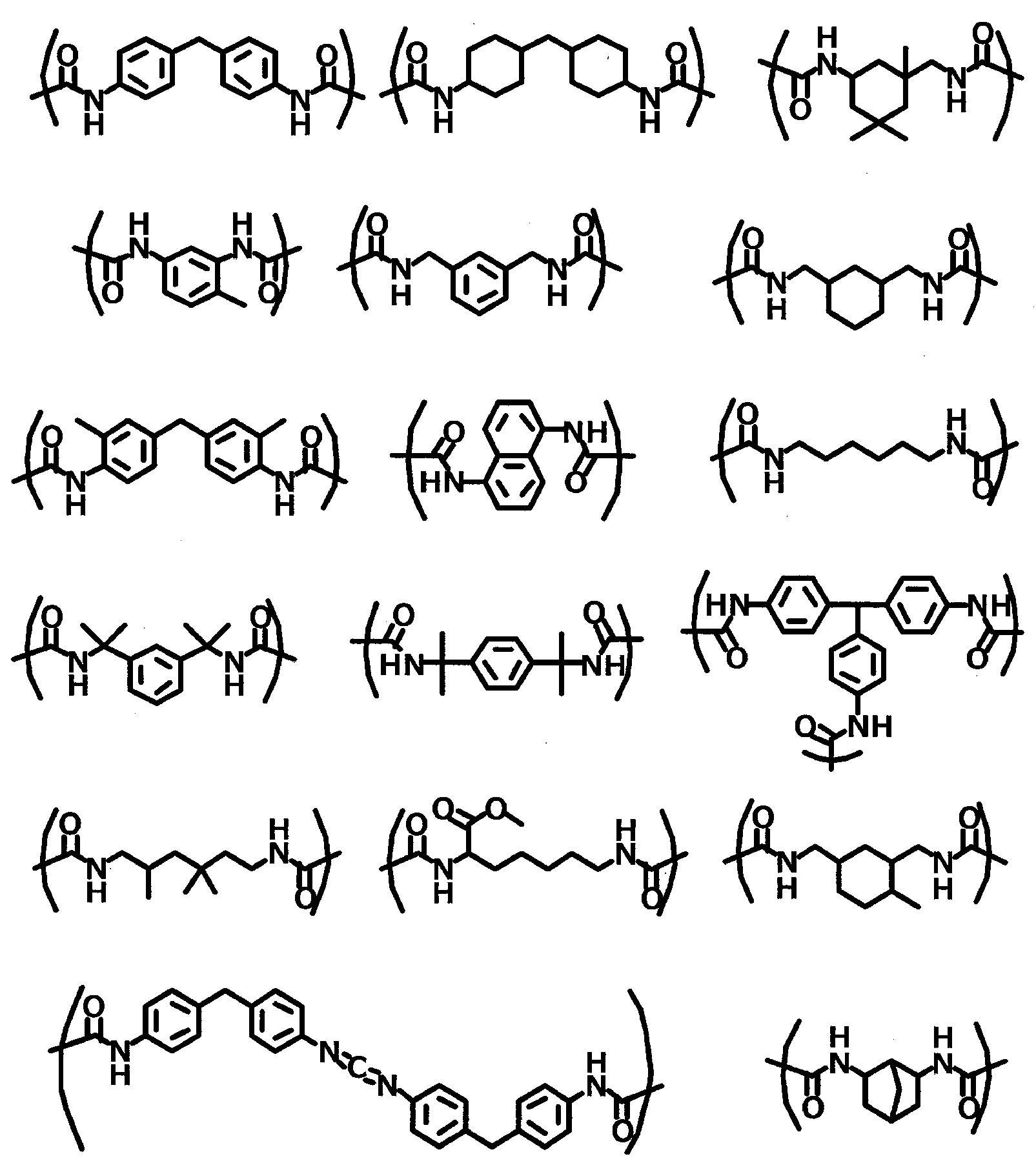
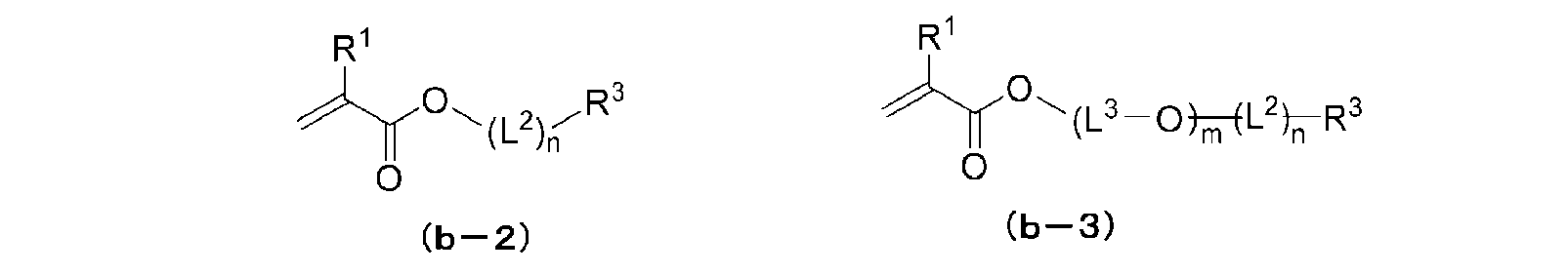
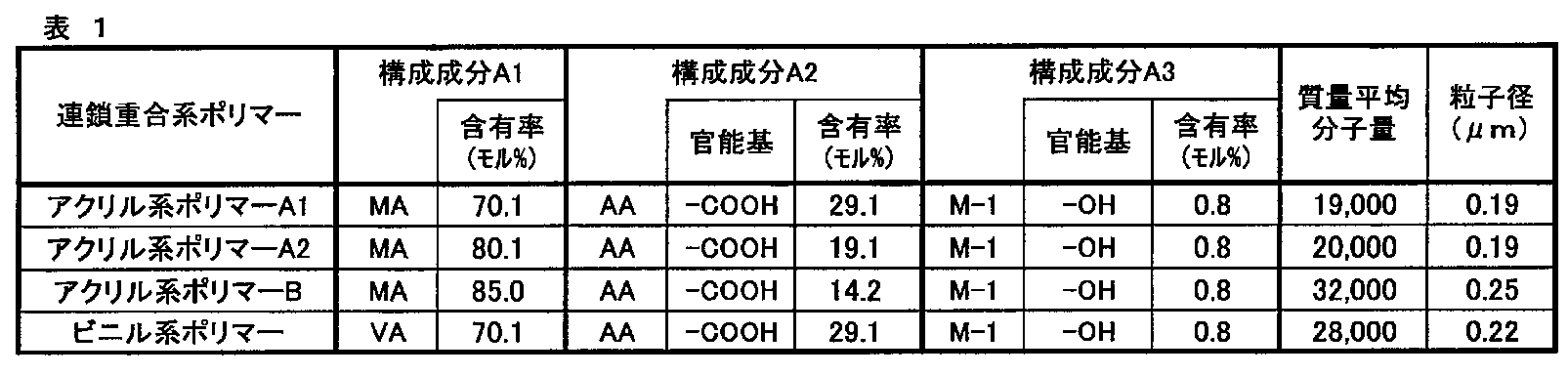
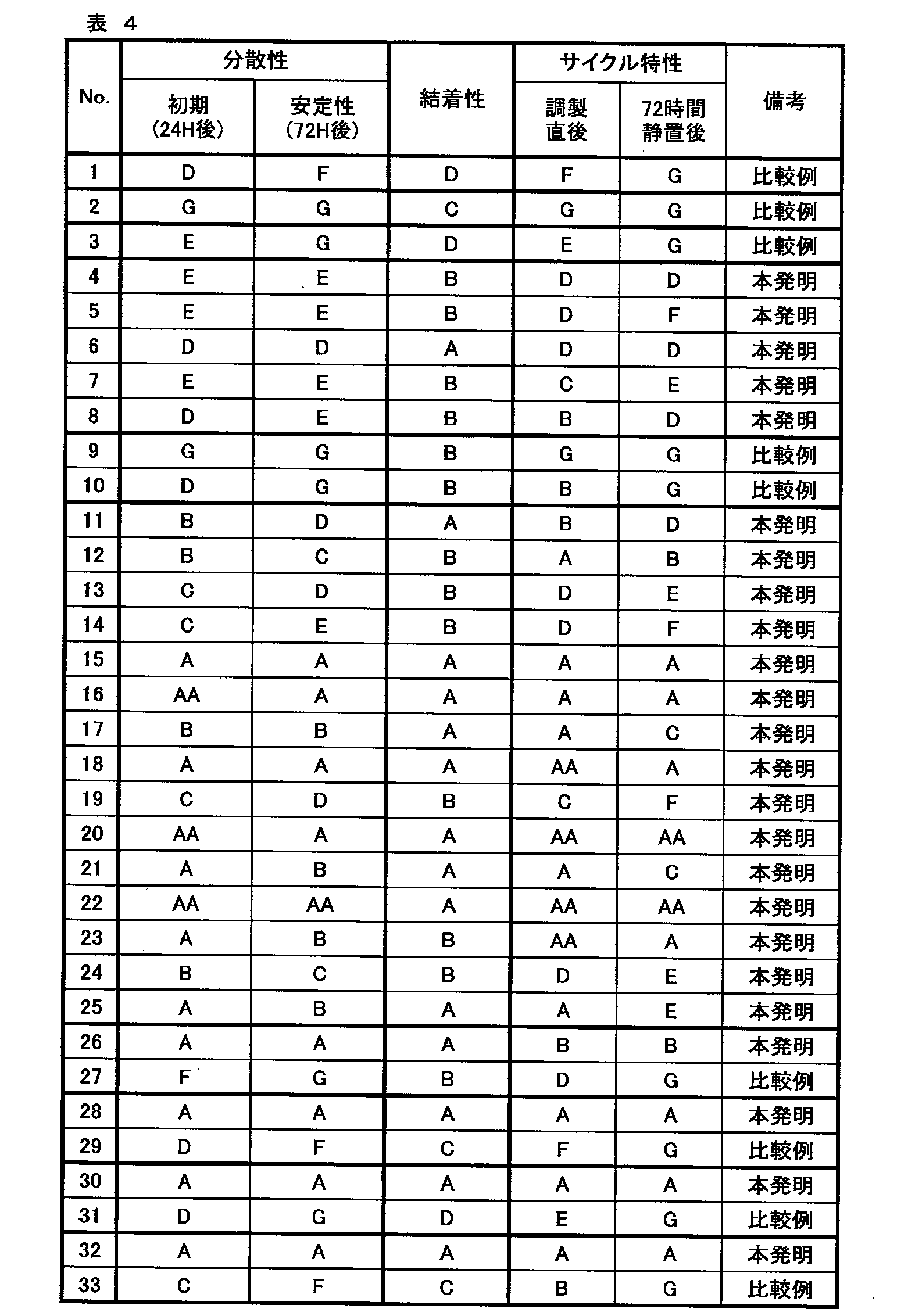
WO2021014852A1 - 無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 - Google Patents
無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 Download PDFInfo
- Publication number
- WO2021014852A1 WO2021014852A1 PCT/JP2020/024291 JP2020024291W WO2021014852A1 WO 2021014852 A1 WO2021014852 A1 WO 2021014852A1 JP 2020024291 W JP2020024291 W JP 2020024291W WO 2021014852 A1 WO2021014852 A1 WO 2021014852A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- solid electrolyte
- binder
- inorganic solid
- containing composition
- group
- Prior art date
Links
- 239000007784 solid electrolyte Substances 0.000 title claims abstract description 389
- 229910003480 inorganic solid Inorganic materials 0.000 title claims abstract description 300
- 239000000203 mixture Substances 0.000 title claims abstract description 275
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 38
- 239000011230 binding agent Substances 0.000 claims abstract description 297
- 239000003960 organic solvent Substances 0.000 claims abstract description 112
- 238000001179 sorption measurement Methods 0.000 claims abstract description 88
- 238000000034 method Methods 0.000 claims abstract description 42
- -1 ester compound Chemical class 0.000 claims description 174
- 229920000642 polymer Polymers 0.000 claims description 154
- 239000007787 solid Substances 0.000 claims description 111
- 239000011149 active material Substances 0.000 claims description 99
- 239000002245 particle Substances 0.000 claims description 83
- 239000007773 negative electrode material Substances 0.000 claims description 75
- 150000002430 hydrocarbons Chemical class 0.000 claims description 73
- 239000007774 positive electrode material Substances 0.000 claims description 52
- 229920000058 polyacrylate Polymers 0.000 claims description 37
- 239000002002 slurry Substances 0.000 claims description 30
- 229930195733 hydrocarbon Natural products 0.000 claims description 28
- 239000004215 Carbon black (E152) Substances 0.000 claims description 26
- 229920002635 polyurethane Polymers 0.000 claims description 26
- 239000004814 polyurethane Substances 0.000 claims description 26
- 239000012454 non-polar solvent Substances 0.000 claims description 21
- 230000000737 periodic effect Effects 0.000 claims description 14
- 229910052731 fluorine Inorganic materials 0.000 claims description 11
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 10
- 239000003792 electrolyte Substances 0.000 claims description 7
- 125000001153 fluoro group Chemical group F* 0.000 claims description 6
- 239000011737 fluorine Substances 0.000 claims description 3
- 229910021645 metal ion Inorganic materials 0.000 claims description 2
- 239000006185 dispersion Substances 0.000 abstract description 52
- 239000010410 layer Substances 0.000 description 245
- 239000000470 constituent Substances 0.000 description 96
- 125000004432 carbon atom Chemical group C* 0.000 description 86
- 125000001424 substituent group Chemical group 0.000 description 52
- 230000015572 biosynthetic process Effects 0.000 description 50
- 150000001875 compounds Chemical class 0.000 description 50
- 238000003786 synthesis reaction Methods 0.000 description 43
- 238000002360 preparation method Methods 0.000 description 38
- 229910052744 lithium Inorganic materials 0.000 description 33
- 239000000463 material Substances 0.000 description 33
- 229910052751 metal Inorganic materials 0.000 description 31
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 30
- 125000005647 linker group Chemical group 0.000 description 29
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 27
- 239000002184 metal Substances 0.000 description 27
- 229910052710 silicon Inorganic materials 0.000 description 26
- 239000011248 coating agent Substances 0.000 description 25
- 229920002554 vinyl polymer Polymers 0.000 description 25
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 24
- 238000000576 coating method Methods 0.000 description 24
- 125000000524 functional group Chemical group 0.000 description 24
- 239000000126 substance Substances 0.000 description 24
- PTTPXKJBFFKCEK-UHFFFAOYSA-N 2-Methyl-4-heptanone Chemical compound CC(C)CC(=O)CC(C)C PTTPXKJBFFKCEK-UHFFFAOYSA-N 0.000 description 22
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 22
- 229910052782 aluminium Inorganic materials 0.000 description 22
- 125000000217 alkyl group Chemical group 0.000 description 21
- 239000002033 PVDF binder Substances 0.000 description 20
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 20
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 20
- 239000000243 solution Substances 0.000 description 20
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 19
- 229920001577 copolymer Polymers 0.000 description 19
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 19
- 229910052799 carbon Inorganic materials 0.000 description 18
- 238000011156 evaluation Methods 0.000 description 18
- 239000010703 silicon Substances 0.000 description 17
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 16
- 125000003118 aryl group Chemical group 0.000 description 16
- 229910001416 lithium ion Inorganic materials 0.000 description 16
- 239000012752 auxiliary agent Substances 0.000 description 15
- 239000002994 raw material Substances 0.000 description 15
- 238000007155 step growth polymerization reaction Methods 0.000 description 15
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 15
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 14
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 14
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 14
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 14
- 239000010936 titanium Substances 0.000 description 14
- 125000001931 aliphatic group Chemical group 0.000 description 13
- 125000002947 alkylene group Chemical group 0.000 description 13
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 13
- 239000011888 foil Substances 0.000 description 13
- 229920000233 poly(alkylene oxides) Chemical group 0.000 description 13
- 125000004429 atom Chemical group 0.000 description 12
- 239000010408 film Substances 0.000 description 12
- 125000000623 heterocyclic group Chemical group 0.000 description 12
- 238000005259 measurement Methods 0.000 description 12
- 239000003575 carbonaceous material Substances 0.000 description 11
- 229910052719 titanium Inorganic materials 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical group C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 10
- 239000011889 copper foil Substances 0.000 description 10
- 125000005843 halogen group Chemical group 0.000 description 10
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 10
- 239000002904 solvent Substances 0.000 description 10
- 229910052717 sulfur Inorganic materials 0.000 description 10
- 238000012360 testing method Methods 0.000 description 10
- 229910052718 tin Inorganic materials 0.000 description 10
- 229910000314 transition metal oxide Inorganic materials 0.000 description 10
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 9
- 229920000049 Carbon (fiber) Polymers 0.000 description 9
- 239000004793 Polystyrene Substances 0.000 description 9
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 9
- 238000001035 drying Methods 0.000 description 9
- 229910002804 graphite Inorganic materials 0.000 description 9
- 239000010439 graphite Substances 0.000 description 9
- 150000002500 ions Chemical class 0.000 description 9
- 125000004430 oxygen atom Chemical group O* 0.000 description 9
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 8
- 229910045601 alloy Inorganic materials 0.000 description 8
- 239000000956 alloy Substances 0.000 description 8
- 239000004917 carbon fiber Substances 0.000 description 8
- 239000002131 composite material Substances 0.000 description 8
- 229910052802 copper Inorganic materials 0.000 description 8
- 239000010949 copper Substances 0.000 description 8
- IIEWJVIFRVWJOD-UHFFFAOYSA-N ethylcyclohexane Chemical compound CCC1CCCCC1 IIEWJVIFRVWJOD-UHFFFAOYSA-N 0.000 description 8
- 239000011521 glass Substances 0.000 description 8
- 239000001257 hydrogen Substances 0.000 description 8
- 229910052739 hydrogen Inorganic materials 0.000 description 8
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 8
- 229910003002 lithium salt Inorganic materials 0.000 description 8
- 159000000002 lithium salts Chemical class 0.000 description 8
- 229910044991 metal oxide Inorganic materials 0.000 description 8
- 238000002156 mixing Methods 0.000 description 8
- 229910052759 nickel Inorganic materials 0.000 description 8
- 239000004417 polycarbonate Substances 0.000 description 8
- 229920000515 polycarbonate Polymers 0.000 description 8
- 229920000728 polyester Polymers 0.000 description 8
- 239000010935 stainless steel Substances 0.000 description 8
- 229910001220 stainless steel Inorganic materials 0.000 description 8
- 229910018091 Li 2 S Inorganic materials 0.000 description 7
- 239000006230 acetylene black Substances 0.000 description 7
- 125000005370 alkoxysilyl group Chemical group 0.000 description 7
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 7
- 150000004770 chalcogenides Chemical class 0.000 description 7
- 230000006866 deterioration Effects 0.000 description 7
- 230000006870 function Effects 0.000 description 7
- 229910052733 gallium Inorganic materials 0.000 description 7
- 229910052732 germanium Inorganic materials 0.000 description 7
- HCDGVLDPFQMKDK-UHFFFAOYSA-N hexafluoropropylene Chemical group FC(F)=C(F)C(F)(F)F HCDGVLDPFQMKDK-UHFFFAOYSA-N 0.000 description 7
- 229920003048 styrene butadiene rubber Polymers 0.000 description 7
- 229910052723 transition metal Inorganic materials 0.000 description 7
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 6
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 6
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical group OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 6
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 229910052796 boron Inorganic materials 0.000 description 6
- XUPYJHCZDLZNFP-UHFFFAOYSA-N butyl butanoate Chemical compound CCCCOC(=O)CCC XUPYJHCZDLZNFP-UHFFFAOYSA-N 0.000 description 6
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 6
- 239000013078 crystal Substances 0.000 description 6
- 150000004678 hydrides Chemical class 0.000 description 6
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 6
- 150000004706 metal oxides Chemical class 0.000 description 6
- 239000000178 monomer Substances 0.000 description 6
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 6
- 238000004062 sedimentation Methods 0.000 description 6
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 6
- 235000002639 sodium chloride Nutrition 0.000 description 6
- 238000003756 stirring Methods 0.000 description 6
- 150000007824 aliphatic compounds Chemical class 0.000 description 5
- 125000003342 alkenyl group Chemical group 0.000 description 5
- 125000003277 amino group Chemical group 0.000 description 5
- 150000001491 aromatic compounds Chemical class 0.000 description 5
- 150000001721 carbon Chemical group 0.000 description 5
- 238000007334 copolymerization reaction Methods 0.000 description 5
- 239000002270 dispersing agent Substances 0.000 description 5
- 125000001033 ether group Chemical group 0.000 description 5
- 230000001747 exhibiting effect Effects 0.000 description 5
- 229920002313 fluoropolymer Polymers 0.000 description 5
- 239000004811 fluoropolymer Substances 0.000 description 5
- 238000010438 heat treatment Methods 0.000 description 5
- 230000006872 improvement Effects 0.000 description 5
- 229910052738 indium Inorganic materials 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- 229910052752 metalloid Inorganic materials 0.000 description 5
- 229910052698 phosphorus Inorganic materials 0.000 description 5
- 238000006068 polycondensation reaction Methods 0.000 description 5
- 229920000139 polyethylene terephthalate Polymers 0.000 description 5
- 239000005020 polyethylene terephthalate Substances 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 229920006132 styrene block copolymer Polymers 0.000 description 5
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 description 4
- 229920000178 Acrylic resin Polymers 0.000 description 4
- 239000004925 Acrylic resin Substances 0.000 description 4
- 229910000838 Al alloy Inorganic materials 0.000 description 4
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 4
- 229910018130 Li 2 S-P 2 S 5 Inorganic materials 0.000 description 4
- 239000004642 Polyimide Substances 0.000 description 4
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 4
- 125000003545 alkoxy group Chemical group 0.000 description 4
- 125000000304 alkynyl group Chemical group 0.000 description 4
- 229910052787 antimony Inorganic materials 0.000 description 4
- 125000002029 aromatic hydrocarbon group Chemical group 0.000 description 4
- 125000004104 aryloxy group Chemical group 0.000 description 4
- 239000011324 bead Substances 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical group C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- NNBZCPXTIHJBJL-UHFFFAOYSA-N decalin Chemical compound C1CCCC2CCCCC21 NNBZCPXTIHJBJL-UHFFFAOYSA-N 0.000 description 4
- 238000002296 dynamic light scattering Methods 0.000 description 4
- 238000010304 firing Methods 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 150000004820 halides Chemical class 0.000 description 4
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 4
- 230000014759 maintenance of location Effects 0.000 description 4
- 239000002905 metal composite material Substances 0.000 description 4
- 229910001317 nickel manganese cobalt oxide (NMC) Inorganic materials 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- 239000003495 polar organic solvent Substances 0.000 description 4
- 229920005569 poly(vinylidene fluoride-co-hexafluoropropylene) Polymers 0.000 description 4
- 229920001721 polyimide Polymers 0.000 description 4
- 229920002223 polystyrene Polymers 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 125000000542 sulfonic acid group Chemical group 0.000 description 4
- 125000004434 sulfur atom Chemical group 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 4
- 125000003396 thiol group Chemical group [H]S* 0.000 description 4
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 description 4
- 238000002834 transmittance Methods 0.000 description 4
- 229910052720 vanadium Inorganic materials 0.000 description 4
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 3
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 229910000881 Cu alloy Inorganic materials 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 229910001228 Li[Ni1/3Co1/3Mn1/3]O2 (NCM 111) Inorganic materials 0.000 description 3
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 3
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 3
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 3
- 239000004952 Polyamide Substances 0.000 description 3
- 239000004698 Polyethylene Substances 0.000 description 3
- 229920002396 Polyurea Polymers 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 229910006404 SnO 2 Inorganic materials 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 description 3
- 125000002252 acyl group Chemical group 0.000 description 3
- 125000004423 acyloxy group Chemical group 0.000 description 3
- 229910021383 artificial graphite Inorganic materials 0.000 description 3
- 125000003710 aryl alkyl group Chemical group 0.000 description 3
- 125000000732 arylene group Chemical group 0.000 description 3
- BTANRVKWQNVYAZ-UHFFFAOYSA-N butan-2-ol Chemical compound CCC(C)O BTANRVKWQNVYAZ-UHFFFAOYSA-N 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 239000011247 coating layer Substances 0.000 description 3
- 239000004020 conductor Substances 0.000 description 3
- 125000004093 cyano group Chemical group *C#N 0.000 description 3
- 125000000753 cycloalkyl group Chemical group 0.000 description 3
- 150000001993 dienes Chemical class 0.000 description 3
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 3
- 229910001873 dinitrogen Inorganic materials 0.000 description 3
- 238000007599 discharging Methods 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000007772 electrode material Substances 0.000 description 3
- 150000002170 ethers Chemical class 0.000 description 3
- 238000005227 gel permeation chromatography Methods 0.000 description 3
- 230000009477 glass transition Effects 0.000 description 3
- 239000002241 glass-ceramic Substances 0.000 description 3
- 125000001841 imino group Chemical group [H]N=* 0.000 description 3
- 230000001965 increasing effect Effects 0.000 description 3
- HSZCZNFXUDYRKD-UHFFFAOYSA-M lithium iodide Inorganic materials [Li+].[I-] HSZCZNFXUDYRKD-UHFFFAOYSA-M 0.000 description 3
- GLNWILHOFOBOFD-UHFFFAOYSA-N lithium sulfide Chemical compound [Li+].[Li+].[S-2] GLNWILHOFOBOFD-UHFFFAOYSA-N 0.000 description 3
- 238000000691 measurement method Methods 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 229940043265 methyl isobutyl ketone Drugs 0.000 description 3
- 239000004570 mortar (masonry) Substances 0.000 description 3
- 229910021382 natural graphite Inorganic materials 0.000 description 3
- 229910052758 niobium Inorganic materials 0.000 description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 description 3
- CYQAYERJWZKYML-UHFFFAOYSA-N phosphorus pentasulfide Chemical compound S1P(S2)(=S)SP3(=S)SP1(=S)SP2(=S)S3 CYQAYERJWZKYML-UHFFFAOYSA-N 0.000 description 3
- 229920002239 polyacrylonitrile Polymers 0.000 description 3
- 229920002647 polyamide Polymers 0.000 description 3
- 229920000573 polyethylene Polymers 0.000 description 3
- 238000006116 polymerization reaction Methods 0.000 description 3
- 229920000346 polystyrene-polyisoprene block-polystyrene Polymers 0.000 description 3
- 239000011241 protective layer Substances 0.000 description 3
- 229920005989 resin Polymers 0.000 description 3
- 239000011347 resin Substances 0.000 description 3
- 235000012239 silicon dioxide Nutrition 0.000 description 3
- 239000011029 spinel Substances 0.000 description 3
- 229910052596 spinel Inorganic materials 0.000 description 3
- 229920000468 styrene butadiene styrene block copolymer Polymers 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 239000011593 sulfur Substances 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- 239000008096 xylene Substances 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 description 2
- KBPLFHHGFOOTCA-UHFFFAOYSA-N 1-Octanol Chemical compound CCCCCCCCO KBPLFHHGFOOTCA-UHFFFAOYSA-N 0.000 description 2
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 2
- ULQISTXYYBZJSJ-UHFFFAOYSA-N 12-hydroxyoctadecanoic acid Chemical compound CCCCCCC(O)CCCCCCCCCCC(O)=O ULQISTXYYBZJSJ-UHFFFAOYSA-N 0.000 description 2
- HXVNBWAKAOHACI-UHFFFAOYSA-N 2,4-dimethyl-3-pentanone Chemical compound CC(C)C(=O)C(C)C HXVNBWAKAOHACI-UHFFFAOYSA-N 0.000 description 2
- RXGUIWHIADMCFC-UHFFFAOYSA-N 2-Methylpropyl 2-methylpropionate Chemical compound CC(C)COC(=O)C(C)C RXGUIWHIADMCFC-UHFFFAOYSA-N 0.000 description 2
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 2
- SVTBMSDMJJWYQN-UHFFFAOYSA-N 2-methylpentane-2,4-diol Chemical compound CC(O)CC(C)(C)O SVTBMSDMJJWYQN-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical class [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- HCFAJYNVAYBARA-UHFFFAOYSA-N 4-heptanone Chemical compound CCCC(=O)CCC HCFAJYNVAYBARA-UHFFFAOYSA-N 0.000 description 2
- DLFVBJFMPXGRIB-UHFFFAOYSA-N Acetamide Chemical compound CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- 229910015902 Bi 2 O 3 Inorganic materials 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 2
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 2
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 2
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- FFOPEPMHKILNIT-UHFFFAOYSA-N Isopropyl butyrate Chemical compound CCCC(=O)OC(C)C FFOPEPMHKILNIT-UHFFFAOYSA-N 0.000 description 2
- 229910018251 LaSi 2 Inorganic materials 0.000 description 2
- 229910018119 Li 3 PO 4 Inorganic materials 0.000 description 2
- 229910000733 Li alloy Inorganic materials 0.000 description 2
- 229910008323 Li-P-S Inorganic materials 0.000 description 2
- 229910015643 LiMn 2 O 4 Inorganic materials 0.000 description 2
- 229910015701 LiNi0.85Co0.10Al0.05O2 Inorganic materials 0.000 description 2
- 229910006736 Li—P—S Inorganic materials 0.000 description 2
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 2
- ATHHXGZTWNVVOU-UHFFFAOYSA-N N-methylformamide Chemical compound CNC=O ATHHXGZTWNVVOU-UHFFFAOYSA-N 0.000 description 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 2
- 239000005062 Polybutadiene Substances 0.000 description 2
- 239000004721 Polyphenylene oxide Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- 229910004298 SiO 2 Inorganic materials 0.000 description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 2
- 239000002174 Styrene-butadiene Substances 0.000 description 2
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical compound CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 2
- PFYQFCKUASLJLL-UHFFFAOYSA-N [Co].[Ni].[Li] Chemical compound [Co].[Ni].[Li] PFYQFCKUASLJLL-UHFFFAOYSA-N 0.000 description 2
- 125000002777 acetyl group Chemical class [H]C([H])([H])C(*)=O 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 125000004442 acylamino group Chemical group 0.000 description 2
- 238000004220 aggregation Methods 0.000 description 2
- 230000002776 aggregation Effects 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 2
- 125000004450 alkenylene group Chemical group 0.000 description 2
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 2
- 125000003282 alkyl amino group Chemical group 0.000 description 2
- 125000005103 alkyl silyl group Chemical group 0.000 description 2
- 125000004390 alkyl sulfonyl group Chemical group 0.000 description 2
- 125000004414 alkyl thio group Chemical group 0.000 description 2
- 125000005336 allyloxy group Chemical group 0.000 description 2
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 2
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 2
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 description 2
- 238000005280 amorphization Methods 0.000 description 2
- 150000001450 anions Chemical class 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000012300 argon atmosphere Substances 0.000 description 2
- 125000001769 aryl amino group Chemical group 0.000 description 2
- 125000005161 aryl oxy carbonyl group Chemical group 0.000 description 2
- 125000005104 aryl silyl group Chemical group 0.000 description 2
- 125000005110 aryl thio group Chemical group 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 238000006664 bond formation reaction Methods 0.000 description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 2
- 229920005549 butyl rubber Polymers 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 2
- 239000006229 carbon black Substances 0.000 description 2
- 235000019241 carbon black Nutrition 0.000 description 2
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 2
- 239000002134 carbon nanofiber Substances 0.000 description 2
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 238000007156 chain growth polymerization reaction Methods 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 229910052804 chromium Inorganic materials 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 229920006037 cross link polymer Polymers 0.000 description 2
- 238000004132 cross linking Methods 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- WJTCGQSWYFHTAC-UHFFFAOYSA-N cyclooctane Chemical compound C1CCCCCCC1 WJTCGQSWYFHTAC-UHFFFAOYSA-N 0.000 description 2
- 239000004914 cyclooctane Substances 0.000 description 2
- BGTOWKSIORTVQH-UHFFFAOYSA-N cyclopentanone Chemical compound O=C1CCCC1 BGTOWKSIORTVQH-UHFFFAOYSA-N 0.000 description 2
- DIOQZVSQGTUSAI-UHFFFAOYSA-N decane Chemical compound CCCCCCCCCC DIOQZVSQGTUSAI-UHFFFAOYSA-N 0.000 description 2
- 238000000151 deposition Methods 0.000 description 2
- 229940028356 diethylene glycol monobutyl ether Drugs 0.000 description 2
- 239000008151 electrolyte solution Substances 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 230000002708 enhancing effect Effects 0.000 description 2
- JBKVHLHDHHXQEQ-UHFFFAOYSA-N epsilon-caprolactam Chemical compound O=C1CCCCCN1 JBKVHLHDHHXQEQ-UHFFFAOYSA-N 0.000 description 2
- 235000019441 ethanol Nutrition 0.000 description 2
- WDAXFOBOLVPGLV-UHFFFAOYSA-N ethyl isobutyrate Chemical compound CCOC(=O)C(C)C WDAXFOBOLVPGLV-UHFFFAOYSA-N 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 239000000706 filtrate Substances 0.000 description 2
- VOZRXNHHFUQHIL-UHFFFAOYSA-N glycidyl methacrylate Chemical compound CC(=C)C(=O)OCC1CO1 VOZRXNHHFUQHIL-UHFFFAOYSA-N 0.000 description 2
- 229910021385 hard carbon Inorganic materials 0.000 description 2
- 238000005984 hydrogenation reaction Methods 0.000 description 2
- 230000001771 impaired effect Effects 0.000 description 2
- 239000011261 inert gas Substances 0.000 description 2
- 229910010272 inorganic material Inorganic materials 0.000 description 2
- 239000011147 inorganic material Substances 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 239000002608 ionic liquid Substances 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N iron Substances [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- RGFNRWTWDWVHDD-UHFFFAOYSA-N isobutyl butyrate Chemical compound CCCC(=O)OCC(C)C RGFNRWTWDWVHDD-UHFFFAOYSA-N 0.000 description 2
- 125000001261 isocyanato group Chemical group *N=C=O 0.000 description 2
- HJOVHMDZYOCNQW-UHFFFAOYSA-N isophorone Chemical compound CC1=CC(=O)CC(C)(C)C1 HJOVHMDZYOCNQW-UHFFFAOYSA-N 0.000 description 2
- 239000001989 lithium alloy Substances 0.000 description 2
- 229910003473 lithium bis(trifluoromethanesulfonyl)imide Inorganic materials 0.000 description 2
- FUJCRWPEOMXPAD-UHFFFAOYSA-N lithium oxide Chemical compound [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 2
- 229910001947 lithium oxide Inorganic materials 0.000 description 2
- 229910001386 lithium phosphate Inorganic materials 0.000 description 2
- VDVLPSWVDYJFRW-UHFFFAOYSA-N lithium;bis(fluorosulfonyl)azanide Chemical compound [Li+].FS(=O)(=O)[N-]S(F)(=O)=O VDVLPSWVDYJFRW-UHFFFAOYSA-N 0.000 description 2
- QSZMZKBZAYQGRS-UHFFFAOYSA-N lithium;bis(trifluoromethylsulfonyl)azanide Chemical compound [Li+].FC(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)F QSZMZKBZAYQGRS-UHFFFAOYSA-N 0.000 description 2
- VGYDTVNNDKLMHX-UHFFFAOYSA-N lithium;manganese;nickel;oxocobalt Chemical compound [Li].[Mn].[Ni].[Co]=O VGYDTVNNDKLMHX-UHFFFAOYSA-N 0.000 description 2
- 239000011572 manganese Substances 0.000 description 2
- 238000003701 mechanical milling Methods 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 238000002844 melting Methods 0.000 description 2
- 230000008018 melting Effects 0.000 description 2
- UAEPNZWRGJTJPN-UHFFFAOYSA-N methylcyclohexane Chemical compound CC1CCCCC1 UAEPNZWRGJTJPN-UHFFFAOYSA-N 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- 238000000465 moulding Methods 0.000 description 2
- CLNYHERYALISIR-UHFFFAOYSA-N nona-1,3-diene Chemical compound CCCCCC=CC=C CLNYHERYALISIR-UHFFFAOYSA-N 0.000 description 2
- YWXLSHOWXZUMSR-UHFFFAOYSA-N octan-4-one Chemical compound CCCCC(=O)CCC YWXLSHOWXZUMSR-UHFFFAOYSA-N 0.000 description 2
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 2
- 239000011368 organic material Substances 0.000 description 2
- JCGNDDUYTRNOFT-UHFFFAOYSA-N oxolane-2,4-dione Chemical compound O=C1COC(=O)C1 JCGNDDUYTRNOFT-UHFFFAOYSA-N 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 125000005328 phosphinyl group Chemical group [PH2](=O)* 0.000 description 2
- 125000005499 phosphonyl group Chemical group 0.000 description 2
- 239000011574 phosphorus Substances 0.000 description 2
- 229920002857 polybutadiene Polymers 0.000 description 2
- 229920000570 polyether Polymers 0.000 description 2
- 229940113115 polyethylene glycol 200 Drugs 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 238000003825 pressing Methods 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- 238000010298 pulverizing process Methods 0.000 description 2
- HNJBEVLQSNELDL-UHFFFAOYSA-N pyrrolidin-2-one Chemical group O=C1CCCN1 HNJBEVLQSNELDL-UHFFFAOYSA-N 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 229910052814 silicon oxide Inorganic materials 0.000 description 2
- 239000002210 silicon-based material Substances 0.000 description 2
- 229910052709 silver Inorganic materials 0.000 description 2
- 239000004332 silver Substances 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- 229910052715 tantalum Inorganic materials 0.000 description 2
- 229920002725 thermoplastic elastomer Polymers 0.000 description 2
- 125000000101 thioether group Chemical group 0.000 description 2
- IMFACGCPASFAPR-UHFFFAOYSA-N tributylamine Chemical compound CCCCN(CCCC)CCCC IMFACGCPASFAPR-UHFFFAOYSA-N 0.000 description 2
- ZIBGPFATKBEMQZ-UHFFFAOYSA-N triethylene glycol Chemical compound OCCOCCOCCO ZIBGPFATKBEMQZ-UHFFFAOYSA-N 0.000 description 2
- TWQULNDIKKJZPH-UHFFFAOYSA-K trilithium;phosphate Chemical compound [Li+].[Li+].[Li+].[O-]P([O-])([O-])=O TWQULNDIKKJZPH-UHFFFAOYSA-K 0.000 description 2
- 229910052721 tungsten Inorganic materials 0.000 description 2
- PXXNTAGJWPJAGM-UHFFFAOYSA-N vertaline Natural products C1C2C=3C=C(OC)C(OC)=CC=3OC(C=C3)=CC=C3CCC(=O)OC1CC1N2CCCC1 PXXNTAGJWPJAGM-UHFFFAOYSA-N 0.000 description 2
- 229910052726 zirconium Inorganic materials 0.000 description 2
- CYSGHNMQYZDMIA-UHFFFAOYSA-N 1,3-Dimethyl-2-imidazolidinon Chemical compound CN1CCN(C)C1=O CYSGHNMQYZDMIA-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- APQXWKHOGQFGTB-UHFFFAOYSA-N 1-ethenyl-9h-carbazole Chemical class C12=CC=CC=C2NC2=C1C=CC=C2C=C APQXWKHOGQFGTB-UHFFFAOYSA-N 0.000 description 1
- 125000006219 1-ethylpentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- IGGDKDTUCAWDAN-UHFFFAOYSA-N 1-vinylnaphthalene Chemical class C1=CC=C2C(C=C)=CC=CC2=C1 IGGDKDTUCAWDAN-UHFFFAOYSA-N 0.000 description 1
- 229940114072 12-hydroxystearic acid Drugs 0.000 description 1
- JVYDLYGCSIHCMR-UHFFFAOYSA-N 2,2-bis(hydroxymethyl)butanoic acid Chemical compound CCC(CO)(CO)C(O)=O JVYDLYGCSIHCMR-UHFFFAOYSA-N 0.000 description 1
- SBASXUCJHJRPEV-UHFFFAOYSA-N 2-(2-methoxyethoxy)ethanol Chemical compound COCCOCCO SBASXUCJHJRPEV-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- WAEVWDZKMBQDEJ-UHFFFAOYSA-N 2-[2-(2-methoxypropoxy)propoxy]propan-1-ol Chemical compound COC(C)COC(C)COC(C)CO WAEVWDZKMBQDEJ-UHFFFAOYSA-N 0.000 description 1
- SCVJRXQHFJXZFZ-KVQBGUIXSA-N 2-amino-9-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-3h-purine-6-thione Chemical compound C1=2NC(N)=NC(=S)C=2N=CN1[C@H]1C[C@H](O)[C@@H](CO)O1 SCVJRXQHFJXZFZ-KVQBGUIXSA-N 0.000 description 1
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 1
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 description 1
- AKRJXOYALOGLHQ-UHFFFAOYSA-N 2-methylheptan-4-one Chemical compound CCCC(=O)CC(C)C AKRJXOYALOGLHQ-UHFFFAOYSA-N 0.000 description 1
- IFVLZKSUYNTRHJ-UHFFFAOYSA-N 2-methylpropyl 2,2-dimethylpropanoate Chemical compound CC(C)COC(=O)C(C)(C)C IFVLZKSUYNTRHJ-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- QCAHUFWKIQLBNB-UHFFFAOYSA-N 3-(3-methoxypropoxy)propan-1-ol Chemical compound COCCCOCCCO QCAHUFWKIQLBNB-UHFFFAOYSA-N 0.000 description 1
- ULMZOZMSDIOZAF-UHFFFAOYSA-N 3-hydroxy-2-(hydroxymethyl)propanoic acid Chemical class OCC(CO)C(O)=O ULMZOZMSDIOZAF-UHFFFAOYSA-N 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000004070 6 membered heterocyclic group Chemical group 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HRPVXLWXLXDGHG-UHFFFAOYSA-N Acrylamide Chemical compound NC(=O)C=C HRPVXLWXLXDGHG-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 1
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 1
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- OKJADYKTJJGKDX-UHFFFAOYSA-N Butyl pentanoate Chemical compound CCCCOC(=O)CCCC OKJADYKTJJGKDX-UHFFFAOYSA-N 0.000 description 1
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical compound C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 description 1
- 101100433727 Caenorhabditis elegans got-1.2 gene Proteins 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 229920008712 Copo Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- VFZRZRDOXPRTSC-UHFFFAOYSA-N DMBA Natural products COC1=CC(OC)=CC(C=O)=C1 VFZRZRDOXPRTSC-UHFFFAOYSA-N 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical compound S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- ZAFNJMIOTHYJRJ-UHFFFAOYSA-N Diisopropyl ether Chemical compound CC(C)OC(C)C ZAFNJMIOTHYJRJ-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- 229910015136 FeMn Inorganic materials 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 229910005191 Ga 2 O 3 Inorganic materials 0.000 description 1
- 229910005557 Gd—Si Inorganic materials 0.000 description 1
- 229910005839 GeS 2 Inorganic materials 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical group CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 1
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 1
- WVRPFQGZHKZCEB-UHFFFAOYSA-N Isopropyl 2-methylpropanoate Chemical compound CC(C)OC(=O)C(C)C WVRPFQGZHKZCEB-UHFFFAOYSA-N 0.000 description 1
- 229910020797 La-Si Inorganic materials 0.000 description 1
- 229910018092 Li 2 S-Al 2 S 3 Inorganic materials 0.000 description 1
- 229910018127 Li 2 S-GeS 2 Inorganic materials 0.000 description 1
- 229910018133 Li 2 S-SiS 2 Inorganic materials 0.000 description 1
- 229910008745 Li2O-B2O3-P2O5 Inorganic materials 0.000 description 1
- 229910008590 Li2O—B2O3—P2O5 Inorganic materials 0.000 description 1
- 229910009293 Li2S-GeS2-Ga2S3 Inorganic materials 0.000 description 1
- 229910009290 Li2S-GeS2-P2S5 Inorganic materials 0.000 description 1
- 229910009301 Li2S-P2S5-SiS2 Inorganic materials 0.000 description 1
- 229910009324 Li2S-SiS2-Li3PO4 Inorganic materials 0.000 description 1
- 229910009326 Li2S-SiS2-Li4SiO4 Inorganic materials 0.000 description 1
- 229910009318 Li2S-SiS2-LiI Inorganic materials 0.000 description 1
- 229910009331 Li2S-SiS2-P2S5 Inorganic materials 0.000 description 1
- 229910009328 Li2S-SiS2—Li3PO4 Inorganic materials 0.000 description 1
- 229910009353 Li2S—GeS2—Al2S3 Inorganic materials 0.000 description 1
- 229910009108 Li2S—GeS2—Ga2S3 Inorganic materials 0.000 description 1
- 229910009110 Li2S—GeS2—P2S5 Inorganic materials 0.000 description 1
- 229910009102 Li2S—GeS2—Sb2S5 Inorganic materials 0.000 description 1
- 229910009130 Li2S—GeS2—ZnS Inorganic materials 0.000 description 1
- 229910009148 Li2S—Li2O—P2S5 Inorganic materials 0.000 description 1
- 229910009142 Li2S—Li3PO4—P2S5 Inorganic materials 0.000 description 1
- 229910009145 Li2S—LiI—Li2O—P2S5 Inorganic materials 0.000 description 1
- 229910009181 Li2S—LiI—P2S5 Inorganic materials 0.000 description 1
- 229910009284 Li2S—P2S5—SiS2 Inorganic materials 0.000 description 1
- 229910007282 Li2S—SiS2—Al2S3 Inorganic materials 0.000 description 1
- 229910007295 Li2S—SiS2—Li3PO4 Inorganic materials 0.000 description 1
- 229910007290 Li2S—SiS2—Li4SiO4 Inorganic materials 0.000 description 1
- 229910007289 Li2S—SiS2—LiI Inorganic materials 0.000 description 1
- 229910007298 Li2S—SiS2—P2S5 Inorganic materials 0.000 description 1
- 229910012323 Li3.5Zn0.25GeO4 Inorganic materials 0.000 description 1
- 229910012329 Li3BO3—Li2SO4 Inorganic materials 0.000 description 1
- 229910012425 Li3Fe2 (PO4)3 Inorganic materials 0.000 description 1
- 229910010640 Li6BaLa2Ta2O12 Inorganic materials 0.000 description 1
- 229910010093 LiAlO Inorganic materials 0.000 description 1
- 229910013063 LiBF 4 Inorganic materials 0.000 description 1
- 229910013184 LiBO Inorganic materials 0.000 description 1
- 229910012851 LiCoO 2 Inorganic materials 0.000 description 1
- 229910011281 LiCoPO 4 Inorganic materials 0.000 description 1
- 229910010701 LiFeP Inorganic materials 0.000 description 1
- 229910010707 LiFePO 4 Inorganic materials 0.000 description 1
- 229910013641 LiNbO 3 Inorganic materials 0.000 description 1
- 229910013716 LiNi Inorganic materials 0.000 description 1
- 229910012752 LiNi0.5Mn0.5O2 Inorganic materials 0.000 description 1
- 229910015705 LiNi0.85Co0.10 Inorganic materials 0.000 description 1
- 229910013870 LiPF 6 Inorganic materials 0.000 description 1
- 229910012305 LiPON Inorganic materials 0.000 description 1
- 229910012465 LiTi Inorganic materials 0.000 description 1
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- OHLUUHNLEMFGTQ-UHFFFAOYSA-N N-methylacetamide Chemical compound CNC(C)=O OHLUUHNLEMFGTQ-UHFFFAOYSA-N 0.000 description 1
- 229910018098 Ni-Si Inorganic materials 0.000 description 1
- 229910018529 Ni—Si Inorganic materials 0.000 description 1
- TYBCSQFBSWACAA-UHFFFAOYSA-N Nonan-4-one Chemical compound CCCCCC(=O)CCC TYBCSQFBSWACAA-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 240000007320 Pinus strobus Species 0.000 description 1
- 239000004962 Polyamide-imide Substances 0.000 description 1
- 229920000604 Polyethylene Glycol 200 Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920000265 Polyparaphenylene Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- AZFUASHXSOTBNU-UHFFFAOYSA-N Propyl 2-methylpropanoate Chemical compound CCCOC(=O)C(C)C AZFUASHXSOTBNU-UHFFFAOYSA-N 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- YZCKVEUIGOORGS-IGMARMGPSA-N Protium Chemical compound [1H] YZCKVEUIGOORGS-IGMARMGPSA-N 0.000 description 1
- VKCLPVFDVVKEKU-UHFFFAOYSA-N S=[P] Chemical compound S=[P] VKCLPVFDVVKEKU-UHFFFAOYSA-N 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- 229910020346 SiS 2 Inorganic materials 0.000 description 1
- 229910005790 SnSiO Inorganic materials 0.000 description 1
- DHXVGJBLRPWPCS-UHFFFAOYSA-N Tetrahydropyran Chemical group C1CCOCC1 DHXVGJBLRPWPCS-UHFFFAOYSA-N 0.000 description 1
- 229910010413 TiO 2 Inorganic materials 0.000 description 1
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 1
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical class C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 description 1
- 238000002441 X-ray diffraction Methods 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- GEIAQOFPUVMAGM-UHFFFAOYSA-N ZrO Inorganic materials [Zr]=O GEIAQOFPUVMAGM-UHFFFAOYSA-N 0.000 description 1
- NFOVOLLZYFKJCK-UHFFFAOYSA-J [Fe+4].[O-]P([O-])(=O)OP([O-])([O-])=O Chemical class [Fe+4].[O-]P([O-])(=O)OP([O-])([O-])=O NFOVOLLZYFKJCK-UHFFFAOYSA-J 0.000 description 1
- YWJVFBOUPMWANA-UHFFFAOYSA-H [Li+].[V+5].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O Chemical compound [Li+].[V+5].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O YWJVFBOUPMWANA-UHFFFAOYSA-H 0.000 description 1
- JFBZPFYRPYOZCQ-UHFFFAOYSA-N [Li].[Al] Chemical compound [Li].[Al] JFBZPFYRPYOZCQ-UHFFFAOYSA-N 0.000 description 1
- GLMOMDXKLRBTDY-UHFFFAOYSA-A [V+5].[V+5].[V+5].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O Chemical class [V+5].[V+5].[V+5].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O GLMOMDXKLRBTDY-UHFFFAOYSA-A 0.000 description 1
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 238000012644 addition polymerization Methods 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 125000005090 alkenylcarbonyl group Chemical group 0.000 description 1
- 125000005193 alkenylcarbonyloxy group Chemical group 0.000 description 1
- 125000004448 alkyl carbonyl group Chemical group 0.000 description 1
- 125000005196 alkyl carbonyloxy group Chemical group 0.000 description 1
- 125000005529 alkyleneoxy group Chemical group 0.000 description 1
- 125000005087 alkynylcarbonyl group Chemical group 0.000 description 1
- 125000005198 alkynylcarbonyloxy group Chemical group 0.000 description 1
- 125000004419 alkynylene group Chemical group 0.000 description 1
- 229910003481 amorphous carbon Inorganic materials 0.000 description 1
- 239000002518 antifoaming agent Substances 0.000 description 1
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 1
- 125000005129 aryl carbonyl group Chemical group 0.000 description 1
- 125000005199 aryl carbonyloxy group Chemical group 0.000 description 1
- 125000004391 aryl sulfonyl group Chemical group 0.000 description 1
- 229910052789 astatine Inorganic materials 0.000 description 1
- RYXHOMYVWAEKHL-UHFFFAOYSA-N astatine atom Chemical compound [At] RYXHOMYVWAEKHL-UHFFFAOYSA-N 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 125000001231 benzoyloxy group Chemical group C(C1=CC=CC=C1)(=O)O* 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- QNRMTGGDHLBXQZ-UHFFFAOYSA-N buta-1,2-diene Chemical compound CC=C=C QNRMTGGDHLBXQZ-UHFFFAOYSA-N 0.000 description 1
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical class C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 1
- FCDMDSDHBVPGGE-UHFFFAOYSA-N butyl 2,2-dimethylpropanoate Chemical compound CCCCOC(=O)C(C)(C)C FCDMDSDHBVPGGE-UHFFFAOYSA-N 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 159000000007 calcium salts Chemical class 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical compound C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 description 1
- 239000002041 carbon nanotube Substances 0.000 description 1
- 229910021393 carbon nanotube Inorganic materials 0.000 description 1
- 125000005708 carbonyloxy group Chemical group [*:2]OC([*:1])=O 0.000 description 1
- 239000006182 cathode active material Substances 0.000 description 1
- 230000011748 cell maturation Effects 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 235000013495 cobalt Nutrition 0.000 description 1
- GIPIUENNGCQCIT-UHFFFAOYSA-K cobalt(3+) phosphate Chemical class [Co+3].[O-]P([O-])([O-])=O GIPIUENNGCQCIT-UHFFFAOYSA-K 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 239000002482 conductive additive Substances 0.000 description 1
- 239000006258 conductive agent Substances 0.000 description 1
- 229920001940 conductive polymer Polymers 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000002788 crimping Methods 0.000 description 1
- 239000003431 cross linking reagent Substances 0.000 description 1
- 150000004292 cyclic ethers Chemical class 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- CGZZMOTZOONQIA-UHFFFAOYSA-N cycloheptanone Chemical compound O=C1CCCCCC1 CGZZMOTZOONQIA-UHFFFAOYSA-N 0.000 description 1
- PDXRQENMIVHKPI-UHFFFAOYSA-N cyclohexane-1,1-diol Chemical compound OC1(O)CCCCC1 PDXRQENMIVHKPI-UHFFFAOYSA-N 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 150000001983 dialkylethers Chemical class 0.000 description 1
- KORSJDCBLAPZEQ-UHFFFAOYSA-N dicyclohexylmethane-4,4'-diisocyanate Chemical compound C1CC(N=C=O)CCC1CC1CCC(N=C=O)CC1 KORSJDCBLAPZEQ-UHFFFAOYSA-N 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000003618 dip coating Methods 0.000 description 1
- SZXQTJUDPRGNJN-UHFFFAOYSA-N dipropylene glycol Chemical compound OCCCOCCCO SZXQTJUDPRGNJN-UHFFFAOYSA-N 0.000 description 1
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 1
- 239000002612 dispersion medium Substances 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 238000010332 dry classification Methods 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000006125 ethylsulfonyl group Chemical group 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- DWYMPOCYEZONEA-UHFFFAOYSA-L fluoridophosphate Chemical compound [O-]P([O-])(F)=O DWYMPOCYEZONEA-UHFFFAOYSA-L 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 229910003472 fullerene Inorganic materials 0.000 description 1
- XPFVYQJUAUNWIW-UHFFFAOYSA-N furfuryl alcohol Chemical compound OCC1=CC=CO1 XPFVYQJUAUNWIW-UHFFFAOYSA-N 0.000 description 1
- 239000006232 furnace black Substances 0.000 description 1
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 229910021389 graphene Inorganic materials 0.000 description 1
- 238000005087 graphitization Methods 0.000 description 1
- 239000001307 helium Substances 0.000 description 1
- 229910052734 helium Inorganic materials 0.000 description 1
- SWQJXJOGLNCZEY-UHFFFAOYSA-N helium atom Chemical compound [He] SWQJXJOGLNCZEY-UHFFFAOYSA-N 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- GNOIPBMMFNIUFM-UHFFFAOYSA-N hexamethylphosphoric triamide Chemical compound CN(C)P(=O)(N(C)C)N(C)C GNOIPBMMFNIUFM-UHFFFAOYSA-N 0.000 description 1
- XXMIOPMDWAUFGU-UHFFFAOYSA-N hexane-1,6-diol Chemical compound OCCCCCCO XXMIOPMDWAUFGU-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-M hexanoate Chemical compound CCCCCC([O-])=O FUZZWVXGSFPDMH-UHFFFAOYSA-M 0.000 description 1
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical group [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 1
- 229910000037 hydrogen sulfide Inorganic materials 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- ZFAVADMJZHASIM-UHFFFAOYSA-N hydroxymethyl butanoate Chemical compound CCCC(=O)OCO ZFAVADMJZHASIM-UHFFFAOYSA-N 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 description 1
- 238000009616 inductively coupled plasma Methods 0.000 description 1
- WBJZTOZJJYAKHQ-UHFFFAOYSA-K iron(3+) phosphate Chemical class [Fe+3].[O-]P([O-])([O-])=O WBJZTOZJJYAKHQ-UHFFFAOYSA-K 0.000 description 1
- JSLCOZYBKYHZNL-UHFFFAOYSA-N isobutyric acid butyl ester Natural products CCCCOC(=O)C(C)C JSLCOZYBKYHZNL-UHFFFAOYSA-N 0.000 description 1
- LRDFRRGEGBBSRN-UHFFFAOYSA-N isobutyronitrile Chemical compound CC(C)C#N LRDFRRGEGBBSRN-UHFFFAOYSA-N 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229940024423 isopropyl isobutyrate Drugs 0.000 description 1
- LVHBHZANLOWSRM-UHFFFAOYSA-N itaconic acid Chemical class OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 1
- 239000003350 kerosene Substances 0.000 description 1
- 239000003273 ketjen black Substances 0.000 description 1
- 229910052746 lanthanum Inorganic materials 0.000 description 1
- FZLIPJUXYLNCLC-UHFFFAOYSA-N lanthanum atom Chemical compound [La] FZLIPJUXYLNCLC-UHFFFAOYSA-N 0.000 description 1
- 239000002346 layers by function Substances 0.000 description 1
- 229910052745 lead Inorganic materials 0.000 description 1
- 229920005610 lignin Polymers 0.000 description 1
- 229910000625 lithium cobalt oxide Inorganic materials 0.000 description 1
- BFZPBUKRYWOWDV-UHFFFAOYSA-N lithium;oxido(oxo)cobalt Chemical compound [Li+].[O-][Co]=O BFZPBUKRYWOWDV-UHFFFAOYSA-N 0.000 description 1
- URIIGZKXFBNRAU-UHFFFAOYSA-N lithium;oxonickel Chemical compound [Li].[Ni]=O URIIGZKXFBNRAU-UHFFFAOYSA-N 0.000 description 1
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 238000007578 melt-quenching technique Methods 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 239000007769 metal material Substances 0.000 description 1
- 125000005395 methacrylic acid group Chemical group 0.000 description 1
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 1
- GYNNXHKOJHMOHS-UHFFFAOYSA-N methyl-cycloheptane Natural products CC1CCCCCC1 GYNNXHKOJHMOHS-UHFFFAOYSA-N 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- YCOWFDCCOFCZPM-UHFFFAOYSA-N n,n'-dihydroxyoxamide Chemical compound ONC(=O)C(=O)NO YCOWFDCCOFCZPM-UHFFFAOYSA-N 0.000 description 1
- YKYONYBAUNKHLG-UHFFFAOYSA-N n-Propyl acetate Natural products CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 1
- QJQAMHYHNCADNR-UHFFFAOYSA-N n-methylpropanamide Chemical compound CCC(=O)NC QJQAMHYHNCADNR-UHFFFAOYSA-N 0.000 description 1
- 125000001038 naphthoyl group Chemical group C1(=CC=CC2=CC=CC=C12)C(=O)* 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- 239000011331 needle coke Substances 0.000 description 1
- JEMDLNFQNCQAKN-UHFFFAOYSA-N nickel;oxomanganese Chemical compound [Ni].[Mn]=O JEMDLNFQNCQAKN-UHFFFAOYSA-N 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- WSGCRAOTEDLMFQ-UHFFFAOYSA-N nonan-5-one Chemical compound CCCCC(=O)CCCC WSGCRAOTEDLMFQ-UHFFFAOYSA-N 0.000 description 1
- 125000002801 octanoyl group Chemical group C(CCCCCCC)(=O)* 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 125000003566 oxetanyl group Chemical group 0.000 description 1
- 125000001820 oxy group Chemical group [*:1]O[*:2] 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- BPUBBGLMJRNUCC-UHFFFAOYSA-N oxygen(2-);tantalum(5+) Chemical compound [O-2].[O-2].[O-2].[O-2].[O-2].[Ta+5].[Ta+5] BPUBBGLMJRNUCC-UHFFFAOYSA-N 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 238000005192 partition Methods 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000011301 petroleum pitch Substances 0.000 description 1
- 238000005191 phase separation Methods 0.000 description 1
- 125000006678 phenoxycarbonyl group Chemical group 0.000 description 1
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 1
- 125000003170 phenylsulfonyl group Chemical group C1(=CC=CC=C1)S(=O)(=O)* 0.000 description 1
- DPTATFGPDCLUTF-UHFFFAOYSA-N phosphanylidyneiron Chemical class [Fe]#P DPTATFGPDCLUTF-UHFFFAOYSA-N 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003018 phosphorus compounds Chemical class 0.000 description 1
- 230000010399 physical interaction Effects 0.000 description 1
- 239000011295 pitch Substances 0.000 description 1
- IUGYQRQAERSCNH-UHFFFAOYSA-N pivalic acid Chemical compound CC(C)(C)C(O)=O IUGYQRQAERSCNH-UHFFFAOYSA-N 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229910052699 polonium Inorganic materials 0.000 description 1
- HZEBHPIOVYHPMT-UHFFFAOYSA-N polonium atom Chemical compound [Po] HZEBHPIOVYHPMT-UHFFFAOYSA-N 0.000 description 1
- 229920002587 poly(1,3-butadiene) polymer Polymers 0.000 description 1
- 229920000162 poly(ureaurethane) Polymers 0.000 description 1
- 229920001197 polyacetylene Polymers 0.000 description 1
- 229920001281 polyalkylene Polymers 0.000 description 1
- 229920002312 polyamide-imide Polymers 0.000 description 1
- 229920000767 polyaniline Polymers 0.000 description 1
- 229920001748 polybutylene Polymers 0.000 description 1
- 238000012643 polycondensation polymerization Methods 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920001195 polyisoprene Polymers 0.000 description 1
- 239000005518 polymer electrolyte Substances 0.000 description 1
- 239000003505 polymerization initiator Substances 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 229920000123 polythiophene Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- PMFKTHJAJBPRNM-UHFFFAOYSA-N propan-2-yl 2,2-dimethylpropanoate Chemical compound CC(C)OC(=O)C(C)(C)C PMFKTHJAJBPRNM-UHFFFAOYSA-N 0.000 description 1
- FVSKHRXBFJPNKK-UHFFFAOYSA-N propionitrile Chemical compound CCC#N FVSKHRXBFJPNKK-UHFFFAOYSA-N 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940090181 propyl acetate Drugs 0.000 description 1
- HUAZGNHGCJGYNP-UHFFFAOYSA-N propyl butyrate Chemical compound CCCOC(=O)CCC HUAZGNHGCJGYNP-UHFFFAOYSA-N 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 238000010526 radical polymerization reaction Methods 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- 229920005604 random copolymer Polymers 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000007151 ring opening polymerisation reaction Methods 0.000 description 1
- 229910052707 ruthenium Inorganic materials 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- 239000011863 silicon-based powder Substances 0.000 description 1
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- DVQHRBFGRZHMSR-UHFFFAOYSA-N sodium methyl 2,2-dimethyl-4,6-dioxo-5-(N-prop-2-enoxy-C-propylcarbonimidoyl)cyclohexane-1-carboxylate Chemical compound [Na+].C=CCON=C(CCC)[C-]1C(=O)CC(C)(C)C(C(=O)OC)C1=O DVQHRBFGRZHMSR-UHFFFAOYSA-N 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 229910052979 sodium sulfide Inorganic materials 0.000 description 1
- GRVFOGOEDUUMBP-UHFFFAOYSA-N sodium sulfide (anhydrous) Chemical compound [Na+].[Na+].[S-2] GRVFOGOEDUUMBP-UHFFFAOYSA-N 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 238000004611 spectroscopical analysis Methods 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 238000013112 stability test Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- FDDDEECHVMSUSB-UHFFFAOYSA-N sulfanilamide Chemical group NC1=CC=C(S(N)(=O)=O)C=C1 FDDDEECHVMSUSB-UHFFFAOYSA-N 0.000 description 1
- 150000004763 sulfides Chemical class 0.000 description 1
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 1
- 239000003115 supporting electrolyte Substances 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 238000004381 surface treatment Methods 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 229910001936 tantalum oxide Inorganic materials 0.000 description 1
- 229910052714 tellurium Inorganic materials 0.000 description 1
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical compound [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 125000004149 thio group Chemical group *S* 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 229910000319 transition metal phosphate Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- PZJJKWKADRNWSW-UHFFFAOYSA-N trimethoxysilicon Chemical group CO[Si](OC)OC PZJJKWKADRNWSW-UHFFFAOYSA-N 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 description 1
- 238000007740 vapor deposition Methods 0.000 description 1
- 229920001567 vinyl ester resin Chemical class 0.000 description 1
- KAKZBPTYRLMSJV-UHFFFAOYSA-N vinyl-ethylene Natural products C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 1
- 238000010333 wet classification Methods 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0568—Liquid materials characterised by the solutes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0561—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of inorganic materials only
- H01M10/0562—Solid materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/052—Li-accumulators
- H01M10/0525—Rocking-chair batteries, i.e. batteries with lithium insertion or intercalation in both electrodes; Lithium-ion batteries
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0567—Liquid materials characterised by the additives
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/056—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes
- H01M10/0564—Accumulators with non-aqueous electrolyte characterised by the materials used as electrolytes, e.g. mixed inorganic/organic electrolytes the electrolyte being constituted of organic materials only
- H01M10/0566—Liquid materials
- H01M10/0569—Liquid materials characterised by the solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/058—Construction or manufacture
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/05—Accumulators with non-aqueous electrolyte
- H01M10/058—Construction or manufacture
- H01M10/0585—Construction or manufacture of accumulators having only flat construction elements, i.e. flat positive electrodes, flat negative electrodes and flat separators
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M10/00—Secondary cells; Manufacture thereof
- H01M10/42—Methods or arrangements for servicing or maintenance of secondary cells or secondary half-cells
- H01M10/4235—Safety or regulating additives or arrangements in electrodes, separators or electrolyte
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/13—Electrodes for accumulators with non-aqueous electrolyte, e.g. for lithium-accumulators; Processes of manufacture thereof
- H01M4/139—Processes of manufacture
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/027—Negative electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M2004/026—Electrodes composed of, or comprising, active material characterised by the polarity
- H01M2004/028—Positive electrodes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0017—Non-aqueous electrolytes
- H01M2300/0025—Organic electrolyte
- H01M2300/0028—Organic electrolyte characterised by the solvent
- H01M2300/0037—Mixture of solvents
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M2300/00—Electrolytes
- H01M2300/0088—Composites
- H01M2300/0091—Composites in the form of mixtures
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/386—Silicon or alloys based on silicon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/387—Tin or alloys based on tin
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/48—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides
- H01M4/52—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron
- H01M4/525—Selection of substances as active materials, active masses, active liquids of inorganic oxides or hydroxides of nickel, cobalt or iron of mixed oxides or hydroxides containing iron, cobalt or nickel for inserting or intercalating light metals, e.g. LiNiO2, LiCoO2 or LiCoOxFy
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Definitions
- the present invention relates to an inorganic solid electrolyte-containing composition, an all-solid-state secondary battery sheet, an all-solid-state secondary battery electrode sheet and an all-solid-state secondary battery, and an all-solid-state secondary battery sheet and an all-solid-state secondary battery. Regarding the manufacturing method.
- the negative electrode, the electrolyte, and the positive electrode are all made of solid, and the safety and reliability, which are the problems of the battery using the organic electrolytic solution, can be greatly improved. It is also said that it will be possible to extend the service life. Further, the all-solid-state secondary battery can have a structure in which electrodes and electrolytes are directly arranged side by side and arranged in series. Therefore, it is possible to increase the energy density as compared with a secondary battery using an organic electrolytic solution, and it is expected to be applied to an electric vehicle, a large storage battery, or the like.
- any of the constituent layers is formed by using an inorganic solid electrolyte or an active material and a binder (binding agent). It has been proposed to form with a material containing (consisting layer forming material).
- Patent Document 1 includes a solid electrolyte, a non-polar solvent-insoluble first binder, and a non-polar solvent-soluble second binder, and the first binder and the second binder. Solid electrolyte mixed solutions having different SP values are described.
- Patent Document 2 describes an electrode active material, a conductive agent, a binder (solubility parameter is less than 12 (cal / cm 3 ) 1/2 ), and a solubility parameter of 12 to 17 (cal / cm 3 ) 1.
- An electrode composition containing a polymer substance of / 2 is described.
- the constituent layer forming material When forming a constituent layer of an all-solid secondary battery with solid particles (inorganic solid electrolyte, active material, conductive auxiliary agent, etc.), the constituent layer forming material is excellently dispersed by dispersing the solid particles in an organic solvent or the like. It is desirable to show sex. However, in general, even if the constituent layer-forming material exhibits excellent dispersibility immediately after preparation, the reaggregation or sedimentation of solid particles gradually progresses with time, and the dispersibility tends to decrease. In particular, in terms of further sophistication of the required characteristics of the all-solid-state secondary battery and flexibility of manufacturing conditions, not only the initial dispersibility of the constituent layer forming material is improved, but also the decrease in dispersibility over time is suppressed. There is an increasing demand for high dispersion stability. In such a situation, the conventional constituent layer forming material is not sufficient not only in initial dispersibility but also in dispersion stability, and there is room for improvement.
- the present inventors set the adsorption rate for the inorganic solid electrolyte to a specific value different from each other.
- the inorganic solid electrolyte can be highly dispersed in the organic solvent (initial stage).
- an inorganic solid electrolyte-containing composition capable of maintaining a high degree of dispersibility (excellent in dispersion stability) can be prepared by suppressing reaggregation or precipitation of the inorganic solid electrolyte over time (excellent in dispersibility). Furthermore, it has been found that this inorganic solid electrolyte-containing composition can be suitably used as a material for forming a constituent layer of an all-solid secondary battery or the like because it exhibits excellent initial dispersibility and dispersion stability.
- the present invention has been further studied based on these findings and has been completed.
- An inorganic solid electrolyte-containing composition containing an inorganic solid electrolyte having conductivity of a metal belonging to Group 1 or Group 2 of the periodic table, a binder, and an organic solvent.
- the binder contains at least two kinds of binder A and binder B having different adsorption rates for the inorganic solid electrolyte.
- An inorganic solid electrolyte-containing composition having an adsorption rate of binder A of 15% or more and an adsorption rate of binder B of less than 10%.
- ⁇ 2> The inorganic solid electrolyte-containing composition according to ⁇ 1>, wherein the adsorption rate of the binder A is 50% or more and the adsorption rate of the binder B is less than 5%.
- ⁇ 3> The inorganic solid electrolyte-containing composition according to ⁇ 1> or ⁇ 2>, wherein at least one of the binder A and the binder B is in the form of particles.
- ⁇ 4> The inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 3>, wherein the binder A is in the form of particles.
- ⁇ 5> The inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 4>, wherein the solubility of the binder A in a non-polar solvent is 1% by mass or less.
- ⁇ 6> The inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 5>, wherein the solubility of the binder B in a non-polar solvent is 2% by mass or more.
- ⁇ 7> In any one of ⁇ 1> to ⁇ 6>, the content of the binder A in the composition containing the inorganic solid electrolyte is higher than the content of the binder B in the composition containing the inorganic solid electrolyte.
- the inorganic solid electrolyte-containing composition according to the above.
- ⁇ 8> The composition containing an inorganic solid electrolyte according to any one of ⁇ 1> to ⁇ 7>, wherein the polymer forming the binder B is a fluorine-based polymer, a hydrocarbon-based polymer, polyurethane, or a (meth) acrylic polymer.
- Stuff. ⁇ 9> The inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 8>, wherein the polymer forming the binder A is a polyurethane or a (meth) acrylic polymer.
- ⁇ 10> The composition containing an inorganic solid electrolyte according to any one of ⁇ 1> to ⁇ 9>, wherein the organic solvent contains at least two kinds of organic solvents.
- ⁇ 11> The inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 10>, wherein the organic solvent contains at least one organic solvent having a ClogP value of 1.5 or more and less than 3.
- the organic solvent may be any one of ⁇ 1> to ⁇ 11>, which comprises an organic solvent having a ClogP value of 3 or more and less than 6 and an organic solvent having a ClogP value of 1.5 or more and less than 3.
- the inorganic solid electrolyte-containing composition according to.
- ⁇ 15> The inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 14>, which contains an active material.
- ⁇ 16> The inorganic solid electrolyte-containing composition according to ⁇ 15>, wherein the adsorption rate of the binder A to the active material is 10% or more.
- ⁇ 17> The inorganic solid electrolyte-containing composition according to ⁇ 15> or ⁇ 16>, wherein the peel strength of the binder B with respect to the current collector is 0.1 N / mm or more.
- An all-solid-state secondary battery sheet having a layer composed of the inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 17> above.
- An electrode sheet for an all-solid secondary battery having an active material layer composed of the composition containing the inorganic solid electrolyte according to any one of ⁇ 15> to ⁇ 17> above.
- An all-solid-state secondary battery including a positive electrode active material layer, a solid electrolyte layer, and a negative electrode active material layer in this order.
- At least one layer of the positive electrode active material layer, the solid electrolyte layer, and the negative electrode active material layer is a layer composed of the inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 17>.
- Solid secondary battery. ⁇ 21> A method for producing a sheet for an all-solid secondary battery, which forms a film of the inorganic solid electrolyte-containing composition according to any one of ⁇ 1> to ⁇ 17> above.
- ⁇ 22> A method for manufacturing an all-solid-state secondary battery, wherein the all-solid-state secondary battery is manufactured through the manufacturing method according to ⁇ 21> above.
- the present invention can provide an inorganic solid electrolyte-containing composition which is excellent in initial dispersibility and dispersion stability and is suitably used as a material for forming a constituent layer of an all-solid secondary battery or the like. Further, the present invention can provide an all-solid-state secondary battery sheet, an all-solid-state secondary battery electrode sheet, and an all-solid-state secondary battery having a layer composed of the inorganic solid electrolyte-containing composition. Furthermore, the present invention can provide a sheet for an all-solid secondary battery and a method for producing an all-solid secondary battery using this inorganic solid electrolyte-containing composition.
- the numerical range represented by using "-" means a range including the numerical values before and after "-" as the lower limit value and the upper limit value.
- the indication of a compound is used to mean that the compound itself, its salt, and its ion are included.
- it is meant to include a derivative which has been partially changed, such as by introducing a substituent, as long as the effect of the present invention is not impaired.
- (meth) acrylic means one or both of acrylic and methacrylic. The same applies to (meth) acrylate.
- substituents include, for example, Substituent Z described later.
- substituents when there are a plurality of substituents, etc. indicated by specific reference numerals, or when a plurality of substituents, etc. are specified simultaneously or selectively, the respective substituents, etc. may be the same or different from each other. It means good. Further, even if it is not particularly specified, it means that when a plurality of substituents and the like are adjacent to each other, they may be connected to each other or condensed to form a ring.
- the inorganic solid electrolyte-containing composition of the present invention contains an inorganic solid electrolyte having conductivity of metal ions belonging to Group 1 or Group 2 of the periodic table, a binder, and an organic solvent.
- the inorganic solid electrolyte-containing composition of the present invention contains, as a binder, at least two kinds of binders A and B having different adsorption rates for the inorganic solid electrolytes contained in the composition.
- the binder A and the binder B each have a specific adsorption rate described later, and the difference between the adsorption rates of both binders is at least 5%.
- the binder contained in the inorganic solid electrolyte-containing composition of the present invention is such that solid particles such as an inorganic solid electrolyte (further, an active material and a conductive additive that can coexist) are contained in the inorganic solid electrolyte-containing composition (for example, coexisting). It has a function of binding inorganic solid electrolytes (inorganic solid electrolytes and active substances, active substances), and further stably dispersing them in an organic solvent. Further, the binder contained in the inorganic solid electrolyte-containing composition of the present invention binds solid particles to each other, at least in the solid electrolyte layer formed of the inorganic solid electrolyte-containing composition, and further comprises a current collector and solid particles. It functions as a binder to bind.
- solid particles such as an inorganic solid electrolyte (further, an active material and a conductive additive that can coexist) are contained in the inorganic solid electrolyte-containing composition (for example,
- solid particles can be highly dispersed in an organic solvent, and the dispersibility (initial dispersibility) immediately after preparation (for example, 24 hours after preparation) is high. Moreover, high initial dispersibility can be maintained for a long period of time by suppressing reaggregation or sedimentation of solid particles over time. The details of the reason are not yet clear, but it can be considered as follows. That is, by using the binder A showing a high adsorption rate and the binder B showing a low adsorption rate in combination with a difference of at least 5% in the adsorption rate, solid particles are bound to each binder in the organic solvent.
- the inorganic solid electrolyte-containing composition of the present invention can also firmly bind solid particles when formed as a constituent layer. Moreover, when the active material layer is formed from this inorganic solid electrolyte-containing composition, in addition to binding the solid particles to each other, the solid particles and the current collector can be firmly bound to each other.
- the constituent layer formed by using the inorganic solid electrolyte-containing composition of the present invention by improving the initial dispersibility and dispersion stability of the inorganic solid electrolyte-containing composition and further enhancing the binding property of the solid particles.
- the all-solid-state secondary battery equipped with the above exhibits excellent battery performance (cycle characteristics).
- the composition containing an inorganic solid electrolyte of the present invention is preferably a slurry in which the inorganic solid electrolyte is dispersed in an organic solvent.
- the viscosity difference ⁇ - ⁇ between the slurry viscosity ⁇ at a shear rate of 10 / s and the slurry viscosity ⁇ at a shear rate of 200 / s is 400 mPa ⁇ s or more. preferable.
- the slurry viscosity ⁇ is an index indicating the difficulty of reaggregation or sedimentation of solid particles when the inorganic solid electrolyte-containing composition is in a high viscosity state, and the slurry viscosity ⁇ is used to produce an inorganic solid electrolyte-containing composition. It is used as an index showing the ease of maintaining the (dispersion) uniformity of the inorganic solid electrolyte-containing composition when the film (particularly coating) is applied.
- the slurry viscosities ⁇ and ⁇ are values calculated by the method described in Examples.
- the viscosity difference ⁇ - ⁇ is preferably 500 mPa ⁇ s or more, more preferably 700 mPa ⁇ s or more, and more preferably 800 mPa ⁇ s or more in terms of further improving the initial dispersibility and dispersion stability. Is more preferable.
- the upper limit of the viscosity difference ⁇ - ⁇ is not particularly limited, but is preferably 100,000 mPa ⁇ s or less, and more preferably 2,000 mPa ⁇ s or less, from the viewpoint of suppressing uneven film formation.
- the slurry viscosity ⁇ and the slurry viscosity ⁇ are appropriately set within a range that satisfies the above-mentioned viscosity difference ⁇ - ⁇ , and are not particularly limited.
- the slurry viscosity ⁇ is preferably 600 to 20,000 mPa ⁇ s, and more preferably 800 to 2,500 mPa ⁇ s.
- the slurry viscosity ⁇ is preferably 100 to 3,000 mPa ⁇ s, and more preferably 200 to 1,000 mPa ⁇ s.
- the slurry viscosity can be appropriately set by changing or adjusting the solid content concentration of the inorganic solid electrolyte-containing composition, the type or content of solid particles or binder, the type of organic solvent, etc., and the dispersion conditions.
- the inorganic solid electrolyte-containing composition of the present invention is a material for forming a solid electrolyte layer or an active material layer (material for forming a constituent layer) of a sheet for an all-solid secondary battery, an electrode sheet for an all-solid secondary battery, or an all-solid secondary battery. ) Can be preferably used.
- the inorganic solid electrolyte-containing composition of the present invention is not particularly limited, but has a water content (also referred to as water content) of preferably 500 ppm or less, more preferably 200 ppm or less, and more preferably 100 ppm or less. Is more preferable, and 50 ppm or less is particularly preferable.
- a water content also referred to as water content
- the water content indicates the amount of water contained in the inorganic solid electrolyte-containing composition (mass ratio to the inorganic solid electrolyte-containing composition).
- the mixture is filtered through a 0.02 ⁇ m membrane filter and curled fisher. The value shall be the value measured using titration.
- the inorganic solid electrolyte-containing composition of the present invention contains an inorganic solid electrolyte.
- the inorganic solid electrolyte is an inorganic solid electrolyte
- the solid electrolyte is a solid electrolyte capable of transferring ions inside the solid electrolyte. Since it does not contain organic substances as the main ionic conductive material, it is an organic solid electrolyte (polymer electrolyte represented by polyethylene oxide (PEO), organic represented by lithium bis (trifluoromethanesulfonyl) imide (LiTFSI), etc. It is clearly distinguished from electrolyte salts).
- PEO polyethylene oxide
- LiTFSI lithium bis (trifluoromethanesulfonyl) imide
- the inorganic solid electrolyte is a solid in a steady state, it is usually not dissociated or liberated into cations and anions. In this respect, it is clearly distinguished from the electrolyte or the inorganic electrolyte salts (LiPF 6 , LiBF 4 , Lithium bis (fluorosulfonyl) imide (LiFSI), LiCl, etc.) that are dissociated or liberated into cations and anions in the polymer. Will be done.
- the inorganic solid electrolyte is not particularly limited as long as it has the conductivity of the ions of the metal belonging to the Group 1 or Group 2 of the periodic table, and is generally one having no electron conductivity.
- the inorganic solid electrolyte preferably has lithium ion ionic conductivity.
- a solid electrolyte material usually used for an all-solid secondary battery can be appropriately selected and used.
- examples of the inorganic solid electrolyte include (i) sulfide-based inorganic solid electrolyte, (ii) oxide-based inorganic solid electrolyte, (iii) halide-based inorganic solid electrolyte, and (iV) hydride-based solid electrolyte.
- a sulfide-based inorganic solid electrolyte is preferable from the viewpoint that a better interface can be formed between the active material and the inorganic solid electrolyte.
- the sulfide-based inorganic solid electrolyte contains a sulfur atom, has ionic conductivity of a metal belonging to Group 1 or Group 2 of the Periodic Table, and is electronically insulated. Those having sex are preferable.
- the sulfide-based inorganic solid electrolyte preferably contains at least Li, S and P as elements and has lithium ion conductivity, but other than Li, S and P may be used depending on the purpose or case. It may contain elements.
- Examples of the sulfide-based inorganic solid electrolyte include a lithium ion conductive inorganic solid electrolyte satisfying the composition represented by the following formula (S1).
- L a1 M b1 P c1 S d1 A e1 (S1)
- L represents an element selected from Li, Na and K, with Li being preferred.
- M represents an element selected from B, Zn, Sn, Si, Cu, Ga, Sb, Al and Ge.
- A represents an element selected from I, Br, Cl and F.
- a1 to e1 indicate the composition ratio of each element, and a1: b1: c1: d1: e1 satisfy 1 to 12: 0 to 5: 1: 2 to 12: 0 to 10.
- a1 is preferably 1 to 9, more preferably 1.5 to 7.5.
- b1 is preferably 0 to 3, more preferably 0 to 1.
- the d1 is preferably 2.5 to 10, more preferably 3.0 to 8.5.
- e1 is preferably 0 to 5, more preferably 0 to 3.
- composition ratio of each element can be controlled by adjusting the blending amount of the raw material compound when producing the sulfide-based inorganic solid electrolyte as described below.
- the sulfide-based inorganic solid electrolyte may be non-crystal (glass) or crystallized (glass-ceramic), or only a part thereof may be crystallized.
- Li-PS-based glass containing Li, P and S, or Li-PS-based glass ceramics containing Li, P and S can be used.
- Sulfide-based inorganic solid electrolytes include, for example, lithium sulfide (Li 2 S), phosphorus sulfide (for example, diphosphorus pentasulfide (P 2 S 5 )), simple phosphorus, simple sulfur, sodium sulfide, hydrogen sulfide, and lithium halide (for example). It can be produced by the reaction of at least two or more raw materials in sulfides of LiI, LiBr, LiCl) and the element represented by M (for example, SiS 2 , SnS, GeS 2 ).
- the ratio of Li 2 S and P 2 S 5 is, Li 2 S: at a molar ratio of P 2 S 5, preferably 60: 40 ⁇ It is 90:10, more preferably 68:32 to 78:22.
- the lithium ion conductivity can be made high.
- the lithium ion conductivity can be preferably 1 ⁇ 10 -4 S / cm or more, and more preferably 1 ⁇ 10 -3 S / cm or more. There is no particular upper limit, but it is practical that it is 1 ⁇ 10 -1 S / cm or less.
- Li 2 S-P 2 S 5 Li 2 S-P 2 S 5 -LiCl, Li 2 S-P 2 S 5 -H 2 S, Li 2 S-P 2 S 5 -H 2 S-LiCl, Li 2 S-LiI-P 2 S 5 , Li 2 S-LiI-Li 2 O-P 2 S 5 , Li 2 S-LiBr-P 2 S 5 , Li 2 S-Li 2 O-P 2 S 5 , Li 2 S-Li 3 PO 4- P 2 S 5 , Li 2 S-P 2 S 5- P 2 O 5 , Li 2 S-P 2 S 5- SiS 2 , Li 2 S-P 2 S 5- SiS 2- LiCl, Li 2 S-P 2 S 5- SnS, Li 2 S-P 2 S 5- Al 2 S 3 , Li 2 S-GeS 2 , Li 2 S-GeS 2 , Li 2 S-Ge
- the mixing ratio of each raw material does not matter.
- an amorphization method can be mentioned.
- the amorphization method include a mechanical milling method, a solution method and a melt quenching method. This is because processing at room temperature is possible and the manufacturing process can be simplified.
- the oxide-based inorganic solid electrolyte contains an oxygen atom, has ionic conductivity of a metal belonging to Group 1 or Group 2 of the Periodic Table, and is electronically insulated. Those having sex are preferable.
- the oxide-based inorganic solid electrolyte preferably has an ionic conductivity of 1 ⁇ 10 -6 S / cm or more, more preferably 5 ⁇ 10 -6 S / cm or more, and 1 ⁇ 10 -5 S / cm or more. It is particularly preferable that it is / cm or more.
- the upper limit is not particularly limited, but it is practical that it is 1 ⁇ 10 -1 S / cm or less.
- Li xa La ya TiO 3 [xa satisfies 0.3 ⁇ xa ⁇ 0.7, and ya satisfies 0.3 ⁇ ya ⁇ 0.7.
- LLT Li xb Layb Zr zb M bb mb Onb
- M bb is one or more elements selected from Al, Mg, Ca, Sr, V, Nb, Ta, Ti, Ge, In and Sn.
- Xb satisfies 5 ⁇ xb ⁇ 10, yb satisfies 1 ⁇ yb ⁇ 4, zb satisfies 1 ⁇ zb ⁇ 4, mb satisfies 0 ⁇ mb ⁇ 2, and nb satisfies 5 ⁇ nb ⁇ 20. Satisfies.); Li xc Byc M cc zc Onc (M cc is one or more elements selected from C, S, Al, Si, Ga, Ge, In and Sn.
- Xc is 0 ⁇ xc ⁇ 5 , Yc satisfies 0 ⁇ yc ⁇ 1, zc satisfies 0 ⁇ zc ⁇ 1, nc satisfies 0 ⁇ nc ⁇ 6); Li xd (Al, Ga) yd (Ti, Ge) zd Si.
- Li xf Si yf O zf (xf satisfies 1 ⁇ xf ⁇ 5, yf satisfies 0 ⁇ yf ⁇ 3 , zf satisfies 1 ⁇ zf ⁇ 10);.
- Li xg S yg O zg (xg satisfies 1 ⁇ xg ⁇ 3, yg satisfies 0 ⁇ yg ⁇ 2, zg satisfies 1 ⁇ zg ⁇ 10.
- Li 7 La 3 Zr 2 O 12 (LLZ) having a garnet-type crystal structure.
- Phosphorus compounds containing Li, P and O are also desirable.
- lithium phosphate Li 3 PO 4
- LiPON in which a part of oxygen of lithium phosphate is replaced with nitrogen
- LiPOD 1 (D 1 is preferably Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zr, Nb, Mo, Ru, Ag, Ta, W, Pt and one or more elements selected from Au) and the like.
- LiA 1 ON (A 1 is one or more elements selected from Si, B, Ge, Al, C and Ga) and the like can also be preferably used.
- the halide-based inorganic solid electrolyte contains a halogen atom, has the conductivity of an ion of a metal belonging to Group 1 or Group 2 of the Periodic Table, and has electrons. A compound having an insulating property is preferable.
- the halide-based inorganic solid electrolyte is not particularly limited, and examples thereof include compounds such as Li 3 YBr 6 and Li 3 YCl 6 described in LiCl, LiBr, LiI, ADVANCED MATERIALS, 2018, 30, 1803075. Of these, Li 3 YBr 6 and Li 3 YCl 6 are preferable.
- the hydride-based inorganic solid electrolyte contains a hydrogen atom, has ionic conductivity of a metal belonging to Group 1 or Group 2 of the Periodic Table, and is electronically insulated. A compound having a property is preferable.
- the hydride-based inorganic solid electrolyte is not particularly limited, and examples thereof include LiBH 4 , Li 4 (BH 4 ) 3 I, and 3 LiBH 4- LiCl.
- the inorganic solid electrolyte is preferably particles.
- the particle size (volume average particle size) of the inorganic solid electrolyte is not particularly limited, but is preferably 0.01 ⁇ m or more, more preferably 0.1 ⁇ m or more, and preferably 0.3 ⁇ m or more. More preferred.
- the upper limit is preferably 100 ⁇ m or less, more preferably 50 ⁇ m or less, and further preferably 10 ⁇ m or less.
- the particle size of the inorganic solid electrolyte is measured by the following procedure.
- Inorganic solid electrolyte particles are prepared by diluting a 1% by mass dispersion in a 20 mL sample bottle with water (diisobutylketone (DIBK) in the case of a water-unstable substance).
- DIBK diisobutylketone
- the diluted dispersion sample is irradiated with 1 kHz ultrasonic waves for 10 minutes, and immediately after that, it is used for the test.
- data was captured 50 times using a laser diffraction / scattering particle size distribution measuring device LA-920 (trade name, manufactured by HORIBA) at a temperature of 25 ° C. using a measuring quartz cell. Obtain the volume average particle size.
- JIS Z 8828 2013 "Particle size analysis-Dynamic light scattering method" as necessary. Five samples are prepared for each level and the average value is adopted.
- the inorganic solid electrolyte may contain one kind or two or more kinds.
- the mass (mg) (grain amount) of the inorganic solid electrolyte per unit area (cm 2 ) of the solid electrolyte layer is not particularly limited. It can be appropriately determined according to the designed battery capacity, and can be, for example, 1 to 100 mg / cm 2 .
- the amount of the inorganic solid electrolyte is preferably such that the total amount of the active material and the inorganic solid electrolyte is in the above range.
- the content of the inorganic solid electrolyte in the composition containing the inorganic solid electrolyte is not particularly limited, but is 50 at a solid content of 100% by mass in terms of dispersibility, reduction of interfacial resistance and binding property. It is preferably mass% or more, more preferably 70% by mass or more, and particularly preferably 90% by mass or more. From the same viewpoint, the upper limit is preferably 99.9% by mass or less, more preferably 99.5% by mass or less, and particularly preferably 99% by mass or less.
- the content of the inorganic solid electrolyte in the inorganic solid electrolyte-containing composition is such that the total content of the active material and the inorganic solid electrolyte is in the above range. Is preferable.
- the solid content is a component that does not disappear by volatilizing or evaporating when the composition containing an inorganic solid electrolyte is dried at 150 ° C. under a pressure of 1 mmHg for 6 hours. Say. Typically, it refers to a component other than the organic solvent described later.
- the composition containing an inorganic solid electrolyte of the present invention has at least a binder having an adsorption rate of 15% or more for the inorganic solid electrolyte (also referred to as binder A) and a binder having an adsorption rate of less than 10% for the inorganic solid electrolyte (also referred to as binder B). It may contain other binders (for example, a binder having an adsorption rate of 10% or more and less than 15% with respect to an inorganic solid electrolyte).
- the binder A, the binder B, and the other binders may be one kind or a plurality of kinds, respectively.
- the number of binders contained in the inorganic solid electrolyte-containing composition of the present invention may be 2 or more, and usually 2 to 4 are preferable, and there are two binders, one binder A and one binder B. Is more preferable.
- the adsorption rate of the binder is an index indicating the degree to which the binder is adsorbed to the inorganic solid electrolyte contained in the inorganic solid electrolyte-containing composition in which the binder is used in an organic solvent.
- the adsorption of the binder to the inorganic solid electrolyte includes not only physical adsorption but also chemical adsorption (adsorption by chemical bond formation, adsorption by transfer of electrons, etc.).
- the organic solvent usually uses DIBK and may be different from the organic solvent contained in the inorganic solid electrolyte-containing composition in which the binder is used.
- the adsorption rate with respect to the inorganic solid electrolyte having the same composition (type and content) as the composition (type and content) of the inorganic solid electrolyte in the inorganic solid electrolyte-containing composition It becomes.
- the adsorption rate for the plurality of types of binder is similarly used.
- the adsorption rate of the binder is a value calculated by the method described in Examples.
- the adsorption rate with respect to the inorganic solid electrolyte is determined by the type of polymer forming the binder (structure and composition of the polymer chain), the type or content of functional groups of the polymer, and the form of the binder (particulate binder or soluble binder). ) Etc., it can be set appropriately.
- the adsorption rate of the binder A is 15% or more, preferably 25% or more, more preferably 50% or more, still more preferably 55% or more.
- the binder A exhibits the above adsorption rate, it can be adsorbed on the inorganic solid electrolyte in the composition containing the inorganic solid electrolyte to form a binder, and the binding property of the inorganic solid electrolyte can be enhanced.
- the upper limit of the adsorption rate is not particularly limited, but is actually 99.9%, and is preferably 70% or less in terms of dispersibility.
- Binder A is not particularly limited in its solubility in a non-polar solvent (usually DIBK), and may be soluble or insoluble in a non-polar solvent.
- the binder A preferably has a solubility in a non-polar solvent of 2% by mass or less and 1% by mass or less in terms of improving the binding property and reducing the resistance of the all-solid-state secondary battery. Is more preferable.
- the binder A exhibiting such solubility is insoluble in a non-polar solvent, and is preferably a particulate binder described later in at least an inorganic solid electrolyte-containing composition.
- the solubility of the binder is a value calculated by the method described in Examples.
- the adsorption rate of the binder A to the active material is not particularly limited. From the viewpoint of further improving the binding property, it is preferably 10% or more, more preferably 20 to 99.9%, and further preferably 30 to 99%.
- the adsorption rate of the binder to the active material is an index indicating the degree to which the binder is adsorbed to the active material contained in the inorganic solid electrolyte-containing composition in which the binder is used in an organic solvent.
- the adsorption of the binder to the active material includes not only physical adsorption but also chemical adsorption (adsorption by chemical bond formation, adsorption by transfer of electrons, etc.).
- the organic solvent usually uses DIBK and may be different from the organic solvent contained in the inorganic solid electrolyte-containing composition in which the binder is used. Therefore, when the composition containing an inorganic solid electrolyte contains a plurality of types of active material, the case where a plurality of types of binders are used is the same as the adsorption rate of the binder to the inorganic solid electrolyte described above.
- the adsorption rate of the binder with respect to the active material is a value calculated by the method described in Examples.
- the adsorption rate for the active material can be appropriately set in the same manner as the adsorption rate for the inorganic solid electrolyte.
- the peel strength of the binder A with respect to the current collector is not particularly limited and is appropriately set.
- it can be set in the same range as the peel strength of the binder B described later with respect to the current collector.
- the content of the binder A in the inorganic solid electrolyte-containing composition is not particularly limited, but is 0.1 to 10.0% by mass at 100% by mass of the solid content in terms of binding property and dispersibility. Is more preferable, 0.2 to 5.0% by mass is more preferable, and 0.3 to 4.0% by mass is further preferable.
- the binder A is a particulate binder, its content is appropriately set within the above range, but the lower limit thereof is set in the composition containing an inorganic solid electrolyte in consideration of the solubility of the binder A.
- the content is preferably insoluble (the content can be maintained in the form of particles).
- the content of the binder A may be the same as or lower than the content of the binder B described later, but is preferably high.
- the difference in content between the binder A and the binder B is not particularly limited, and can be, for example, -5.0 to 5.0% by mass, and is 0. It is preferably 0.0% by mass or more, more preferably 0.0 to 4.0% by mass, still more preferably 0.2 to 2.0% by mass.
- the ratio of the contents of the binder A and the binder B is not particularly limited, but is preferably 0.5 to 30, for example, 1.2 to 1. It is more preferably 10.
- Binder B The adsorption rate of the binder B with respect to the inorganic solid electrolyte is less than 10%, preferably less than 8%, more preferably less than 5%, further preferably 4% or less, and particularly preferably 0% or more and 3% or less.
- Binder B exhibits the above adsorption rate, it is difficult to adsorb to the inorganic solid electrolyte in the composition containing the inorganic solid electrolyte, and excessive formation of aggregates can be suppressed to enhance the dispersibility.
- Binder B is not particularly limited in its solubility in a non-polar solvent (usually DIBK), and may be soluble or insoluble in a non-polar solvent.
- the binder B preferably has a solubility in a non-polar solvent of 2% by mass or more, more preferably 3% by mass or more, and 5% by mass or more in terms of dispersibility and binding property. It is more preferable to have.
- the upper limit of the solubility is not particularly limited, and can be, for example, 25% by mass, preferably 20% by mass or less.
- the binder B exhibiting such solubility is preferably a soluble binder described later in the composition containing an inorganic solid electrolyte.
- the adsorption rate of the binder B to the active material is not particularly limited, but is 0 to 99.9% in terms of binding property. It is preferably, more preferably 2 to 50%, still more preferably 3 to 25%.
- the peel strength of the binder B with respect to the current collector is not particularly limited. For example, in terms of obtaining high adhesion to the current collector, it is preferably 0.1 N / mm or more, more preferably 0.2 N / mm or more, and more preferably 0.3 N / mm or more. More preferred.
- the upper limit is not particularly limited, but for example, it is practically 10 N / mm or less, and preferably 2.0 N / mm or less.
- the current collector subject to the peel strength is not particularly limited as long as it is described later, and examples thereof include copper foil, aluminum foil, and stainless steel (SUS) foil.
- the peel strength is a value calculated in the same manner as the method described in Examples (peeling strength against copper foil) regardless of the type of current collector. In the present invention, the peel strength can be appropriately set in the same manner as the adsorption rate for the inorganic solid electrolyte.
- the content of the binder B in the inorganic solid electrolyte-containing composition is not particularly limited, but is preferably 0.01 to 15% by mass with respect to 100% by mass of the solid content in terms of dispersibility and binding property. , 0.05 to 5% by mass, more preferably 0.1 to 2.0% by mass.
- the mass of B)] is preferably in the range of 1,000 to 1. This ratio is more preferably 500 to 2, and even more preferably 100 to 10.
- binder A and binder B The combination of the binder A and the binder B is not particularly limited, but the difference between the adsorption rate of the binder A to the inorganic solid electrolyte and the adsorption rate of the binder B to the inorganic solid electrolyte in terms of binding property and dispersibility (binder).
- a combination in which the adsorption rate of A-the adsorption rate of the binder B) is larger than 5% is preferable.
- the combination of the binder A and the binder B is more preferably a combination in which the difference in adsorption rates between the two binders is 10% or more, and is 45% or more, in that the binding property and the dispersibility can be balanced in a well-balanced manner. Is even more preferable, a combination of 50% or more is further preferable, and a combination of 55% or more is particularly preferable.
- the upper limit of the difference in adsorption rate is not particularly limited and can be, for example, 80%. As a combination having a difference in each adsorption rate, the binder A and the binder B can be appropriately combined.
- a binder having an adsorption rate of 50% or more examples thereof include a combination of A and a binder B having an adsorption rate of less than 5%.
- the solubilitys of the binder A and the binder B to be used in combination are appropriately set and are not particularly limited, but from the viewpoint of reducing the resistance of the all-solid secondary battery, at least one of the binder A and the binder B is preferably a non-polar solvent. It is set to be insoluble (particulate binder), and more preferably, the binder A is set to be insoluble in a non-polar solvent, and the binder B is set to be soluble in a non-polar solvent.
- the combination of the binder A and the binder B in the polymer type will be described later.
- the polymers forming the binders A and B are not particularly limited as long as they satisfy the above adsorption rate with respect to the inorganic solid electrolyte, and are, for example, step-growth polymerization (polycondensation, polycondensation, etc.) of polyurethane, polyurea, polyamide, polyimide, polyester, polyether, polycarbonate and the like. Examples thereof include step-growth or addition condensation) polymers, and chain polymerization polymers such as fluoropolymers (fluorine-containing polymers), hydrocarbon polymers, vinyl polymers, and (meth) acrylic polymers.
- polyurethane or (meth) acrylic polymer is preferable, and polyurethane is more preferable in terms of dispersibility and binding property.
- polymer forming the binder B a fluoropolymer, a hydrocarbon polymer, a polyurethane or a (meth) acrylic polymer is preferable, and a fluoropolymer and a hydrocarbon polymer are more preferable, in terms of dispersibility and binding property.
- the combination of the binder A and the binder B include combinations of the above-mentioned preferable polymers forming each binder.
- the main chain of a polymer means a linear molecular chain in which all other molecular chains constituting the polymer can be regarded as a branched chain or a pendant with respect to the main chain. Although it depends on the mass average molecular weight of the molecular chain regarded as a branched chain or a pendant chain, the longest chain among the molecular chains constituting the polymer is typically the main chain. However, the terminal group of the polymer terminal is not included in the main chain. Further, the side chain of the polymer means a molecular chain other than the main chain, and includes a short molecular chain and a long molecular chain.
- the step-growth polymerization polymer has at least one bond of urethane bond, urea bond, amide bond, imide bond, ester bond, ether bond and carbonate bond in the main chain of the polymer.
- the polymer having such a bond include polyurethane, polyurea, polyamide, polyimide, polyester, polyether and polycarbonate polymers, and copolymers thereof.
- the copolymer may be a block copolymer having each of the above polymers as a segment, or a random copolymer in which each component constituting two or more of the above polymers is randomly bonded.
- the above-mentioned bond contained in the main chain contributes to the improvement of the binding property of solid particles and the like in the constituent layer of the all-solid-state secondary battery and the like by forming a hydrogen bond.
- the hydrogen bonds may be formed between the above bonds, or may be formed by the above bonds and other partial structures of the main chain.
- the above bonds preferably have hydrogen atoms that form hydrogen bonds (the nitrogen atoms of each bond are unsubstituted) in that hydrogen bonds can be formed with each other.
- the bond is not particularly limited as long as it is contained in the main chain of the polymer, and may be any of the modes contained in the structural unit (repeating unit) and / or the mode contained as a bond connecting different structural units. ..
- the above-mentioned bond contained in the main chain is not limited to one type, and may be two or more types.
- the binding mode of the main chain is not particularly limited, and may have two or more kinds of bonds at random, and the segmented main chain of a segment having a specific bond and a segment having another bond. It may be a chain.
- the main chain having the above bond is not particularly limited, but a main chain having at least one segment of urethane bond, urea bond, amide bond, imide bond and ester bond is preferable, and a main chain composed of polyamide, polyurea or polyurethane is preferable. Chains are more preferred, and main chains made of polyurethane are even more preferred.
- the main chain having the above bond contains two or more types (preferably 2 to 8 types, more preferably 2 types) of constituent components represented by any of the following formulas (I-1) to (I-4).
- a main chain formed by step-growth polymerization with a diamine compound that leads to is preferable.
- the combination of each component is appropriately selected according to the polymer species.
- the main chain made of polycarbonate, a configuration component formula (I-3) as a constituent or R P1 is represented by the following formula was introduced oxygen atoms at both ends of R P1 (I-2)
- Examples thereof include a main chain having a constituent component represented by the following formula (I-2) and a constituent component represented by the following formula (I-3).
- One kind of component in the combination of components means the number of kinds of components represented by any one of the following formulas, and has two kinds of components represented by one of the following formulas. However, it is not interpreted as two kinds of constituents.
- RP1 and RP2 each indicate a molecular chain having a molecular weight or mass average molecular weight of 20 or more and 200,000 or less.
- the molecular weight of this molecular chain cannot be uniquely determined because it depends on the type and the like, but for example, 30 or more is preferable, 50 or more is more preferable, 100 or more is further preferable, and 150 or more is particularly preferable.
- the upper limit is preferably 100,000 or less, more preferably 10,000 or less.
- the molecular weight of the molecular chain is measured for the starting compound before it is incorporated into the main chain of the polymer.
- the molecular chains that can be taken as RP1 and RP2 are not particularly limited, but hydrocarbon chains, polyalkylene oxide chains, polycarbonate chains or polyester chains are preferable, hydrocarbon chains or polyalkylene oxide chains are more preferable, and hydrocarbon chains. , Polyester oxide chains or polypropylene oxide chains are more preferred.
- the hydrocarbon chain that can be taken as RP1 and RP2 means a chain of hydrocarbons composed of carbon atoms and hydrogen atoms, and more specifically, at least two compounds composed of carbon atoms and hydrogen atoms. It means a structure in which an atom (for example, a hydrogen atom) or a group (for example, a methyl group) is eliminated.
- the hydrocarbon chain also includes a chain having a group containing an oxygen atom, a sulfur atom or a nitrogen atom in the chain, for example, a hydrocarbon group represented by the following formula (M2).
- M2 hydrocarbon group represented by the following formula
- This hydrocarbon chain may have a carbon-carbon unsaturated bond and may have a ring structure of an aliphatic ring and / or an aromatic ring. That is, the hydrocarbon chain may be a hydrocarbon chain composed of a hydrocarbon selected from an aliphatic hydrocarbon and an aromatic hydrocarbon.
- Such a hydrocarbon chain may satisfy the above molecular weight, and both a chain composed of a low molecular weight hydrocarbon group and a hydrocarbon chain composed of a hydrocarbon polymer (also referred to as a hydrocarbon polymer chain).
- hydrocarbon chains A low molecular weight hydrocarbon chain is a chain composed of ordinary (non-polymerizable) hydrocarbon groups, and examples of the hydrocarbon groups include aliphatic or aromatic hydrocarbon groups, and specific examples thereof.
- the aliphatic hydrocarbon group is not particularly limited, and for example, from a hydrogen-reduced product of an aromatic hydrocarbon group represented by the following formula (M2), or a partial structure of a known aliphatic diisosoane compound (for example, from isophorone). Narumoto) and the like.
- the hydrocarbon group contained in each of the constituent components of each example described later is also mentioned.
- the aromatic hydrocarbon group include a hydrocarbon group contained in each of the constituent components described below, and a phenylene group or a hydrocarbon group represented by the following formula (M2) is preferable.
- X represents a single bond, -CH 2- , -C (CH 3 ) 2- , -SO 2- , -S-, -CO- or -O-, and is a viewpoint of binding property. Therefore, -CH 2- or -O- is preferable, and -CH 2- is more preferable.
- the above-mentioned alkylene group and alkylene group exemplified here may be substituted with a substituent Z, preferably a halogen atom (more preferably a fluorine atom).
- RM2 to RM5 each represent a hydrogen atom or a substituent, and a hydrogen atom is preferable.
- the substituent which can be taken as RM2 to RM5 is not particularly limited, but for example, an alkyl group having 1 to 20 carbon atoms, an alkenyl group having 1 to 20 carbon atoms, -OR M6 , -N ( RM6 ) 2 , and so on.
- -SR M6 ( RM6 represents a substituent, preferably an alkyl group having 1 to 20 carbon atoms or an aryl group having 6 to 10 carbon atoms), a halogen atom (for example, a fluorine atom, a chlorine atom, a bromine atom). Can be mentioned.
- the ⁇ N ( RM6 ) 2 is an alkylamino group (preferably 1 to 20 carbon atoms, more preferably 1 to 6 carbon atoms) or an arylamino group (preferably 6 to 40 carbon atoms, 6 to 20 carbon atoms). More preferred).
- a hydrocarbon polymer chain may be a polymer chain in which (at least two) polymerizable hydrocarbons are polymerized, and may be a chain composed of a hydrocarbon polymer having a larger number of carbon atoms than the above-mentioned low molecular weight hydrocarbon chain.
- the chain is not particularly limited, but is preferably a chain composed of a hydrocarbon polymer composed of 30 or more, more preferably 50 or more carbon atoms.
- the upper limit of the number of carbon atoms constituting the hydrocarbon polymer is not particularly limited, and can be, for example, 3,000.
- the hydrocarbon polymer chain is preferably a chain composed of an aliphatic hydrocarbon having a main chain satisfying the above number of carbon atoms, and is composed of an aliphatic saturated hydrocarbon or an aliphatic unsaturated hydrocarbon. It is more preferable that the chain is made of a polymer (preferably an elastomer). Specific examples of the polymer include a diene polymer having a double bond in the main chain and a non-diene polymer having no double bond in the main chain.
- diene polymer examples include a styrene-butadiene copolymer, a styrene-ethylene-butadiene copolymer, a copolymer of isobutylene and isoprene (preferably butyl rubber (IIR)), a butadiene polymer, an isoprene polymer, and ethylene.
- IIR butyl rubber
- non-diene polymer include olefin polymers such as ethylene-propylene copolymer and styrene-ethylene-butylene copolymer, and hydrogen-reduced products of the above-mentioned diene polymer.
- the hydrocarbon to be a hydrocarbon chain preferably has a reactive group at its terminal, and more preferably has a polycondensable terminal reactive group.
- the polycondensation or polyaddition-capable terminal reactive group forms a group bonded to RP1 or RP2 of each of the above formulas by polycondensation or polyaddition.
- Examples of such a terminal reactive group include an isocinate group, a hydroxy group, a carboxy group, an amino group and an acid anhydride, and a hydroxy group is preferable.
- hydrocarbon polymers having terminal reactive groups examples include NISSO-PB series (manufactured by Nippon Soda), Claysol series (manufactured by Tomoe Kosan), and PolyVEST-HT series (manufactured by Ebonic) under the trade names.
- Poly-bd series manufactured by Idemitsu Kosan Co., Ltd.
- poly-ip series manufactured by Idemitsu Kosan Co., Ltd.
- EPOL manufactured by Idemitsu Kosan Co., Ltd.
- Polytail series manufactured by Mitsubishi Chemical Corporation
- polyalkylene oxide chain examples include chains composed of known polyalkylene oxides.
- the carbon number of the alkyleneoxy group is preferably 1 to 10, more preferably 1 to 6, and even more preferably 2 or 3 (polyethylene oxide chain or polypropylene oxide chain).
- the polyalkylene oxide chain may be a chain composed of one kind of alkylene oxide or a chain composed of two or more kinds of alkylene oxides (for example, a chain composed of ethylene oxide and propylene oxide).
- polycarbonate chain or polyester chain examples include known chains made of polycarbonate or polyester.
- the polyalkylene oxide chain, the polycarbonate chain, or the polyester chain each preferably has an alkyl group (preferably 1 to 12 carbon atoms, more preferably 1 to 6 carbon atoms) at the terminal.
- Polyalkylene oxide chain which can be taken as R P1 and R P2, end of the polycarbonate chain and a polyester chain, appropriately changing the constituents as R P1 and R P2 are represented by the formulas above the embeddable ordinary chemical structure be able to.
- the terminal oxygen atom of the polyalkylene oxide chain is removed and incorporated as RP1 or RP2 of the above-mentioned constituents.
- RN is a hydrogen atom, inside or at the end of the alkyl group contained in the molecular chain. It may have an alkyl group having 1 to 6 carbon atoms or an aryl group having 6 to 10 carbon atoms).
- RP1 and RP2 are divalent molecular chains, but at least one hydrogen atom is substituted with -NH-CO-, -CO-, -O-, -NH- or -N ⁇ .
- the molecular chain may be trivalent or higher.
- R P1 among the molecular chain is preferably a hydrocarbon is a chain, more preferably a hydrocarbon chain of low molecular weight, more preferably a hydrocarbon chain comprised of hydrocarbon groups aliphatic or aromatic, Hydrocarbon chains consisting of aromatic hydrocarbon groups are particularly preferred.
- RP2 is preferably a low molecular weight hydrocarbon chain (more preferably an aliphatic hydrocarbon group) or a molecular chain other than a low molecular weight hydrocarbon chain, preferably a low molecular weight hydrocarbon chain and a low molecular weight hydrocarbon chain. It is more preferable to include a molecular chain other than the hydrocarbon chain having a molecular weight.
- formula (I-3), component represented by any one of formula (I-4) and formula (I-6) are components R P2 is a hydrocarbon group chain of low molecular weight And, RP2 contains at least two kinds of constituents which are molecular chains other than low molecular weight hydrocarbon chains.
- constituent components represented by the above formula (I-1) are shown below.
- the raw material compound (diisocyanate compound) for deriving the constituent component represented by the above formula (I-1) include the diisocyanate compound represented by the formula (M1) described in International Publication No. 2018/20827. Specific examples thereof include Polymeric 4,4'-diphenylmethane diisocyanate and the like.
- the constituent component represented by the formula (I-1) and the raw material compound derived from the constituent component are not limited to those described in the following specific examples and the above documents.
- the raw material compound (carboxylic acid or its acid chloride, etc.) that derives the constituents represented by the above formula (I-2) is not particularly limited, and is described in, for example, paragraph [0074] of International Publication No. 2018/020827. , Carboxylic acid or acid chloride compounds and specific examples thereof.
- the constituents represented by the above formula (I-3) or formula (I-4) are shown below.
- the raw material compound (diol compound or diamine compound) for deriving the constituent component represented by the above formula (I-3) or formula (I-4) is not particularly limited, and for example, International Publication No. 2018 / Examples of each compound described in 020827 and specific examples thereof are given, and dihydroxyoxamid is also mentioned.
- the constituent components represented by the formula (I-3) or the formula (I-4) and the raw material compounds derived thereto are not limited to those described in the following specific examples and the above documents.
- the number of repetitions is an integer of 1 or more, and is appropriately set within a range satisfying the molecular weight or the number of carbon atoms of the molecular chain.
- R P3 represents an aromatic or aliphatic linking group (tetravalent), preferred linking group represented by any one of the following formulas (i) ⁇ (iix).
- X 1 represents a single bond or a divalent linking group.
- divalent linking group an alkylene group having 1 to 6 carbon atoms (for example, methylene, ethylene, propylene) is preferable.
- propylene 1,3-hexafluoro-2,2-propanediyl is preferable.
- RX and RY represent hydrogen atoms or substituents, respectively.
- * indicates the binding site with the carbonyl group in formula (1-5).
- the substituents can take as R X and R Y, not particularly limited, include later-described substituent Z, an alkyl group (carbon number is preferably from 1 to 12, more preferably 1 to 6, 1-3 More preferably) or an aryl group (preferably 6 to 22 carbon atoms, more preferably 6 to 14 carbon atoms, even more preferably 6 to 10 carbon atoms).
- the carboxylic acid dianhydride represented by the above formula (I-5) and the raw material compound (diamine compound) leading to the constituent components represented by the above formula (I-6) are not particularly limited, and for example, Examples thereof include the compounds described in WO2018 / 020827 and WO2015 / 046313 and specific examples thereof.
- R P1 , R P2 and R P3 may each have a substituent.
- substituent group is not particularly limited, for example, include substituents Z to be described later, the substituents which can take as R M2 are preferably exemplified.
- the polymer constituting the binder is a chain composed of a hydrocarbon group having a low molecular weight of RP2 as a constituent component represented by the formula (I-3) or the formula (I-4), preferably the formula (I-3).
- the constituent component preferably having a group containing an ether group and / or a carbonyl group, more preferably a carboxy group
- a functional group preferably having the following formula (I)
- R P2 is the component (preferably the polyalkylene oxide chain as a molecular chain and has a structure component) represented by the following formula (I-3B) It is preferable that RP2 has at least three kinds of constituents (preferably constituents represented by the following formula (I-3C)) which are the above-mentioned hydrocarbon polymer chains as molecular chains. preferable.
- RP1 is as described above.
- RP2A represents a chain consisting of a low molecular weight hydrocarbon group (preferably an aliphatic hydrocarbon group), and when it is a polymer forming a binder A, it is preferably a functional group described later. It has at least one group selected from the functional group group (I), more preferably an ether group and / or a carbonyl group, and more preferably a carboxy group. Examples thereof include bis (hydroxymethyl) acetic acid compounds such as 2,2-bis (hydroxymethyl) butyric acid.
- RP2B represents a polyalkylene oxide chain.
- RP2C represents a hydrocarbon polymer chain.
- R P2A hydrocarbon group of low molecular weight
- R P2C hydrocarbon polymer chain which can be taken as a polyalkylene oxide chain
- R P2C hydrocarbon polymer chain which can be taken as a polyalkylene oxide chain
- R P2B are respectively taken as R P2 in the above formula (I-3)
- R P2A hydrocarbon group of low molecular weight
- R P2C hydrocarbon polymer chain which can be taken as a polyalkylene oxide chain
- R P2B hydrocarbon polymer chain which can be taken as a polyalkylene oxide chain
- R P2B hydrocarbon polymer chain which can be taken as a polyalkylene oxide chain
- R P2B hydrocarbon polymer chain which can be taken as a polyalkylene oxide chain
- R P2B are respectively taken as R P2 in the above formula (I-3)
- the step-growth polymerization polymer may have components other than the components represented by the above formulas.
- a constituent component is not particularly limited as long as it can be sequentially polymerized with the raw material compound that derives the constituent component represented by each of the above formulas.
- the (total) content of the constituents represented by the above formulas (1-1) to (I-6) in the step-growth polymerization polymer is not particularly limited, but is preferably 5 to 100% by mass. It is more preferably 10 to 100% by mass, further preferably 50 to 100% by mass, and even more preferably 80 to 100% by mass.
- the upper limit of the content may be, for example, 90% by mass or less regardless of the above 100% by mass.
- the content of the constituent components other than the constituent components represented by the above formulas in the step-growth polymerization polymer is not particularly limited, but is preferably 50% by mass or less.
- the step-growth polymerization polymer has a component represented by any of the above formulas (I-1) to (I-6), its content is not particularly limited and can be set in the following range. That is, the component represented by the formula (I-1) or the formula (I-2) or the component derived from the carboxylic acid dianhydride represented by the formula (I-5) in the step-growth polymerization polymer.
- the content is not particularly limited, and is preferably 10 to 50 mol%, more preferably 20 to 50 mol%, and even more preferably 30 to 50 mol%.
- the content of the constituents represented by the formula (I-3), the formula (I-4) or the formula (I-6) in the step-growth polymerization polymer is not particularly limited and is 10 to 50 mol%. It is preferably 20 to 50 mol%, more preferably 30 to 50 mol%.
- the component in which RP2 is a chain composed of a low molecular weight hydrocarbon group (for example, represented by the above formula (I-3A)).
- the content of the constituent components) in the step-growth polymerization polymer is not particularly limited, but is preferably, for example, 0 to 50 mol%, more preferably 1 to 30 mol%, and 2 to 20 mol%. It is more preferably%, and even more preferably 4 to 10 mol%.
- the component in which RP2 is the polyalkylene oxide chain as a molecular chain (for example, represented by the above formula (I-3B)).
- the content of the component) in the step-growth polymerization polymer is not particularly limited, but is preferably, for example, 0 to 50 mol%, more preferably 10 to 45 mol%, and 20 to 43 mol%. Is more preferable.
- the component in which RP2 is the hydrocarbon polymer chain as a molecular chain for example, represented by the above formula (I-3C)
- the content of the constituent component) in the step-growth polymerization polymer is not particularly limited, but is preferably, for example, 0 to 50 mol%, more preferably 1 to 45 mol%, and 3 to 40 mol%. It is more preferably 3 to 30 mol%, further preferably 3 to 20 mol%, still more preferably 3 to 10 mol%.
- the above-mentioned content of each constituent component shall be the total content.
- the step-growth polymerization polymer can be synthesized by selecting a raw material compound by a known method according to the type of bond possessed by the main chain and subjecting the raw material compound to polyaddition or polycondensation.
- a synthesis method for example, International Publication No. 2018/151118 can be referred to.
- polyurethane, polyurea, polyamide, and polyimide polymers that can be used as step-growth polymerization polymers, in addition to those synthesized in Examples, for example, International Publication No. 2018/020827, International Publication No. 2015/046313, and further. Examples thereof include the polymers described in JP-A-2015-08480.
- a chain-growth polymerization polymer suitable as a polymer forming a binder is a polymer obtained by chain-polymerizing one or more kinds of monomers having a non-aromatic carbon-carbon double bond.
- the above-mentioned fluoropolymer, hydrocarbon polymer, vinyl polymer or (meth) acrylic polymer is preferable, and the (meth) acrylic polymer is more preferable.
- the fluoropolymer examples include polytetrafluoroethylene (PTFE), polyvinylidene fluoride (PVdF), and a copolymer of polyvinylidene fluoride and hexafluoropropylene (PVdF-HFP).
- PTFE polytetrafluoroethylene
- PVdF polyvinylidene fluoride
- PVdF-HFP a copolymer of polyvinylidene fluoride and hexafluoropropylene
- the copolymerization ratio [PVdF: HFP] (mass ratio) of PVdF and HFP is not particularly limited, but is preferably 9: 1 to 5: 5 in terms of dispersion stability, 9: 1. ⁇ 7: 3 is more preferable.
- hydrocarbon polymer examples include polyethylene, polypropylene, natural rubber, polybutadiene, polyisoprene, polystyrene, polystyrene butadiene copolymer, styrene-based thermoplastic elastomer, polybutylene, acrylonitrile butadiene copolymer, or hydrogenation thereof (hydrogenation). Chemistry) Polymers can be mentioned.
- the styrene-based thermoplastic elastomer or its hydride is not particularly limited, and for example, styrene-ethylene-butylene-styrene block copolymer (SEBS), styrene-isoprene-styrene block copolymer (SIS), hydride SIS.
- SEBS styrene-ethylene-butylene-styrene block copolymer
- SIS styrene-isoprene-styrene block copolymer
- SIS hydride SIS
- Styrene-butadiene-styrene block copolymer SBS
- hydrogenated SBS styrene-ethylene-ethylene-propylene-styrene block copolymer
- SEEPS styrene-ethylene-propylene-styrene block copolymer
- SEPS styrene-ethylene-propylene-styrene block copolymer
- SBR styrene-butadiene rubber
- HSBR hydride styrene-butadiene rubber
- the hydrocarbon polymer having no unsaturated group for example, 1,2-butadiene constituent
- vinyl-based polymer examples include polymers containing, for example, 50 mol% or more of vinyl-based monomers other than the (meth) acrylic compound (M1).
- vinyl-based monomer examples include vinyl compounds described later.
- Specific examples of the vinyl polymer include polyvinyl alcohol, polyvinyl acetal, polyvinyl acetate, and a copolymer containing these.
- this vinyl-based polymer is a constituent component derived from the (meth) acrylic compound (M1) that forms the (meth) acrylic polymer described later, and further a constituent component derived from the macromonomer described later. It is preferable to have (MM).
- the content of the constituent component derived from the vinyl-based monomer is preferably the same as the content of the constituent component derived from the (meth) acrylic compound (M1) in the (meth) acrylic polymer.
- the content of the constituent component derived from the (meth) acrylic compound (M1) is not particularly limited as long as it is less than 50 mol% in the polymer, but is preferably 0 to 40 mol%, and is preferably 5 to 35 mol%. Is more preferable.
- the content of the component (MM) is preferably the same as the content in the (meth) acrylic polymer.
- the (meth) acrylic polymer at least one (meth) acrylic compound (M1) selected from a (meth) acrylic acid compound, a (meth) acrylic acid ester compound, a (meth) acrylamide compound and a (meth) acrylic nitrile compound. ) Is (co) polymerized to obtain a polymer. Further, a (meth) acrylic polymer composed of a copolymer of the (meth) acrylic compound (M1) and another polymerizable compound (M2) is also preferable.
- the other polymerizable compound (M2) is not particularly limited, and examples thereof include vinyl compounds such as styrene compounds, vinylnaphthalene compounds, vinylcarbazole compounds, allyl compounds, vinyl ether compounds, vinyl ester compounds, and dialkyl itaconate compounds.
- Examples of the vinyl compound include "vinyl-based monomers" described in JP-A-2015-88486.
- the content of the other polymerizable compound (M2) in the (meth) acrylic polymer is not particularly limited, but can be, for example, less than 50 mol%.
- the compound represented by the following formula (b-1) is preferable.
- R 1 is a hydrogen atom, a hydroxy group, a cyano group, a halogen atom, an alkyl group (preferably having 1 to 24 carbon atoms, more preferably 1 to 12 carbon atoms, particularly preferably 1 to 6 carbon atoms), and an alkenyl group (2 carbon atoms).
- ⁇ 24 is preferred, 2-12 is more preferred, 2-6 is particularly preferred), an alkynyl group (2-24 carbon atoms is preferred, 2-12 is more preferred, 2-6 is particularly preferred), or an aryl group (preferably 2-6).
- 6 to 22 carbon atoms are preferable, and 6 to 14 carbon atoms are more preferable).
- a hydrogen atom or an alkyl group is preferable, and a hydrogen atom or a methyl group is more preferable.
- R 2 represents a hydrogen atom or a substituent.
- the substituent that can be taken as R 2 is not particularly limited, but an alkyl group (preferably having 1 to 30 carbon atoms, more preferably 1 to 24 carbon atoms, particularly preferably 1 to 12 chains, and preferably a branched chain but a straight chain).
- Alkyl group (preferably 2 to 12 carbon atoms, more preferably 2 to 6 carbon atoms), aryl group (preferably 6 to 22 carbon atoms, more preferably 6 to 14 carbon atoms), aralkyl group (preferably 7 to 23 carbon atoms, 7) ⁇ 15 is more preferable), an aliphatic heterocyclic group containing a cyano group, a hydroxy group, a sulfanyl group, and an oxygen atom (preferably having 2 to 12 carbon atoms, more preferably 2 to 6).
- an epoxy group-containing group, an oxetane group-containing group, a tetrahydrofuryl group-containing group and the like are preferable.
- L 1 is a linking group and is not particularly limited, but for example, an alkylene group having 1 to 6 carbon atoms (preferably 1 to 3), an alkenylene group having 2 to 6 carbon atoms (preferably 2 to 3), and a carbon number of carbons.
- the linking group may have any substituent.
- the number of atoms constituting the linking group and the number of linking atoms are as described later. Examples of the optional substituent include the Substituent Z described later, and examples thereof include an alkyl group and a halogen atom.
- n is 0 or 1, preferably 1. However, when ⁇ (L 1 ) n ⁇ R 2 indicates one kind of substituent (for example, an alkyl group), n is set to 0 and R 2 is used as a substituent (alkyl group).
- (meth) acrylic compound (M1) a compound represented by the following formula (b-2) or (b-3) is also preferable.
- R 1, n has the same meaning as the above formula (b-1).
- R 3 is synonymous with R 2 .
- L 2 is a linking group and has the same meaning as L 1 described above.
- L 3 is a linking group and is synonymous with L 1 , but an alkylene group having 1 to 6 carbon atoms (preferably 1 to 3) is preferable.
- m is an integer of 1 to 200, preferably an integer of 1 to 100, and more preferably an integer of 1 to 50.
- R 1 unsubstituted carbon atom
- the substituent is not particularly limited, for example, the groups that can be taken as R 1.
- the substituent is within a range that does not impair the effect of the present invention. May have.
- the substituent include a substituent Z described later and a group selected from the functional group group (I).
- a halogen atom a hydroxy group, a sulfanyl group, an acyl group, an acyloxy group, an alkoxy group, and the like.
- examples thereof include an aryloxy group, an allyloyl group and an allyloyloxy group.
- the polymer constituting the binder is a chain polymerization type polymer, preferably an addition polymerization type polymer, it is preferable to have a component (MM) derived from a macromonomer having a mass average molecular weight of 1,000 or more.
- MM component derived from a macromonomer having a mass average molecular weight of 1,000 or more.
- the mass average molecular weight of the macromonomer is preferably 2,000 or more, and more preferably 3,000 or more.
- the upper limit is preferably 500,000 or less, more preferably 100,000 or less, and particularly preferably 30,000 or less.
- the polymer constituting the binder has a component (MM) derived from a macromonomer having a mass average molecular weight in the above range, it can be more uniformly dispersed in an organic solvent, particularly a non-polar solvent.
- the mass average molecular weight of the constituent component (MM) can be identified by measuring the mass average molecular weight of the macromonomer incorporated when synthesizing the polymer constituting the binder.
- the SP value of the macromonomer is not particularly limited, but is preferably 10 or less, and more preferably 9.5 or less. There is no particular lower limit, but it is practical that it is 5 or more.
- the SP value is an index showing the property of dispersing in an organic solvent.
- the binding property with the solid particles is improved, and the affinity with the solvent is enhanced thereby stably dispersed. be able to.
- SP value unless otherwise indicated, determined by Hoy method (H.L.Hoy JOURNAL OF PAINT TECHNOLOGY Vol.42, No.541,1970,76-118, and POLYMER HANDBOOK 4 th, 59 chapters, VII page 686 See the formula below in Table5, Table6 and Table6).
- the SP value is shown with the unit omitted, but the unit is cal 1/2 cm -3/2 .
- the SP value of the component (MM) is almost the same as the SP value of the macromonomer, and may be evaluated accordingly.
- the SP value (SP P ) of the polymer is the SP value of each repeating unit constituting the polymer, SP 1 , SP 2, ...,
- the macromonomer is not particularly limited as long as it has a mass average molecular weight of 1,000 or more, but a macromonomer having a polymerized chain bonded to a polymerizable group such as a group having an ethylenically unsaturated bond is preferable.
- the polymerized chain of the macromonomer constitutes a side chain (graft chain) with respect to the main chain of the polymer.
- the polymerized chain has a function of further improving the dispersibility in an organic solvent. As a result, both suppression of interfacial resistance between solid particles and improvement of binding property can be further achieved.
- the polymerizable group contained in the macromonomer is not particularly limited, and details will be described later. Examples thereof include various vinyl groups and (meth) acryloyl groups, and (meth) acryloyl groups are preferable.
- the polymer chain contained in the macromonomer is not particularly limited, and ordinary polymer components can be applied.
- a (meth) acrylic resin chain, a polyvinyl resin chain, a polysiloxane chain, a polyalkylene ether chain, a hydrocarbon chain and the like can be mentioned, and a (meth) acrylic resin chain or a polysiloxane chain is preferable.
- the chain of the (meth) acrylic resin preferably contains a component derived from the (meth) acrylic compound selected from the (meth) acrylic acid compound, the (meth) acrylic acid ester compound and the (meth) acrylonitrile compound, and two types thereof. It may be a polymer of the above (meth) acrylic compounds.
- the (meth) acrylic compound is synonymous with the above (meth) acrylic compound (M1).
- the polysiloxane chain is not particularly limited, and examples thereof include siloxane polymers having an alkyl group or an aryl group.
- Examples of the hydrocarbon chain include the chains made of the above-mentioned hydrocarbon-based polymers.
- the macromonomer preferably has a polymerizable group represented by the following formula (b-11).
- R 11 is synonymous with R 1 . * Is the connection position.
- the macromonomer preferably has a linking group that links the polymerizable group and the polymerized chain.
- This linking group is usually incorporated into the side chain of the macromonomer.
- Linking group is not particularly limited, include groups described in the linking group L 1 in the above formula (b-1).
- the number of atoms constituting the linking group is preferably 1 to 36, more preferably 1 to 24, further preferably 1 to 12, and preferably 1 to 6.
- the number of connecting atoms of the linking group is preferably 10 or less, and more preferably 8 or less.
- the lower limit is 1 or more.
- the macromonomer preferably has a polymerizable moiety represented by any of the following formulas (b-12a) to (b-12c).
- R b2 is synonymous with R 1 .
- RN2 is synonymous with RN1 described later.
- the benzene ring of the formula (b-12c) may be substituted with any substituent Z.
- the structural part existing at the end of the bond position of * is not particularly limited as long as it satisfies the molecular weight as a macromonomer, but the above-mentioned polymerized chain (preferably may be bonded via a linking group) is preferable.
- the linking group and the polymerized chain may each have a substituent Z, for example, a halogen atom (fluorine atom) or the like.
- the carbon atom to which R 11 or R b2 is not bonded is represented as an unsubstituted carbon atom, but as described above, it may have a substituent.
- the substituent is not particularly limited, for example, the groups that can be taken as R 1.
- the macromonomer is preferably a compound represented by the following formula (b-13a).
- R b2 is synonymous with R 1 .
- na is not particularly limited, but is preferably an integer of 1 to 6, more preferably 1 or 2, and even more preferably 1.
- Ra represents a substituent when na is 1, and a linking group when na is 2 or more.
- the substituent that can be taken as Ra is not particularly limited, but the above-mentioned polymerized chain is preferable, and the (meth) acrylic resin chain or the polysiloxane chain is more preferable.
- Ra may be directly bonded to the oxygen atom (—O—) in the formula (b-13a), or may be bonded via a linking group.
- the linking group is not particularly limited, and examples thereof include the above-mentioned linking group that links the polymerizable group and the polymerized chain.
- the linking group is not particularly limited, but for example, an alcan linking group having 1 to 30 carbon atoms, a cycloalcan linking group having 3 to 12 carbon atoms, and an aryl linking group having 6 to 24 carbon atoms.
- R is a hydrogen atom or an alkyl group having 1 to 6 carbon atoms
- a silylene group (-Si (R Si) 2- : R Si represents a hydrogen atom or an alkyl group having 1 to 6 carbon atoms), a carbonyl group and an imino group
- RN1 represents a hydrogen atom or a substituent, preferably a hydrogen atom and 1 to 6 carbon atoms.
- An alkyl group of 6 or an aryl group having 6 to 10 carbon atoms), or a combination thereof is preferable.
- macromonomers other than the above-mentioned macromonomers include "macromonomers (X)" described in JP-A-2015-88486.
- the (meth) acrylic polymer is a component derived from the (meth) acrylic compound (M1), a component derived from the vinyl compound (M2), a component derived from the macromonomer (MM), and a compound that derives these components. It can contain other polymerizable constituents, preferably a constituent derived from a (meth) acrylic compound (M1), and more preferably a constituent (MM) derived from a macromonomer, preferably a vinyl compound (M2). Derived constituents and other constituents may be included.
- the content of the component derived from the (meth) acrylic compound (M1) in the (meth) acrylic polymer is not particularly limited, but is preferably 1 to 99.9 mol%, preferably 5 to 99 mol%. It is more preferably present, and particularly preferably 10 to 99 mol%.
- the content of the component (MM) in the (meth) acrylic polymer is not particularly limited, but is preferably 0 to 60 mol%, more preferably 0.2 to 50 mol%, and 0. It is more preferably 5 to 40 mol%. When the content is in the above range, the dispersibility of the inorganic solid electrolyte-containing composition, the binding property between solid particles, and the ionic conductivity can be exhibited at a high level.
- the content of the constituent component derived from the vinyl compound (M2) in the (meth) acrylic polymer is not particularly limited, but is preferably 0 to 30 mol%, more preferably 0 to 20 mol%. , 0-10 mol% is particularly preferable.
- the (meth) acrylic polymer has a functional group described later, any of the above-mentioned constituent components may have a functional group, and the content of the constituent component having a functional group is as described later, but the above-mentioned content Also meet the amount.
- the polymer forming the binder preferably has a functional group for enhancing the wettability or adsorptivity of solid particles such as an inorganic solid electrolyte to the surface, especially when applied as a polymer forming the binder A.
- a functional group include a group that exhibits a physical interaction such as a hydrogen bond on the surface of the solid particle and a group that can form a chemical bond with a group existing on the surface of the solid particle. It is more preferable to have at least one group selected from the following functional group group (I).
- the sulfonic acid group and the phosphoric acid group may be salts thereof, and examples thereof include sodium salts and calcium salts.
- the alkoxysilyl group may be a silyl group in which the Si atom is substituted with at least one alkoxy group (preferably having 1 to 12 carbon atoms), and other substituents on the Si atom include an alkyl group and an aryl. Group etc. can be mentioned.
- the alkoxysilyl group for example, the description of the alkoxysilyl group in the substituent Z described later can be preferably applied.
- the group having a condensed ring structure of 3 or more rings is preferably a group having a cholesterol ring structure or a group having a condensed ring structure of 3 or more aromatic rings, and a cholesterol residue or a pyrenyl group is more preferable.
- Carboxy group, a sulfonic acid group (-SO 3 H), phosphoric acid group (-PO 4 H 2), hydroxy group and an alkoxysilyl group has a high adsorptivity of the inorganic solid electrolyte or the cathode active material, 3 or more rings condensed
- a group having a ring structure has high adsorptivity with a negative electrode active material or the like.
- the amino group (-NH 2 ), sulfanil group and isocyanato group have high adsorptivity with the inorganic solid electrolyte.
- the polymer forming the binder may have a functional group selected from the functional group group (I) in any of the constituent components forming the polymer, and may be present in either the main chain or the side chain of the polymer. You may be doing it.
- the content of the functional group selected from the functional group group (I) in the polymer forming the binder A is not particularly limited, but for example, the constituent component having the functional group selected from the functional group group (I).
- the ratio of the total constituent components constituting the polymer forming the binder is preferably 0.01 to 50 mol%, preferably 0.02 to 49 mol%, more preferably 0.1 to 40 mol%, and 1 to 1 to 40 mol%. 30 mol% is more preferable, and 3 to 25 mol% is particularly preferable.
- the polymer forming the binder when applied as the polymer forming the binder B, it may or may not have the above functional groups as long as it satisfies the adsorption rate in the above range.
- the content of the functional group selected from the functional group group (I) in the polymer forming the binder B is not particularly limited, but the binder of the constituent component having the functional group selected from the functional group group (I).
- the proportion of the total constituent components constituting the polymer forming the polymer is preferably 20 mol% or less, preferably 5 mol% or less, more preferably 1 mol% or less, still more preferably 0.7 mol% or less.
- the polymer (each constituent and raw material compound) forming the binder may have a substituent.
- the substituent is not particularly limited, but preferably, a group selected from the following substituent Z can be mentioned.
- -Substituent Z- Alkyl groups preferably alkyl groups having 1 to 20 carbon atoms, such as methyl, ethyl, isopropyl, t-butyl, pentyl, heptyl, 1-ethylpentyl, benzyl, 2-ethoxyethyl, 1-carboxymethyl, etc.
- alkenyl groups preferably alkyl groups having 1 to 20 carbon atoms, such as methyl, ethyl, isopropyl, t-butyl, pentyl, heptyl, 1-ethylpentyl, benzyl, 2-ethoxyethyl, 1-carboxymethyl, etc.
- an alkenyl group having 2 to 20 carbon atoms for example, vinyl, allyl, oleyl, etc.
- an alkynyl group preferably an alkynyl group having 2 to 20 carbon atoms, for example, ethynyl, butadynyl, phenylethynyl, etc.
- a cycloalkyl group having 3 to 20 carbon atoms for example, cyclopropyl, cyclopentyl, cyclohexyl, 4-methylcyclohexyl, etc., is usually used in the present specification to include a cycloalkyl group.
- An aryl group (preferably an aryl group having 6 to 26 carbon atoms, for example, phenyl, 1-naphthyl, 4-methoxyphenyl, 2-chlorophenyl, 3-methylphenyl, etc.), an aralkyl group (preferably having 7 carbon atoms).
- ⁇ 23 aralkyl groups eg, benzyl, phenethyl, etc.
- heterocyclic groups preferably heterocyclic groups having 2 to 20 carbon atoms, more preferably 5 or 5 having at least one oxygen atom, sulfur atom, nitrogen atom. It is a 6-membered heterocyclic group.
- the heterocyclic group includes an aromatic heterocyclic group and an aliphatic heterocyclic group.
- a tetrahydropyran ring group for example, a tetrahydropyran ring group, a tetrahydrofuran ring group, 2-pyridyl, 4-pyridyl, 2-. Imidazolyl, 2-benzoimidazolyl, 2-thiazolyl, 2-oxazolyl, pyrrolidone group, etc.), alkoxy group (preferably an alkoxy group having 1 to 20 carbon atoms, for example, methoxy, ethoxy, isopropyloxy, benzyloxy, etc.), aryloxy group (Preferably, an aryloxy group having 6 to 26 carbon atoms, for example, phenoxy, 1-naphthyloxy, 3-methylphenoxy, 4-methoxyphenoxy, etc., is used in the present specification to include an aryloxy group.
- alkoxy group preferably an alkoxy group having 1 to 20 carbon atoms, for example, methoxy, ethoxy, is
- a heterocyclic oxy group (a group in which an —O— group is bonded to the heterocyclic group), an alkoxycarbonyl group (preferably an alkoxycarbonyl group having 2 to 20 carbon atoms, for example, ethoxycarbonyl, 2-ethylhexyloxycarbonyl).
- aryloxycarbonyl groups preferably aryloxycarbonyl groups with 6-26 carbon atoms, such as phenoxycarbonyl, 1-naphthyloxycarbonyl, 3-me It contains a tylphenoxycarbonyl, 4-methoxyphenoxycarbonyl, etc.
- an amino group preferably an amino group having 0 to 20 carbon atoms, an alkylamino group, an arylamino group, for example, amino (-NH 2 ), N, N-dimethyl.
- Sulfamoyl group (preferably a sulfamoyl group having 0 to 20 carbon atoms, for example, N, N-dimethylsulfamoyl, N-phenylsulfamoyl, etc.)
- Acyl group alkylcarbonyl group, alkenylcarbonyl group, alkynylcarbonyl group, arylcarbonyl group, heterocyclic carbonyl group, preferably acyl group having 1 to 20 carbon atoms, for example, acetyl, propionyl, butyryl, octanoyl, hexadeca.
- acyloxy groups (alkylcarbonyloxy groups, alkenylcarbonyloxy groups, alkynylcarbonyloxy groups, arylcarbonyloxy groups, heterocyclic carbonyloxy groups, etc., preferably carbon.
- acyloxy groups such as acetyloxy, propionyloxy, butyryloxy, octanoyloxy, hexadecanoyloxy, acryloyloxy, methacryloxy, crotonoyloxy, benzoyloxy, naphthoyloxy, nicotineoloxy, etc.), Allyloxy group (preferably an allyloxy group having 7 to 23 carbon atoms, for example, benzoyloxy, etc.), a carbamoyl group (preferably a carbamoyl group having 1 to 20 carbon atoms, for example, N, N-dimethylcarbamoyl, N- Phenylcarbamoyl, etc.), acylamino groups (preferably acylamino groups having 1 to 20 carbon atoms, such as acetylamino, benzoylamino, etc.), alkylthio groups (preferably alkylthio groups having 1 to 20 carbon atoms
- arylthio groups preferably arylthio groups having 6 to 26 carbon atoms, for example, phenylthio, 1-naphthylthio, 3-methylphenylthio, 4-methoxyphenylthio, etc.
- heterocyclic thio groups the above heterocycle.
- a group having an —S— group bonded to the group an alkylsulfonyl group (preferably an alkylsulfonyl group having 1 to 20 carbon atoms, for example, methylsulfonyl, ethylsulfonyl, etc.), an arylsulfonyl group (preferably having 6 to 22 carbon atoms).
- Aryll sul Honyl groups such as benzenesulfonyl, alkylsilyl groups (preferably alkylsilyl groups having 1 to 20 carbon atoms, such as monomethylsilyl, dimethylsilyl, trimethylsilyl, triethylsilyl, etc.), arylsilyl groups (preferably 6 carbon atoms).
- Arylsilyl groups of ⁇ 42 such as triphenylsilyl
- alkoxysilyl groups preferably alkoxysilyl groups having 1 to 20 carbon atoms, such as monomethoxysilyl, dimethoxysilyl, trimethoxysilyl, triethoxysilyl, etc.
- An aryloxysilyl group preferably an aryloxysilyl group having 6 to 42 carbon atoms, for example, triphenyloxysilyl group
- R P 2
- a phosphinyl group preferably a phosphinyl group having 0 to 20 carbon atoms,
- -P (R P) 2) a sulfo group (sulfonic acid group), and carboxy groups, hydroxy group, sulfanyl group, a cyano group, a halogen atom (e.g. fluorine atom, a chlorine atom, a bromine atom, an iodine atom) of Be done.
- a halogen atom e.g. fluorine atom, a chlorine atom, a bromine atom, an iodine atom
- RP is a hydrogen atom or a substituent (preferably a group selected from the substituent Z). Further, each group listed in these substituents Z may be further substituted with the above-mentioned substituent Z.
- the alkyl group, alkylene group, alkenyl group, alkenylene group, alkynyl group and / or alkynylene group and the like may be cyclic or chain-like, or may be linear or branched.
- the polymer forming the binder may be a non-crosslinked polymer or a crosslinked polymer. Further, when the cross-linking of the polymer proceeds by heating or application of a voltage, the molecular weight may be larger than the above molecular weight. Preferably, the polymer has a mass average molecular weight in the above range at the start of use of the all-solid-state secondary battery.
- the binder may be soluble in an organic solvent or may be insoluble (particles) in an organic solvent.
- insoluble in an organic solvent means that the solubility in a non-polar solvent calculated by the method described in Examples is 1% by mass or less, and is soluble in an organic solvent. Means that the solubility in a non-polar solvent calculated by the method described in Examples is more than 1% by mass, preferably 2% by mass or more.
- the binder may be present in the composition containing an inorganic solid electrolyte, for example, dissolved in an organic solvent (the dissolved binder is referred to as a dissolved binder), and is not dissolved in the organic solvent and exists in a solid state.
- the binder A is a particulate binder in the composition containing an inorganic solid electrolyte, and further in the solid electrolyte layer or the active material layer (coating and drying layer), in terms of dispersibility and battery characteristics, and further, the battery. It is preferable in terms of resistance.
- the binder B is preferably a soluble binder in the composition containing an inorganic solid electrolyte from the viewpoint of dispersibility.
- the binder is a particulate binder
- its shape is not particularly limited and may be flat, amorphous or the like, but spherical or granular is preferable.
- the particle size of the particulate binder is not particularly limited, but is preferably 1000 nm or less, more preferably 500 nm or less, and further preferably 300 nm or less.
- the lower limit is 1 nm or more, preferably 5 nm or more, more preferably 10 nm or more, and further preferably 50 nm or more.
- the average particle size of the particulate binder can be measured in the same manner as the average particle size of the inorganic solid electrolyte.
- the particle size of the particulate binder in the constituent layers of the all-solid secondary battery is measured in advance by, for example, disassembling the battery and peeling off the constituent layer containing the particulate binder, and then measuring the constituent layers.
- the measurement can be performed by excluding the measured value of the particle size of the particles other than the particulate binder.
- the particle size of the particulate binder can be adjusted, for example, by the type of organic solvent, the content and content of constituents in the polymer, and the like.
- the water concentration of the binder is preferably 100 ppm (mass basis) or less.
- the polymer may be crystallized and dried, or the binder dispersion may be used as it is.
- the polymer forming the binder is preferably amorphous.
- the term "amorphous" as a polymer typically means that no endothermic peak due to crystal melting is observed when measured at the glass transition temperature.
- the mass average molecular weight of the polymer forming the binder is not particularly limited. For example, 15,000 or more is preferable, 30,000 or more is more preferable, and 50,000 or more is further preferable. As the upper limit, 5,000,000 or less is practical, but 4,000,000 or less is preferable, and 3,000,000 or less is more preferable.
- the mass average molecular weight of the polymer forming the binder B is not particularly limited and is preferably in the above range, but in the above range, 20,000 to 5,000,000 in terms of dispersibility and binding property. More preferably, 50,000 to 4,000,000 is even more preferable, and 200,000 to 3,000,000 is particularly preferable.
- the molecular weights of the polymer, polymer chain and macromonomer refer to the mass average molecular weight and the number average molecular weight in terms of standard polystyrene by gel permeation chromatography (GPC) unless otherwise specified.
- GPC gel permeation chromatography
- the measurement method the value measured by the method of the following condition 1 or condition 2 (priority) is basically used. However, an appropriate eluent may be appropriately selected and used depending on the type of polymer or macromonomer.
- polymer forming the binder examples include those synthesized in Examples, but the present invention is not limited thereto.
- the inorganic solid electrolyte-containing composition of the present invention contains an organic solvent as a dispersion medium for dispersing or dissolving each of the above components.
- the organic solvent may be any organic compound that is liquid in the environment of use, and examples thereof include various solvents. Specific examples thereof include alcohol compounds, ether compounds, amide compounds, amine compounds, ketone compounds, and aromatic compounds. Examples thereof include aliphatic compounds, nitrile compounds and ester compounds.
- the organic solvent may be a non-polar organic solvent (hydrophobic organic solvent) or a polar organic solvent (hydrophilic organic solvent), but a non-polar organic solvent is preferable because it can exhibit excellent dispersibility.
- the non-polar organic solvent generally refers to a property having a low affinity for water, but in the present invention, it is preferable that the organic solvent has a ClogP value of 1.5 to 6, for example, an ester compound, a ketone compound, and an ether. Examples thereof include compounds, aromatic compounds and aliphatic compounds.
- alcohol compounds include methyl alcohol, ethyl alcohol, 1-propyl alcohol, 2-propyl alcohol, 2-butanol, ethylene glycol, propylene glycol, glycerin, 1,6-hexanediol, cyclohexanediol, sorbitol, xylitol, 2 -Methyl-2,4-pentanediol, 1,3-butanediol, 1,4-butanediol can be mentioned.
- ether compound examples include alkylene glycol (triethylene glycol, etc.), alkylene glycol monoalkyl ether (ethylene glycol monomethyl ether, ethylene glycol monobutyl ether, diethylene glycol, dipropylene glycol, propylene glycol monomethyl ether, diethylene glycol monomethyl ether, triethylene glycol, polyethylene.
- alkylene glycol triethylene glycol, etc.
- alkylene glycol monoalkyl ether ethylene glycol monomethyl ether, ethylene glycol monobutyl ether, diethylene glycol, dipropylene glycol, propylene glycol monomethyl ether, diethylene glycol monomethyl ether, triethylene glycol, polyethylene.
- Glycol propylene glycol monomethyl ether, dipropylene glycol monomethyl ether, tripropylene glycol monomethyl ether, diethylene glycol monobutyl ether, diethylene glycol monobutyl ether, etc.), alkylene glycol dialkyl ether (ethylene glycol dimethyl ether, etc.), dialkyl ether (dimethyl ether, diethyl ether, diisopropyl ether, etc.) , Dibutyl ether, etc.), cyclic ether (tetrahexyl, dioxane (including 1,2-, 1,3- and 1,4-isomers), etc.).
- amide compound examples include N, N-dimethylformamide, N-methyl-2-pyrrolidone, 2-pyrrolidinone, 1,3-dimethyl-2-imidazolidinone, ⁇ -caprolactam, formamide, N-methylformamide and acetamide. , N-Methylacetamide, N, N-dimethylacetamide, N-methylpropanamide, hexamethylphosphoric triamide and the like.
- Examples of the amine compound include triethylamine, diisopropylethylamine, tributylamine and the like.
- Examples of the ketone compound include acetone, methyl ethyl ketone, methyl isobutyl ketone (MIBK), cyclopentanone, cyclohexanone, cycloheptanone, dipropyl ketone, dibutyl ketone, diisopropyl ketone, diisobutyl ketone (DIBK), isobutyl propyl ketone, sec-. Examples thereof include butyl propyl ketone, pentyl propyl ketone and butyl propyl ketone.
- Examples of the aromatic compound include benzene, toluene, xylene and the like.
- Examples of the aliphatic compound include hexane, heptane, octane, decane, cyclohexane, methylcyclohexane, ethylcyclohexane, cyclooctane, decalin, paraffin, gasoline, naphtha, kerosene, and light oil.
- Examples of the nitrile compound include acetonitrile, propionitrile, isobutyronitrile and the like.
- ester compound examples include ethyl acetate, butyl acetate, propyl acetate, propyl butyrate, isopropyl butyrate, butyl butyrate, isobutyl butyrate, butyl pentanate, ethyl isobutyrate, propyl isobutyrate, isopropyl isobutyrate, isobutyl isobutyrate, and pivalic acid.
- Examples thereof include propyl, isopropyl pivalate, butyl pivalate, and isobutyl pivalate.
- ether compounds, ketone compounds, aromatic compounds, aliphatic compounds and ester compounds are preferable, and ester compounds, ketone compounds or ether compounds are more preferable.
- the above organic solvent may contain one kind alone, but it is preferable to contain two or more kinds from the viewpoint of initial dispersibility, particularly dispersion stability.
- the number of types of the organic solvent contained in the inorganic solid electrolyte-containing composition is more preferably 2 to 4, and even more preferably 2 or 3.
- the organic solvent is composed of two or more kinds of compounds (organic solvents)
- the combination thereof is not particularly limited, but it is preferable that these are compatible with each other without phase separation.
- the hydrocarbon compound is a compound composed of a hydrocarbon among the above-mentioned aromatic compounds and aliphatic compounds.
- the proportion of the hydrocarbon compound in the organic solvent is preferably 25% by mass or more, more preferably 50% by mass or more, further preferably 60% by mass or more, further preferably 70% by mass or more, and further preferably 80% by mass. % Or more is particularly preferable, and 90% by mass or more is most preferable. It is also one of the preferred embodiments that the organic solvent is composed of a hydrocarbon compound.
- the carbon number of the compound constituting the organic solvent is not particularly limited, and is preferably 2 to 30, more preferably 4 to 20, further preferably 6 to 15, and particularly preferably 7 to 12.
- the compound constituting the organic solvent preferably has a CLogP value of 1 or more, more preferably 1.5 or more, further preferably 2 or more, and particularly preferably 3 or more.
- the upper limit is not particularly limited, but it is practically 10 or less, and preferably 6 or less.
- the CLogP value is a value obtained by calculating the common logarithm LogP of 1-octanol and the partition coefficient P to water.
- Known methods and software can be used for calculating the CRogP value, but unless otherwise specified, the structure is drawn using ChemDraw of PerkinElmer, and the calculated value is used.
- the ClogP value of the organic solvent is the sum of the products of the ClogP value of each organic solvent and the mass fraction.
- the organic solvent is one of preferred embodiments containing at least one organic solvent B having a ClogP value of 1.5 or more and less than 3.
- a solvent satisfying the ClogP value can be appropriately selected from each of the above compounds, and examples thereof include DBE and butyl butyrate.
- the ratio of the organic solvent B in the organic solvent is preferably 10% by mass or more, more preferably 20% by mass or more, further preferably 30% by mass or more, still more preferably 50% by mass or more.
- the upper limit value is not particularly limited and may be 100% by mass, preferably 99.9% by mass or less, and more preferably 99% by mass or less.
- the initial dispersibility is that the organic solvent contains at least one organic solvent A having a ClogP value of 3 or more and less than 6 and an organic solvent B having a ClogP value of 1.5 or more and less than 3.
- the organic solvent A a solvent satisfying the ClogP value can be appropriately selected from each of the above compounds, but a non-polar solvent, specifically, an ester compound, a ketone compound, an ether compound and the like are preferable.
- the organic solvent B is as described above.
- the ClogP value of the entire solvent containing the organic solvent A and the organic solvent B is not particularly limited, but is synonymous with the ClogP value of the compound constituting the organic solvent, and the preferable range is also the same.
- the ratio of the organic solvent A to the organic solvent is preferably 0.1% by mass or more, more preferably 0.5% by mass or more, further preferably 1% by mass or more, and particularly preferably 5% by mass or more.
- the upper limit value is not particularly limited, and is preferably 95% by mass or less, more preferably 80% by mass or less.
- the ratio of the organic solvent B in the organic solvent is the same as the ratio of the organic solvent B in the organic solvent described above, except that the upper limit is not 100% by mass.
- the mass ratio of the content of the organic solvent A to the content of the organic solvent B is not particularly limited, but is, for example, 1:99 to 99: 1. It is preferably 10:90 to 90:10, and more preferably.
- the organic solvent preferably has a boiling point of 50 ° C. or higher at normal pressure (1 atm), and more preferably 70 ° C. or higher.
- the upper limit is preferably 250 ° C. or lower, and more preferably 220 ° C. or lower.
- the content of the organic solvent in the composition containing the inorganic solid electrolyte is not particularly limited and can be appropriately set.
- the composition containing an inorganic solid electrolyte 20 to 80% by mass is preferable, 30 to 70% by mass is more preferable, and 40 to 60% by mass is particularly preferable.
- the inorganic solid electrolyte-containing composition of the present invention may also contain an active material capable of inserting and releasing ions of a metal belonging to Group 1 or Group 2 of the periodic table.
- the active material include a positive electrode active material and a negative electrode active material, which will be described below.
- an inorganic solid electrolyte-containing composition containing an active material positive electrode active material or negative electrode active material
- an electrode layer composition positive electrode layer composition or negative electrode layer composition
- the positive electrode active material is preferably one capable of reversibly inserting and releasing lithium ions.
- the material is not particularly limited as long as it has the above characteristics, and may be a transition metal oxide, an element that can be composited with Li such as sulfur, or the like.
- the 1 (Ia) group elements of the transition metal oxide to elemental M b (Table metal periodic other than lithium, the elements of the 2 (IIa) group, Al, Ga, In, Ge , Sn, Pb, Elements such as Sb, Bi, Si, P and B) may be mixed.
- the mixing amount is preferably 0 to 30 mol% relative to the amount of the transition metal element M a (100 mol%). That the molar ratio of li / M a was synthesized were mixed so that 0.3 to 2.2, more preferably.
- transition metal oxide examples include (MA) a transition metal oxide having a layered rock salt type structure, (MB) a transition metal oxide having a spinel type structure, (MC) a lithium-containing transition metal phosphoric acid compound, and (MD). ) Lithium-containing transition metal halide phosphoric acid compound, (ME) lithium-containing transition metal silicic acid compound, and the like.
- transition metal oxide having a layered rock salt structure examples include LiCoO 2 (lithium cobalt oxide [LCO]), LiNi 2 O 2 (lithium nickel oxide), LiNi 0.85 Co 0.10 Al 0. 05 O 2 (Lithium Nickel Cobalt Aluminate [NCA]), LiNi 1/3 Co 1/3 Mn 1/3 O 2 (Lithium Nickel Manganese Cobalt Oxide [NMC]) and LiNi 0.5 Mn 0.5 O 2 ( Manganese nickel oxide).
- LiCoO 2 lithium cobalt oxide
- LiNi 2 O 2 lithium nickel oxide
- LiNi 0.85 Co 0.10 Al 0. 05 O 2 Lithium Nickel Cobalt Aluminate [NCA]
- LiNi 1/3 Co 1/3 Mn 1/3 O 2 Lithium Nickel Manganese Cobalt Oxide [NMC]
- LiNi 0.5 Mn 0.5 O 2 Manganese nickel oxide
- (MB) Specific examples of the transition metal oxide having a spinel structure, LiMn 2 O 4 (LMO) , LiCoMnO 4, Li 2 FeMn 3 O 8, Li 2 CuMn 3 O 8, Li 2 CrMn 3 O 8 and Li 2 Nimn 3 O 8 can be mentioned.
- Examples of the (MC) lithium-containing transition metal phosphate compound include olivine-type iron phosphate salts such as LiFePO 4 and Li 3 Fe 2 (PO 4 ) 3 , iron pyrophosphates such as LiFeP 2 O 7 , and LiCoPO 4.
- Examples thereof include cobalt phosphates of the above, and monoclinic panocycon-type vanadium phosphate salts such as Li 3 V 2 (PO 4 ) 3 (lithium vanadium phosphate).
- (MD) as the lithium-containing transition metal halogenated phosphate compound for example, Li 2 FePO 4 F such fluorinated phosphorus iron salt, Li 2 MnPO 4 hexafluorophosphate manganese salts such as F and Li 2 CoPO 4 F Fluorophosphate cobalts and the like.
- Examples of the (ME) lithium-containing transition metal silicic acid compound include Li 2 FeSiO 4 , Li 2 MnSiO 4 , and Li 2 CoSiO 4 .
- a transition metal oxide having a (MA) layered rock salt type structure is preferable, and LCO or NMC is more preferable.
- the shape of the positive electrode active material is not particularly limited, but is preferably in the form of particles.
- the particle size (volume average particle size) of the positive electrode active material is not particularly limited. For example, it can be 0.1 to 50 ⁇ m.
- the particle size of the positive electrode active material particles can be measured in the same manner as the particle size of the above-mentioned inorganic solid electrolyte.
- a normal crusher or classifier is used to adjust the positive electrode active material to a predetermined particle size. For example, a mortar, a ball mill, a sand mill, a vibrating ball mill, a satellite ball mill, a planetary ball mill, a swirling airflow type jet mill or a sieve is preferably used.
- wet pulverization in which an organic solvent such as water or methanol coexists can also be performed. It is preferable to perform classification in order to obtain a desired particle size.
- the classification is not particularly limited, and can be performed using a sieve, a wind power classifier, or the like. Both dry and wet classifications can be used.
- the positive electrode active material obtained by the firing method may be used after being washed with water, an acidic aqueous solution, an alkaline aqueous solution, or an organic solvent.
- the positive electrode active material one type may be used alone, or two or more types may be used in combination.
- the mass (mg) (grain amount) of the positive electrode active material per unit area (cm 2 ) of the positive electrode active material layer is not particularly limited. It can be appropriately determined according to the designed battery capacity, and can be, for example, 1 to 100 mg / cm 2 .
- the content of the positive electrode active material in the composition containing an inorganic solid electrolyte is not particularly limited, and is preferably 10 to 97% by mass, more preferably 30 to 95% by mass, and 40 to 93% by mass in terms of solid content of 100% by mass. More preferably, 50 to 90% by mass is particularly preferable.
- the negative electrode active material is preferably one capable of reversibly inserting and releasing lithium ions.
- the material is not particularly limited as long as it has the above characteristics, and examples thereof include a carbonaceous material, a metal oxide, a metal composite oxide, a simple substance of lithium, a lithium alloy, and a negative electrode active material capable of forming an alloy with lithium. .. Among them, a carbonaceous material, a metal composite oxide or a simple substance of lithium is preferably used from the viewpoint of reliability.
- the carbonaceous material used as the negative electrode active material is a material substantially composed of carbon.
- carbon black such as acetylene black (AB), graphite (artificial graphite such as natural graphite and vapor-grown graphite), and PAN (polyacrylonitrile) -based resin or furfuryl alcohol resin.
- a carbonaceous material obtained by firing a resin can be mentioned.
- various carbon fibers such as PAN-based carbon fibers, cellulose-based carbon fibers, pitch-based carbon fibers, vapor-grown carbon fibers, dehydrated PVA (polypoly alcohol) -based carbon fibers, lignin carbon fibers, graphitic carbon fibers and activated carbon fibers.
- carbonaceous materials can also be divided into non-graphitizable carbonaceous materials (also referred to as hard carbon) and graphite-based carbonaceous materials depending on the degree of graphitization. Further, the carbonaceous material preferably has the plane spacing or density and the size of crystallites described in JP-A No. 62-22066, JP-A No. 2-6856, and JP-A-3-45473.
- the carbonaceous material does not have to be a single material, and a mixture of natural graphite and artificial graphite described in JP-A-5-90844, graphite having a coating layer described in JP-A-6-4516, and the like should be used. You can also.
- As the carbonaceous material hard carbon or graphite is preferably used, and graphite is more preferably used.
- the metal or semi-metal element oxide applied as the negative electrode active material is not particularly limited as long as it is an oxide capable of storing and releasing lithium, and is a composite of a metal element oxide (metal oxide) and a metal element.
- metal oxide metal oxide
- examples thereof include oxides or composite oxides of metal elements and semi-metal elements (collectively referred to as metal composite oxides) and oxides of semi-metal elements (semi-metal oxides).
- metal composite oxides oxides or composite oxides of metal elements and semi-metal elements
- oxides of semi-metal elements semi-metal elements
- amorphous oxides are preferable, and chalcogenides, which are reaction products of metal elements and elements of Group 16 of the periodic table, are also preferable.
- the metalloid element means an element exhibiting properties intermediate between the metalloid element and the non-metalloid element, and usually contains six elements of boron, silicon, germanium, arsenic, antimony and tellurium, and further selenium. , Polonium and astatine.
- amorphous means an X-ray diffraction method using CuK ⁇ rays, which has a broad scattering band having an apex in a region of 20 ° to 40 ° in 2 ⁇ value, and a crystalline diffraction line is used. You may have.
- the strongest intensity of the crystalline diffraction lines found at the 2 ⁇ value of 40 ° to 70 ° is 100 times or less the diffraction line intensity at the apex of the broad scattering band seen at the 2 ⁇ value of 20 ° to 40 °. It is preferable that it is 5 times or less, and it is particularly preferable that it does not have a crystalline diffraction line.
- the amorphous oxide of the metalloid element or the chalcogenide is more preferable, and the elements of the groups 13 (IIIB) to 15 (VB) of the periodic table (for example).
- Al, Ga, Si, Sn, Ge, Pb, Sb and Bi) alone or a combination of two or more (composite) oxides, or chalcogenides are particularly preferred.
- preferable amorphous oxides and chalcogenides include, for example, Ga 2 O 3 , GeO, PbO, PbO 2 , Pb 2 O 3 , Pb 2 O 4 , Pb 3 O 4 , Sb 2 O 3 , Sb 2.
- Examples of the negative electrode active material that can be used in combination with the amorphous oxide negative electrode active material centering on Sn, Si, and Ge include a carbonaceous material capable of occluding and / or releasing lithium ions or lithium metal, lithium alone, and lithium.
- a negative electrode active material that can be alloyed with an alloy or lithium is preferably used.
- the oxide of a metal or a metalloid element contains at least one of titanium and lithium as a constituent component from the viewpoint of high current density charge / discharge characteristics.
- the lithium-containing metal composite oxide include a composite oxide of lithium oxide and the metal (composite) oxide or the chalcogenide, and more specifically, Li 2 SnO 2.
- the negative electrode active material for example, a metal oxide, contains a titanium element (titanium oxide).
- Li 4 Ti 5 O 12 lithium titanate [LTO]
- Li 4 Ti 5 O 12 has excellent rapid charge / discharge characteristics due to small volume fluctuations during storage and release of lithium ions, and electrode deterioration is suppressed and lithium ion secondary It is preferable in that the life of the battery can be improved.
- the lithium alloy as the negative electrode active material is not particularly limited as long as it is an alloy usually used as the negative electrode active material of the secondary battery, and examples thereof include a lithium aluminum alloy.
- the negative electrode active material that can be alloyed with lithium is not particularly limited as long as it is usually used as the negative electrode active material of the secondary battery. Such an active material has a large expansion and contraction due to charge and discharge, so that the binding property of solid particles is lowered. However, in the present invention, high binding property can be achieved by using the above-mentioned binders A and B in combination.
- Examples of such an active material include a (negative electrode) active material (alloy) having a silicon element or a tin element, and each metal such as Al and In, and a negative electrode active material having a silicon element that enables a higher battery capacity (negative electrode).
- a silicon element-containing active material (Silicon element-containing active material) is preferable, and a silicon element-containing active material having a silicon element content of 50 mol% or more of all the constituent elements is more preferable.
- a negative electrode containing these negative electrode active materials Si negative electrode containing a silicon element-containing active material, Sn negative electrode containing an active material having a tin element, etc.
- a carbon negative electrode graphite, acetylene black, etc.
- silicon element-containing active material examples include silicon materials such as Si and SiOx (0 ⁇ x ⁇ 1), and silicon-containing alloys containing titanium, vanadium, chromium, manganese, nickel, copper, lanthanum, and the like (for example,).
- LaSi 2 , VSi 2 , La-Si, Gd-Si, Ni-Si) or organized active material (eg LaSi 2 / Si), as well as other silicon and tin elements such as SnSiO 3 , SnSiS 3 Examples include active materials containing.
- SiOx itself can be used as a negative electrode active material (semi-metal oxide), and since Si is generated by the operation of an all-solid-state secondary battery, a negative electrode active material that can be alloyed with lithium (its). It can be used as a precursor substance).
- the negative electrode active material having a tin element include Sn, SnO, SnO 2 , SnS, SnS 2 , and the active material containing the silicon element and the tin element.
- a composite oxide with lithium oxide for example, Li 2 SnO 2 can also be mentioned.
- the above-mentioned negative electrode active material can be used without particular limitation, but in terms of battery capacity, a negative electrode active material that can be alloyed with silicon is a preferable embodiment as the negative electrode active material.
- a negative electrode active material that can be alloyed with silicon is a preferable embodiment as the negative electrode active material.
- the above-mentioned silicon material or silicon-containing alloy (alloy containing a silicon element) is more preferable, and silicon (Si) or a silicon-containing alloy is further preferable.
- the chemical formula of the compound obtained by the above firing method can be calculated from the inductively coupled plasma (ICP) emission spectroscopic analysis method as a measurement method and the mass difference of the powder before and after firing as a simple method.
- ICP inductively coupled plasma
- the shape of the negative electrode active material is not particularly limited, but it is preferably in the form of particles.
- the volume average particle size of the negative electrode active material is not particularly limited, but is preferably 0.1 to 60 ⁇ m.
- the volume average particle diameter of the negative electrode active material particles can be measured in the same manner as the average particle diameter of the inorganic solid electrolyte. In order to obtain a predetermined particle size, a normal crusher or classifier is used as in the case of the positive electrode active material.
- the negative electrode active material may be used alone or in combination of two or more.
- the mass (mg) (grain amount) of the negative electrode active material per unit area (cm 2 ) of the negative electrode active material layer is not particularly limited. It can be appropriately determined according to the designed battery capacity, and can be, for example, 1 to 100 mg / cm 2 .
- the content of the negative electrode active material in the composition containing an inorganic solid electrolyte is not particularly limited, and is preferably 10 to 90% by mass, more preferably 20 to 85% by mass, and 30 to 30% by mass in terms of solid content of 100% by mass. It is more preferably 80% by mass, and even more preferably 40 to 75% by mass.
- the negative electrode active material layer when the negative electrode active material layer is formed by charging the secondary battery, instead of the negative electrode active material, a metal belonging to Group 1 or Group 2 of the periodic table generated in the all-solid-state secondary battery Ions can be used.
- the negative electrode active material layer can be formed by combining these ions with electrons and precipitating them as a metal.
- the surfaces of the positive electrode active material and the negative electrode active material may be surface-coated with another metal oxide.
- the surface coating agent include metal oxides containing Ti, Nb, Ta, W, Zr, Al, Si or Li. Specific examples thereof include spinel titanate, tantalum oxide, niobate oxide, lithium niobate compound and the like.
- the surface of the electrode containing the positive electrode active material or the negative electrode active material may be surface-treated with sulfur or phosphorus.
- the particle surface of the positive electrode active material or the negative electrode active material may be surface-treated with active light rays or an active gas (plasma or the like) before and after the surface coating.
- the inorganic solid electrolyte-containing composition of the present invention may appropriately contain a conductive auxiliary agent, and in particular, a silicon atom-containing active material as a negative electrode active material is preferably used in combination with the conductive auxiliary agent.
- the conductive auxiliary agent is not particularly limited, and those known as general conductive auxiliary agents can be used.
- graphites such as natural graphite and artificial graphite, carbon blacks such as acetylene black, ketjen black and furnace black, amorphous carbon such as needle coke, vapor-grown carbon fibers or carbon nanotubes, which are electron conductive materials.
- It may be a carbon fiber such as graphene or fullerene, a metal powder such as copper or nickel, or a metal fiber, and a conductive polymer such as polyaniline, polypyrrole, polythiophene, polyacetylene, or polyphenylene derivative. May be used.
- the active material and the conductive auxiliary agent when the battery is charged and discharged, the ions of the metal belonging to the first group or the second group of the periodic table (preferably Li).
- a conductive auxiliary agent is one that does not insert and release ions) and does not function as an active material.
- conductive auxiliary agents those that can function as active materials in the active material layer when the battery is charged and discharged are classified as active materials instead of conductive auxiliary agents. Whether or not the battery functions as an active material when it is charged and discharged is not unique and is determined by the combination with the active material.
- the conductive auxiliary agent may contain one kind or two or more kinds.
- the shape of the conductive auxiliary agent is not particularly limited, but is preferably in the form of particles.
- the content of the conductive auxiliary agent in the inorganic solid electrolyte-containing composition is preferably 0 to 10% by mass.
- the inorganic solid electrolyte-containing composition of the present invention preferably contains a lithium salt (supporting electrolyte).
- the lithium salt the lithium salt usually used in this kind of product is preferable, and there is no particular limitation.
- the lithium salt described in paragraphs 882 to 985 of JP2015-088486 is preferable.
- the content of the lithium salt is preferably 0.1 part by mass or more, more preferably 5 parts by mass or more, based on 100 parts by mass of the solid electrolyte.
- the upper limit is preferably 50 parts by mass or less, and more preferably 20 parts by mass or less.
- the inorganic solid electrolyte-containing composition of the present invention does not have to contain a dispersant other than this binder, but may contain a dispersant.
- the dispersant those usually used for all-solid-state secondary batteries can be appropriately selected and used. In general, compounds intended for particle adsorption, steric repulsion and / or electrostatic repulsion are preferably used.
- the composition containing an inorganic solid electrolyte of the present invention contains an ionic liquid, a thickener, and a cross-linking agent (such as those that undergo a cross-linking reaction by radical polymerization, condensation polymerization, or ring-opening polymerization) as other components other than the above components.
- a cross-linking agent such as those that undergo a cross-linking reaction by radical polymerization, condensation polymerization, or ring-opening polymerization
- Polymerization initiators such as those that generate acids or radicals by heat or light
- defoaming agents leveling agents, dehydrating agents, antioxidants and the like
- the ionic liquid is contained in order to further improve the ionic conductivity, and known ones can be used without particular limitation.
- a polymer other than the above polymer, a commonly used binder and the like may be contained.
- the composition containing an inorganic solid electrolyte of the present invention is a mixture of an inorganic solid electrolyte, the above binder, an organic solvent, and optionally a lithium salt, and any other components, for example, by mixing them in various commonly used mixers.
- a slurry preferably as a slurry.
- the mixing method is not particularly limited, and the mixture may be mixed all at once or sequentially.
- the mixing environment is not particularly limited, and examples thereof include under dry air and under an inert gas.
- the composition for forming an active material layer of the present invention can be a dispersion liquid containing solid particles that are highly dispersed over a long period of time by suppressing (re) aggregation of solid particles.
- the sheet for an all-solid-state secondary battery of the present invention is a sheet-like molded body capable of forming a constituent layer of an all-solid-state secondary battery, and includes various aspects depending on its use.
- a sheet preferably used for a solid electrolyte layer also referred to as a solid electrolyte sheet for an all-solid secondary battery
- an electrode or a sheet preferably used for a laminate of an electrode and a solid electrolyte layer (an electrode for an all-solid secondary battery).
- Sheet and the like.
- these various sheets may be collectively referred to as an all-solid-state secondary battery sheet.
- the solid electrolyte sheet for an all-solid secondary battery of the present invention may be a sheet having a solid electrolyte layer, and even a sheet having a solid electrolyte layer formed on a base material does not have a base material and is a solid electrolyte layer. It may be a sheet formed of.
- the solid electrolyte sheet for an all-solid secondary battery may have another layer in addition to the solid electrolyte layer. Examples of other layers include a protective layer (release sheet), a current collector, a coat layer, and the like.
- the solid electrolyte sheet for an all-solid secondary battery of the present invention for example, a sheet having a layer composed of the inorganic solid electrolyte-containing composition of the present invention, a normal solid electrolyte layer, and a protective layer on a substrate in this order.
- the solid electrolyte layer contained in the solid electrolyte sheet for an all-solid secondary battery is preferably formed of the inorganic solid electrolyte-containing composition of the present invention.
- the content of each component in the solid electrolyte layer is not particularly limited, but is preferably synonymous with the content of each component in the solid content of the inorganic solid electrolyte-containing composition of the present invention.
- the thickness of each layer constituting the solid electrolyte sheet for the all-solid-state secondary battery is the same as the thickness of each layer described in the all-solid-state secondary battery described later.
- the base material is not particularly limited as long as it can support the solid electrolyte layer, and examples thereof include a material described in the current collector described later, a sheet body (plate-like body) such as an organic material and an inorganic material.
- a material described in the current collector described later a sheet body (plate-like body) such as an organic material and an inorganic material.
- the organic material include various polymers, and specific examples thereof include polyethylene terephthalate, polypropylene, polyethylene, and cellulose.
- the inorganic material include glass, ceramic and the like.
- the electrode sheet for an all-solid-state secondary battery of the present invention may be an electrode sheet having an active material layer, and the active material layer is formed on a base material (current collector).
- the sheet may be a sheet that does not have a base material and is formed from an active material layer.
- This electrode sheet is usually a sheet having a current collector and an active material layer, but an embodiment having a current collector, an active material layer and a solid electrolyte layer in this order, and a current collector, an active material layer and a solid electrolyte. An embodiment having a layer and an active material layer in this order is also included.
- the solid electrolyte layer and the active material layer of the electrode sheet are preferably formed of the inorganic solid electrolyte-containing composition of the present invention.
- the content of each component in the solid electrolyte layer or the active material layer is not particularly limited, but the content of each component in the solid content of the inorganic solid electrolyte-containing composition (composition for electrode layer) of the present invention is preferable. Synonymous with quantity.
- the thickness of each layer constituting the electrode sheet of the present invention is the same as the thickness of each layer described in the all-solid-state secondary battery described later.
- the electrode sheet of the present invention may have the other layers described above.
- the all-solid-state secondary battery sheet of the present invention at least one of the solid electrolyte layer and the active material layer is formed of the inorganic solid electrolyte-containing composition of the present invention, and the solid particles in this layer are firmly bonded to each other.
- the active material layer formed of the inorganic solid electrolyte-containing composition of the present invention is firmly bonded to the current collector.
- the sheet for an all-solid-state secondary battery of the present invention is suitably used as a sheet capable of forming a constituent layer of an all-solid-state secondary battery.
- the method for producing the sheet for an all-solid secondary battery of the present invention is not particularly limited, and the sheet can be produced by forming each of the above layers using the inorganic solid electrolyte-containing composition of the present invention.
- a film is formed (coating and drying) on a base material or a current collector (which may be via another layer) to form a layer (coating and drying layer) composed of an inorganic solid electrolyte-containing composition.
- the method can be mentioned. Thereby, an all-solid-state secondary battery sheet having a base material or a current collector and a coating dry layer can be produced.
- the coating dry layer is a layer formed by applying the inorganic solid electrolyte-containing composition of the present invention and drying the organic solvent (that is, the inorganic solid electrolyte-containing composition of the present invention is used.
- the organic solvent may remain as long as the effects of the present invention are not impaired, and the residual amount may be, for example, 3% by mass or less in each layer.
- each step such as coating and drying will be described in the following method for producing an all-solid-state secondary battery.
- the coating dry layer obtained as described above can also be pressurized.
- the pressurizing conditions and the like will be described later in the method for manufacturing an all-solid-state secondary battery.
- the base material, the protective layer (particularly the release sheet) and the like can be peeled off.
- the all-solid secondary battery of the present invention has a positive electrode active material layer, a negative electrode active material layer facing the positive electrode active material layer, and a solid electrolyte layer arranged between the positive electrode active material layer and the negative electrode active material layer.
- the positive electrode active material layer is preferably formed on the positive electrode current collector and constitutes the positive electrode.
- the negative electrode active material layer is preferably formed on the negative electrode current collector to form the negative electrode. It is preferable that at least one layer of the negative electrode active material layer, the positive electrode active material layer and the solid electrolyte layer is formed of the inorganic solid electrolyte-containing composition of the present invention, and all the layers are the inorganic solid electrolyte-containing composition of the present invention.
- the mode in which it is formed is also preferable.
- the active material layer or the solid electrolyte layer formed of the inorganic solid electrolyte-containing composition of the present invention is preferably one in the solid content of the inorganic solid electrolyte-containing composition of the present invention with respect to the component species contained therein and the content ratio thereof. Is the same as.
- a known material can be used.
- the thicknesses of the negative electrode active material layer, the solid electrolyte layer, and the positive electrode active material layer are not particularly limited.
- each layer is preferably 10 to 1,000 ⁇ m, more preferably 20 ⁇ m or more and less than 500 ⁇ m, respectively, in consideration of the dimensions of a general all-solid-state secondary battery.
- the thickness of at least one of the positive electrode active material layer and the negative electrode active material layer is more preferably 50 ⁇ m or more and less than 500 ⁇ m.
- the positive electrode active material layer and the negative electrode active material layer may each have a current collector on the opposite side of the solid electrolyte layer.
- the all-solid-state secondary battery of the present invention may be used as an all-solid-state secondary battery with the above structure, but in order to form a dry battery, it should be further enclosed in a suitable housing.
- the housing may be made of metal or resin (plastic).
- a metallic material for example, one made of aluminum alloy or stainless steel can be mentioned.
- the metallic housing is divided into a positive electrode side housing and a negative electrode side housing, and electrically connected to the positive electrode current collector and the negative electrode current collector, respectively. It is preferable that the housing on the positive electrode side and the housing on the negative electrode side are joined and integrated via a gasket for preventing a short circuit.
- FIG. 1 is a schematic cross-sectional view showing an all-solid-state secondary battery (lithium ion secondary battery) according to a preferred embodiment of the present invention.
- the all-solid-state secondary battery 10 of the present embodiment has a negative electrode current collector 1, a negative electrode active material layer 2, a solid electrolyte layer 3, a positive electrode active material layer 4, and a positive electrode current collector 5 in this order when viewed from the negative electrode side. ..
- Each layer is in contact with each other and has an adjacent structure.
- the lithium ions (Li + ) accumulated in the negative electrode are returned to the positive electrode side, and electrons are supplied to the operating portion 6.
- a light bulb is used as a model for the operating portion 6, and the light bulb is turned on by electric discharge.
- the all-solid-state secondary battery having the layer structure shown in FIG. 1 When the all-solid-state secondary battery having the layer structure shown in FIG. 1 is placed in a 2032 type coin case, the all-solid-state secondary battery is referred to as an all-solid-state secondary battery electrode sheet, and the all-solid-state secondary battery electrode sheet is referred to as an all-solid-state secondary battery electrode sheet.
- a battery manufactured by putting it in a 2032 type coin case is sometimes called an all-solid-state secondary battery.
- the positive electrode active material layer, the solid electrolyte layer, and the negative electrode active material layer are all formed of the inorganic solid electrolyte-containing composition of the present invention.
- the all-solid-state secondary battery 10 exhibits excellent battery performance.
- the inorganic solid electrolyte and the binder contained in the positive electrode active material layer 4, the solid electrolyte layer 3, and the negative electrode active material layer 2 may be of the same type or different from each other.
- either or both of the positive electrode active material layer and the negative electrode active material layer may be simply referred to as an active material layer or an electrode active material layer.
- either or both of the positive electrode active material and the negative electrode active material may be collectively referred to as an active material or an electrode active material.
- the binding property of the solid particles is enhanced as described above, and the solid particles have poor contact with each other and are collected. It is possible to suppress the peeling of solid particles from the electric body. Further, it is possible to suppress an increase in the interfacial resistance between the solid particles and an increase in the interfacial resistance between the solid particles and the current collector. Therefore, the all-solid-state secondary battery of the present invention exhibits excellent battery performance.
- the negative electrode active material layer can be a lithium metal layer.
- the lithium metal layer include a layer formed by depositing or molding a lithium metal powder, a lithium foil, a lithium vapor deposition film, and the like.
- the thickness of the lithium metal layer can be, for example, 1 to 500 ⁇ m regardless of the thickness of the negative electrode active material layer.
- the positive electrode current collector 5 and the negative electrode current collector 1 are preferably electron conductors.
- either or both of the positive electrode current collector and the negative electrode current collector may be collectively referred to as a current collector.
- a current collector As a material for forming the positive electrode current collector, in addition to aluminum, aluminum alloy, stainless steel, nickel and titanium, the surface of aluminum or stainless steel is treated with carbon, nickel, titanium or silver (a thin film is formed). Of these, aluminum and aluminum alloys are more preferable.
- As a material for forming the negative electrode current collector in addition to aluminum, copper, copper alloy, stainless steel, nickel and titanium, carbon, nickel, titanium or silver is treated on the surface of aluminum, copper, copper alloy or stainless steel.
- aluminum, copper, copper alloy and stainless steel are more preferable.
- the shape of the current collector is usually a film sheet, but a net, a punched body, a lath body, a porous body, a foam body, a molded body of a fiber group, or the like can also be used.
- the thickness of the current collector is not particularly limited, but is preferably 1 to 500 ⁇ m. Further, it is also preferable that the surface of the current collector is made uneven by surface treatment.
- a functional layer, a member, or the like is appropriately interposed or arranged between or outside each of the negative electrode current collector, the negative electrode active material layer, the solid electrolyte layer, the positive electrode active material layer, and the positive electrode current collector. You may. Further, each layer may be composed of a single layer or a plurality of layers.
- the all-solid-state secondary battery can be manufactured by a conventional method. Specifically, the all-solid-state secondary battery can be manufactured by forming each of the above layers using the inorganic solid electrolyte-containing composition or the like of the present invention. Moreover, since the inorganic solid electrolyte-containing composition of the present invention is excellent in dispersion stability, the preparation of the inorganic solid electrolyte-containing composition and the film formation of each layer are continuously performed in time (immediately after the composition is prepared). An all-solid-state secondary battery with suppressed deterioration of battery performance can be manufactured without implementation. As described above, the present invention can manufacture an all-solid-state secondary battery exhibiting excellent battery performance and preferably a small electric resistance under flexible manufacturing conditions. The details will be described below.
- the inorganic solid electrolyte-containing composition of the present invention is appropriately applied onto a base material (for example, a metal foil serving as a current collector) to form a coating film (film formation).
- a method including (via) a step a method for producing a sheet for an all-solid-state secondary battery of the present invention
- an inorganic solid electrolyte-containing composition containing a positive electrode active material is applied as a positive electrode material (composition for a positive electrode layer) on a metal foil which is a positive electrode current collector to form a positive electrode active material layer.
- a positive electrode sheet for a solid secondary battery is produced.
- an inorganic solid electrolyte-containing composition for forming the solid electrolyte layer is applied onto the positive electrode active material layer to form the solid electrolyte layer.
- an inorganic solid electrolyte-containing composition containing a negative electrode active material is applied as a negative electrode material (composition for the negative electrode layer) on the solid electrolyte layer to form a negative electrode active material layer.
- a negative electrode current collector metal foil
- an all-solid secondary battery having a structure in which a solid electrolyte layer is sandwiched between the positive electrode active material layer and the negative electrode active material layer can be obtained. Can be done. This can be enclosed in a housing to obtain a desired all-solid-state secondary battery.
- a negative electrode active material layer, a solid electrolyte layer and a positive electrode active material layer are formed on the negative electrode current collector, and the positive electrode current collectors are superposed to manufacture an all-solid secondary battery. You can also do it.
- a positive electrode sheet for an all-solid-state secondary battery is produced. Further, an inorganic solid electrolyte-containing composition containing a negative electrode active material is applied as a negative electrode material (composition for a negative electrode layer) on a metal foil which is a negative electrode current collector to form a negative electrode active material layer. A negative electrode sheet for a solid secondary battery is manufactured. Next, a solid electrolyte layer is formed on the active material layer of any one of these sheets as described above.
- the other of the positive electrode sheet for the all-solid-state secondary battery and the negative electrode sheet for the all-solid-state secondary battery is laminated on the solid electrolyte layer so that the solid electrolyte layer and the active material layer are in contact with each other.
- an all-solid-state secondary battery can be manufactured.
- the following method can be mentioned. That is, as described above, a positive electrode sheet for an all-solid-state secondary battery and a negative electrode sheet for an all-solid-state secondary battery are produced. Separately from this, an inorganic solid electrolyte-containing composition is applied onto a base material to prepare a solid electrolyte sheet for an all-solid secondary battery composed of a solid electrolyte layer.
- the positive electrode sheet for the all-solid-state secondary battery and the negative electrode sheet for the all-solid-state secondary battery are laminated so as to sandwich the solid electrolyte layer peeled from the base material. In this way, an all-solid-state secondary battery can be manufactured.
- the solid electrolyte layer or the like can also be formed by, for example, forming an inorganic solid electrolyte-containing composition or the like on a substrate or an active material layer by pressure molding under the pressure conditions described later.
- the inorganic solid electrolyte-containing composition of the present invention may be used as any one of the positive electrode layer composition, the inorganic solid electrolyte-containing composition and the negative electrode layer composition, and all of them are the present invention. It is preferable to use the composition containing an inorganic solid electrolyte of.
- the method for applying the composition containing an inorganic solid electrolyte is not particularly limited and can be appropriately selected.
- coating preferably wet coating
- spray coating spin coating coating
- dip coating coating dip coating coating
- slit coating stripe coating
- bar coating coating can be mentioned.
- the inorganic solid electrolyte-containing composition may be subjected to a drying treatment after being applied to each of them, or may be subjected to a drying treatment after being applied in multiple layers.
- the drying temperature is not particularly limited.
- the lower limit is preferably 30 ° C. or higher, more preferably 60 ° C. or higher, and even more preferably 80 ° C. or higher.
- the upper limit is preferably 300 ° C.
- the organic solvent can be removed and a solid state (coating dry layer) can be obtained. Further, it is preferable because the temperature is not raised too high and each member of the all-solid-state secondary battery is not damaged. As a result, in an all-solid-state secondary battery, it is possible to obtain excellent overall performance, good binding properties, and good ionic conductivity even without pressurization.
- the solid particles are firmly bound to each other, and a coating dry layer having a small interfacial resistance between the solid particles can be formed.
- Examples of the pressurizing method include a hydraulic cylinder press machine and the like.
- the pressing force is not particularly limited, and is generally preferably in the range of 5 to 1500 MPa.
- the applied inorganic solid electrolyte-containing composition may be heated at the same time as pressurization.
- the heating temperature is not particularly limited, and is generally in the range of 30 to 300 ° C. It can also be pressed at a temperature higher than the glass transition temperature of the inorganic solid electrolyte.
- the binder when the inorganic solid electrolyte and the binder coexist, the binder can be pressed at a temperature higher than the glass transition temperature of the binder. However, in general, the temperature does not exceed the melting point of the above-mentioned binder.
- the pressurization may be carried out in a state where the coating solvent or the organic solvent has been dried in advance, or may be carried out in a state where the solvent or the organic solvent remains.
- each composition may be applied at the same time, and the application drying press may be performed simultaneously and / or sequentially. After coating on separate substrates, they may be laminated by transfer.
- the atmosphere during pressurization is not particularly limited, and may be any of air, dry air (dew point ⁇ 20 ° C. or lower), inert gas (for example, argon gas, helium gas, nitrogen gas) and the like.
- the pressing time may be short (for example, within several hours) and high pressure may be applied, or medium pressure may be applied for a long time (1 day or more).
- an all-solid-state secondary battery restraint screw tightening pressure, etc.
- the press pressure may be uniform or different with respect to the pressed portion such as the sheet surface.
- the press pressure can be changed according to the area or thickness of the pressed portion. It is also possible to change the same part step by step with different pressures.
- the pressed surface may be smooth or roughened.
- the all-solid-state secondary battery manufactured as described above is preferably initialized after manufacturing or before use.
- the initialization is not particularly limited, and can be performed, for example, by performing initial charging / discharging with the press pressure increased, and then releasing the pressure until the pressure reaches the general working pressure of the all-solid-state secondary battery.
- the all-solid-state secondary battery of the present invention can be applied to various applications.
- the application mode is not particularly limited, but for example, when mounted on an electronic device, a laptop computer, a pen input computer, a mobile computer, an electronic book player, a mobile phone, a cordless phone handset, a pager, a handy terminal, a mobile fax, or a mobile phone. Examples include copying, mobile printers, headphone stereos, video movies, LCD TVs, handy cleaners, portable CDs, mini discs, electric shavers, transceivers, electronic notebooks, calculators, memory cards, portable tape recorders, radios, backup power supplies, etc.
- Other consumer products include automobiles, electric vehicles, motors, lighting equipment, toys, game equipment, road conditioners, watches, strobes, cameras, medical equipment (pacemakers, hearing aids, shoulder massagers, etc.). Furthermore, it can be used for various munitions and space. It can also be combined with a solar cell.
- Macromonomer M- which is a reaction of acrylic acid (manufactured by Fujifilm Wako Pure Chemical Industries, Ltd.) with a polymer polymerized with glycidyl methacrylate (manufactured by Tokyo Chemical Industry Co., Ltd.) at a ratio of 1: 0.99: 0.01 (molar ratio). I got 1.
- the SP value of this macromonomer M-1 was 9.3, and the number average molecular weight was 11,000.
- the SP value and the number average molecular weight of the macromonomer are values calculated by the above method.
- Synthesis Example 2 Synthesis of Acrylic Polymer A2 and Preparation of Binder Dispersion Solution of Acrylic Polymer A2
- the amount of MA was changed from 12.4 g to 12.6 g
- the amount of MA in another container was changed from 222.8 g to 256.2 g
- the amount of AA was changed from 6.7 g to 6.8 g in another container.
- a dispersion of a binder composed of acrylic polymer A2 was prepared in the same manner as in Synthesis Example 1 except that the amount of AA was changed from 75.0 g to 47.0 g.
- Synthesis Example 3 Synthesis of Acrylic Polymer B and Preparation of Binder Dispersion Solution of Acrylic Polymer B
- the amount of MA was changed from 12.4 g to 12.7 g
- the amount of MA in another container was changed from 222.8 g to 272.9 g
- the amount of AA was changed from 6.7 g to 6.9 g in another container.
- a dispersion of a binder composed of acrylic polymer B was prepared in the same manner as in Synthesis Example 1 except that the amount of AA in AA was changed from 75.0 g to 33.0 g.
- Synthesis Example 4 Synthesis of Vinyl Polymer and Preparation of Binder Dispersion Solution Consisting of Vinyl Polymer
- a binder made of a vinyl polymer was used in Synthesis Example 1 except that vinyl acetate (VA, manufactured by Fujifilm Wako Pure Chemical Industries, Ltd.) was used in the same number of moles instead of methyl acrylate. The dispersion was adjusted.
- This emulsion was heated at 85 ° C. for 120 minutes while flowing nitrogen gas. After the obtained residue, 50 g of heptane was added and further heated at 85 ° C. for 60 minutes. This operation was repeated 4 times to remove THF. In this way, a heptane dispersion of a binder made of polyurethane 1 was obtained.
- Synthesis Examples 6 to 10 Synthesis of Polyurethanes 2 to 6 and Preparation of Binder Dispersion Solution Consists of Polyurethanes 2 to 6
- Synthesis Example 5 the same applies to Synthesis Example 5, except that the content of the compounds leading to the constituents a to d and the constituents a to d are changed to be the compounds and the contents shown in Table 2.
- Polyurethanes 2 to 6 were synthesized, respectively.
- a dispersion of a binder composed of each polyurethane was prepared in the same manner as in Synthesis Example 5.
- Polyurethane 4 was dissolved in heptane and obtained as a solution.
- Tables 1 and 2 show the composition (content of constituent components), mass average molecular weight, and particle size of each binder of each synthesized polymer.
- the mass average molecular weight of each polymer was measured by the above method (condition 2).
- the particle size of each binder was measured by the above method.
- the polyurethane 4 is described as "dissolved” in the "particle diameter” column of Table 2.
- the constituent components A1 and A2 are constituent components derived from the (meth) acrylic acid ester compound (M1) or the vinyl compound (M2), and the constituent component A3 is a constituent component derived from the macromonomer.
- the component a corresponds to the component represented by the above formula (I-1).
- the components b to d correspond to the components represented by the above formulas (I-3A), (I-3B) or (I-3C), respectively.
- PVdF Polyvinylidene fluoride (trade name) (mass average molecular weight 180,000, manufactured by Sigma-Aldrich)
- SBR Hydrogenated styrene-butadiene rubber (DYNARON1321P (trade name), mass average molecular weight 230,000, manufactured by JSR Corporation)
- SEBS Styrene-ethylene-butylene-styrene block copolymer (trade name, mass average molecular weight 100,000, manufactured by Sigma-Aldrich)
- Li 2 S lithium sulfide
- Aldrich Corp. purity> 99.98%
- P 2 S 5. diphosphorus pentasulfide 3.90 g was weighed, placed in an agate mortar, and mixed for 5 minutes using an agate mortar.
- a sulfide-based inorganic solid electrolyte of yellow powder is obtained by setting a container on a planetary ball mill P-7 (trade name, manufactured by Fritsch) manufactured by Fritsch, and performing mechanical milling at a temperature of 25 ° C. at a rotation speed of 510 rpm for 20 hours.
- Li-PS-based glass hereinafter sometimes referred to as LPS. 6.20 g was obtained.
- the particle size of the Li-PS-based glass was 2.5 ⁇ m.
- a SE to the inorganic solid electrolyte Binder 0.5 g of the inorganic solid electrolyte (LPS) and 0.26 g of the binder used for preparing each of the inorganic solid electrolyte-containing compositions shown in Table 3 were placed in a 15 mL vial, and 25 g of DIBK was added while stirring with a mix rotor. Further, the mixture was further stirred at room temperature and 80 rpm for 30 minutes.
- Adsorption rate A SE binder is the average value of the suction rate obtained by performing twice the above measurement.
- Adsorption rate (%) [(0.26-BX ⁇ 25/2) /0.26] ⁇ 100
- the adsorption rate ASE was measured using the inorganic solid electrolyte and the binder taken out from the formed inorganic solid electrolyte layer, the same value was obtained.
- Adsorption rate A AM binder is the average value of the suction rate obtained by performing twice the above measurement.
- Adsorption rate (%) [(0.08-BY ⁇ 8/2) /0.08] ⁇ 100
- Example 1 In Example 1, an inorganic solid electrolyte-containing composition, a composition for a negative electrode layer, and a composition for a positive electrode layer were prepared using the prepared or prepared binder, and their initial dispersibility and dispersion stability were evaluated.
- Si (trade name, Silicon Powder 1 to 5 ⁇ m, manufactured by Alfa Aesar) 7.00 g
- acetylene black (trade name, AB powder, manufactured by Denka) 0.500 g
- the binder dispersion made of acrylic polymer A1 prepared in Synthesis Example 1 was 0.105 g in terms of solid content
- the binder dispersion made of acrylic polymer B prepared in Synthesis Example 3 as binder B was 0.052 g in terms of solid content.
- the container was set on the planetary ball mill P-7 and mixed at a temperature of 25 ° C. and a rotation speed of 200 rpm for 15 minutes to obtain the negative composition No. 5 was prepared.
- the negative electrode composition No. In the preparation of 5, the composition for the negative electrode was used except that the binder dispersion or the binder solution composed of the polymers shown in Table 3 was used as the binder A and the binder B, and the active material and the organic solvent shown in Table 3 were appropriately changed. No. In the same manner as in the preparation of No. 5, the negative electrode composition No. 1 to 4 and 6 to 27 were prepared, respectively. However, the negative electrode composition No. 2 using two kinds of organic solvents. In 22, 23 and 25, the ratio (mass ratio) of the organic solvent used is shown by using “/” in parentheses in the “type” column of Table 3.
- ⁇ Composition No. for positive electrode Preparation of 28 and 30> 160 zirconia beads having a diameter of 5 mm were put into a 45 mL container made of zirconia (manufactured by Fritsch), 2.00 g of LPS synthesized in Synthesis Example 11 and 12.3 g of DIBK (diisobutyl ketone) as an organic solvent were put.
- the container was set on a planetary ball mill P-7 manufactured by Fritsch, and mixed at a temperature of 25 ° C. and a rotation speed of 300 rpm for 2 hours. After that, as an active material, NMC (LiNi 1/3 Co 1/3 Mn 1/3 O 2 , positive electrode composition No.
- composition (slurry) No. for the positive electrode layer. 28 and 30 were prepared respectively.
- the positive electrode composition No. 2 was used except that the amount of LPS used was changed to 2.047 g (0.5% increase with respect to the total solid content) without using the binder B.
- the positive electrode composition No. 29 and 31 were prepared, respectively.
- Inorganic solid electrolyte-containing composition No. 32 In the preparation of No. 32, the inorganic solid electrolyte-containing composition No. 32 was prepared except that the amount of LPS used was changed to 2.01 g (0.5% increase with respect to the total solid content) without using the binder B. Inorganic solid electrolyte-containing composition No. 32, similar to the preparation of No. 32. 33 was prepared.
- the viscosity measured at a shear rate of 10 / s (rotation speed 2.5 rpm) at a temperature of 22 ° C. was defined as the slurry viscosity ⁇ , and was similarly measured at a shear rate of 200 / s (rotation speed 50 rpm).
- the viscosity was defined as the slurry viscosity ⁇ .
- the viscosity difference ⁇ - ⁇ between the slurry viscosity ⁇ and the slurry viscosity ⁇ thus obtained was calculated.
- Si Silicon Powerer (trade name, particle size 1-5 ⁇ m, manufactured by Alfa Aesar)
- Sn Tin powder (particle diameter 3.5 ⁇ m, manufactured by Alfa Aesar)
- NMC LiNi 1/3 Co 1/3 Mn 1/3 O 2 (lithium nickel manganese cobalt oxide)
- NCA LiNi 0.85 Co 0.10 Al 0.05 O 2 (Lithium Nickel Cobalt Aluminate)
- LPS Li-PS-based glass synthesized in Synthesis Example 11
- Acrylic A1 Acrylic polymer A1 synthesized in Synthesis Example 1
- Acrylic A2 Acrylic polymer A2 synthesized in Synthesis Example 2
- Acrylic B Acrylic polymer B synthesized in Synthesis Example 3
- Vinyl-based Vinyl-based polymer synthesized in Synthesis Example 4
- Urethane 1 to 6 Polyurethane 1 to 6 synthesized in Synthesis Examples 5 to 10.
- the "morphology" column of the binder A and the binder B shows the state of the binder in the inorganic solid electrolyte-containing composition.
- Dispersion stability was evaluated for each of the prepared compositions. For dispersion stability, each composition was placed in a sedimentation tube having an inner diameter of 5 mm and allowed to stand at 25 ° C. for 72 hours, and then the supernatant (supernatant) separated from the composition and the composition (maintain the dispersed state after the separation). The distance X of the interface with the (state) was measured and evaluated using the same evaluation criteria as the initial dispersibility. The results are shown in Table 4. In this test, the dispersion stability passed the evaluation rank "E" or higher.
- Example 2 In Example 2, each composition prepared in Example 1 was used to prepare a solid electrolyte sheet for an all-solid secondary battery and an electrode sheet for an all-solid secondary battery to manufacture an all-solid secondary battery.
- the composition No. 1 for the negative electrode layer immediately after preparation obtained above. 1 to 27 are each applied to a copper foil having a thickness of 20 ⁇ m with the above-mentioned baker type applicator (trade name: SA-201 baker type applicator, manufactured by Tester Sangyo Co., Ltd.) and heated at 100 ° C. for 1 hour for the negative electrode layer.
- the composition was dried, and the negative electrode sheet No. 1 for an all-solid secondary battery having a laminated structure of a negative electrode active material layer / copper foil. 1 to 27 were prepared respectively.
- the thickness of the negative electrode active material layer was 100 ⁇ m.
- composition No. 1 for the positive electrode layer immediately after preparation by an applicator (trade name: SA-201 Baker type applicator, manufactured by Tester Sangyo Co., Ltd.) on an aluminum foil having a thickness of 20 ⁇ m.
- 28 to 31 are each applied and dried by heating at 100 ° C. for 1 hour to form a positive electrode active material layer, and the positive electrode sheet No. for an all-solid secondary battery is formed. 28 to 31 were prepared respectively.
- the thickness of the positive electrode active material layer was 100 ⁇ m.
- a disk-shaped indium (In) sheet (thickness 20 ⁇ m) with a diameter of 9 mm and a disk-shaped lithium (Li) sheet (thickness 20 ⁇ m) with a diameter of 9 mm are placed in a cylinder in this order.
- the removed SUS rod was reinserted into the cylinder and fixed under a pressure of 50 MPa.
- an all-solid state having a structure of copper foil (thickness 20 ⁇ m) -negative electrode active material layer (thickness 80 ⁇ m) -solid electrolyte layer (thickness 200 ⁇ m) -counter electrode layer (In / Li sheet, thickness 30 ⁇ m).
- a secondary battery (half cell) was obtained.
- a positive electrode sheet (No. 28) for an all-solid-state secondary battery was punched into a disk shape having a diameter of 10 mm and placed in a PET cylinder having an inner diameter of 10 mm.
- Each solid electrolyte sheet for an all-solid secondary battery prepared on the positive electrode active material layer side in the cylinder was punched into a disk shape having a diameter of 10 mm and placed in the cylinder, and a 10 mm SUS rod was inserted from both end openings of the cylinder.
- the collector side of the positive electrode sheet for the all-solid-state secondary battery and the aluminum foil side of the solid electrolyte sheet for the all-solid-state secondary battery were pressurized by applying a pressure of 350 MPa with a SUS rod.
- the SUS rod on the solid electrolyte sheet side for the all-solid secondary battery is once removed, and the aluminum foil of the solid electrolyte sheet for the all-solid secondary battery is gently peeled off, and then the negative electrode sheet for the all-solid secondary battery (No. 22).
- No. 22 was punched into a disk shape having a diameter of 10 mm and inserted onto the solid electrolyte layer of the solid electrolyte sheet for an all-solid secondary battery in a cylinder.
- the removed SUS rod was reinserted into the cylinder and fixed under a pressure of 50 MPa.
- An all-solid-state secondary battery (full cell) having a configuration was obtained.
- This tape is peeled off at a speed of 30 mm / s at an angle of 90 ° with respect to the surface of the solid electrolyte layer or the active material layer using a pressure crimping) and a tensile tester (ZTS-50N, manufactured by Imada).
- the average peeling force at the time was defined as the peeling strength (unit: N / mm).
- the measured peel strength was applied to the following evaluation criteria to evaluate the binding property of the solid electrolyte layer or the active material layer.
- the peel strength is 0.1 N / mm or more (evaluation levels A and B), the binding property of the solid particles in the solid electrolyte layer or the active material layer, and further the active material layer and the current collector It can be said that it has excellent binding properties.
- the peel strength is 0.15 N / mm or more (evaluation level A).
- Discharge capacity retention rate (%) (Discharge capacity in the 20th cycle / Discharge capacity in the 1st cycle) x 100
- -Evaluation criteria- AA 80% or more, 100% or less A: 70% or more, less than 80% B: 60% or more, less than 70% C: 50% or more, less than 60% D: 40% or more, less than 50% E: 30% or more , Less than 40% F: 20% or more, less than 30% G: less than 20%
- the inorganic solid electrolyte-containing composition that does not contain the two types of binders specified in the present invention may be inferior in dispersion stability and may not have sufficient initial dispersibility. Therefore, the battery performance of the all-solid-state secondary battery (particularly the battery characteristics after being left for 72 hours) and the binding property are not satisfactory.
- the inorganic solid electrolyte-containing composition containing the two types of binders specified in the present invention is excellent not only in initial dispersibility but also in dispersion stability.
- each sheet prepared using these inorganic solid electrolyte-containing compositions has strong binding properties between solid particles and further, a binding property between the active material layer and the current collector, and these sheets are composed of the constituent layers.
- the all-solid-state secondary battery provided as a battery exhibits high battery performance (discharge capacity retention rate).
- an all-solid-state battery having a solid electrolyte layer or active material formed using the composition immediately after preparation even if the solid electrolyte layer or active material formed using the composition left to stand for 72 hours after preparation is provided. Since the deterioration of the battery characteristics can be effectively suppressed with respect to the next battery, it can be seen that the composition is excellent in dispersion stability (prevention of deterioration) and agrees with the result of the above dispersion stability test.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Manufacturing & Machinery (AREA)
- Physics & Mathematics (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Inorganic Chemistry (AREA)
- Materials Engineering (AREA)
- Secondary Cells (AREA)
- Conductive Materials (AREA)
Abstract
Description
<1>周期律表第1族若しくは第2族に属する金属のイオンの伝導性を有する無機固体電解質と、バインダーと、有機溶媒とを含有する無機固体電解質含有組成物であって、
バインダーが、上記無機固体電解質に対する吸着率が異なる少なくとも2種のバインダーA及びバインダーBを含み、
バインダーAの吸着率が15%以上であり、バインダーBの吸着率が10%未満である、無機固体電解質含有組成物。
<2>バインダーAの吸着率が50%以上であり、バインダーBの吸着率が5%未満である、<1>に記載の無機固体電解質含有組成物。
<3>バインダーA及びバインダーBの少なくとも一方が粒子状である、<1>又は<2>に記載の無機固体電解質含有組成物。
<4>バインダーAが粒子状である、<1>~<3>のいずれか1つに記載の無機固体電解質含有組成物。
<5>バインダーAの、非極性溶媒に対する溶解度が1質量%以下である、<1>~<4>のいずれか1つに記載の無機固体電解質含有組成物。
<6>バインダーBの、非極性溶媒に対する溶解度が2質量%以上である、<1>~<5>のいずれか1つに記載の無機固体電解質含有組成物。
<7>無機固体電解質含有組成物中の、バインダーAの含有量が、無機固体電解質含有組成物中の、バインダーBの含有量よりも高い、<1>~<6>のいずれか1つに記載の無機固体電解質含有組成物。
<8>バインダーBを形成するポリマーが、フッ素系ポリマー、炭化水素系ポリマー、ポリウレタン又は(メタ)アクリルポリマーである、<1>~<7>のいずれか1つに記載の無機固体電解質含有組成物。
<9>バインダーAを形成するポリマーが、ポリウレタン又は(メタ)アクリルポリマーである、<1>~<8>のいずれか1つに記載の無機固体電解質含有組成物。
<10>有機溶媒が、少なくとも2種の有機溶媒を含む、<1>~<9>のいずれか1つに記載の無機固体電解質含有組成物。
<11>有機溶媒が、ClogP値が1.5以上3未満である有機溶媒を少なくとも1種含む、<1>~<10>のいずれか1つに記載の無機固体電解質含有組成物。
<12>有機溶媒が、ClogP値が3以上6未満である有機溶媒と、ClogP値が1.5以上3未満である有機溶媒とを含む、<1>~<11>のいずれか1つに記載の無機固体電解質含有組成物。
<13>上記有機溶媒の少なくとも1種が、エステル化合物、ケトン化合物又はエーテル化合物から選択される少なくとも1種を含む、<1>~<12>のいずれか1つに記載の無機固体電解質含有組成物。
<14>せん断速度10/sのスラリー粘度ηαと、せん断速度200/sのスラリー粘度ηβとの粘度差ηα-ηβが400mPa・s以上である、<1>~<13>のいずれか1つに記載の無機固体電解質含有組成物。
<15>活物質を含有する、<1>~<14>のいずれか1つに記載の無機固体電解質含有組成物。
<16>バインダーAの、上記活物質への吸着率が10%以上である、<15>に記載の無機固体電解質含有組成物。
<17>バインダーBの、集電体に対する剥離強度が0.1N/mm以上である、<15>又は<16>に記載の無機固体電解質含有組成物。
<19>上記<15>~<17>のいずれか1つに記載の無機固体電解質含有組成物で構成した活物質層を有する全固体二次電池用電極シート。
<20>正極活物質層と固体電解質層と負極活物質層とをこの順で具備する全固体二次電池であって、
正極活物質層、固体電解質層及び負極活物質層の少なくとも1つの層が、上記<1>~<17>のいずれか1つに記載の無機固体電解質含有組成物で構成した層である、全固体二次電池。
<21>上記<1>~<17>のいずれか1つに記載の無機固体電解質含有組成物を製膜する、全固体二次電池用シートの製造方法。
<22>上記<21>に記載の製造方法を経て全固体二次電池を製造する、全固体二次電池の製造方法。
本明細書において化合物の表示(例えば、化合物と末尾に付して呼ぶとき)については、この化合物そのもののほか、その塩、そのイオンを含む意味に用いる。また、本発明の効果を損なわない範囲で、置換基を導入するなど一部を変化させた誘導体を含む意味である。
本発明において、(メタ)アクリルとは、アクリル及びメタアクリルの一方又は両方を意味する。(メタ)アクリレートについても同様である。
本明細書において、置換又は無置換を明記していない置換基、連結基等(以下、置換基等という。)については、その基に適宜の置換基を有していてもよい意味である。よって、本明細書において、単に、YYY基と記載されている場合であっても、このYYY基は、置換基を有しない態様に加えて、更に置換基を有する態様も包含する。これは置換又は無置換を明記していない化合物についても同義である。好ましい置換基としては、例えば後述する置換基Zが挙げられる。
本明細書において、特定の符号で示された置換基等が複数あるとき、又は複数の置換基等を同時若しくは択一的に規定するときには、それぞれの置換基等は互いに同一でも異なっていてもよいことを意味する。また、特に断らない場合であっても、複数の置換基等が隣接するときにはそれらが互いに連結したり縮環したりして環を形成していてもよい意味である。
本発明の無機固体電解質含有組成物は、周期律表第1族若しくは第2族に属する金属のイオンの伝導性を有する無機固体電解質と、バインダーと、有機溶媒とを含有する。
本発明の無機固体電解質含有組成物は、バインダーとして、この組成物が含有する無機固体電解質に対する吸着率が互いに異なる少なくとも2種のバインダーA及びバインダーBを含有している。このバインダーA及びバインダーBは、それぞれ後述する特定の吸着率を有し、かつ両バインダーの吸着率の差が少なくとも5%となっている。
本発明の無機固体電解質含有組成物が含有するバインダーは、無機固体電解質含有組成物中において、無機固体電解質(更には、共存しうる、活物質、導電助剤)等の固体粒子同士(例えば、無機固体電解質同士、無機固体電解質と活物物質、活物質同士)を結着させ、更には、有機溶媒中に安定して分散させる機能を有する。また、本発明の無機固体電解質含有組成物が含有するバインダーは、少なくとも無機固体電解質含有組成物で形成した固体電解質層中において、固体粒子同士を結着させ、更には集電体と固体粒子とを結着させる結着剤として、機能する。
その理由の詳細はまだ明らかではないが、次のように考えられる。すなわち、高い吸着率を示すバインダーAと低い吸着率を示すバインダーBとを少なくとも5%の吸着率の差となる組み合わせで併用することにより、各バインダーに、有機溶媒中における、固体粒子の結着性向上又は分散性改善を機能分化させて発現させることがきる。その結果、調製直後だけでなく経時においても、固体粒子の過度な(再)凝集及び沈殿等を抑制できる。これにより、無機固体電解質含有組成物の初期分散性と分散安定性とを両立することができると考えられる。また、本発明の無機固体電解質含有組成物は、構成層としたときに固体粒子を強固に結着させることもできる。しかも、この無機固体電解質含有組成物で活物質層を形成すると、固体粒子同士の結着に加えて、固体粒子と集電体とを強固に結着させることもできる。このような、無機固体電解質含有組成物の初期分散性及び分散安定性の改善、更には固体粒子の結着性等の強化により、本発明の無機固体電解質含有組成物を用いて形成した構成層を備えた全固体二次電池は優れた電池性能(サイクル特性)を示す。
本発明において、スラリー粘度ηαは無機固体電解質含有組成物が高粘度状態にある場合の固体粒子の再凝集若しくは沈降のしにくさを示す指標として、スラリー粘度ηβは無機固体電解質含有組成物を製膜(特に塗布)する際の無機固体電解質含有組成物の(分散)均一性の維持しやすさを示す指標として、用いる。スラリー粘度ηα及びηβは実施例に記載の方法により算出される値とする。
この無機固体電解質含有組成物が上記範囲の粘度差を示すことにより、高粘度状態の分散性と製膜時での分散性を更に高めて、全固体二次電池等に優れた電池性能を付与できる。
粘度差ηα-ηβは、初期分散性及び分散安定性の更なる改善の点で、500mPa・s以上であることが好ましく、700mPa・s以上であることがより好ましく、800mPa・s以上であることが更に好ましい。粘度差ηα-ηβの上限は、特に制限されないが、製膜ムラ抑制の点で、100,000mPa・s以下であることが好ましく、2,000mPa・s以下であることがより好ましい。
スラリー粘度ηα及びスラリー粘度ηβは、それぞれ、上記粘度差ηα-ηβを満たす範囲で適宜に設定され、特に制限されない。例えば、スラリー粘度ηαは、600~20,000mPa・sであることが好ましく、800~2,500mPa・sであることがより好ましい。スラリー粘度ηβは、100~3,000mPa・sであることが好ましく、200~1,000mPa・sであることがより好ましい。
スラリー粘度は、無機固体電解質含有組成物の固形分濃度、固体粒子若しくはバインダーの種類若しくは含有量、有機溶媒の種類等、更には分散条件等の、変更若しくは調整により、適宜に設定できる。
本発明の無機固体電解質含有組成物は、無機固体電解質を含有する。
本発明において、無機固体電解質とは、無機の固体電解質のことであり、固体電解質とは、その内部においてイオンを移動させることができる固体状の電解質のことである。主たるイオン伝導性材料として有機物を含むものではないことから、有機固体電解質(ポリエチレンオキシド(PEO)などに代表される高分子電解質、リチウムビス(トリフルオロメタンスルホニル)イミド(LiTFSI)などに代表される有機電解質塩)とは明確に区別される。また、無機固体電解質は定常状態では固体であるため、通常カチオン及びアニオンに解離又は遊離していない。この点で、電解液、又は、ポリマー中でカチオン及びアニオンに解離若しくは遊離している無機電解質塩(LiPF6、LiBF4、リチウムビス(フルオロスルホニル)イミド(LiFSI)、LiClなど)とも明確に区別される。無機固体電解質は周期律表第1族若しくは第2族に属する金属のイオンの伝導性を有するものであれば、特に限定されず、電子伝導性を有さないものが一般的である。本発明の全固体二次電池がリチウムイオン電池の場合、無機固体電解質は、リチウムイオンのイオン伝導性を有することが好ましい。
上記無機固体電解質は、全固体二次電池に通常使用される固体電解質材料を適宜選定して用いることができる。例えば、無機固体電解質としては、(i)硫化物系無機固体電解質、(ii)酸化物系無機固体電解質、(iii)ハロゲン化物系無機固体電解質、及び、(iV)水素化物系固体電解質が挙げられ、活物質と無機固体電解質との間により良好な界面を形成することができる観点から、硫化物系無機固体電解質が好ましい。
硫化物系無機固体電解質は、硫黄原子を含有し、かつ、周期律表第1族若しくは第2族に属する金属のイオン伝導性を有し、かつ、電子絶縁性を有するものが好ましい。硫化物系無機固体電解質は、元素として少なくともLi、S及びPを含有し、リチウムイオン伝導性を有しているものが好ましいが、目的又は場合に応じて、Li、S及びP以外の他の元素を含んでもよい。
La1Mb1Pc1Sd1Ae1 (S1)
式中、LはLi、Na及びKから選択される元素を示し、Liが好ましい。Mは、B、Zn、Sn、Si、Cu、Ga、Sb、Al及びGeから選択される元素を示す。Aは、I、Br、Cl及びFから選択される元素を示す。a1~e1は各元素の組成比を示し、a1:b1:c1:d1:e1は1~12:0~5:1:2~12:0~10を満たす。a1は1~9が好ましく、1.5~7.5がより好ましい。b1は0~3が好ましく、0~1がより好ましい。d1は2.5~10が好ましく、3.0~8.5がより好ましい。e1は0~5が好ましく、0~3がより好ましい。
硫化物系無機固体電解質は、例えば硫化リチウム(Li2S)、硫化リン(例えば五硫化二燐(P2S5))、単体燐、単体硫黄、硫化ナトリウム、硫化水素、ハロゲン化リチウム(例えばLiI、LiBr、LiCl)及び上記Mで表される元素の硫化物(例えばSiS2、SnS、GeS2)の中の少なくとも2つ以上の原料の反応により製造することができる。
酸化物系無機固体電解質は、酸素原子を含有し、かつ、周期律表第1族若しくは第2族に属する金属のイオン伝導性を有し、かつ、電子絶縁性を有するものが好ましい。
酸化物系無機固体電解質は、イオン伝導度として、1×10-6S/cm以上であることが好ましく、5×10-6S/cm以上であることがより好ましく、1×10-5S/cm以上であることが特に好ましい。上限は特に制限されないが、1×10-1S/cm以下であることが実際的である。
またLi、P及びOを含むリン化合物も望ましい。例えばリン酸リチウム(Li3PO4); リン酸リチウムの酸素の一部を窒素で置換したLiPON; LiPOD1(D1は、好ましくは、Ti、V、Cr、Mn、Fe、Co、Ni、Cu、Zr、Nb、Mo、Ru、Ag、Ta、W、Pt及びAuから選ばれる1種以上の元素である。)等が挙げられる。
更に、LiA1ON(A1は、Si、B、Ge、Al、C及びGaから選ばれる1種以上の元素である。)等も好ましく用いることができる。
ハロゲン化物系無機固体電解質は、ハロゲン原子を含有し、かつ、周期律表第1族若しくは第2族に属する金属のイオンの伝導性を有し、かつ、電子絶縁性を有する化合物が好ましい。
ハロゲン化物系無機固体電解質としては、特に制限されないが、例えば、LiCl、LiBr、LiI、ADVANCED MATERIALS,2018,30,1803075に記載のLi3YBr6、Li3YCl6等の化合物が挙げられる。中でも、Li3YBr6、Li3YCl6を好ましい。
水素化物系無機固体電解質は、水素原子を含有し、かつ、周期律表第1族若しくは第2族に属する金属のイオン伝導性を有し、かつ、電子絶縁性を有する化合物が好ましい。
水素化物系無機固体電解質としては、特に制限されないが、例えば、LiBH4、Li4(BH4)3I、3LiBH4-LiCl等が挙げられる。
無機固体電解質の粒子径の測定は、以下の手順で行う。無機固体電解質粒子を、水(水に不安定な物質の場合はジイソブチルケトン(DIBK))を用いて20mLサンプル瓶中で1質量%の分散液を希釈調製する。希釈後の分散液試料は、1kHzの超音波を10分間照射し、その直後に試験に使用する。この分散液試料を用い、レーザ回折/散乱式粒度分布測定装置LA-920(商品名、HORIBA社製)を用いて、温度25℃で測定用石英セルを使用してデータ取り込みを50回行い、体積平均粒子径を得る。その他の詳細な条件等は必要によりJIS Z 8828:2013「粒子径解析-動的光散乱法」の記載を参照する。1水準につき5つの試料を作製しその平均値を採用する。
固体電解質層を形成する場合、固体電解質層の単位面積(cm2)当たりの無機固体電解質の質量(mg)(目付量)は特に制限されるものではない。設計された電池容量に応じて、適宜に決めることができ、例えば、1~100mg/cm2とすることができる。
ただし、無機固体電解質含有組成物が後述する活物質を含有する場合、無機固体電解質の目付量は、活物質と無機固体電解質との合計量が上記範囲であることが好ましい。
ただし、無機固体電解質含有組成物が後述する活物質を含有する場合、無機固体電解質含有組成物中の無機固体電解質の含有量は、活物質と無機固体電解質との合計含有量が上記範囲であることが好ましい。
本明細書において、固形分(固形成分)とは、無機固体電解質含有組成物を、1mmHgの気圧下、窒素雰囲気下150℃で6時間乾燥処理したときに、揮発若しくは蒸発して消失しない成分をいう。典型的には、後述の有機溶媒以外の成分を指す。
本発明の無機固体電解質含有組成物は、少なくとも、無機固体電解質に対する吸着率が15%以上のバインダー(バインダーAともいう。)と、無機固体電解質に対する吸着率が10%未満のバインダー(バインダーBともいう。)とを含んでいればよく、その他のバインダー(例えば、無機固体電解質に対する吸着率が10%以上15%未満のバインダー)を含有していてもよい。本発明において、バインダーA、バインダーB及びその他のバインダーは、それぞれ、1種でもよく、複数種でもよい。
本発明の無機固体電解質含有組成物が含有するバインダー数は、2種以上であればよく、通常、2~4種が好ましく、1種のバインダーAと1種のバインダーBとの2種であることがより好ましい。
したがって、無機固体電解質含有組成物が複数種の無機固体電解質を含有する場合、無機固体電解質含有組成物中の無機固体電解質の組成(種類及び含有量)と同じ組成を有する無機固体電解質に対する吸着率となる。バインダーA又はバインダーBを複数種用いる場合も、同様に、複数種のバインダーについての吸着率とする。
本発明において、バインダーの吸着率は実施例に記載の方法により算出される値とする。
本発明において、無機固体電解質に対する吸着率は、バインダーを形成するポリマーの種類(ポリマー鎖の構造及び組成)、ポリマーが有する官能基の種類若しくは含有量、バインダーの形態(粒子状バインダー若しくは溶解型バインダー)等により、適宜に設定できる。
バインダーAの吸着率は、15%以上であり、25%以上が好ましく、50%以上がより好ましく、55%以上が更に好ましい。バインダーAが上記吸着率を示すと、無機固体電解質含有組成物中において無機固体電解質に吸着して結着体を形成し、無機固体電解質の結着性を強化できる。一方、吸着率の上限は、特に制限されないが、実際には99.9%であり、分散性の点で、70%以下が好ましい。
バインダーAは、非極性溶媒(通常、DIBK)に対する溶解度について、特に制限されず、非極性溶媒に可溶性でも不溶性でもよい。本発明において、バインダーAは、結着性改善、更には全固体二次電池の抵抗低減の点で、非極性溶媒に対する溶解度が2質量%以下であることが好ましく、1質量%以下であることがより好ましい。このような溶解度を示すバインダーAは、非極性溶媒に不溶となり、少なくとも無機固体電解質含有組成物中において後述する粒子状バインダーであることが好ましい。本発明において、バインダーの溶解度は実施例に記載の方法により算出される値とする。
無機固体電解質含有組成物において、バインダーAの含有量は、後述するバインダーBの含有量に対して同じでも低くてもよいが、高いことが好ましい。これにより、無機固体電解質含有組成物の結着性及び分散性を改善でき、更に全固体二次電池の抵抗上昇を抑制できる。バインダーAとバインダーBとの含有量の差(バインダーAの含有量-バインダーBの含有量)は、特に制限されず、例えば、-5.0~5.0質量%とすることができ、0.0質量%以上が好ましく、0.0~4.0質量%がより好ましく、0.2~2.0質量%が更に好ましい。
また、バインダーAとバインダーBとの含有量の比(バインダーAの含有量/バインダーBの含有量)は、特に制限されないが、例えば、0.5~30であることが好ましく、1.2~10であることがより好ましい。
バインダーBの、無機固体電解質に対する吸着率は、10%未満であり、8%未満が好ましく、5%未満がより好ましく、4%以下が更に好ましく、0%以上3%以下が特に好ましい。バインダーBが上記吸着率を示すと、無機固体電解質含有組成物中において無機固体電解質に吸着しにくく凝集体の過度な形成を抑制して、分散性を高めることができる。
バインダーBは、非極性溶媒(通常、DIBK)に対する溶解度について、特に制限されず、非極性溶媒に可溶性でも不溶性でもよい。本発明において、バインダーBは、分散性及び結着性の点で、非極性溶媒に対する溶解度が2質量%以上であることが好ましく、3質量%以上であることがより好ましく、5質量%以上であることが更に好ましい。一方、この溶解度の上限値は、特に制限されず、例えば、25質量%とすることができ、20質量%以下であることが好ましい。このような溶解度を示すバインダーBは、無機固体電解質含有組成物中において、後述する溶解型バインダーであることが好ましい。
本発明の無機固体電解質含有組成物が活物質を含有する場合、バインダーBの、集電体に対する剥離強度は、特に制限されない。例えば集電体との高い密着性が得られる点で、0.1N/mm以上であることが好ましく、0.2N/mm以上であることがより好ましく、0.3N/mm以上であることが更に好ましい。上限は、特に限定されないが、例えば、10N/mm以下であることが実際的であり、2.0N/mm以下であることが好ましい。上記剥離強度の対象となる集電体は、特に制限されず後述するものであればよいが、銅箔、アルミニウム箔、ステンレス鋼(SUS)箔等が挙げられる。剥離強度は、集電体の種類に関わらず、実施例に記載の方法(銅箔に対する剥離強度)と同様にして算出される値とする。本発明において、剥離強度は無機固体電解質に対する吸着率と同様にして、適宜に設定できる。
本発明において、バインダーA及びバインダーBの合計質量に対する、無機固体電解質と活物質の合計質量(総量)の質量比[(無機固体電解質の質量+活物質の質量)/(バインダーAの質量+バインダーBの質量)]は、1,000~1の範囲が好ましい。この比率は更に500~2がより好ましく、100~10が更に好ましい。
バインダーAとバインダーBとの組み合わせは、特に制限されないが、結着性と分散性との点で、バインダーAの無機固体電解質に対する吸着率とバインダーBの無機固体電解質に対する吸着率との差(バインダーAの吸着率-バインダーBの吸着率)が5%よりも大きくなる組み合わせが好ましい。バインダーAとバインダーBとの組み合わせは、結着性と分散性とをバランスよく両立できる点で、両バインダーの吸着率の差が、10%以上となる組み合わせがより好ましく、45%以上となる組み合わせがより一層好ましく、50%以上となる組み合わせが更に好ましく、55%以上となる組み合わせが特に好ましい。この吸着率の差の上限値は、特に制限されず、例えば、80%とすることができる。各吸着率の差となる組み合わせとしては、上記バインダーAと上記バインダーBとを適宜に組み合わせることができ、例えば、吸着率の差が45%以上となる組み合わせとして、吸着率が50%以上のバインダーAと吸着率が5%未満のバインダーBとの組み合わせが挙げられる。
併用されるバインダーA及びバインダーBの上記溶解度は、適宜に設定され、特に制限されないが、全固体二次電池の抵抗低減の点で、好ましくは、バインダーA及びバインダーBの少なくとも一方が非極性溶媒に不溶性(粒子状バインダー)となるように設定され、より好ましくは、バインダーAが非極性溶媒に不溶性となり、バインダーBが非極性溶媒に可溶性となるように、設定される。
ポリマー種類における、バインダーAとバインダーBとの組み合わせについては後述する。
バインダーA及びBを形成するポリマーは、無機固体電解質に対する上記吸着率を満たす限り、特に制限されず、例えば、ポリウレタン、ポリウレア、ポリアミド、ポリイミド、ポリエステル、ポリエーテル、ポリカーボネート等の逐次重合(重縮合、重付加若しくは付加縮合)系ポリマー、更には、フッ素系ポリマー(含フッ素ポリマー)、炭化水素系ポリマー、ビニル系ポリマー、(メタ)アクリルポリマー等の連鎖重合系ポリマーが挙げられる。
バインダーAを形成するポリマーとしては、ポリウレタン又は(メタ)アクリルポリマーが好ましく、分散性及び結着性の点で、ポリウレタンがより好ましい。バインダーBを形成するポリマーとしては、分散性及び結着性の点で、フッ素系ポリマー、炭化水素系ポリマー、ポリウレタン又は(メタ)アクリルポリマーが好ましく、フッ素系ポリマー、炭化水素系ポリマーがより好ましい。バインダーAとバインダーBとの組み合わせは、各バインダーを形成する上記好ましいポリマー同士の組み合わせが挙げられる。
- 逐次重合系ポリマー -
逐次重合系ポリマーは、ポリマーの主鎖に、ウレタン結合、ウレア結合、アミド結合、イミド結合、エステル結合、エーテル結合及びカーボネート結合のうちの少なくとも1つの結合を有している。このような結合を有するポリマーとしては、ポリウレタン、ポリウレア、ポリアミド、ポリイミド、ポリエステル、ポリエーテル及びポリカーボネートの各ポリマー、又は、これらの共重合体が挙げられる。共重合体は、上記各ポリマーをセグメントとするブロック共重合体、上記各ポリマーのうち2つ以上のポリマーを構成する各構成成分がランダムに結合したランダム共重合体でもよい。
主鎖が含む上記結合は、水素結合を形成することにより、全固体二次電池等の構成層中の固体粒子等の結着性向上に寄与する。これらの結合がポリマー内で水素結合を形成する場合、水素結合は、上記結合同士で形成されてもよく、上記結合と主鎖が有するそれ以外の部分構造とで形成されてもよい。上記結合は、互いに水素結合を形成可能な点で、水素結合を形成する水素原子を有していること(各結合の窒素原子が無置換であること)が好ましい。
上記結合は、ポリマーの主鎖中に含まれる限り特に制限されるものでなく、構成単位(繰り返し単位)中に含まれる態様及び/又は異なる構成単位同士を繋ぐ結合として含まれる態様のいずれでもよい。また、主鎖に含まれる上記結合は、1種に限定されず、2種以上であってもよい。この場合、主鎖の結合様式は、特に制限されず、2種以上の結合をランダムに有していてもよく、特定の結合を有するセグメントと他の結合を有するセグメントとのセグメント化された主鎖でもよい。
RP1及びRP2としてとりうる上記分子鎖は、特に制限されないが、炭化水素鎖、ポリアルキレンオキシド鎖、ポリカーボネート鎖又はポリエステル鎖が好ましく、炭化水素鎖又はポリアルキレンオキシド鎖がより好ましく、炭化水素鎖、ポリエチレンオキシド鎖又はポリプロピレンオキシド鎖が更に好ましい。
低分子量の炭化水素鎖は、通常の(非重合性の)炭化水素基からなる鎖であり、この炭化水素基としては、例えば、脂肪族若しくは芳香族の炭化水素基が挙げられ、具体的には、アルキレン基(炭素数は1~12が好ましく、1~6がより好ましく、1~3が更に好ましい)、アリーレン基(炭素数は6~22が好ましく、6~14が好ましく、6~10がより好ましい)、又はこれらの組み合わせからなる基が好ましい。RP2としてとりうる低分子量の炭化水素鎖を形成する炭化水素基としては、アルキレン基がより好ましく、炭素数2~6のアルキレン基が更に好ましく、炭素数2又は3のアルキレン基が特に好ましい。
芳香族の炭化水素基は、例えば、後掲する各例示の構成成分が有する炭化水素基が挙げられ、フェニレン基又は下記式(M2)で表される炭化水素基が好ましい。
RM2~RM5は、それぞれ、水素原子又は置換基を示し、水素原子が好ましい。RM2~RM5としてとりうる置換基としては、特に制限されないが、例えば、炭素数1~20のアルキル基、炭素数1~20のアルケニル基、-ORM6、―N(RM6)2、-SRM6(RM6は置換基を示し、好ましくは炭素数1~20のアルキル基又は炭素数6~10のアリール基を示す。)、ハロゲン原子(例えば、フッ素原子、塩素原子、臭素原子)が挙げられる。-N(RM6)2としては、アルキルアミノ基(炭素数は、1~20が好ましく、1~6がより好ましい)又はアリールアミノ基(炭素数は、6~40が好ましく、6~20がより好ましい)が挙げられる。
末端反応性基を有する炭化水素ポリマーとしては、例えば、いずれも商品名で、NISSO-PBシリーズ(日本曹達社製)、クレイソールシリーズ(巴工業社製)、PolyVEST-HTシリーズ(エボニック社製)、poly-bdシリーズ(出光興産社製)、poly-ipシリーズ(出光興産社製)、EPOL(出光興産社製)及びポリテールシリーズ(三菱化学社製)等が好適に用いられる。
ポリカーボネート鎖又はポリエステル鎖としては、公知のポリカーボネート又はポリエステルからなる鎖が挙げられる。
ポリアルキレンオキシド鎖、ポリカーボネート鎖又はポリエステル鎖は、それぞれ、末端にアルキル基(炭素数は1~12が好ましく、1~6がより好ましい)を有することが好ましい。
RP1及びRP2としてとりうるポリアルキレンオキシド鎖、ポリカーボネート鎖及びポリエステル鎖の末端は、RP1及びRP2として上記各式で表される構成成分に組み込み可能な通常の化学構造に適宜に変更することができる。例えば、実施例で合成したポリウレタン1~6のように、ポリアルキレンオキシド鎖の末端酸素原子は取り除かれて上記構成成分のRP1又はRP2として組み込まれる。
上記各式において、RP1及びRP2は2価の分子鎖であるが、少なくとも1つの水素原子が-NH-CO-、-CO-、-O-、-NH-又は-N<で置換されて、3価以上の分子鎖となっていてもよい。
RP2は、上記分子鎖の中でも、低分子量の炭化水素鎖(より好ましくは脂肪族の炭化水素基)、又は低分子量の炭化水素鎖以外の分子鎖が好ましく、低分子量の炭化水素鎖及び低分子量の炭化水素鎖以外の分子鎖をそれぞれ含む態様がより好ましい。この態様においては、式(I-3)、式(I-4)及び式(I-6)のいずれかで表される構成成分は、RP2が低分子量の炭化水素基鎖である構成成分と、RP2が低分子量の炭化水素鎖以外の分子鎖である構成成分の少なくとも2種を含む。
なお、下記具体例において、構成成分中に繰り返し構造を有する場合、その繰り返し数は1以上の整数であり、上記分子鎖の分子量又は炭素原子数を満たす範囲で適宜に設定される。
なお、バインダーを構成するポリマー中における上記各式で表される構成成分の含有量は後述する。
逐次重合系ポリマー中の、上記各式で表される構成成分以外の構成成分の含有量は、特に限定されないが、50質量%以下であることが好ましい。
すなわち、逐次重合系ポリマー中の、式(I-1)若しくは式(I-2)で表される構成成分、又は式(I-5)で表されるカルボン酸二無水物由来の構成成分の含有量は、特に制限されず、10~50モル%であることが好ましく、20~50モル%であることがより好ましく、30~50モル%であることが更に好ましい。
逐次重合系ポリマー中の、式(I-3)、式(I-4)又は式(I-6)で表される構成成分の含有量は、特に制限されず、10~50モル%であることが好ましく、20~50モル%であることがより好ましく、30~50モル%であることが更に好ましい。
式(I-3)又は式(I-4)で表される構成成分のうち、RP2が分子鎖として上記ポリアルキレンオキシド鎖である構成成分(例えば上記式(I-3B)で表される構成成分)の、逐次重合系ポリマー中の含有量は、特に制限されないが、例えば、0~50モル%であることが好ましく、10~45モル%であることがより好ましく、20~43モル%であることが更に好ましい。
式(I-3)又は式(I-4)で表される構成成分のうち、RP2が分子鎖として上記炭化水素ポリマー鎖である構成成分(例えば上記式(I-3C)で表される構成成分)の、逐次重合系ポリマー中の含有量は、特に制限されないが、例えば、0~50モル%であることが好ましく、1~45モル%であることがより好ましく、3~40モル%であることが更に好ましく、3~30モル%であることが更に好ましく、3~20モル%であることが更に好ましく、3~10モル%であることが更に好ましい。
バインダーを形成するポリマーとして好適な連鎖重合系のポリマーは、非芳香族性の炭素-炭素二重結合を有する1種又は2種以上のモノマーが連鎖重合してなるポリマーである。中でも、上述の、含フッ素ポリマー、炭化水素系ポリマー、ビニル系ポリマー又は(メタ)アクリルポリマーが好ましく、(メタ)アクリルポリマーがより好ましい。
ビニル系ポリマーとしては、(メタ)アクリル化合物(M1)以外のビニル系モノマーを例えば50モル%以上含有するポリマーが挙げられる。ビニル系モノマーとしては、後述するビニル化合物等が挙げられる。ビニル系ポリマーとしては、具体的には、例えば、ポリビニルアルコール、ポリビニルアセタール、ポリ酢酸ビニル、又はこれらを含む共重合体等が挙げられる。
このビニル系ポリマーは、ビニル系モノマー由来の構成成分以外に、後述する(メタ)アクリルポリマーを形成する(メタ)アクリル化合物(M1)由来の構成成分、更には後述するマクロモノマーに由来する構成成分(MM)を有することが好ましい。ビニル系モノマー由来の構成成分の含有量は、(メタ)アクリルポリマーにおける(メタ)アクリル化合物(M1)由来の構成成分の含有量と同じであることが好ましい。(メタ)アクリル化合物(M1)由来の構成成分の含有量は、ポリマー中、50モル%未満であれば特に制限されないが、0~40モル%であることが好ましく、5~35モル%であることがより好ましい。構成成分(MM)の含有量は(メタ)アクリルポリマーにおける含有量と同じであることが好ましい。
(メタ)アクリルポリマー中におけるその他の重合性化合物(M2)の含有量は、特に制限されないが、例えば50モル%未満とすることができる。
酸素原子を含有する脂肪族複素環基は、エポキシ基含有基、オキセタン基含有基、テトラヒドロフリル基含有基などが好ましい。
R3は、R2と同義である。
L2は、連結基であり、上記L1と同義である。
L3は、連結基であり、上記L1と同義であるが、炭素数1~6(好ましくは1~3)のアルキレン基が好ましい。
mは1~200の整数であり、1~100の整数であることが好ましく、1~50の整数であることがより好ましい。
また、式(b-1)~(b-3)において、アルキル基、アリール基、アルキレン基、アリーレン基など置換基を取ることがある基については、本発明の効果を損なわない範囲で置換基を有していてもよい。置換基としては、例えば後述する置換基Z及び官能基群(I)から選択される基が挙げられ、具体的には、ハロゲン原子、ヒドロキシ基、スルファニル基、アシル基、アシルオキシ基、アルコキシ基、アリールオキシ基、アリーロイル基、アリーロイルオキシ基等が挙げられる。
-SP値の定義-
本発明において、SP値は、特に断らない限り、Hoy法によって求める(H.L.Hoy JOURNAL OF PAINT TECHNOLOGY Vol.42,No.541,1970,76-118、及びPOLYMER HANDBOOK 4th、59章、VII 686ページ Table5、Table6及びTable6中の下記式参照)。また、SP値については単位を省略して示しているが、その単位はcal1/2cm-3/2である。なお、構成成分(MM)のSP値は、マクロモノマーのSP値とほぼ変わらず、それにより評価してもよい。
本発明において、ポリマーのSP値(SPP)は、ポリマーを構成する各繰り返し単位のSP値を、それぞれ、SP1、SP2・・・、各繰り返し単位の質量分率をW1、W2・・・とした場合、下記式で算出される値とする。
SPp 2=(SP1 2×W1)+(SP2 2×W2)+・・・
(メタ)アクリル樹脂の鎖は、(メタ)アクリル酸化合物、(メタ)アクリル酸エステル化合物及び(メタ)アクリロニトリル化合物から選ばれる(メタ)アクリル化合物に由来する構成成分を含むことが好ましく、2種以上の(メタ)アクリル化合物の重合体であってもよい。(メタ)アクリル化合物は上記(メタ)アクリル化合物(M1)と同義である。ポリシロキサン鎖は、特に限定されないが、アルキル基若しくはアリール基を有するシロキサンの重合体が挙げられる。炭化水素鎖としては、上述した炭化水素系ポリマーからなる鎖が挙げられる。
本発明において、連結基を構成する原子の数は、1~36であることが好ましく、1~24であることがより好ましく、1~12であることが更に好ましく、1~6であることが特に好ましい。連結基の連結原子数は10以下であることが好ましく、8以下であることがより好ましい。下限としては、1以上である。上記連結原子数とは所定の構造部間を結ぶ最少の原子数をいう。例えば、-CH2-C(=O)-O-の場合、連結基を構成する原子の数は6となるが、連結原子数は3となる。
*の結合位置の先に存在する構造部としては、マクロモノマーとしての分子量を満たせば特に限定されないが、(好ましくは連結基を介して結合してもよい)上記重合鎖が好ましい。このとき、連結基及び重合鎖はそれぞれ置換基Zを有していてもよく、例えば、ハロゲン原子(フッ素原子)などを有していてもよい。
上記式(b-11)で表される重合性基及び上記式(b-12a)~(b-12c)のいずれかで表される重合性部位において、重合性基を形成する炭素原子であってR11又はRb2が結合していない炭素原子は無置換炭素原子として表しているが、上述のように、置換基を有していてもよい。置換基としては、特に制限されないが、例えば、R1としてとりうる上記基が挙げられる。
naは特に限定されないが、好ましくは1~6の整数であり、より好ましくは1又は2であり、更に好ましくは1である。
Raとしてとりうる置換基としては、特に限定されないが、上記重合鎖が好ましく、(メタ)アクリル樹脂の鎖又はポリシロキサン鎖がより好ましい。
Raは、式(b-13a)中の酸素原子(-O-)に直接結合していてもよく、連結基を介して結合していてもよい。この連結基としては、特に限定されないが、上述の、重合性基と重合鎖とを連結する連結基が挙げられる。
構成成分(MM)の、(メタ)アクリルポリマー中の含有量は、特に限定されないが、0~60モル%であることが好ましく、0.2~50モル%であることがより好ましく、0.5~40モル%であることが更に好ましい。上記範囲の含有量であると、無機固体電解質含有組成物の分散性と固体粒子間等の結着性とイオン伝導性とを高い水準で発揮できる。
ビニル化合物(M2)に由来する構成成分の、(メタ)アクリルポリマー中の含有量は、特に限定されないが、0~30モル%であることが好ましく、0~20モル%であることがより好ましく、0~10モル%であることが特に好ましい。
(メタ)アクリルポリマーが後述する官能基を有する場合、上記構成成分のいずれが官能基を有していてもよく、官能基を有する構成成分の含有量は、後述する通りであるが、上記含有量も満たす。
バインダーを形成するポリマーは、特にバインダーAを形成するポリマーとして適用される場合、無機固体電解質等の固体粒子の表面への濡れ性又は吸着性を高めるための官能基を有することが好ましい。このような官能基としては、固体粒子の表面において水素結合等の物理的相互作用を示す基及び固体粒子の表面に存在する基と化学結合を形成し得る基が挙げられ、具体的には、下記官能基群(I)から選択される基を少なくとも1つ有することがより好ましい。ただし、固体粒子の表面への濡れ性又は吸着性をより効果的に発現する観点からは、官能基同士で結合を形成することが可能な2種以上の基を有さないことが好ましい。
<官能基群(I)>
カルボキシ基、スルホン酸基(-SO3H)、リン酸基(-PO4H2)、アミノ基(-NH2)、ヒドロキシ基、スルファニル基、イソシアナト基、アルコキシシリル基及び3環以上の縮環構造を有する基
アルコキシシリル基は、少なくとも一つのアルコキシ基(炭素数は1~12が好ましい。)でSi原子が置換されたシリル基であればよく、Si原子上のその他の置換基としては、アルキル基、アリール基等が挙げられる。アルコキシシリル基としては、例えば、後述の置換基Zにおけるアルコキシシリル基の記載が好ましく適用できる。
3環以上の縮環構造を有する基は、コレステロール環構造を有する基、又は3環以上の芳香族環が縮環した構造を有する基が好ましく、コレステロール残基又はピレニル基がより好ましい。
バインダーAを形成するポリマー中における官能基群(I)から選択される官能基の含有量は、特に制限されないが、例えば、上記官能基群(I)から選択される官能基を有する構成成分の、バインダーを形成するポリマーを構成する全構成成分中の割合は、0.01~50モル%が好ましく、0.02~49モル%が好ましく、0.1~40モル%がより好ましく、1~30モル%が更に好ましく、3~25モル%が特に好ましい。
一方、バインダーを形成するポリマーは、バインダーBを形成するポリマーとして適用される場合、上述の範囲の吸着率を満たす限り、上記官能基を有していてもよく、有さないことが好ましい。バインダーBを形成するポリマー中における官能基群(I)から選択される官能基の含有量は、特に制限されないが、上記官能基群(I)から選択される官能基を有する構成成分の、バインダーを形成するポリマーを構成する全構成成分中の割合は、20モル%以下が好ましく、5モル%以下が好ましく、1モル%以下がより好ましく、0.7モル%以下が更に好ましい。
- 置換基Z -
アルキル基(好ましくは炭素数1~20のアルキル基、例えばメチル、エチル、イソプロピル、t-ブチル、ペンチル、ヘプチル、1-エチルペンチル、ベンジル、2-エトキシエチル、1-カルボキシメチル等)、アルケニル基(好ましくは炭素数2~20のアルケニル基、例えば、ビニル、アリル、オレイル等)、アルキニル基(好ましくは炭素数2~20のアルキニル基、例えば、エチニル、ブタジイニル、フェニルエチニル等)、シクロアルキル基(好ましくは炭素数3~20のシクロアルキル基、例えば、シクロプロピル、シクロペンチル、シクロヘキシル、4-メチルシクロヘキシル等、本明細書においてアルキル基というときには通常シクロアルキル基を含む意味であるが、ここでは別記する。)、アリール基(好ましくは炭素数6~26のアリール基、例えば、フェニル、1-ナフチル、4-メトキシフェニル、2-クロロフェニル、3-メチルフェニル等)、アラルキル基(好ましくは炭素数7~23のアラルキル基、例えば、ベンジル、フェネチル等)、ヘテロ環基(好ましくは炭素数2~20のヘテロ環基で、より好ましくは、少なくとも1つの酸素原子、硫黄原子、窒素原子を有する5又は6員環のヘテロ環基である。ヘテロ環基には芳香族ヘテロ環基及び脂肪族ヘテロ環基を含む。例えば、テトラヒドロピラン環基、テトラヒドロフラン環基、2-ピリジル、4-ピリジル、2-イミダゾリル、2-ベンゾイミダゾリル、2-チアゾリル、2-オキサゾリル、ピロリドン基等)、アルコキシ基(好ましくは炭素数1~20のアルコキシ基、例えば、メトキシ、エトキシ、イソプロピルオキシ、ベンジルオキシ等)、アリールオキシ基(好ましくは炭素数6~26のアリールオキシ基、例えば、フェノキシ、1-ナフチルオキシ、3-メチルフェノキシ、4-メトキシフェノキシ等、本明細書においてアリールオキシ基というときにはアリーロイルオキシ基を含む意味である。)、ヘテロ環オキシ基(上記ヘテロ環基に-O-基が結合した基)、アルコキシカルボニル基(好ましくは炭素数2~20のアルコキシカルボニル基、例えば、エトキシカルボニル、2-エチルヘキシルオキシカルボニル、ドデシルオキシカルボニル等)、アリールオキシカルボニル基(好ましくは炭素数6~26のアリールオキシカルボニル基、例えば、フェノキシカルボニル、1-ナフチルオキシカルボニル、3-メチルフェノキシカルボニル、4-メトキシフェノキシカルボニル等)、アミノ基(好ましくは炭素数0~20のアミノ基、アルキルアミノ基、アリールアミノ基を含み、例えば、アミノ(-NH2)、N,N-ジメチルアミノ、N,N-ジエチルアミノ、N-エチルアミノ、アニリノ等)、スルファモイル基(好ましくは炭素数0~20のスルファモイル基、例えば、N,N-ジメチルスルファモイル、N-フェニルスルファモイル等)、アシル基(アルキルカルボニル基、アルケニルカルボニル基、アルキニルカルボニル基、アリールカルボニル基、ヘテロ環カルボニル基を含み、好ましくは炭素数1~20のアシル基、例えば、アセチル、プロピオニル、ブチリル、オクタノイル、ヘキサデカノイル、アクリロイル、メタクリロイル、クロトノイル、ベンゾイル、ナフトイル、ニコチノイル等)、アシルオキシ基(アルキルカルボニルオキシ基、アルケニルカルボニルオキシ基、アルキニルカルボニルオキシ基、アリールカルボニルオキシ基、ヘテロ環カルボニルオキシ基を含み、好ましくは炭素数1~20のアシルオキシ基、例えば、アセチルオキシ、プロピオニルオキシ、ブチリルオキシ、オクタノイルオキシ、ヘキサデカノイルオキシ、アクリロイルオキシ、メタクリロイルオキシ、クロトノイルオキシ、ベンゾイルオキシ、ナフトイルオキシ、ニコチノイルオキシ等)、アリーロイルオキシ基(好ましくは炭素数7~23のアリーロイルオキシ基、例えば、ベンゾイルオキシ等)、カルバモイル基(好ましくは炭素数1~20のカルバモイル基、例えば、N,N-ジメチルカルバモイル、N-フェニルカルバモイル等)、アシルアミノ基(好ましくは炭素数1~20のアシルアミノ基、例えば、アセチルアミノ、ベンゾイルアミノ等)、アルキルチオ基(好ましくは炭素数1~20のアルキルチオ基、例えば、メチルチオ、エチルチオ、イソプロピルチオ、ベンジルチオ等)、アリールチオ基(好ましくは炭素数6~26のアリールチオ基、例えば、フェニルチオ、1-ナフチルチオ、3-メチルフェニルチオ、4-メトキシフェニルチオ等)、ヘテロ環チオ基(上記ヘテロ環基に-S-基が結合した基)、アルキルスルホニル基(好ましくは炭素数1~20のアルキルスルホニル基、例えば、メチルスルホニル、エチルスルホニル等)、アリールスルホニル基(好ましくは炭素数6~22のアリールスルホニル基、例えば、ベンゼンスルホニル等)、アルキルシリル基(好ましくは炭素数1~20のアルキルシリル基、例えば、モノメチルシリル、ジメチルシリル、トリメチルシリル、トリエチルシリル等)、アリールシリル基(好ましくは炭素数6~42のアリールシリル基、例えば、トリフェニルシリル等)、アルコキシシリル基(好ましくは炭素数1~20のアルコキシシリル基、例えば、モノメトキシシリル、ジメトキシシリル、トリメトキシシリル、トリエトキシシリル等)、アリールオキシシリル基(好ましくは炭素数6~42のアリールオキシシリル基、例えば、トリフェニルオキシシリル等)、ホスホリル基(好ましくは炭素数0~20のリン酸基、例えば、-OP(=O)(RP)2)、ホスホニル基(好ましくは炭素数0~20のホスホニル基、例えば、-P(=O)(RP)2)、ホスフィニル基(好ましくは炭素数0~20のホスフィニル基、例えば、-P(RP)2)、スルホ基(スルホン酸基)、カルボキシ基、ヒドロキシ基、スルファニル基、シアノ基、ハロゲン原子(例えばフッ素原子、塩素原子、臭素原子、ヨウ素原子等)が挙げられる。RPは、水素原子又は置換基(好ましくは置換基Zから選択される基)である。
また、これらの置換基Zで挙げた各基は、上記置換基Zが更に置換していてもよい。
上記アルキル基、アルキレン基、アルケニル基、アルケニレン基、アルキニル基及び/又はアルキニレン基等は、環状でも鎖状でもよく、また直鎖でも分岐していてもよい。
バインダーを形成するポリマーは、非架橋ポリマーであっても架橋ポリマーであってもよい。また、加熱又は電圧の印加によってポリマーの架橋が進行した場合には、上記分子量より大きな分子量となっていてもよい。好ましくは、全固体二次電池の使用開始時にポリマーが上記範囲の質量平均分子量であることである。
本発明において、有機溶媒に対して不溶であるとは、実施例に記載の方法により算出される、非極性溶媒に対する溶解度が1質量%以下であることを意味し、有機溶媒に対して可溶であるとは、実施例に記載の方法により算出される、非極性溶媒に対する溶解度が1質量%を越えていること、好ましくは2質量%以上であることを意味する。
バインダーは、無機固体電解質含有組成物中において、例えば有機溶媒に溶解して存在していてもよく(溶解して存在するバインダーを溶解型バインダーという。)、有機溶媒に溶解せず固体状で存在(好ましくは分散)していてもよい(固体状で存在するバインダーを粒子状バインダーという。)。
本発明において、バインダーAは、無機固体電解質含有組成物中、更には固体電解質層又は活物質層(塗布乾燥層)において粒子状バインダーであることが、分散性及び電池特性の点、更には電池抵抗の点で、好ましい。一方、バインダーBは、無機固体電解質含有組成物中において溶解型バインダーであることが、分散性の点で、好ましい。
なお、全固体二次電池の構成層における粒子状バインダーの粒子径は、例えば、電池を分解して粒子状バインダーを含有する構成層を剥がした後、その構成層について測定を行い、予め測定していた粒子状バインダー以外の粒子の粒子径の測定値を排除することにより、測定することができる。
粒子状バインダーの粒子径は、例えば、有機溶媒の種類、ポリマー中の構成成分の含有量及び含有量等により、調整できる。
バインダーを形成するポリマーは、非晶質であることが好ましい。本発明において、ポリマーが「非晶質」であるとは、典型的には、ガラス転移温度で測定したときに結晶融解に起因する吸熱ピークが見られないことをいう。
バインダーBを形成するポリマーの質量平均分子量は、特に制限されず上記範囲にあることが好ましいが、上記範囲のうち、分散性及び結着性の点で、20,000~5,000,000がより好ましく、50,000~4,000,000が更に好ましく、200,000~3,000,000が特に好ましい。
本発明において、ポリマー、ポリマー鎖及びマクロモノマーの分子量については、特に断らない限り、ゲルパーミエーションクロマトグラフィー(GPC)によって標準ポリスチレン換算の質量平均分子量及び数平均分子量をいう。その測定法としては、基本として下記条件1又は条件2(優先)の方法により測定した値とする。ただし、ポリマー又はマクロモノマーの種類によっては適宜適切な溶離液を選定して用いればよい。
(条件1)
カラム:TOSOH TSKgel Super AWM-H(商品名、東ソー社製)を2本つなげる
キャリア:10mMLiBr/N-メチルピロリドン
測定温度:40℃
キャリア流量:1.0ml/min
試料濃度:0.1質量%
検出器:RI(屈折率)検出器
(条件2)
カラム:TOSOH TSKgel Super HZM-H、TOSOH TSKgel Super HZ4000、TOSOH TSKgel Super HZ2000(いずれも商品名、東ソー社製)をつないだカラムを用いる。
キャリア:テトラヒドロフラン
測定温度:40℃
キャリア流量:1.0ml/min
試料濃度:0.1質量%
検出器:RI(屈折率)検出器
本発明の無機固体電解質含有組成物は、上記の各成分を分散又は溶解させる分散媒として、有機溶媒を含有する。
有機溶媒としては、使用環境において液状を示す有機化合物であればよく、例えば、各種溶媒が挙げられ、具体例としては、アルコール化合物、エーテル化合物、アミド化合物、アミン化合物、ケトン化合物、芳香族化合物、脂肪族化合物、ニトリル化合物、エステル化合物等が挙げられる。
有機溶媒としては、非極性有機溶媒(疎水性の有機溶媒)でも極性有機溶媒(親水性の有機溶媒)でもよいが、優れた分散性を発現できる点で、非極性有機溶媒が好ましい。非極性有機溶媒とは、一般に水に対する親和性が低い性質をいうが、本発明においては、ClogP値が1.5~6の有機溶媒であることが好ましく、例えば、エステル化合物、ケトン化合物、エーテル化合物、香族化合物、脂肪族化合物等が挙げられる。
ケトン化合物としては、例えば、アセトン、メチルエチルケトン、メチルイソブチルケトン(MIBK)、シクロペンタノン、シクロヘキサノン、シクロヘプタノン、ジプロピルケトン、ジブチルケトン、ジイソプロピルケトン、ジイソブチルケトン(DIBK)、イソブチルプロピルケトン、sec-ブチルプロピルケトン、ペンチルプロピルケトン、ブチルプロピルケトンなどが挙げられる。
芳香族化合物としては、例えば、ベンゼン、トルエン、キシレン等が挙げられる。
脂肪族化合物としては、例えば、ヘキサン、ヘプタン、オクタン、デカン、シクロヘキサン、メチルシクロヘキサン、エチルシクロヘキサン、シクロオクタン、デカリン、パラフィン、ガソリン、ナフサ、灯油、軽油等が挙げられる。
ニトリル化合物としては、例えば、アセトニトリル、プロピオニトリル、イソブチロニトリルなどが挙げられる。
エステル化合物としては、例えば、酢酸エチル、酢酸ブチル、酢酸プロピル、酪酸プロピル、酪酸イソプロピル、酪酸ブチル、酪酸イソブチル、ペンタン酸ブチル、イソ酪酸エチル、イソ酪酸プロピル、イソ酪酸イソプロピル、イソ酪酸イソブチル、ピバル酸プロピル、ピバル酸イソプロピル、ピバル酸ブチル、ピバル酸イソブチルなどが挙げられる。
例えば、炭化水素化合物を含むことが好ましい態様の1つである。炭化水素化合物は上述の芳香族化合物と脂肪族化合物のうち炭化水素で構成された化合物である。この態様において、有機溶媒中に占める炭化水素化合物の割合は、25質量%以上が好ましく、50質量%以上がより好ましく、60質量%以上がより一層好ましく、70質量%以上が更に好ましく、80質量%以上が特に好ましく、90質量%以上が最も好ましい。有機溶媒は炭化水素化合物からなることも好ましい態様の1つである。
本発明において、CLogP値とは、1-オクタノールと水への分配係数Pの常用対数LogPを計算によって求めた値である。CLogP値の計算に用いる方法やソフトウェアについては公知のものを用いることができるが、特に断らない限り、PerkinElmer社のChemDrawを用いて構造を描画し、算出した値とする。
有機溶媒を2種以上含有する場合、有機溶媒のClogP値は、各有機溶媒のClogP値と質量分率との積の和とする。
有機溶媒中に占める有機溶媒Bの割合は、10質量%以上が好ましく、20質量%以上がより好ましく、30質量%以上がより一層好ましく、50質量%以上が更に好ましい。上限値は、特に制限されず、100質量%とすることもでき、99.9質量%以下が好ましく、99質量%以下がより好ましい。
有機溶媒Aとしては、上記各化合物からClogP値を満たす溶媒を適宜に選択することができるが、非極性溶媒、具体的にはエステル化合物、ケトン化合物、エーテル化合物等が好ましい。有機溶媒Bとしては上述の通りである。
有機溶媒A及び有機溶媒Bを含む溶媒全体としてのClogP値は、特に制限されないが、有機溶媒を構成する化合物の上記ClogP値と同義であり、好ましい範囲も同じである。
有機溶媒中に占める有機溶媒Aの割合は、0.1質量%以上が好ましく、0.5質量%以上がより好ましく、1質量%以上が更に好ましく、5質量%以上が特に好ましい。上限値は、特に制限されず、95質量%以下が好ましく、80質量%以下がより好ましい。有機溶媒中に占める有機溶媒Bの割合は、上限が100質量%とならないこと以外は、上述の、有機溶媒中に占める有機溶媒Bの割合と同じである。また、有機溶媒Aの含有量と有機溶媒Bの含有量との質量比(有機溶媒Aの含有量:有機溶媒Bの含有量)は、特に制限されないが、例えば、1:99~99:1であることが好ましく、10:90~90:10であることがより好ましい。
本発明の無機固体電解質含有組成物には、周期律表第1族若しくは第2族に属する金属のイオンの挿入放出が可能な活物質を含有することもできる。活物質としては、以下に説明するが、正極活物質及び負極活物質が挙げられる。
本発明において、活物質(正極活物質又は負極活物質)を含有する無機固体電解質含有組成物を電極層用組成物(正極層用組成物又は負極層用組成物)ということがある。
正極活物質は、可逆的にリチウムイオンを挿入及び放出できるものが好ましい。その材料は、上記特性を有するものであれば、特に制限はなく、遷移金属酸化物、又は、硫黄などのLiと複合化できる元素などでもよい。
中でも、正極活物質としては、遷移金属酸化物を用いることが好ましく、遷移金属元素Ma(Co、Ni、Fe、Mn、Cu及びVから選択される1種以上の元素)を有する遷移金属酸化物がより好ましい。また、この遷移金属酸化物に元素Mb(リチウム以外の金属周期律表の第1(Ia)族の元素、第2(IIa)族の元素、Al、Ga、In、Ge、Sn、Pb、Sb、Bi、Si、P及びBなどの元素)を混合してもよい。混合量としては、遷移金属元素Maの量(100モル%)に対して0~30モル%が好ましい。Li/Maのモル比が0.3~2.2になるように混合して合成されたものが、より好ましい。
遷移金属酸化物の具体例としては、(MA)層状岩塩型構造を有する遷移金属酸化物、(MB)スピネル型構造を有する遷移金属酸化物、(MC)リチウム含有遷移金属リン酸化合物、(MD)リチウム含有遷移金属ハロゲン化リン酸化合物及び(ME)リチウム含有遷移金属ケイ酸化合物等が挙げられる。
(MB)スピネル型構造を有する遷移金属酸化物の具体例として、LiMn2O4(LMO)、LiCoMnO4、Li2FeMn3O8、Li2CuMn3O8、Li2CrMn3O8及びLi2NiMn3O8が挙げられる。
(MC)リチウム含有遷移金属リン酸化合物としては、例えば、LiFePO4及びLi3Fe2(PO4)3等のオリビン型リン酸鉄塩、LiFeP2O7等のピロリン酸鉄類、LiCoPO4等のリン酸コバルト類並びにLi3V2(PO4)3(リン酸バナジウムリチウム)等の単斜晶ナシコン型リン酸バナジウム塩が挙げられる。
(MD)リチウム含有遷移金属ハロゲン化リン酸化合物としては、例えば、Li2FePO4F等のフッ化リン酸鉄塩、Li2MnPO4F等のフッ化リン酸マンガン塩及びLi2CoPO4F等のフッ化リン酸コバルト類が挙げられる。
(ME)リチウム含有遷移金属ケイ酸化合物としては、例えば、Li2FeSiO4、Li2MnSiO4、Li2CoSiO4等が挙げられる。
本発明では、(MA)層状岩塩型構造を有する遷移金属酸化物が好ましく、LCO又はNMCがより好ましい。
焼成法によって得られた正極活物質は、水、酸性水溶液、アルカリ性水溶液、有機溶剤にて洗浄した後使用してもよい。
正極活物質層を形成する場合、正極活物質層の単位面積(cm2)当たりの正極活物質の質量(mg)(目付量)は特に制限されるものではない。設計された電池容量に応じて、適宜に決めることができ、例えば、1~100mg/cm2とすることができる。
負極活物質は、可逆的にリチウムイオンを挿入及び放出できるものが好ましい。その材料は、上記特性を有するものであれば、特に制限はなく、炭素質材料、金属酸化物、金属複合酸化物、リチウム単体、リチウム合金、リチウムと合金形成可能な負極活物質等が挙げられる。中でも、炭素質材料、金属複合酸化物又はリチウム単体が信頼性の点から好ましく用いられる。
これらの炭素質材料は、黒鉛化の程度により難黒鉛化炭素質材料(ハードカーボンともいう。)と黒鉛系炭素質材料に分けることもできる。また炭素質材料は、特開昭62-22066号公報、特開平2-6856号公報、同3-45473号公報に記載される面間隔又は密度、結晶子の大きさを有することが好ましい。炭素質材料は、単一の材料である必要はなく、特開平5-90844号公報記載の天然黒鉛と人造黒鉛の混合物、特開平6-4516号公報記載の被覆層を有する黒鉛等を用いることもできる。
炭素質材料としては、ハードカーボン又は黒鉛が好ましく用いられ、黒鉛がより好ましく用いられる。
Sn、Si、Geを中心とする非晶質酸化物負極活物質に併せて用いることができる負極活物質としては、リチウムイオン又はリチウム金属を吸蔵及び/又は放出できる炭素質材料、リチウム単体、リチウム合金、リチウムと合金化可能な負極活物質が好適に挙げられる。
負極活物質、例えば金属酸化物は、チタン元素を含有すること(チタン酸化物)も好ましく挙げられる。具体的には、Li4Ti5O12(チタン酸リチウム[LTO])がリチウムイオンの吸蔵放出時の体積変動が小さいことから急速充放電特性に優れ、電極の劣化が抑制されリチウムイオン二次電池の寿命向上が可能となる点で好ましい。
一般的に、これらの負極活物質を含有する負極(ケイ素元素含有活物質を含有するSi負極、スズ元素を有する活物質を含有するSn負極等)は、炭素負極(黒鉛及びアセチレンブラックなど)に比べて、より多くのLiイオンを吸蔵できる。すなわち、単位質量あたりのLiイオンの吸蔵量が増加する。そのため、電池容量を大きくすることができる。その結果、バッテリー駆動時間を長くすることができるという利点がある。
ケイ素元素含有活物質としては、例えば、Si、SiOx(0<x≦1)等のケイ素材料、更には、チタン、バナジウム、クロム、マンガン、ニッケル、銅、ランタン等を含むケイ素含有合金(例えば、LaSi2、VSi2、La-Si、Gd-Si、Ni-Si)、又は組織化した活物質(例えば、LaSi2/Si)、他にも、SnSiO3、SnSiS3等のケイ素元素及びスズ元素を含有する活物質等が挙げられる。なお、SiOxは、それ自体を負極活物質(半金属酸化物)として用いることができ、また、全固体二次電池の稼働によりSiを生成するため、リチウムと合金化可能な負極活物質(その前駆体物質)として用いることができる。
スズ元素を有する負極活物質としては、例えば、Sn、SnO、SnO2、SnS、SnS2、更には上記ケイ素元素及びスズ元素を含有する活物質等が挙げられる。また、酸化リチウムとの複合酸化物、例えば、Li2SnO2を挙げることもできる。
負極活物質層を形成する場合、負極活物質層の単位面積(cm2)当たりの負極活物質の質量(mg)(目付量)は特に制限されるものではない。設計された電池容量に応じて、適宜に決めることができ、例えば、1~100mg/cm2とすることができる。
正極活物質及び負極活物質の表面は別の金属酸化物で表面被覆されていてもよい。表面被覆剤としてはTi、Nb、Ta、W、Zr、Al、Si又はLiを含有する金属酸化物等が挙げられる。具体的には、チタン酸スピネル、タンタル系酸化物、ニオブ系酸化物、ニオブ酸リチウム系化合物等が挙げられ、具体的には、Li4Ti5O12、Li2Ti2O5、LiTaO3、LiNbO3、LiAlO2、Li2ZrO3、Li2WO4、Li2TiO3、Li2B4O7、Li3PO4、Li2MoO4、Li3BO3、LiBO2、Li2CO3、Li2SiO3、SiO2、TiO2、ZrO2、Al2O3、B2O3等が挙げられる。
また、正極活物質又は負極活物質を含む電極表面は硫黄又はリンで表面処理されていてもよい。
更に、正極活物質又は負極活物質の粒子表面は、上記表面被覆の前後において活性光線又は活性気体(プラズマ等)により表面処理を施されていてもよい。
本発明の無機固体電解質含有組成物は、導電助剤を適宜含有してもよく、特に負極活物質としてのケイ素原子含有活物質は導電助剤と併用されることが好ましい。
導電助剤としては、特に制限はなく、一般的な導電助剤として知られているものを用いることができる。例えば、電子伝導性材料である、天然黒鉛、人造黒鉛などの黒鉛類、アセチレンブラック、ケッチェンブラック、ファーネスブラックなどのカーボンブラック類、ニードルコークスなどの無定形炭素、気相成長炭素繊維若しくはカーボンナノチューブなどの炭素繊維類、グラフェン若しくはフラーレンなどの炭素質材料であってもよいし、銅、ニッケルなどの金属粉、金属繊維でもよく、ポリアニリン、ポリピロール、ポリチオフェン、ポリアセチレン、ポリフェニレン誘導体などの導電性高分子を用いてもよい。
本発明において、活物質と導電助剤とを併用する場合、上記の導電助剤のうち、電池を充放電した際に周期律表第一族若しくは第二族に属する金属のイオン(好ましくはLiイオン)の挿入と放出が起きず、活物質として機能しないものを導電助剤とする。したがって、導電助剤の中でも、電池を充放電した際に活物質層中において活物質として機能しうるものは、導電助剤ではなく活物質に分類する。電池を充放電した際に活物質として機能するか否かは、一義的ではなく、活物質との組み合わせにより決定される。
導電助剤の形状は、特に制限されないが、粒子状が好ましい。
本発明の無機固体電解質含有組成物が導電助剤を含む場合、無機固体電解質含有組成物中の導電助剤の含有量は、0~10質量%が好ましい。
本発明の無機固体電解質含有組成物は、リチウム塩(支持電解質)を含有することも好ましい。
リチウム塩としては、通常この種の製品に用いられるリチウム塩が好ましく、特に制限はなく、例えば、特開2015-088486の段落0082~0085記載のリチウム塩が好ましい。
本発明の無機固体電解質含有組成物がリチウム塩を含む場合、リチウム塩の含有量は、固体電解質100質量部に対して、0.1質量部以上が好ましく、5質量部以上がより好ましい。上限としては、50質量部以下が好ましく、20質量部以下がより好ましい。
本発明の無機固体電解質含有組成物は、上述のバインダーが分散剤としても機能するため、このバインダー以外の分散剤を含有していなくてもよいが、分散剤を含有してもよい。分散剤としては、全固体二次電池に通常使用されるものを適宜選定して用いることができる。一般的には粒子吸着と立体反発及び/又は静電反発を意図した化合物が好適に使用される。
本発明の無機固体電解質含有組成物は、上記各成分以外の他の成分として、適宜に、イオン液体、増粘剤、架橋剤(ラジカル重合、縮合重合又は開環重合により架橋反応するもの等)、重合開始剤(酸又はラジカルを熱又は光によって発生させるものなど)、消泡剤、レベリング剤、脱水剤、酸化防止剤等を含有することができる。イオン液体は、イオン伝導度をより向上させるため含有されるものであり、公知のものを特に制限されることなく用いることができる。また、上記ポリマー以外のポリマー、通常用いられる結着剤等を含有していてもよい。
本発明の無機固体電解質含有組成物は、無機固体電解質、上記バインダー、有機溶媒、更には適宜に、リチウム塩、任意の他の成分を、例えば通常用いる各種の混合機で混合することにより、混合物として、好ましくはスラリーとして、調製することができる。
混合方法は特に制限されず、一括して混合してもよく、順次混合してもよい。混合する環境は特に制限されないが、乾燥空気下又は不活性ガス下等が挙げられる。
本発明の活物質層形成用組成物は、固体粒子の(再)凝集を抑えて、長期に亘って高度に分散した固体粒子を含有した分散液とすることができる。
本発明の全固体二次電池用シートは、全固体二次電池の構成層を形成しうるシート状成形体であって、その用途に応じて種々の態様を含む。例えば、固体電解質層に好ましく用いられるシート(全固体二次電池用固体電解質シートともいう。)、電極、又は電極と固体電解質層との積層体に好ましく用いられるシート(全固体二次電池用電極シート)等が挙げられる。本発明において、これら各種のシートをまとめて全固体二次電池用シートということがある。
本発明の全固体二次電池用固体電解質シートとして、例えば、基材上に、本発明の無機固体電解質含有組成物で構成した層、通常固体電解質層と、保護層とをこの順で有するシートが挙げられる。全固体二次電池用固体電解質シートが有する固体電解質層は、本発明の無機固体電解質含有組成物で形成されることが好ましい。この固体電解質層中の各成分の含有量は、特に限定されないが、好ましくは、本発明の無機固体電解質含有組成物の固形分中における各成分の含有量と同義である。全固体二次電池用固体電解質シートを構成する各層の厚さは、後述する全固体二次電池において説明する各層の厚さと同じである。
本発明の全固体二次電池用シートを用いて全固体二次電池を製造すると、優れた電池性能を示す。
本発明の全固体二次電池用シートの製造方法は、特に制限されず、本発明の無機固体電解質含有組成物を用いて、上記の各層を形成することにより、製造できる。例えば、好ましくは基材若しくは集電体上(他の層を介していてもよい。)に、製膜(塗布乾燥)して無機固体電解質含有組成物からなる層(塗布乾燥層)を形成する方法が挙げられる。これにより、基材若しくは集電体と、塗布乾燥層とを有する全固体二次電池用シートを作製することができる。ここで、塗布乾燥層とは、本発明の無機固体電解質含有組成物を塗布し、有機溶媒を乾燥させることにより形成される層(すなわち、本発明の無機固体電解質含有組成物を用いてなり、本発明の無機固体電解質含有組成物から有機溶媒を除去した組成からなる層)をいう。活物質層及び塗布乾燥層は、本発明の効果を損なわない範囲であれば有機溶媒が残存していてもよく、残存量としては、例えば、各層中、3質量%以下とすることができる。
本発明の全固体二次電池用シートの製造方法において、塗布、乾燥等の各工程については、下記全固体二次電池の製造方法において説明する。
また、本発明の全固体二次電池用シートの製造方法においては、基材、保護層(特に剥離シート)等を剥離することもできる。
本発明の全固体二次電池は、正極活物質層と、この正極活物質層に対向する負極活物質層と、正極活物質層及び負極活物質層の間に配置された固体電解質層とを有する。正極活物質層は、好ましくは正極集電体上に形成され、正極を構成する。負極活物質層は、好ましくは負極集電体上に形成され、負極を構成する。
負極活物質層、正極活物質層及び固体電解質層の少なくとも1つの層が本発明の無機固体電解質含有組成物で形成されることが好ましく、全ての層が本発明の無機固体電解質含有組成物で形成される態様も好ましい。本発明の無機固体電解質含有組成物で形成された活物質層又は固体電解質層は、好ましくは、含有する成分種及びその含有量比について、本発明の無機固体電解質含有組成物の固形分におけるものと同じである。なお、活物質層又は固体電解質層が本発明の無機固体電解質含有組成物で形成されない場合、公知の材料を用いることができる。
負極活物質層、固体電解質層及び正極活物質層の厚さは、それぞれ、特に制限されない。各層の厚さは、一般的な全固体二次電池の寸法を考慮すると、それぞれ、10~1,000μmが好ましく、20μm以上500μm未満がより好ましい。本発明の全固体二次電池においては、正極活物質層及び負極活物質層の少なくとも1層の厚さが、50μm以上500μm未満であることが更に好ましい。
正極活物質層及び負極活物質層は、それぞれ、固体電解質層とは反対側に集電体を備えていてもよい。
本発明の全固体二次電池は、用途によっては、上記構造のまま全固体二次電池として使用してもよいが、乾電池の形態とするためには更に適当な筐体に封入して用いることが好ましい。筐体は、金属性のものであっても、樹脂(プラスチック)製のものであってもよい。金属性のものを用いる場合には、例えば、アルミニウム合金又は、ステンレス鋼製のものを挙げることができる。金属性の筐体は、正極側の筐体と負極側の筐体に分けて、それぞれ正極集電体及び負極集電体と電気的に接続させることが好ましい。正極側の筐体と負極側の筐体とは、短絡防止用のガスケットを介して接合され、一体化されることが好ましい。
全固体二次電池10においては、正極活物質層、固体電解質層及び負極活物質層のいずれも本発明の無機固体電解質含有組成物で形成されている。この全固体二次電池10は優れた電池性能を示す。正極活物質層4、固体電解質層3及び負極活物質層2が含有する無機固体電解質及びバインダーは、それぞれ、互いに同種であっても異種であってもよい。
本発明において、正極活物質層及び負極活物質層のいずれか、又は、両方を合わせて、単に、活物質層又は電極活物質層と称することがある。また、正極活物質及び負極活物質のいずれか、又は両方を合わせて、単に、活物質又は電極活物質と称することがある。
リチウム金属層としては、リチウム金属の粉末を堆積又は成形してなる層、リチウム箔及びリチウム蒸着膜等が挙げられる。リチウム金属層の厚さは、上記負極活物質層の上記厚さにかかわらず、例えば、1~500μmとすることができる。
本発明において、正極集電体及び負極集電体のいずれか、又は、両方を合わせて、単に、集電体と称することがある。
正極集電体を形成する材料としては、アルミニウム、アルミニウム合金、ステンレス鋼、ニッケル及びチタンなどの他に、アルミニウム又はステンレス鋼の表面にカーボン、ニッケル、チタンあるいは銀を処理させたもの(薄膜を形成したもの)が好ましく、その中でも、アルミニウム及びアルミニウム合金がより好ましい。
負極集電体を形成する材料としては、アルミニウム、銅、銅合金、ステンレス鋼、ニッケル及びチタンなどの他に、アルミニウム、銅、銅合金又はステンレス鋼の表面にカーボン、ニッケル、チタンあるいは銀を処理させたものが好ましく、アルミニウム、銅、銅合金及びステンレス鋼がより好ましい。
集電体の厚さは、特に制限されないが、1~500μmが好ましい。また、集電体表面は、表面処理により凹凸を付けることも好ましい。
全固体二次電池は、常法によって、製造できる。具体的には、全固体二次電池は、本発明の無機固体電解質含有組成物等を用いて、上記の各層を形成することにより、製造できる。しかも、本発明の無機固体電解質含有組成物は分散安定性に優れているから、無機固体電解質含有組成物の調製と各層の成膜とを時間的に連続して(組成物の調製直後に)実施しなくても、電池性能の劣化を抑えた全固体二次電池を製造できる。このように、本発明は、優れた電池性能を示し、好適には小さな電気抵抗を示す全固体二次電池を柔軟な製造条件で製造できる。以下、詳述する。
例えば、正極集電体である金属箔上に、正極用材料(正極層用組成物)として、正極活物質を含有する無機固体電解質含有組成物を塗布して正極活物質層を形成し、全固体二次電池用正極シートを作製する。次いで、この正極活物質層の上に、固体電解質層を形成するための無機固体電解質含有組成物を塗布して、固体電解質層を形成する。更に、固体電解質層の上に、負極用材料(負極層用組成物)として、負極活物質を含有する無機固体電解質含有組成物を塗布して、負極活物質層を形成する。負極活物質層の上に、負極集電体(金属箔)を重ねることにより、正極活物質層と負極活物質層の間に固体電解質層が挟まれた構造の全固体二次電池を得ることができる。これを筐体に封入して所望の全固体二次電池とすることもできる。
また、各層の形成方法を逆にして、負極集電体上に、負極活物質層、固体電解質層及び正極活物質層を形成し、正極集電体を重ねて、全固体二次電池を製造することもできる。
また別の方法として、次の方法が挙げられる。すなわち、上記のようにして、全固体二次電池用正極シート及び全固体二次電池用負極シートを作製する。また、これとは別に、無機固体電解質含有組成物を基材上に塗布して、固体電解質層からなる全固体二次電池用固体電解質シートを作製する。更に、全固体二次電池用正極シート及び全固体二次電池用負極シートで、基材から剥がした固体電解質層を挟むように積層する。このようにして、全固体二次電池を製造することができる。
上記の製造方法においては、正極層用組成物、無機固体電解質含有組成物及び負極層用組成物のいずれか1つに本発明の無機固体電解質含有組成物を用いればよく、いずれも、本発明の無機固体電解質含有組成物を用いることが好ましい。
無機固体電解質含有組成物の塗布方法は特に制限されず、適宜に選択できる。例えば、塗布(好ましくは湿式塗布)、スプレー塗布、スピンコート塗布、ディップコート塗布、スリット塗布、ストライプ塗布、バーコート塗布が挙げられる。
このとき、無機固体電解質含有組成物は、それぞれ塗布した後に乾燥処理を施してもよいし、重層塗布した後に乾燥処理をしてもよい。乾燥温度は特に制限されない。下限は30℃以上が好ましく、60℃以上がより好ましく、80℃以上が更に好ましい。上限は、300℃以下が好ましく、250℃以下がより好ましく、200℃以下が更に好ましい。このような温度範囲で加熱することで、有機溶媒を除去し、固体状態(塗布乾燥層)にすることができる。また、温度を高くしすぎず、全固体二次電池の各部材を損傷せずに済むため好ましい。これにより、全固体二次電池において、優れた総合性能を示し、かつ良好な結着性と、非加圧でも良好なイオン伝導度を得ることができる。
また、塗布した無機固体電解質含有組成物は、加圧と同時に加熱してもよい。加熱温度としては特に制限されず、一般的には30~300℃の範囲である。無機固体電解質のガラス転移温度よりも高い温度でプレスすることもできる。一方、無機固体電解質とバインダーが共存する場合、バインダーのガラス転移温度よりも高い温度でプレスすることもできる。ただし、一般的には上述のバインダーの融点を越えない温度である。
加圧は塗布溶媒又は有機溶媒を予め乾燥させた状態で行ってもよいし、溶媒又は有機溶媒が残存している状態で行ってもよい。
なお、各組成物は同時に塗布してもよいし、塗布乾燥プレスを同時及び/又は逐次行ってもよい。別々の基材に塗布した後に、転写により積層してもよい。
プレス時間は短時間(例えば数時間以内)で高い圧力をかけてもよいし、長時間(1日以上)かけて中程度の圧力をかけてもよい。全固体二次電池用シート以外、例えば全固体二次電池の場合には、中程度の圧力をかけ続けるために、全固体二次電池の拘束具(ネジ締め圧等)を用いることもできる。
プレス圧はシート面等の被圧部に対して均一であっても異なる圧であってもよい。
プレス圧は被圧部の面積又は厚さに応じて変化させることができる。また同一部位を段階的に異なる圧力で変えることもできる。
プレス面は平滑であっても粗面化されていてもよい。
上記のようにして製造した全固体二次電池は、製造後又は使用前に初期化を行うことが好ましい。初期化は特に制限されず、例えば、プレス圧を高めた状態で初充放電を行い、その後、全固体二次電池の一般使用圧力になるまで圧力を解放することにより、行うことができる。
本発明の全固体二次電池は種々の用途に適用することができる。適用態様には特に制限はないが、例えば、電子機器に搭載する場合、ノートパソコン、ペン入力パソコン、モバイルパソコン、電子ブックプレーヤー、携帯電話、コードレスフォン子機、ページャー、ハンディーターミナル、携帯ファックス、携帯コピー、携帯プリンター、ヘッドフォンステレオ、ビデオムービー、液晶テレビ、ハンディークリーナー、ポータブルCD、ミニディスク、電気シェーバー、トランシーバー、電子手帳、電卓、メモリーカード、携帯テープレコーダー、ラジオ、バックアップ電源などが挙げられる。その他民生用として、自動車、電動車両、モーター、照明器具、玩具、ゲーム機器、ロードコンディショナー、時計、ストロボ、カメラ、医療機器(ペースメーカー、補聴器、肩もみ機など)などが挙げられる。更に、各種軍需用、宇宙用として用いることができる。また、太陽電池と組み合わせることもできる。
アクリルポリマーA1、A2及びB、ポリウレタン1~6を以下に示す。ただし、各ポリマーにおける構成成分の含有率(モル%)は表1又は表2に示す。なお、ビニル系ポリマーは、アクリルポリマーA1のアクリル酸メチル構成成分を酢酸ビニル構成成分に変更したものであるので記載を省略する。
還流冷却管、ガス導入コックを付した2L三口フラスコに、下記マクロモノマーM-1の40質量%ヘプタン溶液を7.2g、アクリル酸メチル(MA)を12.4g、アクリル酸(AA)を6.7g、ヘプタン(和光純薬工業社製)を207g、アゾイソブチロニトリル1.4gを添加し、流速200mL/minにて窒素ガスを10分間導入した後に、100℃に昇温した。別容器にて調製した液(マクロモノマーM-1の40質量%ヘプタン溶液を846g、アクリル酸メチルを222.8g、アクリル酸を75.0g、ヘプタン300.0g、アゾイソブチロニトリル2.1gを混合した液)を4時間かけて滴下した。滴下完了後、アゾイソブチロニトリル0.5gを添加した。その後100℃で2時間攪拌した後、室温まで冷却し、ろ過することでアクリルポリマーA1の分散液を得た。固形成分濃度は39.2%であった。
12-ヒドロキシステアリン酸(和光純薬工業社製)の自己縮合体(GPCポリスチレンスタンダード数平均分子量:2,000)にグリシジルメタクリレート(東京化成工業社製)を反応させマクロモノマーとしてそれをメタクリル酸メチルとグリシジルメタクリレート(東京化成工業社製)と1:0.99:0.01(モル比)の割合で重合したポリマーにアクリル酸(富士フイルム和光純薬社製)を反応させたマクロモノマーM-1を得た。このマクロモノマーM-1のSP値は9.3、数平均分子量は11,000であった。マクロモノマーのSP値及び数平均分子量は上記方法により算出した値である。
合成例1において、MAの量を12.4gから12.6g、別容器におけるMAの量を222.8gから256.2gに変更し、AAの量を6.7gから6.8g、別容器におけるAAの量を75.0gから47.0gに変更したこと以外は、合成例1と同様にして、アクリルポリマーA2からなるバインダーの分散液を調整した。
合成例1において、MAの量を12.4gから12.7g、別容器におけるMAの量を222.8gから272.9gに変更し、AAの量を、6.7gから6.9g、別容器におけるAAの量を75.0gから33.0gに変更したこと以外は、合成例1と同様にして、アクリルポリマーBからなるバインダーの分散液を調整した。
合成例1において、アクリル酸メチルの代わりに酢酸ビニル(VA、富士フイルム和光純薬社製)を同モル数で用いたこと以外は、合成例1と同様にして、ビニル系ポリマーからなるバインダーの分散液を調整した。
200mL3つ口フラスコに、ポリエチレングリコール(商品名:ポリエチレングリコール200、富士フイルム和光純薬社製)4.46gと、2,2-ビス(ヒドロキシメチル)酪酸(東京化成社製)0.17g、NISSO-PB GI-1000(商品名、日本曹達社製)6.69gとを加え、THF(テトラヒドロフラン)74gに溶解した。この溶液に、ジフェニルメタンジイソシアネート(富士フイルム和光純薬社製)6.98gを加えて60℃で撹拌し、均一に溶解させた。得られた溶液に、ネオスタンU-600(商品名、日東化成社製)560mgを添加して60℃で5時間攪伴し、ポリウレタン1の20質量%THF溶液(ポリマー溶液)を得た。
次に、上記で得られたポリマー溶液に対してTHF74gを加えた溶液に、150rpmで撹拌しながら、ヘプタン222gを10分間かけて滴下し、ポリウレタン1の乳化液を得た。窒素ガスをフローしながらこの乳化液を85℃で120分加熱した。得られた残留物後にヘプタン50gを加えて更に85℃で60分加熱した。この操作を4回繰り返し、THFを除去した。こうして、ポリウレタン1からなるバインダーのヘプタン分散液を得た。
合成例5において、構成成分a~dを導く化合物及び構成成分a~dの含有率を、表2に記載の化合物及び含有率となるように変更したこと以外は、合成例5と同様にして、ポリウレタン2~6をそれぞれ合成した。次いで、合成例5と同様にして、各ポリウレタンからなるバインダーの分散液を調製した。なお、ポリウレタン4はヘプタンに溶解しており溶液として得た。
各ポリマーの質量平均分子量は上記方法(条件2)により測定した。また、各バインダーの粒子径は上記方法により測定した。なお、ポリウレタン4については表2の「粒子径」欄に「溶解」と表記した。
表中、構成成分A1~A3欄には、各構成単位を導く化合物名を下記の略号で示した。
- 構成成分A1 -
MA:アクリル酸メチルVA:酢酸ビニル
- 構成成分A2 -
AA:アクリル酸
- 構成成分A3 -
M-1:上記合成例1で合成したマクロモノマーM-1
表中、構成成分a~d欄には、各構成単位を導く化合物名を下記の略号で示した。
- 構成成分a -
MDI:ジフェニルメタンジイソシアネートH12MDI:ジシクロヘキシルメタン-4,4’-ジイソシアナート(東京化成工業社製)
- 構成成分b -
DMBA:2,2-ビス(ヒドロキシメチル)酪酸
- 構成成分c -
PEG200:ポリエチレングリコール200(商品名、数平均分子量200)
- 構成成分d -
GI-1000:水添ポリブタジエンポリオール NISSO-PB GI-1000(商品名、数平均分子量1500)
以下のポリマーは市販品を用いた。各ポリマーを溶媒:DIBKに溶解して濃度3.0質量%のバインダー溶液を調製した。なお、PVdFポリマーは上記溶媒に溶解せず、分散液として調製した。
PVdF:ポリビニリデンジフルオリド(商品名)(質量平均分子量180,000、Sigma Aldrich社製)
SBR:水添スチレンブタジエンゴム(DYNARON1321P(商品名)、質量平均分子量230,000、JSR社製)
SEBS:スチレン-エチレン-ブチレン-スチレンブロック共重合体(商品名、質量平均分子量100,000、Sigma Aldrich社製)
PVDF-HFP(6:4):ポリビニレンジフルオリドとヘキサフルオロプロピレンとの共重合体((商品名)、共重合比[PVdF:HFP](質量比)=6:4、質量平均分子量280,000、Sigma Aldrich社製)
PVDF-HFP(8:2):ポリビニレンジフルオリドとヘキサフルオロプロピレンとの共重合体((商品名)、共重合比[PVdF:HFP](質量比)=8:2、質量平均分子量300,000、Sigma Aldrich社製)
硫化物系無機固体電解質は、T.Ohtomo,A.Hayashi,M.Tatsumisago,Y.Tsuchida,S.Hama,K.Kawamoto,Journal of Power Sources,233,(2013),pp231-235、及び、A.Hayashi,S.Hama,H.Morimoto,M.Tatsumisago,T.Minami,Chem.Lett.,(2001),pp872-873の非特許文献を参考にして合成した。
具体的には、アルゴン雰囲気下(露点-70℃)のグローブボックス内で、硫化リチウム(Li2S、Aldrich社製、純度>99.98%)2.42g及び五硫化二リン(P2S5、Aldrich社製、純度>99%)3.90gをそれぞれ秤量し、メノウ製乳鉢に投入し、メノウ製乳棒を用いて、5分間混合した。Li2S及びP2S5の混合比は、モル比でLi2S:P2S5=75:25とした。
ジルコニア製45mL容器(フリッチュ社製)に、直径5mmのジルコニアビーズを66g投入し、上記の硫化リチウムと五硫化二リンの混合物全量を投入し、アルゴン雰囲気下で容器を完全に密閉した。フリッチュ社製遊星ボールミルP-7(商品名、フリッチュ社製)に容器をセットし、温度25℃で、回転数510rpmで20時間メカニカルミリングを行うことで、黄色粉体の硫化物系無機固体電解質(Li-P-S系ガラス、以下、LPSと表記することがある。)6.20gを得た。Li-P-S系ガラスの粒子径は2.5μmであった。
表3に示す各無機固体電解質含有組成物の調製に用いた無機固体電解質(LPS)0.5gとバインダー0.26gを15mLのバイアル瓶に入れ、ミックスローターで撹拌しながら、DIBKを25g添加し、更に室温、80rpmで30分撹拌した。撹拌後の分散液を孔径1μmのフィルターでろ過し、ろ液2gを乾燥し、乾固したバインダーの質量BX(無機固体電解質に吸着しなかったバインダーの質量)を計測した。
こうして得たバインダーの質量BX及び用いたバインダーの質量0.26gから、下記式により、バインダーの無機固体電解質に対する吸着率を算出した。
バインダーの吸着率ASEは、上記測定を2回行って得られた吸着率の平均値とする。
吸着率(%)=[(0.26-BX×25/2)/0.26]×100
なお、成膜した無機固体電解質層から取り出した無機固体電解質及びバインダーを用いて吸着率ASEを測定したところ同様の値が得られた。
各無機固体電解質含有組成物に併用する表3に示すバインダーA及びバインダーBについて、上述のようにして、測定した両バインダーの吸着率ASEから吸着率の差(バインダーAの吸着率ASE-バインダーBの吸着率ASE)を算出した。その結果を表3に示す。
表3に示す各無機固体電解質含有組成物(電極層用組成物)の調製に用いた活物質1.6gとバインダー0.08gを15mLのバイアル瓶に入れ、ミックスローターで撹拌しながら、DIBKを8g添加し、更に室温、80rpmで30分撹拌した。撹拌後の分散液を孔径1μmのフィルターでろ過し、ろ液2gを乾燥し、乾固したバインダーの質量(活物質に吸着しなかったバインダーの質量)質量BYを計測した。
こうして得たバインダーの質量BY及び用いたバインダーの質量0.08gから、下記式により、バインダーの活物質に対する吸着率を算出した。
バインダーの吸着率AAMは、上記測定を2回行って得られた吸着率の平均値とする。
吸着率(%)=[(0.08-BY×8/2)/0.08]×100
なお、成膜した活物質層から取り出した活物質及びバインダーを用いて吸着率AAMを測定したところ同様の値が得られた。
各バインダーをガラス瓶内に規定量秤量し、DIBK100gを添加し、25℃の温度下、ミックスローター上において80rpmの回転速度で24時間攪拌した。こうして得られた24時間攪拌後の混合液の透過率を以下条件により測定した。
この試験(透過率測定)をバインダー溶解量を変更して行い、透過率が99.8%となる上限濃度X(質量%)をバインダーの非極性溶媒に対する溶解度とする。
<透過率測定条件>
動的光散乱(DLS)測定
装置:大塚電子製DLS測定装置 DLS-8000
レーザ波長、出力:488nm/100mW
サンプルセル:NMR管
銅箔(商品名:C1100、宝泉社製)上に、各バインダーを有機溶媒(DIBK)に溶解させた溶液(固形分濃度:10質量%)を滴下した後に乾燥(温度:100℃、時間:180分)して、厚さ50μmの乾燥膜(幅:10mm、長さ:50mm)を作製した。
引っ張り試験機(ZTS-50N、イマダ社製)を用いて、得られた乾燥膜を銅箔の塗布面に対して角度90°の方向に30mm/sの速度で引き剥がした際の平均の引き剥がし力を剥離強度(単位:N/mm)とした。
実施例1では、調製若しくは準備したバインダーを用いて、無機固体電解質含有組成物、負極層用組成物及び正極層用組成物を調製して、その初期分散性及び分散安定性を評価した。
ジルコニア製45mL容器(フリッチュ社製)に、直径5mmのジルコニアビーズを180個投入し、合成例11で合成したLPS2.80g、有機溶媒としてキシレン12.3gを投入した。フリッチュ社製遊星ボールミルP-7に容器をセットし、温度25℃、回転数300rpmで2時間混合した。その後、活物質としてSi(商品名、Silicon Powder 1~5μm、Alfa Aesar社製)7.00g、導電助剤としてアセチレンブラック(商品名、AB粉状、デンカ社製)0.500g、バインダーAとして合成例1で調製したアクリルポリマーA1からなるバインダー分散液を固形分換算で0.105gと、バインダーBとして合成例3で調製したアクリルポリマーBからなるバインダー分散液を固形分換算で0.052gとを、容器に投入し、同様に、遊星ボールミルP-7に容器をセットして、温度25℃、回転数200rpmで15分間混合して、負極用組成物No.5を調製した。
上記負極用組成物No.5の調製において、バインダーA及びバインダーBとして表3に記載のポリマーからなるバインダー分散液又はバインダー溶液を用い、更に表3に示す活物質及び有機溶媒に適宜変更したこと以外は、負極用組成物No.5の調製と同様にして、負極用組成物No.1~4、6~27をそれぞれ調製した。
ただし、2種の有機溶媒を用いた負極用組成物No.22、23及び25において、有機溶媒の使用割合(質量比)を表3の「種類」欄の括弧内に「/」を用いて示す。
ジルコニア製45mL容器(フリッチュ社製)に、直径5mmのジルコニアビーズを160個投入し、合成例11で合成したLPS2.00g、有機溶媒としてDIBK(ジイソブチルケトン)12.3gを、投入した。フリッチュ社製遊星ボールミルP-7に容器をセットし、温度25℃、回転数300rpmで2時間混合した。その後、更に、活物質として、NMC(LiNi1/3Co1/3Mn1/3O2、正極用組成物No.28)又はNCA(LiNi0.85Co0.10Al0.05O2、正極用組成物No.30)7.10g、アセチレンブラック0.240g、バインダーAとして合成例7で調製したウレタン3からなるバインダー分散液を固形分換算で0.095gと、バインダーBとしてPVdF-HFP(フッ化ビニリデンとヘキサフルオロプロピレンとの共重合体、PVdF:HFP=8:2(質量比)(アルケマ社製))0.047gとを容器に投入し、同様に、遊星ボールミルP-7にセットし、温度25℃、回転数200rpmで15分間混合を続けた。こうして、正極層用組成物(スラリー)No.28及び30をそれぞれ調製した。
<正極用組成物No.29及び31の調製>
正極用組成物No.28及び30の調製において、バインダーBを用いず、LPSの使用量を2.047g(固形分全量に対して0.5%増)に変更したこと以外は、正極用組成物No.28及び30の調製と同様にして、正極用組成物No.29及び31をそれぞれ調製した。
ジルコニア製45mL容器(フリッチュ社製)に、直径5mmのジルコニアビーズを180個投入し、合成例11で合成したLPS2.00g、有機溶媒としてDIBK(ジイソブチルケトン)12.3gを投入した。その後、バインダーAとして合成例7で調製したウレタン3からなるバインダー分散液を固形分換算で0.020gと、バインダーBとしてPVdF-HFP(フッ化ビニリデンとヘキサフルオロプロピレンとの共重合体、PVdF:HFP=8:2(質量比)(アルケマ社製))0.010gとを容器に投入し、フリッチュ社製遊星ボールミルP-7にセットした。温度25℃、回転数200rpmで15分間混合して、無機固体電解質含有組成物No.32を調製した。
<無機固体電解質含有組成物No.33の調製>
無機固体電解質含有組成物No.32の調製において、バインダーBを用いず、LPSの使用量を2.01g(固形分全量に対して0.5%増)に変更したこと以外は、無機固体電解質含有組成物No.32の調製と同様にして、無機固体電解質含有組成物No.33を調製した。
調製した各組成物(スラリー)について、スラリー粘度ηα及びηβを下記方法により測定して、粘度差ηα―ηβを算出した。その結果(単位はmPa・sである。)を表3に示す。
具体的には、E型粘度計(TV-35、東機産業社製)、標準コーンロータ(1”34’×R24)、サンプル(スラリー)1.1mLをアプライして、回転数1~100rpmの範囲で粘度を測定した。温度22℃において、せん断速度10/s(回転数2.5rpm)で測定した粘度をスラリー粘度ηαとし、同様にせん断速度200/s(回転数50rpm)で測定した粘度をスラリー粘度ηβとした。
こうして求めたスラリー粘度ηαとスラリー粘度ηβとの粘度差ηα-ηβを算出した。
表中、「-」は該当する成分を有していないことを示す。
「含有量」及び「溶解度」の単位は「質量%」である。
「吸着率の差」は、無機固体電解質に対する、バインダーAの吸着率ASEとバインダーBの吸着率ASEの差を示す。
「形態」欄中の、「負極」は負極用組成物を、「正極」は正極用組成物を、「SE」は無機固体電解質含有組成物を、それぞれ表す。
Si:Silicon Powder(商品名、粒子径1~5μm、Alfa Aesar社製)
Sn:スズ粉末(粒子径3.5μm、Alfa Aesar社製)
NMC:LiNi1/3Co1/3Mn1/3O2(ニッケルマンガンコバルト酸リチウム)
NCA:LiNi0.85Co0.10Al0.05O2(ニッケルコバルトアルミニウム酸リチウム)
LPS:合成例11で合成したLi-P-S系ガラス
アクリルA1:合成例1で合成したアクリルポリマーA1
アクリルA2:合成例2で合成したアクリルポリマーA2
アクリルB:合成例3で合成したアクリルポリマーB
ビニル系:合成例4で合成したビニル系ポリマー
ウレタン1~6:合成例5~10で合成したポリウレタン1~6
SBR:スチレンブタジエンゴム
PVdF:ポリビニレンジフルオリド
SEBS:スチレン-エチレン-ブチレン-スチレンブロック共重合体
PVDF-HFP(6-4):ポリビニレンジフルオリドとヘキサフルオロプロピレンとの共重合体(共重合比[PVdF:HFP](質量比)=6:4)
PVDF-HFP(8-2):ポリビニレンジフルオリドとヘキサフルオロプロピレンとの共重合体(共重合比[PVdF:HFP](質量比)=8:2)
バインダーA及びバインダーBの「形態」欄には、バインダーの無機固体電解質含有組成物中での状態を示す。具体的には、バインダーが有機溶媒に固体状で分散している(粒子状バインダーである)場合を「粒子状」と表記し、有機溶媒に溶解している(溶解型バインダーである)場合を「溶解」と表記する。
また、バインダーA又はBとして2種のバインダーを含有する場合、「/」を用いて併記した。
DIBK:ジイソブチルケトン
MIBK:メチルイソブチルケトン
DBE:ジブチルエーテル
ECH:エチルシクロヘキサン
Hep:ヘプタン
なお、「有機溶媒」欄において、2種の有機溶媒を含有する場合、有機溶媒の略号「/」を用いて併記した。ClogP値は、2種の有機溶媒全体としての値とともに、各有機溶媒の値を「/」を用いて併記した。
調製した各組成物について、初期分散性を評価した。
各組成物を、内径5mmの沈降管に入れて、25℃で24時間静置した後に、組成物から分離した清澄(上澄み)と組成物(清澄分離後に分散状態を維持している状態)との界面の距離Xを測定した。具体的には、沈降管の底面から清澄層の表面(投入した組成物の表面)までの距離を100とした場合に、底面から上記界面までの距離Xを百分率で算出し、下記評価基準のいずれに含まれるかで評価した。結果を表4に示す。本試験において、初期分散性は評価ランク「E」以上が合格である。
- 評価ランク -
AA:98%以上、100%以下
A:95%以上、 98%未満
B:90%以上、 95%未満
C:85%以上、 90%未満
D:80%以上、 85%未満
E:75%以上、 80%未満
F:70%以上、 75%未満
G:70%未満
調製した各組成物について、分散安定性を評価した。
分散安定性は、各組成物を、内径5mmの沈降管に入れて、25℃で72時間静置した後に、組成物から分離した清澄(上澄み)と組成物(清澄分離後に分散状態を維持している状態)との界面の距離Xを測定し、初期分散性と同じ評価基準で、評価した。結果を表4に示す。本試験において、分散安定性は評価ランク「E」以上が合格である。
実施例2では、実施例1で調製した各組成物を用いて、全固体二次電池用固体電解質シート及び全固体二次電池用電極シートを作製し、全固体二次電池を製造した。
上記の各組成物(スラリー)を調製直後に用いて全固体二次電池用固体電解質シート及び全固体二次電池用電極シートを作製し、これを用いて、以下のようにして、固体電解質層及び活物質層として組成物調製直後塗工層を備えた全固体二次電池をそれぞれ製造した。
上記で得られた調製直後の負極層用組成物No.1~27を、それぞれ、厚さ20μmの銅箔上に、上記ベーカー式アプリケーター(商品名:SA-201ベーカー式アプリケーター、テスター産業社製)により塗布し、100℃1時間加熱して負極層用組成物を乾燥し、負極活物質層/銅箔の積層構造を有する全固体二次電池用負極シートNo.1~27をそれぞれ作製した。負極活物質層の厚さは100μmであった。
厚さ20μmのアルミニウム箔上にアプリケーター(商品名:SA-201ベーカー式アプリケーター、テスター産業社製)により調製直後の正極層用組成物No.28~31をそれぞれ塗布し、100℃で1時間加熱乾燥して、正極活物質層を形成し、全固体二次電池用正極シートNo.28~31をそれぞれ作製した。正極活物質層の厚さは100μmであった。
厚さ20μmのアルミニウム箔上にアプリケーター(商品名:SA-201ベーカー式アプリケーター、テスター産業社製)により調製直後の無機固体電解質含有組成物No.32及び33をそれぞれ塗布し、100℃で1時間加熱乾燥して、固体電解質層を形成し、全固体二次電池用固体電解質シートNo.32及び33をそれぞれ作製した。固体電解質層の厚さは30μmであった。
作製した各全固体二次電池用負極シートを直径10mmの円盤状に打ち抜き、内径10mmのポリエチレンテレフタラート(PET)製の円筒に入れた。円筒内の負極活物質層側に合成例11で合成したLPSを30mg入れ、円筒の両端開口から直径10mmのステンレス鋼(SUS)棒を挿入した。全固体二次電池用負極シートの集電体側と、LPSをSUS棒により、350MPaの圧力を加えて加圧した。LPS側のSUS棒を一旦外し、直径9mmの円盤状のインジウム(In)シート(厚さ20μm)と、直径9mmの円盤状のリチウム(Li)シート(厚さ20μm)を、この順で円筒内のLPSの上に挿入した。外していたSUS棒を円筒内に再度挿入し、50MPaの圧力をかけた状態で固定した。このようにして、銅箔(厚さ20μm)-負極活物質層(厚さ80μm)-固体電解質層(厚さ200μm)-対極層(In/Liシート、厚さ30μm)の構成を有する全固体二次電池(ハーフセル)を得た。
作製した各全固体二次電池用正極シートを直径10mmの円盤状に打ち抜き、内径10mmのPET製の円筒に入れた。円筒内の正極活物質層側に合成例11で合成したLPSを30mg入れ、円筒の両端開口から直径10mmのSUS棒を挿入した。全固体二次電池用正極シートの集電体側と、LPSをSUS棒により、350MPaの圧力を加えて加圧した。LPS側のSUS棒を一旦外し、直径9mmの円盤状のInシート(厚さ20μm)と、直径9mmの円盤状のLiシート(厚さ20μm)を、この順で円筒内のLPSの上に挿入した。外していたSUS棒を円筒内に再度挿入し、50MPaの圧力をかけた状態で固定した。このようにして、アルミニウム箔(厚さ20μm)-正極活物質層(厚さ80μm)-固体電解質層(厚さ200μm)-対極層(In/Liシート、厚さ30μm)の構成を有する全固体二次電池(ハーフセル)を得た。
全固体二次電池用正極シート(No.28)を直径10mmの円盤状に打ち抜き、内径10mmのPET製の円筒に入れた。円筒内の正極活物質層側に作製した各全固体二次電池用固体電解質シートを直径10mmの円盤状に打ち抜いて円筒内に入れ、円筒の両端開口から10mmのSUS棒を挿入した。全固体二次電池用正極シートの集電体側と、全固体二次電池用固体電解質シートのアルミニウム箔側とをSUS棒により、350MPaの圧力を加えて加圧した。全固体二次電池用固体電解質シート側のSUS棒を一旦外して全固体二次電池用固体電解質シートのアルミニウム箔を静かに剥離し、その後、全固体二次電池用負極シート(No.22)を直径10mmの円盤状に打ち抜き、円筒内の全固体二次電池用固体電解質シートの固体電解質層上に挿入した。外していたSUS棒を円筒内に再度挿入し、50MPaの圧力をかけた状態で固定した。このようにして、アルミニウム箔(厚さ20μm)-正極活物質層(厚さ80μm)-固体電解質層(厚さ25μm)-負極活物質層(厚さ80μm)-銅箔(厚さ20μm)の構成を有する全固体二次電池(フルセル)を得た。
実施例1で調製した各組成物(スラリー)を、温度25℃、露点-50℃の環境下に72時間静置した後に用いて作製した全固体二次電池用固体電解質シート及び全固体二次電池用電極シートを用いて、上記全固体二次電池の製造1と同様にして、固体電解質層及び活物質層として組成物経時後塗工層を備えた全固体二次電池をそれぞれ製造した。
実施例1で調製した各組成物を調製直後に用いて作製した全固体二次電池用固体電解質シート及び全固体二次電池用電極シートについて、固体電解質層又は活物質層の結着性を評価した。
具体的には、各シートの固体電解質層又は活物質層の表面にテープ(幅:1cm、長さ:5cm、商品名:ポリイミドテープ、日東電工社製)を張り付け(圧力0.1MPaで5分加圧圧着)、引っ張り試験機(ZTS-50N、イマダ社製)を用いて、このテープを固体電解質層又は活物質層の表面に対して角度90°の方向に30mm/sの速度で引き剥がした際の平均の引き剥がし力を剥離強度(単位:N/mm)とした。
測定した剥離強度を下記評価基準にあてはめて、固体電解質層又は活物質層の結着性を評価した。本試験において、剥離強度が0.1N/mm以上(評価レベルA及びB)であると、固体電解質層又は活物質層中の固体粒子の結着性、更には活物質層と集電体との結着性に優れるということができる。本発明においては、剥離強度が0.15N/mm以上(評価レベルA)であるとより好ましい。
-評価基準-
A:0.15N/mm以上
B:0.1N/mm以上、0.15N/mm未満
C:0.05N/mm以上、0.1N/mm未満
D:0.05N/mm未満
上記で作製した全固体二次電池を用い、30℃の環境下、充電電流値0.13mA及び放電電流値0.13mAの条件で4.3V~3.0Vの充放電を1回行った(初期化した)。
その後、サイクル試験として、25℃の環境下、充放電電流値0.39mAの条件で4.3V~3.0Vの充放電を繰り返した。1回の充放電を1サイクルとする。
1サイクル目の放電容量と20サイクル目の放電容量とを測定し、下記式により放電容量維持率を求め、この放電容量維持率を下記評価基準にあてはめて、全固体二次電池のサイクル特性を評価した。
本試験においては、調製直後の組成物を用いて製造した全固体二次電池においても、72時間静置後の組成物を用いて製造した全固体二次電池においても、評価レベル「F」以上が合格である。
放電容量維持率(%)=(20サイクル目の放電容量/1サイクル目の放電容量)×100
AA:80%以上、100%以下
A:70%以上、 80%未満
B:60%以上、 70%未満
C:50%以上、 60%未満
D:40%以上、 50%未満
E:30%以上、 40%未満
F:20%以上、 30%未満
G:20%未満
すなわち、本発明で規定する2種のバインダーを含有しない無機固体電解質含有組成物は、分散安定性に劣り、初期分散性も十分ではない場合もある。そのため、全固体二次電池の電池性能(特に72時間放置後の電池特性)、更には結着性も満足できるものではない。
これに対して、本発明で規定する2種のバインダーを含有する無機固体電解質含有組成物は初期分散性だけでなく、分散安定性にも優れる。また、これらの無機固体電解質含有組成物を用いて作製した各シートは固体粒子同士の結着、更には活物質層と集電体との結着性が強固であり、これらのシートを構成層として備えた全固体二次電池は高い電池性能(放電容量維持率)を示すことが分かる。
特に、調製後72時間静置した組成物を用いて形成した固体電解質層又は活物質を備えていても、調製直後の組成物を用いて形成した固体電解質層又は活物質を備えた全固体二次電池に対して電池特性の低下を効果的に抑制できることから、組成物の分散安定性(劣化防止)に優れ、上記分散安定性試験の結果と合致することが分かる。
2 負極活物質層
3 固体電解質層
4 正極活物質層
5 正極集電体
6 作動部位
10 全固体二次電池
Claims (22)
- 周期律表第1族若しくは第2族に属する金属のイオンの伝導性を有する無機固体電解質と、バインダーと、有機溶媒とを含有する無機固体電解質含有組成物であって、
前記バインダーが、前記無機固体電解質に対する吸着率が異なる少なくとも2種のバインダーA及びバインダーBを含み、
前記バインダーAの吸着率が15%以上であり、前記バインダーBの吸着率が10%未満である、無機固体電解質含有組成物。 - 前記バインダーAの吸着率が50%以上であり、前記バインダーBの吸着率が5%未満である、請求項1に記載の無機固体電解質含有組成物。
- 前記バインダーA及び前記バインダーBの少なくとも一方が粒子状である、請求項1又は2に記載の無機固体電解質含有組成物。
- 前記バインダーAが粒子状である、請求項1~3のいずれか1項に記載の無機固体電解質含有組成物。
- 前記バインダーAの、非極性溶媒に対する溶解度が1質量%以下である、請求項1~4のいずれか1項に記載の無機固体電解質含有組成物。
- 前記バインダーBの、非極性溶媒に対する溶解度が2質量%以上である、請求項1~5のいずれか1項に記載の無機固体電解質含有組成物。
- 前記無機固体電解質含有組成物中の、前記バインダーAの含有量が、前記無機固体電解質含有組成物中の、前記バインダーBの含有量よりも高い、請求項1~6のいずれか1項に記載の無機固体電解質含有組成物。
- 前記バインダーBを形成するポリマーが、フッ素系ポリマー、炭化水素系ポリマー、ポリウレタン又は(メタ)アクリルポリマーである、請求項1~7のいずれか1項に記載の無機固体電解質含有組成物。
- 前記バインダーAを形成するポリマーが、ポリウレタン又は(メタ)アクリルポリマーである、請求項1~8のいずれか1項に記載の無機固体電解質含有組成物。
- 前記有機溶媒が、少なくとも2種の有機溶媒を含む、請求項1~9のいずれか1項に記載の無機固体電解質含有組成物。
- 前記有機溶媒が、ClogP値が1.5以上3未満である有機溶媒を少なくとも1種含む、請求項1~10のいずれか1項に記載の無機固体電解質含有組成物。
- 前記有機溶媒が、ClogP値が3以上6未満である有機溶媒と、ClogP値が1.5以上3未満である有機溶媒とを含む、請求項1~11のいずれか1項に記載の無機固体電解質含有組成物。
- 前記有機溶媒の少なくとも1種が、エステル化合物、ケトン化合物又はエーテル化合物から選択される少なくとも1種を含む、請求項1~12のいずれか1項に記載の無機固体電解質含有組成物。
- せん断速度10/sのスラリー粘度ηαと、せん断速度200/sのスラリー粘度ηβとの粘度差ηα-ηβが400mPa・s以上である、請求項1~13のいずれか1項に記載の無機固体電解質含有組成物。
- 活物質を含有する、請求項1~14のいずれか1項に記載の無機固体電解質含有組成物。
- 前記バインダーAの、前記活物質への吸着率が10%以上である、請求項15に記載の無機固体電解質含有組成物。
- 前記バインダーBの、集電体に対する剥離強度が0.1N/mm以上である、請求項15又は16に記載の無機固体電解質含有組成物。
- 請求項1~17のいずれか1項に記載の無機固体電解質含有組成物で構成した層を有する全固体二次電池用シート。
- 請求項15~17のいずれか1項に記載の無機固体電解質含有組成物で構成した活物質層を有する全固体二次電池用電極シート。
- 正極活物質層と固体電解質層と負極活物質層とをこの順で具備する全固体二次電池であって、
前記正極活物質層、前記固体電解質層及び前記負極活物質層の少なくとも1つの層が、請求項1~17のいずれか1項に記載の無機固体電解質含有組成物で構成した層である、全固体二次電池。 - 請求項1~17のいずれか1項に記載の無機固体電解質含有組成物を製膜する、全固体二次電池用シートの製造方法。
- 請求項21に記載の製造方法を経て全固体二次電池を製造する、全固体二次電池の製造方法。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP20843068.6A EP4002514A4 (en) | 2019-07-19 | 2020-06-22 | COMPOSITION CONTAINING INORGANIC SOLID ELECTROLYTE, FULL SOLID SECONDARY BATTERY SHEET, FULLY SOLID SECONDARY BATTERY ELECTRODE SHEET, FULLY SOLID SECONDARY BATTERY, METHOD FOR PRODUCTION OF FULLY SOLID SECONDARY BATTERY SHEET, AND METHOD FOR PRODUCTION OF SECONDARY FULL SOLID BATTERY |
| CN202080050424.2A CN114144918B (zh) | 2019-07-19 | 2020-06-22 | 含有无机固体电解质的组合物、全固态二次电池、片材、及电极片、以及全固态二次电池用片材及全固态二次电池的制造方法 |
| JP2021533872A JP7320062B2 (ja) | 2019-07-19 | 2020-06-22 | 無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 |
| KR1020227001360A KR20220018604A (ko) | 2019-07-19 | 2020-06-22 | 무기 고체 전해질 함유 조성물, 전고체 이차 전지용 시트, 전고체 이차 전지용 전극 시트 및 전고체 이차 전지, 및, 전고체 이차 전지용 시트 및 전고체 이차 전지의 제조 방법 |
| US17/572,543 US20220140395A1 (en) | 2019-07-19 | 2022-01-10 | Inorganic solid electrolyte-containing composition, sheet for all-solid state secondary battery, electrode sheet for all-solid state secondary battery, and all-solid state secondary battery, and manufacturing methods for sheet for all-solid state secondary battery and all-solid state secondary battery |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2019-133417 | 2019-07-19 | ||
| JP2019133417 | 2019-07-19 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US17/572,543 Continuation US20220140395A1 (en) | 2019-07-19 | 2022-01-10 | Inorganic solid electrolyte-containing composition, sheet for all-solid state secondary battery, electrode sheet for all-solid state secondary battery, and all-solid state secondary battery, and manufacturing methods for sheet for all-solid state secondary battery and all-solid state secondary battery |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021014852A1 true WO2021014852A1 (ja) | 2021-01-28 |
Family
ID=74193158
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2020/024291 WO2021014852A1 (ja) | 2019-07-19 | 2020-06-22 | 無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20220140395A1 (ja) |
| EP (1) | EP4002514A4 (ja) |
| JP (1) | JP7320062B2 (ja) |
| KR (1) | KR20220018604A (ja) |
| CN (1) | CN114144918B (ja) |
| WO (1) | WO2021014852A1 (ja) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023054425A1 (ja) * | 2021-09-29 | 2023-04-06 | 富士フイルム株式会社 | 電極組成物、全固体二次電池用電極シート及び全固体二次電池、並びに、電極組成物、全固体二次電池用電極シート及び全固体二次電池の製造方法 |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20220042169A (ko) * | 2019-08-30 | 2022-04-04 | 후지필름 가부시키가이샤 | 무기 고체 전해질 함유 조성물, 전고체 이차 전지용 시트 및 전고체 이차 전지 및, 전고체 이차 전지용 시트 및 전고체 이차 전지의 제조 방법 |
| JP7263525B2 (ja) * | 2019-08-30 | 2023-04-24 | 富士フイルム株式会社 | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池並びに、全固体二次電池用シート及び全固体二次電池の製造方法 |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6222066A (ja) | 1985-07-23 | 1987-01-30 | Wako Pure Chem Ind Ltd | ラテツクス凝集反応測定装置 |
| JPH026856A (ja) | 1988-06-27 | 1990-01-11 | Motonobu Shibata | 触媒担体およびその製造方法 |
| JPH0345473A (ja) | 1989-07-11 | 1991-02-27 | Toyoda Mach Works Ltd | 四輪操舵装置 |
| JPH0590844A (ja) | 1991-09-26 | 1993-04-09 | Toshiba Corp | 歪補償器 |
| JPH064516A (ja) | 1992-06-17 | 1994-01-14 | Toshiba Corp | 割当て決定支援方式 |
| WO2012063827A1 (ja) * | 2010-11-09 | 2012-05-18 | 株式会社村田製作所 | 全固体電池用スラリー、全固体電池用グリーンシート、全固体電池、および全固体電池用スラリーの製造方法 |
| JP5354205B2 (ja) | 2009-12-22 | 2013-11-27 | 日本ゼオン株式会社 | 電気化学素子用電極および電気化学素子 |
| WO2015046313A1 (ja) | 2013-09-25 | 2015-04-02 | 富士フイルム株式会社 | 固体電解質組成物および全固体二次電池用のバインダー、ならびにこれらを用いた電池用電極シートおよび全固体二次電池 |
| JP2015088486A (ja) | 2013-09-25 | 2015-05-07 | 富士フイルム株式会社 | 固体電解質組成物、これを用いた電池用電極シートおよび全固体二次電池 |
| WO2016136089A1 (ja) * | 2015-02-27 | 2016-09-01 | 富士フイルム株式会社 | 固体電解質組成物、電池用電極シート及びその製造方法、並びに全固体二次電池及びその製造方法 |
| JP6262503B2 (ja) | 2013-11-26 | 2018-01-17 | 三星電子株式会社Samsung Electronics Co.,Ltd. | 全固体二次電池および全固体二次電池の製造方法 |
| WO2018020827A1 (ja) | 2016-07-26 | 2018-02-01 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シートおよび全固体二次電池、固体電解質含有シートおよび全固体二次電池の製造方法、ならびに、セグメント化ポリマー、ポリマーおよびセグメント化ポリマーの非水溶媒分散物 |
| WO2018151118A1 (ja) | 2017-02-16 | 2018-08-23 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シート及びその製造方法、全固体二次電池及びその製造方法、並びに、ポリマー及びその非水溶媒分散物 |
| WO2019097903A1 (ja) * | 2017-11-17 | 2019-05-23 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シート及び全固体二次電池、並びに、固体電解質含有シート及び全固体二次電池の製造方法 |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5354205Y2 (ja) | 1973-03-15 | 1978-12-25 | ||
| JP4612664B2 (ja) * | 2007-09-25 | 2011-01-12 | セイコーエプソン株式会社 | 全固体二次電池、全固体二次電池の製造方法 |
| CN111406340B (zh) | 2017-11-17 | 2024-01-16 | 富士胶片株式会社 | 固体电解质组合物、片材、电极片、电池及制造方法 |
| EP3713002A4 (en) | 2017-11-17 | 2020-12-09 | FUJIFILM Corporation | SOLID ELECTROLYTE COMPOSITION, SHEET CONTAINING SOLID ELECTROLYTE, FULLY SOLID SECONDARY BATTERY ELECTRODE SHEET, FULLY SOLID SECONDARY BATTERY, PROCESS FOR PRODUCING SHEET CONTAINING SOLID ELECTRODE, AND PROCESS FOR SOLID PRODUCTION FOR SECONDARY BATTERY |
-
2020
- 2020-06-22 WO PCT/JP2020/024291 patent/WO2021014852A1/ja unknown
- 2020-06-22 EP EP20843068.6A patent/EP4002514A4/en active Pending
- 2020-06-22 KR KR1020227001360A patent/KR20220018604A/ko not_active Application Discontinuation
- 2020-06-22 CN CN202080050424.2A patent/CN114144918B/zh active Active
- 2020-06-22 JP JP2021533872A patent/JP7320062B2/ja active Active
-
2022
- 2022-01-10 US US17/572,543 patent/US20220140395A1/en active Pending
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6222066A (ja) | 1985-07-23 | 1987-01-30 | Wako Pure Chem Ind Ltd | ラテツクス凝集反応測定装置 |
| JPH026856A (ja) | 1988-06-27 | 1990-01-11 | Motonobu Shibata | 触媒担体およびその製造方法 |
| JPH0345473A (ja) | 1989-07-11 | 1991-02-27 | Toyoda Mach Works Ltd | 四輪操舵装置 |
| JPH0590844A (ja) | 1991-09-26 | 1993-04-09 | Toshiba Corp | 歪補償器 |
| JPH064516A (ja) | 1992-06-17 | 1994-01-14 | Toshiba Corp | 割当て決定支援方式 |
| JP5354205B2 (ja) | 2009-12-22 | 2013-11-27 | 日本ゼオン株式会社 | 電気化学素子用電極および電気化学素子 |
| WO2012063827A1 (ja) * | 2010-11-09 | 2012-05-18 | 株式会社村田製作所 | 全固体電池用スラリー、全固体電池用グリーンシート、全固体電池、および全固体電池用スラリーの製造方法 |
| WO2015046313A1 (ja) | 2013-09-25 | 2015-04-02 | 富士フイルム株式会社 | 固体電解質組成物および全固体二次電池用のバインダー、ならびにこれらを用いた電池用電極シートおよび全固体二次電池 |
| JP2015088486A (ja) | 2013-09-25 | 2015-05-07 | 富士フイルム株式会社 | 固体電解質組成物、これを用いた電池用電極シートおよび全固体二次電池 |
| JP2015088480A (ja) | 2013-09-25 | 2015-05-07 | 富士フイルム株式会社 | 固体電解質組成物および全固体二次電池用のバインダー、ならびにこれらを用いた電池用電極シートおよび全固体二次電池 |
| JP6262503B2 (ja) | 2013-11-26 | 2018-01-17 | 三星電子株式会社Samsung Electronics Co.,Ltd. | 全固体二次電池および全固体二次電池の製造方法 |
| WO2016136089A1 (ja) * | 2015-02-27 | 2016-09-01 | 富士フイルム株式会社 | 固体電解質組成物、電池用電極シート及びその製造方法、並びに全固体二次電池及びその製造方法 |
| WO2018020827A1 (ja) | 2016-07-26 | 2018-02-01 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シートおよび全固体二次電池、固体電解質含有シートおよび全固体二次電池の製造方法、ならびに、セグメント化ポリマー、ポリマーおよびセグメント化ポリマーの非水溶媒分散物 |
| WO2018151118A1 (ja) | 2017-02-16 | 2018-08-23 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シート及びその製造方法、全固体二次電池及びその製造方法、並びに、ポリマー及びその非水溶媒分散物 |
| WO2019097903A1 (ja) * | 2017-11-17 | 2019-05-23 | 富士フイルム株式会社 | 固体電解質組成物、固体電解質含有シート及び全固体二次電池、並びに、固体電解質含有シート及び全固体二次電池の製造方法 |
Non-Patent Citations (2)
| Title |
|---|
| H. L. HOY, JOURNAL OF PAINT TECHNOLOGY, vol. 42, no. 541, 1970, pages 76 - 118 |
| T. OHTOMOA. HAYASHIM. TATSUMISAGOY. TSUCHIDAS. HAMAK. KAWAMOTO, JOURNAL OF POWER SOURCES, vol. 233, 2013 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023054425A1 (ja) * | 2021-09-29 | 2023-04-06 | 富士フイルム株式会社 | 電極組成物、全固体二次電池用電極シート及び全固体二次電池、並びに、電極組成物、全固体二次電池用電極シート及び全固体二次電池の製造方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20220018604A (ko) | 2022-02-15 |
| US20220140395A1 (en) | 2022-05-05 |
| EP4002514A4 (en) | 2022-12-28 |
| JPWO2021014852A1 (ja) | 2021-01-28 |
| JP7320062B2 (ja) | 2023-08-02 |
| CN114144918A (zh) | 2022-03-04 |
| CN114144918B (zh) | 2024-08-27 |
| EP4002514A1 (en) | 2022-05-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2021066060A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021039950A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021100362A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021039948A1 (ja) | 電極用組成物の製造方法、全固体二次電池用電極シートの製造方法及び全固体二次電池の製造方法 | |
| JP7104800B2 (ja) | 全固体二次電池の製造方法、並びに、全固体二次電池用電極シート及びその製造方法 | |
| WO2021039468A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021014852A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021060541A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021157278A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| JP7554316B2 (ja) | 全固体二次電池及び全固体二次電池用シート | |
| WO2021193826A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021166968A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021085549A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021039947A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の各製造方法 | |
| WO2020129802A1 (ja) | 固体電解質組成物、固体電解質含有シート及び全固体二次電池、並びに、固体電解質含有シート及び全固体二次電池の製造方法 | |
| WO2023054425A1 (ja) | 電極組成物、全固体二次電池用電極シート及び全固体二次電池、並びに、電極組成物、全固体二次電池用電極シート及び全固体二次電池の製造方法 | |
| WO2021039946A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、全固体二次電池用シート及び全固体二次電池の製造方法、並びに、複合ポリマー粒子 | |
| WO2021020031A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| JPWO2020067108A1 (ja) | 全固体二次電池の負極用組成物、全固体二次電池用負極シート及び全固体二次電池、並びに、全固体二次電池用負極シート及び全固体二次電池の製造方法 | |
| WO2022071124A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| JP7407286B2 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| JP7373674B2 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2021060542A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2024071056A1 (ja) | 無機固体電解質含有組成物、全固体二次電池用シート及び全固体二次電池、並びに、全固体二次電池用シート及び全固体二次電池の製造方法 | |
| WO2022085733A1 (ja) | 電極組成物、全固体二次電池用電極シート及び全固体二次電池、並びに、全固体二次電池用電極シート及び全固体二次電池の製造方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 20843068 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2021533872 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20227001360 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2020843068 Country of ref document: EP Effective date: 20220221 |