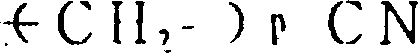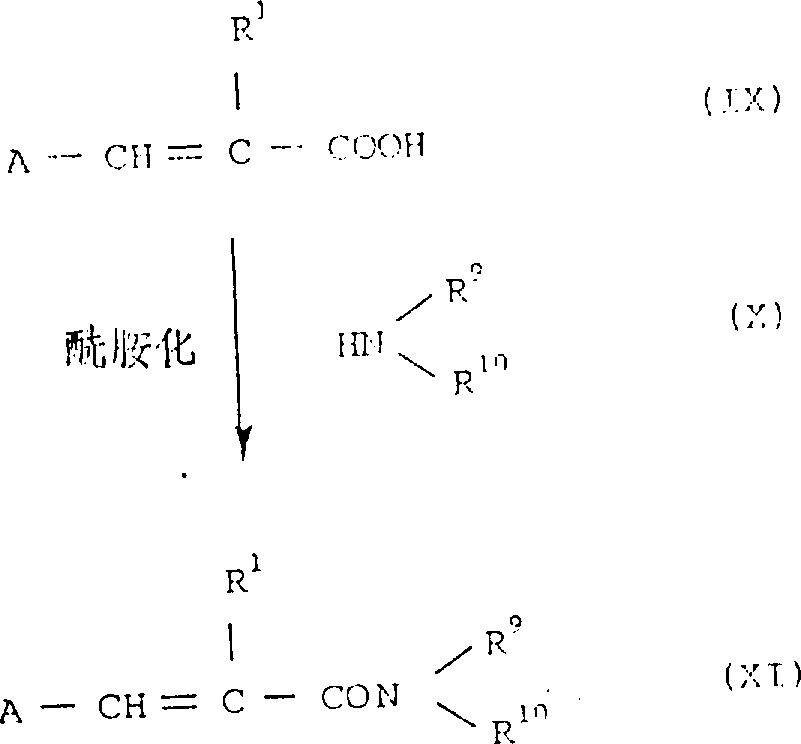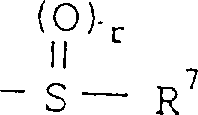CN1031992C - 苯醌衍生物及其药用 - Google Patents
苯醌衍生物及其药用 Download PDFInfo
- Publication number
- CN1031992C CN1031992C CN90107622A CN90107622A CN1031992C CN 1031992 C CN1031992 C CN 1031992C CN 90107622 A CN90107622 A CN 90107622A CN 90107622 A CN90107622 A CN 90107622A CN 1031992 C CN1031992 C CN 1031992C
- Authority
- CN
- China
- Prior art keywords
- group
- compound
- formula
- representative
- low alkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C50/00—Quinones
- C07C50/26—Quinones containing groups having oxygen atoms singly bound to carbon atoms
- C07C50/28—Quinones containing groups having oxygen atoms singly bound to carbon atoms with monocyclic quinoid structure
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/16—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms
- C07D295/18—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms acylated on ring nitrogen atoms by radicals derived from carboxylic acids, or sulfur or nitrogen analogues thereof
- C07D295/182—Radicals derived from carboxylic acids
- C07D295/185—Radicals derived from carboxylic acids from aliphatic carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/01—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms
- C07C255/31—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms having cyano groups bound to acyclic carbon atoms of a carbon skeleton containing rings other than six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/01—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms
- C07C255/32—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms having cyano groups bound to acyclic carbon atoms of a carbon skeleton containing at least one six-membered aromatic ring
- C07C255/41—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms having cyano groups bound to acyclic carbon atoms of a carbon skeleton containing at least one six-membered aromatic ring the carbon skeleton being further substituted by carboxyl groups, other than cyano groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/57—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing cyano groups and carboxyl groups, other than cyano groups, bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C317/00—Sulfones; Sulfoxides
- C07C317/44—Sulfones; Sulfoxides having sulfone or sulfoxide groups and carboxyl groups bound to the same carbon skeleton
- C07C317/46—Sulfones; Sulfoxides having sulfone or sulfoxide groups and carboxyl groups bound to the same carbon skeleton the carbon skeleton being further substituted by singly-bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/51—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton
- C07C323/55—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton the carbon skeleton being unsaturated and containing rings other than six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/51—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton
- C07C323/56—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atoms of the thio groups bound to acyclic carbon atoms of the carbon skeleton the carbon skeleton containing six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/62—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C45/00—Preparation of compounds having >C = O groups bound only to carbon or hydrogen atoms; Preparation of chelates of such compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C47/00—Compounds having —CHO groups
- C07C47/52—Compounds having —CHO groups bound to carbon atoms of six—membered aromatic rings
- C07C47/575—Compounds having —CHO groups bound to carbon atoms of six—membered aromatic rings containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C50/00—Quinones
- C07C50/38—Quinones containing —CHO or non—quinoid keto groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/42—Unsaturated compounds containing hydroxy or O-metal groups
- C07C59/54—Unsaturated compounds containing hydroxy or O-metal groups containing six-membered aromatic rings and other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/64—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings
- C07C59/66—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings the non-carboxylic part of the ether containing six-membered aromatic rings
- C07C59/68—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings the non-carboxylic part of the ether containing six-membered aromatic rings the oxygen atom of the ether group being bound to a non-condensed six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/76—Unsaturated compounds containing keto groups
- C07C59/90—Unsaturated compounds containing keto groups containing singly bound oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C65/00—Compounds having carboxyl groups bound to carbon atoms of six—membered aromatic rings and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C65/21—Compounds having carboxyl groups bound to carbon atoms of six—membered aromatic rings and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups containing ether groups, groups, groups, or groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C66/00—Quinone carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/734—Ethers
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/738—Esters of keto-carboxylic acids or aldehydo-carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/36—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D211/40—Oxygen atoms
- C07D211/44—Oxygen atoms attached in position 4
- C07D211/46—Oxygen atoms attached in position 4 having a hydrogen atom as the second substituent in position 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/54—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
- C07D213/55—Acids; Esters
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/22—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D277/30—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/38—Nitrogen atoms
- C07D277/44—Acylated amino or imino radicals
- C07D277/46—Acylated amino or imino radicals by carboxylic acids, or sulfur or nitrogen analogues thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D309/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings
- C07D309/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D309/04—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/02—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings
- C07D333/04—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom
- C07D333/06—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom not condensed with other rings not substituted on the ring sulphur atom with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring carbon atoms
- C07D333/24—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/14—The ring being saturated
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/12—Systems containing only non-condensed rings with a six-membered ring
- C07C2601/16—Systems containing only non-condensed rings with a six-membered ring the ring being unsaturated
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- Gastroenterology & Hepatology (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Heterocyclic Compounds Containing Sulfur Atoms (AREA)
- Pyridine Compounds (AREA)
- Pyrane Compounds (AREA)
- Hydrogenated Pyridines (AREA)
- Thiazole And Isothizaole Compounds (AREA)
- Medicinal Preparation (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
用于治疗肝病的下式苯醌化合物:
式中各基团定义详见说明书。
Description
本发明涉及具有很好的药物活生的苯醌衍生物:
具体讲,本发明涉及一种用作肝病治疗剂的新的苯醌衍生物。
由于每种肝病的病因、症状、及病理不同,且包括很多未知因素,目前很难为这类病找到好的治疗剂。
目前,广泛应用于治疗和预防肝病的代表性药物及临床上常用药物包括甘草甜素制剂,尽管该类制剂据信对肝病综合症、肝硬变或肝炎以及对肝手术后的保护有效,但其疗效不是很强。更糟的是其具有甾类副作用。而且其有效的剂型为静脉注射,当口服给药时无活性。
因此,急需得到一种安全性高且口服有效的药物。
在上述的情况下,本发明者已经开始了寻找肝病治疗剂的研究。
结果,他们发现下面将叙述的苯醌衍生物可达到本发明的目的。
具有药理治性的苯醌衍生物的实例包括在如日本专利公开特许(Japanese Patent Laid—OpenNos.)223150/1987,223150/1987和177934/1983中所描述的化合物。
日本专利公开特许223150/1987中所述的苯醌衍生物的化学结构与本发明的化合物(1)不同,其具有止喘活性,因此在药理活性上也与本发明的化合物不同。
日本专利公开特许177934/1983还介绍了一个苯醌衍生物,其疗效和化学结构均不同于本发明的化合物。
日本专利公开特许185921/1988也介绍了一种肝病治疗剂,然而其化学结构不同于本发明的化合物。
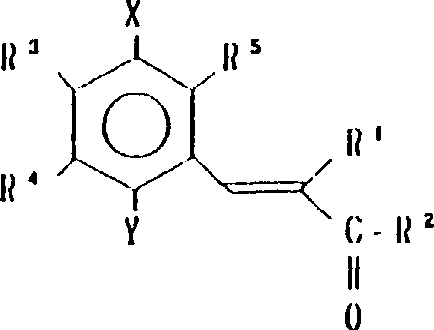
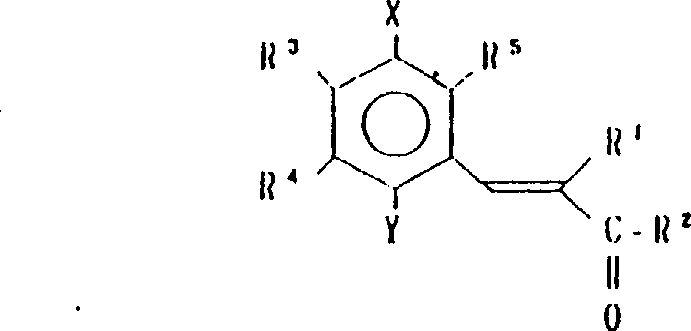
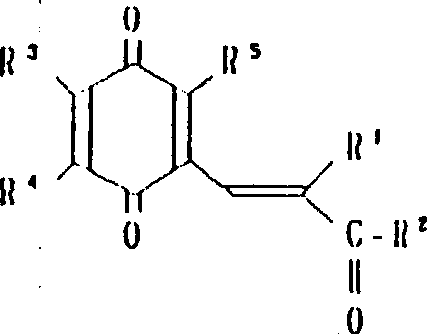
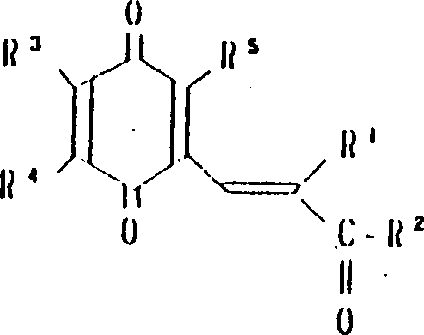
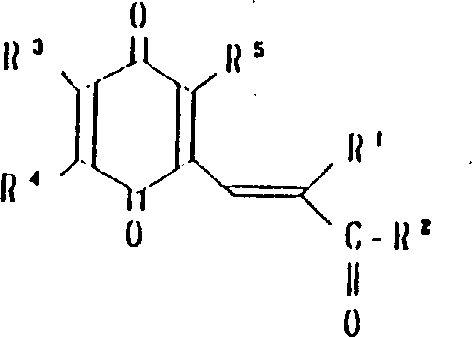
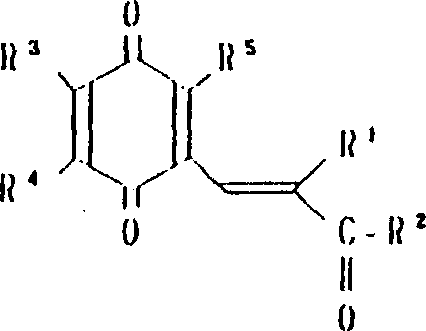
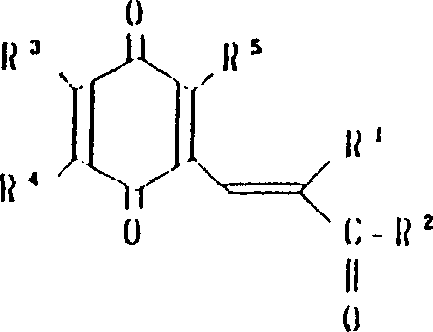
本发明涉及到由下列的通式(1)所代表的苯醌衍生物及其药理上可接受的盐,其中,A为下式所代表的基团:(其中,R3,R4和R5可相同或不同,其可以是氢原予、低级烷基、或是低级烷氧基)。A或者是下式所代表的基团:(其中R3,R4和R5可相同或不同,其可以是氢原子、低级烷基、或低级烷氧基,X和Y可以相同或不同,可以是羟基、由式(OCH2)nOR6(其中n为0或1,R6为低级烷基)所代表的基团或是酰基)。
R1为2-20个碳原子的烷基,环烷基,环烷基烷基,烯基,炔基,芳基烷基,由式
所代表的基团(其中P为1-10的整数),杂芳基烷基,由式
所代表的基团(其中q为1-6的整数),由式
所代表的基团1(其中r为0或是1-2的整数,R7为低级烷基,环烷基或是芳基),或是由式—O—R8所代表的基团(其中R8为低级烷基或芳基),或是由式
所代表的基团(其中,S为1-3的整数)。
R2为由式—OR8所代表的基团(其中R8为氢原子或低级烷基)或由下式所代表的基团:(其中R9和R10可相同或不同,其可以是氢原子,低级烷基、羟基烷基或杂芳基,R9和R10可相互连接与氮原子链合成环,还可再含有一个氮原子和/或氧原子,该环可被取代)。
本发明的化合物中的R3、R4、R5、R6、R7和R8中所用的术语“低级烷基”是指:分1—8个碳原子的直链或支链烷基,其实例包括甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、戊基(正戊基)、异戊基、新戊基、叔戊基、1-甲基丁基、2—甲基丁基、3—甲基丁基、1,2—二甲基丙基、己基、异己基、1—甲基戊基、2—甲基戊基、3—甲基戊基、1,1—二甲基丁基、1,2—二甲基丁基、2,2—二甲基丁基、1,3—二甲基丁基、2,3—二甲基丁基、3,3—二甲基丁基、1—乙基丁基、2—乙基丁基、1,1,2—三甲基丙基、1,2,2—三甲基丙基、1—乙基—1—甲基—丙基、1—乙基—2—甲基丙基和辛基,其中,甲基、乙基、丙基、异丙基等为优选。
R3,R4和R5中所用的术语“低级烷氧基”指由上述低级烷基衍生物得到的烷氧基。如:甲氧基、乙氧基和正丙氧基,其中,甲氧基为最优选者。
R3,R4和R5合起来的优选实例包括:R5为甲基而R3和R4各为甲氧基,R3,R4和R5各为甲氧基,R3为甲氧基,R4为乙氧基而R5为甲基。
R1中所用的术语“烷基”是指含有2-20个碳原子的烷基,含2-12个碳原子的烷基为优选,含7-12个碳原子的烷基最优选。
术语“环烷基”是指:例如含有3-6个碳原子的环烷基。
环烷基的优选实例包括:由上述含3-6个碳原子的环烷基衍生来的环烷基甲基。
芳基烷基的代表性实例包括芳基,其中苯环可以被一或多个低级烷基如甲基、乙基、低级烷氧基如甲氧基、羟基、羧基或卤素所取代。
杂芳基烷基的代表性实例包括杂芳基甲基,其中杂环可被低级烷基如甲基,低级烷氧基如甲氧基,羟基或卤素所取代。杂芳基的实例包括那些含有氮、氧或硫原子者,如:噻唑基,吡喃基,噻二唑基,及吡啶基。
在由式
所代表的基团中,P为1-10的整数,P最优选1-4的整数。
在由式
所代表的基团中,q和B如上定义,q最优选1-4的整数,在B的定义中,R7的优选实例包括甲基,环己基和苯基。
当X和Y均为羟基时,本发明的化合物为氢醌化合物。
在X和Y的定义中,R6最优选甲基,酰基可以是由脂肪、芳香和杂环化合物衍生得到者,酰基优选者包括如:低级烷酰基、如:甲酰、乙酰、丙酰、丁酰、戊酰、异戊酰及新酰基;芳酰基如苯甲酰、甲苯酰、及萘甲酰基:和杂环芳酰基如糖酰、烟酰和异烟酰基。优选的酰基的实例包括:含1-6个碳原予的低级烷基衍生得到的酰基。例如:乙酰、丙酰和丁酰基。
药理上适宜的盐包括:例如:苯醌衍生物与无机酸如盐酸、氢溴酸、硫酸和磷酸所形成的盐、与有机酸所成的盐如:乙酸盐、马来酸盐,酒石酸盐,甲磺酸盐,苯磺酸盐,及甲苯磺酸盐;以及与氨基酸,如精氨酸、天冬氨酸及谷氨酸所形成的盐。
同时,本发明的某些化合物为金属盐形式,如Na、K、Ca、或Mg盐,这些金属盐也在本发明中药理可接受的盐的范围中。
而且,从化学结构可见,本发明的化合物各有一个双键,因而可以立体异构体的形式存在(顺式和反式异构体),所以这些化合物在本发明的范围内。
本发明的化合物的代表性的制备过程如下叙述:
在上述分子式(II)和(III)中,X,Y,R1,R2,R3和R4分别如前述定义。
在上述反应式中,化合物(II)和(III)均为本发明的化合物,由上述反应式可见,化合物(III)的苯醌衍生物可以用氧化剂处理化合物(II)的氢醌衍生物来制备,而化合物(II)可通过还原化合物(III)来制备。
为氧化化合物(II)的氢醌衍生物,用氯化铁六水合物或氧化铅为氧化剂,在该例中,氧化剂的优选用量为氢醌的摩尔数的3-10倍,优选的溶剂包括例如:苯、乙酸乙酯、二噁烷、乙醇和1,2-二甲氧基乙烷、均可含有水,反应在0-80℃进行,优选20-40℃,反应时间通常约为1—12小时。
另一方面,为使苯醌化合物还原成本发明所需的氢醌化合物,优选的还原剂包括,例如:硼氢化钠和亚硫酸氧钠。而各含有适量水的乙醇、四氢呋喃、乙酸乙酯和1,2-二甲氧基乙醇为优选溶剂,反应温度优选0-40℃,更优选0-20℃。
制备方法2
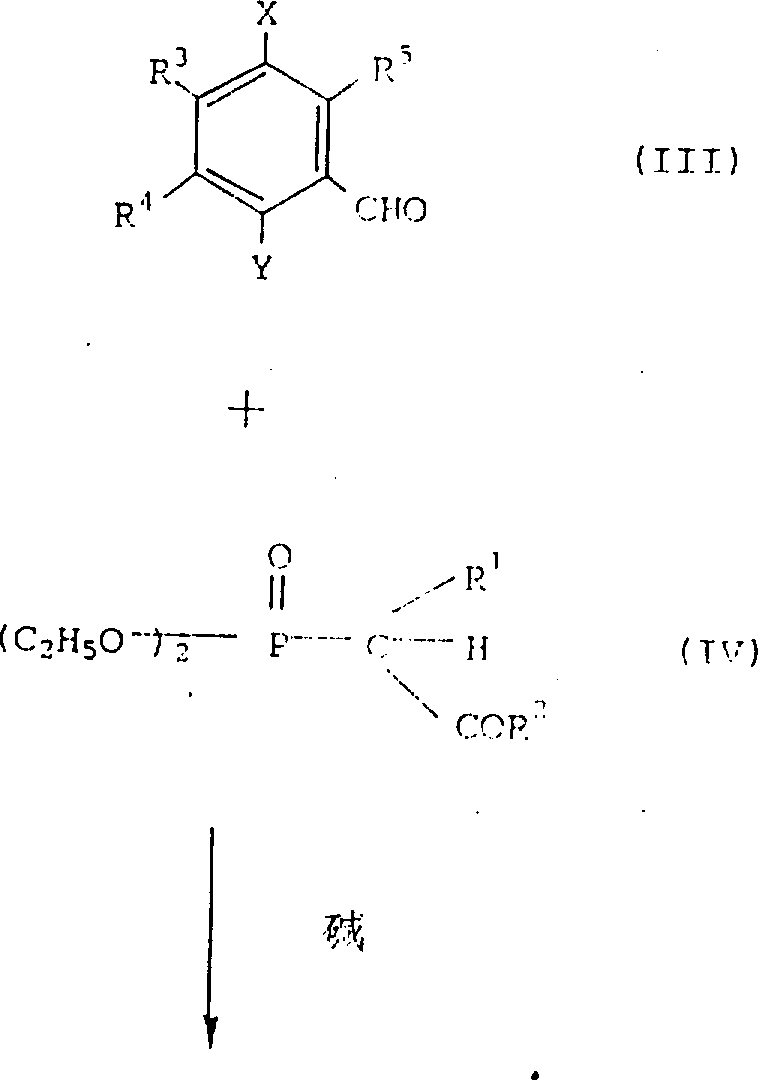
在上述式中,R1,R2,R3,R4,R5,X和Y分别如前述定义。
具体的,由通式(III)所代表的醛衍生物在碱存在下,与通式(IV)所代表的膦酸酯经过Wittig反应(见,如,J.A.C.S.,83,1733(1961))制备得到通式(II)所代表的所要物质。
该反应中所用的碱包括碱金属氢化物如:氢化钠、氢化钾及碱金属的醇化物,如甲醇钠,乙醇钠及叔丁氢化钾,优选的反应溶剂包括:苯、甲苯、二氯甲烷、四氢呋喃、二噁烷、二甲氧基乙烷及二甲基甲酰胺,反应温度优选0-100℃,较优选20-80℃。
制备方法3
具体地,式(V)所代表的化合物按常法用碱皂化,式(VI)所代表的化合物被脱甲氧甲基化以制备式(VII)所代表的化合物。
皂化过程在使用如:常用的醇苛性苏打或苛性钾下进行。脱甲氧甲基过程在,例如:丙酮、二噁烷、二甲氧基乙烷或其水溶液中进行,同时有矿物酸存在,如:盐酸或硫酸,或是有机酸如:对甲苯磺酸或樟脑磺酸。反应温度优选20-80℃。
在氧化过程中,当使用氧化剂如氯化铁六水合物进行直接氧化过程中,脱甲氧甲基化和氧化同时进行,因此使本发明的所要物质之一的、分子式(VIII)所代表的苯醌化合物可以在一步中制备完成。
制备方法4
具体地,羧酸或由通式(IX)所代表的反应衍生物可与通式(X)所代表的氨基化合物进行酰胺化反应,因此得到所要化合物之一的化合物(XI)。
化合物(IX)的反应衍生物包括:例如:酰基卤如酰基氯,酰基溴;酰基叠氮;用N-羟基苯并三唑,N-羟基琥珀酰亚胺等活化的酯;对称酸酐;以及用烷基羧酸,对甲苯磺酸等得到的混合酸酐等。
当游离羧酸用做化合物(IX)时,反应优选在缩合剂存在下进行,如:二环己基碳化二亚胺,1,1'-碳酰二咪唑等。
反应进行中使用化合物(IX)或其反应衍生物且化合物(X)的比例为与化合物(IX)等摩尔数,或其中-种的摩尔数值稍许过量,反应在惰性有机溶剂中进行,如:吡啶、四氢呋喃、二噁烷、乙醚、苯、甲苯、二甲苯、二氯甲烷、二氯乙烷、氯仿、二甲基甲酰胺、乙酸乙酯或乙腈。
在反应中,加入碱,如三乙基胺、吡啶、甲基吡啶、二甲基吡啶、N,N-二甲基苯胺、碳酸钾、或氢氧化钠,根据反应衍生物的种类,常有助于反应的顺利进行。
反应温度根据反应衍生物的种类而变化,并无特定限制。
方法4所制备的氢醌衍生物和苯醌衍生物可以根据上述的制备方法1分别进行氧化和还原,以分别制备苯醌和氢醌衍生物。
起始物的制备方法:
具体讲,起始物可通过三乙基亚磷酸盐与α-卤代羧酸衍生物(XVII)反应进行制备。
当R2为式-OR8所代表的基团时,其中R8如前述定义,起始物也可按下列方法进行制备。上式中,R1和R8分别如前述定义,X为卤原子。
具体讲,起始物可通过将三乙基膦酰乙酸酯(XIX)用卤代烷在碱存在下进行烷基化反应来制备(J.Org.Chem.)30,2208(1965))。
在该例中,碱金属氢化物,如氢化钠或氢化钾或碱金属醇化物如:甲醇钠、乙醇钠或叔丁醇钾可用做碱、优选的溶剂包括二甲基甲酰胺,二甲基乙酰胺,N-甲基吡咯烷酮、四氢呋喃及1,2-二甲氧基乙烷,反应温度为20-80℃,优选40-60℃。
具体讲,起始物可根据常规方法,用通式(XXII)所代表的卤化物(其中Hal为氯、溴、碘等)与金属烷基化合物中的阴离子及甲酰剂进行制备,烷基金属化合物包括例如丁基锂、仲丁基锂和LDA,而甲酰剂包括:例如二甲基甲酰胺、和N-甲基甲酰苯胺,在该反应中所用的溶剂包括,例如:乙醚、四氢呋喃和二甲氧基乙烷,反应温度约为-80-0℃。
具体讲,棓酸的三烷基醚(XXIII)可以在浓氢溴酸乙酸合溶剂中加热,选择地断裂4位的醚键,因此得到通式(XXIV)的化合物。
随后,在碱存在下,使该化合物与适宜的卤代烷反应,同时进行成醚和成酯反应,因此得到通戊(XXV)的化合物。碱金属氢化物如氢化钠或氢化钾以及碱金属碳酸盐如碳酸钠、或碳酸钾可用做碱。
溶剂优选二甲基甲酰胺、二甲基乙酰胺、四氧呋喃、二噁烷等,反应温度为30-80℃。上述化合物用碱金属皂化,随后用常规方法进行溴化,得到溴代化合物(XXVII )。根据Meyer等人所提出的方法(见Chem.Ber.,89,511(1956)),该化合物在有催化量的金属铜存在时,在浓碱溶液中加热回流转化成酚化合物(XXVIII)。
通式(XXIX)所代表的化合物可根据Minami等的方法(见Chem.Phar.Bull.,28(5),1648(1980))进行制备。例如,通过化合物(XXVIII)与卤代羧酸酯(如氯代羧酸乙酯或氯代羧酸异丁酯)在碱存在下反应,制备混合酸酐,随后用硼氢化钠或硼氢化锂将该混合酸酐还原,得到通式(XXIX)的化合物。所用的碱包括,例如:有机碱如:三乙基胺,吡啶及二异丙基乙基胺,和无机碱如碳酸钠和碳酸钾。四氢呋喃、乙醚、二噁烷、二甲氧基乙烷等可用做溶剂,反应温度优选0-30℃。
化合物(XXIX)可以根据常规方法进行溴化和甲氧甲基化,将其转化成所要化合物(XXXI),溴化在氯仿、苯、甲醇或乙酸乙酯等溶剂中在0-30℃的反应温度下进行。甲氧甲基化反应可以使化合物(XXX)与氯代甲基甲基醚在例如二甲基甲酰胺、二甲基乙酰胺、N-甲基吡咯烷酮、四氧呋喃、二甲氧基乙烷、二氯代甲烷或甲苯等溶剂中,在碱金属氢化物,如氢化钠或氢化钾,以及有机碱如二异丙基乙基胺或二甲基氨基吡啶的存在下进行。
具体说,3,4,5-三烷氧基酚(XXXIII)按上述方法进行甲氧甲基化,产物用金属烷基化合物处理,得到阴离予,随后使其与甲酰剂反应,得到甲酰化合物(XXXV)。在该例中,乙醚、四氢呋喃等用做反应溶剂、反应温度优选0-30℃,该甲酰化合物(XXXV)与过酸进行Baeyer-Villiger反应,制备得到0-甲酰化合物。该0-甲酰化合物经水解得到酚化合物,其如此烷基化得到通式(XXXVI)所代表的化合物。在Baeyer-Villiger反应中所用的过酸包括,例如:过乙酸、过苯甲酸和m-氯代过苯甲酸。二氯甲烷、氯仿、二氯乙烷等为优选的反应溶剂,反应温度优选0-20℃。根据上述方法,化合物(XXXVI)可通过重甲酰化,很容易转变成甲酰化合物(XXXVII)。
本发明的作用将根据下列的本发明中具有代表性化合物的药理实验进行详细叙述。
药理实验
试验1 对大鼠D-半乳糖胺(GalN)诱导的急性肝炎模型的作用。
(1)方法
给Fischer(F344)雄性大鼠(重约180g),皮下注射300mg/Kg的GalN,诱导急性肝炎,每种试验化合物分散于0.5%的甲基纤维素水溶液中,并在GalN注射1小时后,将上述溶液以100mg/kg的剂量给大鼠口服。
大鼠注射GalN 48小时后,取大鼠尾部血样利用肝网质试验(HPT)测定凝血时间,同时利用酶法测定血浆中GPT活性。
每种化合物对GalN诱导的肝炎的抑制百发率列于表1中:
(2)结果
结果见表1.其化合物对应表4和表5。
表1(第1部分)
实施例2:对小鼠丙酸菌属痤疮(P.acnes)-脂多糖(LPS)诱导的爆发性肝炎模型的作用。
(1)方法
给五周龄的雄性Balb/c小鼠静脉注射经热灭活的P.acnes 1mg/小鼠于P.acnes注射7天后,以1微克/小鼠的剂量再给其静注LPS,以诱导爆发性肝炎,将各试验化合物分散于0.5%的甲基纤维素溶液中,并将其于静注LPS前30分钟时,以100mg/kg的剂量给小鼠口服。
于LPS静注后24小时,测定存活率及存活小鼠的血浆GPT活性,用各试验化合物治疗的小鼠相对于用P.acnes-LPS诱导的致死率和肝损伤的结果列于表2中。
(2)结果
结果见表2。
同表1表2中化合物号与表4和表5相同。
表2(第1部分)
试验化合物对P.acnes-LPS
诱导的死亡和肝中毒的作用
| 试验化合物 | 存活率(%) | GPT(存活率) |
| 对照组(%)/试验化合物组(%) | 对照组(%)/试验化合物组(%) | |
| 化合物号20化合物号79化合物号125化合物实例号14化合物号135化合物号137化合物号141化合物号142化合物号144 | 30/708/5040/9040/1000/500/300/2522/10036/100 | 848±316/293±651639/1009±196353±67/219±52485±139/297±50------------------761/381±19696±160/280±31 |
表2(第2部分)
| 试验化合物 | 存活率(%) | GPT(存活率) |
| 对照组(%)/试验化合物组(%) | 对照组(%)/试验化合物组(%) | |
| 化合物号149化合物实例号4化合物号176化合物号183化合物号217 | 40/6722/6422/800/220/80 | 353±67/209±66761/358±40761/297±50-------- |
实验实例3
毒性试验
将实例4中所制备的本发明的化合物,实例15中所制备的化合物No.137,和实例15中所制备的化合物No.142给七周龄的雄性SLC:SD大鼠口服给药一周(剂量300mg/kg),结果,未发现化合物致死现象。
由实验实例1和2显然可见,本发明的化合物为肝病的一种有效的治疗剂。
因此,本发明的化合物可有效地用做动物包括人类的各种肝病的治疗剂和预防剂。具体用于治疗和预防,例如:慢性肝炎,急性肝炎,肝中毒,病毒性肝炎,酒精中毒性肝炎,黄疸及晚期的肝硬变。
同时,由实验实例3可见,本发明的化合物毒性很低,即安全性高,在许多病例中,由于疾病的性质,本发明的化合物必须长期反复给药,从这方面看,本发明有很高的价值。
当本发明的化合物用做肝病的治疗剂和预防剂时,其可以口服给药,如粉剂、颗粒剂、胶囊剂,糖浆等剂型,也可非肠道给药,如:栓剂、注射剂、外用制剂及滴剂等剂型。本发明化合物的剂量因症状、年令、及肝病的种类等的不同而显著变化,通常,本发明化合物的给药剂量约为成人每天0.1-1,000mg,优选2-500mg,更优选5-150mg,可一次或分几次给药。
利用药剂学中通常使用的载体,根据常规方法可以将本发明的化合物制成药物制剂。
具体地,当制备口服固体制剂时,将活性成分与赋形剂混合,如需要,还可混入粘合剂、崩解剂、润滑剂、着色剂、矫味剂等,随后制成剂、包衣片、颗粒剂、粉剂和胶囊剂。
赋形剂包括,例如:乳糖、玉米淀粉、果糖、葡萄糖、山梨醇、结晶纤维素及二氧化硅。粘合剂的实例包括:聚乙烯醇、聚乙烯醚、乙基纤维素、甲基纤维素、阿拉伯胶、黄蓍胶、明胶、紫胶、羟丙基纤维素、羟丙甲基纤维素、柠檬酸钙、糊精及果胶。润滑剂包括,例如:硬脂酸镁、滑石、聚乙烯醇、二氧化硅和氢化植物油,药剂学中允许使用的使何着色剂均可用做着色剂,矫味剂包括,例如:可可粉,薄荷醇,芳香粉,薄荷粉,冰片及粉状肉桂皮,当然,糖衣,明胶包衣,以及(若需要)其它包衣可用于这些片剂和颗粒剂上。
当制备注射剂时,往活性成分中加入PH调节剂,缓冲剂,稳定剂,增溶剂等,随后,按常规方法制备皮下,肌肉和静脉注射剂。
本发明代表性实例将在此叙述,当然本发明并不限于此。
由于本发明的化合物具有双键,其可以顺式或反式异构体存在,在下列实例中,本发明的化合物除特别说明外,以反式异构体存在。
制备本发明的所要物质的最后步骤将以实例叙述,而实例中所用的起始物的制备步骤将在实例前的参考实例中叙述。
化学结构式中的下列符号具有如下意义:
Me:甲基
Et:乙基
n-pr:正丙基
MOMO:甲氧基亚甲氧基
iso-Pr:异丙基
Oct:辛基。
参考实例:
将100g的3,4,5-三乙氧基苯甲酸溶于150ml 48%HBr水溶液和300ml乙酸中,将所得溶液在100℃边搅拌边加热2小时,将反应混合物冷却,过滤,分离出沉淀,用水洗,固体用11乙醇重结晶,得到50g白色固体为所需化合物。
参考实例2
将参考实例1中所制备的50g 3,5-二乙氧基-4-羟基苯甲酸溶于300ml DMF(二甲基甲酰胺)中,并加153g碳酸钾,于室温下加41.3ml碘甲烷,将所得的混合物于50℃搅拌6小时后,冷却,倾入冰水,用乙酸乙酯提取,提取物用水洗,无水硫酸镁干燥,蒸除溶剂,得到63g所需化合物粗品(白色固体)。
1H-NMR(CDCl3)δ1.45(t,J=7Hz,6H),
3.87(s,3H),3.88(s,3H),4.12(q,J=7Hz,
4H),7.22(s,2H)
参考实例3
将实例2中所制备的63g 3,5-二乙氧基-4-甲氧基苯甲酸甲酯溶于200ml乙醇和80ml水中,并加44g苛性苏打,混和物于79℃搅拌2小时冷却,用稀盐酸弱酸化,用二氯甲烷提取,有机相经水洗,无水硫酸镁干燥,蒸除溶剂,得到48g所需化合物粗品(白色固体)。
1H-NMR(CDCl3)δ;1.48(t,J=7Hz,6H),
3.95(s,3H),4.15(q,J=7Hz,4H),7.36
(s,2H)
参考实例4
2-溴-3,5-二乙氧-4-甲氧苯甲酸
将48g参考实例3中所制备的3,5-二乙氧-3-甲氧基苯甲酸溶于300ml氯仿,加6ml水,随后在氯仿回流下,于8小时内,将13.4ml溴滴加其中,真空浓缩溶剂,得到68g所需化合物粗品(浅黄色固体)。
1H-NMR(CDCl3)δ;1.46(t,J=7Hz,3H),
1.47(t,J=7Hz,3H),3.95(s,3H),4.08
(q,J=7Hz,4H),7.34(s,1H)
参考实例5
3,5-二乙氧基-2-羟基-4-甲氧基苯甲酸
将68g参考实例4中所制备的2-溴-3,5-二乙氧基-4-甲氧基苯甲酸分散于260ml水中,随后加32g苛性苏打,和0.88g铜粉,混合物于120℃加热搅拌3小时,随后冷却,加碳,通过硅藻土过滤,滤液用140ml 6N盐酸中和,并加入11氯仿,混合物经液-液分离,有机相用水洗,干燥,并浓缩,得到53g所需化合物粗品(褐色固体)。
1H-NMR(CDCl3)δ;1.40(t,J=7Hz,3H),
1.43(t,J=7Hz,3H),4.00(s,3H),4.02
(q,J=7Hz,2H),4.13(q,J=7Hz,2H),
7.11(s,1H)
参考实例6
2,4-二乙氧基-3-甲氧基-6-甲基苯酚
将53g参考实例5中所制备的3,5-二乙氧基-2-羟基-3-甲氧基苯甲酸和45.5g三乙基胺溶于400ml的四氢呋喃中,于冰浴冷却下边搅拌边滴加含有48.4g氯代甲酸乙酯的100ml的四氢呋喃溶液,待滴加完毕后,将所形成的结晶过滤分离出,并用100ml四氢呋喃洗,将母液与结晶洗涤液合并,冰浴搅拌下,往其中加入含30.3g硼氢化钠的10%的水溶液,滴加完毕后,混合物于室温下搅拌1小时,用稀盐酸中和,用乙酸乙酯洗,用无水硫酸镁干燥并浓缩,浓缩物用硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=95∶5),得到40g所需的无色油状物质。
1H-NMR(CDCl3)δ;1.38(t,J=7Hz,3H),
1.40(t,J=7Hz,3H),2.19(s,3H),3.84
(s,3H),3.98(q,J=7Hz,2H),4.18(q,
J=7Hz,2H),5.45(s,1H),6.39(s,1H)
参考实例7
5-溴-2,4-二乙氧基-3-甲氧基-6-甲基苯酚
将40g参考实例6中所制备的2,4-二乙氧-3-甲氧基-6-甲基苯酚溶于200ml氯仿中,冰浴冷却搅拌下加10ml溴,向反应混合物加冰水,混合物经液-液分离,有机相用生理盐水饱和,无水硫酸镁干燥,溶剂蒸除得到54g所需的浅黄色油状物质。
1H-NMR(CDCl3)δ;1.38(t,J=7Hz,3H),
1.40(t,J=7Hz,3H),2.28(s,3H),3.87
(s,3H),3.97(q,J=7Hz,2H),4.16
(q,J=7Hz,2H),5.71(br,1H)
参考实例8
将参考实例7中所制备的54g 5-溴-2,4-二乙氧基-3-甲氧基-6-甲基酚溶于250ml二甲基甲酰胺,并在冰浴,搅拌下加8.5g氢化钠(55%油混悬液),混合物室温搅拌30分钟,再用冰冷却,滴加17.1g甲氧基亚甲基氯,滴加完毕,混合物于室温下再搅拌30分钟,加冰水,混合物用乙酸乙酯提取,有机相用水洗,无水硫酸镁干燥、浓缩,并用硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=95∶5),得到43.6g所需的无色油状物。
1H-NMR(CDCl3)δ;1.37(t,J=7Hz,3H),
1.41(t,J=7Hz,3H),2.36(s,3H),
3.58(s,3H),3.88(s,3H),4.02(q,J=
7Hz,2H),4.03(q,J=7Hz,2H),5.04
(s,2H)
参考实例9
将参考实例8中所制备的43.6g 5-溴-2,4-二氧基-6-甲基-1-甲氧基亚甲氯基苯溶于220ml四氢呋喃中,于-70℃滴加100ml正丁基锂(1.6M正已烷溶液混合物于-40℃搅拌30分钟,并滴加11.9g二甲基甲酰胺,后应混合物的温度恢复到室温,加氯化铵水溶液,随通后/用乙酸乙酯提取,有机相用水洗,干燥、浓缩,硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=90∶10)得到19.2 g所需的无色油状物。
1H-NMR;1.40(t,J=7Hz,6H),2.49(s,3H),
3.58(s,3H),3.88(s,3H),4.10(q,J=
7Hz,2H),4.18(q,J=7Hz,2H),5.01
(s,2H),10.37(s,lH)
参考实例10
1-甲氧基亚甲氧基-3,4,5-三甲氧基苯酚
将20g 3,4,5-三甲氧苯酚溶于100ml四氢呋喃,冰浴冷却搅拌下加7.1g氢化钠(55%油混悬液),随后在冰浴冷却下,加12.4ml甲氧甲基氯,混合物于室温下搅拌30分钟,反应混合物倾入冰水中,用乙酸乙酯提取,有机相用水洗、无水硫酸镁干燥、蒸除溶剂,残余物经硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=85∶15)得到30.4g所需的无色油状物。
参考实例11
2-甲氧基亚甲氧基-4,5,6-三甲氧基苯甲醛
将30.4g参考实例10中所制备的1-甲氧基亚甲氧基-3,4,5-三甲氧基苯酚溶于250ml无水乙醚中,于-20℃往其中滴加100ml正丁基锂(1.6M正己烷溶液),滴加完毕,混合物于室温下搅拌2小时,加14.6ml二甲基甲酰胺,加100ml冰水,混合物用乙酸乙酯提取,有机相用水洗,无水硫酸镁干燥,蒸除溶剂,残余物用硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=6∶4)得到25.5g所需的浅黄色油状物。
1H-NMR;3.48(s,3H),3.78(s,3H),3.88
(s,3H),3.92(s,3H),5.20 (s,2H),6.50
(s,1H),10.22(s,1H)
参考实例12
将参考实例11中所制备的12.8g 2-甲氧基亚甲氧基-4,5,6-三甲基苯甲醛溶于100ml二氯甲烷中,于室温搅拌下加入8.7g间氯过苯甲酸,混合物回流30分钟,并用冰冷却,加100ml硫代硫酸饱和水溶液,过滤分离出沉淀的结晶,母液用硫酸氢钠饱和水溶液洗,无水硫酸镁干燥,并浓缩,残余物与50ml乙醇、40ml水和21.3g氢氧化钾混合,混合物回流搅拌1小时,待其冷却后,倾入稀盐酸,用乙酸乙酯提取,有机相经水洗,无水硫酸镁干燥,蒸除溶剂得到11.5g所需化合物粗品(浅黄色油状物)。
参考实例13
将实例12所制备的11.5g 2-甲氧基亚甲氧基-4,5,6-三甲氧基苯酚和23.0g碳酸钾混悬于100ml二甲基甲酰胺中,混悬液于45℃搅拌,并滴加5.2ml碘甲烷,滴加完毕后,混合物加热30分钟,冷却,过滤分离,往母液中加1l水,混合物用乙酸乙酯提取,有机相用水洗,无水硫酸镁干燥,蒸除溶剂,残余物经硅胶柱层析纯化(流动相:正己烷、乙酸乙酯=85∶15),得到6.2g所需无色油状物。
NMR(CDO3)δ;3.52(s,3H),3.78(s,3H),
3.82(s,6H),3.94(s,3H),5.16(s,2H),
6.50(s,1H)
参考实例14
2-甲氧基亚甲氧基-3,4,5,6-四甲氧基苯甲醛
将参考实例13中所制备的6.2g 1-甲氧基亚甲氧基-2,3,4,5-四甲氧基苯溶于50ml无水乙醚,于-20℃边搅拌边加18ml正丁基锂(1.6M正己烷溶液),混合物于0℃搅拌30分钟后,温度恢复到-20℃,并滴加3.5ml二甲基甲酰胺,待加100ml水后,混合物用乙酸乙酯提取,有机相用水洗,无水硫酸镁干燥,并浓缩,残余物经硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=7∶3)得到5.6g所需的浅黄色油状物。
1H-NMR(δ);3.56(s,3H),3.84(s,3H),
3.86(s,3H),3.90(s,3H),4.02(s,3H),
5.12(s,2H),10.06(s,1H)
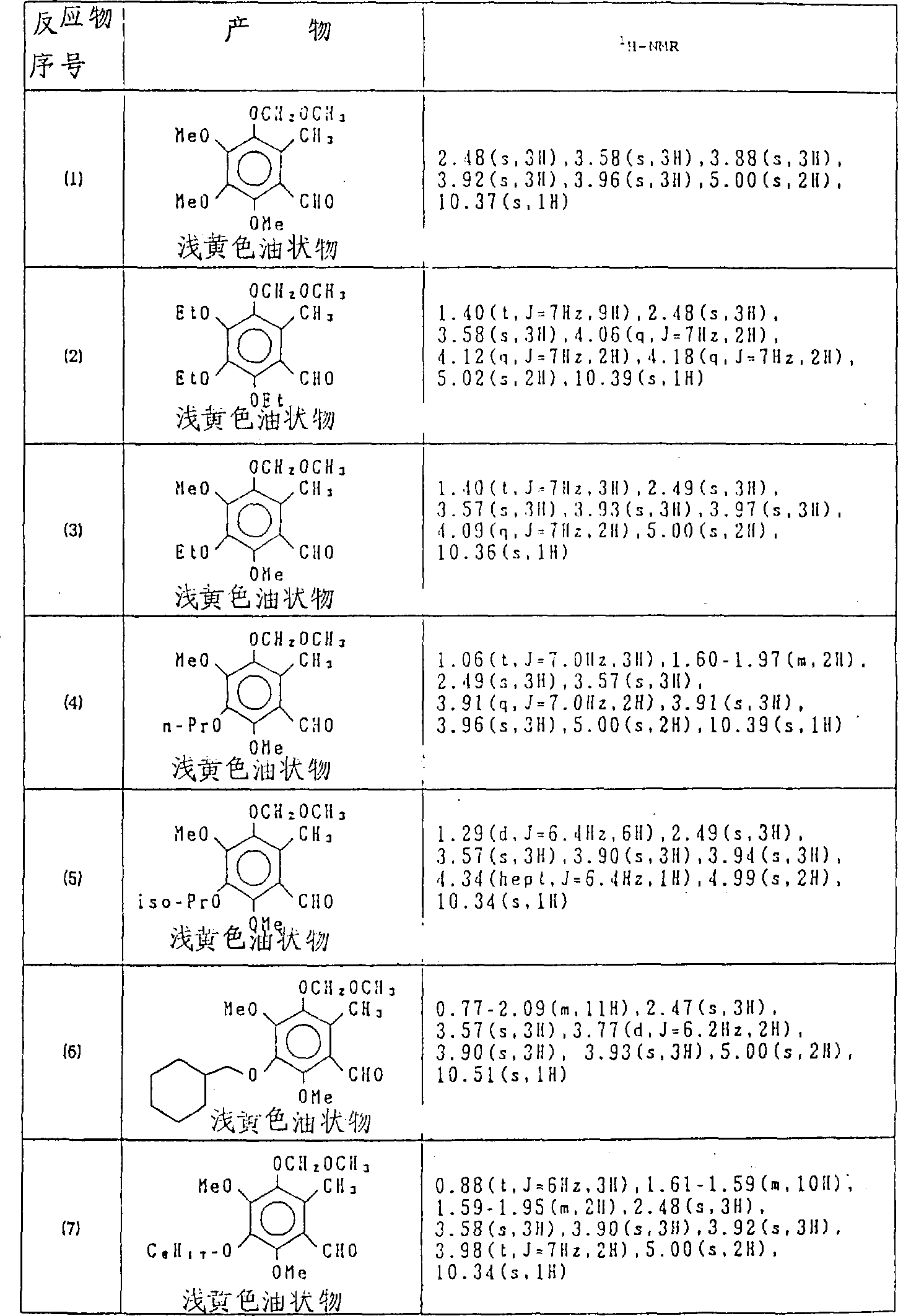
下列起始物(1)-(7)按与参考实例2-9同样的方法制备,化合物的具体数据列于表3中。
实施例1
将0.6g氢化钠(60%油混悬液)分散于5ml二甲基甲酰胺中,向该混悬液中滴加8.8g二乙基膦酰基-2-十一烷酸乙酯,待反应物混合物均匀后,将按参考实例1-9的方法所制备的2.7g的5-甲氧基亚甲氧基-6-甲基-2,3,4-三甲氧基苯甲醛(化合物No.(1))于室温下滴加进反应混合物,滴加完成后,混合物于60-70℃加热1小时,倾入冰水用乙酸乙酯提取,有机相用水洗,用无水硫酸镁干燥,蒸除溶剂,残余物经硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=95∶5),得到3.9g所需化合物。
1N-NMR(δ):0.86(t,J=6Hz,3H),1.00-1.50
(n,14H),1.36(t,J=7Hz,3H),2.08(s,3H),
2.00-2.25(m,2H),3.58(s,3H),3.68(s,3H),
3.88(s,6H),4.24(q,J=7Hz,2H),5.04
(s,2H),7.33 (s,1H)
化合物Nos.1-8,与实例1同法制备。
实施例2
将3.9g实例1中所制备的(E)3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-壬基-2-丙烯酸乙酯溶于30ml乙醇和5ml水中,并加1.7g氢氧化钠,混合物回流搅拌1小时,冷却,用正己烷提取,水相用1N稀盐酸酸化,用二氯甲烷提取,所得有机相用水洗,无水硫酸镁干燥,蒸除溶剂,得到3.6g所需的无色油状物。
1H-NMR(δ):0.86(t,J=6Hz,3H),1.01-1.59
(m,14H),2.00-2.28(m,2H),2.10(s,3H),
3.59(s,3H),3.69(s,3H),3.88(s,6H),
5.04(s,2H),7.50(s,1H)
化合物Nos9-16,与实例2同法制备。
实例3
(E)-3-〔5-(1-羟基-6-甲基-2,3,4-三甲氧基)-苯基〕-2-壬基-2-丙烯酸
将3.6g实例2中所制备的(E)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-壬基-2-丙烯酸溶于30ml丙酮和6N的盐酸中,所得溶液于70℃加热搅拌1小时,将反应混合物冷却,加100ml水,混合物用二氯甲烷提取,有机相用水洗,无水硫酸镁干燥,浓缩,得到3.4g所需的无色油状物。
1H-NMR(δ):0.86(t,J=6Hz,3H),1.01-1.60
(m,14H),2.01-2.32(m,2H),2.07(s,3H),
3.68(s,3H),3.89(s,3H),3.97(s,3H),
7.57(s,1H)
化合物Nos.17-124,如实例3的方法制备
实例4
将3.4g实例3所制备的(E)-3-〔5-(1-羟基-6-甲基-2,3,4-三甲氧基)苯基〕-2-壬基-2-丙烯酸溶于100ml乙酸乙酯中,并加3.4g氯化铁六水合物,混合物于室温搅拌2小时,加200ml水,所得的混合物分成两液相,有机相经水洗,无水硫酸镁干燥,蒸除溶剂,残余物经硅胶柱层析纯化(流动相:二氯甲烷∶甲醇=95∶5),并用正己烷重结晶,得到2.8g所需橙色固体化合物。
熔点:68℃。
1H-NMR(δ):0.86(t,J=6Hz,3H),1.02-1.60
(m,14H),1.96(d,J=2Hz,3H),2.01-2.22
(m,2H),3.99(s,3H),4.01(s,3H),7.20
(bs,1H)
化合物Nos.130-237如实例4方法制备
实例5
N-〔(E)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-壬基-2-丙烯酰基〕吗啉
将1.0g实例2中所制备的(E)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-壬基-2-丙烯酸和1.0ml三乙基胺溶于19ml四氢呋喃中,并在冰浴冷却下,搅拌着滴加0.45ml二乙基膦酰氯,所得混合物于室温下搅拌30分钟,往其中加1.0ml吗啉,继续搅拌2小时,往反应混合物中加水,混合物用乙酸乙酯提取,有机相用水洗,随后经无水硫酸镁干燥;蒸除溶剂,残余物经硅胶硅层析纯化(流动相:正己烷∶乙酸乙酯=3∶2),得到0.93g所需的无色油状物。
1H-NMR(δ):0.86(t,J=6Hz,3H),
100-1.50(m,14H),2.12(s,3H),2.00-2.20
(m,2H),3.50-3.80(m,8H),3.56(s,3H),
3.68(s,3H),388(s,6H),5.04(s,2H),
6.12(s,1H)
实例6
N-〔(E)-3-〔5(2,3-二甲氧基-6-甲基-1,4-醌基)〕-2-壬基-2-丙烯酰基〕吗啉
将0.93g实例5中所制备的N-〔(E)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-壬基-2-丙烯酰基〕吗啉溶于10ml乙酸乙酯中,并加10g氯化铁六水合物,混合物室温搅拌8小时,蒸除溶剂,残余物经硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=1∶2),得到0.75g所需的橙色油状物。
1H-NMR(δ):0.86(t,J=6Hz,3H),1.00-1.50
(m,14H),1.94(q,J=2Hz,3H),2.00-2.20
(m,2H),3.50-3.80(m,8H),3.96(s,3H),
3.98(s,3H),5.88(bs,1H)
化合物Nos.238-245如实例6的方法制备
实例7
N-〔(E)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-苄基-2-丙烯酰基〕-N'-甲基哌嗪
将按实例1和2方法所制备的1.0g的(E)-3-(5-(6-甲基-1-甲氧基亚甲氯基-2,3,4-三甲氧基)苯基〕-2-苄基-2-丙烯酸和1.0ml三乙基胺溶于20ml四氢呋喃中,在冰浴冷却下,不断搅拌着,往其中滴加0.50ml二乙基膦酰氯,混合物于室温下搅拌30分钟后,加1.0ml的N-甲基哌嗪,混合物搅拌2小时,往混合物中加水,所得混合物用乙酸乙酯提取,有机相经水洗,无水硫酸镁干燥,蒸除溶剂,残余物经硅胶柱层析纯化(流动相:氯仿∶乙醇=95∶5),得到0.92g所需的无色油状物。
1H-NMR(δ):1.70-2.30(m,4H),2.12(s,3H),
2.14(s,3H),3.44(s,2H),3.30-3.60
(m,4H),3.56(s,3H),3.72(s,3H),3.92
(s,3H),3.94(s,3H),5.04(s,2H),6.16
(s,1H),6.96-7.20(m,5H)
实例8
将实例7中所制备的0.92g N-〔(E)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-苄基-2-丙烯酰〕-N'-甲基哌嗪溶于10ml丙酮和2ml的6N盐酸中,所得溶液于70℃搅拌30分钟,真空蒸除溶剂,利用甲苯共沸物将水蒸除,得到0.91g所需的无色非晶型化合物。
1H-NMR(δ):2.04(s,3H),2.70(s,3H),
3.00-3.30(m,4H),3.30-3.60(m,4H),3.36
(s,2H),3.69(s,3H),3.84(s,3H),3.88
(s,3H),6.36(s,1H),6.90-7.30(m,5H)
实例9
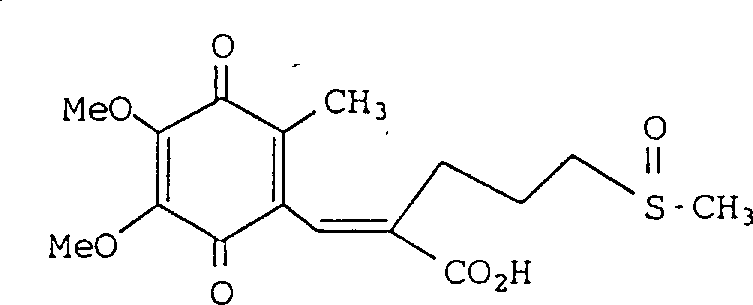
将按实例1-4方法所制备的1.8g(E)-3-〔5-(2,3-二甲氧基-6-甲基-1,4-醌基)〕-2-(3-甲基亚磺酰基)丙基-2-丙烯酸(化合物No.183)溶于50ml二氯甲烷中,所得溶液于-30℃冷却,边搅拌边分次将1.0g间氯过苯甲酸以小量加入,混合物于-30℃再搅拌30分钟,加水,有机相用水洗,无水硫酸镁干燥,蒸除溶剂,残余物经硅胶柱层析纯化(流动相:氯仿∶甲醇=90∶10),得到1.5g所需橙色固体化合物。1H-NMR(δ):1.60-1.85(m,2H),1.94(s,3H),1.90-2.60(m,4H),2.56(s,3H ),3.92(s,3H),3.96(s,3H),7.10(s,1H)
实例10
(E)-3-〔5-(3-乙氧基-2-甲氧基-6-甲基-1,4-氢醌基)〕-2-环乙基甲基-2-丙烯酸
将按实例1-4方法所制备的1.0g(E)-3-〔5-(3-乙氧基-2-甲氧基-6-甲基-1,4-醌基)〕-2-环己基甲基-2-丙烯酸(化合物No.159)溶于50ml乙酸乙酯中,加含10g亚硫酸氢钠的100ml水溶液,混合物转入分离器中并猛烈振摇,当红色有机相转成无色时,相分离完成,有机相经水洗,无水硫酸镁干燥,蒸除溶剂,得到1.0g所需的白色固体化合物。1H-NMR(δ):0.40-1.86(m,11H),1.38(t,
J=7Hz,3H),2.08(s,3H),2.01-2.17(m,2H),
3.86(s,3H),4.06(q,J=7Hz,2H),7.56
(s,1H)
化合物Nos.125-128按实例10的方法制备
实例11
(E)-3-〔5-(3-乙氧基-2-甲氧基-6-甲基-1,4-醌基)〕-2-环己基甲基-2-丙烯酸乙酯。
将按实例1方法所制备的2.0g(E)-3-〔5-(2,4-二甲氧基-3-乙氧基-1-甲氧基亚甲氧基-6-甲基)苯基)-2-环己基甲基-2-丙烯酸乙酯(化合物No.123)溶于50ml乙酸乙酯中,加20g氯化铁六水合物,等混合物于室温搅拌10小时后,加100ml乙酸乙酯和100ml水,有机相用水洗,无水硫酸镁干燥,蒸除溶剂,残余物经硅胶柱层析纯化,(流动相:正己烷∶乙酸乙酯=9∶1),得到1.4g所需的橙色油状物。
1H-NMR(δ):0.51-1.83(m,11H),1.33(t,
J=7Hz,3H),1.37(t,J=7Hz,3H),
1.93 (d,J=2Hz,3H),1.91-2.07(m,2H),
4.04(s,3H),4.20(q,J=7Hz,2H),4.23
(q,J=7Hz,2H),7.17(bs,1H)
化合物No.246按实例11的方法制备。
实例12
将实例10中所制备的1.0g(E)-3-〔5-(3-乙氧基-2-甲氧基-6-甲基-1,4-氢醌基)〕-2-环己基甲基-2-丙烯酸溶于10ml吡啶中,加2ml醋酐,混合物于室温下搅拌1小时后,加冰水,再搅拌30分钟,用6N盐酸弱酸化,用乙酸乙酯提取,提取物用水洗,无水硫酸镁干燥,蒸除溶剂,残余物用硅胶柱层析纯化,(流动相:氯仿:乙醇=95:5)得到1.2g所需的浅黄色非晶型化合物。
1H-NMR(δ):0.40-1.86(m,11H),1.31(t,
J=7Hz,3H),1.98(s,3H),2.01-2.17(m,2H),
2.23(s,3H),2.34(s,3H),3.86(s,3H),
4.04(q,J=7Hz,2H),7.39(bs,1H)
化合物No.129如实例12方法制备。
实例13
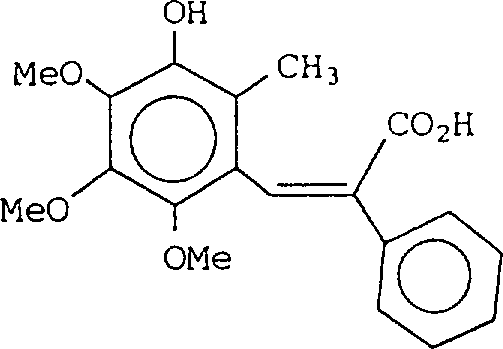
将0.69g氢化钠(60%油混悬液)分散于15ml二甲基甲酰胺中,于室温下滴加7.0g二乙基膦酰-2-苯基乙酸乙酯,待反应混合物均匀后,于室温下滴加3.15g 5-甲氧基亚甲氧基-6-甲基-2,3,4-三甲氧基苯甲醛(化合物No.(1)),反应于150℃进行5小时,反应混合物倾入冰水,用乙酸乙酯提取,有机相用水洗,无水硫酸镁干燥,蒸除溶剂,残余物用硅胶柱层析纯化(流动相:正己烷∶乙酸乙酯=90∶10),因此先得到1.8gE(反式)异构体,随后得到1.0g所需的Z(顺式)异构体,其为无色油。
1N-NMR(δ):1.00(t,J=7.5Hz,3H),2.17
(s,3H),3.56(s,3H),3.69(s,3H),3.86
(s,6H),4.06(q,J=7.5Hz,2H),5.01
(s,2H),6.86(s,1H),7.11-7.53(m,5H)
实例14
将实例13中所制备的1g(Z)-3-〔5-(6-甲基-1-甲氧基亚甲氧基-2,3,4-三甲氧基)苯基〕-2-苯基-2-丙烯酸乙酯,按实例2的方法用氢氧化钠水解,随后,按实例3的方法,于丙酮/6N盐酸中脱甲氧甲基,得到0.5g所需的白色固体化合物。
1N-NMR(δ):2.14(s,3H),3.70(s,3H),
3.85(s,3H),3.96(s,3H),6.90(s,1H),
7.17-7.57(m,5H)
实例15
下列表4和表5中所列的化合物,系按实例1-14的方法所制备的。
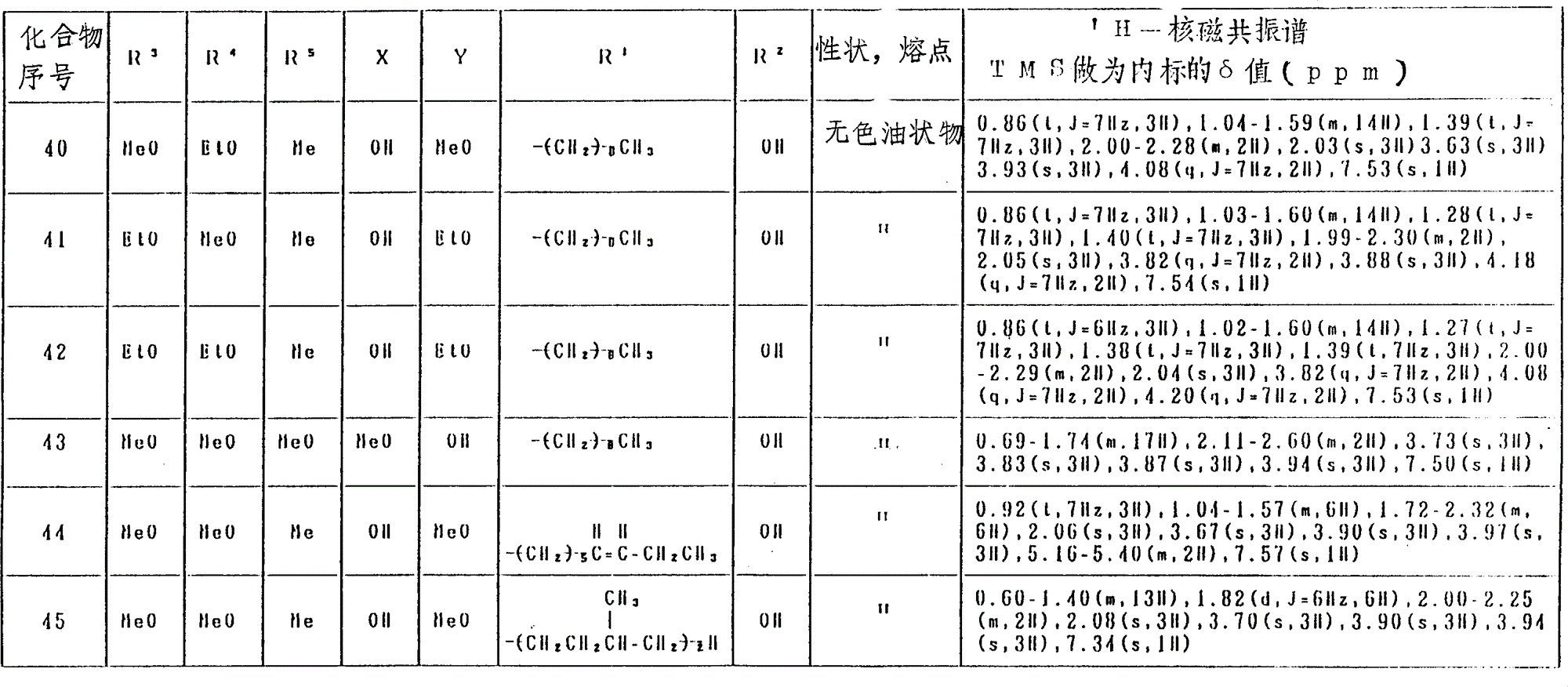
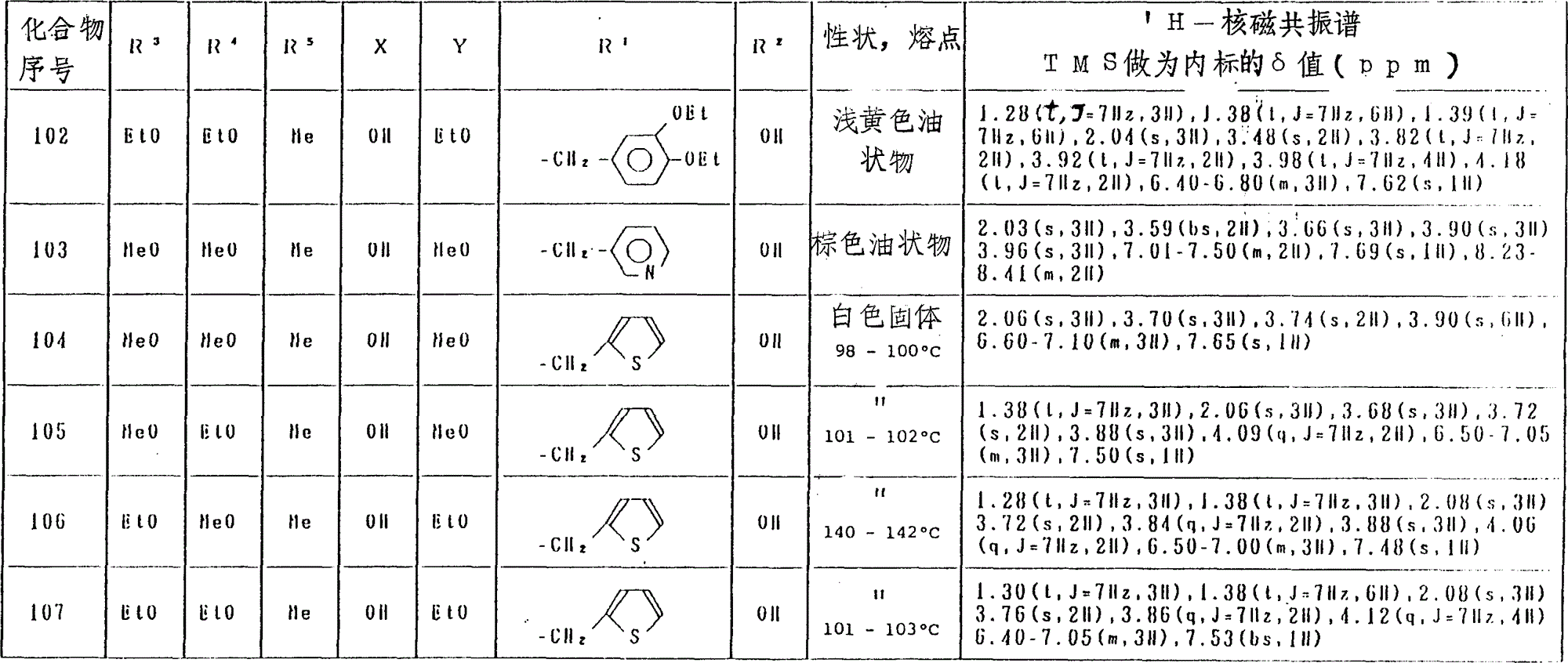
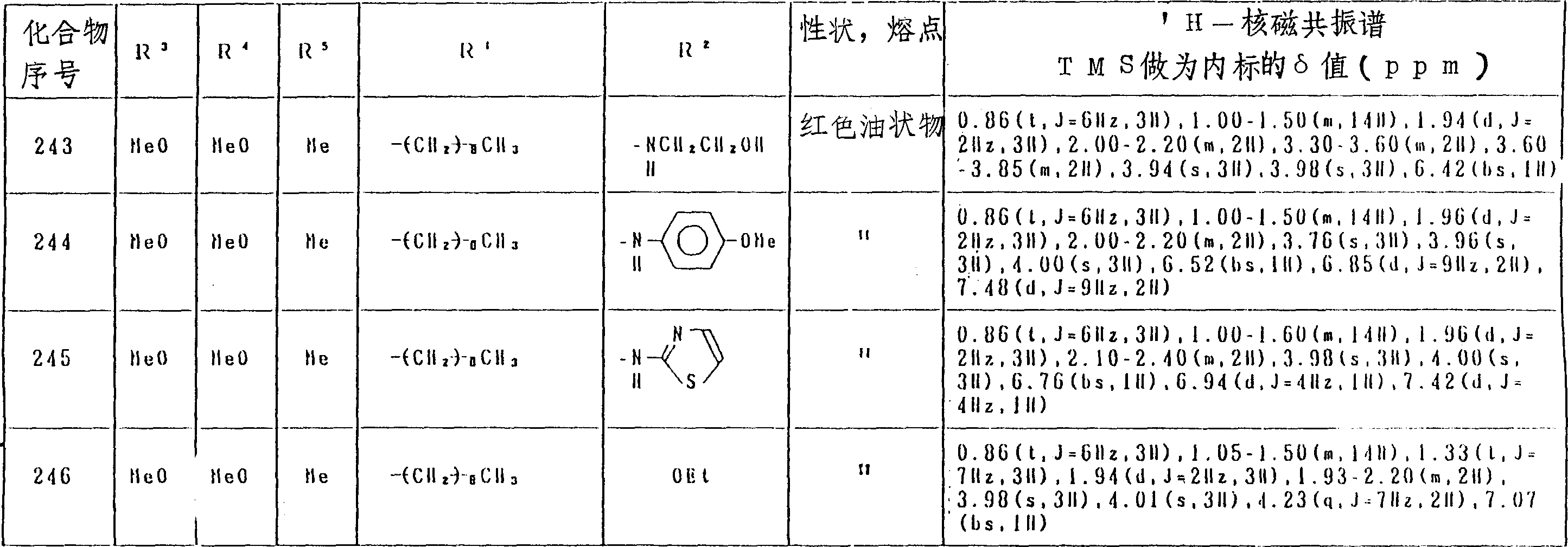
氢醌化合物(化合物No.1-129)和苯醌化合物(化合物No.130-246)分别列于表4和表5中。
表4(第2部分)
表4(第18部分)
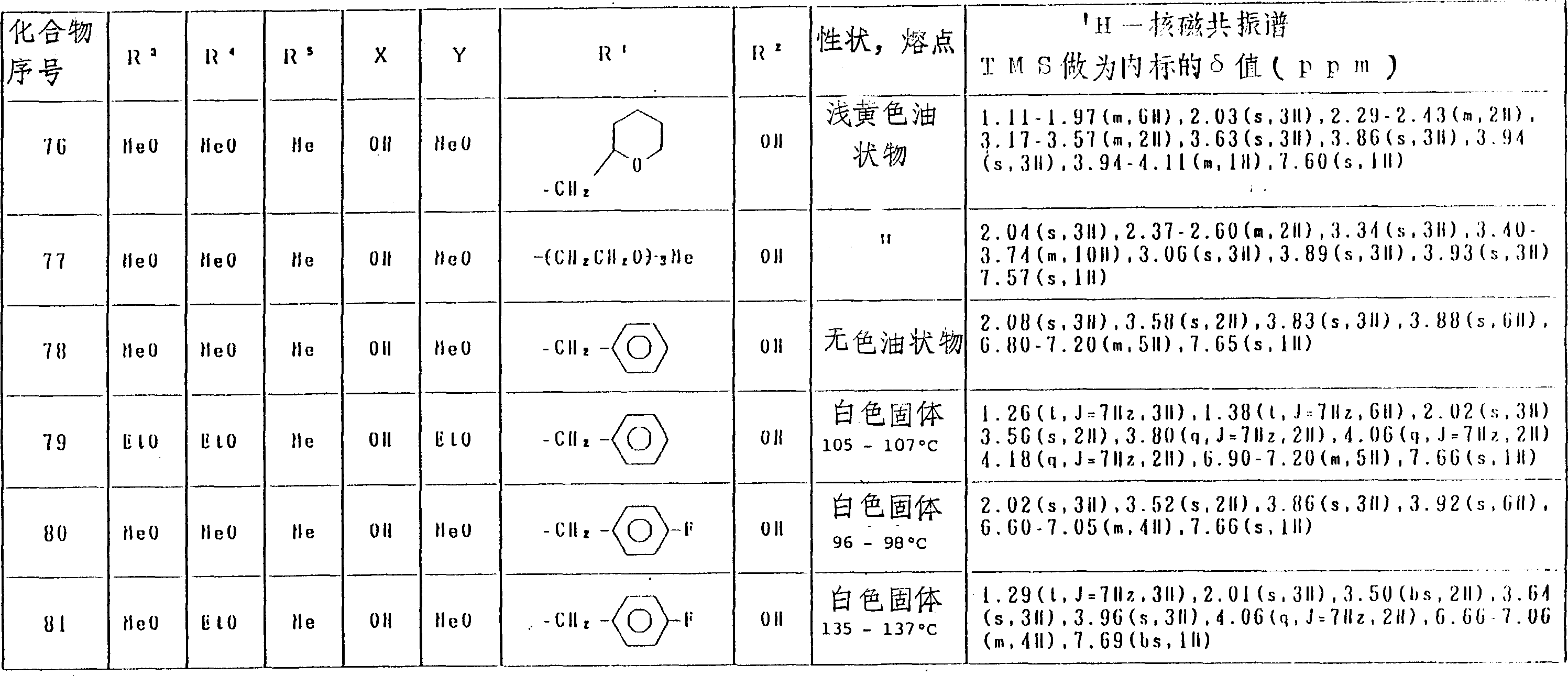
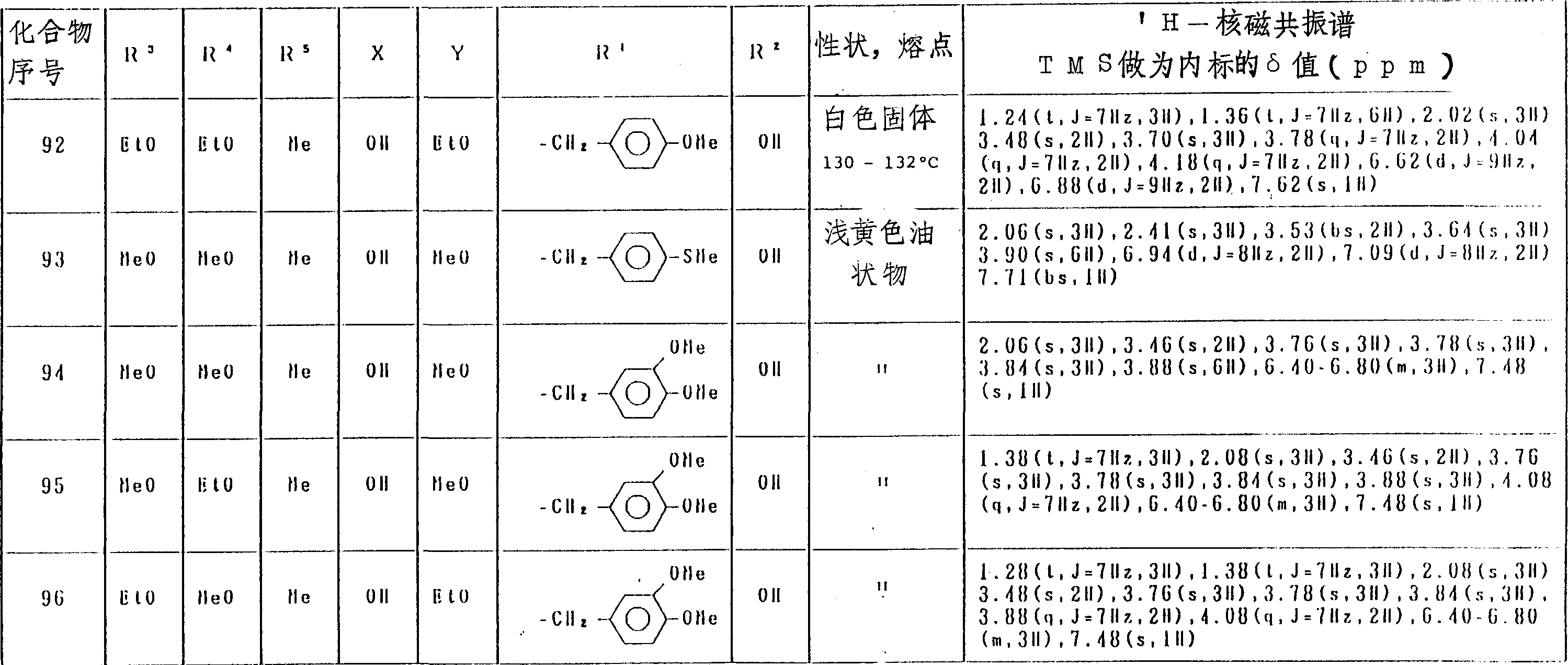
表5(第6部分)
表5(第10部分)
表5(第11部分)
表5(第17部分)
Claims (24)
其中R3,R4和R5可相同或不同,可以是氢原子,低级烷基,或低级烷氧基或是由下式所代表的基团,其中R3,R4和R5可相同或不同,可以是氢原子,低级烷基或低级烷氧基,X和Y可相同或不同,可以是羟基或由式(OCH2)nOR6,其中n为0或1,R6为低级烷基所代表的基团,或是酰基,
R1为含2—20个碳原子的烷基、环烷基、环烷基烷基、烯基、炔基、芳基烷基,由式(CH2)pCN,其中P为1—10的整数,所代表的基团,杂芳基烷基,由式(CH2)qB所代表的基团,其中q为1—6的整数,B为由下式所代表的基团:
其中r为0或1—2的整数,R7为低级烷基、环烷基或芳基,或是由式—O—R8所代表的基团,其中R8为低级烷基或芳基,或是由式(CH2—CH2—O)s,其中S为1—3的整数,所代表的基团;
R9和R10可相同或不同,可以是氢原子、低级烷基、羟基烷基、或杂芳基,R9和R10可相互连接并与氮原子键合成环,可以再含有一个氮原子和/或氧原子,还可以被取代,该方法包括:i)为制备A为下式所代表的基团,X,Y,R1,R2,R3,R4和R5 如上定义的式I化合物,将式III化合物与式(IV)化合物在碱存在下,于惰性有机溶剂中,于0—100℃下,反应;或ii)为制备A为下式所代表的基团,R1,R2,R3,R4和R5 如上定义的式I化合物,将式i)所制得的式I化合物,在惰性有机溶剂中,于0—80℃,用氧化剂氧化1—2小时;和
iii)如需要,将i)和ii)中所制得的式I化合物与无机酸或有机酸或氨基酸形成药用盐。
2.权利要求1 i)的方法,其中碱选自:碱金属氢化物及碱金属的醇化物。
3.权利要求2的方法,其中碱金属氢化物选自氢化钠、氢化钾、碱金属的醇化物选自甲醇钠、乙醇钠及叔丁醇钾。
4.权利要求1 i)的方法,其中惰性有机溶剂选自苯、甲苯、二氯甲烷、四氢呋喃、二恶烷、二甲氧基乙烷及二甲基甲酰胺。
5.权利要求1 ii)的方法,其中氧化剂可选自氯化铁六水合物或氧化铝,隋性有机溶剂选自苯、乙酸乙酯、二噁烷、乙醇和1,2—二甲氧基乙基,且均可含有水。
7.权利要求1 i)中的方法,其中A由下式表示:其中R3、R4和R5可相同或不同,可以是氢原子,低级烷基或低级烷氧基,X和Y可以相同或不同,可以是羟基,由式(OCH2)nOR6所代表的基团其中n为0或1,R6为低级烷基,或是酰基。
9.权利要求8的方法,其中R2为羟基。
10.权利要求1的方法,其中R1为含有2—12个碳原子的烷基,而R2为羟基。
11.权利要求1的方法,其中R1为含有7—12个碳原子的烷基,R2为羟基。
12.权利要求1的方法,其中R1为壬基。
13.权利要求1的方法,其中R1为壬基,R2为羟基。
14.权利要求1的方法,其中R1为3—甲基丁基。
15.权利要求1的方法,其中R1为环烷基。
16.权利要求1的方法,其中环烷基烷基为环己基甲基。
17.权利要求1的方法,其中R3,R4和R5各自独立地为低级烷基或低级烷氧基。
18.权利要求1的方法,其中R3和R4为低级烷氧基,R5为低级烷基。
19.权利要求1的方法,其中R3和R4为甲氧基,R5为甲基。
20.权利要求2中的方法,其中R3和R4为甲氧基,R5为壬基,R2为羟基。
21.权利要求1的方法,其中R3,R4和R5均为甲氧基,R2为羟基,R1为3—甲基丁基。
22.权利要求1的方法,其中R3为甲氧基,R4为乙氧基,R5为甲基,R1为3—甲基丁基,R2为羟基。
23.权利要求1的方法,其中R3为甲氧基,R4为乙氧基,R5为甲基,R1为环己基甲基,R2为羟基。
24.权利要求7的方法,其中R3和R4均为甲氧基,R1为壬基,R2为羟基。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP232761/89 | 1989-09-11 | ||
| JP23276189 | 1989-09-11 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1050182A CN1050182A (zh) | 1991-03-27 |
| CN1031992C true CN1031992C (zh) | 1996-06-12 |
Family
ID=16944337
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN90107622A Expired - Fee Related CN1031992C (zh) | 1989-09-11 | 1990-09-11 | 苯醌衍生物及其药用 |
Country Status (22)
| Country | Link |
|---|---|
| US (2) | US5210239A (zh) |
| EP (1) | EP0419905B1 (zh) |
| JP (1) | JP2919030B2 (zh) |
| KR (1) | KR920005815B1 (zh) |
| CN (1) | CN1031992C (zh) |
| AT (1) | ATE133156T1 (zh) |
| AU (1) | AU637138B2 (zh) |
| CA (1) | CA2024479C (zh) |
| DD (1) | DD299637A5 (zh) |
| DE (1) | DE69024896T2 (zh) |
| DK (1) | DK0419905T3 (zh) |
| ES (1) | ES2082811T3 (zh) |
| FI (1) | FI102273B (zh) |
| GR (1) | GR3019614T3 (zh) |
| HU (1) | HU208105B (zh) |
| IE (1) | IE903241A1 (zh) |
| MX (1) | MXPA95001266A (zh) |
| NO (1) | NO174292C (zh) |
| NZ (1) | NZ235193A (zh) |
| PT (1) | PT95266B (zh) |
| RU (2) | RU2001904C1 (zh) |
| ZA (1) | ZA907179B (zh) |
Families Citing this family (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IE920595A1 (en) * | 1991-03-11 | 1992-09-23 | Eisai Co Ltd | Quinone derivatives |
| WO1995012818A1 (en) * | 1993-11-04 | 1995-05-11 | Baxter Diagnostics Inc. | Tetrahydroxyquinone as an activator component for activated partial thromboplastine time test of blood coagulation and as a detector of blood coagulation disorders |
| IL111613A0 (en) * | 1993-11-12 | 1995-01-24 | Rhone Poulenc Rorer Ltd | Substituted phenyl compounds, their preparation and pharmaceutical compositions containing them |
| EP0801949A1 (en) * | 1996-04-15 | 1997-10-22 | Eisai Co., Ltd. | Hypoglycemic quinone derivative |
| EP0813866A3 (en) * | 1996-06-17 | 1999-01-20 | Eisai Co., Ltd. | Therapeutic agent for joint diseases |
| CA2368801A1 (en) * | 1999-04-30 | 2000-11-09 | Slil Biomedical Corporation | Conjugates as therapies for cancer and prostate diseases |
| US6649587B1 (en) | 1999-04-30 | 2003-11-18 | Slil Biomedical Corporation | Polyamine analog conjugates and quinone conjugates as therapies for cancers and prostate diseases |
| US6482943B1 (en) | 1999-04-30 | 2002-11-19 | Slil Biomedical Corporation | Quinones as disease therapies |
| IL155524A0 (en) | 2000-10-26 | 2003-11-23 | Fournier Lab Ireland Ltd | Combination of fenofibrate and coenzyme q10 for the treatment of endothelial dysfunction |
| EP1337504A4 (en) * | 2000-11-08 | 2005-10-05 | Cellgate Inc | NOVEL AMINO ACID CONJUGATES AND POLYAMINE ANALOGUES USEFUL AS ANTICANCING AGENTS |
| JP4230770B2 (ja) * | 2001-01-18 | 2009-02-25 | ウェリケム バイオテック インコーポレーテッド | 免疫性疾患を治療するための新規な1,2−ジフェニルエテン誘導体 |
| US8026280B2 (en) | 2001-03-27 | 2011-09-27 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors |
| US7842727B2 (en) * | 2001-03-27 | 2010-11-30 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors |
| US7312247B2 (en) * | 2001-03-27 | 2007-12-25 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors |
| US6495719B2 (en) | 2001-03-27 | 2002-12-17 | Circagen Pharmaceutical | Histone deacetylase inhibitors |
| CA2486303C (en) * | 2002-05-22 | 2013-04-30 | Errant Gene Therapeutics, Llc | Histone deacetylase inhibitors based on alpha-ketoepoxide compounds |
| CA2506504A1 (en) * | 2002-11-20 | 2004-06-03 | Errant Gene Therapeutics, Llc | Treatment of lung cells with histone deacetylase inhibitors |
| CA2583700A1 (en) | 2004-08-11 | 2006-02-23 | Arqule, Inc. | Quinone prodrug compositions and methods of use |
| US8614228B2 (en) | 2004-08-11 | 2013-12-24 | Arqule, Inc. | Quinone prodrug compositions and methods of use |
| WO2006052916A2 (en) * | 2004-11-08 | 2006-05-18 | Errant Gene Therapeutics, Inc. | Histone deacetylase inhibitors |
| DK2564843T3 (en) * | 2005-06-01 | 2019-03-11 | Bioelectron Tech Corp | Redox-active therapeutics for the treatment of mitochondrial diseases and other conditions as well as modulation of energy biomarkers |
| PL1933821T3 (pl) * | 2005-09-15 | 2021-01-11 | Ptc Therapeutics, Inc. | Warianty łańcucha końcowego środków terapeutycznych o aktywności redoks do leczenia chorób mitochondrialnych i innych stanów oraz do modulacji biomarkerów energetycznych |
| JP5374162B2 (ja) | 2006-02-22 | 2013-12-25 | エジソン ファーマシューティカルズ, インコーポレイテッド | ミトコンドリア病および他の症状の処置のためのレドックス活性化治療の側鎖変異体およびエネルギーバイオマーカーの調節 |
| TWI401081B (zh) * | 2007-04-30 | 2013-07-11 | Arqule Inc | 苯醌化合物的羥基磺酸鹽及其用途 |
| JP5646327B2 (ja) * | 2007-09-26 | 2014-12-24 | インディアナ・ユニバーシティ・リサーチ・アンド・テクノロジー・コーポレーション | 癌および血管新生の治療のための、化学療法剤と組み合わせたベンゾキノン誘導体e3330 |
| US11331294B2 (en) | 2007-09-26 | 2022-05-17 | Indiana University Research And Technology Corporation | Benzoquinone derivative E3330 in combination with chemotherapeutic agents for the treatment of bladder cancer |
| US8314153B2 (en) | 2008-09-10 | 2012-11-20 | Edison Pharmaceuticals, Inc. | Treatment of pervasive developmental disorders with redox-active therapeutics |
| EP3520860A1 (en) * | 2011-05-26 | 2019-08-07 | Indiana University Research & Technology Corporation | Quinone compounds for treating ape1 mediated diseases |
| US20140128398A1 (en) * | 2011-06-03 | 2014-05-08 | Indiana University Research And Technology Corporation | Compounds, compositions and methods for treating oxidative dna damage disorders |
| DK2913321T3 (da) * | 2014-02-27 | 2021-10-25 | Emerald Health Pharmaceuticals Inc | Hidtil ukendte cannabigerolderivater |
| RU2017144823A (ru) | 2015-05-21 | 2019-06-24 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Способы нацеливания ape1/ref-1 для ингибирования генов трансдукции сигналов гипоксии |
| CN108712903A (zh) | 2015-12-17 | 2018-10-26 | 生物电子技术有限公司 | 用于治疗氧化应急障碍的氟烷基、氟代烷氧基、苯氧基、杂芳氧基、烷氧基和胺1,4-苯醌衍生物 |
| WO2019157163A1 (en) | 2018-02-08 | 2019-08-15 | Indiana University Research And Technology Corporation | Targeting ocular diseases with novel ape1/ref-1 inhibitors |
| KR102013574B1 (ko) * | 2018-10-25 | 2019-08-23 | 주식회사 바이오톡스텍 | 하이드로퀴논 유도체를 포함하는 비만 또는 비알콜성 지방간염의 예방 또는 치료용 약학 조성물 |
| CN112694393B (zh) * | 2020-12-25 | 2022-06-17 | 浙江神洲药业有限公司 | 一种再生四氯苯醌的制备方法 |
| KR102678127B1 (ko) * | 2021-07-22 | 2024-06-25 | 경희대학교 산학협력단 | 신규한 퀴논 유도체 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS58177934A (ja) * | 1982-04-13 | 1983-10-18 | Takeda Chem Ind Ltd | ベンゾキノン誘導体 |
| US4808339A (en) * | 1982-04-13 | 1989-02-28 | Takeda Chemical Industries, Ltd. | Benzoquinone derivatives |
| JP2832979B2 (ja) * | 1988-02-15 | 1998-12-09 | 武田薬品工業株式会社 | 不飽和カルボン酸アミド誘導体 |
| US5286750A (en) * | 1989-10-11 | 1994-02-15 | Basf Aktiengesellschaft | Phenylacetic acid derivatives and fungicides containing them |
-
1990
- 1990-08-24 FI FI904206A patent/FI102273B/fi not_active IP Right Cessation
- 1990-08-31 US US07/576,054 patent/US5210239A/en not_active Expired - Lifetime
- 1990-08-31 CA CA002024479A patent/CA2024479C/en not_active Expired - Fee Related
- 1990-09-05 ES ES90117119T patent/ES2082811T3/es not_active Expired - Lifetime
- 1990-09-05 DE DE69024896T patent/DE69024896T2/de not_active Expired - Fee Related
- 1990-09-05 AT AT90117119T patent/ATE133156T1/de not_active IP Right Cessation
- 1990-09-05 DK DK90117119.9T patent/DK0419905T3/da active
- 1990-09-05 EP EP90117119A patent/EP0419905B1/en not_active Expired - Lifetime
- 1990-09-06 IE IE324190A patent/IE903241A1/en unknown
- 1990-09-06 AU AU62258/90A patent/AU637138B2/en not_active Ceased
- 1990-09-06 NZ NZ235193A patent/NZ235193A/en unknown
- 1990-09-07 NO NO903909A patent/NO174292C/no unknown
- 1990-09-10 HU HU905847A patent/HU208105B/hu not_active IP Right Cessation
- 1990-09-10 ZA ZA907179A patent/ZA907179B/xx unknown
- 1990-09-10 RU SU904831237A patent/RU2001904C1/ru active
- 1990-09-10 DD DD90343921A patent/DD299637A5/de not_active IP Right Cessation
- 1990-09-10 PT PT95266A patent/PT95266B/pt not_active IP Right Cessation
- 1990-09-10 JP JP2237051A patent/JP2919030B2/ja not_active Expired - Lifetime
- 1990-09-11 KR KR1019900014310A patent/KR920005815B1/ko not_active IP Right Cessation
- 1990-09-11 CN CN90107622A patent/CN1031992C/zh not_active Expired - Fee Related
-
1992
- 1992-08-20 RU SU925052502A patent/RU2049771C1/ru active
-
1993
- 1993-03-01 US US08/022,688 patent/US5385942A/en not_active Expired - Lifetime
-
1995
- 1995-03-09 MX MXPA95001266A patent/MXPA95001266A/es unknown
-
1996
- 1996-04-09 GR GR960401009T patent/GR3019614T3/el unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1031992C (zh) | 苯醌衍生物及其药用 | |
| CN1173945C (zh) | 5-硫代-ω-取代的苯基-前列腺素E衍生物、其制备方法及含有它们作为活性成分的药物 | |
| CN1032203C (zh) | 制备取代2-吡啶酮和吡啶-2-硫酮的方法 | |
| CN1027068C (zh) | α-取代的4-(喹啉-2-基-甲氧基)苯乙酸和酯的制备方法 | |
| CN1146536C (zh) | 新的环己烷衍生物 | |
| CN1030757A (zh) | 苯并噻唑衍生物 | |
| CN85108888A (zh) | 环氧丙烷酮类化合物的制备方法 | |
| CN1030582A (zh) | 苯并吡喃衍生物 | |
| CN1173497A (zh) | 含杂环碳酸衍生物 | |
| CN1014409B (zh) | 生产抗坏血酸衍生物的方法 | |
| CN1077954A (zh) | 新的苯并吡喃衍生物 | |
| CN1198827C (zh) | 精选的K-252a衍生物 | |
| CN1049500A (zh) | 苯并吡喃化合物,其生产方法及药物组合物 | |
| CN1016507B (zh) | 新的福斯克林衍生物的制备方法 | |
| CN1140167A (zh) | 取代的2-氨基四氢萘和3-氨基苯并二氢吡喃的制法 | |
| CN85109587A (zh) | 具有抗过敏活性的新的苯并噻吩类和苯并呋喃类化合物的制备方法 | |
| CN1049499A (zh) | 具有粘液调节和抗局部缺血性质的2,3-二氢-5-氧-4,6,7-三甲基苯并呋喃的2-(rs)-衍生物及其作为抗氧化药剂的用途 | |
| CN1045972A (zh) | 5-取代-1,4-二氢-4-氧代-二氮杂萘-3-羧酸(酯)抗菌药 | |
| CN1653081A (zh) | 甾族化合物的c-17螺甾内酯化和6,7氧化 | |
| CN1079745A (zh) | 新的9-氟-7-氧代-7H-吡啶并[1,2,3-d,e][1,4]苯并嗪-6-羧酸及其酯 | |
| CN1058494C (zh) | 新的洋芫荽黄素化合物,其制备方法和含它们的药用组合物 | |
| CN88101674A (zh) | 色酮衍生物 | |
| CN1092647C (zh) | 新抗病毒的取代嘧啶二酮同素碳环核苷衍生物及其制备方法与含有其为活性成分的组合物 | |
| CN1309655A (zh) | 可用于治疗脂血异常、动脉粥样硬化和糖尿病的环状化合物、药物组合物和制备方法 | |
| CN1805947A (zh) | 合成2-羟基-n , n-二甲基-3-[[2-[[1 ( r )-(5-甲基-2-呋喃基)丙基]氨基]-3 , 4-二氧代-1-环丁烯-1-基]氨基] 苯甲酰胺 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |