect is obtained, and if it is 5% by mass or less, unreacted substances in the film remain. Therefore, the defect (stick) in which the films are fused after the strain relief annealing does not occur, so that the B compound is preferably in the range of 0.1 to 5% by mass in terms of B 2 O 3 .
- B compounds include boric acid, orthoboric acid, metaboric acid, tetraboric acid, sodium metaborate, sodium tetraborate and the like, and these can be used alone or in combination.
- generates a borate ion may be sufficient, and the borate ion may superpose
- nitric acid compounds in addition to the above-described inorganic components, one or more selected from the following nitric acid compounds, silane coupling agents, and phosphorus compounds are used in a ratio of 30% by mass or less in the dry film. It can also be contained.
- the ratios of nitrate compound, silane coupling agent and phosphorus compound in the dry film are shown in NO 3 conversion (nitric acid compound), solid content conversion (silane coupling agent) and P 2 O 5 conversion (phosphorus compound), respectively. It is a thing.
- Such nitric acid compounds, silane coupling agents and phosphorus compounds contribute effectively to the improvement of corrosion resistance.
- the ratio in the dry film is 30% by mass or less in total, unreacted substances remain in the film. And the water resistance is not lowered, so the total content is preferably 30% by mass or less.
- Nitrite system Nitrous acid (HNO 2 ), potassium nitrite, calcium nitrite, silver nitrite, sodium nitrite, barium nitrite, ethyl nitrite, isoamyl nitrite, isobutyl nitrite, isopropyl nitrite, nitrous acid -t- Butyl, nitrite-n-butyl, nitrite-n-propyl.
- HNO 2 Nitrous acid
- potassium nitrite calcium nitrite
- silver nitrite sodium nitrite
- barium nitrite barium nitrite
- ethyl nitrite isoamyl nitrite
- isobutyl nitrite isopropyl nitrite
- nitrous acid -t- Butyl nitrite-n-butyl, nitrite-n-propyl.
- silane coupling agent As a silane coupling agent, the following are advantageously adapted. ⁇ Vinyl vinyl trichlorosilane, vinyltrimethoxysilane, vinyltriethoxysilane. Epoxy type 2- (3,4-epoxycyclohexyl) ethyltrimethoxysilane, 3-glycidoxypropyltrimethoxysilane, 3-glycidoxypropylmethyldiethoxysilane, 3-glycidoxypropyltriethoxysilane. ⁇ Styryl p-styryltrimethoxysilane.
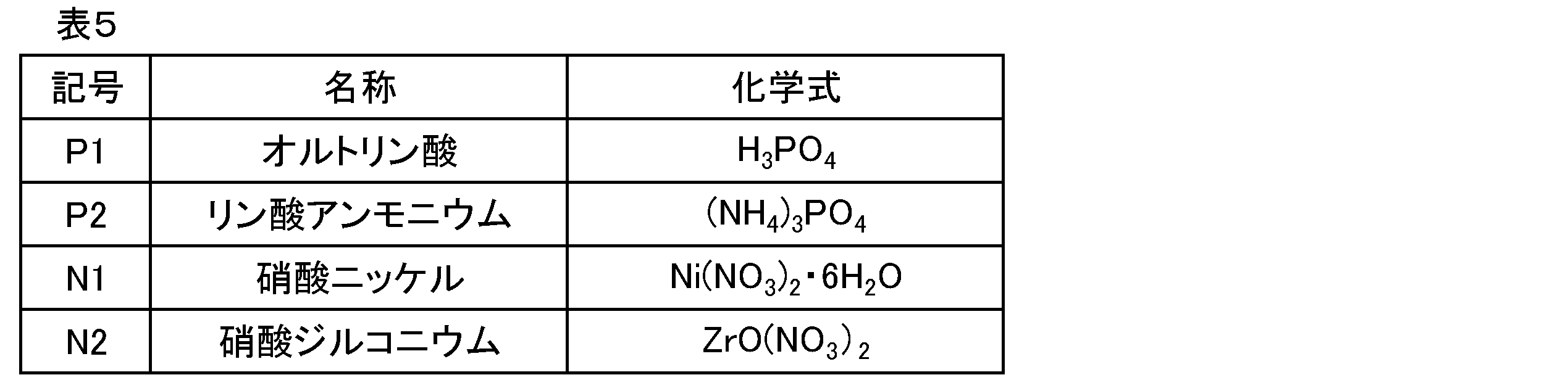
- phosphoric acid and phosphate as shown below are advantageously suitable as the phosphorus compound.
- Hf, HfO 2 , TiO 2 , Fe 2 O 3, and the like may be mixed as impurities in the inorganic component. If the total amount of these impurities is 1% by mass or less in the dry film, No particular problem arises.
- the organic resin is preferably blended at a rate of 5 to 40% by mass so that the content of the inorganic component as described above in the dry film is 60 to 95% by mass.
- the organic resin is not particularly limited, and any conventionally known organic resin is advantageously adapted. Examples thereof include aqueous resins (emulsion, dispersion, water-soluble) such as acrylic resin, alkyd resin, polyolefin resin, styrene resin, vinyl acetate resin, epoxy resin, phenol resin, polyester resin, urethane resin, and melamine resin. Particularly preferred is an emulsion of acrylic resin or ethylene acrylic resin.
- Such an organic resin contributes effectively to the improvement of corrosion resistance, scratch resistance and punching ability, but if the blending ratio in the dry film is 5% by mass or more, the addition effect is large, while it is 40% by mass or less. In this case, the scratch resistance and Tig weldability after strain relief annealing will not be deteriorated, so the blending ratio of the organic resin is preferably about 5 to 40% by mass in terms of solid content.
- the ratio in a dry film is a ratio of each component in the film formed on the surface by apply
- the additive in addition to the above-described components, it does not hinder the inclusion of commonly used additives, other inorganic compounds, and organic compounds.
- the additive is added in order to further improve the performance and uniformity of the insulating coating, and examples thereof include a surfactant, a rust inhibitor, a lubricant, and an antioxidant.
- the compounding quantity of this additive it is preferable that the compounding ratio in a dry film shall be about 10 mass% or less from a viewpoint of maintaining sufficient film characteristics.
- the electromagnetic steel sheet as a material is not particularly limited, and any conventionally known steel sheet is suitable. That is, a so-called soft iron plate (electric iron plate) having a high magnetic flux density, a general cold-rolled steel plate such as SPCC, and a non-oriented electrical steel plate containing Si or Al for increasing the specific resistance are advantageously suitable.
- the present invention is suitable for application to a steel plate containing Si alone or (Si + Al) in an amount of about 0.1 to 10% by mass and / or a steel plate having an iron loss W 15/50 of about 7 W / kg or less.
- an insulating film does not prescribe
- a degreasing treatment such as alkali or pickling treatment such as hydrochloric acid, sulfuric acid or phosphoric acid.
- a degreasing treatment such as alkali or pickling treatment such as hydrochloric acid, sulfuric acid or phosphoric acid.
- a degreasing treatment such
- a roll coater such as a roll coater, a flow coater, a spray, and a knife coater that are generally used in industry can be applied as a method for applying the insulating coating treatment liquid.
- a baking method such as a hot air method, an infrared method, an induction heating method, or the like, which is usually performed, is possible.
- the baking temperature may be a normal level, and may be about 150 to 350 ° C. at the reached steel plate temperature.
- the electrical steel sheet with an insulating coating of the present invention can be subjected to strain relief annealing to remove, for example, strain due to punching.
- a preferable strain relief annealing atmosphere an atmosphere in which iron is hardly oxidized such as an N 2 atmosphere and a DX gas atmosphere is applied.
- the corrosion resistance can be further improved by setting the dew point high, for example, Dp: about 5 to 60 ° C., and slightly oxidizing the surface and the cut end face.
- a preferable strain relief annealing temperature is 700 to 900 ° C., more preferably 700 to 800 ° C.
- the holding time of the strain relief annealing temperature is preferably longer, but more preferably 2 hours or longer.
- the adhesion amount of the insulating coating is not particularly limited, but is preferably about 0.05 to 5 g / m 2 per side.
- the adhesion amount that is, the total solid mass of the insulating coating of the present invention can be measured from the weight reduction after removing the coating by alkali peeling. In addition, when the amount of adhesion is small, the amount of adhesion was measured by the alkali stripping method. Using a standard sample with a known amount of adhesion, the relationship between the detected intensity of Zr or Si by X-ray fluorescence analysis and the amount of adhesion was determined. Based on this calibration curve, the fluorescent X-ray analysis intensity of Zr or Si can be converted into an adhesion amount according to the coating composition.
- the adhesion amount is 0.05 g / m 2 non-above, it is possible to satisfy the insulation with corrosion, whereas if 5 g / m 2 or less, not only the adhesion is improved, blistering during paint baking is It does not occur and the paintability is not reduced. More preferably, it is 0.1 to 3.0 g / m 2 .
- the insulating coating is preferably formed on both sides of the steel sheet, but depending on the purpose, only one side may be formed. Further, depending on the purpose, only one side may be applied, and the other side may be another insulating film.
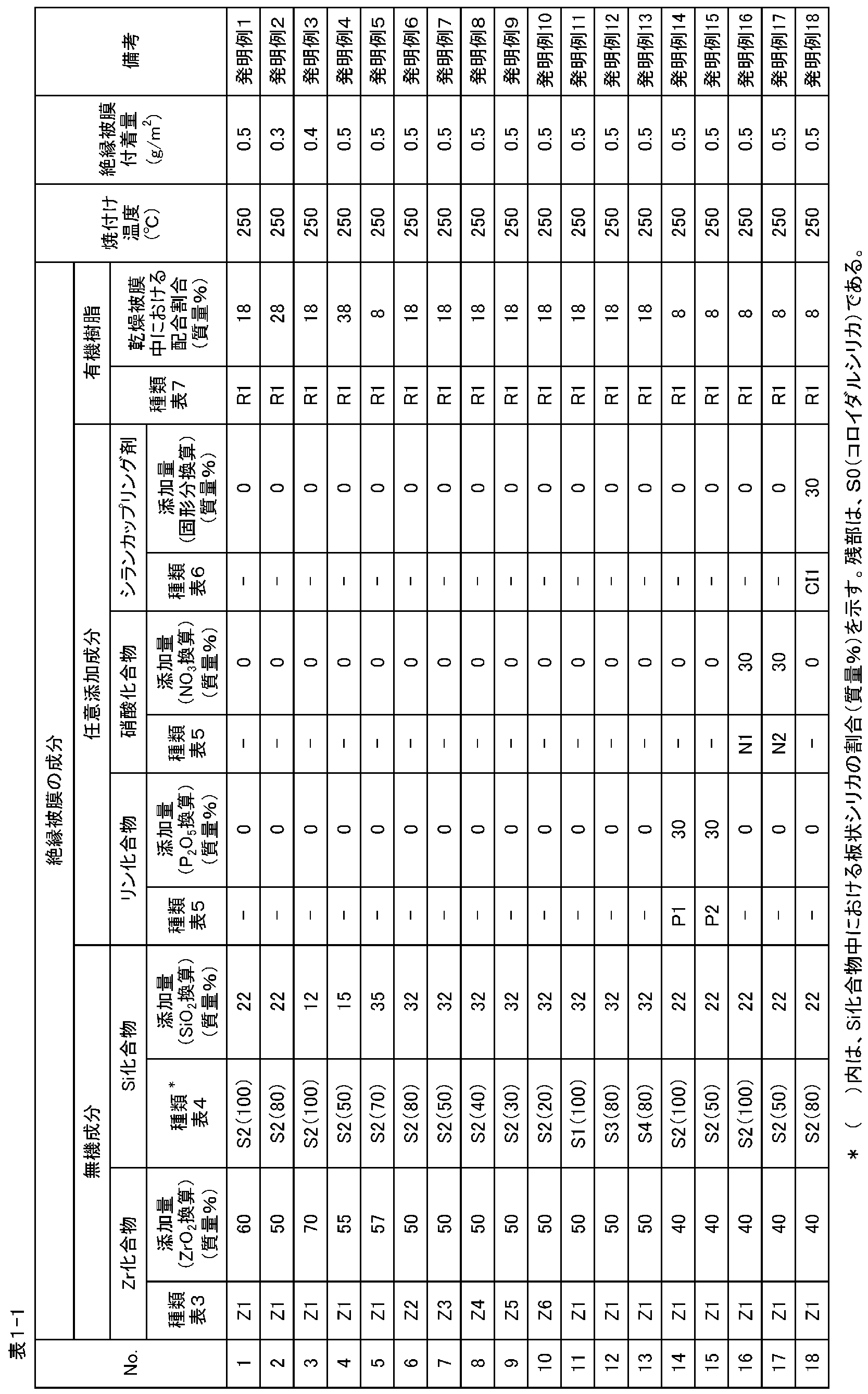
- Example 1 Si compound containing Zr compound and plate-like silica, nitric acid compound, silane coupling agent, phosphorus compound and addition so that the components of the insulating coating after drying are in the proportions shown in Table 1-1 and Table 1-2
- the agent was added to deionized water together with the organic resin to obtain a treatment liquid.
- concentration with respect to the amount of deionized water was 50 g / l.
- Each of these treatment liquids was rolled on a test piece surface cut into a size of width: 150 mm and length: 300 mm from a 0.5 mm thick electromagnetic steel sheet [A230 (JIS C 2552 (2000))]. After coating and baking in a hot air baking oven at the baking temperatures (reachable steel plate temperatures) shown in Table 1-1 and Table 1-2, the coating was allowed to cool to room temperature to form insulating coatings on both sides.
- Table 2 shows the results of examining the corrosion resistance and powder blowing resistance of the thus obtained semi-organic insulating coating-coated electrical steel sheet. Furthermore, after performing strain relief annealing at 750 ° C. for 2 hours in a nitrogen atmosphere, the scratch resistance, sticking property, Tig weldability, punchability, water resistance, and appearance after strain relief annealing are investigated, The obtained results are also shown in Table 2.
- Table 3 shows the types of Zr compounds
- Table 4 shows the types of Si compounds
- Table 5 shows the types of phosphorus compounds and nitrate compounds
- Table 6 shows the types of silane coupling agents
- Table 7 shows the types of organic resins. Respectively, as shown.
- the evaluation method for each characteristic is as follows. ⁇ Corrosion resistance> A wet test (50 ° C., relative humidity ⁇ 98%) was performed on the specimen, and the red rust generation rate after 48 hours was visually observed and evaluated by the area ratio. (Criteria) ⁇ : Red rust area ratio less than 5% ⁇ : Red rust area ratio 5% or more, less than 15% ⁇ : Red rust area ratio 15% or more, less than 40% ⁇ : Red rust area ratio 40% or more, less than 60% ⁇ : Red rust area ratio 60 %more than
- ⁇ Tig weldability> The test material was laminated at a pressure of 9.8 MPa (100 kgf / cm 2 ) so as to have a thickness of 30 mm, and Tig welding was performed on the end face portion (length 30 mm) under the following conditions.
- the superiority or inferiority was determined by the magnitude of the welding speed at which the number of blow holes satisfied 5 or less per bead.
- the semi-organic insulating coated steel sheet obtained according to the present invention is excellent in corrosion resistance and powder blowing resistance, as well as scratch resistance and sticking resistance after strain relief annealing. In addition, it was excellent in Tig weldability, punchability and water resistance, and also in appearance after strain relief annealing.
- Comparative Examples 1 and 2 in which the Zr compound was out of the proper range were particularly inferior in corrosion resistance, powder blowing resistance and scratch resistance after annealing. Moreover, the comparative example 1 was inferior also in the Tig weldability and the external appearance after annealing.
- Comparative Example 3 in which the Si compound is less than the lower limit is inferior in corrosion resistance, powder blowing resistance, post-annealing scratch resistance, and Tig weldability
- Comparative Example 4 in which the Si compound exceeds the upper limit is particularly powder resistant. And scratch resistance after annealing.
- Comparative Examples 5 to 11 containing a large amount of a nitric acid compound, a silane coupling agent, and a phosphorus compound exceeding the proper range are all inferior in corrosion resistance, powder blowing resistance, scratch resistance after annealing, and water resistance. It was.
- Comparative Example 12 using only colloidal silica containing no plate-like silica as the Si compound is particularly inferior in corrosion resistance, punchability, water resistance and appearance after annealing, and is also resistant to powder blowing and scratching after annealing. However, it was not as good as the invention examples.
- Example 2 Si components including Zr compound, B compound and plate-like silica, nitric acid compound, silane coupling agent, phosphorus so that the components of the insulating coating after drying are in the proportions shown in Tables 8-1 and 8-2.
- Compounds and additives were added to deionized water together with the organic resin to obtain a treatment liquid.
- concentration with respect to the amount of deionized water was 50 g / l.
- Each of these treatment liquids was rolled on a test piece surface cut into a size of width: 150 mm and length: 300 mm from a 0.5 mm thick electromagnetic steel sheet [A230 (JIS C 2552 (2000))]. After coating and baking in a hot air baking oven at the baking temperatures (reachable steel plate temperatures) shown in Tables 8-1 and 8-2, the film was allowed to cool to room temperature to form insulating coatings on both sides.
- Table 9 shows the results of examining the corrosion resistance and the powder blowing resistance of the thus obtained semi-organic insulating coated electromagnetic steel sheet. Furthermore, after performing strain relief annealing at 750 ° C. for 2 hours in a nitrogen atmosphere, the scratch resistance, sticking property, Tig weldability, punchability, water resistance, and appearance after strain relief annealing are investigated, The obtained results are also shown in Table 9. The types of the B compound are as shown in Table 10. The evaluation method for each characteristic is the same as that in the first embodiment.
- the semi-organic insulating coated electrical steel sheet obtained according to the present invention is excellent in corrosion resistance and powder blowing resistance, as well as scratch resistance and sticking resistance after strain relief annealing. In addition, it was excellent in Tig weldability, punchability and water resistance, and also in appearance after strain relief annealing.
- Comparative Examples 1 and 2 in which the Zr compound was out of the proper range were particularly inferior in corrosion resistance, powder blowing resistance and scratch resistance after annealing.
- the comparative example 1 was inferior also in the Tig weldability and the external appearance after annealing.
- the comparative example 3 with which the B compound exceeded the upper limit was especially inferior to sticking property.
- Comparative Example 4 in which the Si compound was less than the lower limit was inferior in corrosion resistance and Tig weldability
- Comparative Example 5 in which the Si compound exceeded the upper limit was particularly inferior in powder blowing resistance and scratch resistance after annealing.
- all of Comparative Examples 6 to 12 containing a large amount of a nitric acid compound, a silane coupling agent, and a phosphorus compound exceeding the appropriate range were inferior in corrosion resistance and water resistance.
- Comparative Example 13 using only colloidal silica containing no plate-like silica as the Si compound is particularly inferior in corrosion resistance, water resistance and appearance after annealing, and is also resistant to powder blowing, scratch resistance after annealing, sticking and The punchability did not reach the invention example.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Metallurgy (AREA)
- Mechanical Engineering (AREA)
- Nanotechnology (AREA)
- Inorganic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Power Engineering (AREA)
- Dispersion Chemistry (AREA)
- Chemical Treatment Of Metals (AREA)
- Laminated Bodies (AREA)
- Soft Magnetic Materials (AREA)
- Other Surface Treatments For Metallic Materials (AREA)
- Paints Or Removers (AREA)
Abstract
Description
(1) 溶接性、耐熱性を重視し、歪取り焼鈍に耐える無機被膜、
(2) 打抜性、溶接性の両立を目指し歪取り焼鈍に耐える樹脂含有の無機被膜(すなわち、半有機被膜)、
(3) 特殊用途で歪取り焼鈍不可の有機被膜
の3種に分類されるが、汎用品として歪取り焼鈍に耐えるのは、上記(1), (2)に示した無機成分を含む被膜であり、両者ともクロム化合物を含むものであった。
例えば、特許文献1には、少なくとも1種の2価金属を含む重クロム酸塩系水溶液に、該水溶液中のCrO3:100重量部に対し有機樹脂として酢酸ビニル/ベオバ比が90/10~40/60の割合になる樹脂エマルジョンを樹脂固形分で5~120重量部および有機還元剤を10~60重量部の割合で配合した処理液を、基地鉄板の表面に塗布し、常法による焼付けを施して得た電気絶縁被膜を有する電磁鋼板が記載されている。
これらの問題は、300℃以下の比較的低温で焼き付けた場合に発生しやすい問題であり、特に200℃以下の場合には、その発生が顕著であった。一方で、焼付け温度は消費エネルギーおよび製造コストの低減等の観点から、できるだけ低くすべきである。
本発明は、上記の知見に立脚するものである。
1.表面に、無機成分と有機樹脂からなる半有機絶縁被膜をそなえる電磁鋼板であって、該無機成分としてZr化合物および板状シリカを含むSi化合物をそれぞれ、乾燥被膜中における比率で、Zr化合物(ZrO2換算):20~70質量%、板状シリカを含むSi化合物(SiO2換算):10~50質量%含有し、残部が有機樹脂であることを特徴とする半有機絶縁被膜付き電磁鋼板。
(1) Zr化合物および板状シリカを含むSi化合物をそれぞれ、乾燥被膜中における比率で、Zr化合物(ZrO2換算):20~70質量%、板状シリカを含むSi化合物(SiO2換算):10~50質量%、
(2) 必要に応じて、さらにB化合物を、乾燥被膜中における比率で、B化合物(B2O3換算):0.1~5質量%、
(3) 必要に応じて、さらに硝酸化合物(NO3換算)、シランカップリング剤(固形分換 算)およびリン化合物(P2O5換算)のうちから選んだ一種または二種以上を、乾燥被膜中における比率で合計30質量%以下
含有し、
残部が実質的に(すなわち、不純物や公知の添加剤の含有を許容して)有機樹脂である半有機絶縁被膜付き電磁鋼板であり、さらに
前記板状シリカの平均粒子径が10~600nmであることおよび/または板状シリカのアスペクト比(平均長さ/平均厚み比)が2~400であることが好ましい。
まず、本発明において、半有機被膜の無機成分である、Zr化合物および板状シリカを含むSi化合物、さらにはB化合物の配合割合を、前記の範囲に限定した理由について説明する。
なお、これらの成分の質量%は、乾燥被膜中における比率である。
Zr化合物は、酸素との結合力が強く、Fe表面の酸化物、水酸化物などと強固に結合することができる。また、Zr化合物は3つ以上の結合手を持つため、Zr同士、もしくは他の無機化合物とネットワークを形成することでクロムを使用することなく強靭な被膜を形成することができる。しかしながら、Zr化合物の乾燥被膜中における比率が、ZrO2換算で20質量%に満たないと密着性が劣化し、耐食性、耐粉吹き性が劣化するだけでなく、Si化合物に起因した焼鈍後外観の劣化が発生する。一方、70質量%を超えると耐食性および耐粉吹き性が劣化し、また歪取り焼鈍板での耐キズ性も劣化する。それ故、Zr化合物はZrO2換算で20~70質量%の範囲に限定した。
本発明の板状シリカは、葉状シリカや鱗片状シリカとも呼ばれるもので、SiO2の薄層が多数積層された層状珪酸構造を有しており、非結晶性または微結晶性を有するものが多い。かような板状シリカは、一般的なシリカ粒子たとえばコロイダルシリカ等と比較して、層状の形態をとるために腐食物質透過抑制性に優れ、さらに、水酸基が多いために密着性に優れ、かつ軟質であることから滑り性に優れる。このため、耐食性や打抜性の向上により効果的である。
板状シリカは、薄層の一次粒子が積層した凝集粒子を作製し、この凝集粒子を粉砕することによって得ることができる。
ここに、板状シリカの平均粒子径は10~600nm程度とすることが好ましい。より好ましくは100~450nmの範囲である。また、かかる板状シリカのアスペクト比(長さ/厚み比)は2~400程度とすることが好ましい。より好ましくは10~100の範囲である。
また、シリカが、楕円形や多角形の場合には、同様に、何ヶ所かSEM観察して平面における平均粒子径および断面における平均厚みを求め、(平均粒子径)/(平均厚み)によりアスペクト比を求める。
上記した板状シリカのSi化合物全体における配合割合が50質量%に満たないと、本発明で目指したほど良好な耐食性および打抜性が得られないので、板状シリカの配合割合は50質量%以上とすることが好ましい。勿論、Si化合物が全て板状シリカであってもよい。特に好適な範囲は50~100質量%の範囲である。
なお、板状シリカの配合割合は、例えばFIB断面において、板状シリカ部のSiO2量(B)とそれ以外の領域でのSiO2量(A)を測定し、B/(A+B)×100により求めることができる。また、それぞれの領域におけるSiO2量は、EDS等で求めた平均Si濃度と領域面積から求めることができる。
ここに、かかるSi化合物の乾燥被膜中における含有量がSiO2換算値で10質量%に満たないと十分な耐食性が得られず、一方50質量%を超えると耐粉吹き性が劣化し、また歪取り焼鈍板での耐キズ性も劣化するのでSi化合物は10~50質量%の範囲に限定した。
B化合物:B2O3換算で0.1~5質量%
B化合物は、Zr化合物を単独で添加した場合の問題の解決に有利に寄与する。すなわち、Zr化合物を単独で添加した場合には耐食性や耐粉吹き性が劣化し、また歪取り焼鈍板での耐キズ性が著しく劣化する傾向が見られた。この理由は、Zr化合物単独では、焼付けた際の体積収縮が大きいために被膜割れが生じやすく、部分的に素地が露出する箇所が発生するためと考えられる。
これに対し、B化合物をZr化合物に適量配合することにより、Zr単独の場合に発生していた被膜割れが効果的に緩和され、耐粉吹き性を著しく改善することができる。
ここに、B化合物の乾燥被膜中における比率がB2O3換算で0.1質量%以上であればその添加効果を有し、一方5質量%以下であれば被膜中の未反応物が残存することがなく、歪取り焼鈍後に被膜同士が融着する不具合(スティック)が発生しないので、B化合物はB2O3換算で0.1~5質量%の範囲とすることが好ましい。
かような硝酸化合物、シランカップリング剤およびリン化合物は、耐食性の改善に有効に寄与するが、乾燥被膜中における比率が合計で30質量%以下であれば、未反応物が被膜中に残存することがなく、耐水性を低下させることがないので、含有量は合計で30質量%以下とすることが好ましい。なお、これらの成分の効果を十分に発揮させるには、乾燥被膜中における比率で1質量%以上含有させることが好ましい。
・硝酸系
硝酸(HNO3)、硝酸カリウム(KNO3)、硝酸ナトリウム(NaNO3)、硝酸アンモニウム(NH4NO3)、硝酸カルシウム(Ca(NO3)2)、硝酸銀(AgNO3)、硝酸鉄(II)(Fe(NO3)2)、硝酸鉄(III)(Fe(NO3)3)、硝酸銅(II)(Cu(NO3)2)、硝酸バリウム(Ba(NO3)2)、硝酸アルミニウム(Al(NO3)3)、硝酸マグネシウム(Mg(NO3)2)、硝酸亜鉛(Zn(NO3)2)、硝酸ニッケル(II)(Ni(NO3)2)、硝酸ジルコニウム(ZrO(NO3)2)。
・亜硝酸系
亜硝酸(HNO2)、亜硝酸カリウム、亜硝酸カルシウム、亜硝酸銀、亜硝酸ナトリウム、亜硝酸バリウム、亜硝酸エチル、亜硝酸イソアミル、亜硝酸イソブチル、亜硝酸イソプロピル、亜硝酸-t-ブチル、亜硝酸-n-ブチル、亜硝酸-n-プロピル。
・ビニル系
ビニルトリクロルシラン、ビニルトリメトキシシラン、ビニルトリエトキシシラン。
・エポキシ系
2-(3,4-エポキシシクロヘキシル)エチルトリメトキシシラン、3-グリシドキシプロピルトリメトキシシラン、3-グリシドキシプロピルメチルジエトキシシラン、3-グリシドキシプロピルトリエトキシシラン。
・スチリル系
p-スチリルトリメトキシシラン。
・メタクリロキシ系
3-メタクリロキシプロピルメチルジメトキシシラン、3-メタクリロキシプロピルトリメトキシシラン、3-メタクリロキシプロピルメチルジエトキシシラン、3-メタクリロキシプロピルトリエトキシシラン。
・アクリロキシ系
3-アクリロキシプロピルトリメトキシシラン。
・アミノ系
N-2-(アミノエチル)-3-アミノプロピルメチルジメトキシシラン、N-2-(アミノエチル)-3-アミノプロピルトリメトキシシラン、N-2-(アミノエチル)-3-アミノプロピルトリエトキシシラン、3-アミノプロピルトリメトキシシラン、3-アミノプロピルトリエトキシシラン、3-トリエトキシシリル-N-(1,3-ジメチル-ブチリデン)プロピルアミンとその部分加水分解物、N-フェニル-3-アミノプロピルトリメトキシシラン、N-(ビニルベンジル)-2-アミノエチル-3-アミノプロピルトリメトキシシランの塩酸塩、特殊アミノシラン。
・ウレイド系
3-ウレイドプロピルトリエトキシシラン。
・クロロプロピル系
3-クロロプロピルトリメトキシシラン。
・メルカプト系
3-メルカプトプロピルメチルジメトキシシラン、3-メルカプトプロピルトリメトキシシラン。
・ポリスルフィド系
ビス(トリエトキシシリルプロピル)テトラスルフィド。
・イソシアネート系
3-イソシアネートプロピルトリエトキシシラン。
・リン酸
オルトリン酸、無水リン酸、直鎖状ポリリン酸、環状メタリン酸。
・リン酸塩
リン酸マグネシウム、リン酸アルミニウム、リン酸カルシウム、リン酸亜鉛。
本発明において、有機樹脂としては特に制限はなく、従来から使用されている公知のものいずれもが有利に適合する。例えば、アクリル樹脂、アルキッド樹脂、ポリオレフイン樹脂、スチレン樹脂、酢酸ビニル樹脂、エポキシ樹脂、フェノール樹脂、ポリエステル樹脂、ウレタン樹脂、メラミン樹脂等の水性樹脂(エマルジョン、ディスパーション、水溶性)が挙げられる。特に好ましくはアクリル樹脂やエチレンアクリル酸樹脂のエマルジョンである。
ここに、添加剤は、絶縁被膜の性能や均一性を一層向上させるために添加されるもので、界面活性剤や防錆剤、潤滑剤、酸化防止剤等が挙げられる。なお、かかる添加剤の配合量は、十分な被膜特性を維持する観点から、乾燥被膜中の配合割合が10質量%程度以下とすることが好ましい。
すなわち、磁束密度の高いいわゆる軟鉄板(電気鉄板)やSPCC等の一般冷延鋼板、また比抵抗を上げるためにSiやAlを含有させた無方向性電磁鋼板などいずれもが有利に適合する。特に、Si単独または(Si+Al)量が0.1~10質量%程度含有された鋼板および/または鉄損W15/50が7W/kg以下程度の鋼板に適用して好適である。
本発明では、素材である電磁鋼板の前処理については特に規定しない。すなわち、未処理でもよいが、アルカリなどの脱脂処理、塩酸、硫酸、リン酸などの酸洗処理を施すことは有利である。
そして、この電磁鋼板の表面に、Zr化合物および板状シリカを含むSi化合物、さらにはB化合物、またさらには硝酸化合物、シランカップリング剤およびリン化合物のうちから選んだ一種または二種以上や、必要に応じて添加剤等を、有機樹脂と共に所定の割合で配合した処理液を塗布し、焼き付けることにより絶縁被膜を形成させる。絶縁被膜用処理液の塗布方法は、一般工業的に用いられるロールコーター、フローコーター、スプレー、ナイフコーター等種々の方法が適用可能である。また、焼き付け方法についても、通常実施されるような熱風式、赤外式、誘導加熱式等が可能である。焼付け温度も通常レベルであればよく、到達鋼板温度で150~350℃程度であればよい。
実施例1
乾燥後の絶縁被膜の成分が表1-1,表1-2に示す割合になるように、Zr化合物および板状シリカを含むSi化合物、さらには硝酸化合物、シランカップリング剤、リン化合物や添加剤を、有機樹脂と共に脱イオン水に添加し、処理液とした。なお、脱イオン水量に対する添加濃度は50g/lとした。
これらの各処理液を、板厚:0.5mmの電磁鋼板〔A230(JIS C 2552(2000))〕から幅:150mm、長さ:300mmの大きさに切り出した試験片の表面にロールコーターで塗布し、熱風焼付け炉により表1-1,表1-2に示す焼付け温度(到達鋼板温度)で焼付けした後、常温に放冷して、絶縁被膜を両面に形成した。
さらに、窒素雰囲気中にて750℃、2時間の歪取り焼鈍を行ったのちの耐キズ性、スティッキング性、Tig溶接性、打抜性、耐水性および歪取り焼鈍後の外観について調査を行い、得られた結果を表2に併記する。
<耐食性>
供試材に対して湿潤試験(50℃、相対湿度≧98%)を行い、48時間後の赤錆発生率を目視で観察し、面積率で評価した。
(判定基準)
☆:赤錆面積率 5%未満
◎:赤錆面積率 5%以上、15%未満
○:赤錆面積率 15%以上、40%未満
△:赤錆面積率 40%以上、60%未満
×:赤錆面積率 60%以上
試験条件;フェルト接触面幅20mm×10mm、荷重:0.4MPa(3.8kg/cm2)、被膜表面を100回単純往復。試験後の擦り跡を目視観察し、被膜の剥離状態および粉吹き状態を評価した。
(判定基準)
◎:被膜残存率 ほとんど擦り跡が認められない
○:被膜残存率 若干の擦り跡および若干の粉吹きが認められる程度
△:被膜残存率 被膜の剥離が進行し擦り跡および粉吹きがはっきりわかる程度
×:被膜残存率 地鉄が露出するほど剥離し粉塵が甚大
試験条件;N2雰囲気、750℃で2時間保持して焼鈍したサンプル表面を鋼板せん断エッジで引っかき、キズ、粉吹きの程度を判定した。
(判定基準)
◎:キズ、粉吹きの発生がほとんど認められない
○:若干の擦り跡および若干の粉吹きが認められる程度
△:擦り跡および粉吹きがはっきりわかる程度
×:地鉄が露出するほど剥離し粉塵が甚大
50mm角の供試材10枚を重ねて荷重:20kPa(200g/cm2)をかけながら窒素雰囲気下で750℃,2時間の条件にて焼鈍を行った。ついで、供試材(鋼板)上に500gの分銅を落下させ、5分割するときの落下高さを調査した。
(判定基準)
◎:10cm以下
○:10cm超、15cm以下
△:15cm超、30cm以下
×: 30cm超
供試材を30mmの厚みになるように9.8MPa(100kgf/cm2)の圧力にて積層し、その端面部(長さ30mm)に対して、次の条件でTig溶接を実施した。
・溶接電流:120A
・Arガス流量:6リットル/min
・溶接速度:10、20、30、40、50、60、70、80、90、100cm/min
(判定基準)
ブローホールの数が1ビードにつき5個以下を満足する溶接速度の大小で優劣を判定した。
◎:60cm/min以上
○:40cm/min以上、60cm/min未満
△:20cm/min以上、40cm/min未満
×:20cm/min未満
供試材に対して、15mmφスチールダイスを用いて、かえり高さが50μmに達するまで打ち抜きを行い、その打ち抜き数で評価した。
(判定基準)
◎:120万回以上
○:50万回以上、120万回未満
△:10万回以上、50万回未満
×:10万回未満
供試材を、沸騰水蒸気中に30分暴露させ、外観変化を観察した。
(判定基準)
◎:変化なし
○:目視で若干の変色が認められる程度
△:目視で変色がはっきり認められる程度
×:被膜溶解
供試材に対して、N2雰囲気中にて750℃,2時間保持後、常温まで冷却した鋼板の外観を目視観察した。
(判定基準)
◎:図1(a)に示すように、焼鈍後の外観が完全に均一な場合
○:図1(b)に示すように、焼鈍後の外観にムラが認められる場合
△:図1(c)に示すように、焼鈍後の外観に斑模様が認められる場合
×:図1(d)に示すように、焼鈍後の外観に顕著な斑模様が認められる場合
これに対し、Zr化合物が適正範囲から外れた比較例1,2は、特に耐食性、耐粉吹き性および焼鈍後耐キズ性に劣っていた。また比較例1は、Tig溶接性や焼鈍後外観にも劣っていた。
また、Si化合物が下限に満たない比較例3は、耐食性や耐粉吹き性、焼鈍後耐キズ性、Tig溶接性に劣り、一方Si化合物が上限を超えた比較例4は、特に耐粉吹き性、焼鈍後耐キズ性に劣っていた。
さらに、硝酸化合物やシランカップリング剤およびリン化合物を適正範囲を超えて多量に含有させた比較例5~11はいずれも、耐食性、耐粉吹き性、焼鈍後耐キズ性および耐水性に劣っていた。
なお、Si化合物として、板状シリカを含まないコロイダルシリカのみを用いた比較例12は、特に耐食性、打抜性、耐水性および焼鈍後外観に劣り、また耐粉吹き性および焼鈍後耐キズ性も発明例には及ばなかった。
乾燥後の絶縁被膜の成分が表8-1,表8-2に示す割合になるように、Zr化合物、B化合物および板状シリカを含むSi化合物、さらには硝酸化合物、シランカップリング剤、リン化合物や添加剤を、有機樹脂と共に脱イオン水に添加し、処理液とした。なお、脱イオン水量に対する添加濃度は50g/lとした。
これらの各処理液を、板厚:0.5mmの電磁鋼板〔A230(JIS C 2552(2000))〕から幅:150mm、長さ:300mmの大きさに切り出した試験片の表面にロールコーターで塗布し、熱風焼付け炉により表8-1,表8-2に示す焼付け温度(到達鋼板温度)で焼付けした後、常温に放冷して、絶縁被膜を両面に形成した。
さらに、窒素雰囲気中にて750℃、2時間の歪取り焼鈍を行ったのちの耐キズ性、スティッキング性、Tig溶接性、打抜性、耐水性および歪取り焼鈍後の外観について調査を行い、得られた結果を表9に併記する。
なお、B化合物の種類は表10に示したとおりである。
また、各特性の評価方法は、実施例1の場合と同じである。
これに対し、Zr化合物が適正範囲から外れた比較例1,2は、特に耐食性、耐粉吹き性および焼鈍後耐キズ性に劣っていた。また比較例1は、Tig溶接性や焼鈍後外観にも劣っていた。
また、B化合物が上限を超えた比較例3は、特にスティッキング性に劣っていた。
Si化合物が下限に満たない比較例4は、耐食性やTig溶接性に劣り、一方Si化合物が上限を超えた比較例5は、特に耐粉吹き性、焼鈍後耐キズ性に劣っていた。
さらに、硝酸化合物やシランカップリング剤およびリン化合物を適正範囲を超えて多量に含有させた比較例6~12はいずれも、耐食性および耐水性に劣っていた。
なお、Si化合物として、板状シリカを含まないコロイダルシリカのみを用いた比較例13は、特に耐食性、耐水性および焼鈍後外観に劣り、また耐粉吹き性、焼鈍後耐キズ性、スティッキング性および打抜性も発明例には及ばなかった。
Claims (5)
- 表面に、無機成分と有機樹脂からなる半有機絶縁被膜をそなえる電磁鋼板であって、該無機成分としてZr化合物および板状シリカを含むSi化合物をそれぞれ、乾燥被膜中における比率で、Zr化合物(ZrO2換算):20~70質量%、板状シリカを含むSi化合物(SiO2換算):10~50質量%含有し、残部が有機樹脂であることを特徴とする半有機絶縁被膜付き電磁鋼板。
- 前記板状シリカの平均粒子径が10~600nmであることを特徴とする請求項1に記載の半有機絶縁被膜付き電磁鋼板。
- 前記板状シリカのアスペクト比(長さ/厚み比)が2~400であることを特徴とする請求項1または2に記載の半有機絶縁被膜付き電磁鋼板。
- 前記無機成分として、さらにB化合物を、乾燥被膜中における比率で、B化合物(B2O3換算):0.1~5質量%含有することを特徴とする請求項1ないし3のいずれかに記載の半有機絶縁被膜付き電磁鋼板。
- 前記被膜中に、さらに硝酸化合物(NO3換算)、シランカップリング剤(固形分換算)およびリン化合物(P2O5換算)のうちから選んだ一種または二種以上を、乾燥被膜中における比率で合計30質量%以下で含有することを特徴とする請求項1ないし4のいずれかに記載の半有機絶縁被膜付き電磁鋼板。
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/810,798 US9534136B2 (en) | 2010-07-22 | 2011-07-21 | Magnetic steel sheet with semi-organic insulation coating |
| CN2011800351407A CN103003470A (zh) | 2010-07-22 | 2011-07-21 | 具有半有机绝缘覆膜的电磁钢板 |
| RU2013107785/02A RU2534461C2 (ru) | 2010-07-22 | 2011-07-21 | Лист из магнитной стали с полуорганическим изоляционным покрытием |
| MX2013000564A MX344265B (es) | 2010-07-22 | 2011-07-21 | Lámina de acero magnetico con recubrimiento de aislante semi-organico. |
| BR112013000120-8A BR112013000120B1 (pt) | 2010-07-22 | 2011-07-21 | chapa de aço magnético com revestimento de isolamento semiorgânico |
| CA2803150A CA2803150C (en) | 2010-07-22 | 2011-07-21 | Magnetic steel sheet with semi-organic insulation coating |
| EP11809461.4A EP2597176B1 (en) | 2010-07-22 | 2011-07-21 | Electromagnetic steel sheet with semi-organic insulating coating film |
| KR1020137002933A KR101472228B1 (ko) | 2010-07-22 | 2011-07-21 | 반유기 절연 피막이 형성된 전기 강판 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010165380A JP5589639B2 (ja) | 2010-07-22 | 2010-07-22 | 半有機絶縁被膜付き電磁鋼板 |
| JP2010-165380 | 2010-07-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012011286A1 true WO2012011286A1 (ja) | 2012-01-26 |
Family
ID=45496718
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/004123 WO2012011286A1 (ja) | 2010-07-22 | 2011-07-21 | 半有機絶縁被膜付き電磁鋼板 |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US9534136B2 (ja) |
| EP (1) | EP2597176B1 (ja) |
| JP (1) | JP5589639B2 (ja) |
| KR (1) | KR101472228B1 (ja) |
| CN (1) | CN103003470A (ja) |
| BR (1) | BR112013000120B1 (ja) |
| CA (1) | CA2803150C (ja) |
| MX (1) | MX344265B (ja) |
| RU (1) | RU2534461C2 (ja) |
| TW (1) | TWI538800B (ja) |
| WO (1) | WO2012011286A1 (ja) |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IN2014CN04062A (ja) * | 2011-11-04 | 2015-09-04 | Tata Steel Uk Ltd | |
| JP5974636B2 (ja) * | 2012-05-29 | 2016-08-23 | Jfeスチール株式会社 | 絶縁被膜付き電磁鋼板 |
| DE102013208618A1 (de) * | 2013-05-10 | 2014-11-13 | Henkel Ag & Co. Kgaa | Chromfreie Beschichtung zur elektrischen Isolierung von kornorientiertem Elektroband |
| EP3000915B1 (en) * | 2013-05-23 | 2017-09-13 | JFE Steel Corporation | Electromagnetic steel sheet having insulating coating film attached thereto |
| EP3072936A1 (de) * | 2015-03-24 | 2016-09-28 | Voestalpine Stahl GmbH | Coil und Elektroband oder -blech |
| US10526672B2 (en) | 2015-04-07 | 2020-01-07 | Jfe Steel Corporation | Electrical steel sheet with insulating coating |
| JP6539728B2 (ja) * | 2015-04-24 | 2019-07-03 | 日本パーカライジング株式会社 | 金属材料用表面処理剤及び金属材料 |
| RU2689353C1 (ru) * | 2015-09-02 | 2019-05-27 | ДжФЕ СТИЛ КОРПОРЕЙШН | Обрабатывающий раствор для получения изоляционного покрытия и способ изготовления металла, имеющего изоляционное покрытие |
| WO2017155028A1 (ja) * | 2016-03-09 | 2017-09-14 | 新日鐵住金株式会社 | 表面処理鋼板および表面処理鋼板の製造方法 |
| JP6103114B2 (ja) * | 2016-06-03 | 2017-03-29 | Jfeスチール株式会社 | 絶縁被膜付き電磁鋼板 |
| KR20190097246A (ko) | 2017-03-23 | 2019-08-20 | 닛폰세이테츠 가부시키가이샤 | 전자 강판 |
| US20220106689A1 (en) * | 2019-02-14 | 2022-04-07 | Jfe Steel Corporation | Electrical steel sheet having insulating coating |
| US20230033301A1 (en) | 2019-11-21 | 2023-02-02 | Nippon Steel Corporation | Non-oriented electrical steel sheet and method for producing same |
| TWI734645B (zh) * | 2020-11-12 | 2021-07-21 | 中國鋼鐵股份有限公司 | 皮膜處理液及使用皮膜處理液的鈦鎳合金盤元的抽線預處理方法 |
| KR20230095527A (ko) * | 2021-12-22 | 2023-06-29 | 주식회사 포스코 | 전기강판 절연 피막 조성물, 전기강판, 및 이의 제조 방법 |
| CN114525502A (zh) * | 2022-02-16 | 2022-05-24 | 上海岱森化工科技有限公司 | 无方向性电磁钢板用绝缘皮膜处理剂 |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0636476B2 (ja) | 1987-12-10 | 1994-05-11 | 富士通株式会社 | プリント配線板組合せ方法 |
| JPH1046350A (ja) | 1996-07-30 | 1998-02-17 | Kawasaki Steel Corp | 耐食性に優れた歪取り焼鈍が可能なクロム化合物を含まない絶縁被膜を電磁鋼板の表面に形成する方法 |
| JPH10130858A (ja) | 1996-10-28 | 1998-05-19 | Kawasaki Steel Corp | 低温焼き付けで製造でき、歪取り焼鈍が可能で沸騰水蒸気暴露性、耐溶剤性が良好な絶縁被膜付き電磁鋼板 |
| JP2944849B2 (ja) | 1993-05-21 | 1999-09-06 | 新日本製鐵株式会社 | 被膜特性の極めて良好な無方向性電磁鋼板の製造方法 |
| JP2003253462A (ja) * | 2002-03-07 | 2003-09-10 | Kansai Paint Co Ltd | 鋼板用表面処理組成物 |
| JP2003268563A (ja) * | 2002-03-14 | 2003-09-25 | Kansai Paint Co Ltd | 亜鉛又は亜鉛系合金メッキ用表面処理組成物 |
| JP2005268630A (ja) * | 2004-03-19 | 2005-09-29 | Jfe Steel Kk | クロムフリー絶縁被膜付き電磁鋼板 |
| JP2007197824A (ja) | 2005-12-28 | 2007-08-09 | Jfe Steel Kk | 絶縁被膜を有する電磁鋼板 |
| JP2007197820A (ja) | 2005-12-28 | 2007-08-09 | Jfe Steel Kk | 絶縁被膜を有する電磁鋼板およびその製造方法 |
| JP2009194314A (ja) * | 2008-02-18 | 2009-08-27 | Jfe Steel Corp | 絶縁被膜を有する電磁鋼板 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6036476B2 (ja) | 1982-11-09 | 1985-08-20 | 川崎製鉄株式会社 | 溶接時における悪臭の発生がない電気絶縁被膜を有する電気鉄板 |
| SU1733501A1 (ru) * | 1990-04-11 | 1992-05-15 | Буйский Химический Завод | Способ получени электроизол ционных покрытий |
| JP3604306B2 (ja) * | 1999-10-01 | 2004-12-22 | 住友金属工業株式会社 | 絶縁皮膜付き電磁鋼板 |
| JP3823125B2 (ja) * | 2001-11-08 | 2006-09-20 | 壽男 小田島 | 電磁鋼板用クロムフリー表面処理剤及び表面処理電磁鋼板 |
| WO2005090636A1 (ja) * | 2004-03-19 | 2005-09-29 | Jfe Steel Corporation | 絶縁被膜を有する電磁鋼板 |
| RU2357994C2 (ru) * | 2004-10-18 | 2009-06-10 | Ниппон Стил Корпорейшн | Термостойкое клеящее изоляционное покрытие и лист электротехнической стали с таким покрытием, магнитный сердечник, где используется лист электротехнической стали, и способ его получения |
| JP5082601B2 (ja) * | 2007-06-05 | 2012-11-28 | 住友金属工業株式会社 | 電磁鋼板の絶縁皮膜形成用処理液 |
-
2010
- 2010-07-22 JP JP2010165380A patent/JP5589639B2/ja active Active
-
2011
- 2011-07-21 KR KR1020137002933A patent/KR101472228B1/ko active IP Right Grant
- 2011-07-21 BR BR112013000120-8A patent/BR112013000120B1/pt active IP Right Grant
- 2011-07-21 EP EP11809461.4A patent/EP2597176B1/en active Active
- 2011-07-21 US US13/810,798 patent/US9534136B2/en active Active
- 2011-07-21 CN CN2011800351407A patent/CN103003470A/zh active Pending
- 2011-07-21 RU RU2013107785/02A patent/RU2534461C2/ru active
- 2011-07-21 WO PCT/JP2011/004123 patent/WO2012011286A1/ja active Application Filing
- 2011-07-21 CA CA2803150A patent/CA2803150C/en active Active
- 2011-07-21 MX MX2013000564A patent/MX344265B/es active IP Right Grant
- 2011-07-22 TW TW100125994A patent/TWI538800B/zh active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0636476B2 (ja) | 1987-12-10 | 1994-05-11 | 富士通株式会社 | プリント配線板組合せ方法 |
| JP2944849B2 (ja) | 1993-05-21 | 1999-09-06 | 新日本製鐵株式会社 | 被膜特性の極めて良好な無方向性電磁鋼板の製造方法 |
| JPH1046350A (ja) | 1996-07-30 | 1998-02-17 | Kawasaki Steel Corp | 耐食性に優れた歪取り焼鈍が可能なクロム化合物を含まない絶縁被膜を電磁鋼板の表面に形成する方法 |
| JPH10130858A (ja) | 1996-10-28 | 1998-05-19 | Kawasaki Steel Corp | 低温焼き付けで製造でき、歪取り焼鈍が可能で沸騰水蒸気暴露性、耐溶剤性が良好な絶縁被膜付き電磁鋼板 |
| JP2003253462A (ja) * | 2002-03-07 | 2003-09-10 | Kansai Paint Co Ltd | 鋼板用表面処理組成物 |
| JP2003268563A (ja) * | 2002-03-14 | 2003-09-25 | Kansai Paint Co Ltd | 亜鉛又は亜鉛系合金メッキ用表面処理組成物 |
| JP2005268630A (ja) * | 2004-03-19 | 2005-09-29 | Jfe Steel Kk | クロムフリー絶縁被膜付き電磁鋼板 |
| JP2007197824A (ja) | 2005-12-28 | 2007-08-09 | Jfe Steel Kk | 絶縁被膜を有する電磁鋼板 |
| JP2007197820A (ja) | 2005-12-28 | 2007-08-09 | Jfe Steel Kk | 絶縁被膜を有する電磁鋼板およびその製造方法 |
| JP2009194314A (ja) * | 2008-02-18 | 2009-08-27 | Jfe Steel Corp | 絶縁被膜を有する電磁鋼板 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2597176A4 * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2597176A4 (en) | 2014-12-24 |
| CA2803150C (en) | 2016-07-05 |
| EP2597176A1 (en) | 2013-05-29 |
| RU2534461C2 (ru) | 2014-11-27 |
| BR112013000120B1 (pt) | 2020-07-07 |
| JP2012025999A (ja) | 2012-02-09 |
| MX2013000564A (es) | 2013-02-21 |
| EP2597176B1 (en) | 2016-09-07 |
| MX344265B (es) | 2016-12-09 |
| US20130115444A1 (en) | 2013-05-09 |
| TW201206695A (en) | 2012-02-16 |
| CN103003470A (zh) | 2013-03-27 |
| RU2013107785A (ru) | 2014-08-27 |
| BR112013000120A2 (pt) | 2016-05-24 |
| TWI538800B (zh) | 2016-06-21 |
| CA2803150A1 (en) | 2012-01-26 |
| US9534136B2 (en) | 2017-01-03 |
| KR101472228B1 (ko) | 2014-12-11 |
| KR20130031914A (ko) | 2013-03-29 |
| JP5589639B2 (ja) | 2014-09-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5589639B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| JP5640352B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| KR101608572B1 (ko) | 절연 피막이 형성된 전기 강판 | |
| JP4461861B2 (ja) | クロムフリー絶縁被膜付き電磁鋼板 | |
| JP5830833B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| JP5741190B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| JP5125073B2 (ja) | 絶縁被膜を有する電磁鋼板 | |
| KR100816695B1 (ko) | 절연 피막을 갖는 전자 강판 | |
| JP5598307B2 (ja) | 無機質絶縁被膜付き電磁鋼板 | |
| JP5494240B2 (ja) | 無機質絶縁被膜付き電磁鋼板 | |
| JP2007119799A (ja) | 絶縁被膜を有する電磁鋼板 | |
| JP5598289B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| JP5471081B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| JP2012097339A (ja) | 無機質絶縁被膜付き電磁鋼板 | |
| EP4050122A1 (en) | Electromagnetic steel sheet with insulation coating film | |
| JP5125072B2 (ja) | 絶縁被膜を有する電磁鋼板 | |
| JP5708435B2 (ja) | 半有機絶縁被膜付き電磁鋼板 | |
| JP2012117104A (ja) | 無機質絶縁被膜付き電磁鋼板 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11809461 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2787/MUMNP/2012 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2803150 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2013/000564 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13810798 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2011809461 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011809461 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20137002933 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2013107785 Country of ref document: RU Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013000120 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013000120 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130122 |