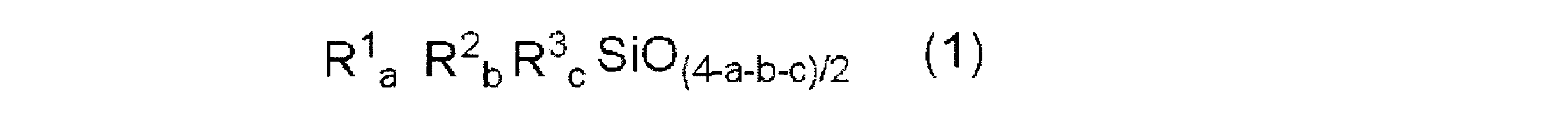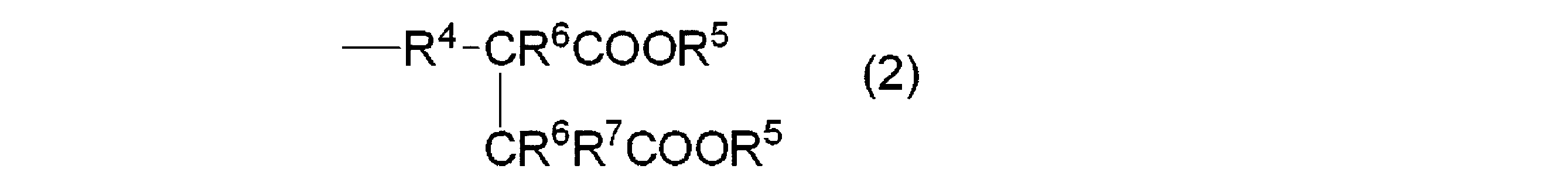WO2015159773A1 - 化粧料 - Google Patents
化粧料 Download PDFInfo
- Publication number
- WO2015159773A1 WO2015159773A1 PCT/JP2015/060963 JP2015060963W WO2015159773A1 WO 2015159773 A1 WO2015159773 A1 WO 2015159773A1 JP 2015060963 W JP2015060963 W JP 2015060963W WO 2015159773 A1 WO2015159773 A1 WO 2015159773A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cosmetic
- acid
- cosmetic according
- carboxylic acid
- modified silicone
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/891—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0241—Containing particulates characterized by their shape and/or structure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
- A61K8/29—Titanium; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/342—Alcohols having more than seven atoms in an unbroken chain
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/361—Carboxylic acids having more than seven carbon atoms in an unbroken chain; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4973—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom
- A61K8/498—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom having 6-membered rings or their condensed derivatives, e.g. coumarin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/58—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing atoms other than carbon, hydrogen, halogen, oxygen, nitrogen, sulfur or phosphorus
- A61K8/585—Organosilicon compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
- A61Q1/02—Preparations containing skin colorants, e.g. pigments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/60—Particulates further characterized by their structure or composition
- A61K2800/61—Surface treated
- A61K2800/614—By macromolecular compounds
Definitions
- the present invention relates to a cosmetic comprising an aqueous phase and having hydrophobic powder dispersed in the aqueous phase.
- Cosmetics containing an aqueous phase as a continuous phase give a fresh and refreshing feeling.
- basic cosmetics such as emulsions, base cosmetics, sunscreen agents, foundations, eye shadows, and other makeup cosmetics. As widely used.
- Non-Patent Document 1 describes that carboxyl-modified silicone neutralized with triethanolamine has excellent emulsifying power, but does not describe the function as a dispersant for dispersing hydrophobic powder in an aqueous phase. Not.
- the present invention provides a cosmetic in which a hydrophobic powder is well dispersed in an aqueous phase, provides a refreshing feeling during use, and can form a cosmetic film excellent in water resistance on the skin.
- the purpose is to do.
- the object of the present invention is to: (A) hydrophobic powder, (B) carboxylic acid-modified silicone, (C) a basic compound, and (D) It is achieved by a cosmetic comprising water, wherein the (A) hydrophobic powder is dispersed in an aqueous phase.
- the cosmetic of the present invention preferably contains (A) the hydrophobic powder in the range of 1 to 40% by mass.
- mass% is synonymous with “wt%”, and the standard is the total mass (total weight) of the cosmetic of the present invention unless otherwise specified.
- the cosmetic of the present invention preferably contains the carboxylic acid-modified silicone (B) in the range of 0.01 to 20 parts by mass with respect to 10 parts by mass of the hydrophobic powder.
- the (B) carboxylic acid-modified silicone has the following structural formula (A): ⁇ Where Rc is represented by the general formula: -R 1- (OR 2 ) p- (O) wR 3 -COOH (R 1 represents a linear or branched alkylene group having 2 to 22 carbon atoms, and R 2 represents a carbon number of 2 Represents a linear or branched alkylene group of 4 to 4, R 3 represents a bond (-) or a linear or branched alkylene group of 1 to 22 carbon atoms, p represents a number of 0 to 200, w represents a number of 0 or 1, and represents a carboxyl group-containing organic group, R is the same or different and represents an alkyl or alkoxy group having 1 to 22 carbon atoms or a phenyl group, R ′ is Rc or R; Each of a and b is a number in the range of 0 or more, and a + b is a number in the range of 0 to
- the cosmetic of the present invention preferably contains (D) water in the range of 20 to 95% by mass.
- the pH of the cosmetic of the present invention is preferably 7.1 to 9.5.
- the cosmetic of the present invention may further contain (G) a polyhydric alcohol.
- the cosmetic of the present invention preferably further comprises (E) an oil.
- the cosmetic of the present invention preferably contains the oil (E) in the range of 3 to 60% by mass.
- the cosmetic of the present invention preferably further comprises (F) a salt of a higher fatty acid.
- the cosmetic of the present invention preferably contains the salt of the higher fatty acid (F) in the range of 0.1 to 12% by mass.
- the higher fatty acid is preferably at least one selected from the group consisting of isostearic acid, hexyldecanoic acid and oleic acid.
- the cosmetic of the present invention may further contain (H) a water-soluble thickener.
- the cosmetic of the present invention preferably contains no surfactant other than the (B) carboxylic acid-modified silicone. Moreover, when the cosmetics of this invention contain the salt of the said (F) higher fatty acid, it is preferable not to contain surfactant other than the said (B) carboxylic acid modified silicone and the said (F) salt of a higher fatty acid.
- the cosmetic of the present invention is preferably in the form of an oil-in-water emulsion composition.
- the present invention provides (A) a hydrophobic powder, (B) a carboxylic acid-modified silicone, (C) a basic compound and (D) water in advance so that the (A) hydrophobic powder is water.
- the present invention also relates to a method for producing a cosmetic comprising a step of producing an aqueous dispersion dispersed in a phase.
- the cosmetic production method includes (A) a hydrophobic powder, (B) a carboxylic acid-modified silicone, (C) a basic compound, (D It is preferable to include a step of preparing an aqueous dispersion in which the hydrophobic powder (A) is dispersed in an aqueous phase by previously mixing water) and (G) polyhydric alcohol.
- the pH of the aqueous dispersion is preferably in the range of 6.5 to 14.0.
- the cosmetic production method comprises (G) a polyhydric alcohol, (A) a hydrophobic powder, and (B) a carboxylic acid-modified silicone. You may include the process of mixing beforehand and obtaining a mixture, and the process of mixing (C) a basic compound and (D) water with the said mixture.
- the manufacturing method of the cosmetics which concern on this invention includes (G) polyhydric alcohol, (A) hydrophobic powder, and (B) carboxylic acid modified silicone.
- the pH of the hydrophobic powder dispersed in the aqueous phase is 6.5 to 14.0.
- a step of producing an aqueous dispersion in the range may be included.
- the cosmetic of the present invention has an aqueous phase, and the hydrophobic powder is well dispersed in the aqueous phase. Therefore, the hydrophobic powder does not aggregate over time.
- the cosmetic of the present invention can give a refreshing feeling during use and can form a cosmetic film having excellent water resistance on the skin. Furthermore, water-based cosmetics that were difficult to design using hydrophobic powders due to dispersibility in the past have excellent dispersibility and appearance of the hydrophobic powder, and are derived from the hydrophobic powder on the skin. Excellent water repellency and water resistance can be realized.
- the inventors of the present invention combined hydrophobic powder, carboxylic acid-modified silicone, basic compound, and water so that the carboxylic acid-modified silicone acts as a surfactant or dispersant in the water phase.
- the active powder can be dispersed well, and when it is applied to the skin, the carboxylic acid modification site of the carboxylic acid modified silicone changes from carboxylate to free carboxylic acid due to the weak acidity of the skin itself.
- the present invention has been completed by discovering that the dispersibility is lowered and the hydrophobic powder can be deposited on the skin to form a hydrophobic film on the skin.
- hydrophobic powder, carboxylic acid-modified silicone, and polyhydric alcohol are mixed uniformly, and further added with a basic compound and water to adjust the pH to 6.5 to 14.0, thereby dispersibility.
- a basic compound and water to adjust the pH to 6.5 to 14.0, thereby dispersibility.
- the cosmetics excellent in long-term storage stability can be obtained suitably.
- the cosmetic of the present invention contains an oil agent
- the cosmetic of the present invention when the cosmetic of the present invention is applied to the skin, the oil agent is deposited on the skin together with the hydrophobic powder to form a hydrophobic film, thereby improving the adhesion. Can do.
- the cosmetic of the present invention contains an oil, it is preferable to contain a surfactant as an emulsifier.
- the cosmetic of the present invention contains a salt of a higher fatty acid
- the dispersion of the hydrophobic powder in the aqueous phase can be made better before the application to the skin due to the surface active action of the salt.
- the cosmetic composition of the present invention contains an oil agent
- the salt of the higher fatty acid functions as a good emulsifier
- the salt changes to a free higher fatty acid
- the hydrophobic powder and It can be deposited on the skin together with an oil agent to form a hydrophobic film with better water resistance.
- the cosmetic of the present invention contains a polyhydric alcohol and / or a water-soluble thickener, the dispersibility of the hydrophobic powder in the aqueous phase can be further enhanced.
- the cosmetic of the present invention does not contain a surfactant other than the carboxylic acid-modified silicone, and when the cosmetic of the present invention contains a salt of a higher fatty acid, the interface other than the carboxylic acid-modified silicone and the salt of the higher fatty acid.
- the water resistance of the cosmetic film obtained on the skin can be further increased by the cosmetic of the present invention.
- Surfactants other than the surfactant component ((B) component or (B) and (F) component) that are essentially included in the cosmetic of the present invention are not particularly limited, but anionic, Various surfactants of cationic, nonionic and amphoteric are listed.
- the above “not including” means substantially not including, and “substantially” means up to 5% by mass or less of the total mass of the cosmetic of the present invention. It can be included. However, a smaller content is advantageous, preferably 3% by mass or less, more preferably 2% by mass or less, and still more preferably 1% by mass or less. Most preferably, the cosmetic of the present invention does not contain any surfactant other than the surfactant (component (B), or components (B) and (F)) that is essential.
- the cosmetic of the present invention contains at least one (A) hydrophobic powder.
- the “powder” in the present invention is generally used as a component of cosmetics, and includes white and colored pigments, fine particles (including so-called nanoparticles) such as an ultraviolet scattering agent, and extender pigments.
- White and colored pigments are used for coloring cosmetics, while extender pigments are used for improving the feel of cosmetics.
- white and colored pigments usually used in cosmetics, and extender pigments can be used without particular limitation. It is preferable to blend one kind or two or more kinds of powders.
- the powder shape (spherical, rod-like, needle-like, plate-like, indeterminate shape, spindle shape, etc.), particle size (fog-like, fine particles, pigment grade, etc.), and particle structure (porous, nonporous, etc.)
- the average primary particle diameter is in the range of 1 nm to 100 ⁇ m.
- the powder examples include inorganic powders, organic powders, surfactant metal salt powders (metal soaps), colored pigments, pearl pigments, metal powder pigments, and those obtained by combining these.
- inorganic powder titanium oxide, zirconium oxide, zinc oxide, cerium oxide, magnesium oxide, barium sulfate, calcium sulfate, magnesium sulfate, calcium carbonate, magnesium carbonate, talc, mica, kaolin, sericite, Muscovite, synthetic mica, phlogopite, saucite, biotite, lithia mica, silicic acid, anhydrous silicic acid, aluminum silicate, sodium silicate, sodium magnesium silicate, magnesium silicate, magnesium aluminum silicate, calcium silicate , Barium silicate, strontium silicate, metal tungstate, hydroxyapatite, vermiculite, hydrite, bentonite, montmorillonite, hectorite, zeolite, ceramic powder, di
- Inorganic yellow pigments inorganic black pigments such as black iron oxide and carbon black, inorganic purple pigments such as manganese violet and cobalt violet, inorganic green pigments such as chromium hydroxide, chromium oxide, cobalt oxide and cobalt titanate, bitumen and ultramarine blue Inorganic blue pigments such as Red No. 3 and Red No. 104 Red 106, Red 201, Red 202, Red 204, Red 205, Red 220, Red 226, Red 227, Red 228, Red 230, Red 401, Red 505, Yellow 4 No. 5, Yellow No. 5, Yellow No. 202, Yellow No. 203, Yellow No. 204, Yellow No. 401, Blue No. 1, Blue No. 2, Blue No.
- tar dyes are raked, and natural dyes such as carminic acid, laccaic acid, calsamine, bradylin, crocin
- natural dyes such as carminic acid, laccaic acid, calsamine, bradylin, crocin
- the metal powder pigment include metal powders such as aluminum, gold, silver, copper, platinum, and stainless steel.
- a hydrophobic powder is used. Therefore, when the surface of the powder itself is not hydrophobic, it is preferable to subject the surface to a hydrophobic treatment. Note that these hydrophobic powders may be combined.
- the hydrophobizing treatment is not particularly limited, and examples thereof include treating the powder with various hydrophobizing surface treatment agents.
- methylhydrogenpolysiloxane in Japanese cosmetics name: methicone
- Dimethicone / methicone copolymer
- dimethylpolysiloxane dimethylpolysiloxane in the Japanese cosmetic label
- silicone resin treatment silicone gum treatment
- acrylic silicone treatment fluorinated silicone treatment
- Metal soap treatment such as zinc stearate treatment; silane treatment such as silane coupling agent treatment and alkyl silane treatment; fluorine compound treatment such as perfluoroalkyl silane, perfluoroalkyl phosphate salt and perfluoropolyether treatment; N - Examples include amino acid treatment such as lauroyl-L-lysine treatment; oil agent treatment such as squalane treatment; acrylic treatment such as alkyl acrylate treatment, and the like. These two or more types can also be used in combination.
- treatment with a silicone compound is preferable from the viewpoint of water resistance and ease of dispersion with a carboxylic acid-modified silicone.
- methyl hydrogen polysiloxane, (dimethicone / methicone) copolymer, dimethyl polysiloxane, alkyl Treatment with silane is particularly preferred.
- Silicone elastomer powder can also be used as the hydrophobic powder.
- the silicone elastomer powder is a crosslinked product of a linear diorganopolysiloxane mainly composed of diorganosiloxy units (D units), and an organohydrogenpolysiloxane having a silicon-bonded hydrogen atom at the side chain or terminal and a side chain or A diorganopolysiloxane having an unsaturated hydrocarbon group such as an alkenyl group at the terminal can be suitably obtained by a crosslinking reaction under a hydrosilylation reaction catalyst.
- Silicone elastomer powder is softer and more resilient than silicone resin powder consisting of T units and Q units, and also has excellent oil absorption, so that it absorbs fats and oils on the skin and prevents breakup of makeup. it can.
- the silicone elastomer powder can take various shapes such as a spherical shape, a flat shape, and an indefinite shape.
- the silicone elastomer powder may be in the form of an oil dispersion.
- the cosmetic of the present invention is a silicone elastomer powder having a particle shape, and has an average primary particle diameter measured by observation using an electron microscope and / or laser diffraction / scattering method of 0.1. Silicone elastomer powder that falls within a range of ⁇ 50 ⁇ m and has a spherical primary particle shape can be suitably blended.
- the silicone elastomer constituting the silicone elastomer powder preferably has a hardness of 80 or less, more preferably 65 or less according to the type A durometer of JIS K 6253 “Testing method for hardness of vulcanized rubber and thermoplastic rubber”. .
- the silicone elastomer powder may be optionally subjected to a surface treatment with a silicone resin, silica or the like.
- the surface treatment include, for example, JP-A-2-243612, JP-A-8-12545, JP-A-8-12546, JP-A-8-12524, JP-A-9-241511, JP-A-9-241511. Examples thereof include those described in JP-A-10-36219, JP-A-11-193331, JP-A-2000-281523, and the like.
- the silicone elastomer powder corresponds to the cross-linked silicone powder listed in “Composition Component Standards by Cosmetic Variety”.
- silicone elastomer powders examples include Trefill E-506S, Trefill E-508, 9701 Cosmetic Powder, 9702 Powder manufactured by Toray Dow Corning. These silicone elastomer powders may be surface-treated, and examples of the surface treatment agent include inorganic oxides such as methylhydrogenpolysiloxane, silicone resin, metal soap, silane coupling agent, silica, and titanium oxide. , Fluorine compounds such as perfluoroalkyl silane and perfluoroalkyl phosphate ester salts.
- hydrophobic powders finely divided inorganic powders that have been hydrophobized are preferably used from the viewpoint of preventing ultraviolet rays, and among them, hydrophobized fine particle titanium oxide and / or hydrophobized fine particle zinc oxide are preferable.
- the particle diameter of the hydrophobized fine particle titanium oxide and / or the hydrophobized fine particle zinc oxide is preferably 1 to 200 nm from the viewpoint of the ultraviolet ray preventing effect and dispersibility, and more preferably 10 to 80 nm.
- a hydrophobic treated inorganic pigment powder, a pearl pigment powder or the like may be used as the hydrophobic inorganic particles, and the hydrophobic fine particle inorganic powder and the hydrophobic treated inorganic pigment powder may be used. You may use it combining pigment powder etc.
- the blending amount of the hydrophobic powder in the cosmetic of the present invention is not particularly limited, but is preferably 1 to 40% by mass, more preferably 2 to 35% by mass based on the total mass of the cosmetic. 3 to 30% by mass is even more preferable, 4 to 25% by mass is still more preferable, and 5 to 20% by mass is even more preferable.
- the cosmetic of the present invention contains at least one (B) carboxylic acid-modified silicone.
- the carboxylic acid-modified silicone in the cosmetic of the present invention is present in the cosmetic in a state where the carboxylic acid-modified site is anionized due to the presence of the basic compound described later. Therefore, the carboxylic acid-modified silicone functions as a surfactant or a dispersant, and (A) the hydrophobic powder can be well dispersed in the cosmetic of the present invention.
- the carboxylic acid-modified silicone contained in the cosmetic of the present invention is not particularly limited as long as it is an organosiloxane in which at least one carboxyl group-containing organic group is introduced into the side chain or terminal.
- the carboxyl group-containing organic group is introduced into the side chain of the organosiloxane.
- carboxylic acid-modified silicones those having a carboxyl group-containing organic group grafted to the silicone main chain, those having a carboxyl group-containing organic group added to one end of the silicone main chain, and carboxyl groups containing at both ends of the silicone main chain
- a silicone main chain is grafted with a carboxyl group-containing organic group.
- carboxylic acid-modified silicone has a long-chain alkyl group, blending stability with an organic oil agent such as hydrocarbon oil or an organic cosmetic raw material (particularly, a UV absorber) may be improved.
- a linking group may be present between the carboxyl group and the silicon atom, and examples of the linking group include divalent or higher valent organic groups such as an alkylene group and a polyoxyalkylene group which may have a hetero atom. However, it is not particularly limited. Further, it may be a carboxylic acid-modified silicone in which (n-1) carboxyl groups are bonded to a silicon atom by an n-valent linking group (n is a number of 3 or more). Specifically, the silicone having a carboxyl group in the main chain or side chain of the silicone through the following linking group is included in the carboxylic acid-modified silicone of the present invention.
- organopolysiloxane having a silicon-bonded carboxyl group-containing organic group described in JP-A-11-504665 Wherein R is a C 1 -C 12 alkylene group, a C 1 -C 12 alkyleneoxy group, an oxygen atom, a sulfur atom, —NH—, —NR′— (R ′ is a C 1 -C 6 alkyl group) or A divalent group containing these combinations)
- R is a C 1 -C 12 alkylene group, a C 1 -C 12 alkyleneoxy group, an oxygen atom, a sulfur atom, —NH—, —NR′— (R ′ is a C 1 -C 6 alkyl group) or A divalent group containing these combinations)
- R 1 to R 4 are the same or different and each may have a hetero atom-containing substituent, a linear or branched alkylene group having 2 to 22 carbon atoms, an alkenylene group or an arylene group
- a group, X represents —O— or NH—, and M represents a hydrogen atom
- Organopolysiloxane having the following carboxyl group-containing organic group described in JP-T-2005-524747: Wherein B represents an alkylene residue having 2 to 30 carbon atoms, optionally substituted by one or more alkyl groups having 1 to 30 carbon atoms; R ′ represents a hydrogen atom or an alkyl group having 1 to 30 carbon atoms, E is absent or has 1 to 5 carbon atoms, preferably 1 to 3 carbon atoms, optionally substituted by one or more alkyl groups having 1 to 30 carbon atoms.
- An alkylene residue; M is a hydrogen atom
- An organopolysiloxane having a carboxyl group-containing organic group represented by the following average composition formula described in JP-A-2009-263634: [Wherein R 1 is selected from an alkyl group having 1 to 30 carbon atoms, a fluoroalkyl group having 1 to 30 carbon atoms, an aryl group having 6 to 30 carbon atoms, and an aralkyl group having 6 to 30 carbon atoms.
- R 2 is a group represented by the following formula (2), and when c is 0, R 2 is bonded to at least one terminal of the organopolysiloxane, (Wherein R 4 is a divalent hydrocarbon group having 2 to 20 carbon atoms and having or not having an oxygen atom, R 5 is a hydrogen atom, and R 6 are independently a hydrogen atom or An alkyl group having 1 to 6 carbon atoms, and R 7 is a hydrogen atom or an alkyl group having 1 to 6 carbon atoms)
- R 3 is a group represented by the following formula (3), Wherein R 2 is as defined above, and R 8 is independently of each other an alkyl group having 1 to 30 carbon atoms, a fluoroalkyl group having 1 to 30 carbon atoms, or an aryl having 6 to 30 carbon atoms.
- a group selected from a group and an aralkyl group having 6 to 30 carbon atoms Q is C d H 2d (where d is an integer of 1 to 5, preferably an integer of 2 to 4) or an oxygen atom; k is an integer of 0 to 500, preferably 1 to 100, more preferably 5 to 60, and h is an integer of 0 to 3, preferably 0).
- At least one silicon atom in the side chain or terminal of the silicone main chain is represented by the general formula: -R 1- (OR 2 ) p- (O) wR 3 -COOH (wherein R 1 represents a linear or branched alkylene group having 2 to 22 carbon atoms, R 2 represents a linear or branched alkylene group having 2 to 4 carbon atoms, and R 3 represents a bond)
- R 1 represents a linear or branched alkylene group having 2 to 22 carbon atoms
- R 2 represents a linear or branched alkylene group having 2 to 4 carbon atoms
- R 3 represents a bond
- a carboxyl group-containing organic group represented by a hand (-) or a linear or branched alkylene group having 1 to 22 carbon atoms
- p represents a number of 0 to 200
- w represents a number of 0 or 1.
- R 1 is a straight chain or branched chain group having 2 to 22 carbon atoms, preferably a straight chain alkylene group having 2 to 12 carbon atoms, particularly preferably 2 to 10 carbon atoms.
- examples of the linear or branched alkylene group having 2 to 4 carbon atoms of R 2 include ethylene, propylene, trimethylene, and butylene groups, and an ethylene group is particularly preferable.
- Examples of the linear or branched alkylene group having 1 to 22 carbon atoms of R 3 include methylene, ethylene, ethylethylene, propylene, trimethylene, tetramethylene, pentamethylene, hexamethylene, heptamethylene, octamethylene, nonamethylene and decamethylene. , Undecamethylene, dodecamethylene, tridecamethylene, tetradecamethylene, pentadecamethylene, hexadecamethylene, and the like, among which 1 to 12 carbon atoms, especially the sum of carbon atoms of R 1 and R 3 Is preferably 2 to 22.
- p represents a number of 0 to 200, preferably a number of 0 to 20, and particularly preferably a number of 0 to 10.
- W represents a number of 0 or 1, and is preferably 0.
- the carboxyl group-containing organic group is represented by the structural formula — (C n H 2n ) —COOH, and one carboxyl group is a straight chain having 3 to 44 carbon atoms. Alternatively, it preferably has a structure bonded to a silicon atom via a branched alkylene group.
- n is a number of 3 to 44, preferably a number of 3 to 20, and particularly preferably a number of 3 to 16.
- Rc represents a carboxyl group-containing organic group represented by the general formula: —R 1 — (OR 2 ) p- (O) wR 3 —COOH), and R's are the same or different and have 1 to 22 represents an alkyl or alkoxy group or a phenyl group
- R ′ is Rc or R
- a and b are each a number in the range of 0 or more
- the carboxylic acid-modified silicone disclosed in JP-A-8-109263 a part of the carboxylic acid-modified silicone disclosed in International Publication No. 2009/22621, etc. (other than those having a siloxane dendron structure)
- the carboxylic acid-modified silicone represented by the formula (A) and preferably used in the present invention is included.
- carboxylic acid-modified silicone represented by the structural formula (A) include a + b in the range of 0 to 500, particularly b> 0, and the silicone main chain has the above general formula.
- Carboxylic acid-modified silicone grafted with a carboxyl group-containing organic group represented by -R 1- (OR 2 ) p- (O) wR 3 -COOH or b 0, and R ′ at both ends of the silicone main chain
- a carboxylic acid-modified silicone which is a carboxyl group-containing organic group represented by the general formula: -R 1- (OR 2 ) p- (O) wR 3 -COOH can be mentioned.
- “b> a” means that more than half of the side chain moieties have a carboxyl group-containing organic group, and a + b is preferably a number in the range of 1 to 500.
- b is a number in the range of 1 to 200 or in the range of 1 to 50. Most preferably it is a number.
- R is preferably a methyl group, an alkoxy group or a phenyl group, but is stable in blending with an organic oil agent such as hydrocarbon oil or an organic cosmetic raw material (particularly a UV absorber). From the viewpoint of properties, it may have a long chain alkyl group having 6 to 22 carbon atoms in part.
- the modification rate by the carboxyl group-containing organic group is not particularly limited. However, if a + b is a number in the range of 0 to 500, including the case where the carboxyl group-containing organic group is bonded to both ends of the silicone main chain. It is preferable that the molecule has an average of 2 to 100 carboxyl group-containing organic groups in the molecule.
- such a carboxylic acid-modified silicone is converted into a carboxylic acid by a known method, for example, an addition reaction of dimethylpolysiloxane having an Si—H group and an unsaturated carboxylic acid ester compound under a platinum catalyst, and further saponification.
- a method for obtaining a carboxylic acid-modified silicone having both ends by an equilibration reaction using tetramethyldisiloxane, cyclic siloxane and an acidic catalyst (Silicone Handbook, edited by Kunio Ito, Nikkan Kogyo Shimbun, pages 166-167) Can be manufactured.
- a commercially available carboxylic acid-modified silicone can be used as it is or after removing the solvent.
- Specific examples thereof include SF8418, BY16-880, BY16-754, BY16-750, FV2056, FZ-3806 (manufactured by Toray Dow Corning), TSF-4770, TSF4771 (manufactured by Momentive Performance Materials), X-22-162A, X-22-162C, X-22-3701E, X-22-3710 (Shin-Etsu Chemical Co., Ltd.).
- the cosmetic composition of the present invention preferably contains the (B) carboxylic acid-modified silicone in a range of 0.01 to 20 parts by mass with respect to 10 parts by mass of the hydrophobic powder, and in a range of 0.1 to 15 parts by mass. Preferably, it is contained in the range of 1 to 10 parts by mass.
- the cosmetic of the present invention contains at least one (C) basic compound.
- the basic compound used in the present invention is not particularly limited as long as it is a compound that exhibits basicity when dissolved in water, and various inorganic compounds and organic compounds can be used. You may mix
- organic compound examples include monoethanolamine, triethanolamine, 2-amino-2-methyl-1,3-propanediol, aminomethylpropanol, arginine, guanidine and the like.
- Examples of the inorganic compound include sodium hydroxide, sodium carbonate, potassium hydroxide, potassium carbonate, calcium hydroxide, calcium carbonate, ammonia and the like, and among these, potassium hydroxide can be particularly preferably used.
- the blending amount of the basic compound in the cosmetic of the present invention is not particularly limited, but in the case of a monovalent base with respect to 1 mol of carboxylic acid groups contained in the blended carboxylic acid-modified silicone, a carboxylic acid is used.
- the group / monovalent base (molar ratio) is preferably from 1 / 0.5 to 1 / 1.5. Specifically, based on the total mass of the cosmetic, 0.01 to 5% by mass is preferable, 0.03 to 4% by mass is more preferable, 0.05 to 4% by mass is even more preferable, and 0.08 to 3% by weight is even more preferred.
- the cosmetic of the present invention contains (D) water. Water forms an aqueous phase in the cosmetic of the present invention.
- the blending amount of water in the cosmetic of the present invention is not particularly limited, but is preferably 20 to 95% by mass, more preferably 40 to 80% by mass based on the total mass of the cosmetic, and 45 to 70 mass% is even more preferred, 47-65 mass% is even more preferred, and 50-60 mass% is even more preferred.
- the pH of the cosmetic of the present invention is preferably weakly alkaline, specifically in the range of 7.1 to 9.5, more preferably in the range of 7.2 to 8.5.
- the (B) carboxylic acid-modified silicone has a good function as a surfactant by anionizing the carboxylic acid-modified site of the (B) carboxylic acid-modified silicone. Can be demonstrated. Therefore, (A) the hydrophobic powder is well dispersed in the aqueous phase and can maintain a stable dispersion state over time.
- the cosmetic of the present invention can contain at least one (G) polyhydric alcohol.
- the polyhydric alcohol By including the polyhydric alcohol, it is possible to adjust the moisturizing feeling and feeling of use of the cosmetic of the present invention, and the hydrophobic powders of the component (B) and the component (A) are mixed with the polyhydric alcohol in advance. By doing so, it can be more uniformly dispersed in the aqueous phase.
- polyhydric alcohol examples include sorbitol, xylitol, propylene glycol, dipropylene glycol, 1,3-butylene glycol, glycerin, diglycerin, polyethylene glycol and the like. These polyhydric alcohols can be used alone or Two or more types can be used in combination.
- the component (B) and the hydrophobic powder of the component (A) are mixed in advance together with the polyhydric alcohol, it is preferable in that the liquid polyhydric alcohol can be uniformly dispersed in the aqueous phase.
- propylene glycol and dipropylene are preferred.
- Glycol, 1,3-butylene glycol, glycerin, diglycerin and combinations thereof are preferred.
- the blending amount of the polyhydric alcohol in the cosmetic of the present invention is not particularly limited, but is preferably 0.3 to 30% by mass, and preferably 0.5 to 25% by mass based on the total mass of the cosmetic. Is more preferable, 1 to 20% by mass is still more preferable, 2 to 20% by mass is still more preferable, and 3 to 15% by mass is still more preferable.
- the cosmetic of the present invention can contain at least one (E) oil.
- the “oil” in the present invention is generally used as a cosmetic ingredient and is not particularly limited.
- the oil agent is usually liquid at room temperature, but it may be a solid such as wax, and may be a high-viscosity and viscous gum or paste described later.
- the oil agent is preferably at least one liquid at 5 to 100 ° C. selected from the group consisting of silicone oil, nonpolar organic compounds and low polar organic compounds.
- Silicone oil is hydrophobic and its molecular structure may be cyclic, linear or branched. Viscosity at 25 ° C. of the silicone oil is generally in the range of 0.65 ⁇ 100,000mm 2 / s, preferably in the range of 0.65 ⁇ 10,000mm 2 / s.
- silicone oils examples include linear organopolysiloxanes, cyclic organopolysiloxanes, and branched organopolysiloxanes. Among these, volatile, linear organopolysiloxane, cyclic organopolysiloxane, and branched organopolysiloxane are preferable.
- the linear organopolysiloxane may be a trimethylsiloxy group-blocked dimethylpolysiloxane having a molecular chain at both ends (a low-viscosity dimethylsilicone such as 2 mPa ⁇ s or 6 mPa ⁇ s to a high viscosity such as 1 million mPa ⁇ s).
- a low-viscosity dimethylsilicone such as 2 mPa ⁇ s or 6 mPa ⁇ s to a high viscosity such as 1 million mPa ⁇ s.
- Organohydrogenpolysiloxane trimethylsiloxy group-capped methylphenyl polysiloxane with both molecular chains, trimethylsiloxy group-capped dimethylsiloxane / methylphenylsiloxane copolymer with both molecular chains, trimethylsiloxy group-capped diphenylpolysiloxane with both molecular chains , Trimethylsiloxy group-blocked dimethylsiloxane / diphenylsiloxane copolymer, both ends of molecular chain, Trimethylpentaphenyltrisiloxane, Phenyl (trimethylsiloxy) siloxane, Trimethylsiloxy group-blocked methyl at both ends of molecular chain Alkylpolysiloxane, trimethylsiloxy group-capped dimethylpolysiloxane / methylalkylsiloxane copolymer with both ends of molecular chain, trimethylsiloxy group-
- Cyclic organopolysiloxanes include hexamethylcyclotrisiloxane (D3), octamethylcyclotetrasiloxane (D4), decamethylcyclopentasiloxane (D5), dodecamethylcyclohexasiloxane (D6), 1,1-diethylhexamethyl.
- Cyclotetrasiloxane phenylheptamethylcyclotetrasiloxane, 1,1-diphenylhexamethylcyclotetrasiloxane, 1,3,5,7-tetravinyltetramethylcyclotetrasiloxane, 1,3,5,7-tetramethylcyclotetra Siloxane, 1,3,5,7-tetracyclohexyltetramethylcyclotetrasiloxane, tris (3,3,3-trifluoropropyl) trimethylcyclotrisiloxane, 1,3,5,7-tetra (3-methacryloxypro L) Tetramethylcyclotetrasiloxane, 1,3,5,7-tetra (3-acryloxypropyl) tetramethylcyclotetrasiloxane, 1,3,5,7-tetra (3-carboxypropyl) tetramethylcyclotetrasiloxane 1,3,5,7-tetra (3-vinyloxypropy
- branched organopolysiloxane examples include methyltristrimethylsiloxysilane, ethyltristrimethylsiloxysilane, propyltristrimethylsiloxysilane, tetrakistrimethylsiloxysilane, and phenyltristrimethylsiloxysilane.
- nonpolar organic compound and the low polarity organic compound hydrocarbon oil and fatty acid ester oil are preferable. These are components that are widely used as base materials for makeup cosmetics.
- hydrocarbon oils examples include liquid paraffin, light liquid isoparaffin, heavy liquid isoparaffin, petrolatum, n-paraffin, isoparaffin, isododecane, isohexadecane, polyisobutylene, hydrogenated polyisobutylene, polybutene, ozokerite, ceresin, and microcrystalline wax.
- paraffin wax polyethylene wax, polyethylene / polypropylene wax, squalane, squalene, pristane, polyisoprene and the like.
- fatty acid ester oils include hexyldecyl octoate, cetyl octanoate, isopropyl myristate, isopropyl palmitate, butyl stearate, hexyl laurate, myristyl myristate, oleyl oleate, decyl oleate, octyldodecyl myristate, Hexyldecyl dimethyloctanoate, cetyl lactate, myristyl lactate, diethyl phthalate, dibutyl phthalate, lanolin acetate, propylene glycol dioleate, glyceryl tri-2-ethylhexanoate, trimethylolpropane tri-2-ethylhexanoate, ditritrihexanoate triethylhexanoate Methylolpropane, (isostearic acid / se
- a higher alcohol having 10 to 30 carbon atoms can be used as the low polarity organic compound.
- a higher alcohol is used as an emulsion stabilizing component, the amount of hydrophilic surfactant can be reduced, and water resistance can be further improved.
- the higher alcohol is a saturated or unsaturated monohydric aliphatic alcohol, and the hydrocarbon group portion may be either linear or branched, but is more preferably linear. preferable.
- Examples of the higher alcohol having 10 to 30 carbon atoms include lauryl alcohol, myristyl alcohol, palmityl alcohol, stearyl alcohol, behenyl alcohol, hexadecyl alcohol, oleyl alcohol, isostearyl alcohol, hexyldodecanol, octyldodecanol, cetostearyl.
- Examples include alcohol, 2-decyltetradecinol, cholesterol, sitosterol, phytosterol, lanosterol, lanolin alcohol, hydrogenated lanolin alcohol, and the like.
- the blending amount of the oil agent in the cosmetic of the present invention is not particularly limited, but is preferably 3 to 60% by mass, more preferably 4 to 50% by mass based on the total mass of the cosmetic. 40% by weight is even more preferred, 6-30% by weight is even more preferred, and 7-20% by weight is even more preferred.
- the cosmetic of the present invention may contain at least one salt of (F) a higher fatty acid.
- Higher fatty acid salts can be used as emulsifiers.
- the higher fatty acid component used for forming the salt may be composed of only the liquid higher fatty acid, or may be composed of a mixed system of liquid higher fatty acid and solid higher fatty acid, but preferably contains liquid higher fatty acid. In the case of only a solid higher fatty acid, the elongation and spread are not sufficient, and the washing-off property with soap is lowered.
- the content of the liquid higher fatty acid is preferably at least 30% by mass or more, more preferably 50% by mass or more, and still more preferably 90% by mass or more, based on the total mass of the higher fatty acid. Improve and washability with soap increases. Further, when the higher fatty acid component is only liquid higher fatty acid, there is no need to provide a heating step in preparation of the cosmetic, which is economically advantageous and the quality of the cosmetic is stable.
- the type of higher fatty acid is not particularly limited.
- liquid fatty acids such as isostearic acid, hexyldecanoic acid and oleic acid are preferable, and isostearic acid is more preferable from the viewpoint of the function of the salt as an emulsifier, oxidation stability, and feeling of use.
- isostearic acid means one or a mixture of two or more branched stearic acids.
- 5,7,7-trimethyl-2- (1,3,3-trimethylbutyl) octanoic acid is converted to a branched aldehyde having 9 carbons by oxo reaction of isobutylene dimer, and then carbon is obtained by aldol condensation of this aldehyde.
- It can be produced by using a branched unsaturated aldehyde of formula 18 and then carrying out a hydrogenation reaction and an oxidation reaction (hereinafter referred to as “aldol condensation type”).
- Aldol condensation-type isostearic acid is commercially available, for example, from Nissan Chemical Industries.
- 2-heptylundecanoic acid can be produced by dimerizing nonyl alcohol by a gerbet reaction (Guerbet reaction: also called Guerbet reaction) and then oxidizing it.
- 2-Heptylundecanoic acid is commercially available from, for example, Mitsubishi Chemical Corporation, and similar compounds having slightly different branch positions are commercially available from Nissan Chemical Industries.
- a type in which the starting alcohol is not a straight chain alcohol but is branched at two locations is also commercially available from Nissan Chemical Industries (hereinafter generally referred to as “gerbet reaction type”).
- isostearic acid called emery type can be used.
- Emery type isostearic acid is obtained by hydrogenating unsaturated fatty acid by-product when synthesizing dimer acid from oleic acid, and has 18 carbon atoms and methyl group in the side chain, and its structure is uncertain (See, for example, J. Amer. Oil Chem. Soc. 51, 522 (1974)). Specific examples thereof include those commercially available from Emery, Inc., USA, and isostearic acid manufactured by Higher Alcohol Industry. Mention may be made of the acid EX.
- the starting material of dimer acid which is a starting material of emery type isostearic acid may include not only oleic acid but also linoleic acid, linolenic acid, and the like. In the present invention, this emery type is particularly preferably used.
- the solid higher fatty acid that can be used in combination with the liquid higher fatty acid is usually one having 10 to 25 carbon atoms, particularly preferably one having 11 to 22 carbon atoms.
- Specific examples thereof include stearic acid, Examples include behenic acid, hydroxystearic acid, palmitic acid, myristic acid, lauric acid and the like.
- the higher fatty acid may be used in advance as a higher fatty acid salt neutralized with a base, but in the production process of the cosmetic of the present invention, the higher fatty acid is added together with (C) a basic compound, In the production process, both components may be neutralized to form a higher fatty acid salt.
- the higher fatty acid component and the basic compound are usually added so as to be in the same amount, but they are not necessarily in the same amount, and the pH of the aqueous phase of the prepared cosmetic is 7
- the blending amount of both is suitably adjusted so that the higher fatty acid / basic compound (molar ratio) is in the range of 1 / 0.5 to 1 / 1.5, for example, so that it is in the range of 1 to 9.5. May be.
- the amount of the higher fatty acid salt in the cosmetic of the present invention is not particularly limited, but is preferably 0.1 to 12% by mass, and preferably 0.5 to 10% by mass based on the total mass of the cosmetic. Is more preferable, and 1 to 5% by mass is even more preferable.
- the cosmetic of the present invention can contain at least one (H) water-soluble thickener.
- the viscosity and feeling of use of the cosmetic of the present invention can be adjusted, and the storage stability is further improved.
- water-soluble thickener examples include carboxyvinyl polymer, sodium polyacrylate, polyethylene glycol, acrylic acid / alkyl methacrylate copolymer, polyoxyethylene polyoxypropylene block copolymer, polyvinyl alcohol, polyvinyl pyrrolidone, polyvinyl methyl ether.
- the blending amount of the water-soluble thickener in the cosmetic of the present invention is not particularly limited, but is preferably 0.01 to 3% by mass based on the total mass of the cosmetic, 0.05 to 2 % By mass is more preferred, 0.1-2% by mass is even more preferred, 0.3-1.5% by mass is even more preferred, and 0.5-1% by mass is even more preferred.
- Optional ingredients in the cosmetic of the present invention, other components that are used in normal cosmetics within a range that does not interfere with the effects of the present invention: for example, other than ultraviolet absorber, organic resin, hydrophilic powder, component (G) Moisturizer, thickener other than (H) component, preservative, antibacterial agent, fragrance, salt, antioxidant, pH adjuster other than (C) component, chelating agent, cooling agent, anti-inflammatory agent, physiologically active ingredient (Whitening agents, cell activators, rough skin improving agents, blood circulation promoters, skin astringents, antiseborrheic agents, etc.), vitamins, amino acids, nucleic acids, hormones, inclusion compounds, and the like can be added.
- Other components are not particularly limited.
- the production process of the cosmetic according to the present invention is optional, and is composed of (A) a hydrophobic powder, (B) a carboxylic acid-modified silicone, (C) a basic compound, and (D) water by mixing the respective components. (A) It will not specifically limit, if the cosmetics in which hydrophobic powder disperse
- the cosmetic of the present invention can be produced, for example, through a step of producing an aqueous dispersion in which the component (A) is dispersed in an aqueous phase by mixing the components (A) to (D). As needed, you may further mix at least 1 sort (s) selected from the group which consists of said (E), (F), (G), and (H) component.
- the manufacturing method of the cosmetics of this invention includes the process of disperse
- the pH of the aqueous dispersion is preferably in the range of 6.5 to 14.0, more preferably in the range of 7.0 to 12.0, and in the range of 7.5 to 10.0. Is even more preferred.
- the method for producing a cosmetic according to the present invention includes a step of previously mixing (G) a polyhydric alcohol, (A) a hydrophobic powder, and (B) a carboxylic acid-modified silicone (a slurry composition as a precursor). It is preferable to include, in particular, by mixing a mixture containing the components (A), (B) and (G) obtained by the premixing step, (C) a basic compound and (D) water, to a pH of 6 More preferably, the method further comprises a step of producing an aqueous dispersion having a hydrophobic powder dispersed in an aqueous phase of .5 to 14.0.
- the initial dispersibility of (A) hydrophobic powder in the aqueous phase is improved.
- the composition (aqueous dispersion) containing the mixture obtained by the preliminary mixing step and in which (A) the hydrophobic powder is dispersed in the aqueous phase has a pH of 6.5 using the component (C) and the like.
- the composition may be the cosmetic itself, or may be used as a cosmetic precursor (premix or cosmetic raw material) in the production of another cosmetic.
- composition (aqueous dispersion) obtained by the step of producing an aqueous dispersion in which the powder is dispersed in the aqueous phase may be the cosmetic itself, or a cosmetic precursor (premix or cosmetic). As a raw material), it may be used for the production of another cosmetic.
- the cosmetic of the present invention contains the component (F)
- the higher fatty acid as the raw material of the component (F) and the basic compound (C) are added separately, and in the production process of the cosmetic, It is preferable to form a salt from the viewpoint of operability and quality stability.
- the heating step required when using solid higher fatty acid which is economically advantageous and of quality. Contributes to stabilization.
- the cosmetic of the present invention may be in the form of cream, gel, emulsion, or liquid.
- the cosmetic of the present invention is, for example, a basic cosmetic such as a milk, a cream, or a cosmetic liquid, or a base makeup.
- Can be used as makeup cosmetics such as cosmetics, sunscreen agents, foundations, eye shadows, eyeliners, water fountains, etc., and can also be used as sunscreen agents for hair and scalp, temporary hair dyes, etc. .
- a hydrophobic fine particle inorganic powder is used as the hydrophobic powder, An aqueous dispersion suitable as a raw material for foundation can be obtained.
- the cosmetic of the present invention is preferably in the form of an oil-in-water emulsion composition.
- the water constituting the continuous phase is in direct contact with the skin, so that it is fresh and refreshing to use.
- the oil-in-water emulsion composition of the present invention has a hydrophobic powder that is difficult to design using a hydrophobic powder due to dispersibility in the prior art.
- the oil-in-water emulsion composition is applied on the skin, it achieves excellent water repellency and water resistance derived from hydrophobic powder, and It is possible to provide cosmetics that are excellent in makeup (long lasting).
- the cosmetic of the present invention is preferably applied onto weakly acidic skin, for example, preferably applied onto skin having a pH of 5.1 to 7.0.
- the pH of the application surface after 30 minutes is preferably 7.0 or less, and further, the pH Is more preferably 6.7 or less.
- the weak acidity inherent to the skin surface causes the carboxylic acid modification site of the carboxylic acid modified silicone to become non-ionized, reducing the surface active function and reducing the dispersion function of the hydrophobic powder. To do. Therefore, the hydrophobic powder can no longer be stably dispersed in the aqueous phase and is deposited on the skin. In this way, a hydrophobic cosmetic coating is formed on the skin.
- the cosmetic of the present invention contains an oil
- the oil when the skin is applied, the oil is also deposited on the skin together with the hydrophobic powder to form a hydrophobic cosmetic film.
- the decorative coating contains not only hydrophobic powder but also an oil agent, it has higher water resistance and adhesion.
- the cosmetic of the present invention contains a salt of a higher fatty acid
- the dispersion of the hydrophobic powder in the aqueous phase is more enhanced by the surface active action of the salt. It can be good.
- a salt of a higher fatty acid functions as a good emulsifier, but when applied to the skin, the salt changes to a free higher fatty acid, and the higher fatty acid deposits on the skin together with the hydrophobic powder, making it hydrophobic.
- the cosmetic coating contains not only a hydrophobic powder but also a higher fatty acid (and may further contain an oil agent), and thus has higher water resistance.
- the cosmetic of the present invention can form a cosmetic film having excellent water resistance on the skin, so that it does not easily lose its makeup due to sweat, rain, etc., and has excellent makeup.
- the skin is slightly acidic and the composition applied on the skin is also weakly acidic.
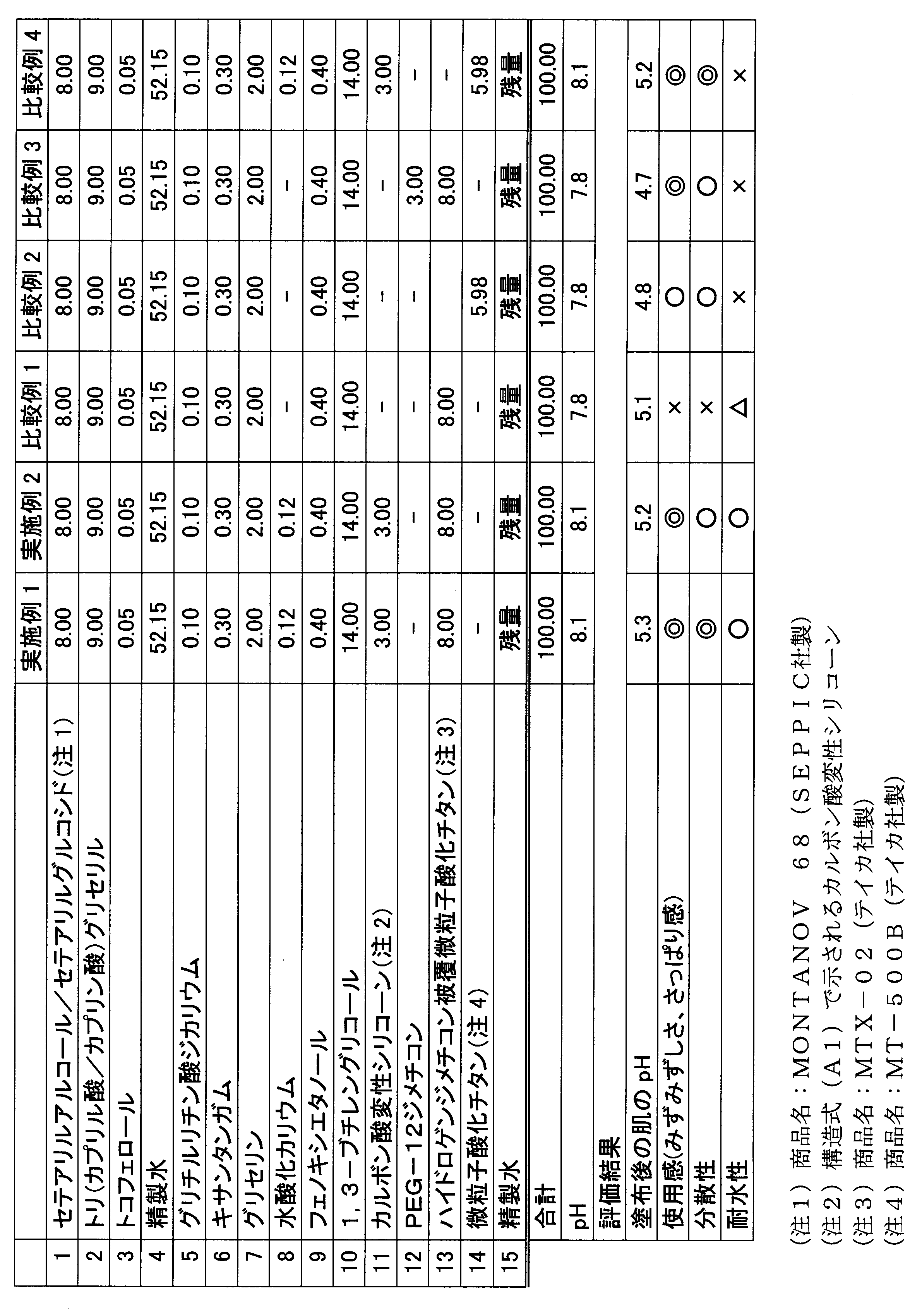
- Example 1 and Comparative Examples 1 to 4 Components (1) to (3) were heated and mixed to form an oil phase.
- Components (4) to (9) were heated and mixed to form an aqueous phase.
- the oil phase and the aqueous phase were mixed and emulsified at a specified temperature (80 ° C.).
- a premixed component (10) to (14) was added to and mixed with the emulsion, then component (15) was mixed and cooled to room temperature to obtain an oil-in-water emulsion cosmetic.
- the amount of untreated titanium oxide of component (14) was adjusted to be the same as the amount of titanium oxide that is the hydrophobic powder of component (13).
- Example 2 Components (1) to (3) were heated and mixed to form an oil phase. Components (4) to (10) were heated and mixed to form an aqueous phase. The oil phase and the aqueous phase were mixed and emulsified at a specified temperature (80 ° C.). A premixed component (11) to (14) was added to and mixed with the emulsion, then component (15) was mixed and cooled to room temperature to obtain an oil-in-water emulsion cosmetic.
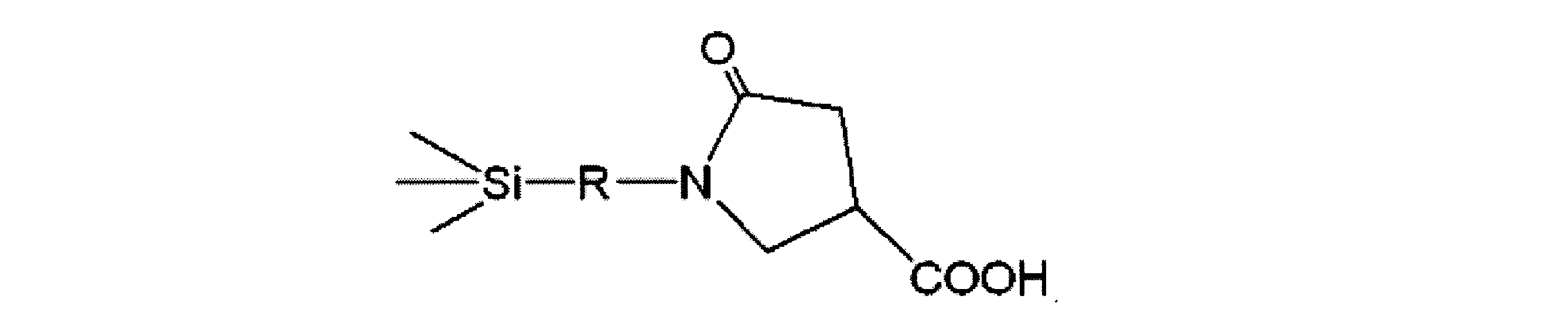
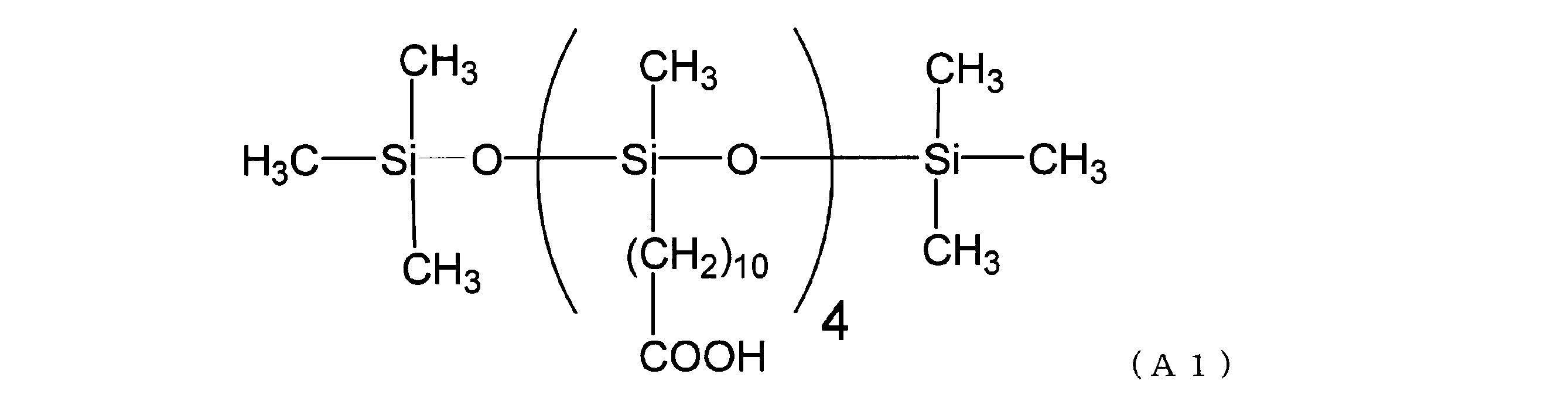
- Carboxylic acid-modified silicone (A1) As the carboxylic acid-modified silicone, one represented by the following structural formula (A1) was used.
- the carboxylic acid-modified silicone of the structural formula (A1) is a methyl hydrosilane having a side chain portion consisting only of a methylhydrogensiloxy unit represented by SiH (CH 3 ) O and having both molecular chain ends blocked with a trimethylsiloxy group.
- Genpolysiloxane and undecylenic acid can be synthesized by hydrosilylation reaction in the presence of a platinum catalyst in such an amount that the molar ratio of undecylenic acid to silicon-bonded hydrogen atoms is 1 or more.
- the evaluation object was stored in a thermostatic bath at 50 ° C. for 30 days, and the dispersion state of the hydrophobic powder was visually observed and evaluated according to the following criteria.
- Examples 1 and 2 which contain the hydrophobic powder in the aqueous phase together with the carboxylic acid-modified silicone and the basic compound show the dispersibility of the hydrophobic powder. While excellent in use feeling and water resistance, Comparative Example 1 containing no carboxylic acid-modified silicone was poor in dispersibility and could not obtain a uniform emulsion. Comparative Examples 2 and 4 using untreated titanium oxide without hydrophobization treatment and Comparative Example 3 using polyether modified silicone PEG-12 dimethicone instead of carboxylic acid modified silicone are inferior in water resistance. It was. Comparing Examples 1 and 2, Example 1 in which 1,3-butylene glycol, which is a polyhydric alcohol, hydrophobic powder, and carboxylic acid-modified silicone were mixed in advance was superior in dispersibility.
- Example 3 Oil-in-water emulsified cosmetic (sunscreen) The components shown in Table 3 below were mixed by the following production method to obtain an oil-in-water emulsion cosmetic.
- Phase A (components 1 to 5) was mixed, and premixed phase B (components 6 to 12) was added and emulsified. After emulsification, Phase C (components 13 to 19) was added and mixed. Furthermore, after adding and mixing the D phase (components 20 to 22) previously mixed, the E phase (component 23) was added, mixed and degassed to obtain an oil-in-water emulsion cosmetic.
- Example 3 From the evaluation results of Example 3, the salt of higher fatty acid (isostearic acid) (which is in a salt state due to the presence of potassium hydroxide) and a hydrophilic surfactant (cetearyl used in Example 1) It can be seen that the water resistance is further improved when no alcohol and cetearyl glucoside) are used.
- Example 4 and Comparative Examples 5 to 6 Aqueous foundation (two-layer separation type) Each component shown in the following Table 4 was mixed by the following production method to obtain an aqueous foundation.
- Phase A (components 1 to 6) was mixed to form an aqueous phase. To this was added and mixed the phase B (components 7 to 14) previously mixed, and then the phase C (component 15) was added and mixed to obtain an aqueous foundation.
- the evaluation object was stored for 30 days in a thermostatic bath at 50 ° C., and the redispersibility of the hydrophobic powder was visually observed and evaluated according to the following criteria.
- X The hydrophobic powder does not disperse in the aqueous phase as it is immediately after preparation.
- Example 4 had excellent redispersibility of the hydrophobic powder after standing and separation, and also had good water resistance.
- Comparative Example 5 containing no carboxylic acid-modified silicone the hydrophobic powder was not uniformly dispersed even when shaken, and could not be uniformly applied to the skin.
- Comparative Example 6 using hydrophilic untreated titanium oxide did not repel water and was inferior in water resistance.
- Example 5 Aqueous dispersion (for sunscreen cosmetics) The components shown in Table 5 below were mixed by the following production method to obtain an aqueous dispersion of hydrophobized fine particle titanium oxide.
- Example 5 In the aqueous dispersion of Example 5, the hydrophobized fine particle titanium oxide was uniformly dispersed in the aqueous phase without aggregation.
- Example 6 Oil-in-water emulsified cosmetic using an aqueous dispersion Oil-in-water emulsified cosmetic using the aqueous dispersion of Example 5 and mixing the components shown in Table 6 below by the following production method. Got.
- the oil-in-water emulsified cosmetic of Example 6 was able to easily disperse the hydrophobized fine particle titanium oxide uniformly in the outer aqueous phase by using an aqueous dispersion, and had water resistance. .
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Emergency Medicine (AREA)
- Dermatology (AREA)
- Cosmetics (AREA)
Abstract
Description
(A)疎水性粉体、
(B)カルボン酸変性シリコーン、
(C)塩基性化合物、及び、
(D)水
を含み、前記(A)疎水性粉体が水相中に分散していることを特徴とする化粧料によって達成される。
Rcは、一般式:-R1-(OR2)p-(O)w-R3-COOH(R1 は炭素数2~22の直鎖又は分岐鎖のアルキレン基を表し、R2 は炭素数2~4の直鎖又は分岐鎖のアルキレン基を表し、R3 は結合手(-)又は炭素数1~22の直鎖若しくは分岐鎖のアルキレン基を表し、pは0~200の数を表し、wは0又は1の数を表す)で示されるカルボキシル基含有有機基を表し、
Rは、同一又は異なり、炭素数1~22のアルキル若しくはアルコキシ基又はフェニル基を表し、
R´は、Rc又はRであり、
a、bは各々0以上の範囲の数であり、a+bは0~1000の範囲の数である。但し、b=0のとき、R´の少なくとも一方はRcである。}
で表されるものであることが好ましい。
本発明の化粧料は少なくとも1種の(A)疎水性粉体を含む。
本発明の化粧料は少なくとも1種の(B)カルボン酸変性シリコーンを含む。
特開2002-114849号公報に記載の、以下のいずれかのカルボキシル基含有有機基を有するオルガノポリシロキサン:
特表2005-524747号公報に記載の以下のカルボキシル基含有有機基を有するオルガノポリシロキサン:
R’は、水素原子、又は1~30個の炭素原子を有するアルキル基を表し、
Eは、存在しないか、又は1~5個の炭素原子、好ましくは1~3個の炭素原子を有し、任意に、1~30個の炭素原子を有する1個以上のアルキル基によって置換されたアルキレン残基であり;Mは、水素原子である)、
特開2009-263643号公報に記載の、以下の平均組成式で表されるカルボキシル基含有有機基を有するオルガノポリシロキサン:
R2は下記式(2)で表される基であり、cが0の場合にはR2は該オルガノポリシロキサンの少なくとも一の末端に結合されており、
R3は下記式(3)で表される基であり、
本発明の化粧料は少なくとも1種の(C)塩基性化合物を含む。
本発明の化粧料は(D)水を含む。水は、本発明の化粧料中において水相を形成する。
本発明の化粧料は少なくとも1種の(G)多価アルコールを含むことができる。
本発明の化粧料は少なくとも1種の(E)油剤を含むことができる。
本発明の化粧料は少なくとも1種の(F)高級脂肪酸の塩を含むことができる。
本発明の化粧料は少なくとも1種の(H)水溶性増粘剤を含むことができる。
本発明の化粧料には、本発明の効果を妨げない範囲で、通常の化粧料に使用されるその他の成分:例えば、紫外線吸収剤、有機樹脂、親水性粉体、(G)成分以外の保湿剤、(H)成分以外の増粘剤、防腐剤、抗菌剤、香料、塩類、酸化防止剤、(C)成分以外のpH調整剤、キレート剤、清涼剤、抗炎症剤、生理活性成分(美白剤、細胞賦活剤、肌荒れ改善剤、血行促進剤、皮膚収斂剤、抗脂漏剤等)、ビタミン類、アミノ酸類、核酸、ホルモン、包接化合物等を添加することができる。その他の成分は特に限定されるものではない。
本発明の化粧料の製造工程は任意であり、前記各成分を混合して、(A)疎水性粉体、(B)カルボン酸変性シリコーン、(C)塩基性化合物及び(D)水からなり、(A)疎水性粉体が水相中に分散した化粧料を調製できる限り、特に限定されない。
本発明の化粧料は、クリーム状、ゲル状、乳液状、液状のいずれの形態であってもよい、本発明の化粧料は、例えば、乳液、クリーム、美容液等の基礎化粧料、下地化粧料、サンスクリーン剤、ファンデーション、アイシャドー、アイライナー、水おしろい等のメイクアップ化粧料として使用することができ、また毛髪・頭皮用のサンスクリーン剤、一時染毛剤等として使用することもできる。
イソステアリン酸2.5gと水酸化カリウム0.02gを100gの精製水に溶解しpH7.9のイソステアリン酸カリウム溶液を調製し、女性パネリストの前腕内側(塗布前のpHは5.4)及び人工皮膚(バイオスキンプレート;ビューラックス社製)上にゴム製指サックを装着した指で、0.5mg/cm2の塗布量で塗布し、30分後のpHを皮膚用pH計スキンチェッカーMJ-120(佐藤商事社製)を用いて測定した。結果を下記表1に示す。
下記表2に示す各成分を以下の製造方法により混合し水中油型乳化化粧料を得た。
成分(1)~(3)を加熱・混合し油相を形成した。また、成分(4)~(9)を加熱・混合し水相を形成した。油相と水相を規定温度(80℃)にて混合・乳化した。乳化物に成分(10)~(14)を予め混合したものを添加・混合し、次いで成分(15)を混合し、室温まで冷却し水中油型乳化化粧料を得た。なお、成分(14)の未処理の酸化チタンの量は成分(13)の疎水性粉体である酸化チタンの量と同一となるように調整された。
成分(1)~(3)を加熱・混合し油相を形成した。また、成分(4)~(10)を加熱・混合し水相を形成した。油相と水相を規定温度(80℃)にて混合・乳化した。乳化物に成分(11)~(14)を予め混合したものを添加・混合し、次いで成分(15)を混合し、室温まで冷却し水中油型乳化化粧料を得た。
カルボン酸変性シリコーンとしては、下記構造式(A1)で表されるものを使用した。なお、構造式(A1)のカルボン酸変性シリコーンは、側鎖部分がSiH(CH3)Oで示されるメチルハイドロジェンシロキシ単位のみからなり、分子鎖両末端がトリメチルシロキシ基で封鎖されたメチルハイドロジェンポリシロキサンとウンデシレン酸を、ケイ素原子結合水素原子に対するウンデシレン酸のモル比が1以上となる量で、白金触媒の存在下、ヒドロシリル化反応させることにより合成することができる。
20名の女性パネリストの顔面に評価対象(下地化粧料)を塗布し、塗布時の使用感(みずみずしさ、さっぱり感)について、下記の基準に従って官能評価を行った。
◎:20名中16名以上が、みずみずしい、さっぱりしていると回答
○:20名中11~15名が、みずみずしい、さっぱりしていると回答
△:20名中6~10名が、みずみずしい、さっぱりしていると回答
×:20名中5名以下が、みずみずしい、さっぱりしていると回答
評価対象を50℃の恒温槽中で30日間保管し、疎水性粉体の分散状態を目視観察し、下記の基準に従って、評価した。
◎:30日間保管しても疎水性粉体が均一に分散している
○:30日間経過後には疎水性粉体の凝集が生じる
△:15日間経過後に疎水性粉体の凝集が生じる
×:調製直後から疎水性粉体が凝集したままで水相に分散しない
20名の女性パネリストの前腕内側に評価対象を0.5mg/cm2の塗布量で均一に塗布し、30分後に、精製水の水滴を静かに滴下して、表面に形成された水滴を写真撮影して、その形状に基づいて接触角をθ/2法で測定し、下記の基準で評価した。
◎:接触角の平均値が55°以上
○:接触角の平均値が40°以上55°未満
△:接触角の平均値が20°以上40°未満
×:接触角の平均値が20°未満
下記表3に示す各成分を以下の製造方法により混合し水中油型乳化化粧料を得た。
A相(成分1~5)を混合し、予め混合したB相(成分6~12)を添加し、乳化した。乳化後、C相(成分13~19)を添加・混合した。更に、予め混合したD相(成分20~22)を添加・混合した後、E相(成分23)を添加・混合・脱気して、水中油型乳化化粧料を得た。
下記表4に示す各成分を以下の製造方法により混合し水性ファンデーションを得た。
(注7)商品名:SA-レッドR-516PS(100%)(三好化成株式会社製)
(注8)商品名:SA-イエローLL-100P(100%)(三好化成株式会社製)
(注9)商品名:SA-ブラックBL-100P(100%)(三好化成株式会社製)
20名の女性パネリストの顔面に評価対象(下地化粧料)を塗布し、塗布時の使用感(みずみずしさ、さっぱり感)について、下記の基準に従って官能評価を行った。
◎:20名中16名以上が、みずみずしい、さっぱりしていると回答
○:20名中11~15名が、みずみずしい、さっぱりしていると回答
△:20名中6~10名が、みずみずしい、さっぱりしていると回答
×:20名中5名以下が、みずみずしい、さっぱりしていると回答
評価対象を50℃の恒温槽中で30日間保管し、疎水性粉体の再分散性を目視観察し、下記の基準に従って、評価した。
◎:30日間保管しても10回以内の振とうにより速やかに均一に分散する。
○:30日間保管しても30回以内の振とうにより均一に分散する。
△:30日間保管後30回振とうしても均一に分散しない。
×:調製直後から疎水性粉体の凝集したまま水相に分散しない。
20名の女性パネリストの前腕内側に評価対象を0.5mg/cm2の塗布量で均一に塗布し、30分後に、pH7.0の水の水滴を静かに滴下して、表面に形成された水滴を写真撮影して、その形状に基づいて接触角をθ/2法で測定し、下記の基準で評価した。
◎:接触角の平均値が55°以上
○:接触角の平均値が40°以上55°未満
△:接触角の平均値が20°以上40°未満
×:接触角の平均値が20°未満
下記表5に示す各成分を以下の製造方法により混合し疎水化微粒子酸化チタンの水性分散体を得た。
成分4~5を混合し、これに成分1~3の混合物を加え混合・分散し、水性分散体を得た。
実施例5の水性分散体を使用し、下記表6に示す各成分を以下の製造方法により混合し水中油型乳化化粧料を得た。
成分(1)~(3)を加熱・混合し油相を形成した。また、成分(4)~(9)を加熱・混合し水相を形成した。油相と水相を規定温度(80℃)にて混合・乳化した。乳化物に成分(10)を添加・混合し、次いで成分(11)を混合し、室温まで冷却し水中油型乳化化粧料を得た。
Claims (21)
- 下記の成分:
(A)疎水性粉体、
(B)カルボン酸変性シリコーン、
(C)塩基性化合物、及び、
(D)水
を含み、前記(A)疎水性粉体が水相中に分散していることを特徴とする化粧料。 - 前記(A)疎水性粉体を化粧料の全質量に対して1~40質量%の範囲で含む、請求項1記載の化粧料。
- 前記(B)カルボン酸変性シリコーンを疎水性粉体10質量部に対して0.01~20質量部の範囲で含む、請求項1又は2記載の化粧料。
- 前記(B)カルボン酸変性シリコーンが、下記構造式(A):
Rcは、一般式:-R1-(OR2)p-(O)w-R3-COOH(R1 は炭素数2~22の直鎖又は分岐鎖のアルキレン基を表し、R2 は炭素数2~4の直鎖又は分岐鎖のアルキレン基を表し、R3 は結合手(-)又は炭素数1~22の直鎖若しくは分岐鎖のアルキレン基を表し、pは0~200の数を表し、wは0又は1の数を表す)で示されるカルボキシル基含有有機基を表し、
Rは、同一又は異なり、炭素数1~22のアルキル基若しくはアルコキシ基又はフェニル基を表し、
R´は、Rc又はRであり、
a、bは各々0以上の範囲の数であり、a+bは0~1000の範囲の数である。但し、b=0のとき、R´の少なくとも一方はRcである。}
で表される、請求項1乃至3のいずれかに記載の化粧料。 - 前記構造式(A)において、b>aであり、b>0であり、a+bが1~500の範囲の数である、請求項4記載の化粧料。
- 前記(D)水を化粧料の全質量に対して20~95質量%の範囲で含む、請求項1乃至5のいずれかに記載の化粧料。
- pHが7.1~9.5である、請求項1乃至6のいずれかに記載の化粧料。
- (G)多価アルコールを更に含む、請求項1乃至7のいずれかに記載の化粧料。
- (E)油剤を更に含む、請求項1乃至8のいずれかに記載の化粧料。
- 前記(E)油剤を化粧料の全質量に対して3~60質量%の範囲で含む、請求項9記載の化粧料。
- (F)高級脂肪酸の塩を更に含む、請求項1乃至10のいずれかに記載の化粧料。
- 前記高級脂肪酸が、イソステアリン酸、ヘキシルデカン酸及びオレイン酸からなる群から選択される1種以上である、請求項11記載の化粧料。
- 前記(F)高級脂肪酸の塩を0.1~12質量%の範囲で含む、請求項11又は12記載の化粧料。
- (H)水溶性増粘剤を更に含む、請求項1乃至13のいずれかに記載の化粧料。
- 前記(B)カルボン酸変性シリコーン及び前記(F)高級脂肪酸の塩以外の界面活性剤を含まない請求項9乃至12のいずれかに記載の化粧料。
- 水中油型乳化組成物の形態である、請求項9乃至15のいずれかに記載の化粧料。
- (A)疎水性粉体、(B)カルボン酸変性シリコーン、(C)塩基性化合物及び(D)水を予め混合することにより、前記(A)疎水性粉体が水相中に分散した水性分散体を製造する工程を含む、請求項1乃至16のいずれかに記載の化粧料の製造方法。
- (A)疎水性粉体、(B)カルボン酸変性シリコーン、(C)塩基性化合物、(D)水及び(G)多価アルコールを予め混合することにより、前記(A)疎水性粉体が水相中に分散した水性分散体を製造する工程を含む、請求項8乃至16のいずれかに記載の化粧料の製造方法。
- 前記水性分散体のpHが6.5~14.0の範囲にある、請求項17又は請求項18記載の化粧料の製造方法。
- (G)多価アルコール、(A)疎水性粉体及び(B)カルボン酸変性シリコーン予め混合して混合物を得る工程、並びに、
(C)塩基性化合物及び(D)水を、前記混合物と混合する工程
を含む、請求項8乃至16のいずれかに記載の化粧料の製造方法。 - (G)多価アルコール、(A)疎水性粉体及び(B)カルボン酸変性シリコーン予め混合する工程して得られた混合物と、(C)塩基性化合物及び(D)水を混合することにより、疎水性粉体が水相中に分散したpHが6.5~14.0の範囲にある水性分散体を製造する工程を含む、請求項8乃至16のいずれかに記載の化粧料の製造方法。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/303,743 US10130579B2 (en) | 2014-04-15 | 2015-04-08 | Cosmetic material |
| CN201580019520.XA CN106163495B (zh) | 2014-04-15 | 2015-04-08 | 化妆品 |
| KR1020167028758A KR102390517B1 (ko) | 2014-04-15 | 2015-04-08 | 화장료 |
| EP15779652.5A EP3132789B1 (en) | 2014-04-15 | 2015-04-08 | Cosmetic |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014-084003 | 2014-04-15 | ||
| JP2014084003A JP6586554B2 (ja) | 2014-04-15 | 2014-04-15 | 化粧料 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015159773A1 true WO2015159773A1 (ja) | 2015-10-22 |
Family
ID=54323984
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/060963 WO2015159773A1 (ja) | 2014-04-15 | 2015-04-08 | 化粧料 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US10130579B2 (ja) |
| EP (1) | EP3132789B1 (ja) |
| JP (1) | JP6586554B2 (ja) |
| KR (1) | KR102390517B1 (ja) |
| CN (1) | CN106163495B (ja) |
| WO (1) | WO2015159773A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022075372A1 (ja) | 2020-10-09 | 2022-04-14 | ダウ・東レ株式会社 | カルボン酸共変性オルガノポリシロキサンおよびその用途 |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101819467B1 (ko) * | 2016-08-26 | 2018-01-18 | 코스맥스 주식회사 | 압전 화장료 조성물 |
| KR20190037454A (ko) * | 2017-09-29 | 2019-04-08 | (주)아모레퍼시픽 | 프라이밍 화장료 조성물 및 이의 제조방법 |
| KR102613596B1 (ko) | 2018-01-29 | 2023-12-13 | 이시하라 산교 가부시끼가이샤 | 이산화 티탄 수성 분산체 및 이의 제조 방법 |
| JP7284148B2 (ja) * | 2018-03-30 | 2023-05-30 | 株式会社 資生堂 | 水中油滴型の微細エマルション型化粧料 |
| JP7371854B2 (ja) | 2018-08-15 | 2023-10-31 | ジェイオーコスメティックス株式会社 | 水中油型乳化化粧料 |
| EP3838254A4 (en) * | 2018-08-15 | 2022-05-11 | Dow Toray Co., Ltd. | AQUEOUS DISPERSION COMPOSITION AND ITS USE |
| CN112804988B (zh) * | 2018-08-15 | 2023-07-18 | 陶氏东丽株式会社 | 化妆料 |
| CN112789022B (zh) * | 2018-08-15 | 2023-08-25 | 陶氏东丽株式会社 | 水包油型乳化化妆料 |
| JP7348189B2 (ja) * | 2018-08-15 | 2023-09-20 | ダウ・東レ株式会社 | 水中油型乳化化粧料 |
| JP7371853B2 (ja) | 2019-04-10 | 2023-10-31 | ジェイオーコスメティックス株式会社 | 水中油型クレンジング化粧料 |
| EP3960152A4 (en) * | 2019-04-24 | 2023-06-07 | Dow Toray Co., Ltd. | COSMETIC PREPARATIONS |
| JP7376147B2 (ja) | 2019-07-31 | 2023-11-08 | 三好化成株式会社 | 化粧料の製造方法、及び化粧料 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001089347A (ja) * | 1999-09-21 | 2001-04-03 | Noevir Co Ltd | 疎水性粉体含有水中油型乳化組成物 |
| JP2004026669A (ja) * | 2002-06-21 | 2004-01-29 | Kao Corp | 皮膚化粧料 |
| WO2009025146A1 (ja) * | 2007-07-26 | 2009-02-26 | Shiseido Company Ltd. | ゲル組成物及び化粧料 |
| JP2010138074A (ja) * | 2007-04-13 | 2010-06-24 | Dai Ichi Kogyo Seiyaku Co Ltd | 水系分散体及び水系化粧料 |
| JP2013121947A (ja) * | 2011-11-11 | 2013-06-20 | Shiseido Co Ltd | 水中油型乳化日焼け止め化粧料 |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0753646B2 (ja) | 1989-01-31 | 1995-06-07 | 東レ・ダウコーニング・シリコーン株式会社 | 化粧料 |
| US5596061A (en) | 1993-12-28 | 1997-01-21 | Mona Industries, Inc. | Organosilicone having a carboxyl functional group thereon |
| JP3178970B2 (ja) | 1994-06-30 | 2001-06-25 | 東レ・ダウコーニング・シリコーン株式会社 | 制汗・消臭剤 |
| JPH0812546A (ja) | 1994-06-30 | 1996-01-16 | Toray Dow Corning Silicone Co Ltd | 日焼け防止用化粧料 |
| JP3354296B2 (ja) | 1994-06-30 | 2002-12-09 | 東レ・ダウコーニング・シリコーン株式会社 | メーキャップ化粧料 |
| JPH08109263A (ja) | 1994-10-12 | 1996-04-30 | Kao Corp | カルボン酸多価金属塩変性オルガノ(ポリ)シロキサン、その製造法並びにこれを含有するゲル化剤及び化粧料 |
| JP3607404B2 (ja) | 1996-03-08 | 2005-01-05 | 東レ・ダウコーニング・シリコーン株式会社 | シリコーンゴムサスペンジョンおよびその製造方法 |
| US5939478A (en) | 1997-07-21 | 1999-08-17 | Dow Corning Corporation | Silicone polyether stabilized silicone latex solvent thickening |
| US6238656B1 (en) | 1998-06-23 | 2001-05-29 | Dow Corning Toray Silicone Corporation, Ltd. | Cosmetic raw materials, cosmetic products, and methods of manufacturing cosmetic products |
| JP4025454B2 (ja) | 1998-06-23 | 2007-12-19 | 東レ・ダウコーニング株式会社 | 化粧品原料、化粧品、および化粧品の製造方法 |
| JP2002114849A (ja) | 2000-10-05 | 2002-04-16 | Kao Corp | オルガノポリシロキサン |
| US6887836B2 (en) | 2002-05-09 | 2005-05-03 | The Procter & Gamble Company | Home care compositions comprising a dicarboxy functionalized polyorganosiloxane |
| JP2004091423A (ja) | 2002-09-03 | 2004-03-25 | Yuskin Seiyaku Kk | 水中油型乳化化粧料 |
| JP5239122B2 (ja) * | 2006-03-03 | 2013-07-17 | 信越化学工業株式会社 | オルガノポリシロキサン粉体処理剤及び該処理剤により処理された粉体並びに該粉体を含有する化粧料 |
| JP4865388B2 (ja) * | 2006-04-07 | 2012-02-01 | 株式会社 資生堂 | 表面処理酸化亜鉛粉体及びこれを含有する化粧料 |
| JP4116666B1 (ja) | 2007-07-23 | 2008-07-09 | 壽義 梶田 | ミシン目付きロールペーパー用ロールペーパーホルダー |
| US20110182846A1 (en) * | 2007-08-10 | 2011-07-28 | Shiseido Company Ltd. | Surface Treating Agent, Surface-Treated Powder, And Cosmetic |
| JP5353357B2 (ja) | 2008-04-03 | 2013-11-27 | 信越化学工業株式会社 | カルボキシル基を有するオルガノポリシロキサン |
| JP5368394B2 (ja) | 2010-08-11 | 2013-12-18 | 信越化学工業株式会社 | 新規オルガノポリシロキサン化合物およびそれを含む化粧料 |
| JP5197723B2 (ja) * | 2010-11-26 | 2013-05-15 | 株式会社 資生堂 | 日焼け止め水中油型乳化化粧料 |
| JP5554308B2 (ja) | 2011-10-27 | 2014-07-23 | 株式会社 資生堂 | 日焼け止め水中油型乳化化粧料 |
| JP6369887B2 (ja) * | 2011-12-27 | 2018-08-08 | 東レ・ダウコーニング株式会社 | 新規な共変性オルガノポリシロキサン、それを含有してなる処理剤および外用剤 |
| JP5357280B2 (ja) * | 2012-01-16 | 2013-12-04 | 株式会社 資生堂 | 油中水型乳化皮膚化粧料 |
| WO2013115099A1 (ja) * | 2012-01-30 | 2013-08-08 | 株式会社 資生堂 | メーキャップ化粧料 |
-
2014
- 2014-04-15 JP JP2014084003A patent/JP6586554B2/ja active Active
-
2015
- 2015-04-08 CN CN201580019520.XA patent/CN106163495B/zh active Active
- 2015-04-08 WO PCT/JP2015/060963 patent/WO2015159773A1/ja active Application Filing
- 2015-04-08 US US15/303,743 patent/US10130579B2/en active Active
- 2015-04-08 EP EP15779652.5A patent/EP3132789B1/en active Active
- 2015-04-08 KR KR1020167028758A patent/KR102390517B1/ko active IP Right Grant
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001089347A (ja) * | 1999-09-21 | 2001-04-03 | Noevir Co Ltd | 疎水性粉体含有水中油型乳化組成物 |
| JP2004026669A (ja) * | 2002-06-21 | 2004-01-29 | Kao Corp | 皮膚化粧料 |
| JP2010138074A (ja) * | 2007-04-13 | 2010-06-24 | Dai Ichi Kogyo Seiyaku Co Ltd | 水系分散体及び水系化粧料 |
| WO2009025146A1 (ja) * | 2007-07-26 | 2009-02-26 | Shiseido Company Ltd. | ゲル組成物及び化粧料 |
| JP2013121947A (ja) * | 2011-11-11 | 2013-06-20 | Shiseido Co Ltd | 水中油型乳化日焼け止め化粧料 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP3132789A4 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2022075372A1 (ja) | 2020-10-09 | 2022-04-14 | ダウ・東レ株式会社 | カルボン酸共変性オルガノポリシロキサンおよびその用途 |
| KR20230084532A (ko) | 2020-10-09 | 2023-06-13 | 다우 도레이 캄파니 리미티드 | 카복실산 공변성 오가노폴리실록산 및 그의 용도 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6586554B2 (ja) | 2019-10-09 |
| US20170035681A1 (en) | 2017-02-09 |
| KR102390517B1 (ko) | 2022-04-27 |
| EP3132789A1 (en) | 2017-02-22 |
| US10130579B2 (en) | 2018-11-20 |
| EP3132789B1 (en) | 2024-01-17 |
| JP2015203026A (ja) | 2015-11-16 |
| CN106163495B (zh) | 2020-04-14 |
| KR20160136349A (ko) | 2016-11-29 |
| EP3132789A4 (en) | 2017-12-13 |
| CN106163495A (zh) | 2016-11-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6586554B2 (ja) | 化粧料 | |
| JP6615935B2 (ja) | 化粧料 | |
| JP5841348B2 (ja) | 新規なオルガノポリシロキサン、それを含む界面活性剤、エマルジョン組成物、粉体処理剤、油性原料の増粘剤、ゲル化剤、ゲル状組成物、化粧料用原料並びに、これらを含む外用剤及び化粧料 | |
| JP5841347B2 (ja) | 新規なオルガノポリシロキサン、それを含む界面活性剤、エマルジョン組成物、粉体処理剤、油性原料の増粘剤、ゲル化剤、ゲル状組成物、化粧料用原料並びに、これらを含む外用剤及び化粧料 | |
| JP7371854B2 (ja) | 水中油型乳化化粧料 | |
| JP2004169015A (ja) | 粉体組成物及び油中粉体分散物.並びにそれらを有する化粧料 | |
| JP5394424B2 (ja) | 固形乳化化粧料 | |
| WO2001092376A1 (fr) | Compose de silicone et preparation cosmetique | |
| JP2002038013A (ja) | 粉体組成物、その油中粉体分散物及びそれらを含有する化粧料 | |
| EP3476382B1 (en) | Agent for treating powder for cosmetic, powder for cosmetic, and cosmetic formulated using said powder | |
| JP2011088832A (ja) | 化粧料 | |
| KR20130108150A (ko) | 신규 오르가노폴리실록산, 이것을 포함하는 화장료 및 오르가노폴리실록산의 제조 방법 | |
| JP3727901B2 (ja) | 水中油型乳化化粧料 | |
| WO2020036064A1 (ja) | 水中油型乳化化粧料 | |
| JP2012162700A (ja) | ペースト状組成物及びこれを含有する化粧料 | |
| JP7422353B2 (ja) | 水中油型乳化化粧料 | |
| JP2004269418A (ja) | 化粧料 | |
| JP7422354B2 (ja) | 化粧料 | |
| JP7048480B2 (ja) | マイクロエマルション組成物及びその硬化物、並びに、それを含んだ化粧料 | |
| US20220183956A1 (en) | Oil-in-water cleansing cosmetic composition | |
| JP6923487B2 (ja) | 分散性粉体 | |
| JP2015113344A (ja) | 化粧料 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15779652 Country of ref document: EP Kind code of ref document: A1 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2015779652 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15303743 Country of ref document: US Ref document number: 2015779652 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 20167028758 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |