WO2004104055A1 - エチレン系重合体 - Google Patents
エチレン系重合体 Download PDFInfo
- Publication number
- WO2004104055A1 WO2004104055A1 PCT/JP2004/007124 JP2004007124W WO2004104055A1 WO 2004104055 A1 WO2004104055 A1 WO 2004104055A1 JP 2004007124 W JP2004007124 W JP 2004007124W WO 2004104055 A1 WO2004104055 A1 WO 2004104055A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- zirconium dichloride
- ethylene
- compound
- cyclopentagenenyl
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F210/00—Copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F210/16—Copolymers of ethene with alpha-alkenes, e.g. EP rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/65912—Component covered by group C08F4/64 containing a transition metal-carbon bond in combination with an organoaluminium compound
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/65916—Component covered by group C08F4/64 containing a transition metal-carbon bond supported on a carrier, e.g. silica, MgCl2, polymer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/64—Titanium, zirconium, hafnium or compounds thereof
- C08F4/659—Component covered by group C08F4/64 containing a transition metal-carbon bond
- C08F4/6592—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring
- C08F4/65922—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not
- C08F4/65927—Component covered by group C08F4/64 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not two cyclopentadienyl rings being mutually bridged
Definitions
- the present invention relates to a novel ethylene polymer, a polymerization catalyst and a polymerization method for producing the ethylene polymer, and uses of the ethylene polymer. Specifically, (1) an ethylene polymer satisfying specific properties, (2) at least one kind of a transition metal compound containing a salicylaldimine ligand and a transition metal compound containing a cyclopentagenil ligand, and The above ethylene using a polymerization catalyst in which at least one compound selected from an organometallic compound, an organoaluminumoxy compound, and a compound that forms an ion pair by reacting with a transition metal compound is supported on a solid support.
- the present invention relates to a method for producing a polymer, and (3) use of the ethylene polymer for a blow molded article, a pipe, and the like.
- Polyolefins such as polyethylene and polypropylene are environmentally friendly, clean materials composed of carbon and hydrogen, and have excellent workability and physical properties. Due to this characteristic, it has been used in a wide range of fields, such as automobiles, electrical equipment parts, food packaging, beverages, cosmetics, medical containers, civil engineering, and agricultural materials. In recent years, however, demands for physical properties of polyolefins have been diversified, and polyolefins having various properties have been desired. Further, further improvement in productivity is desired. In particular, when performing hollow molding or sheet molding, a high melt tension (hereinafter, sometimes referred to as melt tension ( ⁇ )) ⁇ swell ratio is required.
- melt tension ( ⁇ ) melt tension
- Polyethylene produced by a high-pressure method and polyethylene obtained by a Cr-based Phillips catalyst are known as ethylene polymers with good physical properties.
- the strength and impact strength are low due to the presence of a large amount of long chain branches. descend.
- these high-pressure polyethylenes have a flow activation energy, which is a measure of temperature dependence for resin fluidization during molding. There was a problem that it was expensive.
- ethylene based polymers such as ethylene polymer and ethylene / ⁇ -olefin copolymer
- a titanium compound and an organic aluminum compound are used as a polymerization catalyst for producing a polymer.
- Ziegler-type titanium-based catalysts comprising a catalyst compound are widely known. More recently
- Meta-opened catalysts comprising a compound (aluminoxane) have become known
- the swell ratio is 1.35 or less, which is not a sufficiently high value.
- the present inventors have also
- Japanese Unexamined Patent Publication No. 2002-105132 discloses that a long chain component
- a method for producing a polyolefin containing is disclosed. Force, even in this case,
- high-density polyethylene has been conventionally produced by a low-pressure method using a Ziegler-type medium, generally by a slurry polymerization method.
- high-density polyethylenes when producing high-density polyethylene having an arbitrary molecular weight distribution for the purpose of controlling moldability and physical properties, polymerization is carried out in multiple stages, and the polymer usually formed in each stage is produced. The polymerization is carried out by controlling the molecular weight and density of the polymer.
- a multi-stage slurry polymerization step consisting of a low-molecular-weight polyethylene polymerization step and a high-molecular-weight polyethylene polymerization step.
- the operation is complicated due to the multi-stage process and must use large amounts of hydrogen keep low molecular weight polyethylene production formation process, such as process ⁇ Pi co: 'it should be solved in strike surface is left. '
- the present inventors have conducted research on improving the melt tension as described above and rationalizing the process of polyolefin by a multi-stage polymerization method, and found that using a specific catalyst for polymerization of olefins, the length and introduction amount of long-chain branching were reduced. By controlling, they found polyolefins that exhibited excellent 'melt tension' and overcame the conventional resin deficiencies of low flow activation energy. Disclosure of the invention
- the ethylene polymer of the present invention is a copolymer obtained from ethylene and ⁇ -olefin having 3 to 10 carbon atoms, and satisfies the following requirements (1), (ii), (iii) and (iv) simultaneously. It is said that it is worth to do.
- melt flow rate [MFR 2 (g / 10min)] at 2.16 kg load in C shall be in the range of 0.01 to: 10.
- the activation energy of the flow-[Ea] force must be less than 30 (KJ / mol).
- (b-3) at least one compound selected from the group consisting of a compound and a force that reacts with the transition metal compound (Al) or (A2) to form an ion pair;
- (C) It is characterized by being preferably obtained by copolymerizing ethylene and ⁇ -olefin having 3 to 10 carbon atoms in the presence of a solid catalyst component supported on a solid carrier.
- the present invention relates to a blow molded article comprising the above-mentioned ethylene polymer.
- the blow molded articles they are suitably used for drums, large containers, large gasoline tanks, large industrial chemical cans or large bottle containers.
- the present invention also relates to a pipe or a pipe joint made of the ethylene polymer.
- FIG. 1 is a diagram in which the melt tension (MT) and the value of MFR2 of the ethylene polymers obtained in Examples and Comparative Examples are plotted.
- MT melt tension
- MFR2 the value of MFR2 of the ethylene polymers obtained in Examples and Comparative Examples are plotted.
- the ethylene polymer of the present invention is a copolymer obtained from ethylene and forefin having 3 to 10 carbon atoms, and simultaneously satisfies the following requirements), (ii), (iii) and (iv). It is characterized by that.
- melt flow rate [MFR2 (g / 10min)] at 2.16 kg load in C shall be in the range of 0.01 to: 10.
- the activation energy of the flow-[Ea] force must be less than 30 (KJ / mol).
- the ethylene polymer of the present invention contains, as an essential component, an ethylene homopolymer or a copolymer obtained from ethylene and ⁇ -olefin having 3 to 3 carbon atoms, that is, a structural unit derived from ethylene as an essential component. Also includes structural units derived from 3 to 10 ⁇ -olefins.
- the olefin having 3 to 10 carbon atoms may be one kind or two or more kinds.
- fins include, for example, propylene, 1-butene, 1-pentene, 3-methyl-1-butene, 1-hexene, 4-methyl-1- Pentane, 3-methyl-1-pentene, 1-otaten, 1-decene and the like.
- olefins it is preferable that at least one structural unit selected from 1-hexene, 4-methyl-1-pentene and 1-octene is included. It is particularly preferred to include a structural unit derived from 1-hexene.
- the structural unit derived from ⁇ -olefin having 3 to 10 carbon atoms is usually 0 to 5.0 mol%, preferably 0.05 to 1.0 mol%, particularly preferably 0.1 to 1.0 mol%. It is an ethylene polymer containing 0.5 mol%.
- the ethylene polymer of the present invention is characterized by simultaneously satisfying the above requirements ((ii), (iii) and (iv). Each requirement will be described in detail below.
- melt rate of the ethylene polymer of the present invention at 190 ° C. under a load of 2.16 kg [MFR 2 (g / 10 min)] is in the range of 0.01 to: 10.
- the ethylene polymer of the present invention has a melt flow rate and a melt tension [MT (g)] of the following inequality within the range of the melt flow rate [MFR 2 (g / 10min)] under the load of 2.16 kg.
- MT (g) melt flow rate and a melt tension [MT (g)] of the following inequality within the range of the melt flow rate [MFR 2 (g / 10min)] under the load of 2.16 kg.
- Eq-1 preferably satisfies the following inequality (Eq-2), particularly preferably the following inequality (Eq-3).
- the flow activation energy-[Ea] of the ethylene polymer of the present invention is less than 30 (KJ / mol), preferably 10 to 28 (KJ / mol), particularly preferably 20 to 28 (KJ / mol). .
- Ea is in such a range, it is advantageous in terms of excellent fluidity.
- the ethylene polymer of the present invention has a slur ratio of 1.36 or more, preferably 1.40 or more. When the swell ratio is in such a range, a uniform thickness distribution can be obtained when performing hollow molding. 'The ethylene polymer of the present invention preferably satisfies the following requirements (v) to (vii) in addition to the requirements') to (iv).
- the density of the ethylene polymer of the present invention is in the range of 910 to 970 (kg / m3), preferably 920 to 970 (kg / ms), and more preferably 930 to 970 (kgZm3).
- the intrinsic viscosity of the ethylene polymer of the present invention is in the range of 1.0 to 5.0 (dl / .g), preferably 1.5 to 3.0 (dl / g).
- the intrinsic viscosity ([7j] (dl / g)) of the ethylene polymer of the present invention and the melt flow rate [MFR 20 (g / lOmin)] at 190 ° C and 21.6 kg load are represented by the following inequality (Eq4 Satisfies. Further, it preferably satisfies the inequality (Eq-5). ':
- Melt flow rate at 190 ° C and 21.6kg load [MFR 20 (g / 10 min)] is usually in the range of 1 to 100 (g / 10 min), preferably 1 to 50 (g / 10 min), particularly preferably 2 to 30 (g / 10 min).
- the ethylene polymer of the present invention which satisfies the above requirements) to (iv), and preferably satisfies the parameter range defined by the above requirements (v) to (vii), can be produced under the production conditions per se described in Examples of the present application described below or It is possible to arbitrarily create different variations based on minor fluctuations of condition factors or these resin blends.
- Specific examples of the variation of the condition factors include the requirements of the catalyst components such as the structure of the transition metal compound [I] and [ ⁇ ] used, the ratio supported, the type of the carrier and the cocatalyst component used, the polymerization temperature, and the coexisting hydrogen.
- the polymerization conditions such as the amount of the molecular weight modifier such as the amount of the comonomer to be added.
- the range of physical properties can be expanded by combining with multistage polymerization. More specifically, by reducing the polymerization temperature, increasing the amount of comonomer added, or changing the structure of the transition metal compound [I] used, for example, the amount of long-chain branching that can be introduced into the polymer is reduced. It is possible to increase, in this case
- the activation energy (Ea) of the flow can be controlled by changing the combination of transition metal catalysts.
- it contains a transition metal compound having the structure of [I] and two cyclopentagenenyl skeletons as ligands. Ea can be suppressed to 30 KJ / mol or less when the compound is a group III transition metal compound.
- the ethylene polymer of the present invention includes :
- (C) It is preferably obtained by copolymerizing ethylene and the above-mentioned ⁇ -olefin having 3 to 10 carbon atoms in the presence of a polymerization catalyst supported on a solid carrier. .
- M represents a transition metal atom belonging to Group 4 of the periodic table, specifically, titanium, zirconium, or hafnium, and preferably zirconium.
- n represents an integer of 1 to 4, preferably 2, n is a number satisfying the valence of M, and R! to R 6 are a hydrogen atom and a halogen atom which may be the same or different from each other.
- X is a hydrogen atom, a halogen atom, a hydrocarbon group, an oxygen-containing group, a zeo-containing group, a nitrogen-containing group, a boron-containing group, an aluminum-containing group, a phosphorus-containing group, a halogen-containing group, a heterocyclic compound residue, silicon A group, a germanium-containing group, or a tin-containing group, and when n is 2 or more, a plurality of groups represented by X may be the same or different from each other, and a plurality of groups represented by X They may combine to form a ring.
- halogen atom examples include fluorine, chlorine, bromine, and iodine.
- hydrocarbon group examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, t-butyl, neopentyl, and n-hexyl.
- the hydrocarbon group may have a hydrogen atom substituted with a halogen, for example, a carbon atom number of 1 to 30, such as a trifluoromethinole group, a pentafluorophenol group, and a chlorophenyl group, preferably Represents 1 to 20 halogenated hydrocarbon groups. : ⁇
- the hydrocarbon group may be substituted with another hydrocarbon group, for example, an aryl group-substituted alkyl group such as a benzyl group, a Tamyl group, a 2,2-diphenylethyl group, and a triphenylmethyl group. And the like.
- the hydrocarbon group may be a heterocyclic compound residue; an alkoxy group, an aryloxy group, an ester group, an ether group, an acyl group, a carboxyl group, (2) a carbonate group, a hydroxy group, a peroxy group, or a carboxylic anhydride.
- Oxygen-containing groups such as chemical groups; amino groups, imino groups, amide groups, imide groups, hydrazino groups, hydrazono groups, nitro 'groups, nitroso groups, cyano groups, isocyano groups, cyanate ester groups, amidino groups, diazo groups
- Nitrogen-containing groups such as those in which an amino group or an amino group has become an ammonium salt; boron-containing groups such as a porandil group, a boranthryl group, or a diboranyl group; a mercapto group, a thioester group, a dithioester group, an alkylthio group, an arylthio group, a thioacyl group, or a thioacyl group.
- Ether group thiocyanate ester group, isothiocyanate ester group, sulfone ester ⁇ -containing groups such as ter, sulfonamide, thiocarboxyl, dithiocarboxyl, sulfo, sulfonyl, sulfinyl, sulfenyl; phosphorus-containing groups such as phosphide, phosphoryl, thiophosphoryl and phosphato groups , A silicon-containing group, a germanium-containing group, or a tin-containing group. '
- carbon atoms such as methyl group, ethyl group, n-propyl group, isopropyl group, n-butyl group, isobutyl group, sec-butyl group, tert-butyl group, neopentyl group and n-hexyl group
- a linear or branched alkyl group having 1 to 30, preferably 1 to 20 carbon atoms such as a fuel group, a naphthyl group, a biphenyl group, a terphenyl group, a phenanthryl group, and an anthracenyl group;
- an alkyl group or alkoxy group having 20 to 20 carbon atoms a substituted aryl group substituted with 1 to 5 substituents such as an aryl group or an aryloxy group having 6 to 30 carbon atoms, preferably 6 to 20 carbon atoms.
- oxygen-containing group examples include the same as those exemplified above.
- nitrogen-containing group examples include the same as those exemplified above.
- boron-containing group examples include the same as those exemplified above.
- zirconium-containing group examples include the same as those exemplified above.
- heterocyclic compound residues include residues such as nitrogen-containing compounds such as pyrrole, pyridine, pyrimidine, quinoline, and triazine; oxygen-containing compounds such as furan and pyran; sulfur-containing compounds such as thiophene; Examples include groups in which a substituent of the formula compound is further substituted with a substituent such as an alkyl group or an alkoxy group having 1 to 30, preferably 1 to 20 carbon atoms.
- Silicone-containing groups include silyl groups, siloxy groups, hydride-substituted silyl groups, hydrocarbon-substituted siloxy groups, etc., specifically, methylsilyl group, dimethyl / resilyl group, trimethylsilyl group, ethylsilyl group, And a triethylsilyl group, a diphenylmethylsilyl group, a triphenylsilyl group, a dimethylphenylsilyl group, a dimethyl-t-butylsilyl group, and a dimethyl (p-fluorophenyl) silyl group.
- a methylsilyl group, dimethyl, silyl group, trimethylsilyl group, ethylsilyl group, getylsilyl group, triethylsilyl group, dimethylphenylsilyl group, triphenylsilyl group and the like are preferable.
- a trimethinoresilyl group, a triethylsilyl group, a triphenylsilyl group, and a dimethylphenylsilyl group are preferred.
- Specific examples of the hydrocarbon-substituted siloxy group include a trimethylsiloxy group.
- Examples of the germanium-containing group and the tin-containing group include those in which the silicon of the silicon-containing group is replaced with germanium and tin.
- Ri ⁇ R 6 includes two or more of these groups, preferably mutually adjacent groups, consolidated to aliphatic ring, aromatic ring or forms form a hydrocarbon ring containing a hetero atom such as nitrogen atom And these rings may further have a substituent.
- Ri groups represented by Ri ⁇ R 6. Further, Ri each other when m is 2 or more, R 2 together, R 3 together.,
- R4, R5, and R6 may be the same or different from each other.
- n is a number that satisfies the valence of M, specifically 0 to 5, preferably 1 to 4, Preferably it is an integer of 1-3.
- X is a hydrogen atom, a halogen atom, a hydrocarbon group, an oxygen-containing group, a zeo-containing group, a nitrogen-containing group, a boron-containing group, an aluminum-containing group, a phosphorus-containing group, a halogen-containing group, a heterocyclic compound residue, silicon A group, a germanium-containing group, or a tin-containing group, and when n is 2 or more, a plurality of groups represented by X may be the same or different from each other, and a plurality of groups represented by X They may combine to form a ring. '
- halogen atom examples include fluorine, chlorine, bromine, and iodine.
- Examples of the hydrocarbon group, the Ri ⁇ R 6 Ru include the same ones as exemplified. Among them, those having 1 to 20 carbon atoms are preferred.
- heterocyclic compound residue oxygen-containing groups, examples of Iou-containing group and nitrogen-containing groups include the same ones as exemplified in the Ri ⁇ R 6.
- boron-containing group examples include BR 4 (R represents hydrogen, an alkyl group, an aryl group which may have a substituent, a halogen atom, or the like).
- the phosphorus-containing group examples include a trialkylphosphine group such as a trimethylphosphine group, a triptylphosphine group, and a tricyclohexylphosphine group; a triarylphosphine group such as a triphenylphosphine group and a tolylphosphine group; Examples include, but are not limited to, phosphite groups (phosphide groups) such as a phyto group, an ethyl phosphite group, and a phenyl phosphite group; a phosphonic acid group; and a phosphinic acid group.
- phosphite groups phosphide groups
- silicon-containing group examples include the same as those exemplified above in 1 to 16 .
- germanium-containing group examples include the same as those exemplified above in 1 to 16 .
- tin-containing group examples include the same as those exemplified above in 1 to 16 .
- halogen-containing group PF 6, BF 4 Fluorine-containing groups such as, C10 4, SbCl 6 as chlorine containing groups, and an iodine-containing group such as I0 4, is limited to Absent.
- the aluminum-containing group include, but are not limited to, A1R 4 (R represents a hydrogen, an alkyl group, an aryl group which may have a substituent, a halogen atom, or the like).
- R represents a hydrogen, an alkyl group, an aryl group which may have a substituent, a halogen atom, or the like.
- n is 2 or more, a plurality of groups represented by X may be the same or different from each other, and a plurality of groups represented by X may be bonded to each other to form a ring.
- (A1) a Group 4 transition metal complex preferable as the Group 4 transition metal compound represented by the general formula [I] is represented by the following general formula [ ⁇ ].
- M represents a transition metal atom belonging to Group 4 of the periodic table
- m represents an integer of 1 to 4
- 'R 1 ' is represented by the following general formula [IV] or [V];
- 2 to R 6 may be the same or different, and each represents a hydrogen atom, a halogen atom, a hydrocarbon group, a residue of a heterocyclic compound, an oxygen-containing group, a nitrogen-containing group, a boron-containing group, an i-containing group> A containing group, a silicon-containing group, a germanium-containing group, or a tin-containing group, and two or more of these may be connected to each other to form a ring.
- n is a number that satisfies the valency of M
- X is a hydrogen atom, a halogen atom, a hydrocarbon group, an oxygen-containing group, an iodine-containing group, a nitrogen-containing group, a boron-containing group, an aluminum-containing group, a phosphorus-containing group.
- n 2 or more
- the groups represented by X are the same or different.
- a plurality of groups represented by X may combine with each other to form a ring.
- R a represents a hydrogen atom, an aliphatic hydrocarbon group or an alicyclic hydrocarbon group
- Rb and Rc represent a hydrogen atom or a methyl group, and may be the same or different from each other)
- Ri ' is an aliphatic hydrocarbon group or an alicyclic hydrocarbon group represented by the general formula [IV] or [V ⁇ , and examples thereof include those having 1 to 30 carbon atoms. Specifically, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, tert-amyl, 1,2-dimethylpropyl, isoamyl, 1-methylbutyl , 2-methylbutyl, neopentyl, n-hexyl, 1,3-dimethylbutyl, 3,3-dimethylbutyl, n-heptyl, 1-methylhexyl, n-octyl, 1,5-dimethylhexyl, 2- Ethylhexyl, 1-methylheptyl, n-nonyl, n-decyl, n-pentade
- R 2 to R 6 are group 4 transition metal compounds represented by the general formula [I]. R 2 to R 6 described above can be used without limitation.
- R 2 to R 6 When m is 2 or more, two of the groups represented by R 2 to R 6 may be linked. Further, when m is 2 or more, R 2 , R 3 , R 4 , R 5, and R 6 may be the same or different.
- X is a hydrogen atom, a halogen atom, a hydrocarbon group, an oxygen-containing group, a zeo-containing group, a nitrogen-containing group, a boron-containing group, an aluminum-containing group, a phosphorus-containing group, a halogen-containing group, a heterocyclic compound residue, silicon
- n is 2 or more
- a plurality of groups represented by X may be the same or different, and a plurality of groups represented by X They may combine to form a ring.
- n is a number that satisfies the valence of M, and is specifically an integer of 0 to 5, preferably 1 to 4, and more preferably 1 to 3.
- X is a hydrogen atom, a halogen atom, a hydrocarbon group, an oxygen-containing group, a zeo-containing group, a nitrogen-containing group, a boron-containing group, an aluminum-containing group, a phosphorus-containing group, a halogen-containing group, a heterocyclic compound residue, silicon X, which is the same as the group 4 transition metal compound represented by the general formula [I], can be exemplified.
- n is 2 or more, a plurality of groups represented by X may be the same or different from each other, and a plurality of groups represented by X may be bonded to each other to form a ring.
- Group 4 transition metal compound represented by the general formula [I] or [ ⁇ ] two or more different compounds can be used.
- the method for producing such a transition metal compound (A1) can be, for example, the method described in Japanese Patent Application Laid-Open No. 11-315109 or EP0874005A1 by the present applicant without any particular limitation.
- Me represents a methyl group
- Et represents an ethyl group
- tBu represents a tert-butyl group
- Ph represents a phenyl group.
- a transition metal compound in which zirconium metal is replaced with a metal other than zirconium such as titanium or hafnium in the above compounds may be used.
- the Group 4 transition metal compound represented by the general formula [ ⁇ ] according to the present invention is a crosslinkable meta-acene compound as described below.
- R 7 to R 2 o are selected from hydrogen, a hydrocarbon group, and a silicon-containing group, and may be the same or different, and adjacent substituents to R 7 to R 20 are bonded to each other.
- M may be a Group 4 transition metal atom
- Y may be a Group 14 atom
- Q may be coordinated with a halogen, a hydrocarbon group, an anion ligand or a lone pair of electrons. The same or different combinations may be selected from the available neutral ligands, and j is an integer of 1 to 4.
- examples of the hydrocarbon group of R 7 to R 20 include an alkyl group having 1 to 20 carbon atoms, an alkyl group having 7 to 20 carbon atoms, and an aryl group having 6 to 20 carbon atoms.
- Examples of the silicon-containing group include a trimethylsilyl group, a triethylsilyl group, a diphenylmethylsilyl group, and a dimethylphenylsilyl group.
- a cyclohexyl group, a cyclopentyl group, an adamantyl group and the like in which Ri 9 and R 20 are bonded to each other to form a ring are also exemplified.
- At least one of the forces R 9 R 20 mentioned as a preferred form of Ri 9 and R 20 is an unsubstituted aryl group or a substituted aryl group.
- R 19 and R 20 may be the same or different. More specifically, when R 19 and R 20 are an unsubstituted aryl group or a substituted aryl group, a carbon atom such as a phenyl group, a naphthyl group, a biphenyl group, a terphenyl group, a phenanthryl group, an anthracenyl group, etc.
- aryl group having a number of 6 to 30, preferably 6 to 20, or tolyl iso-propylphenyl, tert-butylphenyl, ethynolephenyl, dimethylphenyl, di-tert-butylphenyl;
- alkyl-substituted aryl group such as an aryl group.
- these aryl groups include a halogen atom, an alkyl group or an alkoxy group having 1 to 30, preferably 1 to 20 carbon atoms, an aryl group having 6 to 30, preferably 6 to 20 carbon atoms, or an aryloxy group. Substituents such as:!
- chlorophenyl group dichlorophenyl group, fluorophenyl group, difluorophenyl group, trifluoromethylphenyl group, di (trifluoromethyl) phenyl group, etc. And a halogen-containing hydrocarbon group.
- the covalent bonding atom Y connecting the cyclopentagenil ligand and the fluorenyl ligand is a Group 14 atom, a carbon atom, a silicon atom, a germanium atom, and an alkylene group, a substituted alkylene group, a silylene group, And a substituted silylene group.
- -C (C 6 H 5 ) 2- -C (C 6 H 5 ) (p-CH 3 C 6 H 5 )-, -C (p-CH 3 C 6 H 5 ) (pC H 3 C 6 H 5 )-, -C (tert-BuC 6 H 5 ) (tert-BuC 6 H 5 )-, -SiC (C 6 H 5 ) 2-, -Si (C 6 H 5 ) (p-CH 3 C 6 H 5 )-, -Si (p-CH 3 C 6 3 ⁇ 4) (p- CH 3 C 6 H 5 )-, -Si (tert-Bu C 6 H 5 ) (tert-BuC 6 H 5) - the number of carbon atoms of the like is bridge portion consisting etc. 6-20 unsaturated hydrocarbon group.
- Q is selected from the same or different combinations of halogen, a hydrocarbon group, an anion ligand or a neutral ligand capable of coordinating with a lone pair of electrons.
- j is an integer of 1 to 4, and when j is 2 or more, Qs may be the same or different.
- halogen examples include fluorine, chlorine, bromine, and iodine
- hydrocarbon group examples include hydrocarbon group, and specific examples of the hydrocarbon group are the same as those described above.
- anion ligand examples include an alkoxy group such as methoxy, tert-butoxy and phenoxy, a carboxylate group such as acetate and benzoate, and a sulfonate group such as mesylate and tosylate.
- neutral ligands examples include organic phosphorus compounds such as trimethylphosphine, triethylphosphine, triphenylphosphine, and diphenylmethylphosphine, tetrahydrofuran, getyl ether, and dioxane.
- ethers such as 1,2-dimethoxetane. It is preferable that at least one of Q ′ is a halogen or an alkyl group.
- cross-linked meta-acene compound of the present invention is not limited to the above-mentioned exemplified compounds at all, but includes all compounds satisfying the requirements of the general formula [ ⁇ ].
- the crosslinkable meta-opencen compound represented by the above general formula [ ⁇ ] of the present invention can be produced by a known method, and the production method is not particularly limited. Known WO01 / 27174 by the present applicant is an example of the production method. Further, in the present invention, in addition to the Group 4 transition metal compound represented by the general formula [[], a Group 4 transition metal compound ( ⁇ 2 ′) containing a cyclopentagenyl skeleton as a ligand is used. It can also be used to produce the ethylene polymer of the present invention.
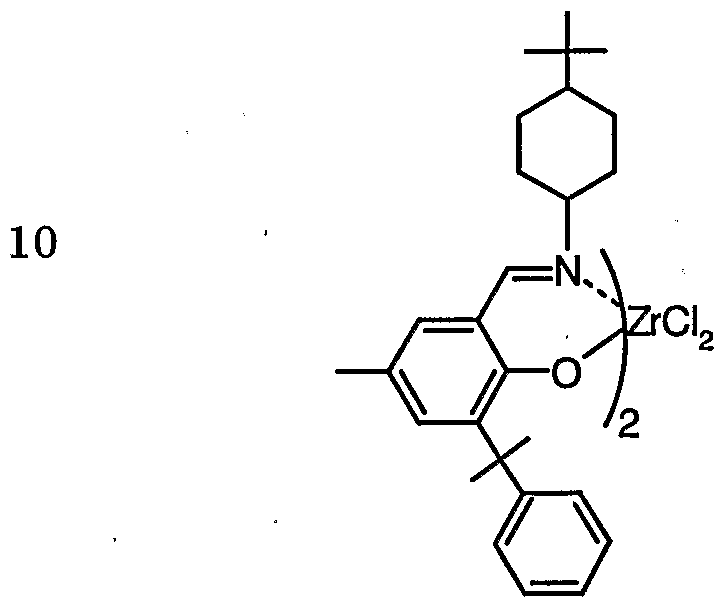
- examples of preferably used meta-mouth compounds include bis (methylcyclopentageninole) dinoreconidium dichloride and bis (dimethylmetholene).
- Norecyclopentageninole) dinorecone dichloride bis (dimethylcyclopentagenenyl) zirconium ethoxycyclolide bis (dimethylcyclopentagenenyl) zirconium bis (trifluoromethanesulfonato), bis (ethylcyclopentagenenyl) zirconium dichloride , Bis (protyl cyclopentagenenyl) zirconium dichloride, bis (methylpropylcyclopentadienyl ⁇ /) dinoreconicum dichloride, bis (butynolecyclopentadiene / di) dinoleconium dichloride, bis (methylbutylcyclopentene) Tageyl) zirconium dichloride
- meta-opencene compounds ( ⁇ 2 ′) two cyclopentagenenyl skeletons are contained as ligands, and the two cyclopentagenenyl skeletons are bonded to each other through a group 14 atom. It is more preferable to use a chiral meta metacene compound having a C2 symmetry ( ⁇ 2 "), which is preferably a Group 4 transition metal compound. A chiral meta meta sense compound having C2 symmetry is preferred.
- Examples of the compound ( ⁇ 2 ") include rac-ethylene-bis (indenyl) zirconium dichloride, rac-ethylene-bis (tetrahydroinginyl) zirconium dichloride, rac-dimethylsilylene-bis (2,3,5-trimethylcyclochloride).
- Pentajyl) zirconium dichloride rac-dimethylsilylene-bis [1- (4-phenylindur)] zirconium dichloride, rac-dimethylsilylene-bis [1- (2-methyl-4-phen) Ruindeyuru)] zirconium dichloride, rac- dimethylsilylene - bis ⁇ 1- [2-Methyl-4- (l-naphthyl) indul] ⁇ zircoium dichloride, rac-dimethylsilylylene-bis ⁇ 1- [2-methyl-4- (2-naphthyl) indur:
- ⁇ zirconium dichloride Rac-dimethylsilylene-bis ⁇ 1- [2-methyl-4- (1-anthryl) indur]] zirconium dichloride, rac-dimethylsilylene-bis ⁇ 1- [2-methyl-4- (9-anthryl) ) Indenyl:] ⁇ zirconium dichloride, rac
- Group 4 transition metal compounds ( ⁇ 2) and ( ⁇ 2 ′) two or more different compounds can be used.
- (A1) a group 4 transition metal compound represented by the general formula [I] and ( ⁇ 2) a group 4 transition metal compound represented by the general formula [ ⁇ ] are used.
- the compounds disclosed in Japanese Patent Application Laid-Open No. 11-315109 and EP0874005A1 by the present applicant can be used without limitation.
- (b-1) As the organometallic compound, an organoaluminum compound is preferable, and one type is used alone or two or more types are used in combination.
- (b-2) As the organoaluminum oxy compound, aluminoxane prepared from trialkylaluminum or tricycloalkylaluminum is preferable. trimethylaluminum or triisobutyl Organoaluminoxy compounds prepared from aluminum are particularly preferred. Such organic aluminum oxy compounds are used singly or in combination of two or more.
- (B-3) Compounds which form an ion pair by reacting with the transition metal compounds (Al) and (A2) are described in JP-A-1-501950, JP-A-1-502036.
- an extremely high polymerization activity for the olefin compound can be obtained by using together with an organoaluminoxy compound (b-2) such as methylaluminoxane as a co-catalyst component.
- an organoaluminoxy compound (b-2) such as methylaluminoxane
- an ionizing ionic compound (b-3) such as triphenylcarboditetrakis (pentafluorophenyl) borate
- a olefin polymer having good activity and a very high molecular weight can be obtained.
- the catalyst for polymerization of olefins comprises the group 4 transition metal compounds (A1) and (A2), (b-1) an organometallic compound, and (b-2) an organoaluminoxy compound. And (b-3) at least one compound (B) selected from a transition metal compound (A1) and a compound capable of forming an ion pair by reacting with the transition metal compound (A2), (C) is used by being carried. .
- the solid carrier (C) used in the present invention is an inorganic or organic compound and is a granular or fine solid.
- the inorganic compound is preferably a porous oxide, an inorganic chloride, a clay, a clay mineral, or an ion-exchange layered compound.
- porous oxide specifically Si_rei_2, Al2_rei 3, MgO, ZrO, Ti_rei_2, B 2 0 3, CaO, ZnO, BaO, Th0 2 , etc., or a composite or a mixture containing them ⁇ ⁇ .
- a composite or a mixture containing them ⁇ ⁇ . such as natural or synthetic Zeoraito, Si0 2 -MgO, S1O2-AI2O3 , Si0 2 -Ti0 2, Si0 2 -V 2 0 5, Si0 2 -Cr 2 0 3, Si0 2 -Ti0 2 -MgO Etc.
- Si0 2 -MgO, S1O2-AI2O3 Si0 2 -Ti0 2, Si0 2 -V 2 0 5, Si0 2 -Cr 2 0 3, Si0 2 -Ti0 2 -MgO Etc.
- the above inorganic oxides may contain small amounts of Na 2 C0 3, K2CO3, CaCOs , MgC0 3, Na 2 S0 4, A1 2 ( S_rei_4) 3, BaS_rei_4, KN0 3, Mg (N0 3 ) 2, A1 (N0 3) 3, Na 2 0, K 2 0, carbonates such L12O, sulfates, nitrates, pointing also contain oxide component supported stomach.
- the carrier preferably used in the present invention has a particle size of 0.2 to 300 ⁇ , preferably.
- the specific surface area is in the range of 50 to 200 m 2 / g, preferably 100 to 100 pm 2 / g, and the pore volume is in the range of 0.3 to 30 cm 3 / g.
- Such a carrier is used by calcining at 100 to: 1000 ° C, preferably 150 to 700 ° C, if necessary.
- the inorganic chloride MgCl 2 , MgBr 2 , MnCl 2 , MnBr 2 and the like are used.
- the inorganic chloride may be used as it is, or may be used after being pulverized by a ball mill or a vibration mill.
- a precipitate obtained by dispersing the inorganic chloride in a fine particle form with a precipitation agent can be used.
- the clay used in the present invention is usually composed mainly of a clay mineral.
- the ion-exchangeable layered compound used in the present invention is a compound having a crystal structure in which planes formed by ionic bonds and the like are stacked in parallel with weak bonding force, and the ions contained therein are exchangeable. is there.
- Most clay minerals are ion-exchangeable layered compounds. Manako, these clays, clay minerals, and ion-exchangeable layered compounds are not limited to natural ones, and artificially synthesized ones can also be used.
- Examples of the clay, clay mineral or ion-exchangeable layered compound include clay, clay mineral, and ion crystalline compounds having a layered crystal structure such as hexagonal dense packing type, antimony type, CdCl2 type, and Cdl type 2. Examples can be given.
- Such clays and clay minerals include kaolin, bentonite, kibushi clay, gairome clay, alofen, hissingelite, pyrophyllite, pummo group, montmorillonite group, vermiculite, lyotadiite group, palygorskite, kaolinite, nacrite, Deitsukaito, halloysite, and examples of the ion-exchange layered compounds, ⁇ -Zr (HAs04) 2 ' H 2 0, a-Zr (HP0 4) 2> a-Zr (KP0 4) 2 -3H 2 0, a-Ti (HP0 4 ) 2, a-Ti (HAs04) 2 - H 2 0; a-Sn (HP0 4) 2 'H 2 0, y-Zr (HP0 4) 2, ⁇ - ⁇ ( ⁇ 0 4) 2, y-Ti (NH 4 P0 4) 2 'polyvalent metal crystalline acid salts of
- 'Such clays, clay minerals or ion-exchangeable layered compounds are preferably those with a pore volume of 0.1 cc / g or more, with a radius of 20 A or more measured by the mercury intrusion method, and 0.3 to 5 cc / g. Particularly preferred.
- the pore volume is measured by a mercury intrusion method using a mercury porosimeter in a range of pore radius of 20 to 3 ⁇ 104 ⁇ .
- the clay and clay mineral used in the present invention are preferably subjected to a chemical treatment.
- a chemical treatment any of a surface treatment for removing impurities adhering to the surface and a treatment for affecting the crystal structure of the clay can be used.
- Specific examples of the chemical treatment include an acid treatment, an alkali treatment, a salt treatment, and an organic substance treatment.
- Acid treatment removes surface impurities and increases the surface area by eluting cations such as Al, Fe, and Mg in the crystal structure.
- Alkali treatment destroys the crystal structure of clay and causes a change in the structure of clay.
- an ion complex, a molecular complex, an organic derivative, and the like are formed, and the surface area and the interlayer distance can be changed.
- the ion-exchangeable layered compound used in the present invention utilizes ion-exchange properties.
- exchangeable ions By exchanging exchangeable ions between layers with another large bulky ion, even a layered compound in which the layers are expanded is used. Good.
- Such bulky ions serve as pillars supporting the layered structure, and are usually called pillars.
- Introducing another substance between the layers of the layered compound in this way is called intercalation.
- Guest compounds to be intercalated include cationic inorganic compounds such as TiCl 4 and ZrCU, and metal alcohols such as Ti (OR) 4 , Zr (OR) 4 , PO (OR) 3 and B (OR) 3.
- Sid (R is such hydrocarbons group), [Ali 3 0 4 ( OH) 24] 7+, [Zr 4 (OH) 14 p +, [Fe 3 0 (OCOCH 3) 6 ] + or the like of a metal hydroxide Ions and the like. These compounds are used alone or in combination of two or more.
- metal alkoxides (R is a hydrocarbon group, etc.) such as Si (OR) 4 , Al (OR) 3 , Ge (OR) 4 are obtained by hydrolysis.
- polymer product, colloids such Si_ ⁇ 2 Inorganic compounds and the like can coexist.
- the leaf include oxides formed by intercalating the above metal hydroxide ions between layers and then heating and dehydrating.
- the clay, clay mineral, and ion-exchangeable layered compound used in the present invention may be used as they are, or may be used after performing a treatment such as ball milling or sieving. Further, it may be used after newly adsorbing and adsorbing water, or after dehydrating by heating. Further, they may be used alone or in combination of two or more.
- clays or clay minerals preferred are clays or clay minerals, and particularly preferred are montmorillonite, permiculite, pectrite, teniolite and synthetic mica.
- the organic compound examples include a granular or fine particulate solid having a particle size in the range of 10 to 300 ⁇ .
- a (co) polymer produced mainly from olefins having 2 to 14 carbon atoms, such as ethylene, propylene, 1-butene and 4-methyl-1-pentene, or vinylcyclohexane, styrene (Co) polymers produced as a main component and modified products thereof.
- the catalyst for polymerization of olefins according to the present invention comprises the above transition metal compounds (A1) and (A2), (b-1) an organometallic compound, (b-2) an organoaluminoxy compound, and (; b-3 )
- a specific organic compound component (D) may be contained, if necessary, together with at least one compound (B) selected from ionized ionic compounds and the solid carrier (C).
- the compounds disclosed in Japanese Patent Application Laid-Open No. 11-315109 or EP0874005 A1 by the present applicant can be used without limitation.
- a method in which a catalyst component in which the component (A1), the component (A2), and the component (B) are supported on a carrier (C) is added to a polymerizer.
- a catalyst component in which the component (A1), the component (A2), and the component (B) are supported on a carrier (C) is added to a polymerizer.
- at least two or more of the respective catalyst components may be brought into contact with each other in advance.
- the unsupported component (B) may be added in an arbitrary order, if necessary.
- the components (B) may be the same or different.
- the solid catalyst component in which the component (A1), the component (A2) and the component (B) are supported on the carrier (C) of the above [1] and [2] can be used as a preparative polymerized olefin.
- a catalyst component may be further supported on the polymerized solid catalyst component.
- the loading of the component (A1) and the component (B) on the carrier (C) or the loading of the component (A2) and the component (B) on the carrier (C) in the above [1] is performed according to a known method. Easy to do: can do. Further, the loading of the component (A1), the component (A2) and the component (B) on the carrier (C) in the above [2] is preferably performed as follows.
- 'One is a method in which the component (A1) and the component (A2) are preliminarily brought into contact, and then the component (B) is brought into contact with the carrier (C) on which the component (B) is supported. ) [Or component (A2)] is brought into contact with the carrier (C) supporting component (B), and then component (A2) [or component (Al)] is added to component (A1) [or component (A2)] and ingredients
- the former is brought into contact with the carrier (C).
- the components (A1) and (A2) are dissolved in an inert hydrocarbon-based solvent at an arbitrary ratio, and then the inert hydrocarbon is added to the carrier (C) supporting the component (B).
- This is a method in which the component (A1), the component (A2,) and the component (B) are supported on a carrier (C) by contact in a system solvent.
- the inert hydrocarbon solvent used for the support include aliphatic hydrocarbons such as propane, butane, pentane, hexane, heptane, octane, decane, dodecane, and kerosene, cyclopentane, cyclohexane, and methylcyclohexane.
- aliphatic hydrocarbons such as propane, butane, pentane, hexane, heptane, octane, decane, dodecane, and kerosene, cyclopentane, cyclohexane, and methylcyclohexane.
- examples thereof include alicyclic hydrocarbons such as pentane, aromatic hydrocarbons such as benzene, toluene, and xylene; halogenated hydrocarbons such as ethylene chloride, chlorobenzene, and dichloromethane; and mixtures thereof.
- the pre-contact time between component (A1) and component (A2) is usually 0 to 5 hours, preferably 0 to 1 hour, particularly preferably 0 to 20 minutes, and the subsequent contact time with the carrier (C) supporting the component (B) is usually 0 to 24 hours, preferably 0 to 24 hours. 5 hours, particularly preferably 0 to 2 hours.
- These loading operations are usually performed at -50 to 200 ° C, preferably at -50 to 50 ° C, particularly preferably at 0 to 40 ° C.
- the components (A1) and (A2) can be arbitrarily determined from the molecular weight and molecular weight distribution of the polyolefin to be produced, and the molar ratio of the components (A1) and (A2) [(Al) / (A2)] It can be determined from the olefin polymerization activity of each of (A1) and component (A2). '[(A1) / (A2)] [, usually 0.03 to 30, preferably f, 0.06 to: 15. '
- the total transition metal atoms (M) in the components (A1) and (A2) supported on the carrier (C) can be determined by inductively coupled plasma emission spectrometry (ICP analysis).
- Component (b-1) has a molar ratio [(bl) / M] of component (b-1) to all transition metal atoms (M) in component (A1) and component (A2). 0.01-: It is used in such an amount that it becomes 100000, preferably 0.05-50000.
- the component (b-2) is formed by the molar ratio of the aluminum atom in the component (b-2) to all transition metal atoms (M) in the component (A1) and the component (A2) [(b-2) / M] is usually used in an amount of 10 to 500,000, preferably 20 to 100,000.
- Component (b-3) has a molar ratio [(b-3) / M] of component (b-3) to all transition metal atoms (M) in components (A1) and (A2). 1 to: It is used in an amount such that it becomes 10, preferably 1 to 5.
- Component (D) is such that when component (B) is component (b-1), the molar ratio [(D) ba b-1)] is usually 0.01 to 10, preferably 0.1 to 5.
- Component (B) In the case of component (b-2), the amount is such that the molar ratio [(D) / (b-2)] is usually 0.001-2, preferably 0.005-1.
- the component (B) is the component (b-3), it is used in such an amount that the molar ratio [(D) / (b-3)] is usually 0.01 to 10, preferably 0.1 to 5.
- an olefin polymer is obtained by polymerizing or copolymerizing the olefin in the presence of the above-mentioned polymerization catalyst.
- the polymerization can be carried out by any of a liquid phase polymerization method such as solution polymerization and suspension polymerization or a gas phase polymerization method.
- Aliphatic hydrocarbons such as propane, butane, pentane, hexane, heptane, octane, decane, dodecane and kerosene; cyclopentane, cyclohexane, methylcyclopentane
- component (A1) ⁇ Pi (A2) is 1 liter of the reaction volume, usually 10-1 2 ⁇ : 10-1 mol, preferably the 10- 8: 10-2 employed in amounts such that the mole can also include an organic compound component of the above specific optionally (D).
- the polymerization temperature of the olefin using such a olefin polymerization catalyst is usually in the range of -50 to + 200 ° C, preferably 0 to + 170 ° C, and particularly preferably +60 to + 170 ° C. is there.
- the polymerization pressure is usually normal pressure ⁇ lOOkg / cm 2, preferably under conditions of normal pressure ⁇ 50 kg / cm @ 2, the polymerization reaction is a batch, semi-continuous, also be carried out in the methods of have deviation of continuous Can be. It is also possible to carry out the polymerization in two or more stages under different reaction conditions.
- the molecular weight of the obtained olefin polymer can be adjusted by changing the power of causing hydrogen to exist in the polymerization system or the polymerization temperature. Furthermore, it can be adjusted by the difference of the component (B) used.
- the oresin which can be polymerized by such an olefin polymerization catalyst is as described above, but if necessary, a cyclic olefin having 3 to 30, preferably 5 to 20 carbon atoms, such as cyclopentene, Cycloheptene, norbornene, 5-methyl-2-norbornene, tetracyclododecene, etc .; polar monomers such as acrylic acid, methacrylic acid, fumaric acid, maleic anhydride; methyl acrylate, ethyl acrylate, methyl methacrylate J3-unsaturated carboxylic acid esters such as methacrylic acid, ethyl methacrylate, methacrylic acid, etc .; vinyl esters such as butyl acetate and vinyl propionate; unsaturated glycidyls such as glycidyl acrylate and glycidyl methacrylate; Halogenated olefins such as bur may
- olefin burcyclohexane, gen or polyene or the like is used. You can also. Further, as the olefin, an aromatic vinyl compound such as a sullen or a functional group-containing styrene derivative such as a dibutyl benzene can be used in combination.
- the ethylene polymer particles obtained in this manner may be velvet-shaped by the following method.
- Ethylene-based polymer and other components to be added as required are in a suitable good solvent (for example, hydrocarbon solvents such as hexane, heptane, decane, cyclohexane, benzene, toluene, and xylene). And then removing the solvent, followed by mechanical blending using an extruder, a kneader or the like, and cutting to a predetermined size.
- a suitable good solvent for example, hydrocarbon solvents such as hexane, heptane, decane, cyclohexane, benzene, toluene, and xylene.
- the ethylene polymer according to the present invention has a range that does not impair the object of the present invention: weather-resistant stabilizer, heat-resistant stabilizer, antistatic agent, anti-slip agent, anti-blocking agent, anti-fog agent, lubricant , Dyes, nucleating agents, plasticizers, antioxidants, hydrochloric acid absorbents, additives such as antioxidants, carbon black, titanium oxide, titanium yellow, phthalocyanine, isoindolinone, quinacridone compounds, condensed azo compounds. Ultramarine, Co: Baltic blue and other pigments may be incorporated as necessary.
- the ethylene-based polymer according to the present invention can be formed into a blow molded article, an inflation molded article, a cast molded article, an extruded laminate molded article, an extruded molded article such as a pipe or a deformed article, a foamed molded article, an injection molded article, and the like. . Further, it can be used for fibers, monofilaments, non-woven fabrics and the like.
- These molded articles include molded articles (laminates and the like) including a part composed of an ethylene-based polymer and a part composed of another resin.
- the ethylene polymer may be one crosslinked in the molding process. It is preferable that the ethylene-based polymer according to the present invention be used in a blow-molded product and an extruded product such as a pipe or a deformed product, among the above-mentioned molded products, because excellent properties are provided.
- the ethylene-based polymer of the present invention can be formed into bottle containers, industrial chemical cans, gasoline tanks, and the like by professional molding.
- These molded articles include molded articles (laminates and the like) including a portion composed of an ethylene-based polymer and a portion composed of another resin. Is included. Also, it can be molded with a single layer of an ethylene-based polymer. 'It is possible to use the bleaching agent typified by the chlorine bleaching agent in the content liquid, and also suitably the surfactant.
- the ethylene polymer of the present invention can be molded into a pipe or a pipe joint formed by injection molding.
- These molded articles include molded articles (laminates and the like) including a portion made of an ethylene (co) polymer and a portion made of another resin. Moreover, it can also shape
- These compacts can be colored using a coloring agent such as titanium oxide or phthalocyanine. Further, it can be used as a molded article having a function by adding additives such as an antioxidant and an antistatic agent.
- a coloring agent such as titanium oxide or phthalocyanine.
- the hexene content in the ethylene / hexene copolymer was determined using FT-IR (SHIMAZU FTIR-8200D).
- the transition metal atom concentration and A1 concentration in the solid component and the solid catalyst component were determined by inductively coupled plasma emission spectrometry (ICP analysis).
- a program was created using Microsoft's Ethacel (registered trademark) 97 Visual 'Basic.
- the two curves to be separated were assumed to be log-normal, and the molecular weight distribution curve was separated into two curves with different molecular weights by convergence calculation.
- the recombined curve of the two separated curves is compared with the molecular weight curve actually measured by GPC, and the calculation is performed while changing the initial value so that the two almost match.
- the calculation is performed by dividing Log (molecular weight) into 0.02 intervals.
- the intensity was normalized so that the area of the actually measured molecular weight curve and the area of the separated curve recombined were 1 and the actual measured intensity (height) and recombined curve for each 'molecular weight
- the value obtained by dividing the absolute value of the difference from the intensity (height) of the sample by the actually measured intensity (height) is 0.4 or less, preferably 0.2 or less, more preferably 0.1 or less in the range of a molecular weight of 10,000 to 1,000,000: Yes, the separation calculation of the curve is repeated until the maximum position of the two separated peaks is 0.2 or less, preferably 0.1 or less.
- the difference between MwZMn of the peak separated on the low molecular weight side and MwZMn of the peak separated on the high molecular weight side is set to 1.5 or less.
- the polymer blend ratio (BR) of the low molecular weight component and the high molecular weight component was calculated by the following equation.
- Melt flow rate is measured as follows. That is, an orifice that meets the dimensions specified in JIS K7210 is attached to an automatic MFR measurement meter manufactured by Tester Sangyo Co., Ltd. manufactured according to JIS K7210, and the barrel (the part where the sample is placed) is heated to 190 ° C. Hold. Put 4 g of sample in the barrel, attach a piston, remove bubbles, and preheat for 6 minutes. After preheating, apply a load of 2.16 Kg or 21.6 kg to extrude the sample, calculate the weight of the sample extruded per 10 minutes, and use it as the melt flow rate.
- MT measurement is measured as follows. That is, an orifice that meets the dimensions specified in JIS K7210 is attached to an automatic MFR measurement meter manufactured by Tester Sangyo Co., Ltd. manufactured according to JIS K7210, and the barrel (the part where the sample is placed) is heated to 190 ° C. Hold. Put 4 g of sample in the barrel, attach a piston
- Melt tension (MT) is determined by measuring stress when a molten ethylene polymer is stretched at a constant speed. That is, the measurement was performed using an MT measuring machine manufactured by Toyo Seiki Seisakusho under the conditions of a resin temperature of 190 ° C., an extrusion speed of 15 mm / min, a winding speed of 7.85 m / min, a nozzle diameter of 2.09 mmq), and a nozzle length of 8 mm.
- Ea is measured as follows. That is, use a TA Instruments Stress Rheometer SR-5000 at a temperature of 150 ° C, 190 ° C, and 230 under nitrogen atmosphere at a measurement frequency of O.lrad / sec to 100rad / sec. Melt viscoelasticity is measured in the range. A parallel plate with a diameter of 25 mm is used as a jig for applying shear to the resin.
- the Group 4 transition metal compounds (A1) and (A2) used in this example and comparative example were synthesized by the method described above.
- silica manufactured by Asahi Glass Co., Ltd.
- the temperature was raised to 114 ° C over 30 minutes, and the reaction was carried out at that temperature for 4 hours. Thereafter, the mixture was cooled to 60 ° C. and warmed, and the supernatant was removed by decantation.
- the solid thus obtained was washed with toluene three times, and toluene was added thereto.
- Toluene 15 was prepared, and a part of the obtained solid component (E) was sampled and the concentration was examined.
- the result was a slurry concentration of 0.1189 g / ml and an A1 concentration of 0.8377 mmol / ml.
- the polymer was dried under reduced pressure at 80 ° C. for 10 hours.
- the amount of the obtained ethylene / 1-hexene copolymer was 28.5 g, and the polymerization activity per 1 mmol of zirconium was 190 kg / mmol.
- the hexene content was 0.48 mol%, [rj] was 3.54 dl / g, Mw, Mw / Mn, and BR were 235,000, 15.25, and 53/47, respectively.
- the mixture was washed three times with heptane, and 100 ml of heptane was added to prepare a heptane slurry of solid catalyst component (G).
- a part of the heptane slurry of the obtained solid catalyst component (G) was sampled and the concentration was examined, the Zr concentration was 0.000105 mmol / ml, and the A1 concentration was 0.0328 mmol / ml.
- the polymer was dried under reduced pressure at 80 ° C. for 10 hours.
- the amount of the obtained ethylene / 1-hexene copolymer was 18.7 g, and the polymerization activity per 1 mmol of zirconium was 234 kg / mmol.
- Hexene content is 0.32mol%
- [77] was 3.39 dl / g, Mw, Mw / Mn, and BR were 240,000, 15.91, and 54/46, respectively.
- A1 concentration 0.0280 mmol / ml.
- the temperature was raised to C, and polymerization was carried out for 3 hours while supplying an ethylene / hydrogen mixed gas (hydrogen concentration: 0.06 mol%) so that the total pressure became 8 kg / cm2-G.
- the polymerization was stopped by adding a small amount of methanol.
- the obtained polymer was washed with hexane and dried under reduced pressure at 80 ° C for 10 hours.
- the amount of the obtained ethylene / 1-hexene copolymer was 22.6 g, and the polymerization activity per mmol of zirconium was 452 kg / mmol.
- the hexene content was 0.67 mol%, [rj] was 3.89 dl / g, Mw, Mw / Mn, and BR were 207,000, 18.64, and 56/44, respectively.
- the supernatant was removed by decantation, washed three times with heptane, and 100 ml of heptane was added to prepare a heptane slurry of the solid catalyst component (J).
- the Zr concentration was 0.000121 mmol / ml
- the A1 concentration was 0.0307 mmol / ml.
- a 1-liter autoclave made of SUS sufficiently purged with nitrogen was charged with 500 ml of heptane at room temperature, and the liquid and gas phases were saturated with ethylene. Thereafter, 3 ml of 1-hexene: 1.0 ml of a heptane solution (0.5 mmol / ml in terms of aluminum atom) of triisobutylaluminum (manufactured by Tosoh Finechem) and 0.207 ml of solid catalyst component (J) (in terms of zirconium) (0.000025 mmol), and the mixture was heated to 80 ° C. and polymerized for 3 hours while supplying ethylene so that the total pressure became 8 kg / cm 2 -G. Termination of the polymerization was carried out by adding a small amount of methanol.
- the polymer was dried under reduced pressure at 80 ° C. for 10 hours.
- the amount of the obtained ethylene 1-hexene copolymer was 30.2 g, and the polymerization activity per 1 mmol of zirconium was 1210 kg / mmol.
- Hexene content 0.85mol%, [77] is 3.10dl / g, Mw, Mw / Mn, BR respectively 337000, 12.3 9, Q was 41/59
- a 1-liter autoclave made of SUS sufficiently purged with nitrogen was charged with 500 ml of heptane at room temperature, and the liquid and gas phases were saturated with ethylene. Thereafter, 3 ml of 1-hexene, 1.0 ml of a heptane solution of triisobutylaluminum (manufactured by Tosoichi Finechem) (0.5 mmol / ml in terms of aluminum atom), 0.214 ml of solid catalyst component (K) (0.000025 in terms of zirconium) mmol), and the mixture was heated to 80 ° C., and polymerization was performed for 3 hours while supplying an ethylene / hydrogen mixed gas (hydrogen concentration: 0.06 mo 1%) so that the total pressure became 8 kg / cm 2 -G.
- the polymerization was stopped by adding a small amount of methanol. -After the obtained polymer was washed with hexane, it was dried under reduced pressure at 80 ° C for 10 hours. The amount of the obtained ethylene / hexene copolymer was 14.0 g, and the polymerization activity per 1 mmol of zirconium was 560 kg / mmol. The hexene content is 0.60 mol%,
- [77] was 4.001dl / g, Mw, Mw / Mn, and BR were 189,000, 12.68, and 43/57, respectively.
- Embodiment 6 is a diagrammatic representation of Embodiment 6
- a 1-liter autoclave made of SUS sufficiently purged with nitrogen was charged with 500 ml of heptane at a moderate temperature, and the liquid phase and the gas phase were saturated with ethylene. Thereafter, 3 ml of 1-hexene, 1.0 ml of a heptane solution of triisobutylaluminum (manufactured by Tosoichi Finechem Co., Ltd., 0.5 mmol / ml in terms of aluminum atom), and 0.208 ml of solid catalyst component (L) (in terms of zirconium) (0.000025 mmol), and the mixture was heated to 80 ° C. and polymerized for 3 hours while supplying an ethylene / hydrogen mixed gas (hydrogen concentration: 0.06 mol%) so that the total pressure became 8 kg / cm 2 -G. The polymerization was stopped by adding a small amount of methanol.
- the obtained polymer was washed with hexane, it was dried under reduced pressure at 80 ° C. for 10 hours.
- the amount of the obtained ethylene / hexene copolymer was 14.6 g, and the polymerization activity per 1 mmol of zirconium was 584 kg / mmol.
- the hexene content was 0.55 mol%, C77] was 4.34 dl / g, Mw, Mw / Mn, and BR were 234000, 24.67, and 41/59, respectively.
- the supernatant was removed by decantation, washed three times with heptane, and 100 ml of heptane was added to prepare a heptane slurry of the solid catalyst component (N).
- the Zr concentration was 0.000270 mmol / ml
- the A1 concentration was 0.0689 mmol / ml.
- the temperature was raised, and polymerization was carried out for 107 minutes while supplying an ethylene / hydrogen mixed gas (hydrogen concentration: 2.53 mol%) so that the total pressure became 8 kg / cm2-G. After the polymerization, the pressure was released, the atmosphere was replaced with nitrogen, and the used ethylene / hydrogen mixed gas was removed.
- the solid component thus obtained was washed three times with toluene, and toluene was added to prepare a toluene slurry of the solid component (O).
- a part of the obtained solid component (o) was sampled, and the concentration was examined.
- the slurry concentration was 0.08123 g./ml
- the A1 concentration was 0.7339 mmol / ml.
- the supernatant was removed by decantation, washed three times with heptane, and 100 ml of heptane was added to prepare a heptane slurry of the solid catalyst component (P).
- the Zr concentration was 0.000126 mmol / ml and the A1 concentration was 0.0347 mmol / ml.
- a 1-liter autoclave made of SUS sufficiently purged with nitrogen was charged with 500 ml of heptane at room temperature, and the liquid phase and the gas phase were saturated with an ethylene / hydrogen mixed gas (hydrogen concentration: 0.101 mol%). Then, 3 ml of 1-hexene, 0.5 ml of a solution of triisobutylaluminum (manufactured by Tosoh Finechem Co., Ltd.) in heptane (1.0 mmol / ml in terms of aluminum atom), and 0.59 ml of solid catalyst component (P) (0.000075 mmol in terms of zirconium) ) Additionally, the temperature was raised to 65 ° C, and polymerization was performed for 3 hours while supplying an ethylene / hydrogen mixed gas (hydrogen concentration: 0.101 mol%) so that the total pressure became 8 kg / cm 2 -G. Was. Termination of the polymerization was carried out by adding a small amount of methanol.
- Table 1 shows the yield of the obtained ethylene / hexene copolymer, the polymerization activity per lmmol of zirconium, the hexene content, [7]], Mw, Mw / Mn, and the polymer blend ratio (BR).
- Polymerization was carried out in the same manner as in Example 8 except that the hydrogen concentration in the ethylene / hydrogen mixed gas was changed to 0.153 mol%.
- the obtained polymer was washed with hexane, purified, and dried under reduced pressure at 80 ° C for 10 hours.
- Table 1 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per mmol of zirconium, hexene content, [j], Mw, Mw / Mn and BR.
- Example 8 was the same as Example 8 except that the charged amount of the solid catalyst component (P) was changed to 0.79 ml (zirconium conversion O.OOOOlOmmol), the hydrogen concentration in the ethylene / hydrogen mixed gas was changed to 0.064 mol%, and the polymerization temperature was changed to 60. Was polymerized in the same manner. The resulting polymer was washed with hexane and dried under reduced pressure at 80 ° C for 10 hours.
- P solid catalyst component
- Table 1 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [rj], Mw, Mw / Mn and BR.
- Example 8 Same as in Example 8, except that the charged amount of the solid catalyst component (P) was changed to 1.19 ml (0.00015 mmol in terms of zirconium), 1-hexene was changed to Oml, and the hydrogen concentration in the ethylene / hydrogen mixed gas was changed to 0.062 mol%. To carry out polymerization. The obtained polymer was washed with hexane and then dried under reduced pressure at 80 ° C for 10 hours.
- Table 1 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [77], Mw, Mw / Mn and BR.
- Table 2 shows the yield of the obtained ethylene hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [], Mw, Mw / Mn and BR.
- Polymerization was carried out in the same manner as in Polymerization Example 13 except that the hydrogen concentration in the ethylene Z hydrogen mixed gas was changed to 0.10 and 1 mol%. The obtained polymer was washed with hexane and then dried under reduced pressure at 80 ° C for 10 hours.
- Table 2 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [i]], Mw, Mw / Mn and BR. [Example 15]
- Polymerization was carried out in the same manner as in Polymerization Example 13, except that the hydrogen concentration in the ethylene / hydrogen mixed gas was changed to 0.153 mol%. After the obtained polymer was washed with hexane, it was dried under reduced pressure at 80 ° C. for 10 hours.
- Table 2 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [77], Mw, Mw / Mn and BR. [Example 16]
- Example 17 Polymerization was carried out in the same manner as in Example 13, except that the charged amount of the solid catalyst component (Q) was changed to 1.26 ml (0.00015 mmol in terms of zirconium) and 1-hexene was changed to 0 ml. After washing the obtained polymer with hexane, the polymer was dried under reduced pressure at 80 ° C. for 10 hours.
- Table 2 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [77], Mw, Mw / Mn and BR.
- Table 2 shows the yield of the obtained ethylene / hexene copolymer, polymerization activity per 1 mmol of zirconium, hexene content, [77], Mw, Mw / Mn and BR :.
- Table 3 shows the physical properties of the ethylene polymer synthesized in the above Examples.
- Tables 4 and 5 show physical property values of conventionally known ethylene polymers for comparison. 5 Regarding the known resins described in these comparative examples or reference examples, it is not possible to deviate from the provisions of the relationship between MFR 2 and MT and / or that Ea is 30 KJ / mol or more and out of the range of the request. it is obvious. .
- Reference example 3 Product name Mirason (Mitsui Chemicals) Brand name MR102J
- the ethylene polymer of the present invention is excellent in moldability, and provides a molded article excellent in mechanical strength and outer strength. When used for the blow molded article of the ethylene polymer according to the present invention and the extruded article such as a pipe or a deformed article, excellent properties are provided.
- the olefin polymerization catalyst and the polymerization method according to the present invention can produce an ethylene-based polymer having the above-mentioned excellent physical properties with high polymerization activity even in single-stage polymerization.
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP04733978.3A EP1632505B1 (en) | 2003-05-20 | 2004-05-19 | Ethylene polymer |
| US10/553,726 US7452946B2 (en) | 2003-05-20 | 2004-05-19 | Ethylene polymer |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003141246 | 2003-05-20 | ||
| JP2003-141246 | 2003-05-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004104055A1 true WO2004104055A1 (ja) | 2004-12-02 |
Family
ID=33475025
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2004/007124 WO2004104055A1 (ja) | 2003-05-20 | 2004-05-19 | エチレン系重合体 |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US7452946B2 (ja) |
| EP (1) | EP1632505B1 (ja) |
| CN (1) | CN100354322C (ja) |
| WO (1) | WO2004104055A1 (ja) |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006080578A1 (ja) * | 2005-01-31 | 2006-08-03 | Mitsui Chemicals, Inc. | エチレン系樹脂及びこれから得られる成形体 |
| WO2008016059A1 (fr) * | 2006-07-31 | 2008-02-07 | Mitsui Chemicals, Inc. | Film ou stratifié comprenant une résine d'éthylène ou une composition de résine d'éthylène |
| JP2008031385A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 熱可塑性樹脂用改質材および樹脂組成物 |
| JP2008031377A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 液体包装用フィルム |
| JP2008031380A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 易引裂き性フィルムおよびその製造方法ならびにその用途 |
| JP2008031374A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 積層体用樹脂組成物および積層体 |
| JP2008031379A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 低臭気性フィルムおよびその製造方法ならびにその用途 |
| JP2008031386A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 表面保護フィルム用樹脂組成物および表面保護フィルム |
| JP2008031383A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | ラミネート紙用樹脂組成物およびラミネート紙 |
| JP2008031381A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 肉厚フィルムおよびその製造方法ならびにその用途 |
| US7838611B2 (en) | 2005-12-19 | 2010-11-23 | Tosoh Corporation | Ethylene polymer, catalyst for ethylene polymer production, and method for producing ethylene polymer |
| CN101341176B (zh) * | 2005-12-19 | 2011-08-17 | 东曹株式会社 | 乙烯类聚合物、用于制备乙烯类聚合物的催化剂及制备乙烯类聚合物的方法 |
| US8129489B2 (en) * | 2004-08-16 | 2012-03-06 | Mitsui Chemicals, Inc. | Ethylene polymer and use thereof |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1734058A1 (en) * | 2005-06-13 | 2006-12-20 | Total Petrochemicals Research Feluy | Ethylene and alpha-olefins polymerisation catalyst system based on fluorenyl ligand |
| EP1930348A1 (en) * | 2006-12-05 | 2008-06-11 | Ineos Manufacturing France SAS | Supported catalyst system |
| DE112008003114T5 (de) * | 2007-11-30 | 2010-10-14 | Sumitomo Chemical Co., Ltd. | Olefinpolymerisationskatalysator und Verfahren zur Herstellung von Olefinpolymer |
| CN101878235B (zh) * | 2007-11-30 | 2012-08-15 | 住友化学株式会社 | 乙烯-α-烯烃共聚物及成型体 |
| US20120123006A1 (en) * | 2009-05-29 | 2012-05-17 | Sumitomo Chemical Company, Limited | Resin composition for crosslinking/foam molding, crosslinked molded foam, member for footwear, and footwear |
| DE112011100914T5 (de) | 2010-03-15 | 2013-01-03 | Sumitomo Chemical Co., Ltd. | Ethylen-a-Olefin-Copolymer und Formteil |
| WO2012053261A1 (ja) * | 2010-10-21 | 2012-04-26 | 三井化学株式会社 | エチレン系重合体粒子の製造方法ならびに該エチレン系重合体粒子から得られる延伸成形体 |
| JP6774184B2 (ja) | 2014-02-13 | 2020-10-21 | 三井化学株式会社 | エチレン・α−オレフィン・非共役ポリエン共重合体およびその用途、並びにその製造方法 |
| JP6253765B2 (ja) * | 2014-03-28 | 2017-12-27 | 三井化学株式会社 | オレフィン系樹脂およびその製造方法 |
| US20200239663A1 (en) * | 2015-08-31 | 2020-07-30 | Prime Polymer Co., Ltd. | Laminate and method for producing the same, and sealant film |
| CN106674471B (zh) | 2015-11-11 | 2019-09-03 | 万华化学集团股份有限公司 | 一种热塑性聚氨酯弹性体及其制备方法、用途和制品 |
| CN108610440A (zh) * | 2016-12-13 | 2018-10-02 | 中国石油天然气股份有限公司 | 烯烃聚合催化剂及其制备方法 |
| EP3555148B1 (en) | 2016-12-15 | 2023-04-05 | Borealis AG | Catalyst system for producing polyethylene copolymers in a high temperature solution polymerization process |
| JP7213183B2 (ja) | 2016-12-15 | 2023-01-26 | ボレアリス エージー | 高温溶液重合プロセスでポリエチレンコポリマーを製造する為の新規な触媒系 |
| US11028192B2 (en) * | 2017-03-27 | 2021-06-08 | Exxonmobil Chemical Patents Inc. | Solution process to make ethylene copolymers |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002008306A1 (fr) * | 2000-07-26 | 2002-01-31 | Mitsui Chemicals, Inc. | Polymere et son procede de production |
| JP2002212217A (ja) * | 2001-01-11 | 2002-07-31 | Mitsui Chemicals Inc | オレフィン重合用触媒およびオレフィンの重合方法 |
| JP2002275332A (ja) * | 2001-03-15 | 2002-09-25 | Mitsui Chemicals Inc | ポリプロピレン樹脂組成物およびその用途 |
| JP2002275342A (ja) * | 2001-03-15 | 2002-09-25 | Mitsui Chemicals Inc | ポリオレフィン樹脂組成物 |
| JP2003020306A (ja) * | 2001-07-10 | 2003-01-24 | Mitsui Chemicals Inc | 新規なオレフィン重合用触媒および該触媒を用いたオレフィンの重合方法 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07278221A (ja) | 1994-02-18 | 1995-10-24 | Mitsui Petrochem Ind Ltd | エチレン系重合体およびその製造方法 |
| CA2142748C (en) | 1994-02-18 | 1999-02-23 | Shin-Ichi Kojoh | Ethylene polymer and process for preparing the same |
| KR100418533B1 (ko) | 1995-01-24 | 2004-06-11 | 이.아이,듀우판드네모아앤드캄파니 | 알파-올레핀및올레핀중합체및그의제조방법 |
| DE69633436T3 (de) * | 1995-07-24 | 2016-06-16 | Mitsui Chemicals, Inc. | Ethylen-Copolymer-Zusammensetzung |
| JP3530020B2 (ja) | 1997-04-25 | 2004-05-24 | 三井化学株式会社 | オレフィン重合用触媒、遷移金属化合物、オレフィンの重合方法およびα−オレフィン・共役ジエン共重合体 |
| TW420693B (en) | 1997-04-25 | 2001-02-01 | Mitsui Chemicals Inc | Olefin polymerization catalysts, transition metal compounds, and <alpha>-olefin/conjugated diene copolymers |
| EP0926169B1 (en) | 1997-12-25 | 2005-02-23 | Mitsui Chemicals, Inc. | Ethylene/Alpha-olefin copolymer and process for preparing the same |
| EP0982362A4 (en) * | 1998-03-10 | 2002-03-06 | Mitsui Chemicals Inc | COMPOSITION OF ETHYLENE COPOLYMER AND USE THEREOF |
| JP2002105132A (ja) | 2000-07-26 | 2002-04-10 | Mitsui Chemicals Inc | ポリマーおよびその製造方法 |
| SG96700A1 (en) * | 2001-09-27 | 2003-06-16 | Sumitomo Chemical Co | Catalyst component for addition polymerization, process for producing catalyst for addition polymerization and process for producing addition polymer |
| US7205358B2 (en) * | 2002-04-24 | 2007-04-17 | Sumitomo Chemical Company, Limited | Method for producing a extruded ethylene copolymer |
| EP2465879B1 (en) | 2002-09-27 | 2019-04-24 | Mitsui Chemicals, Inc. | Bridged metallocene compound for olefin polymerization and method of polymerizing olefin using the same |
-
2004
- 2004-05-19 EP EP04733978.3A patent/EP1632505B1/en not_active Expired - Fee Related
- 2004-05-19 WO PCT/JP2004/007124 patent/WO2004104055A1/ja not_active Application Discontinuation
- 2004-05-19 CN CNB2004800138872A patent/CN100354322C/zh active Active
- 2004-05-19 US US10/553,726 patent/US7452946B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002008306A1 (fr) * | 2000-07-26 | 2002-01-31 | Mitsui Chemicals, Inc. | Polymere et son procede de production |
| JP2002212217A (ja) * | 2001-01-11 | 2002-07-31 | Mitsui Chemicals Inc | オレフィン重合用触媒およびオレフィンの重合方法 |
| JP2002275332A (ja) * | 2001-03-15 | 2002-09-25 | Mitsui Chemicals Inc | ポリプロピレン樹脂組成物およびその用途 |
| JP2002275342A (ja) * | 2001-03-15 | 2002-09-25 | Mitsui Chemicals Inc | ポリオレフィン樹脂組成物 |
| JP2003020306A (ja) * | 2001-07-10 | 2003-01-24 | Mitsui Chemicals Inc | 新規なオレフィン重合用触媒および該触媒を用いたオレフィンの重合方法 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1632505A4 * |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8129489B2 (en) * | 2004-08-16 | 2012-03-06 | Mitsui Chemicals, Inc. | Ethylene polymer and use thereof |
| EP1849805A4 (en) * | 2005-01-31 | 2009-03-11 | Mitsui Chemicals Inc | ETHYLENIC RESIN AND FORM BODY OBTAINED THEREFROM |
| EP1849805A1 (en) * | 2005-01-31 | 2007-10-31 | Mitsui Chemicals, Inc. | Ethylenic resin and formed article obtained therefrom |
| WO2006080578A1 (ja) * | 2005-01-31 | 2006-08-03 | Mitsui Chemicals, Inc. | エチレン系樹脂及びこれから得られる成形体 |
| US7858723B2 (en) | 2005-01-31 | 2010-12-28 | Mitsui Chemicals, Inc. | Ethylene-based resin and molded object obtained therefrom |
| CN101111527B (zh) * | 2005-01-31 | 2010-09-15 | 三井化学株式会社 | 乙烯系树脂和由其得到的成型体 |
| KR100932149B1 (ko) * | 2005-01-31 | 2009-12-16 | 미쓰이 가가쿠 가부시키가이샤 | 에틸렌계 수지 및 이것으로부터 얻어지는 성형체 |
| CN101341176B (zh) * | 2005-12-19 | 2011-08-17 | 东曹株式会社 | 乙烯类聚合物、用于制备乙烯类聚合物的催化剂及制备乙烯类聚合物的方法 |
| US7838611B2 (en) | 2005-12-19 | 2010-11-23 | Tosoh Corporation | Ethylene polymer, catalyst for ethylene polymer production, and method for producing ethylene polymer |
| JP2008031377A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 液体包装用フィルム |
| JP2008031381A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 肉厚フィルムおよびその製造方法ならびにその用途 |
| JP2008031383A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | ラミネート紙用樹脂組成物およびラミネート紙 |
| JP2008031386A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 表面保護フィルム用樹脂組成物および表面保護フィルム |
| JP2008031379A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 低臭気性フィルムおよびその製造方法ならびにその用途 |
| JP2008031374A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 積層体用樹脂組成物および積層体 |
| JP2008031380A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 易引裂き性フィルムおよびその製造方法ならびにその用途 |
| JP2008031385A (ja) * | 2006-07-31 | 2008-02-14 | Mitsui Chemicals Inc | 熱可塑性樹脂用改質材および樹脂組成物 |
| WO2008016059A1 (fr) * | 2006-07-31 | 2008-02-07 | Mitsui Chemicals, Inc. | Film ou stratifié comprenant une résine d'éthylène ou une composition de résine d'éthylène |
| US8299193B2 (en) * | 2006-07-31 | 2012-10-30 | Mitsui Chemicals, Inc. | Film or laminate comprising ethylene-based resin or ethylene-based resin composition |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1632505B1 (en) | 2016-09-07 |
| EP1632505A4 (en) | 2007-07-25 |
| US7452946B2 (en) | 2008-11-18 |
| CN1791619A (zh) | 2006-06-21 |
| EP1632505A1 (en) | 2006-03-08 |
| US20060199925A1 (en) | 2006-09-07 |
| CN100354322C (zh) | 2007-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004104055A1 (ja) | エチレン系重合体 | |
| JP4694975B2 (ja) | オレフィンの重合方法 | |
| JP5154754B2 (ja) | エチレン系重合体およびこれから得られる成形体 | |
| JP5405806B2 (ja) | オレフィン重合用触媒およびそれを用いたエチレン系重合体の製造方法 | |
| SK5482002A3 (en) | Catalyst systems and their use in a polymerization process | |
| JP5587556B2 (ja) | エチレン系重合体、該エチレン系重合体を含む熱可塑性樹脂組成物およびこれらから得られる成形体 | |
| JP2005002333A (ja) | エチレン系重合体 | |
| KR20190071187A (ko) | 장기 내압 특성이 우수한 에틸렌계 중합체 및 이를 이용한 파이프 | |
| JP6049383B2 (ja) | 易カット性ラミネートフィルム | |
| JP6885451B2 (ja) | オレフィン重合体の製造方法 | |
| JP5498926B2 (ja) | 長鎖分岐を有するプロピレン系重合体の製造方法 | |
| JP5256245B2 (ja) | 長鎖分岐を有するプロピレン系重合体の製造方法 | |
| JP2009197226A (ja) | エチレン共重合体、該エチレン共重合体を含む熱可塑性樹脂組成物およびこれらから得られる成形体 | |
| JP5285893B2 (ja) | 溶融物性が改良されたプロピレン系重合体の製造方法 | |
| JP2009173798A (ja) | エチレン系重合体、該エチレン系重合体を含む熱可塑性樹脂組成物およびこれらから得られる成形体 | |
| JP6326202B2 (ja) | オレフィン重合用固体触媒成分およびそれを用いたオレフィン系重合体の製造方法 | |
| JP2010126557A (ja) | オレフィンブロック重合体の製造方法 | |
| JP2013224408A (ja) | オレフィン重合用触媒の製造方法およびこれにより得られるオレフィン重合用触媒を用いたエチレン系重合体の製造方法 | |
| JP6559490B2 (ja) | オレフィン重合用触媒およびエチレン系重合体の合成方法 | |
| JP5769788B2 (ja) | エチレン系重合体、該エチレン系重合体を含む熱可塑性樹脂組成物およびこれらから得られる成形体 | |
| JP5622606B2 (ja) | プロピレン系重合体の製造方法 | |
| JP2005008711A (ja) | 超高分子量オレフィン系重合体の製造方法 | |
| JP5979047B2 (ja) | プロピレン重合用触媒およびプロピレン系重合体の製造方法 | |
| JP6506950B2 (ja) | オレフィン重合用触媒ならびにそれを用いたオレフィンの重合方法 | |
| JP6142766B2 (ja) | α−オレフィン重合用触媒及びそれを用いるα−オレフィン重合体の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 10553726 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2004733978 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004733978 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 5297/DELNP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 20048138872 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004733978 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: JP |
|
| WWP | Wipo information: published in national office |
Ref document number: 10553726 Country of ref document: US |