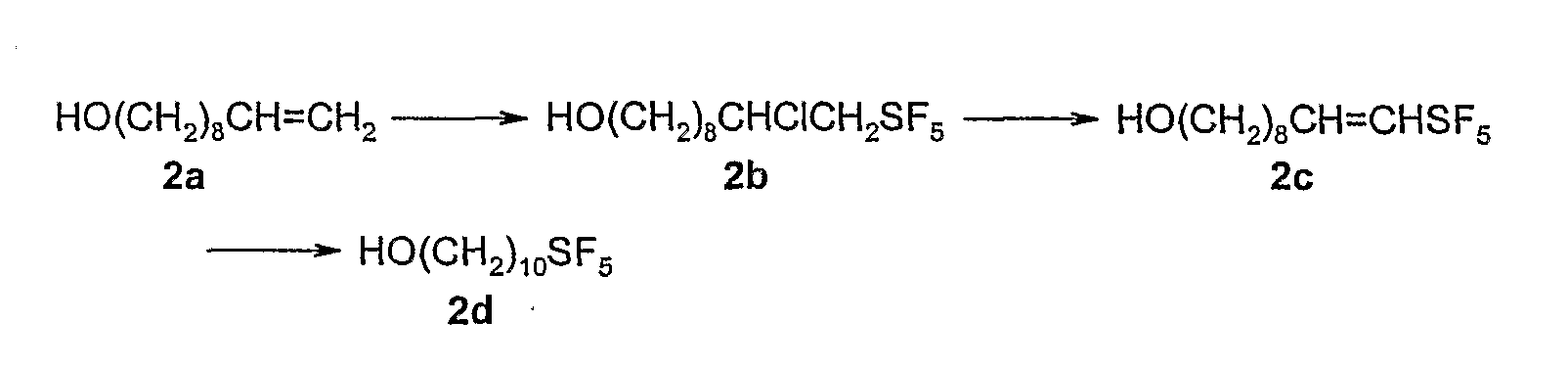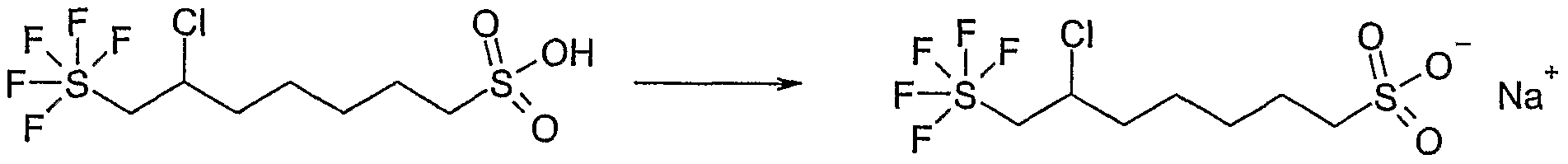WO2006072401A1 - Fluortenside - Google Patents
Fluortenside Download PDFInfo
- Publication number
- WO2006072401A1 WO2006072401A1 PCT/EP2005/013859 EP2005013859W WO2006072401A1 WO 2006072401 A1 WO2006072401 A1 WO 2006072401A1 EP 2005013859 W EP2005013859 W EP 2005013859W WO 2006072401 A1 WO2006072401 A1 WO 2006072401A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- och
- formula
- use according
- integer
- Prior art date
Links
- 0 C1C2C1CC(C1)C1*2 Chemical compound C1C2C1CC(C1)C1*2 0.000 description 1
- OZFYUGSGXHKDEN-UHFFFAOYSA-N FC(OCCCCCCCCCCBr)(F)F Chemical compound FC(OCCCCCCCCCCBr)(F)F OZFYUGSGXHKDEN-UHFFFAOYSA-N 0.000 description 1
- JCZNZADMYMGXQC-UHFFFAOYSA-N OS(CCCCCCCCCCOC(F)(F)F)(=O)=O Chemical compound OS(CCCCCCCCCCOC(F)(F)F)(=O)=O JCZNZADMYMGXQC-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/03—Ethers having all ether-oxygen atoms bound to acyclic carbon atoms
- C07C43/04—Saturated ethers
- C07C43/13—Saturated ethers containing hydroxy or O-metal groups
- C07C43/137—Saturated ethers containing hydroxy or O-metal groups containing halogen
-
- A—HUMAN NECESSITIES
- A62—LIFE-SAVING; FIRE-FIGHTING
- A62D—CHEMICAL MEANS FOR EXTINGUISHING FIRES OR FOR COMBATING OR PROTECTING AGAINST HARMFUL CHEMICAL AGENTS; CHEMICAL MATERIALS FOR USE IN BREATHING APPARATUS
- A62D1/00—Fire-extinguishing compositions; Use of chemical substances in extinguishing fires
- A62D1/0071—Foams
- A62D1/0085—Foams containing perfluoroalkyl-terminated surfactant
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/01—Sulfonic acids
- C07C309/02—Sulfonic acids having sulfo groups bound to acyclic carbon atoms
- C07C309/03—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C309/07—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton containing oxygen atoms bound to the carbon skeleton
- C07C309/09—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton containing oxygen atoms bound to the carbon skeleton containing etherified hydroxy groups bound to the carbon skeleton
- C07C309/10—Sulfonic acids having sulfo groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton containing oxygen atoms bound to the carbon skeleton containing etherified hydroxy groups bound to the carbon skeleton with the oxygen atom of at least one of the etherified hydroxy groups further bound to an acyclic carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C381/00—Compounds containing carbon and sulfur and having functional groups not covered by groups C07C301/00 - C07C337/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/03—Ethers having all ether-oxygen atoms bound to acyclic carbon atoms
- C07C43/04—Saturated ethers
- C07C43/12—Saturated ethers containing halogen
- C07C43/123—Saturated ethers containing halogen both carbon chains are substituted by halogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/62—Halogen-containing esters
- C07C69/65—Halogen-containing esters of unsaturated acids
- C07C69/653—Acrylic acid esters; Methacrylic acid esters; Haloacrylic acid esters; Halomethacrylic acid esters
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
- C07D213/16—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom containing only one pyridine ring
- C07D213/20—Quaternary compounds thereof
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K23/00—Use of substances as emulsifying, wetting, dispersing, or foam-producing agents
- C09K23/007—Organic compounds containing halogen
Definitions
- the present invention is the use of end groups Y, wherein Y is CF 3 O or F 5 S- as a hydrophobic end group in surface-active compounds, corresponding new
- Fluid surfactants have a superior surface energy lowering ability, which is used, for example, in the hydrophobization of surfaces such as textile impregnation, hydrophobization of glass, or aircraft de-icing.
- fluoride surfactants contain perfluoroalkyl substituents which are degraded in the environment by biological and other oxidation processes to perfluoroalkanecarboxylic acids and sulfonic acids. These are considered persistent and are z. T. suspected of causing damage to health (G.L. Kennedy, Jr., J.L. Butenhoff, G.W. Olsen, J.C.
- a first object of the present invention is therefore the
- end groups Y wherein Y stands for CF 3 O or F 5 S- as a hydrophobic end group in surface-active compounds.
- the end group Y in the surface-active compounds is bound to a saturated or unsaturated, branched or unbranched hydrocarbon unit.
- the hydrocarbon units may be aliphatic or aromatic units optionally provided with heteroatoms.
- the compounds to be used according to the invention preferably contain no further fluorinated groups in addition to the stated fluorinated end groups.
- the end group Y occurs several times in the surface-active compound and the surface-active compound is preferably an oligomer or polymer.
- the end group Y in the surface-active compound occurs only once, twice or three times, compounds in which the end group occurs only once being particularly preferred.
- the compounds to be used according to the invention are preferably low molecular weight compounds of the formula I.
- Y is CF 3 O- or F 5 S-
- - Spacer represents a saturated or unsaturated, branched or unbranched hydrocarbon moiety
- - X represents a cationic, nonionic, ampothere or anionic polar group or a polymerizable group.
- the compound of the formula I is selected from the compounds of the formula Ia, Ib or Ic,
- Y is CF 3 O- or F 5 S-
- n is an integer from the range 1 to 30
- X is a cationic, nonionic, ampothere or anionic polar group or a polymerizable group
- (Hal) is F, Cl, Br or I, and corresponding salts of the compounds of the formula Ia, Ib or Ic.
- n is particularly preferably an integer from the range from 4 to 24, and particularly preferably an integer from the range from 6 to 18. In this case, it is in a variant of the invention again preferred when n is an even number.
- Particularly preferred according to the invention is the use of the abovementioned compounds as surfactant.
- the counterion is an alkali metal ion, preferably Li + , Na + or K + , an alkaline earth metal ion or NH 4 + . If the compounds of the formula I are cationic compounds or cationically salinatable compounds, it is preferred if the counterion used is a
- Halide such as Cl “ , Br, I " , or CH 3 SO 3 “ , CF 3 SO 3 “ , CH 3 PhSO 3 ' or PhSO 3 " is present.
- Pentafluorosulfurane methanesulfonates SF 5 CX 2 SO 3 - with X H and F, respectively, in BH Ward, JA Schlueter, U. Geiser, HH Wang,
- Vort ⁇ ile of the compounds according to the invention or use according to the invention of the compounds mentioned or of the compositions according to the invention can be in particular: a surface activity which corresponds to the conventional hydrocarbon
- Another object of the present invention are the corresponding novel compounds of formula I, in particular compounds of formula IIa, IIb or Hc
- n is an integer from the range 1 to 30 and
- X is a cationic, nonionic, ampothere or anionic polar group or a polymerizable group
- (Hal) is F, Cl, Br or I, and corresponding salts of the compounds of formula IIa, IIb or Hc, wherein CF 3 -O -CH 2 -COOH is excluded, or compound according to formula IIIa, IIIb or IHc
- n is an integer from the range 1 to 30 and X is a cationic, nonionic, ampothere or anionic polar group or a polymerisable group, (Hal) is F, Cl, Br or I, and corresponding salts of the compounds Formula IIIa, HIb or IHc, where the compounds F 5 S- (CH 2 ) 1 -CO 2 M, F 5 S- (CH 2 ) 2 - CO 2 M, F 5 S- (CH 2 ) 1 -SO 3 M, F 5 S- (CH 2) 2 -SO 3 M, F 5 S- (CH 2) 1 -CONH 2, F 5 S- (CH 2) 2 -OH, and R 5 S- (CH 2) 3 -OH, F 5 S-CH 2 -CHCl- (CH 2 ) 8 -X 1 where M is H or an alkali metal ion, preferably Li + , Na + or K + , or NH 4 + and X 1 for OH, OC (OO) CH 3 , Br, C
- n is preferably a number from the range 4 to 28, particularly preferably a number from the range 8 to 24.
- X is an anionic polar group selected from -COOM, -SO 3 M, -OSO 3 M, -PO 3 M 2 , - OPO 3 M 2 , - (OCH 2 CH 2 ) m -O- (CH 2 ) o -COOM, - (OCH 2 CH 2 ) m -O- (CH 2 ) o -SO 3 M, - (OCH 2 CH 2 ) m -O- (CH 2 ) o -OSO 3 M, - (OCH 2 CH 2 ) m -O- (CH 2 ) o -PO 3 M 2 , - (OCH 2 CH 2 ) m -O- (CH 2 ) o -PO 3 M 2 , - (OCH 2 CH 2 ) m -O- (CH 2 ) o -OPO 3 M 2 , where M is H or an alkali metal ion, preferably Li + , Na + or K + ,
- the preferred anionic groups include in particular - COOM, -SO 3 M, -OSO 3 M, and - (OCH 2 CH 2 ) m -O- (CH 2 ) o -COOM, - (OCH 2 CH 2 ) m - O- (CH 2) 0 -SO 3 m, and - (OCH 2 CH 2) m -O- (CH 2) o -OSO 3 m, wherein each of these groups may be preferred per se.
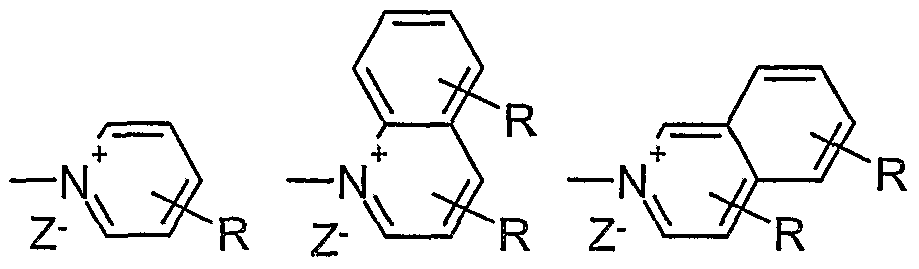
- X is a cationic polar group selected from -NR 1 R 2 R 3 + Z-, -PR 1 R 2 R 3 + Z-,
- R is H or C 1-4 -alkyl in any position
- Z - is Cl-, Br-, I-, CH 3 SO 3 -, CF 3 SO 3 -, CH 3 PhSO 3 -, PhSO 3 -
- R 1, R 2 and R 3 each independently are H, C I-30 -alkyl
- Ar is an unsubstituted or mono- or polysubstituted aromatic ring or fused ring systems having 6 to 18 carbon atoms, in which also one or two CH groups may be replaced by N.
- the preferred cationic groups include in particular -NR 1 R 2 R 3 + Z and
- Each of these groups may be preferred per se.
- X is a nonionic polar group selected from -Cl, -Br, -I, - (OCH 2 CH 2 ) H rOH, -O- (glycoside) o , - (OCH 2 CH 2 ) m -OCH 2 -CHOH-CH 2 -OH,
- n is 0 or 1
- o is an integer in the range of 1 to 10
- p is 1 or 2
- R 1 and R 2 are each independently are C 1-3 o-alkyl, Ar or -CH 2 Ar and,
- Ar is an unsubstituted, mono- or polysubstituted aromatic ring or fused ring systems with 6 to 18 C
- Glycoside stands for an etherified carbohydrate, preferably for a mono-, tri- or oligo-glucoside, all Z in each case independently of one another represent -H, -Cl, -F, -NR 1 R 2 ,
- the preferred non-ionic polar groups include in particular - (OCH 2 CH 2 ) m -OH and -O- (glycoside) o , wherein each of these groups can be taken alone taken.
- those compounds of the formulas I, II or III may be preferred or preferably used in which X is a polymerizable group selected from
- m is an integer from the range from 0 to 1000 and R and R 1 is H or Ci -4 alkyl.
- R and R 1 is H or Ci -4 alkyl.
- those compounds may be preferred or preferably used in which X is an amphoteric group selected from the functional groups of
- X is a group selected from
- Particularly preferred compounds include the compounds shown in the following table:
- the compounds which can be used according to the invention as surfactants are in particular suitable for use as water repellents, for example for surface modification of textiles, paper, glass, porous building materials or adsorbents, or as surface mediators or emulsifiers, in particular for the production of fluoropolymers, or as viscosity reducers or Emulsifier, in particular in paints, lacquers or preparations for surface coating, or as foam stabilizer, in particular in preparations which are known as "fire-extinguishing foams", or in metalworking for covering galvanic baths against the escape of caustic vapors, or as wetting agents in the Production of photographic films and papers, or as leveling agents in self-gloss emulsions, or as fire-extinguishing agents, and for dirt-repellent equipment.
- the compounds which can be used according to the invention as surfactants are suitable for washing and cleaning applications, in particular of textiles. Cleaning and polishing hard surfaces is also a possible field of application for the compounds which can be used according to the invention as surfactant.
- the compounds which can be used according to the invention as surfactants can advantageously be used in cosmetic products, such as foam baths and hair shampoos or as emulsifiers in creams and lotions.
- a further field of application for the compounds which can be used according to the invention as surfactant is flotation, ie the application and separation of ores and minerals from deaf rock.
- preferred the According to the invention can be used as a surfactant compounds as emulsifiers in foods. Further fields of application lie in the metal treatment, as leather auxiliary, the building chemistry and in the plant protection.
- surfactants of the invention are also useful as antimicrobial agents, especially as reagents for antimicrobial surface modification.
- X is a cationic polar group or a polymerizable group.
- the compounds to be used according to the invention are usually introduced into correspondingly prepared preparations for use.
- Corresponding agents which are also the subject of the present invention, contain at least one surface-active compound having at least one end group Y, where Y is CF 3 O- or F 5 S- and a suitable carrier for the respective purpose and optionally other specific active ingredients and possibly adjuvants.
- Preferred agents are dyestuff and lacquer preparations, fire-extinguishing agents, lubricants, detergents and cleaners, de-icers or water repellents for textile finishing or glass treatment.
- the agents are water repellents for finishing textiles and carpets.
- water repellents based on polysiloxanes, fluorocarbons or mixtures of aluminum or
- the water takes on the spherical shape due to the cohesive forces and rolls off the textile surface.
- agents according to the invention are dyestuff and lacquer preparations, fire-extinguishing agents (powders and foams), lubricants, detergents and de-icers.
- the preparation of the compounds to be used according to the invention can be carried out by methods known to the skilled person from the literature.
- the aliphatic OCF 3 group can be obtained, for example, from alcohols via the fluorodeulfurization of xanthates (K. Kanie, Y. Tanaka, K. Suzuki, M. Kuroboshi, T. Hiyama, Bull. Chem. Soc. Jpn., 2000, 73, 471-484, P. Kirsch, Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications, Wiiey-VCH, Weinheim, 2004, pp. 67 ff., Pp. 144 f.).
- the corresponding disclosure to the mentioned method in the cited references thus expressly also belongs to the disclosure content of the present application.
- the introduction of the hydrophilic, anionic, cationic, reactive or polymerizable end group is possible via the corresponding ⁇ -OCF 3 alcohol by methods known to the person skilled in the art. Examples are given in the following scheme:
- the aliphatic SF 5 group may, for. B. at terminal double bonds via the radical addition of SF 5 CI or SF 5 Br are inserted.
- Optional subsequent dehydrohalogenation and hydrogenation allow the variation of the end groups according to formulas IIIa, INb or INc.
- the first two of these reaction steps are described in the literature (R. Winter, PG Nixon, GL Gard, DH Radford, NR Holcomb, DW Grainger, J. Fluorine Chem. 2001, 107, 23-30), catalytic hydrogenations in the presence of an SF 5 function as well (P. Kirsch, M. Bremer, M. Heckmeier, K. Tarumi, Angew Chem 1999, 111, 2174-2178; Angew Chem. Int.
- the present invention therefore relates to a process for preparing a compound of the formula I, which comprises first preparing a compound of the formula IV
- 2a and 2b are prepared as described in R. Winter, P.G. Nixon, G.L. Gard, D.H. Radford, N.R. Holcomb, D.W. Grainger, J. Fluorine Chem. 2001, 107, 23-30.
- methyl iodide 35.9 g, 253 mmol, 1.2 eq
- the reaction mixture is slowly warmed to RT and stirred for 24 h.
- 10% NH 4 Cl solution 200 ml
- the phases are separated and the organic phase is washed and concentrated to dryness.
- the reaction mixture was diluted with water and methyl t-butyl ether and then filtered off.
- the organic phase is dried and purified by column chromatography with heptane.
- the crude sulfonic acid is suspended in 100 ml of ethanol and treated with 1, 4 g (35.22 mmol, 1.2 eq) of sodium hydroxide and heated to 97 ° C for 1 h. After cooling the suspension precipitated crystals, which are filtered off and dried.
- the biochemical degradability of the compounds is determined according to the Zahn-Wellens test according to the publication of the European Commission: Classification, Packaging and Labeling of Dangerous Substances in the European Union, Part II - Test Methods, Annex V - Methods for
- test substances approx. 100 to 200 mg / l as DOC Aeration: with purified air Processing of the samples: filtration (medium-hard filter)
- Example 5 The sodium-10-trifluoromethoxy-decane-1-sulfonate from Example 5 is investigated. The measured values are reproduced in the following table and are shown graphically in FIG.
- Used measuring module ring
- the surfactant according to the invention produces the same surface tension at a significantly lower concentration compared to the hydrocarbon surfactant.
- the curve extrapolation suggests that the final value in the case of the surfactant according to the invention should also be significantly lower than in the case of the hydrocarbon surfactant.
- FIG. 1 shows the biochemical degradability of sodium -10-trifluoromethoxy-decane-1-sulfonate in the Zahn-Wellems test (DOC values) and the fluoride release during the test according to example 8.
- FIG. 2 shows the change in the surface tension of water as a function of the surfactant concentration for sodium-10-trifluoromethoxy-decane-1-sulfonate and sodium decanesulfonate according to Example 9.
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/813,314 US8008358B2 (en) | 2005-01-05 | 2005-12-22 | Fluorosurfactants |
| JP2007549816A JP2008526792A (ja) | 2005-01-05 | 2005-12-22 | フルオロ界面活性剤 |
| EP05822049A EP1833786B1 (de) | 2005-01-05 | 2005-12-22 | Fluortenside |
| DE502005009054T DE502005009054D1 (de) | 2005-01-05 | 2005-12-22 | Fluortenside |
| AT05822049T ATE457978T1 (de) | 2005-01-05 | 2005-12-22 | Fluortenside |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE102005000858A DE102005000858A1 (de) | 2005-01-05 | 2005-01-05 | Fluortenside |
| DE102005000858.5 | 2005-01-05 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2006072401A1 true WO2006072401A1 (de) | 2006-07-13 |
Family
ID=35997784
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2005/013859 WO2006072401A1 (de) | 2005-01-05 | 2005-12-22 | Fluortenside |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US8008358B2 (de) |
| EP (2) | EP1833786B1 (de) |
| JP (1) | JP2008526792A (de) |
| KR (1) | KR20070091642A (de) |
| CN (1) | CN101098853A (de) |
| AT (1) | ATE457978T1 (de) |
| DE (2) | DE102005000858A1 (de) |
| ES (1) | ES2341356T3 (de) |
| TW (1) | TW200635938A (de) |
| WO (1) | WO2006072401A1 (de) |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008003444A1 (de) * | 2006-07-04 | 2008-01-10 | Merck Patent Gmbh | Fluortenside |
| DE102008024221A1 (de) | 2008-05-19 | 2009-11-26 | Merck Patent Gmbh | Herstellung von CF3O-Gruppen enthaltenden Verbindungen |
| DE102008031599A1 (de) | 2008-07-07 | 2010-01-14 | Merck Patent Gmbh | Fluortenside |
| DE102009034194A1 (de) | 2009-07-22 | 2011-01-27 | Merck Patent Gmbh | Materialien für elektronische Vorrichtungen |
| CN101481438B (zh) * | 2009-02-06 | 2011-03-16 | 苏州大学 | 无规共聚含氟大分子乳化剂及其制备方法 |
| US8049022B2 (en) | 2006-07-04 | 2011-11-01 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Fluorosurfactants |
| DE102011114651A1 (de) | 2010-10-29 | 2012-05-03 | Merck Patent Gmbh | Oberflächenaktive Substanzen mit terminaler Pentafluorsulfoxy-Gruppe |
| WO2012084118A1 (de) | 2010-12-21 | 2012-06-28 | Merck Patent Gmbh | Derivate von perfluoroalkoxy-sulfosuccinaten als oberflächenaktive tenside |
| WO2014012661A1 (de) | 2012-07-18 | 2014-01-23 | Merck Patent Gmbh | Fluortenside |
| WO2014023397A2 (de) | 2012-08-06 | 2014-02-13 | Merck Patent Gmbh | Tensidmischungen |
| WO2014095036A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Fluortenside |
| DE102014115381A1 (de) | 2014-10-22 | 2016-04-28 | Abcr Gmbh | Verfahren zur Trifluoromethoxylierung sowie trifluoromethoxylierte Verbindungen |
| DE102016013066A1 (de) | 2016-11-03 | 2018-05-03 | Merck Patent Gmbh | Fluortenside |
| US10315989B2 (en) | 2014-02-21 | 2019-06-11 | Merck Patent Gmbh | Fluorinated tensides |
| US10392332B2 (en) | 2014-07-28 | 2019-08-27 | Merck Patent Gmbh | Fluorinated tensides |
| US10464874B2 (en) | 2014-02-21 | 2019-11-05 | Merck Patent Gmbh | Fluorinated tensides |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102006031262A1 (de) * | 2006-07-04 | 2008-01-10 | Merck Patent Gmbh | Fluortenside |
| DE102006031143A1 (de) * | 2006-07-04 | 2008-01-24 | Merck Patent Gmbh | Fluortenside |
| DE102006032391A1 (de) * | 2006-07-04 | 2008-01-17 | Merck Patent Gmbh | Fluortenside |
| DE102008027930A1 (de) * | 2008-06-12 | 2009-12-17 | Merck Patent Gmbh | Fluortenside |
| US8524104B1 (en) * | 2008-08-28 | 2013-09-03 | Ansul, Incorporated | Fluoroalkenyl sulfate surfactants |
| DE102009030846A1 (de) | 2009-06-26 | 2010-12-30 | Merck Patent Gmbh | Fluortenside |
| US9920141B2 (en) | 2009-12-18 | 2018-03-20 | Solvay Specialty Polymers Italy S.P.A. | Process for manufacturing a dispersion of a vinylidene fluoride polymer |
| WO2011082770A2 (de) | 2010-01-07 | 2011-07-14 | Merck Patent Gmbh | Fluortenside |
| GB201012661D0 (en) * | 2010-07-28 | 2010-09-15 | Dow Corning | Organosilicon hydrophobing agents |
| DE102011114650A1 (de) | 2010-10-29 | 2012-05-03 | Merck Patent Gmbh | Oberflächenaktive Substanzen mit terminaler Pentafluorsulfoxy-Gruppe |
| DE102011117844A1 (de) | 2010-12-02 | 2012-06-14 | Merck Patent Gmbh | Hydrolysierbare Tenside |
| JP6246830B2 (ja) | 2012-12-14 | 2017-12-13 | ビーエーエスエフ ソシエタス・ヨーロピアBasf Se | 50nm以下のライン間寸法を有するパターン化材料を処理する際におけるアンチパターン崩壊を回避するための、界面活性剤及び疎水剤を含む組成物の使用 |
| US9126889B2 (en) | 2013-09-04 | 2015-09-08 | Honeywell International Inc. | Fluorosurfactants having improved biodegradability |
| CN105924375B (zh) * | 2016-04-19 | 2018-04-20 | 巨化集团技术中心 | 一种含氟乳化剂的制备方法 |
| CN110787720B (zh) * | 2019-11-04 | 2023-04-25 | 济南齐氟新材料技术有限公司 | 一种含氟双子表面活性剂及其制备方法 |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3102903A (en) * | 1961-10-31 | 1963-09-03 | Du Pont | Carbonylic compounds containing the sf5 function |
| US4849540A (en) * | 1988-06-24 | 1989-07-18 | The United States Of America As Represented By The Secretary Of The Navy | Pentafluorothio polynitroaliphatic explosives |
| US6110976A (en) * | 1996-02-06 | 2000-08-29 | 3M Innovative Properties Company | Perfluoro (alkoxycycloalkane) carbonyl fluoride compositions and their use |
| US20030138380A1 (en) * | 1995-06-07 | 2003-07-24 | Alexey Kabalnov | Gas emulsions stabilized with fluorinated ethers having low Ostwald coefficients |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE1420576A1 (de) * | 1956-07-25 | 1968-12-19 | Monsanto Co | Verfahren zur Herstellung von Mischpolymerisaten |
| GB941392A (en) | 1961-05-16 | 1963-11-13 | Ici Ltd | Pentafluorosulphur-substituted aldehydes |
| GB941393A (en) * | 1961-05-19 | 1963-11-13 | Ici Ltd | Pentafluorosulphur-substituted carboxylic acids |
| US5159105A (en) * | 1990-02-28 | 1992-10-27 | Minnesota Mining And Manufacturing Company | Higher pentafluorosulfanyl-fluoroaliphatic carbonyl and sulfonyl fluorides, and derivatives |
| JPH09111286A (ja) | 1995-10-13 | 1997-04-28 | Nikko Chemical Co Ltd | エマルション組成物及び洗浄剤組成物並びに洗浄方法 |
| JP3801398B2 (ja) | 1999-11-01 | 2006-07-26 | 信越化学工業株式会社 | 反射防止膜材料及びパターン形成方法 |
| JP3320402B2 (ja) * | 2000-06-26 | 2002-09-03 | クラリアント ジャパン 株式会社 | 現像欠陥防止プロセス及び材料 |
| AU2002331258A1 (en) | 2001-07-25 | 2003-02-17 | Ciba Specialty Chemicals Holding Inc. | Perfluoroalkyl-substituted amines, acids, amino acids and thioether acids |
| PL368805A1 (en) * | 2001-08-03 | 2005-04-04 | Glaxo Group Limited | Surfactant compounds and uses thereof |
| WO2004011422A1 (en) | 2002-07-25 | 2004-02-05 | University Of Florida | Method for incorporation of pentafluorosulfanyl (sf5) substituents into aliphatic and aromatic compounds |
| BRPI0413500A (pt) * | 2003-08-12 | 2006-10-10 | Hoffmann La Roche | compostos, processos para a sua preparação, composição farmacêutica que compreende os mesmos, utilização de um composto, métodos para o tratamento e profilaxia de artrite, diabetes, distúrbios alimentares e obesidade |
| US6870068B1 (en) * | 2003-11-14 | 2005-03-22 | Air Products And Chemicals, Inc. | Synthesis of pentafluorosulfuranyl substituted alkanes |
-
2005
- 2005-01-05 DE DE102005000858A patent/DE102005000858A1/de not_active Withdrawn
- 2005-12-22 AT AT05822049T patent/ATE457978T1/de not_active IP Right Cessation
- 2005-12-22 US US11/813,314 patent/US8008358B2/en not_active Expired - Fee Related
- 2005-12-22 WO PCT/EP2005/013859 patent/WO2006072401A1/de active Application Filing
- 2005-12-22 EP EP05822049A patent/EP1833786B1/de not_active Not-in-force
- 2005-12-22 JP JP2007549816A patent/JP2008526792A/ja active Pending
- 2005-12-22 DE DE502005009054T patent/DE502005009054D1/de active Active
- 2005-12-22 EP EP10001399A patent/EP2208723A1/de not_active Withdrawn
- 2005-12-22 KR KR1020077015473A patent/KR20070091642A/ko not_active Application Discontinuation
- 2005-12-22 ES ES05822049T patent/ES2341356T3/es active Active
- 2005-12-22 CN CNA200580045990XA patent/CN101098853A/zh active Pending
-
2006
- 2006-01-05 TW TW095100464A patent/TW200635938A/zh unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3102903A (en) * | 1961-10-31 | 1963-09-03 | Du Pont | Carbonylic compounds containing the sf5 function |
| US4849540A (en) * | 1988-06-24 | 1989-07-18 | The United States Of America As Represented By The Secretary Of The Navy | Pentafluorothio polynitroaliphatic explosives |
| US20030138380A1 (en) * | 1995-06-07 | 2003-07-24 | Alexey Kabalnov | Gas emulsions stabilized with fluorinated ethers having low Ostwald coefficients |
| US6110976A (en) * | 1996-02-06 | 2000-08-29 | 3M Innovative Properties Company | Perfluoro (alkoxycycloalkane) carbonyl fluoride compositions and their use |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8049022B2 (en) | 2006-07-04 | 2011-11-01 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Fluorosurfactants |
| WO2008003444A1 (de) * | 2006-07-04 | 2008-01-10 | Merck Patent Gmbh | Fluortenside |
| US8067625B2 (en) | 2006-07-04 | 2011-11-29 | Merck Patent Gesellschaft Mit Beschrankter Haftung | Fluorosurfactants |
| DE102008024221A1 (de) | 2008-05-19 | 2009-11-26 | Merck Patent Gmbh | Herstellung von CF3O-Gruppen enthaltenden Verbindungen |
| WO2009141053A1 (de) * | 2008-05-19 | 2009-11-26 | Merck Patent Gmbh, | Herstellung von cf3o-gruppen enthaltenden verbindungen |
| DE102008031599A1 (de) | 2008-07-07 | 2010-01-14 | Merck Patent Gmbh | Fluortenside |
| CN101481438B (zh) * | 2009-02-06 | 2011-03-16 | 苏州大学 | 无规共聚含氟大分子乳化剂及其制备方法 |
| US10125214B2 (en) | 2009-07-22 | 2018-11-13 | Merck Patent Gmbh | Materials for electronic devices |
| WO2011009522A2 (de) | 2009-07-22 | 2011-01-27 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| DE102009034194A1 (de) | 2009-07-22 | 2011-01-27 | Merck Patent Gmbh | Materialien für elektronische Vorrichtungen |
| DE102011114651A1 (de) | 2010-10-29 | 2012-05-03 | Merck Patent Gmbh | Oberflächenaktive Substanzen mit terminaler Pentafluorsulfoxy-Gruppe |
| WO2012084118A1 (de) | 2010-12-21 | 2012-06-28 | Merck Patent Gmbh | Derivate von perfluoroalkoxy-sulfosuccinaten als oberflächenaktive tenside |
| WO2014012661A1 (de) | 2012-07-18 | 2014-01-23 | Merck Patent Gmbh | Fluortenside |
| US9481643B2 (en) | 2012-07-18 | 2016-11-01 | Merck Patent Gmbh | Fluorosurfactants |
| WO2014023397A2 (de) | 2012-08-06 | 2014-02-13 | Merck Patent Gmbh | Tensidmischungen |
| US10590262B2 (en) | 2012-08-06 | 2020-03-17 | Merck Patent Gmbh | Surfactant mixtures |
| WO2014095036A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Fluortenside |
| US10315989B2 (en) | 2014-02-21 | 2019-06-11 | Merck Patent Gmbh | Fluorinated tensides |
| US10464874B2 (en) | 2014-02-21 | 2019-11-05 | Merck Patent Gmbh | Fluorinated tensides |
| US10392332B2 (en) | 2014-07-28 | 2019-08-27 | Merck Patent Gmbh | Fluorinated tensides |
| DE102014115381A1 (de) | 2014-10-22 | 2016-04-28 | Abcr Gmbh | Verfahren zur Trifluoromethoxylierung sowie trifluoromethoxylierte Verbindungen |
| WO2018083110A1 (de) | 2016-11-03 | 2018-05-11 | Merck Patent Gmbh | Fluortenside |
| DE102016013066A1 (de) | 2016-11-03 | 2018-05-03 | Merck Patent Gmbh | Fluortenside |
| US11535589B2 (en) | 2016-11-03 | 2022-12-27 | Merck Patent Gmbh | Fluorinated tensides |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20070091642A (ko) | 2007-09-11 |
| DE102005000858A1 (de) | 2006-07-20 |
| EP2208723A1 (de) | 2010-07-21 |
| ES2341356T3 (es) | 2010-06-18 |
| JP2008526792A (ja) | 2008-07-24 |
| EP1833786B1 (de) | 2010-02-17 |
| DE502005009054D1 (de) | 2010-04-01 |
| US8008358B2 (en) | 2011-08-30 |
| ATE457978T1 (de) | 2010-03-15 |
| US20080149878A1 (en) | 2008-06-26 |
| TW200635938A (en) | 2006-10-16 |
| EP1833786A1 (de) | 2007-09-19 |
| CN101098853A (zh) | 2008-01-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1833786B1 (de) | Fluortenside | |
| DE102006031143A1 (de) | Fluortenside | |
| DE102006031151A1 (de) | Fluortenside | |
| WO2008003444A1 (de) | Fluortenside | |
| DE102006031262A1 (de) | Fluortenside | |
| EP2035131A2 (de) | Fluortenside | |
| EP2282988A1 (de) | Fluortenside | |
| EP2445871A1 (de) | Fluortenside | |
| WO2011082770A2 (de) | Fluortenside | |
| DE1668368A1 (de) | Neue Kohlenstoff-Sauerstoff-Fluor-Verbindungen und Verfahren zu deren Herstellung | |
| EP2655324A1 (de) | Derivate von perfluoroalkoxy-sulfosuccinaten als oberflächenaktive tenside | |
| DE102008031599A1 (de) | Fluortenside | |
| WO2015124290A1 (de) | Fluortenside | |
| WO2014012661A1 (de) | Fluortenside | |
| WO2016015830A1 (de) | Fluortenside | |
| DE2749329A1 (de) | Fluorhaltige alkyl-sulfato-betaine und verfahren zu deren herstellung | |
| EP3535241A1 (de) | Fluortenside | |
| EP2768924A1 (de) | Verwendung von tris(2-hydroxyphenyl)-methan-derivaten für die tertiäre erdölförderung | |
| DE102011114650A1 (de) | Oberflächenaktive Substanzen mit terminaler Pentafluorsulfoxy-Gruppe | |
| WO2019063452A1 (de) | Verfahren zur herstellung von fluorierten verbindungen | |
| DD283994A5 (de) | Verfahren zur herstellung neuer fluortenside |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005822049 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11813314 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007549816 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580045990.X Country of ref document: CN Ref document number: 1020077015473 Country of ref document: KR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2827/KOLNP/2007 Country of ref document: IN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005822049 Country of ref document: EP |