WO2016047569A1 - 鑑査支援システム、錠剤分包装置 - Google Patents
鑑査支援システム、錠剤分包装置 Download PDFInfo
- Publication number
- WO2016047569A1 WO2016047569A1 PCT/JP2015/076587 JP2015076587W WO2016047569A1 WO 2016047569 A1 WO2016047569 A1 WO 2016047569A1 JP 2015076587 W JP2015076587 W JP 2015076587W WO 2016047569 A1 WO2016047569 A1 WO 2016047569A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- tablet
- inspection
- image
- unit
- packaging
- Prior art date
Links
Images
Classifications
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H40/00—ICT specially adapted for the management or administration of healthcare resources or facilities; ICT specially adapted for the management or operation of medical equipment or devices
- G16H40/60—ICT specially adapted for the management or administration of healthcare resources or facilities; ICT specially adapted for the management or operation of medical equipment or devices for the operation of medical equipment or devices
- G16H40/67—ICT specially adapted for the management or administration of healthcare resources or facilities; ICT specially adapted for the management or operation of medical equipment or devices for the operation of medical equipment or devices for remote operation
-
- G—PHYSICS
- G06—COMPUTING; CALCULATING OR COUNTING
- G06Q—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR ADMINISTRATIVE, COMMERCIAL, FINANCIAL, MANAGERIAL OR SUPERVISORY PURPOSES; SYSTEMS OR METHODS SPECIALLY ADAPTED FOR ADMINISTRATIVE, COMMERCIAL, FINANCIAL, MANAGERIAL OR SUPERVISORY PURPOSES, NOT OTHERWISE PROVIDED FOR
- G06Q20/00—Payment architectures, schemes or protocols
- G06Q20/38—Payment protocols; Details thereof
- G06Q20/40—Authorisation, e.g. identification of payer or payee, verification of customer or shop credentials; Review and approval of payers, e.g. check credit lines or negative lists
- G06Q20/401—Transaction verification
- G06Q20/4014—Identity check for transactions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61J—CONTAINERS SPECIALLY ADAPTED FOR MEDICAL OR PHARMACEUTICAL PURPOSES; DEVICES OR METHODS SPECIALLY ADAPTED FOR BRINGING PHARMACEUTICAL PRODUCTS INTO PARTICULAR PHYSICAL OR ADMINISTERING FORMS; DEVICES FOR ADMINISTERING FOOD OR MEDICINES ORALLY; BABY COMFORTERS; DEVICES FOR RECEIVING SPITTLE
- A61J3/00—Devices or methods specially adapted for bringing pharmaceutical products into particular physical or administering forms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61J—CONTAINERS SPECIALLY ADAPTED FOR MEDICAL OR PHARMACEUTICAL PURPOSES; DEVICES OR METHODS SPECIALLY ADAPTED FOR BRINGING PHARMACEUTICAL PRODUCTS INTO PARTICULAR PHYSICAL OR ADMINISTERING FORMS; DEVICES FOR ADMINISTERING FOOD OR MEDICINES ORALLY; BABY COMFORTERS; DEVICES FOR RECEIVING SPITTLE
- A61J7/00—Devices for administering medicines orally, e.g. spoons; Pill counting devices; Arrangements for time indication or reminder for taking medicine
- A61J7/0076—Medicament distribution means
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61J—CONTAINERS SPECIALLY ADAPTED FOR MEDICAL OR PHARMACEUTICAL PURPOSES; DEVICES OR METHODS SPECIALLY ADAPTED FOR BRINGING PHARMACEUTICAL PRODUCTS INTO PARTICULAR PHYSICAL OR ADMINISTERING FORMS; DEVICES FOR ADMINISTERING FOOD OR MEDICINES ORALLY; BABY COMFORTERS; DEVICES FOR RECEIVING SPITTLE
- A61J7/00—Devices for administering medicines orally, e.g. spoons; Pill counting devices; Arrangements for time indication or reminder for taking medicine
- A61J7/04—Arrangements for time indication or reminder for taking medicine, e.g. programmed dispensers
- A61J7/0409—Arrangements for time indication or reminder for taking medicine, e.g. programmed dispensers with timers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65B—MACHINES, APPARATUS OR DEVICES FOR, OR METHODS OF, PACKAGING ARTICLES OR MATERIALS; UNPACKING
- B65B35/00—Supplying, feeding, arranging or orientating articles to be packaged
- B65B35/10—Feeding, e.g. conveying, single articles
- B65B35/14—Feeding, e.g. conveying, single articles by agitators or vibrators
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65B—MACHINES, APPARATUS OR DEVICES FOR, OR METHODS OF, PACKAGING ARTICLES OR MATERIALS; UNPACKING
- B65B35/00—Supplying, feeding, arranging or orientating articles to be packaged
- B65B35/10—Feeding, e.g. conveying, single articles
- B65B35/26—Feeding, e.g. conveying, single articles by rotary conveyors
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B65—CONVEYING; PACKING; STORING; HANDLING THIN OR FILAMENTARY MATERIAL
- B65B—MACHINES, APPARATUS OR DEVICES FOR, OR METHODS OF, PACKAGING ARTICLES OR MATERIALS; UNPACKING
- B65B57/00—Automatic control, checking, warning, or safety devices
-
- G—PHYSICS
- G07—CHECKING-DEVICES
- G07F—COIN-FREED OR LIKE APPARATUS
- G07F17/00—Coin-freed apparatus for hiring articles; Coin-freed facilities or services
- G07F17/0092—Coin-freed apparatus for hiring articles; Coin-freed facilities or services for assembling and dispensing of pharmaceutical articles
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H10/00—ICT specially adapted for the handling or processing of patient-related medical or healthcare data
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H10/00—ICT specially adapted for the handling or processing of patient-related medical or healthcare data
- G16H10/60—ICT specially adapted for the handling or processing of patient-related medical or healthcare data for patient-specific data, e.g. for electronic patient records
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H20/00—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance
- G16H20/10—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance relating to drugs or medications, e.g. for ensuring correct administration to patients
- G16H20/13—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance relating to drugs or medications, e.g. for ensuring correct administration to patients delivered from dispensers
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H40/00—ICT specially adapted for the management or administration of healthcare resources or facilities; ICT specially adapted for the management or operation of medical equipment or devices
- G16H40/60—ICT specially adapted for the management or administration of healthcare resources or facilities; ICT specially adapted for the management or operation of medical equipment or devices for the operation of medical equipment or devices
- G16H40/63—ICT specially adapted for the management or administration of healthcare resources or facilities; ICT specially adapted for the management or operation of medical equipment or devices for the operation of medical equipment or devices for local operation
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16H—HEALTHCARE INFORMATICS, i.e. INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR THE HANDLING OR PROCESSING OF MEDICAL OR HEALTHCARE DATA
- G16H20/00—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance
- G16H20/10—ICT specially adapted for therapies or health-improving plans, e.g. for handling prescriptions, for steering therapy or for monitoring patient compliance relating to drugs or medications, e.g. for ensuring correct administration to patients
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P90/00—Enabling technologies with a potential contribution to greenhouse gas [GHG] emissions mitigation
- Y02P90/30—Computing systems specially adapted for manufacturing
Definitions
- the present invention relates to an inspection support system and a tablet packaging device that support inspection of a dispensing performed based on prescription data.
- a tablet package comprising a plurality of tablet cassettes for storing predetermined types of tablets, and capable of packaging the tablets stored in each tablet cassette in dosage units based on prescription data.
- An apparatus is known (see, for example, Patent Document 1).
- the tablet packaging device may be provided with a handing unit for dispensing tablets, which are put in a plurality of squares arranged in a matrix, in dosage units (see, for example, Patent Document 1). .
- an inspection operation is performed by a pharmacist to confirm whether or not the tablets packaged by the tablet packaging device are appropriate for the prescription data.
- An object of the present invention is to provide an inspection support system and a tablet packaging device that can support an inspection operation of a pharmacist.
- the inspection support system according to the present invention was photographed before a tablet dispensed from one or both of a tablet cassette and a hand-rolling unit based on prescription data was packaged with a packaging material in a tablet packaging device.
- the photographed image of the tablet is displayed in a package unit (single package unit), and the result of the inspection process executed based on the identification information of the tablet and the prescription data included in the photographed image of the tablet is displayed.
- An inspection display processing unit is provided.
- FIG. 1 is a diagram showing a configuration of an inspection support system according to an embodiment of the present invention.
- FIG. 2 is a schematic diagram showing the configuration of the tablet packaging device according to the embodiment of the present invention.
- FIG. 3 is a schematic diagram showing the configuration of the tablet packaging device according to the embodiment of the present invention.
- FIG. 4 is a diagram illustrating an example of a tablet rotation unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 5 is a diagram illustrating an example of a tablet rotation unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 6 is a diagram showing an example of a packaging result by the tablet packaging apparatus according to the embodiment of the present invention.
- FIG. 1 is a diagram showing a configuration of an inspection support system according to an embodiment of the present invention.
- FIG. 2 is a schematic diagram showing the configuration of the tablet packaging device according to the embodiment of the present invention.
- FIG. 3 is a schematic diagram showing the configuration of the tablet packaging device according to the embodiment of the
- FIG. 7 is a flowchart illustrating an example of the procedure of inspection support processing executed by the inspection support system according to the embodiment of the present invention.
- FIG. 8 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 9 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 10 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 11 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 12 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 13 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 14 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 15 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 16 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 17 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 18 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 14 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 15 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of
- FIG. 19 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 20 is a diagram illustrating an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 21 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 22 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 23 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 24A is a diagram showing a transition of the area of the tablet in the photographed image photographed by the inspection support system according to the embodiment of the present invention.
- FIG. 24A is a diagram showing a transition of the area of the tablet in the photographed image photographed by the inspection support system according to the embodiment of the present invention.
- FIG. 24B is a diagram showing a transition of the area of the tablet in the photographed image photographed by the inspection support system according to the embodiment of the present invention.
- FIG. 25 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 26 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 27 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 28 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 29 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 30 is a flowchart illustrating an example of a procedure of information registration processing executed by the inspection support system according to the embodiment of the present invention.
- FIG. 31 is a diagram showing an example of selection setting information used in the inspection support system according to the embodiment of the present invention.
- FIG. 32 is a flowchart illustrating an example of a procedure of device selection processing executed by the inspection support system according to the embodiment of the present invention.
- FIG. 33A is a diagram showing an example of a medicine package dispensed by the inspection support system according to the embodiment of the present invention.
- FIG. 33B is a diagram showing an example of a medicine package dispensed by the inspection support system according to the embodiment of the present invention.
- FIG. 33C is a diagram showing an example of a medicine package dispensed by the inspection support system according to the embodiment of the present invention.
- FIG. 34 is a flowchart showing an example of the procedure of automatic inspection processing executed by the inspection support system according to the embodiment of the present invention.
- FIG. 35A is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 35B is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 36 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 37A is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 37B is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 38 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 39A is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 39B is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 40 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 41 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 42A is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 42B is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 43 is a diagram showing a configuration of a packaging unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 44 is a diagram showing the configuration of the sealing device of the packaging unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 45 is a diagram showing the configuration of the sealing device of the packaging unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 46 is a diagram showing a configuration of the sealing device of the packaging unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 47 is a diagram showing an example of a medicine package dispensed from the tablet packaging device according to the embodiment of the present invention.
- FIG. 48 is a diagram showing a configuration of a packaging unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 49 is a diagram showing a configuration of a packaging unit of the tablet packaging device according to the embodiment of the present invention.
- FIG. 50A is a diagram illustrating an example of a photographed image photographed by the tablet packaging device according to the embodiment of the present invention.
- FIG. 50B is a diagram illustrating an example of a photographed image photographed by the tablet packaging device according to the embodiment of the present invention.
- FIG. 51 is a flowchart illustrating an example of a procedure of a method for forming a packaging bag executed by the tablet packaging device according to the embodiment of the present invention.
- FIG. 52 is a flowchart illustrating an example of a procedure of a subroutine relating to a second vertical seal forming process executed by the tablet packaging device according to the embodiment of the present invention.
- FIG. 53 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 54 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 55 is a diagram for explaining an example of pattern matching processing.
- FIG. 56 is a flowchart showing an example of the procedure of the image inspection process executed by the inspection support system according to the embodiment of the present invention.
- FIG. 57 is a flowchart showing an example of a procedure of a similar medicine registration process executed by the inspection support system according to the embodiment of the present invention.
- FIG. 58 is a diagram showing an example of a display screen displayed by the inspection support system according to the embodiment of the present invention.
- FIG. 59 is a diagram showing an example of a normal image used in the inspection support system according to the embodiment of the present invention.
- FIG. 60 is a diagram showing an example of a normal image used in the inspection support system according to the embodiment of the present invention.
- FIG. 61A is a diagram for explaining a capsule image processing function of the inspection support system according to the embodiment of the present invention.
- FIG. 61B is a diagram for explaining the capsule image processing function of the inspection support system according to the embodiment of the present invention.
- FIG. 61C is a diagram for explaining a capsule image processing function of the inspection support system according to the embodiment of the present invention.
- FIG. 61D is a diagram for explaining a capsule image processing function of the inspection support system according to the embodiment of the present invention.
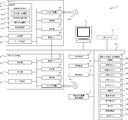
- an inspection support system 1 includes a server 2, one or a plurality of client terminals 3, and one or a plurality of tablets. It comprises a device 4 and one or more dispensing devices 5.
- the server 2 alone may be regarded as an inspection support system according to the present invention.
- the server 2, the client terminal 3, the tablet packaging device 4, and the dispensing device 5 are each connected to be communicable wirelessly or by wire via a communication network N 1 such as a LAN or the Internet.
- the server 2 is connected to a host system 6 such as an electronic medical record system or a prescription input terminal for inputting prescription data to the server 2 via the communication network N1. It is also conceivable that the server 2 can read prescription data from a prescription, or that the server 2 can input prescription data by a user operation.
- the server 2 is a personal computer including a control unit 21, a storage unit 22, a communication I / F 23, a display unit 24, an operation unit 25, a drive device 26, a code reading unit 27, and the like.
- the server 2 is arranged inside or outside a medical institution where the inspection support system 1 is used.
- the control unit 21 includes control devices such as a CPU, a ROM, a RAM, and an EEPROM (registered trademark).
- the CPU is a processor that executes various arithmetic processes.
- the ROM is a non-volatile storage unit in which information such as a control program for causing the CPU to execute various processes is stored in advance.
- the RAM is a volatile storage unit, and the EEPROM is a non-volatile storage unit.
- the RAM and the EEPROM are used as a temporary storage memory (working area) for various processes executed by the CPU.
- the said control part 21 performs various processes according to the various control programs previously stored in the said ROM, the said EEPROM, or the said memory
- the storage unit 22 is a storage unit such as a hard disk device or an SSD (Solid State Drive) that stores various data. Specifically, an inspection support program for causing a computer such as the control unit 21 to execute an inspection support process (see FIG. 7) described later is stored in the storage unit 22 in advance.
- the storage unit 22 also stores various databases such as a pharmaceutical master, a patient master, a cassette master, and a pharmacy master.
- the medicine master includes medicine ID, medicine code, medicine name, YJ code, JAN code (or RSS code), medicine bottle code, classification (dosage form: powder, tablet, liquid medicine, topical medicine, etc.), tablet shape (Capsule tablets, spherical tablets, flat tablets (disc-shaped tablets), etc.), tablet size, specific gravity, chemical type (ordinary drugs, poisonous drugs, narcotics, powerful drugs, antipsychotics, therapeutic drugs, etc.) Contains information about each drug, such as shape drugs and precautions.
- the patient master includes information about the patient such as patient ID, name, sex, age, medical history, prescription drug history, family information, medical department, ward, and hospital room.
- the pharmacy master includes pharmacy information such as a pharmacy name, a pharmacist name, and a pharmacist ID.
- the cassette master is information indicating the correspondence between the cassette identification information of each of the later-described drug cassettes 41 included in the tablet packaging device 4 and the tablets assigned to each of the drug cassettes 41.
- a medicine code for each medicine, a medicine code, a medicine name, a JAN code, an RSS code, a medicine bottle code, a dosage form, a unit, a specific gravity, a medicine type, a compounding change, a shaping medicine, a precaution, an allergy
- a drug database in which information such as information and attached document information is associated is stored separately from the drug master.
- information such as identification information of the tablet formed on the tablet, the shape of the tablet, and the appearance image (front and back) of the tablet are stored.
- the drug database is read from a recording medium such as a CD or a DVD by the drive device 26, or received from an external device via the communication network N1, and stored in the data storage unit 22.
- the drug database is used in the inspection support system 1 when information is taken into various masters such as the drug master, or when referring to information on a package insert of each drug.
- the medicine database can be displayed in response to a user operation on the server 2 or the client terminal 3.
- medical agent database from the external apparatus or website via the said communication network N1 as needed may be sufficient.
- the communication I / F 23 is wirelessly or wired between the client terminal 2, the tablet packaging device 4, and the external device such as the dispensing device 5 via the communication network N1 according to a predetermined communication protocol.
- a communication interface having a network card or the like for executing data communication.
- the display unit 24 is a display unit such as a liquid crystal monitor that displays various information and an operation screen in accordance with a control instruction from the control unit 21.
- the operation unit 25 is an operation unit such as a keyboard, a mouse, and a touch panel that accepts a user operation, and inputs an operation signal corresponding to the user operation to the control unit 21.
- the operation unit 25 receives various operation inputs such as a prescription data selection operation on the display screen displayed on the display unit 24 and a prescription data issuing operation for requesting the start of dispensing of the prescription data.
- the drive device 26 can read the inspection support program from a computer-readable recording medium 261 on which the inspection support program is recorded.
- the recording medium 261 is a CD, DVD, BD, or USB memory
- the drive device 26 is a CD drive, DVD drive, BD drive, USB port, or the like.
- the inspection support program read from the recording medium 261 using the drive device 26 is stored in the storage unit 22 by the control unit 21.
- the code reading unit 27 is a barcode reader capable of reading code information (such as a barcode or a two-dimensional code).
- code information such as a barcode or a two-dimensional code
- the code reading unit 27 is used to read prescription data from the code information described in a prescription.
- Prescription data read from the prescription is stored in the storage unit 22 by the control unit 21.
- the control unit 21 includes an inspection display processing unit 211, a list display processing unit 212, an operation display processing unit 213, and a re-execution processing unit 214. Specifically, the control unit 21 executes various processes in accordance with the inspection support program, thereby causing the inspection display processing unit 211, the list display processing unit 212, the operation display processing unit 213, and the re-execution process. It functions as the unit 214.
- the control unit 21 generates prescription data (dispensing data) for dispensing for executing dispensing processing such as packaging processing by the tablet packaging device 4 and the dispensing device 5 based on the prescription data.
- the prescription data is also input to the tablet packaging device 4 and the dispensing device 5. Thereby, in the tablet packaging device 4 and the dispensing device 5, a dispensing process such as a packaging process is executed based on the prescription data.
- the inspection display processing unit 211 is used before the tablets dispensed from the tablet cassette 41 or the hand handling unit 42 described later based on the prescription data in the tablet packaging device 4 are packaged with a packaging material such as a packaging paper.
- the photographed image of the tablet photographed at the time is displayed on the client terminal 3 or the like in a package unit (single package unit).
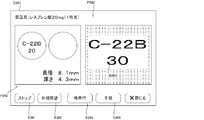
- the inspection display processing unit 211 displays the result of the inspection processing executed based on the identification information (characters or symbols) of the tablets included in the photographed image of the tablets and the prescription data, etc.
- the identification information recognized by the character recognition process from the photographed image of the tablet is collated with the identification information of the tablet included in the prescription data.
- an image of identification information included in the photographed image of the tablet may be collated with a normal image registered in advance corresponding to the identification information of the tablet included in the prescription data.
- the list display processing unit 212 displays a list of the cause of the error together with the taking date and the taking time of the tablet that caused the error. For example, the list display processing unit 212 displays an NG list area A37 (see FIG. 11) on the inspection details screen D31 described later on the client terminal 3 or the like.
- the operation display processing unit 213 is displayed after a predetermined operation on the first display screen or the first screen on which the inspection result of the medicine dispensed by the tablet packaging device 4 and the dispensing device 5 is displayed.
- an operation unit for individually re-executing part or all of the dispensing process based on the prescription data executed by the tablet packaging device 4 and the dispensing device 5 is displayed.
- the operation display processing unit 213 displays an inspection detail screen D31 (FIG. 11) described later, which is the first display screen on which the inspection display result is displayed by the inspection display processing unit 211.
- the operation key K33, the operation key K34, the input field K35, and the operation key described later are used as the operation unit. K36 or the like can be displayed.
- the re-execution processing unit 214 is a part or all of the dispensing process executed by the tablet packaging device 4 and the dispensing device 5 in response to a user operation on the operation unit displayed by the operation display processing unit 213. Is re-executed by the tablet packaging device 4 and the dispensing device 5. More specifically, in the present embodiment, the re-execution processing unit 214 performs the packaging performed by the tablet packaging device 4 in response to a user operation on the operation unit displayed by the operation display processing unit 213. Part or all of the processing can be reexecuted by the tablet packaging device 4.
- the client terminal 3 is a personal computer including a control unit 31, a storage unit 32, a communication I / F 33, a display unit 34, an operation unit 35, a drive device 36, a code reading unit 37, and the like.
- Each of the client terminals 3 is an operation terminal that is arranged in a medical institution where the inspection support system 1 is used and is operated by a user such as a pharmacist.
- the control unit 31 includes control devices such as a CPU, a ROM, a RAM, and an EEPROM.
- the CPU is a processor that executes various arithmetic processes.
- the ROM is a non-volatile storage unit in which information such as a control program for causing the CPU to execute various processes is stored in advance.
- the RAM is a volatile storage unit, and the EEPROM is a non-volatile storage unit.
- the RAM and the EEPROM are used as a temporary storage memory (working area) for various processes executed by the CPU.
- the said control part 31 performs various processes according to the various control programs previously stored in the said ROM, the said EEPROM, or the said memory
- the storage unit 32 is a non-volatile storage unit such as a hard disk or SSD in which various application programs executed by the control unit 31 and various data are stored. Specifically, the storage unit 32 stores an operating system (OS) and application programs such as browser software.
- the browser software displays various operation screens on the display unit 34 by accessing the server 2 via the communication network N1, and performs an input operation on the operation screen using the operation unit 35.
- Application software for transmission to the server 2. Specifically, when address information such as a URL (Universal Resource Locator) corresponding to the server 2 is input at a predetermined position on the operation screen displayed by the browser software, the control unit 31 receives the address information.
- the server 2 is accessed based on the above.
- the communication I / F 33 is a communication interface having a network card or the like that performs data communication with an external device such as the server 2 via the communication network N1 in a wireless or wired manner according to a predetermined communication protocol. is there.
- the display unit 34 is a display unit such as a liquid crystal display or an organic EL display that displays various types of information in accordance with control instructions from the control unit 31.
- the operation unit 35 is an operation unit operated by a user in order to input various information to the client terminal 3.
- the operation unit 35 includes a keyboard and a mouse (pointing device) that accept input operations on various operation screens displayed on the display unit 34.
- the operation unit 35 may include a touch panel that receives touch operations on various operation screens displayed on the display unit 34, or a voice input device that receives input of various types of information by voice recognition.
- the drive device 36 can read the OS or the browser software from a computer-readable recording medium 361 in which the OS or the browser software is recorded.
- the recording medium 361 is a CD, DVD, BD, or USB memory
- the drive device 36 is a CD drive, DVD drive, BD drive, USB port, or the like.
- the OS or the browser software read from the recording medium 361 by using the drive device 36 is stored in the storage unit 32 by the control unit 31.
- the code reading unit 37 is a barcode reader capable of reading code information (such as a barcode or a two-dimensional code).
- code information such as a barcode or a two-dimensional code.
- the code reading unit 37 is used for reading the code information described in the medicine package 451.
- the server 2 and the client terminal 3 constitute a server client system, and the server 2 executes various processes according to user operations of the client terminal 3. To do. Therefore, “display”, “operation”, “selection”, “input” and the like described below are performed using the display unit 34 and the operation unit 35 of the client terminal 3. For example, the control unit 21 of the server 2 displays various screens on the client terminal 3 by transmitting data described in a page description language such as HTML to the client terminal 3.
- a page description language such as HTML
- the inspection support program is installed in any one or a plurality of the server 2, the client terminal 3, and the tablet packaging device 4, and the inspection support processing described later is performed on the server. 2, it is also conceivable that the client terminal 3 and the tablet packaging device 4 are executed in cooperation.
- the inspection support system 1 is provided with an inspection support system having an inspection support function for executing inspection support processing (see FIG. 7) described later, separately from the server 2.
- the inspection support system is an information processing apparatus such as a personal computer or a tablet terminal, for example, and includes the server 2, the client terminal 3, the tablet packaging device 4, and the dispensing device provided in the inspection support system 1. It is possible to communicate with 5 or the like. It is also conceivable that the inspection support system is mounted on a dispensing device such as the tablet packaging device 4.
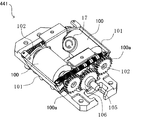
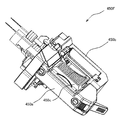
- the tablet packaging device 4 is a dispensing device used for dispensing a medicine. Specifically, as shown in FIG. 1 and FIG. 2, the tablet packaging device 4 includes a control unit 40, a tablet cassette 41, a hand handling unit 42, a separation unit 43, a rotation unit 44, a packaging unit 45, a photographing unit. Unit 46, passage detection unit 47, and the like.
- the tablet packaging device 4 can automatically dispense tablets from either one or both of the tablet cassette 41 and the manual handling unit 42 based on the prescription data, and can be packaged in dosage units. is there.
- the dashed-dotted line in FIG. 2 has shown the movement path
- the imaging unit 46 includes cameras 461 to 467 provided in a tablet movement path from the tablet cassette 41 to the packaging unit 45 and a tablet movement path from the hand-making unit 42 to the packaging unit 45. .
- the cameras 461 to 467 are arranged so that the tablets delivered from the tablet cassette 41 or the hand-rolling unit 42 are divided into one or a plurality of tablets before the packaging unit 45 wraps the tablets. Used for shooting. Note that images taken by the cameras 461 to 467 are color or monochrome.
- the captured images of the tablets by the cameras 461 to 467 are stored by the control unit 40 in the storage unit 49 which is a storage device such as a hard disk provided in the tablet packaging device 4 and transmitted to the server 2. Is done.
- the camera 461 is used to photograph the tablet supplied from the tablet cassette 41 to the rotating unit 44.
- the camera 462 and the camera 463 photograph a plurality of different areas (for example, the front surface and the back surface) of the outer periphery of the tablet from a tablet rotated by a tablet rotation unit 441 described later provided in the rotation unit 44.
- the camera 464 is used to photograph the tablet accommodated in the hand handling unit 42.
- the camera 465 is used to take an image of the tablet supplied from the hand handling unit 42 to the separation unit 43.
- the camera 466 is used to photograph the tablet supplied from the separation unit 43 to the rotation unit 44.
- the camera 467 is used to photograph tablets in a taking unit (packaging unit) stored in a storage unit 443 described later provided in the rotating unit 44.
- the camera 467 is used to photograph the tablet falling from the rotating unit 44 during the dropping.
- it may replace with the said storage part 443 and the chemical
- the camera 467 may be arranged at a position where the imaging range including the medicine introduction unit 80, the tablet, and the packaging material can be imaged instead of the tablet stored in the storage unit 443. It is done.
- the passage detection unit 47 detects the passage of the tablet in the tablet movement path from the tablet cassette 41 to the packaging unit 45 and the tablet movement path from the hand handling unit 42 to the packaging unit 45. It includes passage detection sensors 471 to 475 such as optical sensors for detection. The tablet detection signals from the passage detection sensors 471 to 475 are input to the control unit 40.
- the passage detection sensor 471 detects a tablet dispensed from the tablet cassette 41
- the passage detection sensor 472 detects a tablet falling from the tablet cassette 41 to the rotation unit 44.
- the passage detection sensor 473 detects a tablet dispensed from the hand handling unit 42
- the passage detection sensor 474 detects a tablet falling from the separation unit 43 to the rotation unit 44.
- the passage detection sensor 475 detects a tablet that falls in a storage unit 443 from a tablet rotation unit 441 described later in the rotation unit 44.
- the control unit 40 includes a processor such as a CPU and a storage unit such as a RAM and an EEPROM, and controls the tablet packaging device 4 in an integrated manner. Specifically, the control unit 40 controls one or more types of tablets corresponding to the prescription data by controlling the operation of the tablet packaging device 4 based on the prescription data input from the server 2. A sachet process is performed to sachet at each dose.
- control unit 40 executes a photographing process for photographing an image by the photographing unit 46 in accordance with the detection timing of the tablet by the passage detecting unit 47.
- the camera 461 performs imaging.
- the photographing by the camera 462 and the camera 463 is performed at a photographing interval (several ms) set in advance during execution of the packaging process in the tablet packaging device 4.
- the photographing by the camera 464 is performed when an operation input to the effect that the hand-working operation of the tablets to the hand-making unit 42 is completed.
- a tablet dispensed from the hand-rolling unit 42 is detected by the passage detection sensor 473
- photographing is performed by the camera 465
- a tablet falling from the separation unit 43 to the tablet rotating unit 441 is detected as the passage.
- photographing by the camera 466 is performed.
- the passage detection sensor 475 detects a tablet corresponding to the number of doses
- photographing by the camera 467 is performed.
- the camera 467 performs imaging every time the tablet falling from the rotating unit 44 is detected by the passage detection sensor 475. The tablet falling from the rotating unit 44 is photographed.
- the shooting timings by the cameras 461 to 467 are not limited to these, and may be set in advance according to the use of each shot image.
- the control unit 40 pays out from the tablet cassette 41 or the hand handling unit 42 toward the packaging unit 45 based on the detection results of the passage detection sensors 471 to 475. It is possible to judge the progress of tablets. Accordingly, the control unit 40 can determine to which position the tablet has moved and notify (display) it to the user, for example, when the dispensing of the tablet has not been performed normally.
- an exterior member on a path along which the tablet moves is formed of a transparent or translucent member, and a configuration in which the movement state of the tablet can be visually recognized from the outside is also considered. It is done.
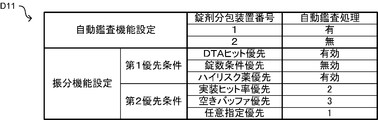
- the control unit 40 can execute an automatic inspection process based on the image of the tablet imaged by the camera 462 or the camera 463 and the prescription data.
- the automatic inspection process when the identification information of the tablet included in the photographed image of the tablet is recognized, it is verified whether the identification information of the tablet matches the medicine information included in the prescription data. .
- the control unit 40 appropriately performs the packaging process based on the photographed image photographed by the camera 462 or the camera 463 before the tablets are packaged in the medicine package 451 in the packaging process. Check whether it was done.
- the automatic inspection process for automatically inspecting the suitability of the packaging process the control when the image inspection process for comparing the identification information of the tablet included in the photographed image with the prescription data is executed.
- the unit 40 is an example of an image inspection processing unit. Note that various conventional techniques such as a technique of reading the identification information by a pattern matching process are used as a method of acquiring the identification information of the tablet from the photographed image of the tablet in the automatic inspection process. Then, when the collation result is coincident, it is judged as appropriate, and when the collation result is inconsistent, it is judged as an error.
- control unit 40 executes the automatic inspection process using the identification information of the tablet acquired from the captured image of the tablet
- the control unit 40 captures the tablet captured by the camera 462 or the camera 463.
- the original image when the identification information is read from the tablet is transmitted to the server 2 together with the result of the automatic inspection process (match or error).
- the control unit 40 extracts the region of the tablet from the captured image captured by the camera 462 or 463, and reads the identification information of the tablet based on the extracted trimmed image. It is possible. In this case, the control unit 40 transmits the trimmed image as the original image to the server 2 instead of or together with the captured image.
- the tablet cassette 41 is a cassette for storing a predetermined type of tablet, and the tablet packaging device 4 is provided with a plurality of the tablet cassettes 41. In the tablet packaging device 4, the tablets included in the prescription data are automatically dispensed from the tablet cassette 41.
- the hand-rolling unit 42 includes a mass provided in a matrix, and each mass is loaded with a tablet that is not suitable for dispensing from the tablet cassette 41, such as a half tablet. And the said hand-rolling unit 42 can pay out the said tablet accommodated in each said square in the said square unit.
- the separation unit 43 can separate the tablets dispensed from the manual handling unit 42 one by one and supply them to the rotation unit 44.
- Various configurations are conceivable as the configuration of the separation unit 43. For example, a tablet placed in alignment with a V-shaped groove is conveyed by vibrating the groove, thereby leading the tip of the groove. One tablet at a time.
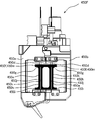
- the rotation unit 44 includes six tablet rotation units 441, a unit rotation unit 442, and a storage unit 443, as shown in FIGS.
- the unit rotating part 442 is rotatably supported by a base part (not shown).
- FIG. 3 is a schematic diagram showing the rotating unit 44 as viewed from above.
- Each tablet rotating unit 441 can displace the posture of the tablet by rotating one tablet supplied from the tablet cassette 41 or the hand-rolling unit 42.
- the unit rotating part 442 six tablet rotating parts 441 are arranged at intervals of 60 ° around a predetermined rotation axis, and the unit rotating part 442 moves the tablet rotating part 441 around the predetermined rotation axis. Can be rotated.
- the unit rotating unit 442 includes a tablet dropping position P1 from the separation unit 43, a tablet falling position P2 from the tablet cassette 41, and a position where the camera 462 can capture images. It is possible to move sequentially to six locations: P3, a position P4 that can be photographed by the camera 463, a preliminary position P5, and a drop position P6 to the storage section 443.
- the tablet packaging device 4 As shown in FIG. 4, when one tablet 17 dispensed from the tablet cassette 41 falls into the tablet rotating part 441, or dispensed from the separation unit 43.
- the unit rotation unit 442 rotates the tablet rotation unit 441 by 60 °.
- each tablet dispensed from the tablet cassette 41 or the hand handling unit 42 is individually placed on the tablet rotating unit 441, and the drop position from the drop position P1 or the drop position P2. Move sequentially toward P6.
- the tablet 17 is photographed by the camera 462 and the camera 463 at the photographable position P3 and the photographable position P4.
- the storage unit 443 is used to temporarily store a tablet of a dosage unit in the middle of dropping from the rotating unit 44 to the packaging unit 45.
- the bottom is opened, and the tablets in the taking unit stored in the storing unit 443 are supplied to the packaging unit 45.
- an illumination device 468 and an illumination device 469 that illuminate the tablet are fixed above the rotating unit 44 at a position where the camera 462 and the camera 463 can be photographed.
- the illumination device 468 and the illumination device 469 irradiate the tablet rotation unit 441 with light at different angles or different illuminances so that images with different illumination environments are captured by the camera 462 and the camera 463, respectively.
- the illumination device 468 performs illumination suitable for reading the identification information of the tablet formed by stamping the tablet.
- the illumination device 468 illuminates the tablet so that the identification information of the tablet is clearly captured. It is conceivable that the light is irradiated from the side or obliquely from above.
- the tablet may be irradiated with light from a plurality of directions. This is because, when the identification information of the tablet formed on the tablet is stamped, unevenness is generated by the marking on the surface of the tablet, and the way the identification information is reflected in the photographed image of the tablet is the light against the unevenness. This is because it may change depending on the irradiation angle.
- the camera 462 and the illumination device 468 are examples of the first photographing unit.
- the illumination device 469 performs illumination suitable for reading the identification information of the tablet formed by printing on the tablet. For example, the illumination device 469 applies the illumination to the tablet so that the identification information of the tablet is clearly captured. It is considered that the light is irradiated from above.
- the identification information of the tablet formed on the tablet is printing, the identification information is formed on the surface of the tablet by a paint or a developer, so that the tablet has no unevenness. This is because the way the identification information is captured in the photographed image of the tablet is difficult to change.
- the camera 463 and the illumination device 469 are examples of the second photographing unit. Note that whether the identification information of the tablet such as characters or symbols formed on the tablet is formed on the tablet by stamping or printing is registered in advance in the pharmaceutical master or the like. It is conceivable that the medicine master is stored not only in the storage unit 22 of the server 2 but also in the storage unit 49 provided in the tablet packaging device 4.
- a stamp flag is set to “1” for a tablet whose identification information is stamped, and a print flag is set to “1” for a tablet whose identification information is print. It is possible.
- the stamp flag is set to “1” for a tablet whose identification information is stamped, and the stamp flag is set to “0” for a tablet whose identification information is print. May be.
- the picked-up image suitable for the formation method (stamping or printing) of the identification information of the said tablet with respect to a tablet among the picked-up images by the said camera 462 and the said camera 463 is used for the said automatic inspection process. .
- the marking is formed on the surface of the tablet by unevenness, and the coating or developer is applied to the surface of the tablet. This includes printing on which identification information is formed. Further, among images captured by the camera 462 and the camera 463, an image having a high degree of coincidence such as pattern matching when recognizing the identification information of the tablet, or an image in which the identification information of the tablet is clearly captured It may be used for automatic inspection processing.
- a medicine code in which information such as shape medicine, precautions, allergy information, and package insert information is associated is stored separately from the drug master.
- information such as shape medicine, precautions, allergy information, and package insert information is associated is stored separately from the drug master.
- the drug database for the tablet, information such as identification information of the tablet formed on the tablet, the shape of the tablet, and the appearance image (front and back) of the tablet are stored.
- the drug database is read from a recording medium such as a CD or a DVD by a drive device (not shown) provided in the tablet packaging device or received from an external device such as the server 2 via the communication network N1. And stored in the storage unit 49. Further, the control unit 40 may be configured to be able to read out the medicine database as needed from an external device such as the server 2 or a website via the communication network N1.
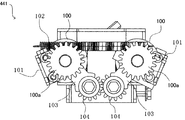
- the tablet rotating unit 441 includes a pair of rotating rollers 100, a pair of supporting plates 101 that individually support the rotating rollers 100, and the pair of supporting plates 101. And a spring 102 for urging them in a direction approaching each other.
- the arm 103 extends from both ends of each of the support plates 101, and a gear 104 that meshes with each other is formed at the tip part. Accordingly, in a normal state, the rotating rollers 100 are in a state of being close to each other, and the tablet 17 can be supported by the pair of rotating rollers 100.
- the pair of rotating rollers 100 are moved toward and away from each other in synchronization with the rotation of the gear 104, and the drug 17 is rotatably supported in the contacted or approached state, and the drug 17 is separated from the pair of rotating rollers 100 in the separated state. It falls toward the storage part 443.
- a driven gear 100a is integrated with one end portion of the rotating shaft of each of the rotating rollers 100.
- a driving gear 106 integrated with one end of the driving shaft 105 is engaged with each driven gear 100a.
- a driven roller (not shown) is integrated with the other end of the drive shaft 105.
- the driven roller is composed of a magnet gear.
- the tablet rotating unit 441 moves to the photographing position P3 of the camera 462 or the photographing position P4 of the camera 463, the driving force from the predetermined driving motor is applied to the driven roller via the connecting magnet gear. Communicated. Accordingly, the driving force of the predetermined driving motor is transmitted to the pair of rotating rollers 100 via the driven roller and the driving shaft 105, and the pair of rotating rollers 100 are rotated in the same direction in synchronization. Therefore, when the tablet 17 is placed on the pair of rotating rollers 100, the tablet 17 rotates. Therefore, the tablet rotating unit 441 can change the posture of the tablet 17 photographed by the camera 462 and the camera 463, and can photograph the outer peripheral surface of the tablet 17 from different directions. In other words, the tablet 17 rotated by the tablet rotating unit 441 is photographed intermittently or continuously by the camera 462 and the camera 463, so that the outer periphery including identification information formed on the tablet 17 by engraving or the like. The surface can be photographed.
- the configuration of the tablet rotation unit 441 described here is merely an example, and the tablet rotation unit 441 can capture an image from which the tablet identification information can be read by the camera 462, the camera 463, and the like. Any configuration capable of displacing the tablet in a stable state may be used. However, it is desirable to photograph in the state where the identification information of the tablet appears clearly in the photographed image. Moreover, in the said tablet packaging apparatus 4, if it can image
- control unit 40 may compare all the captured images with the normal image associated with the tablet, or try to read the identification information of the tablet from all the captured images.
- the processing becomes heavy and the reading accuracy of the identification information of the tablet is lowered.
- the tablet is a flat tablet
- a photographed image taken by the camera 462 or the camera 463 from the direction perpendicular to the front and back surfaces of the flat tablet is compared with the normal image.
- the accuracy of the automatic inspection process is increased. This is because, when the tablet is a flat tablet, the identification information of the tablet is located on one or both of the front and back surfaces of the flat tablet.
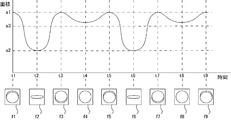
- the control unit 40 selects a plurality of photographed images to be used for acquisition of identification information of the tablets, or a photographed image to be compared with the normal image among the photographed images. It may be specified according to the area of the tablet included in the photographed image.
- the control unit 40 determines whether or not the tablet is a flat tablet according to the transition of the area of the tablet in each of the captured images. For example, when the ratio of the maximum value and the minimum value of the area of the tablet in each of the captured images is equal to or greater than a preset threshold value, that is, when the amount of change in the area of the tablet is large, the tablet is a flat tablet. Judging that there is.
- the control unit 40 may determine whether the tablet is a flat tablet based on the drug master or the drug database.
- the control unit 40 changes the tablet area transition (inclination) from an increase to a decrease.
- the previous photographed image is specified as a photographed image that maximizes the area of the tablet.
- two photographed images in which the area of the tablet is maximized are specified from the plurality of photographed images corresponding to one round of the outer peripheral surface of the tablet. That is, the two photographed images with the largest tablet area have the highest tablet area in the photographed images corresponding to one round of the outer peripheral surface of the tablet photographed at a predetermined photographing interval. It is up to two images.
- the photographed image immediately before the image is specified as an image obtained by capturing the front or back surface of the tablet from the front.
- FIG. 24A is a diagram showing the transition of the area of the tablet when the state that the flat tablet as the tablet is rotated by the tablet rotating unit 441 is continuously photographed.
- the area of the tablet in each of the captured images transitions between an area a1 that is the maximum value and an area a2 that is the minimum value according to the rotation of the tablet in the tablet rotation unit 441.
- the front or back surface to which the identification information of the tablet is attached is slightly inclined with respect to the camera 462 or the camera 463 of the imaging unit 46, and the front or back surface of the tablet and the side surface of the tablet are When both are photographed, the area of the tablet in the photographed image becomes the maximum area a1.
- the captured images f1 to f9 shown in FIG. 24A will be described as an example. However, in the tablet packaging device 4, one or a plurality of captured images are also captured between the captured images f1 to f9. Is done.
- the tablet In the captured images f1, f3, f5, f7, and f9 corresponding to times t1, t3, t5, t7, and t9 when the area of the tablet is the maximum area a1, the tablet The front surface or back surface and part of the side surface are included.
- the captured images f2 and f6 corresponding to the times t2 and t6 when the area of the tablet is the minimum area a2 include only the side surface of the tablet.
- the captured images f4 and f8 corresponding to the times t4 and t8 where the area of the tablet is less than the area a1 and the area a3 is predetermined for each type of the tablet include the front or back surface of the tablet.
- the area a3 is a value that is smaller than the area a1 and larger than the area a2, and is a value that serves as an index for distinguishing between the area of the front or back surface of the tablet and the area of the side surface of the tablet. It is a set value.
- the area a3 is a value that is smaller by a predetermined value than the area when the front or back surface of the tablet is captured from the front.
- the said control part 40 is image
- a photographed image having the smallest tablet area is extracted.
- the control unit 40 specifies an odd-numbered captured image extracted as the captured image f4 among the extracted captured images, and specifies an even-numbered extracted captured image as the captured image f8.
- the control unit 40 specifies the photographed image f4 as an image of the front surface of the tablet and the photographed image f8 as a back surface of the tablet.
- the control unit 40 compares the captured images f4 and f8 obtained by capturing the front and back surfaces of the tablet from the front with the normal image, or the identification information of the tablet is high from the captured images f4 and f8. It is possible to read with accuracy.
- the shooting time per tablet taken by the camera 462 and the camera 463 in the packaging process is, for example, between the times t1 to t9 among various tablets registered in the pharmaceutical master. It is conceivable that the required time is determined to be longer than the required time on the basis of the tablet with the longest required time. As a result, at least one of the photographed image f4 and the photographed image f8 is photographed as the photographed images of the front and back surfaces of the tablet.
- the said control part 40 is the said picked-up image image
- the smooth value is a simple moving average value of tablet areas in a plurality of photographed images.
- FIG. 24B is a diagram illustrating an example of transition of the area of the tablet when continuously capturing a state in which the flat tablet which is the tablet is rotated by the tablet rotating unit 441.
- FIG. 24B shows the result of a total of 20 shootings at shooting timings t11 to t30.
- the solid line indicates the measured value of the area of the tablet in the captured image captured at each of the imaging timings t11 to t30
- the alternate long and short dash line indicates the tablet in the captured image captured at each of the imaging timings t11 to t30.
- the smooth value of the area is shown.
- the smooth value is a peak value at the photographing timing t19
- the photographed image photographed at the photographing timing t19 is specified as a photographed image of the front or back surface of the tablet. More specifically, an odd-numbered peak value among the smooth values is specified as a captured image of the front surface of the tablet, and an even-numbered peak value is specified as a captured image of the back surface of the tablet.
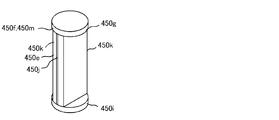
- the packing unit 45 packs a tablet of a dosage unit supplied from either one or both of the tablet cassette 41 and the hand handling unit 42 via the rotating unit 44 with a medicine package 451 which is a packaging material.
- a medicine package 451 which is a packaging material.
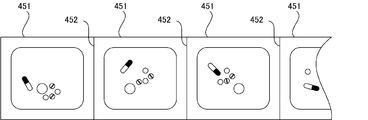
- FIG. 6 is a view showing an example of the medicine package 451 dispensed from the tablet packaging device 4. As shown in FIG. 6, each medicine package 451 is packed with a plurality of tablets in dosage units, and a dotted line for easily separating each medicine package 451 between each medicine package 451. (Perforation) 452 is formed.
- the medicine package 451 used for packaging in the packaging unit 45 or the packaging paper (roll paper) for forming the medicine package 451 is a consumable that is replenished or replaced as necessary.
- the tablet packaging device 4 has a function of detecting the remaining amount of the medicine package 451 or the packaging paper, and the remaining amount is appropriately notified to the server 2 by the control unit 40.
- the blister pack is a packaging material in which a plurality of closed packaging parts are formed by joining a packaging member having a plurality of packaging regions with one side open to a flat plate-like member.
- the tablets stored in the tablet cassette 41 are also examples of consumables, and have a function of detecting the remaining amount of tablets stored in the tablet cassette 41. The remaining amount is appropriately notified to the server 2 by the control unit 40.
- the packaging unit 45 is provided with a printing unit 453 for printing information on each medicine package 451, and the name of the patient and the time of taking are printed on the surface of each medicine package 451 by the printing unit 453. Information such as prescription drugs or prescription amounts can be printed.
- the ink ribbon used for printing in the printing unit 453 is a consumable item that is replenished or replaced as necessary.
- the tablet packaging device 4 has a function of detecting the remaining amount of the ink ribbon, and the remaining amount is appropriately notified to the server 2 by the control unit 40.
- the dispensing device 5 is a device used when dispensing a drug based on prescription data, similarly to the tablet packaging device 4.
- the dispensing device 5 includes, for example, a powder packaging device, a liquid dispensing device, a sheet dispensing device, and a picking auxiliary device.
- the powder packaging device has a plurality of powder cassettes containing a plurality of types of powders, and can automatically package the powders stored in the powder cassettes by a predetermined amount according to prescription data. It is.
- the liquid medicine dispenser has a plurality of medicine bottles containing a plurality of kinds of liquid medicines, and dispenses a necessary amount of liquid medicine from the medicine bottles according to prescription data.
- the sheet dispensing device dispenses from a plurality of sheet cassettes containing PTP sheets pre-packed with tablets or heat seals according to prescription data.
- the picking assist device is used when a pharmacist manually dispenses, reads a drug name from identification information (such as a barcode) attached to a drug shelf or a drug bottle, and the read drug name and prescription data The drug name included in is checked against.
- an inspection operation is performed by a pharmacist to confirm whether or not the tablets packaged by the tablet packaging device 4 are appropriate for the prescription data.
- the inspection support system 1 the inspection work of the pharmacist is supported by executing an inspection support process (see FIG. 7) described later.
- the inspection support process is performed when a login operation by a pharmacist having a preset final inspection authority is performed on the client terminal 3 or an inspection start operation for performing a final inspection process after the login operation is performed. It is executed when it is broken. Note that “display” and “operation” described below are performed using the display unit 33 and the operation unit 34 of the client terminal 3 on which the login operation has been performed.
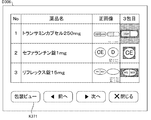
- step S11 the control unit 21 causes the client terminal 3 to display an inspection wait list screen D1 for displaying a list of prescription data to be inspected.
- FIG. 8 is a diagram showing an example of the inspection waiting list screen D1.
- a list display area A11 in which a list of prescription data to be inspected is displayed on the inspection waiting list screen D1.
- the list display area A11 is connected to the inspection support system 1 together with prescription ID (prescription identification information), patient name (patient identification information), taking start date, and number of days included in the prescription data.
- Status information of a plurality of dispensing devices such as the tablet packaging device 4 and the dispensing device 5 is displayed.
- the powder dispensing device is pre-registered as No. 1 machine and the sheet dispensing device is No. 3 machine, and the tablet packaging device Assume that 4 is registered in advance as Unit 2.
- the status information is classified in advance into “Packaging complete”, “Dispensing complete”, “Packaging in progress”, “In operation”, “Waiting for start”, “Out of stock”, and the like.
- “Packaging complete” and “Dispensing complete” indicate that the dispensing of the medicine has been completed, and the background or characters are displayed in a first specific color such as a predetermined blue color.
- “Packaging” and “in operation” indicate that the dispensing of the medicine is being executed, and is displayed together with the waiting time until the end.
- “Waiting for start” is a waiting state until the start of dispensing of a medicine, and is displayed together with a waiting time until the start of dispensing.
- “Out of stock” indicates that dispensing cannot be started because consumables such as the medicine used in the dispensing based on the prescription data, the medicine package 451, the packaging paper, or the ink ribbon are out of stock.
- the background or characters are displayed in a second specific color such as a predetermined red color.
- the inspection waiting list screen D1 displays operation keys K11 to K12 for receiving user operations.
- the operation key K11 is an operation key for displaying an inspection history screen D2 for displaying an inspection history in the server 2
- the operation key K12 is an inspection screen for executing inspection processing in the server 2.
- It is an operation unit for displaying D3 (an example of a first display screen). That is, the control unit 21 may start inspection of dispensing performed by a plurality of dispensing devices such as the tablet packaging device 4 and the dispensing device 5 in accordance with the operation on the inspection waiting list screen D1. Is possible.
- step S12 the control unit 21 determines whether or not a display operation of the inspection history screen D2 has been performed. Specifically, the control unit 21 determines that the display operation of the inspection history screen D2 has been performed when the operation key K11 on the inspection waiting list screen D1 is operated. And if the said control part 21 judges that display operation of the said inspection history screen D2 was performed (S12: Yes), it will transfer a process to step S13, and if display operation of the said inspection history screen D2 is not performed (S12: No), the process proceeds to step S14.
- step S13 the control unit 21 displays the inspection history screen D2 on the client terminal 3. In this way, the control unit 21 displays the inspection history of the dispensing performed by a plurality of dispensing devices such as the tablet packaging device 4 and the dispensing device 5 according to the user operation on the inspection waiting list screen D1. It can be displayed.
- FIG. 9 is a diagram showing an example of the inspection history screen D2.
- the inspection history screen D2 displays a list display area A21 in which a list of prescription data that has already been inspected is displayed.
- information such as a prescription ID (prescription identification information), a patient name (patient identification information), a start date of taking, an inspector, and an inspection date / time included in the prescription data is displayed.
- the determination result is appropriate as the inspection result of the medicine dispensed by each of the tablet packaging device 4 and the dispensing device 5 connected to the inspection support system 1. “OK” indicating “?” Or “NG” indicating that the determination result is an error is displayed.
- the inspection result corresponding to the tablet packaging device 4 is a result of the automatic inspection processing.
- “OK” is displayed in a third specific color such as blue where the background or characters are predetermined
- “NG” is displayed in a fourth specific color such as red where the background or characters are predetermined.
- the content of the treatment performed in response to the determination result being an error is displayed.
- an operation key K21 for selecting one of “patient ID” and “period” as a search item, and a search content for the item selected by the operation key K21 are input.
- An input field K22, an operation key K23 for executing a search, and the like are displayed.
- the control unit 21 searches for prescription data corresponding to the search content and displays the prescription data in the list display area A21.
- an operation key K24 for displaying an inspection screen similar to the inspection screen D3 described later for displaying details of the inspection history of the prescription data displayed in the list display area A21 is displayed. ing.
- step S14 the control unit 21 determines whether or not the inspection start operation has been performed. Specifically, the control unit 21 determines that the inspection start operation has been performed when the operation key K12 is operated in a state where the prescription data is selected on the inspection waiting list screen D1. And if the said control part 21 judges that the said inspection start operation was performed (S14: Yes), it will transfer a process to step S15, and if the said inspection start operation is not performed (S14: No), a process will be performed. The process proceeds to step S16.
- step S15 the control unit 21 causes the client terminal 3 to display the inspection screen D3 for performing inspection on the prescription data selected on the inspection waiting list screen D1.
- FIG. 10 shows an example of the inspection screen D3.
- a basic information area A31, an inspection result area A32, and a device information area A33 are displayed on the inspection screen D3.
- information such as prescription ID (prescription identification information), patient name (patient identification information), gender, age, taking date, and usage included in the prescription data is displayed.
- the inspection result area A32 the contents of the prescription data selected on the inspection waiting list screen D1 and the inspection results for the prescription data are displayed for each record (Rp1 to Rp3). Specifically, in the inspection result area A32, a medicine name, a dose, a form, a number of a dispensing device, a dose, and a determination result are displayed.
- the said determination result is a determination result of the suitability in the automatic inspection process performed in each of the said tablet packaging apparatus 4 and the said dispensing apparatus 5 like the said automatic inspection process performed in the said tablet packaging apparatus 4, for example. Yes, the data is input to the server 2 from the tablet packaging device 4 and the dispensing device 5 as appropriate.
- the said automatic inspection process may be performed by the said control part 21 of the said server 2 which acquired various information, such as an image, from the said tablet packaging apparatus 4 and the said dispensing apparatus 5.
- the user can collectively check the inspection results of the plurality of dispensing devices 5 including the tablet packaging device 4 that executes dispensing based on the prescription data.
- Rp1 is a medicine dispensed by the tablet packaging device 4
- Rp2 is a medicine dispensed by the powder medicine packaging device in the dispensing device 5
- Rp3 is the dispensing It is conceivable that the device 5 is a medicine dispensed by the sheet dispensing device.
- the device information area A33 remaining amount information of consumables in the tablet packaging device 4 and the dispensing device 5 connected to the server 2 is displayed.
- the remaining amount information of the consumables displayed in the device information area A33 is appropriately input to the server 2 from each of the tablet packaging device 4 and the dispensing device 5, or the tablet packaging device 4 by the server 2 And read from each of the dispensing devices 5.
- the user can easily check the remaining amount of consumables in the tablet packaging device 4 and the dispensing device 5.
- the remaining amount of “wrapping paper” is small and the remaining amount of “ink ribbon” is sufficient as the consumable information of the tablet packaging device 4 of “No. 2”. It is shown.
- the control unit 21 can display information on consumables in a plurality of dispensing devices such as the tablet packaging device 4 and the dispensing device 5 in a list on the inspection screen D3. Therefore, the user can easily manage the entire consumables of the inspection support system 1. Thereby, arrangement of the consumables can be performed without omission, and a plurality of dispensing devices such as the tablet packaging device 4 and the dispensing device 5 can be continuously operated even by a small number of people.
- step S16 the control unit 21 determines whether or not a detail confirmation operation for confirming details on the determination result displayed on the inspection screen D3 has been performed. Specifically, in the inspection screen D3, when a display location of a determination result corresponding to any of the records included in the prescription data displayed on the inspection screen D3 is selected, the detailed confirmation operation Is determined to have been performed. If the control unit 21 determines that the detail confirmation operation has been performed (S16: Yes), the process proceeds to step S17. If the detail confirmation operation has not been performed (S16: No), the process is performed. To step S18.
- step S17 the control unit 21 displays an inspection detail screen D31 (an example of a second display screen) on which details about the determination result displayed on the inspection screen D3 are displayed.
- the inspection screen D3 and the inspection details screen D31 are displayed by the inspection display processing unit 211 of the control unit 21.
- FIG. 11 is a diagram showing an example of the inspection details screen D31.
- the basic information area A31, prescription information area A35, dispensing details area A36, NG list area A37, and the like are displayed on the inspection detail screen D31.
- the prescription information area A35 as prescription information of the record corresponding to the determination result, a medicine name (medicine identification information), the number of tablets for each taking time, an image of the medicine, and the like are displayed.
- the contents of the tablets packaged in the medicine package 451 corresponding to the taking time (number of tablets at each taking time) of each taking day are displayed. More specifically, the control unit 21 shoots the tablets in an arrangement form in which the dosing date is displayed in one of the columns or rows and the dosing time is displayed in the other in the dispensing detail area A36 of the inspection detail screen D31. Display an image.
- control unit 21 preliminarily displays the photographed images of the tablets in the packaging unit corresponding to the combination in a parallel direction in the order of the dosing timing at the display locations corresponding to each combination of the dosing date and the dosing time. Display them in the specified display order.
- the display order of the photographed images of the tablets is the same order as the order of the tablets displayed in the prescription information part A35.
- the dispensation detail area A36 displayed in this way when the determination result of the automatic inspection process is appropriate, the photographed images of the same type of tablets are arranged in the arrangement direction of the dosing days (left and right direction in FIG. 11). Will be lined up. Therefore, the pharmacist can easily confirm the suitability of each photographed image of the tablet.
- the background of the display area of the photographed image of the tablet causing the error is a fifth specific color such as red which is determined in advance. Is displayed. That is, the control unit 21 displays the photographed image of the tablet causing the error in the dispensing detail area A36 so that it can be identified. Thereby, the photographed image of the tablet is displayed in a package unit and the result of the automatic inspection process is displayed, so that the efficiency of the inspection operation of the pharmacist can be improved. In particular, the pharmacist can easily identify and check the photographed image of the tablet causing the error by referring to the display of the fifth specific color.
- a list of errors occurring in the dispensing detail area A36 is displayed.
- the control unit 21 displays a list of the causes of the error together with the taking date and the taking time of the tablet that caused the error.
- This processing is executed by the list display processing unit 212 of the control unit 21.
- the cause of the error displayed in the NG list area A37 is specified in any of the tablet packaging device 4, the dispensing device 5, and the server 2 based on the result of the automatic inspection process.
- the cause of the error is specified as “wrong mistake”.
- the cause of the error is identified as “lag behind”.
- the photographed images of the tablets contained in all the medicine packages 451 are displayed in the dispensing detail area A36.
- the automatic inspection is performed.
- the medication date corresponding to only the medicine package 451 for which the processing result is an error, the medication time, and the photographed image of the tablet are displayed, and the information of the medicine package 451 for which the result of the automatic inspection process is appropriate is not displayed. It is also possible. As a result, the user can easily grasp only the portion that needs to be confirmed by looking at the inspection detail screen D31, and can improve the efficiency of inspection work.
- the inspection detail screen D31 displays operation parts such as an operation key K33, an operation key K34, an input field K35, an operation key K36, and a check box K37 along with the dispensing detail area A36.
- the operation unit is an example of an operation unit for individually re-executing part or all of the dispensing process based on the prescription data executed by the tablet packaging device 4, and the display of the operation unit is the control This is executed by the operation display processing unit 213 of the unit 21.
- a packaging process is executed as the dispensing process
- the operation unit is an operation unit for re-executing a part or all of the packaging process.
- the operation key K33 is an example of a first operation unit for re-executing all the dispensing processes (packaging processes) corresponding to the records.
- the operation key K34 is a second operation unit for re-executing a part of the dispensing process (packaging process) determined to be an error in the automatic inspection process among the dispensing processes (packaging process) corresponding to the record. It is an example.
- the input field K35 and the operation key K36 re-execute a part of the dispensing process (packaging process) selected by the user operation on the input field K35 among the dispensing process (packaging process) corresponding to the record. It is an example of the 3rd operation part for.
- the input field K35 it is possible to specify the range (what number-what number) of the medicine package 451 to be re-executed, or specify only the specific medicine package 451, etc. It is. Thereby, as will be described later, the pharmacist selectively uses any two or three of the operation key K33, the operation key K34, the input field K35, and the operation key K36.
- the packaging apparatus 4 can arbitrarily execute part or all of the packaging processing.
- the check box K37 is an operation key for performing a setting not to re-execute the dispensing process of the medicine causing the error when the cause of the error is “lag behind”.
- the control unit 21 causes the error even when the operation key K33, the operation key K34, or the operation key K36 is operated. Re-execution is not performed for drugs that are “lag behind”. Thereby, for example, when an error of “rearward shift” occurs in the tablet packaging device 4, it is possible to cope with an operation in which the pharmacist manually moves the tablet between the preceding and following medicine packages 451.
- step S18 the control unit 21 determines whether an approval operation for the result of the automatic inspection processing of the prescription data has been performed. Specifically, the control unit 21 determines whether or not the approval key K31 displayed on the inspection screen D3 has been operated. If the control unit 21 determines that the approval operation has been performed (S18: Yes), the process proceeds to step 19, and if the approval operation has not been performed (S19: No), the process is performed. The process proceeds to S20.
- step S19 the control unit 21 executes an approval process for approving the result of the automatic inspection process of the prescription data.
- the control unit 21 indicates that the result of the automatic inspection process of the prescription data has been approved and identification information (name or ID) of the user who is the pharmacist who performed the approval operation. Then, the result of the automatic inspection process is stored in the storage unit 22 in association with the prescription data.
- step S20 the control unit 21 determines whether or not a full reissue operation has been performed on the inspection detail screen D31. Specifically, when the operation key K33 displayed on the inspection detail screen D31 is operated, it is determined that the all-reissue operation has been performed. Here, if the said control part 21 judges that the said all-reissue operation was performed (S20: Yes), it will transfer a process to step S21, and if the said all-reissue operation is not performed (S20: No) Then, the process proceeds to step S22.
- step S21 the control unit 21 performs a control instruction for re-executing the dispensing process corresponding to the record corresponding to the determination result, and the tablet packaging device 4 that has performed the dispensing process. Alternatively, it is transmitted to the dispensing device 5.
- prescription data for dispensing corresponding to the record is re-input to the tablet packaging device 4 or the dispensing device 5 as the control instruction.
- all the dispensing processes corresponding to the records are re-executed.
- the entire tablet packaging process corresponding to the record is re-executed.
- step S22 the control unit 21 determines whether or not a reissue operation for only NG packages has been performed on the inspection detail screen D31. Specifically, when the operation key K34 displayed on the inspection details screen D31 is operated, it is determined that the reissuing operation for only the NG package has been performed.
- the control unit 21 determines that the reissue operation for only the NG package has been performed (S22: Yes)
- the process proceeds to step S23, and the reissue operation for only the NG package must be performed. If (S22: No), the process proceeds to step S24.
- step S23 the control unit 21 issues a control instruction to re-execute only the dispensing process (NG package) in which an error has occurred among the dispensing corresponding to the record corresponding to the determination result. Then, the data is transmitted to the tablet packaging device 4 or the dispensing device 5 that has executed the dispensing process.
- the control instruction a part of the prescription data corresponding to the dispensing process in which the error has occurred among the prescription data for dispensing corresponding to the record is retransmitted to the tablet packaging device 4 or the dispensing device 5. Entered.
- step S24 the control unit 21 determines whether or not a reissue operation for only the designated package has been performed on the inspection detail screen D31. Specifically, when the operation key K36 displayed on the inspection details screen D31 is operated, it is determined that the reissuing operation for only the designated package has been performed.
- the control unit 21 determines that the reissue operation for only the designated package has been performed (S24: Yes)
- the process proceeds to step S25, and the reissue operation for only the designated package must be performed. If (S24: No), the process proceeds to step S26.
- step S25 the control unit 21 re-executes only the dispensing process (designated package) specified in the input field K35 among the dispensing corresponding to the record corresponding to the determination result.
- a control instruction is transmitted to the tablet packaging device 4 or the dispensing device 5 that has executed the dispensing process.
- some prescription data corresponding to the dispensing process specified in the input field K35 among the prescription data for dispensing corresponding to the record is the tablet packaging device 4 or the dispensing device 5. Will be re-entered.
- the said tablet packaging apparatus 4 or the said dispensing apparatus 5 only the one part dispensing process designated by the said input column K35 is re-executed.
- a part of the tablet packaging process corresponding to the record is re-executed.
- step S ⁇ b> 26> the control unit 21 determines whether a preset inspection end operation for ending the inspection support process has been performed. For example, the control unit 21 determines that the inspection end operation has been performed when an operation key corresponding to “end” on the inspection waiting list screen D1 is operated. Here, if the said control part 21 judges that the said inspection completion operation was performed (S26: Yes), the said inspection assistance process will be complete
- the control unit 21 executes the processing of steps S20 to S25, so that the dispensing that has already been performed in each of the tablet packaging device 4 and the dispensing device 5 is performed. Part or all of the processing can be individually re-executed by each of the tablet packaging device 4 and the dispensing device 5. This process is executed by the re-execution processing unit 214 of the control unit 21. Thereby, the work efficiency of the pharmacist in the hospital or pharmacy where the inspection support system 1 is used can be improved.
- the automatic inspection processing based on the tablet identification information read from the photographed image of the tablet photographed by the photographing unit 46 and the prescription data or the photographing unit 46 The automatic inspection process based on the photographed image of the tablet and the normal image is executed, and the packaging process is performed in units of the medicine package 451 by the user's arbitrary operation corresponding to the execution result of the automatic inspection process. Re-execution is possible. Therefore, the labor of the pharmacist who performs the final inspection of the medicine package 451 dispensed from the tablet packaging device 4 can be omitted, and the efficiency of the inspection operation of the pharmacist can be improved.
- the said control part 21 mainly demonstrated the case where the dispensing in the said tablet packaging apparatus 4 was re-executed
- the said control part 21 is a user with respect to the said inspection screen D3 or the said inspection screen D3. Any part or all of the dispensing performed by a plurality of dispensing devices such as the tablet packaging device 4 and the dispensing device 5 according to a user operation on the inspection detail screen D31 or the like displayed according to the operation. It can be re-executed.
- the control unit 21 can re-execute part or all of the powder packaging processing performed by the powder packaging device.
- the powder packaging device distributes the powder dispensed from the powder cassette by the feeder every time it is taken, a powder cassette containing powder, a feeder that automatically dispenses a predetermined amount from the powder cassette, A dispensing unit, and a packaging unit that wraps the powdered medicine distributed by the dispensing unit for each time of use with a packaging paper.
- the distribution unit includes, for example, a rotating disk on which a powder supplied from the feeder is placed, and a scraping member that scrapes the powder placed on the rotating disk by a predetermined amount.
- a predetermined amount of powder is supplied from the feeder in a state where the rotating disk is stopped, and the powder is scraped out by the scraping member, so that the packaging processing can be executed in a single package unit. is there.
- the control unit 21 performs a part of the packaging process corresponding to the record of the prescription data in accordance with a user operation on the inspection detail screen D31 or The whole can be re-executed by the tablet packaging device 4. At this time, it is conceivable that the control unit 21 has a repackaging collation function for collating the medicine package 451 re-executed by the tablet packaging device 4.
- the control unit 21 overlaps the inspection detail screen D31 and re-executes the dispensing as shown in FIG. It is conceivable to display a collation screen D32 that prompts reading of code information (one-dimensional code or two-dimensional code) described in the medicine package 451.
- code information one-dimensional code or two-dimensional code
- the control unit 21 The dispensing result of the record is changed to “corrected”. Thereby, when the inspection history screen D2 is displayed, “NG ⁇ corrected” is displayed for the record.
- the suitability of the code information is determined based on whether or not both the code information of the medicine package 451 before re-execution and the code information of the medicine package 451 after re-execution have been read.
- the suitability of the code information is determined based on the code of the medicine package 451 corresponding to before and after the medicine package 451 to be reexecuted among the plurality of medicine packages 451 obtained as a result of packaging corresponding to the record. It may be determined depending on whether or not information has been read. Thereby, the mistake of the replacement position of the said medicine package 451 before and behind re-execution is suppressed.
- both of the code information printed on one or both of the first and last medicine package 451 before re-execution and the code information printed on the medicine package 451 after re-execution are read. It is also conceivable that the determination is made depending on whether or not the operation is performed.
- the control unit 21 supplies the packaging unit 45 from the tablet cassette 41 or the hand-rolling unit 42 using the cameras 461 to 467 of the photographing unit 46 of the tablet packaging device 4. It is conceivable to have a progress monitor function capable of appropriately confirming the photographed image of the tablet photographed on the route of the tablet to be used. Specifically, the captured image of the tablet imaged by the imaging unit 46 is transmitted from the tablet packaging device 4 to the server 2 and stored in the storage unit 22 of the server 2.
- the control unit 21 is displayed on the operation screen such as the inspection screen D3 or the inspection detail screen D31, and when the operation key (not shown) corresponding to the progress monitoring function is operated, It is possible to display on the client terminal 4 a progress monitor screen D4 for displaying a photographed image of the tablet photographed by the cameras 461 to 467.
- the user can take a photographed image of the tablet at each time point that is photographed by the photographing unit 46 regarding the dispensing of the tablet in the tablet packaging device 4. It is possible to easily confirm the location and cause of the error.
- FIG. 13 is a diagram showing an example of the progress monitor screen D4.
- the progress monitor screen D4 shown in FIG. 13 displays an image display area A41 and a guidance display area A42 together with the basic information area A31 and the prescription information area A35.
- photographed images of tablets photographed by the cameras 461 to 467 of the photographing unit 46 are displayed in association with the identification codes of the cameras 461 to 467.
- a display area A411 where a photographed image of a tablet photographed by the camera 461 is displayed, and a display area where a photographed image of a tablet photographed by the cameras 464 to 466 is displayed.
- a 412 a display area A 413 where the photographed images of the tablets photographed by the cameras 462 and 463 are displayed, and a display area A 414 where the photographed images of the tablets photographed by the camera 467 are displayed.
- guidance display area A42 guidance information for indicating the shooting locations of the cameras 461 to 467 in the tablet packaging device 4 is displayed. Thereby, the user can easily grasp the position of the image displayed in the image display area A41.
- the said control part 21 memorize
- Prescription identification information such as a prescription ID is associated and stored.
- the said control part 21 is the said picked-up image corresponding to the designated prescription ID, when prescription ID of the said prescription data is designated even after completion
- the control unit 21 also has a hand check confirmation function for checking the state of the tablet after the hand making operation in the hand making unit 42. Specifically, when the tablet packaging apparatus 4 performs the tablet packaging using the hand-rolling unit 42, the tablet-handling unit 4 uses the camera 464 to perform the hand-rolling after the tablet-handling operation to the hand-rolling unit 42. An image of the unit 42 is taken.
- the control unit 40 uses the camera 464 when an operation key for inputting that the tablet manual operation to the manual operation unit 42 is completed is operated. Then, an image of the hand handling unit 42 is taken.
- the hand-rolling unit 42 is configured to be pulled out from the tablet packaging device 4 when used and stored in the tablet packaging device 4 after being tableted
- the camera 464 is provided inside the apparatus 4, and the control unit 40 causes the camera 464 to take an image of the manual handling unit 42 at the timing when the manual handling unit 42 is accommodated in the tablet packaging device 4. It is also possible to shoot.
- the tablet packaging device 4 transmits a hand-rolled image taken by the camera 464 to the server 2 together with information for identifying the prescription data. Accordingly, in the server 2, the handmade image is stored in the storage unit 22 in association with the prescription data by the control unit 21. The dispensing management device 2 may read the handmade image stored in the tablet packaging device 4.
- control unit 21 is currently displayed when an operation key (not shown) corresponding to the manual check monitor function is operated, which is displayed on the operation screen such as the inspection screen D3 or the inspection detail screen D31.
- the manual image corresponding to the prescription data is read from the storage unit 22.
- the control unit 21 causes the client terminal 3 to display a manual confirmation screen D5 on which the manual image is displayed.
- FIG. 14 is a diagram showing an example of the manual confirmation monitor screen D5.
- the basic information area A31 and the prescription information area A35, as well as the manual operation unit image area A51 and the operation explanation area A52 are displayed.
- the manual unit image area A51 the manual image obtained by photographing the manual unit 42 from above with the camera 464 is displayed. Thereby, the user can confirm the state immediately after hand-working the tablet on the hand-working unit 42.
- an explanation of the operation content in the manual unit image area A51 is displayed.
- the fact that the image is enlarged by the pinch-out operation and the image is reduced by the pinch-in operation is displayed together with the finger gesture and the image.
- the control unit 21 also has a cassette filling monitor function for confirming the state of the tablet filling operation of the tablet into the tablet cassette 41.
- the tablet packaging device 4 includes a workbench for filling tablets into the tablet cassette 41 and a camera for taking an image of the tablet cassette 41 placed on the workbench. It is possible.
- the work table is provided in front of the tablet packaging device 4 and, as shown in FIG. 15, when the tablet is filled in the tablet cassette 41, the work table is separated from the tablet packaging device 4. It is pulled out and can be stored in the tablet packaging device 4 when not in use.
- the control unit 40 uses the camera to control the tablet.
- An image of the cassette 41 is taken.
- the said tablet packaging apparatus 4 transmits the cassette image image
- the cassette image is stored in the storage unit 22 in association with the tablet by the control unit 21.
- the dispensing management device 2 may read the cassette image stored in the tablet packaging device 4.
- the control unit 21 performs an operation corresponding to the cassette filling monitor function displayed on the operation screen after a tablet to be displayed is selected on the operation screen such as the inspection screen D3 or the inspection detail screen D31.
- a key (not shown) is operated, the cassette image corresponding to the tablet is read from the storage unit 22.
- the control unit 21 causes the client terminal 3 to display a cassette filling monitor screen D6 on which the cassette image is displayed.
- the user checks the cassette image photographed by the camera, and the cause of the error is the tablet to the tablet cassette 41. It is possible to easily confirm whether or not this is a filling error.
- the control unit 21 shows the inspection result of the selected type of tablet in FIG. Displayed on the pop-up screen D25.
- the inspection result is displayed in such a manner that an erroneous tablet can be identified.
- the background region A251 is displayed in red on the tablet whose inspection result is an error.
- the control unit 21 displays a cassette filling monitor screen D6 that displays the cassette image corresponding to the tablet. .
- FIG. 15 shows an example of the cassette filling monitor screen D6.
- a cassette image area A61 is displayed together with the basic information area A31, the prescription information area A35, and the operation explanation area A52.
- the cassette image area A61 the cassette image obtained by photographing the tablet cassette 41 from above with the camera is displayed. Thereby, the user can confirm the filling state of the tablet into the tablet cassette 41.
- FIG. 26 is a view showing a cassette filling monitor screen D26 which is another example of the cassette filling monitor screen D6.
- the cassette image is displayed in the cassette image area A61 as in the cassette filling monitor screen D6, and the filling person who filled the tablet cassette 41 and the last filling date and time, etc. Is also displayed.
- FIGS. 16 and 17 are diagrams showing an inspection detail screen D301 which is another example of the inspection detail screen D31.
- FIG. 16 is a diagram showing the inspection details screen D301 when the inspection result is appropriate, and
- FIG. 17 is the inspection detail screen D301 when an error occurs in the inspection result.
- the inspection details screen D301 displays a basic information area A311, a dispensing details area A312 and an inspection result area A313.
- the basic information area A31 is an area in which patient information, prescription data, and the like are displayed in the same manner as the basic information portion A31.
- the dispensing detail area A312 is displayed instead of the dispensing detail area A36, and displays the contents of the tablets packaged in the medicine package 451 in units of the medicine package 451.
- the medicine name, type, result, and normal image are displayed.
- the type it is displayed so that it is possible to identify whether the tablet corresponding to the medicine name has been dispensed from either the tablet cassette 41 or the manual handling unit 42.
- the tablet dispensing source is the tablet cassette 41
- “C” or “K” is displayed as an identification code
- the handing unit (Detachable Tablet Adapter) 45 is “D”.
- “hand” or the like is displayed as an identification code.
- medical agent name is appropriate is displayed separately on the said result.
- the normal image is a registered image registered in advance in the medicine master or the like as a correct image of a tablet.
- a registered image registered in advance in the medicine master or the like as a correct image of a tablet.
- two images corresponding to the front and back surfaces of each tablet are registered as the registered images in association with each tablet.
- the registered image may be a single image in which an area including the identification information of the tablet is photographed on the outer peripheral surface of the tablet.
- a photographed image of the tablet photographed by the photographing unit 46 before being packaged in each medicine package 451 is displayed in association with each tablet.
- the control unit 21 is arranged in such a manner that the tablet name (medicine name) is displayed in one of the columns or rows and the packaging order (first package, second package,...) Is displayed on the other side, A photographed image of the tablet corresponding to each combination of the tablet name and the packaging order is displayed.
- the tablet name is displayed side by side in the up-down direction, and the packaging order is displayed in the left-right direction.
- the control unit 21 includes at least an original image obtained by reading the tablet identification information by the tablet packaging device 4 or an original image compared with the normal image, and the tablet packaging device 4 includes the original image.
- a plurality of photographed images corresponding to different outer peripheral surfaces of the tablets photographed for each tablet are displayed for each tablet. Thereby, for example, when it is difficult to determine the suitability of the tablet by using only the photographed image of one tablet, the pharmacist can determine the suitability by referring to the photographed images of the plurality of tablets.
- the dispensation detail area A312 since the original image of the tablet and the captured image of the tablet are displayed side by side, the pharmacist compares the registered image of the tablet with the captured image of the tablet to determine whether the tablet is appropriate. Can be easily determined.
- the said control part 21 acquires the identification information contained in the picked-up image of the said tablet, and detects the inclination of the character contained in the said identification information,
- the upper and lower sides of the character contained in the said identification information are the said inspection details.
- the photographed image of the tablet is rotationally corrected so as to coincide with the top and bottom of the screen D301 and displayed in the dispensing detail area A312. That is, the control unit 21 displays the photographed image of the tablet with the direction of characters in the identification information of the tablet aligned. Thereby, the user can easily confirm the identification information of the tablet together with the image of each of the tablets.
- the rotation correction of the photographed image of the tablet may be executed by the tablet packaging device 4.
- the scrolling key K311 is displayed in the dispensing detail area A312 and the control unit 21 controls the scroll key K311.
- information on the number of each package of the medicine package 451 displayed in the dispensing detail area A312 is scroll-displayed.
- the dispensing details area A312 together with the package number information such as the first package, the second package,... Indicating the number of packages of the medicine package 451, or instead of the package number information, May be displayed.
- the result of the automatic inspection process is an error among the medicines included in the record of the prescription data displayed on the inspection detail screen D301.
- the background of one or more regions A314 corresponding to the medicine is displayed in a sixth specific color such as a predetermined red color.
- the dispensing detail area A312 in FIG. 17 shows a case in which a medicine name, a captured image of a tablet, and a display area in the order of the medicine package 451 are set as the area A314. Furthermore, when the number of the medicine packages 451 exceeds the number that can be displayed simultaneously in the dispensing detail area A312, and there is a medicine whose result of the automatic inspection process is an error, the control unit 21 The scroll key K311 is automatically moved to display information corresponding to the medicine package 451 in which the error has occurred in the dispensing detail area A312.
- the result of the automatic inspection processing for all medicines included in the prescription data record displayed on the inspection details screen D301 is displayed.
- “OK” indicating that the result of the automatic inspection processing is appropriate is displayed in FIG. 16 together with a background of a seventh specific color such as a predetermined blue color
- FIG. “NG” indicating that the result of the inspection process is an error is displayed together with a background of an eighth specific color such as a predetermined red color different from the seventh specific color.
- the results of the inspection process are individually displayed for each tablet included in the record of the prescription data.
- the results of the inspection process are individually displayed for each tablet included in the record of the prescription data.
- “transamamine capsules 250 mg” and “cephalanthin tablets 1 mg” “ ⁇ ” indicating that the result of the inspection process is appropriate is displayed, and “Reflex tablets 15 mg” "” Is displayed as "x” indicating that an error has occurred in the result of the inspection process.
- the pharmacist can grasp
- the control unit 21 captures the photographed image of the tablet that is the target of the selection operation as shown in FIG. It is possible to display on the client terminal 3 an enlarged screen D33 for enlarging and displaying as an enlarged image P331. Further, on the enlarged screen D33, an enlarged image obtained by enlarging the normal image corresponding to the tablet that is the target of the selection operation and the enlarged image P331 may be displayed side by side vertically or horizontally. Note that the enlargement screen D33 displays the packaging order (-packet) of the medicine package 451 in which the tablets selected by the selection operation are packaged.
- the control unit 21 performs the packaging order of the medicine packages 451 on the tablets displayed on the enlarged screen D33 in response to a predetermined user operation such as a vertical or horizontal flick operation on the enlarged screen D33. It is possible to change according to For example, when the tablet packaged in the seventh medicine package 451 is displayed on the enlarged screen D33, the sixth or eighth medicine package according to the predetermined user operation The tablets packaged in 451 are displayed. In addition, it is also conceivable that the control unit 21 sequentially displays only the tablets causing the error in response to the predetermined user operation.
- control unit 21 switches the medicine pack 451 to be displayed according to one flick operation in the vertical direction and the left / right direction with respect to the enlarged screen D33, and selects the type of tablet to be displayed according to the other flick operation. Switching to another packaged tablet is also conceivable.
- FIGS. 19 and 20 are diagrams showing an inspection detail screen D302 which is another example of the inspection detail screen D31.
- FIG. 19 is a diagram showing the inspection detail screen D302 when the inspection result is appropriate, and
- FIG. 20 is the inspection detail screen D302 when an error occurs in the inspection result.
- the inspection details screen D302 displays the basic information area A311 and the inspection result area A313 similar to the inspection details screen D301, and also in the dispensing details area A312. Instead, a dispensing detail area A322 is displayed.
- the medicine name, type, result, and normal image are displayed as in the dispensing detail area A312.
- the dispensing detail area A322 unlike the dispensing detail area A312, only one image including the identification information of the tablet is displayed as a photographed image of the tablet. Thereby, the number of packages that can be displayed simultaneously in the dispensing detail area A322 can be increased. This is because, in general, the identification information of the tablet can be confirmed when the outer peripheral surface of the medicine is viewed from one direction, and an image of the other outer peripheral surface is often unnecessary. .
- the control unit 21 captures an image of the tablet that is the target of the selection operation as illustrated in FIG. Can be enlarged and displayed on the client terminal 3 as an enlarged image P341.
- the control unit 21 displays, on the enlarged screen D34, an enlarged image P342 obtained by enlarging an image including the identification information in the normal image corresponding to the tablet that is the target of the selection operation, together with the enlarged image P341. Display.
- the arrangement direction of the enlarged image P341 and the enlarged image P342 may be either the vertical direction or the horizontal direction. Further, as the enlarged image P342, a plurality of images registered in advance as the normal image corresponding to the tablet that is the target of the selection operation may be displayed.
- the control unit 21 may alternately display a plurality of captured images (for example, a front surface image and a back surface image) of the tablet in response to a selection operation of the captured image of the tablet in the dispensing detail area A322. It is done. Thereby, it is possible to confirm the state where the tablet is viewed from a plurality of directions while suppressing the size of the display area at the normal time.
- a plurality of captured images for example, a front surface image and a back surface image
- the control unit 21 displays the photographed images of the tablets corresponding to the plurality of medicine packages 451 delivered in a series of packaging processes in the order of the first package, the second package,.
- the control unit 21 arranges the arrangement order of the medicine packages 451 to be displayed for each dosing period (see FIG. 27), and the second array that arranges the dosing day and the dosing period in order (FIG. 27). 28) can be arbitrarily selected according to a user operation or the like.
- the control unit 21 switches between the first array and the second array in accordance with, for example, a setting operation in an initial setting of the server 2 or the tablet packaging device 4.
- the medicine package 451 corresponding to nine times of morning, noon, and night between July 1 and July 3 is paid out based on one record of the prescription data.
- the first arrangement as shown in FIG. 27, the morning of July 1, the morning of July 2, the morning of July 3, the day of July 1, the day of July
- the shot images of tablets packaged in the medicine package 451 are displayed side by side in the order of noon, noon on July 3, night on July 1, night on July 2, and night on July 3. Is done.
- the second arrangement as shown in FIG. 28, the morning of July 1, the day of July 1, the night of July 1, the morning of July 2, the day of July 2.
- the shot images of the tablets packaged in the medicine package 451 are displayed side by side in the order of night of July 2, morning of July 3, noon of July 3, and night of July 3.
- the display mode of the inspection detail screen D301 or D302 can be changed in accordance with the operation in the medical institution where the inspection support system 1 is used, and a system configuration excellent in versatility is realized.
- the inspection object in the second inspection mode is the first and last one or both of the medicine packages 451 packaged based on the prescription data.
- the inspection target in the second inspection mode is the first and last one or both of the medicine packages 451 corresponding to the change of the dosing pattern in the prescription data.
- the inspection target in the second inspection mode is the medicine package 451 in which the supply source of the contained tablet is a preset supply source (for example, the handmade unit 42 or the specific tablet cassette 41). Also good.
- the second inspection mode is applied to the medicine package 451 in which at least one type of tablet whose supply source is the manual handling unit 42 or the specific tablet cassette 41 is included.
- the switching between the first inspection mode and the second inspection mode is not limited to the unit of the medicine pack 451, but may be a unit of a series of the medicine packs 451 packaged based on one prescription data. Good.
- the second inspection mode the case where a part of the tablets of the medicine package 451 is to be inspected has been described as an example.
- the second inspection mode when tablets are continuously dispensed from the same tablet cassette 41 in the packaging process based on the prescription data, only the first dispensed tablet from the tablet cassette 41 May be subject to inspection.
- the tablet packaging device 4 has a third inspection mode in which the presence or absence of the automatic inspection processing is automatically switched by the control unit 40.
- the automatic inspection process is not executed for the tablets dispensed from the tablet cassette 41 in the packaging process, and the automatic inspection process is executed for the tablets dispensed from the manual handling unit 42.
- the manual handling unit 42 that is highly prone to human error is used
- the automatic inspection process is executed to suppress dispensing errors, and the manual cassette unit 42 is not used and the tablet cassette 41 is used.
- the automatic inspection process is not executed and the time required for the packaging process is shortened.
- the tablet imaging by the camera 462 or 463 of the imaging unit 46 may be executed, but may be omitted.
- the inspection method is changed depending on whether or not the handing unit 42 is used for the packaging process.
- the control unit 40 when the supply source of at least one type of tablets among the tablets included in the prescription data to be subjected to the packaging process is the handing unit 42, that is, in the packaging process,
- an image inspection process for collating the tablet identification information acquired from the photographed image with the prescription data is executed as the automatic inspection process.
- the said control part 40 when performing the said image inspection process is an example of an image inspection process part here.
- the control unit 40 performs a count inspection process for comparing the number of dispensed tablets and the prescription data. Execute.
- the said control part 40 when performing the said count inspection process here is an example of a count inspection process part.
- the control unit 40 detects the number of tablets contained in the medicine package 451 for each medicine package 451 using the passage detection sensor 475.
- the said control part 40 when detecting the number of the said tablets using the said passage detection sensor 475 is an example of a tablet number detection part.
- the passage detection sensor 475 is disposed at a position where the tablet is detected at a timing after the tablet is dispensed from the rotating unit 44 and before being stored in the medicine package 451. That is, the control unit 40 detects the number of tablets using the passage detection sensor 475 on the downstream side in the tablet dispensing direction from the position where the tablets are photographed by the cameras 462 and 463.
- the medicine package 451 is photographed with a camera or the like, and the control unit 40 detects the number of the tablets contained in the medicine package 451 from the photographed image. It is also possible to do. Then, the control unit 40 collates the number of tablets to be accommodated in the medicine package 451 with the number of tablets detected by the passage detection sensor 475 based on the prescription data.
- the image inspection process is executed as the automatic inspection process, and dispensing errors are suppressed with high accuracy.
- the time required for the packaging process is shortened because the image inspection process is not executed, and the counting inspection process is executed. As a result, dispensing errors are also suppressed.
- the tablet packaging device 4 does not execute the image inspection process as the automatic inspection process when a malfunction occurs in the components such as the camera 462 or 463 used for the image inspection process. It is also conceivable to have a failure mode for executing the counting inspection process as one of the inspection modes. For example, when a defect is detected in a component used for the image inspection process, the control unit 40 displays an operation screen for selecting whether or not to switch to the failure mode, and according to a user operation It is conceivable to execute the presence / absence of switching to the failure mode. As a result, even if a defect occurs in the component for executing the image inspection process of the tablet packaging device 4, the normal packaging process is performed using the tablet packaging device 4. It is possible.
- the failure mode may be an inspection mode in which both the image inspection process and the count inspection process are not executed.
- the tablet having the same identification information attached to the front surface and the back surface is the same among the front surface and the back surface of the tablet. It is conceivable to have a display change function for enlarging one of the photographed images within the region where the photographed images on the front and back surfaces are displayed and displaying them on the inspection detail screen D301.
- control unit 21 determines whether the identification information on the front surface and the back surface of the tablet is the same by comparing the identification information of the tablet acquired from the front surface and the back surface of the tablet, respectively.
- information on whether the front and back surfaces are the same is registered in advance in the medicine master, and the control unit 21 stores medicine information on the tablet included in the prescription data as a payout target. It is also conceivable to determine whether the front and back surfaces of the tablet are the same based on the drug master.
- the said control part 21 displays the picked-up image corresponding to both the front and back of the said tablet on the said inspection detailed screen D301 about the tablet from which the said identification information attached
- the inspection support system 1 includes an error-time image display function for displaying a plurality of types of captured images obtained by photographing the tablets that are the target of the error from different angles. It is possible. Below, the case where the said server 2 is equipped with the said image display function at the time of an error is demonstrated first, and the case where the said tablet packaging apparatus 4 is equipped with the said image display function at the time of an error is demonstrated in a back
- the control unit 21 uses the tablet packaging device 4 as the captured image of the tablet that causes the error. A plurality of photographed images corresponding to different outer peripheral surfaces of the tablets photographed for each tablet can be displayed for each tablet. Such display processing is executed by the inspection display processing unit 211 of the control unit 21.
- photographing by the camera 462 and the camera 463 is performed at a preset photographing interval (for example, every several ms). Therefore, a plurality of photographed images obtained by photographing a plurality of different regions on the outer peripheral surface of the tablet are obtained as the photographed images of the tablet rotated by the tablet rotating unit 441 provided in the rotating unit 44.
- the imaging interval is an interval set in advance so that a plurality of different areas on the outer peripheral surface of the tablet are imaged by the camera 462 and the camera 463.
- the identification information of the tablet read from the photographed image is used, the identification information of the tablet is read from any of a plurality of photographed images of the tablet photographed for each tablet, It is determined whether the identification information matches the prescription data. For example, identification information formed by marking or printing for each tablet is registered in the pharmaceutical master, and in the automatic inspection process, identification information of the tablet read from the pharmaceutical master and the captured image The drug name of the tablet is specified based on the above, and it is determined whether or not the drug name matches the drug name of the tablet included in the prescription data. Further, in the automatic inspection process performed using the photographed image and the normal image, whether any of the plurality of photographed images of the tablets photographed for each tablet matches the regular image. No or the degree of coincidence is determined.
- the control unit 40 obtains the identification information of the tablet from the tablet among the photographed images of the tablet photographed by the camera 462 or the camera 463.
- One or a plurality of photographed images including the original image when read or the original image compared with the normal image are transmitted to the server 2 together with the result of the automatic inspection process.
- the control unit 40 has a plurality of different images on the outer peripheral surface of the tablet among the photographed images of the tablet photographed by the camera 462 or the camera 463.
- a plurality of photographed images in which areas are photographed are transmitted to the server 2 together with the result of the automatic inspection process. Note that all the captured images of each tablet may be transmitted from the tablet packaging device 4 to the server 2.
- the error of the automatic inspection process includes the first error that the tablet specified by the identification information included in the captured image does not match the tablet included in the prescription data, and the identification information included in the captured image.
- a second error that does not match the identification information of all tablets registered in the pharmaceutical master is included. Therefore, for the tablet whose result of the automatic inspection process is the first error, the control unit 40 uses the tablet when the identification information is read, as in the case where the result of the automatic inspection process is appropriate. It is conceivable that the captured image or the captured image compared with the normal image is transmitted to the server 2.
- the said control part 40 is several picked-up image by which several different area
- the control unit 21 transmits the image to the display unit 24 of the server 2 or the display unit 34 of the client terminal 3.
- the inspection details screen D301 see FIG. 17
- the inspection details screen D302 see FIG. 20
- the control unit 21 causes the storage unit 22 to store each photographed image acquired from the tablet packaging device 4 in association with the prescription data together with the result of the automatic inspection process.
- the control unit 21 reads the identification information of the tablets for the tablets for which the result of the automatic inspection processing is appropriate or the first error is detected. At least a photographed image compared with the normal image is displayed, and for a tablet whose result of the automatic inspection process is the second error, any photographed image of the photographed image of the tablet is displayed. It can be displayed. For example, for the tablet whose result of the automatic inspection process is the second error, the control unit 21 displays one or both of the first photographed image and the last photographed image among the photographed images of the tablet. It is possible.
- control unit 21 includes identification information having a high degree of pattern matching coincidence with the identification information of the tablet in the captured image of the tablet for the tablet in which the result of the automatic inspection process is the second error. It is also conceivable to display a photographed image to be displayed or to display a photographed image in which characters are detected among the photographed images of the tablets.
- control unit 21 performs a user operation on the inspection detail screen D301 or the inspection detail screen D302 to select any one of the photographed images of the tablets for which the result of the automatic inspection processing is appropriate.
- the enlarged image of the photographed image of the tablet is displayed on the enlarged screen D33 (see FIG. 18) and the enlarged screen D34 (see FIG. 21).
- the user operation is, for example, an operation of selecting a photographed image of the tablet for which the result of the automatic inspection process is appropriate on the inspection details screen D301 or the inspection details screen D302.
- the control unit 21 performs the user operation for selecting any one of the photographed images of the tablets in which the result of the automatic inspection processing is an error. Then, an error detail screen D35 on which a plurality of photographed images about the tablet is displayed is displayed.
- the user operation is, for example, an operation of selecting a photographed image of the tablet in which the result of the automatic inspection process is an error on the inspection details screen D301 or the inspection details screen D302.
- the control unit 21 does not use the error details screen D35 for the tablet whose result of the automatic inspection process is the first error, but similar to the case where the result of the automatic inspection process is appropriate. It is also conceivable to display the enlarged screen D33 or the enlarged screen D34.
- the control unit 21 may display on the error detail screen D35 whether the error is the first error or the second error. Conceivable. For example, when the result of the automatic inspection process is the first error, the control unit 21 reads from the photographed image of the tablet together with a message such as “drug type error” indicating that the error is the first error. It is conceivable to display the drug name of the tablet corresponding to the identified identification information on the error detail screen D35. In addition, when the result of the automatic inspection process is the second error, the control unit 21 displays a message such as “unknown drug type” indicating that the error is the second error on the error detail screen D35. It is possible.
- FIG. 22 is a diagram showing an example of the error detail screen D35.
- the result of the automatic inspection process is an error together with the drug name of the tablet selected on the inspection detail screen D301 or the inspection detail screen D302.
- a plurality of captured images P351 to P353 in which different regions on the outer peripheral surface of the tablet are captured are displayed as captured images of the tablets.
- the control unit 21 has a predetermined number of captured images continuously captured at the capturing interval, or a plurality corresponding to a predetermined interval. Is displayed on the error detail screen D35, and the captured images of different regions on the outer peripheral surface of the tablet are displayed.
- the control unit 21 captures the captured image when the identification information is read on the error detail screen D35 or the normal image. It is conceivable to display one or a plurality of photographed images including the photographed image compared with. Thereby, the pharmacist can determine suitability by referring to the captured images of the plurality of tablets displayed on the error detail screen D35, for example.
- rotation correspondence information indicating a correspondence relationship between the rotation amount (rotation time) of the roller 100 in the tablet rotation unit 441 and the rotation amount of the tablet is stored in the storage unit 22, and the control unit 21 includes: Based on the rotation correspondence information and the photographing interval, it is conceivable to select a plurality of photographed images in which different regions on the outer peripheral surface of the tablet are photographed. Specifically, when the photographing by the camera 462 or the camera 463 is performed every 1 ms, and it is known that the tablet rotates 10 ° every 10 ms in the rotation correspondence information, the control unit 21 may select a plurality of captured images captured every 45 ms, or select a plurality of captured images captured every 90 ms.
- the said control part 21 selects the some picked-up image from which the attitude
- the control unit 21 when the control unit 21 includes an error in the result of the automatic inspection process, a plurality of captured images in which different areas on the outer peripheral surface of the tablet are captured are displayed.
- the error detail screen D35 can be displayed. Therefore, when the result of the automatic inspection process is an error, the pharmacist can grasp the reason for the error by referring to the plurality of captured images.
- the number of captured images displayed on the error detail screen D35 is not limited to this.
- the control unit 21 sequentially displays a plurality of captured images of the tablet one by one or a plurality of sheets in response to a flick operation in the horizontal direction or the vertical direction with respect to the error detail screen D35.
- a slide bar that can be slid in the horizontal direction or the vertical direction is displayed on the error detail screen D35, and the control unit 21 selects a plurality of captured images of the tablet according to the slide operation of the slide bar. It is also conceivable to sequentially display every sheet or every plurality of sheets.
- FIG. 23 is a diagram showing an example of the enlarged screen D36. Specifically, in the enlarged screen D36 shown in FIG. 23, as in the enlarged screen D34 (see FIG. 18), the captured image P361 of the selected tablet and the enlarged image of the normal image corresponding to the tablet are displayed. P362 is displayed side by side in the vertical direction or the horizontal direction. Note that the enlarged image P362 may be displayed together with the captured images P351 to P353 on the error detail screen D35.
- control unit 21 sets the error for the tablet in response to a user operation performed after displaying a photographed image of the tablet that caused the error in the automatic inspection process.
- a releasable release processing unit 215 is included.
- the cancellation processing unit 215 can cancel the error and record user identification information when the user operation is performed in association with the prescription data.
- said control part 21 functions as the said cancellation
- the error detail screen D35 and the enlarged screen D36 display an error cancel key K351 for canceling an error that is a result of the automatic inspection process.
- release key K351 is operated, the said control part 21 will cancel
- the automatic inspection process for the prescription data is performed by operating the error release key K351. Even when an error has occurred, the inspection of the prescription data can be normally terminated.
- the control unit 21 indicates that the error has been canceled and the identification information (name or ID) of the pharmacist who is currently logged in as the automatic inspection. Along with the result of processing, it is recorded in the storage unit 22 in association with the prescription data. Thereby, the said control part 21 becomes possible [outputting the information of the user who canceled the error among the results of the above-mentioned automatic inspection processing about the above-mentioned prescription data] (display).
- the control unit 21 associates information such as the date and time when the error cancellation key K351 is operated or an arbitrarily input error cancellation reason with the prescription data together with the result of the automatic inspection process. Recording in the storage unit 22 is also conceivable.
- the control unit 21 When the error cancel key K351 is operated, the control unit 21 confirms that the confirmation operation for the error has been performed without canceling the error (the determination result of the user) and the confirmation operation. It is also conceivable that the identification information of the executed pharmacist is recorded in the storage unit 22 in association with the prescription data together with the result of the automatic inspection process. Thereby, the control unit 21 determines the result of the automatic inspection process in the tablet packaging device 4, the determination result of the user as to whether or not the pharmacist has performed the confirmation operation, and the identification information of the pharmacist who performed the confirmation operation. It is possible to output (display) information.
- the control unit 21 determines the inspection image of the image of the photographic image stored in the storage unit 22 for the photographic image of the tablet in which the error is canceled by the operation of the error release key K351 or the like. It is conceivable to delete at the end of the support process (see FIG. 7) (S26: Yes). Thereby, the storage capacity that needs to be secured in the storage unit 22 can be suppressed.
- control unit 21 selects only one or a plurality of photographed images arbitrarily selected by a user operation on the error detail screen D35 from among the photographed images of the tablets whose error has been canceled by the operation of the error cancel key K351. It is also conceivable to store in the storage unit 22 and delete other captured images.
- the tablet packaging device 4 includes an operation display unit 48 such as a liquid crystal display and a touch panel, and a storage unit 49 such as a hard disk or an SSD. Then, like the control unit 21, the control unit 40 displays, on the operation display unit 48, the inspection details screens D301 and D302, the enlarged screens D34 and D35, the error details screen D35, the enlarged screen D36, and the like. Can be displayed. That is, when the result of the automatic inspection process is an error, the control unit 40 was photographed for each tablet in the tablet packaging device 4 as a photographed image of the tablet that caused the error.
- the operation display unit 48 it is possible to cause the operation display unit 48 to display the error detail screen D35 in which a plurality of captured images corresponding to different outer peripheral surfaces of the tablets are displayed for each tablet.
- the said control part 40 when performing the process which concerns here is an example of an inspection display process part.
- the said control part 40 is a tablet when the said identification information is read similarly to the case where the result of the said automatic inspection process is appropriate about the tablet whose result of the said automatic inspection process is the said 1st error. It is conceivable that the photographed image of the tablet or the photographed image of the tablet compared with the normal image is displayed on the operation display unit 48. And about the tablet whose result of the said automatic inspection process is the said 2nd error, the said control part 40 is several picked-up image by which several different area
- control unit 40 sequentially displays a plurality of photographed images of the tablet one by one or a plurality of sheets in response to a flick operation in the horizontal direction or the vertical direction with respect to the error detail screen D35.
- a slide bar that can be slid in the left-right direction or the up-down direction is displayed on the error detail screen D35, and the control unit 40 selects a plurality of captured images of the tablet according to the slide operation of the slide bar. It is also conceivable to sequentially display every sheet or every plurality of sheets.
- the control unit 40 indicates that the error has been cancelled, and identification information (name or ID) of a pharmacist who is currently logged in to the tablet packaging device 4. Is recorded in the storage unit 49 in association with the prescription data together with the result of the automatic inspection process. Further, when the control unit 40 transmits the result of the automatic inspection process and the photographed image of the tablet to the server 2, the control unit 40 also transmits information indicating that the error has been canceled and identification information of the pharmacist. Thereby, in the server 2, when the control unit 21 outputs (displays) the result of the automatic inspection processing for the prescription data, the fact that the error has been canceled and the identification information of the pharmacist are output. (Display).
- the control unit 40 confirms that the error has been confirmed without releasing the error, and the pharmacist who has performed the confirmation operation. It is also conceivable that the identification information is recorded in the storage unit 49 in association with the prescription data together with the result of the automatic inspection process. Thereby, the control unit 40 outputs (displays) information such as the result of the automatic inspection process in the tablet packaging device 4, the presence / absence of the confirmation operation by the pharmacist, and the identification information of the pharmacist who performed the confirmation operation. It becomes possible to do. For example, when transmitting the result of the automatic inspection process and the photographed image of the tablet to the server 2, the control unit 40 also transmits that the confirmation operation for the error has been performed and the identification information of the pharmacist. .
- the control unit 40 stores the result of the automatic inspection process for the prescription data and the data of the photographed image in the storage unit 49.
- the control unit 21 uses the photographed image data stored in the storage unit 22, the photographed image of the tablet with the normal result of the automatic inspection process, the operation of the error release key K351, and the like.
- the photographed image for which the error has been canceled by the above is deleted at the end of the inspection support process (see FIG. 7) (S26: Yes).
- the storage unit 22 of the server 2 or the storage unit 49 of the tablet packaging device 4 stores the result of the automatic inspection process and the data of the photographed image. It is conceivable that the result of the automatic inspection process and the data of the photographed image are erased at the end of the inspection support process.
- the printing unit 453 may print character information such as a patient's name, medication time, medicine name, or prescription amount on the medicine package 451.
- the medicine package 451 may be printed with a one-dimensional code or a two-dimensional code for a medicine inspection code for determining the suitability of the packaging processing when the medicine package 451 is packaged.
- the tablet packaging device 4 has a print inspection function for automatically inspecting whether or not the printing on the medicine package 451 by the printing unit 453 is normally performed.
- the tablet packaging device 4 includes an image reading unit 50 that reads an image of the medicine package 451.
- the image reading unit 50 is arranged on the downstream side in the movement direction of the medicine package 451 in the packaging unit 45 from the position where the printing unit 453 performs printing on the medicine package 451. More specifically, the image reading unit 50 is on the downstream side in the movement direction of the medicine package 451 with respect to the position where printing on the medicine package 451 is performed by the printing unit 453, and the tablet on the medicine package 451. Is provided at a position before being accommodated.
- the image reading unit 50 may be provided at a position after the tablet is accommodated in the medicine package 451.
- the image reading unit 50 includes an image sensor such as a CCD or CIS that reads an image from the medicine package 451. Then, the control unit 40 acquires character information printed on the medicine package 451 by a known character recognition process (OCR) or the like based on the image read by the image reading unit 50. In addition, when the drug code is included in the image read by the image reading unit 50, the control unit 40 reads drug information from the drug code.
- OCR character recognition process
- the control unit 40 reads the character information or drug information that is the print contents of the medicine package 451 read by the image reading unit 50 and the character information or drug information that should be printed on the medicine package 451. And match. And if the said collation result does not correspond, the said control part 40 will display that on the said operation display part 48, and will alert
- the print content collation process is not limited to the tablet packaging device 4 and may be executed by the server 2. Specifically, an image of the medicine package 451 read by the image reading unit 50 is input from the tablet packaging device 4 to the server 2. Then, the control unit 21 of the server 2 is printed on the medicine package 451 based on the image of the medicine package 451 and the prescription data read by the image reading unit 50 in the tablet packaging device 4. Judge the suitability of the information.
- the packaging processing is performed in the tablet packaging device 4. It is necessary to execute again. At this time, when the medicine package 451 includes a plurality of tablets and only a part of the tablets is inspected by the automatic inspection process, the inspection result is appropriate. The tablets will be wasted. Or the operation
- the control unit 40 when the control unit 40 re-executes the packaging process for the medicine package 451 including the tablet whose inspection result is an error, the inspection result is already stored in the medicine package 451 already packaged. It is determined whether or not an appropriate tablet is included. Then, when a tablet whose inspection result is appropriate is included, the control unit 40 causes the operation display unit 48 to display a guidance notification that prompts the user to insert the tablet into the hand-rolling unit 42. For example, in the guidance notification, guidance about which tablet should be put into which position among the tablets contained in the medicine package 451 is displayed as an image or text.
- the control unit 40 executes the packaging process using the tablets placed in the handing unit 42 when the packaging process for the medicine package 451 is re-executed. Thereby, the tablet with the proper inspection result can be used effectively without wasting it, and the user does not need to return the tablet to the tablet cassette 41.
- the type of tablet whose inspection result is not appropriate is supplied from the tablet cassette 41 when the tablet cassette 41 corresponding to the tablet exists.
- the tablet with the inspection result being appropriate is input to the handing unit 42. Is displayed, and the packaging process is executed by using the manual handling unit 42.
- the control unit 40 executes the automatic inspection process and transmits the inspection result and the photographed image of the automatic inspection process to the server 2.
- the control unit 21 displays the inspection result of the automatic inspection process for the re-executed packaging process on the inspection detail screen D31, D301 or D302.
- the control unit 21 sequentially, in the inspection details screen D31, D301, or D302, automatically or in response to a user operation, the inspection results of the automatic inspection processing executed in the past for the same prescription data. It may be possible to switch and display.
- control unit 21 may rewrite and display only the tablets for which the packaging process has been re-executed among the past inspection results of the automatic inspection process with the latest inspection results of the automatic inspection process. Even in this case, it is conceivable that the control unit 21 accumulates the inspection results of the automatic inspection process for the same prescription data and stores them in the storage unit 22. Thereby, in the said server 2, it is possible to refer the log
- Capsule image processing function When the tablet dispensed in the packaging process is a capsule tablet, the automatic inspection process is executed based on the identification information attached to the left and right capsule pieces constituting the capsule tablet Is done. However, when there is identification information stamped or printed on the left and right capsule pieces constituting the capsule lock, the position of the identification information may be shifted in the circumferential direction.
- the control unit 40 has a capsule image processing function for acquiring a captured image (divided image) of each capsule piece of the capsule tablet when the tablet is a capsule tablet. Can be considered.
- the control unit 40 determines whether or not the tablet is a capsule tablet in accordance with the transition of the area of the tablet in the plurality of captured images obtained by capturing the tablet. For example, when the ratio of the maximum value and the minimum value of the area of the tablet in each of the captured images is equal to or less than a preset threshold value, that is, when the amount of change in the area of the tablet is extremely small, the tablet is a capsule. It can be considered to be a lock.
- the tablet is determined to be a capsule tablet when there is a line segment for dividing the tablet in each of the photographed images or when the left and right images of the tablet have different colors. It is also possible.
- the control unit 40 may determine whether or not the tablet is a capsule tablet based on the drug master or the drug database.
- the image of the said capsule tablet in each said picked-up image is divided
- the divided image is an image obtained by dividing the captured image along a line segment indicating a boundary line between the capsule pieces or along the center of the capsule lock.
- the control unit 40 verifies the suitability of the capsule lock based on the divided images of the capsule pieces.
- a divided normal image divided into left and right as a normal image of the capsule tablet, or a divided normal image divided into four vertically and horizontally is stored, and the control unit 40 It is conceivable to collate the divided image obtained by dividing the captured image of the capsule lock into left and right, or divided into four divided vertically and horizontally with the divided normal image.
- the identification information of the tablets included in the divided image may be individually read and compared with the identification information of the tablets included in the divided correct image to check the suitability of the capsule tablets.
- control unit 40 together with the result of the automatic inspection process, the divided image compared with at least the divided normal image among the divided images of the capsule pieces, or the identification information when the identification information is read.
- the divided image is transmitted to the server 2.
- the control unit 21 generates a composite image by synthesizing the divided images including identification information of each capsule piece of the capsule lock as a captured image of the capsule lock, It is conceivable to display the composite image on the inspection details screen D302. Thereby, the user can confirm the identification information of each capsule piece of the capsule lock on the inspection detail screen D302 by the composite image.
- the control unit 21 determines that the capsule lock image in which identification information on the right side of the capsule lock exists and the capsule lock It is also conceivable to display on the inspection detail screen D301 an image of the capsule lock in which identification information on the left side is present.
- FIG. 61A is a diagram showing an example of a captured image of a capsule tablet in which “Altat 75” and “TZ321” are formed along the circumferential direction as characters of the identification information in the left and right capsule pieces. As shown in FIG. 61A, in each of the captured images of the capsule lock, only a part of the characters of the identification information of the capsule lock appears.
- the control unit 40 combines a plurality of captured images corresponding to different positions in the circumferential direction of the capsule lock. It is conceivable to generate a composite image and determine the suitability of the capsule lock using the composite image. Hereinafter, this point will be described.
- the medicine master as the identification information corresponding to each capsule piece of the capsule tablet, a divided normal image including the characters “Altat 75” and a divided normal image including the characters “TZ321” are stored. It is assumed that
- control unit 40 performs a distortion correction process, a combined feature point extraction process, an image combining process, and a boundary for a plurality of captured images of the capsule lock captured by the camera 462 or 463 in the packaging process. Perform removal processing.
- the control unit 40 expands each captured image in the short direction of the capsule lock to correct the character distortion of the identification information. More specifically, the contraction degree of the character of the identification information in the short direction increases as it approaches the end from the center in the short direction of the capsule lock. Therefore, in the distortion correction processing, the control unit 40 follows the weighting set in advance based on the diameter of the capsule lock or the like, and the character of the identification information becomes closer to the end from the center in the short direction of the capsule lock.
- the correction processing of the photographed image is executed so that the degree of expansion in the short side direction becomes large.
- the control unit 40 sets target points at intervals that gradually narrow from the center in the short direction of the capsule lock in the captured image toward the end, so that the target points are equally spaced. It is conceivable to expand the photographed image. Thereby, for example, as shown in FIG. 61B, a photographed image in which the distortion of the character of the identification information is corrected is obtained.
- the control unit 40 trims an image of a preset necessary range including the identification information from each of the captured images after the distortion correction process, and the trimming is performed.
- feature points for synthesizing each photographed image are extracted.
- the control unit 40 generates a composite image by sequentially superimposing and synthesizing the captured images based on the feature points.
- a combined image obtained by combining the captured images is obtained.
- the control unit 40 executes a process for removing a boundary line generated by combining the captured images from the synthesized image.
- the boundary line is removed using a well-known gradient domain method.
- FIG. 61D a captured image in which the character distortion of the identification information is corrected is obtained.
- the control unit 40 determines the suitability of the capsule lock by executing the image inspection process using the composite image as the captured image. Specifically, the control unit 40 extracts a character area (for example, areas A41 and A42 in FIG. 61D) from the composite image, and performs the identification information by matching each of the image of the character area and the divided normal image. The suitability of the characters is determined. Thereby, in the said tablet packaging apparatus 4, it becomes possible to judge the suitability of the said capsule tablet by the said image inspection process also about the capsule tablet in which the character of identification information is formed in the circumferential direction.
- a character area for example, areas A41 and A42 in FIG. 61D
- control unit 21 does not include the identification information of the tablet in at least one of the two captured images of the tablet on the inspection detail screen D301, the imaging is performed. It is conceivable to have an abbreviated display function for omitting the display of an image and enlarging and displaying the other captured image. In particular, it is conceivable to exclude the information of the specific identification information set in advance from each of the photographed images of the tablets on the inspection detail screen D301 and determine the presence or absence of the identification information in each of the photographed images.
- the control unit 21 acquires information excluding the information of the specific identification information from the identification information of the tablet acquired from the captured image of the tablet as the identification information of the tablet. Thereby, when only the specific identification information is included in the photographed image of the tablet, the identification information of the tablet is not acquired from the photographed image of the tablet.
- the control unit 21 displays the photographed images of the front and back surfaces.
- the control unit 21 displays a photographed image that does not include the identification information. Is omitted and only the captured image including the identification information is displayed. At this time, the control unit 21 enlarges and displays the photographed image within an area where the photographed images on the front and back surfaces are displayed.
- the display of the captured image of the surface where only the specific identification information exists is omitted, and the captured image of the other surface is enlarged. Therefore, the pharmacist can easily confirm the identification information of the tablet.
- the inspection support system 1 images of various tablets registered in advance in the pharmaceutical master are used for collation and result display in the automatic inspection process.
- the tablet packaging device 4 has an information registration function for supporting a registration operation for registering identification information, shape information, and the like of the tablet in the pharmaceutical master. Note that the information on the pharmaceutical master registered in the tablet packaging device 4 is also transmitted to the server 2 and used in the server 2.
- the information registration function is realized by the information registration process being executed by the control unit 40.
- the identification information of the tablet is acquired from a captured image satisfying a preset condition among the plurality of captured images of the tablet, and the identification information of the tablet corresponds to the type of the tablet
- the data is registered with the pharmaceutical master or the like.
- the collation source data includes, as the normal image, an image of the tablet including at least the identification information area of the tablet among the captured images.
- the normal image and the captured image are included. Appropriateness of the tablet is determined by collation with the image.
- the identification information of the tablet registered as the normal image is not limited to an image of a part of the region including the identification information in the captured image, and may be an image of the entire region of the captured image.
- the said control part 40 when performing the said information registration process is an example of a registration process part.
- FIG. 30 is a flowchart showing an example of the procedure of the information registration process executed by the control unit 40.
- the control unit 40 can execute the information registration process as part of an initial setting process in the tablet packaging device 4.
- the said control part 40 is when the information of the tablet contained in the said prescription data used as the object of the said packaging process is not registered into the said pharmaceutical master, or when the inspection result by the said automatic inspection process is an error
- step S31 the control unit 40 executes a process of acquiring a plurality of captured images obtained by capturing the outer periphery of the tablet from different directions. Specifically, the control unit 40 pays out the tablets from the tablet cassette 41 in which the tablets to be registered are accommodated or from the hand handling unit 42. Then, the control unit 40 uses the camera 462 and the camera 463 of the photographing unit 46 to indicate a plurality of states in which the tablet is rotated by the rotation unit 44, as in the case of the packaging process. Shoot once.
- photography process which concerns here is an example of an imaging
- control unit 40 displays a selection screen for selecting whether the identification information of the tablet is formed on the tablet by stamping or printing before photographing the tablet. To display. Next, the control unit 40 specifies whether the identification information of the tablet is stamped or printed in accordance with a user's selection operation on the operation display unit 48.
- the said control part 40 when performing the specific process which concerns here is an example of a specific process part.
- the control unit 40 It is also possible to specify whether the identification information of the tablet is stamped or printed based on a medicine database.
- the control unit 40 includes the first photographing unit including the camera 462 and the lighting device 468 and the second photographing unit including the camera 463 and the lighting device 469 according to a method of forming the identification information. Select one of the above and shoot the tablet. Specifically, when it is specified that the identification information of the tablet is a stamp, photographing of the tablet is performed using the first photographing unit suitable for the marking, and the identification information of the tablet is printed. If specified, photographing of the tablet is executed using the second photographing unit suitable for the printing. As described above, the control unit 40 registers the first image as the collation source data, and the first photographing unit or the second photographing unit suitable for the tablet identification information forming method (engraving or printing). Select a unit and take a pill. Accordingly, a normal image registered in the pharmaceutical master as the tablet collation source data is acquired from a captured image in which the identification information of the tablet is clearly captured. Thereby, in the said image inspection process, the precision of the matching process performed using the said normal image increases.
- the control unit 40 displays the plurality of captured images corresponding to the tablet. It is conceivable to read out from the storage unit 49 as a photographed image of a tablet to be registered. That is, the control unit 40 is not limited to executing the information registration process before executing the packaging process, but registers the tablet information in the pharmaceutical master as a series of processes at the time of the packaging process. It is also possible to do. Note that in this case, the control unit 40 causes the camera 462 and 463 to photograph the tablet in the packaging process, and the camera 462 or the printing suitable for the marking or printing selected by the user operation as described above. It can be considered that the photographed image photographed at 463 is read out as a photographed image of a tablet to be registered.
- the control unit 40 determines whether or not the tablet is in the longitudinal rotation state. If the tablet is in the longitudinal rotation state, the control unit 40 applies vibration to the tablet to change the orientation of the tablet. Execute the process.
- the said control part 40 performs not only the said information registration process, but the same process is performed also when image
- the control unit 40 determines whether or not the aspect ratio of the tablet in a predetermined number or more of the captured images among the plurality of captured images is equal to or greater than a predetermined ratio. Specifically, when the predetermined number is five and the predetermined ratio is 1.2, the control unit 40 determines that the vertical / horizontal value of the tablet is 1.2 in the five or more captured images. When it is above, it is determined that the tablet is rotating in the longitudinal rotation state.
- the lateral direction of the tablet is the axial direction of the rotating roller 100.
- the longitudinal direction of the tablet is a direction perpendicular to the facing direction of the rotating roller 100 and the camera 462 or 463 and the axial direction of the rotating roller 100.
- the said control part 40 when performing the process which concerns here is an example of a vibration process part.
- the rotation unit 44 or the tablet rotation unit 441 is provided with a vibration unit that applies vibration to the rotation unit 44 or the tablet rotation unit 441 so as to vibrate the tablet. It is conceivable that the unit 40 drives the excitation unit so that the tablet falls.
- the said vibration part gives the vibration of the axial direction of the said rotation roller 100 to the said tablet rotation part 441, for example.
- control unit 40 may perform a swing process in which the rotation unit 44 is reciprocated by a predetermined width using the unit rotation unit 442 of the rotation unit 44.
- the tablet is tilted by the tablet rotating unit 441 to change the orientation of the tablet, and the front and back surfaces of the tablet are photographed by the camera 462 or 463.
- the tablet orientation may be changed using the vibration unit, and the control unit 40 is necessary for acquiring the captured images of the front and back surfaces of the tablet.
- the rotation unit 442 does not execute the rotation operation of the tablet rotation unit 441 to the next step.
- the said control part 40 acquires the image
- Step S32 Each time step S32 is executed, the control unit 40 sequentially selects the captured images to be processed in steps S33 to S35 from among the plurality of captured images corresponding to the tablets. In steps S33 to S35, various processes are performed on the captured image selected in step S32.
- step S33 the control unit 40 specifies a tablet region where the tablet is present from the photographed image.
- the control unit 40 can acquire an area excluding black in the photographed image as the tablet area.
- the control unit 40 acquires, as the tablet region, a region that is lighter than the reference color with the black color of the rotating roller 100 as a reference color (threshold value) in the captured image.
- the reference color is represented by component values of R (red), G (green), and B (blue).
- step S34 the control unit 40 acquires the area value of the tablet region specified from the captured image.
- the control unit 40 selects a circumscribed region of the tablet region having the largest area value among the tablet regions as a tablet region corresponding to the tablet. .
- step S35 the control unit 40 acquires an index value such as an average value or a deviation value of gray values in the tablet region.
- the gray value is a value when the color of each pixel included in a grayscale image is expressed by a plurality of gradations such as 256 gradations or 65536 gradations from white to black. For example, the gray value “0” is white, and the gray value “255” is black. The gray value “255” is white, and the gray value “0” is black.
- the control unit 40 converts the captured image into a grayscale image, and the index value of the gray value of the tablet region of the converted captured image To get.
- the index value is used to extract identification information such as printed characters or stamps in the tablet area.
- the ratio of the area occupied by the identification information on the front or back surface of the tablet is considered to be small. Therefore, for example, when the gray value “0” is white, the gray value “255” is black, and the color of the tablet is closer to black than the identification information, the gray value The average value is larger than the gradation value of the identification information. Therefore, when the average value of the gray values is equal to or greater than a predetermined threshold, the control unit 40 may extract an area where the gray value is smaller than the average value among the tablet areas as the identification information. Is possible.
- the control unit 40 may extract, as the identification information, an area in which the gray value is larger than the average value among the tablet areas. Is possible.
- step S36 the control unit 40 determines whether or not the processing in steps S33 to S35 has been completed for all the photographed images corresponding to the tablets.
- S36: Yes the process proceeds to step S37
- S36: No the process is Control goes to step S32.
- step S37 the control unit 40 specifies one or a plurality of photographed images including identification information of the tablet among the plurality of photographed images corresponding to the tablets as registration images.
- the registration image is specified for each of the front and back surfaces of the tablet.
- the control unit 40 as shown in FIG. 24A, is an odd-numbered image out of a plurality of photographed images identified as the photographed images f4 or f8 obtained by capturing the front and back surfaces of the tablet from the front. It can be considered that the photographed image is determined as the front image and the even-numbered photographed image is determined as the back image.
- Processing in steps S38 to S39 which will be described later, is executed for one or more registration images corresponding to the front surface of the tablet and one or more registration images corresponding to the back surface of the tablet.
- the control unit 40 acquires the edge amount included in the tablet region of each of the photographed images, and specifies the reference photographed image having the largest edge amount among the photographed images as the registration image.
- the edge amount is obtained by counting the number of edges where the difference between the gradation values of adjacent pixels is greater than or equal to a preset threshold value. Thereafter, the control unit 40 captures a ratio in which the ratio of the edge amount of the photographed image to the edge amount of the reference photographed image among the plurality of photographed images is equal to or greater than a preset threshold (for example, 80% or 90%).
- a preset threshold for example, 80% or 90%
- control unit 40 may register the reference photographed image as a normal image of the tablet with the medicine master. Further, the reference photographed image registered as the normal image is transmitted to the server 2 by the control unit 40 and registered in the medicine master stored in the storage unit 22 of the server 2. Thereby, the server 2 can display the reference captured image as the normal image in the inspection support process.
- control unit 40 registers a photographed image selected by a user operation among the plurality of photographed images corresponding to the tablets as the normal image in the medicine master. For example, in step S37, the control unit 40 displays a plurality of the captured images corresponding to the tablets on the operation display unit 48, and selects the captured image by a user operation using the operation display unit 48. Accept and register the captured image with the pharmaceutical master.
- step S ⁇ b> 37 the control unit 40 displays the captured image registered as the normal image on the operation display unit 48, and in the captured image by a user operation using the operation display unit 48.
- the designation of the upward position (the correct orientation of the identification information) is accepted.
- the said control part 40 rotates the said picked-up image so that the upward position designated by the said user operation may become an upper part, and registers into the said pharmaceutical master as the said normal image. That is, the correct position is indicated as the upward position in the normal image by the direction of the normal image that is the verification source data.
- control unit 40 is configured to register the image inspection process by registering the normal image rotated so that the upward position in the photographed image is at the top as the verification source data.
- the correctness of the identification information can be determined by comparing the positive image with the captured image in a state where the direction of the identification information included in the normal image is correct.
- the control unit 40 registers the captured image as the normal image without rotating it, and associates information such as coordinates indicating the position in the upward direction with the positive image and a part of the collation source data. It is also possible to register as. In this way, even when information indicating the upward position in the normal image is registered in the medicine master as a part of the verification source data, the control unit 40 adds the correct image to the normal image in the image inspection process.
- the correctness of the identification information can be determined by comparing the positive image and the captured image with the orientation of the identification information included being correct.
- an initial normal image of the tablet in which the identification information of the tablet is photographed in the correct orientation is stored in advance in the medicine database.
- the control unit 40 may determine an upward position (correct orientation of the identification information) in the normal image based on the initial normal image stored in the medicine database instead of the user operation. Conceivable. For example, the control unit 40 executes a matching process for collating the positive image in each rotation state with the initial normal image while gradually rotating the normal image. Then, the control unit 40 determines that the state in which the difference between the positive image and the initial positive image is the smallest is the correct state of the positive image according to the collation result between the positive image and the initial positive image. Then, the normal image in that state is registered in the medicine master.
- control unit 40 registers the captured image as the normal image without rotating the image according to the collation result between the normal image and the initial normal image, and the upward direction. It is also conceivable that information such as coordinates indicating a position is registered as a part of the collation source data in association with the normal image.
- the control unit 40 can register information registered in the medicine database as initial information of the medicine master, and for the tablets not registered in the information registration process, It is also conceivable that the initial positive image in the database is registered in the medicine master as the normal image.
- step S38 the control unit 40 identifies the shape of the tablet based on the registration image, and creates tablet model data corresponding to the shape.
- the tablet model data includes information used to specify the shape of the tablet, such as the height, width, and area value of the tablet.
- the tablet model data includes information such as the position and range of the identification information included in the tablet.
- control unit 40 individually registers information on the front and back surfaces of the tablet in the tablet model. Specifically, the control unit 40 creates a tablet model corresponding to the surface of the tablet based on the one or more registration images corresponding to the surface extracted in the step S37, and in the step S37. A tablet model corresponding to the back surface of the tablet is created based on the one or more registration images corresponding to the extracted back surface.
- the tablet model data is, for example, for checking the suitability of the shape or size of the tablet in the automatic inspection process, or for specifying the existence area of the identification information of the tablet in the photographed image in the automatic inspection process. Used for.
- the control unit 40 acquires information such as the position and range of the identification information in the tablet based on the plurality of registration images. It is also possible. For example, a value obtained by averaging values indicating the position and range of the identification information specified based on each registration image is created as the tablet model. Further, a value indicating the position and range of the identification information specified based on each of the registration images is set in advance corresponding to the ratio between the edge amount of the registration image and the edge amount of the reference photographed image. The tablet model may be created in consideration of weighting. Thereby, the said control part 40 can produce the said tablet model with high precision even when the omission etc. have arisen in the said identification information in a part of the said image for registration.
- step S39 the control unit 40 acquires the identification information of the tablet from the reference photographed image or the registration image, and the medicine identification information such as a medicine name or a medicine code indicating the type of the tablet. And register with the pharmaceutical master. Thereby, the identification information of the tablet acquired in the information registration process is registered in the medicine master as verification source data of the identification information of the tablet in the automatic inspection process.
- the control unit 40 acquires the identification information on the surface of the tablet based on one or a plurality of the registration images corresponding to the surface, and the identification information on the back surface of the tablet corresponds to the back surface. Obtained based on the plurality of registration images.
- step S40 the control unit 40 acquires the minimum value and the maximum value of the area value of the tablet from one or a plurality of the registration images.
- the minimum value and the maximum value of the area value of the tablet are used to acquire the tablet region from the photographed image in the automatic inspection process, or are used to calculate the circumferential length of the tablet.
- the minimum value and the maximum value of the area value of the tablet may be acquired not only from the registration image but also from a plurality of the captured images.
- step S41 the control unit 40 acquires a minimum value and a maximum value of each color component of the tablet from one or a plurality of registration images.
- the control unit 40 acquires a minimum value and a maximum value for each color component of RGB.
- the minimum value and the maximum value of each color component of the tablet may be acquired from a plurality of the captured images, not limited to the registration image.
- step S42 the control unit 40 associates the shape information of the tablet that is the processing target of the information registration process with the medicine identification information such as a medicine name or a medicine code indicating the type of the tablet. And register with the Pharmaceutical Master.
- the shape information of the tablet includes the height, width, and area value of the tablet specified in step S38, and the minimum value and maximum value of the area value of the tablet acquired in step S39. The minimum value and the maximum value of each color component of the tablet acquired in step S40 are included.
- the control unit 40 in the automatic inspection process, captures the image of the tablet and the shape information of the tablet. It is conceivable to determine the suitability of the tablet based on the above. More specifically, in the automatic inspection process, the control unit 40 determines the shape of the tablet included in the captured image and the shape of the tablet included in the tablet model corresponding to the tablet included in the prescription data. Match. And when the collation result of the shape of the tablet based on the tablet model is coincident, the control unit 40 is subsequently registered in the information registration process with the identification information of the tablet included in the photographed image. The identification information of the tablet is collated.
- the control unit 40 does not determine the suitability of the tablet based on the identification information included in the captured image,
- the inspection result of the automatic inspection process is determined to be an error. That is, in the automatic inspection process, only when the collation result of the tablet shape is appropriate, the suitability determination of the tablet based on the tablet identification information is executed. Therefore, in the tablet packaging device 4, it is possible to omit as much as possible the process of acquiring the identification information from the photographed image and the process of heavy load such as the collation of the tablet based on the identification information.
- the said control part 21 has an apparatus selection function which selects the said tablet packaging apparatus 4 which performs the said packaging process based on the said prescription data based on the preset conditions.
- the device selection function will be described with reference to FIGS. 31 and 32.
- the plurality of tablet packaging devices 4 that can be connected to the inspection support system 1 include a tablet packaging device 4 (hereinafter referred to as “No. 1 tablet packaging device 401), and tablet packaging device 4 (hereinafter referred to as second tablet packaging device 402) that can perform the packaging process but cannot perform the image inspection process. May be.
- the second tablet packaging device 402 does not have the imaging unit 46.
- the first tablet packaging device 401 can execute the image inspection process and can switch between valid and invalid of the image inspection process, the execution of the image inspection process is set to invalid.
- the tablet packaging device 4 is recognized as the second tablet packaging device 402 by the server 2.
- tablette packaging device 4 when the first tablet packaging device 401 and the second tablet packaging device 402 are collectively referred to, they are simply referred to as “tablet packaging device 4”.
- selection setting information D11 used for selection of the tablet packaging device 4 is stored.
- FIG. 31 shows an example of the selection setting information D11.
- the selection setting information D11 includes items of automatic inspection function setting and distribution function setting.
- the selection setting information D11 is input from the first tablet packaging device 401 to the control unit 21 of the server 2 or read from the first tablet packaging device 401 by the control unit 21 of the server 2. It is.
- the distribution function setting items include a first priority condition and a second priority condition set in advance.
- the first priority condition is a condition for determining whether or not to execute the automatic inspection process
- the second priority condition is that the tablet packaging device 4 is controlled regardless of the necessity of the automatic inspection process. It is a condition for selecting.
- the control unit 21 can set each of the first priority conditions to either valid or invalid according to a user operation. Further, the control unit 21 can set priorities in the plurality of second priority conditions for each of the second priority conditions in accordance with a user operation.
- the first priority condition includes items of “DTA hit priority”, “lock number condition priority”, and “high risk drug priority”.
- DTA hit priority is a condition under which the first tablet packaging device 401 is selected when the manual handling unit 42 is used in the packaging process.
- Tablet number condition priority is a condition for selecting the first tablet packaging device 401 when the number of tablets per package is equal to or greater than a preset threshold value in the packaging process.
- High-risk medicine priority is a condition for selecting the first tablet packaging device 401 when the medicine to be packaged in the packaging process is a preset high-risk medicine. In the example shown in FIG. 31, “DTA hit priority” and “high-risk medicine priority” are set valid, and “lock number condition priority” is set invalid.
- the second priority condition includes items of “mounting hit rate priority”, “empty buffer rate priority”, and “arbitrarily specified priority”.
- “Mounting hit rate priority” is a condition for selecting the tablet packaging device 4 having the largest number of types of medicines filled in the tablet cassette 41 among the medicines to be packaged in the packaging process.
- the “empty buffer rate priority” is a condition for selecting the tablet packaging device 4 with the smallest amount of prescription data waiting as a target of the packaging process.
- “Optional designation priority” is a condition for selecting the tablet packaging device 4 arbitrarily designated by the user. In the example shown in FIG.
- the priority of “arbitrarily specified priority” is set to “1”
- the priority of “mounting hit rate priority” is set to “2”
- the priority of “empty buffer priority” is set to “3”.
- the second priority condition is not limited to the setting of the priority, but may be alternatively selected.
- the said control part 21 makes the said tablet packaging apparatus 4 which performs the said packaging process corresponding to each said prescription data, at least one of the content of the said prescription data, the content of the said packaging process, and the said selection setting information D11.
- a device selection process for selecting based on the above is executed.
- the said control part 21 when performing the said apparatus selection process is an example of a selection process part.
- FIG. 32 is a flowchart showing an example of the device selection process.
- the control unit 21 executes the device selection process for prescription data that requires the packaging process using the tablet packaging device 4 in the prescription data.
- step S51 the control unit 21 determines whether at least one of the content of the prescription data and the content of the packaging process satisfies any of the conditions of the first priority condition. To do. When the control unit 21 determines that any one of the first priority conditions is satisfied, the control unit 21 proceeds to step S511. When the control unit 21 determines that all the first priority conditions are not satisfied, the process proceeds to step S52. To migrate.
- “DTA hit priority” and “high risk drug priority” are set to be effective. Therefore, when the handing unit 42 is used in the packaging process for the prescription data, or when the medicine to be packaged is a high-risk drug, it is determined that the first priority condition is satisfied.
- step S511 the control unit 21 performs the image inspection processing as the first tablet packaging device 401 as the tablet packaging device 4 that executes the packaging processing based on the prescription data. And the prescription data is transmitted to the selected tablet packaging device 4. That is, when at least one of the first priority conditions is satisfied, the first tablet packaging device 401 is selected. Therefore, when it is desirable to execute the image inspection process, the packaging process can be performed using the first tablet packaging device 401.
- Step S52> When it is determined in step S51 that the first priority condition is not satisfied, in the subsequent steps S52 to 53, the tablet packaging device 4 is selected according to the second priority condition. Specifically, in step S52, the control unit 21 adds 1 to the value of the counter N used to specify the priority value of the second priority condition. The initial value of the counter N is 0, and is reset to 0 at the start or end of the device selection process.
- step S53 the control unit 21 determines which one of the tablet packaging devices 4 is based on the second priority condition of the counter N whose preset priority value is the second priority condition. It is determined whether or not can be selected. For example, when there is one tablet packaging device 4 that matches the second priority condition, it is determined that the tablet packaging device 4 can be selected.
- the said control part 21 judges that the said tablet packaging apparatus 4 can be selected (S53: Yes)
- step S53: No the process is returned to step S52.
- the tablet packaging device 4 cannot be selected even when the value of the counter N is the same as the number of the second priority conditions (here, “3”), the tablet packaging It is conceivable that the specific tablet packaging device 4 set in advance among the devices 4 is selected. Further, the value of the counter N is a case where the plurality of tablet packaging devices 4 are narrowed down as selection targets from the plurality of tablet packaging devices 4 based on the second priority condition, and the second priority Even if the number of conditions is the same, if it is determined that the tablet packaging device 4 cannot be selected, the tablet packaging device with the largest or smallest tablet packaging device number among the tablet packaging devices 4 It is conceivable that the device 4 is selected.
- step S54 the control unit 21 selects the tablet packaging device 4 that executes the packaging process based on the second priority condition in which the priority is a value of a counter N, The prescription data is transmitted to the selected tablet packaging device 4. Thereby, the said packaging process is performed by the said tablet packaging apparatus 4 selected according to the said priority of a said 2nd priority condition.
- step S54 it is conceivable that the first tablet packaging device 401 is not selected. That is, in step S54, the control unit 21 may select one of the second tablet packaging devices 402 based on the second priority condition in which the priority is the value of the counter N. Thereby, when all the first priority conditions are not satisfied, the second tablet packaging device 402 in which the image inspection process is not executed is used, and the dispensing process based on the prescription data is quickly performed. Done.
- the type of each tablet and the prescription data are included based on a photographed image photographed by the camera 462 or the camera 463 before the tablet is packaged in the medicine package 451. It is checked whether the result of the packaging process is appropriate or not by determining whether or not the collation result with the type of tablet to be matched is the same. That is, in the image inspection process, whether or not the medicine package 451 obtained as a result of the packaging process is appropriate is determined by determining whether or not the tablet before being packaged in the medicine package 451 is appropriate. To be judged. On the other hand, the image inspection process is not limited to this.
- the image inspection process includes a photographing unit that photographs the medicine package 451 after the tablets are packaged in the medicine package 451, and is based on a photographed image photographed by the photographing unit. Then, it may be determined whether or not the collation result between the type of each tablet and the type of the tablet included in the prescription data is the same. Further, in the image inspection process, it may be determined whether or not the collation result between the number of tablets of each tablet and the number of tablets of the tablet included in the prescription data is coincident based on a photographed image of the tablet. . That is, the image inspection process may be a process for determining whether or not part or all of each step in the packaging process has been appropriately performed based on the captured image of the tablet or the medicine package 451.
- the tablet packaging device 4 determines whether or not the image inspection process has been executed as the automatic inspection process in the first or last medicine package 451 of the series of medicine packages 451 delivered in the packaging process. It is conceivable to have an inspection recording function for recording. For example, as shown in FIG. 2, the first tablet packaging device 401 records the presence / absence of execution of the image inspection process in the medicine package 451 after the packaging process, separately from the printing unit 453. It is conceivable to have a stamp unit 454 that can. The stamp unit 454 can record inspected information such as characters or images indicating that the image inspection process has been executed on the medicine package 451 by using a stamp. For example, as shown in FIG. 33A and FIG.
- the inspection existence information includes a character string “automatic check target” indicating that it is a check target by the image inspection process, or a check by the image inspection process.
- the stamp unit 454 records “NG” in the medicine package 45 as the inspected information as shown in FIG. 33C and the stamp for recording “automatic check target” or “CH” as described above. It is also conceivable to provide a stamp to be used.
- the result of the image inspection process is OK, as described above, “automatic check target” or “CH” is recorded in the medicine package 451, and when the result of the image inspection process is an error.
- “NG” is recorded in the medicine package 451 as the inspected information.
- the fact that the image inspection process has not been executed is indicated by the fact that the inspection existence information such as “automatic check target”, “CH”, or “NG” is not recorded in the medicine package 451.
- control unit 40 uses the printing unit 453 to set “medical package” to “automatic check target” or “CH” as information indicating whether or not the image inspection process is performed. It is also possible to print to 451. Furthermore, a configuration in which the image inspection process for tablets already packaged in the medicine package 451 is completed at a timing when the inspection presence information can be recorded in the medicine package 451 by the printing unit 453 is also conceivable. In this case, the control unit 40 uses the printing unit 453 to print “automatic check target” or “CH” on the medicine package 451 as the inspection existence information when the result of the image inspection processing is OK. If the result of the image inspection process is an error, “NG” may be printed as the inspection existence information.
- the tablet packaging device 4 executes the automatic inspection function for determining the suitability of the tablets contained in the medicine package 451 packaged by the packaging process.
- the automatic inspection process For determining the suitability of the tablets contained in the medicine package 451 packaged by the packaging process.
- the packaging process based on the prescription data is executed by the control unit 40, the tablets are photographed by the cameras 461 to 467 of the photographing unit 46. Further, when the packaging process is executed, the control unit 40 counts the number of the tablets detected by the passage detection sensor 475 of the passage detection unit 47 for each medicine package 451. In addition, the said control part 40 when counting the number of the said tablets is an example of a tablet number detection part. And when the said packaging process is complete
- Step S61 the control unit 40 determines whether or not the image inspection process needs to be executed, and determines that the image inspection process needs to be executed (S61: Yes), the process proceeds to step S62.
- the control unit 40 determines whether or not the image inspection process is performed according to a current inspection mode (any one of the first inspection mode to the third inspection mode) switched by the inspection mode switching function.
- the said step S61 is abbreviate
- Step S62> the control unit 40 identifies the tablet identification information acquired from the photographed image for each tablet stored in the medicine package 451 and the tablet included in the prescription data. An image inspection process for collating the identification information is executed.
- control unit 40 determines whether or not the shape of the tablet matches the shape of the tablet registered in the pharmaceutical master in association with the tablet included in the prescription data. And the said control part 40 judges that the collation result is inconsistent, when the shape of the said tablet does not correspond. On the other hand, when the shape of the tablet matches, the control unit 40 acquires the identification information of the tablet from the captured image, and associates the identification information with the tablet included in the prescription data to the medicine master. An image inspection process for collating the registered identification information of the tablet is executed. The said control part 40 when performing the process which concerns here is an example of an image inspection process part.
- the control unit 40 extracts an image of the tablet including the identification information of the tablet from the captured image, and identifies the tablet image in the captured image and the tablet in the normal image. Matching process with information image is executed. Moreover, the character of identification information may be recognized from the said picked-up image by a well-known character recognition process (OCR), and collation may be performed using the character of the identification information.
- OCR character recognition process
- the control unit 40 specifies the captured images f4 and f8 corresponding to the front and back surfaces of the tablet as shown in FIG. The identification information is acquired from the captured images f4 and f8.
- the control unit 40 acquires the identification information from the left and right capsule pieces of the capsule tablet.
- step S63 if the collation result in step S62 is the same (S63: Yes), the control unit 40 moves the process to step S64, and if the collation result is inconsistent ( S63: No), the process proceeds to step S631.
- step S64 the control unit 40 records in the storage unit 49 that the inspection result of the tablet in the prescription data is appropriate. That is, the control unit 40 determines that the inspection result of the image inspection process is appropriate when the shape and identification information of the tablet match the shape and identification information of the tablet registered in the pharmaceutical master. To do.
- step S631 the control unit 40 determines whether or not another tablet type not included in the prescription data is detected as the tablet type. For example, when the type of the tablet is not detected, a defect or the like may occur in the identification information attached to the tablet. In addition, since it is not possible to perform collation by the image inspection process for the tablets without the identification information, the type of the other tablets is not detected even when the identification information is not attached to the tablets. Become. On the other hand, if another tablet type is detected, the tablet is clearly incorrect. Therefore, in step S631, when the other tablet type is detected (S631: Yes), the process proceeds to step S632, and when the other tablet type is not detected (S631). : No), the process proceeds to step S633.
- step S632 For tablets without the identification information, it is determined whether or not the appearances such as the shape, size, and color of the tablets registered in the pharmaceutical master coincide with each other. If yes, the process moves to step S632, and if the appearances do not match, the process can move to step S633.
- step S631 it is determined in step S631 whether or not the degree of matching (matching rate) obtained as a result of the matching process is less than a preset threshold value. If the value is less than the threshold, the process may proceed to step S632.
- step S632 the control unit 40 records in the storage unit 49 that the inspection result of the image inspection process for the tablet is an error. That is, the control unit 40 determines that the inspection result of the image inspection process is an error when the identification information of the tablet is clearly different from the identification information of the tablet registered in the pharmaceutical master.
- step S633 the control unit 40 indicates that the inspection result of the image inspection process for the tablet may not be appropriate and that a visual check is necessary as the inspection result of the tablet. Record in the storage unit 49. That is, if the identification information of the tablet is not clearly different from the identification information of the tablet registered in the pharmaceutical master, the control unit 40 needs to check the inspection result of the image inspection process. Judge. In the image inspection process, it is considered that the inspection result of the image inspection process is determined to be necessary check even when the degree of coincidence in the collation result of the identification information of the tablet is a predetermined value or less. It is done.
- Step S65 when the control unit 40 determines that the image inspection process has been completed for all the tablets of the medicine package 451 (S65: Yes), the process proceeds to Step S66 and ends. If not (S65: No), the process returns to step S62.
- step S66 the control unit 40 determines the number of tablets detected by the passage detection sensor 475 in the packaging process and the number of tablets to be packaged in the medicine package 451 in the prescription data.
- a count inspection process is performed to determine whether or not the two match.
- the passage detection sensor 475 (refer to FIG. 2) is downstream of the photographing position of the tablet by the cameras 462 and 463 and before the tablet is accommodated in the medicine package 451. Is arranged at a position where it can be detected. Thereby, in the count inspection process, it is determined whether or not the number of tablets counted at least downstream of the tablet photographing position and before the medicine package 451 is sealed is appropriate.
- the said control part 40 when performing the said count inspection process which concerns here is an example of a count inspection process part.
- step S67 if the collation result in step S66 is the same (S67: Yes), the control unit 40 moves the process to step S68, and if the collation result is inconsistent ( S67: No), the process proceeds to step S671.
- step S68 the control unit 40 associates the fact that the inspection result of the count inspection process for the tablet is appropriate with the tablet that was the object of collation in step S66. 49.
- step S671 the control unit 40 indicates that the inspection result of the counting inspection process for the tablet is an error in association with the tablet that was the target of verification in the step S66. 49.
- both the image inspection process and the count inspection process are performed, and when both inspection results are appropriate.
- the inspection result of the automatic inspection process is determined to be appropriate and one of the inspection results is an error, the inspection result of the automatic inspection process is an error.
- the said control part 40 will perform the result of the said image inspection process with the said picked-up image together with the said server 2 when the said automatic inspection process is performed about the tablet of all the said medicine packages 451 packaged by the said packaging process.
- the number of the tablets used in the counting inspection process is also transmitted to the server 2 by the control unit 40. Accordingly, the server 2 can display the result of the image inspection process, the result of the count inspection process, the captured image, and the like as necessary in the inspection support process executed by the control unit 21. Is possible.
- FIG. 35A, FIG. 36, and FIG. 37A are diagrams showing an inspection detail screen D303 that is another example of the inspection detail screen D302 (see FIGS. 19 and 20).
- FIG. 35A is a display example when the inspection result by the automatic inspection process is appropriate
- FIG. 36 is a display example when the inspection result by the automatic inspection process is an error
- FIG. It is an example of a display when the inspection result by inspection processing is a check required.
- the control unit 21 receives approval of the inspection result of the automatic inspection processing and stores it in association with the prescription data. Further, as shown in FIG. 35B, the control unit 21 displays “approval OK” as the inspection result of the automatic inspection process in the inspection result area A313 on the inspection details screen D303. Furthermore, as shown in FIG. 35B, the control unit 21 displays “ ⁇ ” indicating that the inspection result of the automatic inspection process is appropriate in the result display field A321 on the inspection detail screen D303. As shown in FIG. 35B, when the inspection result by the automatic inspection process is “approved OK”, it is displayed that the approval key K311 cannot be operated due to grayout or the like.
- the inspection detail screen D303 indicates that the inspection result area A313 has an error as the inspection result of the automatic inspection process. “NG” is displayed.
- the inspection result area A313 is a check required as the inspection result of the automatic inspection process. “CHECK” indicating the effect is displayed.
- the control unit 21 does not acquire the tablet identification information in the image inspection process, that is, there is character printing or inscription on the tablet like the tablet “Tic Tac” shown in FIG. 37B.
- the approval key K311 can be operated as shown in FIGS. 37A and 37B, but the inspection result of the automatic inspection process is an error. In some cases, as shown in FIG. 36, the approval key K311 cannot be operated due to grayout or the like.
- operation keys K312 to K314 are displayed on the inspection details screen D303.
- the operation key K312 is an operation key for re-executing the packaging process for one or more medicine packages 451.
- the control unit 21 displays a reissue operation screen D304 for setting a re-execution method of the packaging process.
- FIG. 38 shows an example of the reissue operation screen D304.
- the medicine package 451 to be reexecuted can be arbitrarily designated. For example, continuous designation or individual designation is possible. As will be described later, when the medicine package 451 to be re-executed by the packaging process has already been designated on another screen, the medicine package number of the designated medicine package 451, etc. Identification information is displayed in the input field of the designated package.
- the reissue operation screen D304 displays a reissue key K315 for starting execution of reissue, and the control unit 21 is designated according to the operation of the reissue key K315.
- a control instruction for re-executing the packaging process of the medicine package 451 is transmitted to the tablet packaging device 4. Thereby, in the tablet packaging device 4, the packaging process for the medicine package 451 designated on the reissue operation screen D304 is executed again according to the control instruction.
- the operation key K313 is an operation key for displaying the manual confirmation screen D5.
- the operation key K313 can be operated when the handing unit 42 is used in the packaging process, and when the handing unit 42 is not used in the packaging process, the operation key K313 is grayed out. Operation becomes impossible. Then, when the operation key K313 is operated, the control unit 21 displays the manual confirmation screen D5 (see FIG. 14) on which the manual image is displayed.
- the operation key K314 is an operation unit for changing the display size of the list display of inspection results on the inspection detail screen D303. And when the said display size is changed by operation of the said operation key K314, the said control part 21 will change the number of the said medicine packs 451 displayed simultaneously on the said inspection detail screen D303.
- the operation key K314 four types of display sizes of “large”, “medium”, “small”, and “minimal” can be arbitrarily selected. As a result, when referring to the result of the automatic inspection process, the user enlarges and confirms each of the captured images, or displays according to the purpose such as grasping the entire result of the automatic inspection process. Aspects are selectable.
- the control unit 21 displays the enlarged screen D34 corresponding to the tablet.
- Display. 39A and 39B show an enlarged screen D341 as another example of the enlarged screen D34.
- the enlarged image P342 corresponding to the normal image and the enlarged image P343 of the photographed image corresponding to the selected tablet are displayed side by side. Yes.
- the control unit 21 may display the enlarged screen D34 only for the photographed image of the tablet whose inspection result of the automatic inspection process is an error or needs to be checked among the photographed images.
- the enlarged screen D34 may be displayed for the photographed image of the tablet.
- control unit 21 sequentially switches the plurality of photographed images corresponding to the tablets at predetermined intervals as shown in FIGS. 39A and 39B for the enlarged image P343 displayed on the enlarged screen D341. It is possible to display. More specifically, the control unit 21 starts switching the enlarged image P343 when the enlarged screen D341 is displayed. Accordingly, the user can determine the suitability of the tablet by referring to the plurality of captured images obtained by capturing the outer periphery of the tablet from different angles.
- operation keys K361 to K364 are displayed.
- the control unit 21 temporarily stops automatic switching of the enlarged image P343 when the operation key K361 is operated, and resumes automatic switching of the enlarged image P343 when the operation key K361 is operated again. Note that the control unit 21 may start automatic switching of the enlarged image P343 when the operation key K361 is operated after the enlarged screen D341 is displayed.
- the control unit 21 determines to be appropriate by visual confirmation. Is recorded as the inspection result of the tablet. Further, when the operation key K363 is operated, the control unit 21 selects and records the medicine package 451 containing the tablet being displayed as the reissue target.
- the medicine package 451 containing the tablet being displayed as the reissue target As shown in FIG. 36, on the inspection details screen D303, a character or a design indicating that it is the reissuance target It is conceivable that additional information A324 such as the above is displayed in association with each other.
- the control unit 21 records that a measure such as manually replacing a tablet contained in the medicine package 451 including the displayed tablet is executed. . Also in this case, as shown in FIG. 37A, for the tablet selected as a target on which the manual treatment is performed, a character or a design indicating that it is a target for the manual treatment on the inspection details screen D303. It is conceivable that additional information A325 such as the above is displayed in association with each other.
- the control unit 21 makes the identification information similar to the tablet as shown in FIG. It is conceivable to display a similar medicine list P361 indicating information on one or more types of tablets on the enlarged screen D341.
- the said control part 21 makes the said similar chemical
- the operation key K362 is an operation key that simultaneously accepts an operation of sequentially switching tablets to be displayed on the enlarged screen D341 in the front-rear direction and an operation of inputting appropriateness by visual confirmation. It may be included.
- the control unit 21 determines that the inspection result of the automatic inspection process is OK and an error. Alternatively, both the tablets that need to be checked are displayed on the inspection detail screen D303.
- the control unit 21 determines that the inspection result of the automatic inspection process is an error or a check required. It is conceivable that only certain tablets are displayed on the inspection detail screen D303. That is, the display of the tablet with the proper inspection result of the automatic inspection process is omitted.
- the control unit 21 may display an inspection details screen D305 in which inspection results of the image inspection processing and the count inspection processing are displayed together.
- 42A and 42B are diagrams showing an example of the inspection details screen D305.
- the counting inspection process is executed for all the medicine packages 451 by the automatic inspection processing, and the first medicine package 451 for each time of taking is taken.
- the image inspection process is executed for only the image, and the inspection result is displayed in the first array (see FIG. 27).
- a part or all of the image inspection processing is omitted, and a “number mode” indicating that the counting inspection processing is being executed is displayed in the display area A327.
- the control unit 21 displays the photographed image used in the image inspection process for only the first medicine package 451 for each dose period.
- the tablets in the medicine package 451 that have not been subjected to the image inspection process are photographed by the cameras 462 and 463, but the photographed images of the tablets are not displayed.
- the photographed image used in the image inspection process is an image trimmed to extract the tablet region from the photographed images actually photographed by the cameras 462 and 463.
- the control unit 21 displays the photographed image used in the image inspection process for the first medicine package 451 for each dose period.
- the control unit 21 displays, as it is, a captured image that is actually captured by the cameras 462 and 463 and is not trimmed when the image inspection process is performed for a tablet that has not been subjected to the image inspection process. Thereby, the user can easily grasp whether or not the image inspection process is executed.
- the said control part 21 is performed about all the medicine packages 451 about the said count inspection process, in the said inspection detail screen D305, the number of tablets used by the said count inspection process of each said medicine package 451 is displayed. It is displayed in the lock number display area A326. Thereby, the user can easily grasp that there is no problem in the number of tablets detected by the counting inspection process.
- control unit 21 can display the inspection results by the image inspection process and the lock number inspection process on the inspection detail screen D305. Specifically, if an error has occurred in the image inspection process, for the tablet in which the error has occurred, the background of the captured image is red indicating that the result of the automatic inspection process is an error, etc. Displayed in the sixth specific color. In addition, when an error has occurred in the counting inspection process, for the medicine package 451 in which the error has occurred, for example, the sixth such as red indicating that the background of the display area of the number of tablets is an error. Displayed in a specific color.
- the tablet packaging device 4 includes a camera for photographing the medicine package 451 after the tablet packaging in the packaging unit 45
- the control unit 21 is photographed by the camera. It is also conceivable to display the captured image after packaging on the detailed screen D305. For example, the post-packaging photographed image is displayed side by side below the lock number display area A326.
- the tablet packaging device 4 determines whether or not the tablet to be accommodated in the medicine package 451 when the medicine package 451 is closed is present on the downstream side of the medicine package 451 to be closed.
- a detection unit 476 is provided for detecting.
- the control unit 40 detects the back shift of the tablet to be stored in the medicine package 451 based on the detection result by the detection unit 476.
- the back shift detection function will be described with reference to FIGS. 43 to 52.
- the packaging unit 45 is provided below the rotating unit 44, and is capable of packaging tablets M dispensed from the rotating unit 44 one by one.
- the packaging unit 45 includes a packaging paper supply unit 450A and a packaging mechanism 450B.
- the wrapping paper supply unit 450A is a mechanism that unwinds the wrapping paper S wound around the roll shaft 450C and sends it to the packaging mechanism 450B side.
- the wrapping paper S is a sheet-like long heat-fusible sheet, and is wound around a roll shaft 450C in a state of being folded in the short direction.
- the packaging mechanism 450B includes a sheet support portion 450D, a guide member 450E, and a seal device 450F.
- the wrapping mechanism 450B wraps the medicine M supplied from the rotating unit 44 side by crimping the wrapping paper S sent from the wrapping paper supply unit 450A into a bag shape. Is possible.
- the guide member 450E functions as a guide for guiding the wrapping paper S sent from the wrapping paper supply unit 42.
- the sealing device 50 crimps a portion on one end side (downstream side) in the longitudinal direction of the wrapping paper S that is supplied while being guided by the guide member 44b to form a half bag or a half bag.
- the open portion of the paper S can be crimped and closed to form a bag.
- the medicine package 451 containing the medicine M can be formed as shown in FIG.
- the seal device 450F is a vertical seal (first vertical seal AS1 or second vertical seal AS3) that closes the downstream side in the traveling direction of the wrapping paper S in the medicine package 451 to be created. ) And a horizontal seal WS2 are formed.
- a half-sack-shaped wrapping paper S (medicine package 451) having an open portion at a portion upstream of the wrapping paper S in the traveling direction is formed.
- the open portion is closed by the sealing device 450F. That is, when a part of the lateral seal WS2 is unsealed, the unsealed portion is closed by the sealing device 450F and is closed in the short direction of the wrapping paper S on the upstream side in the traveling direction of the wrapping paper S.
- a vertical seal (second vertical seal AS3) is formed and sealed.
- the sealing device 450F has a main part constituted by a pair of roller frames 450a and 450b.
- the sealing device 450F is provided with a protective cover 450c on the roller frame 50a side. When the protective cover 450c is removed, the roller frames 50a and 50b are in contact with each other as shown in FIG. In FIG.
- the roller frames 450a and 450b are substantially “U” -shaped (gate-shaped) when viewed from the front, and are constituted by metal frames.
- the roller frames 450a and 450b are provided with support shafts 450d extending in the vertical direction, and a vertical seal member 450e and a horizontal seal member 450f are attached thereto.
- the vertical seal member 450e and the horizontal seal member 450f are attached so as to be rotatable with respect to the support shaft 450d.
- the vertical seal member 450e and the horizontal seal member 450f are connected to separate power sources (not shown) via separate power transmission mechanisms (not shown), and can be rotated independently of each other.
- the bag length of the medicine package 451 can be changed by changing the rotation speed of the vertical seal member 450e and the rotation speed of the horizontal seal member 450f.
- the vertical seal member 450e is made of metal and has a substantially linear shape when viewed from the front as shown in FIG. As shown in FIG. 46, the vertical seal member 450e has a disc-shaped lower end portion 450i and a plate-shaped heating unit 450k.
- the heating part 450k is located between an upper end part 450g and a lower end part 450i forming a lateral seal member 450f described later, and is substantially perpendicular to both.
- a heater 450h and a cut line forming part 450j are linearly arranged from the upper end part 450g side to the lower end part 450i side.
- the heaters 450h and 450h can heat-seal the wrapping paper S. Therefore, a seal (vertical seal) extending in the short direction of the wrapper paper S by rotating the longitudinal seal members 450e, 450e arranged in parallel and passing the folded wrapper paper S between them. Can be formed.
- the cut line forming part 450j can form a perforation on the wrapping paper S.
- the cut line forming part 450j on the roller frame 450b side is constituted by a cutter for forming a perforation, and the cut line forming part 450j on the roller frame 450a side is provided corresponding to the cutter. It is constituted by.
- the horizontal seal member 450f includes the above-described upper end portion 450g and heater 450m.
- the upper end part 450g is a disk-shaped member provided on the upper side of the heating part 450k of the vertical seal member 450e.
- a heater 450m is provided over the entire periphery. Therefore, by rotating the horizontal seal members 450f and 450f arranged in parallel and passing the folded folding paper S between the upper end portions 450g and 450g, a seal extending in the longitudinal direction of the packaging paper S ( Can be formed.
- the vertical sealing members 450e and 450e and the horizontal sealing members 450f and 450f are disposed in a substantially “B” -shaped (rectangular) region surrounded by the roller frames 450a and 450b. They are arranged substantially in parallel with a predetermined clearance.
- the sealing device 450F forms the medicine package 451 by rotating the vertical seal members 450e and 450e and the horizontal seal members 450f and 450f and passing the wrapping paper S through these clearances to form the horizontal seal and the vertical seal. Is possible.
- the rotation unit 44 is provided with a medicine introduction unit 80 for supplying tablets M individually dispensed from the rotation unit 44 to the packaging unit 45.
- the medicine introduction unit 80 may be anything as long as it can supply the tablet M into the wrapping paper S, but is configured by a hopper in this embodiment.
- the medicine introduction part 80 is inserted into the open portion of the unsealed medicine package 451 formed of the wrapping paper S by the sealing device 450F, Tablet M can be introduced into Specifically, the drug introduction section 80 has a proximal end portion directed toward the rotation unit 44 and a distal end portion inserted into an unsealed medicine package 451 that is being formed by the seal device 450F. Has been placed. That is, the medicine introduction unit 80 is inserted into the inner side of the folding paper S folded in half on the upstream side in the transport direction of the packaging paper S with respect to the sealing device 450F.
- the medicine package 451 accommodated in the medicine package 451 may be affected by heat from the heaters 450h and 450m. Therefore, as shown in FIG. 49, in the tablet packaging device 4, it is possible to blow air to the packaging paper S at a position where the tablets are supplied from the drug introduction unit 80 to the packaging paper S.
- a ventilation fan 51 is provided. Thereby, the said wrapping paper S is cooled by the ventilation from the said ventilation fan 51, and the influence of the heat from the said heaters 450h and 450m is suppressed.
- the blower fan 51 is disposed above the passage path of the wrapping paper S, and the inside of the wrapping paper S from the opening portion of the wrapping paper S. It is also possible to blow.
- the blower fan 51 may be arranged at a position where air can be blown from other directions such as below the passage path of the wrapping paper S.
- the packaging unit 45 is provided with a detection unit 476 that detects the presence of the tablet M in the introduction route of the tablet M by the medicine introduction unit 80.
- the detection unit 476 includes a camera 476a and an illuminating device 476b that can photograph the introduction route of the tablet M by the medicine introduction unit 80.
- the camera 476a is arranged so as to be able to photograph (detect) the inside of the packaging paper S on the upstream side of the sealing device 450F in the transport direction of the packaging paper S.
- the camera 476a is arranged from the proximal end side to the distal end side of the medicine introduction unit 80.
- the drug introduction part 80 is located on the upstream side with respect to the seal device 450F. Therefore, the camera 476a is arranged so as to be able to photograph (detect) an area on the upstream side in the transport direction of the packaging paper S with respect to the sealing device 450F.
- the lighting device 476b includes a light source such as a light emitting diode or a light bulb. The illumination device 476b is arranged so as to be able to irradiate the internal region of the drug introduction part 80 from the proximal end side to the distal end side of the drug introduction part 80, similarly to the camera 476a.
- the control unit 40 stores an image captured by the camera 476a in the storage unit 22 and also transmits it to the server 2.
- the said control part 40 will overflow the said tablet M from the medicine package 451 which should originally package the tablet M based on the detection data input from the said detection part 476. Therefore, it is possible to determine the occurrence of defective packaging.
- the control unit 40 forms the medicine package 451 in accordance with the control flow shown in FIG.
- specific operation and control will be described with reference to FIG.
- step S71 a vertical seal for closing the upstream end of the medicine package 451 at the leading position in the traveling direction of the wrapping paper S (hereinafter referred to as “first vertical seal AS1”). Is also formed by vertical seal members 450e and 450e (see FIG. 47). Thereafter, the control flow proceeds to step S72.
- a horizontal seal WS2 (see FIG. 47) is formed to close the end opposite to the fold of the wrapping paper S supplied in a folded state.
- the horizontal seal is formed by rotating the horizontal seal members 450f and 450f and passing the wrapping paper S between them.
- step S73 it is confirmed whether or not the horizontal seal WS2 is formed until the wrapping paper S reaches the position (sealing position) where the medicine package 451 is to be sealed.
- step S73: YES the control flow proceeds to step S74, and it is determined that the sealing position has not been reached (step S73: NO). If so, the control flow is returned to step S72.
- step S74 a vertical seal (hereinafter also referred to as “second vertical seal AS3”) for closing the downstream end of the packing paper S in the traveling direction in the medicine package 451 will be described in detail later. It is formed in accordance with the subroutine of FIG.
- the second vertical seal AS3 also functions as the first vertical seal AS1 of the medicine package 451 to be formed next. Therefore, the second vertical seal AS3 functions as a seal that forms a boundary of the medicine package 451 formed continuously in the longitudinal direction of the wrapping paper S.
- the control flow proceeds to step S75.
- step S75 it is confirmed whether or not the medicine package 451 sealed with the second vertical seal AS3 in step S74 is the final one. If the medicine package 451 sealed in step S74 is not the final one (step S75: NO), the control flow is returned to step S72, and if it is the final one (step S75: YES), a series of controls are performed. The flow is complete.
- step S74-1 the rotation of the vertical seal members 450e and 450e is started so that the heating units 450k and 450k are in a positional relationship so as to form the second vertical seal AS3. Thereafter, the control flow proceeds to step S74-2.
- step S74-2 has the time (contact start timing) reached when the heating parts 450k, 450k of the vertical seal members 450e, 450e start to contact the wrapping paper S as shown in FIG. It is confirmed whether or not.
- the contact start timing has been reached can be confirmed by various methods.
- a method of confirming whether or not an angle or a posture at which the heating parts 450k and 450k of the vertical seal members 450e and 450e start to come into contact can be considered.
- step S74-3 control for temporarily stopping the rotation of the vertical seal members 450e and 450e is performed. Accordingly, the vertical seal members 450e and 450e are temporarily stopped in a posture in which the heating units 450k and 450k start to contact each other. Thereafter, the control flow proceeds to step S74-4.
- step S74-4 a vertical seal (first vertical seal) that has already been formed by the seal device 450F inside the inner region of the medicine introduction unit 80 and inside the wrapping paper S by the detection unit 476.
- the presence / absence of the tablet M in the region upstream of the transport direction with respect to the seal AS1 or the second vertical seal AS3) is detected (sealing detection).
- the illumination device 450b is turned on, and the internal region of the medicine introduction unit 80 is illuminated. Data detected by the detection unit 476 is input to the control unit 40.
- the detection data may be any data as long as it is effective for determining the presence or absence of the tablet M, but in this embodiment, image data captured by the camera 467a is input to the control unit 40 as detection data. . Specifically, when the tablet M does not exist, an image of only the drug introduction unit 80 is acquired as shown in FIG. 50A, and when the tablet M exists, the tablet M as shown in FIG. 50B. An image containing is acquired. Such image data is input to the control unit 40 as detection data. Thereafter, the control flow proceeds to step S74-5.
- step S74-5 the presence or absence of the tablet M based on the detection data (image data) obtained by the seal detection performed in step S74-4 by the determination unit 102 of the control unit 40 A determination is made about.
- the presence / absence of the tablet M is determined by a technique utilizing image data such as image analysis.
- the determination of the presence or absence of the tablet M may be performed by any method. For example, when the tablet M is not present, an image obtained by the camera 92 is prepared as a master image and is actually obtained by the camera 476a. The presence of the tablet M may be determined using the image and the master image.
- a different master image is selected depending on the type of the wrapping paper S. It is desirable to prepare. Specifically, the wrapping paper S is folded in two and supplied in a state where the two surfaces overlap each other, but the wrapping paper S is transparent on both sides, or is transparent on one side. Some have an opaque portion (for example, a band having a color such as white) provided on the other surface. It is obvious that there is a difference in the image obtained by the camera 476a between the case where the wrapping paper S is used as in the former and the case in which the wrapping paper S has an opaque portion as in the latter. Therefore, it is desirable to prepare a master image according to the type of the packaging paper S in order to cope with the difference in the packaging paper S. After the determination on the tablet M is made as described above, the control flow proceeds to step S74-6.
- step S74-6 it is confirmed whether or not the tablet M is detected as a result of the determination in step S74-5.
- step S74-6 YES
- step S74-6: NO the tablet M to be packaged in the previous medicine package 451 (the medicine package 451a on the lower side in the figure).
- step S74-7 control for resuming the rotation of the vertical seal members 450e and 450e once stopped in step S74-3 is performed. Thereby, as for the vertical seal members 450e and 450e, the heating parts 450k and 450k begin to come into surface contact, and the second vertical seal AS3 is formed.
- step S74-8 it is confirmed whether or not the formation of the second vertical seal AS3 is completed.
- step S74-8: YES it is determined that the formation of the second vertical seal AS3 is completed.
- step S74-8: NO the control in step S74-8 is continued.
- step S74-9 processing for dealing with the fact that leakage of the tablet M has been confirmed in step S74-6 described above (processing for defective packaging) is performed. Specifically, a process of notifying that a packaging failure has occurred by a method such as voice, image display, or lamp lighting is executed as a packaging failure process. Thereby, a series of control flows shown in FIG. 52 is completed.
- the tablet M for packaging into the next medicine package 451 is introduced after the timing of starting the sealing of the medicine package 451 into which the tablet M has been introduced. It is determined that a packaging failure has occurred on the condition that the presence of the tablet M is detected by the detection unit 476 within a period before the timing. By doing in this way, the packaging failure by the tablet M overflowing from the medicine package 451 which should be originally packaged can be detected accurately, and the labor required for inspection can be suppressed to the minimum.
- the sealing of the medicine package 451 in which the tablet M is introduced is started at the timing when the vertical seal member 450e contacts the packaging paper S to form the second vertical seal AS3.
- the timing is set, and at this timing, the joining of the wrapping paper S by the sealing device 450F is interrupted, and the tablet M is detected by the detection unit 476.
- the packaging failure due to the tablet M leaking out when the medicine package 451 is sealed can be detected with higher detection accuracy. Further, it is possible to suppress problems such as the tablet M leaking from the medicine package 451 being bitten between the vertical seal members 450e and 450e.
- the detection unit 476 detects the tablet M by regarding the timing at which the vertical seal member 450e contacts the packaging paper S to form the second vertical seal AS3 as the timing at which the medicine package 451 is sealed.
- the present invention is not limited to this, and similar processing may be performed by regarding other timings as timings at which the medicine package 451 starts to be sealed.
- the example in which the detection unit 476 detects the tablet M at the timing when the sealing of the medicine package 451 is started has been described, but the present invention is not limited to this, and the The presence of the tablet M may be detected at an arbitrary timing (for example, after the sealing) within a period until the sealing is completed after the sealing is started.
- this invention is not limited to this. Specifically, when the presence or absence of the tablet M is detected by the detection unit 476, sealing by the vertical seal member 450e may not be stopped, or the seal formation speed by the vertical seal member 450e may be reduced.
- the roller-like vertical seal members 450e and 450e and the horizontal seal members 450f and 450f can be individually driven and controlled as the seal device 450F so as to be compatible with medicine packages 451 having different lengths.
- the present invention is not limited to this example. That is, when the bag length of the medicine package 451 may be constant, the vertical seal members 450e and 450e and the horizontal seal members 450f and 450f may be driven integrally with the seal device 450F.
- the packaging paper S is sandwiched and sealed by the roller-shaped vertical seal members 450e and 450e and the horizontal seal members 450f and 450f to form the medicine package 451, but the present invention is limited to this.
- the medicine package 451 may be formed by sealing the wrapping paper S in another form and method.
- the medicine package 451 is formed by folding the wrapping paper S in two and sealing the doubled portion is shown, but the present invention is not limited to this. Absent. Specifically, the medicine package 451 may be formed by supplying two sheets of wrapping paper S and overlapping and joining them.
- the detection unit 476 includes a camera 476a as a detection device for detecting the tablet M, but the present invention is not limited to this, and the presence of the tablet M can be detected. Any thing can be used.
- the detection unit 476 an optical sensor, an infrared sensor, or the like that is capable of detecting the presence or absence of the tablet M inside the wrapping paper S and upstream of the sealing device 450F in the conveyance direction of the wrapping paper S. It may be provided.
- an optical sensor or the like is used as the detection unit 476, it is preferable to take measures so that sufficient detection accuracy can be obtained in consideration of the characteristics of these sensors.
- the detection distance of the optical sensor or the like when the detection distance of the optical sensor or the like is set short, there is a concern that the detection accuracy of the presence of the small tablet M is lowered. That is, when the size of the tablet M is small, the distance from the optical sensor or the like to the surface of the tablet M is larger than when the size of the tablet M is large. Therefore, when the detection distance of the optical sensor or the like is set short, there is a concern that the detection accuracy of the small tablet M is lowered. On the other hand, if the detection distance of the optical sensor or the like is set long, there is a concern that the vertical seal member 450e or the like may be detected as the tablet M. Therefore, when an optical sensor or the like is employed in the detection unit 476, it is preferable to take measures such as setting the detection distance in consideration of the size of the tablet M to be handled.
- the detection unit 476 is disposed at any position as long as it can detect the presence of the tablet M on the inner side of the packaging paper S and upstream of the sealing device 450F in the conveyance direction of the packaging paper S. May be. Specifically, as shown by a two-dot chain line in FIG. 49, a detection unit 476X similar to the detection unit 476 is arranged on the distal end side of the drug introduction unit 80, or is detected further upstream than the drug introduction unit 80. The part 476Y may be arranged.
- a drop sensor for detecting the drop of the tablet M may be provided in the medicine introduction unit 80 or the like. If the determination condition is that the tablet M is detected by the drop sensor and the leakage of the tablet M is not detected based on the detection result by the detection unit 476, whether the tablet M dispensed for packaging has been accurately packaged. Whether or not can be detected with higher accuracy.
- the detection unit 476 is used to detect that the tablet M is accurately packaged at the time of sealing the medicine package 451 is shown, but the detection unit 476 is used for other purposes. May be.
- the presence of the tablet M may be detected by the detection unit 476 when the tablet M for packaging is to be introduced from the drug introduction unit 80 into the packaging paper S. That is, the detection unit 476 may be used not only for the above-described sealing detection, but also for introduction detection for detecting the introduction of the tablet M into the packaging paper S by the medicine introduction unit 80. In such a case, the detection unit 476 can be effectively used to check whether or not the tablet M is supplied into the wrapping paper S.
- the present invention is not limited to this. That is, after the timing of starting the sealing of the medicine package 451 into which the tablet M has been introduced, the timing at which the tablet M for packaging into the medicine package 451 to be formed next through the medicine introduction unit 80 is introduced. As long as it is within the previous period (hereinafter also referred to as “detectable period”), the tablet M may be detected by the detection unit 476 at any timing.
- the detection of the tablet M by the detection unit 476 is performed after the sealing of the medicine package 451 into which the tablet M has been introduced (after the formation of the second vertical seal AS3), and then the medicine package 451 is formed. It may be performed within a period before the timing at which the tablet M is introduced.
- the detection by the detection unit 476 is not only detected at a predetermined timing (time point) within the above-described detection period, but is also continued for a predetermined period included in the above-described detectable period, It may be detected intermittently within a possible period.
- the seal device 450F is exemplified by a roller-like member composed of the vertical seal members 450e and 450e and the horizontal seal members 450f and 450f, which can be joined by sandwiching the wrapping paper S.
- the present invention is not limited, and a paper that can be joined to the wrapping paper S by other methods can be used instead of the sealing device 450F.
- a pair of plate-like heating bodies having a planar shape such as a T-shape, and a paper sheet S sandwiched between the heating bodies can be joined instead of the sealing device 450F of the present embodiment. You may use for.
- the input timing when the tablet is input is the detection timing of the tablet by the passage detection sensor 475.
- the loading timing is set in advance as the timing at which the tablet dispensed from the sheet feeding cassette 41 or the hand handling unit 42 in the tablet packaging device 4 is loaded from the rotating unit 44 into the medicine introduction unit 80. Is the timing.
- the imaging range of the camera 476a includes the medicine introduction unit 80, the tablet, and the medicine package 451.
- the tablet introduced into the medicine introduction unit 80 or the medicine package 451 is photographed, but photographed at the contact start timing. In the photographed image, the tablet is not photographed because the tablet moves to the downstream side of the vertical seal member 450e.
- the control unit 40 stores an image captured by the camera 476a in the storage unit 22 and transmits it to the server 2. Thereby, in the tablet packaging device 4 or the server 2, the control unit 40 or the control unit 21 can realize a packaging view display function for displaying the captured image.
- the control unit 21 selects the medicine A medicine pack individual information screen D306 showing a list of tablets contained in the package 451 is displayed.
- FIG. 53 is a diagram showing an example of the medicine package individual information screen D306.
- the medicine package individual information screen D306 displays the medicine name, the correct image, the photographed image, etc. of each tablet contained in the third medicine package 451 in the packaging process. Is done.
- the background of the display area of the captured image is the automatic image for the tablet whose inspection result of the automatic inspection process is an error. It is displayed in the sixth specific color such as red indicating that the result of the inspection process is an error.
- the medicine pack individual information screen D306 displays an operation key K371, and the control unit 21 determines that the tablet stored in the medicine package 451 is in the detection unit in response to the operation of the operation key K371.
- a packaging screen D307 on which a photographed image photographed by 476 is displayed is displayed.
- FIG. 54 shows an example of the packaging screen D307.
- the packaging part D307 is photographed by the detection part 476.
- Photographed images of the tablets are displayed in the order of photographing.
- the imaging range captured by the captured image includes the tablet introduction unit 80, the tablet, and the medicine package 451. Thereby, the user can easily grasp the state when each tablet falls onto the medicine package 451 with reference to the packaging screen D307.
- the control unit 21 may cause the packaging view D307 to display a photographed image photographed at the contact start timing instead of the photographed image photographed at the insertion timing or together with the photographed image. In addition, when the occurrence of a back shift is detected by the back shift detection function, the control unit 21 displays a message indicating that the back shift has occurred and a photographed image captured at the contact start timing. It may be displayed on the view D307.
- the configurations and functions described in the first embodiment and the second embodiment can be arbitrarily combined.
- the subject having the configuration and processing function described in the first embodiment and the second embodiment is not limited to that described here, and the server 2 and the client terminal included in the inspection support system 1 3 and at least one of the tablet packaging devices 4 and the like.
- a process corresponding to the automatic inspection process is executed by the control unit 21 in the server 2
- a process corresponding to the inspection support process is executed by the control unit 40 in the tablet packaging device 4. Can be considered.
- the inspection support system 1 includes the tablet identification information and the prescription data acquired from the photographed image by the control unit 40.
- An image inspection process is performed in which the identification information of the tablet corresponding to the type of tablet is collated using an image. Specifically, in the image inspection process, an image corresponding to tablet identification information included in the photographed image and a normal image registered in advance corresponding to tablet identification information included in the prescription data are included. A matching process for matching by image comparison is executed.
- the said normal image corresponding to the identification information of the said tablet contained in the said prescription data is a registration image previously registered into the said pharmaceutical master etc. as a correct image of the tablet corresponding to the said identification information.
- the identification information of the target tablet of the image inspection process is “ABC111”, and the correct image P11 corresponding to the tablet is registered in the medicine master, and the imaging A case where a captured image P12 of a tablet whose identification information is “ABC114” is captured as an image will be described as an example.
- the matching result is detected by comparing the positive image P11 and the captured image P12, and the matching result is obtained when the matching score is equal to or higher than a preset threshold value. Are determined to match.
- the number of black pixels included in the photographed image P12 among the black pixels included in the positive image P11 is counted, and the total number of black pixels of the positive image P11 is counted.
- the ratio of the number of black pixels included in the captured image P12 is calculated as the degree of coincidence. For this reason, when the captured image P12 includes most of the normal image P11, the degree of matching is increased even when the captured image P12 includes extra black pixels. Results may be consistent.
- the composite image P13 in FIG. 55 is an image obtained by combining the normal image P11 and the captured image P12.
- the identification information “ABC111” in the normal image P11 and the identification information “ABC114” in the captured image P12 differ by one character, but the captured image of the positive image P11
- the only areas not included in the image P12 are the areas P21 and P22. Therefore, there is a possibility that the degree of coincidence increases and the collation result becomes coincident.
- a high-accuracy image inspection is performed by executing an image inspection process (see FIG. 56) described later by the control unit 40 of the tablet packaging device 4. Processing is realized.
- the image inspection process is executed instead of steps S62 to S64 in the automatic inspection process (see FIG. 34).
- the image inspection process may be executed by the control unit 21 of the server 2.
- the medicine master is stored in the storage unit 49, and in the medicine master, similar tablets are previously stored in association with similar drugs. For example, tablets whose tablet identification information is different by only one letter are associated with each other as similar drugs.
- the same similar group code is set for similar tablets.
- a portion similar to another tablet among the tablets is set in advance as a similar portion.
- the same similar group code is set for the tablet corresponding to the identification information “ABC111” and the tablet corresponding to the identification information “ABC114”, and “1”, “ It is assumed that the first digit region corresponding to “4” is set as the similar portion.
- the similar group code and the similar part are registered in the pharmaceutical master by the control unit 40 in accordance with a user operation using the operation display unit 48, for example.
- the inspection support system 1 a photographed image of a tablet to be packaged is acquired in the packaging process performed by the tablet packaging device 4 before the image inspection process is performed. Further, the control unit 40 also determines whether or not the tablet is in the vertical rotation state when acquiring the captured image in the packaging process, and if the tablet is in the vertical rotation state, A process for changing the orientation of the tablet is executed. And the said control part 40 specifies the imaging
- the photographed image used in the image inspection process is an image including at least the identification information of the tablet among the images photographed by the camera 462 or 463. For example, a part of the photographed image is trimmed. An image, or the captured image itself.
- step S81 the control unit 40 executes a first matching process for matching the entire captured image with the normal image. Specifically, the control unit 40 associates the normal image registered with the pharmaceutical master in association with the tablet that is the target of the image inspection process, and the tablet imaged in the packaging process. The degree of coincidence is acquired by executing the first matching process on a captured image.
- the collation target in the first matching process may be a preset identification information area where the identification information exists in each of the normal image and the captured image.
- step S82 the control unit 40 determines whether or not the degree of coincidence acquired in step S81 is greater than or equal to a preset first threshold value.
- the first threshold value is a value set in advance to determine that the collation result of the first matching process is coincident.
- the process proceeds to step S83, and if it is determined that the degree of coincidence is less than the first threshold ( S82: No), the process proceeds to step S821.
- step S821 the control unit 40 determines whether or not the degree of coincidence acquired in step S81 is greater than or equal to a preset second threshold value.
- the second threshold is a value lower than the first threshold, and is a value set in advance to determine that the collation result of the first matching process is inconsistent. If it is determined that the degree of coincidence is greater than or equal to the second threshold (S821: Yes), the process moves to step S822, and if it is determined that the degree of coincidence is less than the second threshold ( S821: No), the process proceeds to step S823.
- Step S822 If the degree of coincidence acquired in step S81 is less than the first threshold and greater than or equal to the second threshold, the collation result between the positive image and the captured image is identical or inconsistent. This is a situation in which it is not possible to determine clearly. Therefore, in step S822, the control unit 40 records in the storage unit 49 as a result of the image inspection process for the tablet that the visual check is necessary (check required).
- Step S823 On the other hand, when the degree of coincidence acquired in Step S81 is less than the second threshold, it is clearly determined that the collation result between the normal image and the captured image is inconsistent. This is a possible situation. Therefore, in step S823, the control unit 40 records in the storage unit 49 that the collation result is inconsistent (error) as a result of the image inspection process for the tablet.
- step S83 the control unit 40 determines whether or not another tablet is registered as a similar drug in association with the tablet that is the target of the image inspection process in the drug master. Specifically, the control unit 40 determines whether or not a tablet having the same group code as the tablet that is the target of the image inspection process exists in the pharmaceutical master. If it is determined that the similar drug exists (S83: Yes), the process proceeds to step S84. If it is determined that the similar drug does not exist (S83: No), the process proceeds to step S86. To do.
- the identification information “ABC111” of the primary image P11 and the identification information “ABC114” of the captured image P12 include the characters “1” and “4”.
- the regions P21 and P22 exist as differences in the regions. However, the difference is an extremely small portion in the entire character of the identification information, and since the influence on the matching degree is small, the matching degree may be equal to or higher than the first threshold value. Note that the higher the first threshold value is set, the higher the possibility that the collation result of the first matching process becomes inconsistent due to the influence of extremely small noise.
- the control unit 40 determines whether or not there is a similar drug in the tablet that is the target of the image inspection process in the step S83. If the similar drug exists, a more detailed image inspection process is executed in the subsequent step S84. Therefore, the value of the first threshold value can be set to a certain level so as not to be greatly affected by extremely small noise.
- step S84 the control unit 40 executes a second matching process for matching only the similar portion of the captured image with the normal image. Specifically, the control unit 40 executes a second matching process set in advance with a region corresponding to the similar portion in each of the normal image and the captured image as a target of a collation range. In the second matching process, after the similar part of the photographed image is binarized, a difference between the similar part in the photographed image and the similar part in the normal image is detected, and the similarity which is a collation range The ratio of the matching area excluding the place where the difference is generated in the place is acquired as the matching degree.
- a value obtained by subtracting the number of pixels detected as the difference in the similar part from the number of pixels in the similar part in the positive image is calculated, and the ratio of the calculated value to the number of pixels in the similar part in the positive image Is calculated as the degree of coincidence. That is, in the second matching process, unlike the first matching process, even if the similar part of the photographed image includes most of the similar parts of the positive image, the second matching process is redundant in the photographed image. If the black pixel is included, the degree of coincidence is calculated in consideration of the difference. Therefore, the degree of coincidence may be lowered and the collation result may be inconsistent.
- a plurality of the similar portions may be set for the tablet.
- the second matching is performed. The degree of coincidence between the captured image and the normal image is acquired for each similar portion by the processing.
- a region corresponding to “1” of the first digit character preset as a similar portion of the identification information “ABC111” of the tablet to be subjected to the image inspection process (enlarged in FIG. 55). Only the (range) is the target of the second matching process. As described above, by limiting the target region of the second matching process, the degree of coincidence in which the influence of the difference in the similar part is largely reflected is detected. Further, in addition to the identification information “ABC114”, when a tablet such as the identification information “ABO111” is registered in the medicine master as a similar drug, the region corresponding to “C” in the captured image is also corrected. The second matching process for matching with an image is executed.
- step S85 the control unit 40 determines whether or not the degree of coincidence in the first matching process executed in step S84 is greater than or equal to a preset third threshold value.
- the third threshold value is a value set in advance to determine that the matching result of the similar part is coincident.
- the third threshold value may be the same value as the first threshold value, or may be a value greater than or equal to the first threshold value or less than the first threshold value.
- step S86 if it is determined that the degree of coincidence is equal to or greater than the third threshold (S85: Yes), the process proceeds to step S86, and if it is determined that the degree of coincidence is less than the third threshold ( S85: No), the process proceeds to step S822.
- the process may move to step S823.
- the second matching process is executed for a plurality of the similar portions in the step S84, it is determined whether or not the degree of coincidence in all the similar portions is equal to or greater than the third threshold value. If all the matching degrees are equal to or greater than the third threshold, the process proceeds to step S86.
- step S86 the control unit 40 determines that the collation result in the image inspection process is the same, and sets “OK” as the image inspection result to the target tablet for the image inspection process in the prescription data. Record in association.
- the image inspection process As described above, in the image inspection process according to the present embodiment, after the first matching process is performed on the whole of the photographed image and the normal image in the image inspection process for the tablet having a similar medicine.
- the second matching process is executed for the similar part.
- the inspection accuracy in the image inspection process for tablets containing similar drugs can be increased.
- only the first matching process for the whole of the captured image and the normal image is executed for a tablet having no similar medicine. Thereby, about the tablet in which a similar medicine does not exist, it is possible to reduce the processing load of the said image inspection process, or to shorten the time required for the said image inspection process.
- step S81 the case where the first matching process is performed on the entire captured image has been described.
- the second matching process may be performed on the entire captured image in the same manner as in step S84. That is, in the step S81, it is conceivable that the degree of coincidence considering the difference is detected for the entire captured image and the normal image.
- step S84 the case where the second matching process is executed for a similar part in the photographed image has been described.
- the first matching process is executed for a similar part in the photographed image as in step S81. May be. That is, in the step S84, it is conceivable that the amount of overlapping portions in the similar portions of the captured image and the normal image is detected as the degree of coincidence.
- the second matching process is executed only for the entire photographed image and the normal image.
- the degree of coincidence acquired is lower than that in the first matching process for detecting the number of black pixels overlapping the positive image.
- the second matching process is executed only for the entire photographed image and the normal image.
- control unit 40 is normal when the degree of coincidence detected in the second matching process is equal to or higher than the first threshold value, abnormal when it is lower than the second threshold value, and the first threshold value. If it is less than the second threshold value, it is determined that the check is necessary.
- step S91 the control unit 40 is similar in shape, such as height, width, and area, to a tablet to be registered in the information registration process (hereinafter referred to as “registered tablet”). It is determined whether or not a tablet (hereinafter referred to as “first similar target tablet”) exists in the pharmaceutical master. Here, if it is determined that the first similar target tablet exists (S91: Yes), the process proceeds to step S92, and if it is determined that the first similar target tablet does not exist (S91: No), The similar medicine registration process ends. In step S91, it may be determined that there are a plurality of the first similar target tablets.
- step S92 the control unit 40 determines whether or not there is a tablet (hereinafter referred to as “second similar tablet”) having a character area similar to that of the registration target tablet among the first similar target tablets. to decide. Specifically, the control unit 40 acquires the sum of gray values for each of the captured image and the normal image, and the character areas are similar when the difference between the sums of the gray values is within a preset threshold value. Judge that. If it is determined that the second similar tablet exists (S92: Yes), the process proceeds to step S93. If it is determined that the second similar tablet does not exist (S92: No), the similar The medicine registration process ends. In step S92, it may be determined that there are a plurality of the second similar target tablets.
- step S93 the control unit 40 determines whether or not there is a tablet having a tablet color similar to the registration target tablet (hereinafter referred to as “third similar tablet”) among the second similar target tablets. to decide. Specifically, the control unit 40 acquires the sum of each of the R value, the G value, and the B value for each of the captured image and the positive image, and the difference between the sums of the R value, the G value, and the B value. Is within the preset threshold value, it is determined that the tablet colors are similar. If it is determined that the tablet colors are similar (S93: Yes), the process proceeds to step S94. If it is determined that the tablet colors are not similar (S93: No), the similar drug registration process is performed. finish. In step S93, it may be determined that there are a plurality of the third similar target tablets.
- step S94 the control unit 40 executes a matching process on the whole of the normal image of the third similar target tablet and the captured image of the registration target tablet. Specifically, the control unit 40 performs the first matching process or the second matching process as the matching process for the normal image of the third similar target tablet and the captured image of the registration target tablet. Execute to obtain the degree of coincidence.
- the said collation process is the same content as the process performed in said step S81 of the said image inspection process among the said 1st matching process or the said 2nd matching process.
- step S95 the control unit 40 determines that the result of the collation process in step S94 is the same among the third similar target tablets (hereinafter referred to as “fourth similar target tablet”). It is determined whether or not exists. Specifically, it is determined whether or not the degree of coincidence corresponding to each of the third similar target tablets is greater than or equal to the first threshold value.
- the process proceeds to step S96, and when it is determined that the fourth similar target tablet does not exist (S95: No), The similar medicine registration process ends.
- steps S94 to S95 it is determined whether or not the collation result is determined to be the same in steps S81 to S82 of the image inspection process (see FIG. 56).
- step S95 it may be determined that there are a plurality of the fourth similar target tablets.
- step S96 the control unit 40 registers the registration target tablet and one or a plurality of the fourth similar target tablets as similar drugs in the pharmaceutical master. Specifically, the control unit 40 registers one or a plurality of similar group codes attached to one or a plurality of the fourth similar target tablets as the similar group codes of the registration target tablets. That is, the tablet whose collation result is determined to be the same in steps S81 to S82 of the image inspection process (see FIG. 56) is registered as a similar drug to the registration target tablet.
- control unit 40 registers a similar portion with each of the similar drugs in the registration target tablet, for example, in accordance with a user operation in the step S96, or the control unit 40 performs the collation processing of the step S94. Register automatically based on results.
- the similar medicine registration process is executed by the control unit 40, the similar medicine of each tablet is automatically registered in the medicine master without requiring a user operation. Work burden is reduced.
- the tablet packaging device 4 may execute the second matching process for detecting a difference between the captured image and the normal image.
- the control unit 40 transmits information regarding the difference between the captured image and the normal image to the server 2 together with the result of the image inspection process and the captured image.
- the control unit 21 can also execute the second matching process on the captured image and the normal image in the image inspection process to obtain a difference between the captured image and the normal image.
- the control unit 21 displays the difference between the captured image and the normal image when displaying the result of the image inspection process. Specifically, the control unit 21 displays the difference between the captured image and the normal image so that the difference can be visually recognized, superimposed on the enlarged image P343 displayed as an enlarged image of the captured image on the enlarged screen D341. Can be considered. In other words, the control unit 40 displays the difference on the captured image side instead of the normal image on the enlarged screen D341.
- FIG. 58 is a diagram showing another example of the enlarged screen D341.
- the control unit 21 highlights and displays the difference area A343 including the extra area and the deficient area in a manner distinguishable from other areas in the enlarged image P343.
- an area including the identification information of the tablet in the photographed image of the tablet is enlarged and displayed.
- the outer edge of the difference area A343 may be colored in a specific color such as red.
- the difference area A343 is displayed with the specific color.
- the extra area and the insufficient area are displayed in a manner that can be individually identified for the difference area A343.
- the extra area and the insufficient area may be displayed with different color borders or different color fills.
- the display of the difference area A343 may be highlighted by blinking or the like.
- control unit 21 displays the shortage area and the extra area only when the result of the image inspection process (see FIG. 56) is an error (step S823) or a check required (step S822). Can be displayed.
- the result of the image inspection process is an error or a check required, the user can easily grasp the reason by viewing the enlarged image P341 on the enlarged screen D341.
- the control unit 21 does not display when the result of the image inspection process is an error, and displays the insufficient area and the extra area only when the result of the image inspection process is a check required. It is also possible to display on D341. Thereby, the user can easily grasp the reason from the enlarged screen D341 when the result of the image inspection process is a check required, and is unnecessary in the enlarged screen D341 in other cases. Information display is omitted.
- control unit 21 in the server 2 the difference between the photographed image and the normal image is displayed by the control unit 21 in the server 2 has been described.
- control unit 40 It is also conceivable to display the difference between the captured image and the normal image on the operation display unit 48.
- a photographed image obtained by photographing a tablet with the camera 462 or 463 is registered as a normal image of the tablet.
- the identification information of the tablet is a stamp
- a shadow position in the tablet on the tablet rotating unit 441 photographed by the camera 462 or 463 or a light / dark unevenness due to the unevenness of the stamp is detected by the camera 462. Or it may change with the direction of the said tablet seen from 463. Therefore, a difference in the position of a shadow or brightness unevenness included in the normal image may affect the accuracy of the image inspection process.
- the shadow in the said tablet may arise, for example, when there is a height difference between a pair of said rotation rollers 100 of the said tablet rotation part 441.
- the shadow of the tablet may occur on the side of the rotating roller 100 that is close to the camera 462 or 463 in the pair of rotating rollers 100.
- the illumination device 468 emits light from the side or obliquely upward toward the tablet of the tablet rotating unit 441. Therefore, the tablet is rotated by the rotating roller 100. There is a risk of shadowing.
- each of the normal images is an image taken in a plurality of states in which the directions of the tablets as viewed from the camera 462 or 463 are different, that is, in a plurality of states in which the positions of shadows or unevenness in brightness are different.
- FIG. 59 is a diagram showing normal images P31 to P38, which are examples of the plurality of normal images. More specifically, each of the normal images P31 to P38 has an angle of identification information of the tablet with respect to the camera 462 or 463 of 0 °, 45 °, 90 °, 135 °, 180 °, 225 °, 270 °. It is registered in the medicine master as a normal image corresponding to the case of 315 °. In the regular images P31 to P38, a shadow is formed in the right direction in FIG. 59, and the positional relationship between the identification information “ABC111” and the shadow in each of the regular images P31 to P38 is different. Further, although not represented in FIG. 59, when the identification information of the tablet is a stamp, the brightness of the stamp may be different for each of the normal images P31 to P38.
- the collation with the said picked-up image and the said positive image uses the positive image of the angle close
- the control unit 40 gradually rotates the captured image by 360 °, and sets one specific positive image (for example, the positive image P31) set in advance among the normal images. ) Is detected, and the rotation angle of the photographed image when the coincidence is the highest is detected as a specific angle indicating the tilt angle of the photographed image.
- the said control part 40 specifies the positive image corresponding to the angle nearest to the said specific angle among the said positive images, and performs the said image inspection process using the normal image and the said picked-up image. As a result, the influence of shadows or brightness unevenness included in the normal image is reduced, and the inspection accuracy in the image inspection process is increased.
- control part 40 collates using the positive image corresponding to the angle nearest to the said specific angle among several said positive images, each said positive image and the said picked-up image are collated separately. Compared to the case, the processing load is reduced or the processing time is shortened.
- the control unit 40 individually obtains the degree of coincidence between each of the positive images and the captured image, and determines that the collation results are coincident when all the coincidence degrees are equal to or greater than the first threshold value. This is also conceivable as another embodiment.
- control unit 40 selects a positive image corresponding to an angle closest to the specific angle from the plurality of positive images, it may be considered to select the plurality of positive images. Specifically, depending on the interval between the angles of the plurality of positive images, there may be no positive image corresponding to an angle that approximates the specific angle. Therefore, it is conceivable that the control unit 40 selects, for example, two or three normal images closest to the specific angle among the plurality of normal images. In this case, the control unit 40 individually obtains the degree of coincidence between each of the selected positive images and the photographed image, and the matching result matches when all the degree of coincidence is equal to or greater than the first threshold value. It is judged that. This reduces the processing load compared to when each of the positive images is individually compared with the photographed image, and suppresses the influence of shadows or unevenness in the normal image while suppressing the influence of the image inspection. Inspection accuracy in processing can be increased.
- control unit 40 selects a plurality of the positive images when the difference between the shooting angle corresponding to the positive image closest to the specific angle and the specific angle is equal to or greater than a preset threshold value. However, if it is less than the threshold value, it is conceivable to select one normal image.
- a photographed image obtained by photographing a tablet with the camera 462 or 463 is registered as a normal image of the tablet. Further, in the image inspection process, a matching process between a captured image obtained by photographing a tablet with the camera 462 or 463 and the normal image is executed.
- the thickness of the character of the identification information included in the photographed image changes depending on, for example, the matching accuracy of the focus (focus) when the photographed image is photographed. is there.
- the identification information of the tablet is stamped, the thickness of the character of the identification information may change depending on the orientation of the tablet viewed from the camera 462 or 463. Therefore, the difference in character thickness of the identification information may affect the accuracy of the image inspection process.
- each of the positive images is an image in which the thickness of the character of the tablet identification information included in the captured image is changed with respect to the captured image captured by the camera 462 or 463 and registered as the normal image. It is.
- FIG. 60 is a diagram showing normal images P41 to P45 which are examples of the plurality of normal images.
- the control unit 40 uses the captured image of the tablet as a normal image P41, and the normal image P42 in which the thickness of the character of the identification information in the normal image P41 is thickened in two stages. Then, a normal image P43 is generated in which the thickness of the character of the identification information is increased by a predetermined level.
- the thickness of characters in the regular image P42 is four times that of the regular image P41
- the thickness of characters in the regular image P43 is twice that of the regular image P41.
- control unit 40 uses the photographed image as a normal image P41, and a normal image P44 in which the thickness of the character of the identification information in the normal image P41 is narrowed in two stages, and the thickness of the character of the identification information.
- a normal image P45 with a predetermined height is generated.
- the thickness of characters in the regular image P44 is 1/4 times that of the regular image P41
- the thickness of characters in the regular image P45 is 1/2 times that of the regular image P41.
- control unit 40 registers a plurality of corrected positive images acquired as a result of executing an illumination cancellation process, a contrast correction process, a character area extraction process, and the like for the plurality of positive images in the pharmaceutical master. It is possible. Specifically, in the illumination cancellation process, a fitting process is performed on a plurality of the positive images so that a preset luminance change occurs, thereby removing the influence of illumination at the time of capturing the captured image. . For example, in the fitting process, a locally bright region or a locally dark region is suppressed from the plurality of normal images.
- the range of the minimum value and the maximum value of the luminance in the plurality of positive images is expanded to a range from 0 to 255, thereby enhancing the contrast between the character and the background.
- a black region is extracted from the plurality of the normal images using a discriminant analysis method (Otsu's threshold method), and the black region is extracted as a character region of the identification information.
- the captured image and the normal image are collated using a plurality of the normal images.
- the control unit 40 individually executes a matching process between each of the normal images and the captured image.
- the said control part 40 specifies the highest matching degree detected by each of the said matching processes as a collation result of the said captured image, and determines the propriety of the said tablet according to the said matching degree.
- the case where a plurality of the normal images having different character thicknesses as the normal images are registered in the medicine master has been described as an example, but the present invention is not limited thereto.
- the control unit 40 generates a plurality of captured images having different character thicknesses in the captured image from the captured image.
- the control unit 40 individually performs a matching process on each captured image with the normal image registered in the pharmaceutical master, and specifies the highest matching degree as a matching result of the captured image.
- the suitability of the tablet can be determined according to the degree of coincidence.
- a plurality of the normal images having different character thicknesses as the normal images are registered in the medicine master, and the control unit 40 performs a plurality of shootings in which the character thicknesses in the captured images are different from the captured images. It is also possible to generate an image.
- the control unit 40 individually executes matching processing between each of the captured images and each of the normal images, specifies the highest matching degree as a matching result of the captured images, and according to the matching degree It can be considered to determine the suitability of the tablets.
- Photographed image of the tablet taken before the tablets dispensed from one or both of the tablet cassette and the hand-rolling unit based on the prescription data are packaged by the packaging material in the tablet packaging device.
- An inspection support system comprising: an inspection display processing unit that displays a result of inspection processing executed based on the identification information of the tablet read from the photographed image of the tablet and the prescription data.
- Supplementary Note 3 Supplementary Note 1 or 2 further comprising a list display processing unit that, when the result of the inspection process is an error, lists the cause of the error together with the date and time of taking the tablet that caused the error. 2.
- the inspection support system according to 2. Thereby, the pharmacist can easily grasp the cause of the error, the taking date and the taking time of the tablet by referring to the list display.
- the inspection display processing unit displays the date of taking in one of the columns or rows and the time of taking the other in the arrangement form in which the date of taking is displayed in the display location corresponding to each combination of the time taken and the time taken.
- the inspection support system according to any one of supplementary notes 1 to 3, wherein the photographic images of the tablets corresponding to the combination are arranged and displayed in a predetermined display order in a direction parallel to the order of the dosing timing.
- the inspection display processing unit includes the tablets corresponding to each combination of the tablet name and the packaging order in an arrangement format in which the tablet name is displayed in one of the columns or rows and the packaging order is displayed in the other.
- the inspection support system according to any one of appendices 1 to 3, wherein the photographed image is displayed.
- the inspection display processing unit includes at least an original image in which the identification information of the tablet is read, and a plurality of pieces corresponding to different outer peripheral surfaces of the tablet photographed for each tablet in the tablet packaging device.
- the inspection support system according to any one of appendices 1 to 5, wherein a photographed image is displayed for each tablet. Thereby, for example, when it is difficult to determine the suitability of the tablet by using only the photographed image of one tablet, the pharmacist can determine the suitability by referring to the photographed images of the plurality of tablets.
- the inspection display processing unit was imaged for each tablet in the tablet packaging device as a captured image of the tablet that caused the error.
- the inspection support system according to any one of appendices 1 to 6, wherein a plurality of photographed images corresponding to different outer peripheral surfaces of the tablet can be displayed for each tablet. Thereby, the pharmacist can determine suitability by referring to, for example, photographed images of a plurality of the tablets.
- Appendix 10 The inspection support system according to any one of appendices 1 to 9, wherein the inspection display processing unit displays a registered image registered in advance as a correct image of the tablet together with a photographed image of the tablet. Accordingly, the pharmacist can easily determine whether the photographed image of the tablet is appropriate by comparing the registered images.
- the inspection support system is connected to a plurality of dispensing devices used in the dispensing process executed based on the prescription data, and the inspection result of the medicine dispensed by the dispensing device is displayed.
- the inspection result of the medicine dispensed by the dispensing device is displayed.
- An operation display processing unit for displaying the operation unit, and a part or all of the dispensing process executed by the dispensing device in response to a user operation on the operation unit displayed by the operation display processing unit.
- the inspection support system according to any one of appendices 1 to 12, further comprising a re-execution processing unit for re-execution.
- the pharmacist can re-execute part or all of the dispensing process individually for each of the dispensing devices in the inspection support system, so that the work efficiency of the pharmacist is improved.
- First tablet packaging capable of executing a packaging process for storing tablets in a packaging material based on prescription data and capable of performing a predetermined image inspection process for checking the suitability of the packaging process
- a device a second tablet packaging device in which the packaging process is executable and the image inspection process is not executable or the function of the image inspection process is disabled, and the contents of the prescription data and Selection that causes the first tablet packaging device to execute the packaging process based on the prescription data when it is determined that the first priority condition set in advance is satisfied based on at least one of the contents of the packaging process
- An inspection support system including a processing unit.
- the first tablet packaging device was photographed by the photographing unit for photographing the tablet and acquiring a photographed image of the tablet before the tablet was accommodated in the packaging material.
- the imaging unit captures a plurality of captured images corresponding to different outer peripheral surfaces of the tablets for each tablet, and the image inspection processing unit includes the plurality of captured images of the tablets.
- the second tablet packaging device detects the number of tablets before or after the tablets are stored in the packaging material, and is detected by the tablet number detection unit.
- An inspection support system according to any one of appendices 16 to 18, further comprising: a counting inspection processing unit that executes a counting inspection processing for comparing the number of the tablets that have been made and the prescription data.
- Each of the first tablet packaging device and the second tablet packaging device includes a tablet cassette and a handing unit used for supplying the tablets to be packaged in the packaging process,
- the first priority condition is that the handing unit is used in the packaging process, the number of tablets of the tablets to be packaged in the packaging process is greater than or equal to a preset threshold value, or the packaging 20.
- the inspection support system according to any one of supplementary notes 16 to 19, which includes at least one of the tablets that are packaged by the processing being a high-risk drug set in advance.
- the selection processing unit selects the first tablet packaging device or the second tablet packaging device according to a preset second priority condition, and The packaging process based on the prescription data is executed, and the second priority condition selects an apparatus with many types of the tablets filled in the tablet cassette among the tablets included in the prescription data.
- the inspection support system according to appendix 20 including either selecting a device with a small number of waiting times for the present packaging process or selecting a device arbitrarily designated by the user.
- the selection processing unit selects the first tablet packaging device or the second tablet packaging device according to a preset second priority condition, and 21.
- the inspection support system according to any one of supplementary notes 16 to 20, wherein the packaging process based on prescription data is executed.
- the identification information of the tablet is acquired from the photographed image of the tablet taken before the tablet is stored in the packaging material based on the prescription data in the tablet packaging device, and the identification information of the tablet and the prescription
- An image inspection processing unit for executing an image inspection process for verifying data, and a counting inspection process for verifying the number of the tablets detected on the downstream side of the tablet photographing position in the tablet dispensing route and the prescription data
- An inspection support system comprising: a count inspection processing unit that executes
- a pre-registered image corresponding to a photographed image of the tablet taken before the tablet is stored in the packaging material based on the prescription data and the kind of the tablet included in the prescription data in the tablet packaging device.
- An image inspection processing unit for executing an image inspection process for collating the original image, and an inspection capable of displaying a difference between the identification information of the tablet detected in the image inspection process by the image inspection processing unit and the normal image
- An inspection support system comprising a display processing unit.
- the image inspection processing unit executes a first verification process for performing image verification between the captured image of the tablet and the normal image, and the verification result by the first verification process is identical, and the image verification is performed.
- a first verification process for performing image verification between the captured image of the tablet and the normal image
- the verification result by the first verification process is identical
- the image verification is performed.
- an area corresponding to a preset similar position similar to the similar drug in the captured image an area corresponding to the similar position in the normal image
- the tablet packaging device according to appendix 24, which executes a second collation process for performing image collation with.
- a tablet packaging apparatus capable of performing a packaging process for storing a tablet in a packaging material based on prescription data, wherein the drug introduction unit guides the tablet to the packaging material, and the tablet is the packaging material
- a tablet packaging device comprising: an imaging unit that captures an imaging range including the drug introduction unit, the tablet, and the packaging material when housed in a container.
- a tablet packaging apparatus capable of performing a packaging process for storing tablets in a packaging material based on prescription data, wherein the tablet rotating unit rotates the tablet, and the tablet rotating unit rotates the tablet.
- An imaging processing unit that performs imaging processing for imaging a tablet a plurality of times, and identification information for the tablet from a captured image that satisfies a preset condition among a plurality of captured images of the tablet imaged by the imaging processing unit.
- a tablet packaging apparatus comprising: a registration processing unit that acquires and registers the identification information of the tablet as verification source data corresponding to the type of tablet.
- the identification information of the tablet is an image of a part or all of the image of the tablet, and the verification source data is a normal image registered in advance corresponding to the type of the tablet.
- Item 30. The tablet packaging device according to appendix 29, wherein the image inspection process is executed using the photographed image.
- the first imaging unit provided corresponding to the case where the tablet being rotated by the tablet rotating unit can be photographed and the identification information of the tablet is stamped, and the identification information of the tablet are printed
- An imaging unit including a second imaging unit provided corresponding to the case, and a specific processing unit that specifies whether the identification information of the tablet registered by the registration processing unit is stamped or printed according to a user operation And when the photographing processing unit captures the photographed image used when the registration processing unit registers the verification source data, the first processing unit according to a specific result by the specific processing unit.
- the tablet packaging device according to appendix 29, wherein either the photographing unit or the second photographing unit is selected to photograph the tablet.
- Supplementary Note 33 Any one of Supplementary Notes 28 to 32, wherein the registration processing unit registers a photographed image selected by a user operation among a plurality of photographed images of the tablet photographed by the photographing processing unit as the verification source data.
- a tablet packaging device according to claim 1.
- the registration processing unit accepts a user operation for designating an upward position in the captured image registered as the verification source data, and compares the information indicating the upward position in the captured image with the verification.
- the tablet packaging device according to any one of appendices 29 to 33, which can be registered as a part of the original data.
- the registration processing unit compares the initial correct image with the photographed image stored in a medicine database in which the correspondence between at least the tablet and the initial correct image corresponding to the tablet is stored in advance.
- the tablet packaging device according to any one of supplementary notes 29 to 34, wherein information indicating the position in the upward direction in the photographed image can be registered as a part of the verification source data.
- the registration processing unit accepts a user operation for designating an upward position in the captured image registered as the verification source data so that the upward position in the captured image is at the top.
- the tablet packaging device according to any one of supplementary notes 29 to 35, wherein a positive image obtained by rotating the photographed image can be registered as the verification source data.
- the image inspection processing unit executes a first verification process for performing image verification between the captured image and the verification source data, and the verification result by the first verification process is identical, and the image verification process is performed.
- a similar drug similar to the target tablet an area corresponding to a preset similar position similar to the similar drug in the captured image and an area corresponding to the similar position in the verification source data
- the tablet packaging device according to any one of supplementary notes 29 to 37, which executes a second collation process for performing image collation with.
- the first matching process is a first matching process for detecting, as a degree of coincidence, a ratio of black pixels included in the photographed image in the number of black pixels in the matching source data.
- the second matching process Note 38 is the second matching process for detecting the ratio of the matching area excluding a portion where a difference is generated between the matching source data and the photographed image in the matching range of the matching source data and the photographed image as the degree of coincidence.
- a tablet packaging device according to claim 1.
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Medical Informatics (AREA)
- Primary Health Care (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Business, Economics & Management (AREA)
- Medicinal Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Mechanical Engineering (AREA)
- General Business, Economics & Management (AREA)
- Accounting & Taxation (AREA)
- General Physics & Mathematics (AREA)
- Physics & Mathematics (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Finance (AREA)
- Theoretical Computer Science (AREA)
- Strategic Management (AREA)
- Computer Security & Cryptography (AREA)
- Medical Preparation Storing Or Oral Administration Devices (AREA)
- Medical Treatment And Welfare Office Work (AREA)
Abstract
Description
以下、上述の各実施形態から抽出される発明の概要について付記する。なお、以下で説明する各構成及び各処理機能を取捨選択して任意に組み合わせることも可能である。
Claims (39)
- 錠剤分包装置において処方データに基づいて錠剤カセット及び手撒きユニットのいずれか一方又は両方から払い出された錠剤が包装材で分包される前に撮影された前記錠剤の撮影画像を分包単位で表示すると共に、前記錠剤の撮影画像に含まれる前記錠剤の識別情報と前記処方データとに基づいて実行される鑑査処理の結果を表示する鑑査表示処理部を備える鑑査支援システム。
- 前記鑑査表示処理部は、前記鑑査処理の結果がエラーである場合に、前記エラーの原因となった前記錠剤の撮影画像を識別可能に表示させる請求項1に記載の鑑査支援システム。
- 前記鑑査処理の結果がエラーである場合に、前記エラーの原因を前記エラーの原因となった前記錠剤の服用日及び服用時期と共にリスト表示するリスト表示処理部を更に備える請求項1又は2に記載の鑑査支援システム。
- 前記鑑査表示処理部は、列又は行の一方に服用日が表示され他方に服用時期が表示される配置形式で前記服用日及び前記服用時期の組み合わせ各々に対応する表示箇所に前記組み合わせに対応する分包単位の前記錠剤の撮影画像を前記服用時期の並び順に平行な方向に予め定められた表示順で並べて表示させる請求項1~3のいずれかに記載の鑑査支援システム。
- 前記鑑査表示処理部は、列又は行の一方に錠剤名が表示され他方に分包順番が表示される配置形式で前記錠剤名及び前記分包順番の組み合わせ各々に対応する前記錠剤の撮影画像を表示させる請求項1~3のいずれかに記載の鑑査支援システム。
- 前記鑑査表示処理部は、前記鑑査処理で用いられた前記錠剤の識別情報が含まれる元画像を少なくとも含み、前記錠剤分包装置において前記錠剤ごとに撮影された前記錠剤の異なる外周面に対応する複数の撮影画像を前記錠剤ごとに表示させる請求項1~5のいずれかに記載の鑑査支援システム。
- 前記鑑査表示処理部は、前記鑑査処理の結果がエラーである場合に、前記エラーの原因となった前記錠剤の撮影画像として、前記錠剤分包装置において前記錠剤ごとに撮影された前記錠剤の異なる外周面に対応する複数の撮影画像を前記錠剤ごとに表示可能である請求項1~6のいずれかに記載の鑑査支援システム。
- 前記エラーの原因となった前記錠剤の前記撮影画像の表示後に行われるユーザー操作に応じて前記錠剤についての前記エラーを解除可能な解除処理部を更に備える請求項7に記載の鑑査支援システム。
- 前記解除処理部は、前記エラーを解除すると共に、前記ユーザー操作が行われたときのユーザーの識別情報を前記処方データと対応付けて記録する請求項8に記載の鑑査支援システム。
- 前記鑑査表示処理部は、前記錠剤の正しい画像として予め登録された登録画像を前記錠剤の撮影画像と共に表示させる請求項1~9のいずれかに記載の鑑査支援システム。
- 前記鑑査表示処理部は、前記処方データに含まれる前記錠剤ごとについて前記鑑査処理の結果を個別に表示する請求項1~10のいずれかに記載の鑑査支援システム。
- 前記鑑査表示処理部は、前記錠剤の識別情報の文字の向きを揃えて前記錠剤の撮影画像を表示させる請求項1~11のいずれかに記載の鑑査支援システム。
- 前記鑑査支援システムが、前記処方データに基づいて実行される調剤処理で用いられる複数の調剤機器に接続されており、 前記調剤機器で調剤された薬剤の鑑査結果が表示される第1表示画面又は前記第1画面における予め定められた操作後に表示される第2表示画面に、前記調剤機器で実行された前記処方データに基づく調剤処理の一部又は全部を個別に再実行させるための操作部を表示させる操作表示処理部と、 前記操作表示処理部により表示される前記操作部に対するユーザー操作に応じて、前記調剤機器で実行された調剤処理の一部又は全部を前記調剤機器に再実行させる再実行処理部と、 を備える請求項1~12のいずれかに記載の鑑査支援システム。
- 前記操作表示処理部が、前記鑑査表示処理部により前記鑑査処理の結果が表示される前記第1表示画面に、前記錠剤分包装置で実行された分包処理の一部又は全部を再実行させるための前記操作部を表示させ、 前記再実行処理部が、前記操作表示処理部により表示される前記操作部に対するユーザー操作に応じて、前記錠剤分包装置で実行された分包処理の一部又は全部を前記錠剤分包装置に再実行させる請求項13に記載の鑑査支援システム。
- 前記操作表示処理部が、前記分包処理の全部を再実行させるための第1操作部と、前記分包処理のうち前記鑑査処理においてエラーと判定された一部の分包処理のみを再実行させるための第2操作部と、前記分包処理のうちユーザー操作により選択される一部の分包処理を再実行させるための第3操作部とのいずれか二つ又は三つを表示させる請求項14に記載の鑑査支援システム。
- 処方データに基づいて錠剤を包装材に収容する分包処理が実行可能であり、前記分包処理の適否を鑑査する予め定められた画像鑑査処理が実行可能な第1錠剤分包装置と、 前記分包処理が実行可能であり、前記画像鑑査処理が実行不能である又は前記画像鑑査処理の機能が無効に設定されている第2錠剤分包装置と、 前記処方データの内容及び前記分包処理の内容の少なくとも一方に基づいて、予め設定された第1優先条件を充足すると判断した場合に、前記処方データに基づく前記分包処理を前記第1錠剤分包装置に実行させる選択処理部と、
を備える鑑査支援システム。
-
前記第1錠剤分包装置が、
前記錠剤が前記包装材に収容される前に前記錠剤を撮影して前記錠剤の撮影画像を取得する撮影部と、
前記撮影部により撮影された前記錠剤の撮影画像から前記錠剤の識別情報を取得し、前記錠剤の識別情報と前記処方データとを照合する画像鑑査処理を実行する画像鑑査処理部と、
を備える請求項16に記載の鑑査支援システム。
-
前記撮影部が、前記錠剤の異なる外周面に対応する複数の撮影画像を前記錠剤ごとに撮影するものであり、
前記画像鑑査処理部が、複数の前記撮影画像のうち前記錠剤の識別情報の取得に用いられる前記撮影画像を前記撮影画像に含まれる錠剤の面積に応じて特定する請求項17に記載の鑑査支援システム。
- 前記第2錠剤分包装置が、
前記錠剤が前記包装材に収容される前又は収容された後で前記錠剤の数を検出する錠数検出部と、
前記錠数検出部によって検出された前記錠剤の数と前記処方データとを照合する計数鑑査処理を実行する計数鑑査処理部と、
を備える請求項16~18のいずれかに記載の鑑査支援システム。
-
前記第1錠剤分包装置及び前記第2錠剤分包装置各々が、前記分包処理で分包される前記錠剤を供給するために用いられる錠剤カセット及び手撒きユニットを備え、
前記第1優先条件が、前記分包処理で前記手撒きユニットが使用されること、前記分包処理で分包される前記錠剤の錠数が予め設定された閾値以上であること、又は前記分包処理で分包される前記錠剤が予め設定されたハイリスク薬であることの少なくとも一つを含む請求項16~19のいずれかに記載の鑑査支援システム。
-
前記選択処理部が、前記第1優先条件が充足しない場合は、予め設定された第2優先条件に従って前記第1錠剤分包装置又は前記第2錠剤分包装置を選択し、前記処方データに基づく前記分包処理を実行させるものであり、
前記第2優先条件が、前記処方データに含まれる前記錠剤のうち前記錠剤カセットに充填されている前記錠剤の種類が多い装置を選択すること、現在の前記分包処理の待機数が少ない装置を選択すること、又はユーザーにより任意に指定された装置を選択することのいずれかを含む請求項20に記載の鑑査支援システム。
-
前記選択処理部が、前記第1優先条件が充足しない場合は、予め設定された第2優先条件に従って前記第1錠剤分包装置又は前記第2錠剤分包装置を選択し、前記処方データに基づく前記分包処理を実行させる請求項16~20のいずれかに記載の鑑査支援システム。
-
錠剤分包装置において処方データに基づいて錠剤が包装材に収容される前に撮影された前記錠剤の撮影画像から前記錠剤の識別情報を取得し、前記錠剤の識別情報と前記処方データとを照合する画像鑑査処理を実行する画像鑑査処理部と、
前記錠剤の払出経路における前記錠剤の撮影位置の下流側で検出される前記錠剤の数と前記処方データとを照合する計数鑑査処理を実行する計数鑑査処理部と、
を備える鑑査支援システム。
-
錠剤分包装置において処方データに基づいて錠剤が包装材に収容される前に撮影された前記錠剤の撮影画像と前記処方データに含まれる前記錠剤の種類に対応して予め登録された正画像とを照合する画像鑑査処理を実行する画像鑑査処理部と、
前記画像鑑査処理部による前記画像鑑査処理において検出される前記錠剤の識別情報と前記正画像との差分を表示可能な鑑査表示処理部と、
を備える鑑査支援システム。
-
前記画像鑑査処理部が、前記錠剤の撮影画像と前記正画像との画像照合を行う第1照合処理を実行し、前記第1照合処理による照合結果が一致であり、前記画像鑑査処理の対象の前記錠剤に類似する類似薬が存在する場合には、前記撮影画像における前記類似薬と類似する予め設定された類似箇所に対応する領域と前記正画像における前記類似箇所に対応する領域との画像照合を行う第2照合処理を実行する請求項24に記載の錠剤分包装置。
-
処方データに基づいて錠剤を包装材に収容する分包処理が実行可能な錠剤分包装置であって、
前記錠剤を前記包装材に導く薬剤導入部と、 前記錠剤が前記包装材に収容される際に前記薬剤導入部、前記錠剤、及び前記包装材を含む撮影範囲を撮影する撮影部と、
を備える錠剤分包装置。
-
処方データに基づいて錠剤を包装材に収容する分包処理が実行可能な錠剤分包装置であって、
前記錠剤を回転させる錠剤回転部と、
前記錠剤回転部による回転中の前記錠剤を複数回撮影する撮影処理を実行する撮影処理部と、
前記撮影処理部により撮影される前記錠剤の複数の撮影画像のうち予め設定された条件を充足する撮影画像から前記錠剤の識別情報を取得し、前記錠剤の識別情報を前記錠剤の種類に対応する照合元データとして登録する登録処理部と、
を備える錠剤分包装置。
-
前記分包処理において撮影される前記錠剤の撮影画像から取得される前記錠剤の識別情報と前記処方データに含まれる前記錠剤の種類に対応する前記照合元データとを照合する画像鑑査処理を実行する画像鑑査処理部を更に備える請求項27に記載の錠剤分包装置。
-
前記錠剤の識別情報が、前記錠剤の撮影画像の一部又は全部の領域の画像であり、
前記照合元データが、前記錠剤の種類に対応して予め登録された正画像であり、
前記画像鑑査処理部が、前記錠剤の識別情報と前記正画像とを照合する請求項28に記載の錠剤分包装置。
-
前記錠剤回転部による回転中の前記錠剤が撮影可能であり、前記錠剤の識別情報が刻印である場合に対応して設けられる第1撮影ユニット及び前記錠剤の識別情報が印刷である場合に対応して設けられる第2撮影ユニットを含む撮影部と、
前記登録処理部により登録される前記錠剤の識別情報が刻印及び印刷のいずれであるかをユーザー操作に応じて特定する特定処理部と、
を備え、
前記画像鑑査処理部が、前記特定処理部によって特定された前記刻印又は前記印刷のいずれかに対応する前記第1撮影ユニット又は前記第2撮影ユニットを用いて撮影された前記錠剤の撮影画像を用いて前記画像鑑査処理を実行する請求項29に記載の錠剤分包装置。
-
前記錠剤回転部による回転中の前記錠剤が撮影可能であり、前記錠剤の識別情報が刻印である場合に対応して設けられる第1撮影ユニット及び前記錠剤の識別情報が印刷である場合に対応して設けられる第2撮影ユニットを含む撮影部と、
前記登録処理部により登録される前記錠剤の識別情報が刻印及び印刷のいずれであるかをユーザー操作に応じて特定する特定処理部と、
を備え、
前記撮影処理部が、前記登録処理部により前記照合元データが登録される際に用いられる前記撮影画像を撮影する際に、前記特定処理部による特定結果に応じて前記第1撮影ユニット及び前記第2撮影ユニットのいずれかを選択して前記錠剤を撮影する請求項29に記載の錠剤分包装置。
-
前記錠剤回転部による回転中の前記錠剤が撮影可能である撮影部と、
前記錠剤に振動を与える加振部と、
前記撮影部により撮影される前記錠剤の撮影画像各々に基づいて、前記撮影部に対して前記錠剤の側面が正対する状態で前記錠剤が回転しているか否かを判定する状態判定処理部と、 前記状態判定処理部によって前記状態で前記錠剤が回転していると判断された場合に前記加振部により前記錠剤に振動を与える加振処理部と、
を更に備える請求項29に記載の錠剤分包装置。
-
前記登録処理部が、前記撮影処理部により撮影される前記錠剤の複数の撮影画像のうちユーザー操作により選択された撮影画像を前記照合元データとして登録する請求項28~32のいずれかに記載の錠剤分包装置。
-
前記登録処理部が、前記照合元データとして登録される前記撮影画像における上方向の位置を指定するユーザー操作を受け付けて、前記撮影画像における前記上方向の位置を示す情報を前記照合元データの一部として登録可能である請求項29~33のいずれかに記載の錠剤分包装置。
-
前記登録処理部が、少なくとも前記錠剤と前記錠剤に対応する初期正画像との対応関係が予め記憶された薬品データベースに記憶されている前記初期正画像と前記撮影画像との照合結果に応じて、前記撮影画像における前記上方向の位置を示す情報を前記照合元データの一部として登録可能である請求項29~34のいずれかに記載の錠剤分包装置。
-
前記登録処理部が、前記照合元データとして登録される前記撮影画像における上方向の位置を指定するユーザー操作を受け付けて、前記撮影画像における前記上方向の位置が上部になるように前記撮影画像を回転させた正画像を前記照合元データとして登録可能である請求項29~35のいずれかに記載の錠剤分包装置。
-
前記登録処理部が、少なくとも前記錠剤と前記錠剤に対応する初期正画像との対応関係が予め記憶された薬品データベースに記憶されている前記初期正画像と前記撮影画像との照合結果に応じて、前記撮影画像における前記上方向の位置が上部になるように前記撮影画像を回転させた正画像を前記照合元データとして登録可能である請求項29~36のいずれかに記載の錠剤分包装置。
-
前記画像鑑査処理部が、前記撮影画像と前記照合元データとの画像照合を行う第1照合処理を実行し、前記第1照合処理による照合結果が一致であり、前記画像鑑査処理の対象の前記錠剤に類似する類似薬が存在する場合には、前記撮影画像における前記類似薬と類似する予め設定された類似箇所に対応する領域と前記照合元データにおける前記類似箇所に対応する領域との画像照合を行う第2照合処理を実行する請求項29~37のいずれかに記載の錠剤分包装置。
-
前記第1照合処理が、前記照合元データにおける黒画素の数のうち前記撮影画像に含まれる黒画素の割合を一致度として検出する第1マッチング処理であり、
前記第2照合処理が、前記照合元データ及び前記撮影画像の照合範囲において前記照合元データと前記撮影画像とに差分が生じている箇所を除く一致領域の割合を一致度として検出する第2マッチング処理である請求項38に記載の錠剤分包装置。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201580051768.4A CN107072881B (zh) | 2014-09-25 | 2015-09-17 | 检查辅助系统、片剂分包装置 |
| US15/514,198 US20170305589A1 (en) | 2014-09-25 | 2015-09-17 | Inspection assistance system and tablet packaging device |
| EP15843749.1A EP3199139A4 (en) | 2014-09-25 | 2015-09-17 | Inspection assistance system and tablet packaging device |
| KR1020177008206A KR102382736B1 (ko) | 2014-09-25 | 2015-09-17 | 감사 지원 시스템, 정제 분포 장치 |
| JP2016550159A JP6705379B2 (ja) | 2014-09-25 | 2015-09-17 | 鑑査支援システム |
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-195760 | 2014-09-25 | ||
| JP2014195760 | 2014-09-25 | ||
| JP2014209496 | 2014-10-10 | ||
| JP2014-209496 | 2014-10-10 | ||
| JP2015-109232 | 2015-05-28 | ||
| JP2015109232 | 2015-05-28 | ||
| JP2015-133109 | 2015-07-01 | ||
| JP2015133109 | 2015-07-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016047569A1 true WO2016047569A1 (ja) | 2016-03-31 |
Family
ID=55581096
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/076587 WO2016047569A1 (ja) | 2014-09-25 | 2015-09-17 | 鑑査支援システム、錠剤分包装置 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20170305589A1 (ja) |
| EP (1) | EP3199139A4 (ja) |
| JP (7) | JP6705379B2 (ja) |
| KR (1) | KR102382736B1 (ja) |
| CN (1) | CN107072881B (ja) |
| TW (1) | TWI660725B (ja) |
| WO (1) | WO2016047569A1 (ja) |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2017196260A (ja) * | 2016-04-28 | 2017-11-02 | 株式会社湯山製作所 | 調剤制御システム及び調剤制御方法 |
| WO2017217366A1 (ja) * | 2016-06-17 | 2017-12-21 | 株式会社湯山製作所 | 鑑査支援システム、薬品払出装置 |
| WO2018025852A1 (ja) * | 2016-08-05 | 2018-02-08 | 株式会社湯山製作所 | 薬剤分包装置 |
| EP3299997A3 (en) * | 2016-09-27 | 2018-07-11 | RxSafe, LLC | Verification system for a pharmacy packaging system |
| JPWO2017119276A1 (ja) * | 2016-01-06 | 2018-11-08 | 株式会社湯山製作所 | 鑑査支援システム、錠剤分包装置、分包制御プログラム |
| WO2018221065A1 (ja) * | 2017-05-30 | 2018-12-06 | 富士フイルム株式会社 | 薬剤検査支援装置及び薬剤検査支援方法 |
| WO2019039015A1 (ja) * | 2017-08-25 | 2019-02-28 | 富士フイルム株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| WO2019039016A1 (ja) * | 2017-08-25 | 2019-02-28 | 富士フイルム株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| CN110603567A (zh) * | 2017-05-30 | 2019-12-20 | 富士胶片富山化学株式会社 | 配药监查辅助装置以及配药监查辅助方法 |
| WO2019244729A1 (ja) * | 2018-06-19 | 2019-12-26 | 株式会社湯山製作所 | 薬剤仕分装置 |
| JP2020121144A (ja) * | 2020-04-03 | 2020-08-13 | 株式会社湯山製作所 | 薬剤分包システム |
| JP2020175245A (ja) * | 2017-04-14 | 2020-10-29 | 株式会社湯山製作所 | 薬剤仕分装置 |
| WO2020246534A1 (ja) | 2019-06-07 | 2020-12-10 | 株式会社湯山製作所 | 薬剤鑑査支援装置、薬剤鑑査支援システム及び薬剤鑑査方法 |
| JPWO2021010276A1 (ja) * | 2019-07-17 | 2021-01-21 | ||
| WO2021145266A1 (ja) | 2020-01-17 | 2021-07-22 | 富士フイルム富山化学株式会社 | 画像処理装置及び方法 |
| JP2022084581A (ja) * | 2016-07-29 | 2022-06-07 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | 電子目視検査が組み込まれた組み立てライン |
| US11595595B2 (en) | 2016-09-27 | 2023-02-28 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| WO2023120366A1 (ja) * | 2021-12-24 | 2023-06-29 | 大倉工業株式会社 | 一包化鑑査装置、一包化鑑査プログラムおよび一包化鑑査方法 |
| JP7426449B1 (ja) | 2022-09-21 | 2024-02-01 | 三菱電機Itソリューションズ株式会社 | 服薬支援システム、服薬支援方法、および服薬支援プログラム |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10427809B2 (en) | 2012-06-01 | 2019-10-01 | Rxsafe Llc | Pharmacy packaging system |
| US10427810B2 (en) | 2012-06-01 | 2019-10-01 | Rxsafe Llc | Pharmacy packaging system |
| US20170305589A1 (en) * | 2014-09-25 | 2017-10-26 | Yuyama Mfg. Co., Ltd. | Inspection assistance system and tablet packaging device |
| JP6761622B2 (ja) | 2015-07-02 | 2020-09-30 | 株式会社タカゾノテクノロジー | 薬剤包装装置および薬剤供給装置 |
| JP6333871B2 (ja) * | 2016-02-25 | 2018-05-30 | ファナック株式会社 | 入力画像から検出した対象物を表示する画像処理装置 |
| JP6861825B2 (ja) * | 2017-08-22 | 2021-04-21 | 富士フイルム富山化学株式会社 | 薬剤識別装置、画像処理装置、画像処理方法及びプログラム |
| EP3678089A4 (en) * | 2017-08-31 | 2020-09-09 | FUJIFILM Toyama Chemical Co., Ltd. | DRUG INSPECTION DEVICE, IMAGE PROCESSING DEVICE, IMAGE PROCESSING EXPERIENCE AND PROGRAM |
| MX2020003804A (es) | 2017-10-13 | 2020-08-13 | Rxsafe Llc | Mecanismo de alimentacion universal para empacadora automatica. |
| JP7376786B2 (ja) * | 2017-11-08 | 2023-11-09 | 株式会社湯山製作所 | 薬剤カセット取扱装置、及び薬剤払出装置 |
| WO2019170685A1 (en) * | 2018-03-06 | 2019-09-12 | Blisterpartner B.V. | System and method for medication preparation and verification |
| TWI695347B (zh) * | 2018-07-30 | 2020-06-01 | 台灣基督長老教會馬偕醫療財團法人馬偕紀念醫院 | 經由藥物包裝及/或標籤以分類及辨識該藥物的方法及系統 |
| WO2020050109A1 (ja) * | 2018-09-03 | 2020-03-12 | 株式会社日立ハイテクノロジーズ | 表示装置、情報端末、個人情報の保護方法、およびプログラムとそれを記録する記録媒体 |
| CN109044845B (zh) * | 2018-09-29 | 2021-11-16 | 河北盛世天昕电子科技有限公司 | 一种配药余量告警方法及装置 |
| JP2020098420A (ja) * | 2018-12-17 | 2020-06-25 | ソニー株式会社 | 画像処理装置、画像処理方法及びプログラム |
| CN113301883A (zh) * | 2019-02-08 | 2021-08-24 | 富士胶片富山化学株式会社 | 药剂识别系统、药剂识别装置、药剂识别方法以及程序 |
| EP3962835A4 (en) * | 2019-05-03 | 2023-10-18 | Rxsafe Llc | PHARMACY PACKAGING SYSTEM AND POUCH |
| IT201900012216A1 (it) * | 2019-07-18 | 2021-01-18 | Bliss Srl | Sistema per il controllo di farmaci di tipo perfezionato |
| US11189372B2 (en) * | 2019-09-19 | 2021-11-30 | Rx-V Inc. | Verification system for medication packaging and method |
| KR20220126771A (ko) * | 2020-02-21 | 2022-09-16 | 어플라이드 머티어리얼스, 인코포레이티드 | 제품들의 검사를 위한 방법 및 시스템 |
| EP3901960B1 (en) * | 2020-04-21 | 2023-09-06 | Deutsche Post AG | Validation method and validation apparatus for sealed unit |
| WO2022231959A1 (en) * | 2021-04-26 | 2022-11-03 | Ackley Machine Corporation | Conveyer apparatus for pellet-shaped articles |
| JP2023080743A (ja) * | 2021-11-30 | 2023-06-09 | NeoX株式会社 | 薬剤数管理システム |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006155599A (ja) * | 2004-10-28 | 2006-06-15 | Yuyama Manufacturing Co Ltd | 調剤支援装置及び調剤支援方法 |
| JP2011104077A (ja) * | 2009-11-17 | 2011-06-02 | Yuyama Manufacturing Co Ltd | 薬剤払出装置 |
| WO2012005004A1 (ja) * | 2010-07-09 | 2012-01-12 | パナソニック株式会社 | 錠剤監査支援方法及び錠剤監査支援装置 |
| JP2014067342A (ja) * | 2012-09-27 | 2014-04-17 | Fujifilm Corp | 薬剤検査支援装置及び方法 |
Family Cites Families (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6535637B1 (en) | 1997-04-04 | 2003-03-18 | Esco Electronics, Inc. | Pharmaceutical pill recognition and verification system |
| US7006893B2 (en) * | 1999-09-22 | 2006-02-28 | Telepharmacy Solutions, Inc. | Systems for dispensing medical products |
| US7040077B2 (en) * | 2001-09-28 | 2006-05-09 | Yuyama Mfg. Co. Ltd. | Medicine packing apparatus |
| JP2005209132A (ja) | 2004-01-26 | 2005-08-04 | Konishi Seisakusho:Kk | 薬品監査装置 |
| JP4429752B2 (ja) * | 2004-01-30 | 2010-03-10 | 株式会社湯山製作所 | 錠剤収納取出装置 |
| CN101076311B (zh) * | 2004-10-28 | 2011-05-04 | 株式会社汤山制作所 | 配药辅助装置及配药辅助方法 |
| DK1889197T3 (da) * | 2005-05-16 | 2012-02-13 | Intelligent Hospital Systems Ltd | Automatisk farmaceutisk blandesystem |
| JP5044906B2 (ja) * | 2005-08-25 | 2012-10-10 | 株式会社湯山製作所 | 薬剤払出装置 |
| CA2978267A1 (en) * | 2007-08-23 | 2009-02-23 | Dfm, Llc | Translumenally implantable heart valve with formed in place support |
| US8554579B2 (en) * | 2008-10-13 | 2013-10-08 | Fht, Inc. | Management, reporting and benchmarking of medication preparation |
| JP5163985B2 (ja) * | 2008-11-14 | 2013-03-13 | 学校法人早稲田大学 | 粒状物品種検査装置 |
| EP2497715B1 (en) * | 2009-11-02 | 2016-04-27 | Yuyama Mfg. Co., Ltd. | Medicine packing device and method for packaging medicine |
| KR101172242B1 (ko) * | 2010-06-09 | 2012-08-07 | 가부시키가이샤 유야마 세이사쿠쇼 | 약제 분포 장치 |
| JP4699563B1 (ja) * | 2010-10-20 | 2011-06-15 | オオクマ電子株式会社 | 錠剤検査装置 |
| US8943779B2 (en) | 2011-04-28 | 2015-02-03 | Yuyama Mfg. Co., Ltd. | Medicine inspection device, and medicine packaging device |
| KR20130002696A (ko) * | 2011-06-29 | 2013-01-08 | (주)제이브이엠 | 불량약품 검사장치 |
| US20130170732A1 (en) * | 2011-08-08 | 2013-07-04 | Makoto Gotou | Medicine pack inspecting system |
| WO2013105198A1 (ja) * | 2012-01-11 | 2013-07-18 | パナソニックヘルスケア株式会社 | 薬剤供給装置、並びに薬剤供給装置における薬剤鑑査方法 |
| KR20140102007A (ko) * | 2013-02-13 | 2014-08-21 | (주)제이브이엠 | 약제 불출 시스템 및 그 제어방법 |
| JP2014053791A (ja) * | 2012-09-07 | 2014-03-20 | Panasonic Corp | 画像処理装置、画像処理プログラムおよび画像撮影装置 |
| US20140156298A1 (en) * | 2012-12-05 | 2014-06-05 | Meadwestvaco Corporation | Modularization for prescription fulfillment and adherence |
| US9311451B2 (en) * | 2013-02-18 | 2016-04-12 | Lck Co., Ltd. | Medication dispensing apparatus for preventing medication dispensing error |
| DE102013014919A1 (de) * | 2013-07-15 | 2015-01-15 | Ewald Dörken Ag | Bikomponentenfaser zur Herstellung von Spinnvliesen |
| JP2016527060A (ja) * | 2013-08-09 | 2016-09-08 | パーセプティメッド インコーポレイテッドPerceptimed, Inc. | 遠隔医薬検証 |
| JP5457599B1 (ja) | 2013-09-12 | 2014-04-02 | 株式会社Windy | 薬剤分包システム |
| JP6100136B2 (ja) * | 2013-09-30 | 2017-03-22 | 富士フイルム株式会社 | 薬剤認識装置及び方法 |
| JP6167053B2 (ja) * | 2014-02-28 | 2017-07-19 | 富士フイルム株式会社 | 検査装置及び検査方法、並びに検査方法をコンピュータに実行させるプログラム |
| US20170305589A1 (en) * | 2014-09-25 | 2017-10-26 | Yuyama Mfg. Co., Ltd. | Inspection assistance system and tablet packaging device |
| JP6908673B2 (ja) * | 2018-05-22 | 2021-07-28 | 日本金銭機械株式会社 | 自動紙幣取扱いシステムおよび自動紙幣取扱い方法 |
| JP7115598B1 (ja) * | 2021-06-30 | 2022-08-09 | 株式会社デンソーウェーブ | 搬送ロボットシステム |
| JP7126297B1 (ja) * | 2021-07-06 | 2022-08-26 | 竹本油脂株式会社 | 合成繊維用処理剤及び合成繊維 |
-
2015
- 2015-09-17 US US15/514,198 patent/US20170305589A1/en not_active Abandoned
- 2015-09-17 KR KR1020177008206A patent/KR102382736B1/ko active IP Right Grant
- 2015-09-17 EP EP15843749.1A patent/EP3199139A4/en not_active Withdrawn
- 2015-09-17 JP JP2016550159A patent/JP6705379B2/ja active Active
- 2015-09-17 CN CN201580051768.4A patent/CN107072881B/zh active Active
- 2015-09-17 TW TW104130807A patent/TWI660725B/zh active
- 2015-09-17 WO PCT/JP2015/076587 patent/WO2016047569A1/ja active Application Filing
-
2020
- 2020-02-12 JP JP2020021508A patent/JP6825732B2/ja active Active
-
2021
- 2021-01-13 JP JP2021003777A patent/JP7010395B2/ja active Active
-
2022
- 2022-01-12 JP JP2022003255A patent/JP7441445B2/ja active Active
-
2023
- 2023-01-24 JP JP2023009038A patent/JP7445177B2/ja active Active
- 2023-01-24 JP JP2023009037A patent/JP2023038320A/ja active Pending
-
2024
- 2024-02-21 JP JP2024024724A patent/JP2024050995A/ja active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006155599A (ja) * | 2004-10-28 | 2006-06-15 | Yuyama Manufacturing Co Ltd | 調剤支援装置及び調剤支援方法 |
| JP2011104077A (ja) * | 2009-11-17 | 2011-06-02 | Yuyama Manufacturing Co Ltd | 薬剤払出装置 |
| WO2012005004A1 (ja) * | 2010-07-09 | 2012-01-12 | パナソニック株式会社 | 錠剤監査支援方法及び錠剤監査支援装置 |
| JP2014067342A (ja) * | 2012-09-27 | 2014-04-17 | Fujifilm Corp | 薬剤検査支援装置及び方法 |
Cited By (60)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2017119276A1 (ja) * | 2016-01-06 | 2018-11-08 | 株式会社湯山製作所 | 鑑査支援システム、錠剤分包装置、分包制御プログラム |
| JP2017196260A (ja) * | 2016-04-28 | 2017-11-02 | 株式会社湯山製作所 | 調剤制御システム及び調剤制御方法 |
| KR20190019079A (ko) * | 2016-06-17 | 2019-02-26 | 가부시키가이샤 유야마 세이사쿠쇼 | 감사 지원 시스템, 약품 불출 장치 |
| WO2017217366A1 (ja) * | 2016-06-17 | 2017-12-21 | 株式会社湯山製作所 | 鑑査支援システム、薬品払出装置 |
| KR102365329B1 (ko) * | 2016-06-17 | 2022-02-22 | 가부시키가이샤 유야마 세이사쿠쇼 | 감사 지원 시스템, 약품 불출 장치 |
| JPWO2017217366A1 (ja) * | 2016-06-17 | 2019-04-11 | 株式会社湯山製作所 | 鑑査支援システム、薬品払出装置 |
| EP3473232A4 (en) * | 2016-06-17 | 2020-04-29 | Yuyama Mfg. Co., Ltd. | INSPECTION SUPPORT SYSTEM AND DRUG DISPENSER |
| CN109195568A (zh) * | 2016-06-17 | 2019-01-11 | 株式会社汤山制作所 | 检验辅助系统和药品分发装置 |
| US11823371B2 (en) | 2016-07-29 | 2023-11-21 | Regeneron Pharmaceuticals, Inc. | Assembly line with integrated electronic visual inspection |
| JP7288105B2 (ja) | 2016-07-29 | 2023-06-06 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | 電子目視検査が組み込まれた組み立てライン |
| JP2022084581A (ja) * | 2016-07-29 | 2022-06-07 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | 電子目視検査が組み込まれた組み立てライン |
| JP7479545B2 (ja) | 2016-07-29 | 2024-05-08 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | 電子目視検査が組み込まれた組み立てライン |
| CN109195567A (zh) * | 2016-08-05 | 2019-01-11 | 株式会社汤山制作所 | 药剂分包装置 |
| KR102387007B1 (ko) * | 2016-08-05 | 2022-04-18 | 가부시키가이샤 유야마 세이사쿠쇼 | 약제 분할 포장 장치 |
| KR20190039068A (ko) * | 2016-08-05 | 2019-04-10 | 가부시키가이샤 유야마 세이사쿠쇼 | 약제 분할 포장 장치 |
| JPWO2018025852A1 (ja) * | 2016-08-05 | 2019-06-06 | 株式会社湯山製作所 | 薬剤分包装置 |
| WO2018025852A1 (ja) * | 2016-08-05 | 2018-02-08 | 株式会社湯山製作所 | 薬剤分包装置 |
| TWI725218B (zh) * | 2016-08-05 | 2021-04-21 | 日商湯山製作所股份有限公司 | 藥劑分包裝置 |
| CN109195567B (zh) * | 2016-08-05 | 2021-04-06 | 株式会社汤山制作所 | 药剂分包装置 |
| EP3494952A4 (en) * | 2016-08-05 | 2020-01-15 | Yuyama Mfg. Co., Ltd. | DEVICE FOR PACKAGING MEDICATION PACKS |
| US10881581B2 (en) | 2016-08-05 | 2021-01-05 | Yuyama Mfg. Co., Ltd. | Medication sachet packaging device |
| AU2017221799C1 (en) * | 2016-09-27 | 2022-06-30 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| US11039091B2 (en) | 2016-09-27 | 2021-06-15 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| US11595595B2 (en) | 2016-09-27 | 2023-02-28 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| US10187593B2 (en) | 2016-09-27 | 2019-01-22 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| EP3299997A3 (en) * | 2016-09-27 | 2018-07-11 | RxSafe, LLC | Verification system for a pharmacy packaging system |
| AU2017221799B2 (en) * | 2016-09-27 | 2022-03-17 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| US10594956B2 (en) | 2016-09-27 | 2020-03-17 | Rxsafe Llc | Verification system for a pharmacy packaging system |
| JP2020175245A (ja) * | 2017-04-14 | 2020-10-29 | 株式会社湯山製作所 | 薬剤仕分装置 |
| US11504747B2 (en) | 2017-04-14 | 2022-11-22 | Yuyama Mfg. Co., Ltd. | Drug sorting device, sorting container, and drug return method |
| US11404153B2 (en) | 2017-05-30 | 2022-08-02 | Fujifilm Toyama Chemical Co., Ltd. | Drug inspection assisting apparatus and drug inspection assisting method |
| EP3633609A4 (en) * | 2017-05-30 | 2020-04-08 | FUJIFILM Toyama Chemical Co., Ltd. | AUXILIARY DEVICE ASSESSMENT DEVICE AND AUXILIARY ASSESSMENT METHOD |
| US11594322B2 (en) | 2017-05-30 | 2023-02-28 | Fujifilm Toyama Chemical Co., Ltd. | Dispensing audit support apparatus and dispensing audit support method |
| CN110603567A (zh) * | 2017-05-30 | 2019-12-20 | 富士胶片富山化学株式会社 | 配药监查辅助装置以及配药监查辅助方法 |
| WO2018221065A1 (ja) * | 2017-05-30 | 2018-12-06 | 富士フイルム株式会社 | 薬剤検査支援装置及び薬剤検査支援方法 |
| CN110612090B (zh) * | 2017-05-30 | 2021-08-20 | 富士胶片富山化学株式会社 | 药剂检查辅助装置以及药剂检查辅助方法 |
| CN110612090A (zh) * | 2017-05-30 | 2019-12-24 | 富士胶片富山化学株式会社 | 药剂检查辅助装置以及药剂检查辅助方法 |
| JPWO2018221065A1 (ja) * | 2017-05-30 | 2020-03-19 | 富士フイルム富山化学株式会社 | 薬剤検査支援装置及び薬剤検査支援方法 |
| JPWO2019039015A1 (ja) * | 2017-08-25 | 2020-09-24 | 富士フイルム富山化学株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| JPWO2019039302A1 (ja) * | 2017-08-25 | 2020-08-27 | 富士フイルム富山化学株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| WO2019039302A1 (ja) * | 2017-08-25 | 2019-02-28 | 富士フイルム株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| WO2019039016A1 (ja) * | 2017-08-25 | 2019-02-28 | 富士フイルム株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| WO2019039015A1 (ja) * | 2017-08-25 | 2019-02-28 | 富士フイルム株式会社 | 薬剤検査支援装置、画像処理装置、画像処理方法及びプログラム |
| US11423246B2 (en) | 2017-08-25 | 2022-08-23 | Fujifilm Toyama Chemical Co., Ltd. | Medicine inspection assistance device, image processing device, image processing method, and program |
| WO2019244729A1 (ja) * | 2018-06-19 | 2019-12-26 | 株式会社湯山製作所 | 薬剤仕分装置 |
| JP7264105B2 (ja) | 2018-06-19 | 2023-04-25 | 株式会社湯山製作所 | 薬剤仕分装置 |
| JP7469729B2 (ja) | 2018-06-19 | 2024-04-17 | 株式会社湯山製作所 | 薬剤仕分装置、及び、薬剤仕分装置の制御方法 |
| JP2020110732A (ja) * | 2018-06-19 | 2020-07-27 | 株式会社湯山製作所 | 薬剤仕分装置 |
| JP2020096819A (ja) * | 2018-06-19 | 2020-06-25 | 株式会社湯山製作所 | 薬剤仕分装置 |
| JPWO2019244729A1 (ja) * | 2018-06-19 | 2020-06-25 | 株式会社湯山製作所 | 薬剤仕分装置 |
| WO2020246534A1 (ja) | 2019-06-07 | 2020-12-10 | 株式会社湯山製作所 | 薬剤鑑査支援装置、薬剤鑑査支援システム及び薬剤鑑査方法 |
| KR20220017390A (ko) | 2019-06-07 | 2022-02-11 | 가부시키가이샤 유야마 세이사쿠쇼 | 약제 감사 지원 장치, 약제 감사 지원 시스템 및 약제 감사 방법 |
| JP7343585B2 (ja) | 2019-07-17 | 2023-09-12 | 富士フイルム富山化学株式会社 | 識別支援システム、識別支援クライアント、識別支援サーバ、及び識別支援方法 |
| WO2021010276A1 (ja) * | 2019-07-17 | 2021-01-21 | 富士フイルム富山化学株式会社 | 識別支援システム、識別支援クライアント、識別支援サーバ、及び識別支援方法 |
| JPWO2021010276A1 (ja) * | 2019-07-17 | 2021-01-21 | ||
| WO2021145266A1 (ja) | 2020-01-17 | 2021-07-22 | 富士フイルム富山化学株式会社 | 画像処理装置及び方法 |
| JP2020121144A (ja) * | 2020-04-03 | 2020-08-13 | 株式会社湯山製作所 | 薬剤分包システム |
| WO2023120366A1 (ja) * | 2021-12-24 | 2023-06-29 | 大倉工業株式会社 | 一包化鑑査装置、一包化鑑査プログラムおよび一包化鑑査方法 |
| JP7310045B1 (ja) | 2021-12-24 | 2023-07-18 | 大倉工業株式会社 | 一包化鑑査装置、一包化鑑査プログラムおよび一包化鑑査方法 |
| JP7426449B1 (ja) | 2022-09-21 | 2024-02-01 | 三菱電機Itソリューションズ株式会社 | 服薬支援システム、服薬支援方法、および服薬支援プログラム |
Also Published As
| Publication number | Publication date |
|---|---|
| TWI660725B (zh) | 2019-06-01 |
| EP3199139A4 (en) | 2018-05-30 |
| JP7445177B2 (ja) | 2024-03-07 |
| JP6705379B2 (ja) | 2020-06-03 |
| US20170305589A1 (en) | 2017-10-26 |
| JP7010395B2 (ja) | 2022-01-26 |
| JP2021053511A (ja) | 2021-04-08 |
| JP7441445B2 (ja) | 2024-03-01 |
| JP2020075172A (ja) | 2020-05-21 |
| KR20170082503A (ko) | 2017-07-14 |
| CN107072881A (zh) | 2017-08-18 |
| TW201625195A (zh) | 2016-07-16 |
| JP2022036241A (ja) | 2022-03-04 |
| JP2024050995A (ja) | 2024-04-10 |
| JP2023038320A (ja) | 2023-03-16 |
| EP3199139A1 (en) | 2017-08-02 |
| JP2023038321A (ja) | 2023-03-16 |
| CN107072881B (zh) | 2020-07-03 |
| JPWO2016047569A1 (ja) | 2017-07-13 |
| KR102382736B1 (ko) | 2022-04-06 |
| JP6825732B2 (ja) | 2021-02-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7445177B2 (ja) | 画像処理装置、プログラム | |
| JP7401813B2 (ja) | 支援システム、プログラム | |
| JP5916032B2 (ja) | 薬剤検査支援装置及び方法 | |
| TWI746577B (zh) | 檢查輔助系統 | |
| TWI679010B (zh) | 藥劑檢查支援裝置及藥劑檢查支援方法 | |
| JP7156304B2 (ja) | 調剤支援システム | |
| JP6766546B2 (ja) | 薬品払出装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15843749 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016550159 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20177008206 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15514198 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015843749 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2015843749 Country of ref document: EP |