EP2050841B1 - Alkalisches Galvanikbad mit einer Filtrationsmembran - Google Patents
Alkalisches Galvanikbad mit einer Filtrationsmembran Download PDFInfo
- Publication number
- EP2050841B1 EP2050841B1 EP09152660.8A EP09152660A EP2050841B1 EP 2050841 B1 EP2050841 B1 EP 2050841B1 EP 09152660 A EP09152660 A EP 09152660A EP 2050841 B1 EP2050841 B1 EP 2050841B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- bath
- filtration membrane
- electroplating bath
- zinc
- anode
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Revoked
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D3/00—Electroplating: Baths therefor
- C25D3/02—Electroplating: Baths therefor from solutions
- C25D3/56—Electroplating: Baths therefor from solutions of alloys
- C25D3/565—Electroplating: Baths therefor from solutions of alloys containing more than 50% by weight of zinc
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D17/00—Constructional parts, or assemblies thereof, of cells for electrolytic coating
- C25D17/02—Tanks; Installations therefor
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D17/00—Constructional parts, or assemblies thereof, of cells for electrolytic coating
- C25D17/002—Cell separation, e.g. membranes, diaphragms
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D21/00—Processes for servicing or operating cells for electrolytic coating
- C25D21/06—Filtering particles other than ions
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D21/00—Processes for servicing or operating cells for electrolytic coating
- C25D21/16—Regeneration of process solutions
- C25D21/22—Regeneration of process solutions by ion-exchange
Definitions
- the invention relates to an alkaline electroplating bath for applying zinc alloys to substrates, in which the anode space and the cathode space are separated from one another by a filtration membrane.
- zinc alloys can be deposited on substrates in a consistently high quality.
- the electroplating bath is operated with zinc alloy baths containing organic additives such as brighteners and wetting agents and complexing agents in addition to soluble zinc salts and optionally other metal salts selected from iron, nickel, cobalt and tin salts as defined in claim 1.
- Bath dilution reduces the concentration of impurities in proportion to the degree of dilution.
- a dilution is easy to carry out, but has the disadvantage that the amount of electrolyte removed from the bath has to be supplied to cost-intensive disposal.
- a complete new approach of the bath can be considered in this context as a special case of Badver Mednung.
- Alkaline Zn baths contain a factor of 5 to 10 lower proportion of organic additives as acidic baths. Accordingly, contamination by decomposition products is generally less critical. In the case of alkaline alloy baths, however, the addition of significant amounts of organic complexing agents is required to complex the alloying additive (Fe, Co, Ni, Sn). These are oxidatively degraded at the anode and the accumulated decomposition products have a negative effect on the production process.
- the EP 1 369 505 A2 discloses a method for purifying a zinc-nickel electrolyte in a galvanic process in which a portion of the process bath used in the process is evaporated until phase separation into a lower phase, at least one middle phase and an upper phase occurs, and the lower one and the upper phase are separated. This process requires several stages and is disadvantageous in terms of its energy requirements from a cost point of view.
- the WO 00/06807 and WO 01/96631 describe electroplating baths for applying zinc-nickel coatings. To avoid the undesirable decomposition of additives at the anode, it is proposed to separate the anode from the alkaline electrolyte through an ion exchange membrane.
- the baths known in the prior art have the disadvantage that in the anodic decomposition of the nitrogen-containing complexing agent cyanide is formed and accumulates in non-negligible concentration.
- the WO 2005/073438 which belongs to the prior art according to Article 54 (3) EPC, describes an apparatus for the electrolytic deposition of zinc or alloys of zinc with at least one other metal selected from iron, cobalt, nickel and manganese on a workpiece comprising: (a) a basin divided by a separator into a cathode compartment and an anode compartment; (b) the workpiece immersed in an aqueous alkaline-pH cathode liquid contained in the cathode compartment and comprising zincate ions and optionally ions of the metal other than zinc; and (c) an alkaline pH aqueous anode liquid contained in the anode compartment and surrounding an anode of the anode liquid insoluble metal; wherein the separator is made of an open-cell material and satisfies the following three tests: (1) the pore diameters have a dimension between 10 nm and 50 ⁇ m; (2) In a basin containing an aqueous solution of 120 g / l NaOH,
- the invention has for its object to provide an alkaline electroplating bath, which does not have the aforementioned disadvantages.
- the life of the bath is to be increased, the anodic decomposition of organic constituents of the bath to be minimized and, when it is used, a layer thickness of consistently high quality to be obtained on the coated substrate.
- the invention relates to an alkaline electroplating bath for applying zinc alloys on substrates having a cathode and an anode, comprising a zinc alloy bath, wherein the anode space and the cathode space are separated from each other by a filtration membrane, wherein the size of the pores of the filtration membrane in the range of 0.001 to 1 , 0 microns and wherein the zinc alloy bath comprises the following components: 80-250 g / l NaOH or KOH; 5-20 g / l zinc in the form of the soluble zinc salt; 0.02-10 g / l of the alloying metal Ni, Fe, Co, Sn in the form of the soluble metal salts; 2-200 g / l complexing agent selected from polyalkenylamines, alkanolamines, polyhydroxycarboxylates; 0.1-5 g / l aromatic or heteroaromatic brightener.
- filtration membranes are used.
- the size of the pores of this Filtration membranes is in a range of 0.001 to 1.0 microns, depending on the type of membrane (nano- or ultrafiltration membrane).
- filtration membranes are used with a pore size in the range of 0.05 to 0.5 microns.
- the pore size is in a range of 0.1 to 0, 3 microns.
- the filtration membrane contained in the alkaline electroplating bath according to the invention may consist of various organic or inorganic, alkali-resistant materials. These materials are, for example, ceramics, polytetrafluoroethylene (PTFE), polysulfones and polypropylene.
- PTFE polytetrafluoroethylene
- polysulfones polysulfones
- polypropylene polypropylene
- filtration membranes made of polypropylene.
- the filtration membrane in the alkaline electroplating bath according to the invention is designed as a flat membrane.
- the alkaline electroplating bath according to the invention can also be realized with other membrane forms, examples being hoses, capillaries and hollow fibers.
- Such baths are for example in US 5,417,840 .
- US 4,421,611 US 4,877,496 or US 6,652,728 described.
- the alkaline electroplating bath according to the invention has the advantage that in it also baths for the deposition of zinc alloys can be used, which are suitable for use in the from WO 00/06807 and WO 01/96631 known alkaline zinc-nickel bath with an ion exchange membrane are not suitable.
- sold by the applicant bath "Protedur Ni-75" to call which is characterized by a particularly high efficiency.
- the previously used anodes can be used further. These are mostly nickel anodes.
- the use of these anodes is less expensive than that from the WO 00/06807 known electroplating bath, in which special platinum-plated titanium anodes must be used in addition.
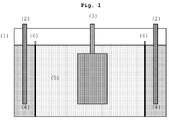
- the anode space is preferably made smaller than the cathode space, since the essential processes take place there.
- Both baths were operated with and without filtration membrane in 5-liter tanks.
- the filtration membrane used was the Abwa-Tec polymer membrane P150F, which has a pore size of 0.12 ⁇ m.
- the membrane was placed in the anode to cathode bath with the anolyte and catholyte being identical, ie, no special anolyte was added.
- iron sheets (7 ⁇ 10 cm), which are usually used for Hull cell tests, were used as workpieces to be coated and coated at a current density of 2 A / dm 2 .
- the baths were operated in serial connection. The movement of the iron sheets was mechanical, at a speed of 1.4 m / min.
- Table 2 shows the Hull cell layer thickness in a new approach and old approach depending on the throughput with and without filtration membrane. The layer thickness measurements were made after adjustment of the baths.
- the dots are on the Hullzellenblechen 3 cm from the bottom edge and 2.5 cm from the left or right side edge. On the left side is the high current density (point A) and on the right the low current density (point B).
- the average layer thickness is about 35% higher in a new batch in the high current density range and about 19% higher in the low current density range as if one had not used a filtration membrane. In the old batch, it is on average 17% and 12% higher than without filtration membrane.
- a filtration membrane is produced after a throughput of> 1000 Ah / l introduced after a short time comparable to a new approach current efficiency.
- composition of the aforementioned baths were analyzed according to the tests described above. Of particular interest was their cyanide content. This was much lower when using the baths according to the invention with a filtration membrane as baths without membrane. As shown in Table 4 below, a bath without the membrane had a cyanide content of 680 mg / L (new batch) and 790 mg / L (bath of> 1000 Ah / L), respectively, while the corresponding membrane baths contained a cyanide Content of 96 mg / L or 190 mg / L.
- the cyanide content of an old batch ie a bath with> 1000 Ah / l can be reduced when it is provided with a filtration membrane and operated.
- the cyanide content was reduced from 670 mg / l to 190 mg / l.
- the color of the baths was also evaluated. It was found that the color of a freshly prepared bath without membrane changed from initially violet-orange to brown within 15 Ah / l, whereby it remained purple or violet-orange over the entire time when using a filtration membrane. The old batch remained brown without using a membrane and the color changed to orange-brown after 15 Ah / l using a filtration membrane. Violet is also the color of freshly applied baths, which then turn to orange (after a few Ah / l) and at high throughput in brown.
- the voltage between anode and cathode was measured. It was about 3 V and was only about 50-100 mV higher in both approaches using a filtration membrane. Is used instead of the filtration membrane an ion exchange membrane, as in the WO 00/06807 described is, the voltage is higher by at least 500 mV. This again shows the advantage of using a filtration membrane instead of an ion exchange membrane.
- the use of filtration membranes over the use of ion exchange membranes offers many advantages.
- the coating process carried out therewith is more cost-effective, since no platinized anodes have to be used, catholyte and anolyte can have the same composition, and thus no circulation for the anolyte is required.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Electroplating And Plating Baths Therefor (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
- Electrodes For Compound Or Non-Metal Manufacture (AREA)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP09152660.8A EP2050841B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
| ES09152660.8T ES2574158T3 (es) | 2005-04-26 | 2005-04-26 | Baño galvánico alcalino con una membrana de filtración |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP05009127A EP1717353B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
| EP09152660.8A EP2050841B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05009127A Division EP1717353B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2050841A1 EP2050841A1 (de) | 2009-04-22 |
| EP2050841B1 true EP2050841B1 (de) | 2016-05-11 |
Family
ID=35530823
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05009127A Active EP1717353B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
| EP09152660.8A Revoked EP2050841B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05009127A Active EP1717353B1 (de) | 2005-04-26 | 2005-04-26 | Alkalisches Galvanikbad mit einer Filtrationsmembran |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US8293092B2 (ja) |
| EP (2) | EP1717353B1 (ja) |
| JP (1) | JP4955657B2 (ja) |
| KR (1) | KR101301275B1 (ja) |
| CN (3) | CN104911676B (ja) |
| AT (1) | ATE429528T1 (ja) |
| BR (1) | BRPI0610765B1 (ja) |
| CA (1) | CA2600273C (ja) |
| DE (1) | DE502005007138D1 (ja) |
| ES (2) | ES2324169T3 (ja) |
| WO (1) | WO2006114305A1 (ja) |
Families Citing this family (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090107545A1 (en) * | 2006-10-09 | 2009-04-30 | Soltaix, Inc. | Template for pyramidal three-dimensional thin-film solar cell manufacturing and methods of use |
| ITTO20070704A1 (it) | 2007-10-05 | 2009-04-06 | Create New Technology S R L | Sistema e metodo di placcatura di leghe metalliche mediante tecnologia galvanica |
| US8177944B2 (en) | 2007-12-04 | 2012-05-15 | Ebara Corporation | Plating apparatus and plating method |
| DE102008056776A1 (de) | 2008-11-11 | 2010-05-12 | Enthone Inc., West Haven | Galvanisches Bad und Verfahren zur Abscheidung von zinkhaltigen Schichten |
| ES2404844T3 (es) | 2010-05-07 | 2013-05-29 | Dr.Ing. Max Schlötter Gmbh & Co. Kg | Regeneración de electrolitos de cinc-níquel alcalinos mediante la eliminación de iones cianuro |
| IT1405319B1 (it) * | 2010-12-27 | 2014-01-03 | Fontana R D S R L | Procedimento di rivestimento di pezzi metallici filettati |
| KR101420865B1 (ko) * | 2012-10-12 | 2014-07-18 | 주식회사 익스톨 | 금속 도금장치 |
| EP2784189A1 (en) | 2013-03-28 | 2014-10-01 | Coventya SAS | Electroplating bath for zinc-iron alloys, method for depositing zinc-iron alloy on a device and such a device |
| JP6142408B2 (ja) | 2015-03-13 | 2017-06-07 | 奥野製薬工業株式会社 | 治具用電解剥離剤 |
| BR112015028629A2 (pt) * | 2015-07-22 | 2017-07-25 | Dipsol Chem | método de eletrogalvanização de liga de zinco |
| EP3042985B1 (en) | 2015-07-22 | 2019-04-10 | Dipsol Chemicals Co., Ltd. | Zinc alloy plating method |
| CA3024991A1 (en) * | 2016-05-24 | 2017-11-30 | Coventya, Inc. | Ternary zinc-nickel-iron alloys and alkaline electrolytes for plating such alloys |
| CA3032224A1 (en) * | 2016-07-29 | 2018-02-01 | Simon Fraser University | Methods of electrochemical deposition |
| EP3358045A1 (de) * | 2017-02-07 | 2018-08-08 | Dr.Ing. Max Schlötter GmbH & Co. KG | Verfahren zur galvanischen abscheidung von zink- und zinklegierungsüberzügen aus einem alkalischen beschichtungsbad mit reduziertem abbau von organischen badzusätzen |
| ES2969188T3 (es) | 2017-06-14 | 2024-05-16 | Dr Ing Max Schloetter Gmbh & Co Kg | Procedimiento para la deposición galvánica de revestimientos de aleaciones de cinc-níquel a partir de un baño de aleación de cinc-níquel alcalino con degradación reducida de aditivos |
| EP3461933B1 (en) | 2017-09-28 | 2019-09-04 | ATOTECH Deutschland GmbH | Method for electrolytically depositing a zinc-nickel alloy layer on at least a substrate to be treated |
| US11165091B2 (en) | 2018-01-23 | 2021-11-02 | City University Of Hong Kong | Battery system and a method of forming a battery |
| CA3127517A1 (en) * | 2019-01-24 | 2020-07-30 | Atotech Deutschland Gmbh | Membrane anode system for electrolytic zinc-nickel alloy deposition |
| WO2020166062A1 (ja) | 2019-02-15 | 2020-08-20 | ディップソール株式会社 | 亜鉛又は亜鉛合金電気めっき方法及びシステム |
| RU2712582C1 (ru) * | 2019-07-16 | 2020-01-29 | Федеральное государственное бюджетное образовательное учреждение высшего образования "Ивановский государственный химико-технологический университет" | Электролит для электроосаждения цинк-железных покрытий |
| EP4273303A1 (en) * | 2022-05-05 | 2023-11-08 | Atotech Deutschland GmbH & Co. KG | Method for depositing a zinc-nickel alloy on a substrate, an aqueous zinc-nickel deposition bath, a brightening agent and use thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3945900A (en) | 1972-05-02 | 1976-03-23 | Dorr-Oliver Incorporated | Electro ultrafiltration process and apparatus |
| EP0649918A1 (en) | 1993-10-21 | 1995-04-26 | Mcgean-Rohco, Inc. | Alkaline zinc-nickel alloy plating baths |
| US5616246A (en) | 1990-10-30 | 1997-04-01 | Minnestoa Mining & Manufacturing Company | Hydrophilic membranes for electrochemical devices and method for preparing same |
| US6383352B1 (en) | 1998-11-13 | 2002-05-07 | Mykrolis Corporation | Spiral anode for metal plating baths |
| EP1553211A1 (en) | 2002-07-25 | 2005-07-13 | Shinryo Electronics Co., Ltd. | Tin-silver-copper plating solution, plating film containing the same, and method for forming the plating film |
Family Cites Families (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB381931A (en) * | 1931-07-11 | 1932-10-11 | Mond Nickel Co Ltd | Improvements relating to electro-plating and the electrodeposition of metals |
| IE39814B1 (en) * | 1973-08-03 | 1979-01-03 | Parel Sa | Electrochemical process and apparatus |
| US4250002A (en) * | 1979-09-19 | 1981-02-10 | Hooker Chemicals & Plastics Corp. | Polymeric microporous separators for use in electrolytic processes and devices |
| US4421611A (en) | 1982-09-30 | 1983-12-20 | Mcgean-Rohco, Inc. | Acetylenic compositions and nickel plating baths containing same |
| JPS6353285A (ja) | 1986-08-22 | 1988-03-07 | Nippon Hyomen Kagaku Kk | 亜鉛−ニツケル合金めつき液 |
| JPH01116094A (ja) * | 1987-10-28 | 1989-05-09 | Eagle Ind Co Ltd | 隔膜鍍金法 |
| JPH02141596A (ja) * | 1988-11-21 | 1990-05-30 | Yuken Kogyo Kk | ジンケート型亜鉛合金メッキ浴 |
| JPH0444375A (ja) * | 1990-06-12 | 1992-02-14 | Zexel Corp | レーザ共振器のアライメント装置 |
| US5082538A (en) | 1991-01-09 | 1992-01-21 | Eltech Systems Corporation | Process for replenishing metals in aqueous electrolyte solutions |
| CN2175238Y (zh) * | 1993-09-29 | 1994-08-24 | 北京科技大学 | 锌-镍合金电镀用阳极 |
| US5631102A (en) * | 1996-02-12 | 1997-05-20 | Wilson Greatbatch Ltd. | Separator insert for electrochemical cells |
| JPH11200099A (ja) | 1998-01-08 | 1999-07-27 | Toyo Kohan Co Ltd | 不溶性陽極を用いるめっき方法およびめっき装置 |
| DE19834353C2 (de) | 1998-07-30 | 2000-08-17 | Hillebrand Walter Gmbh & Co Kg | Alkalisches Zink-Nickelbad |
| DE19840019C1 (de) | 1998-09-02 | 2000-03-16 | Atotech Deutschland Gmbh | Wäßriges alkalisches cyanidfreies Bad zur galvanischen Abscheidung von Zink- oder Zinklegierungsüberzügen sowie Verfahren |
| JP2000087299A (ja) * | 1998-09-08 | 2000-03-28 | Ebara Corp | 基板メッキ装置 |
| JP4060012B2 (ja) * | 1999-07-19 | 2008-03-12 | 日本エレクトロプレイテイング・エンジニヤース株式会社 | カップ式めっき装置 |
| DE60023190T3 (de) | 2000-06-15 | 2016-03-10 | Coventya, Inc. | Zink-nickel-elektroplattierung |
| FR2839729B1 (fr) * | 2002-05-16 | 2005-02-11 | Univ Toulouse | Procede de protection d'un substrat en acier ou alliage d'aluminium contre la corrosion permettant de lui conferer des proprietes tribologiques, et substrat obtenu |
| DE10225203A1 (de) | 2002-06-06 | 2003-12-18 | Goema Ag | Verfahren und Vorrichtung zur Spülwasserrückführung und Reinigung eines Prozessbades |
| EP1639155B1 (en) | 2003-06-03 | 2016-11-02 | Coventya, Inc. | Zinc and zinc-alloy electroplating |
| JP4120497B2 (ja) * | 2003-06-27 | 2008-07-16 | Jfeスチール株式会社 | 電気亜鉛系めっき鋼板 |
| FR2864553B1 (fr) | 2003-12-31 | 2006-09-01 | Coventya | Installation de depot de zinc ou d'alliages de zinc |
| US7442286B2 (en) * | 2004-02-26 | 2008-10-28 | Atotech Deutschland Gmbh | Articles with electroplated zinc-nickel ternary and higher alloys, electroplating baths, processes and systems for electroplating such alloys |
-
2005
- 2005-04-26 ES ES05009127T patent/ES2324169T3/es active Active
- 2005-04-26 AT AT05009127T patent/ATE429528T1/de not_active IP Right Cessation
- 2005-04-26 EP EP05009127A patent/EP1717353B1/de active Active
- 2005-04-26 DE DE502005007138T patent/DE502005007138D1/de active Active
- 2005-04-26 ES ES09152660.8T patent/ES2574158T3/es active Active
- 2005-04-26 EP EP09152660.8A patent/EP2050841B1/de not_active Revoked
-
2006
- 2006-04-26 CN CN201510173898.7A patent/CN104911676B/zh active Active
- 2006-04-26 CN CNA2006800094924A patent/CN101146934A/zh active Pending
- 2006-04-26 CA CA2600273A patent/CA2600273C/en active Active
- 2006-04-26 KR KR1020077019889A patent/KR101301275B1/ko active IP Right Grant
- 2006-04-26 BR BRPI0610765A patent/BRPI0610765B1/pt active IP Right Grant
- 2006-04-26 CN CN201510173915.7A patent/CN104911651A/zh active Pending
- 2006-04-26 JP JP2008508150A patent/JP4955657B2/ja active Active
- 2006-04-26 WO PCT/EP2006/003883 patent/WO2006114305A1/en active Application Filing
- 2006-04-26 US US11/912,591 patent/US8293092B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3945900A (en) | 1972-05-02 | 1976-03-23 | Dorr-Oliver Incorporated | Electro ultrafiltration process and apparatus |
| US5616246A (en) | 1990-10-30 | 1997-04-01 | Minnestoa Mining & Manufacturing Company | Hydrophilic membranes for electrochemical devices and method for preparing same |
| EP0649918A1 (en) | 1993-10-21 | 1995-04-26 | Mcgean-Rohco, Inc. | Alkaline zinc-nickel alloy plating baths |
| US6383352B1 (en) | 1998-11-13 | 2002-05-07 | Mykrolis Corporation | Spiral anode for metal plating baths |
| EP1553211A1 (en) | 2002-07-25 | 2005-07-13 | Shinryo Electronics Co., Ltd. | Tin-silver-copper plating solution, plating film containing the same, and method for forming the plating film |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2008539329A (ja) | 2008-11-13 |
| DE502005007138D1 (de) | 2009-06-04 |
| US8293092B2 (en) | 2012-10-23 |
| BRPI0610765B1 (pt) | 2017-04-04 |
| JP4955657B2 (ja) | 2012-06-20 |
| ATE429528T1 (de) | 2009-05-15 |
| CN101146934A (zh) | 2008-03-19 |
| CN104911676B (zh) | 2017-11-17 |
| CN104911676A (zh) | 2015-09-16 |
| ES2324169T3 (es) | 2009-07-31 |
| EP1717353A1 (de) | 2006-11-02 |
| KR101301275B1 (ko) | 2013-08-29 |
| US20090107845A1 (en) | 2009-04-30 |
| CA2600273A1 (en) | 2006-11-02 |
| CA2600273C (en) | 2014-08-12 |
| EP2050841A1 (de) | 2009-04-22 |
| CN104911651A (zh) | 2015-09-16 |
| WO2006114305A1 (en) | 2006-11-02 |
| ES2574158T3 (es) | 2016-06-15 |
| BRPI0610765A2 (pt) | 2010-07-20 |
| KR20070122454A (ko) | 2007-12-31 |
| EP1717353B1 (de) | 2009-04-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2050841B1 (de) | Alkalisches Galvanikbad mit einer Filtrationsmembran | |
| EP0862665B1 (de) | Verfahren zur elektrolytischen abscheidung von metallschichten | |
| EP2116634B1 (de) | Modifizierter Kupfer-Zinn-Elektrolyt und Verfahren zur Abscheidung von Bronzeschichten | |
| EP3666931A1 (de) | Verfahren zur herstellung eines mit einer beschichtung aus chrom und chromoxid beschichteten metallbands auf basis einer elektrolytlösung mit einer dreiwertigen chromverbindung | |
| EP3481976B1 (de) | Verfahren zur galvanischen abscheidung von zink- und zinklegierungsüberzügen aus einem alkalischen beschichtungsbad mit reduziertem abbau von organischen badzusätzen | |
| DE4023444A1 (de) | Cyanid-freies verfahren zur herstellung eines galvanischen kupferueberzuges | |
| DE102010044551A1 (de) | Anode sowie deren Verwendung in einem alkalischen Galvanikbad | |
| EP3250733B1 (de) | Herstellung von chromschichten auf tiefdruckzylindern | |
| EP3067444B1 (de) | Abscheidung von dekorativen palladium-eisen-legierungsbeschichtungen auf metallischen substanzen | |
| EP2635724A1 (de) | Verfahren zur abscheidung von hartchrom aus cr(vi)-freien elektrolyten | |
| DE3029364A1 (de) | Verfahren zur herstellung von kathoden mit niedriger wasserstoffueberspannung und ihre verwendung | |
| EP1080252B1 (de) | Verfahren zur galvanischen verkupferung von substraten | |
| EP3415665B1 (de) | Verfahren zur galvanischen abscheidung von zink-nickel-legierungsüberzügen aus einem alkalischen zink-nickel-legierungsbad mit reduziertem abbau von additiven | |
| EP2384800B1 (de) | Regeneration alkalischer Zinknickelelektrolyte durch Entfernen von Cyanidionen | |
| DE102009023124A1 (de) | Verfahren zur galvanischen Kupferbeschichtung und Vorrichtung zur Durchführung eines solchen Verfahrens | |
| DE102019202899B3 (de) | Wässrige Formulierung zum Herstellen einer Schicht aus Gold und Silber | |
| DE2646697A1 (de) | Verfahren zum behandeln von verzinktem metallband | |
| DE2929305C2 (de) | Verfahren und Vorrichtung zur kontinuierlichen galvanischen Abscheidung von Mangan auf Stahl | |
| DE19610361A1 (de) | Bad und Verfahren für die galvanische Abscheidung von Halbglanznickel | |
| EP2878711A1 (de) | Verfahren zur galvanischen Abscheidung von Nickel und entsprechender Elektrolyt | |
| DE2063991A1 (de) | Wässriges Chrombad und Verfahren zum Verchromen | |
| AT222969B (de) | Verfahren und Lösung zur elektrolytischen Abscheidung einer Legierung auf Metalloberflächen | |
| WO2023041670A1 (de) | Chrom-indium-, chrom-bismut- und chrom-antimon-beschichtung, verfahren zur herstellung und verwendung | |
| DE102020131371A1 (de) | Rutheniumlegierungsschicht und deren Schichtkombinationen | |
| EP2431500A1 (de) | Regeneration alkalischer Zinknickelelektrolyte durch Entfernen von Cyanidionen mit Hilfe von löslichen quartären Ammoniumverbindungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 1717353 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU MC NL PL PT RO SE SI SK TR |
|
| 17P | Request for examination filed |
Effective date: 20090923 |
|
| 17Q | First examination report despatched |
Effective date: 20091022 |
|
| AKX | Designation fees paid |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU MC NL PL PT RO SE SI SK TR |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: ATOTECH DEUTSCHLAND GMBH |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20151216 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 1717353 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LI LT LU MC NL PL PT RO SE SI SK TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 798744 Country of ref document: AT Kind code of ref document: T Effective date: 20160515 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: GERMAN |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2574158 Country of ref document: ES Kind code of ref document: T3 Effective date: 20160615 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 502005015224 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20160511 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160912 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160812 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R026 Ref document number: 502005015224 Country of ref document: DE |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| 26 | Opposition filed |
Opponent name: CONVENTYA HOLDING SAS Effective date: 20170208 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 13 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20170426 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170430 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170426 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170430 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170426 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20170430 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 14 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170426 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170430 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MM01 Ref document number: 798744 Country of ref document: AT Kind code of ref document: T Effective date: 20170426 |
|
| PLCK | Communication despatched that opposition was rejected |
Free format text: ORIGINAL CODE: EPIDOSNREJ1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170426 |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |
|
| APBM | Appeal reference recorded |
Free format text: ORIGINAL CODE: EPIDOSNREFNO |
|
| APBP | Date of receipt of notice of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA2O |
|
| APBQ | Date of receipt of statement of grounds of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA3O |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20050426 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160511 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160511 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160911 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CZ Payment date: 20210426 Year of fee payment: 17 Ref country code: DE Payment date: 20210420 Year of fee payment: 17 Ref country code: IT Payment date: 20210427 Year of fee payment: 17 Ref country code: FR Payment date: 20210423 Year of fee payment: 17 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20210621 Year of fee payment: 17 |
|
| APBU | Appeal procedure closed |
Free format text: ORIGINAL CODE: EPIDOSNNOA9O |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R103 Ref document number: 502005015224 Country of ref document: DE Ref country code: DE Ref legal event code: R064 Ref document number: 502005015224 Country of ref document: DE |
|
| RDAF | Communication despatched that patent is revoked |
Free format text: ORIGINAL CODE: EPIDOSNREV1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT REVOKED |
|
| RDAG | Patent revoked |
Free format text: ORIGINAL CODE: 0009271 |
|
| REG | Reference to a national code |
Ref country code: FI Ref legal event code: MGE |
|
| 27W | Patent revoked |
Effective date: 20211029 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MA03 Ref document number: 798744 Country of ref document: AT Kind code of ref document: T Effective date: 20211029 |