WO2016104724A1 - ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びに歯科用製品 - Google Patents
ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びに歯科用製品 Download PDFInfo
- Publication number
- WO2016104724A1 WO2016104724A1 PCT/JP2015/086272 JP2015086272W WO2016104724A1 WO 2016104724 A1 WO2016104724 A1 WO 2016104724A1 JP 2015086272 W JP2015086272 W JP 2015086272W WO 2016104724 A1 WO2016104724 A1 WO 2016104724A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- region
- shielding material
- light shielding
- sintered body
- zirconia sintered
- Prior art date
Links
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 title claims abstract description 306
- 239000000203 mixture Substances 0.000 title claims abstract description 61
- 229940023487 dental product Drugs 0.000 title claims description 19
- 239000000463 material Substances 0.000 claims abstract description 113
- RUDFQVOCFDJEEF-UHFFFAOYSA-N yttrium(III) oxide Inorganic materials [O-2].[O-2].[O-2].[Y+3].[Y+3] RUDFQVOCFDJEEF-UHFFFAOYSA-N 0.000 claims abstract description 34
- 239000003381 stabilizer Substances 0.000 claims abstract description 32
- 229910002077 partially stabilized zirconia Inorganic materials 0.000 claims description 23
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 11
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 11
- 238000010304 firing Methods 0.000 claims description 10
- 229910052726 zirconium Inorganic materials 0.000 claims description 10
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims description 8
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 7
- 239000002131 composite material Substances 0.000 claims description 7
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 claims description 7
- 229910052710 silicon Inorganic materials 0.000 claims description 7
- 239000010703 silicon Substances 0.000 claims description 7
- 229910052814 silicon oxide Inorganic materials 0.000 claims description 7
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims description 7
- 239000003086 colorant Substances 0.000 claims description 5
- 239000003795 chemical substances by application Substances 0.000 claims description 4
- 239000004053 dental implant Substances 0.000 claims description 4
- 230000007547 defect Effects 0.000 abstract description 4
- 230000000903 blocking effect Effects 0.000 abstract 2
- 239000010410 layer Substances 0.000 description 69
- 239000000843 powder Substances 0.000 description 53
- 238000000465 moulding Methods 0.000 description 19
- 239000000047 product Substances 0.000 description 11
- 230000008859 change Effects 0.000 description 10
- 238000004519 manufacturing process Methods 0.000 description 8
- 238000005259 measurement Methods 0.000 description 8
- 238000010586 diagram Methods 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 239000002245 particle Substances 0.000 description 7
- 238000005245 sintering Methods 0.000 description 7
- 230000007423 decrease Effects 0.000 description 6
- 238000011049 filling Methods 0.000 description 4
- 239000000049 pigment Substances 0.000 description 4
- 239000002002 slurry Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 229910010413 TiO 2 Inorganic materials 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 239000013078 crystal Substances 0.000 description 3
- 239000003973 paint Substances 0.000 description 3
- 229910052720 vanadium Inorganic materials 0.000 description 3
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 2
- 229910052693 Europium Inorganic materials 0.000 description 2
- 229910052688 Gadolinium Inorganic materials 0.000 description 2
- 229910004298 SiO 2 Inorganic materials 0.000 description 2
- 229910006501 ZrSiO Inorganic materials 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 239000000919 ceramic Substances 0.000 description 2
- RKTYLMNFRDHKIL-UHFFFAOYSA-N copper;5,10,15,20-tetraphenylporphyrin-22,24-diide Chemical compound [Cu+2].C1=CC(C(=C2C=CC([N-]2)=C(C=2C=CC=CC=2)C=2C=CC(N=2)=C(C=2C=CC=CC=2)C2=CC=C3[N-]2)C=2C=CC=CC=2)=NC1=C3C1=CC=CC=C1 RKTYLMNFRDHKIL-UHFFFAOYSA-N 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000001513 hot isostatic pressing Methods 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 2
- 238000003825 pressing Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- LIVNPJMFVYWSIS-UHFFFAOYSA-N silicon monoxide Chemical compound [Si-]#[O+] LIVNPJMFVYWSIS-UHFFFAOYSA-N 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 229910052727 yttrium Inorganic materials 0.000 description 2
- 229910052845 zircon Inorganic materials 0.000 description 2
- GFQYVLUOOAAOGM-UHFFFAOYSA-N zirconium(iv) silicate Chemical compound [Zr+4].[O-][Si]([O-])([O-])[O-] GFQYVLUOOAAOGM-UHFFFAOYSA-N 0.000 description 2
- ODINCKMPIJJUCX-UHFFFAOYSA-N Calcium oxide Chemical compound [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 1
- 229910052684 Cerium Inorganic materials 0.000 description 1
- 229910052691 Erbium Inorganic materials 0.000 description 1
- 229910052777 Praseodymium Inorganic materials 0.000 description 1
- 229910052772 Samarium Inorganic materials 0.000 description 1
- 229910052771 Terbium Inorganic materials 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 229910052787 antimony Inorganic materials 0.000 description 1
- 238000001354 calcination Methods 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 229910000420 cerium oxide Inorganic materials 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 238000011960 computer-aided design Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000004993 emission spectroscopy Methods 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 238000010335 hydrothermal treatment Methods 0.000 description 1
- 238000009616 inductively coupled plasma Methods 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 239000013067 intermediate product Substances 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 238000000462 isostatic pressing Methods 0.000 description 1
- 238000003475 lamination Methods 0.000 description 1
- 230000007257 malfunction Effects 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- BMMGVYCKOGBVEV-UHFFFAOYSA-N oxo(oxoceriooxy)cerium Chemical compound [Ce]=O.O=[Ce]=O BMMGVYCKOGBVEV-UHFFFAOYSA-N 0.000 description 1
- SIWVEOZUMHYXCS-UHFFFAOYSA-N oxo(oxoyttriooxy)yttrium Chemical compound O=[Y]O[Y]=O SIWVEOZUMHYXCS-UHFFFAOYSA-N 0.000 description 1
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229910052573 porcelain Inorganic materials 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 238000007790 scraping Methods 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229910052718 tin Inorganic materials 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 238000002834 transmittance Methods 0.000 description 1
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 229910001928 zirconium oxide Inorganic materials 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B18/00—Layered products essentially comprising ceramics, e.g. refractory products
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
- C04B35/48—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics based on zirconium or hafnium oxides, zirconates, zircon or hafnates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/08—Artificial teeth; Making same
- A61C13/082—Cosmetic aspects, e.g. inlays; Determination of the colour
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/08—Artificial teeth; Making same
- A61C13/083—Porcelain or ceramic teeth
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C5/00—Filling or capping teeth
- A61C5/70—Tooth crowns; Making thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C7/00—Orthodontics, i.e. obtaining or maintaining the desired position of teeth, e.g. by straightening, evening, regulating, separating, or by correcting malocclusions
- A61C7/12—Brackets; Arch wires; Combinations thereof; Accessories therefor
- A61C7/14—Brackets; Fixing brackets to teeth
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C8/00—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C8/00—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools
- A61C8/0012—Means to be fixed to the jaw-bone for consolidating natural teeth or for fixing dental prostheses thereon; Dental implants; Implanting tools characterised by the material or composition, e.g. ceramics, surface layer, metal alloy
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/01—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics
- C04B35/48—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on oxide ceramics based on zirconium or hafnium oxides, zirconates, zircon or hafnates
- C04B35/486—Fine ceramics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/0003—Making bridge-work, inlays, implants or the like
- A61C13/0022—Blanks or green, unfinished dental restoration parts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61C—DENTISTRY; APPARATUS OR METHODS FOR ORAL OR DENTAL HYGIENE
- A61C13/00—Dental prostheses; Making same
- A61C13/08—Artificial teeth; Making same
- A61C13/09—Composite teeth, e.g. front and back section; Multilayer teeth
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3217—Aluminum oxide or oxide forming salts thereof, e.g. bauxite, alpha-alumina
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3224—Rare earth oxide or oxide forming salts thereof, e.g. scandium oxide
- C04B2235/3225—Yttrium oxide or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3231—Refractory metal oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3232—Titanium oxides or titanates, e.g. rutile or anatase
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3231—Refractory metal oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3239—Vanadium oxides, vanadates or oxide forming salts thereof, e.g. magnesium vanadate
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3231—Refractory metal oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3244—Zirconium oxides, zirconates, hafnium oxides, hafnates, or oxide-forming salts thereof
- C04B2235/3246—Stabilised zirconias, e.g. YSZ or cerium stabilised zirconia
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3231—Refractory metal oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3244—Zirconium oxides, zirconates, hafnium oxides, hafnates, or oxide-forming salts thereof
- C04B2235/3248—Zirconates or hafnates, e.g. zircon
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/32—Metal oxides, mixed metal oxides, or oxide-forming salts thereof, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/327—Iron group oxides, their mixed metal oxides, or oxide-forming salts thereof
- C04B2235/3279—Nickel oxides, nickalates, or oxide-forming salts thereof
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/34—Non-metal oxides, non-metal mixed oxides, or salts thereof that form the non-metal oxides upon heating, e.g. carbonates, nitrates, (oxy)hydroxides, chlorides
- C04B2235/3418—Silicon oxide, silicic acids, or oxide forming salts thereof, e.g. silica sol, fused silica, silica fume, cristobalite, quartz or flint
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/96—Properties of ceramic products, e.g. mechanical properties such as strength, toughness, wear resistance
- C04B2235/9646—Optical properties
- C04B2235/9653—Translucent or transparent ceramics other than alumina
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/70—Aspects relating to sintered or melt-casted ceramic products
- C04B2235/96—Properties of ceramic products, e.g. mechanical properties such as strength, toughness, wear resistance
- C04B2235/9646—Optical properties
- C04B2235/9661—Colour
Definitions
- the present invention claims priority based on Japanese patent application: Japanese Patent Application No. 2014-265066 (filed on Dec. 26, 2014), the entire contents of which are incorporated herein by reference. It shall be.
- the present invention relates to a zirconia sintered body.
- the present invention also relates to a zirconia composition and a zirconia calcined body for producing the zirconia sintered body.
- this invention relates to the dental product containing a zirconia sintered compact.
- ceramics such as zirconia have been used as dental prostheses (coated crowns, crowns, crowns, dentures, etc.) in place of metal from the viewpoint of aesthetics and safety.
- Patent Document 1 discloses a highly transparent zirconia sintered body.
- the translucent yttria-containing zirconia sintered body described in Patent Document 1 is made of zirconia containing more than 4 mol% and not more than 7 mol% of yttria, and has a sintered body particle size of 2.0 ⁇ m or less and a relative density of 99.
- the total light transmittance for visible light having a wavelength of 600 nm at a thickness of 5% or more and 1 mm is 40% or more.
- FIG. 4 is a schematic diagram showing an example of use of a dental prosthesis.
- the zirconia sintered body is manufactured as a dental prosthesis (crown) 201.
- the dental prosthesis 201 is placed on the patient's abutment teeth 202 protruding from the gums 203.
- the content of the stabilizer when the content of the stabilizer (particularly yttria) increases, the transparency of the zirconia sintered body tends to increase.
- the content of the stabilizer if the content of the stabilizer is different, the shrinkage rate during sintering of the partially stabilized zirconia will be different. Therefore, even if a zirconia sintered body whose transparency is changed by partially changing the content of the stabilizer is used, since the shrinkage rate is partially different, the zirconia sintered body has defects. It will occur. For this reason, the zirconia sintered compact used for the dental prosthesis 201 cannot be produced by changing the content of the stabilizer.
- the abutment tooth 202 is transparent from the outside of the dental prosthesis 201. I can see it. In this case, the treated tooth (that is, a part of the dental prosthesis 201) appears dark. Therefore, it is desirable that the transparency of the dental prosthesis 201 can be adjusted according to the color of the patient's abutment tooth.
- a partially stabilized zirconia sintered body containing 4 mol% to 7 mol% yttria as a stabilizer is provided.
- the zirconia sintered body contains a light shielding material.
- the zirconia sintered body has a first region and a second region having a higher light shielding material content than the first region. The difference between the yttria content in the first region and the yttria content in the second region is 1 mol% or less.
- a composition containing partially stabilized zirconia containing 4 mol% to 7 mol% yttria as a stabilizer and a light shielding material containing partially stabilized zirconia containing 4 mol% to 7 mol% yttria as a stabilizer and a light shielding material.
- the composition has a first region and a second region in which the content of the light shielding material is higher than that of the first region.
- the light shielding material is at least one selected from the group including a composite oxide of silicon oxide, aluminum oxide, titanium oxide, zirconium and silicon.
- a calcined body containing partially stabilized zirconia containing 4 mol% to 7 mol% yttria as a stabilizer and a light shielding material.
- the calcined body has a first region and a second region in which the content of the light shielding material is higher than that of the first region.
- the light shielding material is at least one selected from the group including a composite oxide of silicon oxide, aluminum oxide, titanium oxide, zirconium and silicon.
- a dental product having the zirconia sintered body according to the first aspect is provided.
- a zirconia sintered body having a transparency change similar to that of natural teeth can be produced. Even if the transparency is changed, a zirconia sintered body in which generation of defects is suppressed can be obtained. Moreover, it is possible to produce a dental product that has the same appearance as that of a natural tooth but is difficult to see through the abutment tooth according to the color of the abutment tooth of the patient.
- the schematic diagram which shows an example of the zirconia sintered compact, calcined body, and composition which concern on 1st Embodiment of this indication.
- the schematic diagram which shows an example of the zirconia sintered compact, calcined body, and composition which concern on 2nd Embodiment of this indication.
- the schematic diagram which shows an example of the zirconia sintered compact, calcined body, and composition which concern on 3rd Embodiment of this indication.
- the schematic sectional drawing which shows the usage example of a dental prosthesis.
- the L * value in chromaticity according to the L * a * b * color system measured with white as the background is defined as the first L * value and measured with black as the background.
- L * a * b * a L * value in the chromaticity due to color system and the second L * value when the value obtained by deducting the second L * value from the first L * value as [Delta] L, the first ⁇ L in this area is larger than ⁇ L in the second area.
- ⁇ L in the first region is 0.8 or more larger than ⁇ L in the second region.
- ⁇ L in the first region is 8-12.
- ⁇ L in the second region is 4 to 11.
- ⁇ L in the second region is 7.5 or less.
- the first region and the second region are stacked.
- the zirconia sintered body further has a third region between the first region and the second region.
- ⁇ L of the third region is larger than ⁇ L of the second region and smaller than ⁇ L of the first region.
- the content rate of the light shielding material in the third region is higher than the content rate of the light shielding material in the first region and lower than the content rate of the light shielding material in the second region.
- the zirconia sintered body further has a fourth region between the third region and the second region.
- the ⁇ L of the fourth region is larger than the ⁇ L of the second region and smaller than the ⁇ L of the third region.
- the content rate of the light shielding material in the fourth region is higher than the content rate of the light shielding material in the third region and lower than the content rate of the light shielding material in the second region.
- the thickness of the first region is 30% to 40% of the total thickness in the stacking direction of each region.
- the thickness of the second region is 30% to 40% of the whole.
- the thickness of the third region is 10% to 20% of the whole.
- the thickness of the fourth region is 10% to 20% of the whole.
- the light shielding material is at least one selected from the group comprising silicon oxide, aluminum oxide, titanium oxide, zirconium and silicon composite oxide, a colorant, and a fluorescent agent. .
- the partially stabilized zirconia contains 5.3 mol% to 6.2 mol% yttria as a stabilizer.
- the composition further includes a third region between the first region and the second region.
- the content rate of the light shielding material in the third region is higher than the content rate of the light shielding material in the first region, and is lower than the content rate of the light shielding material in the second region.
- the composition further includes a fourth region between the third region and the second region.
- the content rate of the light shielding material in the fourth region is higher than the content rate of the light shielding material in the third region, and is lower than the content rate of the light shielding material in the second region.
- the variation in the yttria content in each region is 1 mol% or less.
- the composition has a plate shape having a first surface and a second surface opposite to the first surface.
- the first region is on the first surface side.
- the second region is on the second surface side.
- the calcined body further has a third region between the first region and the second region.
- the content rate of the light shielding material in the third region is higher than the content rate of the light shielding material in the first region, and is lower than the content rate of the light shielding material in the second region.
- the calcined body further includes a fourth region between the third region and the second region.
- the content rate of the light shielding material in the fourth region is higher than the content rate of the light shielding material in the third region, and is lower than the content rate of the light shielding material in the second region.
- the variation in the content of yttria in each region is 1 mol% or less.
- the calcined body has a plate-like shape having a first surface and a second surface opposite to the first surface.
- the first region is on the first surface side.
- the second region is on the second surface side.
- a calcined body obtained by firing the composition according to the second viewpoint at 800 ° C. to 1200 ° C. is provided.
- a zirconia sintered body obtained by firing the composition according to the second viewpoint at 1400 ° C. or higher.
- a zirconia sintered body obtained by firing the calcined body according to the third viewpoint at 1400 ° C. or higher.
- the dental product has a crown shape.
- the first region is arranged on the cut end side of the crown shape.
- region is distribute
- the second region covers at least a part of the side surface of the abutment tooth.
- the dental product is a dental prosthesis, an orthodontic product, or a dental implant product.
- the zirconia sintered body according to the first embodiment of the present disclosure is a sintered body in which partially stabilized zirconia crystal particles containing zirconium oxide (ZrO 2 ; zirconia) and a stabilizer thereof are mainly sintered, and the partially stabilized zirconia is used as a matrix phase.
- the main crystal phase of zirconia is at least one of a tetragonal system and a cubic system.
- Zirconia may contain both tetragonal and cubic systems. It is preferable that the zirconia sintered body does not substantially contain a monoclinic system in a stage where the hydrothermal treatment test is not performed.
- the zirconia sintered body of the present disclosure includes not only a sintered body obtained by sintering molded zirconia particles under normal pressure or non-pressurization, but also HIP (Hot Isostatic Pressing) processing, etc. A sintered body densified by high-temperature pressure treatment is also included.
- Examples of the stabilizer in partially stabilized zirconia include calcium oxide (CaO), magnesium oxide (MgO), yttrium oxide (Y 2 O 3 ) (hereinafter referred to as “yttria”), cerium oxide (CeO 2 ), and the like. The oxide of this is mentioned.
- yttria yttrium oxide
- CeO 2 cerium oxide
- the content of yttria is preferably 4 mol% to 7 mol%, and more preferably 5.3 mol% to 6.2 mol%, for example, with respect to partially stabilized zirconia. .
- the phase transition to the monoclinic crystal can be suppressed and the transparency of the zirconia sintered body can be increased.
- the content of the stabilizer in the zirconia sintered body can be measured by, for example, inductively coupled plasma (ICP) emission spectroscopy, fluorescent X-ray analysis, or the like. Note that zirconia partially stabilized by adding a stabilizer is called partially stabilized zirconia (PSZ).
- the stabilizer is uniformly distributed. That is, in the zirconia sintered body, the content of the stabilizer is preferably constant. For example, in the zirconia sintered body, it is preferable that the content of the stabilizer is not changed stepwise or partially. This is because when the content of the stabilizer is partially different, the shrinkage rate during sintering is different, and a defect occurs in the zirconia sintered body.
- the variation of the stabilizer is preferably 1 mol% or less, and more preferably 0.5 mol% or less.
- Transparency of zirconia sintered body can be expressed using the L * values of chromaticity (color space) in the L * a * b * color system (JISZ8729).
- L * a * b * table in which a sample manufactured as in the examples described later was measured with the sample background (underlay) white (the opposite side of the sample from the measurement device was white) Let the L * value of the color system be the first L * value.
- L * a * b * color system measured for the same sample for which the first L * value was measured, with the sample background (underlay) set to black (with the opposite side of the sample to the measuring device black) the L * value and the second of the L * value.
- the difference between the first L * value and the second L * value is denoted as [Delta] L.
- ⁇ L represents the transparency of the zirconia sintered body. A large ⁇ L indicates that the zirconia sintered body has high transparency, and a small ⁇ L indicates that the zirconia sintered body has low transparency.
- a concealment ratio measuring sheet used for measurement related to the paint can be used.
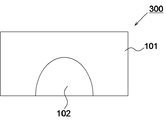
- FIG. 1 is a schematic diagram illustrating an example of a zirconia sintered body according to the first embodiment of the present disclosure.
- the zirconia sintered body 100 has a first region 101 and a second region 102 having different transparency.
- the transparency of the first region 101 is higher than the transparency of the second region 102.
- ⁇ L of the first region 101 is preferably 8 or more, and more preferably 9 or more.
- ⁇ L of the first region 101 is preferably 12 or less, and more preferably 11 or less.
- ⁇ L of the second region 102 is preferably 4 or more, and more preferably 6 or more, for example.
- ⁇ L of the second region 102 is, for example, preferably 11 or less, and more preferably 10 or less.
- ⁇ L of the second region 102 is preferably 7.5 or less, and more preferably 7 or less. 6.5 or less is more preferable.
- the difference between ⁇ L of the first region 101 and ⁇ L of the second region 102 is preferably 0.8 or more, more preferably 1 or more, more preferably 1.5 or more, and 2 or more. More preferably.
- the difference between ⁇ L of the first region 101 and ⁇ L of the second region 102 is preferably 4 or less, and more preferably 3 or less.
- the content difference of the stabilizer (for example, yttria) in the first region 101 and the second region 102 is small.
- the difference in the content of the stabilizer in the first region 101 and the second region 102 is preferably 1 mol% or less, more preferably 0.5 mol%, and substantially no significant difference can be detected. And more preferred.
- the first region 101 and the second region 102 are preferably layered.
- the first region 101 and the second region 102 are preferably stacked.
- the transparency is preferably lowered stepwise from the first region 101 toward the second region 102.
- the thickness (T1 + T2) of the zirconia sintered body can be set to 10 mm to 20 mm, for example.
- the thickness T1 of the first region 101 can be 30% to 70% or 40% to 60% of the thickness of the zirconia sintered body (T1 + T2).
- the thickness T2 of the second region 102 may be 30% to 70% or 40% to 60% of the thickness of the zirconia sintered body (T1 + T2).
- the zirconia sintered body 100 can contain a light shielding material that reduces the transparency of the zirconia sintered body.
- the content of the light shielding material is preferably different partially or stepwise.
- the second region 102 can contain a light shielding material that reduces the transparency of the zirconia sintered body.
- the first region 101 may contain a light shielding material. It is preferable that the content ratio of the light-shielding material tends to decrease stepwise from the second region 102 toward the first region 101 side.
- the content of the light shielding material in the second region 102 is preferably higher than the content of the light shielding material in the first region 101.
- the light shielding material is preferably one that does not significantly reduce the physical properties such as strength and fracture toughness of the zirconia sintered body.
- the light shielding material include silicon oxide (SiO 2 ; silica), aluminum oxide (Al 2 O 3 ; alumina), titanium oxide (TiO 2 ; titania), zirconium and silicon composite oxide (for example, zircon (ZrSiO 4 )).
- a colorant (pigment), and at least one selected from the group comprising a fluorescent agent can be used.
- the colorant include P, Ti, V, Cr, Mn, Fe, Co, Ni, Zn, Y, Zr, Sn, Sb, Bi, Ce, Pr, Sm, Eu, Gd, Tb, and Er.
- the light shielding material can be used in combination of a plurality of compounds. The content rate of a light shielding material can be adjusted according to the kind of light shielding material.
- the light shielding material contained in the second region 102 and the light shielding material contained in the first region 101 may be the same or different.
- the L * value is preferably in the range of 64 to 76.
- the a * value is preferably in the range of ⁇ 6 to 3.
- the b * value is preferably in the range of 3 to 27.
- a dental prosthesis covering the abutment tooth in a region with low transparency can be produced.
- the tendency of the transparency change of a dental prosthesis can be made to resemble the tendency of the transparency change in a natural tooth.
- a zirconia sintered body according to the second embodiment of the present disclosure will be described.
- a zirconia sintered body having two regions has been described.
- a zirconia sintered body having three or more regions will be described.
- FIG. 2 is a schematic diagram illustrating an example of a zirconia sintered body according to the second embodiment of the present disclosure.
- the zirconia sintered body 200 further includes a third region 103 and a fourth region 104 between the first region 101 and the second region 102 in addition to the first region 101 and the second region 102.
- the four regions have a layered shape, and are laminated in the order of the second region 102, the fourth region 104, the third region 103, and the first region 101. ing.
- the transparency of the first region 101 and the second region 102 can be the same as in the first embodiment.
- the transparency of the third region 103 and the fourth region 104 is preferably lower than the transparency of the first region 101 and higher than the transparency of the second region 102. It is preferable that ⁇ L of each region decreases in order from the first region 101 toward the second region 102. Thereby, the transparency change similar to a natural tooth is reproducible.
- the fourth region 104 is preferably transparent so that the abutment teeth are difficult to see through.
- the transparency of the third region 103 is lower than the transparency of the first region 101.
- the transparency of the fourth region 104 is preferably lower than the transparency of the third region 103.
- the transparency of the second region 102 is preferably lower than the transparency of the fourth region 104.
- ⁇ L of the first region 101 is preferably 8 or more, and more preferably 9 or more.
- ⁇ L of the first region 101 is preferably 12 or less, and more preferably 11 or less.
- ⁇ L of the third region 103 is preferably 5 or more, and more preferably 6 or more.
- ⁇ L of the third region 103 is preferably 11 or less, and more preferably 10 or less.
- ⁇ L of the fourth region 104 is preferably 6 or more, and more preferably 7 or more.
- ⁇ L of the fourth region 104 is preferably 10 or less, and more preferably 9 or less.
- ⁇ L of the second region 102 is preferably 7 or more, and more preferably 8 or more.
- ⁇ L of the second region 102 is preferably 12 or less, and more preferably 11 or less.
- the ⁇ L of the third region 103 is preferably smaller than the ⁇ L of the first region 101 by 0.3 or more, more preferably 0.5 or more.
- ⁇ L of the fourth region 104 is preferably 0.3 or more smaller than ⁇ L of the third region 103, and more preferably 0.5 or more.
- ⁇ L of the second region 102 is preferably 0.2 or more smaller than ⁇ L of the fourth region 104, more preferably 0.3 or more.
- the thickness of the zirconia sintered body (thickness in the stacking direction of each region; T1 + T3 + T4 + T2) can be, for example, 10 mm to 20 mm.
- the thickness T1 of the first region 101 can be 30% to 40% of the thickness (T1 + T3 + T4 + T2) of the zirconia sintered body.
- the thickness T3 of the third region 103 can be 10% to 20% of the thickness of the zirconia sintered body (T1 + T3 + T4 + T2).
- the thickness T4 of the fourth region 104 can be 10% to 20% of the thickness of the zirconia sintered body (T1 + T3 + T4 + T2).
- the thickness T2 of the second region 102 may be 30% to 40% of the thickness of the zirconia sintered body (T1 + T3 + T4 + T2).
- each region can contain a light shielding material. It is preferable that the content ratio of the light shielding material in each region tends to decrease stepwise from the second region 102 toward the first region 101.
- the content ratio of the light shielding material in the second region 102 is preferably higher than the content ratio of the light shielding material in the fourth region 104.
- the content ratio of the light shielding material in the fourth region 104 is preferably higher than the content ratio of the light shielding material in the third region 103.
- the content ratio of the light shielding material in the third region 103 is preferably higher than the content ratio of the light shielding material in the first region 101.
- FIG. 2 shows a form in which the zirconia sintered body 200 has four regions (layers), but the number of regions is not limited to four.
- the zirconia sintered body may have three regions, or may have five or more regions.
- the same effect as that of the first embodiment can be obtained.
- the dental prosthesis which has a more natural transparency change can be produced.
- FIG. 3 is a schematic diagram illustrating an example of a zirconia sintered body according to the third embodiment of the present disclosure.
- the zirconia sintered body 300 has a first region 101 and a second region 102.
- the transparency of the first region 101 and the second region 102 can be the same as in the first embodiment.
- the second region 102 can have a shape that covers the side surface of the abutment tooth.
- the second region 102 has a semicircular shape or a semielliptical shape.
- the same effect as in the first embodiment can be obtained.
- the dental prosthesis which can hide an abutment tooth in the area
- the change in the transparency and the content of the light shielding material between adjacent regions does not necessarily need to form a clear boundary, and may be continuous.
- the composition and the calcined body serve as a precursor (intermediate product) of the above-described zirconia sintered body of the present invention.
- the calcined body is obtained by firing (that is, calcining) the composition at a temperature that does not lead to sintering.
- the calcined body includes a molded product.
- a dental product for example, a crown-shaped prosthesis obtained by processing a calcined zirconia disk with a CAD / CAM (Computer-Aided Design / Computer-Aided Manufacturing) system is also included in the calcined body.
- the composition of the present invention includes a powder, a fluid obtained by adding the powder to a solvent, and a molded body obtained by molding the powder into a predetermined shape. That is, the composition may be in the form of a powder, or a paste or a wet composition (that is, it may be in a solvent or may contain a solvent).
- the composition may contain additives such as a binder and a pigment. In addition, in the calculation of the content rate, the mass of additives such as a solvent and a binder is not considered.
- the composition of the present invention is a molded body, it may be molded by any molding method, and may be molded by, for example, press molding, injection molding, stereolithography, or multistage. It may be molded.
- the composition of the present invention may be subjected to press molding and further subjected to CIP (Cold ⁇ ⁇ ⁇ Isostatic Pressing).
- composition and the calcined body contain partially stabilized zirconia and a light shielding material.
- the kind and content of the stabilizer in the composition and calcined body can be the same as described above.
- the type of the light shielding material can be the same as described above.
- the variation in the content of the stabilizer is small.
- the variation in the content of the stabilizer is preferably 1 mol% or less, more preferably 0.5 mol%, and even more preferably when a significant difference cannot be detected.
- the molded composition (molded composition) and calcined body of the present disclosure have the same configuration as the zirconia sintered body to be produced.
- the molding composition and the calcined body when producing a zirconia sintered body as shown in FIGS. 1 to 3, the molding composition and the calcined body have the configuration and shape as shown in FIGS.
- each region of the molding composition and the calcined body is given a name corresponding to each region of the zirconia sintered body.
- the first region of the molding composition and the calcined body is a region corresponding to the first region of the zirconia sintered body.
- composition and the calcined body will be described by taking as an example a molding composition and a calcined body for producing a zirconia sintered body according to the second embodiment as shown in FIG.
- the molding composition and the calcined body have a first region 101 and a second region 102 in which the content of the light shielding material is different from that of the first region 101.
- the first region 101 and the second region 102 are preferably layered.
- the first region 101 and the second region 102 are preferably stacked.
- the molding composition and the calcined body can have a plate shape having a first surface and a second surface opposite to the first surface. In this case, the first region can be disposed on the first surface side.
- the second region can be disposed on the second surface side.
- the content ratio of the light-shielding material tends to decrease stepwise from the second region 102 toward the first region 101.
- the content ratio of the light shielding material in the second region 102 is preferably higher than the content ratio of the light shielding material in the first region 101.
- the third region 103 and the fourth region 104 are provided between the first region 101 and the second region 102. You can also have.
- the light shielding material content in the third region 103 and the fourth region 104 is preferably higher than the light shielding material content in the first region 101 and lower than the light shielding material content in the second region 102.
- the content ratio of the light shielding material in the second region 102 is preferably higher than the content ratio of the light shielding material in the fourth region 104.
- the content ratio of the light shielding material in the fourth region 104 is preferably higher than the content ratio of the light shielding material in the third region 103.
- the content ratio of the light shielding material in the third region 103 is preferably higher than the content ratio of the light shielding material in the first region 101.
- the thickness of each region in the molding composition and calcined body is preferably set so as to correspond to the thickness of each region in the zirconia sintered body to be produced.
- composition of the present disclosure becomes the calcined body of the present disclosure by firing the composition of the present invention at, for example, 800 ° C. to 1200 ° C. under normal pressure.
- composition and calcined body of the present disclosure become the zirconia sintered body of the present disclosure by firing at 1400 ° C. to 1650 ° C., for example, under normal pressure.
- the zirconia sintered body of the present disclosure can be produced.
- the dental product includes the zirconia sintered body or calcined body according to the above embodiment.
- Dental products include prosthetics such as ceramic frames and full cantour crowns.
- the dental prosthesis preferably has a crown shape.
- the dental prosthesis may further include porcelain (for example, a glass material) laminated on the zirconia sintered body or the calcined body.
- the dental prosthesis preferably has a second region on the cervical side.
- the dental prosthesis preferably has a first region on the cut end side. It is preferable that the dental prosthesis has higher transparency from the tooth neck side toward the cut end side.
- the second region is preferably formed so as to cover the side surface of the abutment tooth.
- the dental prosthesis can be formed so as to cover the side surface of the abutment tooth in the second and fourth regions having low transparency.
- the dental product can also include, for example, orthodontic products (for example, orthodontic brackets) and dental implant products (for example, dental implant abutments).
- orthodontic products for example, orthodontic brackets
- dental implant products for example, dental implant abutments
- zirconia sintered body a zirconia calcined body and a zirconia composition
- a method for producing a dental product will be described.
- the zirconia sintered body, the calcined body, and the composition will be described by taking as an example a form having first and second regions as shown in the figure.
- a zirconia and a stabilizer are wet mixed in water to form a slurry.
- the slurry is dried and granulated.
- the granulated product is calcined to produce a primary powder.
- the primary powder is divided into the number of layers.
- a predetermined amount of a light shielding material corresponding to the position of each layer is added to and mixed with each primary powder.
- zirconia is grind
- the slurry is dried and granulated to produce a secondary powder.
- a plurality of powders having different light shielding material contents are sequentially laminated.
- the layers are preferably stacked so that the addition rate of the light shielding material is increased or decreased stepwise in the order of stacking.
- the upper surface of the first layer of powder is flattened.
- a method of leveling the surface flat for example, a method of vibrating a mold or scraping the upper surface of the first layer of powder can be employed.
- the press treatment is not performed until all the layers are laminated.
- the powder of the second layer is filled on the powder of the first layer.
- a vibration is given to the mold. The vibration is transmitted to the powder in the mold.
- a mechanical vibration can be applied to the mold, the mold can be shaken manually, or the mold can be struck with a hammer or the like.
- the powder of the first layer and the powder of the second layer are partially mixed at the boundary between the powder of the first layer and the powder of the second layer. That is, in the case of mechanical vibration, the frequency, amplitude, etc. of the vibration are such that the powder particle size, particle size distribution, particle shape, etc. It can be set appropriately depending on the situation.
- the upper surface of the second layer powder is leveled in the same manner as the first layer powder. This operation is repeated until all layers are laminated.
- the mold is filled with the powder of the first layer to a predetermined thickness (for example, 25% to 45% of the total thickness).
- a predetermined thickness for example, 25% to 45% of the total thickness.
- the upper surface of the powder of the first layer is leveled, but no press treatment is performed.
- the powder of the second layer is filled on the first layer of powder to a predetermined thickness (for example, 5% to 25% of the total thickness).
- a vibration is given to the mold. By this vibration, a first boundary layer in which the first layer powder and the second layer powder are mixed is formed at the boundary between the upper surface of the first layer powder and the lower surface of the second layer powder. I guess that.
- the upper surface of the second layer of powder is leveled.
- the pressing treatment is not performed on the powder of the second layer.
- the powder of the third layer is filled on the second layer of powder to a predetermined thickness (for example, 5% to 25% of the total thickness).
- a vibration is given to the mold. This vibration forms a second boundary layer in which the second layer powder and the third layer powder are mixed at the boundary between the upper surface of the second layer powder and the lower surface of the third layer powder. I guess that.
- the upper surface of the third layer of powder is leveled flat. Before the powder filling of the fourth layer, the third layer powder is not pressed.
- the powder of the fourth layer is filled to a predetermined thickness (for example, 25% to 45% of the total thickness) on the powder of the third layer.
- a vibration is given to the mold. This vibration forms a third boundary layer in which the third layer powder and the fourth layer powder are mixed at the boundary between the upper surface of the third layer powder and the lower surface of the fourth layer powder. I guess that.
- press molding is performed to produce a molded product as the composition of the present invention.
- the molded product may be further subjected to CIP treatment.
- a boundary layer in which the upper and lower layer powders are mixed can be formed between adjacent layers. Conceivable. Thereby, in a sintered compact, the adhesiveness between adjacent layers can be improved. The amount of shrinkage or shrinkage rate during heat treatment can be made equal in each layer, preventing peeling between layers during heat treatment, and preventing the sintered body from deforming to the target shape. can do. Furthermore, the difference in transparency and color between adjacent layers can be reduced. Thereby, in a sintered compact, transparency and a color can be changed naturally in the lamination direction.
- the composition is fired at, for example, 1400 ° C. to 1600 ° C., preferably 1450 ° C. to 1550 ° C., to sinter the zirconia powder to produce the zirconia sintered body of the present invention.
- the composition is fired at, for example, 800 ° C. to 1200 ° C. to prepare a calcined body.
- the calcined body is fired at, for example, 1400 ° C. to 1650 ° C., preferably 1450 ° C. to 1600 ° C. to sinter the zirconia powder to produce the zirconia sintered body of the present invention.
- the molding may be performed by cutting or the like at the stage of the calcined body, or may be performed after sintering. Molding can be performed with a CAD / CAM system.
- the method for producing a dental product is the same as the method for producing a sintered body, except that the calcined body or the sintered body is formed into a dental product shape such as a crown shape.
- composition based on a laminated body of 4 layers, a calcined body, and the sintered compact were illustrated, it is not limited to 4 layers.
- drawings are for facilitating explanation of the positional relationship and direction of each point, and the shapes and dimensions are not limited to the forms shown in the drawings.
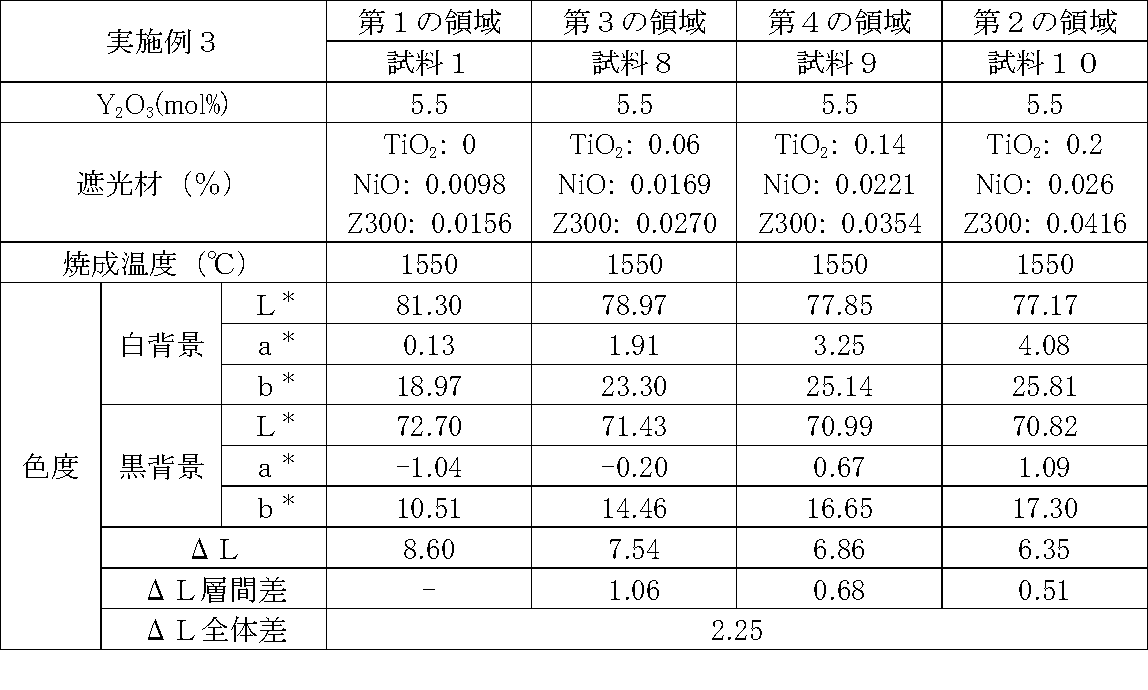
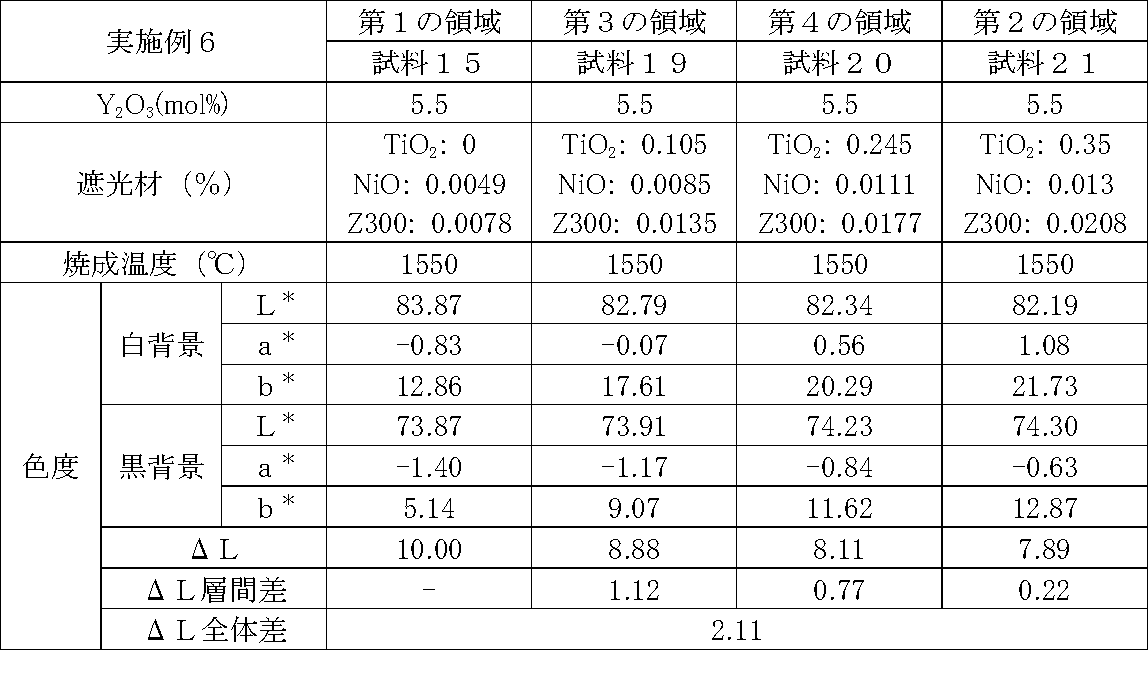
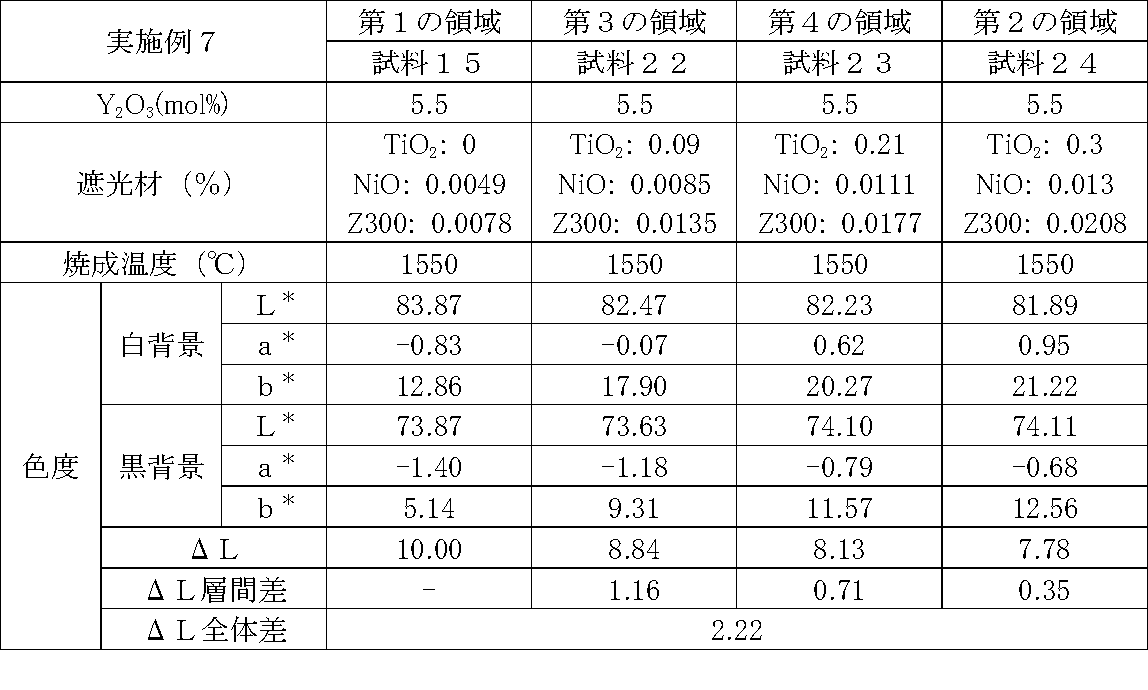
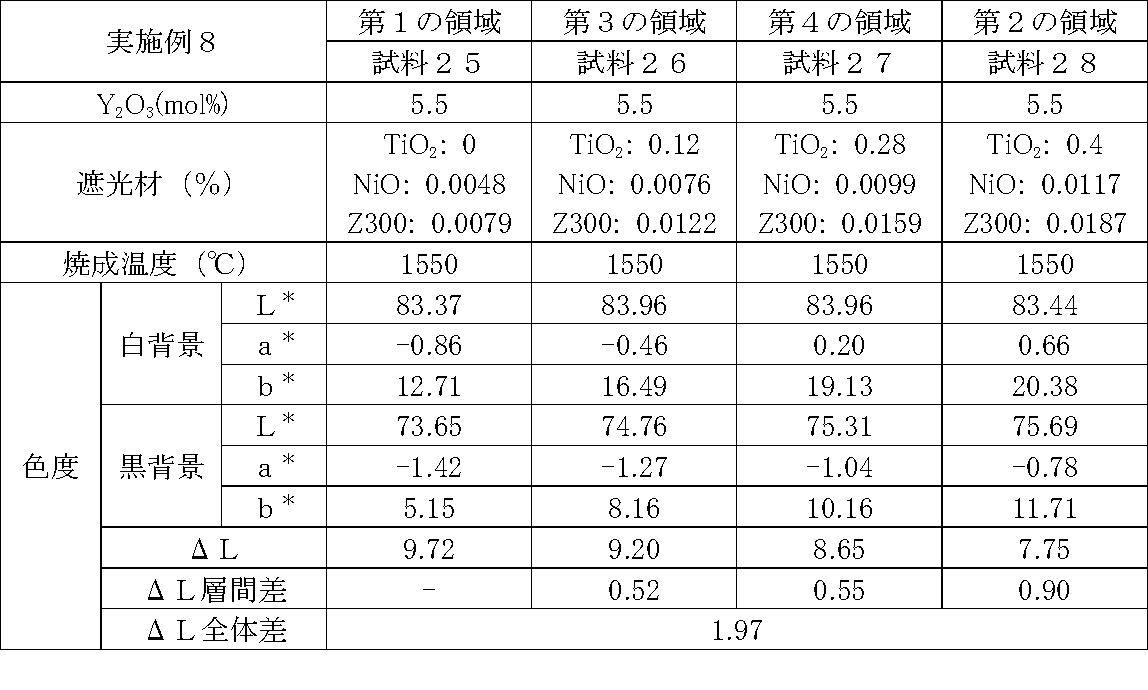
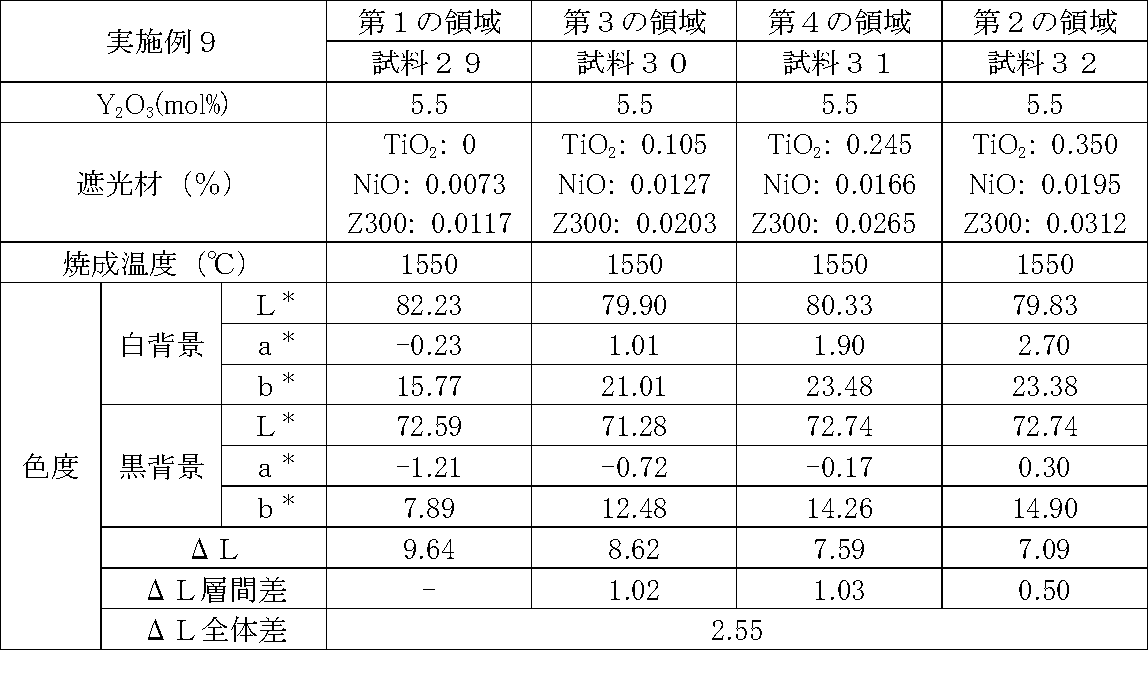
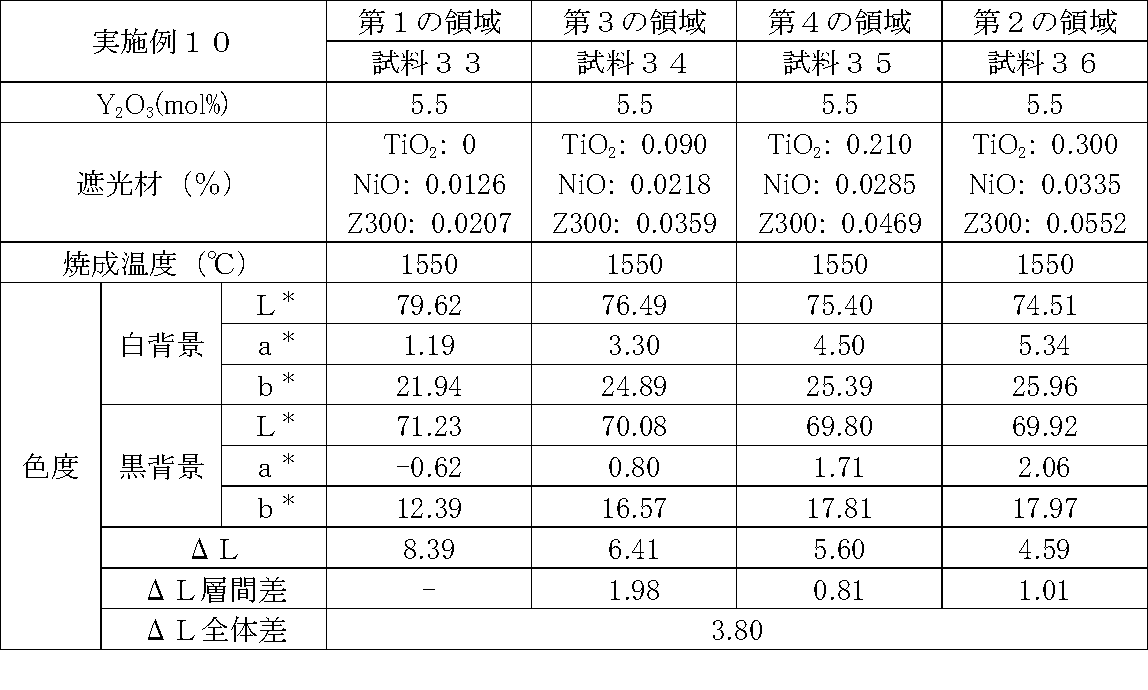
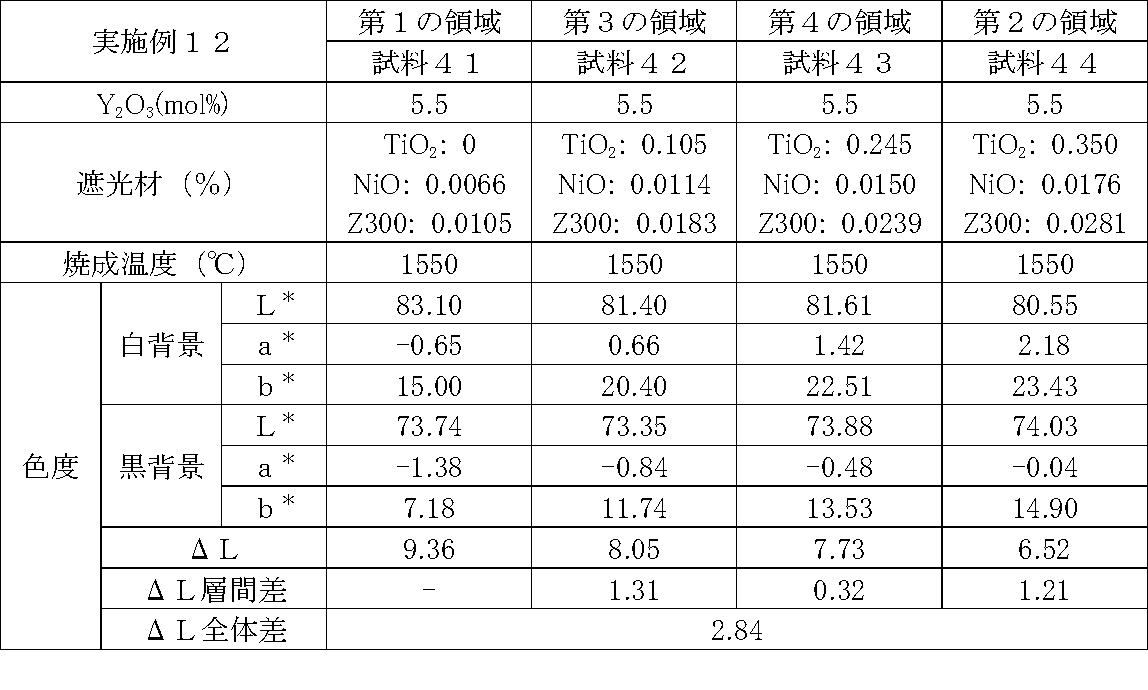
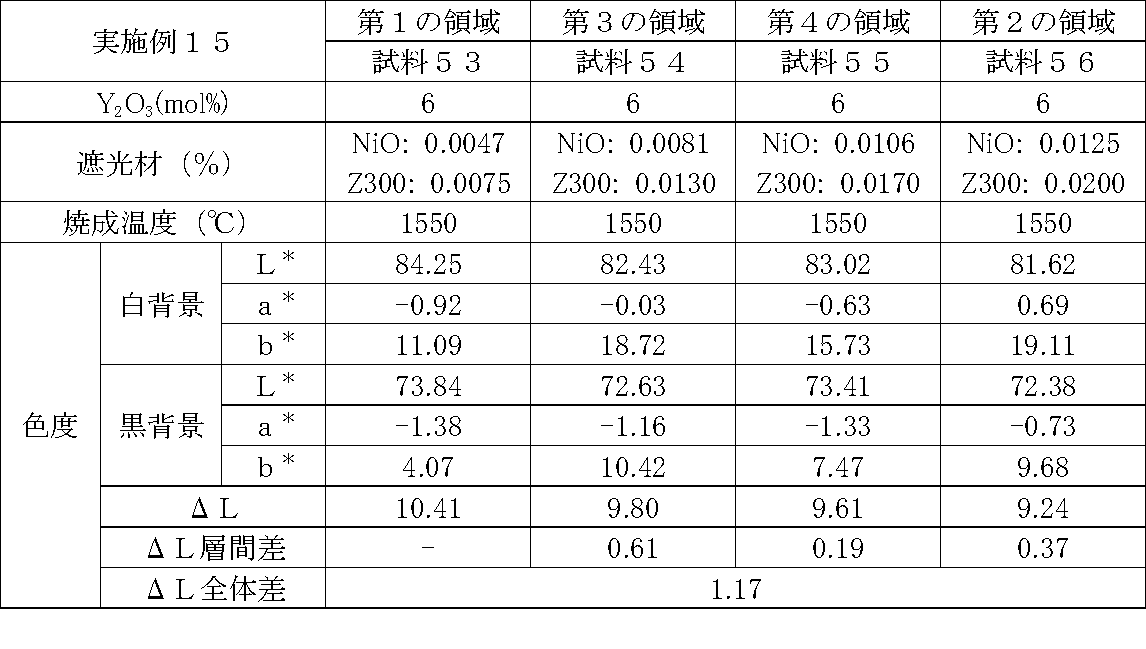
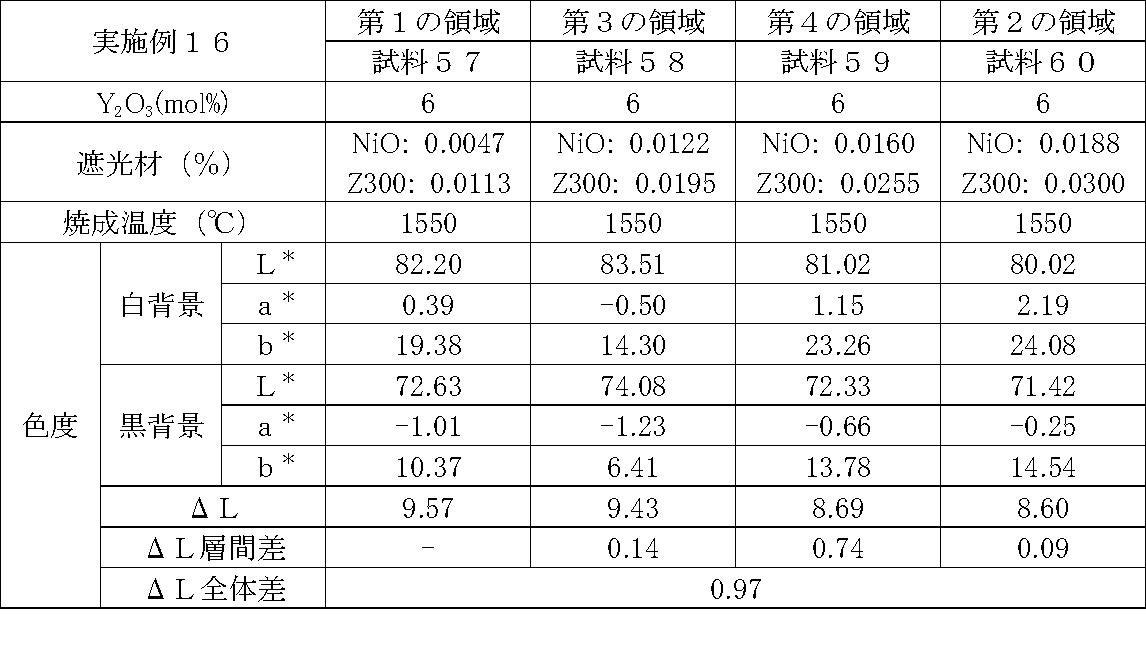
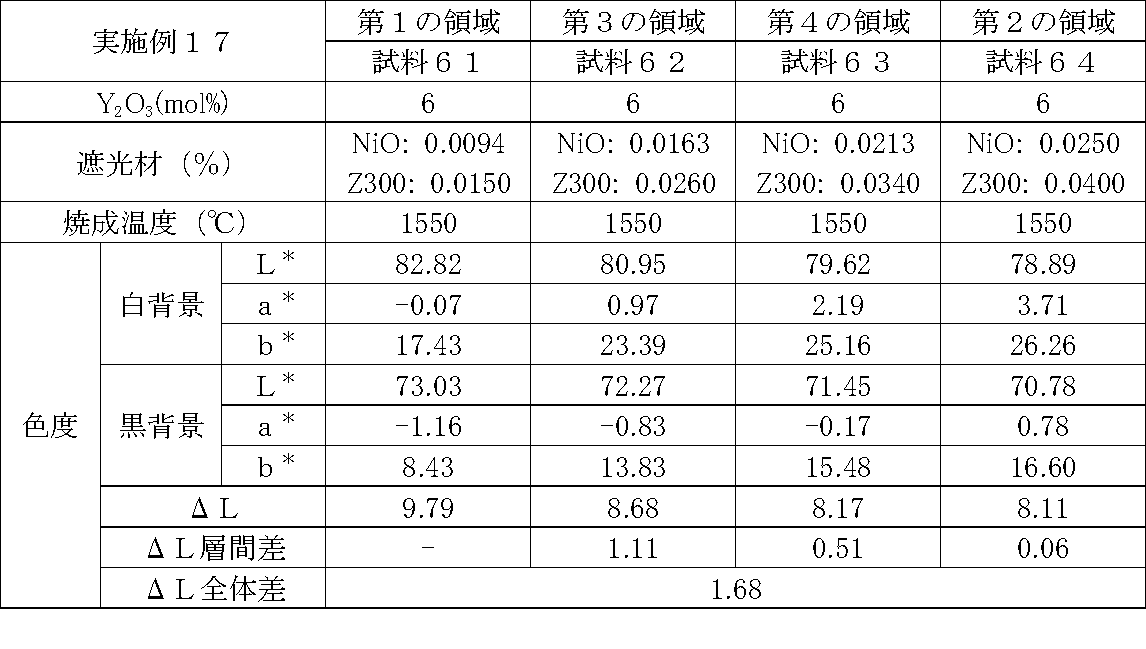
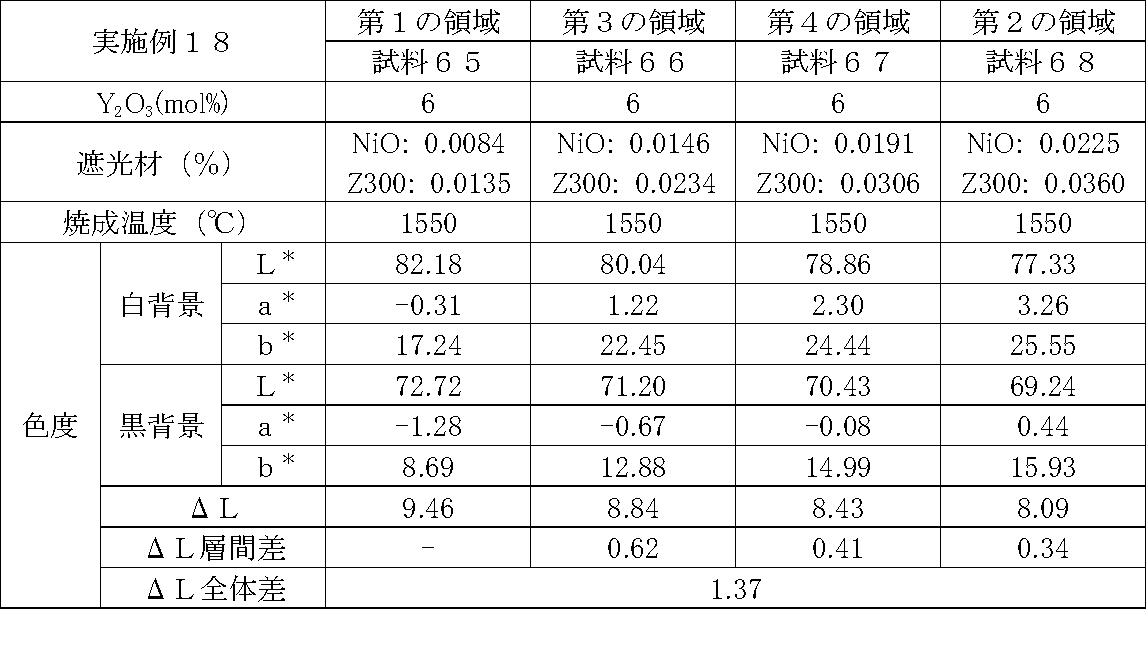
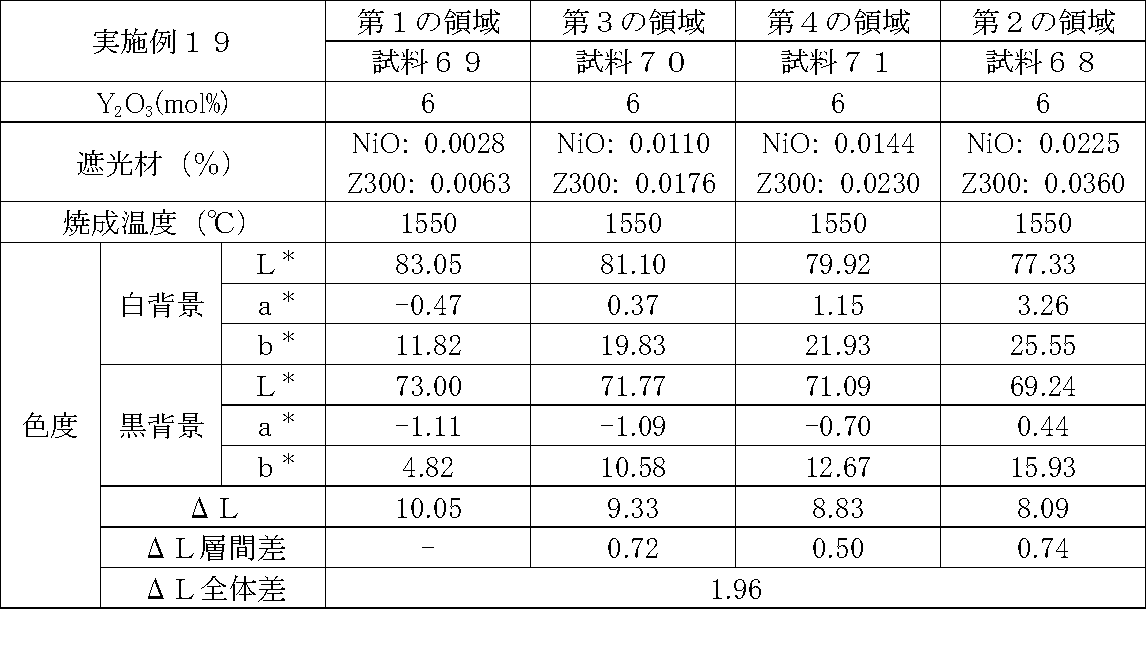
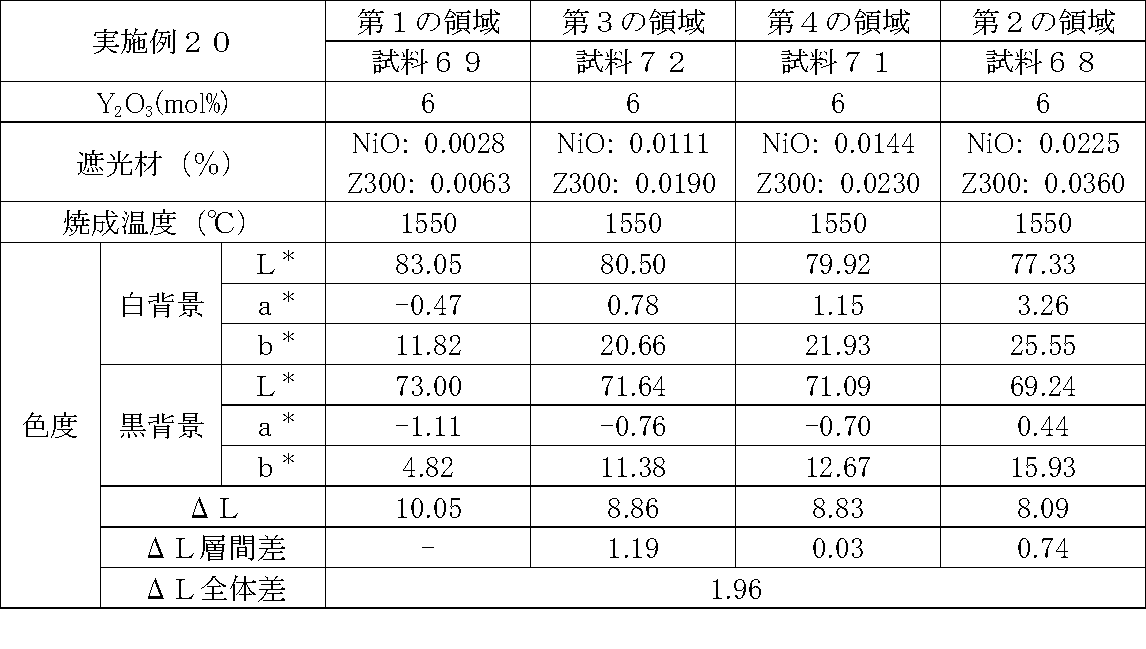
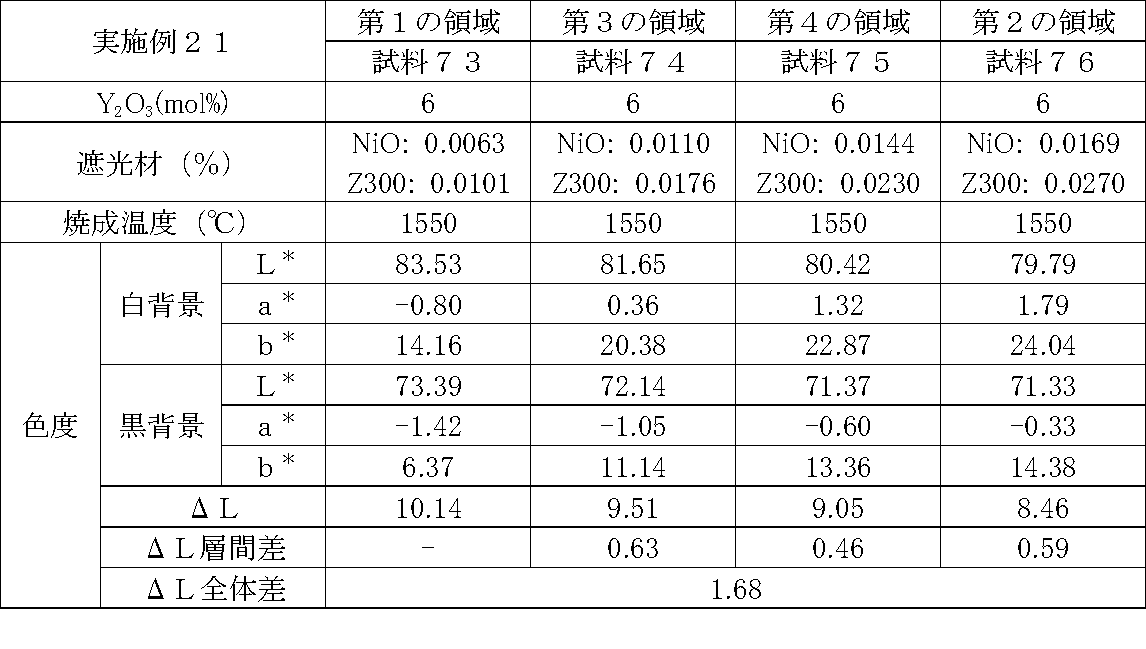
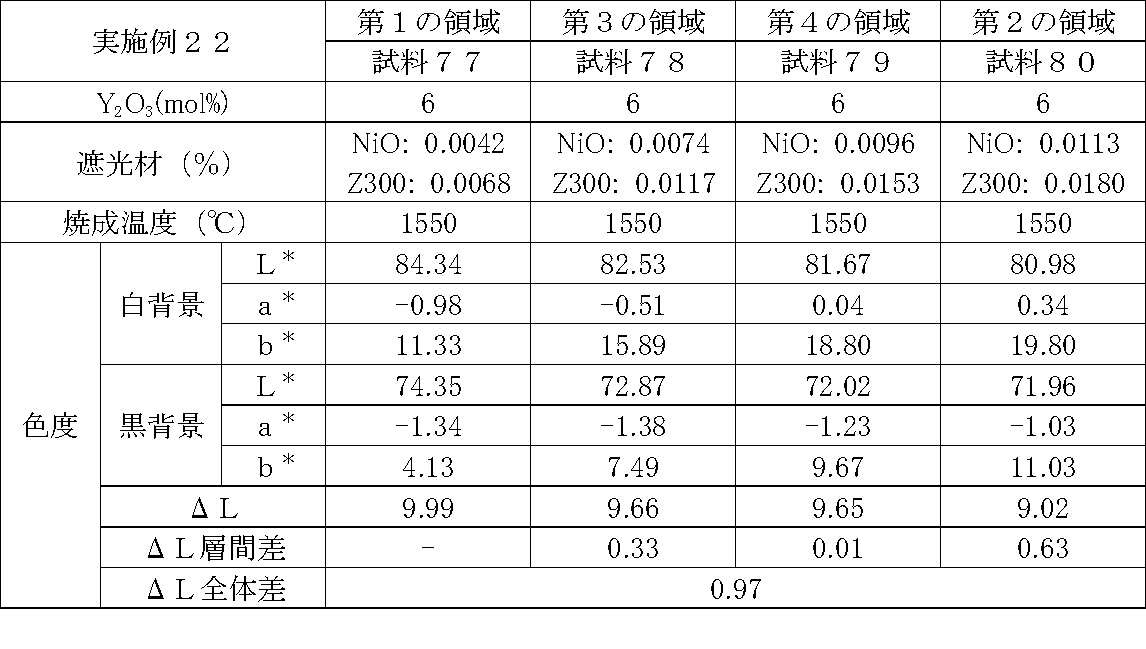
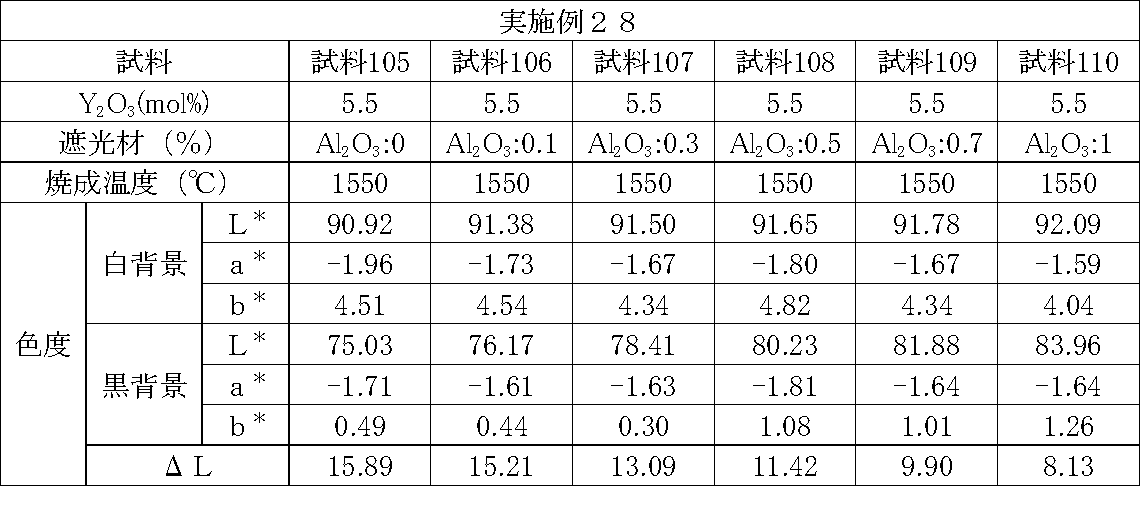
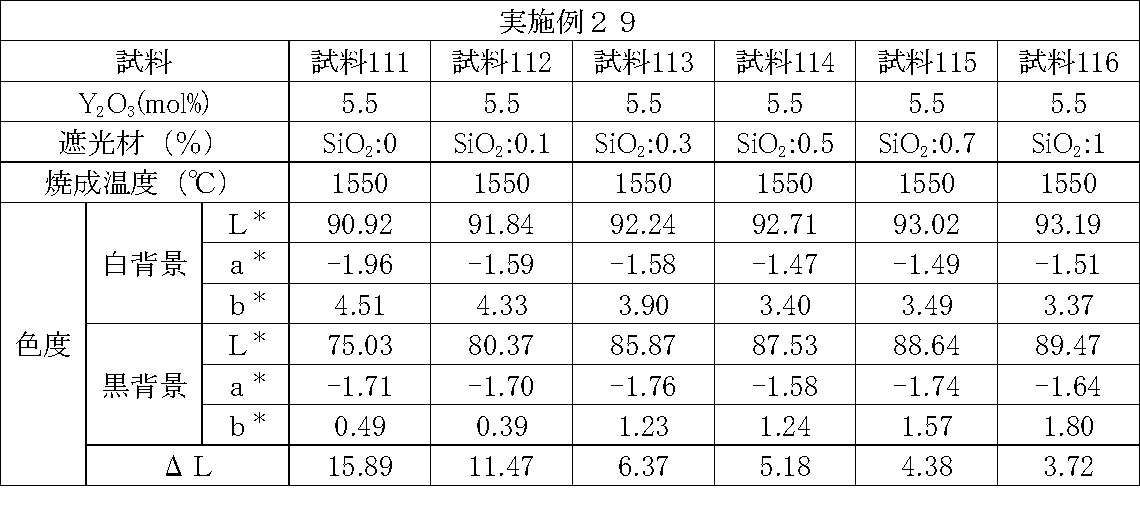
- Examples 1 to 25 A zirconia sintered body was produced, and the chromaticity of each sample was measured.
- samples of zirconia sintered bodies corresponding to the respective regions in the zirconia sintered body having a four-layer structure as in the second embodiment shown in FIG. 2 were prepared, and the chromaticity of each sample was measured. did.
- Data on the composition of the sample and measurement results are shown in Tables 1 to 25.
- the “white background” indicates chromaticity measured using a white hiding ratio measurement paper used for measurement relating to paint as a background (underlay).
- the “black background” indicates chromaticity measured using a white hiding ratio measurement paper used for measurement relating to paint as a background (underlay).
- ⁇ L is the difference between the L * value measured on a white background and the L * value measured on a black background.
- the “ ⁇ L interlayer difference” is a difference in ⁇ L between adjacent layers. The difference between the ⁇ L of the n layer and the ⁇ L of the (n + 1) layer is described in the (n + 1) layer column.
- the “ ⁇ L overall difference” is a difference between ⁇ L in the first region and ⁇ L in the second region. In Tables 1 to 25, the same sample number is assigned to the same sample.
- partially stabilized zirconia containing 5.5 mol% or 6 mol% of yttria as a stabilizer was prepared.
- the light shielding material shown in the table was added to and mixed with the partially stabilized zirconia.
- the addition amount of the light shielding material shown in the table is the mass of the light shielding material relative to the mass of the partially stabilized zirconia.
- the TiO 2 is 0.3%, indicating that the TiO 2 is added 0.3g against partially stabilized zirconia 100 g.
- Z300 is a complex oxide of zirconium and vanadium ((Zr, V) O 2 ); “Z-300 Yellow” manufactured by Nissho Pigment Industries Co., Ltd.).
- 1.3 g of the mixture was put in a cylindrical mold having a diameter of about 18 mm and molded at a pressure of 30 kN, and then subjected to a CIP treatment of 170 MPa for 1 minute to prepare a molding composition.
- the molding composition was fired at the firing temperature shown in the table for 2 hours to produce a disk-shaped zirconia sintered body.
- the surface of the sintered body was polished and finished to a mirror surface (# 2000 or more) to prepare a sample having a thickness of 1.2 mm.
- the chromaticity of each sample was measured using a chromaticity measuring machine (SPECTROTOPOMETER CM-3610A manufactured by KONIKA MINOLTA) and analysis software (Spectra Magic NX).
- the partially stabilized zirconia used for the samples in each region uses the same product in which yttria is uniformly mixed, and it is considered that no significant variation in the yttria content is detected between the samples in each region.
- Examples 1 to 25 basically, the addition amount of the light shielding material was increased from the sample corresponding to the first region to the sample corresponding to the second region. As a result, it was confirmed that ⁇ L sequentially decreased from the sample corresponding to the first region to the sample corresponding to the second region. Thereby, it was confirmed that if a composition of each layer is laminated to produce a zirconia sintered body, a zirconia sintered body whose transparency changes stepwise can be produced. That is, it was confirmed that a dental prosthesis having a more natural transparency change can be produced. Since the content of the stabilizer in each layer is equal, the shrinkage rate during sintering in each layer can be made equal. Thereby, generation
- the abutment tooth When a dental prosthesis having a ⁇ L of 7.5 or less in the second region is produced, the abutment tooth is transparent in the second region even if the color of the abutment tooth is dark (darkened). It was confirmed that it was not visible. Therefore, it was found that when it is not desired to watermark the abutment tooth, if the ⁇ L of the region covering the abutment tooth is set to 7.5 or less, the abutment tooth can be transparent.
- the difference in ⁇ L between layers was in the range of 0.01-2. Further, the range of ⁇ L in the whole was 0.9 to 4. Under such conditions, it was possible to confirm a change in transparency like a natural tooth from the first region toward the second region.
- NiO and Z300 are compounds that are also used as pigments, but it was found that they can also be used as light-shielding materials that reduce transparency.
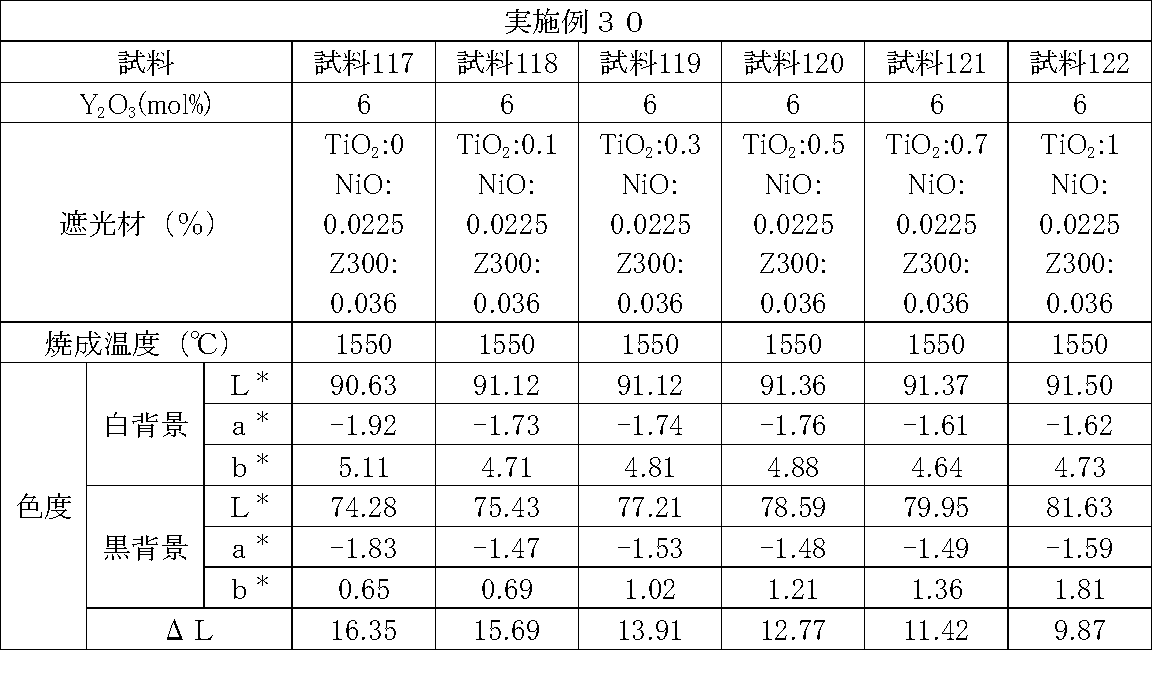
- Example 26 to 31 Samples with different types and contents of the light shielding material were prepared, and the chromaticity of each sample was measured.
- the method for preparing each sample and the method for measuring chromaticity are the same as in Examples 1 to 25.
- zircon (ZrSiO 4 ), titanium oxide (TiO 2 ), alumina (Al 2 O 3 ), and silicon dioxide (SiO 2 ) were used as the light shielding material.
- titanium oxide (TiO 2 ) and a colorant shown in the table were used as a light shielding material.
- Tables 26 to 31 show sample preparation conditions and measurement results.
- ⁇ L tended to decrease by increasing the content rate. That is, the transparency tended to decrease. Thereby, it was confirmed that these compounds can be used as a light shielding material. It was also confirmed that the transparency can be adjusted by adjusting the content of the light shielding material.
- zirconia compositions zirconia calcined bodies and zirconia sintered bodies, and dental products
- identification by structures or characteristics other than the above-mentioned structures or characteristics is required, the identification by these production methods is also taken into consideration. Is possible.
- a partially stabilized zirconia sintered body containing 4 mol% to 7 mol% yttria as a stabilizer The L * value in chromaticity according to the L * a * b * color system measured with a white background is defined as the first L * value.
- the L * value in chromaticity according to the L * a * b * color system measured with a black background is the second L * value, When the value obtained by deducting the second L * value from the first L * value as [Delta] L, The zirconia sintered body, wherein ⁇ L in the first region is larger than ⁇ L in the second region.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Ceramic Engineering (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Veterinary Medicine (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Manufacturing & Machinery (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Structural Engineering (AREA)
- Composite Materials (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Dental Prosthetics (AREA)
- Compositions Of Oxide Ceramics (AREA)
- Inorganic Chemistry (AREA)
- Dental Preparations (AREA)
Abstract
Description
本発明は、日本国特許出願:特願2014-265066号(2014年12月26日出願)に基づき優先権を主張するものであり、同出願の全記載内容は引用をもって本書に組み込み記載されているものとする。
本発明は、ジルコニア焼結体に関する。また、本発明は、当該ジルコニア焼結体を製造するためのジルコニア組成物及びジルコニア仮焼体に関する。さらに、本発明は、ジルコニア焼結体を含む歯科用製品に関する。
ジルコニア焼結体を作製し、各試料について色度を測定した。実施例1~25においては、図2に示す第2実施形態のような4層構成のジルコニア焼結体における各領域に対応するジルコニア焼結体の試料を作製し、各試料について色度を測定した。試料の組成に関するデータ及び測定結果を表1~25に示す。「白背景」とは、塗料に関する測定に使用する白色の隠ぺい率測定用紙を背景(下敷き)にして測定した色度であることを示す。「黒背景」とは、塗料に関する測定に使用する白色の隠ぺい率測定用紙を背景(下敷き)にして測定した色度であることを示す。「ΔL」とは、白背景で測定したL*値と黒背景で測定したL*値の差である。「ΔL層間差」とは、隣接する層間のΔLの差である。n層のΔLと(n+1)層のΔLとの差を(n+1)層の欄に記載してある。「ΔL全体差」とは、第1の領域のΔLと第2の領域のΔLとの差である。表1~25において、同一の試料には同一の試料番号を付してある。
遮光材の種類及び含有率を変化させた試料を作製し、各試料について色度を測定した。各試料の作製方法及び色度の測定方法は実施例1~25と同様である。実施例26~31においては、遮光材としてジルコン(ZrSiO4)、酸化チタン(TiO2)、アルミナ(Al2O3)、及び二酸化ケイ素(SiO2)を用いた。実施例30~31においては、遮光材として酸化チタン(TiO2)及び表に示す着色剤を用いた。
安定化剤として4mol%~7mol%のイットリアを含有する部分安定化ジルコニアの焼結体であって、
白色を背景にして測定したL*a*b*表色系による色度におけるL*値を第1のL*値とし、
黒色を背景にして測定したL*a*b*表色系による色度におけるL*値を第2のL*値とし、
前記第1のL*値から前記第2のL*値を控除した値をΔLとするとき、
前記第1の領域におけるΔLは、前記第2の領域におけるΔLよりも大きい、ジルコニア焼結体。
[付記2]
前記第1の領域におけるΔLは、前記第2の領域におけるΔLよりも0.8以上大きい、付記に記載のジルコニア焼結体。
[付記3]
前記第2の領域におけるΔLは7.5以下である、付記に記載のジルコニア焼結体。
[付記4]
遮光材を含有し、
前記第2の領域における前記遮光材の含有率は、前記第1の領域における前記遮光材の含有率よりも高い、付記に記載のジルコニア焼結体。
[付記5]
前記第1の領域におけるΔLは8~12であり、
前記第2の領域におけるΔLは4~11である、付記に記載のジルコニア焼結体。
[付記6]
前記第1の領域におけるイットリアの含有率と前記第2の領域におけるイットリアの含有率の差は1mol%以下である、付記に記載のジルコニア焼結体。
101 第1の領域
102 第2の領域
103 第3の領域
104 第4の領域
201 歯科用補綴物
202 支台歯
203 歯茎
Claims (31)
- 安定化剤として4mol%~7mol%のイットリアを含有する部分安定化ジルコニアの焼結体であって、
遮光材を含有し、
第1の領域と、
前記第1の領域よりも前記遮光材の含有率が高い第2の領域と、を有し、
前記第1の領域におけるイットリアの含有率と前記第2の領域におけるイットリアの含有率の差は1mol%以下である、ジルコニア焼結体。 - 白色を背景にして測定したL*a*b*表色系による色度におけるL*値を第1のL*値とし、
黒色を背景にして測定したL*a*b*表色系による色度におけるL*値を第2のL*値とし、
前記第1のL*値から前記第2のL*値を控除した値をΔLとするとき、
前記第1の領域におけるΔLは、前記第2の領域におけるΔLよりも大きい、請求項1に記載のジルコニア焼結体。 - 前記第1の領域におけるΔLは、前記第2の領域におけるΔLよりも0.8以上大きい、請求項2に記載のジルコニア焼結体。
- 前記第1の領域におけるΔLは8~12であり、
前記第2の領域におけるΔLは4~11である、請求項2又は3に記載のジルコニア焼結体。 - 前記第2の領域におけるΔLは7.5以下である、請求項2~4のいずれか一項に記載のジルコニア焼結体。
- 前記第1の領域と前記第2の領域とは積層されている、請求項2~5のいずれか一項に記載のジルコニア焼結体。
- 前記第1の領域と前記第2の領域との間に第3の領域をさらに有し、
前記第3の領域のΔLは、前記第2の領域のΔLよりも大きく、前記第1の領域のΔLよりも小さい、請求項2~6のいずれか一項に記載のジルコニア焼結体。 - 前記第3の領域における前記遮光材の含有率は、前記第1の領域における前記遮光材の含有率よりも高く、前記第2の領域における前記遮光材の含有率よりも低い、請求項7に記載のジルコニア焼結体。
- 前記第3の領域と前記第2の領域との間に第4の領域をさらに有し、
前記第4の領域のΔLは、前記第2の領域のΔLよりも大きく、前記第3の領域のΔLよりも小さい、請求項7又は8に記載のジルコニア焼結体。 - 前記第4の領域における前記遮光材の含有率は、前記第3の領域における前記遮光材の含有率よりも高く、前記第2の領域における前記遮光材の含有率よりも低い、請求項9に記載のジルコニア焼結体。
- 各領域の積層方向に厚さにおいて、
前記第1の領域の厚さは全体の30%~40%であり、
前記第2の領域の厚さは全体の30%~40%であり、
前記第3の領域の厚さは全体の10%~20%であり、
前記第4の領域の厚さは全体の10%~20%である、請求項9又は10に記載のジルコニア焼結体。 - 前記遮光材は、酸化ケイ素、酸化アルミニウム、酸化チタン、ジルコニウム及びケイ素の複合酸化物、着色剤、及び蛍光剤を含む群から選択される少なくとも1つである、請求項1~11のいずれか一項に記載のジルコニア焼結体。
- 前記部分安定化ジルコニアは前記安定化剤として5.3mol%~6.2mol%のイットリアを含有する、請求項1~12のいずれか一項に記載のジルコニア焼結体。
- 安定化剤として4mol%~7mol%のイットリアを含有する部分安定化ジルコニアと、
遮光材と、を含有し、
第1の領域と、
前記遮光材の含有率が前記第1の領域よりも高い第2の領域と、を有し、
前記遮光材は、酸化ケイ素、酸化アルミニウム、酸化チタン、ジルコニウム及びケイ素の複合酸化物を含む群から選択される少なくとも1つである、組成物。 - 前記第1の領域と前記第2の領域との間に第3の領域をさらに有し、
前記第3の領域における前記遮光材の含有率は、前記第1の領域における前記遮光材の含有率よりも高く、前記第2の領域における前記遮光材の含有率よりも低い、請求項14に記載の組成物。 - 前記第3の領域と前記第2の領域との間に第4の領域をさらに有し、
前記第4の領域における前記遮光材の含有率は、前記第3の領域における前記遮光材の含有率よりも高く、前記第2の領域における前記遮光材の含有率よりも低い、請求項15に記載の組成物。 - 各領域におけるイットリアの含有率のばらつきは1mol%以下である、請求項14~16のいずれか一項に記載の組成物。
- 第1面、及び前記第1面とは反対側の第2面を有する板状形状を有し、
前記第1の領域は前記第1面側にあり、
前記第2の領域は前記第2面側にある、請求項14~17のいずれか一項に記載の組成物。 - 安定化剤として4mol%~7mol%のイットリアを含有する部分安定化ジルコニアと、
遮光材と、を含有し、
第1の領域と、
前記遮光材の含有率が前記第1の領域よりも高い第2の領域と、を有し、
前記遮光材は、酸化ケイ素、酸化アルミニウム、酸化チタン、ジルコニウム及びケイ素の複合酸化物を含む群から選択される少なくとも1つである、仮焼体。 - 前記第1の領域と前記第2の領域との間に第3の領域をさらに有し、
前記第3の領域における前記遮光材の含有率は、前記第1の領域における前記遮光材の含有率よりも高く、前記第2の領域における前記遮光材の含有率よりも低い、請求項19に記載の仮焼体。 - 前記第3の領域と前記第2の領域との間に第4の領域をさらに有し、
前記第4の領域における前記遮光材の含有率は、前記第3の領域における前記遮光材の含有率よりも高く、前記第2の領域における前記遮光材の含有率よりも低い、請求項20に記載の仮焼体。 - 各領域におけるイットリアの含有率のばらつきは1mol%以下である、請求項19~21のいずれか一項に記載の仮焼体。
- 第1面、及び前記第1面とは反対側の第2面を有する板状形状を有し、
前記第1の領域は前記第1面側にあり、
前記第2の領域は前記第2面側にある、請求項19~22のいずれか一項に記載の仮焼体。 - 請求項14~18のいずれか一項に記載の組成物を800℃~1200℃で焼成して得られる、仮焼体。
- 請求項14~18のいずれか一項に記載の組成物を1400℃以上で焼成して得られる、ジルコニア焼結体。
- 請求項19~23のいずれか一項に記載の仮焼体を1400℃以上で焼成して得られる、ジルコニア焼結体。
- 請求項1~13のいずれか一項に記載のジルコニア焼結体を有する、歯科用製品。
- 歯冠形状を有する、請求項27に記載の歯科用製品。
- 前記第1の領域は歯冠形状の切端側に配され、
前記第2の領域は歯冠形状の歯頚部側に配される、請求項28に記載の歯科用製品。 - 前記第2の領域は、少なくとも支台歯の側面の一部を覆う、請求項29に記載の歯科用製品。
- 歯科用補綴物、歯列矯正用製品、又は歯科インプラント用製品である、請求項27~30のいずれか一項に記載の歯科用製品。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201580071088.9A CN107108373A (zh) | 2014-12-26 | 2015-12-25 | 氧化锆组合物、氧化锆预烧体和氧化锆烧结体、以及齿科用制品 |
| JP2016566525A JP6741592B2 (ja) | 2014-12-26 | 2015-12-25 | ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びに歯科用製品 |
| US15/538,993 US10703076B2 (en) | 2014-12-26 | 2015-12-25 | Zirconia composition, zirconia semi-sintered body and zirconia sintered body, as well as dental product |
| CN202311042598.6A CN117064764A (zh) | 2014-12-26 | 2015-12-25 | 氧化锆组合物、氧化锆预烧体和氧化锆烧结体、以及齿科用制品 |
| EP15873292.5A EP3246300A4 (en) | 2014-12-26 | 2015-12-25 | Zirconia composition, zirconia calcined body, zirconia sintered body, and dental product |
| KR1020177017811A KR102613105B1 (ko) | 2014-12-26 | 2015-12-25 | 지르코니아 조성물, 지르코니아 가소체 및 지르코니아 소결체, 그리고 치과용 제품 |
| US16/892,400 US11077646B2 (en) | 2014-12-26 | 2020-06-04 | Zirconia composition, zirconia semi-sintered body and zirconia sintered body, as well as dental product |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014265066 | 2014-12-26 | ||
| JP2014-265066 | 2014-12-26 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/538,993 A-371-Of-International US10703076B2 (en) | 2014-12-26 | 2015-12-25 | Zirconia composition, zirconia semi-sintered body and zirconia sintered body, as well as dental product |
| US16/892,400 Division US11077646B2 (en) | 2014-12-26 | 2020-06-04 | Zirconia composition, zirconia semi-sintered body and zirconia sintered body, as well as dental product |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016104724A1 true WO2016104724A1 (ja) | 2016-06-30 |
Family
ID=56150736
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/086272 WO2016104724A1 (ja) | 2014-12-26 | 2015-12-25 | ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びに歯科用製品 |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US10703076B2 (ja) |
| EP (1) | EP3246300A4 (ja) |
| JP (2) | JP6741592B2 (ja) |
| KR (1) | KR102613105B1 (ja) |
| CN (2) | CN107108373A (ja) |
| WO (1) | WO2016104724A1 (ja) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017185163A (ja) * | 2016-02-19 | 2017-10-12 | 株式会社松風 | 色調再現性が高い多層構造歯科切削加工用セラミックスブランク |
| WO2018056330A1 (ja) * | 2016-09-20 | 2018-03-29 | クラレノリタケデンタル株式会社 | ジルコニア組成物、仮焼体及び焼結体、並びにそれらの製造方法 |
| WO2019131787A1 (ja) * | 2017-12-26 | 2019-07-04 | クラレノリタケデンタル株式会社 | ミルブランク及び歯科用補綴物、ならびにその製造方法 |
| EP3326574B1 (en) | 2016-11-28 | 2019-10-16 | Shofu Inc. | Dental mill blank with intermediate color layer at 30% position |
| JP2019534080A (ja) * | 2016-10-19 | 2019-11-28 | デンツプライ・シロナ・インコーポレイテッド | 歯科修復物、ブランク、及びブランクの製造の方法 |
| WO2020129918A1 (ja) * | 2018-12-19 | 2020-06-25 | クラレノリタケデンタル株式会社 | ジルコニア焼結体 |
| JP2020109069A (ja) * | 2018-12-31 | 2020-07-16 | Yamakin株式会社 | 歯科用セラミックス材料 |
| JP2021509318A (ja) * | 2017-12-28 | 2021-03-25 | ヴェリコム カンパニー リミテッドVericom Co., Ltd. | 歯科用コンポジットブランク及びその製造方法 |
| EP3892461A1 (en) | 2020-03-06 | 2021-10-13 | Shofu Inc. | Zirconia mill blank having layers with different contents of yttria and alumina |
| JP7022875B1 (ja) * | 2020-09-25 | 2022-02-18 | クラレノリタケデンタル株式会社 | 歯科用被加工体 |
| JP7026295B1 (ja) * | 2020-09-25 | 2022-02-25 | クラレノリタケデンタル株式会社 | ジルコニア焼結体の製造方法 |
| JP2022516876A (ja) * | 2018-12-28 | 2022-03-03 | ヴェリコム カンパニー リミテッド | 複数層を有する歯科用コンポジットブランク及びその製造方法 |
| CN114599765A (zh) * | 2020-01-27 | 2022-06-07 | 第一稀元素化学工业株式会社 | 稳定氧化锆烧结体及氧化锆粉末 |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102540474B1 (ko) * | 2016-02-19 | 2023-06-05 | 소후 인코포레이티드 | 색조 재현성이 높은 다층 구조 치과 절삭 가공용 세라믹스 블랭크 |
| US11613502B2 (en) * | 2016-11-07 | 2023-03-28 | The Regents Of The University Of Colorado | Core-shell ceramic particle colloidal gel and solid oxide fuel cell electrolyte |
| BR112020008314B1 (pt) * | 2017-10-26 | 2023-01-17 | Universidade Do Minho | Implante dentário com gradiente funcional e respetivo processo para sua obtenção. |
| JP7213829B2 (ja) * | 2017-12-26 | 2023-01-27 | クラレノリタケデンタル株式会社 | 歯科用に好適なジルコニア仮焼体 |
| EP3593753A1 (de) * | 2018-07-09 | 2020-01-15 | VITA-ZAHNFABRIK H. Rauter GmbH & Co. KG | Mehrschichtiger formkörper |
| US11161789B2 (en) | 2018-08-22 | 2021-11-02 | James R. Glidewell Dental Ceramics, Inc. | Highly translucent zirconia material, device, methods of making the same, and use thereof |
| WO2020138197A1 (ja) * | 2018-12-26 | 2020-07-02 | クラレノリタケデンタル株式会社 | 歯科用ミルブランク及び歯科用補綴物 |
| EP3936490A4 (en) * | 2019-03-06 | 2022-11-30 | Kuraray Noritake Dental Inc. | ZIRCONIA SINTERED BODY WITH HIGH LINEAR LIGHT TRANSMITTANCE |
| JP2021121576A (ja) * | 2019-03-26 | 2021-08-26 | 東ソー株式会社 | ジルコニア焼結体及びその製造方法 |
| CN110317059B (zh) * | 2019-07-09 | 2021-07-06 | 成都贝施美医疗科技股份有限公司 | 一种各层收缩均匀的分层氧化锆瓷块技术 |
| KR20220083684A (ko) * | 2019-10-17 | 2022-06-20 | 쿠라레 노리타케 덴탈 가부시키가이샤 | 치과용으로 바람직한 지르코니아 성형체 |
| KR20220116439A (ko) * | 2019-12-20 | 2022-08-23 | 쿠라레 노리타케 덴탈 가부시키가이샤 | 지르코니아 소결체의 제조 방법 |
| US20210236389A1 (en) * | 2020-02-03 | 2021-08-05 | James R. Glidewell Dental Ceramics, Inc. | Method of Masking a Dental Support Structure of a Dental Prosthesis Made of Highly Translucent Ceramic Material |
| KR102178904B1 (ko) * | 2020-09-04 | 2020-11-13 | 김정석 | 지르코니아 소재를 이용한 인공치표면에 치아 교정 유지장치의 접착력을 증가시키고 치아를 보호하는 구조체 및 이를 이용한 치아 교정장치 |
| US11672632B2 (en) | 2020-10-05 | 2023-06-13 | Pritidenta Gmbh | Multi-layered zirconia dental blank with reverse layers, process for its preparation and uses thereof |
| WO2022074494A1 (en) | 2020-10-09 | 2022-04-14 | 3M Innovative Properties Company | Set of colored porous zirconia dental mill blanks and process of production |
| KR102286271B1 (ko) * | 2020-10-27 | 2021-08-05 | 주식회사 하스 | 치아 보철수복을 위한 기성 크라운 제조방법 |
| CN115745601B (zh) * | 2022-11-25 | 2024-03-01 | 辽宁爱尔创生物材料有限公司 | 氧化锆烧结体及其制备方法和在牙科材料中的应用 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11100305A (ja) * | 1997-09-29 | 1999-04-13 | Kuraray Co Ltd | 歯科用修復材料 |
| JP2004035332A (ja) * | 2002-07-03 | 2004-02-05 | Advance Co Ltd | セラミックスブロック |
| JP2007126360A (ja) * | 2005-11-01 | 2007-05-24 | Shiyoufuu:Kk | セラミックス歯冠用陶材組成物 |
| JP2007314536A (ja) * | 2006-05-23 | 2007-12-06 | Ivoclar Vivadent Ag | 着色されたブランクおよび歯型部品の調製のためのプロセス |
| WO2014181828A1 (ja) * | 2013-05-10 | 2014-11-13 | クラレノリタケデンタル株式会社 | ジルコニア焼結体、ジルコニア組成物及びジルコニア仮焼体並びにこれらの製造方法、及び歯科用補綴物 |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11305A (ja) | 1997-06-11 | 1999-01-06 | Asahi Optical Co Ltd | 前方視型内視鏡 |
| KR200174757Y1 (ko) * | 1999-09-22 | 2000-03-15 | 태웅마이크로주식회사 | 이동통신단말기용 알에프칩 인덕터 |
| US6878456B2 (en) * | 2001-12-28 | 2005-04-12 | 3M Innovative Properties Co. | Polycrystalline translucent alumina-based ceramic material, uses, and methods |
| JP5396691B2 (ja) | 2007-03-08 | 2014-01-22 | 東ソー株式会社 | 透光性イットリア含有ジルコニア焼結体及びその製造方法並びにその用途 |
| CN100484508C (zh) * | 2006-11-02 | 2009-05-06 | 温宁 | 新型牙科氧化锆修复体及其制备工艺 |
| EP2191809A1 (en) * | 2008-11-27 | 2010-06-02 | 3M Innovative Properties Company | Dental ceramic article, process for production and use thereof |
| KR101324467B1 (ko) | 2012-06-12 | 2013-11-06 | (주)에큐세라 | 다양한 색상과 투광성이 있는 기능성 지르코니아 블록 |
| EP3006418B1 (en) * | 2013-05-10 | 2022-01-05 | Kuraray Noritake Dental Inc. | Process for preparing a zirconia sintered body |
-
2015
- 2015-12-25 KR KR1020177017811A patent/KR102613105B1/ko active IP Right Grant
- 2015-12-25 JP JP2016566525A patent/JP6741592B2/ja active Active
- 2015-12-25 CN CN201580071088.9A patent/CN107108373A/zh active Pending
- 2015-12-25 EP EP15873292.5A patent/EP3246300A4/en active Pending
- 2015-12-25 WO PCT/JP2015/086272 patent/WO2016104724A1/ja active Application Filing
- 2015-12-25 US US15/538,993 patent/US10703076B2/en active Active
- 2015-12-25 CN CN202311042598.6A patent/CN117064764A/zh active Pending
-
2020
- 2020-06-04 US US16/892,400 patent/US11077646B2/en active Active
- 2020-07-27 JP JP2020126265A patent/JP7112455B2/ja active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11100305A (ja) * | 1997-09-29 | 1999-04-13 | Kuraray Co Ltd | 歯科用修復材料 |
| JP2004035332A (ja) * | 2002-07-03 | 2004-02-05 | Advance Co Ltd | セラミックスブロック |
| JP2007126360A (ja) * | 2005-11-01 | 2007-05-24 | Shiyoufuu:Kk | セラミックス歯冠用陶材組成物 |
| JP2007314536A (ja) * | 2006-05-23 | 2007-12-06 | Ivoclar Vivadent Ag | 着色されたブランクおよび歯型部品の調製のためのプロセス |
| WO2014181828A1 (ja) * | 2013-05-10 | 2014-11-13 | クラレノリタケデンタル株式会社 | ジルコニア焼結体、ジルコニア組成物及びジルコニア仮焼体並びにこれらの製造方法、及び歯科用補綴物 |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017185163A (ja) * | 2016-02-19 | 2017-10-12 | 株式会社松風 | 色調再現性が高い多層構造歯科切削加工用セラミックスブランク |
| US11873254B2 (en) | 2016-09-20 | 2024-01-16 | Kuraray Noritake Dental Inc. | Zirconia composition, pre-sintered body and sintered body, and method of producing the same |
| WO2018056330A1 (ja) * | 2016-09-20 | 2018-03-29 | クラレノリタケデンタル株式会社 | ジルコニア組成物、仮焼体及び焼結体、並びにそれらの製造方法 |
| CN109689593A (zh) * | 2016-09-20 | 2019-04-26 | 可乐丽则武齿科株式会社 | 氧化锆组合物、预煅烧体和烧结体、以及其制造方法 |
| KR20190047701A (ko) * | 2016-09-20 | 2019-05-08 | 쿠라레 노리타케 덴탈 가부시키가이샤 | 지르코니아 조성물, 가소체 및 소결체, 그리고 그들의 제조 방법 |
| KR102462031B1 (ko) | 2016-09-20 | 2022-11-02 | 쿠라레 노리타케 덴탈 가부시키가이샤 | 지르코니아 조성물, 가소체 및 소결체, 그리고 그들의 제조 방법 |
| JPWO2018056330A1 (ja) * | 2016-09-20 | 2019-08-15 | クラレノリタケデンタル株式会社 | ジルコニア組成物、仮焼体及び焼結体、並びにそれらの製造方法 |
| US11535564B2 (en) | 2016-09-20 | 2022-12-27 | Kuraray Noritake Dental Inc. | Zirconia composition, pre-sintered body and sintered body, and method of producing the same |
| JP7198667B2 (ja) | 2016-09-20 | 2023-01-04 | クラレノリタケデンタル株式会社 | ジルコニア組成物、仮焼体及び焼結体、並びにそれらの製造方法 |
| US11884595B2 (en) | 2016-09-20 | 2024-01-30 | Kuraray Noritake Dental Inc. | Zirconia composition, pre-sintered body and sintered body, and method of producing the same |
| JP2019534080A (ja) * | 2016-10-19 | 2019-11-28 | デンツプライ・シロナ・インコーポレイテッド | 歯科修復物、ブランク、及びブランクの製造の方法 |
| JP7413017B2 (ja) | 2016-10-19 | 2024-01-15 | デンツプライ・シロナ・インコーポレイテッド | 歯科修復物、ブランク、及びブランクの製造の方法 |
| EP3326574B1 (en) | 2016-11-28 | 2019-10-16 | Shofu Inc. | Dental mill blank with intermediate color layer at 30% position |
| JPWO2019131787A1 (ja) * | 2017-12-26 | 2020-12-24 | クラレノリタケデンタル株式会社 | ミルブランク及び歯科用補綴物、ならびにその製造方法 |
| JP7278963B2 (ja) | 2017-12-26 | 2023-05-22 | クラレノリタケデンタル株式会社 | ミルブランク及び歯科用補綴物、ならびにその製造方法 |
| CN111511309A (zh) * | 2017-12-26 | 2020-08-07 | 可乐丽则武齿科株式会社 | 研磨坯料和牙科用修补物、以及其制备方法 |
| US11413123B2 (en) | 2017-12-26 | 2022-08-16 | Kuraray Noritake Dental Inc. | Mill blank and dental prosthesis, and methods respectively for producing these products |
| WO2019131787A1 (ja) * | 2017-12-26 | 2019-07-04 | クラレノリタケデンタル株式会社 | ミルブランク及び歯科用補綴物、ならびにその製造方法 |
| JP2021509318A (ja) * | 2017-12-28 | 2021-03-25 | ヴェリコム カンパニー リミテッドVericom Co., Ltd. | 歯科用コンポジットブランク及びその製造方法 |
| JP7186462B2 (ja) | 2017-12-28 | 2022-12-09 | ヴェリコム カンパニー リミテッド | 歯科用コンポジットブランク及びその製造方法 |
| CN113164239A (zh) * | 2018-12-19 | 2021-07-23 | 可乐丽则武齿科株式会社 | 氧化锆烧结体 |
| JPWO2020129918A1 (ja) * | 2018-12-19 | 2021-10-21 | クラレノリタケデンタル株式会社 | ジルコニア焼結体 |
| JP7237989B2 (ja) | 2018-12-19 | 2023-03-13 | クラレノリタケデンタル株式会社 | ジルコニア焼結体 |
| WO2020129918A1 (ja) * | 2018-12-19 | 2020-06-25 | クラレノリタケデンタル株式会社 | ジルコニア焼結体 |
| JP2022516876A (ja) * | 2018-12-28 | 2022-03-03 | ヴェリコム カンパニー リミテッド | 複数層を有する歯科用コンポジットブランク及びその製造方法 |
| US11744684B2 (en) | 2018-12-28 | 2023-09-05 | Vericom Co., Ltd. | Dental composite blank having multiple layers and method for manufacturing same |
| JP7386555B2 (ja) | 2018-12-28 | 2023-11-27 | ヴェリコム カンパニー リミテッド | 複数層を有する歯科用コンポジットブランク及びその製造方法 |
| JP2020109069A (ja) * | 2018-12-31 | 2020-07-16 | Yamakin株式会社 | 歯科用セラミックス材料 |
| CN114599765A (zh) * | 2020-01-27 | 2022-06-07 | 第一稀元素化学工业株式会社 | 稳定氧化锆烧结体及氧化锆粉末 |
| CN114599765B (zh) * | 2020-01-27 | 2024-03-08 | 第一稀元素化学工业株式会社 | 稳定氧化锆烧结体及氧化锆粉末 |
| EP3892461A1 (en) | 2020-03-06 | 2021-10-13 | Shofu Inc. | Zirconia mill blank having layers with different contents of yttria and alumina |
| WO2022065452A1 (ja) * | 2020-09-25 | 2022-03-31 | クラレノリタケデンタル株式会社 | ジルコニア焼結体の製造方法 |
| WO2022065448A1 (ja) * | 2020-09-25 | 2022-03-31 | クラレノリタケデンタル株式会社 | 歯科用被加工体 |
| JP7026295B1 (ja) * | 2020-09-25 | 2022-02-25 | クラレノリタケデンタル株式会社 | ジルコニア焼結体の製造方法 |
| CN116209409A (zh) * | 2020-09-25 | 2023-06-02 | 可乐丽则武齿科株式会社 | 氧化锆烧结体的制造方法 |
| JP7022875B1 (ja) * | 2020-09-25 | 2022-02-18 | クラレノリタケデンタル株式会社 | 歯科用被加工体 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20200298536A1 (en) | 2020-09-24 |
| JP7112455B2 (ja) | 2022-08-03 |
| US11077646B2 (en) | 2021-08-03 |
| US10703076B2 (en) | 2020-07-07 |
| JP6741592B2 (ja) | 2020-08-19 |
| EP3246300A1 (en) | 2017-11-22 |
| CN107108373A (zh) | 2017-08-29 |
| US20180002235A1 (en) | 2018-01-04 |
| KR102613105B1 (ko) | 2023-12-12 |
| CN117064764A (zh) | 2023-11-17 |
| JPWO2016104724A1 (ja) | 2017-11-02 |
| KR20170101920A (ko) | 2017-09-06 |
| EP3246300A4 (en) | 2018-07-04 |
| JP2020180048A (ja) | 2020-11-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6741592B2 (ja) | ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びに歯科用製品 | |
| JP7233395B2 (ja) | ジルコニア焼結体及びジルコニア仮焼体 | |
| JP7277495B2 (ja) | 歯科用ジルコニア焼結体、積層体及び歯科加工用ジルコニア仮焼体、並びに歯科用補綴物 | |
| KR102428052B1 (ko) | 소결 거동이 조정된 다층 산화물 세라믹체 | |
| JP6352593B2 (ja) | ジルコニア焼結体、ジルコニア組成物及びジルコニア仮焼体、並びに歯科用補綴物 | |
| JP7213829B2 (ja) | 歯科用に好適なジルコニア仮焼体 | |
| JP7213268B2 (ja) | 歯科用に好適なジルコニア仮焼体 | |
| JP7001310B1 (ja) | 歯科用に好適なジルコニア仮焼体 | |
| WO2016068288A1 (ja) | ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びに歯科用製品 | |
| JP6584766B2 (ja) | ジルコニア焼結体及び歯科用製品 | |
| JP6761250B2 (ja) | ジルコニア焼結体及び歯科用製品、並びにそれらの製造方法 | |
| JP6920573B1 (ja) | ジルコニア組成物、ジルコニア仮焼体及びジルコニア焼結体、並びにそれらの製造方法 | |
| JP6818112B2 (ja) | ジルコニア仮焼体及びジルコニア焼結体並びに積層体 | |
| WO2023127945A1 (ja) | 歯科用に好適なジルコニア仮焼体 | |
| JP6975216B2 (ja) | ジルコニア焼結体 | |
| JP7139407B2 (ja) | ジルコニア仮焼体及びジルコニア焼結体並びに積層体 | |
| JP7061514B2 (ja) | ジルコニア焼結体 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15873292 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016566525 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15538993 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20177017811 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015873292 Country of ref document: EP |