EP1268045B1 - Nouvelles membranes constituees d'un melange de polymeres, destinees a etre utilisees dans des piles a combustible - Google Patents
Nouvelles membranes constituees d'un melange de polymeres, destinees a etre utilisees dans des piles a combustible Download PDFInfo
- Publication number
- EP1268045B1 EP1268045B1 EP01911711A EP01911711A EP1268045B1 EP 1268045 B1 EP1268045 B1 EP 1268045B1 EP 01911711 A EP01911711 A EP 01911711A EP 01911711 A EP01911711 A EP 01911711A EP 1268045 B1 EP1268045 B1 EP 1268045B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- polymer
- polyether
- membrane
- membrane according
- sulphonated
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 CC(*)(*)c(cc1)cc([N+]([O-])=O)c1OC(C=CC(S(C(C)(*)*)(=O)=O)=C)=C* Chemical compound CC(*)(*)c(cc1)cc([N+]([O-])=O)c1OC(C=CC(S(C(C)(*)*)(=O)=O)=C)=C* 0.000 description 2
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/20—Manufacture of shaped structures of ion-exchange resins
- C08J5/22—Films, membranes or diaphragms
- C08J5/2206—Films, membranes or diaphragms based on organic and/or inorganic macromolecular compounds
- C08J5/2275—Heterogeneous membranes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/02—Details
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/52—Polyethers
- B01D71/522—Aromatic polyethers
- B01D71/5222—Polyetherketone, polyetheretherketone, or polyaryletherketone
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/66—Polymers having sulfur in the main chain, with or without nitrogen, oxygen or carbon only
- B01D71/68—Polysulfones; Polyethersulfones
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1023—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having only carbon, e.g. polyarylenes, polystyrenes or polybutadiene-styrenes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1027—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having carbon, oxygen and other atoms, e.g. sulfonated polyethersulfones [S-PES]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/102—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer
- H01M8/1032—Polymeric electrolyte materials characterised by the chemical structure of the main chain of the ion-conducting polymer having sulfur, e.g. sulfonated-polyethersulfones [S-PES]
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1039—Polymeric electrolyte materials halogenated, e.g. sulfonated polyvinylidene fluorides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1041—Polymer electrolyte composites, mixtures or blends
- H01M8/1044—Mixtures of polymers, of which at least one is ionically conductive
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/1016—Fuel cells with solid electrolytes characterised by the electrolyte material
- H01M8/1018—Polymeric electrolyte materials
- H01M8/1041—Polymer electrolyte composites, mixtures or blends
- H01M8/1053—Polymer electrolyte composites, mixtures or blends consisting of layers of polymers with at least one layer being ionically conductive
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2381/00—Characterised by the use of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing sulfur with or without nitrogen, oxygen, or carbon only; Polysulfones; Derivatives of such polymers
- C08J2381/02—Polythioethers; Polythioether-ethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2427/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers
- C08J2427/02—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment
- C08J2427/12—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
- C08J2427/16—Homopolymers or copolymers of vinylidene fluoride
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
Definitions
- the present invention relates to new blend polymer membranes based on sulfonated aromatic aryl polymers and their use as Polymer electrolyte membrane in fuel cells, especially in low-temperature fuel cells ,
- Fuel cell technology has great application potential in areas Space travel, road vehicles, submarines and stationary energy supply.
- the motor vehicles with fuel cell drives are considered A beacon of hope for improved environmental protection in the transport sector.
- Besides some technical problems, however, is a particular problem of the "cost-benefit ratio" from.
- the development of inexpensive elements with required performance for fuel cells of great interest is a particular problem of the "cost-benefit ratio" from.
- a polymer electrolyte membrane fuel cell Cell units consisting of current arrester, gas distributor, electrodes and Polymer electrolyte membrane.
- the electrodes usually contain platinum as Catalyst.
- Such fuel cells work with gaseous hydrogen or with methanol (DMFC Direct Methanol Fuel Cell).
- membranes do not only have to adequate chemical and mechanical stability as well as high Have proton conductivity, but also be inexpensive to manufacture. For this The reason are inexpensive raw materials with excellent properties Functionalization and the inexpensive processes for membrane production crucial roles.
- perfluorinated cation exchange membranes used so far show here strong deficits.
- this material is very expensive and has a high methanol permeability, which the application of these membranes in methanol fuel cells very limited.
- PBI polybenzimidazole
- PES polyether sulfone
- PBI treated with phosphoric acid Wang, J.S .; Wang, J.-T .; Savinell, R.F. .; Litt, M .; Moaddel, H .; Rogers, C .: Acid Doped Polybenzimidazoles, A New Polymer electrolyte; The Electrochemical Society, Spring Meeting, San Francisco, May 22-27, Extended Abstracts, vol. 94-1, 982-983 (1994)].
- phosphoric acid molecules are attached through hydrogen bonds Polymer attached and on the other hand by protonation of the imidazole groups tied up in the membrane.
- the problem is that the Phosphoric acid gradually with the water, which during the operation of the Fuel cell is created, from which PBI-Materix is eliminated.
- the PBI phosphoric acid membrane has a very low modulus of elasticity, which is why unsatisfactory membrane stability is to be expected in fuel cells.
- blended polymer membrane development is that the membrane structure and properties can be optimized by varying the blend component and the mixing ratio.
- DE-A-4422158 describes the blend polymer membranes made from sulfonated Polyether ketone (PEK) and unmodified polyether sulfone (PES).
- PEK sulfonated Polyether ketone
- PES unmodified polyether sulfone
- DE-A-4422158 describes three or four component blends made from sulfonated PEK, PES, polyvinylpyrrolidone (PVP) and polyglycol dimethyl ether (PG) are described show better water absorption, but without a quantitative indication do.
- Blended polymer membranes made from sulfonated aryl polymer PEEK or PSU with aminated polysulfone (PSU) are known from the prior art.
- Cui, W describes in the development and characterization of cation exchange membranes made of aryl polymers (VDI-Verlag; ISBN 3-18-359603-2) that the aminated polysulfones are a weak polybase and consequently a polyacid-base mixture can be produced in the solution. Both the ionic interaction and the hydrogen bonds, ie the ring structures, which have a physical crosslinking, are located between the blend components.
- This blend polymer membrane was tested in PEMFC and DMFC and resulted in a current density of 1.0-1.2 A / cm 2 at a voltage of 0.7V in H 2 / O 2 -PEMFC and from 0.4 to 0.6 A / cm 2 in air / H 2 -PEMFC. In DMFC, this membrane also showed a comparable UI characteristic such as Nafion-117. Against this background, the development of the blend polymer membranes crosslinked by ionic interaction for use in fuel cells has good prospects for low-temperature fuel cells.
- the present invention has for its object an inexpensive To provide polymer blend from the polymer electrolyte membranes for Fuel cells can be produced, these being at least the same or have improved performance over the prior art.
- the above task is solved by new ones through ionic interaction crosslinked blend polymer membranes based on modified polyether sulfone and Polyether ether sulfone, e.g. aminated polyethersulfone as amplifier and sulfonated aryl polymer as a functional polymer and a plasticizer.
- modified polyether sulfone and Polyether ether sulfone e.g. aminated polyethersulfone as amplifier and sulfonated aryl polymer as a functional polymer and a plasticizer.
- the functional polymer used according to the invention is sulfonated aryl polymers such as sulfonated PEEK (SPEEK), sulfonated PEK (SPEK), sulfonated PEEKK (SPEEKK), sulfonated PES (SPES) sulfonated PEES (SPEES).
- SPEEK sulfonated PEEK
- SPEK sulfonated PEK
- SPEEKK sulfonated PEEKK
- SPES sulfonated PES
- the blend polymer membrane can be made of PBI and modified Polyether sulfone or modified polyether ether sulfone can be produced.
- This Blended polymer membrane like PBI membrane, is functionalized by phosphoric acid.
- Aryl polymers of this type contain aromatic building blocks selected from the group and temperature stable bonds selected from the group
- the sulfonation of aryl polymers is known. So describes EP 0574791 Manufacture of sulfonated PEEK. From EP-A-008895, EP-A-041780 and EP 0576807 the production of sulfonated PEK is known. The production of sulfonated PEEKK is from E. Müller in "Crosslinked PEEKK sulfonamides for Separation of aliphatic / aromatic mixtures "[diploma thesis, 1995, Hoechst AG, Frankfurt / Main] known. The production is described in EP-A-0008894 and EP-A-0112724 described by polyethersulfone.
- the degree of sulfonation is preferably between 0.1 and 100%
- the functional polymer according to the invention is used in amounts between 30 and 99.9 % By weight based on the total polymer used.
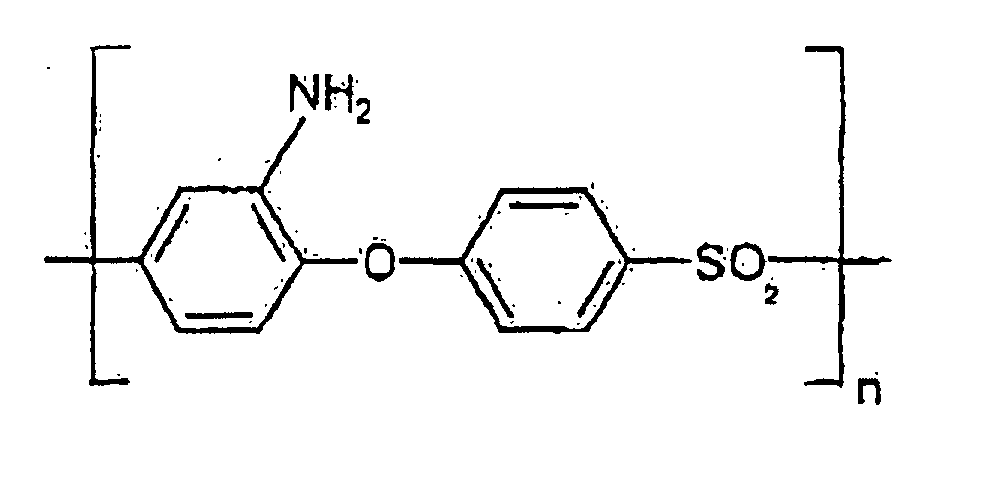
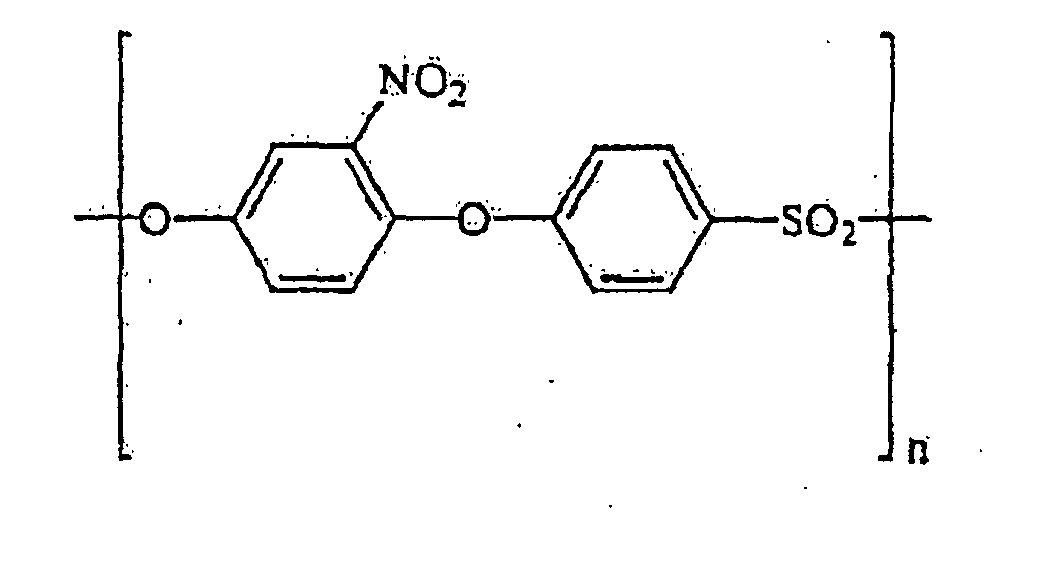
- the reinforcing polymer used according to the invention is aminated polyether sulfone and polyether ether sulfone containing the structural units or where x is independently an integer 0, 1, 2, 3 or 4, or nitrated polyether sulfone and polyether ether sulfone containing the structural units or where x is independently an integer 0, 1, 2, 3 or 4
- Aminated polyether sulfones and polyether ether sulfones containing structural units of the types are particularly preferred or
- Nitrated polyether sulfones and polyether ether sulfones containing structural units of the types are particularly preferred or
- the reinforcing polymer according to the invention is used in amounts between 0.1 and 70 % By weight, preferably 10 to 50% by weight, based on the total polymer used.
- polyacid-polybase blends or polyacid-polyacid blends can be represented as follows: PES-NH 2 + aryl polymer-SO 3 H ⁇ PES- (NH 3 ) + - SO 3 aryl polymer PES-NO 2 + aryl polymer- SO 3 H ⁇ PES- (NO 2 -H) + - SO 3 aryl polymer eg: PES-NH 2 + PES- SO 3 H ⁇ PES- (NH 3 ) + - SO 3 -PES PES-NO 2 + PES- SO 3 H ⁇ PES- (NO 2 -H) + - SO 3 -PES
- the sulfonated polyether sulfone (PES-SO3H) and the nitrided PES (PES-N02) are both polyacid and therefore completely miscible with each other.
- the compatibility between PES-SO3H and aminated PES (PES-NH 2 ) is harmless due to the complete miscibility of the polyacid-polybase mixture.
- PES-NH 2 and PES-NO 2 serve as "macromolecular counterions" to reinforce the membrane. Although the ionic compound is dissolved in water at an elevated temperature, the interaction remains because of the location of the "macromolecular counterions" at the sites.
- the membrane is reinforced by these "macromolecular counterions" at an elevated temperature and, on the other hand, the ion transport is favored because of this resolution.
- the membrane according to the invention thus has the favorable properties for use in fuel cells at elevated temperature.
- Polyether sulfones (PES) are commercially available and are characterized by high thermal, chemical resistance and mechanical stability. The polarity of the polymer promotes water absorption.
- the production processes for nitrated and aminated polyether sulfone and nitrided and aminated polyether ether sulfone are described in German patent application No. 10010002.3, which was filed at the same time.
- plasticizers are understood to be those which Reduce the brittleness of the membrane made from the polymer blend.
- suitable Plasticizers must be used under the conditions prevailing in a fuel cell be intimate.
- the plasticizers with the functional and Reinforcement polymers to be miscible and compatible, as well as in the same dipolar solvent for example dimethylformamide (DMF), dimethyl oxide (DMSO), N-methylpyrrolidone (NMP) or N, N-dimethylacetamide (DMAC).
- PVDF polyvinylidene fluoride
- the plasticizer content is up to 5% by weight, preferably between 0.001 and 3 wt .-%, in particular between 0.1 and 2 wt .-%, based on the Total polymers.
- the PVDF is commercially available and is characterized by excellent chemical and thermal stability.
- the chemical structure of PVDF can be described as follows [CF 2 ⁇ CF 2 ] n
- the production of the three-component blend polymer membrane according to the invention is also carried out according to the following procedure.
- the preparation of the blend polymer membranes according to the invention is as follows carried out: A solution of a homogeneous polymer mixture containing the sulfonated aryl polymer, the aminated PES or the nitrided PES and Plasticizer is poured onto a carrier and then a uniform thick film pulled out with a squeegee. The solvent in the film is e.g. by Evaporation removed. Dimethylformamide is particularly suitable as a solvent (DMF), dimethyl oxide (DMSO), N-methylpyrrolidone (NMP) or N, N-dimethylacetamide (DMAC). The dry film is then removed from the carrier detached and the membrane produced is conditioned.
- DMF solvent
- DMSO dimethyl oxide
- NMP N-methylpyrrolidone
- DMAC N-dimethylacetamide
- the starting polymer is cheaper and Modification process of PES and PEES and the production of the Blend polymer membrane are simple.
- the membrane structure or properties such as conductivity, Membrane swelling can be adjusted depending on the application Comparison to the membranes made of sulfonated aryl polymer blend polymer membranes according to the invention improved mechanical and have thermal properties.
- the blend polymer membrane according to the invention can consist of one layer or of several identical or different layers (multilayer), for. B. from double layer of a) sulfonated aryl polymer and aminated polysulfone and b) sulfonated aryl polymer and nitrided polysulfone.
- various multilayer membranes according to the invention the at least two different layers selected from the group SPEK and NH 2 -PES, SPEK and NO 2 -PES SPEK and NH 2 -PES and plasticizers, SPEK and NO 2 -PES and plasticizers.
- the mechanical properties of the comparison membrane are shown in the table below: Modulus of elasticity (N / mm 2 ); in moisture; 23 ° C 300 N / mm 2 Modulus of elasticity (N / mm 2 ); in water; 80 ° C 50 N / mm 2 Elongation at break (%) in moisture; 23 ° C 175% Elongation at break (%) in water; 80 ° C 255%
- the permeability of the comparison membrane is as follows: temperature (° C) Methanol / H 2 O (g / m 2 .d) (32g / 1000g H 2 O) O 2 permeability [cm 3 50 ⁇ m / (m 2 d bar)] H 2 permeability [cm 3 50 ⁇ m / (m 2 d bar)] 40 61.9 2350 9880 60 151 5250 18800 80 333 8590 36300 100 674 14300 65200 120 1266 23300 113100
- the SPEEK has an ion exchange capacity of 1.73 meq / g.
- the NH 2 -PES has a degree of substitution of 45% (1.9 meq / g).
- the blend polymer membrane TE-4 consists of 90% by weight SPEEK and 10% by weight NH 2 -PES, TE-5 consists of 85% by weight SPEEK and 15% by weight NH 2 -PES.
- the material permeability in the membrane TE-5 is 2.35 [g.50 ⁇ m / (m 2 .d)] at 40 ° C.
- the SPEEKK has an ion exchange capacity of 1.65 meq / g.
- the NH 2 -PES has a degree of substitution of 45% (1.9 meq / g).
- the blend polymer membrane TE-8 consists of 90% by weight SPEEKK and 10% by weight NH 2 -PES, TE-9 consists of 85% by weight SPEEKK and 15% by weight NH 2 -PES.
- the methanol permeability in the membrane TE-8 is 4.11 [g.50 ⁇ m / (m 2 .d)] at 40 ° C.
- the SPEK has an ion exchange capacity of 2.13 meq / g.
- the NO 2 -PES is the degree of substitution of 50% (1.97 meq / g).
- the blend polymer membrane TE-23 consists of 80% by weight SPEK and 20% by weight NO 2 -PES, TE-24 from 75% by weight SPEK and 25% by weight NO 2 -PES TE-25 from 70% by weight % SPEK and 30% by weight NO 2 -PES.
- Three-component blend polymer membranes made of SPEK, NH 2 -PES (IEC 1.9 meq / g) and PVDF.
- the glass transition temperature was determined by means of DSC and the decomposition temperature was determined by means of TGA.
- Such a membrane with the double layer consisting of a total of four components.
- One of the double layers consists of sulfonated PEK, aminated PES and PVDF, the other of sulfonated PEK, nitrided PES and PVDF.
- the component PES-NO 2 is very stable against oxidation under oxygen
- PES-NH 2 is very stable against reduction under hydrogen or methanol. This improves chemical stability.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- General Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Sustainable Development (AREA)
- Sustainable Energy (AREA)
- Electrochemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Composite Materials (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Fuel Cell (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
Claims (20)
- Membrane contenantA) au moins un polymère de fonction sur la base d'un ou de plusieurs arylpolymères porteur de groupes d'acide sulfonique,B) au moins un polymère renforçant sur la base d'un ou de plusieurs polyéthersulfones aminés et/ou polyétheréther-sulfones aminés ou polyéthersulfones nitrés et/ou polyétheréther-sulfones nitrés, lequel améliore par son interaction avec le polymère de fonction la stabilité de la membrane contre le gonflement etC) au moins un plastifiant qui réduit la fragilité des polymères cités ci-avant.
- Membrane selon la revendication 1, caractérisée en ce que l'arylpolymère sulfoné utilisé est un polyétheréther-cétone sulfoné, un cétone de polyéther sulfoné, un cétone de polyétheréther-cétone sulfoné, un polyéthersulfone sulfoné et un polyétheréther-sulfone sulfoné ou un PBI.
- Membrane selon l'une des revendications 1 à 3, caractérisée en ce que l'arylpolymère sulfoné présente un degré de sulfonation entre 0,1 et 100 %.
- Membrane selon l'une des revendications 1 à 4, caractérisée en ce que l'arylpolymère sulfoné est utilisé dans des quantités entre 30 et 99,9 % en poids, rapporté au polymère total.
- Membrane selon l'une des revendications 1 à 9, caractérisée en ce que le polymère renforçant selon l'invention est utilisé dans des quantités entre 0,1 et 70 % en poids, rapporté au polymère total.
- Membrane selon l'une des revendications 1 à 10, caractérisée en ce que le plastifiant réduit la fragilité de la membrane fabriquée à partir du blend polymère.
- Membrane selon l'une des revendications 1 à 11, caractérisée en ce que le plastifiant est inerte dans les conditions qui règnent dans une pile à combustible.
- Membrane selon l'une des revendications 1 à 12, caractérisée en ce que le plastifiant est miscible et compatible avec le polymère de fonction et le polymère renforçant.
- Membrane selon l'une des revendications 1 à 13, caractérisée en ce que le plastifiant est soluble dans du diméthylformadide (DMF), du diméthylsulfoxyde (DMSO), du N-méthylpyrrolidone (NMP) ou du N,N-diméthylacétamide (DMCA).
- Membrane selon l'une des revendications 1 à 14, caractérisée en ce que le plastifiant utilisé est un polyfluorure de vinylidène (PVDF) linéaire.
- Membrane selon l'une des revendications 1 à 15, caractérisée en ce que la teneur en plastifiant s'élève à jusqu'à 5 % en poids, rapporté au poids du polymère total.
- Membrane selon l'une des revendications 1 à 16, caractérisée en ce que la membrane se compose de plusieurs couches pareilles ou différentes (multicouches).
- Membrane selon la revendication 17, caractérisée en ce que la membrane se compose d'au moins deux couches différentes choisies dans le groupe cétone de polyéther sulfoné et polyéthersulfone aminé, cétone de polyéther sulfoné et polyéthersulfone nitré, cétone de polyéther sulfoné et polyéthersulfone aminé et plastifiant, cétone de polyéther sulfoné et polyéthersulfone nitré et plastifiant.
- Utilisation de la membrane selon l'une ou plusieurs des revendications 1 à 18 pour la fabrication d'unités membrane-électrode (MEA) pour piles à combustibles, en particulier pour les piles à basse températures sollicitées à des températures de fonctionnement de 10 °C à 200 °C.
- Pile à combustible contenant une membrane d'électrolyte polymère selon l'une ou plusieurs des revendications 1 à 18.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DK01911711T DK1268045T3 (da) | 2001-03-01 | 2001-03-01 | Nye blandingspolymermembraner til anvendelse i brændselsceller |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE10010001A DE10010001A1 (de) | 2000-03-02 | 2000-03-02 | Neue Blendpolymermembranen zum Einsatz in Brennstoffzellen |
| DE10010001 | 2000-03-02 | ||
| PCT/EP2001/002311 WO2001064322A1 (fr) | 2000-03-02 | 2001-03-01 | Nouvelles membranes constituees d'un melange de polymeres, destinees a etre utilisees dans des piles a combustible |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1268045A1 EP1268045A1 (fr) | 2003-01-02 |
| EP1268045B1 true EP1268045B1 (fr) | 2004-06-30 |
Family
ID=7633133
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP01911711A Expired - Lifetime EP1268045B1 (fr) | 2000-03-02 | 2001-03-01 | Nouvelles membranes constituees d'un melange de polymeres, destinees a etre utilisees dans des piles a combustible |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US6869980B2 (fr) |
| EP (1) | EP1268045B1 (fr) |
| JP (1) | JP2003526716A (fr) |
| KR (1) | KR100734800B1 (fr) |
| CN (1) | CN1227056C (fr) |
| AT (1) | ATE270141T1 (fr) |
| CA (1) | CA2401838C (fr) |
| DE (2) | DE10010001A1 (fr) |
| ES (1) | ES2228818T3 (fr) |
| MX (1) | MXPA02008584A (fr) |
| WO (1) | WO2001064322A1 (fr) |
Families Citing this family (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE478913T1 (de) * | 2000-06-02 | 2010-09-15 | Stanford Res Inst Int | Polymermembranzusammensetzung |
| DE10140147A1 (de) * | 2001-08-16 | 2003-03-06 | Celanese Ventures Gmbh | Verfahren zur Herstellung einer Blend-Membran aus verbrücktem Polymer und Brennstoffzelle |
| JP3561250B2 (ja) | 2001-09-21 | 2004-09-02 | 株式会社日立製作所 | 燃料電池 |
| JP4399586B2 (ja) * | 2002-01-08 | 2010-01-20 | 本田技研工業株式会社 | 高分子電解質膜、該高分子電解質膜を備える膜電極構造体及び該膜電極構造体を備える固体高分子型燃料電池 |
| US7534515B2 (en) | 2002-01-23 | 2009-05-19 | Polyfuel, Inc. | Acid-base proton conducting polymer blend membrane |
| AU2003209080A1 (en) * | 2002-02-06 | 2003-09-02 | Battelle Memorial Institute | Polymer electrolyte membranes for use in fuel cells |
| US7625652B2 (en) * | 2002-04-25 | 2009-12-01 | Basf Fuel Cell Gmbh | Multilayer electrolyte membrane |
| CN100509875C (zh) * | 2002-05-13 | 2009-07-08 | 复合燃料公司 | 磺化共聚物 |
| KR100977234B1 (ko) | 2002-05-13 | 2010-08-23 | 더 유니버시티 오브 노스 플로리다 보드 오브 트러스티즈 | 술폰화된 공중합체 |
| WO2004004037A1 (fr) * | 2002-06-28 | 2004-01-08 | Sumitomo Chemical Company, Limited | Feuilles polymeres, procedes d'obtention desdites feuilles et leur utilisation |
| US7019819B2 (en) | 2002-11-13 | 2006-03-28 | Molecular Imprints, Inc. | Chucking system for modulating shapes of substrates |
| US8003725B2 (en) | 2002-08-12 | 2011-08-23 | Exxonmobil Chemical Patents Inc. | Plasticized hetero-phase polyolefin blends |
| US7531594B2 (en) | 2002-08-12 | 2009-05-12 | Exxonmobil Chemical Patents Inc. | Articles from plasticized polyolefin compositions |
| US7271209B2 (en) | 2002-08-12 | 2007-09-18 | Exxonmobil Chemical Patents Inc. | Fibers and nonwovens from plasticized polyolefin compositions |
| CA2492839C (fr) | 2002-08-12 | 2011-02-01 | Exxonmobil Chemical Patents Inc. | Compositions de polyolefines plastifiees |
| US7998579B2 (en) | 2002-08-12 | 2011-08-16 | Exxonmobil Chemical Patents Inc. | Polypropylene based fibers and nonwovens |
| DE10246461A1 (de) * | 2002-10-04 | 2004-04-15 | Celanese Ventures Gmbh | Protonenleitende Polymermembran enthaltend Polyazolblends und deren Anwendung in Brennstoffzellen |
| US7641840B2 (en) * | 2002-11-13 | 2010-01-05 | Molecular Imprints, Inc. | Method for expelling gas positioned between a substrate and a mold |
| DE10301810A1 (de) * | 2003-01-20 | 2004-07-29 | Sartorius Ag | Membran-Elektroden-Einheit, Polymermembranen für eine Membran-Elektroden-Einheit und Polymerelektrolyt-Brennstoffzellen sowie Verfahren zur Herstellung derselben |
| JP4852828B2 (ja) * | 2003-07-31 | 2012-01-11 | 東洋紡績株式会社 | 電解質膜・電極構造体 |
| US8192813B2 (en) | 2003-08-12 | 2012-06-05 | Exxonmobil Chemical Patents, Inc. | Crosslinked polyethylene articles and processes to produce same |
| JP2008500701A (ja) * | 2004-05-22 | 2008-01-10 | フオスター・ミラー・インコーポレイテツド | 固体ポリマー電解質膜 |
| JP5028736B2 (ja) * | 2004-09-02 | 2012-09-19 | 東レ株式会社 | 高分子電解質材、ならびにそれを用いた高分子電解質膜、膜電極複合体および高分子電解質型燃料電池 |
| US8389615B2 (en) | 2004-12-17 | 2013-03-05 | Exxonmobil Chemical Patents Inc. | Elastomeric compositions comprising vinylaromatic block copolymer, polypropylene, plastomer, and low molecular weight polyolefin |
| DE102005001599A1 (de) * | 2005-01-12 | 2006-07-20 | Basf Ag | Funktionalisierte Polyarylether |
| FR2883292B1 (fr) * | 2005-03-16 | 2008-01-04 | Inst Nat Polytech Grenoble | Extrusion de polymeres ioniques a groupements ioniques acides |
| FR2883293B1 (fr) * | 2005-03-16 | 2007-05-25 | Inst Nat Polytech Grenoble | Extrusion de polymeres ioniques a groupements ioniques alcalins |
| ATE555166T1 (de) | 2005-07-15 | 2012-05-15 | Exxonmobil Chem Patents Inc | Elastomere zusammensetzungen |
| US7615300B2 (en) * | 2005-08-30 | 2009-11-10 | The Board of Regents University and Community College System of Nevada on Behalf of the University of Nevada | Development of novel proton-conductive polymers for proton exchange membrane fuel cell (PEMFC) technology |
| US7316554B2 (en) * | 2005-09-21 | 2008-01-08 | Molecular Imprints, Inc. | System to control an atmosphere between a body and a substrate |
| US20070065700A1 (en) * | 2005-09-22 | 2007-03-22 | Sri International | High temperature polymer electrolyte membranes |
| CA2878756A1 (fr) | 2005-09-30 | 2007-04-12 | Battelle Memorial Institute | Polymeres utilises dans des composants de pile a combustible |
| KR100707163B1 (ko) * | 2005-10-12 | 2007-04-13 | 삼성에스디아이 주식회사 | 고체산, 이를 포함하는 고분자 전해질막 및 이를 채용한연료전지 |
| KR101324413B1 (ko) * | 2006-02-27 | 2013-11-01 | 삼성에스디아이 주식회사 | 고온 고분자 전해질 연료전지 스택의 기동 방법 및 이를이용하는 연료전지 시스템 |
| WO2007123805A2 (fr) * | 2006-04-03 | 2007-11-01 | Molecular Imprints, Inc. | Système d'impression lithographique |
| JP5151074B2 (ja) * | 2006-06-08 | 2013-02-27 | 株式会社日立製作所 | 固体高分子電解質膜,膜電極接合体およびそれを用いた燃料電池 |
| KR100815117B1 (ko) | 2006-06-30 | 2008-03-20 | 한국과학기술원 | 연료전지용 고분자 전해질 막의 제조방법 |
| WO2008076637A1 (fr) * | 2006-12-14 | 2008-06-26 | Arkema Inc. | Polyélectrolytes stables à haute température ayant des groupes aromatiques de squelette |
| ITMI20071034A1 (it) | 2007-05-23 | 2008-11-24 | St Microelectronics Srl | Sintesi e caratterizzazione di una nuova membrana a scambio protonico (pem) per applicazioni in celle a combustibile |
| KR100986493B1 (ko) * | 2008-05-08 | 2010-10-08 | 주식회사 동진쎄미켐 | 연료전지용 고분자 전해질 막 |
| US20100096764A1 (en) * | 2008-10-20 | 2010-04-22 | Molecular Imprints, Inc. | Gas Environment for Imprint Lithography |
| KR101118202B1 (ko) | 2009-10-01 | 2012-03-28 | 한국과학기술연구원 | 연료전지용 고분자 전해질막 및 이의 제조방법 |
| US8815467B2 (en) | 2010-12-02 | 2014-08-26 | Basf Se | Membrane electrode assembly and fuel cells with improved lifetime |
| DE102010055143B4 (de) | 2010-12-18 | 2022-12-01 | Umicore Galvanotechnik Gmbh | Direktkontakt-Membrananode für die Verwendung in Elektrolysezellen |
| US10139116B2 (en) | 2014-06-16 | 2018-11-27 | Core Energy Recovery Solutions Inc. | Blended membranes for water vapor transport and methods for preparing same |
| JP6530630B2 (ja) * | 2015-03-31 | 2019-06-12 | 株式会社Ihi | 高分子アロイ電解質膜およびその製造方法 |
| CN105789657B (zh) * | 2016-04-27 | 2018-09-11 | 工业和信息化部电子第五研究所华东分所 | 一种用于钒电池的改性pes-pvp共混阴离子交换膜及其制备方法 |
| CN106750436B (zh) * | 2017-01-18 | 2019-07-23 | 吉林大学 | 一种表面磺化聚醚醚酮微纳米粒子/磺化聚醚醚酮复合膜及其制备方法 |
| CN110197918A (zh) * | 2018-02-27 | 2019-09-03 | 湖南省银峰新能源有限公司 | 一种全钒液流电池用全氟磺酸复合膜及其制备方法和用途 |
| CN110731026A (zh) * | 2018-05-17 | 2020-01-24 | 松下知识产权经营株式会社 | 液流电池 |
| WO2020116650A1 (fr) * | 2018-12-07 | 2020-06-11 | Agc株式会社 | Perfluoro-polymère, composition liquide, film électrolytique à polymère solide, corps lié membrane et électrode, et pile à combustible de type à polymère solide |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3936997A1 (de) * | 1989-11-07 | 1991-05-08 | Hoechst Ag | Semipermeable membran aus polyetherketonen |
| DE19813613A1 (de) * | 1998-03-27 | 1999-09-30 | Jochen Kerres | Modifiziertes Polymer und modifizierte Polymermembran |
| DE19817374A1 (de) * | 1998-04-18 | 1999-10-21 | Univ Stuttgart Lehrstuhl Und I | Engineering-Ionomerblends und Engineering-Ionomermembranen |
| DE19817376A1 (de) * | 1998-04-18 | 1999-10-21 | Univ Stuttgart Lehrstuhl Und I | Säure-Base-Polymerblends und ihre Verwendung in Membranprozessen |
-
2000
- 2000-03-02 DE DE10010001A patent/DE10010001A1/de not_active Withdrawn
-
2001
- 2001-03-01 AT AT01911711T patent/ATE270141T1/de not_active IP Right Cessation
- 2001-03-01 JP JP2001563213A patent/JP2003526716A/ja not_active Withdrawn
- 2001-03-01 WO PCT/EP2001/002311 patent/WO2001064322A1/fr active IP Right Grant
- 2001-03-01 CA CA002401838A patent/CA2401838C/fr not_active Expired - Fee Related
- 2001-03-01 CN CNB018058868A patent/CN1227056C/zh not_active Expired - Fee Related
- 2001-03-01 KR KR1020027011424A patent/KR100734800B1/ko not_active IP Right Cessation
- 2001-03-01 MX MXPA02008584A patent/MXPA02008584A/es not_active Application Discontinuation
- 2001-03-01 EP EP01911711A patent/EP1268045B1/fr not_active Expired - Lifetime
- 2001-03-01 DE DE50102740T patent/DE50102740D1/de not_active Expired - Lifetime
- 2001-03-01 US US10/220,899 patent/US6869980B2/en not_active Expired - Fee Related
- 2001-03-01 ES ES01911711T patent/ES2228818T3/es not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| EP1268045A1 (fr) | 2003-01-02 |
| CN1406150A (zh) | 2003-03-26 |
| CA2401838C (fr) | 2007-09-18 |
| US6869980B2 (en) | 2005-03-22 |
| KR100734800B1 (ko) | 2007-07-03 |
| DE10010001A1 (de) | 2001-09-06 |
| MXPA02008584A (es) | 2003-02-24 |
| KR20020084165A (ko) | 2002-11-04 |
| CA2401838A1 (fr) | 2001-09-07 |
| ATE270141T1 (de) | 2004-07-15 |
| JP2003526716A (ja) | 2003-09-09 |
| WO2001064322A1 (fr) | 2001-09-07 |
| ES2228818T3 (es) | 2005-04-16 |
| US20030187081A1 (en) | 2003-10-02 |
| DE50102740D1 (de) | 2004-08-05 |
| CN1227056C (zh) | 2005-11-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1268045B1 (fr) | Nouvelles membranes constituees d'un melange de polymeres, destinees a etre utilisees dans des piles a combustible | |
| EP1073690B1 (fr) | Melanges constitues d'acides, de bases et de polymeres, et leur utilisation dans des procedes a membrane | |
| DE60020915T2 (de) | Polymere Kompositmembran und Verfahren zu ihrer Herstellung | |
| EP1337319B1 (fr) | Nouvelles membranes presentant de meilleures proprietes mecaniques, destinees a un usage dans des piles a combustible | |
| DE60114776T2 (de) | Verfahren zur herstellung einer polymerelektrolytmembran | |
| DE60214166T2 (de) | Polymerelektrolyt für eine brennstoffzelle des festpolymertyps und brennstoffzelle | |
| EP1425336B1 (fr) | Procede de fabrication d'une membrane constituee d'un melange a base de polymere ponte et pile a combustible associee | |
| DE60013843T2 (de) | Verbesserte polymerelektrolytmembrane aus gemischten dispersionen | |
| EP0688824B1 (fr) | Allignes polymériques homogènes à la base de polyéther cétones aromatiques sulfonés | |
| EP2009728B1 (fr) | Procédé de fabrication d'un polymère poly(1,3,4-oxadiazol) sulfoné | |
| DE60025101T2 (de) | Protonenleitendes Polymer, Herstellungsverfahren dafür, fester Polymerelektrolyt und Elektrode | |
| EP1144100A2 (fr) | Composition polymere, membrane la contenant, son procede de production et son utilisation | |
| DE10296922T5 (de) | Elektrodenstruktur für Polymerelektrolytbrennstoffzellen, Verfahren zum Herstellen derselben und Polymerelektrolytbrennstoffzelle | |
| DE102008043936A1 (de) | Verstärkte Verbundmembran für Polymerelektrolytbrennstoffzelle | |
| DE102012212420A1 (de) | Membran mit laminierter Struktur und orientierungsgesteuerten Nanofaser-Verstärkungszusatzstoffen für Brennstoffzellen | |
| DE10155543C2 (de) | Protonenleitende Elektrolytmembran, Verfahren zu ihrer Herstellung und deren Verwendung | |
| DE10296598T5 (de) | Brennstoffzelle mit polymerem Elektrolyt | |
| WO2003060012A1 (fr) | Polymeres a chaine principale fonctionnalises | |
| DE102020213449A1 (de) | Membran-Elektrodeneinheit mit verbesserter Beständigkeit und Protonenleitfähigkeit und Verfahren zu deren Herstellung | |
| DE19847782A1 (de) | Verfahren zum Betrieb einer Brennstoffzelle | |
| DE60212209T2 (de) | Brennstoffzelle und membran-elektrodenanordnung dafür | |
| DE60004148T2 (de) | Verfahren zur Herstellung von meta-verknüpftem Polyanilin | |
| DE102017208805A1 (de) | Verfahren zur Herstellung von Blockcopolychinoxalinen mit Sulfon- oder Phosphonsäure-Gruppen und deren Einsatz als protonenleitende Membran | |
| DE10296977T5 (de) | Elektrodenstruktur für Polymerelektrolytbrennstoffzellen | |
| DE10134793A1 (de) | Membranen für Ionentransport |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20021002 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE TR |
|
| 17Q | First examination report despatched |
Effective date: 20030314 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE TR |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20040630 Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20040630 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20040630 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: GERMAN |
|
| REF | Corresponds to: |
Ref document number: 50102740 Country of ref document: DE Date of ref document: 20040805 Kind code of ref document: P |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20040930 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20040930 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20050301 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050301 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FD4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050331 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2228818 Country of ref document: ES Kind code of ref document: T3 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| ET | Fr: translation filed | ||
| 26N | No opposition filed |
Effective date: 20050331 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PUE Owner name: PEMEAS GMBH Free format text: CELANESE VENTURES GMBH##65926 FRANKFURT (DE) -TRANSFER TO- PEMEAS GMBH#INDUSTRIEPARK HOECHST#65926 FRANKFURT AM MAIN (DE) |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E |
|
| NLS | Nl: assignments of ep-patents |
Owner name: PEMEAS GMBH Effective date: 20050815 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20041130 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DK Payment date: 20080313 Year of fee payment: 8 Ref country code: ES Payment date: 20080328 Year of fee payment: 8 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EBP |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090331 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20100323 Year of fee payment: 10 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20090302 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20100313 Year of fee payment: 10 Ref country code: FR Payment date: 20100324 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20100224 Year of fee payment: 10 Ref country code: AT Payment date: 20100312 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20090302 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20100304 Year of fee payment: 10 Ref country code: DE Payment date: 20100312 Year of fee payment: 10 Ref country code: BE Payment date: 20100322 Year of fee payment: 10 |
|
| BERE | Be: lapsed |
Owner name: *PEMEAS G.M.B.H. Effective date: 20110331 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: V1 Effective date: 20111001 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20110301 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110301 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20111130 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110331 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111001 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110331 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110331 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110331 Ref country code: NL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20111001 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 50102740 Country of ref document: DE Effective date: 20111001 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110301 Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20110301 |