WO2014147782A1 - 排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 - Google Patents
排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 Download PDFInfo
- Publication number
- WO2014147782A1 WO2014147782A1 PCT/JP2013/058015 JP2013058015W WO2014147782A1 WO 2014147782 A1 WO2014147782 A1 WO 2014147782A1 JP 2013058015 W JP2013058015 W JP 2013058015W WO 2014147782 A1 WO2014147782 A1 WO 2014147782A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- hydrophilic surface
- water
- surface treatment
- group
- hydrophilic
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D129/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal, or ketal radical; Coating compositions based on hydrolysed polymers of esters of unsaturated alcohols with saturated carboxylic acids; Coating compositions based on derivatives of such polymers
- C09D129/02—Homopolymers or copolymers of unsaturated alcohols
- C09D129/04—Polyvinyl alcohol; Partially hydrolysed homopolymers or copolymers of esters of unsaturated alcohols with saturated carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/07—Aldehydes; Ketones
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/09—Carboxylic acids; Metal salts thereof; Anhydrides thereof
- C08K5/092—Polycarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/34—Heterocyclic compounds having nitrogen in the ring
- C08K5/3467—Heterocyclic compounds having nitrogen in the ring having more than two nitrogen atoms in the ring
- C08K5/3477—Six-membered rings
- C08K5/3492—Triazines
- C08K5/34922—Melamine; Derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/541—Silicon-containing compounds containing oxygen
- C08K5/5415—Silicon-containing compounds containing oxygen containing at least one Si—O bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/541—Silicon-containing compounds containing oxygen
- C08K5/5415—Silicon-containing compounds containing oxygen containing at least one Si—O bond
- C08K5/5419—Silicon-containing compounds containing oxygen containing at least one Si—O bond containing at least one Si—C bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/541—Silicon-containing compounds containing oxygen
- C08K5/5425—Silicon-containing compounds containing oxygen containing at least one C=C bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/541—Silicon-containing compounds containing oxygen
- C08K5/5435—Silicon-containing compounds containing oxygen containing oxygen in a ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/54—Silicon-containing compounds
- C08K5/544—Silicon-containing compounds containing nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
- C08L101/02—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
- C08L101/025—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups containing nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
- C08L101/02—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
- C08L101/06—Compositions of unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups containing oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L101/00—Compositions of unspecified macromolecular compounds
- C08L101/12—Compositions of unspecified macromolecular compounds characterised by physical features, e.g. anisotropy, viscosity or electrical conductivity
- C08L101/14—Compositions of unspecified macromolecular compounds characterised by physical features, e.g. anisotropy, viscosity or electrical conductivity the macromolecular compounds being water soluble or water swellable, e.g. aqueous gels
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/02—Homopolymers or copolymers of acids; Metal or ammonium salts thereof
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/24—Homopolymers or copolymers of amides or imides
- C09D133/26—Homopolymers or copolymers of acrylamide or methacrylamide
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D201/00—Coating compositions based on unspecified macromolecular compounds
- C09D201/02—Coating compositions based on unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D201/00—Coating compositions based on unspecified macromolecular compounds
- C09D201/02—Coating compositions based on unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
- C09D201/025—Coating compositions based on unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups containing nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D201/00—Coating compositions based on unspecified macromolecular compounds
- C09D201/02—Coating compositions based on unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups
- C09D201/06—Coating compositions based on unspecified macromolecular compounds characterised by the presence of specified groups, e.g. terminal or pendant functional groups containing oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D5/00—Coating compositions, e.g. paints, varnishes or lacquers, characterised by their physical nature or the effects produced; Filling pastes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/61—Additives non-macromolecular inorganic
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/60—Additives non-macromolecular
- C09D7/63—Additives non-macromolecular organic
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D7/00—Features of coating compositions, not provided for in group C09D5/00; Processes for incorporating ingredients in coating compositions
- C09D7/40—Additives
- C09D7/66—Additives characterised by particle size
- C09D7/67—Particle size smaller than 100 nm
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F28—HEAT EXCHANGE IN GENERAL
- F28F—DETAILS OF HEAT-EXCHANGE AND HEAT-TRANSFER APPARATUS, OF GENERAL APPLICATION
- F28F13/00—Arrangements for modifying heat-transfer, e.g. increasing, decreasing
- F28F13/18—Arrangements for modifying heat-transfer, e.g. increasing, decreasing by applying coatings, e.g. radiation-absorbing, radiation-reflecting; by surface treatment, e.g. polishing
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F28—HEAT EXCHANGE IN GENERAL
- F28F—DETAILS OF HEAT-EXCHANGE AND HEAT-TRANSFER APPARATUS, OF GENERAL APPLICATION
- F28F17/00—Removing ice or water from heat-exchange apparatus
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F28—HEAT EXCHANGE IN GENERAL
- F28F—DETAILS OF HEAT-EXCHANGE AND HEAT-TRANSFER APPARATUS, OF GENERAL APPLICATION
- F28F19/00—Preventing the formation of deposits or corrosion, e.g. by using filters or scrapers
- F28F19/02—Preventing the formation of deposits or corrosion, e.g. by using filters or scrapers by using coatings, e.g. vitreous or enamel coatings
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F28—HEAT EXCHANGE IN GENERAL
- F28F—DETAILS OF HEAT-EXCHANGE AND HEAT-TRANSFER APPARATUS, OF GENERAL APPLICATION
- F28F19/00—Preventing the formation of deposits or corrosion, e.g. by using filters or scrapers
- F28F19/02—Preventing the formation of deposits or corrosion, e.g. by using filters or scrapers by using coatings, e.g. vitreous or enamel coatings
- F28F19/04—Preventing the formation of deposits or corrosion, e.g. by using filters or scrapers by using coatings, e.g. vitreous or enamel coatings of rubber; of plastics material; of varnish
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/34—Silicon-containing compounds
- C08K3/36—Silica
-
- F—MECHANICAL ENGINEERING; LIGHTING; HEATING; WEAPONS; BLASTING
- F28—HEAT EXCHANGE IN GENERAL
- F28F—DETAILS OF HEAT-EXCHANGE AND HEAT-TRANSFER APPARATUS, OF GENERAL APPLICATION
- F28F2245/00—Coatings; Surface treatments
- F28F2245/02—Coatings; Surface treatments hydrophilic
Definitions
- the present invention relates to a hydrophilic surface treatment agent, a hydrophilized surface treatment method and a hydrophilic surface-treated heat of an aluminum-containing metal heat exchanger (hereinafter simply referred to as a heat exchanger) excellent in drainage of condensed water. It relates to an exchanger.
- water droplets agglomerated between the fins induce corrosion of the aluminum forming the fins, causing white powdered aluminum oxide to adhere to the fin surface, or leaving the heat exchanger surface moist for a long time.
- bacteria and the like easily propagate on the surface.
- the white powder of aluminum oxide adhering to the fins and the water droplets accumulated in the fin gaps are scattered in the room by the blower of the heat exchanger and give the user an unpleasant feeling.
- an anticorrosion film for example, a phosphoric acid chromate film, a chromic acid chromate film, or a non-chromate film
- a hydrophilic film is formed on the anticorrosion film by using a hydrophilic inorganic compound such as water glass or colloidal silica, and an organic compound such as a surfactant and a water-soluble resin, alone or in combination.
- a hydrophilic film may be directly formed without forming the anticorrosion film.
- Japanese Patent Laid-Open No. 9-296121 discloses a hydrophilizing agent for a metal material containing a water-soluble polymer having an acetal structure and a hydrophilic polymer having at least one hydrophilic group.
- JP-A-11-343450 discloses a hydrophilic treatment composition containing a water-soluble polymer, a trivalent chromium compound, and a zirconium or titanium compound.
- the hydrophilic film necessarily has excellent drainage performance, and development of a hydrophilic film having further excellent drainage performance is desired. Furthermore, since the above-mentioned hydrophilic film significantly reduces drainage after aged use, it is desired to develop a hydrophilic film that exhibits stable drainage for a long period of time.
- WO2009 / 044912 contains a colloidal silica sol (A), an acrylic polymer (B), a reactive silane coupling agent (C), and a curing agent, and has a mass ratio of ⁇ (A) / (B) ⁇ .
- a hydrophilic coating agent characterized in that 5/95 to 95/5 and ⁇ (A + B) / (C) ⁇ is 30/70 to 95/5 is disclosed.
- the above hydrophilic film exhibits antifogging properties due to high hydrophilicity, hardness and durability, but does not exhibit sufficient performance with respect to drainage.
- Japanese Patent Application Laid-Open No. 2005-162533 discloses a method for producing modified colloidal silica, characterized in that colloidal silica obtained by hydrolysis and condensation of a hydrolyzable silicon compound is modified with a modifying agent. Yes.
- the above production method aims to improve long-term dispersion stability by modifying colloidal silica.
- the silane coupling agent exceeds the weight ratio of 0.03 with respect to colloidal silica, problems such as an increase in secondary particle system, formation of aggregates, gelation, etc. occur.
- the amount of silane coupling agent (wherein organoalkoxysilane is a kind of silane coupling agent) relative to colloidal silica does not correspond to sufficient surface modification, and exhibits drainage. It is not enough for this.
- Japanese Patent Application Laid-Open No. 7-233270 discloses a polymer obtained by emulsion polymerization of an acrylic monomer, a polymerizable silane coupling agent, and colloidal silica, and 20 to 70% by weight per total solid content.
- the weight ratio of the silane coupling agent to colloidal silica is 0.5 at maximum, and the surface modification with the silane coupling agent is insufficient.
- a silane coupling agent having a hydrophobic group in the molecule is used, and if the weight ratio is 0.5 or more, the silica surface is covered with the hydrophobic coupling agent. Is lost and the hydrophilicity of the coating film surface cannot be obtained. Therefore, although there is no problem in the water drop property in an agricultural film or the like, the drainage property of the aluminum-containing metal heat exchanger having a complicated structure is insufficient.
- Japanese Patent Application Laid-Open No. 2001-323250 discloses an aqueous medium containing silica fine particles and a vinyl alcohol polymer, the silica fine particles being coated with the vinyl alcohol polymer, and coated particles having an average particle diameter of 5 to 1000 nm.
- a hydrophilizing agent dispersed therein is disclosed.
- the above method provides a hydrophilic treatment method for a heat exchanger excellent in hydrophilic sustainability and deodorization.
- the silica fine particles are coated with a vinyl alcohol polymer, the silica fine particles have excellent deodorizing properties, but the drainage is inferior to colloidal silica surface-modified with a silane coupling agent.
- an alkali silicate, a low molecular organic compound having a carbonyl group, an acrylamide copolymer and a silane coupling agent are the main components, and the solid content of the acrylamide copolymer is 25-30.
- a hydrophilic film forming agent for aluminum or its fin material by weight% is disclosed.
- the present invention is intended to solve the above-mentioned problems of the prior art. That is, it is intended to provide a hydrophilic surface treatment agent that can be used for hydrophilic surface treatment of a heat exchanger and can maintain good drainage, and a hydrophilic surface treatment method and a hydrophilic surface-treated heat exchanger. .
- the present inventors have found that evaluation based on such dynamic behavior is important from the viewpoint of improving drainage in an application of an aluminum-containing metal heat exchanger. And, as a result of intensive studies through an evaluation method called a sliding angle at which a water droplet moves by tilting a sample, the present inventors have performed a hydrophilic surface treatment with a hydrophilic surface treatment agent containing a specific component at a specific mixing ratio. Thus, the inventors have found that the above problems can be solved, and have completed the present invention.
- the hydrophilic surface treatment agent excellent in drainage of the condensed water of the present invention is a water-soluble resin having at least one functional group of amide group, hydroxyl group and carboxyl group, or a water-soluble resin having an amide bond in the skeleton.
- the particle size of the colloidal silica (B) used is more preferably in the range of 1 to 100 nm, and still more preferably in the range of 2 to 20 nm.
- the organoalkoxysilane (C) contains a glycidyl group.
- the hydrophilic surface treatment agent has a pH in the range of 1 to 7 and is used for the hydrophilic surface treatment.
- the water-soluble resin (A) is more preferably polyvinyl alcohol or a copolymer thereof.
- the crosslinking agent (D) used is more preferably a crosslinking agent containing at least one functional group selected from the group consisting of a carboxyl group, an aldehyde group, a glycidyl group, a methylol ether group and an isocyanate group.
- the hydrophilic surface treatment agent may contain a metal compound (F) containing at least one metal element selected from the group consisting of V, Ti, Zr, and Cr in order to improve corrosion resistance.
- F metal compound
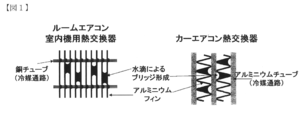
- FIG. 1 is a diagram showing an example of a heat exchanger to which the present invention is applied.
- the aluminum-containing metal used as the material for the heat exchanger of the present invention is aluminum and aluminum alloys, such as aluminum-magnesium alloys, aluminum-silicon alloys and aluminum-manganese alloys. The material is then molded into tubes, fins, and hollow plates used in heat exchangers such as air conditioners.
- Heat exchangers formed from these parts include heat exchangers for automotive air conditioners, in which the fins and the tubes through which the refrigerant passes are both made of aluminum-containing metal, and expanded pipes in combination with processed aluminum sheets and copper pipes.
- An example is a heat exchanger of an air conditioner for home appliances that joins fins and pipes.
- the heat exchanger to which the present invention is applied has a well-known structure for those skilled in the art, and FIG. 1 shows an example of the well-known structure. More specifically, the heat exchanger shown in FIG. 1 is a so-called finned tube heat exchanger. And as this figure shows, in this kind of heat exchanger, the bridge
- a room air conditioner first performs a hydrophilic surface treatment on an aluminum sheet (referred to as a precoat).
- the sheet is molded into the shape of the heat exchanger in FIG.
- a car air conditioner is first formed by processing an aluminum sheet into the shape of the heat exchanger shown in FIG. This heat exchanger is subjected to a hydrophilic surface treatment (called post-coating).
- post-coating a hydrophilic surface treatment
- undercoat ⁇ Good performance can be obtained by applying a hydrophilic surface treating agent to the surface of the aluminum-containing metal substrate.
- a hydrophilic surface treating agent to the surface of the aluminum-containing metal substrate.
- it may be coated with an undercoat chemical conversion layer as a base for the hydrophilic surface treatment film.
- This undercoat chemical conversion layer is formed on the surface of the aluminum-containing metal substrate from a chemical conversion treatment, for example, a chemical conversion treatment agent containing at least one metal element selected from the group consisting of chromium, zirconium, titanium, and vanadium.
- the undercoat chemical conversion layer is preferably formed in a coating amount of 2 to 500 mg / m 2 or a thickness of 0.002 to 0.5 ⁇ m.
- the hydrophilic surface treatment agent includes a water-soluble resin having at least one functional group of an amide group, a hydroxyl group and a carboxyl group, or a water-soluble resin (A) having an amide bond in the skeleton, Colloidal silica (B), An organoalkoxysilane and / or a hydrolyzate thereof (C); A water-soluble resin (A) and a crosslinkable crosslinker (D); Water (E), and ⁇ (B) + (C) ⁇ / ⁇ (A) + (B) + (C) + (D) ⁇ is a solid content ratio (mass ratio) of 0.1 to 0.5, (C) / (B) is a hydrophilic surface treatment agent for an aluminum-containing metal heat exchanger having a fixed component ratio of 0.5 to 4.0 (mass ratio).
- Water-soluble resin (A) examples include homopolymers and copolymers of acrylamide compounds. . Copolymers include one or more copolymers of acrylamide monomers and one or more copolymers of other comonomers.
- the comonomer used here is selected from anionic, nonionic, and cationic addition polymerization monomers that can be polymerized with acrylamide, such as (meth) acrylic acid, itaconic acid, maleic acid, vinyl sulfonic acid, styrene sulfonic acid, sulfone.
- acrylamide such as (meth) acrylic acid, itaconic acid, maleic acid, vinyl sulfonic acid, styrene sulfonic acid, sulfone.
- Anionic unsaturated monomers such as ethyl acrylate, sulfoethyl methacrylate, N-methylene sulfonic acid acrylamide, 2-acrylamido-2-methylpropane sulfonic acid, acid phosphooxyethyl methacrylate or salts thereof; (meth) acrylamide, N-methylol (Meth) acrylamide, hydroxyethyl (meth) acrylate, vinylpyrrolidone, acroylmorpholine, polyethylene glycol acrylate, polyethylene glycol acrylate alkylphenyl ether
- Nonionic unsaturated monomers such as aminoethyl (meth) acrylate, N, N-dimethylaminoethyl acrylate, N-hydroxypropylaminoethyl (meth) acrylate, hydroxymethylaminoethyl methacrylate, vinylimidazole, vinylpyridine, N , N-diallylamine,
- examples of the water-soluble resin (A) include polyvinyl alcohol containing a hydroxyl group or a derivative thereof, and polyacrylic acid containing a carboxyl group or a derivative thereof.
- water-soluble resin (A) having an amide bond in the skeleton examples include water-soluble polyamide and water-soluble nylon.
- Water-soluble polyamides include basic polyamides obtained from polyalkylene polyamines and aliphatic dicarboxylic acids such as adipic acid, and epoxy-modified polyamides produced by reacting the basic polyamides with epichlorohydrin. It is done.
- water-soluble nylon examples include cationic nylon containing an amino group and nonionic nylon containing ethylene oxide.
- the water-soluble resin (A) is more preferably polyvinyl alcohol or a derivative thereof. That is, it includes partially saponified products and completely saponified products of polyvinyl acetate, and partially saponified products and completely saponified products of copolymers of vinyl acetate and other monomers, and modified products of polyvinyl alcohol. There is no particular limitation on the comonomer copolymerized with vinyl acetate.
- cationic comonomer include aminoethyl (Meth) acrylate, N-hydroxypropylaminoethyl (meth) acrylate, vinylimidazole, N, N-dimethyldiallylamine and the like can be used.
- the weight average molecular weight of polyvinyl alcohol or a derivative thereof is 5,000 to 200,000, more preferably 10,000 to 100,000, still more preferably 20,000 to 50,000. If the weight average molecular weight is less than 5,000, a sufficient network structure cannot be formed in the film and the durability cannot be exhibited. If the molecular weight exceeds 200,000, the viscosity increases, and the colloidal silica in the hydrophilic film is contained in the film. It becomes difficult to disperse uniformly and the drainage performance decreases.
- the weight average molecular weight is a weight average molecular weight measured by a GPC-LALLS method. The measuring method is shown below.
- GPC Apparatus Waters type 244 gel permeation chromatography column: Tosoh TSK-gel-GMPW (Inner diameter 8mm, length 30cm, 2) Solvent: 0.1 M Tris buffer (pH 7.9) Flow rate: 0.5 ml / min Temperature: 23 ° C Sample concentration: 0.040% Filtration: 0.45 ⁇ m Mysori disk W-25-5 made by Tosoh Injection volume: 0.2ml Detection sensitivity (differential refractive index detector): 4 times 2) LALLS Apparatus: Chromatrix KMX-6 type low angle laser light scattering photometer Temperature: 23 ° C Wavelength: 633nm Second virial coefficient ⁇ concentration: 0 mol / g Refractive index density change (dn / dc): 0.159 ml / g Filter: MILLIPORE 0.45 ⁇ m filter HAWP01300 Gain: 800mV
- the saponification degree of polyvinyl alcohol used for water-soluble resin (A) or its derivative (s) is 95 mol%, Preferably 98 mol% or more is preferable. If the degree of saponification is low and a large amount of acetyl groups are added, the solubility in water decreases. Moreover, when there are many acetyl groups, reaction with a crosslinking agent does not advance enough, but since a crosslinking degree falls, durability deteriorates and the drainage property after durability falls.
- the degree of saponification refers to a value measured according to the polyvinyl alcohol test method JIS-K-6726.
- colloidal silica (B) used is a dispersion of a silicic acid compound containing silicon and oxygen as main components, and the kind thereof is not particularly limited.
- colloidal silica obtained by a method of removing sodium, potassium, or lithium from an alkali silicate such as sodium silicate, potassium silicate, or lithium silicate by an ion exchange method to form a sol is used.
- colloidal silica obtained by dispersing fine particle silica called vapor phase silica in water it is also possible to use colloidal silica obtained by dispersing fine particle silica called vapor phase silica in water.
- the average particle size of the colloidal silica (B) is preferably in the range of 1 to 100 nm, more preferably in the range of 2 to 20 nm. Appropriate irregularities on the surface of the hydrophilic film are effective for the drainage of condensed water. If the particle size is too small, the effect of colloidal silica will not be sufficiently exerted. The colloidal silica that has been used is likely to flow out after durability and is difficult to hold.
- the average particle diameter of colloidal silica is the number average particle diameter and is measured by a nitrogen adsorption method.
- Organoalkoxysilane (C) and / or its hydrolyzate (C) examples include phenylmethoxysilane, diphenylethoxysilane, phenyltrimethoxysilane, phenyltriethoxysilane, ⁇ - (3,4-epoxycyclohexyl) ethyltrimethoxysilane, and ⁇ -glycid.
- the organoalkoxysilane (C) more preferably contains a glycidyl group.
- the water-soluble resin (A) is firmly bonded to improve durability in an actual use environment, and good drainage can be maintained and exhibited even after durability.
- the surface of the colloidal silica is modified by bonding the silanol group of the organoalkoxysilane (C) with the silanol group of the colloidal silica (B) to form a surface with better drainage of condensed water.
- Crosslinker (D) capable of crosslinking with water-soluble resin (A) ⁇
- the crosslinking agent (D) used is not particularly limited as long as the water-soluble resin (A) can be crosslinked, but is preferably selected from the group consisting of a carboxyl group, an aldehyde group, a glycidyl group, a methylol ether group, and an isocyanate group. It is a water-soluble crosslinking agent containing at least one functional group.
- the crosslinking agent can be selected from methylol melamine, methylol urea, methylolated polyacrylamide, polyethylene glycol diglycidyl ether, glyoxal, butanetetracarboxylic acid, and the like.
- a crosslinking agent containing a carboxyl group particularly a crosslinking agent containing two or more carboxyl groups in one molecule is more preferable because it exhibits a high degree of crosslinking.
- the component (E) contains water as a main component (for example, 80% by volume or more, 90% by volume or more, 95% by volume or more based on the volume of all solvents). Although water is usually used, a solvent that can be mixed with water such as alcohol may be added.
- the hydrophilic surface treatment agent may contain a metal compound (F) containing at least one metal element selected from the group consisting of V, Ti, Zr, and Cr in order to improve corrosion resistance.
- a metal compound (F) containing at least one metal element selected from the group consisting of V, Ti, Zr, and Cr in order to improve corrosion resistance.
- the compound containing Cr include trivalent chromium compounds such as chromium sulfate, chromium nitrate, chromium biphosphate, chromium fluoride, chromium acetate, and chromium formate. Trivalent chromium compounds also include reduced products of chromic compounds such as chromic anhydride and dichromic acid.
- zirconium compound examples include zirconium compounds such as zirconium nitrate, zirconium oxynitrate, zirconyl acetate, zirconyl sulfate, zirconyl ammonium carbonate, zirconyl potassium carbonate, zirconyl sodium carbonate, zirconia sol, zirconate hydrofluoric acid or salts thereof.
- zirconic acid and its salt which are produced by ion-exchange and alkali neutralizing the aqueous solution of a water-soluble zirconium salt are also mentioned.
- titanium compounds include titanyl sulfate, titanyl nitrate, titanium nitrate, titanyl chloride, titania sol, titanium oxide, potassium potassium oxalate, titanium lactate, titanium tetraisopropoxide, titanium acetylacetonate, diisopropyltitanium bisacetylacetone, titanium hydrofluoric acid, It can be selected from titanium compounds such as salts thereof.
- metatitanic acid obtained by thermally hydrolyzing an aqueous solution of titanyl sulfate, orthotitanic acid obtained by alkali neutralization, and salts thereof are also included.
- vanadium compound examples include vanadium pentoxide, ammonium metavanadate, sodium metavanadate, vanadium oxytrichloride, vanadium trioxide, vanadium dioxide, vanadium oxysulfate, vanadium oxyacetylacetonate, vanadium acetylacetonate, vanadylacetylacetonate, Examples include vanadium trichloride, phosphovanadomolybdic acid, and vanadium sulfate.
- the metal compound is metal-crosslinked with the water-soluble resin (A) to increase the crosslink density of the hydrophilic film and improve the corrosion resistance. Thereby, even when there is no undercoat, good corrosion resistance is exhibited, and excellent drainage can be maintained even after durability.
- an antibacterial agent and / or an antifungal agent may be added to prevent the generation of spoiled odor due to the growth of mold, bacteria, and yeast. it can.
- the antibacterial and fungicidal agent that can be used in the present invention is one that can withstand the heating in removing the water that is the solvent of the treating agent in the drying step after applying the treating agent of the present invention, that is, up to 100 ° C. It is desirable that the fungicide and fungicide have no decomposition point at temperature.
- antifungal agent contained in the aqueous hydrophilic surface treatment agent of the present invention examples include 5-chloro-2-methyl-4-isothiazolin-3-one, 2-methyl-4-isothiazolin-3-one, 2 -(4-thiocyanomethylthio) benzothiazole, 2,2-dibromo-3-nitrilopropionamide, sodium ethylenebis (dithiocarbamate), sodium-2-pyridinethiol-1-oxide, zinc-2-pyridinethiol-1 -Oxide, 2,2'-dithiobis (pyridine-1-oxide), 2,4,5,6-tetrachloroisophthalonitrile, 2-methylcarbonylaminobenzimidazole, 2,3,5,6-tetrachloro- 4- (methylsulfonyl) -pyridine, 2- (4-thiazolyl) -benzimidazole, N- (fluoro Dichloromethylthio) sulfamide, p-chloro-m
- hydrophilic surface treatment agent may contain a surfactant and a pigment for improving the treatment property and coloring.
- ⁇ Blending amount (A) to (D) ⁇
- the (B) + (C) and the total solid content ratio (mass ratio), ⁇ (B) + (C) ⁇ / ⁇ (A) + (B) + (C) + (D) ⁇ are 0. 1 to 0.5. Further, it is more preferably 0.2 to 0.5. If ⁇ (B) + (C)) / ⁇ (A) + (B) + (C) + (D) ⁇ exceeds 0.5, the film cannot be sufficiently fixed in the film and will be washed away. Moreover, when ((B) + (C)) / ((A) + (B) + (C) + (D)) is less than 0.1, both hydrophilicity and drainage are insufficient.
- the ratio (mass ratio) of the colloidal silica (B) and the organoalkoxysilane (C), (C) / (B) is 0.5 (preferably more than 0.5) to 4.0. More preferably, it is 1.0 to 3.0. If (C) / (B) is less than 0.5, the surface modification of the colloidal silica cannot be performed sufficiently, and if (C) / (B) exceeds 4.0, the surface of the hydrophilic film is not sufficiently uneven. It becomes.
- pretreatment such as heating and stirring may be performed in advance using (B) and (C) in separate containers. Heating is preferably 30 to 80 ° C. in order to promote hydrolysis efficiently.
- the hydrophilic surface treatment agent contains the metal compound, good corrosion resistance can be obtained without applying the base film.
- the amount of addition is not particularly limited, but the blending ratio of the solid content of the metal compound when the solid content of the raw material blended in the hydrophilic surface treating agent is 100 is 0.1 to 50% by mass, preferably 1 to 30% by mass, more preferably 10 to 20% by mass. If the added amount of the metal compound is small, the corrosion resistance is lowered, so that it is easily corroded, and the drainage after durability is lowered. On the other hand, if the amount added is excessive, the colloidal silica cannot be sufficiently dispersed on the outermost surface and the drainage performance is lowered.
- the blending ratio of the solid content of the antibacterial and antifungal agent is preferably 0.1 to 10% by mass when the solid content of the raw material blended in the hydrophilic surface treatment agent is 100, More preferably, the content is 0.1 to 5% by mass.
- hydrophilic surface treatment agent As the coating method, various coating methods such as dipping, roll coating, spin coating, and bar coating can be used, and among these, the dipping method is preferable.
- the pH of the hydrophilic surface treatment agent is preferably in the range of 1 to 7, more preferably 1 to 4, for the hydrophilic surface treatment. When pH exceeds 7, the stability of a structural component may fall.
- the pH refers to a value measured with a pH measuring instrument: Toa DKK Co., Ltd., pH meter MM-60R.
- the hydrophilic treatment agent applied is preferably heated at 100 to 180 ° C., more preferably 120 to 160 ° C., to form a hydrophilic film.
- the hydrophilic surface treating agent for aluminum-containing metal heat exchangers provided by the present invention is excellent in odor control and corrosion resistance. Furthermore, it is possible to maintain good drainage of condensed water over a long period of time.
- the mechanism for maintaining good dew condensation drainage with the hydrophilic surface treating agent of the present invention is understood as follows.
- the hydrophilic film formed by the hydrophilic treatment agent shows good hydrophilicity, but the unevenness on the outermost surface by colloidal silica plays a role of increasing hydrophilicity. Furthermore, since the surface of colloidal silica is modified with organoalkoxysilane, it is considered that it also plays a role of improving drainage (dynamic hydrophilicity). It is not clear why dynamic hydrophilicity is improved.
- contact angle hysteresis which is the difference between the contact angle of the tip of the water droplet that slides down (advance contact angle) and the contact angle of the rear end of the water droplet (retraction contact angle).
- colloidal silica modified with organoalkoxysilane any of factors such as chemical heterogeneity, molecular arrangement at the solid-liquid interface, and the three-dimensional shape of the surface reduce contact angle hysteresis. I believe.
- An aluminum test piece ⁇ Test piece A1050 manufactured by Partec Co., Ltd. ⁇ is immersed in a 20 g / L aqueous solution of an alkaline degreasing agent Fine Cleaner 315E (manufactured by Nihon Parkerizing Co., Ltd.) maintained at 60 ° C. for 120 seconds to obtain oil content, etc. After removing the contaminants on the surface, it was washed with tap water for 15 seconds.
- Fine Cleaner 315E manufactured by Nihon Parkerizing Co., Ltd.
- an A solution 40 g / L and a B agent 40 g / L of a hexavalent chromium-free chemical conversion treatment agent Pulcoat 3700 were added, and an aqueous solution containing 1.5 g / L of a neutralizing agent NT-4058 was kept at 50 ° C. What was immersed in the thing for 120 second and formed the base film was used as a test sample. These test samples are treated with a hydrophilic treatment agent without drying and used as samples for evaluation.
- Tables 1 to 5 show raw materials used for the preparation of the hydrophilic surface treatment agent.
- the raw materials were mixed in water with the compositions shown in Table 6 and Table 7 to obtain hydrophilic surface treatment agents.
- the compounding quantity (mass%) of the raw material in Table 6 and Table 7 makes the total amount of the solid content of the raw material mix
- a method for preparing the hydrophilic surface treating agent first, a predetermined colloidal silica and an organoalkoxysilane were mixed to prepare a silicon compound dispersion, and then water and various other components were added as a solvent. Note that the pH of each example was in the range of 1-7.
- each test sample was coated by immersion treatment, and then dried in an oven without washing with water to form a hydrophilic surface treatment film.
- the oven was dried at 150 ° C. for 10 minutes.
- test samples prepared in Examples 1 to 21 and Comparative Examples 1 to 6 were tested and evaluated by the following methods.
- Corrosion resistance Salt spray test method JIS
- the rust area of the fin portion after exposure to 720 hours in the corrosion resistance test based on Z-2371 (the ratio of the white rust generation area to the total area) was evaluated by visual observation.
- the evaluation criteria are as follows. Rank Corrosion resistance evaluation result ⁇ White rust generation area less than 1% ⁇ White rust generation area 1 to less than 5% ⁇ White rust generation area 5 to less than 30% ⁇ White rust generation area 30% or more
- Water resistance The amount of the film of the sample immersed in running water for 72 hours at the initial stage and room temperature was measured, and the remaining rate of the film amount after running water was calculated as a percentage.
- Water resistance (%) 100 x coating amount after running water (g / m 2 ) / initial coating amount (g / m 2 ) Rank Water resistance measurement result ⁇ 100-85% ⁇ 84-70% ⁇ 69-55% ⁇ 54% or less
- Table 8 summarizes the evaluation results of samples subjected to hydrophilic surface treatment with the hydrophilic surface treatment agents having the compositions of Examples 1 to 21 and Comparative Examples 1 to 6.
- the ratio of water-soluble resin (A), colloidal silica (B), organoalkoxysilane (C), and cross-linking agent (D) is appropriately adjusted, including a predetermined compound.
- the treated agent of the present invention exhibits excellent contact angle and sliding angle even after endurance, and also exhibits excellent corrosion resistance and water resistance.
- polyvinyl alcohol is used as the water-soluble resin (A)
- the particle size of the colloidal silica (B) is controlled to 2 to 20 nm
- an organoalkoxysilane containing a glycidyl group is used as the organoalkoxysilane (C).
- (B) + (C) ⁇ / ⁇ (A) + (B) + (C) + (D) ⁇ is in the range of 0.2 to 0.5
- (C) / (B) is 1. Examples 1, 4 and 10 with 0 to 3.0 exhibited extremely excellent overall performance. Further, by adding a metal compound to the hydrophilic surface treating agent, excellent corrosion resistance is exhibited even without the base chemical conversion treatment (Example 19).
Abstract
Description
コロイダルシリカ(B)と、
オルガノアルコキシシラン及び/又はその加水分解物(C)と、
水溶性樹脂(A)と架橋可能な架橋剤(D)と、
水(E)と
を配合してなり、
{(B)+(C)}/{(A)+(B)+(C)+(D)}が、固形分比率(質量比)で0.1~0.5であり、
(C)/(B)が、固形分比率(質量比)で0.5~4.0である
ことを特徴とするアルミニウム含有金属製熱交換器の親水性表面処理剤である。
≪熱交換器の説明≫
本発明の熱交換器の材料として使用されるアルミニウム含有金属は、アルミニウム、及びアルミニウム合金、例えば、アルミニウム-マグネシウム合金、アルミニウム-シリコン合金及びアルミニウム-マンガン合金等である。そして、当該材料は、例えばエアコンディショナーのような熱交換器に用いられるチューブ、フィン、及び中空プレートに成型加工される。これらの部品から形成される熱交換器としては、フィン及び冷媒が通るチューブ共にアルミニウム含有金属で形成される自動車用エアコンの熱交換器や、加工したアルミニウムシートを銅製のパイプと組み合わせパイプを拡管してフィンとパイプを接合する家電用エアコンの熱交換器が例として挙げられる。ここで、本発明が適用される熱交換器は、当業者にとって周知構造であり、図1は、当該周知構造の一例を示したものである。より具体的には、図1に示される熱交換器は、いわゆるフィンチューブ型熱交換器と称されるものである。そして、当該図に示されるように、この種の熱交換器においては、フィンの間に水滴によるブリッジが形成される場合がある。ここで、このようなフィンチューブ型熱交換器に親水性表面処理をする場合、一般的に、ルームエアコンは、まずアルミニウムシートを親水性表面処理する(プレコートと呼ばれる)。そのシートを成型加工して図1の熱交換器の形とする。また、一般的に、カーエアコンは、まずアルミニウムシートを成型加工して図1の熱交換器の形とする。この熱交換器を親水性表面処理する(ポストコートと呼ばれる)。但し、これら構造や処理法はあくまで一例に過ぎず、本発明を限定するものではない。
アルミニウム含有金属基体の表面に親水性表面処理剤を塗布することで良好な性能が得られる。しかし、耐食性向上のために親水性表面処理皮膜の下地として、アンダーコート化成層で被覆してもよい。このアンダーコート化成層は、アルミニウム含有金属基体の表面に、化成処理、例えば、クロム、ジルコニウム、チタン、バナジウム、からなる群から選ばれる少なくとも一種の金属元素を含む化成処理剤から形成される。アンダーコート化成層は、2~500mg/m2の塗布量又は0.002~0.5μmの厚さに形成されることが好ましい。他方、製造プロセスの簡素化やコスト削減等の観点からは、当該下地皮膜形成処理を省略することが好ましいといえるが、本発明によれば、当該下地皮膜が存在しなくても、十分に満足できる排水性を達成できる。
親水性表面処理剤は、アミド基、ヒドロキシル基及びカルボキシル基の少なくとも1種以上の官能基を有する水溶性樹脂、又は骨格中にアミド結合を有する水溶性樹脂(A)と、
コロイダルシリカ(B)と、
オルガノアルコキシシラン及び/又はその加水分解物(C)と、
水溶性樹脂(A)と架橋可能な架橋剤(D)と、
水(E)と、を配合してなり、
{(B)+(C)}/{(A)+(B)+(C)+(D)}が、固形分比率(質量比)で0.1~0.5であり、
(C)/(B)が、固定分比率で0.5~4.0(質量比)である
アルミニウム含有金属製熱交換器の親水性表面処理剤である。
アミド基、ヒドロキシル基及びカルボキシル基の少なくとも1種以上の官能基を有する水溶性樹脂、又は骨格中にアミド結合を有する水溶性樹脂(A)は、例えば、アクリルアミド化合物のホモポリマー及びコポリマーが挙げられる。コポリマーとしては、アクリルアミドモノマーの1種以上と他のコモノマーの1種以上の共重合体を包含する。ここで用いられるコモノマーは、アクリルアミドと重合可能なアニオン性、ノニオン性、カチオン性の付加重合モノマーから選ばれ、例えば(メタ)アクリル酸、イタコン酸、マレイン酸、ビニルスルホン酸、スチレンスルホン酸、スルホエチルアクリレート、スルホエチルメタクリレート、N-メチレンスルホン酸アクリルアミド、2-アクリルアミド-2-メチルプロパンスルホン酸、アシッドホスホオキシエチルメタクリレート又はこれらの塩等のアニオン性不飽和モノマー;(メタ)アクリルアミド、N-メチロール(メタ)アクリルアミド、ヒドロキシエチル(メタ)アクリレート、ビニルピロリドン、アクロイルモルホリン、ポリエチレングリコールアクリレート、ポリエチレングリコールアクリレートアルキルフェニルエーテル等のノニオン性の不飽和モノマー;並びにアミノエチル(メタ)アクリレート、N,N-ジメチルアミノエチルアクリレート、N-ヒドロキシプロピルアミノエチル(メタ)アクリレート、ヒドロキシメチルアミノエチルメタクリレート、ビニルイミダゾール、ビニルピリジン、N,N-ジアリルアミン、N,N-ジアリル-N,N-ジメチルアンモニウムクロリド等のカチオン性不飽和モノマー等から選ぶことができる。
1)GPC
装置:Waters製244型ゲル浸透クロマトグラフ
カラム:東ソー製TSK-gel-GMPW
(内径8mm、長さ30cm、2本)
溶媒:0.1M-トリス緩衝液(pH7.9)
流速:0.5ml/min
温度:23℃
試料濃度:0.040%
ろ過:東ソー製0.45μmマイショリディスクW-25-5
注入量:0.2ml
検出感度(示差屈折率検出器):4倍
2)LALLS
装置:Chromatrix製KMX-6型低角度レーザー光散乱光度計
温度:23℃
波長:633nm
第2ビリアル係数×濃度:0mol/g
屈折率濃度変化(dn/dc):0.159ml/g
フィルター:MILLIPORE製0.45μmフィルターHAWP01300
ゲイン:800mV
使用されるコロイダルシリカ(B)は、ケイ素と酸素を主構成成分とする珪酸化合物の分散物であって、その種類は特に限定されない。一般には、珪酸ナトリウム、珪酸カリウム、珪酸リチウム等のアルカリ珪酸塩からイオン交換法によりナトリウム、カリウム、又はリチウムを取り除いてゾル化する等の方法により得られるコロイダルシリカが用いられる。また、気相シリカと呼ばれる微粒子シリカを水に分散してコロイダルシリカとしたものも使用できる。
使用されるオルガノアルコキシシラン(C)は、例えば、フェニルメトキシシラン、ジフェニルエトキシシラン、フェニルトリメトキシシラン、フェニルトリエトキシシラン、β-(3,4-エポキシシクロヘキシル)エチルトリメトキシシラン、γ-グリシドキシプロピルメチルジエトキシシラン、γ-グリシドキシプロピルトリエトキシシラン、N-β(アミノエチル)γ-アミノプロピルトリメトキシシラン、γ-アミノプロピルトリエトキシシラン、γ-メタクリロキシプロピルメチルジエトキシシラン、γ-メルカプトプロピルメチルジメトキシシラン、p-スチリルトリメトキシシラン、γ-アクリロキシプロピルトリメトキシシラン、N-フェニル-γ-アミノプロピルトリメトキシシラン、γ―ウレイドプロピルトリエトキシシラン、γ―クロロプロピルトリメトキシシラン、ビス(トリエトキシシリルプロピル)テトラスルフィド、γ-イソシアネートプロピルトリエトキシシラン、γ-トリエトキシシリル-N-(1,3-ジメチルブチリデン)プロピルアミン、N-(ビニルベンジルアミン)-β-アミノエチル-γ-アミノプロピルトリメトキシシラン等が挙げられる。
使用される架橋剤(D)は、水溶性樹脂(A)を架橋可能な限り特に限定されないが、好適には、カルボキシル基、アルデヒド基、グリシジル基、メチロールエーテル基及びイソシアネート基からなる群から選ばれる少なくとも1種の官能基を含む水溶性架橋剤である。架橋剤としては、メチロールメラミン、メチロール尿素、メチロール化ポリアクリルアミド、ポリエチレングリコールジグリシジルエーテル、グリオキザール、ブタンテトラカルボン酸、等から選ぶことができる。その中でも、カルボキシル基を含有する架橋剤、特に1分子中にカルボキシル基を2個以上含む架橋剤が高い架橋度を示すためにより好ましい。
成分(E)は水を主成分とする(例えば、全溶媒の容量を基準として、80容量%以上、90容量%以上、95容量%以上)。尚、通常は水が用いられるが、アルコール等の水と混合可能な溶剤を添加してもよい。
親水性表面処理剤は、耐食性を向上するためにV、Ti、Zr、及びCrからなる群から選ばれる少なくとも1種の金属元素を含む金属化合物(F)を含有してもよい。Crを含む化合物としては、例えば3価のクロム化合物である硫酸クロム、硝酸クロム、重りん酸クロム、フッ化クロム、酢酸クロム、蟻酸クロム等が挙げられる。3価のクロム化合物には無水クロム酸、重クロム酸等のクロム酸化合物の還元物も含まれる。ジルコニウム化合物としては、例えば硝酸ジルコニウム、オキシ硝酸ジルコニウム、酢酸ジルコニル、硫酸ジルコニル、炭酸ジルコニルアンモニウム、炭酸ジルコニルカリウム、炭酸ジルコニルナトリウム、ジルコニアゾル、ジルコンフッ酸若しくはその塩等のジルコニウム化合物等が挙げられる。また、水溶性ジルコニウム塩の水溶液を、イオン交換やアルカリ中和して作られるジルコン酸及びその塩も挙げられる。チタン化合物としては、例えば硫酸チタニル、硝酸チタニル、硝酸チタン、塩化チタニル、チタニアゾル、酸化チタン、シュウ酸チタンカリウム、チタンラクテート、チタンテトライソプロポキシド、チタンアセチルアセトネート、ジイソプロピルチタニウムビスアセチルアセトン、チタンフッ酸若しくはその塩等のチタン化合物等から選ぶことができる。また、硫酸チタニルの水溶液を、熱加水分解させて得られるメタチタン酸や、アルカリ中和で得られるオルソチタン酸及びこれらの塩も挙げられる。バナジウム化合物としては、例えば五酸化バナジウム、メタバナジン酸アンモニウム、メタバナジン酸ナトリウム、オキシ三塩化バナジウム、三酸化バナジウム、二酸化バナジウム、オキシ硫酸バナジウム、バナジウムオキシアセチルアセトネート、バナジウムアセチルアセトネート、バナジルアセチルアセトネート、三塩化バナジウム、リンバナドモリブデン酸、硫酸バナジウム等が挙げられる。金属化合物は水溶性樹脂(A)と金属架橋して親水性皮膜の架橋密度を上げ、耐食性を向上する。これにより、下地皮膜がない場合でも良好な耐食性を発揮し、耐久後も優れた排水性を維持することが可能となる。
また、本発明の水性処理剤に、その効果を損わない限り、防菌剤、及び/又は防かび剤を添加して、カビ、バクテリア、酵母の繁殖による腐敗臭の発生を防止することができる。本発明に用い得る防菌防かび剤は、本発明の処理剤を塗布した後の乾燥工程において、処理剤の溶媒である水を除去する際の加熱に耐え得るもの、すなわち、100℃までの温度に分解点を有していない防菌防かび剤であることが望ましい。本発明の水性親水性表面処理剤に含有させる防菌防かび剤は、例えば、5-クロロ-2-メチル-4-イソチアゾリン-3-オン、2-メチル-4-イソチアゾリン-3-オン、2-(4-チオシアノメチルチオ)ベンゾチアゾール、2,2-ジブロモ-3-ニトリロプロピオンアミド、ソディウムエチレンビス(ジチオカーバメート)、ソディウム-2-ピリジンチオール-1-オキサイド、ジンク-2-ピリジンチオール-1-オキサイド、2,2′-ジチオビス(ピリジン-1-オキサイド)、2,4,5,6-テトラクロロイソフタロニトリル、2-メチルカルボニルアミノベンツイミダゾール、2,3,5,6-テトラクロロ-4-(メチルスルホニル)-ピリジン、2-(4-チアゾリル)-ベンツイミダゾール、N-(フルオロジクロロメチルチオ)スルファミド、p-クロロ-m-キシレノール、デヒドロ酢酸、o-フェニルフェノール、1,2-ベンツイソチアゾリン-3-オン、2-ブロモ-2-ニトロプロパン-1,3-ジオール、メタほう酸バリウム、ジヨードメチル-p-トルエンスルフォン、2-n-オクチル-4-イソチアゾリン-3-オン、及び10,10′-オキシビスフェノキシアルシン、1,2-ジブロモ-2,4-ジシアノブタン、等から選ぶことができる。
前記(B)+(C)と全体の固形分比率(質量比)、{(B)+(C)}/{(A)+(B)+(C)+(D)}は、0.1~0.5である。また、より好ましくは0.2~0.5である。{(B)+(C))/{(A)+(B)+(C)+(D)}が0.5を超えると皮膜中に十分に固定化できずに流去してしまう。また、((B)+(C))/((A)+(B)+(C)+(D))が0.1を下回ると親水性、排水性共に不十分となる。
親水性表面処理剤が前記金属化合物を含有することによって、前記下地皮膜を塗布しなくても良好な耐食性が得られるようになる。添加量は特に限定しないが、親水性表面処理剤中に配合される原料の固形分を100としたときの金属化合物の固形分の配合比として、0.1~50質量%、好ましくは1~30質量%、更に好ましくは10~20質量%である。金属化合物の添加量が少ないと耐食性が低下するために腐食しやすくなり、耐久後の排水性が低下する。また、添加量が過剰になると最表面へのコロイダルシリカの分散が十分にできず排水性が低下する。
他の成分に関しては、必要性等に応じ、適宜、適当量を配合すればよい。例えば、防菌防カビ剤の固形分の配合比としては、親水性表面処理剤中に配合される原料の固形分を100としたときに、0.1~10質量%であることが好ましく、より好ましくは0.1~5質量%である。
塗布方法は、ディッピング、ロールコート、スピンコート、バーコート等の各種塗布方法を用いることができるが、その中でもディッピング法が好ましい。
ランク 接触角測定結果
◎ 0°~10°
○ 11°~20°
△ 21°~30°
× 31°以上
ランク 接触角測定結果
◎ 0°~20°
○ 21°~40°
△ 41°~60°
× 61°以上
ランク 滑落角測定結果
◎ 0°~10°
○ 11°~20°
△ 21°~30°
× 31°以上
ランク 滑落角測定結果
◎ 0°~20°
○ 21°~40°
△ 41°~60°
× 61°以上
Z-2371に基づく、耐食性試験に720時間暴露後のフィン部の錆面積(全体の面積に対する白錆発生面積の割合)を目視観察により評価した。評価基準は下記の通りである。
ランク 耐食性評価結果
◎ 白錆発生面積 1%未満
○ 白錆発生面積 1~5%未満
△ 白錆発生面積 5~30%未満
× 白錆発生面積 30%以上
耐水性(%)=100×流水後皮膜量(g/m2)/初期皮膜量(g/m2)
ランク 耐水性測定結果
◎ 100―85%
○ 84―70%
△ 69―55%
× 54%以下
Claims (13)
- アミド基、ヒドロキシル基及びカルボキシル基の少なくとも1種以上の官能基を有する水溶性樹脂、又は骨格中にアミド結合を有する水溶性樹脂(A)と、
コロイダルシリカ(B)と、
オルガノアルコキシシラン及び/又はその加水分解物(C)と、
水溶性樹脂(A)と架橋可能な架橋剤(D)と、
水(E)と
を配合してなり、
{(B)+(C)}/{(A)+(B)+(C)+(D)}が、固形分比率(質量比)で0.1~0.5であり、
(C)/(B)が、固形分比率(質量比)で0.5~4.0である
ことを特徴とするアルミニウム含有金属製熱交換器の親水性表面処理剤。 - 前記コロイダルシリカ(B)の平均粒径が1~100nmの範囲である請求項1に記載の親水性表面処理剤。
- 前記コロイダルシリカ(B)の平均粒径が2~20nmの範囲である請求項1に記載の親水性表面処理剤。
- 前記オルガノアルコキシシラン(C)がグリシジル基を含有することを特徴とする請求項1~3のいずれか一項に記載の親水性表面処理剤。
- 前記親水性表面処理剤のpHが1~7である請求項1~4のいずれか一項に記載の親水性表面処理剤。
- 前記水溶性樹脂(A)が、ポリビニルアルコール、又はその誘導体であることを特徴とする請求項1~5のいずれか一項に記載の親水性表面処理剤。
- 前記水溶性樹脂(A)の重量平均分子量が5,000~200,000であることを特徴とする請求項6に記載の親水性表面処理剤。
- 前記水溶性樹脂(A)のケン化度が95mol%以上であることを特徴とする請求項6又は7に記載の親水性表面処理剤。
- 前記架橋剤(D)が、カルボキシル基、アルデヒド基、グリシジル基、メチロールエーテル基及びイソシアネート基からなる群から選ばれる少なくとも1種の官能基を含む架橋剤であることを特徴とする請求項1~8のいずれか一項に記載の親水性表面処理剤。
- 前記親水性表面処理剤が、V、Ti、Zr及びCrからなる群から選ばれる少なくとも1種の金属元素を含む金属化合物(F)を含有することを特徴とする請求項1~9のいずれか一項に記載の親水性表面処理剤。
- 請求項1~10のいずれか一項に記載の親水性表面処理剤でアルミニウム含有金属製熱交換器を表面処理する表面処理工程を含む、表面に親水性皮膜を有するアルミニウム含有金属製熱交換器の製造方法。
- 前記表面処理工程に先立ち、下地化成処理を実施しない、請求項11に記載の製造方法。
- 請求項1~10のいずれか一項に記載の親水性表面処理剤で表面処理された、表面に親水性皮膜を有するアルミニウム含有金属製熱交換器。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020147035915A KR101555162B1 (ko) | 2013-03-21 | 2013-03-21 | 배수성이 우수한 알루미늄 함유 금속제 열교환기의 친수성 표면 처리제 |
| US14/418,646 US9534132B2 (en) | 2013-03-21 | 2013-03-21 | Hydrophilic surface treatment agent for aluminum-containing metal heat exchangers having excellent drainage |
| IN95DEN2015 IN2015DN00095A (ja) | 2013-03-21 | 2013-03-21 | |
| CN201380033594.XA CN104411788B (zh) | 2013-03-21 | 2013-03-21 | 排水性优异的含有铝的金属制热交换器的亲水性表面处理剂 |
| EP13879100.9A EP2977417B1 (en) | 2013-03-21 | 2013-03-21 | Hydrophilic surface treatment agent for aluminum-containing metal heat exchangers having excellent drainage |
| HUE13879100A HUE035627T2 (en) | 2013-03-21 | 2013-03-21 | Hydrophilic Surface Treatment Agent for Aluminum Heat Exchanger with Excellent Conductivity |
| AU2013382832A AU2013382832A1 (en) | 2013-03-21 | 2013-03-21 | Hydrophilic surface treatment agent for aluminum-containing metal heat exchangers having excellent drainage |
| JP2013547425A JP5497971B1 (ja) | 2013-03-21 | 2013-03-21 | 排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 |
| PCT/JP2013/058015 WO2014147782A1 (ja) | 2013-03-21 | 2013-03-21 | 排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/JP2013/058015 WO2014147782A1 (ja) | 2013-03-21 | 2013-03-21 | 排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014147782A1 true WO2014147782A1 (ja) | 2014-09-25 |
Family
ID=50941694
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/058015 WO2014147782A1 (ja) | 2013-03-21 | 2013-03-21 | 排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US9534132B2 (ja) |
| EP (1) | EP2977417B1 (ja) |
| JP (1) | JP5497971B1 (ja) |
| KR (1) | KR101555162B1 (ja) |
| CN (1) | CN104411788B (ja) |
| AU (1) | AU2013382832A1 (ja) |
| HU (1) | HUE035627T2 (ja) |
| IN (1) | IN2015DN00095A (ja) |
| WO (1) | WO2014147782A1 (ja) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017086342A1 (ja) * | 2015-11-20 | 2017-05-26 | 株式会社Uacj | プレコートフィン及び熱交換器 |
| WO2017213097A1 (ja) * | 2016-06-09 | 2017-12-14 | パナソニックIpマネジメント株式会社 | 防汚被覆膜および防汚被覆体 |
| JPWO2017017789A1 (ja) * | 2015-07-28 | 2018-02-22 | 三菱電機株式会社 | 熱交換器及び冷凍サイクル装置 |
| JP2018070677A (ja) * | 2016-10-25 | 2018-05-10 | 日本パーカライジング株式会社 | 表面処理剤、皮膜及び表面処理方法 |
| WO2018230431A1 (ja) * | 2017-06-12 | 2018-12-20 | 株式会社デンソー | 熱交換器およびコルゲートフィン |
| JP2019002589A (ja) * | 2017-06-12 | 2019-01-10 | 株式会社デンソー | 熱交換器およびコルゲートフィン |
| JP2019211115A (ja) * | 2018-05-31 | 2019-12-12 | 株式会社デンソー | 熱交換器およびコルゲートフィン |
| JP2020117777A (ja) * | 2019-01-24 | 2020-08-06 | 日本パーカライジング株式会社 | 金属表面処理剤、並びに皮膜を有する金属材料及びその製造方法 |
| WO2021131374A1 (ja) * | 2019-12-26 | 2021-07-01 | 日本パーカライジング株式会社 | 金属表面処理剤、並びに皮膜を有する金属材料及びその製造方法 |
| WO2023189618A1 (ja) * | 2022-03-28 | 2023-10-05 | 日本パーカライジング株式会社 | 表面処理剤 |
Families Citing this family (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105189816B (zh) * | 2013-04-26 | 2018-06-08 | 日本帕卡濑精株式会社 | 水性亲水化表面处理剂、亲水性皮膜和亲水化表面处理方法 |
| WO2016021071A1 (ja) * | 2014-08-08 | 2016-02-11 | 日本パーカライジング株式会社 | アルミニウム含有金属材料用親水化処理剤 |
| WO2016065366A1 (en) * | 2014-10-24 | 2016-04-28 | Brighton Technologies Llc | Method and device for detecting substances on surfaces |
| US10837712B1 (en) * | 2015-04-15 | 2020-11-17 | Advanced Cooling Technologies, Inc. | Multi-bore constant conductance heat pipe for high heat flux and thermal storage |
| JP6145592B1 (ja) * | 2017-03-21 | 2017-06-14 | 日本パーカライジング株式会社 | アルミニウム含有金属材料用親水化処理剤 |
| CN107129739B (zh) * | 2017-06-22 | 2019-10-18 | 杭州固邦防腐科技有限公司 | 一种高分子特种防腐涂料 |
| KR102580285B1 (ko) * | 2018-07-10 | 2023-09-19 | 엘지전자 주식회사 | 열교환 시스템 |
| CN112714674B (zh) | 2018-07-20 | 2022-12-09 | 布赖顿技术有限责任公司 | 用于从液体微滴分配系统收集的样本数据确定微滴的质量的方法和装置 |
| US11338220B2 (en) | 2018-12-03 | 2022-05-24 | Exaeris Water Innovations, Llc | Atmospheric water generator apparatus |
| WO2020144360A1 (en) * | 2019-01-11 | 2020-07-16 | Nouryon Chemicals International B.V. | Stain resistant coating |
| CN110204936A (zh) * | 2019-03-14 | 2019-09-06 | 上海丰野表面处理剂有限公司 | 一种汽车散热器用涂料及对散热器进行处理的工艺方法 |
| KR102558305B1 (ko) | 2020-11-13 | 2023-07-24 | 한양대학교 산학협력단 | 발수-친수성 패턴 표면을 갖는 면상 발열 열교환기 핀 |
| KR20240014112A (ko) | 2022-07-22 | 2024-02-01 | 전남대학교산학협력단 | 금속재료용 항곰팡이성 친수화처리제, 이를 포함하는 항곰팡이성 친수화처리액, 그 제조방법 및 항곰팡이성 친수피막형성방법 |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01223188A (ja) * | 1988-03-02 | 1989-09-06 | Kansai Paint Co Ltd | 熱交換器フイン材用親水化処理剤 |
| JPH0693209A (ja) | 1991-11-13 | 1994-04-05 | Sanyo Chem Ind Ltd | アルミニウム製熱交換器またはそのフィン材用親水性皮膜形成剤 |
| JPH06228459A (ja) * | 1993-01-29 | 1994-08-16 | Kansai Paint Co Ltd | 親水化処理用組成物および親水化処理方法 |
| JPH06264001A (ja) * | 1993-03-11 | 1994-09-20 | Nippon Light Metal Co Ltd | アルミニウム材料の表面に親水性を付与する表面処理 |
| JPH07233270A (ja) | 1994-02-24 | 1995-09-05 | Kishimoto Akira | 防曇性に優れた農業用フィルム及びその製造方法 |
| JPH08291269A (ja) * | 1995-02-23 | 1996-11-05 | Furukawa Electric Co Ltd:The | 親水化処理用組成物及び親水化処理熱交換器用部材 |
| JPH09296121A (ja) | 1996-05-02 | 1997-11-18 | Nippon Parkerizing Co Ltd | 金属材料用水性表面親水性化処理剤および表面処理方法 |
| JPH1030069A (ja) * | 1996-03-28 | 1998-02-03 | Nippon Light Metal Co Ltd | 水性親水性付与剤及びその付与剤を使用する熱交換器用プレコートフィン材の製造方法 |
| JPH11343450A (ja) | 1998-02-19 | 1999-12-14 | Nippon Parkerizing Co Ltd | アルミニウムまたはアルミニウム合金の親水化処理用組成物および親水化処理方法 |
| JP2001172547A (ja) * | 1999-12-21 | 2001-06-26 | Kansai Paint Co Ltd | 熱交換器フィン材用親水化処理組成物 |
| JP2001247822A (ja) * | 2000-03-06 | 2001-09-14 | Kansai Paint Co Ltd | 親水化処理用組成物及び親水化処理方法 |
| JP2001323250A (ja) | 2000-05-12 | 2001-11-22 | Nippon Paint Co Ltd | 親水化処理剤およびその製造方法 |
| JP2005162533A (ja) | 2003-12-02 | 2005-06-23 | Fuso Chemical Co Ltd | 変性コロイダルシリカの製造方法 |
| WO2009044912A1 (ja) | 2007-10-05 | 2009-04-09 | Harima Chemicals, Inc. | 親水性被覆剤、親水性被膜、及び親水性基材 |
| JP2009079889A (ja) * | 2007-09-05 | 2009-04-16 | Fujifilm Corp | フィン材 |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4478909A (en) * | 1980-10-24 | 1984-10-23 | Toray Industries, Inc. | Anti-fogging coating film |
| JPS6357673A (ja) * | 1986-08-29 | 1988-03-12 | Toyo Ink Mfg Co Ltd | アルミニウムフイン用塗料 |
| JPS63262238A (ja) * | 1987-04-20 | 1988-10-28 | スカイアルミニウム株式会社 | 熱交換器フイン材 |
| CA2021933A1 (en) * | 1989-07-27 | 1991-01-28 | Hiroshi Hosono | Process for preparation of anti-fogging coating |
| JPH04263794A (ja) * | 1990-12-28 | 1992-09-18 | Showa Alum Corp | アルミニウム・フィン付き熱交換器 |
| JPH06300482A (ja) * | 1991-06-19 | 1994-10-28 | Nippon Parkerizing Co Ltd | 熱交換器 |
| JPH05202313A (ja) | 1992-01-24 | 1993-08-10 | Kansai Paint Co Ltd | 親水化処理剤 |
| JPH07268274A (ja) | 1994-04-01 | 1995-10-17 | Kansai Paint Co Ltd | 親水化処理用組成物および親水化処理方法 |
| EP0937757A1 (en) | 1998-02-19 | 1999-08-25 | Nihon Parkerizing Co., Ltd. | Composition and method for hydrophilic treatment of aluminium or aluminium alloy, and use of the composition |
| JP4942251B2 (ja) * | 2001-03-27 | 2012-05-30 | 日本ペイント株式会社 | 親水化処理剤、親水化処理方法及び親水化処理されたアルミニウム材又はアルミニウム合金材 |
| JP2005336394A (ja) | 2004-05-28 | 2005-12-08 | Mitsubishi Polyester Film Copp | 積層ポリエステルフィルム |
| JP5337394B2 (ja) | 2007-05-15 | 2013-11-06 | 富士フイルム株式会社 | 親水性コーティング組成物及びこれを用いた親水性部材 |
| US7851535B2 (en) | 2007-07-26 | 2010-12-14 | Henkel Corporation | Mold release agents employing polyvinyl alcohol |
| JP2009233872A (ja) * | 2008-03-25 | 2009-10-15 | Fujifilm Corp | 親水性部材 |
| JP5427382B2 (ja) | 2008-03-25 | 2014-02-26 | 富士フイルム株式会社 | 親水性部材、フィン材、アルミニウム製フィン材、熱交換器およびエアコン |
| JP2009235130A (ja) * | 2008-03-25 | 2009-10-15 | Fujifilm Corp | 親水性膜形成用組成物および親水性部材 |
| BRPI1006964A2 (pt) * | 2009-01-21 | 2016-04-12 | Nihon Parkerizing | agente hidrofilizante para material de metal contendo alumínio, método de hidrofilização, e material de metal contendo alumínio hidrofilizado. |
-
2013
- 2013-03-21 HU HUE13879100A patent/HUE035627T2/en unknown
- 2013-03-21 CN CN201380033594.XA patent/CN104411788B/zh active Active
- 2013-03-21 AU AU2013382832A patent/AU2013382832A1/en not_active Abandoned
- 2013-03-21 JP JP2013547425A patent/JP5497971B1/ja not_active Expired - Fee Related
- 2013-03-21 KR KR1020147035915A patent/KR101555162B1/ko active IP Right Grant
- 2013-03-21 IN IN95DEN2015 patent/IN2015DN00095A/en unknown
- 2013-03-21 EP EP13879100.9A patent/EP2977417B1/en active Active
- 2013-03-21 WO PCT/JP2013/058015 patent/WO2014147782A1/ja active Application Filing
- 2013-03-21 US US14/418,646 patent/US9534132B2/en active Active
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH01223188A (ja) * | 1988-03-02 | 1989-09-06 | Kansai Paint Co Ltd | 熱交換器フイン材用親水化処理剤 |
| JPH0693209A (ja) | 1991-11-13 | 1994-04-05 | Sanyo Chem Ind Ltd | アルミニウム製熱交換器またはそのフィン材用親水性皮膜形成剤 |
| JPH06228459A (ja) * | 1993-01-29 | 1994-08-16 | Kansai Paint Co Ltd | 親水化処理用組成物および親水化処理方法 |
| JPH06264001A (ja) * | 1993-03-11 | 1994-09-20 | Nippon Light Metal Co Ltd | アルミニウム材料の表面に親水性を付与する表面処理 |
| JPH07233270A (ja) | 1994-02-24 | 1995-09-05 | Kishimoto Akira | 防曇性に優れた農業用フィルム及びその製造方法 |
| JPH08291269A (ja) * | 1995-02-23 | 1996-11-05 | Furukawa Electric Co Ltd:The | 親水化処理用組成物及び親水化処理熱交換器用部材 |
| JPH1030069A (ja) * | 1996-03-28 | 1998-02-03 | Nippon Light Metal Co Ltd | 水性親水性付与剤及びその付与剤を使用する熱交換器用プレコートフィン材の製造方法 |
| JPH09296121A (ja) | 1996-05-02 | 1997-11-18 | Nippon Parkerizing Co Ltd | 金属材料用水性表面親水性化処理剤および表面処理方法 |
| JPH11343450A (ja) | 1998-02-19 | 1999-12-14 | Nippon Parkerizing Co Ltd | アルミニウムまたはアルミニウム合金の親水化処理用組成物および親水化処理方法 |
| JP2001172547A (ja) * | 1999-12-21 | 2001-06-26 | Kansai Paint Co Ltd | 熱交換器フィン材用親水化処理組成物 |
| JP2001247822A (ja) * | 2000-03-06 | 2001-09-14 | Kansai Paint Co Ltd | 親水化処理用組成物及び親水化処理方法 |
| JP2001323250A (ja) | 2000-05-12 | 2001-11-22 | Nippon Paint Co Ltd | 親水化処理剤およびその製造方法 |
| JP2005162533A (ja) | 2003-12-02 | 2005-06-23 | Fuso Chemical Co Ltd | 変性コロイダルシリカの製造方法 |
| JP2009079889A (ja) * | 2007-09-05 | 2009-04-16 | Fujifilm Corp | フィン材 |
| WO2009044912A1 (ja) | 2007-10-05 | 2009-04-09 | Harima Chemicals, Inc. | 親水性被覆剤、親水性被膜、及び親水性基材 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2977417A4 |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2017017789A1 (ja) * | 2015-07-28 | 2018-02-22 | 三菱電機株式会社 | 熱交換器及び冷凍サイクル装置 |
| WO2017086342A1 (ja) * | 2015-11-20 | 2017-05-26 | 株式会社Uacj | プレコートフィン及び熱交換器 |
| CN108291788A (zh) * | 2015-11-20 | 2018-07-17 | 株式会社Uacj | 预涂翅片及热交换器 |
| WO2017213097A1 (ja) * | 2016-06-09 | 2017-12-14 | パナソニックIpマネジメント株式会社 | 防汚被覆膜および防汚被覆体 |
| JPWO2017213097A1 (ja) * | 2016-06-09 | 2019-04-04 | パナソニックIpマネジメント株式会社 | 防汚被覆膜および防汚被覆体 |
| JP2018070677A (ja) * | 2016-10-25 | 2018-05-10 | 日本パーカライジング株式会社 | 表面処理剤、皮膜及び表面処理方法 |
| JP2019002589A (ja) * | 2017-06-12 | 2019-01-10 | 株式会社デンソー | 熱交換器およびコルゲートフィン |
| WO2018230431A1 (ja) * | 2017-06-12 | 2018-12-20 | 株式会社デンソー | 熱交換器およびコルゲートフィン |
| JP2019211115A (ja) * | 2018-05-31 | 2019-12-12 | 株式会社デンソー | 熱交換器およびコルゲートフィン |
| JP2020117777A (ja) * | 2019-01-24 | 2020-08-06 | 日本パーカライジング株式会社 | 金属表面処理剤、並びに皮膜を有する金属材料及びその製造方法 |
| WO2021131374A1 (ja) * | 2019-12-26 | 2021-07-01 | 日本パーカライジング株式会社 | 金属表面処理剤、並びに皮膜を有する金属材料及びその製造方法 |
| JP2021105200A (ja) * | 2019-12-26 | 2021-07-26 | 日本パーカライジング株式会社 | 金属表面処理剤、並びに皮膜を有する金属材料及びその製造方法 |
| WO2023189618A1 (ja) * | 2022-03-28 | 2023-10-05 | 日本パーカライジング株式会社 | 表面処理剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20150008502A (ko) | 2015-01-22 |
| AU2013382832A1 (en) | 2015-02-05 |
| CN104411788B (zh) | 2016-07-06 |
| CN104411788A (zh) | 2015-03-11 |
| EP2977417A1 (en) | 2016-01-27 |
| KR101555162B1 (ko) | 2015-09-22 |
| IN2015DN00095A (ja) | 2015-05-29 |
| JP5497971B1 (ja) | 2014-05-21 |
| EP2977417B1 (en) | 2017-08-02 |
| JPWO2014147782A1 (ja) | 2017-02-16 |
| US20150232681A1 (en) | 2015-08-20 |
| HUE035627T2 (en) | 2018-05-28 |
| EP2977417A4 (en) | 2016-11-09 |
| US9534132B2 (en) | 2017-01-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5497971B1 (ja) | 排水性に優れたアルミニウム含有金属製熱交換器の親水性表面処理剤 | |
| JP4008620B2 (ja) | アルミニウム合金製熱交換器 | |
| CN1730725B (zh) | 熔剂钎焊的铝制热交换器的表面处理法 | |
| EP3305847B1 (en) | Aqueous resin dispersion, production method for aqueous resin dispersion, hydrophilization agent, hydrophilization method, metal material, and heat exchanger | |
| CN102892927B (zh) | 铝材制热交换器的耐腐蚀处理方法 | |
| JP4688602B2 (ja) | フラックスろう付けされたアルミ熱交換器の表面処理方法 | |
| JP4102028B2 (ja) | 金属材料の親水化処理剤、親水化処理液、親水化処理方法、金属材料および熱交換器 | |
| JP2002285139A (ja) | 親水化処理剤、親水化処理方法及び親水化処理されたアルミニウム材又はアルミニウム合金材 | |
| JP2000248381A (ja) | アルミニウム材の親水性処理方法及びその下地処理剤並びに親水性塗料 | |
| AU4384701A (en) | Treatment agent for hydrophilicity and method for preparing thereof | |
| JP2011153745A (ja) | 熱交換器の表面処理方法、表面処理剤、及びアルミニウム製熱交換器 | |
| US8821651B2 (en) | Method for surface treating a heat exchanger, hydrophilizing treatment agent, and aluminum heat exchanger | |
| JP5616669B2 (ja) | アルミニウム基材用耐食処理剤、及びそれを用いたアルミニウム基材の耐食処理方法 | |
| JP2000328038A (ja) | 親水化処理用組成物及び親水化処理方法 | |
| JP2002285140A (ja) | 親水化処理剤、親水化処理方法及び親水化処理されたアルミニウム材又はアルミニウム合金材 | |
| JP2015124390A (ja) | 熱交換器の表面処理方法 | |
| JP2000248225A (ja) | 親水化処理用組成物及び親水化処理方法 | |
| JP2000256579A (ja) | 親水性塗料組成物及びその組成物からなる親水性皮膜 | |
| JP4467264B2 (ja) | フィン用塗料組成物およびフィン材 | |
| JP2019073749A (ja) | 親水化表面処理アルミニウム含有金属材及び熱交換器 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2013547425 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13879100 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20147035915 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2013879100 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013879100 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14418646 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2013382832 Country of ref document: AU Date of ref document: 20130321 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |