WO2005121025A1 - 金属酸化物ナノ多孔体、それを得るための被覆組成物、及びそれらの製造方法 - Google Patents
金属酸化物ナノ多孔体、それを得るための被覆組成物、及びそれらの製造方法 Download PDFInfo
- Publication number
- WO2005121025A1 WO2005121025A1 PCT/JP2005/011003 JP2005011003W WO2005121025A1 WO 2005121025 A1 WO2005121025 A1 WO 2005121025A1 JP 2005011003 W JP2005011003 W JP 2005011003W WO 2005121025 A1 WO2005121025 A1 WO 2005121025A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- metal
- oxide
- fixed point
- average
- Prior art date
Links
- 229910044991 metal oxide Inorganic materials 0.000 title claims abstract description 35
- 150000004706 metal oxides Chemical class 0.000 title claims abstract description 35
- 238000000034 method Methods 0.000 title claims description 129
- 239000008199 coating composition Substances 0.000 title 1
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N iron oxide Inorganic materials [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 claims abstract description 57
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims abstract description 11
- 229910052761 rare earth metal Inorganic materials 0.000 claims abstract description 7
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims abstract description 6
- 229910000272 alkali metal oxide Inorganic materials 0.000 claims abstract description 3
- 150000001875 compounds Chemical class 0.000 claims description 197
- 229910052751 metal Inorganic materials 0.000 claims description 147
- 239000002184 metal Substances 0.000 claims description 138
- 239000000463 material Substances 0.000 claims description 99
- 238000000576 coating method Methods 0.000 claims description 84
- 239000011248 coating agent Substances 0.000 claims description 83
- 239000000843 powder Substances 0.000 claims description 60
- 238000005259 measurement Methods 0.000 claims description 55
- 238000001465 metallisation Methods 0.000 claims description 49
- 238000002156 mixing Methods 0.000 claims description 36
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 30
- 239000002994 raw material Substances 0.000 claims description 26
- 239000004744 fabric Substances 0.000 claims description 23
- 150000002736 metal compounds Chemical class 0.000 claims description 21
- 239000000203 mixture Substances 0.000 claims description 20
- 241000894007 species Species 0.000 claims description 20
- 229910000510 noble metal Inorganic materials 0.000 claims description 19
- 229910001404 rare earth metal oxide Inorganic materials 0.000 claims description 19
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 18
- 150000003839 salts Chemical class 0.000 claims description 18
- 241000234282 Allium Species 0.000 claims description 16
- 235000002732 Allium cepa var. cepa Nutrition 0.000 claims description 16
- 229910052738 indium Inorganic materials 0.000 claims description 16
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 claims description 16
- 238000010438 heat treatment Methods 0.000 claims description 15
- 229910052742 iron Inorganic materials 0.000 claims description 15
- 229910052719 titanium Inorganic materials 0.000 claims description 15
- 239000010936 titanium Substances 0.000 claims description 15
- 238000010894 electron beam technology Methods 0.000 claims description 14
- 239000002244 precipitate Substances 0.000 claims description 10
- 238000004611 spectroscopical analysis Methods 0.000 claims description 9
- 238000001228 spectrum Methods 0.000 claims description 9
- 239000010409 thin film Substances 0.000 claims description 9
- 150000002739 metals Chemical class 0.000 claims description 8
- 239000010970 precious metal Substances 0.000 claims description 7
- -1 tita Chemical compound 0.000 claims description 7
- 230000005540 biological transmission Effects 0.000 claims description 6
- 239000011800 void material Substances 0.000 claims description 6
- 230000001133 acceleration Effects 0.000 claims description 5
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 claims description 5
- 150000002910 rare earth metals Chemical class 0.000 claims description 5
- 150000001540 azides Chemical class 0.000 claims description 4
- 241001071864 Lethrinus laticaudis Species 0.000 claims description 2
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 claims 1
- 241000255925 Diptera Species 0.000 claims 1
- 239000011232 storage material Substances 0.000 claims 1
- 230000009466 transformation Effects 0.000 claims 1
- 229910000287 alkaline earth metal oxide Inorganic materials 0.000 abstract 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 abstract 1
- 235000013980 iron oxide Nutrition 0.000 abstract 1
- VBMVTYDPPZVILR-UHFFFAOYSA-N iron(2+);oxygen(2-) Chemical class [O-2].[Fe+2] VBMVTYDPPZVILR-UHFFFAOYSA-N 0.000 abstract 1
- 239000011104 metalized film Substances 0.000 description 86
- 239000010408 film Substances 0.000 description 75
- 239000000243 solution Substances 0.000 description 52
- 238000001035 drying Methods 0.000 description 47
- 239000007788 liquid Substances 0.000 description 47
- 230000000052 comparative effect Effects 0.000 description 26
- 239000007789 gas Substances 0.000 description 19
- 239000002002 slurry Substances 0.000 description 19
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 17
- 229910052744 lithium Inorganic materials 0.000 description 17
- 239000000523 sample Substances 0.000 description 16
- 238000012360 testing method Methods 0.000 description 16
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 12
- 239000000047 product Substances 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- 239000002253 acid Substances 0.000 description 11
- 230000005484 gravity Effects 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 11
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 10
- 239000008267 milk Substances 0.000 description 10
- 210000004080 milk Anatomy 0.000 description 10
- 235000013336 milk Nutrition 0.000 description 10
- 239000002131 composite material Substances 0.000 description 9
- 238000004458 analytical method Methods 0.000 description 7
- 239000007769 metal material Substances 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 6
- 239000002585 base Substances 0.000 description 6
- 238000000975 co-precipitation Methods 0.000 description 6
- 150000004678 hydrides Chemical class 0.000 description 6
- 239000004615 ingredient Substances 0.000 description 6
- 229910052697 platinum Inorganic materials 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- 238000011156 evaluation Methods 0.000 description 5
- 239000012528 membrane Substances 0.000 description 5
- 238000001000 micrograph Methods 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- 229910052684 Cerium Inorganic materials 0.000 description 4
- GWXLDORMOJMVQZ-UHFFFAOYSA-N cerium Chemical compound [Ce] GWXLDORMOJMVQZ-UHFFFAOYSA-N 0.000 description 4
- 238000000635 electron micrograph Methods 0.000 description 4
- 150000004767 nitrides Chemical class 0.000 description 4
- 238000007254 oxidation reaction Methods 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 229910052705 radium Inorganic materials 0.000 description 4
- HCWPIIXVSYCSAN-UHFFFAOYSA-N radium atom Chemical compound [Ra] HCWPIIXVSYCSAN-UHFFFAOYSA-N 0.000 description 4
- 238000001179 sorption measurement Methods 0.000 description 4
- 241000283690 Bos taurus Species 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- QVQLCTNNEUAWMS-UHFFFAOYSA-N barium oxide Chemical compound [Ba]=O QVQLCTNNEUAWMS-UHFFFAOYSA-N 0.000 description 3
- 238000004364 calculation method Methods 0.000 description 3
- HTXDPTMKBJXEOW-UHFFFAOYSA-N dioxoiridium Chemical compound O=[Ir]=O HTXDPTMKBJXEOW-UHFFFAOYSA-N 0.000 description 3
- 238000002474 experimental method Methods 0.000 description 3
- 238000010304 firing Methods 0.000 description 3
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 3
- 229910052737 gold Inorganic materials 0.000 description 3
- 239000010931 gold Substances 0.000 description 3
- 150000004820 halides Chemical class 0.000 description 3
- 229910000457 iridium oxide Inorganic materials 0.000 description 3
- 229910052987 metal hydride Inorganic materials 0.000 description 3
- 150000004681 metal hydrides Chemical class 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 239000011148 porous material Substances 0.000 description 3
- 238000010298 pulverizing process Methods 0.000 description 3
- 239000010802 sludge Substances 0.000 description 3
- 229910052727 yttrium Inorganic materials 0.000 description 3
- VWQVUPCCIRVNHF-UHFFFAOYSA-N yttrium atom Chemical compound [Y] VWQVUPCCIRVNHF-UHFFFAOYSA-N 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 238000000441 X-ray spectroscopy Methods 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 229910052792 caesium Inorganic materials 0.000 description 2
- TVFDJXOCXUVLDH-UHFFFAOYSA-N caesium atom Chemical compound [Cs] TVFDJXOCXUVLDH-UHFFFAOYSA-N 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 235000013399 edible fruits Nutrition 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-M hydroxide Chemical compound [OH-] XLYOFNOQVPJJNP-UHFFFAOYSA-M 0.000 description 2
- 150000004694 iodide salts Chemical class 0.000 description 2
- 238000005272 metallurgy Methods 0.000 description 2
- 229910052762 osmium Inorganic materials 0.000 description 2
- SYQBFIAQOQZEGI-UHFFFAOYSA-N osmium atom Chemical compound [Os] SYQBFIAQOQZEGI-UHFFFAOYSA-N 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- 239000002893 slag Substances 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- RUDFQVOCFDJEEF-UHFFFAOYSA-N yttrium(III) oxide Inorganic materials [O-2].[O-2].[O-2].[Y+3].[Y+3] RUDFQVOCFDJEEF-UHFFFAOYSA-N 0.000 description 2
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 240000006108 Allium ampeloprasum Species 0.000 description 1
- 235000005254 Allium ampeloprasum Nutrition 0.000 description 1
- 241000731807 Anadia Species 0.000 description 1
- 241001040682 Anaides Species 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- GYHNNYVSQQEPJS-UHFFFAOYSA-N Gallium Chemical compound [Ga] GYHNNYVSQQEPJS-UHFFFAOYSA-N 0.000 description 1
- 244000287680 Garcinia dulcis Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 241001313637 Isotria Species 0.000 description 1
- 240000006550 Lantana camara Species 0.000 description 1
- 244000075898 Lantana strigocamara Species 0.000 description 1
- 241001579016 Nanoa Species 0.000 description 1
- 241000549556 Nanos Species 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 241000242583 Scyphozoa Species 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 241000270666 Testudines Species 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- 229910052770 Uranium Inorganic materials 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 239000010407 anodic oxide Substances 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- CETPSERCERDGAM-UHFFFAOYSA-N ceric oxide Chemical compound O=[Ce]=O CETPSERCERDGAM-UHFFFAOYSA-N 0.000 description 1
- 229910000422 cerium(IV) oxide Inorganic materials 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 238000005352 clarification Methods 0.000 description 1
- 238000002485 combustion reaction Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- KZHJGOXRZJKJNY-UHFFFAOYSA-N dioxosilane;oxo(oxoalumanyloxy)alumane Chemical compound O=[Si]=O.O=[Si]=O.O=[Al]O[Al]=O.O=[Al]O[Al]=O.O=[Al]O[Al]=O KZHJGOXRZJKJNY-UHFFFAOYSA-N 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000005684 electric field Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 210000000887 face Anatomy 0.000 description 1
- 210000003746 feather Anatomy 0.000 description 1
- 229910052733 gallium Inorganic materials 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- FUJCRWPEOMXPAD-UHFFFAOYSA-N lithium oxide Chemical compound [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 1
- 229910001947 lithium oxide Inorganic materials 0.000 description 1
- SWAIALBIBWIKKQ-UHFFFAOYSA-N lithium titanium Chemical compound [Li].[Ti] SWAIALBIBWIKKQ-UHFFFAOYSA-N 0.000 description 1
- 230000001050 lubricating effect Effects 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 229910052863 mullite Inorganic materials 0.000 description 1
- 210000001331 nose Anatomy 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- LZMJNVRJMFMYQS-UHFFFAOYSA-N poseltinib Chemical compound C1CN(C)CCN1C(C=C1)=CC=C1NC1=NC(OC=2C=C(NC(=O)C=C)C=CC=2)=C(OC=C2)C2=N1 LZMJNVRJMFMYQS-UHFFFAOYSA-N 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- CHWRSCGUEQEHOH-UHFFFAOYSA-N potassium oxide Chemical compound [O-2].[K+].[K+] CHWRSCGUEQEHOH-UHFFFAOYSA-N 0.000 description 1
- 229910001950 potassium oxide Inorganic materials 0.000 description 1
- 238000003825 pressing Methods 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 229910052703 rhodium Inorganic materials 0.000 description 1
- 239000010948 rhodium Substances 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 238000004621 scanning probe microscopy Methods 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 230000003068 static effect Effects 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 1
- XJDNKRIXUMDJCW-UHFFFAOYSA-J titanium tetrachloride Chemical compound Cl[Ti](Cl)(Cl)Cl XJDNKRIXUMDJCW-UHFFFAOYSA-J 0.000 description 1
- DNYWZCXLKNTFFI-UHFFFAOYSA-N uranium Chemical compound [U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U] DNYWZCXLKNTFFI-UHFFFAOYSA-N 0.000 description 1
- 238000004876 x-ray fluorescence Methods 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/38—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals
- B01J23/54—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of noble metals combined with metals, oxides or hydroxides provided for in groups B01J23/02 - B01J23/36
- B01J23/56—Platinum group metals
- B01J23/63—Platinum group metals with rare earths or actinides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/92—Chemical or biological purification of waste gases of engine exhaust gases
- B01D53/94—Chemical or biological purification of waste gases of engine exhaust gases by catalytic processes
- B01D53/9445—Simultaneously removing carbon monoxide, hydrocarbons or nitrogen oxides making use of three-way catalysts [TWC] or four-way-catalysts [FWC]
- B01D53/945—Simultaneously removing carbon monoxide, hydrocarbons or nitrogen oxides making use of three-way catalysts [TWC] or four-way-catalysts [FWC] characterised by a specific catalyst
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J23/00—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00
- B01J23/10—Catalysts comprising metals or metal oxides or hydroxides, not provided for in group B01J21/00 of rare earths
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/0215—Coating
- B01J37/0225—Coating of metal substrates
- B01J37/0226—Oxidation of the substrate, e.g. anodisation
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G23/00—Compounds of titanium
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G23/00—Compounds of titanium
- C01G23/003—Titanates
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G25/00—Compounds of zirconium
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G25/00—Compounds of zirconium
- C01G25/006—Compounds containing, besides zirconium, two or more other elements, with the exception of oxygen or hydrogen
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G49/00—Compounds of iron
- C01G49/009—Compounds containing, besides iron, two or more other elements, with the exception of oxygen or hydrogen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D1/00—Coating compositions, e.g. paints, varnishes or lacquers, based on inorganic substances
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20707—Titanium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/207—Transition metals
- B01D2255/20715—Zirconium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/20—Metals or compounds thereof
- B01D2255/209—Other metals
- B01D2255/2092—Aluminium
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/90—Physical characteristics of catalysts
- B01D2255/92—Dimensions
- B01D2255/9202—Linear dimensions
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2255/00—Catalysts
- B01D2255/90—Physical characteristics of catalysts
- B01D2255/92—Dimensions
- B01D2255/9207—Specific surface
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/30—Catalysts, in general, characterised by their form or physical properties characterised by their physical properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/0009—Use of binding agents; Moulding; Pressing; Powdering; Granulating; Addition of materials ameliorating the mechanical properties of the product catalyst
- B01J37/0027—Powdering
- B01J37/0036—Grinding
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/70—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data
- C01P2002/72—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data by d-values or two theta-values, e.g. as X-ray diagram
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/85—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by XPS, EDX or EDAX data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/04—Particle morphology depicted by an image obtained by TEM, STEM, STM or AFM
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/16—Pore diameter
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/10—Internal combustion engine [ICE] based vehicles
- Y02T10/12—Improving ICE efficiencies
Definitions
- the present invention relates to a metallization formed by two or more metallizations, a method for obtaining the metallization, and a method for producing them.
- metal such as CAM, such as ana, dia, and ceria, and platinum, , Radii and other precious metals are commonly used.
- the unmetallized material obtained by moisture has been generally used, for example, 0 82 55
- 20027997 (2) discloses a method of preparing a compound metal by coprecipitation from a solution of aum and / or rhodium and baking it in the atmosphere to obtain a powder of a composite metal compound. It has been done. Also, 733 (3) discloses a method of obtaining a composite metalized powder by mixing a cerium aqueous solution and an aqueous aluminum solution, adding an iron oxide powder to the mixture, followed by drying and drying. .
- the metallization obtained by using the liquid of the metallization of the metallization as it is is not uniform and does not have sufficient adhesiveness and thermal properties.
- the liquid of the metal is mixed at a high temperature and subjected to a heat treatment that substantially co-precipitates, and the surprisingly obtained metallized material has a diameter of less than zero and is composed of metal.
- High-level adherence to base materials such as high-density cams made of metal, which can improve adhesion and heat resistance to various types, and can also form thin films. They discovered that a coating having excellent thermal properties could be obtained in the past, and completed the process.
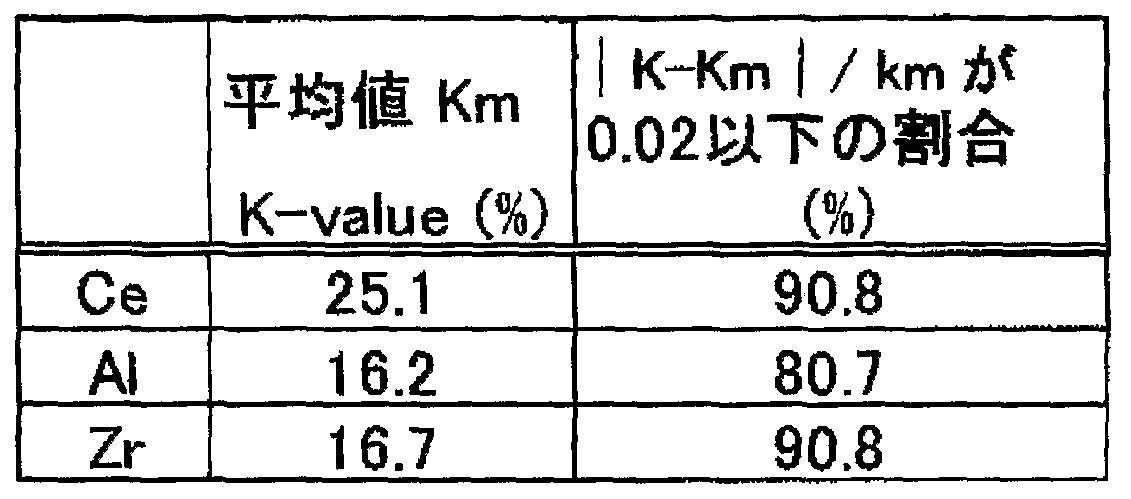
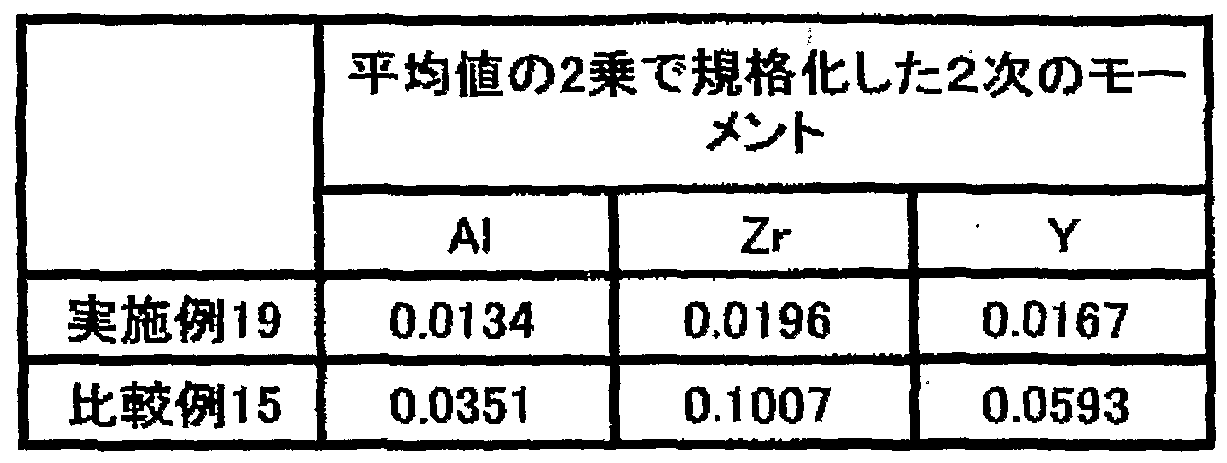
- the average value of n, the average value x, and the next measurement "2" are the following ()
- X is the average of the relative ratio X represented by x (X is the fixed point), and v is the next value around the average x represented by v ((XX) 2), “x 2 indicates a second-order metric of the average value x.
- the above-mentioned compound satisfies at least one of the following () and ().
- Acceleration voltage 5 and electron beam X-equivalent analyzer were used to analyze all elements belonging to 0a.
- the value represented by 5 is the following (3) at the measurement point 65 above the fixed point for all of the above-mentioned elements.
- the quadratic () obtained by the above is the following (7) at the fixed point 60 below the fixed point
- the light compound has a diameter of 5 or less, more preferably has a diameter of 5 or less.
- the light emitting device further comprises a light fluoride and a noble metal supported on the surface.
- the light compound may further include a powder of a second compound contained in the compound and having an average diameter of On, and the noble metal carried on the surface of the powder of the second compound. It is preferable to further provide
- It may be such a light-emitting compound or a powder.
- Ming hydride is a novel compound having the same component identity as that of the prior art, and can be obtained only by the following Ming method.
- Gold formed by two or more primary compounds selected from the group consisting of ana, dia, tita, iron oxide, rare earth oxides, acaides, and acaides The law of the compound,
- the material of the one fluoride is ana, dia, tita, iron oxide, rare earth oxide, alkali and hydride, and aluminum.
- it is at least a species selected from the group consisting of: chromium, indium, titanium, iron, rare earths, genus Aca and salts of the genus Aca.
- at least one species selected from the group consisting of rare earth oxides, oxides and iodides of iodides are: (a) adium, indium, tita, iron, rare earths, akali and Preferably, it is a five metal containing at least a species selected from the group consisting of Aca salts.
- the method of the present invention further includes the step of removing the P metal.
- the powder of the second compound having an average diameter of Onn5 may further be included in the above-mentioned compound.
- a step of removing the noble metal is further included. In such a case, the step of preparing a raw material containing the material of the second metallurgy is performed,
- the method further comprises a step of subjecting the mixture to a heat treatment without substantially coprecipitating after mixing under a strength of nnn to obtain the powder of the second compound.
- It is composed of two or more first compounds selected from the group consisting of ana, ana, dia, tita, iron oxide, rare earth oxide, acaide, and acaide, and has a diameter of less than zero. It is intended to obtain a metal compound in which the above-mentioned compound is dispersed in the material in the composition, and the raw material containing the material of the first compound described in the above 2 or more, and 0 It is characterized by being mixed under bravery.
- the material is composed of two or more first compounds selected from the group consisting of ana, dia, tita, iron oxide, rare earth oxide, acaide, and aide, and has a diameter. 0 to obtain a metallized material in which the compound is dispersed in the material
- the material of the one compound is ana, dia, tita, iron oxide, rare-earth element oxide, acide and aide of the compound, aum, Preferably, it is at least a species selected from the group consisting of um, titan, iron, rare earth, genus Aca and genus A.
- the surface of the powder of the second compound be coated with a noble metal.
- FIG. 2 is a graph showing an example of a good measurement result of the X skew.
- 3 is an electron microscope true showing an example of a place where good analysis was performed.
- Fig. 4 is a graph showing an example of a measurement result of the X-skew.
- Fig. 7 shows the results obtained in the constant straight line of the obtained metallized film. This is a graph showing cloth.
- Reference numeral 9 denotes a microscope showing the state of the surface of the metallized film obtained by comparison.
- 0 is a graph showing the actual data of the values on the constant line of the metal oxide film obtained by comparison.
- FIG. 9 is a graph showing the height of the obtained metallized film.
- R is a graph showing the data of the height () of the comparatively obtained metallized film.
- g F is a graph showing the cloth of height () in the metallized film obtained by comparison.
- FIG. 9 is a graph showing the secondary () cloth in the metallized film obtained by comparison.
- g R is a class showing high () data in the metallized film obtained by comparison.
- gn is a graph showing the height) cloth in the metal film obtained in Comparative n.
- R is a graph showing the of the measurement straight line portion directly above.
- a is an electron micrograph showing the surface of the metallized material (4 mm) obtained in the experiment.
- Fig. 7 is a graph showing the measurement straight line portion directly above the measurement lines shown in Figs. Is an electron microscope photograph showing the surface of the metallized material (4 mm) obtained in the comparison.
- Fig. 7 is a graph showing the measurement straight line portion directly above the measurement lines shown in Figs. n is the metal (4 mesh) obtained in comparison n.
- 1 and 2 are graphs showing the portion of the measurement straight line just above.
- Numeral 1 is an electron microscope which shows the position of the metallization obtained in the experiment.
- Numeral 1 is an electron micrograph showing the position of the metallization obtained in Example 4.
- 1 is a graph showing c of X obtained by the compound of the present invention.
- A is a graph showing the degree of X pics obtained for the compound of Comparative 4.
- A a graph showing the frequency ratio of X-pics obtained for the comparative compound.
- AQ is a microscope showing the state of the surface of the metal film obtained in the embodiment.
- 9 is a micrograph showing the state of the surface of the metallized film obtained in Example 2.
- Example 5 is a micrograph showing the state of the surface of the metal film obtained in Example 3.
- Reference numeral 52 is a graph showing the relationship between the degree of courage in the mixing process and the dispersibility of metallization.
- 53 is a graph showing the relationship between the courage and the dispersibility of metallization during the mixing process.
- Example 54 is an electron micrograph showing the position of the metallized film obtained in Example 4.
- Numeral 56 is an electron microscope showing the position of the metal film obtained in Example 4.
- Reference numeral 58 denotes a class indicating the ratio of X peaks obtained by the oxide film of Example 4.
- 60 is a graph showing the results of the thermal test (3 comparisons with 3).
- 6 is a graph showing the results of the thermal test (of 4 comparisons).
- 62 is a graph showing the results of the thermal test (of 8 comparisons).
- 63 is a graph showing the results of the sex test.
- 66 is a graph showing a metallized cloth with nitrogen adsorption.
- 67 is a graph showing a cloth of metallized X. Good for implementing
- the first compound to be specified is two or more metal compounds selected from the group consisting of ana, zirconia, titania, iron oxide, rare earth oxide, azide, and azide.
- the earth element oxides include oxides such as cerium, rata, neo, yttrium, and platinum.
- the acaides include oxides such as titanium, sodium, uranium, and cerium.
- the alkali include oxides such as lithium, strontium, um, and gallium.
- the composite metal compound is a composite metal compound containing at least two species selected from the group consisting of ana, dia, and sea atar.
- the points at which the obtained metallized sludge is useful as an exhaust gas are as follows: Tha, Ana, Anorantana, Zia Nora Tana, Zi Zia, Zi It A, Zi
- Ratanaso isotria, iron oxide, iron oxide, ceriano, iron oxide, dianoide, iron oxide are preferred, and iron oxide is particularly preferred.
- the fact that the obtained metal oxide sludge is suitable as a material for use in exhaust gas, especially for oxygen, includes anadium, ananodium, barium oxide, and lithium oxide.
- metal compound in such various kinds of mixed metal compounds, and the metal compound is adjusted accordingly.
- the nitride is made of the above-mentioned one compound, has a diameter below, and is characterized in that the compound is dispersed in the material in the constitution.
- the light metal has a very small diameter, lower diameter, preferably lower, more preferable lower, and thus has a higher adhesion to various metals than the higher metal oxide. Performance and thermal properties, as well as the area and the qualities of precious metals, as well as the performance as a catalyst.
- the above-mentioned compound is dispersed in the material when constituting. In other words, it is considered that two or more halides of the light nitride n are substantially uniformly dispersed (dispersed) in the atom, and are obtained by coprecipitation.
- the shape of the hydride is not particularly limited, and it may be a powder, a powder, or a molded product obtained by molding them into a predetermined shape.
- the thickness of the film is 300 degrees, and more preferably 50 degrees, because it is possible to form a good uniform coating. Further, as described above, since it is possible to form a coating made of a compound, it is possible to provide a high level of adhesion to a high-density cam, for which it was difficult to form a sufficient coating. From this viewpoint, it is particularly preferable that the thickness of the film is about 30 degrees.
- the diameter is particularly limited, and is adjusted accordingly. Generally, 0 to 0 to 5 is preferable. Metals with a diameter less than 0 are metal It is more difficult to manufacture with a powder of material, but on the other hand, only the coatings exceeding 50 tend to be very thick and have the problem of reduced adhesion.
- the light compound satisfies the condition () described below.
- the first compound (r is preferably an element belonging to the above-mentioned a) and the electron microscope (electron beam / electron microscope 0) is preferably used.
- the electron microscope electron beam / electron microscope 0
- a spectrum is obtained by onion X-ray spectroscopy, and the degree of X-pic of each element in the obtained skew is converted into a relative ratio.
- the mean value of the relative value of the relative ratio X and the next value around the average value x are the following
- a microscope with an electric field (G) and an accelerating voltage of 20 V is used.
- G electric field
- S scanning electron microscope
- X Donegis X-ray spectroscopy
- the area between 6 and the count of the X-rays is defined as the appropriate area for measurement, that is, the area where the sample can be determined I do.
- the signs of the X fruits found here include () a low physical number, 2) a low number of cows, especially in the onion area of oxygen X, and 3) a high number of copper compared to that. There is a characteristic. Microscope as an example of a place where such analysis cannot be performed (Shown from n to fixed point), and 2 shows an example of good measurement results of X-skew. This time, the measuring station
- Fig. 3 shows a microscopic microscope (showing, 005n fixed points) of an example of a place where good analysis was performed, and Fig. 4 shows an example of a measurement result of x-cut.
- the main metal element composing the material from the F-skut is a metal element belonging to 0a in the main material).
- the number of counts represented by () in the X 2) One of the elements of the genus is selected based on the two points of the two elements that are negligible in other elements. Then, determine the green onion ( ⁇ 2-0 ⁇ 3 e) that is enough to get the selected pic
- the total number of X-ray counts is added up, and this is taken as the degree of the X-pic of the genus element. Based on the result, the following ratio around the above-mentioned degree ratio X, its average value X, and the average value is obtained. Then, the secondary ment 2 having the average value is sequentially output, and it is determined whether or not the condition represented by () is satisfied.
- the compound of the formula satisfies the condition represented by) This indicates that the main metal oxides that make up the material are extremely dispersed in the composition. Such a high level of homogeneity is never achieved with a metallization that can be obtained as it is by using a liquid of the metallization-id metal, but is first achieved by the method described below. Surprisingly, in the case of a compound that satisfies the condition represented by the above (), the adhesion and heat resistance to various types are dramatically improved as compared with a compound that does not satisfy the condition.
- the light-emitting compound satisfies one of the following conditions ()))), and it is particularly preferable that the metal oxide obtained by using an id liquid as a raw material described below is used.
- the light compound satisfies the condition () described below.
- the light-emitting material be satisfied.Second (), in the above-mentioned element a, the acceleration voltage V and the range above 0 using the electron beam X-equivalent analyzer. (2)
- () means the average value at the fixed point.
- a portion corresponding to the crack formed in the above is excluded from the fixed points, and the above-mentioned is obtained.
- the method of recognizing the portion where the amount of the portion of the X-equalizer is raised by a predetermined amount is a method of recognizing the fissure by observation with an optical electron microscope. The method is preferably employed.
- the X-equilibrium analyzer used here may be any ordinary species, and the above-mentioned compounds are not particularly limited.
- the compound of formula (5) satisfies the condition () described below.
- the third () is to use a tip having a 5 for the scanning microscope (), and to piggy out the image of the surface at an interval of A.
- the fact that the light fluoride satisfies the condition of () above indicates that the high cloth is biased toward the small part and the surface is fine and convex. This indicates that the second-order n-number contains a relatively large value and the surface or the surface itself is not slippery, but the state of the surface convexity is much more detailed on the surface convexity.
- the surface of the metallized material which is obtained by the method described later, is never obtained with a metallized material that can be obtained as it is by using a liquid of the metallized material as it is, and is formed by the method described later. Surprisingly, the adhesiveness and heat resistance to various types are improved as compared to the compounds that do not satisfy the condition).
- the fluorinated compound satisfies the condition () described below.
- the light source is filled with a light fluoride. Satisfying the condition that the ratio of the length of the intersecting void (excluding) formed in the above is 0, particularly preferably 5) of the length of the constant line.
- the light compound satisfies the condition () described below.
- the luminous compound is satisfied with the fifth element A.
- the acceleration voltage, the electron beam, and the electron microscope are used. Calculate the skew by onion X spectroscopy and convert the X-pic degree of each element in the obtained skew into a relative ratio.
- the next item in the list of all the above elements is (8 0 ⁇ (8
- the metal oxide satisfies the above (V) indicates that the main metal oxides composing the metal are very uniformly dispersed in the metal oxide. This high level of homogeneity is not always possible with a metallization that can be obtained directly from a liquid of the metallization-id metal, but is first achieved by the method described below, and Surprisingly, in the compounds satisfying the above (), the adhesiveness and heat resistance to various types are improved as compared with the compounds not satisfying the requirement.
- the metallized metal satisfy the conditions of the above (1) to (V), and also satisfy the above two conditions after the conditions (1) and (V). I like it.
- the main metallizations that make up the material are (uniformly) distributed in a very uniform way.
- the adhesion and thermal properties of the metal to various types can be improved synergistically.
- the compound of the present invention is explained, but the precious metal may be further carried on the surface of the compound of the present invention.
- Such metals include platinum, indium, radium, osmium, indium, and gold.
- platinum, indium, and radium are preferred in that the resulting metallized material is useful as an exhaust gas.
- the amount of the metal is not particularly limited, and the obtained metal
- the amount is adjusted according to the temperature, but the amount is generally 0.0 ° with respect to the first compound.
- the powder of a second compound having an average diameter of 0.5 may be mixed.
- a second compound examples include ana, dia, sea, tita,, ratana, and ita, and those having a diameter of 0 are sufficient for production using a metal compound. Difficulty, on the other hand, tends to cause problems that the thickness of the coating exceeding the limit is very large and the adhesion is reduced.Also, although the amount of the second compound powder is not particularly limited, An amount of 30 to 7 degrees in the resulting metallization is preferred.
- the precious metal may be further carried at the end of the second compound.
- examples of such metals include platinum, indium, radium, osmium, indium, and gold.However, platinum, indium, and radium are useful in terms of the fact that the resulting metallized material is useful as an exhaust gas. Preferred.
- the amount of the metal is not particularly limited and is adjusted according to the degree of metallization to be obtained. However, the amount is generally from 0 to 0 degrees with respect to the second compound 00.
- the heat treatment is performed so as to substantially co-precipitate, and the compound is dispersed into the material in a configuration having a diameter of 0 or less.
- a raw material for obtaining the above-mentioned light-emitting compound wherein the raw material contains the material of the two or more first light-emitting materials. c It is characterized by being mixed under the above bravery.
- Ming a method for obtaining said chloride, comprising:
- Examples of the material of the above-mentioned one compound include: ana, dia, tita, iron oxide, rare earth oxide, iodide, iodide, adium, dia, tita, iron, iron, and the like. A few species selected from the group consisting of the rare earth, the genus Aca and the genus Aca are preferred.
- Id containing at least a species selected from the group consisting of ana, dia, titania, iron oxide, rare earth oxides, acaides, and acidoides
- A a metal containing at least a species selected from the group consisting of aum, indium, titanium, iron, rare earths, akali and genus aka;
- the metal oxide described herein is the above-described compound having a diameter of 520, and preferably a compound having a diameter of 5. If the diameter of the element is less than 5, it is difficult in principle to produce an element having a size smaller than that of the metal body. The problem of lower reactivity occurs.
- the shape of the element is not particularly limited, and examples include a child, a rod, a feather, a sphere, and an irregular shape.
- the medium for adjusting the degree of id is not particularly limited, and includes water, a, and the like, but is determined in consideration of a single chemical.
- halides may be further contained in the above-mentioned iodine solution as a hydrogen solution.
- a metal an acid salt, an acid chloride, an acid salt, a sulfate, or an inorganic material
- a constituent is preferably used by dissolving it in water, ammonia, or the like.
- the salt of the metal used as a raw material will be a component that becomes the above-mentioned compound by the treatment described below, that is, a group consisting of aum, um, titanium, iron, rare earth, genus Aca and genus Akari It is a salt of two or more metals selected from the above, and conforms to the above-mentioned two or more metals and their combinations.
- acid salts of the above genus Salts, chlorides, acid salts, sulfates, inorganics (e.g., aum, oxidium, cesium, indium, oxium, titanium tetrachloride, uchitaium, tita, yttrium), etc.
- inorganics e.g., aum, oxidium, cesium, indium, oxium, titanium tetrachloride, uchitaium, tita, yttrium
- a water-soluble salt of is preferably used.
- the metal liquid containing two or more golds to be disclosed there is no particular limitation, and examples thereof include water, water (e.g., methano, tano, and so on).
- water e.g., methano, tano, and so on.
- the metal to be clarified is not particularly limited, but the metal P is preferably 3.0 to 6.0 from the viewpoint that metal ions are more stably present in the liquid.
- the powder of the second compound having an average diameter of 0.05 or more may also be further contained.
- a compound similar to the above-mentioned compound is suitably used.
- a material obtained by pulverizing a liquid containing a metal as the second compound by oxidation after the second compound is preferably used. Further, it is desirable that only the coating having the following properties be used.
- the above-mentioned metal can be applied to its surface.
- a specific method for producing such a metal is not particularly limited, for example, the above-mentioned powder obtained by dissolving a noble metal (acid salt, chloride, acid salt) or a noble metal body in water, a, or the like is removed. After firing, the method of firing and firing is preferably used.
- the degree is preferably within 5 and n, and the condition is preferably about 3 to 60 at 25 to 30 C in an oxidized atmosphere (air, air). Such a metal process may be repeated until desired.
- the coating is obtained by mixing on the raw material nnns, as described above, preferably on O O Sec, particularly preferably on O os, and below.
- the degree of susceptibility is S
- the obtained metallized material does not satisfy the above-mentioned property, and the adhesiveness and thermal property are not improved.
- the degree of courage is not particularly limited
- the apparatus used here is not particularly limited as long as it can be mixed under the above conditions, but a naiser is preferably used.
- the mixing time at this level is not particularly limited, but is generally about 2 to 5 degrees.
- the () of the raw material to be mixed below this level is adjusted according to the (Miya) of the target metallized material, the cloth and the method of forming, but the degree of 550 ° is generally used. The preference is five degrees. gn
- the above-mentioned components are extremely uniformly dispersed.
- the heat treatment is carried out so as to substantially co-precipitate the material, thereby obtaining the above-mentioned compound, which has high adhesion to each of cam turtles and the like. It is possible to form a cover of a metal material having a high thermal property, and it is also possible to form a cover formed on a grave. For this reason, according to Ming, it is possible to form a metal covering that has a high level of adhesion and excellent thermal properties even on metal tombs such as metal cams and high-density cams. .
- qualitatively co-precipitating means that the elemental element of the raw material is converted into a metallized substance by heat treatment that does not substantially pass through a hydroxide, more specifically, before the treatment.
- the ratio of hydroxide in the original part is 5 (more preferably,
- the above-mentioned compound can be obtained by reducing the amount of the raw material by a certain treatment to form a metal compound.
- the mixture be mixed at the above-mentioned temperature and then be treated, and then be shorter, more preferably 0 or less, and more preferably less than 0. If the above-mentioned upper limit is exceeded during this period, the results are reduced, and the metallized substances are collected before the treatment and in the heat treatment step, and a metallized substance with sufficiently improved adhesion and thermal properties is obtained. become.
- n to Sn are used. And within n, especially within 5).
- high-speed drying tends to be sufficiently achieved.
- the degree of drying in the drying period is too rapid, and the degree of moisture is too fast than the degree, so that cracks, cracks, etc. It tends to cause a significant decrease in adhesion.
- the distance between n exceeds the above upper limit, the result is reduced, metal compounds are collected during the drying process, and a metal compound with sufficiently improved adhesion and heat property tends to be obtained.
- the metal content with insufficient adhesion and heat property tends to be obtained, while the above-mentioned upper limit is exceeded.On the other hand, the properties such as tagging due to high temperature and ambient atmosphere are exceeded. It tends to be easier.
- the time is above, is not sufficiently achieved, and a metalized material having sufficiently improved adhesion and heat property tends to be obtained.
- the above upper limit is exceeded, the result of decreases, and In this case, metallized materials tend to be collected, and a metallized material with sufficiently improved adhesion and heat property tends to be obtained.
- the material used here is not particularly limited, and is selected according to the type of metallization to be obtained.
- noses cam eater, high-density cam, etc.
- omfita pit, and put are preferably used.
- the quality is not particularly limited, when the gas is used as the gas n, it is preferable that the gas be made of a metal such as ceramics such as zirai, silicon carbide, or mullite, or a tester including coumium. Adopted to.
- a metal material if the surface is subjected to a treatment of about n to about n to form an oxidized surface, the adhesion tends to be further improved.
- the amount of the raw material to be applied in the light metal method is not particularly limited, and is adjusted according to the amount of the obtained metal material. However, the amount of the metal material constituting the material per volume unit is up to gnn degree. A certain amount is preferred.
- a specific method of forming the base is not particularly limited.
- a method based on the base or a method of attaching a surface to the step or the like is preferably used.
- the condition is that 9 n ⁇ in oxidation (for example, air) 3 6 at a time
- a method of heat-treating and mixing such that the above-mentioned compounds are mixed under the above-described temperature and substantially not coprecipitated is preferably adopted.
- the method of forming the metal is not particularly limited, but a method of obtaining the metallized product by pulverizing after drying as required is preferably used.
- the aforementioned metal may be applied to the surface of the metal oxide obtained above.
- Specific methods for forming the metal include, but are not particularly limited to, for example, the above-mentioned method in which a noble metal (acid salt, chloride, acid salt) or a noble metal body is dissolved in water, a The following method is preferably used.
- the condition for removing the metal in the step of removing the metal is as follows: "n is preferably within a degree, and the condition is about 250 in an oxidized atmosphere (for example, air). Also, the metal process may be repeated until desired.
- a light metal compound makes it possible to form a heat-resistant metal material cover with high adhesion to each of the cameras and the like. It is also possible to form a coating on the surface. Therefore, according to the present invention, it is possible to form a cover of a metal material having excellent thermal properties with a high level of adhesion even to a base such as a metal camfi and a high-density cam.
- the bright oxide can directly form a coating made of a metal oxide on various types of thin films.
- a metallized covering can be formed in various ways by the following method. That is, first, the slurs are obtained by mixing both of the light compounds under a strength of 0, more preferably under a strength of 0 OO sec. If the degree of decay is less than O sec, the resulting metallized material tends to have poor adhesion and thermal properties.
- the apparatus used here is not particularly limited as long as it can be mixed under the above conditions, but a nizer is preferably used.
- the mixing time under this level is not particularly limited, but is generally about 2 and preferably about 5 or less.
- the body of the light compound and both of the above-mentioned two compounds, which are about ⁇ 50, and the binder of the second compound as a binder are mixed. Is preferred.
- Examples of such a second liquid as an indica include a solution obtained by dissolving (acid, acid, hydrochloride, or the like) of aum, indium, cerium, titanium, or silicon in water, a, or the like.
- examples of the id of the third compound include ids of ana, dia, sea, tita, and the like.
- the amount of such an iron is not particularly limited, an amount of 30 degrees is preferable in the metallization constituting the coating from which the amount is obtained.
- the degree of () of the compound mixed at a low temperature in the above range is adjusted according to the intended coating, which is appropriate for the method, but a temperature of 206 ° is common.
- the medium for adjusting the degree of the mixture is not particularly limited, and includes water, a, and the like. Water is preferred.
- the obtained slurry is reduced to about 2 or less (for example, 20 OO) in order to sufficiently remove air bubbles mixed in the obtained slurry. You may give it further.
- the slurry obtained as described above is spread and then heat-treated to obtain a coating.
- a coating As used herein, there is no particular limitation, and the aforementioned is selected depending on the type of the obtained coating.
- the amount of slurry to be cloth is not particularly limited, and the resulting coating
- the amount of metallization constituting the coating is preferably n to 3 degrees with respect to the base volume.
- a specific method of fabricating the slag is not particularly limited, and for example, a method of forming the slag or a tin n method is preferably used on a surface of the slur by a sp.
- the step of heat-treating the slurried cloth is the same as the step of heat-treating the slurries described above. In this case, it is desirable that the time from obtaining the slurries to slurrying the slurries and treating the slurry is short. It is preferably within a degree, and more preferably within 3 degrees.
- the thickness of the coating made of a metal oxide formed using a light oxide is not particularly limited, and is adjusted accordingly. However, even in the case where the coating is thinned, a metal camfighter may be used. Since it is possible to form a single coating having excellent adhesion to each of the above, it is preferable that the coating has a temperature of up to 3 nO, more preferably 5 °, and F The degree is particularly preferred. Furthermore, according to the present invention, it is possible to form a coating made of a nitrided body, so that it is difficult to form a sufficient coating in the past. It is possible to form a coating having the following formula. From this viewpoint, it is particularly preferable that the thickness is approximately 3 to 3 degrees.
- a precious metal may be applied to the coated surface obtained in this way, This is the same process as the above-mentioned process of depositing a noble metal on the surface of a compound.
- the noble metal used in this process may be the same as or different from the noble metal carried by the metal. You may still return this until you have reached the desired value.
- the boxer used was a boxer (box type 2.5, manufactured by Tokushu Kogyo Co., Ltd.).
- the amount of metallization generated when drying, drying, and metallization is expressed as () with respect to.
- lantana a), ear (), potassium oxide (O), oxide a, iron oxide eO) Lantern, yttrium, potassium, and um were used, respectively. Then, the required amount of was collected and dissolved into a solution (a, Y,, a, e) to obtain a solution containing the required amount without performing solidity.
- a metal put (e0C, 50X5X03) consisting of a test tube containing coumium was used to evaluate the component identity, surface, adhesion and heat resistance.
- the intermediate treatment was performed in o to form oxidation on the surface. Also, to evaluate the cross-sectional properties, a 40-meter
- a dilite having a shape of 5 mm and a dilite having a shape of 5 mm were used.
- the density of eh group (e20C5) was used to evaluate the durability.
- the mixture was mixed and removed as about 20 (20) to obtain a coating.
- the coating obtained above was immediately added to the coating obtained above.
- the 0 Lloyd was heated to 150 C to remove the vapor, and further heated to 500 C to remove the components, and then crushed with milk to obtain a powder having an average diameter of about 150.
- the obtained 0-end is passed through a filter, it is collected, and the end is subjected to 60 in an air atmosphere at 300, and then crushed with milk to obtain an average of 70, and the attritor (
- the 0-end solution e obtained above was used to obtain e 35 of 0 e in the obtained coating. Then, the mixture was diluted with methano to prepare a 2-id solution. Then, the obtained id solution was mixed with a nailizer at a temperature of 20000 sec, and then mixed (about 20 m) and mixed to remove to obtain a coating.
- the 0-loyd solution and the e-solution were mixed to obtain 0 and 7 e A 0 Ce 35 R R in the coating to be obtained, and the mixture was diluted with methano 9 to prepare a high-aid solution. Then, the obtained solution A was mixed at a temperature of 20000 ec with a nailizer, and then mixed and removed as approximately gentle (20 p) to obtain a coating.
- the resulting coating was heated to 150C to remove steam, further gsped to 25 ° C to remove the components, and pulverized with milk to obtain a powder having an average diameter of about 70.
- the obtained result is passed through
- the powder was recovered, and the powder was subjected to 60C at 00C in an air atmosphere, and then pulverized with milk to obtain a powder having a mean diameter of about 7 (00 obtained from 3 of metal, All of the ana of 50) were mixed with) 00, the amide as an ida (520n, solid form 25) 009, and 509 were mixed to prepare a compound having a solidity of 50.
- the silicon is further attached to an attritor.
- a metal stamp which had been oxidized on its surface by performing a treatment at 000 ° C. was immediately taken out of the slurry obtained above, and the slurry on the surface was removed by gravity. Then, after placing the plate horizontally, drying at room temperature is 50, drying at 2-60 seconds (60-100 ° C) is 5-0, drying at 250 ° C in the air atmosphere is about 30, and the air speed is 2-10. It was relocated by 50m at 50m. After this treatment was repeated two times, a coating was obtained in which the metallization was reduced to about 0 5 with respect to the evaluation put sheets at 550 to 60 in an air atmosphere. Observation of the covering of the metalized material in the obtained coating with an electron microscope) revealed that the film had a thickness of 0, indicating that a very uniform metalized film was formed.
- the coating was obtained in the same manner as in Example 3 except that the gradual (S.) was used instead of the case with the Niza.
- the resulting coating is placed on a high-temperature putter heated to 35 ° C, dried (3 minutes), dried (metallized and removed), and then crushed with milk to reduce the average diameter to about 30%. 7 got the hydride (4)
- a metallized product was obtained in the same manner as in the above, except that the solution was changed to (0S.c) slowly instead of using a nailizer.
- the composite metallized product was obtained in the same manner as in Example 4 except for drying and drying at 5 ° C for 3 to 4 hours in an air atmosphere.
- a 55 is spread on a high-temperature putter heated to 335 C, dried in 3 minutes (removed) (removed and metal), and then crushed with milk to obtain an average diameter of about 5 Got.
- the obtained A powder was n-filtered by R and a, and recovered. The A powder was recovered at 30 ° C in an air atmosphere, and then pulverized with milk to obtain an average of 7, and the atrium () was used. Average adjusted to 2 powders (3 for metal
- the metal put which had been subjected to a treatment between 1 C and oxidized on its surface, was immediately taken out to the coating obtained above, and the surface was removed by gravity and by gravity.
- 6 was obtained at 5 C in an air atmosphere, and a coating as metallurgy was obtained for the evaluation put sheets.
- the resulting coating is coated on a heated high-temperature putter, dried in 3 minutes (removed) and removed from metal, and then crushed with milk to obtain an average diameter of about 70. A metal powder was obtained.
- the obtained noble metal, 50) OO of ana as, and anide (52, solid 25) 009 as ida were mixed with 5 to prepare a compound having a solids degree of 5.
- the obtained compound After the two were combined under 9 On degree by the Niza, further adjustment was made to the attritor (). Further, the obtained compound was combined with a niser under a temperature of about 20 sec. The mixture was reduced to about 20 (20), mixed and removed 5 to obtain a slurry.
- the metal put which had been subjected to a treatment between nOn and oxidization was formed on the surface thereof was immediately taken out from the slurry obtained above, and the slurry on the surface was removed by gravity and by gravity. Then, after placing the pump horizontally, drying at room temperature is 5; drying at n 5 S (6 to 0 C) is 5 to 5; at 9 noC in the air atmosphere; 5 n by S. After repeating such treatment twice, a coating was obtained in which the metallization was about 0 5 with respect to the evaluation plate in the air atmosphere.
- a composite metal powder was obtained in the same manner as in Example 8, except that the liquid was changed to 9 (s) instead of being changed by a nailizer. And. Using the obtained metal compound powder, the compound is slowly replaced by a nitrogenizer, and then slowly ns. . A coating was obtained in the same manner as in Example 8 except that the coating was performed.
- a composite metal powder was obtained in the same manner as in Example 9 except that the solution 5 was replaced with a nailizer and the solution was slowly (0S.c). Then, by using the obtained metal compound powder, the compound is gently pressed by replacing the compound with a nailizer.
- a metallized film was obtained in the same manner as in the embodiment, except that the courage at the time of the use of the Nizer was set to 0 O Osec.
- a metallized film was obtained in the same manner except that the courage at the time of the use of the Niza was set to 4000 SeC.
- the metallized film was obtained in the same manner except that the metallized film was carried out slowly (0 sec) in place of the case of using a nizer.
- the metallized film is carried in the same manner as in the above except that the degree is reduced to 70 C. I got it.
- a metallized film was obtained in the same manner as described above, except that the two-solution Z solution and the Ce solution were combined so as to obtain ZCe4042.
- a metallized film was obtained in the same manner as in the embodiment except that the courage at the time of the use of the Nyzer was set to 0OOOOsec.
- Metallic film is carried in the same manner as above, except that the strength at the time of the use of the Nizer is set to be 400 SeC.
- the 2-ide solution was mixed so as to obtain ZCe0420 and diluted with methano to prepare a 2-loyd solution. Then, the obtained solution was combined with a nitrogen solution at a temperature of c below, and mixed and removed as about 20 n to obtain a coating. Next, the obtained coating 1 was heated to remove the vapor and further heated at 250 to remove the components, and then crushed with milk to obtain a compound powder having an average diameter of about 70.
- a Niza Niza
- a metallized powder was obtained in the same manner as in Example 7, except that the process was repeated.
- a metallized film was obtained in the same manner as described above, except that the solution was mixed with the solution so that OZOaO4 was obtained.
- a metallized film was obtained in the same manner as in Example 8 except that the metallized film was carried out in a similar manner to Example 8 except that the former was used instead of the case using a nailizer. (9)
- a metal film was obtained in the same manner as described above, except that the solution of the solution Z was mixed with the solution of the solution Z so that zY40402 was obtained.
- a metallized film was obtained in the same manner as in Example 9 except that a gentler (0S.c) was used instead of a case using a nailizer.
- a metallized film was obtained in the same manner as in the embodiment except that the mixture was mixed with the 2-id solution so as to obtain a solution of 60.
- a metallized film was obtained in the same manner as in Example 20, except that a gentler (Sec) was used instead of the case using a nizer.
- a metallized film was obtained in the same manner as in Example 2 except that a gentler (Sec) was used instead of a case using a nailizer.
- a metallized film was obtained in the same manner as in the embodiment except that the mixture of 2 and Z liquids was used so as to obtain 2 zOO40.
- the metallized film was carried in the same manner as in Example 22 except that the film was gently pushed (0 SeC) instead of the case of using a nailizer.
- a metallized film was obtained in the same manner as in the embodiment except that the 2e60 solution of 2 and e in the metallized film to be obtained was mixed with a solution of 2 eide2.
- a metallized film was obtained in the same manner as in Example 23, except that the metallized film was carried out in the same manner as in Example 23 except that a gentler () was used instead of a case using a nailizer.
- a metallized film was obtained in the same manner as in Example 6 except that the liquid Z and the Ce liquid were mixed so that ZCe40402 was obtained.
- a metallized film was obtained in the same manner as in Example 2 except that the metallized film was carried out in the same manner as in Example 2 except that the process was carried out loosely (.c) instead of using a nizer.
- the metal qualities obtained by the method described in the Examples and the metal hydrides obtained by the methods described in Example 4 and Comparative Example 4 were evaluated for their component identity in the following manner. That is, according to the above method (), a high voltage of 2 V and an electron beam microscope (J20E, manufactured by HONDA KK) were used for these oxide films and metal oxide powders. The onion X spectroscopy was used to determine the skew, and the degree of X peak at zCe contained in the film was determined.
- the area between 6 and 6 is determined to be an appropriate area for measurement, that is, the fixed area, based on the total number of X-rays obtained by X, with respect to the sample length. Adopted as an area
- the averaged x obtained in this way is a secondary quadratic metric "x2".
- the component identity was evaluated as follows. That is, a microscopic image is shown at 5. Two constant lines of 0.5 are set arbitrarily on the surface of the obtained metallized film (constant straight line 2), and contained in the film and contained in z Ce. At the measured e, an acceleration voltage of 5 V, an electron beam X-Eq analyzer (manufactured by JEOL Ltd., JX820) is used to analyze at the measurement points on the above-mentioned constant line, and (K va e), the value at the fixed point) and the value at the fixed point were determined. In addition, in the part corresponding to the crack, it was determined whether or not e contained in was detected. Fixed straight line Ho The results obtained in Fig. 6 are the actual data of the values), (
- the metallized film obtained by mixing the liquid under high altitude, drying the substrate at a high speed and drying at a high speed is contained in the film.
- the condition represented by (3) was satisfied at the measurement point above the fixed point, and it was confirmed that the metal oxide was extremely uniformly dispersed in the thin film.
- the metal identity (5) formed on the surface of the put by the method described in Comparative Example was evaluated for component identity in the same manner as described above. That is, two constant straight lines (5) are set arbitrarily on the surface of the obtained metallized film exhibiting the microscopic result (constant straight line 2), and ZC contained in the film. It is contained in.
- the metallization film obtained by using the conventional slurry containing ana- lanodiazide powder obtained by co-precipitation as it is does not include the metal source contained in the film. All of the fixed points satisfying the condition represented by (3) above It was confirmed that the total was less than 65.
- the surface condition of the metallized material (5) formed on the surface of the put by the method described in the embodiment was evaluated as follows. 5 In other words, 8 arbitrarily set a constant line of 2 on the surface of the obtained metal film showing the microscope (5, 3, 9
- the image of the surface is measured with a pig tip using the snap tip, and the height () and its
- the obtained liquid was mixed at a high altitude, then dried at a high speed, and then dried at a high speed.
- the film was below OO, and it was confirmed that the surface of the film was fine and convex.
- the case of the fixed point n below 0.05 of the secondary was 54, and the case where the convexity of the film was further increased
- pulverization was polymorphic.
- the metallized material (5) formed on the surface of the put by the method described in the comparative example was used to evaluate the surface state in the same manner as described above. That is, the image of the metallized compound film surface obtained in the comparison was obtained in the same manner as described above. The number of scans was measured and the height was determined. The obtained results are shown in Fig. 23) ”)), 2)) 2f" ).
- the cross-sectional properties of the metallized film formed on the four-mesi-thick camouflage jirai by the method described in the embodiment were evaluated as follows. That is, in the electron microscope of the surface of the obtained metallized film shown in FIG. The four measurement straight lines 1 to were drawn, and the ratio of the intersection of the voids formed in the film with these constant straight lines was determined.
- Fig. 33 shows the graph obtained by measuring the measurement straight line n to 4 minutes of the four trees just above the electron microscope. The part protruding above the border (the black part directly above corresponds to the air gap) .
- the metallization obtained by mixing the liquid at a high altitude, then drying the substrate at a high speed and drying at a high speed was as described above. It is 4.3 at the point where the constant straight line intersects with the voids, confirming that the metallized material has continuity and that the thin film has few voids.
- the cross-sectional properties were evaluated for the metallized film formed on the surface of the dilite having the four-meccam shape by the method described in the embodiment. That is, in the electron microscope of the surface of the obtained metallized film shown in, four measurement lines e 4 of a total of 4.55 were drawn on the film, and these constant lines were formed on the film. The percentage of voids that intersect was determined.
- Fig. 35 shows a graph of the measured values of the four measurement straight lines ne 4) directly above the electron microscope.
- the cross-sectional properties of the metallized film formed on the five-layer cam-shaped jilai by the method described in the embodiment were evaluated. That is, in the electron microscope of the surface of the obtained metallized film shown in 36, a total of 474.5 were placed on the film. The five measurement straight lines (ne to 5) were drawn, and the ratio of the intersection of the voids formed in the film with these constant straight lines was determined. Fig. 37 shows the graph of the measured values of the five measurement straight lines (ne to 5) just above the electron microscope.
- the cross-sectional properties were evaluated in the same manner as described above for the metallized film formed on the 4-layer cam-shaped jelly by the method described in Comparative. That is, in the electron microscope of the surface of the metallized film obtained at 0 shown in 38, four measurement lines (ne to) of 556 ⁇ were drawn on the film in total, and these constant lines were The ratio of the thickness of the portion intersecting with the void formed in the film was determined.
- Fig. 39 shows a graph showing the four measurement straight lines (ne4) just above the electron microscope.
- the metal oxide (400-mesh) obtained by using the slurry containing the conventional ana- diazide powder obtained by co-precipitation had a constant straight line.
- the gap was 424 at the intersection of the gaps, confirming that there are many gaps.
- the metallization obtained by the method described in Example 4 was evaluated for component identity as follows. That is, in accordance with the above-described method, the metallization obtained in Example 4 was measured at a fixed point of the electron microscope shown at 4243 to a fixed point of 0 to 100). The spectrum was determined by onion X spectroscopy using J20, manufactured by HONDA KK, and the degree of X peak in ZCe contained in was determined. Then, as a result, the above-mentioned ratio X, its average value, the next ment 2 next to the average value x, and the second-order metric X of the average value X are sequentially obtained, and are expressed by the above (8). It was determined whether the conditions were satisfied.
- the green onion X skew obtained with the compound of 4 is converted to 4 (with the results of all the fixed points superimposed) and the fixed point (the X ratio X at Ho 0 to Ho o 0 is set to 458).
- the average value of the second-order methons 2x2 to 9 is 9 respectively.
- the metal identity obtained by the method described in Comparative 4 was evaluated for component identity in the same manner as described above. That is, at the metallized fixed points (five measurement points) obtained in Comparison 4, the risks were determined by green onion X spectroscopy, and the degree of X peaks at zCe contained in was determined. Was. Then, based on the result, the aforementioned ratio X, its average value X, the next ment "2 around the average value X" 2, and the second-order metric vx in which the average value x is classified are sequentially output, and the (8 It was determined whether the condition represented by) was satisfied.
- the metal qualities obtained by the method described in the comparative example were evaluated for the component identity in the same manner as described in the above item 5. That is, a spectrum was obtained by onion X spectroscopy at a fixed point (five measurements at five points) of the metallization obtained by comparison, and the degree of X peak at ZCe contained in was obtained. And, based on the result,
- the X ratio at the fixed point 0 5) obtained at the compound of formula 5 is set to 47, and the average value X to the graded second order v X 9, respectively.
- the property of the metalized film formed on the surface of the put by the method described in 39 was evaluated as follows. That is, 48), 492), 503), and 5 (9) are each set to a constant straight line of 5 for each of the compound films showing microscopic truth, and are contained in zCe contained in the film. e is analyzed at the measurement points on the above-mentioned constant line using an X-Equipment analyzer (manufactured by JEOL Ltd., JX820) with an accelerating voltage of 5 V and an electron beam. The average of the values at points) and the value of at fixed points were determined. In addition, it was determined whether or not e contained in was detected in the portion corresponding to the crack. The results obtained are shown in Table 252.
- the portion between the cow and the 6 counts is a region appropriate for the measurement, that is, the sample size, based on the total number of X-rays obtained by X, regarding the sample size. Was adopted as a deemed area.
- the green onion X sock obtained on the metallized film obtained in step 57 is superimposed with the results of all the fixed points.
- 57 is defined in terms of the peak and the peak of the adjacent element.
- K Kr K is the average value Km
- the metal elements contained in the film has the fixed point satisfying the condition (3) below 65 in the case of a fixed point. Was confirmed and confirmed.
- the thermal properties of the metal () formed on the glass by the methods described in Examples 3, Comparative Example 3, and Comparative Example 30 were evaluated as follows. That is, the sample was heated to 500 C 90 O C 00 O C in a compound atmosphere (in the atmosphere) and held at each of 5 points, and then the specific area (B area) was measured.
- the sample was heated to 900 C in an atmosphere (in the atmosphere), and the specific area (area) after measurement was measured.
- the results obtained are as follows.
- the compound of the present invention obtained by mixing the metal liquor at a high speed and then drying and drying at a high speed is obtained. As a result, it was confirmed and confirmed that the thermal properties, especially at 500 C and 90 oC, were greatly improved.
- the durability was evaluated in the following manner for the metallized film made to have the above-mentioned metal density by the method described in 2. That is, first, a metal density cam made of metal
- the liquid on the surface was removed by gravity and. After placing the density cam in a horizontal position, the dryness at room temperature is 5 ⁇ , the drying speed at 2 ⁇ 5s (60 ⁇ 0C) is 5 ⁇ 0, and the drying at 30 ° C in the air atmosphere is about 5 ⁇ 0. 6, and the wind speed was 25 to 50 Dns, and the speed was reduced to 5 to 0. Repeat this process 3 times The metal-made metal density cam was immersed in the metal.
- the metal-made density obtained in this manner is heated in an infrared image furnace, and octa is used as a material in the space 2500, sea bon 2, and ox d.
- a decay test was conducted on ca bon ⁇ cases.
- the exit temperature was set at 600 tonnes as the temperature condition, and gas (CO C C, C) was used as the measurement target, and the following equation was calculated based on the gas degrees.
- the adhesion of each of the metalized materials () formed on the put by the method described in Example 02 was evaluated as follows. That is, the metallized material formed on the surface was subjected to ultrasonic waves to give 30 X4 ultrasonic waves, and the () of the film during that period was measured.
- the compound film of the present invention obtained by mixing the raw materials at a high altitude, laying down, drying at a high speed, and removing the raw It has been confirmed that the adhesion to the steel has greatly improved.
- the metal oxide film obtained by the method described and the metal oxide powder obtained by the method described in Example 7 were confirmed to have oxygen adsorption and X as described below.
- nitrogen adsorption is a method in which the amount of gas desorbed from a solid surface at a certain equilibrium vapor pressure is determined by a static volume.
- Equivalent data can be obtained by introducing a gas into the charged suspension kept at a constant temperature at each time or by removing the gas from the suspension. It is detached by force The amount corresponds to the difference between the gas removed and the gas required to satisfy the value on the sample side.
- a QO automatic gas analyzer (PP) was used. Then, using the materials from ⁇ 05 to ⁇ 5, the PP at measurement temperature of 96 C and atmospheric pressure was subjected to elemental adsorption in the range of ⁇ 00, and the PP was continuously measured in the range of 0 to ⁇ . It was possible to obtain by pressing the gas, and the pore cloth was obtained by an analysis called J. It is assumed that J is the most effective data for calculation and all the holes are cylindrical. The result of the compound obtained by the method described is shown in FIG.
- FIG. 67 shows the result of the metalization. Also, for comparison, the results are shown in 67 for a (6) and aum ().
- the light compound obtained by the light method had a fine diameter of 2 or less.
- the heat treatment is carried out such that the raw material containing the material of the metallization is mixed at a high temperature and then substantially co-precipitated, so that the diameter is reduced Cage
- the novel metallized compound can be obtained since the compound is dispersed in the material.
- the adhesiveness and heat property to each material such as cam eater are improved, and a coating formed on the substrate is also possible.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Environmental & Geological Engineering (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Geology (AREA)
- Biomedical Technology (AREA)
- Analytical Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Combustion & Propulsion (AREA)
- Catalysts (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
- Oxygen, Ozone, And Oxides In General (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN200580018897XA CN1964919B (zh) | 2004-06-11 | 2005-06-09 | 金属氧化物纳米多孔体、用于得到它的被覆组合物以及它们的制造方法 |
| EP05751325A EP1777197B1 (en) | 2004-06-11 | 2005-06-09 | Metal oxide nanoporous body, coating composition for obtaining same, methods for producing those |
| JP2006514615A JP4918857B2 (ja) | 2004-06-11 | 2005-06-09 | 金属酸化物ナノ多孔体、それを得るための被覆組成物、及びそれらの製造方法 |
| US11/628,493 US7935653B2 (en) | 2004-06-11 | 2005-06-09 | Metal oxide nanoporous material, coating composition to obtain the same, and methods of manufacturing them |
Applications Claiming Priority (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004-173595 | 2004-06-11 | ||
| JP2004173595 | 2004-06-11 | ||
| JP2004303957 | 2004-10-19 | ||
| JP2004303853 | 2004-10-19 | ||
| JP2004-303853 | 2004-10-19 | ||
| JP2004-303805 | 2004-10-19 | ||
| JP2004-303957 | 2004-10-19 | ||
| JP2004303805 | 2004-10-19 | ||
| JP2005103318 | 2005-03-31 | ||
| JP2005-103318 | 2005-03-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005121025A1 true WO2005121025A1 (ja) | 2005-12-22 |
Family
ID=35502974
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2005/011003 WO2005121025A1 (ja) | 2004-06-11 | 2005-06-09 | 金属酸化物ナノ多孔体、それを得るための被覆組成物、及びそれらの製造方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US7935653B2 (ja) |
| EP (1) | EP1777197B1 (ja) |
| JP (1) | JP4918857B2 (ja) |
| CN (1) | CN1964919B (ja) |
| WO (1) | WO2005121025A1 (ja) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006232656A (ja) * | 2005-01-28 | 2006-09-07 | Toyota Central Res & Dev Lab Inc | 金属酸化物ナノ多孔体、触媒担体及びそれを用いた水素生成反応用触媒 |
| JP2006346528A (ja) * | 2005-06-13 | 2006-12-28 | Toyota Central Res & Dev Lab Inc | 排ガス浄化用触媒担体、排ガス浄化用触媒、及び排ガス浄化用触媒担体の製造方法 |
| EP1840086A2 (en) | 2006-03-31 | 2007-10-03 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Method of producing nanoporous metal oxide material |
| JP2008246416A (ja) * | 2007-03-30 | 2008-10-16 | Toyota Central R&D Labs Inc | 改質用触媒担体及びその製造方法 |
| JP2009178675A (ja) * | 2008-01-31 | 2009-08-13 | Toyota Central R&D Labs Inc | 排ガス浄化用触媒 |
| US7618919B2 (en) | 2005-01-28 | 2009-11-17 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Catalyst support and method of producing the same |
| US7838460B2 (en) | 2005-01-28 | 2010-11-23 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Nanoporous metal oxide material, catalyst support, and catalyst for hydrogen production reaction using the same |
| WO2011030875A1 (ja) * | 2009-09-11 | 2011-03-17 | 株式会社豊田中央研究所 | 複合金属酸化物多孔体、それを用いた触媒、およびそれらの製造方法 |
| JP2011140432A (ja) * | 2009-12-08 | 2011-07-21 | Toyota Motor Corp | 金属複合酸化物の合成方法、及び、当該合成方法により得られる金属複合酸化物 |
| JP2014024058A (ja) * | 2012-06-20 | 2014-02-06 | Toyota Central R&D Labs Inc | 排ガス浄化用触媒担体、それを用いた排ガス浄化用触媒、及び排ガス浄化用触媒担体の製造方法 |
| CN107838427A (zh) * | 2017-10-31 | 2018-03-27 | 成都易态科技有限公司 | 多孔烧结金属复合薄膜及其制备方法 |
| CN108557754A (zh) * | 2018-04-13 | 2018-09-21 | 杭州电子科技大学 | 一种自支撑金属纳米孔薄膜的制备方法 |
Families Citing this family (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7820583B2 (en) * | 2006-08-24 | 2010-10-26 | Millennium Inorganic Chemicals, Inc. | Nanocomposite particle and process of preparing the same |
| US20100190640A1 (en) * | 2007-06-27 | 2010-07-29 | Takeshi Nobukawa | Catalytic support and exhaust-gas converting catalyst (as amended) |
| FR2928364B1 (fr) * | 2008-03-05 | 2011-10-14 | Rhodia Operations | Composition a base d'un oxyde de zirconium,d'un oxyde de titane ou d'un oxyde mixte de zirconium et de titane sur un support en alumine,procedes de preparation et utilisation comme catalyseur |
| JP5526502B2 (ja) * | 2008-07-16 | 2014-06-18 | 日産自動車株式会社 | 排気ガス浄化用触媒及びその製造方法 |
| US20100081855A1 (en) * | 2008-09-30 | 2010-04-01 | Fina Technology, Inc. | Semi-Supported Dehydrogenation Catalyst |
| EP2404669B1 (en) * | 2009-03-06 | 2019-11-13 | Umicore Shokubai Japan Co., Ltd. | Catalyst for purification of exhaust gas |
| PT2251453E (pt) | 2009-05-13 | 2014-03-13 | Sio2 Medical Products Inc | Retentor de vaso |
| WO2013170052A1 (en) | 2012-05-09 | 2013-11-14 | Sio2 Medical Products, Inc. | Saccharide protective coating for pharmaceutical package |
| US7985188B2 (en) | 2009-05-13 | 2011-07-26 | Cv Holdings Llc | Vessel, coating, inspection and processing apparatus |
| US9458536B2 (en) | 2009-07-02 | 2016-10-04 | Sio2 Medical Products, Inc. | PECVD coating methods for capped syringes, cartridges and other articles |
| US11624115B2 (en) | 2010-05-12 | 2023-04-11 | Sio2 Medical Products, Inc. | Syringe with PECVD lubrication |
| EP2600382A1 (en) * | 2010-07-27 | 2013-06-05 | Hitachi High-Technologies Corporation | Aberration correction device and charged particle beam device employing same |
| US9878101B2 (en) | 2010-11-12 | 2018-01-30 | Sio2 Medical Products, Inc. | Cyclic olefin polymer vessels and vessel coating methods |
| KR101305182B1 (ko) * | 2010-11-30 | 2013-09-12 | 현대자동차주식회사 | 귀금속을 활용한 고성능 촉매 |
| US9272095B2 (en) | 2011-04-01 | 2016-03-01 | Sio2 Medical Products, Inc. | Vessels, contact surfaces, and coating and inspection apparatus and methods |
| EP2776603B1 (en) | 2011-11-11 | 2019-03-06 | SiO2 Medical Products, Inc. | PASSIVATION, pH PROTECTIVE OR LUBRICITY COATING FOR PHARMACEUTICAL PACKAGE, COATING PROCESS AND APPARATUS |
| US11116695B2 (en) | 2011-11-11 | 2021-09-14 | Sio2 Medical Products, Inc. | Blood sample collection tube |
| JP5576420B2 (ja) * | 2012-03-21 | 2014-08-20 | トヨタ自動車株式会社 | 排ガス浄化触媒 |
| WO2013162029A1 (ja) * | 2012-04-27 | 2013-10-31 | トヨタ自動車株式会社 | 酸化鉄-ジルコニア系複合酸化物とその製造方法、および排ガス浄化触媒 |
| WO2014071061A1 (en) | 2012-11-01 | 2014-05-08 | Sio2 Medical Products, Inc. | Coating inspection method |
| WO2014078666A1 (en) | 2012-11-16 | 2014-05-22 | Sio2 Medical Products, Inc. | Method and apparatus for detecting rapid barrier coating integrity characteristics |
| WO2014085346A1 (en) | 2012-11-30 | 2014-06-05 | Sio2 Medical Products, Inc. | Hollow body with inside coating |
| US9764093B2 (en) | 2012-11-30 | 2017-09-19 | Sio2 Medical Products, Inc. | Controlling the uniformity of PECVD deposition |
| US9662450B2 (en) | 2013-03-01 | 2017-05-30 | Sio2 Medical Products, Inc. | Plasma or CVD pre-treatment for lubricated pharmaceutical package, coating process and apparatus |
| CN105392916B (zh) | 2013-03-11 | 2019-03-08 | Sio2医药产品公司 | 涂布包装材料 |
| US9937099B2 (en) | 2013-03-11 | 2018-04-10 | Sio2 Medical Products, Inc. | Trilayer coated pharmaceutical packaging with low oxygen transmission rate |
| EP2971227B1 (en) | 2013-03-15 | 2017-11-15 | Si02 Medical Products, Inc. | Coating method. |
| EP3693493A1 (en) | 2014-03-28 | 2020-08-12 | SiO2 Medical Products, Inc. | Antistatic coatings for plastic vessels |
| EP3337915B1 (en) | 2015-08-18 | 2021-11-03 | SiO2 Medical Products, Inc. | Pharmaceutical and other packaging with low oxygen transmission rate |
| DE102016202610A1 (de) * | 2016-02-19 | 2017-08-24 | Dürr Systems GmbH | Verfahren zur Herstellung eines porösen Funktionskörpers |
| CN112384477A (zh) * | 2018-06-21 | 2021-02-19 | 学校法人工学院大学 | 氧化铁粉末、组合物、陶瓷器、氧化铁粉末前体、氧化铁粉末前体的制造方法及氧化铁粉末的制造方法 |
| DE102020103292A1 (de) * | 2020-02-10 | 2021-08-12 | Umicore Ag & Co. Kg | Verwendung von Ultraschall zur Reinigung von Wandflussfiltersubstraten |
| CN116535957B (zh) * | 2023-05-16 | 2024-03-19 | 深圳市嘉卓成科技发展有限公司 | 一种抗菌抗病毒双组份聚氨酯涂料的制备方法 |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1045412A (ja) * | 1996-07-31 | 1998-02-17 | Sumitomo Chem Co Ltd | 耐熱性遷移アルミナ及びその製造方法 |
| JP2001224962A (ja) * | 1999-12-06 | 2001-08-21 | Toyota Motor Corp | 超臨界流体を用いる触媒の製造方法及び得られた触媒 |
| JP2002001106A (ja) * | 2000-06-20 | 2002-01-08 | Ebara Corp | 除湿又は熱交換用機能素子とその製造方法 |
| EP1175935A2 (en) | 2000-07-27 | 2002-01-30 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Composite oxide, process for producing the same, catalyst for purifying exhaust gas, and process for producing the same |
| JP2002282689A (ja) | 2001-01-19 | 2002-10-02 | Toyota Motor Corp | 触媒担体と触媒及びそれらの製造方法 |
| WO2004043585A2 (en) | 2002-11-13 | 2004-05-27 | Conocophillips Company | Modification of the pore structure of metal oxide and mixed metal oxide supports for catalyst synthesis |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4757045A (en) * | 1983-02-14 | 1988-07-12 | Engelhard Corporation | Catalysts with support coatings having increased macroporosity and method for producing the catalysts |
| US4791090A (en) * | 1986-05-02 | 1988-12-13 | W. R. Grace & Co.-Conn. | Hydroprocessing catalyst and support having bidisperse pore structure |
| US5128291A (en) * | 1990-12-11 | 1992-07-07 | Wax Michael J | Porous titania or zirconia spheres |
| JPH07300315A (ja) | 1994-04-28 | 1995-11-14 | Nissan Motor Co Ltd | 複合体、その複合体を用いた触媒体及びそれらの製造方法 |
| US5962367A (en) * | 1994-11-04 | 1999-10-05 | Dequing Chemical Industry And Technologies Co., Ltd. | Titania (TiO2) support and process for preparation and use of the same |
| JP3262044B2 (ja) | 1996-10-07 | 2002-03-04 | 株式会社豊田中央研究所 | 複合酸化物担体および複合酸化物含有触媒 |
| JP4053623B2 (ja) * | 1996-12-27 | 2008-02-27 | 阿南化成株式会社 | ジルコニウム−セリウム系複合酸化物及びその製造方法 |
| JP3643948B2 (ja) | 1999-03-15 | 2005-04-27 | 株式会社豊田中央研究所 | チタニア−ジルコニア系粉末およびその製造方法 |
| JP3632739B2 (ja) | 1999-03-30 | 2005-03-23 | 株式会社豊田中央研究所 | 酸化チタン−リン酸チタン系粉末、排ガス浄化用触媒担体及び排ガス浄化用触媒 |
| EP1172139B1 (en) * | 2000-07-14 | 2006-10-11 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Catalyst for purifying exhaust gas |
| JP4006976B2 (ja) * | 2000-11-15 | 2007-11-14 | 株式会社豊田中央研究所 | 複合酸化物粉末とその製造方法及び触媒 |
| US20030186805A1 (en) * | 2002-03-28 | 2003-10-02 | Vanderspurt Thomas Henry | Ceria-based mixed-metal oxide structure, including method of making and use |
| US7165553B2 (en) * | 2003-06-13 | 2007-01-23 | Philip Morris Usa Inc. | Nanoscale catalyst particles/aluminosilicate to reduce carbon monoxide in the mainstream smoke of a cigarette |
| US7618919B2 (en) * | 2005-01-28 | 2009-11-17 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Catalyst support and method of producing the same |
-
2005
- 2005-06-09 WO PCT/JP2005/011003 patent/WO2005121025A1/ja active Application Filing
- 2005-06-09 JP JP2006514615A patent/JP4918857B2/ja not_active Expired - Fee Related
- 2005-06-09 EP EP05751325A patent/EP1777197B1/en not_active Expired - Fee Related
- 2005-06-09 US US11/628,493 patent/US7935653B2/en active Active
- 2005-06-09 CN CN200580018897XA patent/CN1964919B/zh not_active Expired - Fee Related
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH1045412A (ja) * | 1996-07-31 | 1998-02-17 | Sumitomo Chem Co Ltd | 耐熱性遷移アルミナ及びその製造方法 |
| JP2001224962A (ja) * | 1999-12-06 | 2001-08-21 | Toyota Motor Corp | 超臨界流体を用いる触媒の製造方法及び得られた触媒 |
| JP2002001106A (ja) * | 2000-06-20 | 2002-01-08 | Ebara Corp | 除湿又は熱交換用機能素子とその製造方法 |
| EP1175935A2 (en) | 2000-07-27 | 2002-01-30 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Composite oxide, process for producing the same, catalyst for purifying exhaust gas, and process for producing the same |
| JP2002282689A (ja) | 2001-01-19 | 2002-10-02 | Toyota Motor Corp | 触媒担体と触媒及びそれらの製造方法 |
| WO2004043585A2 (en) | 2002-11-13 | 2004-05-27 | Conocophillips Company | Modification of the pore structure of metal oxide and mixed metal oxide supports for catalyst synthesis |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1777197A4 |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006232656A (ja) * | 2005-01-28 | 2006-09-07 | Toyota Central Res & Dev Lab Inc | 金属酸化物ナノ多孔体、触媒担体及びそれを用いた水素生成反応用触媒 |
| US7618919B2 (en) | 2005-01-28 | 2009-11-17 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Catalyst support and method of producing the same |
| US7838460B2 (en) | 2005-01-28 | 2010-11-23 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Nanoporous metal oxide material, catalyst support, and catalyst for hydrogen production reaction using the same |
| JP2006346528A (ja) * | 2005-06-13 | 2006-12-28 | Toyota Central Res & Dev Lab Inc | 排ガス浄化用触媒担体、排ガス浄化用触媒、及び排ガス浄化用触媒担体の製造方法 |
| EP1840086A2 (en) | 2006-03-31 | 2007-10-03 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Method of producing nanoporous metal oxide material |
| JP2007269616A (ja) * | 2006-03-31 | 2007-10-18 | Toyota Central Res & Dev Lab Inc | 金属酸化物ナノ多孔体の製造方法 |
| EP1840086A3 (en) * | 2006-03-31 | 2008-07-30 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Method of producing nanoporous metal oxide material |
| JP2008246416A (ja) * | 2007-03-30 | 2008-10-16 | Toyota Central R&D Labs Inc | 改質用触媒担体及びその製造方法 |
| JP2009178675A (ja) * | 2008-01-31 | 2009-08-13 | Toyota Central R&D Labs Inc | 排ガス浄化用触媒 |
| US9556023B2 (en) | 2009-09-11 | 2017-01-31 | Kabushiki Kaisha Toyota Chuo Kenkyusho | Porous composite metal oxide, catalyst using the same, and methods for producing the porous composite metal oxide and the catalyst |
| JPWO2011030875A1 (ja) * | 2009-09-11 | 2013-02-07 | 株式会社豊田中央研究所 | 複合金属酸化物多孔体、それを用いた触媒、およびそれらの製造方法 |
| JP5534249B2 (ja) * | 2009-09-11 | 2014-06-25 | 株式会社豊田中央研究所 | 複合金属酸化物多孔体、それを用いた触媒、およびそれらの製造方法 |
| WO2011030875A1 (ja) * | 2009-09-11 | 2011-03-17 | 株式会社豊田中央研究所 | 複合金属酸化物多孔体、それを用いた触媒、およびそれらの製造方法 |
| JP2011140432A (ja) * | 2009-12-08 | 2011-07-21 | Toyota Motor Corp | 金属複合酸化物の合成方法、及び、当該合成方法により得られる金属複合酸化物 |
| US9126846B2 (en) | 2009-12-08 | 2015-09-08 | Toyota Jidosha Kabushiki Kaisha | Method of synthesizing metal composite oxide and metal composite oxide obtained by same |
| JP2014024058A (ja) * | 2012-06-20 | 2014-02-06 | Toyota Central R&D Labs Inc | 排ガス浄化用触媒担体、それを用いた排ガス浄化用触媒、及び排ガス浄化用触媒担体の製造方法 |
| CN107838427A (zh) * | 2017-10-31 | 2018-03-27 | 成都易态科技有限公司 | 多孔烧结金属复合薄膜及其制备方法 |
| CN107838427B (zh) * | 2017-10-31 | 2024-05-14 | 成都易态科技有限公司 | 多孔烧结金属复合薄膜及其制备方法 |
| CN108557754A (zh) * | 2018-04-13 | 2018-09-21 | 杭州电子科技大学 | 一种自支撑金属纳米孔薄膜的制备方法 |
| CN108557754B (zh) * | 2018-04-13 | 2020-11-10 | 杭州电子科技大学 | 一种自支撑金属纳米孔薄膜的制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1777197B1 (en) | 2013-01-09 |
| EP1777197A1 (en) | 2007-04-25 |
| US20070215009A1 (en) | 2007-09-20 |
| US7935653B2 (en) | 2011-05-03 |
| CN1964919A (zh) | 2007-05-16 |
| CN1964919B (zh) | 2010-08-25 |
| EP1777197A4 (en) | 2009-04-29 |
| JPWO2005121025A1 (ja) | 2008-04-10 |
| JP4918857B2 (ja) | 2012-04-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2005121025A1 (ja) | 金属酸化物ナノ多孔体、それを得るための被覆組成物、及びそれらの製造方法 | |
| Qian et al. | Flexible membranes of Ag-nanosheet-grafted polyamide-nanofibers as effective 3D SERS substrates | |
| DE69720650T4 (de) | Kohlenstofffaser, Acrylfaser und Verfahren zu deren Herstellung | |
| JP6094230B2 (ja) | 焼結鉱の顕微鏡画像解析方法 | |
| CN107478638B (zh) | 银纳米颗粒组装的单层反蛋白石结构及其制备方法和用途 | |
| Barranco et al. | Influence of the microstructure and topography on the barrier properties of oxide scales generated on blasted Ti6Al4V surfaces | |
| Centeno et al. | The formation of chlorine-induced alterations in daguerreotype image particles: a high resolution SEM-EDS study | |
| JP6240788B2 (ja) | 発色処理された基材およびこのための基材の発色処理方法 | |
| CN107860760A (zh) | 氧化石墨烯/银纳米颗粒/金字塔形pmma三维柔性拉曼增强基底及制备方法和应用 | |
| CN105950134A (zh) | 一种制作高清晰、灵敏识别指纹的材料及指纹识别方法 | |
| Qi et al. | Superhydrophobic brocades modified with aligned ZnO nanorods | |
| Veerman | Registration and mapping of plastically deformed metal surfaces by means of photoelectrons | |
| JP5419049B2 (ja) | 微小構造体及びその製造方法 | |
| CN109370567A (zh) | 一种硅球碳点粉末的制备方法及其在潜手印识别中的应用 | |
| Todorov et al. | Is Difflugia nebeloides (Amoebozoa: Arcellinida) really a Difflugia? Re-description and new combination | |
| DE102015120088A1 (de) | Festkörperelektrolyt und Gassensor | |
| GB2544241B (en) | (Fingerprint) powder composition | |
| WO1997034333A1 (de) | Beschichtung von porösen elektroden mit dünnen elektrolytschichten | |
| JP6539085B2 (ja) | 銅粉およびその製造法 | |
| Bernady et al. | The investigation of 15th century paint layers on two stained glass windows from the Dominican Monastery in Krakow, Poland | |
| JP7197408B2 (ja) | 石炭灰中の未燃炭素の評価方法 | |
| JP6996634B2 (ja) | サーミスタ、及び、サーミスタの製造方法 | |
| Enculescu et al. | Influence of geometrical properties on light emission of ZnO nanowires | |
| JP5983167B2 (ja) | クラック評価方法 | |
| CN111925547A (zh) | 一种含氟聚合物复合涂层的聚乙烯膜及其制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006514615 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11628493 Country of ref document: US Ref document number: 2007215009 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580018897.X Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005751325 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005751325 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 11628493 Country of ref document: US |