WO2013172346A1 - 環境汚染物質を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸エチルエステル及びその製造方法 - Google Patents
環境汚染物質を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸エチルエステル及びその製造方法 Download PDFInfo
- Publication number
- WO2013172346A1 WO2013172346A1 PCT/JP2013/063425 JP2013063425W WO2013172346A1 WO 2013172346 A1 WO2013172346 A1 WO 2013172346A1 JP 2013063425 W JP2013063425 W JP 2013063425W WO 2013172346 A1 WO2013172346 A1 WO 2013172346A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- fatty acid
- unsaturated fatty
- highly unsaturated
- ethyl ester
- less
- Prior art date
Links
- 235000021122 unsaturated fatty acids Nutrition 0.000 title claims abstract description 95
- 150000004670 unsaturated fatty acids Chemical class 0.000 title claims abstract description 88
- 239000003344 environmental pollutant Substances 0.000 title claims abstract description 22
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 6
- 150000002013 dioxins Chemical class 0.000 claims abstract description 50
- 238000004821 distillation Methods 0.000 claims abstract description 45
- 125000004494 ethyl ester group Chemical group 0.000 claims abstract description 39
- 235000014113 dietary fatty acids Nutrition 0.000 claims abstract description 35
- 229930195729 fatty acid Natural products 0.000 claims abstract description 35
- 239000000194 fatty acid Substances 0.000 claims abstract description 35
- 150000004665 fatty acids Chemical class 0.000 claims abstract description 33
- 238000004440 column chromatography Methods 0.000 claims abstract description 17
- 239000010409 thin film Substances 0.000 claims abstract description 16
- 239000000470 constituent Substances 0.000 claims abstract description 13
- 235000021588 free fatty acids Nutrition 0.000 claims abstract description 8
- 150000004826 dibenzofurans Chemical class 0.000 claims abstract description 6
- 239000003921 oil Substances 0.000 claims description 65
- 235000019198 oils Nutrition 0.000 claims description 65
- 238000000034 method Methods 0.000 claims description 54
- 238000000526 short-path distillation Methods 0.000 claims description 23
- 235000021323 fish oil Nutrition 0.000 claims description 20
- 239000003925 fat Substances 0.000 claims description 18
- 235000019197 fats Nutrition 0.000 claims description 18
- 239000002994 raw material Substances 0.000 claims description 18
- MBMBGCFOFBJSGT-KUBAVDMBSA-N all-cis-docosa-4,7,10,13,16,19-hexaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCC(O)=O MBMBGCFOFBJSGT-KUBAVDMBSA-N 0.000 claims description 17
- 238000000199 molecular distillation Methods 0.000 claims description 16
- 235000020673 eicosapentaenoic acid Nutrition 0.000 claims description 13
- 235000013305 food Nutrition 0.000 claims description 13
- 235000020669 docosahexaenoic acid Nutrition 0.000 claims description 12
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical class N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 claims description 11
- XYBSIYMGXVUVGY-UHFFFAOYSA-N 2,2',4,4'-Tetrabromodiphenyl ether Chemical compound BrC1=CC(Br)=CC=C1OC1=CC=C(Br)C=C1Br XYBSIYMGXVUVGY-UHFFFAOYSA-N 0.000 claims description 10
- QWVDUBDYUPHNHY-UHFFFAOYSA-N 2,2',4,5'-Tetrabromodiphenyl ether Chemical compound BrC1=CC(Br)=CC=C1OC1=CC(Br)=CC=C1Br QWVDUBDYUPHNHY-UHFFFAOYSA-N 0.000 claims description 10
- WHPVYXDFIXRKLN-UHFFFAOYSA-N 2,4-dibromophenyl 2,4,5-tribromophenyl ether Chemical compound BrC1=CC(Br)=CC=C1OC1=CC(Br)=C(Br)C=C1Br WHPVYXDFIXRKLN-UHFFFAOYSA-N 0.000 claims description 10
- NSKIRYMHNFTRLR-UHFFFAOYSA-N 2,4-dibromophenyl 2,4,6-tribromophenyl ether Chemical compound BrC1=CC(Br)=CC=C1OC1=C(Br)C=C(Br)C=C1Br NSKIRYMHNFTRLR-UHFFFAOYSA-N 0.000 claims description 9
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 9
- 239000013589 supplement Substances 0.000 claims description 9
- 238000000746 purification Methods 0.000 claims description 8
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 claims description 7
- 235000020777 polyunsaturated fatty acids Nutrition 0.000 claims description 7
- YUFFSWGQGVEMMI-JLNKQSITSA-N (7Z,10Z,13Z,16Z,19Z)-docosapentaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCCCCC(O)=O YUFFSWGQGVEMMI-JLNKQSITSA-N 0.000 claims description 5
- 241000283153 Cetacea Species 0.000 claims description 5
- JAZBEHYOTPTENJ-JLNKQSITSA-N all-cis-5,8,11,14,17-icosapentaenoic acid Chemical compound CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O JAZBEHYOTPTENJ-JLNKQSITSA-N 0.000 claims description 5
- JAZBEHYOTPTENJ-UHFFFAOYSA-N eicosapentaenoic acid Natural products CCC=CCC=CCC=CCC=CCC=CCCCC(O)=O JAZBEHYOTPTENJ-UHFFFAOYSA-N 0.000 claims description 5
- 229940106134 krill oil Drugs 0.000 claims description 5
- 230000000813 microbial effect Effects 0.000 claims description 5
- 235000021294 Docosapentaenoic acid Nutrition 0.000 claims description 4
- 229940090949 docosahexaenoic acid Drugs 0.000 claims description 4
- 229960005135 eicosapentaenoic acid Drugs 0.000 claims description 4
- 239000004480 active ingredient Substances 0.000 claims description 3
- 235000021342 arachidonic acid Nutrition 0.000 claims description 3
- 229940114079 arachidonic acid Drugs 0.000 claims description 3
- 150000002148 esters Chemical class 0.000 claims description 3
- 230000003301 hydrolyzing effect Effects 0.000 claims description 3
- HOBAELRKJCKHQD-UHFFFAOYSA-N (8Z,11Z,14Z)-8,11,14-eicosatrienoic acid Natural products CCCCCC=CCC=CCC=CCCCCCCC(O)=O HOBAELRKJCKHQD-UHFFFAOYSA-N 0.000 claims description 2
- 235000021298 Dihomo-γ-linolenic acid Nutrition 0.000 claims description 2
- HOBAELRKJCKHQD-QNEBEIHSSA-N dihomo-γ-linolenic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCCCC(O)=O HOBAELRKJCKHQD-QNEBEIHSSA-N 0.000 claims description 2
- 150000004827 dibenzo-1,4-dioxins Chemical class 0.000 abstract 1
- 229920000064 Ethyl eicosapentaenoic acid Polymers 0.000 description 12
- SSQPWTVBQMWLSZ-AAQCHOMXSA-N ethyl (5Z,8Z,11Z,14Z,17Z)-icosapentaenoate Chemical compound CCOC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CC SSQPWTVBQMWLSZ-AAQCHOMXSA-N 0.000 description 12
- 238000005259 measurement Methods 0.000 description 11
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- 239000003063 flame retardant Substances 0.000 description 10
- 241001125048 Sardina Species 0.000 description 9
- 235000019512 sardine Nutrition 0.000 description 9
- 239000010779 crude oil Substances 0.000 description 8
- 239000002253 acid Substances 0.000 description 7
- 239000003814 drug Substances 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- 150000003071 polychlorinated biphenyls Chemical class 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 231100000770 Toxic Equivalency Factor Toxicity 0.000 description 5
- 125000004432 carbon atom Chemical group C* 0.000 description 5
- 230000032050 esterification Effects 0.000 description 5
- 238000005886 esterification reaction Methods 0.000 description 5
- 235000019441 ethanol Nutrition 0.000 description 5
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 239000012141 concentrate Substances 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- ITNKVODZACVXDS-YNUSHXQLSA-N ethyl (4Z,7Z,10Z,13Z,16Z,19Z)-docosahexaenoate Chemical compound CCOC(=O)CC\C=C/C\C=C/C\C=C/C\C=C/C\C=C/C\C=C/CC ITNKVODZACVXDS-YNUSHXQLSA-N 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 231100000419 toxicity Toxicity 0.000 description 4
- 230000001988 toxicity Effects 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- KVGZZAHHUNAVKZ-UHFFFAOYSA-N 1,4-Dioxin Chemical compound O1C=COC=C1 KVGZZAHHUNAVKZ-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 239000003054 catalyst Substances 0.000 description 3
- 239000004927 clay Substances 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 238000004817 gas chromatography Methods 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 235000014593 oils and fats Nutrition 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 238000007670 refining Methods 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 description 2
- 239000010775 animal oil Substances 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 238000011109 contamination Methods 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- TXCDCPKCNAJMEE-UHFFFAOYSA-N dibenzofuran Chemical compound C1=CC=C2C3=CC=CC=C3OC2=C1 TXCDCPKCNAJMEE-UHFFFAOYSA-N 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- JLYXXMFPNIAWKQ-GNIYUCBRSA-N gamma-hexachlorocyclohexane Chemical compound Cl[C@H]1[C@H](Cl)[C@@H](Cl)[C@@H](Cl)[C@H](Cl)[C@H]1Cl JLYXXMFPNIAWKQ-GNIYUCBRSA-N 0.000 description 2
- 125000005456 glyceride group Chemical group 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000006317 isomerization reaction Methods 0.000 description 2
- 229960002809 lindane Drugs 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 125000005575 polycyclic aromatic hydrocarbon group Chemical group 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- SQGYOTSLMSWVJD-UHFFFAOYSA-N silver(1+) nitrate Chemical compound [Ag+].[O-]N(=O)=O SQGYOTSLMSWVJD-UHFFFAOYSA-N 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 150000003626 triacylglycerols Chemical class 0.000 description 2
- -1 triglyceride fatty acid Chemical class 0.000 description 2
- HOBAELRKJCKHQD-YHTMAJSVSA-N (8e,11e,14e)-icosa-8,11,14-trienoic acid Chemical compound CCCCC\C=C\C\C=C\C\C=C\CCCCCCC(O)=O HOBAELRKJCKHQD-YHTMAJSVSA-N 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- VHNPZYZQKWIWOD-UHFFFAOYSA-N 2,2',4,4',5,6'-Hexabromodiphenyl ether Chemical compound BrC1=CC(Br)=CC(Br)=C1OC1=CC(Br)=C(Br)C=C1Br VHNPZYZQKWIWOD-UHFFFAOYSA-N 0.000 description 1
- DHUMTYRHKMCVAG-UHFFFAOYSA-N 2,4-dibromophenyl 3,4-dibromophenyl ether Chemical compound BrC1=CC(Br)=CC=C1OC1=CC=C(Br)C(Br)=C1 DHUMTYRHKMCVAG-UHFFFAOYSA-N 0.000 description 1
- ISPYQTSUDJAMAB-UHFFFAOYSA-N 2-chlorophenol Chemical compound OC1=CC=CC=C1Cl ISPYQTSUDJAMAB-UHFFFAOYSA-N 0.000 description 1
- UPNBETHEXPIWQX-UHFFFAOYSA-N 4-bromophenyl 2,4-dibromophenyl ether Chemical compound C1=CC(Br)=CC=C1OC1=CC=C(Br)C=C1Br UPNBETHEXPIWQX-UHFFFAOYSA-N 0.000 description 1
- UHPMCKVQTMMPCG-UHFFFAOYSA-N 5,8-dihydroxy-2-methoxy-6-methyl-7-(2-oxopropyl)naphthalene-1,4-dione Chemical compound CC1=C(CC(C)=O)C(O)=C2C(=O)C(OC)=CC(=O)C2=C1O UHPMCKVQTMMPCG-UHFFFAOYSA-N 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 241000228212 Aspergillus Species 0.000 description 1
- 240000002791 Brassica napus Species 0.000 description 1
- 235000004977 Brassica sinapistrum Nutrition 0.000 description 1
- 241000273930 Brevoortia tyrannus Species 0.000 description 1
- 241000252203 Clupea harengus Species 0.000 description 1
- 241000238424 Crustacea Species 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241000239366 Euphausiacea Species 0.000 description 1
- 241000223218 Fusarium Species 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 235000004431 Linum usitatissimum Nutrition 0.000 description 1
- 240000006240 Linum usitatissimum Species 0.000 description 1
- 239000004367 Lipase Substances 0.000 description 1
- 102000004882 Lipase Human genes 0.000 description 1
- 108090001060 Lipase Proteins 0.000 description 1
- JJWSNOOGIUMOEE-UHFFFAOYSA-N Monomethylmercury Chemical compound [Hg]C JJWSNOOGIUMOEE-UHFFFAOYSA-N 0.000 description 1
- 241000235575 Mortierella Species 0.000 description 1
- 241000228143 Penicillium Species 0.000 description 1
- 241000223252 Rhodotorula Species 0.000 description 1
- 241000269851 Sarda sarda Species 0.000 description 1
- CRPUJAZIXJMDBK-UHFFFAOYSA-N Toxaphene Natural products C1CC2C(=C)C(C)(C)C1C2 CRPUJAZIXJMDBK-UHFFFAOYSA-N 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 235000020661 alpha-linolenic acid Nutrition 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- 239000012159 carrier gas Substances 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 150000008280 chlorinated hydrocarbons Chemical class 0.000 description 1
- 235000012716 cod liver oil Nutrition 0.000 description 1
- 239000003026 cod liver oil Substances 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 238000004042 decolorization Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- 238000004332 deodorization Methods 0.000 description 1
- 230000001877 deodorizing effect Effects 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- 239000008157 edible vegetable oil Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000010932 ethanolysis reaction Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 235000019688 fish Nutrition 0.000 description 1
- 229940013317 fish oils Drugs 0.000 description 1
- VZCCETWTMQHEPK-QNEBEIHSSA-N gamma-linolenic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/CCCCC(O)=O VZCCETWTMQHEPK-QNEBEIHSSA-N 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 235000013402 health food Nutrition 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 235000019514 herring Nutrition 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 238000004896 high resolution mass spectrometry Methods 0.000 description 1
- 238000003987 high-resolution gas chromatography Methods 0.000 description 1
- 239000002917 insecticide Substances 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 229960004488 linolenic acid Drugs 0.000 description 1
- 235000019421 lipase Nutrition 0.000 description 1
- 235000020978 long-chain polyunsaturated fatty acids Nutrition 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- NFBOHOGPQUYFRF-UHFFFAOYSA-N oxanthrene Chemical compound C1=CC=C2OC3=CC=CC=C3OC2=C1 NFBOHOGPQUYFRF-UHFFFAOYSA-N 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 229910001961 silver nitrate Inorganic materials 0.000 description 1
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 238000001256 steam distillation Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- OEJNXTAZZBRGDN-UHFFFAOYSA-N toxaphene Chemical compound ClC1C(Cl)C2(Cl)C(CCl)(CCl)C(=C)C1(Cl)C2(Cl)Cl OEJNXTAZZBRGDN-UHFFFAOYSA-N 0.000 description 1
- PIILXFBHQILWPS-UHFFFAOYSA-N tributyltin Chemical compound CCCC[Sn](CCCC)CCCC PIILXFBHQILWPS-UHFFFAOYSA-N 0.000 description 1
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 1
- SBXWFLISHPUINY-UHFFFAOYSA-N triphenyltin Chemical compound C1=CC=CC=C1[Sn](C=1C=CC=CC=1)C1=CC=CC=C1 SBXWFLISHPUINY-UHFFFAOYSA-N 0.000 description 1
- 235000021081 unsaturated fats Nutrition 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/03—Preparation of carboxylic acid esters by reacting an ester group with a hydroxy group
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/52—Esters of acyclic unsaturated carboxylic acids having the esterified carboxyl group bound to an acyclic carbon atom
- C07C69/587—Monocarboxylic acid esters having at least two carbon-to-carbon double bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/20—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/20—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids
- A61K31/202—Carboxylic acids, e.g. valproic acid having a carboxyl group bound to a chain of seven or more carbon atoms, e.g. stearic, palmitic, arachidic acids having three or more double bonds, e.g. linolenic
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/02—Nutrients, e.g. vitamins, minerals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/32—Bonded phase chromatography
- B01D15/325—Reversed phase
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C51/00—Preparation of carboxylic acids or their salts, halides or anhydrides
- C07C51/42—Separation; Purification; Stabilisation; Use of additives
- C07C51/43—Separation; Purification; Stabilisation; Use of additives by change of the physical state, e.g. crystallisation
- C07C51/44—Separation; Purification; Stabilisation; Use of additives by change of the physical state, e.g. crystallisation by distillation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C51/00—Preparation of carboxylic acids or their salts, halides or anhydrides
- C07C51/42—Separation; Purification; Stabilisation; Use of additives
- C07C51/47—Separation; Purification; Stabilisation; Use of additives by solid-liquid treatment; by chemisorption
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C57/00—Unsaturated compounds having carboxyl groups bound to acyclic carbon atoms
- C07C57/02—Unsaturated compounds having carboxyl groups bound to acyclic carbon atoms with only carbon-to-carbon double bonds as unsaturation
- C07C57/03—Monocarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C57/00—Unsaturated compounds having carboxyl groups bound to acyclic carbon atoms
- C07C57/02—Unsaturated compounds having carboxyl groups bound to acyclic carbon atoms with only carbon-to-carbon double bonds as unsaturation
- C07C57/03—Monocarboxylic acids
- C07C57/12—Straight chain carboxylic acids containing eighteen carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/48—Separation; Purification; Stabilisation; Use of additives
- C07C67/52—Separation; Purification; Stabilisation; Use of additives by change in the physical state, e.g. crystallisation
- C07C67/54—Separation; Purification; Stabilisation; Use of additives by change in the physical state, e.g. crystallisation by distillation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/48—Separation; Purification; Stabilisation; Use of additives
- C07C67/56—Separation; Purification; Stabilisation; Use of additives by solid-liquid treatment; by chemisorption
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B3/00—Refining fats or fatty oils
- C11B3/12—Refining fats or fatty oils by distillation
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23D—EDIBLE OILS OR FATS, e.g. MARGARINES, SHORTENINGS, COOKING OILS
- A23D9/00—Other edible oils or fats, e.g. shortenings, cooking oils
- A23D9/02—Other edible oils or fats, e.g. shortenings, cooking oils characterised by the production or working-up
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/115—Fatty acids or derivatives thereof; Fats or oils
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/215—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids
- A61K31/22—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin
- A61K31/23—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin of acids having a carboxyl group bound to a chain of seven or more carbon atoms
- A61K31/232—Esters, e.g. nitroglycerine, selenocyanates of carboxylic acids of acyclic acids, e.g. pravastatin of acids having a carboxyl group bound to a chain of seven or more carbon atoms having three or more double bonds, e.g. etretinate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/56—Materials from animals other than mammals
- A61K35/60—Fish, e.g. seahorses; Fish eggs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/56—Materials from animals other than mammals
- A61K35/612—Crustaceans, e.g. crabs, lobsters, shrimps, krill or crayfish; Barnacles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D3/00—Distillation or related exchange processes in which liquids are contacted with gaseous media, e.g. stripping
- B01D3/14—Fractional distillation or use of a fractionation or rectification column
- B01D3/143—Fractional distillation or use of a fractionation or rectification column by two or more of a fractionation, separation or rectification step
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/52—Esters of acyclic unsaturated carboxylic acids having the esterified carboxyl group bound to an acyclic carbon atom
- C07C69/533—Monocarboxylic acid esters having only one carbon-to-carbon double bond
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B3/00—Refining fats or fatty oils
- C11B3/16—Refining fats or fatty oils by mechanical means
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11C—FATTY ACIDS FROM FATS, OILS OR WAXES; CANDLES; FATS, OILS OR FATTY ACIDS BY CHEMICAL MODIFICATION OF FATS, OILS, OR FATTY ACIDS OBTAINED THEREFROM
- C11C3/00—Fats, oils, or fatty acids by chemical modification of fats, oils, or fatty acids obtained therefrom
- C11C3/003—Fats, oils, or fatty acids by chemical modification of fats, oils, or fatty acids obtained therefrom by esterification of fatty acids with alcohols
Definitions
- the present invention relates to the production of highly unsaturated fatty acids or highly unsaturated fatty acid ethyl esters produced from oils and fats containing highly unsaturated fatty acids as raw materials, such as environmental pollutants contained in oils and fats, particularly dioxins, brominated flame retardants, etc. It relates to a method for reducing the amount. Furthermore, the present invention relates to foods, supplements, pharmaceuticals, cosmetics and feeds made from fats and oils prepared according to the method.
- EPA eicosapentaenoic acid, C20: 5, n-3, all-cis-5,8,11,14,17-eicosapentaenoic acid
- DHA docosahexaenoic acid, C22: 6 n-3, all-cis-4,7,10,13,16,19-docosahexaenoic acid
- EPA and DHA are known for various physiological functions and are used as ingredients for pharmaceuticals, health foods, foods, feeds, and the like.
- Various refining steps are applied to utilize EPA and / or DHA in marine oil.
- Non-Patent Document 1 describes the removal of the insecticide DDT and its metabolites from fish oil by molecular distillation.
- Non-Patent Document 2 describes that chlorinated hydrocarbons and free fatty acids can be removed from fats or oils using vacuum stripping or thin film distillation.
- Non-Patent Document 3 describes the use of physical purification and molecular distillation for the removal of undesirable substances such as free fatty acids from oil compositions.
- Patent Document 1 and Non-Patent Document 4 describe a method for reducing environmental pollutants in fats and oils: a process of adding a volatile working fluid to the mixture, and the volatile work to which the mixture has been added. A method is described that includes a process that is subjected to at least one stripping process with a fluid.
- Non-Patent Document 5 describes that highly unsaturated fatty acids are thermally decomposed when fish oil is deodorized at high temperature.
- Non-Patent Document 6 describes that dioxins, free fatty acids, and cholesterol are removed from fish oil by a short stroke distillation method and used as a raw material for ethyl ester.
- Patent No. 3905538 (WO2004 / 007654)
- the present invention relates to a method for reducing environmental pollutants contained in fats and oils, particularly dioxins and brominated flame retardants, and dioxins and brominated products, when producing ethyl esters from fats and oils containing highly unsaturated fatty acids as constituent fatty acids. It is an object to provide an ethyl ester having a low flame retardant content.
- the present invention suppresses denaturation of highly unsaturated fatty acids by performing molecular distillation or short-path distillation under certain conditions in the purification of fats and oils containing highly unsaturated fatty acids that are easily denatured, such as oxidation and isomerization, as constituent fatty acids.
- environmental pollutants can be reduced to a very low concentration.
- the amount of environmental pollutants in fats and oils can be reduced to a total toxic equivalent of less than 0.2 pg-TEQ / g.
- ethyl ester is obtained from the raw material. Dioxins in the ethyl ester can be further reduced by subjecting the ethyl ester to distillation and column chromatography.
- the gist of the present invention is the following highly unsaturated fatty acids or ethyl esters of highly unsaturated fatty acids and methods for producing the same, and feeds, foods, pharmaceuticals and the like containing them.
- a highly unsaturated fatty acid or an ethyl ester of a highly unsaturated fatty acid which is produced using a fat and oil containing a highly unsaturated fatty acid as a constituent fatty acid as a raw material oil, and has a reduced content of environmental pollutants.
- the content of polychlorinated dibenzoparadoxine (PCDD) and polychlorinated dibenzofuran (PCDF) is less than 0.05 pg-TEQ / g
- coplanar PCB (Co-PCB) is less than 0.03 pg-TEQ / g.
- Highly unsaturated fatty acids or ethyl esters of highly unsaturated fatty acids are highly unsaturated fatty acids.
- the content of brominated flame retardant contained is less than 0.18 ng / g for BDE-47, less than 0.03 ng / g for BDE-100, and less than 0.05 ng / g for BDE-49 Or (1) highly unsaturated fatty acids or ethyl esters of highly unsaturated fatty acids, wherein the amount of BDE-99 is reduced to less than 0.05 ng / g.
- the concentration of the highly unsaturated fatty acid in the fatty acid is 80 area% or more, 85 area% or more, 90 area% or more, 95 area% or more, or 96 area% or more, (1) or (2) Ethyl ester of highly unsaturated fatty acid.
- the highly unsaturated fatty acid is one of eicosapentaenoic acid, docosahexaenoic acid, docosapentaenoic acid, dihomo- ⁇ -linolenic acid, arachidonic acid, or a combination thereof (1) to (4)
- a pharmaceutical, supplement or food containing the highly unsaturated fatty acid or ethyl ester of the highly unsaturated fatty acid according to any one of (1) to (5) as an active ingredient.
- the concentration of the highly unsaturated fatty acid in the fatty acid of the highly unsaturated fatty acid or ethyl ester of the highly unsaturated fatty acid is 80 area% or more, 85 area% or more, 90 area% or more, 95 area% or more, or 96 area % Or more, the method of (7) or (8).
- a highly unsaturated fatty acid or highly unsaturated fatty acid produced by any of the methods (7) to (16) and having a dioxin content of less than 0.07 pg-TEQ / g or 0.05 pg-TEQ / g Saturated fatty acid ethyl ester.
- the content of brominated flame retardant contained is such that the amount of BDE-47 is less than 0.18 ng / g, the amount of BDE-100 is less than 0.03 ng / g, and the amount of BDE-49 is 0.05 ng / g Or a highly unsaturated fatty acid or ethyl ester of a highly unsaturated fatty acid according to (17), wherein the amount of BDE-99 is reduced to less than 0.05 ng / g.
- the method of the present invention reduces environmental pollutants, especially dioxins, contained in fish oil to a very low level without affecting the content of highly unsaturated fatty acids in fatty acids by distillation at high temperature, high vacuum, and short time. Therefore, it is possible to provide various products such as feeds, foods, supplements, and pharmaceuticals made from fish oil and the like that do not need to worry about contamination by dioxins.
- the highly unsaturated fatty acid is a fatty acid having 18 or more carbon atoms and 3 or more double bonds, more preferably a fatty acid having 20 or more carbon atoms, 3 or more or 4 or more double bonds, particularly preferably. It is a fatty acid having 20 or more carbon atoms and 5 or more double bonds.
- ⁇ -linolenic acid (18: 3, n-3), ⁇ -linolenic acid (18: 3, n-6), dihomo- ⁇ -linolenic acid (20: 3, n-6), arachidonic acid (20: 4, n-6), eicosapentaenoic acid (20: 5, n-3), docosapentaenoic acid (22: 5, n-6), docosahexaenoic acid (22: 6, n-3), etc. Illustrated. These are known to be contained in a large amount in certain microbial oils, vegetable oils, marine animal oils, and the like.
- fish oil such as sardine oil, tuna oil, bonito oil, menhaden oil, cod liver oil, herring oil, caperin oil, and crustacean marine animal oil such as krill, egoma, flax, soybean, rapeseed And oils produced by microorganisms belonging to the genus Mortierella, the genus Penicillium, the genus Aspergillus, the genus Rhodotorula and the genus Fusarium.
- the method of the present invention is particularly suitable for oils and fats derived from marine products where contamination by dioxins is a concern, such as fish oil, krill oil, or marine mammal oil.
- an oil containing a highly unsaturated fatty acid as a constituent fatty acid means triglyceride or phospholipid.
- these fats and oils may be pretreated before being subjected to molecular distillation or short path distillation. Examples of such pretreatment include a degumming step, a decolorization step using activated clay and activated carbon, and a water washing step.
- environmental pollutants include polychlorinated biphenyl (PCB), DDT, polychlorinated triphenyl (PCT), dibenzo-dioxin (PCDD) and dibenzo-furan (PCDF), chlorophenol and hexachlorocyclohexane (HCH). ), Toxaphene, dioxins, brominated flame retardants, polyaromatic hydrocarbons (PAH), organotin compounds (eg tributyltin, triphenyltin), organomercury compounds (eg methylmercury) and the like.
- PCB polychlorinated biphenyl
- PCT polychlorinated triphenyl
- PCDD dibenzo-dioxin
- PCDF dibenzo-furan
- HH hexachlorocyclohexane
- Toxaphene dioxins
- brominated flame retardants polyaromatic hydrocarbons (PAH)
- organotin compounds eg tribu
- dioxins mean the total of polychlorinated dibenzopararadixin (PCDD), polychlorinated dibenzofuran (PCDF) and coplanar PCB (Co-PCB) shown in Table 1, and the content of each component is measured.
- PCDD polychlorinated dibenzopararadixin
- PCDF polychlorinated dibenzofuran
- Co-PCB coplanar PCB
- the toxicity equivalents (pg-TEQ / g) calculated by multiplying the respective measured values by the toxicity equivalents were expressed.
- brominated flame retardant is a general term for compounds as shown in Table 9.
- BDE-100, BDE-49, BDE-99, BDE-47, etc. which are relatively abundant in fish oil, can be used as indicators.
- dioxins are removed by thin film distillation.
- molecular distillation or short path distillation (SPD) is preferable.
- SPD short path distillation
- thin-film distillations distillation performed by placing a condenser within a distance shorter than the mean free path of vapor molecules evaporated from the heating surface at a constant pressure under high vacuum ( ⁇ 0.1 Pa) is called molecular distillation.
- Short stroke distillation was developed to increase the distillation capability of molecular distillation.
- Short-path distillation is performed at a pressure in the middle vacuum range higher than 0.1 Pa, and the condenser is placed at the same distance as the mean free path of the evaporated molecules.
- Method Molecular distillation or short path distillation is suitable for removing unnecessary components from triglycerides containing EPA, DHA, etc., which are sensitive to heat, because the distillate is exposed to a high temperature for a very short time.
- the molecular or short path distillation of the present invention is carried out at a temperature of 200 to 270 ° C., preferably 220 to 260 ° C., particularly preferably 220 to 250 ° C.
- the pressure is 5 Pa, preferably 2 Pa, more preferably 1 Pa.
- the flow rate is 20 to 200 (kg / h) / m 2 , preferably 25 to 120 (kg / h) / m 2 . If the flow rate is too small, the productivity is lowered. Therefore, it is preferable to flow the maximum amount within a range where removal is possible while confirming that dioxins are removed. If distillation is performed under such conditions, although it is a highly unsaturated fatty acid that is weak in temperature, quality degradation hardly occurs.
- PCDD and PCDF could be less than the measurement limit, that is, substantially zero.
- 0 is, for example, less than 0.043 pg-TEQ / g when calculated from the numerical value of the detection limit in Table 6, that is, less than 0.05 pg-TEQ / g.
- Coplanar PCBs can also be reduced to less than 0.2 pg-TEQ / g, further less than 0.1 pg-TEQ / g, less than 0.05 pg-TEQ / g, less than 0.02 pg-TEQ / g, and less than 0.01 pg-TEQ / g.
- brominated flame retardants when BDE-100, BDE-49, BDE-99, BDE-47, etc. are used as indicators, these are less than 0.05 ⁇ g / g, preferably less than 0.03 ⁇ g / g, and further 0.02 ⁇ g / g. Can be reduced to less than
- Embodiments of the method of the present invention are described below. It is preferable to use a raw material oil that has been degummed by a method such as washing with water.
- the raw oil washed with water is subjected to molecular distillation or short-path distillation as it is under the above-mentioned conditions to remove cholesterol, free fatty acids, environmental pollutants and the like as fractions to obtain a residue containing triglycerides.
- This residue may be used as it is, or may be used after decoloring treatment with activated carbon, activated clay, etc., or deodorizing treatment with steam distillation or the like.
- the refined fats and oils thus produced can be used as ingredients for feeds, foods and supplements.
- an ethyl ester with a reduced content of dioxins can be produced using the above residue as a raw material.
- Ethyl alcohol and a catalyst or enzyme are added to the residue and reacted to form a triglyceride fatty acid and an ester of ethyl alcohol.
- Any known method may be used for ethyl esterification. After the ethyl esterification, further purification may be performed as necessary.
- methods such as molecular distillation, rectification, and column chromatography can be used.
- JP-A-5-222392 family patent EP0610506
- JP-A-4-41457 family patent EP0460917
- JP-A-6-33088 JP-A-6-33088 and the like.
- the rectification is carried out in a high-vacuum with three or more distillation columns, and is obtained by separating EPA ethyl ester and / or DHA ethyl ester as the main distillate from the more volatile initial distillate and the less volatile residue. be able to.
- the temperature is 150 to 200 ° C., preferably 160 to 190 ° C., more preferably 170 to 190 ° C.
- the pressure is 1 to 300 Pa, preferably 1 to 200 Pa, more preferably 1 to 133 Pa. is there. It is preferable to obtain a main fraction of 160 to 190 ° C., preferably 170 to 190 ° C. with a vacuum of 1 to 133 Pa.
- Particularly preferred is a method of performing column chromatography after rectification. The inventors of the present application have found that when EPA and DHA are concentrated by rectification, the concentration of dioxins is relatively increased, but by performing column chromatography, the level can be lowered from the level before rectification. .
- silica gel, ion exchange resin, activated clay, silver nitrate, and the like can be used, but column chromatography with a reverse phase distribution system is particularly preferable.
- an alkyl group-bonded silica filler (ODS column or the like) or the like can be used so as to constitute a reverse phase distribution system, and water, alcohol, or ketones can be used as the solvent system. Methanol is preferred. These may be used alone or in combination.
- PCDD and PCDF can be below the measurement limit, that is, substantially zero (less than 0.043 pg-TEQ / g when calculated from the detection limit), and coplanar PCB is also less than 0.1 pg-TEQ / g Furthermore, it can be reduced to less than 0.03 pg-TEQ / g and less than 0.01 pg-TEQ / g.
- the amount of BDE-47 is less than 0.18 ng / g, the amount of BDE-100 is less than 0.03 ng / g, the amount of BDE-49 is less than 0.05 ng / g, or BDE-99
- the amount can be reduced to less than 0.05 ng / g.
- BDE-100, BDE-49, BDE-99, and BDE-47 can be reduced to less than 0.05 ⁇ g / g, preferably less than 0.03 ⁇ g / g, and even less than 0.02 ⁇ g / g.
- the purity of the EPA ethyl ester and / or DHA ethyl ester concentration of 96 area% or more is preferable.
- Such free fatty acids and fatty acid esters having a high concentration of highly unsaturated fatty acids and a low concentration of environmental pollutants are suitable for pharmaceuticals and supplement raw materials containing highly unsaturated fatty acids as active ingredients.
- the highly unsaturated fatty acid in the glyceride can be concentrated by a method (such as WO2009 / 17102) that selectively concentrates the highly unsaturated fatty acid by lipase reaction.
- a method such as WO2009 / 17102
- the highly unsaturated fatty acid can be obtained by hydrolyzing the ester of the highly unsaturated fatty acid produced by the above method.
- Measurement of Dioxins and Brominated Flame Retardant In the examples of the present invention, the measurement of dioxins was requested to the Japan Food Analysis Center. The measuring method is based on “Provisional Guidelines for Measuring Dioxins in Food (February 2008)” (1999 Hygien No. 138, Hygien No. 200). Measurement of brominated flame retardants was requested from eurofins, an analysis company for foods. The measuring method is based on high resolution mass spectrometry (HRGC / HRMS method).
- the acid value (AV) was measured according to the standard method for analyzing fats and oils (2003 version) (edited by the Japan Oil Chemists' Society).
- Fatty acid composition measurement Fish oil used as a raw material and the fatty acid composition of the oil after processing by short-path distillation were measured by gas chromatography after the fish oil was ethyl esterified. That is, 1 mL of 1N sodium ethylate / ethanol solution was added to 40 ⁇ L of fish oil and stirred for about 30 seconds. Thereafter, 1 mL of 1N hydrochloric acid was added for neutralization, 2 mL of hexane and 3 mL of saturated aqueous ammonium sulfate solution were added, and after stirring and standing, the upper layer was measured by gas chromatography.
- the distillation apparatus used is a short path distillation apparatus (SPD) KD 10 (manufactured by UIC GmbH, distillation surface area of 0.1 m 2 ).
- the distillation conditions (temperature, pressure, flow rate) are shown in Table 2.
- the distillation temperature was fixed at 250 ° C., the pressure was varied from 0.4 to 3.0 Pa, and the flow rate was varied from 25 to 121 (kg / hr) / m 2 .
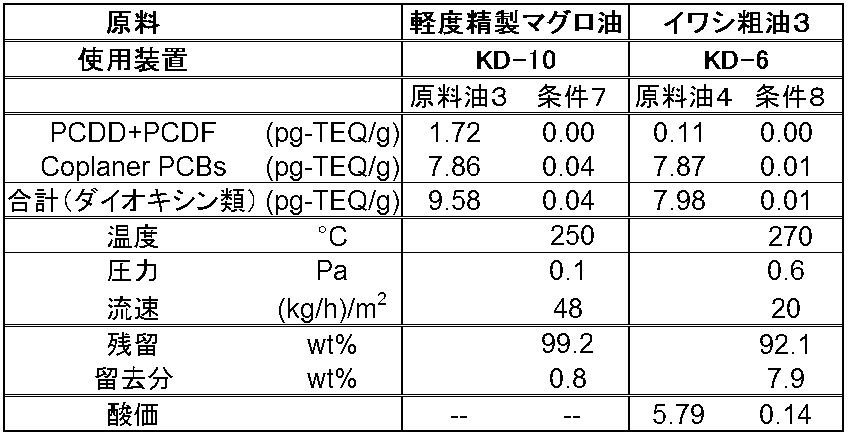
- Table 2 shows the raw oil and dioxin concentrations after distillation and the acid value. Regardless of the acid value of the feedstock and the amount of dioxins in the feedstock, the dioxins content can be reduced to 0.1 pg-TEQ / g or less under any conditions. It was.
- dioxins were removed under the distillation conditions shown in Table 3 for tuna oil (refined oil subjected to degumming and deoxidation treatment). As shown in Table 3, even tuna oil has a dioxin content of 0.1 pg-TEQ / or less by the method of the present invention (distillation temperature is 250 ° C., pressure is 0.1 Pa, flow rate is 48 (kg / hr) / m 2 ). It was possible to reduce. Further, dioxins were removed from sardine crude oil using another short path distillation apparatus (SPD) KD 6 (manufactured by UIC GmbH, distillation surface area 0.06 m 2 ).
- SPD short path distillation apparatus
- Dioxins were removed from the sardine crude oil under the distillation conditions shown in Table 4 using a centrifugal molecular distillation apparatus MS380 (manufactured by Nippon Vehicle Manufacturing Co., Ltd., distillation surface area 0.11 m 2 ). As shown in Table 4, even when a molecular distillation apparatus was used, the dioxins content could be reduced to 0.2 pg-TEQ / or less as in Examples 1 and 2.
- SPD short-path distillation device KD1800 (made by UIC GmbH, distillation surface 18 m 2 ) was used to perform short path distillation.
- the conditions were a degree of vacuum of 0.7 to 1 Pa, a body temperature of 250 ° C., and a feed rate of about 2000 kg / H (flow rate 110 (kg / h) / m 2 ).
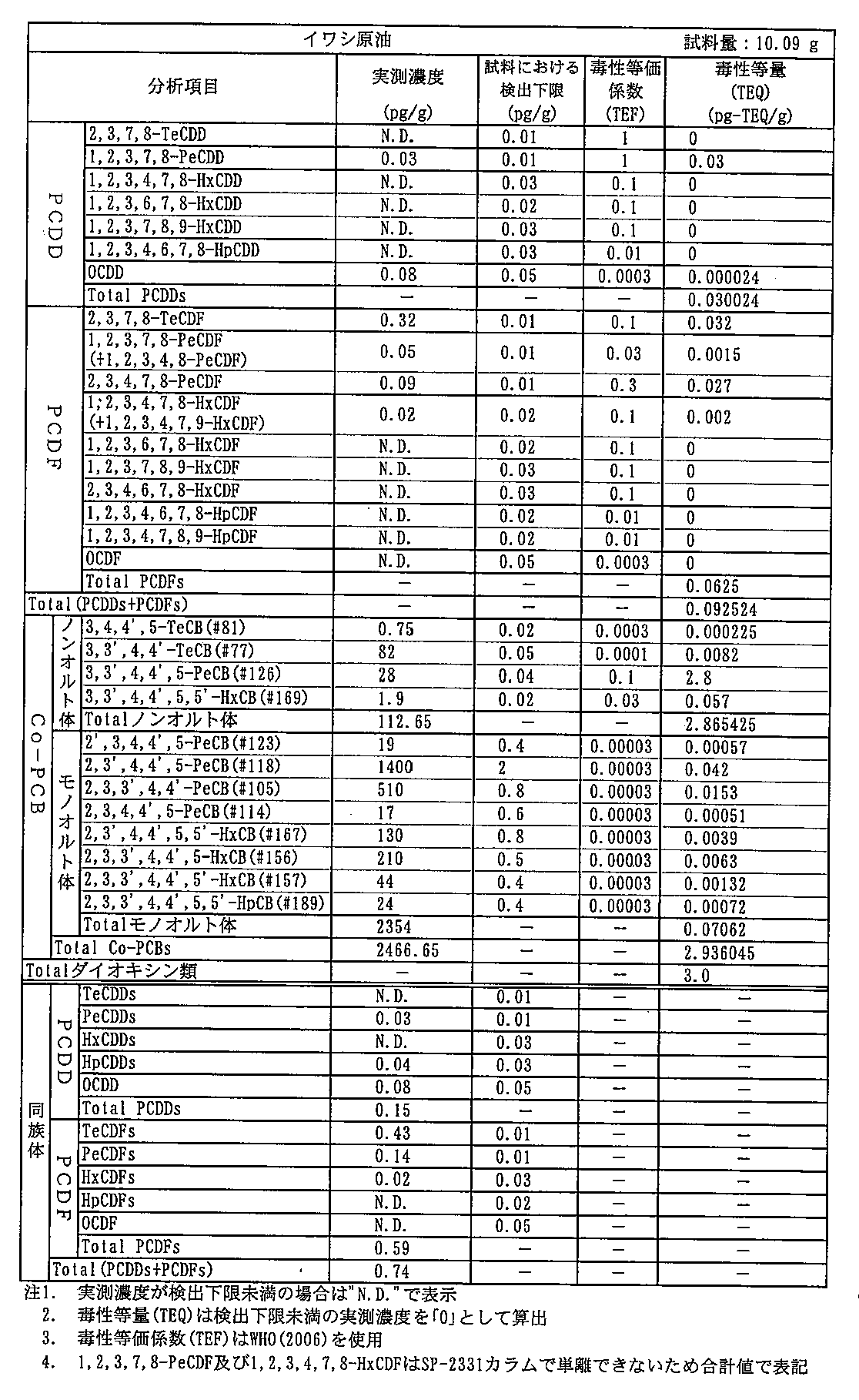
- Tables 5 and 6 show the measurement results of dioxins in crude oil and oil after distillation (SPD oil), respectively.
- the total amount of dioxins was reduced from 3.0 pg-TEQ / g to 0.014 pg-TEQ / g in terms of toxicity equivalent. Further, the acid value of the oil after distillation was 0.2 or less, and the fatty acid composition was not changed.
- EPA ethyl ester was produced using the oil after distillation produced in Example 4 as a raw material.
- the oil distilled by SPD was subjected to an ethanolysis reaction with ethyl alcohol in the presence of an alkali catalyst to obtain an ethyl ester.
- Table 8 shows the results of measuring dioxins of this ethyl ester in the same manner as in Example 4.
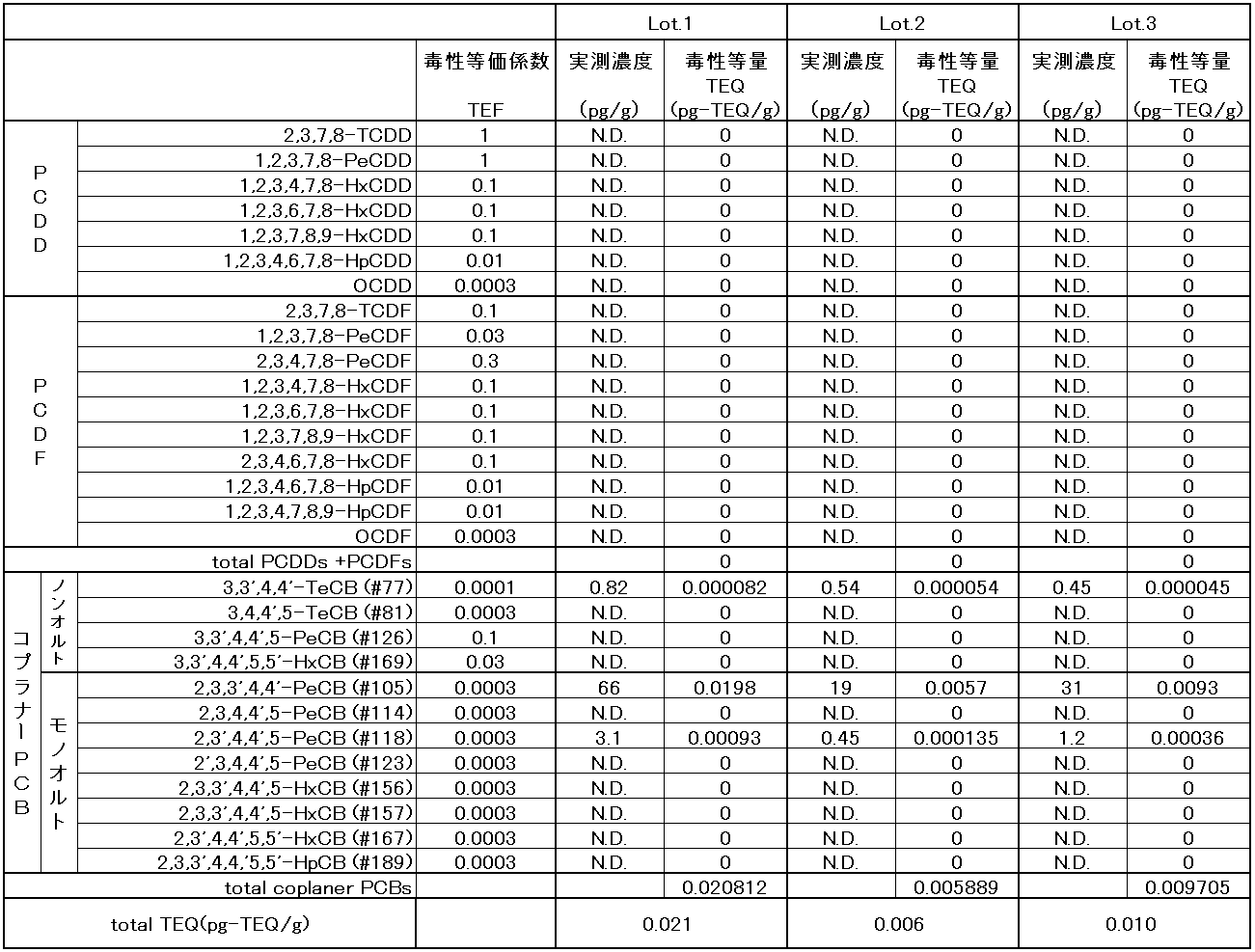
- Dioxins are not concentrated in the ethyl esterification and subsequent purification steps, and by using the raw material treated in Example 4, the total amount of dioxins is 0.07 pg-TEQ / g or less in terms of toxic equivalents.
- Table 8 shows the results of using three different lots of raw materials. It was confirmed that by the method of the present invention, 0.006 to 0.021 pg-TEQ / g of EPA ethyl ester can be produced stably with a total amount of dioxins equivalent to toxicity. Even if ND below the measurement limit is calculated by inserting the value of the measurement limit, it only increases by 0.005, which means that 0.01 to 0.026 pg-TEQ / g EPA ethyl ester can be produced. .
- the effect of rectification and column chromatography in the purification step after ethyl ester on the concentration of dioxins was confirmed.
- Sardine crude oil that had been deacidified and decolorized and not thin-film distilled was used as the raw material oil. This is because it is easier to observe the effect of the refining treatment when using fats and oils containing a little more dioxins.
- the feedstock was ethyl esterified with an alkali catalyst.
- the ethyl ester was first subjected to a rectification step, and a fraction containing ethyl ester of a fatty acid having 20 carbon atoms was collected. Subsequently, a fraction of eicosapentaenoic acid ethyl ester was collected by ODS column chromatography. Dioxins were measured at each stage.
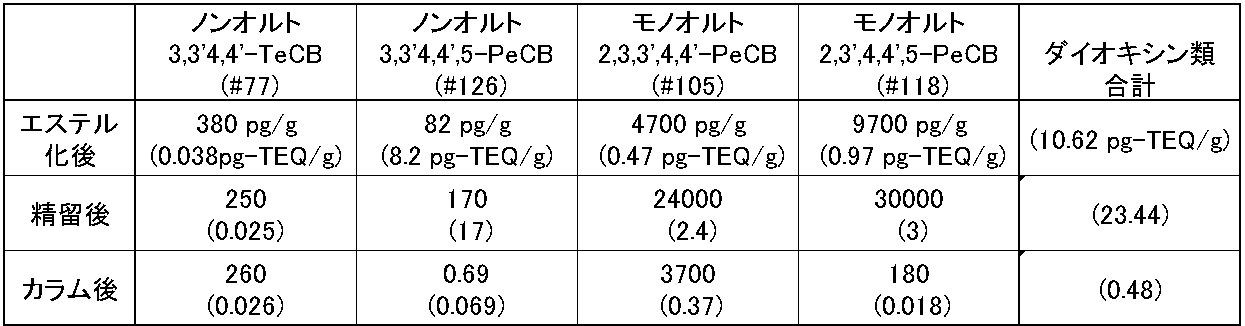
- Table 9 shows the total of 4 components (absolute amount and toxic equivalent amount) and dioxins (toxic equivalent amount) with large fluctuations. Compared with the esterification, the components # 105 and # 118 were greatly concentrated by the rectification process. On the other hand, it declined rather in # 77. In rectification, eicosapentaenoic acid ethyl ester and the like and dioxins behave in a similar manner, and it is considered that dioxins are concentrated with the concentration of eicosapentaenoic acid ethyl ester and the like. As a result of column processing, # 77 remained unchanged or increased slightly, while other components decreased greatly. In the column treatment, it is considered that ethyl ester and dioxins showed different behavior and could be separated. Therefore, by combining rectification and column treatment, it is possible to simultaneously concentrate the target ethyl ester and reduce dioxins.
- fats and oils containing highly unsaturated fatty acids as constituent fatty acids such as fish oil containing EPA and DHA, particularly dioxins and brominated flame retardants can be greatly reduced.
- Such fats and oils can be provided. It can be used for feed, food, supplements, pharmaceuticals, and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Epidemiology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Crystallography & Structural Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Food Science & Technology (AREA)
- Polymers & Plastics (AREA)
- Nutrition Science (AREA)
- Emergency Medicine (AREA)
- Mycology (AREA)
- Wood Science & Technology (AREA)
- Microbiology (AREA)
- Analytical Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Diabetes (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Coloring Foods And Improving Nutritive Qualities (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Fats And Perfumes (AREA)
- Edible Oils And Fats (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Cell Biology (AREA)
- Developmental Biology & Embryology (AREA)
- Immunology (AREA)
Abstract
Description
特許文献1、非特許文献4には、油脂中の環境汚染物質を低減させるための方法であって:揮発性作業流体を該混合物に添加する過程、および該混合物が添加された該揮発性作業流体とともに少なくとも1回のストリッピング処理過程に付される過程を含む方法が記載されている。
非特許文献5には、魚油を高温で脱臭すると高度不飽和脂肪酸が熱分解することが記載されている。
非特許文献6には、短行程蒸留方式で魚油からダイオキシンや遊離脂肪酸、コレステロールを除去し、エチルエステルの原料とすることが記載されている。
本発明は、下記の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル及びその製造方法、及びそれらを含有する飼料、食品、医薬品等を要旨とする。
(2)さらに、含まれる臭素化難燃剤の含有量がBDE-47の量が0.18ng/g未満、BDE-100の量が0.03ng/g未満、BDE-49の量が0.05ng/g未満、又は、BDE-99の量が0.05ng/g未満に低減されている(1)の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
(3)脂肪酸中に占める高度不飽和脂肪酸の濃度が80面積%以上、85面積%以上、90面積%以上、95面積%以上、又は96面積%以上である、(1)又は(2)の高度不飽和脂肪酸のエチルエステル。
(4)高度不飽和脂肪酸を構成脂肪酸として含有する油脂が魚油、オキアミ油、海洋哺乳類油又は微生物油である(1)ないし(3)いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
(5)高度不飽和脂肪酸がエイコサペンタエン酸、ドコサヘキサエン酸、ドコサペンタエン酸、ジホモ-γ-リノレン酸、アラキドン酸のいずれか、又はこれらの組み合わせである(1)ないし(4)いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
(6)(1)ないし(5)いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルを有効成分として含有する医薬品、サプリメント又は食品。
b) 得られた高度不飽和脂肪酸含有油脂を加水分解又はエチルエステル化し、
c) 精留及びカラムクロマトグラフィにより精製することにより、
ポリ塩化ジベンゾパラジオキシン(PCDD)、ポリ塩化ジベンゾフラン(PCDF)及びコプラナーPCB(Co-PCB)の含有量を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルを製造する方法。
(8)さらに、臭素化難燃剤の含有量も低下させる(7)の方法。
(9)高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルの脂肪酸中に占める高度不飽和脂肪酸の濃度が80面積%以上、85面積%以上、90面積%以上、95面積%以上、又は96面積%以上である、(7)又は(8)の方法。
(10)高度不飽和脂肪酸を構成脂肪酸として含有する原料油が魚油、オキアミ油、海洋哺乳類油又は微生物油である(7)ないし(9)いずれかの方法。
(11)前記薄膜蒸留が、200~270℃、220~260℃、又は220~250℃の温度で実行される、(7)ないし(10)いずれかの方法。
(12)前記薄膜蒸留が、5Pa以下、2Pa以下、又は1Pa以下の圧力で実行される、(7)ないし(11)いずれかの方法。
(13)前記薄膜蒸留が、流速20~200(kg/h)/m2、又は25~120(kg/h)/m2で実行される、(7)ないし(12)いずれかの方法。
(14)前期薄膜蒸留が分子蒸留又は短行程蒸留である(7)ないし(13)いずれかの方法。
(15)精留が、3塔以上の蒸留塔にて行うものである(7)ないし(14)のいずれかの方法。
(16)カラムクロマトグラフィによる精製が、逆相分配系のカラムクロマトグラフィを用いるものである(7)ないし(15)のいずれかの方法。
(18)さらに、含まれる臭素化難燃剤の含有量が、BDE-47の量が0.18ng/g未満、BDE-100の量が0.03ng/g未満、BDE-49の量が0.05ng/g未満、又は、BDE-99の量が0.05ng/g未満に低減されている(17)の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
(19)脂肪酸中に占める高度不飽和脂肪酸の濃度が80面積%以上、85面積%以上、90面積%以上、95面積%以上、又は96面積%以上である(17)又は(18)の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
(20)(21)ないし(23)いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルを含有する医薬品、サプリメント、又は食品。
本発明において、高度不飽和脂肪酸とは、炭素数18以上、二重結合数3以上の脂肪酸であり、より好ましくは炭素数20以上、二重結合数3以上又は4以上の脂肪酸、特に好ましくは炭素数20以上、二重結合数5以上の脂肪酸である。具体的にはα‐リノレン酸(18:3,n‐3)、γ‐リノレン酸(18:3,n‐6)、ジホモ-γ-リノレン酸(20:3、n-6)、アラキドン酸(20:4,n‐6)、エイコサペンタエン酸(20:5,n‐3)、ドコサペンタエン酸(22:5,n‐6)、ドコサヘキサエン酸(22:6,n‐3)などが例示される。
これらはある種の微生物油、植物油や海産動物油などに多く含まれることが知られている。具体的には、イワシ油、マグロ油、カツオ油、メンヘイデン油、タラ肝油、ニシン油、カペリン油、およびサーモン油などの魚油やオキアミなどの甲殻類の海産動物油、エゴマ、アマ、大豆、菜種などの植物油、モルティエレラ(Mortierella)属、ペニシリューム(Penicillium)属、アスペルギルス(Aspergillus)属、ロードトルラ(Rhodotorula)属、フザリューム(Fusarium)属に属する微生物が産生する油脂などが例示される。
本発明の方法は、特にダイオキシン類による汚染が懸念される海産物由来の油脂、例えば、魚油、オキアミ油、又は海洋哺乳類油に適する。
本明細書において高度不飽和脂肪酸を構成脂肪酸として含有する油脂とはトリグリセリド、又はリン脂質を意味する。
これらの油脂を本発明の原料油として用いる場合、分子蒸留又は短行程蒸留に付す前に、前処理をしてもよい。このような前処理としては、脱ガム工程、活性白土や活性炭を用いた脱色工程、水洗工程などが例示される。
本発明においてダイオキシン類とは、表1に示した、ポリ塩化ジベンゾパラジオキシン(PCDD)、ポリ塩化ジベンゾフラン(PCDF)及びコプラナーPCB(Co-PCB)の合計を意味し、各成分の含有量を測定し、それぞれの実測値に毒性等量をかけて合計したて算出した毒性等量(pg-TEQ/g)で表した。
また、臭素化難燃剤についても測定した。臭素化難燃剤とは、表9に示すような化合物の総称である。環境中のダイオキシン類の汚染が低下し始めているのに対し、臭素化難燃剤についてはまだ増加傾向が見られており、注目すべき物質である。魚油中に比較的多く含まれるBDE-100、BDE-49、BDE-99、BDE-47などを指標とすることができる。
分子蒸留又は短行程蒸留は、被蒸留物が高温にさらされる時間が非常に短いので、熱に弱いEPA,DHA等を含有するトリグリセリドから不要成分を除去するのに適している。
このような条件で蒸留することにより、ダイオキシン類のうち、PCDD及びPCDFは測定限界未満、すなわち実質的に0にすることができた。ここで0とは例えば、表6検出限界の数値から計算すると0.043pg-TEQ/g未満であり、すなわち、0.05pg-TEQ/g未満にすることができる。コプラナーPCBも0.2pg-TEQ/g未満、さらに0.1 pg-TEQ/g未満、0.05 pg-TEQ/g未満、0.02 pg-TEQ/g未満、0.01pg-TEQ/g未満まで低下させることができる。
また、臭素化難燃剤では、BDE-100、BDE-49、BDE-99、BDE-47などを指標とすると、これらを0.05μg/g未満、好ましくは0.03μg/g未満、さらに0.02μg/g未満に低下させることができる。
原料油は水洗などの方法で脱ガム処理を施したものを用いるのが好ましい。水洗した原料油をそのまま上述の条件で分子蒸留又は短行程蒸留に付し、コレステロール、遊離脂肪酸、環境汚染物質などを留分として除去し、トリグリセリドを含む残留分を得る。この残留分はそのまま用いてもよいし、さらに活性炭、活性白土などによる脱色処理、水蒸気蒸留などによる脱臭処理をして用いることもできる。こうして製造した精製油脂は飼料、食品、サプリメントの成分として用いることができる。
残留分にエチルアルコールと触媒又は酵素を加え、反応させ、トリグリセリドの構成脂肪酸とエチルアルコールのエステルを生成させる。エチルエステル化は公知のどのような方法を用いてもかまわない。
エチルエステル化した後、必要に応じてさらに精製してもよい。EPAエチルエステルやDHAエチルエステルの純度を高めるためには、さらに分子蒸留、精留、カラムクロマトグラフィなどの方法を用いることができる。具体的には、特開平5-222392(ファミリーパテントEP0610506)、特開平4-41457(ファミリーパテントEP0460917)、特開平6-33088等に記載の方法で精製することができる。
精留は高真空下で3塔以上の蒸留塔により行い、EPAエチルエステル及び/又はDHAエチルエステルを主留として、より揮発性の高い初留及びより揮発性の低い残留と分離することにより得ることができる。精留の条件としては、温度は150~200℃、好ましくは160~190℃、さらに好ましくは170~190℃であり、圧力は1~300Pa、好ましくは1~200Pa、さらに好ましくは1~133Paである。1~133Paの真空度で、160~190℃、好ましくは170~190℃の主留を得るのが好ましい。
特に好ましいのは、精留後にカラムクロマトグラフィを行う方法である。本願発明者らは、精留によりEPAやDHAを濃縮すると、相対的にダイオキシン類の濃度が上昇するが、カラムクロマトグラフィを行うことにより、精留前のレベルよりも低下させることができることを見出した。カラムクロマトグラフィにはシリカゲル、イオン交換樹脂、活性白土、硝酸銀などを用いることができるが、逆相分配系のカラムクロマトグラフィが特に好ましい。逆相分配系を構成するように、たとえばアルキル基結合シリカ充填剤(ODSカラム等)等を用い、また、溶媒系としては、水、アルコール、ケトン類を用いることができる。好ましくはメタノールである。これらは単独、もしくは混合してもちいてもよい。
上述の精留、カラムクロマトグラフィを組み合わせることにより、高度不飽和脂肪酸を濃縮する一方で環境汚染物質の濃度を低下させることができる。高度不飽和脂肪酸、例えば、EPAエチルエステル及び/又はDHAエチルエステルの濃度を80面積%以上、85面積%以上、90面積%以上、95面積%以上さらに96面積%以上の純度に高め、かつ、ダイオキシン類のうち、PCDD及びPCDFは測定限界以下、すなわち、実質的に0(検出限界からの計算では0.043pg-TEQ/g未満)にすることができ、コプラナーPCBも0.1pg-TEQ/g未満、さらに0.03 pg-TEQ/g未満、0.01 pg-TEQ/g未満まで低下させることができる。あるいは、臭素化難燃剤では、BDE-47の量で0.18ng/g未満、BDE-100の量で0.03ng/g未満、BDE-49の量で0.05ng/g未満、又は、BDE-99の量で0.05ng/g未満に低下させることができる。BDE-100、BDE-49、BDE-99、BDE-47を0.05μg/g未満、好ましくは0.03μg/g未満、さらに0.02μg/g未満に低下させることができる。医薬品としては、EPAエチルエステル及び/又はDHAエチルエステルの濃度が96面積%以上の純度が好ましい。
これらのような高度不飽和脂肪酸の濃度が高く、環境汚染物質の濃度が低い遊離脂肪酸や脂肪酸エステルは、高度不飽和脂肪酸を有効成分とする医薬品やサプリメント原料に適する。
高度不飽和脂肪酸は上記の方法で製造した高度不飽和脂肪酸のエステルを加水分解して得ることができる。
ダイオキシン類、臭素化難燃剤の測定
本発明の実施例において、ダイオキシン類の測定は財団法人日本食品分析センターに依頼した。測定方法は、「食品のダイオキシン類の測定方法暫定ガイドライン(平成20年2月)」(平成11年衛食第138号、衛乳第200号)による。
臭素化難燃剤の測定は、食品等の分析会社であるeurofinsに依頼した。測定方法は高分解能質量分析法(HRGC/HRMS法)による。
本発明の実施例において、酸価(AV)の測定は基準油脂分析試験法(2003年度版)(社団法人日本油化学会編)に準じて行った。
原料に用いた魚油及び短行程蒸留による処理をした後の油の脂肪酸組成は、魚油をエチルエステル化してガスクロマトグラフィーにて測定した。すなわち、魚油40μLに1Nナトリウムエチラート/エタノール溶液1mLを加え、約30秒間撹拌した。その後、1N塩酸を1mL加えて中和し、ヘキサン2mL、飽和硫酸アンモニア水溶液3mLを加え、撹拌、静置後、上層をガスクロマトグラフィーにて測定した。
ガスクロマトグラフィー分析条件
機種;Agilent 6850 GC system (Agilent社)
カラム;DB-WAX J&W 123-7032E
カラム温度;200℃
注入温度 ;300℃
注入方法 ;スプリット
スプリット比;100:1
検出器温度:300℃
検出器:FID
キャリアーガス:ヘリウム
原料油と蒸留後のダイオキシン類濃度と酸価を表2に示した。
原料油の酸価の高低にかかわらず、また、原料油のダイオキシン類の含有量の多寡にかかわらず、いずれの条件でもダイオキシン類の含有量を0.1pg-TEQ/g以下に低下させることができた。
また、別の短行程蒸留装置(SPD) KD 6 (UIC GmbH社製、蒸留表面積0.06 m2)を用いてイワシ粗油からダイオキシン類の除去を行った。表3に示すとおり、この装置でも本発明の方法(蒸留温度を270℃、圧力0.6Pa、流速20(kg/hr)/m2)により、ダイオキシン類の含有量を0.1pg-TEQ/以下に低下させることができた。
原油と蒸留後(SPD油)の油中のダイオキシン類の測定結果をそれぞれ表5、6に示す。ダイオキシン類の総量を毒性等量で3.0pg-TEQ/gから0.014 pg-TEQ/gに低下させることができた。また、蒸留後の油の酸価は0.2以下であり、脂肪酸組成に変化はなかった。
製造方法は、SPDにより蒸留処理した油をアルカリ触媒下エチルアルコールとエタノリシス反応を行い、エチルエステルにした。温水で水洗した後、脱水し、精留(真空度13Pa、主留の温度約176℃)により主留を得、さらに逆相分配系(ODS)のカラムクロマトグラフィーを用いたHPLC処理し、溶剤を留去し、EPA純度97%のEPAエチルエステルを製造した。
このエチルエステルのダイオキシン類を実施例4と同様に測定した結果を表8に示す。エチルエステル化及びその後の精製行程でダイオキシン類が濃縮することはなく、実施例4の処理をした原料を用いることでダイオキシン類の総量が毒性等量で0.07pg-TEQ/g以下のEPAエチルエステルを製造することができた。表8は3つの異なるロットの原料を用いて実施した結果を示したものである。本発明の方法により、安定してダイオキシン類の総量が毒性等量で0.006~0.021pg-TEQ/gのEPAエチルエステルを製造することができることを確認した。測定限界以下のNDについて、測定限界の数値を挿入して計算しても、値として0.005上昇するだけであり、0.011~0.026 pg-TEQ/gのEPAエチルエステルを製造することができることを意味する。
原料油をアルカリ触媒によりエチルエステル化した。エチルエステルをまず精留工程にかけ、炭素数20の脂肪酸のエチルエステルを含む留分を採取した。続いて、ODSのカラムクロマトグラフィにより、エイコサペンタエン酸エチルエステルの画分を採取した。
それぞれの段階で、ダイオキシン類を測定した。
カラム処理により、#77は不変、あるいは、やや増加しているのに対し、他の成分は大きく低下した。カラム処理においては、エチルエステルとダイオキシン類が異なる挙動を示して、分離できたものと考えられる。
したがって、精留とカラム処理を組み合わせることによって、目的のエチルエステルを濃縮することとダイオキシン類を低減させることを同時に達成することができる。
Claims (20)
- 高度不飽和脂肪酸を構成脂肪酸として含有する油脂を原料油として製造され、環境汚染物質の含有量が低減された、高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルであって、含まれるダイオキシン類のうち、ポリ塩化ジベンゾパラジオキシン(PCDD)及びポリ塩化ジベンゾフラン(PCDF)の含有量が0.05pg-TEQ/g未満、コプラナーPCB(Co-PCB)が0.03pg-TEQ/g未満である、高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- さらに、含まれる臭素化難燃剤の含有量がBDE-47の量が0.18ng/g未満、BDE-100の量が0.03ng/g未満、BDE-49の量が0.05ng/g未満、又は、BDE-99の量が0.05ng/g未満に低減されている請求項1の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- 脂肪酸中に占める高度不飽和脂肪酸の濃度が80面積%以上、85面積%以上、90面積%以上、95面積%以上、又は96面積%以上である、請求項1又は2の高度不飽和脂肪酸のエチルエステル。
- 高度不飽和脂肪酸を構成脂肪酸として含有する油脂が魚油、オキアミ油、海洋哺乳類油又は微生物油である請求項1ないし3いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- 高度不飽和脂肪酸がエイコサペンタエン酸、ドコサヘキサエン酸、ドコサペンタエン酸、ジホモ-γ-リノレン酸、アラキドン酸のいずれか、又はこれらの組み合わせである請求項1ないし4いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- 請求項1ないし5いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルを有効成分として含有する医薬品、サプリメント又は食品。
- a) 高度不飽和脂肪酸を構成脂肪酸として含有する原料油から、薄膜蒸留により遊離脂肪酸及び環境汚染物質を除去し、
b) 得られた高度不飽和脂肪酸含有油脂を加水分解又はエチルエステル化し、
c) 精留及びカラムクロマトグラフィにより精製することにより、
ポリ塩化ジベンゾパラジオキシン(PCDD)、ポリ塩化ジベンゾフラン(PCDF)及びコプラナーPCB(Co-PCB)の含有量を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルを製造する方法。 - さらに、臭素化難燃剤の含有量も低下させる請求項7の方法。
- 高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルの脂肪酸中に占める高度不飽和脂肪酸の濃度が80面積%以上、85面積%以上、90面積%以上、95面積%以上、又は96面積%以上である、請求項7又は8の方法。
- 高度不飽和脂肪酸を構成脂肪酸として含有する原料油が魚油、オキアミ油、海洋哺乳類油又は微生物油である請求項7ないし9いずれかの方法。
- 前記薄膜蒸留が、200~270℃、220~260℃、又は220~250℃の温度で実行される、請求項7ないし10いずれかの方法。
- 前記薄膜蒸留が、5Pa以下、2Pa、又は1Pa以下の圧力で実行される、請求項7ないし11いずれかの方法。
- 前記薄膜蒸留が、流速20~200(kg/h)/m2、又は25~120(kg/h)/m2で実行される、請求項7ないし12いずれかの方法。
- 前期薄膜蒸留が分子蒸留又は短行程蒸留である請求項7ないし13いずれかの方法。
- 精留が、3塔以上の蒸留塔にて行うものである請求項7ないし14のいずれかの方法。
- カラムクロマトグラフィによる精製が、逆相分配系のカラムクロマトグラフィを用いるものである請求項7ないし15のいずれかの方法。
- 請求項7ないし16いずれかの方法により製造された、ダイオキシン類の含有量が0.07pg-TEQ/g未満、又は0.05 pg-TEQ/g未満である、高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- さらに、含まれる臭素化難燃剤の含有量がBDE-47の量が0.18ng/g未満、BDE-100の量が0.03ng/g未満、BDE-49の量が0.05ng/g未満、又は、BDE-99の量が0.05ng/g未満である請求項17の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- 脂肪酸中に占める高度不飽和脂肪酸の濃度が80面積%以上、85面積%以上、90面積%以上、95面積%以上、又は96面積%以上である請求項17又は18の高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステル。
- 請求項21ないし23いずれかの高度不飽和脂肪酸又は高度不飽和脂肪酸のエチルエステルを含有する医薬品、サプリメント、又は食品。
Priority Applications (19)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020217007317A KR102381331B1 (ko) | 2012-05-14 | 2013-05-14 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| KR1020227010218A KR102453616B1 (ko) | 2012-05-14 | 2013-05-14 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| US14/400,652 US9540306B2 (en) | 2012-05-14 | 2013-05-14 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| EP13790465.2A EP2851361B1 (en) | 2012-05-14 | 2013-05-14 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| ES13790465T ES2900618T3 (es) | 2012-05-14 | 2013-05-14 | Acido graso altamente insaturado o etil éster de ácido graso altamente insaturado con contaminantes ambientales reducidos y método para la producción de los mismos |
| CA2873160A CA2873160C (en) | 2012-05-14 | 2013-05-14 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| KR1020147034425A KR102117725B1 (ko) | 2012-05-14 | 2013-05-14 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| DK13790465.2T DK2851361T3 (da) | 2012-05-14 | 2013-05-14 | Højumættet fedtsyre eller højumættet fedtsyreethylester med reduceret indhold af miljøforurenende stoffer, og fremgangsmåde til fremstilling deraf |
| KR1020227034817A KR20220140655A (ko) | 2012-05-14 | 2013-05-14 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| JP2013546117A JP5592570B2 (ja) | 2012-05-14 | 2013-05-14 | 環境汚染物質を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸エチルエステル及びその製造方法 |
| KR1020207005902A KR102117806B1 (ko) | 2012-05-14 | 2013-05-14 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| KR1020207014617A KR102229106B1 (ko) | 2012-05-14 | 2013-05-14 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| CN201380024834.XA CN104302615B (zh) | 2012-05-14 | 2013-05-14 | 减低了环境污染物质的高度不饱和脂肪酸或高度不饱和脂肪酸乙酯及其制造方法 |
| EP21206640.1A EP3971165A1 (en) | 2012-05-14 | 2013-05-14 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| US15/366,522 US10399922B2 (en) | 2012-05-14 | 2016-12-01 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| US16/019,992 US10399923B2 (en) | 2012-05-14 | 2018-06-27 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| US16/514,191 US11034643B2 (en) | 2012-05-14 | 2019-07-17 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| US17/317,179 US11603347B2 (en) | 2012-05-14 | 2021-05-11 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| US18/164,765 US20230183165A1 (en) | 2012-05-14 | 2023-02-06 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012110809 | 2012-05-14 | ||
| JP2012-110809 | 2012-05-14 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/400,652 A-371-Of-International US9540306B2 (en) | 2012-05-14 | 2013-05-14 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
| US15/366,522 Division US10399922B2 (en) | 2012-05-14 | 2016-12-01 | Highly unsaturated fatty acid or highly unsaturated fatty acid ethyl ester with reduced environmental pollutants, and method for producing same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013172346A1 true WO2013172346A1 (ja) | 2013-11-21 |
Family
ID=49583751
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/063425 WO2013172346A1 (ja) | 2012-05-14 | 2013-05-14 | 環境汚染物質を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸エチルエステル及びその製造方法 |
Country Status (13)
| Country | Link |
|---|---|
| US (6) | US9540306B2 (ja) |
| EP (2) | EP3971165A1 (ja) |
| JP (8) | JP5592570B2 (ja) |
| KR (6) | KR102229106B1 (ja) |
| CN (5) | CN106810446A (ja) |
| CA (2) | CA3225865A1 (ja) |
| CL (1) | CL2014003053A1 (ja) |
| DE (1) | DE202013012875U1 (ja) |
| DK (1) | DK2851361T3 (ja) |
| ES (1) | ES2900618T3 (ja) |
| HU (1) | HUE057283T2 (ja) |
| PE (2) | PE20142380A1 (ja) |
| WO (1) | WO2013172346A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016502573A (ja) * | 2012-11-02 | 2016-01-28 | プロノヴァ・バイオファーマ・ノルゲ・アーエスPronova BioPharma Norge AS | 油組成物からの好ましくない成分の除去 |
| JP2017502114A (ja) * | 2013-12-04 | 2017-01-19 | 日本水産株式会社 | ジホモ−γ−リノレン酸含有微生物油及びジホモ−γ−リノレン酸含有微生物菌体 |
| WO2017038860A1 (ja) * | 2015-08-31 | 2017-03-09 | 日本水産株式会社 | 遊離多価不飽和脂肪酸含有組成物及びその製造方法 |
| JP2019533735A (ja) * | 2016-09-23 | 2019-11-21 | シージェイ チェイルジェダン コーポレイション | 脂肪酸エチルエステルの製造方法 |
| WO2020138282A1 (ja) | 2018-12-26 | 2020-07-02 | 日清ファルマ株式会社 | エイコサペンタエン酸アルキルエステル含有組成物及びその製造方法 |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102229106B1 (ko) * | 2012-05-14 | 2021-03-18 | 닛폰 스이산 가부시키가이샤 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| ITUB20153877A1 (it) * | 2015-09-24 | 2017-03-24 | Tiberio Bruzzese | Metodo di purificazione di gliceridi di acidi grassi, composizioni che ne derivano, e loro uso |
| CN106349070A (zh) * | 2016-08-31 | 2017-01-25 | 山东康曼生物科技有限公司 | 一种从低含量乙酯鱼油生产欧米加‑3‑酸乙酯90的工艺 |
| PL3649219T3 (pl) * | 2017-07-07 | 2023-02-06 | Bunge Loders Croklaan B.V. | Sposób wytwarzania kompozycji uwodornionego tłuszczu |
| CN109438220B (zh) * | 2018-10-31 | 2021-04-13 | 苏州赛分科技有限公司 | 一种从鱼油中纯化epa的方法 |
| CN111302940A (zh) * | 2020-04-08 | 2020-06-19 | 燕园泽龙(涿鹿)生物科技有限公司 | 高纯度α-亚麻酸乙酯的生产方法 |
| JP7289994B2 (ja) * | 2021-03-17 | 2023-06-12 | 株式会社ニッスイ | 精製の方法および精製された製品 |
| CN114057574A (zh) * | 2021-12-03 | 2022-02-18 | 浙江工商大学 | 一种制备高纯度epa乙酯的方法 |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0460917A2 (en) | 1990-06-04 | 1991-12-11 | Nippon Suisan Kaisha, Ltd. | Method of producing eicosapentaenoic acid or an ester derivative thereof |
| JPH0441457B2 (ja) | 1982-02-15 | 1992-07-08 | Baruto Re Hoore Yan | |
| JPH05222392A (ja) | 1991-10-28 | 1993-08-31 | Nippon Suisan Kaisha Ltd | 高純度エイコサペンタエン酸またはそのエステルの製造方法 |
| JPH0633088B2 (ja) | 1990-02-20 | 1994-05-02 | 金沢樹脂工業株式会社 | 組立式フォークリフト用パレット |
| JPH09510091A (ja) * | 1994-03-08 | 1997-10-14 | ノルスク・ヒドロ・アクシェセルスカープ | 精油組成物 |
| JPH1095744A (ja) * | 1996-09-20 | 1998-04-14 | Nof Corp | 高度不飽和脂肪酸又はそのアルキルエステルの製造方法 |
| JPH10310551A (ja) * | 1997-05-12 | 1998-11-24 | Nippon Wax Polymer Kaihatsu Kenkyusho:Kk | エイコサペンタエン酸又はそのエステルの精製方法 |
| JPH11209785A (ja) * | 1998-01-21 | 1999-08-03 | Ikeda Shokken Kk | 高純度高度不飽和脂肪酸の低級アルコールエステル精製方法 |
| JP2001302584A (ja) * | 2000-02-17 | 2001-10-31 | Sumitomo Chem Co Ltd | 脂肪酸エステルの製造方法および脂肪酸エステルを含む燃料 |
| WO2004007654A1 (en) | 2002-07-11 | 2004-01-22 | Pronova Biocare As | A process for decreasing environmental pollutants in an oil or a fat, a volatile environmental pollutants decreasing working fluid, a health supplement, and an animal feed product |
| WO2009017102A1 (ja) | 2007-07-30 | 2009-02-05 | Nippon Suisan Kaisha, Ltd. | Epa濃縮油およびdha濃縮油の製造方法 |
| WO2011080503A2 (en) * | 2009-12-30 | 2011-07-07 | Equateq Limited | Simulated moving bed chromatographic separation process |
| JP2011522913A (ja) * | 2008-05-15 | 2011-08-04 | プロノヴァ バイオファーマ ノルゲ アーエス | オキアミ油加工方法 |
Family Cites Families (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57149400A (en) | 1981-03-12 | 1982-09-14 | Kureha Chemical Ind Co Ltd | Manufacture of high purity long chain highly unsaturated fatty acid ester |
| JPS5888339A (ja) | 1981-11-20 | 1983-05-26 | Kagakuhin Kensa Kyokai | エイコサペンタエン酸又はそのエステルとドコサヘキサエン酸又はそのエステルの分離精製方法 |
| JPS60208940A (ja) | 1984-03-31 | 1985-10-21 | Nippon Zeon Co Ltd | 長鎮不飽和脂肪酸化合物の分離精製法 |
| GB8819110D0 (en) | 1988-08-11 | 1988-09-14 | Norsk Hydro As | Antihypertensive drug & method for production |
| JP3290152B2 (ja) | 1990-06-04 | 2002-06-10 | 日本水産株式会社 | 高濃度エイコサペンタエン酸またはそのエステル |
| JPH07105223B2 (ja) | 1991-03-11 | 1995-11-13 | 日本碍子株式会社 | 高温電池集合体 |
| US5215630A (en) | 1991-06-04 | 1993-06-01 | Nippon Suisan Kaisha, Ltd. | Method of purifying eicosapentaenoic acid or the ester derivative thereof by fractional distillation |
| JP2988754B2 (ja) | 1991-09-03 | 1999-12-13 | 日本水産株式会社 | 高真空連続蒸留装置 |
| JP3614177B2 (ja) | 1992-07-21 | 2005-01-26 | 日本水産株式会社 | 高純度ドコサヘキサエン酸またはそのエステルの 製造方法 |
| JPH08100191A (ja) * | 1994-09-30 | 1996-04-16 | Nisshin Flour Milling Co Ltd | 高度不飽和脂肪酸またはそのエステルの精製方法 |
| NO314788B1 (no) | 1994-11-15 | 2003-05-26 | K D Pharma Gmbh | Framgangsmåte for utvinning av en eller flere forbindelser fra en blanding |
| JPH09302380A (ja) * | 1996-05-10 | 1997-11-25 | Nippon Wax Polymer Kaihatsu Kenkyusho:Kk | エイコサペンタエン酸又はそのエステルの精製方法 |
| CN1072711C (zh) * | 1998-01-05 | 2001-10-10 | 山东禹王制药有限公司 | 一种高度不饱和脂肪酸的工业化生产方法 |
| JP4727039B2 (ja) * | 1998-06-05 | 2011-07-20 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | 汚染物質低下魚油 |
| DE19923558A1 (de) | 1999-05-21 | 2000-11-23 | K D Pharma Bexbach Gmbh | Verfahren zur Herstellung von geruchs- und geschmacksfreien ungesättigten Fettsäuren aus Naturölen und deren Verwendung |
| ES2159257B1 (es) | 1999-11-19 | 2002-04-16 | Kd Iqa S L | Nuevo procedimiento de preparacion del ester etilico del acido eicosa pentaenoico. |
| EP1125914A1 (en) | 2000-02-14 | 2001-08-22 | Nisshin Flour Milling Co., Ltd. | Process for separating and purifying eicosapentaenoic acid or its ester |
| CA2336513C (en) | 2000-02-17 | 2010-08-24 | Tatsuo Tateno | Process for producing fatty acid esters and fuels comprising fatty acid ester |
| US20040022923A1 (en) * | 2000-07-19 | 2004-02-05 | Baldur Hjaltason | Marine oils with reduced levels of contaminants |
| FR2894772B1 (fr) * | 2005-12-20 | 2008-03-14 | Pierre Fabre Medicament Sa | Procede d'enrichissement en dha |
| DE102006012866B4 (de) | 2006-03-19 | 2009-04-09 | Uic Gmbh | Verfahren zur Abtrennung leichtflüchtiger Komponenten aus einem Stoffgemisch sowie Vorrichtung zur Durchführung dieses Verfahrens |
| EP2147088A4 (en) * | 2007-04-26 | 2010-05-05 | Patrick Adlercreutz | FERTILIZED MILK OILS COMPRISING MULTIPLE-UNSATURATED FATTY ACIDS, COMPRISING EICOSAPENTAIC ACID (EPA) AND DOCOSAHEXAIC ACID (DHA), AND MANUFACTURING METHOD THEREFOR |
| DE602007013776D1 (de) | 2007-11-23 | 2011-05-19 | Reemtsma H F & Ph | Verfahren zur Herstellung rauchloser Tabakartikel und rauchloser Tabakartikel zum oralen Genuss |
| EP2548456B1 (en) | 2008-03-20 | 2015-07-08 | Virun, Inc. | Emulsions including a PEG-derivative of tocopherol |
| KR101357298B1 (ko) | 2008-06-20 | 2014-01-28 | 에이케이 앤 엠엔 바이오팜 주식회사 | 오메가-3계 고도불포화 지방산의 고순도 정제방법 |
| JP2010118383A (ja) * | 2008-11-11 | 2010-05-27 | Nikon Corp | 照明装置、露光装置、及びデバイス製造方法 |
| PH12019500030A1 (en) | 2009-03-09 | 2020-02-10 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| CL2009001343A1 (es) | 2009-06-02 | 2009-07-10 | Golden Omega S A | Proceso de obtencion concentrado de esteres de epa y dha a partir de aceite marino, que comprende agregar al aceite alcali y agua a menos de 100 grados celsius, agregar solvente, separar fase de refinado, agregar acido, separar la fase no acuosa y agregar alcohol y un catalizador a menos de 150 grados celsius, desolventilizar y destilar. |
| EP2563164B1 (en) | 2010-03-23 | 2016-06-29 | Virun, Inc. | Nanoemulsion including sucrose fatty acid ester |
| JP5905821B2 (ja) | 2010-05-28 | 2016-04-20 | 日本水産株式会社 | リパーゼによる高度不飽和脂肪酸含有油脂の製造方法 |
| ES2733134T3 (es) * | 2010-06-09 | 2019-11-27 | Fermentalg | Composiciones que comprenden ácido eicosapentaenoico adecuado para una alta purificación |
| US9145533B2 (en) | 2010-09-24 | 2015-09-29 | Pronova Blopharm Norge AS | Process for concentrating omega-3 fatty acids |
| JP5650506B2 (ja) | 2010-11-22 | 2015-01-07 | 優章 荒井 | 電解水の製造装置 |
| EP2673371A1 (en) * | 2011-02-11 | 2013-12-18 | E.I. Du Pont De Nemours And Company | An eicosapentaenoic acid concentrate |
| GB201111591D0 (en) * | 2011-07-06 | 2011-08-24 | Equateq Ltd | Further new process |
| ES2395320B1 (es) | 2011-07-14 | 2013-12-18 | Soluciones Extractivas Alimentarias, S.L. | Nuevo método para la reducción de contaminantes en grasas y aceites a partir de aceites y sus derivados. |
| WO2013068846A1 (en) * | 2011-11-09 | 2013-05-16 | Pronova Biopharma Norge As | Membrane-based processes for reducing at least one impurity and making a concentrate comprising at least one natural component from a marine fatty acid oil mixture, and compositions resulting thereof |
| US8258330B1 (en) * | 2012-01-04 | 2012-09-04 | Naturalis, S.A. | Carrier fluid composition comprising fatty acids ethyl esters and process for reducing the concentration of persistent organic pollutants in fish oil |
| KR102229106B1 (ko) * | 2012-05-14 | 2021-03-18 | 닛폰 스이산 가부시키가이샤 | 환경 오염 물질을 저감시킨 고도 불포화 지방산 또는 고도 불포화 지방산 에틸에스테르 및 그 제조 방법 |
| ES2875965T3 (es) * | 2013-12-04 | 2021-11-11 | Nippon Suisan Kaisha Ltd | Aceite microbiano que contiene ácido dihomo-gamma-linolénico y biomasa microbiana que contiene ácido dihomo-gamma-linolénico |
-
2013
- 2013-05-14 KR KR1020207014617A patent/KR102229106B1/ko active Application Filing
- 2013-05-14 EP EP21206640.1A patent/EP3971165A1/en active Pending
- 2013-05-14 WO PCT/JP2013/063425 patent/WO2013172346A1/ja active Application Filing
- 2013-05-14 CN CN201710031298.6A patent/CN106810446A/zh active Pending
- 2013-05-14 DK DK13790465.2T patent/DK2851361T3/da active
- 2013-05-14 PE PE2014002012A patent/PE20142380A1/es active IP Right Grant
- 2013-05-14 KR KR1020227010218A patent/KR102453616B1/ko active IP Right Grant
- 2013-05-14 CN CN202110295239.6A patent/CN113045422A/zh active Pending
- 2013-05-14 CN CN201710701544.4A patent/CN107602378B/zh active Active
- 2013-05-14 CN CN202110299534.9A patent/CN113025434A/zh active Pending
- 2013-05-14 KR KR1020227034817A patent/KR20220140655A/ko not_active Application Discontinuation
- 2013-05-14 CN CN201380024834.XA patent/CN104302615B/zh active Active
- 2013-05-14 KR KR1020207005902A patent/KR102117806B1/ko active IP Right Grant
- 2013-05-14 KR KR1020217007317A patent/KR102381331B1/ko active IP Right Grant
- 2013-05-14 HU HUE13790465A patent/HUE057283T2/hu unknown
- 2013-05-14 CA CA3225865A patent/CA3225865A1/en active Pending
- 2013-05-14 KR KR1020147034425A patent/KR102117725B1/ko active IP Right Grant
- 2013-05-14 CA CA2873160A patent/CA2873160C/en active Active
- 2013-05-14 PE PE2019001187A patent/PE20190969A1/es unknown
- 2013-05-14 JP JP2013546117A patent/JP5592570B2/ja active Active
- 2013-05-14 US US14/400,652 patent/US9540306B2/en active Active
- 2013-05-14 EP EP13790465.2A patent/EP2851361B1/en active Active
- 2013-05-14 ES ES13790465T patent/ES2900618T3/es active Active
- 2013-05-14 DE DE202013012875.5U patent/DE202013012875U1/de not_active Expired - Lifetime
-
2014
- 2014-06-03 JP JP2014114769A patent/JP6309828B2/ja active Active
- 2014-11-10 CL CL2014003053A patent/CL2014003053A1/es unknown
-
2016
- 2016-12-01 US US15/366,522 patent/US10399922B2/en active Active
-
2017
- 2017-07-04 JP JP2017130947A patent/JP6434573B2/ja active Active
-
2018
- 2018-04-03 JP JP2018071685A patent/JP6389579B2/ja active Active
- 2018-06-27 US US16/019,992 patent/US10399923B2/en active Active
- 2018-09-27 JP JP2018181274A patent/JP6774469B2/ja active Active
-
2019
- 2019-07-17 US US16/514,191 patent/US11034643B2/en active Active
-
2020
- 2020-10-02 JP JP2020167387A patent/JP2021004254A/ja active Pending
-
2021
- 2021-05-11 US US17/317,179 patent/US11603347B2/en active Active
-
2022
- 2022-07-01 JP JP2022106906A patent/JP2022132317A/ja not_active Withdrawn
-
2023
- 2023-02-06 US US18/164,765 patent/US20230183165A1/en active Pending
-
2024
- 2024-02-28 JP JP2024028104A patent/JP2024069262A/ja active Pending
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0441457B2 (ja) | 1982-02-15 | 1992-07-08 | Baruto Re Hoore Yan | |
| JPH0633088B2 (ja) | 1990-02-20 | 1994-05-02 | 金沢樹脂工業株式会社 | 組立式フォークリフト用パレット |
| EP0460917A2 (en) | 1990-06-04 | 1991-12-11 | Nippon Suisan Kaisha, Ltd. | Method of producing eicosapentaenoic acid or an ester derivative thereof |
| JPH05222392A (ja) | 1991-10-28 | 1993-08-31 | Nippon Suisan Kaisha Ltd | 高純度エイコサペンタエン酸またはそのエステルの製造方法 |
| EP0610506A1 (en) | 1991-10-28 | 1994-08-17 | Nippon Suisan Kabushiki Kaisha | Process for producing high-purity eicosapentaenoic acid or ester thereof |
| JPH09510091A (ja) * | 1994-03-08 | 1997-10-14 | ノルスク・ヒドロ・アクシェセルスカープ | 精油組成物 |
| JPH1095744A (ja) * | 1996-09-20 | 1998-04-14 | Nof Corp | 高度不飽和脂肪酸又はそのアルキルエステルの製造方法 |
| JPH10310551A (ja) * | 1997-05-12 | 1998-11-24 | Nippon Wax Polymer Kaihatsu Kenkyusho:Kk | エイコサペンタエン酸又はそのエステルの精製方法 |
| JPH11209785A (ja) * | 1998-01-21 | 1999-08-03 | Ikeda Shokken Kk | 高純度高度不飽和脂肪酸の低級アルコールエステル精製方法 |
| JP2001302584A (ja) * | 2000-02-17 | 2001-10-31 | Sumitomo Chem Co Ltd | 脂肪酸エステルの製造方法および脂肪酸エステルを含む燃料 |
| WO2004007654A1 (en) | 2002-07-11 | 2004-01-22 | Pronova Biocare As | A process for decreasing environmental pollutants in an oil or a fat, a volatile environmental pollutants decreasing working fluid, a health supplement, and an animal feed product |
| JP3905538B2 (ja) | 2002-07-11 | 2007-04-18 | プロノヴァ・バイオケア・アーエス | 油または脂肪中の環境汚染物質の低減方法、揮発性環境汚染物質低減作業流体、健康サプリメントおよび動物飼料製品 |
| WO2009017102A1 (ja) | 2007-07-30 | 2009-02-05 | Nippon Suisan Kaisha, Ltd. | Epa濃縮油およびdha濃縮油の製造方法 |
| JP2011522913A (ja) * | 2008-05-15 | 2011-08-04 | プロノヴァ バイオファーマ ノルゲ アーエス | オキアミ油加工方法 |
| WO2011080503A2 (en) * | 2009-12-30 | 2011-07-07 | Equateq Limited | Simulated moving bed chromatographic separation process |
Non-Patent Citations (9)
| Title |
|---|
| "JOCS Standard Methods for the Analysis of Fats, Oils and Related Materials", 2003, JAPAN OIL CHEMISTS' SOCIETY |
| "Provisional Guidelines on the Methods of Measuring Dioxins in Foods (February 2008", EISHOKU NO. 138 AND EINYU NO. 200, 1999 |
| ANTHONY P. BIMBO: "Guidelines for characterization of food-grade fish oil", INFORM, vol. 9, no. 5, 1998, pages 473 - 483 |
| HARALD BREIVIK; OLAV THORSTAD: "Removal of organic environmental pollutants from fish oil by short-path distillation", LIPID TECHNOLOGY, vol. 17, no. 3, 2005, pages 55 - 58 |
| JIRI CMOLIK; JAN POKOMY: "Physical refining of edible oils", EUR. J. LIPID SCI. TECHNOL., vol. 102, no. 7, 2000, pages 472 - 486 |
| K. JULSHAMN; L. KARLSEN; O.R. BRAEKKAN: "Removal of DDT and its metabolites from fish oils by molecular distillation", FISKERIDIREKTORATETSSKRIFTER: SERIE TEKNOLOGISKE UNDERSOKELSER, vol. 5, no. 15, 1973 |
| RYUJI YAMAMURA ET AL.: "High Purification of Polyunsaturated Fatty Acids", JOURNAL OF JAPAN OIL CHEMISTS' SOCIETY, vol. 47, no. 5, 1998, pages 449 - 456, XP008175194 * |
| VERONIQUE FOURNIER ET AL.: "Thermal degradation of long-chain polyunsaturated fatty acids during deodorization of fish oil.", EUR. J. LIPID SCI. TECHNOL., vol. 108, 2006, pages 33 - 42 |
| YUSHI, vol. 62, no. 11, 2009, pages 38 - 39 |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016502573A (ja) * | 2012-11-02 | 2016-01-28 | プロノヴァ・バイオファーマ・ノルゲ・アーエスPronova BioPharma Norge AS | 油組成物からの好ましくない成分の除去 |
| JP2022061976A (ja) * | 2013-12-04 | 2022-04-19 | 日本水産株式会社 | ジホモ-γ-リノレン酸含有微生物油及びジホモ-γ-リノレン酸含有微生物菌体 |
| JP2019208512A (ja) * | 2013-12-04 | 2019-12-12 | 日本水産株式会社 | ジホモ−γ−リノレン酸含有微生物油及びジホモ−γ−リノレン酸含有微生物菌体 |
| JP7466514B2 (ja) | 2013-12-04 | 2024-04-12 | 株式会社ニッスイ | ジホモ-γ-リノレン酸含有微生物油及びジホモ-γ-リノレン酸含有微生物菌体 |
| US11856952B2 (en) | 2013-12-04 | 2024-01-02 | Nippon Suisan Kaisha, Ltd. | Microbial oil, production method for microbial oil, concentrated microbial oil, and production method for concentrated microbial oil |
| US11330817B2 (en) | 2013-12-04 | 2022-05-17 | Nippon Suisan Kaisha, Ltd. | Microbial oil, production method for microbial oil, concentrated microbial oil, and production method for concentrated microbial oil |
| JP2017502114A (ja) * | 2013-12-04 | 2017-01-19 | 日本水産株式会社 | ジホモ−γ−リノレン酸含有微生物油及びジホモ−γ−リノレン酸含有微生物菌体 |
| US10750741B2 (en) | 2013-12-04 | 2020-08-25 | Nippon Suisan Kaisha, Ltd. | Dihomo-γ-linolenic acid-containing microbial oil and dihomo-γ-linolenic acid-containing microbial biomass |
| JP6995807B2 (ja) | 2013-12-04 | 2022-01-17 | 日本水産株式会社 | ジホモ-γ-リノレン酸含有微生物油及びジホモ-γ-リノレン酸含有微生物菌体 |
| US10626347B2 (en) | 2015-08-31 | 2020-04-21 | Nippon Suisan Kaisha, Ltd. | Free polyunsaturated fatty acid-containing composition and manufacturing method therefor |
| JPWO2017038860A1 (ja) * | 2015-08-31 | 2018-06-14 | 日本水産株式会社 | 遊離多価不飽和脂肪酸含有組成物及びその製造方法 |
| WO2017038860A1 (ja) * | 2015-08-31 | 2017-03-09 | 日本水産株式会社 | 遊離多価不飽和脂肪酸含有組成物及びその製造方法 |
| US11193085B2 (en) | 2015-08-31 | 2021-12-07 | Nippon Suisan Kaisha, Ltd. | Free-polyunsaturated-fatty-acid-containing composition and method for manufacturing same |
| US11414622B2 (en) | 2015-08-31 | 2022-08-16 | Nippon Suisan Kaisha, Ltd. | Free polyunsaturated fatty acid-containing composition and manufacturing method therefor |
| US11976254B2 (en) | 2015-08-31 | 2024-05-07 | Nissui Corporation | Free polyunsaturated fatty acid-containing composition and manufacturing method therefor |
| JP2019533735A (ja) * | 2016-09-23 | 2019-11-21 | シージェイ チェイルジェダン コーポレイション | 脂肪酸エチルエステルの製造方法 |
| WO2020138282A1 (ja) | 2018-12-26 | 2020-07-02 | 日清ファルマ株式会社 | エイコサペンタエン酸アルキルエステル含有組成物及びその製造方法 |
| KR20210108948A (ko) | 2018-12-26 | 2021-09-03 | 닛신 파마 가부시키가이샤 | 에이코사펜타엔산 알킬에스테르 함유 조성물 및 그 제조 방법 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6389579B2 (ja) | 環境汚染物質を低減させた高度不飽和脂肪酸又は高度不飽和脂肪酸エチルエステル及びその製造方法 | |
| JP3905538B2 (ja) | 油または脂肪中の環境汚染物質の低減方法、揮発性環境汚染物質低減作業流体、健康サプリメントおよび動物飼料製品 | |
| US10696924B1 (en) | Composition containing highly unsaturated fatty acid or alkyl ester thereof and a method for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2013546117 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13790465 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2013790465 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013790465 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2873160 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014003053 Country of ref document: CL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14400652 Country of ref document: US Ref document number: 002012-2014 Country of ref document: PE |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20147034425 Country of ref document: KR Kind code of ref document: A |