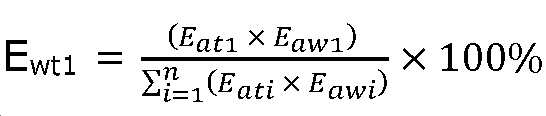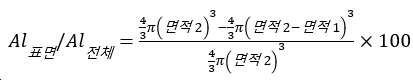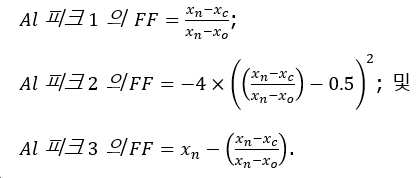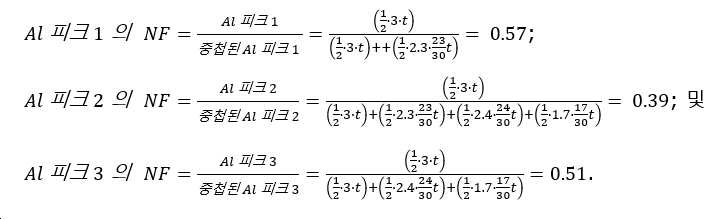KR20220031663A - 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 - Google Patents
재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 Download PDFInfo
- Publication number
- KR20220031663A KR20220031663A KR1020227003786A KR20227003786A KR20220031663A KR 20220031663 A KR20220031663 A KR 20220031663A KR 1020227003786 A KR1020227003786 A KR 1020227003786A KR 20227003786 A KR20227003786 A KR 20227003786A KR 20220031663 A KR20220031663 A KR 20220031663A
- Authority
- KR
- South Korea
- Prior art keywords
- active material
- content
- material powder
- surface layer
- particle
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000007774 positive electrode material Substances 0.000 title claims abstract description 49
- 229910001416 lithium ion Inorganic materials 0.000 title claims abstract description 8
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 title claims abstract description 7
- SOXUFMZTHZXOGC-UHFFFAOYSA-N [Li].[Mn].[Co].[Ni] Chemical compound [Li].[Mn].[Co].[Ni] SOXUFMZTHZXOGC-UHFFFAOYSA-N 0.000 title description 2
- 239000002131 composite material Substances 0.000 title 1
- 239000000843 powder Substances 0.000 claims abstract description 114
- 239000002245 particle Substances 0.000 claims abstract description 112
- 239000002344 surface layer Substances 0.000 claims abstract description 96
- 229910052782 aluminium Inorganic materials 0.000 claims abstract description 50
- 229910052744 lithium Inorganic materials 0.000 claims abstract description 44
- 229910052723 transition metal Inorganic materials 0.000 claims abstract description 42
- 239000006182 cathode active material Substances 0.000 claims abstract description 40
- -1 lithium transition metal Chemical class 0.000 claims abstract description 38
- 229910052717 sulfur Inorganic materials 0.000 claims abstract description 36
- 229910010093 LiAlO Inorganic materials 0.000 claims abstract description 28
- 229910052759 nickel Inorganic materials 0.000 claims abstract description 21
- 229910052748 manganese Inorganic materials 0.000 claims abstract description 20
- 229910052751 metal Inorganic materials 0.000 claims abstract description 16
- 239000002184 metal Substances 0.000 claims abstract description 16
- 239000002019 doping agent Substances 0.000 claims abstract description 15
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims abstract description 12
- 229910052760 oxygen Inorganic materials 0.000 claims abstract description 12
- 239000001301 oxygen Substances 0.000 claims abstract description 12
- 239000011593 sulfur Substances 0.000 claims abstract description 9
- 150000001875 compounds Chemical class 0.000 claims description 56
- 238000000034 method Methods 0.000 claims description 38
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 36
- 239000000203 mixture Substances 0.000 claims description 20
- 238000004458 analytical method Methods 0.000 claims description 18
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 13
- 229910052799 carbon Inorganic materials 0.000 claims description 12
- 238000010438 heat treatment Methods 0.000 claims description 10
- 238000004519 manufacturing process Methods 0.000 claims description 10
- 238000002156 mixing Methods 0.000 claims description 10
- 239000012298 atmosphere Substances 0.000 claims description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 9
- 238000000026 X-ray photoelectron spectrum Methods 0.000 claims description 6
- 229910052721 tungsten Inorganic materials 0.000 claims description 3
- 229910052796 boron Inorganic materials 0.000 claims description 2
- 229910052791 calcium Inorganic materials 0.000 claims description 2
- 229910052804 chromium Inorganic materials 0.000 claims description 2
- 229910052749 magnesium Inorganic materials 0.000 claims description 2
- 229910052758 niobium Inorganic materials 0.000 claims description 2
- 230000001590 oxidative effect Effects 0.000 claims description 2
- 238000010183 spectrum analysis Methods 0.000 claims description 2
- 229910052712 strontium Inorganic materials 0.000 claims description 2
- 229910052719 titanium Inorganic materials 0.000 claims description 2
- 229910052720 vanadium Inorganic materials 0.000 claims description 2
- 229910052727 yttrium Inorganic materials 0.000 claims description 2
- 229910052725 zinc Inorganic materials 0.000 claims description 2
- 229910052726 zirconium Inorganic materials 0.000 claims description 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims 2
- 229910052698 phosphorus Inorganic materials 0.000 claims 2
- 239000011574 phosphorus Substances 0.000 claims 2
- FWFGVMYFCODZRD-UHFFFAOYSA-N oxidanium;hydrogen sulfate Chemical compound O.OS(O)(=O)=O FWFGVMYFCODZRD-UHFFFAOYSA-N 0.000 claims 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 abstract description 27
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 abstract description 5
- 238000004833 X-ray photoelectron spectroscopy Methods 0.000 description 43
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 37
- 239000011572 manganese Substances 0.000 description 30
- 101150088727 CEX1 gene Proteins 0.000 description 28
- 238000009616 inductively coupled plasma Methods 0.000 description 21
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 16
- 230000008569 process Effects 0.000 description 15
- 238000004381 surface treatment Methods 0.000 description 15
- 238000012360 testing method Methods 0.000 description 14
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 12
- 238000009826 distribution Methods 0.000 description 12
- 238000004841 transmission electron microscopy energy-dispersive X-ray spectroscopy Methods 0.000 description 12
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 11
- 239000000243 solution Substances 0.000 description 11
- 238000004364 calculation method Methods 0.000 description 10
- 239000002243 precursor Substances 0.000 description 10
- 150000003624 transition metals Chemical class 0.000 description 10
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 238000000975 co-precipitation Methods 0.000 description 9
- 230000002427 irreversible effect Effects 0.000 description 9
- 238000005259 measurement Methods 0.000 description 9
- 238000005245 sintering Methods 0.000 description 9
- 239000003792 electrolyte Substances 0.000 description 8
- 230000014759 maintenance of location Effects 0.000 description 8
- 239000011164 primary particle Substances 0.000 description 8
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- 101100439211 Caenorhabditis elegans cex-2 gene Proteins 0.000 description 6
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 6
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 239000002002 slurry Substances 0.000 description 6
- 230000001351 cycling effect Effects 0.000 description 5
- 239000013067 intermediate product Substances 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 239000006258 conductive agent Substances 0.000 description 4
- 239000011888 foil Substances 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- 238000012545 processing Methods 0.000 description 4
- 239000011163 secondary particle Substances 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 3
- 101001030537 Homo sapiens FERM domain-containing protein 6 Proteins 0.000 description 3
- 229910021529 ammonia Inorganic materials 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- VNTQORJESGFLAZ-UHFFFAOYSA-H cobalt(2+) manganese(2+) nickel(2+) trisulfate Chemical compound [Mn++].[Co++].[Ni++].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O VNTQORJESGFLAZ-UHFFFAOYSA-H 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 229910002804 graphite Inorganic materials 0.000 description 3
- 239000010439 graphite Substances 0.000 description 3
- 239000012535 impurity Substances 0.000 description 3
- 238000010884 ion-beam technique Methods 0.000 description 3
- 239000008213 purified water Substances 0.000 description 3
- 238000001878 scanning electron micrograph Methods 0.000 description 3
- 238000007086 side reaction Methods 0.000 description 3
- 238000003860 storage Methods 0.000 description 3
- 238000010998 test method Methods 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 2
- 229910013870 LiPF 6 Inorganic materials 0.000 description 2
- 239000002033 PVDF binder Substances 0.000 description 2
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 2
- 239000011149 active material Substances 0.000 description 2
- TYYRFZAVEXQXSN-UHFFFAOYSA-H aluminium sulfate hexadecahydrate Chemical compound O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.O.[Al+3].[Al+3].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O TYYRFZAVEXQXSN-UHFFFAOYSA-H 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 238000003490 calendering Methods 0.000 description 2
- 229940063834 carboxymethylcellulose sodium Drugs 0.000 description 2
- 230000000875 corresponding effect Effects 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000007772 electrode material Substances 0.000 description 2
- 238000005430 electron energy loss spectroscopy Methods 0.000 description 2
- 238000000921 elemental analysis Methods 0.000 description 2
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 238000012886 linear function Methods 0.000 description 2
- 238000011068 loading method Methods 0.000 description 2
- 239000011255 nonaqueous electrolyte Substances 0.000 description 2
- 238000010606 normalization Methods 0.000 description 2
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000035945 sensitivity Effects 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- 239000006245 Carbon black Super-P Substances 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 241000446313 Lamella Species 0.000 description 1
- 229910012258 LiPO Inorganic materials 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 241000156302 Porcine hemagglutinating encephalomyelitis virus Species 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- KFDQGLPGKXUTMZ-UHFFFAOYSA-N [Mn].[Co].[Ni] Chemical compound [Mn].[Co].[Ni] KFDQGLPGKXUTMZ-UHFFFAOYSA-N 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 239000010405 anode material Substances 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 238000000498 ball milling Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000010406 cathode material Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- CKFRRHLHAJZIIN-UHFFFAOYSA-N cobalt lithium Chemical compound [Li].[Co] CKFRRHLHAJZIIN-UHFFFAOYSA-N 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 239000011889 copper foil Substances 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000001186 cumulative effect Effects 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 238000013480 data collection Methods 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 239000011262 electrochemically active material Substances 0.000 description 1
- 239000011883 electrode binding agent Substances 0.000 description 1
- 239000011267 electrode slurry Substances 0.000 description 1
- ZZUFCTLCJUWOSV-UHFFFAOYSA-N furosemide Chemical compound C1=C(Cl)C(S(=O)(=O)N)=CC(C(O)=O)=C1NCC1=CC=CO1 ZZUFCTLCJUWOSV-UHFFFAOYSA-N 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- INHCSSUBVCNVSK-UHFFFAOYSA-L lithium sulfate Inorganic materials [Li+].[Li+].[O-]S([O-])(=O)=O INHCSSUBVCNVSK-UHFFFAOYSA-L 0.000 description 1
- 238000000386 microscopy Methods 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000007773 negative electrode material Substances 0.000 description 1
- 229920006254 polymer film Polymers 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 238000011076 safety test Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000007873 sieving Methods 0.000 description 1
- 238000007581 slurry coating method Methods 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000003746 solid phase reaction Methods 0.000 description 1
- 238000010671 solid-state reaction Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229920003048 styrene butadiene rubber Polymers 0.000 description 1
- 238000005211 surface analysis Methods 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- RBTVSNLYYIMMKS-UHFFFAOYSA-N tert-butyl 3-aminoazetidine-1-carboxylate;hydrochloride Chemical compound Cl.CC(C)(C)OC(=O)N1CC(N)C1 RBTVSNLYYIMMKS-UHFFFAOYSA-N 0.000 description 1
- RBYFNZOIUUXJQD-UHFFFAOYSA-J tetralithium oxalate Chemical compound [Li+].[Li+].[Li+].[Li+].[O-]C(=O)C([O-])=O.[O-]C(=O)C([O-])=O RBYFNZOIUUXJQD-UHFFFAOYSA-J 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 229910021642 ultra pure water Inorganic materials 0.000 description 1
- 239000012498 ultrapure water Substances 0.000 description 1
- 238000010947 wet-dispersion method Methods 0.000 description 1
- 238000004804 winding Methods 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Complex oxides containing nickel and at least one other metal element
- C01G53/42—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2
- C01G53/44—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese
- C01G53/50—Complex oxides containing nickel and at least one other metal element containing alkali metals, e.g. LiNiO2 containing manganese of the type (MnO2)n-, e.g. Li(NixMn1-x)O2 or Li(MyNixMn1-x-y)O2
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
- H01M4/0471—Processes of manufacture in general involving thermal treatment, e.g. firing, sintering, backing particulate active material, thermal decomposition, pyrolysis
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/581—Chalcogenides or intercalation compounds thereof
- H01M4/5815—Sulfides
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/01—Crystal-structural characteristics depicted by a TEM-image
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
- C01P2002/54—Solid solutions containing elements as dopants one element only
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/85—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by XPS, EDX or EDAX data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/03—Particle morphology depicted by an image obtained by SEM
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/51—Particles with a specific particle size distribution
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/61—Micrometer sized, i.e. from 1-100 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/80—Particles consisting of a mixture of two or more inorganic phases
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/40—Electric properties
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/028—Positive electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2220/00—Batteries for particular applications
- H01M2220/20—Batteries in motive systems, e.g. vehicle, ship, plane
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Composite Materials (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
Abstract
Description
도 1.2는 다결정질 형태를 갖는 입자에 대한 SEM 이미지이다.
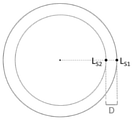
도 2는 본 발명에 따른 양극 활물질 입자의 단면에 대한 개략적 이미지이고, 여기서 LS1은 입자의 단면의 주변의 제1 지점 위치이고, LS2는 상기 입자의 기하학적 중심과 LS1 사이의 가상선에 위치한 제2 지점 위치이다. D는 LS1과 LS2 사이의 거리로 정의된 표면층의 두께이다(cfr. 구현예 9).
도 3.1은 핏팅 과정 전 EX1의 XPS Al 피크 역합성곱이다(x-축: 결합 에너지, y-축: 수).
도 3.2는 핏팅 과정 후 EX1의 XPS Al 피크 역합성곱이다(x-축: 결합 에너지, y-축: 수).
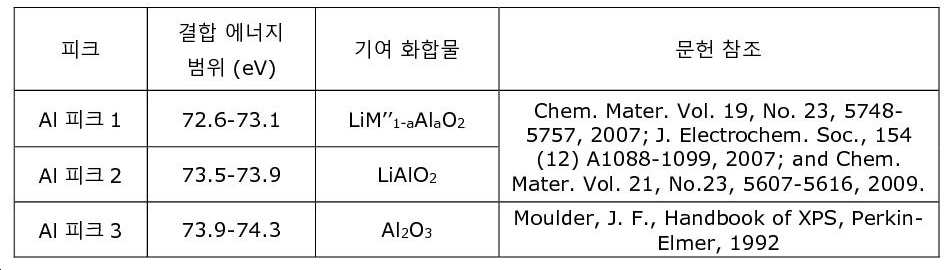
도 4.1은 EX1, CEX1, 및 CEX3의 Al2p 및 Ni3p XPS 스펙트럼이다(x-축: 결합 에너지, y-축: 수).
도 4.2는 각각 LiM"1-aAlaO2, LiAlO2, 및 Al2O3 상의 Al2p3 XPS 피크에 대한 결합 에너지 범위이다. 수직 점선은 CEX1에 대한 Al2p3 XPS 피크 위치를 나타내는 반면, 수직 실선은 EX1에 대한 Al2p3 XPS 피크 위치를 나타낸다.
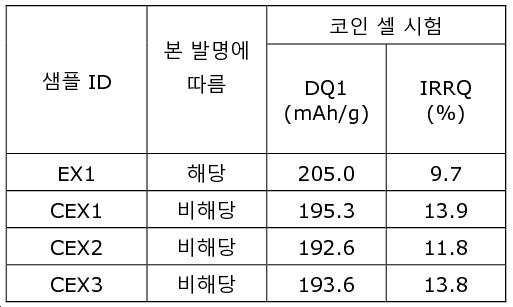
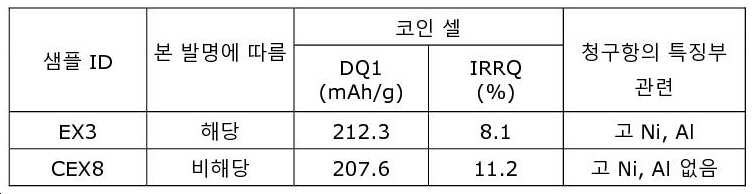
도 5는 25℃ 및 45℃에서 2.7V 내지 4.2V 범위의 EX1, CEX1, 및 CEX3에 대한 풀 셀 시험 결과이다(x-축: 사이클 수, y-축: 용량 보유율).
도 6.1은 EX1의 S에 대한 TEM-EDS 분석 결과이다(x-축: 거리(0은 표면층의 시작점임), y-축: 원소(원자 비율)).
도 6.2는 EX1의 Al에 대한 TEM-EDS 분석 결과이다(x-축: 거리(0은 표면층의 시작점임), y-축: 원소(원자 비율)).
Claims (16)
- 리튬 전이 금속-기반 산화물 입자를 포함하는, 리튬-이온 배터리에 적합한 양극 활물질 분말로서, 상기 입자가 코어 및 표면층을 포함하고, 상기 표면층이 상기 코어의 상부에 있고, 상기 입자가 원소 Li, 금속 M' 및 산소를 포함하고, 여기서 금속 M'는 식 M'=(NizMnyCox)1-kAk를 갖고, 상기 식에서 A는 도펀트이고, 0.60≤z<0.86, 0.05≤x≤0.20, x+y+z+k=1, 및 k≤0.01이고, 상기 양극 활물질 분말이 5μm 내지 15μm 범위의 중간 입자 크기 D50 및 10nm 내지 200nm 범위의 표면층 두께를 갖고,
상기 표면층이
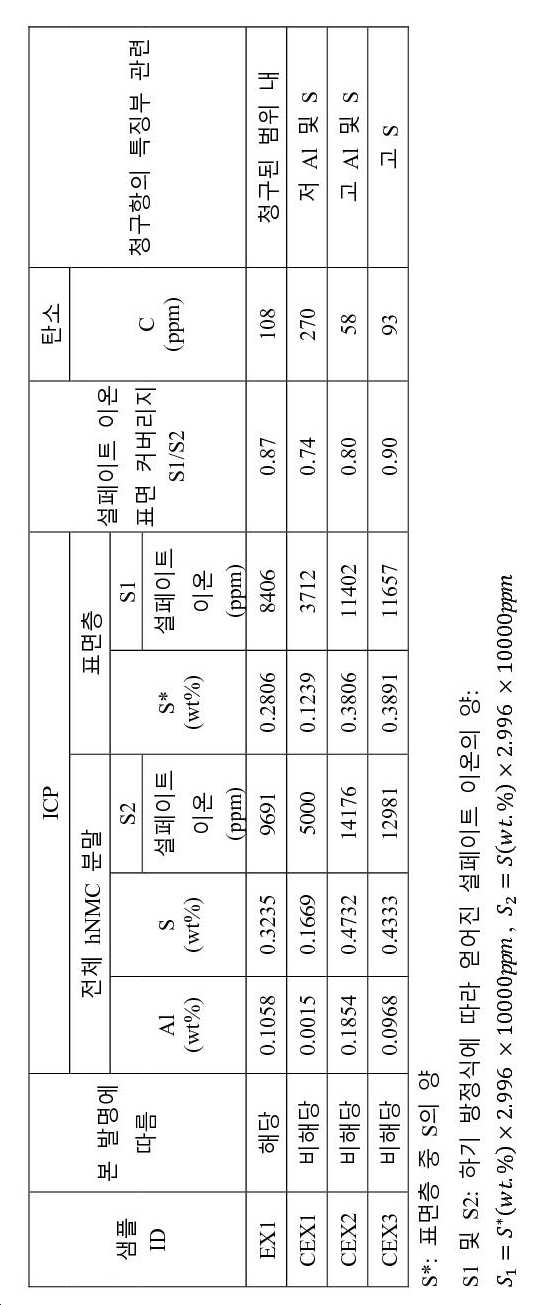
- 양극 활물질 분말의 총 중량에 대해 0.150wt% 이상 및 0.375wt% 이하의 함량의 황, 및
- 양극 활물질 분말의 총 중량에 대해 0.05wt% 이상 및 0.15wt% 이하의 함량의 알루미늄
을 포함하고, 상기 리튬 전이 금속-기반 산화물 입자의 표면층이 LiAlO2 상(phase) 및 LiM"1-aAlaO2 상을 포함하고, 여기서 M"이 Ni, Mn, 및 Co를 포함하고, 상기 LiAlO2 상이 양극 활물질 분말 중 M'의 총 원자 함량에 대해 0.10at% 이상 및 0.30at% 이하의 함량으로 표면층에 존재하고, 상기 LiM"1-aAlaO2 상이 양극 활물질 분말 중 M'의 총 원자 함량에 대해 0.14at% 미만의 함량으로 표면층에 존재하는, 양극 활물질 분말. - 제1항에 있어서, 상기 입자가 원소 Li, M' 및 산소를 포함하고, 여기서 M'는 식 M'=NizMnyCoxAk를 갖고, 상기 식에서 A는 도펀트이고, 0.70≤z<0.86, 0.05≤y≤0.20, 0.05≤x≤0.20, x+y+z+k=1, 및 k≤0.01인, 양극 활물질 분말.
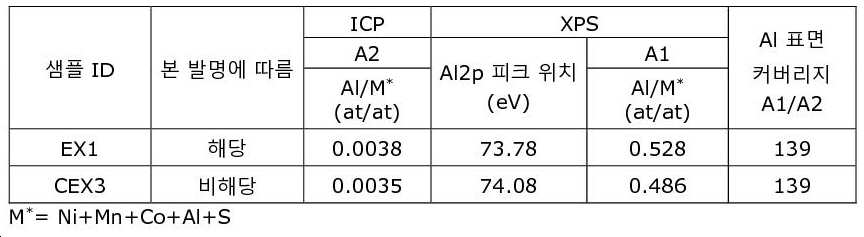
- 제1항 또는 제2항에 있어서, 상기 리튬 전이 금속-기반 산화물 입자가 100 이상인 Al 표면 커버리지 A1/A2를 갖고, 여기서 A1은 표면층에 함유된 원소 Al, Ni, Mn, Co, 및 S의 원자 비율 Al/(Ni+Mn+Co+Al+S)이고, 상기 원자 비율 A1은 XPS 스펙트럼 분석에 의해 얻어지고, A2는 ICP에 의해 얻어지는 원자 비율 Al/(Ni+Mn+Co+Al+S)인, 양극 활물질 분말.
- 제1항 내지 제3항 중 어느 한 항에 있어서, 200ppm 미만의 탄소 함량을 갖는 양극 활물질 분말.
- 제1항 내지 제4항 중 어느 한 항에 있어서, 0.96 이상 및 1.05 이하의 Li/(Ni+Mn+Co+A) 원자 비율 또는 Li/(Ni+Mn+Co+A+Al) 원자 비율을 갖는 양극 활물질 분말.
- 제3항 내지 제5항 중 어느 한 항에 있어서, 상기 표면층이 73.0±0.2eV 내지 74.5±0.2eV, 바람직하게는 73.6±0.2eV 내지 74.1±0.2eV의 결합 에너지 범위에서 최대 피크 세기를 갖는 Al2p 피크를 나타내고, 상기 세기가 XPS 스펙트럼 분석에 의해 얻어지는, 양극 활물질 분말.
- 제1항 내지 제6항 중 어느 한 항에 있어서, 상기 리튬 전이 금속-기반 산화물 입자의 표면층이 4500ppm 이상 및 11250ppm 이하의 함량의 설페이트 이온(SO4 2-)을 갖는, 양극 활물질 분말.
- 제1항 내지 제7항 중 어느 한 항에 있어서, 상기 리튬 전이 금속-기반 산화물 입자가 0.85 초과 및 1.00 이하인 설페이트 이온 표면 커버리지 S1/S2를 갖고, S1이 표면층에 함유된 설페이트 이온의 양이고, S2가 입자 중의 설페이트 이온의 총량인, 양극 활물질 분말.
- 제1항 내지 제8항 중 어느 한 항에 있어서, 일반식 Li1+a'((Niz'My'Cox'AlvSw)1-kAk)1-aO2를 갖고, 여기서 오로지 A만이 도펀트이고, 0.60≤z'<0.86, 0.05≤y'≤0.20, 0.05≤x'≤0.20, 및 x'+y'+z'+v+w+k=1, 0.0018≤v≤0.0053, 0.006≤w≤0.012, -0.05≤a'≤0.05, 및 k≤0.01인, 양극 활물질 분말.
- 제1항 내지 제9항 중 어느 한 항에 있어서, A가 Al, B, S, Mg, Zr, Nb, W, Si, Ba, Sr, Ca, Zn, Cr, V, Y, 및 Ti 중 하나 이상이고, A의 각 원소의 양이 양극 활물질 분말의 총 중량에 대해 100ppm을 초과하는, 양극 활물질 분말.
- 제1항 내지 제10항 중 어느 한 항에 있어서, 표면층의 두께가 하기와 같이 정의되는 최소 거리 D에 해당하는, 양극 활물질 분말:
여기서, LS1은 입자의 중심의 제1 지점 위치이고, LS2는 상기 제1 지점 위치와 상기 입자의 기하학적 중심 사이에 정의된 선의 제2 지점 위치이고,
제2 지점 위치 LS2에서 TEM-EDS에 의해 측정된 S의 함량은 제1 지점 위치에서 측정된 S의 함량의 0at% 이상 및 5.0at% 미만이고, 상기 S의 제2 함량(S2)은
와 같이 정의되고, S3는 상기 선의 제3 지점 위치(LS3)에서의 S의 함량(at%)이고, 상기 제3 지점은 상기 입자의 기하학적 중심과 제2 지점 위치 LS2 사이의 임의의 위치에 위치한다. - 제11항 또는 제12항에 있어서, Al이 하기와 같이 정의되는 함량 l로 표면층에 존재하는, 양극 활물질 분말:
여기서,
- 는 ICP에 의해 측정된, 분말 중 M* 함량에 대한 Al 함량의 원자 비율이고,
- 이고,
여기서,
- Al표면은 EDS에 의해 측정된, 표면층 중 Al의 함량(at%)이고;
- Al전체는 EDS에 의해 측정된, 상기 분말의 입자 중 Al의 총 함량(at%)이고;
- 면적1은 D에 걸쳐 단면-TEM-EDS에 의해 측정된 Al/M* 함량의 적분이고:
- Al(x)는 단면-TEM EDS에 의해 측정된, 단면 입자의 지점 x에서의 Al의 원자 함량이고,
- M*(x)은 단면-TEM EDS에 의해 측정된, 단면 입자의 지점 x에서의 Ni, Mn, Co, Al, 및 S의 원자 함량이고,
- x는 상기 제1 지점 위치와 제2 지점 위치 사이의, TEM에 의해 측정된 nm로 표현되는 거리이고;
- 면적2는 거리 C에 걸쳐 크로스-SEM EDS에 의해 측정된 Al/M* 함량의 적분이며:
- Al(x)는 단면-TEM EDS에 의해 측정된, 단면 입자의 지점 x에서의 Al의 원자 함량이고,
- M*(x)은 단면-TEM EDS에 의해 측정된, 단면 입자의 지점 x에서의 Ni, Mn, Co, Al, 및 S의 원자 함량이고,
- x는 상기 제1 지점 위치(x = 0nm)와 상기 입자의 기하학적 중심(x = C) 사이의, nm로 표현되고 TEM에 의해 측정된 거리이고, 여기서 C는 바람직하게는 2.5μm 내지 7.5μm의 범위이다. - 제1항 내지 제13항 중 어느 한 항에 따른 양극 활물질의 제조 방법으로서,
a) 리튬 전이 금속-기반 산화물 화합물을 준비하는 단계,
b) 상기 리튬 전이 금속-기반 산화물 화합물을 설페이트 이온의 공급원 및 물과 혼합함으로써 혼합물을 얻는 단계, 및
c) 노(furnace)에서 산화 분위기 하에 350℃ 내지 500℃ 미만, 바람직하게는 최대 450℃의 온도에서 1시간 내지 10시간의 시간 동안 혼합물을 가열하여 본 발명에 따른 양극 활물질 분말을 얻는 단계
의 연속 단계를 포함하는, 상기 양극 활물질의 제조 방법. - 제1항 내지 제13항 중 어느 한 항에 따른 양극 활물질 분말을 포함하는 배터리.
- 전기 차량 또는 하이브리드 전기 차량에서의 제15항에 따른 배터리의 용도.
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP19184165 | 2019-07-03 | ||
| EP19184186.5 | 2019-07-03 | ||
| EP19184186 | 2019-07-03 | ||
| EP19184165.9 | 2019-07-03 | ||
| PCT/EP2020/068718 WO2021001498A1 (en) | 2019-07-03 | 2020-07-02 | Lithium nickel manganese cobalt composite oxide as a positive electrode active material for rechargeable lithium ion batteries |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20220031663A true KR20220031663A (ko) | 2022-03-11 |
| KR102790741B1 KR102790741B1 (ko) | 2025-04-03 |
Family
ID=71409430
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020227003786A Active KR102790741B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003773A Active KR102628723B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003734A Active KR102785075B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003754A Active KR102801181B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003758A Active KR102806753B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
Family Applications After (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020227003773A Active KR102628723B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003734A Active KR102785075B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003754A Active KR102801181B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| KR1020227003758A Active KR102806753B1 (ko) | 2019-07-03 | 2020-07-02 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
Country Status (10)
| Country | Link |
|---|---|
| US (5) | US12294077B2 (ko) |
| EP (5) | EP3994747B1 (ko) |
| JP (5) | JP7410984B2 (ko) |
| KR (5) | KR102790741B1 (ko) |
| CN (5) | CN114051487B (ko) |
| ES (2) | ES3055642T3 (ko) |
| FI (1) | FI3994749T3 (ko) |
| HU (2) | HUE070260T2 (ko) |
| PL (3) | PL3994748T3 (ko) |
| WO (5) | WO2021001505A1 (ko) |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102652332B1 (ko) * | 2019-03-06 | 2024-03-27 | 주식회사 엘지에너지솔루션 | 음극 활물질 및 이를 포함하는 리튬이차전지 |
| KR102734799B1 (ko) * | 2019-07-03 | 2024-11-27 | 유미코아 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| CN114051487B (zh) * | 2019-07-03 | 2024-03-22 | 尤米科尔公司 | 作为用于可再充电锂离子电池的正电极活性材料的锂镍锰钴复合氧化物 |
| US20240250257A1 (en) * | 2021-05-27 | 2024-07-25 | Umicore | Lithium nickel-based composite oxide as a positive electrode active material for rechargeable lithium-ion batteries |
| CA3242587A1 (en) * | 2021-12-16 | 2023-06-22 | Umicore | A positive electrode active material for rechargeable solid-state batteries |
| CN114530595B (zh) * | 2022-01-21 | 2024-08-27 | 珠海冠宇电池股份有限公司 | 一种磷酸锰铁锂正极材料及包括该材料的正极片和电池 |
| CN115448386B (zh) * | 2022-11-14 | 2023-02-28 | 宜宾锂宝新材料有限公司 | 一种中空结构前驱体、正极材料及其制备方法 |
| CN116169261B (zh) * | 2022-12-22 | 2026-02-24 | 北京当升材料科技股份有限公司 | 单晶型多元正极材料及其制备方法和锂离子电池 |
| KR20250133735A (ko) * | 2023-01-05 | 2025-09-08 | 유미코아 | 리튬 복합 산화물 제조에 사용되는 수산화리튬 기반 분말, 상기 분말 및 전이 금속 수산화물 또는 옥시수산화물을 포함하는 조성물 및 상기 분말의 제조 방법 |
| KR20250062877A (ko) * | 2023-10-31 | 2025-05-08 | 삼성에스디아이 주식회사 | 양극 활물질과 그 제조 방법, 이를 포함하는 양극, 및 리튬 이차 전지 |
| WO2025132335A1 (en) * | 2023-12-18 | 2025-06-26 | Umicore | Positive electrode active material powder and method for manufacturing a positive electrode active material powder |
| WO2025164382A1 (ja) * | 2024-02-02 | 2025-08-07 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池用正極活物質および非水電解質二次電池 |
| WO2025164374A1 (ja) * | 2024-02-02 | 2025-08-07 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池用正極活物質および非水電解質二次電池 |
| WO2025164411A1 (ja) * | 2024-02-02 | 2025-08-07 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池用正極活物質および非水電解質二次電池 |
| WO2025228856A1 (en) * | 2024-04-29 | 2025-11-06 | Umicore | Positive electrode composite material |
| WO2026048786A1 (ja) * | 2024-08-30 | 2026-03-05 | パナソニックIpマネジメント株式会社 | 正極活物質および正極活物質の製造方法 |
| CN118833881B (zh) * | 2024-09-20 | 2025-02-18 | 河南科隆新能源股份有限公司 | 一种基于调控奥斯瓦尔德熟化速率的极窄分布正极材料前驱体及其制备方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20140051926A (ko) * | 2011-08-05 | 2014-05-02 | 아사히 가라스 가부시키가이샤 | 리튬 이온 이차 전지용 정극 활물질 |
| KR20170118091A (ko) * | 2015-01-23 | 2017-10-24 | 유미코아 | 고전압 리튬 이온 배터리용 리튬 니켈-망간-코발트 산화물 캐소드 분말 |
| KR20190008120A (ko) * | 2017-07-14 | 2019-01-23 | 유미코아 | 충전식 리튬 이온 배터리용 Ni계 캐소드 재료 |
Family Cites Families (80)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6787232B1 (en) | 1998-04-30 | 2004-09-07 | Massachusetts Institute Of Technology | Intercalation compounds and electrodes for batteries |
| JP3649953B2 (ja) | 1999-06-23 | 2005-05-18 | 三洋電機株式会社 | 活物質、電極、非水電解液二次電池及び活物質の製造方法 |
| JP2002015739A (ja) | 2000-04-26 | 2002-01-18 | Mitsubishi Chemicals Corp | リチウム二次電池用正極材料 |
| JP4995382B2 (ja) | 2001-07-05 | 2012-08-08 | 日本化学工業株式会社 | リチウムコバルト系複合酸化物、その製造方法、、リチウム二次電池正極活物質及びリチウム二次電池 |
| US6878490B2 (en) | 2001-08-20 | 2005-04-12 | Fmc Corporation | Positive electrode active materials for secondary batteries and methods of preparing same |
| KR100490549B1 (ko) * | 2003-02-07 | 2005-05-17 | 삼성에스디아이 주식회사 | 카본 화합물이 흡착된 정극 활물질 및 이를 채용한 리튬전지 |
| US7566479B2 (en) | 2003-06-23 | 2009-07-28 | Lg Chem, Ltd. | Method for the synthesis of surface-modified materials |
| EP1742281B1 (en) * | 2004-04-27 | 2011-09-07 | Mitsubishi Chemical Corporation | Layered lithium nickel manganese cobalt composite oxide powder for material of positive electrode of lithium secondary battery, process for producing the same, positive electrode of lithium secondary battery therefrom, and lithium secondary battery |
| JP4923397B2 (ja) | 2004-09-06 | 2012-04-25 | 日産自動車株式会社 | 非水電解質リチウムイオン二次電池用正極材料およびその製造方法 |
| JP4997700B2 (ja) * | 2004-12-13 | 2012-08-08 | 三菱化学株式会社 | リチウム二次電池正極材料用リチウムニッケルマンガン系複合酸化物粉体及びその製造方法、並びにそれを用いたリチウム二次電池用正極及びリチウム二次電池 |
| JP4211865B2 (ja) | 2006-12-06 | 2009-01-21 | 戸田工業株式会社 | 非水電解質二次電池用Li−Ni複合酸化物粒子粉末及びその製造方法、並びに非水電解質二次電池 |
| US20110003200A1 (en) * | 2006-12-26 | 2011-01-06 | Mitsubishi Chemical Corporation | Lithium transition metal based compound powder, method for manufacturing the same, spray-dried substance serving as firing precursor thereof, lithium secondary battery positive electrode by using the same, and lithium secondary battery |
| JP5153156B2 (ja) * | 2007-02-13 | 2013-02-27 | 三洋電機株式会社 | 非水電解質二次電池用正極の製造方法 |
| EP2202828B1 (en) * | 2007-09-04 | 2013-12-11 | Mitsubishi Chemical Corporation | Lithium transition metal-type compound powder, method for manufacturing the same and lithium secondary battery positive electrode and lithium secondary battery using the same |
| JP5515211B2 (ja) | 2007-12-14 | 2014-06-11 | ソニー株式会社 | 正極活物質の製造方法 |
| JP5251401B2 (ja) | 2008-09-29 | 2013-07-31 | 住友金属鉱山株式会社 | 非水系電解質二次電池用正極活物質及びその製造方法、並びに非水系電解質二次電池 |
| JP2010129471A (ja) | 2008-11-28 | 2010-06-10 | Sony Corp | 正極活物質および非水電解質電池 |
| CN101447566B (zh) * | 2008-12-29 | 2010-08-11 | 清华大学深圳研究生院 | 层状-尖晶石共生结构锂离子电池正极材料及制备方法 |
| JP5584456B2 (ja) | 2009-12-10 | 2014-09-03 | 日本化学工業株式会社 | リチウム二次電池用正極活物質、その製造方法及びリチウム二次電池 |
| JP2012003891A (ja) * | 2010-06-15 | 2012-01-05 | Nissan Motor Co Ltd | 活物質の製造方法 |
| EP2606524B1 (en) * | 2010-08-17 | 2016-10-12 | Umicore | Aluminum dry-coated and heat treated cathode material precursors |
| EP2421077B1 (en) * | 2010-08-17 | 2013-10-23 | Umicore | Positive electrode materials combining high safety and high power in a Li rechargeable battery |
| CA2807229A1 (en) * | 2010-08-17 | 2012-02-23 | Umicore | Alumina dry-coated cathode material precursors |
| JP2013175412A (ja) * | 2012-02-27 | 2013-09-05 | Sumitomo Electric Ind Ltd | 非水電解質電池 |
| CN102627332B (zh) * | 2012-04-20 | 2014-05-28 | 湖北万润新能源科技发展有限公司 | 氧化物固溶体及其制备方法以及锂离子电池正极材料及其制备方法 |
| JP5204913B1 (ja) | 2012-04-27 | 2013-06-05 | 三井金属鉱業株式会社 | 層構造を有するリチウム金属複合酸化物 |
| CN103515606B (zh) | 2012-06-21 | 2016-09-14 | 中国科学院宁波材料技术与工程研究所 | 高能量密度锂离子电池氧化物正极材料及其制备方法 |
| CN102832389B (zh) | 2012-09-25 | 2015-04-15 | 湖南长远锂科有限公司 | 表面改性的锂离子电池高镍正极活性材料及其制备方法 |
| JP6365305B2 (ja) * | 2012-11-21 | 2018-08-01 | 日本電気株式会社 | リチウム二次電池 |
| US9761907B2 (en) * | 2013-05-07 | 2017-09-12 | Dowa Holdings Co., Ltd. | Positive-electrode active-material powder and manufacturing method therefor |
| KR101785262B1 (ko) * | 2013-07-08 | 2017-10-16 | 삼성에스디아이 주식회사 | 양극 활물질, 그 제조방법, 이를 채용한 양극 및 리튬이차전지 |
| CN103441261A (zh) * | 2013-08-14 | 2013-12-11 | 湖南化工研究院 | 一种低so42-含量的锰酸锂正极材料的制备方法 |
| TWI521778B (zh) * | 2013-09-05 | 2016-02-11 | 烏明克公司 | 用於含高鋰和錳的陰極材料之碳酸鹽先質 |
| JP6207329B2 (ja) | 2013-10-01 | 2017-10-04 | 日立マクセル株式会社 | 非水二次電池用正極材料及びその製造方法、並びに該非水二次電池用正極材料を用いた非水二次電池用正極合剤層、非水二次電池用正極及び非水二次電池 |
| TWI525889B (zh) | 2013-10-31 | 2016-03-11 | Lg化學股份有限公司 | 陰極活性物質、其製備方法、含有其的陰極及含有所述陰極的鋰二次電池 |
| JP6359323B2 (ja) | 2013-12-27 | 2018-07-18 | 住友化学株式会社 | リチウムイオン二次電池正極用の表面修飾リチウム含有複合酸化物 |
| CN103715424B (zh) | 2014-01-06 | 2016-06-08 | 中国科学院宁波材料技术与工程研究所 | 一种核壳结构正极材料及其制备方法 |
| US11557754B2 (en) * | 2014-01-27 | 2023-01-17 | Sumitomo Chemical Company, Limited | Positive electrode active material for lithium secondary batteries, positive electrode for lithium secondary batteries, and lithium secondary battery |
| TWI581489B (zh) | 2014-02-27 | 2017-05-01 | 烏明克公司 | 具有經氧化表面之含硫酸鹽的可充電電池陰極 |
| KR101847003B1 (ko) * | 2014-03-31 | 2018-04-10 | 히타치 긴조쿠 가부시키가이샤 | 리튬이온 이차전지용 양극 활물질, 그 제조 방법 및 리튬이온 이차전지 |
| JP6128393B2 (ja) * | 2014-06-04 | 2017-05-17 | トヨタ自動車株式会社 | 非水電解液二次電池および該電池の製造方法 |
| EP3167390A1 (en) | 2014-07-08 | 2017-05-17 | Nestec S.A. | Systems and methods for providing animal health, nutrition, and/or wellness recommendations |
| CN104183834A (zh) * | 2014-08-15 | 2014-12-03 | 南京师范大学 | 一种锂硫电池正极用硫/二氧化硅核壳纳米结构的制备方法 |
| GB2539612B (en) * | 2014-09-03 | 2021-12-01 | Mitsui Mining & Smelting Co Ltd | Positive electrode active material for lithium secondary cell |
| CN104347853B (zh) | 2014-09-24 | 2017-04-12 | 秦皇岛中科远达电池材料有限公司 | 一种锰酸锂复合正极材料、其制备方法及锂离子电池 |
| KR101758992B1 (ko) * | 2014-10-02 | 2017-07-17 | 주식회사 엘지화학 | 리튬 이차전지용 양극활물질, 이의 제조방법 및 이를 포함하는 리튬 이차전지 |
| US10388944B2 (en) * | 2014-10-06 | 2019-08-20 | Hitachi Metals, Ltd. | Positive electrode active material for lithium ion secondary battery, and positive electrode for lithium ion secondary battery and lithium ion secondary battery comprising the same |
| PL3204973T3 (pl) * | 2014-10-08 | 2019-09-30 | Umicore | Materiał katodowy zawierający zanieczyszczenia o korzystnej morfologii i sposób wytwarzania z węglanu metalu zawierającego zanieczyszczenia |
| GB2533161C (en) * | 2014-12-12 | 2019-07-24 | Nexeon Ltd | Electrodes for metal-ion batteries |
| KR20160081545A (ko) | 2014-12-31 | 2016-07-08 | 주식회사 에코프로 | 양극활물질 및 이의 제조 방법 |
| PL3248233T3 (pl) | 2015-01-23 | 2021-12-27 | Umicore | Proszki katodowe tlenku metalu i litu do wysokonapięciowych akumulatorów litowo-jonowych |
| JP2016162601A (ja) * | 2015-03-02 | 2016-09-05 | Jx金属株式会社 | リチウムイオン電池用正極活物質の製造方法、リチウムイオン電池用正極活物質、リチウムイオン電池用正極及びリチウムイオン電池 |
| JP6723074B2 (ja) | 2015-06-01 | 2020-07-15 | マクセルホールディングス株式会社 | リチウムイオン二次電池 |
| TWI586025B (zh) * | 2015-07-02 | 2017-06-01 | 烏明克公司 | 基於鈷之鋰金屬氧化物陰極材料 |
| KR20200021564A (ko) * | 2015-09-08 | 2020-02-28 | 유미코아 | 충전식 배터리용 Li 전이 금속 산화물 캐소드의 제조를 위한 전구체 및 방법 |
| KR102115685B1 (ko) * | 2015-09-30 | 2020-05-27 | 유미코아 | 충전식 배터리용 리튬 전이금속 산화물 캐소드 재료를 위한 전구체 |
| KR102699209B1 (ko) * | 2015-11-05 | 2024-08-26 | 스미또모 가가꾸 가부시끼가이샤 | 리튬 2 차 전지용 정극 활물질, 리튬 2 차 전지용 정극 활물질의 제조 방법, 리튬 2 차 전지용 정극 및 리튬 2 차 전지 |
| KR102580235B1 (ko) | 2015-12-22 | 2023-09-19 | 삼성에스디아이 주식회사 | 양극 활물질, 이를 채용한 리튬 전지, 및 상기 양극 활물질의 제조방법 |
| TWI633692B (zh) | 2016-03-31 | 2018-08-21 | 烏明克公司 | 供汽車應用的鋰離子電池組 |
| CN106058188A (zh) | 2016-07-14 | 2016-10-26 | 中南大学 | 一种具有核壳结构的锂离子电池复合正极材料LiNi1‑x‑yMxAlyO2及其制备方法 |
| KR20230122178A (ko) | 2016-08-25 | 2023-08-22 | 토프쉐 에이/에스 | 고 전압 2차 배터리를 위한 캐소드 활성 물질 |
| JP6412094B2 (ja) | 2016-12-26 | 2018-10-24 | 住友化学株式会社 | リチウム二次電池用正極活物質、リチウム二次電池用正極及びリチウム二次電池 |
| EP3437149B1 (en) * | 2017-02-22 | 2019-10-02 | Lionano Inc. | Core-shell electroactive materials |
| EP3589585A1 (en) * | 2017-03-03 | 2020-01-08 | Umicore | PRECURSOR AND METHOD FOR PREPARING Ni BASED CATHODE MATERIAL FOR RECHARGEABLE LITHIUM ION BATTERIES |
| US12315915B2 (en) | 2017-07-14 | 2025-05-27 | Umicore | Ni based cathode material for rechargeable lithium-ion batteries |
| CN108023077A (zh) | 2017-11-30 | 2018-05-11 | 宁波容百新能源科技股份有限公司 | 一种快离子导体包覆改性高镍正极材料及其制备方法 |
| WO2019123306A1 (en) * | 2017-12-22 | 2019-06-27 | Umicore | A positive electrode material for rechargeable lithium ion batteries and methods of making thereof |
| PL3728129T3 (pl) * | 2017-12-22 | 2022-01-17 | Umicore | Materiał elektrody dodatniej do akumulatorów litowo-jonowych |
| CN112004779B (zh) | 2018-03-29 | 2023-02-07 | 尤米科尔公司 | 用于制备可再充电锂离子蓄电池的正极材料的方法 |
| CN108807887A (zh) | 2018-05-31 | 2018-11-13 | 电子科技大学 | 一种铝氟双重改性锂离子电池正极材料及其制备方法 |
| CN109037649A (zh) | 2018-08-13 | 2018-12-18 | 河北省科学院能源研究所 | 一种掺杂包覆改性的镍钴铝酸锂正极材料及其制备方法 |
| CN108751265B (zh) * | 2018-09-04 | 2021-04-20 | 北京当升材料科技股份有限公司 | 一种锂离子电池正极材料及其前驱体的制备方法 |
| WO2020066848A1 (ja) | 2018-09-28 | 2020-04-02 | パナソニックIpマネジメント株式会社 | 非水電解質二次電池用正極活物質、非水電解質二次電池、及び非水電解質二次電池用正極活物質の製造方法 |
| KR20210061423A (ko) | 2018-10-02 | 2021-05-27 | 바스프 에스이 | 부분 코팅된 전극 활물질의 제조 방법 |
| KR102263998B1 (ko) * | 2018-11-02 | 2021-06-11 | 삼성에스디아이 주식회사 | 리튬 이차 전지용 양극 활물질, 이의 제조 방법 및 이를 포함하는 리튬 이차 전지 |
| CN109742344B (zh) * | 2018-12-21 | 2022-07-19 | 贵州振华新材料股份有限公司 | 低游离锂的氧化铝包覆高镍正极材料、制法和应用 |
| WO2020149910A1 (en) * | 2019-01-17 | 2020-07-23 | Camx Power Llc | Polycrystalline metal oxides with enriched grain boundaries |
| KR102734799B1 (ko) * | 2019-07-03 | 2024-11-27 | 유미코아 | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
| CN114051487B (zh) * | 2019-07-03 | 2024-03-22 | 尤米科尔公司 | 作为用于可再充电锂离子电池的正电极活性材料的锂镍锰钴复合氧化物 |
| JP7513983B2 (ja) * | 2020-09-17 | 2024-07-10 | マツダ株式会社 | リチウムイオン二次電池及びその製造方法 |
-
2020
- 2020-07-02 CN CN202080048390.3A patent/CN114051487B/zh active Active
- 2020-07-02 WO PCT/EP2020/068730 patent/WO2021001505A1/en not_active Ceased
- 2020-07-02 JP JP2021577622A patent/JP7410984B2/ja active Active
- 2020-07-02 US US17/623,676 patent/US12294077B2/en active Active
- 2020-07-02 JP JP2021577598A patent/JP7328372B2/ja active Active
- 2020-07-02 CN CN202080048758.6A patent/CN114072937B/zh active Active
- 2020-07-02 ES ES20736670T patent/ES3055642T3/es active Active
- 2020-07-02 EP EP20735596.7A patent/EP3994747B1/en active Active
- 2020-07-02 ES ES20735596T patent/ES3009908T3/es active Active
- 2020-07-02 PL PL20736670.9T patent/PL3994748T3/pl unknown
- 2020-07-02 WO PCT/EP2020/068728 patent/WO2021001503A1/en not_active Ceased
- 2020-07-02 KR KR1020227003786A patent/KR102790741B1/ko active Active
- 2020-07-02 US US17/623,694 patent/US12368161B2/en active Active
- 2020-07-02 US US17/623,720 patent/US12288872B2/en active Active
- 2020-07-02 HU HUE20735596A patent/HUE070260T2/hu unknown
- 2020-07-02 CN CN202080051238.0A patent/CN114127992B/zh active Active
- 2020-07-02 CN CN202080047688.2A patent/CN114072360B/zh active Active
- 2020-07-02 US US17/623,729 patent/US12362350B2/en active Active
- 2020-07-02 EP EP20736670.9A patent/EP3994748B1/en active Active
- 2020-07-02 KR KR1020227003773A patent/KR102628723B1/ko active Active
- 2020-07-02 FI FIEP20736671.7T patent/FI3994749T3/fi active
- 2020-07-02 PL PL20735596.7T patent/PL3994747T3/pl unknown
- 2020-07-02 HU HUE20736671A patent/HUE062129T2/hu unknown
- 2020-07-02 KR KR1020227003734A patent/KR102785075B1/ko active Active
- 2020-07-02 WO PCT/EP2020/068718 patent/WO2021001498A1/en not_active Ceased
- 2020-07-02 EP EP20735595.9A patent/EP3994746A1/en active Pending
- 2020-07-02 JP JP2022500092A patent/JP7343682B2/ja active Active
- 2020-07-02 JP JP2021577596A patent/JP7343625B2/ja active Active
- 2020-07-02 KR KR1020227003754A patent/KR102801181B1/ko active Active
- 2020-07-02 CN CN202080048348.1A patent/CN114041216B/zh active Active
- 2020-07-02 WO PCT/EP2020/068714 patent/WO2021001496A1/en not_active Ceased
- 2020-07-02 EP EP20736673.3A patent/EP3994750A1/en active Pending
- 2020-07-02 KR KR1020227003758A patent/KR102806753B1/ko active Active
- 2020-07-02 WO PCT/EP2020/068724 patent/WO2021001501A1/en not_active Ceased
- 2020-07-02 EP EP20736671.7A patent/EP3994749B1/en active Active
- 2020-07-02 US US17/623,682 patent/US12381206B2/en active Active
- 2020-07-02 JP JP2022500096A patent/JP7376673B2/ja active Active
- 2020-07-02 PL PL20736671.7T patent/PL3994749T3/pl unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20140051926A (ko) * | 2011-08-05 | 2014-05-02 | 아사히 가라스 가부시키가이샤 | 리튬 이온 이차 전지용 정극 활물질 |
| KR20170118091A (ko) * | 2015-01-23 | 2017-10-24 | 유미코아 | 고전압 리튬 이온 배터리용 리튬 니켈-망간-코발트 산화물 캐소드 분말 |
| KR20190008120A (ko) * | 2017-07-14 | 2019-01-23 | 유미코아 | 충전식 리튬 이온 배터리용 Ni계 캐소드 재료 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102790741B1 (ko) | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 | |
| JP2021517719A (ja) | 充電式リチウム二次電池用正極活物質としてのリチウム遷移金属複合酸化物 | |
| KR102734799B1 (ko) | 재충전 가능한 리튬 이온 배터리용 양극 활물질로서의 리튬 니켈 망간 코발트 복합 산화물 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20220203 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PA0201 | Request for examination | ||
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240805 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20250312 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20250331 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20250331 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration |