WO2011001795A1 - 金属積層構造体および金属積層構造体の製造方法 - Google Patents
金属積層構造体および金属積層構造体の製造方法 Download PDFInfo
- Publication number
- WO2011001795A1 WO2011001795A1 PCT/JP2010/059695 JP2010059695W WO2011001795A1 WO 2011001795 A1 WO2011001795 A1 WO 2011001795A1 JP 2010059695 W JP2010059695 W JP 2010059695W WO 2011001795 A1 WO2011001795 A1 WO 2011001795A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- metal layer
- metal
- layer
- thickness
- tungsten
- Prior art date
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/01—Layered products comprising a layer of metal all layers being exclusively metallic
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B15/00—Layered products comprising a layer of metal
- B32B15/20—Layered products comprising a layer of metal comprising aluminium or copper
-
- C—CHEMISTRY; METALLURGY
- C23—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; CHEMICAL SURFACE TREATMENT; DIFFUSION TREATMENT OF METALLIC MATERIAL; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL; INHIBITING CORROSION OF METALLIC MATERIAL OR INCRUSTATION IN GENERAL
- C23C—COATING METALLIC MATERIAL; COATING MATERIAL WITH METALLIC MATERIAL; SURFACE TREATMENT OF METALLIC MATERIAL BY DIFFUSION INTO THE SURFACE, BY CHEMICAL CONVERSION OR SUBSTITUTION; COATING BY VACUUM EVAPORATION, BY SPUTTERING, BY ION IMPLANTATION OR BY CHEMICAL VAPOUR DEPOSITION, IN GENERAL
- C23C28/00—Coating for obtaining at least two superposed coatings either by methods not provided for in a single one of groups C23C2/00 - C23C26/00 or by combinations of methods provided for in subclasses C23C and C25C or C25D
- C23C28/02—Coating for obtaining at least two superposed coatings either by methods not provided for in a single one of groups C23C2/00 - C23C26/00 or by combinations of methods provided for in subclasses C23C and C25C or C25D only coatings only including layers of metallic material
- C23C28/021—Coating for obtaining at least two superposed coatings either by methods not provided for in a single one of groups C23C2/00 - C23C26/00 or by combinations of methods provided for in subclasses C23C and C25C or C25D only coatings only including layers of metallic material including at least one metal alloy layer
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D3/00—Electroplating: Baths therefor
- C25D3/66—Electroplating: Baths therefor from melts
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/10—Electroplating with more than one layer of the same or of different metals
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D5/00—Electroplating characterised by the process; Pretreatment or after-treatment of workpieces
- C25D5/10—Electroplating with more than one layer of the same or of different metals
- C25D5/12—Electroplating with more than one layer of the same or of different metals at least one layer being of nickel or chromium
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D7/00—Electroplating characterised by the article coated
- C25D7/06—Wires; Strips; Foils
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D7/00—Electroplating characterised by the article coated
- C25D7/12—Semiconductors
- C25D7/123—Semiconductors first coated with a seed layer or a conductive layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2311/00—Metals, their alloys or their compounds
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/34—Arrangements for cooling, heating, ventilating or temperature compensation ; Temperature sensing arrangements
- H01L23/36—Selection of materials, or shaping, to facilitate cooling or heating, e.g. heatsinks
- H01L23/373—Cooling facilitated by selection of materials for the device or materials for thermal expansion adaptation, e.g. carbon
- H01L23/3735—Laminates or multilayers, e.g. direct bond copper ceramic substrates
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/34—Arrangements for cooling, heating, ventilating or temperature compensation ; Temperature sensing arrangements
- H01L23/36—Selection of materials, or shaping, to facilitate cooling or heating, e.g. heatsinks
- H01L23/373—Cooling facilitated by selection of materials for the device or materials for thermal expansion adaptation, e.g. carbon
- H01L23/3736—Metallic materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L2924/00—Indexing scheme for arrangements or methods for connecting or disconnecting semiconductor or solid-state bodies as covered by H01L24/00
- H01L2924/0001—Technical content checked by a classifier
- H01L2924/0002—Not covered by any one of groups H01L24/00, H01L24/00 and H01L2224/00
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L33/00—Semiconductor devices with at least one potential-jump barrier or surface barrier specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof
- H01L33/48—Semiconductor devices with at least one potential-jump barrier or surface barrier specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof characterised by the semiconductor body packages
- H01L33/64—Heat extraction or cooling elements
- H01L33/641—Heat extraction or cooling elements characterized by the materials
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/12—All metal or with adjacent metals
- Y10T428/12493—Composite; i.e., plural, adjacent, spatially distinct metal components [e.g., layers, joint, etc.]
- Y10T428/12771—Transition metal-base component
- Y10T428/12806—Refractory [Group IVB, VB, or VIB] metal-base component
- Y10T428/12826—Group VIB metal-base component
Definitions
- the present invention relates to a metal laminate structure and a method for producing the metal laminate structure.
- heat sink heat dissipation substrate
- Patent Document 1 Japanese Patent Laid-Open No. 6-268115
- a Cu plate having a diameter of 200 mm and a thickness of 0.465 mm manufactured using a rolling method, and a Mo plate having the same diameter as the Cu plate and a thickness of 0.090 mm are disclosed.
- a Cu / Mo / Cu clad material having a thickness of 1.02 mm is produced by hot pressing in a state in which Cu plates / Mo plates / Cu plates are stacked in this order, and this can be used as a heat dissipation substrate of a semiconductor device. (Patent Document 1, paragraphs [0034] to [0049], etc.).
- Patent Document 1 also describes that a highly reliable clad material can be obtained by the same manufacturing method even when W is used instead of Mo (paragraph [0033] of Patent Document 1).
- the Mo plate and the W plate have a problem that it is difficult to reduce the overall thickness of the heat dissipation substrate because there is a restriction on the thickness that can be rolled.
- the manufacturing method in which the Cu plate and the Mo plate are installed in the hot press device each time the clad material is manufactured, and the clad material is taken out from the hot press device after the hot press is efficient.
- a heat dissipation substrate cannot be manufactured.
- an object of the present invention is to provide a metal laminate structure and a method for producing the metal laminate structure that can be made thin and can be efficiently manufactured.
- the present invention includes a first metal layer, a second metal layer, and a third metal layer, wherein the first metal layer is disposed on one surface of the second metal layer, The metal layer is disposed on the other surface of the second metal layer, the first metal layer includes at least one of tungsten and molybdenum, the second metal layer includes copper, The metal layer is a metal laminated structure including at least one of tungsten and molybdenum.
- the first metal layer is formed on one surface of the second metal layer by plating
- the third metal layer is the other surface of the second metal layer. It is preferable to be formed by plating.
- the thickness of the first metal layer and the third metal layer with respect to the sum of the thickness of the first metal layer, the thickness of the second metal layer, and the thickness of the third metal layer
- the sum ratio with the thickness of the metal layer is preferably 0.2 or more and 0.8 or less.
- the metal laminate structure of the present invention includes a fourth metal layer disposed on the side of the first metal layer opposite to the second metal layer and a second metal of the third metal layer. It is preferable that the fifth metal layer is provided on the side opposite to the layer installation side, and that the fourth metal layer and the fifth metal layer each contain copper.
- the thickness of the first metal layer, the thickness of the second metal layer, the thickness of the third metal layer, the thickness of the fourth metal layer, and the thickness of the fifth metal layer is preferably 0.2 or more and 0.8 or less.
- cobalt is contained between the first metal layer and the fourth metal layer and between at least one of the third metal layer and the fifth metal layer. It is preferable to include a layer.
- the thickness of the cobalt-containing layer is preferably 0.05 ⁇ m or more and 3 ⁇ m or less.
- the total thickness of the metal laminate structure is 20 ⁇ m or more and 400 ⁇ m or less.
- the metal laminated structure of this invention is used for a heat sink.
- the present invention is a method for producing any one of the above-described metal laminated structures, the step of forming the first metal layer on one surface of the second metal layer by plating, Forming a third metal layer on the other surface of the metal layer by plating.
- the plating is preferably molten salt bath plating.
- the present invention it is possible to provide a metal laminate structure and a method for producing the metal laminate structure that can be made thin and can be efficiently manufactured.
- FIG. 2 is a schematic configuration diagram of an apparatus used in Examples 1 to 4.
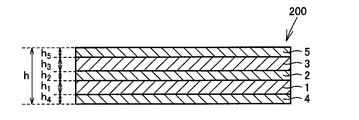
- the metal laminated structure 100 includes a first metal layer 1, a second metal layer 2 installed on the first metal layer 1, and a third metal installed on the second metal layer 2. It is comprised from the laminated structure with the metal layer 3 of this. That is, in the metal laminate structure 100, the first metal layer 1 is disposed on one surface of the second metal layer 2, and the third metal layer 3 is disposed on the other surface of the second metal layer 2. is set up.
- the first metal layer 1 is a metal layer made of a metal containing at least one of tungsten and molybdenum.
- the thickness of the metal multilayer structure 100 is reduced to make the metal multilayer structure 100 efficient.
- the first metal layer 1 is preferably a tungsten layer or a molybdenum layer formed by plating.
- the second metal layer 2 is a metal layer made of a metal containing copper, and in particular, from the viewpoint of efficiently manufacturing the metal multilayer structure 100 by reducing the thickness of the metal multilayer structure 100,
- the second metal layer 2 is preferably a copper thin plate such as a copper foil.
- the third metal layer 3 is a metal layer made of a metal containing at least one of tungsten and molybdenum.
- the metal multilayer structure 100 is efficiently manufactured by reducing the thickness of the metal multilayer structure 100.
- the third metal layer 3 is preferably a tungsten layer or a molybdenum layer formed by plating.
- the overall thickness h of the metal laminated structure 100 is preferably 20 ⁇ m or more and 400 ⁇ m or less.
- the thickness h of the metal multilayer structure 100 is 20 ⁇ m or more and 400 ⁇ m or less, the tendency that the metal multilayer structure 100 can be efficiently manufactured by reducing the thickness of the metal multilayer structure 100 increases.
- the sum of the metal laminated structure 100, a first thickness h 1 and the second thickness h 3 of the thick h 2 and the third metal layer 3 a metal layer and second metal layer 1 (h 1 + ratio of the sum (h 1 + h 3) and h 2 + h 3 first thickness of the metal layer 1 with respect to) h 1 and the thickness h 3 of the third metal layer 3 [(h 1 + h 3 ) / (h 1 + h 2 + h 3 )] is preferably 0.2 or more and 0.8 or less. When the above ratio is 0.2 or more and 0.8 or less, the linear expansion of the metal laminated structure 100 does not become too large and the thermal conductivity does not become too small.
- the heat dissipation function of the metal multilayer structure 100 can be sufficiently exhibited without causing a large difference in thermal expansion between the metal multilayer structure 100 and the semiconductor substrate. There is a tendency to be able to.
- the central portion in the thickness direction of the metal multilayer structure 100 (in this example, a portion that is 1 ⁇ 2 of the total thickness h of the metal multilayer structure 100). It is preferable that the upper part and the lower part from the central part in the thickness direction of the laminated structure are symmetrical with respect to the central part in the thickness direction of the laminated structure.
- “symmetry” means that the material and thickness of a layer that appears when the metal layered structure 100 proceeds from the center in the thickness direction to the upper end surface in the vertical direction are the same. It is a concept that includes not only the case where it is completely the same as the case where it proceeds downward from the center in the thickness direction to the lower end surface, but also the case where it is equivalent.
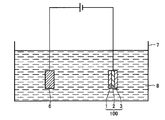
- a molten salt bath 8 containing at least one of tungsten and molybdenum is accommodated in a container 7.
- the molten salt bath 8 is not particularly limited as long as at least one of tungsten and molybdenum can be deposited by electrolysis of the molten salt bath 8. A preferred configuration of the molten salt bath 8 will be described later.
- the counter electrode 6 can be used without particular limitation as long as it is a conductive electrode.
- a metal electrode or the like can be used.
- the copper foil as the second metal layer 2 is used as a cathode
- the counter electrode 6 is used as an anode
- a voltage is applied between the copper foil as the second metal layer 2 and the counter electrode 6 to melt.
- tungsten and / or molybdenum in the molten salt bath 8 is deposited on both sides of the copper foil as the second metal layer 2, respectively, and the first metal layer 1 and A third metal layer 3 is formed.
- the copper foil as the second metal layer 2 after the formation of the first metal layer 1 and the third metal layer 3 is taken out from the molten salt bath 8, and the first metal layer 1 and The molten salt bath 8 adhering to the third metal layer 3 is washed and removed. Then, for example, by washing with a predetermined acid, the oxide films formed on the surfaces of the first metal layer 1 and the third metal layer 3 are removed.
- the metal laminated structure 100 shown in FIG. 1 can be manufactured.
- metal laminated structure 100 shown in FIG. 1 can also be manufactured as follows, for example.
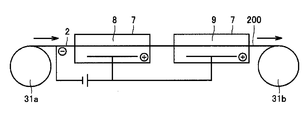
- the copper as the second metal layer 2 so that the copper foil as the second metal layer 2 passes through the molten salt bath 8 accommodated in the container 7.
- the foil is stretched between the first roll 31a and the second roll 31b.
- the copper foil as the second metal layer 2 is fed out from the first roll 31a, and the molten salt bath 8 is electrolyzed while the copper foil is continuously immersed in the molten salt bath 8 accommodated in the container 7.
- tungsten and / or molybdenum is deposited on both sides of the copper foil, and the metal laminated structure 100 is formed by molten salt bath plating.
- the metal laminated structure 100 formed by depositing tungsten and / or molybdenum on both surfaces of the copper foil is wound around the second roll 31b and collected.
- the metal multilayer structure 100 is continuously formed by continuously depositing tungsten and / or molybdenum on the surface of the second metal layer 2, the metal multilayer structure 100 is more efficient. Can be manufactured automatically.
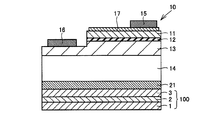
- FIG. 4 shows a schematic cross-sectional view of an example of an LED element which is an example of a semiconductor device using the metal laminated structure of the present invention.
- the LED element shown in FIG. 4 includes the metal laminate structure 100 shown in FIG. 1 and the LED structure 10 installed on the metal laminate structure 100, and the metal laminate structure 100 and the LED structure.
- the body 10 is joined by a joining layer 21.
- the LED structure 10 is disposed on the semiconductor substrate 14, the n-type semiconductor layer 13 disposed on the semiconductor substrate 14, the active layer 12 disposed on the n-type semiconductor layer 13, and the active layer 12.
- the LED structure 10 includes a p-type semiconductor layer 11, an n-type semiconductor layer 13, and an active layer 12, and the active layer 12 is disposed between the p-type semiconductor layer 11 and the n-type semiconductor layer 13.
- any structure that emits light from the active layer 12 by current injection can be used without particular limitation.
- a conventionally known LED structure can be used.
- the p-type semiconductor layer 11, the active layer 12 and the n-type semiconductor layer 13 are each a group III element (at least one selected from the group consisting of Al, In and Ga) and a group V. It is preferable to use a group III-V nitride semiconductor which is a compound with an element (nitrogen). In this case, blue light can be emitted from the active layer 12.
- the LED structure 10 capable of emitting blue light from the active layer 12
- a GaN substrate or a sapphire substrate is used as the semiconductor substrate 14 shown in FIG. 4, and p-type GaN is used as the p-type semiconductor layer 11.
- An LED structure using an undoped InGaN layer as the active layer 12 and an n-type GaN layer as the n-type semiconductor layer 13 can be used.
- the metal laminated structure of the present invention is not limited to the LED element, and can be applied to a semiconductor device other than the LED element such as a semiconductor laser element or a field effect transistor.
- a semiconductor substrate 14 used in the semiconductor device other than the LED structure 10 capable of emitting blue light from the active layer 12 for example, a silicon substrate, a silicon carbide substrate, a gallium arsenide substrate, or the like is used. Can do.
- the p-type semiconductor layer 11 is a semiconductor layer having a p-type conductivity type doped with a p-type impurity
- the n-type semiconductor layer 13 is an n-type conductivity type doped with an n-type impurity. Needless to say, this is a semiconductor layer.
- the active layer 12 may have either a p-type or n-type conductivity type, and is an undoped semiconductor layer in which neither p-type impurity nor n-type impurity is doped. Also good.
- the semiconductor substrate 14 and the n-type semiconductor layer 13 between the n-type semiconductor layer 13 and the active layer 12, between the active layer 12 and the p-type semiconductor layer 11, and between the p-type semiconductor layer 11 and the translucent electrode.
- Other layers may be included between at least one of the layers 17, between the translucent electrode 17 and the p-electrode 15, and between the n-type semiconductor layer 13 and the n-electrode 16.
- the bonding layer 21 for example, a layer made of a conductive material having a higher thermal conductivity than eutectic solder can be used.
- a metal that has low electrical resistance, high thermal conductivity, and is difficult to oxidize and in particular, at least one selected from the group consisting of gold, silver, copper, and nickel It is more preferable to use a layer containing.
- the n-electrode 16 of the LED element having the above configuration is used as a cathode, the p-electrode 15 is used as an anode, and a voltage is applied between these electrodes, whereby the p-electrode 15 to the n-electrode 16 are formed inside the LED structure 10.
- Current is passed toward Thereby, light can be generated in the active layer 12 between the p-type semiconductor layer 11 and the n-type semiconductor layer 13 of the LED structure 10.
- the LED element of the structure shown in FIG. 4 can be manufactured as follows, for example.
- the n-type semiconductor layer 13 and the active layer are formed on the surface of the semiconductor substrate 14 as shown in the schematic cross-sectional view of FIG.
- Layer 12 and p-type semiconductor layer 11 are formed by epitaxial growth in this order, for example, by MOCVD.
- a semi-transparent electrode is formed on the p-type semiconductor layer 11 by using, for example, a lift-off method. 17 and p electrode 15 are formed, and n electrode 16 is formed on n type semiconductor layer 13.
- the metal laminated structure 100 is bonded to the back surface of the semiconductor substrate 14 after the formation of the p-electrode 15 and the n-electrode 16 by the bonding layer 21.
- the semiconductor substrate 14 after the formation of the bonding layer 21 is cut by, for example, a circular rotary blade or the like to divide into individual LED elements having a schematic cross section shown in FIG. As described above, the LED element having the configuration shown in FIG. 4 can be obtained.
- the metal laminated structure 100 having a thickness that is significantly thinner than the conventional one can be used as a heat sink of the semiconductor device, the material cost of the metal laminated structure 100 can be reduced, Since the metal laminated structure 100 can be easily cut when divided into elements, workability is improved. Furthermore, since the thickness of the metal laminated structure 100 can be reduced, the thickness of the semiconductor device itself can also be reduced.
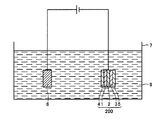
- the metal laminated structure 200 includes a fourth metal layer 4 installed on the opposite side of the first metal layer 1 to the installation side of the second metal layer 2, and the third metal layer 3 of the third metal layer 3.
- the fifth metal layer 5 is provided on the side opposite to the installation side of the second metal layer 2.
- the fourth metal layer 4 and the fifth metal layer 5 for example, a metal layer made of a metal containing copper can be used.
- the total thickness h of the metal laminated structure 200 is preferably 20 ⁇ m or more and 400 ⁇ m or less.
- the thickness h of the metal multilayer structure 200 is 20 ⁇ m or more and 400 ⁇ m or less, the tendency that the metal multilayer structure 200 can be efficiently manufactured by reducing the thickness of the metal multilayer structure 200 increases.
- the first metal layer 1 of thickness h 1 and the second thickness h 2 of the metal layer 2 and the third thickness h 3 of the metal layer 3 fourth metal layer 4 of the thickness h 4 and the thickness h 1 of the sum (h 1 + h 2 + h 3 + h 4 + h 5) the first metal layer to 1 between the thickness h 5 of the fifth metal layer 5 third metal the ratio of the sum of the thickness h 3 of the layer 3 (h 1 + h 3) [(h 1 + h 3) / (h 1 + h 2 + h 3 + h 4 + h 5)] is 0.2 to 0.8 It is preferable.
- the linear expansion of the metal laminated structure 200 does not become too large and the thermal conductivity does not become too small.
- the heat dissipation function of the metal multilayer structure 200 can be sufficiently exhibited without causing a large difference in thermal expansion between the metal multilayer structure 200 and the semiconductor substrate. There is a tendency to be able to.
- the central portion in the thickness direction of the metal multilayer structure 200 (in this example, a portion that is 1 ⁇ 2 of the total thickness h of the metal multilayer structure 200). It is preferable that the upper part and the lower part from the central part in the thickness direction of the laminated structure are symmetrical with respect to the central part in the thickness direction of the laminated structure.
- the metal laminated structure 200 shown in FIG. 5 can be manufactured as follows, for example.
- tungsten and / or molybdenum in the molten salt bath 8 is deposited on both surfaces of the second metal layer 2 such as a copper foil, respectively, and the first metal is obtained by molten salt bath plating.
- Layer 1 and third metal layer 3 are formed.
- the second metal layer 2 after the formation of the first metal layer 1 and the third metal layer 3 is taken out from the molten salt bath 8, and the first metal layer 1 and the third metal layer 1 are removed with, for example, ion exchange water.
- the molten salt bath 8 adhering to the metal layer 3 is washed and removed. Then, for example, by washing with a predetermined acid, the oxide films formed on the surfaces of the first metal layer 1 and the third metal layer 3 are removed.
- the second metal layer 2 after the formation of the first metal layer 1 and the third metal layer 3 in the electroplating solution 9 accommodated in the container 7.
- the counter electrode 6 is immersed.
- the electroplating solution 9 includes metal atoms that constitute the fourth metal layer 4 and the fifth metal layer 5, and the metal that constitutes the fourth metal layer 4 and the fifth metal layer 5. Is not particularly limited as long as it can be deposited by electrolysis of the electroplating solution 9.
- the electroplating solution 9 For example, a commercially available copper sulfate plating solution can be used.
- the second metal layer 2 is used as a cathode and the counter electrode 6 is used as an anode
- a voltage is applied between the second metal layer 2 and the counter electrode 6 to electrolyze the electroplating solution 9.
- copper in the electroplating solution 9 is deposited on the surface of the first metal layer 1 and the surface of the third metal layer 3 to form the fourth metal layer 4 and the fifth metal layer 5.
- the metal laminated structure 200 is produced.
- the metal laminated structure 200 after the formation of the fourth metal layer 4 and the fifth metal layer 5 is taken out from the electroplating solution 9, and the fourth metal layer 4 and the fifth metal, for example, with ion exchange water or the like.
- the electroplating solution 9 adhering to the layer 5 is removed by washing, and then the oxidation formed on the respective surfaces of the fourth metal layer 4 and the fifth metal layer 5 by washing with a predetermined acid, for example.
- the film can be removed.
- the metal laminated structure 200 shown in FIG. 5 can be manufactured.
- metal laminated structure 200 shown in FIG. 5 can also be manufactured as follows, for example.
- the copper foil as the second metal layer 2 passes through the molten salt bath 8 accommodated in the container 7 and the electroplating solution 9 accommodated in the container 7, respectively. As described above, the copper foil is stretched between the first roll 31a and the second roll 31b.
- the copper foil is fed out from the first roll 31a and electrolyzed in the molten salt bath 8 while continuously immersing the copper foil in the molten salt bath 8 accommodated in the container 7, respectively.
- Tungsten and / or molybdenum is deposited, and the first metal layer 1 and the third metal layer 3 are respectively formed on both sides of the copper foil by molten salt bath plating.
- the copper foil after forming the first metal layer 1 and the third metal layer 3 in the electroplating solution 9 accommodated in the container 7 is replaced with the copper foil in the electroplating solution 9 accommodated in the container 7.
- the electroplating solution 9 is electrolyzed while being continuously immersed.
- copper is deposited on the surfaces of the first metal layer 1 and the third metal layer 3, respectively, and the fourth metal is deposited on the surfaces of the first metal layer 1 and the third metal layer 3 by electroplating.
- the layer 4 and the fifth metal layer 5 are formed to form a metal laminated structure 200.
- the metal laminated structure 200 is wound around the second roll 31b and collected.
- the 4th metal layer 4 and the 5th metal layer 5 were each formed using the electroplating liquid 9, but the formation method of the 4th metal layer 4 and the 5th metal layer 5 is these. Needless to say, it is not limited to.
- the metal multilayer structure 200 can also be formed by forming the fourth metal layer 4 and the fifth metal layer 5 by a conventionally known vapor phase method such as sputtering.
- the fourth metal layer 4 and the fifth metal layer 5 may be formed by combining, for example, formation of the electroplating solution by electrolysis and formation by a vapor phase method such as a sputtering method.
- the metal laminated structure is not limited to the above-described three-layer or five-layer structure, and the first metal layer 1, the second metal layer 2, and the third metal layer 3 are the same. It may be included in the order.
- a metal layer made of nickel or the like is formed on the surface of the fourth metal layer 4 of the metal multilayer structure 200 opposite to the installation side of the first metal layer 1. 41 may be provided.
- cobalt containing cobalt is included between the first metal layer 1 and the fourth metal layer 4 and / or between the third metal layer 3 and the fifth metal layer 5 of the metal laminated structure 200. Layers may be installed.
- the metal laminated structure 200 including the cobalt-containing layer can be manufactured, for example, as follows.
- the surface of the first metal layer 1 and the third metal layer 3 are immersed.
- the surface of the metal layer 3 is degreased.
- the first metal layer 1 and the third metal layer 3 are immersed in an alkaline aqueous solution as an anode and electrolysis is performed, so that the surface of the first metal layer 1 and the surface of the third metal layer 3 are removed. Each oxide film is removed.
- the first metal layer 1 and the third metal layer 3 after the removal of the oxide film are immersed in a cobalt plating solution made of a cobalt sulfate aqueous solution or the like as a cathode, and electrolysis is performed. Cobalt is deposited on the surface of the layer 1 and the surface of the third metal layer 3 to form a cobalt-containing layer.
- the cobalt layer formed above is immersed in a copper sulfate plating solution as a cathode and electrolysis is performed, thereby depositing copper on the surface of the cobalt layer, and the fourth metal layer 4 and the fifth metal layer 5 made of copper. Each metal layer 5 is formed.
- the cobalt-containing layers are formed between the first metal layer 1 and the fourth metal layer 4 and between the third metal layer 3 and the fifth metal layer 5 of the metal multilayer structure 200, respectively. be able to.
- molten salt bath 8 used in the present invention for example, potassium fluoride (KF), boron oxide (B 2 O 3 ), and tungsten oxide (WO 3 ) are mixed at a molar ratio of, for example, 67: 26: 7.
- KF potassium fluoride
- B 2 O 3 boron oxide
- WO 3 tungsten oxide
- a molten salt bath produced by melting the mixture can be used.
- the molten salt bath 8 for example, KF, K 2 MoO 4, and B 2 O 3 are used, for example, 81: 9.
- a molten salt bath prepared by melting a mixture mixed at a molar ratio of 10 at a temperature of about 850 ° C. can be used.
- the molten salt bath 8 may be, for example, KF, WO 3 , K 2 MoO 4 , B 2 O 3 , It is also possible to use a molten salt bath or the like prepared by melting a mixture prepared by mixing at a molar ratio of, for example, 80: 4: 5: 10 at a temperature of about 850 ° C., for example.
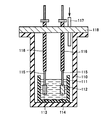
- the KF powder, the B 2 O 3 powder and the WO 3 powder after drying are put into a SiC crucible 111 which has been dried at 500 ° C. for two days or more, respectively.
- the crucible 111 was sealed in a quartz vacuum-resistant container 110.
- the crucible 111 was held at 500 ° C. with the cover 118 made of SUS316L in the upper opening of the vacuum resistant container 110, and the inside of the vacuum resistant container 110 was evacuated for one day or more.
- the molten salt bath precursor 112 was produced by melting.
- a rod-shaped electrode including a tungsten plate 113 (surface: 20 cm 2 ) as an anode and a rod-shaped electrode including a nickel plate 114 (surface: 20 cm 2 ) as a cathode are respectively inserted from openings provided in the lid 118. Then, the tungsten plate 113 and the nickel plate 114 were respectively immersed in the molten salt bath precursor 112 in the crucible 111.
- lead wires 115 are connected to the tungsten plate 113 and the nickel plate 114, respectively, and tungsten wires are used for the lead wires 115 inside the vacuum resistant vessel 110.
- a copper wire was used as the external lead wire 115.
- at least a part of the lead wire 115 was covered with an alumina covering material 116.
- the molten salt bath precursor 112 was electrolyzed to precipitate impurities on the nickel plate 114, thereby removing impurities from the molten salt bath precursor 112 to prepare a molten salt bath.
- a current having a current density of 3 A / dm 2 was passed between the tungsten plate 113 and the copper foil for 17 minutes. Then, constant current electrolysis of the molten salt bath was performed, and tungsten was deposited on both sides of the copper foil to form a tungsten layer having a thickness of 5 ⁇ m. Then, the copper foil after forming the tungsten layer is taken out from the apparatus shown in FIG. 9, and the surface of the tungsten layer is removed by washing the molten salt bath adhering to the tungsten layer with ion exchange water, and then washed with an acid. The heat sink of Example 1 was produced by removing the oxide film formed on the surface of the tungsten layer.
- Example 1 And about the heat sink of Example 1, the linear expansion coefficient (ppm / degreeC) to a horizontal direction was measured. The results are shown in Table 1. The linear expansion coefficient (ppm / ° C.) was measured with a thermomechanical analyzer (TMA), measured from room temperature to 150 ° C., and the average value was calculated.
- TMA thermomechanical analyzer
- a constant current electrolysis of the molten salt bath is performed by flowing a current having a current density of 3 A / dm 2 for 136 minutes between the copper foil and the tungsten plate 113 of the apparatus shown in FIG.
- a heat sink of Example 2 was produced in the same manner as Example 1 except that tungsten was deposited on both surfaces of the copper foil to form a 40 ⁇ m thick tungsten layer.
- Example 3 A heat sink of Example 3 was produced in the same manner as Example 1 except that a copper foil having a thickness of 10 ⁇ m was used. And also about the heat sink of Example 3, it carried out similarly to Example 1, and measured the linear expansion coefficient (ppm / degreeC) to a horizontal direction. The results are shown in Table 1.
- the copper foil after the tungsten layer is formed is taken out from the apparatus shown in FIG. 9, and the surface of the tungsten layer is removed by washing the molten salt bath adhering to the tungsten layer with ion exchange water, and then washed with an acid. As a result, the oxide film formed on the surface of the tungsten layer was removed.
- the surface of the tungsten layer after removal of the oxide film was washed by immersing it in an alkali degreasing solution (A screen A220 manufactured by Okuno Pharmaceutical Co., Ltd.) at 50 ° C. for 20 minutes.
- an alkali degreasing solution A screen A220 manufactured by Okuno Pharmaceutical Co., Ltd.
- each of the oxide layers was removed from the surface of the tungsten layer by performing immersion (alkali anodic electrolysis) by immersing the washed tungsten layer as an anode in an alkaline aqueous solution.
- an anode made of a single cobalt plate was immersed in a cobalt plating solution made of a cobalt sulfate aqueous solution, and the copper foil after alkaline anode electrolysis was immersed as a cathode so as to face the anode.
- a cobalt sulfate aqueous solution in which 200 g of cobalt sulfate and 100 g of sulfuric acid were dissolved in water per liter of the cobalt plating solution was used.
- cobalt is deposited on each surface of the tungsten layer of the copper foil after the formation of the tungsten layer as the cathode, so that each of the tungsten layers on both sides of the copper foil is deposited.
- a cobalt layer having a thickness of 0.5 ⁇ m was formed on the surface.
- the copper foil after the formation of the cobalt layer is taken out of the cobalt plating solution, and the cobalt plating solution adhering to the tungsten layer is washed and removed with ion exchange water, and then washed with an acid on the surface of the tungsten layer.
- the formed oxide film was removed.
- the foil was immersed so as to face the counter electrode.
- the heat sinks of Examples 1 to 4 manufactured as described above can be manufactured as thin as 100 ⁇ m or less as a whole, and can be efficiently manufactured by electroplating using a plating solution. It was.
- the tungsten layer was added to the sum of the total thickness of the copper layer (including the thickness of the copper foil) and the total thickness of the tungsten layer. Since the ratio of the total thickness was 0.2 or more and 0.8 or less, it was confirmed that the heat sink was an excellent heat sink in which linear expansion did not become too large and thermal conductivity did not become too small.
- a mixture is prepared by mixing potassium fluoride (KF) powder, boron oxide (B 2 O 3 ) powder, and tungsten oxide (WO 3 ) powder in a molar ratio of 67: 26: 7, and the mixture is made of SiC.
- KF potassium fluoride
- B 2 O 3 boron oxide
- WO 3 tungsten oxide
- potassium fluoride (KF) powder, boron oxide (B 2 O 3 ) powder and tungsten oxide (WO 3 ) powder are each weighed in a glove box in an Ar (argon) atmosphere, and SiC contained in the same glove box. It was put into a made crucible.
- the above mixture was melted by heating a SiC crucible charged with the above mixture to 850 ° C. using a mantle heater to prepare a molten salt bath.
- a copper foil (cathode) having a thickness of 40 ⁇ m was immersed in the molten salt bath together with the counter electrode (anode) made of a tungsten plate so as to face the counter electrode.
- a nickel wire was welded to each of the anode and the cathode, and a current could be supplied from the nickel wire between the anode and the cathode.
- tungsten was deposited on the surface of the copper foil serving as the cathode to form a tungsten layer made of tungsten precipitates with a thickness of 30 ⁇ m.
- the copper foil after the formation of the tungsten layer is taken out from the molten salt bath, and the molten salt bath adhering to the tungsten layer is washed and removed with ion exchange water, and then the acid is removed.
- the oxide film formed on the surface of the tungsten layer was removed by washing with.
- the heat sink of Example 5 was produced by the above.
- Example 5 produced as described above can be produced with a total thickness as thin as 100 ⁇ m and can be produced efficiently by molten salt bath plating.
- the ratio of the total thickness of the tungsten layer to the sum of the total thickness of the copper layer (including the thickness of the copper foil) and the total thickness of the tungsten layer is 0. 6 and in the range of 0.2 or more and 0.8 or less, it was confirmed that this was an excellent heat sink in which linear expansion did not become too large and thermal conductivity did not become too small.
- each of the five wafers was manufactured as follows. First, an n-type GaN layer, an undoped InGaN active layer, and a p-type GaN layer are epitaxially grown in this order by MOCVD on the surface of a sapphire substrate having a circular surface with a diameter of 100 mm and a thickness of 100 ⁇ m. It was.
- part of each of the n-type GaN layer, the undoped InGaN active layer, and the p-type GaN layer was removed by photoetching until a part of the surface of the n-type GaN layer was exposed.
- the wafers described above were produced by forming an n-electrode on the n-type GaN layer, a semi-transparent electrode on the p-type GaN layer, and a p-electrode on the semi-transparent electrode by lift-off.
- each of the heat sinks of Examples 1 to 5 produced above was joined to the back surface of the wafer produced above on the side opposite to the LED structure formation side by eutectic solder, and 10 mm ⁇ 10 mm with a circular rotary blade.
- the LED elements of Examples 1 to 5 were obtained by dividing into LED elements of a size having a square surface.
- a copper plate and a tungsten plate are joined by pressure welding, and a heat sink composed of a laminated structure of copper (20 ⁇ m) / tungsten (60 ⁇ m) / copper (20 ⁇ m) having a total thickness of 1 mm and the LED structure are bonded by the above eutectic solder.
- a comparative LED element was produced in the same manner as described above except that the LED element was formed by bonding.
- the LED elements of Examples 1 to 5 are the LEDs of the comparative example. It was confirmed that the material cost of the heat sink can be reduced as compared with the element, and the workability is improved because the wafer can be easily cut with a circular rotary blade.
- the metal laminated structure and the method for producing the metal laminated structure of the present invention may be used for a heat sink of a semiconductor device, for example.
- SYMBOLS 1 1st metal layer, 2nd 2nd metal layer, 3rd metal layer, 4th 4th metal layer, 5th 5th metal layer, 6 counter electrode, 7 container, 8 molten salt, 9 electroplating liquid
- SYMBOLS 10 LED structure, 11 p-type semiconductor layer, 12 active layer, 13 n-type semiconductor layer, 14 semiconductor substrate, 15 p electrode, 16 n electrode, 17 translucent electrode, 31a 1st roll, 31b 2nd roll , 41 bonding layer, 100, 200 metal laminated structure, 110 vacuum resistant container, 111 Crucible, 112 Molten salt bath precursor, 113 Tungsten plate, 114 Nickel plate, 115 Lead wire, 116 Coating material, 117 Gas inlet, 118 Lid
Abstract
Description
さらに、本発明は、上記のいずれかの金属積層構造体を製造する方法であって、第2の金属層の一方の表面上に第1の金属層をめっきにより形成する工程と、第2の金属層の他方の表面上に第3の金属層をめっきにより形成する工程とを含む金属積層構造体の製造方法である。
図1に、本発明の金属積層構造体の一例の模式的な断面図を示す。ここで、金属積層構造体100は、第1の金属層1と、第1の金属層1上に設置された第2の金属層2と、第2の金属層2上に設置された第3の金属層3との積層構造体から構成されている。すなわち、金属積層構造体100において、第1の金属層1は第2の金属層2の一方の表面上に設置され、第3の金属層3は第2の金属層2の他方の表面上に設置されている。
以下、図1に示す金属積層構造体100の製造方法の一例について説明するが、本発明の金属積層構造体の製造方法はこれに限定されるものでないことは言うまでもない。
図4に、本発明の金属積層構造体を用いた半導体装置の一例であるLED素子の一例の模式的な断面図を示す。ここで、図4に示すLED素子は、図1に示す金属積層構造体100と、金属積層構造体100上に設置されたLED構造体10とを備えており、金属積層構造体100とLED構造体10とは接合層21によって接合されている。
図5に、本発明の金属積層構造体の他の一例の模式的な断面図を示す。ここで、金属積層構造体200は、第1の金属層1の第2の金属層2の設置側とは反対側に設置された第4の金属層4と、第3の金属層3の第2の金属層2の設置側とは反対側に設置された第5の金属層5と、を備えていることに特徴がある。
また、上記においては、電気めっき液9を用いて第4の金属層4および第5の金属層5をそれぞれ形成したが、第4の金属層4および第5の金属層5の形成方法はこれらに限定されないことは言うまでもない。
本発明に用いられる溶融塩浴8としては、たとえば、フッ化カリウム(KF)と酸化ホウ素(B2O3)と酸化タングステン(WO3)とをたとえば67:26:7のモル比で混合した混合物を溶融して作製した溶融塩浴などを用いることができる。
KF粉末319gおよびWO3粉末133gをそれぞれ耐圧容器に封入した後に、耐圧容器を500℃に保持し、耐圧容器の内部を2日間以上真空引きすることによってKF粉末およびWO3粉末をそれぞれ乾燥させた。
厚さ20μmの銅箔を用いて、当該銅箔と図9に示す装置のタングステン板113との間に3A/dm2の電流密度の電流を136分間流して溶融塩浴の定電流電解を行なうことによって、銅箔の両面にそれぞれタングステンを析出させて厚さ40μmのタングステン層を形成したこと以外は実施例1と同様にして、実施例2のヒートシンクを作製した。
厚さ10μmの銅箔を用いたこと以外は実施例1と同様にして、実施例3のヒートシンクを作製した。そして、実施例3のヒートシンクについても、実施例1と同様にして、水平方向への線膨張係数(ppm/℃)を測定した。その結果を表1に示す。
まず、厚さ100μmの銅箔と図9に示す装置のタングステン板113との間に3A/dm2の電流密度の電流を340分間流して溶融塩浴の定電流電解を行なったこと以外は実施例1と同様にして、銅箔の両面にそれぞれタングステンを析出させて厚さ100μmのタングステン層を形成した。
まず、フッ化カリウム(KF)粉末と酸化ホウ素(B2O3)粉末と酸化タングステン(WO3)粉末とを67:26:7のモル比で混合した混合物を作製し、その混合物をSiC製の坩堝(アズワン(株)製)に投入した。
次に、サファイア基板の一方の表面上にLED構造体が形成されたウエハを5枚作製した。
銅板およびタングステン板を圧接により接合して銅(20μm)/タングステン(60μm)/銅(20μm)の積層構造体からなる全体の厚さが1mmのヒートシンクとLED構造体とを上記の共晶半田により接合してLED素子を形成したこと以外は上記と同様にして比較例のLED素子を作製した。
坩堝、112 溶融塩浴前駆体、113 タングステン板、114 ニッケル板、115 リード線、116 被覆材、117 ガス導入口、118 蓋。
Claims (11)
- 第1の金属層(1)と、第2の金属層(2)と、第3の金属層(3)と、を備え、
前記第1の金属層(1)は前記第2の金属層(2)の一方の表面上に設置され、
前記第3の金属層(3)は前記第2の金属層(2)の他方の表面上に設置されており、
前記第1の金属層(1)は、タングステンおよびモリブデンの少なくとも一方を含み、
前記第2の金属層(2)は、銅を含み、
前記第3の金属層(3)は、タングステンおよびモリブデンの少なくとも一方を含む、金属積層構造体(100,200)。 - 前記第1の金属層(1)は前記第2の金属層(2)の一方の表面上にめっきにより形成され、
前記第3の金属層(3)は前記第2の金属層(2)の他方の表面上にめっきにより形成されてなる、請求の範囲第1項に記載の金属積層構造体(100,200)。 - 前記第1の金属層(1)の厚さと前記第2の金属層(2)の厚さと前記第3の金属層(3)の厚さとの和に対する前記第1の金属層(1)の厚さと前記第3の金属層(3)の厚さとの和の比が0.2以上0.8以下である、請求の範囲第1項に記載の金属積層構造体(100,200)。
- 前記第1の金属層(1)の前記第2の金属層(2)の設置側とは反対側に設置された第4の金属層(4)と、
前記第3の金属層(3)の前記第2の金属層(2)の設置側とは反対側に設置された第5の金属層(5)と、を備え、
前記第4の金属層(4)および前記第5の金属層(5)はそれぞれ銅を含む、請求の範囲第1項に記載の金属積層構造体(100,200)。 - 前記第1の金属層(1)の厚さと前記第2の金属層(2)の厚さと前記第3の金属層(3)の厚さと前記第4の金属層(4)の厚さと前記第5の金属層(5)の厚さとの和に対する前記第1の金属層(1)の厚さと前記第3の金属層(3)の厚さとの和の比が0.2以上0.8以下である、請求の範囲第4項に記載の金属積層構造体(100,200)。
- 前記第1の金属層(1)と前記第4の金属層(4)との間、および前記第3の金属層(3)と前記第5の金属層(5)との間の少なくとも一方の間にコバルト含有層を含む、請求の範囲第4項に記載の金属積層構造体(100,200)。
- 前記コバルト含有層の厚さが0.05μm以上3μm以下である、請求の範囲第6項に記載の金属積層構造体(100,200)。
- 前記金属積層構造体(100,200)の全体の厚さが20μm以上400μm以下である、請求の範囲第1項に記載の金属積層構造体(100,200)。
- ヒートシンクに用いられる、請求の範囲第1項に記載の金属積層構造体(100,200)。
- 請求の範囲第1項に記載の金属積層構造体(100,200)を製造する方法であって、
前記第2の金属層(2)の一方の表面上に前記第1の金属層(1)をめっきにより形成する工程と、
前記第2の金属層(2)の他方の表面上に前記第3の金属層(3)をめっきにより形成する工程と、を含む、金属積層構造体(100,200)の製造方法。 - 前記めっきは溶融塩浴めっきである、請求の範囲第10項に記載の記載の金属積層構造体(100,200)の製造方法。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP10793965.4A EP2450182A4 (en) | 2009-06-30 | 2010-06-08 | METALLIC LAMINATE STRUCTURE AND PROCESS FOR PRODUCING METALLIC LAMINATE STRUCTURE |
| US13/381,703 US9199433B2 (en) | 2009-06-30 | 2010-06-08 | Metal laminated structure and method for producing the metal laminated structure |
| CN2010800297910A CN102470639A (zh) | 2009-06-30 | 2010-06-08 | 金属层压构件和制造所述金属层压构件的方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009-155159 | 2009-06-30 | ||
| JP2009155159A JP2011011366A (ja) | 2009-06-30 | 2009-06-30 | 金属積層構造体の製造方法 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011001795A1 true WO2011001795A1 (ja) | 2011-01-06 |
Family
ID=43410875
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2010/059695 WO2011001795A1 (ja) | 2009-06-30 | 2010-06-08 | 金属積層構造体および金属積層構造体の製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9199433B2 (ja) |
| EP (1) | EP2450182A4 (ja) |
| JP (1) | JP2011011366A (ja) |
| KR (1) | KR20120112353A (ja) |
| CN (1) | CN102470639A (ja) |
| TW (1) | TWI462825B (ja) |
| WO (1) | WO2011001795A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011102059A1 (ja) * | 2010-02-19 | 2011-08-25 | 住友電気工業株式会社 | 金属積層構造体および金属積層構造体の製造方法 |
| US9199433B2 (en) | 2009-06-30 | 2015-12-01 | Sumitomo Electric Industries, Ltd. | Metal laminated structure and method for producing the metal laminated structure |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012253125A (ja) * | 2011-06-01 | 2012-12-20 | Sumitomo Electric Ind Ltd | 半導体装置及び配線基板 |
| KR101669087B1 (ko) | 2011-07-29 | 2016-10-25 | 후루카와 덴키 고교 가부시키가이샤 | 전해 동합금박, 그 제조 방법, 그것의 제조에 이용하는 전해액, 그것을 이용한 2차 전지용 음극 집전체, 2차 전지 및 그 전극 |
| JP6233677B1 (ja) * | 2016-08-31 | 2017-11-22 | Jfe精密株式会社 | 放熱板及びその製造方法 |
| CN106868462A (zh) * | 2017-03-01 | 2017-06-20 | 东莞市航晨纳米材料有限公司 | 一种合金复合材料及其制备方法 |
| JP6786090B2 (ja) * | 2019-03-11 | 2020-11-18 | ザ グッドシステム コーポレーション | 放熱板材 |
| WO2020184773A1 (ko) * | 2019-03-11 | 2020-09-17 | 주식회사 더굿시스템 | 방열판재 |
| JP7444814B2 (ja) * | 2021-04-27 | 2024-03-06 | Ngkエレクトロデバイス株式会社 | パッケージ |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04230063A (ja) * | 1990-12-27 | 1992-08-19 | Aichi Steel Works Ltd | 多層ヒートシンク |
| JPH06268115A (ja) | 1993-03-15 | 1994-09-22 | Tokyo Tungsten Co Ltd | 半導体装置用放熱基板の製造方法 |
| JPH11284111A (ja) * | 1998-03-30 | 1999-10-15 | Sumitomo Special Metals Co Ltd | ヒートシンク部材及びその製造方法、並びにヒートシンク部材を用いた半導体パッケージ |
| JP2000124370A (ja) * | 1998-10-15 | 2000-04-28 | Sumitomo Metal Electronics Devices Inc | 半導体装置用パッケージ |
| JP2004249589A (ja) * | 2003-02-20 | 2004-09-09 | Toshiba Corp | 銅−モリブデン複合材料およびそれを用いたヒートシンク |
| JP2006100640A (ja) * | 2004-09-30 | 2006-04-13 | Hitachi Metals Ltd | セラミックス回路基板及びこれを用いたパワー半導体モジュール |
| WO2007046164A1 (ja) * | 2005-10-18 | 2007-04-26 | Eiki Tsushima | クラッド材およびその製造方法、クラッド材の成型方法、クラッド材を用いた放熱基板 |
| JP2007227458A (ja) * | 2006-02-21 | 2007-09-06 | Toyota Motor Corp | 熱電発電装置 |
| JP2007327081A (ja) * | 2006-06-06 | 2007-12-20 | Toyota Central Res & Dev Lab Inc | 接着方法、半導体装置の製造方法、及び半導体装置 |
| WO2008013279A1 (fr) * | 2006-07-28 | 2008-01-31 | Kyocera Corporation | Boîtier de stockage de composant électronique et dispositif électronique |
Family Cites Families (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4025997A (en) * | 1975-12-23 | 1977-05-31 | International Telephone & Telegraph Corporation | Ceramic mounting and heat sink device |
| US4371589A (en) * | 1976-08-24 | 1983-02-01 | Warner London Inc. | Process for depositing protective coating and articles produced |
| US4448854A (en) | 1980-10-30 | 1984-05-15 | The United States Of America As Represented By The United States Department Of Energy | Coherent multilayer crystals and method of making |
| JPH01305544A (ja) | 1988-06-03 | 1989-12-08 | Denki Kagaku Kogyo Kk | 半導体装置 |
| JPH0280589A (ja) | 1988-09-17 | 1990-03-20 | Nisshin Steel Co Ltd | 電気タングステンめっき浴およびその浴によるめっき方法 |
| JPH02256262A (ja) | 1988-12-14 | 1990-10-17 | Sumitomo Metal Ind Ltd | 半導体素子用ヒートシンク |
| US4950554A (en) * | 1989-05-30 | 1990-08-21 | Amax Inc. | Composite copper-molybdenum sheet |
| JPH03218054A (ja) | 1990-01-23 | 1991-09-25 | Mitsubishi Electric Corp | 発熱素子用基板 |
| JPH07211822A (ja) | 1994-01-24 | 1995-08-11 | Kyocera Corp | 半導体素子収納用パッケージ |
| US5445106A (en) | 1994-10-03 | 1995-08-29 | General Electric Company | Method for making high thermal conducting diamond |
| US5525815A (en) | 1994-10-03 | 1996-06-11 | General Electric Company | Diamond film structure with high thermal conductivity |
| JPH08222658A (ja) | 1995-02-17 | 1996-08-30 | Sumitomo Electric Ind Ltd | 半導体素子用パッケージ及びその製造方法 |
| JPH1012767A (ja) | 1996-06-19 | 1998-01-16 | Tokyo Tungsten Co Ltd | 積層構造放熱基板及びその製造方法 |
| JP3505704B2 (ja) | 1999-05-10 | 2004-03-15 | 株式会社アライドマテリアル | 放熱基板およびその製造方法 |
| JP2001024370A (ja) | 1999-07-13 | 2001-01-26 | Japan Aviation Electronics Industry Ltd | Zif型icソケット |
| US6352014B1 (en) * | 1999-12-15 | 2002-03-05 | International Business Machines Corporation | Method for making punches using multi-layer ceramic technology |
| US6965199B2 (en) * | 2001-03-27 | 2005-11-15 | The University Of North Carolina At Chapel Hill | Coated electrode with enhanced electron emission and ignition characteristics |
| JP2003037204A (ja) | 2001-07-25 | 2003-02-07 | Kyocera Corp | 半導体素子収納用パッケージ |
| FR2830328B1 (fr) * | 2001-09-28 | 2003-12-05 | Socomate Internat | Dispositif et procede d'analyse de la structure d'un materiau |
| JP2004088083A (ja) | 2002-06-25 | 2004-03-18 | Matsushita Electric Ind Co Ltd | 半導体発光素子、その製造方法及びその実装方法 |
| US20040140474A1 (en) | 2002-06-25 | 2004-07-22 | Matsushita Electric Industrial Co., Ltd. | Semiconductor light-emitting device, method for fabricating the same and method for bonding the same |
| AU2003262651A1 (en) | 2003-08-13 | 2005-03-10 | Clad Metals Llc | Bonded metal components having uniform thermal conductivity characteristics and method of making same |
| JP4104517B2 (ja) | 2003-09-26 | 2008-06-18 | 本田技研工業株式会社 | 摺動部材 |
| JP2005105354A (ja) | 2003-09-30 | 2005-04-21 | Honda Motor Co Ltd | 摺動部材 |
| US20080105553A1 (en) | 2004-10-01 | 2008-05-08 | Koji Nitta | Molten Salt Bath, Deposit Obtained Using The Molten Salt Bath, Method Of Manufacturing Metal Product, And Metal Product |
| JP5123462B2 (ja) | 2004-10-27 | 2013-01-23 | 住友電気工業株式会社 | 膜形成用配向基板および超電導線材ならびに膜形成用配向基板の製造方法 |
| US8318519B2 (en) | 2005-01-11 | 2012-11-27 | SemiLEDs Optoelectronics Co., Ltd. | Method for handling a semiconductor wafer assembly |
| US9390999B2 (en) | 2005-03-23 | 2016-07-12 | Noriaki Kawamura | Metal substrate/metal impregnated carbon composite material structure and method for manufacturing said structure |
| TWI254419B (en) | 2005-04-08 | 2006-05-01 | Univ Nat Chiao Tung | Compound semiconductor device having copper metallization |
| JP4711724B2 (ja) | 2005-04-19 | 2011-06-29 | 学校法人同志社 | 溶融塩めっき用電解浴及び該電解浴を用いた溶融塩めっき方法 |
| US7695808B2 (en) | 2005-11-07 | 2010-04-13 | 3M Innovative Properties Company | Thermal transfer coating |
| JP4930026B2 (ja) | 2006-12-13 | 2012-05-09 | 富士ゼロックス株式会社 | 積層体、無端状ベルト、定着装置及び画像形成装置 |
| JP2008214728A (ja) | 2007-03-07 | 2008-09-18 | Sumitomo Electric Ind Ltd | セラミックス膜の製造方法 |
| US20080298024A1 (en) | 2007-05-31 | 2008-12-04 | A.L.M.T. Corp. | Heat spreader and method for manufacturing the same, and semiconductor device |
| JP4883534B2 (ja) | 2008-03-26 | 2012-02-22 | 住友電気工業株式会社 | 溶融塩浴、溶融塩浴の製造方法およびタングステン析出物 |
| JP5568883B2 (ja) | 2009-03-27 | 2014-08-13 | 住友電気工業株式会社 | 溶融塩浴および溶融塩浴の製造方法 |
| JP2011011366A (ja) | 2009-06-30 | 2011-01-20 | Sumitomo Electric Ind Ltd | 金属積層構造体の製造方法 |
| JP5580772B2 (ja) | 2011-04-07 | 2014-08-27 | 住友電気工業株式会社 | 金属積層構造体の製造方法 |
-
2009
- 2009-06-30 JP JP2009155159A patent/JP2011011366A/ja active Pending
-
2010
- 2010-06-08 EP EP10793965.4A patent/EP2450182A4/en not_active Withdrawn
- 2010-06-08 US US13/381,703 patent/US9199433B2/en not_active Expired - Fee Related
- 2010-06-08 KR KR1020127001409A patent/KR20120112353A/ko not_active Application Discontinuation
- 2010-06-08 CN CN2010800297910A patent/CN102470639A/zh active Pending
- 2010-06-08 WO PCT/JP2010/059695 patent/WO2011001795A1/ja active Application Filing
- 2010-06-28 TW TW099120982A patent/TWI462825B/zh not_active IP Right Cessation
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04230063A (ja) * | 1990-12-27 | 1992-08-19 | Aichi Steel Works Ltd | 多層ヒートシンク |
| JPH06268115A (ja) | 1993-03-15 | 1994-09-22 | Tokyo Tungsten Co Ltd | 半導体装置用放熱基板の製造方法 |
| JPH11284111A (ja) * | 1998-03-30 | 1999-10-15 | Sumitomo Special Metals Co Ltd | ヒートシンク部材及びその製造方法、並びにヒートシンク部材を用いた半導体パッケージ |
| JP2000124370A (ja) * | 1998-10-15 | 2000-04-28 | Sumitomo Metal Electronics Devices Inc | 半導体装置用パッケージ |
| JP2004249589A (ja) * | 2003-02-20 | 2004-09-09 | Toshiba Corp | 銅−モリブデン複合材料およびそれを用いたヒートシンク |
| JP2006100640A (ja) * | 2004-09-30 | 2006-04-13 | Hitachi Metals Ltd | セラミックス回路基板及びこれを用いたパワー半導体モジュール |
| WO2007046164A1 (ja) * | 2005-10-18 | 2007-04-26 | Eiki Tsushima | クラッド材およびその製造方法、クラッド材の成型方法、クラッド材を用いた放熱基板 |
| JP2007227458A (ja) * | 2006-02-21 | 2007-09-06 | Toyota Motor Corp | 熱電発電装置 |
| JP2007327081A (ja) * | 2006-06-06 | 2007-12-20 | Toyota Central Res & Dev Lab Inc | 接着方法、半導体装置の製造方法、及び半導体装置 |
| WO2008013279A1 (fr) * | 2006-07-28 | 2008-01-31 | Kyocera Corporation | Boîtier de stockage de composant électronique et dispositif électronique |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2450182A4 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9199433B2 (en) | 2009-06-30 | 2015-12-01 | Sumitomo Electric Industries, Ltd. | Metal laminated structure and method for producing the metal laminated structure |
| WO2011102059A1 (ja) * | 2010-02-19 | 2011-08-25 | 住友電気工業株式会社 | 金属積層構造体および金属積層構造体の製造方法 |
| US8993121B2 (en) | 2010-02-19 | 2015-03-31 | Sumitomo Electric Industries, Ltd. | Metal laminated structure and method for producing the same |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2450182A4 (en) | 2016-07-20 |
| KR20120112353A (ko) | 2012-10-11 |
| EP2450182A1 (en) | 2012-05-09 |
| US20120100392A1 (en) | 2012-04-26 |
| TWI462825B (zh) | 2014-12-01 |
| CN102470639A (zh) | 2012-05-23 |
| US9199433B2 (en) | 2015-12-01 |
| TW201111164A (en) | 2011-04-01 |
| JP2011011366A (ja) | 2011-01-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2011001795A1 (ja) | 金属積層構造体および金属積層構造体の製造方法 | |
| US8993121B2 (en) | Metal laminated structure and method for producing the same | |
| JP4980594B2 (ja) | P型窒化ガリウム系化合物半導体の製造方法 | |
| JP2010074122A (ja) | Led用ヒートシンク、led用ヒートシンク前駆体、led素子、led用ヒートシンクの製造方法およびled素子の製造方法 | |
| JP4841909B2 (ja) | 窒化物系半導体発光素子 | |
| TW200834670A (en) | Process for producing III group nitride compound semiconductor, III group nitride compound semiconductor light emitting element, and lamp | |
| TW202037747A (zh) | 結晶性氧化物半導體 | |
| JP2007081312A (ja) | 窒化物系半導体発光素子の製造方法 | |
| KR100872276B1 (ko) | 수직구조 질화물 반도체 발광 소자 및 제조방법 | |
| JP4799975B2 (ja) | 窒化物系半導体発光素子及びその製造方法 | |
| JP2011014917A (ja) | 金属積層構造体 | |
| JP5580772B2 (ja) | 金属積層構造体の製造方法 | |
| JP5428520B2 (ja) | Led素子およびled素子の製造方法 | |
| WO2012060335A1 (ja) | 半導体素子、発光素子、及び半導体素子の製造方法 | |
| JP4799974B2 (ja) | 窒化物系半導体発光素子及びその製造方法 | |
| JP7061214B2 (ja) | 半導体積層体、半導体素子および半導体素子の製造方法 | |
| TWI251944B (en) | Resistance electrode structure, compound semiconductor light emitting device having the same, and LED lamp | |
| WO2022030114A1 (ja) | 半導体積層体、半導体素子および半導体素子の製造方法 | |
| JP2011176260A (ja) | Ledチップとリードフレームとの接合方法 | |
| JP2007088048A (ja) | 窒化物系半導体発光素子及びその製造方法 | |
| CN102593277B (zh) | 垂直式发光二极管及其制造方法 | |
| KR20150073029A (ko) | 오엘이디용 금속봉지재 및 그의 제조방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080029791.0 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10793965 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13381703 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010793965 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20127001409 Country of ref document: KR Kind code of ref document: A |