WO2013180067A1 - 塗料組成物及びこの塗料組成物を塗布して成る塗装金属板、金属容器及び金属蓋 - Google Patents
塗料組成物及びこの塗料組成物を塗布して成る塗装金属板、金属容器及び金属蓋 Download PDFInfo
- Publication number
- WO2013180067A1 WO2013180067A1 PCT/JP2013/064659 JP2013064659W WO2013180067A1 WO 2013180067 A1 WO2013180067 A1 WO 2013180067A1 JP 2013064659 W JP2013064659 W JP 2013064659W WO 2013180067 A1 WO2013180067 A1 WO 2013180067A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyester resin
- coating composition
- resin
- coating
- resistance
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D167/00—Coating compositions based on polyesters obtained by reactions forming a carboxylic ester link in the main chain; Coating compositions based on derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D167/00—Coating compositions based on polyesters obtained by reactions forming a carboxylic ester link in the main chain; Coating compositions based on derivatives of such polymers
- C09D167/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D7/00—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials
- B05D7/14—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials to metal, e.g. car bodies
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B05—SPRAYING OR ATOMISING IN GENERAL; APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D—PROCESSES FOR APPLYING FLUENT MATERIALS TO SURFACES, IN GENERAL
- B05D7/00—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials
- B05D7/24—Processes, other than flocking, specially adapted for applying liquids or other fluent materials to particular surfaces or for applying particular liquids or other fluent materials for applying particular liquids or other fluent materials
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/04—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B15/08—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
- B32B15/09—Layered products comprising a layer of metal comprising metal as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin comprising polyesters
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65D—CONTAINERS FOR STORAGE OR TRANSPORT OF ARTICLES OR MATERIALS, e.g. BAGS, BARRELS, BOTTLES, BOXES, CANS, CARTONS, CRATES, DRUMS, JARS, TANKS, HOPPERS, FORWARDING CONTAINERS; ACCESSORIES, CLOSURES, OR FITTINGS THEREFOR; PACKAGING ELEMENTS; PACKAGES
- B65D25/00—Details of other kinds or types of rigid or semi-rigid containers
- B65D25/14—Linings or internal coatings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D161/00—Coating compositions based on condensation polymers of aldehydes or ketones; Coating compositions based on derivatives of such polymers
- C09D161/20—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/08—Anti-corrosive paints
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/65—Additives macromolecular
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
- C08L2205/025—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group containing two or more polymers of the same hierarchy C08L, and differing only in parameters such as density, comonomer content, molecular weight, structure
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/03—Polymer mixtures characterised by other features containing three or more polymers in a blend
- C08L2205/035—Polymer mixtures characterised by other features containing three or more polymers in a blend containing four or more polymers in a blend
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L61/00—Compositions of condensation polymers of aldehydes or ketones; Compositions of derivatives of such polymers
- C08L61/04—Condensation polymers of aldehydes or ketones with phenols only
- C08L61/06—Condensation polymers of aldehydes or ketones with phenols only of aldehydes with phenols
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L61/00—Compositions of condensation polymers of aldehydes or ketones; Compositions of derivatives of such polymers
- C08L61/20—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C08L61/26—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with heterocyclic compounds
- C08L61/28—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with heterocyclic compounds with melamine
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/13—Hollow or container type article [e.g., tube, vase, etc.]
- Y10T428/1352—Polymer or resin containing [i.e., natural or synthetic]
- Y10T428/1355—Elemental metal containing [e.g., substrate, foil, film, coating, etc.]
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31678—Of metal
- Y10T428/31681—Next to polyester, polyamide or polyimide [e.g., alkyd, glue, or nylon, etc.]
Definitions
- the present invention relates to a polyester-based coating composition, and more specifically, a polyester-based coating composition having excellent resistance to embrittlement with time, workability, corrosion resistance, abrasion resistance, retort resistance, and blocking resistance, and the coating composition.
- the present invention relates to a coated metal plate, a metal lid, and a metal container formed by painting an object.
- the organic coating that protects the metal container or metal lid prevents the metal substrate from being corroded by the contents, etc., and the metal container is subjected to mechanical processing such as neck-in processing, bead processing, and lid tightening processing.
- a metal lid such as an easy open end (hereinafter sometimes referred to as “EOE”), it is subjected to severe processing such as score processing and rivet processing, so it is used for a metal container or metal lid
- EEE easy open end
- severe processing such as score processing and rivet processing
- Patent Document 1 proposes a coating composition in which a predetermined amount of a hydroxyl group-containing polyester resin, a specific phenol resin crosslinking agent, and an acid catalyst is blended. It is described that it is possible to form a coating film that is excellent in the balance of adhesiveness, adhesiveness, curability, hygiene and boiling water resistance.
- Patent Documents 2 and 3 listed below describe resin compositions for paints using a polyester resin containing a molecule in which a pendant carboxyl group is introduced into the molecular chain.
- the water-based resin composition described in Patent Document 2 In the product, it is excellent in curability, workability, retort resistance, hygiene, spray paintability, and water dispersibility, and in the coating resin composition described in Patent Document 3, the adhesion to the metal surface is improved. It is described that it is particularly excellent and can satisfy high workability and corrosion resistance.
- Patent Document 4 includes a polyester resin having a low Tg and a crosslinking agent having 2-methyl-1,3-propanediol, 1,4-butanediol and 1,4-cyclohexanedimethanol as polyalcohol components as essential components.
- a polyester resin composition is proposed that has processability, retort resistance, flavor properties, content resistance properties, feathering properties, and sulfur resistance, as well as high processability required as an internal paint for EOE, It is described that it has feathering properties and can suppress deterioration of workability over time.
- Patent Document 5 proposes a coating composition containing an appropriately vertically branched bivalent alcohol component while appropriately branching the polymer polyester resin, and the workability of the coating film does not decrease with time. It describes that it is excellent in the balance between adhesion to the material, hardness, and workability. Patent Document 4 describes that a polyester resin having a glass transition temperature (Tg) of 20 to 50 ° C. is preferred, but Patent Document 5 does not mention the glass transition temperature.
- Tg glass transition temperature
- an object of the present invention is to further improve the resistance to embrittlement with time, and to obtain a coating film excellent in all of the coating film performance such as workability, corrosion resistance, abrasion resistance, blocking resistance, and retort resistance. It is to provide a composition.
- a coating composition comprising a mixed polyester resin mixed with a polyester resin (B) at ⁇ 20 to 25 ° C. containing a crosslinking agent and a curing catalyst.
- the polyester resin (A) is a polyester resin which has been given the acid value of the resin by a ring-opening addition reaction of a compound having one or more carboxylic anhydride structures in the molecule; 2.
- Tgmix is the glass transition temperature (K) of the mixed polyester resin
- Tga is the glass transition temperature (K) of the polyester resin (A)
- Tgb is the glass transition temperature (K) of the polyester resin (B)
- Wa is the polyester resin.
- the weight fraction of (A) and Wb are the weight fraction of the polyester resin (B), respectively. 4).
- the crosslinking agent comprises a resol type phenol resin and / or an amino resin, and is blended in an amount of 1 to 30 parts by weight with respect to 100 parts by weight of the resin solids of the polyester resins (A) and (B); 5.
- the curing catalyst is blended in an amount of 0.1 to 3.0 parts by weight with respect to 100 parts by weight of the total resin solids; Is preferred.
- coated the said coating composition is also provided.
- the coating composition is preferably formed by coil baking.
- the present invention further provides a metal container or a metal lid on which a coating film made of a coating composition is formed.
- the reason for the occurrence of the embrittlement phenomenon with time which is one of the problems to be solved by the coating composition of the present invention, has not yet been elucidated, but the present inventors presume as follows: Yes. That is, it is considered that the coating film immediately after curing undergoes molecular reorientation (transition to an equilibrium state) due to enthalpy relaxation over time, resulting in embrittlement of the coating film and decrease in workability.
- the crosslink density of the cured coating film is increased and molecular motion is suppressed to suppress enthalpy relaxation, or the molecular motion is continuously maintained even at the storage temperature with time (non-equilibrium state).
- the cured coating film does not have sufficient workability from the beginning, and in the latter method, even though the embrittlement phenomenon with time of the cured coating film can be suppressed, the corrosion resistance of the contents, during the molding process It was found that the abrasion resistance of the coated metal plate, the blocking resistance of the coated metal plate, and the retort resistance during the sterilization treatment were not sufficient, and in any of the methods, sufficient coating performance in practical use could not be obtained.
- the polyester resin (A) capable of suppressing molecular motion to prevent enthalpy relaxation, which is a cause of embrittlement with time
- a cured coating film that increases the crosslinking density By using a combination of a polyester resin (B) having a low Tg, which can maintain molecular motion even at a storage temperature of a long time, embrittlement resistance over time, workability, corrosion resistance, abrasion resistance, blocking resistance, It came to invention of the coating composition which can form the coating film which was excellent in all coating-film performances, such as retort resistance.
- the coated metal plate formed by applying the coating composition of the present invention has the above-described excellent coating film performance, and can be suitably used for a metal container or a metal lid. Furthermore, the metal container and the metal lid of the present invention have excellent corrosion resistance and retort resistance.
- the coated metal plate and the metal lid using the coating composition of the present invention are excellent in workability both in the initial stage and after the lapse of time, and all of corrosion resistance, retort resistance, blocking resistance, and abrasion resistance.
- the metal lid is evaluated only for the metal lid, which is formed by processing a painted metal plate, while the painted metal container is generally formed after the container is molded. Since the coating film on the coated metal lid has a higher degree of processing, if a good evaluation can be obtained with a can lid, naturally a good evaluation can also be obtained with a metal container. Evaluation is conducted only in
- polyester resin (A) used in the coating composition of the present invention has an acid value in the range of 2 to 50 mgKOH / g, particularly 5 to 30 mgKOH / g, and a glass transition temperature (Tg) of 35 to 100 ° C., particularly 50 to As long as it is in the range of 90 ° C., polyester resins composed of various carboxylic acid components and alcohol components can be used. In the present invention, it is important that the acid value of the polyester resin (A) is in the above range, thereby improving the adhesion to the metal substrate and contributing to the improvement of workability.
- the adhesion to the substrate when the adhesion to the substrate is increased, it becomes difficult for corrosion-inducing substances (salts and acids) to reach the metal, and excellent corrosion resistance can be exhibited. Moreover, it contributes to increasing the crosslink density and suppressing the embrittlement of the coating film due to the enthalpy relaxation over time of the cured coating film. If the acid value is smaller than the above range, sufficient adhesion to the substrate may not be obtained, and it may be difficult to achieve both workability and corrosion resistance. The effect of suppressing embrittlement may not be sufficiently obtained. When the acid value is larger than the above range, the water resistance of the coating film may decrease. Moreover, when the glass transition temperature (Tg) of a polyester resin (A) is smaller than the said range, blocking resistance may be inferior. Moreover, when Tg is higher than the said range, the solubility to the solvent of a coating composition may be inferior.
- Tg glass transition temperature
- carboxylic acid component used in the polyester resin (A) examples include aromatic dicarboxylic acids such as terephthalic acid, isophthalic acid, orthophthalic acid, and naphthalenedicarboxylic acid, succinic acid, glutaric acid, adipic acid, azelaic acid, sebacic acid, Aliphatic dicarboxylic acids such as dodecanedioic acid and dimer acid, unsaturated dicarboxylic acids such as (anhydrous) maleic acid, fumaric acid, dodecenyl succinic anhydride, terpene-maleic acid adduct, 1,4-cyclohexanedicarboxylic acid, tetrahydrophthal Acid, hexahydroisophthalic acid, alicyclic dicarboxylic acid such as 1,2-cyclohexene dicarboxylic acid, trihydric or higher carboxylic acid such as (anhydrous) trimellitic acid, (anhydrous) pyrom
- Examples of the alcohol component used in the polyester resin (A) include ethylene glycol, propylene glycol (1,2-propanediol), 1,3-propanediol, 1,4-butanediol, 1,2-butanediol, , 3-butanediol, 2-methyl-1,3-propanediol, neopentyl glycol, 1,5-pentanediol, 1,6-hexanediol, 3-methyl-1,5-pentanediol, 2-ethyl- 2-butyl-1,3-propanediol, 2,4-diethyl-1,5-pentanediol, 1-methyl-1,8-octanediol, 3-methyl-1,6-hexanediol, 4-methyl- 1,7-heptanediol, 4-methyl-1,8-octanediol, 4-propyl-1,8-oc
- a carboxyl group is present in the middle of a molecular chain from the viewpoint of increasing the crosslinking density of the coating film and suppressing molecular motion to prevent enthalpy relaxation, which is a cause of embrittlement over time. It is particularly preferred to include molecules introduced in pendant form.
- a method for obtaining such a polyester is not limited to this. For example, a ring-opening addition reaction of a compound having one or more carboxylic acid anhydride groups in the molecule described in JP-A No. 2003-213201 is performed. Can be mentioned.
- the polyester resin (A) given the resin acid value by the ring-opening addition reaction of the compound having one or more carboxylic acid anhydride structures in the molecule is the total carboxylic acid used in the ring-opening addition reaction.
- the anhydride is 100 mol%
- 10 mol% or more of the carboxylic acid anhydride used is preferably a compound having two or more carboxylic anhydride groups in the molecule.
- a compound having two or more carboxylic anhydride groups in the molecule can modify not only the polyester resin terminal but also the carboxyl group in the resin molecule chain. By modifying the carboxyl group in the resin molecular chain, it becomes possible to impart an acid value without any limitation on the molecular weight.
- the adhesion between the base materials is further improved because of the low molecular weight between carboxyl groups, and a coating film with further excellent corrosion resistance can be obtained.
- the ring-opening addition reaction of a compound having one or more carboxylic acid anhydride structures in the molecule can be carried out by a known synthesis method.
- a method of adding (2) a method in which a carboxylic acid anhydride is added when the polyester resin is less than the target molecular weight (Mn ⁇ 3,000), and the molecular weight is increased to the target in a nitrogen atmosphere, and (3) an acid is added.
- Examples thereof include a method of kneading a polyester resin and a carboxylic acid anhydride that are not mixed with a melt extruder, and acid addition after melting in a batch.
- Examples of the compound having two or more carboxylic anhydride groups in the molecule include pyromellitic anhydride, 1,2,3,4-butanetetracarboxylic dianhydride, 1,2,3,4-pentane. Tetracarboxylic dianhydride, 3,4,3 ′, 4′-benzophenonetetracarboxylic dianhydride, cyclopentanetetracarboxylic dianhydride, 2,3,6,7-naphthalenetetracarboxylic dianhydride, 1,2,5,6-naphthalenetetracarboxylic dianhydride, ethylene glycol bistrimellitic dianhydride, 2,3,2 ′, 3′-diphenyltetracarboxylic dianhydride, thiophene-2,3,4 , 5-tetracarboxylic dianhydride, ethylenetetracarboxylic dianhydride, 4,4'-oxydiphthalic dianhydride, 5- (2,5-dioxo
- Examples of the carboxylic acid monoanhydride compound that can be used in combination with a compound having two or more carboxylic anhydride groups in the molecule include phthalic anhydride, maleic anhydride, succinic anhydride, maleic anhydride, trimellitic anhydride, anhydrous Examples thereof include monoanhydrides such as itaconic acid and citraconic anhydride, hexahydrophthalic anhydride, tetrahydrophthalic anhydride, and the like, and one or more of them can be selected and used. Of these, phthalic anhydride and trimellitic anhydride are preferred in view of versatility.
- the number average molecular weight of the polyester resin (A) used in the present invention is 3,000 to 100,000, preferably 8,000 to 50,000, more preferably 10,000 to 30,000. It is desirable. If the number average molecular weight is less than 3,000, the coating film becomes brittle and the processability may be inferior. If it exceeds 100,000, the coating workability may deteriorate.
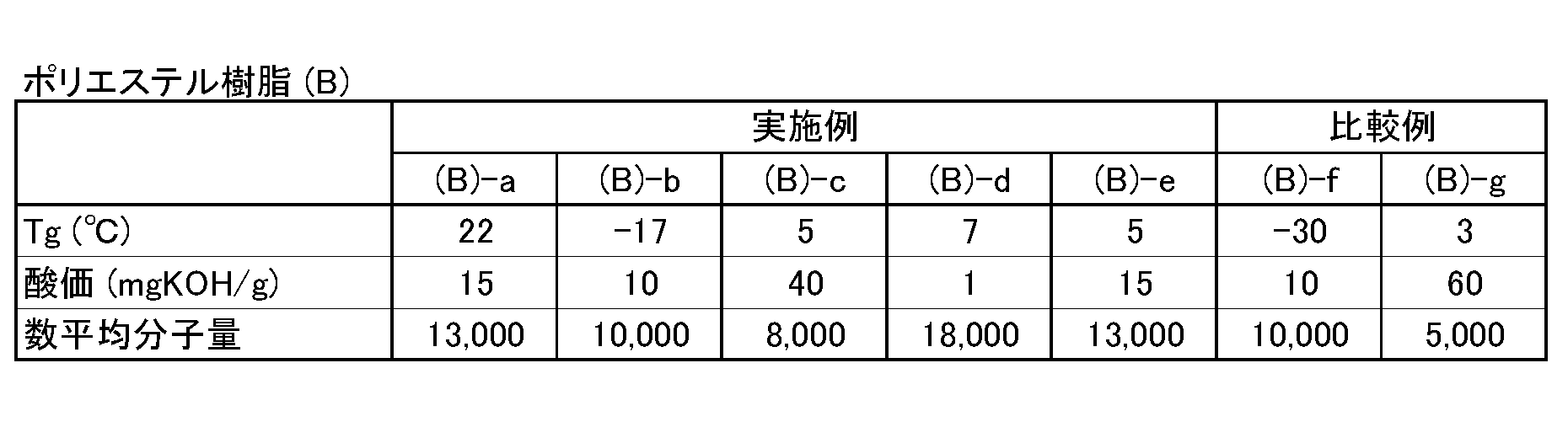
- polyester resin (B) used in the coating composition of the present invention has an acid value in the range of 0 to 50 mgKOH / g, particularly 0 to 20 mgKOH / g, and a glass transition temperature (Tg) of ⁇ 20 to 25 ° C., particularly 0. As long as it is in the range of ⁇ 15 ° C., polyester resins comprising various carboxylic acid components and alcohol components can be used. It is important that the acid value is in the above range, and if it is larger than the above range, the water resistance of the coating film may be lowered. It is important that the glass transition temperature (Tg) is in the above range.
- the glass transition temperature (Tg) is larger than the above range, sufficient embrittlement resistance over time cannot be obtained. Moreover, when smaller than the said range, corrosion resistance, blocking resistance, and retort resistance may become inadequate.
- the carboxylic acid component and the alcohol component the carboxylic acid component and the alcohol component exemplified for the polyester resin (A) can be used.
- the polyester resin (B) preferably contains 2-methyl-1,3-propanediol, 1,4-butanediol and 1,4-cyclohexanedimethanol among the above alcohol components.
- the number average molecular weight of the polyester resin (B) used in the present invention is in the range of 3,000 to 100,000, preferably 8,000 to 50,000, more preferably, like the polyester resin (A). Is preferably in the range of 10,000 to 30,000. If the number average molecular weight of the polyester resin is less than 3,000, the coating film becomes brittle and the workability may be inferior. If it exceeds 100,000, the coating workability may be deteriorated.
- the polyester resin (B) can be appropriately selected from commercially available products.
- Byron 300 Toyobo, molecular weight (Mn) 23,000, acid value 2 or less, Tg 7 ° C.
- Byron 516 Toyobo, molecular weight (Mn) 30,000, acid value 2 or less, Tg ⁇ 17 ° C.
- Byron 560 Toyobo, molecular weight (Mn) 19,000, acid value 2 or less, Tg 7 ° C.
- Byron 630 Toyobo, molecular weight (Mn) 23,000, acid value 2 or less, Tg 7 ° C.
- Byron GK180 Toyobo, molecular weight (Mn) 10,000, acid value 5, Tg 0 ° C.
- Byron GK330 Toyobo, molecular weight (Mn) 17,000, acid value 2 or less, Tg 16 ° C.
- Elitel UE-3223 Unitika, molecular weight (Mn) 2
- a resol type phenol resin and / or an amino resin as a crosslinking agent.
- the resol type phenol resin include, as phenol monomers, o-cresol, p-cresol, p-tert-butylphenol, p-ethylphenol, 2,3-xylenol, 2,5-xylenol, phenol, m-cresol, m -Ethylphenol, 3,5-xylenol, m-methoxyphenol, etc., which can be used singly or in combination, and these phenol monomers and formaldehyde were reacted in the presence of an alkali catalyst Is.
- a metacresol resin is preferable.
- commercially available products such as Sumitrite Resin PR-53893A (solid content 50%) manufactured by Sumitomo Durez Co., Ltd., CKS-3898 (solid content 50%) manufactured by Showa Denko KK can be used.
- amino resin for example, urea resin, melamine resin, benzoguanamine resin, and the like can be used. More specifically, methylated urea resin, methyl etherified urea resin, butyl etherified urea resin, and a mixture of methyl ether and butyl ether Etherified urea resins; methylolated melamine resins, methyl etherified melamine resins, butyl etherified melamine resins, mixed etherified melamine resins of methyl ether and butyl ether, etc .; methylolated benzoguanamine resins, methyl etherified benzoguanamine resins, butyl etherified benzoguanamines Resin, mixed etherified benzoguanamine resin of methyl ether and butyl ether, and the like.
- the crosslinking reaction is promoted, and more precise crosslinking can be efficiently performed at a low temperature and in a short time.
- phosphoric acid compounds such as organic sulfonic acid compounds such as p-toluenesulfonic acid, dodecylbenzenesulfonic acid and camphorsulfonic acid, and neutralized amines thereof can be used.
- NACURE4054J phosphoric acid, manufactured by KING INDUSTRIES
- NACURE5076 diodecylbenzenesulfonic acid, manufactured by KING INDUSTRIES
- Cycat 4040 aromatic sulfonic acid, manufactured by Nippon Cytec Industries
- Cycat 4050 amine Block aromatic sulfonic acid, manufactured by Nippon Cytec Industries
- Cycat 600 aromatic sulfonic acid, manufactured by Nippon Cytec Industries
- Cycat 602 amine block aromatic sulfonic acid, manufactured by Nippon Cytec Industries
- the coating composition of this invention contains a crosslinking agent and a curing catalyst in the mixed polyester resin obtained by mixing the polyester resin (A) and the polyester resin (B).
- the ratio of the polyester resin (B) having a low Tg is 1 or less, the resistance to embrittlement with time deteriorates remarkably, and the post-aging processability of the coated plate cannot maintain the initial processability. This may cause a problem in practical use.
- the glass transition temperature calculated by the above formula (1) of the mixed polyester resin obtained by mixing the polyester resin (A) and the polyester resin (B) is 35 ° C. or more, particularly 40 to 80. It is desirable to be in the range of ° C. When the Tg is smaller than the above range, the blocking resistance is inferior. Moreover, when using as a can inner surface coating material, since a flavor property is required, it is preferable that it is Tg of 40 degreeC or more. From the viewpoint of solubility of the coating composition in a solvent, the upper limit is preferably 80 ° C. or less.
- the crosslinking agent is preferably in the range of 1 to 30 parts by weight, particularly 5 to 20 parts by weight, based on 100 parts by weight of the mixed polyester resin in which the polyester resin (A) and the polyester resin (B) are mixed. is there.
- the addition amount of the crosslinking agent is less than 1 part by weight, the curability becomes insufficient, and the resistance to embrittlement with time, workability, content resistance, retort resistance, and blocking resistance may be inferior.
- the curing catalyst is 0.1 to 3.0 parts by weight, particularly 0.1 to 2. It is suitable to be in the range of 0 part by weight.
- the blending amount of the curing catalyst When the blending amount of the curing catalyst is less than the above range, the effect of promoting the curing reaction obtained by blending the curing catalyst is not sufficiently obtained, while when the blending amount of the curing catalyst is larger than the above range. In some cases, the curing acceleration effect is large and the workability is insufficient.
- Conventionally known additives such as a lubricant, an antifoaming agent, a leveling agent, and a pigment can be added to the coating composition of the present invention in a known formulation.
- the coating composition of this invention can be used in the state which melt
- Organic solvents to be used include aromatic hydrocarbons such as toluene, xylene, and solvesso, esters such as ethyl acetate, butyl acetate, and dibasic acid esters, ketones such as methyl ethyl ketone, methyl isobutyl ketone, cyclohexanone, and isophorone, and butyl cellosolve.
- Alcohol ethers such as butyl carbitol, alcohol ketones such as diacetone alcohol, alcohols such as isopropyl alcohol, n-butanol, amyl alcohol, n-hexanol, etc.
- Alcohol ethers such as butyl carbitol, alcohol ketones such as diacetone alcohol, alcohols such as isopropyl alcohol, n-butanol, amyl alcohol, n-hexanol, etc.
- it can also be made water-based by a conventionally known method and used as an aqueous coating composition.
- a basic compound such as ammonia or an organic amine compound, water, or further an organic solvent is added to form an aqueous coating composition.
- other resins are further used for the purpose of modifying the flexibility and adhesion of the coating film within the range that does not impair the excellent characteristics of the coating composition of the present invention. be able to.
- other resins include ethylene-polymerizable unsaturated carboxylic acid copolymers, ethylene-polymerizable carboxylic acid copolymer ionomers, non-aqueous polyester resins, and the like, and at least one resin selected from these resins. In some cases, the flexibility and adhesion of the coating film can be effectively imparted.
- the coated metal plate of the present invention can be obtained by coating the metal plate with the coating composition of the present invention.
- the metal plate that can be used include hot-drawn steel plate, cold-rolled steel plate, hot-dip galvanized steel plate, electrogalvanized steel plate, alloy-plated steel plate, aluminum zinc alloy-plated steel plate, aluminum plate, tin-plated steel plate, stainless steel plate, copper plate, copper Examples include plated steel sheets, tin-free steel, nickel-plated steel sheets, ultra-thin tin-plated steel sheets, chrome-treated steel sheets, and the like, and various surface treatments and primer treatments are used as necessary.
- the coated metal plate of the present invention can be obtained by coating the coating composition of the present invention on a metal plate by a known coating method such as roll coater coating or spray coating, and baking it by a heating means such as a coil oven.
- the coating composition of the present invention effectively suppresses the embrittlement of the cured coating film over time, so that it is baked at a high temperature in a short time, and is particularly effective when forming a coating film by coil baking, which tends to cause embrittlement over time.
- the thickness of the coating film is not particularly limited, but the dry film thickness is preferably in the range of 3 to 18 ⁇ m, more preferably 3 to 12 ⁇ m.
- the dry film thickness is appropriately determined depending on the use of the coated metal plate.
- the baking conditions of the coating film are appropriately adjusted depending on the polyester resin used, the type of crosslinking agent, the type of solvent used and the type of metal material to be coated, the thickness, the coating speed, and the like.
- the metal container of the present invention is formed by forming a coating film comprising the coating composition of the present invention on the inner surface and / or outer surface of the metal container, and the coating composition of the present invention is particularly excellent in corrosion resistance, flavor properties, etc. Therefore, it is desirable that a coating film made of the coating composition of the present invention is formed on at least the inner surface of the metal container.
- a metal container which forms a coating film all conventionally well-known metal containers can be used, However, It is not limited to this, The three-piece can which has a side seam, a seamless can (two-piece can) etc. can be mentioned.
- the metal container of the present invention can be molded from the above-mentioned painted metal plate. However, in the case of a metal container formed by harsh processing such as a seamless can, the present invention is applied to a pre-formed metal container. It is also possible to mold by applying the coating composition.
- the metal lid of the present invention can be formed from the above-described coated metal plate of the present invention by any conventionally known lid-making method. Generally, it is formed as a stay-on-tab type easy open can lid or a full open type easy open can lid.
- Polyester 0.2g was melt
- Initial workability Evaluated within 8 hours after preparing test coated plates.
- Processability over time Evaluated in the same manner as the initial processing after storage for 1 month in a constant temperature room at 40 ° C. within 8 hours after preparing the test coated plate.
- the evaluation criteria are as follows. ⁇ : Less than 0.5 mA ⁇ : 0.5 mA or more and less than 1.0 mA ⁇ : 1.0 mA or more and less than 3 mA ⁇ : 3 mA or more
- the test coating plate was immersed in water and treated for whitening at 125 ° C. for 30 minutes in an autoclave to evaluate the whitening property.
- Whitening property The whitening state of the coating film was visually evaluated. ⁇ : No whitening ⁇ : Slightly whitening ⁇ : Slightly whitening ⁇ : Remarkably whitening
- polyester resin (A) -a Dimethyl terephthalic acid 446 parts, trimellitic acid 2.2 parts, ethylene glycol 57 parts, propylene glycol 246 parts, 1,4-cyclohexanedimethanol 66 parts, titanium tetrabutoxide 0.2 part were charged into a 3 L four-necked flask, Assemble the polymerization equipment. The temperature was gradually raised to 220 ° C. over 4 hours to perform transesterification. This was subjected to initial polymerization under reduced pressure to 10 mmHg over 30 minutes and the temperature was raised to 250 ° C., and then the latter polymerization was performed for 90 minutes at 1 mmHg or less.
- polyester resin (B) -a 106 parts terephthalic acid, 225 parts isophthalic acid, 4 parts trimellitic anhydride, 143 parts 2-methyl-1,3-propanediol, 86 parts 1,4-butanediol, 92 parts 1,4-cyclohexanedimethanol, titanium tetrabutoxy 0.13 part was charged into a 2 L four-necked flask, and the temperature was gradually raised to 220 ° C. over 4 hours, and water was distilled off for esterification.
- the initial polymerization under reduced pressure was performed to 10 mmHg over 30 minutes and the temperature was raised to 250 ° C., and the latter polymerization was further performed at 1 mmHg or less for 50 minutes to stop the vacuum polymerization, Was taken out to obtain a polyester resin (B) -a of the present invention.
- Table 2 shows Tg, acid value, and number average molecular weight.
- the carboxylic acid anhydride is essential, the polyester resin (A)-having the acid values and Tg shown in Tables 1 and 2 is changed by changing the amount thereof and changing the type or amount of other monomers. b to (A) -i were prepared. Further, polyester resins (B) -b to (B) -g having acid values and Tg shown in Tables 1 and 2 were prepared by changing the types or amounts of the monomers.
- polyester resin (A), polyester resin (B), and solvent were placed in Kolben and heated, and dissolved with good stirring at 100 ° C. or lower. After completely dissolving, the polyester resin solution was cooled to room temperature, and a predetermined cross-linking agent and a curing catalyst were added while stirring well to prepare a coating composition.
- the crosslinker was Sumitrite Durez Co., Ltd. Sumitrite Resin PR-53893A (m-cresol resol type phenol resin) and Nippon Cytec Industries, Ltd. Mycoat 506 (Butylated Melamine Resin), and the curing catalyst was Nippon Cytec Industries. Cycat 602 (amine blocked dodecylbenzene sulfonic acid) manufactured by Co., Ltd. was used. A painted metal plate and a painted metal lid were prepared from each coating composition and evaluated. The results are shown in Tables 3 and 4.
- the coating composition of the present invention is excellent in all film properties such as processability, resistance to embrittlement over time, corrosion resistance, abrasion resistance, blocking resistance, retort resistance, etc. It can be suitably used for a coated metal plate that is baked in a coil oven, where embrittlement with time occurs remarkably.
- the coated metal plate having a coating film from the coating composition of the present invention is excellent in workability, resistance to embrittlement with time, wear resistance, and blocking resistance, and is subjected to severe processing. Can be suitably used for molding.
- the metal container or metal lid of the present invention has excellent corrosion resistance, retort resistance, and flavor, it is suitably used for metal containers that store corrosive contents or contents that require retort sterilization. be able to.
Abstract
Description
例えば、下記特許文献1には、水酸基含有ポリエステル樹脂と特定のフェノール樹脂架橋剤と酸触媒とを所定量配合して成る塗料組成物が提案されており、この塗料は、加工性、と硬度とのバランスに優れ、密着性、硬化性、衛生性及び耐沸騰水性に優れた塗膜を形成できることが記載されている。下記特許文献2及び3には、分子鎖中にペンダント状のカルボキシル基が導入された分子を含むポリエステル樹脂を用いてなる塗料用樹脂組成物が記載されており、特許文献2記載の水系樹脂組成物においては、硬化性、加工性、耐レトルト性、衛生性、スプレー塗装性、水分散性に優れていること、特許文献3記載の塗料用樹脂組成物においては、金属表面への密着性に特に優れ、かつ高度な加工性と耐食性が満足できることが記載されている。
下記特許文献4には、ポリアルコール成分として2-メチル-1,3-プロパンジオール、1,4-ブタンジオール及び1,4-シクロヘキサンジメタノールを必須成分とする低Tgのポリエステル樹脂と架橋剤を含有するポリエステル樹脂組成物が提案されており、加工性、耐レトルト性、フレーバー性、耐内容物性、フェザリング性、耐硫性を備え、更にはEOE用内面塗料として必要な高度な加工性、フェザリング性を備え持ち、加工性の経時低下を抑制し得ることが記載されている。また下記特許文献5には、高分子ポリエステル樹脂を適度に分岐させると共に、上下非対称の2価アルコール成分を含有する塗料組成物が提案されており、塗装被膜の加工性が経時において低下せず、素材との密着性、硬度、加工性のバランスに優れていることが記載されている。
特許文献4にはポリエステル樹脂のガラス転移温度(Tg)が20~50℃であるものが好ましいとの記載があるが、特許文献5ではガラス転移温度については言及されていない。
従って本発明の目的は、耐経時脆化性を更に向上するとともに、加工性、耐食性、耐削れ性、耐ブロッキング性、耐レトルト性等の塗膜性能の全てに優れた塗膜が得られる塗料組成物を提供することである。
本発明の他の目的は、優れた塗膜性能を有し、金属容器又は金属蓋の成形に好適に使用可能な塗装金属板を提供することである。
本発明の更に他の目的は、耐食性、耐レトルト性に優れた金属容器又は金属蓋を提供することである。
本発明の塗料組成物においては、
1.ポリエステル樹脂(A)が、分子内に一つ以上のカルボン酸無水物構造を有する化合物の開環付加反応で樹脂の酸価を与えられたポリエステル樹脂であること、
2.ポリエステル樹脂(A)とポリエステル樹脂(B)の重量比が、(A):(B)=99:1~50:50であること、
3.混合ポリエステル樹脂の、下記式(1)から算出されるガラス転移温度(Tgmix)が35℃以上であること、
1/Tgmix=Wa/Tga+Wb/Tgb ・・・(1)
式中、Tgmixは混合ポリエステル樹脂のガラス転移温度(K)、Tgaはポリエステル樹脂(A)のガラス転移温度(K)、Tgbはポリエステル樹脂(B)のガラス転移温度(K)、Waはポリエステル樹脂(A)の重量分率、Wbはポリエステル樹脂(B)の重量分率、をそれぞれ示す。
4.架橋剤が、レゾール型フェノール樹脂及び/又はアミノ樹脂から成り、前記ポリエステル樹脂(A)及び(B)の樹脂固形分100重量部に対して、1~30重量部の量で配合されること、
5.硬化触媒が、全樹脂固形分100重量部に対して、0.1~3.0重量部の量で配合されること、
が好適である。
本発明の塗装金属板においては、塗料組成物をコイル焼き付けして成ることが好適である。
本発明によれば更に、塗料組成物から成る塗膜が形成されてなる金属容器又は金属蓋が提供される。
即ち、硬化直後の塗膜に経時によるエンタルピー緩和によって分子の再配向(平衡状態への移行)が起こることに起因して、塗膜が脆化し、加工性が低下すると考えられる。この経時脆化現象を抑制するには、硬化塗膜の架橋密度を高めて、分子運動を抑制することによりエンタルピー緩和を抑制すること、或いは経時保存温度においても分子運動を継続維持(非平衡状態の維持)することによってエンタルピー緩和を生じさせないようにすること、の二つの方法が考えられる。
しかしながら、前者の方法では、硬化塗膜は初期から十分な加工性が得られず、また後者の方法では、硬化塗膜の経時脆化現象は抑制できるとしても、内容物の耐食性、成形加工時の耐削れ性、塗装金属板の耐ブロッキング性、殺菌処理時の耐レトルト性が十分でなく、何れの方法においても、実使用上十分な塗膜性能が得られないことが解った。
このような観点から、本発明の塗料組成物においては、経時脆化現象の原因である、エンタルピー緩和を妨げるため分子運動を抑制可能な、架橋密度を高めるポリエステル樹脂(A)、及び硬化塗膜の経時保存温度においても分子運動を継続維持可能な、Tgの低いポリエステル樹脂(B)、を組合せで使用することにより、耐経時脆化性、加工性、耐食性、耐削れ性、耐ブロッキング性、耐レトルト性等の塗膜性能の全てが優れた塗膜を形成可能な塗料組成物の発明に至った。
また本発明の塗料組成物を塗布して成る塗装金属板は、上述した優れた塗膜性能を具備しており、金属容器又は金属蓋に好適に使用することができる。
更に本発明の金属容器及び金属蓋は、優れた耐食性、耐レトルト性を有している。
すなわち、本発明の塗料組成物を用いてなる塗装金属板及び金属蓋は、成形初期及び経時後の何れの場合も加工性に優れ、耐食性、耐レトルト性、耐ブロッキング性、耐削れ性の全てにおいて優れた結果が得られているのに対して(実施例1~20)、本発明の要件を一つでも満たさない場合には、これらの塗膜性能の全てを満たすことはできないことがわかる(比較例1~14)。
尚、実施例及び比較例において、金属蓋においてのみ評価を行っているのは、金属蓋は塗装金属板を加工することにより成形されるのに対して、一般に塗装金属容器は容器を成形した後、塗装を施しており、塗装金属蓋における塗膜の方が加工の程度が大きいことから、缶蓋で良好な評価が得られれば、当然金属容器においても良好な評価が得られるので、金属蓋においてのみ評価を行っている。
[ポリエステル樹脂(A)]
本発明の塗料組成物に用いるポリエステル樹脂(A)は、酸価が2~50mgKOH/g、特に5~30mgKOH/gの範囲にあり、ガラス転移温度(Tg)が35~100℃、特に50~90℃の範囲にある限り、種々のカルボン酸成分及びアルコール成分から成るポリエステル樹脂を用いることができる。
本発明においてポリエステル樹脂(A)の酸価は、上記範囲にあることが重要であり、これにより金属基材との密着性を高め、加工性の向上に寄与する。特に基材との密着が上がると腐食誘因物質(塩類や酸)が金属に到達しにくくなり、優れた耐食性を示すことが可能になる。また、架橋密度を高め硬化塗膜の経時によるエンタルピー緩和による塗膜脆化を抑制することに寄与する。酸価が上記範囲よりも小さい場合には、基材との密着性が十分に得られず加工性、耐食性の両立が困難となる場合があり、また、架橋密度が高まり難く硬化塗膜の経時脆化を抑制する効果が十分に得られない場合がある。酸価が上記範囲よりも大きい場合には、塗膜の耐水性が低下する場合がある。
またポリエステル樹脂(A)のガラス転移温度(Tg)が、上記範囲よりも小さいと、耐ブロッキング性が劣る場合がある。また上記範囲よりもTgが高いと塗料組成物の溶剤への溶解性に劣る場合がある。
このようなポリエステルを得る方法としては、これに限定されないが、例えば特開2003-213201号公報に記載された、分子内に一つ以上のカルボン酸無水物基を有する化合物の開環付加反応を挙げることができる。
このように、分子内に一つ以上のカルボン酸無水物構造を有する化合物の開環付加反応で樹脂酸価を与えられたポリエステル樹脂(A)は、その開環付加反応に使用する全カルボン酸無水物を100モル%としたとき、その使用するカルボン酸無水物のうち10モル%以上は分子内に二つ以上のカルボン酸無水物基を有する化合物であることが好ましい。
分子内に二つ以上のカルボン酸無水物基を有する化合物はポリエステル樹脂末端のみならず、樹脂分子鎖中にカルボキシル基を修飾可能である。カルボキシル基を樹脂分子鎖中に修飾することで、分子量に制限無く酸価を付与することが可能となる。また、カルボキシル基間分子量が低いためか基材密着性が更に向上しており、耐食性に一段と優れた塗膜を得ることができる。
本発明の塗料組成物に用いるポリエステル樹脂(B)は、酸価が0~50mgKOH/g、特に0~20mgKOH/gの範囲にあり、ガラス転移温度(Tg)が-20~25℃、特に0~15℃の範囲にある限り、種々のカルボン酸成分及びアルコール成分から成るポリエステル樹脂を用いることができる。酸価は上記範囲にあることが重要であり、上記範囲より大きい場合、塗膜の耐水性が低下する場合がある。ガラス転移温度(Tg)は上記範囲にあることが重要であり、上記範囲より大きい場合、十分な耐経時脆化性が得られない。また、上記範囲より小さい場合、耐食性、耐ブロッキング性、耐レトルト性が不十分となることがある。
カルボン酸成分及びアルコール成分としては、ポリエステル樹脂(A)について例示したカルボン酸成分及びアルコール成分を用いることができる。
本発明においてポリエステル樹脂(B)は、上記アルコール成分の中でも、2-メチル-1,3-プロパンジオール、1,4-ブタンジオールおよび1,4-シクロヘキサンジメタノールを含有することが好適である。
本発明においては、架橋剤としてレゾール型フェノール樹脂及び/又はアミノ樹脂を用いることが好ましい。
レゾール型フェノール樹脂としては、例えばフェノールモノマーとしてはo-クレゾール、p-クレゾール、p-tert-ブチルフェノール、p-エチルフェノール、2,3-キシレノール、2,5-キシレノール、フェノール、m-クレゾール、m-エチルフェノール、3,5-キシレノール、m-メトキシフェノール等が挙げられ、これらは1種または2種以上を混合して使用でき、これらフェノールモノマーとホルムアルデヒドとをアルカリ触媒の存在下で反応させたものである。また含有するメチロール基の一部ないしは全部を炭素数1~12なるアルコール類でエーテル化したものも使用出来る。その中でも、メタクレゾール樹脂が好ましい。例えば、住友デュレズ(株)製のスミライトレジンPR-53893A(固形分50%)、昭和電工(株)製CKS-3898(固形分50%)等の市販品を用いることができる。
市販品としては、例えば、日本サイテックインダストリーズ(株)製のサイメル303(固形分100%)、サイメル235(固形分100%)、マイコート506(固形分100%)、サイメル1123(固形分100%)、UFR65(固形分100%)、UFR300(固形分60%)等を用いることができる。
本発明においては、硬化触媒を用いることにより、架橋反応が促進され、より緻密な架橋を低温、短時間で効率よく行うことが可能になる。
硬化触媒としてはp-トルエンスルホン酸、ドデシルベンゼンスルホン酸、カンファースルホン酸等の有機スルホン酸化合物、およびこれらのアミン中和体など、リン酸化合物等を使用することができる。
市販されているものでは、NACURE4054J(リン酸、KING INDUSTRIES製、)、NACURE5076(ドデシルベンゼンスルホン酸、KING INDUSTRIES製)、サイキャット4040(芳香族スルホン酸、日本サイテックインダストリーズ製)、サイキャット4050(アミンブロック芳香族スルホン酸、日本サイテックインダストリーズ製)、サイキャット600(芳香族スルホン酸、日本サイテックインダストリーズ製)、サイキャット602(アミンブロック芳香族スルホン酸、日本サイテックインダストリーズ製)等を用いることができる。
本発明の塗料組成物は、前述したポリエステル樹脂(A)及びポリエステル樹脂(B)を混合した混合ポリエステル樹脂に、架橋剤、硬化触媒を含有してなるものである。
ポリエステル樹脂(A)とポリエステル樹脂(B)の配合割合は、重量比で(A):(B)=99:1~50:50、特に98:2~70:30であることが好ましい。Tgの低いポリエステル樹脂(B)の比率が1以下になると耐経時脆化性が著しく低下し、塗装板の経時後加工性は初期の加工性を維持できなくなり、また比率50以上になると耐ブロッキング性が低下し、実使用上問題となる場合がある。
また本発明の塗料組成物においては、ポリエステル樹脂(A)及びポリエステル樹脂(B)を混合した混合ポリエステル樹脂の、前記式(1)で算出されるガラス転移温度が35℃以上、特に40~80℃の範囲にあることが望ましい。上記範囲よりもTgが小さいと、耐ブロッキング性が劣るようになる。また缶内面塗料として用いる場合には、フレーバー性を必要とするため40℃以上のTgであることが好ましい。また塗料組成物の溶剤への溶解性等の観点から、上限は80℃以下であることが好ましい。
また硬化触媒は、混合ポリエステル樹脂の樹脂分及び架橋剤等のその他の樹脂分を含む全樹脂固形分100重量部に対して、0.1~3.0重量部、特に0.1~2.0重量部の範囲にあることが好適である。上記範囲よりも硬化触媒の配合量が少ない場合には、硬化触媒を配合することにより得られる硬化反応を促進する効果が充分得られず、その一方上記範囲よりも硬化触媒の配合量が多い場合には、硬化促進効果が大きく加工性が不十分となる場合がある。
本発明の塗料組成物には、滑剤、消泡剤、レベリング剤、顔料等の従来公知の添加剤を公知の処方で添加することが可能である。
また本発明の塗料組成物においては、従来公知の方法により水性化して水性塗料組成物として使用することもでき、例えば、前述したポリエステル樹脂(A)及び(B)、架橋剤、硬化触媒と共に、アンモニアや有機アミン化合物等の塩基性化合物、水、或いは更に有機溶剤を配合して水性塗料組成物とする。
本発明の塗装金属板は、本発明の塗料組成物を金属板に塗装することにより得られる。
使用し得る金属板としては、例えば、熱延伸鋼板、冷延鋼板、溶融亜鉛メッキ鋼板、電気亜鉛メッキ鋼板、合金メッキ鋼板、アルミニウム亜鉛合金メッキ鋼板、アルミニウム板、スズメッキ鋼板、ステンレス鋼板、銅板、銅メッキ鋼板、ティンフリースチール、ニッケルメッキ鋼板、極薄スズメッキ鋼板、クロム処理鋼板などが挙げられ、必要に応じてこれらに各種表面処理、並びにプライマー処理を行ったものが使用される。
塗膜の厚みは特に限定されないが、乾燥膜厚で3~18μm、更には3~12μmの範囲であることが好ましい。乾燥膜厚は塗装金属板の用途により適宜決められる。塗膜の焼き付け条件は使用したポリエステル樹脂、架橋剤の種類、使用した溶剤や塗装する金属材料の種類、厚さ、塗装速度などにより適宜調節される。
本発明の金属容器は、金属容器の内面及び/又は外面に本発明の塗料組成物から成る塗膜が形成されてなるものであり、本発明の塗料組成物は特に耐食性、フレーバー性等に優れているので、金属容器の少なくとも内面に本発明の塗料組成物から成る塗膜が形成されていることが望ましい。
塗膜を形成する金属容器としては、従来公知の金属容器を全て用いることができ、これに限定されないが、側面継ぎ目を有するスリーピース缶、シームレス缶(ツーピース缶)等を挙げることができる。
本発明の金属容器は、前述した塗装金属板から容器を成形することもできるが、シームレス缶のような過酷な加工により成形される金属容器の場合には、予め成形された金属容器に本発明の塗料組成物を施すことによって成形することもできる。
各測定項目は以下の方法に従った。
ゲル浸透クロマトグラフィー(GPC)によって標準ポリスチレンの検量線を用いて測定した。
示差走査熱量計(DSC)を用いて20℃/分の昇温速度で測定した。
ポリエステル0.2gを20mlのクロロホルムに溶解し、0.1NのKOHエタノール溶液で滴定し、樹脂酸価(mgKOH/g)を求めた。指示薬はフェノールフタレインを用いた。
実施例、比較例の各塗料組成物を厚さ0.28mmの#5182アルミニウム板にバーコーターで乾燥塗膜重量100mg/100cm2塗装し、コイルオーブンで焼き付ける。焼き付け条件はアルミニウム板到達最高温度(PMT)260℃、全焼き付け時間(TOT)28秒である。得られた塗装板は下記の試験方法に基づいて各種試験を行った。
耐経時脆化性は初期加工性と経時加工性を評価した。
・加工性
試験塗装板をアルミニウム板の圧延方向が長辺となるように3×5cmの大きさに切り出す。この試験片の塗装面が外になるように短辺に平行に折り曲げる。25℃の雰囲気下で折り曲げ部の内側に0.28mmのアルミニウム板を2枚挟み、ハゼ折タイプデユポン衝撃試験器を用い衝撃屈曲させる。衝撃屈曲させる接触面が平らな鉄の錘の重さは3.5kgで、これを高さは12.5cmから落下させる。この折り曲げられた先端部分の2cm幅の電流値(mA)を1%塩化ナトリウム水溶液に浸漬したスポンジに接触させ、電圧6.2Vをかけ4秒後に測定した。
経時加工性:試験塗装板を作成後8時間以内に40℃の恒温室に1か月保管後初期加工と同様に評価した。
評価基準は次の通りである。
◎:0.5mA未満
○:0.5mA以上1.0mA未満
△:1.0mA以上3mA未満
×:3mA以上
デュポン衝撃試験器(衝撃棒1/4インチ、錘荷重300gを12.5cmから落下させる。)で凸加工した塗装板を内容物疑似液(クエン酸/エタノール/水=5/10/85)に40℃で7日間浸漬し、腐食の程度を目視で評価した。
◎:腐食なし
○:僅かに腐食
△:少し腐食
×:著しい腐食
試験塗装板を水に浸漬し、オートクレーブ中で125℃30分処理後白化性を評価した。
白化性:塗膜の白化状態を目視で評価した。
◎:白化なし
○:僅かに白化
△:少し白化
×:著しく白化
8×8cmに切断した塗装板の塗装面と塗装面を合わせ加熱圧着プレス機を用い、50℃の温度で0.36MPaの圧力を2分間掛けた後室温まで冷却し、塗装面と塗装面のブロッキング程度を塗装板端面から垂直に剥がしブロッキング程度を評価した。
○:ブロッキングしない
△:少しブロッキングする
×:著しくブロッキングする
摺動試験機 HEIDON-14DR(新東科学(株)製)を用い、荷重を掛けたボールベアリングで塗膜表面を摺動させ、塗膜が削れアルミニウム基材に達するまでの摺動回数で評価した。直径10mmステンレスボール、摺動スピード6000mm/min.、摺動距離10mm、荷重1kg、塗装板温度25℃。
◎:1,000回以上
○:500回以上1,000回未満
△:100回以上500回未満
×:100回未満
前記塗装板を使用し、シェル(開封用タブ取付け前の蓋をシェルと呼称する)成形機にて、強化環状溝のラジアス部の中央パネルからの深さが2.5mm、およびラジアス部の曲率半径が0.50mmである206径シェルを成形した。これに蓋外面側からスコア加工(スコア残存厚み95μm)、リベット加工および開封用タブの取付けを行い、蓋を作製した。
(5)-1 蓋耐経時脆化性
経時加工性:塗装後40℃-1ヶ月保管したものをシェル成形し、コンパウンド塗布後さらに40℃-2週間保管してからエンド成形(スコア加工・タブ取り付け等)を実施した。
前記で得られた蓋について、下記評価を実施した。
塗装直後(RT8時間以内)と1ヶ月保管(40℃)後成形した塗装蓋5個の通電値を測定し、その平均値の差で評価した。通電値の測定条件は電圧6.2Vで4秒通電後の電流値を測定した。
◎:0.2mA以内(数値上昇なし)
○:0.5mA以内(数値上昇なし)
△:1.0mA以内(数値上昇なし)
×:数値上昇あり
両面PETラミネートティンフリースチールを絞りしごきした350g缶に、内容物としてコカコーラをガスボリューム3.75、内容量345gの条件でコールドパック(液温5℃,缶内エアー量2ml以下にする)後、ウォーマー処理(40℃-10分)を実施した。得られたサンプルを37℃の恒温室に6ヶ月間倒立(蓋を下側にして保管)経時後、開缶し蓋内面を観察した。観察は実体顕微鏡及び金属顕微鏡を用いて行い、蓋内面の腐食点,穿孔部有無を評価した。(各n=5枚)
○:腐食なし,軽微腐食(スコア,リベット以外)
×:穿孔或いは穿孔の可能性有
両面PETラミネートティンフリースチールを絞りしごきした350g缶に、水340gを充填し内圧が0.1kgf/cm2となるよう、液体窒素を滴下後にアルミ蓋を巻締めた。この缶に125℃-30minの蒸気・静置レトルト処理を行い加熱終了時点から10分以内に内容液温が40℃未満となるように冷却した。
その蓋を取り外して蓋内面の白化状態を評価した。各条件n=3枚。
◎:白化なし
○:僅かに白化
△:少し白化
×:著しく白化
コンバージョンプレスにて各水準50枚連続加工を行い、リベット側壁部の塗膜状態を目視観察し、塗膜ダメージ部については、金属露出の有無を20%硫酸銅水溶液に1分間浸漬しCu析出により判定した。
○:塗膜ダメージなし
△:表面的ダメージあり(金属露出なし)
×:削れ発生(金属露出あり)
合成例:ポリエステル樹脂(A)-a
ジメチルテレフタル酸446部、トリメリト酸2.2部、エチレングリコール57部、プロピレングリコール246部、1,4-シクロヘキサンジメタノール66部、チタンテトラブトキシド0.2部を3Lの四つ口フラスコに仕込み、重合装置を組む。4時間かけて220℃まで徐々に昇温しエステル交換を行なった。これを30分かけて10mmHgまで減圧初期重合を行なうとともに温度を250℃まで昇温し、更にこのまま1mmHg以下で90分間後期重合を行なった。目標分子量に達したらこれを窒素雰囲気下で220℃に冷却した。次いでエチレングリコールビストリメリテート二無水物20部、無水トリメリト酸5部を相次いで投入し、窒素雰囲気下、200~230℃、1時間攪拌を継続した。これを取り出し本発明のポリエステル樹脂(A)-aを得た。Tg、酸価、数平均分子量は表1に示す。
合成例:ポリエステル樹脂(B)-a
テレフタル酸106部、イソフタル酸225部、無水トリメリト酸4部、2-メチルー1,3-プロパンジオール143部、1,4-ブタンジオール86部、1,4-シクロヘキサンジメタノール92部、チタンテトラブトキシ0.13部を2Lの四つ口フラスコに仕込み、4時間かけて220℃まで徐々に昇温し、水を留出させエステル化を行った。所定量の水を留出させた後、30分かけて10mmHgまで減圧初期重合を行うとともに温度を250℃まで昇温し、更にこのまま1mmHg以下で50分間後期重合を行い、減圧重合を止め、樹脂を取り出し本発明のポリエステル樹脂(B)-aを得た。Tg、酸価、数平均分子量は表2に示す。
まず所定量のポリエステル樹脂(A),ポリエステル樹脂(B)、溶剤をコルベンに入れ加温し、100℃以下で良く攪拌しながら溶解した。完全に溶解した後ポリエステル樹脂溶液を室温まで冷却し、良く攪拌しながら所定の架橋剤、硬化触媒を添加し塗料組成物を調製した。架橋剤は、住友デュレズ(株)製スミライトレジンPR-53893A(m-クレゾールレゾール型フェノール樹脂)及び日本サイテックインダストリーズ(株)製マイコート506(ブチル化メラミン樹脂)を、硬化触媒は日本サイテックインダストリーズ(株)製サイキャット602(アミンブロックドデシルベンゼンスルホン酸)を使用した。各塗料組成物から塗装金属板及び塗装金属蓋を作製し、評価を行った。結果を表3及び表4に示す。
また本発明の塗料組成物から塗膜を有する塗装金属板は、加工性、耐経時脆化性、耐削れ性、耐ブロッキング性に優れており、厳しい加工に賦される、金属容器又は金属蓋の成形に好適に使用することができる。
更に本発明の金属容器又は金属蓋は、優れた耐食性、耐レトルト性、フレーバー性を有することから、腐食性の内容物や、レトルト殺菌が必要な内容物を収納する金属容器に好適に使用することができる。
Claims (10)
- 酸価が2~50mgKOH/g、ガラス転移温度(Tg)が35~100℃であるポリエステル樹脂(A)と、酸価0~50mgKOH/g、ガラス転移温度(Tg)が-20~25℃であるポリエステル樹脂(B)を混合した混合ポリエステル樹脂に、架橋剤、硬化触媒を含有することを特徴とする塗料組成物。
- 前記ポリエステル樹脂(A)が、分子内に一つ以上のカルボン酸無水物構造を有する化合物の開環付加反応で樹脂の酸価を与えられたポリエステル樹脂である請求項1記載の塗料組成物。
- 前記ポリエステル樹脂(A)と前記ポリエステル樹脂(B)の重量比が、(A):(B)=99:1~50:50である請求項1又は2記載の塗料組成物。
- 前記混合ポリエステル樹脂の、下記式(1)から算出されるガラス転移温度(Tgmix)が35℃以上である請求項1~3の何れかに記載の塗料組成物。
1/Tgmix=Wa/Tga+Wb/Tgb ・・・(1)
式中、Tgmixは混合ポリエステル樹脂のガラス転移温度(K)、Tgaはポリエステル樹脂(A)のガラス転移温度(K)、Tgbはポリエステル樹脂(B)のガラス転移温度(K)、Waはポリエステル樹脂(A)の重量分率、Wbはポリエステル樹脂(B)の重量分率、をそれぞれ示す。 - 前記架橋剤が、レゾール型フェノール樹脂及び/又はアミノ樹脂から成り、前記ポリエステル樹脂(A)及び(B)の樹脂固形分100重量部に対して、1~30重量部の量で配合される請求項1~4の何れかに記載の塗料組成物。
- 前記硬化触媒が、全樹脂固形分100重量部に対して、0.1~3.0重量部の量で配合される請求項1~5の何れかに記載の塗料組成物。
- 請求項1~6の何れかに記載の塗料組成物を塗布した塗装金属板。
- 前記塗料組成物をコイル焼き付けして成る請求項7記載の塗装金属板。
- 請求項1~6の何れかに記載の塗料組成物から成る塗膜が形成されてなる金属容器。
- 請求項1~6の何れかに記載の塗料組成物から成る塗膜が形成されてなる金属蓋。
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN9899DEN2014 IN2014DN09899A (ja) | 2012-05-31 | 2013-05-27 | |
| BR112014029736-3A BR112014029736B1 (pt) | 2012-05-31 | 2013-05-27 | Composição de revestimento, placa de metal revestida, recipiente de metal, e, fecho de metal |
| CA2873357A CA2873357C (en) | 2012-05-31 | 2013-05-27 | Coating composition and coated metal plate, metal container and metal closure coated with the coating composition |
| KR1020147033396A KR101752958B1 (ko) | 2012-05-31 | 2013-05-27 | 도료 조성물 및 이 도료 조성물을 도포하여 이루어진 도장 금속판, 금속 용기 및 금속 덮개 |
| US14/404,192 US10508214B2 (en) | 2012-05-31 | 2013-05-27 | Coating composition and coated metal plate, metal container and metal closure coated with the coating composition |
| CN201380028567.3A CN104379687B (zh) | 2012-05-31 | 2013-05-27 | 涂料组合物和涂布有所述涂料组合物的涂装金属板、金属容器和金属盖 |
| EP13796325.2A EP2857465B1 (en) | 2012-05-31 | 2013-05-27 | Coating composition, and coated metal plate, metal container and metal lid formed by coating with said coating composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-124819 | 2012-05-31 | ||
| JP2012124819A JP5462318B2 (ja) | 2012-05-31 | 2012-05-31 | 塗料組成物及びこの塗料組成物を塗布して成る塗装金属板、金属容器及び金属蓋 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013180067A1 true WO2013180067A1 (ja) | 2013-12-05 |
Family
ID=49673261
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/064659 WO2013180067A1 (ja) | 2012-05-31 | 2013-05-27 | 塗料組成物及びこの塗料組成物を塗布して成る塗装金属板、金属容器及び金属蓋 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US10508214B2 (ja) |
| EP (1) | EP2857465B1 (ja) |
| JP (1) | JP5462318B2 (ja) |
| KR (1) | KR101752958B1 (ja) |
| CN (1) | CN104379687B (ja) |
| BR (1) | BR112014029736B1 (ja) |
| CA (1) | CA2873357C (ja) |
| IN (1) | IN2014DN09899A (ja) |
| WO (1) | WO2013180067A1 (ja) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3075801A1 (en) | 2015-04-03 | 2016-10-05 | Holland Novochem Technical Coatings B.V. | Coating composition |
| EP3211049A1 (en) | 2015-04-03 | 2017-08-30 | Holland Novochem Technical Coatings B.V. | Coating composition |
| WO2018005430A1 (en) * | 2016-06-27 | 2018-01-04 | Warner Babcock Institute For Green Chemistry, Llc | Bisphenol a-free crosslinked polymer compositions |
| EP3205495A4 (en) * | 2014-10-06 | 2018-05-02 | Toyo Seikan Group Holdings, Ltd. | Organic resin-coated coated metal plate, and can body and can lid comprising same |
| EP2989016B1 (en) | 2013-10-02 | 2021-08-04 | Swimc Llc | Removable closure and coating system |
| WO2022202831A1 (ja) * | 2021-03-22 | 2022-09-29 | 東洋紡株式会社 | ポリエステル樹脂組成物、水分散体、塗料組成物および塗膜 |
| EP2996954B1 (en) | 2013-10-02 | 2023-04-05 | Swimc Llc | Removable closure and coating system |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6253553B2 (ja) * | 2014-03-27 | 2017-12-27 | 関西ペイント株式会社 | 塗料組成物 |
| JP6280821B2 (ja) * | 2014-06-10 | 2018-02-14 | 株式会社Uacj | 放熱性アルミニウム塗装材 |
| JP5958726B1 (ja) * | 2015-02-06 | 2016-08-02 | 東洋インキScホールディングス株式会社 | 塗料組成物および缶蓋 |
| EP3350241B1 (en) * | 2015-09-14 | 2022-08-24 | LCY Biosciences Inc. | Curable coating compositions using succinic acid |
| CN111100480B (zh) * | 2016-03-30 | 2022-06-10 | 埃卡特有限公司 | 用于粉末涂漆的涂覆有机粘合剂的效应颜料、用于生产所述涂覆的效应颜料的方法及其用途 |
| JP2018021098A (ja) * | 2016-08-01 | 2018-02-08 | 東洋インキScホールディングス株式会社 | 塗料組成物 |
| KR101878068B1 (ko) * | 2016-10-14 | 2018-07-12 | 강남제비스코 주식회사 | Ropp ed cap 내면 도료 조성물 |
| JP7067253B2 (ja) * | 2017-05-11 | 2022-05-16 | 東洋製罐グループホールディングス株式会社 | 水性塗料組成物 |
| EP3623435A4 (en) * | 2017-05-11 | 2021-01-27 | Toyo Seikan Group Holdings, Ltd. | AQUEOUS COATING COMPOSITION AND PROCESS FOR THE PRODUCTION OF COATED METAL SUBSTRATE |
| JP7355485B2 (ja) * | 2018-02-02 | 2023-10-03 | 東洋製罐株式会社 | ワイン用アルミニウム製容器 |
| EP3753991B1 (en) * | 2018-02-13 | 2024-03-06 | Toyo Seikan Group Holdings, Ltd. | Aqueous coating material composition |
| JP7346852B2 (ja) * | 2018-03-14 | 2023-09-20 | 東洋製罐グループホールディングス株式会社 | 水性塗料組成物 |
| WO2020100776A1 (ja) * | 2018-11-13 | 2020-05-22 | 東洋製罐グループホールディングス株式会社 | 塗料組成物及び該塗料組成物から成る塗膜を有する塗装金属基体 |
| WO2020251153A1 (ko) * | 2019-06-14 | 2020-12-17 | 주식회사 케이씨씨 | 하도 도료 조성물 |
| KR102310666B1 (ko) * | 2019-06-14 | 2021-10-12 | 주식회사 케이씨씨 | 하도 도료 조성물 |
| CN110330878B (zh) * | 2019-07-31 | 2022-04-01 | 广东佰朋实业有限公司 | 高拉伸的深冲午餐肉罐头内壁涂料配方、制备及应用工艺 |
| EP4179002A1 (en) * | 2020-07-08 | 2023-05-17 | Bostik, Inc. | Amorphous copolyester resin for industrial coatings and methods for coating a metal surface using such coating compositions |
| JP2022048919A (ja) * | 2020-09-15 | 2022-03-28 | 東洋製罐株式会社 | ワイン用アルミニウム製容器 |
| US20240052195A1 (en) * | 2020-10-27 | 2024-02-15 | Eastman Chemical Company | Polyester polyol compositions for metal packaging coatings |
| EP4236742A4 (en) * | 2020-10-27 | 2024-03-13 | Eastman Chem Co | POLYESTER COMPOSITIONS FOR METAL PACKAGING COATINGS |
| EP4236743A4 (en) * | 2020-10-27 | 2024-03-13 | Eastman Chem Co | POLYESTER COMPOSITIONS FOR METAL PACKAGING COATINGS |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10130571A (ja) * | 1996-10-25 | 1998-05-19 | Kansai Paint Co Ltd | 塗料組成物 |
| JP2001131470A (ja) | 1999-08-25 | 2001-05-15 | Kansai Paint Co Ltd | 塗料組成物 |
| JP2002201411A (ja) | 2000-11-06 | 2002-07-19 | Kansai Paint Co Ltd | 塗料組成物 |
| JP2003089746A (ja) | 2001-07-06 | 2003-03-28 | Toyobo Co Ltd | 水系樹脂組成物、これを含む水系塗料、その塗料を用いた塗膜、その塗膜を用いた金属板および缶 |
| JP2003213201A (ja) | 2002-01-24 | 2003-07-30 | Toyobo Co Ltd | 塗料用樹脂組成物、およびこれを塗布した塗装金属板 |
| JP2003301137A (ja) * | 2002-04-11 | 2003-10-21 | Nippon Ester Co Ltd | 塗料組成物、及び塗装金属板 |
| JP2004346131A (ja) | 2003-05-20 | 2004-12-09 | Toyobo Co Ltd | ポリエステル樹脂および組成物 |
| JP2005126635A (ja) * | 2003-10-27 | 2005-05-19 | Dainippon Ink & Chem Inc | 缶用塗料組成物 |
| JP2006037014A (ja) * | 2004-07-29 | 2006-02-09 | Nippon Ester Co Ltd | 塗料用組成物 |
| JP2006077118A (ja) * | 2004-09-09 | 2006-03-23 | Nippon Ester Co Ltd | 塗料用樹脂組成物 |
| WO2011003761A1 (en) * | 2009-07-07 | 2011-01-13 | Cytec Surface Specialties, S.A. | Low temperature cure powder coating compositions |
| JP2011016360A (ja) * | 2002-08-01 | 2011-01-27 | Valspar Sourcing Inc | 金属基材用塗料組成物 |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6184311B1 (en) * | 1990-03-26 | 2001-02-06 | Courtaulds Coatings (Holdings) Limited | Powder coating composition of semi-crystalline polyester and curing agent |
| US6458439B1 (en) * | 1996-05-17 | 2002-10-01 | The Valspar Corporation | Extrusion coating compositions and method |
| NL1005841C2 (nl) | 1997-04-18 | 1998-10-20 | Dsm Nv | Can- en coilcoatingharsen. |
| US6613840B2 (en) | 2000-12-19 | 2003-09-02 | Dainippon Ink And Chemicals, Inc. | Resin composition for powder coating, powder coating, and coated article therewith |
| CN1404516A (zh) * | 2000-12-21 | 2003-03-19 | Ucb公司 | 涂料用粉末热固性组合物 |
| JP2002302639A (ja) * | 2001-04-06 | 2002-10-18 | Kansai Paint Co Ltd | 缶内面用水性被覆組成物 |
| CN1239648C (zh) * | 2001-04-25 | 2006-02-01 | 三菱瓦斯化学株式会社 | 制备聚酯粉末涂料的方法 |
| EP1273626B1 (en) | 2001-07-06 | 2006-05-17 | Toyo Boseki Kabushiki Kaisha | Aqueous resin composition, aqueous coating material containing the composition, coating provided by the material, and metal plate coated with the material |
| EP1726621A1 (en) * | 2005-05-26 | 2006-11-29 | Cytec Surface Specialties, S.A. | Thermosetting powder compositions |
| US8470447B2 (en) * | 2006-03-06 | 2013-06-25 | Toyo Seikan Kaisha, Ltd. | Easy open ends that can be favorably opened at high temperatures |
| EP2085441A1 (en) * | 2008-01-31 | 2009-08-05 | Cytec Surface Specialties, S.A. | Powder Composition |
| JP5522540B2 (ja) * | 2010-09-15 | 2014-06-18 | 株式会社リコー | トナー、現像剤、現像剤容器、プロセスカートリッジ、画像形成装置及び画像形成方法 |
| CN105765004B (zh) * | 2013-11-25 | 2019-04-09 | 宣伟投资管理有限公司 | 用于高填充粉末涂料的聚酯树脂 |
-
2012
- 2012-05-31 JP JP2012124819A patent/JP5462318B2/ja active Active
-
2013
- 2013-05-27 BR BR112014029736-3A patent/BR112014029736B1/pt active IP Right Grant
- 2013-05-27 KR KR1020147033396A patent/KR101752958B1/ko active IP Right Grant
- 2013-05-27 IN IN9899DEN2014 patent/IN2014DN09899A/en unknown
- 2013-05-27 CN CN201380028567.3A patent/CN104379687B/zh active Active
- 2013-05-27 EP EP13796325.2A patent/EP2857465B1/en active Active
- 2013-05-27 US US14/404,192 patent/US10508214B2/en active Active
- 2013-05-27 CA CA2873357A patent/CA2873357C/en active Active
- 2013-05-27 WO PCT/JP2013/064659 patent/WO2013180067A1/ja active Application Filing
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10130571A (ja) * | 1996-10-25 | 1998-05-19 | Kansai Paint Co Ltd | 塗料組成物 |
| JP2001131470A (ja) | 1999-08-25 | 2001-05-15 | Kansai Paint Co Ltd | 塗料組成物 |
| JP2002201411A (ja) | 2000-11-06 | 2002-07-19 | Kansai Paint Co Ltd | 塗料組成物 |
| JP2003089746A (ja) | 2001-07-06 | 2003-03-28 | Toyobo Co Ltd | 水系樹脂組成物、これを含む水系塗料、その塗料を用いた塗膜、その塗膜を用いた金属板および缶 |
| JP2003213201A (ja) | 2002-01-24 | 2003-07-30 | Toyobo Co Ltd | 塗料用樹脂組成物、およびこれを塗布した塗装金属板 |
| JP2003301137A (ja) * | 2002-04-11 | 2003-10-21 | Nippon Ester Co Ltd | 塗料組成物、及び塗装金属板 |
| JP2011016360A (ja) * | 2002-08-01 | 2011-01-27 | Valspar Sourcing Inc | 金属基材用塗料組成物 |
| JP2004346131A (ja) | 2003-05-20 | 2004-12-09 | Toyobo Co Ltd | ポリエステル樹脂および組成物 |
| JP2005126635A (ja) * | 2003-10-27 | 2005-05-19 | Dainippon Ink & Chem Inc | 缶用塗料組成物 |
| JP2006037014A (ja) * | 2004-07-29 | 2006-02-09 | Nippon Ester Co Ltd | 塗料用組成物 |
| JP2006077118A (ja) * | 2004-09-09 | 2006-03-23 | Nippon Ester Co Ltd | 塗料用樹脂組成物 |
| WO2011003761A1 (en) * | 2009-07-07 | 2011-01-13 | Cytec Surface Specialties, S.A. | Low temperature cure powder coating compositions |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2857465A4 |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2989016B1 (en) | 2013-10-02 | 2021-08-04 | Swimc Llc | Removable closure and coating system |
| EP2996954B1 (en) | 2013-10-02 | 2023-04-05 | Swimc Llc | Removable closure and coating system |
| EP3205495A4 (en) * | 2014-10-06 | 2018-05-02 | Toyo Seikan Group Holdings, Ltd. | Organic resin-coated coated metal plate, and can body and can lid comprising same |
| EP3075801A1 (en) | 2015-04-03 | 2016-10-05 | Holland Novochem Technical Coatings B.V. | Coating composition |
| EP3211049A1 (en) | 2015-04-03 | 2017-08-30 | Holland Novochem Technical Coatings B.V. | Coating composition |
| WO2018005430A1 (en) * | 2016-06-27 | 2018-01-04 | Warner Babcock Institute For Green Chemistry, Llc | Bisphenol a-free crosslinked polymer compositions |
| WO2022202831A1 (ja) * | 2021-03-22 | 2022-09-29 | 東洋紡株式会社 | ポリエステル樹脂組成物、水分散体、塗料組成物および塗膜 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20150125643A1 (en) | 2015-05-07 |
| JP2013249376A (ja) | 2013-12-12 |
| US10508214B2 (en) | 2019-12-17 |
| JP5462318B2 (ja) | 2014-04-02 |
| CA2873357A1 (en) | 2013-12-05 |
| BR112014029736A2 (pt) | 2021-07-06 |
| EP2857465B1 (en) | 2019-09-11 |
| BR112014029736B1 (pt) | 2022-01-11 |
| EP2857465A1 (en) | 2015-04-08 |
| KR101752958B1 (ko) | 2017-07-11 |
| IN2014DN09899A (ja) | 2015-08-07 |
| KR20150016950A (ko) | 2015-02-13 |
| EP2857465A4 (en) | 2016-02-10 |
| CN104379687A (zh) | 2015-02-25 |
| CN104379687B (zh) | 2017-11-10 |
| CA2873357C (en) | 2018-05-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5462318B2 (ja) | 塗料組成物及びこの塗料組成物を塗布して成る塗装金属板、金属容器及び金属蓋 | |
| JP6627771B2 (ja) | 塗装金属板及び有機樹脂被覆塗装金属板 | |
| JP5611697B2 (ja) | 樹脂組成物 | |
| JP6257385B2 (ja) | 塗料組成物 | |
| EP3205495B1 (en) | Organic resin-coated coated metal plate, and can body and can lid comprising same | |
| JP2015209463A (ja) | 塗料組成物及び塗装金属缶 | |
| JP6242000B2 (ja) | 塗料組成物及び金属缶 | |
| JP2017226726A (ja) | ポリエステル樹脂組成物、缶用塗装金属板および缶 | |
| JP2006124497A (ja) | 缶塗料用樹脂組成物及びこれを用いた塗装金属板 | |
| JP6253553B2 (ja) | 塗料組成物 | |
| JP5766416B2 (ja) | 塗料用樹脂組成物 | |
| JP2003213201A (ja) | 塗料用樹脂組成物、およびこれを塗布した塗装金属板 | |
| CN115551784A (zh) | 拉深减薄罐以及拉深减薄罐用涂装金属板 | |
| JP2006143891A (ja) | 水系缶塗料用樹脂組成物及びこれを塗布した塗装金属板 | |
| CN115052944A (zh) | 涂料组合物、涂装金属板以及拉深减薄罐及其制造方法 | |
| JP7235180B2 (ja) | 絞りしごき缶の製造方法及び絞りしごき缶 | |
| WO2023100402A1 (ja) | 塗料組成物 | |
| JP2023056058A (ja) | 絞りしごき缶 | |
| TW202146600A (zh) | 深沖壓罐 | |
| JP6708006B2 (ja) | ポリエステル樹脂、缶塗料用樹脂組成物、缶用塗装金属板および缶 | |
| TW202313409A (zh) | 無縫罐及塗裝金屬板 | |
| JP2021123718A (ja) | 塗料組成物、塗装金属板、並びに、絞りしごき缶及びその製造方法 | |
| JP2020078928A (ja) | 塗装金属板及び絞りしごき缶 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13796325 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2873357 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013796325 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14404192 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20147033396 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: IDP00201407514 Country of ref document: ID |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112014029736 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112014029736 Country of ref document: BR Kind code of ref document: A2 Effective date: 20141127 |
|
| ENPC | Correction to former announcement of entry into national phase, pct application did not enter into the national phase |
Ref document number: 112014029736 Country of ref document: BR Kind code of ref document: A2 Free format text: ANULADA A PUBLICACAO CODIGO 1.3 NA RPI NO 2433 DE 22/08/2017 POR TER SIDO INDEVIDA. |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: 112014029736 Country of ref document: BR Kind code of ref document: A2 Free format text: ESCLARECER, EM ATE 60 (SESSENTA) DIAS, APRESENTANDO DOCUMENTACAO COMPROBATORIA, A EXCLUSAO DO DEPOSITANTE VALSPAR ROCK CO., LTD. QUE CONSTA NA PUBLICACAO INTERNACIONAL WO/2013/180067 DE 05/12/2013 DO QUADRO DE DEPOSITANTES DA PETICAO NO 860140198777 DE 27/11/2014 E PETICAO NO 860140213257 DE 23/12/2014 |
|
| ENP | Entry into the national phase |
Ref document number: 112014029736 Country of ref document: BR Kind code of ref document: A2 Effective date: 20141127 |