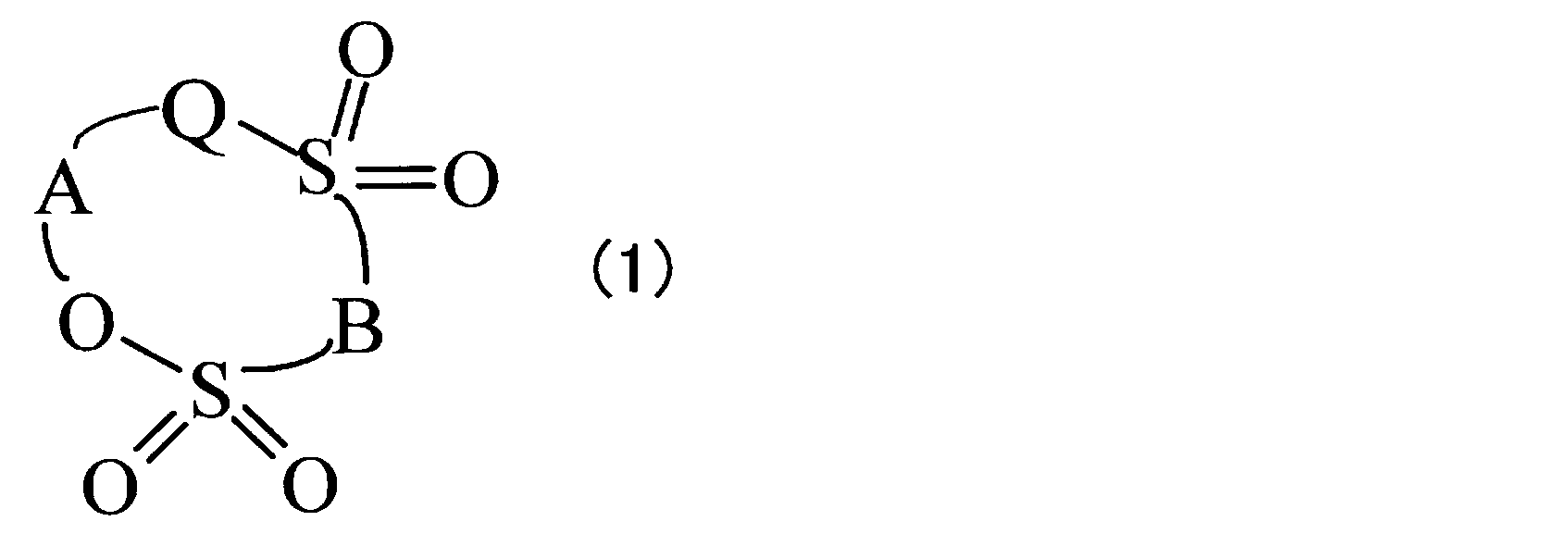WO2011096572A1 - 非水電解液二次電池 - Google Patents
非水電解液二次電池 Download PDFInfo
- Publication number
- WO2011096572A1 WO2011096572A1 PCT/JP2011/052615 JP2011052615W WO2011096572A1 WO 2011096572 A1 WO2011096572 A1 WO 2011096572A1 JP 2011052615 W JP2011052615 W JP 2011052615W WO 2011096572 A1 WO2011096572 A1 WO 2011096572A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- negative electrode
- mass
- secondary battery
- binder
- styrene
- Prior art date
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0567—Liquid materials characterised by the additives
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
- H01M4/583—Carbonaceous material, e.g. graphite-intercalation compounds or CFx
- H01M4/587—Carbonaceous material, e.g. graphite-intercalation compounds or CFx for inserting or intercalating light metals
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/621—Binders
- H01M4/622—Binders being polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
- Such a lithium secondary battery includes, for example, a positive electrode plate having a current collector that holds a positive electrode active material that absorbs and releases lithium ions, and a current collector that holds a negative electrode active material that absorbs and releases lithium ions.
- a negative electrode plate an electrolyte in which a lithium salt such as LiBF 4 or LiPF 6 is dissolved in an aprotic organic solvent, and a separator that is interposed between the positive electrode plate and the negative electrode plate to prevent a short circuit.
- Such a lithium secondary battery has recently been increasingly used not only in a room temperature environment but also in an electronic device used in a wide temperature range environment.
- a lithium secondary battery has recently been increasingly used not only in a room temperature environment but also in an electronic device used in a wide temperature range environment.
- the central processing unit increases in speed, the temperature inside the computer increases, and the battery is used for a long time in a high temperature environment.
- Mobile phones and portable devices are also increasingly used in high temperature environments. Therefore, there has been a strong demand for improving the cycle life of lithium secondary batteries that are repeatedly used in such a high temperature environment.
- the stability with respect to the reaction between the electrode active material and the electrolytic solution is insufficient, and the adhesion of the electrode cannot be maintained sufficiently, or the movement of lithium ions is inhibited. . For this reason, there has been a problem that the capacity retention rate decreases when charging and discharging are repeated for a long time in a high temperature environment.
- An object of the present invention is to provide a non-aqueous electrolyte secondary battery that exhibits a sufficient capacity retention rate even when charging and discharging cycles are repeated in a high temperature environment for a long period of time.
- One embodiment is: A positive electrode capable of inserting and extracting lithium ions; A negative electrode active material layer containing at least a styrene-based polymer as a binder and capable of occluding and releasing lithium ions, wherein the content of the styrene-based polymer is based on the total mass of the negative electrode active material layer A negative electrode of 0.3 to 8.0% by mass with respect to A nonaqueous electrolytic solution containing at least 0.002 to 5.0% by mass of a cyclic sulfonic acid ester having two sulfonyl groups based on the total mass of the nonaqueous electrolytic solution; A separator provided between the positive electrode and the negative electrode; The present invention relates to a non-aqueous electrolyte secondary battery having
- the electrolyte solution does not easily penetrate into the styrene polymer, which is a binder for the negative electrode, contact between the negative electrode active material and the electrolytic solution through the binder is prevented, thereby preventing a side reaction between the negative electrode active material and the electrolytic solution. be able to.
- a cyclic sulfonic acid ester having at least two sulfonyl groups in the electrolytic solution a stable surface film is formed on the surface of the negative electrode active material, and decomposition of the electrolytic solution solvent in a high temperature environment can be prevented. .
- the electrolytic solution is less likely to permeate into the styrene-based polymer, it is possible to prevent the negative electrode active material and the electrolytic solution from contacting with each other through the binder, thereby preventing a side reaction between the negative electrode active material and the electrolytic solution.
- coat is formed in the negative electrode active material surface, and decomposition
- a film having high electrical conductivity is formed at the negative electrode binder / electrolyte interface by containing the styrene polymer and the cyclic sulfonate in a specific range of content in the negative electrode and the non-aqueous electrolyte, respectively. Therefore, when the lithium ions move in the negative electrode, the lithium ions can be smoothly moved through the inside of the film, not the binder layer in which the electrolytic solution is less likely to permeate the lithium ions. For this reason, it is possible to achieve both the smooth movement of lithium ions in the electrode and the suppression of the reaction of the negative electrode active material / electrolytic solution.
- styrene monomer according to the present invention examples include styrene, ⁇ -methylstyrene, dimethylstyrene, vinyltoluene and the like. Among these, styrene is preferable.
- Vinyl monomers such as acrylonitrile, methacrylonitrile, fumaronitrile, A methacrylic acid monomer comprising methacrylic acid, Methacrylate monomers such as methyl methacrylate, ethyl methacrylate, propyl methacrylate, butyl methacrylate, 2-ethylhexyl methacrylate, phenyl methacrylate, benzyl methacrylate, isobornyl methacrylate, Butadiene monomers such as butadiene, isoprene and chloroprene, Acrylic acid monomer consisting of acrylic acid, Acrylic ester monomers such as methyl acrylate, ethyl acrylate, propyl acrylate, butyl acrylate, 2-ethylhexyl acrylate, cyclohexyl acrylate, Unsaturated dicarboxylic acid anhydride type monomers
- the styrene polymer at least a copolymer containing a styrene monomer and a butadiene monomer, or a copolymer containing a styrene monomer and an acrylic acid monomer is preferable. Moreover, it is good also as a copolymer which further contains monomers other than these monomers.
- the styrene polymer binder used in the negative electrode needs to be contained in an amount of 0.3 to 8.0% by mass in the negative electrode binder layer, preferably 0.5 to 6.0% by mass, more preferably 0. It may be contained in the range of 0.8 to 5.0% by mass, more preferably 1.0 to 3.5% by mass. If the content of the styrenic polymer in the negative electrode binder layer is less than 0.3% by mass, sufficient electrode adhesion cannot be obtained. If the amount is more than 8.0% by mass, the effect of increasing the resistance due to the binder that is difficult to soak in the electrolyte becomes larger than the effect of reducing the resistance due to the film formed at the binder / electrolyte interface, and the battery capacity cannot be sufficiently obtained. .
- Metal oxides Metal sulfides such as SnS and FeS 2 ; Metallic lithium, lithium alloy, polyacene, polythiophene, A composite of these and carbon, such as Li 5 (Li 3 N), Li 7 MnN 4 , Li 3 FeN 2 , Li 2.5 Co 0.5 N, lithium nitride such as Li 3 CoN.
- the non-aqueous electrolyte contains at least a cyclic sulfonic acid ester having two sulfonyl groups.
- the cyclic sulfonic acid ester must be contained in the non-aqueous electrolyte in an amount of 0.002 to 5.0% by mass, preferably 0.004 to 4.6% by mass, more preferably 0.1 to 4.2%.
- the content is preferably in the range of 1% by mass, more preferably 1 to 3.6% by mass. When the content of the cyclic sulfonic acid ester is less than 0.002% by mass, a film at the binder / electrolyte interface is not sufficiently formed.
- the film at the binder / electrolyte interface becomes thick, so that solvation / desolvation of lithium ions does not proceed smoothly. As a result, the resistance of the battery is increased and its characteristics are deteriorated.
- a stable surface film is formed on the surface of the binder / electrolytic solution by including a compound represented by the following formula (1).
- Q is an oxygen atom, a methylene group or a single bond
- A is an alkylene group having 1 to 5 carbon atoms, a carbonyl group, a sulfinyl group, a fluoroalkylene group having 1 to 6 carbon atoms, or an ether bond.
- B represents an alkylene group, a fluoroalkylene group, or an oxygen atom.
- Examples of cyclic sulfonic acid esters include compound Nos. Shown in Table 1 below. Examples include, but are not limited to, 1 to 22 organic compounds. Examples of these compounds are shown in Japanese Patent No. 4033074. The compounds shown in Table 1 are described in, for example, US Pat. No. 4,950,768, Japanese Patent Publication No. 5-4496, West German Patent No. 2509738, West German Patent No. 2333859, and the like. It can be obtained using a manufacturing method.
- Nonaqueous solvents used for the nonaqueous electrolyte include ethylene carbonate, propylene carbonate, butylene carbonate, vinylene carbonate, trifluoropropylene carbonate, ⁇ -butyrolactone, 2-methyl- ⁇ -butyrolactone, acetyl- ⁇ -butyrolactone, and ⁇ -valero.
- a mixed system of a cyclic carbonate and a chain carbonate is preferable from the viewpoint of stability to oxidation / reduction.
- an ion conductive polymer electrolyte and an organic electrolyte can be used in combination.
- the ion conductive polymer electrolyte include polyethers such as polyethylene oxide and polypropylene oxide, polyolefins such as polyethylene and polypropylene, polyvinylidene fluoride, polytetrafluoroethylene, polyvinyl fluoride, polyvinyl chloride, polyvinylidene chloride, Polymethyl methacrylate, polymethyl acrylate, polyvinyl alcohol, polymethacrylonitrile, polyvinyl acetate, polyvinyl pyrrolidone, polycarbonate, polyethylene terephthalate, polyhexamethylene aipamide, polycaprolactam, polyurethane, polyethyleneimine, polybutadiene, polystyrene, polyisoprene and these These derivatives can be used alone or in combination.
- the positive electrode active material examples include LiCoO 2 , LiNiO 2 , LiMn 2 O 4 , LiNi 1/3 Co 1/3 Mn 1/3 O 2 , LiNi 0.5 Mn 1.5 O 4 , LiFePO 4 , LiMnPO 4. And lithium-containing composite oxides such as Li 2 MnO 3 .
- the transition metal portion of these lithium-containing composite oxides may be substituted with other elements, or a mixture thereof.
- a polymer containing various monomers constituting the polymer may be used.
- an inorganic solid electrolyte, a mixed material of an organic polymer electrolyte and an inorganic solid electrolyte, an inorganic solid powder bound by an organic binder, or the like can be used.
- a lithium secondary battery is composed of a combination of a positive electrode, a negative electrode, a separator, and a non-aqueous electrolyte.
- a separator a woven fabric, a nonwoven fabric, a polyolefin polymer such as polyethylene or polypropylene, a polyimide, a porous polymer film such as a porous polyvinylidene fluoride film, or an ion conductive polymer electrolyte film can be used alone or in combination.
- the battery can have various shapes such as a cylindrical shape, a square shape, a coin shape, a button shape, and a laminate shape.
- stainless steel, nickel-plated iron, aluminum, titanium, or an alloy thereof and a plated material can be used.
- the material of the laminate resin film aluminum, aluminum alloy, titanium foil, or the like can be used.
- the material of the heat-welded portion of the metal laminate resin film may be any material as long as it is a thermoplastic polymer material such as polyethylene, polypropylene, polyethylene terephthalate.
- the metal laminate resin layer and the metal foil layer are not limited to one layer, and may be two or more layers.
- the positive electrode was produced as follows.
- a positive electrode active material a mixture of Li (Li 0.1 Mn 1.9 ) O 4 and LiNi 0.85 Co 0.15 O 2 in a mass ratio of 85:15 is used, and polyvinylidene fluoride 5 as a binder. The mass% was mixed. This was dispersed in N-methyl-2-pyrrolidone to prepare a slurry. The slurry was applied to both sides of an aluminum current collector with a thickness of 20 ⁇ m uniformly so as to have a thickness of 95 ⁇ m, dried, and then compression molded with a roll press.
- the battery was formed by laminating a positive electrode and a negative electrode via a separator to obtain a laminated exterior type lithium secondary battery.
- the discharge capacity of the produced battery was 98 mAh.
- Examples 21 to 40 Batteries (Examples 21 to 40) were produced in the same manner as in Examples 1 to 20, except that a styrene / acrylic copolymer (styrene polymer) was used as the binder for the negative electrode.
- a styrene / acrylic copolymer styrene polymer
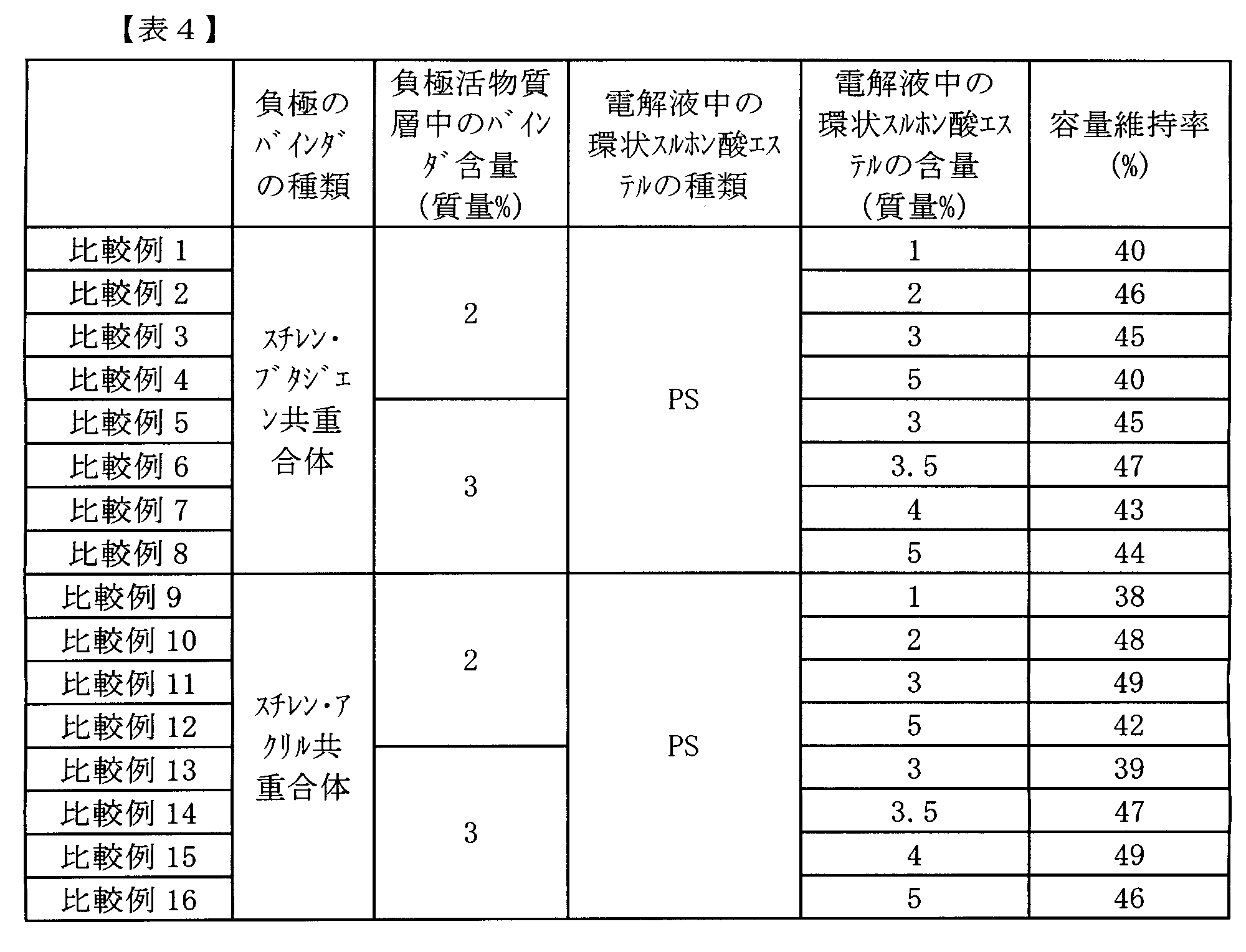
- Batteries (Comparative Examples 1 to 16) were prepared in the same manner as in Examples 1 to 40 except that the electrolyte contained 1,3-propane sultone (hereinafter referred to as “PS”) instead of the cyclic sulfonic acid ester as an additive. 16) was produced.
- PS 1,3-propane sultone
- Batteries (Comparative Examples 17 to 24) were produced in the same manner as in Examples 1 to 20, except that 0.1% by mass or 9.0% by mass of a styrene / butadiene copolymer was contained as a binder for the negative electrode.
- Batteries (Comparative Examples 25 to 32) were prepared in the same manner as in Examples 21 to 40 except that 0.1% by mass or 9.0% by mass of styrene / acrylic copolymer was included as a binder for the negative electrode.
- the negative electrode active material layer contains 0.3 to 8.0% by mass of a styrene polymer as a negative electrode binder, and at least sulfonyl in the electrolytic solution.
- the non-aqueous electrolyte secondary batteries (Examples 1 to 40) containing 0.002 to 5.0% by mass of the cyclic sulfonate ester having two groups were confirmed to have a high capacity retention rate. It was.
- the binder containing the styrenic polymer in the negative electrode is 0.1% by mass (Comparative Examples 17 to 20, Comparative Examples 25 to 28), the amount of the binder in the negative electrode is too small, resulting in poor electrode adhesion. It is considered that the battery capacity has become insufficient. Further, when the binder containing the styrene polymer is 9.0% by mass (Comparative Examples 21 to 24, Comparative Examples 29 to 32), the electrolysis is more effective than the effect of reducing the resistance by the film formed at the binder / electrolyte interface. It is thought that the effect of increasing the resistance due to the use of a binder that hardly penetrates the liquid is increased. As a result, it is considered that the battery capacity is not sufficiently obtained.
- Examples 57 to 72 Batteries (Examples 57 to 72) were prepared in the same manner as in Examples 1 to 20, except that an acrylic / styrene / acrylonitrile copolymer or a carboxylic acid ester-introduced styrene / acrylic copolymer was used as the binder for the negative electrode. Then, a charge / discharge cycle test was conducted in the same manner as in Examples 1 to 20. The results are shown in Table 7.
- Examples 73 to 84 As an additive for an electrolytic solution, compound No. 1 in Table 1 was used. 4, Compound No. 5 or Compound No. Batteries (Examples 73 to 84) were produced in the same manner as in Examples 1 to 40 except that 1% by mass or 3% by mass of the cyclic sulfonate ester compound as shown in FIG.
- Batteries (Comparative Examples 61-65, 71-75) were produced in the same manner as Examples 1 to 40 except that ethylene glycol dimethanesulfonate represented by the following formula (2) was included as an additive for the electrolytic solution.
- Batteries (Comparative Examples 66-70, 76-80) were prepared in the same manner as in Examples 1 to 40 except that the electrolyte contained 1% by mass of benzene and 1,4-butanediol dimethanesulfonate represented by the following formula (3) as an additive. 80).
- Comparative Examples 61-80 were subjected to charge / discharge cycle tests in the same manner as in Examples 1-40. The results are shown in Table 9.
Landscapes
- Chemical & Material Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Engineering & Computer Science (AREA)
- Inorganic Chemistry (AREA)
- Manufacturing & Machinery (AREA)
- General Physics & Mathematics (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- Physics & Mathematics (AREA)
- Materials Engineering (AREA)
- Secondary Cells (AREA)
- Battery Electrode And Active Subsutance (AREA)
Abstract
Description
リチウムイオンを吸蔵・放出することが可能な正極と、
バインダとして少なくともスチレン系重合体を含む負極活物質層を有し、リチウムイオンを吸蔵・放出することが可能な負極であって、前記スチレン系重合体の含量は前記負極活物質層全体の質量に対して0.3~8.0質量%である負極と、
少なくとも、スルホニル基を2個有する環状スルホン酸エステルを、非水電解液全体の質量に対して0.002~5.0質量%、含む非水電解液と、
前記正極と負極間に設けられたセパレータと、
を有する非水電解液二次電池に関する。
負極中に含まれるバインダとしての「スチレン系重合体」とは、スチレン単量体の重合体、又は、スチレン単量体及びスチレン単量体と共重合が可能な単量体の共重合体のことを表す。
アクリロニトリル、メタクリロニトリル、フマロニトリルなどのビニル系の単量体、
メタクリル酸からなるメタクリル酸単量体、
メタクリル酸メチル、メタクリル酸エチル、メタクリル酸プロピル、メタクリル酸ブチル、メタクリル酸-2-エチルヘキシル、メタクリル酸フェニル、メタクリル酸ベンジル、メタクリル酸イソボルニルなどのメタクリル酸エステル系の単量体、
ブタジエン、イソプレン、クロロプレンなどのブタジエン系の単量体、
アクリル酸からなるアクリル酸単量体、
アクリル酸メチル、アクリル酸エチル、アクリル酸プロピル、アクリル酸ブチル、アクリル酸-2-エチルヘキシル、アクリル酸シクロヘキシルなどのアクリル酸エステル系の単量体、
マレイン酸、イタコン酸、シトラコン酸の無水物などの不飽和ジカルボン酸無水物系の単量体、
マレイミド、N-メチルマレイミド、N-ブチルマレイミド、N-フェニルマレイミド、N-シクロヘキシルマレイミドなどの不飽和ジカルボン酸のイミド化合物系の単量体
などが挙げられる。これらの単量体は、単独または2種以上を組み合わせても使用できる。
コークス類、ガラス状炭素類、グラファイト類、難黒鉛化性炭素類、熱分解炭素類、炭素繊維、
Al、Si、Pb、Sn、Zn、Cd、Sb等を主体とした活物質、またはこれらとリチウムとの合金、
LiFe2O3、WO2、MoO2、SiO、SiO2、CuO、SnO、SnO2、Nb3O5、LixTi2-xO4(0≦x≦1)、PbO2、PbO5等の金属酸化物、
SnSやFeS2等の金属硫化物、
金属リチウム、リチウム合金、ポリアセン、ポリチオフェン、
Li5(Li3N)、Li7MnN4、Li3FeN2、Li2.5Co0.5N、Li3CoN等の窒化リチウム等、これらと炭素の複合体。
非水電解液中には少なくとも、スルホニル基を2個有する環状スルホン酸エステルを含む。環状スルホン酸エステルは、非水電解液中に0.002~5.0質量%、含む必要があるが、好ましくは0.004~4.6質量%、より好ましくは0.1~4.2質量%、更に好ましくは1~3.6質量%の範囲で含まれるのが良い。環状スルホン酸エステルの含量が0.002質量%よりも少ないと、バインダ・電解液界面の皮膜が十分に形成されない。また、環状スルホン酸エステルの含量が5質量%よりも多くなると、バインダ・電解液界面の皮膜が厚くなることで、リチウムイオンの溶媒和・脱溶媒和がスムーズに進行しなくなる。その結果、電池の抵抗上昇につながり、その特性が低下する。
(式(1)において、Qは酸素原子、メチレン基または単結合、Aは炭素数1~5のアルキレン基、カルボニル基、スルフィニル基、炭素数1~6のフルオロアルキレン基、またはエーテル結合を介してアルキレン単位もしくはフルオロアルキレン単位結合した炭素数2~6の2価の基を示し、Bはアルキレン基、フルオロアルキレン基、または酸素原子を示す。)。
正極活物質としては、たとえば、LiCoO2、LiNiO2、LiMn2O4、LiNi1/3Co1/3Mn1/3O2、LiNi0.5Mn1.5O4、LiFePO4、LiMnPO4、Li2MnO3などのリチウム含有複合酸化物が挙げられる。これらのリチウム含有複合酸化物の遷移金属部分を他の元素で置換させたものでもよく、またこれらの混合物でもよい。
リチウム二次電池は、正極、負極、セパレータ、非水電解液の組み合わせからなっている。セパレータとしては、織布、不織布、ポリエチレンやポリプロピレンなどのポリオレフィン系、ポリイミド、多孔性ポリフッ化ビニリデン膜などの多孔性ポリマー膜やイオン伝導性ポリマー電解質膜を単独または組み合わせで使用することができる。
図1は、本実施例で作製したリチウム二次電池の構成を示す模式図である。図1に示すように、アルミニウム箔等の金属からなる正極集電体11上にはリチウムイオンを吸蔵、放出し得る正極活物質層12が設けられている。銅箔等の金属からなる負極集電体13上には、リチウムイオンを吸蔵、放出する負極活物質層14が設けられている。そして、電解液15、およびこれを含む不織布、ポリオレフィン微多孔膜などからなるセパレータ16を介して、正極集電体11及び正極活物質層12と、負極集電体13及び負極活物質層14とが対向して配置され構成されている。
負極電極のバインダとして、スチレン・アクリル共重合体(スチレン系重合体)を使用する以外は実施例1~20と同様に電池(実施例21~40)を作製した。
電解液中に、添加剤として環状スルホン酸エステルの代わりに1,3-プロパンスルトン(以下、「PS」と記載する)を含む以外は、実施例1~40と同様に電池(比較例1~16)を作製した。
負極用のバインダとして、スチレン・ブタジエン共重合体を0.1質量%、又は9.0質量%含む以外は、実施例1~20と同様に電池(比較例17~24)を作製した。
負極用のバインダとして、スチレン・アクリル共重合体を0.1質量%、又は9.0質量%含む以外は、実施例21~40と同様に電池(比較例25~32)を作製した。
電解液添加剤として、電解液中に環状スルホン酸エステルを0.001質量%、又は6.0質量%含む以外は実施例1~20と同様に電池(比較例33~40)を作製した。
電解液添加剤として、電解液中に環状スルホン酸エステルを0.001質量%、又は6.0質量%含む以外は実施例21~40と同様に電池(比較例41~48)を作製した。
次に、上記のようにして作製した電池に対して、下記の条件による定電流定電圧方式で、充放電サイクル試験をおこなった。
(1)充電条件
温度:60℃
充電終止電圧:4.2V
充電電流:98mA
全充電時間:2.5時間
(2)放電条件
温度:60℃
放電終止電圧:3.0V
放電電流98mA。
容量維持率(%)は、500サイクル後の放電容量(mAh)の、10サイクル後の放電容量(mAh)に対する割合であり、下記式で表される。
容量維持率(%)=(500サイクル後の放電容量(mAh))/(10サイクル後の放電容量(mAh))
サイクル試験の結果を表2~5に示す。
負極中のバインダとして、メチルメタクリレート・スチレン・ブタジエン共重合体、又はアクリロニトリル・スチレン・ブタジエン共重合体を使用した以外は、実施例1~20と同様に電池(実施例41~56)を作製し、実施例1~20と同様に充放電サイクル試験を行った。結果を表6に示す。
負極用のバインダとして、アクリル・スチレン・アクリロニトリル共重合体、又は、カルボン酸エステル導入スチレン・アクリル共重合体を使用した以外は、実施例1~20と同様に電池(実施例57~72)を作製し、実施例1~20と同様に充放電サイクル試験を行った。結果を表7に示す。
電解液用の添加剤として、表1の化合物No.4、化合物No.5又は化合物No.6に示すような環状スルホン酸エステル化合物を、1質量%、又は3質量%含む以外は実施例1~40と同様に電池(実施例73~84)を作製した。
電解液添加剤としてビニレンカーボネート(VC)を含む以外は実施例1~40と同様に電池(比較例49~60)を作製した。
電解液の添加剤として、下記式(2)に示すエチレングリコールジメタンスルホネートを含む以外は、実施例1~40と同様に電池(比較例61~65、71~75)を作製した。
電解液にベンゼン1質量%と、添加剤として下記式(3)に示す1,4-ブタンジオールジメタンスルホネートを含む以外は実施例1~40と同様に電池(比較例66~70、76~80)を作製した。
12 正極活物質層
13 負極集電体
14 負極活物質層
15 電解液
16 セパレータ
Claims (6)
- リチウムイオンを吸蔵・放出することが可能な正極と、
バインダとして少なくともスチレン系重合体を含む負極活物質層を有し、リチウムイオンを吸蔵・放出することが可能な負極であって、前記スチレン系重合体の含量は前記負極活物質層全体の質量に対して0.3~8.0質量%である負極と、
少なくとも、スルホニル基を2個有する環状スルホン酸エステルを、非水電解液全体の質量に対して0.002~5.0質量%、含む非水電解液と、
前記正極と負極間に設けられたセパレータと、
を有する非水電解液二次電池。 - 前記負極のバインダは、スチレン・ブタジエン共重合体を含む、請求項1又は2に記載の非水電解液二次電池。
- 前記負極のバインダは、スチレン・アクリル共重合体を含む、請求項1又は2に記載の非水電解液二次電池。
- 前記負極は、活物質として炭素質材料を含む、請求項1~4の何れか1項に記載の非水電解液二次電池。
- 前記炭素質材料が黒鉛材料である、請求項5に記載の非水電解液二次電池。
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/577,576 US20130029218A1 (en) | 2010-02-08 | 2011-02-08 | Nonaqueous electrolyte secondary battery |
| EP11739919.6A EP2535974B1 (en) | 2010-02-08 | 2011-02-08 | Nonaqueous electrolyte secondary battery |
| JP2011552861A JPWO2011096572A1 (ja) | 2010-02-08 | 2011-02-08 | 非水電解液二次電池 |
| CN201180008714.1A CN102742063B (zh) | 2010-02-08 | 2011-02-08 | 非水电解液二次电池 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2010-024945 | 2010-02-08 | ||
| JP2010024945 | 2010-02-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011096572A1 true WO2011096572A1 (ja) | 2011-08-11 |
Family
ID=44355564
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2011/052615 WO2011096572A1 (ja) | 2010-02-08 | 2011-02-08 | 非水電解液二次電池 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20130029218A1 (ja) |
| EP (1) | EP2535974B1 (ja) |
| JP (1) | JPWO2011096572A1 (ja) |
| CN (1) | CN102742063B (ja) |
| WO (1) | WO2011096572A1 (ja) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013146819A1 (ja) * | 2012-03-29 | 2013-10-03 | 日本電気株式会社 | 二次電池 |
| WO2014133165A1 (ja) * | 2013-03-01 | 2014-09-04 | 日本電気株式会社 | リチウムイオン二次電池 |
| WO2014133161A1 (ja) * | 2013-03-01 | 2014-09-04 | 日本電気株式会社 | 非水電解液二次電池 |
| WO2014133163A1 (ja) * | 2013-03-01 | 2014-09-04 | 日本電気株式会社 | 非水電解液二次電池 |
| JP2015076389A (ja) * | 2013-10-11 | 2015-04-20 | オートモーティブエナジーサプライ株式会社 | 非水電解質二次電池 |
| JPWO2014133169A1 (ja) * | 2013-03-01 | 2017-02-09 | 日本電気株式会社 | 二次電池用電解液およびそれを用いた二次電池 |
| JPWO2015194559A1 (ja) * | 2014-06-16 | 2017-04-20 | 日本電気株式会社 | 電解液及び二次電池 |
| US9899703B2 (en) | 2013-03-01 | 2018-02-20 | Nec Corporation | Gel electrolyte and polymer secondary battery using same |
| JP2020004542A (ja) * | 2018-06-26 | 2020-01-09 | 日立化成株式会社 | 負極及び電気化学デバイス |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5861634B2 (ja) * | 2010-08-05 | 2016-02-16 | 和光純薬工業株式会社 | 非水系電解液、その製造法、及び該電解液を用いた非水系電解液電池 |
| KR102008671B1 (ko) | 2010-08-05 | 2019-08-08 | 후지필름 와코 준야꾸 가부시키가이샤 | 비수계 전해액 및 그를 사용한 비수계 전해액 전지 |
| US9923196B2 (en) * | 2013-10-03 | 2018-03-20 | Johns Manville | Conductive mat for battery electrode plate reinforcement and methods of use therefor |
| JP6662292B2 (ja) * | 2014-08-07 | 2020-03-11 | 日本電気株式会社 | リチウム二次電池およびその製造方法 |
| US11011774B2 (en) * | 2014-12-16 | 2021-05-18 | Nec Corporation | Lithium-ion secondary battery |
| TWI618287B (zh) * | 2015-07-23 | 2018-03-11 | Copolymer of lithium battery and its positive electrode coating | |
| CN107346818B (zh) * | 2016-05-06 | 2020-04-07 | 宁德新能源科技有限公司 | 阴极极片及其制备方法以及锂离子电池 |
| JP7045545B2 (ja) * | 2018-01-04 | 2022-04-01 | エルジー エナジー ソリューション リミテッド | 絶縁コーティング層が備えられた電極タブを含む二次電池 |
| CN109888385B (zh) * | 2019-01-25 | 2020-11-06 | 厦门大学 | 一种锂金属二次电池用电解液及锂金属二次电池 |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2233859A1 (de) | 1972-07-10 | 1974-01-31 | Asf Agm 6700 Ludwigshafen | Verfahren zur reinigung von rohem carbylsulfat |
| DE2509738A1 (de) | 1975-03-06 | 1976-09-23 | Basf Ag | Verfahren zur herstellung von carbylsulfat |
| US4950768A (en) | 1984-01-16 | 1990-08-21 | Cronyn Marshall W | Cyclic disulfonic ester cross-linking compounds |
| JPH0359832B2 (ja) | 1982-04-02 | 1991-09-11 | Canon Kk | |
| JPH0433074B2 (ja) | 1985-02-27 | 1992-06-02 | Oki Electric Ind Co Ltd | |
| JPH04255670A (ja) * | 1991-01-30 | 1992-09-10 | Matsushita Electric Ind Co Ltd | 非水電解質二次電池用負極 |
| JPH054496B2 (ja) | 1987-07-28 | 1993-01-20 | Mitsui Toatsu Kagaku Kk | |
| JPH11111300A (ja) * | 1997-09-30 | 1999-04-23 | Sanyo Electric Co Ltd | 非水系二次電池用負極 |
| JP3815087B2 (ja) | 1998-10-26 | 2006-08-30 | 宇部興産株式会社 | 非水電解液及びそれを用いたリチウム二次電池 |
| JP3978881B2 (ja) | 1997-08-22 | 2007-09-19 | 宇部興産株式会社 | 非水電解液およびそれを用いたリチウム二次電池 |
| JP2008071559A (ja) * | 2006-09-13 | 2008-03-27 | Nec Tokin Corp | リチウムイオン二次電池 |
| JP2009038018A (ja) * | 2007-07-09 | 2009-02-19 | Sony Corp | 二次電池 |
| JP4229615B2 (ja) | 2002-01-24 | 2009-02-25 | 三菱化学株式会社 | 非水系電解液及びそれを用いたリチウム二次電池 |
| JP2010024945A (ja) | 2008-07-18 | 2010-02-04 | Panasonic Electric Works Co Ltd | ベーンポンプ |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3262704B2 (ja) * | 1995-04-24 | 2002-03-04 | シャープ株式会社 | 非水系二次電池用炭素電極、その製造方法及びそれを用いた非水系二次電池 |
| JPH0973897A (ja) * | 1995-09-06 | 1997-03-18 | Hitachi Chem Co Ltd | 密閉型二次電池 |
| JP4461498B2 (ja) * | 1997-12-16 | 2010-05-12 | パナソニック株式会社 | 非水電解液二次電池およびその負極 |
| JP2000294247A (ja) * | 1999-04-12 | 2000-10-20 | Hitachi Powdered Metals Co Ltd | リチウムイオン二次電池の負極塗膜およびそれを用いたリチウムイオン二次電池 |
| ATE496399T1 (de) * | 2000-03-07 | 2011-02-15 | Teijin Ltd | Lithiumionen-sekundärzelle, separator, zellenpack und ladeverfahren |
| JP4033074B2 (ja) * | 2002-08-29 | 2008-01-16 | 日本電気株式会社 | 二次電池用電解液およびそれを用いた二次電池 |
| JP4291195B2 (ja) * | 2004-03-30 | 2009-07-08 | 宇部興産株式会社 | 非水電解質二次電池 |
| JP2007220496A (ja) * | 2006-02-17 | 2007-08-30 | Hitachi Vehicle Energy Ltd | カルボン酸無水有機化合物を電解液に含むリチウム二次電池 |
| KR100969130B1 (ko) * | 2006-08-25 | 2010-07-07 | 주식회사 엘지화학 | 내수성이 강화된 비수성 전해액 및 이를 포함하는 리튬이차 전지 |
| JP4329806B2 (ja) * | 2006-10-13 | 2009-09-09 | ソニー株式会社 | 二次電池 |
| JP5236875B2 (ja) * | 2006-12-19 | 2013-07-17 | Necエナジーデバイス株式会社 | 非水電解液およびそれを用いた非水電解液二次電池 |
| JP5169400B2 (ja) * | 2008-04-07 | 2013-03-27 | Necエナジーデバイス株式会社 | 非水電解液およびそれを用いた非水電解液二次電池 |
-
2011
- 2011-02-08 US US13/577,576 patent/US20130029218A1/en not_active Abandoned
- 2011-02-08 EP EP11739919.6A patent/EP2535974B1/en active Active
- 2011-02-08 WO PCT/JP2011/052615 patent/WO2011096572A1/ja active Application Filing
- 2011-02-08 JP JP2011552861A patent/JPWO2011096572A1/ja active Pending
- 2011-02-08 CN CN201180008714.1A patent/CN102742063B/zh active Active
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2233859A1 (de) | 1972-07-10 | 1974-01-31 | Asf Agm 6700 Ludwigshafen | Verfahren zur reinigung von rohem carbylsulfat |
| DE2509738A1 (de) | 1975-03-06 | 1976-09-23 | Basf Ag | Verfahren zur herstellung von carbylsulfat |
| JPH0359832B2 (ja) | 1982-04-02 | 1991-09-11 | Canon Kk | |
| US4950768A (en) | 1984-01-16 | 1990-08-21 | Cronyn Marshall W | Cyclic disulfonic ester cross-linking compounds |
| JPH0433074B2 (ja) | 1985-02-27 | 1992-06-02 | Oki Electric Ind Co Ltd | |
| JPH054496B2 (ja) | 1987-07-28 | 1993-01-20 | Mitsui Toatsu Kagaku Kk | |
| JPH04255670A (ja) * | 1991-01-30 | 1992-09-10 | Matsushita Electric Ind Co Ltd | 非水電解質二次電池用負極 |
| JP2548460B2 (ja) | 1991-01-30 | 1996-10-30 | 松下電器産業株式会社 | 非水電解質二次電池用負極 |
| JP3978881B2 (ja) | 1997-08-22 | 2007-09-19 | 宇部興産株式会社 | 非水電解液およびそれを用いたリチウム二次電池 |
| JPH11111300A (ja) * | 1997-09-30 | 1999-04-23 | Sanyo Electric Co Ltd | 非水系二次電池用負極 |
| JP3815087B2 (ja) | 1998-10-26 | 2006-08-30 | 宇部興産株式会社 | 非水電解液及びそれを用いたリチウム二次電池 |
| JP4229615B2 (ja) | 2002-01-24 | 2009-02-25 | 三菱化学株式会社 | 非水系電解液及びそれを用いたリチウム二次電池 |
| JP2008071559A (ja) * | 2006-09-13 | 2008-03-27 | Nec Tokin Corp | リチウムイオン二次電池 |
| JP2009038018A (ja) * | 2007-07-09 | 2009-02-19 | Sony Corp | 二次電池 |
| JP2010024945A (ja) | 2008-07-18 | 2010-02-04 | Panasonic Electric Works Co Ltd | ベーンポンプ |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2535974A4 * |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPWO2013146819A1 (ja) * | 2012-03-29 | 2015-12-14 | 日本電気株式会社 | 二次電池 |
| WO2013146819A1 (ja) * | 2012-03-29 | 2013-10-03 | 日本電気株式会社 | 二次電池 |
| US9899703B2 (en) | 2013-03-01 | 2018-02-20 | Nec Corporation | Gel electrolyte and polymer secondary battery using same |
| WO2014133165A1 (ja) * | 2013-03-01 | 2014-09-04 | 日本電気株式会社 | リチウムイオン二次電池 |
| US9941545B2 (en) | 2013-03-01 | 2018-04-10 | Nec Corporation | Electrolyte solution for secondary batteries, and secondary battery using same |
| WO2014133161A1 (ja) * | 2013-03-01 | 2014-09-04 | 日本電気株式会社 | 非水電解液二次電池 |
| JPWO2014133163A1 (ja) * | 2013-03-01 | 2017-02-09 | 日本電気株式会社 | 非水電解液二次電池 |
| JPWO2014133169A1 (ja) * | 2013-03-01 | 2017-02-09 | 日本電気株式会社 | 二次電池用電解液およびそれを用いた二次電池 |
| JPWO2014133165A1 (ja) * | 2013-03-01 | 2017-02-09 | 日本電気株式会社 | リチウムイオン二次電池 |
| WO2014133163A1 (ja) * | 2013-03-01 | 2014-09-04 | 日本電気株式会社 | 非水電解液二次電池 |
| US9711825B2 (en) | 2013-03-01 | 2017-07-18 | Nec Corporation | Lithium ion secondary battery |
| US9780411B2 (en) | 2013-03-01 | 2017-10-03 | Nec Corporation | Nonaqueous electrolyte solution secondary battery |
| JP2015076389A (ja) * | 2013-10-11 | 2015-04-20 | オートモーティブエナジーサプライ株式会社 | 非水電解質二次電池 |
| JPWO2015194559A1 (ja) * | 2014-06-16 | 2017-04-20 | 日本電気株式会社 | 電解液及び二次電池 |
| US10566661B2 (en) | 2014-06-16 | 2020-02-18 | Nec Corporation | Electrolytic solution and secondary battery |
| JP2020004542A (ja) * | 2018-06-26 | 2020-01-09 | 日立化成株式会社 | 負極及び電気化学デバイス |
| JP7127385B2 (ja) | 2018-06-26 | 2022-08-30 | 昭和電工マテリアルズ株式会社 | 負極及び電気化学デバイス |
Also Published As
| Publication number | Publication date |
|---|---|
| JPWO2011096572A1 (ja) | 2013-06-13 |
| US20130029218A1 (en) | 2013-01-31 |
| CN102742063B (zh) | 2015-04-29 |
| CN102742063A (zh) | 2012-10-17 |
| EP2535974A4 (en) | 2015-04-01 |
| EP2535974B1 (en) | 2016-10-26 |
| EP2535974A1 (en) | 2012-12-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2011096572A1 (ja) | 非水電解液二次電池 | |
| CN109980177B (zh) | 电极极片和包含所述电极极片的电化学装置 | |
| JP4543085B2 (ja) | リチウム2次電池用添加剤 | |
| US20220209298A1 (en) | Electrolyte For Lithium Secondary Battery And Lithium Secondary Battery Including The Same | |
| JP2014199714A (ja) | 非水電解質二次電池用負極およびその非水電解質二次電池 | |
| WO2008140271A1 (en) | Additives for non-aqueous electrolyte and secondary battery using the same | |
| KR102563223B1 (ko) | 전해액 및 전기화학 장치 | |
| KR20160057813A (ko) | 음극 활물질 및 이를 포함하는 리튬 전지 | |
| JP6704457B2 (ja) | リチウム二次電池 | |
| WO2015037367A1 (ja) | 非水電解液二次電池 | |
| CN112400249A (zh) | 一种电解液及电化学装置 | |
| KR101568563B1 (ko) | 리튬 이차 전지 | |
| JP2022519502A (ja) | リチウム二次電池 | |
| TW201633596A (zh) | 非水性電解質鋰二次電池 | |
| KR20210097303A (ko) | 음극 활물질, 이를 포함하는 음극 및 이차전지 | |
| KR20220058026A (ko) | 리튬 이차전지용 전해액 및 이를 포함하는 리튬 이차전지 | |
| US11817584B2 (en) | Binder for the lithium secondary battery, electrode comprising same and lithium secondary battery | |
| KR20130134910A (ko) | 전극 조립체 및 이를 포함하는 리튬 이차전지 | |
| WO2012014255A1 (ja) | リチウムイオン二次電池 | |
| JP2006073253A (ja) | 非水電解質電池 | |
| CN112368872A (zh) | 一种电解液及电化学装置 | |
| CN118231814B (zh) | 二次电池和电子装置 | |
| CN116053461B (zh) | 电化学装置和包括其的电子装置 | |
| KR102555746B1 (ko) | 리튬 이차전지용 비수 전해액 및 이를 포함하는 리튬 이차전지 | |
| WO2023225897A1 (zh) | 电化学装置及包含其的电子装置 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201180008714.1 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11739919 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011552861 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13577576 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2011739919 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011739919 Country of ref document: EP |