WO2007061328A1 - Composition de soins et de prevention pour la cavite buccale - Google Patents
Composition de soins et de prevention pour la cavite buccale Download PDFInfo
- Publication number
- WO2007061328A1 WO2007061328A1 PCT/RU2005/000601 RU2005000601W WO2007061328A1 WO 2007061328 A1 WO2007061328 A1 WO 2007061328A1 RU 2005000601 W RU2005000601 W RU 2005000601W WO 2007061328 A1 WO2007061328 A1 WO 2007061328A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition according
- sodium
- mass
- monofluorophosphate
- calcium
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
- A61K38/46—Hydrolases (3)
- A61K38/48—Hydrolases (3) acting on peptide bonds (3.4)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q11/00—Preparations for care of the teeth, of the oral cavity or of dentures; Dentifrices, e.g. toothpastes; Mouth rinses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/047—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates having two or more hydroxy groups, e.g. sorbitol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
- A61K8/24—Phosphorous; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/345—Alcohols containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/55—Phosphorus compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
- A61K8/66—Enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- the invention relates to dentistry and the perfume industry, and in particular to therapeutic and prophylactic compositions for caring for the oral cavity.
- the invention relates to means for maintaining oral hygiene and can be used to prepare therapeutic and prophylactic toothpastes, gels, oral care fluids, as well as other compositions such as chewing gums, jellies, etc.
- a known composition for the prevention of diseases of the teeth and periodontium including sodium fluoride or sodium monofluorophosphate, sodium carboxymethyl cellulose, titanium dioxide, sodium saccharinate, sorbitol or glycerin, silicon dioxide, food coloring, liquid paraffin, perfume and water, chamomile extract, panthenol, calcium glycerophosphate polyvinylpyrroliddone (RU Cl INb 2188626, IPC 7 A61K 7/16, 2002).
- one or more organic surface-active agents are used to achieve improved hydration, foaming and prophylactic action, to help achieve thorough and complete dispersion of the composition throughout the oral cavity.
- Organic surfactant is anionic, nonionic or ampholytic in nature.
- a detergent material is used as a surface-active agent, which imparts detergent and foaming properties to this composition.
- Various other materials can be incorporated into the oral preparations of this invention, such as whitening agents, preservatives, silicones, chlorophyll compounds, other anti-stone agents and / or ammonia-containing materials such as urea, diammonium phosphate and mixtures thereof.
- the known solution provides for a therapeutic and prophylactic agent in various forms: paste, gel, rinse aid, tablet, chewing composition.
- the manifestation of the therapeutic effect of remineralization is prevented by soft dental deposits, bacterial plaque on the surfaces of the teeth and on the soft tissues of the oral cavity.
- the components contained in the known formulations do not ensure its effective removal for a long period of time, since the components do not include those whose function is to destroy plaque and which remain for a period of time between repeated brushing procedures. Accordingly, bacterial plaque contributes to the occurrence of diseases of the teeth and periodontal disease, prevents the processes of mineral metabolism in the oral cavity, both natural and due to the special use of drugs to strengthen the teeth.
- a therapeutic and prophylactic oral composition prepared according to the invention provides effective cleaning, which in turn opens the way for remineralization of tooth tissues.
- Composition delays the formation of bacterial plaque on the teeth, at least for 10-12 hours, increases enamel resistance and reduces inflammation in the periodontium, since the delay in the appearance of soft plaque on all surfaces of the teeth creates conditions for saturation of tooth enamel with mineral components of saliva, as well as Helps reduce microbial stress on periodontal tissues.
- the composition does not contain synthetic antibacterial drugs and rough abrasives.
- the immediate problem that was solved when creating the invention was to develop a highly effective composition for the prevention of dental diseases of teeth and soft tissues of the oral cavity, in which synergist drugs would enhance each other's action, providing a high therapeutic and prophylactic effect and achieving the above results.
- the therapeutic and prophylactic composition for caring for the oral cavity, containing active and inert components acceptable for oral use includes, among the active components, bromelain in an amount of 0.01 ⁇ l wt.% And xylitol in an amount of l, 5 ⁇ 20 wt.%.
- bromelain is a group of high molecular weight glycoproteins (H. R.
- bromelain is discovered in 1950 by Dr. Heinike.
- the bromelain enzyme provides effective removal of bacterial plaque, has anti-inflammatory and immunocorrective effects, which are associated with both direct proteolytic action of the enzyme and the regulatory action of its utilization products (peptide fragments).
- the enzyme is actively resorbed by the mucosa throughout the gastrointestinal tract. Due to the presence of protease inhibitors, bromelain is safe for viable tissues.
- Bromelain is used to ease inflammation in injuries, to prevent swelling of soft tissues, and to accelerate their recovery from injuries and other injuries. Oral administration of bromelain helps to reduce inflammatory processes and edema, accelerates tissue repair processes. Due to the breakdown of non-viable proteins, bromelain accelerates the healing of wounds and trophic ulcers, helping to cleanse them of necrotic masses. Bromelain has an immunocorrective effect, it is used to facilitate inflammatory processes in injuries, to prevent swelling of soft tissues, and accelerates tissue repair processes, which is associated with a modification of vascular permeability and its ability to influence the metabolism of arachidonic acid.
- bromelain in protecting against viral infections, including influenza viruses (A and B), has been intensively studied (Ivapova V.T. et al. / Vorr.Vigasol. 2003 ser-opt; 48 (5): 14- 8). Bromelain is retained in the oral cavity for a long time, inhibiting the deposition of new plaque, improves the condition of the soft tissues of the oral cavity. This effect is achieved due to the influence of bromelain due to the action of two mechanisms: inhibiting the formation of plaque, it reduces the microbial load on the gums; bromelain, on the other hand, provides an anti-inflammatory effect.
- xylitol The main function of xylitol is to modulate the permeability of tooth enamel.
- the mechanism of involvement of this substance in the biochemical exchange of streptococci is characterized as lethal synthesis, and therefore, xylitol reduces the activity of pathogenic microorganisms and improves the condition of the oral organs, which contributes to the process of remineralization of the teeth.
- Xylitol increases the remineralizing potential, since it promotes the inclusion of calcium in tooth enamel, inhibits plaque formation and reduces the cariogenic potential of microflora.
- xylitol improves taste characteristics, being a polyhydric alcohol, performs the function of a water-retaining component.
- the absence of soft dental deposits provides the teeth with almost constant availability of mineral components contained in toothpaste and saliva. Providing long-term protection against plaque formation, the composition helps prevent the occurrence and development of major dental diseases.
- a remineralizing system is a combination of ingredients that allows minerals to saturate the enamel of healthy teeth and the initial foci of a carious lesion.
- the composition of the active components contains an anti-caries mineral supplement in an amount of 0.05-3.0 wt.%. In the best case, the anti-caries mineral supplement is contained in an amount of 0.2-2.5 wt.%.
- Calcium glycerophosphate and an inorganic or organic salt of magnesium are used as an anti-caries mineral supplement.
- a tooth cleared of plaque is most susceptible to the effects of calcium, phosphorus and magnesium, which can be introduced into the therapeutic composition.
- Calcium and phosphorus are the main building blocks of tooth enamel and are involved in metabolic processes throughout a person’s life.
- the need for additives is due to the need for teeth in these elements during caries and, to an even greater extent, for non-carious lesions of the teeth.
- Non-carious lesions of the teeth are also most often associated with impaired calcium metabolism and occur with adverse effects of an endogenous nature (dysfunction of the thyroid gland, pancreas, sex glands, diseases of the gastrointestinal tract, etc.), as well as under the influence of external adverse effects (ionizing radiation, daily long - more than 6 hours - work with a computer, the influence of production factors: acid fumes, metal dust and their combinations; a number of negative environmental influences), causing their noticeable loss of mineral components in the tissues of the teeth, and then to damage in the form of caries, erosion, wedge-shaped defects, abrasion of the teeth.
- the local use of therapeutic and prophylactic compositions containing calcium-phosphorus additives allows not only to prevent, but also to some extent, compensate for losses in the presence of the disease.
- Calcium glycerophosphate is a source of active intake of phosphorus and calcium in the teeth and periodontal tissues, which accelerates the mineralization processes and increases the anti-carious effect of the composition, enhances the anabolic processes in the tissues.
- Magnesium (from the composition of inorganic or organic salts) is a structural component of teeth and is introduced into the complex as a microelement, which is a cofactor for phosphatases that ensure the inclusion of phosphates in the hard tissues of teeth. Under the influence of phosphatases, hydrolysis of glycerophosphate occurs and its bioavailability increases accordingly.
- the composition may not include fluorides.
- fluorides The presence of fluoride in the composition for caring for the oral cavity is relevant in a situation where soft plaque during the decomposition of easily fermentable carbohydrates produces organic acids.
- the local effect of fluorides is practically not felt, which is reflected in a decrease in their effectiveness for the prevention of caries.
- the proposed composition admits that sodium monofluorophosphate, potassium monofluorophosphate, calcium monofluorophosphate or magnesium monofluorophosphate in an amount of 0.5-1.5 wt.% Are used as an anti-caries mineral supplement.
- the optimum concentration is 0.8-1.1 wt.%.
- a therapeutic and prophylactic composition for caring for the oral cavity in the form of a paste from among inert components contains the following, wt.% Abrasive component - 5-40, moisture-retaining and / or moisturizing component - 5-70, gel-forming component - 0, 5-2.5, at least one surfactant - 0.5-3.0, at least one flavoring agent - 0.5-2, at least one preservative is 0.01-0.5.
- the composition contains, by mass.%, A water-retaining and / or moisturizing component, 5-70, a gel-forming component, 0.5-3.5, at least one surfactant, 0.5 -3.0, at least one flavoring filler - 0.5-2 wt.%, At least one preservative - 0.01-0.5.
- the composition For a liquid form from among inert components, the composition contains, max.% Moisturizing component, 5-70, at least one surfactant, 0.5-3.0, at least one flavoring agent, 0, 5-2, water - the rest.
- the composition For a chewing gum form from among the inert components, the composition contains, wt.% Polymer base, 20-30, at least one polyhydric alcohol, 45-60, at least one flavoring filler, 0.5-2.
- the composition For a jelly-shaped form of inert components, the composition contains, wt.% Fruit sugar, 20-50, at least one structure-forming agent, 1-3, at least one flavoring filler, 0.2-1.
- one or several substances selected from the group can be used as an abrasive component: calcium carbonate, dicalcium phosphate, silicon oxide, aluminum oxide, calcium pyrophosphate, sodium metaphosphate, polymethacrylate, magnesium carbonate.
- an abrasive component calcium carbonate, dicalcium phosphate, silicon oxide, aluminum oxide, calcium pyrophosphate, sodium metaphosphate, polymethacrylate, magnesium carbonate.
- one or more substances of the group can be used as a water-retaining component: sorbitol, glycerin, polyethylene glycol.
- sorbitol glycerin
- polyethylene glycol polyethylene glycol
- carboxymethyl cellulose, hydroxyethyl cellulose, xanthan gum, carrageenan, guar gum can be used as a gel-forming component of a paste or gel.
- one or more substances from the following group can be used: methylparaben, propylparaben, butylparaben or their sodium salts, as well as phenoxyethanol, benzoic acid, sodium benzoate.
- the following surfactants can be used: sodium lauryl sulfate, alkylamidobetaine, PEG-40 hydrogenated castor oil (polyoxyethylene (40) hydrogenated castor oil), polysorbate-20 (polyoxyethylene sorbitan monolaurate).
- one or more substances of the following group can be used as a flavoring filler: oils - peppermint, curly mint, citrus oils, eucalyptus, fir, pear, anise, clove, sage, menthol, carvone, anethole, methyl salicylate, sweeteners - sodium saccharin, lactose, maltose, aspartame, sodium cyclamate.
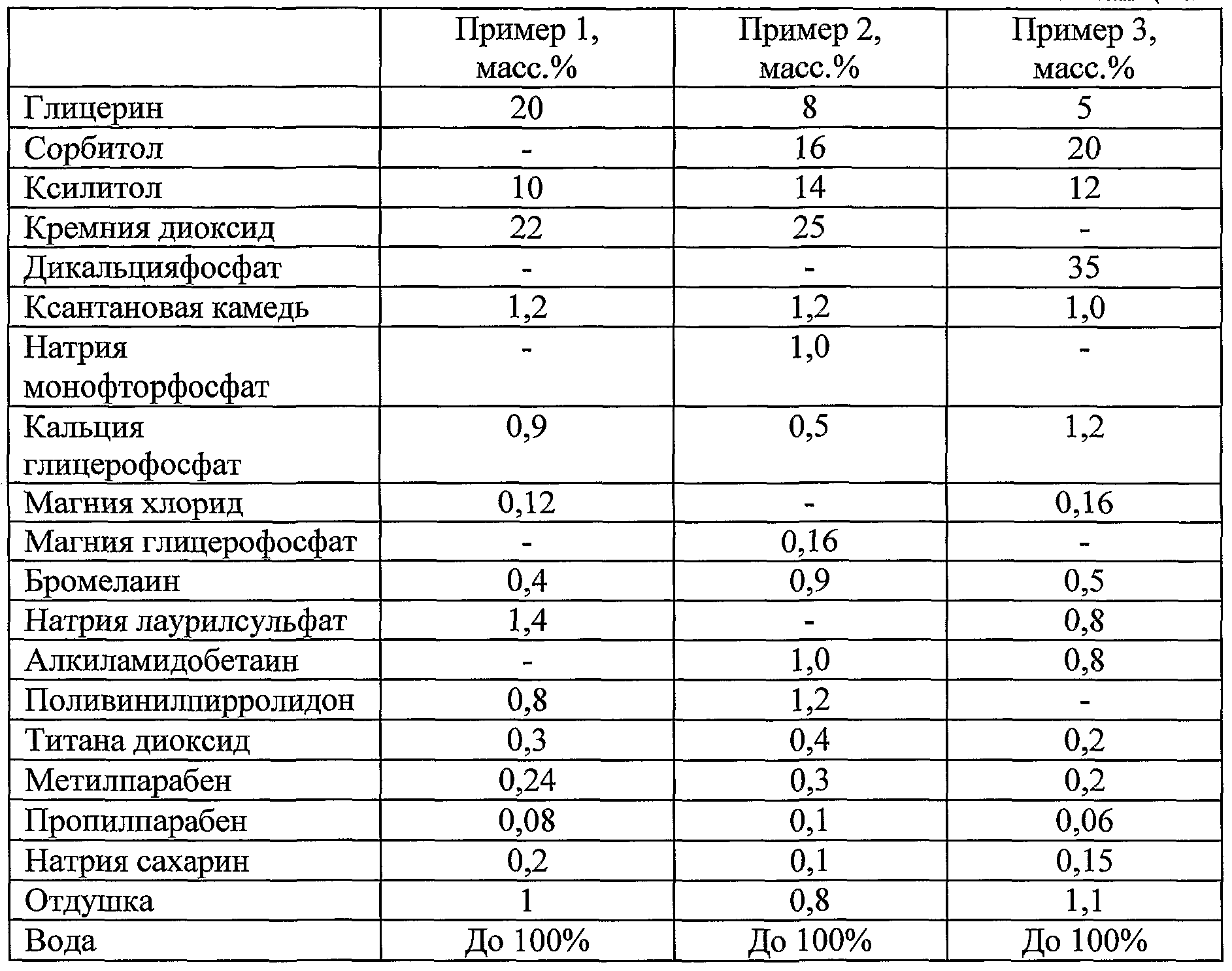
- Toothpaste is prepared as follows.
- the resulting solution is loaded with a suspension of components in glycerol and mixed until a homogeneous mass is formed.
- the composition is evacuated and stirred for 10 minutes until the air is completely removed from the mixture.
- titanium dioxide is added to the mixer and re-vacuum with stirring for 10 minutes until the air is completely removed from the mixture.
- silicon dioxide is added, again vacuumized and stirred for 30-40 minutes.
- the toothpaste is homogenized using a homogenizer pump for 15-20 minutes. Then cool the toothpaste with stirring to a temperature of 40-45 0 C.
- bromelain is loaded and mixed until a homogeneous mass is formed.
- perfume and sodium lauryl sulfate (or amidopropyl betaine in the case of Example 1) are charged and mixed for 30 minutes until a homogeneous mass is obtained.
- the prepared toothpaste is packaged in tubes of polymer material.
- composition in the form of a gel is prepared as follows. Heat the required amount of water in the measuring device - dispenser to
- the mixture is stirred until a clear solution is formed for 20 minutes.
- a suspension of bromelain, hydroxyethyl cellulose, guar gum, calcium glycephosphate in glycerin is separately prepared.
- the aqueous solution of the components is cooled to 50-55 0 C, a suspension of bromelain is added to it and stirred until a homogeneous mass is formed for 30 minutes.
- PEG-40 and hydrogenated castor oil are heated separately to 50-55 0 C, perfume is added and mixed until a homogeneous mixture is formed for 10 minutes.
- the resulting mixture is added to the component solution obtained in the first stage and mixed until a homogeneous gel is obtained for 20-30 minutes.
- the resulting gel is cooled with stirring to a temperature of 20 - 25 0 C and Packed in tubes of polymer material.
- composition in the form of a rinse is prepared as follows. Heat the required amount of water in the measuring device - dispenser to
- magnesium chloride or magnesium glycerophosphate in the case of example 2
- xylitol polyvinylpyrrolidone
- sodium saccharin sodium monofluorophosphate
- sorbitol in the case of examples 1 and 3
- a suspension of bromelain, hydroxyethyl cellulose in glycerol (or propylene glycol in the case of Example 2) is prepared, which is added to the composition obtained in the first stage cooled to 50-55 ° C. The mixture is stirred until a clear solution is formed for 20 minutes.
- PEG-40 hydrogenated castor oil is heated separately to 50-55 0 C, perfume is added, mixed until a homogeneous mixture is formed for 10 minutes and added to the mixture obtained in the previous step at a temperature of 50-55 0 C. The composition is mixed until a clear solution is obtained for 20 minutes.
- Alkylamidobetaine (or sodium lauryl sulfate in the case of Example 3) is added, stirred until a clear solution forms for 20 minutes, which is cooled with stirring to a temperature of 20-25 ° C and bottled.
- compositions presented in table 4 The possibility of the implementation of therapeutic and prophylactic chewing gum is illustrated by examples of the compositions presented in table 4.
- the composition in the form of chewing gum is prepared as follows.
- the base is melted at a temperature of 90 - 95 0 C, overloaded into a mixer equipped with Z-shaped mixers, and cooled to a temperature of 8O 0 C.
- Lecithin, calcium carbonate, magnesium chloride, calcium glycerophosphate are charged and stirred for 10 minutes, non-crystallizing sorbitol is added and stirred for 10 minutes, mannitol and xylitol are added, stirred for 10 minutes, crystalline sorbitol is added, sodium saccharin, starch hydrolyzate are mixed, stirred for 10 minutes.
- the mixture is cooled to a temperature of 55 0 C, add perfume, a prepared suspension of bromelain in glycerin and mix for 20 minutes.
- the mass is unloaded, rolled, cut into plates or cubes of the required size, dried to the required humidity and packaged.
- composition in the form of jelly (marmalade) is prepared as follows.
- sugar-apple mixture When boiling marmalade masses from the sugar-apple mixture, excess moisture is removed at a temperature of 105-107 ° C.
- Sugar-apple mixture has an initial moisture content of more than 45%, sugar-syrup-agar syrup 30-33%.
- the final moisture content of marmalade mass ranges from 24-33% and depends on the formulation and type of products.
- Sugar-syrup-agar syrup is boiled in a coil cooking column until the moisture content is 27-28%, xylitol is added, mixed and cooled to a temperature of 40-50 ° C.
- Aromatic substances calcium glycerophosphate, magnesium chloride, as well as a suspension of bromelain in glycerin are added to the chilled syrup.
- the syrup is gelled into molds. Forms stand, jelly is extracted from them, cut into slices and packaged.
- the effectiveness of the therapeutic and prophylactic toothpastes prepared in accordance with the invention was tested on a group of volunteers in order to evaluate the hygienic and anti-inflammatory effects.
- the compositions were used in examples 1 to 3 of table 6, subject to daily two-time brushing for 2-3 minutes.
- Clinical studies of toothpaste included the study of the cleansing effect and anti-inflammatory effect, assessment of a possible allergenic and locally irritating effect. The study was carried out for three months on the basis of the blind method, that is, when the dentist-epidemiologist who performed the dental examination did not know which group this patient belongs to. The clinical study involved 10 people aged 27 to 42 years.
- IG gingivitis index
- the initial state of periodontal tissues in individuals participating in the testing corresponded to mild and moderate gingivitis.
- the gingivitis index during initial examinations 1 D 3 + 0.12 in the first series, 1.16 + 0.21 - in the second series and 1D 1 + 0.11 - in the third series.
- the decrease in inflammation in periodontal tissues was 18.6% during the period of use of toothpaste with a maximum concentration of bromelain; 38.8% during the period of application of toothpaste with a concentration of bromelain 0.3%; 28.8%, respectively, during the application of the paste containing OD% bromelain.
- the method of acid enamel biopsy according to V.K. Leontiev, V.A. Distil including applying to the enamel a strictly defined amount of demineralizing liquid, collecting it after a certain period of time and subsequent determination of the amount of calcium in the acid demineralizate, makes it possible to determine the acid solubility rate of enamel. Quantitative analysis of the content in ⁇ g / l of calcium and phosphorus in the acid biopsy is carried out by spectrophotometry.
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008542267A JP5684454B2 (ja) | 2005-11-25 | 2005-11-25 | 口腔ケア用の治療及び予防組成物 |
| US12/094,605 US20090016972A1 (en) | 2005-11-25 | 2005-11-25 | Oral cavity care curative and prophylactic composition |
| KR1020087015475A KR101234172B1 (ko) | 2005-11-25 | 2005-11-25 | 구강 치료 및 예방 조성물 |
| PCT/RU2005/000601 WO2007061328A1 (fr) | 2005-11-25 | 2005-11-25 | Composition de soins et de prevention pour la cavite buccale |
| EP05857405.4A EP1952801B1 (en) | 2005-11-25 | 2005-11-25 | Oral cavity care curative and prophylactic composition |
| CNA2005800525225A CN101370475A (zh) | 2005-11-25 | 2005-11-25 | 用于口腔护理的治疗和预防制剂 |
| EA200701677A EA011125B1 (ru) | 2005-11-25 | 2005-11-25 | Лечебно-профилактический состав для ухода за полостью рта |
| NO20082820A NO342401B1 (no) | 2005-11-25 | 2008-06-25 | Legende og profylaktisk sammensetning for pleie av munnhulen |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/RU2005/000601 WO2007061328A1 (fr) | 2005-11-25 | 2005-11-25 | Composition de soins et de prevention pour la cavite buccale |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007061328A1 true WO2007061328A1 (fr) | 2007-05-31 |
Family
ID=38067455
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/RU2005/000601 WO2007061328A1 (fr) | 2005-11-25 | 2005-11-25 | Composition de soins et de prevention pour la cavite buccale |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20090016972A1 (ru) |
| EP (1) | EP1952801B1 (ru) |
| JP (1) | JP5684454B2 (ru) |
| KR (1) | KR101234172B1 (ru) |
| CN (1) | CN101370475A (ru) |
| EA (1) | EA011125B1 (ru) |
| NO (1) | NO342401B1 (ru) |
| WO (1) | WO2007061328A1 (ru) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA010030B1 (ru) * | 2007-07-25 | 2008-06-30 | Общество С Ограниченной Ответственностью "Вдс" | Зубная паста |
| EA012247B1 (ru) * | 2008-07-07 | 2009-08-28 | Общество С Ограниченной Ответственностью "Вдс" | Лечебно-профилактический состав для ухода за полостью рта |
| EP2180895A1 (en) * | 2007-08-30 | 2010-05-05 | Prelief Inc. | Methods for improving healing of an oral lesion using a glycerophosphate salt |
| WO2014059679A1 (en) * | 2012-10-19 | 2014-04-24 | Colgate-Palmolive Company | Processes for preparing toothpaste compositions |
| EP2249795A4 (en) * | 2008-02-07 | 2015-09-23 | Obthestvo S Ogranichennoyj Otvetstvennostjyu Wds | GEL FOR THE REMINERALIZATION OF DENTAL TISSUE |
| RU2641570C2 (ru) * | 2015-01-21 | 2018-01-18 | ОБЩЕСТВО С ОГРАНИЧЕННОЙ ОТВЕТСТВЕННОСТЬЮ "НоваМедика" | Гель, включающий нифедипин и лидокаина гидрохлорид (варианты), применение геля, включающего нифедипин и лидокаина гидрохлорид (варианты), способ приготовления геля, включающего нифедипин и лидокаина гидрохлорид, с использованием нанотехнологии |
Families Citing this family (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PL2705826T3 (pl) | 2006-02-09 | 2020-11-02 | The University Of Melbourne | Kompozycja fluorku i sposoby mineralizacji zębów |
| JP4985975B2 (ja) * | 2007-12-20 | 2012-07-25 | ライオン株式会社 | 口腔用組成物 |
| EA200900764A1 (ru) * | 2009-05-15 | 2010-04-30 | Общество С Ограниченной Ответственностью "Вдс" | Состав для профилактики стоматологических заболеваний |
| JP5397204B2 (ja) * | 2009-12-15 | 2014-01-22 | ライオン株式会社 | 口腔用組成物 |
| EA020907B1 (ru) * | 2011-07-25 | 2015-02-27 | Абдухамит Абдувалиевич Фаттахов | Лечебно-профилактический состав для ухода за полостью рта |
| KR102102130B1 (ko) * | 2012-10-01 | 2020-04-20 | 라이온 가부시키가이샤 | 치마제 조성물 및 치아의 재석회화 촉진제 |
| CN103040636A (zh) * | 2012-12-31 | 2013-04-17 | 龙启知 | 防龋牙膏 |
| WO2014124950A1 (en) * | 2013-02-14 | 2014-08-21 | Glaxo Group Limited | Novel composition |
| EP2956113B1 (en) * | 2013-02-14 | 2019-04-03 | Unilever Plc. | Non-aqueous oral care compositions |
| CA2918441C (en) | 2013-07-23 | 2023-03-21 | The University Of Melbourne | A base and phosphopeptide stabilized amorphous calcium phosphate (acp) and/or amorphous calcium fluoride phosphate (acfp) for use in promoting remineralisation |
| CN103385241A (zh) * | 2013-08-19 | 2013-11-13 | 广州博嘉生物科技有限公司 | 一种复合防腐剂及其用途 |
| US20150287997A1 (en) * | 2013-09-24 | 2015-10-08 | Nanyang Technological University | Nanofiber and use thereof in an electrode |
| FR3011466A1 (fr) * | 2013-10-09 | 2015-04-10 | Roquette Freres | Utilisation d'un polyol dans la remineralisation de l'email |
| EP3086751B1 (en) | 2013-12-24 | 2023-07-12 | The University Of Melbourne | Stabilized stannous compositions |
| CA2953099C (en) | 2014-07-21 | 2021-06-08 | Colgate-Palmolive Company | Abrasive oral care composition |
| US20180008518A1 (en) * | 2014-12-24 | 2018-01-11 | The University Of Melbourne | Mineralization fluoride compositions |
| JP2016204330A (ja) * | 2015-04-27 | 2016-12-08 | サンスター株式会社 | 舌苔除去用ゲルを調製するための組成物、及び舌苔除去用ゲル |
| GB2544782B (en) * | 2015-11-26 | 2020-07-15 | Fontus Health Ltd | Solution |
| CN105726408A (zh) * | 2016-03-28 | 2016-07-06 | 北京世纪鸿成商贸有限公司 | 牙凝胶 |
| CN106074305B (zh) * | 2016-07-29 | 2019-08-23 | 广东俏脸谱文化艺术有限公司 | 一种具有口腔粘膜保护作用的诺丽果牙膏及其制备方法 |
| CN107802641A (zh) * | 2016-09-09 | 2018-03-16 | 李明典 | 口腔软组织(牙龈、粘膜)抗发炎酸痛剂 |
| MY197428A (en) | 2017-03-14 | 2023-06-16 | Univ Melbourne | Treatment for gingivitis |
| FR3070856A1 (fr) * | 2017-09-11 | 2019-03-15 | Yvan ERBS | Produit de soin pour reduire les risques de survenance d'au moins une maladie ou un trouble de la sante |
| JP2020059689A (ja) * | 2018-10-09 | 2020-04-16 | 炭プラスラボ株式会社 | 経口組成物 |
| KR102072442B1 (ko) * | 2019-02-27 | 2020-02-03 | 임이종 | 미네랄 칼슘을 포함하는 구강청결제 및 그 제조방법 |
| KR102067213B1 (ko) * | 2019-02-27 | 2020-01-16 | 임이종 | 미네랄 칼슘을 포함하는 치약 및 그 제조방법 |
| EP3733154A1 (en) * | 2019-05-03 | 2020-11-04 | Omya International AG | Magnesium ion-containing materials as white pigments in oral care compositions |
| JP7439352B2 (ja) | 2019-12-26 | 2024-02-28 | オブスチェストヴォ エス オグラニチェノイ オトヴェットストヴェノスティユ ≪ダブリューディーエス≫ | 歯のエナメル質を再石灰化するための組成物 |
| US20230120190A1 (en) * | 2020-01-29 | 2023-04-20 | Marcelo Rodrigues Teixeira | Oral composition with synergistic association of organic and inorganic components for full maintenance of oral health, process for obtaining and uses thereof |
| CN112274441A (zh) * | 2020-11-03 | 2021-01-29 | 成都大学 | 一种具有美白自修复功能的牙齿抛光膏及其制备方法与应用 |
| CN114652642A (zh) * | 2022-05-09 | 2022-06-24 | 广东新宝电器股份有限公司 | 一种口腔清洁护理组合物 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998002135A1 (en) * | 1996-07-15 | 1998-01-22 | Gillette Canada Inc. | Stabilized stannous-containing compositions for oral care |
| RU2188627C1 (ru) * | 2001-04-26 | 2002-09-10 | ООО НПО "Фитофарм" | Состав для профилактики заболеваний полости рта |
| RU2204379C1 (ru) * | 2002-05-08 | 2003-05-20 | Закрытое акционерное общество "Модум" | Зубная паста |
Family Cites Families (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2726490B2 (ja) * | 1989-05-10 | 1998-03-11 | 花王株式会社 | 口腔用組成物 |
| GB2289841B (en) * | 1994-05-23 | 1998-04-15 | Janina International | Oral care products |
| GB2290234B (en) * | 1994-06-07 | 1998-06-10 | Janina International | Oral care products |
| JPH09295924A (ja) * | 1996-04-30 | 1997-11-18 | Lion Corp | 歯垢の酸中和剤及び口腔用組成物 |
| DE19854086A1 (de) * | 1998-11-24 | 2000-05-25 | Henkel Kgaa | Gestreifte Zahnpasten |
| US6379654B1 (en) * | 2000-10-27 | 2002-04-30 | Colgate Palmolive Company | Oral composition providing enhanced tooth stain removal |
| US6723304B2 (en) * | 2001-11-13 | 2004-04-20 | Noville, Inc. | Oral care compositions comprising diglycerol |
| JP3853785B2 (ja) * | 2002-12-27 | 2006-12-06 | 花王株式会社 | 口腔用組成物 |
| US20050084551A1 (en) * | 2003-09-26 | 2005-04-21 | Jensen Claude J. | Morinda citrifolia-based oral care compositions and methods |
-
2005
- 2005-11-25 KR KR1020087015475A patent/KR101234172B1/ko active IP Right Grant
- 2005-11-25 US US12/094,605 patent/US20090016972A1/en not_active Abandoned
- 2005-11-25 JP JP2008542267A patent/JP5684454B2/ja active Active
- 2005-11-25 WO PCT/RU2005/000601 patent/WO2007061328A1/ru active Application Filing
- 2005-11-25 EP EP05857405.4A patent/EP1952801B1/en active Active
- 2005-11-25 EA EA200701677A patent/EA011125B1/ru active IP Right Revival
- 2005-11-25 CN CNA2005800525225A patent/CN101370475A/zh active Pending
-
2008
- 2008-06-25 NO NO20082820A patent/NO342401B1/no not_active IP Right Cessation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1998002135A1 (en) * | 1996-07-15 | 1998-01-22 | Gillette Canada Inc. | Stabilized stannous-containing compositions for oral care |
| RU2188627C1 (ru) * | 2001-04-26 | 2002-09-10 | ООО НПО "Фитофарм" | Состав для профилактики заболеваний полости рта |
| RU2204379C1 (ru) * | 2002-05-08 | 2003-05-20 | Закрытое акционерное общество "Модум" | Зубная паста |
Non-Patent Citations (4)
| Title |
|---|
| "Zubnaya pasta na travyanoi osnove SanSmail, katalog", SANRAIDER, 30 May 2005 (2005-05-30), XP008185916, Retrieved from the Internet <URL:http://www.shop.alfafarm/RU/product_info.php>products_id=112> * |
| H. R. MAURER, CMLS CELL. MOL. LIFE SCI., vol. 58, 2001, pages 1234 - 1245 |
| IVANOVA V.T. ET AL., VOPR.VIRUSOL, vol. 48, no. 5, September 2003 (2003-09-01), pages 14 - 8 |
| See also references of EP1952801A4 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EA010030B1 (ru) * | 2007-07-25 | 2008-06-30 | Общество С Ограниченной Ответственностью "Вдс" | Зубная паста |
| EP2180895A1 (en) * | 2007-08-30 | 2010-05-05 | Prelief Inc. | Methods for improving healing of an oral lesion using a glycerophosphate salt |
| EP2180895A4 (en) * | 2007-08-30 | 2012-04-11 | Prelief Inc | METHODS FOR IMPROVING THE HEALING OF ORAL INJURY USING GLYCEROPHOSPHATE SALT |
| EP2249795A4 (en) * | 2008-02-07 | 2015-09-23 | Obthestvo S Ogranichennoyj Otvetstvennostjyu Wds | GEL FOR THE REMINERALIZATION OF DENTAL TISSUE |
| EA012247B1 (ru) * | 2008-07-07 | 2009-08-28 | Общество С Ограниченной Ответственностью "Вдс" | Лечебно-профилактический состав для ухода за полостью рта |
| WO2010005338A1 (en) * | 2008-07-07 | 2010-01-14 | Obthestvo S Ogranichennoyj Otvetstvennostjyu «Wds» | Therapeutic and prophylactic formulation for oral care |
| WO2014059679A1 (en) * | 2012-10-19 | 2014-04-24 | Colgate-Palmolive Company | Processes for preparing toothpaste compositions |
| US9254253B2 (en) | 2012-10-19 | 2016-02-09 | Colgate-Palmolive Company | Processes for preparing toothpaste compositions |
| RU2641570C2 (ru) * | 2015-01-21 | 2018-01-18 | ОБЩЕСТВО С ОГРАНИЧЕННОЙ ОТВЕТСТВЕННОСТЬЮ "НоваМедика" | Гель, включающий нифедипин и лидокаина гидрохлорид (варианты), применение геля, включающего нифедипин и лидокаина гидрохлорид (варианты), способ приготовления геля, включающего нифедипин и лидокаина гидрохлорид, с использованием нанотехнологии |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2009517382A (ja) | 2009-04-30 |
| EP1952801A4 (en) | 2015-04-29 |
| NO342401B1 (no) | 2018-05-14 |
| EA200701677A1 (ru) | 2007-12-28 |
| US20090016972A1 (en) | 2009-01-15 |
| EP1952801A1 (en) | 2008-08-06 |
| JP5684454B2 (ja) | 2015-03-11 |
| EP1952801B1 (en) | 2018-07-25 |
| KR101234172B1 (ko) | 2013-02-19 |
| NO20082820L (no) | 2008-06-25 |
| CN101370475A (zh) | 2009-02-18 |
| EA011125B1 (ru) | 2008-12-30 |
| KR20090010154A (ko) | 2009-01-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2007061328A1 (fr) | Composition de soins et de prevention pour la cavite buccale | |
| TWI679991B (zh) | 用於牙齒礦化之組成物及方法 | |
| KR101308920B1 (ko) | 치아의 탈광화를 감소시키기 위한 조성물 및 방법 | |
| US20070098650A1 (en) | Dental formulation | |
| RU2293551C1 (ru) | Состав для профилактики заболеваний полости рта | |
| US9023323B2 (en) | Oral compositions for prevention and reduction of bacterial adhesion to oral surfaces | |
| JP4826790B2 (ja) | 加水分解シルクを含有する口腔用組成物 | |
| US4415550A (en) | Treatment-and-prophylactic tooth paste possessing anticarious effect | |
| AU623283B2 (en) | Oral rinse and method for plaque removal | |
| RU2457829C2 (ru) | Минерально-витаминный комплекс для укрепления эмали зубов, композиция для гигиены полости рта и зубная паста | |
| JP2007197393A (ja) | 歯磨 | |
| WO2000025737A1 (en) | Oral composition exhibiting improved uptake of antibacterial compounds | |
| US11617711B2 (en) | Compositions and methods for dental care | |
| US20050180927A1 (en) | Novel anti-bacterial compositions | |
| KR100704585B1 (ko) | 치약 조성물 | |
| UA85811C2 (ru) | Лечебно-профилактическая смесь для ухода за полостью рта | |
| BR112019021767A2 (pt) | produtos e métodos para o cuidado oral | |
| JPH06219930A (ja) | 口腔用組成物 | |
| RU2535051C1 (ru) | Зубная паста | |
| WO2006110183A2 (en) | Dental formulation | |
| US20220062125A1 (en) | Multipurpose fluorinated biphosphate toothpaste composition | |
| EP1019015B1 (en) | Oral composition exhibiting improved antibacterial uptake and retention | |
| AU2003213864C1 (en) | Novel anti-bacterial compositions | |
| Mason | Dental and mouthwash preparations | |
| JPS6246522B2 (ru) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 200701677 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008542267 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008050860 Country of ref document: EG |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005857405 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020087015475 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580052522.5 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12094605 Country of ref document: US |