KR20140084201A - 알칼리성 포스파타제 및/또는 나트륨이뇨 펩티드를 포함하는 조성물 및 그의 사용 방법 - Google Patents
알칼리성 포스파타제 및/또는 나트륨이뇨 펩티드를 포함하는 조성물 및 그의 사용 방법 Download PDFInfo
- Publication number
- KR20140084201A KR20140084201A KR1020147013214A KR20147013214A KR20140084201A KR 20140084201 A KR20140084201 A KR 20140084201A KR 1020147013214 A KR1020147013214 A KR 1020147013214A KR 20147013214 A KR20147013214 A KR 20147013214A KR 20140084201 A KR20140084201 A KR 20140084201A
- Authority
- KR
- South Korea
- Prior art keywords
- amino acid
- seq
- acid sequence
- polypeptide
- sequence
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 102000002260 Alkaline Phosphatase Human genes 0.000 title claims abstract description 48
- 108020004774 Alkaline Phosphatase Proteins 0.000 title claims abstract description 48
- 238000000034 method Methods 0.000 title claims description 149
- 239000000203 mixture Substances 0.000 title claims description 73
- 108020001621 Natriuretic Peptide Proteins 0.000 title description 3
- 102000004571 Natriuretic peptide Human genes 0.000 title description 3
- 239000000692 natriuretic peptide Substances 0.000 title description 3
- 108090000765 processed proteins & peptides Proteins 0.000 claims abstract description 415
- 102000004196 processed proteins & peptides Human genes 0.000 claims abstract description 394
- 229920001184 polypeptide Polymers 0.000 claims abstract description 387
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 128
- 208000011580 syndromic disease Diseases 0.000 claims abstract description 91
- 208000035475 disorder Diseases 0.000 claims abstract description 79
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 claims abstract description 70
- 229910052708 sodium Inorganic materials 0.000 claims abstract description 70
- 239000011734 sodium Substances 0.000 claims abstract description 70
- 210000000988 bone and bone Anatomy 0.000 claims abstract description 66
- 101710158332 Diuretic hormone Proteins 0.000 claims abstract description 59
- 101710204261 Diuretic hormone class 2 Proteins 0.000 claims abstract description 59
- 102100027842 Fibroblast growth factor receptor 3 Human genes 0.000 claims abstract description 37
- 101710182396 Fibroblast growth factor receptor 3 Proteins 0.000 claims abstract description 35
- 208000003019 Neurofibromatosis 1 Diseases 0.000 claims abstract description 35
- 208000015100 cartilage disease Diseases 0.000 claims abstract description 34
- 208000024834 Neurofibromatosis type 1 Diseases 0.000 claims abstract description 30
- 208000020084 Bone disease Diseases 0.000 claims abstract description 29
- 210000002464 muscle smooth vascular Anatomy 0.000 claims abstract description 20
- 208000029578 Muscle disease Diseases 0.000 claims abstract description 18
- 208000021642 Muscular disease Diseases 0.000 claims abstract description 18
- 208000013403 hyperactivity Diseases 0.000 claims abstract description 16
- 108060003951 Immunoglobulin Proteins 0.000 claims abstract description 13
- 102000018358 immunoglobulin Human genes 0.000 claims abstract description 13
- 230000002232 neuromuscular Effects 0.000 claims abstract description 10
- 125000003275 alpha amino acid group Chemical group 0.000 claims description 413
- 125000000539 amino acid group Chemical group 0.000 claims description 384
- 229940024606 amino acid Drugs 0.000 claims description 233
- 235000001014 amino acid Nutrition 0.000 claims description 223
- 150000001413 amino acids Chemical class 0.000 claims description 180
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 103
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 claims description 67
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 claims description 60
- 201000010099 disease Diseases 0.000 claims description 49
- 241000282414 Homo sapiens Species 0.000 claims description 44
- 239000008194 pharmaceutical composition Substances 0.000 claims description 44
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 claims description 38
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 claims description 38
- 210000004899 c-terminal region Anatomy 0.000 claims description 38
- 239000012634 fragment Substances 0.000 claims description 38
- 238000003776 cleavage reaction Methods 0.000 claims description 34
- 230000007017 scission Effects 0.000 claims description 34
- 229910052720 vanadium Inorganic materials 0.000 claims description 32
- 102000014187 peptide receptors Human genes 0.000 claims description 26
- 239000000556 agonist Substances 0.000 claims description 25
- 235000003704 aspartic acid Nutrition 0.000 claims description 24
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 claims description 24
- 102000005600 Cathepsins Human genes 0.000 claims description 23
- 108010084457 Cathepsins Proteins 0.000 claims description 23
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 claims description 20
- 239000004471 Glycine Substances 0.000 claims description 19
- 235000013922 glutamic acid Nutrition 0.000 claims description 18
- 239000004220 glutamic acid Substances 0.000 claims description 18
- KJBQDYDTQCIAFO-ZLELNMGESA-N (2s)-2-amino-4-methylpentanoic acid;(2s)-2,6-diaminohexanoic acid Chemical compound CC(C)C[C@H](N)C(O)=O.NCCCC[C@H](N)C(O)=O KJBQDYDTQCIAFO-ZLELNMGESA-N 0.000 claims description 17
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 15
- 101710102159 Atrial natriuretic peptide receptor 2 Proteins 0.000 claims description 13
- 102100039341 Atrial natriuretic peptide receptor 2 Human genes 0.000 claims description 13
- 230000002378 acidificating effect Effects 0.000 claims description 13
- 150000001875 compounds Chemical class 0.000 claims description 10
- 239000000539 dimer Substances 0.000 claims description 10
- 108090000625 Cathepsin K Proteins 0.000 claims description 9
- 102000004171 Cathepsin K Human genes 0.000 claims description 9
- SEFLNGWGXSFZMH-DQZFIDPSSA-N (2s)-2-aminobutanedioic acid;(2s,3s)-2-amino-3-methylpentanoic acid Chemical compound CC[C@H](C)[C@H](N)C(O)=O.OC(=O)[C@@H](N)CC(O)=O SEFLNGWGXSFZMH-DQZFIDPSSA-N 0.000 claims description 8
- 230000000926 neurological effect Effects 0.000 claims description 8
- 230000008685 targeting Effects 0.000 claims description 8
- 230000001537 neural effect Effects 0.000 claims description 6
- 229920000805 Polyaspartic acid Polymers 0.000 claims description 5
- 125000000291 glutamic acid group Chemical group N[C@@H](CCC(O)=O)C(=O)* 0.000 claims description 5
- 108010064470 polyaspartate Proteins 0.000 claims description 5
- 108010020346 Polyglutamic Acid Proteins 0.000 claims description 4
- 229920002643 polyglutamic acid Polymers 0.000 claims description 4
- 230000029087 digestion Effects 0.000 claims description 3
- 150000007523 nucleic acids Chemical class 0.000 abstract description 50
- 102000039446 nucleic acids Human genes 0.000 abstract description 39
- 108020004707 nucleic acids Proteins 0.000 abstract description 38
- 238000011282 treatment Methods 0.000 abstract description 30
- 210000000845 cartilage Anatomy 0.000 abstract description 25
- 230000011164 ossification Effects 0.000 abstract description 4
- 108010076504 Protein Sorting Signals Proteins 0.000 description 288
- 125000005647 linker group Chemical group 0.000 description 99
- 101800002899 Soluble alkaline phosphatase Proteins 0.000 description 96
- 101000574445 Homo sapiens Alkaline phosphatase, tissue-nonspecific isozyme Proteins 0.000 description 75
- 102100025683 Alkaline phosphatase, tissue-nonspecific isozyme Human genes 0.000 description 73
- 102000012421 C-Type Natriuretic Peptide Human genes 0.000 description 60
- 101800000060 C-type natriuretic peptide Proteins 0.000 description 60
- 206010058314 Dysplasia Diseases 0.000 description 57
- 230000035772 mutation Effects 0.000 description 55
- 230000004927 fusion Effects 0.000 description 51
- 230000000694 effects Effects 0.000 description 39
- 241000699670 Mus sp. Species 0.000 description 37
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 33
- -1 Phe Chemical compound 0.000 description 33
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 32
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 30
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 28
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 28
- 230000002354 daily effect Effects 0.000 description 28
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 description 28
- 125000002987 valine group Chemical group [H]N([H])C([H])(C(*)=O)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 28
- 108010085793 Neurofibromin 1 Proteins 0.000 description 27
- 210000004027 cell Anatomy 0.000 description 27
- 125000001909 leucine group Chemical group [H]N(*)C(C(*)=O)C([H])([H])C(C([H])([H])[H])C([H])([H])[H] 0.000 description 27
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 26
- 108010041801 2',3'-Cyclic Nucleotide 3'-Phosphodiesterase Proteins 0.000 description 25
- 102100040458 2',3'-cyclic-nucleotide 3'-phosphodiesterase Human genes 0.000 description 25
- 208000009905 Neurofibromatoses Diseases 0.000 description 25
- 102100026379 Neurofibromin Human genes 0.000 description 25
- 201000004931 neurofibromatosis Diseases 0.000 description 25
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 description 24
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 23
- 108091035707 Consensus sequence Proteins 0.000 description 22
- 102000004190 Enzymes Human genes 0.000 description 22
- 108090000790 Enzymes Proteins 0.000 description 22
- 229940088598 enzyme Drugs 0.000 description 22
- 230000033558 biomineral tissue development Effects 0.000 description 21
- 230000001717 pathogenic effect Effects 0.000 description 21
- 206010072610 Skeletal dysplasia Diseases 0.000 description 20
- 101100328886 Caenorhabditis elegans col-2 gene Proteins 0.000 description 19
- 125000000174 L-prolyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])([H])[C@@]1([H])C(*)=O 0.000 description 19
- 108020001507 fusion proteins Proteins 0.000 description 19
- 102000037865 fusion proteins Human genes 0.000 description 19
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Chemical compound CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 18
- 125000000637 arginyl group Chemical group N[C@@H](CCCNC(N)=N)C(=O)* 0.000 description 18
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 18
- 239000011780 sodium chloride Substances 0.000 description 18
- ODKSFYDXXFIFQN-BYPYZUCNSA-N L-arginine Chemical compound OC(=O)[C@@H](N)CCCN=C(N)N ODKSFYDXXFIFQN-BYPYZUCNSA-N 0.000 description 17
- 125000003607 serino group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C(O[H])([H])[H] 0.000 description 17
- 108090000623 proteins and genes Proteins 0.000 description 16
- 230000001965 increasing effect Effects 0.000 description 15
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 14
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 14
- 238000001727 in vivo Methods 0.000 description 14
- 201000008482 osteoarthritis Diseases 0.000 description 14
- 206010061762 Chondropathy Diseases 0.000 description 13
- 208000011231 Crohn disease Diseases 0.000 description 13
- 102000018071 Immunoglobulin Fc Fragments Human genes 0.000 description 13
- 108010091135 Immunoglobulin Fc Fragments Proteins 0.000 description 13
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 13
- 108091028043 Nucleic acid sequence Proteins 0.000 description 13
- 235000018102 proteins Nutrition 0.000 description 13
- 102000004169 proteins and genes Human genes 0.000 description 13
- 238000006467 substitution reaction Methods 0.000 description 13
- 208000024891 symptom Diseases 0.000 description 13
- 102100039339 Atrial natriuretic peptide receptor 1 Human genes 0.000 description 12
- 230000006870 function Effects 0.000 description 12
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 description 12
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 12
- 210000003491 skin Anatomy 0.000 description 12
- 230000001225 therapeutic effect Effects 0.000 description 12
- 101710102163 Atrial natriuretic peptide receptor 1 Proteins 0.000 description 11
- 101001128963 Homo sapiens Protein Dr1 Proteins 0.000 description 11
- 241000699666 Mus <mouse, genus> Species 0.000 description 11
- 102100031227 Protein Dr1 Human genes 0.000 description 11
- 229910052799 carbon Inorganic materials 0.000 description 11
- 230000015556 catabolic process Effects 0.000 description 11
- 238000006731 degradation reaction Methods 0.000 description 11
- 239000001509 sodium citrate Substances 0.000 description 11
- HRXKRNGNAMMEHJ-UHFFFAOYSA-K trisodium citrate Chemical compound [Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O HRXKRNGNAMMEHJ-UHFFFAOYSA-K 0.000 description 11
- 229940038773 trisodium citrate Drugs 0.000 description 11
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 10
- 206010049933 Hypophosphatasia Diseases 0.000 description 10
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 10
- 208000020221 Short stature Diseases 0.000 description 10
- 238000007792 addition Methods 0.000 description 10
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 10
- 235000018417 cysteine Nutrition 0.000 description 10
- 125000000487 histidyl group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C([H])=N1 0.000 description 10
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 10
- 239000012528 membrane Substances 0.000 description 10
- 230000002829 reductive effect Effects 0.000 description 10
- 239000003981 vehicle Substances 0.000 description 10
- 125000000510 L-tryptophano group Chemical group [H]C1=C([H])C([H])=C2N([H])C([H])=C(C([H])([H])[C@@]([H])(C(O[H])=O)N([H])[*])C2=C1[H] 0.000 description 9
- 206010028980 Neoplasm Diseases 0.000 description 9
- 206010029748 Noonan syndrome Diseases 0.000 description 9
- 239000003795 chemical substances by application Substances 0.000 description 9
- 238000012217 deletion Methods 0.000 description 9
- 230000037430 deletion Effects 0.000 description 9
- 230000012010 growth Effects 0.000 description 9
- 238000009396 hybridization Methods 0.000 description 9
- 230000037417 hyperactivation Effects 0.000 description 9
- 230000009984 peri-natal effect Effects 0.000 description 9
- NGVDGCNFYWLIFO-UHFFFAOYSA-N pyridoxal 5'-phosphate Chemical compound CC1=NC=C(COP(O)(O)=O)C(C=O)=C1O NGVDGCNFYWLIFO-UHFFFAOYSA-N 0.000 description 9
- 235000007682 pyridoxal 5'-phosphate Nutrition 0.000 description 9
- 239000011589 pyridoxal 5'-phosphate Substances 0.000 description 9
- 208000007442 rickets Diseases 0.000 description 9
- 230000028327 secretion Effects 0.000 description 9
- 230000003442 weekly effect Effects 0.000 description 9
- 206010010356 Congenital anomaly Diseases 0.000 description 8
- 102000043136 MAP kinase family Human genes 0.000 description 8
- 108091054455 MAP kinase family Proteins 0.000 description 8
- 101100180983 Mus musculus Khdrbs3 gene Proteins 0.000 description 8
- 102000003729 Neprilysin Human genes 0.000 description 8
- 108090000028 Neprilysin Proteins 0.000 description 8
- 208000018737 Parkinson disease Diseases 0.000 description 8
- 241001415513 Salpida Species 0.000 description 8
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 8
- 235000004279 alanine Nutrition 0.000 description 8
- 230000018678 bone mineralization Effects 0.000 description 8
- 239000013078 crystal Substances 0.000 description 8
- 230000007812 deficiency Effects 0.000 description 8
- 231100000673 dose–response relationship Toxicity 0.000 description 8
- 230000014509 gene expression Effects 0.000 description 8
- WHUUTDBJXJRKMK-VKHMYHEASA-L glutamate group Chemical group N[C@@H](CCC(=O)[O-])C(=O)[O-] WHUUTDBJXJRKMK-VKHMYHEASA-L 0.000 description 8
- 238000002887 multiple sequence alignment Methods 0.000 description 8
- 230000037361 pathway Effects 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- 238000005406 washing Methods 0.000 description 8
- 229910052727 yttrium Inorganic materials 0.000 description 8
- 206010002961 Aplasia Diseases 0.000 description 7
- 108010044467 Isoenzymes Proteins 0.000 description 7
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 7
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 7
- SUHOOTKUPISOBE-UHFFFAOYSA-N O-phosphoethanolamine Chemical compound NCCOP(O)(O)=O SUHOOTKUPISOBE-UHFFFAOYSA-N 0.000 description 7
- 230000004913 activation Effects 0.000 description 7
- 238000003556 assay Methods 0.000 description 7
- 230000008901 benefit Effects 0.000 description 7
- 210000004271 bone marrow stromal cell Anatomy 0.000 description 7
- 210000003414 extremity Anatomy 0.000 description 7
- 229910052739 hydrogen Inorganic materials 0.000 description 7
- 125000000741 isoleucyl group Chemical group [H]N([H])C(C(C([H])([H])[H])C([H])([H])C([H])([H])[H])C(=O)O* 0.000 description 7
- 125000003729 nucleotide group Chemical group 0.000 description 7
- 230000001575 pathological effect Effects 0.000 description 7
- 150000003839 salts Chemical class 0.000 description 7
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 6
- 206010003591 Ataxia Diseases 0.000 description 6
- 241000283690 Bos taurus Species 0.000 description 6
- 241000282472 Canis lupus familiaris Species 0.000 description 6
- 206010010904 Convulsion Diseases 0.000 description 6
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 6
- 102100024193 Mitogen-activated protein kinase 1 Human genes 0.000 description 6
- 206010034010 Parkinsonism Diseases 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 241000700159 Rattus Species 0.000 description 6
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 6
- 239000004473 Threonine Substances 0.000 description 6
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 6
- 230000002411 adverse Effects 0.000 description 6
- 201000007201 aphasia Diseases 0.000 description 6
- 210000004369 blood Anatomy 0.000 description 6
- 239000008280 blood Substances 0.000 description 6
- 201000011510 cancer Diseases 0.000 description 6
- 210000001612 chondrocyte Anatomy 0.000 description 6
- 230000007547 defect Effects 0.000 description 6
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 description 6
- 235000011180 diphosphates Nutrition 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- 231100000518 lethal Toxicity 0.000 description 6
- 230000001665 lethal effect Effects 0.000 description 6
- 210000000963 osteoblast Anatomy 0.000 description 6
- 229910052698 phosphorus Inorganic materials 0.000 description 6
- 239000003826 tablet Substances 0.000 description 6
- 238000002560 therapeutic procedure Methods 0.000 description 6
- 239000004474 valine Substances 0.000 description 6
- 239000013598 vector Substances 0.000 description 6
- ZOOGRGPOEVQQDX-UUOKFMHZSA-N 3',5'-cyclic GMP Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=C(NC2=O)N)=C2N=C1 ZOOGRGPOEVQQDX-UUOKFMHZSA-N 0.000 description 5
- 201000010028 Acrocephalosyndactylia Diseases 0.000 description 5
- 108020004414 DNA Proteins 0.000 description 5
- 108010016626 Dipeptides Proteins 0.000 description 5
- 206010013883 Dwarfism Diseases 0.000 description 5
- 208000029663 Hypophosphatemia Diseases 0.000 description 5
- 101100084030 Mus musculus Alpl gene Proteins 0.000 description 5
- 230000036772 blood pressure Effects 0.000 description 5
- 201000005043 chondromalacia Diseases 0.000 description 5
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 5
- 230000008021 deposition Effects 0.000 description 5
- 239000003112 inhibitor Substances 0.000 description 5
- 230000003902 lesion Effects 0.000 description 5
- 230000004048 modification Effects 0.000 description 5
- 238000012986 modification Methods 0.000 description 5
- 239000002773 nucleotide Substances 0.000 description 5
- 102000016914 ras Proteins Human genes 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 201000000980 schizophrenia Diseases 0.000 description 5
- 238000004904 shortening Methods 0.000 description 5
- 241000894007 species Species 0.000 description 5
- 230000002195 synergetic effect Effects 0.000 description 5
- 210000002303 tibia Anatomy 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 5
- 210000000689 upper leg Anatomy 0.000 description 5
- 230000002485 urinary effect Effects 0.000 description 5
- WMSPZRZYIGOEDS-QMMMGPOBSA-N 4-[[(1s)-5-amino-1-carboxypentyl]amino]-3,5-dinitrobenzoic acid Chemical compound NCCCC[C@@H](C(O)=O)NC1=C([N+]([O-])=O)C=C(C(O)=O)C=C1[N+]([O-])=O WMSPZRZYIGOEDS-QMMMGPOBSA-N 0.000 description 4
- 102100024321 Alkaline phosphatase, placental type Human genes 0.000 description 4
- 206010002198 Anaphylactic reaction Diseases 0.000 description 4
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 4
- 208000032170 Congenital Abnormalities Diseases 0.000 description 4
- 241000282326 Felis catus Species 0.000 description 4
- DHCLVCXQIBBOPH-UHFFFAOYSA-N Glycerol 2-phosphate Chemical compound OCC(CO)OP(O)(O)=O DHCLVCXQIBBOPH-UHFFFAOYSA-N 0.000 description 4
- 101000615334 Homo sapiens Antileukoproteinase Proteins 0.000 description 4
- 101000897494 Homo sapiens C-C motif chemokine 27 Proteins 0.000 description 4
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 4
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 4
- 239000004472 Lysine Substances 0.000 description 4
- 229910019142 PO4 Inorganic materials 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- 101710118538 Protease Proteins 0.000 description 4
- 201000001718 Roberts syndrome Diseases 0.000 description 4
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 4
- 230000002159 abnormal effect Effects 0.000 description 4
- 230000005856 abnormality Effects 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- HFVAFDPGUJEFBQ-UHFFFAOYSA-M alizarin red S Chemical compound [Na+].O=C1C2=CC=CC=C2C(=O)C2=C1C=C(S([O-])(=O)=O)C(O)=C2O HFVAFDPGUJEFBQ-UHFFFAOYSA-M 0.000 description 4
- 230000036783 anaphylactic response Effects 0.000 description 4
- 208000003455 anaphylaxis Diseases 0.000 description 4
- 125000000613 asparagine group Chemical group N[C@@H](CC(N)=O)C(=O)* 0.000 description 4
- 230000003197 catalytic effect Effects 0.000 description 4
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 4
- 210000000968 fibrocartilage Anatomy 0.000 description 4
- 230000013595 glycosylation Effects 0.000 description 4
- 238000006206 glycosylation reaction Methods 0.000 description 4
- 230000000004 hemodynamic effect Effects 0.000 description 4
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- 230000003834 intracellular effect Effects 0.000 description 4
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 4
- 229960000310 isoleucine Drugs 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 229930182817 methionine Natural products 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 210000002569 neuron Anatomy 0.000 description 4
- 235000021317 phosphate Nutrition 0.000 description 4
- 108010031345 placental alkaline phosphatase Proteins 0.000 description 4
- 230000004481 post-translational protein modification Effects 0.000 description 4
- 210000002966 serum Anatomy 0.000 description 4
- 230000019491 signal transduction Effects 0.000 description 4
- 238000011287 therapeutic dose Methods 0.000 description 4
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 4
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 3
- 206010067484 Adverse reaction Diseases 0.000 description 3
- RGCKGOZRHPZPFP-UHFFFAOYSA-N Alizarin Natural products C1=CC=C2C(=O)C3=C(O)C(O)=CC=C3C(=O)C2=C1 RGCKGOZRHPZPFP-UHFFFAOYSA-N 0.000 description 3
- 102100025677 Alkaline phosphatase, germ cell type Human genes 0.000 description 3
- 208000025490 Apert syndrome Diseases 0.000 description 3
- 239000004475 Arginine Substances 0.000 description 3
- 108091026890 Coding region Proteins 0.000 description 3
- 206010011878 Deafness Diseases 0.000 description 3
- 206010019280 Heart failures Diseases 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 101000574440 Homo sapiens Alkaline phosphatase, germ cell type Proteins 0.000 description 3
- 208000009289 Jackson-Weiss syndrome Diseases 0.000 description 3
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 3
- 229940124647 MEK inhibitor Drugs 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- 208000001826 Marfan syndrome Diseases 0.000 description 3
- 208000034578 Multiple myelomas Diseases 0.000 description 3
- 208000012902 Nervous system disease Diseases 0.000 description 3
- 201000004404 Neurofibroma Diseases 0.000 description 3
- 102000008763 Neurofilament Proteins Human genes 0.000 description 3
- 108010088373 Neurofilament Proteins Proteins 0.000 description 3
- 208000008589 Obesity Diseases 0.000 description 3
- 201000004014 Pfeiffer syndrome Diseases 0.000 description 3
- 102000004160 Phosphoric Monoester Hydrolases Human genes 0.000 description 3
- 108090000608 Phosphoric Monoester Hydrolases Proteins 0.000 description 3
- 206010035226 Plasma cell myeloma Diseases 0.000 description 3
- 239000004365 Protease Substances 0.000 description 3
- DVEXZJFMOKTQEZ-JYFOCSDGSA-N U0126 Chemical compound C=1C=CC=C(N)C=1SC(\N)=C(/C#N)\C(\C#N)=C(/N)SC1=CC=CC=C1N DVEXZJFMOKTQEZ-JYFOCSDGSA-N 0.000 description 3
- 238000009825 accumulation Methods 0.000 description 3
- 230000006838 adverse reaction Effects 0.000 description 3
- 238000010171 animal model Methods 0.000 description 3
- 210000003423 ankle Anatomy 0.000 description 3
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 239000004202 carbamide Substances 0.000 description 3
- 210000000170 cell membrane Anatomy 0.000 description 3
- 230000002490 cerebral effect Effects 0.000 description 3
- 210000000038 chest Anatomy 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- 238000012937 correction Methods 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 238000010586 diagram Methods 0.000 description 3
- OLSDWRNWUGHKSY-UHFFFAOYSA-J dicalcium;phosphonato phosphate;dihydrate Chemical compound O.O.[Ca+2].[Ca+2].[O-]P([O-])(=O)OP([O-])([O-])=O OLSDWRNWUGHKSY-UHFFFAOYSA-J 0.000 description 3
- 230000001882 diuretic effect Effects 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 210000000959 ear middle Anatomy 0.000 description 3
- 201000010103 fibrous dysplasia Diseases 0.000 description 3
- 208000016354 hearing loss disease Diseases 0.000 description 3
- 208000003906 hydrocephalus Diseases 0.000 description 3
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 3
- 208000011111 hypophosphatemic rickets Diseases 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 230000000968 intestinal effect Effects 0.000 description 3
- 238000001990 intravenous administration Methods 0.000 description 3
- 210000003734 kidney Anatomy 0.000 description 3
- 208000032839 leukemia Diseases 0.000 description 3
- 210000004185 liver Anatomy 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 210000001872 metatarsal bone Anatomy 0.000 description 3
- 238000010369 molecular cloning Methods 0.000 description 3
- 239000000178 monomer Substances 0.000 description 3
- 210000005044 neurofilament Anatomy 0.000 description 3
- 235000020824 obesity Nutrition 0.000 description 3
- 230000002188 osteogenic effect Effects 0.000 description 3
- 208000005368 osteomalacia Diseases 0.000 description 3
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical group [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 3
- 108010011903 peptide receptors Proteins 0.000 description 3
- 238000002732 pharmacokinetic assay Methods 0.000 description 3
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 3
- 239000010452 phosphate Substances 0.000 description 3
- 239000013612 plasmid Substances 0.000 description 3
- 229910052700 potassium Inorganic materials 0.000 description 3
- 230000002028 premature Effects 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- 230000011664 signaling Effects 0.000 description 3
- 210000003625 skull Anatomy 0.000 description 3
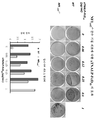
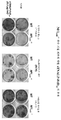
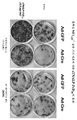
- 238000010186 staining Methods 0.000 description 3
- 238000007920 subcutaneous administration Methods 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 210000002700 urine Anatomy 0.000 description 3
- 230000004584 weight gain Effects 0.000 description 3
- 235000019786 weight gain Nutrition 0.000 description 3
- 238000001262 western blot Methods 0.000 description 3
- ZRMMVODKVLXCBB-UHFFFAOYSA-N 1-n-cyclohexyl-4-n-phenylbenzene-1,4-diamine Chemical compound C1CCCCC1NC(C=C1)=CC=C1NC1=CC=CC=C1 ZRMMVODKVLXCBB-UHFFFAOYSA-N 0.000 description 2
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 2
- 208000009304 Acute Kidney Injury Diseases 0.000 description 2
- 108010011170 Ala-Trp-Arg-His-Pro-Gln-Phe-Gly-Gly Proteins 0.000 description 2
- 208000008822 Ankylosis Diseases 0.000 description 2
- 201000001320 Atherosclerosis Diseases 0.000 description 2
- 208000000412 Avitaminosis Diseases 0.000 description 2
- 206010004146 Basal cell carcinoma Diseases 0.000 description 2
- 206010005003 Bladder cancer Diseases 0.000 description 2
- 208000010392 Bone Fractures Diseases 0.000 description 2
- 206010065687 Bone loss Diseases 0.000 description 2
- 206010006187 Breast cancer Diseases 0.000 description 2
- 208000026310 Breast neoplasm Diseases 0.000 description 2
- 206010007559 Cardiac failure congestive Diseases 0.000 description 2
- 208000024172 Cardiovascular disease Diseases 0.000 description 2
- 206010067380 Costello Syndrome Diseases 0.000 description 2
- ZZZCUOFIHGPKAK-UHFFFAOYSA-N D-erythro-ascorbic acid Natural products OCC1OC(=O)C(O)=C1O ZZZCUOFIHGPKAK-UHFFFAOYSA-N 0.000 description 2
- 208000018035 Dental disease Diseases 0.000 description 2
- 206010048768 Dermatosis Diseases 0.000 description 2
- 208000012239 Developmental disease Diseases 0.000 description 2
- NIQCNGHVCWTJSM-UHFFFAOYSA-N Dimethyl phthalate Chemical compound COC(=O)C1=CC=CC=C1C(=O)OC NIQCNGHVCWTJSM-UHFFFAOYSA-N 0.000 description 2
- 102100031480 Dual specificity mitogen-activated protein kinase kinase 1 Human genes 0.000 description 2
- 101710146526 Dual specificity mitogen-activated protein kinase kinase 1 Proteins 0.000 description 2
- 206010013935 Dysmenorrhoea Diseases 0.000 description 2
- 208000031213 Epidermal nevus syndrome Diseases 0.000 description 2
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 2
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 2
- 206010015995 Eyelid ptosis Diseases 0.000 description 2
- 206010016654 Fibrosis Diseases 0.000 description 2
- 206010017076 Fracture Diseases 0.000 description 2
- 102000018834 GTPase activity proteins Human genes 0.000 description 2
- 108040008737 GTPase activity proteins Proteins 0.000 description 2
- 206010051066 Gastrointestinal stromal tumour Diseases 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 229930186217 Glycolipid Natural products 0.000 description 2
- 206010053759 Growth retardation Diseases 0.000 description 2
- 208000028782 Hereditary disease Diseases 0.000 description 2
- 101000961040 Homo sapiens Atrial natriuretic peptide receptor 2 Proteins 0.000 description 2
- 206010020772 Hypertension Diseases 0.000 description 2
- 206010021135 Hypovitaminosis Diseases 0.000 description 2
- 208000026350 Inborn Genetic disease Diseases 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 206010023198 Joint ankylosis Diseases 0.000 description 2
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 2
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 2
- LRQKBLKVPFOOQJ-YFKPBYRVSA-N L-norleucine Chemical compound CCCC[C@H]([NH3+])C([O-])=O LRQKBLKVPFOOQJ-YFKPBYRVSA-N 0.000 description 2
- 208000005101 LEOPARD Syndrome Diseases 0.000 description 2
- 208000020358 Learning disease Diseases 0.000 description 2
- 206010025323 Lymphomas Diseases 0.000 description 2
- XUMBMVFBXHLACL-UHFFFAOYSA-N Melanin Chemical compound O=C1C(=O)C(C2=CNC3=C(C(C(=O)C4=C32)=O)C)=C2C4=CNC2=C1C XUMBMVFBXHLACL-UHFFFAOYSA-N 0.000 description 2
- 208000024556 Mendelian disease Diseases 0.000 description 2
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 2
- 208000010428 Muscle Weakness Diseases 0.000 description 2
- 206010028372 Muscular weakness Diseases 0.000 description 2
- 208000025966 Neurological disease Diseases 0.000 description 2
- 208000005890 Neuroma Diseases 0.000 description 2
- 101150083321 Nf1 gene Proteins 0.000 description 2
- 206010031243 Osteogenesis imperfecta Diseases 0.000 description 2
- 208000001132 Osteoporosis Diseases 0.000 description 2
- 108090000526 Papain Proteins 0.000 description 2
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 2
- 206010060862 Prostate cancer Diseases 0.000 description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 2
- 208000001647 Renal Insufficiency Diseases 0.000 description 2
- 208000033626 Renal failure acute Diseases 0.000 description 2
- IQFYYKKMVGJFEH-XLPZGREQSA-N Thymidine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 IQFYYKKMVGJFEH-XLPZGREQSA-N 0.000 description 2
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 2
- 229930003268 Vitamin C Natural products 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 201000011040 acute kidney failure Diseases 0.000 description 2
- 208000012998 acute renal failure Diseases 0.000 description 2
- 235000008206 alpha-amino acids Nutrition 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 208000007502 anemia Diseases 0.000 description 2
- 210000000436 anus Anatomy 0.000 description 2
- 239000003125 aqueous solvent Substances 0.000 description 2
- 230000004872 arterial blood pressure Effects 0.000 description 2
- 230000002146 bilateral effect Effects 0.000 description 2
- 230000008468 bone growth Effects 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 244000309464 bull Species 0.000 description 2
- 230000002308 calcification Effects 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 230000000747 cardiac effect Effects 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 208000020832 chronic kidney disease Diseases 0.000 description 2
- 208000024035 chronic otitis media Diseases 0.000 description 2
- 208000022831 chronic renal failure syndrome Diseases 0.000 description 2
- 238000012411 cloning technique Methods 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 210000000860 cochlear nerve Anatomy 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 238000002648 combination therapy Methods 0.000 description 2
- 238000013270 controlled release Methods 0.000 description 2
- 230000002950 deficient Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 230000004882 diastolic arterial blood pressure Effects 0.000 description 2
- FLKPEMZONWLCSK-UHFFFAOYSA-N diethyl phthalate Chemical compound CCOC(=O)C1=CC=CC=C1C(=O)OCC FLKPEMZONWLCSK-UHFFFAOYSA-N 0.000 description 2
- 230000004069 differentiation Effects 0.000 description 2
- 239000002934 diuretic Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 239000002702 enteric coating Substances 0.000 description 2
- 238000009505 enteric coating Methods 0.000 description 2
- 230000002327 eosinophilic effect Effects 0.000 description 2
- 210000002744 extracellular matrix Anatomy 0.000 description 2
- 208000009618 familial spinal neurofibromatosis Diseases 0.000 description 2
- 230000004761 fibrosis Effects 0.000 description 2
- 201000011243 gastrointestinal stromal tumor Diseases 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 239000007897 gelcap Substances 0.000 description 2
- 208000005017 glioblastoma Diseases 0.000 description 2
- 125000000404 glutamine group Chemical group N[C@@H](CCC(N)=O)C(=O)* 0.000 description 2
- 239000001087 glyceryl triacetate Substances 0.000 description 2
- 235000013773 glyceryl triacetate Nutrition 0.000 description 2
- 210000004349 growth plate Anatomy 0.000 description 2
- 231100000001 growth retardation Toxicity 0.000 description 2
- 231100000888 hearing loss Toxicity 0.000 description 2
- 230000010370 hearing loss Effects 0.000 description 2
- 208000019622 heart disease Diseases 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 239000005556 hormone Substances 0.000 description 2
- 229940088597 hormone Drugs 0.000 description 2
- 210000002758 humerus Anatomy 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 2
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 2
- 230000001077 hypotensive effect Effects 0.000 description 2
- FDGQSTZJBFJUBT-UHFFFAOYSA-N hypoxanthine Chemical compound O=C1NC=NC2=C1NC=N2 FDGQSTZJBFJUBT-UHFFFAOYSA-N 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 201000006370 kidney failure Diseases 0.000 description 2
- 201000003723 learning disability Diseases 0.000 description 2
- 210000002414 leg Anatomy 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 206010027191 meningioma Diseases 0.000 description 2
- 230000002503 metabolic effect Effects 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 235000013336 milk Nutrition 0.000 description 2
- 239000008267 milk Substances 0.000 description 2
- 210000004080 milk Anatomy 0.000 description 2
- 238000010172 mouse model Methods 0.000 description 2
- 230000002071 myeloproliferative effect Effects 0.000 description 2
- 208000006943 neurofibromatosis-pheochromocytoma-duodenal carcinoid syndrome Diseases 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 238000002638 palliative care Methods 0.000 description 2
- 229940055729 papain Drugs 0.000 description 2
- 235000019834 papain Nutrition 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 2
- 230000026731 phosphorylation Effects 0.000 description 2
- 238000006366 phosphorylation reaction Methods 0.000 description 2
- 208000031223 plasma cell leukemia Diseases 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 235000017924 poor diet Nutrition 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 238000011321 prophylaxis Methods 0.000 description 2
- 230000007065 protein hydrolysis Effects 0.000 description 2
- 201000003004 ptosis Diseases 0.000 description 2
- 102000005962 receptors Human genes 0.000 description 2
- 108020003175 receptors Proteins 0.000 description 2
- 108010023714 recombinant human bone morphogenetic protein-2 Proteins 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 208000037803 restenosis Diseases 0.000 description 2
- 206010039722 scoliosis Diseases 0.000 description 2
- 230000003248 secreting effect Effects 0.000 description 2
- 208000017520 skin disease Diseases 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- 235000012237 sodium aluminium phosphate Nutrition 0.000 description 2
- 208000005198 spinal stenosis Diseases 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 239000013589 supplement Substances 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 230000004873 systolic arterial blood pressure Effects 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- 229960002622 triacetin Drugs 0.000 description 2
- IQQWMJSNEUUJAY-UHFFFAOYSA-D trialuminum;sodium;dihydrogen phosphate;hydrogen phosphate;tetrahydrate Chemical compound O.O.O.O.[Na+].[Al+3].[Al+3].[Al+3].OP(O)([O-])=O.OP(O)([O-])=O.OP(O)([O-])=O.OP(O)([O-])=O.OP(O)([O-])=O.OP(O)([O-])=O.OP([O-])([O-])=O.OP([O-])([O-])=O IQQWMJSNEUUJAY-UHFFFAOYSA-D 0.000 description 2
- 125000000430 tryptophan group Chemical group [H]N([H])C(C(=O)O*)C([H])([H])C1=C([H])N([H])C2=C([H])C([H])=C([H])C([H])=C12 0.000 description 2
- 229910052721 tungsten Inorganic materials 0.000 description 2
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 2
- 241000701161 unidentified adenovirus Species 0.000 description 2
- 201000005112 urinary bladder cancer Diseases 0.000 description 2
- 210000004291 uterus Anatomy 0.000 description 2
- 230000000304 vasodilatating effect Effects 0.000 description 2
- 235000019154 vitamin C Nutrition 0.000 description 2
- 239000011718 vitamin C Substances 0.000 description 2
- 208000030401 vitamin deficiency disease Diseases 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- RBCOYOYDYNXAFA-UHFFFAOYSA-L (5-hydroxy-4,6-dimethylpyridin-3-yl)methyl phosphate Chemical compound CC1=NC=C(COP([O-])([O-])=O)C(C)=C1O RBCOYOYDYNXAFA-UHFFFAOYSA-L 0.000 description 1
- LDVVTQMJQSCDMK-UHFFFAOYSA-N 1,3-dihydroxypropan-2-yl formate Chemical compound OCC(CO)OC=O LDVVTQMJQSCDMK-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- PZBLUWVMZMXIKZ-UHFFFAOYSA-N 2-o-(2-ethoxy-2-oxoethyl) 1-o-ethyl benzene-1,2-dicarboxylate Chemical compound CCOC(=O)COC(=O)C1=CC=CC=C1C(=O)OCC PZBLUWVMZMXIKZ-UHFFFAOYSA-N 0.000 description 1
- HVCOBJNICQPDBP-UHFFFAOYSA-N 3-[3-[3,5-dihydroxy-6-methyl-4-(3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl]oxydecanoyloxy]decanoic acid;hydrate Chemical class O.OC1C(OC(CC(=O)OC(CCCCCCC)CC(O)=O)CCCCCCC)OC(C)C(O)C1OC1C(O)C(O)C(O)C(C)O1 HVCOBJNICQPDBP-UHFFFAOYSA-N 0.000 description 1
- UZOVYGYOLBIAJR-UHFFFAOYSA-N 4-isocyanato-4'-methyldiphenylmethane Chemical compound C1=CC(C)=CC=C1CC1=CC=C(N=C=O)C=C1 UZOVYGYOLBIAJR-UHFFFAOYSA-N 0.000 description 1
- ODHCTXKNWHHXJC-VKHMYHEASA-N 5-oxo-L-proline Chemical compound OC(=O)[C@@H]1CCC(=O)N1 ODHCTXKNWHHXJC-VKHMYHEASA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 208000021969 Acromelia Diseases 0.000 description 1
- 208000028060 Albright disease Diseases 0.000 description 1
- 201000004384 Alopecia Diseases 0.000 description 1
- 241000272525 Anas platyrhynchos Species 0.000 description 1
- 206010002329 Aneurysm Diseases 0.000 description 1
- 206010003210 Arteriosclerosis Diseases 0.000 description 1
- 241000972773 Aulopiformes Species 0.000 description 1
- 208000008035 Back Pain Diseases 0.000 description 1
- DWRXFEITVBNRMK-UHFFFAOYSA-N Beta-D-1-Arabinofuranosylthymine Natural products O=C1NC(=O)C(C)=CN1C1C(O)C(O)C(CO)O1 DWRXFEITVBNRMK-UHFFFAOYSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 108010049931 Bone Morphogenetic Protein 2 Proteins 0.000 description 1
- 102000008143 Bone Morphogenetic Protein 2 Human genes 0.000 description 1
- 102100022545 Bone morphogenetic protein 8B Human genes 0.000 description 1
- 206010006002 Bone pain Diseases 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 208000014644 Brain disease Diseases 0.000 description 1
- GOJCZVPJCKEBQV-UHFFFAOYSA-N Butyl phthalyl butylglycolate Chemical compound CCCCOC(=O)COC(=O)C1=CC=CC=C1C(=O)OCCCC GOJCZVPJCKEBQV-UHFFFAOYSA-N 0.000 description 1
- 208000000380 Cafe-au-Lait Spots Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 102000005701 Calcium-Binding Proteins Human genes 0.000 description 1
- 108010045403 Calcium-Binding Proteins Proteins 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 206010007747 Cataract congenital Diseases 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 206010008690 Chondrocalcinosis pyrophosphate Diseases 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 206010009269 Cleft palate Diseases 0.000 description 1
- 206010062344 Congenital musculoskeletal anomaly Diseases 0.000 description 1
- 208000025023 Cranial malformation Diseases 0.000 description 1
- 208000009283 Craniosynostoses Diseases 0.000 description 1
- 206010049889 Craniosynostosis Diseases 0.000 description 1
- 108010051219 Cre recombinase Proteins 0.000 description 1
- 201000001200 Crouzon syndrome-acanthosis nigricans syndrome Diseases 0.000 description 1
- 102000053602 DNA Human genes 0.000 description 1
- 208000013558 Developmental Bone disease Diseases 0.000 description 1
- 102100024746 Dihydrofolate reductase Human genes 0.000 description 1
- 208000007590 Disorders of Excessive Somnolence Diseases 0.000 description 1
- 102100023266 Dual specificity mitogen-activated protein kinase kinase 2 Human genes 0.000 description 1
- 101710146529 Dual specificity mitogen-activated protein kinase kinase 2 Proteins 0.000 description 1
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 1
- 208000013805 Dyggve-Melchior-Clausen disease Diseases 0.000 description 1
- 241001061260 Emmelichthys struhsakeri Species 0.000 description 1
- 208000032274 Encephalopathy Diseases 0.000 description 1
- 208000032541 Epidermal naevus Diseases 0.000 description 1
- 206010053177 Epidermolysis Diseases 0.000 description 1
- 241000283073 Equus caballus Species 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- 229920003134 Eudragit® polymer Polymers 0.000 description 1
- 108091008794 FGF receptors Proteins 0.000 description 1
- 102000044168 Fibroblast Growth Factor Receptor Human genes 0.000 description 1
- 102100023600 Fibroblast growth factor receptor 2 Human genes 0.000 description 1
- 101710182389 Fibroblast growth factor receptor 2 Proteins 0.000 description 1
- 206010061159 Foot deformity Diseases 0.000 description 1
- 208000034826 Genetic Predisposition to Disease Diseases 0.000 description 1
- 102100031181 Glyceraldehyde-3-phosphate dehydrogenase Human genes 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- 208000006358 Hand Deformities Diseases 0.000 description 1
- 101000749809 Homo sapiens 2',3'-cyclic-nucleotide 3'-phosphodiesterase Proteins 0.000 description 1
- 101500027325 Homo sapiens Atrial natriuretic peptide Proteins 0.000 description 1
- 101000961044 Homo sapiens Atrial natriuretic peptide receptor 1 Proteins 0.000 description 1
- 101000899368 Homo sapiens Bone morphogenetic protein 8B Proteins 0.000 description 1
- 101500026735 Homo sapiens Brain natriuretic peptide 32 Proteins 0.000 description 1
- 101000796277 Homo sapiens C-type natriuretic peptide Proteins 0.000 description 1
- 101500027331 Homo sapiens Urodilatin Proteins 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010020880 Hypertrophy Diseases 0.000 description 1
- UGQMRVRMYYASKQ-UHFFFAOYSA-N Hypoxanthine nucleoside Natural products OC1C(O)C(CO)OC1N1C(NC=NC2=O)=C2N=C1 UGQMRVRMYYASKQ-UHFFFAOYSA-N 0.000 description 1
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 description 1
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 description 1
- 208000011327 Increased bone mineral density Diseases 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 208000035478 Interatrial communication Diseases 0.000 description 1
- 206010048469 Kidney enlargement Diseases 0.000 description 1
- 208000001182 Kniest dysplasia Diseases 0.000 description 1
- ZGUNAGUHMKGQNY-ZETCQYMHSA-N L-alpha-phenylglycine zwitterion Chemical class OC(=O)[C@@H](N)C1=CC=CC=C1 ZGUNAGUHMKGQNY-ZETCQYMHSA-N 0.000 description 1
- 229930182816 L-glutamine Natural products 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 208000006286 Legius syndrome Diseases 0.000 description 1
- 208000006824 Lichtenstein syndrome Diseases 0.000 description 1
- 241000219745 Lupinus Species 0.000 description 1
- 102000019149 MAP kinase activity proteins Human genes 0.000 description 1
- 108040008097 MAP kinase activity proteins Proteins 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- JLVVSXFLKOJNIY-UHFFFAOYSA-N Magnesium ion Chemical compound [Mg+2] JLVVSXFLKOJNIY-UHFFFAOYSA-N 0.000 description 1
- 208000035450 Malformed Nails Diseases 0.000 description 1
- 201000001853 McCune-Albright syndrome Diseases 0.000 description 1
- 101710113100 Membrane-bound alkaline phosphatase Proteins 0.000 description 1
- 208000036626 Mental retardation Diseases 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 206010062901 Multiple lentigines syndrome Diseases 0.000 description 1
- 101000899367 Mus musculus Bone morphogenetic protein 8A Proteins 0.000 description 1
- 208000007101 Muscle Cramp Diseases 0.000 description 1
- 102000008300 Mutant Proteins Human genes 0.000 description 1
- 108010021466 Mutant Proteins Proteins 0.000 description 1
- 241000475481 Nebula Species 0.000 description 1
- 206010071323 Neuropsychiatric syndrome Diseases 0.000 description 1
- 208000010708 Noonan syndrome with multiple lentigines Diseases 0.000 description 1
- 208000021384 Obsessive-Compulsive disease Diseases 0.000 description 1
- 206010030113 Oedema Diseases 0.000 description 1
- 208000000035 Osteochondroma Diseases 0.000 description 1
- 208000003076 Osteolysis Diseases 0.000 description 1
- 102000004316 Oxidoreductases Human genes 0.000 description 1
- 108090000854 Oxidoreductases Proteins 0.000 description 1
- 208000002193 Pain Diseases 0.000 description 1
- 206010033425 Pain in extremity Diseases 0.000 description 1
- 208000031481 Pathologic Constriction Diseases 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 208000029082 Pelvic Inflammatory Disease Diseases 0.000 description 1
- 208000000450 Pelvic Pain Diseases 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 108091000080 Phosphotransferase Proteins 0.000 description 1
- 208000012641 Pigmentation disease Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920001710 Polyorthoester Polymers 0.000 description 1
- 208000004880 Polyuria Diseases 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 101800001295 Putative ATP-dependent helicase Proteins 0.000 description 1
- 101800001006 Putative helicase Proteins 0.000 description 1
- 206010062237 Renal impairment Diseases 0.000 description 1
- 208000012635 Rhizomelia Diseases 0.000 description 1
- 208000027790 Rib fracture Diseases 0.000 description 1
- 208000005568 Robinow syndrome Diseases 0.000 description 1
- 206010039710 Scleroderma Diseases 0.000 description 1
- YIQKLZYTHXTDDT-UHFFFAOYSA-H Sirius red F3B Chemical compound C1=CC(=CC=C1N=NC2=CC(=C(C=C2)N=NC3=C(C=C4C=C(C=CC4=C3[O-])NC(=O)NC5=CC6=CC(=C(C(=C6C=C5)[O-])N=NC7=C(C=C(C=C7)N=NC8=CC=C(C=C8)S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)O)S(=O)(=O)O)S(=O)(=O)[O-])S(=O)(=O)[O-].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+] YIQKLZYTHXTDDT-UHFFFAOYSA-H 0.000 description 1
- 206010062348 Skull malformation Diseases 0.000 description 1
- 206010041549 Spinal cord compression Diseases 0.000 description 1
- 208000014151 Stomatognathic disease Diseases 0.000 description 1
- 206010042265 Sturge-Weber Syndrome Diseases 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 206010042778 Syndactyly Diseases 0.000 description 1
- 108010076818 TEV protease Proteins 0.000 description 1
- 108020005038 Terminator Codon Proteins 0.000 description 1
- 208000008312 Tooth Loss Diseases 0.000 description 1
- 208000002148 Transposition of Great Vessels Diseases 0.000 description 1
- 206010044443 Transposition of the great vessels Diseases 0.000 description 1
- 208000026911 Tuberous sclerosis complex Diseases 0.000 description 1
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical class O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 1
- 241000469816 Varus Species 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 208000026724 Waardenburg syndrome Diseases 0.000 description 1
- 208000021017 Weight Gain Diseases 0.000 description 1
- PTFCDOFLOPIGGS-UHFFFAOYSA-N Zinc dication Chemical compound [Zn+2] PTFCDOFLOPIGGS-UHFFFAOYSA-N 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 150000001370 alpha-amino acid derivatives Chemical class 0.000 description 1
- 150000001371 alpha-amino acids Chemical class 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 229920000469 amphiphilic block copolymer Polymers 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000019552 anatomical structure morphogenesis Effects 0.000 description 1
- 235000021120 animal protein Nutrition 0.000 description 1
- 230000002547 anomalous effect Effects 0.000 description 1
- 208000022531 anorexia Diseases 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 208000008784 apnea Diseases 0.000 description 1
- 239000008365 aqueous carrier Substances 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 208000011775 arteriosclerosis disease Diseases 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229940072107 ascorbate Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 208000013914 atrial heart septal defect Diseases 0.000 description 1
- 210000002072 atrial myocyte Anatomy 0.000 description 1
- 206010003664 atrial septal defect Diseases 0.000 description 1
- 208000025261 autosomal dominant disease Diseases 0.000 description 1
- 208000021018 autosomal dominant inheritance Diseases 0.000 description 1
- 238000002869 basic local alignment search tool Methods 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- IQFYYKKMVGJFEH-UHFFFAOYSA-N beta-L-thymidine Natural products O=C1NC(=O)C(C)=CN1C1OC(CO)C(O)C1 IQFYYKKMVGJFEH-UHFFFAOYSA-N 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 230000004071 biological effect Effects 0.000 description 1
- 230000031018 biological processes and functions Effects 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 238000007470 bone biopsy Methods 0.000 description 1
- 230000037182 bone density Effects 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 210000002805 bone matrix Anatomy 0.000 description 1
- 201000006715 brachydactyly Diseases 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000000337 buffer salt Substances 0.000 description 1
- 230000003139 buffering effect Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 230000000711 cancerogenic effect Effects 0.000 description 1
- 231100000315 carcinogenic Toxicity 0.000 description 1
- 210000005242 cardiac chamber Anatomy 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 210000003321 cartilage cell Anatomy 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 210000003109 clavicle Anatomy 0.000 description 1
- 210000000078 claw Anatomy 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 230000001149 cognitive effect Effects 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000000599 controlled substance Substances 0.000 description 1
- 230000001054 cortical effect Effects 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 210000002858 crystal cell Anatomy 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 231100000895 deafness Toxicity 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 206010061428 decreased appetite Diseases 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 208000002925 dental caries Diseases 0.000 description 1
- 210000001750 dental cementum Anatomy 0.000 description 1
- 210000004268 dentin Anatomy 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 238000003745 diagnosis Methods 0.000 description 1
- PCYQQSKDZQTOQG-NXEZZACHSA-N dibutyl (2r,3r)-2,3-dihydroxybutanedioate Chemical compound CCCCOC(=O)[C@H](O)[C@@H](O)C(=O)OCCCC PCYQQSKDZQTOQG-NXEZZACHSA-N 0.000 description 1
- 235000015872 dietary supplement Nutrition 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 108020001096 dihydrofolate reductase Proteins 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- FBSAITBEAPNWJG-UHFFFAOYSA-N dimethyl phthalate Natural products CC(=O)OC1=CC=CC=C1OC(C)=O FBSAITBEAPNWJG-UHFFFAOYSA-N 0.000 description 1
- 229960001826 dimethylphthalate Drugs 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 238000003182 dose-response assay Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 230000004064 dysfunction Effects 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 238000001493 electron microscopy Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 230000010102 embolization Effects 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000005221 enamel hypoplasia Effects 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 238000002641 enzyme replacement therapy Methods 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 239000013604 expression vector Substances 0.000 description 1
- 239000012894 fetal calf serum Substances 0.000 description 1
- 210000002082 fibula Anatomy 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 210000003811 finger Anatomy 0.000 description 1
- 235000021323 fish oil Nutrition 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 210000002683 foot Anatomy 0.000 description 1
- 210000000245 forearm Anatomy 0.000 description 1
- 230000022244 formylation Effects 0.000 description 1
- 238000006170 formylation reaction Methods 0.000 description 1
- 230000037433 frameshift Effects 0.000 description 1
- UHBYWPGGCSDKFX-VKHMYHEASA-N gamma-carboxy-L-glutamic acid Chemical compound OC(=O)[C@@H](N)CC(C(O)=O)C(O)=O UHBYWPGGCSDKFX-VKHMYHEASA-N 0.000 description 1
- 210000004051 gastric juice Anatomy 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 238000001415 gene therapy Methods 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 108020004445 glyceraldehyde-3-phosphate dehydrogenase Proteins 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 210000002288 golgi apparatus Anatomy 0.000 description 1
- 230000036449 good health Effects 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 208000024963 hair loss Diseases 0.000 description 1
- 230000003676 hair loss Effects 0.000 description 1
- 210000003128 head Anatomy 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 210000001624 hip Anatomy 0.000 description 1
- 102000045328 human ALPL Human genes 0.000 description 1
- 102000047974 human CNP Human genes 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 230000009610 hypersensitivity Effects 0.000 description 1
- 206010020765 hypersomnia Diseases 0.000 description 1
- 230000002267 hypothalamic effect Effects 0.000 description 1
- 208000003532 hypothyroidism Diseases 0.000 description 1
- 230000002989 hypothyroidism Effects 0.000 description 1
- 210000001621 ilium bone Anatomy 0.000 description 1
- 229940072221 immunoglobulins Drugs 0.000 description 1
- 238000001114 immunoprecipitation Methods 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 238000009830 intercalation Methods 0.000 description 1
- 230000002687 intercalation Effects 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 201000005992 juvenile myelomonocytic leukemia Diseases 0.000 description 1
- 208000017169 kidney disease Diseases 0.000 description 1
- 230000005977 kidney dysfunction Effects 0.000 description 1
- 229940043355 kinase inhibitor Drugs 0.000 description 1
- 210000003127 knee Anatomy 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 210000000867 larynx Anatomy 0.000 description 1
- 231100000225 lethality Toxicity 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 238000004811 liquid chromatography Methods 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 230000004777 loss-of-function mutation Effects 0.000 description 1
- 210000003141 lower extremity Anatomy 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 239000008176 lyophilized powder Substances 0.000 description 1
- 239000006166 lysate Substances 0.000 description 1
- 208000029791 lytic metastatic bone lesion Diseases 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910001425 magnesium ion Inorganic materials 0.000 description 1
- 206010025482 malaise Diseases 0.000 description 1
- 210000004962 mammalian cell Anatomy 0.000 description 1
- 238000005621 mannosylation reaction Methods 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 210000001595 mastoid Anatomy 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 108020004999 messenger RNA Proteins 0.000 description 1
- 208000030159 metabolic disease Diseases 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 210000003789 metatarsus Anatomy 0.000 description 1
- 230000011987 methylation Effects 0.000 description 1
- 238000007069 methylation reaction Methods 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 238000010603 microCT Methods 0.000 description 1
- 239000003595 mist Substances 0.000 description 1
- 201000006417 multiple sclerosis Diseases 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 208000001491 myopia Diseases 0.000 description 1
- 230000004379 myopia Effects 0.000 description 1
- JSOQIZDOEIKRLY-UHFFFAOYSA-N n-propylnitrous amide Chemical compound CCCNN=O JSOQIZDOEIKRLY-UHFFFAOYSA-N 0.000 description 1
- 239000005445 natural material Substances 0.000 description 1
- 239000006225 natural substrate Substances 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000000944 nerve tissue Anatomy 0.000 description 1
- 206010061311 nervous system neoplasm Diseases 0.000 description 1
- 208000007538 neurilemmoma Diseases 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 239000002687 nonaqueous vehicle Substances 0.000 description 1
- 230000006911 nucleation Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 210000003463 organelle Anatomy 0.000 description 1
- 150000002895 organic esters Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 201000008968 osteosarcoma Diseases 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 208000003278 patent ductus arteriosus Diseases 0.000 description 1
- 201000008785 pediatric osteosarcoma Diseases 0.000 description 1
- 230000006320 pegylation Effects 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 150000002994 phenylalanines Chemical class 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 102000020233 phosphotransferase Human genes 0.000 description 1
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 1
- 210000002826 placenta Anatomy 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 238000002264 polyacrylamide gel electrophoresis Methods 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000004626 polylactic acid Substances 0.000 description 1
- 108091033319 polynucleotide Proteins 0.000 description 1
- 102000040430 polynucleotide Human genes 0.000 description 1
- 239000002157 polynucleotide Substances 0.000 description 1
- 208000001061 polyostotic fibrous dysplasia Diseases 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 210000004129 prosencephalon Anatomy 0.000 description 1
- 235000019419 proteases Nutrition 0.000 description 1
- 108020001580 protein domains Proteins 0.000 description 1
- 229940079889 pyrrolidonecarboxylic acid Drugs 0.000 description 1
- 238000001959 radiotherapy Methods 0.000 description 1
- 238000003259 recombinant expression Methods 0.000 description 1
- 230000000306 recurrent effect Effects 0.000 description 1
- 230000008085 renal dysfunction Effects 0.000 description 1
- 230000000241 respiratory effect Effects 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- 235000019515 salmon Nutrition 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 208000005706 short rib-polydactyly syndrome Diseases 0.000 description 1
- 235000021309 simple sugar Nutrition 0.000 description 1
- 231100001055 skeletal defect Toxicity 0.000 description 1
- FGCSIJPPCNCQJB-FAOVPRGRSA-M sodium;(2r,3s,4r,5r)-2,3,4,5,6-pentahydroxyhexanal;chloride Chemical compound [Na+].[Cl-].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O FGCSIJPPCNCQJB-FAOVPRGRSA-M 0.000 description 1
- VVRPOCPLIUDBSA-CNZCJKERSA-M sodium;(3r,5r)-7-[2-(4-fluorophenyl)-3-phenyl-4-(phenylcarbamoyl)-5-propan-2-ylpyrrol-1-yl]-3,5-dihydroxyheptanoate Chemical compound [Na+].C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC([O-])=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 VVRPOCPLIUDBSA-CNZCJKERSA-M 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 230000037436 splice-site mutation Effects 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 230000036262 stenosis Effects 0.000 description 1
- 208000037804 stenosis Diseases 0.000 description 1
- 239000008174 sterile solution Substances 0.000 description 1
- 208000002254 stillbirth Diseases 0.000 description 1
- 231100000537 stillbirth Toxicity 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 210000002536 stromal cell Anatomy 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 230000019635 sulfation Effects 0.000 description 1
- 238000005670 sulfation reaction Methods 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 230000009469 supplementation Effects 0.000 description 1
- 230000008093 supporting effect Effects 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 208000019270 symptomatic heart failure Diseases 0.000 description 1
- 238000002636 symptomatic treatment Methods 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 238000011285 therapeutic regimen Methods 0.000 description 1
- 210000000115 thoracic cavity Anatomy 0.000 description 1
- 210000003813 thumb Anatomy 0.000 description 1
- 229940104230 thymidine Drugs 0.000 description 1
- RWQNBRDOKXIBIV-UHFFFAOYSA-N thymine Chemical class CC1=CNC(=O)NC1=O RWQNBRDOKXIBIV-UHFFFAOYSA-N 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000027 toxicology Toxicity 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 238000006276 transfer reaction Methods 0.000 description 1
- 230000005945 translocation Effects 0.000 description 1
- 208000014903 transposition of the great arteries Diseases 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- YZWRNSARCRTXDS-UHFFFAOYSA-N tripropionin Chemical compound CCC(=O)OCC(OC(=O)CC)COC(=O)CC YZWRNSARCRTXDS-UHFFFAOYSA-N 0.000 description 1
- 201000008827 tuberculosis Diseases 0.000 description 1
- 208000009999 tuberous sclerosis Diseases 0.000 description 1
- 230000034512 ubiquitination Effects 0.000 description 1
- 238000010798 ubiquitination Methods 0.000 description 1
- IUCCYQIEZNQWRS-DWWHXVEHSA-N ularitide Chemical compound C([C@H]1C(=O)NCC(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(NCC(=O)N[C@@H](C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](C)NC(=O)[C@@H](N)[C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(O)=O)=O)[C@@H](C)CC)C1=CC=CC=C1 IUCCYQIEZNQWRS-DWWHXVEHSA-N 0.000 description 1
- 210000000623 ulna Anatomy 0.000 description 1
- 241001515965 unidentified phage Species 0.000 description 1
- 210000004509 vascular smooth muscle cell Anatomy 0.000 description 1
- 229940124549 vasodilator Drugs 0.000 description 1
- 239000003071 vasodilator agent Substances 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 208000006542 von Hippel-Lindau disease Diseases 0.000 description 1
- 230000007279 water homeostasis Effects 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 210000000707 wrist Anatomy 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/575—Hormones
- C07K14/58—Atrial natriuretic factor complex; Atriopeptin; Atrial natriuretic peptide [ANP]; Cardionatrin; Cardiodilatin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/06—Anti-spasmodics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
- C12N9/16—Hydrolases (3) acting on ester bonds (3.1)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/2278—Vasoactive intestinal peptide [VIP]; Related peptides (e.g. Exendin)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/43—Enzymes; Proenzymes; Derivatives thereof
- A61K38/46—Hydrolases (3)
- A61K38/465—Hydrolases (3) acting on ester bonds (3.1), e.g. lipases, ribonucleases
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Genetics & Genomics (AREA)
- Zoology (AREA)
- Biomedical Technology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Gastroenterology & Hepatology (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Wood Science & Technology (AREA)
- Urology & Nephrology (AREA)
- Microbiology (AREA)
- Biotechnology (AREA)
- Dermatology (AREA)
- Physical Education & Sports Medicine (AREA)
- General Engineering & Computer Science (AREA)
- Neurosurgery (AREA)
- Immunology (AREA)
- Neurology (AREA)
- Epidemiology (AREA)
- Cardiology (AREA)
- Endocrinology (AREA)
- Toxicology (AREA)
- Biophysics (AREA)
- Peptides Or Proteins (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Enzymes And Modification Thereof (AREA)
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161549047P | 2011-10-19 | 2011-10-19 | |
| US61/549,047 | 2011-10-19 | ||
| US201261649717P | 2012-05-21 | 2012-05-21 | |
| US61/649,717 | 2012-05-21 | ||
| PCT/US2012/039004 WO2013058833A1 (en) | 2011-10-19 | 2012-05-22 | Compositions comprising alkaline phosphatase and/or natriuretic peptide and methods of use thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20140084201A true KR20140084201A (ko) | 2014-07-04 |
Family
ID=48141235
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020147013214A Withdrawn KR20140084201A (ko) | 2011-10-19 | 2012-05-22 | 알칼리성 포스파타제 및/또는 나트륨이뇨 펩티드를 포함하는 조성물 및 그의 사용 방법 |
Country Status (8)
| Country | Link |
|---|---|
| EP (2) | EP2768526B1 (enExample) |
| JP (1) | JP6118807B2 (enExample) |
| KR (1) | KR20140084201A (enExample) |
| CA (1) | CA2852874A1 (enExample) |
| ES (2) | ES2714406T3 (enExample) |
| HK (1) | HK1201446A1 (enExample) |
| SG (1) | SG11201401605QA (enExample) |
| WO (2) | WO2013058833A1 (enExample) |
Families Citing this family (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| HUE055861T2 (hu) | 2004-04-21 | 2021-12-28 | Alexion Pharma Inc | Csontbejuttató konjugátumok és azok alkalmazási módszere fehérjék csonthoz kötéséhez |
| CA2823066A1 (en) * | 2010-12-27 | 2012-07-05 | Alexion Pharma International Sarl | Compositions comprising natriuretic peptides and methods of use thereof |
| US10052366B2 (en) | 2012-05-21 | 2018-08-21 | Alexion Pharmaceuticsl, Inc. | Compositions comprising alkaline phosphatase and/or natriuretic peptide and methods of use thereof |
| US10822596B2 (en) | 2014-07-11 | 2020-11-03 | Alexion Pharmaceuticals, Inc. | Compositions and methods for treating craniosynostosis |
| CA2967851C (en) | 2014-12-05 | 2024-02-27 | Alexion Pharmaceuticals, Inc. | Treating seizure with recombinant alkaline phosphatase |
| US10603361B2 (en) | 2015-01-28 | 2020-03-31 | Alexion Pharmaceuticals, Inc. | Methods of treating a subject with an alkaline phosphatase deficiency |
| JP6693134B2 (ja) * | 2015-02-02 | 2020-05-13 | 東ソー株式会社 | リンカー配列を有する抗体及びそれを用いた測定法 |
| HK1250040A1 (zh) | 2015-03-31 | 2018-11-23 | Sorriso Pharmaceuticals, Inc. | 多肽 |
| CA2981098A1 (en) * | 2015-03-31 | 2016-10-06 | Vhsquared Limited | Peptide construct having a protease-cleavable linker |
| JP6895421B2 (ja) | 2015-03-31 | 2021-06-30 | ブイエイチスクエアード リミテッド | ポリペプチド |
| TWI773649B (zh) | 2015-07-30 | 2022-08-11 | 美商拜奧馬林製藥公司 | C型利尿納肽變異體治療骨骼發育不良之用途 |
| RU2745528C2 (ru) | 2015-08-17 | 2021-03-26 | Алексион Фармасьютикалз, Инк. | Производство щелочных фосфатаз |
| WO2017058822A1 (en) * | 2015-09-28 | 2017-04-06 | Alexion Pharmaceuticals, Inc. | Identifying effective dosage regimens for tissue non-specific alkaline phosphatase (tnsalp)-enzyme replacement therapy of hypophosphatasia |
| WO2017074466A1 (en) | 2015-10-30 | 2017-05-04 | Alexion Pharmaceuticals, Inc. | Methods for treating craniosynostosis in a patient |
| EP3426286A4 (en) | 2016-03-08 | 2019-12-04 | Alexion Pharmaceuticals, Inc. | PROCESS FOR TREATING HYPOPHOSPHATASIA IN CHILDREN |
| JP7613826B2 (ja) * | 2016-04-01 | 2025-01-15 | アレクシオン ファーマシューティカルズ, インコーポレイテッド | アルカリホスファターゼによって筋力低下を治療すること |
| EP3436020A4 (en) | 2016-04-01 | 2019-12-25 | Alexion Pharmaceuticals, Inc. | METHOD FOR TREATING HYPOPHOSPHATASIE IN TEENS AND ADULTS |
| KR102449167B1 (ko) * | 2016-05-06 | 2022-09-28 | 파세비오 파마수티컬스 인코포레이티드 | 제어된 지속 방출을 위한 elp 융합 단백질 |
| US10988744B2 (en) | 2016-06-06 | 2021-04-27 | Alexion Pharmaceuticals, Inc. | Method of producing alkaline phosphatase |
| EP3500289B1 (en) * | 2016-08-18 | 2024-10-09 | Alexion Pharmaceuticals, Inc. | Asfotase alfa for use in treating tracheobronchomalacia |
| EP3519438A1 (en) | 2016-09-30 | 2019-08-07 | VHsquared Limited | Compositions |
| TW202506185A (zh) | 2017-01-20 | 2025-02-16 | 法商賽諾菲公司 | 抗TGF-β抗體及其用途 |
| AR110755A1 (es) * | 2017-01-20 | 2019-05-02 | Genzyme Corp | Anticuerpos dirigidos a hueso |
| CA3057502A1 (en) | 2017-03-31 | 2018-10-04 | Alexion Pharmaceuticals, Inc. | Methods for treating hypophosphatasia (hpp) in adults and adolescents |
| EP3737750B1 (en) | 2018-01-09 | 2024-06-05 | Theriva Biologics, Inc. | Alkaline phosphatase agents for treatment of neurodevelopmental disorders |
| US11638699B2 (en) | 2018-03-20 | 2023-05-02 | Theriva Biologics, Inc. | Intestinal alkaline phosphatase formulations |
| WO2019183209A1 (en) | 2018-03-20 | 2019-09-26 | Synthetic Biologics, Inc. | Alkaline phosphatase agents for treatment of radiation disorders |
| JP2021519590A (ja) | 2018-03-30 | 2021-08-12 | アレクシオン ファーマシューティカルズ, インコーポレイテッド | 糖タンパク質の製造 |
| JP2021534111A (ja) | 2018-08-10 | 2021-12-09 | アレクシオン ファーマシューティカルズ, インコーポレイテッド | アルカリホスファターゼを用いて神経線維腫症1型および関連状態を治療する方法 |
| CA3137045A1 (en) | 2019-05-06 | 2020-11-12 | Synthetic Biologics, Inc. | Alkaline phosphate-based oncology treatments |
| AU2020294980A1 (en) | 2019-06-21 | 2022-02-17 | Sorriso Pharmaceuticals, Inc. | Polypeptides |
| EP3986931A1 (en) | 2019-06-21 | 2022-04-27 | Sorriso Pharmaceuticals, Inc. | Polypeptides |
| US12433938B2 (en) * | 2019-12-09 | 2025-10-07 | Alexion Pharmaceuticals, Inc. | Alkaline phosphatase polypeptides and methods of use thereof |
| WO2022051555A2 (en) * | 2020-09-03 | 2022-03-10 | Rampart Bioscience, Inc. | Soluble alkaline phosphatase constructs and expression vectors including a polynucleotide encoding for soluble alkaline phosphatase constructs |
| AU2022218782A1 (en) | 2021-02-12 | 2023-08-17 | Alexion Pharmaceuticals, Inc. | Alkaline phosphatase polypeptides and methods of use thereof |
| CA3254704A1 (en) * | 2022-03-11 | 2023-09-14 | Avirmax Biopharma Inc | COMPOSITIONS AND EXPRESSION PROCESSES OF THERAPEUTIC AGENTS |
| AR130935A1 (es) | 2022-11-02 | 2025-02-05 | Novo Nordisk As | Compuestos de cnp |
| CN116286763B (zh) * | 2023-02-28 | 2024-06-25 | 西北工业大学 | 一种热稳定性提高的胱硫醚β-合成酶突变体及应用 |
| TW202513806A (zh) * | 2023-06-07 | 2025-04-01 | 美商拜奧馬林製藥公司 | 高通量篩選與身材矮小相關之基因變異體 |
Family Cites Families (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5352770A (en) | 1990-04-20 | 1994-10-04 | Hisayuki Matsuo | Porcine derived novel physiologically active peptide |
| JP2930380B2 (ja) | 1990-07-13 | 1999-08-03 | 壽之 松尾 | ブタ由来新規生理活性ペプチド(cnp―53) |
| JP3026351B2 (ja) | 1990-07-13 | 2000-03-27 | 壽之 松尾 | ブタcnp遺伝子及び前駆体蛋白 |
| JP3026354B2 (ja) | 1990-09-27 | 2000-03-27 | 壽之 松尾 | ヒトcnp遺伝子及び前駆体蛋白 |
| US5626863A (en) | 1992-02-28 | 1997-05-06 | Board Of Regents, The University Of Texas System | Photopolymerizable biodegradable hydrogels as tissue contacting materials and controlled-release carriers |
| JP2809533B2 (ja) | 1991-01-31 | 1998-10-08 | 壽之 松尾 | Cnp類似体ペプチド |
| WO1994020534A1 (en) | 1993-03-03 | 1994-09-15 | Mayo Foundation For Medical Education And Research | Vasonatrin peptide and analogs thereof |
| WO1998010087A1 (en) | 1996-09-06 | 1998-03-12 | Trustees Of The University Of Pennsylvania | Chimpanzee adenovirus vectors |
| ES2284545T3 (es) | 1999-11-16 | 2007-11-16 | Genzyme Corporation | Vectores y transgenes con elementos reguladores para la administracion de genes del higado. |
| US6407211B1 (en) | 1999-12-17 | 2002-06-18 | Mayo Foundation For Medical Education And Research | Chimeric natriuretic peptides |
| JP2002178279A (ja) | 2000-12-12 | 2002-06-25 | Ulvac Japan Ltd | 基板搬送方法 |
| IL142118A0 (en) | 2001-03-20 | 2002-03-10 | Prochon Biotech Ltd | Method and composition for treatment of skeletal dysplasias |
| EP1395293B1 (en) | 2001-05-14 | 2009-07-22 | Gbp Ip, Llc | Lentiviral vectors encoding clotting factors for gene therapy |
| BRPI0203172B8 (pt) | 2001-09-28 | 2021-05-25 | Nakao Kazuwa | composição farmacêutica para acondroplasia |
| KR100987360B1 (ko) | 2001-11-21 | 2010-10-12 | 더 트러스티스 오브 더 유니버시티 오브 펜실바니아 | 원숭이 아데노바이러스 핵산과 아미노산 서열, 이를함유하는 벡터 및 사용방법 |
| CA2433479A1 (en) | 2002-07-22 | 2004-01-22 | F. Hoffmann-La Roche Ag | Conjugate of a tissue non-specific alkaline phosphatase and dextran, process for its production and use thereof |
| DE60331584D1 (de) | 2002-11-26 | 2010-04-15 | Biocon Ltd | Modifizierte natriuretic verbindungen, konjugate und ihre verwendungen |
| WO2005007809A2 (en) | 2003-05-30 | 2005-01-27 | Alexion Pharmaceuticals, Inc. | Antibodies and fusion proteins that include engineered constant regions |
| EP3620530A1 (en) | 2004-03-31 | 2020-03-11 | Kazuwa Nakao | Composition for increasing body height |
| ES2465141T3 (es) | 2004-03-31 | 2014-06-05 | Kazuwa Nakao | Remedio o medicamento preventivo para la artritis |
| JP2005292718A (ja) | 2004-04-05 | 2005-10-20 | Furukawa Electric Co Ltd:The | 光導波路、光導波路モジュールおよび光導波路の作成方法 |
| HUE055861T2 (hu) | 2004-04-21 | 2021-12-28 | Alexion Pharma Inc | Csontbejuttató konjugátumok és azok alkalmazási módszere fehérjék csonthoz kötéséhez |
| US20070081984A1 (en) | 2005-10-11 | 2007-04-12 | Shunji Tomatsu | Compositions and methods for treating hypophosphatasia |
| JP2008521575A (ja) | 2004-12-01 | 2008-06-26 | ジェンザイム・コーポレイション | 肝臓を標的とした遺伝物質の送達方法 |
| WO2008088422A2 (en) * | 2006-10-25 | 2008-07-24 | Amgen Inc. | Toxin peptide therapeutic agents |
| US20080181903A1 (en) | 2006-12-21 | 2008-07-31 | Pdl Biopharma, Inc. | Conjugate of natriuretic peptide and antibody constant region |
| PL2158319T4 (pl) * | 2007-05-11 | 2016-04-29 | Alexion Pharma Inc | Fosfataza alkaliczna kierowana do kości, zestawy i zastosowanie |
| EP2162464A1 (en) | 2007-06-06 | 2010-03-17 | Boehringer Ingelheim International GmbH | Natriuretic fusion proteins |
| PT2765139T (pt) | 2007-07-20 | 2017-07-14 | Mayo Foundation | Polipéptidos natriuréticos |
| KR20100056517A (ko) * | 2007-09-11 | 2010-05-27 | 몬도바이오테크 래보래토리즈 아게 | 치료제로서의 펩티드의 용도 |
| AR069409A1 (es) | 2007-11-21 | 2010-01-20 | Biomarin Pharm Inc | Variantes de peptidos natriureticos de tipo c |
| HRP20161739T1 (hr) | 2009-05-20 | 2017-03-10 | Biomarin Pharmaceutical Inc. | VARIJANTE NATRIURETIČNOG PEPTIDA C-tipa |
-
2012
- 2012-05-22 SG SG11201401605QA patent/SG11201401605QA/en unknown
- 2012-05-22 WO PCT/US2012/039004 patent/WO2013058833A1/en not_active Ceased
- 2012-05-22 CA CA2852874A patent/CA2852874A1/en not_active Abandoned
- 2012-05-22 KR KR1020147013214A patent/KR20140084201A/ko not_active Withdrawn
- 2012-10-18 ES ES12842640T patent/ES2714406T3/es active Active
- 2012-10-18 WO PCT/US2012/060869 patent/WO2013059491A1/en not_active Ceased
- 2012-10-18 ES ES18201378T patent/ES2944087T3/es active Active
- 2012-10-18 EP EP12842640.0A patent/EP2768526B1/en active Active
- 2012-10-18 HK HK15101938.6A patent/HK1201446A1/xx unknown
- 2012-10-18 JP JP2014537254A patent/JP6118807B2/ja active Active
- 2012-10-18 EP EP18201378.9A patent/EP3488861B1/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| JP6118807B2 (ja) | 2017-04-19 |
| EP2768526A1 (en) | 2014-08-27 |
| EP2768526B1 (en) | 2018-12-05 |
| WO2013058833A8 (en) | 2013-06-13 |
| CA2852874A1 (en) | 2013-04-25 |
| ES2944087T3 (es) | 2023-06-19 |
| WO2013058833A1 (en) | 2013-04-25 |
| EP3488861B1 (en) | 2023-02-15 |
| EP3488861A1 (en) | 2019-05-29 |
| EP2768526A4 (en) | 2015-04-15 |
| JP2015502336A (ja) | 2015-01-22 |
| ES2714406T3 (es) | 2019-05-28 |
| HK1201446A1 (en) | 2015-09-04 |
| SG11201401605QA (en) | 2014-09-26 |
| WO2013059491A1 (en) | 2013-04-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20140084201A (ko) | 알칼리성 포스파타제 및/또는 나트륨이뇨 펩티드를 포함하는 조성물 및 그의 사용 방법 | |
| US10052366B2 (en) | Compositions comprising alkaline phosphatase and/or natriuretic peptide and methods of use thereof | |
| JP6055779B2 (ja) | ナトリウム利尿ペプチドを含む組成物およびその使用方法 | |
| RU2592670C2 (ru) | Комбинированное применение ловушек gdf и активаторов рецепторов эритропоэтина для повышения содержания эритроцитов | |
| TWI748373B (zh) | 使用gdf阱以增加紅血球水平 | |
| KR20120062874A (ko) | ActRIIb 길항제들와 이의 투약 및 용도 | |
| EA033623B1 (ru) | ВАРИАНТЫ, ПРОИСХОДЯЩИЕ ИЗ ActRIIB, И ИХ ПРИМЕНЕНИЕ | |
| EA034563B1 (ru) | Способ лечения или предотвращения перегрузки железом у пациента с талассемией | |
| KR20130118203A (ko) | 매트릭스 무기물화 장애의 처리방법, 조성물 및 키트 | |
| AU2019406214A1 (en) | Use of annexins in preventing and treating muscle membrane injury | |
| CA3166820A1 (en) | Variant actriib proteins and uses thereof | |
| US20240366722A1 (en) | Compositions and methods for treating renal diseases or conditions | |
| JP2019531070A (ja) | ツイスティッド・ガストルレーション・ポリペプチドおよびその用途 | |
| KR20230123932A (ko) | Enpp1 결핍증 및 abcc6 결핍증의 치료 | |
| RU2814047C2 (ru) | Комбинированное применение ловушек gdf и активаторов рецепторов эритропоэтина для повышения содержания эритроцитов | |
| KR20240167914A (ko) | Enpp1 결핍증 및 abcc6 결핍증의 치료 | |
| WO2025042995A1 (en) | Treatment of enpp1 deficiency and abcc6 deficiency in children | |
| BR122024002253A2 (pt) | Polipeptídeo compreendendo sequência de aminoácidos de actriib, ácido nucleico compreendendo uma sequência que codifica o dito polipeptídeo, vetor compreendendo o dito ácido nucléico, célula compreendendo polipeptídeo, ácido nucleico ou vetor, usos terapêuticos do dito polipeptídeo e do dito ácido nucléico e métodos para produzir um polupeptídeo, uma proteína ou uma heteromultímera, piroteína e heteromultímera compreendendo um polipeptídeo actriib variante |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20140516 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
Patent event date: 20160212 Comment text: Notification of Change of Applicant Patent event code: PN23011R01D |
|
| PC1203 | Withdrawal of no request for examination | ||
| WITN | Application deemed withdrawn, e.g. because no request for examination was filed or no examination fee was paid |