WO2018139262A1 - フッ化ビニリデン系樹脂フィルム - Google Patents
フッ化ビニリデン系樹脂フィルム Download PDFInfo
- Publication number
- WO2018139262A1 WO2018139262A1 PCT/JP2018/000969 JP2018000969W WO2018139262A1 WO 2018139262 A1 WO2018139262 A1 WO 2018139262A1 JP 2018000969 W JP2018000969 W JP 2018000969W WO 2018139262 A1 WO2018139262 A1 WO 2018139262A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- vinylidene fluoride
- film
- fluoride resin
- resin film
- inorganic particles
- Prior art date
Links
- 229920005989 resin Polymers 0.000 title claims abstract description 122
- 239000011347 resin Substances 0.000 title claims abstract description 122
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 title claims abstract description 117
- 239000010954 inorganic particle Substances 0.000 claims abstract description 42
- 239000000203 mixture Substances 0.000 claims abstract description 23
- 239000002245 particle Substances 0.000 claims description 25
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 claims description 19
- 229910000019 calcium carbonate Inorganic materials 0.000 claims description 8
- 229920001577 copolymer Polymers 0.000 claims description 8
- 229920001519 homopolymer Polymers 0.000 claims description 5
- 230000003068 static effect Effects 0.000 claims description 5
- 239000000395 magnesium oxide Substances 0.000 claims description 4
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 claims description 4
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 claims description 4
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 claims description 4
- 150000002484 inorganic compounds Chemical class 0.000 claims description 3
- 229910010272 inorganic material Inorganic materials 0.000 claims description 3
- 230000000052 comparative effect Effects 0.000 description 30
- 238000004804 winding Methods 0.000 description 17
- 239000002033 PVDF binder Substances 0.000 description 15
- 239000003990 capacitor Substances 0.000 description 15
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 15
- 238000005259 measurement Methods 0.000 description 13
- 230000015556 catabolic process Effects 0.000 description 11
- 239000000126 substance Substances 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 10
- 239000008188 pellet Substances 0.000 description 10
- 239000000178 monomer Substances 0.000 description 9
- -1 polypropylene Polymers 0.000 description 9
- 229910052791 calcium Inorganic materials 0.000 description 7
- 238000000034 method Methods 0.000 description 7
- 239000000463 material Substances 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- 239000004743 Polypropylene Substances 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 229920001155 polypropylene Polymers 0.000 description 5
- 230000037303 wrinkles Effects 0.000 description 5
- 238000004458 analytical method Methods 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000006057 Non-nutritive feed additive Substances 0.000 description 3
- 238000004898 kneading Methods 0.000 description 3
- 239000000155 melt Substances 0.000 description 3
- 238000002844 melting Methods 0.000 description 3
- 230000008018 melting Effects 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 238000007740 vapor deposition Methods 0.000 description 3
- 239000004721 Polyphenylene oxide Substances 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 238000004891 communication Methods 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000001125 extrusion Methods 0.000 description 2
- 238000009413 insulation Methods 0.000 description 2
- 239000012212 insulator Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000004033 plastic Substances 0.000 description 2
- 229920003023 plastic Polymers 0.000 description 2
- 229920000570 polyether Polymers 0.000 description 2
- 238000010094 polymer processing Methods 0.000 description 2
- BBEAQIROQSPTKN-UHFFFAOYSA-N pyrene Chemical compound C1=CC=C2C=CC3=CC=CC4=CC=C1C2=C43 BBEAQIROQSPTKN-UHFFFAOYSA-N 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 239000011342 resin composition Substances 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- BLTXWCKMNMYXEA-UHFFFAOYSA-N 1,1,2-trifluoro-2-(trifluoromethoxy)ethene Chemical compound FC(F)=C(F)OC(F)(F)F BLTXWCKMNMYXEA-UHFFFAOYSA-N 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- 229920000297 Rayon Polymers 0.000 description 1
- 229920010524 Syndiotactic polystyrene Polymers 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-N acrylic acid group Chemical group C(C=C)(=O)O NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000012937 correction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthrene Natural products C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 1
- XUCNUKMRBVNAPB-UHFFFAOYSA-N fluoroethene Chemical compound FC=C XUCNUKMRBVNAPB-UHFFFAOYSA-N 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- HCDGVLDPFQMKDK-UHFFFAOYSA-N hexafluoropropylene Chemical group FC(F)=C(F)C(F)(F)F HCDGVLDPFQMKDK-UHFFFAOYSA-N 0.000 description 1
- 229920006324 polyoxymethylene Polymers 0.000 description 1
- 239000002964 rayon Substances 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- GCLGEJMYGQKIIW-UHFFFAOYSA-H sodium hexametaphosphate Chemical compound [Na]OP1(=O)OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])OP(=O)(O[Na])O1 GCLGEJMYGQKIIW-UHFFFAOYSA-H 0.000 description 1
- 235000019982 sodium hexametaphosphate Nutrition 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 1
- 239000001577 tetrasodium phosphonato phosphate Substances 0.000 description 1
- 238000009210 therapy by ultrasound Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G4/00—Fixed capacitors; Processes of their manufacture
- H01G4/002—Details
- H01G4/018—Dielectrics
- H01G4/06—Solid dielectrics
- H01G4/14—Organic dielectrics
- H01G4/18—Organic dielectrics of synthetic material, e.g. derivatives of cellulose
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/013—Fillers, pigments or reinforcing additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L27/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers
- C08L27/02—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L27/12—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
- C08L27/16—Homopolymers or copolymers or vinylidene fluoride
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L27/00—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers
- C08L27/02—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment
- C08L27/12—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Compositions of derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
- C08L27/20—Homopolymers or copolymers of hexafluoropropene
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B3/00—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties
- H01B3/18—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances
- H01B3/30—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes
- H01B3/44—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes vinyl resins; acrylic resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01G—CAPACITORS; CAPACITORS, RECTIFIERS, DETECTORS, SWITCHING DEVICES, LIGHT-SENSITIVE OR TEMPERATURE-SENSITIVE DEVICES OF THE ELECTROLYTIC TYPE
- H01G4/00—Fixed capacitors; Processes of their manufacture
- H01G4/002—Details
- H01G4/018—Dielectrics
- H01G4/20—Dielectrics using combinations of dielectrics from more than one of groups H01G4/02 - H01G4/06
- H01G4/206—Dielectrics using combinations of dielectrics from more than one of groups H01G4/02 - H01G4/06 inorganic and synthetic material
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2327/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers
- C08J2327/02—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment
- C08J2327/12—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
- C08J2327/16—Homopolymers or copolymers of vinylidene fluoride
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2327/00—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers
- C08J2327/02—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment
- C08J2327/12—Characterised by the use of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a halogen; Derivatives of such polymers not modified by chemical after-treatment containing fluorine atoms
- C08J2327/20—Homopolymers or copolymers of hexafluoropropene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
- C08K2003/2217—Oxides; Hydroxides of metals of magnesium
- C08K2003/222—Magnesia, i.e. magnesium oxide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
- C08K2003/2227—Oxides; Hydroxides of metals of aluminium
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/24—Acids; Salts thereof
- C08K3/26—Carbonates; Bicarbonates
- C08K2003/265—Calcium, strontium or barium carbonate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K2201/00—Specific properties of additives
- C08K2201/002—Physical properties
- C08K2201/005—Additives being defined by their particle size in general
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02T—CLIMATE CHANGE MITIGATION TECHNOLOGIES RELATED TO TRANSPORTATION
- Y02T10/00—Road transport of goods or passengers
- Y02T10/60—Other road transportation technologies with climate change mitigation effect
- Y02T10/70—Energy storage systems for electromobility, e.g. batteries
Definitions
- the present invention relates to a vinylidene fluoride resin film, and more particularly to a vinylidene fluoride resin film having a large relative dielectric constant.
- Plastic insulators are characterized by high insulation resistance, excellent frequency characteristics, and excellent flexibility, so they are suitable for communication, electronic equipment, power, medium / low-pressure phase advance, inverter, etc. It is expected as a film capacitor and film materials such as a piezoelectric element, a pyroelectric element, and a dielectric for carrying a transfer body.
- a general film capacitor uses a resin having a relatively low dielectric constant such as polypropylene, but it has been proposed to obtain a film capacitor having a high relative dielectric constant by using a vinylidene fluoride resin having a high dielectric constant. .
- Patent Document 1 discloses a vinylidene fluoride resin composition comprising 95 to 30% by weight of vinylidene fluoride resin and 5 to 70% by weight of polyether. Polyoxymethylene is mentioned as this polyether.
- Patent Documents 2 to 5 The technology relating to the film is described in Patent Documents 2 to 5, for example.
- the film described in Patent Document 2 is a high dielectric film formed using a fluororesin containing a total of 95 mol% or more of vinylidene fluoride units and tetrafluoroethylene units, and has high dielectric properties, alternating voltage, and thinning. It is a film for film capacitors that is possible.
- the film described in Patent Document 3 is a high dielectric constant film containing a vinylidene fluoride resin and inorganic particles.
- the film described in Patent Document 4 is a biaxially stretched polypropylene film in which a target surface shape is formed using crystal modification of a polypropylene film.
- Patent Document 5 describes a stretched film made of a syndiotactic polystyrene resin composition.
- the stretched film contains two types of particles having different particle diameters, and at least one of the particles is silica particles.
- the vinylidene fluoride resin film of Patent Document 2 has a risk of causing dielectric breakdown and has a problem in the winding property of the film.
- the biaxially stretched polypropylene film described in Patent Document 4 has a low dielectric constant. For this reason, there is a limit to downsizing capacitors and the like made using such a film.
- Patent Document 5 As a means for improving the winding property of a resin film, as described in Patent Document 5, a method of adding silica particles to the film is known.
- the inorganic particles when inorganic particles are blended with a resin having a small surface energy such as a fluororesin, the inorganic particles have an influence on the film, such as dispersibility, film forming property, and film winding property.
- the influence of inorganic particles in a general resin film cannot be directly applied to the above-described influence when inorganic particles are blended with a resin having a small surface energy.
- melt-kneading a vinylidene fluoride resin and a silicon oxide compound is not preferable because the vinylidene fluoride resin may be decomposed.
- the present invention has been made in view of the above problems, and an object of the present invention is to provide a vinylidene fluoride-based resin film that can be thinned and has improved film winding properties.
- the vinylidene fluoride resin film of the present invention is a vinylidene fluoride resin film produced using a film composition comprising a vinylidene fluoride resin and inorganic particles,
- the vinylidene fluoride resin film has a plurality of protrusions on at least one surface, and among these protrusions, the number of protrusions exceeding 0.10 ⁇ m from the smooth surface where no protrusions are present is the vinylidene fluoride type The number is 7.0 or more and 50 or less per 0.10 mm 2 of the resin film.
- the vinylidene fluoride resin film (hereinafter also simply referred to as “film”) of the present invention is a vinylidene fluoride resin film produced using a film composition containing at least a vinylidene fluoride resin and inorganic particles.
- the film has a plurality of protrusions on at least one surface, and among these protrusions, the number of protrusions exceeding 0.10 ⁇ m from the smooth surface where no protrusion (filler) exists is a vinylidene fluoride type The number is 7.0 or more and 50 or less per 0.10 mm 2 of the resin film.
- composition for film is a composition containing at least a vinylidene fluoride resin and inorganic particles.
- the vinylidene fluoride resin means a polymer mainly composed of a vinylidene fluoride monomer.
- the vinylidene fluoride resin is usually a homopolymer of vinylidene fluoride, but a copolymer may also be used.
- the vinylidene fluoride copolymer preferably contains 90% or more of vinylidene fluoride monomer, more preferably 95% or more, and even more preferably 97% or more. If the content of the monomer other than the vinylidene fluoride monomer is too large, problems such as a decrease in the melting point of the vinylidene fluoride resin and a decrease in the dielectric constant are expected.
- Such a vinylidene fluoride resin is preferable as a material for a high dielectric film because it has a high relative dielectric constant ( ⁇ ).
- the relative dielectric constant (23 ° C., 20 kHz) of the vinylidene fluoride resin of the present embodiment is preferably 6.0 or more, more preferably 8.0 or more, and further preferably 9.0 or more.
- the vinylidene fluoride resin of this embodiment may be a vinylidene fluoride copolymer of a vinylidene fluoride monomer and another monomer, or a vinylidene fluoride alone composed of only one type of vinylidene fluoride monomer. It may be a polymer.
- examples of other monomers include vinyl fluoride, trifluoroethylene, tetrafluoroethylene, chlorotrifluoroethylene, hexafluoropropylene, and perfluoroalkyl vinyl ethers represented by perfluoromethyl vinyl ether.
- hexafluoropropylene is most preferable from the viewpoint of easy processability.
- the vinylidene fluoride-based resin of the present embodiment is preferably a vinylidene fluoride homopolymer, a vinylidene fluoride-hexafluoropropylene copolymer, or a mixture thereof. Further, when the vinylidene fluoride resin of the present embodiment is a mixture of a vinylidene fluoride homopolymer and a vinylidene fluoride-hexafluoropropylene copolymer, the mixing ratio thereof is not particularly limited.
- the molecular structure of the vinylidene fluoride resin of the present embodiment may be linear or branched.
- vinylidene fluoride resin of the present embodiment for example, a known linear polyvinylidene fluoride (for example, KF # 1100 manufactured by Kureha Co., Ltd.) can be suitably used.
- a known linear polyvinylidene fluoride for example, KF # 1100 manufactured by Kureha Co., Ltd.
- the average particle diameter of the inorganic particles in the present embodiment is preferably 0.60 ⁇ m or more and 1.8 ⁇ m or less, more preferably 0.70 ⁇ m or more and 1.6 ⁇ m or less, and further preferably 0.80 ⁇ m or more, 1 .2 ⁇ m or less.
- the reason why the average particle diameter of the inorganic particles is preferably within this range is that when the inorganic particles are too small, the protrusions are small and the winding property of the film may be deteriorated. On the other hand, if the inorganic particles are too large, the protrusions become large and may break during stretching.
- the inorganic particles of this embodiment are preferably particles of one or more inorganic compounds selected from the group consisting of calcium carbonate, aluminum oxide, and magnesium oxide, for example.
- the composition for films contains 2 or more types of inorganic particles, the compounding ratio of these inorganic particles is not specifically limited.
- calcium carbonate known ones such as Softon 2200, Softon 2600, Softon 3200 (all manufactured by Shiroishi Calcium Co., Ltd.), SL-2200 (manufactured by Takehara Chemical Industry Co., Ltd.), and the like can be used.
- aluminum oxide for example, products known as products such as Sumitomo Chemical Co., Ltd., High Purity Chemical Laboratory, Kawai Lime Industry Co., Ltd. can be used.
- magnesium oxide for example, those known as products such as Kanjima Chemical Industry Co., Ltd., Sakai Chemical Industry Co., Ltd. can be used.
- the inorganic particles are 0.050 part by weight or more and 5.0 part by weight or less, preferably 0.050 part by weight or more, 2.5 parts per 100 parts by weight of the vinylidene fluoride resin.
- the content is not more than parts by weight, more preferably not less than 0.10 parts by weight and not more than 1.0 parts by weight. It is preferable that the content of the inorganic particles is within this range from the viewpoint of improving the winding property of the film and stretching the film without breaking.
- the film composition of the present embodiment may contain other resins in addition to the vinylidene fluoride resin and the inorganic particles.
- the polymer processing aid is 0.10 to 10 parts by weight, more preferably 0.50 to 5 parts by weight with respect to 100 parts by weight of the vinylidene fluoride resin. 0.0 part by weight or less, more preferably 1.0 part by weight or more and 3.0 parts by weight or less.
- an acrylic processing aid is preferable from the viewpoint of improving the smoothness and stretchability of the film.
- the vinylidene fluoride resin film according to the present embodiment for example, after mixing inorganic particles with pellets made of vinylidene fluoride resin, melting and extruding this vinylidene fluoride resin, and further stretching Can be obtained by:
- the manufacturing method of a vinylidene fluoride resin film which concerns on this embodiment is not limited to the following method.
- the mixed resin can be obtained by melt extrusion.
- the set temperature of the extruder may be a temperature at which the mixed resin can be melt-extruded, for example, 150 ° C. to 250 ° C.
- melt-extruded resin is cut with, for example, a pelletizer, polyvinylidene fluoride pellets containing inorganic particles can be obtained.
- the polyvinylidene fluoride resin film according to the present embodiment is formed by melt extrusion using the polyvinylidene fluoride pellets containing inorganic particles thus obtained and stretching the obtained unstretched film. Can be obtained.
- Examples of the stretching method include biaxial stretching.
- a method for producing a polyvinylidene fluoride film obtained by biaxial stretching hereinafter also referred to as “biaxially stretched polyvinylidene fluoride film”.
- polyvinylidene fluoride pellets containing inorganic particles are supplied to a known melt extruder.
- a known melt extruder As an extruder in the production of a biaxially stretched polyvinylidene fluoride film, a uniaxial or biaxial extruder can be used.
- the polymer obtained by melting and extruding with an extruder is filtered through a filter.
- a filter can be appropriately selected from filters having openings in a range where the increase in resin pressure does not substantially affect the processability of the film.
- this polymer is extruded into a sheet form from, for example, a T die, and is cooled and solidified on a casting roll to form an unstretched film.
- the biaxially stretched polyvinylidene fluoride film can be produced, for example, using sequential stretching.
- the first longitudinal stretching is performed at a stretching temperature of 110 ° C. or more and 170 ° C. or less, preferably 140 ° C. or more and 165 ° C. or less, and a stretching ratio of 2.0 times or more and 10 times or less, Stretch as 2.5 times or more and 4.0 times or less.
- the stretching temperature in the longitudinal direction is important for suppressing the generation of scratches, and when the stretching temperature in the longitudinal direction is lower than 110 ° C., scratches are generated on the film surface or the film is easily broken.
- the stretching temperature in the longitudinal direction is higher than 170 ° C., the film surface is damaged by heat and the film becomes brittle.
- the temperature of the tenter stretching machine is 110 ° C. or more and 170 ° C. or less, preferably 140 ° C. or more and 165 ° C. or less, and the stretching ratio is 2.0 times or more in the width direction. Stretching is 10 times or less, preferably 3.0 times or more and 5.0 times or less. If the stretching temperature in the width direction is lower than 110 ° C., the film is liable to break, and if the stretching temperature in the width direction is higher than 170, sufficient strength cannot be obtained. Moreover, it is unpreferable also from the point which a stretch spot generate
- the total stretching ratio in the longitudinal direction and the width direction is 4.0 times or more and 20 times or less, preferably 5.0 times or more and 10 times or less. If the total draw ratio is less than 4.0 times, stretch spots are likely to occur, and it is difficult to obtain sufficient strength for the film. On the other hand, if it is larger than 10 times, film breakage tends to occur and it is difficult to produce a stable film.
- a timely magnification can be selected to achieve the target breaking strength, but in order to increase the breaking strength in the width direction, the stretching ratio in the width direction should be set higher than that in the longitudinal direction. Is more preferable.
- the biaxially stretched film is wound into a suitable width and length by a slitting process.
- the manufacturing method of the vinylidene fluoride resin film according to the present embodiment is not limited to the above-described method.
- a vinylidene fluoride resin film raw material
- a polyvinylidene fluoride resin simple substance Inorganic particles may be added.
- the vinylidene fluoride resin film of the present embodiment obtained as described above has a plurality of protrusions on at least one surface of the vinylidene fluoride resin film.
- the protrusions may be formed on both surfaces of the vinylidene fluoride resin film.
- the number of protrusions exceeding 0.10 ⁇ m from the smooth surface among the plurality of protrusions is 7.0 or more per 0.10 mm 2 of vinylidene fluoride resin film, 50 Or less. If the number of protrusions is within this range, there is no risk of air escape failure during winding of the vinylidene fluoride resin film. Moreover, if it is this range, generation
- the smooth surface means a surface on which no protrusion is specifically formed.
- the average protrusion height of the protrusions of the vinylidene fluoride resin film is preferably 0.40 ⁇ m or more and 0.80 ⁇ m or less, more preferably 0.40 ⁇ m or more and 0.60 ⁇ m or less, and still more preferably, It is 0.40 ⁇ m or more and 0.50 ⁇ m or less. When the average protrusion height of the protrusion is within this range, it is preferable from the viewpoint of improving the winding property of the film.
- the “average protrusion height” of the protrusion means an average value of the height from the film smooth surface of the protrusion exceeding 0.10 ⁇ m existing on the vinylidene fluoride resin film surface.
- the number of protrusions was specified using an analysis software from a film smooth surface included in the range by specifying an arbitrary range from a laser microscope (manufactured by Keyence Corporation). The number of protrusions exceeding 10 ⁇ m. In order to accurately count the number of protrusions, it is preferable to count the protrusions by enlarging the field of view at least 10 times or more, preferably 50 times or more.
- the film thickness of the vinylidene fluoride resin film of the present embodiment is preferably 1.0 ⁇ m or more and 8.0 ⁇ m or less, more preferably 1.5 ⁇ m or more and 5.0 ⁇ m or less, and more preferably 1.5 ⁇ m or more. More preferably, it is 3.0 ⁇ m or less. By setting it as this range, products, such as a capacitor produced using the vinylidene fluoride resin film concerning this embodiment, can be reduced in size.
- the average particle diameter of inorganic particles contained in the film composition is d ( ⁇ m), and the film thickness of the vinylidene fluoride resin film is t ( ⁇ m).
- d the average particle diameter of inorganic particles contained in the film composition
- t the film thickness of the vinylidene fluoride resin film
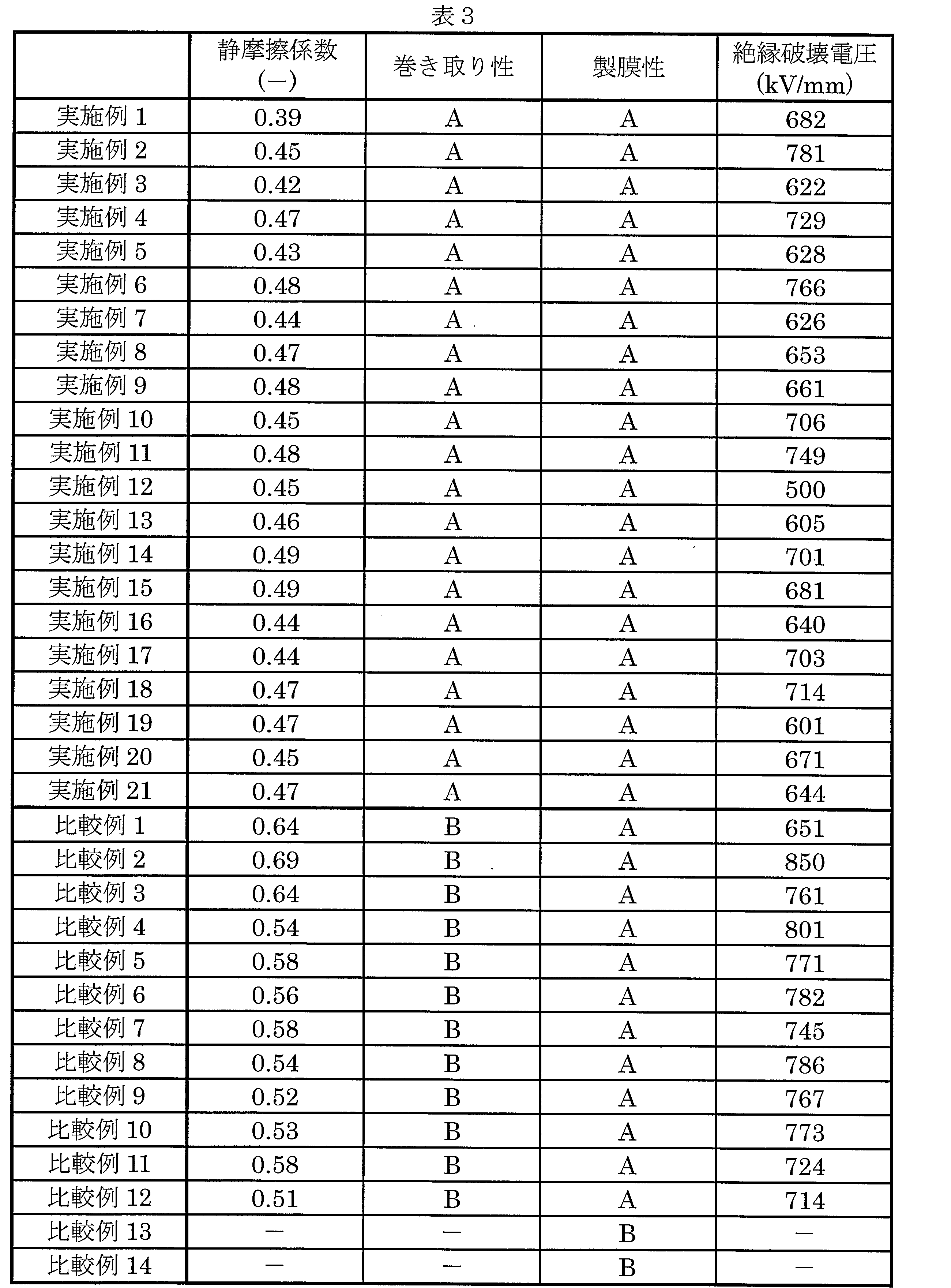
- the vinylidene fluoride resin film of the present embodiment has a static friction coefficient of 0.20 or more and 0.50 or less, more preferably 0.30 or more and 0.50 or less, and still more preferably 0.00. 40 or more and 0.50 or less.
- the static friction coefficient means a friction coefficient when the film starts to slide.
- the vinylidene fluoride resin film of this embodiment is based on vinylidene fluoride resin, it has a high dielectric constant.
- the winding property of the film is improved by forming the protrusions.
- the vinylidene fluoride resin film of this embodiment has a dielectric breakdown voltage of 500 kV / mm or more and a sufficiently high dielectric breakdown strength. Therefore, the vinylidene fluoride resin film of this embodiment can be stably used as a film capacitor as compared with the vinylidene fluoride resin film produced using only the vinylidene fluoride resin.
- Dielectric breakdown strength or “dielectric breakdown strength” is as defined in JIS-C2110 and JIS-C2151, and the breakdown voltage is divided by the thickness of the insulator (in this specification, a film). This is the dielectric breakdown voltage per thickness.
- the metal vapor deposition capacitor is short-circuited even during abnormal discharge, resulting in dielectric breakdown. There is little possibility of doing.
- the metal vapor deposition capacitor is short-circuited even during abnormal discharge, resulting in dielectric breakdown.
- the metal vapor deposition capacitor is short-circuited even during abnormal discharge, resulting in dielectric breakdown.
- the metal vapor deposition capacitor is short-circuited even during abnormal discharge, resulting in dielectric breakdown.
- there is a gap between the films so it is possible to quickly cool and release the generated gas. (Self-healing property) is improved.
- the vinylidene fluoride resin film of the present embodiment is preferably used for a capacitor, for example.
- the static friction coefficient is preferably 0.20 or more and 0.50 or less.
- the average particle diameter of the inorganic particles is d ( ⁇ m) and the film thickness of the vinylidene fluoride resin film is t ( ⁇ m)
- the film composition contains 0.050 part by weight or more and 5.0 parts by weight or less of inorganic particles with respect to 100 parts by weight of the vinylidene fluoride resin. Is preferred.
- the inorganic particles are preferably particles of one or more inorganic compounds selected from the group consisting of calcium carbonate, aluminum oxide, and magnesium oxide.
- the vinylidene fluoride resin is preferably a vinylidene fluoride homopolymer, but a vinylidene fluoride-hexafluoropropylene copolymer, or a copolymer thereof. Mixtures can be used.
- the film thickness is preferably 1.0 ⁇ m or more and 10 ⁇ m or less.
- the average particle diameter of the inorganic particles is 0.50 ⁇ m or more and 2.0 ⁇ m or less.
- the average protrusion height is preferably 0.40 ⁇ m or more and 1.0 ⁇ m or less.
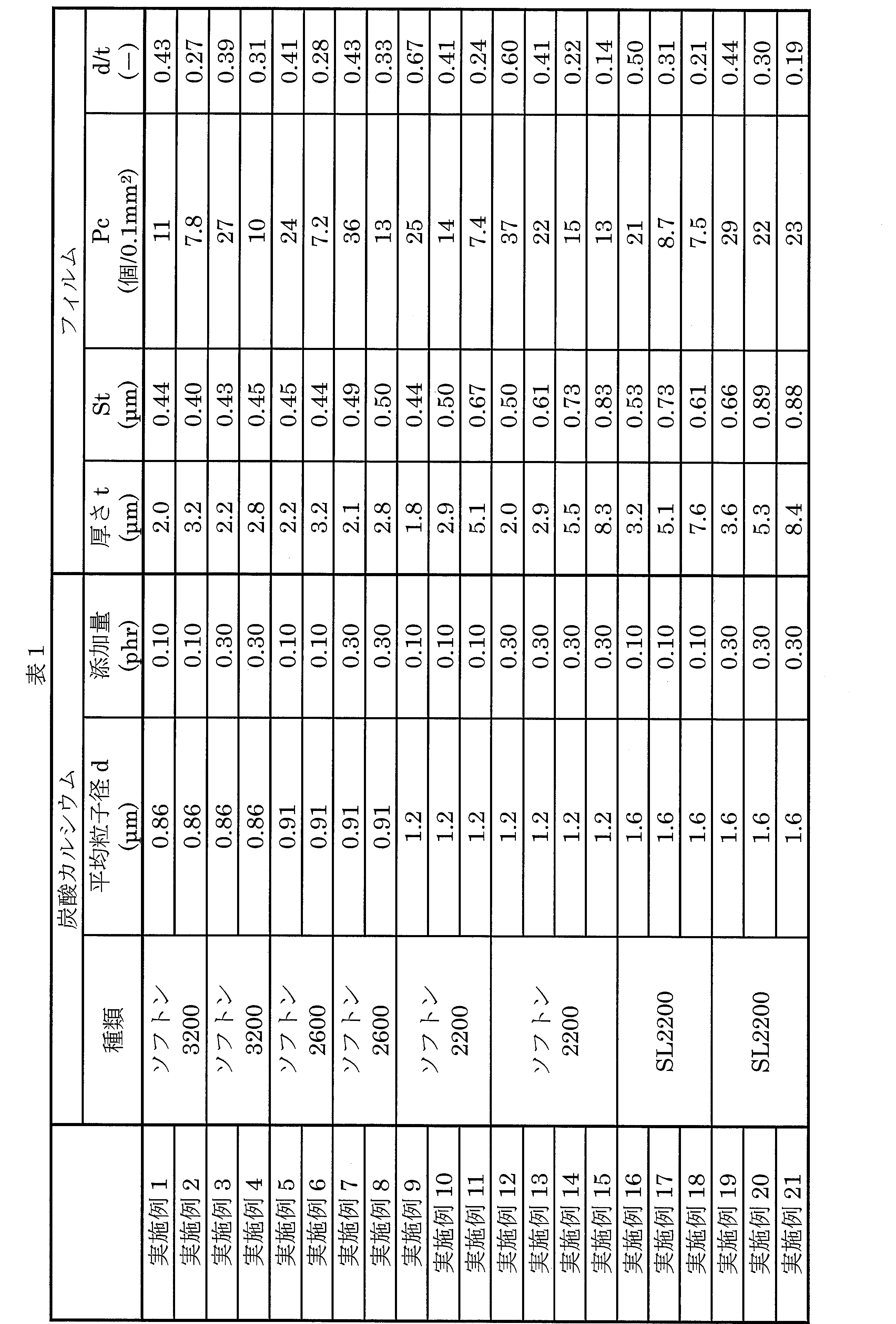
- Example 1 ⁇ Production of film> (Preparation of film composition) Calcium carbonate (Softon 3200, Shiraishi Calcium Co., Ltd.) is used as inorganic particles for 100 parts by weight of linear polyvinylidene fluoride resin (Kureha KF # 1100) having a melt flow rate (MFR) of 2 to 4 g / 10 min. The average particle size was 0.86 ⁇ m) and 0.10 parts by weight were added and mixed uniformly to prepare a film composition.
- Kureha KF # 1100 linear polyvinylidene fluoride resin having a melt flow rate (MFR) of 2 to 4 g / 10 min.
- MFR melt flow rate
- the film composition obtained as described above was supplied to a unidirectional biaxial kneading extruder (TEM-26, manufactured by Toshiba Machine Co., Ltd.) whose temperature was adjusted to 160 ° C. to 230 ° C., and melt kneaded to produce pellets.
- TEM-26 unidirectional biaxial kneading extruder
- Example 2 In the same manner as in Example 1, a biaxially stretched film having a thickness of 3.2 ⁇ m was produced.
- Example 3 A biaxially stretched film having a thickness of 2.2 ⁇ m was produced in the same manner as in Example 1 except that the addition amount of calcium carbonate was changed to 0.30 part by weight.
- Example 4 In the same manner as in Example 3, a biaxially stretched film having a thickness of 2.8 ⁇ m was produced.
- Example 5 A biaxially stretched film having a thickness of 2.2 ⁇ m was produced in the same manner as in Example 1 except that Softon 3200 was changed to Softon 2600 (manufactured by Shiraishi Calcium Co., Ltd., average particle size: 0.91 ⁇ m).
- Example 6 A biaxially stretched film having a thickness of 3.2 ⁇ m was produced in the same manner as in Example 5.
- Example 7 A biaxially stretched film having a thickness of 2.1 ⁇ m was produced in the same manner as in Example 1 except that the amount of Softon 2600 added was changed to 0.30 part by weight.
- Example 8 In the same manner as in Example 7, a biaxially stretched film having a thickness of 2.8 ⁇ m was produced.
- Example 9 A biaxially stretched film having a thickness of 1.8 ⁇ m was produced in the same manner as in Example 1 except that Softon 3200 was changed to Softon 2200 (manufactured by Shiraishi Calcium Co., Ltd., average particle size 1.2 ⁇ m).
- Example 10 In the same manner as in Example 9, a biaxially stretched film having a thickness of 2.9 ⁇ m was produced.
- Example 11 A biaxially stretched film having a thickness of 5.1 ⁇ m was produced in the same manner as Example 10.
- Example 12 A biaxially stretched film having a thickness of 2.0 ⁇ m was produced in the same manner as in Example 9 except that the addition amount of Softon 2200 was changed to 0.30 part by weight.
- Example 13 In the same manner as in Example 12, a biaxially stretched film having a thickness of 2.9 ⁇ m was produced.
- Example 14 A biaxially stretched film having a thickness of 5.5 ⁇ m was produced in the same manner as in Example 12.
- Example 15 A biaxially stretched film having a thickness of 8.3 ⁇ m was produced in the same manner as in Example 12.
- Example 16 A 3.2 ⁇ m biaxially stretched film was produced in the same manner as in Example 1 except that Softon 3200 was changed to SL-2200 (manufactured by Takehara Chemical Industries, Ltd., average particle size 1.6 ⁇ m).
- Example 17 A biaxially stretched film having a thickness of 5.1 ⁇ m was produced in the same manner as in Example 16.
- Example 18 In the same manner as in Example 16, a biaxially stretched film having a thickness of 7.6 ⁇ m was produced.
- Example 19 A biaxially stretched film having a thickness of 3.6 ⁇ m was produced in the same manner as in Example 16 except that the addition amount of SL-2200 was changed to 0.30 part by weight.
- Example 20 A biaxially stretched film having a thickness of 5.3 ⁇ m was produced in the same manner as in Example 19.
- Example 21 In the same manner as in Example 19, a biaxially stretched film having a thickness of 8.4 ⁇ m was produced.
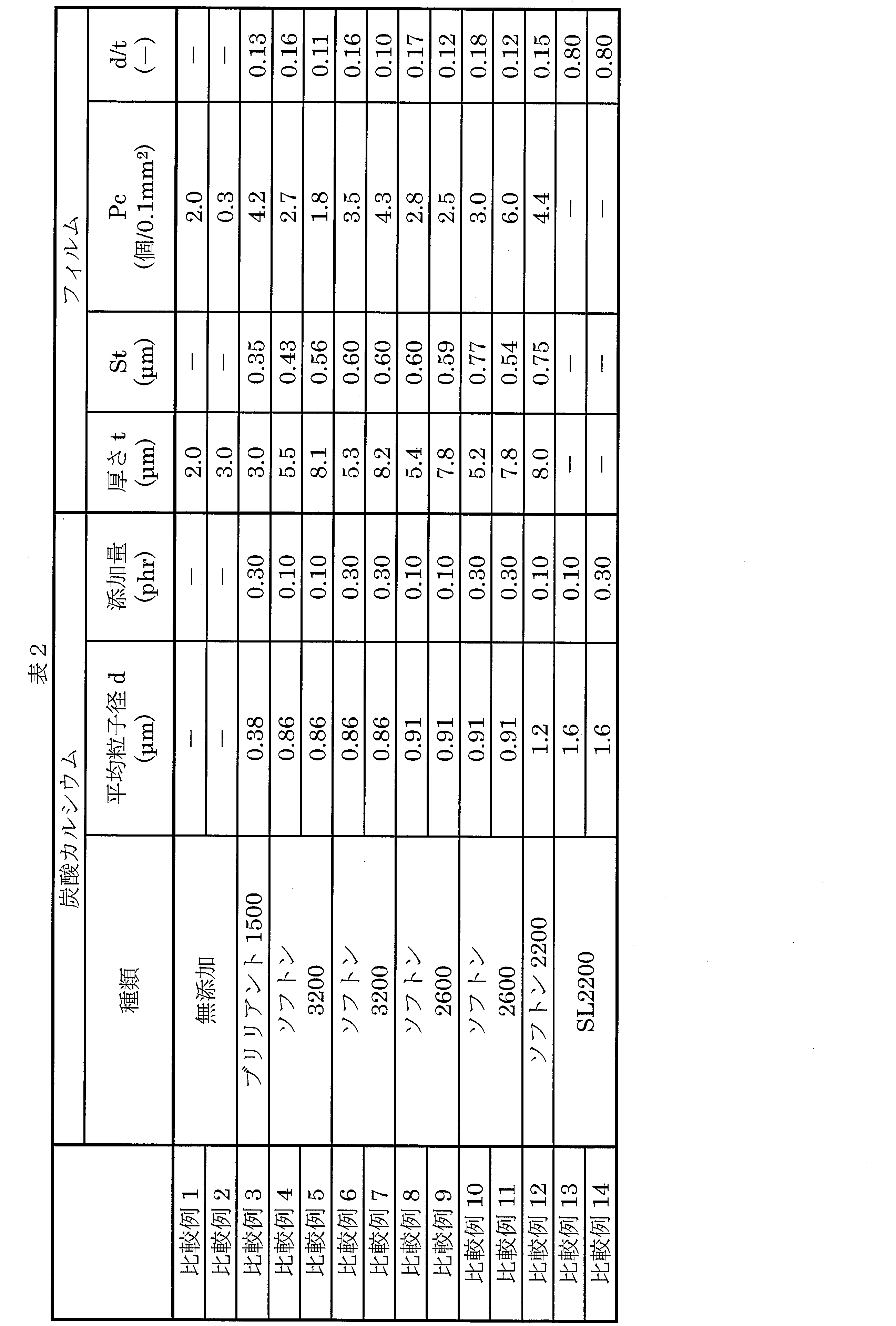
- Example 1 A biaxially stretched film having a thickness of 2.0 ⁇ m was produced in the same manner as in Example 1 except that calcium carbonate was not added.
- Comparative Example 2 In the same manner as in Comparative Example 1, a biaxially stretched film having a thickness of 3.0 ⁇ m was produced.
- Comparative Example 8 A biaxially stretched film having a thickness of 5.4 ⁇ m was produced in the same manner as in Comparative Example 4 except that Softon 3200 was changed to Softon 2600 (manufactured by Shiraishi Calcium Co., Ltd., average particle size: 0.91 ⁇ m).
- Comparative Example 10 A biaxially stretched film having a thickness of 5.2 ⁇ m was produced in the same manner as in Comparative Example 8 except that the amount of Softon 2600 added was changed to 0.30 part by weight.
- Comparative Example 12 A biaxially stretched film having a thickness of 8.0 ⁇ m was prepared in the same manner as in Comparative Example 5 except that Softon 3200 was changed to Softon 2200 (manufactured by Shiraishi Calcium Co., Ltd., average particle size 1.2 ⁇ m).
- Comparative Example 13 A biaxially stretched film having a thickness of 2.0 ⁇ m was tried in the same manner as Comparative Example 1 except that 0.10 parts by weight of SL-2200 (manufactured by Takehara Chemical Industry Co., Ltd., average particle size 1.6 ⁇ m) was added. However, the film broke during stretching and a film was not obtained.
- SL-2200 manufactured by Takehara Chemical Industry Co., Ltd., average particle size 1.6 ⁇ m
- the particle size is measured using a particle size distribution analyzer (“MicroTracMT3300EX II” manufactured by Microtrack Bell Co., Ltd.). Was measured.
- the refractive index of the dispersion solvent was 1.33, and the refractive index of the inorganic particles was 1.49.
- the surface shape of the film was measured using a shape analysis laser microscope (Keyence Co., Ltd. “VK-X250”). From the observed image, the average protrusion height exceeding 0.10 ⁇ m from the smooth surface where no protrusion was present, and The number of protrusions per 0.10 mm 2 was calculated from volume area measurement analysis. The number of measurements per sample was 10 times, and the average value was obtained from all measured values.
- the setting conditions of the shape analysis laser microscope are as follows. The details of the measuring equipment and conditions are as follows.
- Friction measuring machine (TR type” manufactured by Toyo Seiki Seisakusho) Measurement direction: Film longitudinal direction Test speed: 100 mm / min Sliding piece mass: 200 g (Winding property)
- the film was wound up, and wrinkles (longitudinal stripes) or displacement generated in the film after winding up were visually observed.
- wrinkle generated in the film after winding was less than 5
- the film winding property was “good: A”
- the wrinkle was 5 or more
- the film winding property was “inferior: B”. If the film winding property is evaluated as “good: A”, the film is considered to be at a practical level.
- the dielectric breakdown voltage was measured by applying JIS-C2151. The measured value of dielectric breakdown voltage was divided by the film thickness of the sample and expressed in kV / mm. Measuring equipment and conditions are as shown below.
- the vinylidene fluoride resin film according to an embodiment of the present invention is used for communication, electronic equipment, power, medium / low pressure phase advance, automatic external defibrillators, especially for inverters as automobile parts, etc. It can be used as a film capacitor, or a film material such as a piezoelectric element, a pyroelectric element, and a dielectric for carrying a transfer body.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Power Engineering (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Physics & Mathematics (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Inorganic Chemistry (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
Abstract
Description
本実施形態に係るフィルム用組成物とは、少なくとも、フッ化ビニリデン系樹脂と、無機粒子とを含有する組成物である。
以下、本実施形態に係るフッ化ビニリデン系樹脂フィルムの製造方法の一実施形態について説明する。
例えば上記のようにして得られる本実施形態のフッ化ビニリデン系樹脂フィルムは、フッ化ビニリデン系樹脂フィルムの少なくとも一方の面に複数の突起を有している。なお、本実施形態において、突起は、フッ化ビニリデン系樹脂フィルムの両面に形成されていてもよい。
<フィルムの作製>
(フィルム用組成物の調製)
メルトフローレイト(MFR)が2~4g/10分である直鎖状ポリフッ化ビニリデン樹脂(クレハ社製KF#1100)100重量部に対し、無機粒子として、炭酸カルシウム(ソフトン3200、白石カルシウム社製、平均粒子径0.86μm)0.10重量部を添加して均一になるように混合して、フィルム用組成物を調製した。
上記により得られたフィルム用組成物を、160℃~230℃に温調した同方向二軸混練押出機(東芝機械社製、TEM-26)に供給し、溶融混練し、ペレットを作製した。
上記により得られたペレットを単軸押出機(ユニオン・プラスチックス社製)に投入し、樹脂温度270℃~280℃の樹脂混練物を200メッシュのステンレスフィルターに通してTダイから押出し、初めに表面を100℃に保った第一金属ドラム上にキャストし、次に、表面を50℃に保った第二金属ドラム上でさらに冷却し、こうしてフィルム用組成物から未延伸シートを作製した。
延伸ロールを用いて、上記により得られた未延伸シートを縦方向に150~160℃で一軸延伸し、次いで150℃~160℃に設定したテンター延伸機に導入し、横方向に延伸して、厚み2.0μmの二軸延伸フィルムを得た。
実施例1と同様にして、厚み3.2μmの二軸延伸フィルムを作製した。
炭酸カルシウムの添加量を0.30重量部に変えたこと以外は、実施例1と同様にして、厚み2.2μmの二軸延伸フィルムを作製した。
実施例3と同様にして、厚み2.8μmの二軸延伸フィルムを作製した。
ソフトン3200をソフトン2600(白石カルシウム社製、平均粒子径0.91μm)に変えたこと以外は、実施例1と同様にして、厚み2.2μmの二軸延伸フィルムを作製した。
実施例5と同様にして、厚み3.2μmの二軸延伸フィルムを作製した。
ソフトン2600の添加量を0.30重量部に変えたこと以外は、実施例1と同様にして、厚み2.1μmの二軸延伸フィルムを作製した。
実施例7と同様にして、厚み2.8μmの二軸延伸フィルムを作製した。
ソフトン3200をソフトン2200(白石カルシウム社製、平均粒子径1.2μm)に変えたこと以外は、実施例1と同様にして、厚み1.8μmの二軸延伸フィルムを作製した。
実施例9と同様にして、厚み2.9μmの二軸延伸フィルムを製造した。
実施例10と同様にして、厚み5.1μmの二軸延伸フィルムを作製した。
ソフトン2200の添加量を0.30重量部に変えた以外は、実施例9と同様にして、厚み2.0μmの二軸延伸フィルムを作製した。
実施例12と同様にして、厚み2.9μmの二軸延伸フィルムを作製した。
実施例12と同様にして、厚み5.5μmの二軸延伸フィルムを作製した。
実施例12と同様にして、厚み8.3μmの二軸延伸フィルムを作製した。
ソフトン3200をSL-2200(竹原化学工業社製、平均粒子径1.6μm)に変えたこと以外は、実施例1と同様にして、3.2μmの二軸延伸フィルムを作製した。
実施例16と同様にして、厚み5.1μmの二軸延伸フィルムを作製した。
実施例16と同様にして、厚み7.6μmの二軸延伸フィルムを作製した。
SL-2200の添加量を0.30重量部に変えた以外は、実施例16と同様にして、厚み3.6μmの二軸延伸フィルムを作製した。
実施例19と同様にして、厚み5.3μmの二軸延伸フィルムを作製した。
実施例19と同様にして、厚み8.4μmの二軸延伸フィルムを作製した。
炭酸カルシウムを添加しなかったこと以外は、実施例1と同様にして、厚み2.0μmの二軸延伸フィルムを作製した。
比較例1と同様にして、厚み3.0μmの二軸延伸フィルムを作製した。
ソフトン3200をブリリアント1500(白石カルシウム社製、平均粒子径0.38μm)に変えたこと、及びブリリアント1500の添加量を0.30重量部にした以外は、実施例1と同様にして、厚み3.0μmの二軸延伸フィルムを作製した。
ブリリアント1500をソフトン3200に変えたこと、及びソフトン3200の添加量を0.10重量部にした以外は、比較例3と同様にして、厚み5.5μmの二軸延伸フィルムを作製した。
比較例4と同様にして、厚み8.1μmの二軸延伸フィルムを作製した。
ソフトン3200の添加量を0.30重量部に変えたこと以外は、比較例4と同様にして、厚み5.3μmの二軸延伸フィルムを作製した。
比較例6と同様にして、厚み8.2μmの二軸延伸フィルムを作製した。
ソフトン3200をソフトン2600(白石カルシウム社製、平均粒子径0.91μm)に変えたこと以外は、比較例4と同様にして、厚み5.4μmの二軸延伸フィルムを作製した。
比較例8と同様にして、厚み7.8μmの二軸延伸フィルムを作製した。
ソフトン2600の添加量を0.30重量部に変えたこと以外は、比較例8と同様にして、厚み5.2μmの二軸延伸フィルムを作製した。
比較例10と同様にして、厚み7.8μmの二軸延伸フィルムを作製した。
ソフトン3200をソフトン2200(白石カルシウム社製、平均粒子径1.2μm)に変えたこと以外は、比較例5と同様にして、厚み8.0μmの二軸延伸フィルムを作製
した。
SL-2200(竹原化学工業社製、平均粒子径1.6μm)0.10重量部を添加したこと以外は、比較例1と同様にして、厚み2.0μmの二軸延伸フィルムの作製を試みたが、延伸の際に破断してフィルムは得られなかった。
SL-2200の添加量を0.30重量部に変えた以外は、比較例13と同様にして、厚み2.0μmの二軸延伸フィルムの作製を試みたが、延伸の際に破断してフィルムは得られなかった。
実施例1~21及び比較例1~14の各無機粒子に対して、以下の試験を行った。
実施例1~21及び比較例1~14の各フィルムに対して、以下の試験を行った。
フィルムを10枚重ねに折ってから、フィルムの中心をTD方向に5.0mm間隔で40点マーキングした。それから、卓上型厚み計(株式会社山文電気製「TOF-5R」)を用いて、マイクロメータ法によって40点すべてにおけるフィルム厚を測定し、各値を10で割った計40点の平均厚みを算出した。
上記により得られたフィルム厚を用いて、平均粒子径(d)/をフィルム厚(t)の値を算出した。
形状解析レーザ顕微鏡(株式会社キーエンス社製「VK-X250」)を用いてフィルムの表面形状を測定し、観察画像より、突起が存在していない平滑面から0.10μmを超える平均突起高さおよび0.10mm2あたりの突起個数を体積面積計測解析から算出した。なお、1サンプルあたりの測定数を10回とし、全測定値から平均値を求めた。また、形状解析レーザ顕微鏡の設定条件は以下の通りである。なお、測定機器および条件の詳細は以下の通りである。
対物レンズ :50倍
フィルター種別 :ガウシアン
S-フィルター :なし
F-オペレーション:なし
L-フィルター :0.080mm
終末効果補正:有効
1ショット測定範囲:212.347×287.222(μm2)
測定範囲:3×3マス(9ショット)
測定パラメータ:平均突起高さ(St)、0.10mm2あたりの突起個数(Pc)
解析範囲:0.40×0.60(mm2)
高さしきい値:0.10μm
微小領域設定下限値:100Pixel
(摩擦係数)
ガラス盤上にフッ化ビニリデン系樹脂フィルムを貼付け、金属すべり片には厚み40μmのポリプロピレンフィルム(東洋紡株式会社製パイレンフィルム-CT P1011)を貼付け、スプリングなしで行った以外はJIS-K7125:1999を準用して、静摩擦係数を算出した。なお、1サンプルあたりの測定数を5回とし、全測定値から平均値を求めた。測定機器、および条件は以下に示す通りである。
測定方向 :フィルム長手方向
試験速度 :100mm/min
すべり片質量 :200g
(巻き取り性)
フィルムを巻き取り、巻き取り後にフィルムに発生するシワ(縦スジ)またはズレを目視で観察した。巻き取り後のフィルムに発生したシワが5本未満であれば、フィルム巻き取り性「良好:A」、シワが5本以上であれば、フィルム巻き取り性「劣る:B」と評価した。なお、フィルム巻き取り性が「良好:A」と評価されれば、フィルムは実用可能なレベルであると考えられる。
フィルムを延伸して製膜する際に、フィルムが破れずに製膜可能であれば、フィルム製膜性が「良好:A」、破れが発生する等して製膜不能であれば製膜性が「悪い:B」と判断した。
JIS-C2151を準用して絶縁破壊電圧の測定を実施した。絶縁破壊電圧の測定値をサンプルのフィルム厚で除して、kV/mmで表記した。測定機器及び条件は、以下に示す通りである。
Claims (9)
- フッ化ビニリデン系樹脂および無機粒子を含有するフィルム用組成物を用いて作製されたフッ化ビニリデン系樹脂フィルムであって、
当該フッ化ビニリデン系樹脂フィルムの少なくとも一方面に複数の突起を有し、
前記複数の突起のうち、突起が存在していない平滑面から0.10μmを超える突起の数が、前記フッ化ビニリデン系樹脂フィルム0.10mm2あたり7.0個以上、50個以下であることを特徴とするフッ化ビニリデン系樹脂フィルム。 - 静摩擦係数が0.20以上、0.50以下であることを特徴とする請求項1に記載のフッ化ビニリデン系樹脂フィルム。
- 前記無機粒子の平均粒子径をd(μm)、前記フッ化ビニリデン系樹脂フィルムのフィルム厚をt(μm)としたとき、0.14<d/t<0.75の関係式を満たすことを特徴とする請求項1または2に記載のフッ化ビニリデン系樹脂フィルム。
- 前記フィルム用組成物は、前記フッ化ビニリデン系樹脂100重量部に対し、前記無機粒子を0.050重量部以上、5.0重量部以下含有することを特徴とする請求項1~3のいずれか1項に記載のフッ化ビニリデン系樹脂フィルム。
- 前記無機粒子は、炭酸カルシウム、酸化アルミニウムおよび酸化マグネシウムからなる群から選択される1種またはそれ以上の無機化合物の粒子であることを特徴とする請求項1~4のいずれか1項に記載のフッ化ビニリデン系樹脂フィルム。
- 前記フッ化ビニリデン系樹脂は、フッ化ビニリデンの単独重合体もしくはフッ化ビニリデン-ヘキサフルオロプロピレン共重合体、またはそれらの混合物であることを特徴とする請求項1~5のいずれか1項に記載のフッ化ビニリデン系樹脂フィルム。
- フィルム厚が1.0μm以上、10μm以下であることを特徴とする請求項1~6のいずれか1項に記載のフッ化ビニリデン系樹脂フィルム。
- 前記無機粒子の平均粒子径が0.50μm以上、2.0μm以下であることを特徴とする請求項1~7のいずれか1項に記載のフッ化ビニリデン系樹脂フィルム。
- 前記突起の平均突起高さは0.40μm以上、1.0μm以下であることを特徴とする請求項1~8のいずれか1項に記載のフッ化ビニリデン系樹脂フィルム。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/467,559 US10988586B2 (en) | 2017-01-25 | 2018-01-16 | Vinylidene fluoride resin film |
| EP18744722.2A EP3575349B1 (en) | 2017-01-25 | 2018-01-16 | Vinylidene fluoride resin film |
| CN201880004276.3A CN109923158B (zh) | 2017-01-25 | 2018-01-16 | 偏氟乙烯系树脂膜 |
| JP2018564491A JP6814819B2 (ja) | 2017-01-25 | 2018-01-16 | フッ化ビニリデン系樹脂フィルム |
| KR1020197012771A KR102302155B1 (ko) | 2017-01-25 | 2018-01-16 | 불화 비닐리덴계 수지 필름 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2017011677 | 2017-01-25 | ||
| JP2017-011677 | 2017-01-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018139262A1 true WO2018139262A1 (ja) | 2018-08-02 |
Family
ID=62979358
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2018/000969 WO2018139262A1 (ja) | 2017-01-25 | 2018-01-16 | フッ化ビニリデン系樹脂フィルム |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US10988586B2 (ja) |
| EP (1) | EP3575349B1 (ja) |
| JP (1) | JP6814819B2 (ja) |
| KR (1) | KR102302155B1 (ja) |
| CN (1) | CN109923158B (ja) |
| WO (1) | WO2018139262A1 (ja) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6760404B2 (ja) * | 2017-01-31 | 2020-09-23 | ダイキン工業株式会社 | フッ素樹脂フィルム |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS60199046A (ja) | 1984-03-23 | 1985-10-08 | Kureha Chem Ind Co Ltd | フツ化ビニリデン樹脂組成物 |
| JPS61123520A (ja) * | 1984-11-20 | 1986-06-11 | Mitsubishi Petrochem Co Ltd | 滑り性が改良されたプラスチツクフイルムの製造法 |
| JPS61198614A (ja) * | 1985-02-27 | 1986-09-03 | 松下電器産業株式会社 | 金属化フイルムコンデンサ |
| JPS6325024A (ja) * | 1986-07-17 | 1988-02-02 | Toray Ind Inc | 二軸延伸ポリプロピレンフイルム |
| WO2008090947A1 (ja) | 2007-01-26 | 2008-07-31 | Daikin Industries, Ltd. | 高耐電圧を有する高誘電体フィルム |
| JP2008280508A (ja) * | 2007-04-10 | 2008-11-20 | Toray Ind Inc | 二軸配向ポリアリーレンスルフィドフィルム、金属化フィルムおよびコンデンサー |
| JP2009062456A (ja) | 2007-09-06 | 2009-03-26 | Teijin Ltd | 高絶縁性フィルム |
| WO2013146367A1 (ja) | 2012-03-28 | 2013-10-03 | 東レ株式会社 | コンデンサ用二軸延伸ポリプロピレンフィルム、金属化フィルム、およびフィルムコンデンサ |
| JP2014082523A (ja) | 2010-01-20 | 2014-05-08 | Daikin Ind Ltd | 高誘電性フィルム |
| WO2015129851A1 (ja) * | 2014-02-28 | 2015-09-03 | 東レ株式会社 | 二軸配向ポリプロピレンフィルム |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6014774B2 (ja) * | 1978-03-31 | 1985-04-16 | 呉羽化学工業株式会社 | 絶縁耐力の優れたポリ弗化ビニリデンフイルム及びその製造方法 |
| JPS61123520U (ja) * | 1985-01-19 | 1986-08-04 | ||
| JPH10284340A (ja) * | 1997-04-11 | 1998-10-23 | Diafoil Co Ltd | コンデンサ用熱可塑性樹脂フィルム |
| JP2008090947A (ja) | 2006-10-03 | 2008-04-17 | Teac Corp | A/d変換回路及び光ディスク装置 |
| KR101484797B1 (ko) | 2007-06-21 | 2015-01-20 | 데이진 가부시키가이샤 | 절연성 필름 |
| KR20090062456A (ko) | 2007-12-13 | 2009-06-17 | 한국산업기술평가원(관리부서:요업기술원) | 이온발생클러스터용 유전체 세라믹스 및 그 제조방법 |
| JP5679167B2 (ja) * | 2010-10-18 | 2015-03-04 | 信越ポリマー株式会社 | フィルムキャパシタ用フィルム |
| US9151316B2 (en) * | 2012-07-25 | 2015-10-06 | Alan R. Smith | Fastener with unidirectional latch |
| KR20140082523A (ko) | 2012-12-24 | 2014-07-02 | 제일모직주식회사 | 반도체 소자 밀봉용 에폭시 수지 조성물 및 이를 사용하여 밀봉된 반도체 소자 |
| CN106024946A (zh) * | 2016-06-16 | 2016-10-12 | 杭州福斯特光伏材料股份有限公司 | 一种图案化功率增益型太阳能背板材料 |
-
2018
- 2018-01-16 JP JP2018564491A patent/JP6814819B2/ja active Active
- 2018-01-16 WO PCT/JP2018/000969 patent/WO2018139262A1/ja unknown
- 2018-01-16 CN CN201880004276.3A patent/CN109923158B/zh active Active
- 2018-01-16 KR KR1020197012771A patent/KR102302155B1/ko active IP Right Grant
- 2018-01-16 EP EP18744722.2A patent/EP3575349B1/en active Active
- 2018-01-16 US US16/467,559 patent/US10988586B2/en active Active
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS60199046A (ja) | 1984-03-23 | 1985-10-08 | Kureha Chem Ind Co Ltd | フツ化ビニリデン樹脂組成物 |
| JPS61123520A (ja) * | 1984-11-20 | 1986-06-11 | Mitsubishi Petrochem Co Ltd | 滑り性が改良されたプラスチツクフイルムの製造法 |
| JPS61198614A (ja) * | 1985-02-27 | 1986-09-03 | 松下電器産業株式会社 | 金属化フイルムコンデンサ |
| JPS6325024A (ja) * | 1986-07-17 | 1988-02-02 | Toray Ind Inc | 二軸延伸ポリプロピレンフイルム |
| WO2008090947A1 (ja) | 2007-01-26 | 2008-07-31 | Daikin Industries, Ltd. | 高耐電圧を有する高誘電体フィルム |
| JP2008280508A (ja) * | 2007-04-10 | 2008-11-20 | Toray Ind Inc | 二軸配向ポリアリーレンスルフィドフィルム、金属化フィルムおよびコンデンサー |
| JP2009062456A (ja) | 2007-09-06 | 2009-03-26 | Teijin Ltd | 高絶縁性フィルム |
| JP2014082523A (ja) | 2010-01-20 | 2014-05-08 | Daikin Ind Ltd | 高誘電性フィルム |
| WO2013146367A1 (ja) | 2012-03-28 | 2013-10-03 | 東レ株式会社 | コンデンサ用二軸延伸ポリプロピレンフィルム、金属化フィルム、およびフィルムコンデンサ |
| WO2015129851A1 (ja) * | 2014-02-28 | 2015-09-03 | 東レ株式会社 | 二軸配向ポリプロピレンフィルム |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109923158A (zh) | 2019-06-21 |
| EP3575349B1 (en) | 2021-11-10 |
| JPWO2018139262A1 (ja) | 2019-07-25 |
| EP3575349A1 (en) | 2019-12-04 |
| US20190338091A1 (en) | 2019-11-07 |
| EP3575349A4 (en) | 2020-02-19 |
| KR102302155B1 (ko) | 2021-09-14 |
| KR20190065361A (ko) | 2019-06-11 |
| CN109923158B (zh) | 2021-09-28 |
| US10988586B2 (en) | 2021-04-27 |
| JP6814819B2 (ja) | 2021-01-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5062783B2 (ja) | ポリオレフィン製微多孔膜 | |
| JP6304470B1 (ja) | 二軸延伸ポリプロピレンフィルム、コンデンサ用金属化フィルム、及び、コンデンサ | |
| JP5949513B2 (ja) | 二軸延伸ポリオレフィンフィルム、該フィルムの製造方法、金属蒸着ポリオレフィンフィルムおよびフィルムコンデンサ | |
| JP2009185093A (ja) | ポリオレフィン微多孔膜 | |
| KR20090094473A (ko) | 다층 다공막 및 그의 제조 방법 | |
| JP2010254794A (ja) | 微細粗面化ポリプロピレンフィルムおよびその製造方法 | |
| JP6699552B2 (ja) | ポリオレフィン微多孔膜、非水電解液系二次電池用セパレータ、及び非水電解液系二次電池 | |
| JP6439293B2 (ja) | 積層多孔性フィルム、蓄電デバイス用セパレータおよび蓄電デバイス | |
| JP7192973B2 (ja) | ポリプロピレンフィルム、金属層一体型ポリプロピレンフィルム、及び、フィルムコンデンサ | |
| JP6814819B2 (ja) | フッ化ビニリデン系樹脂フィルム | |
| JP6213617B2 (ja) | 二軸延伸ポリオレフィンフィルム、金属蒸着ポリオレフィンフィルムおよびフィルムコンデンサの製造方法 | |
| JP6814820B2 (ja) | フッ化ビニリデン系樹脂フィルム | |
| JP5484713B2 (ja) | ポリフェニレンサルファイド系樹脂フィルム及びその製造方法 | |
| JP2009062472A (ja) | ポリフェニレンスルフィドフィルムおよびそれからなるコンデンサ | |
| WO2017115799A1 (ja) | ポリオレフィン微多孔膜とその製造方法、積層ポリオレフィン微多孔膜、ロール及びポリオレフィン微多孔膜の評価方法 | |
| WO1989008677A1 (en) | Polyphenylene sulfide film and process for its production | |
| JP2015098577A (ja) | 電気絶縁用ポリフェニレンスルフィドフィルム | |
| JP2008282908A (ja) | コンデンサ用二軸配向ポリフェニレンサルファイドフィルムおよびフィルムコンデンサ | |
| JP2006104369A (ja) | 二軸配向ポリフェニレンサルファイドフィルムおよびコンデンサ | |
| JP2004149740A (ja) | 二軸配向ポリフェニレンサルファイドフィルムおよびコンデンサー | |
| JPH048455B2 (ja) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18744722 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2018564491 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20197012771 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2018744722 Country of ref document: EP Effective date: 20190826 |