WO2004010519A1 - リチウム二次電池用正極材料の製造方法 - Google Patents
リチウム二次電池用正極材料の製造方法 Download PDFInfo
- Publication number
- WO2004010519A1 WO2004010519A1 PCT/JP2003/002027 JP0302027W WO2004010519A1 WO 2004010519 A1 WO2004010519 A1 WO 2004010519A1 JP 0302027 W JP0302027 W JP 0302027W WO 2004010519 A1 WO2004010519 A1 WO 2004010519A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- lithium
- positive electrode
- compound
- lithium secondary
- carbonate
- Prior art date
Links
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/04—Processes of manufacture in general
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G45/00—Compounds of manganese
- C01G45/12—Manganates manganites or permanganates
- C01G45/1221—Manganates or manganites with a manganese oxidation state of Mn(III), Mn(IV) or mixtures thereof
- C01G45/1242—Manganates or manganites with a manganese oxidation state of Mn(III), Mn(IV) or mixtures thereof of the type [Mn2O4]-, e.g. LiMn2O4, Li[MxMn2-x]O4
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G51/00—Compounds of cobalt
- C01G51/40—Cobaltates
- C01G51/42—Cobaltates containing alkali metals, e.g. LiCoO2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G51/00—Compounds of cobalt
- C01G51/40—Cobaltates
- C01G51/42—Cobaltates containing alkali metals, e.g. LiCoO2
- C01G51/44—Cobaltates containing alkali metals, e.g. LiCoO2 containing manganese
- C01G51/54—Cobaltates containing alkali metals, e.g. LiCoO2 containing manganese of the type [Mn2O4]-, e.g. Li(CoxMn2-x)04, Li(MyCoxMn2-x-y)O4
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Nickelates
- C01G53/42—Nickelates containing alkali metals, e.g. LiNiO2
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01G—COMPOUNDS CONTAINING METALS NOT COVERED BY SUBCLASSES C01D OR C01F
- C01G53/00—Compounds of nickel
- C01G53/40—Nickelates
- C01G53/42—Nickelates containing alkali metals, e.g. LiNiO2
- C01G53/44—Nickelates containing alkali metals, e.g. LiNiO2 containing manganese
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/364—Composites as mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/362—Composites
- H01M4/366—Composites as layered products
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/50—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese
- H01M4/505—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of manganese of mixed oxides or hydroxides containing manganese for inserting or intercalating light metals, e.g. LiMn2O4 or LiMn2OxFy
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/58—Selection of substances as active materials, active masses, active liquids of inorganic compounds other than oxides or hydroxides, e.g. sulfides, selenides, tellurides, halogenides or LiCoFy; of polyanionic structures, e.g. phosphates, silicates or borates
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
- C01P2002/52—Solid solutions containing elements as dopants
- C01P2002/54—Solid solutions containing elements as dopants one element only
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/88—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by thermal analysis data, e.g. TGA, DTA, DSC
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/61—Micrometer sized, i.e. from 1-100 micrometer
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/11—Powder tap density
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/12—Surface area
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/40—Electric properties
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/021—Physical characteristics, e.g. porosity, surface area
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/131—Electrodes based on mixed oxides or hydroxides, or on mixtures of oxides or hydroxides, e.g. LiCoOx
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/485—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of mixed oxides or hydroxides for inserting or intercalating light metals, e.g. LiTi2O4 or LiTi2OxFy
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Definitions
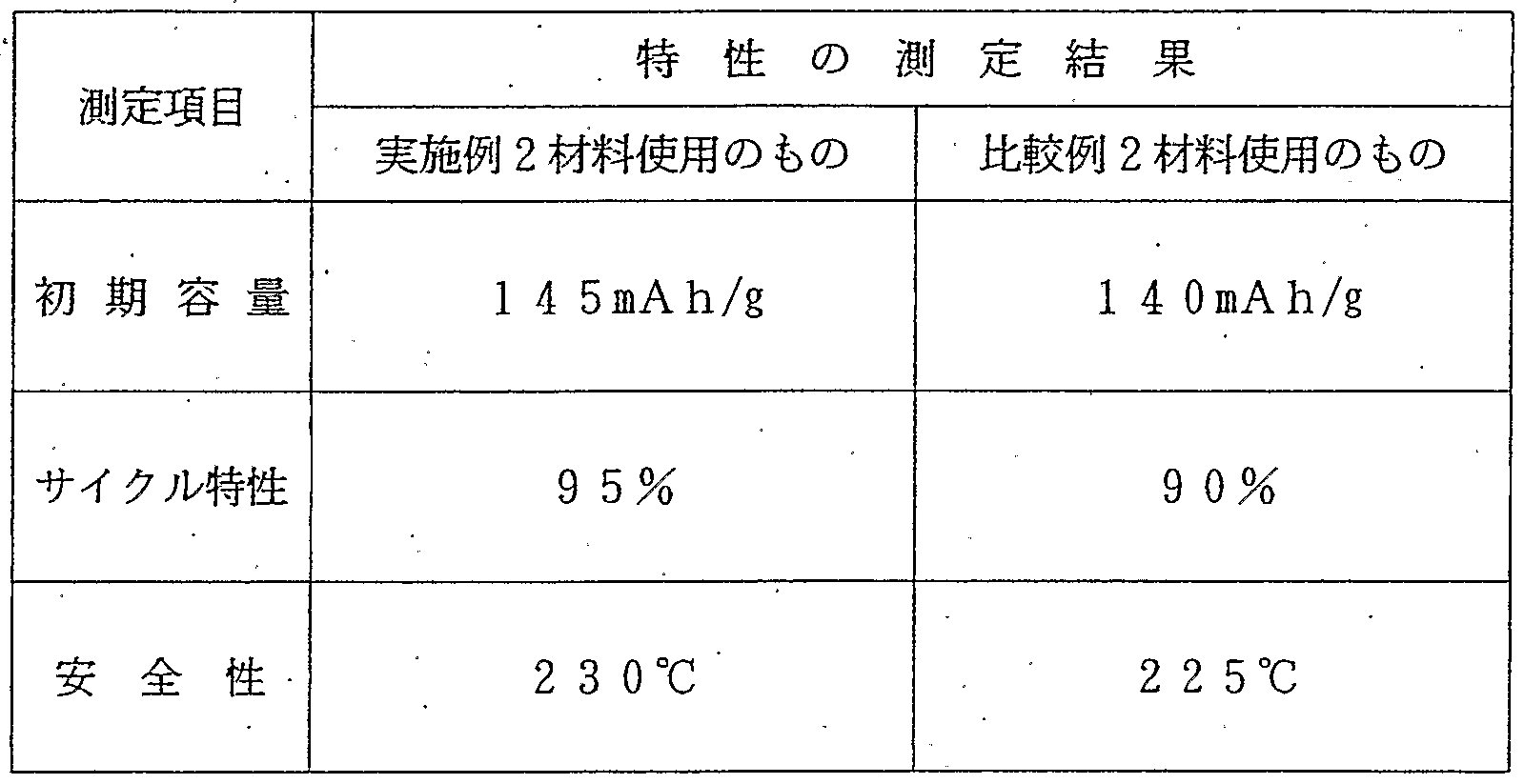
- the present invention relates to a method for producing a positive electrode material for a lithium secondary battery which contributes to the improvement of battery performance such as initial capacity, cycle characteristics and high temperature safety.
- This lithium secondary battery is composed of three basic elements: "positive electrode”, “negative electrode” and “separator holding electrolyte” interposed between both electrodes.
- a slurry obtained by mixing and dispersing an active material, a conductive material, a binder and, if necessary, a plasticizer in a dispersion medium in the positive electrode and the negative electrode' is used to collect metal foil, metal mesh, etc. What is coated on the body is used.
- lithium 'cobalt complex oxide Li x Co 0 2 : 0 x 1 x
- lithium-manganese complex oxide Li x Mn 2 0 4 1 0 ⁇ X ⁇ 1. 2 etc.
- a material capable of occluding and releasing lithium foil or lithium ion for example, a carbonaceous material such as coke-based carbon or graphite-based carbon is generally applied.
- a substance having electron conductivity for example, natural graphite, carbon black, acetylene black, etc.
- a substance having electron conductivity for example, natural graphite, carbon black, acetylene black, etc.
- Fluororesins such as polytetrafluoroethylene (PTFE), polyvinylidene fluoride (PVDF), hexafluoropropylene (HFP), copolymers of these, and the like are generally used.
- organic solvents capable of dissolving the binding material such as acetone, methyl ethyl ketone (MEK), tetrahydrofuran (THF), dimethyl formamide, dimethylacetamide, tetramethyl urea ,-phosphate trimethyl, as the N- methyl-2-pyrrolidone (NM P) such a plasticizer to be added if necessary c used, after the slurry one is deposited is coated on a current collector
- NM P N- methyl-2-pyrrolidone
- the “organic solvent” that can be substituted with the electrolytic solution is suitable, and phthalic acid diesters are preferable.
- copper foil or aluminum foil is generally used as a current collector to which the slurry is applied.
- the slurry necessary for coating is prepared by kneading the active material, the conductive material, the binder, the dispersion medium and the plasticizer at a predetermined ratio.
- Various coating methods such as gravure coating, blade coating, comma coating and dip coating can be applied.
- liquid electrolytes to be held in a separator liquid, polymer or solid electrolytes are known, but liquid electrolytes composed of a solvent and a lithium salt dissolved in the solvent are often used. It is done.
- a solvent polyethylene carbonate, ethylene carbonate, dimethyl sulfoxide, dimethyl lactone, butyl lactone, sulfolane, 1, 2-dimethexetane, tetrahydrofuran, getilka monocarbonate, methyl etylca-ponate, dimethyl ether.
- Organic solvents such as carbonates are suitable, and as lithium salts, LiC 3 F 3 S 0 3 , LiCIO 4, LiB F 4 , LiP F 6 and the like are preferred.
- manganese complex oxides and lithium cobalt complex oxides etc. are mainly composed of compounds (such as manganese oxide and cobalt oxide) and lithium compounds (such as lithium carbonate), which are main components of positive electrode materials for lithium secondary batteries. It is synthesized by mixing at a predetermined ratio and heat treating it.
- a small amount of other element is doped in the positive electrode material for lithium secondary batteries such as the above-mentioned lithium 'mangan complex oxide / lithium' cobalt complex oxide
- a compound such as manganese oxide or cobalt oxide
- a lithium compound such as lithium carbonate
- a compound of a doping element cobalt oxide And manganese carbonate etc. are mixed at a predetermined ratio and heat-treated to synthesize a positive electrode material for a lithium secondary battery.
- the cathode material for lithium secondary batteries such as“ lithium-manganese composite oxide or lithium′-cobalt composite oxide for improving performance ”
- the doping method “lithium secondary such as manganese oxide or cobalt oxide etc.
- a metal compound to be the main component of a battery positive electrode material and a fine powder of a doped element compound such as cobalt oxide, manganese carbonate, etc.
- the compound of the doping element is deposited and deposited on the surface of the metal compound as the main component of the positive electrode material for the secondary battery by a chemical method, and then subjected to such treatment as the main component of the positive electrode material for lithium secondary battery And a lithium compound such as lithium carbonate are mixed and fired to stabilize the positive electrode material for achieving a lithium secondary battery exhibiting excellent initial capacity, cycle characteristics, and safety. It is possible to obtain the knowledge that it can be obtained.
- the present invention has been made based on the above-mentioned findings and the like, and the following items (1) to
- An aqueous solution of a salt of another element is dropped into an alkaline solution, a carbonate solution or a bicarbonate solution in which a metal compound as a main component of a positive electrode material for lithium secondary batteries is suspended,
- the compound of the other element is deposited and deposited on the surface of the metal compound to be the metal compound, and then the main compound having the other element compound deposited and deposited thereon and the lithium compound are mixed and fired.
- “Other elements” are transition metals (Sc, Ti, V, Cr, Mn, Fe, Ni and Cu), alkali metals (L i, Na, K, Rb, Cs and Fr), alkali Cathode material for a lithium secondary battery according to item (1) or (2), which is at least one of alkaline earth metals (Be, Mg, Ca, Sr, Ba and Ra), B and M Manufacturing method.
- a compound according to the present invention characterized in that the molar ratio of the metal to the “other element” in the “compound of the main metal” is 9 9: 1 to 40:60.
- the metal in the “compound of the metal as the main component” is Mn, and the element “Sayaya” is one or more of Co, Ni, M, Mg and Ti.
- the metal in the "compound of the metal as the main component” is Co, and the “other element” is one or more of Mn, Ni, M, Mg and Ti.
- the metal in the “compound of the metal as the main component” is Ni, and the “other element” is one or more of Co, Mn, M, Mg, and Ti.
- Examples of the “compound of the metal that is the main component of the positive electrode material for lithium secondary batteries” include, for example, cobalt oxides that are the main components of the positive electrode materials for lithium-cobalt complex oxide secondary batteries, lithium manganese, In addition to oxides of manganese, which are the main component of positive electrode materials for complex oxide secondary batteries, and oxides of Nigel, which are the main components of positive electrode materials for lithium-nickel composite oxide batteries, and metal oxides And compounds such as carbonates and hydroxides. In addition, hydroxides and oxides produced by coprecipitation method can also be used.
- Examples of the “alkaline solution” for suspending the “compound of metal that is the main component of the positive electrode material for lithium secondary batteries” include an aqueous solution of lithium hydroxide and sodium hydroxide. Aqueous solutions of potassium carbonate and potassium hydroxide, etc. As the “carbonate solution” used for the same purpose, an aqueous solution of sodium carbonate and aqueous potassium carbonate etc. can be used. Examples include aqueous sodium hydrogen carbonate solution and aqueous sodium carbonate carbonate solution.
- lithium hydrogen carbonate aqueous solution prepared by blowing carbon dioxide gas into lithium carbonate aqueous solution can also be used.
- the “salt of other element” is a salt of a doped metal element that is considered to be effective for property improvement, and specifically, transition metals (Sc, Ti, V, Cr, Mn, Fe , Co, Ni and Cu), alkali metals (Li, Na, K, Rb, Cs and Fr), alkaline earth metals (Be, Mg, Ca, Sr, Ba and Ra), sulfuric acid containing B or Al Salt, nitrate, chloride or organic acid salt can be mentioned.
- transition metals Sc, Ti, V, Cr, Mn, Fe , Co, Ni and Cu
- alkali metals Li, Na, K, Rb, Cs and Fr
- alkaline earth metals Be, Mg, Ca, Sr, Ba and Ra
- sulfuric acid containing B or Al Salt nitrate, chloride or organic acid salt
- an aqueous solution of the above-mentioned “salt of other element” is dropped to a solution in which the above “compound of metal to be main component of positive electrode material for lithium secondary battery” is suspended.
- the compound of “other element” is precipitated out on the surface of “”, and in this case, the molar ratio of the metal to the “other element” in the “compound of the main metal” is 9: 1 to 40: It is good to set it as 6 0, and various performance can be obtained stably by this.
- lithium compound which is mixed and fired with the main compound in which other element compounds are deposited and deposited it is not particularly limited, but lithium carbonate which is widely used for manufacturing positive electrode materials for lithium secondary batteries. These firing conditions may also be known conditions applied to the production of a positive electrode material for lithium secondary batteries.
- the method of the present invention mixes the conventional powder compound of the metal to be doped with the positive electrode material powder. Method is not adopted.
- a lithium-manganese composite oxide based positive electrode material Powdery main compounds such as manganese oxide in the case of, cobalt oxide in the case of lithium.cobalt composite oxide type positive electrode material, or nickel oxide in the case of lithium.nickel composite oxide type positive electrode material, alkaline solution, carbonate Suspend in a solution or bicarbonate solution (for example, an aqueous solution of sodium bicarbonate), and add an aqueous solution of a salt of a metal (other element) to be doped, such as cobalt sulfate or manganese sulfate, into it.
- a solution or bicarbonate solution for example, an aqueous solution of sodium bicarbonate
- cobalt carbonate which is a reaction product of cobalt sulfate
- manganese oxide particles which is a main compound
- a doped positive electrode material for lithium secondary battery can be obtained.
- Manganese carbonate prepared by bubbling carbon dioxide gas into an aqueous solution of manganese of manganese metal according to the method disclosed in the above-mentioned Japanese Patent Application Laid-Open No. 2 0 0 0 8 2 0 3 5 1 is oxidized,
- the manganese oxide having an average particle diameter of 1 O m obtained by the above was used as a main raw material for producing a positive electrode material for a lithium secondary battery.
- the resulting material had an average particle size of 10 m, a specific surface area of 0.4 m 2 / g, and a burst density of 2. mm.
- alkali metals represented by Na were less than 500 ppm, and S was also a material with high purity of less than 100 ppm.
- the lithium secondary battery for evaluation has a positive electrode of the above-mentioned positive electrode sample and a lithium foil applied to the counter electrode together with a type 2 coin cell type, and an electrolyte of 1 mol of LiP F 6 Dissolved in a solvent having a ratio of 1: 1 ethylene force / bonate) / DM C (dimethyl carbonate)
- a solvent having a ratio of 1: 1 ethylene force / bonate) / DM C (dimethyl carbonate) The
- D S C differential thermal analysis
- the resulting material had an average particle size of 6 m, a specific surface area of 1.4 m 2 / g, and a tap density of 2.2 g / cc.
- Ca was 500 ppm and S was 150 ppm, it was consistent with the impurity grade of the commercially available oxidized cobalt, and there was no contamination by the reaction.
- lithium carbonate was dissolved in water to form an aqueous solution, and carbon dioxide gas was blown into the solution to prepare 0.3 liters of lithium hydrogen carbonate aqueous solution 6 liters of lithium hydrogen carbonate.
- a commercially available nickel oxide (average particle size 6) was added to this lithium carbonate aqueous solution.
- m, specific surface area 2.0 m 2 / g, tap density 2.4 g / cc) 1 kg is suspended, 0.20 mol / ⁇ cobalt sulfate aqueous solution is dropped to this at a rate of 0.2 _ g / hr
- the mixture was reacted and washed with water to obtain nickel oxide coated with cobalt carbonate on the surface.
- Example 3 material After mixing lithium carbonate so that the Li / (Ni + Co) ratio is 1.00, these are calcined in air at 750 ° C. for 10 hours, and 20% Co-doped lithium nigerate (LiNi 0 2 ) (this is referred to as Example 3 material).
- the average particle size of the obtained material was 8 m.
- the specific surface area was 2.2 mVg, and the tap density was 2. lg / cc.
- Lithium secondary battery for evaluation was a 2 0 3 2 type coin cell manner of applying the lithium foil to both counter With each positive sample the positive electrode, also in the electrolytic solution, 1 mole of LiP F 6 to EC (ethylene Carbonate) ZDMC Dissolved in a solvent with a ratio of 1: 1 (dimethyl carbonate)
- Manganese oxide having the same powder properties as used in Example 1 was used as the main raw material for producing a positive electrode material for lithium secondary batteries, and 1 kg of it was obtained by dissolving lithium carbonate in water. The solution is blown with carbon dioxide gas and suspended in 6 liters of lithium hydrogen carbonate solution of O..35 mol / ⁇ , and 0.20 inol / g aqueous solution of aluminum chloride is added thereto in 0.2 / hr. At the same rate, the same reaction is carried out, and the surface is coated with aluminum hydroxide and oxidized. I got manganese.
- manganese oxide after the above treatment, aluminum hydroxide is uniformly formed on the surface of manganese oxide by SEM (scanning electron microscope) observation and EPMA (electron probe probe microanalyzer). It was confirmed that it was coated, but the molar ratio of Mn to M was 90: 1 0 o
- lithium carbonate is mixed with the above-mentioned “manganese oxide coated with aluminum hydroxide” so that the ratio of Li + Mn + Ajg is 0.55, and then these are mixed to 750 ° C.
- the mixture was calcined in air for 10 hours to obtain 10% Mn-doped lithium manganate (LiMn 2 0 4 ) (this is referred to as Example 3 material).
- the powder properties of the obtained material were an average particle diameter of 1 ⁇ ⁇ specific surface area of 0.8 m 2 / g and a tap density of 2. Og / cc.
- a slurry of 85% of each material, 8% of acetylene black, and 7% of PVDF (polyvinylidene fluoride) was prepared using NMP (N-methyl vinylidene) as a solvent, and this was used as an aluminum foil.
- NMP N-methyl vinylidene
- a positive electrode sample for evaluation of a lithium secondary battery was obtained.
- the lithium secondary battery for evaluation has a positive electrode and the above positive electrode samples and a lithium foil applied to the counter electrode together with a lithium secondary battery, and the electrolyte contains 1 mol of LiP F 6 as EC.
- a solution was used which was dissolved in a solvent having a ratio of 1: 1 ethylene ethylene carbonate: ZDMC (dimethyl carbonate).
- ZDMC dimethyl carbonate
- lithium carbonate was dissolved in water to form an aqueous solution, and carbon dioxide gas was blown into the solution to prepare 6 liters of a 0.35 mol / ⁇ lithium hydrogen carbonate aqueous solution.
- 1 kg of commercially available cobalt oxide having the same powder properties as in Example 2 was suspended in this aqueous lithium carbonate solution, and 0.20 mol / ⁇ aqueous solution of aluminum chloride was dropped thereto at a rate of 0.2 ⁇ / hr.
- the mixture was allowed to react at room temperature for a predetermined time to obtain cobalt oxide having aluminum hydroxide coated on the surface.
- lithium carbonate is mixed with the above-mentioned "Cobalt oxide coated with aluminum hydroxide” so that the Li / (Co + Aj2) ratio is 1.00, and these are mixed at 85.degree.
- the mixture was calcined in air for 0 hour to obtain 5% M-doped lithium cobaltate (LiGoO 2 ) (this is referred to as Example 5 material).
- the powder properties of the obtained material were an average particle diameter of 5111, a specific surface area of 1.5 m 2 / g, and a tap density of 2.2 g / cc.
- a slurry of 7% F (polyvinylidene fluoride) was prepared using NMP (N-methyl vinylidene) as a solvent, this was applied to an aluminum foil, dried, and then press-molded to evaluate a positive electrode sample for lithium secondary battery evaluation.
- the lithium secondary battery for evaluation uses the above positive electrode samples for the positive electrode and a lithium oxide film applied to the counter electrode at the same time as the positive electrode, and the electrolyte solution contains 1 mol of LiP F s as EC (ethylene What was dissolved in a solvent having a ratio of 1: 1 carbonate / DMC (dimethyl carbonate) was used.
- Example 6 First, lithium carbonate was dissolved in water to form an aqueous solution, and carbon dioxide gas was blown into the solution to prepare 6 liters of a 0.35 mol / ⁇ lithium hydrogen carbonate aqueous solution. Subsequently, 1 kg of the commercially available oxidized nickel gel used in Example 3 was suspended in this aqueous solution of lithium carbonate, and an aqueous solution of 0.20 mol / ⁇ of manganese sulfate and sulfate of cobalt sulfate was added thereto to obtain 0.2 ⁇ hr.
- the reaction solution was dropped at a rate and reacted at room temperature for a predetermined time to obtain a nickel oxide coated with manganese carbonate and cobalt carbonate on the surface.
- each carbonate is uniformly formed on the surface of Ni xgel by SEM (scanning electron microscope) observation and EPMA (electron probe probe microanalyzer). It was confirmed that the coating was carried out, but the molar ratio of Ni, Co, and Mn was 60:20:20.
- LiNi 0 2 lithium nickelate
- the material thus obtained had an average particle size of 6 m, a specific surface area of 1.4 m 2 / g, and a tap density of 2. Og / cc.
- Li 'Chiumuni secondary battery for evaluation was a 2 0 3 2 type Koinseru manner that both apply lithium foil as the counter electrode
- a 1 mol ⁇ Li PF s EC Ethylene Carbonate
- the coated manganese carbonate is uniformly coated on the surface of manganese carbonate by the above-mentioned treatment by SEM (scanning electron microscope) observation and EPMA (electron probe microanalyzer).
- SEM scanning electron microscope
- EPMA electron probe microanalyzer
- lithium carbonate is mixed with the above-mentioned “manganese carbonate coated with nickel carbonate” so that the Li / (Mn + Ni) ratio is 1.00, and then these are mixed at 900 ° C.
- the mixture was calcined in the air for time to obtain a lithium-manganese-nickel composite oxide (this is referred to as Example 7 material).
- the average particle size of the obtained material was 8 m / m, the specific surface area was 1.4 m 2 / g, and the tap density was 2. 1 g / cc.
- a slurry of 85% of each material, 8% of acetylene black and 7% of PVDF (polyvinylidene fluoride) was prepared using NMP (N-methyl vinylidene) as a solvent, and this was applied to an aluminum foil. After drying, press molding was performed to obtain a positive electrode sample for evaluation of a lithium secondary battery.
- the lithium secondary battery for evaluation uses the above positive electrode samples as the positive electrode and a lithium secondary battery applied to the counter electrode together with the positive electrode 22 3 type coin cell type, and the electrolyte contains 1 mol of Li 4 PF 6 What was dissolved in a solvent in which the ratio of carbonate) / DMC (dimethyl carbonate) was 1: 1 was used.
- the present invention it is possible to provide a positive electrode material for a lithium secondary battery which enables the production of a lithium secondary battery having excellent initial capacity, cycle characteristics and safety.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Composite Materials (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Secondary Cells (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/521,370 US20050265909A1 (en) | 2002-07-23 | 2003-02-25 | Method for producing positive plate material for lithium secondary cell |
| EP03707039.8A EP1553645B8 (en) | 2002-07-23 | 2003-02-25 | Method for producing positive plate material for lithium secondary cell |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002214450A JP4292761B2 (ja) | 2002-07-23 | 2002-07-23 | リチウム二次電池用正極材料の製造方法 |
| JP2002-214450 | 2002-07-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004010519A1 true WO2004010519A1 (ja) | 2004-01-29 |
Family
ID=30767881
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2003/002027 WO2004010519A1 (ja) | 2002-07-23 | 2003-02-25 | リチウム二次電池用正極材料の製造方法 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US20050265909A1 (ja) |
| EP (1) | EP1553645B8 (ja) |
| JP (1) | JP4292761B2 (ja) |
| KR (1) | KR101015002B1 (ja) |
| CN (1) | CN100340014C (ja) |
| TW (1) | TWI245445B (ja) |
| WO (1) | WO2004010519A1 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102290564A (zh) * | 2011-07-12 | 2011-12-21 | 苏州大学 | 一种二次电池的正极材料及其制备方法 |
| CN103500827A (zh) * | 2013-10-11 | 2014-01-08 | 宁德新能源科技有限公司 | 锂离子电池及其多元正极材料、制备方法 |
| CN104934582A (zh) * | 2015-06-10 | 2015-09-23 | 广东邦普循环科技有限公司 | 一种锂离子电池正极材料湿法包覆钛的方法 |
Families Citing this family (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI279019B (en) * | 2003-01-08 | 2007-04-11 | Nikko Materials Co Ltd | Material for lithium secondary battery positive electrode and manufacturing method thereof |
| JP2005116470A (ja) * | 2003-10-10 | 2005-04-28 | Toyota Central Res & Dev Lab Inc | 非水系リチウム二次電池 |
| JP4916094B2 (ja) * | 2004-03-30 | 2012-04-11 | Jx日鉱日石金属株式会社 | リチウムイオン二次電池正極材料用前駆体とその製造方法並びにそれを用いた正極材料の製造方法 |
| WO2006049001A1 (ja) * | 2004-11-02 | 2006-05-11 | Nippon Mining & Metals Co., Ltd. | リチウム二次電池用正極材料及びその製造方法 |
| CN100344015C (zh) * | 2004-11-06 | 2007-10-17 | 比亚迪股份有限公司 | 一种锂二次电池正极片制备方法及锂离子二次电池 |
| JP2007048711A (ja) * | 2005-08-12 | 2007-02-22 | Sony Corp | 正極活物質およびその製造方法、並びに電池 |
| WO2007083457A1 (ja) * | 2006-01-20 | 2007-07-26 | Nippon Mining & Metals Co., Ltd. | リチウムニッケルマンガンコバルト複合酸化物及びリチウム二次電池 |
| EP2048262B1 (en) * | 2006-07-27 | 2018-09-12 | JX Nippon Mining & Metals Corporation | Lithium-containing transition metal oxide target, process for producing the same and method for producing lithium ion thin-film secondary battery |
| JP5344111B2 (ja) * | 2007-03-30 | 2013-11-20 | 戸田工業株式会社 | 非水電解液二次電池用マンガン酸リチウムの製造方法、並びに非水電解液二次電池 |
| TW200941804A (en) * | 2007-12-12 | 2009-10-01 | Umicore Nv | Homogeneous nanoparticle core doping of cathode material precursors |
| JP4968944B2 (ja) * | 2008-02-01 | 2012-07-04 | 日本化学工業株式会社 | 複合炭酸塩およびその製造方法 |
| US9059465B2 (en) * | 2008-04-17 | 2015-06-16 | Jx Nippon Mining & Metals Corporation | Positive electrode active material for lithium ion battery, positive electrode for secondary battery, and lithium ion battery |
| KR101250710B1 (ko) * | 2008-12-05 | 2013-04-03 | 제이엑스 닛코 닛세키 킨조쿠 가부시키가이샤 | 리튬 이온 전지용 정극 활물질, 동 정극 활물질을 사용한 2 차 전지용 정극 및 2 차 전지 정극을 사용한 리튬 이온 2 차 전지 |
| US9172086B2 (en) | 2008-12-05 | 2015-10-27 | Samsung Sdi Co., Ltd. | Cathode and lithium battery using the same |
| KR101578706B1 (ko) | 2008-12-05 | 2015-12-18 | 삼성에스디아이 주식회사 | 캐소드 및 이를 채용한 리튬 전지 |
| US9051827B1 (en) | 2009-09-02 | 2015-06-09 | Simbol Mining Corporation | Selective removal of silica from silica containing brines |
| US8741256B1 (en) | 2009-04-24 | 2014-06-03 | Simbol Inc. | Preparation of lithium carbonate from lithium chloride containing brines |
| US8637428B1 (en) | 2009-12-18 | 2014-01-28 | Simbol Inc. | Lithium extraction composition and method of preparation thereof |
| US9034294B1 (en) | 2009-04-24 | 2015-05-19 | Simbol, Inc. | Preparation of lithium carbonate from lithium chloride containing brines |
| US10190030B2 (en) | 2009-04-24 | 2019-01-29 | Alger Alternative Energy, Llc | Treated geothermal brine compositions with reduced concentrations of silica, iron and lithium |
| US10935006B2 (en) | 2009-06-24 | 2021-03-02 | Terralithium Llc | Process for producing geothermal power, selective removal of silica and iron from brines, and improved injectivity of treated brines |
| CN101635355B (zh) * | 2009-06-30 | 2011-11-02 | 深圳市源源新材料科技有限公司 | 一种用于生产锂电池的正极材料的制作方法 |
| ES2475740T3 (es) | 2010-02-17 | 2014-07-11 | Simbol, Inc. | Proceso para la preparación de carbonato de litio de alta pureza |
| CN102054976B (zh) * | 2010-12-10 | 2012-11-21 | 王志勇 | 一种锂离子电池正极材料的制备方法 |
| US20130101893A1 (en) * | 2011-10-25 | 2013-04-25 | Apple Inc. | High-voltage lithium-polymer batteries for portable electronic devices |
| CN102659183A (zh) * | 2012-03-06 | 2012-09-12 | 苏州大学 | 正极材料Li1-x-yNaxKyMn2O4制备方法 |
| EP2973802B1 (en) | 2013-03-12 | 2023-08-30 | Apple Inc. | High voltage, high volumetric energy density li-ion battery using advanced cathode materials |
| KR101587055B1 (ko) * | 2013-08-19 | 2016-01-20 | 주식회사 엘지화학 | 수명특성이 우수한 리튬 코발트계 복합 산화물 및 이를 포함하는 이차전지용 양극 활물질 |
| JP6134439B2 (ja) | 2014-03-26 | 2017-05-24 | Jx金属株式会社 | LiCoO2スパッタリングターゲット及びその製造方法並びに正極材薄膜 |
| US9716265B2 (en) | 2014-08-01 | 2017-07-25 | Apple Inc. | High-density precursor for manufacture of composite metal oxide cathodes for Li-ion batteries |
| WO2017058650A1 (en) | 2015-09-30 | 2017-04-06 | Hongli Dai | Cathode-active materials, their precursors, and methods of preparation |
| CN115395008A (zh) | 2016-03-14 | 2022-11-25 | 苹果公司 | 用于锂离子电池的阴极活性材料 |
| JP6430427B2 (ja) * | 2016-03-17 | 2018-11-28 | Jx金属株式会社 | コバルト酸リチウム焼結体及び該焼結体を用いて作製されるスパッタリングターゲット及びコバルト酸リチウム焼結体の製造方法並びにコバルト酸リチウムからなる薄膜 |
| WO2018057584A1 (en) | 2016-09-20 | 2018-03-29 | Apple Inc. | Cathode active materials having improved particle morphologies |
| KR102223565B1 (ko) | 2016-09-21 | 2021-03-04 | 애플 인크. | 리튬 이온 배터리용 표면 안정화된 캐소드 재료 및 이의 합성 방법 |
| US10604414B2 (en) | 2017-06-15 | 2020-03-31 | Energysource Minerals Llc | System and process for recovery of lithium from a geothermal brine |
| WO2019103461A2 (ko) | 2017-11-21 | 2019-05-31 | 주식회사 엘지화학 | 양극활물질 전구체, 그 제조 방법, 이를 이용해 제조된 양극 활물질, 양극 및 이차전지 |
| US11695108B2 (en) | 2018-08-02 | 2023-07-04 | Apple Inc. | Oxide mixture and complex oxide coatings for cathode materials |
| WO2020029028A1 (zh) * | 2018-08-06 | 2020-02-13 | 中天新兴材料有限公司 | 正极材料的制备方法、正极材料及锂离子电池 |
| US11749799B2 (en) | 2018-08-17 | 2023-09-05 | Apple Inc. | Coatings for cathode active materials |
| US11757096B2 (en) | 2019-08-21 | 2023-09-12 | Apple Inc. | Aluminum-doped lithium cobalt manganese oxide batteries |
| CN111342008A (zh) * | 2020-02-25 | 2020-06-26 | 华南理工大学 | 一种氟化钾掺杂富锂锰基材料及其制备方法和应用 |
| CN112670451B (zh) * | 2020-12-29 | 2022-11-11 | 西北工业大学 | 提高水下航行器用锂锰电池能量密度的有机复合正极制备方法 |
| CN114988491A (zh) * | 2022-05-27 | 2022-09-02 | 荆门市格林美新材料有限公司 | 一种双金属交替掺杂的镍锰前驱体及其制备方法和应用 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001202959A (ja) * | 2000-01-18 | 2001-07-27 | Sumitomo Metal Mining Co Ltd | 非水系電解質二次電池用正極活物質およびその製造方法 |
| JP2001256975A (ja) * | 2000-03-14 | 2001-09-21 | Toyota Central Res & Dev Lab Inc | リチウム二次電池正極活物質用リチウムニッケル複合酸化物、その製造方法およびそれを用いたリチウム二次電池 |
| JP2001328818A (ja) * | 2000-05-19 | 2001-11-27 | National Institute For Materials Science | 層状リチウムコバルトマンガン酸化物粒子粉末、およびその製造方法 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4101716A (en) * | 1977-08-15 | 1978-07-18 | Exxon Research & Engineering Co. | Use of high surface area mixed metal oxides of manganese and calcium in electrochemical processes |
| JPH10134811A (ja) * | 1996-10-25 | 1998-05-22 | Nikki Kagaku Kk | リチウム電池正極材の製法 |
| US6242134B1 (en) * | 1996-11-07 | 2001-06-05 | Matsushita Electric Industrial Co., Ltd. | Method of producing positive active material for non-aqueous electrolyte secondary batteries |
| JP2870741B2 (ja) * | 1997-04-14 | 1999-03-17 | 堺化学工業株式会社 | マンガン酸リチウム粒子状組成物及びその製造方法並びにリチウムイオン二次電池 |
| JP4022937B2 (ja) * | 1997-04-24 | 2007-12-19 | 宇部興産株式会社 | リチウムイオン非水電解質二次電池 |
| US6017654A (en) * | 1997-08-04 | 2000-01-25 | Carnegie Mellon University | Cathode materials for lithium-ion secondary cells |
| US6428766B1 (en) * | 1998-10-27 | 2002-08-06 | Toda Kogyo Corporation | Manganese oxide, lithium manganese complex oxide and cobalt-coated lithium manganese complex oxide, and preparation processes thereof |
| DE69903469T2 (de) * | 1998-11-20 | 2003-08-14 | Fmc Corp | Dotierte lithium-mangan-mischoxide und verfahren zur herstellung derselben |
| DE10014884A1 (de) * | 2000-03-24 | 2001-09-27 | Merck Patent Gmbh | Beschichtete Lithium-Mischoxid-Partikel und ein Verfahren zu deren Herstellung |
| GB0117235D0 (en) * | 2001-07-14 | 2001-09-05 | Univ St Andrews | Improvements in or relating to electrochemical cells |
| TWI279019B (en) * | 2003-01-08 | 2007-04-11 | Nikko Materials Co Ltd | Material for lithium secondary battery positive electrode and manufacturing method thereof |
| US20050130042A1 (en) * | 2003-12-11 | 2005-06-16 | Byd America Corporation | Materials for positive electrodes of lithium ion batteries and their methods of fabrication |
-
2002
- 2002-07-23 JP JP2002214450A patent/JP4292761B2/ja not_active Expired - Lifetime
-
2003
- 2003-02-25 WO PCT/JP2003/002027 patent/WO2004010519A1/ja active Application Filing
- 2003-02-25 CN CNB038173883A patent/CN100340014C/zh not_active Expired - Lifetime
- 2003-02-25 KR KR1020057001169A patent/KR101015002B1/ko active IP Right Grant
- 2003-02-25 EP EP03707039.8A patent/EP1553645B8/en not_active Expired - Lifetime
- 2003-02-25 US US10/521,370 patent/US20050265909A1/en not_active Abandoned
- 2003-08-15 TW TW092122476A patent/TWI245445B/zh not_active IP Right Cessation
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001202959A (ja) * | 2000-01-18 | 2001-07-27 | Sumitomo Metal Mining Co Ltd | 非水系電解質二次電池用正極活物質およびその製造方法 |
| JP2001256975A (ja) * | 2000-03-14 | 2001-09-21 | Toyota Central Res & Dev Lab Inc | リチウム二次電池正極活物質用リチウムニッケル複合酸化物、その製造方法およびそれを用いたリチウム二次電池 |
| JP2001328818A (ja) * | 2000-05-19 | 2001-11-27 | National Institute For Materials Science | 層状リチウムコバルトマンガン酸化物粒子粉末、およびその製造方法 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1553645A4 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102290564A (zh) * | 2011-07-12 | 2011-12-21 | 苏州大学 | 一种二次电池的正极材料及其制备方法 |
| CN103500827A (zh) * | 2013-10-11 | 2014-01-08 | 宁德新能源科技有限公司 | 锂离子电池及其多元正极材料、制备方法 |
| CN104934582A (zh) * | 2015-06-10 | 2015-09-23 | 广东邦普循环科技有限公司 | 一种锂离子电池正极材料湿法包覆钛的方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101015002B1 (ko) | 2011-02-16 |
| EP1553645A1 (en) | 2005-07-13 |
| JP2004055472A (ja) | 2004-02-19 |
| TWI245445B (en) | 2005-12-11 |
| TW200507326A (en) | 2005-02-16 |
| EP1553645A4 (en) | 2008-04-09 |
| CN100340014C (zh) | 2007-09-26 |
| CN1672275A (zh) | 2005-09-21 |
| JP4292761B2 (ja) | 2009-07-08 |
| EP1553645B8 (en) | 2019-05-29 |
| KR20050027256A (ko) | 2005-03-18 |
| EP1553645B1 (en) | 2019-03-27 |
| US20050265909A1 (en) | 2005-12-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004010519A1 (ja) | リチウム二次電池用正極材料の製造方法 | |
| EP2214234B1 (en) | Li-ni-based composite oxide particle powder for rechargeable battery with nonaqueous elctrolyte, process for producing the powder, and rechargeable battery with nonaqueous electrolyte | |
| KR101989760B1 (ko) | 정극 활성 물질 전구체 입자 분말 및 정극 활성 물질 입자 분말, 및 비수전해질 이차 전지 | |
| WO2011078389A1 (ja) | 複合酸化物の製造方法、リチウムイオン二次電池用正極活物質およびリチウムイオン二次電池 | |
| CN113302159B (zh) | 制备电极活性材料的方法 | |
| EP2395582A1 (en) | Positive active material for rechargeable lithium battery, method of preparing the same, and rechargeable lithium battery including the same | |
| JP4655599B2 (ja) | 非水系電解質二次電池用正極活物質とその製造方法、およびこれを用いた非水系電解質二次電池 | |
| JP5733571B2 (ja) | リチウム含有複合酸化物の製造方法、正極活物質および二次電池 | |
| WO2008018634A1 (fr) | Manganate de lithium spinelle, procédé servant à produire celui-ci, matière active d'électrode positive utilisant le manganate de lithium spinelle et batterie à électrolyte non aqueux | |
| JP2009263176A (ja) | マグネシウムアルミニウム複合酸化物表面被覆スピネル型マンガン酸リチウム及びその製造方法、並びにそれを使用する正極活物質及び非水電解質電池 | |
| JP5641132B2 (ja) | リチウム含有複合酸化物の製造方法、正極活物質および二次電池 | |
| JP5828282B2 (ja) | 非水電解質二次電池用活物質の製造方法およびそれを用いた二次電池 | |
| JP3495639B2 (ja) | リチウム・マンガン複合酸化物とその製造法並びに該複合酸化物を使用したリチウム二次電池 | |
| EP4110733B1 (en) | Process for making an electrode active material, and electrode active material | |
| JP5003030B2 (ja) | 非水電解質二次電池用正極活物質およびその製造方法、ならびにそれを備えた非水電解質二次電池 | |
| CN115989194A (zh) | 制备掺杂正极活性材料的方法 | |
| WO2023180231A1 (en) | Process for making a doped cathode active material | |
| JP2001297767A (ja) | リチウム二次電池用正極材料及びリチウム二次電池 | |
| US20230406724A1 (en) | Process for making a coated cathode active material | |
| JP2005135861A (ja) | 非水電解質電気化学セル | |
| JP2005135861A5 (ja) | ||
| JP2008063202A (ja) | プロトン含有型ニッケル系遷移金属酸化物の製造方法およびそれを用いた非水電解質二次電池 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): CN KR US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT SE SI SK TR |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 10521370 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003707039 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020057001169 Country of ref document: KR Ref document number: 20038173883 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020057001169 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003707039 Country of ref document: EP |