WO2010143450A1 - π共役高分子組成物 - Google Patents
π共役高分子組成物 Download PDFInfo
- Publication number
- WO2010143450A1 WO2010143450A1 PCT/JP2010/003922 JP2010003922W WO2010143450A1 WO 2010143450 A1 WO2010143450 A1 WO 2010143450A1 JP 2010003922 W JP2010003922 W JP 2010003922W WO 2010143450 A1 WO2010143450 A1 WO 2010143450A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- acidic substance
- conjugated polymer
- salt
- group
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Oc1ccccc1 Chemical compound Oc1ccccc1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/02—Polyamines
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/02—Polyamines
- C08G73/026—Wholly aromatic polyamines
- C08G73/0266—Polyanilines or derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/13—Phenols; Phenolates
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/36—Sulfur-, selenium-, or tellurium-containing compounds
- C08K5/41—Compounds containing sulfur bound to oxygen
- C08K5/42—Sulfonic acids; Derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L65/00—Compositions of macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain; Compositions of derivatives of such polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B1/00—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors
- H01B1/06—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of other non-metallic substances
- H01B1/12—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of other non-metallic substances organic substances
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G9/00—Electrolytic capacitors, rectifiers, detectors, switching devices, light-sensitive or temperature-sensitive devices; Processes of their manufacture
- H01G9/004—Details
- H01G9/022—Electrolytes; Absorbents
- H01G9/025—Solid electrolytes
- H01G9/028—Organic semiconducting electrolytes, e.g. TCNQ
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/322—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed
- C08G2261/3221—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed containing one or more nitrogen atoms as the only heteroatom, e.g. pyrrole, pyridine or triazole
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/322—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed
- C08G2261/3223—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed containing one or more sulfur atoms as the only heteroatom, e.g. thiophene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/70—Post-treatment
- C08G2261/79—Post-treatment doping
- C08G2261/792—Post-treatment doping with low-molecular weight dopants
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/31504—Composite [nonstructural laminate]
- Y10T428/31855—Of addition polymer from unsaturated monomers
Definitions
- the present invention relates to a ⁇ -conjugated polymer composition.
- Conductive polymers are used in a wide range of industrial fields such as electrolytic capacitors, organic EL elements, antistatic films, and touch panels.
- polyaniline a kind of conductive polymer
- polyaniline composition described in Patent Document 1 is a dissolved polyaniline composition in which doped polyaniline is dissolved in a solvent.
- the conductive polyaniline composition disclosed in Patent Document 1 is not necessarily high in heat resistance. For example, when it is left in an inert gas at 105 ° C. for 10 days, the resistance value increases to about 10 times the initial value. There was a problem to do.
- dispersed polyaniline compositions in which doped polyaniline is not dissolved are also known. Furthermore, it is also known that heat resistance is improved by using sulfonic acid as a dopant (Patent Document 2).
- Patent Document 2 the dispersion type polyaniline composition has a drawback that the obtained molded body is brittle when the solid content is only the polyaniline fine particles.
- a binder resin that is a solid content other than polyaniline In order to solve this problem, it is also known to add a binder resin that is a solid content other than polyaniline. However, since the binder resin is an insulator, the conductivity of the obtained molded product is reduced. There are drawbacks.
- the doped polyaniline fine particles aggregate and precipitate. Therefore, when producing a molded body using this dispersion type polyaniline composition, the composition is mixed at the time of production and the polyaniline fine particles are mixed. Has a disadvantage that it must be dispersed in the composition.
- Patent Document 3 It is also known that heat resistance is improved by adding sulfonic acid to a dispersed polyaniline composition.
- Patent Document 3 since the invention described in Patent Document 3 is also a dispersion type, it has the same drawbacks as described above.
- a molded article produced using “conductive composition in which sulfonic acid is added to dispersed polyaniline” described in Patent Document 3 “conductive composition in which sulfonic acid is not added to dispersed polyaniline” As compared with a molded body produced using a material, there is a drawback that the conductivity is greatly reduced.
- An object of the present invention is to provide a soluble polymer composition having high conductivity and high heat resistance and having a ⁇ -conjugated polymer dissolved in a solvent.
- the first ⁇ -conjugated polymer composition of the present invention is (A) solvent, (B) a ⁇ -conjugated polymer doped with a dopant dissolved in the solvent; (C) a ⁇ -conjugated polymer composition comprising at least one of an acidic substance and a salt of the acidic substance, and (d) a phenolic compound, Of the component (c), when containing only the acidic substance, the acidic substance and the phenolic compound are different, and when containing only the salt of the acidic substance, the salt of the acidic substance and the phenolic compound are different, When both the acidic substance and the salt of the acidic substance are included, the phenolic compound is different from at least one of the acidic substance and the salt of the acidic substance.
- the second ⁇ -conjugated polymer composition of the present invention is At least the following (a) to (d) are used as raw materials.
- (c) at least one of an acidic substance and a salt of the acidic substance (d) a phenolic compound (of the component (c))
- the acidic substance and the phenolic compound are different, and in the case of containing only the salt of the acidic substance, the salt of the acidic substance and the phenolic compound are different, and the acidic substance and the acidic substance In the case where both of the acidic substance and the salt of the acidic substance are different from the phenolic compound.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are: Among the components (c), when only the acidic substance is included, the acidic substance is different from the dopant, and when only the acidic substance salt is included, the acidic substance salt is different from the dopant, and the acidic substance and When both of the acidic substance salts are included, the dopant is preferably different from at least one of the acidic substance and the salt of the acidic substance.
- the acidity of the acidic substance is 5.0 or less
- the acidity is 5.0 or less
- both the salt of the acidic substance and the salt of the acidic substance are included, the acidic substance It is preferable that at least one condition is satisfied among the acidity of the substance being 5.0 or less and the salt of the acid substance being a salt of an acid substance having an acidity of 5.0 or less.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the ⁇ -conjugated polymer contains a nitrogen atom,
- the dopant is a sulfonic acid;
- the acidic substance is the same or different sulfonic acid as the dopant,
- the salt of the acidic substance is the same or different sulfonic acid salt as the dopant,
- at least one of the acidic substance and the salt of the acidic substance is a sulfonic acid or a sulfonic acid salt that is the same as or different from the dopant, It is preferable to satisfy the formula (1). 0.21 ⁇ S 1 / N 1 ⁇ 1.2 (1) (In the formula, S 1 is the number of moles of sulfur atoms contained in the composition, and N 1 is the number
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the ⁇ -conjugated polymer contains a nitrogen atom,
- the dopant is a sulfonic acid;
- the acidic substance is the same or different sulfonic acid as the dopant,
- the salt of the acidic substance is the same or different sulfonic acid salt as the dopant,
- at least one of the acidic substance and the salt of the acidic substance is a sulfonic acid or a sulfonic acid salt that is the same as or different from the dopant,
- the formula (2) is satisfied
- the formula (3) is satisfied,
- the acidic substance and the salt of the acidic substance are included, the formula (1)
- S 2 is the sum of the number of moles of sulfur atoms in all acidic substances contained in the composition
- S 3 is the number of moles of sulfur atoms in the salts of all the acidic substances contained in the composition
- S 4 is the sum of moles of sulfur atoms of all acidic substances and salts of acidic substances contained in the composition
- N 2 to N 4 are doped with all dopants contained in the composition. The total number of moles of nitrogen atoms in the ⁇ -conjugated polymer produced.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are: It is preferable that the ⁇ -conjugated polymer contains a nitrogen atom, the dopant is sulfonic acid, and satisfies the formula (5). 0.2 ⁇ S 5 / N 5 ⁇ 0.7 (5) (S 5 is the total number of moles of sulfur atoms of the ⁇ -conjugated polymer doped with the dopant contained in the composition, and N 5 is nitrogen of the ⁇ -conjugated polymer doped with the dopant contained in the composition. (The total number of moles of atoms.)
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the conductivity measured by the four-terminal method of the molded product of ⁇ -conjugated polymer doped with the dopant is 0.01 S / cm or more
- the molded body was prepared by adding 1 ml of a solution obtained by dissolving 500 mg of a ⁇ -conjugated polymer doped with the dopant in 10 g of toluene in a nitrogen atmosphere on a glass substrate on which a patterned indium tin oxide electrode was formed, at 500 rpm for 15 seconds. It is preferably obtained by applying by spin coating and drying at 80 ° C. for 5 minutes in a nitrogen atmosphere.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the conductivity measured by the four-terminal method of the molded body is 0.01 S / cm or more
- the molded body was coated on a glass substrate on which a patterned indium tin oxide electrode was formed, by applying 1 ml of the ⁇ -conjugated polymer composition at 500 rpm for 15 seconds in a nitrogen atmosphere, and at 80 ° C. in a nitrogen atmosphere. It is preferably obtained by drying for 5 minutes.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the doped ⁇ -conjugated polymer is preferably a protonated substituted or unsubstituted polyaniline.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the doped ⁇ -conjugated polymer is a substituted or unsubstituted polyaniline that is protonated with sulfonic acid.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the sulfonic acid is preferably a sulfosuccinic acid derivative represented by the following formula (III).
- M is a hydrogen atom, an organic radical or an inorganic radical, m is the valence of M;
- R 12 and R 13 are each independently a hydrocarbon group or a group represented by — (R 14 O) r—R 15 , R 14 is a hydrocarbon group or a silylene group, and R 15 is a hydrogen atom ,
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the acidic substance is preferably an organic acid.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the acidic substance or the salt of the acidic substance is one or more acidic substances or salts of acidic substances having one or more acidic groups selected from a sulfonic acid group, a phosphoric acid group, a phosphonic acid group, and a carboxy group. It is preferable.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the acidic substance is preferably a cyclic, chain or branched alkyl acid having one or more acidic groups.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the acidic substance is preferably a substituted or unsubstituted aromatic acid having one or more acidic groups.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are: It is preferable that the aromatic acid is an acid having a naphthalene skeleton.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the phenolic compound is preferably a phenolic compound represented by the following formula (1).
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the phenolic compound is preferably a phenolic compound represented by the following formula (2).
- R is an alkyl group having 2 to 10 carbon atoms, an alkenyl group, an alkylthio group, a cycloalkyl group having 3 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, an alkylaryl group, or an arylalkyl group.
- the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention are:
- the phenolic compound is preferably a phenolic compound represented by the following formula (3).
- n is an integer of 1 to 5.
- R is an alkyl group having 2 to 10 carbon atoms, an alkenyl group, an alkylthio group, a cycloalkyl group having 3 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, an alkylaryl group, or an arylalkyl group.
- the first capacitor of the present invention includes the first ⁇ -conjugated polymer composition of the present invention or the second ⁇ -conjugated polymer composition of the present invention.
- the second capacitor of the present invention is produced using the first ⁇ -conjugated polymer composition of the present invention or the second ⁇ -conjugated polymer composition of the present invention.
- the conductive molded body of the present invention is formed by molding the first ⁇ -conjugated polymer composition of the present invention or the second ⁇ -conjugated polymer composition of the present invention.
- the conductive film of the present invention is formed by depositing the first ⁇ -conjugated polymer composition of the present invention or the second ⁇ -conjugated polymer composition of the present invention.
- the surface conductive article of the present invention is obtained by applying the first ⁇ -conjugated polymer composition of the present invention or the second ⁇ -conjugated polymer composition of the present invention to a substrate.
- the substrate is preferably a resin film.
- the conductive article of the present invention is formed by mixing the first ⁇ -conjugated polymer composition of the present invention or the second ⁇ -conjugated polymer composition of the present invention and a substrate.
- the third ⁇ -conjugated polymer composition of the present invention comprises a doped ⁇ -conjugated polymer and an acidic substance or a salt of an acidic substance,
- the doped ⁇ -conjugated polymer is a substituted or unsubstituted polyaniline protonated with a sulfosuccinic acid derivative;
- the acidic substance is an acid having a sulfonic acid group and a naphthalene skeleton, and further includes a phenolic compound.
- a soluble polymer composition having high conductivity and high heat resistance and having a ⁇ -conjugated polymer dissolved in a solvent can be provided.
- the first ⁇ -conjugated polymer composition of the present invention comprises (a) a solvent, (b) a ⁇ -conjugated polymer dissolved in the solvent, (c) at least one of an acidic substance and a salt of an acidic substance, and (d ) Contains phenolic compounds.
- a ⁇ -conjugated polymer is a ⁇ -conjugated polymer doped with a dopant. Details will be described later.
- the first ⁇ -conjugated polymer composition of the present invention is obtained by mixing, for example, a solvent, a ⁇ -conjugated polymer dissolved in the solvent, at least one of an acidic substance and a salt of the acidic substance, and a phenolic compound. be able to.
- the second ⁇ -conjugated polymer composition of the present invention comprises (a) a solvent, (b) a ⁇ -conjugated polymer dissolved in the solvent, (c) at least one of an acidic substance and an acidic substance, and (d) It is a ⁇ -conjugated polymer composition produced by using a phenolic compound as a raw material.
- the acidic substance and the phenolic compound are different, and when only the acidic substance salt is included, the acidic substance salt and the phenolic compound are different, and the acidic substance and the acidic substance salt When both are included, at least one of the acidic substance and the salt of the acidic substance is different from the phenolic compound.
- the second ⁇ -conjugated polymer composition of the present invention comprises, for example, a mixture of a solvent, a ⁇ -conjugated polymer dissolved in the solvent, at least one of an acidic substance and a salt of the acidic substance, and a phenolic compound.
- the composition of the present invention is a concept including both the first ⁇ -conjugated polymer composition of the present invention and the second ⁇ -conjugated polymer composition of the present invention.
- the acidic substance is a dopant and in contrast, when only an acidic substance salt is included, the acidic substance salt is different from a dopant, and when both an acidic substance and an acidic substance salt are included, at least one of the acidic substance and the acidic substance salt is It is preferred that the dopants are different.
- the acidic substance of the acidic substance When the first ⁇ -conjugated polymer composition of the present invention and / or the second ⁇ -conjugated polymer composition of the present invention contains only the acidic substance among the components (c), the acidic substance of the acidic substance
- the acidity is 5.0 or less and includes only the salt of the acidic substance
- the acidity is 5.0 or less and the acid substance salt includes both the acid substance and the acid substance salt. It is preferable that at least one condition is satisfied among the acidic substance salts having an acidity of 5.0 or less and an acidity of 5.0 or less.
- the ⁇ -conjugated polymer contains a nitrogen atom
- the dopant is a sulfonic acid
- the salt of the acidic substance and the acidic substance is a sulfonic acid or a sulfonic acid salt that is the same as or different from the dopant and satisfies the following formula (1): preferable. 0.21 ⁇ S 1 / N 1 ⁇ 1.2 ⁇ (1) (Here, S 1 is the number of moles of sulfur atoms contained in the composition, and N 1 is the number of moles of nitrogen atoms contained in the composition.)
- the ⁇ -conjugated polymer preferably contains a nitrogen atom, the dopant is sulfonic acid, Among c), when only an acidic substance is included, the acidic substance is the same or different sulfonic acid as the dopant, and when only an acidic substance salt is included, the acidic substance salt is the same or different from the dopant.
- the acidic substance and the salt of the acidic substance are the same or different sulfonic acid or sulfonic acid salt as the dopant.
- the formula (2) is satisfied when only the acidic substance is included, the formula (3) is satisfied when only the salt of the acidic substance is included, and the formula (3) is included when the salt of the acidic substance and the acidic substance is included. 4) is satisfied. 0.01 ⁇ S 2 / N 2 ⁇ 0.5 (2) 0.01 ⁇ S 3 / N 3 ⁇ 0.5 (3) 0.01 ⁇ S 4 / N 4 ⁇ 0.5 (4)
- S 2 is the total number of moles of sulfur atoms of all acidic substances contained in the composition
- N 2 is a ⁇ -conjugated polymer doped with all the dopants contained in the composition.
- S 3 is the total number of moles of sulfur atoms in the salts of all acidic substances contained in the composition
- N 3 is the total number of moles in the composition.
- S 4 is the total number of moles of sulfur atoms of all acidic substances and salts of acidic substances contained in the composition.
- N 4 means the total number of moles of nitrogen atoms of the ⁇ -conjugated polymer doped with all dopants contained in the composition.
- the first ⁇ -conjugated polymer composition of the present invention and / or the second ⁇ -conjugated polymer composition of the present invention is a ⁇ -conjugated polymer in which the ⁇ -conjugated polymer contains a nitrogen atom, and the dopant is sulfonic acid. Yes, preferably according to formula (5).
- S 5 is the total number of moles of sulfur atoms in the ⁇ -conjugated polymer which is doped by the dopant contained in the composition
- N 5 is ⁇ -conjugated polymer which is doped by the dopant contained in the composition
- the total number of moles of nitrogen atoms The number of moles of nitrogen and sulfur atoms is a value measured by an organic elemental analysis method.
- the ⁇ -conjugated polymer preferably has a weight average molecular weight of 1,000 or more, more preferably 1,000 to 1,000,000.
- the ⁇ -conjugated polymer is a polymer having a ⁇ -conjugated main chain having a structure in which double bonds and single bonds are alternately arranged.
- Specific examples of the ⁇ -conjugated polymer include substituted or unsubstituted polyaniline, polypyrrole, polythiophene, poly (p-phenylene), poly (p-phenylene vinylene), and derivatives thereof. From the viewpoint of properties, it is preferably a substituted or unsubstituted polyaniline and / or polyaniline derivative.
- the weight average molecular weight of polyaniline is preferably 20,000 or more, more preferably 1,000,000. If the weight molecular weight of polyaniline is less than 20,000, the strength and stretchability of the conductive article obtained from the composition may be reduced.
- the molecular weight distribution is, for example, 1.5 to 10.0. From the viewpoint of electrical conductivity, a smaller molecular weight distribution is preferable, but from the viewpoint of solubility in a solvent and moldability, a wider molecular weight distribution may be preferable.
- the molecular weight and molecular weight distribution can be measured by gel permeation chromatograph (GPC).
- the doped ⁇ -conjugated polymer of the composition of the present invention is dissolved in a solvent.
- dissolved here means that the ⁇ -conjugated polymer is dissolved in a solvent uniformly in molecular units. Thereby, when the solvent is dried, a uniform ⁇ -conjugated polymer film having no clear grain boundary can be obtained.
- the doped ⁇ -conjugated polymer is doped with a dopant.
- the fact that the ⁇ -conjugated polymer is doped with the dopant means that a charge transfer body (carrier) that can freely move into the ⁇ -conjugated polymer is injected by the dopant.
- the doping ratio a of the doped ⁇ -conjugated polymer is preferably 0 ⁇ a ⁇ 1, more preferably 0.7 or less, further preferably 0.2 ⁇ a ⁇ 0.7, and most preferably Is 0.4 ⁇ a ⁇ 0.7.
- the doping rate is generally defined by (number of moles of dopant molecules doped in the conductive polymer) / (monomer unit of the conductive polymer).
- the doped ⁇ -conjugated polymer is a polyaniline complex, preferably 0 ⁇ a ⁇ 1, more preferably 0.7 or less, and further preferably 0.2 ⁇ a ⁇ 0.7. Most preferably, 0.4 ⁇ a ⁇ 0.7.
- a doping rate is 0.2 or less.
- the doping rate is 0.7 or more, the conductivity is lowered.
- a dopant doping rate a of 0.5 means that one molecule of dopant is doped with respect to two nitrogen atoms.
- the conductivity of the molded product of ⁇ -conjugated polymer doped with the dopant is 0.01 S / cm or more. Conductivity is measured by the 4-terminal method.
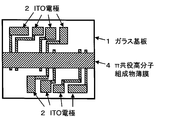
- a molded object is obtained as follows. “500 mg of ⁇ -conjugated polymer doped with dopant” is dissolved in 10 g of toluene to prepare a solution for measuring conductivity. As shown in FIG. 1, 1 ml of a conductivity measuring solution is applied to the upper surface of a glass substrate 1 on which an indium tin oxide (ITO) electrode 2 is formed by patterning. Specifically, it is applied by spin coating. Here, the application by spin coating is performed in a nitrogen atmosphere. In addition, the rotation time of the glass substrate after dropping the conductivity measuring solution on the glass substrate by spin coating is 15 seconds. Moreover, the glass substrate rotation speed of the spin coating method is 500 rpm.
- ITO indium tin oxide
- the glass substrate is dried to form a ⁇ -conjugated polymer thin film.
- the drying is performed in a nitrogen atmosphere.
- the drying time is 5 minutes.
- the drying temperature is 80 ° C.
- the molded product refers to a molded product of ⁇ -conjugated polymer formed on a glass substrate itself.
- electrical conductivity is obtained as follows, for example. After drying the ⁇ -conjugated polymer thin film, as shown in FIG. 2, the portion of the ⁇ -conjugated polymer thin film 3 covering the terminal of the ITO electrode is scraped off in a nitrogen atmosphere to expose the terminal of the ITO electrode on the surface. Using the ITO electrode terminal exposed on the surface, the conductivity is measured by a 4-terminal method using a resistivity meter manufactured by Mitsubishi Chemical Corporation.
- the conductivity of the molded product of the ⁇ -conjugated polymer composition is preferably 0.01 s / cm or more. Conductivity is measured by the 4-terminal method.
- a molded object is obtained as follows. 1 ml of ⁇ -conjugated polymer composition is applied to the upper surface of the glass substrate 1 on which the ITO electrode 2 is formed by patterning as shown in FIG. Specifically, it is applied by spin coating. Here, the application by spin coating is performed in a nitrogen atmosphere. Further, the rotation time of the glass substrate after the ⁇ -conjugated polymer composition is dropped onto the glass substrate by spin coating is 15 seconds. Moreover, the glass substrate rotation speed of the spin coating method is 500 rpm.
- the glass substrate is dried to form a ⁇ -conjugated polymer composition thin film.
- the drying is performed in a nitrogen atmosphere.
- the drying time is 5 minutes.
- the drying temperature is 80 ° C.
- the molded product refers to a molded product itself of a ⁇ -conjugated polymer composition formed on a glass substrate.
- electrical conductivity is obtained as follows, for example. After drying the ⁇ -conjugated polymer composition thin film, as shown in FIG. 3, the portion of the ⁇ -conjugated polymer composition thin film 4 covering the terminal of the ITO electrode is scraped off in a nitrogen atmosphere, and the terminal of the ITO electrode is surfaced. To expose. Using the ITO electrode terminal exposed on the surface, the conductivity is measured by a 4-terminal method using a resistivity meter manufactured by Mitsubishi Chemical Corporation.
- the ⁇ -conjugated polymer doped with a dopant is preferably a substituted or unsubstituted polyaniline that is protonated.
- the dopant include an organic proton acid or a salt thereof.
- the substituent of the substituted polyaniline include linear or branched hydrocarbon groups such as methyl group, ethyl group, hexyl group and octyl group; alkoxyl groups such as methoxy group and phenoxy group; aryloxy group; CF 3 group and the like And halogen-containing hydrocarbon groups.
- the protonated substituted or unsubstituted polyaniline is preferably a substituted or unsubstituted polyaniline that is protonated by an organic protonic acid represented by the following formula (I) or a salt thereof.
- M is a hydrogen atom, an organic free radical or an inorganic free radical.
- organic free radical include a pyridinium group, an imidazolium group, and an anilinium group.
- the inorganic free radical include sodium, lithium, potassium, cesium, ammonium and the like.
- X is an acidic group, for example, a group represented by —SO 3 ⁇ , —PO 3 2 ⁇ , —PO 4 (OH) ⁇ , —OPO 3 2 ⁇ , —OPO 2 (OH) ⁇ , —COO ⁇
- the group represented by —SO 3 — is preferable.
- A is a hydrocarbon group which may contain a substituent.
- the hydrocarbon group include a linear or branched alkyl group having 1 to 24 carbon atoms; an alkenyl group; a cycloalkyl group which may contain a substituent such as cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, menthyl and the like.
- a dicycloalkyl group or polycycloalkyl group that may be condensed such as bicyclohexyl, norbornyl, adamantyl, or the like; an aryl containing an aromatic ring that may contain a substituent such as phenyl, tosyl, thiophenyl, pyrrolinyl, pyridinyl, furanyl, etc.
- a diaryl group or polyaryl group which may be condensed such as naphthyl, anthracenyl, fluorenyl, 1,2,3,4-tetrahydronaphthyl, indanyl, quinolinyl, indonyl, etc .; an alkylaryl group and the like (n + 1 ) The value group is listed It is.
- R 1 is independently a substituent represented by —R 1 , —OR 1 , —COR 1 , —COOR 1 , —CO (COR 1 ), or —CO (COOR 1 ).
- R 1 is a hydrocarbon group that may contain a substituent having 4 or more carbon atoms, a silyl group, an alkylsilyl group, a group represented by — (R 2 O) x —R 3 , or — (OSiR 3 2 ) x- OR 3
- R 2 is an alkylene group
- R 3 is a hydrocarbon group which may be the same or different
- x is an integer of 1 or more).
- hydrocarbon group for R 1 examples include a linear or branched butyl group, pentyl group, hexyl group, heptyl group, octyl group, nonyl group, decyl group, dodecyl group, pentadecyl group, eicosanyl group and the like.
- N is an integer of 2 or more.
- m is the valence of M.
- the organic protonic acid represented by the formula (I) or a salt thereof is preferably a dialkylbenzenesulfonic acid, a dialkylnaphthalenesulfonic acid, a sulfophthalic acid ester, or an organic protonic acid represented by the following formula (II) or a salt thereof. . M (XCR 4 (CR 5 2 COOR 6) COOR 7) p (II)
- R 4 and R 5 are each independently a hydrogen atom, a hydrocarbon group or a group represented by R 8 3 Si— (wherein R 8 is a hydrocarbon group, and three R 8 are the same or different May be).
- Examples of the hydrocarbon group for R 4 and R 5 include a linear or branched alkyl group having 1 to 24 carbon atoms; an aryl group containing an aromatic ring; an alkylaryl group.
- the hydrocarbon group for R 8 is the same as the hydrocarbon group for R 4 and R 5 .
- R 6 and R 7 are each independently a hydrocarbon group or a group represented by — (R 9 O) q —R 10 [wherein R 9 is a hydrocarbon group or a silylene group, and R 10 is hydrogen An atom, a hydrocarbon group, or a group represented by R 11 3 Si— (R 11 is a hydrocarbon group, and three R 11 may be the same or different), and q is an integer of 1 or more There is].
- the hydrocarbon group for R 6 and R 7 include a linear or branched alkyl group having 1 to 24 carbon atoms, preferably 4 or more carbon atoms; an aryl group containing an aromatic ring; an alkylaryl group, and the like.
- Specific examples of the hydrocarbon group of R 6 and R 7 include a linear or branched butyl group, pentyl group, hexyl group, octyl group, decyl group and the like.
- hydrocarbon group for R 9 examples include a linear or branched alkylene group having 1 to 24 carbon atoms; an arylene group containing an aromatic ring; an alkylarylene group; an arylalkylene group.
- the hydrocarbon groups for R 10 and R 11 are the same as those for R 4 and R 5 .
- q is preferably an integer of 1 to 10.
- organic protonic acid represented by the formula (II) or a salt thereof when R 6 and R 7 are a group represented by — (R 9 O) n —R 10 are represented by the following formulae: Acid.
- X is a group represented by —SO 3 , etc.
- the compound represented by the following formula (II) is preferably a sulfosuccinic acid derivative represented by the following formula (III) (hereinafter sometimes simply referred to as a sulfosuccinic acid derivative (III)).
- a sulfosuccinic acid derivative represented by the following formula (III) (hereinafter sometimes simply referred to as a sulfosuccinic acid derivative (III)).
- R 12 and R 13 are each independently a hydrocarbon group or a group represented by — (R 14 O) r —R 15 [wherein R 14 is a hydrocarbon group or a silylene group, and R 15 is a hydrogen group.
- R 14 is a hydrocarbon group or a silylene group
- R 15 is a hydrogen group.
- the hydrocarbon groups for R 12 and R 13 are the same as the hydrocarbon groups for R 6 and R 7 .
- the hydrocarbon group for R 14 is the same as the hydrocarbon group for R 9 .
- the hydrocarbon groups for R 15 and R 16 are the same as the hydrocarbon groups for R 4 and R 5 .
- r is preferably an integer of 1 to 10.
- R 12 and R 13 are a group represented by — (R 14 O) r —R 15
- Specific examples of the organic protonic acid represented by the formula (I) or a salt thereof include an organic proton represented by the formula (II) in the case where R 6 and R 7 are a group represented by — (R 9 O) n —R 10.
- the hydrocarbon group for R 12 and R 13 is the same as the hydrocarbon group for R 6 and R 7 , and is preferably a butyl group, a hexyl group, a 2-ethylhexyl group, a decyl group, or the like.
- the acidic substance may be either an organic acid that is an acid of an organic compound or an inorganic acid that is an acid of an inorganic compound, and is preferably an organic acid.
- the acidic substance contained in the composition of the present invention is preferably an organic acid containing one or more acidic groups which are acidic groups such as sulfonic acid group, phosphoric acid group, phosphonic acid group and carboxy group.
- the organic acid having a sulfonic acid group is preferably a cyclic, linear or branched alkyl sulfonic acid, substituted or unsubstituted aromatic sulfonic acid, or polysulfonic acid having one or more sulfonic acid groups.
- alkylsulfonic acid include methanesulfonic acid, ethanesulfonic acid, and di-2-ethylhexylsulfosuccinic acid.
- the aromatic sulfonic acid include sulfonic acid having a benzene ring, sulfonic acid having a naphthalene skeleton, and sulfonic acid having an anthracene skeleton.
- the aromatic sulfonic acid examples include substituted or unsubstituted benzene sulfonic acid, naphthalene sulfonic acid, and anthracene sulfonic acid.
- the substituent is, for example, a substituent selected from the group consisting of an alkyl group, an alkoxy group, a hydroxy group, a nitro group, a carboxy group, and an acyl group, and one or more substituents may be substituted.
- naphthalene sulfonic acid, dodecylbenzene sulfonic acid, and anthraquinone sulfonic acid can be mentioned.
- the aromatic sulfonic acid substituted or unsubstituted naphthalenesulfonic acid is preferable.
- the polysulfonic acid is a sulfonic acid having a plurality of sulfonic acid groups substituted on the main chain or side chain of the polymer chain.
- polystyrene sulfonic acid is mentioned.
- the alkyl group is preferably a linear or branched alkyl group having 1 to 18 carbon atoms.
- the organic acid having a carboxy group is preferably a cyclic, linear or branched alkyl carboxylic acid having one or more carboxy groups, or a substituted or unsubstituted aromatic carboxylic acid.
- alkyl carboxylic acid include undecylenic acid, cyclohexane carboxylic acid, 2-ethylhexanoic acid and the like.
- aromatic carboxylic acid include substituted or unsubstituted benzene carboxylic acid and naphthalene carboxylic acid.
- the substituent is, for example, a substituent selected from the group consisting of a sulfonic acid group, an alkyl group, an alkoxy group, a hydroxy group, a nitro group, and an acyl group, and one or more substituents may be substituted.
- a substituent selected from the group consisting of a sulfonic acid group, an alkyl group, an alkoxy group, a hydroxy group, a nitro group, and an acyl group and one or more substituents may be substituted.
- a substituent selected from the group consisting of a sulfonic acid group, an alkyl group, an alkoxy group, a hydroxy group, a nitro group, and an acyl group and one or more substituents may be substituted.
- salicylic acid, benzoic acid, naphthoic acid, trimesic acid and the like can be mentioned.
- the alkyl group is preferably a linear or branched alkyl group
- the organic acid having a phosphoric acid group or phosphonic acid group is preferably a cyclic, linear or branched alkyl phosphoric acid or phosphonic acid having one or more phosphoric acid groups or phosphonic acid groups, substituted or unsubstituted aromatic phosphorus. Acid or phosphonic acid.
- alkyl phosphoric acid or phosphonic acid include dodecyl phosphoric acid, bis (2-ethylhexyl) hydrogen phosphate, and the like.
- the aromatic phosphoric acid or phosphonic acid include substituted or unsubstituted benzenesulfonic acid or phosphonic acid, and naphthalenesulfonic acid or phosphonic acid.
- the substituent is, for example, a substituent selected from the group consisting of an alkyl group, an alkoxy group, a hydroxy group, a nitro group, a carboxy group, and an acyl group, and one or more substituents may be substituted.
- substituents for example, phenylphosphonic acid etc. are mentioned.
- the alkyl group is preferably a linear or branched alkyl group having 1 to 18 carbon atoms.
- the salt of the acidic substance contained in the composition of the present invention examples include the salt of the acidic substance.
- the composition of the present invention may contain one or more of the acidic substances or salts thereof.
- the composition of the present invention may contain a plurality of different acidic substances.
- the composition of the present invention may contain a plurality of different acidic substances.
- the composition of the present invention may contain a plurality of different acidic substances and salts of acidic substances.
- the acid substance preferably has an acidity (pKa) of 5.0 or less.
- the lower limit of the acidity (pKa) is not particularly limited. For example, when an acidic substance having an acidity of ⁇ 4.0 or less is added, the ⁇ -conjugated polymer may be deteriorated.
- Acidity is defined by computational chemistry methods.
- the method described in 2224 is used. Specifically, using “TURBOMOLE Version 6.1” (manufactured by COSMO logic), the structure is optimized using TZVP as a basis function, and the COSMO-RS method calculation is performed using this structure using “COSMO therm version C2”. .1 Release 01.10 "(manufactured by COSMO logic).
- the pKa is calculated by inputting the conditions in an aqueous solvent at 25 ° C., the chemical formula of the molecule, and the chemical formula of the deprotonated molecule into “COSMO thermion C2.1 Release 01.10”.
- the content of at least one of the acidic substance and the salt of the acidic substance is preferably 1 to 1000 parts by mass, more preferably 10 to 1000 parts by mass with respect to 100 parts by mass of the doped ⁇ -conjugated polymer. 100 parts by mass.
- the solvent contained in the composition of the present invention is preferably an organic solvent.
- the solvent may be an organic solvent that is substantially immiscible with water (a water-immiscible organic solvent) or a water-soluble organic solvent.
- water-immiscible organic solvents include hydrocarbon solvents such as benzene, toluene, xylene, ethylbenzene, and tetralin; halogen-containing solvents such as methylene chloride, chloroform, carbon tetrachloride, dichloroethane, and tetrachloroethane; ethyl acetate and the like And ester solvents.
- hydrocarbon solvents such as benzene, toluene, xylene, ethylbenzene, and tetralin
- halogen-containing solvents such as methylene chloride, chloroform, carbon tetrachloride, dichloroethane, and tetrachloroethane
- ethyl acetate and the like
- ester solvents ethyl acetate and the like
- water-soluble organic solvent examples include alcohols; ketones such as acetone and methyl ethyl ketone; polar ethers such as tetrahydrofuran and dioxane; and aprotic polar solvents such as N-methylpyrrolidone.
- the doped polyaniline and the phenolic compound described below contained in the composition of the present invention are dissolved in alcohols such as 2-butanol, 2-pentanol, and benzyl alcohol. Alcohol is preferable from the viewpoint of reducing environmental burden unlike toluene.
- a mixed organic solvent of a water-immiscible organic solvent and a water-soluble organic solvent is used in a mass ratio of 99 to 50: 1 to 50 when the composition of the present invention is stored. From the viewpoint of long-term storage, it is possible to prevent generation of gels and the like.
- a low polarity organic solvent can be used as the water-immiscible organic solvent of the mixed organic solvent.
- toluene and chloroform are preferable.
- a water-soluble organic solvent of a mixed organic solvent a highly polar organic solvent can be used.
- methanol, ethanol, isopropyl alcohol, 2-methoxyethanol, 2-ethoxyethanol, acetone, methyl ethyl ketone, methyl isobutyl ketone, tetrahydrofuran or diethyl ether are preferable.
- the proportion of the doped ⁇ -conjugated polymer in the organic solvent is usually 900 g / L or less, preferably 0.01 to 300 g / L or less, depending on the type of the organic solvent. If the content of the doped ⁇ -conjugated polymer is too large, the solution state cannot be maintained, the handling of the molded product becomes difficult, the uniformity of the molded product is impaired, and consequently the electrical properties of the molded product. And mechanical strength and transparency are reduced. On the other hand, if the content of the doped ⁇ -conjugated polymer is too small, only a very thin film can be produced when the film is formed by the method described later, which may make it difficult to produce a uniform conductive film.
- the composition of the present invention further comprises a phenolic compound.
- the phenolic compound is different from the acidic substance or the salt of the acidic substance.
- the phenolic compound contained in the composition of the present invention is not particularly limited, and is a compound represented by ArOH (where Ar is an aryl group or a substituted aryl group).
- Substituted phenols such as-, m- or p-chlorophenol, salicylic acid, hydroxybenzoic acid and hydroxynaphthalene; polyhydric phenolic compounds such as catechol and resorcinol; and polymers such as phenolic resins, polyphenols and poly (hydroxystyrene) A compound etc. can be illustrated.
- the molar concentration of 1 g of ⁇ -conjugated polymer doped with a phenolic compound is preferably in the range of 1 mmol / g to 50 mmol / g.
- the range is preferably 2 mmol / g to 20 mmol / g.
- the composition of the present invention containing a phenolic compound is preferably a phenolic compound represented by the following formula (1), more preferably a phenolic compound represented by formula (1) and a dope.
- the weight ratio of the ⁇ -conjugated polymer is 0.01 to 10.0.
- R is an alkyl group, alkenyl group, cycloalkyl group, alkylthio group, aryl group, alkylaryl group or arylalkyl group having 1 to 20 carbon atoms.
- the —OR substitution position is preferably a meta position or a para position with respect to the phenolic hydroxyl group.
- a phenolic compound represented by the following formula (2) can be used instead of the phenolic compound represented by the above formula (1).
- the composition of the present invention containing a phenolic compound is preferably a phenolic compound represented by the following formula (2), more preferably a phenolic compound represented by formula (2) and a dope.
- the weight ratio of the ⁇ -conjugated polymer is 0.01 to 5.0.
- R is an alkyl group having 2 to 10 carbon atoms, an alkenyl group, an alkylthio group, a cycloalkyl group having 3 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, an alkylaryl group, or an arylalkyl group.
- the phenolic compound represented by Formula (3) can be used instead of the phenolic compound represented by the said Formula (1).
- the composition of the present invention containing a phenolic compound is preferably a phenolic compound represented by the following formula (3), more preferably a phenolic compound represented by formula (3) and a dope.
- the weight ratio of the ⁇ -conjugated polymer is 0.01 to 10.0.
- R is an alkyl group having 2 to 10 carbon atoms, an alkenyl group, an alkylthio group, a cycloalkyl group having 3 to 10 carbon atoms, an aryl group having 6 to 20 carbon atoms, an alkylaryl group, or an arylalkyl group.
- Examples of the alkyl group include methyl, ethyl, propyl, isopropyl, butyl, isobutyl, and tertiary butyl.
- Examples of the alkenyl group include a substituent having an unsaturated bond in the molecule of the alkyl group described above.
- Examples of the cycloalkyl group include cyclopentane and cyclohexane.
- Examples of the alkylthio group include methylthio and ethylthio.
- Examples of the aryl group include phenyl and naphthyl.
- Examples of the alkylaryl group and the arylalkyl group include a substituent obtained by combining the above-described alkyl group and aryl group. Of these groups, R is preferably a methyl or ethyl group.
- the composition of the present invention comprises, for example, 15% by weight or more, 50% by weight or more, 70% by weight or more, and 100% by weight comprising a ⁇ -conjugated polymer, an acidic substance and / or a salt of an acidic substance, a solvent, and a phenolic compound. Also good.
- the composition of the present invention may contain other resins, inorganic materials, curing agents, plasticizers and the like as long as the effects of the present invention are not impaired.
- resins are added as, for example, a binder base material, a plasticizer, a matrix base material, etc., and specific examples thereof include, for example, polyolefins such as polyethylene and polypropylene, chlorinated polyolefins, polystyrenes, polyesters, polyamides, polyacetals, polyethylenes. Examples include terephthalate, polycarbonate, polyethylene glycol, polyethylene oxide, polyacrylic acid, polyacrylic acid ester, polymethacrylic acid ester, and polyvinyl alcohol. Moreover, you may use the precursor which can form thermosetting resins, such as an epoxy resin, a urethane resin, a phenol resin, with resin instead of resin.
- thermosetting resins such as an epoxy resin, a urethane resin, a phenol resin, with resin instead of resin.
- Inorganic materials are added for the purpose of, for example, improving strength, surface hardness, dimensional stability and other mechanical properties.
- Specific examples thereof include silica (silicon dioxide), titania (titanium oxide), alumina ( Aluminum oxide) and the like.
- the curing agent is added, for example, for the purpose of improving strength, surface hardness, dimensional stability and other mechanical properties, and specific examples thereof include, for example, thermosetting agents such as phenol resins, acrylate monomers and photopolymerization. Examples thereof include a photo-curing agent using a property initiator.
- the plasticizer is added for the purpose of improving mechanical properties such as tensile strength and bending strength, and specific examples thereof include phthalates and phosphates.
- composition of the present invention can be prepared by a known method, for example, by the method disclosed in WO05 / 052058.
- a conductive molded body is obtained from the composition of the present invention.
- it is a molded product of a conductive polyaniline composition by applying a composition obtained by adding a small amount of a compound having a phenolic hydroxyl group to a complex of polyaniline and protonic acid dissolved in an organic solvent on glass or the like.
- a film can be obtained.
- the molded article of the conductive polyaniline composition obtained after coating can be made into a conductive article having excellent heat resistance.
- Aerosol OT sodium diisooctylsulfosuccinate, purity 75% or more, manufactured by Wako Pure Chemical Industries, Ltd.
- Aerosol OT sodium diisooctylsulfosuccinate, purity 75% or more, manufactured by Wako Pure Chemical Industries, Ltd.
- the elemental analysis result at the time of removing a volatile matter substantially is shown below.
- the mole fraction of aniline monomer units / sulfosuccinate in the composite is 0.62.
- Production Example 4 [Production of Protonated Monoisopropyl Naphthalenesulfonic Acid]
- Protonated monoisopropyl naphthalene sulfonic acid was prepared in the same manner as in Production Example 2 except that sodium monoisopropyl naphthalene sulfonate (manufactured by Takemoto Yushi) was used in place of sodium di (2-ethylhexyl) sulfosuccinate.
- Production Example 5 [Production of Protonated Diisopropylnaphthalenesulfonic Acid] Protonation was carried out in the same manner as in Production Example 2 except that sodium diisopropylnaphthalenesulfonate (manufactured by Takemoto Yushi) was used in place of sodium di (2-ethylhexyl) sulfosuccinate. Diisopropyl naphthalene sulfonic acid was prepared.
- Production Example 6 [Production of Protonated Triisopropylnaphthalenesulfonic Acid]
- Protonated triisopropyl naphthalene sulfonic acid was prepared in the same manner as in Production Example 2 except that sodium triisopropyl naphthalene sulfonate (manufactured by Takemoto Yushi Co., Ltd.) was used instead of sodium di (2-ethylhexyl) sulfosuccinate.
- Example 1 [Preparation of conductive polyaniline composition] 500 mg of the conductive polyaniline complex obtained in Production Example 1 was dissolved again in 10 g of toluene to prepare a uniform conductive polyaniline complex solution. To this solution, 0.01 g (0.024 mmol) of di (2-ethylhexyl) sulfosuccinic acid obtained in Production Example 2 and 1 g (9.2 mmol) of m-cresol were added to obtain a uniform conductive polyaniline composition. . According to the following method, the acidity (pKa) of di (2-ethylhexyl) sulfosuccinic acid was ⁇ 2.5.
- the portion of the conductive polyaniline thin film 5 covering the terminal of the ITO electrode was scraped off in a nitrogen atmosphere to expose the terminal of the ITO electrode on the surface.
- the resistance of the thin film was measured using a Lorester GP (manufactured by Mitsubishi Chemical Co., Ltd .; resistivity meter by a four-terminal method), and the resistance value immediately after the film formation was defined as an initial value R 0 . did.
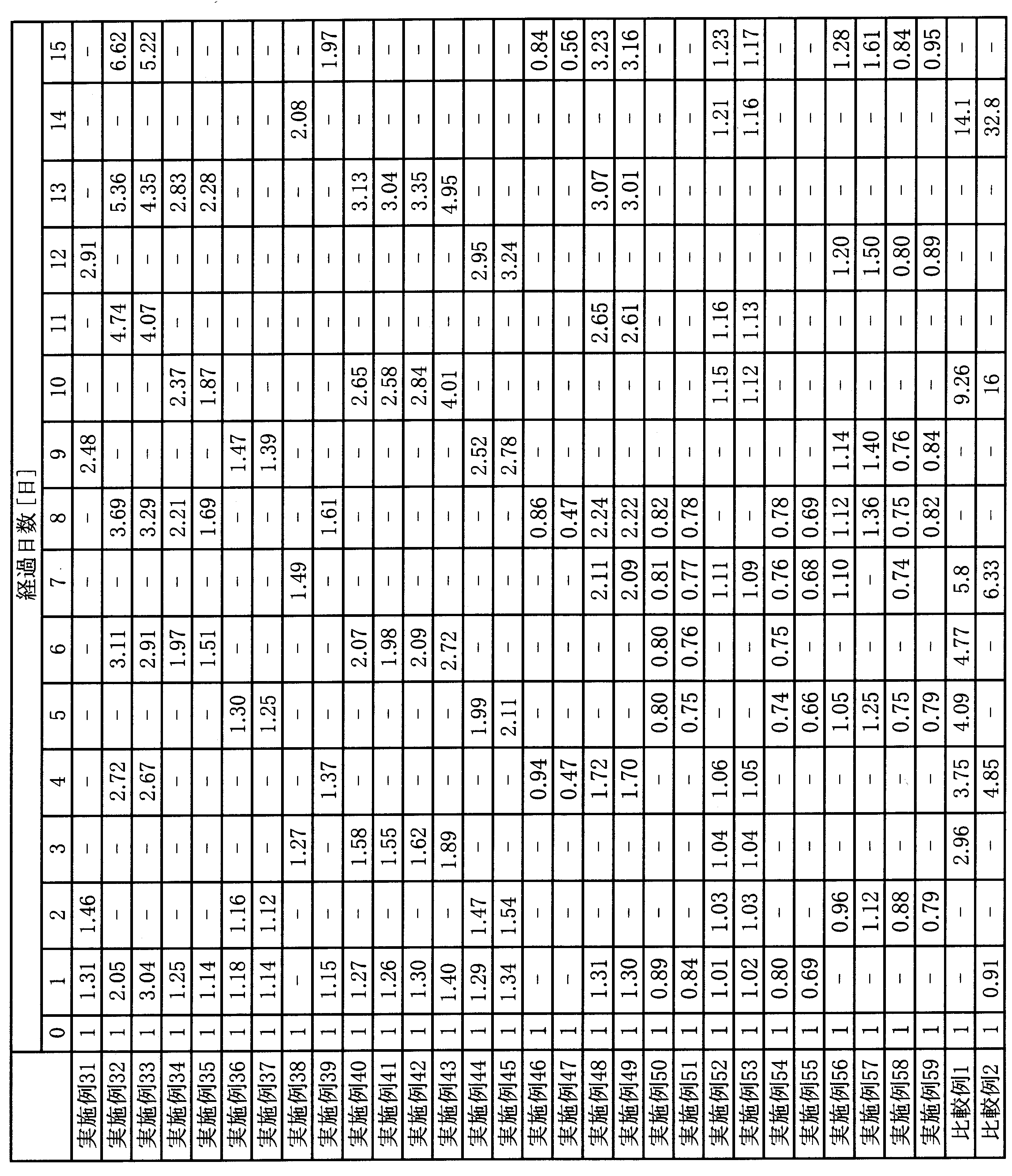
- Example 2 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that the addition amount of di (2-ethylhexyl) sulfosuccinic acid was changed to 0.05 g (0.12 mmol). Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 3 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that the addition amount of di (2-ethylhexyl) sulfosuccinic acid was changed to 0.2 g (0.47 mmol). Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 4 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that the addition amount of di (2-ethylhexyl) sulfosuccinic acid was changed to 0.5 g (1.18 mmol). Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 5 The same procedure as in Example 1 was repeated except that 0.01 g (0.02 mmol) of di (2-ethylhexyl) sulfosuccinic acid aniline salt obtained in Production Example 3 was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 6 The same procedure as in Example 1 was repeated except that 0.05 g (0.10 mmol) of di (2-ethylhexyl) sulfosuccinic acid aniline salt obtained in Preparation Example 3 was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 7 The same procedure as in Example 1 was repeated except that 0.1 g (0.20 mmol) of di (2-ethylhexyl) sulfosuccinic acid aniline salt obtained in Production Example 3 was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 8 The same procedure as in Example 1 was repeated except that 0.2 g (0.39 mmol) of di (2-ethylhexyl) sulfosuccinic acid aniline salt obtained in Production Example 3 was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 9 The same procedure as in Example 1 was repeated except that 0.5 g (0.98 mmol) of di (2-ethylhexyl) sulfosuccinic acid aniline salt obtained in Production Example 3 was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 10 Uniform conductivity was obtained in the same manner as in Example 1 except that 0.1 g (0.23 mmol) of sodium di (2-ethylhexyl) sulfosuccinate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a functional polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 11 The same procedure as in Example 1 was conducted except that 0.2 g (0.45 mmol) of sodium di (2-ethylhexyl) sulfosuccinate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 12 Uniform conductivity was obtained in the same manner as in Example 1, except that 0.3 g (0.68 mmol) of sodium di (2-ethylhexyl) sulfosuccinate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a functional polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 13 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.02 g (0.1 mmol) of methanesulfonic acid (manufactured by Wako Pure Chemical Industries, Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. According to the same method as in Example 1, the acidity (pKa) of methanesulfonic acid was ⁇ 1.5.
- Example 14 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.04 g (0.2 mmol) of methanesulfonic acid (manufactured by Wako Pure Chemical Industries, Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 15 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.08 g (0.4 mmol) of methanesulfonic acid (manufactured by Wako Pure Chemical Industries, Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 16 The same procedure as in Example 1 except that 0.058 g (0.21 mmol) of dodecylphosphoric acid (phosphanol ML200, manufactured by Daiichi Kogyo Seiyaku) represented by the following formula was added instead of di (2-ethylhexyl) sulfosuccinic acid. Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. In addition, according to the method similar to Example 1, the acidity (pKa) of dodecyl phosphoric acid was 0.84.
- dodecylphosphoric acid phosphanol ML200, manufactured by Daiichi Kogyo Seiyaku
- Example 17 Uniform conductivity as in Example 1 except that 0.11 g (0.43 mmol) of dodecyl phosphoric acid (Phosphanol ML200, manufactured by Daiichi Kogyo Seiyaku) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 18 A uniform solution was obtained in the same manner as in Example 1 except that 0.094 g (0.42 mmol) of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. According to the same method as in Example 1, the acidity (pKa) of 2-naphthalenesulfonic acid was ⁇ 2.4.
- Example 19 A uniform solution was obtained in the same manner as in Example 1 except that 0.187 g (0.83 mmol) of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added in place of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 20 Uniform conductive polyaniline as in Example 1 except that 0.08 g (0.43 mmol) of p-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. According to the same method as in Example 1, the acidity (pKa) of p-xylene sulfonic acid was ⁇ 1.8.
- Example 21 Uniform conductive polyaniline as in Example 1 except that 0.16 g (0.86 mmol) of p-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 22 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.05 g (0.41 mmol) of benzoic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. In addition, according to the method similar to Example 1, the acidity (pKa) of benzoic acid was 4.3.
- Example 23 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.1 g (0.82 mmol) of benzoic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 24 Uniform conductive polyaniline composition as in Example 1 except that 0.05 g (0.39 mmol) of cyclohexanecarboxylic acid (Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. In addition, according to the method similar to Example 1, the acidity (pKa) of cyclohexanecarboxylic acid was 4.6.
- Example 25 Uniform conductive polyaniline composition as in Example 1, except that 0.1 g (0.78 mmol) of cyclohexanecarboxylic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 26 Uniform conductive polyaniline as in Example 1 except that 0.08 g (0.42 mmol) of p-toluenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. According to the same method as in Example 1, the acidity (pKa) of p-toluenesulfonic acid was ⁇ 2.2.
- Example 27 Uniform conductive polyaniline as in Example 1 except that 0.16 g (0.84 mmol) of p-toluenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 28 Uniform conductive polyaniline composition as in Example 1, except that 0.065 g (0.41 mmol) of phenylphosphonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. According to the same method as in Example 1, the acidity (pKa) of phenylphosphonic acid was 2.2.
- Example 29 Uniform conductive polyaniline composition as in Example 1, except that 0.13 g (0.82 mmol) of phenylphosphonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2.
- Example 30 Uniform conductive polyaniline in the same manner as in Example 1 except that 0.08 g (0.43 mmol) of m-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 1 and 2. Incidentally, according to the same method as in Example 1, the acidity (pKa) of m-xylene sulfonic acid was ⁇ 2.0.
- Example 31 Uniform conductive polyaniline as in Example 1 except that 0.16 g (0.86 mmol) of m-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 32 Uniform conductive polyaniline composition as in Example 1 except that 0.046 g (0.42 mmol) of ethanesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- ethanesulfonic acid manufactured by Tokyo Chemical Industry Co., Ltd.
- di (2-ethylhexyl) sulfosuccinic acid was prepared.
- a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated.
- Tables 3 and 4 According to the same method as in Example 1, the acidity (pKa) of ethanesulfonic acid was ⁇ 2.6.
- Example 33 Uniform conductive polyaniline composition as in Example 1, except that 0.091 g (0.83 mmol) of ethanesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 34 Uniform conductive polyaniline composition as in Example 1, except that 0.135 g (0.41 mmol) of dodecylbenzenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid A product was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Note that, according to the same method as in Example 1, the acidity (pKa) of dodecylbenzenesulfonic acid was ⁇ 2.8.
- Example 35 A uniform conductive polyaniline composition as in Example 1 except that 0.27 g (0.83 mmol) of dodecylbenzenesulfonic acid (Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid A product was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 36 The same as in Example 1 except that 0.13 g (0.40 mmol) of bis (2-ethylhexyl) hydrogen phosphate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. According to the same method as in Example 1, the acidity (pKa) of bis (2-ethylhexyl) hydrogen phosphate was 1.2.
- Example 37 The same as in Example 1 except that 0.27 g (0.84 mmol) of bis (2-ethylhexyl) hydrogen phosphate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid.
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 38 Example 1 except that 0.10 g (0.40 mmol) of 8-ethoxyquinoline-5-sulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Incidentally, according to the same method as in Example 1, the acidity (pKa) of 8-ethoxyquinoline-5-sulfonic acid was ⁇ 2.1.
- Example 39 Example 1 except that 0.21 g (0.83 mmol) of 8-ethoxyquinoline-5-sulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 40 Uniform conductive polyaniline as in Example 1 except that 0.06 g (0.42 mmol) of 2-ethylhexanoic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Incidentally, according to the same method as in Example 1, the acidity (pKa) of 2-ethylhexanoic acid was 4.5.
- Example 41 Uniform conductive polyaniline in the same manner as in Example 1 except that 0.12 g (0.83 mmol) of 2-ethylhexanoic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 42 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.06 g (0.43 mmol) of salicylic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. did. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. In addition, according to the method similar to Example 1, the acidity (pKa) of salicylic acid was 3.5.
- Example 43 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.12 g (0.87 mmol) of salicylic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. did. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 44 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.075 g (0.41 mmol) of undecylenic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. In addition, according to the method similar to Example 1, the acidity (pKa) of undecylenic acid was 4.6.
- Example 45 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.15 g (0.81 mmol) of undecylenic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 46 Uniform conductive polyaniline as in Example 1 except that 0.072 g (0.41 mmol) of p-phenolsulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Incidentally, according to the same method as in Example 1, the acidity (pKa) of p-phenolsulfonic acid was ⁇ 2.2.
- Example 47 Uniform conductive polyaniline in the same manner as in Example 1, except that 0.144 g (0.83 mmol) of p-phenolsulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 48 A uniform conductive polyaniline composition in the same manner as in Example 1 except that 0.129 g (0.41 mmol) of anthraquinonesulfonic acid Na (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A product was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. According to the same method as in Example 1, the acidity (pKa) of anthraquinone sulfonic acid was ⁇ 0.89.
- Example 49 A uniform conductive polyaniline composition in the same manner as in Example 1 except that 0.259 g (0.83 mmol) of anthraquinonesulfonic acid Na (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A product was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 50 Uniform conductive polyaniline composition as in Example 1, except that 0.104 g (0.41 mmol) of monoisopropylnaphthalenesulfonic acid obtained in Production Example 4 was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Note that, according to the same method as in Example 1, the acidity (pKa) of monoisopropylnaphthalenesulfonic acid was ⁇ 2.3.
- Example 51 Uniform conductive polyaniline composition as in Example 1, except that 0.207 g (0.83 mmol) of monoisopropylnaphthalenesulfonic acid obtained in Production Example 4 was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 52 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that 0.121 g (0.41 mmol) of diisopropylnaphthalenesulfonic acid obtained in Production Example 5 was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Incidentally, according to the same method as in Example 1, the acidity (pKa) of diisopropylnaphthalenesulfonic acid was ⁇ 2.2.
- Example 53 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1, except that 0.224 g (0.83 mmol) of diisopropylnaphthalenesulfonic acid obtained in Production Example 5 was added instead of di (2-ethylhexyl) sulfosuccinic acid. Prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 54 Uniform conductive polyaniline composition as in Example 1, except that 0.139 g (0.41 mmol) of triisopropylnaphthalenesulfonic acid obtained in Production Example 6 was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. Incidentally, according to the same method as in Example 1, the acidity (pKa) of triisopropylnaphthalenesulfonic acid was ⁇ 2.1.
- Example 55 Uniform conductive polyaniline composition as in Example 1, except that 0.277 g (0.83 mmol) of triisopropylnaphthalenesulfonic acid obtained in Preparation Example 6 was added instead of di (2-ethylhexyl) sulfosuccinic acid was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 56 Uniform conductive polyaniline as in Example 1 except that 0.084 g (0.41 mmol) of 3-nitrobenzenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. According to the same method as in Example 1, the acidity (pKa) of 3-nitrobenzenesulfonic acid was ⁇ 2.8.
- Example 57 Uniform conductive polyaniline in the same manner as in Example 1 except that 0.168 g (0.83 mmol) of 3-nitrobenzenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added in place of di (2-ethylhexyl) sulfosuccinic acid. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 58 The same as Example 1 except that 0.128 g (0.41 mmol) of 2-hydroxy-4-benzophenone-5-sulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4. According to the same method as in Example 1, the acidity (pKa) of 2-hydroxy-4-benzophenone-5-sulfonic acid was ⁇ 2.8.
- Example 59 Example 2 except that 0.255 g (0.83 mmol) of 2-hydroxy-4-benzophenone-5-sulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of di (2-ethylhexyl) sulfosuccinic acid. Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Comparative Example 1 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that di (2-ethylhexyl) sulfosuccinic acid was not added. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Comparative Example 2 A uniform conductive polyaniline composition was prepared in the same manner as in Example 1 except that di (2-ethylhexyl) sulfosuccinic acid and m-cresol were not added. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 1, and the obtained thin film was evaluated. The results are shown in Tables 3 and 4.
- Example 60 [Preparation of conductive polyaniline composition] 500 mg of the conductive polyaniline complex obtained in Production Example 1 was dissolved again in 10 g of toluene to prepare a uniform conductive polyaniline complex solution. To this solution, 0.023 g (0.11 mmol) of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.) and 1 g (9.2 mmol) of m-cresol were added to obtain a uniform conductive polyaniline composition. Obtained.
- the portion of the conductive polyaniline thin film 5 covering the terminal of the ITO electrode was scraped off in a nitrogen atmosphere to expose the terminal of the ITO electrode on the surface.
- the resistance of the thin film was measured using a Lorester GP (manufactured by Mitsubishi Chemical Co., Ltd .; resistivity meter by a four-terminal method), and the resistance value immediately after the film formation was defined as an initial value R 0 . did.
- Example 61 A uniform conductive polyaniline composition was prepared in the same manner as in Example 60 except that the amount of 2-naphthalenesulfonic acid hydrate added was changed to 0.047 g (0.22 mmol). Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 62 A uniform conductive polyaniline composition was prepared in the same manner as in Example 60 except that the amount of 2-naphthalenesulfonic acid hydrate added was 0.094 g (0.42 mmol). Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 63 A uniform conductive polyaniline composition was prepared in the same manner as in Example 60 except that the amount of 2-naphthalenesulfonic acid hydrate added was 0.38 g (1.8 mmol). Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 60 is the same as Example 60 except that 0.02 g (0.11 mmol) of p-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Similarly, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 60 is the same as Example 60 except that 0.04 g (0.22 mmol) of p-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Similarly, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 66 Except that 0.08 g (0.43 mmol) of p-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.), Example 60 and Similarly, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 67 Example 60 is the same as Example 60 except that 0.46 g (2.5 mmol) of p-xylenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Similarly, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 68 In the same manner as in Example 60 except that 0.2 g (1.6 mmol) of benzoic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). A uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 69 Except for adding 0.4 g (3.2 mmol) of benzoic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.), the same procedure as in Example 60 was performed. A uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 70 Except that 0.072 g (0.41 mmol) of p-phenolsulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.), Example 60 and Similarly, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 71 Except that 0.144 g (0.83 mmol) of p-phenolsulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.), Example 60 and Similarly, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 72 The same procedure as in Example 60 except that 0.13 g (0.42 mmol) of anthraquinonesulfonic acid Na (Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (Tokyo Chemical Industry Co., Ltd.). Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 73 The same procedure as in Example 60 except that 0.26 g (0.83 mmol) of anthraquinone sulfonic acid Na (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Thus, a uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 74 The same procedure as in Example 60 was conducted, except that 0.104 g (0.41 mmol) of monoisopropylnaphthalenesulfonic acid obtained in Production Example 4 was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 75 The same procedure as in Example 60 was conducted, except that 0.207 g (0.83 mmol) of monoisopropylnaphthalenesulfonic acid obtained in Production Example 4 was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 76 The same procedure as in Example 60 was conducted, except that 0.121 g (0.41 mmol) of diisopropylnaphthalenesulfonic acid obtained in Production Example 5 was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). A uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 77 The same procedure as in Example 60 was conducted, except that 0.242 g (0.83 mmol) of diisopropylnaphthalenesulfonic acid obtained in Production Example 5 was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). A uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 78 The same procedure as in Example 60 was conducted, except that 0.139 g (0.41 mmol) of triisopropylnaphthalenesulfonic acid obtained in Production Example 6 was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 79 The same procedure as in Example 60 was conducted, except that 0.277 g (0.83 mmol) of triisopropylnaphthalenesulfonic acid obtained in Production Example 6 was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.). Uniform conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 80 The same as in Example 60 except that 0.06 g (0.43 mmol) of salicylic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.).
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 81 Uniform in the same manner as in Example 60 except that 0.12 g (0.87 mmol) of salicylic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate (manufactured by Tokyo Chemical Industry Co., Ltd.).
- a conductive polyaniline composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 82 Uniform conductive polyaniline as in Example 60, except that 0.084 g (0.45 mmol) of 3-nitrobenzenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6.
- Example 83 A uniform conductive polyaniline was obtained in the same manner as in Example 60 except that 0.168 g (0.90 mmol) of 3-nitrobenzenesulfonic acid (manufactured by Tokyo Chemical Industry Co., Ltd.) was added instead of 2-naphthalenesulfonic acid hydrate. A composition was prepared. Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. The results are shown in Tables 5 and 6. Comparative Example 3 A uniform conductive polyaniline composition was prepared in the same manner as in Example 60 except that 2-naphthalenesulfonic acid hydrate was not added.
- Example 60 Using the obtained conductive polyaniline composition, a thin film was formed in the same manner as in Example 60, and the obtained thin film was evaluated. As a result, the R / Ro value at the elapsed day 1 is 2, the R / Ro value at the elapsed day 4 is 8, the R / Ro value at the elapsed day 6 is 26, and the elapsed day is 8 days.
- the value of R / Ro is 38, the value of R / Ro at 11 days elapsed is 88, the value of R / Ro at 13 days elapsed is 150, the value of R / Ro at 14 days elapsed is 196
- the value of R / Ro at the elapsed days of 15 days is 246, the value of R / Ro at the elapsed days of 21 days is 930, the value of R / Ro at the elapsed days of 22 days is 1126,
- the value of R / Ro at 26 days elapsed was 2149, the value of R / Ro at 27 days elapsed was 2551, and the value of R / Ro at 28 days elapsed was 3394.
- the molded product obtained from the ⁇ -conjugated polymer composition of the present invention is used in the field of power electronics and optoelectronics, and is used for electrostatic and antistatic materials, transparent electrodes and conductive film materials, electroluminescent element materials, circuit materials, and electromagnetic wave shielding. It can be used for materials, capacitor dielectrics and electrolytes, electrode materials for solar cells and secondary batteries, fuel cell separator materials, etc., or for plating bases, rust inhibitors, and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Polymers & Plastics (AREA)
- Medicinal Chemistry (AREA)
- Electrochemistry (AREA)
- Power Engineering (AREA)
- Engineering & Computer Science (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Physics & Mathematics (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Paints Or Removers (AREA)
- Conductive Materials (AREA)
- Non-Insulated Conductors (AREA)
- Coating Of Shaped Articles Made Of Macromolecular Substances (AREA)
Abstract
Description
この課題を解決するためにポリアニリン以外の固形分であるバインダー樹脂を添加することも知られているが、バインダー樹脂は絶縁体であるため、得られた成形体の導電性が低くなるという別の欠点がある。
また、分散型のポリアニリン組成物ではドープされたポリアニリン微粒子が凝集、沈殿するため、この分散型のポリアニリン組成物を用いて成形体を製造する場合には、製造時にこの組成物を混合しポリアニリン微粒子を組成物中に分散させないといけない欠点がある。
本発明の第一のπ共役高分子組成物は、
(a)溶剤、
(b)前記溶剤に溶解した、ドーパントによりドープされたπ共役高分子、
(c)酸性物質及び酸性物質の塩の少なくとも1つ、並びに
(d)フェノール性化合物を含むπ共役高分子組成物であって、
前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質と前記フェノール性化合物が異なり、前記酸性物質の塩のみを含む場合、前記酸性物質の塩と前記フェノール性化合物が異なり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つとフェノール性化合物が異なる。
少なくとも下記(a)~(d)を原料として用いる。
(a)溶剤
(b)前記溶剤に溶解する、ドーパントによりドープされたπ共役高分子
(c)酸性物質及び酸性物質の塩の少なくとも1つ
(d)フェノール性化合物
(前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質と前記フェノール性化合物が異なり、前記酸性物質の塩のみを含む場合、前記酸性物質の塩と前記フェノール性化合物が異なり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つとフェノール性化合物が異なる。)
前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質は前記ドーパントと異なり、前記酸性物質の塩のみを含む場合、前記酸性物質の塩は前記ドーパントと異なり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つと前記ドーパントが異なることが好ましい。
前記π共役高分子が窒素原子を含み、
前記ドーパントがスルホン酸であり、
前記成分(c)のうち、前記酸性物質のみを含む場合、その酸性物質が前記ドーパントと同一又は異なるスルホン酸であり、
前記酸性物質の塩のみを含む場合、その酸性物質の塩が前記ドーパントと同一又は異なるスルホン酸の塩であり、
前記酸性物質及び前記酸性物質の塩を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つが前記ドーパントと同一又は異なるスルホン酸又はスルホン酸の塩であり、
式(1)を満たすことが好ましい。
0.21≦S1/N1≦1.2…(1)
(式中、S1は組成物に含まれる硫黄原子のモル数であり、N1は組成物に含まれる窒素原子のモル数である。)
前記π共役高分子が窒素原子を含み、
前記ドーパントがスルホン酸であり、
前記成分(c)のうち、前記酸性物質のみを含む場合、その酸性物質が前記ドーパントと同一又は異なるスルホン酸であり、
前記酸性物質の塩のみを含む場合、その酸性物質の塩が前記ドーパントと同一又は異なるスルホン酸の塩であり、
前記酸性物質及び前記酸性物質の塩を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つが前記ドーパントと同一又は異なるスルホン酸又はスルホン酸の塩であり、
前記酸性物質のみを含む場合には、式(2)を満たし、
前記酸性物質の塩のみを含む場合には、式(3)を満たし、
前記酸性物質及び前記酸性物質の塩を含む場合、式(4)を満たすことが好ましい。
0.01≦S2/N2≦0.5…(2)
0.01≦S3/N3≦0.5…(3)
0.01≦S4/N4≦0.5…(4)
(式中、S2は組成物に含まれる全ての酸性物質の硫黄原子のモル数の合計であり、S3は組成物に含まれている全ての酸性物質の塩の硫黄原子のモル数の合計であり、S4は組成物に含まれている全ての酸性物質及び酸性物質の塩の硫黄原子のモル数の合計であり、N2~N4は組成物に含まれる全てのドーパントによりドープされたπ共役高分子の窒素原子のモル数の合計である。)
前記π共役高分子が窒素原子を含み、前記ドーパントがスルホン酸であり、式(5)を満たすことが好ましい。
0.2≦S5/N5≦0.7…(5)
(S5は組成物に含まれる前記ドーパントによりドープされたπ共役高分子の硫黄原子のモル数の合計であり、N5は組成物に含まれる前記ドーパントによりドープされたπ共役高分子の窒素原子のモル数の合計である。)
前記ドーパントによりドープされたπ共役高分子の成形体の、4端子法により測定した導電度が0.01S/cm以上であり、
前記成形体は、パターニングされたインジウム錫酸化物電極を形成したガラス基板上に、窒素雰囲気下で前記ドーパントによりドープされたπ共役高分子500mgをトルエン10gに溶解した溶液1mlを、500rpmで15秒間スピンコート法により塗布し、窒素雰囲気下80℃で5分間乾燥して得られたものであることが好ましい。
成形体の4端子法により測定した導電度が0.01S/cm以上であり、
前記成形体は、パターニングされたインジウム錫酸化物電極を形成したガラス基板上に、窒素雰囲気下で前記π共役高分子組成物1mlを500rpmで15秒間スピンコート法により塗布し、窒素雰囲気下80℃で5分間乾燥して得られたものであることが好ましい。
前記ドープされたπ共役高分子が、プロトネーションされた置換又は非置換ポリアニリンであることが好ましい。
本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物は、
前記ドープされたπ共役高分子が、スルホン酸でプロトネーションされた置換又は非置換ポリアニリンであることが好ましい。
前記スルホン酸が下記式(III)で示されるスルホコハク酸誘導体であることが好ましい。
M(O3SCH(CH2COOR12)COOR13)m (III)
(式(III)において、
Mは、水素原子、有機遊離基又は無機遊離基であり、
mはMの価数であり、
R12及びR13は、それぞれ独立して炭化水素基又は-(R14O)r-R15で表される基であり、R14は炭化水素基又はシリレン基であり、R15は水素原子、炭化水素基又はR16 3Si-で表される基であり、R16は炭化水素基であり、3つのR16は同一又は異なっていてもよく、rは1以上の整数である。)
本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物は、
前記酸性物質が有機酸であることが好ましい。
前記酸性物質又は前記酸性物質の塩が、スルホン酸基、リン酸基、ホスホン酸基及びカルボキシ基から選択される1つ以上の酸性基を有する1つ以上の酸性物質又は酸性物質の塩であることが好ましい。
本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物は、
前記酸性物質が、酸性基を1つ以上有する環状、鎖状又は分岐のアルキル酸であることが好ましい。
前記酸性物質が、酸性基を1つ以上有する置換又は無置換の芳香族酸であることが好ましい。
本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物は、
前記芳香族酸が、ナフタレン骨格を有する酸であることが好ましい。
本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物は、
前記フェノール性化合物が下記式(1)で表されるフェノール性化合物であることが好ましい。
本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物は、
前記フェノール性化合物が下記式(2)で表されるフェノール性化合物であることが好ましい。
Rは、それぞれ炭素数2~10のアルキル基、アルケニル基、アルキルチオ基、炭素数3~10のシクロアルキル基、炭素数6~20のアリール基、アルキルアリール基又はアリールアルキル基である。)
前記フェノール性化合物が下記式(3)で表されるフェノール性化合物であることが好ましい。
Rは、それぞれ炭素数2~10のアルキル基、アルケニル基、アルキルチオ基、炭素数3~10のシクロアルキル基、炭素数6~20のアリール基、アルキルアリール基又はアリールアルキル基である。)
本発明の第二のコンデンサは、本発明の第一のπ共役高分子組成物又は本発明の第二のπ共役高分子組成物を用いて製造される。
本発明の導電性成形体は、本発明の第一のπ共役高分子組成物又は本発明の第二のπ共役高分子組成物を成形してなる。
本発明の導電性フィルムは、本発明の第一のπ共役高分子組成物又は本発明の第二のπ共役高分子組成物を成膜してなる。
本発明の表面導電性物品は、本発明の第一のπ共役高分子組成物又は本発明の第二のπ共役高分子組成物を、基材に塗布してなる。
本発明の表面導電性物品は、前記基材が樹脂フィルムであることが好ましい。
本発明の導電性物品は、本発明の第一のπ共役高分子組成物又は本発明の第二のπ共役高分子組成物と基材を混合してなる。
前記ドープされたπ共役高分子が、スルホコハク酸誘導体でプロトネーションされた置換又は非置換ポリアニリンであり、
前記酸性物質が、スルホン酸基を有し、ナフタレン骨格を有する酸であり、さらにフェノール性化合物を含む。
π共役高分子は、ドーパントによりドープされたπ共役高分子である。詳細は後述する。
酸性物質、酸性物質の塩及びフェノール性化合物の詳細は後述する。
尚、本発明の第一のπ共役高分子組成物は、例えば溶剤とこの溶剤に溶解するπ共役高分子と、酸性物質及び酸性物質の塩の少なくとも1つと、フェノール性化合物を混合して得ることができる。
尚、本発明の第二のπ共役高分子組成物は、例えば、溶剤とこの溶剤に溶解するπ共役高分子と、酸性物質及び酸性物質の塩の少なくとも1つと、フェノール性化合物を混合して得ることができる。
尚、以下、本発明の組成物は、本発明の第一のπ共役高分子組成物及び本発明の第二のπ共役高分子組成物の両方を含む概念である。
0.21≦S1/N1≦1.2・・・(1)
(ここで、S1は組成物に含まれる硫黄原子のモル数であり、N1は組成物に含まれる窒素原子のモル数を意味する。)
0.01≦S2/N2≦0.5…(2)
0.01≦S3/N3≦0.5…(3)
0.01≦S4/N4≦0.5…(4)
0.2≦S5/N5≦0.7…(5)
ここで、S5は組成物に含まれる前記ドーパントによりドープされたπ共役高分子の硫黄原子のモル数の合計であり、N5は組成物に含まれる前記ドーパントによりドープされたπ共役高分子の窒素原子のモル数の合計である。
尚、窒素原子及び硫黄原子のモル数は有機元素分析法により測定した値である。
上記π共役高分子の具体例としては、置換又は無置換のポリアニリン、ポリピロール、ポリチオフェン、ポリ(p-フェニレン)、ポリ(p-フェニレンビニレン)、及びこれらの誘導体等が挙げられ、汎用性及び経済性の点から、好ましくは置換又は無置換のポリアニリン及び/又はポリアニリン誘導体である。
また、分子量分布は、例えば1.5~10.0である。導電率の観点から、分子量分布は小さい方が好ましいが、溶剤への溶解性及び成形性の観点では、分子量分布が広い方が好ましい場合もある。
上記分子量及び分子量分布は、ゲルパーミエーションクロマトグラフ(GPC)により測定できる。
ここでπ共役高分子がドーパントによってドープされるとは、ドーパントによってπ共役高分子に自由に動くことができる電荷移動体(キャリア)が注入されていることを意味する。
ドープされたπ共役高分子のドープ率aは、好ましくは0<a<1であり、より好ましくは0.7以下であり、さらに好ましくは0.2<a<0.7であり、最も好ましくは0.4<a<0.7である。
尚、ドープ率が0.2以下であると導電性及びπ共役高分子の溶剤への溶解性が低下するおそれがある。また、ドープ率が0.7以上になると導電性が低下する。
ドーパントのドープ率aが、0.5であることは、窒素原子2つに対して1分子のドーパントがドープすることを意味する。
ここでの成形体とは、ガラス基板上に形成されたπ共役高分子の成形体自体をいう。尚、導電率は、例えば、以下のようにして得られる。
π共役高分子薄膜を乾燥後、図2に示すように、π共役高分子薄膜3のITO電極の端子を覆っている部分を、窒素雰囲気下で削り取り、ITO電極の端子を表面に露出させる。表面に露出したITO電極の端子を用いて、三菱化学社製の抵抗率計を用いて4端子法で導電度を測定する。
ここで、成形体は、以下のようにして得られる。図1に示す、パターニングによりITO電極2が表面に形成されたガラス基板1の上面に、π共役高分子組成物1mlを塗布する。具体的には、スピンコート法により塗布する。ここでスピンコート法による塗布は、窒素雰囲気下で行う。また、スピンコート法の、ガラス基板にπ共役高分子組成物を滴下した後のガラス基板回転時間は、15秒間である。また、スピンコート法のガラス基板回転速度は、500rpmである。その後、ガラス基板を乾燥してπ共役高分子組成物薄膜を形成する。ここで乾燥は、窒素雰囲気下で行う。また、乾燥時間は、5分間である。また、乾燥温度は、80℃である。
ここでの成形体とは、ガラス基板上に形成されたπ共役高分子組成物の成形体自体をいう。尚、導電率は、例えば、以下のようにして得られる。π共役高分子組成物薄膜を乾燥後、図3に示すように、π共役高分子組成物薄膜4のITO電極の端子を覆っている部分を、窒素雰囲気下で削り取り、ITO電極の端子を表面に露出させる。表面に露出したITO電極の端子を用いて、三菱化学社製の抵抗率計を用いて4端子法で導電度を測定する。
ドーパントとしては、例えば、有機プロトン酸又はその塩が挙げられる。
置換ポリアニリンの置換基としては、例えば、メチル基、エチル基、ヘキシル基、オクチル基等の直鎖又は分岐の炭化水素基;メトキシ基、フェノキシ基等のアルコキシル基;アリーロキシ基;CF3基等の含ハロゲン炭化水素基等が挙げられる。
M(XARn)m (I)
上記有機遊離基としては、例えば、ピリジニウム基、イミダゾリウム基、アニリニウム基等が挙げられる。上記無機遊離基としては、例えばナトリウム、リチウム、カリウム、セシウム、アンモニウム等が挙げられる。
当該炭化水素基としては、例えば炭素数1~24の直鎖若しくは分岐状のアルキル基;アルケニル基;シクロペンチル、シクロヘキシル、シクロヘプチル、シクロオクチル、メンチル等の置換基を含んでいてもよいシクロアルキル基;ビシクロヘキシル、ノルボルニル、アダマンチル等の縮合してもよいジシクロアルキル基若しくはポリシクロアルキル基;フェニル、トシル、チオフェニル、ピローリニル、ピリジニル、フラニル等の置換基を含んでいてもよい芳香環を含むアリール基;ナフチル、アントラセニル、フルオレニル、1,2,3,4-テトラヒドロナフチル、インダニル、キノリニル、インドニル等の縮合していてもよいジアリール基若しくはポリアリール基;アルキルアリール基等であって、対応する(n+1)価の基が挙げられる。
ここで、R1は炭素数が4以上の置換基を含んでもよい炭化水素基、シリル基、アルキルシリル基、-(R2O)x-R3で表される基、又は-(OSiR3 2)x-OR3(R2はアルキレン基、R3はそれぞれ同一でも異なっていてもよい炭化水素基であり、xは1以上の整数である)で表される基である。
R1の炭化水素基の例としては、直鎖若しくは分岐のブチル基、ペンチル基、ヘキシル基、ヘプチル基、オクチル基、ノニル基、デシル基、ドデシル基、ペンタデシル基、エイコサニル基等が挙げられる。
M(XCR4(CR5 2COOR6)COOR7)p (II)
pはMの価数である。
R4及びR5の炭化水素基としては、炭素数1~24の直鎖若しくは分岐状のアルキル基;芳香環を含むアリール基;アルキルアリール基等が挙げられる。
R8の炭化水素基は、R4及びR5の炭化水素基と同様である。
R6及びR7の炭化水素基としては、炭素数1~24、好ましくは炭素数4以上の直鎖若しくは分岐状のアルキル基;芳香環を含むアリール基;アルキルアリール基等が挙げられる。
R6及びR7の炭化水素基の具体例としては、直鎖又は分岐状のブチル基、ペンチル基、ヘキシル基、オクチル基、デシル基等が挙げられる。
また、R10及びR11の炭化水素基としては、R4及びR5の場合と同様である。qは、1~10の整数であることが好ましい。
M(O3SCH(CH2COOR12)COOR13)m (III)
R14の炭化水素基は、R9の炭化水素基と同様である。また、R15及びR16の炭化水素基は、R4及びR5の炭化水素基と同様である。
rは、好ましくは1~10の整数である。
で表される有機プロトン酸又はその塩の具体例は、R6及びR7が-(R9O)n-R10で表される基である場合の式(II)で表される有機プロトン酸又はその塩と同様である。
R12及びR13の炭化水素基は、R6及びR7の炭化水素基と同様であり、ブチル基、ヘキシル基、2-エチルヘキシル基、デシル基等が好ましい。
本発明の組成物が含む酸性物質としては、好ましくはスルホン酸基、リン酸基、ホスホン酸基、カルボキシ基等、酸性の基である酸性基を1つ以上含む有機酸である。
上記アルキルスルホン酸としては、例えば、メタンスルホン酸、エタンスルホン酸、ジ2-エチルヘキシルスルホコハク酸が挙げられる。
上記芳香族スルホン酸としては、例えば、ベンゼン環を有するスルホン酸、ナフタレン骨格を有するスルホン酸、アントラセン骨格を有するスルホン酸が挙げられる。また、上記芳香族スルホン酸としては、置換又は無置換のベンゼンスルホン酸、ナフタレンスルホン酸及びアントラセンスルホン酸が挙げられる。ここで置換基は、例えば、アルキル基、アルコキシ基、ヒドロキシ基、ニトロ基、カルボキシ基、アシル基からなる群から選択される置換基であり、1以上置換していてもよい。例えば、ナフタレンスルホン酸、ドデシルベンゼンスルホン酸、アントラキノンスルホン酸が挙げられる。上記芳香族スルホン酸としては、置換又は無置換のナフタレンスルホン酸が好ましい。
上記ポリスルホン酸は、高分子鎖の主鎖、又は側鎖に複数のスルホン酸基が置換したスルホン酸である。例えば、ポリスチレンスルホン酸が挙げられる。
ここでアルキル基は好ましくは炭素数が1~18の直鎖又は分岐のアルキル基である。
上記アルキルカルボン酸としては、例えば、ウンデシレン酸、シクロヘキサンカルボン酸、2-エチルヘキサン酸等が挙げられる。
上記芳香族カルボン酸としては、置換又は無置換のベンゼンカルボン酸及びナフタレンカルボン酸等が挙げられる。ここで置換基は、例えば、スルホン酸基、アルキル基、アルコキシ基、ヒドロキシ基、ニトロ基、アシル基からなる群から選択される置換基であり、1以上置換していてもよい。例えば、サリチル酸、安息香酸、ナフトエ酸、トリメシン酸等が挙げられる。
ここでアルキル基は好ましくは炭素数が1~18の直鎖又は分岐のアルキル基である。
上記アルキルリン酸又はホスホン酸としては、例えば、ドデシルリン酸、リン酸水素ビス(2-エチルヘキシル)等が挙げられる。
上記芳香族リン酸又はホスホン酸としては、置換又は無置換のベンゼンスルホン酸又はホスホン酸、及びナフタレンスルホン酸又はホスホン酸等が挙げられる。ここで置換基は、例えば、アルキル基、アルコキシ基、ヒドロキシ基、ニトロ基、カルボキシ基、アシル基からなる群から選択される置換基であり、1以上置換していてもよい。例えば、フェニルホスホン酸等が挙げられる。
ここでアルキル基は好ましくは炭素数が1~18の直鎖又は分岐のアルキル基である。
また、本発明の組成物は、上記酸性物質又はその塩を1以上含んでもよい。さらに、本発明の組成物は、異なる複数の酸性物質を含んでいてもよい。また、本発明の組成物は、異なる複数の酸性物質を含んでいてもよい。また、本発明の組成物は、異なる複数の酸性物質及び酸性物質の塩を含んでいてもよい。
of Physical Chemistryの1995年、第99巻、p.2224に記載された方法を用いる。
具体的には、「TURBOMOLE Version 6.1」(COSMO logic社製)を用いて、基底関数にTZVPを用いて構造を最適化し、この構造を用いてCOSMO-RS法計算を「COSMO therm Version C2.1 Release 01.10」(COSMO logic社製)により行う。
ここで、「COSMO therm Version C2.1 Release 01.10」に25℃の水溶媒中との条件と、分子の化学式と、脱プロトンした分子の化学式と、を入力してpKaを算出する。
前記溶剤は、実質的に水に混和しない有機溶剤(水不混和性有機溶剤)でも、水溶性有機溶剤でもよい。
本発明の組成物が含むフェノール性化合物は特に限定されず、ArOH(ここで、Arはアリール基又は置換アリール基である)で示される化合物である。具体的には、フェノール、o-,m-若しくはp-クレゾール、o-,m-若しくはp-エチルフェノール、o-,m-若しくはp-プロピルフェノール、o-,m-若しくはp-ブチルフェノール、o-,m-若しくはp-クロロフェノール、サリチル酸、ヒドロキシ安息香酸、ヒドロキシナフタレン等の置換フェノール類;カテコール、レゾルシノール等の多価フェノール性化合物;及びフェノール樹脂、ポリフェノール、ポリ(ヒドロキシスチレン)等の高分子化合物等を例示することができる。
フェノール性化合物を含む本発明の組成物は、好ましくはフェノール性化合物が下記式(2)で表されるフェノール性化合物であり、さらに好ましくは、式(2)で表されるフェノール性化合物及びドープされたπ共役高分子の重量比(フェノール性化合物/π共役高分子)が0.01~5.0である。
Rは、それぞれ炭素数2~10のアルキル基、アルケニル基、アルキルチオ基、炭素数3~10のシクロアルキル基、炭素数6~20のアリール基、アルキルアリール基又はアリールアルキル基である。)
フェノール性化合物を含む本発明の組成物は、好ましくはフェノール性化合物が下記式(3)で表されるフェノール性化合物であり、さらに好ましくは、式(3)で表されるフェノール性化合物及びドープされたπ共役高分子の重量比(フェノール性化合物/π共役高分子)が0.01~10.0である。
Rは、それぞれ炭素数2~10のアルキル基、アルケニル基、アルキルチオ基、炭素数3~10のシクロアルキル基、炭素数6~20のアリール基、アルキルアリール基又はアリールアルキル基である。)
アルキル基としては、メチル、エチル、プロピル、イソプロピル、ブチル、イソブチル、ターシャルブチル等が挙げられる。
アルケニル基としては、上述したアルキル基の分子内に不飽和結合を有する置換基が挙げられる。
シクロアルキル基としては、シクロペンタン、シクロヘキサン等が挙げられる。
アルキルチオ基としては、メチルチオ、エチルチオ等が挙げられる。
アリール基としては、フェニル、ナフチル等が挙げられる。
アルキルアリール基、及びアリールアルキル基としては、上述したアルキル基とアリール基を組み合わせて得られる置換基等が挙げられる。
これらの基のうち、Rとしては、メチル又はエチル基が好ましい。
これら必須成分の他に、本発明の組成物は、本発明の効果を損なわない範囲で、他の樹脂、無機材料、硬化剤、可塑剤等を含んでもよい。
また樹脂の代わりに、また樹脂と共に、エポキシ樹脂、ウレタン樹脂、フェノール樹脂等の熱硬化性樹脂を形成し得る前駆体を用いてもよい。
例えば、有機溶剤に溶解しているポリアニリンとプロトン酸の複合体に,少量のフェノール性水酸基を有する化合物を添加した組成物を、ガラス等に塗布することで導電性ポリアニリン組成物の成形体である膜を得ることが出来る。ここで塗布前にさらに酸性物質又はその塩を添加することで、塗布後に得られる導電性ポリアニリン組成物の成形体は、耐熱性の優れた導電性物品とすることができる。
[プロトネーションされたポリアニリンの製造]
エーロゾルOT(ジイソオクチルスルホコハク酸ナトリウム、純度75%以上、和光純薬工業製)144gをトルエン4Lに攪拌溶解して調製した溶液を、窒素気流下に置いた30Lのガラス反応器(機械式攪拌器、ジャケット、温度計及び滴下ロート付)に入れ、さらにこの溶液に、150gの原料アニリンを加え、攪拌溶解した。冷媒によるフラスコの攪拌冷却を開始し、1N塩酸12Lを溶液に添加した。次に、溶液温度が-3℃に冷却した状態で、214gの過硫酸アンモニウムを1N塩酸4Lに溶解した溶液を滴下ロートで滴下し、3時間10分で完了した。滴下開始から18時間30分の間、溶液内温を0℃±1℃に保ったまま攪拌を行った。その後、トルエン8Lを加え、溶液温度を19℃に上昇させ静置した。静置により二相に分離した水相(下相)を反応器下部から抜き出し、粗ポリアニリン複合体トルエン溶液を得た。
炭素61.7重量%、水素:8.2重量%、窒素:3.9重量%、硫黄:5.5重量%
[プロトン化されたジ(2-エチルヘキシル)スルホコハク酸の製造]
ジ(2-エチルヘキシル)スルホコハク酸ナトリウム(東京化成工業製)4gを酢酸エチル100mlに攪拌溶解し、完全溶解したところに1N塩酸水溶液50mlを加え、30分攪拌を行った。攪拌後、反応液を分液ロートに移して有機相を取り出し、ジ(2-エチルヘキシル)スルホコハク酸溶液を得た。得られた溶液をエバポレートし、溶媒を除去することで、ジ(2-エチルヘキシル)スルホコハク酸を得た。
[ジ(2-エチルヘキシル)スルホコハク酸アニリン塩の製造]
製造例2で調製したジ(2-エチルヘキシル)スルホコハク酸1g(2.4mmol)に対してアニリン0.22g(2.4mmol)を添加し、10分攪拌することでジ(2-エチルヘキシル)スルホコハク酸アニリン塩を得た。
[プロトネーションされたモノイソプロピルナフタレンスルホン酸の製造]
ジ(2-エチルヘキシル)スルホコハク酸ナトリウムの代わりにモノイソプロピルナフタレンスルホン酸Na(竹本油脂社製)を用いた他は製造例2と同様にして、プロトネーションされたモノイソプロピルナフタレンスルホン酸を調製した。
[プロトネーションされたジイソプロピルナフタレンスルホン酸の製造] ジ(2-エチルヘキシル)スルホコハク酸ナトリウムの代わりにジイソプロピルナフタレンスルホン酸Na(竹本油脂社製)を用いた他は製造例2と同様にして、プロトネーションされたジイソプロピルナフタレンスルホン酸を調製した。
[プロトネーションされたトリイソプロピルナフタレンスルホン酸の製造]
ジ(2-エチルヘキシル)スルホコハク酸ナトリウムの代わりにトリイソプロピルナフタレンスルホン酸Na(竹本油脂社製)を用いた他は製造例2と同様にして、プロトネーションされたトリイソプロピルナフタレンスルホン酸を調製した。
[導電性ポリアニリン組成物の調製]
製造例1で得た導電性ポリアニリン複合体500mgをトルエン10gに再度溶解し、均一な導電性ポリアニリン複合体溶液を調製した。この溶液に製造例2で得たジ(2-エチルヘキシル)スルホコハク酸を0.01g(0.024mmol)及びm-クレゾール1g(9.2mmol)を添加し、均一な導電性ポリアニリン組成物を得た。
尚、下記の方法によると、ジ(2-エチルヘキシル)スルホコハク酸の酸性度(pKa)は-2.5であった。
「TURBOMOLE Version 6.1」(COSMO logic社製)を用いて、基底関数にTZVPを用いて構造を最適化し、この構造を用いてCOSMO-RS法計算を「COSMO therm Version C2.1 Release 01.10」(COSMO logic社製)により行った。ここで、「COSMO therm Version C2.1 Release 01.10」に25℃の水溶媒中との条件と、分子の化学式と、脱プロトンした分子の化学式と、を入力してpKaを算出した。
得られた導電性ポリアニリン組成物約1mlを、図1に示す、パターニングによりITO電極2が表面に形成されたガラス基板1の上面に塗布した。具体的には、スピンコート法により塗布した。ここでスピンコート法による塗布は、窒素雰囲気下で行った。また、スピンコート法の、ガラス基板に導電性ポリアニリン組成物を滴下した後のガラス基板回転時間は、15秒間であった。また、スピンコート法のガラス基板回転速度は、500rpmとした。その後、ガラス基板を乾燥して導電性ポリアニリン薄膜を形成した。ここで乾燥は、窒素雰囲気下で行った。また、乾燥時間は、5分間とした。また、乾燥温度は、80℃とした。導電性ポリアニリン薄膜を乾燥後、図4に示すように、導電性ポリアニリン薄膜5のITO電極の端子を覆っている部分を、窒素雰囲気下で削り取り、ITO電極の端子を表面に露出させた。表面に露出したITO電極の端子を用いて、ロレスターGP(三菱化学社製;四端子法による抵抗率計)を用いて薄膜の抵抗を測定し、成膜直後の抵抗値を初期値R0とした。
得られた導電性ポリアニリン組成物の薄膜をガラス基板のまま、窒素雰囲気下、105℃の条件下で所定時間放置した。所定時間経過後に薄膜の温度を室温に戻してから初期値R0の場合同様にして抵抗の測定を行った。所定時間経過後の抵抗値Rと初期値R0との比R/R0を算出し、薄膜の経時劣化(抵抗の上昇率)を評価した。結果を表1,2に示す。
尚、表中の値は、所定時間経過後の抵抗値Rと初期値R0との比R/R0を示す。
ジ(2-エチルヘキシル)スルホコハク酸の添加量を0.05g(0.12mmol)に変更した以外は実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の添加量を0.2g(0.47mmol)に変更した以外は実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の添加量を0.5g(1.18mmol)に変更した以外は実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例3で得たジ(2-エチルヘキシル)スルホコハク酸アニリン塩を0.01g(0.02mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例3で得たジ(2-エチルヘキシル)スルホコハク酸アニリン塩を0.05g(0.10mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例3で得たジ(2-エチルヘキシル)スルホコハク酸アニリン塩を0.1g(0.20mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例3で得たジ(2-エチルヘキシル)スルホコハク酸アニリン塩を0.2g(0.39mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例3で得たジ(2-エチルヘキシル)スルホコハク酸アニリン塩を0.5g(0.98mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにジ(2-エチルヘキシル)スルホコハク酸ナトリウム(東京化成工業製)0.1g(0.23mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにジ(2-エチルヘキシル)スルホコハク酸ナトリウム(東京化成工業(株)製)0.2g(0.45mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにジ(2-エチルヘキシル)スルホコハク酸ナトリウム(東京化成工業製)0.3g(0.68mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにメタンスルホン酸(和光純薬工業製)0.02g(0.1mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、メタンスルホン酸の酸性度(pKa)は-1.5であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにメタンスルホン酸(和光純薬工業製)0.04g(0.2mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにメタンスルホン酸(和光純薬工業製)0.08g(0.4mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに下記式で表されるドデシルリン酸(フォスファノールML200、第一工業製薬製)0.058g(0.21mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、ドデシルリン酸の酸性度(pKa)は0.84であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにドデシルリン酸(フォスファノールML200、第一工業製薬製)0.11g(0.43mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)0.094g(0.42mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、2-ナフタレンスルホン酸の酸性度(pKa)は-2.4であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)0.187g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにp-キシレンスルホン酸(東京化成工業株式会社製)0.08g(0.43mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、p-キシレンスルホン酸の酸性度(pKa)は-1.8であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにp-キシレンスルホン酸(東京化成工業株式会社製)0.16g(0.86mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに安息香酸(東京化成工業株式会社製)0.05g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、安息香酸の酸性度(pKa)は4.3であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに安息香酸(東京化成工業株式会社製)0.1g(0.82mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにシクロヘキサンカルボン酸(東京化成工業株式会社製)0.05g(0.39mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、シクロヘキサンカルボン酸の酸性度(pKa)は4.6であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにシクロヘキサンカルボン酸(東京化成工業株式会社製)0.1g(0.78mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにp-トルエンスルホン酸(東京化成工業株式会社製)0.08g(0.42mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、p-トルエンスルホン酸の酸性度(pKa)は-2.2であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにp-トルエンスルホン酸(東京化成工業株式会社製)0.16g(0.84mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにフェニルホスホン酸(東京化成工業株式会社製)0.065g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、フェニルホスホン酸の酸性度(pKa)は2.2であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにフェニルホスホン酸(東京化成工業株式会社製)0.13g(0.82mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにm-キシレンスルホン酸(東京化成工業株式会社製)0.08g(0.43mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表1,2に示す。
尚、実施例1と同様の方法によると、m-キシレンスルホン酸の酸性度(pKa)は-2.0であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにm-キシレンスルホン酸(東京化成工業株式会社製)0.16g(0.86mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにエタンスルホン酸(東京化成工業株式会社製)0.046g(0.42mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、エタンスルホン酸の酸性度(pKa)は-2.6であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにエタンスルホン酸(東京化成工業株式会社製)0.091g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにドデシルベンゼンスルホン酸(東京化成工業株式会社製)0.135g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、ドデシルベンゼンスルホン酸の酸性度(pKa)は-2.8であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにドデシルベンゼンスルホン酸(東京化成工業株式会社製)0.27g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにリン酸水素ビス(2-エチルヘキシル)(東京化成工業株式会社製)0.13g(0.40mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、リン酸水素ビス(2-エチルヘキシル)の酸性度(pKa)は1.2であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにリン酸水素ビス(2-エチルヘキシル)(東京化成工業株式会社製)0.27g(0.84mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに8-エトキシキノリン-5-スルホン酸水和物(東京化成工業株式会社製)0.10g(0.40mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、8-エトキシキノリン-5-スルホン酸の酸性度(pKa)は-2.1であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに8-エトキシキノリン-5-スルホン酸水和物(東京化成工業株式会社製)0.21g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに2-エチルヘキサン酸(東京化成工業株式会社製)0.06g(0.42mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、2-エチルヘキサン酸の酸性度(pKa)は4.5であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに2-エチルヘキサン酸(東京化成工業株式会社製)0.12g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにサリチル酸(東京化成工業株式会社製)0.06g(0.43mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、サリチル酸の酸性度(pKa)は3.5であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにサリチル酸(東京化成工業株式会社製)0.12g(0.87mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにウンデシレン酸(東京化成工業株式会社製)0.075g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、ウンデシレン酸の酸性度(pKa)は4.6であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにウンデシレン酸(東京化成工業株式会社製)0.15g(0.81mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにp-フェノールスルホン酸(東京化成工業株式会社製)0.072g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、p-フェノールスルホン酸の酸性度(pKa)は-2.2であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにp-フェノールスルホン酸(東京化成工業株式会社製)0.144g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにアントラキノンスルホン酸Na(東京化成工業株式会社製)0.129g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、アントラキノンスルホン酸の酸性度(pKa)は-0.89であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりにアントラキノンスルホン酸Na(東京化成工業株式会社製)0.259g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例4で得たモノイソプロピルナフタレンスルホン酸0.104g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、モノイソプロピルナフタレンスルホン酸の酸性度(pKa)は-2.3であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例4で得たモノイソプロピルナフタレンスルホン酸0.207g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例5で得たジイソプロピルナフタレンスルホン酸0.121g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、ジイソプロピルナフタレンスルホン酸の酸性度(pKa)は-2.2であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例5で得たジイソプロピルナフタレンスルホン酸0.242g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例6で得たトリイソプロピルナフタレンスルホン酸0.139g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、トリイソプロピルナフタレンスルホン酸の酸性度(pKa)は-2.1であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに製造例6で得たトリイソプロピルナフタレンスルホン酸0.277g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに3-ニトロベンゼンスルホン酸(東京化成工業株式会社製)0.084g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、3-ニトロベンゼンスルホン酸の酸性度(pKa)は-2.8であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに3-ニトロベンゼンスルホン酸(東京化成工業株式会社製)0.168g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに2-ヒドロキシ-4-ベンゾフェノン-5-スルホン酸(東京化成工業株式会社製)0.128g(0.41mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
尚、実施例1と同様の方法によると、2-ヒドロキシ-4-ベンゾフェノン-5-スルホン酸の酸性度(pKa)は-2.8であった。
ジ(2-エチルヘキシル)スルホコハク酸の代わりに2-ヒドロキシ-4-ベンゾフェノン-5-スルホン酸(東京化成工業株式会社製)0.255g(0.83mmol)を添加した他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸を添加しなかった他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
ジ(2-エチルヘキシル)スルホコハク酸及びm-クレゾールを添加しなかった他は、実施例1と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例1と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表3,4に示す。
[導電性ポリアニリン組成物の調製]
製造例1で得た導電性ポリアニリン複合体500mgをトルエン10gに再度溶解し、均一な導電性ポリアニリン複合体溶液を調製した。この溶液に2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)を0.023g(0.11mmol)及びm-クレゾール1g(9.2mmol)を添加し、均一な導電性ポリアニリン組成物を得た。
得られた導電性ポリアニリン組成物約1mlを、図1に示す、パターニングによりITO電極2が表面に形成されたガラス基板1の上面に塗布した。具体的には、スピンコート法により塗布した。ここでスピンコート法による塗布は、窒素雰囲気下で行った。また、スピンコート法の、ガラス基板に導電性ポリアニリン組成物を滴下した後のガラス基板回転時間は、15秒間とした。また、スピンコート法のガラス基板回転速度は、500rpmとした。その後、ガラス基板を乾燥して導電性ポリアニリン薄膜を形成した。ここで乾燥は、窒素雰囲気下で行った。また、乾燥時間は、5分間とした。また、乾燥温度は、80℃とした。導電性ポリアニリン薄膜を乾燥後、図4に示すように、導電性ポリアニリン薄膜5のITO電極の端子を覆っている部分を、窒素雰囲気下で削り取り、ITO電極の端子を表面に露出させた。表面に露出したITO電極の端子を用いて、ロレスターGP(三菱化学社製;四端子法による抵抗率計)を用いて薄膜の抵抗を測定し、成膜直後の抵抗値を初期値R0とした。
得られた導電性ポリアニリン組成物の薄膜をガラス基板のまま、窒素雰囲気下、125℃の条件下で所定時間放置した。所定時間経過後に薄膜の温度を室温に戻してから初期値R0の場合同様にして抵抗の測定を行った。所定時間経過後の抵抗値Rと初期値R0との比R/R0を算出し、薄膜の経時劣化(抵抗の上昇率)を評価した。結果を表5,6に示す。
尚、表中の値は、所定時間経過後の抵抗値Rと初期値R0との比R/R0を示す。
2-ナフタレンスルホン酸水和物の添加量を0.047g(0.22mmol)とした他は実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物の添加量を0.094g(0.42mmol)とした他は実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物の添加量を0.38g(1.8mmol)とした他は実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにp-キシレンスルホン酸(東京化成工業株式会社製)0.02g(0.11mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにp-キシレンスルホン酸(東京化成工業株式会社製)0.04g(0.22mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにp-キシレンスルホン酸(東京化成工業株式会社製)0.08g(0.43mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにp-キシレンスルホン酸(東京化成工業株式会社製)0.46g(2.5mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに安息香酸(東京化成工業株式会社製)0.2g(1.6mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに安息香酸(東京化成工業株式会社製)0.4g(3.2mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにp-フェノールスルホン酸(東京化成工業株式会社製)0.072g(0.41mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにp-フェノールスルホン酸(東京化成工業株式会社製)0.144g(0.83mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにアントラキノンスルホン酸Na(東京化成工業株式会社製)0.13g(0.42mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにアントラキノンスルホン酸Na(東京化成工業株式会社製)0.26g(0.83mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに製造例4で得たモノイソプロピルナフタレンスルホン酸0.104g(0.41mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに製造例4で得たモノイソプロピルナフタレンスルホン酸0.207g(0.83mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに製造例5で得たジイソプロピルナフタレンスルホン酸0.121g(0.41mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに製造例5で得たジイソプロピルナフタレンスルホン酸0.242g(0.83mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに製造例6で得たトリイソプロピルナフタレンスルホン酸0.139g(0.41mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりに製造例6で得たトリイソプロピルナフタレンスルホン酸0.277g(0.83mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにサリチル酸(東京化成工業株式会社製)0.06g(0.43mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物(東京化成工業株式会社製)の代わりにサリチル酸(東京化成工業株式会社製)0.12g(0.87mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物の代わりに3-ニトロベンゼンスルホン酸(東京化成工業株式会社製)0.084g(0.45mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
2-ナフタレンスルホン酸水和物の代わりに3-ニトロベンゼンスルホン酸(東京化成工業株式会社製)0.168g(0.90mmol)を添加した他は、実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果を表5,6に示す。
比較例3
2-ナフタレンスルホン酸水和物を添加しなかった他は実施例60と同様にして均一な導電性ポリアニリン組成物を調製した。
得られた導電性ポリアニリン組成物を用いて、実施例60と同様にして薄膜を形成し、得られた薄膜を評価した。結果は、経過日数1日でのR/Roの値が2、経過日数4日でのR/Roの値が8、経過日数6日でのR/Roの値が26、経過日数8日でのR/Roの値が38、経過日数11日でのR/Roの値が88、経過日数13日でのR/Roの値が150、経過日数14日でのR/Roの値が196、経過日数15日でのR/Roの値が246、経過日数21日でのR/Roの値が930、経過日数22日でのR/Roの値が1126、
経過日数26日でのR/Roの値が2149、経過日数27日でのR/Roの値が2551、経過日数28日でのR/Roの値が3394、であった。
この明細書に記載の文献の内容を全てここに援用する。
Claims (28)
- (a)溶剤、
(b)前記溶剤に溶解した、ドーパントによりドープされたπ共役高分子、
(c)酸性物質及び酸性物質の塩の少なくとも1つ、並びに
(d)フェノール性化合物を含むπ共役高分子組成物であって、
前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質と前記フェノール性化合物が異なり、前記酸性物質の塩のみを含む場合、前記酸性物質の塩と前記フェノール性化合物が異なり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つとフェノール性化合物が異なるπ共役高分子組成物。 - 少なくとも下記(a)~(d)を原料として用いるπ共役高分子組成物。
(a)溶剤
(b)前記溶剤に溶解する、ドーパントによりドープされたπ共役高分子
(c)酸性物質及び酸性物質の塩の少なくとも1つ
(d)フェノール性化合物
(前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質と前記フェノール性化合物が異なり、前記酸性物質の塩のみを含む場合、前記酸性物質の塩と前記フェノール性化合物が異なり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つとフェノール性化合物が異なる。) - 前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質は前記ドーパントと異なり、前記酸性物質の塩のみを含む場合、前記酸性物質の塩は前記ドーパントと異なり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つと前記ドーパントが異なる請求項1又は2に記載のπ共役高分子組成物。
- 前記成分(c)のうち、前記酸性物質のみを含む場合、前記酸性物質の酸性度が5.0以下であり、前記酸性物質の塩のみを含む場合、酸性度が5.0以下である酸性物質の塩であり、前記酸性物質及び前記酸性物質の塩の両方を含む場合、前記酸性物質の酸性度が5.0以下であること及び前記酸性物質の塩が酸性度が5.0以下の酸性物質の塩であることのうち、少なくとも1つの条件を満たす請求項1~3のいずれかに記載のπ共役高分子組成物。
- 前記π共役高分子が窒素原子を含み、
前記ドーパントがスルホン酸であり、
前記成分(c)のうち、前記酸性物質のみを含む場合、その酸性物質が前記ドーパントと同一又は異なるスルホン酸であり、
前記酸性物質の塩のみを含む場合、その酸性物質の塩が前記ドーパントと同一又は異なるスルホン酸の塩であり、
前記酸性物質及び前記酸性物質の塩を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つが前記ドーパントと同一又は異なるスルホン酸又はスルホン酸の塩であり、
式(1)を満たす請求項1又は2に記載のπ共役高分子組成物。
0.21≦S1/N1≦1.2…(1)
(式中、S1は組成物に含まれる硫黄原子のモル数であり、N1は組成物に含まれる窒素原子のモル数である。) - 前記π共役高分子が窒素原子を含み、
前記ドーパントがスルホン酸であり、
前記成分(c)のうち、前記酸性物質のみを含む場合、その酸性物質が前記ドーパントと同一又は異なるスルホン酸であり、
前記酸性物質の塩のみを含む場合、その酸性物質の塩が前記ドーパントと同一又は異なるスルホン酸の塩であり、
前記酸性物質及び前記酸性物質の塩を含む場合、前記酸性物質及び前記酸性物質の塩の少なくとも1つが前記ドーパントと同一又は異なるスルホン酸又はスルホン酸の塩であり、
前記酸性物質のみを含む場合には、式(2)を満たし、
前記酸性物質の塩のみを含む場合には、式(3)を満たし、
前記酸性物質及び前記酸性物質の塩を含む場合、式(4)を満たす請求項1,2又は5に記載のπ共役高分子組成物。
0.01≦S2/N2≦0.5…(2)
0.01≦S3/N3≦0.5…(3)
0.01≦S4/N4≦0.5…(4)
(式中、S2は組成物に含まれる全ての酸性物質の硫黄原子のモル数の合計であり、S3は組成物に含まれている全ての酸性物質の塩の硫黄原子のモル数の合計であり、S4は組成物に含まれている全ての酸性物質及び酸性物質の塩の硫黄原子のモル数の合計であり、N2~N4は組成物に含まれる全てのドーパントによりドープされたπ共役高分子の窒素原子のモル数の合計である。) - 前記π共役高分子が窒素原子を含み、前記ドーパントがスルホン酸であり、式(5)を満たす請求項1~6のいずれかに記載のπ共役高分子組成物。
0.2≦S5/N5≦0.7…(5)
(S5は組成物に含まれる前記ドーパントによりドープされたπ共役高分子の硫黄原子のモル数の合計であり、N5は組成物に含まれる前記ドーパントによりドープされたπ共役高分子の窒素原子のモル数の合計である。) - 前記ドーパントによりドープされたπ共役高分子の成形体の、4端子法により測定した導電度が0.01S/cm以上であり、
前記成形体は、パターニングされたインジウム錫酸化物電極を形成したガラス基板上に、窒素雰囲気下で前記ドーパントによりドープされたπ共役高分子500mgをトルエン10gに溶解した溶液1mlを、500rpmで15秒間スピンコート法により塗布し、窒素雰囲気下80℃で5分間乾燥して得られたものである請求項1~7のいずれかに記載のπ共役高分子組成物。 - 成形体の4端子法により測定した導電度が0.01S/cm以上であり、
前記成形体は、パターニングされたインジウム錫酸化物電極を形成したガラス基板上に、窒素雰囲気下で前記π共役高分子組成物1mlを500rpmで15秒間スピンコート法により塗布し、窒素雰囲気下80℃で5分間乾燥して得られたものである請求項1~8のいずれかに記載のπ共役高分子組成物。 - 前記ドープされたπ共役高分子が、プロトネーションされた置換又は非置換ポリアニリンである請求項1~9のいずれかに記載のπ共役高分子組成物。
- 前記ドープされたπ共役高分子が、スルホン酸でプロトネーションされた置換又は非置換ポリアニリンである請求項1~10のいずれかに記載のπ共役高分子組成物。
- 前記スルホン酸が下記式(III)で示されるスルホコハク酸誘導体である請求項11に記載のπ共役高分子組成物。
M(O3SCH(CH2COOR12)COOR13)m (III)
(式(III)において、
Mは、水素原子、有機遊離基又は無機遊離基であり、
mはMの価数であり、
R12及びR13は、それぞれ独立して炭化水素基又は-(R14O)r-R15で表される基であり、R14は炭化水素基又はシリレン基であり、R15は水素原子、炭化水素基又はR16 3Si-で表される基であり、R16は炭化水素基であり、3つのR16は同一又は異なっていてもよく、rは1以上の整数である。) - 前記酸性物質が有機酸である請求項1~12のいずれかに記載のπ共役高分子組成物。
- 前記酸性物質又は前記酸性物質の塩が、スルホン酸基、リン酸基、ホスホン酸基及びカルボキシ基から選択される1つ以上の酸性基を有する1つ以上の酸性物質又は酸性物質の塩である請求項1~13のいずれかに記載のπ共役高分子組成物。
- 前記酸性物質が、酸性基を1つ以上有する環状、鎖状又は分岐のアルキル酸である請求項1~14のいずれかに記載のπ共役高分子組成物。
- 前記酸性物質が、酸性基を1つ以上有する置換又は無置換の芳香族酸である請求項1~15のいずれかに記載のπ共役高分子組成物。
- 前記芳香族酸が、ナフタレン骨格を有する酸である請求項16に記載のπ共役高分子組成物。
- 請求項1~20のいずれかに記載のπ共役高分子組成物を含むコンデンサ。
- 請求項1~20のいずれかに記載のπ共役高分子組成物を用いて製造されるコンデンサ。
- 請求項1~20のいずれかに記載のπ共役高分子組成物を成形してなる導電性成形体。
- 請求項1~20のいずれかに記載のπ共役高分子組成物を成膜してなる導電性フィルム。
- 請求項1~20のいずれかに記載のπ共役高分子組成物を、基材に塗布してなる表面導電性物品。
- 前記基材が樹脂フィルムである請求項25に記載の表面導電性物品。
- 請求項1~20のいずれかに記載のπ共役高分子組成物と基材を混合してなる導電性物品。
- ドープされたπ共役高分子、及び酸性物質又は酸性物質の塩を含み、
前記ドープされたπ共役高分子が、スルホコハク酸誘導体でプロトネーションされた置換又は非置換ポリアニリンであり、
前記酸性物質が、スルホン酸基を有し、ナフタレン骨格を有する酸であり、さらにフェノール性化合物を含むπ共役高分子組成物。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011518318A JP5839683B2 (ja) | 2009-06-12 | 2010-06-14 | π共役高分子組成物 |
| KR1020167017548A KR101658760B1 (ko) | 2009-06-12 | 2010-06-14 | π 공액 고분자 조성물 |
| CN201080025937.4A CN102803388B (zh) | 2009-06-12 | 2010-06-14 | π共轭高分子组合物 |
| EP10785981.1A EP2441802B1 (en) | 2009-06-12 | 2010-06-14 | Ii-conjugated polymer composition |
| US13/377,672 US8802761B2 (en) | 2009-06-12 | 2010-06-14 | π-conjugated polymer composition |
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009140715 | 2009-06-12 | ||
| JP2009-140715 | 2009-06-12 | ||
| JP2009204330 | 2009-09-04 | ||
| JP2009-204330 | 2009-09-04 | ||
| JP2009-294855 | 2009-12-25 | ||
| JP2009294855 | 2009-12-25 | ||
| JP2010-066496 | 2010-03-23 | ||
| JP2010066496 | 2010-03-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010143450A1 true WO2010143450A1 (ja) | 2010-12-16 |
Family
ID=43308710
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2010/003922 Ceased WO2010143450A1 (ja) | 2009-06-12 | 2010-06-14 | π共役高分子組成物 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8802761B2 (ja) |
| EP (1) | EP2441802B1 (ja) |
| JP (2) | JP5839683B2 (ja) |
| KR (2) | KR101658760B1 (ja) |
| CN (1) | CN102803388B (ja) |
| TW (2) | TWI599603B (ja) |
| WO (1) | WO2010143450A1 (ja) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102757611A (zh) * | 2011-04-26 | 2012-10-31 | 中央大学 | 导电高分子溶液及其制备方法 |
| JP2013095919A (ja) * | 2011-10-28 | 2013-05-20 | National Central Univ | ドープ形態の共役高分子膜の製造及び処理方法 |
| JP2015218217A (ja) * | 2014-05-15 | 2015-12-07 | リグナイト株式会社 | 球状フラン樹脂、球状フラン樹脂の製造方法、レジンコーテッドサンド |
| WO2016006251A1 (ja) * | 2014-07-11 | 2016-01-14 | 出光興産株式会社 | ポリアニリン複合体組成物の製造方法及びポリアニリン複合体組成物 |
| WO2017150407A1 (ja) * | 2016-02-29 | 2017-09-08 | 出光興産株式会社 | 導電性高分子組成物、導電性高分子含有多孔質体及びその製造方法並びに固体電解コンデンサ及びその製造方法 |
| JP2018131636A (ja) * | 2018-06-04 | 2018-08-23 | リグナイト株式会社 | 球状フラン樹脂の製造方法 |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP7020902B2 (ja) * | 2017-12-21 | 2022-02-16 | 出光興産株式会社 | 組成物、導電性膜、導電性膜の製造方法、及びコンデンサ |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04139257A (ja) * | 1990-09-28 | 1992-05-13 | Nitto Denko Corp | 噴霧可能帯電防止材組成物 |
| JPH07238149A (ja) | 1993-09-20 | 1995-09-12 | Korea Res Inst Of Chem Technol Juridical Found | 温度特性の向上された導電性高分子化合物 |
| JPH08231862A (ja) * | 1995-01-09 | 1996-09-10 | Internatl Business Mach Corp <Ibm> | 解凝集した導電性ポリマーの製造方法 |
| WO2005052058A1 (ja) | 2003-11-28 | 2005-06-09 | Idemitsu Kosan Co., Ltd. | 導電性ポリアニリン組成物、その製造方法及びそれからなる成形体 |
| JP2005526876A (ja) | 2002-03-01 | 2005-09-08 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | 添加剤を含有する有機導電性ポリマーの印刷 |
| JP2007224279A (ja) * | 2006-01-26 | 2007-09-06 | Fuji Xerox Co Ltd | 樹脂組成物、樹脂成形品 |
| WO2008018420A1 (en) * | 2006-08-10 | 2008-02-14 | Idemitsu Kosan Co., Ltd. | Conductive polyaniline composition and method for producing the same |
| WO2008038609A1 (fr) * | 2006-09-25 | 2008-04-03 | Idemitsu Kosan Co., Ltd. | Procédé de production d'un composite conducteur de polyaniline |
| JP2008260896A (ja) * | 2007-04-13 | 2008-10-30 | Yokohama Rubber Co Ltd:The | ポリアニリンおよびポリアニリンの製造方法と、それらを使用した導電性組成物、帯電防止塗料および帯電防止材 |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5804100A (en) | 1995-01-09 | 1998-09-08 | International Business Machines Corporation | Deaggregated electrically conductive polymers and precursors thereof |
| US6005070A (en) | 1995-05-30 | 1999-12-21 | International Business Machines Corporation | Methods of fabrication of deaggregated electrically conductive polymers and precursors thereof |
| KR970058474A (ko) * | 1995-12-20 | 1997-07-31 | 정몽원 | 전자파 차폐용 케이스 |
| CN1056165C (zh) * | 1996-12-25 | 2000-09-06 | 中国科学院长春应用化学研究所 | 掺杂态聚苯胺水溶液和水系分散液的制备 |
| US5990249A (en) | 1997-01-16 | 1999-11-23 | International Business Machines Corporation | Methods of fabrication of deaggregated electrically conductive polymers and precursors thereof |
| US6806349B2 (en) | 1999-01-12 | 2004-10-19 | International Business Machines Corporation | Methods of fabrication of deaggregated electrically conductive polymers and precursors thereof |
| JP2000269086A (ja) * | 1999-03-15 | 2000-09-29 | Hitachi Chem Co Ltd | 固体電解コンデンサ用電解質形成用組成物、該組成物を用いた固体電解コンデンサの製造方法及び固体電解コンデンサ |
| JP2001163960A (ja) * | 1999-12-10 | 2001-06-19 | Mitsui Chemicals Inc | 導電性高分子物質の製造方法および固体電解コンデンサ |
| CN1342646A (zh) * | 2000-09-13 | 2002-04-03 | 中国科学院化学研究所 | 一种掺杂剂及其制备方法和用途 |
| JP4139257B2 (ja) | 2003-04-01 | 2008-08-27 | 日本電信電話株式会社 | 無線パケット通信方法および無線パケット通信装置 |
| JP2005139262A (ja) * | 2003-11-05 | 2005-06-02 | Shin Etsu Polymer Co Ltd | 導電性組成物、導電性塗料及び導電性樹脂 |
| US20090011226A1 (en) * | 2005-05-27 | 2009-01-08 | Idemitsu Kosan Co., Ltd | Conductive Polymer Multilayer Body |
| JP5323302B2 (ja) * | 2005-08-10 | 2013-10-23 | 信越ポリマー株式会社 | 導電性高分子塗料の製造方法および導電性塗膜の製造方法 |
| JP5192953B2 (ja) * | 2007-09-14 | 2013-05-08 | 三洋化成工業株式会社 | 磁気ディスク用ガラス基板洗浄剤 |
| WO2009084419A1 (ja) * | 2007-12-27 | 2009-07-09 | Idemitsu Kosan Co., Ltd. | ポリアニリン複合体、その組成物及び成形体 |
-
2010
- 2010-06-14 KR KR1020167017548A patent/KR101658760B1/ko active Active
- 2010-06-14 TW TW105124498A patent/TWI599603B/zh active
- 2010-06-14 EP EP10785981.1A patent/EP2441802B1/en active Active
- 2010-06-14 WO PCT/JP2010/003922 patent/WO2010143450A1/ja not_active Ceased
- 2010-06-14 TW TW099119338A patent/TWI551637B/zh active
- 2010-06-14 CN CN201080025937.4A patent/CN102803388B/zh active Active
- 2010-06-14 KR KR1020117029456A patent/KR101636837B1/ko active Active
- 2010-06-14 JP JP2011518318A patent/JP5839683B2/ja active Active
- 2010-06-14 US US13/377,672 patent/US8802761B2/en active Active
-
2015
- 2015-06-29 JP JP2015129691A patent/JP6069420B2/ja active Active
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH04139257A (ja) * | 1990-09-28 | 1992-05-13 | Nitto Denko Corp | 噴霧可能帯電防止材組成物 |
| JPH07238149A (ja) | 1993-09-20 | 1995-09-12 | Korea Res Inst Of Chem Technol Juridical Found | 温度特性の向上された導電性高分子化合物 |
| JPH08231862A (ja) * | 1995-01-09 | 1996-09-10 | Internatl Business Mach Corp <Ibm> | 解凝集した導電性ポリマーの製造方法 |
| JP2005526876A (ja) | 2002-03-01 | 2005-09-08 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | 添加剤を含有する有機導電性ポリマーの印刷 |
| WO2005052058A1 (ja) | 2003-11-28 | 2005-06-09 | Idemitsu Kosan Co., Ltd. | 導電性ポリアニリン組成物、その製造方法及びそれからなる成形体 |
| JP2007224279A (ja) * | 2006-01-26 | 2007-09-06 | Fuji Xerox Co Ltd | 樹脂組成物、樹脂成形品 |
| WO2008018420A1 (en) * | 2006-08-10 | 2008-02-14 | Idemitsu Kosan Co., Ltd. | Conductive polyaniline composition and method for producing the same |
| WO2008038609A1 (fr) * | 2006-09-25 | 2008-04-03 | Idemitsu Kosan Co., Ltd. | Procédé de production d'un composite conducteur de polyaniline |
| JP2008260896A (ja) * | 2007-04-13 | 2008-10-30 | Yokohama Rubber Co Ltd:The | ポリアニリンおよびポリアニリンの製造方法と、それらを使用した導電性組成物、帯電防止塗料および帯電防止材 |
Non-Patent Citations (1)
| Title |
|---|
| JOURNAL OF PHYSICAL CHEMISTRY, vol. 99, 1995, pages 2224 |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102757611A (zh) * | 2011-04-26 | 2012-10-31 | 中央大学 | 导电高分子溶液及其制备方法 |
| US9437345B2 (en) | 2011-10-28 | 2016-09-06 | National Central University | Methods for fabricating and treating doped conjugated polymer film |
| JP2013095919A (ja) * | 2011-10-28 | 2013-05-20 | National Central Univ | ドープ形態の共役高分子膜の製造及び処理方法 |
| JP2015218217A (ja) * | 2014-05-15 | 2015-12-07 | リグナイト株式会社 | 球状フラン樹脂、球状フラン樹脂の製造方法、レジンコーテッドサンド |
| JPWO2016006251A1 (ja) * | 2014-07-11 | 2017-04-27 | 出光興産株式会社 | ポリアニリン複合体組成物の製造方法及びポリアニリン複合体組成物 |
| KR20170032230A (ko) | 2014-07-11 | 2017-03-22 | 이데미쓰 고산 가부시키가이샤 | 폴리아닐린 복합체 조성물의 제조 방법 및 폴리아닐린 복합체 조성물 |
| WO2016006251A1 (ja) * | 2014-07-11 | 2016-01-14 | 出光興産株式会社 | ポリアニリン複合体組成物の製造方法及びポリアニリン複合体組成物 |
| US10872709B2 (en) | 2014-07-11 | 2020-12-22 | Idemitsu Kosan Co., Ltd. | Method for producing polyaniline complex composition and polyaniline complex composition |
| WO2017150407A1 (ja) * | 2016-02-29 | 2017-09-08 | 出光興産株式会社 | 導電性高分子組成物、導電性高分子含有多孔質体及びその製造方法並びに固体電解コンデンサ及びその製造方法 |
| KR20180118618A (ko) * | 2016-02-29 | 2018-10-31 | 이데미쓰 고산 가부시키가이샤 | 도전성 고분자 조성물, 도전성 고분자 함유 다공질체 및 그 제조 방법 그리고 고체 전해 콘덴서 및 그 제조 방법 |
| JPWO2017150407A1 (ja) * | 2016-02-29 | 2018-12-20 | 出光興産株式会社 | 導電性高分子組成物、導電性高分子含有多孔質体及びその製造方法並びに固体電解コンデンサ及びその製造方法 |
| US10975200B2 (en) | 2016-02-29 | 2021-04-13 | Idemitsu Kosan Co., Ltd. | Conductive polymer composition, conductive-polymer-containing porous body and manufacturing method therefor, and solid electrolytic capacitor and manufacturing method therefor |
| JP7153557B2 (ja) | 2016-02-29 | 2022-10-14 | 出光興産株式会社 | 導電性高分子組成物、導電性高分子含有多孔質体及びその製造方法並びに固体電解コンデンサ及びその製造方法 |
| KR102707123B1 (ko) * | 2016-02-29 | 2024-09-13 | 이데미쓰 고산 가부시키가이샤 | 도전성 고분자 조성물, 도전성 고분자 함유 다공질체 및 그 제조 방법 그리고 고체 전해 콘덴서 및 그 제조 방법 |
| JP2018131636A (ja) * | 2018-06-04 | 2018-08-23 | リグナイト株式会社 | 球状フラン樹脂の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| TWI599603B (zh) | 2017-09-21 |
| EP2441802A4 (en) | 2014-04-30 |
| JP5839683B2 (ja) | 2016-01-06 |
| TWI551637B (zh) | 2016-10-01 |
| US8802761B2 (en) | 2014-08-12 |
| JP2015199969A (ja) | 2015-11-12 |
| CN102803388B (zh) | 2016-03-16 |
| JPWO2010143450A1 (ja) | 2012-11-22 |
| KR20120028325A (ko) | 2012-03-22 |
| KR101636837B1 (ko) | 2016-07-07 |
| EP2441802B1 (en) | 2023-07-12 |
| TW201107391A (en) | 2011-03-01 |
| JP6069420B2 (ja) | 2017-02-01 |
| CN102803388A (zh) | 2012-11-28 |
| US20120135255A1 (en) | 2012-05-31 |
| EP2441802A1 (en) | 2012-04-18 |
| KR101658760B1 (ko) | 2016-09-21 |
| TW201704313A (zh) | 2017-02-01 |
| KR20160084484A (ko) | 2016-07-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6069420B2 (ja) | π共役高分子組成物 | |
| JP4959192B2 (ja) | 導電性ポリアニリン組成物、その製造方法及びそれからなる成形体 | |
| CN103328541B (zh) | 聚苯胺复合物、其制造方法及组合物 | |
| US10872709B2 (en) | Method for producing polyaniline complex composition and polyaniline complex composition | |
| JP5701761B2 (ja) | 導電性組成物 | |
| TWI588179B (zh) | Conductive polymer composition | |
| JP5710388B2 (ja) | ポリアニリン導電性組成物 | |
| JP5492413B2 (ja) | 導電性ポリアニリン組成物及びその製造方法 | |
| JP5608443B2 (ja) | 導電性組成物 | |
| JP5731974B2 (ja) | 導電性組成物 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080025937.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10785981 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011518318 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 20117029456 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010785981 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13377672 Country of ref document: US |