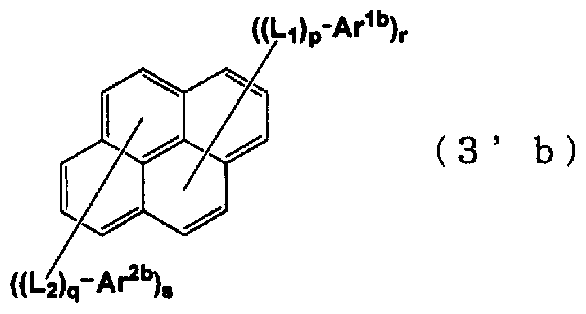WO2007100010A1 - 有機エレクトロルミネッセンス素子 - Google Patents
有機エレクトロルミネッセンス素子 Download PDFInfo
- Publication number
- WO2007100010A1 WO2007100010A1 PCT/JP2007/053806 JP2007053806W WO2007100010A1 WO 2007100010 A1 WO2007100010 A1 WO 2007100010A1 JP 2007053806 W JP2007053806 W JP 2007053806W WO 2007100010 A1 WO2007100010 A1 WO 2007100010A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituted
- unsubstituted
- carbon atoms
- branched
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/622—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing four rings, e.g. pyrene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/623—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing five rings, e.g. pentacene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/625—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing at least one aromatic ring having 7 or more carbon atoms, e.g. azulene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1014—Carbocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1037—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with sulfur
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
- C09K2211/1048—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms with oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
- C09K2211/1051—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms with sulfur
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1092—Heterocyclic compounds characterised by ligands containing sulfur as the only heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1096—Heterocyclic compounds characterised by ligands containing other heteroatoms
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
Definitions
- the present invention relates to an organic electoluminescence device containing a compound having a fluoranthene structure and a condensed ring-containing compound, and in particular, by using the compound having a fluoranthene structure and the condensed ring-containing compound in a light-emitting layer,
- the present invention relates to an organic-electric-luminescence device that can produce blue light emission with a long and high luminous efficiency.
- An organic electoluminescence device (hereinafter abbreviated as electroluminescence) is recombination of holes injected from an anode and electrons injected from a cathode by applying an electric field. It is a self-luminous device that utilizes the principle that fluorescent substances emit light by energy.
- Eastman's Kodak's CW Tang et al. Reported low-voltage-driven organic EL devices using stacked devices (CW Tang, SA Vanslyke, Applied Physics Letters, 51 ⁇ , 913, 1987, etc.) Since then, research on organic EL devices using organic materials as constituent materials has been actively conducted. Tang et al.
- the device structure of the organic EL device is a two-layer type of a hole transport (injection) layer and an electron transport light-emitting layer, or a hole transport (injection) layer, a light-emitting layer, and an electron transport (injection) layer.
- the three-layer type is well known. In such a stacked structure element, the element structure and the formation method are devised in order to increase the recombination efficiency of injected holes and electrons.
- light-emitting materials such as chelate complexes such as tris (8-quinolinolato) aluminum complex, coumarin derivatives, tetraphenylbutadiene derivatives, bisstyrylarylene derivatives, oxadiazole derivatives and the like are known. It has been reported that light emission in the visible range from blue to red can be obtained, and the realization of color display elements is expected. Waiting (for example, Patent Document 1, Patent Document 2, Patent Document 3, etc.).
- Patent Document 4 Devices using bisanthracene derivatives as light-emitting materials are disclosed in Patent Document 4 and Patent Document 5.
- Bisanthracene was insufficient as the power to be used as a blue light-emitting material because its efficiency and lifetime did not reach a practical level.
- Patent Document 6 elements using a symmetric pyrene derivative as a light emitting material are disclosed in Patent Document 6, Patent Document 7, Patent Document 8, and Patent Document 9.
- Such symmetrical pyrene derivatives have been required to improve the lifetime of force elements used as blue light emitting materials.
- Patent Document 10 Devices using a fluoranthene derivative as a light emitting material are disclosed in Patent Document 10, Patent Document 11, Patent Document 12, Patent Document 13, and Patent Document 14.
- a fluoranthene derivative is used as a blue light-emitting material, but there has been a demand for improvement in device lifetime.
- Patent Document 1 Japanese Patent Application Laid-Open No. 8-239655
- Patent Document 2 JP-A-7-183561
- Patent Document 3 JP-A-3-200889
- Patent Document 4 US Patent 3008897 Specification
- Patent Document 5 JP-A-8-12600
- Patent Document 6 JP 2001-118682 A
- Patent Document 7 Japanese Unexamined Patent Application Publication No. 2002-63988
- Patent Document 8 Japanese Patent Application Laid-Open No. 2004-75567
- Patent Document 9 Japanese Patent Laid-Open No. 2004-83481
- Patent Document 10 Japanese Unexamined Patent Application Publication No. 2002-69044
- Patent Document 11 International Publication WO02 / 085822
- Patent Document 12 International Publication WO2005 / 033051
- Patent Document 13 JP-A-10-189247
- Patent Document 14 Japanese Unexamined Patent Application Publication No. 2005-68087
- the present invention has been made to solve the above-described problems, and has a long lifetime and high luminous efficiency.
- An object of the present invention is to provide an organic EL device that can emit blue light.
- the present inventors have conducted intensive research, and as a result, an organic EL layer in which one or more organic thin film layers including at least a light emitting layer are sandwiched between a cathode and an anode.
- the light-emitting layer contains at least one selected from a condensed ring-containing compound represented by the following general formula (1) and at least one selected from a compound having a fluoranthene structure.
- the luminescence element has been found to be capable of producing blue light emission with a long lifetime and high light emission efficiency, and has completed the present invention.
- the light emitting layer has at least one selected from the group consisting of condensed ring-containing compounds represented by the following general formula (1) and at least one selected from compounds having a fluoranthene structure. Containing.
- G 2 is a condensed ring-containing compound, and is a substituted or unsubstituted anthracene structure, a substituted or unsubstituted pyrene structure, a substituted or unsubstituted amine structure, or a substituted or unsubstituted benzene structure. It is a compound which has this.
- the compound having a fluoranthene structure used in the organic EL device of the present invention is preferably represented by the following general formula (2).
- FL is a monovalent group having a fluoranthene structure
- n is an integer of 2 to 4.
- Multiple FLs may be the same or different.
- G 1 is a hydrogen atom when n is 1, and a substituted or unsubstituted aromatic ring group having 6 to 40 carbon atoms, a substituted or unsubstituted carbon atom having 6 to 40 carbon atoms when n is 2 or more.
- the light emitting layer contains at least one selected from a condensed ring-containing compound represented by the following general formula (1) and at least one selected from a compound having a fluoranthene structure.
- the organic electoluminescence device of the present invention provides blue light emission with a long lifetime and high luminous efficiency.
- a protective layer is provided on the surface of the device, or the entire device is protected by silicone oil, resin, etc. It is also possible.
- the organic EL device of the present invention is an organic EL device in which an organic thin film layer having at least one light emitting layer or a plurality of light emitting layers is sandwiched between a cathode and an anode, and the light emitting layer is represented by the following general formula (1). At least one selected from the condensed ring-containing compounds and at least one selected from compounds having a fluoranthene structure. This organic EL device can produce blue light with a long lifetime and high luminous efficiency.
- the condensed ring-containing compound used in the organic EL device of the present invention is represented by the following general formula (1)
- G 2 is a condensed ring-containing compound, and is a substituted or unsubstituted anthracene structure, a substituted or unsubstituted pyrene structure, a substituted or unsubstituted amine structure, or a substituted or unsubstituted benzene structure. It is a compound which has this.
- the organic EL device of the present invention is selected from at least one compound selected from the condensed ring-containing compounds represented by the following general formula (1) and a compound having a fluoranthene structure represented by the following general formula (2) Contains at least one.
- G 2 is a condensed ring-containing compound, and is a substituted or unsubstituted anthracene structure, a substituted or unsubstituted pyrene structure, a substituted or unsubstituted amine structure, or a substituted or unsubstituted benzene structure. It is a compound which has this.
- FL is a monovalent group having a fluoranthene structure
- n is an integer of 2 to 4.
- Multiple FLs may be the same or different.
- G 1 is a substituted or unsubstituted aromatic ring group having 6 to 40 carbon atoms, substituted or unsubstituted Substituted aryl group having 6 to 40 carbon atoms, substituted or unsubstituted diaminoaryl group having 60 to 60 carbon atoms, substituted or unsubstituted triaminoaryl group having 6 to 60 carbon atoms, substituted or unsubstituted A heterocyclic group having 3 to 40 carbon atoms, a substituted or unsubstituted ethenylene group, and containing a single bond.
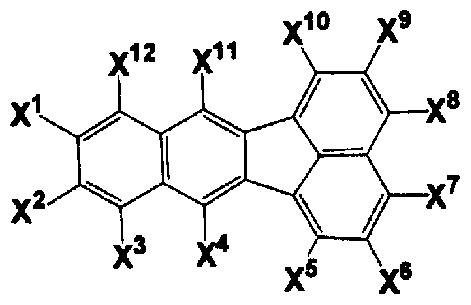
- the compound having a fluoranthene structure is a compound having a structure represented by the following general formula (la).
- ⁇ to ° each independently represents a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic alkyl group having 1 to 30 carbon atoms, a substituted Or an unsubstituted linear, branched or cyclic alkoxy group having 1 to 30 carbon atoms, a substituted or unsubstituted linear, branched or cyclic alkylthio group having 1 to 30 carbon atoms, substituted or Is an unsubstituted linear, branched or cyclic alkenyl group having 2 to 30 carbon atoms, a substituted or unsubstituted linear, branched or cyclic alkenyloxy group having 2 to 30 carbon atoms, substituted or unsubstituted.
- the compound having a fluoranthene structure is a compound having a structure represented by the following general formula (lb).
- ⁇ to X 12 are each independently a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic alkyl group having 1 to 30 carbon atoms, Substituted or unsubstituted linear, branched or cyclic alkoxy group having 1 to 30 carbon atoms, substituted or unsubstituted linear, branched or cyclic alkylthio group having 1 to 30 carbon atoms, substituted Alternatively, an unsubstituted straight chain, branched or cyclic alkenyl group having 2 to 30 carbon atoms, a substituted or unsubstituted straight chain, branched or cyclic alkenyloxy group having 2 to 30 carbon atoms, substituted or unsubstituted Linear, branched or cyclic alkenylthio group having 2 to 30 carbon atoms, substituted or unsubstituted aralkyl group having 7 to
- X 4 and X 11 are not both hydrogen atoms.
- the compound having a fluoranthene structure is represented by the general formula (lb), and G 2 in the general formula (1) is not 9, 10-diphenylanthracene.
- the compound having a fluoranthene structure is a compound having a structure represented by the following general formula (lc).
- ⁇ to X 12 each independently represents a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic alkyl group having 1 to 30 carbon atoms, or a substituted group.
- an unsubstituted linear, branched or cyclic alkoxy group having 1 to 30 carbon atoms a substituted or unsubstituted linear, branched or cyclic alkylthio group having 1 to 30 carbon atoms, substituted or unsubstituted
- X 4 and X 11 are not hydrogen atoms.
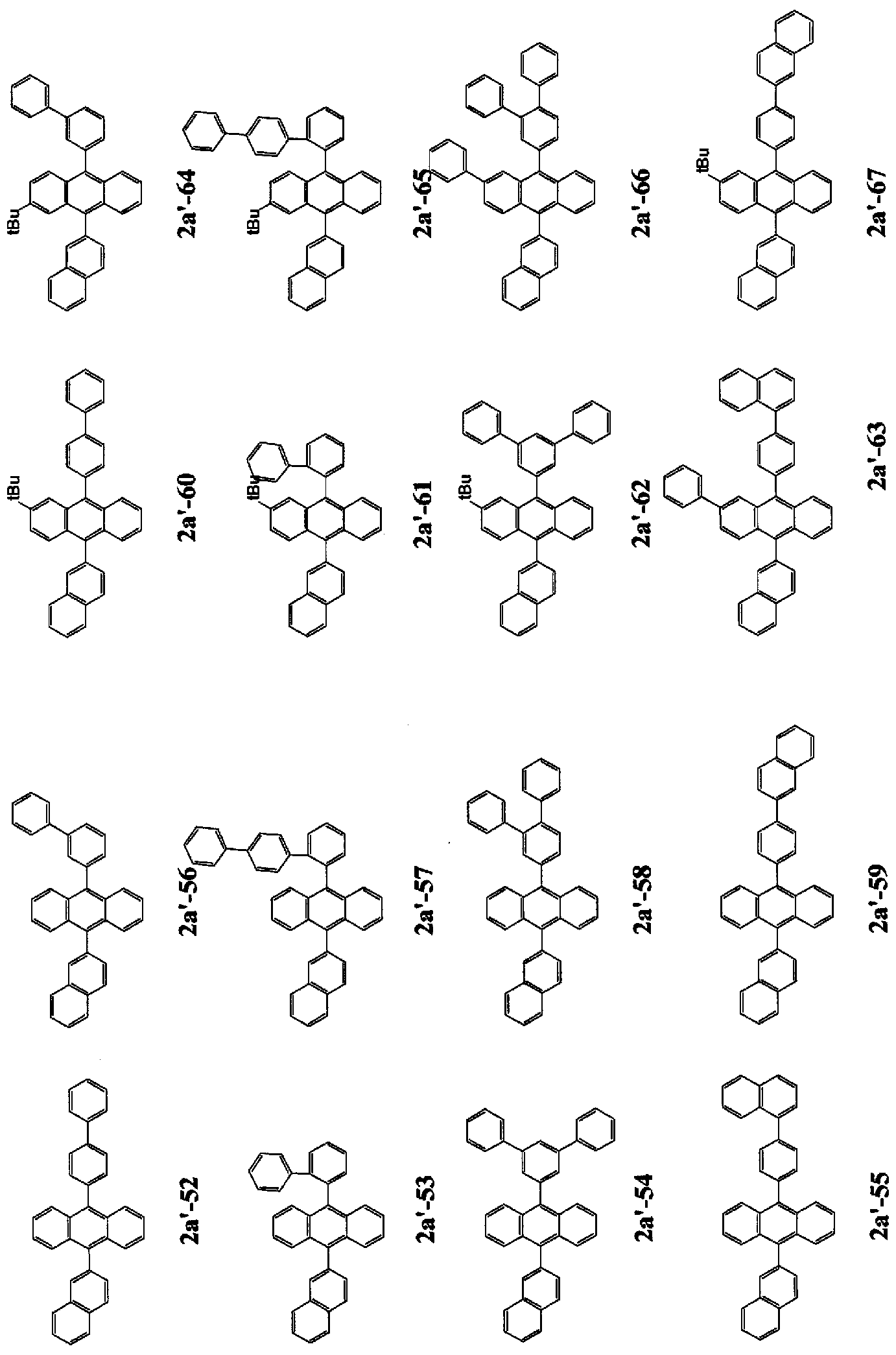
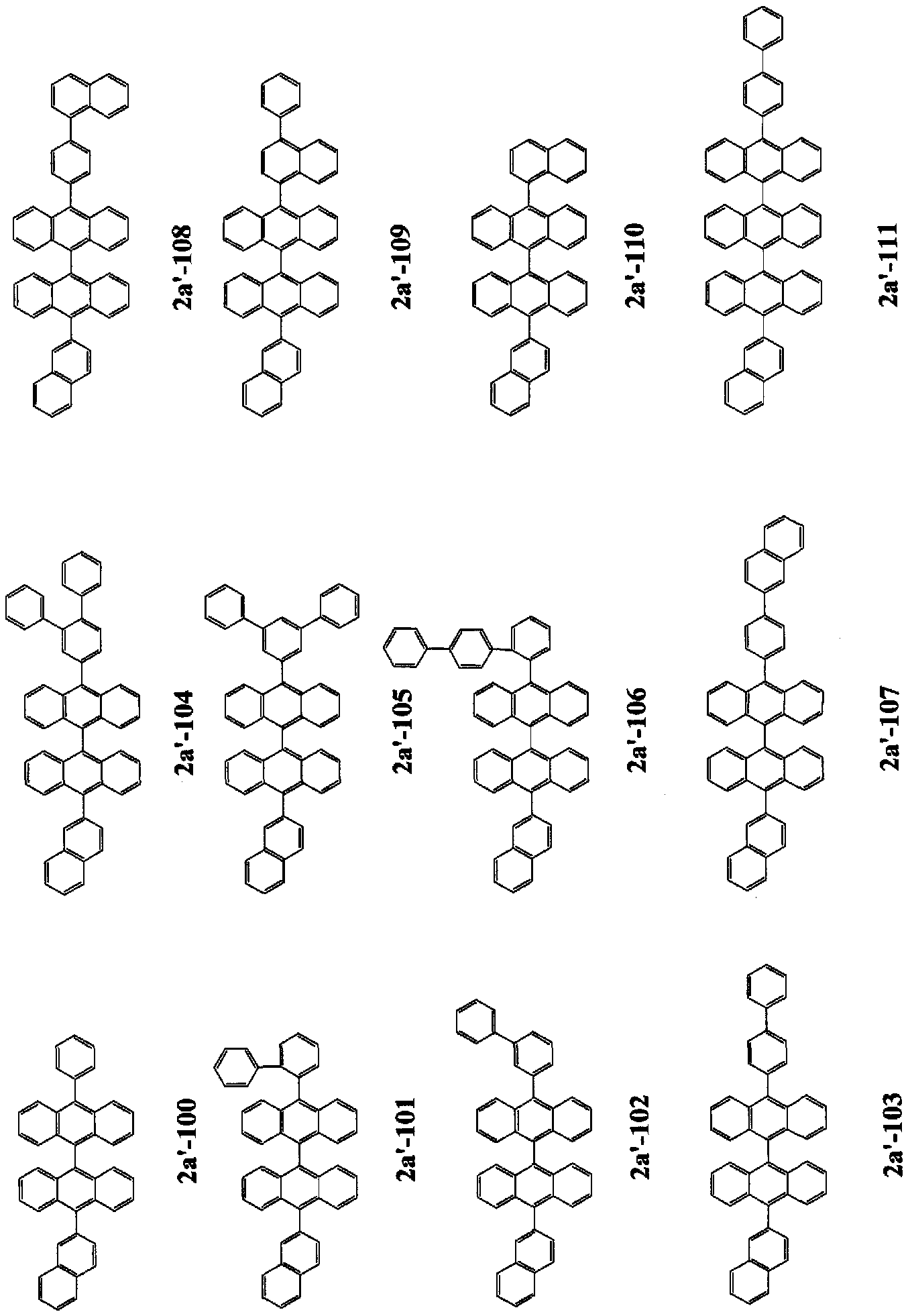
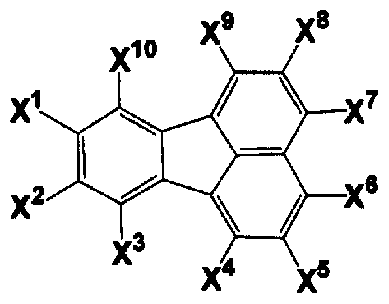
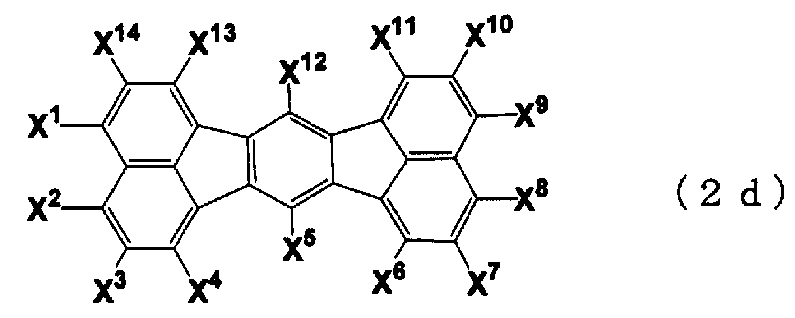
- n is 2 to 4, and a monovalent group derived from a compound in which FL has a structure represented by any one of the following general formulas (2a), (2c), and (2d) It is.
- X ⁇ X 16 is independently a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic carbon atom number.
- _C_ ⁇ _R 2e group represents a substituted or unsubstituted Ararukiru group having 7 to 30 carbon atoms, or substituted or unsubstituted Ariru group having 6 to 30 carbon atoms
- group, R 2e is water Primary atom, substituted or unsubstituted linear, branched or cyclic aralkyl group having 1 to 30 carbon atoms, substituted or unsubstituted linear, branched or cyclic alkenyl group having 2 to 30 carbon atoms, substituted Or an unsubstituted aralkyl group having 7 to 30 carbon atoms, substituted or Represents an unsubstituted aryl group having 6 to 30 carbon atoms, or an amino group
- _ ⁇ COR 3e group wherein R 3 e is a substituted or unsubstituted linear, branched or cyclic carbon atom number) 1-30 alkyl groups, substituted or unsubstituted linear, branche
- n 2 to 4
- FL is a monovalent group derived from a compound having a structure represented by the following general formula (2′b).
- ⁇ to X 12 are each independently a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic alkyl group having 1 to 30 carbon atoms, a substituted group.
- an unsubstituted linear, branched or cyclic alkoxy group having 1 to 30 carbon atoms a substituted or unsubstituted linear, branched or cyclic alkylthio group having 1 to 30 carbon atoms, substituted or Is an unsubstituted linear, branched or cyclic alkenyl group having 2 to 30 carbon atoms, substituted Or an unsubstituted straight chain, branched or cyclic alkenyloxy group having 2 to 30 carbon atoms, a substituted or unsubstituted straight chain, branched or cyclic alkenylthio group having 2 to 30 carbon atoms, substituted or unsubstituted 7 to 30 aralkyl groups, substituted or unsubstituted carbon atoms 7 to 30 aralkyloxy groups, substituted or unsubstituted carbon atoms 7 to 30 aralkylthio groups, substituted or unsubstituted carbon atoms 6
- n is 2 and G 1 is a linking group consisting of a phenylene group, and G 2 in the general formula (1) is 10, 10 ′ bis (2 ) No 9, 9, _Bianthracene.
- alkyl group examples include methyl group, ethyl group, propyl group, isopropyl group, ⁇ butyl group, s butynole group, isobutyl group, t_butyl group, n_pentyl group, n_hexyl group, n_ Heptyl group, n-octyl Nore group, n-Noninore group, n-Desinore group, n-Undecinole group, n-Dodecinole group, n Tridecinole group, n-Tethesinore group, n-Pentadenole group, n-Hexatenanol group, n-Heptadesinore group, n
- a substituted or unsubstituted, linear, alkoxy group and alkylthio group having 1 to 30 carbon atoms of branched or cyclic, respectively - is a group represented by OY 1 and SY 2, the Y 1 and Y 2 Specific examples include the same as those described for the alkyl group, and preferred examples are also the same.
- Examples of the substituted or unsubstituted linear, branched or cyclic alkenyl group having 2 to 30 carbon atoms include, for example, vinyl group, aryl group, 1-buturyl group, 2 butyr group, 3 butenyl group, 1 , 3-butanegenyl group, 1-methylvinyl group, styryl group, 2,2-diphenylenobininore group, 1,2-diphenylvinyl group, 1-methylararyl group, 1,1 dimethylarinole group, 2-methinorealinore group, 1phenenoreali Nore group, 2 phenylenolinole group, 3 phenylenolyl group, 3, 3 diphenylaryl group, 1,2 dimethenoarelinole group, 1-phenolino 1-butenyl group, 3 phenyl 1-butenyl group
- a substituted or unsubstituted linear, branched or cyclic alkenyloxy group having 2 to 30 carbon atoms and a substituted or unsubstituted linear, branched or cyclic alkenyloxy group having 2 to 30 carbon atoms are groups represented by —OY 3 and —SY 4 , respectively. Specific examples of Y 3 and Y 4 include those described for the alkenyl group, and preferred examples are also the same. .
- Examples of the substituted or unsubstituted aralkyl group having 7 to 30 carbon atoms include benzinole group, 1-phenylethyl group, 2-phenylethyl group, 1_phenylisopropyl group, and 2-phenylisopropyl group.
- benzyl group preferred are benzyl group, p_cyanobenzyl group, m_cyanobenzenole group, ocyanobenzyl group, 1 phenylethyl group, 2-phenylethyl group, 1 phenylisopropyl group, 2-phenylisopropyl group and the like.
- a substituted or unsubstituted Ararukiruchio group Ararukiruokishi group and a substituted or unsubstituted having 7 to 30 carbon atoms having 7 to 30 carbon atoms, respectively - is a group represented by SY 6, - OY 5 and Specific examples of Y 5 and Y 6 include the same as those described for the aralkyl group, and preferred examples are also the same.
- Examples of the substituted or unsubstituted aryl group having 6 to 20 carbon atoms include a phenylol group, a 1 naphthyl group, a 2 naphthyl group, a 1 anthryl group, a 2 anthryl group, a 9 anthryl group, and a 1 phenanthrinol.
- phenyl group 1_naphthyl group, 2_naphthyl group, 9-phenanthryl group, 2-biphenylyl group, 3-biphenylyl group, 4-biphenylyl group, p-tolyl group are preferable. 3, 4-xylyl group and the like.
- Examples of 16 substituted or unsubstituted aromatic heterocyclic groups are: 1_pyrrolyl group, 2 —pyrrolyl group, 3_pyrrolinole group, pyraduryl group, 2_pyridinyl group, 3_pyridininole group, 4-pyridinyl group Group, 1_indolyl group, 2_indolyl group, 3_indolyl group, 4_indolinole group, 5_indolyl group, 6_indolyl group, 7_indolyl group, 1_isoindolyl group, 2-isoindolyl group, 3_ Isoindolyl group, 4_isoindolyl group, 5_isoindolyl group, 6_isoindolyl group, 7_isoindolyl group, 2-furinole group, 3-furinole group, 2_benzofuranyl group, 3-benzofuranyl group,
- the substituted or unsubstituted aryloxy group having 6 to 20 carbon atoms and the substituted or unsubstituted aryloxy group having 6 to 20 carbon atoms are groups represented by —OY 7 and —SY 8 respectively. Specific examples of Y 7 and Y 8 are the same as those described for the aryl group, and preferred examples are also the same.
- a substituted or unsubstituted amino group having 2 to 30 carbon atoms it includes an alkylamino group and Ararukiruamino group, expressed as NQ ⁇ 2, specific examples of Q 1 Q 2 are their respective Independently, the same groups as those described for the alkyl group, the aryl group, and the aralkyl group can be given, and preferred examples are also the same.
- CO_ ⁇ _R le group, COR 2e group, and single ⁇ _C_ ⁇ _R 3e R le in groups substitution or unsubstituted linear is R and R 3e, branched or cyclic alkyl group having 1 to 30 carbon atoms
- Specific examples of 30 aryl groups or amino groups include the same specific examples as described above.
- Xi X 16 is preferably a substituted or unsubstituted aryl group having 6 to 20 carbon atoms.
- the compound having a fluoranthene structure contained in the organic electoluminescence device of the present invention is preferably such that n is 2 and the two FLs are different from each other in the general formula (2).
- the compound having a fluoranthene structure contained in the organic electoluminescence device of the present invention is such that n is 2 in the general formula (2), and two FL are the general formulas (2a) to (2d) and ( 2 'b), which is expressed by a shift and is more preferable if they are different from each other.
- G 1 in the general formula (2) is a substituted or unsubstituted aromatic ring group having 6 to 40 carbon atoms.
- a substituted or unsubstituted arylene amino group having 6 to 40 carbon atoms a substituted or unsubstituted diaminoaryl group having 6 to 60 carbon atoms, a substituted or unsubstituted triaminoaryl group having 6 to 60 carbon atoms, A substituted or unsubstituted heterocyclic group having 3 to 40 carbon atoms, or a substituted or unsubstituted etylene group.
- G 1 includes a single bond.
- G 1 is preferably substituted or unsubstituted phenylene, biphenylene, naphthacene, anthresene, etylene, fluorene, thiophene, thiadiazole, pyrazine, ammine, arylenoamine, triaryleneamine, carbazole, pyrrole, thiazole. , Benzothiazol, benzothiadiazole, phenanthral, quinoline, and quinoxaline.
- G 1 is preferably selected from divalent or trivalent groups having the following structure.
- n 2 to 4
- FL is a monovalent group derived from a compound having a structure represented by the following general formula (2′b).
- X ⁇ X 12 each independently represents a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic alkyl group having 1 to 30 carbon atoms, a substituted Or an unsubstituted linear, branched or cyclic alkoxy group having 1 to 30 carbon atoms, a substituted or unsubstituted linear, branched or cyclic alkylthio group having 1 to 30 carbon atoms, substituted or Is an unsubstituted linear, branched or cyclic alkenyl group having 2 to 30 carbon atoms, a substituted or unsubstituted linear, branched or cyclic alkenyloxy group having 2 to 30 carbon atoms, substituted or unsubstituted.
- C ⁇ R group (wherein R 2e is a hydrogen atom, a substituted or unsubstituted linear, branched or cyclic carbon atom) Number 1 to 30 alkyl groups, substituted or unsubstituted Substituted straight, branched or cyclic alkenyl groups having 2 to 30 carbon atoms, substituted or unsubstituted aralkyl groups having 7 to 30 carbon atoms, substituted or unsubstituted aryl groups having 6 to 30 carbon atoms , Or an amino group), OC0R 3e group (wherein R 3e is a substituted or unsubstituted straight chain, branched or cyclic alkyl group having 1 to 30 carbon atoms, substituted or unsubstituted straight chain) Branched or cyclic alkenyl group having 2 to 30 carbon atoms, substituted or unsubstituted straight chain) Branched or cyclic alkenyl group having 2 to 30 carbon atoms,
- n 2 to 4
- FL is a monovalent group derived from a compound having a structure represented by the following general formula (2'b).
- ⁇ to X 12 are each independently a hydrogen atom, a halogen atom, a substituted or unsubstituted linear, branched or cyclic alkyl group having 1 to 30 carbon atoms.
- X ⁇ X 16 location substituent of P contact groups and each group It may be bonded to each other to form a substituted or unsubstituted carbon ring.
- G 1 is one selected from a single bond and a group having the following structure.
- specific examples of X ⁇ X 12 are the same as those described above.
- the organic EL device material of the present invention has a fluoranthene structure represented by the general formula (2)
- the condensed ring-containing compound used together with the compound is represented by the following general formula (1).
- G 2 is a condensed ring-containing compound, and is a substituted or unsubstituted anthracene structure, a substituted or unsubstituted pyrene structure, a substituted or unsubstituted amine structure, or a substituted or unsubstituted benzene structure. It is a compound which has this.
- G 2 is preferably the following substituted or unsubstituted anthracene structure.
- a 1 and A 2 are each independently a substituted or unsubstituted condensed aromatic ring group having 10 to 20 nuclear carbon atoms.
- the substituted or unsubstituted condensed aromatic ring group of A 1 and A 2 for example, 1 naphthyl group, 2 naphthyl group, 1 anthryl group, 2 anthryl group, 9 anthryl group, 1 phenanthryl Group, 2 phenanthryl group, 3 phenanthryl group, 4-phenanthryl group, 9 phenanthrinol group, 1-naphthacenyl group, 2 naphthacenyl group, 9-naphthacenyl group, 1-pyrenyl group, 2-pyrenyl group, 4-pyrenyl group, 3-methyl- Examples include 2-naphthyl group, 4-methyl-1 naphthyl group, 4-methyl-1 anthryl group and the like.
- 1 naphthyl group, 2-naphthyl group, and 9 phenanthryl group are preferable.
- Ar 1 and Ar 2 in the general formula (3a) are each independently a hydrogen atom or a substituted or unsubstituted aromatic ring group having 6 to 50 nuclear carbon atoms.
- Examples of the substituted or unsubstituted aromatic ring group of Ar 1 and Ar 2 include a phenyl group, 1-naphthyl group, 2_naphthyl group, 1_anthryl group, 2 anthrinol group, 9_anthryl group, 1 —Phenanthryl group, 2-phenanthryl group, 3-phenanthryl group, 4-phenanthryl group Group, 9 phenanthrinol group, 1 naphthacenyl group, 2 naphthacenyl group, 9 naphthacenyl group, 1-pyrenyl group, 2-pyrenyl group, 4-pyrenyl group, 2-biphenylyl group, 3-biphenylyl group, 4-biphenylyl group, p terphenyl 4-yl group, p terphenyl _ 3-yl group, p_ terphenyl _ 2-yl group, m_ terphenyl _4-yl group,
- a phenyl group, a 1_naphthyl group, a 2_naphthyl group, a 9-phenanthryl group, a 1_naphthacenyl group, a 2_naphthacenyl group, a 9_naphthacenyl group, a 1-pyrenole group, and a 2-pyrenyl group are preferable.
- 4-pyrenyl group 2-biphenylyl group, 3-biphenylolinole group, 4-biphenylyl group, o-trinole group, m-tolyl group, p-trinole group, p-t-butynolephenyl group.
- R -R 10 ⁇ each independently a hydrogen atom, a substituted or unsubstituted aromatic ring group having 6 to 50 nuclear carbon atoms, a substituted or unsubstituted number of nuclear atoms 5 ⁇ 50 aromatic heterocyclic group, substituted or unsubstituted alkyl group having 1 to 50 carbon atoms, substituted or unsubstituted cycloalkyl group, substituted or unsubstituted alkoxy group having 1 to 50 carbon atoms, substituted or not Is an unsubstituted aralkyl group having 6 to 50 carbon atoms, a substituted or unsubstituted aryloxy group having 5 to 50 nucleus atoms, a substituted or unsubstituted aryloxy group having 5 to 50 nucleus atoms, a substituted or unsubstituted group.
- Examples of substituted or unsubstituted aromatic ring groups of ⁇ ! ⁇ include phenyl group, 1_naphthyl group, 2_naphthyl group, 1_anthryl group, 2_anthrinol group, 9_anthrinol group, 1-Phenanthryl group, 2-Phenanthryl group, 3-Phenanthryl group, 4-Phenanthryl group, 9_Phenanthrinol group, 1_Naphthenyl group, 2_Naphthenyl group, 9_Naphthenyl group, 1-Pyrenyl group, 2-Pyrenyl group 4-pyrenyl group, 2-biphenylyl group, 3-biphenyl group Ruyl group, 4-biphenylyl group, p terfeninore 4-yl group, p terfeninole 3-ynole group, p terfeninole 2-ino
- substituted or unsubstituted aromatic heterocyclic groups of ⁇ ! ⁇ Include 1_pyrrolyl group, 2—pyrrolyl group, 3_pyrrolinole group, pyradul group, 2_pyridinyl group, 3_pyridininole group, 4—pyridinyl group Group, 1_indolyl group, 2_indolyl group, 3_indolyl group, 4_indolinole group, 5_indolyl group, 6_indolyl group, 7_indolyl group, 1_isoindolyl group, 2-isoindolyl group, 3_ Isoindolyl group, 4_isoindolyl group, 5_isoindolyl group, 6_isoindolyl group, 7isoindolyl group, 2furyl group, 3furyl group, 2 benzofuranyl group, 3-benzofuranyl group, 4 monobenzo
- Examples of a substituted or unsubstituted alkyl group of ⁇ ⁇ to ⁇ 1 ⁇ include a methinole group, an ethyl group, a propyl group, an isopropyl group, a ⁇ -butyl group, an s-butynole group, an isobutyl group, a t Butyl group, n-pentyl group, n-hexyl group, n-heptyl group, n-octyl group, hydroxymethyl group, 1-hydroxyethyl group, 2-hydroxyethyl group, 2-hydroxyisobutyl group, 1 , 2-dihydroxyethyl, 1,3-dihydroxyisopropyl, 2,3-dihydroxy_t_butyl, 1,2,3_trihydroxypropyl, chloromethyl, 1_chloroethyl, 2 Diethyl, 2-chlorodiethyl, 1,2 Dichlorodiethyl, 1,
- Examples of substituted or unsubstituted cycloalkyl groups of 1 to! ⁇ include a pinole group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a 4-methylcyclohexinole group, a 1-adamantyl group, a 2-adamantyl group, a 1 norbornyl group, and a 2-nonolebornyl group.
- a substituted or unsubstituted alkoxy group of ⁇ ⁇ to ⁇ 1 ⁇ is a group represented by _ ⁇ , and examples of ⁇ include a substituted or unsubstituted alkyl group of the above 1 ⁇ to 1 ⁇ ° The same example can be given.
- Examples of ⁇ ⁇ to ⁇ 1 ⁇ substituted or unsubstituted aralkyl groups are benzyl, 1_phenylethyl, 2_phenylethyl, 1_phenylisopropyl, 2_phenylisopropyl, phenyl 1 t_butyl group, 1 naphthylmethyl group, 1 _ 1 naphthyltinol group, 2_ 1 naphthylethyl group, 1 _ 1 naphthylisopropyl group, 2_ 1 naphthylisopropyl group, ⁇ -naphthylmethyl group, 1 _ ⁇ -naphthylethyl group, 2_ ⁇ -naphthylethyl group, 1_ ⁇ -naphthylisopropyl group, 2_ ⁇ -naphthylisopropyl group, 1 pyrrolinoremethinole group, 2- (1
- ⁇ ⁇ to ⁇ ⁇ ⁇ 1 ⁇ substituted or unsubstituted aryloxy and allylothio groups are represented as OY 'and SY ", respectively.
- Examples of Y' and ⁇ " include 1 ⁇ to 1 ⁇ Examples are the same as those of the substituted or unsubstituted aromatic ring group of ° and the substituted or unsubstituted aromatic heterocyclic group of ⁇ ! ⁇ .
- ⁇ ⁇ ⁇ substituted or unsubstituted alkoxycarbonyl alkylsulfonyl group 1 ⁇ is expressed as _CO_ ⁇ _Z, examples of same alkyl groups of 1 to! ⁇
- Examples of the halogen atom for R 1 ! ⁇ include fluorine, chlorine, bromine and iodine. Preferably it is a fluorine atom.
- Substituents in the group represented by the following are halogen atoms, hydroxyl groups, nitro groups, cyano groups, alkyl groups, aryl groups, cycloalkyl groups, alkoxy groups, aromatic heterocyclic groups, aralkyl groups, aryloxy groups, and arylthio groups. , An alkoxycarbonyl group, or a carboxyl group.
- a plurality of R 9 and R 1Q may be in contact with each other to form a saturated or unsaturated cyclic structure.
- the G 2 is preferably a compound having a structure represented by the following general formula (3a ′ ′).
- Ar 1 and Ar 2 are each independently a substituted or unsubstituted aromatic ring group having 10 to 30 nuclear carbon atoms (excluding an anthracene residue).
- R ⁇ R 8 each independently represents a hydrogen atom, a substituted or unsubstituted aromatic ring group having 6 to 50 nuclear carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 5 to 50 nucleus atoms, a substituted or unsubstituted Unsubstituted alkyl group having 1 to 50 carbon atoms, substituted or unsubstituted cycloalkyl group, substituted or unsubstituted carbon number:!
- substituted or unsubstituted aralkyl group having 6 to 50 carbon atoms Substituted or unsubstituted aryloxy group having 5 to 50 nuclear atoms, substituted or unsubstituted aryloxy group having 5 to 50 nuclear atoms, substituted or unsubstituted alkoxycarbonyl group having 1 to 50 carbon atoms, substituted or An unsubstituted silyl group, a carboxyl group, a halogen atom, a cyano group, a nitro group or a hydroxyl group.
- Examples of the aromatic ring group having 10 to 30 nuclear carbon atoms of Ar 1 and Ar 2 in the general formula (3a ′ ′) (excluding an anthracene residue) include a substituted or unsubstituted mononaphthyl group and j3 —Naphthyl group, substituted or unsubstituted phenanthrenyl group, substituted or unsubstituted thali Cenyl group, substituted or unsubstituted tetracenyl group, substituted or unsubstituted pyrenyl group, substituted or unsubstituted phenylnaphthyl group, substituted or unsubstituted naphthylnaphthinole group, substituted or unsubstituted naphthylphenyl group, substituted or unsubstituted Substituted phenyl vinylene, substituted or unsubstituted pyrenyl phenyl,
- a substituted or unsubstituted mononaphthyl group and / 3-naphthyl group a substituted or unsubstituted phenyl naphthyl group, a substituted or unsubstituted naphthyl naphthyl group, or a substituted or unsubstituted naphthyl phenyl group is preferred.
- the unsubstituted aromatic ring group having 10 to 30 nuclear carbon atoms is preferably an aromatic residue containing a naphthalene residue.
- G 2 is preferably a compound having a structure represented by the following general formula (3a ′).
- a 1 and A 2 are each independently a hydrogen atom or a substituted or unsubstituted aromatic ring group having 6 to 50 nuclear carbon atoms.
- a 1 and 8 2 ! ⁇ ⁇ ⁇ .
- ⁇ ! ⁇ Is independently a hydrogen atom, a substituted or unsubstituted aromatic ring group having 6 to 50 nuclear carbon atoms, a substituted or unsubstituted aromatic heterocyclic group having 5 to 50 nuclear atoms, a substituted or unsubstituted Is an unsubstituted alkyl group having 1 to 50 carbon atoms, a substituted or unsubstituted cycloalkyl group, a substituted or unsubstituted alkoxy group having 1 to 50 carbon atoms, a substituted or unsubstituted aralkyl group having 6 to 50 carbon atoms, Substituted or unsubstituted aryloxy group having 5 to 50 nuclear atoms, substituted or unsubstituted aryloxy group having 5 to 50 nuclear atoms, substituted or unsubstituted alkoxycarbonyl group having 1 to 50 carbon atoms, substituted or unsubstituted A silyl group
- R 9 and R 1Q may be plural or adjacent to each other to form a saturated or unsaturated cyclic structure.
- a 1 , ⁇ 2 and R ⁇ R “ 1 are the same as those in the general formula (3a).
- the condensed ring-containing compound of G 2 represented by the general formula (1) has the following substitution Or an unsubstituted pyrene structure is preferred.
- Ar lb and Ar are each a substituted or unsubstituted aromatic group having 6 to 50 nuclear carbon atoms.
- aromatic group examples include phenyl group, 1_naphthyl group, 2_naphthyl group, 1-anthryl group, 2_anthryl group, 9_anthrinol group, 9_ (10-phenyl) anthryl group, 9_ (10-naphthinole _ 1 —yl) anthryl group, 9 _ (10-naphthyl -2-yl) anthrinol group, 1-phenanthryl group, 2-phenanthryl group, 3-phenanthryl group, 4-phenanthryl group, 9 1-phenanthrinol group, 1_naphthacenyl group, 2_naphthacenyl group, 9_naphthacenyl group, 1-pyrenyl group, 2-pyrenyl group, 4-pyrenyl group, 2-biphenylyl group, 3-biphenylyl group, 4-biphenylyl group, p-terphenyl 4-
- a phenyl group, a 1 naphthyl group, a 2-naphthyl group, a 9 (10-phenyl) anthryl group, a 9- (10-naphthyl-1-yl) anthryl group, and a 9- (10-naphthyl group are preferable.
- 2-yl) anthryl group 9_phenanthrinol group, 1-pyrenyl group, 2-pyrenylol group, 4-pyrenyl group, 2_biphenylyl group, 3_biphenylyl group, 4_biphenylyl group, o_tolyl group, m-tolyl group, ⁇ -trinole group, p_t-butylphenyl group and the like.
- the aromatic group may be further substituted with a substituent.
- a substituent for example, an alkyl group
- L in the general formula (3b) is a substituted or unsubstituted phenylene group, a substituted or unsubstituted naphthalenylene group, a substituted or unsubstituted fluorenylene group, or a substituted or unsubstituted dibenzosilolylene group, respectively.
- a substituted or unsubstituted phenylene group or A substituted or unsubstituted fluorenylene group is preferred.
- m is an integer of 0 to 2 (preferably 0 to: 1)
- nb is an integer of:! To 4 (preferably 1 to 2)
- s is 0 to 2 (preferably Is an integer from 0 to 1)
- t is an integer from 0 to 4 (preferably 0 to 2).
- L or Ar lb binds to one of 1- to 5-positions of pyrene
- L or Ar 2b is pyrene 6 of: binding to either the 10-position.
- L or pyrene are bonded to different bonding positions on Ar lb and Ar 2b , respectively.
- L or pyrene force are the same on Ar lb and Ar 2b. In the case of bonding at the bonding position, there is no case where L or Ar lb and Ar 2b are in the 6-position, or 2-position and 7-position in the pyrene position.
- fused ring-containing compound G 2 represented by the general formula (1) is preferably a Amin structure: substituted or unsubstituted.
- Ar lc , Ar 2e, and Ar 3e each independently represent a group having an anthracene structure, a phenanthrene structure, or a pyrene structure.
- R le , R 2c , and R each independently represents a hydrogen atom or a substituent.
- Ar le , Ar 2c , Ar 3e are substituted or unsubstituted anthryl phenyl group, anthryl group, phenanthrenyl group, and pyrenyl group, preferably alkyl-substituted or unsubstituted anthryl.
- Pyrenyl and phenanthryl groups are particularly preferred, with phenyl, phenanthryl and pyrenyl being more preferred.
- the substituent represented by R le , R 2e and R 3e is an alkyl group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 10 carbon atoms).
- alkyl group preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 10 carbon atoms.
- an alkenyl group preferably having 2 to 30, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 10 carbon atoms, such as buryl, allyl, 2-butyl, 3_pentenyl, etc.
- alkynyl group preferably carbon number) 2 to 30, more preferably 2 to 20 carbon
- An aryloxy group (preferably having 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, particularly preferably 6 to 12 carbon atoms, and examples thereof include phenyloxy, 1 naphthyloxy, and 2 naphthyloxy. ), Heteroaryloxy groups (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms, such as pyridinoreoxy, pyrazyloxy, pyrimidyloxy, quinolyloxy and the like.
- an acyl group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 12 carbon atoms, and examples thereof include acetyl, benzoyl, formyl, and pivalol. ), An alkoxycarbonyl group (preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 12 carbon atoms, In methoxycarbonyl, and the like ethoxycarbonyl.), ⁇ reel O alkoxycarbonyl group (preferably having 7 to 30 carbon atoms, more preferably Or having 7 to 20 carbon atoms, particularly preferably 7 to 12 carbon atoms, and examples thereof include phenylcarbonyl.
- An acyloleoxy group (preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 10 carbon atoms, such as acetooxy, benzoyloxy, etc.), an acyloleamino group (preferably ) Has 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, and particularly preferably 2 to 10 carbon atoms, and examples thereof include acetylamino benzoylamino).
- An alkoxycarbonylamino group (preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 12 carbon atoms, and examples thereof include methoxycarbonylamino. ),
- An aryloxycarbonylamino group (preferably having a carbon number of 7 to 30, more preferably a carbon number of 7 to 20, particularly preferably a carbon number of 7 to 12; for example, phenylcarbonylcarbonylamino, etc.
- a sulfonylamino group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 12 carbon atoms.
- a sulfamoyl group (preferably having 0 to 30 carbon atoms, more preferably 0 to 20 carbon atoms, and particularly preferably 0 to 12 carbon atoms, such as sulfamoyl, methyl Amoyl, dimethylsulfamoyl, phenylsulfamoyl, etc.), a strong rubamoyl group (preferably having a carbon number of 1-30, more preferably having a carbon number of 1-20, particularly preferably having a carbon number of 1-12: Strength rubamoyl, methyl strength rubamoyl, jetylcarbamoyl, phenylcarbamoyl, etc.),
- An alkylthio group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 12 carbon atoms, such as methylthio, ethylthio and the like),
- a monoruthio group (preferably having 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, particularly preferably 6 to 12 carbon atoms, such as phenylthio), heteroarylthio group (preferably 1-30 carbon atoms, more preferably 1-20 carbon atoms, particularly preferably 1-12 carbon atoms, such as pyridylthio, 2_benzimidazolylthio, 2_benzoxazolinolethio, 2_benzthiazolyl And sulfonyl group (preferably carbon number:!
- Rufiniru group preferably having 1 to 30 carbon atoms, and more favorable Mashiku 1 to 20 carbon atoms, particularly preferably 1 to carbon atoms: is 12, for example methanesulfinyl And benzenesulfiel.
- a ureido group preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms, and examples thereof include ureido, methenoureido, and phenolenoureido).
- a phosphoric acid amide group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms, and examples thereof include jetyl phosphoric acid amide and phenylphosphoric acid amide), hydroxy Group, mercapto group, halogen atom (eg fluorine atom, chlorine atom, bromine atom, iodine atom), cyano group, sulfo group, carboxyl group, nitro group, hydroxamic acid group, sulfino group, hydrazino group, imino group, heterocyclic group (Preferably having 1 to 30 carbon atoms, more preferably 1 to 12 carbon atoms.
- halogen atom eg fluorine atom, chlorine atom, bromine atom, iodine atom
- cyano group eg fluorine atom, chlorine atom, bromine atom, iodine atom
- cyano group eg
- hetero atom examples include a nitrogen atom, an oxygen atom, a sulfur atom, specifically, for example, imidazolyl, pyridyl, quinolyl, furyl, and chenyl. , Piperidinole, morpholino, benzoxazolyl, benzimidazolyl, benzthiazolyl, etc.) And silyl groups (preferably having 3 to 40 carbon atoms, more preferably 3 to 30 carbon atoms, and particularly preferably 3 to 24 carbon atoms, and examples thereof include trimethylsilyl and triphenylsilyl). These substituents may be further substituted.
- R le , R and R 3e are preferably alkyl groups or aryl groups.
- the fused ring-containing compound of G 2 represented by the general formula (1) preferably has the following substituted or unsubstituted benzene structure.
- Ar ", Ar 21 and Ar 31 each represent an arylene group, and Ar 12 , Ar 22 and Ar 32 each independently represent a substituent or a hydrogen atom.
- Ar Ar 21 , Ar 31 , Ar At least one of 12 , Ar 22 , and Ar 32 has a condensed ring reel structure or a condensed ring hetero reel structure.
- Ar represents an arylene group or a heteroarylene group
- Ar 11 , Ar 21 and Ar 31 each represent an arylene group.
- the carbon number of the arylene group is preferably 6-30, more preferably 6-20 force S, and more preferably 6-: 16.
- Masle As the arylene group, for example, a phenylene group, a naphthylene group, an anthrylene group, a phenanthrenylene group, a pyrenylene group, a peryleneylene group, a fluorenylene group, a biphenylene group, a terfenylene group, a rubrenylene group, a chrysylene group , Triphenylenylene group, benzoanthrylene group, benzophenanthrenylene group, diphenylanthrylene group and the like, and these arylene groups may further have a substituent.
- Examples of the substituent on the arylene group include an alkyl group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 10 carbon atoms such as methyl, Til, isopropyl, t-butyl, n_octyl, n-decyl, n_hexadecyl, cyclopropyl, cyclopentyl, cyclohexyl, etc.), alkenyl group (preferably having 2 to 30 carbon atoms, more preferably Has 2 to 20 carbon atoms, particularly preferably 2 to 10 carbon atoms, and examples thereof include bur, allyl, 2-butur, 3_pentur, and the like, alkynyl groups (preferably 2 to 30 carbon atoms).
- alkyl group preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably 1 to 10 carbon atoms such as methyl, Til, isopropyl
- To 10 and examples thereof include methoxy, ethoxy, butoxy, 2-ethylhexyloxy, and the like.
- it has 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, particularly preferably 6 to 12 carbon atoms, such as phenyloxy, 1_naphthylo And a heteroaryloxy group (preferably carbon number:! -30, more preferably carbon number 1-20, particularly preferably carbon number 1-12: Pyridyloxy, pyrazyloxy, pyrimidyloxy, quinolyloxy and the like.
- an acyl group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, and particularly preferably carbon number:! To 12).
- Examples include acetyl, benzoyl, formyl, bivaloyl and the like. It is. ),
- An alkoxycarbonyl group (preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 12 carbon atoms, and examples thereof include methoxycarbonyl and ethoxycarbonyl), aryl.
- An alkoxycarbonyl group (preferably having 7 to 30 carbon atoms, more preferably 7 to 20 carbon atoms, particularly preferably 7 to 12 carbon atoms, such as phenylcarbonyl), an acyloxy group (preferably Has 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 10 carbon atoms, and examples thereof include acetoxy and benzoyloxy), an acylamino group (preferably 2 to 30 carbon atoms, More preferably, it has 2 to 20 carbon atoms, particularly preferably 2 to 10 carbon atoms, and examples thereof include acetylamino-containing benzoylamino.
- Lucoxycarbonylamino group (preferably having 2 to 30 carbon atoms, more preferably 2 to 20 carbon atoms, particularly preferably 2 to 12 carbon atoms, such as methoxycarbonylamino).
- Reyloxycarbonylamino group (preferably having 7 to 30 carbon atoms, more preferably having 7 to 20 carbon atoms, particularly preferably 7 to 12 carbon atoms, and examples thereof include phenylcarbonylcarbonylamino. )
- a sulfonylamino group (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms, and examples thereof include benzenesulfonylamino with methanesulfonylamino.
- a sulfamoyl group (preferably having 0 to 30 carbon atoms, more preferably 0 to 20 carbon atoms, particularly preferably 0 to 12 carbon atoms, such as sulfamoyl, methylsulfamoyl, di- Tilsulfamoyl, phenylsulfamoyl, etc.), rubamoyl groups (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms, Rubamoyl, methylcarbamoyl, jetylcarbamoyl, phenyl-powered rubamoyl, etc.), alkylthio groups (preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms): 12 And, for example, methylthio, ethylthio, etc.), arylthio group (preferably having 6 to 30 carbon atoms, more s
- Heteroarylthio group preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms, such as pyridylthio, 2_benzimidazolylthio, 2_benzoxa Zolylthio, 2_benzthiazolylthio, etc.
- sulfonyl A group preferably having 1 to 30 carbon atoms, more preferably 1 to 20 carbon atoms, particularly preferably 1 to 12 carbon atoms such as mesyl, tosyl, etc.
- a sulfier group preferably carbon number:!
- ureido groups preferably 1 carbon atoms.
- Ar ", at least one of Ar 21, Ar 31, Ar 12 , Ar ⁇ and Ar 32 are Teroariru structure to condensed Ariru structure or condensed ring.
- Ar 11, Ar 21, Ar 31, Ar Preferably, at least one of 12 , ⁇ ⁇ and Ar 32 has a condensed ring reel structure.
- the condensed ring reel structure is preferably a naphthalene structure, anthracene structure, phenanthrene structure, pyrene structure, perylene structure, more preferably a naphthalene structure, anthracene structure, pyrene structure, or a phenanthrene structure.
- Preferred are a phenanthrene structure and an aryl structure having four or more rings, and particularly preferred is a pyrene structure.
- the condensed heteroaryl structure is preferably a quinoline structure, a quinoxaline structure, a quinazoline structure, an atalidine structure, a phenanthridine structure, a phthalazine structure, or a phenantorin structure, and more preferably a quinoline structure, a quinoxaline structure, Quinazoline structure Talazine structure and phenant mouth phosphorus structure.
- the Ar is an arylene group which is a trivalent group (preferably having 6 to 30 carbon atoms, more preferably 6 to 20 carbon atoms, still more preferably 6 to 16 carbon atoms, for example, a phenylene group, a naphthylene group, an anthracite group).
- a selenylene group, a phenanthrene group, a pyrenylene group, a triphenylene group, etc.), a heteroarylene group (a hetero atom is preferably a nitrogen atom, a sulfur atom, an oxygen atom, more preferably a nitrogen atom, preferably a carbon number of 2 to More preferably 30 to 20 carbon atoms, still more preferably 3 to 16 carbon atoms, for example, pyridylene group, pyrazylene group, thiobutyleneylene group, quinolylene group, quinoxarylene group, triadylene group, etc.)
- the group may have a substituent. Examples of the substituent include the groups described for the substituent on Ar 11 .
- Ar is a trivalent group, each of which is a phenylene group (benzenetriyl), a naphthylene group (naphthalenetriyl), an anthracenylene group (anthracentriyl), a pyrenylene group (pyrenetriyl), or a triphenylene group. More preferred is a phenylene group, and more preferred is an unsubstituted (Ar 11 , Ar 21 , Ar 31 is substituted) phenylene group or an alkyl-substituted phenylene group. .
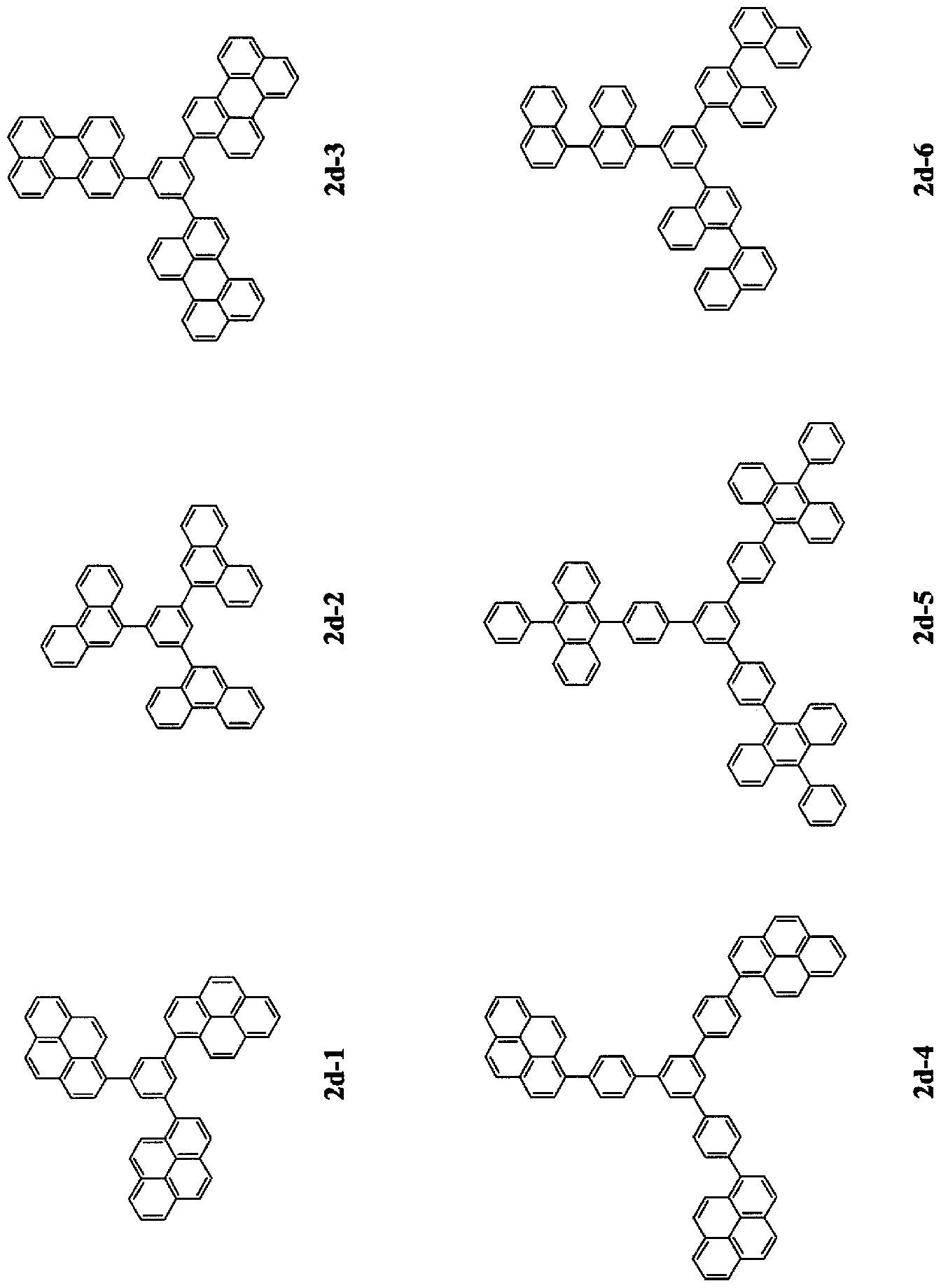
- the fused ring-containing compound of G 2 represented by the general formula (1) is at least one selected from compounds represented by the following general formulas (3 ′ a) to (3 ′ d): preferable.
- Ar la and Ar 2a are each independently a group derived from a substituted or unsubstituted aromatic ring having 6 to 20 nuclear carbon atoms, wherein the aromatic ring is 1 or
- the substituent may be substituted with two or more substituents, which are substituted or unsubstituted aryl groups having 6 to 50 nuclear carbon atoms, substituted or unsubstituted alkyl groups having 1 to 50 carbon atoms, substituted or unsubstituted.
- the aromatic ring is substituted with two or more substituents, the substituents may be the same or different.
- the substituents in contact with P are bonded to each other to form a saturated or unsaturated cyclic structure
- R la to R 8a are each independently a hydrogen atom, a substituted or unsubstituted aryleno group having 6 to 50 nuclear carbon atoms, a substituted or unsubstituted heteroaryl group having 5 to 50 nuclear atoms, a substituted or unsubstituted group.
- Substituted alkyl group having 1 to 50 carbon atoms substituted or unsubstituted alkyl group having 3 to 50 carbon atoms, substituted or unsubstituted alkoxy group having 1 to 50 carbon atoms, substituted or unsubstituted carbon atoms 6 to 50 aralkyl groups, substituted or unsubstituted aryloxy groups having 5 to 50 nuclear atoms, substituted or unsubstituted aryloxy groups having 5 to 50 nuclear atoms, substituted or unsubstituted 1 to 50 carbon atoms It is selected from an alkoxycarbonyl group, a substituted or unsubstituted silyl group, a carboxyl group, a halogen atom, a cyano group, a nitro group and a hydroxy group.
- Ar lb and Ar 2b are each independently a substituted or unsubstituted aryl group having 6 to 50 nuclear carbon atoms.
- L and L are each independently a substituted or unsubstituted phenylene group, substituted or unsubstituted
- It is selected from an unsubstituted naphthalenylene group, a substituted or unsubstituted fluorenylene group, and a substituted or unsubstituted dibenzosilolylene group.
- L or Ar lb is bound to any of the 1-5 positions of pyrene, and L or Ar 2b is 6
- the group is selected from a group having an anthracene structure, a group having a phenanthrene structure, a group having a pyrene structure, and a group having a perylene structure.
- R le , R 2 and R 3e each independently represent a hydrogen atom or a substituent.
- Ar ld , Ar 2d and Ar 3d each independently represent an aryl group having 6 to 50 nuclear carbon atoms, wherein the aryl group is one or more substituents. May be substituted.
- Ar ld , Ar 2d , Ar 3d and at least one of the substituents of these aryl groups have a condensed ring structure having 10 to 20 nuclear carbon atoms or a condensed ring structure having 6 to 20 nuclear carbon atoms.
- Ar represents a trivalent group derived from an aromatic ring or a heteroaromatic ring.
- a specific f row of the anthracene derivative of G 2 represented by the general formula (3a) used in the organic EL device of the present invention is disclosed in Japanese Patent Application Laid-Open No. 2004-356033 [0043] to [0063].
- various known anthracene derivatives such as compounds having two anthracene skeletons in the molecule and compounds having one anthracene skeleton shown on pages 27 to 28 of International Publication WO2005 / 061656. it can.
- a typical example is shown below.
- the f column of the ammine derivative of G 2 represented by the general formula (3c) used in the organic EL device of the present invention is disclosed in Japanese Patent Application Laid-Open No. 2002-324678. [0079] to [0083]
- the following amine derivatives can also be used as the organic EL device material of the present invention.
- the organic EL device emitting layer of the present invention the compound having a fluoranthene structure, 0.01 to 20 weight 0/0, preferably from 0.5 to 20 weight 0/0, is contained Rereru.
- a chalcogenide layer, a metal halide layer or a metal oxide layer is provided on at least one surface of a pair of electrodes.
- the organic electoluminescence material-containing solution of the present invention comprises an organic electoluminescence material and a solvent, and the organic electoluminescence material is a host material and a dopant material, and the dopant material has the general formula It is at least one of compounds having a fluoranthene structure represented by (2), and the host material is represented by the general formulas (3a), (3a,), (3b) to (3d), (3′a) to ( Containing condensed rings represented by 3 'd), (3a'') and (1) It is at least one selected from a compound.
- the configuration (8) is preferably used.
- the compound of the present invention may be used in any of the organic layers described above, but is preferably contained in the light emission band of these constituents.
- the organic EL device of the present invention is manufactured on a light-transmitting substrate.
- the translucent substrate referred to here is a substrate that supports the organic EL element, and is preferably a smooth substrate having a light transmittance in the visible region of 400 to 700 nm of 50% or more.
- a glass plate, a polymer plate, etc. are mentioned.
- the glass plate include soda-lime glass, norlium strontium-containing glass, lead glass, aluminosilicate glass, borosilicate glass, norium borosilicate glass, and quartz.
- a polymer plate May include polycarbonate, acrylic, polyethylene terephthalate, polyethersulfide, polysulfone, and the like.
- the anode of the organic EL device of the present invention plays a role of injecting holes into the hole transport layer or the light emitting layer, and it is effective to have a work function of 4.5 eV or more.
- Specific examples of the anode material used in the present invention include indium tin oxide alloy (ITO), tin oxide (NESA), gold, silver, platinum, copper and the like.
- the anode is preferably a material having a small work function for the purpose of injecting electrons into the electron transport layer or the light emitting layer.
- the anode can be produced by forming a thin film of these electrode materials by a method such as vapor deposition or sputtering.
- the transmittance of the anode for light emission is greater than 10%.
- the sheet resistance of the anode is preferably several hundred ⁇ or less.
- the film thickness of the anode is a force depending on the material. Usually, it is selected in the range of 1011111 to 1/1111, preferably 10 to 200 nm.
- the light emitting layer of the organic EL device has the following functions. That is,
- Injection function Function that can inject holes from the anode or hole injection layer when an electric field is applied, and can inject electrons from the cathode or electron injection layer
- Transport function Function to move injected charges (electrons and holes) by the force of electric field
- Luminous function provides a field for recombination of electrons and holes, and connects it to light emission.
- ease of hole injection and the ease of electron injection, and the transport capability represented by the mobility of holes and electrons may be large or small. I prefer to move the electric charge.
- the light emitting layer is particularly preferably a molecular deposition film, where the molecular deposition film is a thin film formed by deposition from a gas phase material or a material in a solution state or a liquid phase state.
- a film formed by solidification from Usually, this molecular deposited film can be distinguished from the thin film (molecular accumulated film) formed by the LB method by the difference in aggregated structure and higher-order structure and the functional difference resulting from it.
- a binder such as a resin and a material compound are dissolved in a solvent to form a solution, which is then thin-filmed by a spin coating method or the like. Also, the light emitting layer can be formed.
- a known light emitting material other than the light emitting material comprising the compound having the fluoranthene structure of the present invention and the condensed ring-containing compound may be included in the light emitting layer as desired.
- a light emitting layer containing another known light emitting material may be laminated on the light emitting layer containing the light emitting material of the present invention.
- the thickness of the light emitting layer is preferably 5 to 50 nm, more preferably 7 to 50 nm, and most preferably 10 to 50 nm. If the thickness is less than 5 nm, it is difficult to form a light emitting layer, and it may be difficult to adjust the chromaticity. If it exceeds 50 nm, the driving voltage may increase.
- the hole injection 'transport layer is a layer that helps injecting holes into the light emitting layer and transports them to the light emitting region, and has a high ion mobility with a high hole mobility, usually less than 5.5 eV.
- a material that transports holes to the light emitting layer with a lower electric field strength is preferable.
- the mobility force of holes is, for example, 10 4 to: 10 6 V / cm. when an electric field is applied, if the least even 10- 4 cm 2 / V ⁇ a second preferred Rere.
- the aromatic amine derivative of the present invention when used in a hole transport zone, the aromatic amine derivative of the present invention alone may be used as a hole injection or transport layer, or may be mixed with other materials. Les.
- the material for forming the hole injection / transport layer by mixing with the aromatic amine derivative of the present invention is not particularly limited as long as it has the above-mentioned preferred properties.
- a material that is commonly used as a transport material or a known medium force used for a hole injection / transport layer of an organic EL device can be selected and used.
- the above-mentioned materials can be used as the material for the hole injection / transport layer.
- Volphiline compounds (disclosed in JP-A-63-29556965, etc.), aromatic tertiary amine compounds And styrylamine compounds (US Pat. No. 4,127,412, JP-A-53-27033, 54-58445, 54-149634, 54-64299, 55 — See 79450, 55-144250, 56-119132, 61-295558, 61-98353, 63-295695, etc.), especially aromatic third It is preferable to use a secondary amine compound.
- NPD N-(2-naphthyl) -N-phenylamino) biphenol having two condensed aromatic rings described in US Pat. No. 5,061,569 in the molecule.
- Ninore hereinafter abbreviated as NPD
- three triphenylamine units described in JP-A-4-308688 are connected in a starburst type 4, 4, 4, 4 ', one tris (N — (3-Methylphenyl) -N-phenylamino) triphenylamine (hereinafter abbreviated as MTDATA).
- inorganic compounds such as P-type Si and p-type SiC can also be used as the material for the hole injection layer.
- the hole injecting / transporting layer can be formed by thin-filming the above-described compound by a known method such as a vacuum deposition method, a spin coating method, a casting method, or an LB method.
- the thickness of the hole injection / transport layer is not particularly limited, but is usually 5 nm to 5 / im.
- This hole injection 'transport layer may be composed of one or more of the above-mentioned materials as long as it contains the compound of the present invention in the hole transport zone, or the hole
- the hole injection / transport layer made of a compound different from the injection / transport layer may be laminated.
- a layer that assists hole injection or electron injection into the light emitting layer and has a conductivity of 10-1Q S / cm or more is preferable.
- the material for such an organic semiconductor layer include thiophene oligomers, conductive oligomers such as allylamin oligomers disclosed in JP-A-8-193191, and conductive materials such as allylamin dendrimers. Sex dendrimers and the like can be used.
- Electron injection layer is a layer that assists the injection of electrons into the light emitting layer and has a high electron mobility
- the adhesion improving layer is a layer made of a material that has a particularly good adhesion to the cathode among the electron injection layers. is there.
- As a material used for the electron injection layer 8-hydroxyquinoline or a metal complex of its derivative is suitable.
- metal complex of the above 8-hydroxyquinoline or a derivative thereof include metal chelate oxinoid compounds containing a chelate of oxine (generally 8_quinolinol or 8-hydroxyquinoline).
- Alq described in the section of the light emitting material can be used as the electron injection layer.
- examples of the oxadiazole derivative include an electron transfer compound represented by the following general formula.
- Ar 1 , Ar 2 , Ar 3 , Ar 5 , Ar 6 , Ar 9 each represents a substituted or unsubstituted aryl group, which may be the same or different from each other.
- Ar 7 and Ar 8 each represent a substituted or unsubstituted arylene group, which may be the same or different.
- the aryl group includes a phenyl group, a biphenyl group, an anthranyl group, a perylenyl group, A pyrenyl group is mentioned.
- Examples of the arylene group include a phenylene group, a naphthylene group, a biphenylene group, an anthranylene group, a peryleneylene group, and a pyrenylene group.
- Examples of the substituent include an alkyl group having 1 to 10 carbon atoms, an alkoxy group having 1 to 10 carbon atoms, and a cyan group. This electron transfer compound is preferably a thin film-forming compound.
- electron transport compound include the following.
- a preferred form of the organic EL device of the present invention is a device containing a reducing dopant in a region for transporting electrons or an interface region between the cathode and the organic layer.
- the reducing dopant is defined as a substance capable of reducing the electron transporting compound. Accordingly, various materials can be used as long as they have a certain reducibility, such as alkali metals, alkaline earth metals, rare earth metals, alkali metal oxides, alkali metal halides, alkaline earth metals.
- At least one substance can be preferably used.
- preferable reducing dopants include Na (work function: 2.36 eV), K (work function: 2.28 eV), Rb (work function: 2.16 eV), and Cs (work function: 1). 95 eV), at least one alkali metal selected from the group consisting of Ca (work function: 2.9 eV), Sr (work function: 2.0 to 2.5 eV), and Ba (work function: 2.52 eV) Particularly preferred are those having a work function of 2.9 eV or less, including at least one alkaline earth metal selected from the group consisting of: Among these, a more preferable reducing dopant is at least one alkali metal selected from the group consisting of K, Rb and Cs, more preferably Rb or Cs, and most preferably Cs.
- alkali metals in particular, can improve the emission brightness and extend the life of organic EL devices by adding a relatively small amount to the electron injection region where the reducing ability is high.
- reducing dopa with a work function of 2.9 eV or less in particular, combinations of these two or more alkali metals are also preferred, especially combinations containing Cs, such as Cs and Na, Cs and K, Cs and Rb, or Cs and Na and ⁇ . Is preferred.
- Cs such as Cs and Na, Cs and K, Cs and Rb, or Cs and Na and ⁇ .
- an electron injection layer composed of an insulator or a semiconductor may be further provided between the cathode and the organic layer.
- an insulator it is preferable to use at least one metal compound selected from the group consisting of alkali metal chalcogenides, alkaline earth metal chalcogenides, alkali metal halides, and alkaline earth metal halides. Ms. If the electron injection layer is composed of these alkali metal chalcogenides or the like, it is preferable in that the electron injection property can be further improved.
- preferable alkali metal chalcogenides include, for example, LiO, K 0, Na S, Na Se and Na 2 O
- preferable alkaline earth metal chalcogenides include, for example, CaO, BaO, SrO, BeO. , BaS, and CaSe
- Preferred alkali metal halides include, for example, LiF, NaF, KF, LiCl, KC1, and NaCl
- preferred alkaline earth metal halides include fluorides such as CaF, BaF, SrF, MgF and BeF, and halides other than fluorides.
- the inorganic compound constituting the electron transport layer is preferably a microcrystalline or amorphous insulating thin film. If the electron transport layer is composed of these insulating thin films, a more uniform thin film is formed, so that pixel defects such as dark spots can be reduced. Examples of such inorganic compounds include the alkali metal chalcogenides, alkaline earth metal chalcogenides, alkali metal halides, and alkaline earth metal halides described above.
- a metal, an alloy, an electrically conductive compound having a low work function (4 eV or less), and a mixture thereof are used as an electrode material.
- electrode materials include sodium, sodium / potassium alloys, magnesium, lithium, magnesium'silver alloys, aluminum / anolymium oxide, aluminum'lithium alloys, indium, rare earth metals, and the like.
- This cathode can be produced by forming a thin film of these electrode materials by a method such as vapor deposition or sputtering.
- the transmittance of the cathode for light emission is greater than 10 ° / o.
- the sheet resistance as the cathode is preferably several hundred ⁇ / mouth or less, and the film thickness is usually 10 nm 1 ⁇ m, preferably 50 200.
- organic EL devices apply an electric field to ultra-thin films, pixel defects are likely to occur due to leaks and shorts. In order to prevent this, it is preferable to insert an insulating thin film layer between the pair of electrodes.
- Examples of materials used for the insulating layer include aluminum oxide, lithium fluoride, lithium oxide, cesium fluoride, cesium oxide, magnesium oxide, magnesium fluoride, oxidizing power, subsequentlyium, calcium fluoride, aluminum nitride, titanium oxide, Examples thereof include silicon oxide, germanium oxide, silicon nitride, boron nitride, molybdenum oxide, ruthenium oxide, and vanadium oxide.
- a mixture or laminate of these may be used.
- An organic EL device can be produced by forming an anode, a light emitting layer, a hole injection layer as necessary, and an electron injection layer as needed, and further forming a cathode by the materials and formation methods exemplified above. it can.
- organic EL elements can be fabricated in the reverse order from the cathode to the anode.
- an organic EL element having a structure in which an anode, a hole injection layer, a light emitting layer, a electron injection layer, and a cathode are sequentially provided on a light transmitting substrate
- a thin film having an anode material strength is formed on a suitable translucent substrate by a method such as vapor deposition or sputtering so as to have a film thickness of 1 ⁇ m or less, preferably 10 to 2 OO nm.
- a hole injection layer is provided on the anode.
- the hole injection layer can be formed by a vacuum deposition method, a spin coating method, a casting method, an LB method, or the like, but a homogeneous film can be obtained immediately and pinholes are generated. It is preferable to form it by a vacuum evaporation method from the point of being hard to do.
- the deposition conditions vary depending on the compound used (the material of the hole injection layer), the crystal structure of the target hole injection layer, the recombination structure, etc. deposition source temperature 50 to 450 ° C, vacuum degree of 10- 7 ⁇ : 10- 3 Torr, the deposition rate of 0. 01 ⁇ 50nm / sec, a substrate temperature of - 50 to 300 ° C, in the range of thickness of 5 nm to 5 mu m It is preferable to select appropriately.
- a light-emitting layer in which a light-emitting layer is provided on a hole injection layer is also performed using a desired organic light-emitting material by a method such as vacuum deposition, sputtering, spin coating, or cast.
- a vacuum deposition method from the viewpoint that a homogeneous film can be obtained immediately and pinholes are hardly generated.
- the deposition condition varies depending on the compound used, but in general, the medium range of conditions similar to the hole injection layer can be selected.
- the light emitting layer of the EL device of the present invention can be formed by a wet process in which the light emitting material is not only deposited.
- Each organic layer of the light emitting layer can be formed by a dry film forming method such as vacuum deposition, sputtering, plasma, ion plating, or a coating method such as spin coating, dating, casting, bar coating, roll coating, flow coating, or ink jet. Applying power S.
- a material for forming each layer is dissolved or dispersed in an appropriate solvent to prepare a light-emitting organic solution to form a thin film, and any solvent may be used.
- the solvent include halogenated hydrocarbon solvents such as dichloromethane, dichloroethane, chlorophenol, carbon tetrachloride, tetrachloroethane, trichloroethane, chlorobenzene, dichlorobenzene, chlorotolenene, trichlorotolenene, and dibutyl ether.
- Ether solvents such as tetrahydrofuran, tetrahydropyran, dioxane, anisole, dimethoxyethane, methanol, ethanol, propanol, isopropanol, butanol, Alcohol solvents such as pentanomonole, hexanol, cyclohexanol, methinoreserosonoleb, ethinorecerosolev, ethylene glycol, acetone, methyl ethyl ketone, ethyl ketone, 2-hexanone, methyl isobutyl ketone , 2-heptanone, 4-heptanone, diisobutyl ketone, acetonyl acetone, isophorone, cyclohexanone, methyl hexanone, ketone solvent such as acetophenone, benzene, toluene, xylene, ethynolebenzene,
- an electron injection layer is provided on the light emitting layer. Similar to the hole injection layer and the light emitting layer, the necessary power to obtain a homogeneous film is preferably formed by vacuum evaporation.
- the vapor deposition conditions can be selected from the same condition ranges as those for the hole injection layer and the light emitting layer.
- the compound of the present invention differs depending on which layer in the light emission band or the hole transport band is contained, but when the vacuum evaporation method is used, it can be co-deposited with other materials. Moreover, when using a spin coat method, it can be contained by mixing with other materials.
- a cathode can be stacked to obtain an organic EL device.
- the cathode is made of metal, and vapor deposition or sputtering can be used. In order to protect the underlying organic layer from damage during film formation, vacuum deposition is preferred. For the organic EL device described so far, it is preferable to produce the anode negative electrode consistently with a single vacuum.
- the method for forming each layer of the organic EL device of the present invention is not particularly limited. Conventionally known methods such as vacuum deposition and spin coating can be used.
- the organic thin film layer used in the organic EL device of the present invention can be prepared by vacuum evaporation, molecular beam evaporation (MBE), dating of a solution dissolved in a solvent, spin coating, casting, bar It can be formed by a known method such as a coating method or a roll coating method.
- MBE molecular beam evaporation
- an appropriate resin or additive may be used for improving the film formability and preventing pinholes in the film.
- Resins that can be used include insulating resins such as polystyrene, polycarbonate, polyarylate, polyester, polyamide, polyurethane, polysulfone, polymethyl methacrylate, polymethyl acrylate, cellulose, copolymers thereof, poly Examples thereof include photoconductive resins such as _N_bulucarbazole and polysilane, and conductive resins such as polyaniline, polythiophene, and polypyrrole. Examples of the additive include an antioxidant, an ultraviolet absorber, and a plasticizer.
- each organic layer of the organic EL device of the present invention is not particularly limited, but it is necessary to set an appropriate thickness. In general, if the film thickness is too thin, pinholes and the like may occur, and there is a risk that sufficient light emission brightness may not be obtained even when an electric field is applied.
- the film thickness is usually in the range of 5nm to 10 / im, but the range of 10nm to 0.2 / im is more preferred.
- a protective layer may be provided on the surface of the device, or the entire device may be protected with silicone oil, resin, etc. Is possible.
- a direct current voltage When a direct current voltage is applied to the organic EL element, light emission can be observed by applying a voltage of 5 to 40 V with the anode set to + and the cathode set to one polarity. In addition, even when a voltage is applied with the opposite polarity, no current flows and no light emission occurs. Furthermore, when AC voltage is applied, uniform light emission is observed only when the anode is + and the cathode is of the same polarity.
- the alternating current waveform to be applied may be arbitrary.
- a transparent electrode made of a chemical compound was provided. This glass substrate was ultrasonically cleaned with isopropyl alcohol and then irradiated with ultraviolet rays and ozone.
- the glass substrate with a transparent electrode was mounted on a substrate holder in a vapor deposition tank of a vacuum vapor deposition apparatus, and the degree of vacuum in the vacuum tank was reduced to 1 ⁇ 10 ⁇ 3 Pa.
- N N-bis [4,- ⁇ N— (naphthyl-1-yl) -N-phenyl ⁇ aminobiphenyl-4-yl] -N-phenylamine
- the layer was deposited at a deposition rate of 2 nm Zsec and a film thickness of 20 nm. This film functions as a hole transport layer.
- the compound (2a'-55) and the compound (1-13) are deposited at a deposition rate of 2 nm / sec and 0.2 nm / sec, respectively, with a film thickness of 40 nm and a weight ratio of (2a '- 55): (1 3) was co-deposited to be 40: 2.
- This film functions as a light emitting layer.
- tris (8-hydroxyquinolino) aluminum was deposited at a deposition rate of 2 nm / sec and a film thickness of 20 nm to form an electron transport layer.
- an electron injection layer was formed from lithium fluoride at a deposition rate of 0.1 nm / sec and a film thickness of 1 nm.
- a cathode layer was formed by depositing aluminum at a deposition rate of 2 nm / sec and a film thickness of 200 nm.
- Example 2 instead of the compound (2a′-55) and the compound (1-13) in Example 1, the compound (2a′-59) in Example 2 was used instead of the compound (2a′-55) in Example 1.
- compound (11-3) in a weight ratio of 40: 2 in Example 3, compound (2a'-59) and compound (11-7) in a weight ratio of 40: 2, in Example 4
- the compound (2a'-101) and the compound (1 79) were used in a weight ratio of 40: 2
- Example 5 the compound (2a-7) and the compound (1-27) were used in a weight ratio of 40: 2.
- an organic EL device was produced.
- Example 6 instead of the compound (2a′-55) and the compound (1-3) of Example 1, the compound (2a′-57) and the compound ( 1) 36) in a weight ratio of 40: 2, in Example 7, compound (2b-9) and compound (11-5) in a weight ratio of 40: 2, in Example 8, compound (2b-26) and compound (1 97) in a weight ratio of 40: 2, in Example 9, compound (2c-1) and compound (1-8) were in a weight ratio of 40: 2, and in Example 10, compound (2d-1) and compound (1-13) ) was used in the same manner as in Example 1 except that 40) was used at a weight ratio of 40: 2.
- Comparative Examples 1 to 3 only the compound (2a′-59) in Comparative Example 1 was used in the light-emitting layer instead of the compound (2a′55) and the compound (1 3) of Example 1, respectively.
- compound (A) is bis (2-methyl-8-quinolinolato) aluminum 1 ⁇ oxo-bis (2-methyl-8-quinolinolato) aluminum, and compound ( ⁇ ) is: :! -Tetra-butylperylene.
- a transparent electrode made of indium tin oxide having a thickness of 120 nm was provided on a glass substrate having a thickness of 25 mm X 75 mm X 1.1 mm. This glass substrate was ultrasonically cleaned with isopropyl alcohol and then irradiated with ultraviolet rays and ozone.
- the glass substrate with a transparent electrode was mounted on a substrate holder in a vapor deposition tank of a vacuum vapor deposition apparatus, and the degree of vacuum in the vacuum tank was reduced to 1 ⁇ 10 ⁇ 3 Pa.
- N, N, N ′, N′-tetra (4-biphenyl) benzidine was deposited on the hole injection layer at a deposition rate of 2 nm / sec and a film thickness of 20 nm. This film functions as a hole transport layer.
- tris (8-hydroxyquinolino) aluminum was deposited at a deposition rate of 2 nm / sec and a film thickness of 20 nm to form an electron transport layer.
- an electron injection layer was formed from lithium fluoride at a deposition rate of 0.1 nm / sec and a film thickness of 1 nm.
- an organic EL device was manufactured by forming a cathode layer of aluminum at a deposition rate of 2 nm Zsec and a film thickness of 200 nm.
- An organic EL device was produced in the same manner except that the luminescent material 1 and the luminescent material 2 of Example 11 were changed as shown in Table 4-11 and Table 4-12.
- the emission luminance was Sl80 to 550 cd / m 2 at a voltage of 6.3 V, and the emission peak wavelength (EL ⁇ ) and chromaticity were measured.
- the light color was confirmed to be blue. Further, when allowed to constant current driving the initial emission luminance set LOOcdZm 2, the half-life was 5, 500 hours or more, sufficient to confirm that it was the practical region. The results obtained are shown in Tables 4_1 and 4_2.
- Examples shown in Table 1, Table 2, Table 4 1-1 and Table 4 1-2 From the evaluation results of! To 20, the organic EL device of the present invention has excellent emission brightness and practical use. A half-life in the range can be achieved. In particular, by using an anthracene structure as the condensed ring-containing G 2 and a (lb) structure compound as the fluoranthene structure, the light emission efficiency and the half life are most excellent.
- the compound (C) is benzo [k] fluoranthene.
- a glass substrate with a 25 mm ⁇ 75 mm ⁇ l. 1 mm thick IT ⁇ transparent electrode was ultrasonically cleaned in isopropyl alcohol for 5 minutes, followed by UV ozone cleaning for 30 minutes.
- a hole-injecting / transporting layer having a laminated structure was formed on this substrate.
- a film of polyethylene dioxythiophene * polystyrene sulfonic acid (PEDOT 'PSS) with a film thickness of lOOnm was formed by spin coating.
- the polymer 1 (molecular weight (Mw): 145000)
- a chain solution (0.6% by weight) was formed into a film with a thickness of 20 nm by spin coating and dried at 170 ° C. for 30 minutes.
- Alq film A tris (8-quinolinol) aluminum film (hereinafter abbreviated as “Alq film”) having a thickness of 10 nm was formed on the light emitting layer. This Alq film functions as an electron transport layer.
- Li Li source: manufactured by SAES Getter Co., Ltd.
- Alq Alq
- metal A1 was deposited to form a metal cathode, and an organic EL device was formed.
- This device emitted blue light, and the light emitting surface was uniform.
- the luminous efficiency at this time was 2.9 cd / A.
- a transparent electrode made of indium tin oxide having a thickness of 120 nm was provided on a glass substrate having a thickness of 25 mm X 75 mm X 1.1 mm. This glass substrate was ultrasonically cleaned with isopropyl alcohol and then irradiated with ultraviolet rays and ozone. Next, the glass substrate with a transparent electrode was mounted on a substrate holder in a vapor deposition tank of a vacuum vapor deposition apparatus, and the degree of vacuum in the vacuum tank was reduced to 1 ⁇ 10 ⁇ 3 Pa.
- the above compound (2a-2) [Luminescent material 1] and the above compound (2-28) [Luminescent material 2] were deposited at a deposition rate of 2 nm Zsec and 0.2 nm / sec, respectively, and a film thickness of 40 nm.
- This film functions as a light emitting layer.
- tris (8-hydroxyquinolino) aluminum was deposited at a deposition rate of 2 nm / sec and a film thickness of 20 nm to form an electron transport layer.
- an electron injection layer was formed using lithium fluoride at a deposition rate of 0.1 nm / sec and a film thickness of 1 nm.
- a cathode layer was formed by depositing aluminum with a vapor deposition rate of 2 nm Zsec and a film thickness of 200 nm to manufacture an organic EL device.
- an energization test was performed on this device. As a result, the emission luminance was 250 cd / m 2 at a voltage of 6.5 V, and the emission peak wavelength (EL) was 446.
- the half-life was 10,000 hours or more, and it was confirmed that it was sufficiently practical.
- the light emitting layer comprises at least one selected from compounds having a fluoranthene structure represented by the general formula (1), and a condensed ring-containing compound represented by the general formula (2).
- the organic electoluminescence element of the present invention containing at least one selected from the above has a high emission luminance and a long lifetime. Furthermore, in order to improve the stability of the organic EL device obtained by the present invention with respect to temperature, humidity, atmosphere, etc., a protective layer is provided on the surface of the device, or the entire device is protected by silicone oil, resin, etc. Is also possible.
- the organic EL device of the present invention is useful as a light source such as a flat light emitter of a wall-mounted television and a backlight of a display, which are highly practical. It can also be used as an organic EL device, a hole injection / transport material, and a charge transport material for electrophotographic photoreceptors and organic semiconductors.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2007800068553A CN101390230B (zh) | 2006-02-28 | 2007-02-28 | 有机电致发光元件 |
| EP07737525.1A EP1990844B1 (en) | 2006-02-28 | 2007-02-28 | Organic electroluminescent device |
| JP2008502825A JP5199066B2 (ja) | 2006-02-28 | 2007-02-28 | 有機エレクトロルミネッセンス素子 |
| KR1020087021039A KR101406631B1 (ko) | 2006-02-28 | 2007-02-28 | 유기 전기 발광 소자 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006053942 | 2006-02-28 | ||
| JP2006-053942 | 2006-02-28 | ||
| JP2006312023 | 2006-11-17 | ||
| JP2006-312023 | 2006-11-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007100010A1 true WO2007100010A1 (ja) | 2007-09-07 |
Family
ID=38459111
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2007/053806 WO2007100010A1 (ja) | 2006-02-28 | 2007-02-28 | 有機エレクトロルミネッセンス素子 |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US9214636B2 (ja) |
| EP (1) | EP1990844B1 (ja) |
| JP (1) | JP5199066B2 (ja) |
| KR (1) | KR101406631B1 (ja) |
| CN (2) | CN102394273A (ja) |
| TW (1) | TWI433595B (ja) |
| WO (1) | WO2007100010A1 (ja) |
Cited By (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008015945A1 (en) | 2006-08-04 | 2008-02-07 | Canon Kabushiki Kaisha | ORGANIC LUMINESCENT DEVICE AND BENZO[k]FLUORANTHENE COMPOUND |
| WO2008059713A1 (en) * | 2006-11-15 | 2008-05-22 | Idemitsu Kosan Co., Ltd. | Fluoranthene compound, organic electroluminescent device using the fluoranthene compound, and organic electroluminescent material-containing solution |
| JP2008137978A (ja) * | 2006-12-05 | 2008-06-19 | Canon Inc | カルバゾール誘導体及びこれを用いた有機発光素子 |
| JP2008214308A (ja) * | 2007-03-07 | 2008-09-18 | Canon Inc | 縮合環化合物及びこれを用いた有機発光素子 |
| WO2009008348A1 (ja) * | 2007-07-07 | 2009-01-15 | Idemitsu Kosan Co., Ltd. | 有機el素子 |
| JP2009524653A (ja) * | 2006-01-27 | 2009-07-02 | エルジー・ケム・リミテッド | 新規なアントラセン誘導体、その製造方法およびこれを用いた有機電気素子 |
| WO2009123344A1 (en) * | 2008-04-01 | 2009-10-08 | Canon Kabushiki Kaisha | Organic light-emitting device |
| WO2010058855A1 (en) * | 2008-11-19 | 2010-05-27 | Canon Kabushiki Kaisha | Novel organic compound, light-emitting device, and image display apparatus |
| WO2010134350A1 (ja) | 2009-05-22 | 2010-11-25 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2010134352A1 (ja) | 2009-05-22 | 2010-11-25 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2010143434A1 (ja) | 2009-06-12 | 2010-12-16 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2011077691A1 (ja) * | 2009-12-21 | 2011-06-30 | 出光興産株式会社 | ピレン誘導体を用いた有機エレクトロルミネッセンス素子 |
| JP2011249653A (ja) * | 2010-05-28 | 2011-12-08 | Tdk Corp | 有機el素子 |
| JPWO2011077689A1 (ja) * | 2009-12-21 | 2013-05-02 | 出光興産株式会社 | ピレン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 |
| US8476823B2 (en) | 2009-05-22 | 2013-07-02 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| WO2014007287A1 (ja) | 2012-07-05 | 2014-01-09 | 東レ株式会社 | 発光素子材料および発光素子 |
| WO2014034891A1 (ja) * | 2012-08-31 | 2014-03-06 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2014034864A1 (ja) * | 2012-08-31 | 2014-03-06 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2014057873A1 (ja) * | 2012-10-10 | 2014-04-17 | 東レ株式会社 | ホスフィンオキサイド誘導体およびそれを有する発光素子 |
| WO2014057874A1 (ja) | 2012-10-12 | 2014-04-17 | 東レ株式会社 | フルオランテン誘導体、それを含有する発光素子材料および発光素子 |
| WO2014073306A1 (ja) * | 2012-11-07 | 2014-05-15 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子および電子機器 |
| WO2014073307A1 (ja) * | 2012-11-07 | 2014-05-15 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子および電子機器 |
| US9153790B2 (en) | 2009-05-22 | 2015-10-06 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| KR101621801B1 (ko) * | 2015-01-14 | 2016-05-17 | 삼성디스플레이 주식회사 | 방향족 복소환 화합물 및 이를 포함한 유기막을 구비한 유기 발광 소자 |
| WO2017170313A1 (ja) | 2016-03-29 | 2017-10-05 | 住友化学株式会社 | 発光素子 |
| JP2017183723A (ja) * | 2016-03-29 | 2017-10-05 | 住友化学株式会社 | 発光素子 |
| WO2017221822A1 (ja) | 2016-06-24 | 2017-12-28 | 住友化学株式会社 | 発光素子 |
| WO2021090932A1 (ja) * | 2019-11-08 | 2021-05-14 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び電子機器 |
| WO2021201176A1 (ja) * | 2020-04-01 | 2021-10-07 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子、化合物及び電子機器 |
| WO2021210304A1 (ja) * | 2020-04-15 | 2021-10-21 | 出光興産株式会社 | 化合物、有機エレクトロルミネッセンス素子及び電子機器 |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1138745B1 (en) * | 1999-09-30 | 2007-03-28 | Idemitsu Kosan Company Limited | Organic electroluminescent element |
| KR100874472B1 (ko) * | 2007-02-28 | 2008-12-18 | 에스에프씨 주식회사 | 청색발광화합물 및 이를 이용한 유기전계발광소자 |
| US8154195B2 (en) | 2007-07-07 | 2012-04-10 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and material for organic electroluminescence device |
| WO2009066600A1 (ja) * | 2007-11-19 | 2009-05-28 | Idemitsu Kosan Co., Ltd. | モノベンゾクリセン誘導体、及びそれを含む有機エレクトロルミネッセンス素子用材料、並びにそれを用いた有機エレクトロルミネッセンス素子 |
| KR101528241B1 (ko) * | 2007-12-07 | 2015-06-15 | 삼성디스플레이 주식회사 | 방향족 복소환 화합물, 이를 포함한 유기막을 구비한 유기발광 소자 및 상기 유기발광 소자의 제조 방법 |
| CN101910147B (zh) | 2007-12-28 | 2014-02-19 | 出光兴产株式会社 | 芳胺衍生物及使用该芳胺衍生物的有机电致发光元件 |
| KR101099753B1 (ko) | 2008-02-02 | 2011-12-28 | 에스에프씨 주식회사 | 플루오란텐 유도체 및 이를 포함하는 유기전계발광소자 |
| KR100901887B1 (ko) * | 2008-03-14 | 2009-06-09 | (주)그라쎌 | 신규한 유기 발광 화합물 및 이를 채용하고 있는 유기 발광소자 |
| JP2010121036A (ja) * | 2008-11-19 | 2010-06-03 | Canon Inc | 発光素子及び画像表示装置および新規有機化合物 |
| US20100244677A1 (en) * | 2009-03-31 | 2010-09-30 | Begley William J | Oled device containing a silyl-fluoranthene derivative |
| US8039129B2 (en) * | 2009-04-06 | 2011-10-18 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and material for organic electroluminescence device |
| US8039127B2 (en) * | 2009-04-06 | 2011-10-18 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and material for organic electroluminescence device |
| EP2423206B1 (en) | 2009-04-24 | 2014-01-08 | Idemitsu Kosan Co., Ltd. | Aromatic amine derivative, and organic electroluminescent element comprising same |
| CN102224614B (zh) * | 2009-12-16 | 2014-04-23 | 出光兴产株式会社 | 有机发光介质 |
| CN103012206B (zh) * | 2011-09-28 | 2014-10-01 | 海洋王照明科技股份有限公司 | 双荧蒽有机半导体材料及其制备方法和应用 |
| CN103012163B (zh) * | 2011-09-28 | 2015-07-08 | 海洋王照明科技股份有限公司 | 双荧蒽有机半导体材料及其制备方法和应用 |
| CN103044386B (zh) * | 2011-10-17 | 2014-10-01 | 海洋王照明科技股份有限公司 | 含双荧蒽有机半导体材料、其制备方法及应用 |
| US10680182B2 (en) * | 2012-11-21 | 2020-06-09 | Lg Chem, Ltd. | Fluoranthene compound, and organic electronic device comprising same |
| JP6167176B2 (ja) * | 2013-05-31 | 2017-07-19 | 出光興産株式会社 | 縮合フルオランテン化合物、これを含む有機エレクトロルミネッセンス素子用材料、並びにこれを用いた有機エレクトロルミネッセンス素子及び電子機器 |
| CN106356452B (zh) * | 2015-12-09 | 2018-09-18 | 广东阿格蕾雅光电材料有限公司 | 仅电子有机半导体二极管器件 |
| KR20230117645A (ko) | 2017-04-26 | 2023-08-08 | 오티아이 루미오닉스 인크. | 표면의 코팅을 패턴화하는 방법 및 패턴화된 코팅을포함하는 장치 |
| US11751415B2 (en) | 2018-02-02 | 2023-09-05 | Oti Lumionics Inc. | Materials for forming a nucleation-inhibiting coating and devices incorporating same |
| CN108383693A (zh) * | 2018-02-06 | 2018-08-10 | 陕西师范大学 | 一种有机蓝色荧光材料及其制备方法和应用 |
| CN113785411B (zh) | 2019-03-07 | 2023-04-11 | Oti照明公司 | 用于形成成核抑制涂层的材料和结合所述成核抑制涂层的装置 |
Citations (113)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1110518B (de) | 1959-04-09 | 1961-07-06 | Kalle Ag | Material fuer die elektrophotographische Bilderzeugung |
| US3008897A (en) | 1959-08-07 | 1961-11-14 | Sinclair Refining Co | Hydrocarbon demetallization process |
| US3112197A (en) | 1956-06-27 | 1963-11-26 | Azoplate Corp | Electrophotographic member |
| US3180729A (en) | 1956-12-22 | 1965-04-27 | Azoplate Corp | Material for electrophotographic reproduction |
| US3180703A (en) | 1963-01-15 | 1965-04-27 | Kerr Mc Gee Oil Ind Inc | Recovery process |
| US3189447A (en) | 1956-06-04 | 1965-06-15 | Azoplate Corp | Electrophotographic material and method |
| US3240597A (en) | 1961-08-21 | 1966-03-15 | Eastman Kodak Co | Photoconducting polymers for preparing electrophotographic materials |
| US3257203A (en) | 1958-08-20 | 1966-06-21 | Azoplate Corp | Electrophotographic reproduction material |
| JPS45555B1 (ja) | 1966-03-24 | 1970-01-09 | ||
| US3526501A (en) | 1967-02-03 | 1970-09-01 | Eastman Kodak Co | 4-diarylamino-substituted chalcone containing photoconductive compositions for use in electrophotography |
| US3542544A (en) | 1967-04-03 | 1970-11-24 | Eastman Kodak Co | Photoconductive elements containing organic photoconductors of the triarylalkane and tetraarylmethane types |
| JPS463712B1 (ja) | 1966-04-14 | 1971-01-29 | ||
| US3567450A (en) | 1968-02-20 | 1971-03-02 | Eastman Kodak Co | Photoconductive elements containing substituted triarylamine photoconductors |
| US3615404A (en) | 1968-04-25 | 1971-10-26 | Scott Paper Co | 1 3-phenylenediamine containing photoconductive materials |
| US3615402A (en) | 1969-10-01 | 1971-10-26 | Eastman Kodak Co | Tetra-substituted methanes as organic photoconductors |
| US3658520A (en) | 1968-02-20 | 1972-04-25 | Eastman Kodak Co | Photoconductive elements containing as photoconductors triarylamines substituted by active hydrogen-containing groups |
| JPS4725336B1 (ja) | 1969-11-26 | 1972-07-11 | ||
| US3717462A (en) | 1969-07-28 | 1973-02-20 | Canon Kk | Heat treatment of an electrophotographic photosensitive member |
| US3820989A (en) | 1969-09-30 | 1974-06-28 | Eastman Kodak Co | Tri-substituted methanes as organic photoconductors |
| JPS4935702B1 (ja) | 1969-06-20 | 1974-09-25 | ||
| JPS49105537A (ja) | 1973-01-15 | 1974-10-05 | ||
| JPS5110105B2 (ja) | 1972-02-09 | 1976-04-01 | ||
| JPS5110983B2 (ja) | 1971-09-10 | 1976-04-08 | ||
| JPS5193224A (ja) | 1974-12-20 | 1976-08-16 | ||
| US4012376A (en) | 1975-12-29 | 1977-03-15 | Eastman Kodak Company | Photosensitive colorant materials |
| JPS5327033A (en) | 1976-08-23 | 1978-03-13 | Xerox Corp | Image forming member and image forming method |
| US4127412A (en) | 1975-12-09 | 1978-11-28 | Eastman Kodak Company | Photoconductive compositions and elements |
| JPS5453435A (en) | 1977-10-01 | 1979-04-26 | Yoshikatsu Kume | Portable bicycle equipped with foldable type triangle frame |
| JPS5458445A (en) | 1977-09-29 | 1979-05-11 | Xerox Corp | Electrostatic photosensitive device |
| JPS5459143A (en) | 1977-10-17 | 1979-05-12 | Ibm | Electronic photographic material |
| JPS5464299A (en) | 1977-10-29 | 1979-05-23 | Toshiba Corp | Beam deflector for charged particles |
| JPS54110837A (en) | 1978-02-17 | 1979-08-30 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS54110536A (en) | 1978-02-20 | 1979-08-30 | Ichikoh Ind Ltd | Device for time-lag putting out room lamp for motorcar |
| JPS54112637A (en) | 1978-02-06 | 1979-09-03 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS54119925A (en) | 1978-03-10 | 1979-09-18 | Ricoh Co Ltd | Photosensitive material for electrophotography |
| JPS54149634A (en) | 1978-05-12 | 1979-11-24 | Xerox Corp | Image forming member and method of forming image using same |
| US4175961A (en) | 1976-12-22 | 1979-11-27 | Eastman Kodak Company | Multi-active photoconductive elements |
| JPS5517105A (en) | 1978-07-21 | 1980-02-06 | Konishiroku Photo Ind Co Ltd | Electrophotographic photoreceptor |
| JPS5546760A (en) | 1978-09-29 | 1980-04-02 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS5551086A (en) | 1978-09-04 | 1980-04-14 | Copyer Co Ltd | Novel pyrazoline compound, its preparation, and electrophotographic photosensitive substance comprising it |
| JPS5552063A (en) | 1978-10-13 | 1980-04-16 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5552064A (en) | 1978-10-13 | 1980-04-16 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5574546A (en) | 1978-11-30 | 1980-06-05 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS5579450A (en) | 1978-12-04 | 1980-06-14 | Xerox Corp | Image formation device |
| JPS5585495A (en) | 1978-12-18 | 1980-06-27 | Pacific Metals Co Ltd | Method of composting organic waste |
| JPS5588064A (en) | 1978-12-05 | 1980-07-03 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS5588065A (en) | 1978-12-12 | 1980-07-03 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS55108667A (en) | 1979-02-13 | 1980-08-21 | Ricoh Co Ltd | Electrophotographic receptor |
| US4232103A (en) | 1979-08-27 | 1980-11-04 | Xerox Corporation | Phenyl benzotriazole stabilized photosensitive device |
| JPS55144250A (en) | 1979-04-30 | 1980-11-11 | Xerox Corp | Image formation device |
| JPS55156953A (en) | 1979-05-17 | 1980-12-06 | Mitsubishi Paper Mills Ltd | Organic semiconductor electrophotographic material |
| JPS564148A (en) | 1979-06-21 | 1981-01-17 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS5622437A (en) | 1979-08-01 | 1981-03-03 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5636656A (en) | 1979-09-03 | 1981-04-09 | Mitsubishi Paper Mills Ltd | Electrophotographic material |
| JPS5646234A (en) | 1979-09-21 | 1981-04-27 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5680051A (en) | 1979-12-04 | 1981-07-01 | Ricoh Co Ltd | Electrophotographic receptor |
| US4278746A (en) | 1978-06-21 | 1981-07-14 | Konishiroku Photo Industry Co., Ltd. | Photosensitive elements for electrophotography |
| JPS5688141A (en) | 1979-12-20 | 1981-07-17 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS56119132A (en) | 1979-11-23 | 1981-09-18 | Xerox Corp | Image forming element |
| JPS5711350A (en) | 1980-06-24 | 1982-01-21 | Fuji Photo Film Co Ltd | Electrophotographic receptor |
| JPS5745545A (en) | 1980-09-03 | 1982-03-15 | Mitsubishi Paper Mills Ltd | Electrophotographic receptor |
| JPS5751781A (en) | 1980-07-17 | 1982-03-26 | Eastman Kodak Co | Organic electroluminiscent cell and method |
| JPS57148749A (en) | 1981-03-11 | 1982-09-14 | Fuji Photo Film Co Ltd | Electrophotographic receptor |
| JPS6093445A (ja) | 1983-10-28 | 1985-05-25 | Ricoh Co Ltd | 電子写真用感光体 |
| JPS6094462A (ja) | 1983-10-28 | 1985-05-27 | Ricoh Co Ltd | スチルベン誘導体及びその製造法 |
| JPS60175052A (ja) | 1984-02-21 | 1985-09-09 | Ricoh Co Ltd | 電子写真用感光体 |
| JPS60174749A (ja) | 1984-02-21 | 1985-09-09 | Ricoh Co Ltd | スチリル化合物及びその製造法 |
| JPS6114642A (ja) | 1984-06-29 | 1986-01-22 | Konishiroku Photo Ind Co Ltd | 電子写真感光体 |
| JPS6172255A (ja) | 1984-09-14 | 1986-04-14 | Konishiroku Photo Ind Co Ltd | 電子写真感光体 |
| JPS6198353A (ja) | 1984-10-19 | 1986-05-16 | ゼロツクス コーポレーシヨン | 芳香族エーテル正孔移送層を含む感光装置 |
| JPS61210363A (ja) | 1985-03-15 | 1986-09-18 | Canon Inc | 電子写真感光体 |
| JPS61228451A (ja) | 1985-04-03 | 1986-10-11 | Canon Inc | 電子写真感光体 |
| JPS61295558A (ja) | 1985-06-24 | 1986-12-26 | ゼロツクス コ−ポレ−シヨン | アルコキシアミン電荷移送分子を含有する光導電性像形成部材 |
| JPS6210652A (ja) | 1985-07-08 | 1987-01-19 | Minolta Camera Co Ltd | 感光体 |
| JPS6230255A (ja) | 1985-07-31 | 1987-02-09 | Minolta Camera Co Ltd | 電子写真感光体 |
| JPS6236674A (ja) | 1985-08-05 | 1987-02-17 | Fuji Photo Film Co Ltd | 電子写真感光体 |
| JPS6247646A (ja) | 1985-08-27 | 1987-03-02 | Konishiroku Photo Ind Co Ltd | 感光体 |
| JPS63256965A (ja) | 1987-04-15 | 1988-10-24 | Canon Inc | 静電荷像現像用トナ− |
| JPS63295695A (ja) | 1987-02-11 | 1988-12-02 | イーストマン・コダック・カンパニー | 有機発光媒体をもつ電場発光デバイス |
| JPH01211399A (ja) | 1988-02-19 | 1989-08-24 | Toshiba Corp | スキャン機能付きダイナミックシフトレジスタ |
| JPH02204996A (ja) | 1989-02-01 | 1990-08-14 | Nec Corp | 有機薄膜el素子 |
| US4950950A (en) | 1989-05-18 | 1990-08-21 | Eastman Kodak Company | Electroluminescent device with silazane-containing luminescent zone |
| JPH02282263A (ja) | 1988-12-09 | 1990-11-19 | Nippon Oil Co Ltd | ホール輸送材料 |
| JPH02311591A (ja) | 1989-05-25 | 1990-12-27 | Mitsubishi Kasei Corp | 有機電界発光素子 |
| JPH03200889A (ja) | 1989-06-30 | 1991-09-02 | Ricoh Co Ltd | 電界発光素子 |
| US5061569A (en) | 1990-07-26 | 1991-10-29 | Eastman Kodak Company | Electroluminescent device with organic electroluminescent medium |
| JPH04308688A (ja) | 1991-04-08 | 1992-10-30 | Pioneer Electron Corp | 有機エレクトロルミネッセンス素子 |
| JPH07183561A (ja) | 1993-12-24 | 1995-07-21 | Sharp Corp | 回路内蔵受光素子の作製方法 |
| JPH0812600A (ja) | 1994-04-26 | 1996-01-16 | Tdk Corp | フェニルアントラセン誘導体および有機el素子 |
| JPH08193191A (ja) | 1995-01-19 | 1996-07-30 | Idemitsu Kosan Co Ltd | 有機電界発光素子及び有機薄膜 |
| JPH08239655A (ja) | 1995-03-06 | 1996-09-17 | Idemitsu Kosan Co Ltd | 有機エレクトロルミネッセンス素子 |
| JPH10189247A (ja) | 1996-11-07 | 1998-07-21 | Mitsui Chem Inc | 有機電界発光素子 |
| JP2000178212A (ja) * | 1998-12-17 | 2000-06-27 | Mitsui Chemicals Inc | 炭化水素化合物および有機電界発光素子 |
| JP2000186054A (ja) * | 1998-12-21 | 2000-07-04 | Mitsui Chemicals Inc | 炭化水素化合物および有機電界発光素子 |
| JP2001118682A (ja) | 1999-10-21 | 2001-04-27 | Fujitsu Ltd | 有機エレクトロルミネッセンス素子 |
| JP2001351783A (ja) * | 2000-06-06 | 2001-12-21 | Mitsui Chemicals Inc | 炭化水素化合物および有機電界発光素子 |
| JP2002032468A (ja) | 2000-07-18 | 2002-01-31 | Canon Inc | 情報処理システム及び装置及びそれらの方法並びに記憶媒体 |
| JP2002063988A (ja) | 2000-08-22 | 2002-02-28 | Toray Ind Inc | 発光素子 |
| JP2002069044A (ja) | 2000-08-25 | 2002-03-08 | Idemitsu Kosan Co Ltd | 有機エレクトロルミネッセンス素子 |
| WO2002085822A1 (fr) | 2001-04-19 | 2002-10-31 | Tdk Corporation | Compose destine a un element organique electroluminescent (el) et element organique el |
| JP2002324678A (ja) | 2001-04-26 | 2002-11-08 | Fuji Photo Film Co Ltd | 発光素子 |
| JP2003272862A (ja) * | 2002-03-13 | 2003-09-26 | Fuji Photo Film Co Ltd | 発光素子 |
| JP2004002351A (ja) * | 2002-03-27 | 2004-01-08 | Tdk Corp | 有機el素子 |
| JP2004075567A (ja) | 2002-08-12 | 2004-03-11 | Idemitsu Kosan Co Ltd | オリゴアリーレン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
| JP2004083481A (ja) | 2002-08-27 | 2004-03-18 | Canon Inc | フルオレン化合物及びそれを用いた有機発光素子 |
| JP2004356033A (ja) | 2003-05-30 | 2004-12-16 | Tdk Corp | 有機el素子 |
| JP2005068087A (ja) | 2003-08-26 | 2005-03-17 | Mitsui Chemicals Inc | ベンゾフルオランテン化合物、および該ベンゾフルオランテン化合物を含有する有機電界発光素子 |
| WO2005033051A1 (de) | 2003-09-29 | 2005-04-14 | Basf Aktiengesellschaft | Synthese von phenylsubstituierten fluoranthenen durch diels-alder-reaktion und ihre verwendung |
| WO2005061656A1 (ja) | 2003-12-19 | 2005-07-07 | Idemitsu Kosan Co., Ltd. | 有機エレクトロルミネッセンス素子用発光材料、それを利用した有機エレクトロルミネッセンス素子及び有機エレクトロルミネッセンス素子用材料 |
| JP2005240008A (ja) * | 2004-01-27 | 2005-09-08 | Sony Corp | 有機発光材料および有機電界発光素子 |
| JP3716096B2 (ja) | 1998-04-02 | 2005-11-16 | 三菱重工業株式会社 | 微粉炭セパレータ装置 |
| WO2005115950A1 (ja) | 2004-05-27 | 2005-12-08 | Idemitsu Kosan Co., Ltd. | 非対称ピレン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
| JP3927577B2 (ja) | 1994-11-10 | 2007-06-13 | マイケルスン、ガーリー、ケィー | 電動骨鉗子 |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07138561A (ja) | 1993-11-17 | 1995-05-30 | Idemitsu Kosan Co Ltd | 有機エレクトロルミネッセンス素子 |
| JP4070274B2 (ja) | 1996-11-07 | 2008-04-02 | 三井化学株式会社 | 有機電界発光素子 |
| JP3899566B2 (ja) | 1996-11-25 | 2007-03-28 | セイコーエプソン株式会社 | 有機el表示装置の製造方法 |
| JP3794819B2 (ja) | 1997-04-18 | 2006-07-12 | 三井化学株式会社 | フルオランテン誘導体および有機電界発光素子 |
| US5935721A (en) * | 1998-03-20 | 1999-08-10 | Eastman Kodak Company | Organic electroluminescent elements for stable electroluminescent |
| EP1138745B1 (en) * | 1999-09-30 | 2007-03-28 | Idemitsu Kosan Company Limited | Organic electroluminescent element |
| JP4255610B2 (ja) * | 1999-12-28 | 2009-04-15 | 出光興産株式会社 | 白色系有機エレクトロルミネッセンス素子 |
| US6866947B1 (en) * | 1999-12-28 | 2005-03-15 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device emitting white light |
| JP3961200B2 (ja) | 2000-07-25 | 2007-08-22 | 三星エスディアイ株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2002020693A1 (en) | 2000-09-07 | 2002-03-14 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent element |
| JP4224252B2 (ja) | 2001-04-19 | 2009-02-12 | Tdk株式会社 | 有機el素子用化合物、有機el素子 |
| TWI314947B (en) * | 2002-04-24 | 2009-09-21 | Eastman Kodak Compan | Organic light emitting diode devices with improved operational stability |
| US6841267B2 (en) | 2002-04-24 | 2005-01-11 | Eastman Kodak Company | Efficient electroluminescent device |
| ATE555182T1 (de) | 2002-08-23 | 2012-05-15 | Idemitsu Kosan Co | Organische elektrolumineszenz vorrichtung und anthracenderivat |
| JP4585750B2 (ja) | 2002-08-27 | 2010-11-24 | キヤノン株式会社 | 縮合多環化合物及びそれを用いた有機発光素子 |
| US20040049754A1 (en) * | 2002-09-06 | 2004-03-11 | Sun Microsystems, Inc. | Method and apparatus for filling and connecting filler material in a layout |
| WO2004063159A1 (ja) | 2003-01-10 | 2004-07-29 | Idemitsu Kosan Co., Ltd. | 含窒素複素環誘導体及びそれを用いた有機エレクトロルミネッセンス素子 |
| JP4400134B2 (ja) | 2003-07-31 | 2010-01-20 | Tdk株式会社 | 有機el素子用化合物及び有機el素子 |
| US7368624B2 (en) * | 2004-03-30 | 2008-05-06 | Eastman Kodak Company | Synthesis for polycyclic aromatic hydrocarbon compounds |
| JP5084208B2 (ja) * | 2006-09-15 | 2012-11-28 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び有機エレクトロルミネッセンス素子用材料 |
| EP2166588A4 (en) * | 2007-07-07 | 2011-05-04 | Idemitsu Kosan Co | ORGANIC ELECTROLUMINESCENCE DEVICE |
-
2007
- 2007-02-27 US US11/679,531 patent/US9214636B2/en active Active
- 2007-02-28 CN CN2011103730708A patent/CN102394273A/zh active Pending
- 2007-02-28 WO PCT/JP2007/053806 patent/WO2007100010A1/ja active Application Filing
- 2007-02-28 CN CN2007800068553A patent/CN101390230B/zh not_active Expired - Fee Related
- 2007-02-28 KR KR1020087021039A patent/KR101406631B1/ko active IP Right Grant
- 2007-02-28 EP EP07737525.1A patent/EP1990844B1/en not_active Not-in-force
- 2007-02-28 JP JP2008502825A patent/JP5199066B2/ja not_active Expired - Fee Related
- 2007-03-01 TW TW096107028A patent/TWI433595B/zh not_active IP Right Cessation
-
2013
- 2013-01-10 US US13/738,372 patent/US20130221333A1/en not_active Abandoned
Patent Citations (113)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3189447A (en) | 1956-06-04 | 1965-06-15 | Azoplate Corp | Electrophotographic material and method |
| US3112197A (en) | 1956-06-27 | 1963-11-26 | Azoplate Corp | Electrophotographic member |
| US3180729A (en) | 1956-12-22 | 1965-04-27 | Azoplate Corp | Material for electrophotographic reproduction |
| US3257203A (en) | 1958-08-20 | 1966-06-21 | Azoplate Corp | Electrophotographic reproduction material |
| DE1110518B (de) | 1959-04-09 | 1961-07-06 | Kalle Ag | Material fuer die elektrophotographische Bilderzeugung |
| US3008897A (en) | 1959-08-07 | 1961-11-14 | Sinclair Refining Co | Hydrocarbon demetallization process |
| US3240597A (en) | 1961-08-21 | 1966-03-15 | Eastman Kodak Co | Photoconducting polymers for preparing electrophotographic materials |
| US3180703A (en) | 1963-01-15 | 1965-04-27 | Kerr Mc Gee Oil Ind Inc | Recovery process |
| JPS45555B1 (ja) | 1966-03-24 | 1970-01-09 | ||
| JPS463712B1 (ja) | 1966-04-14 | 1971-01-29 | ||
| US3526501A (en) | 1967-02-03 | 1970-09-01 | Eastman Kodak Co | 4-diarylamino-substituted chalcone containing photoconductive compositions for use in electrophotography |
| US3542544A (en) | 1967-04-03 | 1970-11-24 | Eastman Kodak Co | Photoconductive elements containing organic photoconductors of the triarylalkane and tetraarylmethane types |
| US3567450A (en) | 1968-02-20 | 1971-03-02 | Eastman Kodak Co | Photoconductive elements containing substituted triarylamine photoconductors |
| US3658520A (en) | 1968-02-20 | 1972-04-25 | Eastman Kodak Co | Photoconductive elements containing as photoconductors triarylamines substituted by active hydrogen-containing groups |
| US3615404A (en) | 1968-04-25 | 1971-10-26 | Scott Paper Co | 1 3-phenylenediamine containing photoconductive materials |
| JPS4935702B1 (ja) | 1969-06-20 | 1974-09-25 | ||
| US3717462A (en) | 1969-07-28 | 1973-02-20 | Canon Kk | Heat treatment of an electrophotographic photosensitive member |
| US3820989A (en) | 1969-09-30 | 1974-06-28 | Eastman Kodak Co | Tri-substituted methanes as organic photoconductors |
| US3615402A (en) | 1969-10-01 | 1971-10-26 | Eastman Kodak Co | Tetra-substituted methanes as organic photoconductors |
| JPS4725336B1 (ja) | 1969-11-26 | 1972-07-11 | ||
| JPS5110983B2 (ja) | 1971-09-10 | 1976-04-08 | ||
| JPS5110105B2 (ja) | 1972-02-09 | 1976-04-01 | ||
| JPS49105537A (ja) | 1973-01-15 | 1974-10-05 | ||
| JPS5193224A (ja) | 1974-12-20 | 1976-08-16 | ||
| US4127412A (en) | 1975-12-09 | 1978-11-28 | Eastman Kodak Company | Photoconductive compositions and elements |
| US4012376A (en) | 1975-12-29 | 1977-03-15 | Eastman Kodak Company | Photosensitive colorant materials |
| JPS5327033A (en) | 1976-08-23 | 1978-03-13 | Xerox Corp | Image forming member and image forming method |
| US4175961A (en) | 1976-12-22 | 1979-11-27 | Eastman Kodak Company | Multi-active photoconductive elements |
| JPS5458445A (en) | 1977-09-29 | 1979-05-11 | Xerox Corp | Electrostatic photosensitive device |
| JPS5453435A (en) | 1977-10-01 | 1979-04-26 | Yoshikatsu Kume | Portable bicycle equipped with foldable type triangle frame |
| JPS5459143A (en) | 1977-10-17 | 1979-05-12 | Ibm | Electronic photographic material |
| JPS5464299A (en) | 1977-10-29 | 1979-05-23 | Toshiba Corp | Beam deflector for charged particles |
| JPS54112637A (en) | 1978-02-06 | 1979-09-03 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS54110837A (en) | 1978-02-17 | 1979-08-30 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS54110536A (en) | 1978-02-20 | 1979-08-30 | Ichikoh Ind Ltd | Device for time-lag putting out room lamp for motorcar |
| JPS54119925A (en) | 1978-03-10 | 1979-09-18 | Ricoh Co Ltd | Photosensitive material for electrophotography |
| JPS54149634A (en) | 1978-05-12 | 1979-11-24 | Xerox Corp | Image forming member and method of forming image using same |
| US4278746A (en) | 1978-06-21 | 1981-07-14 | Konishiroku Photo Industry Co., Ltd. | Photosensitive elements for electrophotography |
| JPS5517105A (en) | 1978-07-21 | 1980-02-06 | Konishiroku Photo Ind Co Ltd | Electrophotographic photoreceptor |
| JPS5551086A (en) | 1978-09-04 | 1980-04-14 | Copyer Co Ltd | Novel pyrazoline compound, its preparation, and electrophotographic photosensitive substance comprising it |
| JPS5546760A (en) | 1978-09-29 | 1980-04-02 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS5552063A (en) | 1978-10-13 | 1980-04-16 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5552064A (en) | 1978-10-13 | 1980-04-16 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5574546A (en) | 1978-11-30 | 1980-06-05 | Ricoh Co Ltd | Electrophotographic photoreceptor |
| JPS5579450A (en) | 1978-12-04 | 1980-06-14 | Xerox Corp | Image formation device |
| JPS5588064A (en) | 1978-12-05 | 1980-07-03 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS5588065A (en) | 1978-12-12 | 1980-07-03 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS5585495A (en) | 1978-12-18 | 1980-06-27 | Pacific Metals Co Ltd | Method of composting organic waste |
| JPS55108667A (en) | 1979-02-13 | 1980-08-21 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS55144250A (en) | 1979-04-30 | 1980-11-11 | Xerox Corp | Image formation device |
| JPS55156953A (en) | 1979-05-17 | 1980-12-06 | Mitsubishi Paper Mills Ltd | Organic semiconductor electrophotographic material |
| JPS564148A (en) | 1979-06-21 | 1981-01-17 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS5622437A (en) | 1979-08-01 | 1981-03-03 | Ricoh Co Ltd | Electrophotographic receptor |
| US4232103A (en) | 1979-08-27 | 1980-11-04 | Xerox Corporation | Phenyl benzotriazole stabilized photosensitive device |
| JPS5636656A (en) | 1979-09-03 | 1981-04-09 | Mitsubishi Paper Mills Ltd | Electrophotographic material |
| JPS5646234A (en) | 1979-09-21 | 1981-04-27 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS56119132A (en) | 1979-11-23 | 1981-09-18 | Xerox Corp | Image forming element |
| JPS5680051A (en) | 1979-12-04 | 1981-07-01 | Ricoh Co Ltd | Electrophotographic receptor |
| JPS5688141A (en) | 1979-12-20 | 1981-07-17 | Konishiroku Photo Ind Co Ltd | Electrophotographic receptor |
| JPS5711350A (en) | 1980-06-24 | 1982-01-21 | Fuji Photo Film Co Ltd | Electrophotographic receptor |
| JPS5751781A (en) | 1980-07-17 | 1982-03-26 | Eastman Kodak Co | Organic electroluminiscent cell and method |
| JPS5745545A (en) | 1980-09-03 | 1982-03-15 | Mitsubishi Paper Mills Ltd | Electrophotographic receptor |
| JPS57148749A (en) | 1981-03-11 | 1982-09-14 | Fuji Photo Film Co Ltd | Electrophotographic receptor |
| JPS6093445A (ja) | 1983-10-28 | 1985-05-25 | Ricoh Co Ltd | 電子写真用感光体 |
| JPS6094462A (ja) | 1983-10-28 | 1985-05-27 | Ricoh Co Ltd | スチルベン誘導体及びその製造法 |
| JPS60175052A (ja) | 1984-02-21 | 1985-09-09 | Ricoh Co Ltd | 電子写真用感光体 |
| JPS60174749A (ja) | 1984-02-21 | 1985-09-09 | Ricoh Co Ltd | スチリル化合物及びその製造法 |
| JPS6114642A (ja) | 1984-06-29 | 1986-01-22 | Konishiroku Photo Ind Co Ltd | 電子写真感光体 |
| JPS6172255A (ja) | 1984-09-14 | 1986-04-14 | Konishiroku Photo Ind Co Ltd | 電子写真感光体 |
| JPS6198353A (ja) | 1984-10-19 | 1986-05-16 | ゼロツクス コーポレーシヨン | 芳香族エーテル正孔移送層を含む感光装置 |
| JPS61210363A (ja) | 1985-03-15 | 1986-09-18 | Canon Inc | 電子写真感光体 |
| JPS61228451A (ja) | 1985-04-03 | 1986-10-11 | Canon Inc | 電子写真感光体 |
| JPS61295558A (ja) | 1985-06-24 | 1986-12-26 | ゼロツクス コ−ポレ−シヨン | アルコキシアミン電荷移送分子を含有する光導電性像形成部材 |
| JPS6210652A (ja) | 1985-07-08 | 1987-01-19 | Minolta Camera Co Ltd | 感光体 |
| JPS6230255A (ja) | 1985-07-31 | 1987-02-09 | Minolta Camera Co Ltd | 電子写真感光体 |
| JPS6236674A (ja) | 1985-08-05 | 1987-02-17 | Fuji Photo Film Co Ltd | 電子写真感光体 |
| JPS6247646A (ja) | 1985-08-27 | 1987-03-02 | Konishiroku Photo Ind Co Ltd | 感光体 |
| JPS63295695A (ja) | 1987-02-11 | 1988-12-02 | イーストマン・コダック・カンパニー | 有機発光媒体をもつ電場発光デバイス |
| JPS63256965A (ja) | 1987-04-15 | 1988-10-24 | Canon Inc | 静電荷像現像用トナ− |
| JPH01211399A (ja) | 1988-02-19 | 1989-08-24 | Toshiba Corp | スキャン機能付きダイナミックシフトレジスタ |
| JPH02282263A (ja) | 1988-12-09 | 1990-11-19 | Nippon Oil Co Ltd | ホール輸送材料 |
| JPH02204996A (ja) | 1989-02-01 | 1990-08-14 | Nec Corp | 有機薄膜el素子 |
| US4950950A (en) | 1989-05-18 | 1990-08-21 | Eastman Kodak Company | Electroluminescent device with silazane-containing luminescent zone |
| JPH02311591A (ja) | 1989-05-25 | 1990-12-27 | Mitsubishi Kasei Corp | 有機電界発光素子 |
| JPH03200889A (ja) | 1989-06-30 | 1991-09-02 | Ricoh Co Ltd | 電界発光素子 |
| US5061569A (en) | 1990-07-26 | 1991-10-29 | Eastman Kodak Company | Electroluminescent device with organic electroluminescent medium |
| JPH04308688A (ja) | 1991-04-08 | 1992-10-30 | Pioneer Electron Corp | 有機エレクトロルミネッセンス素子 |
| JPH07183561A (ja) | 1993-12-24 | 1995-07-21 | Sharp Corp | 回路内蔵受光素子の作製方法 |
| JPH0812600A (ja) | 1994-04-26 | 1996-01-16 | Tdk Corp | フェニルアントラセン誘導体および有機el素子 |
| JP3927577B2 (ja) | 1994-11-10 | 2007-06-13 | マイケルスン、ガーリー、ケィー | 電動骨鉗子 |
| JPH08193191A (ja) | 1995-01-19 | 1996-07-30 | Idemitsu Kosan Co Ltd | 有機電界発光素子及び有機薄膜 |
| JPH08239655A (ja) | 1995-03-06 | 1996-09-17 | Idemitsu Kosan Co Ltd | 有機エレクトロルミネッセンス素子 |
| JPH10189247A (ja) | 1996-11-07 | 1998-07-21 | Mitsui Chem Inc | 有機電界発光素子 |
| JP3716096B2 (ja) | 1998-04-02 | 2005-11-16 | 三菱重工業株式会社 | 微粉炭セパレータ装置 |
| JP2000178212A (ja) * | 1998-12-17 | 2000-06-27 | Mitsui Chemicals Inc | 炭化水素化合物および有機電界発光素子 |
| JP2000186054A (ja) * | 1998-12-21 | 2000-07-04 | Mitsui Chemicals Inc | 炭化水素化合物および有機電界発光素子 |
| JP2001118682A (ja) | 1999-10-21 | 2001-04-27 | Fujitsu Ltd | 有機エレクトロルミネッセンス素子 |
| JP2001351783A (ja) * | 2000-06-06 | 2001-12-21 | Mitsui Chemicals Inc | 炭化水素化合物および有機電界発光素子 |
| JP2002032468A (ja) | 2000-07-18 | 2002-01-31 | Canon Inc | 情報処理システム及び装置及びそれらの方法並びに記憶媒体 |
| JP2002063988A (ja) | 2000-08-22 | 2002-02-28 | Toray Ind Inc | 発光素子 |
| JP2002069044A (ja) | 2000-08-25 | 2002-03-08 | Idemitsu Kosan Co Ltd | 有機エレクトロルミネッセンス素子 |
| WO2002085822A1 (fr) | 2001-04-19 | 2002-10-31 | Tdk Corporation | Compose destine a un element organique electroluminescent (el) et element organique el |
| JP2002324678A (ja) | 2001-04-26 | 2002-11-08 | Fuji Photo Film Co Ltd | 発光素子 |
| JP2003272862A (ja) * | 2002-03-13 | 2003-09-26 | Fuji Photo Film Co Ltd | 発光素子 |
| JP2004002351A (ja) * | 2002-03-27 | 2004-01-08 | Tdk Corp | 有機el素子 |
| JP2004075567A (ja) | 2002-08-12 | 2004-03-11 | Idemitsu Kosan Co Ltd | オリゴアリーレン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
| JP2004083481A (ja) | 2002-08-27 | 2004-03-18 | Canon Inc | フルオレン化合物及びそれを用いた有機発光素子 |
| JP2004356033A (ja) | 2003-05-30 | 2004-12-16 | Tdk Corp | 有機el素子 |
| JP2005068087A (ja) | 2003-08-26 | 2005-03-17 | Mitsui Chemicals Inc | ベンゾフルオランテン化合物、および該ベンゾフルオランテン化合物を含有する有機電界発光素子 |
| WO2005033051A1 (de) | 2003-09-29 | 2005-04-14 | Basf Aktiengesellschaft | Synthese von phenylsubstituierten fluoranthenen durch diels-alder-reaktion und ihre verwendung |
| WO2005061656A1 (ja) | 2003-12-19 | 2005-07-07 | Idemitsu Kosan Co., Ltd. | 有機エレクトロルミネッセンス素子用発光材料、それを利用した有機エレクトロルミネッセンス素子及び有機エレクトロルミネッセンス素子用材料 |
| JP2005240008A (ja) * | 2004-01-27 | 2005-09-08 | Sony Corp | 有機発光材料および有機電界発光素子 |
| WO2005115950A1 (ja) | 2004-05-27 | 2005-12-08 | Idemitsu Kosan Co., Ltd. | 非対称ピレン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
Non-Patent Citations (2)
| Title |
|---|
| C. W. TANG; S. A. VANSLYKE, APPLIED PHYSICS LETTERS, vol. 51, 1987, pages 913 |
| See also references of EP1990844A4 |
Cited By (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009524653A (ja) * | 2006-01-27 | 2009-07-02 | エルジー・ケム・リミテッド | 新規なアントラセン誘導体、その製造方法およびこれを用いた有機電気素子 |
| WO2008015945A1 (en) | 2006-08-04 | 2008-02-07 | Canon Kabushiki Kaisha | ORGANIC LUMINESCENT DEVICE AND BENZO[k]FLUORANTHENE COMPOUND |
| US7977869B2 (en) | 2006-08-04 | 2011-07-12 | Canon Kabushiki Kaisha | Organic luminescent device and benzo[k]fluoranthene compound |
| EP2049616A4 (en) * | 2006-08-04 | 2012-10-17 | Canon Kk | ORGANIC LUMINESCENT DEVICE AND BENZO [K] FLUORANTHE COMPOUND |
| EP2049616A1 (en) * | 2006-08-04 | 2009-04-22 | Canon Kabushiki Kaisha | Organic luminescent device and benzo[k]fluoranthene compound |
| WO2008059713A1 (en) * | 2006-11-15 | 2008-05-22 | Idemitsu Kosan Co., Ltd. | Fluoranthene compound, organic electroluminescent device using the fluoranthene compound, and organic electroluminescent material-containing solution |
| US9120748B2 (en) | 2006-11-15 | 2015-09-01 | Idemitsu Kosan Co., Ltd. | Fluoranthene compound, organic electroluminescence device using the same, and solution containing organic electroluminescence material |
| JP2008137978A (ja) * | 2006-12-05 | 2008-06-19 | Canon Inc | カルバゾール誘導体及びこれを用いた有機発光素子 |
| JP2008214308A (ja) * | 2007-03-07 | 2008-09-18 | Canon Inc | 縮合環化合物及びこれを用いた有機発光素子 |
| WO2009008348A1 (ja) * | 2007-07-07 | 2009-01-15 | Idemitsu Kosan Co., Ltd. | 有機el素子 |
| JP2009267378A (ja) * | 2008-04-01 | 2009-11-12 | Canon Inc | 有機発光素子 |
| WO2009123344A1 (en) * | 2008-04-01 | 2009-10-08 | Canon Kabushiki Kaisha | Organic light-emitting device |
| WO2010058855A1 (en) * | 2008-11-19 | 2010-05-27 | Canon Kabushiki Kaisha | Novel organic compound, light-emitting device, and image display apparatus |
| US9153790B2 (en) | 2009-05-22 | 2015-10-06 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| US9812661B2 (en) | 2009-05-22 | 2017-11-07 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| WO2010134352A1 (ja) | 2009-05-22 | 2010-11-25 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2010134350A1 (ja) | 2009-05-22 | 2010-11-25 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| US8476823B2 (en) | 2009-05-22 | 2013-07-02 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| WO2010143434A1 (ja) | 2009-06-12 | 2010-12-16 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| EP3104428A1 (en) | 2009-06-12 | 2016-12-14 | Idemitsu Kosan Co., Ltd | Organic electroluminescence device |
| JPWO2011077691A1 (ja) * | 2009-12-21 | 2013-05-02 | 出光興産株式会社 | ピレン誘導体を用いた有機エレクトロルミネッセンス素子 |
| WO2011077691A1 (ja) * | 2009-12-21 | 2011-06-30 | 出光興産株式会社 | ピレン誘導体を用いた有機エレクトロルミネッセンス素子 |
| US9353027B2 (en) | 2009-12-21 | 2016-05-31 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent element using pyrene derivative |
| JPWO2011077689A1 (ja) * | 2009-12-21 | 2013-05-02 | 出光興産株式会社 | ピレン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 |
| JP5608682B2 (ja) * | 2009-12-21 | 2014-10-15 | 出光興産株式会社 | ピレン誘導体を用いた有機エレクトロルミネッセンス素子 |
| JP5645849B2 (ja) * | 2009-12-21 | 2014-12-24 | 出光興産株式会社 | ピレン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 |
| US9028976B2 (en) | 2010-05-28 | 2015-05-12 | Futaba Corporation | Organic EL device |
| JP2011249653A (ja) * | 2010-05-28 | 2011-12-08 | Tdk Corp | 有機el素子 |
| WO2014007287A1 (ja) | 2012-07-05 | 2014-01-09 | 東レ株式会社 | 発光素子材料および発光素子 |
| KR20150035706A (ko) | 2012-07-05 | 2015-04-07 | 도레이 카부시키가이샤 | 발광 소자 재료 및 발광 소자 |
| WO2014034864A1 (ja) * | 2012-08-31 | 2014-03-06 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2014034891A1 (ja) * | 2012-08-31 | 2014-03-06 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2014057873A1 (ja) * | 2012-10-10 | 2014-04-17 | 東レ株式会社 | ホスフィンオキサイド誘導体およびそれを有する発光素子 |
| WO2014057874A1 (ja) | 2012-10-12 | 2014-04-17 | 東レ株式会社 | フルオランテン誘導体、それを含有する発光素子材料および発光素子 |
| KR20150065175A (ko) | 2012-10-12 | 2015-06-12 | 도레이 카부시키가이샤 | 플루오란텐 유도체, 그것을 함유하는 발광 소자 재료 및 발광 소자 |
| WO2014073307A1 (ja) * | 2012-11-07 | 2014-05-15 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子および電子機器 |
| WO2014073306A1 (ja) * | 2012-11-07 | 2014-05-15 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子および電子機器 |
| KR101621801B1 (ko) * | 2015-01-14 | 2016-05-17 | 삼성디스플레이 주식회사 | 방향족 복소환 화합물 및 이를 포함한 유기막을 구비한 유기 발광 소자 |
| WO2017170313A1 (ja) | 2016-03-29 | 2017-10-05 | 住友化学株式会社 | 発光素子 |
| JP2017183723A (ja) * | 2016-03-29 | 2017-10-05 | 住友化学株式会社 | 発光素子 |
| JP7033397B2 (ja) | 2016-03-29 | 2022-03-10 | 住友化学株式会社 | 発光素子 |
| WO2017221822A1 (ja) | 2016-06-24 | 2017-12-28 | 住友化学株式会社 | 発光素子 |
| WO2021090932A1 (ja) * | 2019-11-08 | 2021-05-14 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び電子機器 |
| US11489128B1 (en) | 2019-11-08 | 2022-11-01 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent element emitting light at high luminous effiency and electronic device |
| WO2021201176A1 (ja) * | 2020-04-01 | 2021-10-07 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子、化合物及び電子機器 |
| WO2021210304A1 (ja) * | 2020-04-15 | 2021-10-21 | 出光興産株式会社 | 化合物、有機エレクトロルミネッセンス素子及び電子機器 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20130221333A1 (en) | 2013-08-29 |
| JPWO2007100010A1 (ja) | 2009-07-23 |
| CN101390230B (zh) | 2011-12-14 |
| TW200746895A (en) | 2007-12-16 |
| JP5199066B2 (ja) | 2013-05-15 |
| US9214636B2 (en) | 2015-12-15 |
| EP1990844A1 (en) | 2008-11-12 |
| KR101406631B1 (ko) | 2014-06-11 |
| CN102394273A (zh) | 2012-03-28 |
| TWI433595B (zh) | 2014-04-01 |
| EP1990844A4 (en) | 2010-05-26 |
| KR20080114702A (ko) | 2008-12-31 |
| US20070243411A1 (en) | 2007-10-18 |
| CN101390230A (zh) | 2009-03-18 |
| EP1990844B1 (en) | 2016-07-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101551591B1 (ko) | 방향족 아민 유도체 및 그들을 이용한 유기 전기 발광 소자 | |
| JP5475450B2 (ja) | 芳香族アミン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2007100010A1 (ja) | 有機エレクトロルミネッセンス素子 | |
| WO2008072400A1 (ja) | 芳香族アミン誘導体及びそれらを用いた有機エレクトロルミネッセンス素子 | |
| WO2008059713A1 (en) | Fluoranthene compound, organic electroluminescent device using the fluoranthene compound, and organic electroluminescent material-containing solution | |
| WO2007111263A1 (ja) | 含窒素複素環誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2006070712A1 (ja) | 有機エレクトロルミネッセンス素子用発光性インク組成物 | |
| WO2008032631A1 (fr) | Dérivé d'amine aromatique et dispositif électroluminescent organique l'employant | |
| WO2007102361A1 (ja) | 芳香族アミン誘導体及びそれらを用いた有機エレクトロルミネッセンス素子 | |
| KR20080105112A (ko) | 함질소 복소환 유도체 및 그것을 이용한 유기 전기발광 소자 | |
| WO2007018004A1 (ja) | 含窒素複素環誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2007080704A1 (ja) | 芳香族アミン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2007007463A1 (ja) | 電子吸引性置換基を有する含窒素複素環誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2006001333A1 (ja) | 多環芳香族系化合物、発光性塗膜形成用材料及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2007007464A1 (ja) | 含窒素複素環誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2008023759A1 (fr) | Dérivés d'amines aromatiques et dispositifs électroluminescents organiques utilisant ces mêmes amines | |
| WO2007099983A1 (ja) | フルオランテン誘導体及びインデノペリレン誘導体を用いた有機エレクトロルミネッセンス素子 | |
| WO2007018007A1 (ja) | 含窒素複素環誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| JP5084208B2 (ja) | 有機エレクトロルミネッセンス素子及び有機エレクトロルミネッセンス素子用材料 | |
| KR20090023411A (ko) | 방향족 아민 유도체 및 그들을 이용한 유기 전기발광 소자 | |
| WO2006006505A1 (ja) | 芳香族アミン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| WO2007105448A1 (ja) | ナフタセン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 | |
| JP2008162921A (ja) | ベンゾチアジアゾール誘導体及びそれを用いた有機エレクトロルミネッセンス素子用 | |
| KR20110117073A (ko) | 유기 전기발광 소자용 재료 및 그것을 사용한 유기 전기발광 소자 | |
| WO2007060795A1 (ja) | アミン系化合物及びそれを利用した有機エレクトロルミネッセンス素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| ENP | Entry into the national phase |
Ref document number: 2008502825 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2007737525 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007737525 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 4438/CHENP/2008 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780006855.3 Country of ref document: CN Ref document number: 1020087021039 Country of ref document: KR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |