WO2015182395A1 - 被覆銅粒子及びその製造方法 - Google Patents
被覆銅粒子及びその製造方法 Download PDFInfo
- Publication number
- WO2015182395A1 WO2015182395A1 PCT/JP2015/063880 JP2015063880W WO2015182395A1 WO 2015182395 A1 WO2015182395 A1 WO 2015182395A1 JP 2015063880 W JP2015063880 W JP 2015063880W WO 2015182395 A1 WO2015182395 A1 WO 2015182395A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- copper
- coated copper
- copper particles
- reaction
- solvent
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F1/00—Metallic powder; Treatment of metallic powder, e.g. to facilitate working or to improve properties
- B22F1/10—Metallic powder containing lubricating or binding agents; Metallic powder containing organic material
- B22F1/102—Metallic powder coated with organic material
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F9/00—Making metallic powder or suspensions thereof
- B22F9/16—Making metallic powder or suspensions thereof using chemical processes
- B22F9/18—Making metallic powder or suspensions thereof using chemical processes with reduction of metal compounds
- B22F9/24—Making metallic powder or suspensions thereof using chemical processes with reduction of metal compounds starting from liquid metal compounds, e.g. solutions
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F9/00—Making metallic powder or suspensions thereof
- B22F9/16—Making metallic powder or suspensions thereof using chemical processes
- B22F9/30—Making metallic powder or suspensions thereof using chemical processes with decomposition of metal compounds, e.g. by pyrolysis
- B22F9/305—Making metallic powder or suspensions thereof using chemical processes with decomposition of metal compounds, e.g. by pyrolysis of metal carbonyls
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B1/00—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors
- H01B1/20—Conductive material dispersed in non-conductive organic material
- H01B1/22—Conductive material dispersed in non-conductive organic material the conductive material comprising metals or alloys
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B13/00—Apparatus or processes specially adapted for manufacturing conductors or cables
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B5/00—Non-insulated conductors or conductive bodies characterised by their form
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F1/00—Metallic powder; Treatment of metallic powder, e.g. to facilitate working or to improve properties
- B22F1/05—Metallic powder characterised by the size or surface area of the particles
- B22F1/054—Nanosized particles
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F2301/00—Metallic composition of the powder or its coating
- B22F2301/10—Copper
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F9/00—Making metallic powder or suspensions thereof
- B22F9/16—Making metallic powder or suspensions thereof using chemical processes
- B22F9/30—Making metallic powder or suspensions thereof using chemical processes with decomposition of metal compounds, e.g. by pyrolysis
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/32—Hydrogen storage
Definitions
- the present invention relates to coated copper particles and a method for producing the same.
- Gold and silver which are noble metals, have characteristics that are relatively difficult to be oxidized. Therefore, when a metal fine particle dispersion is prepared, the contained metal fine particles should be maintained without forming an oxide film on the surface. Is easy.
- copper has a characteristic that it is relatively easily oxidized, and the tendency becomes more remarkable in relation to the size effect and the specific surface area particularly when the fine copper particles having a particle diameter of 200 nm or less are obtained. .
- the contained copper fine particles are in a state where the surface is covered with an oxide film in a short time, and the thickness of the oxide film increases with time, and the majority of the particle size of the copper fine particles Is often converted into an oxide film of copper oxide.
- the activity of the particle surface is in a very high state, and even in a method of heating and baking under an inert atmosphere such as nitrogen gas or under vacuum conditions, a trace amount present in the atmosphere Oxidation proceeds with oxygen, which may inhibit sintering of copper fine particles. Further, an increase in the oxide film during firing may cause volume shrinkage during reduction and reduction in firing density when reduction firing is performed using hydrogen gas or the like at the final stage of firing.
- the melting point drop due to the size effect is 1,064 ° C. as a simple substance when gold is taken as an example, but when the particle diameter is about 2 nm, it becomes about 300 ° C., and the melting point decreases to a temperature that can be used for electronic materials.
- the particle diameter exceeds 20 nm. Therefore, if the particle size is a single nano size of about 2 nm, a melting point drop can be sufficiently expected.
- a surface protective agent that prevents oxidation is essential.
- the required amount of the surface protective agent is several times the volume of copper, which causes a significant volume shrinkage during sintering, making it difficult to obtain a high-density sintered body.
- a method for controlling the generation rate of metal nuclei as a solute has been devised. For example, by gradually releasing substances necessary for particle growth from a reservoir (solid or metal chelate), the degree of supersaturation of the solution is controlled, and new nucleation during particle growth is suppressed. Only the nuclei produced at the very beginning after the grain growth period are grown, so that particles exhibiting monodispersity can be produced.
- a solid or complex compound with sufficiently low solubility or dissolution rate is selected as a method for selecting a reservoir for supplying a solute during particle growth.
- a technique for producing copper fine particles by thermally decomposing a complex compound derived from copper formate is known.
- the decomposition temperature of copper formate is about 220 ° C., but the decomposition temperature can be lowered by forming a complex structure.
- Japanese Patent Application Laid-Open No. 2011-032558 proposes a method for producing metal fine particles by thermal decomposition at 100 ° C. using a complex compound of an amino alcohol that functions as a bidentate ligand.
- Japanese Patent Application Laid-Open No. 2008-013466 or Japanese Patent Application Laid-Open No. 2008-031104 proposes a method for producing metal fine particles by thermal decomposition at 120 ° C. using a complex compound of an aliphatic amine functioning as a monodentate ligand. Yes.
- a method of controlling the particle size by limiting the metal nuclei incorporated into the growth nuclei in a micro reaction field using a surfactant has been devised.
- a method for producing metal or metal oxide fine particles by a reverse micelle method using nano-sized water droplets stably dispersed in an organic solvent by a surfactant as a reaction field has been proposed (for example, JP-A-08-143916). No., JP-A-2009-082828, and Japanese Patent No. 3900414).
- the decomposition temperature of the complex compound is too low, so that a large amount of metal nuclei are generated at an accelerated rate due to heat generated during decomposition, and the copper contained in the reaction solution Since the concentration is relatively high at 1.0 to 2.4 mol / L, particle growth is likely to occur due to the aggregation mechanism, and coarse particles are generated, so that the yield tends to be low. Further, in the technique described in Japanese Patent Application Laid-Open No. 2008-013466 or Japanese Patent Application Laid-Open No.
- the aliphatic amine constituting the copper formate complex simultaneously serves as a dispersion protective agent for metal fine particles, so that particle growth is prevented. It is difficult to produce, and it is difficult to produce copper particles having a particle size of 20 nm to submicron.
- a nucleation is generated to perform reduction while partially dissolving with an organic carboxylic acid using a solid copper compound such as copper oxide having low solubility as a reservoir.
- the speed is limited, and agglomeration at the nucleus growth stage is less likely to occur as compared with a dissolution system such as that disclosed in JP2011-032558A.
- the nucleation time is long, and the carbon chain of the carboxylic acid that is coated is short, so that sufficient interparticle repulsion cannot be obtained. Is difficult to produce, and the surface tends to be oxidized copper particles.
- the micelle is stabilized by using a large amount of a surfactant.
- the size of the reaction field is limited, it is difficult to produce particles having a size of 20 nm or more.
- the reverse micelle method is difficult to increase the concentration of the copper compound in the reaction solution, and is not suitable for producing a large amount of particles.
- the present invention solves the problems of the prior art, and is difficult to achieve with the prior art, coated copper particles having both excellent oxidation resistance and sinterability, low heat treatment temperature, low oxygen It aims at providing the manufacturing method which can obtain the covering copper particle in an environment.
- the present inventors have appropriately set the difference in SP value between the solvent contained in the reaction solution and the amino alcohol that is the complexing agent, so that a homogeneous system can be obtained at the initial stage of the reaction.
- the reaction system can be configured so as to form a two-layer separation structure in the middle of the reaction, whereby high-quality coated copper particles can be produced efficiently.
- the present invention includes the following aspects. (1) Obtaining a reaction liquid containing copper formate, amino alcohol, aliphatic carboxylic acid having an aliphatic group having 5 or more carbon atoms, and a solvent, and thermally decomposing a complex compound formed in the reaction liquid to obtain copper metal A ⁇ SP value that is a difference in SP value between an amino alcohol and a solvent is 4.2 or more, and a method for producing coated copper particles whose surface is coated with an aliphatic carboxylic acid. (2) The method for producing coated copper particles according to (1), wherein the amino alcohol has an SP value of 11.0 or more. (3) The method for producing coated copper particles according to (1) or (2), wherein the temperature of the thermal decomposition treatment is 100 ° C. to 130 ° C.
- the average primary particle diameter D SEM by SEM observation is 0.02 to 0.2 ⁇ m, and the fluctuation of the particle size distribution Coated copper particles having a coefficient (standard deviation SD / average primary particle diameter D SEM ) of 0.1 to 0.5.
- the coated copper particles have a D XRD / D SEM of 0.25 to 1.00.
- a conductive composition for screen printing comprising coated copper particles obtained by the method for producing coated copper particles according to any one of (1) to (6) and a medium.
- a conductive composition for inkjet printing comprising coated copper particles obtained by the method for producing coated copper particles according to any one of (1) to (6) and a medium.
- a circuit-formed product comprising a substrate and a wiring pattern that is a heat-treated product of the conductive composition according to (9) or (10) disposed on the substrate.
- a coated copper particle having both excellent oxidation resistance and sinterability, which has been difficult to achieve with the prior art, and a production method capable of obtaining the coated copper particle in a low heat treatment temperature and low oxygen environment can be provided.
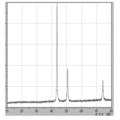
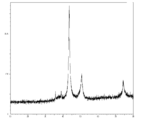
- FIG. 2 is TG-DTA analysis data of coated copper particles produced in Example 1.
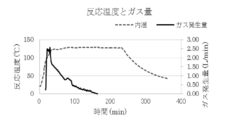
- FIG. 2 is a plot of the reaction temperature and total gas generation during the synthesis of Example 1.
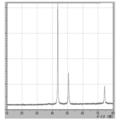
- 2 is FT-IR analysis data of a fraction that flows out during the synthesis of Example 1.
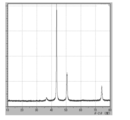
- 3 is XRD data of coated copper particles produced in Reference Example 2.
- 2 is an SEM observation image of coated copper particles produced in Example 1.
- FIG. 3 is an SEM observation image of coated copper particles produced in Example 2.
- 4 is a SEM observation image of coated copper particles produced in Example 4.
- 3 is an SEM observation image of coated copper particles produced in Comparative Example 1.
- 4 is an SEM observation image of coated copper particles produced in Comparative Example 2.
- 4 is a SEM observation image of coated copper particles produced in Comparative Example 3.
- 6 is a SEM observation image of coated copper particles produced in Comparative Example 4.
- 6 is a SEM observation image of coated copper particles produced in Comparative Example 5.
- 6 is a SEM observation image of coated copper particles produced in Example 5.
- 7 is an SEM observation image of coated copper particles produced in Example 6.
- FIG. 6 is an enlarged SEM observation image of coated copper particles produced in Example 6.
- FIG. 6 is an SEM observation image of coated copper particles produced in Example 7.
- 6 is an enlarged SEM observation image of coated copper particles produced in Example 7.
- FIG. It is a SEM observation image of the covering copper particle produced in Example 8.
- 10 is an enlarged SEM observation image of coated copper particles produced in Example 8.
- 10 is an SEM observation image of coated copper particles produced in Example 9.
- 10 is an enlarged SEM observation image of coated copper particles produced in Example 9.
- 2 is an enlarged SEM observation image of coated copper particles produced in Example 1.
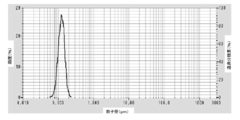
- FIG. It is a particle size distribution measurement data of the coated copper particles produced in Example 1.
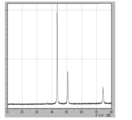
- 7 is XRD data of coated copper particles produced in Comparative Example 6.
- FIG. 7 is XRD data of coated copper particles produced in Comparative Example 7.
- FIG. 7 is an SEM observation image of coated copper particles produced in Comparative Example 6. It is a SEM observation image of the sintered film which paste-coated the copper particle produced by the comparative example 6, and baked it at 500 degreeC and 1 hour by nitrogen atmosphere. 2 is an SEM observation image of coated copper particles produced in Example 1.
- FIG. It is a SEM observation image of the sintered film which baked copper paste A which made the covering copper particle produced in Example 1 into paste at 350 degreeC for 1 hour in nitrogen atmosphere. It is a cross-sectional SEM image of a copper paste A baking film
- membrane. 10 is a SEM observation image of coated copper particles produced in Comparative Example 7.
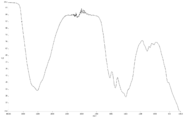
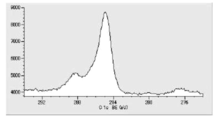
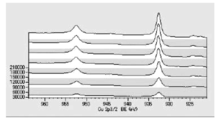
- FIG. 6 is a diagram showing carbon atoms in XPS outermost surface composition analysis data of Test Example 2.
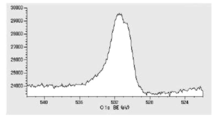
- FIG. 5 is a diagram showing oxygen atoms in XPS outermost surface composition analysis data of Test Example 2.
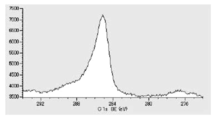
- FIG. 5 is a diagram showing copper atoms in XPS outermost surface composition analysis data of Test Example 2.
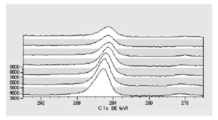
- FIG. 5 is a diagram showing carbon atoms in XPS outermost surface composition analysis data of Test Example 3.
- FIG. 5 is a diagram showing oxygen atoms in XPS outermost surface composition analysis data of Test Example 3.
- FIG. 5 is a diagram showing copper atoms in XPS outermost surface composition analysis data of Test Example 3.
- FIG. 6 is a graph showing the distribution of carbon atoms in the XPS-DepthProfile composition analysis data of Test Example 2.
- FIG. 6 is a diagram showing the distribution of oxygen atoms in the XPS-DepthProfile composition analysis data of Test Example 2.
- FIG. 6 is a diagram showing a distribution of copper atoms in XPS-DepthProfile composition analysis data of Test Example 2.
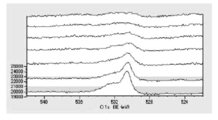
- FIG. 6 is a graph showing the distribution of carbon atoms in the XPS-DepthProfile composition analysis data of Test Example 3.
- 6 is a diagram showing the distribution of oxygen atoms in the XPS-DepthProfile composition analysis data of Test Example 3.
- 6 is a diagram showing a distribution of copper atoms in XPS-DepthProfile composition analysis data of Test Example 3.
- FIG. 6 is a diagram showing the distribution of oxygen atoms in the XPS-DepthProfile composition analysis data of Test Example 3.
- the term “process” is not limited to an independent process, and is included in the term if the intended purpose of the process is achieved even when it cannot be clearly distinguished from other processes.
- a numerical range indicated by using “to” indicates a range including the numerical values described before and after “to” as the minimum value and the maximum value, respectively.
- the content of each component in the composition means the total amount of the plurality of substances present in the composition unless there is a specific notice when there are a plurality of substances corresponding to each component in the composition.
- the method for producing coated copper particles of the present embodiment includes obtaining a reaction solution containing copper formate, amino alcohol, aliphatic carboxylic acid having an aliphatic group having 5 or more carbon atoms, and a solvent, and a complex formed in the reaction solution.
- the surface was coated with an aliphatic carboxylic acid having a ⁇ SP value of 4.2 or more, which is a difference in SP value between the aminoalcohol and the solvent. It is a manufacturing method of a covering copper particle.
- the thermal decomposition and reduction reaction of the copper formate complex proceeds in the liquid phase, and as the reaction proceeds, amino alcohol that is incompatible with this is released from the copper formate complex into the reaction solvent.
- a new reaction field similar to Water-in-Oil Emulsion is formed. Reduced copper particles with excellent oxidation resistance and sinterability, controlled particle size, and uniform particle size are generated by the progress of the nucleus growth reaction while continuously generating copper metal nuclei in the reaction field. It is thought that it is generated.
- supply of a solute is controlled by appropriately controlling the thermal decomposition rate of the copper formate complex. Thereby, it is considered that the growth of metal nuclei is controlled and reduced copper particles having a more uniform particle size are generated.
- the reduced copper particles produced by the physical adsorption are coated with high density.
- the coated copper particles produced in this way are composed of reduced copper particles with almost no oxide film, and the surface is coated with aliphatic carboxylic acid by physical adsorption, so it has an excellent balance between oxidation resistance and sinterability. It is thought that there is.
- covers the copper particle is removed at the temperature of 400 degrees C or less.
- the coated copper particles can be sintered in a low oxygen atmosphere that can be achieved by means such as nitrogen substitution. Therefore, with conventional copper particles that require a reducing atmosphere for sintering, they are also effectively used for sites that have been difficult to apply, for example, sites where hydrogen embrittlement or alteration due to reaction with hydrogen becomes a problem. be able to. Moreover, since it can sinter using existing facilities, such as a nitrogen substitution reflow furnace, it is excellent also in the point of economical efficiency.
- the reaction solution used in the method for producing the coated copper particles of the present embodiment includes copper formate, at least one amino alcohol, and at least one aliphatic carboxylic acid having an aliphatic group having 5 or more carbon atoms, And a solvent.
- the reaction solution may further contain other additives as required.
- Copper formate is composed of divalent copper ions and 2 moles of formate ions per 1 mole of copper ions. Copper formate may be anhydrous or hydrated. In addition, copper formate may be a commercially available product or a newly prepared one. A method for thermally reducing copper formate to obtain fine particles of reduced copper is disclosed in, for example, Japanese Patent Publication No. 61-19682. Formic acid is different from ordinary carboxylic acid and has reducibility. Therefore, when copper formate is thermally decomposed, divalent copper ions can be reduced. For example, anhydrous copper formate is known to thermally decompose at 210 ° C. to 250 ° C. to produce metallic copper when heated in an inert gas.
- the content of copper formate in the reaction solution is not particularly limited and can be appropriately selected according to the purpose and the like.
- the content of copper formate in the reaction solution is preferably 1.0 to 2.5 mol / liter, more preferably 1.5 to 2.5 mol / liter, for example, from the viewpoint of production efficiency. It is particularly preferably 2.0 to 2.5 mol / liter.
- the amino alcohol is not particularly limited as long as it is an alcohol compound having at least one amino group and can form a complex compound with copper formate. Due to the presence of amino alcohol in the reaction solution, a complex compound can be formed from copper formate and solubilized in a solvent.
- the amino alcohol is preferably a monoamino monoalcohol compound, and more preferably a monoamino monoalcohol compound in which the amino group is unsubstituted.
- the amino alcohol is also preferably a monodentate monoamino monoalcohol compound.
- the boiling point of amino alcohol is not particularly limited, but is preferably higher than the reaction temperature of the thermal decomposition treatment. Specifically, the boiling point of amino alcohol is preferably 120 ° C. or higher, and more preferably 130 ° C. or higher.
- the upper limit of the boiling point is not particularly limited, and is, for example, 400 ° C. or lower, preferably 300 ° C. or lower.
- the amino alcohol has an SP value of preferably 11.0 or more, more preferably 12.0 or more, and further preferably 13.0 or more from the viewpoint of polarity.
- the upper limit of the SP value of aminoalcohol is not particularly limited, and is, for example, 18.0 or less, preferably 17.0 or less.
- the SP value in this specification is based on the definition of Hildebrand. According to the report, the SP value is the square root of intermolecular bonding energy E 1 per sample 1mL at 25 ° C..
- the SP value was calculated by the method described in the “Japan Petroleum Institute website” (http://sekiyu-gakkai.or.jp/jp/dictionary/petdicsolvent.html#solubility2). Specifically, it is calculated as follows.
- Intermolecular bonding energy E 1 is a value obtained by subtracting the gas energy from the latent heat of vaporization.
- the latent heat of vaporization Hb is given by the following equation as the boiling point Tb of the sample.
- Hb 21 ⁇ (273 + Tb) From this Hb value, the molar latent heat of vaporization H 25 at 25 ° C. is obtained by the following equation.
- H 25 Hb ⁇ [1 + 0.175 ⁇ (Tb ⁇ 25) / 100]
- Molar latent heat of vaporization H 25 intermolecular bonding energy E from is determined by the following equation.
- E H 25 -596
- Intermolecular bonding energy E 1 per sample 1mL from intermolecular bonding energy E is calculated by the following equation.
- E 1 E ⁇ D / Mw
- D the density of the sample
- Mw the molecular weight of the sample
- SP value is obtained from E 1 by the following equation.
- SP (E 1 ) 1/2 Note that a solvent containing OH groups needs to be corrected by +1 for each OH group. [For example, Mitsubishi Oil Technology, No. 42, p3, p11 (1989)]
- amino alcohols include 2-aminoethanol (boiling point: 170 ° C., SP value: 14.54), 3-amino-1-propanol (boiling point: 187 ° C., SP value: 13.45), 5-amino -1-pentanol (boiling point: 245 ° C., SP value: 12.78), DL-1-amino-2-propanol (boiling point: 160 ° C., SP value: 12.74), N-methyldiethanolamine (boiling point: 247) C., SP value: 13.26) and the like are exemplified, and at least one selected from the group consisting of these is preferable.
- Amino alcohols may be used singly or in combination of two or more.
- the content of amino alcohol in the reaction solution is not particularly limited and can be appropriately selected according to the purpose and the like.
- the content of amino alcohol is, for example, preferably in the range of 1.5 to 4.0 times mol, more preferably in the range of 1.5 to 3.0 times mol with respect to the copper ions in the reaction solution.
- the content of amino alcohol is 1.5 times mol or more with respect to copper ions, sufficient solubility of copper formate can be obtained, and the time required for the reaction can be shortened.
- the contamination of the produced coated copper particles can be suppressed when the amount is 4.0 times mol or less.
- the aliphatic carboxylic acid is not particularly limited as long as it is a long-chain aliphatic carboxylic acid having an aliphatic group having 5 or more carbon atoms.
- the aliphatic group may be linear or branched, and may be either a saturated aliphatic group or an unsaturated aliphatic group.
- the aliphatic group has 5 or more carbon atoms, preferably 5 or more and 17 or less, and more preferably 7 or more and 17 or less. When the aliphatic group has 5 or more carbon atoms, the variation rate that serves as an index of the particle size distribution tends to be small.
- carboxylic acids having a long carbon chain have a strong associative power and contribute to stabilization of the phase similar to the water-in-oil emulsion which is a micro reaction field, so that copper particles having a uniform particle diameter can be efficiently produced. It is done.
- the boiling point of the aliphatic carboxylic acid is preferably higher than the temperature of the thermal decomposition treatment.
- the boiling point of the aliphatic carboxylic acid is preferably 120 ° C. or higher, and more preferably 130 ° C. or higher.
- the upper limit of the boiling point is not particularly limited, and is, for example, 400 ° C. or lower. There exists a tendency for the sinterability of a covering copper particle to improve more that a boiling point is 400 degrees C or less.
- aliphatic carboxylic acid examples include oleic acid, linoleic acid, stearic acid, heptadecanoic acid, lauric acid, and octanoic acid, and are preferably at least one selected from the group consisting of these.
- the aliphatic carboxylic acid may be used alone or in combination of two or more.
- the content of the aliphatic carboxylic acid in the reaction solution is not particularly limited and can be appropriately selected according to the purpose and the like.
- the content of the aliphatic carboxylic acid is, for example, preferably in the range of 2.5 to 25 mol%, more preferably in the range of 5.0 to 15 mol% with respect to the copper ions in the reaction solution. There exists a tendency which can suppress the viscosity raise of a reaction system as content of aliphatic carboxylic acid is 25 mol% or less with respect to a copper ion.
- the solvent constituting the reaction solution is not particularly limited as long as it is selected so that the reduction reaction with formic acid is not excessively inhibited and the ⁇ SP value, which is the difference in SP value from amino alcohol, is 4.2 or more. It can select suitably from the organic solvent used.
- the ⁇ SP value which is the difference between the SP value of the amino alcohol and the SP value of the solvent, is 4.2 or more, coated copper particles having a narrow particle size distribution and a uniform particle diameter are obtained. It is done.
- the ⁇ SP value is 4.2 or more, preferably 4.5 or more, more preferably 5.0 or more, from the viewpoint of the reaction field formability and the quality of the coated copper particles.
- the upper limit of the ⁇ SP value is not particularly limited.
- the ⁇ SP value is 11.0 or less, and preferably 10.0 or less.
- the SP value of the solvent is selected so that the ⁇ SP value is 4.2 or more, but the SP value of the solvent is preferably smaller than the SP value of amino alcohol.
- the SP value of the solvent is preferably 11.0 or less, and more preferably 10.0 or less.
- the lower limit of the SP value of the solvent is not particularly limited.
- the SP value of the solvent is preferably 7.0 or more.
- the boiling point of a solvent is higher than the temperature of a thermal decomposition process.
- the boiling point of the solvent is preferably 120 ° C. or higher, and more preferably 130 ° C. or higher.
- the upper limit of the boiling point is not particularly limited, and for example, the boiling point is 400 ° C. or lower, and preferably 300 ° C. or lower.
- the solvent is also preferably an organic solvent capable of forming an azeotrope with water. If an azeotropic mixture with water can be formed, the water generated in the reaction solution by the thermal decomposition treatment can be easily removed from the reaction system.
- the solvent examples include ethylcyclohexane (boiling point: 132 ° C., SP value: 8.18), C9 cyclohexane [manufactured by Maruzen Petroleum, trade name: Swaclean # 150] (boiling point: 149 ° C., SP value). : 799), n-octane (boiling point: 125 ° C., SP value: 7.54) and the like, and at least one selected from the group consisting of these is preferable.
- the solvent may be used alone or in combination of two or more.
- the solvent is a combination of two or more, it is preferable to include a main solvent that is incompatible with amino alcohol and an auxiliary solvent that is compatible with amino alcohol.
- the main solvent are as described above.
- a preferred embodiment of the boiling point of the auxiliary solvent is the same as that of the main solvent.
- the SP value of the auxiliary solvent is preferably larger than that of the main solvent, and more preferably large enough to be compatible with amino alcohol.
- the auxiliary solvent include glycol ethers such as EO glycol ether, PO glycol ether, and dialkyl glycol ether.
- EO glycol ethers such as methyl diglycol, isopropyl glycol, butyl glycol
- PO glycol ethers such as methyl propylene diglycol, methyl propylene triglycol, propyl propylene glycol, butyl propylene glycol, dimethyl diglycol, etc. And at least one selected from the group consisting of these is preferred.
- cosolvents are all available from Nippon Emulsifier Co., Ltd.
- the SP value of the solvent is calculated as an average SP value considering the SP value and molar volume of each solvent contained in the solvent.
- the average SP value is calculated by the following equation when the solvent is composed of two kinds of solvent 1 and solvent 2.
- ⁇ 3 [V 1 ⁇ ⁇ 1 + V 2 ⁇ ⁇ 2 ] / (V 1 + V 2 ) ⁇ 3 : average SP value of mixed solvent, ⁇ 1 : SP value of solvent 1, V 1 : molar volume of solvent 1, ⁇ 2 : SP value of solvent 2 V 2 : molar volume of solvent 2
- the amount of the solvent contained in the reaction solution is preferably selected so that the concentration of copper ions is 1.0 to 2.5 mol / liter, preferably 1.5 to 2.5 mol / liter. More preferably.
- the copper ion concentration in the reaction solution is 1.0 mol / liter or more, productivity is further improved, and when it is 2.5 mol / liter or less, an increase in the viscosity of the reaction solution is suppressed, and good stirring is achieved. Sex is obtained.
- a complex compound derived from copper formate is generated.
- the structure of a complex compound is not specifically limited, It may consist of 1 type and may contain 2 or more types.
- the structure of the complex compound may change as the thermal decomposition treatment proceeds. That is, the complex compound mainly present in the early stage of the thermal decomposition treatment and the complex compound mainly present in the later stage of the thermal decomposition treatment may have different configurations.
- the complex compound produced in the reaction solution is a complex compound in which two molecules of formate ion and two molecules of amino alcohol are coordinated to one copper ion, and one molecule of formate ion to one copper ion. And a complex compound in which one molecule of an aliphatic carboxylic acid and two molecules of an amino alcohol are coordinated.
- the complex compound produced in the reaction solution produces metallic copper by thermal decomposition treatment. What is necessary is just to select the temperature of a thermal decomposition process suitably according to the structure of a complex compound, etc.
- the thermal decomposition temperature of copper formate is about 220 ° C., but copper formate forms a complex compound with amino alcohol, and as described in, for example, JP-A-2008-013466, the heat The decomposition temperature is considered to be about 110 to 120 ° C.
- the temperature for the thermal decomposition treatment is preferably 100 to 130 ° C, more preferably 110 to 130 ° C. When the temperature of the thermal decomposition treatment is 130 ° C. or lower, the formation of acid amides due to the dehydration reaction between the aliphatic carboxylic acid and amino alcohol is suppressed, and the washability of the resulting coated copper particles tends to be improved.
- Metallic copper is generated by thermal decomposition of the complex compound, and the coated copper particles whose surface is coated with the aliphatic carboxylic acid are obtained by adsorbing the aliphatic carboxylic acid present in the reaction solution on the surface of the generated metallic copper. be able to.
- the adsorption of the aliphatic carboxylic acid on the surface of metallic copper is preferably physical adsorption. This further improves the sinterability of the coated copper particles. By suppressing the formation of copper oxide in the thermal decomposition of the complex compound, physical adsorption of the aliphatic carboxylic acid is promoted.
- the thermal decomposition treatment it is preferable to remove at least a part of water generated in association with the thermal decomposition reaction of the complex compound. By removing water in the thermal decomposition treatment, the production of copper oxide can be suppressed more efficiently.
- the method for removing water is not particularly limited, and can be appropriately selected from conventionally used water removal methods. For example, it is preferable to remove the water produced by azeotropy using an organic solvent capable of forming an azeotrope with water as the solvent.
- the time for the pyrolysis treatment may be appropriately selected according to the temperature of the pyrolysis treatment. For example, it can be 30 to 180 minutes.
- the atmosphere for the pyrolysis treatment is preferably an inert atmosphere such as a nitrogen atmosphere.
- factors that control the particle size distribution of the produced coated copper particles include, for example, the type and amount of aliphatic carboxylic acid, the concentration of copper formate complex, and the ratio of the mixed solvent (main solvent / auxiliary Solvent) and the like.
- Factors that control the size of the coated copper particles are equalized by appropriately maintaining the heating rate that governs the number of metal nuclei generated, that is, the amount of heat input to the reaction system and the stirring rate related to the size of the micro reaction field. be able to.
- the method for producing coated copper particles is a simple operation of preparing a reaction solution containing copper formate, amino alcohol, aliphatic carboxylic acid and a solvent, and performing a thermal decomposition treatment at a desired temperature. And coated copper particles having excellent sinterability can be produced efficiently.
- coated copper particles having a narrow particle size distribution are obtained.
- This can be considered as follows, for example. That is, by setting the ⁇ SP value, which is the difference in SP value between the amino alcohol as a complexing agent for solubilizing copper formate in the reaction solvent and the solvent, to 4.2 or more, the copper formate amino alcohol complex or the formic acid Although it is dissolved in the state of a copper formate amino alcohol complex in which one molecule is substituted with an aliphatic carboxylic acid, when the complex is thermally decomposed to liberate amino alcohol as a complexing agent, the free amino alcohol is not a solvent. Cannot be compatible and begins to form two phases.
- the liberated amino alcohol has a high affinity with copper formate and copper formate amino alcohol complexes, so it acts as a new complexing agent or solvent for copper formate, forming a highly polar inner core (droplet) and polar outside. It becomes presumed that it takes a two-phase structure similar to Water in oil Emulsion surrounded by a low solvent, and this functions as a micro reaction field. Further, water in the reaction system and formic acid eliminated by substitution of the aliphatic carboxylic acid are also present in this microreaction field.
- the metal nucleus, its growing particles, and the copper formate amino alcohol complex that is the source of the metal nucleus, the copper formate amino alcohol complex in which one molecule of formic acid is replaced with an aliphatic carboxylic acid, water and formic acid are sequestered.
- the reaction proceeds.
- the aliphatic carboxylic acid is immobilized as a coating material for the copper metal growth particles and decreases, the thermal decomposition mechanism of the copper formate complex progresses in the reaction formulas 1 to 3 described later. The process proceeds by this mechanism, and the generated gas component changes.
- CuO is produced by hydrolysis of the copper formate amino alcohol complex with water shown in Reaction Formula 5, but is reduced again via Reaction Formula 6 or Reaction Formula 7, so cuprous oxide or copper oxide. Reduced copper particles that do not contain can be produced. Further, since the number of copper atoms contained in the micro reaction field is limited, the particle diameter of the copper particles is controlled to be constant. Then, since copper particles having no copper oxide formed on the surface are generated in the micro reaction field, the aliphatic carboxylic acid present in the micro reaction field is easily physically adsorbed, the particle diameter is uniform, and the oxidation resistance and sintering are facilitated. It can be considered that coated copper particles having excellent binding properties can be obtained efficiently.
- the method for producing coated copper particles may further include a washing step, a separation step, a drying step, and the like of the obtained coated copper particles after the thermal decomposition treatment.
- cleaning process of a covering copper particle the washing
- the organic solvent used in the washing step include alcohol solvents such as methanol and ketone solvents such as acetone. These may be used alone or in combination of two or more.
- the coated copper particles of the present embodiment are produced by the above-described method for producing coated copper particles, and the average primary particle diameter D SEM by SEM observation is 0.02 to 0.2 ⁇ m, and the variation coefficient (standard deviation) of the particle size distribution The value of SD / average primary particle diameter D SEM ) is 0.1 to 0.5.
- the variation coefficient of the particle size distribution is small, and the particle diameter is uniform. Since the coefficient of variation of the particle size distribution of the coated copper particles is small, the effect of excellent dispersibility and easy production of a high-concentration dispersion can be obtained.
- the coated copper particles of the present embodiment are obtained by the above-described method for producing coated copper particles, and the ratio D XRD to the average primary particle size D SEM by SEM observation of the crystal particle size D XRD obtained from powder X-ray analysis. / D SEM is 0.25 to 1.00.
- the difference between the crystal particle diameter and the average primary particle diameter can be reduced by manufacturing the coated copper particles described above. Thereby, it is excellent in oxidation resistance and the effect that sinterability improves more as a result is acquired.
- the coated copper particles of the present embodiment are obtained by the above-described method for producing coated copper particles, so that the surfaces of the copper particles are coated with an aliphatic carboxylic acid.
- the aliphatic carboxylic acid that coats the copper particles is a coating material that localizes on the surface of the copper particles and suppresses oxidation and aggregation, is removed from the particle surface during sintering, and further decomposes or volatilizes below the sintering temperature. Therefore, the remaining in the copper film formed by sintering is suppressed. This is considered to be because, for example, the aliphatic carboxylic acid is physically adsorbed on the surface of the copper particles.
- the copper particle which comprises a covering copper particle has the same particle diameter, it is excellent in a dispersibility. Further, since the difference between the crystallite diameter constituting the copper particles and the SEM observation diameter is small, the coated copper particles are not constituted by aggregation of a plurality of copper particles, and the coating material, impurities, and oxide layer are formed at the boundary of the aggregated particles. Inhibition of sintering due to the presence of.
- the electrically conductive composition of this embodiment contains at least 1 type of the covering copper particle obtained with the manufacturing method of the above-mentioned covering copper particle, and a medium.
- the conductive composition can be suitably used for forming a wiring pattern, and can easily form a wiring pattern excellent in conductivity at a low temperature. That is, this embodiment includes the use of the coated copper particles as a conductive composition.
- the structure of the medium contained in the conductive composition can be appropriately selected according to the purpose of the conductive composition.
- examples of the medium include hydrocarbon solvents, higher alcohol solvents, cellosolve, cellosolve acetate solvents, and the like.
- the solid content concentration of the conductive composition for screen printing can be set to 40 to 95% by mass, for example.
- the solid content of the conductive composition means the total amount of nonvolatile components.
- examples of the medium include hydrocarbon solvents, higher alcohol solvents, cellosolve, cellosolve acetate solvents, and the like.
- the solid content concentration of the conductive composition for inkjet printing can be set to 40 to 90% by mass, for example.
- the conductive composition may further contain other additives as required in addition to the coated copper particles and the medium.
- additives include coupling agents such as silane coupling agents and titanate coupling agents, and dispersants such as polyester dispersants and polyacrylic acid dispersants.
- the circuit formed product of this embodiment includes a base material and a wiring pattern that is a heat-treated product of the conductive composition disposed on the base material.
- the wiring pattern is excellent in conductivity.
- the wiring pattern can be formed at a low temperature, the degree of freedom of choice of base material is great.
- Examples of the material of the base material include polyimide film, glass, ceramics, and metal.
- the thickness in particular of a base material is not restrict
- the thickness of the substrate can be, for example, 0.01 mm to 5 mm.
- the formation of the wiring pattern can be performed, for example, by applying a conductive composition to a desired pattern on a substrate and heat-treating the applied conductive composition.
- a wiring pattern having a desired pattern and excellent conductivity can be efficiently formed at a low temperature.
- the circuit formed product can be manufactured by a manufacturing method including, for example, a step of preparing a base material, a step of applying a conductive composition on the base material, and a step of heat-treating the conductive composition. That is, this embodiment also includes a method for manufacturing a circuit formed product using the conductive composition.
- the method for applying the conductive composition is not particularly limited, and can be performed by, for example, an inkjet printing method, a screen printing method, a flexographic printing method, a dispensing method, or the like.
- the amount of the conductive composition applied can be appropriately selected according to the purpose and the like, and for example, the thickness after the heat treatment can be set to 1 to 100 ⁇ m.
- the heat treatment temperature of the conductive composition can be, for example, 200 to 600 ° C., and preferably 250 to 450 ° C.
- the heat treatment time can be, for example, 1 to 120 minutes, and preferably 5 to 60 minutes.
- the heat treatment atmosphere is preferably a low oxygen atmosphere. Examples of the low oxygen atmosphere include a nitrogen atmosphere and an argon atmosphere. Moreover, it is preferable that oxygen concentration is 1,000 ppm or less.
- Measuring device FE-EPMA JXA-8510F manufactured by JEOL Measurement conditions: acceleration voltage 6KV or 15KV Observation magnification ⁇ 10,000 to ⁇ 75,000
- XRD-6100 manufactured by Shimadzu Measurement conditions: Target Cu Tube voltage 40KV, tube current 30.0mA
- Measuring instrument ULVAC-PHI PHI TRIFT IV type Measurement conditions: Primary ion species Au, acceleration voltage 30 KV
- TG-DTA measurement> Measurement of organic residue and metal content Measuring device: TG8120 manufactured by Rigaku Temperature increase rate: 10 ° C / min Measurement temperature range: 25 °C ⁇ 600 °C Measurement atmosphere: Nitrogen 100ml / min
- Example 1 A 3000 mL glass four-necked flask equipped with a stirrer, a thermometer, a reflux condenser, a 75 mL Dean Stark tube, and a nitrogen introduction tube was placed in an oil bath. There, 484 g (3.1 mol) of copper formate anhydride, 68.1 g (0.11 equivalent / copper formate anhydride) of lauric acid (manufactured by Kanto Chemical Co., Ltd.), and tripropylene glycol monomethyl ether (Tokyo) as a reaction solvent 150 g (manufactured by Kasei Co., Ltd.) (0.23 equivalent / anhydrous copper formate) and 562 g (1.42 equivalent / anhydrous copper formate) of Swclean 150 (manufactured by Gordo) were added and mixed with stirring at 200 rpm.
- the mixture was heated and stirred at 200 rpm in a nitrogen atmosphere until the liquid temperature reached 50 ° C. Thereto, 712 g (3.00 equivalent / copper formate anhydride) of 3-amino-1-propanol (manufactured by Tokyo Chemical Industry Co., Ltd.) was slowly added dropwise. After completion of the dropwise addition, the mixture was heated and stirred at 340 rpm until the liquid temperature became around 120 ° C. The aqueous layer trapped by the Dean-Stark tube was removed as appropriate so that it was not refluxed into the reaction system. As the liquid temperature rose, the reaction solution started to change from dark blue to brown, and carbon dioxide gas bubbling occurred.
- the oil bath temperature control was stopped at the point where the bubbling of the carbon dioxide gas stopped, and the temperature was cooled to room temperature. After cooling to room temperature, 550 g of methanol (manufactured by Kanto Chemical Co., Inc.) was added and mixed. The mixed solution was allowed to stand for 30 minutes or more, and the supernatant was decanted to obtain a precipitate. To this precipitate, 550 g of methanol (manufactured by Kanto Chemical Co., Inc.) and 300 g of acetone (manufactured by Kanto Chemical Co., Ltd.) were added and mixed. This mixed solution was allowed to stand for 30 minutes or more, and the supernatant was decanted to obtain a precipitate, and this operation was repeated once more.
- 550 g of methanol manufactured by Kanto Chemical Co., Inc.
- 300 g of acetone manufactured by Kanto Chemical Co., Ltd.
- the precipitate was transferred to a 500 mL eggplant flask while being washed with 550 g of methanol (manufactured by Kanto Chemical Co., Inc.). The mixture was allowed to stand for 30 minutes or longer, the supernatant was decanted, and the resulting precipitate was placed on a rotary evaporator and vacuum dried at 40 ° C. and 1 kPa or less. After completion of the vacuum drying, the pressure was released while cooling to room temperature and substituting with nitrogen to obtain 194 g of brown coated copper particles. An SEM observation image of the obtained coated copper particles is shown in FIG. An enlarged SEM observation image is shown in FIG. 20A, and a particle size distribution is shown in FIG. 20B.
- Example 2 Coated copper particles were synthesized in the same manner as in Example 1 except that 3-amino-1-propanol was changed to DL-1-amino-2-propanol. An SEM observation image of the obtained coated copper particles is shown in FIG.
- Example 3 Coated copper particles were synthesized in the same manner as in Example 1 except that 3-amino-1-propanol was changed to 5-amino-1-pentanol and the reaction solvent was changed to n-octane.
- Example 4 Coated copper particles were synthesized in the same manner as in Example 1 except that 3-amino-1-propanol was changed to DL-1-amino-2-propanol and the reaction solvent was changed to n-octane. An SEM observation image of the obtained coated copper particles is shown in FIG.
- Example 4 Coated copper particles were synthesized in the same manner as in Example 1 except that 3-amino-1-propanol was changed to 5-amino-1-pentanol. An SEM observation image of the obtained coated copper particles is shown in FIG.
- Coated copper particles were synthesized in the same manner as in Example 1 except that the reaction solvent was changed to n-octanol. An SEM observation image of the obtained coated copper particles is shown in FIG.
- Example 5 Coated copper particles were synthesized in the same manner as in Example 1 except that lauric acid was 68.16 g, oleic acid was not used as a solvent, and the auxiliary solvent was not used, and Swclean # 150 was changed to 712 g. An SEM observation image of the obtained coated copper particles is shown in FIG.
- Example 6 Coated copper particles were synthesized in the same manner as in Example 1 except that 48 g of lauric acid was changed to 16 g. SEM observation images of the obtained coated copper particles are shown in FIGS. 16A and 16B.
- Example 7 Coated copper particles were synthesized in the same manner as in Example 1 except that 48 g of lauric acid was changed to 144 g. SEM observation images of the obtained coated copper particles are shown in FIGS. 17A and 17B.
- Example 8 Coated copper particles were synthesized in the same manner as in Example 1 except that Swagele # 150 was changed to 150 g and methylpropylene triglycol was changed to 562 g as a reaction solvent. SEM observation images of the obtained coated copper particles are shown in FIGS. 18A and 18B.
- Example 9 Coated copper particles were synthesized in the same manner as in Example 1 except that lauric acid was changed to octanoic acid. SEM observation images of the obtained coated copper particles are shown in FIGS. 19A and 19B.
- Coated copper particles were synthesized according to the method described in Example 1 of JP2013-047365A. Specifically, coated copper particles were synthesized using acetic acid as a coating material as follows. 14.3 g (0.1 mol) of cuprous oxide (I) (manufactured by Furukawa Chemicals; particle size: 2 to 4 ⁇ ) as a copper compound, 3.0 g (50 mmol) of acetic acid as a coating material, hydrazine as a reducing agent Hydrate (manufactured by Wako Pure Chemical Industries, Ltd.) 5.0 g (0.1 mol) and 100 ml of isopropanol as a solvent were mixed and added to a 300 ml four-necked flask.
- cuprous oxide (I) manufactured by Furukawa Chemicals; particle size: 2 to 4 ⁇
- acetic acid as a coating material
- hydrazine as a reducing agent Hydrate
- the flask was equipped with a condenser, a thermometer, a nitrogen inlet tube and a stirrer. While agitating nitrogen at 200 ml / min, the temperature was raised to 70 ° C. while stirring, and heating and stirring were continued for 1 hour to reduce cuprous oxide (I) to obtain a coated copper particle dispersion.
- the coated copper particle dispersion was designated as Kiriyama Filter Paper No.
- the powder was filtered off under reduced pressure with 5B.
- the powder separated by filtration was washed 3 times with methanol (manufactured by Kanto Chemical Co., Inc.), dried under reduced pressure at 40 ° C.
- Comparative Example 7 Comparative Example 6 was scaled up, and the reaction time was doubled to synthesize coated copper particles. 71.5 g (0.5 mol) of cuprous oxide (I) (manufactured by Furukawa Chemicals) as the copper compound, 15.0 g (250 mmol) of acetic acid as the coating material, hydrazine monohydrate as the reducing agent (Wako Pure Chemicals) [Industry] 25.0 g (0.5 mol) and 500 ml of isopropanol as a solvent were mixed and added to a 1,000 ml four-necked flask, which was equipped with a condenser, thermometer, nitrogen inlet tube and stirring device.
- I cuprous oxide
- acetic acid 15.0 g (250 mmol)
- hydrazine monohydrate as the reducing agent
- [Industry] 25.0 g (0.5 mol) and 500 ml of isopropanol as a solvent were mixed and added to a 1,000 ml four
- the coated copper particle dispersion was designated as Kiriyama Filter Paper No.
- the powder was filtered off under reduced pressure with 5B.
- the powder separated by filtration was washed 3 times with methanol (manufactured by Kanto Chemical Co., Inc.), dried under reduced pressure at 40 ° C. and 1 kPa or less, cooled to room temperature, purged with nitrogen, and taken out to obtain 62 g of brown powder.
- XRD of the powder was measured (shown in FIG. 22), the raw material cuprous oxide (I) was quantitatively converted to reduced copper.
- An SEM observation image of the obtained coated copper particles is shown in FIG. 26A.
- D K ⁇ / ( ⁇ cos ⁇ ) (1)
- D is a crystal particle diameter
- ⁇ is a wavelength of a measured X-ray (CuK ⁇ : 1.5418 ⁇ )
- ⁇ is expressed by Equation (2).
- the ⁇ b ⁇ B (2)
- b is the half width of the peak
- the crystallite diameter D XRD of the coated copper particles was 48.9 nm. Since the average primary particle size D SEM calculated from the SEM observation result is 85.8 nm, it is 0.57 when calculating D XRD / D SEM, which indicates that the crystallite size with respect to the average primary particle size is relatively large.
- Tof-SIMS surface analysis was performed to investigate the surface composition of the coated copper particles. From the results of the Tof-SIMS surface analysis, free lauric acid was detected almost quantitatively (shown in FIG. 2A), and a small amount of lauric acid bound to 63 Cu and 65 Cu hydroxides. was also detected (shown in FIG. 2B). Since lauric acid bonded to 63 Cu and 65 Cu was not detected, it was found that what was present on the surface of the coated copper particles was lauric acid coated mostly by physical adsorption.
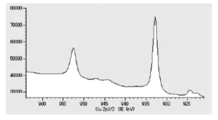
- TG-DTA analysis was performed (FIG. 3). From the results of TG-DTA analysis, it can be seen that the loss on heating is 1.09% by mass, and that almost all of the product is desorbed near the boiling point of lauric acid. This result also suggests that lauric acid is physically adsorbed, and it is assumed that the coated copper particles can exhibit low-temperature sinterability.
- the coating density of the aliphatic carboxylic acid coating the surface of the copper particles was calculated by the following method. If the total amount of the heat loss component is assumed to be lauric acid according to the analysis result of Tof-SIMS, the number of lauric acids contained in the coated copper particles is expressed by formula (3).
- [laurate number] M acid / (M W / N A) ⁇ (3)
- M acid is heated loss measured mass values (g)
- M W is the molecular lauric acid weight (g / mol)
- N A is the Avogadro constant (6.02 ⁇ 10 23 lines / mol).
- the number of particles in 1 g of copper particles is expressed by the following equation (4).
- Number of particles in 1g] M Cu / [( 4 ⁇ r 3/3) ⁇ d ⁇ 10 -21] ⁇ (4)
- M Cu is a calculated mass value (g) obtained from the measured heat loss
- r is the radius (nm) of the primary particle diameter calculated by SEM observation
- the particle surface area in 1 g of copper particles is represented by the formula (5) using the formula (4).
- the coating density of lauric acid in the coated copper particles was 4.23 particles / nm 2 .
- the minimum area is calculated from the van der waals radius of the stearic acid molecule from “Chemistry and Education, Vol. 40, No. 2, (1992) Obtaining the cross-sectional area of the stearic acid molecule—experimental values and calculated values—”.
- the theoretical value of the saturated covered area converted from is about 5.00 lines / nm 2 . From this theoretical value, it is presumed that the coated copper particles of this embodiment have lauric acid localized on the particle surface at a relatively high density. This dense coating effect can be considered as a reason why the lauric acid coating is excellent in oxidation resistance in spite of physical adsorption that is weaker than chemical adsorption.
- Example 1 the reaction mechanism of the present invention was estimated by component analysis of the gas discharged during the reaction and the evaporated distillate, taking Example 1 as an example.
- ⁇ Gas component analysis> Method Gas chromatograph Measuring instrument: GL Science GL320 Detector: Thermal conductivity detector (TCD) Column: Stainless steel column ⁇ 3mm ⁇ 2m Column packing (hydrogen): Molecular Sieve 5A Column packing (carbon dioxide): Active Carbon Carrier gas (hydrogen): N 2 20 mL / min Carrier gas (carbon dioxide): He 50mL / min Measurement temperature: 43-50 ° C Current value: 70 to 120 mA
- the exhaust gas component is carbon dioxide gas
- the reaction temperature is around 120 ° C. (FIG. 4). Therefore, it is considered that the reaction mechanism proceeds through the following reaction equation 1 and the reaction mechanism of reaction equation 2. That is, first, one molecule of lauric acid causes an equilibrium exchange reaction with one molecule of formic acid of a copper formate amino alcohol complex.
- exhaust gas components are hydrogen gas and carbon dioxide gas, and it is considered that the following reaction mechanism proceeds from the component ratio.
- Scheme 4 (HCOO ⁇ ) (HCOO ⁇ ) Cu 2+. (H 2 NC 3 H 6 OH) 2 ⁇ Cu + 2H 2 NC 3 H 6 OH + H 2 + 2CO 2 It is considered that not only reaction formula 1 but also reaction formula 4 proceeds simultaneously as lauric acid is consumed as a covering material of the reduced copper particle growth mechanism rather than the equilibrium exchange reaction of complex formation with copper formate.
- reaction Formula 7 CuO + 2HCOOH ⁇ (HCOO ⁇ ) (HCOO ⁇ ) Cu 2+ + H 2 O
- the regenerated copper formate regenerates according to reaction formula 2 via reaction formula 1. It is thought that water molecules were generated by these side reactions and were discharged as an evaporating fraction. Since this side reaction is a reaction that can occur if formic acid is present in the reaction system, even if copper oxide is unexpectedly formed, the coated copper having no oxide film due to the reduction reaction mechanism of reaction formula 6 and reaction formula 7. It is presumed that particles can be synthesized. In order to verify whether this reduction reaction mechanism can occur in the manufacturing method of the present embodiment, the reaction was intentionally performed by adding copper oxide to the reaction system. The result will be described in Reference Example 2.
- the mixture was heated and stirred at 200 rpm until the liquid temperature reached 50 ° C. Thereto, 21.0 g (3.50 equivalent / copper formate anhydride) of 3-amino-1-propanol (manufactured by Tokyo Chemical Industry Co., Ltd.) was slowly added dropwise. After completion of the dropwise addition, the mixture was heated and stirred at 340 rpm until the liquid temperature became around 120 ° C. As the liquid temperature rises, the reaction solution starts to change from dark blue to brown, and carbon dioxide gas bubbling occurs. The oil bath temperature control was stopped at the point where the bubbling of the carbon dioxide gas stopped, and the temperature was cooled to room temperature.
- This precipitate was transferred to a 100 mL eggplant flask while being washed with 20.0 g of methanol (manufactured by Kanto Chemical Co., Inc.). The mixture was allowed to stand for 30 minutes or more, the supernatant was decanted, and the resulting precipitate was placed on a rotary evaporator and vacuum dried at 40 ° C. and 1 kPa or less. After completion of the vacuum drying, the solution was cooled to room temperature and released under reduced pressure while being purged with nitrogen to obtain 6.5 g of brown copper powder.
- a copper formate amino alcohol complex containing an aliphatic carboxylic acid is formed as in this embodiment (Scheme 8).
- the thermal decomposition temperature of such a copper formate complex is low, aliphatic carboxylic acid and copper formate amino alcohol complex It is described that formic acid eliminated in the exchange reaction is discharged out of the system.
- the reaction conditions are at a low temperature, it is considered that the reduction reaction with formic acid present in the reaction system does not proceed.
- reaction mechanism of the present embodiment has a reaction mechanism for reducing the by-produced copper oxide by the reaction formula 6 and the reaction formula 7 as well as the production of reduced copper by the thermal decomposition reaction of the copper formate complex.
- the coated copper particles to be synthesized are less prone to oxidation.
- the production method of this embodiment does not require strict production control of water and oxygen, which are cited as causes of metal copper oxidation, and is a production method suitable for simpler synthesis.
- the difference between the SP value of the amino alcohol constituting the copper formate amino alcohol complex and the reaction solvent, or the SP value of the mixed solvent of the main solvent and the auxiliary solvent, that is, the ⁇ SP value is 4.2 or more. If it is large, individual coated copper particles can be produced, but if it is 4.2 or less, it does not become individual coated copper particles but forms an aggregate, and as a method for producing coated copper particles, It turns out that it is not suitable. Further, the ratio of the size of the crystal particles in the primary particles, which is defined as crystallinity, is 0.25 or more, and in most cases, about 0.50, and the coated copper particles are composed of one large crystallite. The appearance is shown.
- Example X-ray powder analysis and SEM observation were performed on the coated copper particles obtained in Examples 5 to 8 and Comparative Examples 6 to 7. The results are shown in Table 3.
- the heating rate during the reaction is constant, the reduction reaction rate will be the same, and the amount of metal nuclei generated will be the same. Therefore, it controls the size and stability of the micro-reaction field similar to water-in-oil emulsion. It can be seen that there is a difference in particle size and particle size distribution depending on the factors to be performed. In the direction of stabilizing the micro reaction field, the particle diameter and the fluctuation rate tend to be small, and this tendency becomes larger as the carbon chain of the aliphatic carboxylic acid becomes longer (comparison of Example 1, Example 5 and Example 9).
- Example 1 the number of moles of aliphatic carboxylic acid with respect to copper atoms tends to stabilize, but when the reaction viscosity increases, the particle size increases even if the rate of change is small (Examples 1, 6 and 7). comparison). If the ratio of the main solvent to the auxiliary solvent in the reaction solvent is changed and the average SP value varies from 8.21 to 8.90, even if ⁇ SP with amino alcohol is ensured, the properties of the obtained coated copper particles are improved. It was also confirmed that there was no significant difference (comparison of Example 1 and Example 8).
- JP 2013-047365 A which is similar in that the coated copper particle of the present embodiment and the coating material are aliphatic carboxylic acids, Tried to manufacture.
- These methods are methods for producing coated copper particles using a copper atom source as a reservoir which is a hardly soluble solid.
- a raw material cuprous (I) having a particle size as small as 2 to 4 ⁇ m was used, but unreacted substances remained for a predetermined time. Although the reaction time was quantitatively converted to reduced copper, the average particle size was increased. In addition, the particle size distribution was as large as the fluctuation rates of 0.38 and 0.29.
- Example 2 Characteristics of the fired film by the coated copper particle paste
- the coated copper particles produced under the conditions of Example 1 were dispersed with a solvent to prepare a copper paste composition, and 300 ° C and 350 ° C under a nitrogen atmosphere.
- a copper film (copper paste sintered layer) was prepared by firing for 1 hour, and the electric resistance of the film was measured.
- a copper paste composition having a metal content of 33% by volume was prepared by dispersing and kneading the coated copper particles and the solvent with a mortar so as to have the following composition to form a paste.
- 26A and 26B show SEM observation images of the produced coated copper particles and the fired copper film, and Table 4 shows the measurement results of electric resistance.
- Test Example 4 Characteristics of fired film by mixed paste of coated copper particles and copper powder
- the coated copper particles produced in Example 1 were added as a sintering agent for a commercially available copper powder to prepare a copper paste composition.
- a copper film was prepared by baking at 300 ° C. and 350 ° C. for 1 hour in a nitrogen atmosphere, and the electric resistance of the film was measured.
- Each material was dispersed and kneaded with a three roll mill so as to have the following composition to prepare a copper paste composition having a metal content of 60% by volume.
- Copper paste composition C Coated copper particles of Example 1 100 parts by weight Wet copper powder 2.0 ⁇ m (Mitsui Metals: 1200N) 100 parts by weight Wet copper powder 0.8 ⁇ m (Mitsui Metals: 1050Y) 25 parts by weight Polyacrylic acid dispersant 0. 5 parts by weight Kyowanol M (NH Neochem) 15 parts by weight
- An about 10 ⁇ m wet coating was applied to a 40 ⁇ m thick polyimide film laminated with a 12 ⁇ m copper foil on the back side, and dried and fired in a nitrogen atmosphere to prepare a sample for evaluation.
- the average film thickness of the copper after firing was measured by cross-sectional observation with SEM.
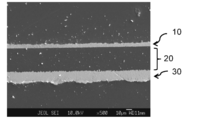
- the film thickness of the copper paste sintered layer was 4.2 ⁇ m.
- An SEM cross-sectional observation image of the evaluation sample used for the measurement is shown in FIG. In FIG. 27, on the surface opposite to the copper foil layer 30 of the polyimide film having a 12 ⁇ m thick copper foil layer 30 on one surface of the 40 ⁇ m thick polyimide film layer 20, a fired product of the copper paste composition A certain copper paste sintered layer 10 is formed.
- the copper paste composition B was coated copper particles coated with the same aliphatic carboxylic acid, and showed a value of 22 ⁇ ⁇ cm, which is one digit higher than the volume resistivity value of the copper bulk.
- the surface composition structure of the sintered copper film produced with the copper paste compositions A and B was subjected to composition analysis by XPS, and the mechanism by which the difference in electrical characteristics occurred was considered.
- the data obtained by the narrow scan in FIGS. 28 and 29 show composition information on the outermost surface of the sintered copper coatings of the copper paste compositions A and B. It can be seen that the outermost surface of the sintered copper film of the copper paste composition B has a higher ratio of reduced copper and a lower ratio of the copper oxide component and the organic component than that of the copper paste composition A.
- the outermost surface of the sintered copper film made of the copper paste composition A is coated with an aliphatic carboxylic acid presumed to be lauric acid, and the amount of organic components increases accordingly. However, although there is an oxide component, a considerable amount of reduced copper is exposed, and it is determined that the contact resistance is not impaired.
- etching was performed with argon ions, and the surface composition in the region of several nm from the outermost surface was clarified by XPS DepthProfile analysis.
- the copper paste composition B is expected to have a lower sintered density because the carbon content does not decrease even in a region deeper than several nm compared to A.
- the mechanism for expressing the low-temperature sinterability of the coated copper particles is not to remove the coating material of the coated copper particles at a low temperature, but to prevent contact between the copper particles, necking and mutual diffusion of copper atoms. It has been found that it is important to be removed moderately.
- the sintered copper film obtained from the coated copper particles of the present invention the aliphatic carboxylic acid in the coating material existing between the particles is efficiently removed while the particles contact, necking, and interdiffusion of copper atoms.
- characteristics close to the resistance value of the copper bulk can be achieved.
- the outermost surface of the sintered copper film is covered with the aliphatic carboxylic acid of the coating material and can be expected as a barrier layer against oxygen.
- the method for producing coated copper particles according to the present invention comprises coated copper particles having excellent oxidation resistance in which the particle diameter is controlled by a unique micro reaction field and the particle surface is covered with a high-density coating layer of aliphatic carboxylic acid. Can be manufactured.
- the obtained coated copper particles are almost composed of reduced copper, and it is considered that the aliphatic carboxylic acid as the coating material is bonded by physical adsorption. Therefore, in order to express the sintering ability by being desorbed near the boiling point of the coating material, for example, a copper sintered film having a volume resistivity close to copper bulk at a relatively low temperature of 300 ° C. or less at normal pressure. Obtainable.
- the coated copper particles can be made into a paste with a solvent and the like, and a wiring pattern can be formed by means such as screen printing, and the firing atmosphere of the formed wiring pattern only needs to be in a low oxygen state.
- a highly versatile production process that can be used can be configured.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Physics & Mathematics (AREA)
- Dispersion Chemistry (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Powder Metallurgy (AREA)
- Manufacture Of Metal Powder And Suspensions Thereof (AREA)
- Conductive Materials (AREA)
- Non-Insulated Conductors (AREA)
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020167027210A KR101709458B1 (ko) | 2014-05-30 | 2015-05-14 | 피복 구리 입자 및 그 제조 방법 |
| US15/307,227 US9744595B2 (en) | 2014-05-30 | 2015-05-14 | Coated copper particles and method for producing the same |
| CN201580028643.XA CN106457407B (zh) | 2014-05-30 | 2015-05-14 | 被覆铜颗粒的制造方法 |
| KR1020167034561A KR101743897B1 (ko) | 2014-05-30 | 2015-05-14 | 피복 구리 입자 및 그 제조 방법 |
| EP15799589.5A EP3150306B1 (en) | 2014-05-30 | 2015-05-14 | Coated copper particles and method for manufacturing same |
| US15/685,729 US20180036805A1 (en) | 2014-05-30 | 2017-08-24 | Coated copper particles and method for producing the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-112794 | 2014-05-30 | ||
| JP2014112794A JP5926322B2 (ja) | 2014-05-30 | 2014-05-30 | 被覆銅粒子及びその製造方法 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/307,227 A-371-Of-International US9744595B2 (en) | 2014-05-30 | 2015-05-14 | Coated copper particles and method for producing the same |
| US15/685,729 Continuation US20180036805A1 (en) | 2014-05-30 | 2017-08-24 | Coated copper particles and method for producing the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015182395A1 true WO2015182395A1 (ja) | 2015-12-03 |
Family
ID=54698732
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/063880 Ceased WO2015182395A1 (ja) | 2014-05-30 | 2015-05-14 | 被覆銅粒子及びその製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US9744595B2 (enExample) |
| EP (1) | EP3150306B1 (enExample) |
| JP (1) | JP5926322B2 (enExample) |
| KR (2) | KR101709458B1 (enExample) |
| CN (2) | CN108161027B (enExample) |
| TW (1) | TWI638769B (enExample) |
| WO (1) | WO2015182395A1 (enExample) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108883466A (zh) * | 2016-03-28 | 2018-11-23 | 协立化学产业株式会社 | 被覆银颗粒及其制造方法、导电性组合物和导体 |
| CN111526952A (zh) * | 2017-11-29 | 2020-08-11 | 国立大学法人北海道大学 | 低温烧结性铜粒子及使用该低温烧结性铜粒子的烧结体的制造方法 |
| US11859112B2 (en) | 2017-11-13 | 2024-01-02 | Kyocera Corporation | Paste composition, semiconductor device, and electrical/electronic component |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3117596B2 (ja) | 1993-12-29 | 2000-12-18 | オルガノ株式会社 | 3分子以上の単糖類分子がグリコシド結合してなる糖類の製造方法 |
| JP6715450B2 (ja) * | 2016-01-13 | 2020-07-01 | 小林 博 | 金属ないしは金属酸化物からなる個々のナノ粒子が、液体の有機化合物に囲まれて該有機化合物中に分散した懸濁体を製造する製造方法 |
| JP7251470B2 (ja) * | 2017-03-15 | 2023-04-04 | 株式会社レゾナック | 接合用金属ペースト、接合体及びその製造方法、並びに半導体装置及びその製造方法 |
| EP3597330A4 (en) * | 2017-03-15 | 2020-11-25 | Hitachi Chemical Company, Ltd. | METAL PASTE FOR BONDING, BONDED BODY AS WELL AS A METHOD FOR MANUFACTURING THE SAME, AND A SEMICONDUCTOR DEVICE AS WELL AS A METHOD FOR MANUFACTURING THE SAME |
| JP6976597B2 (ja) | 2017-04-14 | 2021-12-08 | 学校法人 関西大学 | 銅粒子混合物及びその製造方法、銅粒子混合物分散液、銅粒子混合物含有インク、銅粒子混合物の保存方法及び銅粒子混合物の焼結方法 |
| CN111565870B (zh) * | 2018-01-26 | 2023-04-04 | 日清工程株式会社 | 铜微粒子 |
| US20210069782A1 (en) * | 2018-01-26 | 2021-03-11 | Nisshin Engineering Inc. | Fine particle production method and fine particles |
| JP7139258B2 (ja) * | 2019-01-22 | 2022-09-20 | 大陽日酸株式会社 | 銅微粒子、導電性材料、銅微粒子の製造方法 |
| CN109655521A (zh) * | 2019-01-28 | 2019-04-19 | 中国地质科学院水文地质环境地质研究所 | 基于加速器质谱技术的14c的快速测量方法 |
| JP7269565B2 (ja) * | 2019-03-29 | 2023-05-09 | 学校法人 関西大学 | 導電性インキ組成物及び導電性積層体 |
| US12070801B2 (en) * | 2019-03-29 | 2024-08-27 | Mitsui Mining & Smelting Co., Ltd. | Bonding material and bonded structure |
| JP7244919B2 (ja) * | 2019-08-27 | 2023-03-23 | 協立化学産業株式会社 | 被覆銅ナノ粒子含有組成物、及び物品 |
| EP4205882A4 (en) * | 2020-08-28 | 2024-01-24 | National University Corporation Hokkaido University | FINE OXIDE-CONTAINING COPPER PARTICLES, METHOD FOR MANUFACTURING THEM AND METHOD FOR MANUFACTURING A SINTERED TABLET FROM FINE OXIDE-CONTAINING COPPER PARTICLES |
| CN116329539B (zh) * | 2023-03-03 | 2024-05-28 | 嘉庚创新实验室 | 铜复合材料及其制备方法、铜导电浆料和铜膜 |
| CN118136337B (zh) * | 2024-04-01 | 2025-06-20 | 合肥工业大学 | 一种高收缩率铜导电薄膜的制备方法及其材料 |
| CN119747963A (zh) * | 2024-11-20 | 2025-04-04 | 哈尔滨工业大学(深圳)(哈尔滨工业大学深圳科技创新研究院) | 一种基于热分解铜盐的自还原铜膏及其制备方法和应用 |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004273446A (ja) * | 2003-02-18 | 2004-09-30 | Toray Ind Inc | 導電性ペーストおよびそれを用いたディスプレイパネル用部材の製造方法 |
| JP2008013466A (ja) * | 2006-07-04 | 2008-01-24 | Seiko Epson Corp | ギ酸銅錯体、銅粒子の製造方法および配線基板の製造方法 |
| JP2009082828A (ja) * | 2007-09-28 | 2009-04-23 | Fujifilm Corp | 逆ミセル液、無機ナノ粒子及び無機ナノ粒子の製造方法 |
| JP2011032558A (ja) * | 2009-08-04 | 2011-02-17 | Harima Chemicals Inc | 金属銅微粒子の製造方法 |
| JP2011032509A (ja) * | 2009-07-30 | 2011-02-17 | Dowa Electronics Materials Co Ltd | 金属ナノ粒子分散液 |
| WO2012053456A1 (ja) * | 2010-10-21 | 2012-04-26 | 旭硝子株式会社 | 水素化銅微粒子分散液の製造方法、導電インクおよび導体付き基材の製造方法 |
| JP2013047365A (ja) * | 2011-08-29 | 2013-03-07 | Hitachi Cable Ltd | 銅微粒子分散液およびその製造方法、銅微粒子およびその製造方法、銅微粒子を含む銅ペースト、並びに銅被膜およびその製造方法 |
| JP2014201618A (ja) * | 2013-04-02 | 2014-10-27 | 旭硝子株式会社 | 金属微粒子分散液からなる導電インク、導体付き基材 |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2702796B2 (ja) * | 1990-02-23 | 1998-01-26 | 旭化成工業株式会社 | 銀合金導電性ペースト |
| JPH08143916A (ja) | 1994-11-24 | 1996-06-04 | Nok Corp | 鉄微粒子の製造方法 |
| US6951666B2 (en) * | 2001-10-05 | 2005-10-04 | Cabot Corporation | Precursor compositions for the deposition of electrically conductive features |
| JP3900414B2 (ja) | 2002-02-18 | 2007-04-04 | 富士フイルム株式会社 | ナノ粒子およびナノ粒子の製造方法、並びに、磁気記録媒体 |
| JP4431085B2 (ja) | 2004-06-24 | 2010-03-10 | シャープ株式会社 | 導電性インキ組成物、反射部材、回路基板、電子装置 |
| JP2008019461A (ja) * | 2006-07-11 | 2008-01-31 | Fujifilm Corp | 金属ナノ粒子の製造方法、金属ナノ粒子及び金属ナノ粒子分散物 |
| JP5045015B2 (ja) | 2006-07-28 | 2012-10-10 | セイコーエプソン株式会社 | ギ酸銅の製造方法、銅粒子の製造方法および配線基板の製造方法 |
| JP4961587B2 (ja) * | 2006-10-03 | 2012-06-27 | トヨタ自動車株式会社 | 銅微粒子、銅微粒子製造方法、絶縁材料、配線構造、配線回路板の製造方法、及び電子・電気機器 |
| JP2008205430A (ja) * | 2007-01-26 | 2008-09-04 | Konica Minolta Holdings Inc | 金属パターン形成方法及び金属塩混合物 |
| JP2009158441A (ja) | 2007-12-28 | 2009-07-16 | Namics Corp | 導電性ペースト及びそれを用いた銅膜の形成方法 |
| JP5003668B2 (ja) * | 2008-12-18 | 2012-08-15 | 住友金属鉱山株式会社 | 酸化物被覆銅微粒子の製造方法 |
| JP5759688B2 (ja) | 2010-08-17 | 2015-08-05 | 三井金属鉱業株式会社 | 扁平銅粒子 |
| JP5342597B2 (ja) * | 2011-04-15 | 2013-11-13 | 福田金属箔粉工業株式会社 | 銅超微粒子分散ペーストおよび導電膜の形成方法 |
| CN102220046A (zh) * | 2011-06-01 | 2011-10-19 | 上海大学 | 纳米锡包铜导电墨水的制备方法 |
| KR20150006091A (ko) * | 2012-02-29 | 2015-01-15 | 이슘 리서치 디벨롭먼트 컴퍼니 오브 더 히브루 유니버시티 오브 예루살렘, 엘티디. | 금속 전구체 나노입자들을 함유한 잉크 |
| JP2014001443A (ja) * | 2012-06-21 | 2014-01-09 | Kyoritsu Kagaku Sangyo Kk | 酸化物被覆銅微粒子及びその製造方法 |
| US10214656B2 (en) * | 2014-02-27 | 2019-02-26 | A School Corporation Kansai University | Copper nanoparticles and production method for same, copper nanoparticle fluid dispersion, copper nanoink, copper nanoparticle preservation method, and copper nanoparticle sintering method |
-
2014
- 2014-05-30 JP JP2014112794A patent/JP5926322B2/ja active Active
-
2015
- 2015-05-14 KR KR1020167027210A patent/KR101709458B1/ko active Active
- 2015-05-14 CN CN201711328045.1A patent/CN108161027B/zh active Active
- 2015-05-14 KR KR1020167034561A patent/KR101743897B1/ko active Active
- 2015-05-14 EP EP15799589.5A patent/EP3150306B1/en active Active
- 2015-05-14 US US15/307,227 patent/US9744595B2/en active Active
- 2015-05-14 CN CN201580028643.XA patent/CN106457407B/zh active Active
- 2015-05-14 WO PCT/JP2015/063880 patent/WO2015182395A1/ja not_active Ceased
- 2015-05-22 TW TW104116484A patent/TWI638769B/zh active
-
2017
- 2017-08-24 US US15/685,729 patent/US20180036805A1/en not_active Abandoned
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2004273446A (ja) * | 2003-02-18 | 2004-09-30 | Toray Ind Inc | 導電性ペーストおよびそれを用いたディスプレイパネル用部材の製造方法 |
| JP2008013466A (ja) * | 2006-07-04 | 2008-01-24 | Seiko Epson Corp | ギ酸銅錯体、銅粒子の製造方法および配線基板の製造方法 |
| JP2009082828A (ja) * | 2007-09-28 | 2009-04-23 | Fujifilm Corp | 逆ミセル液、無機ナノ粒子及び無機ナノ粒子の製造方法 |
| JP2011032509A (ja) * | 2009-07-30 | 2011-02-17 | Dowa Electronics Materials Co Ltd | 金属ナノ粒子分散液 |
| JP2011032558A (ja) * | 2009-08-04 | 2011-02-17 | Harima Chemicals Inc | 金属銅微粒子の製造方法 |
| WO2012053456A1 (ja) * | 2010-10-21 | 2012-04-26 | 旭硝子株式会社 | 水素化銅微粒子分散液の製造方法、導電インクおよび導体付き基材の製造方法 |
| JP2013047365A (ja) * | 2011-08-29 | 2013-03-07 | Hitachi Cable Ltd | 銅微粒子分散液およびその製造方法、銅微粒子およびその製造方法、銅微粒子を含む銅ペースト、並びに銅被膜およびその製造方法 |
| JP2014201618A (ja) * | 2013-04-02 | 2014-10-27 | 旭硝子株式会社 | 金属微粒子分散液からなる導電インク、導体付き基材 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3150306A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108883466A (zh) * | 2016-03-28 | 2018-11-23 | 协立化学产业株式会社 | 被覆银颗粒及其制造方法、导电性组合物和导体 |
| US11859112B2 (en) | 2017-11-13 | 2024-01-02 | Kyocera Corporation | Paste composition, semiconductor device, and electrical/electronic component |
| CN111526952A (zh) * | 2017-11-29 | 2020-08-11 | 国立大学法人北海道大学 | 低温烧结性铜粒子及使用该低温烧结性铜粒子的烧结体的制造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101709458B1 (ko) | 2017-02-22 |
| CN108161027A (zh) | 2018-06-15 |
| KR20160120786A (ko) | 2016-10-18 |
| CN106457407A (zh) | 2017-02-22 |
| US9744595B2 (en) | 2017-08-29 |
| CN108161027B (zh) | 2019-01-08 |
| TW201601993A (zh) | 2016-01-16 |
| EP3150306A4 (en) | 2018-01-17 |
| JP2015227476A (ja) | 2015-12-17 |
| JP5926322B2 (ja) | 2016-05-25 |
| EP3150306B1 (en) | 2019-01-30 |
| CN106457407B (zh) | 2017-12-05 |
| KR20160145207A (ko) | 2016-12-19 |
| EP3150306A1 (en) | 2017-04-05 |
| TWI638769B (zh) | 2018-10-21 |
| US20180036805A1 (en) | 2018-02-08 |
| KR101743897B1 (ko) | 2017-06-05 |
| US20170043404A1 (en) | 2017-02-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5926322B2 (ja) | 被覆銅粒子及びその製造方法 | |
| JP6033485B2 (ja) | 被覆銅粒子 | |
| JP6368925B2 (ja) | 被覆銅粒子及びその製造方法 | |
| JP5858374B2 (ja) | 被覆銅微粒子の製造方法 | |
| JP2014001443A (ja) | 酸化物被覆銅微粒子及びその製造方法 | |
| JP6979150B2 (ja) | 被覆銀粒子とその製造方法、導電性組成物、および導電体 | |
| JP5426270B2 (ja) | 金属銅微粒子の製造方法 | |
| JP4897624B2 (ja) | 低温焼結性銀微粉および銀塗料ならびにそれらの製造法 | |
| JP6209249B2 (ja) | 酸化物被覆銅微粒子の製造方法 | |
| JP5232016B2 (ja) | 配線形成用材料 | |
| WO2016080544A1 (ja) | 金属表面の処理方法並びに当該方法により処理された銀被着銅及び複合金属体 | |
| JP6099160B2 (ja) | 複合化合物、及び懸濁液 | |
| JP5176060B2 (ja) | 銀粒子分散液の製造法 | |
| JP5063003B2 (ja) | 銅ナノ粒子の製造方法、銅ナノ粒子、導電性組成物および電子デバイス | |
| JP6387794B2 (ja) | 有機被覆金属ナノ粒子及びその製造方法 | |
| TWI707052B (zh) | 印刷用導電性糊劑及其調製方法與銀奈米粒子分散液之調製方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15799589 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20167027210 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015799589 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15307227 Country of ref document: US Ref document number: 2015799589 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |