WO2006038741A1 - チオエーテル化合物の製造方法 - Google Patents
チオエーテル化合物の製造方法 Download PDFInfo
- Publication number
- WO2006038741A1 WO2006038741A1 PCT/JP2005/018985 JP2005018985W WO2006038741A1 WO 2006038741 A1 WO2006038741 A1 WO 2006038741A1 JP 2005018985 W JP2005018985 W JP 2005018985W WO 2006038741 A1 WO2006038741 A1 WO 2006038741A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- carbon atoms
- represented
- compound
- formula
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/28—Radicals substituted by singly-bound oxygen or sulphur atoms
- C07D213/32—Sulfur atoms
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J31/00—Catalysts comprising hydrides, coordination complexes or organic compounds
- B01J31/16—Catalysts comprising hydrides, coordination complexes or organic compounds containing coordination complexes
- B01J31/24—Phosphines, i.e. phosphorus bonded to only carbon atoms, or to both carbon and hydrogen atoms, including e.g. sp2-hybridised phosphorus compounds such as phosphabenzene, phosphole or anionic phospholide ligands
- B01J31/2404—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring
- B01J31/2442—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems
- B01J31/2447—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems and phosphine-P atoms as substituents on a ring of the condensed system or on a further attached ring
- B01J31/2452—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems and phosphine-P atoms as substituents on a ring of the condensed system or on a further attached ring with more than one complexing phosphine-P atom
- B01J31/2457—Cyclic ligands, including e.g. non-condensed polycyclic ligands, the phosphine-P atom being a ring member or a substituent on the ring comprising condensed ring systems and phosphine-P atoms as substituents on a ring of the condensed system or on a further attached ring with more than one complexing phosphine-P atom comprising aliphatic or saturated rings, e.g. Xantphos
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C319/00—Preparation of thiols, sulfides, hydropolysulfides or polysulfides
- C07C319/14—Preparation of thiols, sulfides, hydropolysulfides or polysulfides of sulfides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/08—Indoles; Hydrogenated indoles with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/62—Oxygen or sulfur atoms
- C07D213/70—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F17/00—Metallocenes
- C07F17/02—Metallocenes of metals of Groups 8, 9 or 10 of the Periodic System
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2531/00—Additional information regarding catalytic systems classified in B01J31/00
- B01J2531/80—Complexes comprising metals of Group VIII as the central metal
- B01J2531/82—Metals of the platinum group
- B01J2531/824—Palladium
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
Definitions
- the present invention relates to an efficient and novel method for producing a thioether compound or a thiol compound useful as a pharmaceutical product or a pharmaceutical intermediate.
- the thioether compounds are disclosed as being useful as pharmaceuticals. For example, it has the inhibitory action of V iracept (AG 1343) HI V-1 protease, which is a thioter compound represented by the following chemical structural formula, and it has been put into practical use as a therapeutic agent for AIDS. It is disclosed. (See Stephen W. Kal dorr et al., Jenol reb med med i n a c i n a c emi s s t ryy, pp 40-3979-3985 (1997))
- Viracept (AG1343) International Publication No. WO 2004/08 1001 discloses a thioether compound useful as a therapeutic and / or prophylactic agent for diabetes, and as a therapeutic and / or prophylactic agent for diabetic complications. Yes. On the other hand, the following production methods are known as methods for producing thioether compounds.
- the reaction is carried out using K 2 C0 3 , NaHC0 3 and triethylamine as a weakly basic base, but the yield of the desired thioether compound is low and industrial. It is not suitable as a simple manufacturing method. Further, it is disclosed that the method for producing a thioether compound disclosed in Non-Patent Document 2 cannot be applied to a thiol nucleophile such as benzenethiol. That is, it can be said that this production method is a production method in which dialle sphere cannot be produced.
- the method for producing a thioether compound by the production method is not suitable as an industrial production method.
- the present invention was also completed by finding an efficient method for introducing a thiol group into a thiol ring or a heteroaryl ring.
- the method for producing a thioether compound and the method for producing a thiol compound of the present invention have excellent points as industrial production methods in the following points.
- a thiol compound substituted with a removable group is reacted with an aryl or heteroaryl compound having a chlorine group, a bromine group or a substituted sulfol group, to give a thioether compound.
- a thiol group can be efficiently introduced into the aryl ring or the heteroaryl ring by efficiently producing and then removing the removable group.
- the present invention relates to (1) to (2 1).
- R 1 is a hydrogen atom, a halogen atom, a cyano group, an alkyl group having 1 to 10 carbon atoms, an alkoxy group having 1 to 10 carbon atoms, an alkylthio group having 1 to 10 carbon atoms, carbon An alkylsphinyl group having 1 to 10 carbon atoms, a 1 to 10 carbon atoms ⁇ Kilsulfonyl group, hydroxyl group, carboxy group, alkoxy group having 2 to 10 carbon atoms NI
- Cicarbol group C2-C10 alkanoyloxy group, aryl group aryl group, arylcarbonyl group, heterocarbonyl group, heteroaryl group, heteroarylcarbonyl group, heteroarylcarbonyloxy group, nitro group
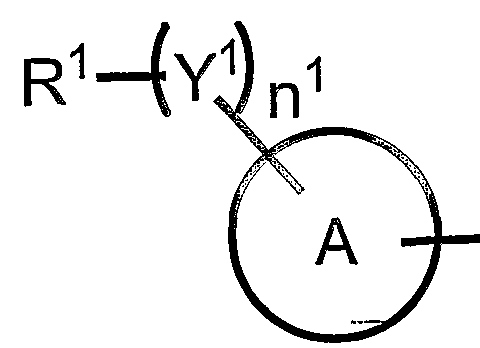
- Y 1 is a sulfur atom, sulfiel Group, sulfonyl group, oxygen atom, carbonyl group, oxycarbonyl group, carbonyloxy group and general formula:
- R 2 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group or a pyridyl group.
- X is a bromine atom, a chlorine atom, a trifluoromethanesulfonyloxy group, a methylsulfonyloxy group, a benzenesulfonyloxy group, a toluenesulfonyloxy group or a ditrobenzenesulfonyloxy group,
- n 1 is 0 or 1
- the group represented by represents an aryl group or a heteroaryl ring group.
- X is a chlorine atom
- I 1 , n 1 and Y 1 have the above-mentioned meanings.
- a palladium compound selected from the group consisting of palladium acetate, P d 2 (dba) 3 and P d (dba) 2 , Cesium carbonate, general formula
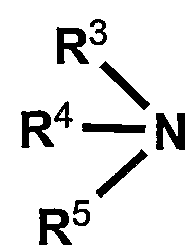
- R 3 R 4 and R 5 may be the same or different and each represents an alkyl group having 1 to 6 carbon atoms, a benzyl group, a vinyl group or a pyridyl group.
- R 3 R 4 and R 5 Selected from the group consisting of 1 5-diazabicyclo [4. 3. 0] nona 1 5 and 1 8-diazabicyclo [5. 4. 0] unde force 1-hen Base, and formula:
- R 6 or R 7 may be the same or different and each represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, an amino group or a phenyl group, n 2 represents 0 to 6, Hydrogen atom, halogen atom, cyano group, amino group, alkyl group having 1 to 10 carbon atoms, trimethylsilyl group, alkoxy group having 1 to 10 carbon atoms, alkylthio group having 1 to 10 carbon atoms, 1 to 1 carbon atom An alkylsulfinyl group having 0 carbon atoms, an alkylsulfonyl group having 1 to 10 carbon atoms, a hydroxyl group, a carboxy group, an alkoxycarbonyl group having 2 to 10 carbon atoms, an alkanoyloxy group having 2 to 10 carbon atoms, an aryl group, Arylene carbon
- Y 2 represents a sulfur atom, a sulfinyl group, a sulfonyl group, an oxygen atom, a carbonyl group, an oxycarbonyl group, a carbo-oxy group, and a general formula: human
- R 7 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group, or a pyridyl group.
- n 3 is 0 or 1
- R 9 is a hydrogen atom, a halogen atom, a cyano group, an amino group, a nitro group, an alkyl group having 1 to 10 carbon atoms, an alkoxy group having 1 to 10 carbon atoms, an alkylthio group having 1 to 10 carbon atoms, An alkylsphinyl group having 1 to 10 carbon atoms, an alkylsulfonyl group having 1 to 10 carbon atoms, a hydroxyl group, a carboxy group, an alkoxycarbonyl group having 2 to 10 carbon atoms, and an alkano having 2 to 10 carbon atoms Iloxy group, Lille group, arylenecarbonyl group, arylenecarbonyloxy group, heteroaryl group, heteroarylcarbonyl group, heteroarylcarbonyloxy group, nitrogen group, alkanoylamino group having 1 to 10 carbon atoms , An arylenecarbonylamino group, a heteroarylcarbonylamino group, or an alkano
- the group represented by represents an aryl group or a heteroaryl ring group. It is made to react with the thiol compound or its salt represented by this.
- the group represented by has the above-mentioned meaning.
- 1 3 or 1 ⁇ may be the same or different and each represents a hydrogen atom, an acetoxy group, A nitro group or an alkoxy group having 1 to 6 carbon atoms is shown. ), A general formula:
- n a is. Of 1 or 2) a group represented by (1 one-naphthyl) methyl group, (2-naphthyl) methyl group, 4-Asetokishifue group, 4 Asetokishifue two group, It represents a phenyl group, a trityl group, a diaminomethyl group, a 2-trimethylsilylethyl group or a 2- (2-ethylhexyloxycarbonyl) ethyl group.
- the thioether compound or its salt as described in (1) characterized by the above-mentioned.
- n a 1 or 2.
- R f is a 4-pyridylethyl group, 4-methoxyphenyl group, 4-1-pyridylmethyl group, benzyl group, 4-acetoxybenzyl group, 4-12 trobenzylyl group, 4-acetoxy group, A phenyl group, a phenyl group, a tritinole group, a diaminomethyl group, a 2-trimethylsilylethyl group or a 2- (2-ethylhexyloxycarbonyl) ethyl group;
- the phosphorus compound has the formula:
- Base is cesium carbonate, diisopropylethylamine, triptylamin, triethylamine, trimethylamine, dibenzylmethylamine, 4-dimethylaminepyridine, tribenzylamine, 1,5-diazabisic mouth [ 4. 3. 0]
- the palladium compound is P d 2 (dba) 3 and the phosphorus compound is represented by the formula:
- the base is cesium carbonate, disopropylethylamine, triptylamin, triethylamine, dibenzylmethylamine, 1,5-diazabicyclo [4. 3. 0] noner 5- Or 1,8-diazabicyclo [5. 4. 0]
- R 1 is a hydrogen atom, a halogen atom, a cyano group, an alkyl group having 1 to 10 carbon atoms, an alkoxy group having 1 to 10 carbon atoms, an alkylthio group having 1 to 10 carbon atoms, carbon Alkylsulfinyl group having 1 to 10 carbon atoms, alkylsulfonyl group having 1 to 10 carbon atoms, hydroxyl group, carboxy group, alkoxycarbonyl group having 2 to 10 carbon atoms, alkanoylo having 2 to 10 carbon atoms Xyl group, aryl group, aryl group, arylcarbonyl group, heteroaryl group, heteroaryl group, heteroarylcarbonyl group, two-necked group, 1 to 10 carbon atoms Arca noisy Rua amino group, ⁇ reel carbonyl ⁇ amino group, a Heteroa Li one Luca Lupo sulfonyl ⁇ amino group or aralkyl force
- R 2 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group or a pyridyl group.
- n 1 is 0 or 1
- a palladium compound selected from the group consisting of palladium acetate, P d 2 (dba) 3 and P d (dba) 2 ,
- R 3 , R 4 and R 5 may be the same or different and each represents an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group or a pyridyl group.
- R a X «R b tt may be the same or different and represents a hydrogen atom, an acetoxy group, a nitro group or an alkoxy group having 1 to 6 carbon atoms.
- n a is. Of 1 or 2) a group represented by (1 one-naphthyl) methyl group, (2-naphthyl) methyl group, 4 Asetokishifue group, 4-Asetokishifue two group, It represents a phenyl group, a trityl group, a diaminomethyl group, a 2-trimethylsilylethyl group or a 2- (2-ethylhexyloxycarbonyl) ethyl group.
- the group represented by has the above-mentioned meaning.
- R 7 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthinole group or a pyridyl group.
- S selected from the group consisting of groups represented by the following formula: S, an alkylene group of 1 to 6 which may be interposed in the carbon chain, methylene group, ethylene group, trimethylene group, tetramethylene group, pentamethylene group , Hexamethylene group, propylene group, ethylethylene group or general formula: ⁇
- Y represents a sulfur atom, a sulfinyl group, a sulfonyl group, an oxygen atom, a carbonyl group, an oxycarbonyl group, a carbonyloxy group, and a general formula:
- R 2 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group, or a pyridyl group, and ⁇ 4 and ⁇ 5 represent an integer of 1 to 6. The sum of both does not exceed 6.
- Base is cesium carbonate, -disopropylethylamine, triptylamin, triethylamine, trimethylamine, dibenzylmethylamine, 4-dimethylaminopyridine, tribenzylamine, 1,5- Jiazabik mouth [4. 3. 0] Nona 5-ene or 1, 8-Diazabicyclo [5. 4. 0] -Unde force 7- Nen A method for producing a compound or a salt thereof.
- the palladium compound is Pd 2 (dba) 3 and the phosphorus compound is represented by the formula:
- the base is cesium carbonate, disopropylethylamine, triptylamamine, triethylamine, trimethylamine, dibenzylmethylamine, 4-dimethylaminopyridine, tribenzylamine, 1,
- the thiol according to (10) characterized in that: 5—Jazabishi black [4. 3. 0] Noner 5—Yen or 1, 8—Jazabishik mouth [5. 4. 0] Unde force _ 7—Yen
- the protecting group represented by R e has the general formula:
- Rc or R d are the same or different and have good hydrogen atom, having 1 to nitro group or C shows a 6 alkoxy group.
- (10) The method for producing a thiol compound or a salt thereof described in (10).
- R a or R b may be the same or different and each represents a hydrogen atom, an acetoxy group, a nitro group or an alkoxy group having 1 to 6 carbon atoms.) ) methyl or (2-naphthyl) when a methyl group, represented by R e
- R e In the presence of one additive selected from the group consisting of a copper compound, an iron compound, a cobalt compound, a silver compound, a titanium compound, or a hydrate thereof, the elimination process of the protecting group is represented by the general formula:
- Magnesium compounds are dimethylmagnesium, jetylmagnesium, di-n-butylmagnesium, di-n-propylmagnesium, n-butylmagnesium chloride, n-butylmagnesiumpromide, methyl Magnesium chloride, Methinoremagnesium Promide, Ethenoremagnesium Chloride, Ethenolemagnesium mouthmide, n-Propyl Magnesium Chloride, n-Propyl Magnesium Promide, Isopropyl Magnesium Chloride or Isopropyl Magnesium Bromide (19) The method for producing a thiol compound or a salt thereof according to (19).
- Halogen atom means a fluorine atom, a chlorine atom, a bromine atom or an iodine atom.
- C1-C6 alkyl group means a linear or branched alkyl group having 1 to 6 carbon atoms, such as a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n— Butyl group, Isoptyl group, sec-Butyl group, tert-Putyl group Pentyl group, isopentyl group, hexyl group or isohexyl group, etc.
- C1-C10 alkyl group means a linear or branched alkyl group having 1 to 10 carbon atoms, such as a methyl group, an ethyl group, an n-propyl group, or an isopropyl group.
- N-butyl group isobutyl group, sec-butyl group, tert-butyl group, pentyl group, isopentyl group, n-hexyl group, isohexyl group, 2-ethylhexyl group, n -heptyl group, An isoheptyl group, an n-octyl group, an isooctyl group, an n-nonyl group, an isonoel group, a decyl group, and the like can be given.
- C1-C6 alkylene group means a linear or branched alkylene group having 1 to 6 carbon atoms, such as a methylene group, an ethylene group, a trimethylene group, a tetramethylene group, a pentamethylene group, Examples include a hexamethylene group, a propylene group, and an ethylethylene group.
- C1-C6 alkoxy group means a linear, branched or cyclic alkoxy group having 1 to 6 carbon atoms, such as a methoxy group, an ethoxy group, an n_propoxy group, an isopropoxy group.
- Group, n-butoxy group, isobutoxy group, sec-butoxy group, tert-butoxy group, n-pentyloxy group, isopentyloxy group, cyclopentinoreoxy group, hexinoreoxy group, isohexyloxy group or cyclohexene A xyloxy group etc. are mentioned.
- Alkoxy group having 1 to 10 carbon atoms means a linear, branched or cyclic alkoxy group having 1 to 10 carbon atoms, such as a methoxy group, an ethoxy group, and an n-propyloxy group.
- C1-C10 alkylthio group means a linear, branched or cyclic alkylthio group having 1 to 10 carbon atoms, such as methylthio group, ethylthio group, n-propylthio group, isopropylthio group.
- n-butylthio group isobutylthio group Group, sec-butylthio group, tert-butylthio group, n-pentylthio group, isopentylthio group, cyclopentylthio group, n-hexylthio group, isohexylthio group, cyclohexylthio group, n_heptylthio group, isoheptylthio group Examples thereof include n-octylthio group, isooctylthio group, n-nonylthio group, isononylthio group, n-decanylthio group, and isodecanylthio group.
- C1-C1 alkylsphinyl group means a straight-chain or branched alkylsf yl group having C1-C10, for example, methylsulfinyl group, ethylsulfier group, n-propyl sulfinyl group, isopropyl sulfinino group, n-butyl sulfininole group, isoptyl sulrefiel group, sec-butyl sulfiel group, tert-butyl sulfinyl group, n-pentyl sulfinino group, isopentyl sulfinyl group, hexyl sulfide El group to n-, hexyl sulfinyl group isohexyl, Petit Rusuru arylsulfinyl group to n primary, Petit Luz sulfide El group isohexyl, n-
- C1-C10 alkylsulfonyl group means a linear, branched or cyclic alkylsulfonyl group having 1 to 10 carbon atoms, such as a methylsulfonyl group or an ethylsulfonyl group.
- N-propyl sulfonyl group isopropyl sulfonyl group, n-butyl sulfonyl group, isobutyl sulfonyl group, sec-butyl sulphonol group, tert-butinolesno sulfonyl group, n-pentyl sulfonyl group, iso Pentylsulfonyl group, cyclopentylsulfonyl group, n-hexylsulfonyl group, isohexylsulfonyl group, cyclohexylsulfonyl group, n-heptylsulfonyl group, isoheptylsulfonyl group, n-octylsulfonyl group Group, isooctylsulfonyl group, n-nonylsulfonyl group, n
- C2-C10 alkoxycarbonyl group means a linear, branched or cyclic alkoxycarbonyl group having 1 to 10 carbon atoms, such as a methoxycarbonyl group, an ethoxycarbonyl group, n-propoxycarbonyl group, isoprobo Xyloxycarbonyl group, n-butoxycarbonyl group, isobutoxycarbonyl group, sec-butoxycananoleptinore group, tert-butoxycanoleboninole group, n-pentinoreoxycarbonyl group, isopentyloxycarbonyl group, cyclopentylo Xoxycarbonyl group, hexyloxycarbonyl group, isohexyloxycarbonyl group, cyclohexyloxycarbonyl group, n -heptyloxycarbonyl group, iso-heptyloxycarbonyl group, n-octyloxy group Examples thereof include a carbonyl group
- C2-C10 alkanoyloxy group means a C2-C10 linear, branched or cyclic alkanoyloxy group, for example, formyloxy group, acetooxy group, propionyl group.
- aryl carbonate group means an aryl carbonate group such as a benzoyl group, a 1-naphthylcarbonyl group, or a 2-naphthylcarbonyl group.
- arylcarbonyloxy group means an arylcarbonyloxy group such as a benzoyloxy group, a 1-naphthylcarbonyloxy group, or a 2-naphthylcarbonyloxy group.
- Heteroarylcarbonyl group means, for example, 2_furylcarbonyl group, 3-furylcarbonyl group, 2-phenylcarbonyl group, 3_phenylcarbonyl group, 2-pyridylcarbonyl group, 3- Pyridylcarbonyl group, 4-pyridylcarbonyl group, 2-pyrrolylcarbonyl group, 3-pyrrolylcarbonyl group, 2-imidazolylcarbonyl group, 4-imidazolylcarbonyl group, 5-imidazolylcarbonyl group, 3-virazolylcarbonyl group, 4-monopyrazolylcarbonyl group, 5-bisazolylcarbonyl group, 2-pyrimidinylcarbonyl group, 4_pyrimidinylcarbonyl group, 5-pyrimidinylcarbonyl group, 2-thiazolylcarbonyl group, 2-oxazolylcarbonyl group, 3-pyridazini group Ruoxy group, 2-birazinylcarbonyl group, 2 It means a hetero
- Heteroarylcarbonyloxy group means, for example, 2_furylcarbonyloxy group, 3-furylcarbonyloxy group, 2-phenylcarbonyloxy group, 3-phenylcarbonyloxy group, 2- Pyridylcarbonyloxy group, 3-pyridylcarbonyloxy group, 4-pyridylcarbonyloxy group, 2-pyrrolylcarbonyloxy group, 3-pyrrolylcarbonyloxy group, 2-imidazolylcarbonyloxy group, 4-imidazolylcarbonyl Oxy group, 5-Imidazolylcarbonyloxy group, 3-Pyrazolylcarbonyloxy group, 4-Pyrazolylcarbonyloxy group, 5-Virazolylcarbonyloxy group, 2-Pyrimidinylcarbonyloxy group, 4-Pyrimidinylcarbonyl Oxy group, 5_pyrimidinylcarbonyloxy group, 2-thiazolylcarbonyl Oxy group, 2-oxazolylcarbonyloxy group,
- C1-C10 alkanoyl group means a C2-C10 linear, branched or cyclic alkanoyl group such as formyl group, acetyl group, pionyl group, butyryl group. , Isobutyryl group, valeryl group, cyclyl pentylcarbonyl group, isopaleryl group, bivaloyl group, hexanol group, dichlorocarbonyl group, heptanol group, octanoyl group, nonanoyl group or decanol group.
- C3-C8 cycloalkyl group means, for example, a C3-C8 cycloalkyl group such as a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group or a cyclohexyl group. Means.
- Heteroarylcarbonylamino group means, for example, 2-furylcarbonylamino group, 3-furylcarbonylamino group, 2-phenylcarbonylamino group, 3-phenylcarbonylamino group, 2- Pyridylcarbonylamino group, 3_pyridyl Carbonylamino group, 4-monopyridylcarbonylamino group, 2-pyrrolylcarbonylaminoamino group, 3-pyrrolylcarbonylamino group, 2-imidazolylcarbonyl!
- arylcarbonylamino group examples include aryloxycarbonyl groups such as a benzoylamino group, a 1-naphthylcarbonylamino group, and a 2-naphthylcarbonylamino group.
- C1-C10 alkanoylamino group means a straight-chain or branched alkanoyl group having 2 to 10 carbon atoms, such as formylamino group, acetylamino group, propionylamino group.
- Butylylamino group isoptyrylamino group, valerylamino group, isovalerylamino group, bivalloylamino group, hexanol amiso group, heptanoylamino group, otanoylamino group, nonanoylamino group or decanoylamino group.
- aryl group means an aryl group such as a phenyl group or a naphthyl group.
- heteroaryl group is a 5-membered or 6-membered monocycle containing 1 or 2 or more, preferably 1 to 3 heteroatoms, which are the same or different from the group consisting of an oxygen atom, a nitrogen atom and a sulfur atom.
- aryl group means an “aryl group substituted with two substituents”.
- heteroaryl ring group means “a heteroaryl group substituted by two substituents”.
- C1-C6 alkanoyl group means a straight-chain or branched alkanoyl group having 1 to 6 carbon atoms, such as an acetyl group, propiol group, butyryl group, isoptyryl group, valeryl group. Group, isovaleryl group or bivaloyl group.
- aryloxy group examples include a phenoxy group, a 1-naphthyloxy group, or a 2-naphthyloxy group.
- R 2 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group or a pyridyl group.
- Y represents a sulfur atom, a sulfinyl group, a sulfonyl group, an oxygen atom, a carbonyl group, an oxycarbonyl group, a carbo-loxy group, and a general formula:
- R 2 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group or a pyridyl group.
- ⁇ 4 and ⁇ 5 are integers of 1 to 6, and the sum of both does not exceed 6.
- the “electron withdrawing group” means a group that attracts ⁇ electrons or ⁇ electrons in the molecule, such as a halogen atom, a nitro group, a carbonyl group, a carboxy group, a cyano group, or a sulfo group.
- Salt means a salt formed by a functional group and an acid or base.
- an alkali metal such as sodium, potassium or lithium
- the salt with organic amine such as triethylamine or triethylamine is an amino group
- the salt with an acid such as hydrochloric acid, sulfuric acid or nitric acid
- the salt is a thiol group
- an alkali metal such as sodium, potassium or lithium
- the salt is a thiol group, an alkali metal such as sodium, potassium or lithium And the salt.
- D b a means dibenzylidaseton (d i b e n z y l i d e n e a c e t o n e).
- Ph means a phenyl group
- I—P r means an isopropyl group.
- Me means a methyl group
- T—Bu means a ter t-butyl group.
- Cy represents a cyclopentadienyl group.
- R 1 is a hydrogen atom, a halogen atom, a cyano group, an alkyl group having 1 to 10 carbon atoms, an alkoxy group having 1 to 10 carbon atoms, an alkylthio group having 1 to 10 carbon atoms, carbon Alkylsulfinyl group having 1 to 10 carbon atoms, alkylsulfol group having 1 to 10 carbon atoms, hydroxyl group, carboxy group, alkoxycarbonyl group having 2 to 10 carbon atoms, alkanoyl having 2 to 10 carbon atoms Roxy group, aryl group, arylcarbonyl carbonyl group, arylcarbonyl group, heteroaryl group, heteroarylcarbonyl group, heteroarylcarbonyloxy group, nitro group, alkanoyl group having 1 to 10 carbon atoms A mino group, an arylcarbonylcarbonyl group, a heteroarylcarbonylamino group or an alkanol group having 1 to 10 carbon atom
- R 2 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group or a pyridyl group.
- X represents a bromine atom, a chlorine atom, a 'trifluoromethanesulfonyloxy group, a methylsulfonyloxy group, a benzenesulphoninoreoxy group, a toluenesulphoninoreoxy group or a two-necked benzenesulphonyloxy group,
- n 1 is 0 or 1
- the group represented by represents an aryl group or a heteroaryl ring group.
- X is a chlorine atom
- R 1 n 1 and Y 1 have the above-mentioned meanings.
- a palladium compound selected from the group consisting of palladium acetate, P d 2 (dba) 3 and P d (dba) 2 ,
- R 3 , R 4 and R 5 may be the same or different and each represents an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group or a pyridyl group.
- 1,5-Diazabicyclo [4. 3. 0] Nona 5-ene and 1,8-Diazabicyclo [5. 4. 0] Wunde force 7-en
- a base selected from: and a formula:
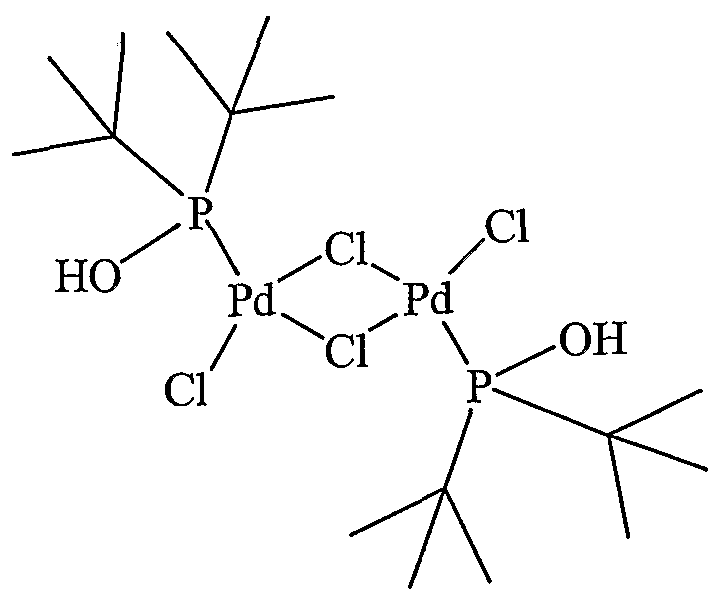
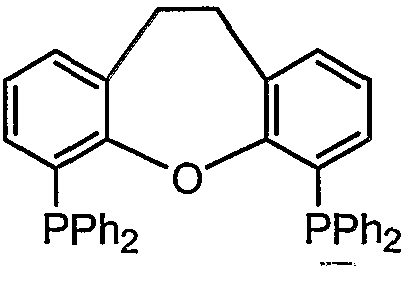
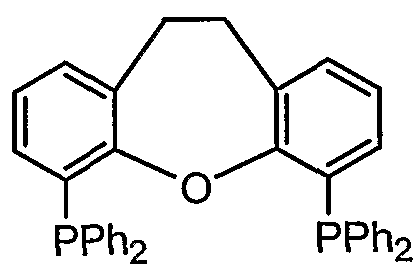
- a compound represented by Fe (alias: DPPF; [1, 1, monobis (diphenylphosphino) pheocene)] [1, 1 '— B is (diphe ny lphosphino) ferroce ri 6]), formula :
- ⁇ -P (i-Pr) 2 compound also known as: 1, 1, monobis (diisopyruphosphino) Huekousen [1, 1, mono B is ⁇ diisopropylphosphino) ierrocene]
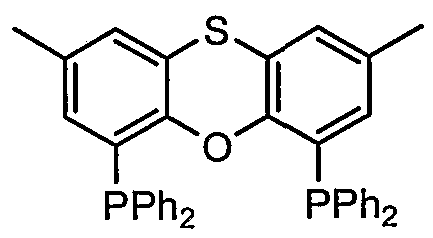
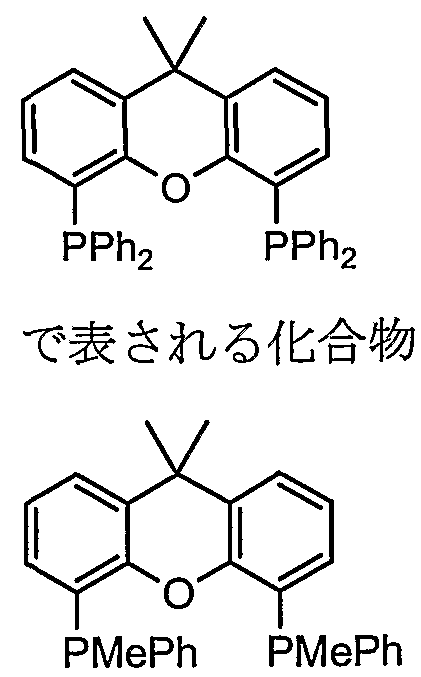
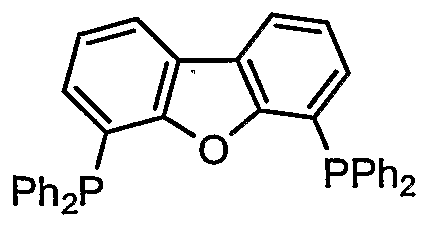
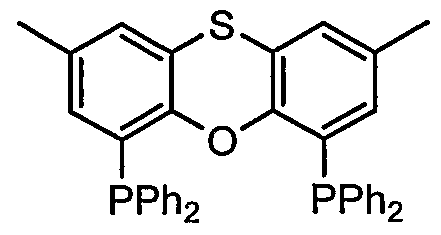
- a compound represented by also known as Th i a x a n t p h o s
- R 6 or R 7 may be the same or different and each represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, an amino group or a phenyl group, n 2 represents 0 to 6,
- R 8 Is a hydrogen atom, a halogen atom, a cyan group, an amino group, an alkyl group having 1 to 10 carbon atoms, a trimethylsilyl group, an alkoxy group having 1 to 10 carbon atoms, an alkylthio group having 1 to 10 carbon atoms, or an alkyl group having 1 to 10 carbon atoms.
- Y 2 represents a sulfur atom, a sulfonyl group, a sulfonyl group, an oxygen atom, a carbonyl group, an oxycarbonyl group, a carbonyloxy group, and a general formula:
- R 7 represents a hydrogen atom, an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group, a naphthyl group, or a pyridyl group.
- ⁇ 3 is 0 or 1
- R 9 is a hydrogen atom, a halogen atom, a cyano group, an amino group, a nitro group, an alkyl group having 1 to 10 carbon atoms, an alkoxy group having 1 to 10 carbon atoms, an alkylthio group having 1 to 10 carbon atoms, An alkylsphinyl group having 1 to 10 carbon atoms, an alkylsulfonyl group having 1 to 10 carbon atoms, a hydroxyl group, a carboxy group, an alkoxycarbonyl group having 2 to 10 carbon atoms, an alkanoylo having 2 to 10 carbon atoms Xyl group, aryl group, arylcarbonyl group, arylcarbonyl group, heteroaryl group, heteroarylcarbonyl group, heteroarylcarbonyloxy group, double-terminal group, alkanoyl group having 1 to 10 carbon atoms A mino group, an arylcarbonylcarbonyl group, a heteroarylcarbony
- the group represented by represents an aryl group or a heteroaryl ring group. Is reacted with a thiol compound represented by
- the group represented by has the above-mentioned meaning.
- the process for producing a thioether compound or a salt thereof represented by the formula: Add 005 equivalents to 0.1 equivalents of phosphorus compound, 0.005 equivalents to 0.1 equivalents of palladium compound, and 1.5 equivalents to 2 equivalents of base, respectively, at 50 ° C or 100 ° C.
- the reaction can be carried out by reacting for 2 to 15 hours.
- the solvent that does not adversely influence the reaction include dioxane, toluene, 2-methyltetrahydrofuran, tetrahydrofuran, N, N-dimethylformamide, dimethylimidazolidinone, N-methylpyrrolidone, dimethyl ether, and diethyl ether. .
- Bases include cesium carbonate, general formula:
- R 3 , R 4 and R 5 may be the same or different and each represents an alkyl group having 1 to 6 carbon atoms, a benzyl group, a phenyl group or a pyridyl group.
- 1,5-Diazabicyclo [4. 3. 0] Nona 1-5- and 1,8-Diazabicyclo [5. 4. 0] Unde force 7- It is done.
- R 3, 1 4 and 1 5, which may be the same or different, showing 1 -C 6 alkyl group, a benzyl group, a phenyl group or a pyridyl group.
- amine derivative represented by the following are tertiary amines such as disopropylethylamine, triptylamamine, triethylamine, trimethylamine, dibenzylmethylamine, 4-dimethylaminopyridine, and tribenzylamine. Can be mentioned.
- the group represented by ⁇ has the above-mentioned meaning.
- the method for producing the thiol compound represented by the following formula or the salt thereof is based on the method for removing the substituent (protective group) ligated to the thiol group disclosed in the following known literature and the method for removing the same. Can be implemented. Alan R. katritz ky et al., Tetrahedron Letters ⁇ 25 12 No. 1223-1226 (1 984)
- R a or R b may be the same or different and each represents a hydrogen atom, an acetoxy group, a nitro group or an alkoxy group having 1 to 6 carbon atoms), (1-naphthyl) ) When it is a methyl group or (2-naphthyl) methyl group,
- R e a protecting group represented by R e has the general formula:
- R d may be the same or different, a hydrogen atom, 1 to several nitro group or C to indicate the 6 alkoxy group.
- R e a group represented by is represented in the R e
- 1.5 liters of potassium alkoxide or sodium alkoxide is used per 1 equivalent of the raw material in a solvent that does not adversely affect the reaction.
- ⁇ 5 equivalents preferably 2-3 equivalents, and can be carried out by reacting at 110 ° C to 120 ° C for 1 hour to 24 hours, preferably 2 hours to 6 hours. it can.
- solvents that do not adversely affect the reaction include diglyme, .triglyme, tetraglyme, dimethyl sulfoxide, 1,3-dimethyl-2-imidazolidinone, N-methinorepyrrolidone, cyclopentyl pentyl methyl ether 1,2-dimethoxetane, N, N-dimethylformamide or N, N-dimethylacetamide, preferably diglyme or N, N-dimethylacetamide.
- 1 £ : or 1 £ 1 may be the same or different and each represents a hydrogen atom, a nitro group, or an alkoxy group having 1 to 6 carbon atoms.
- R e has the general formula:
- R a or R b may be the same or different and each represents a hydrogen atom, an acetoxy group, a nitro group or an alkoxy group having 1 to 6 carbon atoms), (1-naphthyl) ) when it is methyl or (2-naphthyl) methyl group, elimination step of protecting groups that you express in the R e is, copper compounds, iron compounds, cobalt compounds, the addition of the group consisting of silver compound and titanium compound
- one additive selected from the general formula: wherein R g represents a halogen atom or an alkyl group having 1 to 10 carbon atoms, and R h represents an alkyl group having 1 to 10 carbon atoms.
- the additive is added in an amount of 0.05 to 1 equivalent, preferably 0.05 to 0.5 equivalent, relative to 1 mol of the raw material.
- R a or R b may be the same or different and each represents a hydrogen atom, an acetoxy group, a double-headed group, or an alkoxy group having 1 to 6 carbon atoms).
- examples thereof include a benzyl group, a 4-12 benzyl group, a 4-methoxybenzyl group, a 2,4 1-dibenzyl group or a 3,4-dimethoxybenzyl group, and a benzyl group is preferred.
- Examples of the “solvent that does not adversely influence the reaction” used in this step include diglyme, triglyme, tetraglyme, ethyl ether, dioxane, methyltetrahydrofuran, tetrahydrofuran or methyl t-butyl ether, preferably diglyme. is there.
- additives used in this process include CuC 1 2 , CuC 1 2 ⁇ 2H 2 O, F e C l 3 , F e C l 2 , T i C 1 2 ( ⁇ P r O) 2 , Cu (CF 3 SO 2 0) 2 , CoCl 2 , AgN 0 3 or C p 2 Ti C 1 2 (wherein Cp represents a cyclopentadienyl group), preferably CuC 1 2, a CuC l 2 '2H 2 O or C p 2 T i C 1 2 .
- Magnetic compound force such as dimethyl magnesium is used in this step, Jefferies chill magnetic Information & Technology arm, di - n - butyl magnetic Information & Technology arm, di one n- propyl magnesiate ⁇ beam, n- Petit Honoré magnesium chloride Li de, n- Petit Nore magnesium promide, me Chinore Magnesium Chloride, Methinole Magnesium Promide, Ethinore Magnesium Chloride, Ethinore Magne Magnesium Bromide, n-Propinole Magnesium Chloride), n-Propylmagnesium Bromine , Isopropylmagnesium chloride or isopropylmagnesium bromide, preferably di-n-butylmagnesium, n_butinoremagnesium chloride or n-petitenolemagnesium bromide. It's a lomid.
- the product obtained in the above steps is obtained by a method known per se, such as column chromatography using silica gel or an adsorbent resin, liquid chromatography, thin layer chromatography, solvent extraction or recrystallization / reprecipitation. Purification and isolation can be carried out by using conventional separation and purification methods alone or in combination as appropriate.
- the raw material of the manufacturing method of this invention can utilize what was manufactured by the commercial item or the well-known manufacturing method.
- P d 2 (dba) 3 was purchased from Johnson & Matthey.
- X antphos was purchased from Aldrich (A 1 drich).
- Anhydrous K 3 P0 4 , anhydrous K 2 C0 3 , anhydrous Na 2 CO s , and anhydrous C s 2 C 0 3 were purchased from Wako Pure Chemical Industries.
- Other reagents used in this example were purchased from Tokyo Kasei and Strem.
- Thiols, aryl halides, and aryl trifluorosulfonate organic solvents were purchased from Tokyo Kasei, dried over molesieves 4 A, degassed and used.
- High performance liquid chromatography is a high performance liquid chromatogram made by Hitachi D—7000
- NMR data are measured using a Bruker AV-500.
- Examples 1 to 27 Compounds according to Examples 1 to 27 were prepared according to the following basic operation method.
- Aryl sulfonate and its usage 4-trinole trifluoromethanesulphonate (3 58 ⁇ L, 2 mm ⁇ 1)
- the target zeolite compound (C) cannot be obtained even if it is subjected to a known Suzuki-Miyaura reaction using the furobromide which is the starting compound of the present invention. Even if it was obtained, the yield was found to be inappropriate for an industrial production process.
- D_ t— BPF 1, 1 'One bis (di-tert-butylphosphino) Hue port sen (1, 1 1 b i s (d i — t e r t — b u t y l p h o s p h i n o) I e r r o c e n e)
- benzyl phenylthioether (92. 5mg, 0. 462 mmo 1) and copper (II) chloride dihydrate (7.9 mg, 0.0463 mm o 1) were dissolved in diglyme (1 in L) and dibutylmagnesium 1.0 M hexane solution (. 16mL, 1.16mmo 1) was added. After heating to 50 ° C and stirring for 5 hours, analysis by high performance liquid chromatography revealed that the target thiophenol (34.6 mg, yield: 68%), and diphenyldisulfide (15.1 mg, yield) (Rate: 30%). The recovery rate of the raw material benzylphenylthioether was 2% (1.9 mg).
- Table 1 shows that the benzyl group can be removed by using additives.
- T f represents a trifluoromethyl titanium sulfonyl group.
- C p represents a cyclopentagenino group.
- potassium tert-butoxide is industrially useful for elimination of the phenethyl group from the yield of the target thiophenol (a).
- DMS O represents dimethyl sulfoxide.
- DMA N, N indicates dimethylacetamide.
- A: B 50: 50 (0 minute), 50:50 (5 minutes),
- the method for producing a thioether compound according to the present invention uses a bromide, chloride or sulfonate compound that is easy to secure as a raw material, and a target thioether compound in a Suzuki-Miyaura reaction under weak basic conditions. It is characterized by high yield.
- a thioether compound can be produced industrially and efficiently at low cost for chemicals and pharmaceuticals that could not be produced by the conventional Su z uk i -Miyaura reaction. be able to.
- a thiol ether compound can be efficiently produced using a thiol compound having a removable substituent, and a known thiol group protecting group removal method or the present inventors have found If the elimination method of the benzyl group or phenethyl group is used as the protecting group, the introduction of the thiol group into the aryl ring or the heteroaryl ring can be made efficient, which is useful in the field of organic synthesis.
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/664,663 US7683218B2 (en) | 2004-10-08 | 2005-10-07 | Method for producing thioether compound |
| EP05793261A EP1806337A4 (en) | 2004-10-08 | 2005-10-07 | METHOD FOR THE SYNTHESIS OF A THIOETHER COMPOUND |
| AU2005290413A AU2005290413A1 (en) | 2004-10-08 | 2005-10-07 | Method for producing thioether compound |
| CA002582658A CA2582658A1 (en) | 2004-10-08 | 2005-10-07 | Method for producing thioether compound |
| JP2006539361A JPWO2006038741A1 (ja) | 2004-10-08 | 2005-10-07 | チオエーテル化合物の製造方法 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2004295958 | 2004-10-08 | ||
| JP2004-295958 | 2004-10-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006038741A1 true WO2006038741A1 (ja) | 2006-04-13 |
Family
ID=36142795
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2005/018985 WO2006038741A1 (ja) | 2004-10-08 | 2005-10-07 | チオエーテル化合物の製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US7683218B2 (ja) |
| EP (1) | EP1806337A4 (ja) |
| JP (1) | JPWO2006038741A1 (ja) |
| CN (1) | CN101068777A (ja) |
| AU (1) | AU2005290413A1 (ja) |
| CA (1) | CA2582658A1 (ja) |
| WO (1) | WO2006038741A1 (ja) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7534791B2 (en) | 2005-06-17 | 2009-05-19 | H. Lundbeck A/S | Benzo[b]furane and benzo[b]thiophene derivatives |
| US7563908B2 (en) | 2003-12-23 | 2009-07-21 | Jan Kehler | 2-(1H-indolylsulfanyl)-benzyl amine derivatives as SSRI |
| US7629473B2 (en) | 2005-06-17 | 2009-12-08 | H. Lundbeck A/S | 2-(1H-indolylsulfanyl)-aryl amine derivatives |
| WO2013161853A1 (ja) | 2012-04-24 | 2013-10-31 | 中外製薬株式会社 | キナゾリンジオン誘導体 |
| WO2013161851A1 (ja) | 2012-04-24 | 2013-10-31 | 中外製薬株式会社 | ベンズアミド誘導体 |
| WO2015060373A1 (ja) | 2013-10-23 | 2015-04-30 | 中外製薬株式会社 | キナゾリノンおよびイソキノリノン誘導体 |
| CN105218418A (zh) * | 2015-10-01 | 2016-01-06 | 宜春学院 | 一种硫醚的制备方法 |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE517887T1 (de) * | 2003-02-13 | 2011-08-15 | Msd Kk | Neue 2-pyridincarbonsäureamidderivate |
| CN103880720B (zh) * | 2014-04-10 | 2015-10-14 | 顾祥茂 | 一种芳基硫醚化合物的合成方法 |
| CN109843289B (zh) * | 2017-10-09 | 2021-10-29 | 华南农业大学 | 一种抗白色念珠菌的二芳基硫族化合物及其制备和应用 |
| GB201919213D0 (en) * | 2019-12-23 | 2020-02-05 | Ucb Biopharma Sprl | Dihydrocyclopenta-Isoquinoline-Sulfanamide derivatives compounds |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002047278A (ja) | 2000-08-02 | 2002-02-12 | Fuji Photo Film Co Ltd | チオエーテル化合物の製造方法 |
| AU2002952839A0 (en) * | 2002-11-21 | 2002-12-05 | Fujisawa Pharmaceutical Co., Ltd. | Aminoalcohol derivatives |
| ATE517887T1 (de) | 2003-02-13 | 2011-08-15 | Msd Kk | Neue 2-pyridincarbonsäureamidderivate |
-
2005
- 2005-10-07 AU AU2005290413A patent/AU2005290413A1/en not_active Abandoned
- 2005-10-07 CA CA002582658A patent/CA2582658A1/en not_active Abandoned
- 2005-10-07 JP JP2006539361A patent/JPWO2006038741A1/ja not_active Withdrawn
- 2005-10-07 WO PCT/JP2005/018985 patent/WO2006038741A1/ja active Application Filing
- 2005-10-07 US US11/664,663 patent/US7683218B2/en not_active Expired - Fee Related
- 2005-10-07 EP EP05793261A patent/EP1806337A4/en not_active Withdrawn
- 2005-10-07 CN CNA2005800415579A patent/CN101068777A/zh active Pending
Non-Patent Citations (3)
| Title |
|---|
| OGAWA A. ET AL: "Highly regio- and stereocontrolled synthesis of vinyl sulfides via transition-metal-catalyzed hydrothiolation of alkynes with thiols", JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 121, no. 22, 9 June 1999 (1999-06-09), pages 5108 - 5114, XP002995008 * |
| SCHOPFER U. AND SCHLAPBACH A.: "A general palladium-catalyzed synthesis of aromatic and heteroaromatic thioethers", TETRAHEDRON, vol. 57, no. 15, 9 April 2001 (2001-04-09), pages 3069 - 3073, XP004235135 * |
| See also references of EP1806337A4 * |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7563908B2 (en) | 2003-12-23 | 2009-07-21 | Jan Kehler | 2-(1H-indolylsulfanyl)-benzyl amine derivatives as SSRI |
| US7652150B2 (en) | 2003-12-23 | 2010-01-26 | H. Lundbeck A/S | 2-(1H-indolylsulfanyl)-benzyl amine derivatives as SSRIs |
| US7737170B2 (en) | 2003-12-23 | 2010-06-15 | H. Lundbeck A/S | Uses of 2-(1H-indolylsulfanyl)-benzyl amine derivatives as SSRIS |
| US7737171B2 (en) | 2003-12-23 | 2010-06-15 | H. Lundbeck A/S | Uses of 2-(1H-indolylsulfanyl)-benzyl amine derivatives as SSRIS |
| US7534791B2 (en) | 2005-06-17 | 2009-05-19 | H. Lundbeck A/S | Benzo[b]furane and benzo[b]thiophene derivatives |
| US7629473B2 (en) | 2005-06-17 | 2009-12-08 | H. Lundbeck A/S | 2-(1H-indolylsulfanyl)-aryl amine derivatives |
| WO2013161853A1 (ja) | 2012-04-24 | 2013-10-31 | 中外製薬株式会社 | キナゾリンジオン誘導体 |
| WO2013161851A1 (ja) | 2012-04-24 | 2013-10-31 | 中外製薬株式会社 | ベンズアミド誘導体 |
| WO2015060373A1 (ja) | 2013-10-23 | 2015-04-30 | 中外製薬株式会社 | キナゾリノンおよびイソキノリノン誘導体 |
| CN105218418A (zh) * | 2015-10-01 | 2016-01-06 | 宜春学院 | 一种硫醚的制备方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101068777A (zh) | 2007-11-07 |
| EP1806337A1 (en) | 2007-07-11 |
| AU2005290413A1 (en) | 2006-04-13 |
| CA2582658A1 (en) | 2006-04-13 |
| US7683218B2 (en) | 2010-03-23 |
| EP1806337A4 (en) | 2009-11-25 |
| JPWO2006038741A1 (ja) | 2008-05-15 |
| US20080108823A1 (en) | 2008-05-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2006038741A1 (ja) | チオエーテル化合物の製造方法 | |
| CN106061972B (zh) | 5-氟-4-亚氨基-3-(烷基/取代烷基)-1-(芳基磺酰基)-3,4-二氢嘧啶-2(1h)-酮及其制备方法 | |
| WO2007102499A1 (ja) | 4-オキソキノリン化合物の製造方法 | |
| WO2006104088A1 (ja) | 1-(3-(2-(1-ベンゾチオフェン-5-イル)エトキシ)プロピル)アゼチジン-3-オールまたはその塩の製造法 | |
| JP4828863B2 (ja) | (z)−1−フェニル−1−(n,n−ジエチルアミノカルボニル)−2−フタルイミドメチルシクロプロパンの製造方法 | |
| KR101564401B1 (ko) | 브린졸아마이드의 제조방법 | |
| CN102264744A (zh) | 碳青霉烯侧链中间体的改良制造方法 | |
| JP2006348034A (ja) | Mek阻害剤の製造方法 | |
| JP2008056615A (ja) | ビニルエチニルアリールカルボン酸類、その製造方法及びそれを用いた熱架橋性化合物の製造方法 | |
| CN103664959A (zh) | 一种五元双环胍类化合物的制备方法 | |
| WO2016066116A1 (zh) | 阿普斯特的制备方法及其中间体 | |
| CN104507949A (zh) | 高纯度的含氮杂环化合物的制造方法 | |
| KR20160027536A (ko) | 실로도신 합성에 유용한 중간체의 제조방법 | |
| US7728136B2 (en) | Method for the preparation of aryl piperazinyl-heterocyclic compounds | |
| WO2016121959A1 (ja) | イソインドリン化合物またはその塩の製造方法および新規イソインドリン化合物またはその塩 | |
| TWI404711B (zh) | 2-烯基-3-胺基噻吩衍生物及其製造方法 | |
| CN108473431B (zh) | 2-氨基烟酸苄酯衍生物的制造方法 | |
| JP3276523B2 (ja) | ジグアナミン類の製造方法 | |
| JP2000256306A (ja) | 芳香族チオール類の製造法 | |
| CN101300247A (zh) | 3-羟甲基苯并[b]噻吩衍生物及其制备方法 | |
| WO2014108011A1 (zh) | 恩替卡韦中间体及其制备方法 | |
| WO2006019146A1 (en) | Method for producing oxonol compound | |
| KR900001911B1 (ko) | 2-비닐-4, 6-디아미노-s-트리아진의 합성방법 | |
| KR20090082463A (ko) | N-[3-[(2-메톡시페닐)설파닐]-2-메틸프로필]-3,4-디하이드로-2h-1,5-벤즈옥사티에핀-3-아민 제조 방법 | |
| KR20190016208A (ko) | 신규의 설폰아마이드 중간체 및 이를 사용한 실로도신의 제조방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KM KP KR KZ LC LK LR LS LT LU LV LY MA MD MG MK MN MW MX MZ NA NG NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU LV MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2006539361 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2582658 Country of ref document: CA Ref document number: 11664663 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005290413 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2967/DELNP/2007 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005793261 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580041557.9 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005793261 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 11664663 Country of ref document: US |