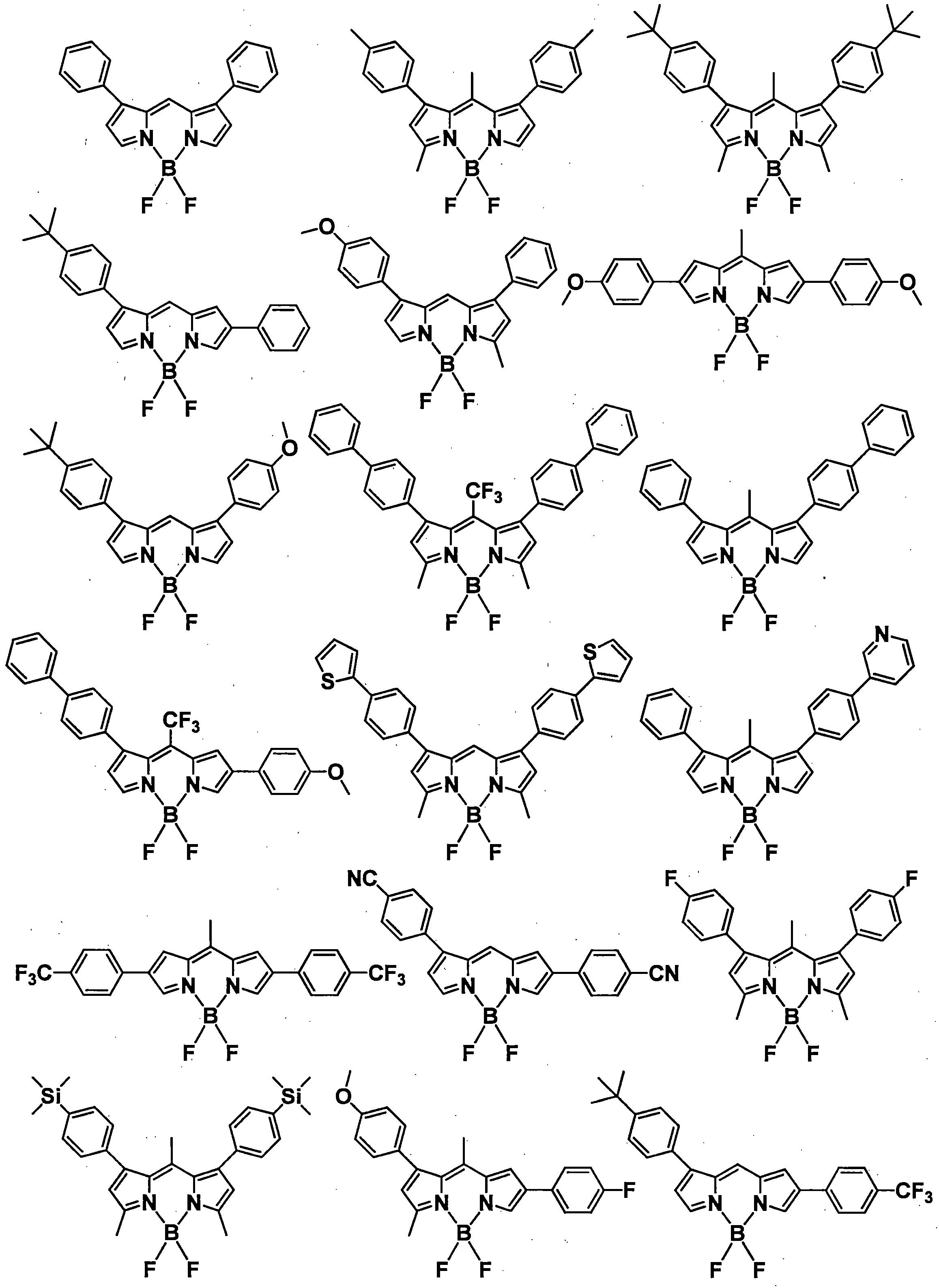WO2017014068A1 - 色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 - Google Patents
色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 Download PDFInfo
- Publication number
- WO2017014068A1 WO2017014068A1 PCT/JP2016/070255 JP2016070255W WO2017014068A1 WO 2017014068 A1 WO2017014068 A1 WO 2017014068A1 JP 2016070255 W JP2016070255 W JP 2016070255W WO 2017014068 A1 WO2017014068 A1 WO 2017014068A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- color conversion
- light
- conversion composition
- substituted
- Prior art date
Links
- 0 C*S(CCCCN(C(C1(C)C)=CC=CC=CC=CC(C2(C)C)N(CCCCS(*)(=O)=O)c3c2c(cccc2)c2cc3)c2c1c(cccc1)c1cc2)(=O)=O Chemical compound C*S(CCCCN(C(C1(C)C)=CC=CC=CC=CC(C2(C)C)N(CCCCS(*)(=O)=O)c3c2c(cccc2)c2cc3)c2c1c(cccc1)c1cc2)(=O)=O 0.000 description 4
- AYQRFGNDSMYZEG-UHFFFAOYSA-N CC(C)COC(c(cc1)c(c(C#N)ccc2-c(cc3)c4c5c3C(OCC(C)C)=O)c2c1-c4ccc5C#N)=O Chemical compound CC(C)COC(c(cc1)c(c(C#N)ccc2-c(cc3)c4c5c3C(OCC(C)C)=O)c2c1-c4ccc5C#N)=O AYQRFGNDSMYZEG-UHFFFAOYSA-N 0.000 description 1
- QJUXUCPDUMQLMG-UHFFFAOYSA-N CC(C)c(cccc1C(C)C)c1N(C(c(c1c2cc3Oc4ccccc4)cc(Oc4ccccc4)c-4c1c3-c(c(Oc(cc1)ccc1-c1ccc3[s]c(-c(cc(cc5)N(c6ccccc6)c6ccccc6)c5O)nc3c1)cc(C(N(c1c(C(C)C)cccc1C(C)C)C1=O)=O)c3c1c1)c3c-4c1Oc1ccccc1)=O)C2=O Chemical compound CC(C)c(cccc1C(C)C)c1N(C(c(c1c2cc3Oc4ccccc4)cc(Oc4ccccc4)c-4c1c3-c(c(Oc(cc1)ccc1-c1ccc3[s]c(-c(cc(cc5)N(c6ccccc6)c6ccccc6)c5O)nc3c1)cc(C(N(c1c(C(C)C)cccc1C(C)C)C1=O)=O)c3c1c1)c3c-4c1Oc1ccccc1)=O)C2=O QJUXUCPDUMQLMG-UHFFFAOYSA-N 0.000 description 1
- PHWPJDAIHBKHLY-VFIVCBTMSA-N CCCCOc1ccc(/C=C(/c(cc2)ccc2/C(/C#N)=C/c(cc2)ccc2OCCCC)\C#N)cc1 Chemical compound CCCCOc1ccc(/C=C(/c(cc2)ccc2/C(/C#N)=C/c(cc2)ccc2OCCCC)\C#N)cc1 PHWPJDAIHBKHLY-VFIVCBTMSA-N 0.000 description 1
- VOFUROIFQGPCGE-UHFFFAOYSA-N CCN(CC)c(cc1O2)ccc1N=C(c1c3cccc1)C2=CC3=O Chemical compound CCN(CC)c(cc1O2)ccc1N=C(c1c3cccc1)C2=CC3=O VOFUROIFQGPCGE-UHFFFAOYSA-N 0.000 description 1
- VBVAVBCYMYWNOU-UHFFFAOYSA-N CCN(CC)c1ccc(C=C(c2nc(cccc3)c3[s]2)C(O2)=O)c2c1 Chemical compound CCN(CC)c1ccc(C=C(c2nc(cccc3)c3[s]2)C(O2)=O)c2c1 VBVAVBCYMYWNOU-UHFFFAOYSA-N 0.000 description 1
- GOLORTLGFDVFDW-UHFFFAOYSA-N CCN(CC)c1ccc(C=C(c2nc3ccccc3[nH]2)C(O2)=O)c2c1 Chemical compound CCN(CC)c1ccc(C=C(c2nc3ccccc3[nH]2)C(O2)=O)c2c1 GOLORTLGFDVFDW-UHFFFAOYSA-N 0.000 description 1
- VCBDMJCBSFUKNZ-UHFFFAOYSA-N COc(cc1)cc(-c2nc3c(cccc4)c4c(cccc4)c4c3[n]2-c2ccccc2)c1O Chemical compound COc(cc1)cc(-c2nc3c(cccc4)c4c(cccc4)c4c3[n]2-c2ccccc2)c1O VCBDMJCBSFUKNZ-UHFFFAOYSA-N 0.000 description 1
- NPRFXRHWTRERQD-UHFFFAOYSA-N C[Si](C(c1ccccc1)=C1c2ccccc2)(C(c2ccccc2)=C1c1ccccc1)c1ccccc1 Chemical compound C[Si](C(c1ccccc1)=C1c2ccccc2)(C(c2ccccc2)=C1c1ccccc1)c1ccccc1 NPRFXRHWTRERQD-UHFFFAOYSA-N 0.000 description 1
- IRZWHILMNSFEAF-UHFFFAOYSA-N OC(c1cc(N=C=S)ccc1C(c(c(O1)c2)ccc2O)=C(C=C2)C1=CC2=O)=O Chemical compound OC(c1cc(N=C=S)ccc1C(c(c(O1)c2)ccc2O)=C(C=C2)C1=CC2=O)=O IRZWHILMNSFEAF-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/63—Additives non-macromolecular organic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/05—Alcohols; Metal alcoholates
- C08K5/053—Polyhydroxylic alcohols
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/09—Carboxylic acids; Metal salts thereof; Anhydrides thereof
- C08K5/098—Metal salts of carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/17—Amines; Quaternary ammonium compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/55—Boron-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/56—Organo-metallic compounds, i.e. organic compounds containing a metal-to-carbon bond
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
- C09D5/22—Luminous paints
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/02—Use of particular materials as binders, particle coatings or suspension media therefor
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F21—LIGHTING
- F21V—FUNCTIONAL FEATURES OR DETAILS OF LIGHTING DEVICES OR SYSTEMS THEREOF; STRUCTURAL COMBINATIONS OF LIGHTING DEVICES WITH OTHER ARTICLES, NOT OTHERWISE PROVIDED FOR
- F21V9/00—Elements for modifying spectral properties, polarisation or intensity of the light emitted, e.g. filters
- F21V9/30—Elements containing photoluminescent material distinct from or spaced from the light source
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L33/00—Semiconductor devices with at least one potential-jump barrier or surface barrier specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof
- H01L33/48—Semiconductor devices with at least one potential-jump barrier or surface barrier specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof characterised by the semiconductor body packages
- H01L33/50—Wavelength conversion elements
- H01L33/501—Wavelength conversion elements characterised by the materials, e.g. binder
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L33/00—Semiconductor devices with at least one potential-jump barrier or surface barrier specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof
- H01L33/48—Semiconductor devices with at least one potential-jump barrier or surface barrier specially adapted for light emission; Processes or apparatus specially adapted for the manufacture or treatment thereof or of parts thereof; Details thereof characterised by the semiconductor body packages
- H01L33/50—Wavelength conversion elements
- H01L33/501—Wavelength conversion elements characterised by the materials, e.g. binder
- H01L33/502—Wavelength conversion materials
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1022—Heterocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
- C09K2211/187—Metal complexes of the iron group metals, i.e. Fe, Co or Ni
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F21—LIGHTING
- F21Y—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES F21K, F21L, F21S and F21V, RELATING TO THE FORM OR THE KIND OF THE LIGHT SOURCES OR OF THE COLOUR OF THE LIGHT EMITTED
- F21Y2115/00—Light-generating elements of semiconductor light sources
- F21Y2115/10—Light-emitting diodes [LED]
Definitions
- the present invention relates to a color conversion composition, a color conversion film, a backlight unit including the same, a display, and illumination.
- the color conversion is to convert light emitted from the light emitter into light having a longer wavelength, for example, to convert blue light emission into green or red light emission.
- a white light source combining such a blue light source and a film having a color conversion function is used as a backlight unit, and a liquid crystal driving portion and a color filter are combined, whereby a full color display can be manufactured. If there is no liquid crystal driving part, it can be used as a white light source as it is, and can be applied as a white light source such as LED lighting.
- An improvement in color reproducibility is an issue for liquid crystal displays that use color conversion methods.
- it is effective to increase the color purity of each color of blue, green, and red by narrowing the half width of each emission spectrum of blue, green, and red of the backlight unit.
- a technique using quantum dots made of inorganic semiconductor fine particles as a component of a color conversion composition has been proposed (for example, see Patent Document 1).
- the technology using quantum dots certainly has a narrow half-value width of the emission spectrum of green and red and improves color reproducibility.
- quantum dots are weak against heat, moisture and oxygen in the air, and have sufficient durability. There wasn't. There are also problems such as the inclusion of cadmium.
- a technique that uses an organic light-emitting material as a component of the color conversion composition instead of quantum dots has also been proposed.
- techniques using an organic light emitting material as a component of the color conversion composition include those using a pyridine-phthalimide condensate (for example, see Patent Document 2), and those using a coumarin derivative (for example, see Patent Document 3).
- the red light emitting material those using a perylene derivative so far (for example, see Patent Document 4), those using a rhodamine derivative (for example, see Patent Document 5), those using a pyromethene derivative (for example, Patent Document 6) To 7).
- the problem to be solved by the present invention is to achieve both improvement in color reproducibility and durability in a color conversion composition used for liquid crystal displays and LED lighting, and in particular, achieves both high color purity emission and durability. It is to let you.
- the present invention is a color conversion composition for converting incident light into light having a longer wavelength than the incident light, and the following components (A) to (C): (A) at least one light emitting material (B) binder resin (C) containing at least one of a tertiary amine, a catechol derivative and a nickel compound,
- the tertiary amine, catechol derivative and nickel compound are color conversion compositions having a molar extinction coefficient ⁇ of 100 or less over the entire wavelength range of from 400 nm to 800 nm.
- the color conversion composition of the present invention and the color conversion film using the same have both high color purity and durability, it is possible to achieve both color reproducibility and durability.
- the schematic cross section which shows an example of the color conversion film of this invention The schematic cross section which shows an example of the color conversion film of this invention.
- the absorption spectrum of the compound of Synthesis Example 1. The emission spectrum of the compound of Synthesis Example 1.
- the color conversion composition of the present invention contains at least one luminescent material.
- the light-emitting material in the present invention refers to a material that emits light having a wavelength different from that of light when irradiated with some light.
- a material exhibiting light emission characteristics with a high quantum yield is preferable.
- Known light-emitting materials such as inorganic phosphors, fluorescent pigments, fluorescent dyes, quantum dots and the like can be mentioned, and among these, organic light-emitting materials are preferable.
- organic light emitting material for example, Compounds having a condensed aryl ring such as naphthalene, anthracene, phenanthrene, pyrene, chrysene, naphthacene, triphenylene, perylene, fluoranthene, fluorene, indene and derivatives thereof; Furan, pyrrole, thiophene, silole, 9-silafluorene, 9,9'-spirobisilafluorene, benzothiophene, benzofuran, indole, dibenzothiophene, dibenzofuran, imidazopyridine, phenanthroline, pyridine, pyrazine, naphthyridine, quinoxaline, pyrrolopyridine
- a compound having a heteroaryl ring such as Borane derivatives; 1,4-distyrylbenzene, 4,4′-bis (2- (4-diphenylaminophenyl
- the organic light emitting material may be a fluorescent light emitting material or a phosphorescent light emitting material, but a fluorescent light emitting material is preferable in order to achieve high color purity.
- a compound having a coordination bond is preferable.
- a compound containing boron such as a boron fluoride complex is also preferable in that the half width is small and highly efficient light emission is possible.
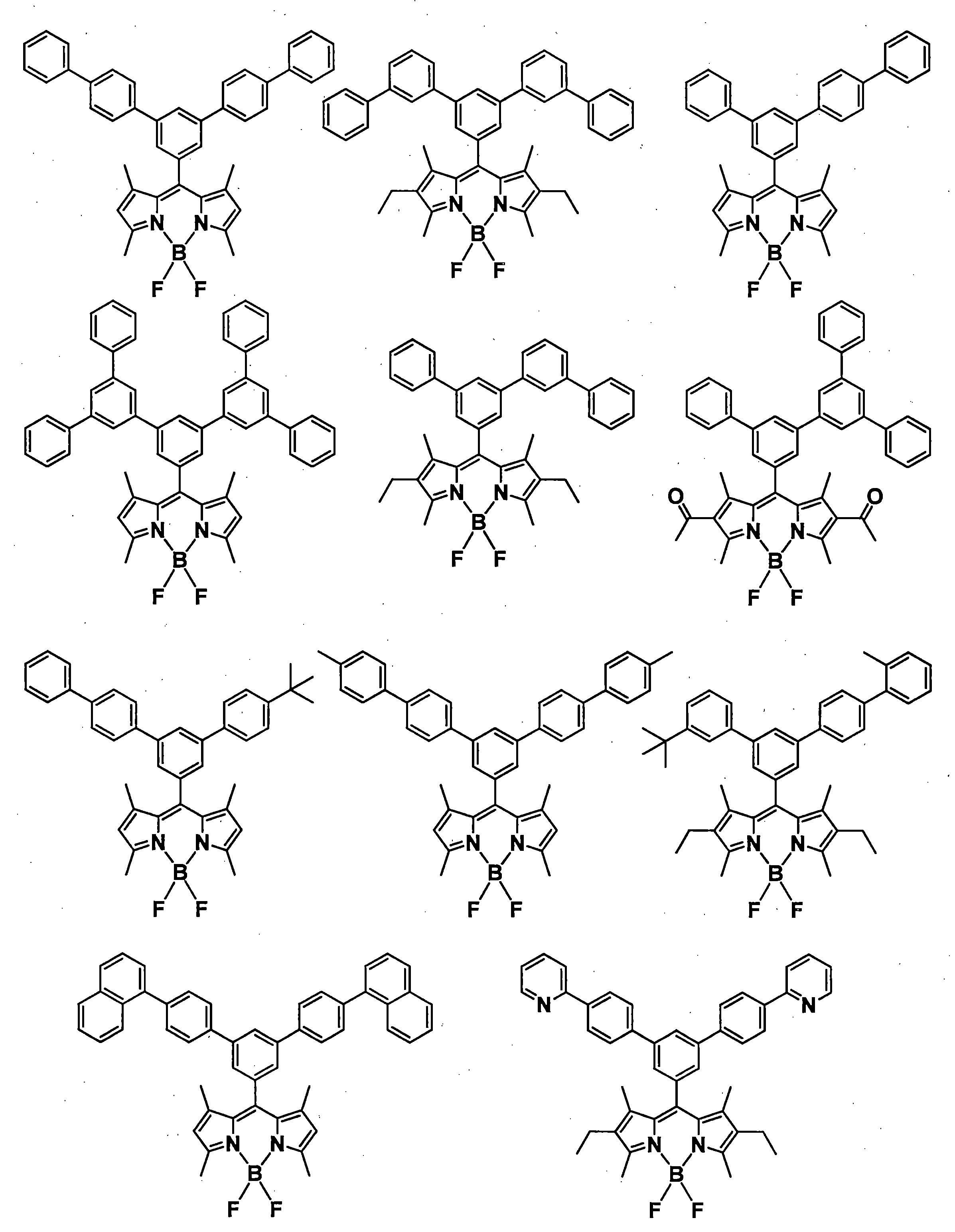
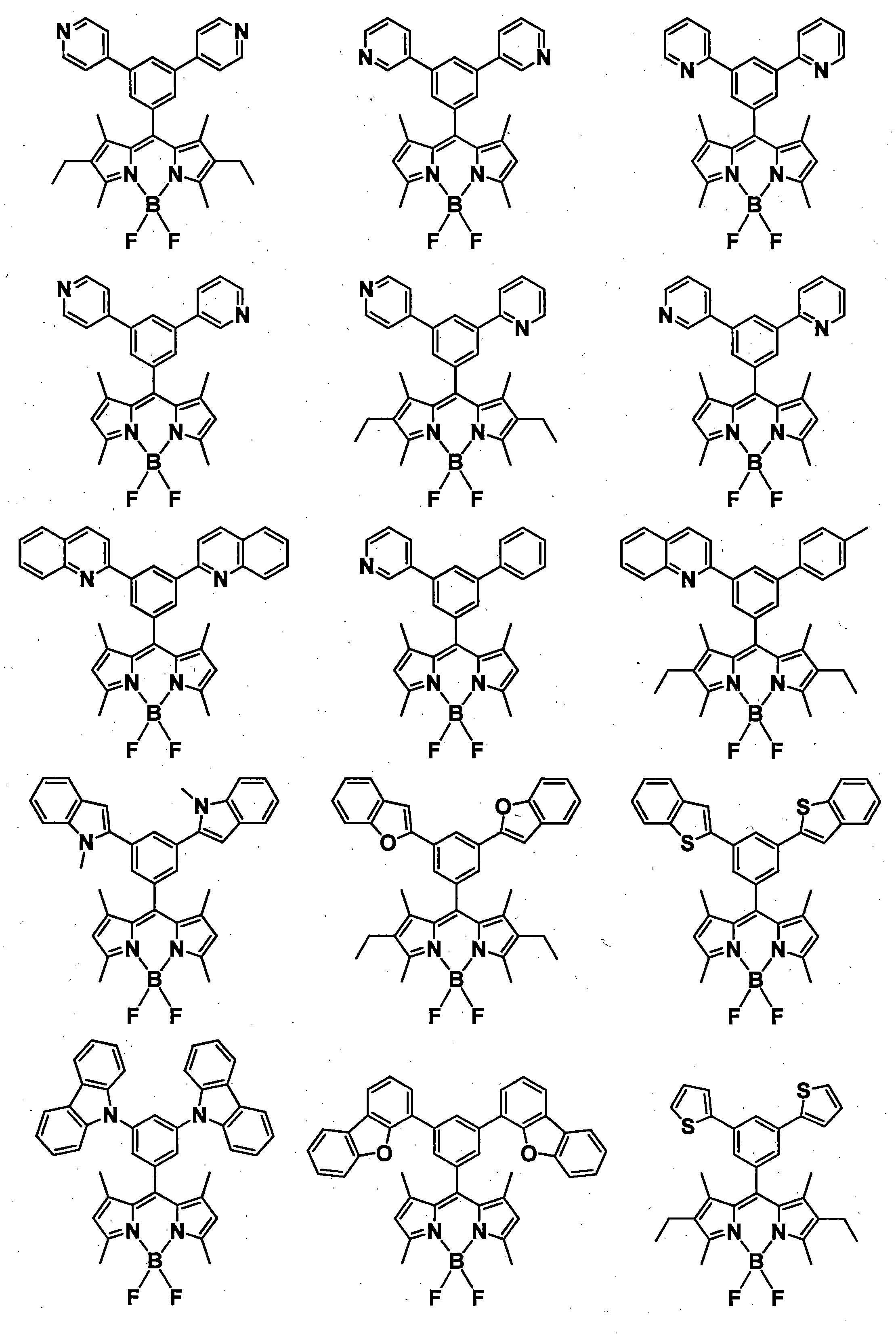
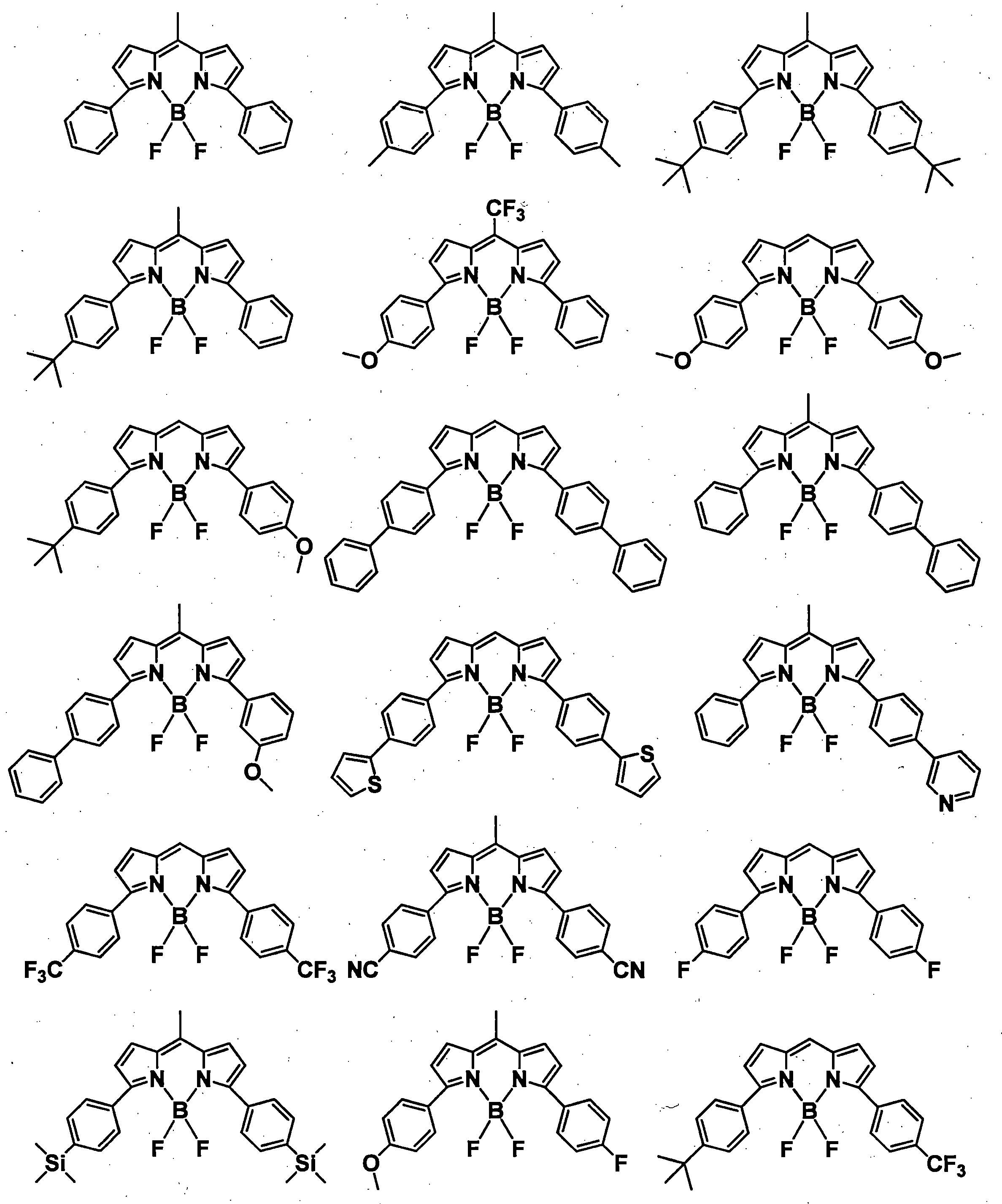
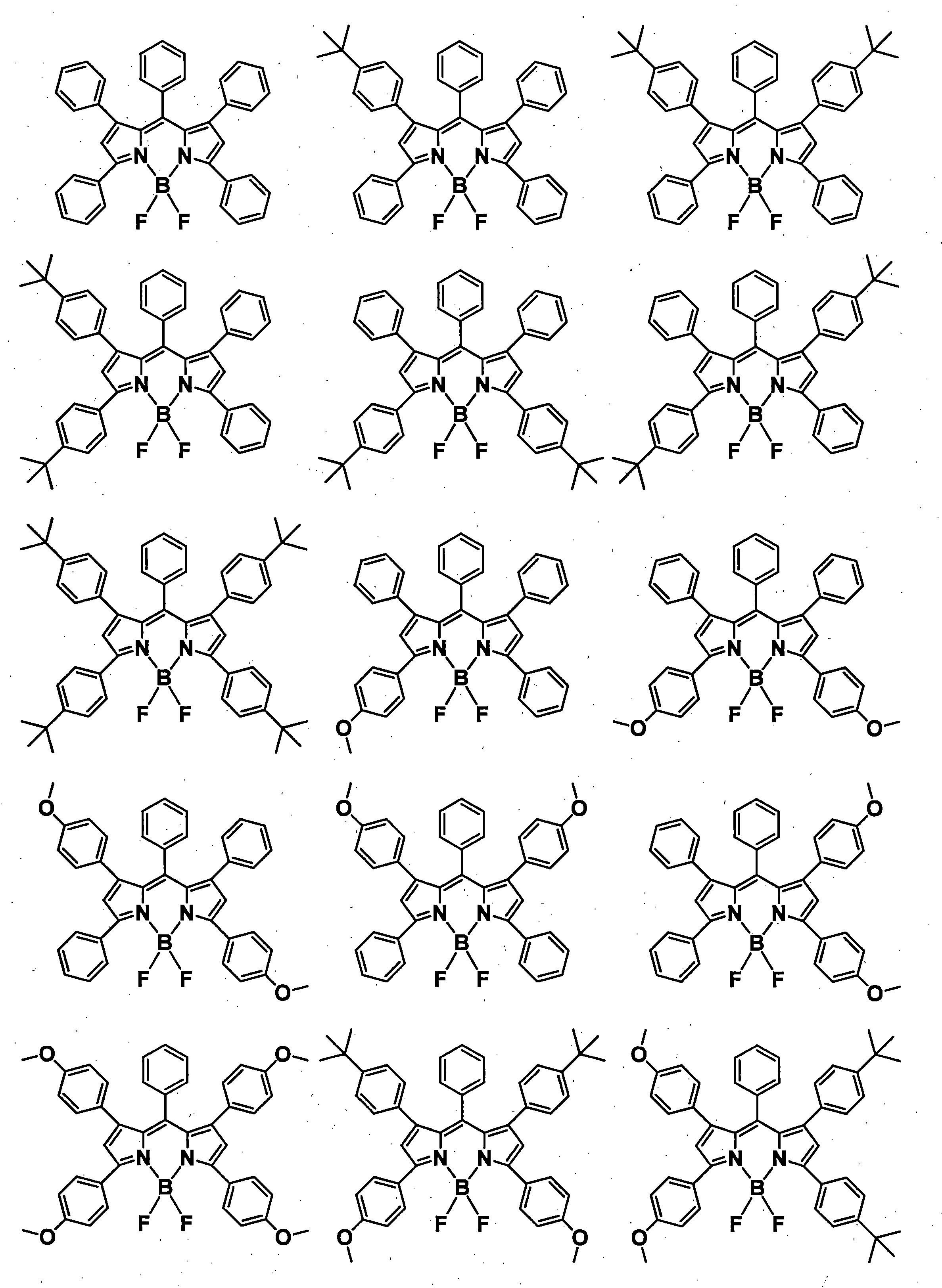
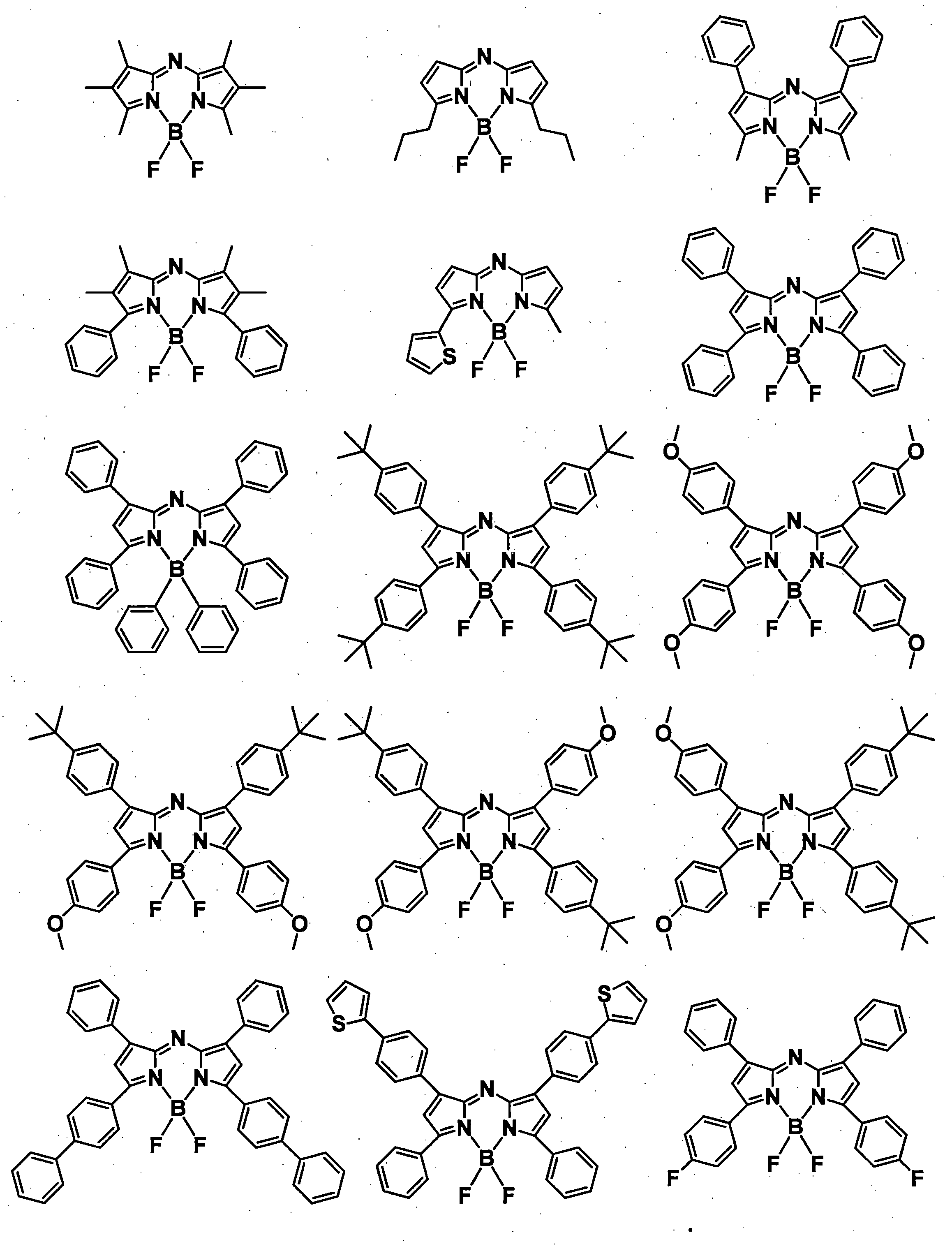
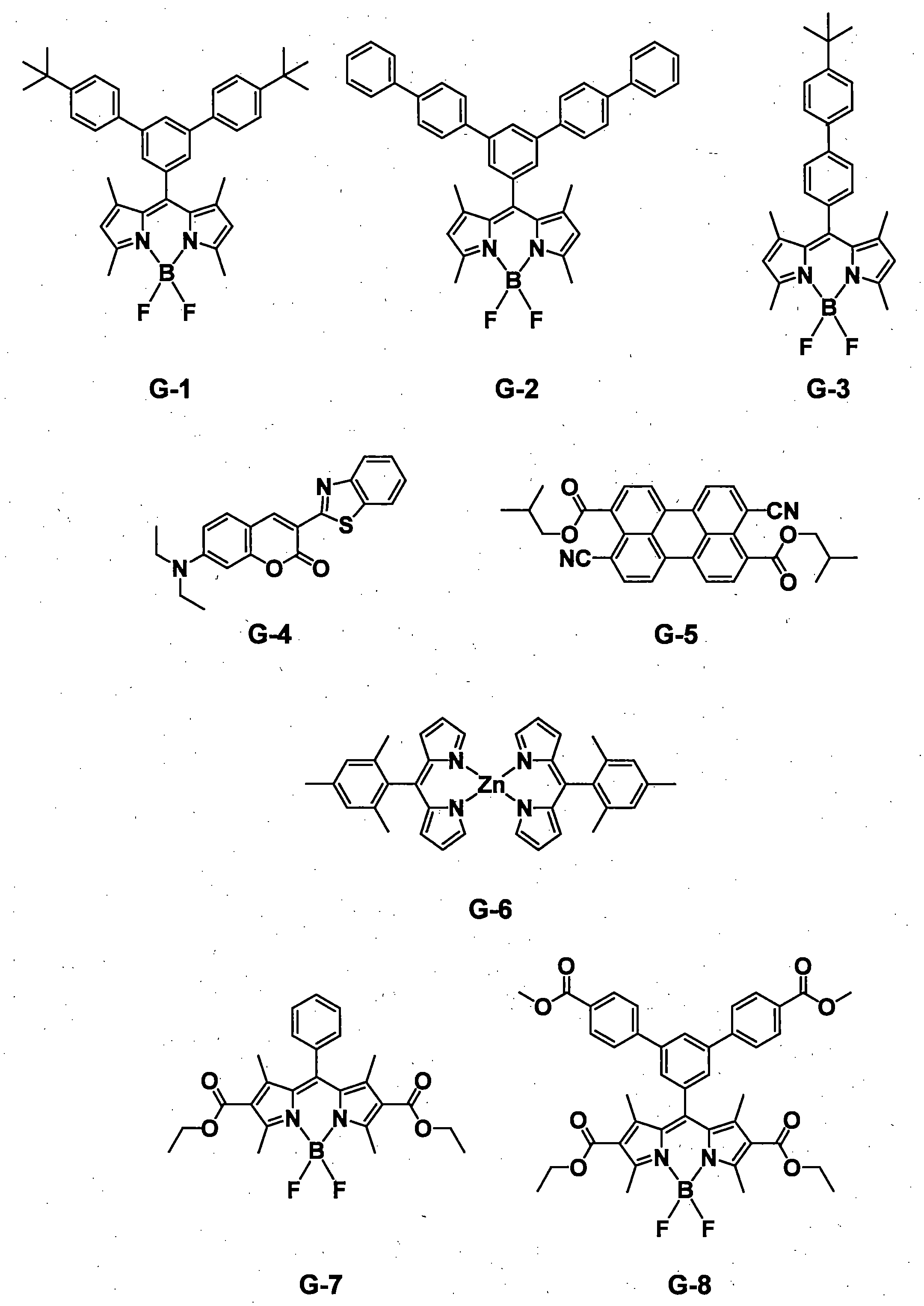
- a pyromethene derivative can be preferably used because it gives a high fluorescence quantum yield and has good durability. More preferably, it is a compound represented by General formula (1).
- R 1 to R 9 may be the same as or different from each other, and may be hydrogen, alkyl group, cycloalkyl group, heterocyclic group, alkenyl group, cycloalkenyl group, alkynyl group, hydroxyl group, thiol group, alkoxy group, alkylthio group, aryl Ether group, arylthioether group, aryl group, heteroaryl group, halogen, cyano group, aldehyde group, carbonyl group, carboxyl group, oxycarbonyl group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group, boryl group, It is selected from a condensed ring and an aliphatic ring formed between a phosphine oxide group and an adjacent substituent.
- hydrogen may be deuterium.
- a substituted or unsubstituted aryl group having 6 to 40 carbon atoms is 6 to 40 carbon atoms including the number of carbon atoms contained in the substituent group substituted on the aryl group. The same applies to the other substituents.
- the substituents in the case of substitution include alkyl groups, cycloalkyl groups, heterocyclic groups, alkenyl groups, cycloalkenyl groups, alkynyl groups, hydroxyl groups, thiol groups, alkoxy groups, alkylthio groups.
- Aryl ether group, aryl thioether group, aryl group, heteroaryl group, halogen, cyano group, aldehyde group, carbonyl group, carboxyl group, oxycarbonyl group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group, boryl Group and a phosphine oxide group are preferable, and specific substituents that are preferable in the description of each substituent are preferable. Moreover, these substituents may be further substituted with the above-mentioned substituents.
- unsubstituted means that a hydrogen atom or a deuterium atom is substituted.
- the alkyl group represents, for example, a saturated aliphatic hydrocarbon group such as a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, a sec-butyl group, or a tert-butyl group, which is a substituent. It may or may not have. There are no particular limitations on the additional substituent when it is substituted, and examples thereof include an alkyl group, a halogen, an aryl group, a heteroaryl group, and the like, and this point is common to the following description.
- the number of carbon atoms of the alkyl group is not particularly limited, but is preferably 1 or more and 20 or less, more preferably 1 or more and 8 or less, from the viewpoint of availability and cost.
- the cycloalkyl group refers to, for example, a saturated alicyclic hydrocarbon group such as a cyclopropyl group, a cyclohexyl group, a norbornyl group, an adamantyl group, which may or may not have a substituent.
- the number of carbon atoms in the alkyl group moiety is not particularly limited, but is preferably in the range of 3 or more and 20 or less.
- the heterocyclic group refers to an aliphatic ring having atoms other than carbon, such as a pyran ring, a piperidine ring, and a cyclic amide, in the ring, which may or may not have a substituent. .
- carbon number of a heterocyclic group is not specifically limited, Preferably it is the range of 2-20.
- alkenyl group refers to an unsaturated aliphatic hydrocarbon group containing a double bond such as a vinyl group, an allyl group, or a butadienyl group, which may or may not have a substituent.
- carbon number of an alkenyl group is not specifically limited, Preferably it is the range of 2-20.
- the cycloalkenyl group refers to an unsaturated alicyclic hydrocarbon group containing a double bond such as a cyclopentenyl group, a cyclopentadienyl group, or a cyclohexenyl group, which may have a substituent. You don't have to.
- the alkynyl group indicates, for example, an unsaturated aliphatic hydrocarbon group containing a triple bond such as an ethynyl group, which may or may not have a substituent.
- the number of carbon atoms of the alkynyl group is not particularly limited, but is preferably in the range of 2 or more and 20 or less.
- the alkoxy group refers to, for example, a functional group having an aliphatic hydrocarbon group bonded through an ether bond such as a methoxy group, an ethoxy group, or a propoxy group, and the aliphatic hydrocarbon group may have a substituent. It may not have.
- carbon number of an alkoxy group is not specifically limited, Preferably it is the range of 1-20.
- the alkylthio group is a group in which an oxygen atom of an ether bond of an alkoxy group is substituted with a sulfur atom.
- the hydrocarbon group of the alkylthio group may or may not have a substituent. Although carbon number of an alkylthio group is not specifically limited, Preferably it is the range of 1-20.
- An aryl ether group refers to a functional group to which an aromatic hydrocarbon group is bonded via an ether bond, such as a phenoxy group, and the aromatic hydrocarbon group may or may not have a substituent. Good. Although carbon number of an aryl ether group is not specifically limited, Preferably, it is the range of 6-40.
- the aryl thioether group is a group in which an oxygen atom of an ether bond of an aryl ether group is substituted with a sulfur atom.
- the aromatic hydrocarbon group in the aryl ether group may or may not have a substituent. Although carbon number of an aryl ether group is not specifically limited, Preferably, it is the range of 6-40.
- the aryl group is, for example, phenyl group, biphenyl group, terphenyl group, naphthyl group, fluorenyl group, benzofluorenyl group, dibenzofluorenyl group, phenanthryl group, anthracenyl group, benzophenanthryl group, benzoanthracene group.
- An aromatic hydrocarbon group such as a nyl group, a chrycenyl group, a pyrenyl group, a fluoranthenyl group, a triphenylenyl group, a benzofluoranthenyl group, a dibenzoanthracenyl group, a perylenyl group, or a helicenyl group.
- a phenyl group, a biphenyl group, a terphenyl group, a naphthyl group, a fluorenyl group, a phenanthryl group, an anthracenyl group, a pyrenyl group, a fluoranthenyl group, and a triphenylenyl group are preferable.
- the aryl group may or may not have a substituent. Although carbon number of an aryl group is not specifically limited, Preferably it is 6-40, More preferably, it is the range of 6-30.
- the aryl group is preferably a phenyl group, a biphenyl group, a terphenyl group, a naphthyl group, a fluorenyl group, a phenanthryl group, or an anthracenyl group, and a phenyl group, a biphenyl group, A terphenyl group and a naphthyl group are more preferable. More preferred are a phenyl group, a biphenyl group, and a terphenyl group, and a phenyl group is particularly preferred.
- the aryl group is preferably a phenyl group, a biphenyl group, a terphenyl group, a naphthyl group, a fluorenyl group, a phenanthryl group, or an anthracenyl group.
- a phenyl group and a naphthyl group are more preferable. Particularly preferred is a phenyl group.
- the heteroaryl group is, for example, pyridyl group, furanyl group, thiophenyl group, quinolinyl group, isoquinolinyl group, pyrazinyl group, pyrimidyl group, pyridazinyl group, triazinyl group, naphthyridinyl group, cinnolinyl group, phthalazinyl group, quinoxalinyl group, quinazolinyl group, Benzofuranyl group, benzothiophenyl group, indolyl group, dibenzofuranyl group, dibenzothiophenyl group, carbazolyl group, benzocarbazolyl group, carbolinyl group, indolocarbazolyl group, benzofurocarbazolyl group, benzothienocarba Zolyl group, dihydroindenocarbazolyl group, benzoquinolinyl group, acridinyl group, dibenzoacridin
- the naphthyridinyl group is any of 1,5-naphthyridinyl group, 1,6-naphthyridinyl group, 1,7-naphthyridinyl group, 1,8-naphthyridinyl group, 2,6-naphthyridinyl group, and 2,7-naphthyridinyl group.
- the heteroaryl group may or may not have a substituent. Although carbon number of heteroaryl group is not specifically limited, Preferably it is 2-40, More preferably, it is the range of 2-30.
- the heteroaryl group includes a pyridyl group, furanyl group, thiophenyl group, quinolinyl group, pyrimidyl group, triazinyl group, benzofuranyl group, benzothiophenyl group, indolyl group , A dibenzofuranyl group, a dibenzothiophenyl group, a carbazolyl group, a benzimidazolyl group, an imidazopyridyl group, a benzoxazolyl group, a benzothiazolyl group, and a phenanthrolinyl group, and a pyridyl group, a furanyl group, a thiophenyl group, and a quinolinyl group are preferred. More preferred. Particularly preferred is a pyridyl group.
- the heteroaryl group includes a pyridyl group, furanyl group, thiophenyl group, quinolinyl group, pyrimidyl group, triazinyl group, benzofuranyl group, benzothiophenyl group, indolyl group, Dibenzofuranyl group, dibenzothiophenyl group, carbazolyl group, benzimidazolyl group, imidazopyridyl group, benzoxazolyl group, benzothiazolyl group, phenanthrolinyl group are preferable, pyridyl group, furanyl group, thiophenyl group, quinolinyl group are more preferable. Particularly preferred is a pyridyl group.
- Halogen refers to an atom selected from fluorine, chlorine, bromine and iodine.
- the carbonyl group, carboxyl group, oxycarbonyl group and carbamoyl group may or may not have a substituent.
- substituents include an alkyl group, a cycloalkyl group, an aryl group, and a heteroaryl group, and these substituents may be further substituted.
- An amino group is a substituted or unsubstituted amino group.
- substituent in the case of substitution include an aryl group, a heteroaryl group, a linear alkyl group, and a branched alkyl group.
- aryl group and heteroaryl group a phenyl group, a naphthyl group, a pyridyl group, and a quinolinyl group are preferable. These substituents may be further substituted.
- carbon number is not specifically limited, Preferably it is 2-50, More preferably, it is 6-40, Most preferably, it is the range of 6-30.
- silyl groups include trimethylsilyl groups, triethylsilyl groups, tert-butyldimethylsilyl groups, propyldimethylsilyl groups, vinyldimethylsilyl groups, and other alkylsilyl groups, phenyldimethylsilyl groups, tert-butyldiphenylsilyl groups, An arylsilyl group such as a phenylsilyl group or a trinaphthylsilyl group is shown. Substituents on silicon may be further substituted. Although carbon number of a silyl group is not specifically limited, Preferably it is the range of 1-30.
- the siloxanyl group refers to a silicon compound group via an ether bond such as a trimethylsiloxanyl group. Substituents on silicon may be further substituted.
- the boryl group is a substituted or unsubstituted boryl group.
- substituent in the case of substitution include an aryl group, a heteroaryl group, a linear alkyl group, a branched alkyl group, an aryl ether group, an alkoxy group, and a hydroxyl group, and among them, an aryl group and an aryl ether group are preferable.
- the phosphine oxide group is a group represented by —P ( ⁇ O) R 10 R 11 .
- R 10 R 11 is selected from the same group as R 1 to R 9 .
- arbitrary adjacent 2 substituents may be bonded to each other to form a conjugated or non-conjugated condensed ring.
- an element selected from nitrogen, oxygen, sulfur, phosphorus, and silicon may be included in addition to carbon.
- the condensed ring may be further condensed with another ring.
- the compound represented by the general formula (1) exhibits a high fluorescence quantum yield and has a small peak half-value width of the emission spectrum, so that efficient color conversion and high color purity can be achieved.
- the compound represented by the general formula (1) has various properties such as luminous efficiency, color purity, thermal stability, light stability, and dispersibility by introducing an appropriate substituent at an appropriate position. ⁇
- the physical properties can be adjusted.
- R 1 , R 3 , R 4 and R 6 are all hydrogen, at least one of R 1 , R 3 , R 4 and R 6 is a substituted or unsubstituted alkyl group or substituted or unsubstituted aryl.
- the group, a substituted or unsubstituted heteroaryl group shows better thermal stability and light stability.
- examples of the alkyl group include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, Preferred are alkyl groups having 1 to 6 carbon atoms such as sec-butyl group, tert-butyl group, pentyl group, hexyl group, and further excellent thermal stability. Therefore, methyl group, ethyl group, n-propyl group, isopropyl group are preferred. N-butyl group, sec-butyl group, and tert-butyl group are preferable. Further, from the viewpoint of preventing concentration quenching and improving the emission quantum yield, a sterically bulky tert-butyl group is more preferable. A methyl group is also preferably used from the viewpoint of ease of synthesis and availability of raw materials.
- the aryl group is preferably a phenyl group, a biphenyl group, a terphenyl group or a naphthyl group, more preferably A phenyl group and a biphenyl group are preferable, and a phenyl group is particularly preferable.
- the heteroaryl group is preferably a pyridyl group, a quinolinyl group or a thiophenyl group, more preferably a pyridyl group or a quinolinyl group. preferable. Particularly preferred is a pyridyl group.
- R 1 , R 3 , R 4 and R 6 may all be the same or different, and a substituted or unsubstituted alkyl group is preferable because of good solubility in a binder resin and a solvent.
- the alkyl group is preferably a methyl group from the viewpoint of ease of synthesis and availability of raw materials.
- R 1 , R 3 , R 4 and R 6 may all be the same or different and when they are substituted or unsubstituted aryl groups or substituted or unsubstituted heteroaryl groups, better thermal stability and In order to exhibit light stability, R 1 , R 3 , R 4 and R 6 may be the same or different from each other, and more preferably a substituted or unsubstituted aryl group.
- R 1 , R 3 , R 4 and R 6 may all be the same or different, and in the case of a substituted or unsubstituted aryl group, for example, R 1 ⁇ R 4 , R 3 ⁇ R 6 , R It is preferable to introduce a plurality of types of substituents such as 1 ⁇ R 3 or R 4 ⁇ R 6 .
- ⁇ indicates a group having a different structure. Since an aryl group that affects the color purity and an aryl group that affects the efficiency can be introduced at the same time, fine adjustment is possible.
- R 1 ⁇ R 3 or R 4 ⁇ R 6 is preferable in terms of improving the light emission efficiency and the color purity in a balanced manner.
- One or more aryl groups that affect color purity can be introduced into each pyrrole ring on both sides, and aryl groups that affect efficiency can be introduced at other positions, thereby maximizing both properties be able to.
- an aryl group substituted with an electron donating group is preferable.
- the electron donating group is an atomic group that donates electrons to a substituted atomic group by an induced effect or a resonance effect in organic electronic theory.
- Examples of the electron donating group include those having a negative value as the Hammett's rule substituent constant ( ⁇ p (para)).
- the Hammett's rule substituent constant ( ⁇ p (para)) can be cited from the Chemical Handbook, Basic Revision 5 (II-380).
- the electron donating group include, for example, an alkyl group ( ⁇ p of methyl group: ⁇ 0.17), an alkoxy group ( ⁇ p of methoxy group: ⁇ 0.27), an amino group ( ⁇ p of —NH 2 : ⁇ 0.66).
- an alkyl group having 1 to 8 carbon atoms or an alkoxy group having 1 to 8 carbon atoms is preferable, and a methyl group, an ethyl group, a tert-butyl group, or a methoxy group is more preferable.
- a tert-butyl group and a methoxy group are particularly preferable, and quenching due to aggregation of molecules is prevented.
- the substitution position of the substituent is not particularly limited, but it is preferable to bond at the meta position or the para position with respect to the bond position with the pyromethene skeleton because it is necessary to suppress the twist of the bond in order to improve the light stability.
- an aryl group having a bulky substituent such as a tert-butyl group, an adamantyl group, or a methoxy group is preferable.
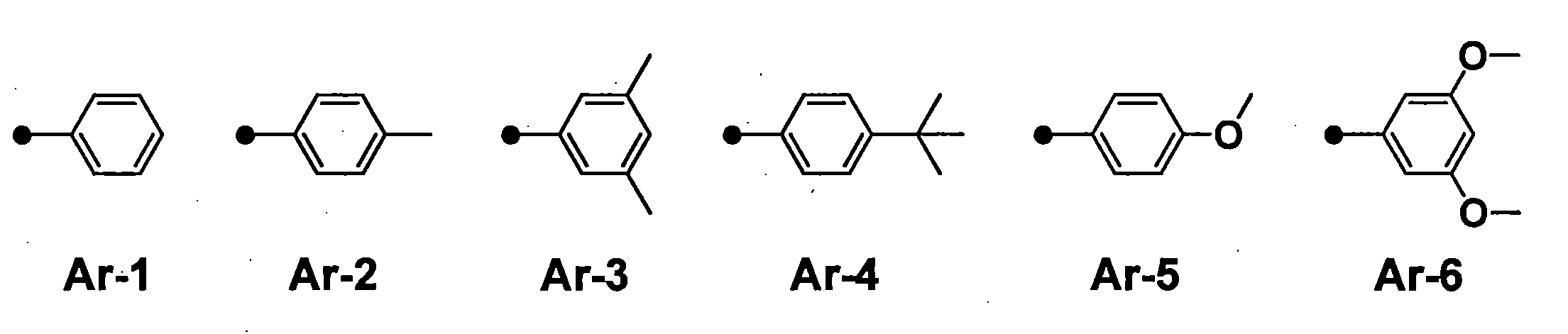
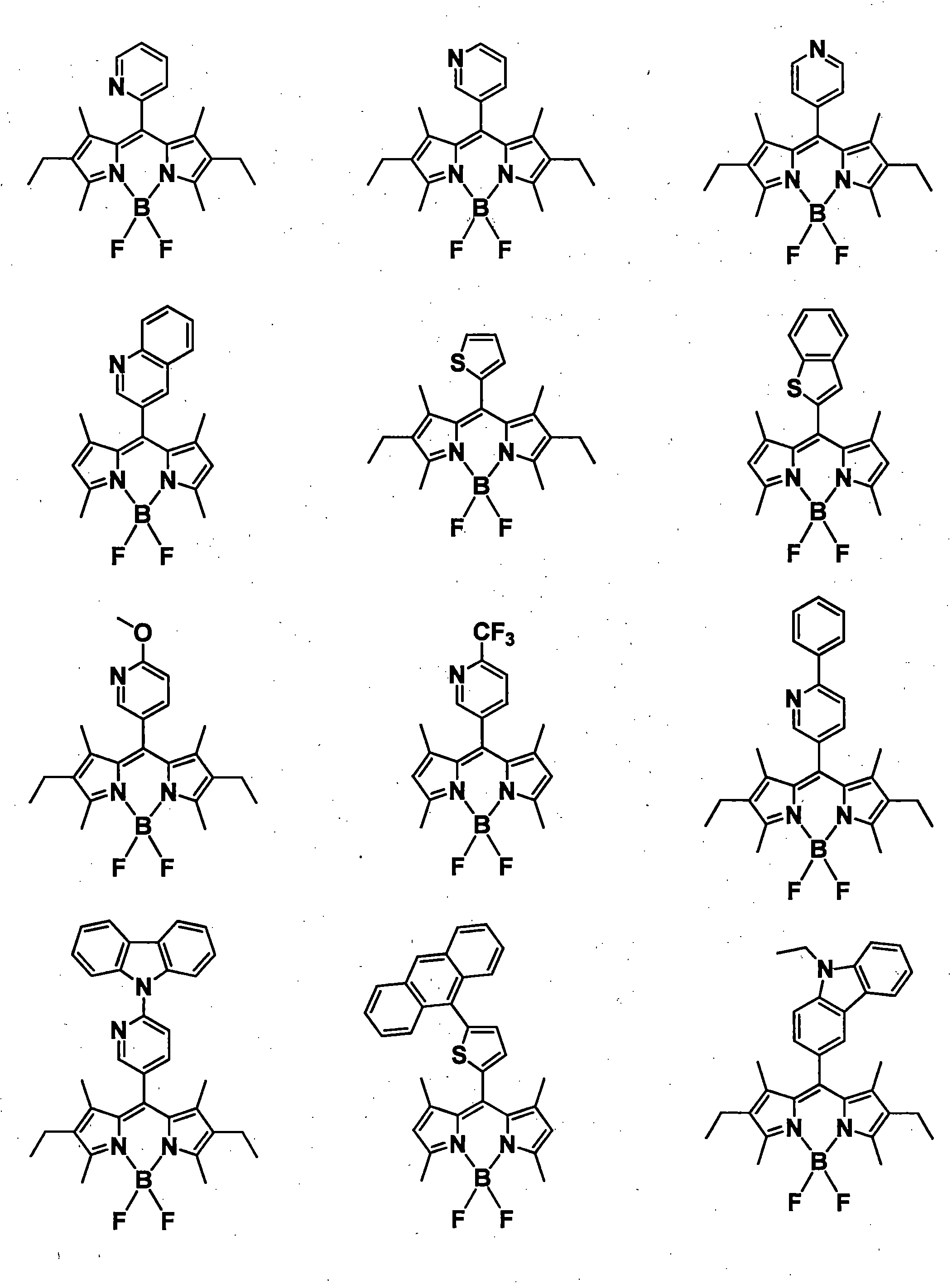
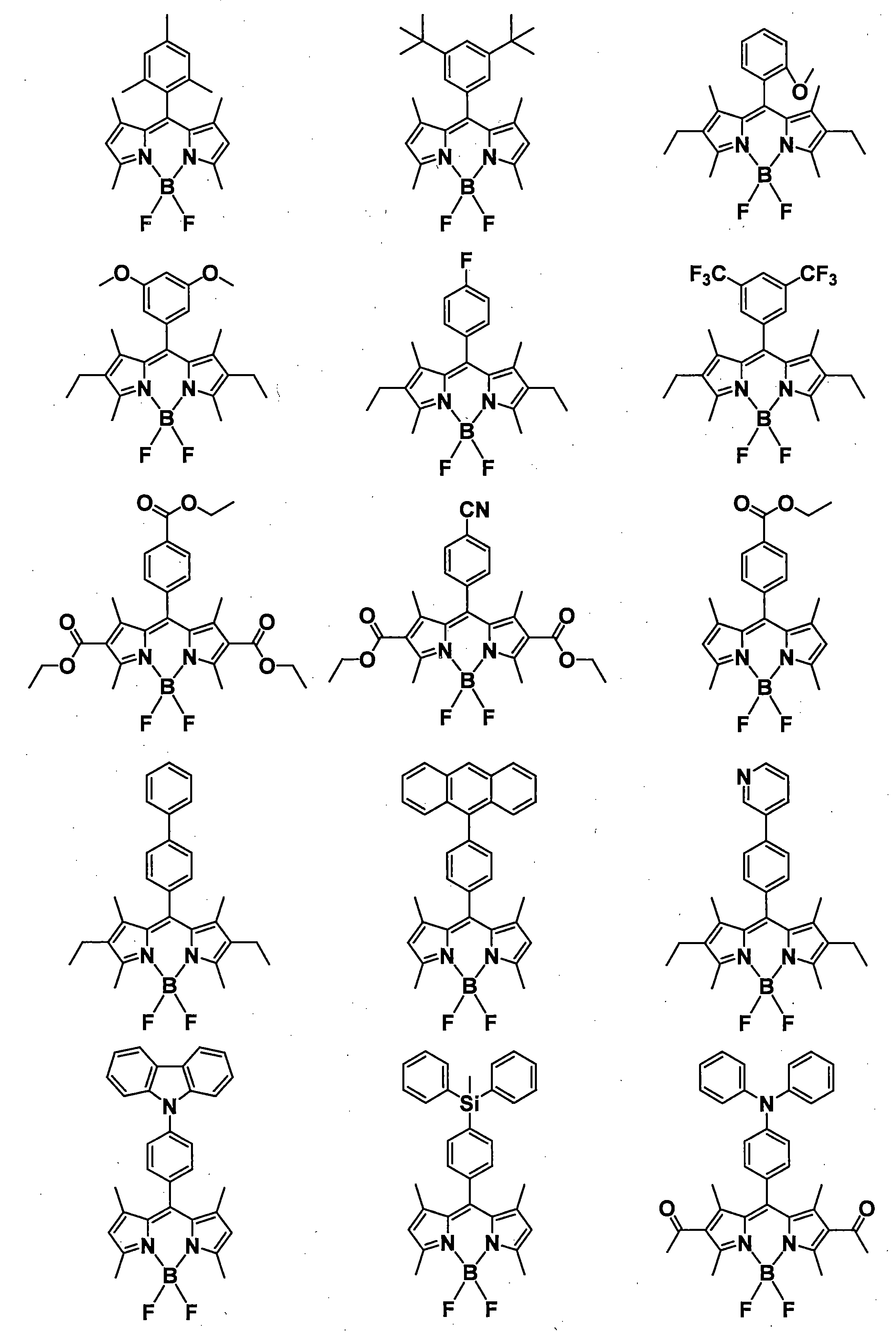
- R 1 , R 3 , R 4 and R 6 may all be the same or different, and in the case of a substituted or unsubstituted aryl group, they are each selected from the following Ar-1 to Ar-6: Is preferred.
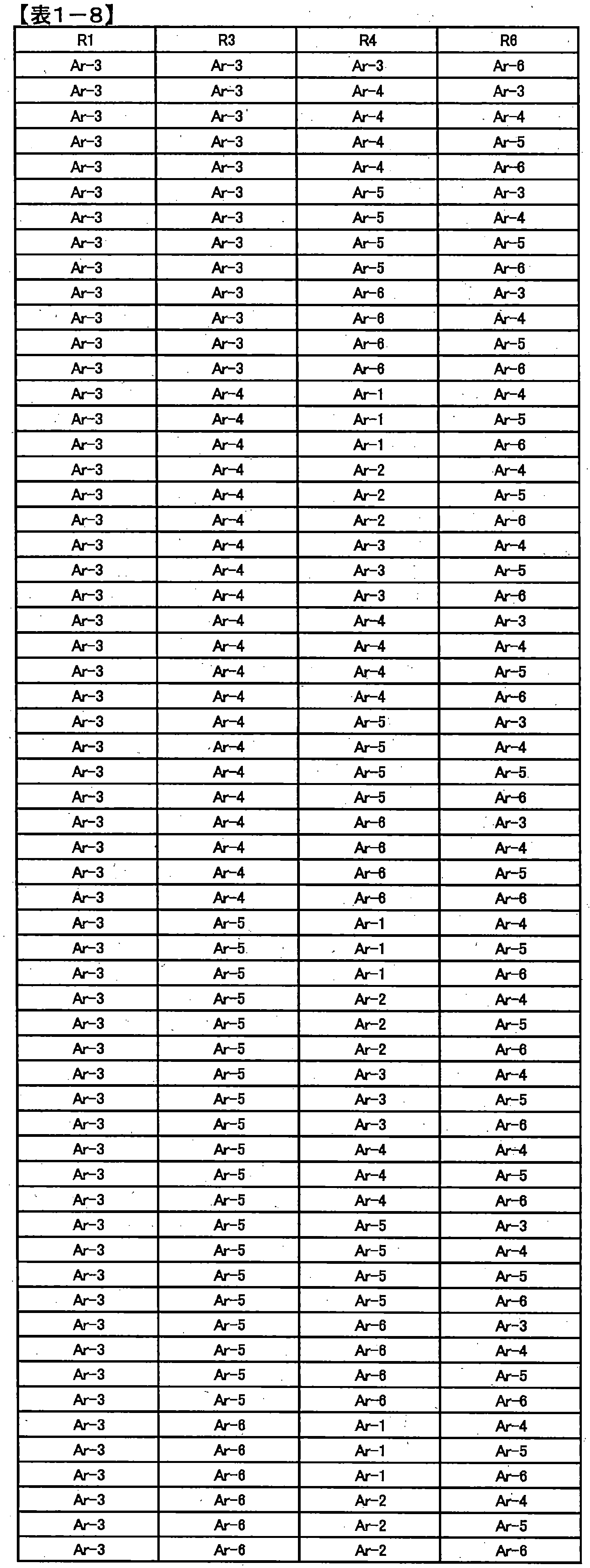
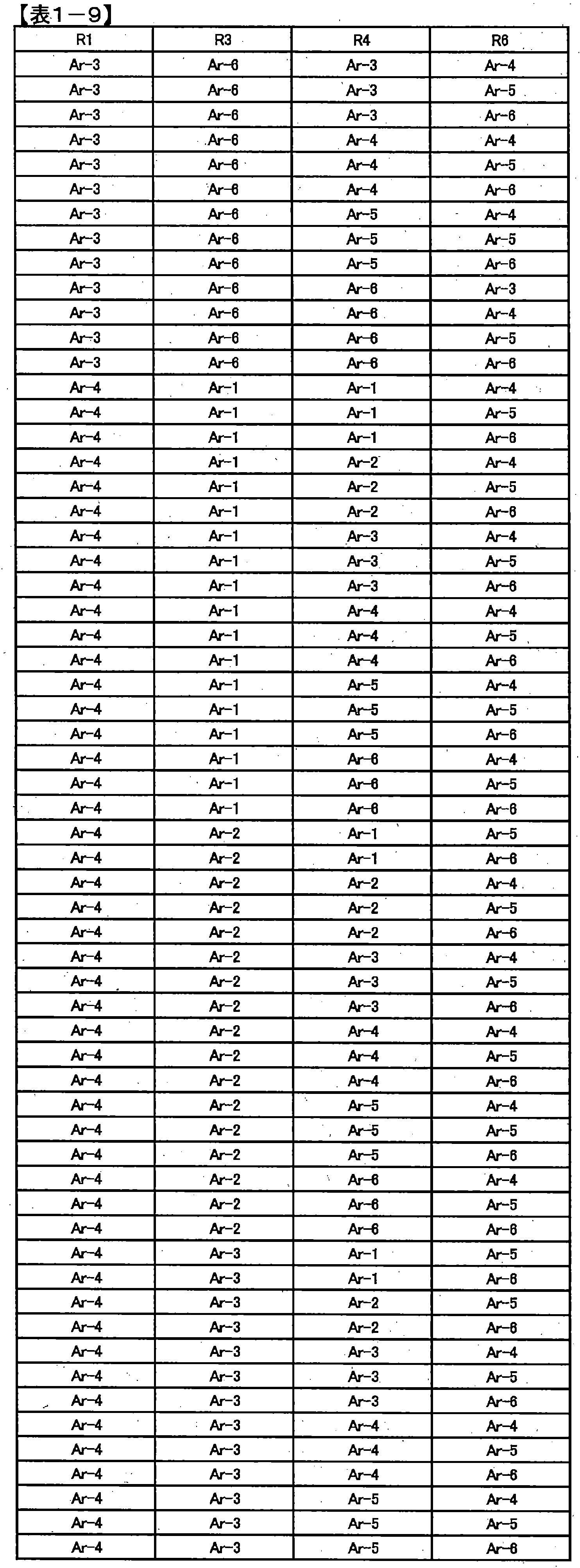
- preferred combinations of R 1 , R 3 , R 4 and R 6 include those shown in Table 1-1 to Table 1-11, but are not limited thereto.
- R 2 and R 5 are preferably hydrogen, an alkyl group, a carbonyl group, an oxycarbonyl group, or an aryl group, but are preferably an alkyl group or hydrogen from the viewpoint of thermal stability, and are easy to obtain a narrow half-value width in the emission spectrum. Hydrogen is more preferred.
- R 8 and R 9 are preferably an alkyl group, an aryl group, a heteroaryl group, fluorine, a fluorine-containing alkyl group, a fluorine-containing heteroaryl group or a fluorine-containing aryl group, which is stable against excitation light and has a higher fluorescence quantum yield. Therefore, it is more preferably a fluorine or fluorine-containing aryl group. In view of ease of synthesis, fluorine is more preferable.
- the fluorine-containing aryl group is an aryl group containing fluorine, and examples thereof include a fluorophenyl group, a trifluoromethylphenyl group, and a pentafluorophenyl group.
- the fluorine-containing heteroaryl group is a heteroaryl group containing fluorine, and examples thereof include a fluoropyridyl group, a trifluoromethylpyridyl group, and a trifluoropyridyl group.
- the fluorine-containing alkyl group is an alkyl group containing fluorine, and examples thereof include a trifluoromethyl group and a pentafluoroethyl group.
- X is preferably C—R 7 from the viewpoint of light stability.
- R 7 When X is C—R 7 , the substituent R 7 greatly affects the durability of the compound represented by the general formula (1), that is, the decrease in emission intensity over time. That is, when R 7 is hydrogen, the reactivity of this hydrogen is high, and it easily reacts with moisture and oxygen in the air, causing decomposition. In addition, when R 7 is a substituent having a large degree of freedom of movement of a molecular chain such as an alkyl group, for example, the reactivity is certainly lowered, but the compounds aggregate with time in the composition. In particular, the emission intensity is reduced due to concentration quenching. Therefore, R 7 is preferably a group that is rigid and has a low degree of freedom of movement and is unlikely to cause aggregation. Specifically, R 7 is a substituted or unsubstituted aryl group, or a substituted or unsubstituted heteroaryl group. It is preferable that
- X is C—R 7 and R 7 is a substituted or unsubstituted aryl group.
- aryl group a phenyl group, a biphenyl group, a terphenyl group, a naphthyl group, a fluorenyl group, a phenanthryl group, and an anthracenyl group are preferable from the viewpoint of not impairing the emission wavelength.
- R 7 is preferably a substituted or unsubstituted phenyl group, a substituted or unsubstituted biphenyl group, a substituted or unsubstituted terphenyl group, or a substituted or unsubstituted naphthyl group.
- a phenyl group, a substituted or unsubstituted biphenyl group, and a substituted or unsubstituted terphenyl group are more preferable. Particularly preferred is a substituted or unsubstituted phenyl group.
- R 7 is preferably a moderately bulky substituent. When R 7 has a certain amount of bulkiness, aggregation of molecules can be prevented, and luminous efficiency and durability are further improved.
- a more preferable example of such a bulky substituent includes a structure represented by the following general formula (2).
- r is hydrogen, alkyl group, cycloalkyl group, heterocyclic group, alkenyl group, cycloalkenyl group, alkynyl group, hydroxyl group, thiol group, alkoxy group, alkylthio group, aryl ether group, aryl thioether group, aryl group, heteroaryl Group, halogen, cyano group, aldehyde group, carbonyl group, carboxyl group, oxycarbonyl group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group, boryl group, and phosphine oxide group.
- k is an integer of 1 to 3. When k is 2 or more, r may be the same or different.
- R is preferably a substituted or unsubstituted aryl group from the viewpoint of giving a higher fluorescence quantum yield.
- aryl groups a phenyl group and a naphthyl group are particularly preferable examples.
- k in the general formula (2) is preferably 1 or 2, and k is more preferably 2 from the viewpoint of further preventing aggregation of molecules.
- the alkyl group in this case, a methyl group, an ethyl group, and a tert-butyl group are particularly preferred from the viewpoint of thermal stability.
- r is preferably a substituted or unsubstituted alkyl group, a substituted or unsubstituted alkoxy group or a halogen, More preferred are a group, an ethyl group, a tert-butyl group, and a methoxy group. From the viewpoint of dispersibility, a tert-butyl group and a methoxy group are particularly preferable, and quenching due to aggregation of molecules is prevented.
- At least one of R 1 to R 7 is preferably an electron withdrawing group.
- at least one of R 1 to R 6 is an electron withdrawing group
- R 7 is an electron withdrawing group
- at least one of R 1 to R 6 is an electron. It is preferably an attracting group and R 7 is an electron withdrawing group.
- the electron-withdrawing group is also called an electron-accepting group, and is an atomic group that attracts electrons from a substituted atomic group by an induced effect or a resonance effect in organic electron theory.
- Examples of the electron-withdrawing group include those that take a positive value as the Hammett's rule substituent constant ( ⁇ p (para)).
- the Hammett's rule substituent constant ( ⁇ p (para)) can be cited from the Chemical Handbook, Basic Revision 5 (II-380).
- a phenyl group also takes a positive value
- the phenyl group is not included in the electron withdrawing group of the present application.
- electron withdrawing groups include, for example, -F ( ⁇ p: +0.20), -Cl ( ⁇ p: +0.28), -Br ( ⁇ p: +0.30), -I ( ⁇ p: +0.30),- CO 2 R 12 ( ⁇ p: when R 12 is an ethyl group +0.45), —CONH 2 ( ⁇ p: +0.38), —COR 12 ( ⁇ p: when R 12 is a methyl group +0.49), —CF 3 ( ⁇ p: +0.51), - SO 2 R 12 ( ⁇ p: when R 12 is a methyl group +0.69), - NO 2 ( ⁇ p : +0.81) , and the like.
- R 12 each independently represents a hydrogen atom, a substituted or unsubstituted aromatic hydrocarbon group having 6 to 30 ring carbon atoms, a substituted or unsubstituted heterocyclic group having 5 to 30 ring atoms, substituted or unsubstituted
- a substituted alkyl group having 1 to 30 carbon atoms and a substituted or unsubstituted cycloalkyl group having 1 to 30 carbon atoms are represented. Specific examples of these groups include the same examples as described above.
- Preferred electron withdrawing groups include fluorine, fluorine-containing aryl groups, fluorine-containing heteroaryl groups, fluorine-containing alkyl groups, substituted or unsubstituted acyl groups, substituted or unsubstituted ester groups, substituted or unsubstituted amide groups, and substituted groups. Or an unsubstituted sulfonyl group or a cyano group is mentioned. This is because they are hardly chemically decomposed.
- More preferred electron-withdrawing groups include fluorine-containing alkyl groups, substituted or unsubstituted acyl groups, substituted or unsubstituted ester groups, and cyano groups. This is because concentration quenching can be prevented and the quantum yield of light emission can be improved. Particularly preferred is a substituted or unsubstituted ester group.
- R 1 to R 9 are the same as those in the general formula (1). However, it binds to L 1 at any m positions of R 1 to R 7 .
- the coupling with L 1 at any position of R 1 ⁇ R 7 refers to a carbon atom and L 1 which connects the R 1 ⁇ R 7 is directly bonded.
- L 1 is a linking group, and is selected from a single bond, an alkylene group, an arylene group, and a heteroarylene group.
- R 10 may be the same or different and each represents a substituted or unsubstituted alkyl group.
- M and n are natural numbers, and m ⁇ n ⁇ 5.
- OR 10 is an alkoxy group, and its bulkiness can prevent quenching due to aggregation of molecules.
- m ⁇ n ⁇ 5 bulky substituents increase, so the effect of preventing aggregation is increased, and high luminous efficiency can be realized.
- m ⁇ 2 is preferable because the aggregation suppressing effect is improved, and m ⁇ 3 is more preferable because the whole molecule is covered with a bulky substituent. More preferably, m ⁇ 4.
- L 1 is a group other than a single bond
- the group serves as a spacer between the pyromethene skeleton and —OR 10 .
- L 1 is preferably selected from an alkylene group, an arylene group, and a heteroarylene group.
- an arylene group and a heteroarylene group having high rigidity are preferable, and a phenylene group is particularly preferable because a twist of a carbon-carbon bond is not excessively increased.
- R 10 examples include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, a sec-butyl group, a tert-butyl group, a pentyl group, and a hexyl group.
- a methyl group is preferable in terms of ease of synthesis.
- R 1 , R 3 , R 4 and R 6 are used for bonding to L 1, and at least three of R 1 , R 3 , R 4 and R 6 are used. More preferably, it is used for bonding to L 1 . Furthermore, when all of R 1 , R 3 , R 4 and R 6 are used for bonding to L 1, it is particularly preferable because the entire molecule can be covered with a bulky substituent.
- R 7 is either a group represented by the general formula (2), or bind to L 1 at a position of R 7.
- R 1 , R 3 , R 4 and R 6 may all be the same or different, and each may be a substituted or unsubstituted alkyl group. Further, there is a case where X is C—R 7 and R 7 is a substituted aryl group, particularly preferably a group represented by the general formula (2).
- R 1 , R 3 , R 4 and R 6 may all be the same or different, and the above-mentioned Ar— 1 to Ar-6, and X is C—R 7 and R 7 is a substituted aryl group, particularly preferably an aryl group substituted with a methoxy group.
- the compound represented by the general formula (1) can be produced, for example, by the method described in JP-T-8-509471 and JP-A-2000-208262. That is, the target pyromethene metal complex is obtained by reacting a pyromethene compound and a metal salt in the presence of a base.
- a method of generating a carbon-carbon bond by using a coupling reaction between a halogenated derivative and a boronic acid or a boronic acid esterified derivative is exemplified. Is not to be done.
- an amino group or a carbazolyl group for example, there is a method of generating a carbon-nitrogen bond by using a coupling reaction between a halogenated derivative and an amine or a carbazole derivative under a metal catalyst such as palladium. Although it is mentioned, it is not limited to this.
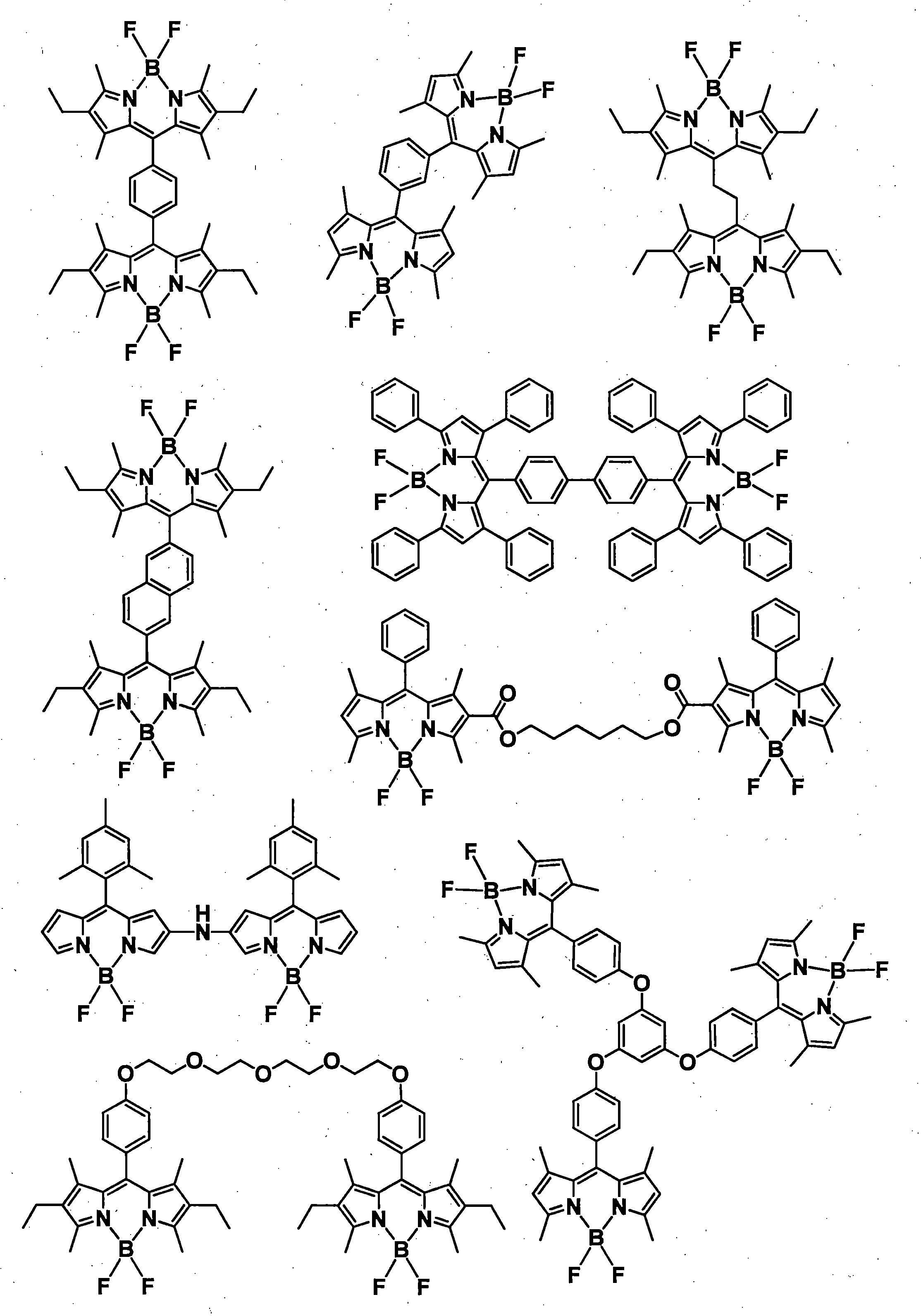
- the color conversion composition of the present invention can contain other compounds as needed in addition to the compound represented by the general formula (1).
- an assist dopant such as rubrene may be contained in order to further increase the energy transfer efficiency from the excitation light to the compound represented by the general formula (1).
- the above-mentioned organic light emitting material can be added.
- known light-emitting materials such as inorganic phosphors, fluorescent pigments, fluorescent dyes, and quantum dots can be added in combination.
- organic light emitting materials other than the compound represented by the general formula (1) are shown below, but are not particularly limited thereto.
- the color conversion composition of the present invention is a luminescent material (hereinafter referred to as “luminescent material (a)”) that emits light observed in a region having a peak wavelength of 500 nm or more and 580 nm or less by using excitation light having a wavelength of 430 nm or more and 500 nm or less.
- luminescent material (a) a luminescent material that emits light observed in a region having a peak wavelength of 500 nm or more and 580 nm or less by using excitation light having a wavelength of 430 nm or more and 500 nm or less.
- green light emission light emission observed in a region having a peak wavelength of 500 nm or more and 580 nm or less.
- the color conversion composition of the present invention comprises (a) a light emitting material that emits light having a peak wavelength of 500 nm to 580 nm by using excitation light having a wavelength in the range of 430 nm to 500 nm, and (b) a wavelength of 430 nm to 500 nm.
- a light emitting material exhibiting light emission observed in a region having a peak wavelength of 580 nm or more and 750 nm or less hereinafter referred to as “light emitting material (b)” when excited by either or both of excitation light in the range and light emission from the light emitting material (a). ) ”).
- light emission observed in a region having a peak wavelength of 580 nm or more and 750 nm or less is referred to as “red light emission”.
- Examples of the luminescent material (a) include coumarin derivatives such as coumarin 6, coumarin 7, coumarin 153, cyanine derivatives such as indocyanine green, fluorescein derivatives such as fluorescein, fluorescein isothiocyanate, carboxyfluorescein diacetate, and phthalocyanine derivatives such as phthalocyanine green.
- coumarin derivatives such as coumarin 6, coumarin 7, coumarin 153
- cyanine derivatives such as indocyanine green
- fluorescein derivatives such as fluorescein, fluorescein isothiocyanate, carboxyfluorescein diacetate
- phthalocyanine derivatives such as phthalocyanine green.
- Perylene derivatives such as diisobutyl-4,10-dicyanoperylene-3,9-dicarboxylate, pyromethene derivatives, stilbene derivatives, oxazine derivatives, naphthalimide derivatives, pyrazine derivatives, benzimidazole derivatives, benzoxazole derivatives, benzothiazoles Derivatives, imidazopyridine derivatives, azole derivatives, compounds having condensed aryl rings such as anthracene and derivatives thereof, aromatic amine derivatives, organic Metal complex compounds, and the like as preferred but not particularly limited thereto.
- pyromethene derivatives are particularly suitable compounds because they give high fluorescence quantum yield and good durability, and among them, the compound represented by the general formula (1) exhibits light emission with high color purity. This is preferable.
- Examples of the luminescent material (b) include cyanine derivatives such as 4-dicyanomethylene-2-methyl-6- (p-dimethylaminostyryl) -4H-pyran, rhodamine B, rhodamine 6G, rhodamine 101, sulforhodamine 101 and the like.
- Rhodamine derivatives pyridine derivatives such as 1-ethyl-2- (4- (p-dimethylaminophenyl) -1,3-butadienyl) -pyridinium-perchlorate, N, N′-bis (2,6-diisopropylphenyl) Perylene derivatives such as -1,6,7,12-tetraphenoxyperylene-3,4: 9,10-bisdicarbimide, porphyrin derivatives, pyromethene derivatives, oxazine derivatives, pyrazine derivatives, naphthacene and dibenzodiindeno Compounds having condensed aryl rings such as perylene, derivatives thereof, organometallic complexes Compounds and the like as preferred but not particularly limited thereto.
- pyromethene derivatives are particularly suitable compounds because they give high fluorescence quantum yield and good durability, and among them, the compound represented by the general formula (1) exhibits
- the content of the component (A) in the color conversion composition of the present invention depends on the molar absorption coefficient of the compound, the fluorescence quantum yield and the absorption intensity at the excitation wavelength, and the thickness and transmittance of the film to be produced. It is preferably 1.0 ⁇ 10 ⁇ 4 to 30 parts by weight, more preferably 1.0 ⁇ 10 ⁇ 3 to 10 parts by weight with respect to 100 parts by weight of component (B). It is particularly preferably 0 ⁇ 10 ⁇ 2 to 5 parts by weight.
- the color conversion composition contains both the light emitting material (a) that emits green light and the light emitting material (b) that emits red light, part of the green light is converted into red light.
- w a and w b are weight percent relative to the weight of the component (B).
- the binder resin forms a continuous phase, and may be any material that is excellent in molding processability, transparency, heat resistance, etc., for example, acrylic acid-based, methacrylic acid-based, polyvinyl cinnamate-based, ring rubber-based, etc.
- Photoreactive resist materials having reactive vinyl groups, epoxy resins, silicone resins (including organopolysiloxane cured products (crosslinked products) such as silicone rubber and silicone gel), urea resins, fluororesins, polycarbonate resins, acrylic resins , Urethane resins, melamine resins, polyvinyl resins, polyamide resins, phenol resins, polyvinyl alcohol resins, polyvinyl butyral resins, cellulose resins, aliphatic ester resins, aromatic ester resins, aliphatic polyolefin resins, aromatic polyolefin resins, etc. Things can be used. These copolymer resins may be used.
- thermosetting resins are more preferable because the film forming process is easy.
- an epoxy resin, a silicone resin, an acrylic resin, a polyester resin, or a mixture thereof can be suitably used.
- Additives such as dispersants and leveling agents for stabilizing coating films, plasticizers, crosslinking agents such as epoxy compounds, curing agents such as amines, acid anhydrides and imidazoles, and silane couplings as film surface modifiers It is also possible to add an adhesion aid such as an agent. It is also possible to add inorganic particles such as silica particles or silicone fine particles as a color conversion material precipitation inhibitor.
- the binder resin is preferably a silicone resin from the viewpoint of heat resistance.
- silicone resins addition reaction curable silicone compositions are preferred.
- the addition reaction curable silicone composition is heated and cured at room temperature or 50 to 200 ° C., and is excellent in transparency, heat resistance, and adhesiveness.
- the addition reaction curable silicone composition is formed by a hydrosilylation reaction of a compound containing an alkenyl group bonded to a silicon atom and a compound having a hydrogen atom bonded to a silicon atom.
- Such materials contain alkenyl groups bonded to silicon atoms such as vinyltrimethoxysilane, vinyltriethoxysilane, allyltrimethoxysilane, propenyltrimethoxysilane, norbornenyltrimethoxysilane, octenyltrimethoxysilane, etc.
- hydrogen atoms bonded to silicon atoms such as methylhydrogenpolysiloxane, dimethylpolysiloxane-CO-methylhydrogenpolysiloxane, ethylhydrogenpolysiloxane, methylhydrogenpolysiloxane-CO-methylphenylpolysiloxane, etc. Examples thereof include those formed by hydrosilylation reaction of the compounds having them.
- other publicly known ones such as those described in JP 2010-159411 A can be used.
- silicone sealing material for general LED applications as a commercially available product.
- Specific examples include OE-6630A / B and OE-6336A / B manufactured by Toray Dow Corning, and SCR-1012A / B and SCR-1016A / B manufactured by Shin-Etsu Chemical Co., Ltd.
- a hydrosilylation reaction retarder such as acetylene alcohol
- fine particles such as fumed silica, glass powder, quartz powder, etc.
- inorganic fillers and pigments such as titanium oxide, zirconia oxide, barium titanate, zinc oxide
- blend adhesiveness imparting agents such as a flame retardant, a heat resistant agent, antioxidant, a dispersing agent, a solvent, a silane coupling agent, and a titanium coupling agent.
- a low molecular weight polydimethylsiloxane component silicone oil or the like to the silicone resin composition for producing the color conversion film.
- Such components are preferably added in an amount of 100 to 2,000 ppm, more preferably 500 to 1,000 ppm, based on the entire composition.
- the binder resin and the compound represented by the general formula (1) in addition to the binder resin and the compound represented by the general formula (1), it is also possible to contain silicone fine particles.
- the binder resin and the compound represented by the general formula (1) the same compounds as described above are preferably used.
- These compounds have a role as a light stabilizer, particularly a singlet oxygen quencher.
- the singlet oxygen quencher is a material that traps and inactivates singlet oxygen formed by activation of oxygen molecules by light energy.
- the presence of the singlet oxygen quencher in the composition can prevent the light emitting material from being deteriorated by singlet oxygen.
- Singlet oxygen is known to be generated by the exchange of electrons and energy between a triplet excited state of a dye such as rose bengal or methylene blue and a ground state oxygen molecule.
- the contained light-emitting material is excited by excitation light, and light is converted by emitting light having a wavelength different from that of the excitation light. Since this excitation-emission cycle is repeated, the probability that singlet oxygen is generated by the interaction between the generated excited species and oxygen contained in the composition increases. Therefore, the probability of collision between the light emitting material and singlet oxygen is also increased, and thus the light emitting material is likely to be deteriorated.
- the compound represented by the general formula (1) has a higher reactivity with singlet oxygen than a compound having a condensed aryl ring such as perylene or a derivative thereof, and the effect of singlet oxygen on durability is large.
- the general formula (1) excellent in quantum yield and color purity is obtained.
- the durability of the represented compound can be improved.
- the compound of component (C) preferably has a small extinction coefficient in the visible light region.
- the molar extinction coefficient ⁇ is 100 or less over the entire wavelength range from 400 nm to 800 nm.
- ⁇ is preferably as small as possible, and more preferably 80 or less. More preferably, it is 60 or less, Most preferably, it is 50 or less.
- the tertiary amine refers to a compound having a structure in which all NH bonds of ammonia are replaced with NC bonds.
- substituent on the nitrogen atom include alkyl group, cycloalkyl group, heterocyclic group, alkenyl group, cycloalkenyl group, alkynyl group, aryl group, heteroaryl group, aldehyde group, carbonyl group, carboxyl group, oxycarbonyl group, It is selected from a condensed ring and an aliphatic ring formed between a carbamoyl group and an adjacent substituent. Moreover, these substituents may be further substituted with the above-mentioned substituents.
- the substituent on the nitrogen atom is preferably a substituted or unsubstituted alkyl group, a substituted or unsubstituted cycloalkyl group, a substituted or unsubstituted aryl group, or a substituted or unsubstituted heteroaryl group from the viewpoint of light stability.
- a substituted or unsubstituted alkyl group, a substituted or unsubstituted cycloalkyl group, or a substituted or unsubstituted aryl group is more preferable.
- the aryl group is preferably a phenyl group or a naphthyl group, and more preferably a phenyl group, since it does not inhibit light from the light source or light emission of the light emitting material. Further, since there is a concern that absorption in the visible region increases when the number of aryl groups on the nitrogen atom increases, among the three substituents on the nitrogen atom, the number of aryl groups is preferably two or less, and one or less. Is more preferable.
- At least one of the three substituents on the nitrogen atom is a substituted or unsubstituted alkyl group, since singlet oxygen can be trapped more efficiently.
- Preferred tertiary amines include triethylamine, 1,4-diazabicyclo [2.2.2. ] Octane, tri-n-butylamine, N, N-diethylaniline, 1,2,2,6,6-pentamethylpiperidine, bis (1,2,2,6,6-pentamethyl-4-piperidyl) sebacate Methacrylic acid 1,2,2,6,6-pentamethyl-4-piperidyl and the like, but are not particularly limited thereto.
- the catechol derivative refers to a compound having two or more hydroxyl groups in the benzene ring, including isomers such as resorcinol and hydroquinone. These compounds can trap singlet oxygen more efficiently than phenol derivatives having one hydroxyl group on the benzene ring.
- substituents on the benzene ring in addition to hydroxyl groups, hydrogen, alkyl groups, cycloalkyl groups, heterocyclic groups, alkenyl groups, cycloalkenyl groups, alkynyl groups, thiol groups, alkoxy groups, alkylthio groups, aryl ether groups, Arylthioether group, aryl group, heteroaryl group, halogen, cyano group, aldehyde group, carbonyl group, carboxyl group, oxycarbonyl group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group, boryl group, phosphine oxide group And a condensed ring formed between adjacent substituents and an aliphatic ring. Moreover, these substituents may be further substituted with the above-mentioned substituents.
- a substituted or unsubstituted alkyl group, a substituted or unsubstituted cycloalkyl group, a substituted or unsubstituted aryl group, a substituted or unsubstituted heteroaryl group, and halogen are preferable from the viewpoint of light stability, and substituted or unsubstituted.
- an alkyl group, a substituted or unsubstituted cycloalkyl group, a substituted or unsubstituted aryl group, and a halogen are more preferable.
- a substituted or unsubstituted alkyl group, a substituted or unsubstituted cycloalkyl group, and a halogen are more preferable because the color change after the reaction with the singlet oxygen quencher is small.
- Particularly preferred is a substituted or unsubstituted alkyl group.
- the position of the hydroxyl group on the benzene ring it is preferable that at least two hydroxyl groups are adjacent to each other. This is because photooxidation is less likely than resorcinol (1,3-substituted) and hydroquinone (1,4-substituted). In addition, since the absorption in the visible region is small even after oxidation, discoloration of the composition can be prevented.
- Preferred catechol derivatives include 4-tert-butylbenzene-1,2-diol, 3,5-di-tert-butylbenzene-1,2-diol and the like, but are not particularly limited thereto. .
- Nickel compounds are compounds containing nickel, and include inorganic salts such as nickel chloride, complexes such as bisacetylacetonato nickel, and organic acid salts such as nickel carbamate, but are not limited to these. is not.
- the organic acid refers to an organic compound having a carboxyl group, a sulfonyl group, a phenolic hydroxyl group, and a thiol group.
- complexes and organic acid salts are preferred in that they are uniformly dispersed in the composition.
- nickel complexes and organic acid nickel salts that can be suitably used as singlet quenchers include acetylacetonate nickel complexes, bisdithio- ⁇ -diketone nickel complexes, dithiolate nickel complexes, aminothiolate nickel Complex, thiocatechol nickel complex, salicylaldehyde oxime nickel complex, thiobisphenolate nickel complex, indoaniline nickel compound, carboxylic acid nickel salt, sulfonic acid nickel salt, phenolic nickel salt, carbamate nickel Examples thereof include, but are not limited to, salts and dithiocarbamic acid nickel salts.
- At least one of nickel salts of organic acids, acetylacetonate nickel complexes, and thiobisphenolate nickel complexes is preferable.
- a nickel salt of an organic acid is preferable because it is easy to synthesize and is inexpensive.
- a sulfonic acid nickel salt is preferable because the molar extinction coefficient in the visible region is small and does not absorb light emitted from a light source or a luminescent material. Furthermore, a nickel salt of an aryl sulfonic acid is more preferable in view of a better singlet quenching effect, and a nickel salt of an alkyl sulfonic acid is preferable from the viewpoint of solubility in a wide variety of solvents.
- aryl group of the aryl sulfonic acid a substituted or unsubstituted phenyl group is preferable, and a phenyl group substituted with an alkyl group is more preferable from the viewpoint of solubility in a solvent and dispersibility.
- acetylacetonate-based nickel complexes and thiobisphenolate-based nickel complexes are preferred because they are soluble in organic solvents and have a low molar extinction coefficient in the visible range.
- the ligands on nickel in these complexes are alkyl groups, cycloalkyl groups, heterocyclic groups, alkenyl groups, cycloalkenyl groups, alkynyl groups, thiol groups, alkoxy groups, alkylthio groups, aryl ether groups, aryl thioether groups, Substituents such as aryl group, heteroaryl group, halogen, cyano group, aldehyde group, carbonyl group, carboxyl group, oxycarbonyl group, carbamoyl group, amino group, nitro group, silyl group, siloxanyl group, boryl group, phosphine oxide group These substituents may be further substituted with the above-mentioned substituents.
- a substituted or unsubstituted alkyl group, a substituted or unsubstituted cycloalkyl group, a substituted or unsubstituted aryl group, a substituted or unsubstituted heteroaryl group, and halogen are preferable from the viewpoint of light stability, and substituted or unsubstituted.
- an alkyl group, a substituted or unsubstituted cycloalkyl group, a substituted or unsubstituted aryl group, and a halogen are more preferable.
- a substituted or unsubstituted alkyl group, a substituted or unsubstituted cycloalkyl group, and a halogen are more preferable because the color change after the reaction with the singlet oxygen quencher is small.
- Particularly preferred is a substituted or unsubstituted alkyl group.
- Nickel compounds having a molar extinction coefficient ⁇ of 100 or less over the entire wavelength range from 400 nm to 800 nm include nickel salts of p-toluylsulfonic acid, acetylacetone nickel (II) complexes, hexafluoroacetylacetone nickel (II) complexes, 2 , 2′-thiobisphenolate-n-butylamine nickel (II) complex, [2,2′-thiobis (4-tert-octylphenolate) -2-ethylhexylamine nickel (II) complex,
- the present invention is not limited to this, and any nickel compound having a molar extinction coefficient ⁇ of 100 or less over the entire wavelength range of 400 to 800 nm can be used.
- component (C) may be used alone or in combination of two or more.
- the content of the component (C) in the color conversion composition of the present invention depends on the molar absorption coefficient of the compound, the fluorescence quantum yield, the absorption intensity at the excitation wavelength, and the thickness and transmittance of the film to be produced. It is 1.0 ⁇ 10 ⁇ 3 parts by weight or more and 30 parts by weight or less, more preferably 1.0 ⁇ 10 ⁇ 2 parts by weight or more and 15 parts by weight or less with respect to 100 parts by weight of the component (B). The amount is more preferably 1.0 ⁇ 10 ⁇ 2 parts by weight to 10 parts by weight, and particularly preferably 1.0 ⁇ 10 ⁇ 1 parts by weight to 10 parts by weight.
- the content of the nickel compound is based on 100 parts by weight of the component (B). 1.0 ⁇ 10 ⁇ 3 to 15 parts by weight, more preferably 1.0 ⁇ 10 ⁇ 2 to 10 parts by weight, and more preferably 1.0 ⁇ 10 ⁇ 1 to 10 parts by weight. It is particularly preferred that the amount is not more than parts by weight.
- the color conversion composition of the present invention includes an antioxidant, a processing and heat stabilizer, a light-resistant stabilizer such as an ultraviolet absorber, and a silane coupling.
- An agent etc. can be contained.
- antioxidants examples include phenolic antioxidants such as 2,6-di-tert-butyl-p-cresol and 2,6-di-tert-butyl-4-ethylphenol, but are not particularly limited. It is not something. These antioxidants may be used alone or in combination.
- processing and heat stabilizer examples include phosphorus stabilizers such as tributyl phosphite, tricyclohexyl phosphite, triethylphosphine, and diphenylbutylphosphine, but are not particularly limited. These stabilizers may be used alone or in combination.
- Examples of the light resistance stabilizer include 2- (5-methyl-2-hydroxyphenyl) benzotriazole, 2- [2-hydroxy-3,5-bis ( ⁇ , ⁇ -dimethylbenzyl) phenyl] -2H- Although benzotriazoles, such as benzotriazole, can be mentioned, it is not specifically limited. These light resistance stabilizers may be used alone or in combination.
- the content of these additives in the color conversion composition of the present invention depends on the molar absorption coefficient of the compound, the fluorescence quantum yield and the absorption intensity at the excitation wavelength, and the thickness and transmittance of the film to be produced. It is 1.0 ⁇ 10 ⁇ 3 to 30 parts by weight, more preferably 1.0 ⁇ 10 ⁇ 2 to 15 parts by weight, with respect to 100 parts by weight of component (B), It is particularly preferably 1.0 ⁇ 10 ⁇ 1 part by weight or more and 10 parts by weight or less.
- the color conversion composition of the present invention may contain a solvent.
- the solvent is not particularly limited as long as it can adjust the viscosity of the resin in a fluid state and does not excessively affect the light emission and durability of the luminescent material.
- toluene is particularly preferably used because it does not affect the deterioration of the compound represented by the general
- ⁇ Method for producing color conversion composition> an example of the manufacturing method of the color conversion composition of this invention is demonstrated.
- a predetermined amount of the aforementioned light emitting material, binder resin, tertiary amine, catechol derivative, nickel compound, solvent, and the like are mixed.
- the color conversion composition is homogeneously mixed and dispersed by a homogenizer, a self-revolving stirrer, a three-roller, a ball mill, a planetary ball mill, a bead mill, etc. Things are obtained.
- Defoaming is preferably carried out under vacuum or reduced pressure conditions after mixing or dispersing. Further, a specific component may be mixed in advance or a process such as aging may be performed. It is also possible to remove the solvent with an evaporator to obtain a desired solid content concentration.
- the configuration of the color conversion film is not limited as long as it includes a layer obtained by curing the color conversion composition.
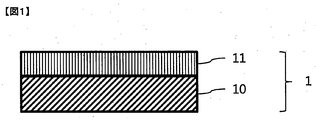
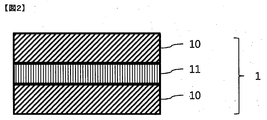
- a typical structural example of the color conversion film as shown in FIG. 1, a laminate of a base layer 10 and a color conversion layer 11 obtained by curing the color conversion composition, or shown in FIG.
- interposed by the some base material layer 10 is mentioned.
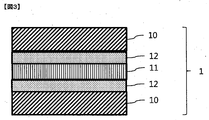
- the color conversion film may be further provided with a barrier film 12 as shown in FIG. 3 in order to prevent the color conversion layer from being deteriorated by oxygen, moisture or heat.
- the thickness of the color conversion film is not particularly limited, but the total of all layers is preferably 1 to 5000 ⁇ m. If it is smaller than 1 ⁇ m, there is a problem that the toughness of the film is reduced. If it exceeds 5000 ⁇ m, cracks are likely to occur and color conversion film molding is difficult. More preferably, it is 10 to 1000 ⁇ m, more preferably 15 to 500 ⁇ m, and particularly preferably 30 to 300 ⁇ m.
- the film thickness relating to the color conversion film in the present invention is the film thickness (average film thickness) measured based on the thickness measurement method A by mechanical scanning in JIS K7130 (1999) plastic-film and sheet-thickness measurement method. ).
- Base material layer As a base material layer, a well-known metal, a film, glass, ceramic, paper etc. can be used without a restriction
- glass and resin films are preferably used because of the ease of producing the color conversion film and the ease of forming the color conversion film. Further, a film having a high strength is preferred so that there is no fear of breakage when handling a film-like substrate.
- Resin films are preferred in terms of their required characteristics and economy, and among these, plastic films selected from the group consisting of PET, polyphenylene sulfide, polycarbonate, and polypropylene are preferred in terms of economy and handleability.
- plastic films selected from the group consisting of PET, polyphenylene sulfide, polycarbonate, and polypropylene are preferred in terms of economy and handleability.
- a polyimide film is preferable in terms of heat resistance.
- the surface of the base material layer may be subjected to a mold release treatment in advance for ease of film peeling.
- the thickness of the base material layer is not particularly limited, but the lower limit is preferably 5 ⁇ m or more, more preferably 25 ⁇ m or more, and further preferably 38 ⁇ m or more. Moreover, as an upper limit, 5000 micrometers or less are preferable and 3000 micrometers or less are more preferable.
- the color conversion layer can be dried using a general heating device such as a hot air dryer or an infrared dryer.
- a general heating device such as a hot air dryer or an infrared dryer is used.
- the heating conditions are usually 40 to 250 ° C. for 1 minute to 5 hours, preferably 60 ° C. to 200 ° C. for 2 minutes to 4 hours. It is also possible to perform heat curing stepwise such as step cure.
- the substrate can be changed as necessary.
- examples of a simple method include a method of performing replacement using a hot plate, a method of using a vacuum laminator and a dry film laminator, and the like, but are not limited thereto.
- the thickness of the color conversion layer is not particularly limited, but is preferably 1 to 1000 ⁇ m, more preferably 10 to 1000 ⁇ m. If it is smaller than 1 ⁇ m, there is a problem that the toughness of the film is reduced. If it exceeds 1000 ⁇ m, cracks are likely to occur and color conversion film molding is difficult. More preferably, it is 10 to 100 ⁇ m, still more preferably 15 to 100 ⁇ m, and particularly preferably 30 to 100 ⁇ m.
- barrier film As the barrier film, it is appropriately used in the case of improving the gas barrier property with respect to the color conversion layer.
- films made of various resins such as polyvinylidene chloride, acrylic resins, silicon resins, melamine resins, urethane resins, fluorine resins, and polyvinyl alcohol resins such as saponified vinyl acetate can be given.
- Examples of the film having a barrier function against moisture include polyethylene, polypropylene, nylon, polyvinylidene chloride, vinylidene chloride and vinyl chloride, vinylidene chloride and acrylonitrile copolymer, fluororesin, and saponified vinyl acetate.
- Examples thereof include films made of various resins such as polyvinyl alcohol resins.
- the barrier film may be provided on both sides of the color conversion layer 12 as shown in FIG. 3, or may be provided only on one side.
- the light diffusing layer In addition, depending on the required functions of the color conversion film, the light diffusing layer, antireflection function, antiglare function, antireflection antiglare function, hard coat function (friction resistance function), antistatic function, antifouling function, electromagnetic wave
- An auxiliary layer having a shield function, an infrared cut function, an ultraviolet cut function, a polarization function, and a toning function may be further provided.
- any excitation light can be used as long as it emits light in a wavelength region that can be absorbed by a light-emitting substance to be mixed such as the compound represented by the general formula (1).
- any excitation light such as a hot cathode tube, a cold cathode tube, a fluorescent light source such as inorganic EL, an organic electroluminescence element light source, an LED light source, an incandescent light source, or sunlight can be used.
- An LED is a suitable excitation light.
- a blue LED having excitation light in the range of 430 to 500 nm is a more suitable excitation light in that the color purity of blue light can be increased.
- the general formula (1) A light-emitting substance such as the represented compound or an organic compound such as a binder resin is not preferable because it is easily photodegraded.
- the excitation light may have one type of emission peak or two or more types of emission peaks, but in order to improve color purity, one having one type of emission peak is preferable. It is also possible to use a combination of a plurality of excitation light sources having different types of emission peaks.
- the light source unit in the present invention includes at least a light source and a color conversion composition or a color conversion film.
- the arrangement method of the light source and the color conversion composition is not particularly limited, and may be configured by directly applying the color conversion composition to the light source, or may be a film separated from the light source, You may take the structure which apply
- the arrangement method of the light source and the color conversion film is not particularly limited, and the light source and the color conversion film may be in close contact with each other, or the remote phosphor that separates the light source and the color conversion film may be used. You may take a fur form. Further, for the purpose of increasing the color purity, a configuration including a color filter may be taken.
- the excitation light in the range of 430 to 500 nm has a relatively low excitation energy and can prevent decomposition of the luminescent substance such as the compound represented by the general formula (1), so that the light source is in the range of 430 to 500 nm.
- a light emitting diode having maximum light emission is preferable.
- the light source unit in the present invention can be used for displays, lighting, interiors, signs, signboards, and the like, but is particularly preferably used for displays and lighting applications.
- compounds G-1 to G-8, R-1 to R-4, and Q-1 to Q-10 are the compounds shown below.
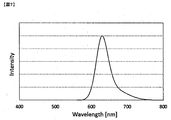
- the fluorescence spectrum of the compound was obtained when an F-2500 type spectrofluorometer (manufactured by Hitachi, Ltd.) was used, and the compound was dissolved in toluene at a concentration of 1 ⁇ 10 ⁇ 6 mol / L and excited at a wavelength of 460 nm. The fluorescence spectrum of was measured.
- a light emitting device equipped with each color conversion film and a blue LED element (manufactured by ProLight; model number PM2B-3LBE-SD, emission peak wavelength: 460 nm) is supplied with a current of 10 mA to light the LED, and a spectral radiance meter (CS- 1000, manufactured by Konica Minolta), emission spectrum, emission intensity and chromaticity at peak wavelength were measured.
- the distance between each color conversion film and the blue LED element was 3 cm.
- a light emitting device equipped with each color conversion film and a blue LED element (manufactured by ProLight; model number PM2B-3LBE-SD, emission peak wavelength: 460 nm) is supplied with a current of 10 mA to light the LED chip, and a spectral radiance meter (CS -1000, manufactured by Konica Minolta) was used to measure the initial luminance.
- the distance between each color conversion film and the blue LED element was 3 cm.
- the light durability was evaluated by continuously irradiating light from the blue LED element at room temperature and observing the time until the luminance decreased by a certain amount.
- the absorbance at each wavelength was calculated, a calibration curve was created from the graph with the absorbance on the vertical axis and the molar concentration (mol / L) on the horizontal axis, and the molar extinction coefficient for each wavelength was calculated. Asked.
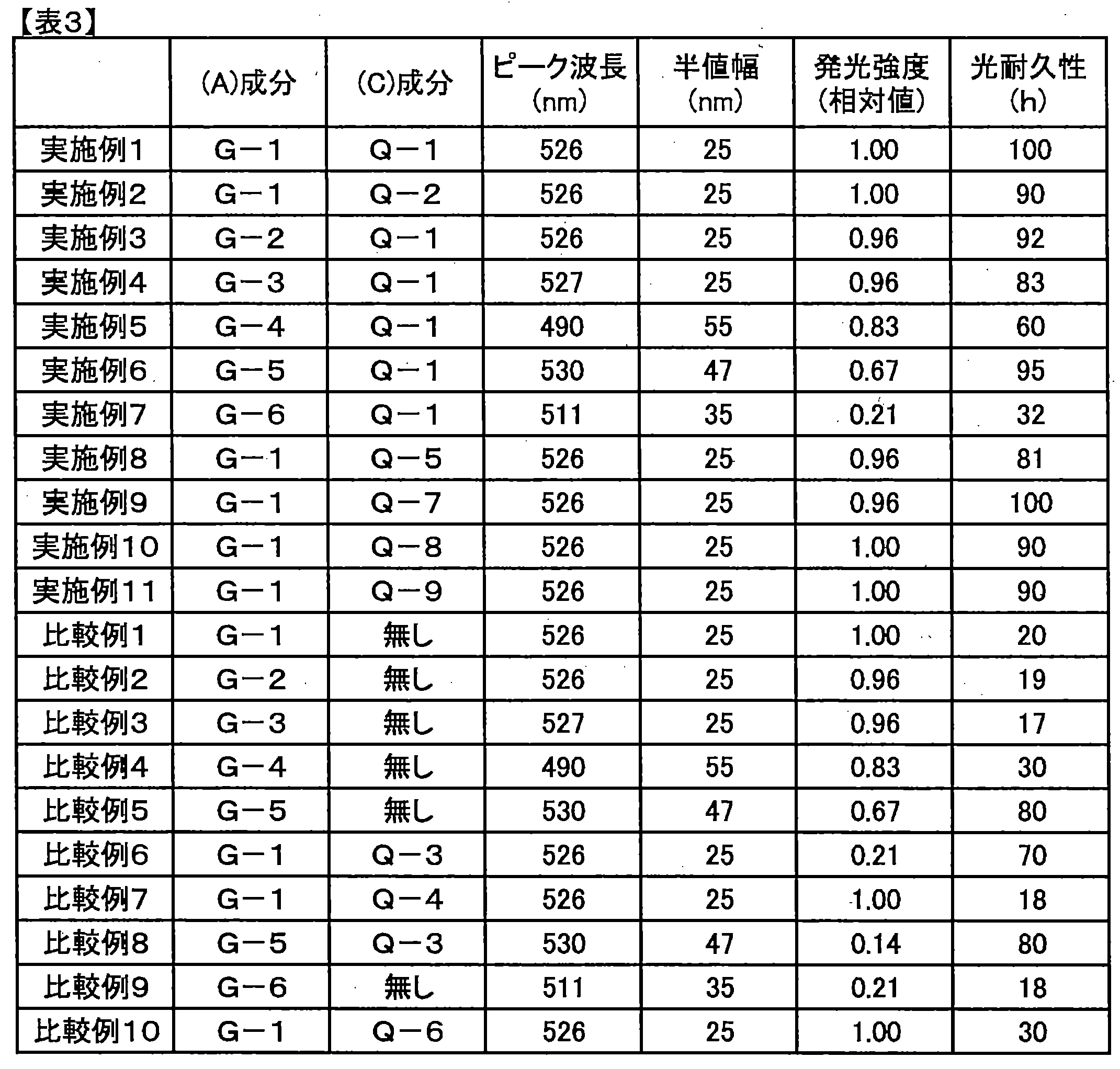
- Table 2 shows the molar extinction coefficients obtained for the compounds Q-1 to Q-10. However, ⁇ max in the table is the maximum value of the molar extinction coefficient ⁇ over the entire wavelength region of wavelengths from 400 nm to 800 nm.
- Synthesis example 1 Synthesis method of compound G-1, 3,5-dibromobenzaldehyde (3.0 g), 4-t-butylphenylboronic acid (5.3 g), tetrakis (triphenylphosphine) palladium (0) (0.4 g), carbonic acid Potassium (2.0 g) was placed in the flask and purged with nitrogen. Degassed toluene (30 mL) and degassed water (10 mL) were added thereto, and the mixture was refluxed for 4 hours. The reaction solution was cooled to room temperature, and the organic layer was separated and washed with saturated brine. The organic layer was dried over magnesium sulfate and filtered, and then the solvent was distilled off. The obtained reaction product was purified by silica gel chromatography to obtain 3,5-bis (4-tert-butylphenyl) benzaldehyde (3.5 g) as a white solid.
- the absorption spectrum of this compound was as shown in FIG. 4, and showed light absorption characteristics with a blue excitation light source (460 nm).
- the fluorescence spectrum was as shown in FIG. 5 and showed a sharp emission peak in the green region.
- the fluorescence quantum yield was 83%, and the compound was capable of efficient color conversion.
- Synthesis example 2 Synthesis Method of Compound R-1 4- (4-t-butylphenyl) -2- (4-methoxyphenyl) pyrrole (300 mg), 2-methoxybenzoyl chloride (201 mg) and toluene (10 ml) were mixed at 120 ° C. under a nitrogen stream at 6 ° C. Heated for hours. After cooling to room temperature, it was evaporated. After washing with 20 ml of ethanol and vacuum drying, 260 mg of 2- (2-methoxybenzoyl) -3- (4-tert-butylphenyl) -5- (4-methoxyphenyl) pyrrole was obtained.
- the absorption spectrum of this compound was as shown in FIG. 6 and showed light absorption characteristics for blue and green excitation light sources.
- the fluorescence spectrum was as shown in FIG. 7 and showed a sharp emission peak in the red region.
- the fluorescence quantum yield was 90%, and the compound was capable of efficient color conversion.
- Example 1 With respect to 100 parts by weight of silicone resin “OE-6630A / B” (manufactured by Dow Corning Toray), 0.20 parts by weight of compound G-1 as component (A), 0.5 parts by weight of curing agent, C) After mixing 1.0 part by weight of compound Q-1 as a component, a planetary stirring and defoaming device “Mazerustar KK-400” (manufactured by Kurabo Industries) is used for stirring and defoaming at 1000 rpm for 20 minutes to produce a film. A color conversion composition as a resin solution was obtained.
- a resin solution for film preparation was applied on “Therapy” BLK (manufactured by Toray Film Processing Co., Ltd.), heated at 130 ° C. for 2 hours, and dried to obtain a color conversion film having a thickness of 200 ⁇ m.
- Examples 2 to 11 and Comparative Examples 1 to 10 A color conversion film was prepared and evaluated in the same manner as in Example 1 except that the compounds described in Table 3 were used as the component (A) and the component (C). The results are shown in Table 3. However, the emission intensity (relative value) in the table is a relative value when the intensity in Comparative Example 1 is 1.00.
- Example 12 0.08 parts by weight of Compound R-1 as component (A), 0.5 parts by weight of curing agent, and Compound Q- After 1 part by weight of 1 was mixed, the mixture was stirred at 300 rpm for 1 hour to produce a composition.
- the above composition was applied on a stretched polyethylene terephthalic acid film (XG5P1 manufactured by Toray Advanced Materials Co., Ltd.) using a bar coating method, and then dried at 120 ° C. for 5 minutes to form a coating layer having an average film thickness of 10 ⁇ m. Formed. Thereafter, a diffusion film (“Texcell” (registered trademark) TDF127, manufactured by Toray Advanced Materials Co., Ltd.) was laminated and then aged at 60 ° C. for 1 hour to obtain a color conversion film.
- Texcell registered trademark
- TDF127 manufactured by Toray Advanced Materials Co., Ltd.
- Examples 13 to 21 and Comparative Examples 11 to 17 A color conversion film was prepared and evaluated in the same manner as in Example 12 except that the compounds described in Table 4 were used as the component (A) and the component (C). The results are shown in Table 4. However, the emission intensity (relative value) in the table is a relative value when the intensity in Comparative Example 11 is 1.00.
- Example 22 As a component (A), 0.20 part by weight of compound G-1, 0.08 part by weight of compound R-1 and 0.5 part of curing agent are added to 100 parts by weight of the two-component thermosetting epoxy acrylic resin. After mixing 1.0 part by weight of compound Q-1 as part by weight and component (C), the mixture was stirred at 300 rpm for 1 hour to produce a composition.
- the above composition was applied on a stretched polyethylene terephthalic acid film (XG5P1 manufactured by Toray Advanced Materials Co., Ltd.) using a bar coating method, and then dried at 120 ° C. for 5 minutes to form a coating layer having an average film thickness of 10 ⁇ m. Formed. Thereafter, a diffusion film (“Texcell” (registered trademark) TDF127, manufactured by Toray Advanced Materials Co., Ltd.) was laminated and then aged at 60 ° C. for 1 hour to obtain a color conversion film.
- Texcell registered trademark
- TDF127 manufactured by Toray Advanced Materials Co., Ltd.
- Examples 23 to 29 and Comparative Examples 18 to 19 A color conversion film was prepared and evaluated in the same manner as in Example 22 except that the compounds described in Table 5 were used as the component (A) and the component (C). The results are shown in Table 5. However, the emission intensity (relative value) in the table is a relative value when the intensity in Comparative Example 18 is 1.00.
- Example 30 With respect to 100 parts by weight of the acrylic resin “KC-7000” (manufactured by Kyoeisha Chemical Co., Ltd.), 0.25 parts by weight of the compound G-1 as the component (A) and 1. After mixing 0 part by weight, the mixture was stirred at 300 rpm for 1 hour to produce a composition.
- barrier film 1 After coating the above composition on a polyethylene terephthalate film (hereinafter referred to as barrier film 1) whose surface has been subjected to alumina vapor deposition using a bar coating method, it is dried at 120 ° C. for 5 minutes, and color conversion with an average film thickness of 10 ⁇ m is performed. A layer was formed.
- thermosetting adhesive layer was formed on another barrier film 1 by coating, and the adhesive layer was laminated on the color conversion layer.
- Texcell (registered trademark) (R) TDF127 manufactured by Toray Advanced Materials Co., Ltd.) was laminated, and then aged at 60 ° C. for 1 hour to obtain a color conversion film.
- Examples 31 to 33 and Comparative Example 22 A color conversion film was prepared and evaluated in the same manner as in Example 27 except that the compounds described in Table 6 were used as the component (A) and the component (C). The results are shown in Table 6. However, the emission intensity (relative value) in the table is a relative value when the intensity in Comparative Example 22 is 1.00.
- Example 34 With respect to 100 parts by weight of the acrylic resin “KC-7000” (manufactured by Kyoeisha Chemical Co., Ltd.), 0.25 parts by weight of the compound G-7 as the component (A) and 1. 1 of the compound Q-1 as the component (C) After 0 part by weight and 1.0 part by weight of compound Q-2 were mixed, the mixture was stirred at 300 rpm for 1 hour to produce a composition.
- composition was applied onto a polyethylene terephthalate film (Toray, U48) using a bar coating method, and then dried at 120 ° C. for 5 minutes to form a color conversion layer having an average film thickness of 16 ⁇ m.
- a polyethylene terephthalate film Toray, U48
- Texcell (registered trademark) (R) TDF127 manufactured by Toray Advanced Materials Co., Ltd.) was laminated, and then aged at 60 ° C. for 1 hour to obtain a color conversion film.
- Examples 35-36 A color conversion film was prepared and evaluated in the same manner as in Example 34 except that the compounds described in Table 7 were used as the component (A) and the component (C). The results are shown in Table 7. However, the emission intensity (relative value) in the table is a relative value when the intensity in Comparative Example 23 described later is 1.00.
- Examples 37 to 41 and Comparative Examples 23 to 24 For 100 parts by weight of the acrylic resin “KC-7000” (manufactured by Kyoeisha Chemical Co., Ltd.), 0.25 parts by weight of the compound described in Table 7 as the component (A) and a film in Table 7 as the component (C) Fabricated and evaluated. The results are shown in Table 7. However, the emission intensity (relative value) in the table is a relative value when the intensity in Comparative Example 23 is 1.00.
Abstract
Description
(A)少なくとも1種の発光材料
(B)バインダー樹脂
(C)3級アミン、カテコール誘導体およびニッケル化合物のうち少なくとも1つ
を含有し、
前記3級アミン、カテコール誘導体およびニッケル化合物は、波長400nm以上800nm以下の波長域全域でモル吸光係数εが100以下である、色変換組成物である。
本発明の色変換組成物は、少なくとも1種の発光材料を含む。ここで、本発明における発光材料とは、何らかの光が照射されたときに、その光とは異なる波長の光を発する材料のことをいう。
ナフタレン、アントラセン、フェナンスレン、ピレン、クリセン、ナフタセン、トリフェニレン、ペリレン、フルオランテン、フルオレン、インデン等の縮合アリール環を有する化合物やその誘導体;
フラン、ピロール、チオフェン、シロール、9-シラフルオレン、9,9’-スピロビシラフルオレン、ベンゾチオフェン、ベンゾフラン、インドール、ジベンゾチオフェン、ジベンゾフラン、イミダゾピリジン、フェナントロリン、ピリジン、ピラジン、ナフチリジン、キノキサリン、ピロロピリジン等のヘテロアリール環を有する化合物やその誘導体;
ボラン誘導体;
1,4-ジスチリルベンゼン、4,4’-ビス(2-(4-ジフェニルアミノフェニル)エテニル)ビフェニル、4,4’-ビス(N-(スチルベン-4-イル)-N-フェニルアミノ)スチルベン等のスチルベン誘導体;
芳香族アセチレン誘導体、テトラフェニルブタジエン誘導体、アルダジン誘導体、ピロメテン誘導体、ジケトピロロ[3,4-c]ピロール誘導体;
クマリン6、クマリン7、クマリン153などのクマリン誘導体;
イミダゾール、チアゾール、チアジアゾール、カルバゾール、オキサゾール、オキサジアゾール、トリアゾールなどのアゾール誘導体およびその金属錯体;
インドシアニングリーン等のシアニン系化合物;
フルオレセイン・エオシン・ローダミン等のキサンテン系化合物やチオキサンテン系化合物;
ポリフェニレン系化合物、ナフタルイミド誘導体、フタロシアニン誘導体およびその金属錯体、ポルフィリン誘導体およびその金属錯体;
ナイルレッドやナイルブルー等のオキサジン系化合物;
ヘリセン系化合物;
N,N’-ジフェニル-N,N’-ジ(3-メチルフェニル)-4,4’-ジフェニル-1,1’-ジアミン等の芳香族アミン誘導体;および
イリジウム(Ir)、ルテニウム(Ru)、ロジウム(Rh)、パラジウム(Pd)、白金(Pt)、オスミウム(Os)、及びレニウム(Re)等の有機金属錯体化合物;
等が好適なものとして挙げられるが、特にこれらに限定されるものではない。
例えば、R1、R3、R4およびR6が全て水素の場合に比べ、R1、R3、R4およびR6の少なくとも1つが置換もしくは無置換のアルキル基や置換もしくは無置換のアリール基、置換もしくは無置換のヘテロアリール基である場合の方が、より良い熱的安定性および光安定性を示す。
_これらの化合物の中でも、ピロメテン誘導体は高い蛍光量子収率を与え、耐久性が良好なので特に好適な化合物であり、中でも一般式(1)で表される化合物は、色純度の高い発光を示すことから好ましい。
_これらの化合物の中でも、ピロメテン誘導体は高い蛍光量子収率を与え、耐久性が良好なので特に好適な化合物であり、中でも一般式(1)で表される化合物は、色純度の高い発光を示すことから好ましい。
バインダー樹脂は、連続相を形成するものであり、成型加工性、透明性、耐熱性等に優れる材料であれば良く、例えばアクリル酸系、メタクリル酸系、ポリケイ皮酸ビニル系、環ゴム系等の反応性ビニル基を有する光硬化型レジスト材料、エポキシ樹脂、シリコーン樹脂(シリコーンゴム、シリコーンゲル等のオルガノポリシロキサン硬化物(架橋物)を含む)、ウレア樹脂、フッ素樹脂、ポリカーボネート樹脂、アクリル樹脂、ウレタン樹脂、メラミン樹脂、ポリビニル樹脂、ポリアミド樹脂、フェノール樹脂、ポリビニルアルコール樹脂、ポリビニルブチラール樹脂、セルロース樹脂、脂肪族エステル樹脂、芳香族エステル樹脂、脂肪族ポリオレフィン樹脂、芳香族ポリオレフィン樹脂などの公知のものを用いることができる。またこれらの共重合樹脂を用いても構わない。これらの樹脂を適宜設計することで、本発明の色変換組成物に有用な樹脂が得られる。
_これらの樹脂の中でも、フィルム化のプロセスが容易であることから熱硬化性樹脂がさらに好ましい。透明性、耐熱性などの観点から、エポキシ樹脂、シリコーン樹脂、アクリル樹脂、ポリエステル樹脂またはこれらの混合物を好適に用いることができる。
発光材料の劣化を防ぎ、耐久性の向上すなわち発光強度の経時的な低下の抑制を達成するために、特定の、3級アミン、カテコール誘導体およびニッケル化合物のうち少なくとも1つが有効である。
本発明の色変換組成物は、前記(A)、(B)、(C)成分以外に、酸化防止剤、加工および熱安定化剤、紫外線吸収剤等の耐光性安定化剤およびシランカップリング剤などを含有することができる。
本発明の色変換組成物は溶媒を含んでいてもよい。溶媒は、流動状態の樹脂の粘度を調整でき、発光物質の発光および耐久性に過度な影響を与えないものであれば、特に限定されない。例えば、水、2-プロパノール、エタノール、トルエン、メチルエチルケトン、メチルイソブチルケトン、シクロヘキサノン、ヘキサン、アセトン、テルピネオール、テキサノール、メチルセルソルブ、エチルセルソルブ、ブチルカルビトール、ブチルカルビトールアセテート、1-メトキシ-2-プロパノール、プロピレングリコールモノメチルエーテルアセテート等が挙げられ、これらの溶媒を2種類以上混合して使用することも可能である。これらの溶媒の中で特にトルエンは、一般式(1)で表される化合物の劣化に影響を与えず、乾燥後の残存溶媒が少ない点で好適に用いられる。
以下に、本発明の色変換組成物の製造方法の一例を説明する。前述した発光材料、バインダー樹脂、3級アミン、カテコール誘導体およびニッケル化合物の少なくとも1つ、溶剤等を所定量混合する。上記の成分を所定の組成になるよう混合した後、ホモジナイザー、自公転型攪拌機、3本ローラー、ボールミル、遊星式ボールミル、ビーズミル等の撹拌・混練機で均質に混合分散することで、色変換組成物が得られる。混合分散後、もしくは混合分散の過程で、真空もしくは減圧条件下で脱泡することも好ましく行われる。また、ある特定の成分を事前に混合することや、エージング等の処理をしても構わない。エバポレーターによって溶剤を除去して所望の固形分濃度にすることも可能である。
本発明において色変換フィルムは色変換組成物を硬化して得られる層を含んでいればその構成に限定はない。色変換フィルムの代表的な構造例として、図1に示すように、基材層10と、色変換組成物を硬化することにより得られる色変換層11との積層体、または、図2に示すように、色変換層11が複数の基材層10によって挟まれた積層体が挙げられる。色変換フィルムには、色変換層の酸素、水分や熱による劣化を防ぐために、さらに図3に示すようにバリアフィルム12を設けても良い。
基材層としては、特に制限無く公知の金属、フィルム、ガラス、セラミック、紙等を使用することができる。具体的には、アルミニウム(アルミニウム合金も含む)、亜鉛、銅、鉄などの金属板や箔、セルロースアセテート、ポリエチレンテレフタレート(PET)、ポリエチレン、ポリエステル、ポリアミド、ポリイミド、ポリフェニレンスルフィド、ポリスチレン、ポリプロピレン、ポリカーボネート、ポリビニルアセタール、アラミド、シリコーン、ポリオレフィン、熱可塑性フッ素樹脂で、テトラフルオロエチレンとエチレンの共重合体(ETFE)などのプラスチックのフィルム、α-ポリオレフィン樹脂、ポリカプロラクトン樹脂、アクリル樹脂、シリコーン樹脂およびこれらとエチレンの共重合樹脂からなるプラスチックのフィルム、前記プラスチックがラミネートされた紙、または前記プラスチックによりコーティングされた紙、前記金属がラミネートまたは蒸着された紙、前記金属がラミネートまたは蒸着されたプラスチックフィルムなどが挙げられる。また、基材が金属板の場合、表面にクロム系やニッケル系などのメッキ処理やセラミック処理されていてもよい。
次に、本発明の色変換フィルムの色変換層の製造方法の一例を説明する。上述した方法で作製した色変換組成物を基材上に塗布し、乾燥させる。塗布は、リバースロールコーター、ブレードコーター、スリットダイコーター、ダイレクトグラビアコーター、オフセットグラビアコーター、キスコーター、ナチュラルロールコーター、エアーナイフコーター、ロールブレードコーター、バリバーロールブレードコーター、トゥーストリームコーター、ロッドコーター、ワイヤーバーコーター、アプリケーター、ディップコーター、カーテンコーター、スピンコーター、ナイフコーター等により行うことができる。色変換層の膜厚均一性を得るためにはスリットダイコーターやディップコーターで塗布することが好ましい。
バリアフィルムとしては、色変換層に対してガスバリア性を向上する場合などにおいて適宜用いられ、例えば、酸化ケイ素、酸化アルミニウム、酸化チタン、酸化タンタル、酸化亜鉛、酸化スズ、酸化インジウム、酸化イットリウム、酸化マグネシウムなどの無機酸化物や、窒化ケイ素、窒化アルミニウム、窒化チタン、炭化窒化ケイ素などの無機窒化物、またはこれらの混合物、またはこれらに他の元素を添加した金属酸化物薄膜や金属窒化物薄膜、あるいはポリ塩化ビニリデン、アクリル系樹脂、シリコン系樹脂、メラミン系樹脂、ウレタン系樹脂、フッ素系樹脂、酢酸ビニルのけん化物等のポリビニルアルコール系樹脂などの各種樹脂から成る膜を挙げることができる。また、水分に対してバリア機能を有する膜としては、例えば、ポリエチレン、ポリプロピレン、ナイロン、ポリ塩化ビニリデン、塩化ビニリデンと塩化ビニル、塩化ビニリデンとアクリロニトリルの共重合物、フッ素系樹脂、酢酸ビニルのけん化物等のポリビニルアルコール系樹脂などの各種樹脂から成る膜を挙げることができる。
励起光の種類は、一般式(1)で表される化合物等の混合する発光物質が吸収可能な波長領域に発光を示すものであればいずれの励起光でも用いることができる。例えば、熱陰極管や冷陰極管、無機ELなどの蛍光性光源、有機エレクトロルミネッセンス素子光源、LED光源、白熱光源、あるいは太陽光などいずれの励起光でも原理的には利用可能であるが、中でもLEDが好適な励起光であり、ディスプレイや照明用途では、青色光の色純度を高められる点で、430~500nmの範囲の励起光を持つ青色LEDがさらに好適な励起光である。励起光の波長範囲がこれより長波長側にあると青色光が欠如するために白色光が形成できなくなり、また励起光の波長範囲がこれより短波長側にあると、一般式(1)で表される化合物等の発光物質あるいはバインダー樹脂等の有機化合物が光劣化しやすいので好ましくない。
本発明における光源ユニットは、少なくとも光源および色変換組成物または色変換フィルムを含む構成である。色変換組成物を含む場合は、光源と色変換組成物の配置方法については特に限定されず、光源に色変換組成物を直接塗布した構成を取っても良いし、光源とは離したフィルムやガラスなどに色変換組成物を塗布した構成を取っても良い。色変換フィルムを含む場合は、光源と色変換フィルムの配置方法については特に限定されず、光源と色変換フィルムを密着させた構成を取っても良いし、光源と色変換フィルムを離したリモートフォスファー形式を取っても良い。また、色純度を高める目的で、さらにカラーフィルターを含む構成を取っても良い。
化合物の1H-NMRは、超伝導FTNMR EX-270(日本電子(株)製)を用い、重クロロホルム溶液にて測定を行った。
化合物の吸収スペクトルは、U-3200形分光光度計(日立製作所(株)製)を用い、化合物をトルエンに1×10-6mol/Lの濃度で溶解させて測定を行った。
化合物の蛍光スペクトルは、F-2500形分光蛍光光度計(日立製作所(株)製)を用い、化合物をトルエンに1×10-6mol/Lの濃度で溶解させ、波長460nmで励起させた際の蛍光スペクトルを測定した。
各色変換フィルムおよび青色LED素子(ProLight社製;型番PM2B-3LBE-SD、発光ピーク波長:460nm)を搭載した発光装置に、10mAの電流を流してLEDを点灯させ、分光放射輝度計(CS-1000、コニカミノルタ社製)を用いて、発光スペクトル、ピーク波長における発光強度および色度を測定した。なお、各色変換フィルムと青色LED素子との距離を3cmとした。
各色変換フィルムおよび青色LED素子(ProLight社製;型番PM2B-3LBE-SD、発光ピーク波長:460nm)を搭載した発光装置に、10mAの電流を流してLEDチップを点灯させ、分光放射輝度計(CS-1000、コニカミノルタ社製)を用いて初期輝度を測定した。なお、各色変換フィルムと青色LED素子との距離を3cmとした。その後、室温下で青色LED素子からの光を連続照射し、輝度が一定量低下するまでの時間を観測することで、光耐久性を評価した。
上記、吸収スペクトルの測定方法と同様に、化合物をトルエンあるいはエタノールに1×10-5mol/L、5×10-5mol/L、1×10-4mol/L、5×10-4mol/L、1×10-3mol/Lの各濃度で溶解させて、それぞれの吸収スペクトルの測定を行った。得られた吸収スペクトルから、各波長における吸光度を算出し、縦軸に吸光度、横軸にモル濃度(mol/L)をとったグラフから、検量線を作成して、波長毎のモル吸光係数を求めた。
化合物G-1の合成方法
3,5-ジブロモベンズアルデヒド(3.0g)、4-t-ブチルフェニルボロン酸(5.3g)、テトラキス(トリフェニルホスフィン)パラジウム(0)(0.4g)、炭酸カリウム(2.0g)をフラスコに入れ、窒素置換した。ここに脱気したトルエン(30mL)および脱気した水(10mL)を加え、4時間還流した。反応溶液を室温まで冷却し、有機層を、分液した後に飽和食塩水で洗浄した。この有機層を硫酸マグネシウムで乾燥し、ろ過後、溶媒を留去した。得られた反応生成物をシリカゲルクロマトグラフィーにより精製し、3,5-ビス(4-t-ブチルフェニル)ベンズアルデヒド(3.5g)を白色固体として得た。
化合物R-1の合成方法
4-(4-t-ブチルフェニル)-2-(4-メトキシフェニル)ピロール300mg、2-メトキシベンゾイルクロリド201mgとトルエン10mlの混合溶液を窒素気流下、120℃で6時間加熱した。室温に冷却後、エバポレートした。エタノール20mlで洗浄し、真空乾燥した後、2-(2-メトキシベンゾイル)-3-(4-t-ブチルフェニル)-5-(4-メトキシフェニル)ピロール260mgを得た。
1H-NMR(CDCl3,ppm):1.19(s,18H)、3.42(s,3H)、3.85(s,6H)、5.72(d,1H)、6.20(t,1H)、6.42-6.97(m,16H),7.89(d,4H)。
シリコーン樹脂“OE-6630A/B”(東レ・ダウコーニング社製)100重量部に対して、(A)成分として化合物G-1を0.20重量部、硬化剤0.5を重量部、(C)成分として化合物Q-1を1.0重量部混合した後、遊星式撹拌・脱泡装置“マゼルスターKK-400”(クラボウ製)を用い、1000rpmで20分間撹拌・脱泡してフィルム作製用樹脂液としての色変換組成物を得た。
(A)成分および(C)成分として表3に記載した化合物を用いた以外は、実施例1と同様にして色変換フィルムを作製して評価した。結果を表3に示す。ただし、表中の発光強度(相対値)は、比較例1における強度を1.00としたときの相対値である。
二液型熱硬化性エポキシ系アクリル樹脂100重量部に対して、(A)成分として化合物R-1を0.08重量部、硬化剤を0.5重量部、(C)成分として化合物Q-1を1.0重量部混合した後、300rpmで1時間攪拌し、組成物を製造した。
(A)成分および(C)成分として表4に記載した化合物を用いた以外は、実施例12と同様にして色変換フィルムを作製して評価した。結果を表4に示す。ただし、表中の発光強度(相対値)は、比較例11における強度を1.00としたときの相対値である。
二液型熱硬化性エポキシ系アクリル樹脂100重量部に対して、(A)成分として化合物G-1を0.20重量部、化合物R-1を0.08重量部、硬化剤を0.5重量部、(C)成分として化合物Q-1を1.0重量部混合した後、300rpmで1時間攪拌し、組成物を製造した。
(A)成分および(C)成分として表5に記載した化合物を用いた以外は、実施例22と同様にして色変換フィルムを作製して評価した。結果を表5に示す。ただし、表中の発光強度(相対値)は、比較例18における強度を1.00としたときの相対値である。
アクリル樹脂“KC-7000”(共栄社化学(株)製)100重量部に対して、(A)成分として化合物G-1を0.25重量部、(C)成分として化合物Q-1を1.0重量部混合した後、300rpmで1時間攪拌し、組成物を製造した。
(A)成分および(C)成分として表6に記載した化合物を用いた以外は、実施例27と同様にして色変換フィルムを作製して評価した。結果を表6に示す。ただし、表中の発光強度(相対値)は、比較例22における強度を1.00としたときの相対値である。
アクリル樹脂“KC-7000”(共栄社化学(株)製)100重量部に対して、(A)成分として化合物G-7を0.25重量部、(C)成分として化合物Q-1を1.0重量部および化合物Q-2を1.0重量部混合した後、300rpmで1時間攪拌し、組成物を製造した。
(A)成分および(C)成分として表7に記載した化合物を用いた以外は、実施例34と同様にして色変換フィルムを作製して評価した。結果を表7に示す。ただし、表中の発光強度(相対値)は、後述の比較例23における強度を1.00としたときの相対値である。
アクリル樹脂“KC-7000”(共栄社化学(株)製)100重量部に対して、(A)成分として表7に記載した化合物を0.25重量部、(C)成分として表7にフィルムを作製して評価した。結果を表7に示す。ただし、表中の発光強度(相対値)は、比較例23における強度を1.00としたときの相対値である。
10 基材層
11 色変換層
12 バリアフィルム
Claims (21)
- 入射光を、その入射光よりも長波長の光に変換する色変換組成物であって、以下の(A)~(C)成分;
(A)少なくとも1種の発光材料
(B)バインダー樹脂
(C)3級アミン、カテコール誘導体およびニッケル化合物のうち少なくとも1つ
を含有し、
前記3級アミン、カテコール誘導体およびニッケル化合物は、波長400nm以上800nm以下の波長域全域でモル吸光係数εが100以下である、
色変換組成物。 - (C)成分がニッケル化合物を含有し、前記ニッケル化合物が、有機酸のニッケル塩、アセチルアセトナート系ニッケル錯体およびチオビスフェノレート系ニッケル錯体のうち少なくとも1つである、請求項1記載の色変換組成物。
- (C)成分がニッケル化合物を含有し、前記ニッケル化合物の含有量が(B)成分100重量部に対して1.0×10-2重量部以上10重量部以下である、請求項1または2に記載の色変換組成物。
- (C)成分が3級アミンを含有し、前記3級アミンの窒素原子上の置換基の少なくとも1つが置換もしくは無置換のアルキル基である、請求項1~3いずれかに記載の色変換組成物。
- (C)成分の含有量が(B)成分100重量部に対して10-2重量部以上15重量部以下である、請求項1~4いずれかに記載の色変換組成物。
- 前記発光材料が、波長430nm以上500nm以下の範囲の励起光を用いることによりピーク波長が500nm以上580nm以下の発光を呈する発光材料を含有する、請求項1~5のいずれかに記載の色変換組成物。
- 前記発光材料が、下記の発光材料(a)および(b)を含有する、請求項1~6のいずれかに記載の色変換組成物。
(a)波長430nm以上500nm以下の範囲の励起光を用いることによりピーク波長が500nm以上580nm以下の発光を呈する発光材料
(b)波長430nm以上500nm以下の範囲の励起光または発光材料(a)からの発光のいずれかまたは両方により励起されることにより、ピーク波長が580nm以上750nm以下の発光を呈する発光材料 - 前記発光材料(a)の含有量waと、発光材料(b)の含有量wbが、wa≧wbの関係である、請求項7記載の色変換組成物。
- 前記発光材料が一般式(1)で表される化合物を含有する、請求項1~8いずれかに記載の色変換組成物。
- 一般式(1)において、XがC-R7であり、R7が置換もしくは無置換のアリール基である、請求項9記載の色変換組成物。
- 一般式(1)において、XがC-R7であり、R7が置換もしくは無置換のフェニル基である、請求項9または10記載の色変換組成物。
- 前記発光材料(a)が、一般式(1)で表される化合物である、請求項6~11いずれか記載の色変換組成物。
- 前記発光材料(b)が、一般式(1)で表される化合物である、請求項7~12いずれか記載の色変換組成物。
- さらに溶媒を含有する、請求項1~13いずれか記載の色変換組成物。
- 前記バインダー樹脂が熱硬化性樹脂であることを特徴とする、請求項1~14記載の色変換組成物。
- 請求項1~15いずれか記載の色変換組成物を硬化して得られる層を含む、色変換フィルム。
- ガスバリア層により被覆されてなる、請求項16記載の色変換フィルム。
- 光源および請求項16または17記載の色変換フィルムを含む、光源ユニット。
- 光源が430nm以上500nm以下の範囲に極大発光を有する発光ダイオードである、請求項18記載の光源ユニット。
- 請求項18または19記載の光源ユニットを含む、ディスプレイ。
- 請求項18または19記載の光源ユニットを含む、照明。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201680041709.3A CN107922835B (zh) | 2015-07-17 | 2016-07-08 | 颜色转换组合物、颜色转换膜以及包含其的背光单元、显示器及照明 |
| JP2016549588A JP6866643B2 (ja) | 2015-07-17 | 2016-07-08 | 色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 |
| SG11201800454TA SG11201800454TA (en) | 2015-07-17 | 2016-07-08 | Color conversion composition, color conversion film and backlight unit, display and lighting device each comprising same |
| KR1020187001253A KR102344621B1 (ko) | 2015-07-17 | 2016-07-08 | 색변환 조성물, 색변환 필름 그리고 그것을 포함하는 백라이트 유닛, 디스플레이 및 조명 |
| US15/745,323 US10800970B2 (en) | 2015-07-17 | 2016-07-08 | Color conversion composition, color conversion film and backlight unit, display and lighting device each comprising same |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015142696 | 2015-07-17 | ||
| JP2015-142696 | 2015-07-17 | ||
| JP2015-142697 | 2015-07-17 | ||
| JP2015142697 | 2015-07-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017014068A1 true WO2017014068A1 (ja) | 2017-01-26 |
Family
ID=57835060
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/070255 WO2017014068A1 (ja) | 2015-07-17 | 2016-07-08 | 色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US10800970B2 (ja) |
| JP (1) | JP6866643B2 (ja) |
| KR (1) | KR102344621B1 (ja) |
| CN (1) | CN107922835B (ja) |
| SG (1) | SG11201800454TA (ja) |
| TW (1) | TWI740835B (ja) |
| WO (1) | WO2017014068A1 (ja) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017104581A1 (ja) * | 2015-12-18 | 2017-06-22 | 東レ株式会社 | 色変換組成物、色変換フィルム、それを含む光源ユニット、ディスプレイおよび照明装置 |
| JP2019026778A (ja) * | 2017-08-01 | 2019-02-21 | Dic株式会社 | インク組成物及びその製造方法、光変換層並びにカラーフィルタ |
| WO2019039142A1 (ja) * | 2017-08-21 | 2019-02-28 | 東レ株式会社 | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイおよび照明装置 |
| JPWO2018155253A1 (ja) * | 2017-02-23 | 2019-12-12 | 東レ株式会社 | 蛍光体シート、それを用いたledチップおよびledパッケージ、ledパッケージの製造方法、ならびにledパッケージを含む発光装置、バックライトユニットおよびディスプレイ |
| WO2021192795A1 (ja) | 2020-03-23 | 2021-09-30 | 東レ株式会社 | 色変換組成物、色変換フィルム、それを含む光源ユニット、ディスプレイおよび照明ならびに化合物 |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017014068A1 (ja) | 2015-07-17 | 2017-01-26 | 東レ株式会社 | 色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 |
| FR3046610B1 (fr) * | 2016-01-08 | 2020-02-21 | Crime Science Technology | Utilisation de 4,4-difluoro-4-bora-3a,4a-diaza-s-indacenes pour la securisation |
| US10604697B2 (en) * | 2016-02-19 | 2020-03-31 | Toray Industries, Inc. | Color conversion sheet, light source unit including the same, display, and lighting apparatus |
| JP6447557B2 (ja) * | 2016-03-24 | 2019-01-09 | 日亜化学工業株式会社 | 発光装置の製造方法 |
| US11340390B2 (en) * | 2017-06-02 | 2022-05-24 | Toray Industries, Inc. | Color conversion sheet, light source unit comprising same, display and lighting device |
| KR102157933B1 (ko) * | 2017-12-27 | 2020-09-18 | 주식회사 엘지화학 | 함질소 고리 화합물 및 이를 포함하는 색변환 필름 |
| JP6891381B2 (ja) | 2018-04-30 | 2021-06-18 | エルジー・ケム・リミテッド | 化合物、これを含む色変換フィルム、バックライトユニットおよびディスプレイ装置 |
| EP3810719B1 (en) | 2018-06-22 | 2022-05-11 | Basf Se | Photostable cyano-substituted boron-dipyrromethene dye as green emitter for display and illumination applications |
| WO2019243288A1 (en) | 2018-06-22 | 2019-12-26 | Basf Se | Extruded pet films with photostable dyes for color conversion |
| WO2020097596A1 (en) * | 2018-11-09 | 2020-05-14 | Li, Ying | Lighting device for horticulture |
| WO2020101299A1 (ko) | 2018-11-12 | 2020-05-22 | 주식회사 엘지화학 | 색변환 필름, 이를 포함하는 백라이트 유닛 및 디스플레이 장치 |
| TW202024282A (zh) | 2018-11-12 | 2020-07-01 | 南韓商Lg化學股份有限公司 | 色彩轉換膜、製備其的方法、包含其的背光單元以及顯示器裝置 |
| KR102388486B1 (ko) | 2019-02-28 | 2022-04-19 | 주식회사 엘지화학 | 화합물, 이를 포함하는 색변환 필름, 백라이트 유닛 및 디스플레이 장치 |
| JP7424594B2 (ja) * | 2020-03-04 | 2024-01-30 | エルジー・ケム・リミテッド | 光学フィルム形成用組成物、光学フィルムおよびこれを含むディスプレイ装置 |
| CN113321671A (zh) * | 2021-01-29 | 2021-08-31 | 南京工业大学 | 一种氟硼二吡咯类固态发光材料及其制备方法和应用、一种蓝光驱动的led |
| CN114460052B (zh) * | 2022-01-11 | 2023-06-20 | 武汉理工大学 | 一种基于荧光碳量子点直接检测丙酮酸钠浓度的方法 |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000256565A (ja) * | 1999-03-08 | 2000-09-19 | Idemitsu Kosan Co Ltd | 蛍光変換膜用樹脂組成物、蛍光変換膜およびカラー化有機エレクトロルミネッセンス素子 |
| JP2002184576A (ja) * | 2000-12-15 | 2002-06-28 | Fuji Electric Co Ltd | 色変換フィルタ基板、および色変換フィルタ基板を具備する色変換カラーディスプレイ |
| WO2003012005A1 (en) * | 2001-08-02 | 2003-02-13 | Ente Per Le Nuove Tecnologie, L'energia E L'ambiente (Enea) | Stabilization of luminescence from organic materials with compounds of phenolic origin |
| JP2008501066A (ja) * | 2004-05-18 | 2008-01-17 | アイファイアー・テクノロジー・コープ | 改良された色変換発光膜 |
| WO2009116456A1 (ja) * | 2008-03-19 | 2009-09-24 | 東レ株式会社 | 発光素子材料及び発光素子 |
| JP2010061824A (ja) * | 2008-09-01 | 2010-03-18 | Toray Ind Inc | 色変換組成物 |
| JP2011241160A (ja) * | 2010-05-17 | 2011-12-01 | Yamamoto Chem Inc | 色変換材料、該材料を含む組成物、該組成物を使用した色変換光学部品および該色変換光学部品を使用した発光素子 |
Family Cites Families (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5446157A (en) | 1990-04-23 | 1995-08-29 | Morgan; Lee R. | Boron difluoride compounds useful in photodynamic therapy and production of laser light |
| US5189029A (en) | 1990-04-23 | 1993-02-23 | Bo-Dekk Ventures, Ltd. | Indacene compounds and methods for using the same |
| US6110566A (en) * | 1997-10-23 | 2000-08-29 | 3M Innovative Properties Company | Stabilization of fluorescent dyes in vinyl articles using hindered amine light stabilizers |
| JP2000212554A (ja) | 1998-11-20 | 2000-08-02 | Idemitsu Kosan Co Ltd | 蛍光変換媒体及びそれを用いた表示装置 |
| JP4005749B2 (ja) | 1999-12-13 | 2007-11-14 | 出光興産株式会社 | 色変換膜及び有機エレクトロルミネッセンス素子 |
| KR100804336B1 (ko) | 2000-11-16 | 2008-02-15 | 후지쯔 가부시끼가이샤 | 잠상용 인쇄 잉크 조성물, 이 잉크 조성물에 의해 기록된잠상을 포함하는 인쇄물, 잠상 정보에 의한 판독 방법 및잠상 정보 판독 장치 |
| JP4948713B2 (ja) | 2001-04-19 | 2012-06-06 | 三井化学株式会社 | 色変換材料、該材料を含む組成物、該組成物を使用した色変換光学部品および該色変換光学部品を使用した発光素子 |
| JP4948714B2 (ja) | 2001-05-24 | 2012-06-06 | 三井化学株式会社 | 色変換材料、該材料を含む組成物、該組成物を使用した色変換光学部品および該色変換光学部品を使用した発光素子 |
| EP1816178A1 (en) * | 2006-02-01 | 2007-08-08 | POLYNT GmbH & Co. KG | Molding compounds containing luminescent pigments |
| JP5221859B2 (ja) | 2006-03-09 | 2013-06-26 | 株式会社Adeka | クマリン化合物を含有してなるフィルム、クマリン化合物とマトリクスを含む色変換層、該色変換層を含む色変換フィルタ、補色層、補色フィルタならびに多色発光デバイス |
| WO2010098098A1 (ja) | 2009-02-27 | 2010-09-02 | 出光興産株式会社 | ピロメテンホウ素錯体化合物及びそれを用いた有機電界発光素子 |
| JP5489280B2 (ja) * | 2010-04-07 | 2014-05-14 | 信越化学工業株式会社 | 光半導体封止用エポキシ組成物 |
| CN102905513A (zh) * | 2010-05-28 | 2013-01-30 | 旭硝子株式会社 | 波长转换膜 |
| JP2012022028A (ja) | 2010-07-12 | 2012-02-02 | Ns Materials Kk | 液晶ディスプレイ |
| EP2487218A1 (en) * | 2011-02-09 | 2012-08-15 | Koninklijke Philips Electronics N.V. | Polymeric matrix with organic phosphor and manufactory thereof |
| JP5527269B2 (ja) | 2011-04-06 | 2014-06-18 | セイコーエプソン株式会社 | インク組成物、インクジェット記録方法、及び記録物 |
| JP6279209B2 (ja) * | 2013-01-17 | 2018-02-14 | 山本化成株式会社 | 波長変換層、及びこれを用いた波長変換フィルタ |
| EP3059288B1 (en) | 2013-10-17 | 2021-12-22 | DIC Corporation | Resin composition and molded article |
| SG11201709751SA (en) | 2015-05-26 | 2017-12-28 | Toray Industries | Pyrromethene-boron complex, color-changing composition, color-changing film, light source unit including same, display, and lighting |
| JP2017011153A (ja) | 2015-06-24 | 2017-01-12 | パナソニックIpマネジメント株式会社 | 波長制御光学部材、発光装置及び照明器具 |
| WO2017014068A1 (ja) | 2015-07-17 | 2017-01-26 | 東レ株式会社 | 色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 |
-
2016
- 2016-07-08 WO PCT/JP2016/070255 patent/WO2017014068A1/ja active Application Filing
- 2016-07-08 CN CN201680041709.3A patent/CN107922835B/zh active Active
- 2016-07-08 US US15/745,323 patent/US10800970B2/en active Active
- 2016-07-08 JP JP2016549588A patent/JP6866643B2/ja active Active
- 2016-07-08 KR KR1020187001253A patent/KR102344621B1/ko active IP Right Grant
- 2016-07-08 SG SG11201800454TA patent/SG11201800454TA/en unknown
- 2016-07-15 TW TW105122308A patent/TWI740835B/zh active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2000256565A (ja) * | 1999-03-08 | 2000-09-19 | Idemitsu Kosan Co Ltd | 蛍光変換膜用樹脂組成物、蛍光変換膜およびカラー化有機エレクトロルミネッセンス素子 |
| JP2002184576A (ja) * | 2000-12-15 | 2002-06-28 | Fuji Electric Co Ltd | 色変換フィルタ基板、および色変換フィルタ基板を具備する色変換カラーディスプレイ |
| WO2003012005A1 (en) * | 2001-08-02 | 2003-02-13 | Ente Per Le Nuove Tecnologie, L'energia E L'ambiente (Enea) | Stabilization of luminescence from organic materials with compounds of phenolic origin |
| JP2008501066A (ja) * | 2004-05-18 | 2008-01-17 | アイファイアー・テクノロジー・コープ | 改良された色変換発光膜 |
| WO2009116456A1 (ja) * | 2008-03-19 | 2009-09-24 | 東レ株式会社 | 発光素子材料及び発光素子 |
| JP2010061824A (ja) * | 2008-09-01 | 2010-03-18 | Toray Ind Inc | 色変換組成物 |
| JP2011241160A (ja) * | 2010-05-17 | 2011-12-01 | Yamamoto Chem Inc | 色変換材料、該材料を含む組成物、該組成物を使用した色変換光学部品および該色変換光学部品を使用した発光素子 |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017104581A1 (ja) * | 2015-12-18 | 2017-06-22 | 東レ株式会社 | 色変換組成物、色変換フィルム、それを含む光源ユニット、ディスプレイおよび照明装置 |
| JPWO2018155253A1 (ja) * | 2017-02-23 | 2019-12-12 | 東レ株式会社 | 蛍光体シート、それを用いたledチップおよびledパッケージ、ledパッケージの製造方法、ならびにledパッケージを含む発光装置、バックライトユニットおよびディスプレイ |
| JP2019026778A (ja) * | 2017-08-01 | 2019-02-21 | Dic株式会社 | インク組成物及びその製造方法、光変換層並びにカラーフィルタ |
| JP7013705B2 (ja) | 2017-08-01 | 2022-02-01 | Dic株式会社 | インク組成物及びその製造方法、光変換層並びにカラーフィルタ |
| WO2019039142A1 (ja) * | 2017-08-21 | 2019-02-28 | 東レ株式会社 | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイおよび照明装置 |
| KR20200034949A (ko) | 2017-08-21 | 2020-04-01 | 도레이 카부시키가이샤 | 색 변환 조성물, 색 변환 시트, 및 그것을 포함하는 광원 유닛, 디스플레이 및 조명 장치 |
| JPWO2019039142A1 (ja) * | 2017-08-21 | 2020-08-06 | 東レ株式会社 | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイおよび照明装置 |
| JP7163773B2 (ja) | 2017-08-21 | 2022-11-01 | 東レ株式会社 | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイおよび照明装置 |
| KR102629175B1 (ko) * | 2017-08-21 | 2024-01-26 | 도레이 카부시키가이샤 | 색 변환 조성물, 색 변환 시트, 및 그것을 포함하는 광원 유닛, 디스플레이 및 조명 장치 |
| WO2021192795A1 (ja) | 2020-03-23 | 2021-09-30 | 東レ株式会社 | 色変換組成物、色変換フィルム、それを含む光源ユニット、ディスプレイおよび照明ならびに化合物 |
| KR20220158226A (ko) | 2020-03-23 | 2022-11-30 | 도레이 카부시키가이샤 | 색변환 조성물, 색변환 필름, 그것을 포함하는 광원 유닛, 디스플레이 및 조명 그리고 화합물 |
| EP4130009A4 (en) * | 2020-03-23 | 2023-12-13 | Toray Industries, Inc. | COLOR CONVERSION COMPOSITION, COLOR CONVERSION FILM, LIGHT SOURCE UNIT, DISPLAY AND ILLUMINATION THEREOF AND CONNECTION |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20180030829A (ko) | 2018-03-26 |
| JP6866643B2 (ja) | 2021-04-28 |
| SG11201800454TA (en) | 2018-03-28 |
| CN107922835B (zh) | 2021-01-08 |
| US10800970B2 (en) | 2020-10-13 |
| TW201708500A (zh) | 2017-03-01 |
| JPWO2017014068A1 (ja) | 2018-04-26 |
| TWI740835B (zh) | 2021-10-01 |
| CN107922835A (zh) | 2018-04-17 |
| US20180208838A1 (en) | 2018-07-26 |
| KR102344621B1 (ko) | 2021-12-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2017014068A1 (ja) | 色変換組成物、色変換フィルムならびにそれを含むバックライトユニット、ディスプレイおよび照明 | |
| JP6299870B2 (ja) | ピロメテンホウ素錯体、色変換組成物、色変換フィルムならびにそれを含む光源ユニット、ディスプレイおよび照明 | |
| JP6760076B2 (ja) | 色変換フィルムならびにそれを含む光源ユニット、ディスプレイおよび照明装置 | |
| JP6380653B2 (ja) | 色変換シート、それを含む光源ユニット、ディスプレイおよび照明装置 | |
| JP6210169B2 (ja) | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイ、照明装置、バックライトユニット、ledチップおよびledパッケージ | |
| JP6939787B2 (ja) | 色変換シート、それを含む光源ユニット、ディスプレイおよび照明装置 | |
| JP6690721B2 (ja) | 色変換組成物、色変換フィルムならびにそれを含む光源ユニット、ディスプレイおよび照明 | |
| JP6693578B2 (ja) | ピロメテンホウ素錯体、色変換組成物、色変換フィルム、光源ユニット、ディスプレイ、照明装置および発光素子 | |
| JP6737010B2 (ja) | 色変換シート、それを含む光源ユニット、ディスプレイおよび照明 | |
| WO2017104581A1 (ja) | 色変換組成物、色変換フィルム、それを含む光源ユニット、ディスプレイおよび照明装置 | |
| JP2019219512A (ja) | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイおよび照明装置 | |
| JP7359151B2 (ja) | ピロメテンホウ素錯体、色変換組成物、色変換フィルム、光源ユニット、ディスプレイおよび照明装置 | |
| JP7163773B2 (ja) | 色変換組成物、色変換シートならびにそれを含む光源ユニット、ディスプレイおよび照明装置 | |
| JP7306265B2 (ja) | 色変換組成物、色変換シート、光源ユニット、ディスプレイおよび照明装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2016549588 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16827630 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20187001253 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15745323 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11201800454T Country of ref document: SG |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 16827630 Country of ref document: EP Kind code of ref document: A1 |