WO2010074270A1 - ブロック共重合体組成物、フィルムおよびブロック共重合体組成物の製造方法 - Google Patents
ブロック共重合体組成物、フィルムおよびブロック共重合体組成物の製造方法 Download PDFInfo
- Publication number
- WO2010074270A1 WO2010074270A1 PCT/JP2009/071692 JP2009071692W WO2010074270A1 WO 2010074270 A1 WO2010074270 A1 WO 2010074270A1 JP 2009071692 W JP2009071692 W JP 2009071692W WO 2010074270 A1 WO2010074270 A1 WO 2010074270A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- block copolymer
- aromatic vinyl
- block
- conjugated diene
- copolymer composition
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L53/00—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L53/02—Compositions of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers of vinyl-aromatic monomers and conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F297/00—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer
- C08F297/02—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type
- C08F297/04—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type polymerising vinyl aromatic monomers and conjugated dienes
- C08F297/044—Macromolecular compounds obtained by successively polymerising different monomer systems using a catalyst of the ionic or coordination type without deactivating the intermediate polymer using a catalyst of the anionic type polymerising vinyl aromatic monomers and conjugated dienes using a coupling agent
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2353/00—Characterised by the use of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives of such polymers
- C08J2353/02—Characterised by the use of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives of such polymers of vinyl aromatic monomers and conjugated dienes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2205/00—Polymer mixtures characterised by other features
- C08L2205/02—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group
- C08L2205/025—Polymer mixtures characterised by other features containing two or more polymers of the same C08L -group containing two or more polymers of the same hierarchy C08L, and differing only in parameters such as density, comonomer content, molecular weight, structure
Definitions
- the present invention relates to a block copolymer composition comprising an aromatic vinyl-conjugated diene-aromatic vinyl block copolymer and a method for producing the same, and more particularly, a sanitary article member such as a paper diaper or a sanitary article.
- the present invention relates to a method for producing a copolymer composition.
- Aromatic vinyl-conjugated diene-aromatic vinyl block copolymers such as styrene-isoprene-styrene block copolymer (SIS) and styrene-butadiene-styrene block copolymer (SBS) are characteristic in various aspects. Since it is a thermoplastic elastomer having properties, it is used in various applications. Aromatic vinyl-conjugated diene-aromatic vinyl block copolymers are particularly elastic and flexible among thermoplastic elastomers, so they are materials for stretchable films used in sanitary products such as disposable diapers and sanitary products. As one of the typical uses of an aromatic vinyl-conjugated diene-aromatic vinyl block copolymer.
- SIS styrene-isoprene-styrene block copolymer
- SBS styrene-butadiene-styrene block copolymer
- stretchable films are used for each part.
- a pants-type diaper which is a type of paper diaper
- stretchable films are disposed at the openings around the legs, the openings around the waist, and the waists on both sides. Even if the wearer of the hygiene product moves violently or wears it for a long time, it is necessary not to cause displacement, so the elastic film used for such applications has a high elastic modulus and Although it is required to have a small permanent elongation, it has been difficult to say that the conventional aromatic vinyl-conjugated diene-aromatic vinyl block copolymer has achieved both of these properties. For this reason, various studies for improving the elastic modulus and permanent elongation of aromatic vinyl-conjugated diene-aromatic vinyl block copolymers have been conducted.
- Patent Document 1 includes 65 to 92 parts by weight of a specific elastomeric monovinylidene aromatic-conjugated diene block copolymer having a monovinylidene aromatic content of less than 50% by weight, and a monovinylidene aromatic content of 50% by weight or more. It is disclosed that an elastomer polymer blend composition comprising 8 to 35 parts by weight of a specific thermoplastic monovinylidene aromatic-conjugated diene block copolymer having an elastomer article exhibiting excellent elasticity and stress relaxation properties is disclosed. Yes.
- Patent Document 2 discloses that a composition obtained by blending a specific aromatic vinyl-conjugated diene block copolymer with a specific polyisoprene or the like is extruded to have anisotropy and flexibility. It is disclosed that an elastic film that is excellent in properties and can be suitably used as a member of sanitary products such as disposable diapers and sanitary products.
- the stretchable film is usually formed by extrusion.
- a conventional aromatic vinyl-conjugated diene-aromatic vinyl block copolymer is formed by extrusion, mechanical properties resulting from molecular orientation are obtained.
- anisotropy of this was likely to occur.
- Such materials that tend to cause anisotropy generally have poor molding stability, and there is a problem that the thickness accuracy deteriorates even when a film is molded.
- an elastic film as a sanitary product such as a disposable diaper or sanitary product, if the elastic film has anisotropy, the wearing feeling of the sanitary product will be different from the normal underwear, etc. There is also the problem of giving a sense of incongruity.
- the present invention has a high elastic modulus and a small permanent elongation at a high level, and further has uniform mechanical properties even when a molding method such as extrusion molding is likely to occur. It is an object of the present invention to provide a block copolymer composition containing an aromatic vinyl-conjugated diene-aromatic vinyl block copolymer which gives a molded article having high isotropic properties.
- an asymmetric aromatic vinyl-conjugated diene-aromatic vinyl block copolymer in which two aromatic vinyl polymer blocks have different specific weight average molecular weights.
- a block copolymer composition comprising a polymer and a branched aromatic vinyl-conjugated diene-aromatic vinyl block copolymer having a specific configuration in a specific ratio, a high elastic modulus It was found that a small permanent elongation is compatible at a high level.
- the film when the inventors form a film using this block copolymer composition, the film has a high elastic modulus and a small permanent elongation that are compatible at a high level, and is repeatedly stretched. Even if it is a case where it is difficult to change the elastic modulus even if it is applied, and even when a molding method such as extrusion molding is likely to cause molecular orientation, the film has uniform mechanical properties over all plane directions. We found that it is highly isotropic.
- the present inventors have also studied the method for producing this block copolymer composition, and after producing an aromatic vinyl-conjugated diene block copolymer having an active end, According to the method in which a part is coupled with a trifunctional or higher functional coupling agent and an aromatic vinyl polymer block is formed at the active terminal of the remaining copolymer, a block copolymer composition having a particularly desirable structure is produced. It was found that it can be obtained with good performance. The present invention has been completed based on these findings.
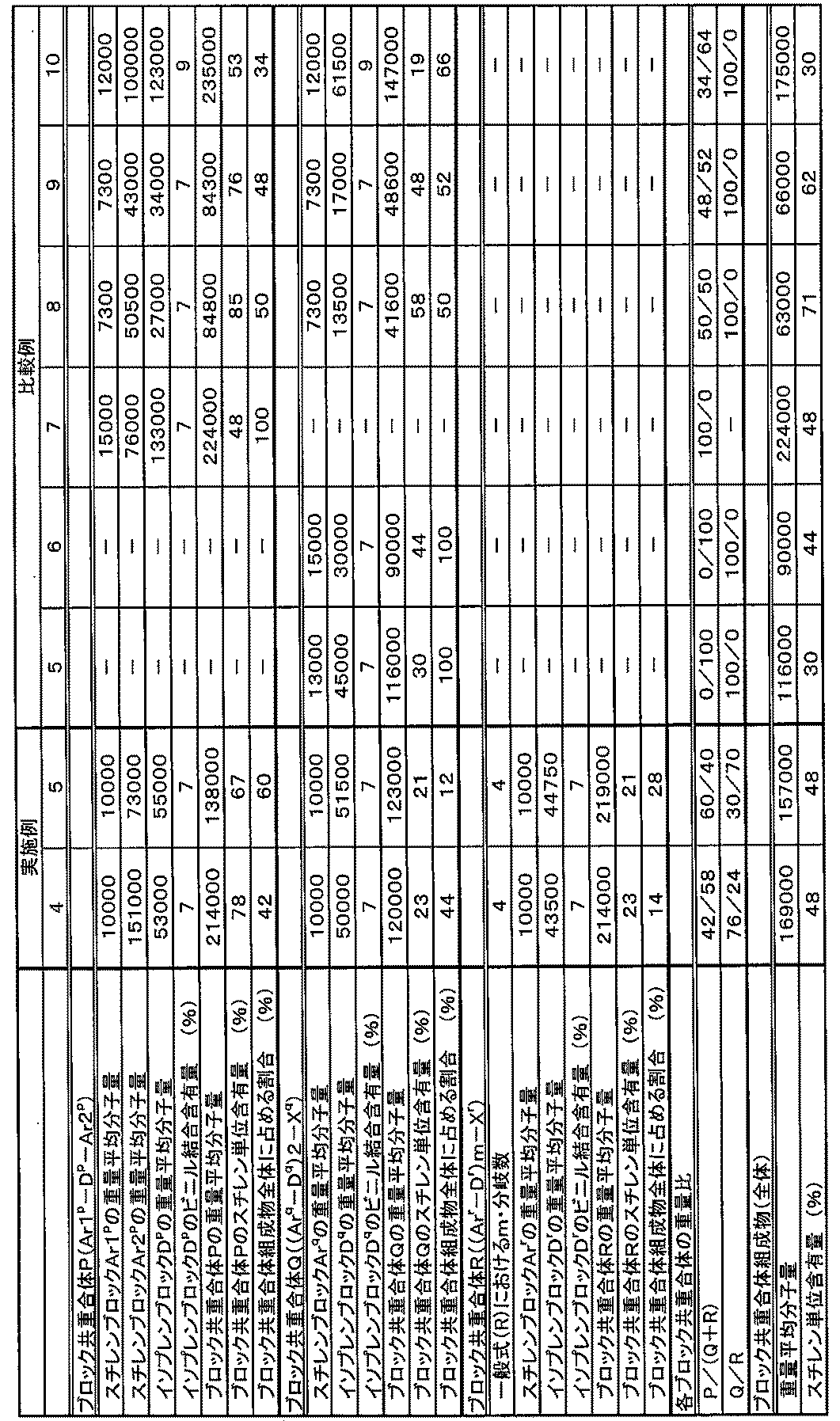
- a block copolymer comprising the block copolymer A represented by the following general formula (A) and the block copolymer B represented by the following general formula (B):
- A block copolymer A
- B block copolymer B
- the weight ratio (A / B) of block copolymer A to block copolymer B is from 36/64 to 85/15.
- Ar1 a -D a -Ar2 a (A) (Ar b -D b ) n -X (B)
- Ar @ 1 a and Ar b are each an aromatic vinyl polymer block having a weight average molecular weight of 6000 ⁇ 15000
- Ar2 a the weight average molecular weight of 40000 to 400000
- An aromatic vinyl polymer block, D a and D b are each a conjugated diene polymer block having a vinyl bond content of 1 to 20 mol%
- X is a residue of a coupling agent
- n is (It is an integer of 3 or more.)
- the proportion of the aromatic vinyl monomer unit in the total repeating units of the polymer component of the block copolymer composition is preferably 27 to 70% by weight.
- a film formed by molding the block copolymer composition is provided.
- a method for producing the above block copolymer composition comprising the following steps (1) to (5).
- the inventors of the present invention have an asymmetric aromatic vinyl-conjugated diene-aromatic vinyl block copolymer in which two aromatic vinyl polymer blocks have different specific weight average molecular weights, and a different specific configuration.
- a block comprising a linear aromatic vinyl-conjugated diene-aromatic vinyl block copolymer having a branched structure and a branched aromatic vinyl-conjugated diene-aromatic vinyl block copolymer having a specific configuration According to the copolymer composition, it was found that a high elastic modulus and a small permanent elongation are compatible at a high level.
- the film when the inventors form a film using this block copolymer composition, the film has a high elastic modulus and a small permanent elongation that are compatible at a high level, and is repeatedly stretched. Even if it is a case where it is difficult to change the elastic modulus even if it is applied, and even when a molding method such as extrusion molding is likely to cause molecular orientation, the film has uniform mechanical properties over all plane directions. We found that it is highly isotropic.
- the present inventors have also studied the method for producing this block copolymer composition, and after producing an aromatic vinyl-conjugated diene block copolymer having an active end, According to a method in which a part is coupled in combination with a bifunctional coupling agent and a trifunctional or higher functional coupling agent, and an aromatic vinyl polymer block is formed at the active terminal of the remaining copolymer, a particularly desirable configuration It has been found that a block copolymer composition having the above can be obtained with good productivity. The present invention has been completed based on these findings.
- P the block copolymer
- Q the block copolymer
- R the following general formula
- Ar1 p , Ar q , and Ar r are each an aromatic vinyl polymer block having a weight average molecular weight of 6000 to 20000, and Ar2 p is , An aromatic vinyl polymer block having a weight average molecular weight of 40,000 to 400,000, and D p , D q , and D r are each a conjugated diene polymer block having a vinyl bond content of 1 to 20 mol%, X q is the residue of a single bond, or a coupling agent, X r is a residue of a coupling agent, m is an integer of 3 or more.)
- the elastic modulus when a molding method having a high elastic modulus and a small permanent elongation, the elastic modulus hardly changes even when repeatedly expanded and contracted, and molecular orientation such as extrusion molding is likely to occur.
- a highly isotropic film having uniform mechanical properties over all plane directions can be obtained.
- the above block copolymer composition has a weight ratio (P / (Q + R)) between the amount of the block copolymer P and the total amount of the block copolymer Q and the block copolymer R of 10/90 to It is preferable that it is 80/20.
- the weight ratio (Q / R) between the amount of the block copolymer Q and the amount of the block copolymer R is preferably 15/85 to 85/15.
- the proportion of the aromatic vinyl monomer unit in the total repeating units of the polymer component of the block copolymer composition is preferably 27 to 70% by weight.
- a film formed by molding the block copolymer composition is provided.
- a method for producing the above block copolymer composition comprising the following steps (6) to (10).
- the block copolymer composition of the present invention can be obtained with high productivity.
- the elastic modulus when a molding method having a high elastic modulus and a small permanent elongation, the elastic modulus hardly changes even when repeatedly expanded and contracted, and molecular orientation such as extrusion molding is likely to occur.
- a highly isotropic film having uniform mechanical properties over all plane directions can be obtained.
- the block copolymer composition of the present invention can be obtained with high productivity.
- the block copolymer composition of the present invention includes an embodiment (first embodiment) containing two block copolymers and an embodiment (second embodiment) containing at least three block copolymers. It can be divided into two aspects. Hereinafter, the block copolymer composition of the present invention will be described in each embodiment.
- the block copolymer composition of this embodiment contains two types of block copolymers.
- the block copolymer A which is one of the two block copolymers constituting the block copolymer composition of this embodiment, has a weight average molecular weight different from each other represented by the following general formula (A).
- Ar1 a is an aromatic vinyl polymer block having a weight average molecular weight of 6000 to 15000
- Ar2 a is an aromatic vinyl polymer block having a weight average molecular weight of 40000 to 400,000.
- Da is a conjugated diene polymer block having a vinyl bond content of 1 to 20 mol%.
- the block copolymer B which is the other of the two block copolymers constituting the block copolymer composition of this embodiment is a branched block copolymer represented by the following general formula (B). It is a coalescence.
- Ar b is an aromatic vinyl polymer block having a weight average molecular weight of 6000 to 15000, and D b is a conjugate having a vinyl bond content of 1 to 20 mol%. It is a diene polymer block, X is a residue of a coupling agent, and n is an integer of 3 or more.
- the aromatic vinyl polymer blocks (Ar1 a , Ar2 a , Ar b ) of the block copolymer A and the block copolymer B are polymer blocks composed of aromatic vinyl monomer units.
- the aromatic vinyl monomer used for constituting the aromatic vinyl monomer unit of the aromatic vinyl polymer block is not particularly limited as long as it is an aromatic vinyl compound, but styrene, ⁇ -methylstyrene, 2 -Methylstyrene, 3-methylstyrene, 4-methylstyrene, 2-ethylstyrene, 3-ethylstyrene, 4-ethylstyrene, 2,4-diisopropylstyrene, 2,4-dimethylstyrene, 4-t-butylstyrene, 5-t-butyl-2-methylstyrene, 2-chlorostyrene, 3-chlorostyrene, 4-chlorostyrene, 4-bromosty
- aromatic vinyl monomers can be used alone or in combination of two or more in each aromatic vinyl polymer block. Further, in each aromatic vinyl polymer block, the same aromatic vinyl monomer may be used, or different aromatic vinyl monomers may be used.
- Each of the aromatic vinyl polymer blocks (Ar1 a , Ar2 a , Ar b ) of the block copolymer A and the block copolymer B may contain a monomer unit other than the aromatic vinyl monomer unit.
- Monomers constituting monomer units other than aromatic vinyl monomer units that can be included in the aromatic vinyl polymer block include 1,3-butadiene and isoprene (2-methyl-1,3-butadiene). Examples thereof include conjugated diene monomers such as ⁇ , ⁇ -unsaturated nitrile monomers, unsaturated carboxylic acid or acid anhydride monomers, unsaturated carboxylic acid ester monomers, and non-conjugated diene monomers.
- the content of monomer units other than aromatic vinyl monomer units in each aromatic vinyl polymer block is preferably 20% by weight or less, more preferably 10% by weight or less, and substantially Particularly preferred is 0% by weight.
- the conjugated diene polymer block (D a , D b ) of the block copolymer A and the block copolymer B is a polymer block composed of conjugated diene monomer units.
- the conjugated diene monomer used for constituting the conjugated diene monomer unit of the conjugated diene polymer block is not particularly limited as long as it is a conjugated diene compound.
- 1,3-butadiene, isoprene, 2 Examples include 3-dimethyl-1,3-butadiene, 2-chloro-1,3-butadiene, 1,3-pentadiene, and 1,3-hexadiene.
- conjugated diene polymer block By constituting the conjugated diene polymer block with isoprene units, a block copolymer composition having excellent flexibility and lower permanent elongation can be obtained.
- conjugated diene monomers can be used alone or in combination of two or more in each conjugated diene polymer block. In each conjugated diene polymer block, the same conjugated diene monomer may be used, or different conjugated diene monomers may be used. Furthermore, you may perform hydrogenation reaction with respect to a part of unsaturated bond of each conjugated diene polymer block.
- the conjugated diene polymer blocks (D a , D b ) of the block copolymer A and the block copolymer B may each contain a monomer unit other than the conjugated diene monomer unit.
- Monomers constituting monomer units other than the conjugated diene monomer unit that can be included in the conjugated diene polymer block include aromatic vinyl monomers such as styrene and ⁇ -methylstyrene, ⁇ , ⁇ -unsaturated monomers, and the like. Examples include saturated nitrile monomers, unsaturated carboxylic acid or acid anhydride monomers, unsaturated carboxylic acid ester monomers, and non-conjugated diene monomers.
- the content of monomer units other than the conjugated diene monomer unit in each conjugated diene polymer block is preferably 20% by weight or less, more preferably 10% by weight or less, and substantially 0% by weight. % Is particularly preferred.
- the block copolymer A constituting the block copolymer composition of this embodiment is an aromatic vinyl polymer block (Ar1 a ) having a relatively small weight average molecular weight as represented by the general formula (A).
- a conjugated diene polymer block having a specific vinyl bond content (D a ) and an aromatic vinyl polymer block having a relatively large weight average molecular weight (Ar 2 a ) are connected in a straight chain in this order.
- the aromatic vinyl polymer block (Ar1 a ) having a relatively small weight average molecular weight has a weight average molecular weight (Mw (Ar1 a )) of 6000 to 15,000, preferably 7000 to 14,000, and preferably 8,000 to 13,000. More preferably. If Mw (Ar1 a ) is outside this range, the resulting composition may have insufficient permanent elongation.
- the aromatic vinyl polymer block (Ar2 a ) having a relatively large weight average molecular weight has a weight average molecular weight (Mw (Ar2 a )) of 40,000 to 400,000, preferably 42,000 to 370000, and preferably 45,000 to More preferably, it is 350,000.
- Mw (Ar2 a) is too small, the elongation of the resulting composition is insufficient, Mw (Ar2 a) is a block copolymer A too large may manufacture is difficult.
- the weight average molecular weight of the polymer or polymer block is determined as a value in terms of polystyrene measured by high performance liquid chromatography.
- the ratio of the weight average molecular weight of polymer block (Ar1 a) (Mw (Ar1 a)) (Mw (Ar2 a) / Mw (Ar1 a)) is not particularly limited but is usually from 2.6 to 67, 4 Is preferably from 40 to 40, more preferably from 4.5 to 35.
- the vinyl bond content of the conjugated diene polymer block (D a ) of the block copolymer A (the ratio of 1,2-vinyl bonds and 3,4-vinyl bonds in all conjugated diene monomer units) is 1 ⁇ 20 mol%, preferably 2 to 15 mol%, more preferably 3 to 10 mol%. If the vinyl bond content is too high, the permanent elongation of the resulting block copolymer composition may increase.
- the weight average molecular weight (Mw (D a )) of the conjugated diene polymer block (D a ) of the block copolymer A is not particularly limited, but is usually 20000-200000, preferably 30000-150,000, preferably 35000. More preferably, it is ⁇ 100,000.
- the content of the aromatic vinyl monomer unit with respect to the total monomer units of the block copolymer A is not particularly limited, but is usually 40 to 90% by weight, preferably 45 to 87% by weight, More preferably, it is ⁇ 85% by weight.
- the weight average molecular weight of the block copolymer A as a whole is not particularly limited, but is usually 50,000 to 500,000, preferably 80000 to 470000, and more preferably 90000 to 450,000.
- the block copolymer B constituting the block copolymer composition of the present embodiment includes an aromatic vinyl polymer block (Ar b ) having a specific weight average molecular weight, as represented by the general formula (B).
- a diblock body (Ar b -D b ) formed by bonding with a conjugated diene polymer block (D b ) having a specific vinyl bond content is terminated at the aromatic vinyl polymer block (Ar b ) side.
- the weight average molecular weights (Mw (Ar b )) of the plurality of aromatic vinyl polymer blocks (Ar b ) included in the block copolymer B are 6000 to 15000, preferably 7000 to 14,000, respectively. More preferably, it is ⁇ 13000. If Mw (Ar b ) is out of this range, the resulting composition may have insufficient permanent elongation.
- the weight average molecular weights (Mw (Ar2 b )) of the aromatic vinyl polymer blocks present in plural in one molecule may be equal or different from each other as long as they are within the above range.
- the weight average molecular weight (Mw (Ar b )) of the plurality of aromatic vinyl polymer blocks is the weight of the aromatic vinyl polymer block (Ar1 a ) having a relatively small weight average molecular weight of the block copolymer A. More preferably, the average molecular weight (Mw (Ar1 a )) is substantially equal.
- the vinyl bond content of the conjugated diene polymer block (D b ) of the block copolymer B is 1 to 20 mol%, preferably 2 to 15 mol%, and preferably 3 to 10 mol%. More preferred. If the vinyl bond content is too high, the permanent elongation of the resulting block copolymer composition may increase.
- the vinyl bond content of the conjugated diene polymer block (D b ) of the block copolymer B is substantially equal to the vinyl bond content of the conjugated diene polymer block (D a ) of the block copolymer A. Is preferred.
- the weight average molecular weight (Mw (D b )) of the conjugated diene polymer block (D b ) of the block copolymer B is not particularly limited, but is usually 20000 to 200000, preferably 30000 to 150,000, preferably 35000. More preferably, it is ⁇ 100,000.
- the weight average molecular weight (Mw (D b )) of the conjugated diene polymer block (D b ) of the block copolymer B is the weight average molecular weight of the conjugated diene polymer block (D a ) of the block copolymer A ( Mw (D a )) is preferably substantially equal. When these weight average molecular weights are substantially equal, the resulting block copolymer composition has a higher elastic modulus and is rich in elasticity.
- the content of the aromatic vinyl monomer unit with respect to the total monomer units of the block copolymer B is not particularly limited, but is usually 10 to 35% by weight, preferably 12 to 32% by weight, More preferably, it is ⁇ 30% by weight.
- the weight average molecular weight of the block copolymer B as a whole is not particularly limited, but is usually 35,000 to 450,000, preferably 50,000 to 400,000, and more preferably 70000 to 350,000.
- the molecular weight distribution represented by (Mw / Mn) is not particularly limited, but is usually 1.1 or less, preferably 1.05 or less.
- the weight ratio (A / B) between the block copolymer A and the block copolymer B contained in the block copolymer composition of this embodiment is 36/64 to 85/15, and 38/62 to 80 / 20 is preferable, and 40/60 to 75/25 is more preferable.
- the block copolymer composition has both a high elastic modulus and a small permanent elongation.
- this ratio is too small, the elastic modulus of the block copolymer composition becomes insufficient, and when this ratio is too large, the permanent elongation of the block copolymer composition increases.
- the block copolymer composition of this embodiment may contain only the block copolymer A and the block copolymer B as the polymer component, but the other than the block copolymer A and the block copolymer B, It may contain a coalescing component.
- the polymer component other than the block copolymer A and the block copolymer B that can be included in the block copolymer composition of the present embodiment include aromatic vinyl-conjugated other than the block copolymer A and the block copolymer B.
- Polymer or thermoplastic elastomer such as polyurethane thermoplastic elastomer, polyamide thermoplastic elastomer, polyester thermoplastic elastomer, polyethylene, polypropylene, polyvinyl chloride, acrylonitrile-styrene copolymer, acrylonitrile-butadiene-styrene copolymer
- Polyphenylene ether Such as a thermoplastic resin.
- the content of the polymer component other than the block copolymer A and the block copolymer B is preferably 20% by weight or less based on the entire polymer component, More preferably, it is 10% by weight or less.
- the proportion of the aromatic vinyl monomer unit in all the repeating units of the entire polymer component contained is 27 to 70% by weight, preferably 30 to 60% by weight, and more preferably 40 to 50% by weight. If the overall aromatic vinyl monomer unit content is too low, the resulting block copolymer composition will have an insufficient elastic modulus, and if the overall aromatic vinyl monomer unit content is too high, it will be obtained. The permanent elongation of the block copolymer composition is increased.
- the total aromatic vinyl monomer unit content is determined by the block copolymer A, the block copolymer B and the other polymer components constituting the block copolymer composition, and the respective aromatic vinyl monomers. It can be easily adjusted by taking into account the content of the units and adjusting their blending amount. In the case where all the polymer components constituting the block copolymer composition are composed only of an aromatic vinyl monomer unit and a conjugated diene monomer unit, Rubber Chem. Technol.
- the weight average molecular weight of the whole polymer component constituting the block copolymer composition of this embodiment is not particularly limited, but is usually 50,000 to 500,000, preferably 60000 to 450,000, and preferably 70000 to 400,000. More preferred. Further, the molecular weight distribution represented by the ratio (Mw / Mn) of the weight average molecular weight (Mw) and the number average molecular weight (Mn) of the entire polymer component constituting the block copolymer composition of this embodiment is not particularly limited. However, it is usually 1.01 to 10, preferably 1.03 to 5, and more preferably 1.05 to 3.
- the melt index of the block copolymer composition of this embodiment is not particularly limited, but is usually 1 to 70 g / 10 as a value measured in accordance with ASTM D-1238 (G condition, 200 ° C., 5 kg). Minutes, preferably 3 to 65 g / 10 minutes, more preferably 5 to 60 g / 10 minutes. If it is this range, the moldability to the film of a block copolymer composition will become favorable.
- the block copolymer composition of this embodiment may contain components other than the polymer component, such as a softener, a tackifier, an antioxidant, an antibacterial agent, a light stabilizer, an ultraviolet absorber, a dye, You may mix
- the method for obtaining the block copolymer composition of this embodiment is not particularly limited.
- block copolymer A and block copolymer B are produced separately, and, if necessary, other polymer components and various additives are blended. They can be produced by mixing them according to conventional methods such as kneading and solution mixing.
- the method for producing the block copolymer composition of the present embodiment described below is preferable.
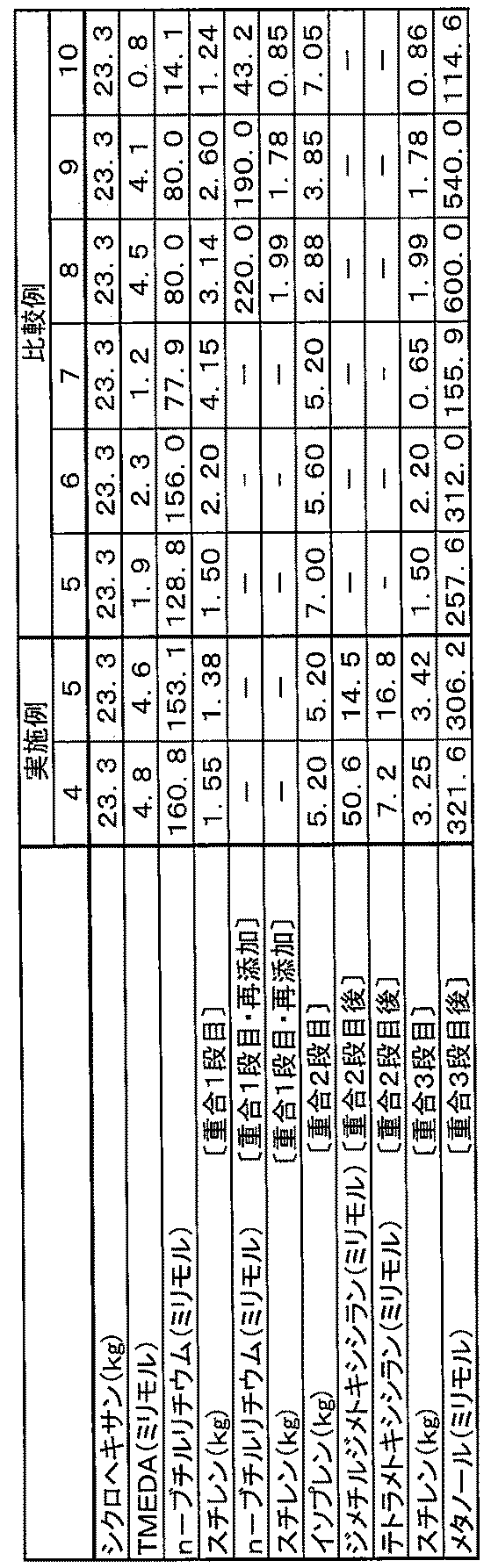
- the method for producing the block copolymer composition of this embodiment comprises the following steps (1) to (5).
- Step (5) of adding vinyl monomer to form block copolymer A Step of recovering the block copolymer composition from the solution obtained in the step (4)
- an aromatic vinyl monomer is polymerized using a polymerization initiator in a solvent.
- the polymerization initiator used is generally an organic alkali metal compound, an organic alkaline earth metal compound known to have anionic polymerization activity for an aromatic vinyl monomer and a conjugated diene monomer, Organic lanthanoid series rare earth metal compounds and the like can be used.
- the organic alkali metal compound an organic lithium compound having one or more lithium atoms in the molecule is particularly preferably used.
- Organic monolithium compounds such as sec-butyllithium, t-butyllithium, hexyllithium, phenyllithium, stilbenelithium, dialkylaminolithium, diphenylaminolithium, ditrimethylsilylaminolithium, methylenedilithium, tetramethylenedilithium, hexamethylene
- Organic dilithium compounds such as dilithium, isoprenyl dilithium, 1,4-dilithio-ethylcyclohexane, and organic trilithium compounds such as 1,3,5-trilithiobenzene It is.
- an organic monolithium compound is particularly preferably used.
- organic alkaline earth metal compound used as the polymerization initiator examples include n-butylmagnesium bromide, n-hexylmagnesium bromide, ethoxycalcium, calcium stearate, t-butoxystrontium, ethoxybarium, isopropoxybarium, ethyl mercaptobarium, Examples thereof include t-butoxybarium, phenoxybarium, diethylaminobarium, barium stearate, and ethylbarium.
- polymerization initiators include lanthanoid series rare earth metal compounds containing neodymium, samarium, gadolinium, etc./alkylaluminum/alkylaluminum halides / alkylaluminum hydrides, titanium, vanadium, samarium, gadolinium. Examples thereof include those having a uniform system in an organic solvent such as a metallocene-type catalyst containing a living polymer and the like and having living polymerizability. In addition, these polymerization initiators may be used individually by 1 type, and may mix and use 2 or more types.
- the amount of the polymerization initiator used may be determined according to the molecular weight of each target block copolymer, and is not particularly limited, but is usually 0.01 to 20 mmol, preferably 100 g per 100 g of all monomers used. Is 0.05 to 15 mmol, more preferably 0.1 to 10 mmol.
- the solvent used for the polymerization is not particularly limited as long as it is inert to the polymerization initiator.
- a chain hydrocarbon solvent, a cyclic hydrocarbon solvent, or a mixed solvent thereof is used.
- chain hydrocarbon solvents include n-butane, isobutane, 1-butene, isobutylene, trans-2-butene, cis-2-butene, 1-pentene, trans-2-pentene, cis-2-pentene, and n-pentane.
- Examples thereof include linear alkanes and alkenes having 4 to 6 carbon atoms such as isopentane, neo-pentane, and n-hexane.
- cyclic hydrocarbon solvent examples include aromatic compounds such as benzene, toluene and xylene; alicyclic hydrocarbon compounds such as cyclopentane and cyclohexane. These solvents may be used alone or in combination of two or more.
- the amount of the solvent used for the polymerization is not particularly limited, but the concentration of the total block copolymer in the solution after the polymerization reaction is usually 5 to 60% by weight, preferably 10 to 55% by weight, more preferably 20 to 50%. Set the weight%.
- a Lewis base compound may be added to a reactor used for polymerization in order to control the structure of each polymer block of each block copolymer.
- the Lewis base compound include ethers such as tetrahydrofuran, diethyl ether, dioxane, ethylene glycol dimethyl ether, ethylene glycol dibutyl ether, diethylene glycol dimethyl ether and diethylene glycol dibutyl ether; tetramethylethylenediamine, trimethylamine, triethylamine, pyridine, quinuclidine and the like.
- Tertiary amines Tertiary amines; alkali metal alkoxides such as potassium-t-amyl oxide and potassium-t-butyl oxide; phosphines such as triphenylphosphine; and the like.
- These Lewis base compounds are used singly or in combination of two or more, and are appropriately selected within a range that does not impair the purpose of this embodiment.
- the timing of adding the Lewis base compound during the polymerization reaction is not particularly limited, and may be appropriately determined according to the structure of each target block copolymer. For example, it may be added in advance before the polymerization is started, or may be added after polymerizing a part of the polymer block. You may add further, after superposing
- the polymerization reaction temperature is usually 10 to 150 ° C., preferably 30 to 130 ° C., more preferably 40 to 90 ° C.
- the time required for polymerization varies depending on the conditions, but is usually within 48 hours, preferably 0.5 to 10 hours.
- the polymerization pressure is not particularly limited as long as it is carried out within a range of pressure sufficient to maintain the monomer and solvent in the liquid phase within the above polymerization temperature range.
- a solution containing an aromatic vinyl polymer having an active terminal can be obtained by polymerizing an aromatic vinyl monomer using a polymerization initiator in a solvent.
- the aromatic vinyl polymer having an active end is composed of an aromatic vinyl polymer block (Ar1 a ) having a relatively small weight average molecular weight of the block copolymer A and a block copolymer.
- the aromatic vinyl polymer block (Ar b ) of the combined B will be constituted. Therefore, the amount of the aromatic vinyl monomer used at this time is determined according to the target weight average molecular weight of these polymer blocks.
- a conjugated diene monomer is added to a solution containing an aromatic vinyl polymer having an active end.
- a conjugated diene polymer chain is formed from the active end, and a solution containing an aromatic vinyl-conjugated diene block copolymer (diblock) having an active end is obtained.
- the amount of the conjugated diene monomer used here is determined so that the resulting conjugated diene polymer chain has the weight average molecular weight of the conjugated diene polymer block (D b ) of the target block copolymer B. .
- the functional group is less than 1 molar equivalent with respect to the active terminal in the solution containing the aromatic vinyl-conjugated diene block copolymer having the active terminal.
- a trifunctional or higher functional coupling agent is added in an amount.
- the coupling agent to be added is not particularly limited as long as it is a coupling agent having three or more functional groups capable of reacting with the active terminal of the polymer in one molecule (that is, a trifunctional or higher functional coupling agent). Any trifunctional or higher functional coupling agent can be used.
- trifunctional coupling agent examples include trifunctional halogenated alkanes such as trichloroethane and trichloropropane; trifunctional halogenated silanes such as methyltrichlorosilane and ethyltrichlorosilane; methyltrimethoxysilane, phenyltrimethoxysilane, And trifunctional alkoxysilanes such as phenyltriethoxysilane;
- the tetrafunctional coupling agent include tetrafunctional halogenated alkanes such as carbon tetrachloride, carbon tetrabromide, and tetrachloroethane; tetrafunctional halogenated silanes such as tetrachlorosilane and tetrabromosilane; tetramethoxysilane, Tetrafunctional alkoxysilanes such as tetraethoxysilane; tetrafunctional tin halides
- Examples of the pentafunctional or higher functional coupling agent include 1,1,1,2,2-pentachloroethane, perchloroethane, pentachlorobenzene, perchlorobenzene, octabromodiphenyl ether, decabromodiphenyl ether, and the like.
- a tetrafunctional coupling agent is preferably used, and among them, a tetrafunctional halogenated silane or a tetrafunctional alkoxysilane is particularly preferably used.
- These coupling agents may be used alone or in combination of two or more.
- the amount of the trifunctional or higher functional coupling agent added is determined according to the ratio of the block copolymer A and the block copolymer B constituting the block copolymer composition, and is based on the active terminal of the polymer.
- the functional group is not particularly limited as long as it is less than 1 molar equivalent, but is usually in the range where the functional group of the coupling agent is 0.15 to 0.90 molar equivalent to the active terminal of the polymer. A range of 0.20 to 0.70 molar equivalent is preferable.
- the amount of functional groups is less than 1 molar equivalent with respect to the active end.
- the conjugated diene polymer blocks are coupled to each other. Bonded through the residue, resulting in the formation of block copolymer B of the block copolymer composition. The remaining part of the aromatic vinyl-conjugated diene block copolymer having an active terminal (diblock body) remains in the solution without being reacted.
- bifunctional coupling agents include bifunctional halogenated silanes such as dichlorosilane, monomethyldichlorosilane, and dimethyldichlorosilane; bifunctional halogenated alkanes such as dichloroethane, dibromoethane, methylene chloride, and dibromomethane; dichlorotin And bifunctional tin halides such as monomethyldichlorotin, dimethyldichlorotin, monoethyldichlorotin, diethyldichlorotin, monobutyldichlorotin, and dibutyldichlorotin.
- bifunctional halogenated silanes such as dichlorosilane, monomethyldichlorosilane, and dimethyldichlorosilane
- bifunctional halogenated alkanes such as dichloroethane, dibromoethane, methylene chloride, and dibromomethane
- an aromatic vinyl monomer is added to the solution obtained as described above.
- an aromatic vinyl monomer is added to the solution, the aromatic vinyl polymer from the end of the aromatic vinyl-conjugated diene block copolymer (diblock) having an active terminal remaining without reacting with the coupling agent A chain is formed.
- This aromatic vinyl polymer chain constitutes an aromatic vinyl polymer block (Ar2 a ) having a relatively large weight average molecular weight of the block copolymer A constituting the block copolymer composition. It is. Therefore, the amount of the aromatic vinyl monomer used at this time is determined in accordance with the target weight average molecular weight of the aromatic vinyl polymer block (Ar2 a ).
- an asymmetric aromatic vinyl-conjugated diene-aromatic vinyl block copolymer that forms the block copolymer A is formed, and as a result, the block copolymer is formed.
- a solution containing the polymer A and the block copolymer B is obtained.
- a solution containing an aromatic vinyl-conjugated diene block copolymer having an active terminal that did not react with the coupling agent (diblock body) was conjugated.
- a diene monomer may be added before the step of adding the aromatic vinyl monomer.
- the weight average molecular weight of the conjugated diene polymer block (D a ) of the block copolymer A can be increased as compared with the case where the conjugated diene monomer is not added.
- a polymerization terminator water, methanol, etc.
- water, methanol, etc. is added to a solution containing an aromatic vinyl-conjugated diene block copolymer having an active end that has not reacted with the coupling agent in an amount less than the equivalent of the active end. Also good.
- the active terminal of the aromatic vinyl-conjugated diene block copolymer (diblock body) is deactivated, and the resulting aromatic vinyl-conjugated diene block copolymer (diblock) Body) will be contained in the block copolymer composition.
- the target block copolymer composition is recovered from the solution containing the block copolymer A and the block copolymer B obtained as described above.
- the recovery method may be any conventional method and is not particularly limited. For example, after completion of the reaction, if necessary, a polymerization terminator such as water, methanol, ethanol, propanol, hydrochloric acid, citric acid is added, and if necessary, an additive such as an antioxidant is added.
- the solution can be recovered by directly applying a known method such as a drying method or steam stripping to the solution.
- the block copolymer composition When the block copolymer composition is recovered as a slurry by applying steam stripping or the like, it is dewatered using an arbitrary dehydrator such as an extruder-type squeezer, and a crumb having a moisture content of a predetermined value or less.
- the crumb may be dried using any dryer such as a band dryer or an expansion extrusion dryer.
- the block copolymer composition obtained as described above may be used after being processed into a pellet shape or the like according to a conventional method.
- the block copolymer A and the block copolymer B can be obtained continuously in the same reaction vessel.
- the target block copolymer composition can be obtained with extremely excellent productivity as compared with the case of individually producing and mixing.
- the weight average molecular weight of each polymer block of each block copolymer has a particularly desirable balance as the block copolymer composition of this embodiment, the obtained block copolymer composition has a high elastic modulus and The composition is highly balanced with small permanent elongation.
- block copolymer composition of this embodiment is not particularly limited.
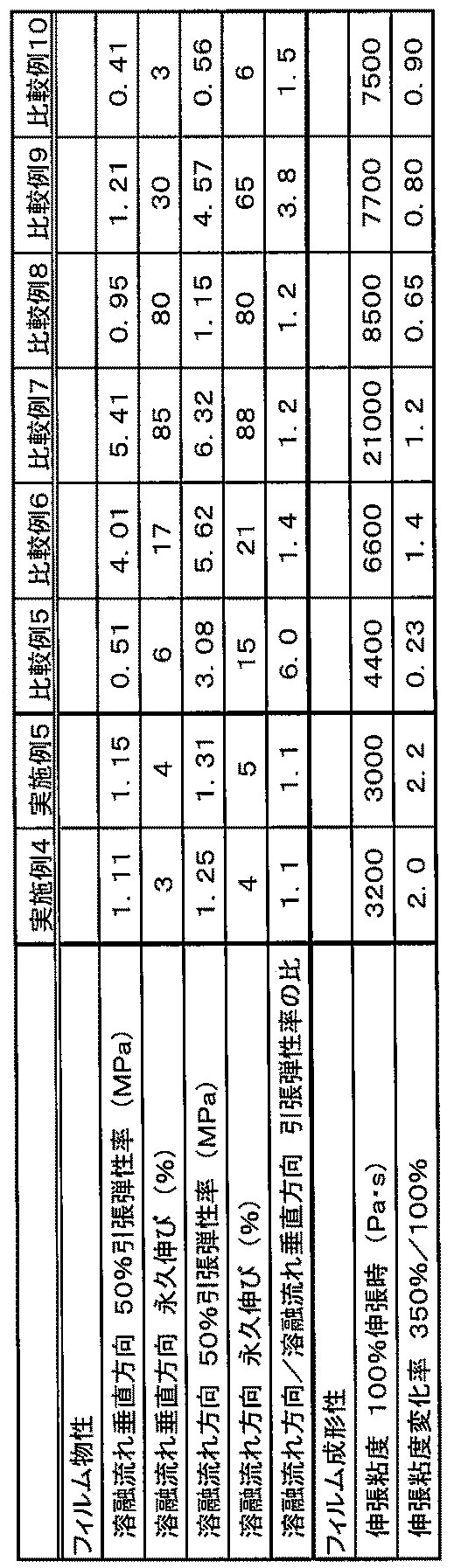
- the block copolymer composition of the present embodiment has both a high elastic modulus and a small permanent elongation, and the elastic modulus hardly changes even when repeatedly stretched. It is particularly preferably used as a material for stretchable films used for sanitary goods such as.
- the method for molding the block copolymer composition of this embodiment into a film is not particularly limited, and a conventionally known film molding method can be applied. From the viewpoint of obtaining a smooth film with good productivity, extrusion molding is possible. Among them, extrusion using a T-die is particularly preferable. As a specific example of extrusion molding using a T-die, a block copolymer composition melted at a temperature of 150 to 250 ° C. is extruded from a T-die mounted on a single screw extruder or a twin screw extruder, and a take-off roll A method of winding while cooling is mentioned. When cooling with a take-up roll, the film may be stretched.
- the film when it is wound, it may be formed into a film while coating the melt of the block copolymer composition on a substrate made of polyethylene terephthalate, polyethylene, polypropylene, nonwoven fabric or release paper, The melt of the combined composition may be formed into a film so as to be sandwiched between these substrates.
- the film thus obtained may be used as it is in an integrated form with the substrate or may be used after being peeled off from the substrate.
- the block copolymer composition of the present embodiment has a homogeneous mechanical property over all plane directions even when a molding method such as extrusion molding that tends to cause molecular orientation is applied. It gives a highly functional film.
- the block copolymer composition of the present embodiment when it is not desired that the obtained film is imparted with anisotropy based on molecular orientation (for example, stretchable used for sanitary products such as paper diapers and sanitary products).
- anisotropy based on molecular orientation for example, stretchable used for sanitary products such as paper diapers and sanitary products.
- molecular orientation for example, stretchable used for sanitary products such as paper diapers and sanitary products.
- the thickness of the film obtained from the block copolymer composition of the present embodiment is adjusted according to the use, but in the case of a film for sanitary products such as paper diapers and sanitary products, usually 0.01 to 50 mm, The thickness is preferably 0.03 to 1 mm, more preferably 0.05 to 0.5 mm.
- the film obtained from the block copolymer composition of this embodiment can be used by being laminated with other members.
- a hot melt adhesive or the like is applied to the film to form a tape, and the nonwoven fabric, woven fabric, plastic film, Alternatively, a stretchable gather member can be formed by adhering to these laminates and reducing the shrinkage of the tape.
- elastic properties such as elastic ship base material, gloves, surgical gloves, finger sack, hemostatic band, contraceptive device, headband, goggles band, rubber band, etc. It can be used as a member.
- the block copolymer composition of this embodiment contains at least three types of block copolymers.
- the block copolymer P which is one of the three types of block copolymers constituting the block copolymer composition of this embodiment, is represented by the following general formula (P) and has different weight average molecular weights.
- Ar1 p is an aromatic vinyl polymer block having a weight average molecular weight of 6000 to 20000
- Ar2 p is an aromatic vinyl polymer block having a weight average molecular weight of 40000 to 400,000
- D p is a conjugated diene polymer block having a vinyl bond content of 1 to 20 mol%.
- the block copolymer Q which is another one of the three block copolymers constituting the block copolymer composition of this embodiment, is a linear block represented by the following general formula (Q) It is a copolymer.
- Ar q is an aromatic vinyl polymer block having a weight average molecular weight of 6000 to 20000
- D q is a conjugated diene weight having a vinyl bond content of 1 to 20 mol%, respectively. It is a combined block
- Xq is a single bond or a residue of a coupling agent.
- the block copolymer R which is still another type of the three block copolymers constituting the block copolymer composition of this embodiment, is a branched block represented by the following general formula (R): It is a copolymer.
- Ar r is an aromatic vinyl polymer block having a weight average molecular weight of 6000 to 20000, and D r is a conjugated diene polymer having a vinyl bond content of 1 to 20 mol%, respectively. It is a combined block, Xr is a residue of a coupling agent, and m is an integer of 3 or more.
- the aromatic vinyl polymer block (Ar1 p , Ar2 p , Ar q , Ar r ) of the block copolymer P, the block copolymer Q, and the block copolymer R is composed of aromatic vinyl monomer units. This is a polymer block.
- the aromatic vinyl monomer used to constitute the aromatic vinyl monomer unit of the aromatic vinyl polymer block may be the same as that described in the section “1. First aspect” above. it can.
- the aromatic vinyl polymer blocks (Ar1 p , Ar2 p , Ar q , Ar r ) of the block copolymer P, the block copolymer Q, and the block copolymer R are each other than an aromatic vinyl monomer unit.
- the monomer unit may be included.
- the monomer constituting the monomer unit other than the aromatic vinyl monomer unit that can be contained in the aromatic vinyl polymer block and the content thereof those described in the above section “1. First embodiment” The same can be said.
- the block copolymer P, block copolymer Q, and block copolymer R conjugated diene polymer blocks (D p , D q , D r ) are polymer blocks composed of conjugated diene monomer units. is there.
- the conjugated diene monomer used for constituting the conjugated diene monomer unit of the conjugated diene polymer block can be the same as that described in the section “1. First aspect”.
- the block copolymer P, the block copolymer Q, and the conjugated diene polymer block (D p , D q , D r ) of the block copolymer R are monomer units other than the conjugated diene monomer unit, respectively. May be included.
- the monomer constituting the monomer unit other than the conjugated diene monomer unit that can be contained in the conjugated diene polymer block and the content thereof are the same as those described in the above section “1. First embodiment”. can do.
- the block copolymer P constituting the block copolymer composition of this embodiment is an aromatic vinyl polymer block (Ar1 p ) having a relatively small weight average molecular weight as represented by the general formula (P).
- a conjugated diene polymer block (D p ) having a specific vinyl bond content and an aromatic vinyl polymer block (Ar 2 p ) having a relatively large weight average molecular weight are linearly linked in this order.
- the aromatic vinyl polymer block (Ar1 p ) having a relatively small weight average molecular weight has a weight average molecular weight (Mw (Ar1 p )) of 6000 to 20000, preferably 7000 to 18000, preferably 8000 to 15000. More preferably. If Mw (Ar1 p ) is outside this range, the resulting composition may have insufficient permanent elongation.
- the aromatic vinyl polymer block (Ar2 p ) having a relatively large weight average molecular weight has a weight average molecular weight (Mw (Ar2 p )) of 40,000 to 400,000, preferably 42,000 to 370000, preferably 45,000 to More preferably, it is 350,000.
- Mw (Ar2 p) is too small, the elongation of the resulting composition is insufficient, Mw (Ar2 p) is a block copolymer P is too large, there is a case preparation is difficult.
- the ratio (Mw (Ar2 p ) / Mw (Ar1 p )) to the weight average molecular weight (Mw (Ar1 p )) of the combined block (Ar1 p ) is not particularly limited, but is usually 2 to 67, and 4 to 40 Preferably, it is 4.5 to 35.
- the vinyl bond content of the conjugated diene polymer block (D p ) of the block copolymer P is as described above. It can be the same as the conjugated diene polymer block (D a ) of the block copolymer A described in the section of “1. First embodiment”.
- the weight average molecular weight (Mw (D p )) of the conjugated diene polymer block (D p ) of the block copolymer P is not particularly limited, but is usually 20,000 to 200,000, preferably 35,000 to 150,000, preferably 45,000. More preferably, it is ⁇ 100,000.
- the content of the aromatic vinyl monomer unit with respect to all the monomer units of the block copolymer P and the weight average molecular weight of the block copolymer P as a whole are the blocks described in the section “1. First aspect” above. It can be the same as the copolymer A.
- the block copolymer Q constituting the block copolymer composition of the present embodiment includes an aromatic vinyl polymer block (Ar q ) having a specific weight average molecular weight, as represented by the general formula (Q).
- An aromatic vinyl polymer block (Ar q ) having a specific weight average molecular weight as represented by the general formula (Q).
- Two diblock bodies (Ar q -D q ) formed by bonding to a conjugated diene polymer block (D q ) having a specific vinyl bond content are bonded to the aromatic vinyl polymer block (Ar q ) side at the end.
- it is a linear aromatic vinyl-conjugated diene-aromatic vinyl block copolymer formed by direct bonding or via a coupling agent residue.
- the weight average molecular weights (Mw (Ar q )) of the two aromatic vinyl polymer blocks (Ar q ) included in the block copolymer Q are 6000 to 20000, preferably 7000 to 18000, respectively. It is more preferably 8000 to 15000. If Mw (Ar q ) is out of this range, the resulting composition may have insufficient permanent elongation.
- the weight average molecular weights (Mw (Ar q )) of the two aromatic vinyl polymer blocks present in one molecule may be equal to or different from each other as long as they are within the above range. Are preferably equal.
- the weight average molecular weight (Mw (Ar q )) of the plurality of aromatic vinyl polymer blocks is the weight of the aromatic vinyl polymer block (Ar1 p ) having a relatively small weight average molecular weight of the block copolymer P. average molecular weight (Mw (Ar1 p)), and more preferably substantially equal.
- the conjugated diene polymer block (D q ) of the block copolymer Q As the vinyl bond content of the conjugated diene polymer block (D q ) of the block copolymer Q, the conjugated diene polymer block (D b ) of the block copolymer B described in the section “1. First aspect” above is used. ).
- the vinyl bond content of the conjugated diene polymer block (D q ) of the block copolymer Q is substantially equal to the vinyl bond content of the conjugated diene polymer block (D p ) of the block copolymer P. Is preferred.
- the weight average molecular weight (Mw (D q )) of the conjugated diene polymer block (D q ) of the block copolymer Q is not particularly limited, but is usually 20,000 to 200,000, preferably 35,000 to 150,000, preferably 45,000. More preferably, it is ⁇ 100,000. When the weight average molecular weight (Mw ( Dq )) is within this range, a block copolymer composition having a lower permanent elongation and a higher elastic modulus and rich in elasticity can be obtained.
- the weight average molecular weight (Mw (D q )) of the conjugated diene polymer block (D q ) of the block copolymer Q is the weight average molecular weight of the conjugated diene polymer block (D p ) of the block copolymer P ( It is preferred that it is substantially equal to Mw ( Dp )).
- Mw ( D q ) The weight average molecular weight of the conjugated diene polymer block (D q ) of the block copolymer Q. It is preferred that it is substantially equal to Mw ( Dp )).
- the resulting block copolymer composition has a higher elastic modulus and is rich in elasticity.
- the content of the aromatic vinyl monomer unit with respect to all the monomer units of the block copolymer Q can be the same as that of the block copolymer B described in the above section “1. First embodiment”.
- the weight average molecular weight of the block copolymer Q as a whole is not particularly limited, but is usually 60000-400000, preferably 80000-300000, and more preferably 100000-200000.
- the block copolymer R constituting the block copolymer composition of the present embodiment includes an aromatic vinyl polymer block (Ar r ) having a specific weight average molecular weight, as represented by the general formula (R).
- a diblock body (Ar r -D r ) formed by bonding with a conjugated diene polymer block (D r ) having a specific vinyl bond content is terminated at the aromatic vinyl polymer block (Ar r ) side.
- the weight average molecular weights (Mw (Ar r )) of the plurality of aromatic vinyl polymer blocks (Ar r ) included in the block copolymer R are 6000 to 20000, preferably 7000 to 18000, respectively. More preferably, it is ⁇ 15000. If Mw (Ar r ) is out of this range, the resulting composition may have insufficient permanent elongation.
- the weight average molecular weight (Mw (Ar r )) of the aromatic vinyl polymer block present in three or more in one molecule may be equal or different from each other as long as it is within the above range. Preferably they are substantially equal.
- the weight average molecular weight (Mw (Ar r )) of the plurality of aromatic vinyl polymer blocks is the weight of the aromatic vinyl polymer block (Ar1 p ) having a relatively small weight average molecular weight of the block copolymer P.
- average molecular weight (Mw (Ar1 p)) and a weight average molecular weight of the aromatic vinyl polymer block (Ar q) of the block copolymer Q (Mw (Ar q)) is preferably equal at least to one substantially, these More preferably, both are substantially equal.
- the vinyl bond content of the conjugated diene polymer block (D r ) of the block copolymer R is the conjugated diene polymer block (D b ) of the block copolymer B described in the section “1. First aspect”. And can be similar. Further, the vinyl bond content of the conjugated diene polymer block (D r ) of the block copolymer R is such that the conjugated diene polymer block (D p ) of the block copolymer P and the conjugated diene polymer of the block copolymer Q It is preferable that the vinyl bond content of at least one of the blocks (D q ) is substantially equal, and it is more preferable that the content of both vinyl bonds is substantially equal.
- the weight average molecular weight (Mw (D r )) of the conjugated diene polymer block (D r ) of the block copolymer R is not particularly limited, but is usually 20000-200000, preferably 35000-150,000, preferably 45000. More preferably, it is ⁇ 100,000. When the weight average molecular weight (Mw (D r )) is within this range, a block copolymer composition having a lower permanent elongation and a higher elastic modulus and rich in elasticity can be obtained.
- the weight average molecular weight (Mw (D r )) of the conjugated diene polymer block (D r ) of the block copolymer R is the weight average molecular weight of the conjugated diene polymer block (D p ) of the block copolymer P ( Mw (D p )) and at least one of the weight average molecular weights (Mw (D q )) of the conjugated diene polymer block (D q ) of the block copolymer Q are preferably substantially equal to both It is more preferable that they are equal.
- the content of the aromatic vinyl monomer unit with respect to the total monomer units of the block copolymer R is not particularly limited, but is usually 10 to 35% by weight, preferably 12 to 32% by weight, More preferably, it is ⁇ 30% by weight.
- the content of the aromatic vinyl monomer unit relative to the total monomer units of the block copolymer R is substantially equal to the content of the aromatic vinyl monomer unit relative to the total monomer units of the block copolymer Q.
- the weight average molecular weight of the block copolymer R as a whole is also not particularly limited, but is usually from 80,000 to 800,000, preferably from 100,000 to 600,000, and more preferably from 120,000 to 400,000.
- the molecular weight distribution represented by the ratio (Mw / Mn) to (Mn) is not particularly limited, but is usually 1.1 or less, preferably 1.05 or less.
- the weight ratio (P / (Q + R)) between the amount of the block copolymer P contained in the block copolymer composition of the present embodiment and the total amount of the block copolymer Q and the block copolymer R is, in particular, Although not limited, it is preferably 10/90 to 80/20, more preferably 36/64 to 80/20, still more preferably 38/62 to 80/20, and 40/60 to 75. Most preferred is / 25.
- the balance between the high elastic modulus and the small permanent elongation in the block copolymer composition becomes particularly excellent.
- this ratio is too small, the elastic modulus of the block copolymer composition may be insufficient, and if this ratio is too large, the permanent elongation of the block copolymer composition may be too large. .
- the weight ratio (Q / R) between the amount of the block copolymer Q and the amount of the block copolymer R contained in the block copolymer composition of the present embodiment is not particularly limited, and may be any value. However, it is preferably 15/85 to 85/15, more preferably 17/83 to 83/17, and still more preferably 20/80 to 80/20.
- the block copolymer Q and the block copolymer R in such a ratio the moldability of the block copolymer composition into a film or the like becomes particularly good, and the block copolymer composition
- a molding method such as extrusion molding in which molecular orientation is likely to occur is applied, a molded body having particularly high isotropic properties is obtained.
- the block copolymer composition of this embodiment may contain only the block copolymers P to R as polymer components, but it contains polymer components other than the block copolymers P to R. Also good.
- the polymer component other than the block copolymers P to R that can be included in the block copolymer composition of this embodiment include an aromatic vinyl-conjugated diene-aromatic vinyl block copolymer other than the block copolymers P to R.
- polymers aromatic vinyl-conjugated diene block copolymers, aromatic vinyl homopolymers, conjugated diene homopolymers, aromatic vinyl-conjugated diene random copolymers, and branched polymers thereof, or polyurethane heat
- Thermoplastic elastomers such as plastic elastomer, polyamide thermoplastic elastomer, polyester thermoplastic elastomer, and thermoplastics such as polyethylene, polypropylene, polyvinyl chloride, acrylonitrile-styrene copolymer, acrylonitrile-butadiene-styrene copolymer, polyphenylene ether Resin etc. are mentioned.
- the content of the polymer components other than the block copolymers P to R is preferably 20% by weight or less, and preferably 10% by weight or less based on the entire polymer component. It is more preferable that
- the proportion of the aromatic vinyl monomer unit in the total repeating units of the entire polymer component contained is the block copolymer described in the section “1. First embodiment” above. It can be the same as the polymer composition.
- the total aromatic vinyl monomer unit content is determined based on the block copolymer P, block copolymer Q, block copolymer R, and other polymer components constituting the block copolymer composition. It is possible to easily adjust the content of the aromatic vinyl monomer unit by taking the content of the aromatic vinyl monomer unit into consideration and adjusting the blending amount thereof.
- the weight average molecular weight and the molecular weight distribution of the entire polymer components constituting the block copolymer composition of this embodiment can be the same as those described in the above section “1. First embodiment”.
- melt index of the block copolymer composition of the present embodiment can be the same as that described in the above section “1. First embodiment”.
- the block copolymer composition of this embodiment may contain a component other than the polymer component, and as a component other than such a polymer component, those described in the above section “1. First embodiment” And can be similar.
- the method for obtaining the block copolymer composition of this embodiment is not particularly limited.
- block copolymer P, block copolymer Q, and block copolymer R are produced separately, and if necessary, other polymer components and various additions It can be manufactured by blending agents and mixing them according to conventional methods such as kneading and solution mixing.
- the method for producing the block copolymer composition of the present embodiment described below is preferable.
- the manufacturing method of the block copolymer composition of this embodiment includes the following steps (6) to (10).
- an aromatic vinyl monomer is polymerized using a polymerization initiator in a solvent.
- the polymerization initiator used, the amount used, the solvent used in the polymerization, and the amount thereof can be the same as those described in the above section “1. First embodiment”.
- a Lewis base compound may be added to a reactor used for polymerization in order to control the structure of each polymer block of each block copolymer.
- the Lewis base compound and the timing of its addition can be the same as those described in the above section “1. First embodiment”.
- the polymerization reaction temperature may be the same as that described in the above section “1. First aspect”.
- a solution containing an aromatic vinyl polymer having an active terminal can be obtained by polymerizing an aromatic vinyl monomer using a polymerization initiator in a solvent.
- the aromatic vinyl polymer having an active end is an aromatic vinyl polymer block (Ar1 p ) having a relatively small weight average molecular weight of the block copolymer P, which constitutes the block copolymer composition,
- the aromatic vinyl polymer block (Ar q ) of the coalescence Q and the aromatic vinyl polymer block (Ar r ) of the block copolymer R are constituted. Therefore, the amount of the aromatic vinyl monomer used at this time is determined according to the target weight average molecular weight of these polymer blocks.
- a conjugated diene monomer is added to a solution containing an aromatic vinyl polymer having an active end.
- a conjugated diene polymer chain is formed from the active end, and a solution containing an aromatic vinyl-conjugated diene block copolymer (diblock) having an active end is obtained.
- the amount of the conjugated diene monomer used here is such that the resulting conjugated diene polymer chain is a conjugated diene polymer block (D q ) of the target block copolymer Q or a conjugated diene polymer of the block copolymer R. Determined to have a weight average molecular weight of the block (D r ).
- the bifunctional coupling agent and the trifunctional or higher functional group are added to the solution containing the aromatic vinyl-conjugated diene block copolymer having an active terminal (diblock body).
- Add the coupling agent in this case, the amount of the coupling agent added is such that the functional group of the coupling agent with respect to the active terminal of the aromatic vinyl-conjugated diene block copolymer (diblock) having an active terminal contained in the solution.
- the total amount of the (functional group capable of reacting with the active terminal of the polymer) is less than 1 molar equivalent.
- the bifunctional coupling agent added in this step is not particularly limited as long as it has two functional groups capable of reacting with the active terminal of the polymer in one molecule.
- dichlorosilane, monomethyldichlorosilane Bifunctional halogenated silanes such as dimethyldichlorosilane
- bifunctional alkoxysilanes such as diphenyldimethoxysilane and diphenyldiethoxysilane
- bifunctional halogenated alkanes such as dichloroethane, dibromoethane, methylene chloride, and dibromomethane
- dichlorotin, Bifunctional tin halides such as monomethyldichlorotin, dimethyldichlorotin, monoethyldichlorotin, diethyldichlorotin, monobutyldichlorotin, dibutyldichlorotin; using dibromobenzene, benzoic acid, CO, 2-chlor
- the type of the trifunctional or higher functional coupling agent added in this step is not particularly limited as long as it has three or more functional groups capable of reacting with the active terminal of the polymer in one molecule.
- any trifunctional or higher functional coupling agent can be used.
- the trifunctional coupling agent may be the same as that described in the above section “1. First aspect”.
- the order in which these coupling agents are added is not particularly limited, and after adding either one, the other may be added. Both may be added simultaneously.
- the total amount of the bifunctional coupling agent and the trifunctional or higher functional coupling agent added is the amount of the block copolymer P in the target block copolymer composition, the block copolymer Q and the block copolymer.
- the active terminal of the aromatic vinyl-conjugated diene block copolymer (diblock body) having an active terminal contained in the solution may be determined according to the weight ratio (P / (Q + R)) to the total amount of R.
- the total amount of functional groups of the coupling agent is less than 1 molar equivalent, but usually the total amount of functional groups of the coupling agent is 0.15 with respect to the active terminal of the polymer.
- the range is from 0.90 to 0.90 molar equivalent, and the range from 0.20 to 0.70 molar equivalent is preferred.
- the amount ratio of the bifunctional coupling agent to be added and the trifunctional or higher functional coupling agent is such that the amount of the block copolymer Q and the amount of the block copolymer R in the target block copolymer composition are as follows.
- the molar ratio of (functional group of bifunctional coupling agent / functional group of trifunctional or higher functional coupling agent) is 0.17. Is preferably in the range of ⁇ 5.7, and more preferably in the range of 0.25 to 4.0.
- the total amount of functional groups of the coupling agent with respect to the active terminal is less than 1 molar equivalent.
- a bifunctional coupling agent and a trifunctional or higher functional coupling agent are added in such an amount, a part of the aromatic vinyl-conjugated diene block copolymer having an active terminal (diblock body) becomes a conjugated diene.
- the polymer blocks are bonded to each other through the residue of each coupling agent, so that a block copolymer Q and a block copolymer R are formed.
- the remaining part of the aromatic vinyl-conjugated diene block copolymer having an active terminal (diblock body) remains in the solution without being reacted.
- an aromatic vinyl monomer is added to the solution obtained as described above.
- an aromatic vinyl monomer is added to the solution, the aromatic vinyl polymer from the end of the aromatic vinyl-conjugated diene block copolymer (diblock) having an active terminal remaining without reacting with the coupling agent A chain is formed.
- This aromatic vinyl polymer chain constitutes an aromatic vinyl polymer block (Ar2 p ) having a relatively large weight average molecular weight of the block copolymer P constituting the block copolymer composition. It is. Therefore, the amount of the aromatic vinyl monomer used at this time is determined according to the target weight average molecular weight of the aromatic vinyl polymer block (Ar2 p ).
- an asymmetric aromatic vinyl-conjugated diene-aromatic vinyl block copolymer constituting the block copolymer P is formed.
- the block copolymer is formed.
- a solution containing the polymers P to R is obtained.
- a solution containing an aromatic vinyl-conjugated diene block copolymer having an active terminal that did not react with the coupling agent (diblock body) was conjugated.
- a diene monomer may be added before the step of adding the aromatic vinyl monomer.
- the weight average molecular weight of the conjugated diene polymer block (D p ) of the block copolymer P can be increased as compared with the case where the conjugated diene monomer is not added.
- a polymerization terminator water, methanol, phenol, etc. is added to the solution containing an aromatic vinyl-conjugated diene block copolymer having an active end that has not reacted with the coupling agent in an amount less than the equivalent of the active end. May be.
- the active terminal of the aromatic vinyl-conjugated diene block copolymer (diblock body) is deactivated, and the resulting aromatic vinyl-conjugated diene block copolymer (diblock) Body) will be contained in the block copolymer composition.
- the target block copolymer composition is recovered from the solution containing the block copolymers P to R obtained as described above.
- the collection method may be in accordance with a conventional method, and may be the same as that described in the above section “1. First embodiment”.
- each block copolymer is produced separately and mixed.
- the target block copolymer composition can be obtained with extremely excellent productivity.
- the weight average molecular weight of each polymer block of each block copolymer has a particularly desirable balance as the block copolymer composition of this embodiment, the obtained block copolymer composition has a high elastic modulus and The composition is highly balanced with small permanent elongation.
- block copolymer composition of the present embodiment is not particularly limited, and can be the same as that described in the above section “1. First embodiment”.
- the method for forming the block copolymer composition of this embodiment into a film is not particularly limited, and a conventionally known film forming method can be applied, which is the same as that described in the above section “1. First embodiment”. be able to.
- the thickness of the film obtained from the block copolymer composition of the present embodiment is adjusted according to the application, but can be the same as that described in the section “1. First embodiment” above.
- the film obtained from the block copolymer composition of this embodiment can be used by being laminated with other members.
- it may be the same as that described in the section “1. First aspect”.
- the apparatus is an HLC8220 manufactured by Tosoh Corporation, the column is a combination of three Shodex KF-404HQ manufactured by Showa Denko (column temperature 40 ° C.), the detector is a differential refractometer and an ultraviolet detector, and the molecular weight is calibrated by a polymer laboratory. The test was carried out using 12 standard polystyrenes (500 to 3 million).
- reaction was completed by introducing a gas flowing out of the reaction vessel into the potassium iodide aqueous solution.
- 50 ml of diethyl ether and 470 mg of lithium aluminum hydride were charged into another reaction vessel purged with nitrogen, and the solution reacted with ozone was slowly added dropwise to the reaction vessel while cooling the reaction vessel with ice water.
- the reaction vessel was placed in a water bath, gradually heated, and refluxed at 40 ° C. for 30 minutes. Thereafter, dilute hydrochloric acid was added dropwise to the reaction vessel little by little while stirring the solution, and the addition was continued until almost no generation of hydrogen was observed.
- the solid product formed in the solution was filtered off, and the solid product was extracted with 100 ml of diethyl ether for 10 minutes.
- the extract and the filtrate obtained by filtration were combined and the solvent was distilled off to obtain a solid sample.
- the weight average molecular weight was measured according to the above-described method for measuring the weight average molecular weight, and the value was taken as the weight average molecular weight of the styrene polymer block.
- Weight average molecular weight of isoprene polymer block The weight average molecular weight of the isoprene polymer block was determined based on the calculated value by subtracting the weight average molecular weight of the corresponding styrene polymer block from the weight average molecular weight of the block copolymer obtained as described above. .
- the tensile modulus of the film at 50% elongation was determined and recorded as the tensile modulus at the second elongation. It can be said that the higher the elastic modulus, the higher the elastic modulus, and the closer the ratio of (tensile elastic modulus in the melt flow direction / tensile elastic modulus in the vertical direction of the melt flow) to 1, the anisotropy of the tensile elastic modulus. It can be said that the ratio of (tensile elastic modulus of the first measurement / tensile elastic modulus of the second measurement) is closer to 1, the smaller the change in the elastic modulus due to expansion and contraction.
- the extension viscosity at 100% extension and 350% extension of the film was measured. If the elongation viscosity at 100% elongation is too high, the moldability is inferior, and the elongation viscosity at 350% elongation is lower than the 100% elongation viscosity (350% elongation viscosity / 100% elongation). When the viscosity value is less than 1, it can be said that the molding stability is poor.
- Example 1 To the pressure resistant reactor, 23.3 kg of cyclohexane, 5.10 mmol of N, N, N ′, N′-tetramethylethylenediamine (hereinafter referred to as TMEDA) and 1.60 kg of styrene were added and stirred at 40 ° C. Thereto was added 169.9 mmol of n-butyllithium, and polymerization was carried out for 1 hour while raising the temperature to 50 ° C. The polymerization conversion of styrene was 100%. Subsequently, 5.20 kg of isoprene was continuously added to the reactor over 1 hour while controlling the temperature so as to maintain 50 to 60 ° C.
- TMEDA N, N, N ′, N′-tetramethylethylenediamine
- Example 1 To 100 parts of the reaction solution obtained as described above, 0.3 part of 2,6-di-tert-butyl-p-cresol as an antioxidant was added and mixed, and the mixed solution was added in small portions from 85 to 95.
- the block copolymer composition of Example 1 was recovered by dropping into warm water heated to ° C to volatilize the solvent to obtain a precipitate, pulverizing the precipitate, and drying with hot air at 85 ° C. did.
- the obtained composition was supplied to a single-screw extruder equipped with an underwater hot cut device at the tip of the extruder to obtain cylindrical pellets having an average diameter of 5 mm and an average length of about 5 mm.
- Example 1 block copolymer composition pellets of Example 1 were melted by heating at 200 ° C. using a twin-screw extruder equipped with a T-die and continuously extruded for 20 minutes to obtain a thickness of 0. Molded into a 2 mm film.
- the film of Example 1 was measured for tensile modulus and permanent elongation. The results are shown in Table 2. The details of the film forming conditions are as follows.
- Composition processing speed 15 kg / hr Film take-off speed: 10 m / min
- Extruder temperature adjusted to 140 ° C. at the inlet and 160 ° C.
- T-die Screw full flight
- Extruder L / D 42
- T-die width 300mm, lip 1mm
- Example 2 To the pressure reactor, 23.3 kg of cyclohexane, 5.56 mmol of TMEDA and 1.55 kg of styrene were added, and 185.0 mmol of n-butyllithium was added to the mixture while stirring at 40 ° C., and the temperature was raised to 50 ° C. The polymerization was carried out for 1 hour. The polymerization conversion of styrene was 100%. Subsequently, 5.20 kg of isoprene was continuously added to the reactor over 1 hour while controlling the temperature so as to maintain 50 to 60 ° C. After completing the addition of isoprene, polymerization was continued for an additional hour. The polymerization conversion rate of isoprene was 100%.
- Example 3 The block copolymer composition of Example 3 was prepared in the same manner as in Example 1 except that the amounts of styrene, n-butyllithium, TMEDA, isoprene, tetrachlorosilane and methanol were changed as shown in Table 1, respectively. It collect
- Comparative Example 4 A block copolymer composition of Comparative Example 4 was prepared in the same manner as in Example 1 except that the amounts of styrene, n-butyllithium, TMEDA, isoprene, tetrachlorosilane and methanol were changed as shown in Table 1. It collect
- Table 2 shows the following. That is, the film obtained from the block copolymer composition of the present invention has both a high elastic modulus and a small permanent elongation, and also has a small anisotropy in tensile elastic modulus and a change in elastic modulus due to expansion and contraction. Small (Examples 1 to 3).
- the composition different from the composition of the block copolymer composition of the present invention when the entire styrene (aromatic vinyl) unit content is increased in order to have a high elastic modulus, the permanent elongation is large. Thus, the change in elastic modulus due to expansion and contraction is large (Comparative Examples 3 and 4).
- Example 4 To the pressure-resistant reactor, 23.3 kg of cyclohexane, 4.80 mmol of N, N, N ′, N′-tetramethylethylenediamine (hereinafter referred to as TMEDA) and 1.55 kg of styrene were added and stirred at 40 ° C. Thereto was added 160.8 mmol of n-butyllithium, and polymerization was carried out for 1 hour while raising the temperature to 50 ° C. The polymerization conversion of styrene was 100%. Subsequently, 5.20 kg of isoprene was continuously added to the reactor over 1 hour while controlling the temperature so as to maintain 50 to 60 ° C.
- TMEDA N, N, N ′, N′-tetramethylethylenediamine
- Example 4 To 100 parts of the reaction solution obtained as described above, 0.3 part of 2,6-di-tert-butyl-p-cresol as an antioxidant was added and mixed, and the mixed solution was added in small portions from 85 to 95.
- the block copolymer composition of Example 4 was recovered by dropping into warm water heated to ° C to volatilize the solvent to obtain a precipitate, pulverizing the precipitate, and drying with hot air at 85 ° C. did.
- the obtained composition was supplied to a single-screw extruder equipped with an underwater hot cut device at the tip of the extruder to obtain cylindrical pellets having an average diameter of 5 mm and an average length of about 5 mm.
- Example 4 block copolymer composition pellets of Example 4 were heated and melted at 200 ° C. using a twin-screw extruder equipped with a T-die and extruded continuously for 20 minutes to obtain a thickness of 0. Molded into a 2 mm film.
- the tensile modulus and permanent elongation of the film of Example 4 were measured, and the film moldability of the block copolymer composition of Example 4 was evaluated.
- the results are shown in Table 5. The details of the film forming conditions are as described above.
- Example 5 The block copolymer of Example 5 was the same as Example 4 except that the amounts of styrene, n-butyllithium, TMEDA, dimethyldichlorosilane, tetramethoxysilane, and methanol were changed as shown in Table 3, respectively. The combined composition was recovered and the film of Example 5 was obtained as pellets. About the block copolymer composition and film of Example 5, the same measurement as Example 4 was performed. The results are shown in Tables 4 and 5.
- Comparative Example 6 and Comparative Example 7 The block copolymer compositions of Comparative Examples 6 and 7 were the same as Comparative Example 5 except that the amounts of styrene, n-butyllithium, TMEDA, isoprene and methanol were changed as shown in Table 3, respectively. Were collected and the films of Comparative Example 6 and Comparative Example 7 were obtained as pellets. The block copolymer compositions and films of Comparative Example 6 and Comparative Example 7 were measured in the same manner as in Example 4. The results are shown in Tables 4 and 5.
- Comparative Example 9 and Comparative Example 10 The block copolymer compositions of Comparative Example 9 and Comparative Example 10 were the same as Comparative Example 8 except that the amounts of styrene, n-butyllithium, TMEDA, isoprene and methanol were changed as shown in Table 3 respectively. Were collected to obtain films of Comparative Example 9 and Comparative Example 10 as pellets. About the block copolymer composition and film of the comparative example 9 and the comparative example 10, the same measurement as Example 4 was performed. The results are shown in Tables 4 and 5.
- Table 4 and Table 5 show the following. That is, the film using the block copolymer composition of the present invention has both a high elastic modulus and a small permanent elongation, and has a small anisotropy in tensile elastic modulus, and a film moldability and a moldability. It is also excellent in stability (Examples 4 and 5).
- a composition other than the block copolymer composition of the present invention if the entire styrene (aromatic vinyl) unit content is increased in order to have a high elastic modulus, permanent elongation is increased. It becomes large (Comparative Examples 6 to 9).
- the present invention relates to elastic films, gloves, elastic bands, condoms, OA equipment, various rolls for office use, anti-vibration sheets for electrical and electronic equipment, anti-vibration rubber, shock absorbing sheets, shock absorbing films and sheets, and residential use. Molding materials used for damping sheets, damping damper materials, etc., adhesive tapes, adhesive sheets, adhesive labels, adhesives used for dust removal rollers, adhesives used for sanitary goods and bookbinding, clothing, sports It can be used for applications such as elastic fiber used for articles.
- the block copolymer composition of the present invention has both a high elastic modulus and a small permanent elongation, and the elastic modulus hardly changes even when repeatedly expanded and contracted, a disposable diaper or a sanitary product It is particularly preferably used as a material for stretchable films used for sanitary goods such as.
Abstract
Description
(Arb-Db)n-X (B)
(一般式(A)および(B)において、Ar1aおよびArbは、それぞれ、重量平均分子量が6000~15000の芳香族ビニル重合体ブロックであり、Ar2aは、重量平均分子量が40000~400000の芳香族ビニル重合体ブロックであり、DaおよびDbは、それぞれ、ビニル結合含有量が1~20モル%の共役ジエン重合体ブロックであり、Xはカップリング剤の残基であり、nは3以上の整数である。)
(1):溶媒中で重合開始剤を用いて、芳香族ビニル単量体を重合する工程
(2):上記(1)の工程で得られる活性末端を有する芳香族ビニル重合体を含有する溶液に、共役ジエン単量体を添加する工程
(3):上記(2)の工程で得られる活性末端を有する芳香族ビニル-共役ジエンブロック共重合体を含有する溶液に、その活性末端に対して官能基が1モル当量未満となる量で、3官能以上のカップリング剤を添加し、ブロック共重合体Bを形成する工程
(4):上記(3)の工程で得られる溶液に、芳香族ビニル単量体を添加し、ブロック共重合体Aを形成する工程
(5):上記(4)の工程で得られる溶液から、ブロック共重合体組成物を回収する工程
(Arq-Dq)2-Xq (Q)
(Arr-Dr)m-Xr (R)
(6):溶媒中で重合開始剤を用いて、芳香族ビニル単量体を重合する工程
(7):上記(6)の工程で得られる活性末端を有する芳香族ビニル重合体を含有する溶液に、共役ジエン単量体を添加する工程
(8):上記(7)の工程で得られる活性末端を有する芳香族ビニル-共役ジエンブロック共重合体を含有する溶液に、その活性末端に対して官能基の総量が1モル当量未満となる量で、2官能のカップリング剤、および3官能以上のカップリング剤を添加し、ブロック共重合体Q、およびブロック共重合体Rを形成する工程
(9):上記(8)の工程で得られる溶液に、芳香族ビニル単量体を添加し、ブロック共重合体Pを形成する工程
(10):上記(9)の工程で得られる溶液から、ブロック共重合体組成物を回収する工程
まず、本発明のブロック共重合体組成物の第1態様について説明する。本態様のブロック共重合体組成物は、2種のブロック共重合体を含有してなるものである。本態様のブロック共重合体組成物を構成する2種のブロック共重合体の一方であるブロック共重合体Aは、下記の一般式(A)で表される、互いに異なる重量平均分子量を持つ2つの芳香族ビニル重合体ブロックを有する、直鎖状の芳香族ビニル-共役ジエン-芳香族ビニルブロック共重合体である。
(2):上記(1)の工程で得られる活性末端を有する芳香族ビニル重合体を含有する溶液に、共役ジエン単量体を添加する工程
(3):上記(2)の工程で得られる活性末端を有する芳香族ビニル-共役ジエンブロック共重合体を含有する溶液に、その活性末端に対して官能基が1モル当量未満となる量で、3官能以上のカップリング剤を添加し、ブロック共重合体Bを形成する工程
(4):上記(3)の工程で得られる溶液に、芳香族ビニル単量体を添加し、ブロック共重合体Aを形成する工程
(5):上記(4)の工程で得られる溶液から、ブロック共重合体組成物を回収する工程
次に、本発明のブロック共重合体組成物の第2態様について説明する。本態様のブロック共重合体組成物は、少なくとも3種のブロック共重合体を含有してなるものである。
本態様のブロック共重合体組成物を構成する3種のブロック共重合体のうちの1種であるブロック共重合体Pは、下記の一般式(P)で表される、互いに異なる重量平均分子量を持つ2つの芳香族ビニル重合体ブロックを有する、直鎖状の芳香族ビニル-共役ジエン-芳香族ビニルブロック共重合体である。
また、ブロック共重合体Qの共役ジエン重合体ブロック(Dq)のビニル結合含有量は、ブロック共重合体Pの共役ジエン重合体ブロック(Dp)のビニル結合含有量と実質的に等しいことが好ましい。
(7):上記(6)の工程で得られる活性末端を有する芳香族ビニル重合体を含有する溶液に、共役ジエン単量体を添加する工程
(8):上記(7)の工程で得られる活性末端を有する芳香族ビニル-共役ジエンブロック共重合体を含有する溶液に、その活性末端に対して官能基の総量が1モル当量未満となる量で、2官能のカップリング剤、および3官能以上のカップリング剤を添加し、ブロック共重合体Q、およびブロック共重合体Rを形成する工程
(9):上記(8)の工程で得られる溶液に、芳香族ビニル単量体を添加し、ブロック共重合体Pを形成する工程
(10):上記(9)の工程で得られる溶液から、ブロック共重合体組成物を回収する工程
流速0.35ml/分のテトラヒドロフランをキャリアとする高速液体クロマトグラフィによりポリスチレン換算分子量として求めた。装置は、東ソー社製HLC8220、カラムは昭和電工社製Shodex KF-404HQを3本連結したもの(カラム温度40℃)、検出器は示差屈折計および紫外検出器を用い、分子量の較正はポリマーラボラトリー社製の標準ポリスチレン(500から300万)の12点で実施した。
上記の高速液体クロマトグラフィにより得られたチャートの各ブロック共重合体に対応するピークの面積比から求めた。
Rubber Chem. Technol.,45,1295(1972)に記載された方法に従い、ブロック共重合体をオゾンと反応させ、水素化リチウムアルミニウムで還元することにより、ブロック共重合体のイソプレン重合体ブロックを分解した。具体的には、以下の手順で行なった。すなわち、モレキュラーシーブで処理したジクロロメタン100mlを入れた反応容器に、試料を300mg溶解した。この反応容器を冷却槽に入れ-25℃としてから、反応容器に170ml/minの流量で酸素を流しながら、オゾン発生器により発生させたオゾンを導入した。反応開始から30分経過後、反応容器から流出する気体をヨウ化カリウム水溶液に導入することにより、反応が完了したことを確認した。次いで、窒素置換した別の反応容器に、ジエチルエーテル50mlと水素化リチウムアルミニウム470mgを仕込み、氷水で反応容器を冷却しながら、この反応容器にオゾンと反応させた溶液をゆっくり滴下した。そして、反応容器を水浴に入れ、徐々に昇温して、40℃で30分間還流させた。その後、溶液を撹拌しながら、反応容器に希塩酸を少量ずつ滴下し、水素の発生がほとんど認められなくなるまで滴下を続けた。この反応の後、溶液に生じた固形の生成物をろ別し、固形の生成物は、100mlのジエチルエーテルで10分間抽出した。この抽出液と、ろ別した際のろ液とをあわせ、溶媒を留去することにより、固形の試料を得た。このようにして得られた試料につき、上記の重量平均分子量の測定法に従い、重量平均分子量を測定し、その値をスチレン重合体ブロックの重量平均分子量とした。
それぞれ上記のようにして求められた、ブロック共重合体の重量平均分子量から、対応するスチレン重合体ブロックの重量平均分子量を引き、その計算値に基づいてイソプレン重合体ブロックの重量平均分子量を求めた。
上記の高速液体クロマトグラフィの測定における、示差屈折計と紫外検出器との検出強度比に基づいて求めた。なお、予め、異なるスチレン単位含有量を有する共重合体を用意し、それらを用いて、検量線を作成した。
プロトンNMRの測定に基づき求めた。
プロトンNMRの測定に基づき求めた。
2枚のフィルムを用いて、一方を成形時の溶融流れ方向に沿って測定し、他方を成形時の溶融流れ垂直方向に沿って測定した。測定手順は以下の通りである。ORIENTEC社製のテンシロン万能試験機RTC-1210を用いて、引張速度300mm/minで100%まで伸張させ、その過程における50%伸張時の引張応力を測定し、50%伸張時におけるフィルムの引張弾性率を求め、1回目伸張時の引張弾性率として記録した。溶融流れ垂直方向の測定については、さらに、次の測定を行なった。1回目の測定で100%伸張させたフィルムを、伸張がない状態まで戻した後、再度、引張速度300mm/minで100%まで伸張させ、その過程における50%伸張時の引張応力を測定し、50%伸張時におけるフィルムの引張弾性率を求め、2回目伸張時の引張弾性率として記録した。なお、引張弾性率が高いものほど高い弾性率を有すると言え、(溶融流れ方向の引張弾性率/溶融流れ垂直方向の引張弾性率)の比が1に近いものほど引張弾性率の異方性が小さいと言え、(1回目の測定の引張弾性率/2回目の測定の引張弾性率)の比が1に近いものほど伸縮による弾性率の変化が小さいと言える。
ASTM 412に準拠して上記のテンシロン万能試験機を用いて測定した。具体的には、サンプル形状はDieAを使用し、伸張前の標線間距離を40mmとしてフィルムを伸び率200%で伸張させ、そのままの状態で10分間保持した後、はね返させることなく急に収縮させて、10分間放置後、標線間距離を測定し、下式に基づいて永久伸びを求めた。
永久伸び(%)=(L1-L0)/L0×100
L0:伸張前の標線間距離(mm)
L1:収縮させて10分間放置後の標線間距離(mm)
なお、この測定では、2枚のフィルムを用いて、一方を成形時の溶融流れ方向に沿って測定し、他方を成形時の溶融流れ垂直方向に沿って測定し、それぞれの値を記録した。
ブロック共重合体組成物の成形性(成形安定性)の指標として、フィルムの伸張粘度を測定した。測定手順は以下の通りである。測定装置としてTAインスツルメント社製のARESレオメーター、測定治具にARES-EVF伸張粘度測定冶具を用い、測定条件として伸張速度10秒-1、測定時間1.5秒、測定温度200℃で行った。サンプルとしてブロック共重合体を、150℃、2分間、熱プレスすることにより作成した厚さ1mmのフィルムを幅10mm、長さ20mm形状に切断したものを用いた。この条件により、フィルムの100%伸張時および350%伸張時の伸張粘度を測定した。100%伸張時の伸張粘度が高すぎると、成形性に劣るといえ、また、350%伸張時の伸張粘度が、100%伸張粘度に比べ低下した場合(350%時伸張粘度/100%時伸張粘度の値が1未満の場合)は、成形安定性に劣るといえる。
耐圧反応器に、シクロヘキサン23.3kg、N,N,N’,N’-テトラメチルエチレンジアミン(以下、TMEDAと称する)5.10ミリモルおよびスチレン1.60kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム169.9ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン5.20kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。次いで、カップリング剤としてテトラクロロシラン29.7ミリモルを添加して2時間カップリング反応を行い、ブロック共重合体Bとなる分岐状のスチレン-イソプレンブロック共重合体を形成させた。この後、50~60℃を保つように温度制御しながら、スチレン3.20kgを1時間にわたり連続的に添加した。スチレンの添加を完了した後、さらに1時間重合し、ブロック共重合体Aとなるスチレン-イソプレン-スチレンブロック共重合体を形成させた。スチレンの重合転化率は100%であった。この後、重合停止剤としてメタノール339.8ミリモルを添加してよく混合し反応を停止した。なお、反応に用いた各試剤の量は、表1にまとめた。得られた反応液の一部を取り出し、各ブロック共重合体およびブロック共重合体組成物の重量平均分子量、各スチレン重合体ブロックの重量平均分子量、各イソプレン重合体ブロックの重量平均分子量、各ブロック共重合体のスチレン単位含有量、ブロック共重合体組成物のスチレン単位含有量ならびにイソプレン重合体ブロックのビニル結合含有量を求めた。これらの値は、表2に示した。
フィルム引き取り速度 :10m/min
押出機温度 :投入口140℃、T-ダイ160℃に調整
スクリュー :フルフライト
押出機L/D :42
T-ダイ :幅300mm、リップ1mm
耐圧反応器に、シクロヘキサン23.3kg、TMEDA5.56ミリモルおよびスチレン1.55kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム185.0ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン5.20kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。次いで、カップリング剤としてテトラクロロシラン35.2ミリモルを添加して2時間カップリング反応を行い、ブロック共重合体Bとなる分岐状のスチレン-イソプレンブロック共重合体を形成させた。次いで、反応器にメタノール9.25ミリモルを添加することにより、一部のスチレン-イソプレンブロック共重合体の活性末端を失活させた。この後、50~60℃を保つように温度制御しながら、スチレン3.25kgを1時間にわたり連続的に添加した。スチレンの添加を完了した後、さらに1時間重合し、ブロック共重合体Aとなるスチレン-イソプレン-スチレンブロック共重合体を形成させた。スチレンの重合転化率は100%であった。この後、重合停止剤としてメタノール370.0ミリモルを添加してよく混合し反応を停止した。得られた反応液の一部を取り出し、実施例1と同様の測定を行なった。これらの値は、表2に示した。以下の操作は、実施例1と同様にして、実施例2のブロック共重合体組成物を回収し、ペレットとして、実施例2のフィルムを得て、このフィルムについて測定を行なった。その結果を表2に示す。
スチレン、n-ブチルリチウム、TMEDA、イソプレン、テトラクロロシランおよびメタノールの量を、それぞれ表1に示すように変更したこと以外は実施例1と同様にして、実施例3のブロック共重合体組成物を回収し、ペレットとして、実施例3のフィルムを得た。実施例3のブロック共重合体組成物およびフィルムについては、実施例1と同様の測定を行った。その結果を表2に示す。
耐圧反応器に、シクロヘキサン23.3kg、TMEDA5.00ミリモルおよびスチレン1.90kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム166.7ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン5.20kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。次いで、カップリング剤としてジメチルジクロロシラン50.0ミリモルおよびテトラクロロシラン12.5ミリモルを添加して2時間カップリング反応を行い、スチレン-イソプレン-スチレンブロック共重合体および分岐状のスチレン-イソプレンブロック共重合体を形成させた。この後、活性末端を有するスチレン-イソプレンブロック共重合体が残留していると考えられる反応液に、メタノール333.4ミリモルを添加してよく混合し、活性末端を失活させた。得られた反応液の一部を取り出し、実施例1と同様の測定を行なった。これらの値は、表2に示した。以下の操作は、実施例1と同様にして、比較例1のブロック共重合体組成物を回収し、ペレットとして、比較例1のフィルムを得て、このフィルムについて測定を行なった。その結果は表2に示す。
耐圧反応器に、シクロヘキサン23.3kg、TMEDA7.90ミリモルおよびスチレン3.00kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム263.2ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン7.00kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。次いで、カップリング剤としてテトラクロロシラン62.4ミリモルを添加して2時間カップリング反応を行い、分岐状のスチレン-イソプレンブロック共重合体を形成させた。この後、活性末端を有するスチレン-イソプレンブロック共重合体が残留していると考えられる反応液に、メタノール526.4ミリモルを添加してよく混合し、活性末端を失活させた。得られた反応液の一部を取り出し、実施例1と同様の測定を行なった。これらの値は、表2に示した。以下の操作は、実施例1と同様にして、比較例2のブロック共重合体組成物を回収し、ペレットとして、比較例2のフィルムを得て、このフィルムについて測定を行なった。その結果は表2に示す。
耐圧反応器に、シクロヘキサン40.0kg、TMEDA0.95ミリモルおよびスチレン4.30kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム63.6ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン5.20kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。さらに引き続き、50~60℃を保つように温度制御しながら、反応器にスチレン0.50kgを10分間にわたり連続的に添加した。スチレンの添加を完了した後、さらに1時間重合した。スチレンの重合転化率は100%であった。この後、重合停止剤としてメタノール127.2ミリモルを添加してよく混合し反応を停止した。得られた反応液の一部を取り出し、実施例1と同様の測定を行なった。これらの値は、表2に示した。以下の操作は、実施例1と同様にして、比較例3のブロック共重合体組成物を回収し、ペレットとして、比較例3のフィルムを得て、このフィルムについて測定を行なった。その結果は表2に示す。
スチレン、n-ブチルリチウム、TMEDA、イソプレン、テトラクロロシランおよびメタノールの量を、それぞれ表1に示すように変更したこと以外は実施例1と同様にして、比較例4のブロック共重合体組成物を回収し、ペレットとして、比較例4のフィルムを得た。比較例4のブロック共重合体組成物およびフィルムについては、実施例1と同様の測定を行った。その結果を表2に示す。
耐圧反応器に、シクロヘキサン23.3kg、N,N,N’,N’-テトラメチルエチレンジアミン(以下、TMEDAと称する)4.80ミリモルおよびスチレン1.55kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム160.8ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン5.20kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。次いで、テトラクロロシラン7.2ミリモルを添加し、30分間カップリング反応を行い、ブロック共重合体Rとなる分岐状のスチレン-イソプレン-スチレンブロック共重合体を形成させた。この後、ジメチルジクロロシラン50.6ミリモルを添加して、さらに1時間カップリング反応を行い、ブロック共重合体Qとなる直鎖状のスチレン-イソプレン-スチレンブロック共重合体を形成させた。この後、50~60℃を保つように温度制御しながら、スチレン3.25kgを1時間にわたり連続的に添加した。スチレンの添加を完了した後、さらに1時間重合し、ブロック共重合体Pとなる直鎖状で非対称なスチレン-イソプレン-スチレンブロック共重合体を形成させた。スチレンの重合転化率は100%であった。この後、重合停止剤としてメタノール321.6ミリモルを添加してよく混合し反応を停止した。なお、反応に用いた各試剤の量は、表3にまとめた。得られた反応液の一部を取り出し、各ブロック共重合体およびブロック共重合体組成物の重量平均分子量、各ブロック共重合体の重量比、各スチレン重合体ブロックの重量平均分子量、各イソプレン重合体ブロックの重量平均分子量、各ブロック共重合体のスチレン単位含有量、ブロック共重合体組成物のスチレン単位含有量ならびにイソプレン重合体ブロックのビニル結合含有量を求めた。これらの値は、表4に示した。
スチレン、n-ブチルリチウム、TMEDA、ジメチルジクロロシラン、テトラメトキシシラン、およびメタノールの量を、それぞれ表3に示すように変更したこと以外は実施例4と同様にして、実施例5のブロック共重合体組成物を回収し、ペレットとして、実施例5のフィルムを得た。実施例5のブロック共重合体組成物およびフィルムについては、実施例4と同様の測定を行った。その結果を表4および表5に示す。
耐圧反応器に、シクロヘキサン23.3kg、TMEDA1.9ミリモルおよびスチレン1.50kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム128.8ミリモルを添加し、50℃に昇温しながら1時間重合した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン7.00kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。この後、50~60℃を保つように温度制御しながら、スチレン1.50kgを1時間にわたり連続的に添加した。スチレンの添加を完了した後、さらに1時間重合し、スチレン-イソプレン-スチレントリブロック共重合体を形成させた。スチレンの重合転化率は100%であった。この後、重合停止剤としてメタノール257.6ミリモルを添加してよく混合し反応を停止した。得られた反応液の一部を取り出し、実施例4と同様の測定を行なった。これらの値は、表4に示した。以下の操作は、実施例4と同様にして、比較例5のブロック共重合体組成物を回収し、ペレットとして、比較例5のフィルムを得て、このフィルムについて測定を行なった。その結果を表5に示す。
スチレン、n-ブチルリチウム、TMEDA、イソプレンおよびメタノールの量を、それぞれ表3に示すように変更したこと以外は比較例5と同様にして、比較例6および比較例7のブロック共重合体組成物を回収し、ペレットとして、比較例6および比較例7のフィルムを得た。比較例6および比較例7のブロック共重合体組成物およびフィルムについては、実施例4と同様の測定を行った。その結果を表4および表5に示す。
耐圧反応器に、シクロヘキサン23.3kg、TMEDA4.50ミリモルおよびスチレン3.14kgを添加し、40℃で攪拌しているところに、n-ブチルリチウム80.0ミリモルを添加し、50℃に昇温しながら1時間重合し、ついでn-ブチルリチウム220.0ミリモルを添加し、続いてスチレン1.99kgを30分にわたり連続添加し、重合を1時間継続した。スチレンの重合転化率は100%であった。引き続き、50~60℃を保つように温度制御しながら、反応器にイソプレン2.88kgを1時間にわたり連続的に添加した。イソプレンの添加を完了した後、さらに1時間重合した。イソプレンの重合転化率は100%であった。さらに次いで、スチレン1.99kgを30分間連続添加し、スチレンの添加を完了した後、さらに1時間重合し、2種の直鎖状のスチレン-イソプレン-スチレンブロック共重合体を形成させた。スチレンの重合転化率は100%であった。この後、重合停止剤としてメタノール600.0ミリモルを添加してよく混合し反応を停止した。得られた反応液の一部を取り出し、実施例4と同様の測定を行なった。これらの値は、表4に示した。以下の操作は、実施例4と同様にして、比較例8のブロック共重合体組成物を回収し、ペレットとして、比較例8のフィルムを得て、このフィルムについて測定を行なった。その結果を表5に示す。
スチレン、n-ブチルリチウム、TMEDA、イソプレンおよびメタノールの量を、それぞれ表3に示すように変更したこと以外は比較例8と同様にして、比較例9および比較例10のブロック共重合体組成物を回収し、ペレットとして、比較例9および比較例10のフィルムを得た。比較例9および比較例10のブロック共重合体組成物およびフィルムについては、実施例4と同様の測定を行った。その結果を表4および表5に示す。
Claims (10)
- 下記の一般式(A)で表されるブロック共重合体Aと、下記の一般式(B)で表されるブロック共重合体Bとを含有してなるブロック共重合体組成物であって、ブロック共重合体Aとブロック共重合体Bとの重量比(A/B)が36/64~85/15であるブロック共重合体組成物。
Ar1a-Da-Ar2a (A)
(Arb-Db)n-X (B)
(一般式(A)および(B)において、Ar1aおよびArbは、それぞれ、重量平均分子量が6000~15000の芳香族ビニル重合体ブロックであり、Ar2aは、重量平均分子量が40000~400000の芳香族ビニル重合体ブロックであり、DaおよびDbは、それぞれ、ビニル結合含有量が1~20モル%の共役ジエン重合体ブロックであり、Xはカップリング剤の残基であり、nは3以上の整数である。) - ブロック共重合体組成物の重合体成分の全繰返し単位において、芳香族ビニル単量体単位が占める割合が27~70重量%である、請求項1に記載のブロック共重合体組成物。
- 請求項1または2に記載のブロック共重合体組成物を成形してなるフィルム。
- 下記の(1)~(5)の工程からなる、請求項1または2に記載のブロック共重合体組成物の製造方法。
(1):溶媒中で重合開始剤を用いて、芳香族ビニル単量体を重合する工程
(2):上記(1)の工程で得られる活性末端を有する芳香族ビニル重合体を含有する溶液に、共役ジエン単量体を添加する工程
(3):上記(2)の工程で得られる活性末端を有する芳香族ビニル-共役ジエンブロック共重合体を含有する溶液に、その活性末端に対して官能基が1モル当量未満となる量で、3官能以上のカップリング剤を添加し、ブロック共重合体Bを形成する工程
(4):上記(3)の工程で得られる溶液に、芳香族ビニル単量体を添加し、ブロック共重合体Aを形成する工程
(5):上記(4)の工程で得られる溶液から、ブロック共重合体組成物を回収する工程 - 下記の一般式(P)で表されるブロック共重合体P、下記の一般式(Q)で表されるブロック共重合体Q、および下記の一般式(R)で表されるブロック共重合体Rを含有してなるブロック共重合体組成物。
Ar1p-Dp-Ar2p (P)
(Arq-Dq)2-Xq (Q)
(Arr-Dr)m-Xr (R)
(一般式(P)、(Q)、および(R)において、Ar1p、Arq、およびArrは、それぞれ、重量平均分子量が6000~20000の芳香族ビニル重合体ブロックであり、Ar2pは、重量平均分子量が40000~400000の芳香族ビニル重合体ブロックであり、Dp、Dq、およびDrは、それぞれ、ビニル結合含有量が1~20モル%の共役ジエン重合体ブロックであり、Xqは、単結合、またはカップリング剤の残基であり、Xrは、カップリング剤の残基であり、mは3以上の整数である。) - ブロック共重合体Pの量と、ブロック共重合体Qおよびブロック共重合体Rの合計量との重量比(P/(Q+R))が、10/90~80/20である、請求項5に記載のブロック共重合体組成物。
- ブロック共重合体Qの量と、ブロック共重合体Rの量との重量比(Q/R)が、15/85~85/15である、請求項5または6に記載のブロック共重合体組成物。
- ブロック共重合体組成物の重合体成分の全繰返し単位において、芳香族ビニル単量体単位が占める割合が27~70重量%である、請求項5~7のいずれかに記載のブロック共重合体組成物。
- 請求項5~8のいずれかに記載のブロック共重合体組成物を成形してなるフィルム。
- 下記の(6)~(10)の工程からなる、請求項5~8のいずれかに記載のブロック共重合体組成物の製造方法。
(6):溶媒中で重合開始剤を用いて、芳香族ビニル単量体を重合する工程
(7):上記(6)の工程で得られる活性末端を有する芳香族ビニル重合体を含有する溶液に、共役ジエン単量体を添加する工程
(8):上記(7)の工程で得られる活性末端を有する芳香族ビニル-共役ジエンブロック共重合体を含有する溶液に、その活性末端に対して官能基の総量が1モル当量未満となる量で、2官能のカップリング剤、および3官能以上のカップリング剤を添加し、ブロック共重合体Q、およびブロック共重合体Rを形成する工程
(9):上記(8)の工程で得られる溶液に、芳香族ビニル単量体を添加し、ブロック共重合体Pを形成する工程
(10):上記(9)の工程で得られる溶液から、ブロック共重合体組成物を回収する工程
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010544186A JP5673105B2 (ja) | 2008-12-26 | 2009-12-25 | ブロック共重合体組成物、フィルムおよびブロック共重合体組成物の製造方法 |
| EP09835066.3A EP2371900B1 (en) | 2008-12-26 | 2009-12-25 | Block copolymer composition, film, and process for producing block copolymer composition |
| US13/141,950 US8598271B2 (en) | 2008-12-26 | 2009-12-25 | Block copolymer composition, film, and method for producing block copolymer composition |
| CN200980152584.1A CN102264832B (zh) | 2008-12-26 | 2009-12-25 | 嵌段共聚物组合物、薄膜以及嵌段共聚物组合物的制造方法 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008-335329 | 2008-12-26 | ||
| JP2008335329 | 2008-12-26 | ||
| JP2009-088533 | 2009-03-31 | ||
| JP2009088533 | 2009-03-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010074270A1 true WO2010074270A1 (ja) | 2010-07-01 |
Family
ID=42287874
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2009/071692 WO2010074270A1 (ja) | 2008-12-26 | 2009-12-25 | ブロック共重合体組成物、フィルムおよびブロック共重合体組成物の製造方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US8598271B2 (ja) |
| EP (1) | EP2371900B1 (ja) |
| JP (1) | JP5673105B2 (ja) |
| CN (1) | CN102264832B (ja) |
| WO (1) | WO2010074270A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012077158A (ja) * | 2010-09-30 | 2012-04-19 | Nippon Zeon Co Ltd | 重合体組成物の製造方法 |
| JP5533866B2 (ja) * | 2009-06-30 | 2014-06-25 | 日本ゼオン株式会社 | 伸縮性フィルム用組成物 |
| JP2016069565A (ja) * | 2014-09-30 | 2016-05-09 | 日本ゼオン株式会社 | ブロック共重合体組成物およびフィルム |
| WO2020045496A1 (ja) * | 2018-08-28 | 2020-03-05 | 日本ゼオン株式会社 | 組成物および弾性体 |
| JP2020050761A (ja) * | 2018-09-27 | 2020-04-02 | 日本ゼオン株式会社 | 芳香族ビニル−共役ジエンブロック共重合体の成形体 |
| WO2020203371A1 (ja) * | 2019-03-29 | 2020-10-08 | 日本ゼオン株式会社 | 非水系二次電池用バインダー組成物、非水系二次電池電極用スラリー組成物、非水系二次電池用電極、及び非水系二次電池 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5772592B2 (ja) * | 2009-03-31 | 2015-09-02 | 日本ゼオン株式会社 | ラベル用粘接着剤組成物 |
| US9575359B2 (en) | 2013-09-24 | 2017-02-21 | Gentex Corporation | Emissive display system |
| CN109504018B (zh) * | 2017-09-14 | 2021-08-03 | 中国石油化工股份有限公司 | 一种二臂苯乙烯-共轭二烯的聚合物组合物以及制备和在沥青改性中的应用 |
| WO2023036918A1 (en) * | 2021-09-10 | 2023-03-16 | Ineos Styrolution Group Gmbh | Organoleptically improved, low film-blocking styrene butadiene block copolymers |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57178722A (en) * | 1981-04-28 | 1982-11-04 | Asahi Chem Ind Co Ltd | Biaxially stretched film, sheet or tube of block copolymer |
| JPS59187048A (ja) * | 1983-03-10 | 1984-10-24 | Nippon Steel Chem Co Ltd | 熱可塑性樹脂組成物 |
| JPS63179956A (ja) * | 1987-01-22 | 1988-07-23 | Asahi Chem Ind Co Ltd | 形状記憶性樹脂組成物 |
| JPS6465152A (en) * | 1987-05-14 | 1989-03-10 | Montedipe Spa | Compound based on vinyl aromatic polymer having high strength and chemical resistance |
| JPS6479251A (en) * | 1984-04-21 | 1989-03-24 | Asahi Chemical Ind | Composition containing block copolymer |
| JPH05287084A (ja) * | 1992-04-13 | 1993-11-02 | Res Dev Corp Of Japan | 三次元共連続ミクロ相分離構造を持つ多成分多相系高分子材料の製造方法 |
| JPH0966554A (ja) * | 1995-08-31 | 1997-03-11 | Daicel Chem Ind Ltd | スチレン系透明シートおよびフィルムの製造方法 |
| JP2003261740A (ja) * | 2002-03-11 | 2003-09-19 | Nippon Zeon Co Ltd | エラストマー組成物の製造方法 |
| JP2006274158A (ja) * | 2005-03-30 | 2006-10-12 | Nippon Zeon Co Ltd | エラストマー組成物及び接着剤組成物 |
| JP2006528273A (ja) | 2003-05-14 | 2006-12-14 | ダウ・グローバル・テクノロジーズ・インコーポレーテツド | ブロックコポリマー組成物およびそれより製造される透明なエラストマー物品 |
| JP2008007654A (ja) | 2006-06-30 | 2008-01-17 | Nippon Zeon Co Ltd | 伸縮性フィルム、伸縮性部材及び衛生用品 |
| WO2009123089A1 (ja) * | 2008-03-31 | 2009-10-08 | 日本ゼオン株式会社 | ブロック共重合体組成物、その製造方法及びフィルム |
Family Cites Families (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA1101586A (en) | 1971-05-17 | 1981-05-19 | Arnel D. Potter | Photosensitive compositions |
| US4104327A (en) | 1975-01-16 | 1978-08-01 | Nippon Zeon Co. Ltd. | Adhesive compositions |
| JPS51125135A (en) | 1975-01-22 | 1976-11-01 | Nippon Zeon Co Ltd | Novel adhesive composition |
| DE2940861A1 (de) | 1979-10-09 | 1981-04-23 | Basf Ag, 6700 Ludwigshafen | Verfahren zur herstellung von mischungen linearer dreiblockcopolymerisate sowie formteile aus diesen |
| JPS5922219B2 (ja) | 1980-07-07 | 1984-05-25 | 東京応化工業株式会社 | フレキソ印刷版用感光性樹脂組成物 |
| EP0058952B1 (en) | 1981-02-20 | 1984-08-22 | Asahi Kasei Kogyo Kabushiki Kaisha | A film, sheet or tube of a block copolymer or a composition containing the same |
| JPH0686586B2 (ja) | 1985-04-05 | 1994-11-02 | 日東電工株式会社 | 感圧性接着剤組成物 |
| JPH07107152B2 (ja) | 1986-09-05 | 1995-11-15 | 積水化学工業株式会社 | 粘着剤組成物 |
| JPH0798889B2 (ja) | 1989-02-23 | 1995-10-25 | 電気化学工業株式会社 | 熱収縮性フィルム |
| US5290842A (en) | 1991-09-03 | 1994-03-01 | Avery Dennison Corporation | Pressure-sensitive adhesives based on preferentially tackified immiscible elastomers |
| JP3168632B2 (ja) | 1991-10-02 | 2001-05-21 | 旭化成株式会社 | ホットメルト型粘着剤組成物 |
| JP3157256B2 (ja) | 1992-03-19 | 2001-04-16 | 旭化成株式会社 | 粘接着用樹脂組成物 |
| JP2705905B2 (ja) | 1994-08-18 | 1998-01-28 | 日本エヌエスシー株式会社 | ホットメルト接着剤組成物 |
| JP3196561B2 (ja) | 1995-04-11 | 2001-08-06 | 日本エヌエスシー株式会社 | 使い捨て製品用ホットメルト接着剤およびそれを用いた使い捨て製品 |
| JPH0978048A (ja) | 1995-09-14 | 1997-03-25 | J S R Shell Elastomer Kk | ホットメルト粘着剤組成物 |
| AU709222B2 (en) | 1996-02-23 | 1999-08-26 | Bostik Findley, Inc. | Improved pressure sensitive labeling adhesive |
| US6329459B1 (en) | 1996-09-23 | 2001-12-11 | Bridgestone Corporation | Extended syndiotactic polystyrene-elastomeric block copolymers |
| JP3184783B2 (ja) | 1997-06-20 | 2001-07-09 | アロン化成株式会社 | 制振材料 |
| KR100528043B1 (ko) | 1997-10-15 | 2005-11-15 | 미네소타 마이닝 앤드 매뉴팩춰링 캄파니 | 이방성 탄성 필름 및 웨브 |
| JP2003342441A (ja) * | 1998-05-13 | 2003-12-03 | Nippon Zeon Co Ltd | ブロック共重合体組成物、その製造方法及び粘着剤組成物 |
| WO2000000546A1 (en) | 1998-06-29 | 2000-01-06 | Shell Internationale Research Maatschappij B.V. | Photo-curable polymer composition and flexographic printing plates containing the same |
| US6344518B1 (en) | 1998-11-10 | 2002-02-05 | Jsr Corporation | Conjugated diolefin copolymer rubber and rubber composition |
| JP4304791B2 (ja) | 1998-11-10 | 2009-07-29 | Jsr株式会社 | 共役ジオレフィン系共重合ゴム及びゴム組成物 |
| JP4044687B2 (ja) | 1998-11-24 | 2008-02-06 | 旭化成ケミカルズ株式会社 | 感光性エラストマー組成物 |
| US6593430B1 (en) | 1999-03-27 | 2003-07-15 | Basf Aktiengesellschaft | Transparent, impact-resistant polystyrene on a styrene-butadiene block copolymer basis |
| EP1158364A3 (de) | 2000-05-03 | 2003-04-23 | BASF Drucksysteme GmbH | Fotopolymerisierbare Flexodruckelemente mit SIS/SBS-Gemischen als Bindemittel zur Herstellung von Flexodruckformen |
| JP4623246B2 (ja) | 2000-08-11 | 2011-02-02 | ジェイエスアール クレイトン エラストマー株式会社 | アスファルト改質用共重合体(組成物)、およびアスファルト組成物 |
| JP3947050B2 (ja) | 2001-06-22 | 2007-07-18 | 株式会社クラレ | ブロック共重合体の製造方法 |
| EP1424359B1 (en) | 2001-08-29 | 2013-02-27 | Zeon Corporation | Elastomer composition and pressure-sensitive adhesives made by using the same |
| DE10158500A1 (de) | 2001-11-28 | 2003-06-12 | Basf Ag | Transparente Styrol-Butadien-Blockcopolymermischungen |
| JP3922446B2 (ja) | 2002-09-19 | 2007-05-30 | 株式会社クラレ | 発泡体およびその用途 |
| JP4518742B2 (ja) | 2003-02-07 | 2010-08-04 | 日本エラストマー株式会社 | 新規な粘接着剤組成物 |
| JP2006520826A (ja) | 2003-02-21 | 2006-09-14 | クレイトン・ポリマーズ・リサーチ・ベー・ベー | 改善されたダイス切断性能を有するラベルストック |
| US7211627B2 (en) | 2003-02-28 | 2007-05-01 | Zeon Corporation | Elastomer composition and pressure-sensitive adhesive compositions |
| EP1473595A1 (en) | 2003-04-29 | 2004-11-03 | KRATON Polymers Research B.V. | Photopolymerizable compositions and flexographic printing plates derived therefrom |
| JP4300868B2 (ja) | 2003-04-30 | 2009-07-22 | 東ソー株式会社 | 制振性発泡体成形用樹脂組成物及び制振性発泡体 |
| JP4539561B2 (ja) | 2003-09-25 | 2010-09-08 | 日本ゼオン株式会社 | 感光性フレキソ版用ブロック共重合体組成物 |
| JP4381844B2 (ja) | 2004-02-18 | 2009-12-09 | 株式会社クラレ | 伸縮性に優れた熱可塑性重合体組成物およびその成形体 |
| EP1775332B1 (en) | 2004-08-05 | 2019-03-06 | Japan Elastomer Co., Ltd. | Pressure-sensitive adhesive composition |
| JP2006089546A (ja) | 2004-09-22 | 2006-04-06 | Kuraray Co Ltd | 伸縮部材 |
| CN101057181B (zh) | 2004-11-11 | 2011-01-12 | 旭化成电子材料株式会社 | 柔性版印刷用感光性树脂组合物 |
| JP2006241177A (ja) | 2005-02-28 | 2006-09-14 | Nippon Zeon Co Ltd | 芳香族ビニル化合物−イソプレンブロック共重合体組成物及び粘・接着剤組成物 |
| US20060205877A1 (en) | 2005-03-11 | 2006-09-14 | Kraton Polymers U.S. Llc | Adhesive compositions comprising mixtures of block copolymers |
| JP2006282683A (ja) | 2005-03-31 | 2006-10-19 | Nippon Zeon Co Ltd | 伸縮性フィルム |
| WO2007072613A1 (ja) | 2005-12-22 | 2007-06-28 | Kraton Jsr Elastomers K.K. | エラストマー構成体およびそれを用いた感光性組成物 |
| JP4889321B2 (ja) | 2006-03-03 | 2012-03-07 | スリーエム イノベイティブ プロパティズ カンパニー | 伸縮性積層部材及び物品 |
| JP5240985B2 (ja) * | 2007-11-06 | 2013-07-17 | 旭化成ケミカルズ株式会社 | ブロック共重合体組成物、その水添物の製造方法 |
-
2009
- 2009-12-25 CN CN200980152584.1A patent/CN102264832B/zh active Active
- 2009-12-25 JP JP2010544186A patent/JP5673105B2/ja active Active
- 2009-12-25 US US13/141,950 patent/US8598271B2/en active Active
- 2009-12-25 WO PCT/JP2009/071692 patent/WO2010074270A1/ja active Application Filing
- 2009-12-25 EP EP09835066.3A patent/EP2371900B1/en active Active
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57178722A (en) * | 1981-04-28 | 1982-11-04 | Asahi Chem Ind Co Ltd | Biaxially stretched film, sheet or tube of block copolymer |
| JPS59187048A (ja) * | 1983-03-10 | 1984-10-24 | Nippon Steel Chem Co Ltd | 熱可塑性樹脂組成物 |
| JPS6479251A (en) * | 1984-04-21 | 1989-03-24 | Asahi Chemical Ind | Composition containing block copolymer |
| JPS63179956A (ja) * | 1987-01-22 | 1988-07-23 | Asahi Chem Ind Co Ltd | 形状記憶性樹脂組成物 |
| JPS6465152A (en) * | 1987-05-14 | 1989-03-10 | Montedipe Spa | Compound based on vinyl aromatic polymer having high strength and chemical resistance |
| JPH05287084A (ja) * | 1992-04-13 | 1993-11-02 | Res Dev Corp Of Japan | 三次元共連続ミクロ相分離構造を持つ多成分多相系高分子材料の製造方法 |
| JPH0966554A (ja) * | 1995-08-31 | 1997-03-11 | Daicel Chem Ind Ltd | スチレン系透明シートおよびフィルムの製造方法 |
| JP2003261740A (ja) * | 2002-03-11 | 2003-09-19 | Nippon Zeon Co Ltd | エラストマー組成物の製造方法 |
| JP2006528273A (ja) | 2003-05-14 | 2006-12-14 | ダウ・グローバル・テクノロジーズ・インコーポレーテツド | ブロックコポリマー組成物およびそれより製造される透明なエラストマー物品 |
| JP2006274158A (ja) * | 2005-03-30 | 2006-10-12 | Nippon Zeon Co Ltd | エラストマー組成物及び接着剤組成物 |
| JP2008007654A (ja) | 2006-06-30 | 2008-01-17 | Nippon Zeon Co Ltd | 伸縮性フィルム、伸縮性部材及び衛生用品 |
| WO2009123089A1 (ja) * | 2008-03-31 | 2009-10-08 | 日本ゼオン株式会社 | ブロック共重合体組成物、その製造方法及びフィルム |
Non-Patent Citations (2)
| Title |
|---|
| RUBBER CHEM. TECHNOL., vol. 45, 1972, pages 1295 |
| See also references of EP2371900A4 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5533866B2 (ja) * | 2009-06-30 | 2014-06-25 | 日本ゼオン株式会社 | 伸縮性フィルム用組成物 |
| JP2012077158A (ja) * | 2010-09-30 | 2012-04-19 | Nippon Zeon Co Ltd | 重合体組成物の製造方法 |
| JP2016069565A (ja) * | 2014-09-30 | 2016-05-09 | 日本ゼオン株式会社 | ブロック共重合体組成物およびフィルム |
| JP7164933B2 (ja) | 2014-09-30 | 2022-11-02 | 日本ゼオン株式会社 | ブロック共重合体組成物およびフィルム |
| WO2020045496A1 (ja) * | 2018-08-28 | 2020-03-05 | 日本ゼオン株式会社 | 組成物および弾性体 |
| JPWO2020045496A1 (ja) * | 2018-08-28 | 2021-08-10 | 日本ゼオン株式会社 | 組成物および弾性体 |
| JP7435451B2 (ja) | 2018-08-28 | 2024-02-21 | 日本ゼオン株式会社 | 組成物および弾性体 |
| JP2020050761A (ja) * | 2018-09-27 | 2020-04-02 | 日本ゼオン株式会社 | 芳香族ビニル−共役ジエンブロック共重合体の成形体 |
| WO2020203371A1 (ja) * | 2019-03-29 | 2020-10-08 | 日本ゼオン株式会社 | 非水系二次電池用バインダー組成物、非水系二次電池電極用スラリー組成物、非水系二次電池用電極、及び非水系二次電池 |
Also Published As
| Publication number | Publication date |
|---|---|
| US20110257336A1 (en) | 2011-10-20 |
| CN102264832B (zh) | 2014-01-08 |
| EP2371900B1 (en) | 2016-10-05 |
| US8598271B2 (en) | 2013-12-03 |
| JP5673105B2 (ja) | 2015-02-18 |
| EP2371900A4 (en) | 2012-10-24 |
| JPWO2010074270A1 (ja) | 2012-06-21 |
| CN102264832A (zh) | 2011-11-30 |
| EP2371900A1 (en) | 2011-10-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5582031B2 (ja) | ブロック共重合体組成物、その製造方法及びフィルム | |
| JP5673105B2 (ja) | ブロック共重合体組成物、フィルムおよびブロック共重合体組成物の製造方法 | |
| JP5494650B2 (ja) | 伸縮性フィルム用組成物 | |
| JP5527220B2 (ja) | ブロック共重合体組成物およびホットメルト粘接着剤組成物 | |
| JP5569310B2 (ja) | 重合体組成物の製造方法 | |
| JP5533866B2 (ja) | 伸縮性フィルム用組成物 | |
| JP6521256B2 (ja) | ブロック共重合体組成物、その製造方法およびフィルム | |
| JP5354272B2 (ja) | ブロック共重合体組成物の製造方法 | |
| JP2006282683A (ja) | 伸縮性フィルム | |
| JP2016069565A (ja) | ブロック共重合体組成物およびフィルム | |
| JP5970812B2 (ja) | ブロック共重合体組成物およびフィルム | |
| WO2021065623A1 (ja) | 水添ブロック共重合体組成物、その製造方法およびフィルム | |
| JP5311050B2 (ja) | 弾性フィラメント用組成物、弾性フィラメントおよび伸縮性シート | |
| US20230111050A1 (en) | Block-copolymer pellets, tacky adhesive composition, and stretch film |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980152584.1 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09835066 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2010544186 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2009835066 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009835066 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13141950 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |