WO2012039372A1 - インクジェット用硬化性組成物及び電子部品の製造方法 - Google Patents
インクジェット用硬化性組成物及び電子部品の製造方法 Download PDFInfo
- Publication number
- WO2012039372A1 WO2012039372A1 PCT/JP2011/071327 JP2011071327W WO2012039372A1 WO 2012039372 A1 WO2012039372 A1 WO 2012039372A1 JP 2011071327 W JP2011071327 W JP 2011071327W WO 2012039372 A1 WO2012039372 A1 WO 2012039372A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- curable composition
- inkjet
- compound
- meth
- group
- Prior art date
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 253
- 238000004519 manufacturing process Methods 0.000 title claims description 19
- 150000001875 compounds Chemical class 0.000 claims abstract description 229
- 239000000047 product Substances 0.000 claims abstract description 114
- QGBSISYHAICWAH-UHFFFAOYSA-N dicyandiamide Chemical compound NC(N)=NC#N QGBSISYHAICWAH-UHFFFAOYSA-N 0.000 claims abstract description 64
- 125000000524 functional group Chemical group 0.000 claims abstract description 55
- 150000004292 cyclic ethers Chemical group 0.000 claims abstract description 52
- 239000003795 chemical substances by application Substances 0.000 claims abstract description 41
- 238000000034 method Methods 0.000 claims abstract description 41
- 239000007795 chemical reaction product Substances 0.000 claims abstract description 6
- 238000006243 chemical reaction Methods 0.000 claims description 77
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 claims description 38
- 239000003999 initiator Substances 0.000 claims description 36
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 claims description 33
- 125000003367 polycyclic group Chemical group 0.000 claims description 20
- -1 dihydroxycyclopentadienyl Chemical group 0.000 claims description 19
- 239000003505 polymerization initiator Substances 0.000 claims description 19
- 125000003700 epoxy group Chemical group 0.000 claims description 13
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 claims description 11
- 125000003118 aryl group Chemical group 0.000 claims description 10
- DTGKSKDOIYIVQL-WEDXCCLWSA-N (+)-borneol Chemical group C1C[C@@]2(C)[C@@H](O)C[C@@H]1C2(C)C DTGKSKDOIYIVQL-WEDXCCLWSA-N 0.000 claims description 5
- OTLDLKLSNZMTTA-UHFFFAOYSA-N octahydro-1h-4,7-methanoindene-1,5-diyldimethanol Chemical compound C1C2C3C(CO)CCC3C1C(CO)C2 OTLDLKLSNZMTTA-UHFFFAOYSA-N 0.000 claims description 5
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 4
- 229910052739 hydrogen Inorganic materials 0.000 claims description 4
- 239000001257 hydrogen Substances 0.000 claims description 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 3
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 claims description 3
- 239000000843 powder Substances 0.000 claims description 3
- 230000008569 process Effects 0.000 claims description 2
- 238000009413 insulation Methods 0.000 abstract description 26
- 238000011417 postcuring Methods 0.000 abstract 1
- 238000001723 curing Methods 0.000 description 41
- 239000011345 viscous material Substances 0.000 description 30
- 239000000758 substrate Substances 0.000 description 27
- 239000004593 Epoxy Substances 0.000 description 21
- 239000003960 organic solvent Substances 0.000 description 16
- 230000001965 increasing effect Effects 0.000 description 15
- 229910000679 solder Inorganic materials 0.000 description 13
- 229920001187 thermosetting polymer Chemical group 0.000 description 13
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 10
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 10
- 239000002904 solvent Substances 0.000 description 10
- 239000011248 coating agent Substances 0.000 description 9
- 238000000576 coating method Methods 0.000 description 9
- 239000011889 copper foil Substances 0.000 description 9
- 239000000463 material Substances 0.000 description 9
- 239000007870 radical polymerization initiator Substances 0.000 description 9
- 239000007787 solid Substances 0.000 description 9
- 238000011156 evaluation Methods 0.000 description 7
- 238000010438 heat treatment Methods 0.000 description 7
- 238000003860 storage Methods 0.000 description 7
- HEQOJEGTZCTHCF-UHFFFAOYSA-N 2-amino-1-phenylethanone Chemical class NCC(=O)C1=CC=CC=C1 HEQOJEGTZCTHCF-UHFFFAOYSA-N 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 6
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 150000005846 sugar alcohols Polymers 0.000 description 6
- 238000003786 synthesis reaction Methods 0.000 description 6
- 239000002518 antifoaming agent Substances 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- 238000006116 polymerization reaction Methods 0.000 description 5
- 238000003892 spreading Methods 0.000 description 5
- 230000007480 spreading Effects 0.000 description 5
- 238000009736 wetting Methods 0.000 description 5
- LWRBVKNFOYUCNP-UHFFFAOYSA-N 2-methyl-1-(4-methylsulfanylphenyl)-2-morpholin-4-ylpropan-1-one Chemical compound C1=CC(SC)=CC=C1C(=O)C(C)(C)N1CCOCC1 LWRBVKNFOYUCNP-UHFFFAOYSA-N 0.000 description 4
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 4
- ISAOCJYIOMOJEB-UHFFFAOYSA-N benzoin Chemical compound C=1C=CC=CC=1C(O)C(=O)C1=CC=CC=C1 ISAOCJYIOMOJEB-UHFFFAOYSA-N 0.000 description 4
- PXKLMJQFEQBVLD-UHFFFAOYSA-N bisphenol F Chemical compound C1=CC(O)=CC=C1CC1=CC=C(O)C=C1 PXKLMJQFEQBVLD-UHFFFAOYSA-N 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- GYZLOYUZLJXAJU-UHFFFAOYSA-N diglycidyl ether Chemical class C1OC1COCC1CO1 GYZLOYUZLJXAJU-UHFFFAOYSA-N 0.000 description 4
- 230000000977 initiatory effect Effects 0.000 description 4
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 4
- 229910052753 mercury Inorganic materials 0.000 description 4
- 230000002194 synthesizing effect Effects 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 3
- UHFFVFAKEGKNAQ-UHFFFAOYSA-N 2-benzyl-2-(dimethylamino)-1-(4-morpholin-4-ylphenyl)butan-1-one Chemical compound C=1C=C(N2CCOCC2)C=CC=1C(=O)C(CC)(N(C)C)CC1=CC=CC=C1 UHFFVFAKEGKNAQ-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- 229920000298 Cellophane Polymers 0.000 description 3
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical class CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- FQYUMYWMJTYZTK-UHFFFAOYSA-N Phenyl glycidyl ether Chemical compound C1OC1COC1=CC=CC=C1 FQYUMYWMJTYZTK-UHFFFAOYSA-N 0.000 description 3
- 244000028419 Styrax benzoin Species 0.000 description 3
- 235000000126 Styrax benzoin Nutrition 0.000 description 3
- 235000008411 Sumatra benzointree Nutrition 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 229960002130 benzoin Drugs 0.000 description 3
- 239000012965 benzophenone Substances 0.000 description 3
- 238000013329 compounding Methods 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 235000019382 gum benzoic Nutrition 0.000 description 3
- 150000002460 imidazoles Chemical class 0.000 description 3
- 238000007689 inspection Methods 0.000 description 3
- 230000001678 irradiating effect Effects 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 229920003986 novolac Polymers 0.000 description 3
- 238000001029 thermal curing Methods 0.000 description 3
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical compound C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 2
- YXIWHUQXZSMYRE-UHFFFAOYSA-N 1,3-benzothiazole-2-thiol Chemical compound C1=CC=C2SC(S)=NC2=C1 YXIWHUQXZSMYRE-UHFFFAOYSA-N 0.000 description 2
- YSUQLAYJZDEMOT-UHFFFAOYSA-N 2-(butoxymethyl)oxirane Chemical compound CCCCOCC1CO1 YSUQLAYJZDEMOT-UHFFFAOYSA-N 0.000 description 2
- LCZVSXRMYJUNFX-UHFFFAOYSA-N 2-[2-(2-hydroxypropoxy)propoxy]propan-1-ol Chemical compound CC(O)COC(C)COC(C)CO LCZVSXRMYJUNFX-UHFFFAOYSA-N 0.000 description 2
- NJWGQARXZDRHCD-UHFFFAOYSA-N 2-methylanthraquinone Chemical compound C1=CC=C2C(=O)C3=CC(C)=CC=C3C(=O)C2=C1 NJWGQARXZDRHCD-UHFFFAOYSA-N 0.000 description 2
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 2
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- QIGBRXMKCJKVMJ-UHFFFAOYSA-N Hydroquinone Chemical compound OC1=CC=C(O)C=C1 QIGBRXMKCJKVMJ-UHFFFAOYSA-N 0.000 description 2
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 229910000831 Steel Inorganic materials 0.000 description 2
- ZJCCRDAZUWHFQH-UHFFFAOYSA-N Trimethylolpropane Chemical compound CCC(CO)(CO)CO ZJCCRDAZUWHFQH-UHFFFAOYSA-N 0.000 description 2
- DAKWPKUUDNSNPN-UHFFFAOYSA-N Trimethylolpropane triacrylate Chemical compound C=CC(=O)OCC(CC)(COC(=O)C=C)COC(=O)C=C DAKWPKUUDNSNPN-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 150000008062 acetophenones Chemical class 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 125000002947 alkylene group Chemical group 0.000 description 2
- 150000004056 anthraquinones Chemical class 0.000 description 2
- 150000008366 benzophenones Chemical class 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000003086 colorant Substances 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- VFHVQBAGLAREND-UHFFFAOYSA-N diphenylphosphoryl-(2,4,6-trimethylphenyl)methanone Chemical compound CC1=CC(C)=CC(C)=C1C(=O)P(=O)(C=1C=CC=CC=1)C1=CC=CC=C1 VFHVQBAGLAREND-UHFFFAOYSA-N 0.000 description 2
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 239000003822 epoxy resin Substances 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- LEQAOMBKQFMDFZ-UHFFFAOYSA-N glyoxal Chemical compound O=CC=O LEQAOMBKQFMDFZ-UHFFFAOYSA-N 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 238000007641 inkjet printing Methods 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 239000000178 monomer Substances 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- 150000002896 organic halogen compounds Chemical class 0.000 description 2
- 125000003566 oxetanyl group Chemical group 0.000 description 2
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 2
- 150000002989 phenols Chemical class 0.000 description 2
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 2
- 229920000647 polyepoxide Polymers 0.000 description 2
- 229920000728 polyester Polymers 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920001296 polysiloxane Polymers 0.000 description 2
- 238000007639 printing Methods 0.000 description 2
- WQGWDDDVZFFDIG-UHFFFAOYSA-N pyrogallol Chemical compound OC1=CC=CC(O)=C1O WQGWDDDVZFFDIG-UHFFFAOYSA-N 0.000 description 2
- 239000010959 steel Substances 0.000 description 2
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 150000003672 ureas Chemical class 0.000 description 2
- QNODIIQQMGDSEF-UHFFFAOYSA-N (1-hydroxycyclohexyl)-phenylmethanone Chemical compound C=1C=CC=CC=1C(=O)C1(O)CCCCC1 QNODIIQQMGDSEF-UHFFFAOYSA-N 0.000 description 1
- DLDWUFCUUXXYTB-UHFFFAOYSA-N (2-oxo-1,2-diphenylethyl) 4-methylbenzenesulfonate Chemical compound C1=CC(C)=CC=C1S(=O)(=O)OC(C=1C=CC=CC=1)C(=O)C1=CC=CC=C1 DLDWUFCUUXXYTB-UHFFFAOYSA-N 0.000 description 1
- PSGCQDPCAWOCSH-UHFFFAOYSA-N (4,7,7-trimethyl-3-bicyclo[2.2.1]heptanyl) prop-2-enoate Chemical compound C1CC2(C)C(OC(=O)C=C)CC1C2(C)C PSGCQDPCAWOCSH-UHFFFAOYSA-N 0.000 description 1
- XKSUVRWJZCEYQQ-UHFFFAOYSA-N 1,1-dimethoxyethylbenzene Chemical compound COC(C)(OC)C1=CC=CC=C1 XKSUVRWJZCEYQQ-UHFFFAOYSA-N 0.000 description 1
- MSAHTMIQULFMRG-UHFFFAOYSA-N 1,2-diphenyl-2-propan-2-yloxyethanone Chemical compound C=1C=CC=CC=1C(OC(C)C)C(=O)C1=CC=CC=C1 MSAHTMIQULFMRG-UHFFFAOYSA-N 0.000 description 1
- YHMYGUUIMTVXNW-UHFFFAOYSA-N 1,3-dihydrobenzimidazole-2-thione Chemical compound C1=CC=C2NC(S)=NC2=C1 YHMYGUUIMTVXNW-UHFFFAOYSA-N 0.000 description 1
- BOCJQSFSGAZAPQ-UHFFFAOYSA-N 1-chloroanthracene-9,10-dione Chemical compound O=C1C2=CC=CC=C2C(=O)C2=C1C=CC=C2Cl BOCJQSFSGAZAPQ-UHFFFAOYSA-N 0.000 description 1
- WJFKNYWRSNBZNX-UHFFFAOYSA-N 10H-phenothiazine Chemical compound C1=CC=C2NC3=CC=CC=C3SC2=C1 WJFKNYWRSNBZNX-UHFFFAOYSA-N 0.000 description 1
- CERJZAHSUZVMCH-UHFFFAOYSA-N 2,2-dichloro-1-phenylethanone Chemical compound ClC(Cl)C(=O)C1=CC=CC=C1 CERJZAHSUZVMCH-UHFFFAOYSA-N 0.000 description 1
- GIMQKKFOOYOQGB-UHFFFAOYSA-N 2,2-diethoxy-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(OCC)(OCC)C(=O)C1=CC=CC=C1 GIMQKKFOOYOQGB-UHFFFAOYSA-N 0.000 description 1
- KWVGIHKZDCUPEU-UHFFFAOYSA-N 2,2-dimethoxy-2-phenylacetophenone Chemical compound C=1C=CC=CC=1C(OC)(OC)C(=O)C1=CC=CC=C1 KWVGIHKZDCUPEU-UHFFFAOYSA-N 0.000 description 1
- BRKORVYTKKLNKX-UHFFFAOYSA-N 2,4-di(propan-2-yl)thioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC(C(C)C)=CC(C(C)C)=C3SC2=C1 BRKORVYTKKLNKX-UHFFFAOYSA-N 0.000 description 1
- BTJPUDCSZVCXFQ-UHFFFAOYSA-N 2,4-diethylthioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC(CC)=CC(CC)=C3SC2=C1 BTJPUDCSZVCXFQ-UHFFFAOYSA-N 0.000 description 1
- LCHAFMWSFCONOO-UHFFFAOYSA-N 2,4-dimethylthioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC(C)=CC(C)=C3SC2=C1 LCHAFMWSFCONOO-UHFFFAOYSA-N 0.000 description 1
- STMDPCBYJCIZOD-UHFFFAOYSA-N 2-(2,4-dinitroanilino)-4-methylpentanoic acid Chemical compound CC(C)CC(C(O)=O)NC1=CC=C([N+]([O-])=O)C=C1[N+]([O-])=O STMDPCBYJCIZOD-UHFFFAOYSA-N 0.000 description 1
- PUBNJSZGANKUGX-UHFFFAOYSA-N 2-(dimethylamino)-2-[(4-methylphenyl)methyl]-1-(4-morpholin-4-ylphenyl)butan-1-one Chemical compound C=1C=C(N2CCOCC2)C=CC=1C(=O)C(CC)(N(C)C)CC1=CC=C(C)C=C1 PUBNJSZGANKUGX-UHFFFAOYSA-N 0.000 description 1
- BXGYYDRIMBPOMN-UHFFFAOYSA-N 2-(hydroxymethoxy)ethoxymethanol Chemical compound OCOCCOCO BXGYYDRIMBPOMN-UHFFFAOYSA-N 0.000 description 1
- LVUNJHSREDQSGA-UHFFFAOYSA-N 2-benzyl-n,n-dimethyl-1-(4-morpholin-4-ylphenyl)butan-2-amine Chemical compound C=1C=C(N2CCOCC2)C=CC=1CC(N(C)C)(CC)CC1=CC=CC=C1 LVUNJHSREDQSGA-UHFFFAOYSA-N 0.000 description 1
- ZCDADJXRUCOCJE-UHFFFAOYSA-N 2-chlorothioxanthen-9-one Chemical compound C1=CC=C2C(=O)C3=CC(Cl)=CC=C3SC2=C1 ZCDADJXRUCOCJE-UHFFFAOYSA-N 0.000 description 1
- KMNCBSZOIQAUFX-UHFFFAOYSA-N 2-ethoxy-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(OCC)C(=O)C1=CC=CC=C1 KMNCBSZOIQAUFX-UHFFFAOYSA-N 0.000 description 1
- SJEBAWHUJDUKQK-UHFFFAOYSA-N 2-ethylanthraquinone Chemical compound C1=CC=C2C(=O)C3=CC(CC)=CC=C3C(=O)C2=C1 SJEBAWHUJDUKQK-UHFFFAOYSA-N 0.000 description 1
- FLFWJIBUZQARMD-UHFFFAOYSA-N 2-mercapto-1,3-benzoxazole Chemical compound C1=CC=C2OC(S)=NC2=C1 FLFWJIBUZQARMD-UHFFFAOYSA-N 0.000 description 1
- BQZJOQXSCSZQPS-UHFFFAOYSA-N 2-methoxy-1,2-diphenylethanone Chemical compound C=1C=CC=CC=1C(OC)C(=O)C1=CC=CC=C1 BQZJOQXSCSZQPS-UHFFFAOYSA-N 0.000 description 1
- QTWJRLJHJPIABL-UHFFFAOYSA-N 2-methylphenol;3-methylphenol;4-methylphenol Chemical compound CC1=CC=C(O)C=C1.CC1=CC=CC(O)=C1.CC1=CC=CC=C1O QTWJRLJHJPIABL-UHFFFAOYSA-N 0.000 description 1
- YTPSFXZMJKMUJE-UHFFFAOYSA-N 2-tert-butylanthracene-9,10-dione Chemical compound C1=CC=C2C(=O)C3=CC(C(C)(C)C)=CC=C3C(=O)C2=C1 YTPSFXZMJKMUJE-UHFFFAOYSA-N 0.000 description 1
- ARXVXVOLXMVYIT-UHFFFAOYSA-N 3-methylbutyl 2-(dimethylamino)benzoate Chemical compound CC(C)CCOC(=O)C1=CC=CC=C1N(C)C ARXVXVOLXMVYIT-UHFFFAOYSA-N 0.000 description 1
- JIGUICYYOYEXFS-UHFFFAOYSA-N 3-tert-butylbenzene-1,2-diol Chemical compound CC(C)(C)C1=CC=CC(O)=C1O JIGUICYYOYEXFS-UHFFFAOYSA-N 0.000 description 1
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical compound C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 1
- 239000006087 Silane Coupling Agent Substances 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- VEBCLRKUSAGCDF-UHFFFAOYSA-N ac1mi23b Chemical compound C1C2C3C(COC(=O)C=C)CCC3C1C(COC(=O)C=C)C2 VEBCLRKUSAGCDF-UHFFFAOYSA-N 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 230000005260 alpha ray Effects 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 125000005577 anthracene group Chemical group 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- RWCCWEUUXYIKHB-UHFFFAOYSA-N benzophenone Chemical compound C=1C=CC=CC=1C(=O)C1=CC=CC=C1 RWCCWEUUXYIKHB-UHFFFAOYSA-N 0.000 description 1
- 230000005250 beta ray Effects 0.000 description 1
- JRPRCOLKIYRSNH-UHFFFAOYSA-N bis(oxiran-2-ylmethyl) benzene-1,2-dicarboxylate Chemical class C=1C=CC=C(C(=O)OCC2OC2)C=1C(=O)OCC1CO1 JRPRCOLKIYRSNH-UHFFFAOYSA-N 0.000 description 1
- VYHBFRJRBHMIQZ-UHFFFAOYSA-N bis[4-(diethylamino)phenyl]methanone Chemical compound C1=CC(N(CC)CC)=CC=C1C(=O)C1=CC=C(N(CC)CC)C=C1 VYHBFRJRBHMIQZ-UHFFFAOYSA-N 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 125000005578 chrysene group Chemical group 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229930003836 cresol Natural products 0.000 description 1
- 125000006841 cyclic skeleton Chemical group 0.000 description 1
- VEIOBOXBGYWJIT-UHFFFAOYSA-N cyclohexane;methanol Chemical compound OC.OC.C1CCCCC1 VEIOBOXBGYWJIT-UHFFFAOYSA-N 0.000 description 1
- WVIIMZNLDWSIRH-UHFFFAOYSA-N cyclohexylcyclohexane Chemical compound C1CCCCC1C1CCCCC1 WVIIMZNLDWSIRH-UHFFFAOYSA-N 0.000 description 1
- 125000000664 diazo group Chemical group [N-]=[N+]=[*] 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 238000010894 electron beam technology Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- ORBFAMHUKZLWSD-UHFFFAOYSA-N ethyl 2-(dimethylamino)benzoate Chemical compound CCOC(=O)C1=CC=CC=C1N(C)C ORBFAMHUKZLWSD-UHFFFAOYSA-N 0.000 description 1
- 230000005251 gamma ray Effects 0.000 description 1
- 238000001879 gelation Methods 0.000 description 1
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 1
- 229940015043 glyoxal Drugs 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical class I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 150000002462 imidazolines Chemical class 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- SLCVBVWXLSEKPL-UHFFFAOYSA-N neopentyl glycol Chemical compound OCC(C)(C)CO SLCVBVWXLSEKPL-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- NWVVVBRKAWDGAB-UHFFFAOYSA-N p-methoxyphenol Chemical compound COC1=CC=C(O)C=C1 NWVVVBRKAWDGAB-UHFFFAOYSA-N 0.000 description 1
- LXTZRIBXKVRLOA-UHFFFAOYSA-N padimate a Chemical compound CCCCCOC(=O)C1=CC=C(N(C)C)C=C1 LXTZRIBXKVRLOA-UHFFFAOYSA-N 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 125000005582 pentacene group Chemical group 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- YNPNZTXNASCQKK-UHFFFAOYSA-N phenanthrene Chemical group C1=CC=C2C3=CC=CC=C3C=CC2=C1 YNPNZTXNASCQKK-UHFFFAOYSA-N 0.000 description 1
- 229950000688 phenothiazine Drugs 0.000 description 1
- 150000004714 phosphonium salts Chemical group 0.000 description 1
- 150000003018 phosphorus compounds Chemical group 0.000 description 1
- 238000000016 photochemical curing Methods 0.000 description 1
- 125000001388 picenyl group Chemical group C1(=CC=CC2=CC=C3C4=CC=C5C=CC=CC5=C4C=CC3=C21)* 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 239000005077 polysulfide Chemical class 0.000 description 1
- 229920001021 polysulfide Chemical class 0.000 description 1
- 150000008117 polysulfides Chemical class 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 125000005581 pyrene group Chemical group 0.000 description 1
- 229940079877 pyrogallol Drugs 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 238000010526 radical polymerization reaction Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000007650 screen-printing Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- 125000005579 tetracene group Chemical group 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- YFDSDPIBEUFTMI-UHFFFAOYSA-N tribromoethanol Chemical compound OCC(Br)(Br)Br YFDSDPIBEUFTMI-UHFFFAOYSA-N 0.000 description 1
- DWWMSEANWMWMCB-UHFFFAOYSA-N tribromomethylsulfonylbenzene Chemical compound BrC(Br)(Br)S(=O)(=O)C1=CC=CC=C1 DWWMSEANWMWMCB-UHFFFAOYSA-N 0.000 description 1
- 125000005580 triphenylene group Chemical group 0.000 description 1
- 238000007740 vapor deposition Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 150000007964 xanthones Chemical class 0.000 description 1
- 229910052724 xenon Inorganic materials 0.000 description 1
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D163/00—Coating compositions based on epoxy resins; Coating compositions based on derivatives of epoxy resins
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41J—TYPEWRITERS; SELECTIVE PRINTING MECHANISMS, i.e. MECHANISMS PRINTING OTHERWISE THAN FROM A FORME; CORRECTION OF TYPOGRAPHICAL ERRORS
- B41J2/00—Typewriters or selective printing mechanisms characterised by the printing or marking process for which they are designed
- B41J2/005—Typewriters or selective printing mechanisms characterised by the printing or marking process for which they are designed characterised by bringing liquid or particles selectively into contact with a printing material
- B41J2/01—Ink jet
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41M—PRINTING, DUPLICATING, MARKING, OR COPYING PROCESSES; COLOUR PRINTING
- B41M3/00—Printing processes to produce particular kinds of printed work, e.g. patterns
- B41M3/006—Patterns of chemical products used for a specific purpose, e.g. pesticides, perfumes, adhesive patterns; use of microencapsulated material; Printing on smoking articles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B41—PRINTING; LINING MACHINES; TYPEWRITERS; STAMPS
- B41M—PRINTING, DUPLICATING, MARKING, OR COPYING PROCESSES; COLOUR PRINTING
- B41M5/00—Duplicating or marking methods; Sheet materials for use therein
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/4007—Curing agents not provided for by the groups C08G59/42 - C08G59/66
- C08G59/4014—Nitrogen containing compounds
- C08G59/4021—Ureas; Thioureas; Guanidines; Dicyandiamides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/02—Printing inks
- C09D11/10—Printing inks based on artificial resins
- C09D11/101—Inks specially adapted for printing processes involving curing by wave energy or particle radiation, e.g. with UV-curing following the printing
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D11/00—Inks
- C09D11/30—Inkjet printing inks
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L31/00—Semiconductor devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation and specially adapted either for the conversion of the energy of such radiation into electrical energy or for the control of electrical energy by such radiation; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof
- H01L31/04—Semiconductor devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation and specially adapted either for the conversion of the energy of such radiation into electrical energy or for the control of electrical energy by such radiation; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof adapted as photovoltaic [PV] conversion devices
- H01L31/042—PV modules or arrays of single PV cells
- H01L31/05—Electrical interconnection means between PV cells inside the PV module, e.g. series connection of PV cells
- H01L31/0504—Electrical interconnection means between PV cells inside the PV module, e.g. series connection of PV cells specially adapted for series or parallel connection of solar cells in a module
- H01L31/0516—Electrical interconnection means between PV cells inside the PV module, e.g. series connection of PV cells specially adapted for series or parallel connection of solar cells in a module specially adapted for interconnection of back-contact solar cells
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K3/00—Apparatus or processes for manufacturing printed circuits
- H05K3/10—Apparatus or processes for manufacturing printed circuits in which conductive material is applied to the insulating support in such a manner as to form the desired conductive pattern
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K3/00—Apparatus or processes for manufacturing printed circuits
- H05K3/22—Secondary treatment of printed circuits
- H05K3/28—Applying non-metallic protective coatings
- H05K3/285—Permanent coating compositions
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05K—PRINTED CIRCUITS; CASINGS OR CONSTRUCTIONAL DETAILS OF ELECTRIC APPARATUS; MANUFACTURE OF ASSEMBLAGES OF ELECTRICAL COMPONENTS
- H05K2203/00—Indexing scheme relating to apparatus or processes for manufacturing printed circuits covered by H05K3/00
- H05K2203/01—Tools for processing; Objects used during processing
- H05K2203/0104—Tools for processing; Objects used during processing for patterning or coating
- H05K2203/013—Inkjet printing, e.g. for printing insulating material or resist
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
Definitions
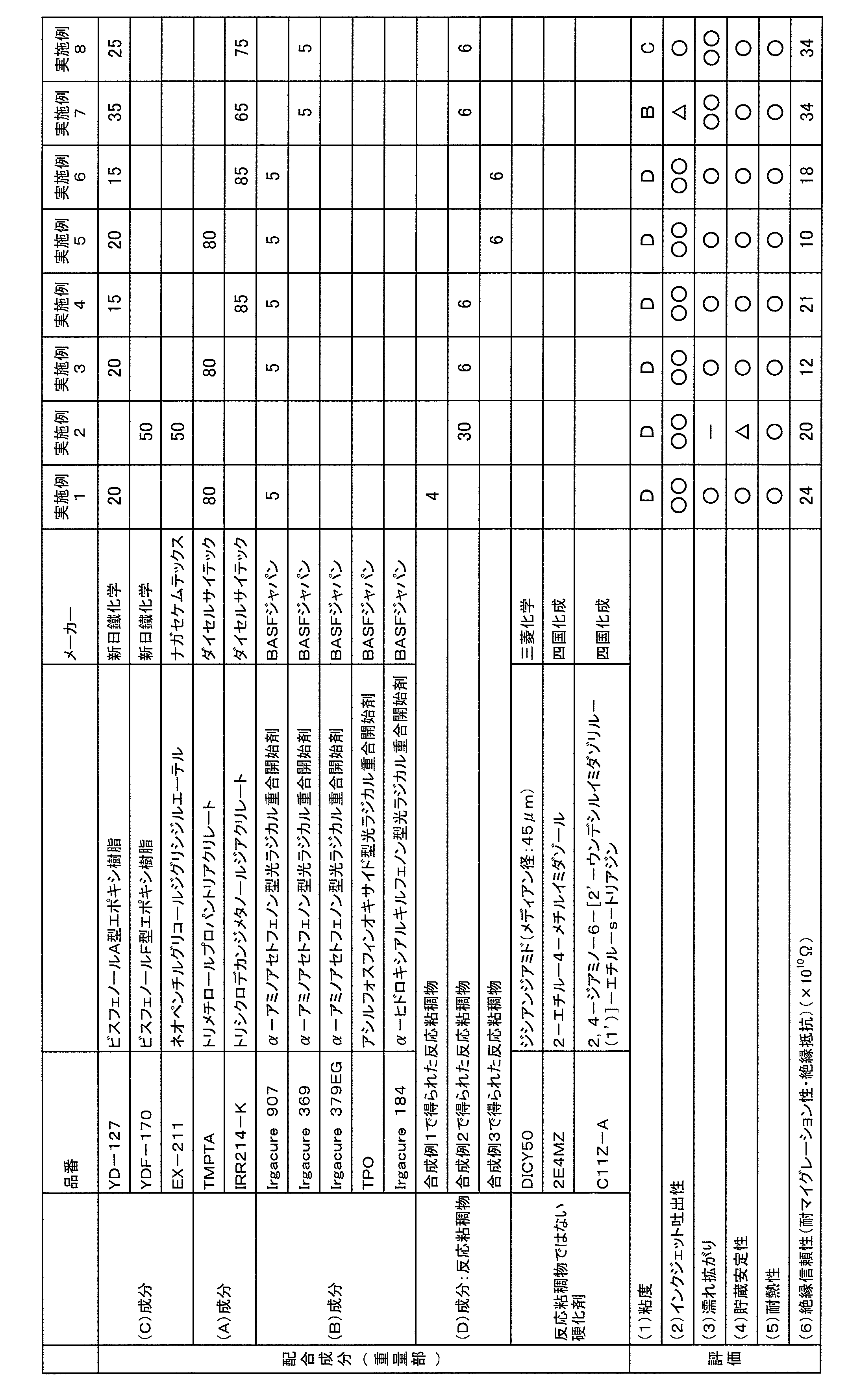
- the present invention is an ink-jet curable composition that is applied by an ink-jet method and is suitably used for forming a cured product layer such as a resist pattern on a substrate, and a curing formed by the ink-jet curable composition.
- the present invention relates to a method for manufacturing an electronic component having a physical layer.
- solder resist pattern which is a patterned solder resist film
- substrate on which wiring is provided on the upper surface
- solder resist pattern which is a patterned solder resist film
- solder resist pattern As a method for forming a fine solder resist pattern, a method of applying a solder resist composition by an ink jet method has been proposed.
- the number of steps is smaller than in the case of forming a solder resist pattern by a screen printing method. For this reason, the solder resist pattern can be easily and efficiently formed by the inkjet method.
- Patent Document 1 discloses inkjet curing comprising a monomer having a (meth) acryloyl group and a thermosetting functional group, a photoreactive diluent having a weight average molecular weight of 700 or less, and a photopolymerization initiator.
- a sex composition is disclosed.
- the viscosity of the curable composition for inkjet at 25 ° C. is 150 mPa ⁇ s or less.
- the viscosity of the curable composition for inkjet described in Patent Document 1 is relatively low. For this reason, the curable composition for inkjet described in Patent Document 1 can be coated on a substrate by an inkjet method.
- the curable composition for inkjet described in Patent Document 1 contains a monomer having a (meth) acryloyl group and a thermosetting functional group, there is a problem that the pot life under an environment of 50 ° C. or higher is short. is there.
- the inkjet curable composition when discharged by an inkjet apparatus, the inkjet curable composition generally remains in the inkjet apparatus for a certain period of time after being supplied into the inkjet apparatus.
- the temperature in the ink jet apparatus may be heated to 50 ° C. or higher in order to improve the ejection property.
- the curable composition for inkjet described in Patent Document 1 the composition is cured in an inkjet apparatus heated to 50 ° C. or more, and the viscosity of the composition is increased. It can be difficult.
- the conventional inkjet curable composition also has a problem that the cured product after curing has low heat resistance and insulation reliability.
- An object of the present invention is a curable composition that is applied by an ink jet method, and includes a compound having a cyclic ether group, and is potted even in an environment in an ink jet apparatus that is heated to 50 ° C. or higher.
- An object is to provide a curable composition for inkjet having a long life and excellent insulation reliability by a cured product after curing, and a method for producing an electronic component using the curable composition for inkjet.
- Another object of the present invention is to provide a curable composition for inkjet which is excellent in heat resistance of a cured product after curing, and a method for producing an electronic component using the curable composition for inkjet.
- a curable composition for inkjet that is applied by an inkjet method and is curable by application of heat, including a compound having a cyclic ether group, and a curing agent
- the curing agent is a reaction viscous product obtained by reacting dicyandiamide and a functional group-containing compound having a functional group capable of reacting with dicyandiamide.
- the functional group-containing compound that reacts with the dicyandiamide preferably has at least one functional group selected from the group consisting of a hydroxyl group, a cyclic ether group, a carboxyl group, and an isocyanate group.
- the functional group-containing compound to be reacted with the dicyandiamide is preferably a compound having an epoxy group.
- the compound having the epoxy group to be reacted with the dicyandiamide preferably has one epoxy group.
- the functional group-containing compound to be reacted with the dicyandiamide preferably has an aromatic skeleton.
- the reaction viscous product is preferably a reaction viscous product obtained by reacting the functional group-containing compound with 1 mol or more and 3 mol or less with respect to 1 mol of dicyandiamide.
- the dicyandiamide reacted with the functional group-containing compound is in a powder form.
- the reaction viscous material is dissolved in the curable composition.
- the curable composition for inkjet that is applied by an inkjet method and is curable by irradiation with light and application of heat, A polyfunctional compound having two or more (meth) acryloyl groups and a photopolymerization initiator are further included.
- the photopolymerization initiator is preferably an ⁇ -aminoalkylphenone type photoradical polymerization initiator.
- the photopolymerization initiator is more preferably an ⁇ -aminoalkylphenone type photoradical polymerization initiator having a dimethylamino group.
- the polyfunctional compound having two or more (meth) acryloyl groups has a polycyclic skeleton and two (meth) acryloyl groups. It is a polyfunctional compound having the above.
- a monofunctional compound having a polycyclic skeleton and having one (meth) acryloyl group is further included.
- the polyfunctional compound having two or more (meth) acryloyl groups has a polycyclic skeleton, and 2 (meth) acryloyl groups.
- a polyfunctional compound having at least one polyfunctional compound further having a polycyclic skeleton and having one (meth) acryloyl group is further included.
- the content of the polyfunctional compound is 20 in a total of 100% by weight of the polyfunctional compound, the monofunctional compound, and the photopolymerization initiator.
- the content of the monofunctional compound is 5% by weight or more and 50% by weight or less.
- the monofunctional compound comprises isobornyl (meth) acrylate, dihydroxycyclopentadienyl (meth) acrylate, dicyclopentenyl (meth) acrylate, dicyclopentenyloxyethyl (meth) acrylate, and dicyclopentanyl (meth) acrylate. It is preferably at least one selected from the group.
- the polyfunctional compound is preferably tricyclodecane dimethanol di (meth) acrylate.
- the reaction viscous product is preferably a reaction product obtained by reacting the functional group of the functional group-containing compound with a part of the active hydrogen of dicyandiamide.
- the viscosity at 25 ° C. measured in accordance with JIS K2283 is preferably 160 mPa ⁇ s or more and 1200 mPa ⁇ s or less.
- coating the inkjet curable composition comprised according to this invention by the inkjet system, and drawing it in a pattern form, and the said inkjet use drawn in the pattern form A method for producing an electronic component is provided, comprising: applying heat to a curable composition and curing the composition to form a cured product layer.
- a method for manufacturing a printed wiring board which is an electronic component having a resist pattern
- the inkjet curable composition is applied by an inkjet method. Then, a resist pattern is formed by drawing in a pattern and applying heat to the curable composition for inkjet drawn in the pattern to be cured.
- an inkjet printer comprising a polyfunctional compound having two or more (meth) acryloyl groups and a photopolymerization initiator.
- light and heat are applied to the curable composition for inkjet drawn in a pattern and cured to form a cured product layer.
- the curable composition for inkjet according to the present invention includes a compound having a cyclic ether group and a curing agent, and the curing agent further reacts dicyandiamide and a functional group-containing compound having a functional group capable of reacting with the dicyandiamide. Since it is a reaction viscous product, it can be applied by an ink jet method.
- the curable composition for inkjet according to the present invention has the above composition, and in particular, because the curing agent is the specific reaction viscous product, the environment within the inkjet apparatus heated to 50 ° C. or higher. But the pot life is long enough. Moreover, since the compound which has a cyclic ether group is contained, the heat resistance of hardened
- the curable composition for inkjet according to the present invention includes a compound (C) having a cyclic ether group and a curing agent (D). It is preferable that the curable composition for inkjet according to the present invention further includes a polyfunctional compound (A) having two or more (meth) acryloyl groups and a photopolymerization initiator (B).
- the curing agent (D) is a reaction viscous product obtained by reacting dicyandiamide with a functional group-containing compound having a functional group capable of reacting with the dicyandiamide.
- (meth) acryloyl group refers to an acryloyl group and a methacryloyl group.
- the curable composition for inkjet according to the present invention contains the compound (C) having a cyclic ether group and the curing agent (D), it can be cured by application of heat. Therefore, the curable composition for inkjet according to the present invention is a thermosetting composition for inkjet.
- the curable composition for inkjet according to the present invention contains the polyfunctional compound (A) and the photopolymerization initiator (B), the curable composition for inkjet according to the present invention is irradiated with light and heat.
- the composition is curable by application of an ink and is a light and thermosetting composition for inkjet.
- the cured curable composition for inkjet according to the present invention can be cured by applying heat to obtain a cured product layer such as a resist pattern which is a cured product.
- a cured product layer such as a resist pattern which is a cured product.
- the curable composition for inkjet according to the present invention contains a polyfunctional compound (A) and a photopolymerization initiator (B)
- a primary cured product is obtained after obtaining a primary cured product by light irradiation.
- a cured product layer such as a resist pattern, which is a cured product, by applying heat to the film.
- the curable composition for inkjet according to the present invention contains the compound (C) having a cyclic ether group and the curing agent (D), and further the curing agent (D) is the reaction viscous product, the cyclic ether.
- the compound (C) having a group and the curing agent (D) are contained, the pot life can be sufficiently lengthened even in an environment in an ink jet apparatus heated to 50 ° C. or higher.
- the curable composition for inkjet before the coating by the inkjet method is heated to 50 ° C. or higher, the viscosity does not easily increase and the thermal curing does not easily proceed.
- the curable composition for inkjet is excellent in stability at high temperatures, and can be stably discharged from the inkjet nozzle. For this reason, a cured product layer such as a uniform resist pattern can be formed.
- the curable composition for inkjet according to the present invention contains the compound (C) having a cyclic ether group in particular, the heat resistance of the cured product after curing can be improved.
- the insulation reliability due to the cured product after curing can be improved.
- the curable composition for inkjet according to the present invention has the above composition, and in particular, the curing agent (D) is the above reaction viscous product, whereby the insulation reliability due to the cured product after curing is increased.
- the inventors have found. For example, when the reaction viscous material using dicyandiamide is used, the insulation reliability by the cured material is considerably higher than when the imidazole curing agent or the like is used.
- the curable composition for inkjet according to the present invention preferably contains a polyfunctional compound (A) having two or more (meth) acryloyl groups.
- the polyfunctional compound (A) is not particularly limited as long as it has two or more (meth) acryloyl groups.
- As the polyfunctional compound (A) a conventionally known polyfunctional compound having two or more (meth) acryloyl groups can be used. Since the polyfunctional compound (A) has two or more (meth) acryloyl groups, polymerization proceeds and cures upon irradiation with light.
- a polyfunctional compound (A) only 1 type may be used and 2 or more types may be used together.
- (meth) acrylic acid adducts of polyhydric alcohols examples include acrylates.
- examples of the polyhydric alcohol include diethylene glycol, triethylene glycol, polyethylene glycol, dipropylene glycol, tripropylene glycol, polypropylene glycol, trimethylol propane, cyclohexane dimethanol, tricyclodecane dimethanol, an alkylene oxide adduct of bisphenol A, and Examples include pentaerythritol.
- the term “(meth) acrylate” refers to acrylate and methacrylate.
- the term “(meth) acryl” refers to acrylic and methacrylic.
- the polyfunctional compound (A) is preferably a polyfunctional compound (A1) having a polycyclic skeleton and having two or more (meth) acryloyl groups.
- the heat and moisture resistance of the cured product of the curable composition for inkjet can be increased. Therefore, the printed wiring board using the curable composition for inkjet according to the present invention can be used for a long period of time, and the reliability of the printed wiring board can be enhanced.
- the polyfunctional compound (A1) is not particularly limited as long as it has a polycyclic skeleton and two or more (meth) acryloyl groups.
- the polyfunctional compound (A1) a conventionally known polyfunctional compound having a polycyclic skeleton and having two or more (meth) acryloyl groups can be used. Since the polyfunctional compound (A1) has two or more (meth) acryloyl groups, the polymerization proceeds and is cured by light irradiation.
- a polyfunctional compound (A1) only 1 type may be used and 2 or more types may be used together.
- Polyfunctional compounds (A1) include polyhydric alcohol (meth) acrylic acid adducts, polyhydric alcohol alkylene oxide-modified (meth) acrylic acid adducts, urethane (meth) acrylates, and polyester (meth). Examples include acrylates.
- Examples of the polyhydric alcohol include diethylene glycol, triethylene glycol, polyethylene glycol, dipropylene glycol, tripropylene glycol, polypropylene glycol, trimethylolpropane, and pentaerythritol.
- polyfunctional compound (A1) examples include tricyclodecane dimethanol di (meth) acrylate, isobornyl dimethanol di (meth) acrylate and dicyclopentenyl dimethanol di (meth) acrylate.
- a polyfunctional compound (A1) is tricyclodecane dimethanol di (meth) acrylate from a viewpoint of improving the heat-and-moisture resistance of hardened
- the term “(meth) acrylate” refers to acrylate and methacrylate.
- the “polycyclic skeleton” indicates a structure having a plurality of cyclic skeletons continuously.
- Examples of the polycyclic skeleton in the polyfunctional compound (A1) and the monofunctional compound (E) include a polycyclic alicyclic skeleton and a polycyclic aromatic skeleton.
- polycyclic alicyclic skeleton examples include a bicycloalkane skeleton, a tricycloalkane skeleton, a tetracycloalkane skeleton, and an isobornyl skeleton.
- polycyclic aromatic skeleton examples include naphthalene ring skeleton, anthracene ring skeleton, phenanthrene ring skeleton, tetracene ring skeleton, chrysene ring skeleton, triphenylene ring skeleton, tetraphen ring skeleton, pyrene ring skeleton, pentacene ring skeleton, picene ring skeleton and And perylene ring skeleton.
- the compounding amount of the polyfunctional compound (A) is appropriately adjusted so as to be appropriately cured by light irradiation, and is not particularly limited.
- the content of the polyfunctional compound (A) is 0% by weight or more, preferably 20% by weight or more, in 100% by weight of the curable composition for inkjet.
- it is 40 weight% or more, More preferably, it is 60 weight% or more, Preferably it is 95 weight% or less, More preferably, it is 90 weight% or less.
- the upper limit of the content of the polyfunctional compound (A) is appropriately adjusted depending on the contents of the components (B) to (D) and other components.
- the polyfunctional compound (A1) and the monofunctional compound (E) to be described later are used in combination, the polyfunctional compound (A1), the monofunctional compound (E), and the photopolymerization initiator (B) in a total of 100% by weight
- the content of the polyfunctional compound (A1) is preferably 20% by weight or more and 70% by weight or less.
- the content of the polyfunctional compound (A1) is more preferably 40% by weight or more, and more preferably 60% by weight or less.
- the curable composition can be more effectively cured by light irradiation.
- the content of the polyfunctional compound (A1) is not more than the above upper limit, the moisture and heat resistance of the cured product is further increased.
- the curable composition for inkjet according to the present invention preferably contains a photopolymerization initiator (B) together with the polyfunctional compound (A).
- the photopolymerization initiator (B) include a photoradical polymerization initiator and a photocationic polymerization initiator.
- the photopolymerization initiator (B) is preferably a radical photopolymerization initiator.
- a photoinitiator (B) only 1 type may be used and 2 or more types may be used together.
- the photo radical polymerization initiator is not particularly limited.
- the photo radical polymerization initiator is a compound for generating radicals upon light irradiation and initiating a radical polymerization reaction.
- Specific examples of the photo radical polymerization initiator include, for example, benzoin, benzoin alkyl ethers, acetophenones, aminoacetophenones, anthraquinones, thioxanthones, ketals, 2,4,5-triarylimidazole dimer, Examples include riboflavin tetrabutyrate, thiol compounds, 2,4,6-tris-s-triazine, organic halogen compounds, benzophenones, xanthones, and 2,4,6-trimethylbenzoyldiphenylphosphine oxide.
- the said radical photopolymerization initiator only 1 type may be used and 2 or more types may be used together.
- benzoin alkyl ethers examples include benzoin methyl ether, benzoin ethyl ether, and benzoin isopropyl ether.
- acetophenones examples include acetophenone, 2,2-dimethoxy-2-phenylacetophenone, 2,2-diethoxy-2-phenylacetophenone, 1,1-dichloroacetophenone, and the like.
- aminoacetophenones examples include 2-methyl-1- [4- (methylthio) phenyl] -2-morpholinopropan-1-one, 2-benzyl-2-dimethylamino-1- (4-morpholinophenyl) -butane
- examples include -1-one and N, N-dimethylaminoacetophene.
- examples of the anthraquinones include 2-methylanthraquinone, 2-ethylanthraquinone, 2-t-butylanthraquinone and 1-chloroanthraquinone.
- Examples of the thioxanthones include 2,4-dimethylthioxanthone, 2,4-diethylthioxanthone, 2-chlorothioxanthone, and 2,4-diisopropylthioxanthone.
- Examples of the ketals include acetophenone dimethyl ketal and benzyl dimethyl ketal.
- Examples of the thiol compound include 2-mercaptobenzoimidazole, 2-mercaptobenzoxazole and 2-mercaptobenzothiazole.
- Examples of the organic halogen compound include 2,2,2-tribromoethanol and tribromomethylphenyl sulfone.
- Examples of the benzophenones include benzophenone and 4,4'-bisdiethylaminobenzophenone.
- the photo radical polymerization initiator is preferably an ⁇ -aminoalkylphenone type photo radical polymerization initiator, and more preferably an ⁇ -aminoalkyl phenone type photo radical polymerization initiator having a dimethylamino group.
- the thermosetting speed can be increased, and the thermosetting property of the light irradiated product of the composition can be increased. Can be improved.
- the present inventors can not only improve the photocurability but also improve the thermosetting property by using an ⁇ -aminoalkylphenone type photoradical polymerization initiator having a dimethylamino group. I found.
- the ⁇ -aminoalkylphenone type photoradical polymerization initiator having a dimethylamino group is a component that greatly contributes to improvement of thermosetting.
- the heat resistance and insulation reliability of the cured product can be enhanced. Even if an electronic component such as a printed wiring board having a resist pattern formed by the curable composition for inkjet according to the present invention is used for a long time under a high humidity condition, the insulation resistance is excellent. Is kept high enough.
- ⁇ -aminoalkylphenone type photo radical polymerization initiator examples include IRGACURE907, IRGACURE369, IRGACURE379, and IRGACURE379EG manufactured by BASF.
- Other ⁇ -aminoalkylphenone type photopolymerization initiators may be used.
- 2-benzyl-2-dimethylamino-1- (4-morpholinophenyl) -butanone examples of the ⁇ -aminoalkylphenone type photo radical polymerization initiator.
- a photopolymerization initiation assistant may be used together with the photoradical polymerization initiator.
- the photopolymerization initiation assistant include N, N-dimethylaminobenzoic acid ethyl ester, N, N-dimethylaminobenzoic acid isoamyl ester, pentyl-4-dimethylaminobenzoate, triethylamine, and triethanolamine.
- Photopolymerization initiation assistants other than these may be used.
- the said photoinitiation adjuvant only 1 type may be used and 2 or more types may be used together.
- a titanocene compound such as CGI-784 (manufactured by Ciba Specialty Chemicals) having absorption in the visible light region may be used to promote the photoreaction.
- the photocationic polymerization initiator is not particularly limited, and examples thereof include sulfonium salts, iodonium salts, metallocene compounds, and benzoin tosylate. As for the said photocationic polymerization initiator, only 1 type may be used and 2 or more types may be used together.
- the content of the photopolymerization initiator (B) with respect to 100 parts by weight of the polyfunctional compound (A) is preferably 0.1 parts by weight or more, more preferably 1 part by weight or more, and further preferably 3 parts by weight or more.
- the amount is preferably 30 parts by weight or less, more preferably 15 parts by weight or less, and still more preferably 10 parts by weight or less.
- the curable composition for inkjet according to the present invention contains a compound (C) having a cyclic ether group so that it can be cured by application of heat.
- the curable composition or the primary cured product of the curable composition can be further cured by applying heat.
- a resist pattern can be formed efficiently and accurately, and the heat resistance and insulation reliability of the cured product can be further increased. Only 1 type may be used for the compound (C) which has a cyclic ether group, and 2 or more types may be used together.
- the compound (C) having a cyclic ether group is not particularly limited as long as it has a cyclic ether group.
- the cyclic ether group in the compound (C) include an epoxy group and an oxetanyl group.
- the cyclic ether group is preferably an epoxy group from the viewpoint of enhancing curability and obtaining a cured product that is more excellent in heat resistance and insulation reliability.
- the compound (C) having a cyclic ether group preferably has two or more cyclic ether groups.
- compounds having an epoxy group include heterocyclic epoxy compounds such as bisphenol S type epoxy compounds, diglycidyl phthalate compounds, triglycidyl isocyanurates, bixylenol type epoxy compounds, biphenol type epoxy compounds, tetraglycidyl xylenoyl Ethane compound, bisphenol A type epoxy compound, hydrogenated bisphenol A type epoxy compound, bisphenol F type epoxy compound, brominated bisphenol A type epoxy compound, phenol novolac type epoxy compound, cresol novolac type epoxy compound, alicyclic epoxy compound, bisphenol A novolac type epoxy compound, chelate type epoxy compound, glyoxal type epoxy compound, amino group-containing epoxy compound, rubber-modified epoxy compound , Dicyclopentadiene phenolic type epoxy compounds, silicone-modified epoxy compounds and ⁇ - caprolactone-modified epoxy compounds and the like.
- heterocyclic epoxy compounds such as bisphenol S type epoxy compounds, diglycidyl phthalate compounds, triglycidyl isocyan
- the compound (C) having a cyclic ether group preferably has an aromatic skeleton.
- a compound having an aromatic skeleton and a cyclic ether group By using a compound having an aromatic skeleton and a cyclic ether group, the thermal stability during storage and ejection of the curable composition is further improved, and gelation is less likely to occur during storage of the curable composition.
- a compound having an aromatic skeleton and a cyclic ether group has a polyfunctional compound (A), a monofunctional compound (E) and a curing agent (C) as compared with a compound having no aromatic skeleton and having a cyclic ether group. ), The insulation reliability is further improved.
- Examples of the compound having an oxetanyl group are exemplified in Japanese Patent No. 3074086.
- the compound (C) having a cyclic ether group is preferably liquid at 25 ° C.
- the viscosity of the compound (C) having a cyclic ether group at 25 ° C. preferably exceeds 300 mPa ⁇ s.
- the viscosity at 25 ° C. of the compound (C) having a cyclic ether group is preferably 80 Pa ⁇ s or less.
- the viscosity of the compound (C) having a cyclic ether group is not more than the above upper limit, the dischargeability of the curable composition is further improved, and the compound (C) having a cyclic ether group and other components The compatibility is further increased, and the insulation reliability is further improved.
- the compounding amount of the compound (C) having a cyclic ether group is appropriately adjusted so as to be appropriately cured by application of heat, and is not particularly limited.
- the content of the compound (C) having a cyclic ether group in 100% by weight of the curable composition for inkjet is preferably 3% by weight or more, preferably 99% by weight or less, more preferably 95% by weight or less, and still more preferably 80%. % By weight or less.
- the content of the compound (C) having a cyclic ether group is more preferably 5% by weight or more, and still more preferably 10% by weight or more.
- the content of the compound (C) having a cyclic ether group in 100% by weight of the curable composition for inkjet is particularly preferably 50. % By weight or less, most preferably 40% by weight or less.
- the content of the compound (C) is not less than the above lower limit, the curable composition can be more effectively cured by application of heat.
- the content of the compound (C) is not more than the above upper limit, the heat resistance of the cured product is further increased.
- the curing agent (D) is a reaction viscous product obtained by reacting dicyandiamide with a functional group-containing compound having a functional group capable of reacting with the dicyandiamide. Since such a hardening
- the said reaction viscous material should just have a viscous property by the reaction viscous material alone before using it for the curable composition for inkjet, and it is not viscous in the curable composition for inkjet. Also good. Moreover, when the said reaction viscous material is taken out from the curable composition for inkjets, this reaction viscous material may be viscous.
- the curable composition for inkjet according to the present invention uses a curing agent (D) that is a reaction viscous product obtained by reacting dicyandiamide with a functional group-containing compound having a functional group capable of reacting with the dicyandiamide. If this hardening
- the reaction viscous product (E1) before blended in the curable composition for inkjet is not blended in an organic solvent or blended in an organic solvent and the reaction viscous product (E1). )
- the amount of the organic solvent blended with respect to 100 parts by weight is preferably 100 parts by weight or less.
- blended with respect to 100 weight part of the said reaction viscous material (E1) shall be 50 weight part or less. Is preferably 20 parts by weight or less, more preferably 10 parts by weight or less, and particularly preferably 1 part by weight or less.
- the reaction viscous product is preferably a reaction product obtained by reacting the functional group-containing compound with a part of the active hydrogen of dicyandiamide.
- the functional group capable of reacting with the dicyandiamide of the functional group-containing compound generally reacts with a part of the active hydrogen of dicyandiamide.
- the dicyandiamide to be reacted with the functional group-containing compound is preferably in a powder form.
- the powdery dicyandiamide is not powdered, and the viscous viscous reaction product is obtained.
- the functional group-containing compound to be reacted with the dicyandiamide includes a hydroxyl group, a cyclic ether group, a carboxyl group, and an isocyanate group. It preferably has at least one functional group selected from the group consisting of:
- the functional group-containing compound reacted with the dicyandiamide is preferably a compound having a cyclic ether group.
- the compound having the cyclic ether group to be reacted with dicyandiamide is preferably a compound having one cyclic ether group.
- the functional group-containing compound reacted with the dicyandiamide is preferably a compound having an epoxy group.
- the compound having an epoxy group to be reacted with dicyandiamide is preferably a compound having one epoxy group.
- the dicyandiamide and The functional group-containing compound to be reacted preferably has an aromatic skeleton, more preferably a compound having an aromatic skeleton and a cyclic ether group, and a compound having an aromatic skeleton and an epoxy group. Particularly preferred.
- the functional group-containing compound examples include phenyl glycidyl ether, butyl glycidyl ether, ortho-cresyl glycidyl ether, meta-cresyl glycidyl ether, para-cresyl glycidyl ether, allyl glycidyl ether, para-t-butylphenyl glycidyl ether, etc.
- Examples thereof include glycidyl ethers, glycidyl (meth) acrylate, and 3,4-epoxycyclohexylmethyl (meth) acrylate.
- the functional group-containing compound is an aromatic ring-containing phenyl glycidyl ether, ortho-cresyl glycidyl ether, meta-cresyl glycidyl ether, para-cresyl glycidyl ether, or para-t-butyl. Preference is given to phenylglycidyl ether.
- the functional group-containing compound is preferably 0.2 mol or more, more preferably 1 mol or more, preferably 4 mol or less, more preferably 3 mol per mol of dicyandiamide. It is desirable to make it react below mol. That is, the reaction viscous product is preferably 0.2 mol or more, more preferably 1 mol or more, preferably 4 mol or less, more preferably 3 mol or less of the functional group-containing compound with respect to 1 mol of the dicyandiamide. It is desirable that the reaction is a viscous reaction product.
- the reaction viscous product is a reaction viscosity obtained by reacting 1 mol to 3 mol of the functional group-containing compound with respect to 1 mol of the dicyandiamide. It is particularly desirable to be a product. There exists a possibility that unreacted dicyandiamide may precipitate that the usage-amount of the said functional group containing compound is less than the said minimum. When the usage-amount of the said functional group containing compound exceeds the said upper limit, there exists a possibility that all the active hydrogens of the said reaction viscous substance may deactivate, and it becomes impossible to harden a compound (C). In addition, in this reaction, it is preferable to make it react at 60 to 140 degreeC in presence of a solvent or a reaction accelerator as needed.
- a solvent may be used to dissolve the dicyandiamide.
- the solvent may be any solvent that can dissolve dicyandiamide.
- Usable solvents include acetone, methyl ethyl ketone, dimethylformamide, methyl cellosolve and the like.
- reaction accelerator In order to promote the reaction between the dicyandiamide and the functional group-containing compound, a reaction accelerator may be used.
- Known reaction accelerators such as phenols, amines, imidazoles, and triphenylphosphine can be used as the reaction accelerator.
- the reaction viscous material is compatible with the compound (C) having a cyclic ether group.
- the compound (A) having two or more (meth) acryloyl groups preferably compatible with the monofunctional compound (E), and further curable composition It is preferably dissolved in the product.
- the reaction viscous product is preferably compatible with the compound (C) having a cyclic ether group, preferably compatible with the compound (A) having two or more (meth) acryloyl groups, It is preferable that it is compatible with the monofunctional compound (E).
- the above-mentioned reaction viscous material is, for example, a non-powder-like reaction viscous material obtained by a reaction between powdered dicyandiamide and the above functional group-containing compound.
- the reaction viscous material is preferably not a solid, preferably not a crystal, and preferably not a crystalline solid.
- the reaction viscous material is preferably in a liquid or semi-solid form.
- the reaction viscous product is preferably transparent or translucent. Whether or not the reaction viscous material is transparent or translucent can be determined by whether or not the object is visible when the object is viewed through the reaction viscous material having a thickness of 5 mm.
- the compounding ratio of the compound (C) having a cyclic ether group and the reaction viscous material is not particularly limited.
- the blending amount of the reaction viscous material is appropriately adjusted so as to be appropriately cured by application of heat, and is not particularly limited.
- the content of the reaction viscous product is preferably 5 parts by weight or more, more preferably 10 parts by weight or more, preferably 60 parts by weight or less, more preferably 100 parts by weight of the compound (C) having a cyclic ether group. 50 parts by weight or less.
- the curable composition for inkjet according to the present invention preferably contains a monofunctional compound (E) having a polycyclic skeleton and having one (meth) acryloyl group. Furthermore, the curable composition for inkjet according to the present invention includes a polyfunctional compound (A1) having a polycyclic skeleton and having two or more (meth) acryloyl groups, a polycyclic skeleton, and (meta And a monofunctional compound (E) having one acryloyl group. In these cases, the heat-and-moisture resistance of the cured product of the ink-jet curable composition is considerably increased.
- an electronic component such as a printed wiring board using the curable composition for inkjet according to the present invention can be used for a longer period of time, and the reliability of the electronic component is further enhanced.
- the use of the monofunctional compound (E) not only increases the heat and moisture resistance of the cured product, but also increases the dischargeability of the curable composition.
- the monofunctional compound (E) having a polycyclic skeleton and having one (meth) acryloyl group it does not have a polycyclic skeleton and has one (meth) acryloyl group. Compared with the case where a monofunctional compound is used, the heat-and-moisture resistance of the cured product is increased.
- the monofunctional compound (E) is not particularly limited as long as it has a polycyclic skeleton and one (meth) acryloyl group.
- the monofunctional compound (E) a conventionally known monofunctional compound having a polycyclic skeleton and one (meth) acryloyl group can be used.
- monofunctional compound (E) only 1 type may be used and 2 or more types may be used together.
- the monofunctional compound (E) examples include isobornyl (meth) acrylate, dihydroxycyclopentadienyl (meth) acrylate, dicyclopentenyl (meth) acrylate, dicyclopentenyloxyethyl (meth) acrylate, and dicyclopenta.
- examples include nyl (meth) acrylate and naphthyl (meth) acrylate.
- the monofunctional compound (E) is composed of isobornyl (meth) acrylate, dihydroxycyclopentadienyl (meth) acrylate, dicyclopentenyl (meth) acrylate, dicyclohexane. It is preferably at least one selected from the group consisting of pentenyloxyethyl (meth) acrylate and dicyclopentanyl (meth) acrylate.
- the content of the monofunctional compound (E) is preferably 5% by weight or more, more preferably 15% by weight or more, preferably 50% by weight or less, more preferably 45% by weight or less. It is.
- the content of the monofunctional compound (E) is 5% by weight or more and 50% by weight or less in the total of 100% by weight of the polyfunctional compound (A1), the monofunctional compound (E), and the photopolymerization initiator (B). It is preferable. In the total of 100% by weight of the polyfunctional compound (A1), the monofunctional compound (E), and the photopolymerization initiator (B), the content of the monofunctional compound (E) is more preferably 15% by weight or more. Is 45% by weight or less. When the content of the monofunctional compound (E) is not less than the above lower limit, the moisture and heat resistance of the cured product is further enhanced. When the content of the monofunctional compound (E) is not more than the above upper limit, the curable composition can be more effectively cured by light irradiation.
- the upper limit of the total content of the polyfunctional compound (A1) and the monofunctional compound (E) is appropriately adjusted depending on the content of the photopolymerization initiator (B).
- the curable composition for inkjet according to the present invention may contain a thermosetting agent other than the reaction viscous material. Furthermore, the curable composition for inkjet according to the present invention may contain a curing accelerator.
- thermosetting agent examples include organic acids, amine compounds, amide compounds, hydrazide compounds, imidazole compounds, imidazoline compounds, phenol compounds, urea compounds, polysulfide compounds, and acid anhydrides.
- a modified polyamine compound such as an amine-epoxy adduct may be used as the thermosetting agent.
- curing accelerator examples include tertiary amines, imidazoles, quaternary ammonium salts, quaternary phosphonium salts, organometallic salts, phosphorus compounds, urea compounds, and the like.
- the curable composition for inkjet according to the present invention various additives may be blended as long as the object of the present invention is not impaired.
- the additive is not particularly limited, and examples thereof include a colorant, a polymerization inhibitor, an antifoaming agent, a leveling agent, and an adhesion imparting agent.
- the curable composition for inkjet according to the present invention may contain an organic solvent as long as the amount is small.
- Examples of the colorant include phthalocyanine / blue, phthalocyanine / green, iodin / green, disazo yellow, crystal violet, titanium oxide, carbon black and naphthalene black.
- Examples of the polymerization inhibitor include hydroquinone, hydroquinone monomethyl ether, tert-butylcatechol, pyrogallol and phenothiazine.
- Examples of the antifoaming agent include silicone-based antifoaming agents, fluorine-based antifoaming agents, and polymer-based antifoaming agents.
- Examples of the leveling agent include silicone leveling agents, fluorine leveling agents, and polymer leveling agents.
- Examples of the adhesion imparting agent include imidazole adhesion imparting agents, thiazole adhesion imparting agents, triazole adhesion imparting agents, and silane coupling agents.
- the viscosity at 25 ° C. measured in accordance with JIS K2283 is preferably 160 mPa ⁇ s or more and 1200 mPa ⁇ s or less.
- the viscosity of the curable composition for inkjet is not less than the above lower limit and not more than the above upper limit, the curable composition for inkjet can be easily and accurately discharged from the inkjet head.
- the curable composition for inkjet is heated to 50 ° C. or higher, the composition can be easily and accurately discharged from the inkjet head.
- the viscosity is preferably 1000 mPa ⁇ s or less, more preferably 500 mPa ⁇ s or less.
- the viscosity preferably exceeds 500 mPa ⁇ s.
- the curable composition for inkjet according to the present invention does not contain an organic solvent, or contains an organic solvent, and the content of the organic solvent in 100% by weight of the curable composition is 50% by weight or less. preferable.
- the content of the organic solvent is more preferably 20% by weight or less, still more preferably 10% by weight or less, and particularly preferably 1% by weight or less. The smaller the content of the organic solvent, the better the resolution when forming the cured product layer.
- the curable composition for inkjet according to the present invention does not contain an organic solvent or contains an organic solvent, and the content of the organic solvent is 50 parts by weight or less with respect to 100 parts by weight of the reaction viscous material (E1). It is preferable that The content of the organic solvent is more preferably 20 parts by weight or less, still more preferably 10 parts by weight or less, and particularly preferably 1 part by weight or less with respect to 100 parts by weight of the reaction viscous product (E1). The smaller the content of the organic solvent, the better the resolution when forming the cured product layer.
- the method for producing an electronic component according to the present invention is characterized by using the above-described curable composition for inkjet. That is, in the method for manufacturing an electronic component according to the present invention, first, the curable composition for inkjet is applied by an inkjet method to draw a pattern. At this time, it is particularly preferable to directly draw the curable composition for inkjet. “Direct drawing” means drawing without using a mask.
- the electronic component include a printed wiring board and a touch panel component.
- the electronic component is preferably a wiring board, and more preferably a printed wiring board.
- An inkjet printer is used for coating the curable composition for inkjet.
- the ink jet printer has an ink jet head.
- the inkjet head has a nozzle.
- the ink jet device preferably includes a heating unit for heating the temperature in the ink jet device or the ink jet head to 50 ° C. or higher. It is preferable that the said inkjet curable composition is coated on the coating object member.
- a substrate etc. are mentioned as said coating object member. Examples of the substrate include a substrate having wirings provided on the upper surface.
- the inkjet curable composition is preferably applied onto a printed circuit board.
- a substrate to a member mainly composed of glass and to produce a glass substrate for a display device such as a liquid crystal display device.
- a conductive pattern such as ITO is provided on glass by a method such as vapor deposition, and a cured product layer is formed on the conductive pattern by an inkjet method by the method for manufacturing an electronic component according to the present invention.
- a pattern is provided on the cured product layer with a conductive ink or the like, the cured product layer becomes an insulating film, and electrical connection is obtained between predetermined patterns in the conductive pattern on the glass.
- the curable composition for inkjet drawn in a pattern and cured is used as a curable composition for inkjets.
- the curable composition for inkjets drawn by pattern shape is used.
- the material is cured by applying light and heat to form a cured product layer.
- an electronic component having a cured product layer can be obtained.
- the cured product layer may be an insulating film or a resist pattern.
- the insulating film may be a patterned insulating film.
- the cured product layer is preferably a resist pattern.
- the resist pattern is preferably a solder resist pattern.
- the method for manufacturing an electronic component according to the present invention is preferably a method for manufacturing a printed wiring board having a resist pattern.
- the inkjet curable composition is applied by an inkjet method, drawn in a pattern, and the inkjet curable composition drawn in a pattern is irradiated with light and heat, cured. It is preferable to form a resist pattern.
- the ink-jet curable composition drawn in a pattern may be primarily cured by irradiation with light to obtain a primary cured product. Thereby, wetting and spreading of the drawn curable composition for inkjet can be suppressed, and a highly accurate resist pattern can be formed.
- the primary cured product when a primary cured product is obtained by light irradiation, the primary cured product may be subjected to main curing by applying heat to obtain a cured product, and a cured product layer such as a resist pattern may be formed.
- the curable composition for inkjet according to the present invention can be cured by light irradiation and heat application.
- the heating temperature for curing by applying heat is preferably 100 ° C. or higher, more preferably 120 ° C. or higher, preferably 250 ° C. or lower, more preferably 200 ° C. or lower.
- the light irradiation may be performed after drawing or may be performed simultaneously with drawing.
- light may be irradiated at the same time as or after the ejection of the curable composition.
- the light source may be arranged so that the light irradiation portion is positioned at the drawing position by the inkjet head.
- the light source for irradiating light is appropriately selected according to the irradiating light.
- the light source include a UV-LED, a low-pressure mercury lamp, a medium-pressure mercury lamp, a high-pressure mercury lamp, an ultrahigh-pressure mercury lamp, a xenon lamp, and a metal halide lamp.
- the irradiated light is generally ultraviolet rays, and may be an electron beam, ⁇ -ray, ⁇ -ray, ⁇ -ray, X-ray, neutron beam, or the like.
- the temperature at the time of application of the inkjet curable composition is not particularly limited as long as the inkjet curable composition has a viscosity at which the inkjet curable composition can be discharged from the inkjet head.
- the temperature at the time of application of the curable composition for inkjet is preferably 50 ° C. or higher, more preferably 60 ° C. or higher, and preferably 100 ° C. or lower.
- the viscosity of the curable composition for inkjet at the time of coating is not particularly limited as long as it can be discharged from the inkjet head.
- the curable composition for inkjet according to the present invention includes a specific reaction viscous material as the curing agent (D), for example, even when the inkjet curable composition is heated in an inkjet head, the inkjet curable composition is used.
- the pot life of the curable composition is sufficiently long and stable ejection is possible.
- the ink-jet curable composition can be heated to a viscosity suitable for coating by the ink-jet method, the use of the ink-jet curable composition according to the present invention suitably produces electronic components such as printed wiring boards. can do.
- Irgacure 907 (manufactured by BASF Japan): ⁇ -aminoalkylphenone type photoradical polymerization initiator having no dimethylamino group
- Irgacure 369 (manufactured by BASF Japan): ⁇ -aminoalkylphenone type photoradical polymerization having dimethylamino group
- Irgacure 379EG (manufactured by BASF Japan): ⁇ -aminoalkylphenone type photoradical polymerization initiator having a dimethylamino group TPO (manufactured by BASF Japan): acylphosphine oxide type photoradical polymerization initiator Irgacure 184 (BASF Japan) Manufactured by: ⁇ -hydroxyalkylphenone type photo radical polymerization initiator
- Example 1 80 parts by weight of trimethylolpropane triacrylate (“TMPTA” manufactured by Daicel Cytec Co., Ltd.) corresponding to the polyfunctional compound (A) and an ⁇ -aminoacetophenone type photoradical polymerization initiator (BASF) corresponding to the photopolymerization initiator (B) 5 parts by weight of “Irgacure 907” manufactured by Japan, 20 parts by weight of a bisphenol A type epoxy resin (“YD-127” manufactured by Nippon Steel Chemical Co., Ltd.) corresponding to the compound (C) having a cyclic ether group, and a synthesis example 4 parts by weight of the reaction viscous product obtained in 1 was mixed to obtain a curable composition for inkjet.
- TMPTA trimethylolpropane triacrylate
- BASF ⁇ -aminoacetophenone type photoradical polymerization initiator
- Example 2 50 parts by weight of a bisphenol F type epoxy resin (“YD-170” manufactured by Nippon Steel Chemical Co., Ltd.) corresponding to the compound (C) having a cyclic ether group, and neopentyl glycol corresponding to the compound (C) having a cyclic ether group 50 parts by weight of diglycidyl ether (“EX-211” manufactured by Nagase ChemteX) and 30 parts by weight of the reaction viscous product obtained in Synthesis Example 2 were mixed to obtain a curable composition for inkjet.
- a bisphenol F type epoxy resin (“YD-170” manufactured by Nippon Steel Chemical Co., Ltd.
- neopentyl glycol corresponding to the compound (C) having a cyclic ether group 50 parts by weight of diglycidyl ether (“EX-211” manufactured by Nagase ChemteX) and 30 parts by weight of the reaction viscous product obtained in Synthesis Example 2 were mixed to obtain a curable composition
- Examples 3 to 16 and Comparative Examples 1 to 5 A curable composition for inkjet was obtained in the same manner as in Example 1 except that the types and blending amounts of the blending components were changed as shown in Tables 1 to 3 below.
- the reaction viscous material is compatible with the polyfunctional compound and is compatible with the compound having a cyclic ether group. Further, it was dissolved in the curable composition.
- Viscosity exceeds 1200 mPa ⁇ s
- B Viscosity exceeds 1000 mPa ⁇ s
- C Viscosity exceeds 500 mPa ⁇ s, 1000 mPa ⁇ s or less
- D Viscosity is 160 mPa ⁇ s or more, 500 mPa ⁇ s or less
- E Viscosity is less than 160 mPa ⁇ s
- the ejection test of the obtained curable composition for inkjet was performed from the inkjet head of a piezo-type inkjet printer with an ultraviolet irradiation device, and evaluated according to the following criteria.
- the head temperature was set to 80 ° C. during the discharge test of the curable composition having a viscosity of 500 mPa ⁇ s or less, and the head temperature was set to 95 ° C. during the discharge test of the curable composition having a viscosity exceeding 500 mPa ⁇ s.
- the head temperature is set to 80 ° C.
- the head temperature is set to 95 ° C. C.
- the curable composition for inkjet (thickness 20 ⁇ m) coated on the substrate was irradiated with ultraviolet rays having a wavelength of 365 nm so that the irradiation energy was 1000 mJ / cm 2 .
- the wet spread of the pattern was visually observed, and the wet spread was determined according to the following criteria.
- Wet spread state is the target line width + 40 ⁇ m or less
- Wet spread state exceeds the target line width + 40 ⁇ m and 75 ⁇ m or less
- the composition layer is wet spread from the drawing portion, and between the lines The interval is lost or the wet spread condition exceeds the target line width +75 ⁇ m.
- An FR-4 substrate with a copper foil having a copper foil attached to the upper surface was prepared.
- the ink-jet curable composition was applied from the ink-jet head of a piezo-type ink-jet printer with an ultraviolet irradiation device so that the line width was 80 ⁇ m and the line-to-line spacing was 80 ⁇ m. I tried to draw a pattern. From the ejection properties from the inkjet head at this time, the storage stability was determined according to the following criteria.
- the head temperature was set to 80 ° C. during the discharge test of the curable composition having a viscosity of 500 mPa ⁇ s or less, and the head temperature was set to 95 ° C. during the discharge test of the curable composition having a viscosity exceeding 500 mPa ⁇ s.
- composition could be ejected from the inkjet head.
- X The composition was cured before ejection, or the viscosity of the composition was increased, and the composition could not be ejected from the inkjet head.
- the curable composition for inkjet (thickness 20 ⁇ m) drawn in a pattern was irradiated with ultraviolet rays having a wavelength of 365 nm so that the irradiation energy was 1000 mJ / cm 2 to obtain a primary cured product.
- the primary cured product was heated at 150 ° C. for 60 minutes to be fully cured to obtain a resist pattern as a cured product.
- the obtained laminate of the substrate and the resist pattern was heated in an oven at 270 ° C. for 5 minutes, and then the appearance of the heated resist pattern was visually inspected. Further, a cellophane tape was attached to the resist pattern after heating, and the cellophane tape was peeled off in the 90-degree direction.
- the heat resistance was determined by the following criteria based on the appearance inspection and the peel test.
- IPC-B-25 comb test pattern B Insulation reliability (migration resistance) IPC-B-25 comb test pattern B was prepared.
- the comb-type test pattern B is heated to 80 ° C., and the curable composition for inkjet is discharged from the inkjet head of the piezo-type inkjet printer with an ultraviolet irradiation device so as to cover the entire surface of the comb-type test pattern B. And coated.
- the head temperature was set to 80 ° C. during the discharge test of the curable composition having a viscosity of 500 mPa ⁇ s or less, and the head temperature was set to 95 ° C. during the discharge test of the curable composition having a viscosity exceeding 500 mPa ⁇ s.
- the coated curable composition for inkjet (thickness 20 ⁇ m) was irradiated with ultraviolet rays having a wavelength of 365 nm so that the irradiation energy was 1000 mJ / cm 2 to obtain a primary cured product.
- the primary cured product was heated at 150 ° C. for 60 minutes to be fully cured to form a resist pattern as a cured product, thereby obtaining a test piece.
- the obtained test piece was subjected to a humidification test for 500 hours under the conditions of applying 85 ° C., 85% relative humidity and 50V direct current.
- the insulation resistance after the humidification test was measured.
- Tables 1 to 3 below “-” indicates no evaluation.
- the curable composition for inkjet which was not heated at 80 degreeC for 12 hours was used.
- * 1 indicates that the insulation resistance is 1 ⁇ 10 9 or more and less than 3 ⁇ 10 10 .
- the photopolymerization initiators of Examples 15 and 16 using a photopolymerization initiator other than the ⁇ -aminoacetophenone type photopolymerization initiator were used.
- the curable composition there is less stickiness of the surface of the primary cured product after light irradiation and before thermal curing, it is excellent in thermal curability during thermal curing, and the insulation reliability evaluation results are also good. It was.
- Example 17 65 parts by weight of tricyclodecane dimethanol diacrylate corresponding to the polyfunctional compound (A), 30 parts by weight of isobornyl acrylate corresponding to the monofunctional compound (E), and Irgacure corresponding to the photopolymerization initiator (B) 907 ( ⁇ -aminoacetophenone type photoradical polymerization initiator, manufactured by BASF Japan) was mixed with 5 parts by weight to obtain a curable composition for inkjet.
- Example 18 to 40 A curable composition for inkjet was obtained in the same manner as in Example 17 except that the types and blending amounts of the blending components were changed as shown in Tables 5 and 6 below.
- the reaction viscous material is compatible with the polyfunctional compound, is compatible with the monofunctional compound, and is a cyclic ether group. And was dissolved in the curable composition.
- Insulation resistance is 3 ⁇ 10 10 ⁇ or more ⁇ : Insulation resistance is 1 ⁇ 10 9 or more and less than 3 ⁇ 10 10 ⁇ : Insulation resistance is less than 1 ⁇ 10 9
- Heat and humidity resistance (heat resistance and moisture resistance)
- a glass epoxy substrate (100 mm ⁇ 100 mm) provided with copper wiring on the upper surface was prepared.
- substrate the curable composition for inkjet was discharged from the inkjet head of the piezo-type inkjet printer with an ultraviolet irradiation device, and was coated on the whole surface.
- the head temperature was set to 80 ° C. during the discharge test of the curable composition having a viscosity of 500 mPa ⁇ s or less, and the head temperature was set to 95 ° C. during the discharge test of the curable composition having a viscosity exceeding 500 mPa ⁇ s.
- the curable composition for inkjet (thickness 20 ⁇ m) coated on the substrate is irradiated with ultraviolet light having a wavelength of 365 nm so that the irradiation energy is 1000 mJ / cm 2, and then heated at 180 ° C. for 1 hour to obtain a cured product. (Thickness 20 ⁇ m) was obtained.
- the resulting laminate of the substrate and the cured product was allowed to stand for 24 hours under conditions of 130 ° C. and relative humidity of 85% RH. Thereafter, the adhesion of the cured product to the substrate was confirmed by a cross-cut tape test (JIS 5400 6.15), and the moisture and heat resistance was determined according to the following criteria. Make a cut of 100 squares at 1 mm intervals with a cutter on the cured product, and then apply cellophane tape (JIS Z1522) to the cured product with the incised part, and attach one end of the tape to a 45 degree angle. And peeled off strongly to confirm the peeled state.
- JIS 5400 6.15 cross-cut tape test
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Medicinal Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Polymers & Plastics (AREA)
- Manufacturing & Machinery (AREA)
- Pest Control & Pesticides (AREA)
- Power Engineering (AREA)
- Electromagnetism (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Physics & Mathematics (AREA)
- General Chemical & Material Sciences (AREA)
- Sustainable Development (AREA)
- General Physics & Mathematics (AREA)
- Computer Hardware Design (AREA)
- Inks, Pencil-Leads, Or Crayons (AREA)
- Ink Jet Recording Methods And Recording Media Thereof (AREA)
- Epoxy Resins (AREA)
- Manufacturing Of Printed Wiring (AREA)
Abstract
Description
本発明に係るインクジェット用硬化性組成物のさらに他の特定の局面では、上記(メタ)アクリロイル基を2個以上有する多官能化合物が、多環骨格を有し、かつ(メタ)アクリロイル基を2個以上有する多官能化合物であり、多環骨格を有し、かつ(メタ)アクリロイル基を1個有する単官能化合物がさらに含まれている。
本発明に係る電子部品の製造方法の他の特定の局面では、上記インクジェット用硬化性組成物として、(メタ)アクリロイル基を2個以上有する多官能化合物と、光重合開始剤とを含むインクジェット用硬化性組成物を用いて、上記硬化物層を形成する工程において、パターン状に描画された上記インクジェット用硬化性組成物に光を照射及び熱を付与し、硬化させて、硬化物層を形成する。
本発明に係るインクジェット用硬化性組成物は、環状エーテル基を有する化合物(C)と、硬化剤(D)とを含む。本発明に係るインクジェット用硬化性組成物は、(メタ)アクリロイル基を2個以上有する多官能化合物(A)と、光重合開始剤(B)とをさらに含むことが好ましい。硬化剤(D)は、ジシアンジアミドと、該ジシアンジアミドと反応しうる官能基を有する官能基含有化合物とを反応させた反応粘稠物である。上記「(メタ)アクリロイル基」の用語は、アクリロイル基とメタクリロイル基とを示す。
光の照射により硬化性組成物を硬化させるために、本発明に係るインクジェット用硬化性組成物は、(メタ)アクリロイル基を2個以上有する多官能化合物(A)を含むことが好ましい。多官能化合物(A)は、(メタ)アクリロイル基を2個以上有すれば特に限定されない。多官能化合物(A)として、(メタ)アクリロイル基を2個以上有する従来公知の多官能化合物を用いることができる。多官能化合物(A)は、(メタ)アクリロイル基を2個以上有するため、光の照射により重合が進行し、硬化する。このため、硬化性組成物を塗工した後に光を照射することにより硬化を進行させることができ、塗工された形状を保持することができ、光が照射された硬化性組成物の一次硬化物及び硬化物が過度に濡れ拡がるのを効果的に抑制することができる。多官能化合物(A)は、1種のみが用いられてもよく、2種以上が併用されてもよい。
光の照射により硬化性組成物を硬化させるために、本発明に係るインクジェット用硬化性組成物は、多官能化合物(A)とともに、光重合開始剤(B)を含むことが好ましい。光重合開始剤(B)としては、光ラジカル重合開始剤及び光カチオン重合開始剤等が挙げられる。光重合開始剤(B)は、光ラジカル重合開始剤であることが好ましい。光重合開始剤(B)は、1種のみが用いられてもよく、2種以上が併用されてもよい。
本発明に係るインクジェット用硬化性組成物は、熱の付与によって硬化可能であるように、環状エーテル基を有する化合物(C)を含む。化合物(C)の使用により、熱の付与により硬化性組成物又は該硬化性組成物の一次硬化物をさらに硬化させることができる。このため、化合物(C)の使用により、レジストパターンを効率的にかつ精度よく形成することができ、更に硬化物の耐熱性及び絶縁信頼性を高めることができる。環状エーテル基を有する化合物(C)は1種のみが用いられてもよく、2種以上が併用されてもよい。
エポキシ基を有する化合物の具体例としては、ビスフェノールS型エポキシ化合物、ジグリシジルフタレート化合物、トリグリシジルイソシアヌレートなどの複素環式エポキシ化合物、ビキシレノール型エポキシ化合物、ビフェノール型エポキシ化合物、テトラグリシジルキシレノイルエタン化合物、ビスフェノールA型エポキシ化合物、水添ビスフェノールA型エポキシ化合物、ビスフェノールF型エポキシ化合物、臭素化ビスフェノールA型エポキシ化合物、フェノールノボラック型エポキシ化合物、クレゾールノボラック型エポキシ化合物、脂環式エポキシ化合物、ビスフェノールAのノボラック型エポキシ化合物、キレート型エポキシ化合物、グリオキザール型エポキシ化合物、アミノ基含有エポキシ化合物、ゴム変性エポキシ化合物、ジシクロペンタジエンフェノリック型エポキシ化合物、シリコーン変性エポキシ化合物及びε-カプロラクトン変性エポキシ化合物等が挙げられる。
硬化剤(D)は、ジシアンジアミドと、該ジシアンジアミドと反応しうる官能基を有する官能基含有化合物とを反応させた反応粘稠物である。このような硬化剤(D)が用いられているので、上記硬化性組成物は、熱の付与により硬化可能である。また、環状エーテル基を有する化合物(C)と硬化剤(D)とを併用した硬化性組成物を熱硬化させることにより、硬化物による絶縁信頼性を高くすることができる。
本発明に係るインクジェット用硬化性組成物は、多環骨格を有し、かつ(メタ)アクリロイル基を1個有する単官能化合物(E)を含むことが好ましい。さらに、本発明に係るインクジェット用硬化性組成物は、多環骨格を有し、かつ(メタ)アクリロイル基を2個以上有する多官能化合物(A1)と、多環骨格を有し、かつ(メタ)アクリロイル基を1個有する単官能化合物(E)とを含むことがより好ましい。これらの場合には、上記インクジェット用硬化性組成物の硬化物の耐湿熱性がかなり高くなる。従って、本発明に係るインクジェット用硬化性組成物を用いたプリント配線板などの電子部品をより一層長期間使用でき、かつ該電子部品の信頼性がより一層高くなる。また、単官能化合物(E)の使用により、硬化物の耐湿熱性が高くなるだけでなく、硬化性組成物の吐出性も高くなる。なお、多環骨格を有し、かつ(メタ)アクリロイル基を1個有する単官能化合物(E)を用いた場合には、多環骨格を有さず、かつ(メタ)アクリロイル基を1個有する単官能化合物を用いた場合と比べて、硬化物の耐湿熱性が高くなる。
本発明に係るインクジェット用硬化性組成物は、上記反応粘稠物以外の熱硬化剤を含んでいてもよい。さらに、本発明に係るインクジェット用硬化性組成物は、硬化促進剤を含んでいてもよい。
次に、本発明に係る電子部品の製造方法について説明する。
攪拌器、温度計、滴下ロートを備えた3つ口フラスコに、メチルセロソルブ50g、ジシアンジアミド15g、及び2,4-ジアミノ-6-[2’-ウンデシルイミダゾリル-(1’)]-エチル-s-トリアジン1gを加え、100℃に加熱してジシアンジアミドを溶解させた。溶解後、ブチルグリシジルエーテル130gを滴下ロートから20分かけて滴下し、1時間反応させた。その後60℃に温度を下げ、減圧にして溶媒を除去し、黄色及び半透明の反応粘稠物を得た。得られた反応粘稠物は溶媒を含んでいなかった。
攪拌器、温度計、滴下ロートを備えた3つ口フラスコに、メチルセロソルブ50g、ジシアンジアミド15g、及び2,4-ジアミノ-6-[2’-ウンデシルイミダゾリル-(1’)]-エチル-s-トリアジン1gを加え、100℃に加熱してジシアンジアミドを溶解させた。溶解後、オルソクレジルグリシジルエーテル40gを滴下ロートから20分かけて滴下し、1時間反応させた。その後60℃に温度を下げ、減圧にして溶媒を除去し、黄色及び半透明の反応粘稠物を得た。得られた反応粘稠物は溶媒を含んでいなかった。
オルソクレジルグリシジルエーテルの滴下量を40gから95gに変更したこと以外は合成例2と同様にして、黄色及び半透明の反応粘稠物を得た。得られた反応粘稠物は溶媒を含んでいなかった。
Irgacure 907(BASFジャパン社製):ジメチルアミノ基を有さないα-アミノアルキルフェノン型光ラジカル重合開始剤
Irgacure 369(BASFジャパン社製):ジメチルアミノ基を有するα-アミノアルキルフェノン型光ラジカル重合開始剤
Irgacure 379EG(BASFジャパン社製):ジメチルアミノ基を有するα-アミノアルキルフェノン型光ラジカル重合開始剤
TPO(BASFジャパン社製):アシルフォスフィンオキサイド型光ラジカル重合開始剤
Irgacure 184(BASFジャパン社製):α-ヒドロキシアルキルフェノン型光ラジカル重合開始剤
多官能化合物(A)に相当するトリメチロールプロパントリアクリレート(ダイセルサイテック社製「TMPTA」)80重量部と、光重合開始剤(B)に相当するα-アミノアセトフェノン型光ラジカル重合開始剤(BASFジャパン社製「Irgacure 907」)5重量部と、環状エーテル基を有する化合物(C)に相当するビスフェノールA型エポキシ樹脂(新日鐵化学社製「YD-127」)20重量部と、合成例1で得られた反応粘稠物4重量部とを混合し、インクジェット用硬化性組成物を得た。
環状エーテル基を有する化合物(C)に相当するビスフェノールF型エポキシ樹脂(新日鐵化学社製「YD-170」)50重量部と、環状エーテル基を有する化合物(C)に相当するネオペンチルグリコールジグリシジルエーテル(ナガセケムテックス社製「EX-211」)50重量部と、合成例2で得られた反応粘稠物30重量部とを混合し、インクジェット用硬化性組成物を得た。
配合成分の種類及び配合量を下記の表1~3に示すように変更したこと以外は実施例1と同様にして、インクジェット用硬化性組成物を得た。
(1)粘度
JIS K2283に準拠して、粘度計(東機産業社製「TVE22L」)を用いて、得られたインクジェット用硬化性組成物の25℃での粘度を測定した。インクジェット用硬化性組成物の粘度を下記の判定基準で判定した。
A:粘度が1200mPa・sを超える
B:粘度が1000mPa・sを超え、1200mPa・s以下
C:粘度が500mPa・sを超え、1000mPa・s以下
D:粘度が160mPa・s以上、500mPa・s以下
E:粘度が160mPa・s未満
紫外線照射装置付きピエゾ方式インクジェットプリンタのインクジェットヘッドから、得られたインクジェット用硬化性組成物の吐出試験を行い、下記の判断基準で評価した。なお、粘度が500mPa・s以下である硬化性組成物の吐出試験時には、ヘッド温度を80℃とし、粘度が500mPa・sを超える硬化性組成物の吐出試験時には、ヘッド温度を95℃とした。
○○:硬化性組成物をヘッドから10時間以上連続して吐出可能であった
○:硬化性組成物をヘッドから10時間以上連続して吐出可能であるが、10時間の連続吐出の間にわずかに吐出むらが生じる
△:硬化性組成物をヘッドから連続して吐出可能であるが、10時間以上連続して吐出不可能であった
×硬化性組成物をヘッドから吐出の初期段階で吐出不可能であった
銅箔が上面に貼り付けられている銅箔付きFR-4基板を用意した。この基板上に銅箔の表面の全体を覆うようにインクジェット用硬化性組成物を、紫外線照射装置付きピエゾ方式インクジェットプリンタのインクジェットヘッドから、ラインの幅80μmでライン間の間隔が80μmとなるように吐出して塗工し、パターン状に描画した。なお、粘度が500mPa・s以下であるインクジェット用硬化性組成物の吐出試験時には、ヘッド温度を80℃とし、粘度が500mPa・sを超えるインクジェット用硬化性組成物の吐出試験時には、ヘッド温度を95℃とした。
○○:濡れ拡がりの状態が、狙いのライン幅+40μm以下
○:濡れ拡がりの状態が、狙いのライン幅+40μmを超え、75μm以下
×:描画部分から組成物層が濡れ拡がっており、ライン間の間隔が無くなっているか、又は濡れ拡がりの状態が、狙いのライン幅+75μmを超える
5μmのメンブレンフィルターを用いて、得られたインクジェット用硬化性組成物をろ過し、ろ過したインクジェット用硬化性組成物を80℃で12時間加熱した。
○:インクジェットヘッドから組成物を吐出できた
×:吐出前に組成物が硬化しているか、又は組成物の粘度が上昇しており、インクジェットヘッドから組成物を吐出できなかった
銅箔が上面に貼り付けられている銅箔付きFR-4基板を用意した。基板上の銅箔上に、インクジェット用硬化性組成物を、紫外線照射装置付きピエゾ方式インクジェットプリンタのインクジェットヘッドから、ラインの幅80μmでライン間の間隔が80μmとなるように吐出して塗工し、パターン状に描画した。なお、粘度が500mPa・s以下である硬化性組成物の吐出試験時には、ヘッド温度を80℃とし、粘度が500mPa・sを超える硬化性組成物の吐出試験時には、ヘッド温度を95℃とした。
○:外観検査において加熱前後でレジストパターンに変化がなく、かつ剥離試験においてレジストパターンが基板から剥離しなかった
×:外観検査においてレジストパターンにクラック、剥離及び膨れの内の少なくとも1つがあるか、又は剥離試験においてレジストパターンが基板から剥離した
IPC-B-25のくし型テストパターンBを用意した。このくし型テストパターンBを80℃に加温して、くし型テストパターンBの表面の全体を覆うようにインクジェット用硬化性組成物を、紫外線照射装置付きピエゾ方式インクジェットプリンタのインクジェットヘッドから、吐出して塗工した。なお、粘度が500mPa・s以下である硬化性組成物の吐出試験時には、ヘッド温度を80℃とし、粘度が500mPa・sを超える硬化性組成物の吐出試験時には、ヘッド温度を95℃とした。
結果を下記の表1~3に示す。なお、下記の表1~3において、「-」は評価していないことを示す。なお、貯蔵安定性の評価を除いて、80℃で12時間加熱していないインクジェット用硬化性組成物を用いた。なお、下記の表3において、*1は、絶縁抵抗が1×109以上、3×1010未満であることを示す。
多官能化合物(A)に相当するトリシクロデカンジメタノールジアクリレート65重量部と、単官能化合物(E)に相当するイソボルニルアクリレート30重量部と、光重合開始剤(B)に相当するIrgacure 907(α-アミノアセトフェノン型光ラジカル重合開始剤、BASFジャパン社製)5重量部とを混合し、インクジェット用硬化性組成物を得た。
配合成分の種類及び配合量を下記の表5,6に示すように変更したこと以外は実施例17と同様にして、インクジェット用硬化性組成物を得た。
実施例17~40について、実施例1~16及び比較例1~5と同様の評価項目について同様にして評価を実施した。但し、絶縁信頼性(耐マイグレーション性)の評価に関しては、絶縁抵抗を下記の基準で判定し、結果を下記の表5,6に示した。
○:絶縁抵抗が3×1010Ω以上
△:絶縁抵抗が1×109以上、3×1010未満
×:絶縁抵抗が1×109未満
銅配線が上面に設けられたガラスエポキシ基板(100mm×100mm)を用意した。この基板上にインクジェット用硬化性組成物を、紫外線照射装置付きピエゾ方式インクジェットプリンタのインクジェットヘッドから吐出して、全面に塗工した。なお、粘度が500mPa・s以下である硬化性組成物の吐出試験時には、ヘッド温度を80℃とし、粘度が500mPa・sを超える硬化性組成物の吐出試験時には、ヘッド温度を95℃とした。
○○:テープの剥離時に硬化物の剥離なし
○:テープの剥離時に硬化物の一部が剥離する
△:テープの剥離時に硬化物の全部が剥離する
×:テープの剥離前に硬化物が剥離している
Claims (22)
- インクジェット方式により塗工され、かつ熱の付与により硬化可能であるインクジェット用硬化性組成物であって、
環状エーテル基を有する化合物と、
硬化剤とを含み、
前記硬化剤が、ジシアンジアミドと、該ジシアンジアミドと反応しうる官能基を有する官能基含有化合物とを反応させた反応粘稠物である、インクジェット用硬化性組成物。 - 前記ジシアンジアミドと反応される前記官能基含有化合物が、水酸基、環状エーテル基、カルボキシル基及びイソシアネート基からなる群から選択された少なくとも1種の官能基を有する、請求項1に記載のインクジェット用硬化性組成物。
- 前記ジシアンジアミドと反応される前記官能基含有化合物が、エポキシ基を有する化合物ある、請求項2に記載のインクジェット用硬化性組成物。
- 前記ジシアンジアミドと反応される前記エポキシ基を有する化合物が、エポキシ基を1個有する、請求項3に記載のインクジェット用硬化性組成物。
- 前記ジシアンジアミドと反応される前記官能基含有化合物が、芳香族骨格を有する、請求項1~4のいずれか1項に記載のインクジェット用硬化性組成物。
- 前記反応粘稠物が、ジシアンジアミド1モルに対して、前記官能基含有化合物を1モル以上、3モル以下反応させた反応粘稠物である、請求項1~5のいずれか1項に記載のインクジェット用硬化性組成物。
- 前記官能基含有化合物と反応される前記ジシアンジアミドが、粉末状である、請求項1~6のいずれか1項に記載のインクジェット用硬化性組成物。
- 前記反応粘稠物が、硬化性組成物中に溶解している、請求項1~7のいずれか1項に記載のインクジェット用硬化性組成物。
- インクジェット方式により塗工され、かつ光の照射と熱の付与とにより硬化可能であるインクジェット用硬化性組成物であって、
(メタ)アクリロイル基を2個以上有する多官能化合物と、
光重合開始剤とをさらに含む、請求項1~8のいずれか1項に記載のインクジェット用硬化性組成物。 - 前記光重合開始剤が、α-アミノアルキルフェノン型光ラジカル重合開始剤である、請求項9に記載のインクジェット用硬化性組成物。
- 前記光重合開始剤が、ジメチルアミノ基を有するα-アミノアルキルフェノン型光ラジカル重合開始剤である、請求項9に記載のインクジェット用硬化性組成物。
- 前記(メタ)アクリロイル基を2個以上有する多官能化合物が、多環骨格を有し、かつ(メタ)アクリロイル基を2個以上有する多官能化合物である、請求項9~11のいずれか1項に記載のインクジェット用硬化性組成物。
- 多環骨格を有し、かつ(メタ)アクリロイル基を1個有する単官能化合物をさらに含む、請求項9~11のいずれか1項に記載のインクジェット用硬化性組成物。
- 前記(メタ)アクリロイル基を2個以上有する多官能化合物が、多環骨格を有し、かつ(メタ)アクリロイル基を2個以上有する多官能化合物であり、
多環骨格を有し、かつ(メタ)アクリロイル基を1個有する単官能化合物をさらに含む、請求項9~11のいずれか1項に記載のインクジェット用硬化性組成物。 - 前記多官能化合物と前記単官能化合物と前記光重合開始剤との合計100重量%中、前記多官能化合物の含有量が20重量%以上、70重量%以下であり、かつ前記単官能化合物の含有量が5重量%以上、50重量%以下である、請求項14に記載のインクジェット用硬化性組成物。
- 前記単官能化合物が、イソボルニル(メタ)アクリレート、ジヒドロキシシクロペンタジエニル(メタ)アクリレート、ジシクロペンテニル(メタ)アクリレート、ジシクロペンテニルオキシエチル(メタ)アクリレート及びジシクロペンタニル(メタ)アクリレートからなる群から選択された少なくとも1種である、請求項13~15のいずれか1項に記載のインクジェット用硬化性組成物。
- 前記多官能化合物が、トリシクロデカンジメタノールジ(メタ)アクリレートである、請求項9~16のいずれか1項に記載のインクジェット用硬化性組成物。
- 前記反応粘稠物が、ジシアンジアミドの活性水素の一部に、前記官能基含有化合物の前記官能基を反応させた反応物である、請求項1~17のいずれか1項に記載のインクジェット用硬化性組成物。
- JIS K2283に準拠して測定された25℃での粘度が160mPa・s以上、1200mPa・s以下である、請求項1~18のいずれか1項に記載のインクジェット用硬化性組成物。
- 請求項1~19のいずれか1項に記載のインクジェット用硬化性組成物を、インクジェット方式にて塗工し、パターン状に描画する工程と、
パターン状に描画された前記インクジェット用硬化性組成物に熱を付与し、硬化させて、硬化物層を形成する工程とを備える、電子部品の製造方法。 - レジストパターンを有する電子部品であるプリント配線板の製造方法であって、
前記インクジェット用硬化性組成物を、インクジェット方式にて塗工し、パターン状に描画し、パターン状に描画された前記インクジェット用硬化性組成物に熱を付与し、硬化させて、レジストパターンを形成する、請求項20に記載の電子部品の製造方法。 - 前記インクジェット用硬化性組成物として、(メタ)アクリロイル基を2個以上有する多官能化合物と、光重合開始剤とを含むインクジェット用硬化性組成物を用いて、
前記硬化物層を形成する工程において、パターン状に描画された前記インクジェット用硬化性組成物に光を照射及び熱を付与し、硬化させて、硬化物層を形成する、請求項20又は21に記載の電子部品の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020127001661A KR101485225B1 (ko) | 2010-09-24 | 2011-09-20 | 잉크젯용 경화성 조성물 및 전자 부품의 제조 방법 |
| JP2011539831A JP5965642B2 (ja) | 2010-09-24 | 2011-09-20 | インクジェット用硬化性組成物及び電子部品の製造方法 |
| EP11826817.6A EP2620478B1 (en) | 2010-09-24 | 2011-09-20 | Curable composition for inkjet and method for producing electronic component |
| CN201180009526.0A CN102753630B (zh) | 2010-09-24 | 2011-09-20 | 喷墨用固化性组合物及电子部件的制造方法 |
| US13/825,535 US10160881B2 (en) | 2010-09-24 | 2011-09-20 | Curable composition for inkjet and method for producing electronic component |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010-214252 | 2010-09-24 | ||
| JP2010214252 | 2010-09-24 | ||
| JP2011015307A JP5416725B2 (ja) | 2010-09-24 | 2011-01-27 | インクジェット用硬化性組成物及びプリント配線板の製造方法 |
| JP2011-015307 | 2011-01-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2012039372A1 true WO2012039372A1 (ja) | 2012-03-29 |
Family
ID=45873859
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/071327 WO2012039372A1 (ja) | 2010-09-24 | 2011-09-20 | インクジェット用硬化性組成物及び電子部品の製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10160881B2 (ja) |
| EP (1) | EP2620478B1 (ja) |
| JP (1) | JP5416725B2 (ja) |
| KR (1) | KR101485225B1 (ja) |
| CN (1) | CN102753630B (ja) |
| TW (1) | TWI518105B (ja) |
| WO (1) | WO2012039372A1 (ja) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104685010A (zh) * | 2012-09-27 | 2015-06-03 | 积水化学工业株式会社 | 喷墨用固化性组合物及电子零件的制造方法 |
| JPWO2013161270A1 (ja) * | 2012-04-24 | 2015-12-21 | コニカミノルタ株式会社 | 活性光線硬化型インクジェットインク、及びこれを用いた画像形成方法 |
| US20160046813A1 (en) * | 2013-11-06 | 2016-02-18 | Sekisui Chemical Co., Ltd. | Method for manufacturing cured film, method for manufacturing electronic component, and electronic component |
| JP2017066170A (ja) * | 2015-09-28 | 2017-04-06 | 積水化学工業株式会社 | 電子部品の製造方法 |
| WO2017169166A1 (ja) * | 2016-03-31 | 2017-10-05 | 太陽インキ製造株式会社 | インクジェット用硬化性組成物、硬化物およびプリント配線板 |
| JP2019522689A (ja) * | 2016-05-17 | 2019-08-15 | マイクロクラフト コリア カンパニー リミテッド | インクジェット樹脂組成物の調製方法 |
| JP2019524900A (ja) * | 2017-06-08 | 2019-09-05 | マイクロクラフト コリア カンパニー リミテッド | インクジェット印刷用樹脂組成物およびそれを使用して調製されたプリント配線板 |
| JP2019189848A (ja) * | 2018-04-20 | 2019-10-31 | 積水化学工業株式会社 | 電子デバイス用光硬化性樹脂組成物 |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5416725B2 (ja) | 2010-09-24 | 2014-02-12 | 積水化学工業株式会社 | インクジェット用硬化性組成物及びプリント配線板の製造方法 |
| JP6374722B2 (ja) * | 2014-07-23 | 2018-08-15 | 積水化学工業株式会社 | インクジェット用熱硬化性接着剤、半導体装置の製造方法及び電子部品 |
| EP3210946B1 (en) * | 2016-02-29 | 2020-07-08 | Agfa-Gevaert | Method of manufacturing an etched glass article |
Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59126428A (ja) * | 1983-01-06 | 1984-07-21 | Dai Ichi Kogyo Seiyaku Co Ltd | エポキシ樹脂用硬化剤 |
| JPS63205313A (ja) * | 1987-02-23 | 1988-08-24 | Hitachi Ltd | エポキシ樹脂用硬化剤 |
| JPH02283718A (ja) * | 1989-04-25 | 1990-11-21 | Dainippon Ink & Chem Inc | エポキシ樹脂組成物、プリプレグ及び積層板 |
| JPH0374086B2 (ja) | 1986-02-01 | 1991-11-25 | ||
| JPH0578454A (ja) * | 1991-09-25 | 1993-03-30 | Hitachi Ltd | エポキシ樹脂用硬化剤 |
| JP2000239418A (ja) * | 1999-02-19 | 2000-09-05 | Hitachi Chem Co Ltd | 印刷配線板用プリプレグ及びそれを用いた積層体 |
| JP2005060520A (ja) * | 2003-08-12 | 2005-03-10 | Konica Minolta Medical & Graphic Inc | 硬化組成物とそれを用いたインクジェット用インク及び硬化物形成方法 |
| JP2009506187A (ja) * | 2005-08-31 | 2009-02-12 | プリンター リミテッド | Uv硬化可能なハイブリッド硬化インクジェットインク組成物およびそれを使用するソルダマスク |
| JP2010111716A (ja) * | 2008-11-04 | 2010-05-20 | Chisso Corp | インクジェット用インク |
| JP2010143982A (ja) * | 2008-12-17 | 2010-07-01 | Chisso Corp | 光硬化性インクジェット用インク |
| JP2011021079A (ja) * | 2009-07-14 | 2011-02-03 | Chisso Corp | インクジェット用インク |
| JP2011026403A (ja) * | 2009-07-23 | 2011-02-10 | Chisso Corp | 光硬化性インクジェット用インク |
Family Cites Families (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4032196B2 (ja) | 1998-08-05 | 2008-01-16 | 富士フイルム株式会社 | インクジェット用油溶性インク |
| CN1293116C (zh) * | 2000-02-14 | 2007-01-03 | 太阳油墨制造株式会社 | 形成消光涂膜用的光固化性·热固化性组合物 |
| JP2001240782A (ja) | 2000-02-29 | 2001-09-04 | Asahi Optical Co Ltd | サーマルタイプのインクジェットプリンター及びインク転写プリンター用油性インク |
| TW562844B (en) | 2001-01-19 | 2003-11-21 | Kolon Inc | Ink composition for solder resist |
| KR20030013924A (ko) * | 2001-08-10 | 2003-02-15 | 주식회사 코오롱 | 솔더 레지스트용 잉크 조성물 |
| JP5116921B2 (ja) * | 2001-02-02 | 2013-01-09 | 太陽ホールディングス株式会社 | 配線板用熱硬化性樹脂組成物とその成形体および多層プリント配線板 |
| JP2002331739A (ja) | 2001-05-09 | 2002-11-19 | Fuji Xerox Co Ltd | インクジェット記録方法およびインクジェット記録装置 |
| CN1446864A (zh) * | 2002-03-26 | 2003-10-08 | 冠品化学股份有限公司 | 一种制造无溶剂感光热固化型油墨的方法 |
| JP2004034545A (ja) | 2002-07-04 | 2004-02-05 | Konica Minolta Holdings Inc | インクジェット記録方法、インク及びインクジェットプリンタ |
| CN1194025C (zh) | 2002-09-28 | 2005-03-23 | 北京科化新材料科技有限公司 | 液化双氰胺及其制备方法 |
| TWI288142B (en) | 2003-05-09 | 2007-10-11 | Taiyo Ink Mfg Co Ltd | Photocuring/thermosetting ink jet composition and printed wiring board using same |
| JP4290510B2 (ja) | 2003-08-22 | 2009-07-08 | 太陽インキ製造株式会社 | インクジェット用光硬化性・熱硬化性組成物とそれを用いたプリント配線板 |
| JP3912405B2 (ja) | 2003-11-11 | 2007-05-09 | 三菱化学株式会社 | 硬化性組成物、硬化物、カラーフィルタ及び液晶表示装置 |
| US7439285B2 (en) | 2004-01-23 | 2008-10-21 | Printar Ltd. | Liquid thermosetting ink |
| JP2005255793A (ja) | 2004-03-10 | 2005-09-22 | Hitachi Chem Co Ltd | 無溶剤1液型の熱硬化性エポキシ樹脂組成物 |
| JP4687224B2 (ja) | 2004-04-23 | 2011-05-25 | 東レ株式会社 | エポキシ樹脂組成物、プリプレグおよび繊維強化複合材料 |
| JP2006117389A (ja) | 2004-10-21 | 2006-05-11 | Seiko Epson Corp | 媒体給送装置の駆動方法、駆動制御プログラム及び記録装置 |
| JP4925656B2 (ja) | 2005-12-12 | 2012-05-09 | レンゴー株式会社 | インクジェット印刷物 |
| JP4873621B2 (ja) | 2006-05-11 | 2012-02-08 | 日本化薬株式会社 | 機能素子及びそれに用いられるネガ型感光性樹脂組成物並びに機能素子の製造方法 |
| JP5034321B2 (ja) * | 2006-05-31 | 2012-09-26 | Dic株式会社 | 紫外線硬化型インクジェット記録用インク組成物 |
| JP4049194B2 (ja) | 2006-06-26 | 2008-02-20 | セイコーエプソン株式会社 | カラーフィルター |
| JP2008106165A (ja) * | 2006-10-26 | 2008-05-08 | Chisso Corp | インクジェット用インクおよび当該インクにより得られる硬化膜 |
| JP2008166451A (ja) | 2006-12-27 | 2008-07-17 | Furukawa Electric Co Ltd:The | チップ保護用フィルム |
| WO2008093071A1 (en) * | 2007-01-29 | 2008-08-07 | Sericol Limited | A printing ink |
| JP5305502B2 (ja) | 2008-03-07 | 2013-10-02 | 日本化薬株式会社 | 機能素子及びそれに用いられるネガ型感光性樹脂組成物並びに機能素子の製造方法 |
| JP5187000B2 (ja) | 2008-05-27 | 2013-04-24 | コニカミノルタホールディングス株式会社 | インクジェット記録装置及びインクジェット記録方法 |
| EP2620479B1 (en) | 2010-09-22 | 2016-06-22 | Sekisui Chemical Co., Ltd. | Curable composition for inkjet, and method for producing electronic component |
| JP5416725B2 (ja) | 2010-09-24 | 2014-02-12 | 積水化学工業株式会社 | インクジェット用硬化性組成物及びプリント配線板の製造方法 |
| JP2012087284A (ja) | 2010-09-24 | 2012-05-10 | Sekisui Chem Co Ltd | インクジェット用硬化性組成物及びプリント配線板の製造方法 |
| JP2013193127A (ja) | 2012-03-22 | 2013-09-30 | Toyota Motor Corp | 成形装置、及び成形方法 |
| JP2014153598A (ja) | 2013-02-12 | 2014-08-25 | Canon Inc | 可変焦点光学素子 |
-
2011
- 2011-01-27 JP JP2011015307A patent/JP5416725B2/ja active Active
- 2011-09-20 CN CN201180009526.0A patent/CN102753630B/zh active Active
- 2011-09-20 WO PCT/JP2011/071327 patent/WO2012039372A1/ja active Application Filing
- 2011-09-20 EP EP11826817.6A patent/EP2620478B1/en active Active
- 2011-09-20 US US13/825,535 patent/US10160881B2/en active Active
- 2011-09-20 KR KR1020127001661A patent/KR101485225B1/ko active IP Right Grant
- 2011-09-23 TW TW100134448A patent/TWI518105B/zh active
Patent Citations (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59126428A (ja) * | 1983-01-06 | 1984-07-21 | Dai Ichi Kogyo Seiyaku Co Ltd | エポキシ樹脂用硬化剤 |
| JPH0374086B2 (ja) | 1986-02-01 | 1991-11-25 | ||
| JPS63205313A (ja) * | 1987-02-23 | 1988-08-24 | Hitachi Ltd | エポキシ樹脂用硬化剤 |
| JPH02283718A (ja) * | 1989-04-25 | 1990-11-21 | Dainippon Ink & Chem Inc | エポキシ樹脂組成物、プリプレグ及び積層板 |
| JPH0578454A (ja) * | 1991-09-25 | 1993-03-30 | Hitachi Ltd | エポキシ樹脂用硬化剤 |
| JP2000239418A (ja) * | 1999-02-19 | 2000-09-05 | Hitachi Chem Co Ltd | 印刷配線板用プリプレグ及びそれを用いた積層体 |
| JP2005060520A (ja) * | 2003-08-12 | 2005-03-10 | Konica Minolta Medical & Graphic Inc | 硬化組成物とそれを用いたインクジェット用インク及び硬化物形成方法 |
| JP2009506187A (ja) * | 2005-08-31 | 2009-02-12 | プリンター リミテッド | Uv硬化可能なハイブリッド硬化インクジェットインク組成物およびそれを使用するソルダマスク |
| JP2010111716A (ja) * | 2008-11-04 | 2010-05-20 | Chisso Corp | インクジェット用インク |
| JP2010143982A (ja) * | 2008-12-17 | 2010-07-01 | Chisso Corp | 光硬化性インクジェット用インク |
| JP2011021079A (ja) * | 2009-07-14 | 2011-02-03 | Chisso Corp | インクジェット用インク |
| JP2011026403A (ja) * | 2009-07-23 | 2011-02-10 | Chisso Corp | 光硬化性インクジェット用インク |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2620478A4 * |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2013161270A1 (ja) * | 2012-04-24 | 2015-12-21 | コニカミノルタ株式会社 | 活性光線硬化型インクジェットインク、及びこれを用いた画像形成方法 |
| US10100218B2 (en) | 2012-09-27 | 2018-10-16 | Sekisui Chemical Co., Ltd. | Curable composition for inkjet, and method for producing electronic part |
| CN104685010A (zh) * | 2012-09-27 | 2015-06-03 | 积水化学工业株式会社 | 喷墨用固化性组合物及电子零件的制造方法 |
| EP2902455A4 (en) * | 2012-09-27 | 2016-05-04 | Sekisui Chemical Co Ltd | CURABLE COMPOSITION FOR INK JET AND METHOD FOR MANUFACTURING ELECTRONIC PART |
| TWI567141B (zh) * | 2012-09-27 | 2017-01-21 | Sekisui Chemical Co Ltd | 噴墨用硬化性組合物及電子零件之製造方法 |
| US11220604B2 (en) * | 2013-11-06 | 2022-01-11 | Sekisui Chemical Co., Ltd. | Method for manufacturing cured film, method for manufacturing electronic component, and electronic component |
| US20160046813A1 (en) * | 2013-11-06 | 2016-02-18 | Sekisui Chemical Co., Ltd. | Method for manufacturing cured film, method for manufacturing electronic component, and electronic component |
| US20220098419A1 (en) * | 2013-11-06 | 2022-03-31 | Sekisui Chemical Co., Ltd. | Method for manufacturing cured film, method for manufacturing electronic component, and electronic component |
| JP2017066170A (ja) * | 2015-09-28 | 2017-04-06 | 積水化学工業株式会社 | 電子部品の製造方法 |
| WO2017169166A1 (ja) * | 2016-03-31 | 2017-10-05 | 太陽インキ製造株式会社 | インクジェット用硬化性組成物、硬化物およびプリント配線板 |
| US10907066B2 (en) | 2016-03-31 | 2021-02-02 | Taiyo Ink Mfg. Co., Ltd. | Curable composition for ink-jet printing, cured object, and printed wiring board |
| JP2019522689A (ja) * | 2016-05-17 | 2019-08-15 | マイクロクラフト コリア カンパニー リミテッド | インクジェット樹脂組成物の調製方法 |
| JP2019524900A (ja) * | 2017-06-08 | 2019-09-05 | マイクロクラフト コリア カンパニー リミテッド | インクジェット印刷用樹脂組成物およびそれを使用して調製されたプリント配線板 |
| US11390766B2 (en) | 2017-06-08 | 2022-07-19 | Microcraft Korea Co., Ltd. | Resin composition for inkjet printing and printed wiring board prepared by using same |
| JP2019189848A (ja) * | 2018-04-20 | 2019-10-31 | 積水化学工業株式会社 | 電子デバイス用光硬化性樹脂組成物 |
| JP7227016B2 (ja) | 2018-04-20 | 2023-02-21 | 積水化学工業株式会社 | 電子デバイス用光硬化性樹脂組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102753630A (zh) | 2012-10-24 |
| KR20130050270A (ko) | 2013-05-15 |
| TWI518105B (zh) | 2016-01-21 |
| EP2620478A1 (en) | 2013-07-31 |
| JP5416725B2 (ja) | 2014-02-12 |
| TW201213379A (en) | 2012-04-01 |
| US20130216726A1 (en) | 2013-08-22 |
| US10160881B2 (en) | 2018-12-25 |
| EP2620478B1 (en) | 2016-02-24 |
| EP2620478A4 (en) | 2014-03-26 |
| CN102753630B (zh) | 2015-04-08 |
| KR101485225B1 (ko) | 2015-01-23 |
| JP2012136681A (ja) | 2012-07-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5860096B2 (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| KR101485225B1 (ko) | 잉크젯용 경화성 조성물 및 전자 부품의 제조 방법 | |
| JP5727623B2 (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP5799144B2 (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP2012092312A (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP2012087298A (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP5735238B2 (ja) | インクジェット用硬化性組成物及びプリント配線板の製造方法 | |
| JP5940828B2 (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP6066558B2 (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP2012167268A (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP6066559B2 (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP2014055288A (ja) | インクジェット用硬化性組成物及び電子部品の製造方法 | |
| JP6039023B2 (ja) | インクジェット用硬化性組成物及びプリント配線板の製造方法 | |
| JP5879416B2 (ja) | インクジェット用硬化性組成物 | |
| JP2013213221A (ja) | インクジェット用硬化性組成物 | |
| JP2012153056A (ja) | インクジェット用硬化性組成物用硬化剤、インクジェット用硬化性組成物及びプリント配線板の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011539831 Country of ref document: JP Ref document number: 201180009526.0 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 20127001661 Country of ref document: KR Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11826817 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011826817 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13825535 Country of ref document: US |