JP4629047B2 - Glp−1アナログ複合タンパク質 - Google Patents
Glp−1アナログ複合タンパク質 Download PDFInfo
- Publication number
- JP4629047B2 JP4629047B2 JP2006533197A JP2006533197A JP4629047B2 JP 4629047 B2 JP4629047 B2 JP 4629047B2 JP 2006533197 A JP2006533197 A JP 2006533197A JP 2006533197 A JP2006533197 A JP 2006533197A JP 4629047 B2 JP4629047 B2 JP 4629047B2
- Authority
- JP
- Japan
- Prior art keywords
- gly
- ser
- glu
- val
- pro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/26—Immunoglobulins [IG], e.g. monoclonal or polyclonal antibodies against material from animals or humans against hormones ; against hormone releasing or inhibiting factors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5063—Compounds of unknown constitution, e.g. material from plants or animals
- A61K9/5068—Cell membranes or bacterial membranes enclosing drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/51—Nanocapsules; Nanoparticles
- A61K9/5107—Excipients; Inactive ingredients
- A61K9/5176—Compounds of unknown constitution, e.g. material from plants or animals
- A61K9/5184—Virus capsids or envelopes enclosing drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/48—Drugs for disorders of the endocrine system of the pancreatic hormones
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/475—Growth factors; Growth regulators
- C07K14/50—Fibroblast growth factor [FGF]
- C07K14/503—Fibroblast growth factor [FGF] basic FGF [bFGF]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/605—Glucagons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Endocrinology (AREA)
- Zoology (AREA)
- Genetics & Genomics (AREA)
- Pharmacology & Pharmacy (AREA)
- Biochemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Diabetes (AREA)
- Toxicology (AREA)
- Gastroenterology & Hepatology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Immunology (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Epidemiology (AREA)
- Virology (AREA)
- Botany (AREA)
- Child & Adolescent Psychology (AREA)
- Physics & Mathematics (AREA)
- Biomedical Technology (AREA)
- Nanotechnology (AREA)
- Optics & Photonics (AREA)
- Emergency Medicine (AREA)
- Cell Biology (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Description
a) (SEQ ID NO:1)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)、
b) (SEQ ID NO:2)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Lys−Asn−Gly−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)、
c) (SEQ ID NO:3)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly−Pro
(配列中、Xaa8はGlyとValから選択される)、
d) (SEQ ID NO:4)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Lys−Asn−Gly−Gly−Pro
(配列中、Xaa8はGlyとValから選択される)、
e) (SEQ ID NO:5)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)、
f) (SEQ ID NO:6)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Lys−Asn−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)
から成るグループから選択された配列を含み、
Ala−Glu−Ser−Lys−Tyr−Gly−Pro−Pro−
Cys−Pro−Pro−Cys−Pro−Ala−Pro−Xaa16−
Xaa17−Xaa18−Gly−Gly−Pro−Ser−Val−Phe−
Leu−Phe−Pro−Pro−Lys−Pro−Lys−Asp−
Thr−Leu−Met−Ile−Ser−Arg−Thr−Pro−
Glu−Val−Thr−Cys−Val−Val−Val−Asp−
Val−Ser−Gln−Glu−Asp−Pro−Glu−Val−
Gln−Phe−Asn−Trp−Tyr−Val−Asp−Gly−
Val−Glu−Val−His−Asn−Ala−Lys−Thr−
Lys−Pro−Arg−Glu−Glu−Gln−Phe−Xaa80−
Ser−Thr−Tyr−Arg−Val−Val−Ser−Val−
Leu−Thr−Val−Leu−His−Gln−Asp−Trp−
Leu−Asn−Gly−Lys−Glu−Tyr−Lys−Cys−
Lys−Val−Ser−Asn−Lys−Gly−Leu−Pro−
Ser−Ser−Ile−Glu−Lys−Thr−Ile−Ser−
Lys−Ala−Lys−Gly−Gln−Pro−Arg−Glu−
Pro−Gln−Val−Tyr−Thr−Leu−Pro−Pro−
Ser−Gln−Glu−Glu−Met−Thr−Lys−Asn−
Gln−Val−Ser−Leu−Thr−Cys−Leu−Val−
Lys−Gly−Phe−Tyr−Pro−Ser−Asp−Ile−
Ala−Val−Glu−Trp−Glu−Ser−Asn−Gly−
Gln−Pro−Glu−Asn−Asn−Tyr−Lys−Thr−
Thr−Pro−Pro−Val−Leu−Asp−Ser−Asp−
Gly−Ser−Phe−Phe−Leu−Tyr−Ser−Arg−
Leu−Thr−Val−Asp−Lys−Ser−Arg−Trp−
Gln−Glu−Gly−Asn−Val−Phe−Ser−Cys−
Ser−Val−Met−His−Glu−Ala−Leu−His−
Asn−His−Tyr−Thr−Gln−Lys−Ser−Leu−
Ser−Leu−Ser−Leu−Gly−Xaa230
(SEQ ID NO:7)
(配列中、16番目のXaaは、Pro、またはGluであり、
17番目のXaaは、Phe、Val、またはAlaであり、
18番目のXaaは、Leu、Glu、またはAlaであり、
80番目のXaaは、Asn、または Alaであり、
230番目のXaaは、Lys、または欠損である)、
以上のSEQ ID NO:7の配列を含む免疫グロブリンのFc部分と融合されている。
7His−Ala−Glu−10Gly−Thr−Phe−Thr−Ser−15Asp−Val−Ser−Ser−Tyr−20Leu−Glu−Gly−Gln−Ala−
25Ala−Lys−Glu−Phe−Ile−30Ala−Trp−Leu−Val−Lys−35Gly−Arg−37Gly(SEQ ID NO:9)
である。
His−Gly−Glu−Gly−Thr−Phe−Thr−Ser−Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−Trp−Leu−Val−Lys−Gly−Arg−Gly−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Ala−Glu−Ser−Lys−Tyr−Gly−Pro−Pro−Cys−Pro (SEQ ID NO:10)
CACGGCGAGGGCACCTTCACCTCCGACGTGTCCTCCTATCTCGAGGAGCAGGCCGCCAAGGAATTCATCGCCTGGCTGGTGAAGGGCGGCGGCGGTGGTGGTGGCTCCGGAGGCGGCGGCTCTGGTGGCGGTGGCAGCGCTGAGTCCAAATATGGTCCCCCATGCCCACCCTGCCCAGCACCTGAGGCCGCCGGGGGACCATCAGTCTTCCTGTTCCCCCCAAAACCCAAGGACACTCTCATGATCTCCCGGACCCCTGAGGTCACGTGCGTGGTGGTGGACGTGAGCCAGGAAGACCCCGAGGTCCAGTTCAACTGGTACGTGGATGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTTCAACAGCACGTACCGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAACGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGGCCTCCCGTCCTCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAGCCACAGGTGTACACCCTGCCCCCATCCCAGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTACCCCAGCGACATCGCCGTGGAGTGGGAAAGCAATGGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTACAGCAGGCTAACCGTGGACAAGAGCAGGTGGCAGGAGGGGAATGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACACAGAAGAGCCTCTCCCTGTCTCTGGGT (SEQ ID NO:20)
CRE−BLAMシステムを用いたヒトGLP−1受容体を発現するHEK−293細胞を、ポリ−d−リジンコーティングされた底面が透明な黒い96穴プレートに、1ウェルあたり10%FBSを含むDMEM培地100 μl中、20,000から40,000細胞の密度で播種した。播種翌日、培地を除き、血漿を含まないDMEM培地80 μlを添加した。播種後3日目に、用量反応曲線を得るために、異なる濃度の様々なGLP−1−Fc異種融合タンパク質を含む0.5%BSA含有血漿不含DMEM培地20 μlをそれぞれのウェルに加えた。通常、3nmolから30nmolまでの異種GLP1−Fc融合タンパク質を含む14個の希釈物を用いて、EC50値を決定する用量反応曲線を作成した。融合タンパク質とのインキュベーション5時間後、β−ラクタマーゼ基質20 μl(CCF2/AM,パンヴェラ社(PanVera LLC))を加え、1時間インキュベートを続け、Cytofluor(蛍光プレートリーダー)上で蛍光を測定した。このアッセイについては、ズロカーニック(Zlokarnik)等、(1998)サイエンス誌(Science)278:84−88で更に述べられている。種々のGLP−1−Fc融合タンパク質をテストし、EC50値を表1に示した。この値は、全実験で、内部標準として行なったVal8−GLP−1(7−37)OHの結果に対する相対値としてもとめた。
CRE−ルシフェラーゼ系を用いたヒトGLP−1受容体を安定に発現するHEK−293細胞を、96穴プレートに1ウェルあたり低血清DMEM F12培地80 μl中、30,000細胞の密度で播種した。播種翌日、0.5%BSAに溶解したテストタンパク質を20μlの一定分量で混合し、細胞と共に5時間インキュベートした。通常、3pmolから3nmolを含む12個の希釈物を各テストタンパク質用に5倍濃度で調整し、細胞に添加し、EC50値を決定する用量反応曲線を作成した。インキュベーション後、ルシフェラーゼ試薬100 μlをそれぞれのプレートに直接加え、2分間穏やかに混合した。プレートをTRILUX社のルミノメーターに置き、ルシフェラーゼ発現の結果により生じる発光を測定した。種々のGLP−1−Fc融合タンパク質をテストし、EC50値を表2に示した。この値は、全実験で、内部標準として行なったVal8−GLP−1(7−37)OHの結果に対する相対値としてもとめた。以下でテストした融合タンパク質は二量体であるため、値をモル濃度にして2倍の違いを考慮して補正した。
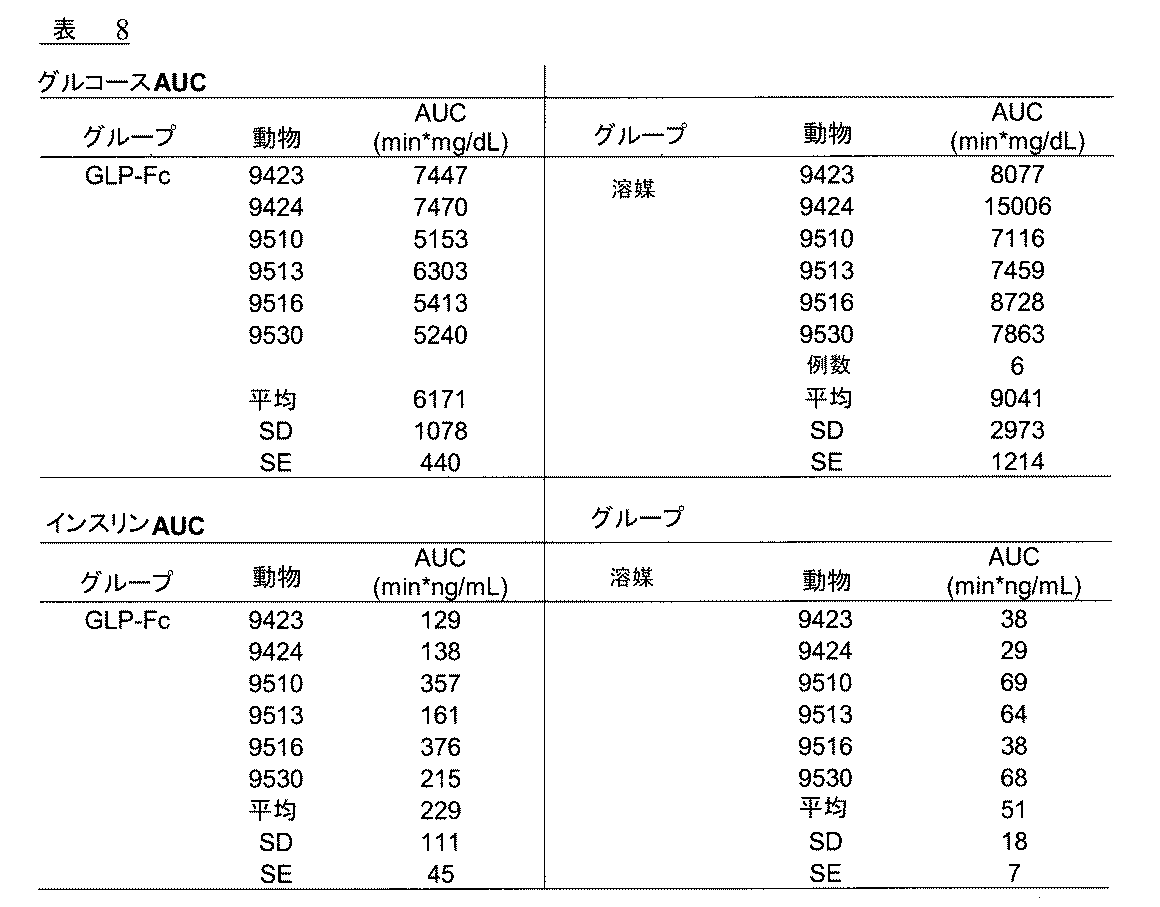
Fc融合タンパク質、Gly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4 (S228P、F234A、L235A)を、ラットにおける静脈内糖負荷試験(IVGTT)にて評価した。3グループそれぞれには、少なくともラット4匹が含まれている。グループIには溶媒を投与し(表3)、グループII にはGly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)、1.79 mg/kgを一回の皮下注射にて投与し(表4)、グループIIIにはGly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4 (S228P,F234A,L235A)、0.179 mg/kgを一回の皮下注射にて投与した(表5)。ラットを、第一日目の朝、皮下注射した。初回の注射から24時間後、ラットの体重1グラムあたり1 μLのグルコース(D50)をボーラス投与した。血液試料を、グルコースボーラス投与2、4、6、10、20および30分後に採取した。
雄のカニクイザルにFc融合タンパク質、Gly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)を0.1 mg/kg皮下注射(SC)した際の薬物動態(PK)を明らかにするために試験を行った。ラジオイムノアッセイ(RIA)抗体はGLPの中間部分に特異的である。酵素免疫測定法(ELISA)は、N末端特異的な捕捉抗体、およびFc特異的な検出抗体を用いる。酵素免疫測定法(ELISA)法およびラジオイムノアッセイ(RIA)法、双方から得られた血漿濃度の結果を、この薬物動態パラメーター値の決定に用いた。
a:観察された最大血漿濃度
b:最大血漿濃度が観察された時間
c:血漿濃度−時間曲線の0から無限大までの曲線下面積(AUC)
d:除去半減期
e:生物学的利用能に対する全身クリアランス
f:生物学的利用能に対する分布容積
SD:標準偏差
カニクイザル(cynomolgus monkey)の所定の血清試料を、Gly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)に対する抗体形成を直接吸着法による酵素免疫測定法(ELISA)法を用いてテストした。マイクロタイタープレートを0.1 μg/mLの濃度のGly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)でコーティングした。サル血清試料をブロッキング溶液で50倍、500倍、1000倍および5000倍に希釈して、ウェル当たり0.05 mLで約1時間インキュベートした。二次抗体、ヤギ抗ヒトFab'2ペルオキシダーゼ結合抗体(ヒトとの交差反応性は75%)を、ブロッキング溶液にて10,000倍に希釈され、ウェル当たり0.05mLで約1時間インキュベートした。テトラメチルベンジジン(tetramethylbenzidine,TMB)基質を用いた発色を、450nm−630nmの吸光度で測定した。2回の計測値を平均した。GLP−1抗体をポジティブコントロールに用い、検出に用いる二次抗体にはヤギ抗ウサギIgG(H+L)ペルオキシダーゼ結合抗体を用いた。抗体形成の可能性を評価するため、血清試料を、投薬前、2回目の投薬の24時間後、1回目、2回目の皮下注射投薬168時間後に採取した。G8E22−CEX−L−hIgG4に対する抗体力価の有無は、投薬前の血清とポジティブコントロールの比較によって解釈された。該当結果は、表7に示す。
段階1(研究第1日)で、溶媒の皮下注射を行った。次いで、5、10、および25 mg/kg/minの段階的な静脈グルコース(20%グルコース)点滴を溶媒注射後直ちに行った。段階2(研究第2日)では、GLP−1融合タンパク質(0.1 mg/kg)の皮下注射を行った。段階3では、GLP−1融合タンパク質の注射後、およそ96時間の段階的な静脈グルコース点滴を行った。
カニューレを常置したラットは、溶媒コントロール(生理食塩水)もしくは3つの処置グループの一つ(GLP−1融合タンパク質、0.0179 mg/kg、0.179 mg/kg、または 1.79 mg/kg)のいずれかに割り当てられた。GLP−1融合タンパク質および溶媒を、皮下注射にて投与した。処置24時間後、前夜絶食させた(16時間)ラットを、段階的静脈グルコース点滴試験の対象とした。段階的グルコース点滴を、ベースラインの生理食塩水期間(20分)、その後それぞれ5および15 mg/kg/minの2回の30分間のグルコース点滴段階で構成した。血漿試料を、グルコース点滴前(ベースライン)20分、10分、0分、およびグルコース点滴後10分、20分、30分、40分、50分、60分に採取した。
Claims (4)
- GLP−1アナログを含む異種融合タンパク質であって、前記GLP−1アナログは、
a) (SEQ ID NO:1)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly−Gly
(配列中、Xaa8はGlyである)、
を含み、前記GLP−1アナログは、
Ala−Glu−Ser−Lys−Tyr−Gly−Pro−Pro−
Cys−Pro−Pro−Cys−Pro−Ala−Pro−Xaa16−
Xaa17−Xaa18−Gly−Gly−Pro−Ser−Val−Phe−
Leu−Phe−Pro−Pro−Lys−Pro−Lys−Asp−
Thr−Leu−Met−Ile−Ser−Arg−Thr−Pro−
Glu−Val−Thr−Cys−Val−Val−Val−Asp−
Val−Ser−Gln−Glu−Asp−Pro−Glu−Val−
Gln−Phe−Asn−Trp−Tyr−Val−Asp−Gly−
Val−Glu−Val−His−Asn−Ala−Lys−Thr−
Lys−Pro−Arg−Glu−Glu−Gln−Phe−Xaa80−
Ser−Thr−Tyr−Arg−Val−Val−Ser−Val−
Leu−Thr−Val−Leu−His−Gln−Asp−Trp−
Leu−Asn−Gly−Lys−Glu−Tyr−Lys−Cys−
Lys−Val−Ser−Asn−Lys−Gly−Leu−Pro−
Ser−Ser−Ile−Glu−Lys−Thr−Ile−Ser−
Lys−Ala−Lys−Gly−Gln−Pro−Arg−Glu−
Pro−Gln−Val−Tyr−Thr−Leu−Pro−Pro−
Ser−Gln−Glu−Glu−Met−Thr−Lys−Asn−
Gln−Val−Ser−Leu−Thr−Cys−Leu−Val−
Lys−Gly−Phe−Tyr−Pro−Ser−Asp−Ile−
Ala−Val−Glu−Trp−Glu−Ser−Asn−Gly−
Gln−Pro−Glu−Asn−Asn−Tyr−Lys−Thr−
Thr−Pro−Pro−Val−Leu−Asp−Ser−Asp−
Gly−Ser−Phe−Phe−Leu−Tyr−Ser−Arg−
Leu−Thr−Val−Asp−Lys−Ser−Arg−Trp−
Gln−Glu−Gly−Asn−Val−Phe−Ser−Cys−
Ser−Val−Met−His−Glu−Ala−Leu−His−
Asn−His−Tyr−Thr−Gln−Lys−Ser−Leu−
Ser−Leu−Ser−Leu−Gly−Xaa230
(SEQ ID NO:7)
(配列中、16番目のXaaは、Gluであり、
17番目のXaaは、Alaであり、
18番目のXaaは、Alaであり、
80番目のXaaは、Asnであり、
230番目のXaaは、欠損である)、
上記のSEQ ID NO:7の配列を含む免疫グロブリンのFc部分と融合されており、該異種融合タンパク質はさらに以下の配列のペプチドリンカーを含み、
Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser (SEQ ID NO:8)、
ここで、ペプチドリンカーのN−末端のグリシンが、前記GLP−1アナログのC−末端のグリシン残基と直接融合しており、ペプチドリンカーのC−末端のセリンが前記Fc部分のN−末端のアラニンと直接融合している、異種融合タンパク質。 - 請求項1に記載の異種融合タンパク質を含有する、医薬組成物。
- インスリン非依存性糖尿病を処置するための、請求項1に記載の異種融合タンパク質を含有する、医薬組成物。
- 体重過多の患者において肥満を処置するためのまたは体重減少を誘導するための、請求項1に記載の異種融合タンパク質を含有する、医薬組成物。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US47788003P | 2003-06-12 | 2003-06-12 | |
| US60/477,880 | 2003-06-12 | ||
| PCT/US2004/015595 WO2005000892A2 (en) | 2003-06-12 | 2004-06-10 | Glp-1 analog fusion plroteins |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2007536902A JP2007536902A (ja) | 2007-12-20 |
| JP4629047B2 true JP4629047B2 (ja) | 2011-02-09 |
Family
ID=33551775
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2006533197A Expired - Lifetime JP4629047B2 (ja) | 2003-06-12 | 2004-06-10 | Glp−1アナログ複合タンパク質 |
Country Status (28)
| Country | Link |
|---|---|
| US (2) | US7452966B2 (ja) |
| EP (2) | EP2368909A1 (ja) |
| JP (1) | JP4629047B2 (ja) |
| KR (1) | KR100758755B1 (ja) |
| CN (2) | CN1802386B (ja) |
| AR (1) | AR044776A1 (ja) |
| AT (1) | ATE525395T1 (ja) |
| AU (1) | AU2004251145C1 (ja) |
| BE (1) | BE2015C007I2 (ja) |
| BR (1) | BRPI0411132B8 (ja) |
| CA (1) | CA2528591C (ja) |
| CY (2) | CY1111991T1 (ja) |
| DK (1) | DK1641823T3 (ja) |
| EA (1) | EA008831B1 (ja) |
| ES (1) | ES2371072T3 (ja) |
| FR (1) | FR15C0010I2 (ja) |
| HR (1) | HRP20110714T1 (ja) |
| HU (1) | HUS1500024I1 (ja) |
| IL (1) | IL171926A (ja) |
| LT (1) | LTC1641823I2 (ja) |
| MX (1) | MXPA05013565A (ja) |
| NZ (1) | NZ543292A (ja) |
| PL (1) | PL1641823T3 (ja) |
| PT (1) | PT1641823E (ja) |
| SI (1) | SI1641823T1 (ja) |
| TW (1) | TW200507870A (ja) |
| UA (1) | UA87458C2 (ja) |
| WO (1) | WO2005000892A2 (ja) |
Families Citing this family (189)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2686899B1 (fr) | 1992-01-31 | 1995-09-01 | Rhone Poulenc Rorer Sa | Nouveaux polypeptides biologiquement actifs, leur preparation et compositions pharmaceutiques les contenant. |
| US7183387B1 (en) | 1999-01-15 | 2007-02-27 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US7459540B1 (en) | 1999-09-07 | 2008-12-02 | Amgen Inc. | Fibroblast growth factor-like polypeptides |
| EP2206720A1 (en) | 2000-04-12 | 2010-07-14 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| HUP0600342A3 (en) * | 2001-10-25 | 2011-03-28 | Genentech Inc | Glycoprotein compositions |
| EP2261250B1 (en) | 2001-12-21 | 2015-07-01 | Human Genome Sciences, Inc. | GCSF-Albumin fusion proteins |
| AU2002364587A1 (en) | 2001-12-21 | 2003-07-30 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| CA2513213C (en) | 2003-01-22 | 2013-07-30 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| US20070161087A1 (en) * | 2003-05-29 | 2007-07-12 | Wolfgang Glaesner | Glp-1 fusion proteins |
| MXPA05013565A (es) * | 2003-06-12 | 2006-03-09 | Lilly Co Eli | Proteinas de fusion analogas al glp-1. |
| ES2567634T3 (es) | 2004-02-09 | 2016-04-25 | Human Genome Sciences, Inc. | Proteínas de fusión de albúmina |
| EA011166B1 (ru) * | 2004-12-22 | 2009-02-27 | Эли Лилли Энд Компани | Композиции слитых белков-аналогов glp-1 |
| PT1881850E (pt) * | 2005-05-13 | 2010-11-26 | Lilly Co Eli | Compostos peguilados de glp-1 |
| CA2658673A1 (en) * | 2005-07-27 | 2008-02-01 | Gerald J. Prud'homme | Composition and method for prevention and treatment of type i and type ii diabetes |
| CA2658678A1 (en) | 2005-08-06 | 2007-02-15 | Gerald J. Prud'homme | Composition and method for prevention and treatment of type i diabetes |
| PL1767545T3 (pl) * | 2005-09-22 | 2010-04-30 | Biocompatibles Uk Ltd | Polipeptydy fuzyjne GLP-1 (glukagonopodobny peptyd-1) o zwiększonej odporności na peptydazy |
| WO2007067828A2 (en) * | 2005-10-24 | 2007-06-14 | Centocor, Inc. | Glp-2 mimetibodies, polypeptides, compositions, methods and uses |
| ES2586236T3 (es) | 2005-11-04 | 2016-10-13 | Glaxosmithkline Llc | Procedimientos para administrar agentes hipoglucémicos |
| EP2570133B1 (en) | 2005-11-07 | 2016-03-23 | Indiana University Research and Technology Corporation | Glucagon analogs exhibiting physiological solubility and stability |
| US8841255B2 (en) | 2005-12-20 | 2014-09-23 | Duke University | Therapeutic agents comprising fusions of vasoactive intestinal peptide and elastic peptides |
| US20130172274A1 (en) | 2005-12-20 | 2013-07-04 | Duke University | Methods and compositions for delivering active agents with enhanced pharmacological properties |
| EP1816201A1 (en) | 2006-02-06 | 2007-08-08 | CSL Behring GmbH | Modified coagulation factor VIIa with extended half-life |
| ES2495741T3 (es) | 2006-04-20 | 2014-09-17 | Amgen, Inc | Compuestos de GLP-1 |
| WO2008028117A2 (en) * | 2006-08-31 | 2008-03-06 | Centocor, Inc. | Glp-2 mimetibodies, polypeptides, compositions, methods and uses |
| EP2059606A4 (en) * | 2006-09-06 | 2010-04-07 | Phasebio Pharmaceuticals Inc | PEPTIDE FUSION THERAPEUTIC COMPOSITIONS |
| US8338376B2 (en) * | 2006-10-20 | 2012-12-25 | Biogen Idec Ma Inc. | Compositions comprising variant LT-B-R-IG fusion proteins |
| AU2007338298B2 (en) | 2006-12-22 | 2013-02-07 | Csl Behring Gmbh | Modified coagulation factors with prolonged in vivo half-life |
| JP2008169195A (ja) * | 2007-01-05 | 2008-07-24 | Hanmi Pharmaceutical Co Ltd | キャリア物質を用いたインスリン分泌ペプチド薬物結合体 |
| AU2011254001B2 (en) * | 2007-01-05 | 2012-08-02 | Covx Technologies Ireland Limited | Glucagon-like protein-1 receptor (GLP-1R) agonist compounds |
| US20090098130A1 (en) * | 2007-01-05 | 2009-04-16 | Bradshaw Curt W | Glucagon-like protein-1 receptor (glp-1r) agonist compounds |
| RU2477286C2 (ru) * | 2007-01-05 | 2013-03-10 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | АНАЛОГИ ГЛЮКАГОНА, ОБЛАДАЮЩИЕ ПОВЫШЕННОЙ РАСТВОРИМОСТЬЮ В БУФЕРАХ С ФИЗИОЛОГИЧЕСКИМ ЗНАЧЕНИЕМ pH |
| EP2526962B1 (en) | 2007-02-12 | 2019-08-14 | CSL Behring GmbH | Therapeutic application of Kazal-type serine protease inhibitors |
| AU2008216265B2 (en) | 2007-02-15 | 2014-04-03 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| JP5431171B2 (ja) * | 2007-03-15 | 2014-03-05 | バイオジェン・アイデック・エムエイ・インコーポレイテッド | 自己免疫障害の処置 |
| TWI423821B (zh) * | 2007-06-19 | 2014-01-21 | Glucose-containing glucagon-like peptide-1 peptide | |
| CA2693504A1 (en) | 2007-08-03 | 2009-02-12 | Eli Lilly And Company | Treatment for obesity |
| US7960336B2 (en) * | 2007-08-03 | 2011-06-14 | Pharmain Corporation | Composition for long-acting peptide analogs |
| US8563527B2 (en) * | 2007-08-20 | 2013-10-22 | Pharmain Corporation | Oligonucleotide core carrier compositions for delivery of nucleic acid-containing therapeutic agents, methods of making and using the same |
| EP2031064A1 (de) * | 2007-08-29 | 2009-03-04 | Boehringer Ingelheim Pharma GmbH & Co. KG | Verfahren zur Steigerung von Proteintitern |
| US8980830B2 (en) * | 2007-10-30 | 2015-03-17 | Indiana University Research And Technology Corporation | Peptide compounds exhibiting glucagon antagonist and GLP-1 agonist activity |
| WO2009058662A2 (en) | 2007-10-30 | 2009-05-07 | Indiana University Research And Technology Corporation | Glucagon antagonists |
| US20100317057A1 (en) * | 2007-12-28 | 2010-12-16 | Novo Nordisk A/S | Semi-recombinant preparation of glp-1 analogues |
| AU2009210570B2 (en) * | 2008-01-30 | 2014-11-20 | Indiana University Research And Technology Corporation | Ester-based insulin prodrugs |
| EP2296690B1 (en) | 2008-06-04 | 2016-11-30 | Amgen, Inc | Fgf21 mutants and uses thereof |
| EP2300037B1 (en) | 2008-06-17 | 2016-03-30 | Indiana University Research and Technology Corporation | Glucagon/glp-1 receptor co-agonists |
| WO2009155257A1 (en) * | 2008-06-17 | 2009-12-23 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility and stability physiological ph buffers |
| DK2300035T3 (en) * | 2008-06-17 | 2015-11-16 | Univ Indiana Res & Tech Corp | Mixed GIP-based agonists for the treatment of metabolic diseases and obesity |
| KR101648734B1 (ko) | 2008-06-24 | 2016-08-18 | 체에스엘 베링 게엠베하 | 연장된 생체내 반감기를 갖는 인자 viii, 폰 빌레브란트 인자 또는 이들의 복합체 |
| EP3412300A1 (en) | 2008-06-27 | 2018-12-12 | Duke University | Therapeutic agents comprising elastin-like peptides |
| JP5878757B2 (ja) | 2008-10-10 | 2016-03-08 | アムジエン・インコーポレーテツド | Fgf21変異体およびその使用 |
| CN102325539A (zh) | 2008-12-19 | 2012-01-18 | 印第安纳大学研究及科技有限公司 | 基于酰胺的胰高血糖素超家族肽前药 |
| US9238878B2 (en) | 2009-02-17 | 2016-01-19 | Redwood Bioscience, Inc. | Aldehyde-tagged protein-based drug carriers and methods of use |
| EP2427207B1 (en) | 2009-05-05 | 2017-08-16 | Amgen, Inc | Fgf21 mutants and uses thereof |
| HRP20240135T1 (hr) * | 2009-05-05 | 2024-04-12 | Amgen Inc. | Fgf21 mutanti i njihove upotrebe |
| WO2010148089A1 (en) | 2009-06-16 | 2010-12-23 | Indiana University Research And Technology Corporation | Gip receptor-active glucagon compounds |
| US8324160B2 (en) * | 2009-06-17 | 2012-12-04 | Amgen Inc. | Chimeric polypeptides and uses thereof |
| CN101993485B (zh) | 2009-08-20 | 2013-04-17 | 重庆富进生物医药有限公司 | 促胰岛素分泌肽类似物同源二聚体及其用途 |
| WO2011043530A1 (ko) * | 2009-10-09 | 2011-04-14 | (주)알테오젠 | Glp-1 유사체의 융합체, 및 이를 유효성분으로 함유하는 당뇨병의 예방 또는 치료용 조성물 |
| EP2679234A3 (en) * | 2009-12-02 | 2014-04-23 | Amgen Inc. | Binding proteins that bind to human FGFR1C, human beta-klotho and both human FGFR1C and human beta-klotho |
| UA109888C2 (uk) * | 2009-12-07 | 2015-10-26 | ІЗОЛЬОВАНЕ АНТИТІЛО АБО ЙОГО ФРАГМЕНТ, ЩО ЗВ'ЯЗУЄТЬСЯ З β-КЛОТО, РЕЦЕПТОРАМИ FGF І ЇХНІМИ КОМПЛЕКСАМИ | |
| US8703701B2 (en) | 2009-12-18 | 2014-04-22 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| EP2528618A4 (en) | 2010-01-27 | 2015-05-27 | Univ Indiana Res & Tech Corp | GLUCAGON ANTAGONISTE AND GIP AGONISTS CONJUGATES AND COMPOSITIONS FOR THE TREATMENT OF METABOLISM DISEASES AND ADIPOSITAS |
| EP2371857A1 (en) | 2010-04-01 | 2011-10-05 | CSL Behring GmbH | Factor XII inhibitors for treating interstitial lung disease |
| CA2796055A1 (en) | 2010-04-15 | 2011-10-20 | Amgen Inc. | Human fgf receptor and .beta.-klotho binding proteins |
| WO2011136361A1 (ja) | 2010-04-30 | 2011-11-03 | 株式会社 三和化学研究所 | 生理活性物質等の生体内安定性向上のためのペプチド及び生体内安定性が向上した生理活性物質 |
| WO2011143208A1 (en) | 2010-05-13 | 2011-11-17 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting g protein-coupled receptor activity |
| CA2797095A1 (en) | 2010-05-13 | 2011-11-17 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting nuclear hormone receptor activity |
| CN101891823B (zh) | 2010-06-11 | 2012-10-03 | 北京东方百泰生物科技有限公司 | 一种Exendin-4及其类似物融合蛋白 |
| RU2580317C2 (ru) | 2010-06-24 | 2016-04-10 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Пептидные пролекарства, принадлежащие к суперсемейству амид-содержащих глюкагонов |
| EP2595647A1 (en) * | 2010-07-20 | 2013-05-29 | Novo Nordisk A/S | N-terminal modified fgf21 compounds |
| US8507428B2 (en) | 2010-12-22 | 2013-08-13 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting GIP receptor activity |
| AU2012205301B2 (en) | 2011-01-14 | 2017-01-05 | Redwood Bioscience, Inc. | Aldehyde-tagged immunoglobulin polypeptides and method of use thereof |
| CN103415301A (zh) | 2011-03-09 | 2013-11-27 | 德国杰特贝林生物制品有限公司 | 用于与包括与人造表面接触的医疗操作一起施用的fxii抑制剂 |
| EP2497489A1 (en) | 2011-03-09 | 2012-09-12 | CSL Behring GmbH | Factor XII inhibitors for the treatment of silent brain ischemia and ischemia of other organs |
| SG10201602394QA (en) | 2011-03-29 | 2016-05-30 | Roche Glycart Ag | Antibody FC Variants |
| BR112013032717A2 (pt) | 2011-06-22 | 2017-01-24 | Univ Indiana Res & Tech Corp | coagonistas do receptor de glucagon/glp-1 |
| RS56173B1 (sr) | 2011-06-22 | 2017-11-30 | Univ Indiana Res & Tech Corp | Koagonisti receptora za glukagon/glp-1 receptora |
| ES3005912T3 (en) | 2011-07-22 | 2025-03-17 | Csl Behring Gmbh | Inhibitory anti-factor xii/xiia monoclonal antibodies and their uses |
| UY34317A (es) | 2011-09-12 | 2013-02-28 | Genzyme Corp | Anticuerpo antireceptor de célula T (alfa)/ß |
| KR20130049671A (ko) | 2011-11-04 | 2013-05-14 | 한미사이언스 주식회사 | 생리활성 폴리펩타이드 결합체 제조 방법 |
| RU2014117678A (ru) | 2011-11-17 | 2015-12-27 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Пептиды глюкагонового суперсемейства, обладающие глюкокортикоидной рецепторной активностью |
| EP2623110A1 (en) | 2012-01-31 | 2013-08-07 | CSL Behring GmbH | Factor XII inhibitors for the treatment of neurological inflammatory disorders |
| WO2013120939A1 (en) | 2012-02-15 | 2013-08-22 | Csl Behring Gmbh | Von willebrand factor variants having improved factor viii binding affinity |
| CA2877358A1 (en) | 2012-06-21 | 2013-12-27 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting gip receptor activity |
| LT3495387T (lt) | 2012-07-13 | 2021-11-25 | Roche Glycart Ag | Bispecifiniai anti-vegf / anti-ang-2 antikūnai ir jų panaudojimas akių kraujagyslių ligoms gydyti |
| EA035987B1 (ru) * | 2012-09-12 | 2020-09-09 | Джензим Корпорейшн | ПОЛИПЕПТИДЫ, СОДЕРЖАЩИЕ Fc С ИЗМЕНЕННЫМ ГЛИКОЗИЛИРОВАНИЕМ И СНИЖЕННОЙ АФФИННОСТЬЮ К Fc-ГАММА РЕЦЕПТОРАМ |
| US9790268B2 (en) | 2012-09-12 | 2017-10-17 | Genzyme Corporation | Fc containing polypeptides with altered glycosylation and reduced effector function |
| JP6026002B2 (ja) | 2012-11-27 | 2016-11-16 | アルテオジェン インコーポレイテッド | タンパク質とFcドメインを融合した融合タンパク質の安定化用組成物 |
| HUE039334T2 (hu) | 2012-11-27 | 2018-12-28 | Biomarin Pharm Inc | Célzott terápiás lizoszómális enzim fúziós fehérjék és azok alkalmazásai |
| CA2901225C (en) | 2013-03-08 | 2023-09-19 | Csl Behring Gmbh | Treatment and prevention of remote ischemia-reperfusion injury |
| CN110256560A (zh) | 2013-03-11 | 2019-09-20 | 建新公司 | 通过糖工程的位点特异性抗体-药物偶联 |
| US9580486B2 (en) | 2013-03-14 | 2017-02-28 | Amgen Inc. | Interleukin-2 muteins for the expansion of T-regulatory cells |
| WO2014158900A1 (en) | 2013-03-14 | 2014-10-02 | Indiana University Research And Technology Corporation | Insulin-incretin conjugates |
| EP2796145B1 (en) | 2013-04-22 | 2017-11-01 | CSL Ltd. | A covalent complex of von willebrand factor and faktor viii linked by a disulphide bridge |
| JP2017501968A (ja) | 2013-06-28 | 2017-01-19 | ツェー・エス・エル・ベーリング・ゲー・エム・ベー・ハー | 第xii因子インヒビターおよびc1−インヒビターを使用する組合せ療法 |
| CN103408669B (zh) * | 2013-08-01 | 2016-01-20 | 江苏泰康生物医药有限公司 | Glp-1类似物融合蛋白,及其制备方法和用途 |
| CN104592381A (zh) * | 2013-10-31 | 2015-05-06 | 江苏万邦生化医药股份有限公司 | 一种利拉鲁肽中间体多肽的制备方法 |
| US20170065678A1 (en) | 2014-03-11 | 2017-03-09 | Novartis Ag | Methods of treating metabolic disorders associated with lipodystrophies and defects in insulin production or signaling |
| PL3129067T3 (pl) | 2014-03-19 | 2023-05-08 | Genzyme Corporation | Specyficzne dla miejsca glikomodyfikowanie ugrupowań celujących |
| AU2015242657B2 (en) * | 2014-03-31 | 2020-05-21 | Hanmi Pharm. Co., Ltd. | Method for improving solubility of protein and peptide by using immunoglobulin Fc fragment linkage |
| BR112016029624A2 (pt) | 2014-06-18 | 2017-10-24 | Csl Behring Gmbh | terapia usando um inibidor do fator xii em um distúrbio neurotraumático |
| CA2953593C (en) | 2014-07-02 | 2023-09-26 | Csl Limited | Modified von willebrand factor |
| GB2538666A (en) | 2014-07-21 | 2016-11-23 | Delinia Inc | Molecules that selectively activate regulatory T cells for the treatment of autoimmune diseases |
| WO2016025385A1 (en) | 2014-08-11 | 2016-02-18 | Delinia, Inc. | Modified il-2 variants that selectively activate regulatory t cells for the treatment of autoimmune diseases |
| ES2822994T3 (es) | 2014-09-24 | 2021-05-05 | Univ Indiana Res & Tech Corp | Conjugados de incretina-insulina |
| CN104293834B (zh) * | 2014-10-11 | 2018-03-23 | 上海兴迪金生物技术有限公司 | GLP‑1或其类似物与抗体Fc片段融合蛋白的制备方法 |
| AR103246A1 (es) | 2014-12-23 | 2017-04-26 | Novo Nordiks As | Derivados de fgf21 y sus usos |
| KR101825048B1 (ko) | 2014-12-31 | 2018-02-05 | 주식회사 제넥신 | GLP 및 면역글로불린 하이브리드 Fc 융합 폴리펩타이드 및 이의 용도 |
| MY188430A (en) | 2015-04-10 | 2021-12-08 | Amgen Inc | Interleukin-2 muteins for the expansion of t-regulatory cells |
| AR105616A1 (es) * | 2015-05-07 | 2017-10-25 | Lilly Co Eli | Proteínas de fusión |
| RU2017145014A (ru) | 2015-05-22 | 2019-06-24 | Цсл Беринг Ленгнау Аг | Усеченные полипептиды фактора фон виллебранда для лечения гемофилии |
| WO2016188905A1 (en) | 2015-05-22 | 2016-12-01 | Csl Behring Recombinant Facility Ag | Methods for preparing modified von willebrand factor |
| TWI622596B (zh) | 2015-10-26 | 2018-05-01 | 美國禮來大藥廠 | 升糖素受體促效劑 |
| KR102670157B1 (ko) | 2015-10-28 | 2024-05-29 | 주식회사유한양행 | 이중 작용 단백질 및 이를 포함하는 약학적 조성물 |
| KR102668200B1 (ko) | 2015-10-28 | 2024-05-23 | 주식회사유한양행 | 지속형 fgf21 융합 단백질 및 이를 포함하는 약학적 조성물 |
| EP3184149A1 (en) | 2015-12-23 | 2017-06-28 | Julius-Maximilians-Universität Würzburg | Soluble glycoprotein v for treating thrombotic diseases |
| KR102844773B1 (ko) | 2015-12-23 | 2025-08-13 | 암젠 인크 | 위 억제 펩티드 수용체 (gipr)에 대한 결합 단백질을 glp-1 효능제와 조합하여 사용하여 대사 장애를 치료하거나 개선시키는 방법 |
| JP6851381B6 (ja) | 2016-01-07 | 2021-04-21 | ツェー・エス・エル・ベーリング・レングナウ・アクチエンゲゼルシャフト | 変異切断型フォンウィルブランド因子 |
| WO2017117630A1 (en) | 2016-01-07 | 2017-07-13 | Csl Limited | Mutated von willebrand factor |
| US20170204154A1 (en) | 2016-01-20 | 2017-07-20 | Delinia, Inc. | Molecules that selectively activate regulatory t cells for the treatment of autoimmune diseases |
| EP3440107A4 (en) | 2016-04-06 | 2020-02-19 | CSL Limited | METHOD FOR TREATING ATHEROSCLEROSIS |
| AU2017257504A1 (en) | 2016-04-26 | 2018-10-25 | R.P. Scherer Technologies, Llc | Antibody conjugates and methods of making and using the same |
| US10336812B2 (en) | 2016-05-10 | 2019-07-02 | Janssen Biotech, Inc. | GDF15 fusion proteins and uses thereof |
| CN106279437B (zh) | 2016-08-19 | 2017-10-31 | 安源医药科技(上海)有限公司 | 高糖基化人凝血因子viii融合蛋白及其制备方法与用途 |
| US11123438B2 (en) | 2016-08-19 | 2021-09-21 | Ampsource Biopharma Shanghai Inc. | Linker peptide for constructing fusion protein |
| CN107759697B (zh) | 2016-08-19 | 2023-03-24 | 安源医药科技(上海)有限公司 | 制备融合蛋白的方法 |
| US11077172B2 (en) | 2016-11-08 | 2021-08-03 | Delinia, Inc. | IL-2 variants for the treatment of psoriasis |
| AU2017358289C1 (en) | 2016-11-10 | 2025-05-08 | Yuhan Corporation | Pharmaceutical composition for preventing or treating hepatitis, hepatic fibrosis, and hepatic cirrhosis comprising fusion proteins |
| CA3043397A1 (en) | 2016-11-11 | 2018-05-17 | CSL Behring Lengnau AG | Truncated von willebrand factor polypeptides for extravascular administration in the treatment or prophylaxis of a blood coagulation disorder |
| SG10201912360SA (en) | 2016-11-11 | 2020-02-27 | CSL Behring Lengnau AG | Truncated von willebrand factor polypeptides for treating hemophilia |
| EP3351262A1 (en) | 2016-12-30 | 2018-07-25 | Istanbul Universitesi Rektorlugu | Curaglutide for in treatment of prediabetes, diabetes, obesity and metabolic diseases associated thereto |
| BR112019014588A2 (pt) | 2017-01-17 | 2020-02-18 | Amgen Inc. | Método de tratamento ou amenização de distúrbios metabólicos usando agonistas do receptor de glp-1 conjugados a antagonistas para receptor do peptídeo inibidor gástrico (gipr) |
| EP4470551A3 (en) | 2017-03-14 | 2025-02-26 | Sunshine Lake Pharma Co., Ltd. | Dual-target fusion proteins comprising the fc portion of an immunoglobulin |
| CN110709520B (zh) | 2017-04-21 | 2024-04-05 | 株式会社柳韩洋行 | 用于产生双功能蛋白质及其衍生物的方法 |
| JP7191043B2 (ja) | 2017-06-01 | 2022-12-16 | イーライ リリー アンド カンパニー | 慢性腎疾患の治療のためのデュラグルチド |
| CN110831969B (zh) | 2017-06-20 | 2024-06-21 | 安进公司 | 使用抑胃肽受体(gipr)结合蛋白与glp-1激动剂的组合治疗或改善代谢障碍的方法 |
| MX2019015544A (es) | 2017-06-21 | 2020-07-28 | Amgen Inc | Metodo para tratar o mejorar trastornos metabolicos con proteinas de fusion de agonistas del receptor de glp-1/proteinas de union antagonistas para el receptor peptidico inhibidor gastrico (gipr). |
| KR20200018690A (ko) | 2017-06-22 | 2020-02-19 | 체에스엘 베링 렝나우 아게 | 절단된 vwf에 의한 fviii 면역원성의 조절 |
| BR112020002871A2 (pt) | 2017-08-15 | 2020-07-28 | Kindred Biosciences, Inc. | variantes fc de igg para uso veterinário |
| MY200364A (en) | 2017-09-22 | 2023-12-21 | Regeneron Pharma | Glucagon-like peptide 1 receptor agonists and uses thereof |
| MX2020005231A (es) | 2017-11-21 | 2020-08-24 | Lilly Co Eli | Metodos de uso y composiciones que contienen dulaglutida. |
| CN109929806B (zh) | 2017-12-19 | 2020-05-08 | 北京吉源生物科技有限公司 | 一种表达glp1和fgf21的干细胞及其用途 |
| US20200376080A1 (en) | 2017-12-22 | 2020-12-03 | Novartis Ag | Methods of treating metabolic disorders with fgf21 variants |
| WO2019125003A1 (ko) * | 2017-12-22 | 2019-06-27 | 케이비바이오메드 주식회사 | 경구용 유전자 전달체 및 이의 용도 |
| WO2019140021A1 (en) | 2018-01-12 | 2019-07-18 | Eli Lilly And Company | Combination therapy |
| AU2019218147B2 (en) | 2018-02-08 | 2023-06-08 | Sunshine Lake Pharma Co., Ltd. | FGF21 variant, fusion protein and application thereof |
| KR20200135618A (ko) | 2019-05-23 | 2020-12-03 | ㈜ 디앤디파마텍 | 폴리펩티드를 포함하는 비알코올성 지방간 질환의 예방 또는 치료용 약학 조성물 |
| CN121591873A (zh) | 2018-07-19 | 2026-03-03 | D&D制药技术股份有限公司 | 包括多肽的药物组合物 |
| CN110878127B (zh) | 2018-09-06 | 2022-06-28 | 浙江柏拉阿图医药科技有限公司 | 长效重组GLP1-Fc-CD47蛋白及其制备和用途 |
| EP3870211B1 (en) | 2018-10-22 | 2025-01-08 | Janssen Sciences Ireland Unlimited Company | Glucagon like peptide 1 (glp1)-growth differentiation factor 15 (gdf15) fusion proteins and uses thereof |
| EP3873519A1 (en) | 2018-10-29 | 2021-09-08 | F. Hoffmann-La Roche AG | Antibody formulation |
| CN111234000B (zh) * | 2018-11-28 | 2023-05-26 | 鲁南制药集团股份有限公司 | 艾塞纳肽类似物 |
| CN111269312B (zh) * | 2018-12-04 | 2023-05-09 | 鲁南制药集团股份有限公司 | 一种异源融合蛋白质 |
| CN109836486B (zh) * | 2019-01-30 | 2020-09-08 | 北京双因生物科技有限公司 | 成纤维生长因子21变体、其融合蛋白及其用途 |
| CN111662373B (zh) * | 2019-03-05 | 2024-05-14 | 广东东阳光药业股份有限公司 | 一种多肽分子及其应用 |
| EP3934679A1 (en) | 2019-03-08 | 2022-01-12 | Amgen Inc. | Growth differentiation factor 15 combination therapy |
| WO2020190591A1 (en) | 2019-03-15 | 2020-09-24 | Eli Lilly And Company | Preserved formulations |
| MA55529A (fr) | 2019-04-03 | 2022-02-09 | Genzyme Corp | Polypeptides de liaison anti-alpha bêta tcr à fragmentation réduite |
| CA3177693A1 (en) | 2019-04-05 | 2020-10-05 | Eli Lilly And Company | Therapeutic uses of dulaglutide |
| JP2022530216A (ja) * | 2019-04-23 | 2022-06-28 | エルジー・ケム・リミテッド | 免疫グロブリンのFc領域およびGDF15を含む融合ポリペプチド |
| AU2020277640A1 (en) | 2019-05-17 | 2022-01-20 | Csl Behring Ag | Haptoglobin for use in treating an adverse secondary neurological outcome following a haemorrhagic stroke |
| CN114072420B (zh) | 2019-07-04 | 2024-06-11 | 康诺贝林伦瑙有限公司 | 用于增加凝血因子viii的体外稳定性的截短的血管性血友病因子(vwf) |
| AU2020328038B2 (en) | 2019-08-13 | 2025-10-09 | Amgen Inc. | Interleukin-2 muteins for the expansion of T-regulatory cells |
| EP4045641A1 (en) | 2019-10-15 | 2022-08-24 | Eli Lilly and Company | Recombinantly engineered, lipase/esterase-deficient mammalian cell lines |
| JP7680442B2 (ja) | 2019-11-11 | 2025-05-20 | ツェー・エス・エル・ベーリング・レングナウ・アクチエンゲゼルシャフト | 第viii因子に対する寛容を誘導するためのポリペプチド |
| CN115322794B (zh) | 2020-01-11 | 2025-09-19 | 北京质肽生物医药科技有限公司 | Glp-1和fgf21的融合蛋白的缀合物 |
| US11981718B2 (en) | 2020-05-27 | 2024-05-14 | Ampsource Biopharma Shanghai Inc. | Dual-function protein for lipid and blood glucose regulation |
| BR112023003310A2 (pt) | 2020-08-24 | 2023-05-02 | Univ Pennsylvania | Vetores virais que codificam fusões de agonistas do receptor de glp-1 e usos dos mesmos no tratamento de doenças metabólicas |
| CN114106194B (zh) * | 2020-08-31 | 2024-01-16 | 中国科学院天津工业生物技术研究所 | 一种用于治疗糖尿病和/或肥胖症的融合蛋白 |
| AU2021381360A1 (en) | 2020-11-20 | 2023-06-22 | Csl Behring Gmbh | Method for treating antibody-mediated rejection |
| CN114685644A (zh) | 2020-12-29 | 2022-07-01 | 苏州康宁杰瑞生物科技有限公司 | 一种人glp-1多肽变体及其应用 |
| US20250339492A1 (en) | 2021-02-01 | 2025-11-06 | Csl Behring Ag | Method of treating or preventing an adverse secondary neurological outcome following a haemorrhagic stroke |
| WO2022234070A1 (en) | 2021-05-07 | 2022-11-10 | Csl Behring Ag | Expression system for producing a recombinant haptoglobin (hp) beta chain |
| TW202317607A (zh) | 2021-07-06 | 2023-05-01 | 大陸商蘇州康寧杰瑞生物科技有限公司 | 融合蛋白及其應用 |
| CA3224743A1 (en) | 2021-07-14 | 2023-01-19 | Beijing Ql Biopharmaceutical Co., Ltd. | Fusion polypeptides for metabolic disorders |
| WO2023225534A1 (en) | 2022-05-18 | 2023-11-23 | Protomer Technologies Inc. | Aromatic boron-containing compounds and related insulin analogs |
| CN114774496B (zh) * | 2022-06-21 | 2022-10-04 | 北京惠之衡生物科技有限公司 | 一种高密度发酵制备glp-1类似物的方法 |
| KR20250058762A (ko) | 2022-09-02 | 2025-04-30 | 체에스엘 베링 아게 | 과도한 발기 반응 또는 발기 부전을 치료하거나 예방하는 데 사용하기 위한 합토글로빈 |
| CN120187750A (zh) | 2022-09-21 | 2025-06-20 | 瑞泽恩制药公司 | 治疗肥胖、糖尿病和肝功能障碍的方法 |
| WO2024068848A1 (en) | 2022-09-28 | 2024-04-04 | Zealand Pharma A/S | Methods for treating obesity |
| WO2024123812A1 (en) | 2022-12-05 | 2024-06-13 | Shattuck Labs, Inc. | Fusion proteins for the treatment of cardiometabolic diseases |
| WO2024132147A1 (en) | 2022-12-22 | 2024-06-27 | Lifearc | Galanin-2 receptor agonists |
| WO2024197132A1 (en) | 2023-03-23 | 2024-09-26 | Eli Lilly And Company | Methods of producing fc-containing proteins |
| WO2025069009A1 (en) | 2023-09-29 | 2025-04-03 | Graviton Bioscience Bv | Rock2 inhibitors in the treatment of obesity |
| WO2025122835A2 (en) | 2023-12-08 | 2025-06-12 | Eli Lilly And Company | Methods of producing fc-containing proteins |
| US20250270274A1 (en) * | 2024-02-26 | 2025-08-28 | Serpentide Inc. | Super long-lasting glp1 or glp1/gip analogue drug for type-2 diabetes and obesity |
| WO2025257801A1 (en) | 2024-06-13 | 2025-12-18 | CSL Innovation Pty Ltd | Heme-binding protein for the treatment of ischemia-reperfusion injury (iri) |
| WO2025256577A1 (zh) * | 2024-06-13 | 2025-12-18 | 上海银诺医药技术有限公司 | 一种改进的glp-1多肽的融合蛋白和应用 |
| WO2026073030A1 (en) | 2024-09-27 | 2026-04-02 | Carmot Therapeutics Inc. | Combination therapy of peptide tyrosine-tyrosine (pyy) analogues and glp-1r agonists |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4399216A (en) | 1980-02-25 | 1983-08-16 | The Trustees Of Columbia University | Processes for inserting DNA into eucaryotic cells and for producing proteinaceous materials |
| NZ201705A (en) | 1981-08-31 | 1986-03-14 | Genentech Inc | Recombinant dna method for production of hepatitis b surface antigen in yeast |
| US4943529A (en) | 1982-05-19 | 1990-07-24 | Gist-Brocades Nv | Kluyveromyces as a host strain |
| AU3145184A (en) | 1983-08-16 | 1985-02-21 | Zymogenetics Inc. | High expression of foreign genes in schizosaccharomyces pombe |
| US4879231A (en) | 1984-10-30 | 1989-11-07 | Phillips Petroleum Company | Transformation of yeasts of the genus pichia |
| GB8610600D0 (en) | 1986-04-30 | 1986-06-04 | Novo Industri As | Transformation of trichoderma |
| WO1988007089A1 (en) * | 1987-03-18 | 1988-09-22 | Medical Research Council | Altered antibodies |
| US5010182A (en) | 1987-07-28 | 1991-04-23 | Chiron Corporation | DNA constructs containing a Kluyveromyces alpha factor leader sequence for directing secretion of heterologous polypeptides |
| AU4005289A (en) | 1988-08-25 | 1990-03-01 | Smithkline Beecham Corporation | Recombinant saccharomyces |
| FR2646437B1 (fr) | 1989-04-28 | 1991-08-30 | Transgene Sa | Nouvelles sequences d'adn, leur application en tant que sequence codant pour un peptide signal pour la secretion de proteines matures par des levures recombinantes, cassettes d'expression, levures transformees et procede de preparation de proteines correspondant |
| ATE144281T1 (de) | 1989-04-28 | 1996-11-15 | Rhein Biotech Proz & Prod Gmbh | Hefezellen der gattung-schwanniomyces |
| EP0402226A1 (en) | 1989-06-06 | 1990-12-12 | Institut National De La Recherche Agronomique | Transformation vectors for yeast yarrowia |
| FR2649120B1 (fr) | 1989-06-30 | 1994-01-28 | Cayla | Nouvelle souche et ses mutants de champignons filamenteux, procede de production de proteines recombinantes a l'aide de ladite souche et souches et proteines obtenues selon ce procede |
| JP3560609B2 (ja) * | 1992-11-13 | 2004-09-02 | イミュネックス・コーポレーション | Elkリガンドと呼ばれる新規なサイトカイン |
| JPH10503371A (ja) * | 1994-07-29 | 1998-03-31 | スミスクライン・ビーチャム・パブリック・リミテッド・カンパニー | 新規化合物 |
| GB9511935D0 (en) | 1995-06-13 | 1995-08-09 | Smithkline Beecham Plc | Novel compound |
| US5723125A (en) * | 1995-12-28 | 1998-03-03 | Tanox Biosystems, Inc. | Hybrid with interferon-alpha and an immunoglobulin Fc linked through a non-immunogenic peptide |
| US6750334B1 (en) | 1996-02-02 | 2004-06-15 | Repligen Corporation | CTLA4-immunoglobulin fusion proteins having modified effector functions and uses therefor |
| US6277819B1 (en) | 1996-08-30 | 2001-08-21 | Eli Lilly And Company | Use of GLP-1 or analogs in treatment of myocardial infarction |
| US6006753A (en) | 1996-08-30 | 1999-12-28 | Eli Lilly And Company | Use of GLP-1 or analogs to abolish catabolic changes after surgery |
| UA65549C2 (uk) | 1996-11-05 | 2004-04-15 | Елі Ліллі Енд Компані | Спосіб регулювання ожиріння шляхом периферійного введення аналогів та похідних glp-1 (варіанти) та фармацевтична композиція |
| US6190909B1 (en) * | 1997-04-17 | 2001-02-20 | Millennium Pharmaceuticals, Inc. | TH2-specific gene |
| SE9802080D0 (sv) | 1998-06-11 | 1998-06-11 | Hellstroem | Pharmaceutical composition for the treatment of functional dyspepsia and/or irritable bowel syndrome and new use of substances therein |
| AU5027299A (en) | 1998-07-31 | 2000-02-28 | Novo Nordisk A/S | Use of glp-1 and analogues for preventing type ii diabetes |
| MY155270A (en) | 1998-09-24 | 2015-09-30 | Lilly Co Eli | Use of glp-1 or analogs in treatment of stroke |
| US6376653B1 (en) * | 1998-09-28 | 2002-04-23 | Smithkline Beecham Plc | Tie2 antagonist antibodies |
| CA2363712C (en) | 1999-05-17 | 2011-05-10 | Conjuchem Inc. | Long lasting insulinotropic peptides |
| ATE424413T1 (de) | 2000-06-16 | 2009-03-15 | Lilly Co Eli | Analoge des glucagon-ähnlichen peptids-1 |
| BR0116024A (pt) * | 2000-12-07 | 2005-12-13 | Lilly Co Eli | Proteìna de fusão heteróloga e uso da mesma |
| WO2002079415A2 (en) * | 2001-03-30 | 2002-10-10 | Lexigen Pharmaceuticals Corp. | Reducing the immunogenicity of fusion proteins |
| US6900292B2 (en) * | 2001-08-17 | 2005-05-31 | Lee-Hwei K. Sun | Fc fusion proteins of human erythropoietin with increased biological activities |
| EP2261250B1 (en) * | 2001-12-21 | 2015-07-01 | Human Genome Sciences, Inc. | GCSF-Albumin fusion proteins |
| MXPA05013565A (es) * | 2003-06-12 | 2006-03-09 | Lilly Co Eli | Proteinas de fusion analogas al glp-1. |
-
2004
- 2004-06-10 MX MXPA05013565A patent/MXPA05013565A/es active IP Right Grant
- 2004-06-10 PL PL04752589T patent/PL1641823T3/pl unknown
- 2004-06-10 NZ NZ543292A patent/NZ543292A/en not_active IP Right Cessation
- 2004-06-10 AT AT04752589T patent/ATE525395T1/de active
- 2004-06-10 US US10/558,627 patent/US7452966B2/en active Active
- 2004-06-10 CN CN200480015953XA patent/CN1802386B/zh not_active Expired - Lifetime
- 2004-06-10 CN CN201010508567.1A patent/CN101974090B/zh not_active Expired - Lifetime
- 2004-06-10 DK DK04752589.4T patent/DK1641823T3/da active
- 2004-06-10 SI SI200431776T patent/SI1641823T1/sl unknown
- 2004-06-10 JP JP2006533197A patent/JP4629047B2/ja not_active Expired - Lifetime
- 2004-06-10 KR KR1020057023668A patent/KR100758755B1/ko not_active Expired - Lifetime
- 2004-06-10 EP EP11166548A patent/EP2368909A1/en not_active Withdrawn
- 2004-06-10 HR HR20110714T patent/HRP20110714T1/hr unknown
- 2004-06-10 ES ES04752589T patent/ES2371072T3/es not_active Expired - Lifetime
- 2004-06-10 PT PT04752589T patent/PT1641823E/pt unknown
- 2004-06-10 EA EA200600015A patent/EA008831B1/ru active Protection Beyond IP Right Term
- 2004-06-10 UA UAA200511830A patent/UA87458C2/ru unknown
- 2004-06-10 BR BRPI0411132A patent/BRPI0411132B8/pt not_active IP Right Cessation
- 2004-06-10 CA CA2528591A patent/CA2528591C/en not_active Expired - Lifetime
- 2004-06-10 WO PCT/US2004/015595 patent/WO2005000892A2/en not_active Ceased
- 2004-06-10 AU AU2004251145A patent/AU2004251145C1/en active Active
- 2004-06-10 EP EP04752589A patent/EP1641823B1/en not_active Expired - Lifetime
- 2004-06-11 TW TW093116970A patent/TW200507870A/zh unknown
- 2004-06-11 AR ARP040102037A patent/AR044776A1/es unknown
-
2005
- 2005-11-14 IL IL171926A patent/IL171926A/en active IP Right Grant
-
2008
- 2008-10-31 US US12/262,832 patent/US8273854B2/en active Active
-
2011
- 2011-11-03 CY CY20111101059T patent/CY1111991T1/el unknown
-
2015
- 2015-01-30 FR FR15C0010C patent/FR15C0010I2/fr active Active
- 2015-01-30 CY CY2015002C patent/CY2015002I2/el unknown
- 2015-02-02 BE BE2015C007C patent/BE2015C007I2/fr unknown
- 2015-02-03 LT LTPA2015007C patent/LTC1641823I2/lt unknown
- 2015-04-20 HU HUS1500024C patent/HUS1500024I1/hu unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4629047B2 (ja) | Glp−1アナログ複合タンパク質 | |
| EP1641483B1 (en) | Fusion proteins | |
| US7576190B2 (en) | FGF-21 fusion proteins | |
| KR100942864B1 (ko) | Glp-1 융합 단백질 | |
| WO2011153965A1 (zh) | Exendin-4及其类似物的融合蛋白,其制备和应用 | |
| HK1149566B (en) | Glp-1 analog fusion plroteins |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20100427 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20100723 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20101012 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20101110 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131119 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4629047 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R153 | Grant of patent term extension |
Free format text: JAPANESE INTERMEDIATE CODE: R153 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |