JP2004511430A - 二重特異性免疫グロブリン様抗原結合蛋白および製造方法 - Google Patents
二重特異性免疫グロブリン様抗原結合蛋白および製造方法 Download PDFInfo
- Publication number
- JP2004511430A JP2004511430A JP2001587003A JP2001587003A JP2004511430A JP 2004511430 A JP2004511430 A JP 2004511430A JP 2001587003 A JP2001587003 A JP 2001587003A JP 2001587003 A JP2001587003 A JP 2001587003A JP 2004511430 A JP2004511430 A JP 2004511430A
- Authority
- JP
- Japan
- Prior art keywords
- antigen
- binding protein
- antigen binding
- protein according
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 102000025171 antigen binding proteins Human genes 0.000 title claims abstract description 159
- 108091000831 antigen binding proteins Proteins 0.000 title claims abstract description 159
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 17
- 230000027455 binding Effects 0.000 claims abstract description 210
- 239000000427 antigen Substances 0.000 claims abstract description 119
- 108091007433 antigens Proteins 0.000 claims abstract description 118
- 102000036639 antigens Human genes 0.000 claims abstract description 118
- 108060003951 Immunoglobulin Proteins 0.000 claims abstract description 14
- 102000018358 immunoglobulin Human genes 0.000 claims abstract description 14
- 230000000295 complement effect Effects 0.000 claims abstract description 11
- 230000003013 cytotoxicity Effects 0.000 claims abstract description 9
- 231100000135 cytotoxicity Toxicity 0.000 claims abstract description 9
- 230000001404 mediated effect Effects 0.000 claims abstract description 8
- 210000004027 cell Anatomy 0.000 claims description 83
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 50
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 43
- 238000000034 method Methods 0.000 claims description 42
- 229920001184 polypeptide Polymers 0.000 claims description 42
- 102000005962 receptors Human genes 0.000 claims description 35
- 108020003175 receptors Proteins 0.000 claims description 35
- 108010053099 Vascular Endothelial Growth Factor Receptor-2 Proteins 0.000 claims description 32
- 101100372762 Rattus norvegicus Flt1 gene Proteins 0.000 claims description 22
- 101100381481 Caenorhabditis elegans baz-2 gene Proteins 0.000 claims description 21
- 230000014509 gene expression Effects 0.000 claims description 16
- 239000013604 expression vector Substances 0.000 claims description 15
- 230000004913 activation Effects 0.000 claims description 14
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 14
- 108020004414 DNA Proteins 0.000 claims description 13
- 239000000126 substance Substances 0.000 claims description 12
- 108010047041 Complementarity Determining Regions Proteins 0.000 claims description 11
- 102000004127 Cytokines Human genes 0.000 claims description 11
- 108090000695 Cytokines Proteins 0.000 claims description 11
- 108091008605 VEGF receptors Proteins 0.000 claims description 11
- 102000009484 Vascular Endothelial Growth Factor Receptors Human genes 0.000 claims description 11
- 230000033115 angiogenesis Effects 0.000 claims description 11
- 230000001580 bacterial effect Effects 0.000 claims description 11
- 108010087819 Fc receptors Proteins 0.000 claims description 9
- 102000009109 Fc receptors Human genes 0.000 claims description 9
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 claims description 9
- 210000004962 mammalian cell Anatomy 0.000 claims description 9
- 102100033177 Vascular endothelial growth factor receptor 2 Human genes 0.000 claims description 8
- 210000000987 immune system Anatomy 0.000 claims description 8
- 101710160107 Outer membrane protein A Proteins 0.000 claims description 7
- 102100033178 Vascular endothelial growth factor receptor 1 Human genes 0.000 claims description 7
- 230000015572 biosynthetic process Effects 0.000 claims description 7
- 239000012636 effector Substances 0.000 claims description 7
- 239000002773 nucleotide Substances 0.000 claims description 7
- 125000003729 nucleotide group Chemical group 0.000 claims description 7
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 claims description 5
- 102000004388 Interleukin-4 Human genes 0.000 claims description 5
- 108090000978 Interleukin-4 Proteins 0.000 claims description 5
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 claims description 5
- 230000004614 tumor growth Effects 0.000 claims description 5
- -1 EGF-R Proteins 0.000 claims description 4
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 claims description 4
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 claims description 4
- 102000000588 Interleukin-2 Human genes 0.000 claims description 4
- 108010002350 Interleukin-2 Proteins 0.000 claims description 4
- 108091008606 PDGF receptors Proteins 0.000 claims description 4
- 108020004511 Recombinant DNA Proteins 0.000 claims description 4
- 210000002540 macrophage Anatomy 0.000 claims description 4
- 102000017420 CD3 protein, epsilon/gamma/delta subunit Human genes 0.000 claims description 3
- 108050005493 CD3 protein, epsilon/gamma/delta subunit Proteins 0.000 claims description 3
- 101100481408 Danio rerio tie2 gene Proteins 0.000 claims description 3
- 102100026122 High affinity immunoglobulin gamma Fc receptor I Human genes 0.000 claims description 3
- 101000913074 Homo sapiens High affinity immunoglobulin gamma Fc receptor I Proteins 0.000 claims description 3
- 101000917826 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor II-a Proteins 0.000 claims description 3
- 101000917824 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor II-b Proteins 0.000 claims description 3
- 101000917858 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-A Proteins 0.000 claims description 3
- 101000917839 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor III-B Proteins 0.000 claims description 3
- 101000914514 Homo sapiens T-cell-specific surface glycoprotein CD28 Proteins 0.000 claims description 3
- 108010002616 Interleukin-5 Proteins 0.000 claims description 3
- 102100029204 Low affinity immunoglobulin gamma Fc region receptor II-a Human genes 0.000 claims description 3
- 102100029185 Low affinity immunoglobulin gamma Fc region receptor III-B Human genes 0.000 claims description 3
- 101100481410 Mus musculus Tek gene Proteins 0.000 claims description 3
- 102100027213 T-cell-specific surface glycoprotein CD28 Human genes 0.000 claims description 3
- 210000001744 T-lymphocyte Anatomy 0.000 claims description 3
- 108010053100 Vascular Endothelial Growth Factor Receptor-3 Proteins 0.000 claims description 3
- 102100033179 Vascular endothelial growth factor receptor 3 Human genes 0.000 claims description 3
- 230000000259 anti-tumor effect Effects 0.000 claims description 3
- 230000003472 neutralizing effect Effects 0.000 claims description 3
- 210000000440 neutrophil Anatomy 0.000 claims description 3
- 108091008598 receptor tyrosine kinases Proteins 0.000 claims description 3
- 102000027426 receptor tyrosine kinases Human genes 0.000 claims description 3
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 claims description 2
- 229910052799 carbon Inorganic materials 0.000 claims description 2
- 101000851018 Homo sapiens Vascular endothelial growth factor receptor 1 Proteins 0.000 claims 6
- 102000013463 Immunoglobulin Light Chains Human genes 0.000 claims 3
- 108010065825 Immunoglobulin Light Chains Proteins 0.000 claims 3
- 102000003675 cytokine receptors Human genes 0.000 claims 2
- 108010057085 cytokine receptors Proteins 0.000 claims 2
- 210000000822 natural killer cell Anatomy 0.000 claims 2
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims 1
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 claims 1
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 claims 1
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 claims 1
- 102100039897 Interleukin-5 Human genes 0.000 claims 1
- 210000005253 yeast cell Anatomy 0.000 claims 1
- 229940072221 immunoglobulins Drugs 0.000 abstract description 5
- 238000013461 design Methods 0.000 abstract description 4
- 230000001419 dependent effect Effects 0.000 abstract description 3
- 108090000623 proteins and genes Proteins 0.000 description 67
- 108010073929 Vascular Endothelial Growth Factor A Proteins 0.000 description 60
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 60
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 60
- 102000004169 proteins and genes Human genes 0.000 description 31
- 239000013598 vector Substances 0.000 description 29
- 102000016549 Vascular Endothelial Growth Factor Receptor-2 Human genes 0.000 description 23
- 241000699666 Mus <mouse, genus> Species 0.000 description 20
- 230000006870 function Effects 0.000 description 18
- 101000595923 Homo sapiens Placenta growth factor Proteins 0.000 description 15
- 102100035194 Placenta growth factor Human genes 0.000 description 15
- 238000003752 polymerase chain reaction Methods 0.000 description 14
- 101001001487 Homo sapiens Phosphatidylinositol-glycan biosynthesis class F protein Proteins 0.000 description 12
- 206010028980 Neoplasm Diseases 0.000 description 12
- 150000001413 amino acids Chemical class 0.000 description 12
- 230000000694 effects Effects 0.000 description 12
- 102000002260 Alkaline Phosphatase Human genes 0.000 description 11
- 108020004774 Alkaline Phosphatase Proteins 0.000 description 11
- 230000026731 phosphorylation Effects 0.000 description 11
- 238000006366 phosphorylation reaction Methods 0.000 description 11
- 239000002246 antineoplastic agent Substances 0.000 description 10
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 9
- 210000002889 endothelial cell Anatomy 0.000 description 9
- 230000003993 interaction Effects 0.000 description 9
- 241000699670 Mus sp. Species 0.000 description 8
- 108010076504 Protein Sorting Signals Proteins 0.000 description 8
- 125000000539 amino acid group Chemical group 0.000 description 8
- 238000003556 assay Methods 0.000 description 8
- 239000012634 fragment Substances 0.000 description 8
- 230000004927 fusion Effects 0.000 description 8
- 239000002609 medium Substances 0.000 description 8
- 239000002953 phosphate buffered saline Substances 0.000 description 8
- 241000588724 Escherichia coli Species 0.000 description 7
- 101000808011 Homo sapiens Vascular endothelial growth factor A Proteins 0.000 description 7
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 7
- 102000058223 human VEGFA Human genes 0.000 description 7
- 239000003446 ligand Substances 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- 230000028327 secretion Effects 0.000 description 7
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 6
- 101710117290 Aldo-keto reductase family 1 member C4 Proteins 0.000 description 6
- 102100035360 Cerebellar degeneration-related antigen 1 Human genes 0.000 description 6
- 241000724791 Filamentous phage Species 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 6
- 201000011510 cancer Diseases 0.000 description 6
- 238000004132 cross linking Methods 0.000 description 6
- 108020001507 fusion proteins Proteins 0.000 description 6
- 102000037865 fusion proteins Human genes 0.000 description 6
- 210000004408 hybridoma Anatomy 0.000 description 6
- 229960000485 methotrexate Drugs 0.000 description 6
- 239000002245 particle Substances 0.000 description 6
- 238000002823 phage display Methods 0.000 description 6
- 239000013612 plasmid Substances 0.000 description 6
- 230000003389 potentiating effect Effects 0.000 description 6
- 238000012216 screening Methods 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- 238000006467 substitution reaction Methods 0.000 description 6
- 241000283973 Oryctolagus cuniculus Species 0.000 description 5
- 230000000903 blocking effect Effects 0.000 description 5
- 230000004186 co-expression Effects 0.000 description 5
- 238000012217 deletion Methods 0.000 description 5
- 230000037430 deletion Effects 0.000 description 5
- 231100000673 dose–response relationship Toxicity 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 230000005764 inhibitory process Effects 0.000 description 5
- 238000003780 insertion Methods 0.000 description 5
- 238000005259 measurement Methods 0.000 description 5
- 230000005012 migration Effects 0.000 description 5
- 238000013508 migration Methods 0.000 description 5
- 238000004091 panning Methods 0.000 description 5
- 238000002360 preparation method Methods 0.000 description 5
- 239000011780 sodium chloride Substances 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- 108020004705 Codon Proteins 0.000 description 4
- 102100024746 Dihydrofolate reductase Human genes 0.000 description 4
- 102000018233 Fibroblast Growth Factor Human genes 0.000 description 4
- 108050007372 Fibroblast Growth Factor Proteins 0.000 description 4
- 241000701024 Human betaherpesvirus 5 Species 0.000 description 4
- 230000004663 cell proliferation Effects 0.000 description 4
- 238000010367 cloning Methods 0.000 description 4
- 229940127089 cytotoxic agent Drugs 0.000 description 4
- 108020001096 dihydrofolate reductase Proteins 0.000 description 4
- 229940126864 fibroblast growth factor Drugs 0.000 description 4
- 239000000833 heterodimer Substances 0.000 description 4
- 238000011534 incubation Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 230000037431 insertion Effects 0.000 description 4
- 229940028885 interleukin-4 Drugs 0.000 description 4
- 239000000651 prodrug Substances 0.000 description 4
- 229940002612 prodrug Drugs 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- 102100034278 Annexin A6 Human genes 0.000 description 3
- 108090001008 Avidin Proteins 0.000 description 3
- 101710132601 Capsid protein Proteins 0.000 description 3
- 101710094648 Coat protein Proteins 0.000 description 3
- 108091026890 Coding region Proteins 0.000 description 3
- 102000001301 EGF receptor Human genes 0.000 description 3
- 108060006698 EGF receptor Proteins 0.000 description 3
- 238000002965 ELISA Methods 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 3
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- 102100021181 Golgi phosphoprotein 3 Human genes 0.000 description 3
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 3
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 3
- 108700005091 Immunoglobulin Genes Proteins 0.000 description 3
- 101710125418 Major capsid protein Proteins 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 101710141454 Nucleoprotein Proteins 0.000 description 3
- 108091000080 Phosphotransferase Proteins 0.000 description 3
- 101710083689 Probable capsid protein Proteins 0.000 description 3
- 108010090804 Streptavidin Proteins 0.000 description 3
- 101150006914 TRP1 gene Proteins 0.000 description 3
- 101710120037 Toxin CcdB Proteins 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 238000001042 affinity chromatography Methods 0.000 description 3
- 229960000723 ampicillin Drugs 0.000 description 3
- AVKUERGKIZMTKX-NJBDSQKTSA-N ampicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=CC=C1 AVKUERGKIZMTKX-NJBDSQKTSA-N 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 230000008901 benefit Effects 0.000 description 3
- 229960002685 biotin Drugs 0.000 description 3
- 235000020958 biotin Nutrition 0.000 description 3
- 239000011616 biotin Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 238000007796 conventional method Methods 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 239000003937 drug carrier Substances 0.000 description 3
- 230000013020 embryo development Effects 0.000 description 3
- 230000003053 immunization Effects 0.000 description 3
- 238000002649 immunization Methods 0.000 description 3
- 238000001727 in vivo Methods 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 208000032839 leukemia Diseases 0.000 description 3
- 230000035772 mutation Effects 0.000 description 3
- 102000020233 phosphotransferase Human genes 0.000 description 3
- 238000000159 protein binding assay Methods 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 239000006228 supernatant Substances 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
- 238000002560 therapeutic procedure Methods 0.000 description 3
- 230000009261 transgenic effect Effects 0.000 description 3
- 230000014616 translation Effects 0.000 description 3
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 3
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 2
- 108090000656 Annexin A6 Proteins 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 208000005623 Carcinogenesis Diseases 0.000 description 2
- 101150074155 DHFR gene Proteins 0.000 description 2
- 238000001712 DNA sequencing Methods 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- 238000012286 ELISA Assay Methods 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 102000000743 Interleukin-5 Human genes 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 2
- 206010027476 Metastases Diseases 0.000 description 2
- 241000609499 Palicourea Species 0.000 description 2
- 102000003992 Peroxidases Human genes 0.000 description 2
- 108010038512 Platelet-Derived Growth Factor Proteins 0.000 description 2
- 102000010780 Platelet-Derived Growth Factor Human genes 0.000 description 2
- 102000011653 Platelet-Derived Growth Factor Receptors Human genes 0.000 description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 2
- 108010073443 Ribi adjuvant Proteins 0.000 description 2
- 230000024932 T cell mediated immunity Effects 0.000 description 2
- 108020005038 Terminator Codon Proteins 0.000 description 2
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 2
- 101150117115 V gene Proteins 0.000 description 2
- 230000003321 amplification Effects 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- 230000036952 cancer formation Effects 0.000 description 2
- 229940041514 candida albicans extract Drugs 0.000 description 2
- 231100000504 carcinogenesis Toxicity 0.000 description 2
- 239000013592 cell lysate Substances 0.000 description 2
- 238000010382 chemical cross-linking Methods 0.000 description 2
- 239000013599 cloning vector Substances 0.000 description 2
- 230000009918 complex formation Effects 0.000 description 2
- 230000002950 deficient Effects 0.000 description 2
- 239000007857 degradation product Substances 0.000 description 2
- UQLDLKMNUJERMK-UHFFFAOYSA-L di(octadecanoyloxy)lead Chemical compound [Pb+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O UQLDLKMNUJERMK-UHFFFAOYSA-L 0.000 description 2
- 230000029087 digestion Effects 0.000 description 2
- 239000000539 dimer Substances 0.000 description 2
- 230000009977 dual effect Effects 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 239000003623 enhancer Substances 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000000684 flow cytometry Methods 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 239000003102 growth factor Substances 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- 230000002163 immunogen Effects 0.000 description 2
- 230000001771 impaired effect Effects 0.000 description 2
- 239000003978 infusion fluid Substances 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 229940100602 interleukin-5 Drugs 0.000 description 2
- 238000007912 intraperitoneal administration Methods 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 210000001161 mammalian embryo Anatomy 0.000 description 2
- 239000003550 marker Substances 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 108020004999 messenger RNA Proteins 0.000 description 2
- 230000009401 metastasis Effects 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 235000013336 milk Nutrition 0.000 description 2
- 239000008267 milk Substances 0.000 description 2
- 210000004080 milk Anatomy 0.000 description 2
- 230000037230 mobility Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 238000002703 mutagenesis Methods 0.000 description 2
- 231100000350 mutagenesis Toxicity 0.000 description 2
- 238000003199 nucleic acid amplification method Methods 0.000 description 2
- 210000001672 ovary Anatomy 0.000 description 2
- 108040007629 peroxidase activity proteins Proteins 0.000 description 2
- YBYRMVIVWMBXKQ-UHFFFAOYSA-N phenylmethanesulfonyl fluoride Chemical compound FS(=O)(=O)CC1=CC=CC=C1 YBYRMVIVWMBXKQ-UHFFFAOYSA-N 0.000 description 2
- 238000003566 phosphorylation assay Methods 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 230000012743 protein tagging Effects 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 230000011664 signaling Effects 0.000 description 2
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 230000008685 targeting Effects 0.000 description 2
- 238000001890 transfection Methods 0.000 description 2
- 230000009466 transformation Effects 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- 239000012137 tryptone Substances 0.000 description 2
- 230000008728 vascular permeability Effects 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 239000012138 yeast extract Substances 0.000 description 2
- HKZAAJSTFUZYTO-LURJTMIESA-N (2s)-2-[[2-[[2-[[2-[(2-aminoacetyl)amino]acetyl]amino]acetyl]amino]acetyl]amino]-3-hydroxypropanoic acid Chemical compound NCC(=O)NCC(=O)NCC(=O)NCC(=O)N[C@@H](CO)C(O)=O HKZAAJSTFUZYTO-LURJTMIESA-N 0.000 description 1
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 1
- KLDBMPMWBVTYGW-AIDJSRAFSA-N 2-aminoacetic acid (2S)-2-amino-3-hydroxypropanoic acid Chemical compound NCC(O)=O.NCC(O)=O.NCC(O)=O.NCC(O)=O.OC[C@H](N)C(O)=O KLDBMPMWBVTYGW-AIDJSRAFSA-N 0.000 description 1
- NDMPLJNOPCLANR-UHFFFAOYSA-N 3,4-dihydroxy-15-(4-hydroxy-18-methoxycarbonyl-5,18-seco-ibogamin-18-yl)-16-methoxy-1-methyl-6,7-didehydro-aspidospermidine-3-carboxylic acid methyl ester Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 NDMPLJNOPCLANR-UHFFFAOYSA-N 0.000 description 1
- XZKIHKMTEMTJQX-UHFFFAOYSA-N 4-Nitrophenyl Phosphate Chemical compound OP(O)(=O)OC1=CC=C([N+]([O-])=O)C=C1 XZKIHKMTEMTJQX-UHFFFAOYSA-N 0.000 description 1
- FHIDNBAQOFJWCA-UAKXSSHOSA-N 5-fluorouridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 FHIDNBAQOFJWCA-UAKXSSHOSA-N 0.000 description 1
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 108010032595 Antibody Binding Sites Proteins 0.000 description 1
- 241000271566 Aves Species 0.000 description 1
- 241000304886 Bacilli Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 102000014914 Carrier Proteins Human genes 0.000 description 1
- 108020004635 Complementary DNA Proteins 0.000 description 1
- 102100031673 Corneodesmosin Human genes 0.000 description 1
- 101710139375 Corneodesmosin Proteins 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- 241000699802 Cricetulus griseus Species 0.000 description 1
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-UWTATZPHSA-N D-alanine Chemical compound C[C@@H](N)C(O)=O QNAYBMKLOCPYGJ-UWTATZPHSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-UHFFFAOYSA-N D-alpha-Ala Natural products CC([NH3+])C([O-])=O QNAYBMKLOCPYGJ-UHFFFAOYSA-N 0.000 description 1
- WEAHRLBPCANXCN-UHFFFAOYSA-N Daunomycin Natural products CCC1(O)CC(OC2CC(N)C(O)C(C)O2)c3cc4C(=O)c5c(OC)cccc5C(=O)c4c(O)c3C1 WEAHRLBPCANXCN-UHFFFAOYSA-N 0.000 description 1
- 206010012689 Diabetic retinopathy Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 241001524679 Escherichia virus M13 Species 0.000 description 1
- 206010015548 Euthanasia Diseases 0.000 description 1
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 1
- 102100039619 Granulocyte colony-stimulating factor Human genes 0.000 description 1
- 101000690301 Homo sapiens Aldo-keto reductase family 1 member C4 Proteins 0.000 description 1
- 101000851181 Homo sapiens Epidermal growth factor receptor Proteins 0.000 description 1
- 101000840258 Homo sapiens Immunoglobulin J chain Proteins 0.000 description 1
- 101001116548 Homo sapiens Protein CBFA2T1 Proteins 0.000 description 1
- 101000851007 Homo sapiens Vascular endothelial growth factor receptor 2 Proteins 0.000 description 1
- 102000003839 Human Proteins Human genes 0.000 description 1
- 108090000144 Human Proteins Proteins 0.000 description 1
- 241000713772 Human immunodeficiency virus 1 Species 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 1
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 1
- 102100029571 Immunoglobulin J chain Human genes 0.000 description 1
- 102000010789 Interleukin-2 Receptors Human genes 0.000 description 1
- 108010038453 Interleukin-2 Receptors Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 102000008072 Lymphokines Human genes 0.000 description 1
- 108010074338 Lymphokines Proteins 0.000 description 1
- 206010025323 Lymphomas Diseases 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- 101710204212 Neocarzinostatin Proteins 0.000 description 1
- 229930193140 Neomycin Natural products 0.000 description 1
- 102000016979 Other receptors Human genes 0.000 description 1
- 238000012408 PCR amplification Methods 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000001888 Peptone Substances 0.000 description 1
- 108010080698 Peptones Proteins 0.000 description 1
- 206010057249 Phagocytosis Diseases 0.000 description 1
- 102000004160 Phosphoric Monoester Hydrolases Human genes 0.000 description 1
- 108090000608 Phosphoric Monoester Hydrolases Proteins 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 1
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 102000004879 Racemases and epimerases Human genes 0.000 description 1
- 108090001066 Racemases and epimerases Proteins 0.000 description 1
- 102000004278 Receptor Protein-Tyrosine Kinases Human genes 0.000 description 1
- 108090000873 Receptor Protein-Tyrosine Kinases Proteins 0.000 description 1
- 108010039491 Ricin Proteins 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- 102000002262 Thromboplastin Human genes 0.000 description 1
- 108010000499 Thromboplastin Proteins 0.000 description 1
- IQFYYKKMVGJFEH-XLPZGREQSA-N Thymidine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 IQFYYKKMVGJFEH-XLPZGREQSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 108010053096 Vascular Endothelial Growth Factor Receptor-1 Proteins 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 230000009824 affinity maturation Effects 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000002491 angiogenic effect Effects 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000003466 anti-cipated effect Effects 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 230000000890 antigenic effect Effects 0.000 description 1
- 230000000712 assembly Effects 0.000 description 1
- 238000000429 assembly Methods 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- 230000001588 bifunctional effect Effects 0.000 description 1
- 108091008324 binding proteins Proteins 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000008512 biological response Effects 0.000 description 1
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical class [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 1
- 235000011132 calcium sulphate Nutrition 0.000 description 1
- 229930195731 calicheamicin Natural products 0.000 description 1
- HXCHCVDVKSCDHU-LULTVBGHSA-N calicheamicin Chemical compound C1[C@H](OC)[C@@H](NCC)CO[C@H]1O[C@H]1[C@H](O[C@@H]2C\3=C(NC(=O)OC)C(=O)C[C@](C/3=C/CSSSC)(O)C#C\C=C/C#C2)O[C@H](C)[C@@H](NO[C@@H]2O[C@H](C)[C@@H](SC(=O)C=3C(=C(OC)C(O[C@H]4[C@@H]([C@H](OC)[C@@H](O)[C@H](C)O4)O)=C(I)C=3C)OC)[C@@H](O)C2)[C@@H]1O HXCHCVDVKSCDHU-LULTVBGHSA-N 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 238000006555 catalytic reaction Methods 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 description 1
- 229960004630 chlorambucil Drugs 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- 230000002860 competitive effect Effects 0.000 description 1
- 230000024203 complement activation Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 230000001143 conditioned effect Effects 0.000 description 1
- 230000021615 conjugation Effects 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 230000009260 cross reactivity Effects 0.000 description 1
- 239000012228 culture supernatant Substances 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 239000002254 cytotoxic agent Substances 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 230000002939 deleterious effect Effects 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 238000002405 diagnostic procedure Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 238000006471 dimerization reaction Methods 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000001378 electrochemiluminescence detection Methods 0.000 description 1
- 230000005670 electromagnetic radiation Effects 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 210000002257 embryonic structure Anatomy 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000010595 endothelial cell migration Effects 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 1
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 1
- 210000003714 granulocyte Anatomy 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000005734 heterodimerization reaction Methods 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 102000045108 human EGFR Human genes 0.000 description 1
- 102000055590 human KDR Human genes 0.000 description 1
- 102000054751 human RUNX1T1 Human genes 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 229940127121 immunoconjugate Drugs 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 230000002779 inactivation Effects 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 208000027866 inflammatory disease Diseases 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 229960000318 kanamycin Drugs 0.000 description 1
- 229930027917 kanamycin Natural products 0.000 description 1
- SBUJHOSQTJFQJX-NOAMYHISSA-N kanamycin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N SBUJHOSQTJFQJX-NOAMYHISSA-N 0.000 description 1
- 229930182823 kanamycin A Natural products 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 239000008297 liquid dosage form Substances 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 238000013507 mapping Methods 0.000 description 1
- 239000008204 material by function Substances 0.000 description 1
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 1
- 229960001924 melphalan Drugs 0.000 description 1
- 230000008172 membrane trafficking Effects 0.000 description 1
- 238000010232 migration assay Methods 0.000 description 1
- 239000003226 mitogen Substances 0.000 description 1
- 230000000394 mitotic effect Effects 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 108091005573 modified proteins Proteins 0.000 description 1
- 102000035118 modified proteins Human genes 0.000 description 1
- 230000009149 molecular binding Effects 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 229940126619 mouse monoclonal antibody Drugs 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 229960000210 nalidixic acid Drugs 0.000 description 1
- MHWLWQUZZRMNGJ-UHFFFAOYSA-N nalidixic acid Chemical compound C1=C(C)N=C2N(CC)C=C(C(O)=O)C(=O)C2=C1 MHWLWQUZZRMNGJ-UHFFFAOYSA-N 0.000 description 1
- QZGIWPZCWHMVQL-UIYAJPBUSA-N neocarzinostatin chromophore Chemical compound O1[C@H](C)[C@H](O)[C@H](O)[C@@H](NC)[C@H]1O[C@@H]1C/2=C/C#C[C@H]3O[C@@]3([C@@H]3OC(=O)OC3)C#CC\2=C[C@H]1OC(=O)C1=C(O)C=CC2=C(C)C=C(OC)C=C12 QZGIWPZCWHMVQL-UIYAJPBUSA-N 0.000 description 1
- 229960004927 neomycin Drugs 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 230000014207 opsonization Effects 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 235000019319 peptone Nutrition 0.000 description 1
- 108700010839 phage proteins Proteins 0.000 description 1
- 230000008782 phagocytosis Effects 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 235000011151 potassium sulphates Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000010839 reverse transcription Methods 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 239000006152 selective media Substances 0.000 description 1
- 239000008299 semisolid dosage form Substances 0.000 description 1
- 239000012679 serum free medium Substances 0.000 description 1
- 235000020183 skimmed milk Nutrition 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 210000004989 spleen cell Anatomy 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- 231100000167 toxic agent Toxicity 0.000 description 1
- 239000003440 toxic substance Substances 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
- 239000011573 trace mineral Substances 0.000 description 1
- 235000013619 trace mineral Nutrition 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000010474 transient expression Effects 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 108010002164 tyrosine receptor Proteins 0.000 description 1
- 241000701447 unidentified baculovirus Species 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 230000006459 vascular development Effects 0.000 description 1
- 210000003556 vascular endothelial cell Anatomy 0.000 description 1
- 210000005166 vasculature Anatomy 0.000 description 1
- 230000032665 vasculature development Effects 0.000 description 1
- UGGWPQSBPIFKDZ-KOTLKJBCSA-N vindesine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(N)=O)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1N=C1[C]2C=CC=C1 UGGWPQSBPIFKDZ-KOTLKJBCSA-N 0.000 description 1
- 229960004355 vindesine Drugs 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 229950009268 zinostatin Drugs 0.000 description 1
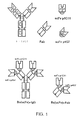
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/46—Hybrid immunoglobulins
- C07K16/468—Immunoglobulins having two or more different antigen binding sites, e.g. multifunctional antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2863—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against receptors for growth factors, growth regulators
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/60—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments
- C07K2317/62—Immunoglobulins specific features characterized by non-natural combinations of immunoglobulin fragments comprising only variable region components
- C07K2317/622—Single chain antibody (scFv)
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Cardiology (AREA)
- Heart & Thoracic Surgery (AREA)
- Peptides Or Proteins (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US20674900P | 2000-05-24 | 2000-05-24 | |
| PCT/US2001/016924 WO2001090192A2 (en) | 2000-05-24 | 2001-05-24 | Bispecific immunoglobulin-like antigen binding proteins and method of production |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2004511430A true JP2004511430A (ja) | 2004-04-15 |
| JP2004511430A5 JP2004511430A5 (cg-RX-API-DMAC7.html) | 2008-07-17 |
Family
ID=22767777
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2001587003A Pending JP2004511430A (ja) | 2000-05-24 | 2001-05-24 | 二重特異性免疫グロブリン様抗原結合蛋白および製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US20020103345A1 (cg-RX-API-DMAC7.html) |
| EP (1) | EP1299419A2 (cg-RX-API-DMAC7.html) |
| JP (1) | JP2004511430A (cg-RX-API-DMAC7.html) |
| AU (1) | AU2001264946A1 (cg-RX-API-DMAC7.html) |
| CA (1) | CA2409991A1 (cg-RX-API-DMAC7.html) |
| WO (1) | WO2001090192A2 (cg-RX-API-DMAC7.html) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005518336A (ja) * | 2001-06-26 | 2005-06-23 | イムクローン システムズ インコーポレイティド | Vegf受容体に結合する二重特異性抗体 |
| JP2005526482A (ja) * | 2001-08-10 | 2005-09-08 | イムクローン システムズ インコーポレイティド | Vegfr−1を発現する幹細胞の単離および動員 |
| JP2009504191A (ja) * | 2005-08-19 | 2009-02-05 | アボット・ラボラトリーズ | 二重可変ドメイン免疫グロブリン及びその使用 |
| JP2010535032A (ja) * | 2007-07-31 | 2010-11-18 | メディミューン,エルエルシー | 多重特異性エピトープ結合性タンパク質およびその用途 |
| JP2012228248A (ja) * | 2005-08-19 | 2012-11-22 | Abbott Lab | 二重可変ドメイン免疫グロブリン及びその使用 |
Families Citing this family (273)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6448077B1 (en) * | 1994-02-10 | 2002-09-10 | Imclone Systems, Inc. | Chimeric and humanized monoclonal antibodies specific to VEGF receptors |
| US20030108545A1 (en) * | 1994-02-10 | 2003-06-12 | Patricia Rockwell | Combination methods of inhibiting tumor growth with a vascular endothelial growth factor receptor antagonist |
| AU2469299A (en) * | 1998-01-23 | 1999-08-09 | Cornell Research Foundation Inc. | Purified populations of stem cells |
| US7754208B2 (en) | 2001-01-17 | 2010-07-13 | Trubion Pharmaceuticals, Inc. | Binding domain-immunoglobulin fusion proteins |
| CA2450954A1 (en) * | 2001-06-20 | 2003-01-03 | Imclone Systems Incorporated | Method of treating atherosclerosis and other inflammatory diseases |
| US20050271663A1 (en) * | 2001-06-28 | 2005-12-08 | Domantis Limited | Compositions and methods for treating inflammatory disorders |
| DK1399484T3 (da) * | 2001-06-28 | 2010-11-08 | Domantis Ltd | Dobbelt-specifik ligand og anvendelse af denne |
| US20060073141A1 (en) * | 2001-06-28 | 2006-04-06 | Domantis Limited | Compositions and methods for treating inflammatory disorders |
| ES2732276T3 (es) | 2001-08-23 | 2019-11-21 | Rsr Ltd | Regiones del epítopo de un receptor de tirotropina (TSH), usos del mismo y anticuerpos para el mismo |
| US20050069549A1 (en) | 2002-01-14 | 2005-03-31 | William Herman | Targeted ligands |
| ATE510561T1 (de) | 2002-03-04 | 2011-06-15 | Imclone Llc | Kdr-spezifische menschliche antikörper und ihre verwendung |
| US7696320B2 (en) | 2004-08-24 | 2010-04-13 | Domantis Limited | Ligands that have binding specificity for VEGF and/or EGFR and methods of use therefor |
| US9321832B2 (en) * | 2002-06-28 | 2016-04-26 | Domantis Limited | Ligand |
| EP2135879A3 (en) * | 2002-06-28 | 2010-06-23 | Domantis Limited | Ligand |
| BR0315117A (pt) * | 2002-10-10 | 2005-08-16 | Merck Patent Gmbh | Anticorpos anti-erb-b biespecìficos e seu uso na terapia de tumores |
| EP1578801A2 (en) * | 2002-12-27 | 2005-09-28 | Domantis Limited | Dual specific single domain antibodies specific for a ligand and for the receptor of the ligand |
| EP2389953A1 (en) | 2003-06-09 | 2011-11-30 | Samuel Waksal | Method of inhibiting receptor tyrosine kinases with an extracellular antagonist and an intracellular antagonist |
| WO2005035754A1 (ja) | 2003-10-14 | 2005-04-21 | Chugai Seiyaku Kabushiki Kaisha | 機能蛋白質を代替する二重特異性抗体 |
| US8298532B2 (en) | 2004-01-16 | 2012-10-30 | Regeneron Pharmaceuticals, Inc. | Fusion polypeptides capable of activating receptors |
| US7534604B2 (en) | 2004-01-16 | 2009-05-19 | Regeneron Pharmaceuticals, Inc. | Fusion polypeptides capable of activating receptors |
| US20050282233A1 (en) * | 2004-03-05 | 2005-12-22 | Ludwig Institute For Cancer Research | Multivalent antibody materials and methods for VEGF/PDGF family of growth factors |
| ATE507240T1 (de) * | 2004-03-05 | 2011-05-15 | Vegenics Pty Ltd | Materialien und verfahren für wachstumsfaktorbindende konstrukte |
| CA2558811A1 (en) * | 2004-03-08 | 2005-09-22 | Zymogenetics, Inc. | Dimeric fusion proteins and materials and methods for producing them |
| US8124085B2 (en) | 2004-05-05 | 2012-02-28 | Merrimack Pharmaceuticals, Inc. | Bispecific binding agents for modulating biological activity |
| EP1773881A4 (en) * | 2004-05-13 | 2008-08-06 | Imclone Systems Inc | Inhibition of macrophage-stimulating protein receptor (RON) |
| TW200607523A (en) | 2004-06-01 | 2006-03-01 | Domantis Ltd | Drug compositions, fusions and conjugates |
| CN102532321B (zh) * | 2004-06-18 | 2018-10-12 | Ambrx公司 | 新颖抗原结合多肽和其用途 |
| US7563443B2 (en) | 2004-09-17 | 2009-07-21 | Domantis Limited | Monovalent anti-CD40L antibody polypeptides and compositions thereof |
| US7972596B2 (en) | 2004-11-18 | 2011-07-05 | Imclone Llc | Antibodies against vascular endothelial growth factor receptor-1 |
| KR101404512B1 (ko) | 2005-01-05 | 2015-01-29 | 에프-스타 비오테크놀로기쉐 포르슝스 운드 엔트비클룽스게스.엠.베.하. | 상보성 결정부위와 다른 분자의 부위에서 처리된 결합성을갖는 합성 면역글로불린 영역 |
| AU2006230099B2 (en) | 2005-03-25 | 2012-04-19 | Gitr, Inc. | GITR binding molecules and uses therefor |
| EP3623473A1 (en) | 2005-03-31 | 2020-03-18 | Chugai Seiyaku Kabushiki Kaisha | Process for production of polypeptide by regulation of assembly |
| US20120237442A1 (en) * | 2005-04-06 | 2012-09-20 | Ibc Pharmaceuticals, Inc. | Design and Construction of Novel Multivalent Antibodies |
| PL2100614T3 (pl) | 2005-06-17 | 2014-02-28 | Imclone Llc | Przeciwciała przeciwko PDGFR alfa do zastosowania w leczeniu guzów nowotworowych |
| EP2937360A1 (en) * | 2005-06-17 | 2015-10-28 | Merck Sharp & Dohme Corp. | Ilt3 binding molecules and uses therefor |
| HUE026303T2 (hu) | 2005-07-25 | 2016-06-28 | Emergent Product Dev Seattle | B-sejt csökkentés CD37-specifikus és CD20-specifikus kapcsoló molekulák alkalmazásával |
| US7612181B2 (en) | 2005-08-19 | 2009-11-03 | Abbott Laboratories | Dual variable domain immunoglobulin and uses thereof |
| TW200730539A (en) * | 2005-12-01 | 2007-08-16 | Domantis Ltd | Noncompetitive domain antibody formats that bind interleukin 1 receptor type 1 |
| MX2008009977A (es) | 2006-02-03 | 2009-02-27 | Imclone Llc | Antagonistas de receptor de factor de crecimiento de tipo insulina (igf-ir) como auxiliares para el tratamiento de cancer de prostata. |
| BRPI0709843A2 (pt) | 2006-03-28 | 2011-07-26 | Biogen Idec Inc | anticorpos de anti-igf-1r e usos dos mesmos |
| KR101463631B1 (ko) | 2006-03-31 | 2014-11-19 | 추가이 세이야쿠 가부시키가이샤 | 항체의 혈중 동태를 제어하는 방법 |
| EP2009101B1 (en) | 2006-03-31 | 2017-10-25 | Chugai Seiyaku Kabushiki Kaisha | Antibody modification method for purifying bispecific antibody |
| MX2008015524A (es) | 2006-06-12 | 2009-01-13 | Trubion Pharmaceuticals Inc | Proteinas de union multivalentes monocatenarias con funcion efectora. |
| US7777008B2 (en) * | 2006-06-19 | 2010-08-17 | Tolerx, Inc. | ILT3 binding molecules and uses therefor |
| AT503889B1 (de) | 2006-07-05 | 2011-12-15 | Star Biotechnologische Forschungs Und Entwicklungsges M B H F | Multivalente immunglobuline |
| EA019595B1 (ru) | 2007-04-17 | 2014-04-30 | Имклоун Элэлси | СПЕЦИФИЧНЫЕ ИНГИБИТОРЫ PDGFRβ |
| JP2010528647A (ja) * | 2007-06-06 | 2010-08-26 | ドマンティス リミテッド | ポリペプチド、抗体可変ドメインおよびアンタゴニスト |
| KR100883430B1 (ko) * | 2007-06-13 | 2009-02-12 | 한국생명공학연구원 | 혈관내피성장인자 수용체를 중화하는 인간 단클론항체 및그 용도 |
| ES2975748T3 (es) | 2007-06-26 | 2024-07-12 | F Star Therapeutics Ltd | Presentación de agentes de unión |
| EP2175884B8 (en) | 2007-07-12 | 2017-02-22 | GITR, Inc. | Combination therapies employing gitr binding molecules |
| EP2197490A2 (en) * | 2007-08-28 | 2010-06-23 | Biogen Idec MA, Inc. | Compositions that bind multiple epitopes of igf-1r |
| RU2510400C9 (ru) | 2007-09-26 | 2014-07-20 | Чугаи Сейяку Кабусики Кайся | Способ модификации изоэлектрической точки антитела с помощью аминокислотных замен в cdr |
| CA2700714C (en) | 2007-09-26 | 2018-09-11 | Ucb Pharma S.A. | Dual specificity antibody fusions |
| EP2050764A1 (en) | 2007-10-15 | 2009-04-22 | sanofi-aventis | Novel polyvalent bispecific antibody format and uses thereof |
| UY31478A1 (es) * | 2007-11-21 | 2009-07-17 | Inhibicion del receptor para la proteina estimulante del macrofago (ron) y métodos para el tratamiento de lo mismo | |
| US9266967B2 (en) | 2007-12-21 | 2016-02-23 | Hoffmann-La Roche, Inc. | Bivalent, bispecific antibodies |
| US20090162359A1 (en) | 2007-12-21 | 2009-06-25 | Christian Klein | Bivalent, bispecific antibodies |
| WO2009120922A2 (en) | 2008-03-27 | 2009-10-01 | Zymogenetics, Inc. | Compositions and methods for inhibiting pdgfrbeta and vegf-a |
| AU2009234277B2 (en) | 2008-04-11 | 2014-12-04 | Aptevo Research And Development Llc | CD37 immunotherapeutic and combination with bifunctional chemotherapeutic thereof |
| EP2113255A1 (en) | 2008-05-02 | 2009-11-04 | f-star Biotechnologische Forschungs- und Entwicklungsges.m.b.H. | Cytotoxic immunoglobulin |
| WO2010033249A2 (en) | 2008-09-22 | 2010-03-25 | Massachusetts Institute Of Technology | Compositions of and methods using ligand dimers |
| CA2737241C (en) * | 2008-09-26 | 2017-08-29 | Ucb Pharma S.A. | Multivalent antibody fusion proteins |
| JP2012507299A (ja) * | 2008-10-31 | 2012-03-29 | バイオジェン・アイデック・エムエイ・インコーポレイテッド | Light標的分子およびその使用 |
| US9238878B2 (en) | 2009-02-17 | 2016-01-19 | Redwood Bioscience, Inc. | Aldehyde-tagged protein-based drug carriers and methods of use |
| PE20120591A1 (es) | 2009-04-02 | 2012-05-23 | Roche Glycart Ag | Anticuerpos multiespecificos que comprenden anticuerpos de longitud completa y fragmentos fab de cadena sencilla |
| WO2010115589A1 (en) | 2009-04-07 | 2010-10-14 | Roche Glycart Ag | Trivalent, bispecific antibodies |
| US9676845B2 (en) | 2009-06-16 | 2017-06-13 | Hoffmann-La Roche, Inc. | Bispecific antigen binding proteins |
| WO2011028952A1 (en) | 2009-09-02 | 2011-03-10 | Xencor, Inc. | Compositions and methods for simultaneous bivalent and monovalent co-engagement of antigens |
| CN102574924A (zh) | 2009-09-03 | 2012-07-11 | 先灵公司 | 抗-gitr抗体 |
| AU2010296018B2 (en) | 2009-09-16 | 2016-05-05 | Genentech, Inc. | Coiled coil and/or tether containing protein complexes and uses thereof |
| JP5955773B2 (ja) | 2009-11-17 | 2016-07-20 | ヤンセン バイオテツク,インコーポレーテツド | 改善された細菌膜タンパク質分泌 |
| US8728985B2 (en) | 2009-11-17 | 2014-05-20 | Centocor Ortho Biotech Inc. | Display of disulfide linked dimeric proteins in filamentous phage |
| EP2555788B1 (en) | 2010-03-24 | 2017-10-11 | Massachusetts Institute of Technology | NEUREGULIN DIMER FOR AN ErbB/HER RECEPTOR FOR USE IN REDUCING CARDIOTOXICITY. |
| TW201138821A (en) | 2010-03-26 | 2011-11-16 | Roche Glycart Ag | Bispecific antibodies |
| US8637641B2 (en) | 2010-07-29 | 2014-01-28 | Xencor, Inc. | Antibodies with modified isoelectric points |
| WO2012025530A1 (en) | 2010-08-24 | 2012-03-01 | F. Hoffmann-La Roche Ag | Bispecific antibodies comprising a disulfide stabilized - fv fragment |
| WO2012025525A1 (en) | 2010-08-24 | 2012-03-01 | Roche Glycart Ag | Activatable bispecific antibodies |
| US9334331B2 (en) | 2010-11-17 | 2016-05-10 | Chugai Seiyaku Kabushiki Kaisha | Bispecific antibodies |
| CA2818621A1 (en) | 2010-11-24 | 2012-05-31 | Glaxo Group Limited | Multispecific antigen binding proteins targeting hgf |
| ES2693232T3 (es) * | 2010-11-30 | 2018-12-10 | Chugai Seiyaku Kabushiki Kaisha | Agente terapéutico que induce citotoxicidad |
| EP2655413B1 (en) | 2010-12-23 | 2019-01-16 | F.Hoffmann-La Roche Ag | Polypeptide-polynucleotide-complex and its use in targeted effector moiety delivery |
| AU2012205301B2 (en) | 2011-01-14 | 2017-01-05 | Redwood Bioscience, Inc. | Aldehyde-tagged immunoglobulin polypeptides and method of use thereof |
| JP5764677B2 (ja) | 2011-02-28 | 2015-08-19 | エフ・ホフマン−ラ・ロシュ・アクチェンゲゼルシャフト | 抗原結合タンパク質 |
| KR101572338B1 (ko) | 2011-02-28 | 2015-11-26 | 에프. 호프만-라 로슈 아게 | 1가 항원 결합 단백질 |
| AR085911A1 (es) | 2011-03-16 | 2013-11-06 | Sanofi Sa | Dosis terapeutica segura de una proteina similar a un anticuerpo con region v dual |
| SI3865581T1 (sl) | 2011-08-05 | 2024-12-31 | Regeneron Pharmaceuticals, Inc. | Miši s humanizirano univerzalno lahko verigo |
| US12466897B2 (en) | 2011-10-10 | 2025-11-11 | Xencor, Inc. | Heterodimeric human IgG1 polypeptides with isoelectric point modifications |
| US10851178B2 (en) | 2011-10-10 | 2020-12-01 | Xencor, Inc. | Heterodimeric human IgG1 polypeptides with isoelectric point modifications |
| RU2681885C2 (ru) | 2011-10-31 | 2019-03-13 | Чугаи Сейяку Кабусики Кайся | Антигенсвязывающая молекула с регулируемой конъюгацией между тяжелой цепью и легкой цепью |
| ES2749349T3 (es) * | 2011-11-07 | 2020-03-19 | Medimmune Llc | Proteínas de unión multiespecíficas y multivalentes y usos de las mismas |
| JP6486686B2 (ja) | 2012-02-10 | 2019-03-20 | ジェネンテック, インコーポレイテッド | 単鎖抗体及び他のヘテロ多量体 |
| RU2015100656A (ru) | 2012-06-27 | 2016-08-20 | Ф. Хоффманн-Ля Рош Аг | Способ получения конъюгатов fc-фрагмента антитела, включающих по меньшей мере одну связывающую группировку, которая специфически связывается с мишенью, и их применения |
| RU2639287C2 (ru) | 2012-06-27 | 2017-12-20 | Ф. Хоффманн-Ля Рош Аг | Способ отбора и получения высокоселективных и мультиспецифичных нацеливающих групп с заданными свойствами, включающих по меньшей мере две различные связывающие группировки, и их применения |
| US9682143B2 (en) * | 2012-08-14 | 2017-06-20 | Ibc Pharmaceuticals, Inc. | Combination therapy for inducing immune response to disease |
| US20150231241A1 (en) | 2012-08-14 | 2015-08-20 | Ibc Pharmaceuticals, Inc. | Combination therapy for inducing immune response to disease |
| US9701759B2 (en) | 2013-01-14 | 2017-07-11 | Xencor, Inc. | Heterodimeric proteins |
| US11053316B2 (en) | 2013-01-14 | 2021-07-06 | Xencor, Inc. | Optimized antibody variable regions |
| US9605084B2 (en) | 2013-03-15 | 2017-03-28 | Xencor, Inc. | Heterodimeric proteins |
| CA3211863A1 (en) | 2013-01-14 | 2014-07-17 | Xencor, Inc. | Novel heterodimeric proteins |
| US10131710B2 (en) | 2013-01-14 | 2018-11-20 | Xencor, Inc. | Optimized antibody variable regions |
| US10487155B2 (en) | 2013-01-14 | 2019-11-26 | Xencor, Inc. | Heterodimeric proteins |
| US10968276B2 (en) | 2013-03-12 | 2021-04-06 | Xencor, Inc. | Optimized anti-CD3 variable regions |
| WO2014113510A1 (en) | 2013-01-15 | 2014-07-24 | Xencor, Inc. | Rapid clearance of antigen complexes using novel antibodies |
| US10519242B2 (en) | 2013-03-15 | 2019-12-31 | Xencor, Inc. | Targeting regulatory T cells with heterodimeric proteins |
| US10544187B2 (en) | 2013-03-15 | 2020-01-28 | Xencor, Inc. | Targeting regulatory T cells with heterodimeric proteins |
| US10106624B2 (en) | 2013-03-15 | 2018-10-23 | Xencor, Inc. | Heterodimeric proteins |
| US10858417B2 (en) | 2013-03-15 | 2020-12-08 | Xencor, Inc. | Heterodimeric proteins |
| ES2881306T3 (es) | 2013-09-27 | 2021-11-29 | Chugai Pharmaceutical Co Ltd | Método para la producción de heteromultímeros de polipéptidos |
| KR20160044060A (ko) | 2013-10-11 | 2016-04-22 | 에프. 호프만-라 로슈 아게 | 다중특이적 도메인 교환된 통상의 가변 경쇄 항체 |
| PT3083671T (pt) | 2013-12-20 | 2020-12-24 | Hutchinson Fred Cancer Res | Moléculas efetoras quiméricas etiquetadas e recetores destas |
| JOP20200094A1 (ar) | 2014-01-24 | 2017-06-16 | Dana Farber Cancer Inst Inc | جزيئات جسم مضاد لـ pd-1 واستخداماتها |
| JOP20200096A1 (ar) | 2014-01-31 | 2017-06-16 | Children’S Medical Center Corp | جزيئات جسم مضاد لـ tim-3 واستخداماتها |
| UY36032A (es) | 2014-03-14 | 2015-10-30 | Novartis Ag | Moléculas de anticuerpo que se unen a lag-3 y usos de las mismas |
| US20170335281A1 (en) | 2014-03-15 | 2017-11-23 | Novartis Ag | Treatment of cancer using chimeric antigen receptor |
| BR112016022385A2 (pt) | 2014-03-28 | 2018-06-19 | Xencor, Inc | anticorpos específicos que se ligam a cd38 e cd3 |
| CN106459206B (zh) | 2014-04-07 | 2024-06-07 | 中外制药株式会社 | 免疫活化抗原结合分子 |
| MX2016014434A (es) | 2014-05-13 | 2017-02-23 | Chugai Pharmaceutical Co Ltd | Molecula de union a antigeno redirigida a celulas t para celulas que tienen funcion de inmunosupresion. |
| GB201411320D0 (en) | 2014-06-25 | 2014-08-06 | Ucb Biopharma Sprl | Antibody construct |
| DK3160513T3 (da) | 2014-06-30 | 2020-04-06 | Glykos Finland Oy | Saccharidderivat af en toksisk payload og antistofkonjugater deraf |
| WO2016014553A1 (en) | 2014-07-21 | 2016-01-28 | Novartis Ag | Sortase synthesized chimeric antigen receptors |
| EP3193915A1 (en) | 2014-07-21 | 2017-07-26 | Novartis AG | Combinations of low, immune enhancing. doses of mtor inhibitors and cars |
| US9777061B2 (en) | 2014-07-21 | 2017-10-03 | Novartis Ag | Treatment of cancer using a CD33 chimeric antigen receptor |
| KR102612313B1 (ko) | 2014-07-21 | 2023-12-12 | 노파르티스 아게 | 인간화 항-bcma 키메라 항원 수용체를 사용한 암의 치료 |
| EP4205749A1 (en) | 2014-07-31 | 2023-07-05 | Novartis AG | Subset-optimized chimeric antigen receptor-containing cells |
| US10851149B2 (en) | 2014-08-14 | 2020-12-01 | The Trustees Of The University Of Pennsylvania | Treatment of cancer using GFR α-4 chimeric antigen receptor |
| WO2016028896A1 (en) | 2014-08-19 | 2016-02-25 | Novartis Ag | Anti-cd123 chimeric antigen receptor (car) for use in cancer treatment |
| BR112017005390A2 (pt) | 2014-09-17 | 2017-12-12 | Novartis Ag | células citotóxicas alvo com receptores quiméricos para imunoterapia adotiva |
| TWI700300B (zh) | 2014-09-26 | 2020-08-01 | 日商中外製藥股份有限公司 | 中和具有第viii凝血因子(fviii)機能替代活性的物質之抗體 |
| TWI701435B (zh) | 2014-09-26 | 2020-08-11 | 日商中外製藥股份有限公司 | 測定fviii的反應性之方法 |
| MA40764A (fr) | 2014-09-26 | 2017-08-01 | Chugai Pharmaceutical Co Ltd | Agent thérapeutique induisant une cytotoxicité |
| EA201790834A1 (ru) | 2014-10-14 | 2018-01-31 | Новартис Аг | Молекулы антител к pd-l1 и их применение |
| SG11201704274QA (en) | 2014-11-26 | 2017-06-29 | Xencor Inc | Heterodimeric antibodies that bind cd3 and cd38 |
| US10259887B2 (en) | 2014-11-26 | 2019-04-16 | Xencor, Inc. | Heterodimeric antibodies that bind CD3 and tumor antigens |
| WO2016086189A2 (en) | 2014-11-26 | 2016-06-02 | Xencor, Inc. | Heterodimeric antibodies that bind cd3 and tumor antigens |
| US20180334490A1 (en) | 2014-12-03 | 2018-11-22 | Qilong H. Wu | Methods for b cell preconditioning in car therapy |
| PL3227332T3 (pl) | 2014-12-03 | 2020-06-15 | F. Hoffmann-La Roche Ag | Wielospecyficzne przeciwciała |
| WO2016105450A2 (en) | 2014-12-22 | 2016-06-30 | Xencor, Inc. | Trispecific antibodies |
| WO2016141387A1 (en) | 2015-03-05 | 2016-09-09 | Xencor, Inc. | Modulation of t cells with bispecific antibodies and fc fusions |
| WO2016159213A1 (ja) | 2015-04-01 | 2016-10-06 | 中外製薬株式会社 | ポリペプチド異種多量体の製造方法 |
| HRP20220893T1 (hr) | 2015-04-08 | 2022-10-14 | Novartis Ag | Cd20 terapije, cd22 terapije, i kombinirane terapije sa stanicom koja eksprimira cd19 kimerni antigenski receptor |
| WO2016172583A1 (en) | 2015-04-23 | 2016-10-27 | Novartis Ag | Treatment of cancer using chimeric antigen receptor and protein kinase a blocker |
| EP3288570A4 (en) * | 2015-04-29 | 2018-11-21 | Fred Hutchinson Cancer Research Center | Modified stem cells and uses thereof |
| US20180207273A1 (en) | 2015-07-29 | 2018-07-26 | Novartis Ag | Combination therapies comprising antibody molecules to tim-3 |
| CN108025051B (zh) | 2015-07-29 | 2021-12-24 | 诺华股份有限公司 | 包含抗pd-1抗体分子的联合疗法 |
| HUE055469T2 (hu) | 2015-07-29 | 2021-11-29 | Novartis Ag | A LAG-3 elleni antitest molekulákat tartalmazó kombinált terápiák |
| BR112018005573A2 (pt) | 2015-09-21 | 2019-01-22 | Aptevo Research And Development Llc | ?polipeptídeos de ligação a cd3? |
| WO2017079768A1 (en) * | 2015-11-08 | 2017-05-11 | Genentech, Inc. | Methods of screening for multispecific antibodies |
| JP6931329B2 (ja) | 2015-11-18 | 2021-09-01 | 中外製薬株式会社 | 免疫抑制機能を有する細胞に対するt細胞リダイレクト抗原結合分子を用いた併用療法 |
| WO2017086419A1 (ja) | 2015-11-18 | 2017-05-26 | 中外製薬株式会社 | 液性免疫応答の増強方法 |
| CA3007030A1 (en) | 2015-12-07 | 2017-06-15 | Xencor, Inc. | Heterodimeric antibodies that bind cd3 and psma |
| JP2019503349A (ja) | 2015-12-17 | 2019-02-07 | ノバルティス アーゲー | Pd−1に対する抗体分子およびその使用 |
| RU2018125603A (ru) | 2015-12-17 | 2020-01-21 | Новартис Аг | Комбинация ингибитора с-мет с молекулой антитела к pd-1 и ее применения |
| MA44140A (fr) | 2015-12-22 | 2021-05-19 | Dana Farber Cancer Inst Inc | Récepteur d'antigène chimérique (car) contre la mésothéline et anticorps contre l'inhibiteur de pd-l1 pour une utilisation combinée dans une thérapie anticancéreuse |
| JP6954842B2 (ja) | 2015-12-25 | 2021-10-27 | 中外製薬株式会社 | 増強された活性を有する抗体及びその改変方法 |
| BR112018009312A8 (pt) | 2015-12-28 | 2019-02-26 | Chugai Pharmaceutical Co Ltd | método para promover eficiência de purificação de polipeptídeo contendo região de fc |
| US20210198368A1 (en) | 2016-01-21 | 2021-07-01 | Novartis Ag | Multispecific molecules targeting cll-1 |
| CN105481981B (zh) * | 2016-01-27 | 2019-03-19 | 中国人民解放军第二军医大学 | 靶向vegf双特异性抗体及其用途 |
| AU2017225733A1 (en) | 2016-03-04 | 2018-09-27 | Novartis Ag | Cells expressing multiple chimeric antigen receptor (CAR) molecules and uses therefore |
| CA3016424A1 (en) | 2016-03-14 | 2017-09-21 | Chugai Seiyaku Kabushiki Kaisha | Cell injury inducing therapeutic drug for use in cancer therapy |
| WO2017165683A1 (en) | 2016-03-23 | 2017-09-28 | Novartis Ag | Cell secreted minibodies and uses thereof |
| EP3443096B1 (en) | 2016-04-15 | 2023-03-01 | Novartis AG | Compositions and methods for selective expression of chimeric antigen receptors |
| US11208632B2 (en) | 2016-04-26 | 2021-12-28 | R.P. Scherer Technologies, Llc | Antibody conjugates and methods of making and using the same |
| CA3016301A1 (en) | 2016-04-28 | 2017-11-02 | Chugai Seiyaku Kabushiki Kaisha | Antibody-containing preparation |
| US20210177896A1 (en) | 2016-06-02 | 2021-06-17 | Novartis Ag | Therapeutic regimens for chimeric antigen receptor (car)- expressing cells |
| CA3026151A1 (en) | 2016-06-14 | 2017-12-21 | Xencor, Inc. | Bispecific checkpoint inhibitor antibodies |
| KR20190020341A (ko) | 2016-06-28 | 2019-02-28 | 젠코어 인코포레이티드 | 소마토스타틴 수용체 2에 결합하는 이종이량체 항체 |
| SG11201900344YA (en) | 2016-07-15 | 2019-02-27 | Novartis Ag | Treatment and prevention of cytokine release syndrome using a chimeric antigen receptor in combination with a kinase inhibitor |
| EP3491016A1 (en) | 2016-07-28 | 2019-06-05 | Novartis AG | Combination therapies of chimeric antigen receptors and pd-1 inhibitors |
| AU2017306267A1 (en) | 2016-08-01 | 2019-02-14 | Novartis Ag | Treatment of cancer using a chimeric antigen receptor in combination with an inhibitor of a pro-M2 macrophage molecule |
| US10793632B2 (en) | 2016-08-30 | 2020-10-06 | Xencor, Inc. | Bispecific immunomodulatory antibodies that bind costimulatory and checkpoint receptors |
| MY209831A (en) | 2016-09-06 | 2025-08-07 | Chugai Pharmaceutical Co Ltd | Methods of using a bispecific antibody that recognizes coagulation factor ix and/or activated coagulation factor ix and coagulation factor x and/or activated coagulation factor x |
| CA3039646A1 (en) | 2016-10-07 | 2018-04-12 | Novartis Ag | Chimeric antigen receptors for the treatment of cancer |
| PE20240950A1 (es) | 2016-10-14 | 2024-05-06 | Xencor Inc | PROTEINAS DE FUSION FC HETERODIMERICAS IL 15/IL 15R(alfa) |
| EP4043485A1 (en) | 2017-01-26 | 2022-08-17 | Novartis AG | Cd28 compositions and methods for chimeric antigen receptor therapy |
| JOP20190189A1 (ar) | 2017-02-02 | 2019-08-01 | Amgen Res Munich Gmbh | تركيبة صيدلانية ذات درجة حموضة منخفضة تتضمن بنيات جسم مضاد يستهدف الخلية t |
| KR20250089564A (ko) | 2017-02-08 | 2025-06-18 | 드래곤플라이 쎄라퓨틱스, 인크. | 천연 킬러 세포의 활성화를 위한 다중-특이적 결합 단백질 및 암 치료에서의 그의 치료적 용도 |
| KR20190120782A (ko) | 2017-02-20 | 2019-10-24 | 드래곤플라이 쎄라퓨틱스, 인크. | Her2, nkg2d 및 cd16에 결합하는 단백질 |
| EP3589647A1 (en) | 2017-02-28 | 2020-01-08 | Novartis AG | Shp inhibitor compositions and uses for chimeric antigen receptor therapy |
| EP3615068A1 (en) | 2017-04-28 | 2020-03-04 | Novartis AG | Bcma-targeting agent, and combination therapy with a gamma secretase inhibitor |
| EP3615055A1 (en) | 2017-04-28 | 2020-03-04 | Novartis AG | Cells expressing a bcma-targeting chimeric antigen receptor, and combination therapy with a gamma secretase inhibitor |
| CN111448323B (zh) * | 2017-06-07 | 2023-12-01 | 长源赋能(上海)生命科技有限公司 | 精确制导的多功能治疗抗体 |
| MX2019015885A (es) | 2017-06-22 | 2020-09-10 | Novartis Ag | Moleculas de anticuerpo que se unen a cd73 y usos de las mismas. |
| KR20200022447A (ko) | 2017-06-27 | 2020-03-03 | 노파르티스 아게 | 항-tim-3 항체의 투여 요법 및 그의 용도 |
| AU2018291497A1 (en) | 2017-06-30 | 2020-01-16 | Xencor, Inc. | Targeted heterodimeric Fc fusion proteins containing IL-15/IL-15Ra and antigen binding domains |
| PE20200757A1 (es) | 2017-07-11 | 2020-07-27 | Compass Therapeutics Llc | Anticuerpos agonistas que se unen a cd137 humano y sus usos |
| WO2019018730A1 (en) | 2017-07-20 | 2019-01-24 | Novartis Ag | DOSAGE REGIMES FOR ANTI-LAG3 ANTIBODIES AND USES THEREOF |
| EP3690050A4 (en) | 2017-09-29 | 2021-06-16 | Chugai Seiyaku Kabushiki Kaisha | MULTISPECIFIC ANTIGENBINDING MOLECULE WITH BLOOD CLOG FACTOR VIII (FVIII) COFACTOR FUNCTION SUBSTITUATING ACTIVITY AND PHARMACEUTICAL FORMULATION WITH THIS MOLECULE AS THE ACTIVE SUBSTANCE |
| US11718679B2 (en) | 2017-10-31 | 2023-08-08 | Compass Therapeutics Llc | CD137 antibodies and PD-1 antagonists and uses thereof |
| SG11202003833TA (en) | 2017-11-01 | 2020-05-28 | Chugai Pharmaceutical Co Ltd | Antibody variant and isoform with lowered biological activity |
| US11312770B2 (en) | 2017-11-08 | 2022-04-26 | Xencor, Inc. | Bispecific and monospecific antibodies using novel anti-PD-1 sequences |
| US10981992B2 (en) | 2017-11-08 | 2021-04-20 | Xencor, Inc. | Bispecific immunomodulatory antibodies that bind costimulatory and checkpoint receptors |
| US20200277378A1 (en) | 2017-11-16 | 2020-09-03 | Novartis Ag | Combination therapies |
| US11851497B2 (en) | 2017-11-20 | 2023-12-26 | Compass Therapeutics Llc | CD137 antibodies and tumor antigen-targeting antibodies and uses thereof |
| WO2019125732A1 (en) | 2017-12-19 | 2019-06-27 | Xencor, Inc. | Engineered il-2 fc fusion proteins |
| EP3737692A4 (en) | 2018-01-09 | 2021-09-29 | Elstar Therapeutics, Inc. | CALRETICULIN AND MODIFIED T-LYMPHOCYTES BINDING CONSTRUCTIONS FOR THE TREATMENT OF DISEASES |
| EP3746116A1 (en) | 2018-01-31 | 2020-12-09 | Novartis AG | Combination therapy using a chimeric antigen receptor |
| PE20210375A1 (es) | 2018-02-08 | 2021-03-02 | Dragonfly Therapeutics Inc | Terapia de combinacion del cancer que incluye proteinas de union multiespecificas que activan a las celulas asesinas naturales |
| CA3090244A1 (en) | 2018-02-08 | 2019-08-15 | Dragonfly Therapeutics, Inc. | Antibody variable domains targeting the nkg2d receptor |
| CN112119093A (zh) | 2018-02-20 | 2020-12-22 | 蜻蜓治疗公司 | 结合cd33、nkg2d和cd16的多特异性结合蛋白和使用方法 |
| EP3765517A1 (en) | 2018-03-14 | 2021-01-20 | Elstar Therapeutics, Inc. | Multifunctional molecules that bind to calreticulin and uses thereof |
| WO2019195623A2 (en) | 2018-04-04 | 2019-10-10 | Xencor, Inc. | Heterodimeric antibodies that bind fibroblast activation protein |
| US20210147547A1 (en) | 2018-04-13 | 2021-05-20 | Novartis Ag | Dosage Regimens For Anti-Pd-L1 Antibodies And Uses Thereof |
| CN112867734A (zh) | 2018-04-18 | 2021-05-28 | Xencor股份有限公司 | 包含IL-15/IL-15Ra Fc融合蛋白和PD-1抗原结合结构域的靶向PD-1的异源二聚体融合蛋白及其用途 |
| US11505595B2 (en) | 2018-04-18 | 2022-11-22 | Xencor, Inc. | TIM-3 targeted heterodimeric fusion proteins containing IL-15/IL-15RA Fc-fusion proteins and TIM-3 antigen binding domains |
| WO2019210153A1 (en) | 2018-04-27 | 2019-10-31 | Novartis Ag | Car t cell therapies with enhanced efficacy |
| CN112384534A (zh) | 2018-05-21 | 2021-02-19 | 指南针制药有限责任公司 | 用于增强nk细胞对靶细胞的杀死的组合物和方法 |
| WO2019226658A1 (en) | 2018-05-21 | 2019-11-28 | Compass Therapeutics Llc | Multispecific antigen-binding compositions and methods of use |
| WO2019227003A1 (en) | 2018-05-25 | 2019-11-28 | Novartis Ag | Combination therapy with chimeric antigen receptor (car) therapies |
| US20210214459A1 (en) | 2018-05-31 | 2021-07-15 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| EP3806962A1 (en) | 2018-06-13 | 2021-04-21 | Novartis AG | Bcma chimeric antigen receptors and uses thereof |
| MA52969A (fr) | 2018-06-19 | 2021-04-28 | Atarga Llc | Molécules d'anticorps se liant au composant du complément 5 et leurs utilisations |
| MY208638A (en) | 2018-07-02 | 2025-05-21 | Amgen Inc | Anti-steap1 antigen-binding protein |
| AU2019297451A1 (en) | 2018-07-03 | 2021-01-28 | Marengo Therapeutics, Inc. | Anti-TCR antibody molecules and uses thereof |
| AR116109A1 (es) | 2018-07-10 | 2021-03-31 | Novartis Ag | Derivados de 3-(5-amino-1-oxoisoindolin-2-il)piperidina-2,6-diona y usos de los mismos |
| WO2020021465A1 (en) | 2018-07-25 | 2020-01-30 | Advanced Accelerator Applications (Italy) S.R.L. | Method of treatment of neuroendocrine tumors |
| BR112021002276A2 (pt) | 2018-08-08 | 2021-05-04 | Dragonfly Therapeutics, Inc. | proteínas de ligação a nkg2d, cd16 e um antígeno associado ao tumor |
| CA3108427A1 (en) | 2018-08-08 | 2020-02-13 | Dragonfly Therapeutics, Inc. | Multi-specific binding proteins that bind bcma, nkg2d and cd16, and methods of use |
| EA202091888A1 (ru) | 2018-08-08 | 2020-10-23 | Драгонфлай Терапьютикс, Инк. | Вариабельные домены антител, нацеленные на рецептор nkg2d |
| EP3861016A2 (en) | 2018-10-03 | 2021-08-11 | Xencor, Inc. | Il-12 heterodimeric fc-fusion proteins |
| AU2019379576A1 (en) | 2018-11-13 | 2021-06-03 | Compass Therapeutics Llc | Multispecific binding constructs against checkpoint molecules and uses thereof |
| MX2021007391A (es) | 2018-12-20 | 2021-09-23 | Novartis Ag | Regimenes de baja dosis extendida para inhibidores de mdm2. |
| WO2020128972A1 (en) | 2018-12-20 | 2020-06-25 | Novartis Ag | Dosing regimen and pharmaceutical combination comprising 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives |
| CN111378045B (zh) * | 2018-12-28 | 2022-08-02 | 长春金赛药业有限责任公司 | 二价双特异性抗体及其制备方法、编码基因、宿主细胞、组合物 |
| JP7483732B2 (ja) | 2019-02-15 | 2024-05-15 | ノバルティス アーゲー | 3-(1-オキソ-5-(ピペリジン-4-イル)イソインドリン-2-イル)ピペリジン-2,6-ジオン誘導体及びその使用 |
| US10871640B2 (en) | 2019-02-15 | 2020-12-22 | Perkinelmer Cellular Technologies Germany Gmbh | Methods and systems for automated imaging of three-dimensional objects |
| ES2982474T3 (es) | 2019-02-15 | 2024-10-16 | Novartis Ag | Derivados de 3-(1-oxoisoindolin-2-il)piperidin-1,6-diona sustituidos y usos de estos |
| WO2020172605A1 (en) | 2019-02-21 | 2020-08-27 | Elstar Therapeutics, Inc. | Antibody molecules that bind to nkp30 and uses thereof |
| CA3130754A1 (en) | 2019-02-21 | 2020-08-27 | Marengo Therapeutics, Inc. | Multifunctional molecules that bind to t cell related cancer cells and uses thereof |
| WO2020172553A1 (en) | 2019-02-22 | 2020-08-27 | Novartis Ag | Combination therapies of egfrviii chimeric antigen receptors and pd-1 inhibitors |
| AU2020232605A1 (en) | 2019-03-01 | 2021-10-21 | Xencor, Inc. | Heterodimeric antibodies that bind ENPP3 and CD3 |
| SG11202110732XA (en) | 2019-03-29 | 2021-10-28 | Atarga Llc | Anti fgf23 antibody |
| IL292347A (en) | 2019-10-21 | 2022-06-01 | Novartis Ag | Combination treatments with ventoclax and tim-3 inhibitors |
| KR20220087498A (ko) | 2019-10-21 | 2022-06-24 | 노파르티스 아게 | Tim-3 억제제 및 그의 용도 |
| IL293215A (en) | 2019-11-26 | 2022-07-01 | Novartis Ag | Chimeric antigen receptors that bind bcma and cd19 and their uses |
| JP2023507190A (ja) | 2019-12-20 | 2023-02-21 | ノバルティス アーゲー | 増殖性疾患を治療するための抗TGFβ抗体及びチェックポイント阻害薬の使用 |
| GB2609554B (en) | 2020-01-03 | 2025-08-20 | Marengo Therapeutics Inc | Anti-TCR antibody molecules and uses thereof |
| EP4090762A1 (en) | 2020-01-17 | 2022-11-23 | Becton, Dickinson and Company | Methods and compositions for single cell secretomics |
| CN114980902A (zh) | 2020-01-17 | 2022-08-30 | 诺华股份有限公司 | 用于治疗骨髓增生异常综合征或慢性粒单核细胞白血病的包含tim-3抑制剂和低甲基化药物的组合 |
| KR20220147109A (ko) | 2020-02-27 | 2022-11-02 | 노파르티스 아게 | 키메라 항원 수용체 발현 세포의 제조 방법 |
| CA3173934A1 (en) | 2020-04-29 | 2021-11-04 | Twinkle R. Christian | Pharmaceutical formulation |
| JP2023524444A (ja) | 2020-04-29 | 2023-06-12 | アムジェン インコーポレイテッド | 医薬製剤 |
| US12157771B2 (en) | 2020-05-06 | 2024-12-03 | Dragonfly Therapeutics, Inc. | Proteins binding NKG2D, CD16 and CLEC12A |
| US11919956B2 (en) | 2020-05-14 | 2024-03-05 | Xencor, Inc. | Heterodimeric antibodies that bind prostate specific membrane antigen (PSMA) and CD3 |
| US20230321067A1 (en) | 2020-06-23 | 2023-10-12 | Novartis Ag | Dosing regimen comprising 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives |
| TW202216761A (zh) | 2020-07-16 | 2022-05-01 | 瑞士商諾華公司 | 抗β細胞素抗體、其片段及多特異性結合分子 |
| WO2022026592A2 (en) | 2020-07-28 | 2022-02-03 | Celltas Bio, Inc. | Antibody molecules to coronavirus and uses thereof |
| EP4188549A1 (en) | 2020-08-03 | 2023-06-07 | Novartis AG | Heteroaryl substituted 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives and uses thereof |
| KR20230166150A (ko) | 2020-08-19 | 2023-12-06 | 젠코어 인코포레이티드 | 항-cd28 조성물 |
| MX2023002280A (es) | 2020-08-24 | 2023-05-23 | Amgen Inc | Formulacion farmaceutica que comprende un bite, anticuerpo biespecifico y metionina. |
| US20230338587A1 (en) | 2020-08-31 | 2023-10-26 | Advanced Accelerator Applications International Sa | Method of treating psma-expressing cancers |
| WO2022043557A1 (en) | 2020-08-31 | 2022-03-03 | Advanced Accelerator Applications International Sa | Method of treating psma-expressing cancers |
| US20240002509A1 (en) | 2020-11-06 | 2024-01-04 | Novartis Ag | ANTIBODY Fc VARIANTS |
| CN116635062A (zh) | 2020-11-13 | 2023-08-22 | 诺华股份有限公司 | 使用表达嵌合抗原受体(car)的细胞的组合疗法 |
| US20240141060A1 (en) | 2021-01-29 | 2024-05-02 | Novartis Ag | Dosage regimes for anti-cd73 and anti-entpd2 antibodies and uses thereof |
| WO2022187539A1 (en) | 2021-03-03 | 2022-09-09 | Dragonfly Therapeutics, Inc. | Methods of treating cancer using multi-specific binding proteins that bind nkg2d, cd16 and a tumor-associated antigen |
| CA3212665A1 (en) | 2021-03-09 | 2022-09-15 | Xencor, Inc. | Heterodimeric antibodies that bind cd3 and cldn6 |
| WO2022192586A1 (en) | 2021-03-10 | 2022-09-15 | Xencor, Inc. | Heterodimeric antibodies that bind cd3 and gpc3 |
| TW202304979A (zh) | 2021-04-07 | 2023-02-01 | 瑞士商諾華公司 | 抗TGFβ抗體及其他治療劑用於治療增殖性疾病之用途 |
| AR125874A1 (es) | 2021-05-18 | 2023-08-23 | Novartis Ag | Terapias de combinación |
| BR112023024757A2 (pt) | 2021-06-01 | 2024-02-15 | Amgen Inc | Método acelerado de preparação de formulações de proteínas liofilizadas |
| EP4363448A1 (en) | 2021-06-30 | 2024-05-08 | Amgen Inc. | Method of reconstituting lyophilized formulation |
| WO2023044483A2 (en) | 2021-09-20 | 2023-03-23 | Voyager Therapeutics, Inc. | Compositions and methods for the treatment of her2 positive cancer |
| WO2023092004A1 (en) | 2021-11-17 | 2023-05-25 | Voyager Therapeutics, Inc. | Compositions and methods for the treatment of tau-related disorders |
| US20230383010A1 (en) | 2022-02-07 | 2023-11-30 | Visterra, Inc. | Anti-idiotype antibody molecules and uses thereof |
| CA3252619A1 (en) | 2022-02-23 | 2023-08-31 | Xencor, Inc. | Anti-CD28 and anti-PSMA antibodies |
| JP2025514115A (ja) | 2022-04-26 | 2025-05-02 | アムジエン・インコーポレーテツド | 凍結乾燥方法 |
| US20250295809A1 (en) | 2022-05-13 | 2025-09-25 | Voyager Therapeutics, Inc. | Compositions and methods for the treatment of her2 positive cancer |
| TW202435912A (zh) | 2022-08-03 | 2024-09-16 | 美商航海家醫療公司 | 用於穿過血腦屏障之組合物及方法 |
| EP4573553A1 (en) | 2022-08-15 | 2025-06-25 | ABSCI Corporation | Quantitative affinity activity specific cell enrichment |
| WO2024168061A2 (en) | 2023-02-07 | 2024-08-15 | Ayan Therapeutics Inc. | Antibody molecules binding to sars-cov-2 |
| AU2024302800A1 (en) | 2023-06-13 | 2025-11-06 | Amgen Inc. | Methods of determining viscosity |
| WO2025122634A1 (en) | 2023-12-05 | 2025-06-12 | Voyager Therapeutics, Inc. | Compositions and methods for the treatment of tau-related disorders |
| WO2025144700A1 (en) | 2023-12-27 | 2025-07-03 | Absci Corporation | Nanobody library screening using bacterial surface display |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10179151A (ja) * | 1996-09-03 | 1998-07-07 | Gsf Forschungszentrum Fuer Umwelt & Gesundheit Gmbh | 二特異的抗体を用いた幹細胞移植における汚染腫瘍細胞の除去 |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6121424A (en) * | 1991-11-25 | 2000-09-19 | Enzon, Inc. | Multivalent antigen-binding proteins |
| US5869620A (en) * | 1986-09-02 | 1999-02-09 | Enzon, Inc. | Multivalent antigen-binding proteins |
| US4946778A (en) * | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| US5202238A (en) * | 1987-10-27 | 1993-04-13 | Oncogen | Production of chimeric antibodies by homologous recombination |
| AU4128089A (en) * | 1988-09-15 | 1990-03-22 | Rorer International (Overseas) Inc. | Monoclonal antibodies specific to human epidermal growth factor receptor and therapeutic methods employing same |
| US5530101A (en) * | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US20010021382A1 (en) * | 1991-03-29 | 2001-09-13 | Genentech, Inc. | Vascular endothelial cell growth factor antagonists |
| US5367057A (en) * | 1991-04-02 | 1994-11-22 | The Trustees Of Princeton University | Tyrosine kinase receptor flk-2 and fragments thereof |
| US5861301A (en) * | 1992-02-20 | 1999-01-19 | American Cayanamid Company | Recombinant kinase insert domain containing receptor and gene encoding same |
| US5965132A (en) * | 1992-03-05 | 1999-10-12 | Board Of Regents, The University Of Texas System | Methods and compositions for targeting the vasculature of solid tumors |
| US6036955A (en) * | 1992-03-05 | 2000-03-14 | The Scripps Research Institute | Kits and methods for the specific coagulation of vasculature |
| US6177401B1 (en) * | 1992-11-13 | 2001-01-23 | Max-Planck-Gesellschaft Zur Forderung Der Wissenschaften | Use of organic compounds for the inhibition of Flk-1 mediated vasculogenesis and angiogenesis |
| US5840299A (en) * | 1994-01-25 | 1998-11-24 | Athena Neurosciences, Inc. | Humanized antibodies against leukocyte adhesion molecule VLA-4 |
| US5840301A (en) * | 1994-02-10 | 1998-11-24 | Imclone Systems Incorporated | Methods of use of chimerized, humanized, and single chain antibodies specific to VEGF receptors |
| US5861499A (en) * | 1994-02-10 | 1999-01-19 | Imclone Systems Incorporated | Nucleic acid molecules encoding the variable or hypervariable region of a monoclonal antibody that binds to an extracellular domain |
| US6448077B1 (en) * | 1994-02-10 | 2002-09-10 | Imclone Systems, Inc. | Chimeric and humanized monoclonal antibodies specific to VEGF receptors |
| ES2259247T3 (es) * | 1998-12-21 | 2006-09-16 | Ludwig Institute For Cancer Research | Anticuerpos frente a vegf-d truncado y sus usos. |
| AU3475100A (en) * | 1999-01-29 | 2000-08-18 | Imclone Systems Incorporated | Antibodies specific to kdr and uses thereof |
| US7740841B1 (en) * | 2000-01-28 | 2010-06-22 | Sunnybrook Health Science Center | Therapeutic method for reducing angiogenesis |
| AU2001283496A1 (en) * | 2000-07-25 | 2002-02-05 | Immunomedics, Inc. | Multivalent target binding protein |
-
2001
- 2001-05-24 JP JP2001587003A patent/JP2004511430A/ja active Pending
- 2001-05-24 WO PCT/US2001/016924 patent/WO2001090192A2/en not_active Ceased
- 2001-05-24 US US09/865,198 patent/US20020103345A1/en not_active Abandoned
- 2001-05-24 CA CA002409991A patent/CA2409991A1/en not_active Abandoned
- 2001-05-24 AU AU2001264946A patent/AU2001264946A1/en not_active Abandoned
- 2001-05-24 EP EP01939426A patent/EP1299419A2/en not_active Withdrawn
-
2004
- 2004-02-13 US US10/778,910 patent/US20040259156A1/en not_active Abandoned
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10179151A (ja) * | 1996-09-03 | 1998-07-07 | Gsf Forschungszentrum Fuer Umwelt & Gesundheit Gmbh | 二特異的抗体を用いた幹細胞移植における汚染腫瘍細胞の除去 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005518336A (ja) * | 2001-06-26 | 2005-06-23 | イムクローン システムズ インコーポレイティド | Vegf受容体に結合する二重特異性抗体 |
| JP2005526482A (ja) * | 2001-08-10 | 2005-09-08 | イムクローン システムズ インコーポレイティド | Vegfr−1を発現する幹細胞の単離および動員 |
| JP2009504191A (ja) * | 2005-08-19 | 2009-02-05 | アボット・ラボラトリーズ | 二重可変ドメイン免疫グロブリン及びその使用 |
| JP2012228248A (ja) * | 2005-08-19 | 2012-11-22 | Abbott Lab | 二重可変ドメイン免疫グロブリン及びその使用 |
| JP2010535032A (ja) * | 2007-07-31 | 2010-11-18 | メディミューン,エルエルシー | 多重特異性エピトープ結合性タンパク質およびその用途 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2001090192A3 (en) | 2003-01-16 |
| EP1299419A2 (en) | 2003-04-09 |
| US20020103345A1 (en) | 2002-08-01 |
| WO2001090192A2 (en) | 2001-11-29 |
| AU2001264946A1 (en) | 2001-12-03 |
| US20040259156A1 (en) | 2004-12-23 |
| CA2409991A1 (en) | 2001-11-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2004511430A (ja) | 二重特異性免疫グロブリン様抗原結合蛋白および製造方法 | |
| US20040242851A1 (en) | Bispecific antibodies that bind to vegf receptors | |
| CN113603783B (zh) | 蛋白酶可切割的双特异性抗体及其用途 | |
| CN109071654B (zh) | 靶向Fc受体样5的抗体及使用方法 | |
| JP2024053016A (ja) | Gタンパク質共役受容体を標的化する抗体および使用の方法 | |
| JP2009526857A (ja) | 機能性抗体 | |
| TWI381848B (zh) | 抗纖維母細胞生長因子受體-3 (fgfr-3) 抗體及包含其之醫藥組合物 | |
| JP2008512352A (ja) | 新規な四価の二重特異性抗体 | |
| US20020064528A1 (en) | Antibodies specific to KDR and uses thereof | |
| CN1345334A (zh) | 对kdr特异的抗体及其应用 | |
| WO1995025167A1 (en) | Anti-egfr single-chain fvs and anti-egfr antibodies | |
| KR102691493B1 (ko) | α-SYN/IGF1R에 대한 이중 특이 항체 및 그 용도 | |
| CN113135995B (zh) | 抗her3单克隆抗体及其应用 | |
| US11891446B2 (en) | Highly potent antibodies binding to death receptor 5 | |
| WO2004003211A1 (en) | Bispecific antibodies that bind to vegf receptors | |
| CA3230246A1 (en) | Bispecific antibody and use thereof | |
| HK40055460B (en) | Protease cleavable bispecific antibodies and uses thereof | |
| HK40055460A (en) | Protease cleavable bispecific antibodies and uses thereof | |
| HK40002168B (en) | Antibodies targeting fc receptor-like 5 and methods of use | |
| HK1243955B (zh) | 靶向g-蛋白偶联受体的抗体和使用方法 | |
| NZ732567B2 (en) | Antibodies targeting g-protein coupled receptor and methods of use |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20080523 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20080523 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20090812 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20090812 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20110920 |