CN100521972C - 用于促进骨生长及维持骨健康的组合物 - Google Patents
用于促进骨生长及维持骨健康的组合物 Download PDFInfo
- Publication number
- CN100521972C CN100521972C CNB028245172A CN02824517A CN100521972C CN 100521972 C CN100521972 C CN 100521972C CN B028245172 A CNB028245172 A CN B028245172A CN 02824517 A CN02824517 A CN 02824517A CN 100521972 C CN100521972 C CN 100521972C
- Authority
- CN
- China
- Prior art keywords
- bone
- aerial part
- plant
- iris
- root
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 66
- 230000037180 bone health Effects 0.000 title claims abstract description 13
- 230000008468 bone growth Effects 0.000 title abstract description 8
- 238000012423 maintenance Methods 0.000 title abstract 3
- 239000003814 drug Substances 0.000 claims abstract description 14
- 208000020084 Bone disease Diseases 0.000 claims abstract description 10
- 235000016709 nutrition Nutrition 0.000 claims abstract description 10
- 230000002265 prevention Effects 0.000 claims abstract description 8
- 210000000988 bone and bone Anatomy 0.000 claims description 51
- 241000196324 Embryophyta Species 0.000 claims description 43
- 235000013305 food Nutrition 0.000 claims description 43
- 239000000419 plant extract Substances 0.000 claims description 29
- 241000234653 Cyperus Species 0.000 claims description 17
- 235000013399 edible fruits Nutrition 0.000 claims description 16
- 240000006891 Artemisia vulgaris Species 0.000 claims description 12
- 235000007542 Cichorium intybus Nutrition 0.000 claims description 12
- 244000298479 Cichorium intybus Species 0.000 claims description 12
- 235000003261 Artemisia vulgaris Nutrition 0.000 claims description 10
- 206010065687 Bone loss Diseases 0.000 claims description 10
- 235000009027 Amelanchier alnifolia Nutrition 0.000 claims description 9
- 244000068687 Amelanchier alnifolia Species 0.000 claims description 9
- 244000020518 Carthamus tinctorius Species 0.000 claims description 9
- 235000005747 Carum carvi Nutrition 0.000 claims description 9
- 240000000467 Carum carvi Species 0.000 claims description 9
- 235000014749 Mentha crispa Nutrition 0.000 claims description 9
- 240000007926 Ocimum gratissimum Species 0.000 claims description 9
- 240000001949 Taraxacum officinale Species 0.000 claims description 9
- 102000007350 Bone Morphogenetic Proteins Human genes 0.000 claims description 8
- 108010007726 Bone Morphogenetic Proteins Proteins 0.000 claims description 8
- 235000003255 Carthamus tinctorius Nutrition 0.000 claims description 8
- 244000163122 Curcuma domestica Species 0.000 claims description 8
- 244000178231 Rosmarinus officinalis Species 0.000 claims description 8
- 229940112869 bone morphogenetic protein Drugs 0.000 claims description 8
- 230000035764 nutrition Effects 0.000 claims description 8
- 244000205574 Acorus calamus Species 0.000 claims description 7
- 235000015265 Iris pallida Nutrition 0.000 claims description 7
- 240000004101 Iris pallida Species 0.000 claims description 7
- 240000006365 Vitis vinifera Species 0.000 claims description 7
- 235000014787 Vitis vinifera Nutrition 0.000 claims description 7
- 230000000975 bioactive effect Effects 0.000 claims description 7
- 238000004113 cell culture Methods 0.000 claims description 7
- 235000003373 curcuma longa Nutrition 0.000 claims description 7
- 229940124597 therapeutic agent Drugs 0.000 claims description 7
- GLZPCOQZEFWAFX-UHFFFAOYSA-N Geraniol Chemical compound CC(C)=CCCC(C)=CCO GLZPCOQZEFWAFX-UHFFFAOYSA-N 0.000 claims description 6
- 241001633663 Iris pseudacorus Species 0.000 claims description 6
- ULDHMXUKGWMISQ-UHFFFAOYSA-N carvone Chemical compound CC(=C)C1CC=C(C)C(=O)C1 ULDHMXUKGWMISQ-UHFFFAOYSA-N 0.000 claims description 6
- 235000015872 dietary supplement Nutrition 0.000 claims description 6
- 239000008194 pharmaceutical composition Substances 0.000 claims description 6
- 235000017807 phytochemicals Nutrition 0.000 claims description 6
- 229930000223 plant secondary metabolite Natural products 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 6
- 235000006011 Bixa Nutrition 0.000 claims description 5
- 241000934840 Bixa Species 0.000 claims description 5
- 244000024873 Mentha crispa Species 0.000 claims description 5
- 235000004066 Ocimum gratissimum Nutrition 0.000 claims description 5
- 235000005187 Taraxacum officinale ssp. officinale Nutrition 0.000 claims description 5
- 240000002657 Thymus vulgaris Species 0.000 claims description 5
- 235000007303 Thymus vulgaris Nutrition 0.000 claims description 5
- 239000002253 acid Substances 0.000 claims description 5
- 210000000845 cartilage Anatomy 0.000 claims description 5
- 210000001541 thymus gland Anatomy 0.000 claims description 5
- 239000001585 thymus vulgaris Substances 0.000 claims description 5
- 235000009051 Ambrosia paniculata var. peruviana Nutrition 0.000 claims description 4
- 244000137121 Amelanchier vulgaris Species 0.000 claims description 4
- 235000001311 Amelanchier vulgaris Nutrition 0.000 claims description 4
- 235000011446 Amygdalus persica Nutrition 0.000 claims description 4
- 235000003097 Artemisia absinthium Nutrition 0.000 claims description 4
- 235000017731 Artemisia dracunculus ssp. dracunculus Nutrition 0.000 claims description 4
- 235000004355 Artemisia lactiflora Nutrition 0.000 claims description 4
- 235000003392 Curcuma domestica Nutrition 0.000 claims description 4
- 240000005153 Dodonaea viscosa Species 0.000 claims description 4
- 244000148992 Lindera benzoin Species 0.000 claims description 4
- 235000004520 Lindera benzoin Nutrition 0.000 claims description 4
- 244000078639 Mentha spicata Species 0.000 claims description 4
- 240000005809 Prunus persica Species 0.000 claims description 4
- 235000015360 Taraxacum officinale ssp. ceratophorum Nutrition 0.000 claims description 4
- 239000001138 artemisia absinthium Substances 0.000 claims description 4
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 claims description 4
- 235000002532 grape seed extract Nutrition 0.000 claims description 4
- 239000001220 mentha spicata Substances 0.000 claims description 4
- 201000008482 osteoarthritis Diseases 0.000 claims description 4
- KDYFGRWQOYBRFD-UHFFFAOYSA-N succinic acid Chemical compound OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 claims description 4
- 235000013976 turmeric Nutrition 0.000 claims description 4
- JCZPMGDSEAFWDY-SQOUGZDYSA-N (2r,3s,4r,5r)-2,3,4,5,6-pentahydroxyhexanamide Chemical compound NC(=O)[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO JCZPMGDSEAFWDY-SQOUGZDYSA-N 0.000 claims description 3
- ULDHMXUKGWMISQ-VIFPVBQESA-N (S)-(+)-Carvone Natural products CC(=C)[C@H]1CC=C(C)C(=O)C1 ULDHMXUKGWMISQ-VIFPVBQESA-N 0.000 claims description 3
- SQDAZGGFXASXDW-UHFFFAOYSA-N 5-bromo-2-(trifluoromethoxy)pyridine Chemical compound FC(F)(F)OC1=CC=C(Br)C=N1 SQDAZGGFXASXDW-UHFFFAOYSA-N 0.000 claims description 3
- 235000006480 Acorus calamus Nutrition 0.000 claims description 3
- 244000144730 Amygdalus persica Species 0.000 claims description 3
- 235000011996 Calamus deerratus Nutrition 0.000 claims description 3
- 235000009024 Ceanothus sanguineus Nutrition 0.000 claims description 3
- 229920001287 Chondroitin sulfate Polymers 0.000 claims description 3
- 235000007516 Chrysanthemum Nutrition 0.000 claims description 3
- 244000035851 Chrysanthemum leucanthemum Species 0.000 claims description 3
- 235000008495 Chrysanthemum leucanthemum Nutrition 0.000 claims description 3
- 244000189548 Chrysanthemum x morifolium Species 0.000 claims description 3
- 241001362421 Epimedium brevicornu Species 0.000 claims description 3
- 241001527234 Eriogonum giganteum Species 0.000 claims description 3
- 240000006927 Foeniculum vulgare Species 0.000 claims description 3
- 235000004204 Foeniculum vulgare Nutrition 0.000 claims description 3
- 239000005792 Geraniol Substances 0.000 claims description 3
- GLZPCOQZEFWAFX-YFHOEESVSA-N Geraniol Natural products CC(C)=CCC\C(C)=C/CO GLZPCOQZEFWAFX-YFHOEESVSA-N 0.000 claims description 3
- 240000000015 Iris germanica Species 0.000 claims description 3
- 235000002971 Iris x germanica Nutrition 0.000 claims description 3
- 241000721662 Juniperus Species 0.000 claims description 3
- 235000008119 Larix laricina Nutrition 0.000 claims description 3
- 244000193510 Larix occidentalis Species 0.000 claims description 3
- 235000008122 Larix occidentalis Nutrition 0.000 claims description 3
- 240000003553 Leptospermum scoparium Species 0.000 claims description 3
- 235000015459 Lycium barbarum Nutrition 0.000 claims description 3
- 241001499733 Plantago asiatica Species 0.000 claims description 3
- 235000009769 Populus balsamifera subsp. balsamifera Nutrition 0.000 claims description 3
- 235000006040 Prunus persica var persica Nutrition 0.000 claims description 3
- 235000013234 Rhus glabra Nutrition 0.000 claims description 3
- 240000008599 Rhus glabra Species 0.000 claims description 3
- 235000016639 Syzygium aromaticum Nutrition 0.000 claims description 3
- 244000223014 Syzygium aromaticum Species 0.000 claims description 3
- 235000006754 Taraxacum officinale Nutrition 0.000 claims description 3
- 244000299461 Theobroma cacao Species 0.000 claims description 3
- 235000009754 Vitis X bourquina Nutrition 0.000 claims description 3
- 235000012333 Vitis X labruscana Nutrition 0.000 claims description 3
- 125000001931 aliphatic group Chemical group 0.000 claims description 3
- 239000003963 antioxidant agent Substances 0.000 claims description 3
- 230000003078 antioxidant effect Effects 0.000 claims description 3
- 235000006708 antioxidants Nutrition 0.000 claims description 3
- 229940059329 chondroitin sulfate Drugs 0.000 claims description 3
- 239000000835 fiber Substances 0.000 claims description 3
- 229940113087 geraniol Drugs 0.000 claims description 3
- 239000004922 lacquer Substances 0.000 claims description 3
- VJQAFLAZRVKAKM-QQUHWDOBSA-N lactucin Chemical compound O[C@@H]1CC(C)=C2C(=O)C=C(CO)[C@@H]2[C@H]2OC(=O)C(=C)[C@@H]21 VJQAFLAZRVKAKM-QQUHWDOBSA-N 0.000 claims description 3
- VJQAFLAZRVKAKM-UHFFFAOYSA-N lactucine Natural products OC1CC(C)=C2C(=O)C=C(CO)C2C2OC(=O)C(=C)C21 VJQAFLAZRVKAKM-UHFFFAOYSA-N 0.000 claims description 3
- 235000015639 rosmarinus officinalis Nutrition 0.000 claims description 3
- 241000592238 Juniperus communis Species 0.000 claims description 2
- 244000010922 Plantago major Species 0.000 claims 2
- 235000015266 Plantago major Nutrition 0.000 claims 2
- 238000000034 method Methods 0.000 abstract description 30
- 239000000047 product Substances 0.000 abstract description 6
- 238000004519 manufacturing process Methods 0.000 abstract description 3
- 230000001737 promoting effect Effects 0.000 abstract 1
- 239000013589 supplement Substances 0.000 abstract 1
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 63
- 239000000284 extract Substances 0.000 description 36
- 230000000694 effects Effects 0.000 description 32
- 102000008143 Bone Morphogenetic Protein 2 Human genes 0.000 description 23
- 108010049931 Bone Morphogenetic Protein 2 Proteins 0.000 description 23
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 22
- 244000068988 Glycine max Species 0.000 description 11
- 210000004027 cell Anatomy 0.000 description 11
- 239000006041 probiotic Substances 0.000 description 11
- 230000000529 probiotic effect Effects 0.000 description 11
- 235000018291 probiotics Nutrition 0.000 description 11
- 238000010521 absorption reaction Methods 0.000 description 10
- 235000015097 nutrients Nutrition 0.000 description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 10
- 235000010469 Glycine max Nutrition 0.000 description 9
- 238000009472 formulation Methods 0.000 description 9
- 235000019197 fats Nutrition 0.000 description 8
- 210000000554 iris Anatomy 0.000 description 7
- 230000011164 ossification Effects 0.000 description 7
- 108090000623 proteins and genes Proteins 0.000 description 7
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- 241000124008 Mammalia Species 0.000 description 5
- 150000001720 carbohydrates Chemical class 0.000 description 5
- 235000014633 carbohydrates Nutrition 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 239000000262 estrogen Substances 0.000 description 5
- 229940011871 estrogen Drugs 0.000 description 5
- 230000001939 inductive effect Effects 0.000 description 5
- 244000005700 microbiome Species 0.000 description 5
- 102000004169 proteins and genes Human genes 0.000 description 5
- 238000012216 screening Methods 0.000 description 5
- 235000013343 vitamin Nutrition 0.000 description 5
- 239000011782 vitamin Substances 0.000 description 5
- 229940088594 vitamin Drugs 0.000 description 5
- 229930003231 vitamin Natural products 0.000 description 5
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 4
- 235000010676 Ocimum basilicum Nutrition 0.000 description 4
- 241000404542 Tanacetum Species 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 4
- 229960004844 lovastatin Drugs 0.000 description 4
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 210000002997 osteoclast Anatomy 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 235000018102 proteins Nutrition 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 150000003722 vitamin derivatives Chemical class 0.000 description 4
- 108010010256 Dietary Proteins Proteins 0.000 description 3
- 102000015781 Dietary Proteins Human genes 0.000 description 3
- 108060001084 Luciferase Proteins 0.000 description 3
- 208000001132 Osteoporosis Diseases 0.000 description 3
- 241001127637 Plantago Species 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 235000013339 cereals Nutrition 0.000 description 3
- 239000000306 component Substances 0.000 description 3
- 235000009508 confectionery Nutrition 0.000 description 3
- 235000021245 dietary protein Nutrition 0.000 description 3
- 239000004459 forage Substances 0.000 description 3
- 235000006539 genistein Nutrition 0.000 description 3
- TZBJGXHYKVUXJN-UHFFFAOYSA-N genistein Natural products C1=CC(O)=CC=C1C1=COC2=CC(O)=CC(O)=C2C1=O TZBJGXHYKVUXJN-UHFFFAOYSA-N 0.000 description 3
- 229940045109 genistein Drugs 0.000 description 3
- ZCOLJUOHXJRHDI-CMWLGVBASA-N genistein 7-O-beta-D-glucoside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=C2C(=O)C(C=3C=CC(O)=CC=3)=COC2=C1 ZCOLJUOHXJRHDI-CMWLGVBASA-N 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000000691 measurement method Methods 0.000 description 3
- 235000013336 milk Nutrition 0.000 description 3
- 239000008267 milk Substances 0.000 description 3
- 210000004080 milk Anatomy 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 238000012856 packing Methods 0.000 description 3
- 238000012827 research and development Methods 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- LDVVTQMJQSCDMK-UHFFFAOYSA-N 1,3-dihydroxypropan-2-yl formate Chemical compound OCC(CO)OC=O LDVVTQMJQSCDMK-UHFFFAOYSA-N 0.000 description 2
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 2
- VOXZDWNPVJITMN-ZBRFXRBCSA-N 17β-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-ZBRFXRBCSA-N 0.000 description 2
- 241000209495 Acorus Species 0.000 description 2
- 241001092083 Amelanchier Species 0.000 description 2
- 235000003840 Amygdalus nana Nutrition 0.000 description 2
- 241000125205 Anethum Species 0.000 description 2
- 235000003826 Artemisia Nutrition 0.000 description 2
- 241000723343 Cichorium Species 0.000 description 2
- 244000075634 Cyperus rotundus Species 0.000 description 2
- 235000016854 Cyperus rotundus Nutrition 0.000 description 2
- 241000893536 Epimedium Species 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- 229920002907 Guar gum Polymers 0.000 description 2
- 108010002352 Interleukin-1 Proteins 0.000 description 2
- 241000546273 Lindera <angiosperm> Species 0.000 description 2
- 239000005089 Luciferase Substances 0.000 description 2
- 235000014435 Mentha Nutrition 0.000 description 2
- 241001072983 Mentha Species 0.000 description 2
- 241001529734 Ocimum Species 0.000 description 2
- 235000011205 Ocimum Nutrition 0.000 description 2
- 241000220299 Prunus Species 0.000 description 2
- 235000011432 Prunus Nutrition 0.000 description 2
- 241000208225 Rhus Species 0.000 description 2
- 241001529742 Rosmarinus Species 0.000 description 2
- 108010073771 Soybean Proteins Proteins 0.000 description 2
- 235000021307 Triticum Nutrition 0.000 description 2
- 244000098338 Triticum aestivum Species 0.000 description 2
- 235000009392 Vitis Nutrition 0.000 description 2
- 241000219095 Vitis Species 0.000 description 2
- 240000008042 Zea mays Species 0.000 description 2
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 2
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 235000009052 artemisia Nutrition 0.000 description 2
- 238000003556 assay Methods 0.000 description 2
- 235000013361 beverage Nutrition 0.000 description 2
- 210000000481 breast Anatomy 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 235000005822 corn Nutrition 0.000 description 2
- 230000002354 daily effect Effects 0.000 description 2
- 235000005911 diet Nutrition 0.000 description 2
- 235000013325 dietary fiber Nutrition 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000000975 dye Substances 0.000 description 2
- 235000018905 epimedium Nutrition 0.000 description 2
- 229960005309 estradiol Drugs 0.000 description 2
- 229930182833 estradiol Natural products 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- 125000005456 glyceride group Chemical group 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 239000003102 growth factor Substances 0.000 description 2
- 235000010417 guar gum Nutrition 0.000 description 2
- 239000000665 guar gum Substances 0.000 description 2
- 229960002154 guar gum Drugs 0.000 description 2
- 239000000787 lecithin Substances 0.000 description 2
- 235000010445 lecithin Nutrition 0.000 description 2
- 229940067606 lecithin Drugs 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 230000001582 osteoblastic effect Effects 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 239000003075 phytoestrogen Substances 0.000 description 2
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 2
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 235000014774 prunus Nutrition 0.000 description 2
- 235000019710 soybean protein Nutrition 0.000 description 2
- 235000021147 sweet food Nutrition 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- GZCWLCBFPRFLKL-UHFFFAOYSA-N 1-prop-2-ynoxypropan-2-ol Chemical compound CC(O)COCC#C GZCWLCBFPRFLKL-UHFFFAOYSA-N 0.000 description 1
- 102000013563 Acid Phosphatase Human genes 0.000 description 1
- 108010051457 Acid Phosphatase Proteins 0.000 description 1
- 102000007469 Actins Human genes 0.000 description 1
- 108010085238 Actins Proteins 0.000 description 1
- 235000001674 Agaricus brunnescens Nutrition 0.000 description 1
- OGSPWJRAVKPPFI-UHFFFAOYSA-N Alendronic Acid Chemical compound NCCCC(O)(P(O)(O)=O)P(O)(O)=O OGSPWJRAVKPPFI-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 235000019737 Animal fat Nutrition 0.000 description 1
- 208000036487 Arthropathies Diseases 0.000 description 1
- 235000007319 Avena orientalis Nutrition 0.000 description 1
- 235000007558 Avena sp Nutrition 0.000 description 1
- 229940122361 Bisphosphonate Drugs 0.000 description 1
- 229940078581 Bone resorption inhibitor Drugs 0.000 description 1
- 102000055006 Calcitonin Human genes 0.000 description 1
- 108060001064 Calcitonin Proteins 0.000 description 1
- WLYGSPLCNKYESI-RSUQVHIMSA-N Carthamin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1[C@@]1(O)C(O)=C(C(=O)\C=C\C=2C=CC(O)=CC=2)C(=O)C(\C=C\2C([C@](O)([C@H]3[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O3)O)C(O)=C(C(=O)\C=C\C=3C=CC(O)=CC=3)C/2=O)=O)=C1O WLYGSPLCNKYESI-RSUQVHIMSA-N 0.000 description 1
- 241000208809 Carthamus Species 0.000 description 1
- 108010076119 Caseins Proteins 0.000 description 1
- 102000004171 Cathepsin K Human genes 0.000 description 1
- 108090000625 Cathepsin K Proteins 0.000 description 1
- 240000000560 Citrus x paradisi Species 0.000 description 1
- 241000212948 Cnidium Species 0.000 description 1
- 235000014375 Curcuma Nutrition 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- -1 Daidezin Chemical compound 0.000 description 1
- 101000874334 Dalbergia nigrescens Isoflavonoid 7-O-beta-apiosyl-glucoside beta-glycosidase Proteins 0.000 description 1
- 108010082495 Dietary Plant Proteins Proteins 0.000 description 1
- 241000158776 Dodonaea Species 0.000 description 1
- 206010013786 Dry skin Diseases 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- 101000757733 Enterococcus faecalis (strain ATCC 700802 / V583) Autolysin Proteins 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 241000893030 Eriogonum Species 0.000 description 1
- 108700039887 Essential Genes Proteins 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 108010068370 Glutens Proteins 0.000 description 1
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 description 1
- 208000012659 Joint disease Diseases 0.000 description 1
- 102000004407 Lactalbumin Human genes 0.000 description 1
- 108090000942 Lactalbumin Proteins 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- UMVSOHBRAQTGQI-UPZYVNNASA-N Lactucopicrin Natural products O=C(O[C@@H]1[C@H]2C(=C)C(=O)O[C@@H]2[C@@H]2C(CO)=CC(=O)C2=C(C)C1)Cc1ccc(O)cc1 UMVSOHBRAQTGQI-UPZYVNNASA-N 0.000 description 1
- 239000005913 Maltodextrin Substances 0.000 description 1
- 229920002774 Maltodextrin Polymers 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 101000757734 Mycolicibacterium phlei 38 kDa autolysin Proteins 0.000 description 1
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 235000008098 Oxalis acetosella Nutrition 0.000 description 1
- 240000007930 Oxalis acetosella Species 0.000 description 1
- 235000019482 Palm oil Nutrition 0.000 description 1
- 235000010240 Paullinia pinnata Nutrition 0.000 description 1
- 108010084695 Pea Proteins Proteins 0.000 description 1
- 102000015731 Peptide Hormones Human genes 0.000 description 1
- 108010038988 Peptide Hormones Proteins 0.000 description 1
- 208000006735 Periostitis Diseases 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 244000046052 Phaseolus vulgaris Species 0.000 description 1
- 108091000080 Phosphotransferase Proteins 0.000 description 1
- 235000010582 Pisum sativum Nutrition 0.000 description 1
- 241001092090 Pittosporum Species 0.000 description 1
- 206010036590 Premature baby Diseases 0.000 description 1
- 235000018992 Prunus glandulosa Nutrition 0.000 description 1
- 240000001619 Prunus glandulosa Species 0.000 description 1
- 235000013999 Prunus japonica Nutrition 0.000 description 1
- 102000014128 RANK Ligand Human genes 0.000 description 1
- 108010025832 RANK Ligand Proteins 0.000 description 1
- IIDJRNMFWXDHID-UHFFFAOYSA-N Risedronic acid Chemical compound OP(=O)(O)C(P(O)(O)=O)(O)CC1=CC=CN=C1 IIDJRNMFWXDHID-UHFFFAOYSA-N 0.000 description 1
- 235000019485 Safflower oil Nutrition 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 235000019486 Sunflower oil Nutrition 0.000 description 1
- 241000245665 Taraxacum Species 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 1
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 1
- 108060008683 Tumor Necrosis Factor Receptor Proteins 0.000 description 1
- 208000035896 Twin-reversed arterial perfusion sequence Diseases 0.000 description 1
- 239000002250 absorbent Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000001785 acacia senegal l. willd gum Substances 0.000 description 1
- 235000020167 acidified milk Nutrition 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 238000000246 agarose gel electrophoresis Methods 0.000 description 1
- 230000004520 agglutination Effects 0.000 description 1
- 229940062527 alendronate Drugs 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 235000021120 animal protein Nutrition 0.000 description 1
- 238000000137 annealing Methods 0.000 description 1
- 244000030166 artemisia Species 0.000 description 1
- 235000021336 beef liver Nutrition 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 150000004663 bisphosphonates Chemical class 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 210000002449 bone cell Anatomy 0.000 description 1
- 230000010072 bone remodeling Effects 0.000 description 1
- 235000021152 breakfast Nutrition 0.000 description 1
- BBBFJLBPOGFECG-VJVYQDLKSA-N calcitonin Chemical compound N([C@H](C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N1[C@@H](CCC1)C(N)=O)C(C)C)C(=O)[C@@H]1CSSC[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1 BBBFJLBPOGFECG-VJVYQDLKSA-N 0.000 description 1
- 229960004015 calcitonin Drugs 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 235000013351 cheese Nutrition 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000002670 chondrogenetic effect Effects 0.000 description 1
- 230000004087 circulation Effects 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 235000013409 condiments Nutrition 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 229960000913 crospovidone Drugs 0.000 description 1
- VFLDPWHFBUODDF-FCXRPNKRSA-N curcumin Chemical compound C1=C(O)C(OC)=CC(\C=C\C(=O)CC(=O)\C=C\C=2C=C(OC)C(O)=CC=2)=C1 VFLDPWHFBUODDF-FCXRPNKRSA-N 0.000 description 1
- 235000013365 dairy product Nutrition 0.000 description 1
- 238000005238 degreasing Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- ORYOIBJWFDNIPD-UHFFFAOYSA-N diacetyl 2,3-dihydroxybutanedioate Chemical compound CC(=O)OC(=O)C(O)C(O)C(=O)OC(C)=O ORYOIBJWFDNIPD-UHFFFAOYSA-N 0.000 description 1
- 230000037213 diet Effects 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000004043 dyeing Methods 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 238000004945 emulsification Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000002357 endometrial effect Effects 0.000 description 1
- 230000007071 enzymatic hydrolysis Effects 0.000 description 1
- 238000006047 enzymatic hydrolysis reaction Methods 0.000 description 1
- 230000009483 enzymatic pathway Effects 0.000 description 1
- 230000001076 estrogenic effect Effects 0.000 description 1
- ZMMJGEGLRURXTF-UHFFFAOYSA-N ethidium bromide Chemical compound [Br-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CC)=C1C1=CC=CC=C1 ZMMJGEGLRURXTF-UHFFFAOYSA-N 0.000 description 1
- 229960005542 ethidium bromide Drugs 0.000 description 1
- 238000003810 ethyl acetate extraction Methods 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 235000012041 food component Nutrition 0.000 description 1
- 239000005428 food component Substances 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000021312 gluten Nutrition 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 229910001867 inorganic solvent Inorganic materials 0.000 description 1
- 239000003049 inorganic solvent Substances 0.000 description 1
- 230000002608 insulinlike Effects 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- GOMNOOKGLZYEJT-UHFFFAOYSA-N isoflavone Chemical compound C=1OC2=CC=CC=C2C(=O)C=1C1=CC=CC=C1 GOMNOOKGLZYEJT-UHFFFAOYSA-N 0.000 description 1
- CJWQYWQDLBZGPD-UHFFFAOYSA-N isoflavone Natural products C1=C(OC)C(OC)=CC(OC)=C1C1=COC2=C(C=CC(C)(C)O3)C3=C(OC)C=C2C1=O CJWQYWQDLBZGPD-UHFFFAOYSA-N 0.000 description 1
- 235000008696 isoflavones Nutrition 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- QCDLLIUTDGNCPO-UHFFFAOYSA-N lactupicrin Natural products C12OC(=O)C(=C)C2C(O)CC(C)=C(C(C=2)=O)C1C=2COC(=O)CC1=CC=C(O)C=C1 QCDLLIUTDGNCPO-UHFFFAOYSA-N 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 150000004668 long chain fatty acids Chemical class 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 238000003670 luciferase enzyme activity assay Methods 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 229940035034 maltodextrin Drugs 0.000 description 1
- 210000001161 mammalian embryo Anatomy 0.000 description 1
- 210000005075 mammary gland Anatomy 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 239000001771 mentha piperita Substances 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- 201000011682 nervous system cancer Diseases 0.000 description 1
- 235000021590 normal diet Nutrition 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 210000000963 osteoblast Anatomy 0.000 description 1
- 210000004409 osteocyte Anatomy 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 239000002540 palm oil Substances 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 230000000849 parathyroid Effects 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 235000019702 pea protein Nutrition 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 239000000813 peptide hormone Substances 0.000 description 1
- 210000003460 periosteum Anatomy 0.000 description 1
- 239000008177 pharmaceutical agent Substances 0.000 description 1
- 102000020233 phosphotransferase Human genes 0.000 description 1
- 150000008442 polyphenolic compounds Chemical class 0.000 description 1
- 235000013824 polyphenols Nutrition 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 235000011962 puddings Nutrition 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000008521 reorganization Effects 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 229940089617 risedronate Drugs 0.000 description 1
- 235000005713 safflower oil Nutrition 0.000 description 1
- 239000003813 safflower oil Substances 0.000 description 1
- 239000000333 selective estrogen receptor modulator Substances 0.000 description 1
- 210000000582 semen Anatomy 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 235000014347 soups Nutrition 0.000 description 1
- 235000021262 sour milk Nutrition 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 235000013599 spices Nutrition 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 238000004659 sterilization and disinfection Methods 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 239000002600 sunflower oil Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 238000007910 systemic administration Methods 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 102000003298 tumor necrosis factor receptor Human genes 0.000 description 1
- 239000005418 vegetable material Substances 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/73—Rosaceae (Rose family), e.g. strawberry, chokeberry, blackberry, pear or firethorn
- A61K36/736—Prunus, e.g. plum, cherry, peach, apricot or almond
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/105—Plant extracts, their artificial duplicates or their derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/23—Apiaceae or Umbelliferae (Carrot family), e.g. dill, chervil, coriander or cumin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
- A61K36/282—Artemisia, e.g. wormwood or sagebrush
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
- A61K36/286—Carthamus (distaff thistle)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/28—Asteraceae or Compositae (Aster or Sunflower family), e.g. chamomile, feverfew, yarrow or echinacea
- A61K36/288—Taraxacum (dandelion)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/48—Fabaceae or Leguminosae (Pea or Legume family); Caesalpiniaceae; Mimosaceae; Papilionaceae
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/53—Lamiaceae or Labiatae (Mint family), e.g. thyme, rosemary or lavender
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/53—Lamiaceae or Labiatae (Mint family), e.g. thyme, rosemary or lavender
- A61K36/534—Mentha (mint)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/54—Lauraceae (Laurel family), e.g. cinnamon or sassafras
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/73—Rosaceae (Rose family), e.g. strawberry, chokeberry, blackberry, pear or firethorn
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/87—Vitaceae or Ampelidaceae (Vine or Grape family), e.g. wine grapes, muscadine or peppervine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/88—Liliopsida (monocotyledons)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/88—Liliopsida (monocotyledons)
- A61K36/882—Acoraceae (Calamus family), e.g. sweetflag or Acorus calamus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/88—Liliopsida (monocotyledons)
- A61K36/89—Cyperaceae (Sedge family)
- A61K36/8905—Cyperus (flatsedge)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/12—Drugs for disorders of the metabolism for electrolyte homeostasis
- A61P3/14—Drugs for disorders of the metabolism for electrolyte homeostasis for calcium homeostasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Abstract
本发明涉及用于维持骨健康或预防、减轻和/或治疗骨病的组合物。本发明也涉及组合物在制造营养物、补充物、治疗剂或药物的用途;并涉及促进骨生长或维持骨健康的方法,所述方法包括施用有效量的该组合物。
Description
本发明涉及用于维持骨健康或预防、减轻和/或治疗骨病的组合物。本发明也涉及该组合物在制造营养物、补充物或药物中的用途;并涉及促进骨生长或维持骨健康的方法,所述方法包括施用有效量的组合物。
发明背景
健康的骨需要包括骨形成和吸收间平衡的有效骨再造。大多数骨病归咎于骨吸收增加,致使主要治疗目的是抑制骨吸收的增加,因此迄今为止研发的大多数药剂是抗吸收的。例如,雌激素抑制可促进破骨细胞产生和分化的细胞因子的产生。正在研发对骨健康有益同时降低对乳腺或子宫内膜组织产生激素副作用危险的SERMs(选择性雌激素反应调节剂)。认为它们通过与雌激素类似的机理在骨中起作用。二膦酸盐化合物(如阿伦膦酸盐、利塞膦酸盐等)集中于骨,是迄今为止最有效的骨吸收抑制剂。它们抑制关键的酶途径,该途径是破骨细胞活性和破骨细胞存活需要的。降钙素是通过抑制破骨细胞活性而抑制骨吸收的多肽激素。新的靶包括阻断TNF受体/配体家族成员及它们的信号途径,特别是阻断RANK/RANKL,抑制骨特异的金属蛋白酶如组织蛋白酶K或抑制特异的激酶。
刺激骨形成的治疗剂的研发落后于骨吸收治疗剂的研发。已知一些化学品或药剂促进人的骨生长。例如,WO 9619246描述了通过间歇施用甲状旁腺素、PTH-相关蛋白或激动剂至少一个月促进患者骨生长的方法。在WO 9619501中,来源于胰的因子抑制骨吸收并刺激骨细胞增殖和增加骨形成。
最近的主要突破是证实骨形态发生蛋白2(BMP-2)在刺激骨形成中作为关键因素的作用及他汀类药物(通过抑制胆固醇合成降低胆固醇的有效药)改善骨形成的作用,改善骨形成部分是通过诱导BMP-2而介导。已显示重组BMP-2的递送诱导骨或软骨形成。在US 6150328中,描述了诱导骨和软骨形成的方法,所述方法包括施用通过培养用编码BMP的DNA转化了的细胞产生的经纯化骨形态发生蛋白。并且,WO 9711095涉及骨形态发生蛋白组合物用于治疗神经肿瘤及骨生长和创伤愈合的用途。除了BMPs外,生长因子如胰岛素样GF(IGF-1)、转化GF-β(TGF-β)、成纤维细胞GFs(FGFs)在骨折愈合和骨缺损中的局部治疗法正在研究中。然而由于小肠的代谢及也由于对其它组织可能有作用,全身施用是有问题的。正提议基因治疗作为一种选择。该备选方法是通过饮食或药物调节剂调节成骨细胞的基因或蛋白质表达而靶向对成骨细胞产生的调节。
虽然证明了这些化学品和药物化合物用于骨病的治疗,但通过提供安全、有效的营养方式来促进骨生长并预防、减轻或治疗哺乳动物的骨/关节病仍然是令人感兴趣的。
发明概述
因此,第一方面,本发明提供了用于预防、减轻和/或治疗骨病或维持哺乳动物骨健康的组合物,所述组合物包含经筛选的诱导骨形态发生蛋白表达的至少一种植物或植物提取物的有效量作为活性成分。
值得注意,现在发现一些植物或植物提取物通过调节骨或软骨组织中内源性生长因子具有促进骨生长的能力。它们具有诱导体内及骨局部环境中骨形态发生蛋白表达的能力,这种能力对骨形成和修复、骨健康的维持或预防、减轻和/或治疗骨病有积极效果。
根据本发明的组合物可用于人或宠物的营养物、补充物、治疗剂或药物的生产。
向个体施用本发明的食物组合物导致在骨折愈合期间改善骨再生。本组合物进一步有助于抑制骨吸收。它有助于在生长期间增加骨形成及骨矿物质密度,并优化骨量峰值。并且,本食物组合物有助于降低骨丢失,特别是哺乳动物中与年龄相关的骨丢失。因此它有助于维持与年龄有关的骨量并降低骨质疏松症的危险。
此外,它有助于构建哺乳动物的软骨,预防宠物或人的骨关节炎,从而使个体的活动或移动性更好。
另一方面,本发明涉及根据其刺激骨形态发生蛋白和/或抑制骨吸收的能力而筛选的植物或植物提取物的用途,用于制备维持哺乳动物骨健康并预防、减轻和/或治疗骨病的组合物。
另外,本发明提供了预防、减轻和/或治疗骨病或维持骨健康的方法,其包括施用有效量的上述组合物。
本发明进一步提供了在生长期间增加骨形成、骨矿物质密度并优化骨量峰值、治疗或预防骨质疏松症、在骨折愈合期间刺激骨再生的方法,其包括施用有效量的上述组合物。
本发明进一步涉及治疗、减轻和/或预防宠物和人骨关节炎的方法,其包括个体食用上述组合物的步骤。
进一步提供了对人或宠物降低骨丢失,特别是与年龄相关的骨丢失的方法,其包括个体食用上述组合物的步骤。
发明详述
关于本发明的第一个目的,根据本发明的植物或植物提取物包含通过诱导骨形态发生蛋白表达而具有合成代谢潜力并可进一步为抗吸收剂的植物化学品。
在一个优选的实施方案中,植物或植物提取物来自植物源的任何部分,如叶、块茎、果实、种子、根、颖果、胚或细胞培养物。植物或植物提取物取决于植物源,可为叶、根和/或果实的干的、冷冻干燥提取物形式,或为新鲜植物,或为通过本领域公知的无机溶剂或有机溶剂提取方法获得的浓缩级分。
选择具有抑制骨吸收和/或诱导骨形成能力的植物或植物提取物,特别是它可选自山胡椒属(Lindera)、蒿属(Artemisia)、菖蒲属(Acorus)、红蓝花属(Carthamus)、葛缕子属(Carum)、蛇床子(Cnidium)、唐棣属(Amelanchier)、姜黄属(Curcuma)、蒲公英属(Taraxacum)、莎草属(Cyperus)、刺柏属(Juniperus)、李属(Prunus)、鸢尾属(Iris)、菊苣属(Cichorium)、车桑子属(Dodonaea)、淫羊藿属(Epimedium)、Eriogonum、大豆(Soya)、薄荷属(Mentha)、罗勒属(Ocimum)、百里香属(thymus)、艾菊属(Tanacetum)、车前草属(Plantago)、绿薄荷(Spearmint)、红木属(Bixa)、葡萄属(Vitis)、迷迭香属(Rosemarinus)、艾菊属(Tanacetum)、盐肤木属(Rhus)和莳萝属(Anethum)。它也可为蘑菇。
在一个最优选的实施方案中,它可例如为美国山胡椒(Linderabenzoin)的地上部分,艾蒿(Artemisia vulgaris)的地上部分,菖蒲(Acoruscalamus)的根状茎,红花(Carthamus tinctorius)的种子或花,卵圆形唐棣(Amelanchier ovalis)的果实,桤叶唐棣(Amelanchier alnifolia)的果实,菊苣(Cichorium intybus)的根,姜黄(Curcuma longa)的根状茎,短角淫羊藿(Epimedium brevicornum)的地上部分,Eriogonum giganteum的地上部分,药蒲公英(Taraxacum officinalis)的叶或根,香附子(Cyperusrotondus)的根状茎,车桑子(Dodoneae viscosa)的叶,白鸢尾(Iris pallida)的细胞培养物,德国鸢尾(Iris germanica)、白鸢尾(Iris pallida)或黄鸢尾(Iris pseudacorus)的根状茎,欧洲刺柏(Juniperus communis)的果实,桃(Prunus persica)的种子,大豆细胞培养物,留兰香(Mentha spicata)的地上部分,丁香萝勒(Ocimum gratissimum)的地上部分,百里香种(Thymus sp.)的地上部分,光叶漆(Rhus glabra)的地上部分,茴香(aneth),红木属,葡萄(Vitis vinifera)的果实,迷迭香(Rosmarinusofficinalis)的地上部分,普通菊蒿(Tanacetum vulgare)的地上部分,葛缕子(Carum carvi),车前草(Plantago maior),Oxydendron arboreum的地上部分。
植物化学品可为染料木黄酮、大豆黄素、莴苣苦素、山莴苣苦素(lactucopicrin)、3-脱氧-莴苣苦素、香叶醇或香芹酮。
本发明的植物或植物提取物可用于制备食物组合物。所述组合物可为营养均衡的食物或宠物食物、膳食补充物、治疗剂或药物组合物的形式。
植物或植物提取物可单独应用或与其它植物如菊苣、茶树、可可树联合应用,或与其它生物活性分子如抗氧化剂、脂肪酸、益生素纤维、葡糖胺、硫酸软骨素联合应用。
在一个实施方案中,制备了用于人消费的食物组合物。该组合物可为营养完全配方、乳制品、冷的或常温稳定的饮料、汤、膳食补充物、膳食替代品和营养棒或甜食。
除了根据本发明的植物提取物外,营养配方可包含蛋白质源。优选应用膳食蛋白质作为蛋白质源。膳食蛋白质可为任何合适的膳食蛋白质;例如动物蛋白(如乳蛋白、肉蛋白和卵蛋白);植物蛋白(如大豆蛋白、小麦蛋白、大米蛋白和豌豆蛋白);游离氨基酸的混合物;或为它们的组合物。特别优选乳蛋白如酪蛋白、乳清蛋白和大豆蛋白。组合物也可包含碳水化合物源和脂肪源。
如果营养配方包括脂肪源,该脂肪源优选地提供营养配方中的约5%至约55%的能量;如提供约20%至50%的能量。组成脂肪源的脂类可为任何合适的脂肪或脂肪混合物。植物脂肪特别适用;如大豆油、棕榈油、椰子油、红花油、葵花子油、玉米油、芸苔油、卵磷脂等。如果需要,也可加入动物性脂肪如乳脂。
营养配方中可加入碳水化合物源。所述碳水化合物源优选地提供占该营养组合物约40%至约80%的能量。可应用任何合适的碳水化合物,如蔗糖、乳糖、葡萄糖、果糖、玉米糖浆固体和麦芽糖糊精及它们的混合物。如果需要也可加入膳食纤维。如果应用碳水化合物,则其优选地包含占营养配方约5%的能量。膳食纤维可来自任何适当来源,包括如大豆、豌豆、燕麦、果胶、瓜尔胶、阿拉伯胶和果糖寡糖。营养配方中也可包括合适的符合适当指标的维生素和矿物质。
如果需要,可在营养配方中掺入一种或多种食物级乳化剂;如单甘油酯和双甘油酯的二乙酰酒石酸酯、卵磷脂及单甘油酯和双甘油酯。同样地可包括合适的盐和稳定剂。植物提取物也可与维生素和矿物质组合。
优选可经肠施用的营养组合物;如以粉剂、片剂、胶囊剂、液态浓缩剂、固体产品或及喝饮料的形式。如果想生产粉剂营养配方,可将经过匀化加工处理的混合物转移到合适的干燥器如喷雾干燥器或冷冻干燥器并转化为粉剂。
在另一实施方案中,营养组合物包含基于乳的谷类并包含益生素的配方。优选地,基于乳的谷类是作为益生素配方的载体的未成熟谷类。
在另一实施方案中,可用至少一种本发明的植物或植物提取物强化普通食品。如发酵乳、酸奶、新鲜乳酪、凝乳、甜食如糖果或甜饮料、甜条、早餐谷片或条、饮料、奶粉、豆制品、非乳发酵品或用于临床营养的营养补充物。
组合物中植物或植物提取物的量可根据植物源和它的用途而改变。在一个优选的实施方案中,其有效日剂量至少为约1mg活性分子/天,更优选地为从1毫克至200毫克活性分子/天。
在一个实施方案中,可制备包含至少一种上述提取物或植物化学品的药物组合物,该药物组合物的量足可使个体达到预期效应。该组合物可为片剂、液体、胶囊剂、软胶囊剂、糊剂、锭剂、胶剂或可饮用溶液或乳剂、干的经口补充物、湿的经口补充物。药物组合物进一步包含适于向靶组织递送不同性质的各活性分子的载体和赋形剂。载体/赋形剂的种类和其量取决于物质的性质和药物递送和/或施用方式。可以理解熟练人员基于他自己的知识可选择适当的组分和植物剂型。
本发明的植物或植物提取物可用于制备宠物食物组合物。所述组合物可作为宠物正常饮食的补充物或作为营养完全的宠物食物组分施用,并且更优选地在低热量宠物食物中。它也可为药物组合物。
植物或植物提取物可单独应用或与其它植物如菊苣、茶树、可可树联合应用,或与其它生物活性分子如抗氧化剂、脂肪酸、益生素纤维、葡糖胺、硫酸软骨素联合应用。
优选地,宠物食物组合物对于15千克的犬,每克干宠物食物包含约0.01至0.5克干植物;对于15千克的犬,每克湿宠物食物包含约0.001至0.1克干植物。
根据本发明的营养完全的宠物食物可为粉状、干燥形式、治疗剂或为湿的、冷的或常温稳定的宠物食物产品。它可冷冻或作为常温稳定产品提供。这些宠物食物可以本领域公知的方式生产。
宠物食物也可任选包含益生素、益生微生物或其它活性剂,如长链脂肪酸。宠物食物中益生素的量优选占低于10%的重量。例如,所包含的益生素可占宠物食物重量的约0.1%至约5%。对于用菊苣作为益生素来源的宠物食物,所包括的菊苣可占食物混合物重量的约0.5%至10%;更优选地占重量的约1%至约5%。
如果应用益生微生物,宠物食物优选每克宠物食物包含约104至约1010个细胞的益生微生物;更优选每克约106至约108个细胞的益生微生物。宠物食物可包含占约0.5%至约20%重量的益生微生物混合物;优选地占重量的约1%至约6%;例如占重量的约3%至约6%。
如果需要,可向宠物食物添加矿物质和维生素以使它们营养完全。此外,根据需要,也可将多种其它成分如糖、盐、香料、调料、调味剂等掺入到宠物食物中。
在另一实施方案中,可制备膳食添加剂以改善宠物食物的质量。作为膳食添加剂,它们无论是湿的还是干的均可被封入胶囊内或可以粉剂形式提供并与正餐一起包装或单独包装。通过举例的方式,含有本发明提取物的粉末可以粉末形式包装入小袋中或包装到凝胶或脂或其它合适的载体中。根据使用者的要求,这些单独的包装单位可与正餐一起提供或为与正餐或治疗剂一同应用的多单位包装。
为达到有益效果,宠物消费的宠物食物的量取决于宠物的大小、宠物的类型和宠物的年龄。但是,每天提供每千克体重约0.5至5克干植物的宠物食物的量对犬和猫一般是足够的。
对人或动物施用上述食物或宠物食物组合物,导致在骨折愈合期间可改善骨再生。它有助于在生长期间刺激骨形成和骨矿物质密度,并优化骨量峰值。特别是它可在儿童期提供最佳骨生长。该食物组合物有助于预防骨丢失,特别是哺乳动物中与年龄相关的骨丢失或长期住院相关的骨丢失。它降低了骨质疏松症的危险并在骨折后改善恢复。此外,它有助于构建哺乳动物软骨,预防宠物和人的骨关节炎,其导致个体活动或移动性更好。
下列实施例仅以阐述的形式给出,不用于限制本发明的主题。除非另有说明,所有百分数均为重量百分数。实施例之前是对附图的简述。
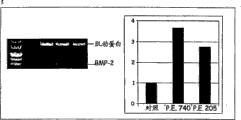
图1:来自美国山胡椒(P.E.740,50μg/ml))或香附子(P.E.205,10μg/ml)的提取物处理48小时后通过RT-PCR测定hPOB-tert细胞中内源性BMP-2mRNA的表达,并显示它们对BMP-2的刺激分别为对照的3.8倍和2.8倍。本测定法以洛伐他汀(0.5μg/ml)作为阳性对照,洛伐他汀显示了诱导BMP-2为对照的2.5倍。
图2:来自丁香罗勒(738)、桤叶唐棣(734)、大豆(Glycine max)(768)、香附子(205)、红花(746)提取物用颅盖测定法(A)和凹陷测定法(pit assay)(B)所测定的抑制值的比较。
实施例
实施例1:骨形成和骨吸收鉴定法
1.骨形成
对91种提取物进行了筛选,其中通过BMP-2(骨形态发生蛋白)基因报道测定法筛选用于骨形成的提取物,通过颅盖测定法筛选用于骨吸收的提取物。这91种提取物对应于30种不同的植物。
材料与方法
●用于筛选测定法的提取物的制备:
磨碎的植物材料用己烷脱脂,然后用醇和水混合物进行提取,不同的水百分比为从10至90%,优选地用50%的水。醇可用甲醇或乙醇,获得提取物1a。
对该第一提取物的残余部分,用α和β糖苷酶进行酶促水解作用。酶可用酸性条件替代。此操作可在温和条件下(室温)进行或用不同酸浓度通过回流进行。含水的水解相用不互溶的溶剂提取,优选用乙酸乙酯提取获得提取物2a。
提取物可干燥、冷冻干燥或以液体形式提供。
在一些情况下,多酚可通过聚乙烯聚吡咯烷酮(PVPP)处理后丢弃,避免筛选测定的假象。
制备提取物之后,每种提取物进行称重,用二甲基亚砜(DMSO)再溶解至终浓度20mg/ml并分装置于-20℃保存。这作为母液应用并随后用各测定法所使用的培养基进行稀释。测定法系统中对一系列的剂量进行检测。
●骨形成测定法
BMP-2萤光素酶测定法—用包含与萤光素酶基因可操作地连接的BMP-2启动子的2T3细胞测定提取物的活性。细胞裂解物中萤光素酶活性增加反映BMP-2启动子活性的增加。提取物最初稀释成100μg/ml,然后进行1/2稀释至0.2μg/ml,进行测定。BMP-2启动子活性通过测定细胞提取物中萤光素酶活性进行测定。
表1 13种植物在刺激BMP-2表达中得到明显的阳性结果
| 拉丁名 | 英文名 | 部分 | 浓度μg/ml | 活性提取物/n<sup>o</sup> |
| Acorus calamus | 菖蒲 | 根状茎 | 5 | MeOH/水/731 |
| Amelanchier ovalis | 卵圆形唐棣 | 果实 | 10 | MeOH/水/219 |
| Artemisia vulgaris | 艾蒿 | 地上部分 | 10 | 乙酸乙酯/225 |
| Cyperus rotundus | 香附子 | 根状茎 | 10 | 乙酸乙酯/205 |
| Taraxacum officinalis | 药蒲公英 | 叶 | 50 | 乙酸乙酯/750 |
| Lindera benzoin | 美国山胡椒 | 地上部分 | 50 | 乙酸乙酯/740 |
| Prunus persica | 桃 | 种子 | 25 | 乙酸乙酯/772 |
| Glycine max | 大豆 | 细胞培养物 | 50 | 乙酸乙酯/768 |
| Iris pallida | 白鸢尾 | 块茎 | 100 | MeOH/水/239 |
| Rosmarinus officianlis | 迷迭香 | 叶 | 50 | MeOH/水/2004乙酸乙酯/2005 |
| Carvi | 葛缕子 | 种子 | 25 | 乙酸乙酯/2074 |
| Thyme | 百里香 | 叶 | 25 | 乙酸乙酯/2067 |
| Mentha spicata | 薄荷 | 叶 | 100 | 乙酸乙酯/2072 |
| Vitis vinifera | 葡萄 | 果实 | 100 | 乙酸乙酯/2069 |
刺激BMP-2的来自同一植物的新制备物和其亚级分的例子是:美国山胡椒、活性提取物/nO乙酸乙酯/740/2059;活性亚级分/nO2060药蒲公英,活性提取物/nO乙酸乙酯/750/2034;活性亚级分/nO2035香附子,活性提取物/nO乙酸乙酯/205/2011;活性亚级分/nO2012、2013白鸢尾,活性提取物/nOMeOH/水/239;活性亚级分/nO760/762/2021、2022
通过在反相硅胶柱上用极性不同的溶剂洗脱而分级分离制备亚级分。纯大豆异黄酮类中的染料木黄酮和大豆黄素(10-6M)刺激BMP-2而雌二醇不刺激BMP-2。
对BMP-2的诱导作用似乎不限于雌激素样活性,因为它是通过植物雌激素进行刺激而不是通过雌激素本身刺激。这表明植物雌激素(如染料木黄酮和大豆黄素)的活性可通过非雌激素机理介导。BMP-2启动子活性不是通过雌二醇进行刺激的,因此植物化合物的动情作用不是本试验需要的活性。
| 植物 | 活性提取物编号 | 浓度(μg/ml) |
| 大豆 | 乙酸乙酯/2001 | 10、50 |
| 迷迭香 | MeOH/水/2004 | 10、50 |
| 迷迭香 | 乙酸乙酯/2005 | 10 |
| 香附子 | 亚级分/2012 | 10、50 |
| 白鸢尾 | 亚级分/2022 | 10 |
| 百里香 | 乙酸乙酯/2067 | 10 |
| 葛缕子 | 乙酸乙酯/2074 | 10 |
在颅盖器官培养物中骨形成的例子
在科学(Science)286:1946-1949(1999)描述了应用的方法。用体外新生鼠颅盖测定法进行4天试验评估了提取物。骨与提取物孵育了整4天。骨形成通过组织学进行评估。
2.骨吸收,颅盖测定法
在新生骨吸收测定法中评估了提取物抑制IL-1(10-10M)刺激的骨吸收的能力。评估了每种提取物在10μg/ml时抑制骨吸收的能力。在新生骨吸收测定法中评估了实施例1制备的提取物抑制IL-1(10-10M)刺激的骨吸收的能力。评估了每种提取物在10μg/ml时抑制骨吸收的能力。
表2 列出了阳性的植物提取物:
| 拉丁名 | 英文名 | 部分 | 活性提取物/n<sup>o</sup> |
| Amelanchier alnifolia | 唐棣 | 果实 | 乙酸乙酯/734 |
| Ocimum gratissimum | 罗勒种 | 叶 | MeOH/H<sub>2</sub>O/737 |
| Ocimum gratissimum | 罗勒种 | 叶 | 乙酸乙酯/738 |
| Carthamus tinctorius | 红花 | 种子 | 乙酸乙酯/746 |
| Cyperus rotundus | 香附子 | 根状茎 | 乙酸乙酯/205 |
| Glycine max | 大豆 | 细胞培养物 | 768 |
下列植物在骨吸收测定法中有活性:桤叶唐棣、丁香罗勒、红花及大豆。香附子抑制骨吸收并诱导BMP-2。
实施例2:植物提取物对人成骨细胞中内源性BMP-2表达的作用
进一步检测了实施例1中BMP-2诱导阳性的植物在人骨膜/前成骨细胞细胞系hPOB-tert中诱导内源性BMP-2表达的能力。这个在成骨细胞的检测证实了实施例1中显示的结果。
例如,用美国山胡椒(提取物740,50μg/ml)和香附子(提取物205,10μg/ml)的提取物处理hPOB-tert细胞48小时,刺激BMP-2表达为对照的3.8和2.8倍(见图1)。用洛伐他汀(0.5μg/ml)作为阳性对照显示其诱导BMP-2为对照的2.5倍,从而验证了本测定法。
细胞长成单层后,细胞与0.05μg/ml的洛伐他汀孵育或与植物提取物孵育。用TRIzol试剂(Gibco)提取总RNA。用第一链cDNA合成试剂盒(Boehringer)对10μg RNA进行反转录。BMP-2cDNA序列用特异寡核苷酸引物(5′:TTGCGGCTGCTCAGCATGTT;3′:CATCTTGCATCTGTTCTCGGAA)在退火温度为55℃的条件下进行35个循环进行扩增。PCR产物通过琼脂糖凝胶电泳进行分离并用溴化乙锭染色进行检测。用NIH图象软件进行定量并用肌动蛋白作为管家基因对结果进行校正。
结果在图1显示。
骨吸收
在颅盖测定法中阳性的植物提取物在骨吸收的第二种测定法中再进行检测,第二种测定法即用在牛骨切片上培养的兔骨混合细胞的凹陷测定法(Tezuka K等人,1992,Biochem.Biophys.Res.Commun.186(2):911-7和Lorget F.等人,2000,Biochem.Biophys.Res.Commun.268(3):899-903)。吸收凹陷通过TRAP(酒石酸抗性的酸性磷酸酶)染色进行观察。观察阳性细胞并计数。
图2显示了两种测定系统对10μg/ml提取物测得的活性的比较
实施例3:干宠物食物
饲料混合物由占约58%重量的玉米、占约5.5%重量的玉米麸、占约22%重量的鸡食、2.5%干菊苣、约10%香附子块茎组成,盐、维生素和矿物质组成其余部分。
饲料混合物装入预调节器中并弄湿。然后将湿饲料装入挤压机-炊具中并弄成胶状。离开挤压机的胶状基质通过模具挤压并压出。压出物切成合适的饲喂犬的小块,110℃干燥约20分钟,并冷却形成小块。
该干燥物食对骨和软骨健康有积极效果并增加犬的活动性。
实施例4:有补充物的湿罐装宠物食物
由73%禽尸体、猪肺和牛肝(磨碎)、16%小麦面粉、2%染料、维生素和无机盐制备混合物。混合物在12℃乳化并压挤成布丁形式,然后在90℃烹熟。冷却至30℃并切成大块。45%的这些大块与55%调味料混合,所述调味料由98%水、1%染料和1%瓜尔胶制成。装入马口铁罐中并125℃消毒40分钟。
作为饲养前与该宠物食物混合的补充物,其所在的额外包装(小袋)中包含25克粉状香附子地上部分,用于添加至日常食物中。对于宠物的对应量为约25克/天,并且这可作为罐头的补充物(如在罐头上面)供应。
Claims (12)
1.用于人和宠物预防、减轻和/或治疗骨病和维持骨健康的食物组合物,所述组合物包含作为活性成分的有效量的至少一种植物或植物提取物,植物或植物提取物选自美国山胡椒(Lindera benzoin)的地上部分,艾蒿(Artemisia vulgaris)的地上部分,菖蒲(Acorus calamus)的根状茎,红花(Carthamus tinctorius)的种子或花,卵圆形唐棣(Amelanchier ovalis)的果实,桤叶唐棣(Amelanchier alnifolia)的果实,菊苣(Cichoriumintybus)的根,姜黄(Curcuma longa)的根状茎,短角淫羊藿(Epimediumbrevicornum)的地上部分,Eriogonum giganteum的地上部分,药蒲公英(Taraxacum officinalis)的叶或根,香附子(Cyperus rotondus)的根状茎,车桑子(Dodoneae viscosa)的叶,白鸢尾(Iris pallida)的细胞培养物,德国鸢尾(Iris germanica)、白鸢尾(Iris pallida)或黄鸢尾(Irispseudacorus)的根状茎,欧洲刺柏(Juniperus communis)的果实,桃(Prunus persica)的种子,留兰香(Mentha spicata)的地上部分,丁香萝勒(Ocimum gratissimum)的地上部分,百里香种(Thymus sp.)的地上部分,光叶漆(Rhus glabra)的地上部分,茴香(aneth),红木属,葡萄(Vitis vinifera)的果实,迷迭香(Rosmarinus officinalis)的地上部分,普通菊蒿(Tanacetum vulgare)的地上部分,葛缕子(Carum carvi),车前草(Plantago major),Oxydendron arboreum的地上部分,所述植物或植物提取物含有植物化学品莴苣苦素、山莴苣苦素、3-脱氧-莴苣苦素、香叶醇或香芹酮。
2.根据权利要求1的组合物,其中所述植物或植物提取物还抑制骨吸收。
3.根据权利要求1或2所述的组合物,所述组合物为营养均衡的食物或宠物食物、膳食补充物、治疗剂或药物组合物的形式。
4.根据权利要求1或2所述的组合物在制备食品中的用途,所述食品对人或宠物在骨折愈合期间有助于骨再生,在生长期间有助于增加骨形成及骨矿物质密度,并优化骨量峰值或减少骨丢失。
5.根据权利要求4中所述的用途,其中所述骨丢失是与年龄相关的骨丢失。
6.根据权利要求4或5中所述的用途,其中所制备的食品有助于构建人或宠物的软骨。
7.根据权利要求4或5中所述的用途,其中所制备的食品有助于预防人或宠物的骨关节炎。
8.包含具有刺激骨形态发生蛋白和/或抑制骨吸收能力的植物化学品的植物或植物提取物在制备对人或宠物预防、减轻和/或治疗骨病或维持骨健康的食物或宠物食物组合物中的用途,其中所述植物和植物提取物选自美国山胡椒(Lindera benzoin)的地上部分,艾蒿(Artemisia vulgaris)的地上部分,菖蒲(Acorus calamus)的根状茎,红花(Carthamus tinctorius)的种子或花,卵圆形唐棣(Amelanchier ovalis)的果实,桤叶唐棣(Amelanchier alnifolia)的果实,菊苣(Cichorium intybus)的根,姜黄(Curcuma longa)的根状茎,短角淫羊藿(Epimedium brevicornum)的地上部分,Eriogonum giganteum的地上部分,药蒲公英(Taraxacumofficinalis)的叶或根,香附子(Cyperus rotondus)的根状茎,车桑子(I)odoneae viscosa)的叶,白鸢尾(Iris pallida)的细胞培养物,德国鸢尾(Iris germanica)、白鸢尾(Iris pallida)或黄鸢尾(Iris pseudacorus)的根状茎,欧洲刺柏(Juniperus eommunis)的果实,桃(Prunus persica)的种子,留兰香(Mentha spicata)的地上部分,丁香萝勒(Ocimumgratissimum)的地上部分,百里香种(Thymus sp.)的地上部分,光叶漆(Rhus glabra)的地上部分,茴香(aneth),红木属,葡萄(Vitis vinifera)的果实,迷迭香(Rosmarinus officinalis)的地上部分,普通菊蒿(Tanacetumvulgare)的地上部分,葛缕子(Carum carvi),车前草(Plantago major),Oxydendron arboreum的地上部分,所述植物化学品选自莴苣苦素、山莴苣苦素、3-脱氧-莴苣苦素、香叶醇或香芹酮。
9.根据权利要求8的用途,其中组合物为如权利要求1至3中任一项所述。
10.根据权利要求8或9的用途,其中植物或植物提取物单独应用或与其它植物联合应用,或与其它生物活性分子联合应用。
11.根据权利要求10的用途,其中所述其它生物活性分子为抗氧化剂、脂肪酸、益生素纤维、葡糖胺、硫酸软骨素。
12.根据权利要求10的用途,其中所述其它植物为菊苣、茶树、可可树。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP01204838A EP1325681A1 (en) | 2001-12-11 | 2001-12-11 | Composition for promotion of bone growth and maintenance of bone health |
| EP01204838.5 | 2001-12-11 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1602159A CN1602159A (zh) | 2005-03-30 |
| CN100521972C true CN100521972C (zh) | 2009-08-05 |
Family
ID=8181405
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB028245172A Expired - Fee Related CN100521972C (zh) | 2001-12-11 | 2002-12-10 | 用于促进骨生长及维持骨健康的组合物 |
Country Status (23)
| Country | Link |
|---|---|
| US (2) | US20050079232A1 (zh) |
| EP (6) | EP1325681A1 (zh) |
| JP (3) | JP4653951B2 (zh) |
| KR (1) | KR20040083466A (zh) |
| CN (1) | CN100521972C (zh) |
| AT (1) | ATE472943T1 (zh) |
| AU (3) | AU2002366734B2 (zh) |
| BR (1) | BR0214838A (zh) |
| CA (1) | CA2469551C (zh) |
| CO (1) | CO5590866A2 (zh) |
| DE (1) | DE60236961D1 (zh) |
| DK (4) | DK1455601T4 (zh) |
| ES (4) | ES2387876T3 (zh) |
| HK (1) | HK1076009A1 (zh) |
| IL (2) | IL161659A0 (zh) |
| MX (1) | MXPA04005583A (zh) |
| NO (1) | NO327135B1 (zh) |
| PL (1) | PL369768A1 (zh) |
| PT (4) | PT2263480E (zh) |
| RU (1) | RU2314717C2 (zh) |
| UA (1) | UA84123C2 (zh) |
| WO (1) | WO2003053167A1 (zh) |
| ZA (1) | ZA200405454B (zh) |
Families Citing this family (61)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1325681A1 (en) * | 2001-12-11 | 2003-07-09 | Société des Produits Nestlé S.A. | Composition for promotion of bone growth and maintenance of bone health |
| AU2003272902A1 (en) * | 2002-10-01 | 2004-04-23 | Takara Bio Inc. | Remedies |
| US7601370B2 (en) * | 2004-07-28 | 2009-10-13 | Abbott Laboratories | Method for controlling body weight in estrogen-insufficient women |
| ES2314692T3 (es) * | 2004-07-28 | 2009-03-16 | Abbott Laboratories | Composiciones nutricionales y metodos para tratar o prevenir la osteoporosis. |
| US7435431B2 (en) * | 2004-07-28 | 2008-10-14 | Abbott Laboratories | Method for controlling body weight in estrogen-insufficient women |
| JP4738007B2 (ja) * | 2005-02-04 | 2011-08-03 | 久光製薬株式会社 | 骨粗鬆症治療用外用剤 |
| US8216801B2 (en) * | 2005-02-25 | 2012-07-10 | The State Of Israel, Ministry Of Agriculture & Rural Development, Agricultural Research Organization, (A.R.O.), The Volcani Center | Methods for treating inflammatory disorders |
| WO2008004118A2 (en) * | 2006-07-06 | 2008-01-10 | Avestha Gengraine Technologies Pvt. Ltd. | Carthamus tinctoris plant extracts for treating osteoporosis and the extraction process thereof |
| CN101646433B (zh) * | 2006-10-24 | 2011-11-16 | 戴维·W·克雷姆平 | 抗再吸收的和造骨的食物增补剂和使用方法 |
| US20080200425A1 (en) * | 2007-02-16 | 2008-08-21 | Kurtz Seymour J | Methods and compositions for regulating bone mineral density |
| KR100902947B1 (ko) * | 2007-03-07 | 2009-06-15 | 주식회사 피앤비 | 바실러스 서브틸리스 모리 배양물을 함유하는 성장촉진용조성물 |
| EP2214481B1 (en) | 2007-10-15 | 2019-05-01 | United Animal Health, Inc. | Method for increasing performance of offspring |
| WO2009106125A1 (en) * | 2008-02-25 | 2009-09-03 | Nestec S.A. | Polyphenols for the treatment of cartilage disorders |
| JP2009269834A (ja) * | 2008-05-01 | 2009-11-19 | Dhc Co | 骨吸収抑制剤並びに骨吸収抑制用飲食品及び医薬部外品 |
| US9771199B2 (en) * | 2008-07-07 | 2017-09-26 | Mars, Incorporated | Probiotic supplement, process for making, and packaging |
| US20100041768A1 (en) * | 2008-08-18 | 2010-02-18 | National Yang-Ming University | Composition and method of adipose cell differentiation inhibition |
| KR101096574B1 (ko) * | 2008-12-12 | 2011-12-20 | 에이치 엘 지노믹스(주) | 유럽종 포도의 씨 추출물을 함유하는 골다공증 예방 또는 치료용 약학 조성물 |
| KR101099021B1 (ko) * | 2009-04-24 | 2011-12-28 | 에이치 엘 지노믹스(주) | 유럽종 포도의 씨 추출물을 함유하는 골관절염 예방 또는 치료용 약학 조성물 |
| US7897184B1 (en) * | 2009-08-13 | 2011-03-01 | Access Business Group International Llc | Topical composition with skin lightening effect |
| RU2509569C2 (ru) * | 2011-07-05 | 2014-03-20 | Общество С Ограниченной Ответственностью "Парафарм" | Композиция для лечения и предупреждения остеоартрита и остеоартроза суставов |
| BR122020009091B1 (pt) | 2011-12-19 | 2021-08-17 | The Coca-Cola Company | Método para purificar reb x |
| KR101318160B1 (ko) * | 2011-12-20 | 2013-10-16 | 주식회사 진생사이언스 | 골형성을 촉진하는 효능을 가지는 dsx 추출물을 유효성분으로 하는 약학 조성물 |
| DE102011122466A1 (de) * | 2011-12-24 | 2013-06-27 | Westfalia-Automotive Gmbh | Hohlträger für ein Anhängesystem eines Kraftfahrzeugs |
| KR20140057709A (ko) * | 2012-10-31 | 2014-05-14 | (주)메디언스 | 키성장 및 골 강화용 조성물 |
| CN108484698A (zh) | 2012-12-19 | 2018-09-04 | 可口可乐公司 | 用于提高莱鲍迪苷x溶解度的组合物和方法 |
| US9820504B2 (en) | 2013-03-08 | 2017-11-21 | Axiom Foods, Inc. | Rice protein supplement and methods of use thereof |
| AU2014225786B2 (en) | 2013-03-08 | 2017-10-12 | Axiom Foods, Inc. | Rice protein supplements |
| US20140272068A1 (en) | 2013-03-14 | 2014-09-18 | Indra Prakash | Beverages containing rare sugars |
| WO2014146135A2 (en) | 2013-03-15 | 2014-09-18 | The Coca-Cola Company | Steviol glycosides, their compositions and their purification |
| US20160029677A1 (en) | 2013-03-15 | 2016-02-04 | The Coca-Cola Company | Novel glucosyl steviol glycosides, their compositions and their purification |
| US10905146B2 (en) | 2013-07-12 | 2021-02-02 | The Coca-Cola Company | Compositions for improving rebaudioside M solubility |
| KR101487761B1 (ko) * | 2013-11-05 | 2015-01-29 | 한국생명공학연구원 | 아르테미시닌을 유효성분으로 포함하는 골다공증 예방 및 치료용 약학적 조성물 |
| EP3683315A1 (en) | 2014-08-19 | 2020-07-22 | Purecircle SDN BHD | Method for preparing rebaudioside m |
| WO2017035527A1 (en) | 2015-08-27 | 2017-03-02 | Purecircle Sdn Bhd | Stevia extracts |
| US11202461B2 (en) | 2014-09-02 | 2021-12-21 | Purecircle Sdn Bhd | Stevia extracts |
| JP2017535289A (ja) * | 2014-11-06 | 2017-11-30 | エヌダブリューオー ステム キュア、エルエルシー | ラクトバチルス・ラムノサス(lactobacillus rhamnosus)を含む栄養補助食品 |
| CA2968973C (en) | 2014-11-29 | 2024-03-05 | The Coca-Cola Company | Novel diterpene glycosides, compositions and purification methods |
| KR101559937B1 (ko) * | 2015-02-25 | 2015-10-14 | 주식회사 진용 | 홍화씨 추출물, 보스웰리아 추출물 및 황금 추출물을 유효성분으로 함유하는 뼈 길이 성장 촉진용 및 골밀도 증가용 식품 조성물 |
| EP4190172A1 (en) | 2015-10-02 | 2023-06-07 | The Coca-Cola Company | Sweetener composition comprising steviol glycoside sweeteners with improved flavor profiles |
| AU2017241771B2 (en) | 2016-03-28 | 2023-02-02 | The Coca-Cola Company | Sweetness and taste improvement of steviol glycoside or mogroside sweeteners with flavonoids |
| AU2017246876B2 (en) | 2016-04-06 | 2021-09-23 | The Coca-Cola Company | Sweetness and taste improvement of steviol glycoside or mogroside sweeteners |
| JP6706959B2 (ja) * | 2016-04-07 | 2020-06-10 | ポーラ化成工業株式会社 | プロテオグリカン産生促進剤 |
| KR101924532B1 (ko) * | 2016-09-07 | 2018-12-03 | 연세대학교 산학협력단 | 서어나무속 추출물을 포함하는 골질환의 예방 또는 치료용 약학 조성물 |
| JP2018080148A (ja) * | 2016-11-18 | 2018-05-24 | 株式会社東洋新薬 | 更年期症状の予防及び/又は改善組成物 |
| US11653606B2 (en) | 2016-12-01 | 2023-05-23 | Purecircle Usa Inc. | Stevia plant and uses thereof |
| CN110868870A (zh) | 2017-05-12 | 2020-03-06 | 艾斯姆食品公司 | 大米产物及制备它们的系统和方法 |
| CN111295101B (zh) | 2017-06-27 | 2023-09-01 | 可口可乐公司 | 口服甜味剂组合物和方法 |
| CN111683671A (zh) | 2017-10-06 | 2020-09-18 | 嘉吉公司 | 制备马黛茶提取物组合物的方法 |
| JP7328670B2 (ja) * | 2018-04-13 | 2023-08-17 | 養命酒製造株式会社 | ロコモティブシンドローム改善用組成物 |
| AU2019263007A1 (en) | 2018-04-30 | 2020-12-03 | The Coca-Cola Company | Methods for improving the solubility of steviol glycoside mixtures, and uses |
| WO2019217310A1 (en) | 2018-05-08 | 2019-11-14 | Epc Natural Products Co., Ltd. | Sweetener and flavor compositions, methods of making and methods of use thereof |
| KR102186886B1 (ko) * | 2018-07-11 | 2020-12-04 | 한국 한의학 연구원 | 프루누스 자마사쿠라 추출물을 유효성분으로 함유하는 골다공증의 예방, 개선 또는 치료용 조성물 |
| WO2020123067A2 (en) | 2018-10-12 | 2020-06-18 | Epc Natural Products Co., Ltd. | Sweetener and flavor compositions, methods of making and methods of use thereof |
| KR20200064776A (ko) * | 2018-11-29 | 2020-06-08 | 경희대학교 산학협력단 | 도인 추출물을 포함하는 골절 예방, 개선 또는 치료용 조성물 |
| WO2020210118A1 (en) | 2019-04-06 | 2020-10-15 | Cargill, Incorporated | Sensory modifiers |
| EP4033915A4 (en) | 2019-09-27 | 2023-05-31 | The Coca-Cola Company | ENHANCED TASTE SWEETENER BLENDS |
| KR102255000B1 (ko) * | 2020-04-29 | 2021-05-24 | 한국 한의학 연구원 | 박하 추출물을 유효성분으로 함유하는 골질환의 예방, 개선 또는 치료용 조성물 |
| KR102612089B1 (ko) * | 2021-01-28 | 2023-12-14 | 대한민국 | 생강나무 잎 추출물의 유효성분을 포함하는 골다공증 예방 및 치료용 조성물 |
| AU2022340820A1 (en) | 2021-09-01 | 2024-03-28 | The Coca-Cola Company | Methods and compositions comprising caffeine and/or a derivative thereof and a polyphenol |
| CN114886115A (zh) * | 2022-04-18 | 2022-08-12 | 绵阳膳意生物科技有限公司 | 一种24节气均衡膳食组合物 |
| WO2024081959A2 (en) | 2022-10-14 | 2024-04-18 | The Coca-Cola Company | Methods and compositions for improving cognitive and mood functions |
Family Cites Families (60)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4556564A (en) * | 1983-03-14 | 1985-12-03 | Ethyl Corporation | Feeding of zeolite A to poultry |
| FR2580929B1 (fr) * | 1984-09-10 | 1988-10-07 | Imbert Nicole | Composition a base de plantes pour soulager et apaiser les douleurs de l'arthrose |
| US6150328A (en) | 1986-07-01 | 2000-11-21 | Genetics Institute, Inc. | BMP products |
| IE69026B1 (en) * | 1987-06-12 | 1996-08-07 | Immuno Ag | Novel proteins with factor VIII activity process for their preparation using genetically-engineered cells and pharmaceutical compositions containing them |
| EP0517836B1 (en) * | 1990-03-02 | 1999-09-22 | The Partnership Of Isaac G. Eliaz And Shmuel Gonen | Method and product for promoting hair growth and treating skin conditions |
| US5162113A (en) * | 1991-05-15 | 1992-11-10 | Oh Jung M | Oral composition for treating bone disorders |
| WO1995008318A2 (fr) * | 1993-09-15 | 1995-03-30 | Valentina Nikolaevna Peidus | Substance medicinale a proprietes anti-infecteuses destinee a etre utilisee dans le traitement de la sterilite et dans la protection de la grossesse |
| US5506211A (en) * | 1994-05-09 | 1996-04-09 | The Uab Research Foundation | Genistein for use in inhibiting osteroclasts |
| JPH08119869A (ja) * | 1994-10-26 | 1996-05-14 | Mikimoto Pharmaceut Co Ltd | 活性酸素抑制剤 |
| CA2205959A1 (en) | 1994-12-19 | 1996-06-27 | Michael Chorev | Continuous low-dose administration of parathyroid hormone or its agonist |
| AU4607596A (en) | 1994-12-20 | 1996-07-10 | Osteosa Inc. | Modulators of bone cell function and uses thereof |
| ZA95908B (en) * | 1995-02-06 | 1995-10-13 | Joshi Yeshwant Dr Kashinath | A synergistic formulation for treatment of rheumatic diseases |
| JP3009599B2 (ja) * | 1995-02-24 | 2000-02-14 | フジッコ株式会社 | フラボノイド配糖体を含む骨粗鬆症治療剤および骨粗鬆症治療用の可食性組成物 |
| CN1055845C (zh) * | 1995-06-05 | 2000-08-30 | 金纪前 | 跌打活血散 |
| WO1997011095A1 (en) | 1995-09-21 | 1997-03-27 | The Trustees Of Columbia University In The City Of New York | Uses of bone morphogenetic proteins |
| FR2740332B1 (fr) * | 1995-10-26 | 1997-12-05 | Oreal | Composition pour la deformation permanente des fibres keratiniques, contenant un antagoniste de substance p ou un antagoniste de cgrp |
| JPH09187248A (ja) * | 1995-11-06 | 1997-07-22 | Nippon Oil & Fats Co Ltd | 抗アレルギー食品 |
| RU2102460C1 (ru) * | 1996-05-13 | 1998-01-20 | Уфимский винодельческий завод | Композиция ингредиентов для бальзама "капова пещера" |
| CN1057218C (zh) * | 1996-07-01 | 2000-10-11 | 谢景龙 | 一种用于治疗疮疡及外伤感染的中药制剂及其制备方法 |
| JPH10130161A (ja) * | 1996-09-06 | 1998-05-19 | Otsuka Pharmaceut Co Ltd | ヘリコバクター・ピロリに対する組成物 |
| JPH10114653A (ja) * | 1996-10-11 | 1998-05-06 | Taishi Shokuhin Kogyo Kk | 骨形成促進および骨塩量減少防止用組成物 |
| CN1068189C (zh) * | 1997-10-13 | 2001-07-11 | 刘国柱 | 玉米红薯果粥粉配方及制作工艺 |
| JPH11130670A (ja) * | 1997-10-27 | 1999-05-18 | Oyo Seikagaku Kenkyusho | 骨粗鬆症治療剤 |
| ATE239687T1 (de) * | 1998-01-09 | 2003-05-15 | Eurovita As | Bestimmte diterpene, extrakte oder konzentrate von curcuma amada, die diese enthalten zur verwendung als medikamente |
| DE19812204A1 (de) * | 1998-03-19 | 1999-11-04 | Plantamed Arzneimittel Gmbh | Verwendung von Extrakten aus Cimicifuga racemosa und Belamcanda sinensis als estrogenartiges organselektives Arzneimittel ohne uterotrope Wirkung |
| US6123944A (en) * | 1998-03-19 | 2000-09-26 | Phytoceutica, Inc. | Icariin preparations |
| DE19814725A1 (de) * | 1998-04-02 | 1999-10-07 | Michael O Ruepp | Verwendung von Echinacea-pallida und Echinacea angustifolia-Extrakten; Löwenzahn-/Taraxacum)-Extrakten; Spitzwegerich-(Plantago)-Extrakten |
| WO1999055351A1 (en) * | 1998-04-23 | 1999-11-04 | Vladimir Leko | Herbal composition and medicament against diabetes mellitus type ii manufactured thereof |
| US5908628A (en) * | 1998-05-01 | 1999-06-01 | Hou; Liping | Compositions with analgesic, antipyretic and antiinflammatory properties |
| RU2138282C1 (ru) * | 1998-05-14 | 1999-09-27 | Суханов Александр Иванович | Полиартритный сбор |
| JP3479224B2 (ja) * | 1998-08-06 | 2003-12-15 | 森下仁丹株式会社 | ハーブ植物を有効成分とする閉経後骨粗鬆症の予防又は治療剤 |
| KR20000019869A (ko) * | 1998-09-16 | 2000-04-15 | 정세영 | 홍화자 추출물을 포함하는 경조직 재생촉진제 조성물 |
| GB9821670D0 (en) * | 1998-10-05 | 1998-12-02 | Muhlbauer Roman C | Plant extracts for the treatment of increased bone resorption |
| JP2000191526A (ja) * | 1998-12-25 | 2000-07-11 | Marumiya:Kk | 骨形成促進及び抗骨粗鬆症組成物 |
| JP2000281567A (ja) * | 1999-01-29 | 2000-10-10 | Sunstar Inc | スチルベン系化合物を含有する医薬及び食品組成物 |
| JP3403350B2 (ja) * | 1999-02-26 | 2003-05-06 | ハウス食品株式会社 | 紅茶抽出物及びこれを含むゲル状食品 |
| US20010024664A1 (en) * | 1999-03-19 | 2001-09-27 | Obukowicz Mark G. | Selective COX-2 inhibition from edible plant extracts |
| AUPQ008399A0 (en) * | 1999-04-28 | 1999-05-27 | Novogen Research Pty Ltd | Cariovascular applications |
| CN1084206C (zh) * | 1999-05-20 | 2002-05-08 | 周金龙 | 一种治疗癌症的药物及其制备方法 |
| AUPQ127399A0 (en) * | 1999-06-29 | 1999-07-22 | Guangzhou University Of Traditional Chinese Medicine | Compositions and methods for treating or preventing osteoporosis |
| US6274176B1 (en) * | 1999-07-01 | 2001-08-14 | Chromak Research, Inc. | Herbal compositions and their use as anti-inflammatory agents for alleviation of arthritis and gout |
| AUPQ138199A0 (en) | 1999-07-02 | 1999-07-29 | Telstra R & D Management Pty Ltd | A search system |
| DE60027296T2 (de) * | 1999-07-09 | 2006-12-28 | Sun Farm Corp., Milford | Diätzusatz zur behandlung von malignitäten und virusinfektionen und verbesserung der immunfunktion |
| GB2355189B (en) * | 1999-08-20 | 2004-07-28 | Yousef Haik Babikian | Herbal preparation for the treatment of diabetes mellitus |
| JP2001064192A (ja) * | 1999-08-25 | 2001-03-13 | Sunstar Inc | ランゲルハンス細胞の遊走抑制剤及び抗原提示抑制剤 |
| US6242012B1 (en) * | 1999-10-19 | 2001-06-05 | Thomas Newmark | Herbal composition for promoting hormonal balance in women and methods of using same |
| WO2001045725A2 (en) * | 1999-12-23 | 2001-06-28 | Ancile Pharmaceuticals, Inc. | Treatment for inflammatory bowel disease (ibd) and related conditions |
| CN1132598C (zh) * | 2000-01-03 | 2003-12-31 | 谭翠玉 | 治疗骨质增生的益肾通痹丸及其制备方法 |
| CN1122518C (zh) * | 2000-01-07 | 2003-10-01 | 徐柏功 | 一种治疗妇科病的药物及其制备方法 |
| CN1101202C (zh) * | 2000-02-22 | 2003-02-12 | 陈永贵 | 一种复方骨质增生中药制剂 |
| RU2162334C1 (ru) * | 2000-04-24 | 2001-01-27 | Титиева Наталья Михайловна | Гомеопатическое лекарственное средство для лечения костно-суставной патологии (остеоартроз, остеохандроз, артрозы, артриты) |
| JP2001302539A (ja) * | 2000-04-26 | 2001-10-31 | Meiji Seika Kaisha Ltd | 骨強化剤、骨強化用食品及び骨強化用飼料組成物 |
| JP2001314173A (ja) * | 2000-05-09 | 2001-11-13 | Kyoto Eiyo Kagaku Kenkyusho:Kk | 食品用組成物 |
| JP2002003382A (ja) * | 2000-06-20 | 2002-01-09 | Nihon Shokken Co Ltd | 関節軟骨再生促進及び軟骨減少防止用栄養補助組成物 |
| AU2001273759A1 (en) * | 2000-08-18 | 2002-02-25 | University Of Bern | Essential oils and chemically related species for the treatment of increased bone resorption |
| CN1120714C (zh) * | 2000-09-27 | 2003-09-10 | 陈忠彦 | 一种治疗股骨头坏死的中药制剂及其制备方法 |
| CN1163258C (zh) * | 2000-12-27 | 2004-08-25 | 孔昭苓 | 一种药物组合物及制备方法 |
| CN1136888C (zh) * | 2001-05-09 | 2004-02-04 | 陈志友 | 治疗骨质增生及腰椎间盘脱出症的外用药物及其制备方法 |
| EP1325681A1 (en) * | 2001-12-11 | 2003-07-09 | Société des Produits Nestlé S.A. | Composition for promotion of bone growth and maintenance of bone health |
| AU2003272902A1 (en) * | 2002-10-01 | 2004-04-23 | Takara Bio Inc. | Remedies |
-
2001
- 2001-12-11 EP EP01204838A patent/EP1325681A1/en not_active Withdrawn
-
2002
- 2002-12-10 DE DE60236961T patent/DE60236961D1/de not_active Expired - Lifetime
- 2002-12-10 US US10/497,809 patent/US20050079232A1/en not_active Abandoned
- 2002-12-10 CA CA2469551A patent/CA2469551C/en not_active Expired - Fee Related
- 2002-12-10 CN CNB028245172A patent/CN100521972C/zh not_active Expired - Fee Related
- 2002-12-10 IL IL16165902A patent/IL161659A0/xx unknown
- 2002-12-10 ES ES10176060T patent/ES2387876T3/es not_active Expired - Lifetime
- 2002-12-10 KR KR10-2004-7008341A patent/KR20040083466A/ko not_active Application Discontinuation
- 2002-12-10 PT PT10176060T patent/PT2263480E/pt unknown
- 2002-12-10 AT AT02805314T patent/ATE472943T1/de active
- 2002-12-10 DK DK02805314.8T patent/DK1455601T4/da active
- 2002-12-10 ES ES10176057T patent/ES2387877T3/es not_active Expired - Lifetime
- 2002-12-10 PT PT02805314T patent/PT1455601E/pt unknown
- 2002-12-10 EP EP02805314.8A patent/EP1455601B2/en not_active Expired - Lifetime
- 2002-12-10 ES ES02805314.8T patent/ES2347774T5/es not_active Expired - Lifetime
- 2002-12-10 BR BR0214838-2A patent/BR0214838A/pt not_active Application Discontinuation
- 2002-12-10 UA UA20040705594A patent/UA84123C2/ru unknown
- 2002-12-10 EP EP10176060A patent/EP2263480B1/en not_active Expired - Lifetime
- 2002-12-10 MX MXPA04005583A patent/MXPA04005583A/es active IP Right Grant
- 2002-12-10 DK DK10176057.7T patent/DK2286675T3/da active
- 2002-12-10 ES ES09172445T patent/ES2387634T3/es not_active Expired - Lifetime
- 2002-12-10 DK DK09172445.0T patent/DK2135517T3/da active
- 2002-12-10 EP EP09172445A patent/EP2135517B1/en not_active Expired - Lifetime
- 2002-12-10 PT PT09172445T patent/PT2135517E/pt unknown
- 2002-12-10 PT PT10176057T patent/PT2286675E/pt unknown
- 2002-12-10 EP EP10176058A patent/EP2272382A1/en not_active Ceased
- 2002-12-10 AU AU2002366734A patent/AU2002366734B2/en not_active Ceased
- 2002-12-10 PL PL02369768A patent/PL369768A1/xx unknown
- 2002-12-10 JP JP2003553934A patent/JP4653951B2/ja not_active Expired - Fee Related
- 2002-12-10 EP EP10176057A patent/EP2286675B1/en not_active Expired - Lifetime
- 2002-12-10 DK DK10176060.1T patent/DK2263480T3/da active
- 2002-12-10 RU RU2004121173/13A patent/RU2314717C2/ru not_active IP Right Cessation
- 2002-12-10 WO PCT/EP2002/014120 patent/WO2003053167A1/en active Application Filing
-
2004
- 2004-04-28 IL IL161659A patent/IL161659A/en not_active IP Right Cessation
- 2004-05-25 NO NO20042151A patent/NO327135B1/no not_active IP Right Cessation
- 2004-06-08 CO CO04053698A patent/CO5590866A2/es not_active Application Discontinuation
- 2004-07-08 ZA ZA2004/05454A patent/ZA200405454B/en unknown
-
2005
- 2005-09-16 HK HK05108118.5A patent/HK1076009A1/xx not_active IP Right Cessation
-
2008
- 2008-04-18 AU AU2008201726A patent/AU2008201726B2/en not_active Ceased
- 2008-08-01 US US12/184,800 patent/US20090060883A1/en not_active Abandoned
-
2009
- 2009-12-04 JP JP2009276784A patent/JP5001342B2/ja not_active Expired - Fee Related
- 2009-12-04 JP JP2009276785A patent/JP2010088442A/ja active Pending
-
2010
- 2010-03-23 AU AU2010201150A patent/AU2010201150B2/en not_active Ceased
Non-Patent Citations (2)
| Title |
|---|
| 大豆异黄酮防治骨质疏松的研究进展 . 李洁.上海预防医学杂志,第12卷第4期. 2000 |
| 大豆异黄酮防治骨质疏松的研究进展 . 李洁.上海预防医学杂志,第12卷第4期. 2000 * |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN100521972C (zh) | 用于促进骨生长及维持骨健康的组合物 | |
| CN100473291C (zh) | 用于骨健康的含有植物提取物的食物或宠物食物组合物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1076009 Country of ref document: HK |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: GR Ref document number: 1076009 Country of ref document: HK |
|
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20090805 Termination date: 20171210 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |