WO2020235658A1 - オリゴヌクレオチド合成に用いるマルチフルオラスブロックマーおよびこれを用いたオリゴヌクレオチド合成方法 - Google Patents
オリゴヌクレオチド合成に用いるマルチフルオラスブロックマーおよびこれを用いたオリゴヌクレオチド合成方法 Download PDFInfo
- Publication number
- WO2020235658A1 WO2020235658A1 PCT/JP2020/020203 JP2020020203W WO2020235658A1 WO 2020235658 A1 WO2020235658 A1 WO 2020235658A1 JP 2020020203 W JP2020020203 W JP 2020020203W WO 2020235658 A1 WO2020235658 A1 WO 2020235658A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- protecting group
- protected
- group
- nucleoside
- alkyl
- Prior art date
Links
- 0 C*C(C(C)=CN1C(C2)OC(CO)C2O)=NC1=O Chemical compound C*C(C(C)=CN1C(C2)OC(CO)C2O)=NC1=O 0.000 description 7
- XNQDICSBXPAPRV-UHFFFAOYSA-N CC(C(N1)=O)=CN(C(C2)OC(COCPOCC=C)C2OP(COC)OCC=C)C1=O Chemical compound CC(C(N1)=O)=CN(C(C2)OC(COCPOCC=C)C2OP(COC)OCC=C)C1=O XNQDICSBXPAPRV-UHFFFAOYSA-N 0.000 description 1
- IPKGDZQHNGZIAS-UHFFFAOYSA-N CC(C(N1)O)=CN(C(C2)OCC2OP(OCC=C)=O)C1=O Chemical compound CC(C(N1)O)=CN(C(C2)OCC2OP(OCC=C)=O)C1=O IPKGDZQHNGZIAS-UHFFFAOYSA-N 0.000 description 1
- ZZXHGGXVFZPPRY-LILUBXCNSA-N CC(C)C(NC(NC(C1)OC(CCCP(OCC=C)OC(C2)C(CO)OC2N(C=C(C)C(N2)=O)C2=O)[C@@H]1OP(COC)OCC=C)=O)=O Chemical compound CC(C)C(NC(NC(C1)OC(CCCP(OCC=C)OC(C2)C(CO)OC2N(C=C(C)C(N2)=O)C2=O)[C@@H]1OP(COC)OCC=C)=O)=O ZZXHGGXVFZPPRY-LILUBXCNSA-N 0.000 description 1
- MVOHONKFLYPSFC-UHFFFAOYSA-N CC(CN(C1OC(CO)CC1)C(N1)=O)C1=O Chemical compound CC(CN(C1OC(CO)CC1)C(N1)=O)C1=O MVOHONKFLYPSFC-UHFFFAOYSA-N 0.000 description 1
- DKVIBEFOBOVION-AFPNSQJFSA-N CC(Nc1ncnc2c1nc[n]2C(C1)OC(CO)[C@@H]1O)=O Chemical compound CC(Nc1ncnc2c1nc[n]2C(C1)OC(CO)[C@@H]1O)=O DKVIBEFOBOVION-AFPNSQJFSA-N 0.000 description 1
- NGHAMMLXEAZBCM-UHFFFAOYSA-N CCC(CC(C)N(C=C(C)C(N1)O)C1=O)OPOCC=C Chemical compound CCC(CC(C)N(C=C(C)C(N1)O)C1=O)OPOCC=C NGHAMMLXEAZBCM-UHFFFAOYSA-N 0.000 description 1
- LZUYVBCJKHBLAE-UHFFFAOYSA-N CCC(CC(N1)O)NC1=O Chemical compound CCC(CC(N1)O)NC1=O LZUYVBCJKHBLAE-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H23/00—Compounds containing boron, silicon, or a metal, e.g. chelates, vitamin B12
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
- C07H19/073—Pyrimidine radicals with 2-deoxyribosyl as the saccharide radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H21/00—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids
- C07H21/04—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids with deoxyribosyl as saccharide radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H1/00—Processes for the preparation of sugar derivatives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
- C07H19/10—Pyrimidine radicals with the saccharide radical esterified by phosphoric or polyphosphoric acids
Definitions
- the present invention relates to a multifluorous blocker in oligonucleotide synthesis and an oligonucleotide synthesis method using the same.
- nucleic acid drugs based on natural or modified oligonucleotides. Chemical synthesis methods are widely used to obtain nucleic acid drugs designed to achieve the desired effect.
- a coupling reaction between amidite and a nucleoside or a deprotection reaction for obtaining the next reaction base point even for synthesizing a relatively short-chain oligonucleotide.
- Dozens of steps of reaction are required. Many of the steps require purification using column chromatography or the like. Since purification is often complicated, it is not easy to synthesize a large amount of oligonucleotide by a widely used liquid phase synthesis method.
- the present invention has been made in view of such circumstances, and a multifluorous blocker used for oligonucleotide synthesis, which can reduce the purification load by using an easily available fluorous tag, and an oligonucleotide synthesis using the same.
- the purpose is to provide a method.
- the first aspect of the present invention is a nucleoside protector represented by the following formula (I).
- B is a natural or modified nucleoside base
- R 1 , R 2 are independently H or protecting groups that can be deprotected under acidic, basic or neutral conditions.
- the F-protector is O (CH 2 ) n (CF 2 ) m CF 3 when the protected part of the nucleoside base B is O, and N when the protected part of the nucleoside base B is N.
- Y is H, OH, halogen, OCH 3 , Methoxyethyl, CN, CF 3, or hydroxyl groups protected by acyl-based protecting groups, ether-based protecting groups, silyl-based protecting groups;
- Z is H, alkyl, O-alkyl, N-alkyl, halogen. There is or forms a ZZ bond with the Y.
- a second aspect of the present invention is a 5'-terminal protected nucleoside phosphoramidite represented by the following formula (II).
- B is a natural or modified nucleoside base
- R 1 is a deprotectable protecting group under acidic, basic or neutral conditions
- R 3 a ring and one R 4 bonded to the nitrogen atom attached to the phosphorus atom is formed by bonding,;
- R 4 is a substituted or unsubstituted aliphatic It is a group, substituted or unsubstituted aromatic group;
- the F-projector is O (CH 2 ) n (CF 2 ) m CF 3 when the protected portion of the nucleoside base B is O, and the nucleoside.
- Y is H, OH, halogen, OCH 3 , methoxyethyl, CN, CF 3, or a hydroxyl group protected by an acyl protecting group, an ether protecting group, a silyl protecting group;
- Z is H. , Alkyl, O-alkyl, N-alkyl, halogen, or form a ZZ bond with the Y.
- a third aspect of the present invention is a fluorolas blocker amidite represented by the following formula (III).
- B is a natural or modified nucleoside base
- R 1 is a deprotectable protecting group under acidic, basic or neutral conditions
- R 3 is a ring of the one of R 5 bonded to the nitrogen atom attached to the phosphorus atom to form a amidite portion is formed by bonding;
- R 5 is a substituted Alternatively, it is an unsubstituted aliphatic group, a substituted or unsubstituted aromatic group;
- Pro is an unprotected, nucleoside base protecting group or an F-projector, and the F-projector is a protected portion of the nucleoside base B.
- Is O (CH 2 ) n (CF 2 ) m CF 3 and NH (C O) (CH 2 ) n (CF) when the protected portion of the nucleoside base B is N.
- n 1 or 2 and m is an integer from 1 to 20;
- X is O or S;
- p is an integer from 0 to 27;
- Y is H, OH, halogen, OCH 3 , methoxyethyl, CN, CF 3, or hydroxyl groups protected by acyl protecting groups, ether protecting groups, silyl protecting groups;
- Z is H, alkyl, O-alkyl, It is N-alkyl, halogen, or forms a ZZ bond with said Y.
- the fluorolas blocker amidite represented by the above formula (III) has at least one F-projector in either Pro or R 1 , R 3 .
- a fourth aspect of the present invention is a multifluorous blocker represented by the following formula (IV).
- B is a natural or modified nucleoside base
- R 1 is a protecting group that can be deprotected under acidic, basic or neutral conditions
- R 3 is a phosphoric acid protecting group.
- R is NH or S and G is an allyl or acyl group.
- t is 0 to 3
- Pro is an unprotected, protecting group or F-projector of the nucleoside base, and the F-projector is O (CH 2 ) when the protected portion of the nucleoside base B is O.
- n 1 or 2
- X is O or S
- l is an integer from 0 to 58
- Y is H, OH, halogen, OCH 3 , Methoxyethyl, CN, CF 3, or a hydroxyl group protected with an acyl protecting group, an ether protecting group, a silyl protecting group
- Z is H, alkyl, O-alkyl, N-alkyl, halogen. There is or forms a ZZ bond with the Y.
- a fifth aspect of the present invention comprises a fluorolas blocker amidite represented by the above formula (III), a 5'-terminal protected nucleoside H-phosphonate represented by the following formula (II'), and the following formula (V).
- B is a natural or modified nucleoside base
- R 1 is a protecting group that can be deprotected under acidic, basic or neutral conditions
- F-projector is a protecting group that can be deprotected under acidic, basic or neutral conditions
- Is O (CH 2 ) n (CF 2 ) m CF 3 when the protected portion of the nucleoside base B is O, and NH (C ) when the protected portion of the nucleoside base B is N.
- Y is H, OH, halogen, OCH 3 , methoxyethyl, A hydroxyl group protected with CN, CF 3, or an acyl-based protecting group, an ether-based protecting group, or a silyl-based protecting group;
- Z is H, alkyl, O-alkyl, N-alkyl, halogen, or , A ZZ bond is formed with the Y.
- the structure of a fluorolas anchor By adjusting the number of introductions, the solubility of the intermediate for oligonucleotide synthesis and the purification load can be reduced. This makes it possible to synthesize oligonucleotides by a more versatile method.
- the nucleoside protector in the first embodiment has a structure represented by the following formula (I).
- B is a natural or modified nucleoside base
- R 1 , R 2 are independently H or protecting groups that can be deprotected under acidic, basic or neutral conditions.
- the F-protector is O (CH 2 ) n (CF 2 ) m CF 3 when the protected part of the nucleoside base B is O, and N when the protected part of the nucleoside base B is N.
- Y is H, OH, halogen, OCH 3 , Methoxyethyl, CN, CF 3, or hydroxyl groups protected by acyl-based protecting groups, ether-based protecting groups, silyl-based protecting groups;
- Z is H, alkyl, O-alkyl, N-alkyl, halogen. There is or forms a ZZ bond with the Y.
- ZZ bonds are substituted or unsubstituted C2-C6 alkylene groups, -S-, -CO-, -CS-, -COO-, -OCONR 6- (R 6 is H or C1-C6 alkyl groups. ), -CONR 6- , -CSNR 6- and the like.
- the nucleoside protecting group according to the above formula (I) protects the base by reacting a commercially available fluorous alcohol with the base portion of the nucleoside in which (1) 3', 5'-hydroxyl group is protected, and (2) (1). ),
- the compound can be synthesized by deprotecting the protecting group of the 3', 5'-hydroxyl group.
- the protection of the base portion can be carried out by applying a Mitsunobu reaction or a reaction using benzenesulfonic acid chloride using a commercially available fluorus alcohol.
- a Mitsunobu reaction or a reaction using benzenesulfonic acid chloride using a commercially available fluorus alcohol.
- 1H, 1H, 2H, 2H-nonafluoro-1-hexanol, 1H, 1H, 2H, 2H-tridecafluoro-1-octanol, 1H, 1H-pentadecafluoro-1-octanol and the like are used. be able to.
- nucleoside protectant described in the above formula (I) can be synthesized more easily and at a lower cost than the conventional method.
- Specific examples of the nucleoside derivative into which the F-projector has been introduced include the following compounds.
- the second embodiment is a 5'-terminal protected nucleoside phosphoramidite represented by the following formula (II).
- B is a natural or modified nucleoside base
- R 1 is a deprotectable protecting group under acidic, basic or neutral conditions
- R 4 is a substituted or unsubstituted aliphatic group , Substituted or unsubstituted aromatic group
- Y is H, OH, halogen, OCH 3 , methoxyethyl, CN, CF 3, or a hydroxyl group protected by an acyl protecting group, an ether protecting group, or a silyl protecting group
- Z is H, alkyl. , O-alkyl, N-
- the 3'-phosphoromidite described in the above formula (II) selectively selects the 5'-hydroxyl group of the 3', 5'-unprotected nucleoside represented by the above formula (I) by a known method.
- NCCH 2 CH 2 OP [N (i-C 3 H 7 ) 2 ] 2 , CH 2 CHCH 2 to the 5'-protected 3'-unprotected nucleoside obtained in (2) and (1).
- It can be synthesized by reacting with a trivalent phosphorylating agent such as OP [N (i-C 3 H 7 ) 2 ] 2 .
- Step (2) can be performed by a known method.
- the third embodiment is a fluorolas blocker amidite represented by the following formula (III).
- B is a natural or modified nucleoside base
- R 1 is a deprotectable protecting group under acidic, basic or neutral conditions
- R 3 is a ring of the one of R 5 bonded to the nitrogen atom attached to the phosphorus atom is formed by bond;
- R 5 is an aliphatic substituted or unsubstituted A group, substituted or unsubstituted aromatic group;
- Pro is an unprotected, nucleoside base protecting group or F-projector, and the F-projector is when the protected portion of the nucleoside base B is O.
- Is O (CH 2 ) n (CF 2 ) m CF 3 and is NH (C O) (CH 2 ) n (CF 2 ) m CF 3 when the protected portion of the nucleoside base B is N.
- n is 1 or 2
- m is an integer from 1 to 20
- p is an integer from 0 to 27
- Y is H, OH, halogen, OCH 3 , methoxyethyl, CN, CF 3, Alternatively, it is a hydroxyl group protected with an acyl protecting group, an ether protecting group, or a silyl protecting group
- Z is H, alkyl, O-alkyl, N-alkyl, halogen, or with Y.
- a ZZ bond is formed between them.
- An example of the ZZ bond is the same as that of the compound represented by the above formula (I).
- the fluorolas blocker amidite represented by the above formula (III) has at least one F-projector in either Pro or R 1 , R 3 .
- blockmer is a dimer or more nucleotide, and a longer chain nucleotide is formed by performing a condensation reaction with amidite, a 3'-or 5'-hydroxyl group-unprotected nucleoside, a nucleotide, or the like. It is a nucleotide unit that serves as a synthetic block for the purpose.
- a multi-fluorous blockmer is a blockmer containing a plurality of fluorous tags.
- the fluorus blocker amidite represented by the above formula (III) is represented by (1) the 5'-terminal protected nucleoside phosphoramidite represented by the above formula (II) or the following formula (II').
- An intermediate was synthesized by reacting 5'-terminal protected nucleoside H-phosphonate with a nucleoside in which a fluorus anchor was bound to the 3'-terminal represented by the following formula (V), and (2) the obtained intermediate.
- B is a natural or modified nucleoside base
- R 1 is a protecting group that can be deprotected under acidic, basic or neutral conditions
- n is 1 or 2
- m is an integer from 1 to 20
- Y is H, OH, halogen, OCH 3 , methoxyethyl, A hydroxyl group protected with CN, CF 3, or an acyl-based protecting group, an ether-based protecting group, or a silyl-based protecting group
- Z is H, alkyl, O-alkyl, N-alkyl, halogen, or , A ZZ bond is formed with the Y.
- the fourth embodiment is a multifluorous blocker represented by the following formula (IV).
- B is a natural or modified nucleoside base
- R 1 is a deprotectable protecting group under acidic, basic or neutral conditions
- Pro is an unprotected, protecting group or F-projector of the nucleoside base, and the F-projector is O (CH 2 ) when the protected portion of the nucleoside base B is O.
- a ZZ bond is formed with the Y.
- An example of the ZZ bond is the same as that of the compound represented by the above formula (I).
- the multifluorous blocker represented by the above formula (IV) has at least one F-projector in either Pro or R 1 , R 3 .
- the fifth embodiment is a fluorolas blocker amidite represented by the above formula (III) or a 5'-terminal protected nucleoside H-phosphonate represented by the following formula (II') and a following formula (V). It is a method for synthesizing a multifluorous blocker represented by the above formula (IV), which comprises a step of coupling a nucleoside having a fluorous anchor bonded to the 3'-terminal represented by the above formula (IV).
- B is a natural or modified nucleoside base
- R 1 is a protecting group that can be deprotected under acidic, basic or neutral conditions
- F-projector is a protecting group that can be deprotected under acidic, basic or neutral conditions
- Is O (CH 2 ) n (CF 2 ) m CF 3 when the protected portion of the nucleoside base B is O, and NH (C ) when the protected portion of the nucleoside base B is N.
- Y is H, OH, halogen, OCH 3 , methoxyethyl, A hydroxyl group protected with CN, CF 3, or an acyl-based protecting group, an ether-based protecting group, or a silyl-based protecting group;
- Z is H, alkyl, O-alkyl, N-alkyl, halogen, or , A ZZ bond is formed with the Y.
- the multifluorous blocker in the present embodiment is (1) a 5'-terminal protected nucleoside phosphoramidite represented by the above formula (II) or a 5'-terminal protected represented by the above formula (II').
- An intermediate was synthesized by reacting a nucleoside H-phosphonate with a nucleoside in which a fluorus anchor was bound to the 3'-terminal represented by the above formula (V), and (2) the 3'-terminal of the obtained intermediate.
- the nucleoside anchor bound to the nucleoside was removed to obtain a 3'-unprotected form, and (3) NCCH 2 CH 2 OP [N (i-C) was added to the unprotected 3'-hydroxyl oxide.
- the multifluorous blocker in the above embodiment can also be synthesized by another method described below.
- a multifluorous blockmer having a desired chain length can be obtained by coupling a multifluorous blockmer represented by the above formula (IV) with the 5'-end deprotected in a so-called one-pot. it can.
- the 5'-terminal of the compound obtained by coupling with 5'-protected fluorous blocker amidite and fluorolas blocker H-phosphonate is deprotected, and again with fluorolas blocker amidite.
- the 5'-terminal may be deprotected and subjected to the above-mentioned one-pot coupling reaction.
- the above reaction can also be carried out by using a multifluorous blocker in which the 3'-terminal of the multifluorous blocker is bound to a solid phase carrier instead of a fluorous anchor, and the multifluorous blocker bound to the solid phase carrier is also used.
- a multifluorous blocker in which the 3'-terminal of the multifluorous blocker is bound to a solid phase carrier instead of a fluorous anchor, and the multifluorous blocker bound to the solid phase carrier is also used.
- the oligonucleotide can be obtained by performing a deprotection reaction at each site. can get.
- the multifluorous blocker represented by the above formula (IV) has at least one F-projector in either Pro or R 1 , R 3 .
- the affinity of the multifluorous blocker can be utilized to simplify the separation and purification of intermediates and products as compared with the case of using a widely used protecting group.
- the nucleoside base in the above embodiment includes a natural base such as an adenyl group, a guanyl group, a cytosynyl group, a timyl group and a uracil group, a 5-methylcytosynyl group, a 5-fluorouracil group, a 7-methylguanyl group, a 7-deazaadenyl group and the like.
- a natural base such as an adenyl group, a guanyl group, a cytosynyl group, a timyl group and a uracil group, a 5-methylcytosynyl group, a 5-fluorouracil group, a 7-methylguanyl group, a 7-deazaadenyl group and the like.
- the "modified nucleoside base” in the present specification includes a base having a reactive functional group such as an amino group, a carbonyl group, a hydroxy
- a fluorous protecting group derived from a fluorous alcohol is introduced into the above-mentioned reactive functional groups.
- the protecting group that protects the nucleoside base include an acyl group, a benzoyl group, and an allyloxycarbonyl group.
- Aliphatic group in the above embodiments include saturated or unsaturated, C 1 -C 18 hydrocarbons which are linear or branched, cyclic C 3 -C 18 saturated or unsaturated hydrocarbon.
- the aromatic group in the present embodiment includes a carbocyclic aromatic ring such as a phenyl group, a carbocyclic aromatic ring such as a naphthyl group, or a carbocyclic aromatic ring condensed with a non-carbon aromatic ring.
- Aliphatic group in the present embodiment the aromatic group, substituted saturated or unsaturated, C 1 -C 8 hydrocarbon or C 3 -C 8 cyclic hydrocarbons, halogen, cyano, nitro, with a substituent such as an aromatic ring It may have been.
- What is bonded to the nitrogen atom bonded to the phosphorus atom is preferably a linear or branched alkyl group, a secondary amino group such as pyrrolidine, diethylamine, or morpholino group, and more preferably an isopropyl group. Is.
- one end of the alkyl group bonded to the nitrogen atom bonded to the phosphorus atom may be a ring formed by bonding to the adjacent nitrogen atom.
- a phosphoric acid protecting group that is widely used in oligonucleotide synthesis can be used.
- -CH 2 CH 2 CN, -CH 2 CH CH 2 , -OCH 3, 2-chlorophenyl group, phenyl group, or R ′ and R ′′ bonded to the nitrogen atom bonded to the phosphorus atom. It may be a ring formed by combining one with one.
- the protecting group other than the above include -CH 2 CH 2 E (E is an electron-withdrawing group) and a fluorine-containing protecting group that can be deprotected under basic conditions.
- the 5'-hydroxyl protecting group in this embodiment contains a protecting group that can be removed under acidic, basic or neutral conditions.
- Protecting groups that can be removed under acidic conditions include ether-based protecting groups containing substituted or unsubstituted trityl groups, pixyl groups, substituted or unsubstituted tetrahydropyranyl (THP) groups, and 4 as typical protecting groups. , 4'-dimethoxytrityl group.
- Examples of protecting groups that can be removed under neutral conditions include silyl protecting groups, specifically, trimethylsilyl group, triethylsilyl group, t-butyldimethylsilyl group, t-butyldiphenylsilyl group, and triphenylsilyl group. including.
- Examples of protecting groups that can be removed under basic conditions include Fmoc group and pivaloyl group.
- Examples of the protecting group other than the above include an alkyl group, an acyl group, an acetyl group, a benzoyl group, a benzyl group, an alkoxyalkyl group, a carbamoyl group and the like.
- a trivalent phosphorylating agent (3'-unprotected) is added to a solution of the 3'-unprotected nucleoside (0.1 to 0.4 M). Add 1.05-2.0 eq of nucleoside) and activator (0.4-0.8 eq of 3'-unprotected nucleoside) and stir at room temperature for 10-20 hours. The obtained 3'-phosphoromidite is purified on silica gel.
- the above formula (V) is used.
- an activator that activates the amidite moiety to 3'-phosphoromidite represented by the formula (II) or the formula (III)
- the above formula (V) is used.
- a nucleoside to which a fluorolas anchor is bound By coupling a nucleoside to which a fluorolas anchor is bound to the 3'-terminal to be formed, a multifluorous blocker represented by the formula (IV) can be obtained.
- Representative activators include, but are not limited to, 1H-tetrazole, S-ethylthiotetrazole, dicyanoimidazole, dichloroimidazole, salts of sulfonic acid and azole or tertiary amines.
- the reaction is carried out in a dried solvent such as dichloromethane, acetonitrile, tetrahydrofuran, DMF, toluene and the like.
- blockmer amidite When synthesizing blocker amidite or multifluorous blockmer in the above embodiment, as another method of extending one base at a time, blockmer amidite that is already a dimer or more is 3'-terminal. It is also possible to extend two or more bases at once by condensing with a nucleoside to which a fluorolas anchor is bound.
- the synthesis of phosphoramidite using the blockmer amidite represented by the above formula (III) can be carried out in a solution (hereinafter referred to as "liquid phase synthesis method").
- liquid phase synthesis method After synthesizing the multifluorous blocker by the method described above, it can be easily purified by column chromatography using commercially available silica gel, silica gel modified with octadecyl or diol, or silica gel for solid-phase extraction of fluorous. it can.
- silica gel for solid-phase extraction of fluorolas 40 ⁇ m of Fluoroflash silica gel, which can be purchased from Aldrich, can be mentioned.
- liquid phase synthesis method it can be synthesized on a large scale (10 times to 100 times or more) as compared with the solid phase synthesis method in which a coupling reaction is carried out on a solid phase resin. Therefore, it is possible to synthesize a multifluorous blocker for oligonucleotide synthesis at a lower cost.
- Liquid separation operation using an organic solvent and water or a fluorus solvent and an organic solvent, a fluorus solvent and water, countercurrent chromatography of these two phases, and simple purification by crystallization / powdering are also applicable.
- the number of F-anchors of the present invention to be introduced as a protecting group into the nucleoside base portion of multifluorous blocker is adjusted, in other words, the F-anchor of the present invention is introduced into some nucleoside base portions, and the rest.
- the affinity with silica gel for fluorolas solid-phase extraction can be changed. This allows the multifluorous blocker to remain on the silica gel side for fluorolas solid-phase extraction, making it easier to separate the reaction by-products and excess reagents.
- the solubility in a solvent can be changed even at the intermediate stage of the synthesis of multifluorous blocker
- purification by affinity chromatography or a fluorous solvent-hydrocarbon-based organic solvent is used instead of purification by silica gel chromatography. Purification by distribution of.
- the purification method can be appropriately selected according to the amount to be synthesized and the chain length thereof.
- oligonucleotides using the multifluorous broccyl represented by the above formula (IV) for example, in the stage of multifluorous brocmer synthesis, trivalent phosphoric acid is reacted with a sulfide agent instead of an oxidizing agent.
- a part of the pentavalent phosphoric acid can be thiophosphate. Therefore, by synthesizing a multifluorous blockmer made into thiophosphate in the middle of the blockmer synthesis, it is possible to surely synthesize an oligonucleotide in which thiophosphate is introduced at a target position.
- Example 1 a base fluorine-protected phosphoramidite, which is an example of the compound represented by the above formula (II), was produced. Further, according to the procedure shown in Example 2, a multifluorous blocker, which is an example of the compound represented by the above formula (IV), was produced. Further, according to the procedure shown in Example 3, an example of a blockmer phosphoramidite hexamer phosphoramidite, which is one of the compounds represented by the above formula (III), and a compound represented by the above formula (IV). Oligonucleotide 19-mer was synthesized using the above multifluorous blocker.
- 3', 5'-bis-O-tert-butyldimethylsilylthymidine 1 (2.4 g, 5.0 mmol), which can be synthesized by a known method, was dissolved in tetrahydrofuran (25 mL) to 0 ° C. There, triphenylphosphine (1.4 g, 5.5 mmol), 40% diethylazodicarboxylate / toluene solution (2.5 mL, 5.5 mmol), and 1H, 1H, 2H, 2H-tridecafluoro-1- Octanol 2 (2.0 g, 1.2 mL, 5.5 mmol) was added, and the mixture was stirred for 12 hours.
- Step 2 3', 5'-Protection-Base Fluorous Protection Nucleoside 3', 5'deprotection
- Step 3 3', 5'-Unprotected-Base Fluorous Protected Nucleoside 5'-Protected
- Step 4 5'-Protected-3'-Unprotected-Base Fluorous Protected Nucleoside amidite
- Step 7 5'-Protecting-3'-Five'-deprotection of fluorolas anchor-binding nucleotide dimer
- Step 8 Synthesis of 5'-protected nucleotide 13-mer by condensation of 5'-protected hexamer blocker amidite with 5'-unprotected-3'-fluorous anchor-linked nucleotide 7-mer
- Step 10 5'-Protected -3'-Fluorous Anchor-Binding Nucleotide 19-Medium Synthesis by Condensation of 5'-Protected Hexomer Block Maramidite with 5'-Unprotected-3'-Fluorous Anchor-Binding Nucleotide 13-Mer
- Step 11 Deprotection of 5'-protecting-3'-fluorous anchor-binding nucleotide 19-mer synthesis
- Step 13 3'-deprotection of a 5', 3'-protected nucleotide tetramer containing a base fluorous-protected nucleoside Acetic acid (51.4 ⁇ L, 0.900 mmol) was added to 5', 3'-protected nucleotide tetramer 17 (1.87 g, 0.900 mmol), and the mixture was cooled to 0 ° C. 1.0 M TBAF in THF (1.80 mL, 1.80 mmol) was added dropwise and the mixture was stirred for 3 hours.
- Step 14 3'-Amiditeization of a 5'-protected nucleotide tetramer containing a base fluorolas-protected nucleoside Compound 18 (1.27 g, 0.65 mmol) was dissolved in a dichloromethane-acetonitrile 1: 1 mixture (16 mL), followed by 1H-tetrazole (59.0 mg, 0.845 mmol) and N-methylimidazole (21.0 ⁇ L, 0. 260 mmol) was added to bring the temperature to 0 ° C.
- Step 15 Containing a base fluorus-protected nucleoside by condensation of a 5'-protected nucleotide tetramer 3'-amidite containing a base fluorus-protected nucleoside with a 5'-unprotected-3'-protected nucleotide trimer 5 ⁇ , 3 ′-Protected nucleotide nucleoside synthesis
- Compound 16 0.537 g, 0.25 mmol
- tetrameric amidite 19 0.288 g, 0.275 mmol
- Step 16 5'-deprotection of a 5', 3'-protected nucleotide dimer containing a base fluorous-protected nucleoside Dichloromethane (4 mL) was added to compound 20 (0.655 g, 0.211 mmol), and the mixture was cooled to 0 ° C.
- Step 17 Base by condensation of 5'-protected nucleotide tetramer 3'-amidite containing basic fluorous-protected nucleoside and 5'-unprotected-3'-protected nucleotide nucleoside containing base fluoro-protected nucleoside.
- a nucleotide 11-mer from which a protecting group has been removed can be obtained by deprotecting the obtained 5', 3'-protected nucleotide 11-mer 22 containing the obtained base fluorine-protected nucleoside by a general-purpose method. it can.
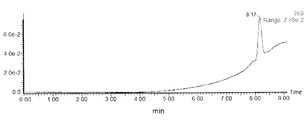
- FIG. (A) is the spectrum of the dTA 19 20mer synthesized by the conventional method
- (b) is the spectrum of the dTA 19 20mer having the fluorous tag of the present invention at the base portion of the 5'-terminal T.
- the 20-mer was obtained with a higher purity than the 20-mer synthesized by the conventional method by deprotecting the product so as to leave the fluorous tag and subjecting it to purification. It was shown that due to the high lipophilicity of fluorous tags, the retention time for separation and purification changes significantly, which makes isolation and purification easier than with conventional methods.
- the multifluorous blocker of the present embodiment can be synthesized by introducing a fluorous anchor using a commercially available fluorocarbon derivative as it is.
- the introduction of fluorolas tags into nucleosides by the conventional method has a limit on the number of fluorolas tags that can be introduced, and is not highly versatile.
- the solubility of the intermediate in the oligonucleotide synthesis and the purification load can be reduced by adjusting the number of introduced fluorous anchors.
- the fluorous tag can be introduced into the nucleoside base moiety or the protecting group at the 3'-terminal of the nucleoside, depending on the length and sequence of the blockmer or oligonucleotide to be synthesized.
- a pentavalent phosphate-bonded moiety that has been oxidized / sulfurized at the blockmer stage can be formed. Therefore, even when only a part of the phosphate bond in the oligonucleotide is in an oxidation / sulfide state different from that of the other part of the phosphate bond, the object is more easily and easily without changing the procedure of oligonucleotide synthesis. Oligonucleotides containing modified phosphate binding moieties can be synthesized.
- the blockmer into which the fluorolas tag used so far has been introduced is affected by the physical properties of the fluorolas tag, so that the purification method at the time of chain length extension in oligonucleotide synthesis has become complicated.
- the method for synthesizing multifluorous bromine and the method for synthesizing an oligonucleotide using multifluorous bromar are silica gel filtration, purification by affinity chromatography, and fluorous solvent-hydrocarbon-based organic solvent, depending on the chain length. There is a great deal of freedom in selecting the purification method, such as the distribution of silica gel. Therefore, the desired oligonucleotide can be obtained by simpler purification than the conventional method for synthesizing an oligonucleotide using a fluorous tag.
- the oligonucleotide synthesis method using the multifluorous blocker of the present embodiment when synthesizing the oligonucleotide of the same N-mer, the method of extending one base at a time by the commonly used liquid phase synthesis method is used. In comparison, the number of steps required can be reduced. Therefore, the yield of the oligonucleotide of the desired length can be improved.
Abstract
Description
本発明の第1の態様は、下記式(I)で表されるヌクレオシド保護体であって、
ステップ12:5´-保護-塩基部フルオラス保護ヌクレオシド-3´-アミダイトと5´-無保護-3´-保護-ヌクレオチド3量体との縮合による、塩基部フルオラス保護ヌクレオシドを含む5´,3´-保護ヌクレオチド4量体合成
得られた塩基部フルオラス保護ヌクレオシドを含む5´,3´-保護ヌクレオチド11量体22に対して汎用される方法で脱保護を行うことで、保護基を除去したヌクレオチド11量体を得ることができる。
Claims (5)
- 下記式(I)で表されるヌクレオシド保護体であって、
- 下記式(II)で表される5´-末端保護ヌクレオシドホスホロアミダイトであって、
- 下記式(III)で表されるフルオラスブロックマーアミダイトであって、
- 下記式(IV)で表されるマルチフルオラスブロックマーであって、
- 請求項3に記載の上記式(III)で表されるフルオラスブロックマーアミダイトまたは、下記式(II´)で表される、5´-末端保護ヌクレオシドH-ホスホネートと、下記式(V)で表される3´-末端にフルオラスアンカーが結合したヌクレオシドとをカップリング反応させる工程を含む、請求項4に記載の下記式(VI)で表されるマルチフルオラスブロックマーの合成方法であって、
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202080037793.8A CN113924309A (zh) | 2019-05-21 | 2020-05-21 | 寡核苷酸合成中使用的多氟嵌段聚体以及使用其的寡核苷酸合成方法 |
| EP20809071.2A EP3960749A4 (en) | 2019-05-21 | 2020-05-21 | MULTIFLUORINE BLOCKERS FOR OLIGONUCLEOTIDE SYNTHESIS AND METHODS OF OLIGONUCLEOTIDE SYNTHESIS USING THE SAME |
| US17/595,613 US20220235089A1 (en) | 2019-05-21 | 2020-05-21 | Multi-Fluorous Blockmer for Oligonucleotide Synthesis, and Oligonucleotide Synthesis Method Using the Same |
| JP2021520862A JP7393807B2 (ja) | 2019-05-21 | 2020-05-21 | オリゴヌクレオチド合成に用いるマルチフルオラスブロックマーおよびこれを用いたオリゴヌクレオチド合成方法 |
| KR1020217038369A KR20220002469A (ko) | 2019-05-21 | 2020-05-21 | 올리고뉴클레오티드 합성에 사용하는 멀티플루오로스블록머 및 이를 사용한 올리고뉴클레오티드 합성방법 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019-095591 | 2019-05-21 | ||
| JP2019095591 | 2019-05-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2020235658A1 true WO2020235658A1 (ja) | 2020-11-26 |
Family
ID=73458301
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2020/020203 WO2020235658A1 (ja) | 2019-05-21 | 2020-05-21 | オリゴヌクレオチド合成に用いるマルチフルオラスブロックマーおよびこれを用いたオリゴヌクレオチド合成方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20220235089A1 (ja) |
| EP (1) | EP3960749A4 (ja) |
| JP (1) | JP7393807B2 (ja) |
| KR (1) | KR20220002469A (ja) |
| CN (1) | CN113924309A (ja) |
| WO (1) | WO2020235658A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022172994A1 (ja) | 2021-02-12 | 2022-08-18 | 味の素株式会社 | オリゴヌクレオチドの製造方法 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20210317158A1 (en) * | 2018-05-02 | 2021-10-14 | Natias Inc. | Optically active segment for use in synthesis of stereocontrolled oligonucleotide, method for producing the same, and method for synthesizing stereocontrolled oligonucleotide using the same |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005070859A1 (ja) | 2004-01-27 | 2005-08-04 | Takeshi Wada | フルオラス担体およびそれを用いたオリゴヌクレオチド誘導体の製造方法 |
| JP2008516938A (ja) * | 2004-10-15 | 2008-05-22 | アストラゼネカ アクチボラグ | 置換アデニンとその使用 |
| JP2009541438A (ja) * | 2006-06-27 | 2009-11-26 | ビオヴィトルム・アクチボラゲット(プブリクト) | 治療用化合物 |
| JP2014510743A (ja) * | 2011-03-31 | 2014-05-01 | シェファー、コンスタンツェ | 核酸の非ウイルスの導入のためのパーフルオロ化化合物 |
| WO2017086397A1 (ja) | 2015-11-17 | 2017-05-26 | 日産化学工業株式会社 | オリゴヌクレオチドの製造方法 |
| WO2019042888A1 (de) * | 2017-08-27 | 2019-03-07 | Caperis Gmbh | Perfluorierte nukleinsäuren als tenside mit spezifischen eigenschaften |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060178507A1 (en) * | 2004-12-30 | 2006-08-10 | Berry & Associates, Inc. | Fluorous oligonucleotide reagents and affinity purification of oligonucleotides |
| JP7431723B2 (ja) * | 2017-08-18 | 2024-02-15 | アジレント・テクノロジーズ・インク | オリゴヌクレオチドのアフィニティー精製のためのオルトエステル組成物 |
-
2020
- 2020-05-21 EP EP20809071.2A patent/EP3960749A4/en active Pending
- 2020-05-21 US US17/595,613 patent/US20220235089A1/en active Pending
- 2020-05-21 WO PCT/JP2020/020203 patent/WO2020235658A1/ja unknown
- 2020-05-21 CN CN202080037793.8A patent/CN113924309A/zh active Pending
- 2020-05-21 JP JP2021520862A patent/JP7393807B2/ja active Active
- 2020-05-21 KR KR1020217038369A patent/KR20220002469A/ko not_active Application Discontinuation
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005070859A1 (ja) | 2004-01-27 | 2005-08-04 | Takeshi Wada | フルオラス担体およびそれを用いたオリゴヌクレオチド誘導体の製造方法 |
| JP2008516938A (ja) * | 2004-10-15 | 2008-05-22 | アストラゼネカ アクチボラグ | 置換アデニンとその使用 |
| JP2009541438A (ja) * | 2006-06-27 | 2009-11-26 | ビオヴィトルム・アクチボラゲット(プブリクト) | 治療用化合物 |
| JP2014510743A (ja) * | 2011-03-31 | 2014-05-01 | シェファー、コンスタンツェ | 核酸の非ウイルスの導入のためのパーフルオロ化化合物 |
| WO2017086397A1 (ja) | 2015-11-17 | 2017-05-26 | 日産化学工業株式会社 | オリゴヌクレオチドの製造方法 |
| WO2019042888A1 (de) * | 2017-08-27 | 2019-03-07 | Caperis Gmbh | Perfluorierte nukleinsäuren als tenside mit spezifischen eigenschaften |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3960749A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022172994A1 (ja) | 2021-02-12 | 2022-08-18 | 味の素株式会社 | オリゴヌクレオチドの製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP7393807B2 (ja) | 2023-12-07 |
| CN113924309A (zh) | 2022-01-11 |
| KR20220002469A (ko) | 2022-01-06 |
| EP3960749A1 (en) | 2022-03-02 |
| EP3960749A4 (en) | 2023-02-22 |
| US20220235089A1 (en) | 2022-07-28 |
| JPWO2020235658A1 (ja) | 2020-11-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2753607B1 (en) | Bicyclo[6.1.0]non-4-yne reagents for chemical modification of oligonucleotides | |
| JP2552048B2 (ja) | ヌクレオチド鎖合成用試薬 | |
| JP7075680B2 (ja) | オリゴヌクレオチド合成用セグメントおよびその製造方法、ならびにそれを用いたオリゴヌクレオチドの合成方法 | |
| JP5698206B2 (ja) | ローダミン標識されたオリゴヌクレオチドを合成するために有用な試薬 | |
| EP2921499B1 (en) | Method for liquid-phase synthesis of nucleic acids | |
| JP3188243B2 (ja) | 5’−修飾オリゴヌクレオチド | |
| US20090012279A1 (en) | Polynucleotide labeling reagent | |
| CN112533892B (zh) | 烷氧基苯基衍生物、核苷保护体和核苷酸保护体、寡核苷酸制造方法以及取代基除去方法 | |
| JPH01301691A (ja) | 新規ヌクレオチド誘導体 | |
| WO2020235658A1 (ja) | オリゴヌクレオチド合成に用いるマルチフルオラスブロックマーおよびこれを用いたオリゴヌクレオチド合成方法 | |
| US5623068A (en) | Synthesis of DNA using substituted phenylacetyl-protected nucleotides | |
| KR20030032924A (ko) | 포스포로티오에이트 트리에스테르의 제조 방법 | |
| JP2794461B2 (ja) | ホスホアミダイト化合物及びそれを用いたオリゴリボヌクレオチドの固相合成法 | |
| JP2024009042A (ja) | 核酸化合物の製造方法、及び、核酸化合物 | |
| JPWO2020235658A5 (ja) | ||
| WO2020158687A1 (ja) | 光応答性ヌクレオチドアナログの製造方法 | |
| JPWO2021039935A5 (ja) | ||
| JP7229539B2 (ja) | 高脂溶性ホスホラミダイトの製造 | |
| JP7075681B2 (ja) | 立体制御オリゴヌクレオチド合成用光学活性セグメントおよびその製造方法、ならびにそれを用いた立体制御オリゴヌクレオチドの合成方法 | |
| JPS6251695A (ja) | ホスホロアミダイト類の合成法 | |
| WO2021080021A1 (ja) | オリゴヌクレオチドを製造する方法 | |
| PL221806B1 (pl) | Sposób wprowadzania acetalowych i acetaloestrowych grup ochronnych oraz związki do realizacji tego sposobu | |
| JP2003292499A (ja) | オリゴヌクレオチドの化学的合成法 | |
| Cramer et al. | Nucleotides, Part LX, Synthesis and Characterization of New 2′‐O‐Methylriboside 3′‐O‐Phosphoramidites Useful for the Solid‐Phase Synthesis of 2′‐O‐Methyloligoribonucleotides | |
| PL222577B1 (pl) | Sposób ochrony funkcji hydroksylowych lub aminowych nowe związki do realizacji tego sposobu oraz sposób otrzymywania nowych związków |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20809071 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2021520862 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20217038369 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2020809071 Country of ref document: EP Effective date: 20211122 |