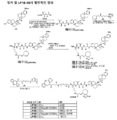
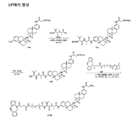
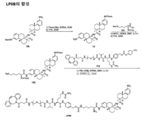
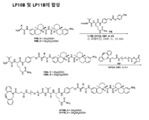
KR20210008008A - 항-msr1 항체 및 이의 사용 방법 - Google Patents
항-msr1 항체 및 이의 사용 방법 Download PDFInfo
- Publication number
- KR20210008008A KR20210008008A KR1020207034456A KR20207034456A KR20210008008A KR 20210008008 A KR20210008008 A KR 20210008008A KR 1020207034456 A KR1020207034456 A KR 1020207034456A KR 20207034456 A KR20207034456 A KR 20207034456A KR 20210008008 A KR20210008008 A KR 20210008008A
- Authority
- KR
- South Korea
- Prior art keywords
- antibody
- formula
- alkyl
- msr1
- antigen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2896—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against molecules with a "CD"-designation, not provided for elsewhere
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
- A61K31/167—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide having the nitrogen of a carboxamide group directly attached to the aromatic ring, e.g. lidocaine, paracetamol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/4164—1,3-Diazoles
- A61K31/417—Imidazole-alkylamines, e.g. histamine, phentolamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4985—Pyrazines or piperazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5383—1,4-Oxazines, e.g. morpholine ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
- A61K31/58—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids containing heterocyclic rings, e.g. danazol, stanozolol, pancuronium or digitogenin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/14—Peptides containing saccharide radicals; Derivatives thereof, e.g. bleomycin, phleomycin, muramylpeptides or vancomycin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/3955—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against proteinaceous materials, e.g. enzymes, hormones, lymphokines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/61—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule the organic macromolecular compound being a polysaccharide or a derivative thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/6807—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug or compound being a sugar, nucleoside, nucleotide, nucleic acid, e.g. RNA antisense
- A61K47/6809—Antibiotics, e.g. antitumor antibiotics anthracyclins, adriamycin, doxorubicin or daunomycin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6889—Conjugates wherein the antibody being the modifying agent and wherein the linker, binder or spacer confers particular properties to the conjugates, e.g. peptidic enzyme-labile linkers or acid-labile linkers, providing for an acid-labile immuno conjugate wherein the drug may be released from its antibody conjugated part in an acidic, e.g. tumoural or environment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/56—Immunoglobulins specific features characterized by immunoglobulin fragments variable (Fv) region, i.e. VH and/or VL
- C07K2317/565—Complementarity determining region [CDR]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/77—Internalization into the cell
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Cardiology (AREA)
- Rheumatology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Vascular Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Urology & Nephrology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Cell Biology (AREA)
- Physical Education & Sports Medicine (AREA)
- Gastroenterology & Hepatology (AREA)
- Pain & Pain Management (AREA)
- Endocrinology (AREA)
Applications Claiming Priority (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862669276P | 2018-05-09 | 2018-05-09 | |
| US62/669,276 | 2018-05-09 | ||
| US201862678200P | 2018-05-30 | 2018-05-30 | |
| US62/678,200 | 2018-05-30 | ||
| US201862769946P | 2018-11-20 | 2018-11-20 | |
| US62/769,946 | 2018-11-20 | ||
| US201962789987P | 2019-01-08 | 2019-01-08 | |
| US62/789,987 | 2019-01-08 | ||
| US201962821362P | 2019-03-20 | 2019-03-20 | |
| US62/821,362 | 2019-03-20 | ||
| PCT/US2019/031383 WO2019217591A1 (en) | 2018-05-09 | 2019-05-08 | Anti-msr1 antibodies and methods of use thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20210008008A true KR20210008008A (ko) | 2021-01-20 |
Family
ID=66625372
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207034456A Ceased KR20210008008A (ko) | 2018-05-09 | 2019-05-08 | 항-msr1 항체 및 이의 사용 방법 |
Country Status (15)
| Country | Link |
|---|---|
| US (2) | US11377502B2 (enExample) |
| EP (1) | EP3790899A1 (enExample) |
| JP (2) | JP7590871B2 (enExample) |
| KR (1) | KR20210008008A (enExample) |
| CN (2) | CN118955710A (enExample) |
| AU (1) | AU2019265703A1 (enExample) |
| BR (1) | BR112020022400A2 (enExample) |
| CA (1) | CA3098453A1 (enExample) |
| CL (1) | CL2020002873A1 (enExample) |
| CO (1) | CO2020015084A2 (enExample) |
| MX (1) | MX2020011546A (enExample) |
| MY (1) | MY209590A (enExample) |
| PH (1) | PH12020551865A1 (enExample) |
| SG (1) | SG11202010909RA (enExample) |
| WO (1) | WO2019217591A1 (enExample) |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MY199937A (en) | 2016-11-08 | 2023-11-29 | Regeneron Pharma | Steroids and protein-conjugates thereof |
| WO2018213082A1 (en) | 2017-05-18 | 2018-11-22 | Regeneron Pharmaceuticals, Inc. | Bis-octahydrophenanthrene carboxamides and protein conjugates thereof |
| KR20200007905A (ko) * | 2017-05-18 | 2020-01-22 | 리제너론 파마슈티칼스 인코포레이티드 | 사이클로덱스트린 단백질 약물 접합체 |
| CN111601619A (zh) | 2017-11-07 | 2020-08-28 | 里珍纳龙药品有限公司 | 用于抗体药物偶联物的亲水性连接体 |
| US12070506B2 (en) | 2018-01-08 | 2024-08-27 | Regeneron Pharmaceuticals, Inc. | Steroids and antibody-conjugates thereof |
| AU2019265703A1 (en) | 2018-05-09 | 2020-11-19 | Regeneron Pharmaceuticals, Inc. | Anti-MSR1 antibodies and methods of use thereof |
| CA3120528A1 (en) | 2018-11-20 | 2020-05-28 | Regeneron Pharmaceuticals, Inc. | Bis-octahydrophenanthrene carboxamide derivatives and protein conjugates thereof for use as lxr agonists |
| SG11202106658XA (en) * | 2018-12-21 | 2021-07-29 | Regeneron Pharma | Rifamycin analogs and antibody-drug conjugates thereof |
| JP2022517767A (ja) | 2019-01-08 | 2022-03-10 | レゲネロン ファーマシューティカルス,インコーポレーテッド | 無痕跡型リンカー及びそのタンパク質-コンジュゲート |
| US20230220065A1 (en) * | 2020-06-16 | 2023-07-13 | Academia Sinica | Antibodies to interleukin-1beta and uses thereof |
| BR112023000489A2 (pt) * | 2020-07-13 | 2023-03-28 | Regeneron Pharma | Análogos de camptotecina conjugados a um resíduo de glutamina em uma proteína, e seu uso |
| CN114605367B (zh) * | 2020-12-03 | 2023-06-27 | 中国人民解放军军事科学院军事医学研究院 | 含香豆素的连接子及含该连接子的抗体偶联药物 |
| US12036286B2 (en) | 2021-03-18 | 2024-07-16 | Seagen Inc. | Selective drug release from internalized conjugates of biologically active compounds |
| EP4313060B1 (en) * | 2021-03-26 | 2025-03-05 | Regeneron Pharmaceuticals, Inc. | Rifamycin analogs in combination with vancomycin and uses thereof |
| US11806405B1 (en) | 2021-07-19 | 2023-11-07 | Zeno Management, Inc. | Immunoconjugates and methods |
| CN115671309B (zh) * | 2021-07-30 | 2025-05-09 | 北京键凯科技股份有限公司 | 一种抗体-siRNA药物偶联物 |
| CN115671308B (zh) * | 2021-07-30 | 2025-11-14 | 北京键凯科技股份有限公司 | 一种靶向性的抗体-聚乙二醇-siRNA药物偶联物 |
| CN115594766A (zh) * | 2021-12-30 | 2023-01-13 | 辽宁键凯科技有限公司(Cn) | 一种缀合物及用其制备的抗体偶联药物 |
| US20230287138A1 (en) * | 2022-01-12 | 2023-09-14 | Regneron Pharmaceuticals, Inc. | Protein-drug conjugates comprising camptothecin analogs and methods of use thereof |
| WO2023173121A1 (en) | 2022-03-11 | 2023-09-14 | Firefly Bio, Inc. | Phenyl maleimide linker agents |
| EP4587030A2 (en) * | 2022-09-15 | 2025-07-23 | Adcentrx Therapeutics Inc. | Novel glucocorticoid receptor agonists and immunoconjugates thereof |
| CN121001751A (zh) | 2023-02-09 | 2025-11-21 | 里珍纳龙药品有限公司 | 借助逆电子需求狄尔斯-阿尔德反应的抗体-药物缀合物 |
| TW202519268A (zh) * | 2023-09-11 | 2025-05-16 | 美商卡蒂那生物科學股份有限公司 | 多肽結合組合物及使用方法 |
| WO2025228390A1 (en) * | 2024-04-30 | 2025-11-06 | Hutchmed Limited | A hydrophilic linker, antibody drug conjugate and use thereof |
| CN120399078A (zh) * | 2025-05-08 | 2025-08-01 | 上海天泽云泰生物医药有限公司 | 靶向cd117的纳米抗体及其应用 |
Family Cites Families (151)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2920999A (en) | 1957-06-07 | 1960-01-12 | Pfizer & Co C | 21-nitrogen derivatives of corticosteroids |
| GB889766A (en) | 1958-03-04 | 1962-02-21 | Olin Mathieson | Improvements in steroids and the synthesis thereof |
| DE1165595B (de) | 1958-03-04 | 1964-03-19 | Olin Mathieson | Verfahren zur Herstellung von Triamcinolonacetonid-21-hemisuccinat und dessen Salzen |
| US3197469A (en) | 1958-08-06 | 1965-07-27 | Pharmaceutical Res Products In | 16, 17-acetals and ketals of 6-halo-16, 17-dihydroxy steroids of the pregnane seriesand intermediates therefor |
| US3007923A (en) | 1959-01-22 | 1961-11-07 | Lab Francais Chimiotherapie | Process for the fluorination of 9beta, 11beta-epoxy and 5alpha, 6alpha-epoxy steroids |
| GB898291A (en) | 1959-03-18 | 1962-06-06 | Upjohn Co | Improvements in or relating to steroids and the manufacture thereof |
| US3033874A (en) | 1960-05-11 | 1962-05-08 | Pfizer & Co C | 17beta-propenoyl steroids and process for preparing same |
| US3033873A (en) | 1960-05-11 | 1962-05-08 | Pfizer & Co C | 21-aminomethyl-pregnenes |
| US3020275A (en) | 1960-06-07 | 1962-02-06 | American Cyanamid Co | 21-nitrogen substituted steroids of the pregnane series and methods of preparing the same |
| DE1169444B (de) | 1961-02-22 | 1964-05-06 | Schering Ag | Verfahren zur Herstellung von ?-16ª-Methylsteroiden |
| US3047468A (en) | 1961-06-06 | 1962-07-31 | American Cyanamid Co | Method of preparing delta1, 4-3-keto steroids of the pregnane series |
| US3383394A (en) | 1965-08-30 | 1968-05-14 | Schering Corp | Novel 17-acylating process and products thereof |
| US3798216A (en) | 1969-03-07 | 1974-03-19 | Fabrication Des Antibiotique S | 9-fluoro-11beta,21-dihydroxy-16alpha,17-(2-propenylidenedioxy)-pregna-1,4-diene-3,20-dione and derivatives thereof |
| DE2047105C3 (de) | 1970-09-18 | 1979-02-15 | Schering Ag, 1000 Berlin Und 4619 Bergkamen | 17-Chlorsteroide und Verfahren zu ihrer Herstellung |
| IT1057859B (it) | 1972-04-28 | 1982-03-30 | Sigma Tao Ind Farmaceutiche Sp | Perivato del triamoinolone |
| SE378110B (enExample) | 1972-05-19 | 1975-08-18 | Bofors Ab | |
| SE378109B (enExample) | 1972-05-19 | 1975-08-18 | Bofors Ab | |
| US4076737A (en) | 1975-02-20 | 1978-02-28 | Ciba-Geigy Corporation | Aldehydes of the pregnane series and derivatives thereof |
| IL73337A (en) | 1984-10-28 | 1988-09-30 | Yissum Res Dev Co | Antitumor steroid-platinum complexes and method for the preparation thereof |
| SE8604059D0 (sv) | 1986-09-25 | 1986-09-25 | Astra Pharma Prod | A method of controlling the epimeric distribution in the preparation of 16,17-acetals of pregnane derivatives |
| IL85035A0 (en) | 1987-01-08 | 1988-06-30 | Int Genetic Eng | Polynucleotide molecule,a chimeric antibody with specificity for human b cell surface antigen,a process for the preparation and methods utilizing the same |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| US5116829A (en) | 1990-04-23 | 1992-05-26 | Kao Corporation | Steroid compounds |
| US5183815A (en) | 1991-01-22 | 1993-02-02 | Merck & Co., Inc. | Bone acting agents |
| EP0940468A1 (en) | 1991-06-14 | 1999-09-08 | Genentech, Inc. | Humanized antibody variable domain |
| DK0678097T3 (da) | 1993-01-08 | 2000-02-07 | Astra Ab | Hidtil ukendte colon- eller ileumspecifikke steroidderivater |
| DE4311987A1 (de) | 1993-04-07 | 1994-10-13 | Schering Ag | Neue Glucocorticoide |
| US5714586A (en) | 1995-06-07 | 1998-02-03 | American Cyanamid Company | Methods for the preparation of monomeric calicheamicin derivative/carrier conjugates |
| US5824669A (en) | 1996-03-22 | 1998-10-20 | Nitromed, Inc. | Nitrosated and nitrosylated compounds and compositions and their use for treating respiratory disorders |
| US5837698A (en) | 1996-05-02 | 1998-11-17 | G. D. Searle & Co. | Steroid nitrite and nitrate ester derivatives useful as anti-inflammatory drugs |
| AU3703900A (en) | 1999-02-24 | 2000-09-14 | Nitromed, Inc. | Nitrosated and nitrosylated steroids for the treatment of cardiovascular diseases and disorders |
| US6596541B2 (en) | 2000-10-31 | 2003-07-22 | Regeneron Pharmaceuticals, Inc. | Methods of modifying eukaryotic cells |
| US20070258987A1 (en) | 2000-11-28 | 2007-11-08 | Seattle Genetics, Inc. | Recombinant Anti-Cd30 Antibodies and Uses Thereof |
| SE0101220D0 (sv) | 2001-04-04 | 2001-04-04 | Astrazeneca Ab | New use |
| US6908934B2 (en) | 2001-06-11 | 2005-06-21 | Merck & Co., Inc. | Therapeutic compounds for treating dyslipidemic conditions |
| ITMI20020148A1 (it) | 2002-01-29 | 2003-07-29 | Nicox Sa | Nuovi corticosteroidi |
| US20050009798A1 (en) | 2002-02-20 | 2005-01-13 | Sepracor Inc. | Carbonate and carbamate modified forms of glucocorticoids in combination with B2 adrenergic agonists |
| US7190468B2 (en) | 2002-07-17 | 2007-03-13 | Hewlett-Packard Development Company, L.P. | Background document rendering system and method |
| SI2357006T1 (sl) | 2002-07-31 | 2016-01-29 | Seattle Genetics, Inc. | Konjugati zdravil in njihova uporaba za zdravljenje raka, avtoimunske bolezni ali infekcijske bolezni |
| EP1562627A4 (en) | 2002-08-23 | 2006-11-02 | Mclean Hospital Corp | CORTICOSTEROID CONJUGATES AND ITS USES |
| BR122012021252B8 (pt) | 2002-09-06 | 2021-05-25 | Cerulean Pharma Inc | polímeros à base de ciclodextrina para o fornecimento de agentes terapêuticos ligados a eles por covalência |
| CN1414008A (zh) | 2002-10-17 | 2003-04-30 | 南开大学 | 含氮甾体化合物及其制备方法 |
| US7015349B2 (en) | 2003-03-26 | 2006-03-21 | The Gillette Company | Reduction of hair growth |
| BR122018071808B8 (pt) | 2003-11-06 | 2020-06-30 | Seattle Genetics Inc | conjugado |
| WO2005063777A1 (en) | 2003-12-23 | 2005-07-14 | Corus Pharma | Benzylphosphate and substituted benzylphosphate prodrugs for the treatment of pulmonary inflammation |
| WO2005079523A2 (en) * | 2004-02-18 | 2005-09-01 | University Of Georgia Research Foundation, Inc. | Novel teleost derived antimicrobal polypeptides |
| AU2005216251B2 (en) | 2004-02-23 | 2011-03-10 | Genentech, Inc. | Heterocyclic self-immolative linkers and conjugates |
| US20050192257A1 (en) | 2004-02-26 | 2005-09-01 | Peyman Gholam A. | Predictors for patients at risk for glaucoma from steroid therapy |
| AR048098A1 (es) | 2004-03-15 | 2006-03-29 | Wyeth Corp | Conjugados de caliqueamicina |
| BRPI0510883B8 (pt) | 2004-06-01 | 2021-05-25 | Genentech Inc | composto conjugado de droga e anticorpo, composição farmacêutica, método de fabricação de composto conjugado de droga e anticorpo e usos de uma formulação, de um conjugado de droga e anticorpo e um agente quimioterapêutico e de uma combinação |
| EP1602931A1 (en) | 2004-06-03 | 2005-12-07 | Universiteit Leiden | SR-A antagonists |
| US7541330B2 (en) | 2004-06-15 | 2009-06-02 | Kosan Biosciences Incorporated | Conjugates with reduced adverse systemic effects |
| ES2315600T3 (es) | 2004-08-09 | 2009-04-01 | Deutsches Krebsforschungszentrum Stiftung Des Offentlichen Rechts | Conjugados de albumina que contienen un ligador glucuronico. |
| TW200616604A (en) | 2004-08-26 | 2006-06-01 | Nicholas Piramal India Ltd | Nitric oxide releasing prodrugs containing bio-cleavable linker |
| EP1817341A2 (en) | 2004-11-29 | 2007-08-15 | Seattle Genetics, Inc. | Engineered antibodies and immunoconjugates |
| WO2006135371A1 (en) | 2005-06-09 | 2006-12-21 | Kosan Biosciences Incorporated | Conjugates with reduced adverse systemic effects |
| US20100209508A1 (en) | 2005-06-14 | 2010-08-19 | Corus Pharma, Inc. | Substituted Phenylphosphates as Mutual Prodrugs of Steroids and ß-Agonists for the Treatment of Title Pulmonary Inflammation and Bronchoconstriction |
| US7750116B1 (en) | 2006-02-18 | 2010-07-06 | Seattle Genetics, Inc. | Antibody drug conjugate metabolites |
| PL2374818T3 (pl) | 2006-06-02 | 2013-05-31 | Regeneron Pharma | Przeciwciała o wysokim powinowactwie przeciw ludzkiemu receptorowi IL 6 |
| US8354508B2 (en) * | 2006-07-21 | 2013-01-15 | Diadexus, Inc. | PRO115 antibody compositions and methods of use |
| EP2109618A1 (en) | 2007-02-05 | 2009-10-21 | Nicox S.A. | Nitric oxide releasing steroids |
| WO2008122039A2 (en) | 2007-04-02 | 2008-10-09 | The Government Of The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Selenocysteine mediated hybrid antibody molecules |
| EP2167963B1 (en) | 2007-05-23 | 2019-04-17 | Ventana Medical Systems, Inc. | Polymeric carriers for immunohistochemistry and in situ hybridization |
| CN104383553A (zh) | 2007-06-25 | 2015-03-04 | 恩多塞特公司 | 含有亲水性间隔区接头的共轭物 |
| EP2170054A4 (en) | 2007-06-28 | 2012-08-08 | Capsutech Ltd | TARGETING CONJUGATES WITH ACTIVE SUBSTANCES ENCAPLED IN CYCLODEXTRIN POLYMERS |
| CN101397328A (zh) | 2007-09-27 | 2009-04-01 | 天津药业研究院有限公司 | 一氧化氮供体型糖皮质激素 |
| PL2214646T3 (pl) | 2007-10-05 | 2021-12-20 | Wayne State University | Dendrymery do przedłużonego uwalniania związków |
| CL2008003802A1 (es) | 2007-12-21 | 2010-02-19 | Schering Corp | Compuestos derivados de tioeteres c-21, agonistas del receptor de glucocorticoide; composicion farmaceutica; y uso para el tratamiento o profilaxis de una enfermedad o afeccion inmune, autoinmune o inflamatoria, tales como artritis reumatoide y esclerosis multiple. |
| WO2009085879A2 (en) | 2007-12-21 | 2009-07-09 | Schering Corporation | C20-c21 substituted glucocorticoid receptor agonists |
| MX2010011808A (es) | 2008-04-30 | 2011-03-04 | Immunogen Inc | Conjugados potentes y enlazadores hidrofilicos. |
| US20090318396A1 (en) | 2008-06-10 | 2009-12-24 | Gilead Sciences, Inc. | Corticosteroid linked beta-agonist compounds for use in therapy |
| CN102099058B (zh) | 2008-07-21 | 2014-10-22 | 宝力泰锐克斯有限公司 | 用于缀合生物分子的新试剂和方法 |
| WO2010010119A1 (en) * | 2008-07-22 | 2010-01-28 | Ablynx Nv | Amino acid sequences directed against multitarget scavenger receptors and polypeptides |
| AU2010221972B2 (en) | 2009-03-09 | 2012-07-05 | Mikasa Seiyaku Co., Ltd. | Steroid compound |
| WO2010126953A1 (en) | 2009-04-29 | 2010-11-04 | Gilead Sciences, Inc. | Corticosteroid linked beta-agonist compounds for use in therapy |
| WO2010132743A1 (en) | 2009-05-15 | 2010-11-18 | Gilead Sciences, Inc. | Corticosteroid beta-agonist compounds for use in therapy |
| JP5954789B2 (ja) | 2009-08-10 | 2016-07-20 | ユーシーエル ビジネス パブリック リミテッド カンパニー | 機能的分子の可逆的共有結合 |
| WO2011020107A2 (en) | 2009-08-14 | 2011-02-17 | Georgetown University | Compositions and methods for detection and treatment of breast cancer |
| GB0917044D0 (en) | 2009-09-29 | 2009-11-18 | Cytoguide As | Agents, uses and methods |
| GB0917054D0 (en) | 2009-09-29 | 2009-11-11 | Cytoguide As | Agents, uses and methods |
| WO2011081937A1 (en) | 2009-12-15 | 2011-07-07 | Gilead Sciences, Inc. | Corticosteroid-beta-agonist-muscarinic antagonist compounds for use in therapy |
| WO2011090940A1 (en) | 2010-01-19 | 2011-07-28 | Cerulean Pharma Inc. | Cyclodextrin-based polymers for therapeutic delivery |
| US9375473B2 (en) * | 2010-02-19 | 2016-06-28 | Cornell Research Foundation, Inc. | Method to treat autoimmune demyelinating diseases and other autoimmune or inflammatory diseases |
| BR112012026213B1 (pt) | 2010-04-15 | 2021-12-28 | Medimmune Limited | Compostos de pirrolobenzodiazepinas, conjugado das mesmas, composição farmacêutica compreendendo o conjugado e uso do mesmo para o tratamento de uma doença proliferativa |
| WO2012005982A2 (en) | 2010-07-06 | 2012-01-12 | Board Of Supervisors Of Louisiana State University And Agricultural And Mechanical College | Reporter for rna polymerase ii termination |
| JPWO2012011591A1 (ja) * | 2010-07-23 | 2013-09-09 | 武田薬品工業株式会社 | 縮合複素環化合物およびその用途 |
| IL315833A (en) | 2010-08-02 | 2024-11-01 | Regeneron Pharma | Mice producing binding proteins containing VL regions |
| CA2809487C (en) | 2010-09-01 | 2019-02-26 | Bayer Cropscience Ag | N-(tetrazol-5-yl)- and n-(triazol-5-yl)arylcarboxamides and use thereof as herbicides |
| WO2012058592A2 (en) | 2010-10-29 | 2012-05-03 | Immunogen, Inc. | Non-antagonistic egfr-binding molecules and immunoconjugates thereof |
| WO2012145632A1 (en) | 2011-04-21 | 2012-10-26 | Cerulean Pharma Inc. | Cyclodextrin-based polymers for therapeutic delivery |
| KR20140037105A (ko) | 2011-05-27 | 2014-03-26 | 암브룩스, 인코포레이티드 | 비-천연 아미노산 연결된 돌라스타틴 유도체를 함유하는 조성물, 이를 수반하는 방법, 및 용도 |
| US8815226B2 (en) | 2011-06-10 | 2014-08-26 | Mersana Therapeutics, Inc. | Protein-polymer-drug conjugates |
| AU2012322613B2 (en) | 2011-10-14 | 2016-04-21 | Medimmune Limited | Pyrrolobenzodiazepines and targeted conjugates |
| EA029046B1 (ru) | 2011-10-14 | 2018-02-28 | Медимьюн Лимитед | Пирролобензодиазепины и промежуточные соединения для их получения, способы их синтеза |
| KR101891859B1 (ko) | 2011-10-14 | 2018-08-24 | 메디뮨 리미티드 | 피롤로벤조디아제핀 |
| CA2850373C (en) | 2011-10-14 | 2019-07-16 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| WO2013068874A1 (en) | 2011-11-11 | 2013-05-16 | Pfizer Inc. | Antibody-drug conjugates |
| WO2013085925A1 (en) | 2011-12-05 | 2013-06-13 | Igenica, Inc. | Antibody-drug conjugates and related compounds, compositions, and methods |
| GB201122325D0 (en) | 2011-12-23 | 2012-02-01 | Cytoguide As | Novel formulations |
| PL2825553T3 (pl) | 2012-03-14 | 2018-12-31 | Regeneron Pharmaceuticals, Inc. | Wieloswoiste cząsteczki wiążące antygen i ich zastosowanie |
| EP2861259A1 (en) | 2012-06-14 | 2015-04-22 | Ambrx, Inc. | Anti-psma antibodies conjugated to nuclear receptor ligand polypeptides |
| HK1208813A1 (en) | 2012-10-11 | 2016-03-18 | Ascendis Pharma A/S | Diagnosis, prevention and treatment of diseases of the joint |
| US9662402B2 (en) | 2012-10-16 | 2017-05-30 | Endocyte, Inc. | Drug delivery conjugates containing unnatural amino acids and methods for using |
| HRP20230690T1 (hr) | 2012-10-23 | 2023-10-13 | Synaffix B.V. | Modificirano antitijelo, konjugat antitijela i postupak za njihovo pripremanje |
| WO2014065575A1 (ko) | 2012-10-25 | 2014-05-01 | 주식회사유한양행 | 4,5-다이하이드로-1h-피라졸 유도체 또는 그의 염 및 이를 포함하는 약학 조성물 |
| HRP20180822T1 (hr) | 2012-12-24 | 2018-08-10 | Abbvie Inc. | Vezujući proteini prolaktinskog receptora i njihovo korištenje |
| KR102274714B1 (ko) | 2013-03-13 | 2021-07-09 | 씨젠 인크. | 사이클로덱스트린 및 항체-약물 포합체 제형 |
| HK1222122A1 (zh) | 2013-04-22 | 2017-06-23 | Avelas Biosciences, Inc. | 选择性药物递送组合物及使用方法 |
| WO2014197854A1 (en) | 2013-06-06 | 2014-12-11 | Igenica Biotherapeutics, Inc. | Novel linkers for antibody-drug conjugates and related compounds, compositions, and methods of use |
| CA2914189C (en) | 2013-06-21 | 2023-03-14 | Innate Pharma | Enzymatic conjugation of polypeptides |
| WO2015005459A1 (ja) | 2013-07-10 | 2015-01-15 | 生化学工業株式会社 | 呼吸器投与用の医薬組成物 |
| TWI641620B (zh) | 2013-08-21 | 2018-11-21 | 再生元醫藥公司 | 抗-prlr抗體及其用途 |
| NZ758050A (en) | 2013-10-15 | 2024-03-22 | Seagen Inc | Pegylated drug-linkers for improved ligand-drug conjugate pharmacokinetics |
| CN103694375B (zh) | 2013-12-13 | 2016-10-05 | 北京大学 | 一种三萜-环糊精共价化合物及其制备方法和用途 |
| JP6527171B2 (ja) | 2014-01-24 | 2019-06-05 | サントル ナショナル ドゥ ラ ルシェルシュ シアンティフィック | クローン病などの大腸菌誘発性炎症性腸疾患治療用の経口摂取可能な化合物、これを調製するプロセスおよび付着防止薬としての使用 |
| US10550190B2 (en) | 2014-04-04 | 2020-02-04 | Merck Sharp & Dohme Corp. | Phosphate based linkers for intracellular delivery of drug conjugates |
| KR102186027B1 (ko) | 2014-04-10 | 2020-12-03 | 다이이치 산쿄 유럽 게엠베하 | 항her3 항체-약물 콘주게이트 |
| EP3148592A2 (en) | 2014-06-02 | 2017-04-05 | Regeneron Pharmaceuticals, Inc. | Antibody-drug conjugates, their preparation and their therapeutic use |
| DK3166638T3 (da) | 2014-06-13 | 2025-10-06 | Glykos Finland Oy | Nyttelagt-polymer-proteinkonjugater |
| BR112017011478A2 (pt) | 2014-12-03 | 2018-02-27 | Genentech, Inc. | composto de conjugado anticorpo-antibiótico, conjugado anticorpo-antibiótico, composição farmacêutica, métodos de tratamento de uma infecção bacteriana e para exterminar staph aureus, processo para a preparação do composto, kit e intermediário antibiótico-ligante |
| CN107206102A (zh) | 2014-12-03 | 2017-09-26 | 豪夫迈·罗氏有限公司 | 抗金黄色葡萄球菌抗体利福霉素缀合物及其用途 |
| MX2017007629A (es) | 2014-12-09 | 2018-05-17 | Abbvie Inc | Compuestos inhibidores de bcl-xl que tienen una baja permeabilidad en las celulas y conjugados de anticuerpo-farmaco que los incluyen. |
| BR112017012342A2 (pt) | 2014-12-09 | 2018-02-27 | Abbvie Inc | compostos inibitórios de bcl-xl e conjugados de anticorpo-fármaco incluindo os mesmos |
| US10233210B2 (en) | 2015-01-30 | 2019-03-19 | Coral Drugs Pvt. Ltd. | Process for preparation of glucocorticoid steroids |
| EP3253212A4 (en) | 2015-02-06 | 2018-09-19 | Sorrento Therapeutics, Inc. | Antibody drug conjugates |
| CA2987641A1 (en) * | 2015-07-06 | 2017-01-12 | Regeneron Pharmaceuticals, Inc. | Multispecific antigen-binding molecules and uses thereof |
| WO2017006279A1 (en) | 2015-07-08 | 2017-01-12 | Aten Porus Lifesciences | Cyclodextrin-polymer complexes and compostions and methods of making and using the same |
| CN117567535A (zh) | 2015-09-10 | 2024-02-20 | 坦伯公司 | 生物正交组合物 |
| WO2017062271A2 (en) | 2015-10-06 | 2017-04-13 | Merck Sharp & Dohme Corp. | Antibody drug conjugate for anti-inflammatory applications |
| WO2017132103A2 (en) | 2016-01-29 | 2017-08-03 | Merck Sharp & Dohme Corp. | Phosphonate linkers and their use to facilitate cellular retention of compounds |
| EP3419670A2 (en) | 2016-02-26 | 2019-01-02 | Regeneron Pharmaceuticals, Inc. | Optimized transglutaminase site-specific antibody conjugation |
| JP7073266B2 (ja) | 2016-03-25 | 2022-05-23 | シージェン インコーポレイテッド | Peg化薬物リンカー及びその中間体を調製するためのプロセス |
| GB201608936D0 (en) | 2016-05-20 | 2016-07-06 | Polytherics Ltd | Novel conjugates and novel conjugating reagents |
| MY194619A (en) | 2016-06-02 | 2022-12-07 | Abbvie Inc | Glucocorticoid receptor agonist and immunoconjugates thereof |
| JP2019522643A (ja) | 2016-06-08 | 2019-08-15 | アッヴィ・インコーポレイテッド | 抗cd98抗体及び抗体薬物コンジュゲート |
| MA46417A (fr) | 2016-09-23 | 2019-07-31 | Regeneron Pharma | Anticorps anti-steap2, conjugués anticorps-médicament, et molécules bispécifiques de liaison à l'antigène qui se lient à steap2 et cd3, et leurs utilisations |
| MY199937A (en) | 2016-11-08 | 2023-11-29 | Regeneron Pharma | Steroids and protein-conjugates thereof |
| IL298414B2 (en) | 2017-02-28 | 2024-12-01 | Immunogen Inc | Maytansinoid derivatives with self-immolative peptide linkers and conjugates thereof |
| KR20200007905A (ko) * | 2017-05-18 | 2020-01-22 | 리제너론 파마슈티칼스 인코포레이티드 | 사이클로덱스트린 단백질 약물 접합체 |
| WO2018213082A1 (en) | 2017-05-18 | 2018-11-22 | Regeneron Pharmaceuticals, Inc. | Bis-octahydrophenanthrene carboxamides and protein conjugates thereof |
| CN118652192A (zh) | 2017-05-24 | 2024-09-17 | 德克萨斯州大学系统董事会 | 用于抗体药物缀合物的接头 |
| WO2019034177A1 (zh) | 2017-08-18 | 2019-02-21 | 四川百利药业有限责任公司 | 具有两种不同药物的抗体药物偶联物 |
| CN109106951A (zh) | 2017-08-18 | 2019-01-01 | 四川百利药业有限责任公司 | 一种喜树碱-抗体偶联物 |
| CN111601619A (zh) * | 2017-11-07 | 2020-08-28 | 里珍纳龙药品有限公司 | 用于抗体药物偶联物的亲水性连接体 |
| US12070506B2 (en) * | 2018-01-08 | 2024-08-27 | Regeneron Pharmaceuticals, Inc. | Steroids and antibody-conjugates thereof |
| BR112020020466A2 (pt) | 2018-04-06 | 2021-01-12 | Seattle Genetics, Inc. | Conjugados de peptídeo de camptotecina |
| AU2019265703A1 (en) | 2018-05-09 | 2020-11-19 | Regeneron Pharmaceuticals, Inc. | Anti-MSR1 antibodies and methods of use thereof |
| CA3120528A1 (en) | 2018-11-20 | 2020-05-28 | Regeneron Pharmaceuticals, Inc. | Bis-octahydrophenanthrene carboxamide derivatives and protein conjugates thereof for use as lxr agonists |
| SG11202106658XA (en) | 2018-12-21 | 2021-07-29 | Regeneron Pharma | Rifamycin analogs and antibody-drug conjugates thereof |
| US20230119539A1 (en) | 2019-01-08 | 2023-04-20 | Regeneron Pharmaceuticals, Inc. | Traceless linkers and protein-conjugates thereof |
| JP2022517767A (ja) | 2019-01-08 | 2022-03-10 | レゲネロン ファーマシューティカルス,インコーポレーテッド | 無痕跡型リンカー及びそのタンパク質-コンジュゲート |
-
2019
- 2019-05-08 AU AU2019265703A patent/AU2019265703A1/en active Pending
- 2019-05-08 CN CN202410707876.3A patent/CN118955710A/zh active Pending
- 2019-05-08 MY MYPI2020005832A patent/MY209590A/en unknown
- 2019-05-08 KR KR1020207034456A patent/KR20210008008A/ko not_active Ceased
- 2019-05-08 JP JP2020562649A patent/JP7590871B2/ja active Active
- 2019-05-08 WO PCT/US2019/031383 patent/WO2019217591A1/en not_active Ceased
- 2019-05-08 US US16/407,099 patent/US11377502B2/en active Active
- 2019-05-08 MX MX2020011546A patent/MX2020011546A/es unknown
- 2019-05-08 SG SG11202010909RA patent/SG11202010909RA/en unknown
- 2019-05-08 CA CA3098453A patent/CA3098453A1/en active Pending
- 2019-05-08 CN CN201980046191.6A patent/CN112533951A/zh active Pending
- 2019-05-08 EP EP19725564.9A patent/EP3790899A1/en active Pending
- 2019-05-08 BR BR112020022400-6A patent/BR112020022400A2/pt unknown
-
2020
- 2020-11-04 PH PH12020551865A patent/PH12020551865A1/en unknown
- 2020-11-05 CL CL2020002873A patent/CL2020002873A1/es unknown
- 2020-11-30 CO CONC2020/0015084A patent/CO2020015084A2/es unknown
-
2022
- 2022-04-22 US US17/727,432 patent/US12497460B2/en active Active
-
2024
- 2024-06-05 JP JP2024091536A patent/JP2024133471A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| US12497460B2 (en) | 2025-12-16 |
| CL2020002873A1 (es) | 2021-03-26 |
| CN118955710A (zh) | 2024-11-15 |
| PH12020551865A1 (en) | 2021-07-19 |
| CA3098453A1 (en) | 2019-11-14 |
| EP3790899A1 (en) | 2021-03-17 |
| JP2024133471A (ja) | 2024-10-02 |
| MX2020011546A (es) | 2021-01-29 |
| WO2019217591A1 (en) | 2019-11-14 |
| JP2021523147A (ja) | 2021-09-02 |
| BR112020022400A2 (pt) | 2021-02-02 |
| SG11202010909RA (en) | 2020-12-30 |
| AU2019265703A1 (en) | 2020-11-19 |
| JP7590871B2 (ja) | 2024-11-27 |
| CO2020015084A2 (es) | 2021-02-26 |
| US20230079407A1 (en) | 2023-03-16 |
| MY209590A (en) | 2025-07-23 |
| CN112533951A (zh) | 2021-03-19 |
| US11377502B2 (en) | 2022-07-05 |
| US20190367631A1 (en) | 2019-12-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12497460B2 (en) | Anti-MSR1 antibodies and methods of use thereof | |
| JP7751054B2 (ja) | リファマイシンアナログおよびその抗体薬物コンジュゲート | |
| KR102877468B1 (ko) | 흔적이 없는 링커 및 이의 단백질-접합체 | |
| KR102713173B1 (ko) | 비스-옥타하이드로페난트렌 카복스아미드 및 이의 단백질 접합체 | |
| KR20230038738A (ko) | 단백질에서 글루타민 잔기에 접합된 캄토테신 유사체 및 그의 용도 | |
| BR112020013492A2 (pt) | Esteroides e conjugados de anticorpo dos mesmos | |
| KR20210094568A (ko) | Lxr 작용물질로서 사용하기 위한 비스-옥타하이드로펜안트렌 카복스아미드 유도체 및 그의 단백질 접합체 | |
| CN115968304A (zh) | 狄尔斯-阿尔德缀合方法 | |
| CN117015381A (zh) | 新颖类固醇有效负载、类固醇接头、含有其的adc及其用途 | |
| RU2847195C1 (ru) | Способы конъюгации дильса-альдера | |
| AU2023310999A1 (en) | Glucocorticoid receptor agonists and conjugates thereof | |
| EA049203B1 (ru) | Аналоги рифамицина и конъюгаты антитело-лекарственное средство с ними | |
| EA045111B1 (ru) | Стероиды и их антитело-конъюгаты |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-2-2-P10-P22-nap-X000 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| B15 | Application refused following examination |
Free format text: ST27 STATUS EVENT CODE: N-2-6-B10-B15-EXM-PE0601 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| PE0601 | Decision on rejection of patent |
St.27 status event code: N-2-6-B10-B15-exm-PE0601 |
|
| E13 | Pre-grant limitation requested |
Free format text: ST27 STATUS EVENT CODE: A-2-3-E10-E13-LIM-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11 | Amendment of application requested |
Free format text: ST27 STATUS EVENT CODE: A-2-2-P10-P11-NAP-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| T11 | Administrative time limit extension requested |
Free format text: ST27 STATUS EVENT CODE: U-3-3-T10-T11-OTH-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| T13 | Administrative time limit extension granted |
Free format text: ST27 STATUS EVENT CODE: U-3-3-T10-T13-OTH-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| T13-X000 | Administrative time limit extension granted |
St.27 status event code: U-3-3-T10-T13-oth-X000 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P12-X000 | Request for amendment of application rejected |
St.27 status event code: A-2-2-P10-P12-nap-X000 |