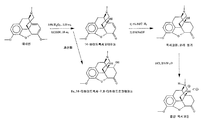
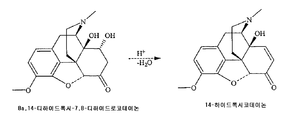
KR20150046393A - 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 - Google Patents
25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 Download PDFInfo
- Publication number
- KR20150046393A KR20150046393A KR1020157009780A KR20157009780A KR20150046393A KR 20150046393 A KR20150046393 A KR 20150046393A KR 1020157009780 A KR1020157009780 A KR 1020157009780A KR 20157009780 A KR20157009780 A KR 20157009780A KR 20150046393 A KR20150046393 A KR 20150046393A
- Authority
- KR
- South Korea
- Prior art keywords
- ppm
- oxycodone
- less
- solution
- oxycodone hydrochloride
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- BQNSLJQRJAJITR-UHFFFAOYSA-N 1,1,2-trichloro-1,2-difluoroethane Chemical compound FC(Cl)C(F)(Cl)Cl BQNSLJQRJAJITR-UHFFFAOYSA-N 0.000 title claims abstract description 119
- 229960003617 oxycodone hydrochloride Drugs 0.000 title claims abstract description 119
- 238000004519 manufacturing process Methods 0.000 title abstract description 32
- YYCRAERBSFHMPL-XFKAJCMBSA-N (4r,4as,7ar,12bs)-4a-hydroxy-9-methoxy-3-methyl-2,4,7a,13-tetrahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-7-one Chemical compound O=C([C@@H]1O2)C=C[C@@]3(O)[C@]4([H])N(C)CC[C@]13C1=C2C(OC)=CC=C1C4 YYCRAERBSFHMPL-XFKAJCMBSA-N 0.000 title description 15
- YYCRAERBSFHMPL-UHFFFAOYSA-N 14beta-Hydroxycodeinone Natural products O1C2C(=O)C=CC3(O)C4CC5=CC=C(OC)C1=C5C23CCN4C YYCRAERBSFHMPL-UHFFFAOYSA-N 0.000 title description 15
- 239000000203 mixture Substances 0.000 claims abstract description 233
- 238000000034 method Methods 0.000 claims description 75
- BRUQQQPBMZOVGD-XFKAJCMBSA-N Oxycodone Chemical compound O=C([C@@H]1O2)CC[C@@]3(O)[C@H]4CC5=CC=C(OC)C2=C5[C@@]13CCN4C BRUQQQPBMZOVGD-XFKAJCMBSA-N 0.000 description 95
- 239000000243 solution Substances 0.000 description 93
- 239000000463 material Substances 0.000 description 86
- 229960002085 oxycodone Drugs 0.000 description 84
- 239000002585 base Substances 0.000 description 57
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 55
- 238000009472 formulation Methods 0.000 description 55
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 54
- 230000002209 hydrophobic effect Effects 0.000 description 47
- 238000013268 sustained release Methods 0.000 description 46
- 239000012730 sustained-release form Substances 0.000 description 46
- 238000000576 coating method Methods 0.000 description 44
- 239000011248 coating agent Substances 0.000 description 39
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 31
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 28
- 239000000523 sample Substances 0.000 description 28
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 27
- 229920003134 Eudragit® polymer Polymers 0.000 description 26
- 229960000240 hydrocodone Drugs 0.000 description 26
- 229920000642 polymer Polymers 0.000 description 26
- 238000004128 high performance liquid chromatography Methods 0.000 description 25
- 239000011159 matrix material Substances 0.000 description 24
- 239000002904 solvent Substances 0.000 description 24
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 23
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 23
- 238000002360 preparation method Methods 0.000 description 23
- 239000012086 standard solution Substances 0.000 description 23
- 238000002347 injection Methods 0.000 description 22
- 239000007924 injection Substances 0.000 description 22
- -1 polyethylene Polymers 0.000 description 21
- 238000004458 analytical method Methods 0.000 description 20
- 238000005984 hydrogenation reaction Methods 0.000 description 19
- 239000011324 bead Substances 0.000 description 18
- 239000004014 plasticizer Substances 0.000 description 18
- 239000003054 catalyst Substances 0.000 description 17
- 239000003795 chemical substances by application Substances 0.000 description 17
- 239000003814 drug Substances 0.000 description 17
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 17
- 238000006243 chemical reaction Methods 0.000 description 16
- 229920001577 copolymer Polymers 0.000 description 16
- 229910052739 hydrogen Inorganic materials 0.000 description 16
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 15
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 15
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 15
- 235000014113 dietary fatty acids Nutrition 0.000 description 15
- 229940079593 drug Drugs 0.000 description 15
- 239000000194 fatty acid Substances 0.000 description 15
- 229930195729 fatty acid Natural products 0.000 description 15
- 230000008569 process Effects 0.000 description 15
- 239000007787 solid Substances 0.000 description 15
- 239000011230 binding agent Substances 0.000 description 14
- OROGSEYTTFOCAN-UHFFFAOYSA-N hydrocodone Natural products C1C(N(CCC234)C)C2C=CC(O)C3OC2=C4C1=CC=C2OC OROGSEYTTFOCAN-UHFFFAOYSA-N 0.000 description 14
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 13
- 239000002253 acid Substances 0.000 description 13
- LLPOLZWFYMWNKH-CMKMFDCUSA-N hydrocodone Chemical compound C([C@H]1[C@H](N(CC[C@@]112)C)C3)CC(=O)[C@@H]1OC1=C2C3=CC=C1OC LLPOLZWFYMWNKH-CMKMFDCUSA-N 0.000 description 13
- 239000002245 particle Substances 0.000 description 13
- 235000002639 sodium chloride Nutrition 0.000 description 13
- 239000008186 active pharmaceutical agent Substances 0.000 description 12
- OROGSEYTTFOCAN-DNJOTXNNSA-N codeine Chemical compound C([C@H]1[C@H](N(CC[C@@]112)C)C3)=C[C@H](O)[C@@H]1OC1=C2C3=CC=C1OC OROGSEYTTFOCAN-DNJOTXNNSA-N 0.000 description 12
- 239000012530 fluid Substances 0.000 description 12
- 150000002430 hydrocarbons Chemical class 0.000 description 12
- 239000010410 layer Substances 0.000 description 12
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 12
- 229920000058 polyacrylate Polymers 0.000 description 12
- 229920001515 polyalkylene glycol Polymers 0.000 description 12
- 239000000126 substance Substances 0.000 description 12
- 239000000829 suppository Substances 0.000 description 12
- 239000006185 dispersion Substances 0.000 description 11
- 150000004665 fatty acids Chemical class 0.000 description 11
- 239000012458 free base Substances 0.000 description 11
- 229930195733 hydrocarbon Natural products 0.000 description 11
- 239000006186 oral dosage form Substances 0.000 description 11
- 150000003839 salts Chemical class 0.000 description 11
- 239000001993 wax Substances 0.000 description 11
- 239000001856 Ethyl cellulose Substances 0.000 description 10
- 229920001249 ethyl cellulose Polymers 0.000 description 10
- 235000019325 ethyl cellulose Nutrition 0.000 description 10
- 229920013821 hydroxy alkyl cellulose Polymers 0.000 description 10
- 239000000049 pigment Substances 0.000 description 10
- 238000003860 storage Methods 0.000 description 10
- 239000003826 tablet Substances 0.000 description 10
- DNIAPMSPPWPWGF-GSVOUGTGSA-N (R)-(-)-Propylene glycol Chemical compound C[C@@H](O)CO DNIAPMSPPWPWGF-GSVOUGTGSA-N 0.000 description 9
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 9
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 9
- 238000004090 dissolution Methods 0.000 description 9
- 239000001257 hydrogen Substances 0.000 description 9
- 239000007858 starting material Substances 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 8
- 239000004480 active ingredient Substances 0.000 description 8
- 229920013820 alkyl cellulose Polymers 0.000 description 8
- 230000015572 biosynthetic process Effects 0.000 description 8
- 229920002678 cellulose Polymers 0.000 description 8
- 239000003153 chemical reaction reagent Substances 0.000 description 8
- XYYVYLMBEZUESM-UHFFFAOYSA-N dihydrocodeine Natural products C1C(N(CCC234)C)C2C=CC(=O)C3OC2=C4C1=CC=C2OC XYYVYLMBEZUESM-UHFFFAOYSA-N 0.000 description 8
- 239000008187 granular material Substances 0.000 description 8
- 239000000314 lubricant Substances 0.000 description 8
- 238000011003 system suitability test Methods 0.000 description 8
- LLPOLZWFYMWNKH-UHFFFAOYSA-N trans-dihydrocodeinone Natural products C1C(N(CCC234)C)C2CCC(=O)C3OC2=C4C1=CC=C2OC LLPOLZWFYMWNKH-UHFFFAOYSA-N 0.000 description 8
- ATRRKUHOCOJYRX-UHFFFAOYSA-N Ammonium bicarbonate Chemical compound [NH4+].OC([O-])=O ATRRKUHOCOJYRX-UHFFFAOYSA-N 0.000 description 7
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 7
- 239000001099 ammonium carbonate Substances 0.000 description 7
- 235000012501 ammonium carbonate Nutrition 0.000 description 7
- 239000001913 cellulose Substances 0.000 description 7
- 235000010980 cellulose Nutrition 0.000 description 7
- 238000005755 formation reaction Methods 0.000 description 7
- 239000004615 ingredient Substances 0.000 description 7
- 239000012528 membrane Substances 0.000 description 7
- 230000003204 osmotic effect Effects 0.000 description 7
- 230000003647 oxidation Effects 0.000 description 7
- 238000007254 oxidation reaction Methods 0.000 description 7
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 6
- 239000012298 atmosphere Substances 0.000 description 6
- 229920002301 cellulose acetate Polymers 0.000 description 6
- 229960004126 codeine Drugs 0.000 description 6
- 150000001875 compounds Chemical class 0.000 description 6
- 239000011162 core material Substances 0.000 description 6
- 230000018044 dehydration Effects 0.000 description 6
- 238000006297 dehydration reaction Methods 0.000 description 6
- 230000001419 dependent effect Effects 0.000 description 6
- 239000003085 diluting agent Substances 0.000 description 6
- 235000019441 ethanol Nutrition 0.000 description 6
- 239000007903 gelatin capsule Substances 0.000 description 6
- 239000008240 homogeneous mixture Substances 0.000 description 6
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 6
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 6
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 6
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 6
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 6
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 6
- 229910052763 palladium Inorganic materials 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 239000012488 sample solution Substances 0.000 description 6
- 238000003756 stirring Methods 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 5
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 5
- 241000209094 Oryza Species 0.000 description 5
- 235000007164 Oryza sativa Nutrition 0.000 description 5
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 5
- DOOTYTYQINUNNV-UHFFFAOYSA-N Triethyl citrate Chemical compound CCOC(=O)CC(O)(C(=O)OCC)CC(=O)OCC DOOTYTYQINUNNV-UHFFFAOYSA-N 0.000 description 5
- 238000010521 absorption reaction Methods 0.000 description 5
- 229960000583 acetic acid Drugs 0.000 description 5
- 239000000654 additive Substances 0.000 description 5
- 239000008367 deionised water Substances 0.000 description 5
- 229910021641 deionized water Inorganic materials 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 150000002191 fatty alcohols Chemical class 0.000 description 5
- 238000001914 filtration Methods 0.000 description 5
- 210000004051 gastric juice Anatomy 0.000 description 5
- 239000008101 lactose Substances 0.000 description 5
- 238000002844 melting Methods 0.000 description 5
- 230000008018 melting Effects 0.000 description 5
- 229920003145 methacrylic acid copolymer Polymers 0.000 description 5
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 5
- 231100000252 nontoxic Toxicity 0.000 description 5
- 230000003000 nontoxic effect Effects 0.000 description 5
- 239000008188 pellet Substances 0.000 description 5
- 239000000546 pharmaceutical excipient Substances 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 235000009566 rice Nutrition 0.000 description 5
- 238000000967 suction filtration Methods 0.000 description 5
- 239000002511 suppository base Substances 0.000 description 5
- 239000001069 triethyl citrate Substances 0.000 description 5
- VMYFZRTXGLUXMZ-UHFFFAOYSA-N triethyl citrate Natural products CCOC(=O)C(O)(C(=O)OCC)C(=O)OCC VMYFZRTXGLUXMZ-UHFFFAOYSA-N 0.000 description 5
- 235000013769 triethyl citrate Nutrition 0.000 description 5
- 239000004215 Carbon black (E152) Substances 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 229920002472 Starch Polymers 0.000 description 4
- 239000013543 active substance Substances 0.000 description 4
- 150000001298 alcohols Chemical class 0.000 description 4
- 230000008878 coupling Effects 0.000 description 4
- 238000010168 coupling process Methods 0.000 description 4
- 238000005859 coupling reaction Methods 0.000 description 4
- 238000002425 crystallisation Methods 0.000 description 4
- 230000008025 crystallization Effects 0.000 description 4
- FLKPEMZONWLCSK-UHFFFAOYSA-N diethyl phthalate Chemical compound CCOC(=O)C1=CC=CC=C1C(=O)OCC FLKPEMZONWLCSK-UHFFFAOYSA-N 0.000 description 4
- 239000000975 dye Substances 0.000 description 4
- 210000001035 gastrointestinal tract Anatomy 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 239000000017 hydrogel Substances 0.000 description 4
- 125000005395 methacrylic acid group Chemical group 0.000 description 4
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 4
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 4
- 230000035699 permeability Effects 0.000 description 4
- 229920000233 poly(alkylene oxides) Polymers 0.000 description 4
- 229920001451 polypropylene glycol Polymers 0.000 description 4
- 229920005990 polystyrene resin Polymers 0.000 description 4
- 238000001694 spray drying Methods 0.000 description 4
- 239000008107 starch Substances 0.000 description 4
- 235000019698 starch Nutrition 0.000 description 4
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 4
- 150000003626 triacylglycerols Chemical class 0.000 description 4
- 239000003643 water by type Substances 0.000 description 4
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 3
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 3
- 229920000178 Acrylic resin Polymers 0.000 description 3
- 239000004925 Acrylic resin Substances 0.000 description 3
- 241000599985 Beijerinckia mobilis Species 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 239000005977 Ethylene Substances 0.000 description 3
- 108010010803 Gelatin Proteins 0.000 description 3
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 3
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 3
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 3
- 239000004677 Nylon Substances 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 229920001800 Shellac Polymers 0.000 description 3
- 235000021355 Stearic acid Nutrition 0.000 description 3
- 230000000996 additive effect Effects 0.000 description 3
- 239000000556 agonist Substances 0.000 description 3
- 125000000217 alkyl group Chemical group 0.000 description 3
- 239000007864 aqueous solution Substances 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 239000004359 castor oil Substances 0.000 description 3
- 235000019438 castor oil Nutrition 0.000 description 3
- 229920003086 cellulose ether Polymers 0.000 description 3
- 238000013375 chromatographic separation Methods 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 239000002552 dosage form Substances 0.000 description 3
- 238000001125 extrusion Methods 0.000 description 3
- 239000012467 final product Substances 0.000 description 3
- 235000019253 formic acid Nutrition 0.000 description 3
- 239000003205 fragrance Substances 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 239000008273 gelatin Substances 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 235000011852 gelatine desserts Nutrition 0.000 description 3
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 3
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 3
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 3
- 239000004922 lacquer Substances 0.000 description 3
- 235000019359 magnesium stearate Nutrition 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- 239000000155 melt Substances 0.000 description 3
- 238000007909 melt granulation Methods 0.000 description 3
- 210000004379 membrane Anatomy 0.000 description 3
- 229940117841 methacrylic acid copolymer Drugs 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 230000007935 neutral effect Effects 0.000 description 3
- 229920001778 nylon Polymers 0.000 description 3
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 3
- 239000002357 osmotic agent Substances 0.000 description 3
- 230000001590 oxidative effect Effects 0.000 description 3
- MUZQPDBAOYKNLO-RKXJKUSZSA-N oxycodone hydrochloride Chemical compound [H+].[Cl-].O=C([C@@H]1O2)CC[C@@]3(O)[C@H]4CC5=CC=C(OC)C2=C5[C@@]13CCN4C MUZQPDBAOYKNLO-RKXJKUSZSA-N 0.000 description 3
- 239000008194 pharmaceutical composition Substances 0.000 description 3
- 230000000704 physical effect Effects 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 239000004810 polytetrafluoroethylene Substances 0.000 description 3
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 3
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 3
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 3
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 3
- 125000001453 quaternary ammonium group Chemical group 0.000 description 3
- 230000035484 reaction time Effects 0.000 description 3
- 239000012087 reference standard solution Substances 0.000 description 3
- 238000012216 screening Methods 0.000 description 3
- 235000013874 shellac Nutrition 0.000 description 3
- 239000004208 shellac Substances 0.000 description 3
- 229940113147 shellac Drugs 0.000 description 3
- ZLGIYFNHBLSMPS-ATJNOEHPSA-N shellac Chemical compound OCCCCCC(O)C(O)CCCCCCCC(O)=O.C1C23[C@H](C(O)=O)CCC2[C@](C)(CO)[C@@H]1C(C(O)=O)=C[C@@H]3O ZLGIYFNHBLSMPS-ATJNOEHPSA-N 0.000 description 3
- 239000002002 slurry Substances 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 229910052708 sodium Inorganic materials 0.000 description 3
- 239000010421 standard material Substances 0.000 description 3
- 239000008117 stearic acid Substances 0.000 description 3
- 210000002784 stomach Anatomy 0.000 description 3
- 230000002459 sustained effect Effects 0.000 description 3
- 239000004408 titanium dioxide Substances 0.000 description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 description 3
- 239000008158 vegetable oil Substances 0.000 description 3
- 229920003169 water-soluble polymer Polymers 0.000 description 3
- 239000012224 working solution Substances 0.000 description 3
- JHPBZFOKBAGZBL-UHFFFAOYSA-N (3-hydroxy-2,2,4-trimethylpentyl) 2-methylprop-2-enoate Chemical compound CC(C)C(O)C(C)(C)COC(=O)C(C)=C JHPBZFOKBAGZBL-UHFFFAOYSA-N 0.000 description 2
- GXMDLXQESSRSFB-ZWKGOLINSA-N (4r,4as,7ar,12bs)-4a-hydroxy-9-methoxy-3-methyl-3-oxido-2,4,5,6,7a,13-hexahydro-1h-4,12-methanobenzofuro[3,2-e]isoquinoline-3-ium-7-one Chemical compound C([C@@]1(O)[C@H]([N+](CC[C@@]112)(C)[O-])C3)CC(=O)[C@@H]1OC1=C2C3=CC=C1OC GXMDLXQESSRSFB-ZWKGOLINSA-N 0.000 description 2
- GVJHHUAWPYXKBD-UHFFFAOYSA-N (±)-α-Tocopherol Chemical compound OC1=C(C)C(C)=C2OC(CCCC(C)CCCC(C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-UHFFFAOYSA-N 0.000 description 2
- KVNYFPKFSJIPBJ-UHFFFAOYSA-N 1,2-diethylbenzene Chemical compound CCC1=CC=CC=C1CC KVNYFPKFSJIPBJ-UHFFFAOYSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 2
- XCYJPXQACVEIOS-UHFFFAOYSA-N 1-isopropyl-3-methylbenzene Chemical compound CC(C)C1=CC=CC(C)=C1 XCYJPXQACVEIOS-UHFFFAOYSA-N 0.000 description 2
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 2
- RYPKRALMXUUNKS-UHFFFAOYSA-N 2-Hexene Natural products CCCC=CC RYPKRALMXUUNKS-UHFFFAOYSA-N 0.000 description 2
- KWSLGOVYXMQPPX-UHFFFAOYSA-N 5-[3-(trifluoromethyl)phenyl]-2h-tetrazole Chemical compound FC(F)(F)C1=CC=CC(C2=NNN=N2)=C1 KWSLGOVYXMQPPX-UHFFFAOYSA-N 0.000 description 2
- 229920002126 Acrylic acid copolymer Polymers 0.000 description 2
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 2
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 2
- 229920001747 Cellulose diacetate Polymers 0.000 description 2
- RGSFGYAAUTVSQA-UHFFFAOYSA-N Cyclopentane Chemical compound C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 2
- YNQLUTRBYVCPMQ-UHFFFAOYSA-N Ethylbenzene Chemical compound CCC1=CC=CC=C1 YNQLUTRBYVCPMQ-UHFFFAOYSA-N 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 229920002774 Maltodextrin Polymers 0.000 description 2
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 2
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 2
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 2
- 239000008896 Opium Substances 0.000 description 2
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 2
- 229920002845 Poly(methacrylic acid) Polymers 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 2
- ATUOYWHBWRKTHZ-UHFFFAOYSA-N Propane Chemical compound CCC ATUOYWHBWRKTHZ-UHFFFAOYSA-N 0.000 description 2
- ZTHYODDOHIVTJV-UHFFFAOYSA-N Propyl gallate Chemical compound CCCOC(=O)C1=CC(O)=C(O)C(O)=C1 ZTHYODDOHIVTJV-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- ZFOZVQLOBQUTQQ-UHFFFAOYSA-N Tributyl citrate Chemical compound CCCCOC(=O)CC(O)(C(=O)OCCCC)CC(=O)OCCCC ZFOZVQLOBQUTQQ-UHFFFAOYSA-N 0.000 description 2
- 229920002494 Zein Polymers 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 239000000908 ammonium hydroxide Substances 0.000 description 2
- 230000036592 analgesia Effects 0.000 description 2
- 239000003963 antioxidant agent Substances 0.000 description 2
- 235000006708 antioxidants Nutrition 0.000 description 2
- 235000013871 bee wax Nutrition 0.000 description 2
- 239000012166 beeswax Substances 0.000 description 2
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 2
- 239000012490 blank solution Substances 0.000 description 2
- IAQRGUVFOMOMEM-UHFFFAOYSA-N but-2-ene Chemical compound CC=CC IAQRGUVFOMOMEM-UHFFFAOYSA-N 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 229920003064 carboxyethyl cellulose Polymers 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229940105329 carboxymethylcellulose Drugs 0.000 description 2
- 239000004203 carnauba wax Substances 0.000 description 2
- 235000013869 carnauba wax Nutrition 0.000 description 2
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 2
- 229940082500 cetostearyl alcohol Drugs 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- RWGFKTVRMDUZSP-UHFFFAOYSA-N cumene Chemical compound CC(C)C1=CC=CC=C1 RWGFKTVRMDUZSP-UHFFFAOYSA-N 0.000 description 2
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- DOIRQSBPFJWKBE-UHFFFAOYSA-N dibutyl phthalate Chemical compound CCCCOC(=O)C1=CC=CC=C1C(=O)OCCCC DOIRQSBPFJWKBE-UHFFFAOYSA-N 0.000 description 2
- 230000001079 digestive effect Effects 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- VDCSGNNYCFPWFK-UHFFFAOYSA-N diphenylsilane Chemical compound C=1C=CC=CC=1[SiH2]C1=CC=CC=C1 VDCSGNNYCFPWFK-UHFFFAOYSA-N 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 229940088679 drug related substance Drugs 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 210000002615 epidermis Anatomy 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000012362 glacial acetic acid Substances 0.000 description 2
- 125000005908 glyceryl ester group Chemical group 0.000 description 2
- 239000001087 glyceryl triacetate Substances 0.000 description 2
- 235000013773 glyceryl triacetate Nutrition 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 239000000852 hydrogen donor Substances 0.000 description 2
- 239000012729 immediate-release (IR) formulation Substances 0.000 description 2
- 239000003701 inert diluent Substances 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- 230000000968 intestinal effect Effects 0.000 description 2
- NNPPMTNAJDCUHE-UHFFFAOYSA-N isobutane Chemical compound CC(C)C NNPPMTNAJDCUHE-UHFFFAOYSA-N 0.000 description 2
- 238000010829 isocratic elution Methods 0.000 description 2
- QWTDNUCVQCZILF-UHFFFAOYSA-N isopentane Chemical compound CCC(C)C QWTDNUCVQCZILF-UHFFFAOYSA-N 0.000 description 2
- 238000012417 linear regression Methods 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 2
- FQXXSQDCDRQNQE-UHFFFAOYSA-N markiertes Thebain Natural products COC1=CC=C2C(N(CC3)C)CC4=CC=C(OC)C5=C4C23C1O5 FQXXSQDCDRQNQE-UHFFFAOYSA-N 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- DCUFMVPCXCSVNP-UHFFFAOYSA-N methacrylic anhydride Chemical compound CC(=C)C(=O)OC(=O)C(C)=C DCUFMVPCXCSVNP-UHFFFAOYSA-N 0.000 description 2
- UAEPNZWRGJTJPN-UHFFFAOYSA-N methylcyclohexane Chemical compound CC1CCCCC1 UAEPNZWRGJTJPN-UHFFFAOYSA-N 0.000 description 2
- GDOPTJXRTPNYNR-UHFFFAOYSA-N methylcyclopentane Chemical compound CC1CCCC1 GDOPTJXRTPNYNR-UHFFFAOYSA-N 0.000 description 2
- 229940016286 microcrystalline cellulose Drugs 0.000 description 2
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 2
- 239000008108 microcrystalline cellulose Substances 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- BKIMMITUMNQMOS-UHFFFAOYSA-N nonane Chemical compound CCCCCCCCC BKIMMITUMNQMOS-UHFFFAOYSA-N 0.000 description 2
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 2
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 2
- 239000003605 opacifier Substances 0.000 description 2
- 229960001027 opium Drugs 0.000 description 2
- 229940105606 oxycontin Drugs 0.000 description 2
- YWAKXRMUMFPDSH-UHFFFAOYSA-N pentene Chemical compound CCCC=C YWAKXRMUMFPDSH-UHFFFAOYSA-N 0.000 description 2
- QMMOXUPEWRXHJS-UHFFFAOYSA-N pentene-2 Natural products CCC=CC QMMOXUPEWRXHJS-UHFFFAOYSA-N 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 229920000191 poly(N-vinyl pyrrolidone) Polymers 0.000 description 2
- 229920001467 poly(styrenesulfonates) Polymers 0.000 description 2
- 229920002401 polyacrylamide Polymers 0.000 description 2
- 229920000193 polymethacrylate Polymers 0.000 description 2
- 229940100467 polyvinyl acetate phthalate Drugs 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- CIBMHJPPKCXONB-UHFFFAOYSA-N propane-2,2-diol Chemical compound CC(C)(O)O CIBMHJPPKCXONB-UHFFFAOYSA-N 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000012898 sample dilution Substances 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 210000000813 small intestine Anatomy 0.000 description 2
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 2
- 235000010234 sodium benzoate Nutrition 0.000 description 2
- 239000004299 sodium benzoate Substances 0.000 description 2
- BBMHARZCALWXSL-UHFFFAOYSA-M sodium dihydrogenphosphate monohydrate Chemical compound O.[Na+].OP(O)([O-])=O BBMHARZCALWXSL-UHFFFAOYSA-M 0.000 description 2
- 229910001379 sodium hypophosphite Inorganic materials 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 239000011550 stock solution Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- HLZKNKRTKFSKGZ-UHFFFAOYSA-N tetradecan-1-ol Chemical compound CCCCCCCCCCCCCCO HLZKNKRTKFSKGZ-UHFFFAOYSA-N 0.000 description 2
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 2
- FQXXSQDCDRQNQE-VMDGZTHMSA-N thebaine Chemical compound C([C@@H](N(CC1)C)C2=CC=C3OC)C4=CC=C(OC)C5=C4[C@@]21[C@H]3O5 FQXXSQDCDRQNQE-VMDGZTHMSA-N 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 229960002622 triacetin Drugs 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 238000005303 weighing Methods 0.000 description 2
- 239000005019 zein Substances 0.000 description 2
- 229940093612 zein Drugs 0.000 description 2
- NOBPDEYWFIONBA-WEZQJLTASA-N (4r,4as,7ar,12br)-4a-hydroxy-9-methoxy-3,7a-dimethyl-1,2,4,5,6,13-hexahydro-4,12-methanobenzofuro[3,2-e]isoquinoline-7-one Chemical compound O=C([C@]1(C)O2)CC[C@@]3(O)[C@H]4CC5=CC=C(OC)C2=C5[C@@]13CCN4C NOBPDEYWFIONBA-WEZQJLTASA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N (R)-alpha-Tocopherol Natural products OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
- ODIGIKRIUKFKHP-UHFFFAOYSA-N (n-propan-2-yloxycarbonylanilino) acetate Chemical compound CC(C)OC(=O)N(OC(C)=O)C1=CC=CC=C1 ODIGIKRIUKFKHP-UHFFFAOYSA-N 0.000 description 1
- OKIRBHVFJGXOIS-UHFFFAOYSA-N 1,2-di(propan-2-yl)benzene Chemical compound CC(C)C1=CC=CC=C1C(C)C OKIRBHVFJGXOIS-UHFFFAOYSA-N 0.000 description 1
- WWRCMNKATXZARA-UHFFFAOYSA-N 1-Isopropyl-2-methylbenzene Chemical compound CC(C)C1=CC=CC=C1C WWRCMNKATXZARA-UHFFFAOYSA-N 0.000 description 1
- HQGPZXPTJWUDQR-UHFFFAOYSA-N 1-ethenyl-5-methylpyrrolidin-2-one Chemical compound CC1CCC(=O)N1C=C HQGPZXPTJWUDQR-UHFFFAOYSA-N 0.000 description 1
- WCOXQTXVACYMLM-UHFFFAOYSA-N 2,3-bis(12-hydroxyoctadecanoyloxy)propyl 12-hydroxyoctadecanoate Chemical compound CCCCCCC(O)CCCCCCCCCCC(=O)OCC(OC(=O)CCCCCCCCCCC(O)CCCCCC)COC(=O)CCCCCCCCCCC(O)CCCCCC WCOXQTXVACYMLM-UHFFFAOYSA-N 0.000 description 1
- CHHHXKFHOYLYRE-UHFFFAOYSA-M 2,4-Hexadienoic acid, potassium salt (1:1), (2E,4E)- Chemical compound [K+].CC=CC=CC([O-])=O CHHHXKFHOYLYRE-UHFFFAOYSA-M 0.000 description 1
- WLQMYDWPKCQDPQ-UHFFFAOYSA-N 2,6-ditert-butyl-4-chlorophenol Chemical compound CC(C)(C)C1=CC(Cl)=CC(C(C)(C)C)=C1O WLQMYDWPKCQDPQ-UHFFFAOYSA-N 0.000 description 1
- SJIXRGNQPBQWMK-UHFFFAOYSA-N 2-(diethylamino)ethyl 2-methylprop-2-enoate Chemical compound CCN(CC)CCOC(=O)C(C)=C SJIXRGNQPBQWMK-UHFFFAOYSA-N 0.000 description 1
- VKNASXZDGZNEDA-UHFFFAOYSA-N 2-cyanoethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCCC#N VKNASXZDGZNEDA-UHFFFAOYSA-N 0.000 description 1
- NEHIITZWJDVKGO-UHFFFAOYSA-N 2-ethoxyethyl 2-methylprop-2-enoate;2-methylprop-2-enoic acid Chemical compound CC(=C)C(O)=O.CCOCCOC(=O)C(C)=C NEHIITZWJDVKGO-UHFFFAOYSA-N 0.000 description 1
- WROUWQQRXUBECT-UHFFFAOYSA-N 2-ethylacrylic acid Chemical compound CCC(=C)C(O)=O WROUWQQRXUBECT-UHFFFAOYSA-N 0.000 description 1
- IMOYOUMVYICGCA-UHFFFAOYSA-N 2-tert-butyl-4-hydroxyanisole Chemical compound COC1=CC=C(O)C=C1C(C)(C)C IMOYOUMVYICGCA-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- ZQDPJFUHLCOCRG-UHFFFAOYSA-N 3-hexene Chemical compound CCC=CCC ZQDPJFUHLCOCRG-UHFFFAOYSA-N 0.000 description 1
- OOWFYDWAMOKVSF-UHFFFAOYSA-N 3-methoxypropanenitrile Chemical compound COCCC#N OOWFYDWAMOKVSF-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Natural products CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 1
- IFTRQJLVEBNKJK-UHFFFAOYSA-N Aethyl-cyclopentan Natural products CCC1CCCC1 IFTRQJLVEBNKJK-UHFFFAOYSA-N 0.000 description 1
- 229920000856 Amylose Polymers 0.000 description 1
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Natural products OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 1
- 239000004255 Butylated hydroxyanisole Substances 0.000 description 1
- 239000005635 Caprylic acid (CAS 124-07-2) Substances 0.000 description 1
- 229920002284 Cellulose triacetate Polymers 0.000 description 1
- VZTBXBNKGIACNJ-PCHTVJFHSA-N Cl.Cl.C1=CC(OC)=C2C=3[C@@]45[C@@H](O2)C(=O)CC[C@@]4(O)[C@@H](CC13)N(C)CC5 Chemical compound Cl.Cl.C1=CC(OC)=C2C=3[C@@]45[C@@H](O2)C(=O)CC[C@@]4(O)[C@@H](CC13)N(C)CC5 VZTBXBNKGIACNJ-PCHTVJFHSA-N 0.000 description 1
- 108020004705 Codon Proteins 0.000 description 1
- 241000967536 Cordiera Species 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- PMPVIKIVABFJJI-UHFFFAOYSA-N Cyclobutane Chemical compound C1CCC1 PMPVIKIVABFJJI-UHFFFAOYSA-N 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- PYGXAGIECVVIOZ-UHFFFAOYSA-N Dibutyl decanedioate Chemical compound CCCCOC(=O)CCCCCCCCC(=O)OCCCC PYGXAGIECVVIOZ-UHFFFAOYSA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- IMROMDMJAWUWLK-UHFFFAOYSA-N Ethenol Chemical compound OC=C IMROMDMJAWUWLK-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 235000000177 Indigofera tinctoria Nutrition 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 239000011786 L-ascorbyl-6-palmitate Substances 0.000 description 1
- QAQJMLQRFWZOBN-LAUBAEHRSA-N L-ascorbyl-6-palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](O)[C@H]1OC(=O)C(O)=C1O QAQJMLQRFWZOBN-LAUBAEHRSA-N 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- CERQOIWHTDAKMF-UHFFFAOYSA-M Methacrylate Chemical compound CC(=C)C([O-])=O CERQOIWHTDAKMF-UHFFFAOYSA-M 0.000 description 1
- SUAKHGWARZSWIH-UHFFFAOYSA-N N,N‐diethylformamide Chemical compound CCN(CC)C=O SUAKHGWARZSWIH-UHFFFAOYSA-N 0.000 description 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 1
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 1
- DXIRRDROGPMTPD-XFKAJCMBSA-N OO[C@@]12C=CC([C@H]3[C@]14C=1C(=C(C=CC1C[C@H]2N(C)CC4)OC)O3)=O Chemical compound OO[C@@]12C=CC([C@H]3[C@]14C=1C(=C(C=CC1C[C@H]2N(C)CC4)OC)O3)=O DXIRRDROGPMTPD-XFKAJCMBSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 102000003840 Opioid Receptors Human genes 0.000 description 1
- 108090000137 Opioid Receptors Proteins 0.000 description 1
- UQCNKQCJZOAFTQ-ISWURRPUSA-N Oxymorphone Chemical compound O([C@H]1C(CC[C@]23O)=O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O UQCNKQCJZOAFTQ-ISWURRPUSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 229920001145 Poly(N-vinylacetamide) Polymers 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 239000004260 Potassium ascorbate Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 239000007868 Raney catalyst Substances 0.000 description 1
- 229910000564 Raney nickel Inorganic materials 0.000 description 1
- 241000220010 Rhode Species 0.000 description 1
- KJTLSVCANCCWHF-UHFFFAOYSA-N Ruthenium Chemical compound [Ru] KJTLSVCANCCWHF-UHFFFAOYSA-N 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- BZHJMEDXRYGGRV-UHFFFAOYSA-N Vinyl chloride Chemical compound ClC=C BZHJMEDXRYGGRV-UHFFFAOYSA-N 0.000 description 1
- 229930003427 Vitamin E Natural products 0.000 description 1
- NNLVGZFZQQXQNW-ADJNRHBOSA-N [(2r,3r,4s,5r,6s)-4,5-diacetyloxy-3-[(2s,3r,4s,5r,6r)-3,4,5-triacetyloxy-6-(acetyloxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6s)-4,5,6-triacetyloxy-2-(acetyloxymethyl)oxan-3-yl]oxyoxan-2-yl]methyl acetate Chemical compound O([C@@H]1O[C@@H]([C@H]([C@H](OC(C)=O)[C@H]1OC(C)=O)O[C@H]1[C@@H]([C@@H](OC(C)=O)[C@H](OC(C)=O)[C@@H](COC(C)=O)O1)OC(C)=O)COC(=O)C)[C@@H]1[C@@H](COC(C)=O)O[C@@H](OC(C)=O)[C@H](OC(C)=O)[C@H]1OC(C)=O NNLVGZFZQQXQNW-ADJNRHBOSA-N 0.000 description 1
- 229940124532 absorption promoter Drugs 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- VEHZBMGMMPZMRJ-UHFFFAOYSA-N acetic acid;2-(diethylamino)acetic acid Chemical compound CC(O)=O.CCN(CC)CC(O)=O VEHZBMGMMPZMRJ-UHFFFAOYSA-N 0.000 description 1
- GJAYYEWRFJQMQK-UHFFFAOYSA-N acetic acid;ethyl carbamate Chemical compound CC(O)=O.CCOC(N)=O GJAYYEWRFJQMQK-UHFFFAOYSA-N 0.000 description 1
- MFOPEVCFSVUADB-UHFFFAOYSA-N acetic acid;methyl carbamate Chemical compound CC(O)=O.COC(N)=O MFOPEVCFSVUADB-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 229920006243 acrylic copolymer Polymers 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 238000005054 agglomeration Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229920003144 amino alkyl methacrylate copolymer Polymers 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O ammonium group Chemical group [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- 150000008064 anhydrides Chemical class 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010385 ascorbyl palmitate Nutrition 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 239000012754 barrier agent Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 150000003938 benzyl alcohols Chemical class 0.000 description 1
- XFOZBWSTIQRFQW-UHFFFAOYSA-M benzyl-dimethyl-prop-2-enylazanium;chloride Chemical compound [Cl-].C=CC[N+](C)(C)CC1=CC=CC=C1 XFOZBWSTIQRFQW-UHFFFAOYSA-M 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 239000003833 bile salt Substances 0.000 description 1
- 239000001045 blue dye Substances 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 239000006189 buccal tablet Substances 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000019282 butylated hydroxyanisole Nutrition 0.000 description 1
- CZBZUDVBLSSABA-UHFFFAOYSA-N butylated hydroxyanisole Chemical compound COC1=CC=C(O)C(C(C)(C)C)=C1.COC1=CC=C(O)C=C1C(C)(C)C CZBZUDVBLSSABA-UHFFFAOYSA-N 0.000 description 1
- 229940043253 butylated hydroxyanisole Drugs 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 238000011088 calibration curve Methods 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 125000005587 carbonate group Chemical group 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 125000004181 carboxyalkyl group Chemical group 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 229920003123 carboxymethyl cellulose sodium Polymers 0.000 description 1
- 229920003090 carboxymethyl hydroxyethyl cellulose Polymers 0.000 description 1
- 229940063834 carboxymethylcellulose sodium Drugs 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 239000013043 chemical agent Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical class OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 1
- 238000000975 co-precipitation Methods 0.000 description 1
- 239000008199 coating composition Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 229920000891 common polymer Polymers 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 239000002826 coolant Substances 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229920006037 cross link polymer Polymers 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 239000003405 delayed action preparation Substances 0.000 description 1
- 229940099371 diacetylated monoglycerides Drugs 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- AFABGHUZZDYHJO-UHFFFAOYSA-N dimethyl butane Natural products CCCC(C)C AFABGHUZZDYHJO-UHFFFAOYSA-N 0.000 description 1
- XNMQEEKYCVKGBD-UHFFFAOYSA-N dimethylacetylene Natural products CC#CC XNMQEEKYCVKGBD-UHFFFAOYSA-N 0.000 description 1
- MSJMDZAOKORVFC-SEPHDYHBSA-L disodium fumarate Chemical compound [Na+].[Na+].[O-]C(=O)\C=C\C([O-])=O MSJMDZAOKORVFC-SEPHDYHBSA-L 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 239000012738 dissolution medium Substances 0.000 description 1
- 238000004821 distillation Methods 0.000 description 1
- LQZZUXJYWNFBMV-UHFFFAOYSA-N dodecan-1-ol Chemical compound CCCCCCCCCCCCO LQZZUXJYWNFBMV-UHFFFAOYSA-N 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- QHZOMAXECYYXGP-UHFFFAOYSA-N ethene;prop-2-enoic acid Chemical compound C=C.OC(=O)C=C QHZOMAXECYYXGP-UHFFFAOYSA-N 0.000 description 1
- MEGHWIAOTJPCHQ-UHFFFAOYSA-N ethenyl butanoate Chemical compound CCCC(=O)OC=C MEGHWIAOTJPCHQ-UHFFFAOYSA-N 0.000 description 1
- GLVVKKSPKXTQRB-UHFFFAOYSA-N ethenyl dodecanoate Chemical compound CCCCCCCCCCCC(=O)OC=C GLVVKKSPKXTQRB-UHFFFAOYSA-N 0.000 description 1
- AFSIMBWBBOJPJG-UHFFFAOYSA-N ethenyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC=C AFSIMBWBBOJPJG-UHFFFAOYSA-N 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 125000005448 ethoxyethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 229920006226 ethylene-acrylic acid Polymers 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000003925 fat Substances 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000007888 film coating Substances 0.000 description 1
- 238000009501 film coating Methods 0.000 description 1
- 239000013020 final formulation Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 235000019634 flavors Nutrition 0.000 description 1
- XUCNUKMRBVNAPB-UHFFFAOYSA-N fluoroethene Chemical compound FC=C XUCNUKMRBVNAPB-UHFFFAOYSA-N 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- WIGCFUFOHFEKBI-UHFFFAOYSA-N gamma-tocopherol Natural products CC(C)CCCC(C)CCCC(C)CCCC1CCC2C(C)C(O)C(C)C(C)C2O1 WIGCFUFOHFEKBI-UHFFFAOYSA-N 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- DMEGYFMYUHOHGS-UHFFFAOYSA-N heptamethylene Natural products C1CCCCCC1 DMEGYFMYUHOHGS-UHFFFAOYSA-N 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 150000004678 hydrides Chemical class 0.000 description 1
- 239000008172 hydrogenated vegetable oil Substances 0.000 description 1
- 230000002706 hydrostatic effect Effects 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 1
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 229940097275 indigo Drugs 0.000 description 1
- COHYTHOBJLSHDF-UHFFFAOYSA-N indigo powder Natural products N1C2=CC=CC=C2C(=O)C1=C1C(=O)C2=CC=CC=C2N1 COHYTHOBJLSHDF-UHFFFAOYSA-N 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 239000001034 iron oxide pigment Substances 0.000 description 1
- LDHBWEYLDHLIBQ-UHFFFAOYSA-M iron(3+);oxygen(2-);hydroxide;hydrate Chemical compound O.[OH-].[O-2].[Fe+3] LDHBWEYLDHLIBQ-UHFFFAOYSA-M 0.000 description 1
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 1
- 239000001282 iso-butane Substances 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- TYQCGQRIZGCHNB-JLAZNSOCSA-N l-ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(O)=C(O)C1=O TYQCGQRIZGCHNB-JLAZNSOCSA-N 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910001386 lithium phosphate Inorganic materials 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000010907 mechanical stirring Methods 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 150000002734 metacrylic acid derivatives Chemical class 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 239000002905 metal composite material Substances 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 1
- AXLHVTKGDPVANO-UHFFFAOYSA-N methyl 2-amino-3-[(2-methylpropan-2-yl)oxycarbonylamino]propanoate Chemical compound COC(=O)C(N)CNC(=O)OC(C)(C)C AXLHVTKGDPVANO-UHFFFAOYSA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- GYNNXHKOJHMOHS-UHFFFAOYSA-N methyl-cycloheptane Natural products CC1CCCCCC1 GYNNXHKOJHMOHS-UHFFFAOYSA-N 0.000 description 1
- 230000011987 methylation Effects 0.000 description 1
- 238000007069 methylation reaction Methods 0.000 description 1
- 239000003094 microcapsule Substances 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- LCEDQNDDFOCWGG-UHFFFAOYSA-N morpholine-4-carbaldehyde Chemical compound O=CN1CCOCC1 LCEDQNDDFOCWGG-UHFFFAOYSA-N 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 229940043348 myristyl alcohol Drugs 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- YNTOKMNHRPSGFU-UHFFFAOYSA-N n-Propyl carbamate Chemical compound CCCOC(N)=O YNTOKMNHRPSGFU-UHFFFAOYSA-N 0.000 description 1
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 1
- 229910052754 neon Inorganic materials 0.000 description 1
- GKAOGPIIYCISHV-UHFFFAOYSA-N neon atom Chemical compound [Ne] GKAOGPIIYCISHV-UHFFFAOYSA-N 0.000 description 1
- 229910000510 noble metal Inorganic materials 0.000 description 1
- QKQQEIVDLRUZRP-UHFFFAOYSA-N northebaine Natural products COC1=CC=C2C(NCC3)CC4=CC=C(OC)C5=C4C23C1O5 QKQQEIVDLRUZRP-UHFFFAOYSA-N 0.000 description 1
- 229960002446 octanoic acid Drugs 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 229960002969 oleic acid Drugs 0.000 description 1
- 239000000014 opioid analgesic Substances 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 239000007935 oral tablet Substances 0.000 description 1
- 229940096978 oral tablet Drugs 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 229960005118 oxymorphone Drugs 0.000 description 1
- 238000010979 pH adjustment Methods 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 230000036407 pain Effects 0.000 description 1
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000000123 paper Substances 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 229940011043 percocet Drugs 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 150000002978 peroxides Chemical class 0.000 description 1
- 229940124531 pharmaceutical excipient Drugs 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 235000011007 phosphoric acid Nutrition 0.000 description 1
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical class OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 238000005498 polishing Methods 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000447 polyanionic polymer Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229920002744 polyvinyl acetate phthalate Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 235000019275 potassium ascorbate Nutrition 0.000 description 1
- 229940017794 potassium ascorbate Drugs 0.000 description 1
- 239000001103 potassium chloride Substances 0.000 description 1
- 235000011164 potassium chloride Nutrition 0.000 description 1
- 229940096992 potassium oleate Drugs 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 235000010241 potassium sorbate Nutrition 0.000 description 1
- 239000004302 potassium sorbate Substances 0.000 description 1
- 229940069338 potassium sorbate Drugs 0.000 description 1
- OTYBMLCTZGSZBG-UHFFFAOYSA-L potassium sulfate Chemical compound [K+].[K+].[O-]S([O-])(=O)=O OTYBMLCTZGSZBG-UHFFFAOYSA-L 0.000 description 1
- 229910052939 potassium sulfate Inorganic materials 0.000 description 1
- 235000011151 potassium sulphates Nutrition 0.000 description 1
- CONVKSGEGAVTMB-RXSVEWSESA-M potassium-L-ascorbate Chemical compound [K+].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] CONVKSGEGAVTMB-RXSVEWSESA-M 0.000 description 1
- MLICVSDCCDDWMD-KVVVOXFISA-M potassium;(z)-octadec-9-enoate Chemical compound [K+].CCCCCCCC\C=C/CCCCCCCC([O-])=O MLICVSDCCDDWMD-KVVVOXFISA-M 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 150000003138 primary alcohols Chemical class 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 239000001294 propane Substances 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 235000010388 propyl gallate Nutrition 0.000 description 1
- 239000000473 propyl gallate Substances 0.000 description 1
- 229940075579 propyl gallate Drugs 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 239000011241 protective layer Substances 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000005086 pumping Methods 0.000 description 1
- 238000000275 quality assurance Methods 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 229940098467 rectal solution Drugs 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 239000001044 red dye Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000003252 repetitive effect Effects 0.000 description 1
- 230000000979 retarding effect Effects 0.000 description 1
- MHOVAHRLVXNVSD-UHFFFAOYSA-N rhodium atom Chemical compound [Rh] MHOVAHRLVXNVSD-UHFFFAOYSA-N 0.000 description 1
- 229940116759 roxicet Drugs 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 235000003441 saturated fatty acids Nutrition 0.000 description 1
- 150000004671 saturated fatty acids Chemical class 0.000 description 1
- 150000003333 secondary alcohols Chemical class 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000007493 shaping process Methods 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- WBHQBSYUUJJSRZ-UHFFFAOYSA-M sodium bisulfate Chemical compound [Na+].OS([O-])(=O)=O WBHQBSYUUJJSRZ-UHFFFAOYSA-M 0.000 description 1
- 229910000342 sodium bisulfate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- CSMWJXBSXGUPGY-UHFFFAOYSA-L sodium dithionate Chemical compound [Na+].[Na+].[O-]S(=O)(=O)S([O-])(=O)=O CSMWJXBSXGUPGY-UHFFFAOYSA-L 0.000 description 1
- 235000010352 sodium erythorbate Nutrition 0.000 description 1
- RYYKJJJTJZKILX-UHFFFAOYSA-M sodium octadecanoate Chemical compound [Na+].CCCCCCCCCCCCCCCCCC([O-])=O RYYKJJJTJZKILX-UHFFFAOYSA-M 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- 235000011008 sodium phosphates Nutrition 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000002195 soluble material Substances 0.000 description 1
- 239000011877 solvent mixture Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 238000005563 spheronization Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 125000004079 stearyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 239000012489 system suitability test solution Substances 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- DOMXUEMWDBAQBQ-WEVVVXLNSA-N terbinafine Chemical compound C1=CC=C2C(CN(C\C=C\C#CC(C)(C)C)C)=CC=CC2=C1 DOMXUEMWDBAQBQ-WEVVVXLNSA-N 0.000 description 1
- 229960002722 terbinafine Drugs 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 229930003945 thebaine Natural products 0.000 description 1
- 239000012815 thermoplastic material Substances 0.000 description 1
- 229920001187 thermosetting polymer Polymers 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- AOBORMOPSGHCAX-DGHZZKTQSA-N tocofersolan Chemical compound OCCOC(=O)CCC(=O)OC1=C(C)C(C)=C2O[C@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C AOBORMOPSGHCAX-DGHZZKTQSA-N 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 238000009901 transfer hydrogenation reaction Methods 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- TWQULNDIKKJZPH-UHFFFAOYSA-K trilithium;phosphate Chemical compound [Li+].[Li+].[Li+].[O-]P([O-])([O-])=O TWQULNDIKKJZPH-UHFFFAOYSA-K 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 238000000825 ultraviolet detection Methods 0.000 description 1
- 238000003828 vacuum filtration Methods 0.000 description 1
- 239000012178 vegetable wax Substances 0.000 description 1
- 229940046009 vitamin E Drugs 0.000 description 1
- 235000019165 vitamin E Nutrition 0.000 description 1
- 239000011709 vitamin E Substances 0.000 description 1
- 230000004584 weight gain Effects 0.000 description 1
- 235000019786 weight gain Nutrition 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 239000002759 woven fabric Substances 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D489/00—Heterocyclic compounds containing 4aH-8, 9 c- Iminoethano-phenanthro [4, 5-b, c, d] furan ring systems, e.g. derivatives of [4, 5-epoxy]-morphinan of the formula:
- C07D489/06—Heterocyclic compounds containing 4aH-8, 9 c- Iminoethano-phenanthro [4, 5-b, c, d] furan ring systems, e.g. derivatives of [4, 5-epoxy]-morphinan of the formula: with a hetero atom directly attached in position 14
- C07D489/08—Oxygen atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/485—Morphinan derivatives, e.g. morphine, codeine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2095—Tabletting processes; Dosage units made by direct compression of powders or specially processed granules, by eliminating solvents, by melt-extrusion, by injection molding, by 3D printing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4833—Encapsulating processes; Filling of capsules
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D489/00—Heterocyclic compounds containing 4aH-8, 9 c- Iminoethano-phenanthro [4, 5-b, c, d] furan ring systems, e.g. derivatives of [4, 5-epoxy]-morphinan of the formula:
- C07D489/02—Heterocyclic compounds containing 4aH-8, 9 c- Iminoethano-phenanthro [4, 5-b, c, d] furan ring systems, e.g. derivatives of [4, 5-epoxy]-morphinan of the formula: with oxygen atoms attached in positions 3 and 6, e.g. morphine, morphinone
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D489/00—Heterocyclic compounds containing 4aH-8, 9 c- Iminoethano-phenanthro [4, 5-b, c, d] furan ring systems, e.g. derivatives of [4, 5-epoxy]-morphinan of the formula:
- C07D489/02—Heterocyclic compounds containing 4aH-8, 9 c- Iminoethano-phenanthro [4, 5-b, c, d] furan ring systems, e.g. derivatives of [4, 5-epoxy]-morphinan of the formula: with oxygen atoms attached in positions 3 and 6, e.g. morphine, morphinone
- C07D489/04—Salts; Organic complexes
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Pain & Pain Management (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Emergency Medicine (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Biomedical Technology (AREA)
- Rheumatology (AREA)
- Inorganic Chemistry (AREA)
- Nutrition Science (AREA)
- Physiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Medicinal Preparation (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Saccharide Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Applications Claiming Priority (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US55749204P | 2004-03-30 | 2004-03-30 | |
| US60/557,492 | 2004-03-30 | ||
| US60153404P | 2004-08-13 | 2004-08-13 | |
| US60/601,534 | 2004-08-13 | ||
| US62007204P | 2004-10-18 | 2004-10-18 | |
| US60/620,072 | 2004-10-18 | ||
| US64862505P | 2005-01-31 | 2005-01-31 | |
| US60/648,625 | 2005-01-31 | ||
| US65177805P | 2005-02-10 | 2005-02-10 | |
| US60/651,778 | 2005-02-10 | ||
| PCT/US2005/010666 WO2005097801A1 (en) | 2004-03-30 | 2005-03-30 | Process for preparing oxycodone hydrochloride having less than 25 ppm 14-hydroxycodeinone |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020147030767A Division KR20140133615A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187021551A Division KR20180088505A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20150046393A true KR20150046393A (ko) | 2015-04-29 |
Family
ID=35124996
Family Applications (12)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187021551A Ceased KR20180088505A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020147030767A Ceased KR20140133615A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020147001423A Withdrawn KR20140021716A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020117022725A Expired - Lifetime KR101200700B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020157009780A Ceased KR20150046393A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020097019843A Expired - Lifetime KR100993911B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020067022569A Expired - Lifetime KR100930730B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산옥시코돈의 제조방법 |
| KR1020197038467A Ceased KR20200000499A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020127017379A Ceased KR20120106784A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020127006697A Ceased KR20120046309A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020137000989A Expired - Lifetime KR101410141B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020107013435A Ceased KR20100069719A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
Family Applications Before (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187021551A Ceased KR20180088505A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020147030767A Ceased KR20140133615A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020147001423A Withdrawn KR20140021716A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020117022725A Expired - Lifetime KR101200700B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
Family Applications After (7)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020097019843A Expired - Lifetime KR100993911B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020067022569A Expired - Lifetime KR100930730B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산옥시코돈의 제조방법 |
| KR1020197038467A Ceased KR20200000499A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020127017379A Ceased KR20120106784A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020127006697A Ceased KR20120046309A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020137000989A Expired - Lifetime KR101410141B1 (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
| KR1020107013435A Ceased KR20100069719A (ko) | 2004-03-30 | 2005-03-30 | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 |
Country Status (38)
Families Citing this family (110)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7776314B2 (en) | 2002-06-17 | 2010-08-17 | Grunenthal Gmbh | Abuse-proofed dosage system |
| US8075872B2 (en) | 2003-08-06 | 2011-12-13 | Gruenenthal Gmbh | Abuse-proofed dosage form |
| DE10361596A1 (de) * | 2003-12-24 | 2005-09-29 | Grünenthal GmbH | Verfahren zur Herstellung einer gegen Missbrauch gesicherten Darreichungsform |
| US20070048228A1 (en) * | 2003-08-06 | 2007-03-01 | Elisabeth Arkenau-Maric | Abuse-proofed dosage form |
| DE10336400A1 (de) | 2003-08-06 | 2005-03-24 | Grünenthal GmbH | Gegen Missbrauch gesicherte Darreichungsform |
| DE102005005446A1 (de) | 2005-02-04 | 2006-08-10 | Grünenthal GmbH | Bruchfeste Darreichungsformen mit retardierter Freisetzung |
| DE102004020220A1 (de) * | 2004-04-22 | 2005-11-10 | Grünenthal GmbH | Verfahren zur Herstellung einer gegen Missbrauch gesicherten, festen Darreichungsform |
| PT1842533E (pt) * | 2003-08-06 | 2013-05-17 | Gruenenthal Gmbh | Forma de dosagem protegida contra abuso |
| US7201920B2 (en) | 2003-11-26 | 2007-04-10 | Acura Pharmaceuticals, Inc. | Methods and compositions for deterring abuse of opioid containing dosage forms |
| TW201509943A (zh) * | 2004-03-30 | 2015-03-16 | Euro Celtique Sa | 含有小於25ppm14-羥可待因酮之羥可酮鹽酸鹽之組成物、醫藥劑型、延遲釋出口服劑型及醫藥上可以接受的包裝 |
| DE102004032049A1 (de) * | 2004-07-01 | 2006-01-19 | Grünenthal GmbH | Gegen Missbrauch gesicherte, orale Darreichungsform |
| DE102004032103A1 (de) * | 2004-07-01 | 2006-01-19 | Grünenthal GmbH | Gegen Missbrauch gesicherte, orale Darreichungsform |
| GB0421149D0 (en) * | 2004-09-23 | 2004-10-27 | Johnson Matthey Plc | Preparation of oxycodone |
| US20080152595A1 (en) * | 2004-11-24 | 2008-06-26 | Acura Pharmaceuticals, Inc. | Methods and compositions for deterring abuse of orally administered pharmaceutical products |
| DE102005005449A1 (de) * | 2005-02-04 | 2006-08-10 | Grünenthal GmbH | Verfahren zur Herstellung einer gegen Missbrauch gesicherten Darreichungsform |
| PT2112153E (pt) * | 2005-03-04 | 2010-12-17 | Euro Celtique Sa | Método de diminuír a quantidade de cetonas insaturadas em alfa-beta em composições de opiáceos |
| US20070082845A1 (en) * | 2005-07-15 | 2007-04-12 | The Penn State Research Foundation | Ferritin as a therapeutic target in abnormal cells |
| EP1971607A2 (en) * | 2005-11-22 | 2008-09-24 | Controlled Chemicals, Inc. | Processes for reducing contaminating michael acceptor levels in oxycodone and other compositions |
| US20070117826A1 (en) * | 2005-11-23 | 2007-05-24 | Forest Laboratories, Inc. | Pharmaceutical formulations comprising ibuprofen, oxycodone, and 14-hydroxycodeinone |
| JP5695296B2 (ja) * | 2006-03-02 | 2015-04-01 | マリンクロッド エルエルシー | 低レベルのアルファ,ベータ−不飽和ケトン化合物を伴うモルフィナン−6−オン生成物の製造方法 |
| SA07280459B1 (ar) | 2006-08-25 | 2011-07-20 | بيورديو فارما إل. بي. | أشكال جرعة صيدلانية للتناول عن طريق الفم مقاومة للعبث تشتمل على مسكن شبه أفيوني |
| WO2008048711A1 (en) * | 2006-10-17 | 2008-04-24 | Penick Corporation | Process for preparing oxymorphone |
| US20080125592A1 (en) * | 2006-10-17 | 2008-05-29 | Penick Corporation | Process for preparing oxymorphone, naltrexone, and buprenorphine |
| CA2674915C (en) | 2006-10-17 | 2015-06-30 | Penick Corporation | Process for preparing oxymorphone |
| CA2671518C (en) * | 2006-12-04 | 2016-06-07 | Noramco, Inc. | Process for preparing oxycodone having reduced levels of 14-hydroxycodeinone |
| AU2007329451B2 (en) * | 2006-12-04 | 2012-08-30 | Noramco, Llc | Process for reducing impurities in oxycodone base |
| GB0624880D0 (en) * | 2006-12-14 | 2007-01-24 | Johnson Matthey Plc | Improved method for making analgesics |
| DE102007011485A1 (de) | 2007-03-07 | 2008-09-11 | Grünenthal GmbH | Darreichungsform mit erschwertem Missbrauch |
| CA2681740A1 (en) * | 2007-03-23 | 2008-10-02 | Mallinckrodt Inc. | Improved preparation of oxymorphone from oripavine |
| CA2684458A1 (en) | 2007-04-16 | 2008-10-30 | Mallinckrodt Inc. | Novel opiate reduction utilizing catalytic hydrogen transfer reaction |
| AU2009207796B2 (en) | 2008-01-25 | 2014-03-27 | Grunenthal Gmbh | Pharmaceutical dosage form |
| CA2720108C (en) * | 2008-03-11 | 2016-06-07 | Depomed, Inc. | Gastric retentive extended-release dosage forms comprising combinations of a non-opioid analgesic and an opioid analgesic |
| US8372432B2 (en) * | 2008-03-11 | 2013-02-12 | Depomed, Inc. | Gastric retentive extended-release dosage forms comprising combinations of a non-opioid analgesic and an opioid analgesic |
| KR101690094B1 (ko) | 2008-05-09 | 2016-12-27 | 그뤼넨탈 게엠베하 | 분무 응결 단계의 사용하에 중간 분말 제형 및 최종 고체 제형을 제조하는 방법 |
| MX342643B (es) * | 2008-09-18 | 2016-10-07 | Purdue Pharma Lp | Formas de dosificacion farmaceutica que comprenden poli (e-caprolactona). |
| CA2765971C (en) * | 2009-07-22 | 2017-08-22 | Gruenenthal Gmbh | Hot-melt extruded controlled release dosage form |
| ES2428938T3 (es) * | 2009-07-22 | 2013-11-12 | Grünenthal GmbH | Forma de dosificación resistente a la manipulación y estabilizada frente a la oxidación |
| EP2473195A4 (en) * | 2009-08-31 | 2013-01-16 | Depomed Inc | PHARMACEUTICAL MAGNETIC RELEASE COMPOSITIONS FOR IMMEDIATE AND EXTENDED RELEASE OF ACETAMINOPHES |
| WO2011041414A1 (en) | 2009-09-30 | 2011-04-07 | Acura Pharmaceuticals, Inc. | Methods and compositions for deterring abuse |
| US8597681B2 (en) | 2009-12-22 | 2013-12-03 | Mallinckrodt Llc | Methods of producing stabilized solid dosage pharmaceutical compositions containing morphinans |
| US9198861B2 (en) | 2009-12-22 | 2015-12-01 | Mallinckrodt Llc | Methods of producing stabilized solid dosage pharmaceutical compositions containing morphinans |
| WO2011095314A2 (en) * | 2010-02-03 | 2011-08-11 | Grünenthal GmbH | Preparation of a powdery pharmaceutical composition by means of an extruder |
| EP2377866B1 (en) | 2010-03-23 | 2014-02-26 | Siegfried AG | Preparation of low impurity opiates in a continuous flow reactor |
| EP2568968B1 (en) | 2010-05-10 | 2017-07-12 | Euro-Celtique S.A. | Manufacturing of active-free granules and tablets comprising the same |
| US9901540B2 (en) | 2010-05-10 | 2018-02-27 | Euro-Celtique S.A. | Combination of active loaded granules with additional actives |
| SE1251371A1 (sv) | 2010-05-10 | 2012-12-27 | Euro Celtique Sa | Farmaceutiska kompositioner innefattande hydromorfon och naloxon |
| WO2011154826A1 (en) | 2010-06-11 | 2011-12-15 | Rhodes Technologies | Process for n-dealkylation of tertiary amines |
| WO2011154827A2 (en) | 2010-06-11 | 2011-12-15 | Rhodes Technologies | Transition metal-catalyzed processes for the preparation of n-allyl compounds and use thereof |
| ES2659168T3 (es) | 2010-07-02 | 2018-03-14 | Johnson Matthey Public Limited Company | Proceso para la síntesis y purificación de oxicodona |
| US8912781B2 (en) * | 2010-07-30 | 2014-12-16 | Cirrus Logic, Inc. | Integrated circuit switching power supply controller with selectable buck mode operation |
| RU2604676C2 (ru) | 2010-09-02 | 2016-12-10 | Грюненталь Гмбх | Устойчивая к разрушению лекарственная форма, содержащая неорганическую соль |
| PL2611425T3 (pl) | 2010-09-02 | 2014-09-30 | Gruenenthal Gmbh | Odporna na ingerencję postać dawki zawierająca polimer anionowy |
| US8741885B1 (en) | 2011-05-17 | 2014-06-03 | Mallinckrodt Llc | Gastric retentive extended release pharmaceutical compositions |
| US8858963B1 (en) | 2011-05-17 | 2014-10-14 | Mallinckrodt Llc | Tamper resistant composition comprising hydrocodone and acetaminophen for rapid onset and extended duration of analgesia |
| US8658631B1 (en) | 2011-05-17 | 2014-02-25 | Mallinckrodt Llc | Combination composition comprising oxycodone and acetaminophen for rapid onset and extended duration of analgesia |
| HRP20171458T1 (hr) | 2011-07-29 | 2017-11-17 | Grünenthal GmbH | Tableta otporna na mijenjanje koja pruža neposredno oslobađanje lijeka |
| PL2736495T3 (pl) | 2011-07-29 | 2018-01-31 | Gruenenthal Gmbh | Tabletka odporna na ingerencję, zapewniająca natychmiastowe uwalnianie leku |
| ES2842453T3 (es) | 2011-09-05 | 2021-07-14 | Siegfried Ltd | Kit que comprende un material de envasado y un producto farmacéutico o nutracéutico sólido contenido en el material de envasado |
| WO2013038268A1 (en) | 2011-09-16 | 2013-03-21 | Purdue Pharma L.P. | Tamper resistant immediate release formulations |
| US20140341984A1 (en) | 2011-09-16 | 2014-11-20 | Purdue Pharma L.P. | Tamper resistant pharmaceutical formulations |
| US20150265536A1 (en) | 2011-12-09 | 2015-09-24 | Purdue Pharma L.P. | Pharmaceutical dosage forms comprising poly(epsilon-caprolactone) and polyethylene oxide |
| WO2013127831A1 (en) | 2012-02-28 | 2013-09-06 | Grünenthal GmbH | Tamper-resistant dosage form comprising pharmacologically active compound and anionic polymer |
| CN109820830A (zh) | 2012-03-02 | 2019-05-31 | 罗德兹制药股份有限公司 | 抗破坏性立即释放型制剂类 |
| DE112013002074T5 (de) | 2012-04-17 | 2015-01-29 | Purdue Pharma L.P. | Systeme und Methoden zur Behandlung einer Opioid-induzierten unerwünschten pharmakodynamischen Reaktion |
| LT2838512T (lt) | 2012-04-18 | 2018-11-12 | GrĆ¼nenthal GmbH | Pažeidimui atspari ir dozės atpalaidavimo nuokrypiui atspari farmacinė vaisto forma |
| US10064945B2 (en) | 2012-05-11 | 2018-09-04 | Gruenenthal Gmbh | Thermoformed, tamper-resistant pharmaceutical dosage form containing zinc |
| WO2014011830A1 (en) | 2012-07-12 | 2014-01-16 | Mallinckrodt Llc | Extended release, abuse deterrent pharmaceutical compositions |
| ES2638178T3 (es) | 2012-07-16 | 2017-10-19 | Rhodes Technologies | Proceso para la síntesis mejorada de opioides |
| KR101946103B1 (ko) | 2012-07-16 | 2019-02-08 | 로드스 테크놀로지즈 | 개선된 오피오이드 합성을 위한 방법 |
| RS58427B1 (sr) | 2012-08-03 | 2019-04-30 | Johnson Matthey Plc | Postupak za pripremu oksikodona |
| RU2018141241A (ru) | 2012-11-30 | 2019-01-24 | Экьюра Фармасьютикалз, Инк. | Саморегулируемое высвобождение фармацевтического ингредиента |
| MX2015010041A (es) | 2013-02-05 | 2015-10-30 | Purdue Pharma Lp | Formulacion farmaceuticas resistentes a la alteracion. |
| US9770710B2 (en) | 2013-02-20 | 2017-09-26 | University Of Washington Through Its Center For Commercialization | Hydrogenation and disproportionation catalysis |
| US10751287B2 (en) | 2013-03-15 | 2020-08-25 | Purdue Pharma L.P. | Tamper resistant pharmaceutical formulations |
| JP6445537B2 (ja) | 2013-05-29 | 2018-12-26 | グリュネンタール・ゲゼルシャフト・ミト・ベシュレンクテル・ハフツング | 1個または複数の粒子を含有する改変防止(tamper−resistant)剤形 |
| AR096438A1 (es) | 2013-05-29 | 2015-12-30 | Gruenenthal Gmbh | Forma de dosificación resistente al uso indebido con perfil de liberación bimodal, proceso |
| US10624862B2 (en) | 2013-07-12 | 2020-04-21 | Grünenthal GmbH | Tamper-resistant dosage form containing ethylene-vinyl acetate polymer |
| GB201313211D0 (en) | 2013-07-24 | 2013-09-04 | Cambrex Karlskoga Ab | New process |
| GB201313915D0 (en) * | 2013-08-02 | 2013-09-18 | Johnson Matthey Plc | Process |
| US9120800B2 (en) | 2013-08-02 | 2015-09-01 | Johnson Matthey Public Limited Company | Process for the preparation of oxymorphone alkaloid and oxymorphone salts |
| WO2015069717A1 (en) | 2013-11-07 | 2015-05-14 | Mallinckrodt Llc | Production of 6-hydroxy morphinans without the isolation of intermediates |
| US9814710B2 (en) | 2013-11-13 | 2017-11-14 | Euro-Celtique S.A. | Hydromorphone and naloxone for treatment of pain and opioid bowel dysfunction syndrome |
| JP6480936B2 (ja) | 2013-11-26 | 2019-03-13 | グリュネンタール・ゲゼルシャフト・ミト・ベシュレンクテル・ハフツング | クライオミリングによる粉末状医薬組成物の調製 |
| US8846923B1 (en) | 2013-12-18 | 2014-09-30 | Cody Laboratories, Inc. | Preparation of 14-hydroxycodeinone sulfate |
| US9062062B1 (en) | 2013-12-18 | 2015-06-23 | Cody Laboratories, Inc. | Synthesis of oxycodone hydrochloride |
| US10227354B2 (en) | 2013-12-18 | 2019-03-12 | Cody Laboratories, Inc. | Conversion of oxycodone base to oxycodone hydrochloride |
| CA2937007C (en) | 2014-01-15 | 2018-11-06 | Rhodes Technologies | Process for improved oxymorphone synthesis |
| US9932348B2 (en) | 2014-01-15 | 2018-04-03 | Rhodes Technologies | Process for improved oxycodone synthesis |
| WO2015120201A1 (en) | 2014-02-05 | 2015-08-13 | Kashiv Pharma, Llc | Abuse-resistant drug formulations with built-in overdose protection |
| WO2015145459A1 (en) | 2014-03-26 | 2015-10-01 | Sun Pharma Advanced Research Company Ltd. | Abuse deterrent immediate release coated reservoir solid dosage form |
| JP2017518980A (ja) | 2014-05-12 | 2017-07-13 | グリュネンタール・ゲゼルシャフト・ミト・ベシュレンクテル・ハフツング | タペンタドールを含む、改変防止即時放出カプセル製剤 |
| CN106456550A (zh) | 2014-05-26 | 2017-02-22 | 格吕伦塔尔有限公司 | 避免乙醇剂量倾泻的多颗粒 |
| WO2016005923A1 (en) | 2014-07-09 | 2016-01-14 | Rhodes Technologies | Reduction of alpha, beta-unsaturated ketone levels in morphinan derivative compositions |
| CA2910865C (en) | 2014-07-15 | 2016-11-29 | Isa Odidi | Compositions and methods for reducing overdose |
| US20160052932A1 (en) | 2014-08-25 | 2016-02-25 | Johnson Matthey Public Limited Company | Processes for Making Opioids Including 14-Hydroxycodeinone and 14-hydroxymorphinone |
| US9849124B2 (en) | 2014-10-17 | 2017-12-26 | Purdue Pharma L.P. | Systems and methods for treating an opioid-induced adverse pharmacodynamic response |
| CN105777766B (zh) * | 2014-12-15 | 2017-11-24 | 北大方正集团有限公司 | Delta‑7溴甲纳曲酮的制备方法 |
| US9918979B2 (en) * | 2015-01-29 | 2018-03-20 | Johnson Matthey Public Limited Company | Process of preparing low ABUK oxymorphone hydrochloride |
| KR20170139158A (ko) | 2015-04-24 | 2017-12-18 | 그뤼넨탈 게엠베하 | 즉시 방출되고 용매 추출 방지된 변조 방지된 투여 형태 |
| GB201513203D0 (en) | 2015-07-27 | 2015-09-09 | Cambrex Charles City Inc | New process |
| US11103581B2 (en) | 2015-08-31 | 2021-08-31 | Acura Pharmaceuticals, Inc. | Methods and compositions for self-regulated release of active pharmaceutical ingredient |
| WO2017042325A1 (en) | 2015-09-10 | 2017-03-16 | Grünenthal GmbH | Protecting oral overdose with abuse deterrent immediate release formulations |
| US9861629B1 (en) | 2015-10-07 | 2018-01-09 | Banner Life Sciences Llc | Opioid abuse deterrent dosage forms |
| US10335405B1 (en) | 2016-05-04 | 2019-07-02 | Patheon Softgels, Inc. | Non-burst releasing pharmaceutical composition |
| US20180104236A1 (en) | 2016-09-26 | 2018-04-19 | Euro-Celtique S. A. | Methods of treatment comprising administering a high daily dose of oxycodone and naloxone in a 2:1 weight ratio |
| US10335375B2 (en) | 2017-05-30 | 2019-07-02 | Patheon Softgels, Inc. | Anti-overingestion abuse deterrent compositions |
| CN110770238B (zh) * | 2017-06-20 | 2022-10-04 | 庄信万丰股份有限公司 | 用于从14-羟可待因酮制备盐酸氧可酮的氢化方法 |
| JP2020526500A (ja) | 2017-06-30 | 2020-08-31 | パーデュー、ファーマ、リミテッド、パートナーシップ | 治療方法及びその剤形 |
| CN110330500B (zh) * | 2019-07-12 | 2021-11-23 | 宜昌人福药业有限责任公司 | 一种6β-羟基-7,8-二氢-吗啡衍生物的立体选择性合成方法 |
| CN113009060B (zh) * | 2021-02-24 | 2022-10-21 | 西南药业股份有限公司 | 高效液相色谱法测定盐酸羟考酮含量的方法 |
Family Cites Families (97)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE286431C (enExample) | ||||
| DE296916C (enExample) | ||||
| US100000A (en) * | 1870-02-22 | Improved sun-bonnet for horses | ||
| US420098A (en) * | 1890-01-28 | Mowing-machine | ||
| US1468805A (en) | 1923-09-25 | Htabtin ereund | ||
| US1485673A (en) * | 1924-03-04 | Ministrator | ||
| US105553A (en) | 1870-07-19 | Improvement in kettle for melting, mixing, and casting metals | ||
| US710223A (en) * | 1901-06-10 | 1902-09-30 | Thew Automatic Shovel Company | Power-shovel. |
| SU64699A1 (ru) | 1944-01-26 | 1944-11-30 | А.И. Лютенберг | Способ получени сол нокислого дигидрооксикодеинона |
| ES212554A1 (es) | 1953-12-01 | 1954-08-16 | Casanovas Albajes Cesar | Un procedimiento para la obtención de un vidrio orgánico |
| US2772270A (en) | 1954-10-21 | 1956-11-27 | M J Lewenstein | 14-hydroxymorphinone and 8, 14-dihydroxydihydromorphinone |
| US2806033A (en) | 1955-08-03 | 1957-09-10 | Lewenstein | Morphine derivative |
| US3173878A (en) * | 1960-02-26 | 1965-03-16 | Ibm | Process of making microcapsules |
| US3173876A (en) | 1960-05-27 | 1965-03-16 | John C Zobrist | Cleaning methods and compositions |
| NL271831A (enExample) | 1960-11-29 | |||
| US3276586A (en) | 1963-08-30 | 1966-10-04 | Rosaen Filter Co | Indicating means for fluid filters |
| US3916898A (en) | 1964-05-20 | 1975-11-04 | Searle & Co | Administration of medicaments and the like |
| NL6714885A (enExample) | 1967-11-02 | 1969-05-06 | ||
| US3541006A (en) | 1968-07-03 | 1970-11-17 | Amicon Corp | Ultrafiltration process |
| US3541005A (en) | 1969-02-05 | 1970-11-17 | Amicon Corp | Continuous ultrafiltration of macromolecular solutions |
| US3845770A (en) | 1972-06-05 | 1974-11-05 | Alza Corp | Osmatic dispensing device for releasing beneficial agent |
| US3916899A (en) | 1973-04-25 | 1975-11-04 | Alza Corp | Osmotic dispensing device with maximum and minimum sizes for the passageway |
| US3905981A (en) | 1973-10-12 | 1975-09-16 | Research Corp | N-dealkylation of tertiary amines |
| US3894026A (en) | 1973-10-16 | 1975-07-08 | Merck & Co Inc | Production of thebaine |
| GB1478759A (en) | 1974-11-18 | 1977-07-06 | Alza Corp | Process for forming outlet passageways in pills using a laser |
| US4077407A (en) | 1975-11-24 | 1978-03-07 | Alza Corporation | Osmotic devices having composite walls |
| US4063064A (en) | 1976-02-23 | 1977-12-13 | Coherent Radiation | Apparatus for tracking moving workpiece by a laser beam |
| US3984026A (en) * | 1976-03-26 | 1976-10-05 | Shoup Russel W | Combined can opening and sealing device |
| US4045440A (en) | 1976-09-16 | 1977-08-30 | The United States Of America As Represented By The Department Of Health, Education And Welfare | Method of producing thebaine from codeine and oripavine from morphine |
| US4200098A (en) | 1978-10-23 | 1980-04-29 | Alza Corporation | Osmotic system with distribution zone for dispensing beneficial agent |
| US4285987A (en) | 1978-10-23 | 1981-08-25 | Alza Corporation | Process for manufacturing device with dispersion zone |
| US4272540A (en) | 1979-04-24 | 1981-06-09 | Sisa, Incorporated | 14-Methoxymorphinan-6-one compounds and therapeutic methods of treating pain and drug dependence with them |
| US4370333A (en) | 1981-06-29 | 1983-01-25 | Sisa, Incorporated | 17-Cyclopropylmethyl-3-hydroxy-14-methoxy 7α-methyl-morphinan-6-one and therapeutic method of treating pain with it |
| US4795813A (en) | 1981-08-17 | 1989-01-03 | The Florida Board Of Regents On Behalf Of The Florida State University | Synthesis of derivatives of codeine and other 3-O-alkylmorphines |
| US4639520A (en) | 1984-03-27 | 1987-01-27 | Mallinckrodt, Inc. | Preparation of 14-hydroxy-N-ethoxy-carbonyl-norcodeinone |
| US5112975A (en) | 1984-03-27 | 1992-05-12 | Mallinckrodt Specialty Chemicals Company | Preparation of noroxymorphone from morphine |
| US4861598A (en) | 1986-07-18 | 1989-08-29 | Euroceltique, S.A. | Controlled release bases for pharmaceuticals |
| US4810699A (en) | 1987-02-20 | 1989-03-07 | American Home Products Corporation | Substituted 1,3,4,9-tetrahydropyrano[3,4,-b]indole-1-acetic acids, pharmaceutical compositions containing them, and methods for treating inflammatory conditions and for analgesic purposes using them |
| KR930001499B1 (ko) * | 1987-07-07 | 1993-03-02 | 오끼뎅끼 고오교오 가부시끼가이샤 | 반도체 장치의 제조방법 |
| DE3812567A1 (de) | 1988-04-15 | 1989-10-26 | Basf Ag | Verfahren zur herstellung pharmazeutischer mischungen |
| HU208633B (en) | 1991-02-04 | 1993-12-28 | Alkaloida Vegyeszeti Gyar | Process for production of analgetic compositions as applicable for blocking of opioid-binding spaces /2-receptors/ causing respiration depression |
| US5215758A (en) | 1991-09-11 | 1993-06-01 | Euroceltique, S.A. | Controlled release matrix suppository for pharmaceuticals |
| US5656295A (en) | 1991-11-27 | 1997-08-12 | Euro-Celtique, S.A. | Controlled release oxycodone compositions |
| US5266331A (en) | 1991-11-27 | 1993-11-30 | Euroceltique, S.A. | Controlled release oxycodone compositions |
| US5286493A (en) | 1992-01-27 | 1994-02-15 | Euroceltique, S.A. | Stabilized controlled release formulations having acrylic polymer coating |
| US5273760A (en) | 1991-12-24 | 1993-12-28 | Euroceltigue, S.A. | Stabilized controlled release substrate having a coating derived from an aqueous dispersion of hydrophobic polymer |
| US5472712A (en) | 1991-12-24 | 1995-12-05 | Euroceltique, S.A. | Controlled-release formulations coated with aqueous dispersions of ethylcellulose |
| US5324351A (en) | 1992-08-13 | 1994-06-28 | Euroceltique | Aqueous dispersions of zein and preparation thereof |
| JP3433871B2 (ja) * | 1996-01-26 | 2003-08-04 | 株式会社デンソー | 集積化半導体歪みセンサ及びその製造方法 |
| AU3508097A (en) | 1996-06-26 | 1998-01-14 | Board Of Regents, The University Of Texas System | Hot-melt extrudable pharmaceutical formulation |
| US5869669A (en) | 1996-07-26 | 1999-02-09 | Penick Corporation | Preparation of 14-hydroxynormorphinones from normorphinone dienol acylates |
| ES2121554B1 (es) * | 1996-12-23 | 1999-06-16 | Univ Santiago Compostela | Procedimiento de obtencion de 14-hidroximofinonas mediante fotooxidac ion de alcaloides morfinicos con sistema alcoxidienico en el anillo c. |
| GB9713703D0 (en) | 1997-06-30 | 1997-09-03 | Johnson Matthey Plc | Preparation of opiates |
| GB9717629D0 (en) | 1997-08-21 | 1997-10-22 | Johnson Matthey Plc | Removal of residual organic solvents |
| US6710223B1 (en) | 1997-09-10 | 2004-03-23 | The Procter & Gamble Company | Method for improving skin condition |
| GB9805516D0 (en) | 1998-03-17 | 1998-05-13 | Johnson Matthey Plc | Preparation of opiates |
| US6454970B1 (en) * | 1998-10-14 | 2002-09-24 | Amic Ab And Gyros Ab | Matrix, method of producing and using the matrix and machine including the matrix |
| US6177567B1 (en) * | 1999-10-15 | 2001-01-23 | Boehringer Ingelheim Chemicals, Inc. | Method for preparing oxycodone |
| US6284769B1 (en) * | 1999-12-03 | 2001-09-04 | The Board Of Trustees Of The University Of Illinois | Nonpeptide kappa opioid receptor antagonists |
| US6133132A (en) * | 2000-01-20 | 2000-10-17 | Advanced Micro Devices, Inc. | Method for controlling transistor spacer width |
| WO2001058447A1 (en) * | 2000-02-08 | 2001-08-16 | Euro-Celtique, S.A. | Controlled-release compositions containing opioid agonist and antagonist |
| AU2001258825A1 (en) | 2000-05-29 | 2001-12-11 | Shionogi And Co., Ltd. | Method for labeling with tritium |
| US7200357B2 (en) * | 2000-10-20 | 2007-04-03 | Universal Electronics Inc. | Automotive storage and playback device and method for using the same |
| UA81224C2 (uk) | 2001-05-02 | 2007-12-25 | Euro Celtic S A | Дозована форма оксикодону та її застосування |
| ATE506064T1 (de) * | 2001-06-05 | 2011-05-15 | Control Delivery Systems | Analgetika mit verzögerter wirkstoffabgabe |
| EP1392250A2 (en) | 2001-06-08 | 2004-03-03 | Endo Pharmaceuticals Inc. | Controlled release dosage forms using acrylic polymer, and process for making the same |
| AU2002316738B2 (en) | 2001-07-18 | 2009-01-08 | Euro-Celtique S.A. | Pharmaceutical combinations of oxycodone and naloxone |
| ES2562682T3 (es) * | 2002-01-15 | 2016-03-07 | Novartis Ag | Sistema para liberar aerosoles del espacio muerto anatómico efectivo |
| ES2327034T3 (es) * | 2002-03-26 | 2009-10-23 | Euro-Celtique S.A. | Composiciones recubiertas con gel de liberacion sostenida. |
| NZ536252A (en) * | 2002-04-29 | 2007-06-29 | Alza Corp | Methods and dosage forms for controlled delivery of oxycodone |
| EP1509050A4 (en) * | 2002-05-27 | 2009-03-18 | Ntt Docomo Inc | MOBILE COMMUNICATION SYSTEM, TRANSMISSION STATION, RECEPTION STATION, RELAY STATION, METHOD FOR DECISION-MAKING THE COMMUNICATION PATH AND PROGRAM FOR DECISION-MAKING THE COMMUNICATION PATH |
| BR0304960A (pt) | 2002-05-31 | 2005-01-04 | Alza Corp | Formas de dosagem e composições para liberação osmótica de dosagens variáveis de oxicodona |
| WO2004004693A1 (en) * | 2002-07-05 | 2004-01-15 | Collgegium Pharmaceutical | Abuse-deterrent pharmaceutical compositions of opiods and other drugs |
| US20040058946A1 (en) * | 2002-07-05 | 2004-03-25 | Buchwald Stephen L. | Abuse-resistant prodrugs of oxycodone and other pharmaceuticals |
| WO2004016618A1 (en) * | 2002-08-15 | 2004-02-26 | Noramco, Inc. | Oxycodone-hydrochloride polymorhs |
| AU2003291103A1 (en) | 2002-11-15 | 2004-06-15 | Branded Products For The Future | Pharmaceutical composition |
| MXPA05005781A (es) * | 2002-11-29 | 2005-12-12 | Forest Laboratories | Combinacion de ibuprofeno y oxicodona para alivio de dolor agudo. |
| FR2850576B1 (fr) * | 2003-02-05 | 2007-03-23 | Ethypharm Sa | Composition comprenant un melange de principes actifs, et procede de preparation |
| ATE454169T1 (de) | 2003-03-13 | 2010-01-15 | Controlled Chemicals Inc | Oxycodon- konjugate mit niedrigerem missbrauch- potential und ausgedehnter tätigkeitsdauer |
| US6864370B1 (en) * | 2003-06-05 | 2005-03-08 | Zhaiwei Lin | Process for manufacturing oxycodone |
| DE10336400A1 (de) | 2003-08-06 | 2005-03-24 | Grünenthal GmbH | Gegen Missbrauch gesicherte Darreichungsform |
| TW201509943A (zh) * | 2004-03-30 | 2015-03-16 | Euro Celtique Sa | 含有小於25ppm14-羥可待因酮之羥可酮鹽酸鹽之組成物、醫藥劑型、延遲釋出口服劑型及醫藥上可以接受的包裝 |
| US20070172958A1 (en) * | 2004-03-30 | 2007-07-26 | Euro-Celtique S.A. | Methods for detecting 14-hydroxy codeinone and codeinone |
| US20050226929A1 (en) | 2004-04-12 | 2005-10-13 | Jianbo Xie | Controlled release opioid analgesic formulation |
| WO2006004672A1 (en) | 2004-06-25 | 2006-01-12 | Tessera, Inc. | Components with posts and pads |
| GB0421149D0 (en) | 2004-09-23 | 2004-10-27 | Johnson Matthey Plc | Preparation of oxycodone |
| PT2112153E (pt) | 2005-03-04 | 2010-12-17 | Euro Celtique Sa | Método de diminuír a quantidade de cetonas insaturadas em alfa-beta em composições de opiáceos |
| US8067597B2 (en) | 2005-06-16 | 2011-11-29 | Mallinckrodt Llc | Synthetic route to 14-hydroxyl opiates through 1-halo-thebaine or analogs |
| EP1971607A2 (en) | 2005-11-22 | 2008-09-24 | Controlled Chemicals, Inc. | Processes for reducing contaminating michael acceptor levels in oxycodone and other compositions |
| US20070117826A1 (en) | 2005-11-23 | 2007-05-24 | Forest Laboratories, Inc. | Pharmaceutical formulations comprising ibuprofen, oxycodone, and 14-hydroxycodeinone |
| JP5695296B2 (ja) | 2006-03-02 | 2015-04-01 | マリンクロッド エルエルシー | 低レベルのアルファ,ベータ−不飽和ケトン化合物を伴うモルフィナン−6−オン生成物の製造方法 |
| SA07280459B1 (ar) | 2006-08-25 | 2011-07-20 | بيورديو فارما إل. بي. | أشكال جرعة صيدلانية للتناول عن طريق الفم مقاومة للعبث تشتمل على مسكن شبه أفيوني |
| AU2007329451B2 (en) | 2006-12-04 | 2012-08-30 | Noramco, Llc | Process for reducing impurities in oxycodone base |
| CA2671518C (en) * | 2006-12-04 | 2016-06-07 | Noramco, Inc. | Process for preparing oxycodone having reduced levels of 14-hydroxycodeinone |
| GB2450691A (en) | 2007-07-02 | 2009-01-07 | Alpharma Aps | One-pot preparation of oxycodone from thebaine |
| ES2659168T3 (es) * | 2010-07-02 | 2018-03-14 | Johnson Matthey Public Limited Company | Proceso para la síntesis y purificación de oxicodona |
| US9062062B1 (en) * | 2013-12-18 | 2015-06-23 | Cody Laboratories, Inc. | Synthesis of oxycodone hydrochloride |
-
2005
- 2005-03-15 TW TW103140205A patent/TW201509943A/zh unknown
- 2005-03-15 TW TW101106699A patent/TWI483944B/zh not_active IP Right Cessation
- 2005-03-15 TW TW094107824A patent/TWI365880B/zh not_active IP Right Cessation
- 2005-03-23 MY MYPI2011004160A patent/MY172063A/en unknown
- 2005-03-23 MY MYPI20051252A patent/MY143205A/en unknown
- 2005-03-23 MY MYPI20090495A patent/MY154985A/en unknown
- 2005-03-29 PE PE2005000361A patent/PE20050880A1/es active IP Right Grant
- 2005-03-30 SI SI200531950T patent/SI2305683T1/sl unknown
- 2005-03-30 ES ES10011787.8T patent/ES2585576T3/es not_active Expired - Lifetime
- 2005-03-30 KR KR1020187021551A patent/KR20180088505A/ko not_active Ceased
- 2005-03-30 EP EP10011793.6A patent/EP2314589B1/en not_active Expired - Lifetime
- 2005-03-30 KR KR1020147030767A patent/KR20140133615A/ko not_active Ceased
- 2005-03-30 LT LTEP10011793.6T patent/LT2314589T/lt unknown
- 2005-03-30 CA CA2913355A patent/CA2913355C/en not_active Expired - Lifetime
- 2005-03-30 CA CA2774121A patent/CA2774121C/en not_active Expired - Lifetime
- 2005-03-30 ME MEP-2016-148A patent/ME02482B/me unknown
- 2005-03-30 EP EP10011792.8A patent/EP2305683B2/en not_active Expired - Lifetime
- 2005-03-30 KR KR1020147001423A patent/KR20140021716A/ko not_active Withdrawn
- 2005-03-30 PL PL10011787.8T patent/PL2319846T3/pl unknown
- 2005-03-30 PT PT100117928T patent/PT2305683E/pt unknown
- 2005-03-30 HU HUE11186168A patent/HUE028233T2/en unknown
- 2005-03-30 HR HRP20110425TT patent/HRP20110425T4/hr unknown
- 2005-03-30 EA EA200601520A patent/EA013208B1/ru not_active IP Right Cessation
- 2005-03-30 ES ES05730345T patent/ES2362282T5/es not_active Expired - Lifetime
- 2005-03-30 RS RS20160561A patent/RS54941B1/sr unknown
- 2005-03-30 ES ES10011793T patent/ES2938727T3/es not_active Expired - Lifetime
- 2005-03-30 KR KR1020117022725A patent/KR101200700B1/ko not_active Expired - Lifetime
- 2005-03-30 PL PL05730345.5T patent/PL1730151T5/pl unknown
- 2005-03-30 ES ES10011788.6T patent/ES2665287T3/es not_active Expired - Lifetime
- 2005-03-30 KR KR1020157009780A patent/KR20150046393A/ko not_active Ceased
- 2005-03-30 AU AU2005230826A patent/AU2005230826C1/en not_active Expired
- 2005-03-30 CN CN201210257643.5A patent/CN102838607B/zh not_active Expired - Lifetime
- 2005-03-30 AP AP2006003738A patent/AP2232A/xx active
- 2005-03-30 US US11/093,626 patent/US7129248B2/en not_active Expired - Lifetime
- 2005-03-30 PT PT05730345T patent/PT1730151E/pt unknown
- 2005-03-30 SI SI200532083A patent/SI2319846T1/sl unknown
- 2005-03-30 EP EP11186168.8A patent/EP2426132B1/en not_active Expired - Lifetime
- 2005-03-30 PT PT111861688T patent/PT2426132E/pt unknown
- 2005-03-30 SG SG10201406658VA patent/SG10201406658VA/en unknown
- 2005-03-30 LT LTEP10011788.6T patent/LT2316837T/lt unknown
- 2005-03-30 EP EP10011788.6A patent/EP2316837B1/en not_active Expired - Lifetime
- 2005-03-30 EP EP10011791A patent/EP2311839A1/en not_active Withdrawn
- 2005-03-30 ES ES10011792.8T patent/ES2535618T3/es not_active Expired - Lifetime
- 2005-03-30 PT PT100117886T patent/PT2316837T/pt unknown
- 2005-03-30 NZ NZ601326A patent/NZ601326A/xx not_active IP Right Cessation
- 2005-03-30 ES ES11186168.8T patent/ES2566363T3/es not_active Expired - Lifetime
- 2005-03-30 CN CN2010101515524A patent/CN101812065B/zh not_active Ceased
- 2005-03-30 DK DK10011787.8T patent/DK2319846T3/en active
- 2005-03-30 PL PL10011792T patent/PL2305683T3/pl unknown
- 2005-03-30 HU HUE10011788A patent/HUE036451T2/hu unknown
- 2005-03-30 ME MEP-2011-91A patent/ME01299B/me unknown
- 2005-03-30 RS RS20110209A patent/RS51693B2/sr unknown
- 2005-03-30 WO PCT/US2005/010666 patent/WO2005097801A1/en not_active Ceased
- 2005-03-30 KR KR1020097019843A patent/KR100993911B1/ko not_active Expired - Lifetime
- 2005-03-30 SI SI200531289T patent/SI1730151T2/sl unknown
- 2005-03-30 CA CA2557845A patent/CA2557845C/en not_active Expired - Lifetime
- 2005-03-30 RS RS20160189A patent/RS54598B1/sr unknown
- 2005-03-30 EP EP10011787.8A patent/EP2319846B1/en not_active Expired - Lifetime
- 2005-03-30 KR KR1020067022569A patent/KR100930730B1/ko not_active Expired - Lifetime
- 2005-03-30 ME MEP-2018-71A patent/ME03068B/me unknown
- 2005-03-30 CN CN201210256799.1A patent/CN102887903B/zh not_active Expired - Lifetime
- 2005-03-30 DE DE602005026786T patent/DE602005026786D1/de not_active Expired - Lifetime
- 2005-03-30 SG SG201101934-6A patent/SG170736A1/en unknown
- 2005-03-30 CN CN2010106226501A patent/CN102127086B/zh not_active Expired - Lifetime
- 2005-03-30 DE DE202005021371U patent/DE202005021371U1/de not_active Expired - Lifetime
- 2005-03-30 CN CN2005800106471A patent/CN1960994B/zh not_active Expired - Lifetime
- 2005-03-30 SG SG201101931-2A patent/SG170735A1/en unknown
- 2005-03-30 JP JP2007506507A patent/JP4912294B2/ja not_active Expired - Lifetime
- 2005-03-30 NZ NZ602862A patent/NZ602862A/en not_active IP Right Cessation
- 2005-03-30 AT AT05730345T patent/ATE501150T1/de active
- 2005-03-30 KR KR1020197038467A patent/KR20200000499A/ko not_active Ceased
- 2005-03-30 KR KR1020127017379A patent/KR20120106784A/ko not_active Ceased
- 2005-03-30 DK DK10011792T patent/DK2305683T3/en active
- 2005-03-30 HU HUE10011787A patent/HUE029913T2/en unknown
- 2005-03-30 DK DK10011788.6T patent/DK2316837T3/en active
- 2005-03-30 BR BRPI0508758A patent/BRPI0508758B8/pt active IP Right Grant
- 2005-03-30 MX MX2013004023A patent/MX363228B/es unknown
- 2005-03-30 PT PT100117878T patent/PT2319846T/pt unknown
- 2005-03-30 DK DK05730345.5T patent/DK1730151T4/da active
- 2005-03-30 SI SI200532046T patent/SI2426132T1/sl unknown
- 2005-03-30 RS RS20180297A patent/RS57000B1/sr unknown
- 2005-03-30 PL PL10011788T patent/PL2316837T3/pl unknown
- 2005-03-30 ME MEP-2016-58A patent/ME02411B/me unknown
- 2005-03-30 KR KR1020127006697A patent/KR20120046309A/ko not_active Ceased
- 2005-03-30 KR KR1020137000989A patent/KR101410141B1/ko not_active Expired - Lifetime
- 2005-03-30 AR ARP050101243A patent/AR049012A1/es active IP Right Grant
- 2005-03-30 EP EP05730345.5A patent/EP1730151B2/en not_active Expired - Lifetime
- 2005-03-30 RS RS20150215A patent/RS53891B1/sr unknown
- 2005-03-30 NZ NZ549430A patent/NZ549430A/en not_active IP Right Cessation
- 2005-03-30 PL PL11186168T patent/PL2426132T3/pl unknown
- 2005-03-30 SI SI200532200T patent/SI2316837T1/en unknown
- 2005-03-30 KR KR1020107013435A patent/KR20100069719A/ko not_active Ceased
- 2005-03-30 DK DK11186168.8T patent/DK2426132T3/en active
-
2006
- 2006-03-29 US US11/391,897 patent/US20060173029A1/en not_active Abandoned
- 2006-08-29 ZA ZA200607181A patent/ZA200607181B/en unknown
- 2006-09-04 CR CR8593A patent/CR8593A/es unknown
- 2006-09-08 MX MX2020008878A patent/MX2020008878A/es unknown
- 2006-09-12 IL IL178038A patent/IL178038A/en active IP Right Grant
- 2006-09-28 MA MA29351A patent/MA28492B1/fr unknown
- 2006-10-26 EC EC2006006953A patent/ECSP066953A/es unknown
- 2006-10-27 NO NO20064935A patent/NO337669B1/no unknown
-
2007
- 2007-01-16 US US11/653,530 patent/US7674798B2/en not_active Expired - Lifetime
- 2007-01-16 US US11/653,531 patent/US7674799B2/en not_active Expired - Lifetime
- 2007-01-16 US US11/653,529 patent/US7683072B2/en not_active Expired - Lifetime
- 2007-03-29 US US11/729,741 patent/US7674800B2/en not_active Expired - Lifetime
- 2007-07-04 FI FI20070284U patent/FI7843U1/fi not_active IP Right Cessation
- 2007-08-02 DK DK200700214U patent/DK200700214U3/da not_active IP Right Cessation
- 2007-09-03 AT AT0052507U patent/AT9952U1/de not_active IP Right Cessation
-
2009
- 2009-01-30 AU AU2009200335A patent/AU2009200335B2/en not_active Expired
- 2009-03-04 US US12/380,800 patent/US20090227615A1/en not_active Abandoned
-
2010
- 2010-02-24 US US12/711,948 patent/US8822687B2/en not_active Expired - Lifetime
- 2010-09-29 US US12/893,681 patent/US20110207762A1/en not_active Abandoned
-
2011
- 2011-03-17 JP JP2011058728A patent/JP5118223B2/ja not_active Expired - Lifetime
- 2011-05-23 CY CY20111100503T patent/CY1111487T1/el unknown
- 2011-06-30 IL IL213889A patent/IL213889A/en active IP Right Grant
- 2011-06-30 IL IL213890A patent/IL213890A/en active IP Right Grant
- 2011-06-30 CR CR20110376A patent/CR20110376A/es unknown
- 2011-06-30 IL IL213888A patent/IL213888A0/en active IP Right Grant
- 2011-06-30 IL IL213891A patent/IL213891A0/en active IP Right Grant
- 2011-07-12 IL IL214050A patent/IL214050A/en active IP Right Grant
- 2011-11-17 JP JP2011251260A patent/JP5378486B2/ja not_active Expired - Lifetime
-
2012
- 2012-02-06 US US13/366,755 patent/US9073933B2/en active Active
- 2012-05-30 JP JP2012123160A patent/JP5543991B2/ja not_active Expired - Lifetime
-
2014
- 2014-03-07 JP JP2014045713A patent/JP2014139205A/ja active Pending
- 2014-10-23 IL IL235298A patent/IL235298A/en active IP Right Grant
-
2015
- 2015-03-30 HR HRP20150358TT patent/HRP20150358T1/hr unknown
- 2015-04-17 CY CY20151100357T patent/CY1116327T1/el unknown
- 2015-05-28 JP JP2015108759A patent/JP2015187134A/ja active Pending
- 2015-05-29 US US14/725,110 patent/US20150258086A1/en not_active Abandoned
- 2015-05-29 US US14/725,210 patent/US20150265598A1/en not_active Abandoned
- 2015-05-29 US US14/725,153 patent/US9522919B2/en not_active Expired - Lifetime
- 2015-09-03 IL IL241117A patent/IL241117B/en active IP Right Grant
-
2016
- 2016-03-01 HR HRP20160223T patent/HRP20160223T1/hr unknown
- 2016-03-02 US US15/058,390 patent/US20160176888A1/en not_active Abandoned
- 2016-03-02 US US15/058,363 patent/US20160175298A1/en not_active Abandoned
- 2016-03-02 US US15/058,960 patent/US20160175300A1/en not_active Abandoned
- 2016-03-02 US US15/058,812 patent/US20160250205A1/en not_active Abandoned
- 2016-03-02 US US15/058,420 patent/US9777011B2/en not_active Expired - Fee Related
- 2016-03-02 US US15/058,379 patent/US20160175299A1/en not_active Abandoned
- 2016-03-02 US US15/058,912 patent/US20160176889A1/en not_active Abandoned
- 2016-03-04 US US15/061,640 patent/US20160264588A1/en not_active Abandoned
- 2016-03-04 US US15/061,416 patent/US20160251363A1/en not_active Abandoned
- 2016-03-04 US US15/061,541 patent/US20160185789A1/en not_active Abandoned
- 2016-03-04 US US15/061,681 patent/US20160185790A1/en not_active Abandoned
- 2016-03-04 US US15/061,733 patent/US20160251365A1/en not_active Abandoned
- 2016-03-04 CY CY20161100188T patent/CY1117228T1/el unknown
- 2016-03-04 US US15/061,588 patent/US20160251364A1/en not_active Abandoned
- 2016-03-04 US US15/061,477 patent/US20160185788A1/en not_active Abandoned
- 2016-07-15 HR HRP20160885TT patent/HRP20160885T1/hr unknown
- 2016-07-20 CY CY20161100711T patent/CY1117841T1/el unknown
-
2017
- 2017-03-08 JP JP2017043530A patent/JP6503392B2/ja not_active Expired - Lifetime
- 2017-09-29 US US15/721,167 patent/US10259819B2/en not_active Expired - Lifetime
-
2018
- 2018-03-08 HR HRP20180407TT patent/HRP20180407T1/hr unknown
- 2018-03-12 CY CY20181100298T patent/CY1120171T1/el unknown
- 2018-07-31 JP JP2018144003A patent/JP2018184462A/ja active Pending
-
2019
- 2019-01-30 US US16/262,683 patent/US10407434B2/en not_active Expired - Lifetime
- 2019-08-30 US US16/557,937 patent/US10689389B2/en not_active Expired - Lifetime
- 2019-11-20 US US16/690,052 patent/US10696684B2/en not_active Expired - Lifetime
-
2020
- 2020-06-12 US US16/900,600 patent/US11384091B2/en not_active Expired - Lifetime
-
2021
- 2021-03-15 US US17/202,320 patent/US11236098B2/en not_active Expired - Lifetime
-
2022
- 2022-07-11 US US17/811,736 patent/US20230159548A1/en not_active Abandoned
-
2023
- 2023-06-07 US US18/330,932 patent/US12060361B2/en not_active Expired - Lifetime
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101410141B1 (ko) | 25 ppm 미만의 14-하이드록시코데이논을 가지는 염산 옥시코돈의 제조방법 | |
| AU2017204741A1 (en) | Process for preparing oxycodone hydrochloride having less than 25 ppm 14-hydroxycodeinone | |
| HK1149561A (en) | Process for preparing oxycodone hydrochloride having less than 25ppm 14-hydroxycodeinone | |
| HK1155729B (en) | Process for preparing oxycodone hydrochloride having less than 25ppm 14-hydroxycodeinone | |
| HK1098745B (en) | Process for preparing oxycodone hydrochloride having less than 25 ppm 14-hydroxycodeinone | |
| HK1149560A (en) | Oxycodone hydrochloride composition having less than 25 ppm 14-hydroxycodeinone |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A107 | Divisional application of patent | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20150415 Application number text: 1020147030767 Filing date: 20141031 |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20150515 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20150728 Patent event code: PE09021S01D |
|
| E90F | Notification of reason for final refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Final Notice of Reason for Refusal Patent event date: 20171222 Patent event code: PE09021S02D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20180625 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20171222 Comment text: Final Notice of Reason for Refusal Patent event code: PE06011S02I Patent event date: 20150728 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |
|
| J201 | Request for trial against refusal decision | ||
| PA0104 | Divisional application for international application |
Comment text: Divisional Application for International Patent Patent event code: PA01041R01D Patent event date: 20180725 Application number text: 1020147030767 Filing date: 20141031 |
|
| PJ0201 | Trial against decision of rejection |
Patent event date: 20180725 Comment text: Request for Trial against Decision on Refusal Patent event code: PJ02012R01D Patent event date: 20180625 Comment text: Decision to Refuse Application Patent event code: PJ02011S01I Appeal kind category: Appeal against decision to decline refusal Decision date: 20180817 Appeal identifier: 2018101003139 Request date: 20180725 |
|
| J121 | Written withdrawal of request for trial | ||
| PC1202 | Submission of document of withdrawal before decision of registration |
Comment text: [Withdrawal of Procedure relating to Patent, etc.] Withdrawal (Abandonment) Patent event code: PC12021R01D Patent event date: 20180817 |
|
| PJ1201 | Withdrawal of trial |
Patent event code: PJ12011R01D Patent event date: 20180817 Comment text: Written Withdrawal of Request for Trial Appeal identifier: 2018101003139 Request date: 20180725 Appeal kind category: Appeal against decision to decline refusal Decision date: 20180817 |