WO2004000950A1 - 処理顔料、その用途及び顔料処理用化合物 - Google Patents
処理顔料、その用途及び顔料処理用化合物 Download PDFInfo
- Publication number
- WO2004000950A1 WO2004000950A1 PCT/JP2003/008019 JP0308019W WO2004000950A1 WO 2004000950 A1 WO2004000950 A1 WO 2004000950A1 JP 0308019 W JP0308019 W JP 0308019W WO 2004000950 A1 WO2004000950 A1 WO 2004000950A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- compound
- pigment
- carbodiimide
- parts
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C3/00—Treatment in general of inorganic materials, other than fibrous fillers, to enhance their pigmenting or filling properties
- C09C3/08—Treatment with low-molecular-weight non-polymer organic compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C1/00—Treatment of specific inorganic materials other than fibrous fillers; Preparation of carbon black
- C09C1/44—Carbon
- C09C1/48—Carbon black
- C09C1/56—Treatment of carbon black ; Purification
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y30/00—Nanotechnology for materials or surface science, e.g. nanocomposites
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09C—TREATMENT OF INORGANIC MATERIALS, OTHER THAN FIBROUS FILLERS, TO ENHANCE THEIR PIGMENTING OR FILLING PROPERTIES ; PREPARATION OF CARBON BLACK ; PREPARATION OF INORGANIC MATERIALS WHICH ARE NO SINGLE CHEMICAL COMPOUNDS AND WHICH ARE MAINLY USED AS PIGMENTS OR FILLERS
- C09C3/00—Treatment in general of inorganic materials, other than fibrous fillers, to enhance their pigmenting or filling properties
- C09C3/10—Treatment with macromolecular organic compounds
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/60—Particles characterised by their size
- C01P2004/64—Nanometer sized, i.e. from 1-100 nanometer
Definitions
- the present invention relates to a treated pigment and its use, and more specifically, to a coloring that requires good fluidity and stable dispersion in a highly concentrated and finely dispersed state in addition to ordinary printing inks and paints.
- Manufacture of treated pigments intended for use as agents and color filters with good contrast and high transmittance, and black matrices with excellent shielding and insulating properties The present invention relates to a dispersed resist composition.
- the present invention also relates to a pigment-treating compound which is suitable for treating an organic pigment or carbon black, particularly having a surface having an adsorption portion for a basic nitrogen-containing group. Background art
- Pigments have begun to be used as colorants not only in various printing inks and paints, but also in fields that require sharper hues, such as color filters and ink jet inks.
- carbon black is widely used as a pigment with a high degree of blackness, but because of its fine primary particle size, its porous nature, and its large specific surface area, it is still difficult to increase the concentration and dispersion stability. There is a problem that lacks. Recently, attempts have been made to use carbon black as a light shielding material for black matrix. Therefore, there is a problem that it is difficult to obtain a black matrix having high insulation from carbon black having high conductivity.
- a dispersion comprising a reaction product of a poly (lower alkylene) imine and a polyester having a free hydroxyl group in which at least two polyester chains are bonded to each poly (lower alkylene) imine chain.
- JP-A-54-37082 discloses a pigment dispersant obtained by reacting a polyalkylenepolyamine compound with a lipoxyl-terminated polylatatatone compound obtained by ring-opening polymerization of a ratatatone compound using hydroxycarboxylic acid as an initiator.
- JP-A-07-207012 a polyallylamine derivative in which a polyester, a polyamide or the like is introduced into an amino group of polyallylamine is disclosed in JP-A-09-169821. Further, in carbon black having a functional group such as a carboxyl group, Has functional groups such as aziridine group, isocyanate group and epoxy group JP-A-09-166869 discloses a method of reacting a dispersant with a carboxyl group to form a daraft. However, even this method has not been able to improve fluidity and dispersion stability at high concentrations. Not in.
- Such treated pigments proposed by the applicant of the present application are effectively used in printing inks, paints, ink jet recording liquids, and the like, and in particular, the resist composition for the color filter and the resist composition for the black matrix described above. Which can be suitably used in It is.
- carbon black provides a film having high insulation and light-shielding properties, and also has improved effects such as improved developability when used as a resist composition.
- the problem to be solved by the present invention is that even a pigment having no functional group capable of reacting with a carpoimide group on its surface, a treated pigment having good dispersion stability and fluidity in a dispersion medium, a dispersion composition thereof, Another object of the present invention is to provide a resist composition having very excellent development characteristics when a pattern such as a color filter or a black matrix is formed. Another object of the present invention is to provide a pigment treating compound suitable for treating such a pigment.
- the present inventors have conducted intensive studies in order to solve the above-mentioned problems, and as a result, a polyester having an adsorbing portion for a basic nitrogen-containing group and, preferably, a pigment having a functional group capable of further reacting with a carbodiimide group, was converted into a polyester. All of the above problems are solved by treating with a carbodiimide compound having at least one side chain selected from the group consisting of a side chain, a polyether side chain, and a polyacryl side chain and a basic nitrogen-containing group. The present invention has been completed, and the present invention has been completed.
- the present invention provides (1) a pigment comprising, in a molecule, at least one side chain selected from the group consisting of a polyester side chain, a polyether side chain and a polyataryl side chain, and a basic nitrogen-containing group.
- the present invention relates to a treated pigment characterized by being treated with at least one carbodiimide compound.
- the present invention provides (2) the above carbodiimide-based compound, wherein
- the treated pigment according to (1) which is a carbodiimide-based compound containing a basic nitrogen-containing group in an amount of 0.02 to 4 mmol.
- the present invention relates to (3) the treated pigment according to (1) or (2), wherein the pigment is a pigment having an adsorption portion for a basic nitrogen-containing group.
- the present invention provides (4) the carbodiimide compound, wherein the pigment has a functional group capable of reacting with a carbodiimide group, and the carbodiimide compound further has at least one carbodiimide group in a molecule.
- the present invention relates to the treated pigment according to any one of (1) to (3).
- the present invention also provides (5) the pigment having a functional group capable of reacting with the carbodiimide group, wherein the pigment is an organic pigment or a carbon black having a functional group capable of reacting with the carbodiimide group;
- the present invention relates to (6) the treated pigment according to any one of (1) to (5), wherein the carbodiimide-based compound has a basic nitrogen-containing group in a main chain.
- the present invention also provides (7) the treated pigment according to any one of (1) to (6), wherein the basic nitrogen-containing group is a tertiary amino group or a nitrogen-containing complex ring group. About.
- the present invention also relates to (8) a pigment dispersion composition comprising the treated pigment according to any one of (1) to (7) and a dispersion medium.
- the present invention provides (9) at least one side chain selected from the group consisting of a polyester chain, a polyether chain, and a polyacryl chain in a molecule, and at least one basic nitrogen-containing group.
- the present invention relates to a compound for pigment treatment, which is a carposide compound.
- the present invention relates to (10) a pigment-treating compound according to (9), which is a carbopimide compound having at least one carbopimide group in the molecule.
- the treated pigment of the present invention is characterized in that at least one selected from the group consisting of a polyester side chain, a polyether side chain, and a polyacryloline side chain comprises an organic pigment or a carbon black having an adsorption portion for a basic nitrogen-containing group on its surface. It is obtained by treating with a carbodiimide compound having a side chain and a basic nitrogen-containing group.
- the treated pigment of the present invention may be in the form of a pigment dispersion obtained using a dispersion medium or the like, or may be in the form of a dry state obtained without using a dispersion medium.
- the carbodiimide compound when the treated pigment is dispersed in a dispersion medium, has at least one type of side chain selected from the group consisting of a polyester side chain, a polyether side chain, and a polyacryl side chain described below.
- the side chain forms a domain in the dispersion medium and acts as a steric hindrance. It is thought that it can be dispersed stably.
- the pigment is carbon black
- the surface of the carbon black is coated with an insulating material such as the above-mentioned carbodiimide compound, and the pigment-dispersed resist composition for a black matrix containing such treated carbon black is It is thought that very excellent high insulation properties can be obtained.
- Examples of the pigment used in the present invention include organic pigments and carbon black generally used in printing inks and resist compositions for color filters.
- Specific examples of the organic pigment include dye lake pigments. , Azo, benzimidazolone, phthalocyanine, quinacridone, anthraquinone, dioxazine, indigo, thioindico, perylene, perinone, diketopyrrolopyrrole, isoindolinone, nitro, Organic pigments such as nitroso-based, anthraquinone-based, flavanthrone-based, quinophthalone-based, pyranthrone-based, and infrared-based pigments.
- the carbon black preferably has a carboxyl group having a pH of 7.0 or less.
- Carbon black having a group can be used. Specifically, one or more carbon blacks as exemplified below can be used.
- MONARCH1400 As Cabot Corp. of carbon black, MONARCH1400, MONARCH 1 300, MONARCH 1 100, MONARCH 1000, MOGUL- L s REGAL 400 R, as possible out to include REGAL 350 R, and the like.
- Color B lack F w 200 s Color B lack Fw 2 s Color B lack Fw 2 VC Color B lack Fwl, Color B lack Fwl 8 s Color B lack S 1 70, Color B lack S 160 N S pecial B lack 6, S pecial B lack 5, S pecia 1 B 1 ack 4 s S pecial B lack 4A, S pecial B lack 550, S pecia 1 B 1 ack 350, Specific B lack 250, Special B lackl O 0, Printexl 50T N Printe xU, Printe xV, Printexl 40U, Printexl 40V and the like. These are all trade names.
- the pigment preferably has an adsorbing portion for a basic nitrogen-containing group, and the adsorbing portion for the basic nitrogen-containing group is typically an acid group, preferably a carboxyl group ⁇ sulfonic acid.
- a functional group capable of reacting with a basic nitrogen-containing group such as a group. Even if the pigment is untreated and has no adsorbing portion with the basic nitrogen-containing group, as a method for introducing a carboxyl group or a sulfonic acid group, introduction of a normal functional group such as a derivative treatment of the pigment surface and a sulfonation treatment. It can be processed and used in any way.
- the pigment is preferably a pigment having a functional group capable of reacting with the carbodiimide group.
- the functional group capable of reacting with the carpoimide group includes carboimide
- it has at least one functional group selected from the group consisting of a xyl group, a sulfonic acid group, a hydroxyl group and an amino group.
- the functional group can be introduced by surface treatment.
- a functional group capable of reacting with the above-mentioned carbodiimide group can be introduced by heat treatment or oxygen / ultraviolet ray treatment and a low-temperature plasma method described in JP-A-58-217595.
- the carpoimide compound used in the present invention comprises at least one side chain selected from the group consisting of a polyester side chain, a polyether side chain and a polyataryl side chain, and at least one basic nitrogen-containing group in the molecule. It is a carbodiimide-based compound.
- carbodiimide compound refers to a compound obtained by introducing the above-mentioned side chain and a basic nitrogen-containing group based on a compound having a carbodiimide group. Not only those compounds having a carbodiimide group in the molecule of the obtained compound but also those not having the carbodiimide group are included.
- the “side chain” refers to a chain that is branched from the main chain when the carbodiimide compound is used as the main chain. Those formed by combining at least one of a polyester chain and a polyester chain in one side chain belong to both the “polyester side chain” and the “polyether side chain”, Those formed by combining at least one of a polyester chain and a polyacrylic chain in one side chain belong to both the “polyester side chain” and the “polyacrylic side chain” and have one side chain. Those formed by combining one or more polyether chains and polyacryl chains in the chain each belong to both “polyether side chains” and “polyacryl side chains”.
- polyester side chains those formed by combining one or more polyester chains, polyether chains and polyacryl chains in one side chain are referred to as “polyester side chains”, “polyether side chains”, and “polyacrylic chains”.
- Side chain “. Like this Polyester chains, polyether chains, and polyacryl chains may be mixed in one of the side chains. Of course, at least one side selected from the group consisting of polyester side chains, polyether side chains, and polyacryl side chains It is a side chain that satisfies the condition.
- the term "basic nitrogen-containing group” includes not only a nitrogen-containing group that forms a quaternary ammonium ion in water, but also a nitrogen-containing group that acts as a Lewis base. Examples include amino groups and nitrogen-containing heterocyclic groups.
- the carbodiimide-based compound particularly preferably, at least one selected from the group consisting of a polyester chain, a polyether chain, and a polyacryl chain by utilizing a reaction between a carbodiimide group and a functional group capable of reacting therewith.
- Compounds introduced into the molecule can be used.
- such a reaction between the carbodiimide group and the functional group is sometimes referred to as a grafting reaction, and the side chain introduced by the method is grafted, and the introduced polyester side chain is grafted.
- Polyester side chains and polyetherene side chains are sometimes referred to as darafted polyether side chains, and polyataline side chains are sometimes referred to as grafted polyacrylic side chains.
- the carpoimide compound having a grafted side chain includes a compound having one or more carpoimide groups in a molecule, and is composed of a polyester side chain, a polyether side chain and a polyataryl side chain by a grafting reaction.
- Compounds obtained by a method of adding one or more at least one type of side chain selected from the group can be mentioned.
- the compound having one or more carpoimide groups in the molecule for example, a compound obtained by carboimidation of a diisocyanate compound by a decarboxylation reaction in an organic solvent in the presence of a carpoimidization catalyst can be used.
- the diisocyanate compound for the decarboxylation reaction include hexamethylene diisocyanate, isophorone diisocyanate, tolylene diisocyanate, diphenylmethane diisocyanate, cyclohexanediisocyanate, and dicyclohexylmethane diisocyanate.
- Xylylene diisocyanate tetramethylene diisocyanate, tetramethyl xylylene diisocyanate and the like, and aliphatic, alicyclic, aromatic, and aromatic aliphatic diisocyanate compounds.
- usable organic solvents are those having a high boiling point and an isocyanate compound. It does not have active hydrogen that reacts with a product or a compound having a carbodiimide group to be formed.
- aromatic hydrocarbons such as toluene, xylene, and dimethyl ether; diethylene glycol diacetate, dipropylene Glycol ether esters such as dalicol dibutyrate, hexylene glycolone resin acetate, glyco / resiacetate, methyl glycol acetate, ethyl diol alcohol acetate, butyl daricol acetate, ethyl glycol acetate, butyl diglycol acetate, etc .; Ketones such as chirptyl ketone, acetophenone, propiophenone, diisobutyl ketone and cyclohexanone; and aliphatic esters such as amyl acetate, propyl propionate
- Examples of usable carpoimidization catalysts include phosphorenes and phosphorene oxides. Specifically, 1-ethyl-3-methyl-13-phospholene oxide, 1-phenylen-3-methinole Examples thereof include 13-phosphorene oxide and 11-phenyl-13-methyl-2-phosphorenoxide.
- Known methods can be used to perform a decarboxylation reaction of the isocyanate group using these materials.
- the reaction can be performed at a reaction temperature of 100 to 200 ° C. in a nitrogen atmosphere.
- Examples of other methods for obtaining the above-mentioned compound having a carbodiimide group include, for example, U.S. Pat. No. 2,914,156, Japanese Patent Publication No. 47-32279, and Japanese Patent Application Laid-Open No. 5-17. Nos. 8954, JP-A-6-56950 and the like.
- a compound obtained by decarboxylating K ( ⁇ 2) mol of a diisocyanate compound can be represented by the following general formula (1).
- ⁇ is a residue excluding the isocyanate group of the diisocyanate compound used in the synthesis of the carbodiimide compound.
- carbodiimide group represented by the above general formula (1) Commercially available compounds having a carbodiimide group represented by the above general formula (1) include carbodiimides made from tetramethylxylylene didisocyanate, such as lipoposite V-03, V-05, etc. Brand name, manufactured by Nisshinbo Co., Ltd.) It is.
- the compound represented by the general formula (1) has an isocyanate group in the molecule.
- the isocyanate group is first eliminated by a reaction. In some cases, it is preferable to carry out the reaction with the carbodiimide group later.
- a component that forms a side chain by grafting described below has a functional group that can react with an isocyanate group in addition to a functional group that can react with a carpoimide group, and an isocyanate group as compared with a carpoimide group When it has a functional group having the same or higher reactivity, it is preferable that the isocyanate group has disappeared.
- a compound having low reactivity with the remaining carbodiimide group and reacting selectively with the isocyanate group first with the isocyanate group is used to form the carbodiimide compound of the present invention. It can also be used as a compound having one or more carbodiimide groups in the molecule.
- the carbodiimide-based compound as the pigment-treating compound of the present invention has a basic nitrogen-containing group in the molecule.
- a method for obtaining such a carbodiimide-based compound utilizes the reaction with the above isocyanate group. Then, a preferred method is to introduce a basic nitrogen-containing group into the carbodiimide group-containing compound.
- a basic nitrogen-containing group is introduced into a carpoimide group-containing compound, a hydroxyl group suitable as a functional group capable of selectively reacting with an isocyanate group and a carpoimide group ⁇ isocyanate group are considered from the above conditions. It is preferable to use a compound having a tertiary amino group or a basic nitrogen-containing heterocyclic group which does not participate in the reaction.
- compounds having one hydroxyl group and a tertiary amino group include, for example, N, N-dialkylalkanolamines such as N, N-dimethylethanolamine and N, N-jetinoleethanolamine.
- N, N-dialkylalkanolamines such as N, N-dimethylethanolamine and N, N-jetinoleethanolamine.
- examples thereof include a compound, an ethylene oxide adduct of a secondary amine compound, and a reaction product of a secondary amine compound and an epoxy compound.
- Examples of the compound having two hydroxyl groups and a tertiary amino group include, for example, N-anolequinolezalcanolamine compounds such as N-methinoresietananolamine, N-ethinoresietananolamine, and primary amamine compounds.
- a reaction product of a primary amine compound and an epoxy compound examples include tertiary basic nitrogen such as hydroxypyridine, pyridinemethanol, pyridineethanol and the like, pyridine, pyrazine, triazine, quinoline and the like.
- tertiary basic nitrogen such as hydroxypyridine, pyridinemethanol, pyridineethanol and the like
- pyridine, pyrazine, triazine, quinoline and the like Compounds having a heterocycle and a hydroxyl group can be used.
- a heterocyclic compound containing a secondary basic nitrogen such as piperidine / piperazine can be used by alkylating and tertiary and containing a hydroxyl group.
- 2 mol of the compound having one hydroxyl group may be reacted with 1 mol of the compound represented by the above general formula (1) to introduce a basic nitrogen-containing group at the terminal of the main chain.
- Such a compound in which a basic nitrogen-containing group is introduced into the main chain is a more preferable compound.
- compounds having no basic nitrogen-containing group can also be used in the reaction, for example, low-molecular-weight monoalcohol compounds such as methanol and ethanol, monoalkylester compounds of (poly) alkyleneglycol, and hydroxyl groups described below.
- low-molecular-weight monoalcohol compounds such as methanol and ethanol, monoalkylester compounds of (poly) alkyleneglycol, and hydroxyl groups described below.
- One example is a polyester compound, a polyether compound, a polyacryl compound, and the like.
- the compound having one or more carbodiimide groups in the molecule obtained by the above method or the like is further introduced with a polyester side chain, a polyether side chain, or a polyatolinole side chain by a grafting reaction to form a darafted polyester.
- a typical method for introducing these side chains into the molecule by a grafting reaction is a functional group capable of reacting with a carbodiimide group, for example, a carboxyl group, a sulfonate group, a phosphate group, a hydroxyl group, an amino group, or the like.
- a method of reacting a polyester compound, a polyester compound or a polyacryl compound having a carbodiimide group of a carbodiimide compound can be used.
- a ring-opening polymer compound of a cyclic ester compound for example, a mono- or polyoxycarboxylic acid such as lactic acid, caproic acid, 12-hydroxystearic acid, dimethylolpropionic acid, dimethylolbutanoic acid, etc.
- a cyclic ester compound for example, a mono- or polyoxycarboxylic acid such as lactic acid, caproic acid, 12-hydroxystearic acid, dimethylolpropionic acid, dimethylolbutanoic acid, etc.
- Ring-opening polymerization of cyclic ester compounds such as force prolacton, V-lactate ratatone, 2-methylcaprolactone, 4-methylcaprolataton, ⁇ -propiolatatone, ⁇ -valerolatone, and j3-methyl- ⁇ -valerolatone.
- a low molecular weight diol compound as an initiator, the cyclic ester compound polyester diol compounds containing a hydroxyl group obtained by ring-opening polymerization, etc.
- a self-polycondensation compound of an oxycarboxylic acid for example, a polyester compound containing a carboxyl group and a hydroxyl group obtained by polycondensing a monooxycarboxylic acid such as lactic acid, caproic acid, and 12-hydroxystearic acid. Etc.).
- a low-molecular-weight diol compound and a low-molecular-weight dicarboxylic acid compound for example, ethylene dalicol, 1,3-propanediol, 1,4-butanediol, 1,6- Linear glycols such as hexanediol, etc .; low molecular diols such as 1,2-propanediol, neopentyl glycol, 3-methyl-1,5-pentanediol, and branched dalicols such as ethylbutylpropanediol
- a compound component and a low-molecular dicarboxylic acid compound component such as an aromatic dicarbonic acid such as a saturated or unsaturated aliphatic dicarboxylic acid such as conodic acid, adipic acid, azelaic acid, sebacic acid, and maleic acid, and phthalic acid.
- hydroxy for example, ethylene dalicol
- a phosphate ester / ester compound of a ring-opening polymer of a cyclic ester compound using a monoalcohol as an initiator for example, a phosphate group obtained by subjecting the above polyester monool compound to a phosphoric acid esterification reaction.
- Polyester diol compound contained (5) ring-opening polymerization compound of a cyclic ester compound using an amino group-containing sulfonic acid compound as an initiator (for example, using the above-mentioned cyclic esterol compound as an initiator with an amino group-containing sulfonic acid compound such as taurine as an initiator)
- a sulfurous acid gas adduct of a ring-opening polymer of a cyclic ester compound in which a monoalcohol is used as an initiator for example, a sulfonic acid group-containing polyester diol obtained by adding sulfurous acid gas to the above polyester monol compound
- the polyester compound having a phosphoric acid group / a sulfonic acid group is preferably a compound having a polyester chain obtained by ring-opening polymerization of polyproprotatone.
- Ring-opening polymerized compounds of cyclic ether compounds using oxycarboxylic acids, monoalcohols, low molecular weight diol compounds, etc. as initiators for example, lactic acid, cabronic acid, 12-hydroxyxtearic acid, dimethylonolepropionic acid, It is obtained by ring-opening polymerization of a cyclic ether compound such as ethylene oxide, propylene oxide, trimethylene oxide, tetrahydrofuran, or tetrahydrobilan using a mono- or polyoxycarboxylic acid such as dimethylolonebutanoic acid as an initiator.
- oxycarboxylic acids for example, lactic acid, cabronic acid, 12-hydroxyxtearic acid, dimethylonolepropionic acid, It is obtained by ring-opening polymerization of a cyclic ether compound such as ethylene oxide, propylene oxide, trimethylene oxide, tetrahydrofuran, or tetrahydrobilan using
- a phosphate ester compound of a ring-opening polymer of a cyclic ether compound using a monoalcohol as an initiator for example, a phosphoric acid group containing a phosphate group obtained by subjecting the above polyether monool compound to an esterification reaction with phosphoric acid; Polyether compounds).
- a ring-opening polymerization compound of a cyclic ether compound using an amino group-containing sulfonic acid compound as an initiator for example, an amino group-containing sulfonic acid compound such as taurine is opened.
- the initiator include a polyether compound having a sulfonic acid group obtained by ring-opening polymerization of the above cyclic ether compound).
- a sulfurous acid gas adduct of a ring-opening polymer of a cyclic ether compound in which a monoalcohol is used as an initiator for example, a sulfonic acid group-containing polyether obtained by adding a sulfurous acid gas to the above polyether monol compound).
- the polyacryl compound having the functional group is selected from a (meth) acrylic monomer having at least one selected from the group consisting of a carboxyl group, a sulfonate group, a phosphate group, a hydroxyl group, and an amino group.
- (Meth) acrylic polymers obtained by polymerizing at least one monomer component are preferred.
- Examples of such a monomer include (meth) acrylic monomers having a carboxyl group: (meth) acrylic acid, crotonic acid, maleic acid, fumaric acid, cytocholanic acid, itaconic acid, etc .; Acrylic monomers: (meth) sulfoethyl (meth) acrylate, sulfopropyl (meth) acrylate, etc .; (meth) acrylic monomers having a phosphoric acid group: 2-metaryloyloxylacid phosphate, tris Acryloyloxyshethyl phosphate, etc .; (meth) acrylic monomers having a hydroxyl group: 2-hydroxyethyl (meth) acrylate, hydroxypropyl (meth) acrylate, hydroxypentinole (meth) (Meth) acrylic acid hydroxyalkyl esters such as atalylate; having amino groups (Meth) acrylyl monomer: (meth) acrylamide, N-methyl
- (Meth) acrylic polymers obtained by polymerizing known monomers such as alkyl esters and styrene-based monomers Door can be.
- it has the above functional group, and further contains both the polyester chain and the polyether chain.
- a polyacrylic compound having a polyester chain or a polyacrylic compound having a polyether chain is reacted using polyalkylene dalycol or a monoalkyl ether compound thereof as an initiator.
- a compound obtained by reacting a cyclic ether compound with a hydroxyl group-containing polyester compound, a polyester chain is contained in the molecule (meth)
- An acrylic monomer or a polyether chain is contained in the molecule (meth)
- a polyacryl compound obtained by using an acrylic monomer can be mentioned.
- polyester compounds polyether compounds and polyataryl compounds
- the compound having a hydroxyl group reacts with the isocyanate group before the carbodiimide group, making it difficult to control the reaction.
- Compounds having two or more may crosslink during the grafting reaction and cause gelation. Therefore, for a compound having only a hydroxyl group as a functional group, an acid anhydride such as phthalic anhydride, maleic anhydride, or succinic anhydride is further reacted to obtain a polyester compound having one carboxyl group, a polyether compound, or a polyether compound. It is desirable to carry out the grafting reaction after obtaining the acrylic compound.
- polyester compounds polyether compounds or polyacrylic compounds which are introduced into the molecule of the carpoimide-based compound as a side chain, those having a basic nitrogen-containing group have a basic nitrogen-containing group simultaneously with the side chain. This means that it can be introduced into carbodiimide compounds.
- the compound in which at least one of the polyester compounds, polyether compounds, and polyacryl compounds exemplified above is introduced into the molecule by grafting may have zero or more preferably one or more carposide groups.
- a side chain is introduced into the polycarboimide compound represented by the general formula (1) by a grafting reaction, a carpoimide compound represented by the following general formulas (2) and (3) can be obtained. it can.
- A is a residue excluding the isocyanate group of the diisocyanate compound used in the synthesis of the polycarboimide compound.
- X is the same or different, and a trivalent linking group obtained by reacting a carbodiimide group with a functional group capable of reacting with the same.
- Y is the same or different and is a polyester chain, polyether chain or polyacryl chain.
- 1, m and n represent an integer of 1 or more.
- a linking group formed by a reaction between a carboxyimide group and a carboxylic acid group is represented by the following general formulas (4) and (5):
- the linking group formed by the reaction of the carboximido group with the amino group is represented by the following general formula (8):
- the linking group formed from the reaction between the sulfonic acid group and the sulfonic acid group is represented by the following general formula (9), and the linking group formed from the reaction between the carbodiimide group and the phosphoric acid group is represented by the following general formula (10). Is done.
- R represents a hydrogen atom or a hydrocarbon group having 1 or more carbon atoms.
- the carbodiimide compounds represented by the general formulas (2) and (3) A compound having a cyanate group but having a functional group capable of reacting with the isocyanate group in the same manner as described above can also be reacted. As a result, the carposimide group in which the isocyanate group has been eliminated first can be obtained. The same compound as that obtained when the component having a side chain is reacted with the compound having the compound can be obtained.
- a carbodiimide-based compound obtained by reacting a compound having one hydroxyl group with cisocyanate groups at both ends of the carbodiimide-based compound represented by the general formulas (2) and (3) is represented by the following general formula ( 11) and (12), and this compound can also be used as the carbodiimide compound in the present invention.
- sigma 1 and sigma 2 are each independently, may have different structures, of a compound having a functional group reactive with Isoshianeto group, a residue excluding the functional group.
- A is a residue excluding the isocyanate group of the diisocyanate compound used in the synthesis of the polycarbodiimide compound.
- X is the same or different and is a trivalent linking group obtained by reacting a carboximide group with a functional group capable of reacting with the same, and Y is the same or different and is a polyester chain, polyether chain or polyacryl chain.
- 1, m and n represent an integer of 1 or more.
- a ring-opening reaction between a hydroxyl-containing ester compound and an acid anhydride a ring-opening reaction between a cyclic ether compound, a ring-opening reaction between a hydroxyl-containing ether compound and an acid anhydride, a reaction between a carbodiimide group and a carboxyl group or a hydroxyl group, etc.
- a conventional method can be used for the reaction between j-isocyanate group and hydroxyl group.
- the above-mentioned carbodiimide-based compounds when having no carbodiimide group in the molecule, are prepared by: (a) reacting with an acid anhydride such as succinic anhydride or trimellitic anhydride to obtain an acid value; Is considered as one means.
- an acid anhydride such as succinic anhydride or trimellitic anhydride
- the developability is improved when the carboxylic acid group in the molecule reacts with the basic nitrogen-containing group introduced into the molecule free.
- any of these means (a) and (b) may be used.
- a polyester compound containing a functional group capable of reacting with a carbodiimide group is first synthesized in order to introduce a polyester side chain by, for example, grafting. Then, the method of reacting with the carbodiimide group to form a side chain was described.Another method is to react the carbodiimide group with a compound having a functional group capable of reacting with the carbodiimide group, and then form a polyester chain. Side chains can also be introduced by introducing them into the molecule.
- a method may be used in which a polyester conjugate or a cyclic polyether compound is subjected to ring-opening polymerization to graft-bond a polyester chain or a polyether chain.
- a radical polymerizable unsaturated double bond for example, a radical polymerizable unsaturated double bond and A compound having a functional group capable of reacting with a carbodiimide group such as an acid group or a hydroxyl group is first reacted with a carbodiimide group to introduce a radical polymerizable unsaturated double bond into the molecule of the carbodiimide compound.
- a method of polymerizing a system monomer and graft-bonding a polyacryl resin may be used. As for the order of these reactions, it is desirable to synthesize under conditions that minimize undesired side reaction products.
- a compound having at least one carbodiimide group (a compound represented by the general formula (2) or (11)) may be used.
- the compound represented by the above general formula (1) and all of the carbodiimide groups are grafted.
- the following method can be mentioned based on the compound represented by the general formula (3), which is a polyester chain, a grafted polyether chain or a grafted polyacryl chain.
- a diol compound is reacted with an isocyanate group at both ends to form a compound having a hydroxyl group at both ends.
- a compound obtained by reacting 1 mol of the compound represented by the general formula (1) with 2 mol of the diol compound is represented by the following general formula (13), while represented by the general formula (3)
- the compound obtained by reacting 1 mol of the compound with 2 mol of the diol compound is represented by the following general formula (14).
- G is the same or different and represents a residue of the diol compound excluding a hydroxyl group reacted with an isocyanate group.
- a diol compound having the above-mentioned carbodiimide group (which is represented by the general formula (13))
- a diisocyanate compound having a grafted polyester chain, a grafted polyether chain or a grafted polyacryl chain (a compound represented by the general formula (3))
- a diisocyanate compound having a lipoimide group (a compound represented by the general formula (1))
- a diol compound having a graft side chain (the compound represented by the general formula (14)).
- the compound obtained by the reaction is a compound having at least one or more grafted side chains and liposide imide groups.
- a side chain can be easily introduced into a carbodiimide compound by using a grafting reaction.However, it is also possible to introduce a side chain of a carbodiimide compound by a method other than the grafting reaction.
- a method of extending the chain of the polycarbodiimide compound represented by the general formula (1) using a chain extender having a polyester chain, a polyether chain or a polyacryl chain can be used.
- Examples of such a chain extender include compounds obtained by subjecting a cyclic polyester compound to ring-opening polymerization with a polyol compound having three or more hydroxyl groups such as trimethylolpropane and pentaerythritol as having a polyesterol chain, A compound obtained by polycondensation of the low molecular weight didiol compound with a partly triol compound and the above low molecular weight dicarboxylic acid, a diol monocarboxylic acid such as dimethyl alcohol and pionic acid, and an epoxy having a polyester chain in the molecule.
- Examples of the compound include a compound obtained by reacting a compound.
- Examples of the compound having a polyether chain include compounds obtained by subjecting the above-mentioned polyol compound to ring-opening polymerization of the above-mentioned cyclic polyether compound.
- Examples of the compound having a polyacryl chain include a compound obtained by reacting a (meth) acrylic polymer having a carboxyl group with the polyol compound.
- a carbodiimide compound having at least one side chain selected from the group consisting of polyester side chains, polyether side chains and polyacryl side chains and a basic nitrogen-containing group in a molecule obtained by reacting the above materials is
- the amount of the basic nitrogen-containing group per 1 g of the carbopimide compound is 0.02 to 4 min O 1, More preferably, it is 0.05 to 3 mmol.
- the amount of the basic nitrogen-containing group per 1 g of the carbodiimide-based compound is within the above range, good pigment dispersion stability can be obtained.
- the polyester chain, polyether chain and polyacryl chain introduced as side chains are not particularly limited as long as they have an action of improving the dispersion stability of the pigment in the dispersion medium.
- the formula weight is preferably at least 200 and at most 100. More preferably, it is not less than 300 and not more than 500.
- the carbopimide equivalent is preferably from 100 to 500,000, and more preferably from 200 to 100,000. preferable.
- the carbodiimide equivalent is in the above range, when an organic pigment or a carbon black having a functional group capable of reacting with a carbodiimide group is used, the effect of improving the dispersion stability becomes high.
- a color filter having good contrast and high transmittance is obtained, and when carbon black is used, a black matrix having higher insulation and shielding properties is obtained. Can be obtained.
- the carbodiimide equivalent means a number represented by (molecular weight of carbodiimide compound) / (number of carbodiimide groups in carbodiimide compound molecule).
- the number average molecular weight of the carbopimide compound used in the present invention is preferably at least 100, and at most 100, more preferably at least 100, and more preferably at least 100. It is less than or equal to 0 0 0. If the number average molecular weight of the carbodiimide compound is too high, it is suitable for dispersing a pigment having a functional group capable of reacting with a carbodiimide group in a dispersing medium, and for preparing a pigment dispersion resist composition. It is difficult to obtain a pigment having a high viscosity, which is not preferable particularly when a pigment dispersion composition having a high concentration is required.
- the number average molecular weight is too low, the dispersion stability of the above pigment in the dispersing medium and, particularly when carbon black is used as the pigment, the insulating property when a black matrix film is finally formed, will be reduced. It is not preferable because it decreases.
- the amount of basic nitrogen-containing groups in the carposimide-based compound, the polyester side chain present in the molecule is preferable to appropriately adjust the number and formula weight of side chains such as side chains and polyacrylic side chains, carbodiimide equivalents, and the like.
- the positive imide compound can be used as a pigment treatment compound for treating an organic pigment having an adsorption portion with a basic nitrogen-containing group and carbon black, and such a pigment treatment compound is also one of the present invention. It is.
- Carposimide-based compounds having one or more lipodiimide groups in the molecule are suitable for pigment dispersion treatment of organic pigments and carbon black having an adsorption site for basic nitrogen-containing groups and a functional group capable of reacting with carbodiimide groups. It is a compound for pigment treatment.
- the treatment is to treat the entire surface or a part of the surface of the dispersed particles of the pigment with the carbodiimide compound of the present invention.
- a mixture of the above-mentioned pigment, phenol diimide-based compound, and, if necessary, a dispersion medium and other additives is roll-milled, kneaded, a high-speed stirrer, bead mill, ball mill, sand mill, ultrasonic disperser And a method of kneading, dispersing and treating using a high-pressure dispersing apparatus.
- the carbodiimide compound In order to perform the above pigment treatment only with a carbodiimide compound without using a dispersion medium, the carbodiimide compound must be at a temperature higher than the melting point of the carbodiimide compound and a carbodiimide compound having a carbodiimide group in the molecule.
- the temperature for accelerating the above reaction is generally 100 ° C. or lower, preferably about 40 to 80 ° C.
- the treated pigment obtained by such a method is coarsely pulverized or chipped, and can be used by dispersing it in a suitable dispersion medium for any use later.
- the treatment is performed even at a relatively low temperature.
- a carbodiimide-based compound having a carbodiimide group in the molecule and is a functional group capable of reacting with the carbodiimide group.
- a pigment dispersion finely treated in a dispersion medium suitably used in the field of inks and paints can be directly processed into a pigment dispersion as an ink or paint.
- ethylene glycol monomethyl ether ethylene glycol monoethynoleate ether, diethylene glycol monomethinole ether, and diethylene glycol monomethyl ether
- anolexylene glycol ether monoalkyl ethers such as thioglycerol, propylene glycol monomethyl ether, propylene glycol / lemonoethynole ether, ethylene glycol monomethyl ether acetate, ethyl glycolone monoethyl ether etherate,
- anolexylene glycol monoalkynoleate acetates such as propylene glycol monomethylate / rareacetate
- diethylene glycol Ethenoles such as oleether, diethylene glycolone methinoleetinoleate / tetrahydrofuran
- ketones such as methiolexylene glycol ether monoalkyl ethers
- Aromatic hydrocarbons such as n-methylpyrrolidone, N, N-dimethylformamide, N, N-dimethylacetamide; alcohols such as isopropynole alcohol and normal propyl alcohol can be These dispersion media can be used alone or in combination of two or more.
- the aqueous medium may be water alone, or may be used in combination with a water-miscible organic dispersion medium.
- the use ratio of the carbodiimide-based compound used for performing the pigment treatment Is preferably 3 parts by mass or more and 100 parts by mass or less, more preferably 5 parts by mass or more, and 50 parts by mass or less with respect to 100 parts by mass of the pigment (total pigment used).
- the treated pigment thus obtained may contain various binder resins, solvents, surfactants, and other various additives as necessary, and may be used in printing inks, paints, black matrix pigment-dispersed resist compositions, It can be suitably used in applications such as pigment-dispersed resist compositions for filters, inkjet inks, writing implement inks, ribbon inks, and liquid developers.
- a pigment-dispersed resist composition for black matrices and color filters will be described in more detail as an example of a preferred use of the treated pigment of the present invention.
- the pigment-dispersed resist composition is a resist composition having an active energy ray-curable property and capable of developing an alkali, and in addition to the treated pigment of the present invention (the treated car pump rack or the treated organic pigment), a film-forming resin. It is mainly composed of a photopolymerizable compound, a photopolymerization initiator, and a solvent, and is obtained by containing various additives such as a polymerization inhibitor as needed.
- Constituent of the pigment-dispersed resist composition for a black matrix according to the present invention As the bon black, in order to form a highly insulating film, it is preferable to use one that is difficult to form an aggregate once dispersed. .
- the treated car pump rack constituting the pigment dispersion resist composition for the black matrix a small car pump rack having an average primary particle size of 0.15 Aim or less is preferable, but such carbon black is dispersed. In such a case, it is preferable to mix a hue to neutral black by mixing a complementary color pigment as necessary.
- a complementary color pigment As an organic pigment having a functional group capable of reacting with a carbodiimide group for obtaining a treated organic pigment constituting the pigment-dispersed resist composition for a color filter of the present invention, in order to form a film having a clear hue, It is preferable to use a higher organic pigment having a functional group capable of reacting with the carpoimide group.
- the film-forming resin constituting the pigment-dispersed resist composition of the present invention is acrylic.
- Carboxyl group-containing unsaturated monomers such as acid, methacrylic acid, itaconic acid, maleic acid, maleic anhydride, monoalkyl maleate, citraconic acid, citraconic anhydride, citraconic acid mono- / rexyl ester; Hydroxyethyl acrylate, 2-hydroxyxethyl methacrylate, aryl acrylate, aryl methacrylate, benzyl acrylate, benzyl methacrylate, glycerol monomono acrylate, glycerol monomethacrylate, N -A carboxyl group-soluble resin having a carboxyl group, which is a copolymer obtained by reacting with at least one selected from the group consisting of feninolema resin, polystyrene mac-mouth monomer and polymethyl methacrylate macro-monomer, I can illustrate this
- Examples of the photopolymerizable compound constituting the pigment-dispersed resist composition of the present invention include a monomer having a photopolymerizable unsaturated bond, an oligomer, and the like.
- Monomers having one photopolymerizable unsaturated bond in the molecule include methyl methacrylate, ptynolemetharate acrylate, 2-ethynolehexynolemethacrylate, methylenolate acrylate, ptynoleate acrylate, and 2-ethynolehexyl acrylate.
- Aralkyl methacrylates or acrylates such as benzyl methacrylate or benzyl acrylate; alkoxyalkyl methacrylates or acrylates such as butoxyshethyl methacrylate or butoxyshethyl acrylate; N, N —Aminoamino quinolemethacrylate or phthalate such as dimethylaminoethyl methacrylate, N, N-dimethylaminoethyl acrylate; diethyleneglycol monoethylene monoethyl enoate; Methacrylic acid esters or acrylic acid esters of polyalkylene glycol monoalkyl ethers, such as ethylene glycol glycol monobutylinoleate ether and dipropylene glycol monomethyl ether glycol; polyalkylene glycol monoesters such as hexaethylene glycol alcohol monophenylene etherate Examples thereof include methacrylates or acrylates of aryl ethers; isoponyl meth
- Monomers having two or more photopolymerizable unsaturated bonds in the molecule include bispheno Ichinore A dimethacrylate, 1,4-butanediol / resin methacrylate, 1,3-butyleneglycol / resin methacrylate, diethyleneglyconoresin methacrylate, glyceryl monodimethacrylate, neopentinole glycol dimethacrylate, polyethylene glycol dimethacrylate , Polypropylene glycol dimethacrylate, tetraethylene glycol dimethacrylate, trimethylonolepropane trimethacrylate, pentaerythritol noretrimethacrylate, pentaerythritol tetramethacrylate, dipentaerythritol tetramethacrylate, dipentaerythritol hexamethacrylate, Dipentaerythritol pentamethacrylate, Bisphenolole A d
- the photopolymerization initiator constituting the pigment-dispersed resist composition of the present invention is not particularly limited.
- the solvent constituting the pigment-dispersed resist composition of the present invention the same solvents as those described above for the dispersion medium can be used.
- the solvent is normal pressure (1.013 ⁇ 10 2 kPa).
- organic solvents include ethylene glycol monomethyl ether, ethylene glycol mono / ethylene monoethyl ether, ethylene glycol monoethyl isopropyl ether, ethylene glycol mono ethyl enolate, diethylene glycol monomethyl ether, and ethylene glycol monomethyl ether.
- diethylene glycolone monoethylenoate / propylene glycol monopropylene monomethineoleatene, propylene glycolone monoethylenoate ether, propylene glycolone monobutynooleatene, diethylene glycol gethyleenoate, diethylene Ether-based organic solvents such as glyco-resin methinolate and diethylene glycol methino-retino ether; ethylene glycol monomethyl ether acetate, ethylene glycol Monoethynoleate enoleate acetate, ethylene glycol / remo noptinoleate enoleacetate, propylene dali cornole monomethyate / leate enoleate acetate, propylene terephthalate oleate enoleate / reatecetate, etc.
- Ester-based organic solvents such as methyl isobutyl ketone, cyclohexanone, 2-heptanone, and S-butyrolactone; methyl 2-hydroxypropionate, ethinole 2-hydroxypropionate, and 2-hydroxy-1-one 2-Methylethyl propionate, 3-Methyl-3-methoxybutylpropionate, 3-Methoxymethyl propionate, 3-Methylethoxypropionate, 3-Ethoxypropionate methylene, 3-Ethoxypropionic acid Etinole, ethoxy acetate, hydroxyacetic acid Organic solvents such as -methylpyrrolidone, ⁇ , ⁇ ⁇ ⁇ ⁇ -dimethylformamide, ⁇ , ⁇ ⁇ ⁇ ⁇ -dimethylacetamide, etc .; They can be used alone or in combination of two or more.
- diethylene dali corn resin methino oleate diethylene glycol ole methyl ethino oleate, ethylene glycol ole monomethino oleate enole acetate, propylene glycol Noremono Mechinore Ether acetate, propylene glycol olemonoethyl ethereoleate, cyclohexanone, 2-heptanone, ethinole 2-hydroxypropionate, 3-methyl-3-methoxybutylpropionate, 3-methoxypropionate Ethyl acid, methyl 3-ethoxypropionate, n-amyl formate and the like are preferred, and propylene glycol monomethyl ether acetate is more preferred.
- these organic solvents are preferably 50% by mass or more of the total organic solvent used in the pigment-dispersed resist composition of the present invention, in view of the solubility of the alkali-soluble resin, pigment dispersibility, coatability, and the like. Preferably, it is 70% by mass or more.
- the organic solvent does not sufficiently evaporate and remains in the dried coating film when pre-betaing the formed coating film.
- the heat resistance of the dried coating film may decrease.
- the organic solvent having a boiling point of less than 100 ° C. is contained in a large amount, it becomes difficult to apply the solvent uniformly and evenly, and a coating film having excellent surface smoothness may not be obtained.
- pigment-dispersed resist composition of the present invention if necessary, various additives such as other photopolymerizable compounds, thermal polymerization inhibitors, ultraviolet absorbers, and antioxidants may be appropriately used. it can.
- the method for producing the pigment-dispersed resist composition of the present invention is an example of a preferred embodiment of the present invention, and the present invention is not limited to this.
- the above-mentioned photopolymerizable compound, photopolymerization initiator, and film are added to the pigment dispersion composition in which the treated pigment is dispersed by the above method.
- a method can be used in which a forming resin, an organic solvent, and other additives are added as required, and mixed using a high-speed stirring device, a high-pressure emulsifier, a bead mill, a three-roll, sand mill, a kneader, or the like.
- the pigment dispersion composition of the present invention has good dispersion stability and fluidity even when the pigment content is high, and the film formed thereby is a thin film when carbon black is used as the pigment.
- the pigment dispersion composition of the present invention can be suitably used as a black matrix material requiring high insulation properties, and a pigment dispersion resist composition for a color filter requiring high transmittance and high contrast. It is.
- the obtained pigment-dispersed resist composition has extremely excellent developing characteristics that an uncured film can be removed cleanly in a short time during development.
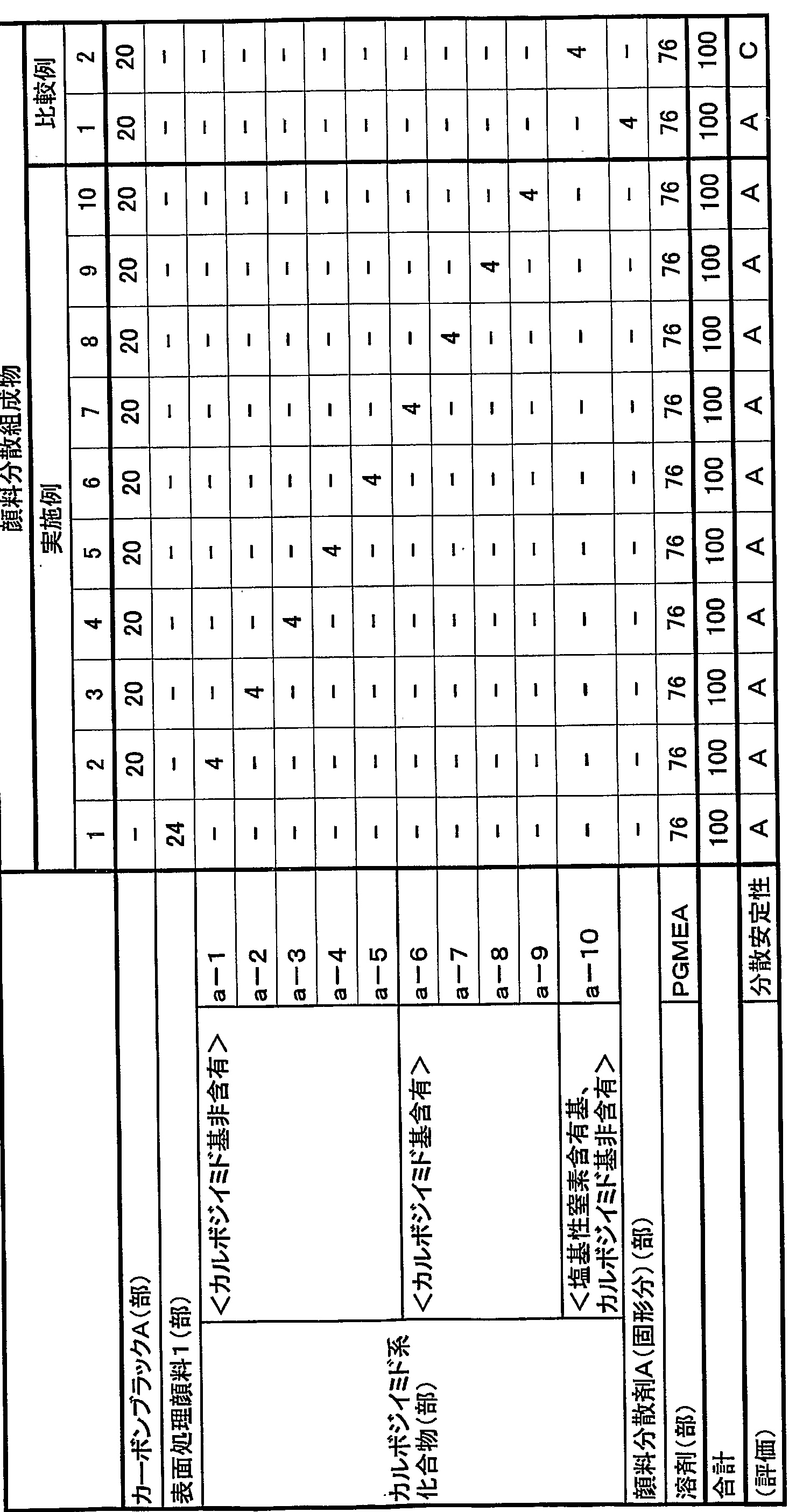
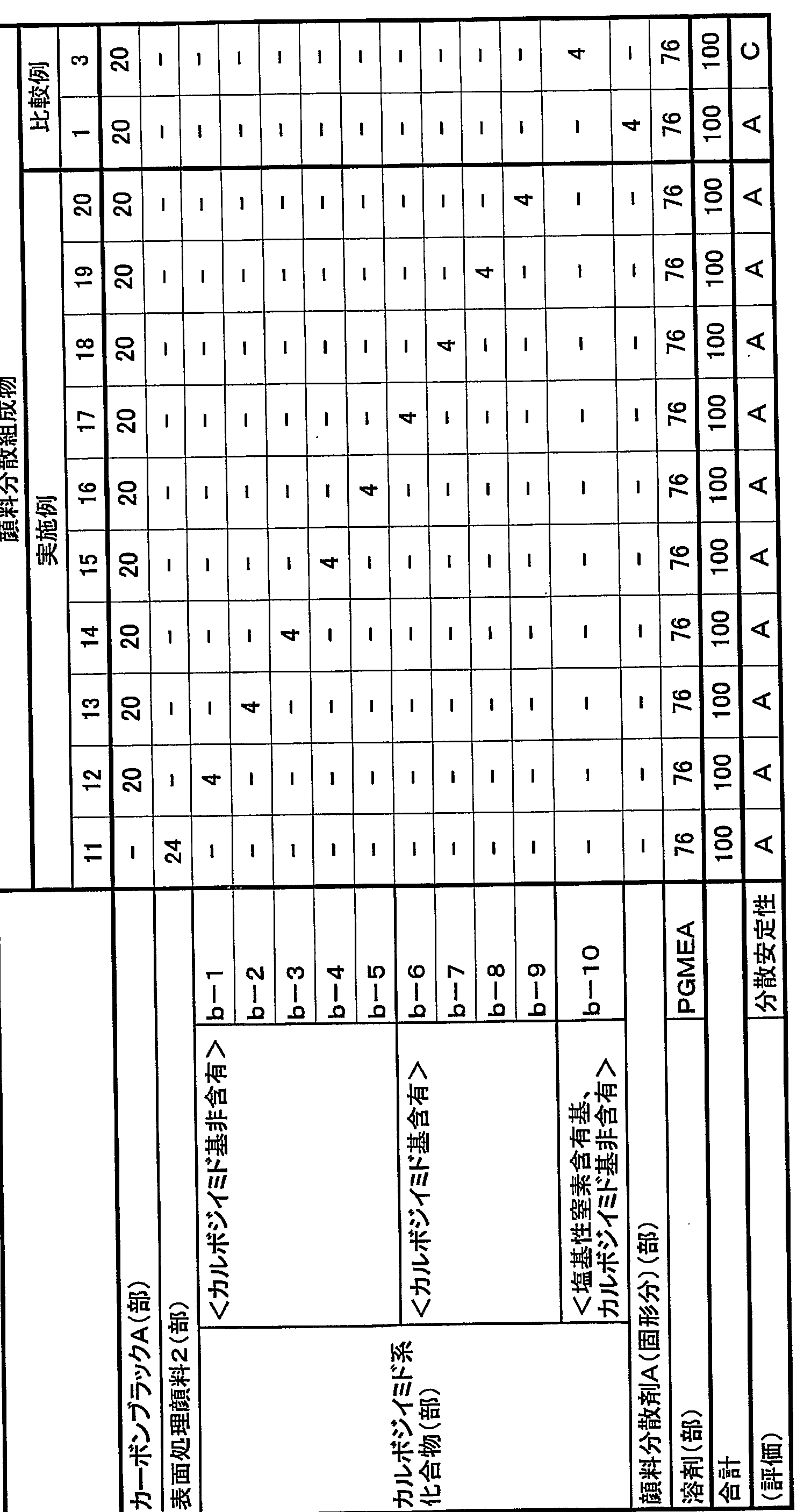
- Example 1 A number average was obtained in the same manner as in Preparation Example a-1, except that the ring-opening polymer of poly (lactic acid) having a molecular weight of 1000 was changed to a ring-opening polymer of poly (lactic acid) having a molecular weight of 1500.
- Compound having a molecular weight of about 550 and a basic nitrogen-containing group content of 0.3600 mmo 1 (Example of preparation a-3)
- Example 1 A number average molecular weight of about 6500 and basic nitrogen content were prepared in the same manner as in Preparation Example a_1, except that the ring-opening polymer of poly-prolatataton having a molecular weight of 1000 was changed to a ring-opening polymer of poly-prolatataton having a molecular weight of 2000. A compound (a-4) having a basis weight of 0.383 mmo1 was obtained.
- Example 1 A number-average molecular weight of about 9100 and basic nitrogen content were prepared in the same manner as in Preparation Example a-1, except that the ring-opening polymer of polyhydraprotone having a molecular weight of 1000 was changed to a ring-opening polymer of polyhydraprotatonone having a molecular weight of 3000. A compound (a-5) having a basis weight of 0.291 92 mmo1 was obtained.
- a four-necked flask equipped with a reflux condenser, a nitrogen gas inlet, a stir bar, and a thermometer was charged with 55 parts of a polycarbodiimide compound having an isocyanate group and a carboimide equivalent of 315, and 8.1 parts of ethylene glycol.
- the mixture was kept at about 100 ° C. for 2 hours to allow the isocyanate group to react with the hydroxyl group, and then 94.5 parts of propylene glycol monomethyl ether acetate was charged. After charging 19.8 parts of the ring-opening polymer of prolactatone, the mixture was kept at about 90 ° C.
- Example Preparation b-1 The same procedure as in Example Preparation b-1 was performed except that the polyethylene oxide polypropylene oxide copolymer having a molecular weight of 1000 was changed to a polyethylene oxide polypropylene oxide copolymer having a molecular weight of 150.
- a compound (b-2) having a number average molecular weight of about 550 and a basic nitrogen-containing group content of 0.3600 mmol was obtained.
- a polycarboimide compound having an isocyanate group and a carbodiimide equivalent of 31.5, 52.5 parts of a methylcarbanolamine, 4.7 parts were charged and kept at about 100 ° C. for 2 hours to allow the isocyanate group to react with the hydroxyl group, and then 99.5 parts of propylene glycol monomethyl ether acetate were charged.
- a number average molecular weight of about 6500 was obtained in the same manner as in Preparation Example b-1, except that the polyethylene oxide-polypropylene oxide copolymer having a molecular weight of 1000 was changed to a polyethylene oxide-polypropylene oxide copolymer having a molecular weight of 2000.
- a compound (b-4) having a basic nitrogen-containing group content of 0.3083 mmol was obtained.
- a number average molecular weight of about 9100 was obtained in the same manner as in Preparation Example b-1, except that the polyethylene oxide-polypropylene oxide copolymer having a molecular weight of 1000 was changed to a polyethylene oxide-polypropylene oxide copolymer having a molecular weight of 3000.
- a compound (b-5) having a basic nitrogen-containing group content of 0.2192 mmol was obtained.
- a four-necked flask equipped with a reflux condenser, a nitrogen gas inlet, a stir bar, and a thermometer was charged with 57 parts of a polycarboimide compound having an isocyanate group equivalent of 315 carbodimid equivalent 315 and 16 parts of methyl jetanolamine.
- the mixture is kept at about 100 ° C for 2 hours to allow the isocyanate group to react with the hydroxyl group.
- 97.7 parts of propylene glycol monomethyl ether acetate is charged, and then a polyethylene oxide having a carboxyl group at the terminal and having a molecular weight of 1000 is prepared.
- a four-necked flask equipped with a reflux condenser, nitrogen gas inlet, stirrer, and thermometer Charge 65 parts of a polycarboimide compound having a cyanoate group-equivalent of 3115, 8.2 parts of methyl jetanolamine, and 9.5 parts of 2-pyridineethanol, and hold at about 100 ° C for 2 hours. Then, 99.7 parts of propylene glycol monomethyl ether acetate is charged to react an isocyanate group and a hydroxyl group, and then a polyethylene oxide compound having a carboxyl group at a terminal having a molecular weight of 2000 and having a molecular weight of 200,000.
- a four-necked flask equipped with a reflux condenser, a nitrogen gas inlet, a stirrer, and a thermometer was charged with 55 parts of a polycarboimide compound having an isocyanate group equivalent of 31.5 and an ethylene glycol of 8.1 parts.
- the mixture was kept at about 100 ° C. for 2 hours to react the isocyanate group with the hydroxyl group, and then charged with 94.5 parts of propylene glycol monomethyl ether acetate.
- a carboxyimide group and a propyloxyl group were allowed to react with each other.
- 94.5 parts of monomethyl ether acetate was charged to obtain a compound (b-10) having a number average molecular weight of about 3700 and a basic nitrogen-containing group content of 0 mmo1.
- a number average molecular weight of about 5500 was obtained in the same manner as in Preparation Example c-11 except that polymethyl methacrylate having a carboxyl group at the terminal and having a molecular weight of 1000 was changed to polymethyl methacrylate having a carboxyl group at the terminal and having a molecular weight of 1500.
- a compound (c-12) having a basic nitrogen-containing group content of 0.360 Ommo 1 was obtained.
- a polycarboleimide compound having an isocyanate group equivalent to 315 is added in a four-necked flask equipped with a reflux condenser, a nitrogen gas inlet, a stirrer, and a thermometer. Charge 6 parts, about 100. C. for 2 hours to allow the isocyanate group to react with the hydroxyl group. Then, 97.7 parts of propylene glycol monomethyl ether acetate were charged, and then a carboxyl group having a molecular weight of 100 After charging 90.5 parts of polymethyl methacrylate and maintaining the temperature at about 90 ° C. to react the carpoimide group with the carboxyl group, 16.7 parts of propylene glycol monomethyl ether acetate was charged. As a result, a compound (c-16) having a number average molecular weight of about 240, a carbodiimide equivalent of 187, and a basic nitrogen-containing group of 0.821.6 mmo 1 was obtained.
- a compound (c-7) having 0, a calposimid equivalent of 3400, and a basic nitrogen-containing group of 0.1141 mmo 1 was obtained.
- a carposimide equivalent having an isocyanate group of 3 15 and a polycarboimide compound of 5 and 5 parts, methinoresiethananolamine 1 0 9 4 parts, isophorone diisocyanate (174.7 parts) were charged and kept at about 100 ° C. for 2 hours to allow the isocyanate group to react with the hydroxyl group, followed by propylene daricol monomethyl ether acetate.
- a four-necked flask equipped with a reflux condenser, a nitrogen gas inlet tube, a stir bar, and a thermometer was charged with 55 parts of a polycarbodiimide compound having a carbodiimide equivalent of 315 cisocyanate groups and 55 parts of ethylene glycol, and 8.1 parts of ethylene glycol.
- the mixture is kept at about 100 for 2 hours to allow the isocyanate group to react with the hydroxyl group, and then 94.5 parts of propylene daricol monomethyl ether acetate is charged, and then a polymethyl having a carboxyl group at a terminal and having a molecular weight of 1000 is prepared.
- This mixture was kneaded with a paint conditioner for 30 minutes to obtain an aqueous pigment dispersion, which was then subjected to a four-necked flask equipped with a reflux condenser, a nitrogen inlet tube, a stirring rod, and a thermometer.
- 50 g of the carbodiimide compound (a-1) and 170 g of propylene glycol monomethyl ether acetate were added with stirring, and reacted at about 90 ° C. for 8 hours.
- Example 1 Dalicol monomethyl ether acetate was removed, and the resultant was placed in a vacuum dryer and dried at 80 ° C. for 2 hours to obtain the surface-treated pigment of Example 1 (surface-treated pigment 1) surface-treated with a carbodiimide compound.
- Table 1 Fee, 1 day and night in a bead mill, and kneaded at a temperature of 60 ° C, to obtain a dispersion composition of the surface treated pigment of Example 1.
- a steel can with a capacity of 100 Om1 filled with 1000 g of stinore beads ( ⁇ 5 ⁇ ) was charged with 50 g of carbon black A (average primary particle diameter 56 nm, pH 3.1) and 450 g of purified water. This mixture was ground for 30 minutes using a paint conditioner to obtain an aqueous pigment dispersion. This aqueous pigment dispersion was transferred to a four-necked flask equipped with a reflux condenser, a nitrogen inlet tube, a stirrer, and a thermometer, and 50 g of the carpoimide compound (b-1), propylene glycol monomethyl ether acetate was stirred with stirring.
- Example 11 surface-treated pigment 2
- the materials having the compositions shown in Table 1 were kneaded with a bead mill for 24 hours at a temperature of 60 ° C. to obtain a dispersion composition of the surface-treated pigment of Example 11.
- Capacity of 1000 g of stinole beads ( ⁇ 5 mm): 50 g of carbon black A (average primary particle diameter 56 nm, pH 3.1) and 450 g of purified water in a stinole can with L 000 m1 I charged. This mixture was kneaded with a paint conditioner for 30 minutes to obtain an aqueous pigment dispersion. The aqueous pigment dispersion was placed in a four-necked flask equipped with a reflux condenser, a nitrogen inlet tube, a stirring rod, and a thermometer.
- the dispersion compositions of the treated carbon blacks of Examples 1 to 30 and Comparative Examples 1 to 4 and other materials were uniformly mixed with a high-speed stirrer so that the compositions of Tables 1 to 6 were obtained.
- the mixture was filtered through a filter of m to obtain pigment-dispersed resist compositions for black matrix of Examples 1 to 30 and Comparative Examples 1 to 4.
- the pigment dispersion resist composition for black matrices for which A or B was evaluated for dispersion stability, was applied on a glass substrate to a thickness of 1 ⁇ using a spin coater, and was heated at 100 ° C. After pre-betaing for 3 minutes, exposure was performed with a high-pressure mercury lamp, and post-beta was further performed at 230 ° C for 30 minutes to obtain a resist pattern formed only of solid portions.
- the optical density (OD value) of the obtained resist pattern in each solid part was measured using a Macbeth densitometer. (TD-931, trade name, manufactured by Macbeth) to evaluate the light-shielding property. Tables 4 to 6 show the evaluation results.
- the surface resistance of the resist pattern obtained by the above method was measured with a resistance meter (R8340 / 8340A S trade name, manufactured by Advantest). Tables 4 to 6 show the evaluation results.
- a pigment dispersion resist composition for a black matrix for which A or B was evaluated for dispersion stability, was applied on a glass substrate to a thickness of 1 / m using a spin coater, and was applied at 100 ° C. Pre-baked for minutes.
- the resulting coating film using a 0. 1 5 ° / 0 Na 2 C0 3 aqueous solution and developed from time to resist composition uncured portion is Ru completely removed, developability according to the following evaluation criteria Was evaluated. Tables 4 to 6 show the evaluation results.
- A It can be completely removed within 30 seconds.
- the pigment dispersant A represents PB821 (trade name, manufactured by Ajinomoto Co.), and PGEA represents propylene glycol monomethyl ether acetate.
- the BMA / MAA copolymer represents a benzyl methacrylate Z methacrylic acid copolymer
- DP EHA represents dipentaerythritol hexane atalylate
- irgacure 907 represents 2-methyl-1 [4- (Methylthio) phenyl] 1-morpholinopropane 11-one (trade name, manufactured by Ciba Specialty Chemicals).
- ⁇ Pigment Organic pigment having a functional group that reacts with carbodiimide group>
- Examples 31 to 34 are pigments surface-treated with the carbodiimide compound (a-1) or (a-6).
- Examples 35 to 46 are surface-treated with the respective carbodiimide compounds while simultaneously dispersing the dispersion medium. It is a dispersion composition of a surface-treated pigment dispersed therein.
- aqueous pigment dispersion In a steel can (100 ml) filled with steel beads (5) (100 g), 50 g of plasma-treated C. I. Pigment Red 122 and purified water (450 g) were placed. Was charged. This mixture was kneaded for 30 minutes using a paint conditioner to obtain an aqueous pigment dispersion. The aqueous pigment dispersion is transferred to a four-necked flask equipped with a reflux condenser, a nitrogen inlet, a stirring bar, and a thermometer, and 50 g of the compound (a-1) or the compound (a-6) is stirred while stirring. 170 g of glycol monomethyl ether acetate was added to the mixture and reacted at about 90 ° C for 8 hours.
- a steel can ( ⁇ 5 ⁇ ) filled with 100 g is filled with 100 g Om 1 and 50 g of plasma-treated C.I. 0 g was charged.
- the mixture was kneaded for 30 minutes using a paint conditioner to obtain an aqueous pigment dispersion.
- This aqueous pigment dispersion was transferred to a four-necked flask equipped with a reflux condenser, a nitrogen inlet, a stirring rod, and a thermometer, and 50 g of the compound (a-1) or the compound (a-6) was mixed with propylene while stirring. 170 g of glycol monomethyl ether acetate was added and reacted at about 90 ° C. for 8 hours.
- the pigment dispersion compositions of Examples 31 to 46 and Comparative Examples 5 and 6 were uniformly mixed with other materials so as to have the composition shown in Table 8, and then a filter having a pore size of 3 / m was used.
- the pigment dispersion resist compositions for color filters of Examples 31 to 46 and Comparative Examples 5 and 6 were obtained.
- Examples 31 to 46, dispersion compositions of the treated organic pigments of Comparative Examples 5 and 6, and Example 3 1 To 46, and the pigment dispersion resist compositions for color filters of Comparative Examples 5 and 6 were each taken in a glass bottle, sealed, and stored at room temperature for 7 days, and the condition was evaluated according to the following evaluation criteria. Tables 7 and 8 show the results.
- the pigment dispersion resist composition for a color filter which was evaluated as A or B in the above dispersion stability, was applied on a glass substrate so as to have a thickness of 1 / m using a spin coater. Pre-beta for minutes. The resulting coating film was developed using a 0.15% Na 2 CO 3 aqueous solution.From the time until the uncured portion of the resist composition was completely removed, the developability was evaluated according to the following evaluation criteria. did. Table 8 shows the results.
- A It can be completely removed within 30 seconds.
- Pigment Dispersant A represents PB 821 (trade name, manufactured by Ajinomoto Co.)
- PGME A represents propylene glycol monomethyl ether acetate
- B MA / MAA copolymer is , Benzyl methacrylate, Z methacrylic acid copolymer
- DPE HA represents dipentaerythritol hexaatalylate
- irgacure 907 represents 2-methyl-1- [4- (methylthio) phenyl] -12-morpholinopro Indicates bread_1_on (product name, Ciba 'Specialty' manufactured by Chemicals).
- Industrial applicability represents PB 821 (trade name, manufactured by Ajinomoto Co.)
- PGME A represents propylene glycol monomethyl ether acetate
- B MA / MAA copolymer is , Benzyl methacrylate, Z methacrylic acid copolymer
- DPE HA represents dipentaery
- a pigment dispersion resist for color filters is used by using an organic pigment (higher-grade organic pigment) having a functional group capable of reacting with a carbodiimide group on the surface of the pigment for adsorption of a basic nitrogen-containing group and, if necessary, a pigment.
- organic pigment higher-grade organic pigment
- a pigment-dispersed resist composition for a color filter having excellent developability, high transmittance, and excellent contrast is used.
- a pigment dispersion resist composition for a black matrix having excellent imageability, high light-shielding properties and high insulation properties can be obtained.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP03733572A EP1535971A4 (en) | 2002-06-25 | 2003-06-25 | TREATED PIGMENT, ITS USE, AND COMPOUND FOR THE TREATMENT OF PIGMENTS |
| JP2004515181A JP4365318B2 (ja) | 2002-06-25 | 2003-06-25 | 処理顔料、その用途 |
| US10/519,079 US7220307B2 (en) | 2002-06-25 | 2003-06-25 | Treated pigment, use thereof, and compound for pigment treatment |
| TW092129501A TW200500420A (en) | 2003-06-25 | 2003-10-24 | Treated pigment, use thereof, and compound for pigment treatment |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002185172 | 2002-06-25 | ||
| JP2002-185172 | 2002-06-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2004000950A1 true WO2004000950A1 (ja) | 2003-12-31 |
Family
ID=29996727
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2003/008019 WO2004000950A1 (ja) | 2002-06-25 | 2003-06-25 | 処理顔料、その用途及び顔料処理用化合物 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US7220307B2 (ja) |
| EP (1) | EP1535971A4 (ja) |
| JP (1) | JP4365318B2 (ja) |
| KR (1) | KR100972318B1 (ja) |
| CN (1) | CN100369980C (ja) |
| WO (1) | WO2004000950A1 (ja) |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006011338A1 (ja) * | 2004-07-06 | 2006-02-02 | Sakata Inx Corp. | 顔料分散組成物、その用途及び顔料処理用化合物 |
| JP2006225431A (ja) * | 2005-02-15 | 2006-08-31 | Sakata Corp | カルボジイミド系化合物及びそれを用いた顔料分散組成物の製造方法 |
| WO2006118201A1 (ja) * | 2005-04-28 | 2006-11-09 | Sakata Inx Corp. | 液体現像剤 |
| WO2009041634A1 (ja) | 2007-09-28 | 2009-04-02 | Sakata Inx Corp. | 液体現像剤の製造方法 |
| JP4977034B2 (ja) * | 2005-11-28 | 2012-07-18 | サカタインクス株式会社 | 液体現像剤 |
| JP5090338B2 (ja) * | 2006-03-22 | 2012-12-05 | サカタインクス株式会社 | 液体現像剤の製造方法及びその方法によって得られる液体現像剤 |
| US8334341B2 (en) | 2005-03-24 | 2012-12-18 | Sakata Inx Corp. | Carbodiimide compound and use thereof |
| US8399170B2 (en) | 2005-06-27 | 2013-03-19 | Sakata Inx Corp. | Process for production of liquid developer, and liquid developer produced by the process |
| WO2014123121A1 (ja) | 2013-02-08 | 2014-08-14 | サカタインクス株式会社 | 液体現像剤 |
| WO2015020128A1 (ja) | 2013-08-07 | 2015-02-12 | サカタインクス株式会社 | 非水性インクジェット用インク組成物及びそれを用いて得られる印刷物 |
| WO2015119145A1 (ja) * | 2014-02-04 | 2015-08-13 | サカタインクス株式会社 | 液体現像剤 |
| WO2018030088A1 (ja) | 2016-08-10 | 2018-02-15 | サカタインクス株式会社 | 非水性インク組成物 |
| WO2018105696A1 (ja) | 2016-12-09 | 2018-06-14 | サカタインクス株式会社 | 非水性インクジェット用インク組成物 |
| JPWO2017134858A1 (ja) * | 2016-02-03 | 2018-12-06 | 富士フイルム株式会社 | 分散組成物、硬化性組成物、遮光膜、カラーフィルタ、固体撮像装置 |
| KR20190049504A (ko) | 2017-10-30 | 2019-05-09 | 사카타 인쿠스 가부시키가이샤 | 흑색 착색 조성물 및 이를 함유하는 흑색 착색 레지스트 조성물 |
| US10683428B2 (en) | 2016-06-29 | 2020-06-16 | Sakata Inx Corporation | Nonaqueous ink-jet magenta ink composition |
| US10767067B2 (en) | 2016-04-28 | 2020-09-08 | Sakata Inx Corporation | Non-aqueous inkjet ink composition |
| WO2021085511A1 (ja) | 2019-10-30 | 2021-05-06 | サカタインクス株式会社 | 非水性インクジェット用インク組成物 |
| WO2021085510A1 (ja) | 2019-10-30 | 2021-05-06 | サカタインクス株式会社 | 非水性インクジェット用インク組成物 |
| WO2021199760A1 (ja) | 2020-03-31 | 2021-10-07 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
| WO2021215341A1 (ja) | 2020-04-24 | 2021-10-28 | サカタインクス株式会社 | 光硬化型水性インクジェット印刷用インク組成物 |
| WO2021261208A1 (ja) | 2020-06-24 | 2021-12-30 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
| WO2022024705A1 (ja) | 2020-07-29 | 2022-02-03 | サカタインクス株式会社 | 活性エネルギー線硬化型インクジェット印刷用インク組成物 |
| WO2022024704A1 (ja) | 2020-07-29 | 2022-02-03 | サカタインクス株式会社 | 活性エネルギー線硬化型インクジェット印刷用インク組成物 |
| WO2022024706A1 (ja) | 2020-07-29 | 2022-02-03 | サカタインクス株式会社 | 活性エネルギー線硬化型インクジェット印刷用インク組成物 |
| WO2022030261A1 (ja) | 2020-08-07 | 2022-02-10 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
| WO2022085246A1 (ja) | 2020-10-23 | 2022-04-28 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5280846B2 (ja) * | 2005-07-22 | 2013-09-04 | キャボット コーポレイション | 顔料組成物の製造法 |
| ES2563310T3 (es) * | 2006-05-31 | 2016-03-14 | The Sherwin-Williams Company | Polímeros dispersantes |
| US9381481B2 (en) * | 2006-09-27 | 2016-07-05 | Basf Se | Polyethylene imine based dendritic dispersant |
| JP2010516860A (ja) * | 2007-01-24 | 2010-05-20 | キャボット コーポレイション | 改質顔料を生成するための方法 |
| US20080249217A1 (en) * | 2007-02-27 | 2008-10-09 | Sze-Ming Lee | Inkjet ink compositions comprising multiple modified pigments |
| CN101688071B (zh) * | 2007-04-24 | 2014-02-12 | 卡伯特公司 | 低结构炭黑及其制造方法 |
| EP2048539A1 (en) * | 2007-09-06 | 2009-04-15 | FUJIFILM Corporation | Processed pigment, pigment-dispersed composition, colored photosensitive composition, color filter, liquid crystal display element, and solid image pickup element |
| EP2231784A2 (en) * | 2007-12-07 | 2010-09-29 | Cabot Corporation | Method of preparing coated pigments |
| US8129448B2 (en) * | 2008-12-18 | 2012-03-06 | Cabot Corporation | Method of preparing polymer modified pigments |
| CN105216460B (zh) * | 2015-10-30 | 2018-05-01 | 河南卓立膜材料股份有限公司 | 一种水性油墨热转印色带及其制备方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002069329A (ja) * | 2000-08-29 | 2002-03-08 | Catalysts & Chem Ind Co Ltd | 樹脂被覆鱗片状無機粒子およびこれを配合した化粧料 |
| JP2002097390A (ja) * | 2000-09-22 | 2002-04-02 | Mitsubishi Chemicals Corp | インクジェット記録液およびインクジェット記録方法 |
| JP2002201381A (ja) * | 2000-12-27 | 2002-07-19 | Sakata Corp | 表面処理したカーボンブラック及びそれを含有してなるブラックマトリックス用レジスト組成物 |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4049610A (en) * | 1974-01-22 | 1977-09-20 | Bayer Aktiengesellschaft | Pigment preparations |
| NZ187714A (en) | 1977-07-15 | 1980-09-12 | Ici Ltd | Dispersing agent reaction product of a polyalkylene imine and a polyester |
| DE4039193A1 (de) * | 1990-12-08 | 1992-06-11 | Bayer Ag | In wasser dispergierbare, elektrolytstabile polyetherester-modifizierte polyurethanionomere |
| US5371148A (en) * | 1993-06-23 | 1994-12-06 | Union Carbide Chemicals & Plastics Technology Corporation | Reactive polymers having pendant flexible side chains prepared from ethylenically unsaturated carbodiimides |
| JP3235930B2 (ja) | 1994-01-10 | 2001-12-04 | ダイセル化学工業株式会社 | 顔料分散剤の製造方法 |
| US5602264A (en) * | 1994-11-28 | 1997-02-11 | The Regents Of The University Of California | Highly reactive, water soluble carbodiimides, intermediates and derivatives thereof |
| JP3856402B2 (ja) | 1995-10-13 | 2006-12-13 | 東京応化工業株式会社 | 遮光膜形成用感光性樹脂組成物、該遮光膜形成用感光性樹脂組成物で形成されたブラックマトリックス、及びその製造方法 |
| JP3718915B2 (ja) | 1995-10-16 | 2005-11-24 | 味の素株式会社 | 顔料分散剤 |
| US6395824B1 (en) * | 1997-07-31 | 2002-05-28 | Basf Aktiengesellschaft | Aqueous dispersions containing polyurethanes with carbodiimide groups |
| DE19954500A1 (de) * | 1999-11-11 | 2001-05-17 | Basf Ag | Carbodiimide mit Carboxyl- oder Caboxylatgruppen |
| DE10000656A1 (de) * | 2000-01-11 | 2001-07-12 | Basf Ag | Carbodiimide mit Carboxyl- oder Carboxylatgruppen |
| JP2003039824A (ja) * | 2001-05-22 | 2003-02-13 | Fuji Photo Film Co Ltd | インクジェット記録用シート |
| JP4247124B2 (ja) | 2002-03-08 | 2009-04-02 | サカタインクス株式会社 | 処理顔料、その用途及び顔料処理用化合物 |
| JP4117140B2 (ja) * | 2002-03-13 | 2008-07-16 | 日清紡績株式会社 | カルボジイミド含有硬化型反応性粒子、その製造方法及び用途 |
| US8440779B2 (en) * | 2004-11-04 | 2013-05-14 | 3M Innovative Properties Company | Carbodiimide compound and compositions for rendering substrates oil and water repellent |
-
2003
- 2003-06-25 JP JP2004515181A patent/JP4365318B2/ja not_active Expired - Fee Related
- 2003-06-25 WO PCT/JP2003/008019 patent/WO2004000950A1/ja active Application Filing
- 2003-06-25 KR KR1020047021091A patent/KR100972318B1/ko active IP Right Grant
- 2003-06-25 EP EP03733572A patent/EP1535971A4/en not_active Withdrawn
- 2003-06-25 US US10/519,079 patent/US7220307B2/en not_active Expired - Lifetime
- 2003-06-25 CN CNB038148366A patent/CN100369980C/zh not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2002069329A (ja) * | 2000-08-29 | 2002-03-08 | Catalysts & Chem Ind Co Ltd | 樹脂被覆鱗片状無機粒子およびこれを配合した化粧料 |
| JP2002097390A (ja) * | 2000-09-22 | 2002-04-02 | Mitsubishi Chemicals Corp | インクジェット記録液およびインクジェット記録方法 |
| JP2002201381A (ja) * | 2000-12-27 | 2002-07-19 | Sakata Corp | 表面処理したカーボンブラック及びそれを含有してなるブラックマトリックス用レジスト組成物 |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP1535971A4 * |
Cited By (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006011338A1 (ja) * | 2004-07-06 | 2006-02-02 | Sakata Inx Corp. | 顔料分散組成物、その用途及び顔料処理用化合物 |
| JP2006225431A (ja) * | 2005-02-15 | 2006-08-31 | Sakata Corp | カルボジイミド系化合物及びそれを用いた顔料分散組成物の製造方法 |
| US8334341B2 (en) | 2005-03-24 | 2012-12-18 | Sakata Inx Corp. | Carbodiimide compound and use thereof |
| AU2006241795B2 (en) * | 2005-04-28 | 2011-10-13 | Sakata Inx Corp. | Liquid developer |
| WO2006118201A1 (ja) * | 2005-04-28 | 2006-11-09 | Sakata Inx Corp. | 液体現像剤 |
| JP4977016B2 (ja) * | 2005-04-28 | 2012-07-18 | サカタインクス株式会社 | 液体現像剤 |
| US8399170B2 (en) | 2005-06-27 | 2013-03-19 | Sakata Inx Corp. | Process for production of liquid developer, and liquid developer produced by the process |
| JP4977034B2 (ja) * | 2005-11-28 | 2012-07-18 | サカタインクス株式会社 | 液体現像剤 |
| JP5090338B2 (ja) * | 2006-03-22 | 2012-12-05 | サカタインクス株式会社 | 液体現像剤の製造方法及びその方法によって得られる液体現像剤 |
| WO2009041634A1 (ja) | 2007-09-28 | 2009-04-02 | Sakata Inx Corp. | 液体現像剤の製造方法 |
| WO2014123121A1 (ja) | 2013-02-08 | 2014-08-14 | サカタインクス株式会社 | 液体現像剤 |
| WO2015020128A1 (ja) | 2013-08-07 | 2015-02-12 | サカタインクス株式会社 | 非水性インクジェット用インク組成物及びそれを用いて得られる印刷物 |
| WO2015119145A1 (ja) * | 2014-02-04 | 2015-08-13 | サカタインクス株式会社 | 液体現像剤 |
| JPWO2015119145A1 (ja) * | 2014-02-04 | 2017-03-23 | サカタインクス株式会社 | 液体現像剤 |
| US11091594B2 (en) | 2016-02-03 | 2021-08-17 | Fujifilm Corporation | Dispersion composition, curable composition, light-shielding film, color filter, and solid-state imaging device |
| JPWO2017134858A1 (ja) * | 2016-02-03 | 2018-12-06 | 富士フイルム株式会社 | 分散組成物、硬化性組成物、遮光膜、カラーフィルタ、固体撮像装置 |
| US10767067B2 (en) | 2016-04-28 | 2020-09-08 | Sakata Inx Corporation | Non-aqueous inkjet ink composition |
| US10683428B2 (en) | 2016-06-29 | 2020-06-16 | Sakata Inx Corporation | Nonaqueous ink-jet magenta ink composition |
| WO2018030088A1 (ja) | 2016-08-10 | 2018-02-15 | サカタインクス株式会社 | 非水性インク組成物 |
| WO2018105696A1 (ja) | 2016-12-09 | 2018-06-14 | サカタインクス株式会社 | 非水性インクジェット用インク組成物 |
| KR20190049504A (ko) | 2017-10-30 | 2019-05-09 | 사카타 인쿠스 가부시키가이샤 | 흑색 착색 조성물 및 이를 함유하는 흑색 착색 레지스트 조성물 |
| WO2021085511A1 (ja) | 2019-10-30 | 2021-05-06 | サカタインクス株式会社 | 非水性インクジェット用インク組成物 |
| WO2021085510A1 (ja) | 2019-10-30 | 2021-05-06 | サカタインクス株式会社 | 非水性インクジェット用インク組成物 |
| WO2021199760A1 (ja) | 2020-03-31 | 2021-10-07 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
| WO2021215341A1 (ja) | 2020-04-24 | 2021-10-28 | サカタインクス株式会社 | 光硬化型水性インクジェット印刷用インク組成物 |
| WO2021261208A1 (ja) | 2020-06-24 | 2021-12-30 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
| WO2022024705A1 (ja) | 2020-07-29 | 2022-02-03 | サカタインクス株式会社 | 活性エネルギー線硬化型インクジェット印刷用インク組成物 |
| WO2022024704A1 (ja) | 2020-07-29 | 2022-02-03 | サカタインクス株式会社 | 活性エネルギー線硬化型インクジェット印刷用インク組成物 |
| WO2022024706A1 (ja) | 2020-07-29 | 2022-02-03 | サカタインクス株式会社 | 活性エネルギー線硬化型インクジェット印刷用インク組成物 |
| WO2022030261A1 (ja) | 2020-08-07 | 2022-02-10 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
| WO2022085246A1 (ja) | 2020-10-23 | 2022-04-28 | サカタインクス株式会社 | 光硬化型インクジェット印刷用インク組成物 |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4365318B2 (ja) | 2009-11-18 |
| US7220307B2 (en) | 2007-05-22 |
| KR100972318B1 (ko) | 2010-07-26 |
| US20050256247A1 (en) | 2005-11-17 |
| EP1535971A1 (en) | 2005-06-01 |
| CN100369980C (zh) | 2008-02-20 |
| CN1662614A (zh) | 2005-08-31 |
| EP1535971A4 (en) | 2010-01-20 |
| KR20050026931A (ko) | 2005-03-16 |
| JPWO2004000950A1 (ja) | 2005-10-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2004000950A1 (ja) | 処理顔料、その用途及び顔料処理用化合物 | |
| JP4247124B2 (ja) | 処理顔料、その用途及び顔料処理用化合物 | |
| JP6005620B2 (ja) | 低粘度、粒子状物質高含有分散体 | |
| TWI398492B (zh) | A pigment dispersion composition, a use thereof, and a pigment treatment compound | |
| JP4114793B2 (ja) | 処理有機顔料、その用途及び顔料処理用化合物 | |
| JP4402590B2 (ja) | 顔料分散組成物、その用途 | |
| JP2006273974A (ja) | 酸性アクリルブロック樹脂 | |
| JP4559209B2 (ja) | カルボジイミド系化合物、顔料分散組成物及びその用途 | |
| JP2006225431A (ja) | カルボジイミド系化合物及びそれを用いた顔料分散組成物の製造方法 | |
| JP4114797B2 (ja) | 処理チタンブラック、処理チタンブラックの分散体及びチタンブラック処理用化合物 | |
| JP7025939B2 (ja) | カラーフィルター用顔料分散組成物及びカラーフィルター用レジスト組成物 | |
| JP2006176657A (ja) | カルボジイミド系化合物及びその用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): CN JP KR US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2004515181 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020047021091 Country of ref document: KR Ref document number: 20038148366 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003733572 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020047021091 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003733572 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10519079 Country of ref document: US |