JP4629047B2 - Glp−1アナログ複合タンパク質 - Google Patents
Glp−1アナログ複合タンパク質 Download PDFInfo
- Publication number
- JP4629047B2 JP4629047B2 JP2006533197A JP2006533197A JP4629047B2 JP 4629047 B2 JP4629047 B2 JP 4629047B2 JP 2006533197 A JP2006533197 A JP 2006533197A JP 2006533197 A JP2006533197 A JP 2006533197A JP 4629047 B2 JP4629047 B2 JP 4629047B2
- Authority
- JP
- Japan
- Prior art keywords
- gly
- ser
- glu
- val
- pro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- DTHNMHAUYICORS-KTKZVXAJSA-N Glucagon-like peptide 1 Chemical class C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 DTHNMHAUYICORS-KTKZVXAJSA-N 0.000 title claims description 52
- 108090000623 proteins and genes Proteins 0.000 title description 47
- 102000004169 proteins and genes Human genes 0.000 title description 31
- 108020001507 fusion proteins Proteins 0.000 claims description 100
- 102000037865 fusion proteins Human genes 0.000 claims description 100
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Natural products NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 34
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 20
- 108060003951 Immunoglobulin Proteins 0.000 claims description 11
- 102000018358 immunoglobulin Human genes 0.000 claims description 11
- 239000004471 Glycine Substances 0.000 claims description 10
- 210000004899 c-terminal region Anatomy 0.000 claims description 7
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 claims description 5
- 235000004279 alanine Nutrition 0.000 claims description 5
- 208000001072 type 2 diabetes mellitus Diseases 0.000 claims description 5
- 230000001939 inductive effect Effects 0.000 claims description 4
- 208000008589 Obesity Diseases 0.000 claims description 3
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 claims description 3
- 235000020824 obesity Nutrition 0.000 claims description 3
- 230000004580 weight loss Effects 0.000 claims description 3
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 claims description 2
- 230000002950 deficient Effects 0.000 claims description 2
- 239000008194 pharmaceutical composition Substances 0.000 claims 3
- 206010033307 Overweight Diseases 0.000 claims 1
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 claims 1
- 210000004027 cell Anatomy 0.000 description 55
- 238000000034 method Methods 0.000 description 35
- 101800000224 Glucagon-like peptide 1 Proteins 0.000 description 30
- 235000018102 proteins Nutrition 0.000 description 30
- 108020004414 DNA Proteins 0.000 description 28
- 102100040918 Pro-glucagon Human genes 0.000 description 27
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 20
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 20
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 20
- 239000008103 glucose Substances 0.000 description 20
- 239000013598 vector Substances 0.000 description 19
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 16
- 238000011282 treatment Methods 0.000 description 16
- 238000001802 infusion Methods 0.000 description 15
- 238000006467 substitution reaction Methods 0.000 description 14
- 238000001727 in vivo Methods 0.000 description 13
- 230000006870 function Effects 0.000 description 12
- 101100335894 Caenorhabditis elegans gly-8 gene Proteins 0.000 description 11
- 108010086246 Glucagon-Like Peptide-1 Receptor Proteins 0.000 description 11
- 102000007446 Glucagon-Like Peptide-1 Receptor Human genes 0.000 description 11
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Chemical compound CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 11
- 239000012636 effector Substances 0.000 description 11
- 238000010254 subcutaneous injection Methods 0.000 description 11
- 239000007929 subcutaneous injection Substances 0.000 description 11
- 241000700159 Rattus Species 0.000 description 10
- 230000000694 effects Effects 0.000 description 10
- 101001100327 Homo sapiens RNA-binding protein 45 Proteins 0.000 description 9
- 102100038823 RNA-binding protein 45 Human genes 0.000 description 9
- 239000002585 base Substances 0.000 description 9
- 150000007523 nucleic acids Chemical group 0.000 description 9
- 102000004196 processed proteins & peptides Human genes 0.000 description 9
- 102000004190 Enzymes Human genes 0.000 description 8
- 108090000790 Enzymes Proteins 0.000 description 8
- 102000004877 Insulin Human genes 0.000 description 8
- 108090001061 Insulin Proteins 0.000 description 8
- 235000001014 amino acid Nutrition 0.000 description 8
- 229940024606 amino acid Drugs 0.000 description 8
- 230000004071 biological effect Effects 0.000 description 8
- 230000015572 biosynthetic process Effects 0.000 description 8
- 229940125396 insulin Drugs 0.000 description 8
- -1 protoplast fusion Substances 0.000 description 8
- 108091006020 Fc-tagged proteins Proteins 0.000 description 7
- 241000282412 Homo Species 0.000 description 7
- 108010076504 Protein Sorting Signals Proteins 0.000 description 7
- 238000010367 cloning Methods 0.000 description 7
- 230000000295 complement effect Effects 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 7
- 239000003623 enhancer Substances 0.000 description 7
- 230000004927 fusion Effects 0.000 description 7
- 230000003993 interaction Effects 0.000 description 7
- 210000004962 mammalian cell Anatomy 0.000 description 7
- 239000002609 medium Substances 0.000 description 7
- 230000008569 process Effects 0.000 description 7
- 230000009466 transformation Effects 0.000 description 7
- 210000002237 B-cell of pancreatic islet Anatomy 0.000 description 6
- 102000016622 Dipeptidyl Peptidase 4 Human genes 0.000 description 6
- 101000930822 Giardia intestinalis Dipeptidyl-peptidase 4 Proteins 0.000 description 6
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 6
- 241000282567 Macaca fascicularis Species 0.000 description 6
- 125000003275 alpha amino acid group Chemical group 0.000 description 6
- 150000001413 amino acids Chemical class 0.000 description 6
- 210000004369 blood Anatomy 0.000 description 6
- 239000008280 blood Substances 0.000 description 6
- 239000013604 expression vector Substances 0.000 description 6
- 238000001990 intravenous administration Methods 0.000 description 6
- 108020004707 nucleic acids Proteins 0.000 description 6
- 102000039446 nucleic acids Human genes 0.000 description 6
- 229920001184 polypeptide Polymers 0.000 description 6
- GCYXWQUSHADNBF-AAEALURTSA-N preproglucagon 78-108 Chemical class C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 GCYXWQUSHADNBF-AAEALURTSA-N 0.000 description 6
- 238000003127 radioimmunoassay Methods 0.000 description 6
- 150000003839 salts Chemical class 0.000 description 6
- 210000002966 serum Anatomy 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- 238000013518 transcription Methods 0.000 description 6
- 230000035897 transcription Effects 0.000 description 6
- 238000002965 ELISA Methods 0.000 description 5
- 101800004266 Glucagon-like peptide 1(7-37) Proteins 0.000 description 5
- NYHBQMYGNKIUIF-UUOKFMHZSA-N Guanosine Chemical compound C1=NC=2C(=O)NC(N)=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O NYHBQMYGNKIUIF-UUOKFMHZSA-N 0.000 description 5
- 239000004472 Lysine Substances 0.000 description 5
- 108091028043 Nucleic acid sequence Proteins 0.000 description 5
- 108020004511 Recombinant DNA Proteins 0.000 description 5
- 239000000427 antigen Substances 0.000 description 5
- 108091007433 antigens Proteins 0.000 description 5
- 102000036639 antigens Human genes 0.000 description 5
- 239000002299 complementary DNA Substances 0.000 description 5
- 206010012601 diabetes mellitus Diseases 0.000 description 5
- 230000008030 elimination Effects 0.000 description 5
- 238000003379 elimination reaction Methods 0.000 description 5
- 239000012634 fragment Substances 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 108020004999 messenger RNA Proteins 0.000 description 5
- 108091008146 restriction endonucleases Proteins 0.000 description 5
- 230000001629 suppression Effects 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 4
- 241000282693 Cercopithecidae Species 0.000 description 4
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 4
- 102000051325 Glucagon Human genes 0.000 description 4
- 108060003199 Glucagon Proteins 0.000 description 4
- 101001015516 Homo sapiens Glucagon-like peptide 1 receptor Proteins 0.000 description 4
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 4
- 239000005089 Luciferase Substances 0.000 description 4
- 241000829100 Macaca mulatta polyomavirus 1 Species 0.000 description 4
- IQFYYKKMVGJFEH-XLPZGREQSA-N Thymidine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 IQFYYKKMVGJFEH-XLPZGREQSA-N 0.000 description 4
- 230000004913 activation Effects 0.000 description 4
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 4
- 238000013459 approach Methods 0.000 description 4
- 230000037396 body weight Effects 0.000 description 4
- 230000001419 dependent effect Effects 0.000 description 4
- MASNOZXLGMXCHN-ZLPAWPGGSA-N glucagon Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 MASNOZXLGMXCHN-ZLPAWPGGSA-N 0.000 description 4
- 229960004666 glucagon Drugs 0.000 description 4
- 230000028993 immune response Effects 0.000 description 4
- 230000006698 induction Effects 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 239000013612 plasmid Substances 0.000 description 4
- 108091033319 polynucleotide Proteins 0.000 description 4
- 102000040430 polynucleotide Human genes 0.000 description 4
- 239000002157 polynucleotide Substances 0.000 description 4
- 239000000523 sample Substances 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- 208000024891 symptom Diseases 0.000 description 4
- 238000001890 transfection Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 241000222120 Candida <Saccharomycetales> Species 0.000 description 3
- 102100024746 Dihydrofolate reductase Human genes 0.000 description 3
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 3
- 108060001084 Luciferase Proteins 0.000 description 3
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- 108700008625 Reporter Genes Proteins 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 241000700605 Viruses Species 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 239000000654 additive Substances 0.000 description 3
- 238000003556 assay Methods 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 230000015556 catabolic process Effects 0.000 description 3
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 3
- 239000013599 cloning vector Substances 0.000 description 3
- 108020001096 dihydrofolate reductase Proteins 0.000 description 3
- 238000010790 dilution Methods 0.000 description 3
- 239000012895 dilution Substances 0.000 description 3
- 239000000539 dimer Substances 0.000 description 3
- 231100000673 dose–response relationship Toxicity 0.000 description 3
- 238000004520 electroporation Methods 0.000 description 3
- 230000012010 growth Effects 0.000 description 3
- 238000003018 immunoassay Methods 0.000 description 3
- 238000011534 incubation Methods 0.000 description 3
- 230000001404 mediated effect Effects 0.000 description 3
- 238000010369 molecular cloning Methods 0.000 description 3
- 238000010899 nucleation Methods 0.000 description 3
- 239000010452 phosphate Substances 0.000 description 3
- 230000036470 plasma concentration Effects 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 238000001742 protein purification Methods 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 230000003248 secreting effect Effects 0.000 description 3
- 230000028327 secretion Effects 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 3
- UHDGCWIWMRVCDJ-UHFFFAOYSA-N 1-beta-D-Xylofuranosyl-NH-Cytosine Natural products O=C1N=C(N)C=CN1C1C(O)C(O)C(CO)O1 UHDGCWIWMRVCDJ-UHFFFAOYSA-N 0.000 description 2
- GZCWLCBFPRFLKL-UHFFFAOYSA-N 1-prop-2-ynoxypropan-2-ol Chemical compound CC(O)COCC#C GZCWLCBFPRFLKL-UHFFFAOYSA-N 0.000 description 2
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 2
- OSJPPGNTCRNQQC-UWTATZPHSA-N 3-phospho-D-glyceric acid Chemical compound OC(=O)[C@H](O)COP(O)(O)=O OSJPPGNTCRNQQC-UWTATZPHSA-N 0.000 description 2
- 102000013563 Acid Phosphatase Human genes 0.000 description 2
- 108010051457 Acid Phosphatase Proteins 0.000 description 2
- 229930024421 Adenine Natural products 0.000 description 2
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 2
- 108700028369 Alleles Proteins 0.000 description 2
- 241000228212 Aspergillus Species 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- DWRXFEITVBNRMK-UHFFFAOYSA-N Beta-D-1-Arabinofuranosylthymine Natural products O=C1NC(=O)C(C)=CN1C1C(O)C(O)C(CO)O1 DWRXFEITVBNRMK-UHFFFAOYSA-N 0.000 description 2
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 2
- 241000283707 Capra Species 0.000 description 2
- 108020004705 Codon Proteins 0.000 description 2
- MIKUYHXYGGJMLM-GIMIYPNGSA-N Crotonoside Natural products C1=NC2=C(N)NC(=O)N=C2N1[C@H]1O[C@@H](CO)[C@H](O)[C@@H]1O MIKUYHXYGGJMLM-GIMIYPNGSA-N 0.000 description 2
- UHDGCWIWMRVCDJ-PSQAKQOGSA-N Cytidine Natural products O=C1N=C(N)C=CN1[C@@H]1[C@@H](O)[C@@H](O)[C@H](CO)O1 UHDGCWIWMRVCDJ-PSQAKQOGSA-N 0.000 description 2
- 241000701022 Cytomegalovirus Species 0.000 description 2
- NYHBQMYGNKIUIF-UHFFFAOYSA-N D-guanosine Natural products C1=2NC(N)=NC(=O)C=2N=CN1C1OC(CO)C(O)C1O NYHBQMYGNKIUIF-UHFFFAOYSA-N 0.000 description 2
- 101710088194 Dehydrogenase Proteins 0.000 description 2
- 108090000204 Dipeptidase 1 Proteins 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 241000233866 Fungi Species 0.000 description 2
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 2
- 102000005731 Glucose-6-phosphate isomerase Human genes 0.000 description 2
- 108010070600 Glucose-6-phosphate isomerase Proteins 0.000 description 2
- BCCRXDTUTZHDEU-VKHMYHEASA-N Gly-Ser Chemical compound NCC(=O)N[C@@H](CO)C(O)=O BCCRXDTUTZHDEU-VKHMYHEASA-N 0.000 description 2
- ZRALSGWEFCBTJO-UHFFFAOYSA-N Guanidine Chemical compound NC(N)=N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 description 2
- 241000238631 Hexapoda Species 0.000 description 2
- 102000018071 Immunoglobulin Fc Fragments Human genes 0.000 description 2
- 108010091135 Immunoglobulin Fc Fragments Proteins 0.000 description 2
- 241001138401 Kluyveromyces lactis Species 0.000 description 2
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 2
- 125000000174 L-prolyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])([H])[C@@]1([H])C(*)=O 0.000 description 2
- 102000003960 Ligases Human genes 0.000 description 2
- 108090000364 Ligases Proteins 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Natural products OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 241000235648 Pichia Species 0.000 description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 description 2
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 2
- 108020005091 Replication Origin Proteins 0.000 description 2
- 241000235070 Saccharomyces Species 0.000 description 2
- 241000235347 Schizosaccharomyces pombe Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 241000256248 Spodoptera Species 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 108091008874 T cell receptors Proteins 0.000 description 2
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 2
- 210000001744 T-lymphocyte Anatomy 0.000 description 2
- 101150006914 TRP1 gene Proteins 0.000 description 2
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 2
- DRTQHJPVMGBUCF-XVFCMESISA-N Uridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-XVFCMESISA-N 0.000 description 2
- MZVQCMJNVPIDEA-UHFFFAOYSA-N [CH2]CN(CC)CC Chemical group [CH2]CN(CC)CC MZVQCMJNVPIDEA-UHFFFAOYSA-N 0.000 description 2
- 229960000643 adenine Drugs 0.000 description 2
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 2
- 125000000539 amino acid group Chemical group 0.000 description 2
- 210000000612 antigen-presenting cell Anatomy 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- IQFYYKKMVGJFEH-UHFFFAOYSA-N beta-L-thymidine Natural products O=C1NC(=O)C(C)=CN1C1OC(CO)C(O)C1 IQFYYKKMVGJFEH-UHFFFAOYSA-N 0.000 description 2
- 102000006635 beta-lactamase Human genes 0.000 description 2
- 230000000903 blocking effect Effects 0.000 description 2
- 239000001506 calcium phosphate Substances 0.000 description 2
- 229910000389 calcium phosphate Inorganic materials 0.000 description 2
- 235000011010 calcium phosphates Nutrition 0.000 description 2
- 230000022131 cell cycle Effects 0.000 description 2
- 210000000170 cell membrane Anatomy 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000003776 cleavage reaction Methods 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- UHDGCWIWMRVCDJ-ZAKLUEHWSA-N cytidine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O1 UHDGCWIWMRVCDJ-ZAKLUEHWSA-N 0.000 description 2
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical compound NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 230000001934 delay Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 210000003527 eukaryotic cell Anatomy 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 230000002068 genetic effect Effects 0.000 description 2
- 230000002641 glycemic effect Effects 0.000 description 2
- 229940029575 guanosine Drugs 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 230000001900 immune effect Effects 0.000 description 2
- 230000005847 immunogenicity Effects 0.000 description 2
- 229940072221 immunoglobulins Drugs 0.000 description 2
- 230000003914 insulin secretion Effects 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000003550 marker Substances 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 150000007522 mineralic acids Chemical class 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 201000000050 myeloid neoplasm Diseases 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 238000007911 parenteral administration Methods 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 102000013415 peroxidase activity proteins Human genes 0.000 description 2
- 108040007629 peroxidase activity proteins Proteins 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 210000001938 protoplast Anatomy 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 230000000241 respiratory effect Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 230000007017 scission Effects 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 229940104230 thymidine Drugs 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 2
- 241000701161 unidentified adenovirus Species 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- DIGQNXIGRZPYDK-WKSCXVIASA-N (2R)-6-amino-2-[[2-[[(2S)-2-[[2-[[(2R)-2-[[(2S)-2-[[(2R,3S)-2-[[2-[[(2S)-2-[[2-[[(2S)-2-[[(2S)-2-[[(2R)-2-[[(2S,3S)-2-[[(2R)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[2-[[(2S)-2-[[(2R)-2-[[2-[[2-[[2-[(2-amino-1-hydroxyethylidene)amino]-3-carboxy-1-hydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxypropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1,5-dihydroxy-5-iminopentylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxybutylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1,3-dihydroxypropylidene]amino]-1-hydroxyethylidene]amino]-1-hydroxy-3-sulfanylpropylidene]amino]-1-hydroxyethylidene]amino]hexanoic acid Chemical compound C[C@@H]([C@@H](C(=N[C@@H](CS)C(=N[C@@H](C)C(=N[C@@H](CO)C(=NCC(=N[C@@H](CCC(=N)O)C(=NC(CS)C(=N[C@H]([C@H](C)O)C(=N[C@H](CS)C(=N[C@H](CO)C(=NCC(=N[C@H](CS)C(=NCC(=N[C@H](CCCCN)C(=O)O)O)O)O)O)O)O)O)O)O)O)O)O)O)N=C([C@H](CS)N=C([C@H](CO)N=C([C@H](CO)N=C([C@H](C)N=C(CN=C([C@H](CO)N=C([C@H](CS)N=C(CN=C(C(CS)N=C(C(CC(=O)O)N=C(CN)O)O)O)O)O)O)O)O)O)O)O)O DIGQNXIGRZPYDK-WKSCXVIASA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- OLXZPDWKRNYJJZ-RRKCRQDMSA-N 2'-deoxyadenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@H]1C[C@H](O)[C@@H](CO)O1 OLXZPDWKRNYJJZ-RRKCRQDMSA-N 0.000 description 1
- OWEFQTXQEHYDEJ-UHFFFAOYSA-N 2,3-dihydroxypropanal diphosphono hydrogen phosphate Chemical compound OCC(O)C=O.OP(O)(=O)OP(O)(=O)OP(O)(O)=O OWEFQTXQEHYDEJ-UHFFFAOYSA-N 0.000 description 1
- JUSXLWAFYVKNLT-UHFFFAOYSA-N 2-bromobenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=CC=C1Br JUSXLWAFYVKNLT-UHFFFAOYSA-N 0.000 description 1
- YRNWIFYIFSBPAU-UHFFFAOYSA-N 4-[4-(dimethylamino)phenyl]-n,n-dimethylaniline Chemical compound C1=CC(N(C)C)=CC=C1C1=CC=C(N(C)C)C=C1 YRNWIFYIFSBPAU-UHFFFAOYSA-N 0.000 description 1
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 1
- 102000007469 Actins Human genes 0.000 description 1
- 108010085238 Actins Proteins 0.000 description 1
- 108010041525 Alanine racemase Proteins 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- 101710187573 Alcohol dehydrogenase 2 Proteins 0.000 description 1
- 101710133776 Alcohol dehydrogenase class-3 Proteins 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 1
- 241000713842 Avian sarcoma virus Species 0.000 description 1
- 241000894006 Bacteria Species 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 1
- 241000222122 Candida albicans Species 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 208000000668 Chronic Pancreatitis Diseases 0.000 description 1
- 108091062157 Cis-regulatory element Proteins 0.000 description 1
- 108091026890 Coding region Proteins 0.000 description 1
- 108020004394 Complementary RNA Proteins 0.000 description 1
- 241000699802 Cricetulus griseus Species 0.000 description 1
- IVOMOUWHDPKRLL-KQYNXXCUSA-N Cyclic adenosine monophosphate Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=CN=C2N)=C2N=C1 IVOMOUWHDPKRLL-KQYNXXCUSA-N 0.000 description 1
- MNQZXJOMYWMBOU-VKHMYHEASA-N D-glyceraldehyde Chemical compound OC[C@@H](O)C=O MNQZXJOMYWMBOU-VKHMYHEASA-N 0.000 description 1
- KDXKERNSBIXSRK-RXMQYKEDSA-N D-lysine group Chemical group N[C@H](CCCCN)C(=O)O KDXKERNSBIXSRK-RXMQYKEDSA-N 0.000 description 1
- 102000053602 DNA Human genes 0.000 description 1
- 102000012410 DNA Ligases Human genes 0.000 description 1
- 108010061982 DNA Ligases Proteins 0.000 description 1
- 241000255581 Drosophila <fruit fly, genus> Species 0.000 description 1
- 108010042407 Endonucleases Proteins 0.000 description 1
- 102000004533 Endonucleases Human genes 0.000 description 1
- 241000206602 Eukaryota Species 0.000 description 1
- 108010087819 Fc receptors Proteins 0.000 description 1
- 102000009109 Fc receptors Human genes 0.000 description 1
- 241000700662 Fowlpox virus Species 0.000 description 1
- 108700039691 Genetic Promoter Regions Proteins 0.000 description 1
- 102000006395 Globulins Human genes 0.000 description 1
- 108010044091 Globulins Proteins 0.000 description 1
- 108010088406 Glucagon-Like Peptides Proteins 0.000 description 1
- 108010073178 Glucan 1,4-alpha-Glucosidase Proteins 0.000 description 1
- 102100022624 Glucoamylase Human genes 0.000 description 1
- 102000030595 Glucokinase Human genes 0.000 description 1
- 108010021582 Glucokinase Proteins 0.000 description 1
- 208000002705 Glucose Intolerance Diseases 0.000 description 1
- 241001149669 Hanseniaspora Species 0.000 description 1
- 241000700721 Hepatitis B virus Species 0.000 description 1
- 108091027305 Heteroduplex Proteins 0.000 description 1
- 229920000209 Hexadimethrine bromide Polymers 0.000 description 1
- 102000005548 Hexokinase Human genes 0.000 description 1
- 108700040460 Hexokinases Proteins 0.000 description 1
- 241000701109 Human adenovirus 2 Species 0.000 description 1
- 208000013016 Hypoglycemia Diseases 0.000 description 1
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 1
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 1
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 1
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 1
- 241000235649 Kluyveromyces Species 0.000 description 1
- 241000235058 Komagataella pastoris Species 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 1
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 1
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- 208000002720 Malnutrition Diseases 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 102000003792 Metallothionein Human genes 0.000 description 1
- 108090000157 Metallothionein Proteins 0.000 description 1
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 description 1
- 241000588652 Neisseria gonorrhoeae Species 0.000 description 1
- 229930193140 Neomycin Natural products 0.000 description 1
- 241000221960 Neurospora Species 0.000 description 1
- 241000221961 Neurospora crassa Species 0.000 description 1
- 241000080590 Niso Species 0.000 description 1
- 102000016387 Pancreatic elastase Human genes 0.000 description 1
- 108010067372 Pancreatic elastase Proteins 0.000 description 1
- 206010033645 Pancreatitis Diseases 0.000 description 1
- 206010033647 Pancreatitis acute Diseases 0.000 description 1
- 206010033649 Pancreatitis chronic Diseases 0.000 description 1
- 241001631646 Papillomaviridae Species 0.000 description 1
- 241000228143 Penicillium Species 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 102000012288 Phosphopyruvate Hydratase Human genes 0.000 description 1
- 108010022181 Phosphopyruvate Hydratase Proteins 0.000 description 1
- 108091000080 Phosphotransferase Proteins 0.000 description 1
- 241001505332 Polyomavirus sp. Species 0.000 description 1
- 108010058003 Proglucagon Proteins 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 108010011939 Pyruvate Decarboxylase Proteins 0.000 description 1
- 102000013009 Pyruvate Kinase Human genes 0.000 description 1
- 108020005115 Pyruvate Kinase Proteins 0.000 description 1
- 230000006819 RNA synthesis Effects 0.000 description 1
- 230000010799 Receptor Interactions Effects 0.000 description 1
- 208000017442 Retinal disease Diseases 0.000 description 1
- 244000253911 Saccharomyces fragilis Species 0.000 description 1
- 241000235343 Saccharomycetales Species 0.000 description 1
- 241000311088 Schwanniomyces Species 0.000 description 1
- 206010039897 Sedation Diseases 0.000 description 1
- 229920005654 Sephadex Polymers 0.000 description 1
- 239000012507 Sephadex™ Substances 0.000 description 1
- 238000012300 Sequence Analysis Methods 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 229940100389 Sulfonylurea Drugs 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- 108091036066 Three prime untranslated region Proteins 0.000 description 1
- 102000006601 Thymidine Kinase Human genes 0.000 description 1
- 108020004440 Thymidine kinase Proteins 0.000 description 1
- 241001149964 Tolypocladium Species 0.000 description 1
- 101710120037 Toxin CcdB Proteins 0.000 description 1
- 241000223259 Trichoderma Species 0.000 description 1
- 102000005924 Triose-Phosphate Isomerase Human genes 0.000 description 1
- 108700015934 Triose-phosphate isomerases Proteins 0.000 description 1
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 1
- 206010067584 Type 1 diabetes mellitus Diseases 0.000 description 1
- IVOMOUWHDPKRLL-UHFFFAOYSA-N UNPD107823 Natural products O1C2COP(O)(=O)OC2C(O)C1N1C(N=CN=C2N)=C2N=C1 IVOMOUWHDPKRLL-UHFFFAOYSA-N 0.000 description 1
- 108020005202 Viral DNA Proteins 0.000 description 1
- IXKSXJFAGXLQOQ-XISFHERQSA-N WHWLQLKPGQPMY Chemical compound C([C@@H](C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(O)=O)NC(=O)[C@@H](N)CC=1C2=CC=CC=C2NC=1)C1=CNC=N1 IXKSXJFAGXLQOQ-XISFHERQSA-N 0.000 description 1
- 241000235013 Yarrowia Species 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 201000003229 acute pancreatitis Diseases 0.000 description 1
- 230000003044 adaptive effect Effects 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 229960005305 adenosine Drugs 0.000 description 1
- 102000030621 adenylate cyclase Human genes 0.000 description 1
- 108060000200 adenylate cyclase Proteins 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- LFVVNPBBFUSSHL-UHFFFAOYSA-N alexidine Chemical compound CCCCC(CC)CNC(=N)NC(=N)NCCCCCCNC(=N)NC(=N)NCC(CC)CCCC LFVVNPBBFUSSHL-UHFFFAOYSA-N 0.000 description 1
- 229950010221 alexidine Drugs 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229910001854 alkali hydroxide Inorganic materials 0.000 description 1
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 description 1
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003368 amide group Chemical group 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 238000012870 ammonium sulfate precipitation Methods 0.000 description 1
- 239000003957 anion exchange resin Substances 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 229940088710 antibiotic agent Drugs 0.000 description 1
- 230000005875 antibody response Effects 0.000 description 1
- 239000003472 antidiabetic agent Substances 0.000 description 1
- 239000004599 antimicrobial Substances 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 235000009582 asparagine Nutrition 0.000 description 1
- 229960001230 asparagine Drugs 0.000 description 1
- 150000001507 asparagine derivatives Chemical class 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 108010051210 beta-Fructofuranosidase Proteins 0.000 description 1
- DRTQHJPVMGBUCF-PSQAKQOGSA-N beta-L-uridine Natural products O[C@H]1[C@@H](O)[C@H](CO)O[C@@H]1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-PSQAKQOGSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000001110 calcium chloride Substances 0.000 description 1
- 229910001628 calcium chloride Inorganic materials 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 238000005341 cation exchange Methods 0.000 description 1
- 230000020411 cell activation Effects 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 230000010307 cell transformation Effects 0.000 description 1
- 230000007969 cellular immunity Effects 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 230000002759 chromosomal effect Effects 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003184 complementary RNA Substances 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 230000009260 cross reactivity Effects 0.000 description 1
- 210000004748 cultured cell Anatomy 0.000 description 1
- 229940095074 cyclic amp Drugs 0.000 description 1
- 230000009089 cytolysis Effects 0.000 description 1
- 230000001461 cytolytic effect Effects 0.000 description 1
- 229940104302 cytosine Drugs 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 238000012217 deletion Methods 0.000 description 1
- 230000037430 deletion Effects 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- 239000005549 deoxyribonucleoside Substances 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 201000006549 dyspepsia Diseases 0.000 description 1
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 238000012869 ethanol precipitation Methods 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000001605 fetal effect Effects 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 229930182830 galactose Natural products 0.000 description 1
- 230000030136 gastric emptying Effects 0.000 description 1
- 230000030135 gastric motility Effects 0.000 description 1
- 238000002523 gelfiltration Methods 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 208000004104 gestational diabetes Diseases 0.000 description 1
- UKVFVQPAANCXIL-FJVFSOETSA-N glp-1 (1-37) amide Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 UKVFVQPAANCXIL-FJVFSOETSA-N 0.000 description 1
- 108010063245 glucagon-like peptide 1 (7-36)amide Proteins 0.000 description 1
- 229960001031 glucose Drugs 0.000 description 1
- 238000007446 glucose tolerance test Methods 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 230000002414 glycolytic effect Effects 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 229960004198 guanidine Drugs 0.000 description 1
- 125000000487 histidyl group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C([H])=N1 0.000 description 1
- 230000004727 humoral immunity Effects 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 230000002218 hypoglycaemic effect Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000006054 immunological memory Effects 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 230000008991 intestinal motility Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 239000001573 invertase Substances 0.000 description 1
- 235000011073 invertase Nutrition 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- 208000002551 irritable bowel syndrome Diseases 0.000 description 1
- 238000001155 isoelectric focusing Methods 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 210000003292 kidney cell Anatomy 0.000 description 1
- 208000017169 kidney disease Diseases 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- 210000005265 lung cell Anatomy 0.000 description 1
- 239000008176 lyophilized powder Substances 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- RLSSMJSEOOYNOY-UHFFFAOYSA-N m-cresol Chemical compound CC1=CC=CC(O)=C1 RLSSMJSEOOYNOY-UHFFFAOYSA-N 0.000 description 1
- 230000012976 mRNA stabilization Effects 0.000 description 1
- 238000013507 mapping Methods 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229940100630 metacresol Drugs 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 231100000350 mutagenesis Toxicity 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 208000010125 myocardial infarction Diseases 0.000 description 1
- 210000004897 n-terminal region Anatomy 0.000 description 1
- 230000003880 negative regulation of appetite Effects 0.000 description 1
- 229960004927 neomycin Drugs 0.000 description 1
- 201000001119 neuropathy Diseases 0.000 description 1
- 230000007823 neuropathy Effects 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 235000018343 nutrient deficiency Nutrition 0.000 description 1
- 229920002113 octoxynol Polymers 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000006174 pH buffer Substances 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 208000033808 peripheral neuropathy Diseases 0.000 description 1
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 1
- 102000020233 phosphotransferase Human genes 0.000 description 1
- 230000029537 positive regulation of insulin secretion Effects 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 201000009104 prediabetes syndrome Diseases 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 230000000069 prophylactic effect Effects 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 238000009163 protein therapy Methods 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000001850 reproductive effect Effects 0.000 description 1
- 238000002271 resection Methods 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 238000004007 reversed phase HPLC Methods 0.000 description 1
- 239000002342 ribonucleoside Substances 0.000 description 1
- 230000036186 satiety Effects 0.000 description 1
- 235000019627 satiety Nutrition 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000036280 sedation Effects 0.000 description 1
- 210000000717 sertoli cell Anatomy 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 238000011895 specific detection Methods 0.000 description 1
- 238000007619 statistical method Methods 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 125000001424 substituent group Chemical group 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 238000004114 suspension culture Methods 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- 150000003548 thiazolidines Chemical class 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
- 230000005030 transcription termination Effects 0.000 description 1
- 238000003151 transfection method Methods 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 229940035893 uracil Drugs 0.000 description 1
- DRTQHJPVMGBUCF-UHFFFAOYSA-N uracil arabinoside Natural products OC1C(O)C(CO)OC1N1C(=O)NC(=O)C=C1 DRTQHJPVMGBUCF-UHFFFAOYSA-N 0.000 description 1
- 229940045145 uridine Drugs 0.000 description 1
- 239000004474 valine Substances 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/26—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against hormones ; against hormone releasing or inhibiting factors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/48—Drugs for disorders of the endocrine system of the pancreatic hormones
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/475—Growth factors; Growth regulators
- C07K14/50—Fibroblast growth factors [FGF]
- C07K14/503—Fibroblast growth factors [FGF] basic FGF [bFGF]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/605—Glucagons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biochemistry (AREA)
- Endocrinology (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Diabetes (AREA)
- Engineering & Computer Science (AREA)
- Toxicology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Veterinary Medicine (AREA)
- Gastroenterology & Hepatology (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Zoology (AREA)
- Public Health (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Obesity (AREA)
- Hematology (AREA)
- Immunology (AREA)
- Child & Adolescent Psychology (AREA)
- Epidemiology (AREA)
- Emergency Medicine (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Description
a) (SEQ ID NO:1)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)、
b) (SEQ ID NO:2)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Lys−Asn−Gly−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)、
c) (SEQ ID NO:3)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly−Pro
(配列中、Xaa8はGlyとValから選択される)、
d) (SEQ ID NO:4)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Lys−Asn−Gly−Gly−Pro
(配列中、Xaa8はGlyとValから選択される)、
e) (SEQ ID NO:5)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)、
f) (SEQ ID NO:6)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Lys−Asn−Gly−Gly
(配列中、Xaa8はGlyとValから選択される)
から成るグループから選択された配列を含み、
Ala−Glu−Ser−Lys−Tyr−Gly−Pro−Pro−
Cys−Pro−Pro−Cys−Pro−Ala−Pro−Xaa16−
Xaa17−Xaa18−Gly−Gly−Pro−Ser−Val−Phe−
Leu−Phe−Pro−Pro−Lys−Pro−Lys−Asp−
Thr−Leu−Met−Ile−Ser−Arg−Thr−Pro−
Glu−Val−Thr−Cys−Val−Val−Val−Asp−
Val−Ser−Gln−Glu−Asp−Pro−Glu−Val−
Gln−Phe−Asn−Trp−Tyr−Val−Asp−Gly−
Val−Glu−Val−His−Asn−Ala−Lys−Thr−
Lys−Pro−Arg−Glu−Glu−Gln−Phe−Xaa80−
Ser−Thr−Tyr−Arg−Val−Val−Ser−Val−
Leu−Thr−Val−Leu−His−Gln−Asp−Trp−
Leu−Asn−Gly−Lys−Glu−Tyr−Lys−Cys−
Lys−Val−Ser−Asn−Lys−Gly−Leu−Pro−
Ser−Ser−Ile−Glu−Lys−Thr−Ile−Ser−
Lys−Ala−Lys−Gly−Gln−Pro−Arg−Glu−
Pro−Gln−Val−Tyr−Thr−Leu−Pro−Pro−
Ser−Gln−Glu−Glu−Met−Thr−Lys−Asn−
Gln−Val−Ser−Leu−Thr−Cys−Leu−Val−
Lys−Gly−Phe−Tyr−Pro−Ser−Asp−Ile−
Ala−Val−Glu−Trp−Glu−Ser−Asn−Gly−
Gln−Pro−Glu−Asn−Asn−Tyr−Lys−Thr−
Thr−Pro−Pro−Val−Leu−Asp−Ser−Asp−
Gly−Ser−Phe−Phe−Leu−Tyr−Ser−Arg−
Leu−Thr−Val−Asp−Lys−Ser−Arg−Trp−
Gln−Glu−Gly−Asn−Val−Phe−Ser−Cys−
Ser−Val−Met−His−Glu−Ala−Leu−His−
Asn−His−Tyr−Thr−Gln−Lys−Ser−Leu−
Ser−Leu−Ser−Leu−Gly−Xaa230
(SEQ ID NO:7)
(配列中、16番目のXaaは、Pro、またはGluであり、
17番目のXaaは、Phe、Val、またはAlaであり、
18番目のXaaは、Leu、Glu、またはAlaであり、
80番目のXaaは、Asn、または Alaであり、
230番目のXaaは、Lys、または欠損である)、
以上のSEQ ID NO:7の配列を含む免疫グロブリンのFc部分と融合されている。
7His−Ala−Glu−10Gly−Thr−Phe−Thr−Ser−15Asp−Val−Ser−Ser−Tyr−20Leu−Glu−Gly−Gln−Ala−
25Ala−Lys−Glu−Phe−Ile−30Ala−Trp−Leu−Val−Lys−35Gly−Arg−37Gly(SEQ ID NO:9)
である。
His−Gly−Glu−Gly−Thr−Phe−Thr−Ser−Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−Trp−Leu−Val−Lys−Gly−Arg−Gly−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Ala−Glu−Ser−Lys−Tyr−Gly−Pro−Pro−Cys−Pro (SEQ ID NO:10)
CACGGCGAGGGCACCTTCACCTCCGACGTGTCCTCCTATCTCGAGGAGCAGGCCGCCAAGGAATTCATCGCCTGGCTGGTGAAGGGCGGCGGCGGTGGTGGTGGCTCCGGAGGCGGCGGCTCTGGTGGCGGTGGCAGCGCTGAGTCCAAATATGGTCCCCCATGCCCACCCTGCCCAGCACCTGAGGCCGCCGGGGGACCATCAGTCTTCCTGTTCCCCCCAAAACCCAAGGACACTCTCATGATCTCCCGGACCCCTGAGGTCACGTGCGTGGTGGTGGACGTGAGCCAGGAAGACCCCGAGGTCCAGTTCAACTGGTACGTGGATGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTTCAACAGCACGTACCGTGTGGTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAACGGCAAGGAGTACAAGTGCAAGGTCTCCAACAAAGGCCTCCCGTCCTCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAGCCACAGGTGTACACCCTGCCCCCATCCCAGGAGGAGATGACCAAGAACCAGGTCAGCCTGACCTGCCTGGTCAAAGGCTTCTACCCCAGCGACATCGCCGTGGAGTGGGAAAGCAATGGGCAGCCGGAGAACAACTACAAGACCACGCCTCCCGTGCTGGACTCCGACGGCTCCTTCTTCCTCTACAGCAGGCTAACCGTGGACAAGAGCAGGTGGCAGGAGGGGAATGTCTTCTCATGCTCCGTGATGCATGAGGCTCTGCACAACCACTACACACAGAAGAGCCTCTCCCTGTCTCTGGGT (SEQ ID NO:20)
CRE−BLAMシステムを用いたヒトGLP−1受容体を発現するHEK−293細胞を、ポリ−d−リジンコーティングされた底面が透明な黒い96穴プレートに、1ウェルあたり10%FBSを含むDMEM培地100 μl中、20,000から40,000細胞の密度で播種した。播種翌日、培地を除き、血漿を含まないDMEM培地80 μlを添加した。播種後3日目に、用量反応曲線を得るために、異なる濃度の様々なGLP−1−Fc異種融合タンパク質を含む0.5%BSA含有血漿不含DMEM培地20 μlをそれぞれのウェルに加えた。通常、3nmolから30nmolまでの異種GLP1−Fc融合タンパク質を含む14個の希釈物を用いて、EC50値を決定する用量反応曲線を作成した。融合タンパク質とのインキュベーション5時間後、β−ラクタマーゼ基質20 μl(CCF2/AM,パンヴェラ社(PanVera LLC))を加え、1時間インキュベートを続け、Cytofluor(蛍光プレートリーダー)上で蛍光を測定した。このアッセイについては、ズロカーニック(Zlokarnik)等、(1998)サイエンス誌(Science)278:84−88で更に述べられている。種々のGLP−1−Fc融合タンパク質をテストし、EC50値を表1に示した。この値は、全実験で、内部標準として行なったVal8−GLP−1(7−37)OHの結果に対する相対値としてもとめた。
CRE−ルシフェラーゼ系を用いたヒトGLP−1受容体を安定に発現するHEK−293細胞を、96穴プレートに1ウェルあたり低血清DMEM F12培地80 μl中、30,000細胞の密度で播種した。播種翌日、0.5%BSAに溶解したテストタンパク質を20μlの一定分量で混合し、細胞と共に5時間インキュベートした。通常、3pmolから3nmolを含む12個の希釈物を各テストタンパク質用に5倍濃度で調整し、細胞に添加し、EC50値を決定する用量反応曲線を作成した。インキュベーション後、ルシフェラーゼ試薬100 μlをそれぞれのプレートに直接加え、2分間穏やかに混合した。プレートをTRILUX社のルミノメーターに置き、ルシフェラーゼ発現の結果により生じる発光を測定した。種々のGLP−1−Fc融合タンパク質をテストし、EC50値を表2に示した。この値は、全実験で、内部標準として行なったVal8−GLP−1(7−37)OHの結果に対する相対値としてもとめた。以下でテストした融合タンパク質は二量体であるため、値をモル濃度にして2倍の違いを考慮して補正した。
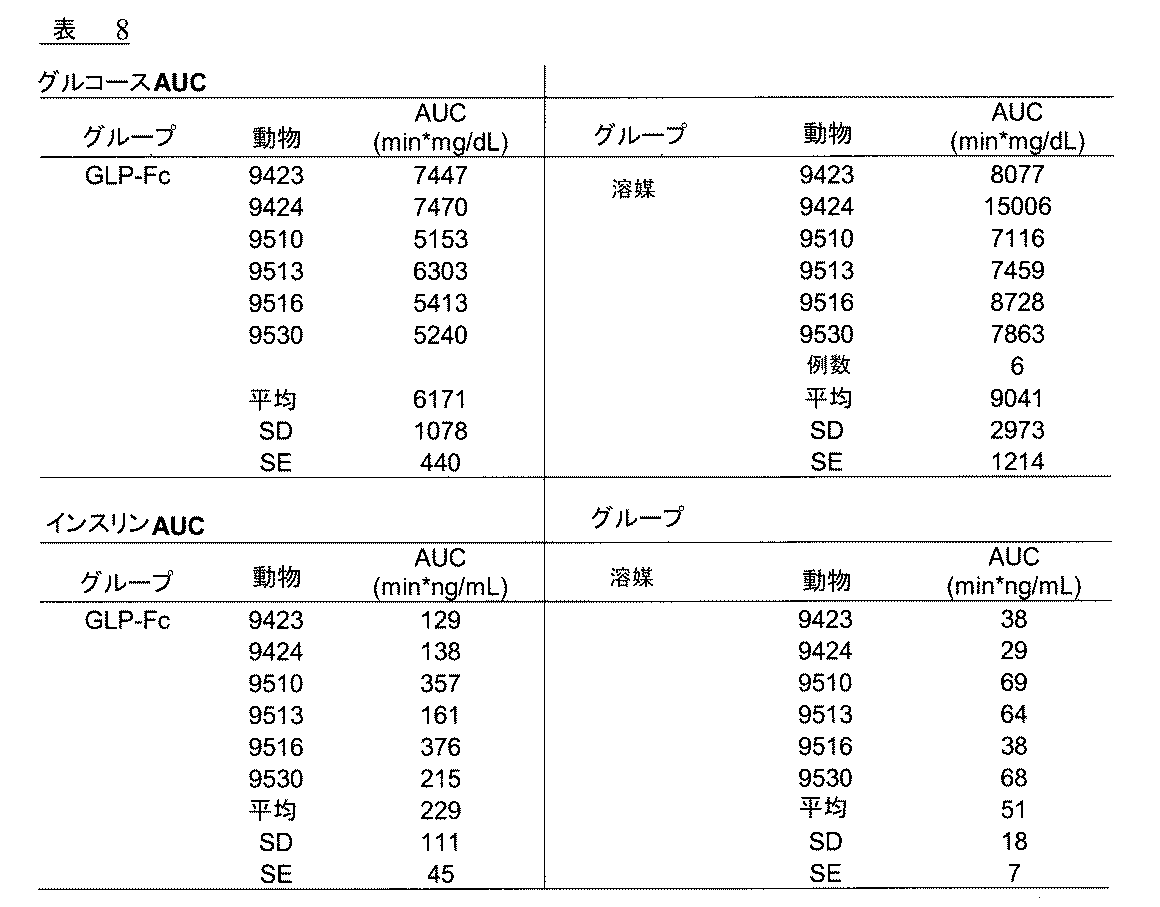
Fc融合タンパク質、Gly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4 (S228P、F234A、L235A)を、ラットにおける静脈内糖負荷試験(IVGTT)にて評価した。3グループそれぞれには、少なくともラット4匹が含まれている。グループIには溶媒を投与し(表3)、グループII にはGly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)、1.79 mg/kgを一回の皮下注射にて投与し(表4)、グループIIIにはGly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4 (S228P,F234A,L235A)、0.179 mg/kgを一回の皮下注射にて投与した(表5)。ラットを、第一日目の朝、皮下注射した。初回の注射から24時間後、ラットの体重1グラムあたり1 μLのグルコース(D50)をボーラス投与した。血液試料を、グルコースボーラス投与2、4、6、10、20および30分後に採取した。
雄のカニクイザルにFc融合タンパク質、Gly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)を0.1 mg/kg皮下注射(SC)した際の薬物動態(PK)を明らかにするために試験を行った。ラジオイムノアッセイ(RIA)抗体はGLPの中間部分に特異的である。酵素免疫測定法(ELISA)は、N末端特異的な捕捉抗体、およびFc特異的な検出抗体を用いる。酵素免疫測定法(ELISA)法およびラジオイムノアッセイ(RIA)法、双方から得られた血漿濃度の結果を、この薬物動態パラメーター値の決定に用いた。
a:観察された最大血漿濃度
b:最大血漿濃度が観察された時間
c:血漿濃度−時間曲線の0から無限大までの曲線下面積(AUC)
d:除去半減期
e:生物学的利用能に対する全身クリアランス
f:生物学的利用能に対する分布容積
SD:標準偏差
カニクイザル(cynomolgus monkey)の所定の血清試料を、Gly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)に対する抗体形成を直接吸着法による酵素免疫測定法(ELISA)法を用いてテストした。マイクロタイタープレートを0.1 μg/mLの濃度のGly8−Glu22−Gly36−GLP−1(7−37)−L−IgG4(S228P,F234A,L235A)でコーティングした。サル血清試料をブロッキング溶液で50倍、500倍、1000倍および5000倍に希釈して、ウェル当たり0.05 mLで約1時間インキュベートした。二次抗体、ヤギ抗ヒトFab'2ペルオキシダーゼ結合抗体(ヒトとの交差反応性は75%)を、ブロッキング溶液にて10,000倍に希釈され、ウェル当たり0.05mLで約1時間インキュベートした。テトラメチルベンジジン(tetramethylbenzidine,TMB)基質を用いた発色を、450nm−630nmの吸光度で測定した。2回の計測値を平均した。GLP−1抗体をポジティブコントロールに用い、検出に用いる二次抗体にはヤギ抗ウサギIgG(H+L)ペルオキシダーゼ結合抗体を用いた。抗体形成の可能性を評価するため、血清試料を、投薬前、2回目の投薬の24時間後、1回目、2回目の皮下注射投薬168時間後に採取した。G8E22−CEX−L−hIgG4に対する抗体力価の有無は、投薬前の血清とポジティブコントロールの比較によって解釈された。該当結果は、表7に示す。
段階1(研究第1日)で、溶媒の皮下注射を行った。次いで、5、10、および25 mg/kg/minの段階的な静脈グルコース(20%グルコース)点滴を溶媒注射後直ちに行った。段階2(研究第2日)では、GLP−1融合タンパク質(0.1 mg/kg)の皮下注射を行った。段階3では、GLP−1融合タンパク質の注射後、およそ96時間の段階的な静脈グルコース点滴を行った。
カニューレを常置したラットは、溶媒コントロール(生理食塩水)もしくは3つの処置グループの一つ(GLP−1融合タンパク質、0.0179 mg/kg、0.179 mg/kg、または 1.79 mg/kg)のいずれかに割り当てられた。GLP−1融合タンパク質および溶媒を、皮下注射にて投与した。処置24時間後、前夜絶食させた(16時間)ラットを、段階的静脈グルコース点滴試験の対象とした。段階的グルコース点滴を、ベースラインの生理食塩水期間(20分)、その後それぞれ5および15 mg/kg/minの2回の30分間のグルコース点滴段階で構成した。血漿試料を、グルコース点滴前(ベースライン)20分、10分、0分、およびグルコース点滴後10分、20分、30分、40分、50分、60分に採取した。
Claims (4)
- GLP−1アナログを含む異種融合タンパク質であって、前記GLP−1アナログは、
a) (SEQ ID NO:1)
His−Xaa8−Glu−Gly−Thr−Phe−Thr−Ser−
Asp−Val−Ser−Ser−Tyr−Leu−Glu−Glu−
Gln−Ala−Ala−Lys−Glu−Phe−Ile−Ala−
Trp−Leu−Val−Lys−Gly−Gly−Gly
(配列中、Xaa8はGlyである)、
を含み、前記GLP−1アナログは、
Ala−Glu−Ser−Lys−Tyr−Gly−Pro−Pro−
Cys−Pro−Pro−Cys−Pro−Ala−Pro−Xaa16−
Xaa17−Xaa18−Gly−Gly−Pro−Ser−Val−Phe−
Leu−Phe−Pro−Pro−Lys−Pro−Lys−Asp−
Thr−Leu−Met−Ile−Ser−Arg−Thr−Pro−
Glu−Val−Thr−Cys−Val−Val−Val−Asp−
Val−Ser−Gln−Glu−Asp−Pro−Glu−Val−
Gln−Phe−Asn−Trp−Tyr−Val−Asp−Gly−
Val−Glu−Val−His−Asn−Ala−Lys−Thr−
Lys−Pro−Arg−Glu−Glu−Gln−Phe−Xaa80−
Ser−Thr−Tyr−Arg−Val−Val−Ser−Val−
Leu−Thr−Val−Leu−His−Gln−Asp−Trp−
Leu−Asn−Gly−Lys−Glu−Tyr−Lys−Cys−
Lys−Val−Ser−Asn−Lys−Gly−Leu−Pro−
Ser−Ser−Ile−Glu−Lys−Thr−Ile−Ser−
Lys−Ala−Lys−Gly−Gln−Pro−Arg−Glu−
Pro−Gln−Val−Tyr−Thr−Leu−Pro−Pro−
Ser−Gln−Glu−Glu−Met−Thr−Lys−Asn−
Gln−Val−Ser−Leu−Thr−Cys−Leu−Val−
Lys−Gly−Phe−Tyr−Pro−Ser−Asp−Ile−
Ala−Val−Glu−Trp−Glu−Ser−Asn−Gly−
Gln−Pro−Glu−Asn−Asn−Tyr−Lys−Thr−
Thr−Pro−Pro−Val−Leu−Asp−Ser−Asp−
Gly−Ser−Phe−Phe−Leu−Tyr−Ser−Arg−
Leu−Thr−Val−Asp−Lys−Ser−Arg−Trp−
Gln−Glu−Gly−Asn−Val−Phe−Ser−Cys−
Ser−Val−Met−His−Glu−Ala−Leu−His−
Asn−His−Tyr−Thr−Gln−Lys−Ser−Leu−
Ser−Leu−Ser−Leu−Gly−Xaa230
(SEQ ID NO:7)
(配列中、16番目のXaaは、Gluであり、
17番目のXaaは、Alaであり、
18番目のXaaは、Alaであり、
80番目のXaaは、Asnであり、
230番目のXaaは、欠損である)、
上記のSEQ ID NO:7の配列を含む免疫グロブリンのFc部分と融合されており、該異種融合タンパク質はさらに以下の配列のペプチドリンカーを含み、
Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser−Gly−Gly−Gly−Gly−Ser (SEQ ID NO:8)、
ここで、ペプチドリンカーのN−末端のグリシンが、前記GLP−1アナログのC−末端のグリシン残基と直接融合しており、ペプチドリンカーのC−末端のセリンが前記Fc部分のN−末端のアラニンと直接融合している、異種融合タンパク質。 - 請求項1に記載の異種融合タンパク質を含有する、医薬組成物。
- インスリン非依存性糖尿病を処置するための、請求項1に記載の異種融合タンパク質を含有する、医薬組成物。
- 体重過多の患者において肥満を処置するためのまたは体重減少を誘導するための、請求項1に記載の異種融合タンパク質を含有する、医薬組成物。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US47788003P | 2003-06-12 | 2003-06-12 | |
| US60/477,880 | 2003-06-12 | ||
| PCT/US2004/015595 WO2005000892A2 (en) | 2003-06-12 | 2004-06-10 | Glp-1 analog fusion plroteins |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2007536902A JP2007536902A (ja) | 2007-12-20 |
| JP4629047B2 true JP4629047B2 (ja) | 2011-02-09 |
Family
ID=33551775
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2006533197A Active JP4629047B2 (ja) | 2003-06-12 | 2004-06-10 | Glp−1アナログ複合タンパク質 |
Country Status (29)
| Country | Link |
|---|---|
| US (2) | US7452966B2 (ja) |
| EP (2) | EP1641823B1 (ja) |
| JP (1) | JP4629047B2 (ja) |
| KR (1) | KR100758755B1 (ja) |
| CN (2) | CN101974090B (ja) |
| AR (1) | AR044776A1 (ja) |
| AT (1) | ATE525395T1 (ja) |
| AU (1) | AU2004251145C1 (ja) |
| BE (1) | BE2015C007I2 (ja) |
| BR (1) | BRPI0411132B8 (ja) |
| CA (1) | CA2528591C (ja) |
| CY (2) | CY1111991T1 (ja) |
| DK (1) | DK1641823T3 (ja) |
| EA (1) | EA008831B1 (ja) |
| ES (1) | ES2371072T3 (ja) |
| FR (1) | FR15C0010I2 (ja) |
| HK (1) | HK1149566A1 (ja) |
| HR (1) | HRP20110714T1 (ja) |
| HU (1) | HUS1500024I1 (ja) |
| IL (1) | IL171926A (ja) |
| LT (1) | LTC1641823I2 (ja) |
| MX (1) | MXPA05013565A (ja) |
| NZ (1) | NZ543292A (ja) |
| PL (1) | PL1641823T3 (ja) |
| PT (1) | PT1641823E (ja) |
| SI (1) | SI1641823T1 (ja) |
| TW (1) | TW200507870A (ja) |
| UA (1) | UA87458C2 (ja) |
| WO (1) | WO2005000892A2 (ja) |
Families Citing this family (165)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7183387B1 (en) | 1999-01-15 | 2007-02-27 | Genentech, Inc. | Polypeptide variants with altered effector function |
| US7459540B1 (en) | 1999-09-07 | 2008-12-02 | Amgen Inc. | Fibroblast growth factor-like polypeptides |
| ES2326964T3 (es) * | 2001-10-25 | 2009-10-22 | Genentech, Inc. | Composiciones de glicoproteina. |
| EP1463752A4 (en) | 2001-12-21 | 2005-07-13 | Human Genome Sciences Inc | ALBUMIN FUSION PROTEINS |
| EP2277889B1 (en) | 2001-12-21 | 2014-07-09 | Human Genome Sciences, Inc. | Fusion proteins of albumin and interferon beta |
| US20070161087A1 (en) * | 2003-05-29 | 2007-07-12 | Wolfgang Glaesner | Glp-1 fusion proteins |
| BRPI0411132B8 (pt) * | 2003-06-12 | 2021-05-25 | Lilly Co Eli | protéina de fusão heteróloga e seus usos |
| BRPI0507026A (pt) | 2004-02-09 | 2007-04-17 | Human Genome Sciences Inc | proteìnas de fusão de albumina |
| US20090232807A1 (en) * | 2004-12-22 | 2009-09-17 | Eli Lilly And Company | Glp-1 analog fusion protein formulations |
| PT1881850E (pt) * | 2005-05-13 | 2010-11-26 | Lilly Co Eli | Compostos peguilados de glp-1 |
| CN101273134B (zh) * | 2005-07-27 | 2012-01-04 | 王庆华 | 用于预防和治疗ⅰ型和ⅱ型糖尿病的组合物和方法 |
| CA2658678A1 (en) | 2005-08-06 | 2007-02-15 | Gerald J. Prud'homme | Composition and method for prevention and treatment of type i diabetes |
| ES2397289T3 (es) * | 2005-09-22 | 2013-03-06 | Biocompatibles Uk Ltd. | Polipéptidos de fusión de GLP-1 (péptido 1 de tipo glucagón) con resistencia a peptidasa incrementada |
| WO2007067828A2 (en) * | 2005-10-24 | 2007-06-14 | Centocor, Inc. | Glp-2 mimetibodies, polypeptides, compositions, methods and uses |
| ES2586236T3 (es) | 2005-11-04 | 2016-10-13 | Glaxosmithkline Llc | Procedimientos para administrar agentes hipoglucémicos |
| WO2007056362A2 (en) | 2005-11-07 | 2007-05-18 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting physiological solubility and stability |
| US20130172274A1 (en) | 2005-12-20 | 2013-07-04 | Duke University | Methods and compositions for delivering active agents with enhanced pharmacological properties |
| US8841255B2 (en) | 2005-12-20 | 2014-09-23 | Duke University | Therapeutic agents comprising fusions of vasoactive intestinal peptide and elastic peptides |
| EP1816201A1 (en) | 2006-02-06 | 2007-08-08 | CSL Behring GmbH | Modified coagulation factor VIIa with extended half-life |
| EP2573111A1 (en) | 2006-04-20 | 2013-03-27 | Amgen Inc. | GLP-1 compounds |
| JP5290177B2 (ja) * | 2006-08-31 | 2013-09-18 | セントカー・インコーポレーテツド | Glp−2ミメティボディ、ポリペプチド、組成物、方法および用途 |
| CA2663047A1 (en) * | 2006-09-06 | 2008-03-13 | Phase Bioscience, Inc. | Therapeutic elastin-like polypeptide (elp) fusion proteins |
| US8338376B2 (en) * | 2006-10-20 | 2012-12-25 | Biogen Idec Ma Inc. | Compositions comprising variant LT-B-R-IG fusion proteins |
| EP3231440A1 (en) | 2006-12-22 | 2017-10-18 | CSL Behring GmbH | Modified coagulation factors with prolonged in vivo half-life |
| AU2011254001B2 (en) * | 2007-01-05 | 2012-08-02 | Covx Technologies Ireland Limited | Glucagon-like protein-1 receptor (GLP-1R) agonist compounds |
| MX2009006564A (es) * | 2007-01-05 | 2009-06-26 | Univ Indiana Res & Tech Corp | Analogos de glucagon que muestran solubilidad potenciada en amortiguadores a ph fisiologico. |
| US20090098130A1 (en) * | 2007-01-05 | 2009-04-16 | Bradshaw Curt W | Glucagon-like protein-1 receptor (glp-1r) agonist compounds |
| JP2008169195A (ja) * | 2007-01-05 | 2008-07-24 | Hanmi Pharmaceutical Co Ltd | キャリア物質を用いたインスリン分泌ペプチド薬物結合体 |
| ES2753183T3 (es) | 2007-02-12 | 2020-04-07 | Csl Behring Gmbh | Aplicación terapéutica de inhibidores de la proteasa de serina de tipo Kazal |
| KR20090119876A (ko) | 2007-02-15 | 2009-11-20 | 인디애나 유니버시티 리서치 앤드 테크놀로지 코퍼레이션 | 글루카곤/glp-1 수용체 공동-항진물질 |
| EP2139509A2 (en) * | 2007-03-15 | 2010-01-06 | Biogen Idec MA, Inc. | Treatment of autoimmune disorders |
| DK2172479T3 (en) * | 2007-06-19 | 2016-12-19 | Glytech Inc | GLP-1 peptide having the sugar chain attached thereto |
| US7960336B2 (en) | 2007-08-03 | 2011-06-14 | Pharmain Corporation | Composition for long-acting peptide analogs |
| EP2185178B1 (en) | 2007-08-03 | 2017-08-23 | Eli Lilly And Company | Use of an fgf-21 compound and a glp-1 compound for the treatment of obesity |
| US8563527B2 (en) * | 2007-08-20 | 2013-10-22 | Pharmain Corporation | Oligonucleotide core carrier compositions for delivery of nucleic acid-containing therapeutic agents, methods of making and using the same |
| EP2031064A1 (de) * | 2007-08-29 | 2009-03-04 | Boehringer Ingelheim Pharma GmbH & Co. KG | Verfahren zur Steigerung von Proteintitern |
| US8981047B2 (en) | 2007-10-30 | 2015-03-17 | Indiana University Research And Technology Corporation | Glucagon antagonists |
| ES2558155T3 (es) * | 2007-10-30 | 2016-02-02 | Indiana University Research And Technology Corporation | Compuestos que muestran actividad antagonista de glucacón y agonista de GLP-1 |
| US20100317057A1 (en) * | 2007-12-28 | 2010-12-16 | Novo Nordisk A/S | Semi-recombinant preparation of glp-1 analogues |
| CN101983066B (zh) * | 2008-01-30 | 2016-06-29 | 印第安那大学科技研究公司 | 基于酯的胰岛素前药 |
| JOP20190083A1 (ar) | 2008-06-04 | 2017-06-16 | Amgen Inc | بولي ببتيدات اندماجية طافرة لـfgf21 واستخداماتها |
| PA8830501A1 (es) | 2008-06-17 | 2010-07-27 | Univ Indiana Res & Tech Corp | Co-agonistas del receptor de glucagon/glp-1 |
| SG192405A1 (en) * | 2008-06-17 | 2013-08-30 | Univ Indiana Res & Tech Corp | Gip-based mixed agonists for treatment of metabolic disorders and obesity |
| WO2009155257A1 (en) * | 2008-06-17 | 2009-12-23 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility and stability physiological ph buffers |
| ES2531464T3 (es) | 2008-06-24 | 2015-03-16 | Csl Behring Gmbh | Factor VIII, factor de von Willebrand o sus complejos con semivida in vivo prolongada |
| CA2953975C (en) | 2008-06-27 | 2019-11-26 | Duke University | Therapeutic agents comprising elastin-like peptides |
| EA032727B1 (ru) | 2008-10-10 | 2019-07-31 | Амген Инк. | Мутантный резистентный к протеолизу полипептид fgf21 и его применение |
| JP2012512903A (ja) | 2008-12-19 | 2012-06-07 | インディアナ ユニバーシティー リサーチ アンド テクノロジー コーポレーション | アミド系グルカゴンスーパーファミリーペプチドプロドラッグ |
| US9238878B2 (en) | 2009-02-17 | 2016-01-19 | Redwood Bioscience, Inc. | Aldehyde-tagged protein-based drug carriers and methods of use |
| US20120052069A1 (en) | 2009-05-05 | 2012-03-01 | Amgen Inc | Fgf21 mutants and uses thereof |
| HRP20240135T1 (hr) | 2009-05-05 | 2024-04-12 | Amgen Inc. | Fgf21 mutanti i njihove upotrebe |
| SG176858A1 (en) | 2009-06-16 | 2012-02-28 | Univ Indiana Res & Tech Corp | Gip receptor-active glucagon compounds |
| WO2010148142A1 (en) * | 2009-06-17 | 2010-12-23 | Amgen Inc. | Chimeric fgf19 polypeptides and uses thereof |
| CN101993485B (zh) | 2009-08-20 | 2013-04-17 | 重庆富进生物医药有限公司 | 促胰岛素分泌肽类似物同源二聚体及其用途 |
| WO2011043530A1 (ko) * | 2009-10-09 | 2011-04-14 | (주)알테오젠 | Glp-1 유사체의 융합체, 및 이를 유효성분으로 함유하는 당뇨병의 예방 또는 치료용 조성물 |
| JP2013512672A (ja) * | 2009-12-02 | 2013-04-18 | アムジエン・インコーポレーテツド | ヒトFGFR1c、ヒトβ−クロト−、ならびにヒトFGFR1cおよびヒトβ−クロト−の両方に結合する結合タンパク質 |
| UA109888C2 (uk) | 2009-12-07 | 2015-10-26 | ІЗОЛЬОВАНЕ АНТИТІЛО АБО ЙОГО ФРАГМЕНТ, ЩО ЗВ'ЯЗУЄТЬСЯ З β-КЛОТО, РЕЦЕПТОРАМИ FGF І ЇХНІМИ КОМПЛЕКСАМИ | |
| US8703701B2 (en) | 2009-12-18 | 2014-04-22 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| EP2528618A4 (en) | 2010-01-27 | 2015-05-27 | Univ Indiana Res & Tech Corp | GLUCAGON ANTAGONISTE AND GIP AGONISTS CONJUGATES AND COMPOSITIONS FOR THE TREATMENT OF METABOLISM DISEASES AND ADIPOSITAS |
| EP2371857A1 (en) | 2010-04-01 | 2011-10-05 | CSL Behring GmbH | Factor XII inhibitors for treating interstitial lung disease |
| AU2011239689A1 (en) | 2010-04-15 | 2012-11-08 | Amgen Inc. | Human FGF receptor and beta-Klotho binding proteins |
| US20130123460A1 (en) | 2010-04-30 | 2013-05-16 | Sanwa Kagaku Kenkyusho Co., Ltd. | Peptide for improving biostability of bioactive substance, and bioactive substance having improved biostability |
| EP2569000B1 (en) | 2010-05-13 | 2017-09-27 | Indiana University Research and Technology Corporation | Glucagon superfamily peptides exhibiting nuclear hormone receptor activity |
| CA2797089A1 (en) | 2010-05-13 | 2011-11-17 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting g protein-coupled receptor activity |
| CN101891823B (zh) | 2010-06-11 | 2012-10-03 | 北京东方百泰生物科技有限公司 | 一种Exendin-4及其类似物融合蛋白 |
| MX2012014576A (es) | 2010-06-24 | 2013-02-21 | Univ Indiana Res & Tech Corp | Profarmacos de peptido de la superfamilia de glucagon basado en amida. |
| CN103415300B (zh) * | 2010-07-20 | 2018-02-23 | 诺沃—诺迪斯克有限公司 | N‑末端修饰的fgf21化合物 |
| MA34885B1 (fr) | 2010-12-22 | 2014-02-01 | Indiana Unversity Res And Technology Corp | Analogues du glucagon presentant una ctivite de recepteur de gip |
| RU2606016C2 (ru) | 2011-01-14 | 2017-01-10 | Редвуд Байосайнс, Инк. | Меченые альдегидом полипептиды иммуноглобулина и способы их применения |
| EP2497489A1 (en) | 2011-03-09 | 2012-09-12 | CSL Behring GmbH | Factor XII inhibitors for the treatment of silent brain ischemia and ischemia of other organs |
| CA2829037C (en) | 2011-03-09 | 2022-05-17 | Csl Behring Gmbh | Factor xii inhibitors for the administration with medical procedures comprising contact with artificial surfaces |
| PT2691417T (pt) | 2011-03-29 | 2018-10-31 | Roche Glycart Ag | Variantes de fc de anticorpos |
| US9156902B2 (en) | 2011-06-22 | 2015-10-13 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| BR112013032717A2 (pt) | 2011-06-22 | 2017-01-24 | Univ Indiana Res & Tech Corp | coagonistas do receptor de glucagon/glp-1 |
| JP6253578B2 (ja) | 2011-07-22 | 2017-12-27 | ツェー・エス・エル・ベーリング・ゲー・エム・ベー・ハー | 阻害性抗第xii/xiia因子モノクローナル抗体及びそれらの使用 |
| UY34317A (es) | 2011-09-12 | 2013-02-28 | Genzyme Corp | Anticuerpo antireceptor de célula T (alfa)/ß |
| KR20130049671A (ko) | 2011-11-04 | 2013-05-14 | 한미사이언스 주식회사 | 생리활성 폴리펩타이드 결합체 제조 방법 |
| CA2847246A1 (en) | 2011-11-17 | 2013-05-23 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting glucocorticoid receptor activity |
| EP2623110A1 (en) | 2012-01-31 | 2013-08-07 | CSL Behring GmbH | Factor XII inhibitors for the treatment of neurological inflammatory disorders |
| PL2814502T3 (pl) | 2012-02-15 | 2018-02-28 | Csl Behring Gmbh | Warianty czynnika von Willebranda mające ulepszone powinowactwo do wiązania czynnika VIII |
| CA2877358A1 (en) | 2012-06-21 | 2013-12-27 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting gip receptor activity |
| HUE056217T2 (hu) | 2012-07-13 | 2022-02-28 | Roche Glycart Ag | Bispecifikus anti-VEGF/anti-ANG-2 antitestek és ezek alkalmazása szemészeti érbetegségek kezelésében |
| US9790268B2 (en) | 2012-09-12 | 2017-10-17 | Genzyme Corporation | Fc containing polypeptides with altered glycosylation and reduced effector function |
| PT3366705T (pt) * | 2012-09-12 | 2023-07-19 | Genzyme Corp | Polipéptidos contendo fc com glicosilação alterada e função efetora reduzida |
| US9474803B2 (en) | 2012-11-27 | 2016-10-25 | Alteogen Inc. | Composition for stabilizing fusion protein in which protein and FC domain are fused |
| HUE039334T2 (hu) | 2012-11-27 | 2018-12-28 | Biomarin Pharm Inc | Célzott terápiás lizoszómális enzim fúziós fehérjék és azok alkalmazásai |
| CA2901225C (en) | 2013-03-08 | 2023-09-19 | Csl Behring Gmbh | Treatment and prevention of remote ischemia-reperfusion injury |
| IL275376B2 (en) | 2013-03-11 | 2024-01-01 | Genzyme Corp | Polypeptides with hyperglycosidic bonds |
| US9580486B2 (en) * | 2013-03-14 | 2017-02-28 | Amgen Inc. | Interleukin-2 muteins for the expansion of T-regulatory cells |
| WO2014158900A1 (en) | 2013-03-14 | 2014-10-02 | Indiana University Research And Technology Corporation | Insulin-incretin conjugates |
| EP2796145B1 (en) | 2013-04-22 | 2017-11-01 | CSL Ltd. | A covalent complex of von willebrand factor and faktor viii linked by a disulphide bridge |
| US20160166660A1 (en) | 2013-06-28 | 2016-06-16 | Csl Behring Gmbh | Combination therapy using a factor xii inhibitor and a c-1 inhibitor |
| CN103408669B (zh) | 2013-08-01 | 2016-01-20 | 江苏泰康生物医药有限公司 | Glp-1类似物融合蛋白,及其制备方法和用途 |
| CN104592381A (zh) * | 2013-10-31 | 2015-05-06 | 江苏万邦生化医药股份有限公司 | 一种利拉鲁肽中间体多肽的制备方法 |
| US20170065678A1 (en) | 2014-03-11 | 2017-03-09 | Novartis Ag | Methods of treating metabolic disorders associated with lipodystrophies and defects in insulin production or signaling |
| SG10201808158UA (en) | 2014-03-19 | 2018-10-30 | Genzyme Corp | Site-specific glycoengineering of targeting moieties |
| EP3889185A3 (en) * | 2014-03-31 | 2022-01-26 | Hanmi Pharm. Co., Ltd. | Composition for improving the solubility of a protein or peptide by using immunoglobulin fc fragment linkage |
| AU2015276089B2 (en) | 2014-06-18 | 2021-03-18 | Csl Behring Gmbh | Therapy using a Factor XII inhibitor in a neurotraumatic disorder |
| CN106659771B (zh) | 2014-07-02 | 2021-09-24 | 康诺贝林伦瑙有限公司 | 修饰的von Willebrand因子 |
| MX2017000821A (es) | 2014-07-21 | 2017-08-18 | Delinia Inc | Moleculas que activan selectivamente celulas t reguladoras para el tratamiento de enfermedades autoinmunes. |
| HUE043038T2 (hu) | 2014-08-11 | 2019-07-29 | Delinia Inc | Regulator T-sejteket szelektíven aktiváló, módosított IL-2-változatok autoimmun betegségek kezelésére |
| CN108271356A (zh) | 2014-09-24 | 2018-07-10 | 印第安纳大学研究及科技有限公司 | 肠降血糖素-胰岛素缀合物 |
| CN104293834B (zh) * | 2014-10-11 | 2018-03-23 | 上海兴迪金生物技术有限公司 | GLP‑1或其类似物与抗体Fc片段融合蛋白的制备方法 |
| RS59154B1 (sr) | 2014-12-23 | 2019-10-31 | Novo Nordisk As | Fgf21 derivati i njihova upotreba |
| KR101825048B1 (ko) * | 2014-12-31 | 2018-02-05 | 주식회사 제넥신 | GLP 및 면역글로불린 하이브리드 Fc 융합 폴리펩타이드 및 이의 용도 |
| MX2017012966A (es) | 2015-04-10 | 2018-06-06 | Amgen Inc | Muteinas de interleuquina 2 para la expansion de celulas t regulatorias. |
| AR105616A1 (es) * | 2015-05-07 | 2017-10-25 | Lilly Co Eli | Proteínas de fusión |
| EP4089109A3 (en) | 2015-05-22 | 2023-03-01 | CSL Behring Lengnau AG | Methods for preparing modified von willebrand factor |
| ES2774011T3 (es) | 2015-05-22 | 2020-07-16 | CSL Behring Lengnau AG | Polipéptidos del factor de von Willebrand truncado para tratar la hemofilia |
| TWI622596B (zh) | 2015-10-26 | 2018-05-01 | 美國禮來大藥廠 | 升糖素受體促效劑 |
| KR20170049319A (ko) | 2015-10-28 | 2017-05-10 | 주식회사유한양행 | 지속형 fgf21 융합 단백질 및 이를 포함하는 약학적 조성물 |
| KR20170049320A (ko) | 2015-10-28 | 2017-05-10 | 주식회사유한양행 | 이중 작용 단백질 및 이를 포함하는 약학적 조성물 |
| EP3184149A1 (en) | 2015-12-23 | 2017-06-28 | Julius-Maximilians-Universität Würzburg | Soluble glycoprotein v for treating thrombotic diseases |
| PE20181327A1 (es) | 2015-12-23 | 2018-08-20 | Amgen Inc | Metodo para tratar o mejorar trastornos metabolicos con proteinas de union para el receptor peptidico inhibidor gastrico (gipr) en combinacion con agonistas de glp-1 |
| SG11201805494WA (en) | 2016-01-07 | 2018-07-30 | Csl Behring Recombinant Facility Ag | Mutated von willebrand factor |
| BR112018013861A2 (pt) | 2016-01-07 | 2018-12-18 | Csl Behring Recombinant Facility Ag | polipeptídeo, complexo, composição farmacêutica, métodos para tratar um distúrbio de coagulação sanguínea, para produzir um polipeptídeo e um dímero de um polipeptídeo, para aumentar a meia vida do fator viii e para aumentar a dimerização do dito polipeptídeo, uso de um polipeptídeo, kit farmacêutico, plasmídeo ou vetor, e, célula hospedeira |
| US20170204154A1 (en) | 2016-01-20 | 2017-07-20 | Delinia, Inc. | Molecules that selectively activate regulatory t cells for the treatment of autoimmune diseases |
| JP7456723B2 (ja) | 2016-04-06 | 2024-03-27 | シーエスエル、リミテッド | アテローム性動脈硬化症の治療方法 |
| CA3019398A1 (en) | 2016-04-26 | 2017-11-02 | R.P. Scherer Technologies, Llc | Antibody conjugates and methods of making and using the same |
| US10336812B2 (en) * | 2016-05-10 | 2019-07-02 | Janssen Biotech, Inc. | GDF15 fusion proteins and uses thereof |
| EP3502143A4 (en) | 2016-08-19 | 2020-07-15 | Ampsource Biopharma Shanghai Inc. | BINDING PEPTIDE FOR THE CONSTRUCTION OF A FUSION PROTEIN |
| CN106279437B (zh) | 2016-08-19 | 2017-10-31 | 安源医药科技(上海)有限公司 | 高糖基化人凝血因子viii融合蛋白及其制备方法与用途 |
| CN107759694B (zh) | 2016-08-19 | 2023-01-13 | 安源医药科技(上海)有限公司 | 双特异性抗体及其制备方法与用途 |
| EA201991142A1 (ru) | 2016-11-08 | 2019-10-31 | Варианты il-2 для лечения аутоиммунных заболеваний | |
| JP7328891B2 (ja) | 2016-11-10 | 2023-08-17 | ユーハン・コーポレイション | 融合タンパク質を含む、肝炎、肝線維症、および肝硬変を予防または処置するための医薬組成物 |
| DK3538134T3 (da) | 2016-11-11 | 2022-02-07 | CSL Behring Lengnau AG | Trunkeret von willebrand-faktor polypeptider til ekstravaskulær administration til behandling eller profylakse af en blodkoagulationsforstyrrelse |
| AU2017358861B2 (en) | 2016-11-11 | 2022-02-17 | CSL Behring Lengnau AG | Truncated von Willebrand Factor polypeptides for treating hemophilia |
| EP3351262A1 (en) | 2016-12-30 | 2018-07-25 | Istanbul Universitesi Rektorlugu | Curaglutide for in treatment of prediabetes, diabetes, obesity and metabolic diseases associated thereto |
| JOP20190177A1 (ar) | 2017-01-17 | 2019-07-16 | Amgen Inc | طريقة لعلاج أو تحسين اضطرابات أيضية باستخدام مساعدات مستقبل glp-1 مقترنة بمناهضات لمستقبل ببتيد مثبط معوي (gipr) |
| EA201992516A1 (ru) | 2017-04-21 | 2020-04-09 | Юхан Корпорейшн | Способ получения бифункциональных белков и их производных |
| SI3630164T1 (sl) | 2017-06-01 | 2023-10-30 | Eli Lilly And Company | Dulaglutid za zdravljenje kronične ledvične bolezni |
| ES2966835T3 (es) | 2017-06-22 | 2024-04-24 | CSL Behring Lengnau AG | Modulación de la inmunogenicidad del FVIII por VWF truncado |
| WO2019060653A1 (en) | 2017-09-22 | 2019-03-28 | Regeneron Pharmaceuticals, Inc. | GLP-1 RECEPTOR AGONISTS (GLUCAGON-LIKE PEPTIDE 1) AND USES THEREOF |
| JOP20200126A1 (ar) | 2017-11-21 | 2020-05-21 | Lilly Co Eli | طرق استخدام وتركيبات تحتوي على دولاجلوتيد |
| US11752173B2 (en) | 2017-12-19 | 2023-09-12 | Beijing Jiyuan Biological Technology Co., Ltd. | FGF21 and GLP1 double gene-modified mesenchymal stem cell and use in treating a metabolic disease |
| WO2019125003A1 (ko) * | 2017-12-22 | 2019-06-27 | 케이비바이오메드 주식회사 | 경구용 유전자 전달체 및 이의 용도 |
| WO2019140021A1 (en) | 2018-01-12 | 2019-07-18 | Eli Lilly And Company | Combination therapy |
| US11679143B2 (en) | 2018-02-08 | 2023-06-20 | Sunshine Lake Pharma Co., Ltd. | FGF21 variant, fusion protein and application thereof |
| KR20200135618A (ko) | 2019-05-23 | 2020-12-03 | ㈜ 디앤디파마텍 | 폴리펩티드를 포함하는 비알코올성 지방간 질환의 예방 또는 치료용 약학 조성물 |
| BR112021000933A2 (pt) | 2018-07-19 | 2021-04-27 | D&D Pharmatech Inc. | polipeptídeo, composição farmacêutica, método de preparação dela e método de prevenção ou tratamento de doenças |
| CN110878127B (zh) | 2018-09-06 | 2022-06-28 | 浙江柏拉阿图医药科技有限公司 | 长效重组GLP1-Fc-CD47蛋白及其制备和用途 |
| CN111234000B (zh) * | 2018-11-28 | 2023-05-26 | 鲁南制药集团股份有限公司 | 艾塞纳肽类似物 |
| CN111269312B (zh) * | 2018-12-04 | 2023-05-09 | 鲁南制药集团股份有限公司 | 一种异源融合蛋白质 |
| CN109836486B (zh) * | 2019-01-30 | 2020-09-08 | 北京双因生物科技有限公司 | 成纤维生长因子21变体、其融合蛋白及其用途 |
| CN111662373A (zh) * | 2019-03-05 | 2020-09-15 | 广东东阳光药业有限公司 | 一种多肽分子及其应用 |
| EP3934679A1 (en) | 2019-03-08 | 2022-01-12 | Amgen Inc. | Growth differentiation factor 15 combination therapy |
| CA3128522C (en) | 2019-03-15 | 2024-04-02 | Eli Lilly And Company | Preserved formulations |
| CA3177693A1 (en) | 2019-04-05 | 2020-10-05 | Eli Lilly And Company | Therapeutic uses of dulaglutide |
| US20220211808A1 (en) | 2019-05-17 | 2022-07-07 | Universitaet Zuerich | Haptoglobin for use in treating an adverse secondary neurological outcome following a haemorrhagic stroke |
| KR20220029733A (ko) | 2019-07-04 | 2022-03-08 | 체에스엘 베링 렝나우 아게 | 응고 인자 viii의 시험관내 안정성을 증가시키기 위한 절단된 폰 빌레브란트 인자 (vwf) |
| EP4045641A1 (en) | 2019-10-15 | 2022-08-24 | Eli Lilly and Company | Recombinantly engineered, lipase/esterase-deficient mammalian cell lines |
| WO2021094344A1 (en) | 2019-11-11 | 2021-05-20 | CSL Behring Lengnau AG | Polypeptides for inducing tolerance to factor viii |
| CN115322794A (zh) | 2020-01-11 | 2022-11-11 | 北京质肽生物医药科技有限公司 | Glp-1和fgf21的融合蛋白的缀合物 |
| US20230372539A1 (en) | 2020-08-24 | 2023-11-23 | The Trustees Of The University Of Pennsylvania | Viral vectors encoding glp-1 receptor agonist fusions and uses thereof in treating metabolic diseases |
| CN114106194B (zh) * | 2020-08-31 | 2024-01-16 | 中国科学院天津工业生物技术研究所 | 一种用于治疗糖尿病和/或肥胖症的融合蛋白 |
| JP2023551193A (ja) | 2020-11-20 | 2023-12-07 | ツェー・エス・エル・ベーリング・ゲー・エム・ベー・ハー | 抗体関連型拒絶反応を処置するための方法 |
| CN114685644A (zh) | 2020-12-29 | 2022-07-01 | 苏州康宁杰瑞生物科技有限公司 | 一种人glp-1多肽变体及其应用 |
| KR20230142569A (ko) | 2021-02-01 | 2023-10-11 | 체에스엘 베링 아게 | 출혈성 뇌졸중 후의 이차 신경학적 이상 반응을 치료 또는 예방하는 방법 |
| WO2022234070A1 (en) | 2021-05-07 | 2022-11-10 | Csl Behring Ag | Expression system for producing a recombinant haptoglobin (hp) beta chain |
| CN117616037A (zh) | 2021-07-06 | 2024-02-27 | 苏州康宁杰瑞生物科技有限公司 | 融合蛋白及其应用 |
| WO2023225534A1 (en) | 2022-05-18 | 2023-11-23 | Protomer Technologies Inc. | Aromatic boron-containing compounds and related insulin analogs |
| CN114774496B (zh) * | 2022-06-21 | 2022-10-04 | 北京惠之衡生物科技有限公司 | 一种高密度发酵制备glp-1类似物的方法 |
| WO2024047219A1 (en) | 2022-09-02 | 2024-03-07 | Csl Behring Ag | Haptoglobin for use in treating or preventing exaggerated erectile response or erectile dysfunction |
| WO2024064842A1 (en) | 2022-09-21 | 2024-03-28 | Regeneron Pharmaceuticals, Inc. | Methods of treating obesity, diabetes, and liver dysfunction |
| WO2024068848A1 (en) | 2022-09-28 | 2024-04-04 | Zealand Pharma A/S | Methods for treating obesity |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4399216A (en) | 1980-02-25 | 1983-08-16 | The Trustees Of Columbia University | Processes for inserting DNA into eucaryotic cells and for producing proteinaceous materials |
| NZ201705A (en) | 1981-08-31 | 1986-03-14 | Genentech Inc | Recombinant dna method for production of hepatitis b surface antigen in yeast |
| US4943529A (en) | 1982-05-19 | 1990-07-24 | Gist-Brocades Nv | Kluyveromyces as a host strain |
| AU3145184A (en) | 1983-08-16 | 1985-02-21 | Zymogenetics Inc. | High expression of foreign genes in schizosaccharomyces pombe |
| US4879231A (en) | 1984-10-30 | 1989-11-07 | Phillips Petroleum Company | Transformation of yeasts of the genus pichia |
| GB8610600D0 (en) | 1986-04-30 | 1986-06-04 | Novo Industri As | Transformation of trichoderma |
| EP0307434B2 (en) * | 1987-03-18 | 1998-07-29 | Scotgen Biopharmaceuticals, Inc. | Altered antibodies |
| US5010182A (en) | 1987-07-28 | 1991-04-23 | Chiron Corporation | DNA constructs containing a Kluyveromyces alpha factor leader sequence for directing secretion of heterologous polypeptides |
| AU4005289A (en) | 1988-08-25 | 1990-03-01 | Smithkline Beecham Corporation | Recombinant saccharomyces |
| FR2646437B1 (fr) | 1989-04-28 | 1991-08-30 | Transgene Sa | Nouvelles sequences d'adn, leur application en tant que sequence codant pour un peptide signal pour la secretion de proteines matures par des levures recombinantes, cassettes d'expression, levures transformees et procede de preparation de proteines correspondant |
| DE68927344T2 (de) | 1989-04-28 | 1997-02-20 | Rhein Biotech Proz & Prod Gmbh | Hefezellen der Gattung-Schwanniomyces |
| EP0402226A1 (en) | 1989-06-06 | 1990-12-12 | Institut National De La Recherche Agronomique | Transformation vectors for yeast yarrowia |
| FR2649120B1 (fr) | 1989-06-30 | 1994-01-28 | Cayla | Nouvelle souche et ses mutants de champignons filamenteux, procede de production de proteines recombinantes a l'aide de ladite souche et souches et proteines obtenues selon ce procede |
| DE69334100T2 (de) * | 1992-11-13 | 2007-07-19 | Immunex Corp., Seattle | Elk ligand, ein cytokin |
| CA2196200A1 (en) * | 1994-07-29 | 1996-02-15 | Michael Joseph Browne | Novel compounds |
| GB9511935D0 (en) | 1995-06-13 | 1995-08-09 | Smithkline Beecham Plc | Novel compound |
| US5723125A (en) * | 1995-12-28 | 1998-03-03 | Tanox Biosystems, Inc. | Hybrid with interferon-alpha and an immunoglobulin Fc linked through a non-immunogenic peptide |
| US6750334B1 (en) | 1996-02-02 | 2004-06-15 | Repligen Corporation | CTLA4-immunoglobulin fusion proteins having modified effector functions and uses therefor |
| US6006753A (en) | 1996-08-30 | 1999-12-28 | Eli Lilly And Company | Use of GLP-1 or analogs to abolish catabolic changes after surgery |
| US6277819B1 (en) | 1996-08-30 | 2001-08-21 | Eli Lilly And Company | Use of GLP-1 or analogs in treatment of myocardial infarction |
| UA65549C2 (uk) * | 1996-11-05 | 2004-04-15 | Елі Ліллі Енд Компані | Спосіб регулювання ожиріння шляхом периферійного введення аналогів та похідних glp-1 (варіанти) та фармацевтична композиція |
| US6190909B1 (en) * | 1997-04-17 | 2001-02-20 | Millennium Pharmaceuticals, Inc. | TH2-specific gene |
| SE9802080D0 (sv) | 1998-06-11 | 1998-06-11 | Hellstroem | Pharmaceutical composition for the treatment of functional dyspepsia and/or irritable bowel syndrome and new use of substances therein |
| AU5027299A (en) | 1998-07-31 | 2000-02-28 | Novo Nordisk A/S | Use of glp-1 and analogues for preventing type ii diabetes |
| MY155270A (en) | 1998-09-24 | 2015-09-30 | Lilly Co Eli | Use of glp-1 or analogs in treatment of stroke |
| US6376653B1 (en) * | 1998-09-28 | 2002-04-23 | Smithkline Beecham Plc | Tie2 antagonist antibodies |
| ATE252601T1 (de) | 1999-05-17 | 2003-11-15 | Conjuchem Inc | Lang wirkende insulinotrope peptide |
| DE60124710T2 (de) | 2000-06-16 | 2007-09-13 | Eli Lilly And Co., Indianapolis | Analoge des glucagon ähnlichen peptid-1 |
| SI1724284T1 (sl) * | 2000-12-07 | 2009-12-31 | Lilly Co Eli | Glp-1 fuzijski proteini |
| US6992174B2 (en) * | 2001-03-30 | 2006-01-31 | Emd Lexigen Research Center Corp. | Reducing the immunogenicity of fusion proteins |
| US6900292B2 (en) * | 2001-08-17 | 2005-05-31 | Lee-Hwei K. Sun | Fc fusion proteins of human erythropoietin with increased biological activities |
| EP2277889B1 (en) * | 2001-12-21 | 2014-07-09 | Human Genome Sciences, Inc. | Fusion proteins of albumin and interferon beta |
| BRPI0411132B8 (pt) * | 2003-06-12 | 2021-05-25 | Lilly Co Eli | protéina de fusão heteróloga e seus usos |
-
2004
- 2004-06-10 BR BRPI0411132A patent/BRPI0411132B8/pt active IP Right Grant
- 2004-06-10 SI SI200431776T patent/SI1641823T1/sl unknown
- 2004-06-10 EP EP04752589A patent/EP1641823B1/en active Active
- 2004-06-10 EP EP11166548A patent/EP2368909A1/en not_active Withdrawn
- 2004-06-10 KR KR1020057023668A patent/KR100758755B1/ko active Protection Beyond IP Right Term
- 2004-06-10 EA EA200600015A patent/EA008831B1/ru active Protection Beyond IP Right Term
- 2004-06-10 CN CN201010508567.1A patent/CN101974090B/zh active Active
- 2004-06-10 AT AT04752589T patent/ATE525395T1/de active
- 2004-06-10 PT PT04752589T patent/PT1641823E/pt unknown
- 2004-06-10 DK DK04752589.4T patent/DK1641823T3/da active
- 2004-06-10 NZ NZ543292A patent/NZ543292A/en unknown
- 2004-06-10 JP JP2006533197A patent/JP4629047B2/ja active Active
- 2004-06-10 PL PL04752589T patent/PL1641823T3/pl unknown
- 2004-06-10 US US10/558,627 patent/US7452966B2/en active Active
- 2004-06-10 CN CN200480015953XA patent/CN1802386B/zh active Active
- 2004-06-10 MX MXPA05013565A patent/MXPA05013565A/es active IP Right Grant
- 2004-06-10 CA CA2528591A patent/CA2528591C/en active Active
- 2004-06-10 AU AU2004251145A patent/AU2004251145C1/en active Active
- 2004-06-10 UA UAA200511830A patent/UA87458C2/ru unknown
- 2004-06-10 WO PCT/US2004/015595 patent/WO2005000892A2/en active Application Filing
- 2004-06-10 ES ES04752589T patent/ES2371072T3/es active Active
- 2004-06-11 TW TW093116970A patent/TW200507870A/zh unknown
- 2004-06-11 AR ARP040102037A patent/AR044776A1/es unknown
-
2005
- 2005-11-14 IL IL171926A patent/IL171926A/en active IP Right Grant
-
2008
- 2008-10-31 US US12/262,832 patent/US8273854B2/en active Active
-
2011
- 2011-04-06 HK HK11103498.8A patent/HK1149566A1/xx unknown
- 2011-10-05 HR HR20110714T patent/HRP20110714T1/hr unknown
- 2011-11-03 CY CY20111101059T patent/CY1111991T1/el unknown
-
2015
- 2015-01-30 CY CY2015002C patent/CY2015002I2/el unknown
- 2015-01-30 FR FR15C0010C patent/FR15C0010I2/fr active Active
- 2015-02-02 BE BE2015C007C patent/BE2015C007I2/fr unknown
- 2015-02-03 LT LTPA2015007C patent/LTC1641823I2/lt unknown
- 2015-04-20 HU HUS1500024C patent/HUS1500024I1/hu unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4629047B2 (ja) | Glp−1アナログ複合タンパク質 | |
| EP1641483B1 (en) | Fusion proteins | |
| US7576190B2 (en) | FGF-21 fusion proteins | |
| KR100942864B1 (ko) | Glp-1 융합 단백질 | |
| US20070161087A1 (en) | Glp-1 fusion proteins | |
| WO2011153965A1 (zh) | Exendin-4及其类似物的融合蛋白,其制备和应用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20100427 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20100723 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20101012 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20101110 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131119 Year of fee payment: 3 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4629047 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R153 | Grant of patent term extension |
Free format text: JAPANESE INTERMEDIATE CODE: R153 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |