WO2014162761A1 - 塗布フィルム - Google Patents
塗布フィルム Download PDFInfo
- Publication number
- WO2014162761A1 WO2014162761A1 PCT/JP2014/051476 JP2014051476W WO2014162761A1 WO 2014162761 A1 WO2014162761 A1 WO 2014162761A1 JP 2014051476 W JP2014051476 W JP 2014051476W WO 2014162761 A1 WO2014162761 A1 WO 2014162761A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- film
- coating
- polyester
- layer
- weight
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/043—Improving the adhesiveness of the coatings per se, e.g. forming primers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/06—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material
- B32B27/08—Layered products comprising a layer of synthetic resin as the main or only constituent of a layer, which is next to another layer of the same or of a different material of synthetic resin
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B27/00—Layered products comprising a layer of synthetic resin
- B32B27/36—Layered products comprising a layer of synthetic resin comprising polyesters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/0427—Coating with only one layer of a composition containing a polymer binder
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J7/00—Chemical treatment or coating of shaped articles made of macromolecular substances
- C08J7/04—Coating
- C08J7/06—Coating with compositions not containing macromolecular substances
- C08J7/065—Low-molecular-weight organic substances, e.g. absorption of additives in the surface of the article
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D1/00—Coating compositions, e.g. paints, varnishes or lacquers, based on inorganic substances
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/10—Coating on the layer surface on synthetic resin layer or on natural or synthetic rubber layer
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2255/00—Coating on the layer surface
- B32B2255/26—Polymeric coating
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/20—Properties of the layers or laminate having particular electrical or magnetic properties, e.g. piezoelectric
- B32B2307/202—Conductive
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2307/00—Properties of the layers or laminate
- B32B2307/40—Properties of the layers or laminate having particular optical properties
- B32B2307/412—Transparent
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B32—LAYERED PRODUCTS
- B32B—LAYERED PRODUCTS, i.e. PRODUCTS BUILT-UP OF STRATA OF FLAT OR NON-FLAT, e.g. CELLULAR OR HONEYCOMB, FORM
- B32B2457/00—Electrical equipment
- B32B2457/20—Displays, e.g. liquid crystal displays, plasma displays
- B32B2457/208—Touch screens
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2367/00—Characterised by the use of polyesters obtained by reactions forming a carboxylic ester link in the main chain; Derivatives of such polymers
- C08J2367/02—Polyesters derived from dicarboxylic acids and dihydroxy compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2429/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal, or ketal radical; Hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Derivatives of such polymer
- C08J2429/02—Homopolymers or copolymers of unsaturated alcohols
- C08J2429/04—Polyvinyl alcohol; Partially hydrolysed homopolymers or copolymers of esters of unsaturated alcohols with saturated carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2433/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers
- C08J2433/04—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers esters
- C08J2433/14—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Derivatives of such polymers esters of esters containing halogen, nitrogen, sulfur, or oxygen atoms in addition to the carboxy oxygen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2461/00—Characterised by the use of condensation polymers of aldehydes or ketones; Derivatives of such polymers
Definitions
- the present invention relates to a coated film, and in particular, relates to a coated film having a polyester film as a base material and, for example, less oligomer precipitation even after being exposed to a high temperature.
- Polyester film is excellent in transparency, dimensional stability, mechanical properties, heat resistance, electrical properties, etc., and is used in various fields.
- a transparent conductive laminate there is one in which a polyester film is used as a base material and an ITO (indium tin oxide) film is formed by sputtering directly or via an anchor layer.
- ITO indium tin oxide
- Such a biaxially stretched polyester film is generally heat-processed.
- Patent Document 1 there are treatments such as leaving for 1 hour at 150 ° C. for low heat shrinkage (Patent Document 1) and performing heat treatment at 150 ° C. for crystallization of ITO (Patent Document 2).
- polyester film when exposed to such high temperature and long time treatment, oligomers (low molecular weight component of polyester, especially ester cyclic trimer) contained in the film are precipitated and crystallized on the film surface. As a result, deterioration of visibility due to whitening of the external appearance of the film, defects in post-processing, contamination of the process and members, and the like occur. Therefore, the characteristics of the transparent conductive laminate based on the polyester film cannot be said to be sufficiently satisfactory.
- oligomers low molecular weight component of polyester, especially ester cyclic trimer
- the curable resin layer is formed by thermosetting, requires high-temperature treatment for dissociation of the blocking agent of the isocyanate resin, and is in a situation where curling and sagging are likely to occur during processing. Care must be taken in handling.
- the present invention has been made in view of the above circumstances, and a problem to be solved is to provide an optical coating film having excellent optical characteristics and visibility even after being processed as a product for an optical member. Specifically, for example, a process under harsh conditions such as a long-time heat treatment at 150 ° C., a sputtering process under a high tension condition, and a durability test under a high-temperature and high-humidity atmosphere. It is an object of the present invention to provide an optical coating film that is excellent in optical characteristics and visibility even after being processed, as an increase in film haze is suppressed to a small level and is processed as a product for optical members.
- the gist of the present invention is that a monolayer polyester film having an ester cyclic trimer content of 0.7% by weight or less or a multilayer polyester film having a polyester surface layer having an ester cyclic trimer content of 0.7% by weight or less.
- the coating film has a coating layer formed from a coating solution containing a polyvalent aldehyde compound on at least one side of the coating film.
- the combination of the polyester layer having a low ester cyclic trimer content and the formation of a specific coating layer results in very little precipitation of the ester cyclic trimer.
- the effect becomes.
- the oligomer precipitation from the surface is suppressed even after high-temperature and long-time treatment, it is possible to obtain a product with an excellent appearance that does not increase haze or generate foreign matter. Value is high.
- the base film of the coated film of the present invention is made of polyester.
- Polyesters include dicarboxylic acids such as terephthalic acid, isophthalic acid, 2,6-naphthalenedicarboxylic acid, adipic acid, sebacic acid, 4,4'-diphenyldicarboxylic acid, 1,4-cyclohexyldicarboxylic acid or esters thereof and ethylene glycol.
- Diethylene glycol, triethylene glycol, propylene glycol, 1,4-butanediol, neopentyl glycol, and glycols such as 1,4-cyclohexanedimethanol are produced by melt polycondensation. Polyesters composed of these acid components and glycol components can be produced by arbitrarily using a commonly used method.
- a transesterification reaction is first carried out between a lower alkyl ester of an aromatic dicarboxylic acid and a glycol, or an aromatic dicarboxylic acid and a glycol are directly esterified to form a substantially aromatic dicarboxylic acid.
- a method is employed in which a bisglycol ester or a low polymer thereof is formed and then subjected to polycondensation by heating under reduced pressure.
- an aliphatic dicarboxylic acid may be copolymerized.
- polyester examples include polyethylene terephthalate, polyethylene-2,6-naphthalate, poly-1,4-cyclohexanedimethylene terephthalate, and the like.
- polyester obtained by copolymerizing the above acid component and glycol component. And may contain other components and additives as necessary.
- the polymerization catalyst for polyester is not particularly limited, and conventionally known compounds can be used.
- examples thereof include antimony compounds, titanium compounds, germanium compounds, manganese compounds, aluminum compounds, magnesium compounds, calcium compounds and the like.
- antimony compounds have the advantage of being inexpensive and having high catalytic activity. Titanium compounds and germanium compounds are preferable because they have high catalytic activity and can be polymerized in a small amount, and since the amount of metal remaining in the film is small, the brightness of the film is increased. Furthermore, since a germanium compound is expensive, it is more preferable to use a titanium compound.
- the single layer polyester film having an ester cyclic trimer content of 0.7% by weight or less or the ester cyclic trimer content is 0.
- a multilayer polyester film having a polyester surface layer of 7% by weight or less is an essential requirement.
- the content of the ester cyclic trimer contained in the polyester film is about 1% by weight in a general production method.
- the effect of preventing the precipitation of the ester cyclic trimer on the film surface is particularly high by specifying the content of the ester cyclic trimer as 0.7% by weight or less in the above-described manner. Is done.
- the content of the ester cyclic trimer exceeds 0.7% by weight, the film haze deteriorates when used in a high temperature treatment for a long time or a processing step under harsh conditions. There arises a problem that the ester cyclic trimer adheres to and accumulates on the transport roll that comes into contact with the roller.
- the ester cyclic trimer content in the present invention is preferably 0.6% by weight or less, particularly preferably 0.5% by weight or less.
- Various known methods can be used as a method for producing a polyester having a low ester cyclic trimer content. Examples thereof include a method of solid-phase polymerization after polyester production. And ester cyclic trimer content can be reduced by lengthening solid phase polymerization time.
- polyester having a reduced ester cyclic trimer content is expensive, it is advantageous to adjust the ester cyclic trimer content to 0.7% by weight or less by polymer blending.
- the blend amount is usually 70% by weight or more, preferably 80% by weight or more.
- the lower limit of the ester cyclic trimer content of the polyester used in the polymer blend is usually 0.1% by weight, preferably 0.2% by weight, from the viewpoint of cost.
- the polyester film may have a single layer structure or a multilayer structure, but in the case of a multilayer structure, a polyester having an ester cyclic trimer content of 0.7% by weight or less may be used only for the surface layer. Therefore, it is advantageous in terms of cost, and the surface layer and the inner layer, or both the surface layer and each layer can be made of different polyesters depending on the purpose.
- the polyester film has a multilayer structure of two types and three layers, and the surface of the multilayer polyester film is designed using a polyester raw material with a small content of the ester cyclic trimer. Can be suppressed.
- the thickness of the 0.7% by weight or less polyester layer is preferably 1.5 ⁇ m or more, more preferably 2.0 ⁇ m or more, and particularly preferably 2.5 ⁇ m or more.
- the heat treatment is performed for a long time at 150 ° C., the sputtering process under high tension, or the high temperature and high humidity atmosphere.
- film haze increases significantly and may not be suitable for optical members in terms of optical properties and visibility after processing as a product .
- the polyester film can contain particles for the purpose of ensuring the film runnability and preventing scratches.
- particles include inorganic particles such as silica, calcium carbonate, magnesium carbonate, calcium phosphate, kaolin, talc, aluminum oxide, titanium oxide, alumina, barium sulfate, calcium fluoride, lithium fluoride, zeolite, and molybdenum sulfide.
- organic particles such as crosslinked polymer particles and calcium oxalate, and precipitated particles obtained by precipitating and finely dispersing a part of a metal compound such as a catalyst during the polyester production process can also be used.
- the shape of the particles to be used is not particularly limited, and any of a spherical shape, a block shape, a rod shape, a flat shape, and the like may be used. Moreover, there is no restriction
- the particle size and content of the particles to be used are selected according to the use and purpose of the film.
- the average particle size (d50) is usually 3 ⁇ m or less, preferably 0.02 ⁇ m to 2.8 ⁇ m, more preferably 0.8.
- the range is from 03 ⁇ m to 2.5 ⁇ m. If the average particle size exceeds 3 ⁇ m, the surface roughness of the film becomes too rough, or the particles easily fall off from the film surface.
- the particle content is usually 3% by weight or less, preferably 0.0003 to 1.0% by weight, more preferably 0.0005 to 0.5% by weight, based on the polyester layer containing the particles.
- the particle content is usually 3% by weight or less, preferably 0.0003 to 1.0% by weight, more preferably 0.0005 to 0.5% by weight, based on the polyester layer containing the particles.
- the transparency of the film becomes high and a good film is obtained, but the slipperiness may be insufficient. In some cases, it is necessary to improve the performance. Further, when the particle content exceeds 3% by weight, the transparency of the film may be insufficient.
- the method for adding particles to the polyester layer is not particularly limited, and a conventionally known method can be adopted.
- it can be added at any stage for producing the polyester constituting each layer, but it is preferably added after completion of esterification or transesterification.
- the thickness of the polyester film is not particularly limited as long as it can be formed into a film, but is usually 10 to 300 ⁇ m, preferably 15 to 250 ⁇ m, more preferably 20 to 200 ⁇ m.
- the haze of the coating film of this invention is 10% or less. More preferably, it is 5% or less, More preferably, it is 4% or less. When the haze of the coated film exceeds 10%, it may be difficult to use in appearance in optical film applications.
- a generally known film forming method can be adopted, and there is no particular limitation.
- a sheet obtained by melt extrusion is stretched 2 to 6 times at 70 to 145 ° C. by a roll stretching method to obtain a uniaxially stretched polyester film, and then the previous stretching direction in the tenter A film can be obtained by stretching 2 to 6 times at 80 to 160 ° C. in the perpendicular direction, and further performing heat treatment at 150 to 250 ° C. for 1 to 600 seconds. Further, at this time, it is preferable to perform relaxation within 20% in the longitudinal direction and / or the transverse direction in the heat treatment zone and / or the cooling zone at the heat treatment outlet.
- the coating layer can be provided by either a so-called off-line coating in which a coating layer is provided later on the formed film or a so-called in-line coating in which a coating layer is provided during film formation. It is preferably provided by in-line coating, particularly by a coating stretching method in which stretching is performed after coating.
- In-line coating is a method of coating in the process of manufacturing a polyester film, and specifically, a method of coating at an arbitrary stage from melt-extrusion of polyester to heat-fixing and winding after stretching.
- it is either a substantially amorphous unstretched sheet obtained by melting and quenching, a stretched uniaxially stretched film, a biaxially stretched film before heat setting, or a film after winding and before winding. Coating.
- sequential biaxial stretching a method of stretching in the transverse direction after coating a uniaxially stretched film stretched in the longitudinal direction (longitudinal direction) is particularly excellent.
- the polyester film before biaxial stretching is first covered with a resin layer constituting the coating layer, and then the base film and the coating layer are firmly adhered by stretching the film and the coating layer simultaneously. It will be.
- the biaxial stretching of the polyester film is that the film is constrained in the longitudinal / lateral direction by gripping the end of the film with a tenter clip etc. High temperature can be applied while maintaining flatness. Therefore, since the heat treatment performed after coating can be performed at a high temperature that cannot be achieved by other methods, the film forming property of the coating layer is improved, and the coating layer and the polyester film are firmly adhered.
- the uniformity of the coating layer, the improvement of the film forming property, and the adhesion between the coating layer and the film often produce preferable characteristics.
- the coating solution to be used is preferably an aqueous solution or an aqueous dispersion for safety reasons in terms of handling, working environment, but water is the main medium and does not exceed the gist of the present invention. If so, an organic solvent may be contained.
- the coated film of the present invention requires a coating layer formed from a coating solution containing a polyvalent aldehyde compound as an essential requirement.
- the coating solution may contain other components.
- the polyvalent aldehyde compound is a compound having two or more aldehyde groups or a functional group derived from an aldehyde group in one molecule.
- aldehyde groups or a functional group derived from an aldehyde group in one molecule.
- the polyvalent aldehyde compound may be unstable and is preferably used in a form in which an aldehyde group is reacted (protected).
- a form reacted with a hydroxyl group-containing compound, particularly a polyhydric alcohol compound is preferable, and among them, a type that forms a cyclic structure when reacted with an aldehyde group is more preferable.
- polyhydric alcohol compound examples include polyhydric alcohols such as ethylene glycol, 1,3-propanediol, and glycerin, and saccharides such as glucose and galactose. Among these, saccharides are preferable in consideration of applicability.
- polyhydric aldehyde compound an aliphatic polyhydric aldehyde compound or a derivative thereof is preferable in consideration of the strength of the coating layer and the coating appearance, and among them, glyoxal that increases the amount of aldehyde functional group is more preferable, in particular, the molecular weight. .
- a preferable form of the polyhydric aldehyde compound is a type in which a polyhydric alcohol compound is reacted with glyoxal, and among them, a compound in which a saccharide is reacted with glyoxal.
- an ammonium group-containing polymer, polyvinyl alcohol and a crosslinking agent should be used in combination from the viewpoint of preventing precipitation of the ester cyclic trimer on the film surface by heating and improving the durability and coating properties of the coating layer. Is preferred. Among these, it is preferable to use an ammonium group-containing polymer in combination in order to further prevent precipitation of an ester cyclic trimer.
- a coating layer formed from a coating solution containing a polyvalent aldehyde compound and an ammonium group-containing polymer, polyvinyl alcohol, and at least one compound selected from the group of crosslinking agents is used. It is a coating film which has.
- the ammonium group-containing polymer refers to a polymer compound having an ammonium group in the molecule, and for example, a polymer containing a monomer having an ammonium group and an unsaturated double bond as a component can be used.
- Such a polymer include a polymer having a constituent represented by the following formula (1) or the following formula (2) as a repeating unit. These homopolymers and copolymers, and other plural components may be copolymerized.
- R 2 is —O— or —NH—
- R 3 is an alkylene group, or other structure capable of forming the structure of formula (2)
- R 1 , R 4 , R 5 , R 6 Are each a hydrogen atom, an alkyl group, a phenyl group or the like, and these alkyl group and phenyl group may be substituted with the following groups.
- Substitutable groups are, for example, hydroxy groups, amide groups, ester groups, alkoxy groups, phenoxy groups, naphthoxy groups, thioalkoxy groups, thiophenoxy groups, cycloalkyl groups, trialkylammonium alkyl groups, cyano groups, and halogens. .
- R ⁇ 1 >, R ⁇ 2 > is respectively independently a hydrogen atom, an alkyl group, a phenyl group, etc.
- alkyl groups and a phenyl group may be substituted by the group shown below.
- Substitutable groups are, for example, hydroxyl group, amide group, ester group, alkoxy group, phenoxy group, naphthoxy group, thioalkoxy group, thiophenoxy group, cycloalkyl group, trialkylammonium alkyl group, cyano group, halogen .
- X ⁇ in the above formulas (1) and (2) can be appropriately selected within a range not impairing the gist of the present invention.
- examples include halogen ions, sulfonates, phosphates, nitrates, alkyl sulfonates, and carboxylates.
- a polymer having a component represented by Formula 1 is preferable because of excellent transparency of the resulting coating layer.
- the coating stretching method may be inferior in heat resistance, and when used in the coating stretching method, X ⁇ is preferably not a halogen.
- a compound represented by the formula 2 or other ammonium base in the polymer skeleton is preferable because of its excellent heat resistance.
- the polymer in which the component represented by the formulas 1 and 2 and the polyethylene glycol-containing (meth) acrylate are copolymerized has a flexible structure, and has a uniform coating layer during coating and stretching. Is preferable.
- a coating layer having excellent uniformity can be obtained by coating a polyethylene glycol-containing (meth) acrylate polymer in a coating solution.
- polyethylene glycol-containing (meth) acrylates include polyethylene glycol monoacrylate, polypropylene glycol monoacrylate, polyethylene glycol diacrylate (the polymerization degree of polyethylene glycol units is preferably in the range of 4 to 14), and polypropylene glycol diacrylate.
- polytetramethylene glycol diacrylate poly (ethylene glycol-tetramethylene glycol) diacrylate, poly (propylene glycol-tetramethylene glycol) diacrylate, polyethylene glycol-polypropylene glycol-polyethylene glycol diacrylate, polypropylene glycol-polybutylene glycol Monomethacrylate, methoxypolyethylene glycol Methacrylate, methoxy polyethylene glycol monoacrylate, octoxy polyethylene glycol-polypropylene glycol monomethacrylate, octoxy polyethylene glycol-polypropylene glycol monoacrylate, lauroxy polyethylene glycol monomethacrylate, lauroxy polyethylene glycol monoacrylate, stearoxy polyethylene glycol monomethacrylate, Examples thereof include polymers starting from stearoxy polyethylene glycol monoacrylate, allyloxy polyethylene glycol monomethacrylate, allyloxy polyethylene glycol monoacrylate and the like.
- the number average molecular weight of the ammonium group-containing polymer is 1,000 to 500,000, preferably 2,000 to 350,000, and more preferably 5,000 to 200,000.
- the molecular weight is less than 1000, the strength of the coating film may be weak or the heat resistance stability may be poor.
- the molecular weight exceeds 500,000, the viscosity of the coating solution increases, and the handleability and applicability may deteriorate.
- polyvinyl alcohol conventionally known polyvinyl alcohol can be used, including modified compounds partially acetalized or brachiralized.
- the degree of polymerization of polyvinyl alcohol is not particularly limited, but is usually 100 or more, preferably in the range of 300 to 40,000. When the degree of polymerization is less than 100, the water resistance of the coating layer may decrease.
- the degree of saponification of polyvinyl alcohol is not particularly limited, but is usually 70 mol% or more, preferably in the range of 70 to 99.9 mol%, more preferably 80 to 97 mol%, particularly preferably 86 to 95 mol%. Some polyvinyl saponification products are practically used.
- crosslinking agent various known crosslinking agents can be used, and examples thereof include melamine compounds, oxazoline compounds, epoxy compounds, isocyanate compounds, carbodiimide compounds, silane coupling compounds, and the like.
- an oxazoline compound, an epoxy compound, an isocyanate compound, and a carbodiimide compound are preferably used from the viewpoint of improving durability adhesion.
- precipitation prevention of the ester cyclic trimer may be inferior.
- a melamine compound is used suitably from a viewpoint of prevention of precipitation of the ester cyclic trimer on the film surface by heating and improvement of durability and applicability of the coating layer.
- an alkylolated melamine derivative a compound obtained by reacting an alcohol with an alkylolated melamine derivative or partially etherified, or a mixture thereof can be used.
- alcohol used for etherification methyl alcohol, ethyl alcohol, isopropyl alcohol, n-butanol, isobutanol and the like are preferably used.
- a melamine compound either a monomer or a multimer more than a dimer may be sufficient, or a mixture thereof may be used.
- a product obtained by co-condensing urea or the like with a part of melamine can be used, and a catalyst can be used to increase the reactivity of the melamine compound.
- the oxazoline compound is particularly preferably a polymer containing an oxazoline group, and can be prepared by polymerization of an addition polymerizable oxazoline group-containing monomer alone or with another monomer.
- Addition-polymerizable oxazoline group-containing monomers include 2-vinyl-2-oxazoline, 2-vinyl-4-methyl-2-oxazoline, 2-vinyl-5-methyl-2-oxazoline, 2-isopropenyl-2-oxazoline, Examples thereof include 2-isopropenyl-4-methyl-2-oxazoline, 2-isopropenyl-5-ethyl-2-oxazoline, and the like, and one or a mixture of two or more thereof can be used.
- 2-isopropenyl-2-oxazoline is preferred because it is easily available industrially.
- the other monomer is not particularly limited as long as it is a monomer copolymerizable with an addition polymerizable oxazoline group-containing monomer.
- alkyl (meth) acrylate (alkyl groups include methyl, ethyl, n-propyl, isopropyl, (Meth) acrylic acid esters such as n-butyl group, isobutyl group, t-butyl group, 2-ethylhexyl group, cyclohexyl group); acrylic acid, methacrylic acid, itaconic acid, maleic acid, fumaric acid, crotonic acid, styrene Unsaturated carboxylic acids such as sulfonic acid and its salts (sodium salt, potassium salt, ammonium salt, tertiary amine salt, etc.); Unsaturated nitriles such as acrylonitrile, methacrylonitrile; (meth) acrylamide, N-alkyl ( (Meth) acrylamide, N, N-dialkyl (meth) acrylamide, Examples of the alkyl group include unsaturated amides such as methyl,
- the amount of the oxazoline group of the oxazoline compound contained in the coating layer is usually in the range of 0.5 to 10 mmol / g, preferably 3 to 9 mmol / g, more preferably 5 to 8 mmol / g. By using it in the above range, the durability of the coating film is improved.
- epoxy compound examples include condensates of epichlorohydrin and ethylene glycol, polyethylene glycol, glycerin, polyglycerin, bisphenol A and other hydroxyl groups and amino groups, such as polyepoxy compounds, diepoxy compounds, monoepoxy compounds, Examples include glycidylamine compounds.
- polyepoxy compound examples include sorbitol polyglycidyl ether, polyglycerol polyglycidyl ether, pentaerythritol polyglycidyl ether, diglycerol polyglycidyl ether, triglycidyl tris (2-hydroxyethyl) isocyanate, glycerol polyglycidyl ether, trimethylolpropane.
- polyglycidyl ether and diepoxy compound examples include neopentyl glycol diglycidyl ether, 1,6-hexanediol diglycidyl ether, resorcin diglycidyl ether, ethylene glycol diglycidyl ether, polyethylene glycol diglycidyl ether, and propylene glycol diglycidyl ether.
- Polypropylene glycol diglycidyl ether polypropylene glycol diglycidyl ether, poly Examples of tetramethylene glycol diglycidyl ether and monoepoxy compounds include allyl glycidyl ether, 2-ethylhexyl glycidyl ether, phenyl glycidyl ether, and glycidyl amine compounds such as N, N, N ′, N′-tetraglycidyl-m-xylyl. Examples include range amine and 1,3-bis (N, N-diglycidylamino) cyclohexane.
- the isocyanate compound is a compound having an isocyanate derivative structure typified by isocyanate or blocked isocyanate.
- isocyanates include aromatic isocyanates such as tolylene diisocyanate, xylylene diisocyanate, methylene diphenyl diisocyanate, phenylene diisocyanate, and naphthalene diisocyanate, and aromatic rings such as ⁇ , ⁇ , ⁇ ′, ⁇ ′-tetramethylxylylene diisocyanate.
- Aliphatic isocyanates such as aliphatic isocyanate, methylene diisocyanate, propylene diisocyanate, lysine diisocyanate, trimethylhexamethylene diisocyanate, hexamethylene diisocyanate, cyclohexane diisocyanate, methylcyclohexane diisocyanate, isophorone diisocyanate, methylene bis (4-cyclohexyl isocyanate), isopropylidene dicyclohexyl diisocyanate
- Alicyclic isocyanates such as bets are exemplified.
- polymers and derivatives such as burettes, isocyanurates, uretdiones, and carbodiimide modified products of these isocyanates are also included. These may be used alone or in combination.
- isocyanates aliphatic isocyanates or alicyclic isocyanates are more preferable than aromatic isocyanates in order to avoid yellowing due to ultraviolet rays.
- the blocking agent When used in the state of blocked isocyanate, the blocking agent includes, for example, bisulfites, phenolic compounds such as phenol, cresol, and ethylphenol, and alcohols such as propylene glycol monomethyl ether, ethylene glycol, benzyl alcohol, methanol, and ethanol.
- Compounds, active methylene compounds such as methyl isobutanoyl acetate, dimethyl malonate, diethyl malonate, methyl acetoacetate, ethyl acetoacetate, acetylacetone, mercaptan compounds such as butyl mercaptan, dodecyl mercaptan, ⁇ -caprolactam, ⁇ -valerolactam, etc.
- Lactam compounds amine compounds such as diphenylaniline, aniline, ethyleneimine, acetanilide, acetic acid amide acid amide compounds, Examples include oxime compounds such as rumaldehyde, acetoaldoxime, acetone oxime, methyl ethyl ketone oxime, and cyclohexanone oxime, and these may be used alone or in combination of two or more.
- the isocyanate compound may be used alone, or may be used as a mixture or a combination with various polymers. In the sense of improving the dispersibility and crosslinkability of the isocyanate compound, it is preferable to use a mixture or a bond with a polyester resin or a urethane resin.
- the carbodiimide compound is more preferably a polycarbodiimide compound having two or more in the molecule for better adhesion and the like.
- the carbodiimide compound can be synthesized by a conventionally known technique, and generally a condensation reaction of a diisocyanate compound is used.
- the diisocyanate compound is not particularly limited, and any of aromatic and aliphatic compounds can be used.
- tolylene diisocyanate, xylene diisocyanate, diphenylmethane diisocyanate, phenylene diisocyanate, naphthalene diisocyanate, hexamethylene diisocyanate examples thereof include trimethylhexamethylene diisocyanate, cyclohexane diisocyanate, methylcyclohexane diisocyanate, isophorone diisocyanate, dicyclohexyl diisocyanate, and dicyclohexylmethane diisocyanate.
- the content of the carbodiimide group contained in the carbodiimide-based compound is a carbodiimide equivalent (weight of the carbodiimide compound to give 1 mol of carbodiimide group [g]), and is usually 100 to 1000, preferably 250 to 800, more preferably 300. It is in the range of ⁇ 700. By using it in the above range, the durability of the coating film is improved.
- cross-linking agents are used in a design that improves the performance of the coating layer by reacting in the drying process or film forming process. It can be inferred that unreacted products of these crosslinking agents, compounds after the reaction, or mixtures thereof exist in the finished coating layer.
- a component for promoting crosslinking for example, a crosslinking catalyst can be used in combination.
- the coating layer it is possible to use a polymer in combination for the purpose of improving the coating appearance and improving the adhesion when various surface functional layers are formed in the coating layer.
- polymer examples include polyester resin, acrylic resin, urethane resin, conductive polymer, polyalkylene glycol, polyalkyleneimine, methylcellulose, oxycellulose, and starches.
- polyester resin acrylic resin, urethane resin, conductive polymer, polyalkylene glycol, polyalkyleneimine, methylcellulose, oxycellulose, and starches.
- particles can be used in combination for the purpose of blocking and improving slipperiness in forming the coating layer.
- the average particle diameter is preferably 1.0 ⁇ m or less, more preferably 0.5 ⁇ m or less, and particularly preferably 0.2 ⁇ m or less from the viewpoint of transparency of the film.
- the lower limit is preferably 0.005 ⁇ m or more, more preferably 0.01 ⁇ m or more, in order to improve the slipperiness more effectively.
- Specific examples of the particles include silica, alumina, kaolin, calcium carbonate, and organic particles. Among these, silica is preferable from the viewpoint of transparency.
- an antifoaming agent a coating property improver, a thickener, an organic lubricant, an antistatic agent, an ultraviolet absorber, and an antioxidant are formed as necessary for forming the coating layer. It is also possible to use a foaming agent, a dye, a pigment and the like in combination.
- the ratio of the polyhydric aldehyde compound is usually in the range of 1 to 50% by weight, preferably 2 to 30% by weight, based on the total nonvolatile components in the coating solution.
- the ratio is out of the above range, precipitation of the ester cyclic trimer after heating cannot be effectively suppressed or the appearance of coating may be deteriorated.
- the proportion of the ammonium group-containing polymer is usually in the range of 5 to 80% by weight, preferably 10 to 70% by weight, as a proportion of all nonvolatile components in the coating solution.
- the ratio is out of the above range, precipitation of the ester cyclic trimer after heating cannot be effectively suppressed or the uniformity of the obtained coating film may be deteriorated.
- the proportion of polyvinyl alcohol is usually in the range of 5 to 80% by weight, preferably 10 to 35% by weight, as a proportion of all nonvolatile components in the coating solution.
- the ratio is out of the above range, precipitation of the ester cyclic trimer after heating cannot be effectively suppressed or the appearance of coating may be deteriorated.
- the ratio of the crosslinking agent is usually in the range of 5 to 80% by weight, preferably 10 to 50% by weight as the ratio to the total nonvolatile components in the coating solution.
- the ratio is out of the above range, precipitation of the ester cyclic trimer after heating cannot be effectively suppressed or the appearance of coating may be deteriorated.
- the thickness of the coating layer is usually in the range of 0.003 ⁇ m to 1 ⁇ m, preferably 0.005 ⁇ m to 0.5 ⁇ m, more preferably 0.00 as the thickness of the coating layer on the finally obtained film. It is in the range of 01 ⁇ m to 0.2 ⁇ m.
- the thickness is less than 0.003 ⁇ m, the amount of ester cyclic trimer precipitated from the film may not be sufficiently reduced.
- it is thicker than 1 ⁇ m, problems such as deterioration of the appearance of the coating layer and easy blocking may occur.
- Examples of methods for applying a coating solution to a polyester film include air doctor coating, blade coating, rod coating, bar coating, knife coating, squeeze coating, impregnation coating, reverse roll coating, transfer roll coating, gravure coating, and kiss roll coating.
- Conventional coating methods such as cast coating, spray coating, curtain coating, calendar coating, and extrusion coating can be used.
- the film may be subjected to chemical treatment, corona discharge treatment, plasma treatment or the like before coating.
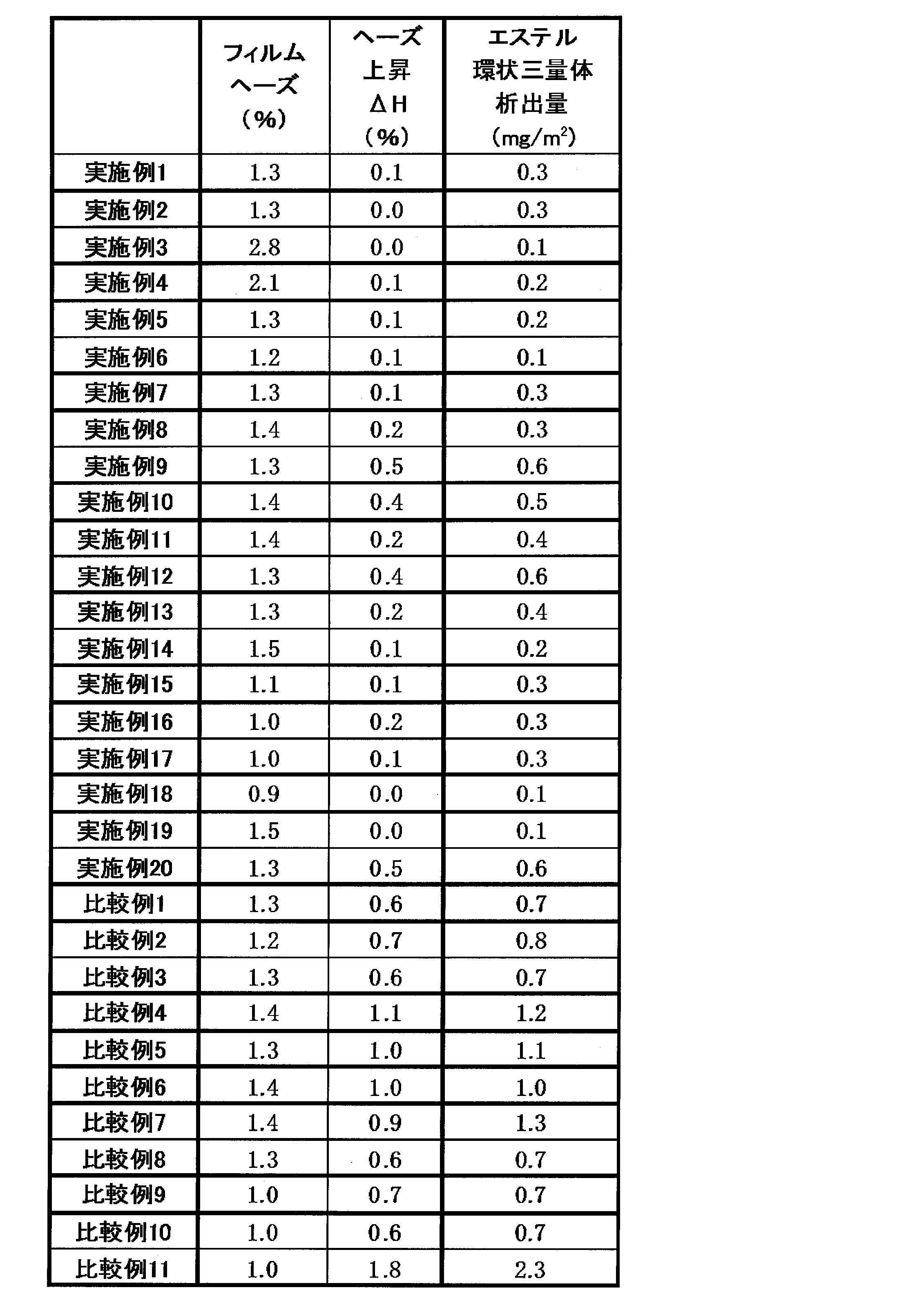
- the film haze change amount ( ⁇ H) in the heat treatment is preferably 0.5% or less, more preferably 0.4% or less, and further preferably, in order to cope with high transparency. Is 0.3% or less.
- ⁇ H exceeds 0.5%, the visibility decreases as the film haze increases due to precipitation of the ester cyclic trimer, and is not suitable for applications that require high visibility, such as for touch panels. It may be appropriate.
- the amount of the ester cyclic trimer extracted from the film surface by dimethylformamide by heat treatment is preferably 0. .6mg / m 2 or less, more preferably 0.4 mg / m 2 or less, more preferably 0.3 mg / m 2 or less. If it exceeds 0.6 mg / m 2 , the amount of ester cyclic trimer deposited increases in the subsequent process, for example, with heat treatment for a long time in a high-temperature atmosphere such as 180 ° C. for 90 minutes, and the transparency of the film There is a concern that the performance may deteriorate or contamination of the process.
- Preparation of the standard sample was performed by accurately weighing the pre-sorted ester cyclic trimer and dissolving in accurately weighed DMF.
- Coating layer thickness The film was fixed with an embedding resin, the cross section was cut with a microtome, and the sample was prepared by staining with 2% osmic acid at 60 ° C. for 2 hours. The obtained sample was observed with a transmission electron microscope (JEM2010 manufactured by JEOL Ltd.), and the thickness of the coating layer was measured. A total of 15 points on the film are measured, and an average of 9 points excluding 3 points from the larger value and 3 points from the smaller value is defined as the coating layer thickness.
- the amount of ester cyclic trimer precipitated on the film surface (mg / m 2 ) was used.
- the amount of ester cyclic trimer in DMF was calculated according to the absolute calibration curve method described in (3) Method for measuring amount of oligomer contained in polyester raw material.
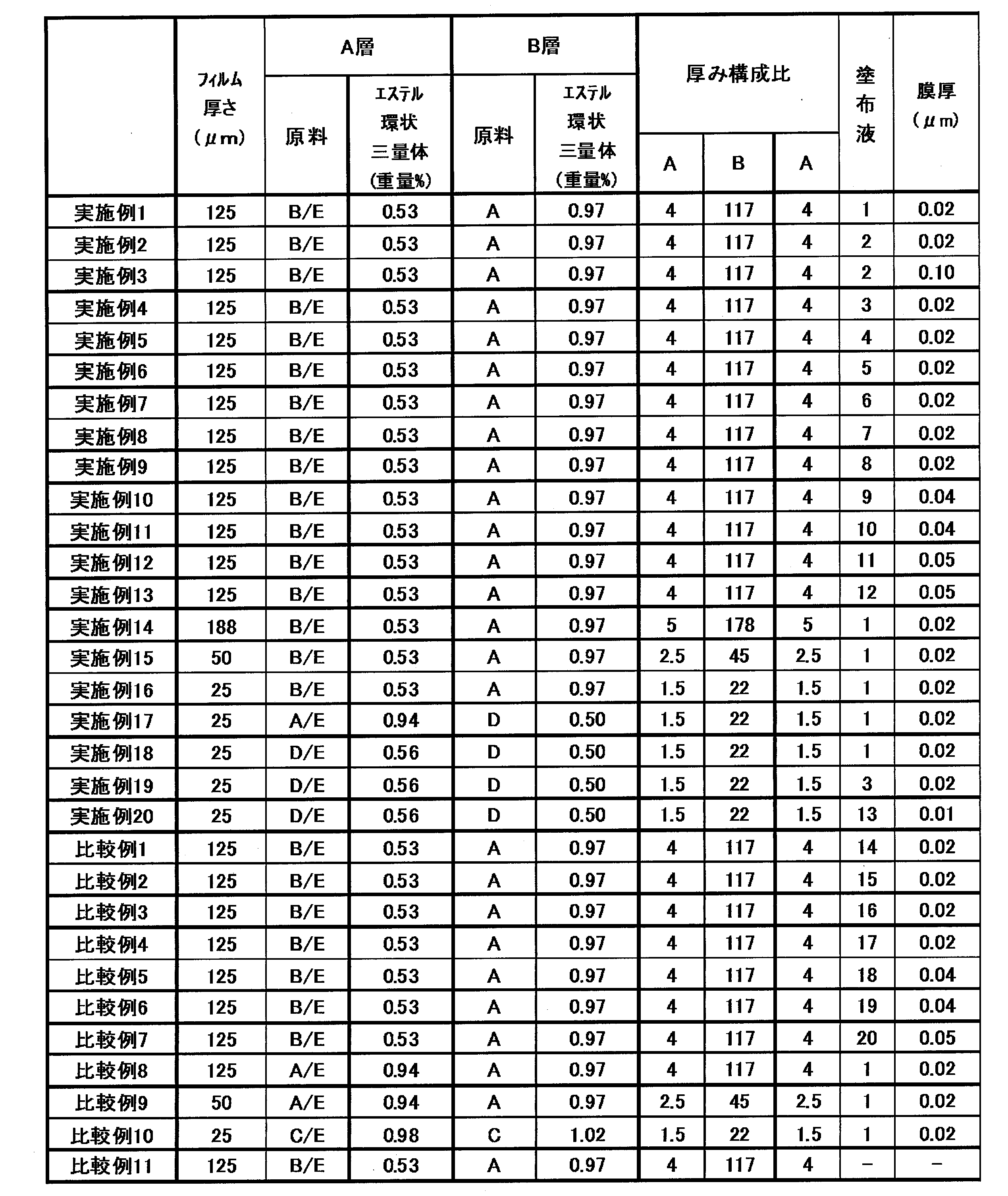
- the polyester used in the examples and comparative examples was prepared as follows. ⁇ Method for producing polyester (A)> 100 parts by weight of dimethyl terephthalate and 60 parts by weight of ethylene glycol are used as starting materials, 0.09 parts by weight of magnesium acetate tetrahydrate as a catalyst is taken in the reactor, the reaction start temperature is set to 150 ° C., and the methanol is gradually distilled off. The reaction temperature was raised to 230 ° C. after 3 hours. After 4 hours, the transesterification reaction was substantially terminated. After adding 0.04 part of ethyl acid phosphate to this reaction mixture, 0.04 part of antimony trioxide was added, and a polycondensation reaction was carried out for 4 hours.
- the obtained polyester (A) had an intrinsic viscosity of 0.63 and an ester cyclic trimer content of 0.97% by weight.
- polyester (A) is pre-crystallized at 160 ° C. in advance, and then solid-phase polymerized in a nitrogen atmosphere at a temperature of 220 ° C., and has an intrinsic viscosity of 0.75 and an ester cyclic trimer content of 0.46% by weight. Polyester (B) was obtained.
- Polyester (C) was pre-crystallized at 160 ° C. in advance, and then solid-phase polymerized in a nitrogen atmosphere at a temperature of 210 ° C., with an intrinsic viscosity of 0.72 and an ester cyclic trimer content of 0.50% by weight. Polyester (D) was obtained.
- polyester (E) ⁇ Method for producing polyester (E)>
- a polyester (C) was obtained using the same method as the production method of polyester 1 except that the polycondensation reaction was stopped at a time corresponding to an intrinsic viscosity of 0.65.
- the obtained polyester (C) had an intrinsic viscosity of 0.65 and an ester cyclic trimer content of 0.82% by weight.
- A1 Multivalent aldehyde compound obtained by reacting glyoxal with anhydrous glucose
- B1 2- (trimethylamino) ethyl methacrylate / ethyl methacrylate / butyl methacrylate / polyethylene glycol-containing monoacrylate having a counter ion of methylsulfonate
- the copolymer having a weight ratio of 75/12/15/30 has a number average molecular weight of 150,000.
- B2 2-hydroxy3-methacryloxypropyltrimethylammonium salt polymer in which the counter ion is methylsulfonate.
- C4 polyglycerol polyglycidyl ether
- F1 silica particles having an average particle size of 0.07 ⁇ m
- F2 alumina-modified silica particles having an average particle size of 0.02 ⁇ m
- the film was stretched 3.4 times in the longitudinal direction using the difference in peripheral speed of the roll while passing through a heated roll group at 85 ° C., and then the aqueous coating solution shown in Table 1 below was formed on one side of the longitudinally stretched film. 1 was applied, led to a tenter stretching machine, stretched 4.0 times in the transverse direction at 100 ° C., heat treated at 230 ° C., and then subjected to a relaxation treatment of 2% in the transverse direction to obtain a film thickness of the coating layer.
- the film haze increase value ( ⁇ H) by heat treatment of the obtained polyester film was small, and the amount of ester cyclic trimer deposited was small and good.
- the properties of this film are shown in Table 3 below.
- Example 1 In Example 1, it manufactured similarly to Example 1 except having changed the coating agent composition into the coating agent composition shown in Table 1, and obtained the polyester film.
- the obtained polyester film had the characteristics shown in Table 3, the film haze increase value ( ⁇ H) by heat treatment was small, and the precipitation amount of the ester cyclic trimer was small, which was good.
- the film was stretched 3.4 times in the longitudinal direction using the difference in peripheral speed of the roll while passing through a heated roll group at 85 ° C., and then the aqueous coating solution shown in Table 1 below was formed on one side of the longitudinally stretched film. 1 was applied, led to a tenter stretching machine, stretched 4.0 times in the transverse direction at 100 ° C., heat treated at 230 ° C., and then subjected to a relaxation treatment of 2% in the transverse direction to obtain a film thickness of the coating layer.
- a biaxially oriented polyethylene terephthalate film with a film thickness of 25 ⁇ m having a coating layer of 0.02 ⁇ m (after drying) was obtained.
- Example 18 Polyester (D) and (E) blended at a weight ratio of 80/20 as the surface layer and polyester (D) alone as the raw material for the intermediate layer were fed to the two extruders, respectively, at 285 ° C.
- the layer A is the outermost layer (surface layer)
- the layer B is the intermediate layer
- the film was coextruded so as to be .5, and solidified by cooling while being in close contact with a mirror-cooling drum having a surface temperature of 40 to 50 ° C. by using an electrostatic contact method, thereby preparing an unstretched polyethylene terephthalate film.
- the film was stretched 3.4 times in the longitudinal direction using the difference in peripheral speed of the roll while passing through a heated roll group at 85 ° C., and then the aqueous coating solution shown in Table 1 below was formed on one side of the longitudinally stretched film. 1 was applied, led to a tenter stretching machine, stretched 4.0 times in the transverse direction at 100 ° C., heat treated at 230 ° C., and then subjected to a relaxation treatment of 2% in the transverse direction to obtain a film thickness of the coating layer.
- a biaxially oriented polyethylene terephthalate film with a film thickness of 25 ⁇ m having a coating layer of 0.02 ⁇ m (after drying) was obtained.
- Example 19 In Example 18, it manufactured like Example 1 except having changed an application agent composition into the application agent composition shown in Table 1, and obtained the polyester film.
- Example 1 In Example 1, it manufactured similarly to Example 1 except having changed the coating agent composition into the coating agent composition shown in Table 1, and obtained the polyester film. When the obtained coated film was evaluated, as shown in Table 3, the film haze due to the heat treatment was greatly increased, and precipitation of the ester cyclic trimer was also large.
- Example 8 In Example 1, as a raw material for the A layer, a polyester film was produced in the same manner as in Example 1 except that polyesters (A) and (E) were changed to mixed raw materials mixed at a ratio of 80% and 20%, respectively. Got. When the obtained coated film was evaluated, as shown in Table 3, the film haze due to the heat treatment was greatly increased, and precipitation of the ester cyclic trimer was also large.
- the film was stretched 3.4 times in the longitudinal direction using the difference in peripheral speed of the roll while passing through a heated roll group at 85 ° C., and then the aqueous coating solution shown in Table 1 below was formed on one side of the longitudinally stretched film. 1 was applied, led to a tenter stretching machine, stretched 4.0 times in the transverse direction at 100 ° C., heat treated at 230 ° C., and then subjected to a relaxation treatment of 2% in the transverse direction to obtain a film thickness of the coating layer.
- a biaxially oriented polyethylene terephthalate film with a film thickness of 25 ⁇ m having a coating layer of 0.02 ⁇ m (after drying) was obtained.
- Comparative Example 11 In Example 1, it manufactured like Example 1 except not providing an application layer, and obtained the polyester film. When the obtained coated film was evaluated, the film haze due to heat treatment was greatly increased, the precipitation of ester cyclic trimer was also large, and there was concern about deterioration of visibility due to process contamination and whitening after heating. It was a thing.
- the coated film of the present invention requires the performance that the film is exposed to a high temperature atmosphere for a long time, and even after a severe heat treatment step, the increase in film haze is as small as possible and the precipitation of the ester cyclic trimer is small.
- a coating film in applications for example, it can be suitably used as a base material of a transparent conductive laminate.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Wood Science & Technology (AREA)
- Coating Of Shaped Articles Made Of Macromolecular Substances (AREA)
- Laminated Bodies (AREA)
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Paints Or Removers (AREA)
Abstract
Description
ポリエステル1gを精秤し、フェノール/テトラクロロエタン=50/50(重量比)の混合溶媒100mlを加えて溶解させ、30℃で測定した。
遠心沈降式粒度分布測定装置(株式会社島津製作所社製SA-CP3型)を使用して測定した等価球形分布における積算(重量基準)50%の値を平均粒径とした。
ポリエステル原料を約200mg秤量し、クロロホルム/HFIP(ヘキサフルオロ-2-イソプロパノル)の比率3:2の混合溶媒2mlに溶解させる。溶解後、クロロホルム20mlを追加した後、メタノール10mlを少しずつ加える。沈殿物を濾過により除去し、更に、沈殿物をクロロホルム/メタノールの比率2:1の混合溶媒で洗浄し、濾液・洗浄液を回収し、エバポレーターにより濃縮、その後、乾固させる。乾固物をDMF(ジメチルホルムアミド)25mlに溶解後、この溶液を液体クロマトグラフィー(株式会社島津製作所製「LC-7A」)に供給して、DMF中のエステル環状三量体量を求め、この値をクロロホルム/HFIP混合溶媒に溶解させたポリエステル原料量で割って、含有エステル環状三量体量(重量%)とする。DMF中のエステル環状三量体量は、標準試料ピーク面積と測定試料ピーク面積のピーク面積比より求めた(絶対検量線法)。
移動相A:アセトニトリル
移動相B:2%酢酸水溶液
カラム:三菱化学株式会社製「MCI GEL ODS 1HU」
カラム温度:40℃
流速:1ml/分
検出波長:254nm
前記(3)の方法で得られたポリエステル原料に含有される含有エステル環状三量体量を基に算出した。
包埋樹脂でフィルムを固定し断面をミクロトームで切断し、2%オスミウム酸で60℃、2時間染色して試料を調整した。得られた試料を、透過型電子顕微鏡(日本電子株式会社製JEM2010)で観察し、塗布層の厚みを測定した。フィルムの計15箇所を測定し、数値の大きい方から3点と、小さい方から3点を除いた9点の平均を塗布層厚みとする。
サンプルの測定面がむき出しとなる状態でケント紙と重ねて固定し、窒素雰囲気下で、180℃で90分間放置して熱処理を行う。
試料フィルムをJIS-K-7136に準じ、株式会社村上色彩技術研究所製ヘーズメーター「HM-150」により、フィルムヘーズを測定した。
先ず、試料フィルムの塗布層が設けられた面とは反対側の面に下記塗布剤組成からなる塗布剤を、硬化後の厚さが3μmになるように塗布し、80℃に設定した熱風乾燥式オーブンにて1分間乾燥させた。次いで、120W/cmのエネルギーの高圧水銀灯を使用し、照射距離100mmにて約7秒間照射し、110mJ/cm2で硬化を行って、フィルム上に活性エネルギー線硬化樹脂層を設けた積層フィルムを得た。
(塗布剤組成)
紫外線硬化型ウレタンアクリレート樹脂(日本合成化学工業株式会社製「紫光7600B」)と、光重合開始剤(チバスペシャルティケミカルズ株式会社製「Irgacure651」)を、重量比で100/5で混合、メチルエチルケトンで濃度30重量%に希釈したものを使用した。
得られた試料のヘーズを前記(7)項の方法で測定した(ヘーズ1)。
次いで、試料の活性エネルギー線硬化樹脂層と反対の面を測定面として、前記(6)項の方法で加熱した後、(7)の方法でヘーズを測定した(ヘーズ2)。
ΔH=(ヘーズ2)-(ヘーズ1)
ΔHが低いほど、高温処理によるオリゴマーの析出が少ないことを示し、良好である。
ポリエステルフィルムを空気中、180℃で90分間加熱する。その後、熱処理をした当該フィルムを上部が開いている縦横10cm、高さ3cmになるように、測定面(塗布層)を内面として箱形の形状を作成する。次いで、上記の方法で作成した箱の中にDMF(ジメチルスルホアミド)4mlを入れて3分間放置した後、DMFを回収する。回収したDMFを液体クロマトグラフィー(株式会社島津製作所製:LC-7A)に供給して、DMF中のエステル環状三量体量を求め、この値を、DMFを接触させたフィルム面積で割って、フィルム表面に析出するエステル環状三量体量(mg/m2)とした。なお、DMF中のエステル環状三量体量は上記(3)ポリエステル原料に含有される含有オリゴマー量の測定方法に記載の絶対検量線法に従い算出した。
<ポリエステル(A)の製造方法>
テレフタル酸ジメチル100重量部とエチレングリコール60重量部とを出発原料とし、触媒として酢酸マグネシウム・四水塩0.09重量部を反応器にとり、反応開始温度を150℃とし、メタノールの留去とともに徐々に反応温度を上昇させ、3時間後に230℃とした。4時間後、実質的にエステル交換反応を終了させた。この反応混合物にエチルアシッドフォスフェート0.04部を添加した後、三酸化アンチモン0.04部を加えて、4時間重縮合反応を行った。すなわち、温度を230℃から徐々に昇温し280℃とした。一方、圧力は常圧より徐々に減じ、最終的には0.3mmHgとした。反応開始後、反応槽の攪拌動力の変化により、極限粘度0.63に相当する時点で反応を停止し、窒素加圧下ポリマーを吐出させた。得られたポリエステル(A)の極限粘度は0.63、エステル環状三量体の含有量は0.97重量%であった。
ポリエステル(A)を、予め160℃で予備結晶化させた後、温度220℃の窒素雰囲気下で固相重合し、極限粘度0.75、エステル環状三量体の含有量が0.46重量%のポリエステル(B)を得た。
テレフタル酸ジメチル100重量部、エチレングリコール60重量部、エチルアシッドフォスフェートを生成ポリエステルに対して30ppm、触媒として酢酸マグネシウム・四水和物を生成ポリエステルに対して100ppmを窒素雰囲気下、260℃でエステル化反応をさせた。引き続いて、テトラブチルチタネートを生成ポリエステルに対して50ppm添加し、2時間30分かけて280℃まで昇温すると共に、絶対圧力0.3kPaまで減圧し、さらに80分、溶融重縮合させ、極限粘度0.61のポリエステル(A)エステル環状三量体の含有量が1.02重量%のポリエステル(C)を得た。
ポリエステル(C)を、予め160℃で予備結晶化させた後、温度210℃の窒素雰囲気下で固相重合し、極限粘度0.72、エステル環状三量体の含有量が0.50重量%のポリエステル(D)を得た。
ポリエステル1の製造方法において、エチルアシッドフォスフェート0.04部を添加後、エチレングリコールに分散させた平均粒子径(d50)が1.6μmのシリカ粒子を0.5部、三酸化アンチモン0.04部を加えて、極限粘度0.65に相当する時点で重縮合反応を停止した以外は、ポリエステル1の製造方法と同様の方法を用いてポリエステル(C)を得た。得られたポリエステル(C)は、極限粘度0.65、エステル環状三量体の含有量は0.82重量%であった。
(A1):グリオキサールに無水グルコースを反応させた多価アルデヒド系化合物(B1):対イオンがメチルスルホネートである、2-(トリメチルアミノ)エチルメタクリレート/エチルメタクリレート/ブチルメタクリレート/ポリエチレングリコール含有モノアクリレートが、重量比で75/12/15/30である共重合ポリマー 数平均分子量が150000
(B2):対イオンがメチルスルホネートである、2-ヒドロキシ3-メタクリルオキシプロピルトリメチルアンモノウム塩ポリマー。数平均分子量が130000
(B3):けん化度=88モル%、重合度500のポリビニルアルコール
(B4):けん化度=93モル%、アセトアセチル基含有ポリビニルアルコール
(B5):数平均分子量が20000の、ポリエチレングリコール含有モノアクリレートポリマー
(C1):ヘキサメトキシメチロールメラミン
(C2):下記方法で合成したブロックポリイソシアネート
ヘキサメチレンジイソシアネート1000部を60℃で攪拌し、触媒としてテトラメチルアンモニウム・カプリエート0.1部を加えた。4時間後、リン酸0.2部を添加して反応を停止させ、イソシアヌレート型ポリイソシアネート組成物を得た。得られたイソシアヌレート型ポリイソシアネート組成物100部、数平均分子量400のメトキシポリエチレングリコール42.3部、プロピレングリコールモノメチルエーテルアセテート29.5部を仕込み、80℃で7時間保持した。その後反応液温度を60℃に保持し、イソブタノイル酢酸メチル35.8部、マロン酸ジエチル32.2部、ナトリウムメトキシドの28%メタノール溶液0.88部を添加し、4時間保持した。n-ブタノール58.9部を添加し、反応液温度80℃で2時間保持し、その後、2-エチルヘキシルアシッドホスフェート0.86部を添加して得られたブロックポリイソシアネート
(C3):オキサゾリン基及びポリアルキレンオキシド鎖を有するアクリルポリマー(オキサゾリン基量=7.7mmol/g)
(C4):ポリグリセロールポリグリシジルエーテル
(F1):平均粒径0.07μmのシリカ粒子
(F2):平均粒径0.02μmのアルミナ変性シリカ粒子
ポリエステル(B)、(E)を重量比で80/20でブレンドしたものを表層、ポリエステル(A)のみのものを中間層の原料として、二台の押出機にそれぞれを供給し、285℃に加熱溶融した後、A層を最外層(表層)、B層を中間層として、2種3層(A/B/A)で厚み構成比がA/B/A=4/117/4になるよう共押出し、静電密着法を用いて表面温度40~50℃の鏡面冷却ドラムに密着させながら冷却固化させて、未延伸ポリエチレンテレフタレートフィルムを作成した。このフィルムを85℃の加熱ロール群を通過させながら、ロール周速差を利用して縦方向に3.4倍延伸した後、この縦延伸フィルムの片面に、下記表1に示す水系の塗布液1を塗布し、テンター延伸機に導き、100℃で横方向に4.0倍延伸し、さらに230℃で熱処理を施した後、横方向に2%の弛緩処理を行い、塗布層の膜厚(乾燥後)が0.02μmの塗布層を有するフィルム厚みが125μmの二軸配向ポリエチレンテレフタレートフィルムを得た。
実施例1において、塗布剤組成を表1に示す塗布剤組成に変更する以外は実施例1と同様にして製造し、ポリエステルフィルムを得た。得られたポリエステルフィルムは表3に示す特性を有し、加熱処理によるフィルムヘーズ上昇値(ΔH)は小さく、エステル環状三量体の析出量は少なく、良好であった。
実施例1において、厚み構成比をA/B/A=5/178/5にし、ポリエステルフィルムの厚みを188μmに変更した以外は、実施例1と同様の方法でポリエステルフィルムを得た。得られたポリエステルフィルムの特性を表3に示す。
実施例1において、厚み構成比をA/B/A=2.5/45/2.5にし、ポリエステルフィルムの厚みを50μmに変更した以外は、実施例1と同様の方法でポリエステルフィルムを得た。得られたポリエステルフィルムの特性を表3に示す。
実施例1において、厚み構成比をA/B/A=2.5/45/2.5にし、ポリエステルフィルムの厚みを50μmに変更した以外は、実施例1と同様の方法でポリエステルフィルムを得た。得られたポリエステルフィルムの特性を表3に示す。
ポリエステル(A)、(E)を重量比で80/20でブレンドしたものを表層、ポリエステル(D)のみのものを中間層の原料として、二台の押出機にそれぞれを供給し、285℃に加熱溶融した後、A層を最外層(表層)、B層を中間層として、2種3層(A/B/A)で厚み構成比がA/B/A=1.5/22/1.5になるよう共押出し、静電密着法を用いて表面温度40~50℃の鏡面冷却ドラムに密着させながら冷却固化させて、未延伸ポリエチレンテレフタレートフィルムを作成した。このフィルムを85℃の加熱ロール群を通過させながら、ロール周速差を利用して縦方向に3.4倍延伸した後、この縦延伸フィルムの片面に、下記表1に示す水系の塗布液1を塗布し、テンター延伸機に導き、100℃で横方向に4.0倍延伸し、さらに230℃で熱処理を施した後、横方向に2%の弛緩処理を行い、塗布層の膜厚(乾燥後)が0.02μmの塗布層を有するフィルム厚みが25μmの二軸配向ポリエチレンテレフタレートフィルムを得た。
ポリエステル(D)、(E)を重量比で80/20でブレンドしたものを表層、ポリエステル(D)のみのものを中間層の原料として、二台の押出機にそれぞれを供給し、285℃に加熱溶融した後、A層を最外層(表層)、B層を中間層として、2種3層(A/B/A)で厚み構成比がA/B/A=1.5/22/1.5になるよう共押出し、静電密着法を用いて表面温度40~50℃の鏡面冷却ドラムに密着させながら冷却固化させて、未延伸ポリエチレンテレフタレートフィルムを作成した。このフィルムを85℃の加熱ロール群を通過させながら、ロール周速差を利用して縦方向に3.4倍延伸した後、この縦延伸フィルムの片面に、下記表1に示す水系の塗布液1を塗布し、テンター延伸機に導き、100℃で横方向に4.0倍延伸し、さらに230℃で熱処理を施した後、横方向に2%の弛緩処理を行い、塗布層の膜厚(乾燥後)が0.02μmの塗布層を有するフィルム厚みが25μmの二軸配向ポリエチレンテレフタレートフィルムを得た。
実施例18において、塗布剤組成を表1に示す塗布剤組成に変更する以外は実施例1と同様にして製造し、ポリエステルフィルムを得た。
実施例1において、塗布剤組成を表1に示す塗布剤組成に変更する以外は実施例1と同様にして製造し、ポリエステルフィルムを得た。得られた塗布フィルムを評価したところ、表3に示すとおり、加熱処理によるフィルムヘーズが大きく上昇し、エステル環状三量体の析出も多いものであった。
実施例1において、A層の原料として、ポリエステル(A)、(E)を、それぞれ80%、20%の割合で混合した混合原料に変更した以外は、実施例1と同様の方法でポリエステルフィルムを得た。得られた塗布フィルムを評価したところ、表3に示すとおり、加熱処理によるフィルムヘーズが大きく上昇し、エステル環状三量体の析出も多いものであった。
比較例8において、厚み構成比をA/B/A=2.5/45/2.5にし、ポリエステルフィルムの厚みを50μmに変更した以外は、比較例9と同様の方法でポリエステルフィルムを得た。得られたポリエステルフィルムの特性を表3に示す。
ポリエステル(C)、(E)を重量比で80/20でブレンドしたものを表層、ポリエステル(C)のみのものを中間層の原料として、二台の押出機にそれぞれを供給し、285℃に加熱溶融した後、A層を最外層(表層)、B層を中間層として、2種3層(A/B/A)で厚み構成比がA/B/A=1.5/22/1.5になるよう共押出し、静電密着法を用いて表面温度40~50℃の鏡面冷却ドラムに密着させながら冷却固化させて、未延伸ポリエチレンテレフタレートフィルムを作成した。このフィルムを85℃の加熱ロール群を通過させながら、ロール周速差を利用して縦方向に3.4倍延伸した後、この縦延伸フィルムの片面に、下記表1に示す水系の塗布液1を塗布し、テンター延伸機に導き、100℃で横方向に4.0倍延伸し、さらに230℃で熱処理を施した後、横方向に2%の弛緩処理を行い、塗布層の膜厚(乾燥後)が0.02μmの塗布層を有するフィルム厚みが25μmの二軸配向ポリエチレンテレフタレートフィルムを得た。
実施例1において、塗布層を設けないこと以外は実施例1と同様にして製造し、ポリエステルフィルムを得た。得られた塗布フィルムを評価したところ、熱処理によるフィルムヘーズが大きく上昇し、エステル環状三量体の析出も多いものであり、工程の汚染や、加熱後の白化による視認性の悪化が懸念されるものであった。
Claims (7)
- エステル環状三量体含有量が0.7重量%以下の単層ポリエステルフィルム又はエステル環状三量体含有量が0.7重量%以下のポリエステル表層を有する多層ポリエステルフィルムの少なくとも片面に多価アルデヒド系化合物を含有する塗布液から形成された塗布層を有することを特徴とする塗布フィルム。
- 単層ポリエステルフィルム又は多層ポリエステルフィルムの厚みが10~300μmであり、多層ポリエステルフィルムの表層の膜厚が1.5μm以上である請求項1に記載の塗布フィルム。
- ポリエステルフィルムの多層構成が2種3層である求項1又は2に記載の塗布フィルム。
- 多価アルデヒド系化合物が多価アルデヒド化合物と多価アルコール化合物とのアセタール化反応物である請求項1~3の何れかに記載の塗布フィルム。
- 多価アルデヒド系化合物の割合が塗布液中の全不揮発成分に対する割合として1~50重量%の範囲である請求項1~4の何れかに記載の塗布フィルム。
- 塗布液が更にアンモニウム基含有ポリマー、ポリビニルアルコール、および架橋剤の群から選ばれる少なくとも一種の化合物を含有する請求項1~5の何れかに記載の塗布フィルム。
- アンモニウム基含有ポリマー、ポリビニルアルコール、架橋剤の各割合が塗布液中の全不揮発成分に対する割合として5~80重量%の範囲である請求項6に記載の塗布フィルム。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020157016138A KR101776598B1 (ko) | 2013-04-06 | 2014-01-24 | 도포 필름 |
| US14/770,212 US20160009884A1 (en) | 2013-04-06 | 2014-01-24 | Coated film |
| EP14779986.0A EP2982509B1 (en) | 2013-04-06 | 2014-01-24 | Applied film |
| CN201480010861.6A CN105008130B (zh) | 2013-04-06 | 2014-01-24 | 涂布膜 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013080022 | 2013-04-06 | ||
| JP2013-080022 | 2013-04-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014162761A1 true WO2014162761A1 (ja) | 2014-10-09 |
Family
ID=51658067
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/051476 WO2014162761A1 (ja) | 2013-04-06 | 2014-01-24 | 塗布フィルム |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20160009884A1 (ja) |
| EP (1) | EP2982509B1 (ja) |
| JP (1) | JP5764277B2 (ja) |
| KR (1) | KR101776598B1 (ja) |
| CN (1) | CN105008130B (ja) |
| WO (1) | WO2014162761A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014210425A (ja) * | 2013-04-06 | 2014-11-13 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
| JP2015025084A (ja) * | 2013-07-27 | 2015-02-05 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101686394B1 (ko) * | 2012-03-26 | 2016-12-14 | 미쓰비시 쥬시 가부시끼가이샤 | 도포 필름 |
| JP2015021014A (ja) * | 2013-07-16 | 2015-02-02 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
| JP5778724B2 (ja) * | 2013-08-09 | 2015-09-16 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
| JP5840182B2 (ja) * | 2013-08-20 | 2016-01-06 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
| CN108025541B (zh) * | 2016-03-19 | 2021-01-15 | 三菱化学株式会社 | 涂布膜 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001162748A (ja) * | 1999-12-07 | 2001-06-19 | Mitsubishi Polyester Film Copp | 蒸着用ポリエステルフィルム |
| JP2007042473A (ja) | 2005-08-04 | 2007-02-15 | Nitto Denko Corp | 導電性積層フィルム、タッチパネル用電極板およびタッチパネル |
| JP2007200823A (ja) | 2006-01-30 | 2007-08-09 | Nitto Denko Corp | 結晶性透明導電性薄膜、その製造方法、透明導電性フィルムおよびタッチパネル |
| JP2007320144A (ja) | 2006-05-31 | 2007-12-13 | Toyobo Co Ltd | 導電性フィルム用積層ポリエステルフィルム |
| JP2007327065A (ja) * | 2007-07-31 | 2007-12-20 | Mitsubishi Polyester Film Copp | 積層ポリエステルフィルム |
| JP2011201172A (ja) * | 2010-03-26 | 2011-10-13 | Mitsubishi Plastics Inc | 光学用二軸延伸ポリエステルフィルムロール |
| JP2011230442A (ja) * | 2010-04-30 | 2011-11-17 | Mitsubishi Plastics Inc | 積層ポリエステルフィルム |
| JP2012025850A (ja) * | 2010-07-23 | 2012-02-09 | Mitsubishi Plastics Inc | 積層ポリエステルフィルム |
Family Cites Families (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2263642A1 (en) * | 1974-03-05 | 1975-10-03 | Faeam | Capacitive proximity alarm - has plug varying resonant frequency of oscillator reference and control coils |
| US4655841A (en) * | 1984-01-04 | 1987-04-07 | Ulano Corporation | Hardener composition |
| US4695606A (en) * | 1986-09-22 | 1987-09-22 | Sun Chemical Corporation | Coating binder additive |
| GB9324989D0 (en) * | 1992-12-23 | 1994-01-26 | Ici Plc | Polymeric film |
| DE19636510A1 (de) * | 1996-09-09 | 1998-03-12 | Clariant Gmbh | Polyvinylalkohol-Kompositionen |
| DE19651325A1 (de) * | 1996-12-11 | 1998-06-18 | Basf Ag | Verfahren zur Herstellung von Glyoxalmonoacetalen |
| US5973043A (en) * | 1997-11-26 | 1999-10-26 | Milliken & Company | Carbamoyl substituted acetals and compositions containing the same |
| WO1999041249A1 (fr) * | 1998-02-17 | 1999-08-19 | Daicel Chemical Industries, Ltd. | Procedes de production de monoacetals de dialdehyde |
| KR100579633B1 (ko) * | 1999-01-21 | 2006-05-12 | 도요 보세키 가부시키가이샤 | 광학용 이접착필름 및 그 롤 |
| AU780389B2 (en) * | 1999-11-11 | 2005-03-17 | Mitsubishi Chemical Corporation | Polyester resin and its production process |
| DE10149063A1 (de) * | 2001-10-05 | 2003-04-17 | Basf Ag | Verfahren zur Herstellung von Diacetalen des Glyoxals |
| JP2003238734A (ja) * | 2002-02-13 | 2003-08-27 | Daicel Chem Ind Ltd | ガスバリア性組成物及びそれを用いて形成したフィルム |
| JP2003301057A (ja) * | 2002-04-09 | 2003-10-21 | Mitsubishi Polyester Film Copp | 光学用易接着ポリエステルフィルム |
| JP2004010875A (ja) * | 2002-06-12 | 2004-01-15 | Mitsubishi Polyester Film Copp | ディスプレイ用ポリエステルフィルム |
| DE10255647A1 (de) * | 2002-11-28 | 2004-06-09 | Basf Ag | Verfahren zur Gewinnung eines aliphatischen Dialdehyd-Monoacetals |
| JP4923385B2 (ja) * | 2004-04-01 | 2012-04-25 | 東レ株式会社 | ポリエステルフィルム |
| CN101001895B (zh) * | 2004-08-18 | 2010-06-16 | 日本合成化学工业株式会社 | 侧链具有1,2-二醇键的聚乙烯醇及其制造方法 |
| EP1785443A4 (en) * | 2004-08-30 | 2008-10-29 | Mitsubishi Polyester Film Corp | POLYESTER FILMS FOR AUTHORIZATION |
| JP2006169467A (ja) * | 2004-12-20 | 2006-06-29 | Mitsubishi Polyester Film Copp | 光学用ポリエステルフィルム |
| JP2006181770A (ja) * | 2004-12-27 | 2006-07-13 | Mitsubishi Polyester Film Copp | 易接着性ポリエステルフィルム |
| JP2007023174A (ja) * | 2005-07-19 | 2007-02-01 | Mitsubishi Polyester Film Copp | 保護フィルム用ポリエステルフィルム |
| CN101421321B (zh) * | 2006-04-19 | 2012-07-11 | 日本合成化学工业株式会社 | 聚乙烯醇类树脂及其用途 |
| JP4976259B2 (ja) * | 2007-10-25 | 2012-07-18 | 帝人デュポンフィルム株式会社 | 偏光フィルム製造工程で支持体として用いられる支持体用フィルム |
| JP2009140532A (ja) * | 2007-12-04 | 2009-06-25 | Toray Ind Inc | 磁気記録媒体用支持体および磁気記録媒体 |
| JP5064479B2 (ja) * | 2008-12-26 | 2012-10-31 | 花王株式会社 | ガスバリア用材料及びガスバリア性成形体とその製造方法 |
| WO2010140575A1 (ja) * | 2009-06-05 | 2010-12-09 | 東洋紡績株式会社 | 容器の胴巻ラベル用二軸配向ポリエステルフィルム及び容器の胴巻用ラベル |
| JP5441583B2 (ja) * | 2009-09-18 | 2014-03-12 | 三菱樹脂株式会社 | 両面積層ポリエステルフィルム |
| JP5281554B2 (ja) * | 2009-11-30 | 2013-09-04 | 三菱樹脂株式会社 | 離型フィルム |
| KR101686394B1 (ko) * | 2012-03-26 | 2016-12-14 | 미쓰비시 쥬시 가부시끼가이샤 | 도포 필름 |
| EP2982509B1 (en) * | 2013-04-06 | 2018-03-28 | Mitsubishi Chemical Corporation | Applied film |
| JP2015021014A (ja) * | 2013-07-16 | 2015-02-02 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
| JP2015139925A (ja) * | 2014-01-28 | 2015-08-03 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
-
2014
- 2014-01-24 EP EP14779986.0A patent/EP2982509B1/en active Active
- 2014-01-24 WO PCT/JP2014/051476 patent/WO2014162761A1/ja active Application Filing
- 2014-01-24 US US14/770,212 patent/US20160009884A1/en not_active Abandoned
- 2014-01-24 CN CN201480010861.6A patent/CN105008130B/zh active Active
- 2014-01-24 KR KR1020157016138A patent/KR101776598B1/ko active IP Right Grant
- 2014-01-28 JP JP2014012875A patent/JP5764277B2/ja active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001162748A (ja) * | 1999-12-07 | 2001-06-19 | Mitsubishi Polyester Film Copp | 蒸着用ポリエステルフィルム |
| JP2007042473A (ja) | 2005-08-04 | 2007-02-15 | Nitto Denko Corp | 導電性積層フィルム、タッチパネル用電極板およびタッチパネル |
| JP2007200823A (ja) | 2006-01-30 | 2007-08-09 | Nitto Denko Corp | 結晶性透明導電性薄膜、その製造方法、透明導電性フィルムおよびタッチパネル |
| JP2007320144A (ja) | 2006-05-31 | 2007-12-13 | Toyobo Co Ltd | 導電性フィルム用積層ポリエステルフィルム |
| JP2007327065A (ja) * | 2007-07-31 | 2007-12-20 | Mitsubishi Polyester Film Copp | 積層ポリエステルフィルム |
| JP2011201172A (ja) * | 2010-03-26 | 2011-10-13 | Mitsubishi Plastics Inc | 光学用二軸延伸ポリエステルフィルムロール |
| JP2011230442A (ja) * | 2010-04-30 | 2011-11-17 | Mitsubishi Plastics Inc | 積層ポリエステルフィルム |
| JP2012025850A (ja) * | 2010-07-23 | 2012-02-09 | Mitsubishi Plastics Inc | 積層ポリエステルフィルム |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2982509A4 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2014210425A (ja) * | 2013-04-06 | 2014-11-13 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
| JP2015025084A (ja) * | 2013-07-27 | 2015-02-05 | 三菱樹脂株式会社 | 積層ポリエステルフィルム |
Also Published As
| Publication number | Publication date |
|---|---|
| CN105008130B (zh) | 2018-03-16 |
| JP5764277B2 (ja) | 2015-08-19 |
| JP2014210425A (ja) | 2014-11-13 |
| EP2982509B1 (en) | 2018-03-28 |
| EP2982509A4 (en) | 2016-11-09 |
| EP2982509A1 (en) | 2016-02-10 |
| CN105008130A (zh) | 2015-10-28 |
| KR101776598B1 (ko) | 2017-09-08 |
| KR20150132081A (ko) | 2015-11-25 |
| US20160009884A1 (en) | 2016-01-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101686394B1 (ko) | 도포 필름 | |
| JP5764277B2 (ja) | 積層ポリエステルフィルム | |
| KR101912260B1 (ko) | 도포 필름 | |
| JP5889244B2 (ja) | 積層ポリエステルフィルム | |
| JP5728147B2 (ja) | 積層ポリエステルフィルム | |
| JP5778724B2 (ja) | 積層ポリエステルフィルム | |
| JP6057724B2 (ja) | 積層ポリエステルフィルム | |
| JP5730938B2 (ja) | 積層ポリエステルフィルム | |
| JP5800851B2 (ja) | 積層ポリエステルフィルム | |
| JP5730961B2 (ja) | 積層ポリエステルフィルム | |
| JP5970116B2 (ja) | 積層ポリエステルフィルム | |
| JP5787913B2 (ja) | 積層ポリエステルフィルム | |
| JP6057739B2 (ja) | 積層ポリエステルフィルム | |
| JP5851443B2 (ja) | 積層ポリエステルフィルム | |
| KR101921898B1 (ko) | 도포 필름 | |
| JP2011224856A (ja) | 積層ポリエステルフィルム | |
| JP2014231152A (ja) | 導電性フィルム用保護フィルムロール | |
| JP2014231151A (ja) | 導電性フィルム用保護フィルム基材 | |
| JP2015205439A (ja) | 導電性フィルム用保護フィルム用基材 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14779986 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20157016138 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2014779986 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14770212 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |