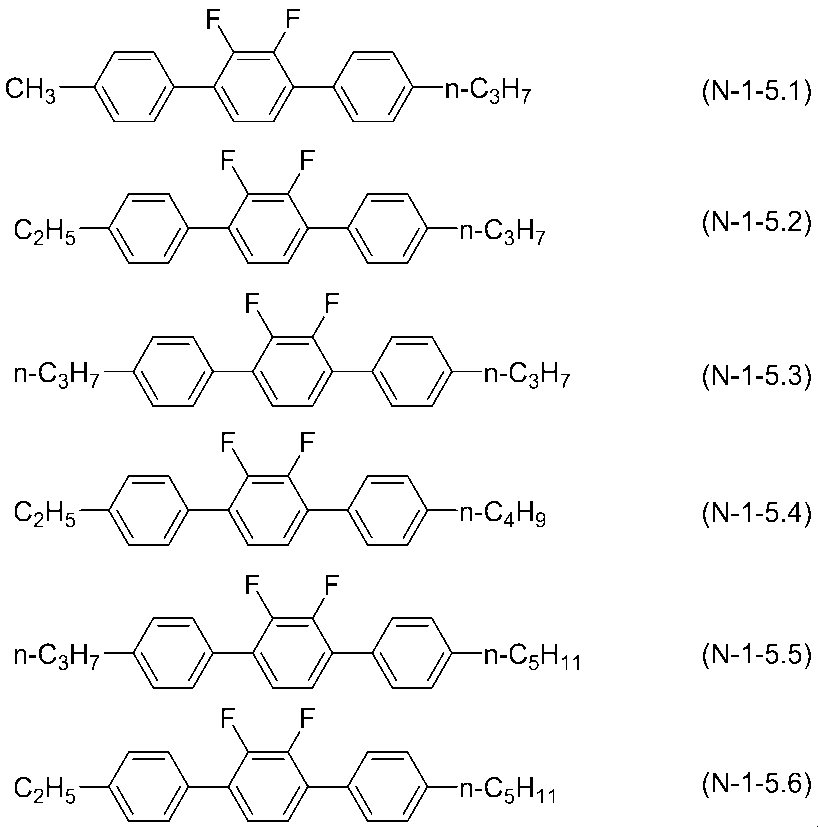WO2016035786A1 - ネマチック液晶組成物及びこれを用いた液晶表示素子 - Google Patents
ネマチック液晶組成物及びこれを用いた液晶表示素子 Download PDFInfo
- Publication number
- WO2016035786A1 WO2016035786A1 PCT/JP2015/074835 JP2015074835W WO2016035786A1 WO 2016035786 A1 WO2016035786 A1 WO 2016035786A1 JP 2015074835 W JP2015074835 W JP 2015074835W WO 2016035786 A1 WO2016035786 A1 WO 2016035786A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mass
- group
- formula
- liquid crystal
- carbon atoms
- Prior art date
Links
- 0 CCC(*)(C(CC1)CCC1c(cc1)ccc1-c(cc1)ccc1OC)N Chemical compound CCC(*)(C(CC1)CCC1c(cc1)ccc1-c(cc1)ccc1OC)N 0.000 description 6
- KELHQTAZSUEQSJ-UHFFFAOYSA-N CC(CC1)CCC1c(cc1)ccc1-c(cc1)ccc1OC Chemical compound CC(CC1)CCC1c(cc1)ccc1-c(cc1)ccc1OC KELHQTAZSUEQSJ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/42—Mixtures of liquid crystal compounds covered by two or more of the preceding groups C09K19/06 - C09K19/40
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3066—Cyclohexane rings in which the rings are linked by a chain containing carbon and oxygen atoms, e.g. esters or ethers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/34—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring
- C09K19/3402—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having oxygen as hetero atom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/12—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings at least two benzene rings directly linked, e.g. biphenyls
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/14—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a carbon chain
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/14—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a carbon chain
- C09K19/16—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a carbon chain the chain containing carbon-to-carbon double bonds, e.g. stilbenes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/14—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a carbon chain
- C09K19/18—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a carbon chain the chain containing carbon-to-carbon triple bonds, e.g. tolans
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/20—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a chain containing carbon and oxygen atoms as chain links, e.g. esters or ethers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/24—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings linked by a chain containing nitrogen-to-nitrogen bonds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/32—Non-steroidal liquid crystal compounds containing condensed ring systems, i.e. fused, bridged or spiro ring systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
- C09K19/542—Macromolecular compounds
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/133—Constructional arrangements; Operation of liquid crystal cells; Circuit arrangements
- G02F1/1333—Constructional arrangements; Manufacturing methods
- G02F1/1337—Surface-induced orientation of the liquid crystal molecules, e.g. by alignment layers
-
- G—PHYSICS
- G02—OPTICS
- G02F—OPTICAL DEVICES OR ARRANGEMENTS FOR THE CONTROL OF LIGHT BY MODIFICATION OF THE OPTICAL PROPERTIES OF THE MEDIA OF THE ELEMENTS INVOLVED THEREIN; NON-LINEAR OPTICS; FREQUENCY-CHANGING OF LIGHT; OPTICAL LOGIC ELEMENTS; OPTICAL ANALOGUE/DIGITAL CONVERTERS
- G02F1/00—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics
- G02F1/01—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour
- G02F1/13—Devices or arrangements for the control of the intensity, colour, phase, polarisation or direction of light arriving from an independent light source, e.g. switching, gating or modulating; Non-linear optics for the control of the intensity, phase, polarisation or colour based on liquid crystals, e.g. single liquid crystal display cells
- G02F1/133—Constructional arrangements; Operation of liquid crystal cells; Circuit arrangements
- G02F1/136—Liquid crystal cells structurally associated with a semi-conducting layer or substrate, e.g. cells forming part of an integrated circuit
- G02F1/1362—Active matrix addressed cells
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K2019/0444—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit characterized by a linking chain between rings or ring systems, a bridging chain between extensive mesogenic moieties or an end chain group
- C09K2019/0448—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit characterized by a linking chain between rings or ring systems, a bridging chain between extensive mesogenic moieties or an end chain group the end chain group being a polymerizable end group, e.g. -Sp-P or acrylate
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/10—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings
- C09K19/12—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing at least two benzene rings at least two benzene rings directly linked, e.g. biphenyls
- C09K2019/121—Compounds containing phenylene-1,4-diyl (-Ph-)
- C09K2019/123—Ph-Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3004—Cy-Cy
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3009—Cy-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/301—Cy-Cy-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3016—Cy-Ph-Ph
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/08—Non-steroidal liquid crystal compounds containing at least two non-condensed rings
- C09K19/30—Non-steroidal liquid crystal compounds containing at least two non-condensed rings containing saturated or unsaturated non-aromatic rings, e.g. cyclohexane rings
- C09K19/3001—Cyclohexane rings
- C09K19/3003—Compounds containing at least two rings in which the different rings are directly linked (covalent bond)
- C09K2019/3027—Compounds comprising 1,4-cyclohexylene and 2,3-difluoro-1,4-phenylene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/04—Liquid crystal materials characterised by the chemical structure of the liquid crystal components, e.g. by a specific unit
- C09K19/06—Non-steroidal liquid crystal compounds
- C09K19/34—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring
- C09K19/3402—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having oxygen as hetero atom
- C09K2019/3422—Non-steroidal liquid crystal compounds containing at least one heterocyclic ring having oxygen as hetero atom the heterocyclic ring being a six-membered ring
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K19/00—Liquid crystal materials
- C09K19/52—Liquid crystal materials characterised by components which are not liquid crystals, e.g. additives with special physical aspect: solvents, solid particles
- C09K19/54—Additives having no specific mesophase characterised by their chemical composition
- C09K19/542—Macromolecular compounds
- C09K2019/548—Macromolecular compounds stabilizing the alignment; Polymer stabilized alignment
Definitions
- Liquid crystal display elements are used in clocks, calculators, various household electrical devices, measuring devices, automotive panels, word processors, electronic notebooks, printers, computers, televisions, and the like.
- Typical liquid crystal display methods include TN (twisted nematic), STN (super twisted nematic), DS (dynamic light scattering), GH (guest / host), and IPS (in-plane switching).
- Type OCB (optical compensation birefringence) type, ECB (voltage controlled birefringence) type, VA (vertical alignment) type, CSH (color super homeotropic) type, FLC (ferroelectric liquid crystal), etc.
- Examples of the driving method include static driving, multiplex driving, simple matrix method, active matrix (AM) method driven by TFT (thin film transistor), TFD (thin film diode), and the like.
- the IPS type, ECB type, VA type, CSH type, and the like have a feature that a liquid crystal material having a negative value of ⁇ is used.
- the VA type display method by AM driving is used for a display element that requires a high speed and a wide viewing angle, such as a television.
- a nematic liquid crystal composition used for a display method such as a VA type is required to have a low voltage drive, a high-speed response, and a wide operating temperature range. That is, ⁇ is negative, the absolute value is large, the viscosity is low, and a high nematic phase-isotropic liquid phase transition temperature (T ni ) is required. Further, from the setting of ⁇ n ⁇ d, which is the product of refractive index anisotropy ( ⁇ n) and cell gap (d), it is necessary to adjust ⁇ n of the liquid crystal material to an appropriate range according to the cell gap. In addition, when applying a liquid crystal display element to a television or the like, since high-speed response is important, a liquid crystal material having a low viscosity ( ⁇ ) is required.
- a liquid crystal composition using the following liquid crystal compounds (A) and (B) having a 2,3-difluorophenylene skeleton (Patent Document 1) as a liquid crystal material having a negative ⁇ is disclosed.
- liquid crystal compounds (C) and (D) are used as compounds having ⁇ of almost 0.
- the high-speed response of this liquid crystal composition is a performance required when used for a liquid crystal television or the like. Was not satisfied.
- the problem to be solved by the present invention is that the viscosity ( ⁇ ) is sufficiently small without reducing the refractive index anisotropy ( ⁇ n) and the nematic phase-isotropic liquid phase transition temperature (T ni ), and the rotational viscosity
- a liquid crystal composition having a negative dielectric anisotropy ( ⁇ ) having a sufficiently small ( ⁇ 1), a large elastic constant (K 33 ), and a large absolute value is provided.
- An object of the present invention is to provide a liquid crystal display element which has no defects or is suppressed, has excellent heat resistance, has excellent display quality, and has a high response speed.
- the present inventor has studied various compounds and found that the above problems can be solved by combining specific compounds, and has completed the present invention.
- the liquid crystal composition of the present invention has the general formula (i) as the first component.
- R L1 and R L2 each independently represents an alkyl group having 1 to 8 carbon atoms, and one or two or more non-adjacent —CH 2 — in the alkyl group are each independently Optionally substituted by —CH ⁇ CH—, —C ⁇ C—, —O—, —CO—, —COO— or —OCO—, n L1 represents 0, 1, 2 or 3,
- a L1 , A L2 and A L3 each independently represent (a) a 1,4-cyclohexylene group (one —CH 2 — present in the group or two or more —CH 2 — not adjacent to each other).
- the group (a), the group (b) and the group (c) may be each independently substituted with a cyano group, a fluorine atom or a chlorine atom
- n L1 is 2 or 3 and a plurality of A L2 are present, they may be the same or different, and when n L1 is 2 or 3, and a plurality of Z L3 are present, May be the same or different, but excludes the compound represented by formula (i).
- the liquid crystal composition characterized by containing the 1 type, or 2 or more types of compound chosen from the compound represented by this is
- the liquid crystal composition of the present invention has a sufficiently low viscosity ( ⁇ ) and a rotational viscosity ( ⁇ 1) without lowering the refractive index anisotropy ( ⁇ n) and the nematic phase-isotropic liquid phase transition temperature (T ni ). ) Is sufficiently small, the elastic constant (K 33 ) is large, and the absolute value has a large negative dielectric anisotropy ( ⁇ ). Therefore, a VA-type liquid crystal display element using this has no display defect or Suppressed, excellent display quality and fast response speed.
- the liquid crystal composition of the present invention has the general formula (i) as the first component.
- R i1 represents an alkyl group having 1 to 8 carbon atoms, preferably an alkyl group having 1 to 5 carbon atoms, more preferably an alkyl group having 1 to 3 carbon atoms, A methyl group is most preferred.
- R i2 represents an alkyl group having 3 to 8 carbon atoms, preferably an alkyl group having 3 to 6 carbon atoms, more preferably an alkyl group having 3 or 4 carbon atoms, and 3 carbon atoms. Most preferred are alkyl groups.
- Examples of the “alkyl group having 1 to 8 carbon atoms” according to the present invention include a methyl group, an ethyl group, a propyl group, a butyl group, an isopentyl group, a neopentyl group, a pentyl group, a hexyl group, a heptyl group, and an octyl group.
- alkyl groups are common and are appropriately selected from the above examples depending on the number of carbon atoms of each alkyl group.
- the alkyl group having 1 to 8 carbon atoms according to the present invention is preferably linear or branched, and more preferably linear.
- the compound represented by the general formula (i) is preferably a compound described below.
- the content of the compound represented by the general formula (i) is preferably 0.1% by mass to 30% by mass, and 0.1% by mass to 25% by mass. It is preferably 0.2% by mass to 20% by mass, preferably 0.2% by mass to 15% by mass, and preferably 0.5% by mass to 10% by mass.
- the content is preferably from 10% by mass to 10% by mass, and preferably from 1% by mass to 8% by mass.
- the lower limit of the content of the first component is, for example, 0.1 as an embodiment of the present invention with respect to the total amount (100% by mass) of the liquid crystal composition of the present invention. % By mass. Moreover, in another embodiment of this invention, it is 0.2 mass%. Or in another embodiment of the present invention, it is 0.5 mass%. Alternatively, in another embodiment of the present invention, the content is 1% by mass.
- the upper limit of the content of the first component is, for example, 30% by mass in one form of the present invention with respect to the total amount of the liquid crystal composition of the present invention. Moreover, in another embodiment of this invention, it is 25 mass%. Or in another embodiment of the present invention, it is 20 mass%. Alternatively, in another embodiment of the present invention, the content is 10% by mass. Furthermore, or in another embodiment of the present invention, it is 8% by mass.
- the liquid crystal composition of the present invention contains one or more compounds selected from the compounds represented by formula (L) as the second component.
- the compound represented by the general formula (L) corresponds to a dielectrically neutral compound ( ⁇ value is ⁇ 2 to 2).
- R L1 and R L2 each independently represents an alkyl group having 1 to 8 carbon atoms, and one or two or more non-adjacent —CH 2 — in the alkyl group are each Independently may be substituted by —CH ⁇ CH—, —C ⁇ C—, —O—, —CO—, —COO— or —OCO—, n L1 represents 0, 1, 2 or 3, A L1 , A L2 and A L3 each independently represent (a) a 1,4-cyclohexylene group (one —CH 2 — present in the group or two or more —CH 2 — not adjacent to each other).
- the group (a), the group (b) and the group (c) may be each independently substituted with a cyano group, a fluorine atom or a chlorine atom
- the compounds represented by the general formula (L) may be used alone or in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to desired properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention. Alternatively, in another embodiment of the present invention, there are two types, three types, four types, five types, six types, seven types, eight types, nine types, 10 types, More than types.
- the content of the compound represented by the general formula (L) is low-temperature solubility, transition temperature, electrical reliability, birefringence, process compatibility, dripping marks, image sticking, It is necessary to appropriately adjust according to required performance such as dielectric anisotropy.
- the content of the compound represented by the general formula (L) is preferably 1% by mass to 80% by mass, preferably 10% by mass to 70% by mass, and 20% by mass to 60% by mass. It is preferably 25% to 55% by weight, preferably 25% to 55% by weight, preferably 25% to 50% by weight, and 30% to 50% by weight. It is preferable that it is 35 mass% to 50 mass%.
- the content of the compound represented by the general formula (L) is preferably 1% by mass or more, and preferably 10% by mass or more, as a lower limit in the composition. It is preferable to contain at least 25% by mass, preferably at least 25% by mass, preferably at least 29% by mass, preferably at least 30% by mass, and preferably at least 35% by mass.
- the upper limit value is preferably 80% by mass or less, preferably 70% by mass or less, preferably 60% by mass or less, preferably 55% by mass or less, and preferably 50% by mass or less. It is preferable to contain.
- the above lower limit value is preferably high and the upper limit value is preferably high. Furthermore, when the composition of the present invention maintains a high Tni and requires a composition having good temperature stability, the above lower limit value is preferably high and the upper limit value is preferably high. Further, when it is desired to increase the dielectric anisotropy in order to keep the driving voltage low, it is preferable that the above lower limit value is lowered and the upper limit value is low.
- R L1 and R L2 are preferably both alkyl groups, and when importance is placed on reducing the volatility of the compound, it is preferably an alkoxy group, and importance is placed on viscosity reduction. In this case, at least one is preferably an alkenyl group.
- R L1 and R L2 are each a linear alkyl group having 1 to 5 carbon atoms or a linear alkyl group having 1 to 5 carbon atoms when the ring structure to which R L1 is bonded is a phenyl group (aromatic).
- a phenyl group aromatic
- Alkyl groups, linear alkoxy groups having 1 to 4 carbon atoms and linear alkenyl groups having 2 to 5 carbon atoms are preferred.
- the total of carbon atoms and oxygen atoms, if present, is preferably 5 or less, and is preferably linear.
- the alkenyl group is preferably selected from groups represented by any of the formulas (R1) to (R5). (The black dots in each formula represent carbon atoms in the ring structure.)
- n L1 is preferably 0 when importance is attached to the response speed, 2 or 3 is preferred for improving the upper limit temperature of the nematic phase, and 1 is preferred for balancing these. In order to satisfy the properties required for the composition, it is preferable to combine compounds having different values.
- it represents a trans-1,4-cyclohexylene group or a 1,4-phenylene group.
- Z L1 and Z L2 are preferably single bonds when the response speed is important.
- the compound represented by the general formula (L) contains one or more compounds represented by the general formula (L-1).
- R L11 , R L12 , A L12 , and A L13 each independently represent the same meaning as R L1 , R L2 , A L2 , and A L3 in the general formula (L), and n L11 is 0 Or 1)
- R L11 and R L12 are preferably a linear alkyl group having 1 to 5 carbon atoms, a linear alkoxy group having 1 to 5 carbon atoms, and a linear alkenyl group having 2 to 5 carbon atoms. .
- the compound represented by the general formula (L-1) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the content of the compound represented by the general formula (L-1) is preferably 1% by mass to 80% by mass, preferably 10% by mass to 70% by mass, and 20% by mass to 60% by mass. It is preferably 25% by mass to 55% by mass, preferably 29% by mass to 55% by mass, more preferably 29% by mass to 50% by mass, and 30% by mass to 50% by mass. It is preferable that it is mass%, and it is preferable that it is 35 mass% to 50 mass%.
- the content of the compound represented by the general formula (L-1) is preferably 1% by mass or more, and preferably 10% by mass or more, as a lower limit in the composition. 20% by mass or more, preferably 25% by mass or more, preferably 29% by mass or more, preferably 30% by mass or more, and preferably 35% by mass or more. .
- the upper limit value is preferably 80% by mass or less, preferably 70% by mass or less, preferably 60% by mass or less, preferably 55% by mass or less, and preferably 50% by mass or less. It is preferable to contain.
- the above lower limit value is preferably high and the upper limit value is preferably high. Furthermore, when the composition of the present invention requires a high Tni and a composition having good temperature stability, it is preferable that the lower limit value is moderate and the upper limit value is moderate. When it is desired to increase the dielectric anisotropy in order to keep the driving voltage low, it is preferable that the lower limit value is low and the upper limit value is low.
- the compound represented by the general formula (L-1) is preferably a compound selected from the group of compounds represented by the general formulas (L-1-1) to (L-1-4). In particular, it is preferable to contain one or more compounds represented by general formula (L-1-1) and / or general formula (L-1-4).
- the compound represented by the general formula (L-1-1) is the following compound.
- R L111 and R L112 each independently represent the same meaning as R L1 and R L2 in the general formula (L).
- R L111 and R L112 are preferably a linear alkyl group having 1 to 5 carbon atoms, a linear alkoxy group having 1 to 4 carbon atoms, and a linear alkenyl group having 2 to 5 carbon atoms. .
- the compound represented by the general formula (L-1-1) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the above lower limit value is preferably high and the upper limit value is preferably high. Furthermore, when the composition of the present invention requires a high Tni and a composition having good temperature stability, it is preferable that the lower limit value is moderate and the upper limit value is moderate. When it is desired to increase the dielectric anisotropy in order to keep the driving voltage low, it is preferable that the lower limit value is low and the upper limit value is low.
- the compound represented by the general formula (L-1-1) is preferably a compound selected from the group of compounds represented by the general formula (L-1-1-1).
- R L1112 represents the same meaning as R L2 in the general formula (L).
- the compound represented by the general formula (L-1-1-1) is selected from the group of compounds represented by the formula (L-1-1-1.1) to the formula (L-1-1-1.3). The selected compound is preferable, and the compound represented by the formula (L-1-1-1.2) or the formula (L-1-1-1.3) is preferable. The compound represented by (1-1-1.3) is preferable.
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-1.3) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, % By mass, 5% by mass, 7% by mass, and 10% by mass.
- the upper limit of the preferable content is 20% by mass, 15% by mass, 13% by mass, 10% by mass, 8% by mass, and 7% by mass with respect to the total amount of the composition of the present invention. % By mass, 6% by mass, 5% by mass, and 3% by mass.
- the compound represented by the general formula (L-1-1) is preferably a compound selected from the group of compounds represented by the general formula (L-1-1-2).
- R L1122 represents the same meaning as R L2 in the general formula (L).
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-2) with respect to the total amount of the composition of the present invention is 1% by mass, 5% by mass, 10% by mass 15% by mass 17% by mass 20% by mass 23% by mass 25% by mass 27% by mass 30% by mass 35% by mass .
- the upper limit of the preferable content is 60% by mass, 55% by mass, 50% by mass, 45% by mass, 42% by mass, and 40% by mass with respect to the total amount of the composition of the present invention. % By mass, 38% by mass, 35% by mass, 33% by mass, and 30% by mass.
- the compound represented by the general formula (L-1-1-2) is a compound represented by the formula (L-1-1-2.1) to the formula (L-1-1-2.4).
- a compound selected from the group is preferable, and a compound represented by Formula (L-1-1-2.2) to Formula (L-1-1-2.4) is preferable.
- the compound represented by the formula (L-1-1-2.2) is preferable because the response speed of the composition of the present invention is particularly improved.
- the content of the compounds represented by formula (L-1-1-2.3) and formula (L-1-1-2.4) is 30% by mass or more in order to improve the solubility at low temperatures. That is not preferable.
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-2.2) with respect to the total amount of the composition of the present invention is 10% by mass, 15% by mass, % By mass, 20% by mass, 23% by mass, 25% by mass, 27% by mass, 30% by mass, 33% by mass, 35% by mass, and 38% by mass It is 40 mass%.
- the upper limit of the preferable content is 60% by mass, 55% by mass, 50% by mass, 45% by mass, 43% by mass, and 40% by mass with respect to the total amount of the composition of the present invention. % By mass, 38% by mass, 35% by mass, 32% by mass, 30% by mass, 27% by mass, 25% by mass and 22% by mass.
- the total of the compound represented by the formula (L-1-1-1.3) and the compound represented by the formula (L-1-1-2.2) with respect to the total amount of the composition of the present invention is preferable.
- the lower limit of the content is 10% by mass, 15% by mass, 20% by mass, 25% by mass, 27% by mass, 30% by mass, 35% by mass, 40% % By mass.
- the upper limit of the preferable content is 60% by mass, 55% by mass, 50% by mass, 45% by mass, 43% by mass, and 40% by mass with respect to the total amount of the composition of the present invention. % By mass, 38% by mass, 35% by mass, 32% by mass, 30% by mass, 27% by mass, 25% by mass and 22% by mass.
- R L1131 and R L1132 each independently represent an alkyl group having 1 to 8 carbon atoms or an alkoxy group having 1 to 8 carbon atoms.
- R L1131 and R L1132 are preferably a linear alkyl group having 1 to 5 carbon atoms, a linear alkoxy group having 1 to 4 carbon atoms, and a linear alkenyl group having 2 to 5 carbon atoms. .
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-3) with respect to the total amount of the composition of the present invention is 1% by mass, 5% by mass, 10% by mass 13% by mass, 15% by mass, 17% by mass, 20% by mass, 23% by mass, 25% by mass, and 30% by mass.
- the upper limit of the preferable content is 60% by mass, 55% by mass, 50% by mass, 45% by mass, 40% by mass, and 37% with respect to the total amount of the composition of the present invention.
- the compounds represented by the general formula (L-1-1-3) are compounds represented by the formulas (L-1-1-3.1) to (L-1-1-3.12).
- the compound is selected from the group represented by formula (L-1-1-3.1), formula (L-1-1-3.3) or formula (L-1-1-3.4). It is preferable that it is a compound represented.
- the compound represented by the formula (L-1-1-3.1) is preferable because the response speed of the composition of the present invention is particularly improved. Further, when obtaining Tni higher than the response speed, the equations (L-1-1-3.3), (L-1-1-3.4), and (L-1-1-3.11) are used. ) And a compound represented by the formula (L-1-1-3.12) are preferably used.
- Formula (L-1-1-3.3), Formula (L-1-1-3.4), Formula (L-1-1-3.11) and Formula (L-1-1-3.12) The total content of the compounds represented by) is not preferably 20% by mass or more in order to improve the solubility at low temperatures.
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-3.1) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, % By mass, 5% by mass, 7% by mass, 10% by mass, 13% by mass, 15% by mass, 18% by mass and 20% by mass.
- the upper limit of the preferable content is 20% by mass, 17% by mass, 15% by mass, 13% by mass, 10% by mass, and 8% by mass with respect to the total amount of the composition of the present invention. % By mass, 7% by mass, and 6% by mass.
- the compound represented by the general formula (L-1-1) is a compound selected from the group of compounds represented by the general formula (L-1-1-4) and / or (L-1-1-5). It is preferable.
- R L1142 and R L1152 each independently represent an alkyl group having 1 to 8 carbon atoms or an alkoxy group having 1 to 8 carbon atoms.
- R L1142 and R L1152 are preferably a linear alkyl group having 1 to 5 carbon atoms, a linear alkoxy group having 1 to 4 carbon atoms, and a linear alkenyl group having 2 to 5 carbon atoms. .
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-4) with respect to the total amount of the composition of the present invention is 1% by mass, 5% by mass, 10% by mass 13% by mass, 15% by mass, 17% by mass, and 20% by mass.
- the upper limit of the preferable content is 25% by mass, 23% by mass, 20% by mass, 17% by mass, 15% by mass, and 13% by mass with respect to the total amount of the composition of the present invention. % By mass, and 10% by mass.
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-5) with respect to the total amount of the composition of the present invention is 1% by mass, 5% by mass, 10% by mass 13% by mass, 15% by mass, 17% by mass, and 20% by mass.
- the upper limit of the preferable content is 25% by mass, 23% by mass, 20% by mass, 17% by mass, 15% by mass, and 13% by mass with respect to the total amount of the composition of the present invention. % By mass, and 10% by mass.
- the compounds represented by the general formulas (L-1-1-4) and (L-1-1-5) are represented by the formulas (L-1-1-4.1) to (L-1-1).
- -5.3) is preferably a compound selected from the group of compounds represented by formula (L-1-1-4.2) or formula (L-1-1-5.2) A compound is preferred.
- the lower limit of the preferable content of the compound represented by the formula (L-1-1-4.2) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, % By mass, 5% by mass, 7% by mass, 10% by mass, 13% by mass, 15% by mass, 18% by mass and 20% by mass.
- the upper limit of the preferable content is 20%, 17% by mass, 15% by mass, 13% by mass, 10% by mass, and 8% by mass with respect to the total amount of the composition of the present invention. %, 7% by mass, and 6% by mass.
- the lower limit of the preferred content of the total content of these compounds is 1% by weight, 2% by weight, 3% by weight, based on the total amount of the composition of the present invention, 5% by mass, 7% by mass, 10% by mass Yes, 13% by mass, 15% by mass, 18% by mass, 20% by mass, 23% by mass, 25% by mass, 27% by mass, 30% by mass, 33% by mass, 35% by mass, and the upper limit is
- the formula (L-1-1-3.1), the formula (L-1-1-3.3) and the formula (L-1-1-3.4) It is preferable to combine two or more compounds selected from the compounds represented by formula (L-1-1-1.3) and formula (L-1- It is preferable to combine two or more compounds selected from the compounds represented by 1-2-2).
- the compound represented by the general formula (L-1-2) is the following compound.
- R L121 and R L122 each independently represent the same meaning as R L1 and R L2 in General Formula (L).
- R L121 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms
- R L122 is an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or a carbon atom.
- An alkoxy group of 1 to 4 is preferable.
- the compound represented by the general formula (L-1-2) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (L-1-2) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass 5% by mass, 7% by mass, and 10% by mass.
- the upper limit of the preferable content is 20% by mass, 15% by mass, 13% by mass, 10% by mass, 8% by mass, and 7% by mass with respect to the total amount of the composition of the present invention. % By mass, 6% by mass, 5% by mass, and 3% by mass.
- the compound represented by the general formula (L-1-2) is a compound selected from the group of compounds represented by the formula (L-1-2.1) to the formula (L-1-2.6).
- it is represented by the formula (L-1-2.1), the formula (L-1-2.3), the formula (L-1-2.4), and the formula (L-1-2.6). It is preferable that it is a compound.
- the compound represented by the general formula (L-1-3) is the following compound.
- R L131 and R L132 each independently represent the same meaning as R L1 and R L2 in General Formula (L).
- R L131 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms
- R L132 is an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or a carbon atom.
- An alkoxy group of 1 to 4 is preferable.
- the compound represented by the general formula (L-1-3) can be used alone, or two or more compounds can be used in combination.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the content of the compound represented by the general formula (L-1-3) is the solubility at low temperature, the transition temperature, the electrical reliability, the birefringence, the process suitability, the dropwise addition. It is necessary to adjust appropriately according to the required performance such as marks, image sticking, and dielectric anisotropy.
- the lower limit of the preferable content of the compound represented by the formula (L-1-3) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass 5% by weight, 7% by weight, 10% by weight, 14% by weight, 16% by weight, 20% by weight, 23% by weight, 26% by weight, 30% Mass%, 35 mass%, and 40 mass%.
- the upper limit of the preferable content of the compound represented by the formula (L-4) with respect to the total amount of the composition of the present invention is 50% by mass, 40% by mass, 35% by mass, 30% % By mass, 20% by mass, 15% by mass, 10% by mass, and 5% by mass.
- the compound represented by the general formula (L-1-3) is preferably, for example, a compound represented by the formula (L-1-3.1) to the formula (L-1-3.3).
- the total amount of the composition of the present invention is The lower limit of the preferred content of both compounds is 15% by weight, 19% by weight, 24% by weight, 30% by weight, and the preferred upper limit is 45% by weight, 40% by weight. 35% by mass, 30% by mass, 25% by mass, 23% by mass, 20% by mass, 18% by mass, 15% by mass and 13% by mass .
- the compound represented by the general formula (L-1-3) is preferably a compound represented by the formula (L-1-3.4) to the formula (L-1-3.6), for example.
- a compound represented by (L-1-3.4) is preferable.
- the lower limit of the preferable content of the compound represented by the formula (L-1-3.4) or the formula (L-1-3.5) with respect to the total amount of the composition of the present invention is 3% by mass. Yes, 5% by mass, 7% by mass, 9% by mass, 11% by mass, 12% by mass, 13% by mass, 18% by mass and 21% by mass. Preferred upper limit values are 45%, 40% by mass, 35% by mass, 30% by mass, 25% by mass, 23% by mass, 20% by mass, and 18% by mass. Yes, 15% by mass, 13% by mass, 10% by mass, and 8% by mass.
- the total amount of the composition of the present invention is The lower limit of the preferred content of both compounds is 15% by weight, 19% by weight, 24% by weight, 30% by weight, and the preferred upper limit is 45% by weight, 40% by weight. 35% by mass, 30% by mass, 25% by mass, 23% by mass, 20% by mass, 18% by mass, 15% by mass and 13% by mass .
- the compound represented by the general formula (L-1-3) is preferably a compound represented by the formula (L-1-3.7) to the formula (L-1-3.10).
- a compound represented by the formula (L-1-3.9) is preferable.
- the compound represented by the general formula (L-1-4) is the following compound.
- R L141 and R L142 each independently represent the same meaning as R L1 and R L2 in General Formula (L).
- R L141 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms
- R L142 is an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or a carbon atom.
- An alkoxy group of 1 to 4 is preferable.
- the compound represented by the general formula (L-1-4) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the content of the compound represented by the general formula (L-1-4) is such that solubility at low temperature, transition temperature, electrical reliability, birefringence, process suitability, dropping It is necessary to adjust appropriately according to the required performance such as marks, image sticking, and dielectric anisotropy.
- the lower limit of the preferable content of the compound represented by the formula (L-1-4) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass 5% by weight, 7% by weight, 10% by weight, 14% by weight, 16% by weight, 20% by weight, 23% by weight, 26% by weight, 30% Mass%, 35 mass%, and 40 mass%.
- the upper limit of the preferable content of the compound represented by the formula (L-1-4) with respect to the total amount of the composition of the present invention is 50% by mass, 40% by mass, and 35% by mass. 30% by mass, 20% by mass, 15% by mass, 10% by mass, and 5% by mass.
- the compound represented by the general formula (L-1-4) is preferably a compound represented by the formula (L-1-4.1) or the formula (L-1-4.2).
- a compound represented by the formula (L-1-4.1) is preferable.
- the lower limit of the preferable content of these compounds with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, 5% by mass, and 7% by mass. is there.
- the upper limit of preferable content of these compounds is 20% by mass, 15% by mass, 13% by mass, 10% by mass, and 9% by mass.
- the compound represented by the general formula (L-1-4) is preferably a compound represented by the formula (L-1-4.3) or the formula (L-1-4.4).
- the lower limit of the preferable content of these compounds with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, 5% by mass, and 7% by mass. is there.
- the upper limit of preferable content of these compounds is 20% by mass, 15% by mass, 13% by mass, 10% by mass, and 9% by mass.
- the compound represented by the general formula (L-1-4) is a compound selected from the group of compounds represented by the formula (L-1-4.5) to the formula (L-1-4.7) And particularly preferred is a compound represented by formula (L-1-4.7).
- the lower limit of the preferable content of these compounds with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, 5% by mass, and 7% by mass. is there.
- the upper limit of preferable content of these compounds is 20% by mass, 15% by mass, 13% by mass, 10% by mass, and 9% by mass.
- the general formula (L) in addition to the compounds represented by the general formula (L-1-1) to the general formula (L-1-4), the general formula (L-2) to the general formula It is preferable to contain one or more compounds represented by (L-4).
- the compound represented by the general formula (L-2) is the following compound.
- R L21 and R L22 each independently represent the same meaning as R L1 and R L2 in the general formula (L).
- R L21 and R L22 are each independently preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms.
- the compound represented by the general formula (L-2) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (L-2) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, Mass%, 7 mass%, and 10 mass%.
- the upper limit of the preferable content is 20% by mass, 15%, 13% by mass, 10% by mass, 8% by mass, and 7% by mass with respect to the total amount of the composition of the present invention. %, 6% by mass, 5% by mass, and 3% by mass.
- the effect is high when the content is set to be large.
- the effect is high when the content is set low.
- the compound represented by the general formula (L-2) is preferably a compound selected from the group of compounds represented by the formula (L-2.1) to the formula (L-2.4)
- a compound represented by the formula (L-2.7) from (L-2.2) is preferable.
- the compound represented by the general formula (L-3) is the following compound.
- R L31 and R L32 each independently represent the same meaning as R L1 and R L2 in General Formula (L), and X L31 and X L32 each independently represent a hydrogen atom or a fluorine atom.
- R L31 and R L32 are each independently preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and one of X L31 and X L32 is a fluorine atom and the other is a hydrogen atom. Is preferred.
- the compound represented by the general formula (L-3) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (L-3) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, % By mass, 7% by mass, 10% by mass, 14% by mass, 16% by mass, 20% by mass, 23% by mass, 26% by mass, 30% by mass It is 35% by mass and 40% by mass.
- the upper limit of the preferable content of the compound represented by the formula (L-3) with respect to the total amount of the composition of the present invention is 50% by mass, 40% by mass, 35% by mass, 30% % By mass, 20% by mass, 15% by mass, 10% by mass, and 5% by mass.
- the compound represented by the general formula (L-3) is preferably a compound represented by the formula (L-3.1) to the formula (L-3.9).
- the types of compounds that can be combined there are no particular restrictions on the types of compounds that can be combined, but 1 to 3 types of these compounds are preferably contained, more preferably 1 to 4 types. Further, since the wide molecular weight distribution of the compound to be selected is also effective for solubility, for example, one type of the compound represented by the formula (L-3.1) or (L-3.2), the formula (L- 3.4) or one type from the compound represented by (L-6.5), one type from the compound represented by formula (L-3.6) or formula (L-3.7), It is preferable to select one compound from the compounds represented by -3.8) or (L-3.9) and combine them as appropriate. Among them, the formula (L-6.1), the formula (L-3.3), the formula (L-3.4), the formula (L-3.6), and the formula (L-3.9) are represented. It is preferable to include a compound.
- the compound represented by the general formula (L-3) is preferably, for example, a compound represented by the formula (L-3.10) to the formula (L-3.17).
- L-3.11) is preferred.
- the lower limit of the preferable content of these compounds with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, 5% by mass, and 7% by mass. is there.
- the upper limit of preferable content of these compounds is 20% by mass, 15% by mass, 13% by mass, 10% by mass, and 9% by mass.
- the compound represented by the general formula (L-4) is the following compound.
- R L41 and R L42 each independently represent the same meaning as R L1 and R L2 in Formula (L), A L41 and A L43 is A L2 and in the general formula (L) independently A L3 represents the same meaning, but the hydrogen atoms on A L41 and A L42 may each independently be substituted with a fluorine atom, Z L41 represents the same meaning as Z L1 in formula (L), X L41 and X L42 each independently represent a fluorine atom or a hydrogen atom.
- R L41 and R L42 are each independently preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 2 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and
- a L41 and A L42 Are each independently preferably a 1,4-cyclohexylene group or a 1,4-phenylene group, the hydrogen atoms on A L41 and A L42 may be each independently substituted with a fluorine atom, and
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, and four kinds.
- the lower limit of the preferable content of the compound represented by the formula (L-4) with respect to the total amount of the composition of the present invention is 1% by mass, 2% by mass, 3% by mass, % By mass, 7% by mass, 10% by mass, 14% by mass, 16% by mass, and 20% by mass.
- the upper limit of the preferable content of the compound represented by the formula (L-7) with respect to the total amount of the composition of the present invention is 30% by mass, 25% by mass, 23% by mass, % By mass, 18% by mass, 15% by mass, 10% by mass, and 5% by mass.
- the content of the compound represented by formula (L-7) is preferably increased, and when an embodiment with low viscosity is desired, the content is It is preferable to reduce the amount.
- the compound represented by the general formula (L-4) is preferably a compound represented by the formula (L-4.1) to the formula (L-4.4). It is preferable that it is a compound represented by 2).
- the compound represented by the general formula (L-4) is preferably a compound represented by the formula (L-4.11) to the formula (L-4.13). It is preferable that it is a compound represented by 11).
- the compound represented by the general formula (L-4) is a compound represented by the formula (L-4.21) to the formula (L-4.23).
- a compound represented by the formula (L-4.21) is preferable.
- the compound represented by the general formula (L-4) is preferably a compound represented by the formula (L-4.31) to the formula (L-4.34), and the formula (L-4. 31) or / and a compound represented by formula (L-4.32).
- the compound represented by the general formula (L-4) is preferably a compound represented by the formula (L-4.41) to the formula (L-4.44), and the formula (L-4. 41) or / and a compound represented by formula (L-4.42).
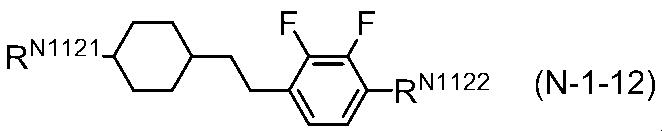
- the liquid crystal composition of the present invention includes, as a third component, one or two selected from compounds represented by general formula (N-1), general formula (N-2), and general formula (N-3) Contains the above compounds. These compounds correspond to dielectrically negative compounds (the sign of ⁇ is negative and the absolute value is greater than 2).
- R N11 , R N12 , R N21 , R N22 , R N31 and R N32 each independently represents an alkyl group having 1 to 8 carbon atoms, and one or non-adjacent 2 in the alkyl group
- Two or more —CH 2 — may be each independently substituted by —CH ⁇ CH—, —C ⁇ C—, —O—, —CO—, —COO— or —OCO—
- a N11 , A N12 , A N21 , A N22 , A N31 and A N32 are each independently (A) 1,4-cyclohexylene group (this is present in the group one -CH 2 - or nonadjacent two or more -CH 2 -.
- the group (a), the group (b) and the group (c) may be each independently substituted with a cyano group, a fluorine atom or a chlorine atom, Z N11 , Z N12 , Z N21 , Z N22 , Z N31 and Z N32 are each independently a single bond, —CH 2 CH 2 —, — (CH 2 ) 4 —, —OCH 2 —, —CH 2 O—.
- XN21 represents a hydrogen atom or a fluorine atom
- T N31 represents —CH 2 — or an oxygen atom
- n N11 , n N12 , n N21 , n N22 , n N31, and n N32 each independently represent an integer of 0 to 3, but n N11 + n N12 , n N21 + n N22, and n N31 + n N32 are each independently When there are a plurality of A N11 to A N32 and Z N11 to Z N32 , they may be the same or different.
- the compounds represented by general formula (N-1), general formula (N-2), and general formula (N-3) are preferably compounds in which ⁇ is negative and the absolute value is larger than 3.
- R N11 , R N12 , R N21 , R N22 , R N31, and R N32 each independently represent carbon.
- An alkyl group having 1 to 8 atoms, an alkoxy group having 1 to 8 carbon atoms, an alkenyl group having 2 to 8 carbon atoms or an alkenyloxy group having 2 to 8 carbon atoms is preferable, and an alkyl having 1 to 5 carbon atoms is preferred.
- An alkenyl group having 5 carbon atoms is more preferable, an alkyl group having 2 to 5 carbon atoms or an alkenyl group having 2 to 3 carbon atoms is further preferable, and an alkenyl group having 3 carbon atoms (propenyl group) is particularly preferable.
- the ring structure to which it is bonded is a phenyl group (aromatic)
- An alkenyl group having 4 to 5 atoms is preferable
- the ring structure to which the alkenyl group is bonded is a saturated ring structure such as cyclohexane, pyran and dioxane
- a straight-chain alkoxy group having 1 to 4 carbon atoms and a straight-chain alkenyl group having 2 to 5 carbon atoms are preferred.
- the total of carbon atoms and oxygen atoms, if present is preferably 5 or less, and is preferably linear.
- the alkenyl group is preferably selected from groups represented by any one of formula (R1) to formula (R5). (The black dots in each formula represent carbon atoms in the ring structure.)
- a N11 , A N12 , A N21 , A N22 , A N31, and A N32 are preferably aromatic when it is required to increase ⁇ n independently, and in order to improve the response speed, fat
- fat Preferably a trans-1,4-cyclohexylene group, 1,4-phenylene group, 2-fluoro-1,4-phenylene group, 3-fluoro-1,4-phenylene group, 3,5 -Difluoro-1,4-phenylene group, 2,3-difluoro-1,4-phenylene group, 1,4-cyclohexenylene group, 1,4-bicyclo [2.2.2] octylene group, piperidine-1 , 4-diyl group, naphthalene-2,6-diyl group, decahydronaphthalene-2,6-diyl group or 1,2,3,4-tetrahydronaphthalene-2,6-diyl group Preferred, it is more preferable that represents the following
- it represents a trans-1,4-cyclohexylene group or a 1,4-phenylene group.
- Z N11, Z N12, Z N21 , Z N22, Z N31 and Z N32 -CH 2 each independently O -, - CF 2 O - , - CH 2 CH 2 -, - CF 2 CF 2 - or a single bond preferably represents an, -CH 2 O -, - CH 2 CH 2 - or a single bond is more preferable, -CH 2 O-or a single bond is particularly preferred.
- XN21 is preferably a fluorine atom.
- T N31 is preferably an oxygen atom.
- n N11 + n N12 , n N21 + n N22 and n N31 + n N32 are preferably 1 or 2, a combination in which n N11 is 1 and n N12 is 0, a combination in which n N11 is 2 and n N12 is 0, n A combination in which N11 is 1 and n N12 is 1, a combination in which n N11 is 2 and n N12 is 1, a combination in which n N21 is 1 and n N22 is 0, n N21 is 2 and n N22 is n A combination in which n N31 is 1 and n N32 is 0, and a combination in which n N31 is 2 and n N32 is 0 are preferable.
- the lower limit of the preferable content of the compound represented by the general formula (N-1) with respect to the total amount of the composition of the present invention is 1% by mass, 10% by mass, 20% by mass, 30% by mass, 40% by mass, 50% by mass, 55% by mass, 60% by mass, 65% by mass, 70% by mass, 75% by mass, 80% by mass %.
- the upper limit of the preferable content is 95% by mass, 85% by mass, 75% by mass, 65% by mass, 55% by mass, 45% by mass, and 35% by mass, 25% by mass and 20% by mass.
- the lower limit of the preferable content of the compound represented by the general formula (N-2) with respect to the total amount of the composition of the present invention is 1% by mass, 10% by mass, 20% by mass, 30% by mass, 40% by mass, 50% by mass, 55% by mass, 60% by mass, 65% by mass, 70% by mass, 75% by mass, 80% by mass %.
- the upper limit of the preferable content is 95% by mass, 85% by mass, 75% by mass, 65% by mass, 55% by mass, 45% by mass, and 35% by mass, 25% by mass and 20% by mass.
- the lower limit of the preferable content of the compound represented by the general formula (N-3) with respect to the total amount of the composition of the present invention is 1% by mass, 10% by mass, 20% by mass, 30% by mass, 40% by mass, 50% by mass, 55% by mass, 60% by mass, 65% by mass, 70% by mass, 75% by mass, 80% by mass %.
- the upper limit of the preferable content is 95% by mass, 85% by mass, 75% by mass, 65% by mass, 55% by mass, 45% by mass, and 35% by mass, 25% by mass and 20% by mass.
- the above lower limit value is preferably low and the upper limit value is preferably low. Furthermore, when the composition of the present invention keeps Tni high and a composition having good temperature stability is required, the above lower limit value is preferably low and the upper limit value is preferably low. When it is desired to increase the dielectric anisotropy in order to keep the driving voltage low, it is preferable that the above lower limit value is increased and the upper limit value is high.
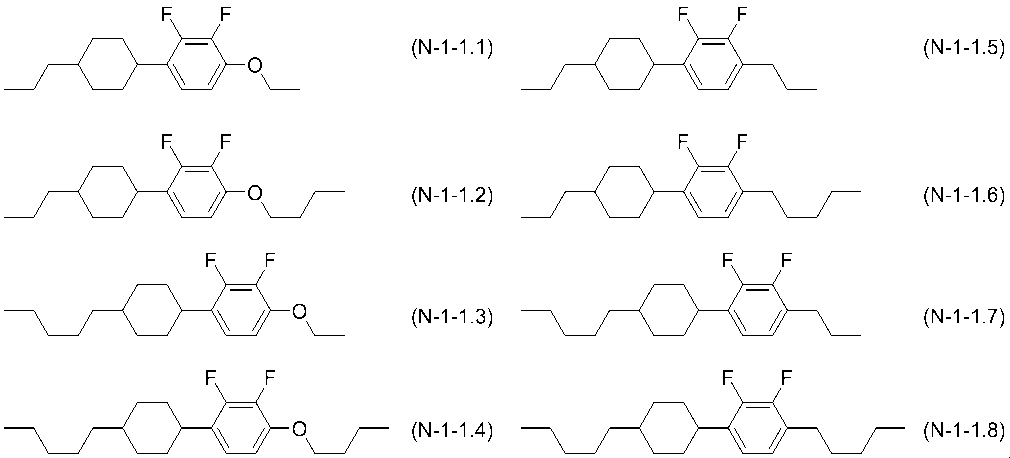
- the compounds represented by general formula (N-1) are represented by general formulas (N-1-1) to (N-1-6) and (N-1-10) to (N-1-20). A compound selected from the group of compounds is preferred.
- the compound represented by the general formula (N-1-1) is the following compound.
- R N111 and R N112 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- R N111 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably a propyl group or a pentyl group.
- RN112 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group or a butoxy group.
- the compound represented by the general formula (N-1-1) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-1) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15% by mass, 17% by mass, 20% by mass, 23% by mass, 25% by mass, 27% by mass, 30% by mass, 33% by mass, and 35% by mass. % By mass.
- the upper limit of the preferable content is 50% by mass, 40% by mass, 38% by mass, 35% by mass, 33% by mass, and 30% with respect to the total amount of the composition of the present invention. % By mass, 28% by mass, 25% by mass, 23% by mass, 20% by mass, 18% by mass, 15% by mass, 13% by mass, 10% by mass 8% by mass 7% by mass 6% by mass 5% by mass 3% by mass
- the compound represented by the general formula (N-1-1) is a compound selected from the group of compounds represented by the formula (N-1-1.1) to the formula (N-1-1.8).
- it is a compound represented by the formulas (N-1-1.1) to (N-1-1.4), and the formula (N-1-1.1) and the formula (N
- the compound represented by -1-1.3) is preferable.
- the compounds represented by the formulas (N-1-1.1) to (N-1-1.4) can be used alone or in combination.
- the lower limit of the preferable content of these compounds alone or with respect to the total amount is 5% by mass, 10% by mass, 13% by mass, 15% by mass, 17% by mass, and 20% by mass.
- the upper limit of the preferable content is 50% by mass, 40% by mass, 38% by mass, 35% by mass, 33% by mass, and 30% with respect to the total amount of the composition of the present invention.
- the compound represented by the general formula (N-1-2) is the following compound.
- R N121 and R N122 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- RN121 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group, a butyl group or a pentyl group.
- RN122 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and includes a methyl group, a propyl group, a methoxy group, an ethoxy group, or a propoxy group. preferable.
- the compound represented by the general formula (N-1-2) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-2) with respect to the total amount of the composition of the present invention is 5% by mass, 7% by mass, and 10% by mass. 13% by mass, 15% by mass, 17% by mass, 20% by mass, 23% by mass, 25% by mass, 27% by mass, 30% by mass, 33% by mass, % By mass, 35% by mass, 37% by mass, 40% by mass and 42% by mass.
- the upper limit of the preferable content is 50% by mass, 48% by mass, 45% by mass, 43% by mass, 40% by mass, and 38% by mass with respect to the total amount of the composition of the present invention.
- the compounds represented by the formula (N-1-2.1) to the formula (N-1-2.13) can be used alone or in combination.
- the lower limit of the preferable content of these compounds alone or with respect to the total amount of is 5% by mass, 10% by mass, 13% by mass, 15% by mass, 17% by mass, 20% % By mass, 23% by mass, 25% by mass, 27% by mass, 30% by mass, 33% by mass and 35% by mass.
- the upper limit of the preferable content is 50% by mass, 40% by mass, 38% by mass, 35% by mass, 33% by mass, and 30% with respect to the total amount of the composition of the present invention.
- the compound represented by the general formula (N-1-3) is the following compound.
- R N131 and R N132 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- R N131 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N132 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-3) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-3) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-3) is a compound selected from the group of compounds represented by the formula (N-1-3.1) to the formula (N-1-3.9).
- it is a compound represented by the formulas (N-1-3.1) to (N-1-3.7), and the formula (N-1-3.1) and the formula (N -1-3.2), formula (N-1-3.3), formula (N-1-3.4) and compounds represented by formula (N-1-3.6) are preferred.
- the compounds represented by formula (N-1-3.1) to formula (N-1-3.4) and formula (N-1-3.6) may be used alone or in combination. Is possible, but the combination of formula (N-1-3.1) and formula (N-1-3.2), formula (N-1-3.3), formula (N-1-3.4) ) And a combination of two or three selected from formula (N-1-3.6) are preferred.
- the compound represented by the general formula (N-1-4) is the following compound.
- R N141 and R N142 each independently represents the same meaning as R N11 and R N12 in the general formula (N-1).
- R N141 and R N142 are each independently an alkyl group having 1 to 5 carbon atoms, an alkenyl group or an alkoxy group having 1 to 4 carbon atoms carbon atoms 4-5 preferably a methyl group, a propyl group, an ethoxy Group or butoxy group is preferred.
- the compound represented by the general formula (N-1-4) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-4) with respect to the total amount of the composition of the present invention is 3% by mass, 5% by mass, and 7% by mass. 10% by mass, 13% by mass, 15% by mass, 17% by mass, and 20% by mass.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention.
- the compound represented by the general formula (N-1-4) is a compound selected from the group of compounds represented by the formula (N-1-4.1) to the formula (N-1-4.8).
- it is a compound represented by the formulas (N-1-4.1) to (N-1-4.4), and the formula (N-1-4.1) and the formula (N
- the compound represented by -1-4.2) is preferable.
- the compounds represented by formulas (N-1-4.1) to (N-1-4.4) can be used singly or in combination, but the compounds of the present invention
- the lower limit of the preferable content of these compounds alone or with respect to the total amount is 3% by mass, 5% by mass, 7% by mass, 10% by mass, 13% by mass, 15% by mass %, 17% by mass, and 20% by mass.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention.
- the compound represented by the general formula (N-1-5) is the following compound.
- R N151 and R N152 each independently represent the same meaning as R N11 and R N12 in formula (N-1).
- R N151 and R N152 are each independently an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethyl group, a propyl group, or a butyl group. Is preferred.
- the lower limit of the preferable content of the compound represented by the formula (N-1-5) with respect to the total amount of the composition of the present invention is 5% by mass, 8% by mass, and 10% by mass. 13 mass%, 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 33% by mass, 30% by mass, 28% by mass, and 25% by mass with respect to the total amount of the composition of the present invention. % By mass, 20% by mass, 18% by mass, 15% by mass, and 13% by mass.
- the compound represented by the general formula (N-1-5) is a compound selected from the group of compounds represented by the formula (N-1-5.1) to the formula (N-1-5.6). It is preferable that the compound represented by formula (N-1-5.2) and formula (N-1-5.4) is preferable.
- the compounds represented by formula (N-1-5.2) and formula (N-1-5.4) can be used alone or in combination, but the composition of the present invention
- the lower limit of the preferable content of these compounds alone or with respect to the total amount of is 5% by mass, 8% by mass, 10% by mass, 13% by mass, 15% by mass, 17 It is 20 mass%.
- the upper limit of the preferable content is 35% by mass, 33% by mass, 30% by mass, 28% by mass, and 25% by mass with respect to the total amount of the composition of the present invention.
- the compound represented by the general formula (N-1-6) is the following compound.
- R N161 and R N162 each independently represent the same meaning as R N11 and R N12 in formula (N-1).
- the compound represented by the general formula (N-1-6) is a compound represented by the following general formula (N-1-6-1) to general formula (N-1-6-14) preferable.
- the content of the compound represented by the general formula (N-1-6) is preferably 1% by mass as the lower limit, more preferably 2% by mass, and 15% as the upper limit. %, More preferably 12% by weight, still more preferably 10% by weight, particularly preferably 8% by weight, and most preferably 7% by weight.
- R N1101 and R N1102 each independently represent the same meaning as R N11 and R N12 in formula (N-1).
- R N1101 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1102 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-10) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-10) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-10) is a compound selected from the group of compounds represented by the formula (N-1-10.1) to the formula (N-1-10.10).
- it is a compound represented by the formulas (N-1-10.1) to (N-1-10.5), and the formula (N-1-10.1) and the formula (N
- the compound represented by (1-10.2) is preferable.
- the compounds represented by the formula (N-1-10.1) and the formula (N-1-10.2) can be used alone or in combination.
- the lower limit of the preferable content of these compounds alone or with respect to the total amount of is 5% by mass, 10% by mass, 13% by mass, 15% by mass, 17% by mass, 20% % By mass.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-11) is the following compound.
- R N1111 and R N1112 each independently represent the same meaning as R N11 and R N12 in formula (N-1).
- R N1111 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1112 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group or a butoxy group.
- the compound represented by the general formula (N-1-11) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-11) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-11) is a compound selected from the group of compounds represented by the formula (N-1-11.1) to the formula (N-1-11.10). It is preferably a compound represented by formulas (N-1-11.1) to (N-1-11.5), and preferably represented by formulas (N-1-11.2) and (N- The compound represented by 1-11.4) is preferable.
- the compounds represented by the formula (N-1-11.2) and the formula (N-1-11.4) can be used alone or in combination.
- the lower limit of the preferable content of these compounds alone or with respect to the total amount of is 5% by mass, 10% by mass, 13% by mass, 15% by mass, 17% by mass, 20% % By mass.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-12) is the following compound.
- R N1121 and R N1122 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- RN1121 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- RN1122 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-12) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-12) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-13) is the following compound.
- R N1131 and R N1132 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- R N1131 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1132 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-13) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-13) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-14) is the following compound.
- R N1141 and R N1142 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- R N1141 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1142 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-14) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-14) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-15) is the following compound.
- R N1151 and R N1152 each independently represent the same meaning as R N11 and R N12 in General Formula (N).
- RN1151 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1152 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-15) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-15) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-16) is the following compound.
- R N1161 and R N1162 each independently represent the same meaning as R N11 and R N12 in formula (N-1).
- R N1161 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1162 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-16) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-16) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-17) is the following compound.
- R N1171 and R N1172 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- RN1171 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1172 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-17) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-17) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-18) is the following compound.
- R N1181 and R N1182 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- RN1181 is preferably an alkyl group having 1 to 5 carbon atoms or an alkenyl group having 2 to 5 carbon atoms, and preferably an ethyl group, a propyl group or a butyl group.
- R N1182 is preferably an alkyl group having 1 to 5 carbon atoms, an alkenyl group having 4 to 5 carbon atoms, or an alkoxy group having 1 to 4 carbon atoms, and preferably an ethoxy group, a propoxy group, or a butoxy group.
- the compound represented by the general formula (N-1-18) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-18) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-19) is the following compound.
- R N1191 and R N1192 each independently represent the same meaning as R N11 and R N12 in formula (N-1)).
- the compound represented by the general formula (N-1-19) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-19) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-1-20) is the following compound.
- R N1201 and R N1202 each independently represent the same meaning as R N11 and R N12 in formula (N-1).
- the compound represented by the general formula (N-1-20) can be used alone, or two or more compounds can be used in combination. There are no particular restrictions on the types of compounds that can be combined, but they are used in appropriate combinations according to the required properties such as solubility at low temperatures, transition temperatures, electrical reliability, and birefringence.
- the kind of the compound used is, for example, one kind as one embodiment of the present invention, two kinds, three kinds, four kinds, and five kinds or more.
- the lower limit of the preferable content of the compound represented by the formula (N-1-20) with respect to the total amount of the composition of the present invention is 5% by mass, 10% by mass, and 13% by mass. 15 mass%, 17 mass%, and 20 mass%.
- the upper limit of the preferable content is 35% by mass, 30% by mass, 28% by mass, 25% by mass, 23% by mass, and 20% by mass with respect to the total amount of the composition of the present invention. Mass%, 18 mass%, 15 mass%, and 13 mass%.
- the compound represented by the general formula (N-2) is preferably a compound selected from the group of compounds represented by the general formulas (N-2-1) to (N-2-6).
- R N311 , R N321 , R N331 , and R N341 each independently represent the same meaning as R N11 in formula (N-3), and R N312 , R N322 , R N332 , and R N342 Each independently represents the same meaning as RN12 in formula (N).
- the liquid crystal composition of the present invention contains two or more compounds represented by the general formulas (N-1) to (N-3), it is represented by the general formulas (N-1) to (N-3). May contain two or more compounds selected from only one of the formulas, or two or more selected from the compounds represented by the general formulas (N-1) to (N-3) You may contain 2 or more types of compounds chosen from a formula.
- the liquid crystal composition of the present invention preferably contains one or more compounds represented by the general formula (N-1), and the general formula (N-1-1) to the general formula (N-1- 5), preferably containing one or more compounds represented by general formula (N-1-10), general formula (N-1-11) or general formula (N-1-19), It is more preferable to contain 2 to 10 species, which are used in combination of compounds represented by the general formula (N-1-1) to the general formula (N-1-5) or the general formula (N-1-10) And a combination of compounds represented by formula (N-1-11).
- the total content of the compounds represented by the general formula (N-1), the general formula (N-2), and the general formula (N-3) is preferably 1 to 90% by mass, and 10 to 90% by mass. %, More preferably 20% by mass to 80% by mass, further preferably 20% by mass to 70% by mass, further preferably 20% by mass to 60% by mass, and further preferably 25% by mass to 60% by mass. 30 mass% to 60 mass% is more preferable.
- the total amount of the compounds represented by the general formula (N-1), the general formula (N-2), and the general formula (N-3) is set as a lower limit in the composition.
- the content is preferably 1% by mass or more, preferably 10% by mass or more, preferably 20% by mass or more, preferably 25% by mass or more, and preferably 30% by mass or more.
- the upper limit is preferably 90% by mass or less, preferably 80% by mass or less, preferably 70% by mass or less, and preferably 60% by mass or less.
- composition of the present invention preferably does not contain a compound having a structure in which oxygen atoms such as a peracid (—CO—OO—) structure are bonded in the molecule.
- the content of the compound having a carbonyl group is preferably 5% by mass or less, preferably 3% by mass or less, based on the total mass of the composition. Is more preferable, and it is still more preferable to set it as 1 mass% or less, and it is most preferable not to contain substantially.
- the content of the compound substituted with chlorine atoms is preferably 15% by mass or less, and preferably 10% by mass or less, based on the total mass of the composition. 8% by mass or less, preferably 5% by mass or less, more preferably 3% by mass or less, and still more preferably substantially not contained.
- the content of a compound in which all the ring structures in the molecule are 6-membered rings is 80 It is preferably at least 90% by mass, more preferably at least 90% by mass, even more preferably at least 95% by mass, and it is composed of only a compound having substantially all 6-membered ring structures in the molecule. Most preferably it constitutes a product.
- the content of the compound having a cyclohexenylene group as a ring structure, and the content of the compound having a cyclohexenylene group as the total mass of the composition is preferably 10% by mass or less, preferably 8% by mass or less, more preferably 5% by mass or less, and preferably 3% by mass or less, and substantially not contained. Further preferred.
- the content of a compound having a 2-methylbenzene-1,4-diyl group in the molecule, in which a hydrogen atom may be substituted with a halogen may be reduced.
- the content of the compound having a 2-methylbenzene-1,4-diyl group in the molecule is preferably 10% by mass or less, more preferably 8% by mass or less based on the total mass of the composition. Is preferably 5% by mass or less, more preferably 3% by mass or less, and still more preferably substantially not contained.
- substantially not contained in the present application means that it is not contained except for an unintentionally contained product.
- the total amount of the compounds represented by the second component and the third component is 80% by mass to 99.9% by mass with respect to the total amount of the liquid crystal composition of the present invention. It is preferably 85% by mass to 99.9% by mass, preferably 90% by mass to 99.8% by mass, more preferably 90% by mass to 99.5% by mass, and 92% by mass.
- the content is preferably from 9% by mass to 99.5% by mass, and more preferably from 92% by mass to 99% by mass.
- the alkenyl group when the compound contained in the composition of the first embodiment of the present invention has an alkenyl group as a side chain, when the alkenyl group is bonded to cyclohexane, the alkenyl group has 2 to 5 carbon atoms.
- the alkenyl group is bonded to benzene, the number of carbon atoms of the alkenyl group is preferably 4 to 5, and the unsaturated bond of the alkenyl group and benzene are directly bonded. Preferably not.
- the value of the dielectric anisotropy ⁇ of the liquid crystal composition in the present invention has a negative dielectric anisotropy, and the absolute value of the dielectric anisotropy is 2 or more.
- the value of the dielectric anisotropy ⁇ is preferably ⁇ 2.0 to ⁇ 6.0 at 25 ° C., more preferably ⁇ 2.5 to ⁇ 5.0, and ⁇ 2.5 to It is particularly preferably ⁇ 4.5, but more specifically, ⁇ 2.5 to ⁇ 3.4 is preferable when the response speed is important, and ⁇ 3 when the drive voltage is important. It is preferably 4 to -4.5.
- the value of the refractive index anisotropy ⁇ n of the liquid crystal composition in the present invention is preferably 0.08 to 0.13 at 25 ° C., more preferably 0.09 to 0.12. More specifically, it is preferably 0.10 to 0.12 when corresponding to a thin cell gap, and preferably 0.08 to 0.10 when corresponding to a thick cell gap.
- the rotational viscosity ( ⁇ 1 ) of the liquid crystal composition in the present invention is preferably 150 or less, more preferably 130 or less, and particularly preferably 120 or less.
- Z as a function of rotational viscosity and refractive index anisotropy shows a specific value.
- ⁇ 1 represents rotational viscosity
- ⁇ n represents refractive index anisotropy.
- Z is preferably 13000 or less, more preferably 12000 or less, and particularly preferably 11000 or less.
- the nematic phase-isotropic liquid phase transition temperature (T ni ) of the liquid crystal composition in the present invention is 60 ° C. or higher, preferably 75 ° C. or higher, more preferably 80 ° C. or higher, and still more preferably 90 ° C. It is above °C.
- the liquid crystal composition of the present invention needs to have a specific resistance of 10 12 ( ⁇ ⁇ m) or more, preferably 10 13 ( ⁇ ⁇ m), and more preferably 10 14 ( ⁇ ⁇ m) or more.
- the liquid crystal composition of the present invention may contain a normal nematic liquid crystal, a smectic liquid crystal, a cholesteric liquid crystal, an antioxidant, an ultraviolet absorber, etc. in addition to the above-mentioned compounds.
- chemical stability it is preferable not to have a chlorine atom in the molecule.
- stability to light such as ultraviolet rays of the liquid crystal composition is required, conjugation represented by a naphthalene ring or the like. It is desirable that the molecule does not have a condensed ring having a long length and an absorption peak in the ultraviolet region.
- composition of the present invention may contain a polymerizable compound in order to produce a liquid crystal display element such as a PS mode, a lateral electric field type PSA mode, or a lateral electric field type PSVA mode.
- a polymerizable compound such as a PS mode, a lateral electric field type PSA mode, or a lateral electric field type PSVA mode.
- the polymerizable compound that can be used include a photopolymerizable monomer that undergoes polymerization by energy rays such as light.
- the structure has, for example, a liquid crystal skeleton in which a plurality of six-membered rings such as biphenyl derivatives and terphenyl derivatives are connected. Examples thereof include a polymerizable compound.
- polymerizable compound examples include the following general formula (X)
- X 21 and X 22 each independently represent a hydrogen atom or a methyl group
- Sp 21 and Sp 22 each independently represent a single bond, an alkylene group having 1 to 8 carbon atoms or —Y— (CH 2 ) s — (wherein s represents an integer of 2 to 7, Y represents O, OCOO, or COO, and Y is bonded to an aromatic ring present in U).
- U represents a linear or branched polyvalent alkylene group having 2 to 20 carbon atoms, a linear or branched polyvalent alkenylene group having 3 to 20 carbon atoms, or a polyvalent cyclic substituent having 5 to 30 carbon atoms.
- the alkylene group in the polyvalent alkylene group or the alkenylene group in the polyvalent alkenylene group may be substituted with —O—, —CO—, —CF 2 — within the range in which the oxygen atom is not adjacent, and has 5 carbon atoms.
- ⁇ 20 alkyl groups (alkylene in the group) The oxygen group may be substituted with an oxygen atom in the range where the oxygen atoms are not adjacent to each other)), or may be substituted with a cyclic substituent, and k represents an integer of 1 to 5.) It is preferable to use 1 type, or 2 or more types of an organic compound.
- X 21 and X 22 each independently represent a hydrogen atom or a methyl group.
- a hydrogen atom is preferable, and importance is placed on reducing the reaction residual amount.
- a methyl group is preferred.
- Sp 21 and Sp 22 are each independently a single bond, an alkylene group having 1 to 8 carbon atoms, or —Y— (CH 2 ) S — (wherein s is 2 to 7 Y represents O, OCOO, or COO, and Y is bonded to an aromatic ring present in U.), but the carbon chain is preferably not so long, and a single bond or An alkylene group having 1 to 5 carbon atoms is preferable, and a single bond or an alkylene group having 1 to 3 carbon atoms is more preferable.
- Sp 21 and Sp 22 represent —O— (CH 2 ) s —
- s is preferably 1 to 5, more preferably 1 to 3, and at least one of Sp 21 and Sp 22 is a single bond. More preferably, it is particularly preferable that both are single bonds.
- U is a linear or branched polyvalent alkylene group having 2 to 20 carbon atoms, a linear or branched polyvalent alkenylene group having 3 to 20 carbon atoms, or a polyvalent having 5 to 30 carbon atoms.
- An alkyl group (the alkylene group in the group may be substituted with an oxygen atom within the range where the oxygen atom is not adjacent), or may be substituted with a cyclic substituent, and may be substituted with two or more cyclic substituents; It is preferable.
- polymerizable compound is represented by the general formula (XX)
- X 201 and X 202 each independently represent a hydrogen atom or a methyl group
- Sp 201 and Sp 202 each independently represent a single bond, an alkylene group having 1 to 8 carbon atoms, or —O— (CH 2 ) s — (wherein s represents an integer of 2 to 7, Represents an aromatic ring).
- X 201 and X 202 are each preferably a diacrylate derivative that represents a hydrogen atom, or a dimethacrylate derivative that has a methyl group, and a compound in which one represents a hydrogen atom and the other represents a methyl group.
- the polymerization rate of these compounds is the fastest for diacrylate derivatives, slow for dimethacrylate derivatives, and intermediate for asymmetric compounds, and a preferred embodiment can be used depending on the application.
- a dimethacrylate derivative is particularly preferable.
- Sp 201 and Sp 202 each independently represent a single bond, an alkylene group having 1 to 8 carbon atoms, or —O— (CH 2 ) s —, but at least one of them is a single bond in a PSA display element.
- a compound in which both represent a single bond or one in which one represents a single bond and the other represents an alkylene group having 1 to 8 carbon atoms or —O— (CH 2 ) s — is preferable.
- 1 to 4 alkyl groups are preferable, and s is preferably 1 to 4.
- Z 201 represents —OCH 2 —, —CH 2 O—, —COO—, —OCO—, —CF 2 O—, —OCF 2 —, —CH 2 CH 2 —, —CF 2 CF 2 — or a single bond
- —COO—, —OCO— or a single bond is more preferred, and a single bond is particularly preferred.
- M 201 represents a 1,4-phenylene group, a trans-1,4-cyclohexylene group or a single bond in which any hydrogen atom may be substituted by a fluorine atom, but the 1,4-phenylene group or the single bond is preferable.
- C represents a ring structure other than a single bond
- Z 201 is preferably a linking group other than a single bond.
- M 201 is a single bond
- Z 201 is preferably a single bond.
- the ring structure between Sp 201 and Sp 202 is specifically preferably the structure described below.
- both ends shall be bonded to Sp 201 or Sp 202.
- the polymerizable compounds containing these skeletons are optimal for PSA-type liquid crystal display elements because of their alignment restriction power after polymerization, and a good alignment state is obtained, so that display unevenness is suppressed or does not occur at all.
- the following general formulas (XX-1) to (XX-4) are particularly preferable, and among them, the general formula (XX-2) is most preferable.
- the polymerizable compound-containing liquid crystal composition obtained by adding the polymerizable compound to the liquid crystal composition of the present invention has a low viscosity ( ⁇ ), a low rotational viscosity ( ⁇ 1 ), and a large elastic constant (K 33 ).
- ⁇ low viscosity
- ⁇ 1 low rotational viscosity
- K 33 large elastic constant
- the polymerizable compound is preferably contained in an amount of 0.01% by mass to 10% by mass, more preferably 0.01% by mass to 5% by mass with respect to the total mass of the liquid crystal composition, and 0.01% by mass. % To 2% by mass is more preferable, and 0.1% to 1% by mass is more preferable.
- the polymerization proceeds even in the absence of a polymerization initiator, but may contain a polymerization initiator in order to accelerate the polymerization.
- the polymerization initiator include benzoin ethers, benzophenones, acetophenones, benzyl ketals, acylphosphine oxides, and the like.
- composition in the present invention can further contain a compound represented by the general formula (Q).
- RQ represents a straight-chain alkyl group or a branched-chain alkyl group having 1 to 22 carbon atoms, and one or more CH 2 groups in the alkyl group are —O—so that oxygen atoms are not directly adjacent to each other.
- MQ represents a trans-1,4-cyclohexylene group, a 1,4
- the compound represented by the general formula (Q) is preferably a compound represented by the following general formula (Qa) to general formula (Qd).
- R Q1 is preferably a linear or branched alkyl group having 1 to 10 carbon atoms
- R Q2 is preferably a linear or branched alkyl group having 1 to 20 carbon atoms
- R Q3 is A straight-chain alkyl group having 1 to 8 carbon atoms, a branched-chain alkyl group, a straight-chain alkoxy group or a branched-chain alkoxy group is preferred
- L Q is preferably a straight-chain alkylene group or branched-chain alkylene group having 1 to 8 carbon atoms.
- compounds represented by general formula (Qa) to general formula (Qd) compounds represented by general formula (Qc) and general formula (Qd) are more preferable.
- the compound represented by the general formula (Q) preferably contains one or two compounds, more preferably contains one to five compounds, and the content thereof is a liquid crystal composition. It is preferably 0.001% by mass to 1% by mass, more preferably 0.001% by mass to 0.1% by mass, and particularly preferably 0.001% by mass to 0.05% by mass with respect to the total mass of .
- the polymerizable compound contained therein is polymerized by ultraviolet irradiation to impart liquid crystal alignment ability, and the amount of transmitted light is controlled using the birefringence of the composition.
- liquid crystal display elements As liquid crystal display elements, AM-LCD (active matrix liquid crystal display element), TN (nematic liquid crystal display element), STN-LCD (super twisted nematic liquid crystal display element), OCB-LCD and IPS-LCD (in-plane switching liquid crystal display element)
- AM-LCD active matrix liquid crystal display element
- TN nematic liquid crystal display element
- STN-LCD super twisted nematic liquid crystal display element
- OCB-LCD OCB-LCD
- IPS-LCD in-plane switching liquid crystal display element
- the two substrates of the liquid crystal cell used in the liquid crystal display element can be made of a transparent material having flexibility such as glass or plastic, and one of them can be an opaque material such as silicon.
- a transparent substrate having a transparent electrode layer can be obtained, for example, by sputtering indium tin oxide (ITO) on a transparent substrate such as a glass plate.
- the color filter can be prepared by, for example, a pigment dispersion method, a printing method, an electrodeposition method, or a dyeing method. A method for producing a color filter by a pigment dispersion method will be described as an example.
- a curable coloring composition for a color filter is applied on the transparent substrate, subjected to patterning treatment, and cured by heating or light irradiation.
- a pixel portion for a color filter can be created.
- a pixel electrode provided with an active element such as a TFT, a thin film diode, or a metal insulator metal specific resistance element may be provided on the substrate.
- the substrate is opposed so that the transparent electrode layer is on the inside.
- the thickness of the obtained light control layer is 1 to 100 ⁇ m. More preferably, the thickness is 1.5 to 10 ⁇ m.
- the polarizing plate it is preferable to adjust the product of the refractive index anisotropy ⁇ n of the liquid crystal and the cell thickness d so that the contrast is maximized.
- the polarizing axis of each polarizing plate can be adjusted so that the viewing angle and contrast are good.
- a retardation film for widening the viewing angle can also be used.
- the spacer examples include columnar spacers made of glass particles, plastic particles, alumina particles, a photoresist material, and the like. Thereafter, a sealant such as an epoxy thermosetting composition is screen-printed on the substrates with a liquid crystal inlet provided, the substrates are bonded together, and heated to thermally cure the sealant.
- a sealant such as an epoxy thermosetting composition is screen-printed on the substrates with a liquid crystal inlet provided, the substrates are bonded together, and heated to thermally cure the sealant.
- a normal vacuum injection method or an ODF method can be used as a method for sandwiching the polymerizable compound-containing composition between the two substrates.
- a display element manufactured using the ODF method In the ODF liquid crystal display device manufacturing process, a sealant such as epoxy photothermal combination curing is drawn on a backplane or frontplane substrate using a dispenser in a closed-loop bank shape, and then removed.
- a liquid crystal display element can be manufactured by bonding a front plane and a back plane after dropping a predetermined amount of the composition under air.
- the composition of the present invention can be suitably used because the composition can be stably dropped in the ODF process.
- an appropriate polymerization rate is desirable in order to obtain good alignment performance of liquid crystals. Therefore, active energy rays such as ultraviolet rays or electron beams are irradiated singly or in combination or sequentially.
- the method of polymerizing by is preferred.
- ultraviolet rays When ultraviolet rays are used, a polarized light source or a non-polarized light source may be used.
- the polymerization is carried out in a state where the polymerizable compound-containing composition is sandwiched between two substrates, at least the substrate on the irradiated surface side must be given adequate transparency to the active energy rays. Don't be.
- the orientation state of the unpolymerized part is changed by changing conditions such as an electric field, a magnetic field, or temperature, and further irradiation with active energy rays is performed. Then, it is possible to use a means for polymerization.
- a means for polymerization In particular, when ultraviolet exposure is performed, it is preferable to perform ultraviolet exposure while applying an alternating electric field to the polymerizable compound-containing composition.
- the alternating electric field to be applied is preferably an alternating current having a frequency of 10 Hz to 10 kHz, more preferably a frequency of 60 Hz to 10 kHz, and the voltage is selected depending on a desired pretilt angle of the liquid crystal display element.
- the pretilt angle of the liquid crystal display element can be controlled by the applied voltage.
- the pretilt angle is preferably controlled from 80 degrees to 89.9 degrees from the viewpoint of alignment stability and contrast.
- the temperature during irradiation is preferably within a temperature range in which the liquid crystal state of the composition of the present invention is maintained. Polymerization is preferably performed at a temperature close to room temperature, that is, typically at a temperature of 15 to 35 ° C.
- a lamp for generating ultraviolet rays a metal halide lamp, a high-pressure mercury lamp, an ultra-high pressure mercury lamp, or the like can be used.
- a wavelength of the ultraviolet-ray to irradiate it is preferable to irradiate the ultraviolet-ray of the wavelength range which is not the absorption wavelength range of a composition, and it is preferable to cut and use an ultraviolet-ray as needed.
- Intensity of ultraviolet irradiation is preferably from 0.1mW / cm 2 ⁇ 100W / cm 2, 2mW / cm 2 ⁇ 50W / cm 2 is more preferable.
- the amount of energy of ultraviolet rays to be irradiated can be adjusted as appropriate, but is preferably 10 mJ / cm 2 to 500 J / cm 2, and more preferably 100 mJ / cm 2 to 200 J / cm 2 .
- the intensity may be changed.
- the time for irradiating with ultraviolet rays is appropriately selected depending on the intensity of the irradiated ultraviolet rays, but is preferably from 10 seconds to 3600 seconds, and more preferably from 10 seconds to 600 seconds.
- the liquid crystal display device using the composition of the present invention is useful for achieving both high-speed response and suppression of display failure, and is particularly useful for a liquid crystal display device for active matrix driving.
- VA mode, PSVA mode, PSA It can be applied to a mode, IPS mode or ECB mode liquid crystal display element.
- n a natural number.
- the measured characteristics are as follows.
- T ni Nematic phase-isotropic liquid phase transition temperature (° C.) ⁇ n: Refractive index anisotropy at 20 ° C. ⁇ : Dielectric anisotropy at 20 ° C. ⁇ : Viscosity at 20 ° C. (mPa ⁇ s) ⁇ 1 : rotational viscosity at 20 ° C. (mPa ⁇ s) K 11 : elastic constant K 11 (pN) at 20 ° C. K 33 : Elastic constant at 20 ° C. K 33 (pN)
- Initial VHR VHR-1 (Toyo Technica), initial voltage holding ratio (%) at 60 ° C. under conditions of frequency 60 Hz and applied voltage 1 V 1 hour after 150 ° C.
- VHR 1 hour after heating at 150 ° C., voltage holding ratio (%) at 60 ° C. under the condition of frequency 60 Hz and applied voltage 1V
- Vsel 8 V
- Vnsel 1 V
- the evaluation of the burn-in of the liquid crystal display element is as follows. After the predetermined fixed pattern is displayed in the display area for 24 hours, the afterimage level of the fixed pattern when the display of the uniform halftone on the entire screen is performed is as follows. The four-step evaluation was performed.
- Examples 1 to 8 and Comparative Examples 1 to 8 are FFS mode liquid crystal display elements having a cell thickness of 3.0 ⁇ m.
- the following Example 9 to Example 16 and Comparative Example 9 to Comparative Example 16 were prepared, measured, and evaluated.
- a liquid crystal display element) was prepared and measured and evaluated.
- Examples 9 to 10 and Comparative Examples 9 to 10 when a pretilt angle was formed, a commonly used rubbing process was performed. Further, in Examples 11 to 16 and Comparative Examples 11 to 16, when the pretilt angle was formed, UV was irradiated at 60 J (365 nm) while applying a rectangular wave voltage of 10 V, 100 Hz. USHIO multi-light was used as the UV light source.
- polymerizable compounds As representative examples of polymerizable compounds, (Pb-1), (Pb-3), (Pa-31), (Pd-29) and (Pd-37) were used.
- the present invention is not limited to these polymerizable compounds.
- Examples 1 to 5 Comparative Examples 1 to 5-2
- LC-1 to LC-5 Examples 1 to 5
- LC-1 ′ to LC-5 ′′ Comparative Examples 1 to 5-2
- Table 1 The composition of the liquid crystal composition and its physical property values are shown in Table 1. The symbol on the left side of the content is a description of the abbreviation for the above compound.
- liquid crystal compositions LC-1, LC-2, LC-3, LC-4 and LC-5 of the present invention contain the compound represented by the general formula (i), represented compared to LC-1 is a liquid crystal composition containing no compounds' ⁇ LC-5 '' (Comparative example), gamma 1 is sufficiently small, because K 33 is large, it is indicative of the response speed gamma 1 Even when / K 33 showed a small value and the response speed was actually measured, the response speed was improved at the same ratio. Further, an FFS mode liquid crystal display element was manufactured using the liquid crystal compositions LC-1 to LC-5, and the voltage holding ratio, image sticking, transmittance, and solubility at low temperature were evaluated by the above-described methods. Excellent evaluation results were shown.
- the liquid crystal composition of the present invention has a nematic phase-isotropic liquid phase transition temperature (T ni ), refractive index anisotropy ( ⁇ n), dielectric anisotropy ( ⁇ ), rotational viscosity ( ⁇ 1 ), the elastic constant (K 33 ) and heat resistance are sufficiently excellent, and it has been confirmed that a liquid crystal display element such as an FFS mode using this has excellent display quality.
- T ni nematic phase-isotropic liquid phase transition temperature
- ⁇ n refractive index anisotropy
- ⁇ 1 dielectric anisotropy
- K 33 rotational viscosity
- heat resistance K 33
- LC-6 which is a liquid crystal composition of the present invention contains a compound represented by the general formula (i)
- LC-6 ′ which is a liquid crystal composition which does not contain the compound represented by the general formula (i) (Comparative example)
- K 33 is large, indicates an index in which gamma 1 / K 33 smaller value of the response speed, even if actually measured response speed, had improved response speed at a ratio of comparable .
- an FFS mode liquid crystal display element was prepared using the liquid crystal composition LC-6, and the voltage holding ratio, image sticking, transmittance, and solubility at low temperatures were evaluated by the above-described methods. showed that.
- LC-7 to LC-9 which are the liquid crystal compositions of the present invention, contain the compound represented by the general formula (i) and therefore do not contain the compound represented by the general formula (i).
- K 33 is large, so ⁇ 1 / K 33, which is an index of response speed, shows a small value. The response speed also improved with the ratio.
- an FFS mode liquid crystal display element is manufactured using the liquid crystal compositions LC-7 and LC-8, and a VA mode liquid crystal display element is manufactured using the LC-9. Evaluation of image sticking, transmittance, and solubility at low temperatures showed extremely excellent evaluation results.
- the liquid crystal composition of the present invention has a nematic phase-isotropic liquid phase transition temperature (T ni ), refractive index anisotropy ( ⁇ n), dielectric anisotropy ( ⁇ ), rotational viscosity ( ⁇ 1 ), the elastic constant (K 33 ) and heat resistance are sufficiently excellent, and it has been confirmed that liquid crystal display elements such as FFS mode and VA mode using this have excellent display quality.
- T ni nematic phase-isotropic liquid phase transition temperature
- ⁇ n refractive index anisotropy
- ⁇ 1 dielectric anisotropy
- K 33 rotational viscosity
- LC-10 which is a liquid crystal composition of the present invention contains a compound represented by general formula (i)
- LC-10 ′ which is a liquid crystal composition which does not contain a compound represented by general formula (i) Since ⁇ 1 is smaller and K 33 is larger than (Comparative Example), ⁇ 1 / K 33, which is an index of response speed, shows a small value, and the response speed is measured at the same ratio even when the response speed is measured. Was also improving.
- a VA mode liquid crystal display device was prepared using the liquid crystal composition LC-10, and the voltage holding ratio, image sticking, transmittance, and solubility at low temperatures were evaluated by the above-described methods. showed that.
- the liquid crystal composition of the present invention has a nematic phase-isotropic liquid phase transition temperature (T ni ), refractive index anisotropy ( ⁇ n), dielectric anisotropy ( ⁇ ), rotational viscosity ( ⁇ 1 ), the elastic constant (K 33 ) and heat resistance were sufficiently excellent, and it was confirmed that a liquid crystal display element such as a VA mode using the same has excellent display quality.
- T ni nematic phase-isotropic liquid phase transition temperature
- ⁇ n refractive index anisotropy
- ⁇ 1 dielectric anisotropy
- K 33 rotational viscosity
- K 33 the elastic constant
- MLC-1 ′ (Comparative Example 11)
- MLC-2 ′ (Comparative Example 12)
- MLC-3 ′ (Comparative Example 13)
- MLC-4 ′ (Comparative Example 14)
- MLC-5 ′ (Comparative Example 15)
- MLC-6 ′ (Comparative Example 16).
- the constitution and physical property evaluation results of the liquid crystal composition were as shown in Table 5 and Table 6.
- MLC-1 to MLC-6 which are liquid crystal compositions of the present invention, are provided with an appropriate pretilt angle of 85 to 88 ° and improved response speed, and liquid crystal display elements such as PSVA and PSA type using the same are displayed. It was confirmed that the quality was excellent.
Landscapes
- Chemical & Material Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Nonlinear Science (AREA)
- General Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Mathematical Physics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Liquid Crystal Substances (AREA)
Abstract
Description
第二成分として、一般式(L)
nL1は0、1、2又は3を表し、
AL1、AL2及びAL3はそれぞれ独立して
(a) 1,4-シクロヘキシレン基(この基中に存在する1個の-CH2-又は隣接していない2個以上の-CH2-は-O-に置き換えられてもよい。)及び
(b) 1,4-フェニレン基(この基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられてもよい。)
(c) (c)ナフタレン-2,6-ジイル基、1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基又はデカヒドロナフタレン-2,6-ジイル基(ナフタレン-2,6-ジイル基又は1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられても良い。)
からなる群より選ばれる基を表し、上記の基(a)、基(b)及び基(c)はそれぞれ独立してシアノ基、フッ素原子又は塩素原子で置換されていても良く、
ZL1及びZL2はそれぞれ独立して単結合、-CH2CH2-、-(CH2)4-、-OCH2-、-CH2O-、-COO-、-OCO-、-OCF2-、-CF2O-、-CH=N-N=CH-、-CH=CH-、-CF=CF-又は-C≡C-を表し、
nL1が2又は3であってAL2が複数存在する場合は、それらは同一であっても異なっていても良く、nL1が2又は3であってZL3が複数存在する場合は、それらは同一であっても異なっていても良いが、一般式(i)で表される化合物を除く。)で表される化合物から選ばれる1種又は2種以上の化合物を含有することを特徴とする液晶組成物を提供し、また、これを用いた液晶表示素子を提供する。
nL1は0、1、2又は3を表し、
AL1、AL2及びAL3はそれぞれ独立して
(a) 1,4-シクロヘキシレン基(この基中に存在する1個の-CH2-又は隣接していない2個以上の-CH2-は-O-に置き換えられてもよい。)及び
(b) 1,4-フェニレン基(この基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられてもよい。)
(c) ナフタレン-2,6-ジイル基、1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基又はデカヒドロナフタレン-2,6-ジイル基(ナフタレン-2,6-ジイル基又は1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられても良い。)
からなる群より選ばれる基を表し、上記の基(a)、基(b)及び基(c)はそれぞれ独立してシアノ基、フッ素原子又は塩素原子で置換されていても良く、
ZL1及びZL2はそれぞれ独立して単結合、-CH2CH2-、-(CH2)4-、-OCH2-、-CH2O-、-COO-、-OCO-、-OCF2-、-CF2O-、-CH=N-N=CH-、-CH=CH-、-CF=CF-又は-C≡C-を表し、
nL1が2又は3であってAL2が複数存在する場合は、それらは同一であっても異なっていても良く、nL1が2又は3であってZL3が複数存在する場合は、それらは同一であっても異なっていても良いが、一般式(i)で表される化合物を除く。
RL11及びRL12は、直鎖状の炭素原子数1~5のアルキル基、直鎖状の炭素原子数1~5のアルコキシ基及び直鎖状の炭素原子数2~5のアルケニル基が好ましい。
RL111及びRL112は、直鎖状の炭素原子数1~5のアルキル基、直鎖状の炭素原子数1~4のアルコキシ基及び直鎖状の炭素原子数2~5のアルケニル基が好ましい。
一般式(L-1-1-1)で表される化合物は、式(L-1-1-1.1)から式(L-1-1-1.3)で表される化合物群から選ばれる化合物であることが好ましく、式(L-1-1-1.2)又は式(L-1-1-1.3)で表される化合物であることが好ましく、特に、式(L-1-1-1.3)で表される化合物であることが好ましい。
本発明の組成物の総量に対しての式(L-1-1-2)で表される化合物の好ましい含有量の下限値は、1質量%であり、5質量%であり、10質量%であり、15質量%であり、17質量%であり、20質量%であり、23質量%であり、25質量%であり、27質量%であり、30質量%であり、35質量%である。好ましい含有量の上限値は、本発明の組成物の総量に対して、60質量%であり、55質量%であり、50質量%であり、45質量%であり、42質量%であり、40質量%であり、38質量%であり、35質量%であり、33質量%であり、30質量%である。
RL1131及びRL1132は、直鎖状の炭素原子数1~5のアルキル基、直鎖状の炭素原子数1~4のアルコキシ基及び直鎖状の炭素原子数2~5のアルケニル基が好ましい。
さらに、一般式(L-1-1-3)で表される化合物は、式(L-1-1-3.1)から式(L-1-1-3.12)で表される化合物群から選ばれる化合物であることが好ましく、式(L-1-1-3.1)、式(L-1-1-3.3)又は式(L-1-1-3.4)で表される化合物であることが好ましい。特に、式(L-1-1-3.1)で表される化合物は本発明の組成物の応答速度を特に改善するため好ましい。また、応答速度よりも高いTniを求めるときは、式(L-1-1-3.3)、式(L-1-1-3.4)、式(L-1-1-3.11)及び式(L-1-1-3.12)で表される化合物を用いることが好ましい。式(L-1-1-3.3)、式(L-1-1-3.4)、式(L-1-1-3.11)及び式(L-1-1-3.12)で表される化合物の合計の含有量は、低温での溶解度を良くするために20質量%以上にすることは好ましくない。
RL1142及びRL1152は、直鎖状の炭素原子数1~5のアルキル基、直鎖状の炭素原子数1~4のアルコキシ基及び直鎖状の炭素原子数2~5のアルケニル基が好ましい。
RL121は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、RL122は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましい。
RL131は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、RL132は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましい。)
一般式(L-1-3)で表される化合物は単独で使用することもできるが、2以上の化合物を組み合わせて使用することもできる。組み合わせることができる化合物の種類に特に制限は無いが、低温での溶解性、転移温度、電気的な信頼性、複屈折率などの求められる性能に応じて適宜組み合わせて使用する。使用する化合物の種類は、例えば本発明の一つの実施形態としては1種類であり、2種類であり、3種類であり、4種類であり、5種類以上である。
RL141は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、RL142は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましい。
RL21及びRL22はそれぞれ独立して炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましい。
RL31及びRL32はそれぞれ独立して炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、XL31及びXL32のうち一方がフッ素原子他方が水素原子であることが好ましい。
式中、RL41及びRL42はそれぞれ独立して炭素原子数1~5のアルキル基、炭素原子数2~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、AL41及びAL42はそれぞれ独立して1,4-シクロヘキシレン基又は1,4-フェニレン基が好ましく、AL41及びAL42上の水素原子はそれぞれ独立してフッ素原子によって置換されていてもよく、ZL41は単結合又はCOO-が好ましく、単結合が好ましく、XL41及びXL42は水素原子が好ましい。
AN11、AN12、AN21、AN22、AN31及びAN32はそれぞれ独立して、
(a) 1,4-シクロヘキシレン基(この基中に存在する1個の-CH2-又は隣接していない2個以上の-CH2-は-O-に置き換えられてもよい。)及び
(b) 1,4-フェニレン基(この基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられてもよい。)
(c) (c)ナフタレン-2,6-ジイル基、1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基又はデカヒドロナフタレン-2,6-ジイル基(ナフタレン-2,6-ジイル基又は1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられても良い。)
からなる群より選ばれる基を表し、上記の基(a)、基(b)及び基(c)はそれぞれ独立してシアノ基、フッ素原子又は塩素原子で置換されていても良く、
ZN11、ZN12、ZN21、ZN22、ZN31及びZN32はそれぞれ独立して単結合、-CH2CH2-、-(CH2)4-、-OCH2-、-CH2O-、-COO-、-OCO-、-OCF2-、-CF2O-、-CH=N-N=CH-、-CH=CH-、-CF=CF-又は-C≡C-を表し、
XN21は水素原子又はフッ素原子を表し、
TN31は-CH2-又は酸素原子を表し、
nN11、nN12、nN21、nN22、nN31及びnN32はそれぞれ独立して0~3の整数を表すが、nN11+nN12、nN21+nN22及びnN31+nN32はそれぞれ独立して1、2又は3であり、AN11~AN32、ZN11~ZN32が複数存在する場合は、それらは同一であっても異なっていても良い。)
一般式(N-1)、一般式(N-2)及び、一般式(N-3)で表される化合物は、Δεが負でその絶対値が3よりも大きな化合物であることが好ましい。
RN111は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、プロピル基又はペンチル基が好ましい。RN112は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基又はブトキシ基が好ましい。
RN121は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基、ブチル基又はペンチル基が好ましい。RN122は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、メチル基、プロピル基、メトキシ基、エトキシ基又はプロポキシ基が好ましい。
RN131は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN132は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN141及びRN142はそれぞれ独立して、炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、メチル基、プロピル基、エトキシ基又はブトキシ基が好ましい。
RN151及びRN152はそれぞれ独立して、炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましくエチル基、プロピル基又はブチル基が好ましい。
一般式(N-1-6)で表される化合物としては、下記一般式(N-1-6-1)~一般式(N-1-6-14)で表される化合物であることが好ましい。
RN1101は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1102は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1111は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1112は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1121は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1122は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1131は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1132は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1141は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1142は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1151は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1152は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1161は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1162は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1171は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1172は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
RN1181は炭素原子数1~5のアルキル基又は炭素原子数2~5のアルケニル基が好ましく、エチル基、プロピル基又はブチル基が好ましい。RN1182は炭素原子数1~5のアルキル基、炭素原子数4~5のアルケニル基又は炭素原子数1~4のアルコキシ基が好ましく、エトキシ基、プロポキシ基又はブトキシ基が好ましい。
一般式(N-1-19)で表される化合物は単独で使用することもできるが、2以上の化合物を組み合わせて使用することもできる。組み合わせることができる化合物の種類に特に制限は無いが、低温での溶解性、転移温度、電気的な信頼性、複屈折率などの求められる性能に応じて適宜組み合わせて使用する。使用する化合物の種類は、例えば本発明の一つの実施形態としては1種類であり、2種類であり、3種類であり、4種類であり、5種類以上である。
一般式(N-1-20)で表される化合物は単独で使用することもできるが、2以上の化合物を組み合わせて使用することもできる。組み合わせることができる化合物の種類に特に制限は無いが、低温での溶解性、転移温度、電気的な信頼性、複屈折率などの求められる性能に応じて適宜組み合わせて使用する。使用する化合物の種類は、例えば本発明の一つの実施形態としては1種類であり、2種類であり、3種類であり、4種類であり、5種類以上である。
一般式(N-3)で表される化合物は一般式(N-3-1)~(N-3-4)で表される化合物群から選ばれる化合物であることが好ましい。
本発明の液晶組成物において、一般式(N-1)~(N-3)で表される化合物を2種以上含有する場合は、一般式(N-1)~(N-3)で表される化合物のうちいずれかの式のみから選ばれる化合物を2種以上含有してもよいし、一般式(N-1)~(N-3)で表される化合物から選ばれる2つ以上の式から選ばれる化合物を2種以上含有してもよい。
Zは、13000以下が好ましく、12000以下がより好ましく、11000以下が特に好ましい。
Sp201及びSp202はそれぞれ独立して、単結合、炭素原子数1~8のアルキレン基又は-O-(CH2)s-(式中、sは2から7の整数を表し、酸素原子は芳香環に結合するものとする。)を表し、
Z201は-OCH2-、-CH2O-、-COO-、-OCO-、-CF2O-、-OCF2-、-CH2CH2-、-CF2CF2-、-CH=CH-COO-、-CH=CH-OCO-、-COO-CH=CH-、-OCO-CH=CH-、-COO-CH2CH2-、-OCO-CH2CH2-、-CH2CH2-COO-、-CH2CH2-OCO-、-COO-CH2-、-OCO-CH2-、-CH2-COO-、-CH2-OCO-、-CY1=CY2-(式中、Y1及びY2はそれぞれ独立して、フッ素原子又は水素原子を表す。)、-C≡C-又は単結合を表し、
M201は1,4-フェニレン基、トランスー1,4-シクロヘキシレン基又は単結合を表し、式中の全ての1,4-フェニレン基は、任意の水素原子がフッ素原子により置換されていても良い。)で表される二官能モノマーが好ましい。
これらの骨格を含む重合性化合物は重合後の配向規制力がPSA型液晶表示素子に最適であり、良好な配向状態が得られることから、表示ムラが抑制されるか、又は、全く発生しない。
本発明の液晶組成物に前記重合性化合物を含有させた重合性化合物含有液晶組成物は、低い粘度(η)、低い回転粘性(γ1)及び大きな弾性定数(K33)が得られるため、これを用いたPSAモード又はPSVAモードの液晶表示素子は表示ムラの抑制と高速応答の両立が実現できる。
RQは炭素原子数1から22の直鎖アルキル基又は分岐鎖アルキル基を表し、該アルキル基中の1つ又は2つ以上のCH2基は、酸素原子が直接隣接しないように、-O-、-CH=CH-、-CO-、-OCO-、-COO-、-C≡C-、-CF2O-、-OCF2-で置換されてよいが、炭素原子数1から10の直鎖アルキル基、直鎖アルコキシ基、1つのCH2基が-OCO-又は-COO-に置換された直鎖アルキル基、分岐鎖アルキル基、分岐アルコキシ基、1つのCH2基が-OCO-又は-COO-に置換された分岐鎖アルキル基が好ましく、炭素原子数1から20の直鎖アルキル基、1つのCH2基が-OCO-又は-COO-に置換された直鎖アルキル基、分岐鎖アルキル基、分岐アルコキシ基、1つのCH2基が-OCO-又は-COO-に置換された分岐鎖アルキル基が更に好ましい。MQはトランス-1,4-シクロへキシレン基、1,4-フェニレン基又は単結合を表すが、トランス-1,4-シクロへキシレン基又は1,4-フェニレン基が好ましい。
(側鎖)
-n -CnH2n+1 炭素原子数nの直鎖状のアルキル基
n- CnH2n+1- 炭素原子数nの直鎖状のアルキル基
-On -OCnH2n+1 炭素原子数nの直鎖状のアルコキシル基
nO- CnH2n+1O- 炭素原子数nの直鎖状のアルコキシル基
-V -CH=CH2
V- CH2=CH-
-V1 -CH=CH-CH3
1V- CH3-CH=CH-
-2V -CH2-CH2-CH=CH3
V2- CH3=CH-CH2-CH2-
-2V1 -CH2-CH2-CH=CH-CH3
1V2- CH3-CH=CH-CH2-CH2
(連結基)
-n- -CnH2n-
-nO- -CnH2n-O-
-On- -O-CnH2n-
-COO- -C(=O)-O-
-OCO- -O-C(=O)-
-CF2O- -CF2-O-
-OCF2- -O-CF2-
(環構造)
Δn :20℃における屈折率異方性
Δε :20℃における誘電率異方性
η :20℃における粘度(mPa・s)
γ1 :20℃における回転粘度(mPa・s)
K11 :20℃における弾性定数K11(pN)
K33 :20℃における弾性定数K33(pN)
初期VHR:VHR-1(東陽テクニカ)を用い、周波数60Hz,印加電圧1Vの条件下で60℃における初期の電圧保持率(%)
150℃1h後VHR:150℃で1h加熱後、周波数60Hz,印加電圧1Vの条件下で60℃における電圧保持率(%)
試料の応答速度を測定する場合、AUTRONIC-MELCHERS社のDMS301を用いて、Vsel=8V、Vnsel=1V、測定温度20℃で実施した。
〇:残像ごく僅かに有るも、許容できるレベル
△:残像有り、許容できないレベル
×:残像有り、かなり劣悪
透過率の評価は、LCD評価システムLCD-5200(大塚電子(株)製)を用いて、液晶表示素子の可視光透過率を測定した。
番号が同一な実施例と比較例との比較において、実施例に対して(実施例が基準となるため「-」と表記。)、
◎:透過率が高い
〇:透過率が同等
×:透過率が低い
低温での溶解性評価は、液晶組成物を調製後、2mLのサンプル瓶に液晶組成物を1g秤量し、これに温度制御式試験槽の中で、次を1サイクル「-25℃(1時間保持)→昇温(0.1℃/毎分)→0℃(1時間保持)→昇温(0.1℃/毎分)→20℃(1時間保持)→降温(-0.1℃/毎分)→0℃(1時間保持)→降温(-0.1℃/毎分)→-25℃」として温度変化を与え続け、目視にて液晶組成物からの析出物の発生を観察し、以下の4段階評価を行った。
また実施例11~実施例16、及び、比較例11~比較例16において、プレチルト角を形成させる場合は、10V、100Hz、矩形波電圧を印加しながら、UVを60J(365nm)照射した。UV光源としてUSHIO社のマルチライトを使用した。
LC-1~LC-5(実施例1~5)、LC-1’~LC-5’’(比較例1~比較例5-2)を調製した。液晶組成物の構成とその物性値は表1のとおりであった。尚、含有量の左側の記号は、上記化合物の略号の記載である。
(実施例6、比較例6)
LC-6(実施例6)、及びLC-6’(比較例6)を調製した。液晶組成物の構成とその物性値は表2のとおりであった。
(実施例7~9、比較例7~9)
LC-7~LC-9(実施例7~9)、LC-7’~LC-9’(比較例7~比較例9)を調製した。液晶組成物の構成とその物性値は表3のとおりであった。
(実施例10、比較例10)
LC-10(実施例10)、及びLC-10’(比較例10)を調製した。液晶組成物の構成とその物性値は表4のとおりであった。
(実施例11~実施例16、比較例11~比較例16)
MLC-1(実施例11)、MLC-2(実施例12)、MLC-3(実施例13)、MLC-4(実施例14)、MLC-5(実施例15)およびMLC-6(実施例16)を調製した。また、MLC-1’(比較例11)、MLC-2’(比較例12)、MLC-3’(比較例13)、MLC-4’(比較例14)、MLC-5’(比較例15)およびMLC-6’(比較例16)を調製した。液晶組成物の構成及び物性評価結果は表5及び表6のとおりであった。
Claims (8)
- 第一成分として、一般式(i)
第二成分として、一般式(L)
nL1は0、1、2又は3を表し、
AL1、AL2及びAL3はそれぞれ独立して
(a) 1,4-シクロヘキシレン基(この基中に存在する1個の-CH2-又は隣接していない2個以上の-CH2-は-O-に置き換えられてもよい。)及び
(b) 1,4-フェニレン基(この基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられてもよい。)
(c) (c)ナフタレン-2,6-ジイル基、1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基又はデカヒドロナフタレン-2,6-ジイル基(ナフタレン-2,6-ジイル基又は1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられても良い。)
からなる群より選ばれる基を表し、上記の基(a)、基(b)及び基(c)はそれぞれ独立してシアノ基、フッ素原子又は塩素原子で置換されていても良く、
ZL1及びZL2はそれぞれ独立して単結合、-CH2CH2-、-(CH2)4-、-OCH2-、-CH2O-、-COO-、-OCO-、-OCF2-、-CF2O-、-CH=N-N=CH-、-CH=CH-、-CF=CF-又は-C≡C-を表し、
nL1が2又は3であってAL2が複数存在する場合は、それらは同一であっても異なっていても良く、nL1が2又は3であってZL3が複数存在する場合は、それらは同一であっても異なっていても良いが、一般式(i)で表される化合物を除く。)で表される化合物から選ばれる1種又は2種以上の化合物を含有する、負の誘電率異方性を有する液晶組成物。 - 第三成分として、一般式(N-1)、一般式(N-2)、及び一般式(N-3)
AN11、AN12、AN21、AN22、AN31及びAN32はそれぞれ独立して、
(a) 1,4-シクロヘキシレン基(この基中に存在する1個の-CH2-又は隣接していない2個以上の-CH2-は-O-に置き換えられてもよい。)及び
(b) 1,4-フェニレン基(この基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられてもよい。)
(c) (c)ナフタレン-2,6-ジイル基、1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基又はデカヒドロナフタレン-2,6-ジイル基(ナフタレン-2,6-ジイル基又は1,2,3,4-テトラヒドロナフタレン-2,6-ジイル基中に存在する1個の-CH=又は隣接していない2個以上の-CH=は-N=に置き換えられても良い。)
からなる群より選ばれる基を表し、上記の基(a)、基(b)及び基(c)はそれぞれ独立してシアノ基、フッ素原子又は塩素原子で置換されていても良く、
ZN11、ZN12、ZN21、ZN22、ZN31及びZN32はそれぞれ独立して単結合、-CH2CH2-、-(CH2)4-、-OCH2-、-CH2O-、-COO-、-OCO-、-OCF2-、-CF2O-、-CH=N-N=CH-、-CH=CH-、-CF=CF-又は-C≡C-を表し、
XN21は水素原子又はフッ素原子を表し、
TN31は-CH2-又は酸素原子を表し、
nN11、nN12、nN21、nN22、nN31及びnN32はそれぞれ独立して0~3の整数を表すが、nN11+nN12、nN21+nN22及びnN31+nN32はそれぞれ独立して1、2又は3であり、AN11~AN32、ZN11~ZN32が複数存在する場合は、それらは同一であっても異なっていても良い。)から選ばれる化合物を1種又は2種以上の化合物を含有する、請求項1又は請求項2に記載の液晶組成物。 - 重合性化合物を1種又は2種以上含有する請求項1から3のいずれか1項に記載の液晶組成物。
- 重合性化合物が一般式(X)
- 請求項1から5のいずれか1項に記載の液晶組成物を用いた液晶表示素子。
- 請求項1から5のいずれか1項に記載の液晶組成物を用いたアクティブマトリックス駆動用液晶表示素子。
- 請求項1から5のいずれか1項に記載の液晶組成物を用いたVAモード、PSAモード、PSVAモード、IPSモード又はFFSモード用液晶表示素子。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/504,416 US10113115B2 (en) | 2014-09-05 | 2015-09-01 | Nematic liquid crystal composition and liquid crystal display device using the same |
| DE112015004038.2T DE112015004038T5 (de) | 2014-09-05 | 2015-09-01 | Nematische Flüssigkristallzusammensetzung und Flüssig-Kristallanzeige unter Verwendung derselben |
| CN201580002880.9A CN105814173A (zh) | 2014-09-05 | 2015-09-01 | 向列液晶组合物及使用其的液晶显示元件 |
| JP2015559067A JP5900718B1 (ja) | 2014-09-05 | 2015-09-01 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| KR1020167012921A KR101826643B1 (ko) | 2014-09-05 | 2015-09-01 | 네마틱 액정 조성물 및 이것을 사용한 액정 표시 소자 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014181213 | 2014-09-05 | ||
| JP2014-181213 | 2014-09-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016035786A1 true WO2016035786A1 (ja) | 2016-03-10 |
Family
ID=55439846
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/074835 WO2016035786A1 (ja) | 2014-09-05 | 2015-09-01 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10113115B2 (ja) |
| JP (1) | JP5900718B1 (ja) |
| KR (1) | KR101826643B1 (ja) |
| CN (1) | CN105814173A (ja) |
| DE (1) | DE112015004038T5 (ja) |
| TW (1) | TW201623585A (ja) |
| WO (1) | WO2016035786A1 (ja) |
Families Citing this family (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10351772B2 (en) * | 2014-05-13 | 2019-07-16 | Dic Corporation | Nematic liquid crystal composition and liquid crystal display element using same |
| CN107075375B (zh) | 2014-12-25 | 2020-11-13 | Dic株式会社 | 向列液晶组合物及使用其的液晶显示元件 |
| TWI724197B (zh) * | 2016-10-25 | 2021-04-11 | 日商迪愛生股份有限公司 | 液晶組成物及液晶顯示元件 |
| JP6380825B1 (ja) * | 2016-12-26 | 2018-08-29 | Dic株式会社 | 液晶組成物及び液晶表示素子 |
| TWI743350B (zh) * | 2017-04-21 | 2021-10-21 | 日商迪愛生股份有限公司 | 液晶組成物及液晶顯示元件 |
| KR20200041880A (ko) * | 2017-09-08 | 2020-04-22 | 디아이씨 가부시끼가이샤 | 배향 조제, 액정 조성물 및 액정 표시 소자 |
| CN115433589A (zh) * | 2017-09-30 | 2022-12-06 | 石家庄诚志永华显示材料有限公司 | 液晶组合物 |
| CN111164184A (zh) * | 2017-10-16 | 2020-05-15 | Dic株式会社 | 向列液晶组合物和使用其的液晶显示元件 |
| WO2020044833A1 (ja) * | 2018-08-28 | 2020-03-05 | Dic株式会社 | 液晶組成物 |
| WO2021002205A1 (ja) * | 2019-07-02 | 2021-01-07 | Dic株式会社 | 液晶組成物及び液晶表示素子 |
| JP2022076640A (ja) * | 2020-11-10 | 2022-05-20 | Dic株式会社 | 液晶組成物及び液晶表示素子 |
Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01240591A (ja) * | 1988-03-18 | 1989-09-26 | Seiko Epson Corp | 液晶組成物 |
| JPH01247482A (ja) * | 1988-03-29 | 1989-10-03 | Seiko Epson Corp | 液晶組成物 |
| JPH0222382A (ja) * | 1988-07-11 | 1990-01-25 | Seiko Epson Corp | 液晶組成物 |
| JPH0812605A (ja) * | 1994-04-28 | 1996-01-16 | Chisso Corp | 液晶性化合物、液晶組成物および液晶表示素子 |
| JP2012136623A (ja) * | 2010-12-27 | 2012-07-19 | Jnc Corp | 液晶組成物および液晶表示素子 |
| JP5333685B2 (ja) * | 2011-06-30 | 2013-11-06 | Dic株式会社 | 化合物、ポリマー、液晶配向層、液晶表示素子及び光学異方体 |
| JP2014043561A (ja) * | 2012-07-30 | 2014-03-13 | Jnc Corp | 液晶組成物、酸化防止剤および液晶表示素子 |
| JP2014141648A (ja) * | 2012-12-27 | 2014-08-07 | Jnc Corp | 液晶組成物、酸化防止剤および液晶表示素子 |
| JP2015157915A (ja) * | 2014-02-25 | 2015-09-03 | Jnc株式会社 | 液晶組成物および液晶表示素子 |
Family Cites Families (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3043033B2 (ja) | 1990-07-13 | 2000-05-22 | 沖電気工業株式会社 | 画像処理方法 |
| DE4027981A1 (de) | 1990-09-04 | 1992-04-30 | Merck Patent Gmbh | Matrix-fluessigkristallanzeige |
| TW304203B (ja) | 1994-04-28 | 1997-05-01 | Chisso Corp | |
| US5599480A (en) | 1994-07-28 | 1997-02-04 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Liquid-crystalline medium |
| JP5295471B2 (ja) | 2000-11-13 | 2013-09-18 | Dic株式会社 | 重合性液晶化合物、該化合物を含有する重合性液晶組成物及びその重合体 |
| JP4175826B2 (ja) | 2002-04-16 | 2008-11-05 | シャープ株式会社 | 液晶表示装置 |
| JP4691893B2 (ja) | 2004-03-24 | 2011-06-01 | Dic株式会社 | トリフルオロナフタレン誘導体を含有する液晶組成物と表示素子及び化合物。 |
| JP4883336B2 (ja) | 2004-07-30 | 2012-02-22 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| US20060238696A1 (en) | 2005-04-20 | 2006-10-26 | Chien-Hui Wen | Method of aligning negative dielectric anisotropic liquid crystals |
| JP2007002132A (ja) | 2005-06-24 | 2007-01-11 | Chisso Corp | 液晶組成物および液晶表示素子 |
| DE502006009061D1 (de) | 2005-08-09 | 2011-04-21 | Merck Patent Gmbh | Flüssigkristallines medium |
| ATE479735T1 (de) | 2006-10-12 | 2010-09-15 | Merck Patent Gmbh | Flüssigkristallanzeige |
| US8114310B2 (en) | 2007-10-22 | 2012-02-14 | Merck Patent Gmbh | Liquid-crystal display |
| US8496850B2 (en) | 2008-09-01 | 2013-07-30 | Jnc Corporation | Liquid crystal composition and liquid crystal display device |
| USRE46763E1 (en) | 2009-01-22 | 2018-03-27 | Jnc Corporation | Liquid crystal composition and liquid crystal display device |
| TWI458706B (zh) | 2009-05-11 | 2014-11-01 | Jnc Corp | 聚合性化合物及含有其之液晶組成物 |
| EP2292720A1 (en) | 2009-09-08 | 2011-03-09 | Merck Patent GmbH | Liquid-crystal display |
| TWI509056B (zh) | 2009-09-29 | 2015-11-21 | Jnc Corp | 液晶組成物及液晶顯示元件 |
| US8741397B2 (en) | 2009-11-09 | 2014-06-03 | Jnc Corporation | Liquid crystal display element, liquid crystal composition, aligning agent, method for producing liquid crystal display element, and use of liquid crystal composition |
| WO2011083677A1 (ja) | 2010-01-06 | 2011-07-14 | Jnc株式会社 | 液晶化合物、液晶組成物および液晶表示素子 |
| JP5725324B2 (ja) | 2010-03-31 | 2015-05-27 | Dic株式会社 | 不純物含有量を低減した液晶組成物 |
| JP5593890B2 (ja) | 2010-07-06 | 2014-09-24 | Dic株式会社 | 液晶組成物および液晶デバイス |
| DE102011105930A1 (de) | 2010-07-21 | 2012-01-26 | Merck Patent Gmbh | Polymerisierbare Mischungen und ihre Verwendung in Flüssigkristallanzeigen |
| JP5743132B2 (ja) | 2010-10-01 | 2015-07-01 | Dic株式会社 | 重合性化合物を含有する液晶組成物及びそれを使用した液晶表示素子 |
| JP5699518B2 (ja) | 2010-10-15 | 2015-04-15 | Dic株式会社 | 重合性化合物を含有する液晶組成物及びそれを使用した液晶表示素子 |
| JP5678587B2 (ja) | 2010-11-04 | 2015-03-04 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| US9080101B2 (en) | 2010-12-24 | 2015-07-14 | Dic Corporation | Polymerizable compound-containing liquid crystal composition and liquid crystal display device using the same |
| KR20240056652A (ko) | 2011-03-29 | 2024-04-30 | 메르크 파텐트 게엠베하 | 액정 매질 |
| JP5234227B2 (ja) | 2011-04-06 | 2013-07-10 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| WO2012144321A1 (ja) | 2011-04-18 | 2012-10-26 | Jnc株式会社 | 液晶組成物および液晶表示素子 |
| JP5834489B2 (ja) | 2011-05-18 | 2015-12-24 | Dic株式会社 | 重合性ナフタレン化合物 |
| JP5678798B2 (ja) | 2011-05-20 | 2015-03-04 | Dic株式会社 | 重合性化合物含有液晶組成物及びそれを使用した液晶表示素子 |
| JP6031781B2 (ja) | 2012-03-01 | 2016-11-24 | Dic株式会社 | 重合性化合物及びそれを用いた液晶組成物 |
| US9157027B2 (en) | 2012-04-24 | 2015-10-13 | Jnc Corporation | Compound having four polymerizable groups, liquid crystal composition and liquid crystal display device |
| US9150787B2 (en) | 2012-07-06 | 2015-10-06 | Jnc Corporation | Liquid crystal composition and liquid crystal display device |
| TWI565790B (zh) | 2012-08-08 | 2017-01-11 | 捷恩智股份有限公司 | 液晶組成物、液晶顯示元件及液晶組成物的使用 |
| CN102876338A (zh) | 2012-09-21 | 2013-01-16 | 深圳市华星光电技术有限公司 | 液晶介质混合物及使用其的液晶显示器 |
| US9127198B2 (en) | 2012-09-21 | 2015-09-08 | Shenzhen China Star Optoelectronics Technology Co., Ltd. | Mixture for liquid crystal medium and liquid crystal display using the same |
| TWI624531B (zh) * | 2013-03-06 | 2018-05-21 | Dainippon Ink & Chemicals | Nematic liquid crystal composition and liquid crystal display element using same |
| JP6153999B2 (ja) | 2013-03-21 | 2017-06-28 | Dic株式会社 | 重合性化合物含有液晶組成物及びこれを使用した液晶表示素子 |
| KR101957753B1 (ko) * | 2013-03-21 | 2019-03-14 | 디아이씨 가부시끼가이샤 | 중합성 화합물 함유 액정 조성물 및 그것을 사용한 액정 표시 소자 |
| WO2014203567A1 (ja) | 2013-06-17 | 2014-12-24 | Dic株式会社 | ネマチック液晶組成物及びこれを用いた液晶表示素子 |
| US10093857B2 (en) * | 2013-10-08 | 2018-10-09 | Dic Corporation | Nematic liquid crystal composition and liquid crystal display device using the same |
| CN109054861A (zh) * | 2014-01-06 | 2018-12-21 | Dic株式会社 | 向列液晶组合物及使用其的液晶显示元件 |
| EP3173457B1 (en) * | 2014-07-25 | 2019-06-05 | DIC Corporation | Liquid-crystal composition and liquid-crystal display element obtained using same |
| WO2016017519A1 (ja) * | 2014-07-29 | 2016-02-04 | Dic株式会社 | 液晶表示素子 |
| WO2016017521A1 (ja) * | 2014-07-29 | 2016-02-04 | Dic株式会社 | 液晶表示素子 |
-
2015
- 2015-09-01 US US15/504,416 patent/US10113115B2/en active Active
- 2015-09-01 WO PCT/JP2015/074835 patent/WO2016035786A1/ja active Application Filing
- 2015-09-01 JP JP2015559067A patent/JP5900718B1/ja active Active
- 2015-09-01 DE DE112015004038.2T patent/DE112015004038T5/de not_active Withdrawn
- 2015-09-01 CN CN201580002880.9A patent/CN105814173A/zh active Pending
- 2015-09-01 KR KR1020167012921A patent/KR101826643B1/ko active IP Right Grant
- 2015-09-04 TW TW104129321A patent/TW201623585A/zh unknown
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01240591A (ja) * | 1988-03-18 | 1989-09-26 | Seiko Epson Corp | 液晶組成物 |
| JPH01247482A (ja) * | 1988-03-29 | 1989-10-03 | Seiko Epson Corp | 液晶組成物 |
| JPH0222382A (ja) * | 1988-07-11 | 1990-01-25 | Seiko Epson Corp | 液晶組成物 |
| JPH0812605A (ja) * | 1994-04-28 | 1996-01-16 | Chisso Corp | 液晶性化合物、液晶組成物および液晶表示素子 |
| JP2012136623A (ja) * | 2010-12-27 | 2012-07-19 | Jnc Corp | 液晶組成物および液晶表示素子 |
| JP5333685B2 (ja) * | 2011-06-30 | 2013-11-06 | Dic株式会社 | 化合物、ポリマー、液晶配向層、液晶表示素子及び光学異方体 |
| JP2014043561A (ja) * | 2012-07-30 | 2014-03-13 | Jnc Corp | 液晶組成物、酸化防止剤および液晶表示素子 |
| JP2014141648A (ja) * | 2012-12-27 | 2014-08-07 | Jnc Corp | 液晶組成物、酸化防止剤および液晶表示素子 |
| JP2015157915A (ja) * | 2014-02-25 | 2015-09-03 | Jnc株式会社 | 液晶組成物および液晶表示素子 |
Also Published As
| Publication number | Publication date |
|---|---|
| US10113115B2 (en) | 2018-10-30 |
| JP5900718B1 (ja) | 2016-04-06 |
| CN105814173A (zh) | 2016-07-27 |
| TW201623585A (zh) | 2016-07-01 |
| JPWO2016035786A1 (ja) | 2017-04-27 |
| US20170253800A1 (en) | 2017-09-07 |
| KR101826643B1 (ko) | 2018-02-07 |
| DE112015004038T5 (de) | 2017-06-22 |
| KR20160074565A (ko) | 2016-06-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6760340B2 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| JP5900718B1 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| JP6245211B2 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| JP5987264B2 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| JP5561570B2 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| JP6206750B1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| JPWO2014136770A1 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| WO2013161096A1 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| JP5170602B1 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| WO2017188005A1 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| WO2014148197A1 (ja) | 重合性化合物含有液晶組成物及びこれを使用した液晶表示素子 | |
| JP6132122B2 (ja) | 液晶組成物及びこれを用いた液晶表示素子 | |
| JP5170603B1 (ja) | ネマチック液晶組成物及びこれを用いた液晶表示素子 | |
| WO2017098955A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| TW201425543A (zh) | 向列型液晶組成物及使用其之液晶顯示元件 | |
| TWI597352B (zh) | Nematic liquid crystal composition and liquid crystal display device using the same | |
| WO2015037519A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| WO2016052006A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| WO2016056314A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| WO2016098480A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| JP2017222749A (ja) | 液晶組成物、及び液晶表示素子 | |
| WO2016056313A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| WO2018207635A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| WO2016098478A1 (ja) | 組成物及びそれを使用した液晶表示素子 | |
| WO2016098479A1 (ja) | 組成物及びそれを使用した液晶表示素子 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2015559067 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15837257 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20167012921 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15504416 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 112015004038 Country of ref document: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 15837257 Country of ref document: EP Kind code of ref document: A1 |