WO2008029688A1 - Polymère hyperbranché et son procédé de production - Google Patents
Polymère hyperbranché et son procédé de production Download PDFInfo
- Publication number
- WO2008029688A1 WO2008029688A1 PCT/JP2007/066767 JP2007066767W WO2008029688A1 WO 2008029688 A1 WO2008029688 A1 WO 2008029688A1 JP 2007066767 W JP2007066767 W JP 2007066767W WO 2008029688 A1 WO2008029688 A1 WO 2008029688A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- hyperbranched polymer
- formula
- carbon atoms
- represented
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/18—Introducing halogen atoms or halogen-containing groups
- C08F8/20—Halogenation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
- C08F2/38—Polymerisation using regulators, e.g. chain terminating agents, e.g. telomerisation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F12/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an aromatic carbocyclic ring
- C08F12/02—Monomers containing only one unsaturated aliphatic radical
- C08F12/04—Monomers containing only one unsaturated aliphatic radical containing one ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F2/00—Processes of polymerisation
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/18—Introducing halogen atoms or halogen-containing groups
- C08F8/20—Halogenation
- C08F8/22—Halogenation by reaction with free halogens
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/30—Introducing nitrogen atoms or nitrogen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/30—Introducing nitrogen atoms or nitrogen-containing groups
- C08F8/32—Introducing nitrogen atoms or nitrogen-containing groups by reaction with amines
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/44—Preparation of metal salts or ammonium salts
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G83/00—Macromolecular compounds not provided for in groups C08G2/00 - C08G81/00
- C08G83/002—Dendritic macromolecules
- C08G83/005—Hyperbranched macromolecules
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F8/00—Chemical modification by after-treatment
- C08F8/26—Removing halogen atoms or halogen-containing groups from the molecule
Definitions
- the present invention relates to a novel hyperbranched polymer and a method for producing the same.
- the hyperbranched polymer of the present invention is characterized by being optically stable. These include paints, inks, adhesives, resin fillers, various molding materials, nanometer-size porous forming agents, chemical mechanical abrasives, functional materials, nanocapsules, photonic crystals, resist materials , Optical materials, electronic materials, information recording materials, printing materials, battery materials, medical materials, magnetic materials, intermediate raw materials, etc.
- Dendritic dendritic
- Conventional dendritic polymers generally have a string-like shape, whereas these dendritic polymers actively introduce branching, so they have a unique structure and are on the order of nanometers.
- the number of terminal groups is the most prominent feature of dendritic polymers, and the number of branches increases as the molecular weight increases. Therefore, the absolute number of terminal groups increases with higher molecular weight dendritic polymers.
- Such dendritic polymers with a large number of terminal groups are greatly affected by the type of terminal group, and thus the glass transition temperature, solubility, thin film formability, etc. vary greatly, resulting in a general linear shape. It has characteristics not found in polymers.
- a reactive functional group is added to a terminal group, a dendritic polymer has a reactive functional group at a very high density. Application as a multifunctional cross-linking agent, metal or metal oxide dispersant or coating agent is expected.
- the ability to set the type of end groups is an important factor in developing the properties of the polymer.
- the advantages of the no-perperbranched polymer over the dendrimer include its ease of synthesis, which is particularly advantageous in industrial production.
- dendrimarker S, protected and deprotected are synthesized repeatedly, whereas hyperbranched polymers are synthesized by one-step polymerization of so-called ABx-type monomers with two or more substituents in one molecule. Is done.
- a hyperbranched polymer by living radical polymerization of a compound having photopolymerization initiating ability and having a bur group is known.
- a method of synthesizing a hyperbranched polymer by photopolymerization of a styrene compound having a dithiocanolevamate group see Non-Patent Documents 1, 2, and 3
- light of an acrylic compound having a dithiocanalbamate group is known.
- Patents l3 ⁇ 4 l Koji Ishizu, Akihide Mori, Macromol. Rapid Commun. 21, 665-668 (2000)
- Non-Patent Document 2 Koji Ishizu, Akihide Mori, Polymer International 50, 9 06-910 (2001)
- Non-Patent Document 3 Koji Ishizu, Yoshihiro Ohta, Susumu Kawauchi, Macromolecules Vol. 35, No. 9, 3781— 3784 (2002)
- Non-Patent Document 4 Koji Ishizu, Takeshi Shibuya, Akihide Mori, Polymer International 51, 424—428 (2002)
- Non-Patent Document 5 Koji Ishizu, Takeshi Shibuya, Susumu Kawauchi, Macromolecules Vol. 36, No. 10, 3505— 3510 (2002)
- Non-Patent Document 6 Koji Ishizu, Takeshi Shibuya, Jaebum Park, Satoshi U chida, Polymer International 53, 259—265 (2004)
- these hyperbranched polymers have dithiocanolevamate groups having photopolymerization initiating ability in their molecules, so they remain in a living state with respect to light and are optically unstable.
- no-perperbranched polymers can be compared with organic synthesis reactions. For example, in order to derivatize to hydroxylation, etherification, etc., it is necessary to convert the dithiocanolevamate group into a reactive functional group. Therefore, a novel hyperbranched polymer that is optically stable and can be derivatized into various compounds has been desired.
- An object of the present invention is to provide a novel hyperbranched polymer that is optically stable and can be derivatized into various polymers using a known reaction, and a method for producing the same. is there.
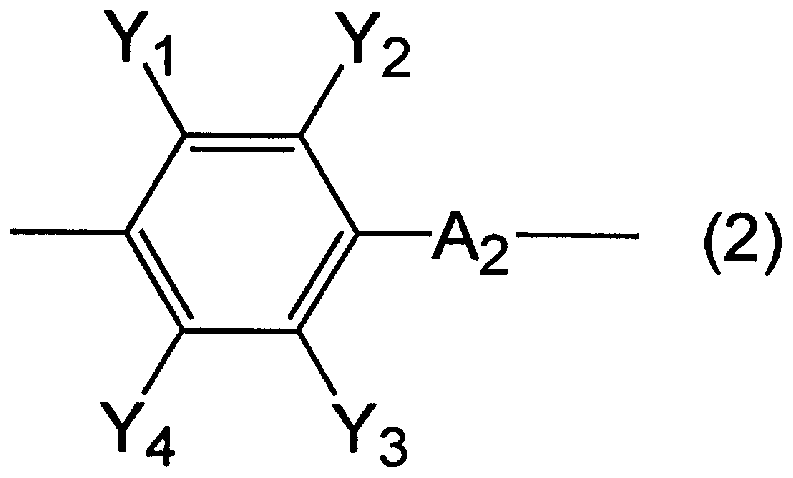
- A is 1 carbon atom which may contain an ether bond or an ester bond! / Represents 30 linear, branched or cyclic alkylene groups, Y, Y, Y or Y
- 1 2 3 4 represents a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a nitro group, a hydroxyl group, an amino group, a carboxyl group, or a cyan group, respectively.
- n is the number of repeating unit structures and represents an integer of 2 to 100,000.
- the hyperbranched polymer according to the first aspect wherein X represents a chlorine atom, a bromine atom or an iodine atom,
- the hyperbranched polymer according to the first aspect which is a structure represented by:
- the hyperbranched polymer according to the first aspect wherein the weight average molecular weight measured in terms of polystyrene by gel permeation chromatography is 500 to 5,000,000,
- equation (4)
- R and A are as defined in the formula (1), and R and R are respectively
- a force representing an alkyl group having 1 to 5 carbon atoms, a hydroxylalkyl group having 1 to 5 carbon atoms or an arylalkyl group having 7 to 12 carbon atoms, or R and R are
- a process for producing a hyperbranched polymer having a halogen atom at the molecular end comprising a step of substituting a halogen atom with a dithio force rubamate group at the molecular end of a hyperbranched polymer obtained by living radical polymerization of a rubamate compound.
- the method for producing a hyperbranched polymer according to the fifth aspect which is the dithiocanolobamate compound strength N, N-jetyldithiocarbamylmethylstyrene, and as a seventh aspect, the substitution reaction is The reaction is carried out by reacting a halogenating agent with a hyperbranched polymer having a dithiocanolevamate group at the molecular end in an organic solvent solution containing a hyperbranched polymer having a dithiopower rubamate group at the molecular end.
- a method for producing a hyperbranched polymer according to 5 aspects
- the halogenating agent includes chlorine, N-chlorosuccinimide, chlorinated isocyanuric acid, phosphorus trichloride, bromine, N-bromosuccinimide, N-bromodaltalimide, N, ⁇ ', ⁇
- the hyperbranched polymer according to the fifth aspect which uses at least one selected from “—trib mouth moisocyanuric acid, ⁇ -sodium bromoisocyanurate, iodine, ⁇ -odosuccinimide, potassium iodate, and periodic acid” Production method,
- the method for producing a hyperbranched polymer according to the fifth aspect wherein at least one of bromine and ⁇ -bromosuccinimide is used as the halogenating agent, and as a tenth aspect, the formula (5):
- R and ⁇ are as defined in the formula (1), and R and R are respectively Hydrogen atom, linear, branched or cyclic alkyl group having 1 to 20 carbon atoms, hydroxyl alkyl group having 1 to 5 carbon atoms, aryl group having 6 to 14 carbon atoms, 7 to 20 carbon atoms An arylalkyl group, an alkylaryl group having 7 to 20 carbon atoms, one (CH 3) —NH (CO) —0—C (CH 2) (where m is an integer of 2 to 6)
- N is the number of repeating unit structures and represents an integer of 2 to 100,000.
- 4 and R represent a hydrogen atom
- A is a structure represented by the formula (3).
- equation (6) As a thirteenth aspect, equation (6):
- R, R, R and R are each a hydrogen atom or a straight chain of 1 to 20 carbon atoms.
- Branched or cyclic alkyl groups hydroxyl alkyl groups of 1 to 5 carbon atoms, aryl groups of 6 to 14 carbon atoms, aryl hydrocarbon groups of 7 to 20 carbon atoms, 7 to 20 carbon atoms Alkylaryl group, one (CH) -NH (CO) — O—
- the hyperbranched polymer according to the thirteenth aspect representing 4 and R force S methyl group
- R is
- the hyperbranched polymer according to the thirteenth aspect which is a structure represented by the formula (3), and as the sixteenth aspect, the weight average molecular weight measured in terms of polystyrene by gel permeation chromatography is 500 to The hyperbranched polymer according to the tenth aspect or the fifteenth aspect, which is 5,000,000,
- a seventeenth aspect includes a step of reacting a hyperbranched polymer having a halogen atom at the molecular end represented by the formula (1) with an amine compound in water and / or an organic solvent solution in the presence of a base. , A method for producing a hyperbranched polymer according to the tenth aspect or the thirteenth aspect,
- a hyperbranched polymer having a halogen atom at the molecular end represented by the formula (1) is reacted with phthalimide in the presence of a base in water and / or an organic solvent solution to form the molecular end.
- the method for producing a hyperbranched polymer according to the eleventh aspect which includes the second step of converting NH into a hyperbranched polymer.
- the hyperbranched polymer having a halogen atom which is a reactive functional group at the molecular end of the present invention is a novel optically stable hyperbranched polymer. Furthermore, the hyperbranched polymer can obtain a hyperbranched polymer derivatized using a known reaction. In addition, the production method of the present invention can easily and efficiently obtain a hyper-branched polymer having these characteristics.
- a hyperbranched polymer having molecular terminals derived from an amino group or an ammonium group is provided.
- a hyperbranched polymer having an ammonium group at the molecular end is soluble in water and alcohol solvents such as methanol and ethanol, and is also useful as a hydrophilizing agent for glass substrates. Further, by aminating the molecular terminals, it becomes possible to induce various functional hyperbranched polymers that can react with an acid such as a carboxylic acid group, and further derivatization is expected.
- the hyperbranched polymer having a halogen atom at the molecular end of the present invention has a structure represented by the formula (1).
- X represents a halogen atom.
- R is a hydrogen atom or methyl
- n is the number of repeating unit structures and represents an integer of 2 to 100,000.
- A represents the structure represented by the formula (2).
- A represents a linear, branched or cyclic alkylene group having 1 to 30 carbon atoms which may contain an ether bond or an ester bond, and Y or Y
- 3 4 represents a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a nitro group, a hydroxyl group, an amino group, a carboxyl group, or a cyan group, respectively.
- alkylene group of A examples include a methylene group, an ethylene group, a normal propylene group, a normal butylene group, a normal alkylene group such as a normal hexylene group, an isopropylene group, an isobutylene group, and a 2-methylpropylene group.
- a branched alkylene group may be mentioned.
- the cyclic alkylene group include alicyclic aliphatic groups having a monocyclic, polycyclic and bridged cyclic structure having 3 to 30 carbon atoms. Specifically, a group having a monocyclo, bicyclo, tricyclo, tetracyclo, pentacyclo structure or the like having 4 or more carbon atoms
- alkyl group having 1 to 20 carbon atoms of Y, Y, Y or ⁇ examples include a methyl group
- alkoxy group having 1 to 20 atoms examples include methoxy group, ethoxy group, isopropoxy group, cyclohexyloxy group, and normal pentyloxy group.
- Y is preferably a hydrogen atom or an alkyl group having 1 to 20 carbon atoms.
- a in the formula (1) is preferably a structure represented by the formula (3).
- the repeating unit structure represented by the formula (8) is linked to the structure of the starting point portion having a bulle group represented by the formula (7). It has the same structure.
- Equation (15) can be considered.
- n which represents the number of repeating unit structures, is 4 or more, many more structures are possible.
- the hyperbranched polymer having the structure represented by the formula (1) of the present invention has a structure in which a repeating unit structure is regularly bonded at three points to form a branched structure, and bonded at two points. , Also includes! / In the case of a linear structure without branching.
- the hyperbranched polymer of the present invention may have a force that can be considered to be a single repeating unit structure or two or more repeating unit structures.
- the arrangement pattern of the copolymer may be a random copolymer, an alternating copolymer, or a block copolymer.
- Weight average molecular weight measured by gel permeation chromatography in terms of polystyrene Mw force 500 is less than 5,000,000, and preferably ⁇ (or less than 1, 000, 1,000,000 , Better than girls ⁇ i 2,000! /, And 500,000, and H is better than girls ⁇ i 3,000! /, And 100,000, and the dispersion Mw (weight average molecular weight ) / Mn (number average molecular weight) is 1.0, 7.0, and is like a girl, il. 1 is 6.0, more like a girl, il. 2 is 5.0 so
- the hyperbranched polymer having a structure represented by the formula (1) of the present invention has a dithiocanolevamate group obtained by living radical polymerization of a dithiocanolevamate compound represented by the formula (4).
- R and A have the same definitions as in formula (1).
- R and R are
- 1 1 2 3 represents an alkyl group having 1 to 5 carbon atoms, a hydroxyalkyl group having 1 to 5 carbon atoms or an arylalkyl group having 7 to 12 carbon atoms.
- R and R are
- Examples of the alkyl group having 1 to 5 carbon atoms include a methyl group, an ethyl group, an isopropyl group, a tbutyl group, a cyclopentyl group, and a normal pentyl group.
- Examples of the hydroxyalkyl group having 1 to 5 carbon atoms include a hydroxymethyl group, a hydroxyethyl group, and a hydroxypropyl group.
- Examples of the arylalkyl group having 7 to 12 carbon atoms include a benzyl group and a phenethyl group.
- Examples of the ring formed by R and R bonded to each other and the nitrogen atom bonded to them include four- and eight-membered rings.
- Examples of the ring include a ring containing 4 to 6 methylene groups. Further, examples of the ring include a ring containing an oxygen atom or sulfur atom and 4 to 6 methylene groups. Specific examples of the ring formed by combining R and R with each other and the nitrogen atom bonded to them include a piperidine ring, a pyrrolidine ring, a morpholine ring, a thiomorpholine ring, and a homopiperidine ring.
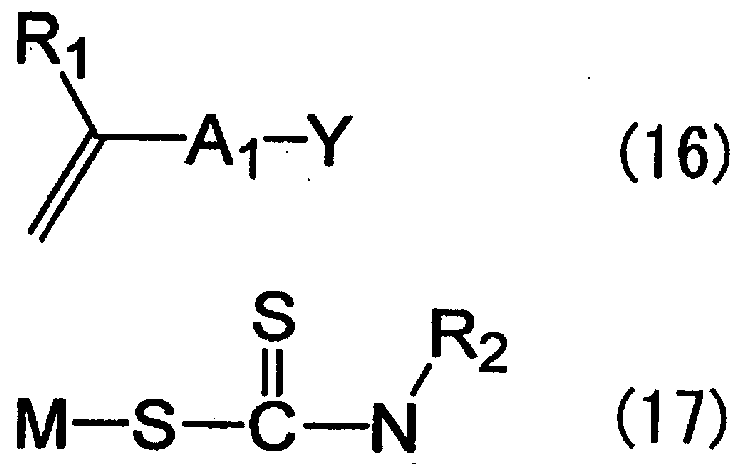
- the compound represented by the formula (4) can be easily obtained by a nucleophilic substitution reaction between the compound represented by the following formula (16) and the compound represented by the formula (17).
- Y represents a leaving group.
- the leaving group include a fluoro group, a black mouth group, a bromo group, a iodine group, a mesyl group, and a tosyl group.
- M represents lithium, sodium, or lithium.
- This nucleophilic substitution reaction is usually preferably carried out in an organic solvent that can dissolve both of the above two types of compounds.
- the compound represented by the formula (4) can be obtained with high purity by performing a liquid separation treatment and a recrystallization treatment with a water / non-aqueous organic solvent.
- the compound represented by the formula (4) is produced by referring to the method described in Macromol. Rapid Commun. 21, 665-668 (2 000) or (also Polymer International 51, 424-428 (2002)). It ’s the power to do.
- a hyperbranched polymer having a dithiocarbamate group at the molecular end can be obtained by subjecting the compound represented by the formula (4) to living radical polymerization.
- Living radical polymerization of the compound represented by the formula (4) can be carried out by a known polymerization method such as bulk polymerization, solution polymerization, suspension polymerization, or emulsion polymerization. Of these polymerization methods, solution polymerization is preferred.
- the polymerization reaction can be carried out at an arbitrary concentration in a solvent capable of dissolving the compound represented by formula (4).
- concentration of the compound represented by the formula (4) is arbitrary, but is, for example, 1 to 80% by mass, preferably 2 to 70% by mass, more preferably 5 to 60% by mass, most preferably 30 to 30% by mass. 50% by mass.
- the solvent is not particularly limited as long as it can dissolve the compound represented by formula (4).
- aromatic hydrocarbons such as benzene, toluene, xylene, and ethylbenzene
- ether compounds such as tetrahydrofuran and diethyl ether
- ketone compounds such as acetone, methyl ethyl ketone, methyl isobutanol ketone, and cyclohexanone
- normal heptane and aliphatic hydrocarbons such as normal hexane and cyclohexane.
- One of these solvents may be used for V, or may be used as a mixture of two or more! /.
- Living radical polymerization of the compound represented by the formula (4) can be performed in a solvent by heating or irradiation with light such as ultraviolet rays. And it is preferable to carry out by irradiation with light such as ultraviolet rays.
- living radical polymerization it is necessary to sufficiently remove oxygen in the reaction system before the start of polymerization, and the system may be replaced with an inert gas such as nitrogen or argon.
- the polymerization time is, for example, 0.1 to 100 hours, preferably 1 to 50 hours, and more preferably 3 to 30 hours. Usually, the conversion rate of the monomer ⁇ compound represented by formula (4) ⁇ increases with the lapse of polymerization time.
- the polymerization temperature is not particularly limited, but is, for example, 0 to 200 ° C, preferably 10 to 150 ° C, more preferably 20 to 100 ° C.
- a chain transfer agent such as mercaptans and sulfides, or a sulfide compound such as tetraethylthiuram disulfide can be used to adjust the molecular weight and molecular weight distribution.
- antioxidants such as hindered phenols, UV absorption of benzotriazoles, etc. ", 4 tert butylcatechol, hydroquinone, nitrophenol, nitrocresol, picric acid, phenothiazine, dithiobenzoyl disulfide
- a polymerization inhibitor such as id can be used.
- a known butyl monomer having no dithiocarbamate group or a compound having an unsaturated double bond can be added in order to adjust the degree of branching and the degree of polymerization.
- These can be used in a proportion of less than 50 mol% with respect to the compound represented by the formula (4).
- Specific examples thereof include styrenes, vinylbiphenyls, urnaphthalenes, buranthracenes, acrylic esters, methacrylic esters, acrylamides, methacrylolamides, vinylol pyrrolidones, phthalonitriles, Mention may be made of maleic acids, maleimides, dibules and tribules.
- the hyperbranched polymer represented by the formula (1) having a halogen atom at the molecular end of the present invention is substituted by replacing the hyperbranched polymer having a dithiopower rubamate group at the molecular end obtained as described above with a halogen atom. Obtainable.
- Halogenating agents that can be used in this reaction include chlorine, N-chlorosuccinimide, chlorinated isocyanuric acid, sulfuryl chloride, tertiary butylno, ivochloride, phosphorus trichloride, phosphorus pentachloride, triphenylphosphine dichloride, cupric chloride.
- Chlorinating agents such as antimony pentachloride, bromine, N-bromosuccinimide, N-bromo gnoretanolimide, N, ⁇ ', ⁇ , monotribromoisocyanuric acid, ⁇ , ⁇ Sodium nullate, ⁇ , ⁇ , potassium monobromoisocyanurate, ⁇ , ⁇ , monodibromoisocyanate, ⁇ -sodium bromoisocyanurate, ⁇ , N'-dibromohydantoin, ⁇ - Methylhydantoin, 1,3-dib-mouthed 5,5, -dimethylhydantoin, 3-bromo-5,5, monodimethylhydantoin, 3-bromo-5,5-dimethylhydantoin, 1-bromo-5
- Brominating agents such as 3-bromo-5,5-dimethylhydantoin sodium, 3-bromo-5,5'-dimethylhydantoin potassium, iodine, N-iodosuccinimide, potassium iodate, potassium periodate, periodate
- An iodinating agent such as acid or iodic acid can be used.
- the amount of the halogenating agent used is 1 to 20 times molar equivalent, preferably 1.5 to 15 times molar equivalent, more preferably 2 to 10 times molar equivalent, relative to the number of dithiorubbamate groups in the hyperbranched polymer. If it is.
- the conditions for the substitution reaction are appropriately selected from a reaction time of 0.01 or 100 hours and a reaction temperature of 0 to 300 ° C. Preferably reaction time 0.
- the reaction temperature is 20 to 150 ° C for 1 to 10 hours.
- the reaction for substituting the dithiopower rubamate group at the molecular end with a halogen atom is preferably carried out in water or an organic solvent.
- the solvent used is preferably a solvent capable of dissolving the hyperbranched polymer having a dithio-rubamate group and the halogenating agent.
- the solvent is the same as the solvent used in the production of the hyperbranched polymer having a dithiorubamate group because the reaction operation is simplified.
- a reaction is preferably performed by heating and refluxing in an organic solvent solution using a halogenating agent such as bromine.
- a halogenating agent such as bromine.
- the organic solvent any organic acid solvent such as acetic acid may be used as long as it does not significantly inhibit the progress of the reaction, aromatic hydrocarbons such as benzene, toluene, xylene, ethylbenzene, 1,2-dichlorobenzene, Ether compounds such as tetrahydrofuran and jetyl ether, ketone compounds such as acetone, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexanone, chloroform, formaldehyde, dichloromethane, 1,2-dichloroethane, noremanole heptane, Aliphatic hydrocarbons such as nonolemanolehexane and cyclohexane can be used.
- the mass of the hyperbranched polymer having a dithiopower rubamate group at the end of the molecule is 0.2 to 1,000 times, preferably 1 to 500 times, more preferably 5 to 100 times, most preferably It is preferable to use 10 or 50 times mass organic solvent.
- the reaction starts Prior to this, it is necessary to sufficiently remove oxygen in the reaction system, and it is better to replace the system with an inert gas such as nitrogen or argon.
- the reaction conditions are appropriately selected from a reaction time of 0.01 to 100 hours and a reaction temperature of 0 to 200 ° C.
- the reaction time is preferably 0.1 to 5 hours, and the reaction temperature is 20 to 150 ° C.
- aqueous solution of a reducing agent such as sodium thiosulfate or sodium sulfite, or sodium hydroxide, potassium hydroxide, hydroxide
- a reducing agent such as sodium thiosulfate or sodium sulfite
- sodium hydroxide potassium hydroxide
- hydroxide sodium hydroxide
- An alkaline aqueous solution such as calcium
- it may be reacted with a compound containing an unsaturated bond such as ethylene, propylene, butene and chlorohexene.
- the amount used may be 0.1 to 50 equivalents, preferably 0.5 to 10 equivalents, more preferably 1 to 3 equivalents, relative to the halogenating agent used.
- the hyperbranched polymer having a halogen atom at the molecular end of the present invention obtained by the reaction as described above can be separated from the solvent from the reaction solution by distilling off the solvent or by solid-liquid separation. Further, the hyperbranched polymer having a halogen atom at the molecular end of the present invention can be precipitated by adding the reaction solution into a poor solvent, and recovered as a powder.
- a part of the molecular terminal may remain as a dithiorubamate group! /.
- R represents a hydrogen atom or a methyl group. Also in formula (5) or formula (6), R represents a hydrogen atom or a methyl group. Also in formula (5) or formula (6)
- A is the formula (2):
- A represents a linear, branched or cyclic alkylene group having 1 to 30 carbon atoms which may contain an ether bond or an ester bond
- Y, Y, Y or Y is
- 1 2 3 4 represents a hydrogen atom, an alkyl group having 1 to 20 carbon atoms, an alkoxy group having 1 to 20 carbon atoms, a nitro group, a hydroxyl group, an amino group, a carboxyl group, or a cyan group, respectively.
- R and R are bonded to each other and are shared with the nitrogen atom bonded to them.
- a ring may be formed.
- n is the number of repeating unit structures and represents an integer of 2 to 100,000.
- X— is the negative value of the halogen atom. Represents
- Examples of the linear alkyl group having 1 to 20 carbon atoms represented by R, R, and R include meso
- Til Til, ethyl, n propyl, n butyl, n pentyl, n hexyl, n heptyl, n octyl, n nonyl, n decyl, n undecyl, n dodecinole, n tridenol Group, n-tetradenole group, n-pentadecinole group, n-hexadecyl group, n-heptadecyl group, n-octadecyl group, n-nonadecyl group, n-eicosyl group and the like.
- Examples of the branched alkyl group include isopropyl group, isobutyl group, sec butyl group, tert butyl group and the like.
- Examples of the cyclic alkyl group include a group having a cyclopentyl ring and a cyclohexyl ring structure.
- Examples of the hydroxylalkyl group having 1 to 5 carbon atoms include a hydroxymethyl group, a hydroxychetyl group, and a hydroxypropyl group.
- Examples of aryl groups having 6 to 14 carbon atoms include phenyl, biphenyl, naphthyl, anthraquino, anthracene, fluorene, fluorenone, indane, phenanthrene, and quinoline groups.
- Examples of the arylalkyl group having 7 to 20 carbon atoms include a benzyl group and a phenethyl group.
- alkylaryl group having 7 to 20 carbon atoms examples include pn butylphenyl group, p- tert butylphenyl group, p- n octylphenyl group, p —n decylphenyl group, p- n dodecylaniline group, p- n tetradecyl group And phenyl group.
- R and R are bonded to each other, and the nitrogen atom bonded to them
- Examples of the ring formed together with the element include a phthalimide group, a pyrrole ring, a piperidine ring, a piperazinol ring, and an imidazole ring.
- R, R and R are bonded to each other and bonded to them.
- Examples of the ring formed with the nitrogen atom include a pyridine ring, a pyrimidine ring, a pyrazine ring, a quinoline ring, and a piperidyl ring.
- examples of the halogen atom in the formula (6) include a chlorine atom, an aromatic atom, and an iodine atom.
- the hyperbranched polymer having the structure represented by the formula (5) having an amino group at the molecular end or the formula (6) having an ammonium group has an amine compound as a hyperbranched polymer having a halogen atom at the molecular end. It can be obtained by reacting. Therefore, the hyperbranched polymer having the structure represented by the formula (5) or the formula (6) is described in detail in the structure of the hyperbranched polymer having a halogen atom at the molecular end represented by the formula (1). It has a similar structure and has a structure in which a halogen atom is replaced with an amino group or an ammonium group.
- the amine compounds that can be used in this reaction are N-methylamine, N-ethylamine, N-n-propylamine, N-isopropylamine, N-n-butylamine, N-n-isobutyramine, N-sec butyramine as primary amines.
- Aminobiphenyl such as aminoanthraquinone, 4-aminobiphenyl, 2-aminobiphenyl, 2-aminofluorenaminofluorene, 1-amino-9fluorenone, 4-amino-9-fluorenone, etc.
- Aminoisoquinoline Aminoisoquinoline of 9 ⁇ Minofu Henin train aromatic amino Hue phenanthrene such as Amin and the like.
- Secondary amines include N, N dimethylamine, N, N jetylamine, N, N di-n-propylamine, N, N di-isopropylamine, N, N di-n-butylamine, N, N- n Isobutyramine, N, N Disec sec Butylamine, N, N—n Pentenoreamine, N Methyl N Ethylamine, N Methyl N—n Propylamine, N—Methyl-N—n Butyramine, N Methyl-N—n—Pentylamine, N Ethyl N Isopropylamine, N Ethyl N—n Butylamine, N Ethyl N—n—Pentylamine, N Methyl-N—n Octylamine, N Methylenole N—n—Decinoleamine, N Methyl-N—n Dodecylamine, N Methyl-N—n Tetradecylamine, N
- N, N di (2-hydroxyethynole) amine, N, N di (3-hydroxypropynole) amine, N, N di (ethoxychechinole) amine, N, N di (propoxychetil) amine and the like can be mentioned.
- Tertiary amines include trimethylamine, triethylamine, tripropynoleamine, tribubutinoreamine, tripentinoreamine, N, N dimethyl-Noctylamine, N, N jetty N- n decylamine, N, N Dimethyl-N- n Dodecylamine, N, N Dimethyl— N— n Tetradecylamine, N, N Dimethyl— N— n Hexadecylamine, N, N Dimethyl-N— n Octadecylamine, N, N Dimethinole N — N Eicosylamine, N, N dimethyl-N— n Aliphatic amines such as dodecylamine, pyridine, virazine, pyrimidine, quinoline, 1-methinoreidamidole, 4, 4'-pipyridinore, 4-methinore 4, 4'-pipyridyl, etc. Of the nitrogen-containing heterocyclic
- the amount of the amine compound that can be used in these reactions is 0.1 to 20 with respect to 1 molar equivalent of the halogen atom in the hyperbranched polymer having a halogen atom at the molecular end. Double molar equivalent, preferably 0.5 to 10 molar equivalent, more preferably 1 to 5 molar equivalent.
- the reaction conditions are appropriately selected from a reaction time of 0.01 to 100 hours and a reaction temperature of 0 to 300 ° C. Preferably, the reaction time is 0.1 to 10 hours and the reaction temperature is 20 to 150 ° C.
- the reaction between the halogen atom at the molecular terminal and the amine compound can be carried out in water or an organic solvent solution in the presence or absence of a base.
- the solvent used is preferably a solvent capable of dissolving the hyperbranched polymer having a halogen atom and the amine compound.
- a hyperbranched polymer having a halogen atom and an amine compound can be dissolved, but a solvent that does not dissolve a hyperbranched polymer having an amino group or an ammonium group at the molecular end is more suitable for isolation. is there.
- organic solvents water and organic acid solvents such as acetic acid are acceptable as long as they do not significantly inhibit the progress of this reaction, and aromatic carbonization such as benzene, toluene, xylene, ethylbenzene, and 1,2-dichlorobenzene.
- Ether compounds such as hydrogen, tetrahydrofuran and jetyl ether, ketone compounds such as acetone, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexanone, chloroform, formaldehyde, dichloromethane, 1,2-dichloroethane, and noranolheptane
- Aliphatic hydrocarbons such as normal hexane, cyclohexane, dimethylformamide, dimethylacetamide, N-methylpyrrolidone, and the like can be used. These solvents may be used alone or in combination of two or more.
- the amount used is 0.2 to 1,000 times, preferably 1 to 500 times, more preferably 5 to 100 times the mass of the hyperbranched polymer having a halogen atom at the molecular end. It is preferable to use an organic solvent having a mass of 10 to 50 times. Further, in this reaction, it is necessary to sufficiently remove oxygen in the reaction system before the start of the reaction, and the system may be replaced with an inert gas such as nitrogen or argon.
- the reaction conditions are appropriately selected from a reaction time of 0.01 to 100 hours and a reaction temperature of 0 to 200 ° C. The reaction time is preferably 0.1! /, 5 hours, and the reaction temperature is 20/150 ° C.
- Suitable bases are generally alkali metal hydroxides and alkaline earth metal hydroxides, alkali metal oxides and alkaline earth metal oxides, alkali metal hydrides and alkaline earth metal hydrides, alkali metals.
- Amides, alkali metal carbonates and alkalis Inorganic compounds such as earth metal carbonates (eg lithium carbonate, potassium carbonate, calcium carbonate), alkali metal bicarbonates (eg sodium bicarbonate), as well as alkali metal alkyls, alkylmagnesium halides, alkali metal alkoxides, alkalis Organometallic compounds such as earth metal alkoxides and dimethoxymagnesium are used. Particularly preferred are potassium carbonate and sodium carbonate.
- the amount used is 0.2 to 10 times equivalent, preferably 0.5 to 10 equivalents, most preferably 1 to 5 equivalents, relative to the mass of the hyperbranched polymer having a halogen atom at the molecular end. It is preferred to use a base.
- the hydrolysis may be performed using a hydrazine derivative such as hydrazine, methyl hydrazine, phenyl hydrazine, etc.
- a hydrazine derivative such as hydrazine, methyl hydrazine, phenyl hydrazine, etc.
- R and R are both hydrogen (22)
- the hyperbranched polymer having an amino group or an ammonium group at the molecular end of the present invention obtained by the reaction method as described above is separated from the solvent by distilling off the solvent or solid-liquid separation from the reaction solution. That power S.
- the hyperbranched polymer of the present invention can be precipitated by adding the reaction solution into a poor solvent and recovered as a powder.
- a part of the molecular end may remain as a halogen atom.
- Pretreatment equipment Automatic sample combustion equipment manufactured by Diane Instruments Co., Ltd. AQF—10 type
- Combustion tube temperature 1000 ° C
- Pretreatment equipment Automatic sample combustion equipment manufactured by Diane Instruments Co., Ltd. AQF—10 type
- Combustion tube temperature 1000 ° C
- This reaction crude powder was redissolved in toluene, separated in a toluene / water system, and then recrystallized from the toluene phase in a freezer at 20 ° C under zero.
- the recrystallized product was filtered and vacuum-dried to obtain 206 g (yield 97%) of the target product as a white powder.
- the purity (relative area percentage) by liquid chromatography was 100%.
- the melting point determined by DSC was 56 ° C.
- reaction solution was added to 3000 g of methanol to reprecipitate the polymer in the form of a highly viscous lump, and then the supernatant was removed by decantation.
- the weight average molecular weight Mw measured by gel permeation chromatography in terms of polystyrene was 6,600, and the degree of dispersion Mw / Mn was 2.2. Elemental analysis was 50.2 wt% carbon, 3.8 wt% hydrogen, less than 1.0 wt% nitrogen and 33.2 wt% bromine.
- Fig. 2 shows the NMR spectrum measurement results. The peak derived from the methylene group of the dithiocarbamate group at 4. ppm and 3.7 ppm disappeared as shown in Figure 1, and the peak derived from the methyl group of 1.3 ppm dithiocarbamate group decreased. It was observed.
- the obtained hyperbranched polymer has a structure represented by the formula (23).
- Fig. 3 shows the NMR spectrum measurement results.
- the peak derived from the methylene group of the 4.4 ppm benzyl group shown in Fig. 2 has shifted to 4.7 ppm, and a new peak derived from the methyl group was observed at 3.3 ppm. This revealed that the bromine atom at the end of the hyperbranched polymer obtained in Example 1 was substituted with almost 100% trimethylammonium group.
- the obtained hyperbranched polymer has a structure represented by the formula (24).
- the obtained brown solid was dissolved in 10 g of black mouth form, reprecipitated with 100 g of methanol, and dried to obtain 1.3 g of a light brown powder.
- the measurement result of H-NMR spectrum is shown in FIG.
- the peak force S derived from the methylene group of the 4.4 ppm benzyl group shown in Fig. 2 has been shifted to 4.7 ppm, and it is newly 7.5 ppm or 7.8 ppm.
- a peak was observed. This revealed that the bromine atom at the end of the hyperbranched polymer obtained in Example 1 was almost 100% phthalimidated.
- the obtained hyperbranched polymer has a structure represented by the formula (25).
- the NMR spectrum measurement results are shown in FIG. Since the broad peak derived from 7.5 ppm to 7.8 ppm of the aromatic ring disappeared in FIG. 4, the phthalimide moiety at the end of the hyperbranched polymer obtained in Example 3 was almost 100%. It became clear that it was converted to amin.
- the obtained hyperbranched polymer has a structure represented by the formula (27).
- the hyperbranched polymer synthesized in Example 2 was added to methanol so as to be 10% by mass.
- a dissolved solution was prepared. This solution is treated with a 0.45 m syringe filter, applied to the entire surface of a glass substrate cleaned with a detergent and ion-exchanged water using a spin coater (300 rpm x 5 seconds, 2500 rpm x 25 seconds), and hot plate And dried at 150 ° C for 5 minutes.
- the contact angle of pure water with the glass substrate surface-treated with the hyperbranched polymer was 21.0 °.
- the contact angle of pure water with respect to the untreated glass substrate was 41.9 °. From this, the glass substrate surface was hydrophilized by coating with the hyperbranched polymer synthesized in Example 2.
- the novel hyperbranched polymer having a halogen atom at the molecular end of the present invention contains a norogen and a rogen atom as a reactive functional group
- the novel hyperbranched polymer as described in Example 2 and Example 5 was used. It can also be used as an intermediate for producing a hyperbranched polymer.
- it can be derived into various hyperbranched polymers using organic synthesis reactions such as hydroxyloleation, etherification, nitrylation, thioetherification, thiolation, and phosphorylation.
- Example 5 can react with an acid having a carboxylic acid group or the like, and has various functional hyperbranches. It can be further derived into polymers and the like. Further, the hyperbranched polymer having an ammonium group at the molecular terminal is soluble in water and alcohol solvents such as methanol and ethanol, and is useful as a hydrophilic treatment agent for glass substrates. Industrial applicability
- the hyperbranched polymer of the present invention includes a coating material, an adhesive material, a resin filler, various molding materials, a nanometer-size porous forming agent, a resist material, an electronic material, a printing material, a battery material, a medical material, an intermediate material. It can be used as a body raw material.
- FIG. 1 is a 1 H-NMR spectrum of the hyperbranched polymer obtained in Reference Example 2.
- FIG. 2 is a 1 H-NMR of the hyperbranched polymer obtained in Example 1.
- Fig. 3 is a 1 H-NMR spectrum of the hyperbranched polymer obtained in Example 2.
- FIG. 4 is a 1 H-NMR spectrum of the hyperbranched polymer obtained in Example 3.
- FIG. 5 is a 1 H-NMR spectrum of the hyperbranched polymer obtained in Example 4.
- FIG. 6 is a 1 H-NMR spectrum of the hyperbranched polymer obtained in Example 5.
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020087028953A KR101406330B1 (ko) | 2006-09-01 | 2007-08-29 | 하이퍼브랜치 폴리머 및 이의 제조방법 |
| JP2008533117A JP5499477B2 (ja) | 2006-09-01 | 2007-08-29 | ハイパーブランチポリマー及びその製造方法 |
| US12/310,543 US8338554B2 (en) | 2006-09-01 | 2007-08-29 | Hyperbranched polymer and method for producing the same |
| EP07806244A EP2058338B1 (en) | 2006-09-01 | 2007-08-29 | Hyperbranched polymer and method for producing the same |
| CN2007800313574A CN101506246B (zh) | 2006-09-01 | 2007-08-29 | 超支化聚合物及其制造方法 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006-237713 | 2006-09-01 | ||
| JP2006237713 | 2006-09-01 | ||
| JP2007115855 | 2007-04-25 | ||
| JP2007-115855 | 2007-04-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008029688A1 true WO2008029688A1 (fr) | 2008-03-13 |
Family
ID=39157119
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2007/066767 WO2008029688A1 (fr) | 2006-09-01 | 2007-08-29 | Polymère hyperbranché et son procédé de production |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8338554B2 (ja) |
| EP (1) | EP2058338B1 (ja) |
| JP (3) | JP5499477B2 (ja) |
| KR (1) | KR101406330B1 (ja) |
| CN (3) | CN102167756B (ja) |
| TW (1) | TWI465465B (ja) |
| WO (1) | WO2008029688A1 (ja) |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009031594A1 (ja) * | 2007-09-03 | 2009-03-12 | Nissan Chemical Industries, Ltd. | ジチオカルバメート基を有する高分子化合物からなる金属微粒子分散剤 |
| WO2009035042A1 (ja) * | 2007-09-12 | 2009-03-19 | Nissan Chemical Industries, Ltd. | ハイパーブランチポリマーの製造方法 |
| WO2009054455A1 (ja) * | 2007-10-26 | 2009-04-30 | Kyusyu University | ニトロキシル基を有するハイパーブランチポリマー |
| WO2009099055A1 (ja) * | 2008-02-07 | 2009-08-13 | Nissan Chemical Industries, Ltd. | 抗菌性ハイパーブランチポリマー |
| WO2009136626A1 (ja) * | 2008-05-07 | 2009-11-12 | 国立大学法人九州大学 | エレクトロクロミック材料 |
| WO2010021386A1 (ja) | 2008-08-22 | 2010-02-25 | 日産化学工業株式会社 | アンモニウム基を有する分岐高分子化合物からなる金属微粒子分散剤 |
| WO2010087379A1 (ja) * | 2009-01-27 | 2010-08-05 | 国立大学法人九州大学 | チオエステル基含有ハイパーブランチポリマー |
| WO2010095618A1 (ja) * | 2009-02-17 | 2010-08-26 | 国立大学法人九州大学 | 金属微粒子-高分子化合物複合体を含む有機層を有する有機電界発光素子 |
| WO2010101252A1 (ja) * | 2009-03-06 | 2010-09-10 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| WO2010101254A1 (ja) * | 2009-03-06 | 2010-09-10 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| JP2011037716A (ja) * | 2009-08-06 | 2011-02-24 | Nissan Chem Ind Ltd | 抗菌性ハイパーブランチポリマー |
| JP2011053416A (ja) * | 2009-09-01 | 2011-03-17 | Univ Of Electro-Communications | 金属ナノ粒子−アンモニウム基含有分岐高分子化合物複合体を含有する感光性組成物 |
| WO2012074051A1 (ja) * | 2010-12-01 | 2012-06-07 | 国立大学法人九州大学 | 含フッ素ハイパーブランチポリマー及びその製造方法 |
| WO2012121165A1 (ja) * | 2011-03-10 | 2012-09-13 | 国立大学法人九州大学 | 塩素化ハイパーブランチポリマーの製造方法 |
| WO2012141215A1 (ja) | 2011-04-12 | 2012-10-18 | 日産化学工業株式会社 | ハイパーブランチポリマー及び金属微粒子を含む無電解めっき下地剤 |
| JP2014043487A (ja) * | 2012-08-24 | 2014-03-13 | Nissan Chem Ind Ltd | エチレンオキサイド鎖を有するハイパーブランチポリマー及びその利用 |
| WO2014042215A1 (ja) | 2012-09-13 | 2014-03-20 | 日産化学工業株式会社 | 無電解めっき下地剤 |
| JP5667558B2 (ja) * | 2009-02-27 | 2015-02-12 | 日産化学工業株式会社 | 有機スイッチング素子及びその製造方法 |
| US9893366B2 (en) | 2012-02-14 | 2018-02-13 | National University Corporation Gunma University | Metal fine particle association and method for producing the same |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102234343B (zh) * | 2010-04-20 | 2015-08-19 | 南开大学 | 单分散的表面功能化聚合物微球树脂及其制备方法 |
| KR102520886B1 (ko) * | 2014-09-05 | 2023-04-14 | 닛산 가가쿠 가부시키가이샤 | 감광성 무전해 도금 하지제 |
| CN106687619B (zh) * | 2014-09-05 | 2019-12-31 | 日产化学工业株式会社 | 光固化性非电解镀基底剂 |
| US11384172B2 (en) * | 2015-12-14 | 2022-07-12 | Jsr Corporation | Polymer, antimicrobial agent, disinfectant, antimicrobial material, disinfectant material, antimicrobial method, and disinfecting method |
| DE112017006823T5 (de) | 2017-01-13 | 2019-10-02 | Maxell Holdings, Ltd. | Hyperverzweigtes Polymer, Metallrückgewinnungsmittel, Metallrückgewinnungsverfahren und Inhibitor der katalytischen Aktivität |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005040233A1 (fr) * | 2003-10-22 | 2005-05-06 | Rhodia Chimie | Procede pour oxyder partiellement ou totalement une ou plusieurs extremites thiocarbonylthio d'un polymere issu d'une polymerisation radicalaire controlee par addition-fragmentation reversible |
| JP2005226051A (ja) * | 2004-02-16 | 2005-08-25 | Toho Chem Ind Co Ltd | 遊離ラジカル源による高分子末端基の置換 |
| WO2006093050A1 (ja) * | 2005-03-03 | 2006-09-08 | Tokyo Institute Of Technology | ハイパーブランチポリマー及びその製造方法 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6252025B1 (en) * | 1998-08-11 | 2001-06-26 | Eastman Kodak Company | Vinyl hyperbranched polymer with photographically useful end groups |
| JP2001114826A (ja) * | 1999-10-20 | 2001-04-24 | Mitsubishi Chemicals Corp | 水溶性カチオン性重合体 |
| US7250475B2 (en) * | 2003-06-26 | 2007-07-31 | Symyx Technologies, Inc. | Synthesis of photoresist polymers |
| EP1698645A1 (en) | 2003-12-22 | 2006-09-06 | Lion Corporation | Hyperbranched polymer, process for producing the same and resist composition containing the hyperbranched polymer |
| CN1218979C (zh) * | 2003-12-30 | 2005-09-14 | 上海交通大学 | 超支化聚对氯甲基苯乙烯接枝的碳纳米管及其制备方法 |
| KR101314794B1 (ko) | 2005-10-25 | 2013-10-08 | 닛산 가가쿠 고교 가부시키 가이샤 | 표면 및/또는 계면이 개질된 폴리머 구조체, 및 그 제조방법 |
-
2007
- 2007-08-29 CN CN2011100481718A patent/CN102167756B/zh active Active
- 2007-08-29 CN CN2011100482369A patent/CN102167757B/zh active Active
- 2007-08-29 JP JP2008533117A patent/JP5499477B2/ja active Active
- 2007-08-29 CN CN2007800313574A patent/CN101506246B/zh active Active
- 2007-08-29 US US12/310,543 patent/US8338554B2/en active Active
- 2007-08-29 EP EP07806244A patent/EP2058338B1/en active Active
- 2007-08-29 WO PCT/JP2007/066767 patent/WO2008029688A1/ja active Application Filing
- 2007-08-29 KR KR1020087028953A patent/KR101406330B1/ko active IP Right Grant
- 2007-08-31 TW TW096132496A patent/TWI465465B/zh active
-
2013
- 2013-07-08 JP JP2013143108A patent/JP5748076B2/ja active Active
- 2013-07-08 JP JP2013143105A patent/JP5610171B2/ja active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005040233A1 (fr) * | 2003-10-22 | 2005-05-06 | Rhodia Chimie | Procede pour oxyder partiellement ou totalement une ou plusieurs extremites thiocarbonylthio d'un polymere issu d'une polymerisation radicalaire controlee par addition-fragmentation reversible |
| JP2005226051A (ja) * | 2004-02-16 | 2005-08-25 | Toho Chem Ind Co Ltd | 遊離ラジカル源による高分子末端基の置換 |
| WO2006093050A1 (ja) * | 2005-03-03 | 2006-09-08 | Tokyo Institute Of Technology | ハイパーブランチポリマー及びその製造方法 |
Non-Patent Citations (13)
| Title |
|---|
| KOJI ISHIZU AND AKIHIDE MORI: "Synthesis of hyperbranched polymers by self-addition free radical vinyl polymerization of photo functional styrene", MACROMOLECULAR RAPID COMMUNICATIONS, vol. 21, no. 10, 2000, pages 665 - 668, XP003002146 * |
| KOJI ISHIZU ET AL., POLYMER INTERNATIONAL, vol. 53, 2004, pages 259 - 265 |
| KOJI ISHIZU; AKIHIDE MORI, MACROMOL. RAPID COMMUN., vol. 21, 2000, pages 665 - 668 |
| KOJI ISHIZU; AKIHIDE MORI, POLYMER INTERNATIONAL, vol. 50, 2001, pages 906 - 910 |
| KOJI ISHIZU; TAKESHI SHIBUYA; AKIHIDE MORI, POLYMER INTERNATIONAL, vol. 51, 2002, pages 424 - 428 |
| KOJI ISHIZU; TAKESHI SHIBUYA; SUSUMU KAWAUCHI, MACROMOLECULES, vol. 36, no. 10, 2002, pages 3505 - 3510 |
| KOJI ISHIZU; YOSHIHIRO OHTA; SUSUMU KAWAUCHI, MACROMOLECULES, vol. 35, no. 9, 2002, pages 3781 - 3784 |
| MACROMOL. RAPID COMMUN., vol. 21, 2000, pages 665 - 668 |
| MACROMOLECULES, vol. 35, no. 9, 2002, pages 3781 - 3784 |
| MACROMOLECULES, vol. 36, no. 10, 2002, pages 3505 - 3510 |
| POLYMER INTERNATIONAL, vol. 51, 2002, pages 424 - 428 |
| See also references of EP2058338A4 |
| TAKAYUKI OTSU ET AL.: "Features of Living Radical Polymerization of Vinyl Monomers in Homogeneous System Using N,N-Diethyldithiocarbamate Derivatives as Photoiniferters", EUROPEAN POLYMER JOURNAL, vol. 31, no. 1, 1995, pages 67 - 78, XP000483750 * |
Cited By (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009031594A1 (ja) * | 2007-09-03 | 2009-03-12 | Nissan Chemical Industries, Ltd. | ジチオカルバメート基を有する高分子化合物からなる金属微粒子分散剤 |
| JP5435229B2 (ja) * | 2007-09-03 | 2014-03-05 | 日産化学工業株式会社 | ジチオカルバメート基を有する高分子化合物からなる金属微粒子分散剤 |
| WO2009035042A1 (ja) * | 2007-09-12 | 2009-03-19 | Nissan Chemical Industries, Ltd. | ハイパーブランチポリマーの製造方法 |
| JP5408435B2 (ja) * | 2007-09-12 | 2014-02-05 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| US8362190B2 (en) | 2007-09-12 | 2013-01-29 | Nissan Chemical Industries, Ltd. | Method for producing hyperbranched polymer |
| US7994258B2 (en) | 2007-10-26 | 2011-08-09 | Nissan Chemical Industries, Ltd. | Hyperbranched polymer having nitroxyl group |
| WO2009054455A1 (ja) * | 2007-10-26 | 2009-04-30 | Kyusyu University | ニトロキシル基を有するハイパーブランチポリマー |
| JP5570220B2 (ja) * | 2007-10-26 | 2014-08-13 | 国立大学法人九州大学 | ニトロキシル基を有するハイパーブランチポリマー |
| WO2009099055A1 (ja) * | 2008-02-07 | 2009-08-13 | Nissan Chemical Industries, Ltd. | 抗菌性ハイパーブランチポリマー |
| JPWO2009136626A1 (ja) * | 2008-05-07 | 2011-09-08 | 日産化学工業株式会社 | エレクトロクロミック材料 |
| WO2009136626A1 (ja) * | 2008-05-07 | 2009-11-12 | 国立大学法人九州大学 | エレクトロクロミック材料 |
| US8773745B2 (en) | 2008-05-07 | 2014-07-08 | Kyushu University | Electrochromic material |
| US20110183837A1 (en) * | 2008-08-22 | 2011-07-28 | Nissan Chemical Industries, Ltd. | Metal fine particle dispersant containing branched polymer compound having ammonium group |
| KR101743637B1 (ko) * | 2008-08-22 | 2017-06-05 | 닛산 가가쿠 고교 가부시키 가이샤 | 암모늄기를 가지는 분지고분자 화합물로 이루어진 금속 미립자 분산제 |
| KR101680259B1 (ko) * | 2008-08-22 | 2016-11-29 | 닛산 가가쿠 고교 가부시키 가이샤 | 암모늄기를 가지는 분지고분자 화합물로 이루어진 금속 미립자 분산제 |
| EP2332640A4 (en) * | 2008-08-22 | 2011-10-26 | Nissan Chemical Ind Ltd | METAL MICROPARTICLE DISPERSION PRODUCTS WITH BRANCHED POLYMER COMPOUND WITH AN AMMONIUM GROUP |
| KR20160062202A (ko) | 2008-08-22 | 2016-06-01 | 닛산 가가쿠 고교 가부시키 가이샤 | 암모늄기를 가지는 분지고분자 화합물로 이루어진 금속 미립자 분산제 |
| JP2015166084A (ja) * | 2008-08-22 | 2015-09-24 | 日産化学工業株式会社 | アンモニウム基を有する分岐高分子化合物からなる金属微粒子分散剤 |
| JP5761995B2 (ja) * | 2008-08-22 | 2015-08-12 | 国立大学法人九州大学 | アンモニウム基を有する分岐高分子化合物からなる金属微粒子分散剤 |
| US20140288264A1 (en) * | 2008-08-22 | 2014-09-25 | Kyushu University | Metal fine particle dispersant containing branched polymer compound having ammonium group |
| US10597491B2 (en) | 2008-08-22 | 2020-03-24 | Nissan Chemical Corporation | Metal fine particle dispersant containing branched polymer compound having ammonium group |
| US8722562B2 (en) | 2008-08-22 | 2014-05-13 | Nissan Chemical Industries, Ltd. | Metal fine particle dispersant containing branched polymer compound having ammonium group |
| WO2010021386A1 (ja) | 2008-08-22 | 2010-02-25 | 日産化学工業株式会社 | アンモニウム基を有する分岐高分子化合物からなる金属微粒子分散剤 |
| JP5553776B2 (ja) * | 2009-01-27 | 2014-07-16 | 国立大学法人九州大学 | チオエステル基含有ハイパーブランチポリマー |
| WO2010087379A1 (ja) * | 2009-01-27 | 2010-08-05 | 国立大学法人九州大学 | チオエステル基含有ハイパーブランチポリマー |
| US20110318554A1 (en) * | 2009-01-27 | 2011-12-29 | Nissan Chemical Industries, Ltd. | Hyperbranched polymer containing thioester groups |
| WO2010095618A1 (ja) * | 2009-02-17 | 2010-08-26 | 国立大学法人九州大学 | 金属微粒子-高分子化合物複合体を含む有機層を有する有機電界発光素子 |
| JP5667558B2 (ja) * | 2009-02-27 | 2015-02-12 | 日産化学工業株式会社 | 有機スイッチング素子及びその製造方法 |
| JP5594482B2 (ja) * | 2009-03-06 | 2014-09-24 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| WO2010101252A1 (ja) * | 2009-03-06 | 2010-09-10 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| WO2010101254A1 (ja) * | 2009-03-06 | 2010-09-10 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| JP5534244B2 (ja) * | 2009-03-06 | 2014-06-25 | 日産化学工業株式会社 | ハイパーブランチポリマーの製造方法 |
| JP2011037716A (ja) * | 2009-08-06 | 2011-02-24 | Nissan Chem Ind Ltd | 抗菌性ハイパーブランチポリマー |
| JP2011053416A (ja) * | 2009-09-01 | 2011-03-17 | Univ Of Electro-Communications | 金属ナノ粒子−アンモニウム基含有分岐高分子化合物複合体を含有する感光性組成物 |
| JP5872484B2 (ja) * | 2010-12-01 | 2016-03-01 | 国立大学法人九州大学 | 含フッ素ハイパーブランチポリマー及びその製造方法 |
| WO2012074051A1 (ja) * | 2010-12-01 | 2012-06-07 | 国立大学法人九州大学 | 含フッ素ハイパーブランチポリマー及びその製造方法 |
| US9243084B2 (en) | 2011-03-10 | 2016-01-26 | Kyushu University | Method for producing chlorinated hyperbranched polymer |
| JP5872540B2 (ja) * | 2011-03-10 | 2016-03-01 | 国立大学法人九州大学 | 塩素化ハイパーブランチポリマーの製造方法 |
| WO2012121165A1 (ja) * | 2011-03-10 | 2012-09-13 | 国立大学法人九州大学 | 塩素化ハイパーブランチポリマーの製造方法 |
| WO2012141215A1 (ja) | 2011-04-12 | 2012-10-18 | 日産化学工業株式会社 | ハイパーブランチポリマー及び金属微粒子を含む無電解めっき下地剤 |
| US9893366B2 (en) | 2012-02-14 | 2018-02-13 | National University Corporation Gunma University | Metal fine particle association and method for producing the same |
| JP2014043487A (ja) * | 2012-08-24 | 2014-03-13 | Nissan Chem Ind Ltd | エチレンオキサイド鎖を有するハイパーブランチポリマー及びその利用 |
| WO2014042215A1 (ja) | 2012-09-13 | 2014-03-20 | 日産化学工業株式会社 | 無電解めっき下地剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5499477B2 (ja) | 2014-05-21 |
| EP2058338A1 (en) | 2009-05-13 |
| CN102167757A (zh) | 2011-08-31 |
| CN102167757B (zh) | 2012-12-26 |
| KR101406330B1 (ko) | 2014-07-14 |
| JP2013231194A (ja) | 2013-11-14 |
| JP5748076B2 (ja) | 2015-07-15 |
| CN101506246A (zh) | 2009-08-12 |
| TW200825109A (en) | 2008-06-16 |
| US8338554B2 (en) | 2012-12-25 |
| US20100048845A1 (en) | 2010-02-25 |
| TWI465465B (zh) | 2014-12-21 |
| CN102167756B (zh) | 2012-12-26 |
| CN101506246B (zh) | 2011-06-22 |
| JPWO2008029688A1 (ja) | 2010-01-21 |
| EP2058338B1 (en) | 2012-08-15 |
| EP2058338A4 (en) | 2010-01-27 |
| KR20090045889A (ko) | 2009-05-08 |
| JP2013253246A (ja) | 2013-12-19 |
| JP5610171B2 (ja) | 2014-10-22 |
| CN102167756A (zh) | 2011-08-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2008029688A1 (fr) | Polymère hyperbranché et son procédé de production | |
| JP5187825B2 (ja) | ハイパーブランチポリマー及びその製造方法 | |
| JP5268644B2 (ja) | ハイパーブランチポリマー及びその製造方法 | |
| JP5630957B2 (ja) | ハイパーブランチポリマー及びその製造方法 | |
| US8362190B2 (en) | Method for producing hyperbranched polymer | |
| JP5983216B2 (ja) | 新規(メタ)アクリル酸エステル、その重合体および該重合体の製造方法 | |
| JP2003026724A (ja) | 重合体の製造方法 | |
| JP5872540B2 (ja) | 塩素化ハイパーブランチポリマーの製造方法 | |
| KR20230139095A (ko) | 화합물, 이를 포함하는 코팅 조성물, 화합물의 제조 방법 및 전자 소자 | |
| JP2008163172A (ja) | 新規(メタ)アクリルモノマー |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780031357.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07806244 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008533117 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020087028953 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12310543 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007806244 Country of ref document: EP |