WO2014126157A1 - ラジカル重合性ポリエーテル、該ラジカル重合性ポリエーテルの製造方法、該ラジカル重合性ポリエーテル及びラジカル重合性ビニル系単量体を含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム - Google Patents
ラジカル重合性ポリエーテル、該ラジカル重合性ポリエーテルの製造方法、該ラジカル重合性ポリエーテル及びラジカル重合性ビニル系単量体を含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム Download PDFInfo
- Publication number
- WO2014126157A1 WO2014126157A1 PCT/JP2014/053344 JP2014053344W WO2014126157A1 WO 2014126157 A1 WO2014126157 A1 WO 2014126157A1 JP 2014053344 W JP2014053344 W JP 2014053344W WO 2014126157 A1 WO2014126157 A1 WO 2014126157A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polyether
- radically polymerizable
- mass
- polymerizable
- film
- Prior art date
Links
- 0 *C(OCC(COI)OCCCC[O+]1CCCC1)=O Chemical compound *C(OCC(COI)OCCCC[O+]1CCCC1)=O 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/04—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers only
- C08G65/22—Cyclic ethers having at least one atom other than carbon and hydrogen outside the ring
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F283/00—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G
- C08F283/06—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G on to polyethers, polyoxymethylenes or polyacetals
- C08F283/065—Macromolecular compounds obtained by polymerising monomers on to polymers provided for in subclass C08G on to polyethers, polyoxymethylenes or polyacetals on to unsaturated polyethers, polyoxymethylenes or polyacetals
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F290/00—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups
- C08F290/08—Macromolecular compounds obtained by polymerising monomers on to polymers modified by introduction of aliphatic unsaturated end or side groups on to polymers modified by introduction of unsaturated side groups
- C08F290/14—Polymers provided for in subclass C08G
- C08F290/142—Polyethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/04—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers only
- C08G65/06—Cyclic ethers having no atoms other than carbon and hydrogen outside the ring
- C08G65/16—Cyclic ethers having four or more ring atoms
- C08G65/20—Tetrahydrofuran
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/26—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers and other compounds
- C08G65/2603—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers and other compounds the other compounds containing oxygen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G65/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G65/02—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring
- C08G65/26—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers and other compounds
- C08G65/2603—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers and other compounds the other compounds containing oxygen
- C08G65/2615—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule from cyclic ethers by opening of the heterocyclic ring from cyclic ethers and other compounds the other compounds containing oxygen the other compounds containing carboxylic acid, ester or anhydride groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2351/00—Characterised by the use of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives of such polymers
- C08J2351/08—Characterised by the use of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Derivatives of such polymers grafted on to macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
Definitions
- the present invention 1) a radical polymerizable polyether obtained by ring-opening copolymerization of glycidyl (meth) acrylate and tetrahydrofuran, and a method for producing the same, 2) a polymerizable composition containing the radical polymerizable polyether and a radical polymerizable vinyl monomer, and 3) a copolymer, a molded article and a film formed by radical polymerization of the polymerizable composition.
- radical polymerization polyethers that can form copolymers having excellent mechanical properties such as excellent transparency, high elongation at break, and high bending strength by radical polymerization with radical polymerizable vinyl monomers.
- the present invention provides a united product, a molded product, and a film.
- Patent Document 1 discloses a radical polymerizable poly (ethylene) group having a (meth) acryloyl group produced by the action of boron trifluoride diethyl ether complex which is glycidyl (meth) acrylate, tetrahydrofuran and Lewis acid. Ethers are disclosed.
- the number average molecular weight is 300 to 3000 and the amount of (meth) acryloyl group introduced is 20 to 98 mol%
- the polymer composed of the radical polymerizable polyether has transparency, weather resistance, and elastic modulus. It is disclosed that the resin has an excellent balance of flexibility.
- Non-Patent Document 1 has a skeleton similar to that of Patent Document 1, and has a mass average molecular weight of 7900 to 18500, a number average molecular weight of 3300 to 10300, and an introduction amount of (meth) acryloyl group of 2.5 to 5.3.
- Mol% radically polymerizable polyether (Table 1 on page 3184), a solvent-soluble graft type block copolymer comprising the radically polymerizable polyether and methyl methacrylate, and the graft type block copolymer.
- a film made by solvent casting is disclosed.
- Patent Document 1 discloses a radical polymerizable polyether. Further, the radical polymerizable polyether can be copolymerized with other radical polymerizable monomers, and various kinds of radical polymerizable polyethers are used. There is a description that an additive can be added. However, what is specifically disclosed in Examples and the like is only a homopolymer of the radical polymerizable polyether, and when copolymerized with a radical polymerizable vinyl monomer as in the present invention. Furthermore, there is no disclosure of a radically polymerizable polyether capable of forming a copolymer having excellent transparency and mechanical properties.
- Non-Patent Document 1 describes that a film made of a copolymer is formed by a solvent casting method using a radical polymerizable polyether, methyl methacrylate (MMA). However, since the ring-opening polymerization does not proceed sufficiently with the described polymerization initiator and polymerization conditions, a high molecular weight product was not obtained.
- MMA methyl methacrylate
- the radical polymerizable polyether As a result of intensive studies by the present inventors, surprisingly, in the radical polymerizable polyether, the number average molecular weight and the amount of (meth) acryloyl group introduced are within a specific range, whereby the radical polymerizable polyether and the radical It has been found that the copolymer obtained by radical polymerization of a polymerizable monomer can be imparted with excellent mechanical properties such as excellent transparency, high elongation at break, and high bending strength. .
- a method is known in which ring-opening polymerization of an epoxy site is performed by adding an acid, base, metal salt, Lewis acid or the like as a catalyst. It is known that an oxonium ion is generated by coordination of a catalyst to an epoxy site, and this generated oxonium ion induces ring-opening polymerization of tetrahydrofuran.
- Non-Patent Document 2 discloses that polymerization with an epoxy compound such as propylene oxide, styrene oxide, cyclohexene oxide and a metal salt of trifluoromethanesulfonic acid composed of yttrium, scandium, lanthanum, etc. It is disclosed that a polyether copolymer comprising an epoxy compound can be obtained.
- an epoxy compound such as propylene oxide, styrene oxide, cyclohexene oxide and a metal salt of trifluoromethanesulfonic acid composed of yttrium, scandium, lanthanum, etc. It is disclosed that a polyether copolymer comprising an epoxy compound can be obtained.
- Non-patent document 1 discloses that a polyether copolymer composed of a ring-opening polymer of tetrahydrofuran and glycidyl methacrylate can be obtained by polymerization using glycidyl methacrylate and a tin hexachloride ether complex. It is disclosed.
- Non-Patent Document 2 the epoxy compound or catalyst used for the reaction is dehydrated, and polymerization is performed in a state where water is not present in the reaction system as much as possible. Even when used, it is not disclosed until a high yield and high molecular weight polymer is obtained.
- the present invention when performing ring-opening copolymerization of tetrahydrofuran, by selecting a glycidyl ester compound as an epoxy compound and using a metal salt of trifluoromethanesulfonic acid as a catalyst, a high molecular weight polyether copolymer is obtained.
- the present invention provides a production method that can be produced at a high yield.
- the present invention is a radical polymerization capable of imparting excellent mechanical properties such as excellent transparency, high elongation at break and high bending strength to a copolymer obtained by radical polymerization of a radical polymerizable monomer. It is an object of the present invention to provide a functional polyether and a method for producing the same.
- the present invention provides the radical polymerizable polyether and the radical polymerizable vinyl monomer capable of forming a copolymer having excellent transparency and high mechanical properties such as high elongation at break and high bending strength. It is an object of the present invention to provide a polymerizable composition containing, and to provide a copolymer, a molded article and a film formed by radical polymerization of the polymerizable composition.
- the present inventors have determined that the number average molecular weight of the radical polymerizable polyether is in a specific range, and further, that the introduction amount of the (meth) acryloyl group is in a specific range. The inventors have found that the problem is solved, and have completed the following present invention.
- the present inventors have a carbonyl group at a site adjacent to the epoxy group represented by the following general formula (1) when producing a polyether copolymer by ring-opening polymerization of tetrahydrofuran.
- the first invention is a radical polymerizable polyether (A) having a mass average molecular weight of 20000 to 100,000 and comprising a polyalkylene ether skeleton and a polytetramethylene ether skeleton having a (meth) acryloyl group as a pendant group.
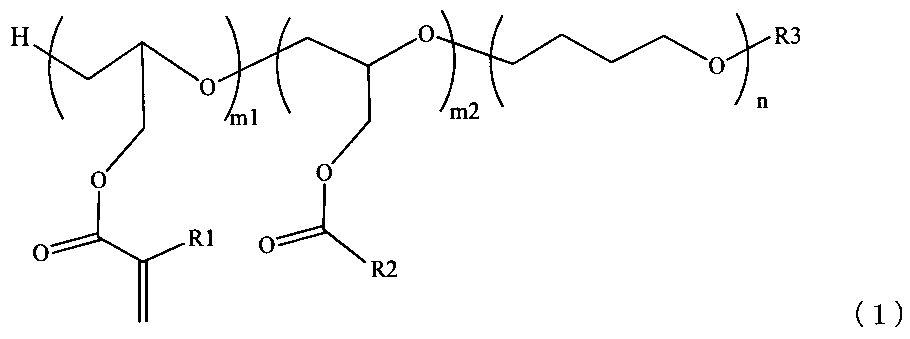
- the second invention is the radically polymerizable polyether (A) of the first invention represented by the following general formula (1) having a mass average molecular weight of 20,000 to 1,000,000.
- M1 is 1 to 7000, m2 is 0 to 7000, n is 1 to 14000, R1 is a methyl group or hydrogen, R2 and R3 are hydrogen or a hydrocarbon group having 1 to 20 carbon atoms)
- the third invention is the radically polymerizable polyether (A) in which m1, m2 and n in the general formula (1) are in a relationship of 0.0005 ⁇ m1 / (m1 + m2 + n) ⁇ 0.20 in the second invention. is there.
- the fourth invention is a radically polymerizable polyether (A) in which m1, m2, and n in the general formula (1) are in a relationship of 0.001 ⁇ m1 / (m1 + m2 + n) ⁇ 0.10 in the second invention. is there.
- the fifth invention contains the radical polymerizable polyether (A) and the radical polymerizable vinyl monomer (B) of any one of the first to fourth inventions as main components, and the components (A) 1 to A polymerizable composition (C) obtained by blending 99 to 1% by mass of the component (B) with respect to 99% by mass.
- the sixth invention is the polymerizable composition (C) according to the fifth invention, wherein the radical polymerizable vinyl monomer (B) is a (meth) acrylic monomer.
- the seventh invention is a copolymer formed by radical polymerization of the polymerizable composition (C) of the fifth or sixth invention.
- the eighth invention is a molded article formed by radical polymerization after shaping the polymerizable composition (C) of the fifth or sixth invention into a desired shape.
- the ninth invention is a film formed by radical polymerization of the polymerizable composition (C) of the fifth or sixth invention.
- the tenth invention is the film according to the ninth invention, wherein the film has a thickness of 0.1 to 3000 ⁇ m.
- the eleventh invention is a film according to the ninth or tenth invention, wherein the elongation at break when the tensile speed is 500 mm / min is 10% or more.
- the twelfth invention is the film according to any one of the ninth to eleventh inventions, wherein the haze is 5% or less when the thickness is 0.5 mm.
- glycidyl ester (a) represented by the following general formula (2) is added to 0.1 to 5 parts of trifluoromethanesulfonic acid metal salt (b) with respect to 100 parts by mass of tetrahydrofuran.
- This is a method for producing a radically polymerizable polyether (A) in which ring-opening polymerization of tetrahydrofuran is effected in the presence of parts by mass.
- R is a linear or branched hydrocarbon group having 1 to 20 carbon atoms, or a hydrocarbon group having an unsaturated bond having 2 to 10 carbon atoms
- the fourteenth invention is the manufacturing method according to the thirteenth invention, wherein the mass ratio (a) / (b) between the glycidyl ester (a) and the metal metal trifluoromethanesulfonate (b) is 2 to 9.
- the fifteenth aspect of the present invention is the manufacturing method according to the thirteenth or fourteenth aspect of the present invention, wherein the glycidyl ester (a) is glycidyl methacrylate.
- the sixteenth invention is a manufacturing method according to any one of the thirteenth to fifteenth inventions, wherein the metal of the trifluoromethanesulfonic acid metal salt (b) is at least one selected from the group consisting of scandium, yttrium and lanthanoids.
- a seventeenth aspect of the invention is a production method according to any one of the thirteenth to sixteenth aspects, wherein the metal salt of trifluoromethanesulfonic acid (b) is ytterbium trifluoromethanesulfonate.
- the radical polymerizable polyether (A) of the present invention has a structure in which a (meth) acryloyl group which is a polymerizable group is bonded to a polyether skeleton in a pendant form.
- a (meth) acryloyl group which is a polymerizable group is bonded to a polyether skeleton in a pendant form.
- the radical polymerizable polyether (A) for example, glycidyl ester (a) is obtained in the presence of trifluoromethanesulfonic acid metal salt (b) to effect ring-opening polymerization of tetrahydrofuran. What is represented by (1) is mentioned.
- the radical polymerizable polyether (A) represented by the following general formula (1) is polymerized using glycidyl (meth) acrylate and tetrahydrofuran using a protonic acid, an ion complex, a Lewis acid, an active halide, etc. as a catalyst, It can be produced by adding a compound having an active hydrogen such as alcohol having 1 to 20 carbon atoms or water to stop the reaction.
- M1 is 1 to 7000, m2 is 0 to 7000, n is 1 to 14000, R1 is a methyl group or hydrogen, R2 and R3 are hydrogen or a hydrocarbon group having 1 to 20 carbon atoms)
- the mass average molecular weight of the radical polymerizable polyether (A) is such that when copolymerized with the radical polymerizable vinyl monomer (B), the copolymer has excellent transparency, high elongation at break, high bending strength, etc. In order to impart the excellent mechanical properties, it is preferable to be 20000 to 1000000.
- the number average molecular weight is preferably 20000 to 100,000. Since the viscosity when mixed with the radical polymerizable vinyl monomer (B) is relatively low and the handling is good, the mass average molecular weight is preferably 20,000 to 200,000 and the number average molecular weight is more preferably 11,000 to 50,000.
- the degree of polymerization of the glycidyl (meth) acrylate-derived skeleton in the radical polymerizable polyether (A) is m1
- the degree of polymerization of the glycidyl ester-derived skeleton having no radical polymerizable functional group is m2
- the degree of polymerization of tetrahydrofuran is n.
- M1 / (m1 + m2 + n) is 0.0005 in order to impart a high elongation at break and high bending strength to the copolymer when copolymerized with the radical polymerizable vinyl monomer (B).
- ⁇ m1 / (m1 + m2 + n) ⁇ 0.20 is preferable.
- 0.001 ⁇ m1 / (m1 + m2 + n) ⁇ 0.10 is more preferable.
- Glycidyl ester (a) represented by the following general formula (2) used for the production of radical polymerizable polyether (A) is glycidol and one having 1 to 20 carbon atoms.
- the compound is not particularly limited as long as it is a compound in which a carboxylic acid composed of a linear or branched hydrocarbon group or a hydrocarbon group having an unsaturated bond having 2 to 10 carbon atoms is bonded by an ester bond.
- the presence of a carbonyl group near the epoxy skeleton is preferable because the oxygen polymerization activity of the epoxy group when the catalyst is coordinated is improved.
- R is a linear or branched hydrocarbon group having 1 to 20 carbon atoms, or a hydrocarbon group having an unsaturated bond having 2 to 10 carbon atoms
- glycidyl ester (a) examples include glycidyl acrylate, glycidyl methacrylate, glycidyl acetate, glycidyl propionate, glycidyl butyrate, and glycidyl stearate. These may be used alone or in combination of two or more.
- the amount of glycidyl ester (a) used can be appropriately determined within the range of 1 to 20 parts by mass with respect to 100 parts by mass of tetrahydrofuran. In order to increase the molecular weight of the resulting polyether copolymer, Is preferably 1.5 to 6.0 parts by mass.
- the trifluoromethanesulfonic acid metal salt (b) is not particularly limited as long as it has a property as a Lewis acid, and since it acts as a Lewis acid even in the presence of water, the metal is scandium.
- a trifluoromethanesulfonic acid metal salt hydrate or anhydride composed of a metal selected from at least one of yttrium and lanthanoid is preferred, and a trifluoromethanesulfonic acid metal salt hydrate composed of the metal is more preferred.
- ytterbium trifluoromethanesulfonate in which the metal is ytterbium (an element belonging to a lanthanoid), is more preferable because of its high activity for ring-opening polymerization of tetrahydrofuran.
- the amount of the trifluoromethanesulfonic acid metal salt (b) used is preferably 0.1 to 5 parts by mass, more preferably 0.2 to 5 parts by mass with respect to 100 parts by mass of tetrahydrofuran. In order to increase the molecular weight of the obtained polyether copolymer, and from the viewpoint of easy catalyst removal, 0.4 to 1.3 parts by mass is most preferable.
- the tetrahydrofuran used in the present invention is not particularly required to reduce the water content by a dehydration operation or the like, and tetrahydrofuran in a water-containing state may be used. Since the ring-opening polymerization may not proceed, the water content is preferably 1% by mass or less.
- a polymerization solvent may not be used, but an organic solvent such as hexane, heptane, octane, or diethyl ether may be used as the polymerization solvent.
- the polymerization temperature can be appropriately determined in the range of ⁇ 100 to 80 ° C. Room temperature to 50 ° C. is more preferable from the viewpoints of suppressing thermal runaway during ring-opening polymerization and suppressing volatilization of tetrahydrofuran.
- the polymerization time can be appropriately determined in the range of 0.1 hour to 144 hours.
- the polymerization time is preferably in the range of 0.5 to 72 hours from the standpoints of production cost and viscosity of the polymerization solution.
- a skeleton derived from the compound having active hydrogen is introduced at the end of the polyether copolymer, so that the polymerization reaction can be stopped.
- the compound having active hydrogen include water and linear or branched alcohols having 1 to 20 carbon atoms.
- the linear or branched alcohol having 1 to 20 carbon atoms include methanol, ethanol, propanol, isopropanol, n-butyl alcohol, iso-butyl alcohol, tert-butyl alcohol and the like.
- radical polymerization may occur depending on the reaction temperature, so polymerization of nitroxy radicals such as hydroquinone derivatives and TEMPO.
- An inhibitor may be added to the polymerization solution in advance. The addition amount is preferably 0.0001 to 1 part by mass with respect to 100 parts by mass of the glycidyl ester (a) having radical polymerizability used in the polymerization reaction.
- Radical polymerizable polyether (A) is blended with radical polymerizable vinyl monomer (B) and contains radical polymerizable polyether (A) and radical polymerizable vinyl monomer (B) as main components.
- the polymerizable composition (C) is obtained, and 99 to 1% by mass of the component (B) is blended with 1 to 99% by mass of the component (A).
- the total of the component (A) and the component (B) in the polymerizable composition (C) is preferably 50 to 100% by mass, more preferably 80 to 99% by mass.
- radical polymerizable vinyl monomer (B) a (meth) acrylic monomer, a styrene monomer or the like can be used.
- (Meth) acrylic monomer refers to a monomer composed of methacrylic acid ester or acrylic acid ester. Specifically, acrylic acid, methyl acrylate, ethyl acrylate, propyl acrylate, acrylic Butyl acrylate, isobutyl acrylate, t-butyl acrylate, pentyl acrylate, hexyl acrylate, cyclohexyl acrylate, 2-ethylhexyl acrylate, stearyl acrylate, methacrylic acid, methyl methacrylate, ethyl methacrylate, propyl methacrylate, Examples include butyl methacrylate, isobutyl methacrylate, t-butyl methacrylate, pentyl methacrylate, hexyl methacrylate, cyclohexyl methacrylate, 2-ethylhexyl methacrylate, and stearyl methacrylate. These can be used
- Styrene monomers include styrene, o-methylstyrene, m-methylstyrene, p-methylstyrene, ⁇ -methylstyrene, p-ethylstyrene, 2,4-dimethylstyrene, pn-butylstyrene, p -Tert-butyl styrene, pn-hexyl styrene, pn-octyl styrene, pn-nonyl styrene, pn-densyl styrene, pn-dodecyl styrene, p-phenyl styrene, 3,4 Mention may be made of dichlorostyrene. These can be used by appropriately selecting one or two or more as necessary.
- composition ratio of the radical polymerizable polyether (A) to the radical polymerizable vinyl monomer (B) in the polymerizable composition (C) is 1 to 99% by mass of the radical polymerizable polyether (A).
- the functional vinyl monomer (B) is in the range of 99 to 1% by mass, the formed copolymer has excellent mechanical properties such as excellent transparency, high elongation at break, and high bending strength. Become.
- composition ratio of the polymerizable composition (C) is 20% by mass or more for the radical polymerizable polyether (A) and 80% by mass or less for the radical polymerizable vinyl monomer (B).
- the copolymer is preferable because it has excellent mechanical properties such as excellent transparency, high elongation at break, and high bending strength, and excellent punchability at room temperature.
- composition ratio of the polymerizable composition (C) is 50 mass% or less for the radical polymerizable polyether (A) and 50 mass% or more for the radical polymerizable vinyl monomer (B).
- the copolymer has excellent mechanical properties such as excellent transparency, high elongation at break, and high bending strength, and a mixture of radical polymerizable polyether (A) and radical polymerizable vinyl monomer (B). This is preferable because the viscosity is relatively low and the handling property is excellent.
- the component polymerizable composition (C) of the polymerizable composition (C) contains a radical polymerizable polyether (A) and a radical polymerizable vinyl monomer (B) as main components.
- a radical polymerizable polyether (A) and a radical polymerizable vinyl monomer (B) as main components.
- polymerization mode photopolymerization, bulk thermal polymerization, solution polymerization, suspension polymerization, emulsion polymerization, and the like can be employed.
- polymerization is performed by appropriately adding water or an organic solvent.
- thermal polymerization initiator examples include benzoyl peroxide, lauroyl peroxide, t-butylperoxyisobutyrate, t-butylperoxy-2-ethylhexanoate, t-butylperoxyneodecanoate, t Organic peroxide polymerization initiators such as hexyl peroxypivalate, diisopropyl peroxydicarbonate, bis (4-t-butylcyclohexyl) peroxydicarbonate; 2,2′-azobisisobutyronitrile, 2 Azo polymerization initiators such as 2,2′-azobis (2,4-dimethylvaleronitrile) and 2,2′-azobis (4-methoxy-2,4-dimethylvaleronitrile). These may be used alone or in combination of two or more.
- photopolymerization initiator examples include 1-hydroxy-cyclohexyl phenyl ketone, 2-hydroxy-2-methyl-1-phenylpropan-1-one, methylphenylglyoxylate, acetophenone, benzophenone, diethoxyacetophenone, 2, 2-dimethoxy-2-phenylacetophenone, 1-phenyl-1,2-propane-dione-2- (o-ethoxycarbonyl) oxime, 2-methyl [4- (methylthio) phenyl] -2-morpholino-1-propanone Benzyl, benzoin methyl ether, benzoin ethyl ether, benzoin isopropyl ether, benzoin isobutyl ether, 2-chlorothioxanthone, isopropylthioxanthone, 2,4,6-trimethylbenzoyldiphenylphosphine oxa De, diphenylphosphine oxide, 2-methyl-benzoyl di
- the polymerizable composition (C) is blended with additives such as a release agent as necessary. can do.
- the blending amount of the mold release agent is preferably 0.001 part by mass or more with respect to 100 parts by mass of the polymerizable composition before adding the mold release agent and the polymerization initiator from the viewpoint of improving the peelability. 0.005 mass parts or more is more preferable.
- release agents include sodium dioctyl sulfosuccinate (trade name: Aerosol OT-100) manufactured by Nippon Cytec Industries, Ltd., diethyl phosphate and monoethyl phosphate manufactured by Johoku Chemical Industry Co., Ltd. And a 55:45 mixture of esters (trade name: JP-502).
- additives other than mold release agents lubricants, plasticizers, antibacterial agents, antifungal agents, light stabilizers, ultraviolet absorbers, bluing agents, dyes, antistatic agents, heat stabilizers, etc., as necessary
- plasticizers plasticizers
- antibacterial agents antifungal agents
- antifungal agents light stabilizers
- ultraviolet absorbers ultraviolet absorbers
- bluing agents dyes
- antistatic agents heat stabilizers, etc.
- the radically polymerizable polyether (A) and the radically polymerizable vinyl monomer (B) are converted into vinyl groups.
- a copolymer By shaping the polymerizable composition (C) and radically polymerizing the radically polymerizable composition (C), the radically polymerizable polyether (A) and the radically polymerizable vinyl monomer (B) are converted into vinyl groups. To obtain a copolymer.
- the polymerizable composition (C) into a desired shape as in (1) and (2) below, for example, radical polymerization is performed to form a molded body having a desired shape such as a film. can do.
- a mold having a cavity shape corresponding to a molded body is prepared, and a polymerizable composition (C) containing a polymerization initiator is injected into the cavity through a gasket or the like, and active energy is applied from one side or both sides of the mold.
- the polymerizable composition (C) is cured by radical polymerization by performing irradiation with radiation, heating, or the like.
- a coating film of the polymerizable composition (C) is continuously formed on the running belt, and is continuously irradiated with active energy rays, heated, etc. from one side or both sides of the belt.
- the coating film of the composition (C) is radical-polymerized and cured, and then the cured coating film is peeled from the belt.
- Polymers, molded products and films formed by radical polymerization of the polymerizable composition (C) have excellent mechanical properties such as excellent transparency, high elongation at break and high bending strength.
- the film in the case of forming a film by radical polymerization of the formed film polymerizable composition (C), it is preferable to form the film so as to have a thickness of 0.1 to 3000 ⁇ m.
- the thickness is 0.1 ⁇ m or more, the mechanical properties are excellent, and when it is 3000 ⁇ m or less, the bending properties are excellent.
- the thickness of the film is more preferably 200 ⁇ m to 1000 ⁇ m.
- this film has excellent mechanical properties such as excellent transparency, high breaking elongation, and high bending strength.
- the breaking elongation at 500 mm / min is preferably 10% or more, and the transparency is preferably 5% or less when the film thickness is 0.5 mm.
- Total light transmittance The total light transmittance was measured using a haze meter (trade name: NDH2000, manufactured by Nippon Denshoku Industries Co., Ltd.) in accordance with JIS K7361-1, an acrylic film cut into a 5 cm square. did.
- the mass mean molecular weight was 45000 by GPC measurement.
- the results of 1 H-NMR measurement are shown below. Proton peaks derived from the ring-opened product of glycidyl methacrylate were observed at 5.55, 6.15, 4.15, 4.05, and 1.9, and proton peaks derived from the ring-opened product of tetrahydrofuran were observed. 3.1 to 3.9 and 1.6 were observed. From the above, it was confirmed that the white solid obtained was a copolymer of poly (3-methacryloxypropene oxide) and polybutylene oxide having a (meth) acryloyl skeleton.
- Example 1 Preparation of Polymerizable Composition 20% by mass of radical polymerizable polyether produced in Production Example 1, 80% by mass of methyl methacrylate (trade name: Acryester M, manufactured by Mitsubishi Rayon Co., Ltd.), radical polymerizable polyether and methacrylic Polymerization was carried out by adding 0.3% by mass of 1-hydroxy-cyclohexyl-phenyl-ketone (trade name: Irgacure 184, manufactured by BASF Japan) as a polymerization initiator to 100% by mass of the monomer mixture combined with methyl acid. Sex composition was obtained.
- methyl methacrylate trade name: Acryester M, manufactured by Mitsubishi Rayon Co., Ltd.
- Irgacure 184 1-hydroxy-cyclohexyl-phenyl-ketone
- sodium dioctylsulfosuccinate (trade name: Aerosol OT-100, manufactured by Nippon Cytec Industries Co., Ltd.) was added and mixed as a mold release agent, and then degassed under reduced pressure.
- Example 2 Except having changed the usage-amount of the radically polymerizable polyether manufactured in manufacture example 1 into the usage-amount shown in Table 2, it carried out similarly to Example 1 and obtained the acrylic film. The evaluation results are shown in Table 2.
- the radically polymerizable polyether had a mass average molecular weight as low as less than 20000, and the copolymer had poor transparency and bending properties.
- Example 1 ' An acrylic film was obtained in the same manner as in Example 1 except that the radical polymerizable polyether produced in Production Example 1 was changed to the radical polymerizable polyether produced in Production Example 1 ′. The evaluation results are shown in Table 2.
- Example 1 [Weather resistance evaluation] Moreover, the weather resistance test was implemented about the film obtained in Example 1 and Example 1 '. The acrylic film cut into 3 cm square was subjected to a weather resistance test using a die plastic metal weather KU-R4-W type (manufactured by Daipura Wintes Co., Ltd.).
- Test cycle irradiation 4 hours (humidity: 70% RH, black panel temperature: 63 ° C.) / Condensation 4 hours (humidity: 98% RH, black panel temperature: 30 ° C.) / 10 seconds shower (70 ° C.) / Dark 4
- the optical properties of the film after 96 hours were evaluated under the conditions of time (humidity: 70% RH, black panel temperature: 65 ° C.) / 10 seconds shower (30 ° C.), UV intensity: 140 mW / cm 2 .
- the evaluation results are shown in Table 4.
- Example 1 ′ maintained a high total light transmittance and a low value of HAZE even after the weather resistance test.
- Example 1 ' since the radical polymerizable polyether (A) was washed with water after synthesis, deterioration of the optical performance after the weather resistance test of the produced film was suppressed.
- Example 5 Preparation of Polymerizable Composition 20% by mass of radical polymerizable polyether produced in Production Example 1, 80% by mass of methyl methacrylate (trade name: Acryester M, manufactured by Mitsubishi Rayon Co., Ltd.), radical polymerizable polyether and methacrylic As a polymerization initiator, 0.3% by mass of t-hexylperoxypivalate (trade name: perhexyl PV) was added to 100% by mass of the monomer mixture combined with methyl acid to obtain a polymerizable composition.
- methyl methacrylate trade name: Acryester M, manufactured by Mitsubishi Rayon Co., Ltd.
- t-hexylperoxypivalate trade name: perhexyl PV
- sodium dioctylsulfosuccinate (trade name: Aerosol OT-100, manufactured by Nippon Cytec Industries, Ltd.) was added and mixed as a release agent, and then degassing was performed under reduced pressure.
- Charpy impact test The Charpy impact strength of the copolymer obtained in Example 5 was measured in accordance with JIS-K7111. The measurement was carried out under conditions of 8 cm long ⁇ 1 cm wide ⁇ 3 mm thick, flat wise and notched. The evaluation results are shown in Table 3.
- Drop weight impact test The impact resistance of the copolymer obtained in Example 5 was evaluated using a DuPont impact tester. As a sample, a resin plate cut into a square with a side of 50 mm was used, and a 50% fracture energy was evaluated according to the standard of JIS-K7211, using a 500 g weight with a punch radius of 7.9 mm and a usus radius of 15.2 mm. The evaluation results are shown in Table 3.
- the mass mean molecular weight was 33800 by GPC measurement.
- the results of 1 H-NMR measurement are shown below. Proton peaks derived from ring-opened glycidyl methacrylate were observed at 5.55, 6.15, 4.15, 4.05, and 1.9, and proton peaks derived from ring-opened tetrahydrofuran. Were observed at 3.1 to 3.9 and 1.6. From the above, it was confirmed that the white solid obtained was a copolymer of poly (3-methacryloxypropene oxide) and polybutylene oxide having a (meth) acryloyl skeleton.
- the (co) polymer comprising a radical polymerizable polyether and a radical polymerizable vinyl monomer has excellent characteristics as described above, so that a flexible display front plate, a solar cell substrate, an organic EL substrate It can be suitably used as a dense optical member such as a lighting cover, a liquid crystal display front plate, and a light guide sheet.
Abstract
Description
1)グリシジル(メタ)アクリレートとテトラヒドロフランとを開環共重合して得られるラジカル重合性ポリエーテル及びその製造方法、
2)該ラジカル重合性ポリエーテルとラジカル重合性ビニル系単量体とを含有する重合性組成物、並びに
3)該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム
に係わり、ラジカル重合性ビニル系単量体とラジカル重合することで、優れた透明性及び高い破断伸度合、高い曲げ強度などの優れた機械特性を有する共重合体が形成できるラジカル重合性ポリエーテル及びその製造方法を提供し、また、該ラジカル重合性ポリエーテルとラジカル重合性ビニル系単量体とを含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルムを提供するものである。
本発明のラジカル重合性ポリエーテル(A)は、ポリエーテル骨格に対し、重合性基である(メタ)アクリロイル基がペンダント状に結合した構造を有する。前記ラジカル重合性ポリエーテル(A)としては、例えばグリシジルエステル(a)を、トリフルオロメタンスルホン酸金属塩(b)の存在下で作用させてテトラヒドロフランの開環重合を行って得られる、下記一般式(1)で表されるものが挙げられる。
ラジカル重合性ポリエーテル(A)の製造のために用いる下記一般式(2)で表されるグリシジルエステル(a)は、グリシドールと炭素数1~20の直鎖状または分岐を有する炭化水素基、炭素数2~10の不飽和結合を有する炭化水素基からなるカルボン酸がエステル結合で結合した化合物であればよく、特に限定されるものではない。エポキシ基骨格のそばにカルボニル基が存在することにより、触媒が配位した際のエポキシ基の酸素の重合活性が向上するので好ましい。
ラジカル重合性ポリエーテル(A)にラジカル重合性ビニル系単量体(B)を配合して、ラジカル重合性ポリエーテル(A)及びラジカル重合性ビニル系単量体(B)を主成分として含有する重合性組成物(C)を得るが、該(A)成分1~99質量%に対して、該(B)成分を99~1質量%配合する。重合性組成物(C)中の該(A)成分及び該(B)成分の合計は、50~100質量%が好ましく、80~99質量%がより好ましい。
重合性組成物(C)は、ラジカル重合性ポリエーテル(A)及びラジカル重合性ビニル系単量体(B)を主成分として含有するものであるが、重合開始剤としてアゾ系開始剤、過酸化物系開始剤などを適宜選択して添加することにより、ラジカル重合性ポリエーテル(A)とラジカル重合性ビニル系単量体(B)とをラジカル重合することができる。
重合性組成物(C)をラジカル重合することにより、ラジカル重合性ポリエーテル(A)及びラジカル重合性ビニル系単量体(B)をビニル基で反応させ、共重合体を得ることができる。
重合性組成物(C)をラジカル重合してフィルムを形成する場合、フィルムの厚みは0.1~3000μmとなるように形成するのが好ましい。フィルムの0.1μm以上であると機械特性に優れ、また、3000μm以下であると曲げ特性に優れる。フィルムの厚みは、200μm~1000μmであることがより好ましい。
化合物の構造を1H-NMRスペクトルにより確認した。
測定溶媒にCDCl3、基準物質にテトラメチルシランを用い、核磁気共鳴装置(JNM EX-270:日本電子(株)製)を使用して測定した。測定温度は室温、積算回数は16回で測定した。
重合体の数平均分子量(Mn)、質量平均分子量(Mw)及び分子量分布(Mw/Mn)を、ポリスチレンをスタンダードとして、GPC(HLC-8220:東ソー(株)製、カラム:TSK-GEL SUPER H-4000とTSK-GEL SUPER H-2000の直列接続)により測定した。
溶離液:クロロホルム、測定温度:40℃、流速:0.6mL/minの条件で測定した。
5cm角に切り出したアクリルフィルムをJIS K7361-1に準拠して、ヘイズメーター(商品名:NDH2000、日本電色工業(株)製)を用いて全光線透過率を測定した。
5cm角に切り出したアクリルフィルムをJIS K7105に準拠して、ヘイズメーター(商品名:NDH2000、日本電色工業(株)製)を用いてヘイズ値を測定した。
スーパーダンベルカッター(商品名:SDK-100D、(株)ダンベル製)を用いてダンベル状1号型のアクリルフィルムの試験片を作製し、両端を切断した。得られた試験片について、動的粘弾性測定装置(商品名:EXSTARDMS6100、エスアイアイ・ナノテクノロジー(株)製)を使用して、周波数1Hz、温度範囲30~150℃、昇温速度2℃/minの測定条件で測定し、得られた温度-tanδ曲線の極大値を示すときの温度をガラス転移温度(Tg)とした。
JIS K6251に準拠して、スーパーダンベルカッター(商品名:SDK-100D、(株)ダンベル製)を用いてダンベル状1号型のアクリルフィルムの試験片を5枚作製した。得られた試験片について、引張試験機(商品名:ストログラフT、(株)東洋精機製作所製)を使用して室温23℃及び引張速度500mm/minで5回の引張試験を実施し、その時の応力歪み曲線の接線の平均値を求め、弾性率とした。
JIS K6251に準拠して、スーパーダンベルカッター(商品名:SDK-100D、(株)ダンベル製)を用いてダンベル状1号型のアクリルフィルムの試験片を5枚作製した。得られた試験片について、引張試験機(商品名:ストログラフT、(株)東洋精機製作所製)を使用して室温23℃及び引張速度500mm/minで5回の引張試験を実施し、破断伸度を平均値として求めた。
5cm角に切り出したアクリルフィルムを用いて、90°曲げを行った。具体的には積層品を5cm×5cmの大きさに切り出し、90°曲率半径が1mmとなるように、2秒間で折り曲げた。目視により下記の2段階評価を行った。
×:フィルム折曲時もしくは折曲途中で破断。
○:破断、白化が無く良好。
攪拌子、温度計、冷却管を備えた1000mlの三口フラスコにテトラヒドロフラン(和光純薬工業(株)製)600g、トリフルオロメタンスルホン酸イッテルビウム(III)水和物(東京化成工業(株)製)2gを添加して、室温で攪拌した。イッテルビウムトリフラート水和物が溶解したのを確認後、グリシジルメタクリレート24g(和光純薬工業(株)製)を添加した。添加後、室温(25℃)で11時間攪拌した。攪拌後、エタノール50g(和光純薬工業(株)製)を添加して反応を停止させた。反応終了後、未反応のテトラヒドロフランや余剰のエタノールをエバポレーターを使用して、留去することで白色固体437g(収率70%)を得た。
1H-NMR
0.95(s´)1.60(m),1.9(s),2.35(s´)3.1-3.9(m),4.05(s),4.25(m),5.55(s),6.13(s)
テトラヒドロフラン、グリシジルメタクリレート、イッテルビウムトリフラート水和物、エタノールの使用量と反応時間を表1に示す使用量と反応時間を変更したこと以外は、製造例1と同様に行った。GPC測定により質量平均分子量は14500であった。また、グリシジルメタクリレート由来骨格の重合度mとテトラヒドロフランの重合度nとしたときのグリシジルメタクリレート由来骨格の導入率m1/(m1+m2+n)は、0.081であった。
製造例1で得られた白化固体60gを分液漏斗に移し、酢酸エチル90ml、純水90mlを加えて分液操作を行った。洗浄後の有機層を無水硫酸マグネシウムで脱水した。これをろ過後、エバポレーターを使用して、酢酸エチルを留去することでラジカル重合ポリエーテルである白色固体を得た。GPC測定と1H-NMRを測定した結果は製造例1と同一であった。
重合性組成物の調製
製造例1で製造したラジカル重合性ポリエーテル20質量%、メタクリル酸メチル80質量%(商品名:アクリエステルM、三菱レイヨン(株)製)、ラジカル重合性ポリエーテルとメタクリル酸メチルを合わせたモノマー混合物100質量%に対して重合開始剤として1-ヒドロキシ-シクロヘキシル-フェニル-ケトン(商品名:イルガキュア184、BASFジャパン(株)製)0.3質量%を添加して重合性組成物を得た。さらに離型剤としてジオクチルスルホコハク酸ナトリウム(商品名:エアロゾルOT-100、日本サイテックインダストリーズ(株)製)0.05質量%を添加、混合し、その後、減圧下で脱気処理を行った。
製造例1で製造したラジカル重合性ポリエーテルの使用量を表2に示す使用量に変更したこと以外は、実施例1と同様に行い、アクリルフィルムを得た。評価結果を表2に示す。
製造例1で製造したラジカル重合性ポリエーテルを製造例2で製造したラジカル重合性ポリエーテルに変更したこと以外は、実施例1と同様に行い、アクリルフィルムを得た。評価結果を表2に示す。
製造例1で製造したラジカル重合性ポリエーテルを製造例1'で製造したラジカル重合性ポリエーテルに変更したこと以外は、実施例1と同様に行い、アクリルフィルムを得た。評価結果を表2に示す。
また、実施例1と実施例1'で得られたフィルムについては、耐候試験を実施した。3cm角に切り出したアクリルフィルムをダイプラ・メタルウエザーKU-R4-W型(ダイプラ・ウィンテス(株)製)にて耐候性試験を行った。試験サイクルは、照射4時間(湿度:70%RH、ブラックパネル温度:63℃)/結露4時間(湿度:98%RH、ブラックパネル温度:30℃)/10秒シャワー(70℃)/暗黒4時間(湿度:70%RH、ブラックパネル温度:65℃)/10秒シャワー(30℃)、UV強度:140mW/cm2の条件で、96時間経過後のフィルムの光学特性を評価した。評価結果を表4に示す。
重合性組成物の調製
製造例1で製造したラジカル重合性ポリエーテル20質量%、メタクリル酸メチル80質量%(商品名:アクリエステルM、三菱レイヨン(株)製)、ラジカル重合性ポリエーテルとメタクリル酸メチルを合わせたモノマー混合物100質量%に対して重合開始剤としてt‐へキシルパーオキシピバレート(商品名:パーヘキシルPV)0.3質量%を添加して重合性組成物を得た。さらに離型剤としてジオクチルスルホコハク酸ナトリウム(商品名:エアロゾルOT-100、日本サイテックインダストリーズ(株)製)0.05質量%を添加、混合し、その後、減圧下で脱気処理を行った。
実施例5で得られた共重合体については、耐衝撃性を評価した。
実施例5で得られた共重合体について、JIS‐K7111も準拠してシャルピー衝撃強度を測定した。測定は、長さ8cm×幅1cm×厚さ3mmの試験片を切出し、フラットワイズ、ノッチ無しの条件で実施した。評価結果を表3に示す。
実施例5で得られた共重合体について、デュポン衝撃試験機によって耐衝撃性を評価した。サンプルとして一辺50mmの正方形に切断した樹脂板を用い、ポンチ半径7.9mm、ウス半径15.2mmにて500gのおもりを用いてJIS‐K7211の規格に準拠して50%破壊エネルギーを評価した。評価結果を表3に示す。
*テトラヒドロフラン(和光純薬工業(株)製)
*トリフルオロメタンスルホン酸イッテルビウム(III)水和物(東京化成工業(株)製)
*グリシジルメタクリレート(和光純薬工業(株)製)
*エタノール(和光純薬工業(株)製)
*メタクリル酸メチル(商品名:アクリエステルM、三菱レイヨン(株)製)
*1-ヒドロキシ-シクロヘキシル-フェニル-ケトン(商品名:イルガキュア184、BASFジャパン(株)製)
*ジオクチルスルホコハク酸ナトリウム(商品名:エアロゾル OT-100、日本サイテックインダストリーズ(株)製)
*酢酸エチル(和光純薬工業(株)製)
*t‐へキシルパーオキシピバレート(商品名:パーヘキシルPV、日油(株)製)
以下、本発明について、実施例を挙げて具体的に説明する。
化合物の構造を1H-NMRスペクトルにより確認した。
測定溶媒にCDCl3、基準物質にテトラメチルシランを用い、核磁気共鳴装置(JNM EX-270:日本電子(株)製)を使用して測定した。測定温度は室温、積算回数は16回で測定した。
重合体の数平均分子量(Mn)、質量平均分子量(Mw)及び分子量分布(Mw/Mn)を、ポリスチレンをスタンダードとして、GPC(HLC-8220:東ソー(株)製、カラム:TSK-GEL SUPER H-4000とTSK-GEL SUPER H-2000の直列接続)により測定した。
溶離液:クロロホルム、測定温度:40℃、流速:0.6mL/minの条件で測定した。
重合時に仕込んだテトラヒドロフラン、メタクリル酸グリシジルの質量の合計とエバポレーター後に回収された固体の質量から算出した。
(2)、(3)の結果をもとに下記の基準で重合性判定を行った。
◎:数平均分子量15000以上かつ収率40%以上
○:数平均分子量15000以上または収率40%以上
×:数平均分子量15000未満または/かつ収率40%未満
攪拌子、温度計、冷却管を備えた1000mlの三口フラスコにテトラヒドロフラン(和光純薬工業(株)製)100g、トリフルオロメタンスルホン酸イッテルビウム(III)水和物((東京化成工業(株)製)0.67gを添加して、室温で攪拌した。トリフルオロメタンスルホン酸イッテルビウム(III)水和物が溶解したのを確認後、メタクリル酸グリシジル4g(和光純薬工業(株)製)を添加した。添加後、室温(25℃)で7時間攪拌した。攪拌後、エタノール8.3g(和光純薬工業(株)製)を添加して反応を停止させた。反応終了後、未反応のテトラヒドロフランや余剰のエタノールをエバポレーターを使用して、留去することで白色固体52g(収率50%)を得た。
1H-NMR
0.95(s´)1.60(m),1.9(s),2.35(s´)3.1-3.9(m),4.05(s),4.25(m),5.55(s),6.13(s)
テトラヒドロフラン、メタクリル酸グリシジル、トリフルオロメタンスルホン酸イッテルビウム(III)水和物、エタノールの使用量と反応時間を表5に示す使用量と反応時間を変更したこと以外は、製造方法実施例1と同様に行った。収率は57%で、GPC測定により質量平均分子量は40900であった。
メタクリル酸グリシジルをR-グリシジルブチレート(東京化成工業(株))に変更し、テトラヒドロフラン、トリフルオロメタンスルホン酸イッテルビウム(III)水和物、エタノールの使用量と反応時間を表5に示す使用量と反応時間を変更したこと以外は、製造方法実施例1と同様に行った。収率は55%で、GPC測定により質量平均分子量は30400であった。
テトラヒドロフラン、メタクリル酸グリシジル、トリフルオロメタンスルホン酸イッテルビウム(III)水和物、エタノールの使用量と反応時間を表5に示す使用量と反応時間を変更したこと以外は、製造方法実施例1と同様に行った。収率は39%で、GPC測定により質量平均分子量は23400であった。
メタクリル酸グリシジルを4-ヒドロキシブチルアクリル酸グリシジルエーテル(日本化成(株))に変更し、テトラヒドロフラン、トリフルオロメタンスルホン酸イッテルビウム(III)水和物、エタノールの使用量と反応時間を表5に示す使用量と反応時間を変更したこと以外は、製造方法実施例1と同様に行った。収率は27%で、GPC測定により質量平均分子量は16700であった。グリシジルエステルをグリシジルエーテルとしたことでカルボニル基がグリシジル基に隣接していないために重合活性向上がなく、製造方法実施例1と比較して、分子量と収率が低かった。
Claims (17)
- 質量平均分子量が20000~100000であり、(メタ)アクリロイル基をペンダント基として有するポリアルキレンエーテル骨格及びポリテトラメチレンエーテル骨格を含むラジカル重合性ポリエーテル(A)。
- 前記一般式(1)のm1とm2とnが0.0005≦m1/(m1+m2+n)≦0.20の関係にある請求項2記載のラジカル重合性ポリエーテル(A)。
- 前記一般式(1)のm1とm2とnが0.001≦m1/(m1+m2+n)≦0.10の関係にある請求項2記載のラジカル重合性ポリエーテル(A)。
- 請求項1~4のいずれかに記載のラジカル重合性ポリエーテル(A)及びラジカル重合性ビニル系単量体(B)を主成分として含有し、該(A)成分1~99質量%に対して、該(B)成分を99~1質量%配合してなる重合性組成物(C)。
- ラジカル重合性ビニル系単量体(B)が(メタ)アクリル系単量体である請求項5記載の重合性組成物(C)。
- 請求項5又は6記載の重合性組成物(C)をラジカル重合して形成される共重合体。
- 請求項5又は6記載の重合性組成物(C)を所望の形状に賦形した後、ラジカル重合して形成される成形体。
- 請求項5又は6記載の重合性組成物(C)をラジカル重合して形成されるフィルム。
- フィルムの厚みが0.1~3000μmである請求項9記載のフィルム。
- 引張速度500mm/minのときの破断伸度が10%以上である請求項9又は10に記載のフィルム。
- フィルムの厚みが0.5mmのときのヘイズが5%以下である請求項9~11のいずれかに記載のフィルム。
- グリシジルエステル(a)とトリフルオロメタンスルホン酸金属塩(b)との質量比(a)/(b)が2~9である請求項13に記載の製造方法。
- グリシジルエステル(a)がメタクリル酸グリシジルである請求項13又は14に記載の製造方法。
- トリフルオロメタンスルホン酸金属塩(b)の金属がスカンジウム、イットリウム及びランタノイドからなる群より選ばれる一種以上である請求項13~15のいずれかに記載の製造方法。
- トリフルオロメタンスルホン酸金属塩(b)がトリフルオロメタンスルホン酸イッテルビウム水和物である請求項13~16のいずれかに記載の製造方法。
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201480019422.1A CN105102504B (zh) | 2013-02-13 | 2014-02-13 | 自由基聚合性聚醚及其制造方法、聚合性组合物、共聚物、成型体和薄膜 |
| US14/767,326 US9718919B2 (en) | 2013-02-13 | 2014-02-13 | Radically polymerizable polyether, method for producing said radically polymerizable polyether, polymerizable composition comprising said radically polymerizable polyether and radically polymerizable vinyl monomer, and copolymer, molded article and film each formed by radical polymerization of said polymerizable composition |
| JP2014508410A JP6229205B6 (ja) | 2013-02-13 | 2014-02-13 | ラジカル重合性ポリエーテル、該ラジカル重合性ポリエーテルの製造方法、該ラジカル重合性ポリエーテル及びラジカル重合性ビニル系単量体を含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-025380 | 2013-02-13 | ||
| JP2013025380 | 2013-02-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014126157A1 true WO2014126157A1 (ja) | 2014-08-21 |
Family
ID=51354156
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/053344 WO2014126157A1 (ja) | 2013-02-13 | 2014-02-13 | ラジカル重合性ポリエーテル、該ラジカル重合性ポリエーテルの製造方法、該ラジカル重合性ポリエーテル及びラジカル重合性ビニル系単量体を含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US9718919B2 (ja) |
| JP (1) | JP6229205B6 (ja) |
| CN (1) | CN105102504B (ja) |
| WO (1) | WO2014126157A1 (ja) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015040257A (ja) * | 2013-08-22 | 2015-03-02 | 三菱レイヨン株式会社 | 透明粘着フィルム、フラットパネルディスプレイ及び太陽電池モジュール |
| JP2015168799A (ja) * | 2014-03-10 | 2015-09-28 | 三菱レイヨン株式会社 | 硬化型接着剤 |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5981302A (ja) * | 1982-10-01 | 1984-05-11 | インペリアル・ケミカル・インダストリ−ズ・ピ−エルシ− | 全体が非イオン系の立体的に安定化した水性重合体分散物の製造方法 |
| JPS6119229A (ja) * | 1984-07-05 | 1986-01-28 | Mitsubishi Electric Corp | 利得制御装置 |
| JPH09500162A (ja) * | 1993-07-16 | 1997-01-07 | イー・アイ・デユポン・ドウ・ヌムール・アンド・カンパニー | ケイ素化合物促進剤を使用する環状エーテル重合 |
| JPH09263632A (ja) * | 1996-03-29 | 1997-10-07 | Hodogaya Chem Co Ltd | アクリル基と水酸基を含有するポリオキシアルキレン誘 導体 |
| JP2001295181A (ja) * | 2000-04-07 | 2001-10-26 | Miyoshi Oil & Fat Co Ltd | アクリル系繊維用帯電防止剤及びアクリル系繊維の耐久帯電防止処理方法 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3816279A (en) * | 1968-08-20 | 1974-06-11 | American Can Co | Photopolymerization of mixtures of epoxide materials and tetrahydrofuran |
| GB8321119D0 (en) * | 1982-10-01 | 1983-09-07 | Ici Plc | Polymerisation process |
| JPS61192729A (ja) * | 1985-02-22 | 1986-08-27 | Mitsui Toatsu Chem Inc | 反応性のオリゴマ− |
-
2014
- 2014-02-13 JP JP2014508410A patent/JP6229205B6/ja active Active
- 2014-02-13 WO PCT/JP2014/053344 patent/WO2014126157A1/ja active Application Filing
- 2014-02-13 CN CN201480019422.1A patent/CN105102504B/zh active Active
- 2014-02-13 US US14/767,326 patent/US9718919B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5981302A (ja) * | 1982-10-01 | 1984-05-11 | インペリアル・ケミカル・インダストリ−ズ・ピ−エルシ− | 全体が非イオン系の立体的に安定化した水性重合体分散物の製造方法 |
| JPS6119229A (ja) * | 1984-07-05 | 1986-01-28 | Mitsubishi Electric Corp | 利得制御装置 |
| JPH09500162A (ja) * | 1993-07-16 | 1997-01-07 | イー・アイ・デユポン・ドウ・ヌムール・アンド・カンパニー | ケイ素化合物促進剤を使用する環状エーテル重合 |
| JPH09263632A (ja) * | 1996-03-29 | 1997-10-07 | Hodogaya Chem Co Ltd | アクリル基と水酸基を含有するポリオキシアルキレン誘 導体 |
| JP2001295181A (ja) * | 2000-04-07 | 2001-10-26 | Miyoshi Oil & Fat Co Ltd | アクリル系繊維用帯電防止剤及びアクリル系繊維の耐久帯電防止処理方法 |
Non-Patent Citations (2)
| Title |
|---|
| N. CHAGNEUX ET AL.: "Synthesis of Laterally Linked Poly(tetrahydrofuran)-Poly (methylmethacrylate) Block Copolymers via Use of a ''Jekyll and Hyde", COMONOMER, MACROMOLECULES, vol. 40, no. 9, pages 3183 - 3189 * |
| TAKEO SAEGUSA ET AL.: "Cationic Homo- and Co-Polymerizations of Ethyl Glycidate", POLYMER JOURNAL, vol. 11, no. 6, June 1979 (1979-06-01), pages 463 - 469 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015040257A (ja) * | 2013-08-22 | 2015-03-02 | 三菱レイヨン株式会社 | 透明粘着フィルム、フラットパネルディスプレイ及び太陽電池モジュール |
| JP2015168799A (ja) * | 2014-03-10 | 2015-09-28 | 三菱レイヨン株式会社 | 硬化型接着剤 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP6229205B6 (ja) | 2018-06-27 |
| JPWO2014126157A1 (ja) | 2017-02-02 |
| US9718919B2 (en) | 2017-08-01 |
| CN105102504B (zh) | 2017-08-04 |
| JP6229205B2 (ja) | 2017-11-15 |
| US20160002399A1 (en) | 2016-01-07 |
| CN105102504A (zh) | 2015-11-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5281710B2 (ja) | 硬化性樹脂組成物、その硬化物並びに光学材料 | |
| TW200846380A (en) | (Meth)acrylate copolymer for syrup and resin composition thereof | |
| KR101852217B1 (ko) | 아크릴계 조성물, 성형체, 휴대 전화 키 시트 및 도광체 | |
| EP2479214B1 (en) | Polymerizable composition and acrylic resin films | |
| JP6438882B2 (ja) | 共重合体の製造方法 | |
| KR20150134398A (ko) | 경화성 수지 조성물, 경화물 및 광학 물품 | |
| JP6229205B6 (ja) | ラジカル重合性ポリエーテル、該ラジカル重合性ポリエーテルの製造方法、該ラジカル重合性ポリエーテル及びラジカル重合性ビニル系単量体を含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム | |
| JP6230532B2 (ja) | メタクリル樹脂組成物並びにその成形品および製造方法 | |
| JP6657619B2 (ja) | 成形体 | |
| JPWO2014126157A6 (ja) | ラジカル重合性ポリエーテル、該ラジカル重合性ポリエーテルの製造方法、該ラジカル重合性ポリエーテル及びラジカル重合性ビニル系単量体を含有する重合性組成物、並びに該重合性組成物をラジカル重合して形成される共重合体、成形体及びフィルム | |
| KR101728922B1 (ko) | 광학 부품용 수지 원료 조성물, 광학 부품용 수지 및 광학 부품 | |
| JP6163962B2 (ja) | 透明粘着フィルム、フラットパネルディスプレイ及び太陽電池モジュール | |
| JP2012184371A (ja) | シリコーン樹脂組成物及びその成形体 | |
| TWI572662B (zh) | 熱可塑性樹脂組成物及其所形成的成型品 | |
| JP6780722B2 (ja) | 分散型アクリル共重合体、分散型アクリル共重合体の製造方法、アクリルシート、及びアクリルシートの製造方法 | |
| JP6269190B2 (ja) | 硬化型接着剤 | |
| JP2013067792A (ja) | ラジカル重合性化合物及びラジカル硬化性組成物 | |
| JP5596390B2 (ja) | アルコール性水酸基を有する可溶性多官能(メタ)アクリル酸エステル共重合体及びその製造方法 | |
| JPS61151212A (ja) | メタクリル系共重合体の製造法 | |
| JP5485095B2 (ja) | メタクリル系重合体組成物および成形品 | |
| JP6398278B2 (ja) | (メタ)アクリル系樹脂キャスト板用組成物、この組成物を注型重合して得られる(メタ)アクリル系樹脂キャスト板及びこの組成物を注型重合する(メタ)アクリル系樹脂キャスト板の製造方法 | |
| JP2023137519A (ja) | ポリデカメチレングリコールジ(メタ)アクリレート、重合体、樹脂組成物、単量体組成物、及び製造方法 | |
| JP2019189733A (ja) | メタクリル系樹脂組成物及びその製造方法 | |
| JPH03218335A (ja) | ポリブチレングリコールジメタクリレート及びそれを含む可撓性・低吸水性組成物 | |
| JP2005290167A (ja) | メタクリル樹脂組成物および透明部材 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480019422.1 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 2014508410 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14751571 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14767326 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14751571 Country of ref document: EP Kind code of ref document: A1 |