WO2016114285A1 - 多能性幹細胞からの角膜上皮細胞の分化誘導 - Google Patents
多能性幹細胞からの角膜上皮細胞の分化誘導 Download PDFInfo
- Publication number
- WO2016114285A1 WO2016114285A1 PCT/JP2016/050784 JP2016050784W WO2016114285A1 WO 2016114285 A1 WO2016114285 A1 WO 2016114285A1 JP 2016050784 W JP2016050784 W JP 2016050784W WO 2016114285 A1 WO2016114285 A1 WO 2016114285A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cells
- cell
- corneal
- corneal epithelial
- epithelial
- Prior art date
Links
- 210000002919 epithelial cell Anatomy 0.000 title claims abstract description 109
- 230000004069 differentiation Effects 0.000 title claims abstract description 92
- 238000000034 method Methods 0.000 title claims abstract description 50
- 210000001778 pluripotent stem cell Anatomy 0.000 title claims abstract description 46
- 230000001939 inductive effect Effects 0.000 title claims abstract description 20
- 210000004027 cell Anatomy 0.000 claims abstract description 284
- 239000012679 serum free medium Substances 0.000 claims abstract description 17
- 239000002609 medium Substances 0.000 claims description 85
- 210000000130 stem cell Anatomy 0.000 claims description 60
- 210000003981 ectoderm Anatomy 0.000 claims description 33
- 102000007354 PAX6 Transcription Factor Human genes 0.000 claims description 31
- 101150081664 PAX6 gene Proteins 0.000 claims description 31
- 101100351026 Drosophila melanogaster ey gene Proteins 0.000 claims description 28
- 238000012258 culturing Methods 0.000 claims description 21
- 108010085895 Laminin Proteins 0.000 claims description 17
- 102000007547 Laminin Human genes 0.000 claims description 17
- 210000000933 neural crest Anatomy 0.000 claims description 17
- 210000002966 serum Anatomy 0.000 claims description 17
- 239000003112 inhibitor Substances 0.000 claims description 14
- 102100023123 Mucin-16 Human genes 0.000 claims description 12
- 101000623901 Homo sapiens Mucin-16 Proteins 0.000 claims description 11
- 239000012634 fragment Substances 0.000 claims description 11
- 210000002950 fibroblast Anatomy 0.000 claims description 10
- 210000004561 lacrimal apparatus Anatomy 0.000 claims description 10
- 210000001161 mammalian embryo Anatomy 0.000 claims description 10
- 238000004519 manufacturing process Methods 0.000 claims description 10
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 claims description 9
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 claims description 9
- 229920002674 hyaluronan Polymers 0.000 claims description 9
- 229960003160 hyaluronic acid Drugs 0.000 claims description 9
- 229930002330 retinoic acid Natural products 0.000 claims description 9
- 229960001727 tretinoin Drugs 0.000 claims description 9
- 108010031318 Vitronectin Proteins 0.000 claims description 8
- 102100035140 Vitronectin Human genes 0.000 claims description 8
- 210000004738 parenchymal cell Anatomy 0.000 claims description 8
- 108010049955 Bone Morphogenetic Protein 4 Proteins 0.000 claims description 7
- 102100024505 Bone morphogenetic protein 4 Human genes 0.000 claims description 7
- 108010035532 Collagen Proteins 0.000 claims description 7
- 102000008186 Collagen Human genes 0.000 claims description 7
- 229920001436 collagen Polymers 0.000 claims description 7
- 239000003102 growth factor Substances 0.000 claims description 7
- 210000000554 iris Anatomy 0.000 claims description 7
- 239000011435 rock Substances 0.000 claims description 7
- 108010010803 Gelatin Proteins 0.000 claims description 6
- 239000008273 gelatin Substances 0.000 claims description 6
- 229920000159 gelatin Polymers 0.000 claims description 6
- 235000019322 gelatine Nutrition 0.000 claims description 6
- 235000011852 gelatine desserts Nutrition 0.000 claims description 6
- 210000000399 corneal endothelial cell Anatomy 0.000 claims description 5
- 210000002175 goblet cell Anatomy 0.000 claims description 5
- 210000000844 retinal pigment epithelial cell Anatomy 0.000 claims description 5
- 102100037362 Fibronectin Human genes 0.000 claims description 4
- 108010067306 Fibronectins Proteins 0.000 claims description 4
- 108010039918 Polylysine Proteins 0.000 claims description 4
- 210000002469 basement membrane Anatomy 0.000 claims description 4
- 210000000744 eyelid Anatomy 0.000 claims description 4
- 239000011159 matrix material Substances 0.000 claims description 4
- 229920000656 polylysine Polymers 0.000 claims description 4
- 108010059616 Activins Proteins 0.000 claims description 3
- 102100026818 Inhibin beta E chain Human genes 0.000 claims description 3
- 108090001012 Transforming Growth Factor beta Proteins 0.000 claims description 3
- 102000009618 Transforming Growth Factors Human genes 0.000 claims description 3
- 108010009583 Transforming Growth Factors Proteins 0.000 claims description 3
- 239000000488 activin Substances 0.000 claims description 3
- 230000001886 ciliary effect Effects 0.000 claims description 3
- 210000001542 lens epithelial cell Anatomy 0.000 claims description 3
- 210000004175 meibomian gland Anatomy 0.000 claims description 3
- 102000045246 noggin Human genes 0.000 claims description 3
- 108700007229 noggin Proteins 0.000 claims description 3
- 210000001328 optic nerve Anatomy 0.000 claims description 3
- 210000001127 pigmented epithelial cell Anatomy 0.000 claims description 3
- 210000001423 scleral cell Anatomy 0.000 claims description 3
- 230000000392 somatic effect Effects 0.000 claims description 3
- 102100030742 Transforming growth factor beta-1 proprotein Human genes 0.000 claims 1
- 210000003560 epithelium corneal Anatomy 0.000 description 30
- 230000006698 induction Effects 0.000 description 29
- 210000001508 eye Anatomy 0.000 description 28
- 102100027881 Tumor protein 63 Human genes 0.000 description 26
- 101710087047 Cytoskeleton-associated protein 4 Proteins 0.000 description 25
- 101710140697 Tumor protein 63 Proteins 0.000 description 25
- 239000001963 growth medium Substances 0.000 description 21
- 239000003550 marker Substances 0.000 description 21
- 210000000981 epithelium Anatomy 0.000 description 16
- 210000004263 induced pluripotent stem cell Anatomy 0.000 description 16
- 210000001982 neural crest cell Anatomy 0.000 description 16
- 210000001519 tissue Anatomy 0.000 description 14
- 230000009786 epithelial differentiation Effects 0.000 description 12
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 9
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 8
- 235000002639 sodium chloride Nutrition 0.000 description 8
- 102000000905 Cadherin Human genes 0.000 description 7
- 108050007957 Cadherin Proteins 0.000 description 7
- 102100023967 Keratin, type I cytoskeletal 12 Human genes 0.000 description 7
- 102100040445 Keratin, type I cytoskeletal 14 Human genes 0.000 description 7
- 108010065086 Keratin-12 Proteins 0.000 description 7
- 238000002955 isolation Methods 0.000 description 7
- 238000002054 transplantation Methods 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- 241001465754 Metazoa Species 0.000 description 6
- -1 NANOG Proteins 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 238000011161 development Methods 0.000 description 6
- 230000018109 developmental process Effects 0.000 description 6
- 238000012744 immunostaining Methods 0.000 description 6
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 230000002207 retinal effect Effects 0.000 description 6
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 description 5
- 101001015006 Homo sapiens Integrin beta-4 Proteins 0.000 description 5
- 101000595669 Homo sapiens Pituitary homeobox 2 Proteins 0.000 description 5
- 102100033000 Integrin beta-4 Human genes 0.000 description 5
- 241000283973 Oryctolagus cuniculus Species 0.000 description 5
- 108010032788 PAX6 Transcription Factor Proteins 0.000 description 5
- 102100037506 Paired box protein Pax-6 Human genes 0.000 description 5
- 102100036090 Pituitary homeobox 2 Human genes 0.000 description 5
- 102100033237 Pro-epidermal growth factor Human genes 0.000 description 5
- 101100247004 Rattus norvegicus Qsox1 gene Proteins 0.000 description 5
- 102000004338 Transferrin Human genes 0.000 description 5
- 108090000901 Transferrin Proteins 0.000 description 5
- 230000004888 barrier function Effects 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 238000012423 maintenance Methods 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 150000003839 salts Chemical class 0.000 description 5
- 210000001082 somatic cell Anatomy 0.000 description 5
- 239000012581 transferrin Substances 0.000 description 5
- 102000009027 Albumins Human genes 0.000 description 4
- 108010088751 Albumins Proteins 0.000 description 4
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 4
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 4
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 4
- 101001094700 Homo sapiens POU domain, class 5, transcription factor 1 Proteins 0.000 description 4
- 101000713275 Homo sapiens Solute carrier family 22 member 3 Proteins 0.000 description 4
- 101000664703 Homo sapiens Transcription factor SOX-10 Proteins 0.000 description 4
- 101000713575 Homo sapiens Tubulin beta-3 chain Proteins 0.000 description 4
- 108010066321 Keratin-14 Proteins 0.000 description 4
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 4
- 102100038895 Myc proto-oncogene protein Human genes 0.000 description 4
- 101710135898 Myc proto-oncogene protein Proteins 0.000 description 4
- 102100035423 POU domain, class 5, transcription factor 1 Human genes 0.000 description 4
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 4
- 102100038808 Transcription factor SOX-10 Human genes 0.000 description 4
- 101710150448 Transcriptional regulator Myc Proteins 0.000 description 4
- 102100036790 Tubulin beta-3 chain Human genes 0.000 description 4
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 4
- 210000004087 cornea Anatomy 0.000 description 4
- 210000003683 corneal stroma Anatomy 0.000 description 4
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- 235000019441 ethanol Nutrition 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- 150000004665 fatty acids Chemical class 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 230000001537 neural effect Effects 0.000 description 4
- 235000015097 nutrients Nutrition 0.000 description 4
- 239000000049 pigment Substances 0.000 description 4
- LWIHDJKSTIGBAC-UHFFFAOYSA-K potassium phosphate Substances [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 4
- 229910052711 selenium Inorganic materials 0.000 description 4
- 239000011669 selenium Substances 0.000 description 4
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 4
- 238000010186 staining Methods 0.000 description 4
- 210000002341 stratified epithelial cell Anatomy 0.000 description 4
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 4
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 3
- 102000004392 Aquaporin 5 Human genes 0.000 description 3
- 108090000976 Aquaporin 5 Proteins 0.000 description 3
- 239000012583 B-27 Supplement Substances 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 3
- 101001052004 Escherichia phage T5 L-shaped tail fiber protein pb1 Proteins 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 102100021084 Forkhead box protein C1 Human genes 0.000 description 3
- 101000818310 Homo sapiens Forkhead box protein C1 Proteins 0.000 description 3
- 101001139134 Homo sapiens Krueppel-like factor 4 Proteins 0.000 description 3
- 101000798114 Homo sapiens Lactotransferrin Proteins 0.000 description 3
- 101000972282 Homo sapiens Mucin-5AC Proteins 0.000 description 3
- 101000972273 Homo sapiens Mucin-7 Proteins 0.000 description 3
- 101000687905 Homo sapiens Transcription factor SOX-2 Proteins 0.000 description 3
- 102000004877 Insulin Human genes 0.000 description 3
- 108090001061 Insulin Proteins 0.000 description 3
- 102100020677 Krueppel-like factor 4 Human genes 0.000 description 3
- 108700021430 Kruppel-Like Factor 4 Proteins 0.000 description 3
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 3
- 102100032241 Lactotransferrin Human genes 0.000 description 3
- 102100022496 Mucin-5AC Human genes 0.000 description 3
- 102100022492 Mucin-7 Human genes 0.000 description 3
- 101150086694 SLC22A3 gene Proteins 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- 102100024270 Transcription factor SOX-2 Human genes 0.000 description 3
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 238000004113 cell culture Methods 0.000 description 3
- 210000001900 endoderm Anatomy 0.000 description 3
- 210000002615 epidermis Anatomy 0.000 description 3
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 3
- 230000000762 glandular Effects 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 238000001727 in vivo Methods 0.000 description 3
- 239000000411 inducer Substances 0.000 description 3
- 229940125396 insulin Drugs 0.000 description 3
- 239000008101 lactose Substances 0.000 description 3
- 108010082117 matrigel Proteins 0.000 description 3
- 230000035800 maturation Effects 0.000 description 3
- 210000003716 mesoderm Anatomy 0.000 description 3
- 210000005036 nerve Anatomy 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 230000035755 proliferation Effects 0.000 description 3
- 108090000623 proteins and genes Proteins 0.000 description 3
- 230000001172 regenerating effect Effects 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 238000003757 reverse transcription PCR Methods 0.000 description 3
- 210000003079 salivary gland Anatomy 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 101150077014 sox10 gene Proteins 0.000 description 3
- 101150055666 sox6 gene Proteins 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 239000013589 supplement Substances 0.000 description 3
- 229930003231 vitamin Natural products 0.000 description 3
- 235000013343 vitamin Nutrition 0.000 description 3
- 239000011782 vitamin Substances 0.000 description 3
- 229940088594 vitamin Drugs 0.000 description 3
- 239000011701 zinc Substances 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- AWDORCFLUJZUQS-ZDUSSCGKSA-N (S)-2-methyl-1-(4-methylisoquinoline-5-sulfonyl)-1,4-diazepane Chemical compound C[C@H]1CNCCCN1S(=O)(=O)C1=CC=CC2=CN=CC(C)=C12 AWDORCFLUJZUQS-ZDUSSCGKSA-N 0.000 description 2
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 2
- PAWQVTBBRAZDMG-UHFFFAOYSA-N 2-(3-bromo-2-fluorophenyl)acetic acid Chemical compound OC(=O)CC1=CC=CC(Br)=C1F PAWQVTBBRAZDMG-UHFFFAOYSA-N 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 229920001353 Dextrin Polymers 0.000 description 2
- 239000004375 Dextrin Substances 0.000 description 2
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 2
- 101001133056 Homo sapiens Mucin-1 Proteins 0.000 description 2
- 101000972286 Homo sapiens Mucin-4 Proteins 0.000 description 2
- 101000854931 Homo sapiens Visual system homeobox 2 Proteins 0.000 description 2
- 108010050345 Microphthalmia-Associated Transcription Factor Proteins 0.000 description 2
- 102100030157 Microphthalmia-associated transcription factor Human genes 0.000 description 2
- 102100034256 Mucin-1 Human genes 0.000 description 2
- 102100022693 Mucin-4 Human genes 0.000 description 2
- 108010063954 Mucins Proteins 0.000 description 2
- 102000015728 Mucins Human genes 0.000 description 2
- 229910019142 PO4 Inorganic materials 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 2
- 102100020676 Visual system homeobox 2 Human genes 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 235000019270 ammonium chloride Nutrition 0.000 description 2
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 2
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 2
- 235000011130 ammonium sulphate Nutrition 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 159000000007 calcium salts Chemical class 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 229910002091 carbon monoxide Inorganic materials 0.000 description 2
- 230000021164 cell adhesion Effects 0.000 description 2
- 239000006143 cell culture medium Substances 0.000 description 2
- 230000024245 cell differentiation Effects 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 235000019425 dextrin Nutrition 0.000 description 2
- ZPWVASYFFYYZEW-UHFFFAOYSA-L dipotassium hydrogen phosphate Chemical compound [K+].[K+].OP([O-])([O-])=O ZPWVASYFFYYZEW-UHFFFAOYSA-L 0.000 description 2
- 229910000396 dipotassium phosphate Inorganic materials 0.000 description 2
- 235000019797 dipotassium phosphate Nutrition 0.000 description 2
- 210000000871 endothelium corneal Anatomy 0.000 description 2
- 210000005081 epithelial layer Anatomy 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 230000004373 eye development Effects 0.000 description 2
- 239000003925 fat Substances 0.000 description 2
- 235000019197 fats Nutrition 0.000 description 2
- 239000011790 ferrous sulphate Substances 0.000 description 2
- 235000003891 ferrous sulphate Nutrition 0.000 description 2
- 210000004602 germ cell Anatomy 0.000 description 2
- 235000012907 honey Nutrition 0.000 description 2
- 229930195733 hydrocarbon Natural products 0.000 description 2
- 150000002430 hydrocarbons Chemical class 0.000 description 2
- 239000012535 impurity Substances 0.000 description 2
- 150000002505 iron Chemical class 0.000 description 2
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 2
- 229910000359 iron(II) sulfate Inorganic materials 0.000 description 2
- 239000000787 lecithin Substances 0.000 description 2
- 235000010445 lecithin Nutrition 0.000 description 2
- 229940067606 lecithin Drugs 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 159000000003 magnesium salts Chemical class 0.000 description 2
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 2
- 235000019341 magnesium sulphate Nutrition 0.000 description 2
- 150000002696 manganese Chemical class 0.000 description 2
- 229940099596 manganese sulfate Drugs 0.000 description 2
- 239000011702 manganese sulphate Substances 0.000 description 2
- 235000007079 manganese sulphate Nutrition 0.000 description 2
- SQQMAOCOWKFBNP-UHFFFAOYSA-L manganese(II) sulfate Chemical compound [Mn+2].[O-]S([O-])(=O)=O SQQMAOCOWKFBNP-UHFFFAOYSA-L 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 2
- 235000019796 monopotassium phosphate Nutrition 0.000 description 2
- GLDOVTGHNKAZLK-UHFFFAOYSA-N octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCO GLDOVTGHNKAZLK-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 230000002093 peripheral effect Effects 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 239000002504 physiological saline solution Substances 0.000 description 2
- 210000004694 pigment cell Anatomy 0.000 description 2
- 210000002826 placenta Anatomy 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 2
- 229920000053 polysorbate 80 Polymers 0.000 description 2
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 2
- GNSKLFRGEWLPPA-UHFFFAOYSA-M potassium dihydrogen phosphate Chemical compound [K+].OP(O)([O-])=O GNSKLFRGEWLPPA-UHFFFAOYSA-M 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 210000003491 skin Anatomy 0.000 description 2
- 239000011684 sodium molybdate Substances 0.000 description 2
- 235000015393 sodium molybdate Nutrition 0.000 description 2
- TVXXNOYZHKPKGW-UHFFFAOYSA-N sodium molybdate (anhydrous) Chemical compound [Na+].[Na+].[O-][Mo]([O-])(=O)=O TVXXNOYZHKPKGW-UHFFFAOYSA-N 0.000 description 2
- 235000010344 sodium nitrate Nutrition 0.000 description 2
- 239000004317 sodium nitrate Substances 0.000 description 2
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 description 2
- XMVONEAAOPAGAO-UHFFFAOYSA-N sodium tungstate Chemical compound [Na+].[Na+].[O-][W]([O-])(=O)=O XMVONEAAOPAGAO-UHFFFAOYSA-N 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 229960002920 sorbitol Drugs 0.000 description 2
- 230000007480 spreading Effects 0.000 description 2
- 238000003892 spreading Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 239000011573 trace mineral Substances 0.000 description 2
- 235000013619 trace mineral Nutrition 0.000 description 2
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- WNWVKZTYMQWFHE-UHFFFAOYSA-N 4-ethylmorpholine Chemical group [CH2]CN1CCOCC1 WNWVKZTYMQWFHE-UHFFFAOYSA-N 0.000 description 1
- UZOVYGYOLBIAJR-UHFFFAOYSA-N 4-isocyanato-4'-methyldiphenylmethane Chemical compound C1=CC(C)=CC=C1CC1=CC=C(N=C=O)C=C1 UZOVYGYOLBIAJR-UHFFFAOYSA-N 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 108010008629 CA-125 Antigen Proteins 0.000 description 1
- 239000002229 CNT20 Substances 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- 206010010996 Corneal degeneration Diseases 0.000 description 1
- 206010011039 Corneal perforation Diseases 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- 206010015548 Euthanasia Diseases 0.000 description 1
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 1
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 1
- 102100024785 Fibroblast growth factor 2 Human genes 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000994375 Homo sapiens Integrin alpha-4 Proteins 0.000 description 1
- 101000984042 Homo sapiens Protein lin-28 homolog A Proteins 0.000 description 1
- 101000711846 Homo sapiens Transcription factor SOX-9 Proteins 0.000 description 1
- 101000801254 Homo sapiens Tumor necrosis factor receptor superfamily member 16 Proteins 0.000 description 1
- 102100032818 Integrin alpha-4 Human genes 0.000 description 1
- 102000011782 Keratins Human genes 0.000 description 1
- 108010076876 Keratins Proteins 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- OLIIUAHHAZEXEX-UHFFFAOYSA-N N-(6-fluoro-1H-indazol-5-yl)-6-methyl-2-oxo-4-[4-(trifluoromethyl)phenyl]-3,4-dihydro-1H-pyridine-5-carboxamide Chemical compound C1C(=O)NC(C)=C(C(=O)NC=2C(=CC=3NN=CC=3C=2)F)C1C1=CC=C(C(F)(F)F)C=C1 OLIIUAHHAZEXEX-UHFFFAOYSA-N 0.000 description 1
- 239000012580 N-2 Supplement Substances 0.000 description 1
- 102000007999 Nuclear Proteins Human genes 0.000 description 1
- 108010089610 Nuclear Proteins Proteins 0.000 description 1
- 206010034277 Pemphigoid Diseases 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 102000001253 Protein Kinase Human genes 0.000 description 1
- 102100025460 Protein lin-28 homolog A Human genes 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 241000978776 Senegalia senegal Species 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 101100289792 Squirrel monkey polyomavirus large T gene Proteins 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 206010042033 Stevens-Johnson syndrome Diseases 0.000 description 1
- 231100000168 Stevens-Johnson syndrome Toxicity 0.000 description 1
- 108010008125 Tenascin Proteins 0.000 description 1
- 102100038126 Tenascin Human genes 0.000 description 1
- 206010043276 Teratoma Diseases 0.000 description 1
- 102100034204 Transcription factor SOX-9 Human genes 0.000 description 1
- 102100033725 Tumor necrosis factor receptor superfamily member 16 Human genes 0.000 description 1
- 206010064996 Ulcerative keratitis Diseases 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000000205 acacia gum Substances 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 235000010419 agar Nutrition 0.000 description 1
- 102000007362 alpha-Crystallins Human genes 0.000 description 1
- 108010007908 alpha-Crystallins Proteins 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 208000008303 aniridia Diseases 0.000 description 1
- 210000002159 anterior chamber Anatomy 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 230000005549 barrier dysfunction Effects 0.000 description 1
- 210000000270 basal cell Anatomy 0.000 description 1
- 239000011324 bead Substances 0.000 description 1
- KXDAEFPNCMNJSK-UHFFFAOYSA-N benzene carboxamide Natural products NC(=O)C1=CC=CC=C1 KXDAEFPNCMNJSK-UHFFFAOYSA-N 0.000 description 1
- 229920002988 biodegradable polymer Polymers 0.000 description 1
- 239000004621 biodegradable polymer Substances 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- GEHJBWKLJVFKPS-UHFFFAOYSA-N bromochloroacetic acid Chemical compound OC(=O)C(Cl)Br GEHJBWKLJVFKPS-UHFFFAOYSA-N 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 230000036952 cancer formation Effects 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 239000002771 cell marker Substances 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 102000006533 chordin Human genes 0.000 description 1
- 108010008846 chordin Proteins 0.000 description 1
- 230000005757 colony formation Effects 0.000 description 1
- 230000004453 corneal transparency Effects 0.000 description 1
- 201000007717 corneal ulcer Diseases 0.000 description 1
- 238000012136 culture method Methods 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000007423 decrease Effects 0.000 description 1
- 229940105990 diglycerin Drugs 0.000 description 1
- GPLRAVKSCUXZTP-UHFFFAOYSA-N diglycerol Chemical compound OCC(O)COCC(O)CO GPLRAVKSCUXZTP-UHFFFAOYSA-N 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 210000001705 ectoderm cell Anatomy 0.000 description 1
- 210000001671 embryonic stem cell Anatomy 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 230000007159 enucleation Effects 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 210000001339 epidermal cell Anatomy 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 235000020776 essential amino acid Nutrition 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 210000002744 extracellular matrix Anatomy 0.000 description 1
- NGOGFTYYXHNFQH-UHFFFAOYSA-N fasudil Chemical compound C=1C=CC2=CN=CC=C2C=1S(=O)(=O)N1CCCNCC1 NGOGFTYYXHNFQH-UHFFFAOYSA-N 0.000 description 1
- 230000003325 follicular Effects 0.000 description 1
- 210000001654 germ layer Anatomy 0.000 description 1
- 210000004907 gland Anatomy 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 210000003780 hair follicle Anatomy 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 239000003276 histone deacetylase inhibitor Substances 0.000 description 1
- 238000002991 immunohistochemical analysis Methods 0.000 description 1
- 201000001371 inclusion conjunctivitis Diseases 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 239000000644 isotonic solution Substances 0.000 description 1
- 206010023332 keratitis Diseases 0.000 description 1
- 201000010666 keratoconjunctivitis Diseases 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 238000013532 laser treatment Methods 0.000 description 1
- 210000003644 lens cell Anatomy 0.000 description 1
- 210000001232 limbus corneae Anatomy 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 239000012092 media component Substances 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 210000002901 mesenchymal stem cell Anatomy 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- PJUIMOJAAPLTRJ-UHFFFAOYSA-N monothioglycerol Chemical compound OCC(O)CS PJUIMOJAAPLTRJ-UHFFFAOYSA-N 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- YOVNFNXUCOWYSG-UHFFFAOYSA-N n-[3-[2-(4-amino-1,2,5-oxadiazol-3-yl)-1-ethylimidazo[4,5-c]pyridin-6-yl]oxyphenyl]-4-(2-morpholin-4-ylethoxy)benzamide Chemical compound C1=C2N(CC)C(C=3C(=NON=3)N)=NC2=CN=C1OC(C=1)=CC=CC=1NC(=O)C(C=C1)=CC=C1OCCN1CCOCC1 YOVNFNXUCOWYSG-UHFFFAOYSA-N 0.000 description 1
- GOQYKNQRPGWPLP-UHFFFAOYSA-N n-heptadecyl alcohol Natural products CCCCCCCCCCCCCCCCCO GOQYKNQRPGWPLP-UHFFFAOYSA-N 0.000 description 1
- 210000005155 neural progenitor cell Anatomy 0.000 description 1
- 210000005157 neural retina Anatomy 0.000 description 1
- 210000001178 neural stem cell Anatomy 0.000 description 1
- 210000004498 neuroglial cell Anatomy 0.000 description 1
- 210000002569 neuron Anatomy 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 108700025694 p53 Genes Proteins 0.000 description 1
- 101710135378 pH 6 antigen Proteins 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 206010033675 panniculitis Diseases 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 229920001277 pectin Polymers 0.000 description 1
- 239000001814 pectin Substances 0.000 description 1
- 235000010987 pectin Nutrition 0.000 description 1
- 210000000578 peripheral nerve Anatomy 0.000 description 1
- 235000019271 petrolatum Nutrition 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920001495 poly(sodium acrylate) polymer Polymers 0.000 description 1
- 239000004626 polylactic acid Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 229940068968 polysorbate 80 Drugs 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 108060006633 protein kinase Proteins 0.000 description 1
- 230000002685 pulmonary effect Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 210000001525 retina Anatomy 0.000 description 1
- 210000003583 retinal pigment epithelium Anatomy 0.000 description 1
- 102000000568 rho-Associated Kinases Human genes 0.000 description 1
- 108010041788 rho-Associated Kinases Proteins 0.000 description 1
- 231100000241 scar Toxicity 0.000 description 1
- 239000000565 sealant Substances 0.000 description 1
- 210000000697 sensory organ Anatomy 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 210000004927 skin cell Anatomy 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- NNMHYFLPFNGQFZ-UHFFFAOYSA-M sodium polyacrylate Chemical compound [Na+].[O-]C(=O)C=C NNMHYFLPFNGQFZ-UHFFFAOYSA-M 0.000 description 1
- 229940054269 sodium pyruvate Drugs 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 239000004575 stone Substances 0.000 description 1
- 238000013517 stratification Methods 0.000 description 1
- 210000005127 stratified epithelium Anatomy 0.000 description 1
- 210000004304 subcutaneous tissue Anatomy 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 206010044325 trachoma Diseases 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 230000008733 trauma Effects 0.000 description 1
- 238000011282 treatment Methods 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical group C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 239000000230 xanthan gum Substances 0.000 description 1
- 235000010493 xanthan gum Nutrition 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 229940082509 xanthan gum Drugs 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0618—Cells of the nervous system
- C12N5/0621—Eye cells, e.g. cornea, iris pigmented cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/30—Nerves; Brain; Eyes; Corneal cells; Cerebrospinal fluid; Neuronal stem cells; Neuronal precursor cells; Glial cells; Oligodendrocytes; Schwann cells; Astroglia; Astrocytes; Choroid plexus; Spinal cord tissue
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/48—Reproductive organs
- A61K35/54—Ovaries; Ova; Ovules; Embryos; Foetal cells; Germ cells
- A61K35/545—Embryonic stem cells; Pluripotent stem cells; Induced pluripotent stem cells; Uncharacterised stem cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0607—Non-embryonic pluripotent stem cells, e.g. MASC
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/10—Cells modified by introduction of foreign genetic material
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/05—Inorganic components
- C12N2500/10—Metals; Metal chelators
- C12N2500/20—Transition metals
- C12N2500/24—Iron; Fe chelators; Transferrin
- C12N2500/25—Insulin-transferrin; Insulin-transferrin-selenium
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2500/00—Specific components of cell culture medium
- C12N2500/90—Serum-free medium, which may still contain naturally-sourced components
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/11—Epidermal growth factor [EGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/115—Basic fibroblast growth factor (bFGF, FGF-2)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/117—Keratinocyte growth factors (KGF-1, i.e. FGF-7; KGF-2, i.e. FGF-12)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/15—Transforming growth factor beta (TGF-β)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/155—Bone morphogenic proteins [BMP]; Osteogenins; Osteogenic factor; Bone inducing factor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/16—Activin; Inhibin; Mullerian inhibiting substance
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/70—Enzymes
- C12N2501/72—Transferases [EC 2.]
- C12N2501/727—Kinases (EC 2.7.)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/45—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from artificially induced pluripotent stem cells
Definitions
- the present invention relates to a method for inducing differentiation of corneal epithelial cells from pluripotent stem cells. More specifically, pluripotent stem cells (particularly, induced pluripotent stem cells) are autonomously differentiated into ectodermal cell types in serum-free medium without using feeder cells, and obtained ocular surface ectoderm lineage The present invention relates to a method for inducing differentiation of cells into corneal epithelial cells.
- ES cells embryonic stem cells
- artificial pluripotent stem cells having the same differentiation pluripotency as ES cells were established by introducing specific factors into somatic cells and undifferentiated stem cells.
- a typical example is iPS cells established by Yamanaka et al. Regenerative medicine using these induced pluripotent stem cells has no ethical problem and can also avoid the problem of rejection by using patient-derived cells as a source.
- epithelial cells can be induced to differentiate from human iPS cells or ES cells (Patent Document 1, Non-Patent Documents 1 and 2). Many of the induced epithelial cells express stratified epithelial cell markers such as keratin 14 and p63, and can be differentiated into stratified epithelial cells such as skin epithelium. The inventors have reported the differentiation of stratified epithelium into corneal epithelium (Patent Documents 2 to 3 and Non-patent Document 3), but cannot form a sheet, and cannot induce corneal epithelial progenitor cells with high proliferation ability. The use of feeder cells has been a problem in clinical application.
- a corneal epithelial sheet having a functional tissue structure layered from human iPS cells and marker expression can be prepared without using feeder cells. No such report has been made so far.
- An object of the present invention is to provide a method for inducing differentiation of pluripotent stem cells (particularly, induced pluripotent stem cells) into ocular surface epithelial series such as corneal epithelium and conjunctival epithelium without using feeder cells or serum, Another object is to provide a functional cultured corneal epithelial cell sheet layered from pluripotent stem cells (particularly, induced pluripotent stem cells).
- the inventors have intensively studied and the inventors have cultivated human iPS cells in a differentiation medium containing KSR or the like without using serum or feeder cells. It was found that colonies (multilayered) composed of concentric layers composed of different ectodermal cell types were formed. Furthermore, from the obtained colonies, the ocular surface ectoderm lineage cells including epithelial stem cells are separated, corneal epithelial progenitor cells are induced to differentiate, and further cultivated to obtain stratified corneal epithelial cells. I found it. The obtained corneal epithelial cells can be collected in a sheet form and transplanted, and it was confirmed to function as a corneal epithelium in vitro and in vivo as well as somatic corneal epithelial cells.
- the present invention has been completed based on the above findings and relates to the following (1) to (15).
- a method for producing a colony comprising: (2) The method according to (1) above, wherein concentric layers composed of different ectodermal cell types are formed by autonomous differentiation of pluripotent stem cells; (3) The method according to (1) or (2) above, wherein the serum-free medium does not contain 0.5 nM or more of BMP4 (Bone morphogenetic protein 4), transforming growth factor, and activin; (4) The method according to (3) above, wherein the serum-free medium does not contain at least one selected from a high concentration retinoic acid, a BMP inhibitor, a TGF ⁇ inhibitor, and Noggin; (5) Using a container in which pluripotent stem cells are coated with at least one selected from collagen, fibronectin, laminin or laminin fragment, vitronectin, basement membrane matrix, gelatin, hyaluronic acid, polylysine, vitronectin, and hyaluronic acid The method according to any one of (1) to (4) above, wherein the method is cultured.
- BMP4
- the colonies each comprise a layer composed of a neuroectodermal lineage cell, a neural crest lineage cell / ocular embryo lineage cell, an ocular surface ectoderm lineage cell, and a surface ectoderm lineage cell.
- Eye-related cells are corneal epithelial cells, retinal pigment epithelial cells, neural retinal cells, conjunctival epithelial cells, limbal epithelial cells, corneal endothelial cells, corneal parenchymal cells, iris parenchymal cells, scleral cells, iris pigmented epithelial cells (7) any one selected from ciliary epithelial cells, optic nerve cells, sublimbal fibroblasts, subconjunctival fibroblasts, lacrimal glands, meibomian glands, goblet cells, lens epithelial cells, and eyelid epithelial cells The method described in;
- epithelial cells obtained by the method of the present invention express these markers, are transplantable, and function as corneal epithelial cells in vitro and in vivo in the same manner as corneal epithelial cells derived from somatic cells. Further, since the method of the present invention does not use animal-derived feeder cells or serum, the resulting cells are highly safe and suitable for clinical application.
- colonies consisting of concentric layers composed of different ectodermal cell types obtained by the method of the present invention not only corneal epithelial cells but also conjunctival epithelial cells, retinal cells, lacrimal gland cells and other eyes. Various cells constituting the cell can also be produced.
- FIG. 1 shows a scheme of a method for inducing differentiation of human iPS cells into ocular surface epithelial / corneal epithelial cells.
- FIG. 2 shows the process of inducing differentiation of human iPS cells into ocular surface epithelial / corneal epithelial cells (A: from upper left, day 0, day 5, day 10, day 15, day 20, day 25, B: 6 weeks) ).
- FIG. 1 shows a scheme of a method for inducing differentiation of human iPS cells into ocular surface epithelial / corneal epithelial cells.
- FIG. 2 shows the process of inducing differentiation of human iPS cells into ocular surface epithelial / corneal epithelial cells (A: from upper left, day 0, day 5, day 10, day 15, day 20, day 25, B: 6 weeks) ).
- FIG. 1 shows a scheme of a method for inducing differentiation of human iPS cells into ocular surface epithelial / corneal epithelial cells.
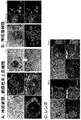
- FIG. 3 shows the characteristics of cells contained in each layer of ectodermal cells autonomously induced to differentiate from human iPS cells (A: expression of pax6 and p63 in layers 1-3, B: layers 2-4) Pax6, p63 expression, C: 1-3 layer E-cadherin, p63 expression, D: 1-3 layer ⁇ III-tublin, p63 expression). From these results, ocular surface epithelial cells co-positive for p63 and pax6 are observed only in the third layer.
- FIG. 4 shows the characteristics of cells contained in each layer of ectodermal cells autonomously differentiated from human iPS cells (A: CHX10-positive neuroretinal cells in the inner two layers and MITF positive in the outer layers.
- B p75 and SOX10 co-positive neural crest cells appear in the two-layer planned area at around the second week of differentiation
- C lens between 2-3 layers at the fourth week of differentiation Cells appear and are widely dispersed in the 2-3 layer through the differentiation induction process.
- FIG. 5 shows marker expression in each layer of ectoderm cells autonomously differentiated from human iPS cells
- first layer (1st) neuroectodermal lineage (Sox2 +, TUBB3 +, Sox6 +)
- second layer ( 2nd) neural crest / follicular cell lineage (pax6 +, Sox10 +, Rx +)
- 3rd layer (3rd) ocular surface ectoderm lineage (pax6 +, p63 +, E-cadherin +, K18)
- FIG. 6 shows corneal epithelium-related marker expression during differentiation induction (1-12 weeks).
- FIG. 7 shows expression of corneal epithelium-related markers in mouse eye development (E9.5-18.5) (PCE: corneal epithelial planned region, OSEpi: eye surface epithelium, CE: corneal epithelium, CS: corneal parenchyma, LV: lens follicle) , LE: lens, AC: anterior chamber, EL: eyelid, OV: eye follicle).
- FIG. 8 shows the isolation of corneal epithelial progenitor cells by FACS at 12 weeks of differentiation culture of the third layer fraction cells (P1: first fraction, P2: second fraction, P3: third fraction, P4: Fourth fraction).
- FIG. 9 shows the expression of corneal epithelial markers in the second fraction (P2) and the third fraction (P3).
- FIG. 10 shows the results of marker expression of the characteristics of corneal epithelial cells differentiated from human iPS cells.
- Corneal epithelial cells are observed in the third fraction (P3), and conjunctival epithelium and other pax6-negative stratified epithelial cells are observed in the second fraction (P2).
- FIG. 11 shows marker expression after mature culture of corneal epithelial cells (derived from the third fraction (P3)) induced to differentiate from human iPS cells.
- A Human iPS cell-derived corneal epithelial cells show a paving stone-like morphology and are stratified into 3-4 layers.
- B Cells after mature culture express markers ZO-1, MUC1, MUC4, and MUC16 essential for corneal barrier function.
- FIG. 12 shows the result of transplanting a human iPS cell-derived corneal epithelial cell sheet into a rabbit eye.
- the human iPS cell-derived corneal epithelial cell sheet can be collected into a sheet (left). From the observation of slits after transplantation), fluorescein staining (upper right) and immunohistochemical analysis (lower right), human iPS cell-derived corneal epithelial cell sheets engraft on the corneal stroma after transplantation and exert corneal barrier function I understand that FIG.
- FIG. 13 shows conjunctival goblet cell-like cells and lacrimal gland-like cells that have emerged during long-term culture in corneal epithelial culture medium.
- A PAS-positive and PAX6-positive cell populations express MUC5AC and K7, which are conjunctival embryo (goblet) cell markers.
- B Gland tissue obtained by three-dimensionally culturing cell aggregates exhibiting gland-like structures in Matrigel expresses AQP5, LTF, and MUC7, which are markers for lacrimal and salivary glands.
- FIG. 14 shows the induction of periocular neural crest cells.
- FIG. 15 shows isolation using a negative marker (CD200) of corneal epithelial progenitor cells derived from iPS cells.
- CD200 negative cells cells isolated as SSEA4-positive and ITG ⁇ 4-positive cells (P3 fraction) were layered and expressed K12, p63, PAX6, and showed the characteristics of differentiated corneal epithelial cells.
- the “pluripotent stem cell” includes all cells having differentiation pluripotency that can be differentiated into all cells other than the placenta, in addition to ES cells and ES cell lines, It includes both induced pluripotent stem cells such as iPS cells.
- “Artificial pluripotent stem cells” are reprogrammed (initialized) to have the same pluripotency as ES cells by introducing specific factors into mammalian somatic cells or undifferentiated stem cells. Say cell.
- “Artificial pluripotent stem cells” were first established by Yamanaka et al. By introducing four factors Oct3 / 4, Sox2, Klf4, and c-Myc into mouse fibroblasts. “IPS cells (Induced Pluripotent Stem Cell)” (Takahashi K, Yamanaka S., Cell, (2006) 126: 663-676). Subsequently, human iPS was also established by introducing the same four factors into human fibroblasts (Takahashi K, Yamanaka S., et al. Cell, (2007) 131: 861-872 .; Okita, K., Ichisaka, T., and Yamanaka, S. (2007).
- Sakurada et al. Are not somatic cells, but induced artificial multiplicity induced more efficiently by introducing Oct3 / 4, Sox2, Klf4, c-Myc, etc. using undifferentiated stem cells present in postnatal tissues as cell sources. Reportable stem cells (JP 2008-307007).
- artificial pluripotent stem cells (Shi Y., Ding S., et al., Cell Stem Cell, (2008) produced by introducing OCT3 / 4, KLF4, low molecular weight compounds into mouse neural progenitor cells, etc. Vol3, Issue 5,568-574), induced pluripotent stem cells (Kim JB., Scholer HR.) Produced by introducing OCT3 / 4, KLF4 into mouse neural stem cells endogenously expressing SOX2, C-MYC.
- JP2008-307007 JP2008-283972
- US2008-2336610 US2009-047263
- WO2007-069666 WO2008-118220
- WO2008-124133 JP2008-151058, WO2009-006930, WO2009-006997, WO2009-007852 and the like.
- “Artificial pluripotent stem cell” used in the present invention is a known artificial pluripotent stem cell or an equivalent induced artificial pluripotent stem cell as long as it satisfies the definition described at the beginning and does not impair the purpose of the present invention. Including all, cell sources, introduction factors, introduction methods, etc. are not particularly limited.
- the cell source is derived from humans (human induced pluripotent stem cells), more preferably, epithelial progenitor cells / stem cells derived from the cells or epithelial cells including corneal epithelium, epidermal cells Derived from the patient in need of treatment.
- humans human induced pluripotent stem cells
- epithelial progenitor cells / stem cells derived from the cells or epithelial cells including corneal epithelium, epidermal cells Derived from the patient in need of treatment.
- Ectodermal Lineage Cell Type A human embryo forms three germ layers at the stage of development, that is, endoderm, mesoderm, and ectoderm. That is, it forms endoderm, mesoderm, and ectoderm.
- the endoderm is the mucosal epithelium, liver, pancreas, etc. of the stomach and small intestine
- the mesoderm is muscle, bone, blood vessels, blood, subcutaneous tissue, heart, kidney, etc.
- the ectoderm is nerve, eye, epidermis, etc.
- the “ectodermal lineage cell type” means a cell lineage type derived from ectoderm, that is, a cell that will form the central nervous system / sensory organ, epidermis, and eyes in the future.
- ectodermal lineage cell types include neuroectodermal lineage cells, neural crest lineage cells / eye embryo lineage cells, ocular surface ectoderm lineage cells, and surface ectoderm lineage cells.
- Neuron ectoderm lineage cells are cells that will differentiate into nerve-related cells in the future, and are characterized as Sox2 +, TUBB3 +, Sox6 + cells.
- Neuronal crest lineage cells are cells that differentiate into neural crest-related cells such as peripheral nerve cells, glial cells, pigment cells, corneal endothelial cells and corneal parenchymal cells, and are characterized as sox10 positive and pax6 negative cells.
- Eye cup series cells are cells that differentiate into eye cup related cells such as the retina, retinal pigment epithelium, and iris pigment epithelium, and are characterized as Rx + cells.
- Opthelial surface ectoderm lineage cells are cells that differentiate into ocular surface cells such as corneal epithelium and conjunctival epithelium, and are characterized as pax6 +, p63 +, E-cadherin + cells.
- Eye-related cells are cells that form eyes derived from ectoderm, and are corneal epithelial cells, retinal pigment epithelial cells, neuroretinal cells, conjunctival epithelial cells, limbal epithelium.
- Cells, corneal endothelial cells, corneal parenchymal cells, iris parenchymal cells, scleral cells, iris pigmented epithelial cells, ciliary epithelial cells, optic nerve cells, sublimbal fibroblasts, subconjunctival fibroblasts, lacrimal gland, meibomian gland, goblet Examples include cells, lens epithelial cells, and eyelid epithelial cells.
- Corneal epithelial cells and corneal epithelial progenitor cells From the surface, the cornea has a three-layer structure of a corneal epithelial layer, a corneal stroma layer, and a corneal endothelial layer.
- a “corneal epithelial cell” is a cell constituting the outermost layer of the cornea, and is composed of 4 to 5 corneal epithelial cell layers.
- “Corneal epithelial cells” are derived from epidermal ectoderm, but the corneal stroma and endothelium are derived from neural crests, and it is thought that individual stem cells exist.
- “corneal epithelial cells” are characterized by the expression of keratin 12, which is a corneal epithelial differentiation marker, in addition to pax6 and p63.
- the “corneal epithelial progenitor cell” is an undifferentiated corneal epithelial cell, characterized by the expression of pax6 and p63, and the differentiation marker keratin 12 (K12) is hardly expressed.
- a marker specific to each cell type is used in order to identify the differentiation-induced cell.
- typical ones will be described.
- Keratin 14 (Cytokeratin 14: K14): Keratin 14 is a representative marker of basal epithelial cells.
- P63 is a nuclear protein belonging to the p53 gene family, but is a typical marker of epithelial progenitor cells and stem cells, and its expression is observed in normal human epidermis and hair follicle basal cells.
- Keratin 12 (Cytokeratin 12: K12): Keratins 12 and 3 are typical specific differentiation markers for corneal epithelium.
- Pax6 (Paired homeobox-6): Pax6 is a transcriptional regulatory factor involved in the formation of the eye, and is a representative marker of corneal epithelium, lens epithelium and retinal cells.
- MUC16 (Mucin 16): MUC16 is a kind of membrane-bound mucin, which is selectively expressed in corneal epithelial cells, and has an important role in maintaining the mucin layer on the ocular surface and expressing the barrier function.
- pluripotent stem cells are autonomously differentiated to form colonies composed of concentric layers each composed of different ectodermal cell types. “Autonomous differentiation (autonomous differentiation)” means that a cell differentiates itself without being stimulated from the outside, such as a differentiation inducer or a differentiation induction promoter.
- serum-free medium means a medium that does not contain unconditioned or unpurified serum, and a medium that contains purified blood-derived components or animal tissue-derived components (for example, growth factors) is a serum-free medium. It corresponds to.
- DMEM medium As a basic medium of differentiation medium, DMEM medium, BME medium, ⁇ MEM medium, serum-free DMEM / F12 medium, BGJb medium, CMRL 1066 medium, Glasgow MEM medium, Improved MEM Zinc Option medium, IMDM medium, Medium 199 medium, Eagle Any medium that can be used for animal cell culture, such as MEM medium, Ham medium, RPMI 1640 medium, Fischer's medium, McCoy's medium, and Williams E medium, can be used, but KnockOut TM DMEM A medium for stem cells such as Medium 154 and StemPro (registered trademark) hESC SFM is preferred.
- the basic medium for maintenance a medium for pluripotent stem cells that does not contain animal or human-derived components is more preferable. Examples of such a medium include mTeSR TM 1 (Japan BD), StemFit (registered trademark), and the like. Commercially available media can also be used.
- the medium may contain “serum substitute”.
- Serum substitutes include, for example, albumin (eg, lipid-rich albumin), transferrin, fatty acid, collagen precursor, trace elements (eg, zinc, selenium), B-27 (registered trademark) supplement, N2 supplement, knockout sealant replacement (KSR: manufactured by Invitrogen), 2-mercaptoethanol, 3′thiolglycerol and the like.
- B-27 supplement the concentration in the medium is 0.01 to 10% by weight, preferably 0.5 to 4% by weight.
- nutrient sources include glycerol, glucose, fructose, sucrose, lactose, honey, starch, dextrin and other carbon sources, fatty acids, fats and oils, lecithin, alcohols and other hydrocarbons, ammonium sulfate, ammonium nitrate, ammonium chloride , Nitrogen sources such as urea and sodium nitrate, salt, potassium salt, phosphate, magnesium salt, calcium salt, iron salt, manganese salt and other inorganic salts, monopotassium phosphate, dipotassium phosphate, magnesium sulfate, sodium chloride , Ferrous sulfate, sodium molybdate, sodium tungstate and manganese sulfate, various vitamins, amino acids and the like.
- the medium does not need to contain differentiation inducers such as BMP4 (Bone morphogenetic protein 4), transforming growth factor, and activin. . That is, the culture medium is substantially free of one or more, preferably two or more, more preferably all of the differentiation inducers. In the case of BMP4, it may be included if it is less than 0.5 nM, but it is preferable that it is not included at all.
- differentiation inducers such as BMP4 (Bone morphogenetic protein 4), transforming growth factor, and activin.
- the medium does not need to contain a differentiation induction promoter such as high concentration retinoic acid, BMP inhibitor, TGF ⁇ inhibitor, and Noggin.
- the high concentration retinoic acid means 1 ⁇ M, particularly about 10 ⁇ M retinoic acid. That is, one or more, preferably two or more, more preferably not all of the differentiation induction promoter.
- the medium does not need to contain Wnt, Wnt signal activator, Chordin and the like.
- the pH of the medium obtained by blending the above components is in the range of 5.5 to 9.0, preferably 6.0 to 8.0, more preferably 6.5 to 7.5.
- pluripotent stem cells are two-dimensionally cultured without using feeder cells.
- the vessel is not particularly limited as long as it is used for cell culture, flask, flask for tissue culture, dish, petri dish, tissue culture dish, multi-dish, microplate, microwell plate, multiplate, multiwell Plates, microslides, chamber slides, petri dishes, tubes, trays, culture bags, and roller bottles can be used.
- the inner surface of the container is coated with at least one selected from collagen, fibronectin, laminin or laminin fragment, vitronectin, basement membrane matrix, gelatin, hyaluronic acid, polylysine, vitronectin, and hyaluronic acid to promote cell adhesion and spreading It is preferable that Among these, it is more preferable to use laminin fragments such as laminin, laminin E8 fragment and laminin 511E8 fragment.
- Culturing is carried out at 36 ° C. to 38 ° C., preferably 36.5 ° C. to 37.5 ° C. under conditions of 1% to 25% O 2 and 1% to 15% CO 2 .
- the culture period for autonomous differentiation is at most 1 week to 8 weeks, preferably 2 weeks to 6 weeks, more preferably 3 weeks to 5 weeks.
- the first layer (neural ectoderm lineage cells (Sox2 +, TUBB3 +, Sox6 +)), the second layer Layer (neural crest lineage cells / eye embryo lineage cells (pax6 +, Sox10 +, Rx +)), layer 3 (ocular surface ectoderm lineage cells (pax6 +, p63 +, E-cadherin +,)), and layer 4 (surface ectoderm) Lineage cells (p63 +, Ecadherin +)) are included.
- Colonies formed by autonomous differentiation of pluripotent stem cells consist of concentric layers composed of different ectodermal cell types.
- the first layer (neural ectoderm lineage) Cell The first layer (neural ectoderm lineage) Cell
- layer 2 neural crest line cell / eye embryo line cell
- layer 3 ocular surface ectoderm line cell
- layer 4 surface ectoderm line cell.
- Differentiated Since the cell types contained in each layer of the colony are different lineages, the cells contained in a specific layer are separated (isolated) and induced to obtain the target eye-related cells. be able to.
- the present invention produces a colony composed of concentric layers composed of different ectodermal cell types by the method described in the preceding paragraph, and separates cells contained in a specific layer from the colony, There is also provided a method for producing an eye-related cell, comprising inducing differentiation into an eye-related cell.
- conjunctival epithelial cells differentiation is performed from the ocular surface epithelial cell lineage of the third layer cells as in the case of corneal epithelium. After culturing in a corneal epithelial differentiation medium, conjunctival epithelial cells can be isolated as pax6 positive, K13 positive, and K12 negative cells among cells isolated as SSEA4 negative and ITGB4 positive cells using FACS.
- retinal pigment epithelial cells are cells derived from neuroectodermal cells, they can be observed as paving stone epithelial cells having pigment in the 1-2 layer during differentiation induction.
- the pigment cells can be visually isolated under a microscope, and the picked-up pigment epithelial colonies can be isolated and cultured in a medium such as DMEM containing 10% FBS on a gelatin-coated culture dish. is there.
- neural crest cells In the case of neural crest cells, they appear in the second layer in the early stage of differentiation induction (about 2 weeks), and after 2 weeks, the cells are collected and simply used by FACS using neural crest cell markers such as p75NTR and ITGA4. Can be separated. The obtained neural crest cells can be induced to differentiate into corneal endothelial cells. In addition, early neural crest cells are induced into periocular neural crest cells by continuing culture. Periocular neural crest cells can be confirmed by the expression of periocular neural crest markers such as PITX2 and FOXC1.
- corneal epithelial cell differentiation from pluripotent stem cells
- Epithelial progenitor cells that can differentiate into corneal epithelium are present in the second and third layers of the colony. Lineage cells). Therefore, corneal epithelial cells can be obtained by inducing differentiation of the third layer of ocular surface ectoderm lineage cells into corneal epithelial progenitor cells, isolating them and culturing them mature.
- concentric colonies are formed from pluripotent stem cells by autonomous differentiation, and then replaced with a differentiation medium for epithelium containing a growth factor and cultured.
- a differentiation medium for epithelium containing a growth factor and cultured.
- ocular surface epithelial stem cells characterized by pax6 +, p63 +, E-cadherin + appear in the third layer.
- the ocular surface epithelial stem cells are separated by pipetting or the like and further cultured in a culture medium for corneal epithelium to induce differentiation into corneal epithelial progenitor cells.
- corneal epithelial progenitor cells are isolated using FACS or the like and matured to obtain corneal epithelial cells.
- the culture is performed using a serum-free medium without using feeder cells.
- the culture medium and the culture conditions will be described.
- the epithelial differentiation medium contains growth factors such as KGF, a Rock inhibitor, bFGF, and a serum substitute in order to promote differentiation into epithelial stem cells.
- any medium (serum-free medium) that can be used for culturing epithelial cells such as DMEM medium, BME medium, and ⁇ MEM medium used for autonomous differentiation, can be used.
- stem cell culture media such as KnockOut TM DMEM, Medium 154, and StemPro (registered trademark) hESC SFM, epithelial cell culture media such as CNT20, Cnt50, CnT-PR, and KSFM, or a mixture of these are desirable.
- 50% serum-free medium for autonomous differentiation and 50% Cnt20 or CntPR were mixed and used.
- Corneal epithelial culture medium contains KGF, a Rock inhibitor, and a serum substitute (eg, B27-supplement etc.) to promote differentiation into corneal epithelial progenitor cells. be able to. Furthermore, FGF2, insulin, and transferrin may be included.
- the “ROCK inhibitor” means a substance that inhibits Rho kinase (ROCK: Rho-associated, coiled-coil containing protein kinase), for example, N- (4-pyridinyl) -4 ⁇ -[(R) -1-aminoethyl] cyclohexane-1 ⁇ -carboxamide (Y-27632), Fasudil (HA1077), (2S) -2-methyl-1-[(4-methyl-5-isoquinolinyl) sulfonyl] hexahydro-1H-1, 4-diazepine (H-1152), 4 ⁇ -[(1R) -1-aminoethyl] -N- (4-pyridyl) benzene-1 zencarboxamide (Wf-536), N- (1H-pyrrolo [2, 3-b] pyridin-4-yl) -4PER (R) -1-aminoethyl] cyclohexane-1lo
- the corneal epithelium can be stratified and matured using the corneal epithelial medium described above, but when cultured for about one week in the medium for mature culture containing serum, And maturation are further promoted.
- the mature culture medium preferably contains serum (if necessary), KGF, a Rock inhibitor, insulin, transferrin, and selenium.
- As the basic medium those similar to the corneal epithelial culture medium can be used.
- Any medium may contain a “serum substitute”.
- Serum substitutes include those described in the previous section, such as albumin (eg, lipid-rich albumin), transferrin, fatty acid, collagen precursor, trace elements (eg, zinc, selenium), B-27 (registered trademark) supplement N2 supplements and the like.
- albumin eg, lipid-rich albumin
- transferrin e.g, transferrin
- fatty acid eg, transferrin
- collagen precursor eg, collagen precursor
- trace elements eg, zinc, selenium
- B-27 registered trademark
- the concentration in the medium is 0.01 to 10% by weight, preferably 0.5 to 4% by weight.
- nutrient sources necessary for maintenance and growth of cells and components necessary for induction of differentiation may be appropriately added to the medium.
- nutrient sources include glycerol, glucose, fructose, sucrose, lactose, honey, starch, dextrin and other carbon sources, fatty acids, fats and oils, lecithin, alcohols and other hydrocarbons, ammonium sulfate, ammonium nitrate, ammonium chloride , Nitrogen sources such as urea and sodium nitrate, salt, potassium salt, phosphate, magnesium salt, calcium salt, iron salt, manganese salt and other inorganic salts, monopotassium phosphate, dipotassium phosphate, magnesium sulfate, sodium chloride , Ferrous sulfate, sodium molybdate, sodium tungstate and manganese sulfate, various vitamins, amino acids and the like.
- the vessel is not particularly limited as long as it is used for cell culture, flask, flask for tissue culture, dish, petri dish, tissue culture dish, multi-dish, microplate, microwell plate, multiplate, multiwell Plates, microslides, chamber slides, petri dishes, tubes, trays, culture bags, and roller bottles can be used.
- the inner surface of the container is coated with at least one selected from collagen, fibronectin, laminin or laminin fragment, vitronectin, basement membrane matrix, gelatin, hyaluronic acid, polylysine, vitronectin, and hyaluronic acid to promote cell adhesion and spreading It is preferable that Among these, it is more preferable to use laminin fragments such as laminin, laminin E8 fragment and laminin 511E8 fragment.
- Culturing is carried out at 36 ° C. to 38 ° C., preferably 36.5 ° C. to 37.5 ° C. under conditions of 1% to 25% O 2 and 1% to 15% CO 2 .
- the culture period is at least 1 week to 8 weeks, preferably 2 weeks to 6 weeks, more preferably 3 weeks to 5 weeks, in the epithelial differentiation medium for inducing differentiation of ocular surface epithelial stem cells.
- the culture in the culture medium for corneal epithelium for inducing differentiation of corneal epithelial progenitor cells is at least 1 week to 12 weeks, preferably 1 week to 8 weeks, more preferably 2 weeks to 6 weeks.
- the stratification / maturation culture in the culture medium for corneal epithelium after isolation of corneal epithelial progenitor cells is at least 4 days, preferably 1 week or more, more preferably 2 to 3 weeks.
- Corneal epithelial progenitor cells can be easily carried out using an antibody specific for each marker according to a conventional method. For example, isolation may be performed using magnetic beads labeled with an antibody, a column on which an antibody is immobilized, and separation using a cell sorter (FACS) using a fluorescently labeled antibody. A commercially available antibody may be used, or may be prepared according to a conventional method.
- FACS cell sorter
- corneal epithelial progenitor cells can be easily isolated by FACS or the like using ITG ⁇ 4 and / or SSEA4 expression as an index.
- negative selection with TRA-1-60 or CD200 is preferably performed before positive selection with ITG ⁇ 4 and SSEA4.
- SSEA4 and CD200 can be used for isolation of corneal epithelial progenitor cells.
- SSEA4 Stage-Specific Embryonic Antigen-4 is known to be expressed in teratomas, human embryonic germ cells (EG), human ES cells, mesenchymal stem cells, erythrocytes and the like.
- SSEA4 is widely used for evaluation by human ES cell differentiation monitoring because its expression on human ES cells decreases with differentiation.
- CD200 has not been known to be expressed in pluripotent stem cells or not expressed in corneal epithelium, and can remove impurities more widely than known TRA-1-60. An excellent marker.
- corneal epithelial progenitor cells When the corneal epithelial progenitor cells are continuously cultured in the culture medium for corneal epithelium, they can differentiate into conjunctival goblet cells and lacrimal gland cells.
- Conjunctival goblet cells can be confirmed by the expression of MUC5AC and K7, which are conjunctival germ cell markers.
- Lacrimal gland cells can be confirmed by the expression of AQP5, LTF, and MUC7, which are markers of lacrimal and salivary glands, and glandular tissue can be constructed by performing three-dimensional culture.
- corneal epithelial cells By culturing corneal epithelial progenitor cells maturely, transplantable stratified corneal epithelial cells can be obtained.
- the obtained corneal epithelial cells are positive for K12, pax6, and p63 and function as corneal epithelium in vitro and in vivo in the same manner as somatic corneal epithelial cells.
- the present invention also provides pluripotent stem cells, particularly functional corneal epithelial cells derived from induced pluripotent stem cells, characterized by K12, pax6, and p63.
- the cell preparation of the present invention may contain scaffold materials and components for assisting cell maintenance / proliferation and administration to the affected area, and other pharmaceutically acceptable carriers.
- Components necessary for cell maintenance / proliferation include media components such as carbon sources, nitrogen sources, vitamins, minerals, salts, various cytokines, and extracellular matrix preparations such as Matrigel TM.
- scaffold materials and components that assist administration to the affected area include biodegradable polymers; for example, collagen, polylactic acid, hyaluronic acid, cellulose, and derivatives thereof, and a complex composed of two or more thereof, an aqueous solution for injection;
- aqueous solution for injection For example, physiological saline, medium, physiological buffer such as PBS, isotonic solution (eg D-sorbitol, D-mannose, D-mannitol, sodium chloride) containing glucose and other adjuvants, etc.
- Adjuvants such as alcohols, specifically ethanol, polyalcohols such as propylene glycol, polyethylene glycol, nonionic surfactants such as polysorbate 80, HCO-50, etc. may be used in combination.
- organic solvents polyvinyl alcohol, polyvinyl pyrrolidone, carboxyvinyl polymer, sodium carboxymethylcellulose, sodium polyacrylate, sodium alginate, water-soluble dextran, sodium carboxymethyl starch, pectin, methylcellulose as necessary , Ethyl cellulose, xanthan gum, gum arabic, casein, agar, polyethylene glycol, diglycerin, glycerin, propylene glycol, petrolatum, paraffin, stearyl alcohol, stearic acid, mannitol, sorbitol, lactose, surfactants acceptable as pharmaceutical additives, It may contain a buffer, an emulsifier, a suspension, a soothing agent, a stabilizer and the like.
- a purified antibody is dissolved in a solvent such as physiological saline, buffer solution, glucose solution, etc., and an adsorption inhibitor such as Tween 80, Tween 20, gelatin or the like is added thereto.
- a solvent such as physiological saline, buffer solution, glucose solution, etc.
- an adsorption inhibitor such as Tween 80, Tween 20, gelatin or the like is added thereto.
- Tween 80, Tween 20, gelatin or the like is added thereto.
- Diseases that can be the subject of the cell preparation of the present invention include, for example, Stevens-Johnson syndrome, pemphigoid, heat / chemical trauma, aniridia, Salzmann corneal degeneration, idiopathic keratoconjunctivitis, post-trachoma scar, Examples include corneal perforation, peripheral corneal ulcer, and corneal epithelial detachment after excimer laser treatment.
- the epithelial progenitor cell group obtained by the method of the present invention and / or the corneal epithelial cell group induced to differentiate from the cell group may be layered to produce a cultured corneal epithelial cell sheet. it can.
- the method of the present invention it is possible to easily obtain a layered sheet-like corneal epithelial cell using a serum-free medium without using feeder cells.
- the obtained cells retain markers specific to corneal epithelial cells such as keratin 12, pax6, and MUC16, and have the same functions as corneal epithelial cells derived from somatic cells.
- the present invention also provides a functional stratified corneal epithelial cell sheet derived from pluripotent stem cells, particularly induced pluripotent stem cells, characterized by being positive for K12, pax6, and MUC16, and a method for producing the same.
- Example 1 Induction of differentiation from human iPS cells into ocular surface epithelial / corneal epithelial cells Ocular surface epithelial / corneal epithelial cells were induced to differentiate from human iPS cells in a serum-free medium without using feeder cells. A scheme of the differentiation induction method is shown in FIG.
- Human iPS cells are 201B7 cell line (iPS cell line prepared by introducing Oct3 / 4, OX2, c-Myc, Klf4 into fibroblasts), and StemFit (Ajinomoto Co., Inc.) is used for laminin. on 6 well plate coated with 511E8 fragments, 200-1500cells / cm 2, 300-1000 cells / cm 2, cells were seeded at a seeding density of 450-700 cells / cm 2, and cultured 7-13 days. Replace the medium with differentiation medium (GMEM containing 10% KSR, 1 mM sodium pyruvate, 0.1 mM non-essential amino acids, 55 ⁇ m monothioglycerol), and then replace with fresh differentiation medium once every two or three days. Cultured for 4 weeks.
- differentiation medium GMEM containing 10% KSR, 1 mM sodium pyruvate, 0.1 mM non-essential amino acids, 55 ⁇ m monothioglycerol
- the medium was replaced with a differentiation medium for epithelium containing growth factors, and further cultured for 4 weeks. 2-3 weeks after replacement with epithelial differentiation medium (50% differentiation medium containing 20 ng / ml KGF, 5 ng / ml FGF2, 1032 ⁇ M % Y27632 and 50% Cnt20 (or CntPR; EGF / FGF2 free (CELLnTEC))
- epithelial differentiation medium 50% differentiation medium containing 20 ng / ml KGF, 5 ng / ml FGF2, 1032 ⁇ M % Y27632 and 50% Cnt20 (or CntPR; EGF / FGF2 free (CELLnTEC)
- the third layer of ocular surface epithelial cells was selectively cultured by removing non-epithelial cells by pipetting, and then replaced with corneal epithelial culture medium after 4 weeks of culture.
- corneal epithelial progenitor cells were isolated by FACS. The isolated corneal epithelial progenitor cells were further added to corneal epithelial culture medium (DMEM containing 20 ng / ml KGF, 10 ⁇ M Y27632, 2% B27-supplemen) Cultivated corneal epithelial cell sheet by culturing for 2-3 weeks at / F12 (2: 1)), and if necessary, after culturing in corneal epithelial culture medium for 1-2 weeks , Medium for maturation culture containing serum (DMEM / F1 containing 20 ng / ml KGF, 10 ⁇ M Y27632, insulin, transferrin, selenium and 5% FBS It was possible to produce a similar cultured corneal epithelial cell sheet by exchanging the cells for 2 weeks (3: 1)) and culturing for about one week.
- DMEM / F1 containing 20 ng / ml KGF, 10 ⁇ M Y27632, insulin,
- FIG. 3 The characteristics of the cells in each layer constituting the colony were examined (FIG. 3). Colonies differentiated into concentric circles in layers, and different lineages of cells existed in each layer. That is, layer 1-3 was positive for pax6, layer 3-4 was p63, E-cadherin positive, and TUBB3 was positive for layer 1-2.
- the CHX10-positive neural retina appeared in the inner layer of the second layer, and the MITF-positive retinal pigment epithelial cells appeared from the outer layer, and they could be isolated as cobblestone cells having the pigment by picking up and culturing.
- Neural crest cells appeared in the second layer as p75 and SOX10 co-positive cells in the early stage of differentiation (second week) (FIG. 4). It was confirmed that lens cells expressing ⁇ -crystallin appeared between the 2-3 layers (4 weeks, 6 weeks) (FIG. 4).
- Ocular surface epithelial ectoderm (pax6 +, p63 +, Ecad +), which is the origin of corneal epithelium, appeared from the third layer.
- this culture method induces human pluripotent stem cells, including ocular surface epithelial cells, to cells related to the eye (retinal, lens, neural crest cells, etc.) in a form that mimics the physiological development process. It was confirmed that is possible.
- Example 2 Induction of differentiation of corneal epithelial progenitor cells from human iPS cells.
- Concentric colonies formed by autonomous differentiation (4 weeks) of human iPS cells according to Example 1 contain growth factors after 4 weeks
- the epithelial differentiation medium was replaced, and ocular surface epithelial stem cells appearing in the third layer were isolated by removing non-epithelial cells by pipetting.
- the corneal epithelial culture medium was exchanged to induce differentiation into corneal epithelial progenitor cells.
- FACS FACS was performed using cell surface markers such as TRA-1-60, SSEA4, and ITGB4 (FIG. 8).
- Corneal epithelial progenitor cells can be isolated by TRA-1-60, SSEA4 and ITGB4 expression, and among TRA-1-60 negative cells, ITGB4 positive, SSEA4 positive (P3 fraction) and ITGB4 positive, SSEA4 It was confirmed that it was selectively recognized as negative (P2 fraction) (right in FIG. 8).
- the cells isolated from the P3 fraction were cultured in corneal epithelial culture medium for 2-3 weeks to obtain stratified corneal epithelial cells.
- the obtained cells expressed markers ZO-1, MUC1, MUC4 and MUC16 essential for the corneal barrier function (FIG. 11B).
- the K14 positive cell purity was about 99%
- the corneal epithelial differentiation marker K12 positive rate was about 60% (FIG. 11C).
- Example 3 Transplantation of Human iPS Cell-Derived Corneal Epithelial Cell Sheet into Rabbit Eyes
- the epithelial cell layer around the corneal limbus of the rabbit was removed by cornea excision to produce a rabbit corneal epithelial stem cell exhaustion model.
- the human iPS cell-derived corneal epithelial cell sheet collected in a sheet form was transplanted.
- the anti-human (H2B) antibody confirmed that the epithelial layer engrafted in the rabbit eye was derived from human iPS cells, and K12, pax6, p63, and MUC16 were It was expressed and it was confirmed that the properties of the corneal epithelium were retained (FIG. 12 right).
- Example 4 Induction of differentiation of conjunctival embryo cell-like cells and lacrimal gland-like cells from human iPS cells. Concentric colonies formed by autonomous differentiation (4 weeks) of human iPS cells according to Example 1 Thereafter, the epithelial differentiation medium containing epidermal growth factor was replaced, and ocular surface epithelial stem cells appearing in the third layer were isolated by removing non-epithelial cells by pipetting. Furthermore, after 8 weeks, the corneal epithelial culture medium was exchanged to induce differentiation into corneal epithelial progenitor cells.
- ocular surface epithelial stem / progenitor cells derived from human pluripotent stem cells are not only corneal epithelium but also conjunctival epithelial cells, conjunctival goblet cells and lacrimal gland cells that are thought to have the same origin as corneal epithelium It was confirmed that differentiation was possible.
- Example 5 Induction of differentiation of periocular neural crest cells from human iPS cells Human iPS cells were autonomously differentiated in a differentiation medium according to Example 1 (4 weeks) to differentiate concentric colonies (multilayer structures). Induced. At 3 weeks after differentiation induction, PITX2-positive and AP2b-positive cell clusters (periocular neural crest cells) were induced mainly in the second layer.
- the cell mass was also inducible when cultured in a medium supplemented with EGF, FGF2 (1-3 weeks), and low concentration (0.5 ⁇ M) retinoic acid (2-3 weeks) in differentiation medium.
- the expression of the marker gene was confirmed by RT-PCR on the 9th, 14th, 21st, 28th and 35th days from the start of differentiation culture. As a result, it was confirmed that after the initial neural crest marker SOX10 disappeared in 4 weeks, expression of periocular neural crest markers such as PITX2 and FOXC1 was induced. Thus, it was shown that early neural crest cells derived from human iPS cells are induced into periocular neural crest cells.
- the periocular neural crest that is the origin of development of neural crest cells, corneal endothelium and iris parenchymal cells Cells could be induced.
- the addition of EGF, FGF, or low concentrations (0.5 ⁇ M) of retinoic acid to the differentiation medium promotes the growth of retinal cells in the second layer, and the addition of a retinoic acid signal further increases the periocular nerve crest. It is thought that cell induction was promoted.
- the retinoic acid signal derived from retinal cells is thought to be important for the development of the periocular neural crest, and this culture system was thought to mimic the development.
- Example 6 Isolation of corneal epithelial progenitor cells derived from human iPS cells According to Example 1, human iPS cells were autonomously differentiated in a differentiation medium to induce differentiation of concentric colonies (multilayer structures). At 14 weeks from differentiation induction, FACS was performed using CD200, SSEA4, and ITG ⁇ 4 as cell surface markers.
- CD200-positive cells were excluded by gating, and SSEA4-positive and ITG ⁇ 4-positive cells (P3 fraction) among CD200-negative cells could be isolated as corneal epithelial progenitor cells.
- the isolated cells were stratified and expressed K12, p63, PAX6 and showed the characteristics of differentiated corneal epithelial cells (FIG. 15).
- corneal epithelial progenitor cells derived from iPS cells can be purified using CD200, SSEA4, ITG ⁇ 4.
- CD200 is not known to be expressed in pluripotent stem cells or corneal epithelium until now, and it can remove impurities more widely than the known TRA-1-60. It is considered an excellent marker.
- a functional and high-purity corneal epithelial cell that can be transplanted and layered is obtained. Since the method of the present invention does not use animal-derived feeder cells or serum, the resulting cells are highly safe and suitable for clinical application. Furthermore, according to the method of the present invention, not only corneal epithelium but also various cells constituting the ocular surface such as conjunctival epithelium can be prepared from pluripotent stem cells such as human iPS cells.
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Zoology (AREA)
- Biotechnology (AREA)
- Genetics & Genomics (AREA)
- Organic Chemistry (AREA)
- Wood Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Cell Biology (AREA)
- General Health & Medical Sciences (AREA)
- Developmental Biology & Embryology (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Ophthalmology & Optometry (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Epidemiology (AREA)
- Reproductive Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Virology (AREA)
- Immunology (AREA)
- Gynecology & Obstetrics (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Materials For Medical Uses (AREA)
Abstract
Description
本出願は,日本特許出願2015-006074(2015年1月15日出願)に基づく優先権を主張しており、この内容は本明細書に参照として取り込まれる。
本発明は多能性幹細胞からの角膜上皮細胞の分化誘導方法に関する。より詳細には、多能性幹細胞(とくに、人工多能性幹細胞)を、フィーダー細胞を用いることなく無血清培地で自律的に外胚葉系細胞種に分化させ、得られた眼表面外胚葉系列細胞を角膜上皮細胞に分化誘導する方法に関する。
(1)多能性幹細胞(とくに、人工多能性幹細胞)を、無血清培地でフィーダー細胞を用いることなく二次元培養することにより、各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを製造する方法;
(2)異なる外胚葉系細胞種で構成される同心円状の層が、多能性幹細胞の自律的分化によって形成されることを特徴とする、上記(1)に記載の方法;
(3)無血清培地が、0.5nM以上のBMP4(Bone morphogenetic protein 4)、トランスフォーミング増殖因子、及びアクチビンを含まないことを特徴とする、上記(1)又は(2)に記載の方法;
(4)無血清培地が、さらに高濃度レチノイン酸、BMP阻害剤、TGFβ阻害剤、及びNogginから選ばれる少なくとも1以上を含まないことを特徴とする、上記(3)に記載の方法;
(5)多能性幹細胞を、コラーゲン、フィブロネクチン、ラミニン又はラミニンフラグメント、ビトロネクチン、基底膜マトリックス、ゼラチン、ヒアルロン酸、ポリリジン、ビトロネクチン、及びヒアルロン酸から選ばれるいずれか1以上でコーティングされた容器を用いて培養することを特徴とする、上記(1)~(4)のいずれかに記載の方法;
(6)コロニーが、それぞれ神経外胚葉系列細胞、神経堤系列細胞/眼胚系列細胞、眼表面外胚葉系列細胞、及び表面外胚葉系列細胞で構成される層を含む、上記(1)~(5)のいずれかに記載の方法;
(7)上記(1)~(6)のいずれかに記載の方法で各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを製造する工程、
前記コロニーから特定の層に含まれる細胞を分離する工程、
前記細胞を眼関連細胞に分化誘導する工程、
を含む、眼関連細胞の製造方法;
(8)眼関連細胞が、角膜上皮細胞、網膜色素上皮細胞、神経網膜細胞、結膜上皮細胞、輪部上皮細胞、角膜内皮細胞、角膜実質細胞、虹彩実質細胞、強膜細胞、虹彩色素上皮細胞、毛様体上皮細胞、視神経細胞、輪部下繊維芽細胞、結膜下繊維芽細胞、涙腺、マイボーム腺、杯細胞、レンズ上皮細胞、及び眼瞼上皮細胞から選ばれるいずれかである、上記(7)に記載の方法;
(9)上記(1)~(6)のいずれかに記載の方法で各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを製造する工程、
成長因子を含む培地で前記コロニーを培養する工程、
眼表面外胚葉系列細胞で構成される層に含まれる細胞を分離し、KGF、Rock阻害剤、及び血清代替物を含む培地で培養して、角膜上皮前駆細胞に分化誘導する工程、
角膜上皮前駆細胞を単離し、角膜上皮細胞に分化誘導する工程、
を含む、角膜上皮細胞の製造方法;
(10)ITGβ4、SSEA4、TRA-1-60、及びCD200から選ばれる1又は2以上のマーカーの発現の有無、好ましくはITGβ4陽性及びSSEA4陽性、より好ましくはTRA-1-60又はCD200陰性で、かつITGβ4陽性及びSSEA4陽性、さらに好ましくはCD200陰性で、かつITGβ4陽性及びSSEA4陽性を指標として角膜上皮前駆細胞を単離することを特徴とする、上記(9)に記載の方法;
(11)上記(9)又は(10)に記載の方法で製造される角膜上皮細胞であって、K12、pax6、及びp63陽性であることを特徴とする角膜上皮細胞;
(12)上記(9)又は(10)に記載の方法で角膜上皮細胞を製造する工程、得られた角膜上皮細胞をさらに重層化培養する工程を含む、角膜細胞シートの製造方法;
(13)多能性幹細胞(とくに、人工多能性幹細胞)由来の各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニー;
(14)上記(1)~(6)のいずれか1項に記載の方法によって製造される、上記(13)記載のコロニー;
(15)多能性幹細胞(とくに、人工多能性幹細胞)由来の重層化角膜上皮細胞シートであって、角膜上皮細胞がK12、pax6、及びMUC16陽性であることを特徴とする重層化角膜上皮細胞シート;
(16)上記(13)又は(14)記載のコロニーの眼表面外胚葉系列細胞で構成される層に含まれる細胞を分離・培養して製造される多能性幹細胞(とくに、人工多能性幹細胞)由来の角膜上皮細胞であって、K12、pax6、及びp63陽性であることを特徴とする角膜上皮細胞。
さらに、本発明の方法で得られた異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーからは、角膜上皮細胞のみならず、結膜上皮細胞、網膜細胞、涙腺細胞などの眼を構成する様々な細胞も作製することができる。
以下、本発明で使用される用語のいくつかについて説明する。
(1)多能性幹細胞
本発明にかかる「多能性幹細胞」とは、胎盤以外のすべての細胞に分化可能な分化多能性を有する細胞すべてを含み、ES細胞やES細胞株のほか、iPS細胞等の人工多能性幹細胞の両方を含む。
ヒトの胚は、発生の段階で3つの胚葉、すなわち内胚葉、中胚葉、外胚葉を形成する。すなわち内胚葉、中胚葉、外胚葉を形成する。このうち内胚葉は、胃や小腸の粘膜上皮、肝臓、膵臓等になり、中胚葉は筋肉、骨、血管や血液、皮下組織、心臓、腎臓等になり、外胚葉は神経、目、表皮等を形成する。本発明にかかる「外胚葉系列細胞種」は、外胚葉に由来する細胞系列種、すなわち、将来的に中枢神経系・感覚器官、表皮、眼を形成する細胞を意味する。
本発明にかかる「眼関連細胞」とは、外胚葉に由来する眼を形成する細胞で、角膜上皮細胞、網膜色素上皮細胞、神経網膜細胞、結膜上皮細胞、輪部上皮細胞、角膜内皮細胞、角膜実質細胞、虹彩実質細胞、強膜細胞、虹彩色素上皮細胞、毛様体上皮細胞、視神経細胞、輪部下繊維芽細胞、結膜下繊維芽細胞、涙腺、マイボーム腺、杯細胞、レンズ上皮細胞、及び眼瞼上皮細胞等を挙げることができる。
角膜は、表面から、角膜上皮層、角膜実質層、角膜内皮層の3層構造をしている。「角膜上皮細胞」は、この角膜の一番外側の層を構成する細胞で、4~5層の角膜上皮細胞層から構成されている。「角膜上皮細胞」は表皮外胚葉に由来するが、角膜の実質と内皮は神経堤由来であり、それぞれ個別の幹細胞が存在すると考えられている。本発明において「角膜上皮細胞」は、pax6及びp63に加えて、角膜上皮分化マーカーであるケラチン12の発現によって特徴づけられる。
本発明では、分化誘導された細胞を同定するために、各細胞種に特異的なマーカーを利用する。以下、代表的なものについて説明する。
本発明においては、まず多能性幹細胞を自律的に分化させ、各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを形成させる。「自律的分化(自律的に分化)」とは、分化誘導剤や分化誘導促進剤等の外部から刺激を受けることなく、細胞が自ら分化することを意味する。
本発明では無血清培地を用いて培養を行う。「無血清培地」とは、無調整又は未精製の血清を含まない培地を意味し、精製された血液由来成分や動物組織由来成分(例えば、増殖因子)が混入している培地は無血清培地に該当する。
維持用の基本培地としては、動物・ヒト由来成分を含まない多能性幹細胞用の培地がより好ましく、そのような培地としては、mTeSRTM1(日本BD社)、StemFit(登録商標)等の市販の培地を使用することもできる。
本発明ではフィーダー細胞を用いることなく多能性幹細胞を二次元培養する。容器は、細胞培養に使用されるものであれば特に限定されず、フラスコ、組織培養用フラスコ、ディッシュ、ペトリデッシュ、組織培養用ディッシュ、マルチディッシュ、マイクロプレート、マイクロウエルプレート、マルチプレート、マルチウェルプレート、マイクロスライド、チャンバースライド、シャーレ、チューブ、トレイ、培養バック、及びローラーボトルを使用することができる。
自律的分化のための培養期間は、長くとも1週~8週間、好ましくは2週~6週間、より好ましくは3週間~5週間である。
無血清培地でフィーダー細胞を用いることなく二次元培養された多能性幹細胞は、自律的に分化して外胚葉系細胞種で構成される層状のコロニーを形成する。コロニーは、中心部から周辺部に向けて、各々異なる外胚葉系細胞種で構成される同心円状の層からなり、第1層(神経外胚葉系列細胞(Sox2+, TUBB3+, Sox6+))、第2層(神経堤系列細胞/眼胚系列細胞(pax6+, Sox10+, Rx+))、第3層(眼表面外胚葉系列細胞(pax6+, p63+, E-cadherin+,))、及び第4層(表面外胚葉系列細胞(p63+, Ecadherin+))を含む。
多能性幹細胞の自律的分化によって形成されたコロニーは、各々異なる外胚葉系細胞種で構成される同心円状の層からなり、第1層(神経外胚葉系列細胞)、第2層(神経堤系列細胞/眼胚系列細胞)、第3層(眼表面外胚葉系列細胞)、及び第4層(表面外胚葉系列細胞)で構成される層状の細胞群に区別される。コロニーの各層に含まれる細胞種は異なる系列であるため、これを利用して、特定の層に含まれる細胞を分離(単離)し、分化誘導することで、目的とする眼関連細胞を得ることができる。
角膜上皮に分化し得る上皮前駆細胞は、コロニーの第2層と第3層に存在し、とくに角膜上皮前駆細胞は第3層(眼表面外胚葉系列細胞)に存在する。よって、第3層の眼表面外胚葉系列細胞を角膜上皮前駆細胞に分化誘導し、これを単離し、成熟培養することで角膜上皮細胞を得ることができる。
この眼表面上皮幹細胞を、ピペッティング等により分離し、さらに角膜上皮培養用培地で培養し、角膜上皮前駆細胞に分化誘導する。次いで、角膜上皮前駆細胞をFACS等を用いて単離し、成熟培養して角膜上皮細胞を得る。
分化誘導のいずれの工程においても、培養は、無血清培地によりフィーダー細胞を用いることなく行う。以下、培地と培養条件について説明する。
(1-1)上皮用分化培地
上皮用分化培地は、上皮系幹細胞への分化を促すために、KGF、Rock阻害剤、bFGF、血清代替物等の成長因子を含む。基本培地は、自律的分化で使用した、DMEM培地、BME培地、α MEM培地など、上皮細胞の培養に用いることのできる培地(無血清培地)あればいずれも用いることができる。また、KnockOutTMDMEM、Medium 154、StemPro(登録商標)hESC SFM等の幹細胞用培地やCNT20、Cnt50、CnT-PR、KSFMなど上皮細胞培養培地、又はこれらを混合した培地が望ましい。後述する実施例では、50%の自律分化用無血清培地と50%のCnt20 あるいは CntPRを混合して使用した。
角膜上皮培養用培地は、角膜上皮前駆細胞への分化誘導を促すために、KGF、Rock阻害剤、及び血清代替物(例えば、B27-supplement等)を含むことができる。さらに、FGF2、インスリン、トランスフェリンを含んでいてもよい。なお、「ROCK阻害剤」とは、Rhoキナーゼ(ROCK: Rho-associated,coiled-coil containing protein kinase)を阻害する物質を意味し、例えば、N-(4-ピリジニル)-4β-[(R)-1-アミノエチル]シクロヘキサン-1α-カルボアミド(Y-27632)、Fasudil(HA1077)、(2S)-2-メチル-1-[(4-メチル-5-イソキノリニル)スルホニル]ヘキサヒドロ-1H-1,4-ジアゼピン(H-1152)、4β-[(1R)-1-アミノエチル]-N-(4-ピリジル)ベンゼン-1ゼンカルボアミド(Wf-536)、N-(1H-ピロロ[2,3-b]ピリジン-4-イル)-4PER(R)-1-アミノエチル]シクロヘキサン-1ロヘカルボアミド(Y-30141)、N-(3-{[2-(4-アミノ-1,2,5-オキサジアゾール-3-イル)-1-エチル-1H-イミダゾ[4, 5-c]ピリジン-6-イル]オキシ}フェニル)-4-{[2-(4-モルホリニル)エチル]-オキシ}ベンズアミド(GSK269962A)、N-(6-フルオロ-1H-インダゾール-5-イル)-6-メチル-2-オキソ-4-[4-(トリフルオロメチル)フェニル]-3,4-ジヒドロ-1H-ピリジン-5-カルボキサミド(GSK429286A)を利用できる。
基本培地としては、上皮用分化培地と同様のものを使用することができる。
角膜上皮は、上記した角膜上皮培地を用いて重層化・成熟化することができるが、最後1週間程度、血清を含む成熟培養用培地を用いて培養すると重層化、成熟化がより促進される。前記成熟培養用培地は、血清(必要に応じて)、KGF、Rock阻害剤、インスリン、トランスフェリン、セレニウムを含むことが好ましい。基本培地としては、角膜上皮培養培地と同様のものを使用することができる。
いずれの工程も、フィーダー細胞を用いることなく培養を行う。容器は、細胞培養に使用されるものであれば特に限定されず、フラスコ、組織培養用フラスコ、ディッシュ、ペトリデッシュ、組織培養用ディッシュ、マルチディッシュ、マイクロプレート、マイクロウエルプレート、マルチプレート、マルチウェルプレート、マイクロスライド、チャンバースライド、シャーレ、チューブ、トレイ、培養バック、及びローラーボトルを使用することができる。
角膜上皮前駆細胞を分化誘導するための、角膜上皮培養用培地での培養は、少なくとも1週~12週間、好ましくは1週~8週間、より好ましくは2週~6週間である。
角膜上皮前駆細胞を単離後の角膜上皮培養用培地における重層化・成熟培養には、少なくとも4日間、好ましくは1週間以上、より好ましくは2週間~3週間である。
角膜上皮前駆細胞は、常法にしたがい各マーカーに特異的な抗体を用いて容易に実施できる。たとえば、抗体で標識された磁気ビーズ、抗体を固相化したカラム、蛍光標識された抗体を用いたセルソーター(FACS)による分離を用いて単離すればよい。抗体は、市販のものを利用してもよいし、常法にしたがい作製してもよい。
角膜上皮前駆細胞を成熟培養することで、移植可能な、重層化した角膜上皮細胞が得られる。得られた角膜上皮細胞は、K12、pax6、及びp63陽性で、体細胞由来の角膜上皮細胞と同様にin vitro及びin vivoで角膜上皮として機能する。本発明は、このK12、pax6、及びp63で特徴づけられる、多能性幹細胞、とくに人工多能性幹細胞由来の機能的な角膜上皮細胞も提供する。
5.1 細胞製剤
本発明の方法によって得られた神経外胚葉系列細胞、神経堤系列細胞/眼胚系列細胞、眼表面外胚葉系列細胞、及び表面外胚葉系列細胞、あるいは、前記細胞から分化誘導された眼関連細胞は、それ自体、研究、再生医療あるいは後述する細胞製剤の原料として利用することができる。
本発明の方法で得られた上皮系前駆細胞群、及び/又は前記細胞群から分化誘導された角膜上皮細胞群を重層化して培養角膜上皮細胞シートを作製することができる。
ヒトiPS細胞より、無血清培地でフィーダー細胞を用いることなく眼表面上皮・角膜上皮細胞を分化誘導した。分化誘導方法のスキームを図1に示す。
培地を分化培地(10%KSR、1 mM sodium pyruvate、0.1 mM non-essential amino acids、55 μm Monothioglycerolを含むGMEM)に交換し、その後2、3日に1回新鮮な分化培地に交換し、約4週間培養した。
実施例1にしたがってヒトiPS細胞の自律的分化(4週間)によって形成された同心円状のコロニーを、4週目以降成長因子を含む上皮用分化培地に交換し、第3層に出現する眼表面上皮幹細胞をピペッティングにより非上皮性細胞を除去することにより単離した。さらに、8週目以降、角膜上皮培養培地に交換し、角膜上皮前駆細胞への分化誘導を行った。
家兎の角膜輪部全周の上皮細胞層を角膜切除術により除去し、家兎角膜上皮幹細胞疲弊症モデルを作製した。その後、シート状に回収したヒトiPS細胞由来角膜上皮細胞シートを移植した。
移植後7日目では、シート移植群においては角膜透明性が保たれており、フルオレセイン染色によりバリア機能が回復していることが示された。一方、コントロール眼では大部分がフルオレセインで染色され、バリア機能不全の状態のままであった(図12中)。
術後7日目にて安楽死させ、眼球摘出後10%中性緩衝ホルマリン液にて化学固定を行った。その後ヘマトキシリン&エオジン染色により角膜組織を観察した。ヒトiPS細胞由来角膜上皮細胞シートは動物眼へ移植後も、角膜実質面に生着していた。凍結切片を用いた免疫染色解析においては、抗ヒト(H2B)抗体により、家兎眼に生着している上皮層がヒトiPS細胞由来であることが確認され、K12、pax6、p63、MUC16を発現しており、角膜上皮の性質を保持していることが確認できた(図12右)。
実施例1にしたがってヒトiPS細胞の自律的分化(4週間)によって形成された同心円状のコロニーを、4週目以降成長因子を含む上皮用分化培地に交換し、第3層に出現する眼表面上皮幹細胞をピペッティングにより非上皮性細胞を除去することにより単離した。さらに、8週目以降、角膜上皮培養培地に交換し、角膜上皮前駆細胞への分化誘導を行った。
実施例1にしたがってヒトiPS細胞を分化培地で自律的に分化させ(4週間)、同心円状のコロニー(多層構造体)を分化誘導した。分化誘導から3週目において、PITX2陽性、AP2b陽性の細胞塊(眼周囲神経堤細胞)が2層目を中心に誘導された。
実施例1にしたがってヒトiPS細胞を分化培地で自律的に分化させ、同心円状のコロニー(多層構造体)を分化誘導した。分化誘導から14週目において、CD200、SSEA4、ITGβ4を細胞表面マーカーとしてFACSを行った。
Claims (16)
- 多能性幹細胞を、無血清培地でフィーダー細胞を用いることなく二次元培養することにより、各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを製造する方法。
- 異なる外胚葉系細胞種で構成される同心円状の層が、多能性幹細胞の自律的分化によって形成されることを特徴とする、請求項1に記載の方法。
- 無血清培地が、0.5nM以上のBMP4(Bone morphogenetic protein 4)、トランスフォーミング増殖因子、及びアクチビンを含まないことを特徴とする、請求項1又は2に記載の方法。
- 無血清培地が、さらに高濃度レチノイン酸、BMP阻害剤、TGFβ阻害剤、及びNogginから選ばれる少なくとも1以上を含まないことを特徴とする、請求項3に記載の方法。
- 多能性幹細胞を、コラーゲン、フィブロネクチン、ラミニン又はラミニンフラグメント、ビトロネクチン、基底膜マトリックス、ゼラチン、ヒアルロン酸、ポリリジン、ビトロネクチン、及びヒアルロン酸から選ばれるいずれか1以上でコーティングされた容器を用いて培養することを特徴とする、請求項1~4のいずれか1項に記載の方法。
- コロニーが、それぞれ神経外胚葉系列細胞、神経堤系列細胞/眼胚系列細胞、眼表面外胚葉系列細胞、及び表面外胚葉系列細胞で構成される層を含む、請求項1~5のいずれか1項に記載の方法。
- 請求項1~6のいずれか1項に記載の方法で各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを製造する工程、
前記コロニーから特定の層に含まれる細胞を分離する工程、
前記細胞を眼関連細胞に分化誘導する工程、
を含む、眼関連細胞の製造方法。 - 眼関連細胞が、角膜上皮細胞、網膜色素上皮細胞、神経網膜細胞、結膜上皮細胞、輪部上皮細胞、角膜内皮細胞、角膜実質細胞、虹彩実質細胞、強膜細胞、虹彩色素上皮細胞、毛様体上皮細胞、視神経細胞、輪部下繊維芽細胞、結膜下繊維芽細胞、涙腺、マイボーム腺、杯細胞、レンズ上皮細胞、及び眼瞼上皮細胞から選ばれるいずれかである、請求項7に記載の方法。
- 請求項1~6のいずれか1項に記載の方法で各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニーを製造する工程、
成長因子を含む培地で前記コロニーを培養する工程、
眼表面外胚葉系列細胞で構成される層に含まれる細胞を分離し、KGF、Rock阻害剤、及び血清代替物を含む培地で培養して、角膜上皮前駆細胞に分化誘導する工程、
角膜上皮前駆細胞を単離し、角膜上皮細胞に分化誘導する工程、
を含む、角膜上皮細胞の製造方法。 - ITGβ4、SSEA4、TRA-1-60、及びCD200から選ばれる1又は2以上のマーカーの発現の有無を指標として角膜上皮前駆細胞を単離することを特徴とする、請求項9に記載の方法。
- 請求項9又は10に記載の方法で製造される角膜上皮細胞であって、K12、pax6、及びp63陽性であることを特徴とする角膜上皮細胞。
- 請求項9又は10に記載の方法で角膜上皮細胞を製造する工程、得られた角膜上皮細胞をさらに重層化培養する工程を含む、角膜細胞シートの製造方法。
- 多能性幹細胞由来の各々異なる外胚葉系細胞種で構成される同心円状の層からなるコロニー。
- 請求項1~6のいずれか1項に記載の方法によって製造される、請求項13記載のコロニー。
- 多能性幹細胞由来の重層化角膜上皮細胞シートであって、角膜上皮細胞がK12、pax6、及びMUC16陽性であることを特徴とする重層化角膜上皮細胞シート。
- 請求項13又は14に記載のコロニーの眼表面外胚葉系列細胞で構成される層に含まれる細胞を分離・培養して製造される多能性幹細胞由来の角膜上皮細胞であって、K12、pax6、及びp63陽性であることを特徴とする角膜上皮細胞。
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2016207513A AU2016207513B2 (en) | 2015-01-15 | 2016-01-13 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
| KR1020177019320A KR102121647B1 (ko) | 2015-01-15 | 2016-01-13 | 다능성 줄기세포로부터의 각막 상피 세포의 분화 유도 방법 |
| CA2973187A CA2973187C (en) | 2015-01-15 | 2016-01-13 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
| CN201680006004.8A CN107208052A (zh) | 2015-01-15 | 2016-01-13 | 由多能干细胞向角膜上皮细胞的分化诱导 |
| JP2016569469A JP6465898B2 (ja) | 2015-01-15 | 2016-01-13 | 多能性幹細胞からの角膜上皮細胞の分化誘導 |
| SG11201705750WA SG11201705750WA (en) | 2015-01-15 | 2016-01-13 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
| US15/542,200 US11066641B2 (en) | 2015-01-15 | 2016-01-13 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
| EP16737356.2A EP3246394B1 (en) | 2015-01-15 | 2016-01-13 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
| US17/348,174 US20210371818A1 (en) | 2015-01-15 | 2021-06-15 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-006074 | 2015-01-15 | ||
| JP2015006074 | 2015-01-15 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/542,200 A-371-Of-International US11066641B2 (en) | 2015-01-15 | 2016-01-13 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
| US17/348,174 Continuation US20210371818A1 (en) | 2015-01-15 | 2021-06-15 | Method for inducing differentiation of corneal epithelial cells from pluripotent stem cells |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016114285A1 true WO2016114285A1 (ja) | 2016-07-21 |
Family
ID=56405827
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2016/050784 WO2016114285A1 (ja) | 2015-01-15 | 2016-01-13 | 多能性幹細胞からの角膜上皮細胞の分化誘導 |
Country Status (9)
| Country | Link |
|---|---|
| US (2) | US11066641B2 (ja) |
| EP (1) | EP3246394B1 (ja) |
| JP (1) | JP6465898B2 (ja) |
| KR (1) | KR102121647B1 (ja) |
| CN (1) | CN107208052A (ja) |
| AU (1) | AU2016207513B2 (ja) |
| CA (1) | CA2973187C (ja) |
| SG (1) | SG11201705750WA (ja) |
| WO (1) | WO2016114285A1 (ja) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018030720A1 (ko) * | 2016-08-10 | 2018-02-15 | 가톨릭대학교 산학협력단 | 단백질 리간드를 이용하여 유도만능줄기세포의 분화를 유도하는 각막 줄기세포 유사 세포주를 배양하는 방법 및 시스템 |
| WO2018030719A1 (ko) * | 2016-08-10 | 2018-02-15 | 가톨릭대학교 산학협력단 | 유도만능줄기세포의 분화를 유도하여 각막 상피 세포를 배양하는 방법 및 시스템 |
| WO2018131491A1 (ja) * | 2017-01-13 | 2018-07-19 | 国立大学法人大阪大学 | 角膜上皮細胞集団の製造方法 |
| WO2018143312A1 (ja) * | 2017-01-31 | 2018-08-09 | 国立大学法人大阪大学 | 多能性幹細胞の分化制御方法 |
| WO2018235786A1 (ja) | 2017-06-19 | 2018-12-27 | 国立大学法人大阪大学 | 角膜内皮細胞マーカー及びその利用 |
| WO2019103125A1 (ja) * | 2017-11-24 | 2019-05-31 | 住友化学株式会社 | 神経系細胞又は神経組織と非神経上皮組織とを含む細胞塊の製造方法及びその細胞塊 |
| WO2019189640A1 (ja) | 2018-03-28 | 2019-10-03 | 国立大学法人大阪大学 | 幹細胞由来涙腺組織の作製方法 |
| WO2019225672A1 (ja) | 2018-05-23 | 2019-11-28 | 国立大学法人大阪大学 | 多能性幹細胞由来結膜細胞の誘導方法 |
| WO2021241658A1 (ja) | 2020-05-26 | 2021-12-02 | 株式会社ヘリオス | 低免疫原性細胞 |
| JP2022534332A (ja) * | 2020-04-20 | 2022-07-29 | 青島瑞思徳生物科技有限公司 | 角膜上皮細胞への間葉系幹細胞の分化を誘導する無血清完全培地 |
| WO2023127824A1 (ja) | 2021-12-27 | 2023-07-06 | 住友ファーマ株式会社 | 神経堤細胞の培養方法及び製造方法 |
| WO2023210609A1 (ja) * | 2022-04-27 | 2023-11-02 | ファーマバイオ株式会社 | 細胞シート作製方法 |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2716200T3 (es) | 2011-12-06 | 2019-06-11 | Astellas Inst For Regenerative Medicine | Método de diferenciación dirigida que produce células endoteliales corneales |
| SG11201809235QA (en) * | 2016-04-20 | 2018-11-29 | Kyoto Prefectural Public Univ Corp | Method for producing cultivated epithelial cell sheet |
| WO2019107485A1 (ja) | 2017-11-30 | 2019-06-06 | 国立大学法人京都大学 | 細胞の培養方法 |
| KR102230614B1 (ko) * | 2018-11-13 | 2021-03-22 | 한국화학연구원 | 세포 배양용 캐리어를 이용하여 제조된 오가노이드 및 이를 이용한 약물 독성 평가방법 |
| ES2823398B2 (es) * | 2019-11-05 | 2021-10-15 | Consejo Superior Investigacion | Composición biopolimérica, procedimiento para su preparación y uso de la misma |
| CN113736735B (zh) * | 2020-05-27 | 2024-02-20 | 深圳华大生命科学研究院 | 体外诱导类角膜缘干细胞的方法及试剂盒 |
| CN111733132B (zh) * | 2020-06-28 | 2021-09-21 | 厦门大学 | 一种诱导人胚胎干细胞定向分化为角膜上皮细胞的培养方法及应用 |
| WO2023123299A1 (en) * | 2021-12-31 | 2023-07-06 | Beijing Theraxyte Bioscience Co., Ltd. | Compositions and methods for culturing stem cells |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012073238A1 (en) * | 2010-12-02 | 2012-06-07 | Technion Research & Development Foundation Ltd. | Methods of generating corneal cells and cell populations comprising same |
| WO2012144582A1 (ja) * | 2011-04-20 | 2012-10-26 | 国立大学法人大阪大学 | 角膜上皮分化指向性iPS細胞 |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2360305A1 (en) | 1999-02-09 | 2000-08-17 | Bristol-Myers Squibb Company | Lactam inhibitors of fxa and method |
| CN1929877A (zh) * | 2004-03-11 | 2007-03-14 | 阿如布勒斯特有限公司 | 角膜上皮片及其制作方法、以及使用该片的移植方法 |
| WO2005090557A1 (ja) * | 2004-03-23 | 2005-09-29 | Daiichi Asubio Pharma Co., Ltd. | 多能性幹細胞の増殖方法 |
| WO2009071601A1 (en) | 2007-12-03 | 2009-06-11 | Obe Therapy Biotechnology | Boropeptide inhibitors of enteropeptidase and their uses in treatment of obesity, overweight and/or diseases associated with an abnormal fat metabolism |
| US20120142103A1 (en) * | 2009-05-18 | 2012-06-07 | Kohji Nishida | Method for inducing differentiation into epithelial progenitor cell/stem cell population and corneal epithelial cell population from induced pluripotent stem cells |
| WO2010140698A1 (ja) | 2009-06-03 | 2010-12-09 | 国立大学法人東北大学 | 多能性幹細胞からの神経堤細胞群の分化誘導方法 |
| US9359592B2 (en) | 2009-10-06 | 2016-06-07 | Snu R&Db Foundation | Method for differentiation into retinal cells from stem cells |
| ES2532201T3 (es) | 2009-12-07 | 2015-03-25 | Ajinomoto Co., Inc. | Derivado de éster de ácido heteroarilcarboxílico |
| WO2012169579A1 (ja) | 2011-06-07 | 2012-12-13 | 味の素株式会社 | ヘテロ環カルボン酸エステル誘導体 |
| CA2848561C (en) | 2011-09-15 | 2019-04-02 | Astellas Pharma Inc. | Guanidinobenzoic acid compound |
| CN103184187A (zh) * | 2011-12-28 | 2013-07-03 | 连祺周 | 人诱导性多能干细胞向角膜上皮样细胞定向分化的方法 |
| US9024044B2 (en) | 2012-06-14 | 2015-05-05 | Ajinomoto Co., Inc. | Heteroarylcarboxylic acid ester derivative |
-
2016
- 2016-01-13 CA CA2973187A patent/CA2973187C/en active Active
- 2016-01-13 JP JP2016569469A patent/JP6465898B2/ja active Active
- 2016-01-13 EP EP16737356.2A patent/EP3246394B1/en active Active
- 2016-01-13 US US15/542,200 patent/US11066641B2/en active Active
- 2016-01-13 CN CN201680006004.8A patent/CN107208052A/zh active Pending
- 2016-01-13 AU AU2016207513A patent/AU2016207513B2/en active Active
- 2016-01-13 KR KR1020177019320A patent/KR102121647B1/ko active IP Right Grant
- 2016-01-13 WO PCT/JP2016/050784 patent/WO2016114285A1/ja active Application Filing
- 2016-01-13 SG SG11201705750WA patent/SG11201705750WA/en unknown
-
2021
- 2021-06-15 US US17/348,174 patent/US20210371818A1/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012073238A1 (en) * | 2010-12-02 | 2012-06-07 | Technion Research & Development Foundation Ltd. | Methods of generating corneal cells and cell populations comprising same |
| WO2012144582A1 (ja) * | 2011-04-20 | 2012-10-26 | 国立大学法人大阪大学 | 角膜上皮分化指向性iPS細胞 |
Non-Patent Citations (5)
| Title |
|---|
| H.SUGA ET AL.: "Self-formation of functional adenohypophysis in three-dimensional culture", NATURE, vol. 480, 2011, pages 57 - 62, XP055067739 * |
| R.HAYASHI ET AL.: "Co-ordinated ocular development from human iPS cells and recovery of corneal function", NATURE, vol. 531, March 2016 (2016-03-01), pages 376 - 380, XP055383265 * |
| R.HAYASHI ET AL.: "Generation of Corneal Epithelial Cells from Induced Pluripotent Stem Cells Derived from Human Dermal Fibroblast and Corneal Limbal Epithelium", PLOS ONE, vol. 7, no. Issue 9, e45435, 2012, pages 1 - 10, XP055143151 * |
| See also references of EP3246394A4 * |
| T.D.BLALOCK ET AL.: "Functions of MUC16 in Corneal Epithelial Cells", INVESTIGATIVE OPHTHALMOLOGY & VISUAL SCIENCE, vol. 48, no. 10, 2007, pages 4509 - 4518, XP055464527 * |
Cited By (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018030720A1 (ko) * | 2016-08-10 | 2018-02-15 | 가톨릭대학교 산학협력단 | 단백질 리간드를 이용하여 유도만능줄기세포의 분화를 유도하는 각막 줄기세포 유사 세포주를 배양하는 방법 및 시스템 |
| CN109563485A (zh) * | 2016-08-10 | 2019-04-02 | 加图立大学校产学协力团 | 通过诱导诱导多能干细胞的分化来培养角膜上皮细胞的方法及系统 |
| KR20180017704A (ko) * | 2016-08-10 | 2018-02-21 | 가톨릭대학교 산학협력단 | 단백질 리간드를 이용하여 유도만능줄기세포의 분화를 유도하는 각막 줄기세포 유사 세포주를 배양하는 방법 및 시스템 |
| KR20180017703A (ko) * | 2016-08-10 | 2018-02-21 | 가톨릭대학교 산학협력단 | 유도만능줄기세포의 분화를 유도하여 각막 상피 세포를 배양하는 방법 및 시스템 |
| KR102015815B1 (ko) * | 2016-08-10 | 2019-08-29 | 가톨릭대학교 산학협력단 | 유도만능줄기세포의 분화를 유도하여 각막 상피 세포를 배양하는 방법 및 시스템 |
| KR102029931B1 (ko) * | 2016-08-10 | 2019-10-08 | 가톨릭대학교 산학협력단 | 단백질 리간드를 이용하여 유도만능줄기세포의 분화를 유도하는 각막 줄기세포 유사 세포주를 배양하는 방법 및 시스템 |
| WO2018030719A1 (ko) * | 2016-08-10 | 2018-02-15 | 가톨릭대학교 산학협력단 | 유도만능줄기세포의 분화를 유도하여 각막 상피 세포를 배양하는 방법 및 시스템 |
| WO2018131491A1 (ja) * | 2017-01-13 | 2018-07-19 | 国立大学法人大阪大学 | 角膜上皮細胞集団の製造方法 |
| AU2017393059B2 (en) * | 2017-01-13 | 2021-06-24 | Osaka University | Method for producing corneal epithelial cell mass |
| KR102241349B1 (ko) * | 2017-01-13 | 2021-04-15 | 고꾸리쯔 다이가꾸 호우징 오사까 다이가꾸 | 각막상피세포 집단의 제조 방법 |
| KR20190094244A (ko) * | 2017-01-13 | 2019-08-12 | 고꾸리쯔 다이가꾸 호우징 오사까 다이가꾸 | 각막상피세포 집단의 제조 방법 |
| CN110168076A (zh) * | 2017-01-13 | 2019-08-23 | 国立大学法人大阪大学 | 角膜上皮细胞群的制造方法 |
| EP3569691A4 (en) * | 2017-01-13 | 2019-12-25 | Osaka University | METHOD FOR PRODUCING CORNEAL EPITHELIC CELL MASS |
| JPWO2018131491A1 (ja) * | 2017-01-13 | 2019-11-21 | 国立大学法人大阪大学 | 角膜上皮細胞集団の製造方法 |
| CN110168076B (zh) * | 2017-01-13 | 2023-04-21 | 国立大学法人大阪大学 | 角膜上皮细胞群的制造方法 |
| JPWO2018143312A1 (ja) * | 2017-01-31 | 2019-12-12 | 国立大学法人大阪大学 | 多能性幹細胞の分化制御方法 |
| CN110234755A (zh) * | 2017-01-31 | 2019-09-13 | 国立大学法人大阪大学 | 多能干细胞的分化调控方法 |
| US11649433B2 (en) | 2017-01-31 | 2023-05-16 | Osaka University | Method for controlling differentiation of pluripotent stem cells |
| WO2018143312A1 (ja) * | 2017-01-31 | 2018-08-09 | 国立大学法人大阪大学 | 多能性幹細胞の分化制御方法 |
| CN110234755B (zh) * | 2017-01-31 | 2023-08-29 | 国立大学法人大阪大学 | 多能干细胞的分化调控方法 |
| US11839697B2 (en) | 2017-06-19 | 2023-12-12 | Osaka University | Corneal endothelial cell marker and use thereof |
| KR102343384B1 (ko) * | 2017-06-19 | 2021-12-24 | 오사카 유니버시티 | 각막 내피 세포 마커 및 그 이용 |
| JP2019000069A (ja) * | 2017-06-19 | 2019-01-10 | 国立大学法人大阪大学 | 角膜内皮細胞マーカー及びその利用 |
| KR20200012934A (ko) * | 2017-06-19 | 2020-02-05 | 오사카 유니버시티 | 각막 내피 세포 마커 및 그 이용 |
| WO2018235786A1 (ja) | 2017-06-19 | 2018-12-27 | 国立大学法人大阪大学 | 角膜内皮細胞マーカー及びその利用 |
| JPWO2019103125A1 (ja) * | 2017-11-24 | 2020-11-19 | 住友化学株式会社 | 神経系細胞又は神経組織と非神経上皮組織とを含む細胞塊の製造方法及びその細胞塊 |
| JP7384671B2 (ja) | 2017-11-24 | 2023-11-21 | 住友化学株式会社 | 神経系細胞又は神経組織と非神経上皮組織とを含む細胞塊の製造方法及びその細胞塊 |
| WO2019103125A1 (ja) * | 2017-11-24 | 2019-05-31 | 住友化学株式会社 | 神経系細胞又は神経組織と非神経上皮組織とを含む細胞塊の製造方法及びその細胞塊 |
| JPWO2019189640A1 (ja) * | 2018-03-28 | 2021-02-25 | 国立大学法人大阪大学 | 幹細胞由来涙腺組織の作製方法 |
| JP7016185B2 (ja) | 2018-03-28 | 2022-02-04 | 国立大学法人大阪大学 | 幹細胞由来涙腺組織の作製方法 |
| WO2019189640A1 (ja) | 2018-03-28 | 2019-10-03 | 国立大学法人大阪大学 | 幹細胞由来涙腺組織の作製方法 |
| CN112189049A (zh) * | 2018-05-23 | 2021-01-05 | 国立大学法人大阪大学 | 来源于多能干细胞的结膜细胞的诱导方法 |
| JP7107595B2 (ja) | 2018-05-23 | 2022-07-27 | 国立大学法人大阪大学 | 多能性幹細胞由来結膜細胞の誘導方法 |
| US20210207087A1 (en) * | 2018-05-23 | 2021-07-08 | Osaka University | Method for inducing differentiation into pluripotent stem cell-derived conjunctival cells |
| JPWO2019225672A1 (ja) * | 2018-05-23 | 2021-05-13 | 国立大学法人大阪大学 | 多能性幹細胞由来結膜細胞の誘導方法 |
| KR20210015789A (ko) | 2018-05-23 | 2021-02-10 | 고꾸리쯔 다이가꾸 호우징 오사까 다이가꾸 | 다능성 줄기세포 유래 결막세포의 유도 방법 |
| WO2019225672A1 (ja) | 2018-05-23 | 2019-11-28 | 国立大学法人大阪大学 | 多能性幹細胞由来結膜細胞の誘導方法 |
| CN112189049B (zh) * | 2018-05-23 | 2023-12-15 | 国立大学法人大阪大学 | 来源于多能干细胞的结膜细胞的诱导方法 |
| JP2022534332A (ja) * | 2020-04-20 | 2022-07-29 | 青島瑞思徳生物科技有限公司 | 角膜上皮細胞への間葉系幹細胞の分化を誘導する無血清完全培地 |
| JP7239686B2 (ja) | 2020-04-20 | 2023-03-14 | 青島瑞思徳生物科技有限公司 | 角膜上皮細胞への間葉系幹細胞の分化を誘導する無血清完全培地 |
| WO2021241658A1 (ja) | 2020-05-26 | 2021-12-02 | 株式会社ヘリオス | 低免疫原性細胞 |
| WO2023127824A1 (ja) | 2021-12-27 | 2023-07-06 | 住友ファーマ株式会社 | 神経堤細胞の培養方法及び製造方法 |
| KR20240125943A (ko) | 2021-12-27 | 2024-08-20 | 스미토모 파마 가부시키가이샤 | 신경능 세포의 배양 방법 및 제조 방법 |
| WO2023210609A1 (ja) * | 2022-04-27 | 2023-11-02 | ファーマバイオ株式会社 | 細胞シート作製方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| US11066641B2 (en) | 2021-07-20 |
| EP3246394B1 (en) | 2022-11-23 |
| KR102121647B1 (ko) | 2020-06-10 |
| US20210371818A1 (en) | 2021-12-02 |
| KR20170092694A (ko) | 2017-08-11 |
| EP3246394A4 (en) | 2018-07-18 |
| CA2973187C (en) | 2021-11-02 |
| JP6465898B2 (ja) | 2019-02-06 |
| CA2973187A1 (en) | 2016-07-21 |
| SG11201705750WA (en) | 2017-08-30 |
| AU2016207513B2 (en) | 2019-03-28 |
| US20180010093A1 (en) | 2018-01-11 |
| JPWO2016114285A1 (ja) | 2017-09-14 |
| EP3246394A1 (en) | 2017-11-22 |
| CN107208052A (zh) | 2017-09-26 |
| AU2016207513A1 (en) | 2017-07-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6465898B2 (ja) | 多能性幹細胞からの角膜上皮細胞の分化誘導 | |
| JP6991517B2 (ja) | 毛様体周縁部様構造体の製造法 | |
| US11542471B2 (en) | Method for producing ciliary marginal zone-like structure | |
| CN105940101A (zh) | 由人多能干细胞特化功能性颅基板衍生物 | |
| KR102297580B1 (ko) | 전안부 조직의 제조 방법 | |
| JP7016185B2 (ja) | 幹細胞由来涙腺組織の作製方法 | |
| JPWO2019225672A1 (ja) | 多能性幹細胞由来結膜細胞の誘導方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16737356 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2016569469 Country of ref document: JP Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2973187 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15542200 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20177019320 Country of ref document: KR Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2016737356 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11201705750W Country of ref document: SG |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2016207513 Country of ref document: AU Date of ref document: 20160113 Kind code of ref document: A |