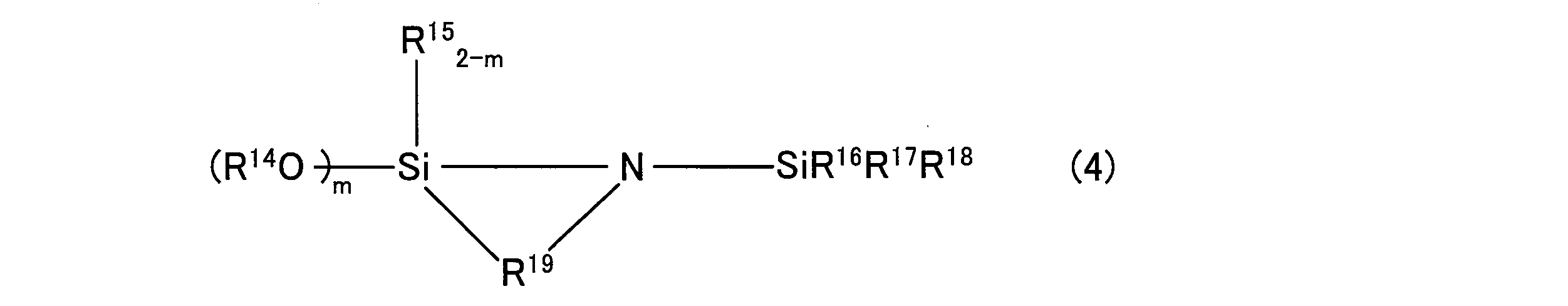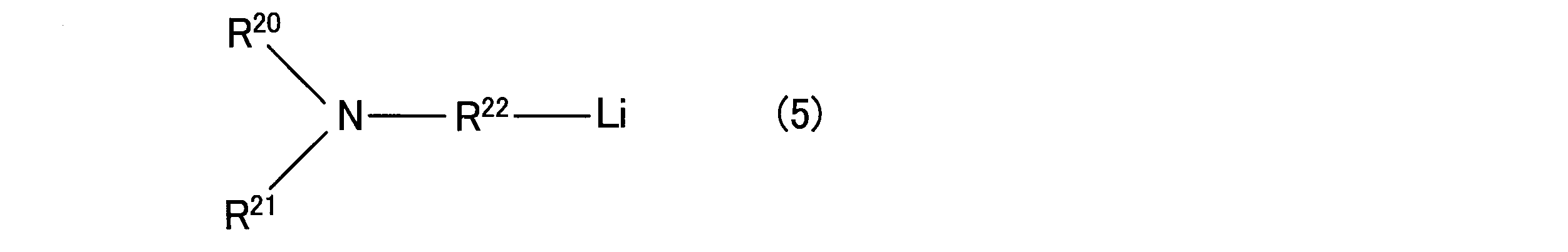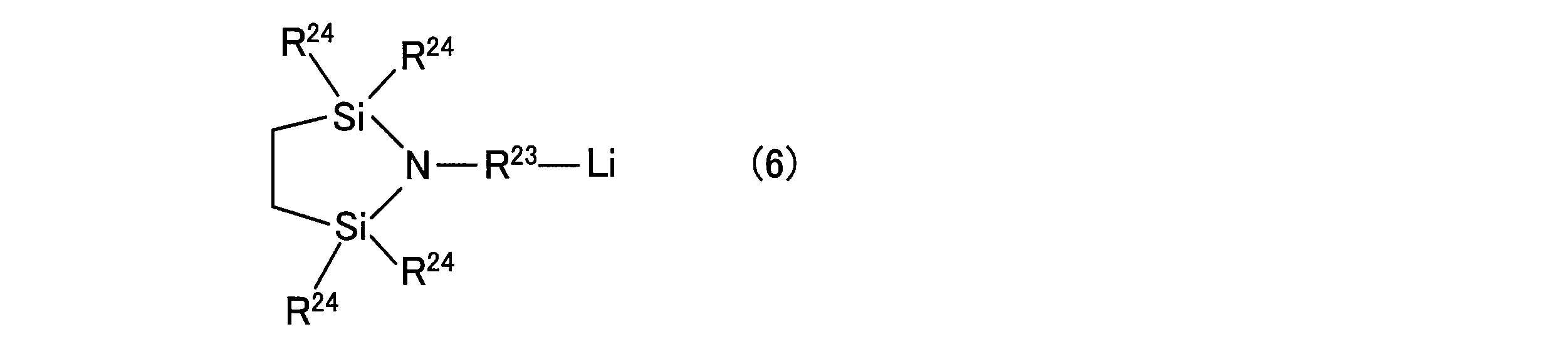WO2017038528A1 - 組成物及び成形体 - Google Patents
組成物及び成形体 Download PDFInfo
- Publication number
- WO2017038528A1 WO2017038528A1 PCT/JP2016/074368 JP2016074368W WO2017038528A1 WO 2017038528 A1 WO2017038528 A1 WO 2017038528A1 JP 2016074368 W JP2016074368 W JP 2016074368W WO 2017038528 A1 WO2017038528 A1 WO 2017038528A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- component
- fiber
- mass
- polymer
- parts
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/04—Reinforcing macromolecular compounds with loose or coherent fibrous material
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/04—Reinforcing macromolecular compounds with loose or coherent fibrous material
- C08J5/0405—Reinforcing macromolecular compounds with loose or coherent fibrous material with inorganic fibres
- C08J5/042—Reinforcing macromolecular compounds with loose or coherent fibrous material with inorganic fibres with carbon fibres
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/04—Reinforcing macromolecular compounds with loose or coherent fibrous material
- C08J5/06—Reinforcing macromolecular compounds with loose or coherent fibrous material using pretreated fibrous materials
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K9/00—Use of pretreated ingredients
- C08K9/08—Ingredients agglomerated by treatment with a binding agent
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L21/00—Compositions of unspecified rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L23/00—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers
- C08L23/02—Compositions of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L23/10—Homopolymers or copolymers of propene
- C08L23/12—Polypropene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2300/00—Characterised by the use of unspecified polymers
- C08J2300/22—Thermoplastic resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2323/00—Characterised by the use of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Derivatives of such polymers
- C08J2323/02—Characterised by the use of homopolymers or copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond; Derivatives of such polymers not modified by chemical after treatment
- C08J2323/10—Homopolymers or copolymers of propene
- C08J2323/12—Polypropene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2347/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, at least one having two or more carbon-to-carbon double bonds; Derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2400/00—Characterised by the use of unspecified polymers
- C08J2400/10—Polymers characterised by the presence of specified groups, e.g. terminal or pendant functional groups
- C08J2400/106—Polymers characterised by the presence of specified groups, e.g. terminal or pendant functional groups containing nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2453/00—Characterised by the use of block copolymers containing at least one sequence of a polymer obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/02—Elements
- C08K3/04—Carbon
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K7/00—Use of ingredients characterised by shape
- C08K7/02—Fibres or whiskers
- C08K7/04—Fibres or whiskers inorganic
- C08K7/06—Elements
Definitions
- the present invention relates to a composition containing fibers and a molded body obtained by molding the composition.
- Fiber reinforced resin is a material in which reinforcing fibers such as glass fibers and carbon fibers are hardened with a resin, and is a composite material having excellent mechanical strength, heat resistance, moldability, and the like. Therefore, FRP is used as a material in a wide range of fields such as aviation, space use, vehicle use, building material use, and sports use.
- CFRP carbon fiber reinforced resin
- FRP using a thermoplastic resin has attracted attention in recent years because it has a feature that a molding cycle can be shortened in addition to the above characteristics.
- thermoplastic resin In FRP using such a thermoplastic resin, continuous fibers are used as reinforcing fibers in order to obtain sufficient mechanical strength (see, for example, Patent Documents 1 and 2).
- carbon long fiber reinforced resin pellets obtained by impregnating a thermoplastic resin while drawing the carbon long fiber under tension to obtain a fiber reinforced resin rod (strand) and then cutting it into an arbitrary length are obtained. It is used (for example, refer patent document 3).
- a method for producing CFRP by impregnating a thermoplastic resin with a mat (nonwoven fabric or the like) made of fibers has been studied (for example, see Patent Document 4).
- FRP molded bodies using continuous fibers or nonwoven fabrics as reinforcing fibers generally have higher impact resistance and bending strength than short fiber composite materials.
- CFRP produced by the above method may have insufficient adhesion between the continuous fiber or nonwoven fabric and the matrix resin, and may be insufficient in terms of improving mechanical properties such as bending strength. It was. For this reason, the CFRP manufactured by the above method sometimes cracks from the interface between the carbon fiber and the matrix resin when a load such as a bending load is applied. The cracks generated in this way propagate to the other interface between the continuous fiber and the matrix resin, thereby causing further cracks and finally traversing the molded body, leading to total destruction.
- some aspects according to the present invention are excellent in adhesion between the fiber and the matrix resin, and in mechanical strength such as impact resistance and bending strength, by solving at least a part of the problems.
- a composition from which a molded body is obtained is obtained.
- the present invention has been made to solve at least a part of the above-described problems, and can be realized as the following aspects or application examples.
- composition according to the present invention is: Containing an amino group-containing polymer (A), fiber (B), and thermoplastic resin (C);
- the fiber (B) is contained in an amount of 70 parts by mass to 250 parts by mass with respect to 100 parts by mass of the thermoplastic resin (C).
- the content rate of the polymer (A) which has the said amino group can be 0.1 mass part or more and 10 mass parts or less with respect to 100 mass parts of said thermoplastic resins (C).
- the fiber (B) may be a carbon fiber.
- the polymer (A) having an amino group may be a conjugated diene polymer.
- the fiber length of the said fiber (B) can be 50 mm or more.
- composition according to the present invention is: A polymer (A) having an amino group; Non-woven fabric (B ′), A thermoplastic resin (C); Can be contained.
- the said nonwoven fabric (B ') can contain a recycled fiber.
- the nonwoven fabric (B ′) can be contained in an amount of 50 to 150 parts by mass with respect to 100 parts by mass of the thermoplastic resin (C).
- the nonwoven fabric (B ′) may contain carbon fibers.
- the polymer (A) having the amino group can be contained in an amount of 0.1 parts by mass or more and 10 parts by mass or less with respect to 100 parts by mass of the thermoplastic resin (C).
- thermoplastic resin (C) may be an olefin resin.
- One aspect of the molded body according to the present invention is: It is produced using the composition of any one of the application examples 1 to 11.
- composition of the present invention it is possible to produce a molded article having excellent adhesion between the fiber and the matrix resin and excellent mechanical strength such as impact resistance and bending strength.
- the polymer (A) having an amino group is referred to as “component (A)”, the fiber (B) as “component (B)”, and the nonwoven fabric (B ′) as “component (B ′)”.
- the thermoplastic resin (C) is sometimes abbreviated as “(C) component”.
- one aspect of the composition according to this embodiment includes a polymer (A) having an amino group, a fiber (B), and a thermoplastic resin (C), and is a matrix resin.
- a configuration is adopted in which the component (B) is contained in an amount of 70 parts by weight to 250 parts by weight with respect to 100 parts by weight of the component (C).
- one aspect of the composition according to the present embodiment employs a configuration in which the polymer (A) having an amino group, the nonwoven fabric (B ′), and the thermoplastic resin (C) are contained.
- each component contained in the composition according to the present embodiment will be described.
- the composition according to this embodiment includes a polymer (A) having an amino group.
- the component (A) is obtained when a load such as a bending load is applied by firmly bonding the component (B) or the component (B ′) and the component (C) in the molded body according to the present embodiment ( It is considered that the occurrence of cracks from the B) component or the interface between the (B ′) component and the (C) component is suppressed, and the mechanical strength such as the bending strength and falling weight impact strength of the molded body is improved.
- the component (A) used in the present embodiment has an amino group.
- the method for imparting an amino group to a polymer is not particularly limited, such as a method for imparting an amino group to a polymer with a modifier having an amino group, a method for obtaining a component (A) by polymerizing a monomer having an amino group, etc. Is exemplified. Moreover, it is good also as a structure into which the amino group was introduce
- the component (A) may have an amino group as a functional group, but may have an alkoxysilyl group.
- amino group means a primary amino group (—NH 2 ), a secondary amino group (—NHR, where R is a hydrocarbon group), and a tertiary amino group (—NRR ′, where R , R ′ represents any one of hydrocarbon groups), and the amino group may be protected by a protecting group.
- hydrocarbon groups an alkyl group is preferable, an alkyl group having 1 to 8 carbon atoms is more preferable, and an alkyl group having 1 to 6 carbon atoms is particularly preferable.
- alkyl groups examples include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, pentyl, heptyl, hexyl, 2,2- Examples thereof include dimethylbutyl group, 2,3-dimethylbutyl group, 2-ethylbutyl group, 2-methylpentyl group, 3-methylpentyl group, heptyl group, octyl group, 2-ethylhexyl group and the like.
- the amount of amino groups per molecular chain of component (A) is preferably 1 or more, more preferably 5 or more, and particularly preferably 10 or more. In order to prevent gelation of the polymer during production, the amount of amino groups per molecular chain is preferably 100 or less, more preferably 50 or less.
- the determination method of the amount of amino groups in a component is not specifically limited, It can obtain
- the weight average molecular weight (Mw) of the component (A) in terms of polystyrene in the gel permeation chromatography (GPC) method is preferably 30,000 to 2,000,000, more preferably 40,000 to 1,000,000, particularly preferably 50,000 to 50. It is ten thousand.
- the melt flow rate (MFR: 230 ° C., 2.16 kg) of component (A) measured in accordance with JIS K7210 is preferably 0.1 to 100 g / 10 min, more preferably 0.2 to 50 g. / 10 min, particularly preferably 0.3 to 30 g / 10 min.
- the component (A) is preferably a conjugated diene polymer having a repeating unit derived from a conjugated diene.
- the component (A) may have a repeating unit derived from a monomer other than the conjugated diene as necessary.
- the component (A) may be a block type polymer in which the same monomer forms a repeating unit, or may be a random type polymer in which different monomers are randomly polymerized. .
- the component (A) It is preferably a coalescence.
- the component (A) is preferably a hydrogenated polymer.
- the structural component of the polymer (A) which has an amino group is explained in full detail.
- the conjugated diene (A) component preferably contains a repeating unit derived from a conjugated diene.
- the conjugated diene include 1,3-butadiene, isoprene, 2,3-dimethyl-1,3-butadiene, 1,3-pentadiene, 2-methyl-1,3-octadiene, 1,3-hexadiene, 1, Examples include 3-cyclohexadiene, 4,5-diethyl-1,3-octadiene, 3-butyl-1,3-octadiene, myrcene, farnesene and chloroprene.
- the conjugated diene preferably contains 1,3-butadiene or isoprene.
- the component may contain repeating units derived from compounds other than conjugated dienes.
- an aromatic alkenyl compound is preferable.
- aromatic alkenyl compounds an unsaturated monomer represented by the following general formula (1) is more preferable in order to produce a molded article having excellent mechanical strength, heat resistance, and cold resistance.
- R 1 is a single bond or a divalent hydrocarbon group having 1 to 3 carbon atoms
- R 3 and R 4 are each independently an alkyl group having 1 to 3 carbon atoms, or 3 to 18 carbon atoms. Or any one of the above trialkylsilyl groups, the other is an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 20 carbon atoms, an aralkyl group having 7 to 20 carbon atoms, or 1 to 100 is an organosiloxy group
- R 2 is a single bond, an alkylene group or alkylidene group having 1 to 20 carbon atoms
- R 5 is a hydrogen atom or a methyl group
- n is an integer of 0 to 3.
- aromatic alkenyl compound examples include styrene, tert-butylstyrene, ⁇ -methylstyrene, p-methylstyrene, p-ethylstyrene, divinylbenzene, 1,1-diphenylstyrene, 1-vinylnaphthalene and 2-vinyl.
- the compound other than the conjugated diene contains styrene or p-methylstyrene.
- the component (A) has a repeating unit derived from a conjugated diene and a repeating unit derived from an aromatic alkenyl compound
- the repeating unit derived from the conjugated diene of the component (A) and a repeating unit derived from an aromatic alkenyl compound The mass ratio is preferably 100: 0 to 20:80 in order to keep the glass transition point of the component (A) moderate and to improve the mechanical strength and cold resistance of the molded body, and 97: 3 More preferably, it is ⁇ 40: 60.
- the component (A) is preferably a block copolymer in order to enhance compatibility with the component (C) and to more firmly bond the component (B) and the component (C).
- a block polymer including two or more polymer blocks selected from the following polymer blocks A to D is more preferable.
- a block A polymer block having a repeating unit amount derived from an aromatic alkenyl compound of 80% by mass or more.
- B block A polymer block having a repeating unit amount derived from a conjugated diene of 80% by mass or more and a vinyl bond content of less than 30 mol%.
- C block A polymer block having a repeating unit amount derived from a conjugated diene of 80% by mass or more and a vinyl bond content of 30 mol% or more and 90 mol% or less.
- D block a random copolymer block of a repeat derived from a conjugated diene and a repeat derived from an aromatic alkenyl compound, and a polymer block other than the above A to C.
- the component (A) contains the C block
- the molecular entanglement and compatibility with the olefin resin that is a kind of the component (C) are improved, so that the mechanical strength of the molded body is further improved.
- the vinyl bond content of the C block is more preferably 50 mol% or more and 90 mol% or less, and particularly preferably 60 mol% or more and 90 mol% or less.
- the C block is hydrogenated.
- the content of the repeating unit derived from a random type or an aromatic alkenyl compound or a repeating unit derived from a conjugated diene is the polymer block.
- a taper type that continuously changes may be used.
- the “vinyl bond content” in the present invention refers to a repeating unit derived from a conjugated diene that is incorporated in the polymer before hydrogenation in 1, 2 bond, 3, 4 bond, and 1, 4 bond bonding modes. Of these, the total proportion of units incorporated in 1, 2 bonds and 3, 4 bonds (based on mol%).
- the vinyl bond content (1,2 bond content and 3,4 bond content) can be calculated by an infrared absorption spectrum method (Morero method).
- Examples of the block polymer containing two or more polymer blocks selected from the polymer blocks A to D include, for example, AB, AC, AD, BC, BD [AB] xY, [AC] xY, [AD] xY, [BC] xY, [BD] xY, [BA ] XY, [CA] xY, [DA] xY, ABDD, ABAA, ACAA, ABC, AD -A, B-A-B, [A-B-D] x-Y, [A-B-A] x-Y, [A-C-A] x-Y, [A-C-B] x -Y, [ADA] x-Y, [BAB] x-Y, ABAB, BABA, [ABAB] xY, ABABABA, [ABABABA] xY, BABB, BABA, BACA -A, BACB, -ADA, [CABAD] xY, [CABAA] xY, [
- a structure surrounded by square brackets and having Y indicates that the block closest to Y is directly coupled to Y.
- [ACB] xY indicates that x [ACB] are directly bonded to Y by the polymer block B.
- composition according to this embodiment is formed into a pellet shape, it is preferable that at least one of A block and B block is included as a block component outside the conjugated diene block copolymer.
- ACA and A- Preferably, it has a structure of CB, [BC] xY, [AC] xY, [AD] xY, ADA.
- a block polymer having a C block or a D block is preferable in that, when an olefin resin is used as the component (C), the compatibility with the resin is excellent, so that a good interface reinforcing effect can be obtained.
- the linking group Y is a structural unit derived from a coupling agent.
- a coupling agent include halogen compounds such as methyldichlorosilane, methyltrichlorosilane, butyltrichlorosilane, tetrachlorosilane, dibromoethane, tetrachlorotin, butyltrichlorotin, tetrachlorogermanium, and bis (trichlorosilyl) ethane.
- An epoxy compound such as epoxidized soybean oil; a carbonyl compound such as diethyl adipate, dimethyl adipate, dimethyl terephthalic acid and diethyl terephthalic acid; a polyvinyl compound such as divinylbenzene; and a polyisocyanate.
- a coupling agent can be used individually by 1 type or in combination of 2 or more types.
- the reaction temperature is preferably 0 to 120 ° C, more preferably 50 to 100 ° C.
- the reaction time is preferably 1 to 30 minutes, more preferably 5 to 20 minutes.
- the component (A) is preferably a hydrogenated polymer (hereinafter also referred to as “hydrogenation”).
- hydrogenation a hydrogenated polymer
- the component (C) by using a hydrogenated polymer as the component (A), the molecular entanglement and compatibility between the component (A) and the olefin resin are remarkable.
- the adhesion between the component (C) and the component (B) is improved.
- the hydrogenation rate of the polymer (hereinafter also referred to as “hydrogenation rate”) can be controlled by changing the amount of the hydrogenation catalyst, the hydrogen pressure during the hydrogenation reaction, or the reaction time.
- the reaction is carried out in the presence of a hydrogenation catalyst at 20 to 150 ° C. under hydrogen pressure of 0.1 to 10 MPa.
- the hydrogenation rate is preferably 60% or more of double bonds such as vinyl bonds, more preferably 80% or more, and particularly preferably 95% or more. Hydrogenation may be performed after the polymerization reaction and then the modifier may be reacted, or the modifier may be reacted after the polymerization reaction and then hydrogenated.
- Examples of the hydrogenation catalyst and a specific hydrogenation method include, for example, JP-A-1-275605, JP-A-5-271326, JP-A-5-271325, JP-A-5-222115, and JP-A-11. -292924, JP-A 2000-37632, JP-A 59-133203, JP-A 63-5401, JP-A 62-218403, JP 7-90017, JP Examples thereof include hydrogenation catalysts and hydrogenation methods described in JP-A Nos. 43-19960 and 47-40473.
- the weight average molecular weight (Mw) of the hydrogenated polymer is preferably 30,000 to 2,000,000, more preferably 40,000 to 1,000,000, and particularly preferably 50,000 to 500,000.
- Mw weight average molecular weight
- the “weight average molecular weight” is a polystyrene equivalent weight average molecular weight measured by gel permeation chromatography (GPC).
- the component (A) can be produced according to the method described in, for example, Japanese Patent No. 5402112, Japanese Patent No. 4840140, International Publication No. 2003/029299, and the like. Moreover, when (A) component is a block polymer, it manufactures according to the method as described in patent 3134504 gazette, patent 3360411 gazette, patent 3988495 gazette, international publication 2014/014052, etc., for example. Can do. Specific examples of the method for producing the component (A) include the following methods (a) to (c). In addition, the polymer obtained by the following method can be hydrogenated by the above-mentioned method as needed.
- a conjugated diene alone or a conjugated diene and an aromatic alkenyl compound are polymerized in the presence of at least one of an organic alkali metal compound and an organic alkaline earth metal compound. This is a method in which a denaturing agent is reacted with the coalescence. Hydrogenation may be performed as necessary.
- the modifier is a compound having an alkoxysilyl group and an amino group, it can be introduced at a high introduction rate.
- the temperature at which the modifier is reacted is preferably 0 to 120 ° C.
- the reaction time is preferably 1 to 30 minutes.
- the amount of the modifier to be reacted with the polymer is preferably 10 mol% or more.
- Examples of the compound having an amino group include compounds represented by the following general formula (2), the following general formula (3), or the following general formula (4).
- R 6 (4-mn) Si (OR 7 ) m X n
- R 6 is an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 20 carbon atoms, an aralkyl group having 7 to 20 carbon atoms, or an organosiloxy group having 1 to 100 carbon atoms. If R 6 is more, each of R 6 may be either different groups in the same group.
- R 7 is an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 20 carbon atoms, or an aralkyl group having 7 to 20 carbon atoms. If R 7 have multiple, each R 7 may be either different groups in the same group.
- X is a group represented by the formula -A-X '(wherein A is an alkylene group having 1 to 20 carbon atoms, and X' is a group containing at least an N atom). When there are a plurality of X, each X may be the same group or different groups. Each X may be an independent substituent or may form a cyclic structure. m and n are integers of 1 to 3. The sum of m and n is an integer of 2 to 4.
- N N-bis (trimethylsilyl) aminopropyltrimethoxysilane
- N N-bis (trimethylsilyl) aminopropylmethyldimethoxysilane
- N N-bis (trimethylsilyl) aminopropylmethyldiethoxysilane
- N N -Bis (trimethylsilyl) aminopropyldimethylmethoxysilane
- 1- 3-triethoxysilylpropyl)- 2,2,5,5-tetramethyl-1-aza-2,5-disilacyclopentane
- 3-morpholinopropyldimethylmethoxysilane 3- (4-
- R 8 to R 11 each independently represents an alkyl group or aryl group having 1 to 20 carbon atoms
- R 12 and R 13 represent an alkylene group having 1 to 20 carbon atoms
- M is an integer of 1 or 2
- n is an integer of 1 to 3.
- the total number of alkoxy groups having 1 to 20 carbon atoms is preferably 4 or more.
- R 14 and R 15 each independently represents an alkyl group or an aryl group having 1 to 20 carbon atoms
- R 19 represents an alkylene group having 1 to 20 carbon atoms
- R 16 , R 17 and R 18 are each independently an alkyl group or aryl group having 1 to 20 carbon atoms, or two of them are bonded to each other to form a silicon atom to which they are bonded. To form a ring.
- m is an integer of 1 or 2.
- Examples of the compound represented by the general formula (4) include 1-trimethylsilyl-2,2-diethoxy-1-aza-2-silacyclopentane and dimethoxysilyl compounds corresponding to these diethoxysilyl compounds, methyl Examples thereof include an ethoxysilyl compound, an ethylethoxysilyl compound, a methylmethoxysilyl compound, and an ethylmethoxysilyl compound.
- Examples of the organic alkali metal compound include an organic lithium compound and an organic sodium compound. Among these, an organolithium compound is preferable.
- Examples of the organic lithium compound include an organic monolithium compound, an organic dilithium compound, and an organic polylithium compound.
- organic lithium compound for example, compounds described in Japanese Patent No. 3988495, International Publication No. 2014/014052 and the like can be used.
- n-butyllithium, sec-butyllithium, tert-butyllithium, and 1,3-phenylene-bis- (3-methyl-1-phenylpentylidene) bislithium are preferable.
- the organic alkali metal compound may be an organic alkali metal compound having an amino group.
- Examples of such a compound include compounds represented by the following general formula (5) or the following general formula (6).
- R 20 and R 21 are both trialkylsilyl groups having 3 to 18 carbon atoms, or one of them is the above trialkylsilyl group and the other is an alkyl having 1 to 20 carbon atoms.
- R 22 represents an alkylene group having 1 to 20 carbon atoms or an alkylidene group.
- R 23 represents an alkylene group or alkylidene group having 1 to 20 carbon atoms.
- R 24 represents an alkyl group having 1 to 20 carbon atoms, an aryl group having 6 to 20 carbon atoms, an aralkyl group having 7 to 20 carbon atoms, or an organosiloxy group having 1 to 100 carbon atoms.
- organic alkali metal compound represented by the general formula (5) or (6) compounds described in Japanese Patent No. 3988495, International Publication No. 2014/014052 and the like can be used.
- organic alkaline earth metal compound examples include an organic magnesium compound, an organic calcium compound, an organic strontium compound, and an organic barium compound.
- organic alkaline earth metal compound examples include, for example, ethylbutylmagnesium, di-n-butylmagnesium, di-n-hexylmagnesium, diethoxycalcium, calcium distearate, di-t-butoxystrontium, diethoxybarium. , Diisopropoxybarium, diethyl mercaptobarium, di-t-butoxybarium, diphenoxybarium, diethylaminobarium, barium distearate, diketylbarium and the like.
- organic alkali metal compounds and organic alkaline earth metal compounds can be used singly or in combination of two or more. Further, the amount of the organic alkali metal compound or organic alkaline earth metal compound used is preferably 0.02 to 15 mass with respect to 100 mass parts in total of monomers such as conjugated diene and other monomers.
- an aliphatic tertiary amine is preferable.
- the aliphatic tertiary amine include an ethylenediamine derivative, a propylenediamine derivative, or polyethyleneimine. Among these, ethylenediamine derivatives are preferable, and N, N, N ′, N′-tetramethylethylenediamine is more preferable.
- a polymer containing a repeating unit derived from an aromatic alkenyl compound undergoes a modification reaction in a solvent.
- the isolated block copolymer may be dissolved in a solvent, or the copolymer solution after completion of the polymerization reaction or hydrogenation reaction may be used as it is.
- the amount of the organic alkali metal compound or organic alkaline earth metal compound is preferably 0.01 to 5 times the molar ratio with respect to the aromatic alkenyl compound. Further, the aliphatic amine compound is preferably 0.8 to 5 times the molar ratio of the organic alkali metal compound or organic alkaline earth metal compound.
- the modifying agent is preferably 0.5 to 2 times the molar ratio of the organic alkali metal compound or organic alkaline earth metal compound.
- the first-stage reaction is a step of mixing an aliphatic amine compound with at least one selected from the group consisting of an organic alkali metal compound and an organic alkaline earth metal compound in a copolymer solution. At least one selected from the group consisting of an organic alkali metal compound and an organic alkaline earth metal compound and the aliphatic amine compound may be added first, or may be added simultaneously.
- the reaction temperature is preferably 20 to 120 ° C.
- the reaction time is preferably 0 to 120 minutes.
- the second stage reaction is a process of mixing a denaturant with the first stage reaction solution.
- the reaction temperature is preferably 20 to 120 ° C.
- the reaction time is preferably 0 to 120 minutes. If necessary, hydrogenation may be performed after the second-stage reaction.
- the conjugated diene alone or the conjugated diene and the aromatic alkenyl compound are polymerized in the presence of at least one of an organic alkali metal compound and an organic alkaline earth metal compound.
- the polymer is added to the polymer together with the peroxide and the modifier in a solution or in a kneader such as an extruder.
- the organic alkali metal compound, the organic alkaline earth metal compound, and the modifier those exemplified in “1.1.5.1. Production method (a)” can be used.
- the peroxide is not particularly limited, and 1,1-di-t-butylperoxy-3,3,5-trimethylcyclohexane, di-t-butyl peroxide, dicumyl peroxide, t-butylcumyl peroxide, 2,5-dimethyl-2,5-di (t-butylperoxy) hexane, 2,5-dimethyl-2,5-di (t-butylperoxy) hexyne-3, 1,3-bis (t-butyl And organic peroxides such as peroxyisopropyl) benzene. These peroxides can be used alone or in combination of two or more.
- the use ratio of the peroxide is preferably 0.001 to 3 parts by mass with respect to 1 part by mass of the modifier.
- the mixing and heating method is not particularly limited, and is an open type mixing roll, a non-open type Banbury mixer, a batch type melt kneader such as a kneader, a single screw extruder, a co-rotating continuous type Examples thereof include a continuous melt kneader such as a twin screw extruder and a different direction rotating type continuous twin screw extruder.
- the heating temperature is preferably 100 to 300 ° C.
- the heating time is preferably 10 to 900 seconds.
- a continuous fiber can be used as the fiber (B).
- the “continuous fiber” in the present invention refers to a fiber or an aggregate of the fibers having a fiber length of 50 mm or more, preferably 100 mm or more, more preferably 300 mm or more.
- the shape of the continuous fiber may be any of roving (tow), rod (rod), braid (braid), stranded (strand), and grid (grid).
- the continuous fibers may be contained in the matrix resin in the form of a woven or knitted fabric, a sheet such as a unidirectionally arranged strand and a multiaxial woven fabric, or a nonwoven fabric. Further, the continuous fiber may be used after being opened.
- the continuous fibers are aligned in one direction in the FRP molded body and are uninterrupted in that direction.
- the continuous fiber is a cloth material in which a unidirectional material is woven in the FRP molded body.
- the mechanical strength is extremely excellent.
- a prepreg in which a continuous fiber aligned in one direction is impregnated with a matrix resin by laminating a plurality of fibers in the fiber axis direction, a direction perpendicular thereto, and an oblique direction, it is possible to ensure isotropic mechanical properties. .
- an FRP molded body using continuous fibers has higher impact resistance and bending strength than short fiber composite materials because it contains continuous fibers.
- an FRP molded article has insufficient interfacial adhesion between the continuous fiber and the matrix resin, cracks will occur from the interface between the continuous fiber and the matrix resin when a load such as a bending load is applied.
- Cheap The cracks generated in this way propagate to the other interface between the continuous fiber and the matrix resin, thereby causing further cracks and finally traversing the molded body, leading to total destruction.
- the added fiber (B) is a long fiber such as a continuous fiber, by adding the component (A) as in the present invention, not only impact resistance but also mechanical properties such as bending strength are obtained. It has become clear that can be effectively improved.
- Component (B) is, for example, alumina fiber, glass fiber, rock wool, potassium titanate fiber, zirconia fiber, ceramic fiber, silicon fiber, silicon nitride fiber, silica-alumina fiber, kaolin fiber, bauxite fiber, cananoid fiber, boron Inorganic fibers such as fiber, boron nitride fiber, magnesia fiber, potassium titanate whisper; polyester fiber, polyamide fiber, polyimide fiber, polyvinyl alcohol modified fiber, polyvinyl chloride fiber, polypropylene fiber, polybenzimidazole fiber, acrylic fiber , Carbon fiber, phenol fiber, nylon fiber, organic fiber such as cellulose (nano) fiber; Among these, carbon fiber is preferable.
- the lower limit value of the fiber diameter of the component (B) is preferably 1 nm or more, more preferably 5 nm or more, and particularly preferably 10 nm or more.
- the upper limit of the fiber diameter is preferably 10 mm or less, more preferably 5 mm or less, still more preferably 3 mm or less, and particularly preferably 1 mm or less.
- the fiber diameter of a component can be measured by a well-known method.
- the fiber diameter can be measured by observing the component (B) with a microscope.
- the fiber diameter of the component (B) in the FRP molded body is measured by observing a filler residue collected by processing such as high-temperature ashing of the molded product, dissolution with a solvent, and decomposition with a reagent under a microscope. can do.
- the surface of the component (B) may be modified with a functional group as necessary.
- a functional group include (meth) acryloyl group, amide group, amino group, isocyanate group, imide group, urethane group, ether group, epoxy group, carboxy group, hydroxy group, and acid anhydride group.
- the method for introducing the above functional group into the component (B) is not particularly limited. However, after the component (B) is introduced by directly reacting the sizing agent, or after the sizing agent is applied or impregnated to the component (B) Examples include a method of solidifying a sizing agent as necessary. Specifically, it can be produced based on the method described in JP2013-147663A.
- Types of sizing agents include, for example, acids, acid anhydrides, alcohols, halogenating reagents, isocyanates, alkoxysilanes, cyclic ethers such as oxirane (epoxy), epoxy resins, urethane resins, urethane-modified epoxy resins, epoxy-modified urethanes Resin, amine-modified aromatic epoxy resin, acrylic resin, polyester resin, phenol resin, polyamide resin, polycarbonate resin, polyimide resin, polyetherimide resin, bismaleimide resin, polysulfone resin, polyethersulfone resin, polyvinyl alcohol resin, polyvinyl The 1 type (s) or 2 or more types chosen from the group which consists of pyrrolidone resins are mentioned.
- the amount of the sizing agent used when the component (B) is made into a fiber bundle is not particularly limited, and is preferably 0.1 to 10 parts by mass with respect to 100 parts by mass of the component (B), for example.
- carbon fibers examples include PAN-based carbon fibers made from polyacrylonitrile fiber, pitch-based carbon fibers made from coal tar and petroleum pitch, cellulose-based carbon fibers made from viscose rayon, cellulose acetate, etc.
- Preferable examples include vapor-grown carbon fibers made from hydrogen or the like, and graphitized fibers thereof.
- the carbon fiber in this invention includes the form which processed the carbon nanotube and the graphene into the fiber form. These carbon fibers may be used alone or in combination of two or more.
- Nonwoven fabric (B ') Generally, when a load such as a bending load is applied to the FRP molded body, the adhesion between the fiber and the matrix resin tends to be insufficient, and cracks are likely to occur from the interface between the fiber and the matrix resin. The cracks generated in this way propagate to the other interface between the fiber and the matrix resin, thereby causing further cracks and finally traversing the molded body, leading to total destruction.
- the adhesion between the component (B ′) and the component (C) is improved, and mechanical properties such as bending strength and impact resistance are effectively improved. It became clear that we could do it.
- the non-woven fabric (B ′) in the present invention has a void portion, and refers to a form in which fiber strands and / or monofilaments (hereinafter, strands and monofilaments are collectively referred to as fineness strands) are dispersed in a planar shape, Examples thereof include a chopped strand mat, a continuous strand mat, a papermaking mat, a carding mat, and an airlaid mat.
- a strand is a collection of a plurality of single fibers arranged in parallel and is also called a fiber bundle.
- the fineness strands usually have no regularity in the dispersed state.
- the component (B ′) By using the component (B ′), the steric hindrance between the fibers increases, the ratio of the fibers can be reduced efficiently, and the shapeability is excellent, so that it is easy to form into a complicated shape. Further, since the voids in the component (B ′) complicate the progress of resin impregnation, the component (A) and the component (C) described later form a more complicated interface, and exhibit excellent bonding ability.
- the component (B ′) is preferably a nonwoven fabric in which fibers are dispersed in a substantially monofilament shape.
- “dispersed in a substantially monofilament” means that the discontinuous fibers constituting the component (B ′) contain 50% by weight or more of fineness strands having less than 100 filaments.
- the fibers are preferably dispersed randomly.
- Such a component (B ′) can be prepared by a known method. For example, the methods described in Japanese Patent Application Laid-Open Nos. 2014-196584 and 2014-125532 can be used.
- recycled fiber As the fiber contained in the component (B ′), recycled fiber can be used.
- the recycled fiber refers to a fiber that can be reused among the collected fibers after removing the matrix resin from the fiber reinforced resin (FRP) that has become waste material.
- methods for decomposing a resin used when recovering fibers from FRP include methods such as thermal decomposition, chemical decomposition, and photodecomposition.
- the sizing agent may be removed in the treatment step, or the functional group on the carbon fiber surface may disappear. Therefore, when recycled fibers collected by recycling are reused as FRP, mechanical properties such as impact resistance and bending strength of FRP are significantly deteriorated as compared with the case of adding unused fibers.
- mechanical properties such as impact resistance and bending strength can be improved by adding the components (A) and (C) as in the present invention.
- the fiber contained in the component (B ′) used in the present embodiment preferably has a fiber length of 1 mm or more and 200 mm or less.
- the lower limit of the fiber length of the fiber is preferably 4 mm or more, more preferably 5 mm or more.
- the upper limit of the fiber length of the fiber is preferably 100 mm or less, more preferably 50 mm or less.
- fibers having the same fiber diameter as the component (B) described above can be used.
- the ratio (aspect ratio) between the fiber length and the fiber diameter of the fibers contained in the component (B ′) is preferably 100 to 30000, more preferably 500 to 10,000.
- the aspect ratio is within the above range, the mechanical properties of the molded body can be further improved.
- the aspect ratio is in the above range, deformation of the molded body and generation of anisotropy can be prevented, and a good appearance can be obtained.
- the lower limit value of the basis weight suitable for the component (B ′) is preferably 50 g / cm 3 or more, more preferably 80 g / cm 3 or more.
- the upper limit value of the basis weight is preferably 300 g / cm 3 or less, more preferably 250 g / cm 3 or less.
- the same material as the component (B) described above can be used.
- carbon fiber is preferable among the above exemplified fibers.
- carbon fibers include PAN-based carbon fibers made from polyacrylonitrile fiber, pitch-based carbon fibers made from coal tar and petroleum pitch, cellulose-based carbon fibers made from viscose rayon, cellulose acetate, etc.
- Preferable examples include vapor-grown carbon fibers made from hydrogen or the like, and graphitized fibers thereof. These carbon fibers may be used alone or in combination of two or more.
- the surface of the fiber contained in the component (B ′) may be modified with a functional group as necessary.
- functional groups include (meth) acryloyl group, amide group, amino group, isocyanate group, imide group, urethane group, ether group, epoxy group, carboxy group, hydroxy group, and acid anhydride group. .
- the method of introducing the above functional group into the fiber is not particularly limited, but the method of introducing the functional group by directly reacting the fiber with the sizing agent, or solidifying the sizing agent as necessary after applying or impregnating the fiber with the sizing agent. Methods and the like. Specifically, it can be produced based on the method described in JP2013-147663A.
- Thermoplastic resin (C) contains a thermoplastic resin (C).
- the component (C) include olefin resins; polyester resins such as polyethylene terephthalate, polybutylene terephthalate, and polylactic acid; acrylic resins; styrene resins such as polystyrene, AS resin, and ABS resin; nylon 6, nylon 6, 6, nylon 12, semi-aromatic polyamide (nylon 6T, nylon 6I, nylon 9T), polyamides such as modified polyamide; polycarbonate, polyacetal, fluororesin, modified polyphenylene ether, polyphenylene sulfide, polyester elastomer, polyarylate, liquid crystal polymer (all Aromatic, semi-aromatic), polysulfone, polyethersulfone, polyetheretherketone, polyetherimide, polyamideimide, polyimide, and the like. Or it can be used in combination of two or more. Among these, an olefin resin is prefer
- the molecular weight of the component (C) is preferably from 50,000 to 1,000,000, more preferably from 10,000 to 900,000, more preferably from 20,000 to 800,000 in terms of weight average molecular weight (Mw). It is particularly preferred.
- the ratio (Mw / Mn) between the weight average molecular weight (Mw) and the number average molecular weight (Mn) is not particularly limited, but is preferably 1 or more and 10 or less, and more preferably 2 or more and 7 or less.
- olefin resin examples include homopolymers of ⁇ -olefins having about 2 to 8 carbon atoms such as ethylene, propylene, 1-butene; these ⁇ -olefins, ethylene, propylene, 1-butene, and 3-methyl.
- binary or ternary (co) polymers with other ⁇ -olefins having about 2 to 18 carbon atoms such as 1-octadecene.
- the olefin resin include, for example, ethylene homopolymers such as branched low-density polyethylene and linear high-density polyethylene, ethylene-propylene copolymer, ethylene-1-butene copolymer, ethylene-propylene- Ethylene-based polymers such as 1-butene copolymer, ethylene-4-methyl-1-pentene copolymer, ethylene-1-hexene copolymer, ethylene-1-heptene copolymer, ethylene-1-octene copolymer Resin; propylene homopolymer, propylene-ethylene copolymer, propylene-ethylene-1-butene copolymer, propylene-ethylene-4-methyl-1-pentene copolymer, propylene-ethylene-1-hexene copolymer Propylene resins such as 1-butene homopolymer, 1-butene-ethylene copolymer, 1-butene-propylene 1-butene-based polymers
- olefin resins may be used alone or in combination of two or more.
- ethylene resins and propylene resins are preferable, propylene resins are more preferable, ethylene-propylene copolymers and propylene homopolymers are further preferable, and propylene homopolymers are particularly preferable.
- the component (A) is a block polymer having a conjugated diene polymer block having a repeating unit amount derived from a conjugated diene of 80% by mass or more and a vinyl bond content of 30% by mol to 90% by mol.
- the propylene-based resin is preferable in that the compatibility with the component (A) is particularly good.
- the vinyl bond content of the polymer block is more preferably from 50 mol% to 90 mol%, particularly preferably from 60 mol% to 90 mol%.
- the component (A) is hydrogenated because compatibility with the propylene-based resin and molecular entanglement are significantly improved.
- the weight average molecular weight (Mw) of the olefin-based resin is preferably from 50,000 to 1,000,000, more preferably from 10,000 to 900,000 in order to improve the mechanical strength of the molded body. It is especially preferable that it is 20,000 or more and 800,000 or less. Further, the ratio (Mw / Mn) of the weight average molecular weight (Mw) to the number average molecular weight (Mn) is preferably 1 or more and 10 or less, and more preferably 2 or more and 7 or less.
- the olefin resin may be a combination of crystalline polyolefin and amorphous polyolefin.
- amorphous polyolefin include homopolymers such as atactic polypropylene and atactic poly-1-butene, copolymers of propylene and other ⁇ -olefins, and 1-butene and other ⁇ -olefins. A copolymer etc. are mentioned.
- composition according to the present embodiment includes, as other additives, an anti-aging agent, an antioxidant, a weathering agent, a metal deactivator, a light stabilizer, a heat stabilizer, and an ultraviolet absorber.
- Agent antibacterial / antifungal agent, deodorant, conductivity enhancer, dispersant, softener, plasticizer, cross-linking agent, co-crosslinking agent, vulcanizing agent, vulcanizing aid, foaming agent, foaming aid, coloring
- An agent, a flame retardant, a vibration damping agent, a nucleating agent, a neutralizing agent, a lubricant, an anti-blocking agent, a dispersant, a fluidity improver, a release agent, and the like can be blended.
- the lower limit value of the content ratio of the component (A) is preferably 0.1 parts by mass or more with respect to 100 parts by mass of the component (C) that is a matrix resin. More preferably, it is 0.5 mass part or more.
- the upper limit of the content rate of a component becomes like this. Preferably it is 10 mass parts or less, More preferably, it is 5 mass parts or less.
- the lower limit value of the content ratio of the component (B) is 70 parts by mass or more, preferably 75 parts by mass with respect to 100 parts by mass of the component (C) that is a matrix resin. That's it.
- the upper limit of the content rate of a component is 250 mass parts or less with respect to 100 mass parts of (C) component which is a matrix resin, Preferably it is 240 mass parts or less, More preferably, it is 230 mass parts or less.
- the lower limit of the content ratio of the component (B ′) is preferably 50 parts by mass or more, more preferably 70 parts per 100 parts by mass of the component (C) that is a matrix resin. More than part by mass.
- the upper limit value of the content ratio of the component (B ′) is preferably 150 parts by mass or less, more preferably 100 parts by mass or less.
- the composition which concerns on this embodiment can be manufactured by mixing (A) component, (B) component, (C) component, and another component as needed.
- the mixing method is not particularly limited, and the (A) component, (B) component, and (C) component may be mixed and kneaded together, or the composition of the (A) component and the (C) component may be mixed. It can be obtained as a master batch and mixed with the component (B) to form a composition.
- coating (A) component as a sizing agent to (B) component you may mix and knead
- composition according to this embodiment can be produced by impregnating the component (A), the component (C), and other components (B ′) as necessary.
- the impregnation method is not particularly limited, and after mixing the component (A) and the component (C), the mixture may be impregnated with the component (B ′). Further, the component (A) as a sizing agent may be applied to the component (B ′) and then impregnated with the component (C).
- the component (A) is used as a sizing agent
- the polymerization solution of the polymer having an amino group obtained by the method for producing the component (A) described above is used as the sizing agent. It may be used as it is.
- the component (A) may be dissolved in a solvent and used as a solution, or dispersed in a dispersion medium and used as an emulsion (latex).
- the component (A) as a sizing agent to the surface of the component (B) or the component (B ′) in advance, in the composition according to the present embodiment, the component (B), the component (B ′) and the component (C The component (A) can be unevenly distributed at the interface with the component. As a result, even if the usage-amount of (A) component is small, the adhesiveness of (B) component or (B ') component, and (C) component can be improved effectively. As a result, it is preferable because the mechanical strength of the molded body can be dramatically improved.
- the liquid medium is not particularly limited.
- Aromatic hydrocarbons such as cyclopentane, cyclopentene and cyclohexane; aliphatic hydrocarbons such as pentane, hexane and heptane; alcohols such as methanol, ethanol, propanol, 2-propanol, ethylene glycol and glycerine; Hydrocarbon liquid media such as halogenated hydrocarbons such as methylene, chloroform and ethylene dichloride can be used.
- aromatic hydrocarbons, alicyclic hydrocarbons, and aliphatic hydrocarbons are preferable, and aliphatic hydrocarbons are more preferable.
- the content ratio of the liquid medium in the composition according to this embodiment is preferably 100 to 10000 parts by mass, more preferably 300 to 5000 parts by mass, and particularly preferably 600 to 3000 parts per 100 parts by mass of the component (A). Part by mass.
- the sizing agent can be prepared by a known emulsification method.
- a surfactant When emulsifying, a surfactant may be added alone or in combination as necessary.
- surfactants include nonionic surfactants such as polyoxyethylene alkyl ether, polyoxyethylene alkyl phenol ether, polyoxyethylene alkyl ester, polyoxyethylene sorbitan alkyl ester; myristic acid, palmitic acid, Alkali metal salts and alkaline earth metal salts of fatty acids such as oleic acid, linolenic acid, stearic acid, lauric acid, araginic acid and ricinic acid; alkalis of resin acids such as rosin acid, disproportionated rosin acid and hydrogenated rosin acid Metal salts and alkaline earth metal salts; amine salts of hydroxyamines of long-chain fatty acid esters having a chain alkyl group or a cyclic alkyl group; alkylbenzene sulfonates such as sodium dodecylbenzenesulfonate,
- Copolymerizable surfactants such as sulfoesters of ⁇ , ⁇ -unsaturated carboxylic acids, sulfate esters of ⁇ , ⁇ -unsaturated carboxylic acids, and sulfoalkylaryl ethers can also be used. Water can be suitably used as the liquid medium.
- the proportion of the surfactant used is preferably 0.5 to 50 parts by mass, more preferably 0.5 to 20 parts by mass with respect to 100 parts by mass of component (A). It is preferable that the amount of the surfactant used be in the above range because the emulsion tends to be more stable.
- the average particle size of the component (A) in the emulsion is preferably 0.02 to 100 ⁇ m, more preferably 0.1 to 10 ⁇ m, and particularly preferably 0.5 to 5 ⁇ m.
- the viscosity of the emulsion can be controlled to be easy to handle, and when the emulsion is stored, formation of a film at the gas-liquid interface can be suppressed.
- the average particle diameter of the component (A) in the emulsion can be measured by using a particle size distribution measuring apparatus based on a laser diffraction / scattering method.
- An example of such a particle size distribution measuring apparatus is Microtrac MT3000 (manufactured by Nikkiso Co., Ltd.).
- coating to (B) component as a sizing agent what dispersed (A) component in the liquid medium as an emulsion, it is preferable to contain water as a liquid medium.
- the proportion of the dispersion medium used in the emulsion is preferably 100 to 10000 parts by mass, more preferably 300 to 5000 parts by mass, and particularly preferably 600 to 3000 parts by mass with respect to 100 parts by mass of component (A).
- additives such as a pH adjuster, an antifoaming agent, a preservative, a crosslinking agent, a chelating agent, an oxygen scavenger, and a dispersing agent, which are usually blended in the latex field, can be blended.
- the pH adjuster include alkali metal hydroxides such as sodium hydroxide and potassium hydroxide; alkali metal carbonates such as sodium carbonate and potassium carbonate; alkali metal hydrogen carbonates such as sodium hydrogen carbonate; Gas; ammonia; organic amine compounds such as trimethylammonium and triethanolamine.
- alkali metal hydroxide, carbon dioxide, or ammonia is preferably used.
- component (A) When component (A) is applied as a sizing agent to component (B) or component (B ′) and mixed with component (C), component (A) is applied to component (B) or component (B ′).
- component (B) may be a single fiber or a fiber bundle, but a fiber bundle is preferable.
- the component (A) After the component (A) is applied to the component (B) or the component (B ′), it may be heated using hot air, a hot plate, a roller, an infrared heater or the like.
- composition according to the present embodiment is obtained by impregnating the component (A), the component (B) or the component (B ′), the component (C), and other components as necessary. It can be produced according to the production method described in JP2013-67051A, the production method described in JP2013-166924A, and the like.
- a conventionally known kneader such as a single screw extruder, a twin screw extruder, a Banbury mixer, a kneader, or a roll, and a kneader formed by combining them are used. be able to.
- a method of kneading each component at once or a multistage divided kneading method in which a certain component is kneaded and then the remaining components are added and kneaded can be employed.
- a twin-screw extruder is particularly preferable, and any of the same-direction rotation type and the different-direction rotation type can be suitably used.
- a twin-screw extruder is particularly preferable, and any of the same-direction rotation type and the different-direction rotation type can be suitably used.
- what connected the twin-screw extruder and the twin-screw extruder what connected the twin-screw extruder and the single-screw extruder, and what connected the continuous kneader and the twin-screw extruder are mentioned.
- the L / D ratio of the effective screw length (L) to the screw diameter (D) of the extruder
- the kneading segment a general-purpose kneading disk segment, a rotor segment, a VCMT (VARIOUS Clearance Mixing Technology) rotor segment, a twist kneading segment, a BMS (Backward Mixing single flight screw) segment, or the like can be used.
- the obtained mixture can be supplied to a single screw extruder or a twin screw extruder and melt kneaded again under the same kneading conditions as described above.
- a step of melt-kneading the mixture of the component (A) and the component (C) and other optional components under the above-described kneading conditions is performed, and the steps are repeated a plurality of times as necessary. You may go.
- a film When producing a film, it is obtained by forming a film using a twin-screw extruder and winding the film with a film take-up winder, but is not limited to the above method, and a known method is used. Can be done.
- the molded body according to the present embodiment is obtained by molding the above-described composition.
- a molding method a method similar to a method for molding a general thermoplastic composition can be applied, but a molding condition is selected so as to suppress breakage of fibers contained in the composition according to the present embodiment. It is preferable.
- the temperature is set to 10 to 30 ° C. higher than the general plasticizing temperature at the time of molding without adding reinforcing fibers to the matrix resin (non-reinforced). It is desirable to reduce shear due to plasticization.
- molded from the composition which concerns on this embodiment can be achieved by taking the conditions which lengthen fiber length at the time of shaping
- a known method can be applied, but conditions for reducing fiber shear due to plasticization can be appropriately selected.
- injection molding, extrusion molding, hollow molding, foam molding, press molding, etc. Can be used.
- the (B) component and the (B ′) component are previously formed into a desired shape such as a sheet, and a molded body is produced by impregnating a molten mixture of the (A) component and the (C) component. You can also.
- a molded body with good mechanical strength can be manufactured.
- the molded body according to the present embodiment makes use of its characteristics, for example, automobile materials such as automobile interior materials, outer plates, bumpers, etc., housings for household electrical products, home appliance parts, packaging materials, building materials, civil engineering materials, fisheries It is suitably used as a material and other industrial materials.
- automobile materials such as automobile interior materials, outer plates, bumpers, etc.
- carbon fiber when carbon fiber is used as a fiber, it can also be used as an electromagnetic wave absorber by adjusting the degree of orientation of the carbon fiber in the resin.
- Weight average molecular weight (Mw) The weight average molecular weight (Mw) is a polystyrene equivalent weight average molecular weight measured by gel permeation chromatography (GPC) (HLC-8120, manufactured by Tosoh Corporation).
- GPC gel permeation chromatography
- Developing solvent THF
- Measurement temperature 40 °C -Column: TSKgel GMHxl
- the amino group content is the content of amino groups (pieces) in one molecular chain of the polymer, and is represented by the following formula.
- ⁇ Amino group amount (amino group (pieces) / polymer 1 molecular chain)
- the amino group amount was calculated by the following method. First, Analy. Chem. The amino group concentration (mol / g) was determined by quantification by the amine titration method described in 564 (1952). That is, after purification of the obtained polymer, it was dissolved in an organic solvent, methyl violet was used as an indicator, and HClO 4 / CH 3 COOH was titrated until the color of the solution changed from purple to light blue. (Mol / g) was determined.
- the amino group amount (mol / g) ⁇ molecular weight (g / mol) is calculated to calculate the content of amino groups (pieces) in one molecular chain of the polymer.
- the molecular weight was calculated
- the component (B) When the component (B) was used, the case where the falling weight impact strength was 20 J or more was judged good, and the case where it was less than 20 J was judged as bad.
- the component (B ′) When the component (B ′) is used, the resin impregnation into the nonwoven fabric tends to be insufficient, and the strength tends to decrease. Therefore, the case where the falling weight impact strength is 10 J or more is good. The case where it was less than was judged as bad.
- Interfacial shear strength was evaluated by a microdroplet method using a composite interface characteristic evaluation device (manufactured by Toei Sangyo Co., Ltd., model number “HM410”). Specifically, the fiber to be measured is set in the apparatus, and a drop of each thermoplastic resin (C) of the type shown in the melted table is formed on the fiber, cooled sufficiently at room temperature, and used for measurement. Samples were obtained. Thereafter, the measurement sample is set again in the apparatus, the drop is sandwiched between the apparatus blades, is run on the apparatus at a speed of 0.12 mm / min, and the maximum pulling load F when the fiber is pulled out from the drop is measured. The interfacial shear strength ⁇ was calculated.
- volume content of reinforcing fibers Based on JIS K7075, the volume content of reinforcing fibers in the molded body was evaluated.
- a hydrogenation catalyst was produced by the following method.
- a 1 L three-necked flask equipped with a stirrer and a dropping funnel was replaced with dry nitrogen, and 200 mL of anhydrous tetrahydrofuran and 0.2 mol of tetrahydrofurfuryl alcohol were added.
- n-butyllithium hereinafter also referred to as “n-BuLi”
- cyclohexane solution 0.2 mol
- Methyldiethoxysilane (39.9 g) was added and allowed to react for 30 minutes. After the solvent was distilled off with a rotary evaporator, the polymer (1-D) having an amino group was obtained by vacuum drying at 60 ° C. for 18 hours.
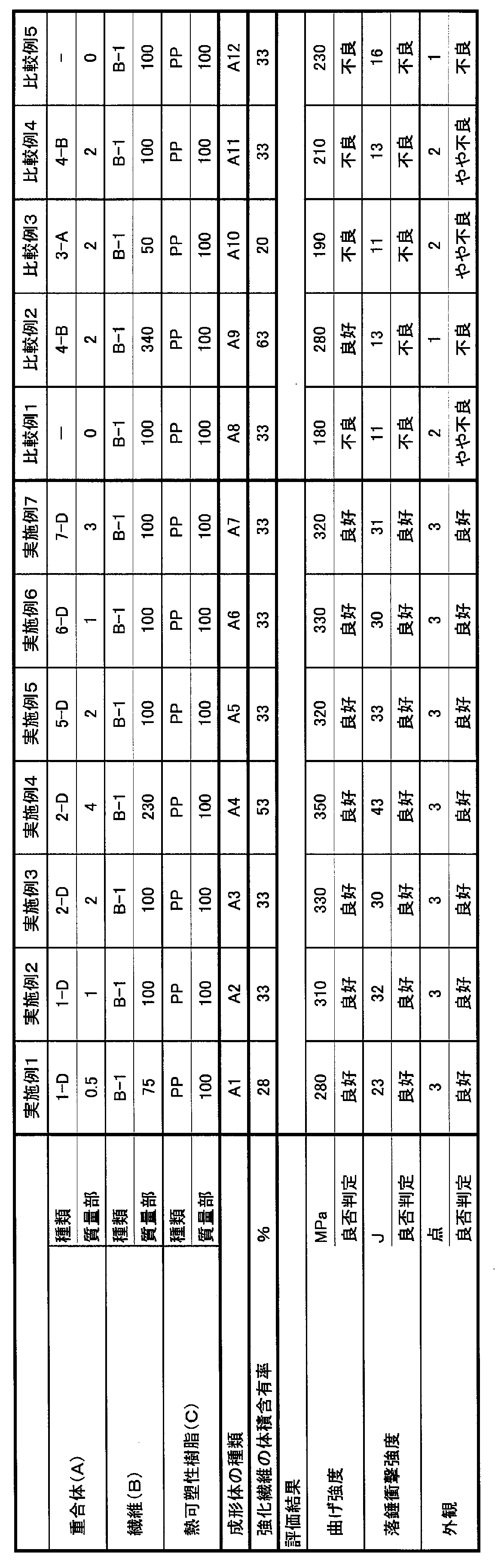
- Table 1 shows the reagents and analysis results used for the reaction of each copolymer obtained.
- Example 1 Pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate which is an antioxidant for the types and parts (A) and (C) of the parts shown in Table 2 ] (Trade name “ADK STAB AO-60”, manufactured by ADEKA) 0.1 parts by mass and tris (2,4-di-tert-butylphenyl) phosphite (trade name “ADK STAB 2112”, manufactured by ADEKA) 0 1 part by mass was added and hand blended at room temperature.
- this mixture was supplied to a twin-screw extruder “TEM26SS” (model name) manufactured by Toshiba Machine Co., Ltd., and melt-kneaded to obtain pellets.
- the temperature set in the cylinder during the melt-kneading was 140 ° C. to 220 ° C. so that the temperature near the base of the hopper was 140 ° C. and the temperature near the outlet was 220 ° C.
- the Technobel double screw extruder “KZW15-30MG” model name
- T-die with a temperature gradient of 140 ° C. to 220 ° C. were set as above.
- the produced film was cut into a size of 30 cm ⁇ 19 cm to produce a press film.
- Carbon fiber (product number “T700SC-12K-50C” manufactured by Toray Industries Inc., bundled with 12,000 filaments having a diameter of about 7 ⁇ m) as a component (B) on a single pressed film. Were stacked in a size of 30 cm ⁇ 19 cm. Furthermore, another produced press film was placed thereon.
- the laminated body thus prepared is subjected to hot press molding at 190 ° C. and a pressure of 3 MPa, whereby the volume content of reinforcing fibers (based on JIS K7075) is 28%, and a film obtained by molding the composition described in Table 2 A shaped molded body (length 30 cm, width 19 cm, thickness 80 ⁇ m) was produced.
- the produced film-like molded body is cut, 14 sheets are laminated so that the fiber directions are 90 °, and heat-press-molded at 190 ° C. and a pressure of 10 MPa to obtain a molded body A1 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm. It was.
- Examples 2, 3, 5 to 7, Comparative Examples 1 and 4 Molded by the same method as in Example 1 except that the thickness of the press film is 40 ⁇ m and the volume content of the reinforcing fiber (according to JIS K7075) is 33%. Each molded product has a length of 15 cm, a width of 15 cm, and a thickness of 1 mm. A2, A3, A5, A6, A7, A8, A11 were obtained.
- Molded body A4 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm except that the thickness of the press film is 30 ⁇ m and the volume content of the reinforcing fibers (according to JIS K7075) is 53%.
- Molded body A9 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm except that the thickness of the press film was 25 ⁇ m and the volume content of the reinforcing fibers (according to JIS K7075) was 63%.
- Molded body A10 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm except that the thickness of the press film was 60 ⁇ m and the volume content of the reinforcing fibers (according to JIS K7075) was 20%.
- the thickness of the press film was 60 ⁇ m and the volume content of the reinforcing fibers (according to JIS K7075) was 20%.
- Antioxidant pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate] (trade name “C”) for the types and parts by weight of component (C) shown in Table 2 0.1 part by mass of ADK STAB AO-60 "manufactured by ADEKA) and 0.1 part by mass of Tris (2,4-di-tert-butylphenyl) phosphite (trade name” ADK STAB 2112 "manufactured by ADEKA)
- component (A) 1 part by mass of acid-modified polypropylene (trade name “Yumex 1001”, manufactured by Sanyo Chemical Industries, Ltd.) was added and dry blended at room temperature.
- this mixture was supplied to a twin-screw extruder “TEM26SS” (model name) manufactured by Toshiba Machine Co., Ltd., and melt-kneaded to obtain pellets.
- the temperature set in the cylinder at the time of melt kneading was 140 ° C. to 220 ° C. so that the temperature near the base of the hopper was 140 ° C. and the temperature near the outlet was 220 ° C.
- the Technobel double screw extruder “KZW15-30MG” model name
- T-die with a temperature gradient of 140 ° C. to 220 ° C. were set as above. Was used to prepare a film having a thickness of 50 ⁇ m.
- the produced film was cut into a size of 30 cm ⁇ 19 cm to produce two press films.
- Molded body A12 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm, formed by the same method as in Example 1 except that the thickness of the press film was 40 ⁇ m and the volume content of the reinforcing fibers (based on JIS K7075) was 33%.
- Table 2 shows the compositions and evaluation results of the compositions of Examples and Comparative Examples.
- Comparative Example 1 since it did not contain the component (A), it was found that it was inferior in terms of bending strength, falling weight impact strength, and appearance as compared with the Example.
- the component (A) does not have an amino group, and the content ratio of the component (B) is too high. Therefore, in the obtained molded product, the component (B) and the component (C) are firmly It was found that the falling weight impact strength was hardly improved even when compared with Comparative Example 1 that did not include the component (A). Moreover, since the content rate of (B) component was too high, the impregnation of resin became inadequate and many voids were observed on the surface in the molded object.
- the component (A) does not have an amino group, and the content ratio of the component (B) is too low. Therefore, the bending strength and falling weight are lower than those of Comparative Example 1 that does not include the component (A). It was found that the impact strength hardly improved.
- Comparative Example 4 since the component (A) does not have an amino group, the component (B) and the component (C) cannot be firmly bonded to each other in the obtained molded body. It was found that the bending strength and drop weight impact strength were hardly improved even in comparison with Comparative Example 1 that did not contain.
- the method for producing the nonwoven fabrics (B′-1) to (B′-3) is as follows.
- a product number “T700SC-12K-50C” (density: 1.80 g / cm 3 ) manufactured by Toray Industries, Inc. was cut with a cartridge cutter to obtain chopped yarns having fiber lengths of 6 mm, 10 mm, and 40 mm, respectively.
- 100 liters of a 1.5 wt% aqueous solution of a product name “sodium n-dodecylbenzenesulfonate” manufactured by Wako Pure Chemical Industries, Ltd. was stirred to prepare a prefoamed dispersion.
- the obtained chopped yarn was put into this dispersion, stirred for 10 minutes, poured into a paper machine having a paper surface, dehydrated by suction, and then dried at a temperature of 150 ° C. for 2 hours to obtain a nonwoven fabric (B '-1) to (B'-3) were obtained.
- the nonwoven fabric (B′-1) is a nonwoven fabric obtained from chopped yarn having a fiber length of 6 mm
- the nonwoven fabric (B′-2) is a nonwoven fabric obtained from chopped yarn having a fiber length of 10 mm.
- the manufacturing method of the nonwoven fabric (B′-4) is as follows. After sandwiching both sides of the molded body obtained in Comparative Example 6 described later with 300 mesh, the crucible is filled at a filling rate of 80% by volume, and heat cracking is performed using an electric furnace at 400 ° C. for 1 hour under cracking gas filling. Thus, a recycled carbon fiber having an average fiber diameter of 7 ⁇ m and an average fiber length of 4 mm was obtained. 100 liters of a 1.5 wt% aqueous solution of a product name “sodium n-dodecylbenzenesulfonate” manufactured by Wako Pure Chemical Industries, Ltd.
- a non-woven fabric (B′-4) made of fibers was obtained.
- Example 8 Pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate, which is an antioxidant, for the types and parts by weight of component (A) and component (C) shown in Table 3. ] (Trade name “ADK STAB AO-60”, manufactured by ADEKA) 0.1 parts by mass and tris (2,4-di-tert-butylphenyl) phosphite (trade name “ADK STAB 2112”, manufactured by ADEKA) 0 1 part by mass was added and hand blended at room temperature.
- this mixture was supplied to a twin-screw extruder “TEM26SS” (model name) manufactured by Toshiba Machine Co., Ltd., and melt-kneaded to obtain pellets.
- the temperature set in the cylinder during the melt-kneading was 140 ° C. to 220 ° C. so that the temperature near the base of the hopper was 140 ° C. and the temperature near the outlet was 220 ° C.
- the Technobel double screw extruder “KZW15-30MG” model name
- T-die with a temperature gradient of 140 ° C. to 220 ° C. were set as above.
- a film having a thickness of 100 ⁇ m was prepared using The produced film was cut into a size of 15 cm ⁇ 15 cm to produce a press film.
- a non-woven fabric (B′-1) as a component (B ′) was arranged in a size of 15 cm ⁇ 15 cm and laminated on the produced single press film. Furthermore, another produced press film was placed thereon. A film obtained by molding the composition described in Table 3 with a volume content of reinforcing fibers (based on JIS K7075) of 33% by hot press molding the laminate thus produced at 190 ° C. and a pressure of 3 MPa. A shaped product (15 cm long, 15 cm wide, 200 ⁇ m thick) was produced.
- Example 9 to 14, Comparative Examples 6 and 10 Except for the components (A) and (B ′) shown in Table 3, molding was performed in the same manner as in Example 8 to obtain molded bodies A14 to A20 and A24 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm, respectively. .
- Example 7 In the same manner as in Example 8, except that the component (A) is as shown in Table 3, the component (B ′) is a non-woven fabric (B′-2), and the thickness of the film produced using a T-die is 50 ⁇ m. It shape
- Example 8 According to the same method as in Example 8, except that the component (A) is as shown in Table 3, the component (B ′) is a non-woven fabric (B′-3), and the thickness of the film produced using a T-die is 150 ⁇ m. It shape
- Antioxidant pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate] (trade name “C”) for the types and parts by weight of component (C) shown in Table 3 0.1 part by mass of ADK STAB AO-60 "manufactured by ADEKA) and 0.1 part by mass of Tris (2,4-di-tert-butylphenyl) phosphite (trade name” ADK STAB 2112 "manufactured by ADEKA)
- component (A) 3 parts by mass of acid-modified polypropylene (trade name “Yumex 1001”, manufactured by Sanyo Chemical Industries, Ltd.) was added and dry blended at room temperature.
- this mixture was supplied to a twin-screw extruder “TEM26SS” (model name) manufactured by Toshiba Machine Co., Ltd., and melt-kneaded to obtain pellets.
- the temperature set in the cylinder at the time of melt kneading was 140 ° C. to 220 ° C. so that the temperature near the base of the hopper was 140 ° C. and the temperature near the outlet was 220 ° C.
- the Technobel double screw extruder “KZW15-30MG” model name
- T-die with a temperature gradient of 140 ° C. to 220 ° C. were set as above.
- a film having a thickness of 100 ⁇ m was prepared using The produced film was cut into a size of 15 cm ⁇ 15 cm to produce two press films.
- the same methods as in Example 8 were used to mold the nonwoven fabrics (B′-1) and (B′-4), respectively, to obtain molded bodies A23 and A25 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm, respectively.
- Table 3 shows the compositions and evaluation results of the compositions of Examples and Comparative Examples.
- Comparative Example 7 since the component (A) does not have an amino group, the component (B ′) and the component (C) cannot be firmly bonded to each other in the obtained molded body. It was found that the falling weight impact strength was hardly improved even when compared with Comparative Example 6 containing no component.
- Comparative Example 8 since the component (A) does not have an amino group, it was found that the bending strength and drop weight impact strength are hardly improved even when compared with Comparative Example 6 that does not include the component (A).
- the solution was transferred to a rotary evaporator N-11 (trade name) manufactured by Tokyo Rika Kikai Co., Ltd., and each aqueous emulsified dispersion (sizing was adjusted so that the concentration of the polymer (A) was 7% by dissolving the cyclohexane. Agent).
- the average particle size of the polymer (A) was determined by using a dynamic light scattering particle size distribution analyzer (model “FPAR-1000”, manufactured by Otsuka Electronics Co., Ltd.). The particle size distribution of the sizing agent) was measured, and the average particle diameter D50 was determined from the particle size distribution.
- Carbon fiber B-0 (trade name “Torayca T700SC-12000-50C” manufactured by Toray Industries, Ltd., where the sizing agent has been removed with acetone) was continuously immersed in a treatment bath filled with the prepared aqueous emulsion dispersion (sizing agent). Then, a predetermined amount of the polymer (A) having an amino group was adhered to the fiber (B). Subsequently, it was dried by continuously passing through an oven at 120 ° C. for 5 minutes. The fiber (B) to which the obtained polymer (A) having an amino group was attached was subjected to the above-described interfacial shear strength measurement and used for producing a carbon fiber film.
- Examples 15 and 16, Comparative Examples 13 and 14 For the types and parts by weight of component (C) shown in Table 4, pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate], an antioxidant (trade name “ 0.1 part by mass of ADK STAB AO-60 ”(manufactured by ADEKA) and 0.1 part by mass of Tris (2,4-di-tert-butylphenyl) phosphite (trade name“ ADK STAB 2112 ”, manufactured by ADEKA) And hand blended at room temperature.
- component (C) shown in Table 4 pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate]
- an antioxidant trade name “ 0.1 part by mass of ADK STAB AO-60 ”(manufactured by ADEKA)
- this mixture was formed into a film having a thickness of 40 ⁇ m using a Technobel double screw extruder “KZW15-30MG” (model name) and a T die with a temperature gradient of 140 ° C. to 220 ° C. of the cylinder. Created. The produced film was cut into a size of 30 cm ⁇ 19 cm to produce two press films.
- KZW15-30MG model name
- Example 2 15 cm in length, respectively, in the same manner as in Example 1 except that the produced film for press and the carbon fiber film produced using the fiber (B) to which the polymer (A) having an amino group was adhered were used. Molded bodies B1, B2, B4, and B5 having a width of 15 cm and a thickness of 1 mm were obtained.
- Example 12 15 cm in length, 15 cm in width, in the same manner as in Example 15 except that the carbon fiber B-0 was used as it was without applying the sizing agent containing the component (A) and the component (C) described in Table 4 was used. A molded body B3 having a thickness of 1 mm was obtained.
- Table 4 shows the compositions and evaluation results of the compositions of the examples and comparative examples.
- Comparative Example 12 since it did not contain the component (A), it was found that it was inferior in terms of interfacial shear strength, bending strength, falling weight impact strength and appearance as compared with the Examples.
- a product number “T700SC-12K-50C” manufactured by Toray Industries, Inc. was immersed in acetone to remove the sizing agent, and then cut with a cartridge cutter to obtain chopped yarns having fiber lengths of 6 mm, 10 mm, and 40 mm, respectively.
- 100 liters of a 1.5 wt% aqueous solution of a product name “sodium n-dodecylbenzenesulfonate” manufactured by Wako Pure Chemical Industries, Ltd. was stirred to prepare a prefoamed dispersion.
- the obtained chopped yarn was put into this dispersion, stirred for 10 minutes, poured into a paper machine having a paper surface, dehydrated by suction, and then dried at a temperature of 150 ° C. for 2 hours to obtain a nonwoven fabric (B '-5), (B'-6) and (B'-7) were obtained, respectively.
- the nonwoven fabric (B′-5) is a nonwoven fabric obtained from chopped yarn having a fiber length of 6 mm
- the nonwoven fabric (B′-6) is a nonwoven fabric obtained from chopped yarn having a fiber length of 10 mm.
- a non-woven fabric (B'-8) was obtained in the same manner as described above except that non-woven fabric (B'-4) (non-woven fabric made of recycled carbon fiber) was used.
- Example 17 The nonwoven fabric (B′-5) was immersed in a treatment bath filled with the prepared aqueous emulsion dispersion (sizing agent), and a predetermined amount of the polymer (1-D) was adhered to the nonwoven fabric (B′-5). . Subsequently, it was continuously passed through an oven at 120 ° C. for 5 minutes and dried to produce a nonwoven fabric (B′-5) to which the polymer (1-D) was adhered.
- pentaerythritol tetrakis [3- (3,5-di-tert-butyl-4-hydroxyphenyl) propionate], which is an antioxidant, is used for the types and parts by weight of component (C) shown in Table 5.
- component (C) shown in Table 5 0.1 part by mass of trade name “Adeka Stub AO-60” (manufactured by ADEKA) and 0.1 tris (2,4-di-tert-butylphenyl) phosphite (trade name “Adeka Stub 2112”, manufactured by ADEKA) 0.1 Part by mass was added and hand blended at room temperature.
- this mixture was formed into a film having a thickness of 100 ⁇ m using a Technobel twin screw extruder “KZW15-30MG” (model name) with a temperature gradient of 140 ° C. to 220 ° C. and a T die.
- KZW15-30MG model name
- the produced film was cut into a size of 15 cm ⁇ 15 cm to produce two press films.
- a molded body B6 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm was obtained in the same manner as in Example 8.
- Examples 18 to 21, Comparative Examples 15 to 19 Except for the components (A) and (B ′) shown in Table 5, molding was carried out in the same manner as in Example 8 to obtain molded bodies B7 to B15 having a length of 15 cm, a width of 15 cm, and a thickness of 1 mm, respectively.
- Table 5 shows the compositions and evaluation results of the compositions of Examples and Comparative Examples.
- the present invention is not limited to the above embodiment, and various modifications can be made.
- the present invention includes configurations that are substantially the same as the configurations described in the embodiments (for example, configurations that have the same functions, methods, and results, or configurations that have the same objects and effects).
- the present invention also includes a configuration in which a non-essential part of the configuration described in the above embodiment is replaced with another configuration.
- the present invention includes a configuration that achieves the same effect as the configuration described in the above embodiment or a configuration that can achieve the same object.
- the present invention includes a configuration obtained by adding a known technique to the configuration described in the above embodiment.
Landscapes
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Polymers & Plastics (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Materials Engineering (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Inorganic Chemistry (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Reinforced Plastic Materials (AREA)
- Graft Or Block Polymers (AREA)
Abstract
Description
本発明に係る組成物の一態様は、
アミノ基を有する重合体(A)と、繊維(B)と、熱可塑性樹脂(C)と、を含有し、
前記熱可塑性樹脂(C)100質量部に対して前記繊維(B)を70質量部以上250質量部以下含有することを特徴とする。
適用例1の組成物において、
前記アミノ基を有する重合体(A)の含有割合が、前記熱可塑性樹脂(C)100質量部に対して0.1質量部以上10質量部以下であることができる。
適用例1または適用例2の組成物において、
前記繊維(B)が炭素繊維であることができる。
適用例1ないし適用例3のいずれか一例の組成物において、
前記アミノ基を有する重合体(A)が共役ジエン系重合体であることができる。
適用例1ないし適用例4のいずれか一例の組成物において、
前記繊維(B)の繊維長が50mm以上であることができる。
本発明に係る組成物の一態様は、
アミノ基を有する重合体(A)と、
不織布(B’)と、
熱可塑性樹脂(C)と、
を含有することができる。
適用例6の組成物において、
前記不織布(B’)がリサイクル繊維を含有することができる。
適用例6または適用例7の組成物において、
前記熱可塑性樹脂(C)100質量部に対して、前記不織布(B’)を50質量部以上150質量部以下含有することができる。
適用例6ないし適用例8のいずれか一例の組成物において、
前記不織布(B’)が炭素繊維を含むことができる。
適用例6ないし適用例9のいずれか一例の組成物において、
前記熱可塑性樹脂(C)100質量部に対して、前記アミノ基を有する重合体(A)を0.1質量部以上10質量部以下含有することができる。
適用例1ないし適用例10のいずれか一例の組成物において、
前記熱可塑性樹脂(C)がオレフィン系樹脂であることができる。
本発明に係る成形体の一態様は、
適用例1ないし適用例11のいずれか一例の組成物を用いて作成されることを特徴とする。
一般的に、連続繊維や不織布等を用いたFRP成形体は曲げ荷重などの負荷が印加された場合、連続繊維や不織布等とマトリックス樹脂との接着性が不十分であると、界面から亀裂が発生しやすい。このようにして発生した亀裂が繊維とマトリックス樹脂との他の界面に伝播することにより、さらに亀裂を誘発し、最終的に成形体を横断して全体破壊に至る。
本実施形態に係る組成物は、アミノ基を有する重合体(A)を含む。(A)成分は、本実施形態に係る成形体において(B)成分や(B’)成分と(C)成分とを強固に接着させることにより、曲げ荷重などの負荷が印加された場合の(B)成分や(B’)成分と(C)成分の界面から亀裂の発生を抑制し、成形体の曲げ強度、落錘衝撃強度等の機械的強度を向上させると考えられる。
(A)成分は、共役ジエンに由来する繰り返し単位を含むことが好ましい。共役ジエンとしては、例えば1,3-ブタジエン、イソプレン、2,3-ジメチル-1,3-ブタジエン、1,3-ペンタジエン、2-メチル-1,3-オクタジエン、1,3-ヘキサジエン、1,3-シクロヘキサジエン、4,5-ジエチル-1,3-オクタジエン、3-ブチル-1,3-オクタジエン、ミルセン、ファルネセン及びクロロプレン等を挙げることができる。機械的強度や耐寒性に優れた成形体を得るために、共役ジエンとしては、1,3-ブタジエン又はイソプレンを含むことが好ましい。
(A)成分は、共役ジエン以外の化合物に由来する繰り返し単位を含むものであってもよい。このような化合物としては、芳香族アルケニル化合物が好ましい。芳香族アルケニル化合物の中でも、機械的強度、耐熱性、及び耐寒性に優れた成形体を作製するために、下記一般式(1)で示される不飽和単量体がより好ましい。
(A)成分は、(C)成分との相容性を高め、(B)成分と(C)成分をより強固に接着させるために、ブロック共重合体であることが好ましい。さらに、下記A~Dの重合体ブロックの中から選ばれた2種以上の重合体ブロックを含むブロック重合体であることがより好ましい。
Bブロック:共役ジエンに由来する繰り返し単位量が80質量%以上であって、かつ、ビニル結合含量が30モル%未満の重合体ブロック。
Cブロック:共役ジエンに由来する繰り返し単位量が80質量%以上であって、かつ、ビニル結合含量が30モル%以上90モル%以下の重合体ブロック。
Dブロック:共役ジエンに由来する繰り返しと芳香族アルケニル化合物に由来する繰り返しとのランダム共重合体ブロックであって、上記A~C以外の重合体ブロック。
本実施形態に係る成形体の耐候性及び機械的強度を向上させるために、(A)成分は水素添加(以下、「水添」ともいう。)された重合体であることが好ましい。特に(C)成分としてオレフィン系樹脂を用いた場合、水素添加された重合体を(A)成分として使用することにより、(A)成分とオレフィン系樹脂との分子の絡み合い及び相容性を顕著に向上させ、(C)成分と(B)成分との接着性をより向上させることができる。
(A)成分は、例えば特許第5402112号公報、特許第4840140号公報、国際公開第2003/029299号等に記載の方法に従って製造することができる。また、(A)成分がブロック重合体である場合、例えば、特許第3134504号公報、特許第3360411号公報、特許第3988495号公報、国際公開第2014/014052号等に記載の方法に従って製造することができる。具体的な(A)成分の製造方法としては、例えば下記(a)~(c)の方法が挙げられる。なお、以下の方法によって得られた重合体を、必要に応じて上述の方法により水素添加することができる。
製造方法(a)は、共役ジエンを単独で、あるいは、共役ジエン及び芳香族アルケニル化合物を、有機アルカリ金属化合物及び有機アルカリ土類金属化合物のうち少なくとも一種の存在下で重合し、得られた重合体に変性剤を反応させる方法である。必要に応じて水素添加を行ってもよい。変性剤がアルコキシシリル基とアミノ基を有する化合物である場合、高い導入率で導入することができる。
R6 (4-m-n)Si(OR7)mXn (2)
式(2)中、R6は炭素数1~20のアルキル基、炭素数6~20のアリール基、炭素数7~20のアラルキル基又は炭素数1~100のオルガノシロキシ基である。R6が複数ある場合は、各々のR6は同一の基でも異なる基でもよい。R7は炭素数1~20のアルキル基、炭素数6~20のアリール基又は炭素数7~20のアラルキル基である。R7が複数ある場合は、各々のR7は同一の基でも異なる基でもよい。
製造方法(b)は、共役ジエンと芳香族アルケニル化合物、又は共役ジエンと芳香族アルケニル化合物と他の単量体とを重合して得られた重合体に、少なくとも1つの有機アルカリ金属化合物及び/又は有機アルカリ土類金属化合物と、少なくとも1つの脂肪族アミン化合物との存在下で変性剤を反応させる方法である。有機アルカリ金属化合物、有機アルカリ土類金属化合物や変性剤としては、「1.1.5.1.製造方法(a)」で例示されたものが使用できる。
製造方法(c)は、共役ジエンを単独で、あるいは、共役ジエン及び芳香族アルケニル化合物を、有機アルカリ金属化合物及び有機アルカリ土類金属化合物のうち少なくとも一種の存在下で重合し、得られた重合体を、過酸化物と変性剤とともに、溶液中又は押し出し機等の混練機中で変性剤を重合体に付加させる方法である。有機アルカリ金属化合物、有機アルカリ土類金属化合物や変性剤としては、「1.1.5.1.製造方法(a)」で例示されたものが使用できる。
繊維(B)としては、連続繊維を使用することができる。本発明における「連続繊維」とは、繊維長が50mm以上、好ましくは100mm以上、より好ましくは300mm以上である繊維もしくは当該繊維の集合体のことをいう。連続繊維が集合体である場合、連続繊維の形状は、ロービング(トウ)、棒状(ロッド)、組紐状(ブレイド)、より線状(ストランド)、格子状(グリッド)のいずれであってもよい。また、連続繊維は、織編物、ストランドの一方向配列シート状物及び多軸織物等のシート状、又は不織布状でマトリックス樹脂中に含有されていてもよい。また、連続繊維は開繊して用いてもよい。
一般的に、FRP成形体は曲げ荷重などの負荷が印加された場合、繊維とマトリックス樹脂との接着性が不十分となりやすく、繊維とマトリックス樹脂との界面から亀裂が発生しやすい。このようにして発生した亀裂が繊維とマトリックス樹脂との他の界面に伝播することにより、さらに亀裂を誘発し、最終的に成形体を横断して全体破壊に至ることがあった。ところが、本願発明のように(A)成分を添加することにより(B’)成分と(C)成分との接着性が向上し、曲げ強度及び耐衝撃性などの機械的特性を効果的に向上できることが明らかとなった。
本実施形態に係る組成物は、熱可塑性樹脂(C)を含有する。(C)成分としては、例えばオレフィン系樹脂;ポリエチレンテレフタレート、ポリブチレンテレフタレート、ポリ乳酸等のポリエステル系樹脂;アクリル系樹脂;ポリスチレン、AS樹脂、ABS樹脂等のスチレン系樹脂;ナイロン6、ナイロン6,6、ナイロン12、半芳香族ポリアミド(ナイロン6T、ナイロン6I、ナイロン9T)、変性ポリアミド等のポリアミド;ポリカーボネート、ポリアセタール、フッ素樹脂、変性ポリフェニレンエーテル、ポリフェニレンサルファイド、ポリエステルエラストマー、ポリアリレート、液晶ポリマー(全芳香族系、半芳香族系)、ポリスルホン、ポリエーテルスルホン、ポリエーテルエーテルケトン、ポリエーテルイミド、ポリアミドイミド、ポリイミドが例示され、これらから選ばれる1種又は2種以上を組み合わせて用いることができる。これらの中でも、オレフィン系樹脂が好ましい。
本実施形態に係る組成物には、上記成分以外に、その他の添加剤として、老化防止剤、酸化防止剤、耐候剤、金属不活性剤、光安定剤、熱安定剤、紫外線吸収剤、防菌・防黴剤、防臭剤、導電性付与剤、分散剤、軟化剤、可塑剤、架橋剤、共架橋剤、加硫剤、加硫助剤、発泡剤、発泡助剤、着色剤、難燃剤、制振剤、造核剤、中和剤、滑剤、ブロッキング防止剤、分散剤、流動性改良剤、離型剤等を配合することができる。
本実施形態に係る組成物において、(A)成分の含有割合の下限値は、マトリックス樹脂である(C)成分100質量部に対して、好ましくは0.1質量部以上、より好ましくは0.5質量部以上である。(A)成分の含有割合の上限値は、好ましくは10質量部以下、より好ましくは5質量部以下である。(A)成分の含有割合が前記範囲にあることにより、(A)成分が(B)成分や(B’)成分と(C)成分を強固に接着させることができる。その結果、曲げ荷重などの負荷が印加された場合の(B)成分や(B’)成分と(C)成分との界面から亀裂の発生を抑制し、成形体の曲げ強度、落錘衝撃強度等の機械的強度を向上させると考えられる。
本実施形態に係る組成物は、(A)成分、(B)成分、(C)成分、及び必要に応じてその他の成分を混合することにより製造することができる。混合の方法は、特に限定されず、(A)成分、(B)成分、(C)成分を一括で混合、混練してもよいし、(A)成分と(C)成分との組成物を得てこれをマスターバッチとして、(B)成分と混合して組成物としてもよい。また、(B)成分にサイジング剤としての(A)成分を塗布した後、(C)成分と混合、混練してもよい。
(A)成分をサイジング剤として使用する場合、上述した(A)成分の製造方法により得られたアミノ基を有する重合体の重合溶液をサイジング剤としてそのまま使用してもよい。また、該重合溶液から(A)成分を分離した後、(A)成分を溶媒に溶解させて溶液として使用したり、分散媒へ分散させてエマルジョン(ラテックス)として使用してもよい。(A)成分をサイジング剤としてあらかじめ(B)成分や(B’)成分の表面へ塗布することにより、本実施形態に係る組成物中において、(B)成分や(B’)成分と(C)成分との界面に(A)成分を偏在させることができる。その結果、(A)成分の使用量が少量であっても効果的に(B)成分や(B’)成分と(C)成分との接着性を向上させることができる。その結果、成形体の機械的強度を飛躍的に向上させることができるため好ましい。
本実施形態に係る組成物は、(A)成分、(B)成分又は(B’)成分、(C)成分及び必要に応じて上記その他の成分等を含浸させることによる、特開2013-67051号公報に記載の製造方法、特開2013-166924号公報に記載の製造方法等に準じて製造することができる。
本実施形態に係る成形体は、上述の組成物を成形して得られるものである。成形方法としては、一般的な熱可塑性組成物の成形方法と同様の方法を適用することができるが、本実施形態に係る組成物に含まれる繊維の折損が抑制できるような成形条件を選択することが好ましい。繊維長をできるだけ維持する成形条件としては、マトリックス樹脂に対して強化繊維を添加していない(非強化の)状態で成形する際の一般的可塑化温度より10~30℃高めの温度設定とするなど、可塑化による剪断を低減することが望ましい。このように成形時においては繊維長を長くするような条件をとることで、本実施形態に係る組成物から成形される成形体中に分散される繊維強化樹脂成形体を達成できる。
以下、本発明を実施例に基づいて具体的に説明するが、本発明はこれらの実施例に限定されるものではない。実施例、比較例中の「部」及び「%」は、特に断らない限り質量基準である。なお、本発明の組成物の物性の測定方法は以下の通りである。
(1)ビニル結合含量等
ビニル結合含量(1,2結合含量及び3,4結合含量)は、赤外吸収スペクトル法(モレロ法)によって求めた。ただし、ビニル結合含量の単位は、モル%基準である。スチレン単位とp-メチルスチレン単位の合計含有量は、赤外吸収スペクトル法(モレロ法)により、検量線を作成して求めた。ただし、スチレン単位の含有量の単位は、質量%基準である。
水添率は、四塩化炭素を溶媒として用い、400MHz、1H-NMRスペクトルから算出した。
重量平均分子量(Mw)は、ゲルパーミエーションクロマトグラフィー(GPC)(東ソー(株)製、HLC-8120)法により測定された、ポリスチレン換算の重量平均分子量である。
・展開溶媒:THF
・測定温度:40℃
・カラム:TSKgel GMHxl
アミノ基量は、重合体1分子鎖中のアミノ基(個)の含有量であり、下記式により表される。
・アミノ基量=(アミノ基(個)/重合体1分子鎖)
アミノ基量は、以下の手法で算出した。まず、Analy.Chem.564(1952)記載のアミン滴定法による定量によりアミノ基濃度(mol/g)を求めた。即ち、得られた重合体を精製後、有機溶剤に溶解し、指示薬としてメチルバイオレットを用い、溶液の色が紫色から水色に変化するまでHClO4/CH3COOHを滴定することにより、アミノ基量(mol/g)を求めた。このアミノ基量(mol/g)を元に、アミノ基量(mol/g)×分子量(g/mol)と計算を行うことにより、重合体1分子鎖中のアミノ基(個)の含有量を算出した。なお、分子量は、GPC法で求めたポリスチレン換算の数平均分子量から求めた。
(1)曲げ強度
実施例や比較例で組成物を成形して得られた成形体を、ユニバーサルカッターを用いて、大きさが10mm×150mm×1mm(=幅×長さ×厚さ)となるよう切り出して試験片を作成した。試験はISO179に準じて、支点間距離64mm、試験速度2mm/minで行った。試験温度は23℃、単位は「MPa」である。
・(B)成分を使用した場合には、曲げ強度が250MPa以上である場合を良好、250MPa未満である場合を不良と判断した。
・(B’)成分を使用した場合には、不織布への樹脂含浸が不十分になる傾向があり、強度が低下する傾向があるため、曲げ強度が160MPa以上である場合を良好、160MPa未満を不良と判断した。
実施例や比較例で組成物を成形して得られた成形体を、ユニバーサルカッターを用いて、大きさが80mm×55mm×1mm(=幅×長さ×厚さ)となるよう切り出して試験片を作成した。その後、島津製作所社製高速衝撃試験機「HITS-P10」(型式名)にセットし、落錘試験(重錘のポンチ先端直径:12.7mm、受け台穴径:43mm、試験速度:6.7m/秒、試験温度:23℃)に供し、JIS K7211-2に準じてパンクチャーエネルギー量を測定した。単位は「J」である。
・(B)成分を使用した場合には、落錘衝撃強度が20J以上である場合を良好、20J未満である場合を不良と判断した。
・(B’)成分を使用した場合には、不織布への樹脂含浸が不十分になる傾向があり、強度が低下する傾向があるため、落錘衝撃強度が10J以上である場合を良好、10J未満である場合を不良と判断した。
複合材界面特性評価装置(東栄産業株式会社製、型番「HM410」)を用いてマイクロドロップレット法により界面せん断強度を評価した。具体的には、測定対象とする繊維を装置にセッティングし、溶融した表中に示す種類のそれぞれの熱可塑性樹脂(C)のドロップを繊維上に形成させ、室温で十分に冷却し、測定用の試料を得た。その後、再度測定試料を装置にセットし、ドロップを装置ブレードで挟み、装置上で0.12mm/分の速度で走行させ、繊維をドロップから引き抜く際の最大引き抜き荷重Fを測定し、下記式により界面せん断強度τを算出した。界面せん断強度が20MPa以上である場合を良好、20MPa未満である場合を良好と判断した。
τ=F/πDL
上記式において、
F:炭素繊維から熱可塑性樹脂が剥離する際に生じる最大応力(N)
D:1本の炭素繊維の直径(m)
L:炭素繊維の軸方向における熱可塑性樹脂の直径(m)
をそれぞれ表す。
<(B)成分を用いた場合>
フィルム状材料(縦30cm、横19cm、厚み80μm)の製造時において、縦10mm×横10mmの100mm2をマイクロスコープにより表面観察し、以下の基準で評価した。
・「3点」:ボイドが観察されることはなく、樹脂含浸が良好と判断する。
・「2点」:観察されたボイドが1個以上10個未満であり、樹脂含浸がやや不良と判断する。
・「1点」:観察されたボイドが10個以上であり、樹脂含浸が不良と判断する。
<(B’)成分を用いた場合>
外観は、成形体の表面を目視で観測し、以下の基準で評価した。
・「3点」:成形体の表面が滑らかであり、光沢がある。良好と判断する。
・「2点」:成形体の表面に凹凸がみえるが、光沢がある。やや不良と判断する。
・「1点」:成形体の表面に多数の凹凸がみえ、光沢もない。不良と判断する。
JIS K7075に準拠し、成形体中の強化繊維の体積含有率を評価した。
以下の方法により、水添触媒を製造した。
撹拌機、滴下漏斗を備えた1L容量の三つ口フラスコを乾燥窒素で置換し、無水テトラヒドロフラン200mL及びテトラヒドロフルフリルアルコール0.2モルを加えた。その後、n-ブチルリチウム(以下「n-BuLi」ともいう。)/シクロヘキサン溶液(0.2モル)を三つ口フラスコ中に15℃にて滴下して反応を行い、テトラヒドロフルフリルオキシリチウムのテトラヒドロフラン溶液を得た。
[合成例1]
窒素置換された内容積50リットルの反応容器に、シクロヘキサン(25kg)、テトラヒドロフラン(750g)、p-メチルスチレン(750g)、及びn-ブチルリチウム(7.0g)を加え、50℃からの断熱重合を行った。反応完結後、温度を20℃として、1,3-ブタジエン(3,750g)を加え、断熱重合を行った。30分後、p-メチルスチレン(500g)を加え重合を行った。水素圧1.0MPaを保つようにして30分反応させて重合停止した。次いで、四塩化ケイ素(1.7g)を加え、15分後に上記水添触媒(5.4g)、及びジエチルアルミニウムクロライド(2.1g)を加え、水素圧1.0MPaを保つようにして1時間反応させた。反応後、反応液を70℃、常圧に戻して反応容器より抜き出し、重合体溶液を得た。脱溶媒槽に水と上記重合体溶液を加え(重合体溶液100質量部に対して、水200質量部の割合)、脱溶媒槽の液相の温度:95℃で、2時間スチームストリッピング(スチーム温度:190℃)により脱溶媒を行い、110℃に調温された熱ロールにより乾燥を行うことで水添共役ジエンブロック重合体(1-B)を得た。撹拌機を備えた7L容量のセパラブルフラスコを乾燥窒素で置換し、上記共役ジエンブロック重合体(1-B)(500g)をシクロヘキサン(4,000g)に溶解させた。次いで、N,N,N’,N’-テトラメチルエチレンジアミンを(13.8g)及びs-ブチルリチウム(7.6g)を加えて15分撹拌した後、N,N-ビス(トリメチルシリル)アミノプロピルメチルジエトキシシラン(39.9g)を加え、30分間反応させた。ロータリーエバポレーターで溶剤を留去した後、60℃で18時間真空乾燥してアミノ基を有する重合体(1-D)を得た。
合成例1と同様の手法で、表1に示す成分と量を使用してアミノ基を有する重合体(2-D)、アミノ基を有する重合体(5-D)、アミノ基を有する重合体(6-D)およびアミノ基を有する重合体(7-D)を得た。
窒素置換された内容積50リットルの反応容器に、シクロヘキサン(25kg)、テトラヒドロフラン(750g)、スチレン(750g)、及びn-ブチルリチウム(7.0g)を加え、50℃からの断熱重合を行った。反応完結後、温度を20℃として、1,3-ブタジエン(3,750g)を加え、断熱重合を行った。30分後、スチレン(500g)を加え、さらに重合を行い30分間反応させた。その後、合成例1と同様の方法により脱溶媒と乾燥を行うことで重合体(3-A)を得た。
窒素置換された内容積50リットルの反応容器に、シクロヘキサン(25kg)、テトラヒドロフラン(750g)、スチレン(750g)、及びn-ブチルリチウム(7.0g)を加え、50℃からの断熱重合を行った。反応完結後、温度を20℃として、1,3-ブタジエン(3,750g)を加え、断熱重合を行った。30分後、スチレン(500g)を加え重合を行った。水素圧1.0MPaを保つようにして30分反応させて重合停止した。次いで、四塩化ケイ素(1.7g)を加え、15分後に上記水添触媒(5.4g)、及びジエチルアルミニウムクロライド(2.1g)を加え、水素圧1.0MPaを保つようにして1時間反応させた。反応後、反応液を70℃、常圧に戻して反応容器より抜き出し、重合体溶液を得た。その後、合成例1と同様の方法により脱溶媒と乾燥を行うことで水添重合体(4-B)を得た。
実施例1~4及び比較例1~4では、(A)成分と(C)成分の混合物を(B)成分に含浸させることにより得られるフィルム状材料から作製された成形体について評価した。
表2に示す種類、質量部の(A)成分及び(C)成分に対して、酸化防止剤であるペンタエリスリトールテトラキス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオナート](商品名「アデカスタブAO-60」、ADEKA社製)0.1質量部と、トリス(2,4-ジ-tert-ブチルフェニル)ホスファイト(商品名「アデカスタブ2112」、ADEKA社製)0.1質量部とを添加し、室温でハンドブレンドした。次いで、この混合物を、東芝機械社製二軸押出機「TEM26SS」(型式名)に供給して溶融混練し、ペレットを得た。なお、溶融混練の際のシリンダー設定温度は、ホッパーの根元付近の温度を140℃とし、出口付近の温度が220℃となるように140℃~220℃の温度勾配をつけた。その後、上記ペレットを十分に乾燥した後、上記と同様にシリンダーの設定温度を140℃~220℃と温度勾配をつけたテクノベル社製二軸押出機「KZW15-30MG」(型式名)とTダイを用いて厚さ50μmのフィルムを作成した。作成したフィルムを30cm×19cmの大きさに裁断してプレス用フィルムを作成した。
プレス用フィルムの厚みを40μmにし、強化繊維の体積含有率(JIS K7075に準拠)を33%にした以外は実施例1と同じ方法により成形し、それぞれ縦15cm、横15cm、厚み1mmの成形体A2、A3、A5、A6、A7、A8、A11を得た。
プレス用フィルムの厚みを30μmにし、強化繊維の体積含有率(JIS K7075に準拠)を53%にした以外は実施例1と同じ方法により成形し、縦15cm、横15cm、厚み1mmの成形体A4を得た。
プレス用フィルムの厚みを25μmにし、強化繊維の体積含有率(JIS K7075に準拠)を63%にした以外は実施例1と同じ方法により成形し、縦15cm、横15cm、厚み1mmの成形体A9を得た。
プレス用フィルムの厚みを60μmにし、強化繊維の体積含有率(JIS K7075に準拠)を20%にした以外は実施例1と同じ方法により成形し、縦15cm、横15cm、厚み1mmの成形体A10を得た。
表2に示す種類、質量部の(C)成分に対して、酸化防止剤であるペンタエリスリトールテトラキス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオナート](商品名「アデカスタブAO-60」、ADEKA社製)0.1質量部と、トリス(2,4-ジ-tert-ブチルフェニル)ホスファイト(商品名「アデカスタブ2112」、ADEKA社製)0.1質量部、(A)成分の代わりに酸変性ポリプロピレン(商品名「ユーメックス 1001」、三洋化成工業社製)1質量部とを添加し、室温でドライブレンドした。次いで、この混合物を、東芝機械社製二軸押出機「TEM26SS」(型式名)に供給して溶融混練し、ペレットを得た。なお、溶融混練の際のシリンダー設定温度はホッパーの根元付近の温度を140℃とし、出口付近の温度が220℃となるように、140℃~220℃の温度勾配をつけた。その後、上記ペレットを十分に乾燥した後、上記と同様にシリンダーの設定温度を140℃~220℃と温度勾配をつけたテクノベル社製二軸押出機「KZW15-30MG」(型式名)とTダイを用いて厚さ50μmのフィルムを作成した。作成したフィルムを30cm×19cmの大きさに裁断してプレス用フィルムを二枚作成した。プレス用フィルムの厚みを40μmにし、強化繊維の体積含有率(JIS K7075に準拠)を33%にした以外は実施例1と同じ方法により成形し、縦15cm、横15cm、厚み1mmの成形体A12を得た。
表2に各実施例、各比較例の組成物の組成及び評価結果を示した。
[繊維(B)]
・B-1:東レ社製、品番「T700SC-12K-50C」、直径が約7μmであるフィラメントが12000本集束した束状のもの。
[熱可塑性樹脂(C)]
・PP:日本ポリプロ社製、ポリプロピレン「ノバテック MA1B」(商品名)
実施例8~14及び比較例6~8、比較例10では、(A)成分と(C)成分の混合物を(B’)成分に含浸させることにより得られるフィルム状材料から作製された成形体について評価した。
不織布(B’-1)~(B’-3)の製造方法は以下の通りである。東レ社製、品番「T700SC-12K-50C」(密度:1.80g/cm3)をカートリッジカッターでカットし、繊維長6mm、10mm、40mmのチョップド糸をそれぞれ得た。和光純薬工業(株)製、品名「n-ドデシルベンゼンスルホン酸ナトリウム」の1.5wt%水溶液100リットルを攪拌し、予め泡立てた分散液を作製した。この分散液に、得られたチョップド糸を投入し、10分間撹拌した後、抄紙面を有する抄紙機に流し込み、吸引により脱水して、その後、150℃の温度で2時間乾燥し、不織布(B’-1)~(B’-3)をそれぞれ得た。不織布(B’-1)は繊維長6mmのチョップド糸から得られた不織布であり、不織布(B’-2)は繊維長10mmのチョップド糸から得られた不織布であり、不織布(B’-3)は、繊維長40mmのチョップド糸から得られた不織布である。
表3に示す種類、質量部の(A)成分及び(C)成分に対して、酸化防止剤であるペンタエリスリトールテトラキス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオナート](商品名「アデカスタブAO-60」、ADEKA社製)0.1質量部と、トリス(2,4-ジ-tert-ブチルフェニル)ホスファイト(商品名「アデカスタブ2112」、ADEKA社製)0.1質量部とを添加し、室温でハンドブレンドした。次いで、この混合物を、東芝機械社製二軸押出機「TEM26SS」(型式名)に供給して溶融混練し、ペレットを得た。なお、溶融混練の際のシリンダー設定温度は、ホッパーの根元付近の温度を140℃とし、出口付近の温度が220℃となるように140℃~220℃の温度勾配をつけた。その後、上記ペレットを十分に乾燥した後、上記と同様にシリンダーの設定温度を140℃~220℃と温度勾配をつけたテクノベル社製二軸押出機「KZW15-30MG」(型式名)とTダイを用いて厚さ100μmのフィルムを作製した。作製したフィルムを15cm×15cmの大きさに裁断してプレス用フィルムを作製した。
(A)成分と(B’)成分を表3に示すものとした以外は実施例8と同じ方法により成形し、それぞれ縦15cm、横15cm、厚み1mmの成形体A14~A20、A24を得た。
(A)成分を表3に示すものとし、(B’)成分を不織布(B’-2)とし、Tダイを用いて作製したフィルムの厚みを50μmとした以外は実施例8と同じ方法により成形し、それぞれ縦15cm、横15cm、厚み1mmの成形体A21を得た。
(A)成分を表3に示すものとし、(B’)成分を不織布(B’-3)とし、Tダイを用いて作製したフィルムの厚みを150μmとした以外は実施例8と同じ方法により成形し、それぞれ縦15cm、横15cm、厚み1mmの成形体A22を得た。
表3に示す種類、質量部の(C)成分に対して、酸化防止剤であるペンタエリスリトールテトラキス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオナート](商品名「アデカスタブAO-60」、ADEKA社製)0.1質量部と、トリス(2,4-ジ-tert-ブチルフェニル)ホスファイト(商品名「アデカスタブ2112」、ADEKA社製)0.1質量部、(A)成分の代わりに酸変性ポリプロピレン(商品名「ユーメックス 1001」、三洋化成工業社製)3質量部とを添加し、室温でドライブレンドした。次いで、この混合物を、東芝機械社製二軸押出機「TEM26SS」(型式名)に供給して溶融混練し、ペレットを得た。なお、溶融混練の際のシリンダー設定温度はホッパーの根元付近の温度を140℃とし、出口付近の温度が220℃となるように、140℃~220℃の温度勾配をつけた。その後、上記ペレットを十分に乾燥した後、上記と同様にシリンダーの設定温度を140℃~220℃と温度勾配をつけたテクノベル社製二軸押出機「KZW15-30MG」(型式名)とTダイを用いて厚さ100μmのフィルムを作製した。作製したフィルムを15cm×15cmの大きさに裁断してプレス用フィルムを二枚作製した。実施例8と同じ方法によりそれぞれ不織布(B’-1)、(B’-4)を用いて成形し、縦15cm、横15cm、厚み1mmの成形体A23、A25をそれぞれ得た。
表3に各実施例、各比較例の組成物の組成及び評価結果を示した。
[熱可塑性樹脂(C)]
・PP:日本ポリプロ社製、ポリプロピレン「ノバテック MA1B」(商品名)
実施例15~16及び比較例12~14では、(A)成分を含むサイジング剤を(B)成分に塗布することにより得られるフィルム状材料から作製された成形体について評価した。
三角フラスコへ表4に示す重合体(A)100g及びシクロヘキサン900gを投入し、室温で溶解させて重合体(A)の溶液を作製した。その後、2Lガラスビーカーへ水350g、上記溶解液640g、花王社製エマルゲン147(商品名、ポリオキシエチレンラウリルエーテル)の25%水溶液を25.6g順次投入し、特殊機化工業社製ホモミキサーMARKII(商品名)を用いて13,000rpmにて10分間撹拌した。その後、東京理化器械社製ロータリーエバポレーター N-11(商品名)に移液し、シクロヘキサンを脱溶させ、重合体(A)の濃度が7%になるように調整した各水系乳化分散液(サイジング剤)を得た。なお、重合体(A)の平均粒子径は、動的光散乱式粒度分布測定装置(大塚電子株式会社製、型式「FPAR-1000」)を用いて、上記で得られた水系乳化分散液(サイジング剤)の粒度分布を測定し、その粒度分布から平均粒子径D50として求めた。
表4に示す種類および質量部の(C)成分に対して、酸化防止剤であるペンタエリスリトールテトラキス[3-(3,5-ジ-tert-ブチル-4-ヒドロキシフェニル)プロピオナート](商品名「アデカスタブAO-60」、ADEKA社製)0.1質量部と、トリス(2,4-ジ-tert-ブチルフェニル)ホスファイト(商品名「アデカスタブ2112」、ADEKA社製)0.1質量部とを添加し、室温でハンドブレンドした。次いで、この混合物を、シリンダーの設定温度を140℃~220℃と温度勾配をつけたテクノベル社製二軸押出機「KZW15-30MG」(型式名)とTダイを用いて厚さ40μmのフィルムを作成した。作成したフィルムを30cm×19cmの大きさに裁断してプレス用フィルムを二枚作成した。
(A)成分含むサイジング剤を塗布せず炭素繊維B-0をそのまま使用し、表4に記載された(C)成分を使用した以外は実施例15と同様にして、縦15cm、横15cm、厚み1mmの成形体B3を得た。
表4に各実施例、各比較例の組成物の組成及び評価結果を示した。
[熱可塑性樹脂(C)]
・PP:日本ポリプロ社製、ポリプロピレン「ノバテック MA1B」(商品名)
実施例17~21及び比較例15~19では、上記で作成した(A)成分を含むサイジング剤を(B’)成分に塗布することにより得られるフィルム状成形体から作製された成形体について評価した。
東レ社製、品番「T700SC-12K-50C」をアセトンに浸漬させてサイジング剤を除去した後、カートリッジカッターでカットし、繊維長6mm、10mm、40mmのチョップド糸をそれぞれ得た。和光純薬工業(株)製、品名「n-ドデシルベンゼンスルホン酸ナトリウム」の1.5wt%水溶液100リットルを攪拌し、予め泡立てた分散液を作製した。この分散液に、得られたチョップド糸を投入し、10分間撹拌した後、抄紙面を有する抄紙機に流し込み、吸引により脱水して、その後、150℃の温度で2時間乾燥し、不織布(B’-5)、(B’-6)、(B’-7)をそれぞれ得た。不織布(B’-5)は繊維長6mmのチョップド糸から得られた不織布であり、不織布(B’-6)は繊維長10mmのチョップド糸から得られた不織布であり、不織布(B’-7)は、繊維長40mmのチョップド糸から得られた不織布である。
不織布(B’-5)を、調整した水系乳化分散液(サイジング剤)を満たした処理浴に浸漬し、不織布(B’-5)に所定量の重合体(1-D)を付着させた。引き続き連続的に120℃のオーブンに5分間通して乾燥し、重合体(1-D)が付着した不織布(B’-5)を作製した。
(A)成分と(B’)成分を表5に示すものとした以外は実施例8と同じ方法により成形し、それぞれ縦15cm、横15cm、厚み1mmの成形体B7~B15を得た。
表5に各実施例、各比較例の組成物の組成及び評価結果を示した。
[熱可塑性樹脂(C)]
・PP:日本ポリプロ社製、ポリプロピレン「ノバテック MA1B」(商品名)
Claims (12)
- アミノ基を有する重合体(A)と、繊維(B)と、熱可塑性樹脂(C)と、を含有し、
前記熱可塑性樹脂(C)100質量部に対して前記繊維(B)を70質量部以上250質量部以下含有する、組成物。 - 前記アミノ基を有する重合体(A)の含有割合が、前記熱可塑性樹脂(C)100質量部に対して0.1質量部以上10質量部以下である、請求項1に記載の組成物。
- 前記繊維(B)が炭素繊維である、請求項1または請求項2に記載の組成物。
- 前記アミノ基を有する重合体(A)が共役ジエン系重合体である、請求項1ないし請求項3のいずれか一項に記載の組成物。
- 前記繊維(B)の繊維長が50mm以上である、請求項1ないし請求項4のいずれか一項に記載の組成物。
- アミノ基を有する重合体(A)と、
不織布(B’)と、
熱可塑性樹脂(C)と、
を含有する、組成物。 - 前記不織布(B’)がリサイクル繊維を含有する、請求項6に記載の組成物。
- 前記熱可塑性樹脂(C)100質量部に対して、前記不織布(B’)を50質量部以上150質量部以下含有する、請求項6または請求項7に記載の組成物。
- 前記不織布(B’)が炭素繊維を含む、請求項6ないし請求項8のいずれか一項に記載の組成物。
- 前記熱可塑性樹脂(C)100質量部に対して、前記アミノ基を有する重合体(A)を0.1質量部以上10質量部以下含有する、請求項6ないし請求項9のいずれか一項に記載の組成物。
- 前記熱可塑性樹脂(C)がオレフィン系樹脂である、請求項1ないし請求項10のいずれか一項に記載の組成物。
- 請求項1ないし請求項11のいずれか一項に記載の組成物を用いて作成された成形体。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201680050714.0A CN107922744A (zh) | 2015-09-02 | 2016-08-22 | 一种组合物和成型体 |
| JP2016563871A JP6187794B2 (ja) | 2015-09-02 | 2016-08-22 | 組成物及び成形体 |
| US15/756,653 US10570259B2 (en) | 2015-09-02 | 2016-08-22 | Composition and formed article |
| EP16841560.2A EP3345969A4 (en) | 2015-09-02 | 2016-08-22 | COMPOSITION AND FORM BODY |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-172649 | 2015-09-02 | ||
| JP2015172649 | 2015-09-02 | ||
| JP2015243818 | 2015-12-15 | ||
| JP2015-243818 | 2015-12-15 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017038528A1 true WO2017038528A1 (ja) | 2017-03-09 |
Family
ID=58187433
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/074368 WO2017038528A1 (ja) | 2015-09-02 | 2016-08-22 | 組成物及び成形体 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US10570259B2 (ja) |
| EP (1) | EP3345969A4 (ja) |
| JP (3) | JP6187794B2 (ja) |
| CN (1) | CN107922744A (ja) |
| TW (1) | TW201714952A (ja) |
| WO (1) | WO2017038528A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019108543A (ja) * | 2017-12-19 | 2019-07-04 | 上野製薬株式会社 | 熱可塑性樹脂プリプレグおよび複合材料 |
| WO2019189820A1 (ja) * | 2018-03-30 | 2019-10-03 | 日鉄ケミカル&マテリアル株式会社 | 熱可塑性樹脂組成物、繊維強化プラスチック成形用材料および成形物 |
| JP2020176244A (ja) * | 2019-04-22 | 2020-10-29 | 東洋インキScホールディングス株式会社 | ポリオレフィン樹脂組成物および成形体 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3936650B1 (en) * | 2019-03-07 | 2024-02-21 | Kuraray Co., Ltd. | Continuous long-fiber non-woven fabric, layered body, and composite material and production method therefor |
| JP6849850B1 (ja) * | 2019-09-25 | 2021-03-31 | 三ツ星ベルト株式会社 | ゴム組成物および摩擦伝動ベルト |
| WO2022185387A1 (ja) * | 2021-03-01 | 2022-09-09 | 中央精機株式会社 | 車両用ホイール、および、車両用ホイールの製造方法 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005213479A (ja) * | 2004-02-02 | 2005-08-11 | Idemitsu Kosan Co Ltd | ポリオレフィン系炭素繊維強化樹脂組成物及びそれからなる成形品 |
| JP2013227459A (ja) * | 2012-04-26 | 2013-11-07 | Henkel Japan Ltd | ホットメルト接着剤 |
| WO2014087767A1 (ja) * | 2012-12-05 | 2014-06-12 | 日本製紙株式会社 | 複合材料及びそれを用いた成形体 |
| JP2014234509A (ja) * | 2013-06-05 | 2014-12-15 | 三菱レイヨン株式会社 | 炭素繊維熱可塑性樹脂プリプレグおよび炭素繊維強化複合材料 |
| WO2015186667A1 (ja) * | 2014-06-02 | 2015-12-10 | 日本エラストマー株式会社 | 共役ジエン系重合体、その製造方法及びそれを含む組成物 |
Family Cites Families (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2134909B (en) | 1983-01-20 | 1986-08-20 | Asahi Chemical Ind | Catalytic hydrogenation of conjugated diene polymer |
| JPS59133203A (ja) | 1983-01-20 | 1984-07-31 | Asahi Chem Ind Co Ltd | 重合体の水添方法 |
| JPS62218403A (ja) | 1986-03-19 | 1987-09-25 | Nippon Zeon Co Ltd | 共役ジエン系重合体の水素化方法 |
| JPS635401A (ja) | 1986-06-26 | 1988-01-11 | Omron Tateisi Electronics Co | プログラム温度調節器の比例制御方法 |
| JP2718059B2 (ja) | 1988-04-28 | 1998-02-25 | 日本合成ゴム株式会社 | 重合体の水素添加方法および触媒 |
| US5039755A (en) | 1990-05-29 | 1991-08-13 | Shell Oil Company | Selective hydrogenation of conjugated diolefin polymers |
| JP3134504B2 (ja) | 1991-05-30 | 2001-02-13 | ジェイエスアール株式会社 | 水添ジエン系共重合体およびその組成物 |
| JPH05112657A (ja) | 1991-10-21 | 1993-05-07 | Mitsubishi Kasei Corp | 熱可塑性樹脂強化用炭素繊維強化樹脂組成物および炭素繊維強化熱可塑性樹脂複合材 |
| JP3166137B2 (ja) | 1991-11-28 | 2001-05-14 | ジェイエスアール株式会社 | オレフィン性不飽和重合体の水素化方法および水素添加触媒 |
| US5270274A (en) | 1991-11-28 | 1993-12-14 | Japan Synthetic Rubber Co., Ltd. | Catalyst composition for hydrogenating olefinically unsaturated polymers |
| JP3227678B2 (ja) | 1992-01-29 | 2001-11-12 | ジェイエスアール株式会社 | オレフィン性不飽和重合体の水素添加方法および水素添加触媒 |
| JP3326772B2 (ja) | 1992-01-29 | 2002-09-24 | ジェイエスアール株式会社 | オレフィン性不飽和重合体の水素化方法および水素添加触媒 |
| JPH0732465A (ja) | 1993-07-21 | 1995-02-03 | Nippon Steel Corp | 繊維強化熱可塑性樹脂張り出しまたは深絞り成形品およびその成形方法 |
| WO1995021202A1 (fr) * | 1994-02-01 | 1995-08-10 | Asahi Kasei Kogyo Kabushiki Kaisha | Nouveau polymere modifie contenant un motif structurel moleculaire cyclique |
| US5596041A (en) | 1994-04-04 | 1997-01-21 | Japan Synthetic Rubber Co., Ltd. | Hydrogenated block copolymer and hydrogenated block copolymer compositon |
| JP3360411B2 (ja) | 1994-04-04 | 2002-12-24 | ジェイエスアール株式会社 | 水添ブロック共重合体組成物 |
| JP3572823B2 (ja) | 1996-09-26 | 2004-10-06 | 東海化成工業株式会社 | 繊維強化熱可塑性樹脂成形体の製造方法 |
| JP3951439B2 (ja) | 1998-04-10 | 2007-08-01 | Jsr株式会社 | オレフイン性不飽和化合物の水素添加触媒および水素添加方法 |
| JP3777810B2 (ja) | 1998-07-24 | 2006-05-24 | Jsr株式会社 | オレフイン性不飽和化合物の水素添加触媒および水素添加方法 |
| JP3988495B2 (ja) | 2001-03-26 | 2007-10-10 | Jsr株式会社 | 水添変性重合体及びその製造方法並びにそれを含む組成物 |
| ES2307678T3 (es) | 2001-03-26 | 2008-12-01 | Jsr Corporation | Polimero modificado hidrogenado, procedimiento para producir el mismo y composicion que contiene el mismo. |
| CN1274719C (zh) | 2001-09-27 | 2006-09-13 | Jsr株式会社 | 共轭二烯(共)聚合橡胶、(共)聚合橡胶的制造方法、橡胶组合物、复合体以及轮胎 |
| US7419721B2 (en) * | 2003-12-19 | 2008-09-02 | Ppg Industries Ohio, Inc. | Sizing composition and glass fiber reinforced thermoplastic resin |
| WO2006016512A1 (ja) | 2004-08-10 | 2006-02-16 | Jsr Corporation | ゴム組成物およびタイヤ |
| CN101305055A (zh) * | 2005-09-29 | 2008-11-12 | 东丽株式会社 | 纤维增强热塑性树脂组合物、其制造方法、及热塑性树脂用碳纤维 |
| WO2007037260A1 (ja) | 2005-09-29 | 2007-04-05 | Toray Industries, Inc. | 繊維強化熱可塑性樹脂組成物、その製造方法、及び熱可塑性樹脂用炭素繊維 |
| JP2008169344A (ja) | 2007-01-15 | 2008-07-24 | Toray Ind Inc | 熱可塑性樹脂組成物 |
| JP5402112B2 (ja) | 2009-03-11 | 2014-01-29 | Jsr株式会社 | ゴム組成物及び空気入りタイヤ |
| US20110319519A1 (en) | 2009-03-11 | 2011-12-29 | Jsr Corporation | Rubber composition and pneumatic tire |
| EP2476799B1 (en) | 2009-09-09 | 2020-01-01 | Mitsubishi Chemical Corporation | Carbon fiber bundle and method for producing same |
| JP5855401B2 (ja) | 2011-09-21 | 2016-02-09 | 三菱レイヨン株式会社 | 繊維強化熱可塑性樹脂成形品の製造方法 |
| JP5899949B2 (ja) | 2012-01-18 | 2016-04-06 | 三菱レイヨン株式会社 | 炭素繊維の製造方法 |
| JP6107155B2 (ja) | 2012-01-20 | 2017-04-05 | 東レ株式会社 | プリプレグ |
| ES2688886T3 (es) | 2012-01-20 | 2018-11-07 | Toray Industries, Inc. | Composición de resina de polipropileno reforzada con fibras, material de moldeo y material preimpregnado |
| JP5933478B2 (ja) * | 2012-03-28 | 2016-06-08 | 積水化成品工業株式会社 | 積層発泡シート |
| JP6229654B2 (ja) | 2012-07-20 | 2017-11-15 | Jsr株式会社 | 変性共役ジエン系重合体、重合体組成物、架橋体及びタイヤの製造方法 |
| US10093777B2 (en) | 2012-12-26 | 2018-10-09 | Toray Industries, Inc. | Fiber-reinforced resin sheet, integrated molded product and process for producing same |
| JP5626330B2 (ja) | 2012-12-26 | 2014-11-19 | 東レ株式会社 | 繊維強化樹脂シート、成形体、一体化成形品およびそれらの製造方法、ならびに実装部材 |
| JP2014196584A (ja) | 2013-03-29 | 2014-10-16 | トヨタ自動車株式会社 | 不織布の製造方法および複合材料の製造方法 |
| JPWO2016009858A1 (ja) | 2014-07-16 | 2017-04-27 | Jsr株式会社 | 成形体の製造方法 |
| JP2017048314A (ja) | 2015-09-02 | 2017-03-09 | Jsr株式会社 | 組成物及び成形体 |
| JP2017048313A (ja) | 2015-09-02 | 2017-03-09 | Jsr株式会社 | 組成物及び成形体 |
-
2016
- 2016-08-09 TW TW105125305A patent/TW201714952A/zh unknown
- 2016-08-22 US US15/756,653 patent/US10570259B2/en active Active
- 2016-08-22 CN CN201680050714.0A patent/CN107922744A/zh active Pending
- 2016-08-22 WO PCT/JP2016/074368 patent/WO2017038528A1/ja active Application Filing
- 2016-08-22 EP EP16841560.2A patent/EP3345969A4/en not_active Withdrawn
- 2016-08-22 JP JP2016563871A patent/JP6187794B2/ja active Active
-
2017
- 2017-02-17 JP JP2017027723A patent/JP2017110236A/ja active Pending
- 2017-02-17 JP JP2017027722A patent/JP6187795B2/ja active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005213479A (ja) * | 2004-02-02 | 2005-08-11 | Idemitsu Kosan Co Ltd | ポリオレフィン系炭素繊維強化樹脂組成物及びそれからなる成形品 |
| JP2013227459A (ja) * | 2012-04-26 | 2013-11-07 | Henkel Japan Ltd | ホットメルト接着剤 |
| WO2014087767A1 (ja) * | 2012-12-05 | 2014-06-12 | 日本製紙株式会社 | 複合材料及びそれを用いた成形体 |
| JP2014234509A (ja) * | 2013-06-05 | 2014-12-15 | 三菱レイヨン株式会社 | 炭素繊維熱可塑性樹脂プリプレグおよび炭素繊維強化複合材料 |
| WO2015186667A1 (ja) * | 2014-06-02 | 2015-12-10 | 日本エラストマー株式会社 | 共役ジエン系重合体、その製造方法及びそれを含む組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3345969A4 * |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2019108543A (ja) * | 2017-12-19 | 2019-07-04 | 上野製薬株式会社 | 熱可塑性樹脂プリプレグおよび複合材料 |
| JP7312548B2 (ja) | 2017-12-19 | 2023-07-21 | 上野製薬株式会社 | 熱可塑性樹脂プリプレグおよび複合材料 |
| WO2019189820A1 (ja) * | 2018-03-30 | 2019-10-03 | 日鉄ケミカル&マテリアル株式会社 | 熱可塑性樹脂組成物、繊維強化プラスチック成形用材料および成形物 |
| CN111971343A (zh) * | 2018-03-30 | 2020-11-20 | 日铁化学材料株式会社 | 热塑性树脂组合物、纤维强化塑料成型用材料及成型物 |
| JPWO2019189820A1 (ja) * | 2018-03-30 | 2021-04-15 | 日鉄ケミカル&マテリアル株式会社 | 熱可塑性樹脂組成物、繊維強化プラスチック成形用材料および成形物 |
| JP7273795B2 (ja) | 2018-03-30 | 2023-05-15 | 日鉄ケミカル&マテリアル株式会社 | 熱可塑性樹脂組成物、繊維強化プラスチック成形用材料および成形物 |
| JP2020176244A (ja) * | 2019-04-22 | 2020-10-29 | 東洋インキScホールディングス株式会社 | ポリオレフィン樹脂組成物および成形体 |
| JP7338222B2 (ja) | 2019-04-22 | 2023-09-05 | 東洋インキScホールディングス株式会社 | ポリオレフィン樹脂組成物および成形体 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN107922744A (zh) | 2018-04-17 |
| JP6187794B2 (ja) | 2017-08-30 |
| JP6187795B2 (ja) | 2017-08-30 |
| TW201714952A (zh) | 2017-05-01 |
| EP3345969A1 (en) | 2018-07-11 |
| JPWO2017038528A1 (ja) | 2017-09-07 |
| US10570259B2 (en) | 2020-02-25 |
| US20180258239A1 (en) | 2018-09-13 |
| EP3345969A4 (en) | 2019-01-16 |
| JP2017110236A (ja) | 2017-06-22 |
| JP2017110235A (ja) | 2017-06-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6187795B2 (ja) | 組成物及び成形体 | |
| JP2016065354A (ja) | サイジング剤、組成物及び成形体 | |
| CN104066783B (zh) | 纤维增强聚丙烯树脂组合物、成型材料及预浸料坯 | |
| KR101669377B1 (ko) | 수지 강화용 탄소 섬유속 및 그의 제조 방법, 및 탄소 섬유 강화 열가소성 수지 조성물 및 그의 성형품 | |
| CN105722911B (zh) | 树脂组合物及其成型体 | |
| KR101288676B1 (ko) | 섬유 처리용 수분산체 | |
| Fu et al. | Combined effect of interfacial strength and fiber orientation on mechanical performance of short Kevlar fiber reinforced olefin block copolymer | |
| KR20150099710A (ko) | 섬유 강화 열가소성 수지 성형품, 섬유 강화 열가소성 수지 성형 재료, 및 섬유 강화 열가소성 수지 성형 재료의 제조 방법 | |
| CN103030884A (zh) | 一种汽车搪塑仪表板骨架用的聚丙烯组合物及其制备方法 | |
| JP2017048313A (ja) | 組成物及び成形体 | |
| JPWO2018101022A1 (ja) | 繊維強化樹脂用組成物及びその製造方法、繊維強化樹脂、並びに成形体 | |
| JP2011162767A (ja) | 炭素繊維強化ポリフェニレンスルフィド樹脂組成物、ならびにそれを用いた成形材料および成形品 | |
| WO2014045981A1 (ja) | 強化繊維束及びそれを用いた複合材料 | |
| CN107141609A (zh) | 一种耐刮擦聚丙烯复合材料及其制备方法 | |
| JP2008138294A (ja) | 繊維、繊維の構造体およびそれら製造方法 | |
| JP2011021184A (ja) | 樹脂組成物およびその製造方法 | |
| JP2017048314A (ja) | 組成物及び成形体 | |
| JP2010202797A (ja) | 天然繊維を含有する樹脂組成物及び成形体 | |
| JP3326957B2 (ja) | 繊維強化熱可塑性組成物及びその製造方法 | |
| JP2018058947A (ja) | 繊維強化樹脂用組成物、繊維強化樹脂及び成形体 | |
| JP2018058948A (ja) | 繊維強化樹脂用組成物、繊維強化樹脂及び成形体 | |
| JP2008133328A (ja) | ポリフェニレンエーテル系樹脂組成物の製造方法 | |
| JP2012149246A (ja) | ポリプロピレン樹脂組成物、およびその製造方法、ならびにそれを成形してなる成形体 | |
| JP5311050B2 (ja) | 弾性フィラメント用組成物、弾性フィラメントおよび伸縮性シート | |
| CN104559205B (zh) | 一种原位增韧聚丙烯合金的制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2016563871 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16841560 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15756653 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2016841560 Country of ref document: EP |