BRPI0815331B1 - Composição farmacêutica compreendendo um derivado de benzeno glucopiranosila substituída,bem como seu uso - Google Patents
Composição farmacêutica compreendendo um derivado de benzeno glucopiranosila substituída,bem como seu uso Download PDFInfo
- Publication number
- BRPI0815331B1 BRPI0815331B1 BRPI0815331-0A BRPI0815331A BRPI0815331B1 BR PI0815331 B1 BRPI0815331 B1 BR PI0815331B1 BR PI0815331 A BRPI0815331 A BR PI0815331A BR PI0815331 B1 BRPI0815331 B1 BR PI0815331B1
- Authority
- BR
- Brazil
- Prior art keywords
- dpp
- pharmaceutical composition
- benzene
- derivative
- inhibitor
- Prior art date
Links
- ILTRPILDWNCOMQ-UHFFFAOYSA-N CC#CC[n]1c(N(CCC2)CC2N)nc(N(C)C(N2Cc3nc(cccc4)c4cn3)=O)c1C2=O Chemical compound CC#CC[n]1c(N(CCC2)CC2N)nc(N(C)C(N2Cc3nc(cccc4)c4cn3)=O)c1C2=O ILTRPILDWNCOMQ-UHFFFAOYSA-N 0.000 description 1
- ZSBOMTDTBDDKMP-UHFFFAOYSA-N CN(C(C=C(N1Cc2ccccc2C#N)N(CCC2)CC2N)=O)C1=O Chemical compound CN(C(C=C(N1Cc2ccccc2C#N)N(CCC2)CC2N)=O)C1=O ZSBOMTDTBDDKMP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7028—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages
- A61K31/7034—Compounds having saccharide radicals attached to non-saccharide compounds by glycosidic linkages attached to a carbocyclic compound, e.g. phloridzin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7048—Compounds having saccharide radicals and heterocyclic rings having oxygen as a ring hetero atom, e.g. leucoglucosan, hesperidin, erythromycin, nystatin, digitoxin or digoxin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
- A61K31/52—Purines, e.g. adenine
- A61K31/522—Purines, e.g. adenine having oxo groups directly attached to the heterocyclic ring, e.g. hypoxanthine, guanine, acyclovir
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/141—Intimate drug-carrier mixtures characterised by the carrier, e.g. ordered mixtures, adsorbates, solid solutions, eutectica, co-dried, co-solubilised, co-kneaded, co-milled, co-ground products, co-precipitates, co-evaporates, co-extrudates, co-melts; Drug nanoparticles with adsorbed surface modifiers
- A61K9/145—Intimate drug-carrier mixtures characterised by the carrier, e.g. ordered mixtures, adsorbates, solid solutions, eutectica, co-dried, co-solubilised, co-kneaded, co-milled, co-ground products, co-precipitates, co-evaporates, co-extrudates, co-melts; Drug nanoparticles with adsorbed surface modifiers with organic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/16—Agglomerates; Granulates; Microbeadlets ; Microspheres; Pellets; Solid products obtained by spray drying, spray freeze drying, spray congealing,(multiple) emulsion solvent evaporation or extraction
- A61K9/1605—Excipients; Inactive ingredients
- A61K9/1617—Organic compounds, e.g. phospholipids, fats
- A61K9/1623—Sugars or sugar alcohols, e.g. lactose; Derivatives thereof; Homeopathic globules
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2013—Organic compounds, e.g. phospholipids, fats
- A61K9/2018—Sugars, or sugar alcohols, e.g. lactose, mannitol; Derivatives thereof, e.g. polysorbates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2072—Pills, tablets, discs, rods characterised by shape, structure or size; Tablets with holes, special break lines or identification marks; Partially coated tablets; Disintegrating flat shaped forms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4858—Organic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/48—Drugs for disorders of the endocrine system of the pancreatic hormones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/48—Drugs for disorders of the endocrine system of the pancreatic hormones
- A61P5/50—Drugs for disorders of the endocrine system of the pancreatic hormones for increasing or potentiating the activity of insulin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- Diabetes (AREA)
- Biophysics (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Endocrinology (AREA)
- Dermatology (AREA)
- Heart & Thoracic Surgery (AREA)
- Urology & Nephrology (AREA)
- Cardiology (AREA)
- Oncology (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Ophthalmology & Optometry (AREA)
- Gastroenterology & Hepatology (AREA)
- Emergency Medicine (AREA)
- Communicable Diseases (AREA)
- Child & Adolescent Psychology (AREA)
- Vascular Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Saccharide Compounds (AREA)
Abstract
Description
- - para prevenir, reduzir a evolução, retardar, ou tratar um distúrbio metabólico;
- - para melhorar o controle glicêmico e/ou reduzir a glicose plasmática de jejum, a glicose plasmática pós-prandial e/ou a hemoglobina glicosilada HbA1c;
- - para prevenir, reduzir, retardar ou reverter a evolução de tolerância à glicose prejudicada, glicose sanguínea de jejum prejudicada, resistência à insulina e/ou da síndrome metabólica associada ao diabetes melito tipo 2;
- - para prevenir, reduzir a evolução, retardar ou tratar de uma condição ou distúrbio selecionado do grupo que consiste em complicações do diabetes melito;
- - para reduzir o peso corporal ou prevenir o aumento do peso corporal ou facilitar a redução do peso corporal;
- - para prevenir ou tratar a degeneração das células beta pancreáticas e/ou melhorar e/ou restaurar a funcionalidade das células beta pancreáticas e/ou restaurar a funcionalidade da secreção de insulina pancreática;
- - para prevenir, reduzir, retardar ou tratar doenças ou condições atribuídas a uma acumulação anormal de gordura hepática;
- - manter e/ou melhorar a sensibilidade à insulina e/ou para tratar ou prevenir a hiperinsulinemia e/ou resistência à insulina,
- (1) Wright, E.M. (2001) Am. J. Renal Physiol. 280, F10-F18;
- (2) Wright, E.M. et al. (2004) Pflugers Arch. 447(5):510-8;
- (3) You, G. et al. (1995) J. Biol. Chem. 270 (49) 29365-29371;
- (4) Pajor AM, Wright EM (1992) J Biol. Chem. 267(6):3557-3560;
- (5) Zhou, L. et al. (2003) J. Cell. Biochem. 90:339-346;
- (6) Diez-Sampedro, A. et al. (2003) Proc. Natl. Acad. Sci. USA 100(20), 11753-11758;
- (7) Tabatabai, N.M. (2003) Kidney Int. 64, 1320-1330;
- (8) Curtis, R.A.J. (2003) US Patent Appl. 2003/0054453;
- (9) Bruss,M. & Bonisch,H. (2001) Cloning and functional characterization of a new human sugar transporter in kidney (Genbank Acc. No. AJ305237);
- (10) Rossetti, L. Et al. (987) J. Clin. Invest. 79, 1510-1515;
- (11) Gouvea, W.L. (1989) Kidney Int. 35(4):1041-1048.
- sitagliptina (MK-0431) que tem a fórmula estrutural A abaixo é (3R)-3-amino-1-[3-(trifluormetil)-5,6,7,8-tetra-hidro-5H-[1,2,4]triazol[4,3-a]pirazin-7-il]-4-(2,4,5-trifluorfenil)butan-1-ona, também conhecida como (2R)-4-oxo-4-[3-(trifluormetil)-5,6-di-hidro[1,2,4]triazol[4,3-a]pirazin-7(8H)-il]-1-(2,4,5-trifluorfenil)butan-2-amina,Em uma modalidade, a sitagliptina está na forma de seu sal de di-hidrogenofosfato, isto é, fosfato de sitagliptina. Em uma outra modalidade, o fosfato de sitagliptina está na forma de um anidrato ou mono-hidrato cristalino. Uma classe desta modalidade refere-se ao mono-hidrato de fosfato de sitagliptina. A base livre da sitagliptina e sais farmaceuticamente aceitáveis da mesma estão descritos na Patente US N° 6.699.871 e no Exemplo 7 do documento WO 03/004498. O mono-hidrato de fosfato de sitagliptina cristalino está descrito no documento WO 2005/003135 e no documento WO 2007/050485. Para detalhes, por exemplo sobre um processo para produzir ou formular este composto ou um sal do mesmo, é feito portanto referência a esses documentos. Uma formulação de comprimido para a sitagliptina encontra-se comercialmente disponível sob o nome comercial Januvia®.
Para detalhes, por exemplo sobre um processo para produzir ou formular este composto ou um sal do mesmo, é feito portanto referência a esses documentos. Uma formulação de comprimido para vildagliptina está comercialmente disponível sob o nome comercial Galvus®.
Esses compostos e métodos para sua preparação estão descritos no documento WO 03/037327. O sal de mesilato do primeiro composto assim como polimorfos cristalinos do mesmo estão descritos no documento WO 2006/100181. O sal de fumarato do último composto assim como polimorfos cristalinos do mesmo estão descritos no documento WO 2007/071576. Esses compostos podem ser formulados como uma composição farmacêutica da maneira descrita no documento WO 2007/017423. Para detalhes, por exemplo sobre um processo para produzir, formular ou usar esses compostos ou um sal dos mesmos, é feito portanto referência a esses documentos.
Este composto e métodos para sua preparação estão descritos nos documentos WO 2005/116014 e US 7291618. Para detalhes, por e-xemplo sobre o processo de fabricação, formulação e uso deste composto ou um sal do mesmo, é feito portanto referência a esses documentos.
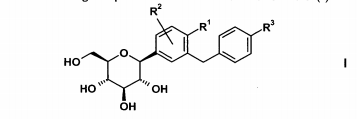
glucopiranosila substituída de fórmula (I)onde R1 representa Cl, metila ou ciano; R2 representa H, metila, metóxi ou hidróxi e R3 representa etila, ciclopropila, etinila, etóxi, (R)-tetra-hidrofuran-3-ilóxi ou (S)-tetra-hidrofuran-3-ilóxi,seja, em uma primeira modalidade (modalidade A), em combinação com
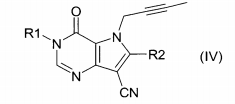
um inibidor de DPP IV de fórmula (I)ou de fórmula (II) ou de fórmula (III) ou de fórmula (IV) onde R1 representa ([1,5]naftiridin-2-il)metila, (quinazolin-2-il)metila, (quinoxalin-6-il)metila, (4-metil-quinazolin-2-il)metila, 2-ciano-benzila, (3-ciano-quinolin-2-il)metila, (3-ciano-piridin-2-il)metila, (4-metil-pirimidin-2-il)metila, ou (4,6-dimetil-pirimidin-2-il)metil e R2 representa 3-(R)-amino-piperidin-1-ila, (2-amino-2-metil-propil)-metilamino ou (2-(S)-amino-propil)-metilamino, ou seu sal farmaceuticamente aceitável;
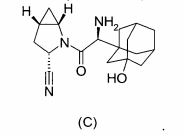
(2S)-1-{[2-(5-Metil-2-fenil-oxazol-4-il)-etilamino]-acetil}-pirrolidina-2-carbonitrila, (2S)-1-{[1,1,-Dimetil-3-(4-piridin-3-il-imidazol-1-il)-propilamino]-acetil}-pirrolidina-2-carbonitrila,
(S)-1-((2S,3S,11 bS)-2-Amino-9,10-dimetóxi-1,3,4,7,11 b-hexa-hidro-2H-pirido[2,1-a]isoquinolin-3-il)-4-fluormetil-pirrolidin-2-ona,
(3,3-Difluorpirrolidin-1-il)-((2S,4S)-4-(4-(pirimidin-2-il)piperazin-1-il)pirrolidin-2-il)metanona,
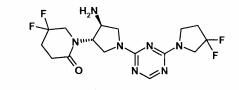
(1((3S,4S)-4-amino-1-(4-(3,3-difluorpirrolidin-1-il)-1,3,5-triazin-2-il)pirrolidin-3-il)-5,5-difluorpiperidin-2-ona,
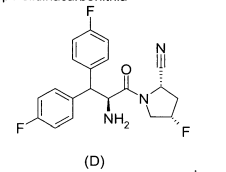
(2S,4S)-1 -{2-[(3S,1 R)-3-(1 H-1,2,4-Triazol-1 -ilmetil)ciclopentilamino]-acetil}-4-fluorpirrolidina-2-carbonitrila, e
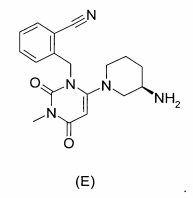
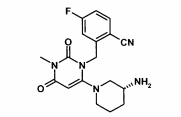
(R)-2-[6-(3-Amino-piperidin-1-il)-3-metil-2,4-dioxo-3,4-di-hidro-2H-pirimidin-1-ilmetil]-4-flúor-benzonitriia,
ou um sal farmaceuticamente aceitável das mesmas.
- - prevenir, reduzir a evolução, retardar ou tratar um distúrbio metabólico selecionado do grupo que consiste em diabetes melito tipo 1, diabetes melito tipo 2, tolerância à glicose prejudicada (IGT), glicose sanguínea de jejum prejudicada (IFG), hiperglicemia, hiperglicemia pós-prandial, excesso de peso, obesidade e síndrome metabólica; ou
- - melhorar o controle glicêmico e/ou reduzir a glicose plasmática de jejum, a glicose plasmática pós-prandial e/ou a hemoglobina glicosilada HbA1c; ou
- - prevenir, reduzir, retardar ou reverter a evolução de tolerância à glicose prejudicada (IGT), glicose sanguínea de jejum prejudicada (IFG), resistência à insulina e/ou da síndrome metabólica associada ao diabetes melito tipo 2; ou
- - prevenir, reduzir a evolução, retardar ou tratar uma condição ou distúrbio selecionado do grupo que consiste em complicações do diabetes melito tais como cataratas e doenças microvasculares e macrovascula-res, tais como nefropatia, retinopatia, neuropatia, isquemia tecidual, arteriosclerose, infarto do miocárdio, acidente vascular cerebral e doença arterial oclusiva periférica; ou
- - reduzir o peso corporal ou prevenir o aumento do peso corporal ou facilitar a redução do peso corporal; ou
- - prevenir, reduzir, retardar ou tratar a degeneração das células beta pancreáticas e/ou a diminuição da funcionalidade das células beta pancreáticas e/ou melhorar e/ou restaurar a funcionalidade das células beta pancreáticas e/ou restaurar a funcionalidade da secreção de insulina pancreática; ou
- - prevenir, reduzir, retardar ou tratar doenças ou condições a-tribuídas a uma acumulação anormal de gordura hepática; ou
- - manter e/ou melhorar a sensibilidade à insulina e/ou tratar ou prevenir a hiperinsulinemia e/ou resistência à insulina;
- - prevenir, reduzir a evolução, retardar ou tratar um distúrbio metabólico selecionado do grupo que consiste em diabetes melito tipo 1, diabetes melito tipo 2, tolerância à glicose prejudicada (IGT), glicose sanguínea de jejum prejudicada (IFG), hiperglicemia, hiperglicemia pós-prandial, excesso de peso, obesidade e síndrome metabólica; ou
- - melhorar o controle glicêmico e/ou reduzir a glicose plasmática de jejum, a glicose plasmática pós-prandial e/ou a hemoglobina glicosilada HbA1c; ou
- - prevenir, reduzir, retardar ou reverter a evolução de tolerância à glicose prejudicada (IGT), glicose sanguínea de jejum prejudicada (IFG), resistência à insulina e/ou da síndrome metabólica associada ao dia-betes melito tipo 2; ou
- - prevenir, reduzir a evolução, retardar ou tratar uma condição ou distúrbio selecionado do grupo que consiste em complicações do diabetes melito tais como cataratas e doenças microvasculares e macrovasculares, tais como nefropatia, retinopatia, neuropatia, isquemia tecidual, arteriosclerose, infarto do miocárdio, acidente vascular cerebral e doença arterial oclusiva periférica; ou
- - reduzir o peso corporal ou prevenir o aumento do peso corporal ou facilitar a redução do peso corporal; ou
- - prevenir, reduzir, retardar ou tratar a degeneração das células beta pancreáticas e/ou a diminuição da funcionalidade das células beta pancreáticas e/ou melhorar e/ou restaurar a funcionalidade das células beta pancreáticas e/ou restaurar a funcionalidade da secreção de insulina pancreática; ou
- - prevenir, reduzir, retardar ou tratar doenças ou condições a-tribuídas a uma acumulação anormal de gordura hepática; ou
- - manter e/ou melhorar a sensibilidade à insulina e/ou tratar ou prevenir a hiperinsulinemia e/ou resistência à insulina;
HOMA-IR = [insulina sérica de jejum (μU/mL)] x [glicose plasmática de jejum (mmol/L)/22,5]
- 1. obesidade abdominal, definida como circunferência da cintura > 40 polegadas ou 102 cm em homens, e > 35 polegadas ou 94 cm em mulheres; ou com respeito à etnia japonesa ou pacientes japoneses, definida como circunferência da cintura > 85 cm em homens e > 90 cm em mulheres;
- 2. triglicerídeos: = 150 mg/dL
- 3. colesterol HDL < 40 mg/dL em homens
- 4. pressão sanguínea > 130/85 mm Hg (SBP > 130 ou DBP > 85)
- 5. glicose sanguínea de jejum = 110 mg/dL
De preferência R1 representa cloro ou ciano; em particular cloro.
De preferência R2 representa H.
De preferência R3 representa etila, ciclopropila, etinila, (R)-tetra-hidrofuran-3-ilóxi ou (S)-tetra-hidrofuran-3-i\óxi. Ainda mais preferivelmente R3 representa ciclopropila, etinila, (R)-tetra-hidrofuran-3-ilóxi ou (S)-tetra-hidrofuran-3-ilóxi.
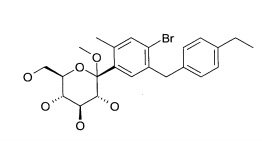
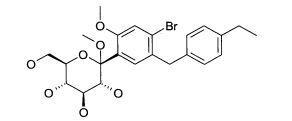
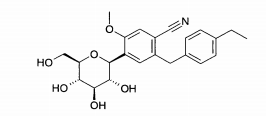
- (1) 6-(4-Etilbenzil)-4-(β-D-glucopiranos-1-il)-2-metóxi-benzonitrila
- (2) 2-(4-Etilbenzil)-4-(β-D-glucopiranos-1-il)-5-metóxi-benzonitrila
- (3) 1 -Ciano-2-(4-etilbenzil)-4-(β-D-glucopiranos-1 -il)-5-metil-benzeno
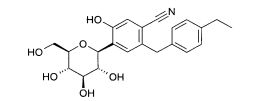
- (4) 2-(4-Etilbenzil)-4-(β-D-glucopiranos-1-il)-5-hidróxi-benzonitrila
- (5) 2-(4-Etilbenzil)-4-(β-D-glucopiranos-1 -il)-benzonitrila
- (6) 2-(4-Ciclopropil-benzil)-4-(β -D-glucopiranos-1 -il)-benzonitrila
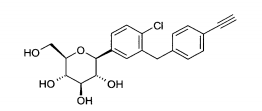
- (7) 1 -cloro-4-(β-D-glucopiranos-1 -il)-2-(4-etinil-benzil)-benzeno
- (8) 1 -cloro-4-(β-D-glucopiranos-1 -il)-2-[4-((R)-tetra-hidrofuran-3-ilóxl)-benzil]-benzeno
- (9) 1 -cloro-4-(β-D-glucopiranos-1 -il)-2-[4-{(S)-tetra-hidrofuran-3-ilóxl)-benzil]-benzeno
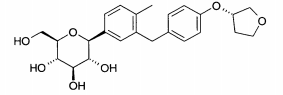
- (10) 1-Metil-2-[4-(R)-tetra-hidrofuran-3-ilóxi)-benzil]-4-(3-D-glucopiranos-1-il)-benzeno
- (11) 1-Metil-2-[4-(S)-tetra-hidrofuran-3-ilóxi)-benzil]-4-(β-D-glucopiranos-1-il)-benzeno
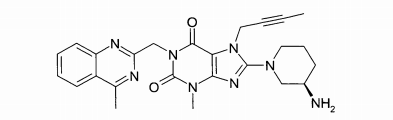
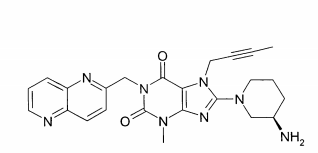
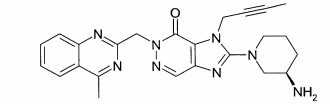
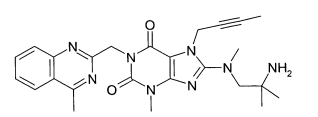
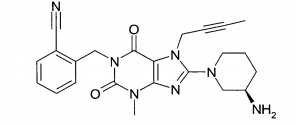
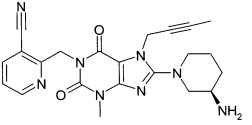
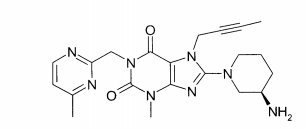
(A): 1 -[(4-metil-quinazolin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8-(3-(R)-amino-piperidin-1-il)-xantina (conforme WO 2004/018468, exemplo 2(142)):(B): 1 -[([1,5]naftiridin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8-((R)- 3-amino-piperidin-1-il)-xantina (conforme WO 2004/018468, exemplo 2(252)): (C):1-[(quinazolin-2-il)metil]-3-metil-7-(2-butin-1-il)-8-((R)-3-amino-piperidin-1 -il)-xantina (conforme WO 2004/018468, exemplo 2(80)): (D): 2-((R)-3-amino-piperidin-1 -il)-3-(but-2-inil)-5-(4-metil- quinazolin-2-ilmetil)-3,5-di-hidro-imidazo[4,5-d]piridazin-4-ona (conforme WO 2004/050658, exemplo 136): (E): 1-[(4-metil-quinazolin-2-il)metil]-3-metil-7-(2-butin-1-il)-8- [(2-amino-2-metil-propil)-metilamino]-xantina (conforme WO 2006/029769, exemplo 2(1)): (F): 1 -[(3-ciano-quinolin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8-((R)-3-amino-piperidin-1-il)-xantina (conforme WO 2005/085246, exemplo 1(30)): (G): 1 -(2-ciano-benzil)-3-metil-7-(2-butin-1 -il)-8-((R)-3-amino- piperidin-1-il)-xantina (conforme WO 2005/085246, exemplo 1(39)): (H): 1 -[(4-metil-quinazolin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8- [(S)-(2-amino-propil)-metilamino]-xantina (conforme WO 2006/029769, e-xemplo 2(4)): (I): 1 -[(3-ciano-piridin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8-((R)-3-amino-piperidin-1-il)-xantina (conforme WO 2005/085246, exemplo 1(52)): (J): 1 -[(4-metil-pirimidin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8-((R)-3-amino-piperidin-1-il)-xantina (conforme WO 2005/085246, exemplo 1(81)): (K): 1-[(4,6-dimetil-pirimidin-2-il)metil]-3-metil-7-(2-butin-1-il)- 8-((R)-3-amino-piperidin-1-il)-xantina (conforme WO 2005/085246, exemplo 1(82)): (L): 1 -[(quinoxalin-6-il)metil]-3-metil-7-(2-butin-1 -il)-8-((R)-3-amino-piperidin-1-il)-xantina (conforme WO 2005/085246, exemplo 1(83)):
Tabela 1
- (a) uma concentração de glicose sanguínea de jejum ou de glicose sérica maior que 110 mg/dL, em particular maior que 125 mg/dL;
- (b) um nível de glicose plasmática pós-prandial maior ou igual a 140 mg/dL;
- (c) um valor de HbA1c maior ou igual a 6,5 %, em particular maior ou igual a 8,0 %.
- - resistência à insulina,
- - hiperinsulinemia,
- - pré-diabetes,
- - diabetes melito tipo 2, em particular diabetes melito tipo 2tardio,
- - diabetes melito tipo 1.
- (a) obesidade (incluindo obesidade classe I, II e/ou III), obesidade visceral e/ou obesidade abdominal,
- (b) nível sanguíneo de triglicerídeos ≥150 mg/dL,
- (c) nível sanguíneo de colesterol HDL < 40 mg/dL em mulheres e < 50 mg/dL em homens,
- (d) uma pressão sanguínea sistólica >130 mm Hg e uma pressão sanguínea diastólica ≥ 85 mm Hg,
- (e) um nível de glicose sanguínea de jejum ≥110 mg/dL.
- (a) um primeiro recipiente contendo uma forma de dosagem compreendendo o derivado de benzeno glucopiranosila substituída e pelo menos um veículo farmaceuticamente aceitável, e
- (b) um segundo recipiente contendo uma forma de dosagem compreendendo o inibidor de DPP IV e pelo menos um veículo farmaceuticamente aceitável.
as composições farmacêuticas e os métodos de acordo com esta invenção podem ser testados em animais geneticamente hiperinsulinêmi-cos ou diabéticos como camundongos db/db, camundongos ob/ob, ratos Zucker obesos (fa/fa) ou ratos Zucker obesos diabéticos (ZDF). Além disso, eles podem ser testados em animais com diabetes induzido experimentalmente como ratos HanWistar ou Sprague Dawley pré-tratados com estreptozotocina.
fBu ter-butil
dba dibenzilidenoacetona
DMF dimetilformamida
DMSOdimetil sulfóxido
NMP N-metil-2-pirrolidona
THF tetra-hidrofurano
Preparação dos compostos de partida:
Exemplo I
Rendimento: 27,2 g (96% da teoria)
Espectro de massa (ESI-): m/z = 339/341 (Br) [M-H]-
O composto a seguir pode ser obtido de maneira análoga ao Exemplo I:
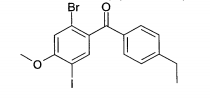
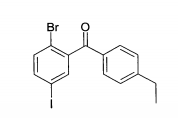
(1) (2-Bromo-5-iodo-4-metóxi-fenil)-(4-etil-fenil)-metanonaEspectro de massa (ESI+): m/z = 445/447 (Br) [M+H]+
O material de partida, (2-bromo-4-metóxi-fenil)-(4-etil-fenil)-metanona, é preparado da maneira descrita nos Exemplos II e III.
Exemplo II
Rendimento: 30,8 g (97% da teoria)
Espectro de massa (ESI+): m/z = 415/417 (Br) [M+H]+
Os compostos a seguir podem ser obtidos de maneira análoga ao Exemplo II:
(1) (2-Bromo-5-iodo-4-metil-fenil)-(4-etil-fenil)-metanonaEspectro de massa (ESI+): m/z = 429/431 (Br) [M+H]+ (2) (2-Bromo-4-flúor-fenil)-(4-etil-fenil)-metanona Espectro de massa (ESI+): m/z = 307/309 (Br) [M+H]+
Exemplo III
Rendimento: 33,7 g (75% da teoria)
Espectro de massa (ESI+): m/z = 319/321 (Br) [M+H]+
Exemplo IV
Rendimento: 21 g (68% da teoria)
Espectro de massa (ESI+): m/z = 418/420 (Br) [M+NH4]+
Os compostos a seguir podem ser obtidos de maneira análoga ao Exemplo IV:
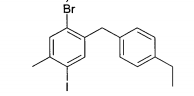
(1) 4-Bromo-5-(4-etilbenzil)-1 -iodo-2-metil-benzenoEspectro de massa (ESI+): m/z = 432/434 (Br) [M+NH4]+ (2) 4-Bromo-5-(4-etilbenzil)-1 -iodo-2-metóxi-benzenoEspectro de massa (ESI+): m/z = 448/450 (Br) [M+NH4]+
Exemplo V
Rendimento: 10,0 g (58% da teoria)
Espectro de massa (ESI+): m/z = 330/332 (Br) [M+H]+
Exemplo VI
Rendimento: 41,0 g (82% da teoria)
Exemplo VIl
Rendimento: 38,0 g (79% da teoria)
Exemplo VIII
Rendimento: 7,8 g (93% da teoria)
Os compostos a seguir podem ser obtidos de maneira análoga ao Exemplo VIII:
(1) 1 -Bromo-2-(4-etilbenzil)-4-(1 -metóxi-D-glucopiranos-1 -il)-benzenoEspectro de massa (ESI-): m/z = 511/513 (Br) [M+HCOO]-
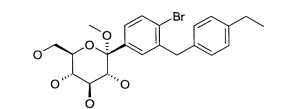
(2) 1 -Bromo-2-(4-etilbenzil)-4-(1 -metóxi-D-glucopiranos-1 -il)-5-metil-benzeno
(3) 1 -Bromo-2-(4-etilbenzil)-4-(1 -metóxi-D-glucopiranos-1 -il)-5-metóxi-benzenoExemplo IX
Rendimento: 0,5 g (7% da teoria)
Exemplo X
Rendimento: 6,78 g (60% da teoria)
Espectro de massa (ESI+): m/z = 610/612 (Br) [M+NH4]+
Os compostos a seguir podem ser obtidos de maneira análoga ao Exemplo X:
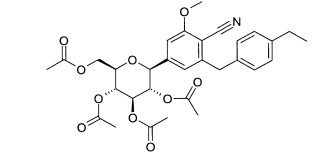
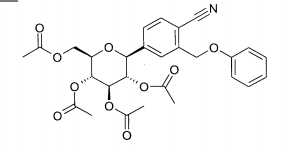
(1) 1 -Bromo-2-(4-etilbenzil)-4-(2,3,4,6-tetra-O-acetil-β-D-glucopiranos-1 -il)-benzenoEspectro de massa (ESI+): m/z = 622/624 [M+NH4]+
(2) 1-Bromo-2-(4-etilbenzil)-4-(2,3,4,6-tetra-O-acetil-β-D-glucopiranos-1-il)-5-metóxi-benzenoEspectro de massa (ESI+): m/z = 652/654 (Br) [M+NH4]+
(3) 6-(4-Etilbenzil)-4-(2,3,4,6-tetra-O-acetil-β-D-glucopiranos-1-il)-2-metóxi-benzonitrilaEspectro de massa (ESI+): m/z = 599 [M+NH4]+
A redução é conduzida em 6-(4-etilbenzil)-4-(1-metóxi-D-glucopiranos-1-il)-2-metóxi-benzonitrila por analogia com o procedimento descrito acima.
(4) 1 -Bromo-2-(4-etilbenzil)-4-(β-D-glucopiranos-1 -il)-5-metil-benzenoEspectro de massa (ESI+): m/z = 468/470 (Br) [M+NH4]+
Este composto é isolado com os grupos hidroxila livres depois de terminada a redução de acordo com o procedimento descrito acima.
Exemplo XI
Rendimento: 4,10 g (84% da teoria)
Espectro de massa (ESI+): m/z = 557 [M+NH4]+
Alternativamente, o composto também pode ser obtido empregando-se os procedimentos descritos nos Exemplos XII e 3
Exemplo XII
Rendimento: 1,1 g (75% da teoria)
Espectro de massa (ESI+): m/z = 583 [M+NH4]+
Este composto também pode ser preparado usando-se os procedimentos descritos para os Exemplos XI e 3.
Exemplo XIII
Rendimento: 0,52 g (75% da teoria)
Espectro de massa (ESI+): m/z = 543/545 (Br) [M+NH4]+
Exemplo XIV
Rendimento: 2,92 g (60% da teoria)
Espectro de massa (ESI-): m/z = 207 (Cl) [M+HCOO]-
Preparação dos compostos finais:
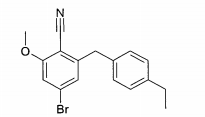
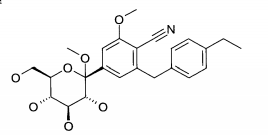
Exemplo (1): 6-(4-Etilbenzil)-4-(β-D-glucopiranos-1-il)-2-metóxi-benzonitrila
Rendimento: 65 mg (57% da teoria)
Espectro de massa (ESI+): m/z = 431 [M+NH4]+
O composto a seguir é obtido de maneira análoga ao Exemplo1:
Exemplo (2): 2-(4-Etilbenzil)-4-(β-D-glucopiranos-1 -il)-5-metóxi-benzonitrilaEspectro de massa (ESI+): m/z = 431 [M+NH4]+
Exemplo (3): 1 -Ciano-2-(4-etilbenzil)-4-(β-D-glucopiranos-1 -il)-5-metil-benzeno
Rendimento: 0,30 g (85% da teoria)
Espectro de massa (ESI+): m/z = 415 [M+NH4]+
Exemplo (4): 2-(4-Etilbenzil)-4-(β-D-qlucopiranos-1-il)-5-hidróxi-benzonitrila
Rendimento: 0,25 g (46 % da teoria)
Espectro de massa (ESI+): m/z = 398 [M-H]"
Exemplo (5): 2-(4-Etilbenzil)-4-(β-D-glucopiranos-1-il)-benzoni-trila
Rendimento: 0,51 g (81% da teoria)
Espectro de massa (ESI+): m/z = 401 [M+NH4]+
Exemplo (6): 2-(4-Ciclopropil-benzil)-4-(β-D-glucopiranos-1-il)- benzonitrila
Rendimento: 0,91 g (76% da teoria)
Espectro de massa (ESI+): m/z = 413 [M+NH4]+
Exemplo (7): 1 -cloro-4-(β-D-glucopiranos-1 -il)-2-(4-etinil-benzil)- benzenoO composto (7) pode ser vantajosamente preparado de acordo com o exemplo 12 descrito no documento WO 2005/092877.
Exemplo 1: ampola seca contendo 75 mg de substância ativa por 10 ml Composição:
Substância ativa 75,0 mg
Manitol 50,0 mg
água para injeção ad 10,0 ml
Preparação:
A substância ativa e manitol são dissolvidos em água. Depois de acondicionada, a solução é liofilizada. Para produzir uma solução pronta para uso, o produto é dissolvido em água para injeção.
Substância ativa 35,0 mg
Manitol 100,0 mg
água para injeção ad 2,0 ml
Preparação:
A substância ativa e manitol são dissolvidos em água. Depois de acondicionada, a solução é liofilizada.
Para produzir uma solução pronta para uso, o produto é dissolvido em água para injeção.
Composição:Preparação:
(1), (2) e (3) são misturados e granulados com uma solução aquosa de (4). (5) é adicionado ao material granulado seco. A partir desta mistura são prensados comprimidos biplanares, facetados em ambos os lados e com um entalhe em um lado.
Diâmetro dos comprimidos: 9 mm.
Preparação: (1), (2) e (3) são misturados e granulados com uma solução aquosa de (4). (5) é adicionado ao material granulado seco. A partir desta mistura são prensados comprimidos biplanares, facetados em ambos os lados e com um entalhe em um lado.
Diâmetro dos comprimidos: 12 mm.
Composição:Preparação:
(1) é triturado com (3). Esta trituração é adicionada à mistura de (2) e (4) com agitação vigorosa. Esta mistura em pó é acondicionada em cápsulas de gelatina de tamanho 3 em uma máquina de encher cápsulas.
Claims (10)
- Composição farmacêutica, caracterizada pelo fato que compreende o derivado de benzeno glicopiranosil-substituído 1-cloro-4-(β-D-glucopiranos-1-il)-2-[4-((S)-tetra-hidrofuran-3-ilóxi)-benzil]-benzeno em combinação com um inibidor de DPP IV 1-[(4-metil-quinazolin-2-il)metil]-3-metil-7-(2-butin-1 -il)-8-(3-(R)-amino-piperidin-1 -il)-xantina, ou sal farmaceuticamente aceitável do mesmo.
- Composição farmacêutica de acordo com a reivindicação 1, caracterizada pelo fato de que a composição é adequada para uso combinado ou simultâneo ou sequencial de derivado de benzeno glicopiranosil-substituído e de inibidor DPP IV.
- Composição farmacêutica de acordo com a reivindicação 1 ou 2, caracterizada pelo fato de que o derivado de benzeno glicopiranosil-substituído e o inibidor de DPP IV estão presentes em forma de dosagem única.
- Composição farmacêutica de acordo com qualquer uma das reivindicações 1 a 3, caracterizada pelo fato de que o derivado de benzeno glicopiranosil-substituído e o inibidor de DPP IV estão presentes em uma forma de dosagem separada.
- Composição farmacêutica, de acordo com uma das reivindicações 1 a 4, caracterizada pelo fato de que compreende uma quantidade de 5 a 50 mg do derivado de benzeno glicopiranosil-substituído.
- Composição farmacêutica, de acordo com a reivindicação 5, caracterizada pelo fato de que compreende uma quantidade de 5 mg, 10 mg, 15 mg, 20 mg, 25 mg ou 50 mg do derivado de benzeno glicopiranosil-substituído.
- Composição farmacêutica, de acordo com uma das reivindicações 1 a 6, caracterizada pelo fato de que compreende uma quantidade de 0,5 a 10 mg do inibidor de DPP IV.
- Composição farmacêutica, de acordo com a reivindicação 7, caracterizada pelo fato de que compreende uma quantidade de 1 mg, 2,5 mg ou 5 mg do inibidor DPP IV.
- Composição farmacêutica, de acordo com qualquer uma das reivindicações 1 a 4, caracterizada pelo fato de que compreende uma
quantidade de 5 a 50 mg do derivado de benzeno glicopiranosil-substituído e uma quantidade de 0,5 a 10 mg do inibidor de DPP IV. - Composição farmacêutica, de acordo com qualquer uma das reivindicações 1 a 9, caracterizada pelo fato de que a composição farmacêutica é formulada para administração oral na forma sólida.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07114459 | 2007-08-16 | ||
| EP07114459.6 | 2007-08-16 | ||
| PCT/EP2008/060736 WO2009022007A1 (en) | 2007-08-16 | 2008-08-15 | Pharmaceutical composition comprising a glucopyranosyl-substituted benzene derivative |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| BRPI0815331A2 BRPI0815331A2 (pt) | 2020-06-09 |
| BRPI0815331B1 true BRPI0815331B1 (pt) | 2021-04-06 |
| BRPI0815331B8 BRPI0815331B8 (pt) | 2021-05-25 |
Family
ID=39870130
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| BRPI0815331A BRPI0815331B8 (pt) | 2007-08-16 | 2008-08-15 | composição farmacêutica compreendendo um derivado de benzeno glicopiranosil-substituído, bem como seu uso |
Country Status (39)
| Country | Link |
|---|---|
| US (1) | US8551957B2 (pt) |
| EP (4) | EP2187879B1 (pt) |
| JP (1) | JP5595914B2 (pt) |
| KR (1) | KR101491554B1 (pt) |
| CN (3) | CN104288166A (pt) |
| AR (2) | AR067970A1 (pt) |
| AU (1) | AU2008288407B2 (pt) |
| BR (1) | BRPI0815331B8 (pt) |
| CA (1) | CA2696558C (pt) |
| CL (1) | CL2008002427A1 (pt) |
| CO (1) | CO6251239A2 (pt) |
| CY (2) | CY1118308T1 (pt) |
| DK (1) | DK2187879T3 (pt) |
| EA (1) | EA018608B1 (pt) |
| EC (1) | ECSP109977A (pt) |
| ES (1) | ES2602748T3 (pt) |
| HK (2) | HK1201721A1 (pt) |
| HR (1) | HRP20170022T1 (pt) |
| HU (2) | HUE030158T2 (pt) |
| IL (1) | IL202886A (pt) |
| LT (2) | LT2187879T (pt) |
| MA (1) | MA31612B1 (pt) |
| ME (1) | ME02573B (pt) |
| MX (1) | MX2010001696A (pt) |
| MY (1) | MY152037A (pt) |
| NL (1) | NL300872I2 (pt) |
| NO (1) | NO2017020I2 (pt) |
| NZ (1) | NZ583242A (pt) |
| PE (1) | PE20090938A1 (pt) |
| PL (1) | PL2187879T3 (pt) |
| PT (1) | PT2187879T (pt) |
| RS (1) | RS55205B1 (pt) |
| SI (1) | SI2187879T1 (pt) |
| TN (1) | TN2010000073A1 (pt) |
| TW (2) | TW201436798A (pt) |
| UA (1) | UA100384C2 (pt) |
| UY (1) | UY31296A1 (pt) |
| WO (1) | WO2009022007A1 (pt) |
| ZA (1) | ZA200909105B (pt) |
Families Citing this family (79)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7407955B2 (en) | 2002-08-21 | 2008-08-05 | Boehringer Ingelheim Pharma Gmbh & Co., Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| US7501426B2 (en) | 2004-02-18 | 2009-03-10 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, their preparation and their use as pharmaceutical compositions |
| CA2557801C (en) * | 2004-03-16 | 2013-06-25 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzol derivatives, drugs containing said compounds, the use thereof and method for the production thereof |
| DE102004054054A1 (de) | 2004-11-05 | 2006-05-11 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Verfahren zur Herstellung chiraler 8-(3-Amino-piperidin-1-yl)-xanthine |
| US7772191B2 (en) | 2005-05-10 | 2010-08-10 | Boehringer Ingelheim International Gmbh | Processes for preparing of glucopyranosyl-substituted benzyl-benzene derivatives and intermediates therein |
| DE102005035891A1 (de) | 2005-07-30 | 2007-02-08 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-(3-Amino-piperidin-1-yl)-xanthine, deren Herstellung und deren Verwendung als Arzneimittel |
| TW200745075A (en) * | 2005-09-08 | 2007-12-16 | Boehringer Ingelheim Int | Crystalline forms of 1-chloro-4-(β-D-glucopyranos-1-yl)-2-(4-ethynyl-benzyl)-benzene, methods for its preparation and the use thereof for preparing medicaments |
| PE20080697A1 (es) | 2006-05-03 | 2008-08-05 | Boehringer Ingelheim Int | Derivados de benzonitrilo sustituidos con glucopiranosilo, composiciones farmaceuticas que contienen compuestos de este tipo, su uso y procedimiento para su fabricacion |
| PE20080251A1 (es) | 2006-05-04 | 2008-04-25 | Boehringer Ingelheim Int | Usos de inhibidores de dpp iv |
| EP1852108A1 (en) | 2006-05-04 | 2007-11-07 | Boehringer Ingelheim Pharma GmbH & Co.KG | DPP IV inhibitor formulations |
| CN109503584A (zh) | 2006-05-04 | 2019-03-22 | 勃林格殷格翰国际有限公司 | 多晶型 |
| AU2007256633B2 (en) | 2006-06-06 | 2013-09-12 | Intra-Cellular Therapies, Inc. | Organic compounds |
| US8283326B2 (en) * | 2006-10-27 | 2012-10-09 | Boehringer Ingelheim International Gmbh | Crystalline form of 4-(beta-D-glucopyranos-1-yl)-1-methyl-2-[4-((S)-tetrahydrofuran-3-yloxy)-benzyl]-benzene, a method for its preparation and the use thereof for preparing medicaments |
| PE20090603A1 (es) * | 2007-08-16 | 2009-06-11 | Boehringer Ingelheim Int | Composicion farmaceutica que comprende un inhibidor de sglt2 y un inhibidor de dpp iv |
| PE20090938A1 (es) | 2007-08-16 | 2009-08-08 | Boehringer Ingelheim Int | Composicion farmaceutica que comprende un derivado de benceno sustituido con glucopiranosilo |
| JP2011504505A (ja) | 2007-11-21 | 2011-02-10 | デコード ジェネティクス イーエイチエフ | 肺および心血管障害を治療するためのビアリールpde4抑制剤 |
| EP2240028B1 (en) | 2007-12-06 | 2016-07-20 | Intra-Cellular Therapies, Inc. | Pyrazolopyrimidine-4,6-dione derivatives and their use as pharmaceutical |
| PE20091730A1 (es) | 2008-04-03 | 2009-12-10 | Boehringer Ingelheim Int | Formulaciones que comprenden un inhibidor de dpp4 |
| PE20100156A1 (es) * | 2008-06-03 | 2010-02-23 | Boehringer Ingelheim Int | Tratamiento de nafld |
| BRPI0916769A2 (pt) | 2008-07-15 | 2017-09-26 | Theracos Inc | derivados de benzilbenzeno deuterados e métodos de uso |
| UY32030A (es) | 2008-08-06 | 2010-03-26 | Boehringer Ingelheim Int | "tratamiento para diabetes en pacientes inapropiados para terapia con metformina" |
| BRPI0916997A2 (pt) | 2008-08-06 | 2020-12-15 | Boehringer Ingelheim International Gmbh | Inibidor de dpp-4 e seu uso |
| MX2011002558A (es) | 2008-09-10 | 2011-04-26 | Boehringer Ingelheim Int | Terapia de combinacion para el tratamiento de diabetes y estados relacionados. |
| US20200155558A1 (en) | 2018-11-20 | 2020-05-21 | Boehringer Ingelheim International Gmbh | Treatment for diabetes in patients with insufficient glycemic control despite therapy with an oral antidiabetic drug |
| UY32177A (es) * | 2008-10-16 | 2010-05-31 | Boehringer Ingelheim Int | Tratamiento de diabetes en pacientes con control glucémico insuficiente a pesar de la terapia con fármaco, oral o no, antidiabético |
| JP5710492B2 (ja) | 2008-12-06 | 2015-04-30 | イントラ−セルラー・セラピーズ・インコーポレイテッドIntra−Cellular Therapies, Inc. | 有機化合物 |
| MX2011005934A (es) | 2008-12-06 | 2011-12-16 | Intra Cellular Therapies Inc | Compuestos organicos. |
| JP5710493B2 (ja) | 2008-12-06 | 2015-04-30 | イントラ−セルラー・セラピーズ・インコーポレイテッドIntra−Cellular Therapies, Inc. | 有機化合物 |
| EA022310B1 (ru) | 2008-12-23 | 2015-12-30 | Бёрингер Ингельхайм Интернациональ Гмбх | Солевые формы органического соединения |
| AR074990A1 (es) | 2009-01-07 | 2011-03-02 | Boehringer Ingelheim Int | Tratamiento de diabetes en pacientes con un control glucemico inadecuado a pesar de la terapia con metformina |
| AR075204A1 (es) * | 2009-01-29 | 2011-03-16 | Boehringer Ingelheim Int | Inhibidores de dpp-4 y composiciones farmaceuticas que los comprenden, utiles para tratar enfermedades metabolicas en pacientes pediatricos, particularmente diabetes mellitus tipo 2 |
| AU2016213789B2 (en) * | 2009-02-13 | 2018-01-18 | Boehringer Ingelheim International Gmbh | SGLT-2 inhibitor for treating type 1 diabetes mellitus, type 2 diabetes mellitus, impaired glucose tolerance or hyperglycemia |
| UY32427A (es) * | 2009-02-13 | 2010-09-30 | Boheringer Ingelheim Internat Gmbh | Composicion farmaceutica, forma farmaceutica, procedimiento para su preparacion, metodos de tratamiento y usos de la misma |
| AP2011005795A0 (en) * | 2009-02-13 | 2011-08-31 | Boehringer Ingelheim Int | SGLT-2 inhibitor for treating type 1 diabetes mellitus, type 2 diabetes mellitus, impaired glucose tolerance or hyperglycemia. |
| PL2395983T3 (pl) | 2009-02-13 | 2020-09-07 | Boehringer Ingelheim International Gmbh | Kompozycja farmaceutyczna zawierająca inhibitor sglt2, inhibitor dpp-iv i ewentualnie dalszy środek przeciwcukrzycowy oraz jej zastosowania |
| JP5600328B2 (ja) * | 2009-02-13 | 2014-10-01 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | グルコピラノシルジフェニルメタン誘導体を含む医薬組成物、その医薬剤形、それらの調製方法及び患者の血糖コントロールを改善するためのそれらの使用 |
| SG175154A1 (en) | 2009-04-16 | 2011-11-28 | Taisho Pharmaceutical Co Ltd | Pharmaceutical compositions |
| SI2486029T1 (sl) | 2009-09-30 | 2015-10-30 | Boehringer Ingelheim International Gmbh | Postopki za pripravo z glukopiranozilom substituiranih benzil-benzenskih derivatov |
| EA020798B1 (ru) | 2009-09-30 | 2015-01-30 | Бёрингер Ингельхайм Интернациональ Гмбх | СПОСОБ ПОЛУЧЕНИЯ КРИСТАЛЛИЧЕСКОЙ ФОРМЫ 1-ХЛОР-4-(β-D-ГЛЮКОПИРАНОЗ-1-ИЛ)-2-[4-((S)-ТЕТРАГИДРОФУРАН-3-ИЛОКСИ)БЕНЗИЛ]БЕНЗОЛА |
| UY32919A (es) | 2009-10-02 | 2011-04-29 | Boehringer Ingelheim Int | Composición farmacéutica, forma de dosificación farmacéutica, procedimiento para su preparación, mé todos para su tratamiento y sus usos |
| MX364651B (es) | 2009-11-27 | 2019-05-03 | Boehringer Ingelheim Int Gmbh Star | Inhibidores de dpp-iv, tales como la linagliptina, y composiciones farmacéuticas o combinaciones que comprenden los mismos, para usarse en el tratamiento de pacientes diabéticos tipificados genéticamente. |
| EP2368552A1 (en) | 2010-03-25 | 2011-09-28 | Boehringer Ingelheim Vetmedica GmbH | 1-[(3-cyano-pyridin-2-yl)methyl]-3-methyl-7-(2-butyn-1-yl)-8-[3-(r)-amino-piperidin-1-yl]-xanthine for the treatment of a metabolic disorder of a predominantly carnivorous non-human animal |
| EP2566469B1 (en) | 2010-05-05 | 2022-12-21 | Boehringer Ingelheim International GmbH | Combination therapy |
| WO2011153135A1 (en) | 2010-05-31 | 2011-12-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
| WO2011153136A1 (en) | 2010-05-31 | 2011-12-08 | Intra-Cellular Therapies, Inc. | Organic compounds |
| EA201991014A1 (ru) | 2010-06-24 | 2019-09-30 | Бёрингер Ингельхайм Интернациональ Гмбх | Лечение диабета |
| AR083878A1 (es) | 2010-11-15 | 2013-03-27 | Boehringer Ingelheim Int | Terapia antidiabetica vasoprotectora y cardioprotectora, linagliptina, metodo de tratamiento |
| UY33937A (es) | 2011-03-07 | 2012-09-28 | Boehringer Ingelheim Int | Composiciones farmacéuticas que contienen inhibidores de dpp-4 y/o sglt-2 y metformina |
| AU2012252380B2 (en) * | 2011-05-10 | 2016-09-08 | Sandoz Ag | Polymorph of Linagliptin benzoate |
| US20130035298A1 (en) | 2011-07-08 | 2013-02-07 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| US8883800B2 (en) | 2011-07-15 | 2014-11-11 | Boehringer Ingelheim International Gmbh | Substituted quinazolines, the preparation thereof and the use thereof in pharmaceutical compositions |
| WO2013098775A1 (en) * | 2011-12-28 | 2013-07-04 | Dr. Reddy's Laboratories Limited | Improved process for preparation of pure linagliptin |
| US9555001B2 (en) | 2012-03-07 | 2017-01-31 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition and uses thereof |
| US9192617B2 (en) | 2012-03-20 | 2015-11-24 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| WO2013171167A1 (en) | 2012-05-14 | 2013-11-21 | Boehringer Ingelheim International Gmbh | A xanthine derivative as dpp -4 inhibitor for use in the treatment of podocytes related disorders and/or nephrotic syndrome |
| JP6374862B2 (ja) * | 2012-05-24 | 2018-08-15 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | 自己免疫性糖尿病、特に、ladaの治療に使用するためのdpp−4阻害剤としてのキサンチン誘導体 |
| WO2013174767A1 (en) | 2012-05-24 | 2013-11-28 | Boehringer Ingelheim International Gmbh | A xanthine derivative as dpp -4 inhibitor for use in modifying food intake and regulating food preference |
| PL2981269T3 (pl) | 2013-04-04 | 2024-02-05 | Boehringer Ingelheim Vetmedica Gmbh | Leczenie zaburzeń metabolicznych u zwierząt koniowatych |
| US20160000816A1 (en) * | 2013-04-05 | 2016-01-07 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| US20140303098A1 (en) * | 2013-04-05 | 2014-10-09 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| CA2812519A1 (en) * | 2013-04-05 | 2014-10-05 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| US11813275B2 (en) | 2013-04-05 | 2023-11-14 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| US20140303097A1 (en) * | 2013-04-05 | 2014-10-09 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| CA2812016A1 (en) * | 2013-04-05 | 2014-10-05 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| CN105377266A (zh) * | 2013-04-18 | 2016-03-02 | 勃林格殷格翰国际有限公司 | 药物组合物、治疗方法及其用途 |
| ES2969764T3 (es) | 2013-12-17 | 2024-05-22 | Boehringer Ingelheim Vetmedica Gmbh | Un inhibidor de SGLT-2 para usar en el tratamiento de un trastorno metabólico en animales felinos |
| US9902751B2 (en) | 2013-12-30 | 2018-02-27 | Mylan Laboratories Limited | Process for the preparation of empagliflozin |
| MX2016009421A (es) * | 2014-01-23 | 2016-09-16 | Boehringer Ingelheim Vetmedica Gmbh | Tratamiento de trastornos metabolicos en animales caninos. |
| WO2015128453A1 (en) | 2014-02-28 | 2015-09-03 | Boehringer Ingelheim International Gmbh | Medical use of a dpp-4 inhibitor |
| DK3125882T3 (da) | 2014-04-01 | 2020-06-29 | Boehringer Ingelheim Vetmedica Gmbh | Behandling af metabolske lidelser i hestedyr |
| CN103965020B (zh) * | 2014-05-06 | 2015-09-09 | 启东东岳药业有限公司 | 制备5-碘-2-溴苄醇的方法 |
| US9546175B2 (en) | 2014-08-07 | 2017-01-17 | Intra-Cellular Therapies, Inc. | Organic compounds |
| BR112017003570A2 (pt) | 2014-09-25 | 2017-12-05 | Boehringer Ingelheim Vetmedica Gmbh | tratamento de combinação de inibidores de sglt2 e agonistas de dopamina para prevenir distúrbios metabólicos em animais equinos |
| BR112018003749B1 (pt) | 2015-08-27 | 2023-10-31 | Boehringer Ingelheim Vetmedica Gmbh | Composições farmacêuticas líquidas compreendendo inibidores sglt-2, kit de peças e processo para a produção da composição farmacêutica líquida |
| KR20230028565A (ko) * | 2016-03-16 | 2023-02-28 | 베링거 인겔하임 인터내셔날 게엠베하 | 엠파글리플로진을 포함하는 약제학적 조성물 및 이의 용도 |
| WO2017211979A1 (en) | 2016-06-10 | 2017-12-14 | Boehringer Ingelheim International Gmbh | Combinations of linagliptin and metformin |
| WO2020041531A2 (en) * | 2018-08-21 | 2020-02-27 | Kaleido Biosciences, Inc. | Oligosaccharide compositions and methods of use thereof for reducing ammonia levels |
| EP3852730A1 (en) | 2018-09-19 | 2021-07-28 | Galenicum Health S.L.U. | Pharmaceutical compositions of empagliflozin |
| WO2021214023A1 (en) | 2020-04-22 | 2021-10-28 | Bayer Aktiengesellschaft | Combination of finerenone and a sglt2 inhibitor for the treatment and/or prevention of cardiovascular and/or renal diseases |
Family Cites Families (236)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3174901A (en) | 1963-01-31 | 1965-03-23 | Jan Marcel Didier Aron Samuel | Process for the oral treatment of diabetes |
| NL167151C (nl) | 1971-04-09 | 1981-11-16 | Acf Chemiefarma Nv | Werkwijze ter bereiding van geneesmiddelen met anti-parasitaire werking op basis van halogeen bevatten- de 2,2'-methyleendifenolderivaten, alsmede werkwijze ter bereiding van deze geneeskrachtige verbindingen. |
| NO154918C (no) | 1977-08-27 | 1987-01-14 | Bayer Ag | Analogifremgangsmaate til fremstilling av terapeutisk aktive derivater av 3,4,5-trihydroksypiperidin. |
| DE2758025A1 (de) | 1977-12-24 | 1979-07-12 | Bayer Ag | Neue derivate von 3,4,5-trihydroxypiperidin, verfahren zu ihrer herstellung und ihre verwendung |
| DE2951135A1 (de) | 1979-12-19 | 1981-06-25 | Hoechst Ag, 6230 Frankfurt | Sulfonylharnstoffe, verfahren zu ihrer herstellung, pharmazeutische praeparate auf basis dieser verbindungen und ihre verwendung |
| US4786755A (en) | 1985-06-03 | 1988-11-22 | Warner-Lambert Company | Diphenic acid monoamides |
| US4602023A (en) | 1985-06-03 | 1986-07-22 | Warner-Lambert Company | Diphenic acid monoamides |
| US4786023A (en) | 1987-08-19 | 1988-11-22 | Harris Leverett D | Drafting implement holder |
| DE122010000020I1 (de) | 1996-04-25 | 2010-07-08 | Prosidion Ltd | Verfahren zur Senkung des Blutglukosespiegels in Säugern |
| WO1998031697A1 (en) | 1997-01-15 | 1998-07-23 | Sankyo Company, Limited | Aryl c-glycoside compounds and sulfated esters thereof |
| ES2288807T1 (es) | 1998-02-02 | 2008-02-01 | Trustees Of Tufts College | Uso de inhibidores de dipeptidasa para regular el metabolismo de la glucosa. |
| CO5150173A1 (es) | 1998-12-10 | 2002-04-29 | Novartis Ag | Compuestos n-(glicilo sustituido)-2-cianopirrolidinas inhibidores de peptidasa de dipeptidilo-iv (dpp-iv) los cuales son efectivos en el tratamiento de condiciones mediadas por la inhibicion de dpp-iv |
| US6613806B1 (en) | 1999-01-29 | 2003-09-02 | Basf Corporation | Enhancement of the efficacy of benzoylbenzenes |
| DE60009929T2 (de) | 1999-08-31 | 2005-03-31 | Kissei Pharmaceutical Co., Ltd., Matsumoto | Glucopyranosyloxypyrazol-derivate, diese enthaltende arzneimittel und zwischenprodukte zu deren herstellung |
| PH12000002657B1 (en) | 1999-10-12 | 2006-02-21 | Bristol Myers Squibb Co | C-aryl glucoside SGLT2 inhibitors |
| US6515117B2 (en) * | 1999-10-12 | 2003-02-04 | Bristol-Myers Squibb Company | C-aryl glucoside SGLT2 inhibitors and method |
| PT1248604E (pt) | 2000-01-21 | 2007-01-31 | Novartis Ag | Associações compreendendo inibidor de dipeptidilpeptidase-iv |
| JP4456768B2 (ja) | 2000-02-02 | 2010-04-28 | 壽製薬株式会社 | C−配糖体を含有する薬剤 |
| US6627611B2 (en) | 2000-02-02 | 2003-09-30 | Kotobuki Pharmaceutical Co Ltd | C-glycosides and preparation of thereof as antidiabetic agents |
| US6395767B2 (en) | 2000-03-10 | 2002-05-28 | Bristol-Myers Squibb Company | Cyclopropyl-fused pyrrolidine-based inhibitors of dipeptidyl peptidase IV and method |
| US6683056B2 (en) | 2000-03-30 | 2004-01-27 | Bristol-Myers Squibb Company | O-aryl glucoside SGLT2 inhibitors and method |
| GB0014969D0 (en) | 2000-06-19 | 2000-08-09 | Smithkline Beecham Plc | Novel method of treatment |
| AU2001268958B2 (en) | 2000-07-04 | 2006-03-09 | Novo Nordisk A/S | Heterocyclic compounds, which are inhibitors of the enzyme dpp-iv |
| ES2269456T3 (es) | 2000-09-29 | 2007-04-01 | Kissei Pharmaceutical Co., Ltd. | Derivados de glucopiranosiloxibencilbenceno y composiciones medicinales que contienen los mismos. |
| AU2002223127A1 (en) | 2000-11-30 | 2002-06-11 | Kissei Pharmaceutical Co., Ltd. Intellectual Property | Glucopyranosyloxybenzyl benzene derivatives, medicinal compositions containing the same and intermediates in the production thereof |
| WO2002051836A1 (fr) | 2000-12-27 | 2002-07-04 | Kyowa Hakko Kogyo Co., Ltd. | Inhibiteur de dipeptidyl peptidase iv |
| ES2326158T3 (es) | 2000-12-28 | 2009-10-02 | Kissei Pharmaceutical Co., Ltd. | Derivados de glucopiranosiloxipirazol y su utilizacion como medicamentos. |
| TWI255817B (en) | 2001-02-14 | 2006-06-01 | Kissei Pharmaceutical | Glucopyranosyloxybenzylbenzene derivatives and medicinal use thereof |
| SK288003B6 (sk) | 2001-02-24 | 2012-10-02 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthin derivatives, method for the preparation thereof, pharmaceutical composition containing thereof and their use |
| US6936590B2 (en) * | 2001-03-13 | 2005-08-30 | Bristol Myers Squibb Company | C-aryl glucoside SGLT2 inhibitors and method |
| JP3698067B2 (ja) | 2001-03-30 | 2005-09-21 | Jsr株式会社 | 電子吸引性基および電子供与性基を有するモノマー、それを用いた共重合体、ならびにプロトン伝導膜 |
| WO2002083857A2 (en) | 2001-04-10 | 2002-10-24 | Millennium Pharmaceuticals, Inc. | 68723, sodium/glucose cotransporter family members and uses therefor |
| HUP0600232A2 (en) | 2001-04-11 | 2006-08-28 | Bristol Myers Squibb Co | Amino acid complexes of c-aryl glucosides for treatment of diabetes and method |
| EP1406873B1 (en) | 2001-06-27 | 2007-09-26 | Smithkline Beecham Corporation | Fluoropyrrolidines as dipeptidyl peptidase inhibitors |
| WO2003004496A1 (en) | 2001-07-03 | 2003-01-16 | Novo Nordisk A/S | Dpp-iv-inhibiting purine derivatives for the treatment of diabetes |
| US6869947B2 (en) | 2001-07-03 | 2005-03-22 | Novo Nordisk A/S | Heterocyclic compounds that are inhibitors of the enzyme DPP-IV |
| UA74912C2 (en) | 2001-07-06 | 2006-02-15 | Merck & Co Inc | Beta-aminotetrahydroimidazo-(1,2-a)-pyrazines and tetratriazolo-(4,3-a)-pyrazines as inhibitors of dipeptylpeptidase for the treatment or prevention of diabetes |
| EP1432720A1 (en) | 2001-09-05 | 2004-06-30 | Bristol-Myers Squibb Company | O-pyrazole glucoside sglt2 inhibitors and method of use |
| EP1463727A2 (en) | 2001-09-19 | 2004-10-06 | Novo Nordisk A/S | Heterocyclic compounds that are inhibitors of the enzyme dpp-iv |
| WO2003031458A1 (en) | 2001-10-12 | 2003-04-17 | Dana-Farber Cancer Institute | Methods for synthesis of diarylmethanes |
| ATE407678T1 (de) | 2001-10-17 | 2008-09-15 | Boehringer Ingelheim Pharma | Pyrimidinderivate, arzneimittel enthaltend diese verbindungen, deren verwendung und verfahren zu ihrer herstellung |
| US6861440B2 (en) | 2001-10-26 | 2005-03-01 | Hoffmann-La Roche Inc. | DPP IV inhibitors |
| DE60323823D1 (de) | 2002-01-11 | 2008-11-13 | Novo Nordisk As | Verfahren und zusammensetzung zur behandlung von diabetes, hypertonie, chronischer herzinsuffizienz und mit flüssigkeitsretention einhergehenden zuständen |
| GB0206215D0 (en) | 2002-03-15 | 2002-05-01 | Novartis Ag | Organic compounds |
| JP4424203B2 (ja) | 2002-04-26 | 2010-03-03 | 味の素株式会社 | 糖尿病予防・治療剤 |
| DE10231370B4 (de) | 2002-07-11 | 2006-04-06 | Sanofi-Aventis Deutschland Gmbh | Thiophenglycosidderivate, diese Verbindungen enthaltende Arzneimittel und Verfahren zur Herstellung dieser Arzneimittel |
| TWI254635B (en) | 2002-08-05 | 2006-05-11 | Yamanouchi Pharma Co Ltd | Azulene derivative and salt thereof |
| US7375087B2 (en) | 2002-08-08 | 2008-05-20 | Kissei Pharmaceutical Co., Ltd. | Pyrazole derivative, medicinal composition containing the same, medicinal use thereof, and intermediate for production thereof |
| US7407955B2 (en) | 2002-08-21 | 2008-08-05 | Boehringer Ingelheim Pharma Gmbh & Co., Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the preparation thereof and their use as pharmaceutical compositions |
| CA2496249C (en) | 2002-08-21 | 2012-01-24 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-amino-piperidin-1-yl]-xanthines, the production thereof and the use of the same as medicaments |
| DE10238477A1 (de) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue Purinderivate, deren Herstellung und deren Verwendung als Arzneimittel |
| DE10238470A1 (de) | 2002-08-22 | 2004-03-04 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue Xanthinderivate, deren Herstellung und deren Verwendung als Arzneimittel |
| US7482337B2 (en) | 2002-11-08 | 2009-01-27 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Xanthine derivatives, the preparation thereof and their use as pharmaceutical compositions |
| DE10251927A1 (de) | 2002-11-08 | 2004-05-19 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue Xanthinderivate, deren Herstellung und deren Verwendung als Arzneimittel |
| JP3567162B1 (ja) | 2002-11-20 | 2004-09-22 | 日本たばこ産業株式会社 | 4−オキソキノリン化合物及びそのhivインテグラーゼ阻害剤としての利用 |
| TR200401663T1 (tr) | 2002-11-20 | 2005-04-21 | Japan Tobacco Inc. | 4-oksokuinolin bileşiği ve bunun HIV integraz inhibitörü olarak kullanımı. |
| DE10254304A1 (de) | 2002-11-21 | 2004-06-03 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue Xanthinderivate, deren Herstellung und deren Verwendung als Arzneimittel |
| UY28103A1 (es) * | 2002-12-03 | 2004-06-30 | Boehringer Ingelheim Pharma | Nuevas imidazo-piridinonas sustituidas, su preparación y su empleo como medicacmentos |
| US7109192B2 (en) * | 2002-12-03 | 2006-09-19 | Boehringer Ingelheim Pharma Gmbh & Co Kg | Substituted imidazo-pyridinones and imidazo-pyridazinones, the preparation thereof and their use as pharmaceutical compositions |
| JP4651934B2 (ja) | 2002-12-04 | 2011-03-16 | キッセイ薬品工業株式会社 | ベンジルフェノール誘導体、それを含有する医薬組成物およびその医薬用途 |
| US7420079B2 (en) | 2002-12-09 | 2008-09-02 | Bristol-Myers Squibb Company | Methods and compounds for producing dipeptidyl peptidase IV inhibitors and intermediates thereof |
| DE10258007B4 (de) | 2002-12-12 | 2006-02-09 | Sanofi-Aventis Deutschland Gmbh | Aromatische Fluorglycosidderivate, diese Verbindungen enthaltende Arzneimittel und Verfahren zur Herstellung dieser Arzneimittel |
| DE10258008B4 (de) | 2002-12-12 | 2006-02-02 | Sanofi-Aventis Deutschland Gmbh | Heterocyclische Fluorglycosidderivate, diese Verbindungen enthaltende Arzneimittel und Verfahren zur Herstellung dieser Arzneimittel |
| MXPA05007052A (es) | 2003-01-03 | 2005-08-18 | Squibb Bristol Myers Co | Metodos para producir c-aril-glucosidos como inhibidores de sglt2. |
| BRPI0406761A (pt) | 2003-01-14 | 2005-12-20 | Arena Pharm Inc | Derivados de arila e heteroarila 1,2,3-trissubstituìdos como moduladores do metabolismo e a profilaxia e tratamento de distúrbios relacionados a estes tais como diabetes e hiper-glicemia |
| WO2004076470A2 (en) | 2003-02-27 | 2004-09-10 | Bristol-Myers Squibb Company | A non-cryogenic process for forming glycosides |
| TWI310033B (en) | 2003-03-14 | 2009-05-21 | Yamanouchi Pharma Co Ltd | C-glycoside derivatives or salts thereof |
| US7674486B2 (en) | 2003-05-14 | 2010-03-09 | Indus Biotech Pvt. Ltd. | Synergistic composition for the treatment of diabetes mellitus |
| WO2005000860A2 (en) | 2003-06-03 | 2005-01-06 | The Regents Of The University Of California | Compositions and methods for treatment of disease with acetylated disaccharides |
| JP2004359630A (ja) | 2003-06-06 | 2004-12-24 | Yamanouchi Pharmaceut Co Ltd | ジフルオロジフェニルメタン誘導体及びその塩 |
| DE10327439A1 (de) | 2003-06-18 | 2005-01-05 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue Imidazopyridazinon- und Imidazopyridonderivate, deren Herstellung und deren Verwendung als Arzneimittel |
| DE602004030244D1 (de) | 2003-06-20 | 2011-01-05 | Hoffmann La Roche | Itoren |
| JO2625B1 (en) | 2003-06-24 | 2011-11-01 | ميرك شارب اند دوم كوربوريشن | Phosphoric acid salts of dipeptidyl betidase inhibitor 4 |
| KR20060037391A (ko) | 2003-07-21 | 2006-05-03 | 스미스클라인 비참 코포레이션 | (2S,4S)-4-플루오로-1-[4-플루오로-베타-(4-플루오로페닐)-L-페닐알라닐]-2-피롤리딘카르보니트릴 p-톨루엔설폰산염 및 이의 무수 결정 형태 |
| CA2549015A1 (en) | 2003-08-01 | 2005-02-10 | Janssen Pharmaceutica N.V. | Substituted fused heterocyclic c-glycosides |
| PL2896397T5 (pl) | 2003-08-01 | 2023-06-19 | Mitsubishi Tanabe Pharma Corporation | Nowe związki mające działanie inhibitujące zależny od sodu transporter glukozy |
| JP4131216B2 (ja) | 2003-08-20 | 2008-08-13 | Jsr株式会社 | ポリアリーレンおよびその製造方法、ならびに高分子固体電解質およびプロトン伝導膜 |
| US7375090B2 (en) | 2003-08-26 | 2008-05-20 | Boehringer Ingelheim International Gmbh | Glucopyranosyloxy-pyrazoles, pharmaceutical compositions containing these compounds, the use thereof and processed for the preparation thereof |
| ATE422204T1 (de) | 2003-08-26 | 2009-02-15 | Boehringer Ingelheim Int | Glucopyranosyloxy-pyrazole, diese verbindungen enthaltende arzneimittel, deren verwendung und verfahren zu ihrer herstellung |
| US20050085680A1 (en) | 2003-10-21 | 2005-04-21 | Joseph Auerbach | Method for industrial decontamination |
| US7526806B2 (en) * | 2003-11-05 | 2009-04-28 | Cisco Technology, Inc. | Method and system for addressing intrusion attacks on a computer system |
| EP2839832A3 (en) * | 2003-11-17 | 2015-06-24 | Novartis AG | Use of dipeptidyl peptidase IV inhibitors |
| DE10355304A1 (de) | 2003-11-27 | 2005-06-23 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue 8-(Piperazin-1-yl)-und 8-([1,4]Diazepan-1-yl)-xanthine, deren Herstellung und deren Verwendung als Arzneimittel |
| DE10359098A1 (de) | 2003-12-17 | 2005-07-28 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue 2-(Piperazin-1-yl)- und 2-([1,4]Diazepan-1-yl)-imidazo[4,5-d]pyridazin-4-one, deren Herstellung und deren Verwendung als Arzneimittel |
| DE10361133A1 (de) | 2003-12-22 | 2005-07-21 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Glucopyranosyloxy-substituierte Aromaten, diese Verbindungen enthaltende Arzneimittel, deren Verwendung und Verfahren zu ihrer Herstellung |
| US7371732B2 (en) | 2003-12-22 | 2008-05-13 | Boehringer Ingelheim International Gmbh | Glucopyranosyloxy-substituted aromatic compounds, medicaments containing such compounds, their use and process for their manufacture |
| DE10360835A1 (de) | 2003-12-23 | 2005-07-21 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Bicyclische Imidazolverbindungen, deren Herstellung und deren Verwendung als Arzneimittel |
| DK3023095T3 (en) | 2004-01-20 | 2018-08-20 | Novartis Ag | Directly pressed wording and approach |
| EA010854B1 (ru) | 2004-02-18 | 2008-12-30 | Бёрингер Ингельхайм Интернациональ Гмбх | 8-[3-аминопиперидин-1-ил]ксантины, их получение и их применение в качестве ингибиторов dpp-iv |
| DE102004008112A1 (de) * | 2004-02-18 | 2005-09-01 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-Amino-piperidin-1-yl]-xanthine, deren Herstellung und Verwendung als Arzneimittel |
| US7501426B2 (en) | 2004-02-18 | 2009-03-10 | Boehringer Ingelheim International Gmbh | 8-[3-amino-piperidin-1-yl]-xanthines, their preparation and their use as pharmaceutical compositions |
| DE102004009039A1 (de) | 2004-02-23 | 2005-09-08 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-[3-Amino-piperidin-1-yl]-xanthine, deren Herstellung und Verwendung als Arzneimittel |
| NZ549629A (en) | 2004-03-04 | 2010-06-25 | Kissei Pharmaceutical | Fused heterocycle derivative, medicinal composition containing the same, and medicinal use thereof |
| ZA200606792B (en) | 2004-03-04 | 2007-12-27 | Kissei Pharmaceutical | Fused heterocycle derivative, medicinal composition containing the same, and medicinal use thereof |
| CN102127053A (zh) * | 2004-03-15 | 2011-07-20 | 武田药品工业株式会社 | 二肽基肽酶抑制剂 |
| CA2557801C (en) | 2004-03-16 | 2013-06-25 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzol derivatives, drugs containing said compounds, the use thereof and method for the production thereof |
| WO2005097798A1 (de) | 2004-04-10 | 2005-10-20 | Boehringer Ingelheim International Gmbh | Neue 2-amino-imidazo[4,5-d]pyridazin-4-one und 2-amino-imidazo[4,5-c]pyridin-4-one, deren herstellung und deren verwendung als arzneimittel |
| US7741082B2 (en) | 2004-04-14 | 2010-06-22 | Bristol-Myers Squibb Company | Process for preparing dipeptidyl peptidase IV inhibitors and intermediates therefor |
| DE102004022970A1 (de) | 2004-05-10 | 2005-12-01 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue Imidazolderivate, deren Herstellung und deren Verwendung als Intermediate zur Herstellung von Arzneimitteln und Pestiziden |
| ATE437870T1 (de) | 2004-05-12 | 2009-08-15 | Pfizer Prod Inc | Prolinderivate und deren verwendung als dipeptidylpeptidase-iv-inhibitoren |
| US7214702B2 (en) | 2004-05-25 | 2007-05-08 | Bristol-Myers Squibb Company | Process for producing a dipeptidyl peptidase IV inhibitor |
| TWI354569B (en) | 2004-05-28 | 2011-12-21 | Bristol Myers Squibb Co | Coated tablet formulation and method |
| WO2005117861A1 (en) | 2004-06-04 | 2005-12-15 | Novartis Ag | Use of organic compounds |
| US7393836B2 (en) | 2004-07-06 | 2008-07-01 | Boehringer Ingelheim International Gmbh | D-xylopyranosyl-substituted phenyl derivatives, medicaments containing such compounds, their use and process for their manufacture |
| EP1783122A4 (en) | 2004-07-08 | 2008-12-10 | Astellas Pharma Inc | PROCESS FOR PREPARING AZULATE DERIVATIVES AND INTERMEDIATE PRODUCTS FOR THEIR SYNTHESIS |
| US20080269311A1 (en) | 2004-07-14 | 2008-10-30 | Edwin Bernard Villhauer | Combination of Dpp-Iv Inhibitors and Compounds Modulating 5-Ht3 and/or 5-Ht4 Receptors |
| DE102004034690A1 (de) | 2004-07-17 | 2006-02-02 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Methyliden-D-xylopyranosyl-und Oxo-D-xylopyranosyl-substituierte Phenyle, diese Verbindungen enthaltende Arzneimittel, deren Verwendung und Verfahren zu ihrer Herstellung |
| TW200606129A (en) | 2004-07-26 | 2006-02-16 | Chugai Pharmaceutical Co Ltd | Novel cyclohexane derivative, its prodrug, its salt and diabetic therapeutic agent containing the same |
| JP2008508213A (ja) | 2004-07-27 | 2008-03-21 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | D−グルコピラノシル−フェニル置換環状体、そのような化合物を含有する医薬品、それらの使用及びその製造方法 |
| WO2006018150A1 (de) | 2004-08-11 | 2006-02-23 | Boehringer Ingelheim International Gmbh | D-xylopyranosyl-phenyl-substituierte cyclen, diese verbindungen enthaltende arzneimittel, deren verwendung und verfahren zu ihrer herstellung |
| DE102004043944A1 (de) | 2004-09-11 | 2006-03-30 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue 8-(3-Amino-piperidin-1-yl)-7-(but-2-inyl)-xanthine, deren Herstellung und deren Verwendung als Arzneimittel |
| DE102004044221A1 (de) | 2004-09-14 | 2006-03-16 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Neue 3-Methyl-7-butinyl-xanthine, deren Herstellung und deren Verwendung als Arzneimittel |
| AR051446A1 (es) | 2004-09-23 | 2007-01-17 | Bristol Myers Squibb Co | Glucosidos de c-arilo como inhibidores selectivos de transportadores de glucosa (sglt2) |
| DE102004048388A1 (de) | 2004-10-01 | 2006-04-06 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | D-Pyranosyl-substituierte Phenyle, diese Verbindungen enthaltende Arzneimittel, deren Verwendung und Verfahren zu ihrer Herstellung |
| JP4959569B2 (ja) * | 2004-10-12 | 2012-06-27 | グレンマーク・ファーマシューティカルズ・エスエー | 新規なジペプチジルペプチダーゼiv阻害剤、それらを含む医薬品組成物、およびそれらを調製するためのプロセス |
| AU2005299808B2 (en) | 2004-10-25 | 2009-08-20 | Novartis Ag | Combination of DPP-IV inhibitor, PPAR antidiabetic and metformin |
| DE102004054054A1 (de) | 2004-11-05 | 2006-05-11 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Verfahren zur Herstellung chiraler 8-(3-Amino-piperidin-1-yl)-xanthine |
| WO2006068163A1 (ja) | 2004-12-24 | 2006-06-29 | Dainippon Sumitomo Pharma Co., Ltd. | 二環性ピロール誘導体 |
| US7687469B2 (en) | 2004-12-16 | 2010-03-30 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzene derivatives, medicaments containing such compounds, their use and process for their manufacture |
| DE102004063099B4 (de) | 2004-12-22 | 2009-02-12 | Sanofi-Aventis Deutschland Gmbh | Verfahren zur Herstellung von Thiophenglycosidderivaten |
| DOP2006000008A (es) | 2005-01-10 | 2006-08-31 | Arena Pharm Inc | Terapia combinada para el tratamiento de la diabetes y afecciones relacionadas y para el tratamiento de afecciones que mejoran mediante un incremento de la concentración sanguínea de glp-1 |
| GT200600008A (es) | 2005-01-18 | 2006-08-09 | Formulacion de compresion directa y proceso | |
| TW200637869A (en) | 2005-01-28 | 2006-11-01 | Chugai Pharmaceutical Co Ltd | The spiroketal derivatives and the use as therapeutical agent for diabetes of the same |
| WO2006089872A1 (en) | 2005-02-23 | 2006-08-31 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted ( (hetero)arylethynyl-benzyd-benzene derivatives and use thereof as sodium-dependent glucose cotransporter 2 (sglt2) inhibitors |
| EP1888571A2 (en) | 2005-03-22 | 2008-02-20 | F. Hoffmann-Roche AG | New salt and polymorphs of a dpp-iv inhibitor |
| WO2006108842A1 (en) | 2005-04-15 | 2006-10-19 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted (heteroaryloxy-benzyl)-benzene derivatives as sglt inhibitors |
| UA91546C2 (uk) | 2005-05-03 | 2010-08-10 | Бьорінгер Інгельхайм Інтернаціональ Гмбх | КРИСТАЛІЧНА ФОРМА 1-ХЛОР-4-(β-D-ГЛЮКОПІРАНОЗ-1-ИЛ)-2-[4-((S)-ТЕТРАГІДРОФУРАН-3-ІЛОКСИ)-БЕНЗИЛ]-БЕНЗОЛУ, СПОСІБ ЇЇ ОДЕРЖАННЯ ТА ЇЇ ЗАСТОСУВАННЯ ПРИ ПРИГОТУВАННІ ЛІКАРСЬКИХ ЗАСОБІВ |
| US7723309B2 (en) | 2005-05-03 | 2010-05-25 | Boehringer Ingelheim International Gmbh | Crystalline forms of 1-chloro-4-(β-D-glucopyranos-1-yl)-2-[4-((R)-tetrahydrofuran-3-yloxy)-benzyl]-benzene, a method for its preparation and the use thereof for preparing medicaments |
| US7772191B2 (en) | 2005-05-10 | 2010-08-10 | Boehringer Ingelheim International Gmbh | Processes for preparing of glucopyranosyl-substituted benzyl-benzene derivatives and intermediates therein |
| GT200600218A (es) | 2005-06-10 | 2007-03-28 | Formulación y proceso de compresión directa | |
| MY152185A (en) | 2005-06-10 | 2014-08-29 | Novartis Ag | Modified release 1-[(3-hydroxy-adamant-1-ylamino)-acetyl]-pyrrolidine-2(s)-carbonitrile formulation |
| WO2007000445A1 (en) | 2005-06-29 | 2007-01-04 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzyl-benzene derivatives, medicaments containing such compounds, their use and process for their manufacture |
| DE602006014411D1 (de) | 2005-07-27 | 2010-07-01 | Boehringer Ingelheim Pharma | Glucopyranosyl-substituierte ((hetero)cycloalyklethynyl-benzyl)-benzenderivative und deren verwendung als inhibitoren des natriumabhängigen glucose-cotransporters (sglt) |
| UY29694A1 (es) | 2005-07-28 | 2007-02-28 | Boehringer Ingelheim Int | Metodos para prevenir y tratar trastornos metabolicos y nuevos derivados de pirazol-o-glucosido |
| DE102005035891A1 (de) | 2005-07-30 | 2007-02-08 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 8-(3-Amino-piperidin-1-yl)-xanthine, deren Herstellung und deren Verwendung als Arzneimittel |
| KR20080031936A (ko) | 2005-08-04 | 2008-04-11 | 노파르티스 아게 | 빌다글립틴의 염 |
| EP1917001A2 (en) | 2005-08-11 | 2008-05-07 | F.Hoffmann-La Roche Ag | Pharmaceutical composition comprising a dpp-iv inhibitor |
| DE602006017566D1 (de) | 2005-08-30 | 2010-11-25 | Boehringer Ingelheim Pharma | Glucopyranosyl-substituierte benzyl-derivate, medikamente mit solchen verbindungen, ihre verwendung und herstellungsverfahren dafür |
| TW200745075A (en) | 2005-09-08 | 2007-12-16 | Boehringer Ingelheim Int | Crystalline forms of 1-chloro-4-(β-D-glucopyranos-1-yl)-2-(4-ethynyl-benzyl)-benzene, methods for its preparation and the use thereof for preparing medicaments |
| ME02005B (me) * | 2005-09-14 | 2012-08-31 | Takeda Pharmaceuticals Co | Inhibitori dipeptidil peptidaze za lečenje dijabetesa |
| ES2445180T5 (es) | 2005-09-14 | 2022-02-01 | Takeda Pharmaceuticals Co | Administración de inhibidores de dipeptidil peptidasa |
| AR056195A1 (es) | 2005-09-15 | 2007-09-26 | Boehringer Ingelheim Int | Procedimientos para preparar derivados de (etinil-bencil)-benceno sustituidos de glucopiranosilo y compuestos intermedios de los mismos |
| CA2622642C (en) | 2005-09-16 | 2013-12-31 | Takeda Pharmaceutical Company Limited | Dipeptidyl peptidase inhibitors |
| TW200745079A (en) | 2005-09-16 | 2007-12-16 | Takeda Pharmaceuticals Co | Polymorphs of benzoate salt of 2-[[6-[(3R)-3-amino-1-piperidinyl]-3,4-dihydro-3-methyl-2,4-dioxo-1(2H)-pyrimidinyl]methyl]-benzonitrile and methods of use therefor |
| US8143217B2 (en) | 2005-09-20 | 2012-03-27 | Novartis Ag | Use of DPP-IV inhibitor to reduce hypoglycemic events |
| JOP20180109A1 (ar) | 2005-09-29 | 2019-01-30 | Novartis Ag | تركيبة جديدة |
| JP2009513633A (ja) | 2005-10-25 | 2009-04-02 | メルク エンド カムパニー インコーポレーテッド | 糖尿病および高血圧を治療するためのジペプチジルペプチダーゼ−4インヒビターと降圧薬との組合せ |
| EP1785745A1 (de) | 2005-11-10 | 2007-05-16 | Omron Electronics Manufacturing of Germany GmbH | Verfahren und Bedieneinheit zum Konfigurieren und Überwachen einer Einrichtung mit funktionaler Sicherheit |
| CA2633167A1 (en) | 2005-12-16 | 2007-07-12 | Merck & Co., Inc. | Pharmaceutical compositions of combinations of dipeptidyl peptidase-4 inhibitors with metformin |
| WO2007071576A1 (en) | 2005-12-21 | 2007-06-28 | F. Hoffmann-La Roche Ag | New salt and polymorph of dpp-iv inhibitor |
| US20090192138A1 (en) | 2005-12-23 | 2009-07-30 | Daniel Kaspar Baeschlin | compounds |
| US20090042863A1 (en) | 2005-12-28 | 2009-02-12 | Takeda Pharmaceutical Company Limited | Therapeutic Agent for Diabetes |
| JP2009522374A (ja) | 2006-01-06 | 2009-06-11 | ノバルティス アクチエンゲゼルシャフト | 糖尿病処置のためのビルダグリプチンの使用 |
| AR059489A1 (es) * | 2006-02-15 | 2008-04-09 | Boehringer Ingelheim Vetmed | Derivados de benzonitrilo sustituidos con glucopiranosilo, composiciones farmaceuticas que contienen compuestos de este tipo, su uso y procedimiento para su fabricacion |
| US20100029941A1 (en) | 2006-03-28 | 2010-02-04 | Takeda Pharmaceutical Company Limited | Preparation of (r)-3-aminopiperidine dihydrochloride |
| PE20071221A1 (es) | 2006-04-11 | 2007-12-14 | Arena Pharm Inc | Agonistas del receptor gpr119 en metodos para aumentar la masa osea y para tratar la osteoporosis y otras afecciones caracterizadas por masa osea baja, y la terapia combinada relacionada a estos agonistas |
| PE20080697A1 (es) | 2006-05-03 | 2008-08-05 | Boehringer Ingelheim Int | Derivados de benzonitrilo sustituidos con glucopiranosilo, composiciones farmaceuticas que contienen compuestos de este tipo, su uso y procedimiento para su fabricacion |
| PE20080251A1 (es) | 2006-05-04 | 2008-04-25 | Boehringer Ingelheim Int | Usos de inhibidores de dpp iv |
| EP1852108A1 (en) | 2006-05-04 | 2007-11-07 | Boehringer Ingelheim Pharma GmbH & Co.KG | DPP IV inhibitor formulations |
| CN109503584A (zh) | 2006-05-04 | 2019-03-22 | 勃林格殷格翰国际有限公司 | 多晶型 |
| KR20070111099A (ko) | 2006-05-16 | 2007-11-21 | 영진약품공업주식회사 | 시타글립틴 염산염의 신규 결정형, 이의 제조 방법과 이를포함하는 약학적 조성물 |
| ATE499090T1 (de) | 2006-06-16 | 2011-03-15 | Lek Pharmaceuticals | Pharmazeutische zusammensetzung mit hydrochlorothiazid und telmisartan |
| WO2007149797A2 (en) | 2006-06-19 | 2007-12-27 | Novartis Ag | Use of organic compounds |
| WO2007148185A2 (en) | 2006-06-21 | 2007-12-27 | Pfizer Products Inc. | Substituted 3 -amino- pyrrolidino-4 -lactams as dpp inhibitors |
| EP2035395A2 (en) | 2006-06-27 | 2009-03-18 | Glenmark Pharmaceuticals S.A. | Novel processes for the preparation of dpp iv inhibitors |
| WO2008017670A1 (en) | 2006-08-08 | 2008-02-14 | Boehringer Ingelheim International Gmbh | Pyrrolo [3, 2 -d] pyrimidines as dpp-iv inhibitors for the treatment of diabetes mellitus |
| US8039441B2 (en) | 2006-08-15 | 2011-10-18 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted cyclopropylbenzene derivatives, pharmaceutical compositions containing such compounds, their use as SGLT inhibitors and process for their manufacture |
| CA2663279C (en) | 2006-09-13 | 2016-05-17 | Takeda Pharmaceutical Company Limited | Use of 2-6-(3-amino-piperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2h-pyrimidin-1-ylmethyl-4-fluoro-benzonitrile for treating diabetes, cancer, autoimmune disorders and hiv infection |
| WO2008031749A1 (en) | 2006-09-15 | 2008-03-20 | F. Hoffmann-La Roche Ag | Process for the preparation of pyrido[2,1-a]isoquinoline derivatives comprising optical resolution of an enamine |
| WO2008031750A2 (en) | 2006-09-15 | 2008-03-20 | F. Hoffmann-La Roche Ag | Process for the preparation of pyrido[2,1-a]isoquinoline derivatives by catalytic asymmetric hydrogenation of an enamine |
| EP2074130A1 (en) | 2006-09-21 | 2009-07-01 | Boehringer Ingelheim International GmbH | Glucopyranosyl-substituted difluorobenzyl-benzene derivatives, medicaments containing such compounds, their use and process for their manufacture |
| US8283326B2 (en) | 2006-10-27 | 2012-10-09 | Boehringer Ingelheim International Gmbh | Crystalline form of 4-(beta-D-glucopyranos-1-yl)-1-methyl-2-[4-((S)-tetrahydrofuran-3-yloxy)-benzyl]-benzene, a method for its preparation and the use thereof for preparing medicaments |
| US7956201B2 (en) | 2006-11-06 | 2011-06-07 | Hoffman-La Roche Inc. | Process for the preparation of (S)-4-fluoromethyl-dihydro-furan-2-one |
| US7879806B2 (en) | 2006-11-06 | 2011-02-01 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzyl-benzonitrile derivates, medicaments containing such compounds, their use and process for their manufacture |
| US20100298243A1 (en) | 2006-11-09 | 2010-11-25 | Boehringer Ingelheim International Gmbh | Combination therapy with sglt-2 inhibitors and their pharmaceutical compositions |
| WO2008062273A2 (en) | 2006-11-20 | 2008-05-29 | Cadila Pharmaceuticals Limited | Solid oral dosage form having antidiabetic drug combination |
| WO2008089892A1 (de) | 2007-01-26 | 2008-07-31 | Sanofi-Aventis | Phenothiazin derivate, verfahren zu ihrer herstellung und ihre verwendung als arzneimittel |
| AR065033A1 (es) | 2007-01-26 | 2009-05-13 | Boehringer Ingelheim Int | Metodos para prevenir y tratar trastornos neurodegenerativos |
| TW200836774A (en) | 2007-02-01 | 2008-09-16 | Takeda Pharmaceutical | Solid preparation |
| GEP20125410B (en) | 2007-02-01 | 2012-02-27 | Takeda Pharmaceutical | Solid preparation comprising alogliptin and pioglitazone |
| WO2008101938A1 (en) | 2007-02-21 | 2008-08-28 | Boehringer Ingelheim International Gmbh | Glucopyranosyl-substituted benzyl-benzonitrile derivatives, medicaments containing such compounds, their use and process for their manufacture |
| US7879807B2 (en) | 2007-02-21 | 2011-02-01 | Boehringer Ingelheim International Gmbh | Tetrasubstituted glucopyranosylated benzene derivatives, medicaments containing such compounds, their use and process for their manufacture |
| ITFI20070042A1 (it) | 2007-02-21 | 2008-08-22 | Laboratori Guidotti Spa | Formulazione farmaceutica e compressa comprendente detta formulazione. |
| CA2681092A1 (en) | 2007-03-15 | 2008-09-18 | Nectid, Inc. | Anti-diabetic combinations comprising a slow release biguanide composition and an immediate release dipeptidyl peptidase iv inhibitor composition |
| TW200904454A (en) | 2007-03-22 | 2009-02-01 | Bristol Myers Squibb Co | Methods for treating obesity employing an SGLT2 inhibitor and compositions thereof |
| PE20090185A1 (es) | 2007-03-22 | 2009-02-28 | Bristol Myers Squibb Co | Formulaciones farmaceuticas que contienen un inhibidor sglt2 |
| JP5616630B2 (ja) | 2007-04-03 | 2014-10-29 | 田辺三菱製薬株式会社 | ジペプチジルペプチダーゼ4阻害化合物と甘味料との併用 |
| PE20090696A1 (es) | 2007-04-20 | 2009-06-20 | Bristol Myers Squibb Co | Formas cristalinas de saxagliptina y procesos para preparar las mismas |
| JP2010528023A (ja) | 2007-05-18 | 2010-08-19 | ブリストル−マイヤーズ スクイブ カンパニー | Sglt2阻害剤の結晶構造およびその製造方法 |
| PE20090938A1 (es) | 2007-08-16 | 2009-08-08 | Boehringer Ingelheim Int | Composicion farmaceutica que comprende un derivado de benceno sustituido con glucopiranosilo |
| PE20090987A1 (es) | 2007-08-16 | 2009-08-14 | Boehringer Ingelheim Int | Composicion farmaceutica que comprende un derivado de pirazol-o-glucosido |
| FR2920023B1 (fr) | 2007-08-16 | 2013-02-08 | Sanofi Aventis | Derives de l'indol-2-one disubstitues en 3, leur preparation et leur application en therapeutique |
| PE20090597A1 (es) | 2007-08-16 | 2009-06-06 | Boehringer Ingelheim Int | Composicion farmaceutica que comprende un derivado de pirazol-o-glucosido |
| PE20090603A1 (es) | 2007-08-16 | 2009-06-11 | Boehringer Ingelheim Int | Composicion farmaceutica que comprende un inhibidor de sglt2 y un inhibidor de dpp iv |
| WO2009024542A2 (en) | 2007-08-17 | 2009-02-26 | Boehringer Ingelheim International Gmbh | Purin derivatives for use in the treatment of fab-related diseases |
| US9024009B2 (en) | 2007-09-10 | 2015-05-05 | Janssen Pharmaceutica N.V. | Process for the preparation of compounds useful as inhibitors of SGLT |
| CN107115523A (zh) | 2007-11-16 | 2017-09-01 | 诺沃—诺迪斯克有限公司 | 包含glp‑1肽或毒蜥外泌肽‑4和基础胰岛素肽的药物组合物 |
| CN101234105A (zh) | 2008-01-09 | 2008-08-06 | 北京润德康医药技术有限公司 | 一种含有二甲双胍和维格列汀的药用组合物及其制备方法 |
| CL2008003653A1 (es) | 2008-01-17 | 2010-03-05 | Mitsubishi Tanabe Pharma Corp | Uso de un inhibidor de sglt derivado de glucopiranosilo y un inhibidor de dppiv seleccionado para tratar la diabetes; y composicion farmaceutica. |
| EP2249643A4 (en) | 2008-02-05 | 2013-10-09 | Merck Sharp & Dohme | PHARMACEUTICAL COMPOSITIONS OF A METFORMIN AND DIPEPTIDYL PEPTIDASE-IV INHIBITOR ASSOCIATION |
| US8551524B2 (en) | 2008-03-14 | 2013-10-08 | Iycus, Llc | Anti-diabetic combinations |
| CA2719507C (en) | 2008-03-31 | 2018-03-27 | Metabolex, Inc. | Oxymethylene aryl compounds and uses thereof |
| PE20091730A1 (es) | 2008-04-03 | 2009-12-10 | Boehringer Ingelheim Int | Formulaciones que comprenden un inhibidor de dpp4 |
| PE20100156A1 (es) | 2008-06-03 | 2010-02-23 | Boehringer Ingelheim Int | Tratamiento de nafld |
| UY32030A (es) | 2008-08-06 | 2010-03-26 | Boehringer Ingelheim Int | "tratamiento para diabetes en pacientes inapropiados para terapia con metformina" |
| BRPI0917675A2 (pt) | 2008-08-15 | 2015-12-01 | Boehringer Ingelheim Int | compostos orgânicos para cura de ferida |
| MX2011002558A (es) | 2008-09-10 | 2011-04-26 | Boehringer Ingelheim Int | Terapia de combinacion para el tratamiento de diabetes y estados relacionados. |
| UY32177A (es) | 2008-10-16 | 2010-05-31 | Boehringer Ingelheim Int | Tratamiento de diabetes en pacientes con control glucémico insuficiente a pesar de la terapia con fármaco, oral o no, antidiabético |
| WO2010045656A2 (en) | 2008-10-17 | 2010-04-22 | Nectid, Inc. | Novel sglt2 inhibitor dosage forms |
| EA022310B1 (ru) | 2008-12-23 | 2015-12-30 | Бёрингер Ингельхайм Интернациональ Гмбх | Солевые формы органического соединения |
| AR074990A1 (es) | 2009-01-07 | 2011-03-02 | Boehringer Ingelheim Int | Tratamiento de diabetes en pacientes con un control glucemico inadecuado a pesar de la terapia con metformina |
| AR075204A1 (es) | 2009-01-29 | 2011-03-16 | Boehringer Ingelheim Int | Inhibidores de dpp-4 y composiciones farmaceuticas que los comprenden, utiles para tratar enfermedades metabolicas en pacientes pediatricos, particularmente diabetes mellitus tipo 2 |
| PL2395983T3 (pl) | 2009-02-13 | 2020-09-07 | Boehringer Ingelheim International Gmbh | Kompozycja farmaceutyczna zawierająca inhibitor sglt2, inhibitor dpp-iv i ewentualnie dalszy środek przeciwcukrzycowy oraz jej zastosowania |
| UY32427A (es) | 2009-02-13 | 2010-09-30 | Boheringer Ingelheim Internat Gmbh | Composicion farmaceutica, forma farmaceutica, procedimiento para su preparacion, metodos de tratamiento y usos de la misma |
| AP2011005795A0 (en) | 2009-02-13 | 2011-08-31 | Boehringer Ingelheim Int | SGLT-2 inhibitor for treating type 1 diabetes mellitus, type 2 diabetes mellitus, impaired glucose tolerance or hyperglycemia. |
| CA2752437C (en) | 2009-02-13 | 2017-07-11 | Boehringer Ingelheim International Gmbh | Antidiabetic medications |
| JP5600328B2 (ja) | 2009-02-13 | 2014-10-01 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | グルコピラノシルジフェニルメタン誘導体を含む医薬組成物、その医薬剤形、それらの調製方法及び患者の血糖コントロールを改善するためのそれらの使用 |
| CN106075451A (zh) | 2009-05-27 | 2016-11-09 | 阿斯利康(瑞典)有限公司 | 使用sglt2抑制剂及其组合物在对先前用其它抗糖尿病药进行的治疗具有耐受的患者中治疗ii型糖尿病的方法 |
| JP2012530135A (ja) | 2009-06-15 | 2012-11-29 | メルク・シャープ・エンド・ドーム・コーポレイション | ジペプチジルペプチダーゼ−4阻害剤及びピオグリタゾンの併用医薬組成物 |
| EA020798B1 (ru) | 2009-09-30 | 2015-01-30 | Бёрингер Ингельхайм Интернациональ Гмбх | СПОСОБ ПОЛУЧЕНИЯ КРИСТАЛЛИЧЕСКОЙ ФОРМЫ 1-ХЛОР-4-(β-D-ГЛЮКОПИРАНОЗ-1-ИЛ)-2-[4-((S)-ТЕТРАГИДРОФУРАН-3-ИЛОКСИ)БЕНЗИЛ]БЕНЗОЛА |
| SI2486029T1 (sl) | 2009-09-30 | 2015-10-30 | Boehringer Ingelheim International Gmbh | Postopki za pripravo z glukopiranozilom substituiranih benzil-benzenskih derivatov |
| UY32919A (es) | 2009-10-02 | 2011-04-29 | Boehringer Ingelheim Int | Composición farmacéutica, forma de dosificación farmacéutica, procedimiento para su preparación, mé todos para su tratamiento y sus usos |
| EP2482812B1 (en) | 2009-10-02 | 2023-01-11 | Boehringer Ingelheim International GmbH | Pharmaceutical compositions comprising bi-1356 and metformin |
| DK2498759T3 (en) | 2009-11-13 | 2018-11-26 | Astrazeneca Ab | TABLE FORMULATIONS WITH IMMEDIATE RELEASE |
| MX364651B (es) | 2009-11-27 | 2019-05-03 | Boehringer Ingelheim Int Gmbh Star | Inhibidores de dpp-iv, tales como la linagliptina, y composiciones farmacéuticas o combinaciones que comprenden los mismos, para usarse en el tratamiento de pacientes diabéticos tipificados genéticamente. |
| EP2547339A1 (en) | 2010-03-18 | 2013-01-23 | Boehringer Ingelheim International GmbH | Combination of a gpr119 agonist and the dpp-iv inhibitor linagliptin for use in the treatment of diabetes and related conditions |
| EP2368552A1 (en) | 2010-03-25 | 2011-09-28 | Boehringer Ingelheim Vetmedica GmbH | 1-[(3-cyano-pyridin-2-yl)methyl]-3-methyl-7-(2-butyn-1-yl)-8-[3-(r)-amino-piperidin-1-yl]-xanthine for the treatment of a metabolic disorder of a predominantly carnivorous non-human animal |
| EP2552442A1 (en) | 2010-03-30 | 2013-02-06 | Boehringer Ingelheim International GmbH | Pharmaceutical composition comprising an sglt2 inhibitor and a ppar- gamma agonist and uses thereof |
| EP2566469B1 (en) | 2010-05-05 | 2022-12-21 | Boehringer Ingelheim International GmbH | Combination therapy |
| BR112012028091A2 (pt) | 2010-05-05 | 2016-08-02 | Boehringer Ingelheim Int | composições farmacêuticas compreendendo pioglitazona e linagliptina |
| EA201991014A1 (ru) | 2010-06-24 | 2019-09-30 | Бёрингер Ингельхайм Интернациональ Гмбх | Лечение диабета |
| US20120283169A1 (en) | 2010-11-08 | 2012-11-08 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| AR083878A1 (es) | 2010-11-15 | 2013-03-27 | Boehringer Ingelheim Int | Terapia antidiabetica vasoprotectora y cardioprotectora, linagliptina, metodo de tratamiento |
| US20130035281A1 (en) | 2011-02-09 | 2013-02-07 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
| UY33937A (es) | 2011-03-07 | 2012-09-28 | Boehringer Ingelheim Int | Composiciones farmacéuticas que contienen inhibidores de dpp-4 y/o sglt-2 y metformina |
| US20130035298A1 (en) | 2011-07-08 | 2013-02-07 | Boehringer Ingelheim International Gmbh | Pharmaceutical composition, methods for treating and uses thereof |
-
2008
- 2008-08-14 PE PE2008001382A patent/PE20090938A1/es active IP Right Grant
- 2008-08-14 CL CL2008002427A patent/CL2008002427A1/es unknown
- 2008-08-15 AR ARP080103592A patent/AR067970A1/es not_active Application Discontinuation
- 2008-08-15 LT LTEP08787264.4T patent/LT2187879T/lt unknown
- 2008-08-15 RS RS20160963A patent/RS55205B1/sr unknown
- 2008-08-15 HU HUE08787264A patent/HUE030158T2/en unknown
- 2008-08-15 UY UY31296A patent/UY31296A1/es active IP Right Grant
- 2008-08-15 US US12/673,327 patent/US8551957B2/en active Active
- 2008-08-15 TW TW103116634A patent/TW201436798A/zh unknown
- 2008-08-15 UA UAA201002970A patent/UA100384C2/ru unknown
- 2008-08-15 NZ NZ583242A patent/NZ583242A/xx unknown
- 2008-08-15 EP EP08787264.4A patent/EP2187879B1/en active Active
- 2008-08-15 KR KR20107003411A patent/KR101491554B1/ko active IP Right Review Request
- 2008-08-15 TW TW97131324A patent/TWI472325B/zh active
- 2008-08-15 BR BRPI0815331A patent/BRPI0815331B8/pt active IP Right Grant
- 2008-08-15 AU AU2008288407A patent/AU2008288407B2/en active Active
- 2008-08-15 ME MEP-2016-255A patent/ME02573B/me unknown
- 2008-08-15 EP EP16172707.8A patent/EP3106156A1/en not_active Withdrawn
- 2008-08-15 WO PCT/EP2008/060736 patent/WO2009022007A1/en active Application Filing
- 2008-08-15 CN CN201410482423.1A patent/CN104288166A/zh active Pending
- 2008-08-15 EP EP13180262.1A patent/EP2698152A1/en not_active Withdrawn
- 2008-08-15 CN CN201410482945.1A patent/CN104353077A/zh active Pending
- 2008-08-15 SI SI200831745A patent/SI2187879T1/sl unknown
- 2008-08-15 JP JP2010520594A patent/JP5595914B2/ja active Active
- 2008-08-15 ES ES08787264.4T patent/ES2602748T3/es active Active
- 2008-08-15 MY MYPI20100681 patent/MY152037A/en unknown
- 2008-08-15 EP EP21189804.4A patent/EP3939577A1/en active Pending
- 2008-08-15 EA EA201000321A patent/EA018608B1/ru active Protection Beyond IP Right Term
- 2008-08-15 CN CN200880102911A patent/CN101784270A/zh active Pending
- 2008-08-15 MX MX2010001696A patent/MX2010001696A/es active IP Right Grant
- 2008-08-15 PT PT87872644T patent/PT2187879T/pt unknown
- 2008-08-15 PL PL08787264T patent/PL2187879T3/pl unknown
- 2008-08-15 DK DK08787264.4T patent/DK2187879T3/en active
- 2008-08-15 CA CA2696558A patent/CA2696558C/en active Active
-
2009
- 2009-12-21 ZA ZA200909105A patent/ZA200909105B/xx unknown
- 2009-12-22 IL IL202886A patent/IL202886A/en active IP Right Grant
-
2010
- 2010-02-05 CO CO10012795A patent/CO6251239A2/es active IP Right Grant
- 2010-02-12 TN TNP2010000073A patent/TN2010000073A1/fr unknown
- 2010-02-15 MA MA32622A patent/MA31612B1/fr unknown
- 2010-02-19 EC EC2010009977A patent/ECSP109977A/es unknown
- 2010-11-11 HK HK15101597.8A patent/HK1201721A1/xx unknown
- 2010-11-11 HK HK15102815.2A patent/HK1203351A1/xx unknown
-
2012
- 2012-08-23 AR ARP120103117A patent/AR087657A2/es not_active Application Discontinuation
-
2016
- 2016-12-09 CY CY20161101277T patent/CY1118308T1/el unknown
-
2017
- 2017-01-09 HR HRP20170022TT patent/HRP20170022T1/hr unknown
- 2017-05-05 NO NO2017020C patent/NO2017020I2/no unknown
- 2017-05-08 LT LTPA2017014C patent/LTC2187879I2/lt unknown
- 2017-05-08 NL NL300872C patent/NL300872I2/nl unknown
- 2017-05-09 CY CY2017017C patent/CY2017017I1/el unknown
- 2017-05-10 HU HUS1700020C patent/HUS1700020I1/hu unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| BRPI0815331B1 (pt) | Composição farmacêutica compreendendo um derivado de benzeno glucopiranosila substituída,bem como seu uso | |
| US20230285432A1 (en) | Pharmaceutical composition, methods for treating and uses thereof | |
| ES2930655T3 (es) | Usos terapéuticos de empagliflozina | |
| WO2009022009A1 (en) | Pharmaceutical composition comprising a pyrazole-o-glucoside derivative | |
| US20130096076A1 (en) | Pharmaceutical composition comprising a sglt2 inhibitor in combination with a dpp-iv inhibitor | |
| WO2009022008A1 (en) | Pharmaceutical composition comprising a pyrazole-o-glucoside derivative | |
| BRPI1013638A2 (pt) | composição farmacêutica que compreende derivados de glicopiranosildifenil-metano, sua forma de dosagem farmacêutica, processo para sua preparação, e seus usos para melhor controle glicèmico em um paciente | |
| TW200838548A (en) | Pharmaceutical composition comprising a pyrazole-O-glucoside derivative | |
| JP2010536734A6 (ja) | Dpp iv阻害剤と併用するsglt2阻害剤を含む医薬組成物 | |
| BRPI0706375A2 (pt) | forma cristalina de 1'-(1-metiletil)-4'-[(2 flúor-4-metoxifenil)metil]-5'-metil-1h-pirazol-3'-o-bet a-d-glucopiranosìdeo, um método para sua preparação e o uso da mesma para preparo de medicamentos | |
| BR112012009376B1 (pt) | composição farmacêutica sólida compreendendo um inibidor de sglt-2 e sua forma de dosagem farmacêutica | |
| JP2013523681A (ja) | Sglt2インヒビター及びppar−ガンマアゴニストを含む医薬組成物並びにその使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| B06F | Objections, documents and/or translations needed after an examination request according [chapter 6.6 patent gazette] | ||
| B07D | Technical examination (opinion) related to article 229 of industrial property law [chapter 7.4 patent gazette] |
Free format text: DE ACORDO COM O ARTIGO 229-C DA LEI NO 10196/2001, QUE MODIFICOU A LEI NO 9279/96, A CONCESSAO DA PATENTE ESTA CONDICIONADA A ANUENCIA PREVIA DA ANVISA. CONSIDERANDO A APROVACAO DOS TERMOS DO PARECER NO 337/PGF/EA/2010, BEM COMO A PORTARIA INTERMINISTERIAL NO 1065 DE 24/05/2012, ENCAMINHA-SE O PRESENTE PEDIDO PARA AS PROVIDENCIAS CABIVEIS. |
|
| B07E | Notification of approval relating to section 229 industrial property law [chapter 7.5 patent gazette] | ||
| B06U | Preliminary requirement: requests with searches performed by other patent offices: procedure suspended [chapter 6.21 patent gazette] | ||
| B09A | Decision: intention to grant [chapter 9.1 patent gazette] | ||
| B16A | Patent or certificate of addition of invention granted [chapter 16.1 patent gazette] |
Free format text: PRAZO DE VALIDADE: 10 (DEZ) ANOS CONTADOS A PARTIR DE 06/04/2021, OBSERVADAS AS CONDICOES LEGAIS. |
|
| B16C | Correction of notification of the grant [chapter 16.3 patent gazette] |
Free format text: PRAZO DE VALIDADE: 20 (VINTE) ANOS CONTADOS A PARTIR DE 15/08/2008 OBSERVADAS AS CONDICOES LEGAIS. PATENTE CONCEDIDA CONFORME ADI 5.529/DF |
|
| B17A | Notification of administrative nullity (patentee has 60 days time to reply to this notification) |
Free format text: REQUERENTE DA NULIDADE: EMS.S.A. - 870210091672 - 04/10/2021 |